User login
Eyes on ESC ‘21: Hope for EMPEROR-Preserved, guidelines remade
There will be so much more to the annual congress of the European Society of Cardiology, which begins Aug. 27 with an all-virtual format, than detailed primary results of EMPEROR-Preserved, a trial that could mark a turning point for heart failure (HF) medical therapy.
Also among the featured Hot Line and Late-Breaking Science sessions are – along with many other studies – explorations of arrhythmia management (ablation or guided by loop recorder); secondary prevention, including by vaccination; oral anticoagulation, notably after transcatheter valve procedures; and colchicine or thrombosis prophylaxis in hospitalized patients with COVID-19.
There will even be a head-to-head comparison of two long-familiar left atrial appendage (LAA) occluders, and a population-based, randomized trial of sodium restriction through wide-scale use of a potassium-based salt substitute.
The congress will also introduce four guideline documents at sessions throughout the Congress, one on each day. They cover new and modified recommendations for heart failure; pacing, including cardiac resynchronization therapy (CRT); cardiovascular (CV) disease prevention; and, with cosponsorship from the European Association for Cardio-Thoracic Surgery, valvular heart disease.
The virtues of virtual
That next year’s Congress is slated for Aug. 27-30 in Barcelona should be welcome news for anyone whose “what if” curiosity about all-virtual conferences has already been satisfied. But with experience comes wisdom, as the medical societies have learned that online scientific meetings have some winning qualities that may be worth keeping, as least for a while.
“I think there is no doubt that the digital format will continue, for several reasons. One is that this pandemic is not over,” ESC Congress program committee chair Stephan Windecker, MD, Bern (Switzerland) University Hospital, , told this news organization. “As long as it is not over, the digital format is here to stay.”
But it also appears that people who haven’t been able to attend the congress in person are keen to log in and engage online, Dr. Windecker said. The 2020 all-virtual conference drew a much younger pool of registrants, on average, than did the live conferences before the pandemic.
“I think that’s an indication of people that may be in training, in early stages of their career, or they don’t have the support from departments or from their practice, or other financial means.” But they are able to participate via computer, tablet, or smartphone, he said.
“Another advantage is that the recorded content can be replayed at the convenience of whoever wants to consume it at a later point in time,” he added. “Those are just some examples why the digital format is likely to stay,” on its own or in a new age of hybrid meetings.
New and updated guidelines
Leading off the guideline series is the document on diagnosis and treatment of acute and chronic HF, which leveraged the past few busy years of HF clinical trials to arrive at a number of new recommendations and strengthened level-of-evidence ratings. It covers both drug and device therapy of HF with reduced ejection fraction (HFrEF) and acute decompensated HF, and tweaks and further enshrines the concept of HF with mildly reduced ejection fraction (HFmrEF).
Several updated recommendations for both long-used and novel medications, notably the sodium-glucose cotransporter 2 inhibitors, will be included because of the recently appreciated evidence-based impact in HFrEF, Dr. Windecker noted.
“I think it will be particularly interesting to look for the SGLT2 inhibitors as not a completely new class of drugs, but certainly one where there has been a lot of new evidence, to look at how those drugs will be integrated in the overall care pathway.”
A top-line preview of the new HF guideline limited to drug therapy, presented at July’s Heart Failure Association of the European Society of Cardiology (ESC-HFA), provided a simple answer to a common question in the new, bountiful age of HFrEF medications: Which meds, initiated in what order?
As it happens, the new recommendation for first-line HFrEF drug therapy is not a silver bullet, but a shotgun – prompt initiation of at least four meds, one from each of four drug classes: renin-angiotensin system inhibitors, beta-blockers, mineralocorticoid receptor antagonists (MRA), and SGLT2 inhibitors. Each class, as described in the document, is to be started as soon as safely feasible, in a sequence deemed appropriate for each individual patient.
Spotlight on EMPEROR-Preserved
The world already knows that the trial, which tested the SGLT2 inhibitor empagliflozin (Jardiance, Boehringer Ingelheim/Eli Lilly) on top of standard therapy, “met” its primary endpoint in almost 6,000 patients with HF with preserved ejection fraction (HFpEF), who included some with HFmrEF by more contemporary definitions.
That means patients in EMPEROR-Preserved assigned to take empagliflozin showed significantly fewer events that made up the study’s primary endpoint, a composite of CV death or HF hospitalization. It appears to be the first clearly significant overall medical therapy benefit for a clinical primary endpoint in a major randomized HFpEF drug trial.
And that, pending fuller presentation of trial results at the Congress on Aug. 27, could be a huge deal for the half of HF patients with left ventricular ejection fractions (LVEF) higher than the HFrEF range.
Those early top-line results weren’t a decisive bombshell for a field now filled with hope for a practice-changing empagliflozin outcome in EMPEROR-Preserved, which isn’t a certainty. They were more like the “boom” of a mortar launching a rocket of fireworks that may explode into a chrysanthemum or green comet or, sometimes, turn out to be no more than a dud. The promise of the early cursory results critically depends on further details.
“Provided there is a compelling benefit, this is what everyone has been waiting for in this condition for decades,” Mikhail N. Kosiborod, MD, director of cardiometabolic research at Saint Luke’s Mid-America Heart Institute, Kansas City, Mo., said.
“Already knowing that the trial met the primary endpoint is obviously very intriguing and encouraging,” he added. “But there are things we don’t know, such as: What is the magnitude of benefit? And whether that benefit, whatever the magnitude, is driven by reductions in both heart failure hospitalizations and cardiovascular death, or only one of the two.”
For example: “If we see an impressive benefit for reduction of hospitalizations, but not a significant reduction in death, that would still be a huge advance. That’s because, to date, we don’t have any drug for HFpEF that has convincingly demonstrated a compelling reduction in heart failure hospitalization or improvement in symptoms, function, or quality of life,” observed Dr. Kosiborod, who wasn’t part of EMPEROR-Preserved.
There have been “suggestions” from HFrEF trials that empagliflozin and dapagliflozin (Farxiga, AstraZeneca) “have very comparable effects on at least the endpoint of cardiovascular death or hospitalization for heart failure,” he said. “So, my expectation would be that whatever is observed in EMPEROR-Preserved is likely a class effect, as well.”
Following EMPEROR-Preserved on the agenda is EMPEROR-Pooled, a patient-level combined analysis of the EMPEROR series of trials that spans the range of HF, regardless of ejection fraction or diabetes status, primarily exploring the effects of empagliflozin on renal function.
Other offerings, Friday, Aug. 27
Scheduled immediately after EMPEROR-Preserved is a presentation on the SMART-MI trial, which should clarify whether management guided by continuous ambulatory monitoring is effective in patients considered at especially high arrhythmic risk. Entry called for recent myocardial infarction and an LVEF of 36%-50% with evidence of cardiac autonomic dysfunction.
The trial randomly assigned 400 such patients to be or not be implanted with a Reveal LINQ (Medtronic) loop recorder and followed them for up to 18 months, primarily for detection of potentially serious arrhythmic events. Endpoints that involved mortality, hospitalization or other clinical events were secondary.
In a time slot preceding both SMART-MI and EMPEROR-Preserved, the GUIDE-HF trial is following a projected 3,600 patients with HF implanted with a CardioMEMS HF System (Abbott) pulmonary artery (PA) pressure sensor to explore the its value for guiding management.
The trial’s three cohorts, followed for at least 12 months, include randomized sensor-monitored and control groups of patients with New York Heart Association class 2-4 symptoms, as well as a third observational set of patients in NYHA class 3. That’s the indication for which the CardioMEMS monitor gained approval in the United States in 2014 based on the 2011 CHAMPION trial, and which fared just as well in the 2017 CHAMPION Post-Approval Study.
The Friday Hot Lines also include Dal-GenE, which has entered about 6,000 patients with recent MI to test the once-abandoned cholesterol ester transfer protein (CETP) inhibitor dalcetrapib (DalCor) for any secondary-prevention benefits when used selectively. The trial’s hook: All its patients are confirmed to have the AA genotype of the rs1967309 variant in the ADCY9 gene, which has been associated with a pronounced clinical response to CETP inhibition.
Saturday, Aug. 28
The direct oral anticoagulants (DOACs) have largely replaced vitamin K antagonists in patients with nonvalvular atrial fibrillation (AFib). But whether DOACs are similarly preferable in the growing world population of people who have undergone transcatheter aortic valve replacement (TAVR or TAVI), an issue explored with variable results in the ATLANTIS and GALILEO trials, is far from settled.
The ENVISAGE-TAVI AF trial explored the question for the factor X inhibitor edoxaban (Savaysa, Lixiana, Daiichi-Sankyo) in 1,400 patients with AFib and a transfemoral TAVR in the previous 5 days, who were randomly assigned to the DOAC or standard management along with discretionary antiplatelet therapy. They’ve been followed for up to 3 years for a composite endpoint of clinical events – including death, MI, and stroke – and for major bleeding.
The day will also feature MASTER DAPT, a comparison of two dual-antiplatelet therapy (DAPT) regimens in an estimated 4,300 patients considered to be high-risk for bleeding who had received the sirolimus-eluting Ultimaster (Terumo) coronary stent, which has a bioresorbable polymer coating.
Investigators have randomly assigned patients to receive either very-short-duration DAPT, for about a month after stenting, followed by a P2Y12 inhibitor alone for up to a year after the procedure; or a more conventional regimen of a P2Y12 inhibitor for 6-12 months with aspirin maintained for a total of 12 months.
Later that day, investigators from the FIGARO-DKD trial will present their results based on 7,437 patients with type 2 diabetes and chronic kidney disease (CKD), a much fuller version than the top-line findings announced by sponsor Bayer 3 months ago.
Those top-line results suggested that patients assigned to receive the nonsteroidal nonselective mineralocorticoid receptor antagonist (MRA) finerenone (Kerendia) on top of standard care benefited with a drop in risk for the primary endpoint of CV death or nonfatal CV events.
Finerenone was recently approved in the United States for treating patients with both type 2 diabetes and CKD based on the published FIDELIO-DKD trial, which had seen less CKD progression and fewer CV events in such patients who took the novel MRA.
Although similar in design to FIGARO-DKD, FIDELIO-DKD had entered fewer patients with early-stage diabetic kidney disease (DKD). That led researchers to pool the two trials’ populations to create a cohort that spans the spectrum of DKD severity. An analysis of the pooled cohort, dubbed FIDELITY, is on the schedule after FIGARO-DKD.
After FIDELITY is the prospective APAF-CRT trial that is following a projected 1,830 patients with permanent, symptomatic AFib and a recent hospitalization for AFib or HF and who were not good candidates for standard ablation. They were assigned to receive either atrioventricular junctional ablation followed by CRT, with or without a defibrillation, on top of optimal meds – a so-called “ablate-and-pace” strategy – or an implantable cardioverter defibrillator with rate-control drug therapy.
The new analysis represents the trial’s second phase in which mortality was followed for 4 years as the primary endpoint, in contrast to the previously reported initial phase that followed the first 102 patients for 2 years for the composite primary endpoint of death, worsening HF, and HF hospitalization. The first phase had halted enrollment before reaching its planned target of 280 patients after an interim analysis showed a significant benefit for ablate and pace.
Next up: DECAAF 2, a randomized assessment of whether catheter ablation for AFib guided by delayed gadolinium enhancement on MRI, a proxy for scar tissue, can be more effective than standard AFib ablation by pulmonary vein isolation alone. An estimated 900 patients with persistent AFib who had never before undergone ablation for the arrhythmia were randomly assigned to one strategy or the other and followed for AFib recurrence over 18 months.
Sunday, Aug. 29
The TOMAHAWK trial aimed to clarify the optimal timing of invasive coronary angiography for resuscitated patients with non–ST-segment elevation out-of-hospital cardiac arrest, a broad population in a setting for which there is little randomized-trial guidance. Investigators randomly assigned 558 such patients to undergo immediate invasive angiography or to direct intensive care unit admission for initial standard care with discretionary delayed angiography. Patients were followed for all-cause mortality, with other clinical events and neurologic outcomes as secondary endpoints.
Next on the schedule, the RIPCORD-2 trial randomly assigned 1,100 patients with stable known or suspected coronary artery disease (CAD) to undergo conventional angiography alone or with added direct pressure-wire measurement of fractional flow reserve to guide management decisions. Primary outcomes include health care costs and patient-reported quality of life at 1 year.
Slated for later that day, the Asymptomatic Carotid Surgery Trial-2 (ACST-2) has entered an estimated 3600 patients with a substantial carotid artery narrowing not associated with symptoms but for which either carotid endarterectomy (CEA) or carotid artery stenting (CAS) was considered anatomically feasible. There also must have been “substantial uncertainty” regarding the optimal procedure choice.
The trial, conducted in 40 countries primarily in Europe and North America and launched in 2008, randomly assigned the patients to undergo either CEA or CAS, in both cases with appropriate medical therapy, and followed them for periprocedural events and up to 10 years for strokes and stroke-related events.
The LOOP study, which is to directly follow ACST-2, has explored whether screening for AFib using the Medtronic Reveal LINQ monitor in older patients with non-AFib stroke risk factors – with oral anticoagulation prescribed for those who test positive – can lower their risk for stroke or systemic embolism. It randomly assigned 6,000 such patients to care guided by the loop recorder or to standard care.
On a somewhat larger scale, the Salt Substitute and Stroke Study (SSaSS) randomly assigned a total of 20,996 people in about 600 villages across northern China and Tibet to sodium-restriction intervention and control groups by village. All participants had a history of stroke or were aged at least 60 years with uncontrolled hypertension.
As described by the trial’s online portal, participants in villages assigned to the intervention group were given a supply of a low-sodium, potassium-supplementing salt substitute to replace their own salt supplies, along with education on the health benefits of sodium restriction. Participants in control villages continued their normal diets and, at the trial’s beginning, received “advice to reduce their salt intake.” All were required to own a telephone.
Clinical events, including strokes and hospitalizations throughout a 5-year follow-up, were tracked by phone calls made to all participants every 6 months and were documented at follow-up home visits.
Sunday is also to feature a Late-Breaking Trials session with a focus on COVID-19, which leads off with COLCOVID, a test of colchicine in patients hospitalized for suspected SARS-CoV-2 infection and in acute respiratory distress.
The 1,279 participants in Argentina were randomly assigned to receive or not receive the potent anti-inflammatory agent on top of antivirals and other standard management and followed for death or new need for mechanical ventilation. A successful outcome would contrast with the RECOVERY trial, which terminated a colchicine group of patients hospitalized with COVID-19 because of a lack of efficacy earlier this year.
COLCOVID is to be followed by the MICHELLE trial of rivaroxaban (Xarelto, Bayer/Janssen) prophylaxis, compared with no preventive oral anticoagulant, in 320 patients who, when hospitalized with COVID-19, had been on parenteral anticoagulants because of an elevated risk for venous thromboembolism. The trial, conducted in Brazil, called for postdischarge rivaroxaban at a once-daily dosage of 10 mg for about 1 month.
The session also includes a presentation called “Insights into the Effects of the COVID-19 Pandemic: Comprehensive Analysis from the GUIDE-HF Trial,” the primary outcomes of which will be reported on the first day of the Congress.
Following is a presentation on the PREPARE-IT study of icosapent ethyl (Vascepa, Amarin), given at high dosages intended to be anti-inflammatory, compared with placebo, in an estimated 4,000 adults. The trial has two groups: A prevention group of adults living and circulating in the community; and a treatment group of patients aged at least 40 years with confirmed symptomatic SARS-CoV-2 infection for whom the need for hospitalization isn’t clear.
Monday, Aug. 30
The final day of the Congress features a trial called Influenza Vaccination after Myocardial Infarction (IAMI), which has tested the secondary preventive effect of influenza vaccination by randomly assigning 2,571 patients to receive a standard vaccine or a saline placebo injection on one occasion.
Entry to the international trial called for a diagnosis of MI with or without ST-segment elevation, or stable CAD and age at least 75 years with other risk factors. The patients were followed for death, MI, stent thrombosis, and a slew of secondary endpoints over 12 months.
Monday offerings continue later in a time block leading off with the STEP trial, which has randomly assigned an estimated 8,000 patients at 40 centers in China who are 60 to 80 years of age with a systolic blood pressure of 140 to <190 mm Hg to be on standard guideline-based therapy or an intensive drug-management strategy.
The systolic BP goals are 130 to <150 mm Hg for standard care and 110 to <130 mm Hg for the intensive regimen. The composite primary endpoint includes death and clinical events related to acute coronary syndromes, HF, revascularization, and stroke.
Following on heels of STEP, the Amulet IDE trial – the first major randomized comparison of two transcatheter LAA closure devices – entered 1,878 patients with nonvalvular AFib who were considered high-risk for bleeding and stroke or systemic embolism.
They were randomly assigned in the noninferiority trial to receive either the AMPLATZER Amulet (Abbott Medical Devices) or the WATCHMAN (Boston Scientific) closure devices and were followed for safety and efficacy for up to 5 years.
Both LAA closure devices, intended to make patients with AFib less reliant on oral anticoagulation, are now available on both sides of the Atlantic – as well as many other countries – after the Amulet’s United States market approval on Aug. 16, based largely on the Amulet IDE trial.
Rounding out the final Hot Line set is one of the latest efforts to show the efficacy and safety of a very short DAPT period after coronary stenting in patients with acute coronary syndromes, the STOPDAPT-2 ACS trial.
The study assigned 3,008 patients in Japan to receive aspirin and clopidogrel for either 1 month or 1 year after implantation with an everolimus-eluting cobalt-chromium stent and followed them for up to 5 years for a composite of MI, CV death, stent thrombosis, stroke, and bleeding.
The trial follows the published STOPDAPT-2 trial that showed superiority for the 1-month DAPT regimen in a predominantly stable-CAD population treated with the same kind of stent.
Program structure and format
A total of 15 online channels are to be available in the morning, European time, their schedules running in parallel. Presentations often are prerecorded, but also include live sessions at 8:00 a.m. Central time and 12 p.m. CET (2:00 a.m. and 6:00 a.m. Eastern time) to liven up the channel offerings, Dr. Windecker observed, and to make them more immediate and potentially interactive.
Many of the parallel channels are devoted throughout the Congress to particular silos of cardiology; for example, arrhythmias and device therapy is on channel 3; CAD and acute care is on 5; HF is on 6; and preventive cardiology is on 9.
Other channels swing across different topics from day to day, such as channel 1, which covers COVID-19 topics on the first and third day of the meeting, “advances in science” on day 2, and “digital health, public health, health economics” on day 4.
The focus each day, starting at 2:00 p.m. CET (8:00 a.m. ET) and continuing into the evening in Europe, shifts over to the Prime Time live program, which features the Hot Line and guideline presentations and many of the live abstract presentations.
Dr. Kosiborod, not a researcher with the EMPEROR trials, is chair of the Dapagliflozin in Preserved Ejection Fraction Heart Failure ( PRESERVED-HF ) trial, which is scheduled for presentation at the September 2021 Heart Failure Society of American meeting.
A version of this article first appeared on Medscape.com.
There will be so much more to the annual congress of the European Society of Cardiology, which begins Aug. 27 with an all-virtual format, than detailed primary results of EMPEROR-Preserved, a trial that could mark a turning point for heart failure (HF) medical therapy.
Also among the featured Hot Line and Late-Breaking Science sessions are – along with many other studies – explorations of arrhythmia management (ablation or guided by loop recorder); secondary prevention, including by vaccination; oral anticoagulation, notably after transcatheter valve procedures; and colchicine or thrombosis prophylaxis in hospitalized patients with COVID-19.
There will even be a head-to-head comparison of two long-familiar left atrial appendage (LAA) occluders, and a population-based, randomized trial of sodium restriction through wide-scale use of a potassium-based salt substitute.
The congress will also introduce four guideline documents at sessions throughout the Congress, one on each day. They cover new and modified recommendations for heart failure; pacing, including cardiac resynchronization therapy (CRT); cardiovascular (CV) disease prevention; and, with cosponsorship from the European Association for Cardio-Thoracic Surgery, valvular heart disease.
The virtues of virtual
That next year’s Congress is slated for Aug. 27-30 in Barcelona should be welcome news for anyone whose “what if” curiosity about all-virtual conferences has already been satisfied. But with experience comes wisdom, as the medical societies have learned that online scientific meetings have some winning qualities that may be worth keeping, as least for a while.
“I think there is no doubt that the digital format will continue, for several reasons. One is that this pandemic is not over,” ESC Congress program committee chair Stephan Windecker, MD, Bern (Switzerland) University Hospital, , told this news organization. “As long as it is not over, the digital format is here to stay.”
But it also appears that people who haven’t been able to attend the congress in person are keen to log in and engage online, Dr. Windecker said. The 2020 all-virtual conference drew a much younger pool of registrants, on average, than did the live conferences before the pandemic.
“I think that’s an indication of people that may be in training, in early stages of their career, or they don’t have the support from departments or from their practice, or other financial means.” But they are able to participate via computer, tablet, or smartphone, he said.
“Another advantage is that the recorded content can be replayed at the convenience of whoever wants to consume it at a later point in time,” he added. “Those are just some examples why the digital format is likely to stay,” on its own or in a new age of hybrid meetings.
New and updated guidelines
Leading off the guideline series is the document on diagnosis and treatment of acute and chronic HF, which leveraged the past few busy years of HF clinical trials to arrive at a number of new recommendations and strengthened level-of-evidence ratings. It covers both drug and device therapy of HF with reduced ejection fraction (HFrEF) and acute decompensated HF, and tweaks and further enshrines the concept of HF with mildly reduced ejection fraction (HFmrEF).
Several updated recommendations for both long-used and novel medications, notably the sodium-glucose cotransporter 2 inhibitors, will be included because of the recently appreciated evidence-based impact in HFrEF, Dr. Windecker noted.
“I think it will be particularly interesting to look for the SGLT2 inhibitors as not a completely new class of drugs, but certainly one where there has been a lot of new evidence, to look at how those drugs will be integrated in the overall care pathway.”
A top-line preview of the new HF guideline limited to drug therapy, presented at July’s Heart Failure Association of the European Society of Cardiology (ESC-HFA), provided a simple answer to a common question in the new, bountiful age of HFrEF medications: Which meds, initiated in what order?
As it happens, the new recommendation for first-line HFrEF drug therapy is not a silver bullet, but a shotgun – prompt initiation of at least four meds, one from each of four drug classes: renin-angiotensin system inhibitors, beta-blockers, mineralocorticoid receptor antagonists (MRA), and SGLT2 inhibitors. Each class, as described in the document, is to be started as soon as safely feasible, in a sequence deemed appropriate for each individual patient.
Spotlight on EMPEROR-Preserved
The world already knows that the trial, which tested the SGLT2 inhibitor empagliflozin (Jardiance, Boehringer Ingelheim/Eli Lilly) on top of standard therapy, “met” its primary endpoint in almost 6,000 patients with HF with preserved ejection fraction (HFpEF), who included some with HFmrEF by more contemporary definitions.
That means patients in EMPEROR-Preserved assigned to take empagliflozin showed significantly fewer events that made up the study’s primary endpoint, a composite of CV death or HF hospitalization. It appears to be the first clearly significant overall medical therapy benefit for a clinical primary endpoint in a major randomized HFpEF drug trial.
And that, pending fuller presentation of trial results at the Congress on Aug. 27, could be a huge deal for the half of HF patients with left ventricular ejection fractions (LVEF) higher than the HFrEF range.
Those early top-line results weren’t a decisive bombshell for a field now filled with hope for a practice-changing empagliflozin outcome in EMPEROR-Preserved, which isn’t a certainty. They were more like the “boom” of a mortar launching a rocket of fireworks that may explode into a chrysanthemum or green comet or, sometimes, turn out to be no more than a dud. The promise of the early cursory results critically depends on further details.
“Provided there is a compelling benefit, this is what everyone has been waiting for in this condition for decades,” Mikhail N. Kosiborod, MD, director of cardiometabolic research at Saint Luke’s Mid-America Heart Institute, Kansas City, Mo., said.
“Already knowing that the trial met the primary endpoint is obviously very intriguing and encouraging,” he added. “But there are things we don’t know, such as: What is the magnitude of benefit? And whether that benefit, whatever the magnitude, is driven by reductions in both heart failure hospitalizations and cardiovascular death, or only one of the two.”
For example: “If we see an impressive benefit for reduction of hospitalizations, but not a significant reduction in death, that would still be a huge advance. That’s because, to date, we don’t have any drug for HFpEF that has convincingly demonstrated a compelling reduction in heart failure hospitalization or improvement in symptoms, function, or quality of life,” observed Dr. Kosiborod, who wasn’t part of EMPEROR-Preserved.
There have been “suggestions” from HFrEF trials that empagliflozin and dapagliflozin (Farxiga, AstraZeneca) “have very comparable effects on at least the endpoint of cardiovascular death or hospitalization for heart failure,” he said. “So, my expectation would be that whatever is observed in EMPEROR-Preserved is likely a class effect, as well.”
Following EMPEROR-Preserved on the agenda is EMPEROR-Pooled, a patient-level combined analysis of the EMPEROR series of trials that spans the range of HF, regardless of ejection fraction or diabetes status, primarily exploring the effects of empagliflozin on renal function.
Other offerings, Friday, Aug. 27
Scheduled immediately after EMPEROR-Preserved is a presentation on the SMART-MI trial, which should clarify whether management guided by continuous ambulatory monitoring is effective in patients considered at especially high arrhythmic risk. Entry called for recent myocardial infarction and an LVEF of 36%-50% with evidence of cardiac autonomic dysfunction.
The trial randomly assigned 400 such patients to be or not be implanted with a Reveal LINQ (Medtronic) loop recorder and followed them for up to 18 months, primarily for detection of potentially serious arrhythmic events. Endpoints that involved mortality, hospitalization or other clinical events were secondary.
In a time slot preceding both SMART-MI and EMPEROR-Preserved, the GUIDE-HF trial is following a projected 3,600 patients with HF implanted with a CardioMEMS HF System (Abbott) pulmonary artery (PA) pressure sensor to explore the its value for guiding management.
The trial’s three cohorts, followed for at least 12 months, include randomized sensor-monitored and control groups of patients with New York Heart Association class 2-4 symptoms, as well as a third observational set of patients in NYHA class 3. That’s the indication for which the CardioMEMS monitor gained approval in the United States in 2014 based on the 2011 CHAMPION trial, and which fared just as well in the 2017 CHAMPION Post-Approval Study.
The Friday Hot Lines also include Dal-GenE, which has entered about 6,000 patients with recent MI to test the once-abandoned cholesterol ester transfer protein (CETP) inhibitor dalcetrapib (DalCor) for any secondary-prevention benefits when used selectively. The trial’s hook: All its patients are confirmed to have the AA genotype of the rs1967309 variant in the ADCY9 gene, which has been associated with a pronounced clinical response to CETP inhibition.
Saturday, Aug. 28
The direct oral anticoagulants (DOACs) have largely replaced vitamin K antagonists in patients with nonvalvular atrial fibrillation (AFib). But whether DOACs are similarly preferable in the growing world population of people who have undergone transcatheter aortic valve replacement (TAVR or TAVI), an issue explored with variable results in the ATLANTIS and GALILEO trials, is far from settled.
The ENVISAGE-TAVI AF trial explored the question for the factor X inhibitor edoxaban (Savaysa, Lixiana, Daiichi-Sankyo) in 1,400 patients with AFib and a transfemoral TAVR in the previous 5 days, who were randomly assigned to the DOAC or standard management along with discretionary antiplatelet therapy. They’ve been followed for up to 3 years for a composite endpoint of clinical events – including death, MI, and stroke – and for major bleeding.
The day will also feature MASTER DAPT, a comparison of two dual-antiplatelet therapy (DAPT) regimens in an estimated 4,300 patients considered to be high-risk for bleeding who had received the sirolimus-eluting Ultimaster (Terumo) coronary stent, which has a bioresorbable polymer coating.
Investigators have randomly assigned patients to receive either very-short-duration DAPT, for about a month after stenting, followed by a P2Y12 inhibitor alone for up to a year after the procedure; or a more conventional regimen of a P2Y12 inhibitor for 6-12 months with aspirin maintained for a total of 12 months.
Later that day, investigators from the FIGARO-DKD trial will present their results based on 7,437 patients with type 2 diabetes and chronic kidney disease (CKD), a much fuller version than the top-line findings announced by sponsor Bayer 3 months ago.
Those top-line results suggested that patients assigned to receive the nonsteroidal nonselective mineralocorticoid receptor antagonist (MRA) finerenone (Kerendia) on top of standard care benefited with a drop in risk for the primary endpoint of CV death or nonfatal CV events.
Finerenone was recently approved in the United States for treating patients with both type 2 diabetes and CKD based on the published FIDELIO-DKD trial, which had seen less CKD progression and fewer CV events in such patients who took the novel MRA.
Although similar in design to FIGARO-DKD, FIDELIO-DKD had entered fewer patients with early-stage diabetic kidney disease (DKD). That led researchers to pool the two trials’ populations to create a cohort that spans the spectrum of DKD severity. An analysis of the pooled cohort, dubbed FIDELITY, is on the schedule after FIGARO-DKD.
After FIDELITY is the prospective APAF-CRT trial that is following a projected 1,830 patients with permanent, symptomatic AFib and a recent hospitalization for AFib or HF and who were not good candidates for standard ablation. They were assigned to receive either atrioventricular junctional ablation followed by CRT, with or without a defibrillation, on top of optimal meds – a so-called “ablate-and-pace” strategy – or an implantable cardioverter defibrillator with rate-control drug therapy.
The new analysis represents the trial’s second phase in which mortality was followed for 4 years as the primary endpoint, in contrast to the previously reported initial phase that followed the first 102 patients for 2 years for the composite primary endpoint of death, worsening HF, and HF hospitalization. The first phase had halted enrollment before reaching its planned target of 280 patients after an interim analysis showed a significant benefit for ablate and pace.
Next up: DECAAF 2, a randomized assessment of whether catheter ablation for AFib guided by delayed gadolinium enhancement on MRI, a proxy for scar tissue, can be more effective than standard AFib ablation by pulmonary vein isolation alone. An estimated 900 patients with persistent AFib who had never before undergone ablation for the arrhythmia were randomly assigned to one strategy or the other and followed for AFib recurrence over 18 months.
Sunday, Aug. 29
The TOMAHAWK trial aimed to clarify the optimal timing of invasive coronary angiography for resuscitated patients with non–ST-segment elevation out-of-hospital cardiac arrest, a broad population in a setting for which there is little randomized-trial guidance. Investigators randomly assigned 558 such patients to undergo immediate invasive angiography or to direct intensive care unit admission for initial standard care with discretionary delayed angiography. Patients were followed for all-cause mortality, with other clinical events and neurologic outcomes as secondary endpoints.
Next on the schedule, the RIPCORD-2 trial randomly assigned 1,100 patients with stable known or suspected coronary artery disease (CAD) to undergo conventional angiography alone or with added direct pressure-wire measurement of fractional flow reserve to guide management decisions. Primary outcomes include health care costs and patient-reported quality of life at 1 year.
Slated for later that day, the Asymptomatic Carotid Surgery Trial-2 (ACST-2) has entered an estimated 3600 patients with a substantial carotid artery narrowing not associated with symptoms but for which either carotid endarterectomy (CEA) or carotid artery stenting (CAS) was considered anatomically feasible. There also must have been “substantial uncertainty” regarding the optimal procedure choice.
The trial, conducted in 40 countries primarily in Europe and North America and launched in 2008, randomly assigned the patients to undergo either CEA or CAS, in both cases with appropriate medical therapy, and followed them for periprocedural events and up to 10 years for strokes and stroke-related events.
The LOOP study, which is to directly follow ACST-2, has explored whether screening for AFib using the Medtronic Reveal LINQ monitor in older patients with non-AFib stroke risk factors – with oral anticoagulation prescribed for those who test positive – can lower their risk for stroke or systemic embolism. It randomly assigned 6,000 such patients to care guided by the loop recorder or to standard care.
On a somewhat larger scale, the Salt Substitute and Stroke Study (SSaSS) randomly assigned a total of 20,996 people in about 600 villages across northern China and Tibet to sodium-restriction intervention and control groups by village. All participants had a history of stroke or were aged at least 60 years with uncontrolled hypertension.
As described by the trial’s online portal, participants in villages assigned to the intervention group were given a supply of a low-sodium, potassium-supplementing salt substitute to replace their own salt supplies, along with education on the health benefits of sodium restriction. Participants in control villages continued their normal diets and, at the trial’s beginning, received “advice to reduce their salt intake.” All were required to own a telephone.
Clinical events, including strokes and hospitalizations throughout a 5-year follow-up, were tracked by phone calls made to all participants every 6 months and were documented at follow-up home visits.
Sunday is also to feature a Late-Breaking Trials session with a focus on COVID-19, which leads off with COLCOVID, a test of colchicine in patients hospitalized for suspected SARS-CoV-2 infection and in acute respiratory distress.
The 1,279 participants in Argentina were randomly assigned to receive or not receive the potent anti-inflammatory agent on top of antivirals and other standard management and followed for death or new need for mechanical ventilation. A successful outcome would contrast with the RECOVERY trial, which terminated a colchicine group of patients hospitalized with COVID-19 because of a lack of efficacy earlier this year.
COLCOVID is to be followed by the MICHELLE trial of rivaroxaban (Xarelto, Bayer/Janssen) prophylaxis, compared with no preventive oral anticoagulant, in 320 patients who, when hospitalized with COVID-19, had been on parenteral anticoagulants because of an elevated risk for venous thromboembolism. The trial, conducted in Brazil, called for postdischarge rivaroxaban at a once-daily dosage of 10 mg for about 1 month.
The session also includes a presentation called “Insights into the Effects of the COVID-19 Pandemic: Comprehensive Analysis from the GUIDE-HF Trial,” the primary outcomes of which will be reported on the first day of the Congress.
Following is a presentation on the PREPARE-IT study of icosapent ethyl (Vascepa, Amarin), given at high dosages intended to be anti-inflammatory, compared with placebo, in an estimated 4,000 adults. The trial has two groups: A prevention group of adults living and circulating in the community; and a treatment group of patients aged at least 40 years with confirmed symptomatic SARS-CoV-2 infection for whom the need for hospitalization isn’t clear.
Monday, Aug. 30
The final day of the Congress features a trial called Influenza Vaccination after Myocardial Infarction (IAMI), which has tested the secondary preventive effect of influenza vaccination by randomly assigning 2,571 patients to receive a standard vaccine or a saline placebo injection on one occasion.
Entry to the international trial called for a diagnosis of MI with or without ST-segment elevation, or stable CAD and age at least 75 years with other risk factors. The patients were followed for death, MI, stent thrombosis, and a slew of secondary endpoints over 12 months.
Monday offerings continue later in a time block leading off with the STEP trial, which has randomly assigned an estimated 8,000 patients at 40 centers in China who are 60 to 80 years of age with a systolic blood pressure of 140 to <190 mm Hg to be on standard guideline-based therapy or an intensive drug-management strategy.
The systolic BP goals are 130 to <150 mm Hg for standard care and 110 to <130 mm Hg for the intensive regimen. The composite primary endpoint includes death and clinical events related to acute coronary syndromes, HF, revascularization, and stroke.
Following on heels of STEP, the Amulet IDE trial – the first major randomized comparison of two transcatheter LAA closure devices – entered 1,878 patients with nonvalvular AFib who were considered high-risk for bleeding and stroke or systemic embolism.
They were randomly assigned in the noninferiority trial to receive either the AMPLATZER Amulet (Abbott Medical Devices) or the WATCHMAN (Boston Scientific) closure devices and were followed for safety and efficacy for up to 5 years.
Both LAA closure devices, intended to make patients with AFib less reliant on oral anticoagulation, are now available on both sides of the Atlantic – as well as many other countries – after the Amulet’s United States market approval on Aug. 16, based largely on the Amulet IDE trial.
Rounding out the final Hot Line set is one of the latest efforts to show the efficacy and safety of a very short DAPT period after coronary stenting in patients with acute coronary syndromes, the STOPDAPT-2 ACS trial.
The study assigned 3,008 patients in Japan to receive aspirin and clopidogrel for either 1 month or 1 year after implantation with an everolimus-eluting cobalt-chromium stent and followed them for up to 5 years for a composite of MI, CV death, stent thrombosis, stroke, and bleeding.
The trial follows the published STOPDAPT-2 trial that showed superiority for the 1-month DAPT regimen in a predominantly stable-CAD population treated with the same kind of stent.
Program structure and format
A total of 15 online channels are to be available in the morning, European time, their schedules running in parallel. Presentations often are prerecorded, but also include live sessions at 8:00 a.m. Central time and 12 p.m. CET (2:00 a.m. and 6:00 a.m. Eastern time) to liven up the channel offerings, Dr. Windecker observed, and to make them more immediate and potentially interactive.
Many of the parallel channels are devoted throughout the Congress to particular silos of cardiology; for example, arrhythmias and device therapy is on channel 3; CAD and acute care is on 5; HF is on 6; and preventive cardiology is on 9.
Other channels swing across different topics from day to day, such as channel 1, which covers COVID-19 topics on the first and third day of the meeting, “advances in science” on day 2, and “digital health, public health, health economics” on day 4.
The focus each day, starting at 2:00 p.m. CET (8:00 a.m. ET) and continuing into the evening in Europe, shifts over to the Prime Time live program, which features the Hot Line and guideline presentations and many of the live abstract presentations.
Dr. Kosiborod, not a researcher with the EMPEROR trials, is chair of the Dapagliflozin in Preserved Ejection Fraction Heart Failure ( PRESERVED-HF ) trial, which is scheduled for presentation at the September 2021 Heart Failure Society of American meeting.
A version of this article first appeared on Medscape.com.
There will be so much more to the annual congress of the European Society of Cardiology, which begins Aug. 27 with an all-virtual format, than detailed primary results of EMPEROR-Preserved, a trial that could mark a turning point for heart failure (HF) medical therapy.
Also among the featured Hot Line and Late-Breaking Science sessions are – along with many other studies – explorations of arrhythmia management (ablation or guided by loop recorder); secondary prevention, including by vaccination; oral anticoagulation, notably after transcatheter valve procedures; and colchicine or thrombosis prophylaxis in hospitalized patients with COVID-19.
There will even be a head-to-head comparison of two long-familiar left atrial appendage (LAA) occluders, and a population-based, randomized trial of sodium restriction through wide-scale use of a potassium-based salt substitute.
The congress will also introduce four guideline documents at sessions throughout the Congress, one on each day. They cover new and modified recommendations for heart failure; pacing, including cardiac resynchronization therapy (CRT); cardiovascular (CV) disease prevention; and, with cosponsorship from the European Association for Cardio-Thoracic Surgery, valvular heart disease.
The virtues of virtual
That next year’s Congress is slated for Aug. 27-30 in Barcelona should be welcome news for anyone whose “what if” curiosity about all-virtual conferences has already been satisfied. But with experience comes wisdom, as the medical societies have learned that online scientific meetings have some winning qualities that may be worth keeping, as least for a while.
“I think there is no doubt that the digital format will continue, for several reasons. One is that this pandemic is not over,” ESC Congress program committee chair Stephan Windecker, MD, Bern (Switzerland) University Hospital, , told this news organization. “As long as it is not over, the digital format is here to stay.”
But it also appears that people who haven’t been able to attend the congress in person are keen to log in and engage online, Dr. Windecker said. The 2020 all-virtual conference drew a much younger pool of registrants, on average, than did the live conferences before the pandemic.
“I think that’s an indication of people that may be in training, in early stages of their career, or they don’t have the support from departments or from their practice, or other financial means.” But they are able to participate via computer, tablet, or smartphone, he said.
“Another advantage is that the recorded content can be replayed at the convenience of whoever wants to consume it at a later point in time,” he added. “Those are just some examples why the digital format is likely to stay,” on its own or in a new age of hybrid meetings.
New and updated guidelines
Leading off the guideline series is the document on diagnosis and treatment of acute and chronic HF, which leveraged the past few busy years of HF clinical trials to arrive at a number of new recommendations and strengthened level-of-evidence ratings. It covers both drug and device therapy of HF with reduced ejection fraction (HFrEF) and acute decompensated HF, and tweaks and further enshrines the concept of HF with mildly reduced ejection fraction (HFmrEF).
Several updated recommendations for both long-used and novel medications, notably the sodium-glucose cotransporter 2 inhibitors, will be included because of the recently appreciated evidence-based impact in HFrEF, Dr. Windecker noted.
“I think it will be particularly interesting to look for the SGLT2 inhibitors as not a completely new class of drugs, but certainly one where there has been a lot of new evidence, to look at how those drugs will be integrated in the overall care pathway.”
A top-line preview of the new HF guideline limited to drug therapy, presented at July’s Heart Failure Association of the European Society of Cardiology (ESC-HFA), provided a simple answer to a common question in the new, bountiful age of HFrEF medications: Which meds, initiated in what order?
As it happens, the new recommendation for first-line HFrEF drug therapy is not a silver bullet, but a shotgun – prompt initiation of at least four meds, one from each of four drug classes: renin-angiotensin system inhibitors, beta-blockers, mineralocorticoid receptor antagonists (MRA), and SGLT2 inhibitors. Each class, as described in the document, is to be started as soon as safely feasible, in a sequence deemed appropriate for each individual patient.
Spotlight on EMPEROR-Preserved
The world already knows that the trial, which tested the SGLT2 inhibitor empagliflozin (Jardiance, Boehringer Ingelheim/Eli Lilly) on top of standard therapy, “met” its primary endpoint in almost 6,000 patients with HF with preserved ejection fraction (HFpEF), who included some with HFmrEF by more contemporary definitions.
That means patients in EMPEROR-Preserved assigned to take empagliflozin showed significantly fewer events that made up the study’s primary endpoint, a composite of CV death or HF hospitalization. It appears to be the first clearly significant overall medical therapy benefit for a clinical primary endpoint in a major randomized HFpEF drug trial.
And that, pending fuller presentation of trial results at the Congress on Aug. 27, could be a huge deal for the half of HF patients with left ventricular ejection fractions (LVEF) higher than the HFrEF range.
Those early top-line results weren’t a decisive bombshell for a field now filled with hope for a practice-changing empagliflozin outcome in EMPEROR-Preserved, which isn’t a certainty. They were more like the “boom” of a mortar launching a rocket of fireworks that may explode into a chrysanthemum or green comet or, sometimes, turn out to be no more than a dud. The promise of the early cursory results critically depends on further details.
“Provided there is a compelling benefit, this is what everyone has been waiting for in this condition for decades,” Mikhail N. Kosiborod, MD, director of cardiometabolic research at Saint Luke’s Mid-America Heart Institute, Kansas City, Mo., said.
“Already knowing that the trial met the primary endpoint is obviously very intriguing and encouraging,” he added. “But there are things we don’t know, such as: What is the magnitude of benefit? And whether that benefit, whatever the magnitude, is driven by reductions in both heart failure hospitalizations and cardiovascular death, or only one of the two.”
For example: “If we see an impressive benefit for reduction of hospitalizations, but not a significant reduction in death, that would still be a huge advance. That’s because, to date, we don’t have any drug for HFpEF that has convincingly demonstrated a compelling reduction in heart failure hospitalization or improvement in symptoms, function, or quality of life,” observed Dr. Kosiborod, who wasn’t part of EMPEROR-Preserved.
There have been “suggestions” from HFrEF trials that empagliflozin and dapagliflozin (Farxiga, AstraZeneca) “have very comparable effects on at least the endpoint of cardiovascular death or hospitalization for heart failure,” he said. “So, my expectation would be that whatever is observed in EMPEROR-Preserved is likely a class effect, as well.”
Following EMPEROR-Preserved on the agenda is EMPEROR-Pooled, a patient-level combined analysis of the EMPEROR series of trials that spans the range of HF, regardless of ejection fraction or diabetes status, primarily exploring the effects of empagliflozin on renal function.
Other offerings, Friday, Aug. 27
Scheduled immediately after EMPEROR-Preserved is a presentation on the SMART-MI trial, which should clarify whether management guided by continuous ambulatory monitoring is effective in patients considered at especially high arrhythmic risk. Entry called for recent myocardial infarction and an LVEF of 36%-50% with evidence of cardiac autonomic dysfunction.
The trial randomly assigned 400 such patients to be or not be implanted with a Reveal LINQ (Medtronic) loop recorder and followed them for up to 18 months, primarily for detection of potentially serious arrhythmic events. Endpoints that involved mortality, hospitalization or other clinical events were secondary.
In a time slot preceding both SMART-MI and EMPEROR-Preserved, the GUIDE-HF trial is following a projected 3,600 patients with HF implanted with a CardioMEMS HF System (Abbott) pulmonary artery (PA) pressure sensor to explore the its value for guiding management.
The trial’s three cohorts, followed for at least 12 months, include randomized sensor-monitored and control groups of patients with New York Heart Association class 2-4 symptoms, as well as a third observational set of patients in NYHA class 3. That’s the indication for which the CardioMEMS monitor gained approval in the United States in 2014 based on the 2011 CHAMPION trial, and which fared just as well in the 2017 CHAMPION Post-Approval Study.
The Friday Hot Lines also include Dal-GenE, which has entered about 6,000 patients with recent MI to test the once-abandoned cholesterol ester transfer protein (CETP) inhibitor dalcetrapib (DalCor) for any secondary-prevention benefits when used selectively. The trial’s hook: All its patients are confirmed to have the AA genotype of the rs1967309 variant in the ADCY9 gene, which has been associated with a pronounced clinical response to CETP inhibition.
Saturday, Aug. 28
The direct oral anticoagulants (DOACs) have largely replaced vitamin K antagonists in patients with nonvalvular atrial fibrillation (AFib). But whether DOACs are similarly preferable in the growing world population of people who have undergone transcatheter aortic valve replacement (TAVR or TAVI), an issue explored with variable results in the ATLANTIS and GALILEO trials, is far from settled.
The ENVISAGE-TAVI AF trial explored the question for the factor X inhibitor edoxaban (Savaysa, Lixiana, Daiichi-Sankyo) in 1,400 patients with AFib and a transfemoral TAVR in the previous 5 days, who were randomly assigned to the DOAC or standard management along with discretionary antiplatelet therapy. They’ve been followed for up to 3 years for a composite endpoint of clinical events – including death, MI, and stroke – and for major bleeding.
The day will also feature MASTER DAPT, a comparison of two dual-antiplatelet therapy (DAPT) regimens in an estimated 4,300 patients considered to be high-risk for bleeding who had received the sirolimus-eluting Ultimaster (Terumo) coronary stent, which has a bioresorbable polymer coating.
Investigators have randomly assigned patients to receive either very-short-duration DAPT, for about a month after stenting, followed by a P2Y12 inhibitor alone for up to a year after the procedure; or a more conventional regimen of a P2Y12 inhibitor for 6-12 months with aspirin maintained for a total of 12 months.
Later that day, investigators from the FIGARO-DKD trial will present their results based on 7,437 patients with type 2 diabetes and chronic kidney disease (CKD), a much fuller version than the top-line findings announced by sponsor Bayer 3 months ago.
Those top-line results suggested that patients assigned to receive the nonsteroidal nonselective mineralocorticoid receptor antagonist (MRA) finerenone (Kerendia) on top of standard care benefited with a drop in risk for the primary endpoint of CV death or nonfatal CV events.
Finerenone was recently approved in the United States for treating patients with both type 2 diabetes and CKD based on the published FIDELIO-DKD trial, which had seen less CKD progression and fewer CV events in such patients who took the novel MRA.
Although similar in design to FIGARO-DKD, FIDELIO-DKD had entered fewer patients with early-stage diabetic kidney disease (DKD). That led researchers to pool the two trials’ populations to create a cohort that spans the spectrum of DKD severity. An analysis of the pooled cohort, dubbed FIDELITY, is on the schedule after FIGARO-DKD.
After FIDELITY is the prospective APAF-CRT trial that is following a projected 1,830 patients with permanent, symptomatic AFib and a recent hospitalization for AFib or HF and who were not good candidates for standard ablation. They were assigned to receive either atrioventricular junctional ablation followed by CRT, with or without a defibrillation, on top of optimal meds – a so-called “ablate-and-pace” strategy – or an implantable cardioverter defibrillator with rate-control drug therapy.
The new analysis represents the trial’s second phase in which mortality was followed for 4 years as the primary endpoint, in contrast to the previously reported initial phase that followed the first 102 patients for 2 years for the composite primary endpoint of death, worsening HF, and HF hospitalization. The first phase had halted enrollment before reaching its planned target of 280 patients after an interim analysis showed a significant benefit for ablate and pace.
Next up: DECAAF 2, a randomized assessment of whether catheter ablation for AFib guided by delayed gadolinium enhancement on MRI, a proxy for scar tissue, can be more effective than standard AFib ablation by pulmonary vein isolation alone. An estimated 900 patients with persistent AFib who had never before undergone ablation for the arrhythmia were randomly assigned to one strategy or the other and followed for AFib recurrence over 18 months.
Sunday, Aug. 29
The TOMAHAWK trial aimed to clarify the optimal timing of invasive coronary angiography for resuscitated patients with non–ST-segment elevation out-of-hospital cardiac arrest, a broad population in a setting for which there is little randomized-trial guidance. Investigators randomly assigned 558 such patients to undergo immediate invasive angiography or to direct intensive care unit admission for initial standard care with discretionary delayed angiography. Patients were followed for all-cause mortality, with other clinical events and neurologic outcomes as secondary endpoints.
Next on the schedule, the RIPCORD-2 trial randomly assigned 1,100 patients with stable known or suspected coronary artery disease (CAD) to undergo conventional angiography alone or with added direct pressure-wire measurement of fractional flow reserve to guide management decisions. Primary outcomes include health care costs and patient-reported quality of life at 1 year.
Slated for later that day, the Asymptomatic Carotid Surgery Trial-2 (ACST-2) has entered an estimated 3600 patients with a substantial carotid artery narrowing not associated with symptoms but for which either carotid endarterectomy (CEA) or carotid artery stenting (CAS) was considered anatomically feasible. There also must have been “substantial uncertainty” regarding the optimal procedure choice.
The trial, conducted in 40 countries primarily in Europe and North America and launched in 2008, randomly assigned the patients to undergo either CEA or CAS, in both cases with appropriate medical therapy, and followed them for periprocedural events and up to 10 years for strokes and stroke-related events.
The LOOP study, which is to directly follow ACST-2, has explored whether screening for AFib using the Medtronic Reveal LINQ monitor in older patients with non-AFib stroke risk factors – with oral anticoagulation prescribed for those who test positive – can lower their risk for stroke or systemic embolism. It randomly assigned 6,000 such patients to care guided by the loop recorder or to standard care.
On a somewhat larger scale, the Salt Substitute and Stroke Study (SSaSS) randomly assigned a total of 20,996 people in about 600 villages across northern China and Tibet to sodium-restriction intervention and control groups by village. All participants had a history of stroke or were aged at least 60 years with uncontrolled hypertension.
As described by the trial’s online portal, participants in villages assigned to the intervention group were given a supply of a low-sodium, potassium-supplementing salt substitute to replace their own salt supplies, along with education on the health benefits of sodium restriction. Participants in control villages continued their normal diets and, at the trial’s beginning, received “advice to reduce their salt intake.” All were required to own a telephone.
Clinical events, including strokes and hospitalizations throughout a 5-year follow-up, were tracked by phone calls made to all participants every 6 months and were documented at follow-up home visits.
Sunday is also to feature a Late-Breaking Trials session with a focus on COVID-19, which leads off with COLCOVID, a test of colchicine in patients hospitalized for suspected SARS-CoV-2 infection and in acute respiratory distress.
The 1,279 participants in Argentina were randomly assigned to receive or not receive the potent anti-inflammatory agent on top of antivirals and other standard management and followed for death or new need for mechanical ventilation. A successful outcome would contrast with the RECOVERY trial, which terminated a colchicine group of patients hospitalized with COVID-19 because of a lack of efficacy earlier this year.
COLCOVID is to be followed by the MICHELLE trial of rivaroxaban (Xarelto, Bayer/Janssen) prophylaxis, compared with no preventive oral anticoagulant, in 320 patients who, when hospitalized with COVID-19, had been on parenteral anticoagulants because of an elevated risk for venous thromboembolism. The trial, conducted in Brazil, called for postdischarge rivaroxaban at a once-daily dosage of 10 mg for about 1 month.
The session also includes a presentation called “Insights into the Effects of the COVID-19 Pandemic: Comprehensive Analysis from the GUIDE-HF Trial,” the primary outcomes of which will be reported on the first day of the Congress.
Following is a presentation on the PREPARE-IT study of icosapent ethyl (Vascepa, Amarin), given at high dosages intended to be anti-inflammatory, compared with placebo, in an estimated 4,000 adults. The trial has two groups: A prevention group of adults living and circulating in the community; and a treatment group of patients aged at least 40 years with confirmed symptomatic SARS-CoV-2 infection for whom the need for hospitalization isn’t clear.
Monday, Aug. 30
The final day of the Congress features a trial called Influenza Vaccination after Myocardial Infarction (IAMI), which has tested the secondary preventive effect of influenza vaccination by randomly assigning 2,571 patients to receive a standard vaccine or a saline placebo injection on one occasion.
Entry to the international trial called for a diagnosis of MI with or without ST-segment elevation, or stable CAD and age at least 75 years with other risk factors. The patients were followed for death, MI, stent thrombosis, and a slew of secondary endpoints over 12 months.
Monday offerings continue later in a time block leading off with the STEP trial, which has randomly assigned an estimated 8,000 patients at 40 centers in China who are 60 to 80 years of age with a systolic blood pressure of 140 to <190 mm Hg to be on standard guideline-based therapy or an intensive drug-management strategy.
The systolic BP goals are 130 to <150 mm Hg for standard care and 110 to <130 mm Hg for the intensive regimen. The composite primary endpoint includes death and clinical events related to acute coronary syndromes, HF, revascularization, and stroke.
Following on heels of STEP, the Amulet IDE trial – the first major randomized comparison of two transcatheter LAA closure devices – entered 1,878 patients with nonvalvular AFib who were considered high-risk for bleeding and stroke or systemic embolism.
They were randomly assigned in the noninferiority trial to receive either the AMPLATZER Amulet (Abbott Medical Devices) or the WATCHMAN (Boston Scientific) closure devices and were followed for safety and efficacy for up to 5 years.
Both LAA closure devices, intended to make patients with AFib less reliant on oral anticoagulation, are now available on both sides of the Atlantic – as well as many other countries – after the Amulet’s United States market approval on Aug. 16, based largely on the Amulet IDE trial.
Rounding out the final Hot Line set is one of the latest efforts to show the efficacy and safety of a very short DAPT period after coronary stenting in patients with acute coronary syndromes, the STOPDAPT-2 ACS trial.
The study assigned 3,008 patients in Japan to receive aspirin and clopidogrel for either 1 month or 1 year after implantation with an everolimus-eluting cobalt-chromium stent and followed them for up to 5 years for a composite of MI, CV death, stent thrombosis, stroke, and bleeding.
The trial follows the published STOPDAPT-2 trial that showed superiority for the 1-month DAPT regimen in a predominantly stable-CAD population treated with the same kind of stent.
Program structure and format
A total of 15 online channels are to be available in the morning, European time, their schedules running in parallel. Presentations often are prerecorded, but also include live sessions at 8:00 a.m. Central time and 12 p.m. CET (2:00 a.m. and 6:00 a.m. Eastern time) to liven up the channel offerings, Dr. Windecker observed, and to make them more immediate and potentially interactive.
Many of the parallel channels are devoted throughout the Congress to particular silos of cardiology; for example, arrhythmias and device therapy is on channel 3; CAD and acute care is on 5; HF is on 6; and preventive cardiology is on 9.
Other channels swing across different topics from day to day, such as channel 1, which covers COVID-19 topics on the first and third day of the meeting, “advances in science” on day 2, and “digital health, public health, health economics” on day 4.
The focus each day, starting at 2:00 p.m. CET (8:00 a.m. ET) and continuing into the evening in Europe, shifts over to the Prime Time live program, which features the Hot Line and guideline presentations and many of the live abstract presentations.
Dr. Kosiborod, not a researcher with the EMPEROR trials, is chair of the Dapagliflozin in Preserved Ejection Fraction Heart Failure ( PRESERVED-HF ) trial, which is scheduled for presentation at the September 2021 Heart Failure Society of American meeting.
A version of this article first appeared on Medscape.com.
Chronic kidney disease tied to worse LAAO outcomes
The presence of chronic kidney disease (CKD) or end-stage renal disease (ESRD) is associated with worse in-hospital and short-term outcomes after left atrial appendage (LAA) closure, a nationwide study shows.
Patients with ESRD were particularly vulnerable, having about 6.5-fold higher odds of in-hospital mortality than those without CKD and about 11.5-fold higher odds than those with CKD, even after adjustment for potential confounders.
Patients with CKD had higher rates of stroke or transient ischemic attack (TIA) and more short-term readmissions for bleeding, Keerat Rai Ahuja, MD, Reading Hospital-Tower Health, West Reading, Pennsylvania, and colleagues reported August 16 in JACC: Cardiovascular Interventions.
CKD and ESRD are known to be associated with an increased risk for stroke and bleeding in patients with atrial fibrillation (AFib), yet data are limited on the safety and efficacy of LAA closure for stroke prevention in AFib patients with CKD or ESRD, they note.
“It’s important to know about CKD and understand that there may be an association with worse levels of CKD and worse outcomes, but the data that strikes me is really that for end-stage renal disease,” Matthew Sherwood, MD, MHS, who was not involved with the study, said in an interview.
He noted that data have not been published for patients with CKD and ESRD enrolled in the pivotal PROTECT-AF and PREVAIL trials of Boston Scientific’s Watchman device or from large clinical registries such as EWOLUTION and the company’s continued access protocol registries.
Further, it’s not well understood what the best strategy is to prevent stroke in AFib patients with ESRD and whether they benefit from anticoagulation with warfarin or any of the newer agents. “Thus, it’s hard to then say: ‘Well they have worse outcomes with Watchman,’ which is true as shown in this study, but they may not have any other options based upon the lack of data for oral anticoagulants in end-stage kidney disease patients,” said Dr. Sherwood, from the Inova Heart and Vascular Institute, Falls Church, Virginia.
The lack of clarity is concerning, given rising atrial fibrillation cases and the prevalence of abnormal renal function in everyday practice. In the present study – involving 21,274 patients undergoing LAA closure between 2016 and 2017 in the Nationwide Readmissions Database – 18.6% of patients had CKD stages I to V and 2.7% had ESRD based on ICD-10 codes.
In-hospital mortality was increased only in patients with ESRD. In all, 3.3% of patients with ESRD and 0.4% of those with no CKD died in hospital (adjusted odds ratio [aOR], 6.48), as did 0.5% of patients with CKD (aOR, 11.43; both P <.001).
“These patients represent a sicker population at baseline and have an inherent greater risk for mortality in cardiac interventions, as noted in other studies of structural heart interventions,” Dr. Ahuja and colleagues write.
Patients with CKD had a higher risk for in-hospital stroke or TIA than patients with no CKD (1.8% vs. 1.3%; aOR, 1.35; P = .038) and this risk continued up to 90 days after discharge (1.7% vs. 1.0%; aOR, 1.67; P = .007).
The in-hospital stroke rate was numerically higher in patients with ESRD compared with no CKD (aOR, 1.18; P = .62).
The authors point out that previous LAA closure and CKD studies have reported no differences in in-hospital or subsequent stroke/TIA rates in patients with and without CKD. Possible explanations are that patients with CKD in the present study had higher CHA2DS2-VASc scores than those without CKD (4.18 vs. 3.62) and, second, patients with CKD and AFib are known to have higher risk for thromboembolic events than those with AFib without CKD.
CKD patients were also more likely than those without CKD to experience in-hospital acute kidney injury or hemodialysis (aOR, 5.02; P <.001).
CKD has been shown to be independently associated with acute kidney injury (AKI) after LAA closure. AKI may have long-term thromboembolic consequences, the authors suggest, with one study reporting higher stroke risk at midterm follow-up in patients with AKI.
“As with other cardiac interventions in patients with CKD, efforts should be made to optimize preoperative renal function, minimize contrast volume, and avoid abrupt hemodynamic changes such as hypotension during the procedure to prevent AKI,” Dr. Ahuja and colleagues write.
Patients with CKD and ESRD had longer index length of stay than those without CKD but had similar rates of other in-hospital complications, such as systemic embolization, bleeding/transfusion, vascular complications, and pericardial tamponade requiring intervention.
Among the short-term outcomes, 30- and 90-day all-cause readmissions were increased in patients with CKD and ESRD compared with those without CKD, and 30-day bleeding readmissions were increased within the CKD cohort.
“With Watchman and left atrial appendage closure, what we see is that they have higher rates of readmission and other problems,” Dr. Sherwood said. “I think we understand that that’s probably related not to the procedure itself, not because the Watchman doesn’t work for end-stage kidney disease, but because the patients themselves are likely higher risk.”
Commonly used risk scores for atrial fibrillation, however, don’t take into account advanced kidney disease, he added.
Besides the inherent limitations of observational studies, Dr. Sherwood and the authors point to the lack of laboratory variables and procedural variables in the database, the fact that CKD was defined using ICD-10 codes, that outcomes were not clinically adjudicated, that unmeasured confounders likely still exist, and that long-term follow-up is lacking.
Dr. Sherwood, who wrote an editorial accompanying the study, said that the release of outcomes data from CKD and ESRD patients in the major clinical trials would be helpful going forward, as would possible involvement with the Kidney Disease Improving Global Outcomes organization.
“One of the main points of this study is that we just need a lot more research diving into this patient population,” he said.
The authors report no relevant financial relationships. Dr. Sherwood reports honoraria from Janssen and Medtronic. Editorial coauthor Sean Pokorney reports research grant support from Gilead, Boston Scientific, Pfizer, Bristol Myers Squibb, Janssen, and the Food and Drug Administration; and advisory board, consulting, and honoraria supports from Medtronic, Boston Scientific, Pfizer, Bristol Myers Squibb, Philips, and Zoll.
A version of this article first appeared on Medscape.com.
The presence of chronic kidney disease (CKD) or end-stage renal disease (ESRD) is associated with worse in-hospital and short-term outcomes after left atrial appendage (LAA) closure, a nationwide study shows.
Patients with ESRD were particularly vulnerable, having about 6.5-fold higher odds of in-hospital mortality than those without CKD and about 11.5-fold higher odds than those with CKD, even after adjustment for potential confounders.
Patients with CKD had higher rates of stroke or transient ischemic attack (TIA) and more short-term readmissions for bleeding, Keerat Rai Ahuja, MD, Reading Hospital-Tower Health, West Reading, Pennsylvania, and colleagues reported August 16 in JACC: Cardiovascular Interventions.
CKD and ESRD are known to be associated with an increased risk for stroke and bleeding in patients with atrial fibrillation (AFib), yet data are limited on the safety and efficacy of LAA closure for stroke prevention in AFib patients with CKD or ESRD, they note.
“It’s important to know about CKD and understand that there may be an association with worse levels of CKD and worse outcomes, but the data that strikes me is really that for end-stage renal disease,” Matthew Sherwood, MD, MHS, who was not involved with the study, said in an interview.
He noted that data have not been published for patients with CKD and ESRD enrolled in the pivotal PROTECT-AF and PREVAIL trials of Boston Scientific’s Watchman device or from large clinical registries such as EWOLUTION and the company’s continued access protocol registries.
Further, it’s not well understood what the best strategy is to prevent stroke in AFib patients with ESRD and whether they benefit from anticoagulation with warfarin or any of the newer agents. “Thus, it’s hard to then say: ‘Well they have worse outcomes with Watchman,’ which is true as shown in this study, but they may not have any other options based upon the lack of data for oral anticoagulants in end-stage kidney disease patients,” said Dr. Sherwood, from the Inova Heart and Vascular Institute, Falls Church, Virginia.
The lack of clarity is concerning, given rising atrial fibrillation cases and the prevalence of abnormal renal function in everyday practice. In the present study – involving 21,274 patients undergoing LAA closure between 2016 and 2017 in the Nationwide Readmissions Database – 18.6% of patients had CKD stages I to V and 2.7% had ESRD based on ICD-10 codes.
In-hospital mortality was increased only in patients with ESRD. In all, 3.3% of patients with ESRD and 0.4% of those with no CKD died in hospital (adjusted odds ratio [aOR], 6.48), as did 0.5% of patients with CKD (aOR, 11.43; both P <.001).
“These patients represent a sicker population at baseline and have an inherent greater risk for mortality in cardiac interventions, as noted in other studies of structural heart interventions,” Dr. Ahuja and colleagues write.
Patients with CKD had a higher risk for in-hospital stroke or TIA than patients with no CKD (1.8% vs. 1.3%; aOR, 1.35; P = .038) and this risk continued up to 90 days after discharge (1.7% vs. 1.0%; aOR, 1.67; P = .007).
The in-hospital stroke rate was numerically higher in patients with ESRD compared with no CKD (aOR, 1.18; P = .62).
The authors point out that previous LAA closure and CKD studies have reported no differences in in-hospital or subsequent stroke/TIA rates in patients with and without CKD. Possible explanations are that patients with CKD in the present study had higher CHA2DS2-VASc scores than those without CKD (4.18 vs. 3.62) and, second, patients with CKD and AFib are known to have higher risk for thromboembolic events than those with AFib without CKD.
CKD patients were also more likely than those without CKD to experience in-hospital acute kidney injury or hemodialysis (aOR, 5.02; P <.001).
CKD has been shown to be independently associated with acute kidney injury (AKI) after LAA closure. AKI may have long-term thromboembolic consequences, the authors suggest, with one study reporting higher stroke risk at midterm follow-up in patients with AKI.
“As with other cardiac interventions in patients with CKD, efforts should be made to optimize preoperative renal function, minimize contrast volume, and avoid abrupt hemodynamic changes such as hypotension during the procedure to prevent AKI,” Dr. Ahuja and colleagues write.
Patients with CKD and ESRD had longer index length of stay than those without CKD but had similar rates of other in-hospital complications, such as systemic embolization, bleeding/transfusion, vascular complications, and pericardial tamponade requiring intervention.
Among the short-term outcomes, 30- and 90-day all-cause readmissions were increased in patients with CKD and ESRD compared with those without CKD, and 30-day bleeding readmissions were increased within the CKD cohort.
“With Watchman and left atrial appendage closure, what we see is that they have higher rates of readmission and other problems,” Dr. Sherwood said. “I think we understand that that’s probably related not to the procedure itself, not because the Watchman doesn’t work for end-stage kidney disease, but because the patients themselves are likely higher risk.”
Commonly used risk scores for atrial fibrillation, however, don’t take into account advanced kidney disease, he added.
Besides the inherent limitations of observational studies, Dr. Sherwood and the authors point to the lack of laboratory variables and procedural variables in the database, the fact that CKD was defined using ICD-10 codes, that outcomes were not clinically adjudicated, that unmeasured confounders likely still exist, and that long-term follow-up is lacking.
Dr. Sherwood, who wrote an editorial accompanying the study, said that the release of outcomes data from CKD and ESRD patients in the major clinical trials would be helpful going forward, as would possible involvement with the Kidney Disease Improving Global Outcomes organization.
“One of the main points of this study is that we just need a lot more research diving into this patient population,” he said.
The authors report no relevant financial relationships. Dr. Sherwood reports honoraria from Janssen and Medtronic. Editorial coauthor Sean Pokorney reports research grant support from Gilead, Boston Scientific, Pfizer, Bristol Myers Squibb, Janssen, and the Food and Drug Administration; and advisory board, consulting, and honoraria supports from Medtronic, Boston Scientific, Pfizer, Bristol Myers Squibb, Philips, and Zoll.
A version of this article first appeared on Medscape.com.
The presence of chronic kidney disease (CKD) or end-stage renal disease (ESRD) is associated with worse in-hospital and short-term outcomes after left atrial appendage (LAA) closure, a nationwide study shows.
Patients with ESRD were particularly vulnerable, having about 6.5-fold higher odds of in-hospital mortality than those without CKD and about 11.5-fold higher odds than those with CKD, even after adjustment for potential confounders.
Patients with CKD had higher rates of stroke or transient ischemic attack (TIA) and more short-term readmissions for bleeding, Keerat Rai Ahuja, MD, Reading Hospital-Tower Health, West Reading, Pennsylvania, and colleagues reported August 16 in JACC: Cardiovascular Interventions.
CKD and ESRD are known to be associated with an increased risk for stroke and bleeding in patients with atrial fibrillation (AFib), yet data are limited on the safety and efficacy of LAA closure for stroke prevention in AFib patients with CKD or ESRD, they note.
“It’s important to know about CKD and understand that there may be an association with worse levels of CKD and worse outcomes, but the data that strikes me is really that for end-stage renal disease,” Matthew Sherwood, MD, MHS, who was not involved with the study, said in an interview.
He noted that data have not been published for patients with CKD and ESRD enrolled in the pivotal PROTECT-AF and PREVAIL trials of Boston Scientific’s Watchman device or from large clinical registries such as EWOLUTION and the company’s continued access protocol registries.
Further, it’s not well understood what the best strategy is to prevent stroke in AFib patients with ESRD and whether they benefit from anticoagulation with warfarin or any of the newer agents. “Thus, it’s hard to then say: ‘Well they have worse outcomes with Watchman,’ which is true as shown in this study, but they may not have any other options based upon the lack of data for oral anticoagulants in end-stage kidney disease patients,” said Dr. Sherwood, from the Inova Heart and Vascular Institute, Falls Church, Virginia.
The lack of clarity is concerning, given rising atrial fibrillation cases and the prevalence of abnormal renal function in everyday practice. In the present study – involving 21,274 patients undergoing LAA closure between 2016 and 2017 in the Nationwide Readmissions Database – 18.6% of patients had CKD stages I to V and 2.7% had ESRD based on ICD-10 codes.
In-hospital mortality was increased only in patients with ESRD. In all, 3.3% of patients with ESRD and 0.4% of those with no CKD died in hospital (adjusted odds ratio [aOR], 6.48), as did 0.5% of patients with CKD (aOR, 11.43; both P <.001).
“These patients represent a sicker population at baseline and have an inherent greater risk for mortality in cardiac interventions, as noted in other studies of structural heart interventions,” Dr. Ahuja and colleagues write.
Patients with CKD had a higher risk for in-hospital stroke or TIA than patients with no CKD (1.8% vs. 1.3%; aOR, 1.35; P = .038) and this risk continued up to 90 days after discharge (1.7% vs. 1.0%; aOR, 1.67; P = .007).
The in-hospital stroke rate was numerically higher in patients with ESRD compared with no CKD (aOR, 1.18; P = .62).
The authors point out that previous LAA closure and CKD studies have reported no differences in in-hospital or subsequent stroke/TIA rates in patients with and without CKD. Possible explanations are that patients with CKD in the present study had higher CHA2DS2-VASc scores than those without CKD (4.18 vs. 3.62) and, second, patients with CKD and AFib are known to have higher risk for thromboembolic events than those with AFib without CKD.
CKD patients were also more likely than those without CKD to experience in-hospital acute kidney injury or hemodialysis (aOR, 5.02; P <.001).
CKD has been shown to be independently associated with acute kidney injury (AKI) after LAA closure. AKI may have long-term thromboembolic consequences, the authors suggest, with one study reporting higher stroke risk at midterm follow-up in patients with AKI.
“As with other cardiac interventions in patients with CKD, efforts should be made to optimize preoperative renal function, minimize contrast volume, and avoid abrupt hemodynamic changes such as hypotension during the procedure to prevent AKI,” Dr. Ahuja and colleagues write.
Patients with CKD and ESRD had longer index length of stay than those without CKD but had similar rates of other in-hospital complications, such as systemic embolization, bleeding/transfusion, vascular complications, and pericardial tamponade requiring intervention.
Among the short-term outcomes, 30- and 90-day all-cause readmissions were increased in patients with CKD and ESRD compared with those without CKD, and 30-day bleeding readmissions were increased within the CKD cohort.
“With Watchman and left atrial appendage closure, what we see is that they have higher rates of readmission and other problems,” Dr. Sherwood said. “I think we understand that that’s probably related not to the procedure itself, not because the Watchman doesn’t work for end-stage kidney disease, but because the patients themselves are likely higher risk.”
Commonly used risk scores for atrial fibrillation, however, don’t take into account advanced kidney disease, he added.
Besides the inherent limitations of observational studies, Dr. Sherwood and the authors point to the lack of laboratory variables and procedural variables in the database, the fact that CKD was defined using ICD-10 codes, that outcomes were not clinically adjudicated, that unmeasured confounders likely still exist, and that long-term follow-up is lacking.
Dr. Sherwood, who wrote an editorial accompanying the study, said that the release of outcomes data from CKD and ESRD patients in the major clinical trials would be helpful going forward, as would possible involvement with the Kidney Disease Improving Global Outcomes organization.
“One of the main points of this study is that we just need a lot more research diving into this patient population,” he said.
The authors report no relevant financial relationships. Dr. Sherwood reports honoraria from Janssen and Medtronic. Editorial coauthor Sean Pokorney reports research grant support from Gilead, Boston Scientific, Pfizer, Bristol Myers Squibb, Janssen, and the Food and Drug Administration; and advisory board, consulting, and honoraria supports from Medtronic, Boston Scientific, Pfizer, Bristol Myers Squibb, Philips, and Zoll.
A version of this article first appeared on Medscape.com.
Increased stroke risk linked with excess sitting in those under 60
While the risk of stroke increased more than fourfold among sedentary people under the age of 60, no significant increase in risk was observed among older individuals, according to the study based on self-reported data from more than 140,000 people. This highlights the need for relevant public health messaging directed at younger people, reported lead author, Raed A. Joundi, MD, DPhil, a stroke fellow at the University of Calgary (Alta.), and colleagues, in the paper published in Stroke.
“Sedentary time has increased over the past 2 decades in the United States and Canada, particularly in the young, raising the importance of characterizing its effect on long-term health , ” the investigators wrote. “A better understanding of the risk of sedentary time specific to stroke may be important for public health campaigns to reduce sedentary behavior.” Dr. Joundi and colleagues reviewed data from the Canadian Community Health Survey, including 143,180 healthy individuals without baseline history of cancer, heart disease, or stroke. Those under the age of 40 years were also excluded from the analysis.
Excess sedentary leisure time was defined as 8 or more hours of sedentary leisure time per day, whereas low physical activity was defined as less than 3.5 metabolic equivalent hours per week. The analysis also included a range of demographic and medical covariates, such as age, sex, marital status, smoking status, presence of hypertension, and others.
After a median follow-up of 9.4 years, 2,965 stroke events occurred, with a median time from survey response of 5.6 years. Risk of stroke among individuals aged younger than 60 years who engaged in low physical activity and excess sedentary leisure time was increased 4.5-fold, compared with individuals with low physical activity who were sedentary less than 4 hours per day (fully adjusted hazard ratio, 4.50; 95% confidence interval, 1.64-12.3).
Findings highlight benefits of physical activity
Similar risk elevations were not observed among individuals aged 60-79 years, or those older than 80. And among people younger than 60, high physical activity appeared to eliminate the additional risk imposed by excess sedentary leisure time.
“Sedentary time is associated with higher risk of stroke in inactive individuals, but not an active individual, ” Dr. Joundi said in an interview. “So it suggests that there’s two ways to lower risk: One would be to lower your sedentary time, and the other would be to engage in physical activity.”
These interpretations are speculative, Dr. Joundi cautioned, as the study was not interventional. Even so, he said that the findings “bring the spotlight back on physical activity,” thereby aligning with previous research.
“The more you exercise, the more that relationship between sedentary time and poor health outcomes is blunted, and in fact, can be completely negated with enough physical activity,” he said.
How exactly physical activity offers such protection remains unclear, Dr. Joundi added. He speculated that regularity of exercise may be key, with each session counteracting the adverse effects of prolonged sedentary time, which may include reduced blood flow, increased insulin resistance, and inflammatory changes that can affect blood vessels.
“This study is particularly a message for younger individuals,” Dr. Joundi said, suggesting that the findings may alter behavior, as many people have witnessed, or are aware of, the long-term impacts of stroke.
“There’s a sort of social or cognitive aversion to stroke, I think, in the general population, because of how disabling it can be, and how it can lower your quality of life,” he said.
Subtle lifestyle changes may be enough.
For those aiming to lower their risk of stroke, Dr. Joundi suggested that subtle lifestyle changes may be enough.
“Ultimately, what we saw is that even minimal amounts of physical activity – walking 3 hours a week, for example – could blunt the impact of sedentary time,” he said. “Doing what you can, even if it’s a small amount, tends to be quite meaningful over a long period of time.”
Daniel T. Lackland, DrPH, professor of epidemiology in the department of neurology at the Medical University of South Carolina, Charleston, offered a similar takeaway, noting that small efforts can lead to great benefits.
“Less intense activity is still better than being sedentary,” he said in an interview. “For many people, if you do get up and you just walk around, move your arms around – do any kind of movement – that’s better than being sedentary.”
Dr. Lackland applauded the practicality of studying sedentary leisure time, versus overall leisure time, as many people can’t control their work environment.
“You can’t do very much about how you work your job,” Dr. Lackland said. “Sometimes we have to sit, and I guess there are things you can do – you can put a treadmill instead of a chair and that kind of thing – but more often than not, you don’t really have that choice to do something. With leisure time, though, you’re in full control. And so what do you do with your leisure time? Do you sit and watch TV, or do you engage in some type of activity? Not necessarily aerobic activity, but some type of activity that would not let you be sedentary. You want to be active as much as you possibly can.”
Dr. Joundi disclosed grant support from the Canadian Institutes of Health Research. The other investigators and Dr. Lackland reported no relevant disclosures.
While the risk of stroke increased more than fourfold among sedentary people under the age of 60, no significant increase in risk was observed among older individuals, according to the study based on self-reported data from more than 140,000 people. This highlights the need for relevant public health messaging directed at younger people, reported lead author, Raed A. Joundi, MD, DPhil, a stroke fellow at the University of Calgary (Alta.), and colleagues, in the paper published in Stroke.
“Sedentary time has increased over the past 2 decades in the United States and Canada, particularly in the young, raising the importance of characterizing its effect on long-term health , ” the investigators wrote. “A better understanding of the risk of sedentary time specific to stroke may be important for public health campaigns to reduce sedentary behavior.” Dr. Joundi and colleagues reviewed data from the Canadian Community Health Survey, including 143,180 healthy individuals without baseline history of cancer, heart disease, or stroke. Those under the age of 40 years were also excluded from the analysis.
Excess sedentary leisure time was defined as 8 or more hours of sedentary leisure time per day, whereas low physical activity was defined as less than 3.5 metabolic equivalent hours per week. The analysis also included a range of demographic and medical covariates, such as age, sex, marital status, smoking status, presence of hypertension, and others.
After a median follow-up of 9.4 years, 2,965 stroke events occurred, with a median time from survey response of 5.6 years. Risk of stroke among individuals aged younger than 60 years who engaged in low physical activity and excess sedentary leisure time was increased 4.5-fold, compared with individuals with low physical activity who were sedentary less than 4 hours per day (fully adjusted hazard ratio, 4.50; 95% confidence interval, 1.64-12.3).
Findings highlight benefits of physical activity
Similar risk elevations were not observed among individuals aged 60-79 years, or those older than 80. And among people younger than 60, high physical activity appeared to eliminate the additional risk imposed by excess sedentary leisure time.
“Sedentary time is associated with higher risk of stroke in inactive individuals, but not an active individual, ” Dr. Joundi said in an interview. “So it suggests that there’s two ways to lower risk: One would be to lower your sedentary time, and the other would be to engage in physical activity.”
These interpretations are speculative, Dr. Joundi cautioned, as the study was not interventional. Even so, he said that the findings “bring the spotlight back on physical activity,” thereby aligning with previous research.
“The more you exercise, the more that relationship between sedentary time and poor health outcomes is blunted, and in fact, can be completely negated with enough physical activity,” he said.
How exactly physical activity offers such protection remains unclear, Dr. Joundi added. He speculated that regularity of exercise may be key, with each session counteracting the adverse effects of prolonged sedentary time, which may include reduced blood flow, increased insulin resistance, and inflammatory changes that can affect blood vessels.
“This study is particularly a message for younger individuals,” Dr. Joundi said, suggesting that the findings may alter behavior, as many people have witnessed, or are aware of, the long-term impacts of stroke.
“There’s a sort of social or cognitive aversion to stroke, I think, in the general population, because of how disabling it can be, and how it can lower your quality of life,” he said.
Subtle lifestyle changes may be enough.
For those aiming to lower their risk of stroke, Dr. Joundi suggested that subtle lifestyle changes may be enough.
“Ultimately, what we saw is that even minimal amounts of physical activity – walking 3 hours a week, for example – could blunt the impact of sedentary time,” he said. “Doing what you can, even if it’s a small amount, tends to be quite meaningful over a long period of time.”
Daniel T. Lackland, DrPH, professor of epidemiology in the department of neurology at the Medical University of South Carolina, Charleston, offered a similar takeaway, noting that small efforts can lead to great benefits.
“Less intense activity is still better than being sedentary,” he said in an interview. “For many people, if you do get up and you just walk around, move your arms around – do any kind of movement – that’s better than being sedentary.”
Dr. Lackland applauded the practicality of studying sedentary leisure time, versus overall leisure time, as many people can’t control their work environment.
“You can’t do very much about how you work your job,” Dr. Lackland said. “Sometimes we have to sit, and I guess there are things you can do – you can put a treadmill instead of a chair and that kind of thing – but more often than not, you don’t really have that choice to do something. With leisure time, though, you’re in full control. And so what do you do with your leisure time? Do you sit and watch TV, or do you engage in some type of activity? Not necessarily aerobic activity, but some type of activity that would not let you be sedentary. You want to be active as much as you possibly can.”
Dr. Joundi disclosed grant support from the Canadian Institutes of Health Research. The other investigators and Dr. Lackland reported no relevant disclosures.
While the risk of stroke increased more than fourfold among sedentary people under the age of 60, no significant increase in risk was observed among older individuals, according to the study based on self-reported data from more than 140,000 people. This highlights the need for relevant public health messaging directed at younger people, reported lead author, Raed A. Joundi, MD, DPhil, a stroke fellow at the University of Calgary (Alta.), and colleagues, in the paper published in Stroke.
“Sedentary time has increased over the past 2 decades in the United States and Canada, particularly in the young, raising the importance of characterizing its effect on long-term health , ” the investigators wrote. “A better understanding of the risk of sedentary time specific to stroke may be important for public health campaigns to reduce sedentary behavior.” Dr. Joundi and colleagues reviewed data from the Canadian Community Health Survey, including 143,180 healthy individuals without baseline history of cancer, heart disease, or stroke. Those under the age of 40 years were also excluded from the analysis.
Excess sedentary leisure time was defined as 8 or more hours of sedentary leisure time per day, whereas low physical activity was defined as less than 3.5 metabolic equivalent hours per week. The analysis also included a range of demographic and medical covariates, such as age, sex, marital status, smoking status, presence of hypertension, and others.
After a median follow-up of 9.4 years, 2,965 stroke events occurred, with a median time from survey response of 5.6 years. Risk of stroke among individuals aged younger than 60 years who engaged in low physical activity and excess sedentary leisure time was increased 4.5-fold, compared with individuals with low physical activity who were sedentary less than 4 hours per day (fully adjusted hazard ratio, 4.50; 95% confidence interval, 1.64-12.3).
Findings highlight benefits of physical activity
Similar risk elevations were not observed among individuals aged 60-79 years, or those older than 80. And among people younger than 60, high physical activity appeared to eliminate the additional risk imposed by excess sedentary leisure time.
“Sedentary time is associated with higher risk of stroke in inactive individuals, but not an active individual, ” Dr. Joundi said in an interview. “So it suggests that there’s two ways to lower risk: One would be to lower your sedentary time, and the other would be to engage in physical activity.”
These interpretations are speculative, Dr. Joundi cautioned, as the study was not interventional. Even so, he said that the findings “bring the spotlight back on physical activity,” thereby aligning with previous research.
“The more you exercise, the more that relationship between sedentary time and poor health outcomes is blunted, and in fact, can be completely negated with enough physical activity,” he said.
How exactly physical activity offers such protection remains unclear, Dr. Joundi added. He speculated that regularity of exercise may be key, with each session counteracting the adverse effects of prolonged sedentary time, which may include reduced blood flow, increased insulin resistance, and inflammatory changes that can affect blood vessels.
“This study is particularly a message for younger individuals,” Dr. Joundi said, suggesting that the findings may alter behavior, as many people have witnessed, or are aware of, the long-term impacts of stroke.
“There’s a sort of social or cognitive aversion to stroke, I think, in the general population, because of how disabling it can be, and how it can lower your quality of life,” he said.
Subtle lifestyle changes may be enough.
For those aiming to lower their risk of stroke, Dr. Joundi suggested that subtle lifestyle changes may be enough.
“Ultimately, what we saw is that even minimal amounts of physical activity – walking 3 hours a week, for example – could blunt the impact of sedentary time,” he said. “Doing what you can, even if it’s a small amount, tends to be quite meaningful over a long period of time.”
Daniel T. Lackland, DrPH, professor of epidemiology in the department of neurology at the Medical University of South Carolina, Charleston, offered a similar takeaway, noting that small efforts can lead to great benefits.
“Less intense activity is still better than being sedentary,” he said in an interview. “For many people, if you do get up and you just walk around, move your arms around – do any kind of movement – that’s better than being sedentary.”
Dr. Lackland applauded the practicality of studying sedentary leisure time, versus overall leisure time, as many people can’t control their work environment.
“You can’t do very much about how you work your job,” Dr. Lackland said. “Sometimes we have to sit, and I guess there are things you can do – you can put a treadmill instead of a chair and that kind of thing – but more often than not, you don’t really have that choice to do something. With leisure time, though, you’re in full control. And so what do you do with your leisure time? Do you sit and watch TV, or do you engage in some type of activity? Not necessarily aerobic activity, but some type of activity that would not let you be sedentary. You want to be active as much as you possibly can.”
Dr. Joundi disclosed grant support from the Canadian Institutes of Health Research. The other investigators and Dr. Lackland reported no relevant disclosures.
FROM STROKE
FDA approves Abbott’s Amplatzer Amulet for AFib
The Food and Drug Administration has approved the Amplatzer Amulet left atrial appendage occluder (Abbott) to treat people with nonvalvular atrial fibrillation who are at increased risk for stroke and systemic embolism.
The Amulet and its competitor, Boston Scientific’s Watchman, are minimally invasive devices used to close off the left atrial appendage (LAA), an area where blood clots tend to form in people with atrial fibrillation.
Amulet uses dual-seal technology to completely and immediately seal the LAA, the company says, whereas the other minimally invasive solution uses a single component to seal the LAA that requires blood-thinning drugs to heal and additional patient monitoring. The Amulet also has the widest range of occluder sizes on the market and is recapturable and repositionable to ensure optimal placement.
“As the world’s population continues to age, we’re seeing a surge in atrial fibrillation cases, and with that comes increased risk of stroke. The approval of Abbott’s Amulet device provides physicians with a treatment option that reduces the risk of stroke and eliminates the need for blood-thinning medication immediately after the procedure, which is incredibly valuable given the bleeding risks associated with these medicines,” Dhanunjaya Lakkireddy, MD, Kansas City Heart Rhythm Institute at HCA Midwest Health, Overland Park, Kan., and principal investigator for the study that led to FDA approval, said in a news release from Abbott.
The FDA approval is supported by findings from the global Amulet IDE trial, a head-to-head comparison of the Amulet and Watchman devices in 1,878 participants with nonvalvular atrial fibrillation. The results will be presented virtually on Aug. 30 at the 2021 annual congress of the European Society of Cardiology.
The Amplatzer Amulet received CE Mark designation in 2013 and is approved for use in more than 80 countries, including in Australia, Canada, and European countries.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved the Amplatzer Amulet left atrial appendage occluder (Abbott) to treat people with nonvalvular atrial fibrillation who are at increased risk for stroke and systemic embolism.
The Amulet and its competitor, Boston Scientific’s Watchman, are minimally invasive devices used to close off the left atrial appendage (LAA), an area where blood clots tend to form in people with atrial fibrillation.
Amulet uses dual-seal technology to completely and immediately seal the LAA, the company says, whereas the other minimally invasive solution uses a single component to seal the LAA that requires blood-thinning drugs to heal and additional patient monitoring. The Amulet also has the widest range of occluder sizes on the market and is recapturable and repositionable to ensure optimal placement.
“As the world’s population continues to age, we’re seeing a surge in atrial fibrillation cases, and with that comes increased risk of stroke. The approval of Abbott’s Amulet device provides physicians with a treatment option that reduces the risk of stroke and eliminates the need for blood-thinning medication immediately after the procedure, which is incredibly valuable given the bleeding risks associated with these medicines,” Dhanunjaya Lakkireddy, MD, Kansas City Heart Rhythm Institute at HCA Midwest Health, Overland Park, Kan., and principal investigator for the study that led to FDA approval, said in a news release from Abbott.
The FDA approval is supported by findings from the global Amulet IDE trial, a head-to-head comparison of the Amulet and Watchman devices in 1,878 participants with nonvalvular atrial fibrillation. The results will be presented virtually on Aug. 30 at the 2021 annual congress of the European Society of Cardiology.
The Amplatzer Amulet received CE Mark designation in 2013 and is approved for use in more than 80 countries, including in Australia, Canada, and European countries.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved the Amplatzer Amulet left atrial appendage occluder (Abbott) to treat people with nonvalvular atrial fibrillation who are at increased risk for stroke and systemic embolism.
The Amulet and its competitor, Boston Scientific’s Watchman, are minimally invasive devices used to close off the left atrial appendage (LAA), an area where blood clots tend to form in people with atrial fibrillation.
Amulet uses dual-seal technology to completely and immediately seal the LAA, the company says, whereas the other minimally invasive solution uses a single component to seal the LAA that requires blood-thinning drugs to heal and additional patient monitoring. The Amulet also has the widest range of occluder sizes on the market and is recapturable and repositionable to ensure optimal placement.
“As the world’s population continues to age, we’re seeing a surge in atrial fibrillation cases, and with that comes increased risk of stroke. The approval of Abbott’s Amulet device provides physicians with a treatment option that reduces the risk of stroke and eliminates the need for blood-thinning medication immediately after the procedure, which is incredibly valuable given the bleeding risks associated with these medicines,” Dhanunjaya Lakkireddy, MD, Kansas City Heart Rhythm Institute at HCA Midwest Health, Overland Park, Kan., and principal investigator for the study that led to FDA approval, said in a news release from Abbott.
The FDA approval is supported by findings from the global Amulet IDE trial, a head-to-head comparison of the Amulet and Watchman devices in 1,878 participants with nonvalvular atrial fibrillation. The results will be presented virtually on Aug. 30 at the 2021 annual congress of the European Society of Cardiology.
The Amplatzer Amulet received CE Mark designation in 2013 and is approved for use in more than 80 countries, including in Australia, Canada, and European countries.
A version of this article first appeared on Medscape.com.
Exercise tied to 50% reduction in mortality after stroke
, with a greater than 50% reduction in risk.
Lead study author Raed A. Joundi, MD, DPhil, of the University of Calgary (Alta.), said he expected results to show exercise was beneficial, but was surprised by the magnitude of the association between physical activity and lower mortality risk.
The impact of physical activity also differed significantly by age; those younger than 75 had a 79% reduction in mortality risk, compared with 32% in those age 75 and older.
“This is even after adjusting for factors such heart disease, respiratory conditions, smoking, and other functional limitations,” said Dr. Joundi.
The study was published online Aug. 11 in the journal Neurology.
For this analysis, the researchers used data on a cohort of people across Canada (excluding the province of Quebec) over 3-9 years. The 895 patients with prior stroke averaged 72 years of age, while the 97,805 in the control group had an average age of 63.
Weekly physical activity averages were evaluated using the self-reporting Canadian Community Health Survey, which was linked with administrative databases to evaluate the association of physical activity with long-term risk for mortality among stroke survivors, compared with controls.
Physical activity was measured in metabolic equivalents (METs); meeting minimum physical activity guidelines was defined as 10 MET-hours/week.
During the study period, more stroke patients than controls died (24.7% vs. 5.7%). However, those who met the physical activity guideline recommendations of 10 MET-hours/week had a lower mortality, both in the stroke survivor group (14.6% vs. 33.2%; adjusted hazard ratio, 0.46; 95% confidence interval, 0.29-0.73) and among control participants (3.6% vs. 7.9%; aHR 0.69; 95% CI, 0.62-0.76).
The largest absolute and relative reduction in mortality was among stroke respondents younger than 75 (10.5% vs. 29%; aHR, 0.21; 95% CI, 0.10-0.43), the researchers note.
There was a significant interaction with age for the stroke patients but not the control group.
“The greatest reduction in mortality was seen between 0 and 10 METs per week … so the main point is that something is better than nothing,” said Dr. Joundi.
Exercise guidelines for the future
Although current guidelines recommend physical activity in stroke survivors, investigators noted that these are largely based on studies in the general population. Therefore, the aim of this research was to get a better understanding of the role of physical activity in the health of stroke survivors in the community, which could ultimately be used to design improved public health campaigns and physical activity interventions.
Given that this is a large study of stroke survivors in the community, Dr. Joundi hopes the results will influence future activity guidelines for those who have suffered a stroke.
“We found a log-linear relationship between physical activity and mortality such that 10 MET-hours/week was associated with large reductions in mortality with most benefit achieved by 20 MET-hours/week,” the authors concluded. “These thresholds could be considered for use in future guidelines for stroke.”
Clinical trials are underway to provide evidence for the implementation of exercise programs after stroke, they add, and offering physical activity programs to stroke survivors in the community “is an increasing priority in the U.S., Canada, and Europe.”
“People are at higher risk of death early on after a stroke but also months and years later, so if we can identify a relatively low-cost and easy intervention like physical activity to improve health and reduce the risk of death for stroke survivors it would be important,” Dr. Joundi said.
Key barriers
Paul George, MD, PhD, a stroke and vascular neurologist at Stanford (Calif.) University, said findings such as these further strengthen the argument that physical exercise is important after stroke.
“Because the study looked specifically at stroke patients, it can provide further guidance on physical activity recommendations that we provide to our patients following stroke,” said Dr. George, who was not associated with the study.
Going forward, he said, more research is needed to identify specifically what is preventing stroke patients from exercising more. What is required, he said, is “future research to determine the key barriers to physical activity following stroke and methods to reduce these will also be important to increasing physical activity in stroke survivors.”
Dr. Joundi said determining how to tailor exercise recommendations to meet the wide range of capabilities of stroke survivors will be another key factor.
“Stroke survivors may have some disabilities, so we need to be able to engage them at an [exercise] level that’s possible for them,” he said.
The study did not include stroke survivors living in long-term care homes.
The study had no targeted funding. Coauthor Eric E. Smith, MD, MPH, reports royalties from UpToDate, and consulting fees from Alnylam, Biogen, and Javelin. Dr. Joundi and the other coauthors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, with a greater than 50% reduction in risk.
Lead study author Raed A. Joundi, MD, DPhil, of the University of Calgary (Alta.), said he expected results to show exercise was beneficial, but was surprised by the magnitude of the association between physical activity and lower mortality risk.
The impact of physical activity also differed significantly by age; those younger than 75 had a 79% reduction in mortality risk, compared with 32% in those age 75 and older.
“This is even after adjusting for factors such heart disease, respiratory conditions, smoking, and other functional limitations,” said Dr. Joundi.
The study was published online Aug. 11 in the journal Neurology.
For this analysis, the researchers used data on a cohort of people across Canada (excluding the province of Quebec) over 3-9 years. The 895 patients with prior stroke averaged 72 years of age, while the 97,805 in the control group had an average age of 63.
Weekly physical activity averages were evaluated using the self-reporting Canadian Community Health Survey, which was linked with administrative databases to evaluate the association of physical activity with long-term risk for mortality among stroke survivors, compared with controls.
Physical activity was measured in metabolic equivalents (METs); meeting minimum physical activity guidelines was defined as 10 MET-hours/week.
During the study period, more stroke patients than controls died (24.7% vs. 5.7%). However, those who met the physical activity guideline recommendations of 10 MET-hours/week had a lower mortality, both in the stroke survivor group (14.6% vs. 33.2%; adjusted hazard ratio, 0.46; 95% confidence interval, 0.29-0.73) and among control participants (3.6% vs. 7.9%; aHR 0.69; 95% CI, 0.62-0.76).
The largest absolute and relative reduction in mortality was among stroke respondents younger than 75 (10.5% vs. 29%; aHR, 0.21; 95% CI, 0.10-0.43), the researchers note.
There was a significant interaction with age for the stroke patients but not the control group.
“The greatest reduction in mortality was seen between 0 and 10 METs per week … so the main point is that something is better than nothing,” said Dr. Joundi.
Exercise guidelines for the future
Although current guidelines recommend physical activity in stroke survivors, investigators noted that these are largely based on studies in the general population. Therefore, the aim of this research was to get a better understanding of the role of physical activity in the health of stroke survivors in the community, which could ultimately be used to design improved public health campaigns and physical activity interventions.
Given that this is a large study of stroke survivors in the community, Dr. Joundi hopes the results will influence future activity guidelines for those who have suffered a stroke.
“We found a log-linear relationship between physical activity and mortality such that 10 MET-hours/week was associated with large reductions in mortality with most benefit achieved by 20 MET-hours/week,” the authors concluded. “These thresholds could be considered for use in future guidelines for stroke.”
Clinical trials are underway to provide evidence for the implementation of exercise programs after stroke, they add, and offering physical activity programs to stroke survivors in the community “is an increasing priority in the U.S., Canada, and Europe.”
“People are at higher risk of death early on after a stroke but also months and years later, so if we can identify a relatively low-cost and easy intervention like physical activity to improve health and reduce the risk of death for stroke survivors it would be important,” Dr. Joundi said.
Key barriers
Paul George, MD, PhD, a stroke and vascular neurologist at Stanford (Calif.) University, said findings such as these further strengthen the argument that physical exercise is important after stroke.
“Because the study looked specifically at stroke patients, it can provide further guidance on physical activity recommendations that we provide to our patients following stroke,” said Dr. George, who was not associated with the study.
Going forward, he said, more research is needed to identify specifically what is preventing stroke patients from exercising more. What is required, he said, is “future research to determine the key barriers to physical activity following stroke and methods to reduce these will also be important to increasing physical activity in stroke survivors.”
Dr. Joundi said determining how to tailor exercise recommendations to meet the wide range of capabilities of stroke survivors will be another key factor.
“Stroke survivors may have some disabilities, so we need to be able to engage them at an [exercise] level that’s possible for them,” he said.
The study did not include stroke survivors living in long-term care homes.
The study had no targeted funding. Coauthor Eric E. Smith, MD, MPH, reports royalties from UpToDate, and consulting fees from Alnylam, Biogen, and Javelin. Dr. Joundi and the other coauthors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, with a greater than 50% reduction in risk.
Lead study author Raed A. Joundi, MD, DPhil, of the University of Calgary (Alta.), said he expected results to show exercise was beneficial, but was surprised by the magnitude of the association between physical activity and lower mortality risk.
The impact of physical activity also differed significantly by age; those younger than 75 had a 79% reduction in mortality risk, compared with 32% in those age 75 and older.
“This is even after adjusting for factors such heart disease, respiratory conditions, smoking, and other functional limitations,” said Dr. Joundi.
The study was published online Aug. 11 in the journal Neurology.
For this analysis, the researchers used data on a cohort of people across Canada (excluding the province of Quebec) over 3-9 years. The 895 patients with prior stroke averaged 72 years of age, while the 97,805 in the control group had an average age of 63.
Weekly physical activity averages were evaluated using the self-reporting Canadian Community Health Survey, which was linked with administrative databases to evaluate the association of physical activity with long-term risk for mortality among stroke survivors, compared with controls.
Physical activity was measured in metabolic equivalents (METs); meeting minimum physical activity guidelines was defined as 10 MET-hours/week.
During the study period, more stroke patients than controls died (24.7% vs. 5.7%). However, those who met the physical activity guideline recommendations of 10 MET-hours/week had a lower mortality, both in the stroke survivor group (14.6% vs. 33.2%; adjusted hazard ratio, 0.46; 95% confidence interval, 0.29-0.73) and among control participants (3.6% vs. 7.9%; aHR 0.69; 95% CI, 0.62-0.76).
The largest absolute and relative reduction in mortality was among stroke respondents younger than 75 (10.5% vs. 29%; aHR, 0.21; 95% CI, 0.10-0.43), the researchers note.
There was a significant interaction with age for the stroke patients but not the control group.
“The greatest reduction in mortality was seen between 0 and 10 METs per week … so the main point is that something is better than nothing,” said Dr. Joundi.
Exercise guidelines for the future
Although current guidelines recommend physical activity in stroke survivors, investigators noted that these are largely based on studies in the general population. Therefore, the aim of this research was to get a better understanding of the role of physical activity in the health of stroke survivors in the community, which could ultimately be used to design improved public health campaigns and physical activity interventions.
Given that this is a large study of stroke survivors in the community, Dr. Joundi hopes the results will influence future activity guidelines for those who have suffered a stroke.
“We found a log-linear relationship between physical activity and mortality such that 10 MET-hours/week was associated with large reductions in mortality with most benefit achieved by 20 MET-hours/week,” the authors concluded. “These thresholds could be considered for use in future guidelines for stroke.”
Clinical trials are underway to provide evidence for the implementation of exercise programs after stroke, they add, and offering physical activity programs to stroke survivors in the community “is an increasing priority in the U.S., Canada, and Europe.”
“People are at higher risk of death early on after a stroke but also months and years later, so if we can identify a relatively low-cost and easy intervention like physical activity to improve health and reduce the risk of death for stroke survivors it would be important,” Dr. Joundi said.
Key barriers
Paul George, MD, PhD, a stroke and vascular neurologist at Stanford (Calif.) University, said findings such as these further strengthen the argument that physical exercise is important after stroke.
“Because the study looked specifically at stroke patients, it can provide further guidance on physical activity recommendations that we provide to our patients following stroke,” said Dr. George, who was not associated with the study.
Going forward, he said, more research is needed to identify specifically what is preventing stroke patients from exercising more. What is required, he said, is “future research to determine the key barriers to physical activity following stroke and methods to reduce these will also be important to increasing physical activity in stroke survivors.”
Dr. Joundi said determining how to tailor exercise recommendations to meet the wide range of capabilities of stroke survivors will be another key factor.
“Stroke survivors may have some disabilities, so we need to be able to engage them at an [exercise] level that’s possible for them,” he said.
The study did not include stroke survivors living in long-term care homes.
The study had no targeted funding. Coauthor Eric E. Smith, MD, MPH, reports royalties from UpToDate, and consulting fees from Alnylam, Biogen, and Javelin. Dr. Joundi and the other coauthors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Short-term approach is best for seizure prevention after intracerebral hemorrhage
(sICH), new research shows.
Investigators created a model that simulated common clinical scenarios to compare four antiseizure drug strategies – conservative, moderate, aggressive, and risk-guided. They used the 2HELPS2B score as a risk stratification tool to guide clinical decisions.
The investigators found that the short-term, early-seizure prophylaxis strategies “dominated” long-term therapy under most clinical scenarios, underscoring the importance of early discontinuation of antiseizure drug therapy.
“The main message here was that strategies that involved long-term antiseizure drug prescription (moderate and aggressive) fail to provide better outcomes in most clinical scenarios, when compared with strategies using short-term prophylaxis (conservative and risk-guided),” senior investigator Lidia M.V.R. Moura, MD, MPH, assistant professor of neurology, Harvard Medical School, Boston, said in an interview.
The study was published online July 26 in JAMA Neurology.
Common complication
“Acute asymptomatic seizures [early seizures ≤7 days after stroke] are a common complication of sICH,” the authors noted.
Potential safety concerns have prompted recommendations against the use of antiseizure medications for primary prophylaxis. However, approximately 40% of U.S. patients with sICH do receive prophylactic levetiracetam before seizure development. For these patients, the duration of prophylaxis varies widely.
“Because seizure risk is a key determinant of which patient groups might benefit most from different prophylaxis strategies, validated tools for predicting early ... and late ... seizure risks could aid physicians in treatment decisions. However, no clinical trials or prospective studies have evaluated the net benefit of various strategies after sICH,” the investigators noted.
“Our patients who were survivors of an intracerebral hemorrhage motivated us to conduct the study,” said Dr. Moura, who is also director of the MGH NeuroValue Laboratory. “Some would come to the clinic with a long list of medications; some of them were taking antiseizure drugs for many years, but they never had a documented seizure.” These patients did not know why they had been taking an antiseizure drug for so long.
“In these conversations, we noted so much variability in indications and variability in patient access to specialty care to make treatment decisions. We noted that the evidence behind our current guidelines on seizure management was limited,” she added.
Dr. Moura and colleagues were “committed to improve outcome for people with neurological conditions by leveraging research methods that can help guide providers and systems, especially when data from clinical trials is lacking,” so they “decided to compare different strategies head to head using available data and generate evidence that could be used in situations with many trade-offs in risks and benefits.”
To investigate, the researchers used a simulation model and decision analysis to compare four treatment strategies on the basis of type of therapy (primary vs. secondary prophylaxis), timing of event (early vs. late seizures), and duration of therapy (1-week [short-term] versus indefinite [long-term] therapy).
These four strategies were as follows:
- Conservative: short-term (7-day) secondary early-seizure prophylaxis with long-term therapy after late seizure
- Moderate: long-term secondary early-seizure prophylaxis or late-seizure therapy
- Aggressive: long-term primary prophylaxis
- Risk-guided: short-term secondary early-seizure prophylaxis among low-risk patients (2HELPS2b score, 0), short-term primary prophylaxis among patients at higher risk (2HELPS2B score ≥1), and long-term secondary therapy for late seizure
The decision tree’s outcome measure was the number of expected quality-adjusted life-years.
Primary prophylaxis was defined as “treatment initiated immediately on hospital admission.” Secondary prophylaxis was defined as “treatment started after a seizure” and was subdivided into secondary early-seizure prophylaxis, defined as treatment started after a seizure occurring in the first 7 days after the stroke, or secondary late-seizure therapy, defined as treatment started or restarted after a seizure occurring after the first poststroke week.
Incorporate early-risk stratification tool
The researchers created four common clinical scenarios and then applied the decision-making model to each. They found that the preferred strategies differed, depending on the particular scenario.
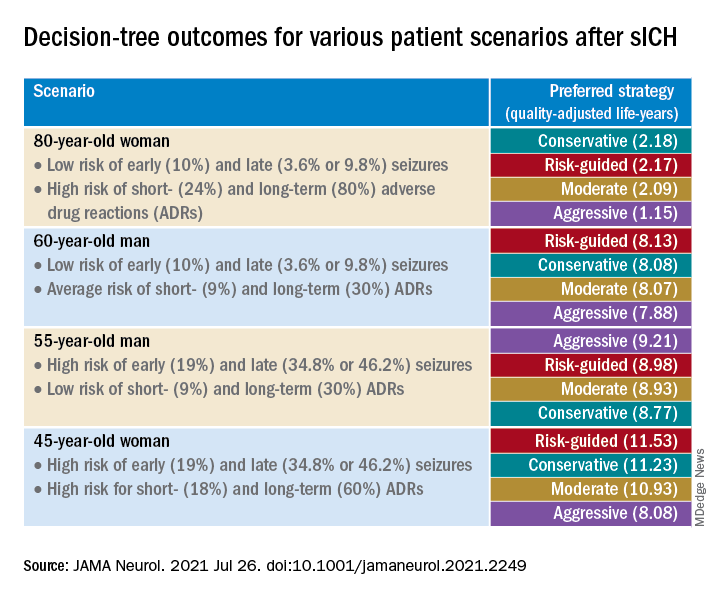
Sensitivity analyses revealed that short-term strategies, including the conservative and risk-guided approaches, were preferable in most cases, with the risk-guided strategy performing comparably or even better than alternative strategies in most cases.
“Our findings suggest that a strategy that incorporates an early-seizure risk stratification tool [2HELPS2B] is favored over alternative strategies in most settings,” Dr. Moura commented.
“Current services with rapidly available EEG may consider using a 1-hour screening with EEG upon admission for all patients presenting with sICH to risk-stratify those patients, using the 2HELPS2B tool,” she continued. “If EEG is unavailable for early-seizure risk stratification, the conservative strategy seems most reasonable.”
‘Potential fallacies’
Commenting on the study, José Biller, MD, professor and chairman, department of neurology, Loyola University Chicago, Maywood, Ill., called it a “well-written and intriguing contribution [to the field], with potential fallacies.”
The bottom line, he said, is that only a randomized, long-term, prospective, multicenter, high-quality study with larger cohorts can prove or disprove the investigators’ assumption.
The authors acknowledged that a limitation of the study was the use of published literature to obtain data to estimate model parameters and that they did not account for other possible factors that might modify some parameter estimates.
Nevertheless, Dr. Moura said the findings have important practical implications because they “highlight the importance of discontinuing antiseizure medications that were started during a hospitalization for sICH in patients that only had an early seizure.”
It is “of great importance for all providers to reassess the indication of antiseizure medications. Those drugs are not free of risks and can impact the patient’s health and quality of life,” she added.
The study was supported by grants from the National Institutes of Health. Dr. Moura reported receiving funding from the Centers for Disease Control and Prevention, the NIH, and the Epilepsy Foundation of America (Epilepsy Learning Healthcare System) as the director of the data coordinating center. Dr. Biller is the editor-in-chief of the Journal of Stroke and Cerebrovascular Diseases and a section editor of UpToDate.
A version of this article first appeared on Medscape.com.
(sICH), new research shows.
Investigators created a model that simulated common clinical scenarios to compare four antiseizure drug strategies – conservative, moderate, aggressive, and risk-guided. They used the 2HELPS2B score as a risk stratification tool to guide clinical decisions.
The investigators found that the short-term, early-seizure prophylaxis strategies “dominated” long-term therapy under most clinical scenarios, underscoring the importance of early discontinuation of antiseizure drug therapy.
“The main message here was that strategies that involved long-term antiseizure drug prescription (moderate and aggressive) fail to provide better outcomes in most clinical scenarios, when compared with strategies using short-term prophylaxis (conservative and risk-guided),” senior investigator Lidia M.V.R. Moura, MD, MPH, assistant professor of neurology, Harvard Medical School, Boston, said in an interview.
The study was published online July 26 in JAMA Neurology.
Common complication
“Acute asymptomatic seizures [early seizures ≤7 days after stroke] are a common complication of sICH,” the authors noted.
Potential safety concerns have prompted recommendations against the use of antiseizure medications for primary prophylaxis. However, approximately 40% of U.S. patients with sICH do receive prophylactic levetiracetam before seizure development. For these patients, the duration of prophylaxis varies widely.
“Because seizure risk is a key determinant of which patient groups might benefit most from different prophylaxis strategies, validated tools for predicting early ... and late ... seizure risks could aid physicians in treatment decisions. However, no clinical trials or prospective studies have evaluated the net benefit of various strategies after sICH,” the investigators noted.
“Our patients who were survivors of an intracerebral hemorrhage motivated us to conduct the study,” said Dr. Moura, who is also director of the MGH NeuroValue Laboratory. “Some would come to the clinic with a long list of medications; some of them were taking antiseizure drugs for many years, but they never had a documented seizure.” These patients did not know why they had been taking an antiseizure drug for so long.
“In these conversations, we noted so much variability in indications and variability in patient access to specialty care to make treatment decisions. We noted that the evidence behind our current guidelines on seizure management was limited,” she added.
Dr. Moura and colleagues were “committed to improve outcome for people with neurological conditions by leveraging research methods that can help guide providers and systems, especially when data from clinical trials is lacking,” so they “decided to compare different strategies head to head using available data and generate evidence that could be used in situations with many trade-offs in risks and benefits.”
To investigate, the researchers used a simulation model and decision analysis to compare four treatment strategies on the basis of type of therapy (primary vs. secondary prophylaxis), timing of event (early vs. late seizures), and duration of therapy (1-week [short-term] versus indefinite [long-term] therapy).
These four strategies were as follows:
- Conservative: short-term (7-day) secondary early-seizure prophylaxis with long-term therapy after late seizure
- Moderate: long-term secondary early-seizure prophylaxis or late-seizure therapy
- Aggressive: long-term primary prophylaxis
- Risk-guided: short-term secondary early-seizure prophylaxis among low-risk patients (2HELPS2b score, 0), short-term primary prophylaxis among patients at higher risk (2HELPS2B score ≥1), and long-term secondary therapy for late seizure
The decision tree’s outcome measure was the number of expected quality-adjusted life-years.
Primary prophylaxis was defined as “treatment initiated immediately on hospital admission.” Secondary prophylaxis was defined as “treatment started after a seizure” and was subdivided into secondary early-seizure prophylaxis, defined as treatment started after a seizure occurring in the first 7 days after the stroke, or secondary late-seizure therapy, defined as treatment started or restarted after a seizure occurring after the first poststroke week.
Incorporate early-risk stratification tool
The researchers created four common clinical scenarios and then applied the decision-making model to each. They found that the preferred strategies differed, depending on the particular scenario.

Sensitivity analyses revealed that short-term strategies, including the conservative and risk-guided approaches, were preferable in most cases, with the risk-guided strategy performing comparably or even better than alternative strategies in most cases.
“Our findings suggest that a strategy that incorporates an early-seizure risk stratification tool [2HELPS2B] is favored over alternative strategies in most settings,” Dr. Moura commented.
“Current services with rapidly available EEG may consider using a 1-hour screening with EEG upon admission for all patients presenting with sICH to risk-stratify those patients, using the 2HELPS2B tool,” she continued. “If EEG is unavailable for early-seizure risk stratification, the conservative strategy seems most reasonable.”
‘Potential fallacies’
Commenting on the study, José Biller, MD, professor and chairman, department of neurology, Loyola University Chicago, Maywood, Ill., called it a “well-written and intriguing contribution [to the field], with potential fallacies.”
The bottom line, he said, is that only a randomized, long-term, prospective, multicenter, high-quality study with larger cohorts can prove or disprove the investigators’ assumption.
The authors acknowledged that a limitation of the study was the use of published literature to obtain data to estimate model parameters and that they did not account for other possible factors that might modify some parameter estimates.
Nevertheless, Dr. Moura said the findings have important practical implications because they “highlight the importance of discontinuing antiseizure medications that were started during a hospitalization for sICH in patients that only had an early seizure.”
It is “of great importance for all providers to reassess the indication of antiseizure medications. Those drugs are not free of risks and can impact the patient’s health and quality of life,” she added.
The study was supported by grants from the National Institutes of Health. Dr. Moura reported receiving funding from the Centers for Disease Control and Prevention, the NIH, and the Epilepsy Foundation of America (Epilepsy Learning Healthcare System) as the director of the data coordinating center. Dr. Biller is the editor-in-chief of the Journal of Stroke and Cerebrovascular Diseases and a section editor of UpToDate.
A version of this article first appeared on Medscape.com.
(sICH), new research shows.
Investigators created a model that simulated common clinical scenarios to compare four antiseizure drug strategies – conservative, moderate, aggressive, and risk-guided. They used the 2HELPS2B score as a risk stratification tool to guide clinical decisions.
The investigators found that the short-term, early-seizure prophylaxis strategies “dominated” long-term therapy under most clinical scenarios, underscoring the importance of early discontinuation of antiseizure drug therapy.
“The main message here was that strategies that involved long-term antiseizure drug prescription (moderate and aggressive) fail to provide better outcomes in most clinical scenarios, when compared with strategies using short-term prophylaxis (conservative and risk-guided),” senior investigator Lidia M.V.R. Moura, MD, MPH, assistant professor of neurology, Harvard Medical School, Boston, said in an interview.
The study was published online July 26 in JAMA Neurology.
Common complication
“Acute asymptomatic seizures [early seizures ≤7 days after stroke] are a common complication of sICH,” the authors noted.
Potential safety concerns have prompted recommendations against the use of antiseizure medications for primary prophylaxis. However, approximately 40% of U.S. patients with sICH do receive prophylactic levetiracetam before seizure development. For these patients, the duration of prophylaxis varies widely.
“Because seizure risk is a key determinant of which patient groups might benefit most from different prophylaxis strategies, validated tools for predicting early ... and late ... seizure risks could aid physicians in treatment decisions. However, no clinical trials or prospective studies have evaluated the net benefit of various strategies after sICH,” the investigators noted.
“Our patients who were survivors of an intracerebral hemorrhage motivated us to conduct the study,” said Dr. Moura, who is also director of the MGH NeuroValue Laboratory. “Some would come to the clinic with a long list of medications; some of them were taking antiseizure drugs for many years, but they never had a documented seizure.” These patients did not know why they had been taking an antiseizure drug for so long.
“In these conversations, we noted so much variability in indications and variability in patient access to specialty care to make treatment decisions. We noted that the evidence behind our current guidelines on seizure management was limited,” she added.
Dr. Moura and colleagues were “committed to improve outcome for people with neurological conditions by leveraging research methods that can help guide providers and systems, especially when data from clinical trials is lacking,” so they “decided to compare different strategies head to head using available data and generate evidence that could be used in situations with many trade-offs in risks and benefits.”
To investigate, the researchers used a simulation model and decision analysis to compare four treatment strategies on the basis of type of therapy (primary vs. secondary prophylaxis), timing of event (early vs. late seizures), and duration of therapy (1-week [short-term] versus indefinite [long-term] therapy).
These four strategies were as follows:
- Conservative: short-term (7-day) secondary early-seizure prophylaxis with long-term therapy after late seizure
- Moderate: long-term secondary early-seizure prophylaxis or late-seizure therapy
- Aggressive: long-term primary prophylaxis
- Risk-guided: short-term secondary early-seizure prophylaxis among low-risk patients (2HELPS2b score, 0), short-term primary prophylaxis among patients at higher risk (2HELPS2B score ≥1), and long-term secondary therapy for late seizure
The decision tree’s outcome measure was the number of expected quality-adjusted life-years.
Primary prophylaxis was defined as “treatment initiated immediately on hospital admission.” Secondary prophylaxis was defined as “treatment started after a seizure” and was subdivided into secondary early-seizure prophylaxis, defined as treatment started after a seizure occurring in the first 7 days after the stroke, or secondary late-seizure therapy, defined as treatment started or restarted after a seizure occurring after the first poststroke week.
Incorporate early-risk stratification tool
The researchers created four common clinical scenarios and then applied the decision-making model to each. They found that the preferred strategies differed, depending on the particular scenario.

Sensitivity analyses revealed that short-term strategies, including the conservative and risk-guided approaches, were preferable in most cases, with the risk-guided strategy performing comparably or even better than alternative strategies in most cases.
“Our findings suggest that a strategy that incorporates an early-seizure risk stratification tool [2HELPS2B] is favored over alternative strategies in most settings,” Dr. Moura commented.
“Current services with rapidly available EEG may consider using a 1-hour screening with EEG upon admission for all patients presenting with sICH to risk-stratify those patients, using the 2HELPS2B tool,” she continued. “If EEG is unavailable for early-seizure risk stratification, the conservative strategy seems most reasonable.”
‘Potential fallacies’
Commenting on the study, José Biller, MD, professor and chairman, department of neurology, Loyola University Chicago, Maywood, Ill., called it a “well-written and intriguing contribution [to the field], with potential fallacies.”
The bottom line, he said, is that only a randomized, long-term, prospective, multicenter, high-quality study with larger cohorts can prove or disprove the investigators’ assumption.
The authors acknowledged that a limitation of the study was the use of published literature to obtain data to estimate model parameters and that they did not account for other possible factors that might modify some parameter estimates.
Nevertheless, Dr. Moura said the findings have important practical implications because they “highlight the importance of discontinuing antiseizure medications that were started during a hospitalization for sICH in patients that only had an early seizure.”
It is “of great importance for all providers to reassess the indication of antiseizure medications. Those drugs are not free of risks and can impact the patient’s health and quality of life,” she added.
The study was supported by grants from the National Institutes of Health. Dr. Moura reported receiving funding from the Centers for Disease Control and Prevention, the NIH, and the Epilepsy Foundation of America (Epilepsy Learning Healthcare System) as the director of the data coordinating center. Dr. Biller is the editor-in-chief of the Journal of Stroke and Cerebrovascular Diseases and a section editor of UpToDate.
A version of this article first appeared on Medscape.com.
Mobile stroke teams treat patients faster and reduce disability
Having a mobile interventional stroke team (MIST) travel to treat stroke patients soon after stroke onset may improve patient outcomes, according to a new study. A retrospective analysis of a pilot program in New York found that
“The use of a Mobile Interventional Stroke Team (MIST) traveling to Thrombectomy Capable Stroke Centers to perform endovascular thrombectomy has been shown to be significantly faster with improved discharge outcomes,” wrote lead author Jacob Morey, a doctoral Candidate at Icahn School of Medicine at Mount Sinai in New York and coauthors in the paper. Prior to this study, “the effect of the MIST model stratified by time of presentation” had yet to be studied.
The findings were published online on Aug. 5 in Stroke.
MIST model versus drip-and-ship
The researchers analyzed 226 patients who underwent endovascular thrombectomy between January 2017 and February 2020 at four hospitals in the Mount Sinai health system using the NYC MIST Trial and a stroke database. At baseline, all patients were functionally independent as assessed by the modified Rankin Scale (mRS, score of 0-2). 106 patients were treated by a MIST team – staffed by a neurointerventionalist, a fellow or physician assistant, and radiologic technologist – that traveled to the patient’s location. A total of 120 patients were transferred to a comprehensive stroke center (CSC) or a hospital with endovascular thrombectomy expertise. The analysis was stratified based on whether the patient presented in the early time window (≤ 6 hours) or late time window (> 6 hours).
Patients treated in the early time window were significantly more likely to be mobile and able to perform daily tasks (mRS ≤ 2) 90 days after the procedure in the MIST group (54%), compared with the transferred group (28%, P < 0.01). Outcomes did not differ significantly between groups in the late time window (35% vs. 41%, P = 0.77).
Similarly, early-time-window patients in the MIST group were more likely to have higher functionality at discharge, compared with transferred patients, based on the on the National Institutes of Health Stroke Scale (median score of 5.0 vs. 12.0, P < 0.01). There was no significant difference between groups treated in the late time window (median score of 5.0 vs. 11.0, P = 0.11).
“Ischemic strokes often progress rapidly and can cause severe damage because brain tissue dies quickly without oxygen, resulting in serious long-term disabilities or death,“ said Johanna Fifi, MD, of Icahn School of Medicine, said in a statement to the American Heart Association. “Assessing and treating stroke patients in the early window means that a greater number of fast-progressing strokes are identified and treated.”
Time is brain
Endovascular thrombectomy is a time-sensitive surgical procedure to remove large blood clots in acute ischemic stroke that has “historically been limited to comprehensive stroke centers,” the authors wrote in their paper. It is considered the standard of care in ischemic strokes, which make up 90% of all strokes. “Less than 50% of Americans have direct access to endovascular thrombectomy, the others must be transferred to a thrombectomy-capable hospital for treatment, often losing over 2 hours of time to treatment,” said Dr. Fifi. “Every minute is precious in treating stroke, and getting to a center that offers thrombectomy is very important. The MIST model would address this by providing faster access to this potentially life-saving, disability-reducing procedure.”
Access to timely endovascular thrombectomy is gradually improving as “more institutions and cities have implemented the [MIST] model.” Dr. Fifi said.
“This study stresses the importance of ‘time is brain,’ especially for patients in the early time window. Although the study is limited by the observational, retrospective design and was performed at a single integrated center, the findings are provocative,” said Louise McCullough, MD, of the University of Texas Health Science Center at Houston said in a statement to the American Heart Association. “The use of a MIST model highlights the potential benefit of early and urgent treatment for patients with large-vessel stroke. Stroke systems of care need to take advantage of any opportunity to treat patients early, wherever they are.”
The study was partly funded by a Stryker Foundation grant.
Having a mobile interventional stroke team (MIST) travel to treat stroke patients soon after stroke onset may improve patient outcomes, according to a new study. A retrospective analysis of a pilot program in New York found that
“The use of a Mobile Interventional Stroke Team (MIST) traveling to Thrombectomy Capable Stroke Centers to perform endovascular thrombectomy has been shown to be significantly faster with improved discharge outcomes,” wrote lead author Jacob Morey, a doctoral Candidate at Icahn School of Medicine at Mount Sinai in New York and coauthors in the paper. Prior to this study, “the effect of the MIST model stratified by time of presentation” had yet to be studied.
The findings were published online on Aug. 5 in Stroke.
MIST model versus drip-and-ship
The researchers analyzed 226 patients who underwent endovascular thrombectomy between January 2017 and February 2020 at four hospitals in the Mount Sinai health system using the NYC MIST Trial and a stroke database. At baseline, all patients were functionally independent as assessed by the modified Rankin Scale (mRS, score of 0-2). 106 patients were treated by a MIST team – staffed by a neurointerventionalist, a fellow or physician assistant, and radiologic technologist – that traveled to the patient’s location. A total of 120 patients were transferred to a comprehensive stroke center (CSC) or a hospital with endovascular thrombectomy expertise. The analysis was stratified based on whether the patient presented in the early time window (≤ 6 hours) or late time window (> 6 hours).
Patients treated in the early time window were significantly more likely to be mobile and able to perform daily tasks (mRS ≤ 2) 90 days after the procedure in the MIST group (54%), compared with the transferred group (28%, P < 0.01). Outcomes did not differ significantly between groups in the late time window (35% vs. 41%, P = 0.77).
Similarly, early-time-window patients in the MIST group were more likely to have higher functionality at discharge, compared with transferred patients, based on the on the National Institutes of Health Stroke Scale (median score of 5.0 vs. 12.0, P < 0.01). There was no significant difference between groups treated in the late time window (median score of 5.0 vs. 11.0, P = 0.11).
“Ischemic strokes often progress rapidly and can cause severe damage because brain tissue dies quickly without oxygen, resulting in serious long-term disabilities or death,“ said Johanna Fifi, MD, of Icahn School of Medicine, said in a statement to the American Heart Association. “Assessing and treating stroke patients in the early window means that a greater number of fast-progressing strokes are identified and treated.”
Time is brain
Endovascular thrombectomy is a time-sensitive surgical procedure to remove large blood clots in acute ischemic stroke that has “historically been limited to comprehensive stroke centers,” the authors wrote in their paper. It is considered the standard of care in ischemic strokes, which make up 90% of all strokes. “Less than 50% of Americans have direct access to endovascular thrombectomy, the others must be transferred to a thrombectomy-capable hospital for treatment, often losing over 2 hours of time to treatment,” said Dr. Fifi. “Every minute is precious in treating stroke, and getting to a center that offers thrombectomy is very important. The MIST model would address this by providing faster access to this potentially life-saving, disability-reducing procedure.”
Access to timely endovascular thrombectomy is gradually improving as “more institutions and cities have implemented the [MIST] model.” Dr. Fifi said.
“This study stresses the importance of ‘time is brain,’ especially for patients in the early time window. Although the study is limited by the observational, retrospective design and was performed at a single integrated center, the findings are provocative,” said Louise McCullough, MD, of the University of Texas Health Science Center at Houston said in a statement to the American Heart Association. “The use of a MIST model highlights the potential benefit of early and urgent treatment for patients with large-vessel stroke. Stroke systems of care need to take advantage of any opportunity to treat patients early, wherever they are.”
The study was partly funded by a Stryker Foundation grant.
Having a mobile interventional stroke team (MIST) travel to treat stroke patients soon after stroke onset may improve patient outcomes, according to a new study. A retrospective analysis of a pilot program in New York found that
“The use of a Mobile Interventional Stroke Team (MIST) traveling to Thrombectomy Capable Stroke Centers to perform endovascular thrombectomy has been shown to be significantly faster with improved discharge outcomes,” wrote lead author Jacob Morey, a doctoral Candidate at Icahn School of Medicine at Mount Sinai in New York and coauthors in the paper. Prior to this study, “the effect of the MIST model stratified by time of presentation” had yet to be studied.
The findings were published online on Aug. 5 in Stroke.
MIST model versus drip-and-ship
The researchers analyzed 226 patients who underwent endovascular thrombectomy between January 2017 and February 2020 at four hospitals in the Mount Sinai health system using the NYC MIST Trial and a stroke database. At baseline, all patients were functionally independent as assessed by the modified Rankin Scale (mRS, score of 0-2). 106 patients were treated by a MIST team – staffed by a neurointerventionalist, a fellow or physician assistant, and radiologic technologist – that traveled to the patient’s location. A total of 120 patients were transferred to a comprehensive stroke center (CSC) or a hospital with endovascular thrombectomy expertise. The analysis was stratified based on whether the patient presented in the early time window (≤ 6 hours) or late time window (> 6 hours).
Patients treated in the early time window were significantly more likely to be mobile and able to perform daily tasks (mRS ≤ 2) 90 days after the procedure in the MIST group (54%), compared with the transferred group (28%, P < 0.01). Outcomes did not differ significantly between groups in the late time window (35% vs. 41%, P = 0.77).
Similarly, early-time-window patients in the MIST group were more likely to have higher functionality at discharge, compared with transferred patients, based on the on the National Institutes of Health Stroke Scale (median score of 5.0 vs. 12.0, P < 0.01). There was no significant difference between groups treated in the late time window (median score of 5.0 vs. 11.0, P = 0.11).
“Ischemic strokes often progress rapidly and can cause severe damage because brain tissue dies quickly without oxygen, resulting in serious long-term disabilities or death,“ said Johanna Fifi, MD, of Icahn School of Medicine, said in a statement to the American Heart Association. “Assessing and treating stroke patients in the early window means that a greater number of fast-progressing strokes are identified and treated.”
Time is brain
Endovascular thrombectomy is a time-sensitive surgical procedure to remove large blood clots in acute ischemic stroke that has “historically been limited to comprehensive stroke centers,” the authors wrote in their paper. It is considered the standard of care in ischemic strokes, which make up 90% of all strokes. “Less than 50% of Americans have direct access to endovascular thrombectomy, the others must be transferred to a thrombectomy-capable hospital for treatment, often losing over 2 hours of time to treatment,” said Dr. Fifi. “Every minute is precious in treating stroke, and getting to a center that offers thrombectomy is very important. The MIST model would address this by providing faster access to this potentially life-saving, disability-reducing procedure.”
Access to timely endovascular thrombectomy is gradually improving as “more institutions and cities have implemented the [MIST] model.” Dr. Fifi said.
“This study stresses the importance of ‘time is brain,’ especially for patients in the early time window. Although the study is limited by the observational, retrospective design and was performed at a single integrated center, the findings are provocative,” said Louise McCullough, MD, of the University of Texas Health Science Center at Houston said in a statement to the American Heart Association. “The use of a MIST model highlights the potential benefit of early and urgent treatment for patients with large-vessel stroke. Stroke systems of care need to take advantage of any opportunity to treat patients early, wherever they are.”
The study was partly funded by a Stryker Foundation grant.
FROM STROKE
FDA panel balks at TriGuard 3 cerebral embolic device for TAVR
A Food and Drug Administration advisory panel struggled to muster support for marketing clearance of the TriGuard 3 (Keystone Heart) device for use during transcatheter aortic valve replacement (TAVR).
The Circulatory Systems Devices Panel of the Medical Devices Advisory Committee took no vote when it met Aug. 3, but weighed evidence for a proposed indication for the device “to minimize the risk of cerebral damage by deflecting embolic debris away from the cerebral circulation” during TAVR.
“While this device may deflect some debris, the data would suggest it may also create issues,” said Keith B. Allen, MD, director of surgical research at the Mid America Heart & Lung Surgeons, Kansas City, Mo. “I am really concerned that our desire and the emotion that surrounds preventing stroke are not being supported by the data.”
TriGuard 3 received CE Mark in Europe in March 2020. It was submitted for 510(k) clearance and seeks to prove substantial equivalence to the predicate Sentinel device (Claret Medical), currently the only approved embolic protection device in the United States.
The device is designed to cover all three major aortic vessels (innominate, left carotid, and left subclavian arteries) and is delivered transfemorally through an 8F sheath, whereas the Sentinel is positioned within the branch vessels, doesn’t cover the left subclavian artery, and is introduced through the radial or brachial artery via a 6F sheath.
TriGuard 3 faced an uphill battle, however, after failing to meet the primary composite efficacy endpoint in the REFLECT phase 2 trial (P = .857), with numeric trends showing higher all-cause mortality or any stroke at 30 days (9.8% vs. 6.7%) than pooled control subjects without embolic protection.
Rates for other components of the endpoint also trended higher with the device: National Institutes of Stroke Stroke Scale score worsening 2-5 days after the procedure, cerebral ischemic lesions on MRI 2-5 days after the procedure, and total cerebral ischemic lesion volume.
The Sentinel device was approved in 2017 after it failed to meet its primary efficacy endpoint of new brain lesion volume on MRI, but death and stroke rates favored the device over control, the panel pointed out.
The sponsor provided additional analyses in the per treatment (PT) population, defined as those with complete three-vessel coverage in at least two of three procedural time points. Compared with pooled control subjects, most of the imaging endpoints favored the TriGuard 3 device, but clinical neurologic event rates continued to favor the control group.
“The data used to demonstrate efficacy are all based on the PT subpopulation of the whole population, and those have to be considered promissory data,” said John Hirshfeld, MD, emeritus professor, University of Pennsylvania, Philadelphia. “This is the group where everything went well and for us to decide that’s achievable in the general population is speculative.”
Safety data
The REFLECT trial did meet its primary safety endpoint, with a 30-day major adverse cardiovascular event rate of 15.9%, compared with a performance goal of 34.4% (P < .0001).
Although prespecified, panel members pushed back, saying that the performance goal was unacceptably high, with several members remarking they’d never heard of a trial adding 9% as a “fudge factor” to a 25% historic control rate to get to the 34% performance target.
Keystone health officials noted that REFLECT was not designed to demonstrate a significant difference in the rate of primary safety events, compared with control. Instead, its purpose was to demonstrate that TriGuard 3 did not increase the risk associated with a TAVR procedure.
The TriGuard 3 device was successfully placed and retrieved in 100% of patients, but complete coverage was not uniform, with 72% of 157 as-treated patients having complete three-vessel coverage post TAVR but 15% having no coverage.
Panel members also expressed concern over device interference during TAVR, which was reported in nearly 10% of all TriGuard patients.
The TriGuard 3 group had 11 major vascular complications, 2 directly related to the device, and 3 stage 3 acute kidney injuries, whereas neither complication occurred in the control group.
Throughout the 9-hour hearing, the panel wrestled with what was described as a highly select patient group and small patient numbers that made it difficult to interpret observed differences. The trial involved 157 TriGuard 3 patients (including 41 from the roll-in phase) and 119 control subjects pooled from phase 2 of the trial (n = 57) and from phase 1 using the early-stage TriGuard HDH device (n = 57).
Pieter Stella, MD, PhD, Utrecht (the Netherlands) Medical Center, also presented “real-world” evidence from 75 patients in the Netherlands using the latest iteration of the device available in Europe with updates to the crimper and additional training materials to prevent the device from torquing during delivery. No strokes were reported, one patient had a transient ischemic attack (TIA), and two patients had a dissection, which resolved without sequelae.
Ralph Brindis, MD, MPH, professor of medicine, University of California, San Francisco, countered that there were only three experienced operators from a single center and that the stroke incidence was physician reported, “not data we can really embrace.”
There was much debate over why enrollment in phase 2 of the RHYTHM trial was temporarily paused in February 2019, briefly restarted, and then prematurely stopped in April 2019.
FDA officials said the study was paused at the recommendation of the data monitoring committee (DMC) because rates of safety events were different between patients and control subjects and operational errors called into question the accuracy of the data being reviewed. Ultimately, both the DMC and FDA recommended study suspension.
During the public hearing, TAVR pioneer Alain Cribier, MD, University of Rouen’s Charles Nicolle Hospital, Mont-Saint-Aignan, France, said the TriGuard 3 is of interest because it can be used with minimal need for manipulation and complete coverage of the cerebral vessels that is achieved by diverting rather than capturing debris. “The rapid and exponential growth of TAVR procedures demands safe TAVR interventions and the use of cerebral protection devices is a step in this direction.”
Others took a dim view. “Given that the Sentinel device has not demonstrated benefit on clinical outcomes, there is significant concern about similar devices, such as the TriGuard 3, providing clinical benefit,” Rita Redberg, MD, Sanket Dhruva, MD, and Robin Ji, University of California, San Francisco, wrote in a letter submitted to the panel.
Commenting further, they added: “With the results from the REFLECT II trial demonstrating no evidence for clinical outcome benefit in TAVR patients, and numerically higher rates for stroke risk, mortality, bleeding risk, and other dangerous adverse complications among those treated, it is concerning and dangerous for patient safety that the TriGUARD 3 cerebral embolic protection device is being considered for FDA 510(k) clearance.”
The FDA panel members reported no financial relationships.
A version of this article first appeared on Medscape.com.
A Food and Drug Administration advisory panel struggled to muster support for marketing clearance of the TriGuard 3 (Keystone Heart) device for use during transcatheter aortic valve replacement (TAVR).
The Circulatory Systems Devices Panel of the Medical Devices Advisory Committee took no vote when it met Aug. 3, but weighed evidence for a proposed indication for the device “to minimize the risk of cerebral damage by deflecting embolic debris away from the cerebral circulation” during TAVR.
“While this device may deflect some debris, the data would suggest it may also create issues,” said Keith B. Allen, MD, director of surgical research at the Mid America Heart & Lung Surgeons, Kansas City, Mo. “I am really concerned that our desire and the emotion that surrounds preventing stroke are not being supported by the data.”
TriGuard 3 received CE Mark in Europe in March 2020. It was submitted for 510(k) clearance and seeks to prove substantial equivalence to the predicate Sentinel device (Claret Medical), currently the only approved embolic protection device in the United States.
The device is designed to cover all three major aortic vessels (innominate, left carotid, and left subclavian arteries) and is delivered transfemorally through an 8F sheath, whereas the Sentinel is positioned within the branch vessels, doesn’t cover the left subclavian artery, and is introduced through the radial or brachial artery via a 6F sheath.
TriGuard 3 faced an uphill battle, however, after failing to meet the primary composite efficacy endpoint in the REFLECT phase 2 trial (P = .857), with numeric trends showing higher all-cause mortality or any stroke at 30 days (9.8% vs. 6.7%) than pooled control subjects without embolic protection.
Rates for other components of the endpoint also trended higher with the device: National Institutes of Stroke Stroke Scale score worsening 2-5 days after the procedure, cerebral ischemic lesions on MRI 2-5 days after the procedure, and total cerebral ischemic lesion volume.
The Sentinel device was approved in 2017 after it failed to meet its primary efficacy endpoint of new brain lesion volume on MRI, but death and stroke rates favored the device over control, the panel pointed out.
The sponsor provided additional analyses in the per treatment (PT) population, defined as those with complete three-vessel coverage in at least two of three procedural time points. Compared with pooled control subjects, most of the imaging endpoints favored the TriGuard 3 device, but clinical neurologic event rates continued to favor the control group.
“The data used to demonstrate efficacy are all based on the PT subpopulation of the whole population, and those have to be considered promissory data,” said John Hirshfeld, MD, emeritus professor, University of Pennsylvania, Philadelphia. “This is the group where everything went well and for us to decide that’s achievable in the general population is speculative.”
Safety data
The REFLECT trial did meet its primary safety endpoint, with a 30-day major adverse cardiovascular event rate of 15.9%, compared with a performance goal of 34.4% (P < .0001).
Although prespecified, panel members pushed back, saying that the performance goal was unacceptably high, with several members remarking they’d never heard of a trial adding 9% as a “fudge factor” to a 25% historic control rate to get to the 34% performance target.
Keystone health officials noted that REFLECT was not designed to demonstrate a significant difference in the rate of primary safety events, compared with control. Instead, its purpose was to demonstrate that TriGuard 3 did not increase the risk associated with a TAVR procedure.
The TriGuard 3 device was successfully placed and retrieved in 100% of patients, but complete coverage was not uniform, with 72% of 157 as-treated patients having complete three-vessel coverage post TAVR but 15% having no coverage.
Panel members also expressed concern over device interference during TAVR, which was reported in nearly 10% of all TriGuard patients.
The TriGuard 3 group had 11 major vascular complications, 2 directly related to the device, and 3 stage 3 acute kidney injuries, whereas neither complication occurred in the control group.
Throughout the 9-hour hearing, the panel wrestled with what was described as a highly select patient group and small patient numbers that made it difficult to interpret observed differences. The trial involved 157 TriGuard 3 patients (including 41 from the roll-in phase) and 119 control subjects pooled from phase 2 of the trial (n = 57) and from phase 1 using the early-stage TriGuard HDH device (n = 57).
Pieter Stella, MD, PhD, Utrecht (the Netherlands) Medical Center, also presented “real-world” evidence from 75 patients in the Netherlands using the latest iteration of the device available in Europe with updates to the crimper and additional training materials to prevent the device from torquing during delivery. No strokes were reported, one patient had a transient ischemic attack (TIA), and two patients had a dissection, which resolved without sequelae.
Ralph Brindis, MD, MPH, professor of medicine, University of California, San Francisco, countered that there were only three experienced operators from a single center and that the stroke incidence was physician reported, “not data we can really embrace.”
There was much debate over why enrollment in phase 2 of the RHYTHM trial was temporarily paused in February 2019, briefly restarted, and then prematurely stopped in April 2019.
FDA officials said the study was paused at the recommendation of the data monitoring committee (DMC) because rates of safety events were different between patients and control subjects and operational errors called into question the accuracy of the data being reviewed. Ultimately, both the DMC and FDA recommended study suspension.
During the public hearing, TAVR pioneer Alain Cribier, MD, University of Rouen’s Charles Nicolle Hospital, Mont-Saint-Aignan, France, said the TriGuard 3 is of interest because it can be used with minimal need for manipulation and complete coverage of the cerebral vessels that is achieved by diverting rather than capturing debris. “The rapid and exponential growth of TAVR procedures demands safe TAVR interventions and the use of cerebral protection devices is a step in this direction.”
Others took a dim view. “Given that the Sentinel device has not demonstrated benefit on clinical outcomes, there is significant concern about similar devices, such as the TriGuard 3, providing clinical benefit,” Rita Redberg, MD, Sanket Dhruva, MD, and Robin Ji, University of California, San Francisco, wrote in a letter submitted to the panel.
Commenting further, they added: “With the results from the REFLECT II trial demonstrating no evidence for clinical outcome benefit in TAVR patients, and numerically higher rates for stroke risk, mortality, bleeding risk, and other dangerous adverse complications among those treated, it is concerning and dangerous for patient safety that the TriGUARD 3 cerebral embolic protection device is being considered for FDA 510(k) clearance.”
The FDA panel members reported no financial relationships.
A version of this article first appeared on Medscape.com.
A Food and Drug Administration advisory panel struggled to muster support for marketing clearance of the TriGuard 3 (Keystone Heart) device for use during transcatheter aortic valve replacement (TAVR).
The Circulatory Systems Devices Panel of the Medical Devices Advisory Committee took no vote when it met Aug. 3, but weighed evidence for a proposed indication for the device “to minimize the risk of cerebral damage by deflecting embolic debris away from the cerebral circulation” during TAVR.
“While this device may deflect some debris, the data would suggest it may also create issues,” said Keith B. Allen, MD, director of surgical research at the Mid America Heart & Lung Surgeons, Kansas City, Mo. “I am really concerned that our desire and the emotion that surrounds preventing stroke are not being supported by the data.”
TriGuard 3 received CE Mark in Europe in March 2020. It was submitted for 510(k) clearance and seeks to prove substantial equivalence to the predicate Sentinel device (Claret Medical), currently the only approved embolic protection device in the United States.
The device is designed to cover all three major aortic vessels (innominate, left carotid, and left subclavian arteries) and is delivered transfemorally through an 8F sheath, whereas the Sentinel is positioned within the branch vessels, doesn’t cover the left subclavian artery, and is introduced through the radial or brachial artery via a 6F sheath.
TriGuard 3 faced an uphill battle, however, after failing to meet the primary composite efficacy endpoint in the REFLECT phase 2 trial (P = .857), with numeric trends showing higher all-cause mortality or any stroke at 30 days (9.8% vs. 6.7%) than pooled control subjects without embolic protection.
Rates for other components of the endpoint also trended higher with the device: National Institutes of Stroke Stroke Scale score worsening 2-5 days after the procedure, cerebral ischemic lesions on MRI 2-5 days after the procedure, and total cerebral ischemic lesion volume.
The Sentinel device was approved in 2017 after it failed to meet its primary efficacy endpoint of new brain lesion volume on MRI, but death and stroke rates favored the device over control, the panel pointed out.
The sponsor provided additional analyses in the per treatment (PT) population, defined as those with complete three-vessel coverage in at least two of three procedural time points. Compared with pooled control subjects, most of the imaging endpoints favored the TriGuard 3 device, but clinical neurologic event rates continued to favor the control group.
“The data used to demonstrate efficacy are all based on the PT subpopulation of the whole population, and those have to be considered promissory data,” said John Hirshfeld, MD, emeritus professor, University of Pennsylvania, Philadelphia. “This is the group where everything went well and for us to decide that’s achievable in the general population is speculative.”
Safety data
The REFLECT trial did meet its primary safety endpoint, with a 30-day major adverse cardiovascular event rate of 15.9%, compared with a performance goal of 34.4% (P < .0001).
Although prespecified, panel members pushed back, saying that the performance goal was unacceptably high, with several members remarking they’d never heard of a trial adding 9% as a “fudge factor” to a 25% historic control rate to get to the 34% performance target.
Keystone health officials noted that REFLECT was not designed to demonstrate a significant difference in the rate of primary safety events, compared with control. Instead, its purpose was to demonstrate that TriGuard 3 did not increase the risk associated with a TAVR procedure.
The TriGuard 3 device was successfully placed and retrieved in 100% of patients, but complete coverage was not uniform, with 72% of 157 as-treated patients having complete three-vessel coverage post TAVR but 15% having no coverage.
Panel members also expressed concern over device interference during TAVR, which was reported in nearly 10% of all TriGuard patients.
The TriGuard 3 group had 11 major vascular complications, 2 directly related to the device, and 3 stage 3 acute kidney injuries, whereas neither complication occurred in the control group.
Throughout the 9-hour hearing, the panel wrestled with what was described as a highly select patient group and small patient numbers that made it difficult to interpret observed differences. The trial involved 157 TriGuard 3 patients (including 41 from the roll-in phase) and 119 control subjects pooled from phase 2 of the trial (n = 57) and from phase 1 using the early-stage TriGuard HDH device (n = 57).
Pieter Stella, MD, PhD, Utrecht (the Netherlands) Medical Center, also presented “real-world” evidence from 75 patients in the Netherlands using the latest iteration of the device available in Europe with updates to the crimper and additional training materials to prevent the device from torquing during delivery. No strokes were reported, one patient had a transient ischemic attack (TIA), and two patients had a dissection, which resolved without sequelae.
Ralph Brindis, MD, MPH, professor of medicine, University of California, San Francisco, countered that there were only three experienced operators from a single center and that the stroke incidence was physician reported, “not data we can really embrace.”
There was much debate over why enrollment in phase 2 of the RHYTHM trial was temporarily paused in February 2019, briefly restarted, and then prematurely stopped in April 2019.
FDA officials said the study was paused at the recommendation of the data monitoring committee (DMC) because rates of safety events were different between patients and control subjects and operational errors called into question the accuracy of the data being reviewed. Ultimately, both the DMC and FDA recommended study suspension.
During the public hearing, TAVR pioneer Alain Cribier, MD, University of Rouen’s Charles Nicolle Hospital, Mont-Saint-Aignan, France, said the TriGuard 3 is of interest because it can be used with minimal need for manipulation and complete coverage of the cerebral vessels that is achieved by diverting rather than capturing debris. “The rapid and exponential growth of TAVR procedures demands safe TAVR interventions and the use of cerebral protection devices is a step in this direction.”
Others took a dim view. “Given that the Sentinel device has not demonstrated benefit on clinical outcomes, there is significant concern about similar devices, such as the TriGuard 3, providing clinical benefit,” Rita Redberg, MD, Sanket Dhruva, MD, and Robin Ji, University of California, San Francisco, wrote in a letter submitted to the panel.
Commenting further, they added: “With the results from the REFLECT II trial demonstrating no evidence for clinical outcome benefit in TAVR patients, and numerically higher rates for stroke risk, mortality, bleeding risk, and other dangerous adverse complications among those treated, it is concerning and dangerous for patient safety that the TriGUARD 3 cerebral embolic protection device is being considered for FDA 510(k) clearance.”
The FDA panel members reported no financial relationships.
A version of this article first appeared on Medscape.com.
Intracranial atherosclerosis finding on MRA linked to stroke
An incidental diagnosis of intracranial atherosclerotic stenosis in stroke-free individuals should trigger a thorough assessment of vascular health, according to the authors of a study identifying risk factors and vascular event risk in asymptomatic ICAS.
That conclusion emerged from data collected on more than 1,000 stroke-free participants in NOMAS (Northern Manhattan Study), a trial that prospectively followed participants who underwent a brain magnetic resonance angiogram (MRA) during 2003-2008.
In ICAS patients with stenosis of at least 70%, even with aggressive medical therapy, the annual stroke recurrence rate is 10%-20% in those with occlusions and at least three or more vascular risk factors. This high rate of recurrent vascular events in patients with stroke caused by ICAS warrants greater focus on primary prevention and targeted interventions for stroke-free individuals at highest risk for ICAS-related events, the investigators concluded.
Identify high-risk ICAS
Using NOMAS data, the investigators, led by Jose Gutierrez, MD, MPH, tested the hypothesis that stroke-free subjects at high risk of stroke and vascular events could be identified through the presence of asymptomatic ICAS. NOMAS is an ongoing, population-based epidemiologic study among randomly selected people with home telephones living in northern Manhattan.
During 2003-2008, investigators invited participants who were at least 50 years old, stroke free, and without contraindications to undergo brain MRA. The 1,211 study members were followed annually via telephone and in-person adjudication of events. A control group of 79 patients with no MRA was also identified with similar rates of hypertension, diabetes, hypercholesterolemia and current smoking.
Mean age was about 71 years (59% female, 65% Hispanic, 45% any stenosis). At the time of MRA, 78% had hypertension, 25% had diabetes, 81% had hypercholesterolemia, and 11% were current smokers.
Researchers rated stenoses in 11 brain arteries as 0, with no stenosis; 1, with less than 50% stenosis or luminal irregularities; 2, 50%-69% stenosis; and 3, at least 70% stenosis or flow gap. Outcomes included vascular death, myocardial infarction, ischemic stroke, cardioembolic stroke, intracranial artery disease stroke (which combined intracranial small and large artery disease strokes), and any vascular events (defined as a composite of vascular death, any stroke, or MI).
Greater stenosis denotes higher risk
Analysis found ICAS to be associated with older age (odds ratio, 1.02 per year; 95% confidence interval, 1.01-1.04), hypertension duration (OR, 1.01 per year; 95% CI, 1.00-1.02), higher number of glucose-lowering drugs (OR, 1.64 per each medication; 95% CI, 1.24-2.15), and HDL cholesterol(OR, 0.96 per mg/dL; 95% CI, 0.92-0.99). Event risk was greater among participants with ICAS of at least 70% (5.5% annual risk of vascular events; HR, 2.1; 95% CI, 1.4-3.2; compared with those with no ICAS), the investigators reported in the Journal of the American College of Cardiology.
Furthermore, 80% of incident strokes initially classified as small artery disease occurred among individuals with evidence of any degree of ICAS at their baseline MRI, the investigators noted. They found also that individuals with ICAS who had a primary care physician at the time of their initial MRI had a lower risk of events. Frequent primary care visits, they observed, might imply greater control of risk factors and other unmeasured confounders, such as health literacy, health care trust, access, and availability.
Incidental ICAS should trigger vascular assessment
An incidental diagnosis of ICAS in stroke-free subjects should trigger a thorough assessment of vascular health, the investigators concluded. They commented also that prophylaxis of first-ever stroke at this asymptomatic stage “may magnify the societal benefits of vascular prevention and decrease stroke-related disability and vascular death in our communities.”
“The big gap in our knowledge,” Tanya N. Turan, MD, professor of neurology at Medical University of South Carolina, Charleston, wrote in an accompanying editorial “is understanding the pathophysiological triggers for an asymptomatic stenosis to become a high-risk symptomatic stenosis. Until that question is answered, screening for asymptomatic ICAS is unlikely to change management among patients with known vascular risk factors.” In an interview, she observed further that “MRI plaque imaging could be a useful research tool to see if certain plaque features in an asymptomatic lesion are high risk for causing stroke. If that were proven, then it would make more sense to screen for ICAS and develop specific therapeutic strategies targeting high-risk asymptomatic plaque.”
Focus on recurrent stroke misplaced
Dr. Gutierrez said in an interview: “In the stroke world, most of what we do focuses on preventing recurrent stroke. Nonetheless, three-fourths of strokes in this country are new strokes, so to me it doesn’t make much sense to spend most of our efforts and attention to prevent the smallest fractions of strokes that occur in our society.”
He stressed that “the first immediate application of our results is that if people having a brain MRA for other reasons are found to have incidental, and therefore asymptomatic, ICAS, then they should be aggressively treated for vascular risk factors.” Secondly, “we hope to identify the patients at the highest risk of prevalent ICAS before they have a stroke. Among them, a brain MRI/MRA evaluating the phenotype would determine how aggressively to treat LDL.”
Dr. Gutierrez, professor of neurology at Columbia University Irving Medical Center, New York, noted that educating patients of their underlying high risk of events may have the effect of engaging them more in their own care. “There is evidence that actually showing people scans increases compliance and health literacy. It’s not yet standard of care, but we hope our future projects will help advance the field in the primary prevention direction,” he said.
This work was supported by the National Institutes of Health. The authors reported that they had no relevant financial disclosures.
An incidental diagnosis of intracranial atherosclerotic stenosis in stroke-free individuals should trigger a thorough assessment of vascular health, according to the authors of a study identifying risk factors and vascular event risk in asymptomatic ICAS.
That conclusion emerged from data collected on more than 1,000 stroke-free participants in NOMAS (Northern Manhattan Study), a trial that prospectively followed participants who underwent a brain magnetic resonance angiogram (MRA) during 2003-2008.
In ICAS patients with stenosis of at least 70%, even with aggressive medical therapy, the annual stroke recurrence rate is 10%-20% in those with occlusions and at least three or more vascular risk factors. This high rate of recurrent vascular events in patients with stroke caused by ICAS warrants greater focus on primary prevention and targeted interventions for stroke-free individuals at highest risk for ICAS-related events, the investigators concluded.
Identify high-risk ICAS
Using NOMAS data, the investigators, led by Jose Gutierrez, MD, MPH, tested the hypothesis that stroke-free subjects at high risk of stroke and vascular events could be identified through the presence of asymptomatic ICAS. NOMAS is an ongoing, population-based epidemiologic study among randomly selected people with home telephones living in northern Manhattan.
During 2003-2008, investigators invited participants who were at least 50 years old, stroke free, and without contraindications to undergo brain MRA. The 1,211 study members were followed annually via telephone and in-person adjudication of events. A control group of 79 patients with no MRA was also identified with similar rates of hypertension, diabetes, hypercholesterolemia and current smoking.
Mean age was about 71 years (59% female, 65% Hispanic, 45% any stenosis). At the time of MRA, 78% had hypertension, 25% had diabetes, 81% had hypercholesterolemia, and 11% were current smokers.
Researchers rated stenoses in 11 brain arteries as 0, with no stenosis; 1, with less than 50% stenosis or luminal irregularities; 2, 50%-69% stenosis; and 3, at least 70% stenosis or flow gap. Outcomes included vascular death, myocardial infarction, ischemic stroke, cardioembolic stroke, intracranial artery disease stroke (which combined intracranial small and large artery disease strokes), and any vascular events (defined as a composite of vascular death, any stroke, or MI).
Greater stenosis denotes higher risk
Analysis found ICAS to be associated with older age (odds ratio, 1.02 per year; 95% confidence interval, 1.01-1.04), hypertension duration (OR, 1.01 per year; 95% CI, 1.00-1.02), higher number of glucose-lowering drugs (OR, 1.64 per each medication; 95% CI, 1.24-2.15), and HDL cholesterol(OR, 0.96 per mg/dL; 95% CI, 0.92-0.99). Event risk was greater among participants with ICAS of at least 70% (5.5% annual risk of vascular events; HR, 2.1; 95% CI, 1.4-3.2; compared with those with no ICAS), the investigators reported in the Journal of the American College of Cardiology.
Furthermore, 80% of incident strokes initially classified as small artery disease occurred among individuals with evidence of any degree of ICAS at their baseline MRI, the investigators noted. They found also that individuals with ICAS who had a primary care physician at the time of their initial MRI had a lower risk of events. Frequent primary care visits, they observed, might imply greater control of risk factors and other unmeasured confounders, such as health literacy, health care trust, access, and availability.
Incidental ICAS should trigger vascular assessment
An incidental diagnosis of ICAS in stroke-free subjects should trigger a thorough assessment of vascular health, the investigators concluded. They commented also that prophylaxis of first-ever stroke at this asymptomatic stage “may magnify the societal benefits of vascular prevention and decrease stroke-related disability and vascular death in our communities.”
“The big gap in our knowledge,” Tanya N. Turan, MD, professor of neurology at Medical University of South Carolina, Charleston, wrote in an accompanying editorial “is understanding the pathophysiological triggers for an asymptomatic stenosis to become a high-risk symptomatic stenosis. Until that question is answered, screening for asymptomatic ICAS is unlikely to change management among patients with known vascular risk factors.” In an interview, she observed further that “MRI plaque imaging could be a useful research tool to see if certain plaque features in an asymptomatic lesion are high risk for causing stroke. If that were proven, then it would make more sense to screen for ICAS and develop specific therapeutic strategies targeting high-risk asymptomatic plaque.”
Focus on recurrent stroke misplaced
Dr. Gutierrez said in an interview: “In the stroke world, most of what we do focuses on preventing recurrent stroke. Nonetheless, three-fourths of strokes in this country are new strokes, so to me it doesn’t make much sense to spend most of our efforts and attention to prevent the smallest fractions of strokes that occur in our society.”
He stressed that “the first immediate application of our results is that if people having a brain MRA for other reasons are found to have incidental, and therefore asymptomatic, ICAS, then they should be aggressively treated for vascular risk factors.” Secondly, “we hope to identify the patients at the highest risk of prevalent ICAS before they have a stroke. Among them, a brain MRI/MRA evaluating the phenotype would determine how aggressively to treat LDL.”
Dr. Gutierrez, professor of neurology at Columbia University Irving Medical Center, New York, noted that educating patients of their underlying high risk of events may have the effect of engaging them more in their own care. “There is evidence that actually showing people scans increases compliance and health literacy. It’s not yet standard of care, but we hope our future projects will help advance the field in the primary prevention direction,” he said.
This work was supported by the National Institutes of Health. The authors reported that they had no relevant financial disclosures.
An incidental diagnosis of intracranial atherosclerotic stenosis in stroke-free individuals should trigger a thorough assessment of vascular health, according to the authors of a study identifying risk factors and vascular event risk in asymptomatic ICAS.
That conclusion emerged from data collected on more than 1,000 stroke-free participants in NOMAS (Northern Manhattan Study), a trial that prospectively followed participants who underwent a brain magnetic resonance angiogram (MRA) during 2003-2008.
In ICAS patients with stenosis of at least 70%, even with aggressive medical therapy, the annual stroke recurrence rate is 10%-20% in those with occlusions and at least three or more vascular risk factors. This high rate of recurrent vascular events in patients with stroke caused by ICAS warrants greater focus on primary prevention and targeted interventions for stroke-free individuals at highest risk for ICAS-related events, the investigators concluded.
Identify high-risk ICAS
Using NOMAS data, the investigators, led by Jose Gutierrez, MD, MPH, tested the hypothesis that stroke-free subjects at high risk of stroke and vascular events could be identified through the presence of asymptomatic ICAS. NOMAS is an ongoing, population-based epidemiologic study among randomly selected people with home telephones living in northern Manhattan.
During 2003-2008, investigators invited participants who were at least 50 years old, stroke free, and without contraindications to undergo brain MRA. The 1,211 study members were followed annually via telephone and in-person adjudication of events. A control group of 79 patients with no MRA was also identified with similar rates of hypertension, diabetes, hypercholesterolemia and current smoking.
Mean age was about 71 years (59% female, 65% Hispanic, 45% any stenosis). At the time of MRA, 78% had hypertension, 25% had diabetes, 81% had hypercholesterolemia, and 11% were current smokers.
Researchers rated stenoses in 11 brain arteries as 0, with no stenosis; 1, with less than 50% stenosis or luminal irregularities; 2, 50%-69% stenosis; and 3, at least 70% stenosis or flow gap. Outcomes included vascular death, myocardial infarction, ischemic stroke, cardioembolic stroke, intracranial artery disease stroke (which combined intracranial small and large artery disease strokes), and any vascular events (defined as a composite of vascular death, any stroke, or MI).
Greater stenosis denotes higher risk
Analysis found ICAS to be associated with older age (odds ratio, 1.02 per year; 95% confidence interval, 1.01-1.04), hypertension duration (OR, 1.01 per year; 95% CI, 1.00-1.02), higher number of glucose-lowering drugs (OR, 1.64 per each medication; 95% CI, 1.24-2.15), and HDL cholesterol(OR, 0.96 per mg/dL; 95% CI, 0.92-0.99). Event risk was greater among participants with ICAS of at least 70% (5.5% annual risk of vascular events; HR, 2.1; 95% CI, 1.4-3.2; compared with those with no ICAS), the investigators reported in the Journal of the American College of Cardiology.
Furthermore, 80% of incident strokes initially classified as small artery disease occurred among individuals with evidence of any degree of ICAS at their baseline MRI, the investigators noted. They found also that individuals with ICAS who had a primary care physician at the time of their initial MRI had a lower risk of events. Frequent primary care visits, they observed, might imply greater control of risk factors and other unmeasured confounders, such as health literacy, health care trust, access, and availability.
Incidental ICAS should trigger vascular assessment
An incidental diagnosis of ICAS in stroke-free subjects should trigger a thorough assessment of vascular health, the investigators concluded. They commented also that prophylaxis of first-ever stroke at this asymptomatic stage “may magnify the societal benefits of vascular prevention and decrease stroke-related disability and vascular death in our communities.”
“The big gap in our knowledge,” Tanya N. Turan, MD, professor of neurology at Medical University of South Carolina, Charleston, wrote in an accompanying editorial “is understanding the pathophysiological triggers for an asymptomatic stenosis to become a high-risk symptomatic stenosis. Until that question is answered, screening for asymptomatic ICAS is unlikely to change management among patients with known vascular risk factors.” In an interview, she observed further that “MRI plaque imaging could be a useful research tool to see if certain plaque features in an asymptomatic lesion are high risk for causing stroke. If that were proven, then it would make more sense to screen for ICAS and develop specific therapeutic strategies targeting high-risk asymptomatic plaque.”
Focus on recurrent stroke misplaced
Dr. Gutierrez said in an interview: “In the stroke world, most of what we do focuses on preventing recurrent stroke. Nonetheless, three-fourths of strokes in this country are new strokes, so to me it doesn’t make much sense to spend most of our efforts and attention to prevent the smallest fractions of strokes that occur in our society.”
He stressed that “the first immediate application of our results is that if people having a brain MRA for other reasons are found to have incidental, and therefore asymptomatic, ICAS, then they should be aggressively treated for vascular risk factors.” Secondly, “we hope to identify the patients at the highest risk of prevalent ICAS before they have a stroke. Among them, a brain MRI/MRA evaluating the phenotype would determine how aggressively to treat LDL.”
Dr. Gutierrez, professor of neurology at Columbia University Irving Medical Center, New York, noted that educating patients of their underlying high risk of events may have the effect of engaging them more in their own care. “There is evidence that actually showing people scans increases compliance and health literacy. It’s not yet standard of care, but we hope our future projects will help advance the field in the primary prevention direction,” he said.
This work was supported by the National Institutes of Health. The authors reported that they had no relevant financial disclosures.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
DOACs best aspirin after ventricular ablation: STROKE-VT
Catheter ablation has been around a lot longer for ventricular arrhythmia than for atrial fibrillation, but far less is settled about what antithrombotic therapy should follow ventricular ablations, as there have been no big, randomized trials for guidance.
But the evidence base grew stronger this week, and it favors postprocedure treatment with a direct oral anticoagulant (DOAC) over antiplatelet therapy with aspirin for patients undergoing radiofrequency (RF) ablation to treat left ventricular (LV) arrhythmias.
The 30-day risk for ischemic stroke or transient ischemia attack (TIA) was sharply higher for patients who took daily aspirin after RF ablation for ventricular tachycardia (VT) or premature ventricular contractions (PVC) in a multicenter randomized trial.
Those of its 246 patients who received aspirin were also far more likely to show asymptomatic lesions on cerebral MRI scans performed both 24 hours and 30 days after the procedure.
The findings show the importance of DOAC therapy after ventricular ablation procedures, a setting for which there are no evidence-based guidelines, “to mitigate the risk of systemic thromboembolic events,” said Dhanunjaya Lakkireddy, MD, Kansas City Heart Rhythm Institute, Overland Park. He spoke at a media presentation on the trial, called STROKE-VT, during the Heart Rhythm Society 2021 Scientific Sessions, held virtually and on-site in Boston.
The risk for stroke and TIA went up in association with several procedural issues, including some that operators might be able to change in order to reach for better outcomes, Dr. Lakkireddy observed.
“Prolonged radiofrequency ablation times, especially in those with low left ventricle ejection fractions, are definitely higher risk,” as are procedures that involved the retrograde transaortic approach for advancing the ablation catheter, rather than a trans-septal approach.
The retrograde transaortic approach should be avoided in such procedures, “whenever it can be avoided,” said Dr. Lakkireddy, who formally presented STROKE-VT at the HRS sessions and is lead author on its report published about the same time in JACC: Clinical Electrophysiology.
The trial has limitations, but “it’s a very important study, and I think that this could become our standard of care for managing anticoagulation after VT and PVC left-sided ablations,” Mina K. Chung, MD, Cleveland Clinic, said as an invited discussant after Dr. Lakkireddy’s presentation.
How patients are treated with antithrombotics after ventricular ablations can vary widely, sometimes based on the operator’s “subjective feeling of how extensive the ablation is,” Christine M. Albert, MD, MPH, Cedars-Sinai Medical Center, Los Angeles, not involved in the study, said during the STROKE-VT media briefing.
That’s consistent with the guidelines, which propose oral anticoagulation therapy after more extensive ventricular ablations and antiplatelets when the ablation is more limited – based more on consensus than firm evidence – as described by Jeffrey R. Winterfield, MD, Medical University of South Carolina, Charleston, and Usha Tedrow, MD, MSc, Brigham and Women’s Hospital, Boston, in an accompanying editorial.
“This is really the first randomized trial data, that I know of, that we have on this. So I do think it will be guideline-influencing,” Dr. Albert said.
“This should change practice,” agreed Jonathan P. Piccini, MD, MHS, Duke University, Durham, N.C., also not part of STROKE-VT. “A lot of evidence in the trial is consistent and provides a compelling story, not to mention that, in my opinion, the study probably underestimates the value of DOACs,” he told this news organization.
That’s because patients assigned to DOACs had far longer ablation times, “so their risk was even greater than in the aspirin arm,” Dr. Piccini said. Ablation times averaged 2,095 seconds in the DOAC group, compared with only 1,708 seconds in the aspirin group, probably because the preponderance of VT over PVC ablations for those getting a DOAC was even greater in the aspirin group.
Of the 246 patients assigned to either aspirin or a DOAC, usually a factor Xa inhibitor, 75% had undergone VT ablation and the remainder ablation for PVCs. Their mean age was 60 years and only 18% were women. None had experienced a cerebrovascular event in the previous 3 months.
The 30-day odds ratio for TIA or ischemic stroke in patients who received aspirin, compared with a DOAC, was 12.6 (95% confidence interval, 4.10-39.11; P < .001).
The corresponding OR for asymptomatic cerebral lesions by MRI at 24 hours was 2.15 (95% CI, 1.02-4.54; P = .04) and at 30 days was 3.48 (95% CI, 1.38-8.80; P = .008).
The rate of stroke or TIA was similar in patients who underwent ablation for VT and for PVCs (14% vs. 16%, respectively; P = .70). There were fewer asymptomatic cerebrovascular events by MRI at 24 hours for those undergoing VT ablations (14.7% and 25.8%, respectively; P = .046); but difference between rates attenuated by 30 days (11.4% and 14.5%, respectively; P = .52).
The OR for TIA or stroke associated with the retrograde transaortic approach, performed in about 40% of the patients, compared with the trans-septal approach in the remainder was 2.60 (95% CI, 1.06-6.37; P = .04).
“The study tells us it’s safe and indeed preferable to anticoagulate after an ablation procedure. But the more important finding, perhaps, wasn’t the one related to the core hypothesis. And that was the effect of retrograde access,” Paul A. Friedman, MD, Mayo Clinic, Rochester, Minn., said as an invited discussant after Dr. Lakkireddy’s formal presentation of the trial.
Whether a ventricular ablation is performed using the retrograde transaortic or trans-septal approach often depends on the location of the ablation targets in the left ventricle. But in some cases it’s a matter of operator preference, Dr. Piccini observed.
“There are some situations where, really, it is better to do retrograde aortic, and there are some cases that are better to do trans-septal. But now there’s going to be a higher burden of proof,” he said. Given the findings of STROKE-VT, operators may need to consider that a ventricular ablation procedure that can be done by the trans-septal route perhaps ought to be consistently done that way.
Dr. Lakkireddy discloses financial relationships with Boston Scientific, Biosense Webster, Janssen Pharmaceuticals, and more. Dr. Chung had “nothing relevant to disclose.” Dr. Piccini discloses receiving honoraria or speaking or consulting fees from Sanofi, Abbott, ARCA Biopharma, Medtronic, Philips, Biotronik, Allergan, LivaNova, and Myokardia; and research in conjunction with Bayer Healthcare, Abbott, Boston Scientific, and Philips. Dr. Friedman discloses conducting research in conjunction with Medtronic and Abbott; holding intellectual property rights with AliveCor, Inference, Medicool, Eko, and Anumana; and receiving honoraria or speaking or consulting fees from Boston Scientific. Dr. Winterfield and Dr. Tedrow had no disclosures.
A version of this article first appeared on Medscape.com.
Catheter ablation has been around a lot longer for ventricular arrhythmia than for atrial fibrillation, but far less is settled about what antithrombotic therapy should follow ventricular ablations, as there have been no big, randomized trials for guidance.
But the evidence base grew stronger this week, and it favors postprocedure treatment with a direct oral anticoagulant (DOAC) over antiplatelet therapy with aspirin for patients undergoing radiofrequency (RF) ablation to treat left ventricular (LV) arrhythmias.
The 30-day risk for ischemic stroke or transient ischemia attack (TIA) was sharply higher for patients who took daily aspirin after RF ablation for ventricular tachycardia (VT) or premature ventricular contractions (PVC) in a multicenter randomized trial.
Those of its 246 patients who received aspirin were also far more likely to show asymptomatic lesions on cerebral MRI scans performed both 24 hours and 30 days after the procedure.
The findings show the importance of DOAC therapy after ventricular ablation procedures, a setting for which there are no evidence-based guidelines, “to mitigate the risk of systemic thromboembolic events,” said Dhanunjaya Lakkireddy, MD, Kansas City Heart Rhythm Institute, Overland Park. He spoke at a media presentation on the trial, called STROKE-VT, during the Heart Rhythm Society 2021 Scientific Sessions, held virtually and on-site in Boston.
The risk for stroke and TIA went up in association with several procedural issues, including some that operators might be able to change in order to reach for better outcomes, Dr. Lakkireddy observed.
“Prolonged radiofrequency ablation times, especially in those with low left ventricle ejection fractions, are definitely higher risk,” as are procedures that involved the retrograde transaortic approach for advancing the ablation catheter, rather than a trans-septal approach.
The retrograde transaortic approach should be avoided in such procedures, “whenever it can be avoided,” said Dr. Lakkireddy, who formally presented STROKE-VT at the HRS sessions and is lead author on its report published about the same time in JACC: Clinical Electrophysiology.
The trial has limitations, but “it’s a very important study, and I think that this could become our standard of care for managing anticoagulation after VT and PVC left-sided ablations,” Mina K. Chung, MD, Cleveland Clinic, said as an invited discussant after Dr. Lakkireddy’s presentation.
How patients are treated with antithrombotics after ventricular ablations can vary widely, sometimes based on the operator’s “subjective feeling of how extensive the ablation is,” Christine M. Albert, MD, MPH, Cedars-Sinai Medical Center, Los Angeles, not involved in the study, said during the STROKE-VT media briefing.
That’s consistent with the guidelines, which propose oral anticoagulation therapy after more extensive ventricular ablations and antiplatelets when the ablation is more limited – based more on consensus than firm evidence – as described by Jeffrey R. Winterfield, MD, Medical University of South Carolina, Charleston, and Usha Tedrow, MD, MSc, Brigham and Women’s Hospital, Boston, in an accompanying editorial.
“This is really the first randomized trial data, that I know of, that we have on this. So I do think it will be guideline-influencing,” Dr. Albert said.
“This should change practice,” agreed Jonathan P. Piccini, MD, MHS, Duke University, Durham, N.C., also not part of STROKE-VT. “A lot of evidence in the trial is consistent and provides a compelling story, not to mention that, in my opinion, the study probably underestimates the value of DOACs,” he told this news organization.
That’s because patients assigned to DOACs had far longer ablation times, “so their risk was even greater than in the aspirin arm,” Dr. Piccini said. Ablation times averaged 2,095 seconds in the DOAC group, compared with only 1,708 seconds in the aspirin group, probably because the preponderance of VT over PVC ablations for those getting a DOAC was even greater in the aspirin group.
Of the 246 patients assigned to either aspirin or a DOAC, usually a factor Xa inhibitor, 75% had undergone VT ablation and the remainder ablation for PVCs. Their mean age was 60 years and only 18% were women. None had experienced a cerebrovascular event in the previous 3 months.
The 30-day odds ratio for TIA or ischemic stroke in patients who received aspirin, compared with a DOAC, was 12.6 (95% confidence interval, 4.10-39.11; P < .001).
The corresponding OR for asymptomatic cerebral lesions by MRI at 24 hours was 2.15 (95% CI, 1.02-4.54; P = .04) and at 30 days was 3.48 (95% CI, 1.38-8.80; P = .008).
The rate of stroke or TIA was similar in patients who underwent ablation for VT and for PVCs (14% vs. 16%, respectively; P = .70). There were fewer asymptomatic cerebrovascular events by MRI at 24 hours for those undergoing VT ablations (14.7% and 25.8%, respectively; P = .046); but difference between rates attenuated by 30 days (11.4% and 14.5%, respectively; P = .52).
The OR for TIA or stroke associated with the retrograde transaortic approach, performed in about 40% of the patients, compared with the trans-septal approach in the remainder was 2.60 (95% CI, 1.06-6.37; P = .04).
“The study tells us it’s safe and indeed preferable to anticoagulate after an ablation procedure. But the more important finding, perhaps, wasn’t the one related to the core hypothesis. And that was the effect of retrograde access,” Paul A. Friedman, MD, Mayo Clinic, Rochester, Minn., said as an invited discussant after Dr. Lakkireddy’s formal presentation of the trial.
Whether a ventricular ablation is performed using the retrograde transaortic or trans-septal approach often depends on the location of the ablation targets in the left ventricle. But in some cases it’s a matter of operator preference, Dr. Piccini observed.
“There are some situations where, really, it is better to do retrograde aortic, and there are some cases that are better to do trans-septal. But now there’s going to be a higher burden of proof,” he said. Given the findings of STROKE-VT, operators may need to consider that a ventricular ablation procedure that can be done by the trans-septal route perhaps ought to be consistently done that way.
Dr. Lakkireddy discloses financial relationships with Boston Scientific, Biosense Webster, Janssen Pharmaceuticals, and more. Dr. Chung had “nothing relevant to disclose.” Dr. Piccini discloses receiving honoraria or speaking or consulting fees from Sanofi, Abbott, ARCA Biopharma, Medtronic, Philips, Biotronik, Allergan, LivaNova, and Myokardia; and research in conjunction with Bayer Healthcare, Abbott, Boston Scientific, and Philips. Dr. Friedman discloses conducting research in conjunction with Medtronic and Abbott; holding intellectual property rights with AliveCor, Inference, Medicool, Eko, and Anumana; and receiving honoraria or speaking or consulting fees from Boston Scientific. Dr. Winterfield and Dr. Tedrow had no disclosures.
A version of this article first appeared on Medscape.com.
Catheter ablation has been around a lot longer for ventricular arrhythmia than for atrial fibrillation, but far less is settled about what antithrombotic therapy should follow ventricular ablations, as there have been no big, randomized trials for guidance.
But the evidence base grew stronger this week, and it favors postprocedure treatment with a direct oral anticoagulant (DOAC) over antiplatelet therapy with aspirin for patients undergoing radiofrequency (RF) ablation to treat left ventricular (LV) arrhythmias.
The 30-day risk for ischemic stroke or transient ischemia attack (TIA) was sharply higher for patients who took daily aspirin after RF ablation for ventricular tachycardia (VT) or premature ventricular contractions (PVC) in a multicenter randomized trial.
Those of its 246 patients who received aspirin were also far more likely to show asymptomatic lesions on cerebral MRI scans performed both 24 hours and 30 days after the procedure.
The findings show the importance of DOAC therapy after ventricular ablation procedures, a setting for which there are no evidence-based guidelines, “to mitigate the risk of systemic thromboembolic events,” said Dhanunjaya Lakkireddy, MD, Kansas City Heart Rhythm Institute, Overland Park. He spoke at a media presentation on the trial, called STROKE-VT, during the Heart Rhythm Society 2021 Scientific Sessions, held virtually and on-site in Boston.
The risk for stroke and TIA went up in association with several procedural issues, including some that operators might be able to change in order to reach for better outcomes, Dr. Lakkireddy observed.
“Prolonged radiofrequency ablation times, especially in those with low left ventricle ejection fractions, are definitely higher risk,” as are procedures that involved the retrograde transaortic approach for advancing the ablation catheter, rather than a trans-septal approach.
The retrograde transaortic approach should be avoided in such procedures, “whenever it can be avoided,” said Dr. Lakkireddy, who formally presented STROKE-VT at the HRS sessions and is lead author on its report published about the same time in JACC: Clinical Electrophysiology.
The trial has limitations, but “it’s a very important study, and I think that this could become our standard of care for managing anticoagulation after VT and PVC left-sided ablations,” Mina K. Chung, MD, Cleveland Clinic, said as an invited discussant after Dr. Lakkireddy’s presentation.
How patients are treated with antithrombotics after ventricular ablations can vary widely, sometimes based on the operator’s “subjective feeling of how extensive the ablation is,” Christine M. Albert, MD, MPH, Cedars-Sinai Medical Center, Los Angeles, not involved in the study, said during the STROKE-VT media briefing.
That’s consistent with the guidelines, which propose oral anticoagulation therapy after more extensive ventricular ablations and antiplatelets when the ablation is more limited – based more on consensus than firm evidence – as described by Jeffrey R. Winterfield, MD, Medical University of South Carolina, Charleston, and Usha Tedrow, MD, MSc, Brigham and Women’s Hospital, Boston, in an accompanying editorial.
“This is really the first randomized trial data, that I know of, that we have on this. So I do think it will be guideline-influencing,” Dr. Albert said.
“This should change practice,” agreed Jonathan P. Piccini, MD, MHS, Duke University, Durham, N.C., also not part of STROKE-VT. “A lot of evidence in the trial is consistent and provides a compelling story, not to mention that, in my opinion, the study probably underestimates the value of DOACs,” he told this news organization.
That’s because patients assigned to DOACs had far longer ablation times, “so their risk was even greater than in the aspirin arm,” Dr. Piccini said. Ablation times averaged 2,095 seconds in the DOAC group, compared with only 1,708 seconds in the aspirin group, probably because the preponderance of VT over PVC ablations for those getting a DOAC was even greater in the aspirin group.
Of the 246 patients assigned to either aspirin or a DOAC, usually a factor Xa inhibitor, 75% had undergone VT ablation and the remainder ablation for PVCs. Their mean age was 60 years and only 18% were women. None had experienced a cerebrovascular event in the previous 3 months.
The 30-day odds ratio for TIA or ischemic stroke in patients who received aspirin, compared with a DOAC, was 12.6 (95% confidence interval, 4.10-39.11; P < .001).
The corresponding OR for asymptomatic cerebral lesions by MRI at 24 hours was 2.15 (95% CI, 1.02-4.54; P = .04) and at 30 days was 3.48 (95% CI, 1.38-8.80; P = .008).
The rate of stroke or TIA was similar in patients who underwent ablation for VT and for PVCs (14% vs. 16%, respectively; P = .70). There were fewer asymptomatic cerebrovascular events by MRI at 24 hours for those undergoing VT ablations (14.7% and 25.8%, respectively; P = .046); but difference between rates attenuated by 30 days (11.4% and 14.5%, respectively; P = .52).
The OR for TIA or stroke associated with the retrograde transaortic approach, performed in about 40% of the patients, compared with the trans-septal approach in the remainder was 2.60 (95% CI, 1.06-6.37; P = .04).
“The study tells us it’s safe and indeed preferable to anticoagulate after an ablation procedure. But the more important finding, perhaps, wasn’t the one related to the core hypothesis. And that was the effect of retrograde access,” Paul A. Friedman, MD, Mayo Clinic, Rochester, Minn., said as an invited discussant after Dr. Lakkireddy’s formal presentation of the trial.
Whether a ventricular ablation is performed using the retrograde transaortic or trans-septal approach often depends on the location of the ablation targets in the left ventricle. But in some cases it’s a matter of operator preference, Dr. Piccini observed.
“There are some situations where, really, it is better to do retrograde aortic, and there are some cases that are better to do trans-septal. But now there’s going to be a higher burden of proof,” he said. Given the findings of STROKE-VT, operators may need to consider that a ventricular ablation procedure that can be done by the trans-septal route perhaps ought to be consistently done that way.
Dr. Lakkireddy discloses financial relationships with Boston Scientific, Biosense Webster, Janssen Pharmaceuticals, and more. Dr. Chung had “nothing relevant to disclose.” Dr. Piccini discloses receiving honoraria or speaking or consulting fees from Sanofi, Abbott, ARCA Biopharma, Medtronic, Philips, Biotronik, Allergan, LivaNova, and Myokardia; and research in conjunction with Bayer Healthcare, Abbott, Boston Scientific, and Philips. Dr. Friedman discloses conducting research in conjunction with Medtronic and Abbott; holding intellectual property rights with AliveCor, Inference, Medicool, Eko, and Anumana; and receiving honoraria or speaking or consulting fees from Boston Scientific. Dr. Winterfield and Dr. Tedrow had no disclosures.
A version of this article first appeared on Medscape.com.