User login
Spondyloarthritides undergoing a renaissance, but challenges exist
Rheumatologists’ understanding of the spectrum of spondyloarthritis is undergoing an evolution that has revealed an underrecognition of the disease, particularly in women, but regulatory recognition of the spectrum of disease in the United States has yet to catch up with reality, according to Dr. Philip Mease.
More sophisticated imaging and laboratory techniques has meant that spondyloarthritis (SpA) is being discovered in up to twice as many people as originally thought, many of whom are women, said Dr. Mease, director of rheumatology research at Swedish Medical Center and clinical professor at the University of Washington, both in Seattle.
It’s not that SpA occurs as frequently in women as in men, but that it presents in a less-severe form with often little visible x-ray damage but with evidence of inflammation on MRI or in lab tests, he said at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
“These patients can be just as significantly impacted as patients with more ‘objective’ ankylosing spondylitis and can benefit as much from therapy such as anti-TNF [tumor necrosis factor] medications,” he said.
Evidence shows the prevalence of all spondyloarthritides in the United States is 0.346%-1.31%, compared with a rheumatoid arthritis prevalence of 0.6%-1.0%. Data from the German spondyloarthritis inception cohort has shown that the burden of disease in ankylosing spondylitis and nonradiographic axial SpA (nr-axSpA) is similar, Dr. Mease said.
The Assessment of SpondyloArthritis Society (ASAS) classification criteria (Ann Rheum Dis. 2009;68:777-83) have gone some way toward identifying patients earlier, he said, but the biggest controversy surrounding the management of nr-axSpA in the United States has been the Food and Drug Administration’s lack of recognition of the disease, making it difficult to treat patients early.
The FDA is concerned about the potential for misdiagnosis and inappropriate use of anti-TNFs for these noninflammatory conditions, he said. “They readily understand that patients who are MRI positive or CRP [C-reactive protein] elevated and x-ray negative exist and are significantly affected by their disease, but they are also aware that there can be similarity of symptomatology between these types of patients and patients with mechanical or degenerative spine disease and/or fibromyalgia.”
As such, there is a need for more information on the natural history of the disease from clinical registries and trials in order to gain more understanding on how to accurately diagnose patients, he said.
More education to U.S. physicians is also needed, he added, noting that this particular controversy is not present in other parts of the world, such as the European Union, where etanercept, adalimumab, and certolizumab pegol have been approved by the European Medicines Agency for the treatment of nr-axSpA.
“There is need for better understanding about this patient population and better education of health-care providers to get them to think about this condition and refer their patients to rheumatologists for evaluation and appropriate therapy,” he added.
Dr. Mease disclosed that he has received research grants, consultation fees, and/or speaker honoraria from AbbVie, Amgen, Biogen, Bristol-Myers Squibb, Celgene, Genentech, GlaxoSmithKline, Janssen, Lilly, Merck, Novartis, Pfizer, and UCB. Global Academy for Medical Education and this news organization are owned by the same parent company.
Rheumatologists’ understanding of the spectrum of spondyloarthritis is undergoing an evolution that has revealed an underrecognition of the disease, particularly in women, but regulatory recognition of the spectrum of disease in the United States has yet to catch up with reality, according to Dr. Philip Mease.
More sophisticated imaging and laboratory techniques has meant that spondyloarthritis (SpA) is being discovered in up to twice as many people as originally thought, many of whom are women, said Dr. Mease, director of rheumatology research at Swedish Medical Center and clinical professor at the University of Washington, both in Seattle.
It’s not that SpA occurs as frequently in women as in men, but that it presents in a less-severe form with often little visible x-ray damage but with evidence of inflammation on MRI or in lab tests, he said at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
“These patients can be just as significantly impacted as patients with more ‘objective’ ankylosing spondylitis and can benefit as much from therapy such as anti-TNF [tumor necrosis factor] medications,” he said.
Evidence shows the prevalence of all spondyloarthritides in the United States is 0.346%-1.31%, compared with a rheumatoid arthritis prevalence of 0.6%-1.0%. Data from the German spondyloarthritis inception cohort has shown that the burden of disease in ankylosing spondylitis and nonradiographic axial SpA (nr-axSpA) is similar, Dr. Mease said.
The Assessment of SpondyloArthritis Society (ASAS) classification criteria (Ann Rheum Dis. 2009;68:777-83) have gone some way toward identifying patients earlier, he said, but the biggest controversy surrounding the management of nr-axSpA in the United States has been the Food and Drug Administration’s lack of recognition of the disease, making it difficult to treat patients early.
The FDA is concerned about the potential for misdiagnosis and inappropriate use of anti-TNFs for these noninflammatory conditions, he said. “They readily understand that patients who are MRI positive or CRP [C-reactive protein] elevated and x-ray negative exist and are significantly affected by their disease, but they are also aware that there can be similarity of symptomatology between these types of patients and patients with mechanical or degenerative spine disease and/or fibromyalgia.”
As such, there is a need for more information on the natural history of the disease from clinical registries and trials in order to gain more understanding on how to accurately diagnose patients, he said.
More education to U.S. physicians is also needed, he added, noting that this particular controversy is not present in other parts of the world, such as the European Union, where etanercept, adalimumab, and certolizumab pegol have been approved by the European Medicines Agency for the treatment of nr-axSpA.
“There is need for better understanding about this patient population and better education of health-care providers to get them to think about this condition and refer their patients to rheumatologists for evaluation and appropriate therapy,” he added.
Dr. Mease disclosed that he has received research grants, consultation fees, and/or speaker honoraria from AbbVie, Amgen, Biogen, Bristol-Myers Squibb, Celgene, Genentech, GlaxoSmithKline, Janssen, Lilly, Merck, Novartis, Pfizer, and UCB. Global Academy for Medical Education and this news organization are owned by the same parent company.
Rheumatologists’ understanding of the spectrum of spondyloarthritis is undergoing an evolution that has revealed an underrecognition of the disease, particularly in women, but regulatory recognition of the spectrum of disease in the United States has yet to catch up with reality, according to Dr. Philip Mease.
More sophisticated imaging and laboratory techniques has meant that spondyloarthritis (SpA) is being discovered in up to twice as many people as originally thought, many of whom are women, said Dr. Mease, director of rheumatology research at Swedish Medical Center and clinical professor at the University of Washington, both in Seattle.
It’s not that SpA occurs as frequently in women as in men, but that it presents in a less-severe form with often little visible x-ray damage but with evidence of inflammation on MRI or in lab tests, he said at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
“These patients can be just as significantly impacted as patients with more ‘objective’ ankylosing spondylitis and can benefit as much from therapy such as anti-TNF [tumor necrosis factor] medications,” he said.
Evidence shows the prevalence of all spondyloarthritides in the United States is 0.346%-1.31%, compared with a rheumatoid arthritis prevalence of 0.6%-1.0%. Data from the German spondyloarthritis inception cohort has shown that the burden of disease in ankylosing spondylitis and nonradiographic axial SpA (nr-axSpA) is similar, Dr. Mease said.
The Assessment of SpondyloArthritis Society (ASAS) classification criteria (Ann Rheum Dis. 2009;68:777-83) have gone some way toward identifying patients earlier, he said, but the biggest controversy surrounding the management of nr-axSpA in the United States has been the Food and Drug Administration’s lack of recognition of the disease, making it difficult to treat patients early.
The FDA is concerned about the potential for misdiagnosis and inappropriate use of anti-TNFs for these noninflammatory conditions, he said. “They readily understand that patients who are MRI positive or CRP [C-reactive protein] elevated and x-ray negative exist and are significantly affected by their disease, but they are also aware that there can be similarity of symptomatology between these types of patients and patients with mechanical or degenerative spine disease and/or fibromyalgia.”
As such, there is a need for more information on the natural history of the disease from clinical registries and trials in order to gain more understanding on how to accurately diagnose patients, he said.
More education to U.S. physicians is also needed, he added, noting that this particular controversy is not present in other parts of the world, such as the European Union, where etanercept, adalimumab, and certolizumab pegol have been approved by the European Medicines Agency for the treatment of nr-axSpA.
“There is need for better understanding about this patient population and better education of health-care providers to get them to think about this condition and refer their patients to rheumatologists for evaluation and appropriate therapy,” he added.
Dr. Mease disclosed that he has received research grants, consultation fees, and/or speaker honoraria from AbbVie, Amgen, Biogen, Bristol-Myers Squibb, Celgene, Genentech, GlaxoSmithKline, Janssen, Lilly, Merck, Novartis, Pfizer, and UCB. Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE ANNUAL PERSPECTIVES IN RHEUMATIC DISEASES
WCD: Look for TNF inhibitor–induced psoriasis in kids
VANCOUVER – Tumor necrosis factor inhibitor–induced psoriasiform dermatitis can occur in pediatric patients after any length of treatment, with documented cases emerging after the very first dose and as late as 63 months into anti-TNF therapy, Dr. Amy S. Paller said at the World Congress of Dermatology.
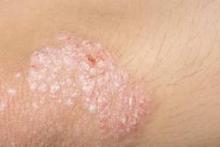
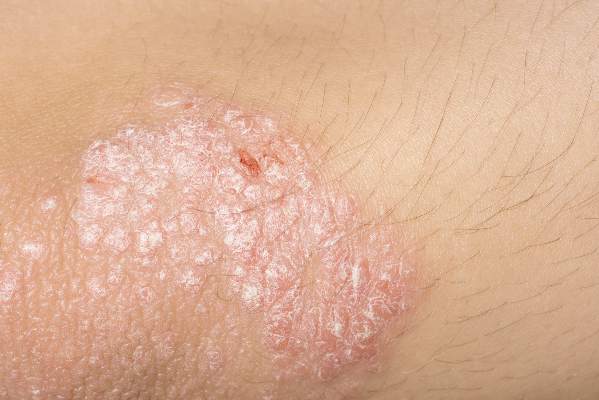
Histopathologically, this medication-induced condition is psoriasis. But it tends to follow a distinctive pattern, favoring the scalp, dorsal hands and feet, nails, and periorificial skin. Palmoplantar pustulosis is not uncommon. The lesions are often secondarily infected with Staphylococcus aureus, according to Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University in Chicago.
The phenomenon was first described in adults. But in the past several years, as the use of tumor necrosis factor (TNF) antagonists has gained increasing traction for treatment of pediatric inflammatory bowel disease and rheumatologic diseases, the dermatologic disorder has become better characterized in youths. In a retrospective study at McMaster Children’s Hospital in Hamilton, Ont., 17 of 172 (10%) infliximab-treated patients with Crohn’s disease developed new-onset psoriasis and another (0.6%) experienced worsening of preexisting psoriasis after anywhere from 1 to 25 infusions. Most patients responded well to topical steroids; however, three discontinued the biologic because of this complication (J Pediatr Gastroenterol Nutr. 2013 May;56[5]:512-8).
It’s now clear that the emergence of TNF inhibitor–induced psoriasis does not adversely affect the response of a patient’s inflammatory bowel disease or juvenile arthritis to the biologic. Also, the risk of recurrent psoriatic eruption is not reduced by concurrent methotrexate.
Researchers find TNF inhibitor–induced psoriasis to be an intriguing puzzle because of its paradoxical nature. After all, the TNF inhibitors are a highly effective treatment for moderate to severe plaque psoriasis. The leading theory as to the underlying basis for TNF inhibitor–induced psoriasis is that it may have a genetic basis, Dr. Paller noted.
The McMaster group found that their pediatric Crohn’s disease patients who developed psoriasis in conjunction with infliximab (Remicade) therapy were more likely than disease-matched controls to be homozygous for one of several specific polymorphisms in the interleukin-23R gene. And investigators at the University of Helsinki have reported that children with inflammatory bowel disease who developed psoriasiform dermatitis while on infliximab only rarely possessed the HLA-Cw*0602 genotype, which is commonly associated with psoriasis (Inflamm Bowel Dis. 2014 Aug;20[8]:1309-15).
The possibility of streptococcal or staphylococcal infection serving as a trigger for TNF inhibitor–induced psoriasis is also being explored, according to Dr. Paller.
She has received research funding from LEO Pharma and Amgen and serves as a consultant to AbbVie.
VANCOUVER – Tumor necrosis factor inhibitor–induced psoriasiform dermatitis can occur in pediatric patients after any length of treatment, with documented cases emerging after the very first dose and as late as 63 months into anti-TNF therapy, Dr. Amy S. Paller said at the World Congress of Dermatology.
Histopathologically, this medication-induced condition is psoriasis. But it tends to follow a distinctive pattern, favoring the scalp, dorsal hands and feet, nails, and periorificial skin. Palmoplantar pustulosis is not uncommon. The lesions are often secondarily infected with Staphylococcus aureus, according to Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University in Chicago.
The phenomenon was first described in adults. But in the past several years, as the use of tumor necrosis factor (TNF) antagonists has gained increasing traction for treatment of pediatric inflammatory bowel disease and rheumatologic diseases, the dermatologic disorder has become better characterized in youths. In a retrospective study at McMaster Children’s Hospital in Hamilton, Ont., 17 of 172 (10%) infliximab-treated patients with Crohn’s disease developed new-onset psoriasis and another (0.6%) experienced worsening of preexisting psoriasis after anywhere from 1 to 25 infusions. Most patients responded well to topical steroids; however, three discontinued the biologic because of this complication (J Pediatr Gastroenterol Nutr. 2013 May;56[5]:512-8).
It’s now clear that the emergence of TNF inhibitor–induced psoriasis does not adversely affect the response of a patient’s inflammatory bowel disease or juvenile arthritis to the biologic. Also, the risk of recurrent psoriatic eruption is not reduced by concurrent methotrexate.
Researchers find TNF inhibitor–induced psoriasis to be an intriguing puzzle because of its paradoxical nature. After all, the TNF inhibitors are a highly effective treatment for moderate to severe plaque psoriasis. The leading theory as to the underlying basis for TNF inhibitor–induced psoriasis is that it may have a genetic basis, Dr. Paller noted.
The McMaster group found that their pediatric Crohn’s disease patients who developed psoriasis in conjunction with infliximab (Remicade) therapy were more likely than disease-matched controls to be homozygous for one of several specific polymorphisms in the interleukin-23R gene. And investigators at the University of Helsinki have reported that children with inflammatory bowel disease who developed psoriasiform dermatitis while on infliximab only rarely possessed the HLA-Cw*0602 genotype, which is commonly associated with psoriasis (Inflamm Bowel Dis. 2014 Aug;20[8]:1309-15).
The possibility of streptococcal or staphylococcal infection serving as a trigger for TNF inhibitor–induced psoriasis is also being explored, according to Dr. Paller.
She has received research funding from LEO Pharma and Amgen and serves as a consultant to AbbVie.
VANCOUVER – Tumor necrosis factor inhibitor–induced psoriasiform dermatitis can occur in pediatric patients after any length of treatment, with documented cases emerging after the very first dose and as late as 63 months into anti-TNF therapy, Dr. Amy S. Paller said at the World Congress of Dermatology.
Histopathologically, this medication-induced condition is psoriasis. But it tends to follow a distinctive pattern, favoring the scalp, dorsal hands and feet, nails, and periorificial skin. Palmoplantar pustulosis is not uncommon. The lesions are often secondarily infected with Staphylococcus aureus, according to Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University in Chicago.
The phenomenon was first described in adults. But in the past several years, as the use of tumor necrosis factor (TNF) antagonists has gained increasing traction for treatment of pediatric inflammatory bowel disease and rheumatologic diseases, the dermatologic disorder has become better characterized in youths. In a retrospective study at McMaster Children’s Hospital in Hamilton, Ont., 17 of 172 (10%) infliximab-treated patients with Crohn’s disease developed new-onset psoriasis and another (0.6%) experienced worsening of preexisting psoriasis after anywhere from 1 to 25 infusions. Most patients responded well to topical steroids; however, three discontinued the biologic because of this complication (J Pediatr Gastroenterol Nutr. 2013 May;56[5]:512-8).
It’s now clear that the emergence of TNF inhibitor–induced psoriasis does not adversely affect the response of a patient’s inflammatory bowel disease or juvenile arthritis to the biologic. Also, the risk of recurrent psoriatic eruption is not reduced by concurrent methotrexate.
Researchers find TNF inhibitor–induced psoriasis to be an intriguing puzzle because of its paradoxical nature. After all, the TNF inhibitors are a highly effective treatment for moderate to severe plaque psoriasis. The leading theory as to the underlying basis for TNF inhibitor–induced psoriasis is that it may have a genetic basis, Dr. Paller noted.
The McMaster group found that their pediatric Crohn’s disease patients who developed psoriasis in conjunction with infliximab (Remicade) therapy were more likely than disease-matched controls to be homozygous for one of several specific polymorphisms in the interleukin-23R gene. And investigators at the University of Helsinki have reported that children with inflammatory bowel disease who developed psoriasiform dermatitis while on infliximab only rarely possessed the HLA-Cw*0602 genotype, which is commonly associated with psoriasis (Inflamm Bowel Dis. 2014 Aug;20[8]:1309-15).
The possibility of streptococcal or staphylococcal infection serving as a trigger for TNF inhibitor–induced psoriasis is also being explored, according to Dr. Paller.
She has received research funding from LEO Pharma and Amgen and serves as a consultant to AbbVie.
EXPERT ANALYSIS FROM WCD 2015
Nail psoriasis therapies lack supporting evidence
VANCOUVER – Evidence-based therapy for nail psoriasis is in a sorry state because of a lack of consensus on a reliable nail psoriasis scoring system for use in clinical trials, according to a coauthor of the Cochrane systematic review of interventions for nail psoriasis.
“The last 12 randomized clinical trials used 21 ways of scoring the results of treatment, so comparing the studies means comparing apples to oranges. Which is the most effective treatment? What should we advise our patients? We don’t know. Comparison is impossible,” Dr. Marcel C. Pasch said at the World Congress of Dermatology.
The Cochrane report (Cochrane Database Syst Rev. 2013 Jan 31;1:CD007633) deemed the evidence for topical therapies as “inconclusive and weak,” even though topicals are the treatment mainstay for this localized expression of psoriasis. Indeed, Dr. Pasch and his coauthors found that no topical therapy has been shown effective in improving nail psoriasis. The Cochrane group concluded that just five therapies rise to the standard of being evidence based in terms of efficacy: the tumor necrosis factor (TNF) inhibitors infliximab (Remicade) and golimumab (Simponi), superficial radiation therapy, Grenz rays, and electron beam therapy. All five are strikingly impractical for use in clinical practice.
“The findings are quite disappointing because nobody sends a patient with psoriasis to the radiotherapist, and while giving an anti-TNF biologic only for the nails will be effective, at least in my country it won’t be reimbursed,” wrote Dr. Pasch, a dermatologist at Radboud University Nijmegen (the Netherlands) Medical Centre.
The presence and severity of nail psoriasis is unrelated to the severity of cutaneous psoriasis. Moreover, nail psoriasis without cutaneous involvement occurs in 5%-10% of psoriasis patients.
Since publication of the Cochrane systematic review, 12 new randomized controlled trials of treatments for nail psoriasis have appeared. Six focused on biologics: the anti-TNF agents certolizumab (Cimzia), etanercept (Enbrel), and adalimumab (Humira); the anti–interleukin-12/23 agent ustekinumab (Stelara); and the interleukin-17A inhibitor secukiumab (Cosentyx). Dr. Pasch said in his opinion all five biologics were supported by convincing studies and now can be added to the short list of evidence-based nail psoriasis therapies.
Of the six recent studies of topical therapies, two provided persuasive evidence of efficacy, in his view: tacrolimus ointment and indigo naturalis extract in oil (Lindioil), a variant of a traditional Chinese medicine therapy, which at this time isn’t commercially available.
In contrast, studies of clobetasol nail lacquer, pulsed dye laser therapy, a nail lacquer based upon chitin from crab shells, and a study of calcitriol ointment versus betamethasone dipropionate ointment failed to be convincing either because of methodologic problems or lack of efficacy, he continued.
These 12 recent randomized clinical trials utilized 21 different nail psoriasis scoring systems.
“Which scoring system is best? The answer is, we don’t know,” Dr. Pasch said.
He and his coinvestigators compared eight different scoring systems in a prospective study and concluded that the Nijmegen–Nail Psoriasis Activity Index Tool (N-NAIL), which Dr. Pasch helped develop, best reflected the clinical severity of nail psoriasis (J Am Acad Dermatol. 2014 Jun;70[6]:1061-6).
However, he added that at present there is no validated scoring system for nail psoriasis. And creation of a single validated scoring system that researchers can agree on as the standard is a prerequisite for making major advances in the treatment of nail psoriasis, in Dr. Pasch’s view.
He is so convinced of this that he has created an organization whose goal is to achieve consensus on one reliable, validated nail psoriasis scoring system for use in clinical trials. At the World Congress of Dermatology, he invited stakeholders – including academic and community dermatologists, patient organizations, and the pharmaceutical industry – to join (www.nailinitiative.org).
Session chair Dr. Peter van de Kerkhof, chairman of dermatology at Radboud University, said he sees the NAPSI (Nail Psoriasis Severity Index) being used in lots of clinical trials in psoriasis. What’s wrong with building a consensus around NAPSI? he asked.
“The problem is not the NAPSI score,” Dr. Pasch replied. “The problem is that in each trial a modified NAPSI score is used, but they are all modified in different ways. We have the single-hand NAPSI, the eight-finger NAPSI, the 10-finger NAPSI, the target NAPSI. The NAPSI doesn’t exist anymore.”
He reported receiving research grants from Pfizer and Janssen-Cilag.
VANCOUVER – Evidence-based therapy for nail psoriasis is in a sorry state because of a lack of consensus on a reliable nail psoriasis scoring system for use in clinical trials, according to a coauthor of the Cochrane systematic review of interventions for nail psoriasis.
“The last 12 randomized clinical trials used 21 ways of scoring the results of treatment, so comparing the studies means comparing apples to oranges. Which is the most effective treatment? What should we advise our patients? We don’t know. Comparison is impossible,” Dr. Marcel C. Pasch said at the World Congress of Dermatology.
The Cochrane report (Cochrane Database Syst Rev. 2013 Jan 31;1:CD007633) deemed the evidence for topical therapies as “inconclusive and weak,” even though topicals are the treatment mainstay for this localized expression of psoriasis. Indeed, Dr. Pasch and his coauthors found that no topical therapy has been shown effective in improving nail psoriasis. The Cochrane group concluded that just five therapies rise to the standard of being evidence based in terms of efficacy: the tumor necrosis factor (TNF) inhibitors infliximab (Remicade) and golimumab (Simponi), superficial radiation therapy, Grenz rays, and electron beam therapy. All five are strikingly impractical for use in clinical practice.
“The findings are quite disappointing because nobody sends a patient with psoriasis to the radiotherapist, and while giving an anti-TNF biologic only for the nails will be effective, at least in my country it won’t be reimbursed,” wrote Dr. Pasch, a dermatologist at Radboud University Nijmegen (the Netherlands) Medical Centre.
The presence and severity of nail psoriasis is unrelated to the severity of cutaneous psoriasis. Moreover, nail psoriasis without cutaneous involvement occurs in 5%-10% of psoriasis patients.
Since publication of the Cochrane systematic review, 12 new randomized controlled trials of treatments for nail psoriasis have appeared. Six focused on biologics: the anti-TNF agents certolizumab (Cimzia), etanercept (Enbrel), and adalimumab (Humira); the anti–interleukin-12/23 agent ustekinumab (Stelara); and the interleukin-17A inhibitor secukiumab (Cosentyx). Dr. Pasch said in his opinion all five biologics were supported by convincing studies and now can be added to the short list of evidence-based nail psoriasis therapies.
Of the six recent studies of topical therapies, two provided persuasive evidence of efficacy, in his view: tacrolimus ointment and indigo naturalis extract in oil (Lindioil), a variant of a traditional Chinese medicine therapy, which at this time isn’t commercially available.
In contrast, studies of clobetasol nail lacquer, pulsed dye laser therapy, a nail lacquer based upon chitin from crab shells, and a study of calcitriol ointment versus betamethasone dipropionate ointment failed to be convincing either because of methodologic problems or lack of efficacy, he continued.
These 12 recent randomized clinical trials utilized 21 different nail psoriasis scoring systems.
“Which scoring system is best? The answer is, we don’t know,” Dr. Pasch said.
He and his coinvestigators compared eight different scoring systems in a prospective study and concluded that the Nijmegen–Nail Psoriasis Activity Index Tool (N-NAIL), which Dr. Pasch helped develop, best reflected the clinical severity of nail psoriasis (J Am Acad Dermatol. 2014 Jun;70[6]:1061-6).
However, he added that at present there is no validated scoring system for nail psoriasis. And creation of a single validated scoring system that researchers can agree on as the standard is a prerequisite for making major advances in the treatment of nail psoriasis, in Dr. Pasch’s view.
He is so convinced of this that he has created an organization whose goal is to achieve consensus on one reliable, validated nail psoriasis scoring system for use in clinical trials. At the World Congress of Dermatology, he invited stakeholders – including academic and community dermatologists, patient organizations, and the pharmaceutical industry – to join (www.nailinitiative.org).
Session chair Dr. Peter van de Kerkhof, chairman of dermatology at Radboud University, said he sees the NAPSI (Nail Psoriasis Severity Index) being used in lots of clinical trials in psoriasis. What’s wrong with building a consensus around NAPSI? he asked.
“The problem is not the NAPSI score,” Dr. Pasch replied. “The problem is that in each trial a modified NAPSI score is used, but they are all modified in different ways. We have the single-hand NAPSI, the eight-finger NAPSI, the 10-finger NAPSI, the target NAPSI. The NAPSI doesn’t exist anymore.”
He reported receiving research grants from Pfizer and Janssen-Cilag.
VANCOUVER – Evidence-based therapy for nail psoriasis is in a sorry state because of a lack of consensus on a reliable nail psoriasis scoring system for use in clinical trials, according to a coauthor of the Cochrane systematic review of interventions for nail psoriasis.
“The last 12 randomized clinical trials used 21 ways of scoring the results of treatment, so comparing the studies means comparing apples to oranges. Which is the most effective treatment? What should we advise our patients? We don’t know. Comparison is impossible,” Dr. Marcel C. Pasch said at the World Congress of Dermatology.
The Cochrane report (Cochrane Database Syst Rev. 2013 Jan 31;1:CD007633) deemed the evidence for topical therapies as “inconclusive and weak,” even though topicals are the treatment mainstay for this localized expression of psoriasis. Indeed, Dr. Pasch and his coauthors found that no topical therapy has been shown effective in improving nail psoriasis. The Cochrane group concluded that just five therapies rise to the standard of being evidence based in terms of efficacy: the tumor necrosis factor (TNF) inhibitors infliximab (Remicade) and golimumab (Simponi), superficial radiation therapy, Grenz rays, and electron beam therapy. All five are strikingly impractical for use in clinical practice.
“The findings are quite disappointing because nobody sends a patient with psoriasis to the radiotherapist, and while giving an anti-TNF biologic only for the nails will be effective, at least in my country it won’t be reimbursed,” wrote Dr. Pasch, a dermatologist at Radboud University Nijmegen (the Netherlands) Medical Centre.
The presence and severity of nail psoriasis is unrelated to the severity of cutaneous psoriasis. Moreover, nail psoriasis without cutaneous involvement occurs in 5%-10% of psoriasis patients.
Since publication of the Cochrane systematic review, 12 new randomized controlled trials of treatments for nail psoriasis have appeared. Six focused on biologics: the anti-TNF agents certolizumab (Cimzia), etanercept (Enbrel), and adalimumab (Humira); the anti–interleukin-12/23 agent ustekinumab (Stelara); and the interleukin-17A inhibitor secukiumab (Cosentyx). Dr. Pasch said in his opinion all five biologics were supported by convincing studies and now can be added to the short list of evidence-based nail psoriasis therapies.
Of the six recent studies of topical therapies, two provided persuasive evidence of efficacy, in his view: tacrolimus ointment and indigo naturalis extract in oil (Lindioil), a variant of a traditional Chinese medicine therapy, which at this time isn’t commercially available.
In contrast, studies of clobetasol nail lacquer, pulsed dye laser therapy, a nail lacquer based upon chitin from crab shells, and a study of calcitriol ointment versus betamethasone dipropionate ointment failed to be convincing either because of methodologic problems or lack of efficacy, he continued.
These 12 recent randomized clinical trials utilized 21 different nail psoriasis scoring systems.
“Which scoring system is best? The answer is, we don’t know,” Dr. Pasch said.
He and his coinvestigators compared eight different scoring systems in a prospective study and concluded that the Nijmegen–Nail Psoriasis Activity Index Tool (N-NAIL), which Dr. Pasch helped develop, best reflected the clinical severity of nail psoriasis (J Am Acad Dermatol. 2014 Jun;70[6]:1061-6).
However, he added that at present there is no validated scoring system for nail psoriasis. And creation of a single validated scoring system that researchers can agree on as the standard is a prerequisite for making major advances in the treatment of nail psoriasis, in Dr. Pasch’s view.
He is so convinced of this that he has created an organization whose goal is to achieve consensus on one reliable, validated nail psoriasis scoring system for use in clinical trials. At the World Congress of Dermatology, he invited stakeholders – including academic and community dermatologists, patient organizations, and the pharmaceutical industry – to join (www.nailinitiative.org).
Session chair Dr. Peter van de Kerkhof, chairman of dermatology at Radboud University, said he sees the NAPSI (Nail Psoriasis Severity Index) being used in lots of clinical trials in psoriasis. What’s wrong with building a consensus around NAPSI? he asked.
“The problem is not the NAPSI score,” Dr. Pasch replied. “The problem is that in each trial a modified NAPSI score is used, but they are all modified in different ways. We have the single-hand NAPSI, the eight-finger NAPSI, the 10-finger NAPSI, the target NAPSI. The NAPSI doesn’t exist anymore.”
He reported receiving research grants from Pfizer and Janssen-Cilag.
EXPERT ANALYSIS FROM WCD 2015
Psoriasis patients more likely to have type D personalities
Incidence of type D personality was significantly more common in patients with moderate to severe psoriasis, compared with a healthy control group, according to Dr. Alejandro Molina-Leyva of Hospital Torrecardenas, Almeria, Spain, and his associates.
People with type D, or distressed, personality tend to be more worried and irritable, and tend to display more negative emotions than do others. Of the 90 patients with moderate to severe psoriasis included in the study, 39% had type D personality, compared with 24% of the 82 members of the control group. The odds ratio for psoriasis patients developing type D personality was 2.1.
Psoriasis patients with type D personalities had significantly worse general, sexual, and psoriasis-related health-related quality of life, compared with psoriasis patients without type D personality. In addition, type D personality psoriasis patients were much more likely to experience anxiety or depression than were healthy people with type D personality, with an OR of 3.2.
“It may be that the higher prevalence of type D personality in moderate to severe psoriasis is, at least in part, the result of accumulated psychic damage over years of evolution of the disease. It is important to conduct prospective studies with incident cases of psoriasis to clarify the relationship between type D personality and psoriasis,” the investigators noted.
Find the full study here in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.12960).
Incidence of type D personality was significantly more common in patients with moderate to severe psoriasis, compared with a healthy control group, according to Dr. Alejandro Molina-Leyva of Hospital Torrecardenas, Almeria, Spain, and his associates.
People with type D, or distressed, personality tend to be more worried and irritable, and tend to display more negative emotions than do others. Of the 90 patients with moderate to severe psoriasis included in the study, 39% had type D personality, compared with 24% of the 82 members of the control group. The odds ratio for psoriasis patients developing type D personality was 2.1.
Psoriasis patients with type D personalities had significantly worse general, sexual, and psoriasis-related health-related quality of life, compared with psoriasis patients without type D personality. In addition, type D personality psoriasis patients were much more likely to experience anxiety or depression than were healthy people with type D personality, with an OR of 3.2.
“It may be that the higher prevalence of type D personality in moderate to severe psoriasis is, at least in part, the result of accumulated psychic damage over years of evolution of the disease. It is important to conduct prospective studies with incident cases of psoriasis to clarify the relationship between type D personality and psoriasis,” the investigators noted.
Find the full study here in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.12960).
Incidence of type D personality was significantly more common in patients with moderate to severe psoriasis, compared with a healthy control group, according to Dr. Alejandro Molina-Leyva of Hospital Torrecardenas, Almeria, Spain, and his associates.
People with type D, or distressed, personality tend to be more worried and irritable, and tend to display more negative emotions than do others. Of the 90 patients with moderate to severe psoriasis included in the study, 39% had type D personality, compared with 24% of the 82 members of the control group. The odds ratio for psoriasis patients developing type D personality was 2.1.
Psoriasis patients with type D personalities had significantly worse general, sexual, and psoriasis-related health-related quality of life, compared with psoriasis patients without type D personality. In addition, type D personality psoriasis patients were much more likely to experience anxiety or depression than were healthy people with type D personality, with an OR of 3.2.
“It may be that the higher prevalence of type D personality in moderate to severe psoriasis is, at least in part, the result of accumulated psychic damage over years of evolution of the disease. It is important to conduct prospective studies with incident cases of psoriasis to clarify the relationship between type D personality and psoriasis,” the investigators noted.
Find the full study here in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.12960).
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Psoriasis and acne worse in winter, milder in summer
Psoriasis and acne appear to be susceptible to seasonal variations of clearing and worsening, with an analysis revealing both conditions maintained a significant trend of summer clearing and winter worsening, according to a research letter published in the Journal of the American Academy of Dermatology.
Using Physician’s Global Assessment scales for psoriasis and acne, Dr. Vanessa Lindsay Pascoe and Dr. Alexandra Boer Kimball, both of Massachusetts General Hospital, Boston, collected data from 5,468 psoriasis patients and 9,301 acne patients between June 2011 and May 2014 in the New England area. Among the psoriasis patient group, 16% were seen in the summer, 25% in the fall, 31% in the winter, and 28% in the spring. The trend was similar for acne patients, with 18% seen in the summer, 25% in the fall, 28% in the winter, and 29% in the spring. There were no significant seasonal differences in age or sex for either group.
The percentage of psoriasis patients with clear/almost clear disease was highest in the summer at 20.4%, while the percentage of patients with moderate/severe disease was highest in the winter at 40.5%. For acne, the percentage of patients with clear/almost clear disease was highest in the fall at 17.5%, and the percentage of patients with moderate/severe disease was highest in the winter at 45.9%. Fewer psoriasis and acne patients presented to the clinic in the summer, which the researchers suggested could be due to disease improvement.
“Although the climate of the Northeastern United States may not generalize to regions with less seasonal variation, providers may consider seasonal adjustment of acne plans as they have traditionally done for psoriasis,” the authors wrote. “For example, they may wait until after winter to taper a systemic antibiotic for acne, just as some providers may wait until spring to change systemic psoriasis treatments.”
Read the full article in the Journal of the American Academy of Dermatology.
Psoriasis and acne appear to be susceptible to seasonal variations of clearing and worsening, with an analysis revealing both conditions maintained a significant trend of summer clearing and winter worsening, according to a research letter published in the Journal of the American Academy of Dermatology.
Using Physician’s Global Assessment scales for psoriasis and acne, Dr. Vanessa Lindsay Pascoe and Dr. Alexandra Boer Kimball, both of Massachusetts General Hospital, Boston, collected data from 5,468 psoriasis patients and 9,301 acne patients between June 2011 and May 2014 in the New England area. Among the psoriasis patient group, 16% were seen in the summer, 25% in the fall, 31% in the winter, and 28% in the spring. The trend was similar for acne patients, with 18% seen in the summer, 25% in the fall, 28% in the winter, and 29% in the spring. There were no significant seasonal differences in age or sex for either group.
The percentage of psoriasis patients with clear/almost clear disease was highest in the summer at 20.4%, while the percentage of patients with moderate/severe disease was highest in the winter at 40.5%. For acne, the percentage of patients with clear/almost clear disease was highest in the fall at 17.5%, and the percentage of patients with moderate/severe disease was highest in the winter at 45.9%. Fewer psoriasis and acne patients presented to the clinic in the summer, which the researchers suggested could be due to disease improvement.
“Although the climate of the Northeastern United States may not generalize to regions with less seasonal variation, providers may consider seasonal adjustment of acne plans as they have traditionally done for psoriasis,” the authors wrote. “For example, they may wait until after winter to taper a systemic antibiotic for acne, just as some providers may wait until spring to change systemic psoriasis treatments.”
Read the full article in the Journal of the American Academy of Dermatology.
Psoriasis and acne appear to be susceptible to seasonal variations of clearing and worsening, with an analysis revealing both conditions maintained a significant trend of summer clearing and winter worsening, according to a research letter published in the Journal of the American Academy of Dermatology.
Using Physician’s Global Assessment scales for psoriasis and acne, Dr. Vanessa Lindsay Pascoe and Dr. Alexandra Boer Kimball, both of Massachusetts General Hospital, Boston, collected data from 5,468 psoriasis patients and 9,301 acne patients between June 2011 and May 2014 in the New England area. Among the psoriasis patient group, 16% were seen in the summer, 25% in the fall, 31% in the winter, and 28% in the spring. The trend was similar for acne patients, with 18% seen in the summer, 25% in the fall, 28% in the winter, and 29% in the spring. There were no significant seasonal differences in age or sex for either group.
The percentage of psoriasis patients with clear/almost clear disease was highest in the summer at 20.4%, while the percentage of patients with moderate/severe disease was highest in the winter at 40.5%. For acne, the percentage of patients with clear/almost clear disease was highest in the fall at 17.5%, and the percentage of patients with moderate/severe disease was highest in the winter at 45.9%. Fewer psoriasis and acne patients presented to the clinic in the summer, which the researchers suggested could be due to disease improvement.
“Although the climate of the Northeastern United States may not generalize to regions with less seasonal variation, providers may consider seasonal adjustment of acne plans as they have traditionally done for psoriasis,” the authors wrote. “For example, they may wait until after winter to taper a systemic antibiotic for acne, just as some providers may wait until spring to change systemic psoriasis treatments.”
Read the full article in the Journal of the American Academy of Dermatology.
Interviews with psoriasis patients reveal disease hardships
Interviewing patients who live with psoriasis can reveal important information not included in psoriasis severity measures, according to Dr. David Pariser of Eastern Virginia Medical School, Norfolk, and his associates.
More than 90% of 101 patients with moderate to severe plaque psoriasis, with and without psoriatic arthritis, reported emotional and social impacts to their lives. Social impacts included avoiding/changing activities, relationships, or trying new things. Emotional impacts included a sense of anger, frustration, embarrassment, and self-consciousness; a lowered sense of self-esteem was common. Patients reported impacts in family, professional, social, and educational areas.
Patients with psoriasis and psoriatic arthritis tended to report both symptoms and well-being complications more often, with 77% reporting physical impacts and 34% reporting educational impacts, compared with 38% and 17%, respectively, for those with psoriasis only.
The study data “provide practitioners with deep insight into how patients with moderate to severe psoriasis are suffering, regardless of whether the concerns are elucidated during patient visits. The burdens in patients’ lives support the use of effective treatment; indeed, patients expressed how they value physicians who are knowledgeable about psoriasis and who will try various therapies,” the investigators concluded.
Find the full study in the Journal of Dermatological Treatment (doi: 10.3109/09546634.2015.1044492).
Interviewing patients who live with psoriasis can reveal important information not included in psoriasis severity measures, according to Dr. David Pariser of Eastern Virginia Medical School, Norfolk, and his associates.
More than 90% of 101 patients with moderate to severe plaque psoriasis, with and without psoriatic arthritis, reported emotional and social impacts to their lives. Social impacts included avoiding/changing activities, relationships, or trying new things. Emotional impacts included a sense of anger, frustration, embarrassment, and self-consciousness; a lowered sense of self-esteem was common. Patients reported impacts in family, professional, social, and educational areas.
Patients with psoriasis and psoriatic arthritis tended to report both symptoms and well-being complications more often, with 77% reporting physical impacts and 34% reporting educational impacts, compared with 38% and 17%, respectively, for those with psoriasis only.
The study data “provide practitioners with deep insight into how patients with moderate to severe psoriasis are suffering, regardless of whether the concerns are elucidated during patient visits. The burdens in patients’ lives support the use of effective treatment; indeed, patients expressed how they value physicians who are knowledgeable about psoriasis and who will try various therapies,” the investigators concluded.
Find the full study in the Journal of Dermatological Treatment (doi: 10.3109/09546634.2015.1044492).
Interviewing patients who live with psoriasis can reveal important information not included in psoriasis severity measures, according to Dr. David Pariser of Eastern Virginia Medical School, Norfolk, and his associates.
More than 90% of 101 patients with moderate to severe plaque psoriasis, with and without psoriatic arthritis, reported emotional and social impacts to their lives. Social impacts included avoiding/changing activities, relationships, or trying new things. Emotional impacts included a sense of anger, frustration, embarrassment, and self-consciousness; a lowered sense of self-esteem was common. Patients reported impacts in family, professional, social, and educational areas.
Patients with psoriasis and psoriatic arthritis tended to report both symptoms and well-being complications more often, with 77% reporting physical impacts and 34% reporting educational impacts, compared with 38% and 17%, respectively, for those with psoriasis only.
The study data “provide practitioners with deep insight into how patients with moderate to severe psoriasis are suffering, regardless of whether the concerns are elucidated during patient visits. The burdens in patients’ lives support the use of effective treatment; indeed, patients expressed how they value physicians who are knowledgeable about psoriasis and who will try various therapies,” the investigators concluded.
Find the full study in the Journal of Dermatological Treatment (doi: 10.3109/09546634.2015.1044492).
VIDEO: Use fiduciary duty to set pain medication boundaries
ORLANDO – Physicians should use the concept of fiduciary duty to set appropriate boundaries with patients taking pain medications, explained Dr. Louis Kuritzky.
Often, patients want treatments that are not in their best interests, noted Dr. Kuritzky of the department of community health and family medicine at the University of Florida, Gainesville.
In an interview at a meeting held by the American Pain Society and Global Academy for Medical Education, Dr. Kuritzky outlined how physicians can take a fiduciary duty approach to set boundaries with patients in a dispassionate manner.
Global Academy and this news organization are owned by the same company. Dr. Kuritzky reported a financial relationship with Lilly.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ORLANDO – Physicians should use the concept of fiduciary duty to set appropriate boundaries with patients taking pain medications, explained Dr. Louis Kuritzky.
Often, patients want treatments that are not in their best interests, noted Dr. Kuritzky of the department of community health and family medicine at the University of Florida, Gainesville.
In an interview at a meeting held by the American Pain Society and Global Academy for Medical Education, Dr. Kuritzky outlined how physicians can take a fiduciary duty approach to set boundaries with patients in a dispassionate manner.
Global Academy and this news organization are owned by the same company. Dr. Kuritzky reported a financial relationship with Lilly.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ORLANDO – Physicians should use the concept of fiduciary duty to set appropriate boundaries with patients taking pain medications, explained Dr. Louis Kuritzky.
Often, patients want treatments that are not in their best interests, noted Dr. Kuritzky of the department of community health and family medicine at the University of Florida, Gainesville.
In an interview at a meeting held by the American Pain Society and Global Academy for Medical Education, Dr. Kuritzky outlined how physicians can take a fiduciary duty approach to set boundaries with patients in a dispassionate manner.
Global Academy and this news organization are owned by the same company. Dr. Kuritzky reported a financial relationship with Lilly.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
EXPERT ANALYSIS FROM PAIN CARE FOR PRIMARY CARE
Higher risk of arrhythmia in psoriasis patients
Patients with psoriasis are at increased risk of arrhythmia, with the risk even greater for younger patients and those with psoriatic arthritis, according to a population-based cohort study conducted in Taiwan.
Dr. Hsien-Yi Chiu of National Taiwan University and Wei-Lun Chang of National Yang-Ming University, both in Taipei, and their colleagues, looked at records from 40,637 patients diagnosed with psoriasis and 162,548 age- and sex-matched controls without psoriasis, over a mean follow-up of about 6 years, for the incidence of arrhythmias over a mean of 6 years.
In an article published in the September issue of the Journal of the American Academy of Dermatology, the investigators reported that those patients with psoriasis were at a significantly higher risk of developing arrhythmia, independent of traditional cardiovascular risk factors (adjusted hazard ratio, 1.34; 95% confidence interval, 1.29-1.39). Increased risk for patients with mild disease (aHR,1.35; 95% CI, 1.30-1.41) was comparable to that of patients with severe disease (aHR, 1.25; 95% CI 1.12-1.39) and more pronounced in the subgroup of patients with psoriatic arthritis (aHR, 1.46; 95% CI, 1.22-1.74). Younger patients, between aged 20 and 39 years, were at a higher risk (aHR, 1.39; 95% CI, 1.26-1.54) than older patients in the cohort (J Am Acad Dermatol. 2015 Sep;73:429-38).
Although previous studies have shown severe psoriasis to be associated with a nearly 60% increase in cardiovascular morbidity and mortality beyond traditional risk factors, less is known about arrhythmias specifically. “Inflammation may contribute to the alteration of cardiomyocyte electrophysiology, such as dysregulation of ion channel function, leading to increased risk of arrhythmia,” the investigators wrote.
The authors noted that limitations of their study were the potential surveillance bias for psoriasis patients due to increased hospital visits, and the fact that alcohol and tobacco use was not captured in the patient data. Treatment with systemic therapies may lower cardiovascular risk in psoriasis patients, they added, which may have explained why the arrhythmia risk among patients with severe disease was similar to those with mild disease.
The findings indicate “that psoriasis can be added to future risk-stratification scores for arrhythmia,” the investigators wrote, adding that patients with psoriasis, “especially young patients and those with PsA [psoriatic arthritis] , should be more closely screened for various types of arrhythmia,” with the hope of earlier intervention leading to reduction of cardiovascular morbidity and mortality.
Patients with psoriasis are at increased risk of arrhythmia, with the risk even greater for younger patients and those with psoriatic arthritis, according to a population-based cohort study conducted in Taiwan.
Dr. Hsien-Yi Chiu of National Taiwan University and Wei-Lun Chang of National Yang-Ming University, both in Taipei, and their colleagues, looked at records from 40,637 patients diagnosed with psoriasis and 162,548 age- and sex-matched controls without psoriasis, over a mean follow-up of about 6 years, for the incidence of arrhythmias over a mean of 6 years.
In an article published in the September issue of the Journal of the American Academy of Dermatology, the investigators reported that those patients with psoriasis were at a significantly higher risk of developing arrhythmia, independent of traditional cardiovascular risk factors (adjusted hazard ratio, 1.34; 95% confidence interval, 1.29-1.39). Increased risk for patients with mild disease (aHR,1.35; 95% CI, 1.30-1.41) was comparable to that of patients with severe disease (aHR, 1.25; 95% CI 1.12-1.39) and more pronounced in the subgroup of patients with psoriatic arthritis (aHR, 1.46; 95% CI, 1.22-1.74). Younger patients, between aged 20 and 39 years, were at a higher risk (aHR, 1.39; 95% CI, 1.26-1.54) than older patients in the cohort (J Am Acad Dermatol. 2015 Sep;73:429-38).
Although previous studies have shown severe psoriasis to be associated with a nearly 60% increase in cardiovascular morbidity and mortality beyond traditional risk factors, less is known about arrhythmias specifically. “Inflammation may contribute to the alteration of cardiomyocyte electrophysiology, such as dysregulation of ion channel function, leading to increased risk of arrhythmia,” the investigators wrote.
The authors noted that limitations of their study were the potential surveillance bias for psoriasis patients due to increased hospital visits, and the fact that alcohol and tobacco use was not captured in the patient data. Treatment with systemic therapies may lower cardiovascular risk in psoriasis patients, they added, which may have explained why the arrhythmia risk among patients with severe disease was similar to those with mild disease.
The findings indicate “that psoriasis can be added to future risk-stratification scores for arrhythmia,” the investigators wrote, adding that patients with psoriasis, “especially young patients and those with PsA [psoriatic arthritis] , should be more closely screened for various types of arrhythmia,” with the hope of earlier intervention leading to reduction of cardiovascular morbidity and mortality.
Patients with psoriasis are at increased risk of arrhythmia, with the risk even greater for younger patients and those with psoriatic arthritis, according to a population-based cohort study conducted in Taiwan.
Dr. Hsien-Yi Chiu of National Taiwan University and Wei-Lun Chang of National Yang-Ming University, both in Taipei, and their colleagues, looked at records from 40,637 patients diagnosed with psoriasis and 162,548 age- and sex-matched controls without psoriasis, over a mean follow-up of about 6 years, for the incidence of arrhythmias over a mean of 6 years.
In an article published in the September issue of the Journal of the American Academy of Dermatology, the investigators reported that those patients with psoriasis were at a significantly higher risk of developing arrhythmia, independent of traditional cardiovascular risk factors (adjusted hazard ratio, 1.34; 95% confidence interval, 1.29-1.39). Increased risk for patients with mild disease (aHR,1.35; 95% CI, 1.30-1.41) was comparable to that of patients with severe disease (aHR, 1.25; 95% CI 1.12-1.39) and more pronounced in the subgroup of patients with psoriatic arthritis (aHR, 1.46; 95% CI, 1.22-1.74). Younger patients, between aged 20 and 39 years, were at a higher risk (aHR, 1.39; 95% CI, 1.26-1.54) than older patients in the cohort (J Am Acad Dermatol. 2015 Sep;73:429-38).
Although previous studies have shown severe psoriasis to be associated with a nearly 60% increase in cardiovascular morbidity and mortality beyond traditional risk factors, less is known about arrhythmias specifically. “Inflammation may contribute to the alteration of cardiomyocyte electrophysiology, such as dysregulation of ion channel function, leading to increased risk of arrhythmia,” the investigators wrote.
The authors noted that limitations of their study were the potential surveillance bias for psoriasis patients due to increased hospital visits, and the fact that alcohol and tobacco use was not captured in the patient data. Treatment with systemic therapies may lower cardiovascular risk in psoriasis patients, they added, which may have explained why the arrhythmia risk among patients with severe disease was similar to those with mild disease.
The findings indicate “that psoriasis can be added to future risk-stratification scores for arrhythmia,” the investigators wrote, adding that patients with psoriasis, “especially young patients and those with PsA [psoriatic arthritis] , should be more closely screened for various types of arrhythmia,” with the hope of earlier intervention leading to reduction of cardiovascular morbidity and mortality.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point: Patients with psoriasis are at an increased risk of developing arrhythmias compared to those without psoriasis.
Major finding: After researchers adjusted for medical history and medication use, patients with psoriasis were at increased risk of overall arrhythmia (adjusted hazard ratio, 1.34; 95% confidence interval, 1.29-1.39).
Data source: A retrospective cohort study using data from almost 41,000 psoriasis patients identified from the Taiwan National Health Insurance Research Database, and almost 163,000 age and sex-matched cohorts from the same database
Disclosures: Study was institutionally funded. Dr. Chiu, Ms. Chang, and three other authors had no disclosures; one author disclosed having conducted clinical trials, or having received honoraria from several companies, including Pfizer and Novartis, and having received speaking fees from AbbVie.
Simple screening tool can help detect psoriatic arthritis
PARK CITY, UTAH – Since most people with psoriatic arthritis (PsA) develop psoriasis before joint symptoms, it’s helpful to have a simple screening test for the condition.
One of Dr. Philip Mease’s favorite PsA screening tools is the Psoriasis Epidemiology Screening Test (PEST), which was first described at the 2009 annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and consists of five simple questions. They are: Have you ever had a swollen joint (or joints)? Has a doctor ever told you that you have arthritis? Do your fingernails or toes have holes or pits? Have you had pain in your heel? Have you had a finger or toe that was completely swollen and painful for no apparent reason? (Clin Exp Rheumatol. 2009;27:469-74).
“Just these five simple questions, or trying to remember a few of them, can help you in your review of systems,” Dr. Mease, director of arthritis research at Swedish Medical Center, Seattle, said at the annual meeting of the Pacific Dermatologic Association. “I think patients appreciate it when you look beyond the skin in your questioning. These can pick up [PsA] with a high sensitivity and specificity” of 0.92 and 0.78, respectively.
He went on to discuss current PsA treatment approaches. According to an evidence review that he and his associates in GRAPPA published in 2009, biologics (anti–tumor necrosis factor inhibitors) as a group were found to be effective in all five domains of the disease: peripheral arthritis, skin and nail disease, axial disease, dactylitis, and enthesitis, while the oral disease-modifying antirheumatic drugs (DMARDs) were effective for peripheral arthritis and skin and nail disease.
Other treatments that were found effective are: psoralen and UVA/UVB for skin and nail disease, physiotherapy for axial disease, intra-articular steroids for peripheral arthritis, and NSAIDs for peripheral arthritis and axial disease (J Rheum. 2009;33:1417-21). “Patients with mild disease can be tried on NSAIDs, especially in a patient with monoarticular disease, but for the most part we need to move on to using systemic medication,” he said. Updated recommendations from GRAPPA include new data regarding ustekinumab, apremilast, and secukinumab, as well as data on comorbidities (J Rheumatol. 2015;42[6]:1052-5).
According to Dr. Mease, controlled trials of DMARDs in PsA patients have yielded treatment effects that range from marginal in the joints to marginal or none at all in the skin. Data from the Methotrexate in Psoriatic Arthritis trial conducted in the United Kingdom and published in 2012 showed no evidence that methotrexate improves inflammatory synovitis in active PsA (Rheumatol. 2012;51:1368-77).
“There were issues with this trial, including the fact that it took 5 years to enroll patients, and many dropped out, so I don’t think it’s a very reliable study,” said Dr. Mease, who is also a professor of medicine in the division of rheumatology at the University of Washington, Seattle. “Currently, Amgen is in the process of starting a trial in which the goal is to enroll 840 subjects with early PsA who are being randomized to methotrexate alone, Enbrel alone, or Enbrel plus methotrexate. This, I think, is going to give us a better answer about the effectiveness of methotrexate. It will also teach us about whether there’s a value in combining an anti-TNF inhibitor with methotrexate. We still don’t know the answer to that question.”
The most recent data on methotrexate come from an open-label trial known as TICOPA, which used a tight control treatment paradigm through 48 weeks of treatment. A subanalysis of 188 patients treated with methotrexate through 12 weeks was presented at the 2015 meeting of the European League Against Rheumatism. It revealed that 41% of patients achieved an ACR 20, 22% achieved minimal disease activity, 62% experienced an improvement in dactylitis, and 25% experienced an improvement in enthesitis.
“So we have a few data suggesting that methotrexate may be modestly effective in treating PsA,” Dr. Mease said. “We often will start with methotrexate unless the patient has really aggressive disease activity. If they get some effect from the drug but not enough, we’ll often add a biologic agent but often keep some methotrexate in the background, even at 10 mg per week, in order to reduce immunogenicity from a biologic.”
Dr. Mease disclosed that he has received research grants, consultation fees, and/or speaker honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Crescendo Bioscience, Genentech, GlaxoSmithKline, Janssen, Eli Lilly, Merck, Novartis, Pfizer, and UCB.
PARK CITY, UTAH – Since most people with psoriatic arthritis (PsA) develop psoriasis before joint symptoms, it’s helpful to have a simple screening test for the condition.
One of Dr. Philip Mease’s favorite PsA screening tools is the Psoriasis Epidemiology Screening Test (PEST), which was first described at the 2009 annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and consists of five simple questions. They are: Have you ever had a swollen joint (or joints)? Has a doctor ever told you that you have arthritis? Do your fingernails or toes have holes or pits? Have you had pain in your heel? Have you had a finger or toe that was completely swollen and painful for no apparent reason? (Clin Exp Rheumatol. 2009;27:469-74).
“Just these five simple questions, or trying to remember a few of them, can help you in your review of systems,” Dr. Mease, director of arthritis research at Swedish Medical Center, Seattle, said at the annual meeting of the Pacific Dermatologic Association. “I think patients appreciate it when you look beyond the skin in your questioning. These can pick up [PsA] with a high sensitivity and specificity” of 0.92 and 0.78, respectively.
He went on to discuss current PsA treatment approaches. According to an evidence review that he and his associates in GRAPPA published in 2009, biologics (anti–tumor necrosis factor inhibitors) as a group were found to be effective in all five domains of the disease: peripheral arthritis, skin and nail disease, axial disease, dactylitis, and enthesitis, while the oral disease-modifying antirheumatic drugs (DMARDs) were effective for peripheral arthritis and skin and nail disease.
Other treatments that were found effective are: psoralen and UVA/UVB for skin and nail disease, physiotherapy for axial disease, intra-articular steroids for peripheral arthritis, and NSAIDs for peripheral arthritis and axial disease (J Rheum. 2009;33:1417-21). “Patients with mild disease can be tried on NSAIDs, especially in a patient with monoarticular disease, but for the most part we need to move on to using systemic medication,” he said. Updated recommendations from GRAPPA include new data regarding ustekinumab, apremilast, and secukinumab, as well as data on comorbidities (J Rheumatol. 2015;42[6]:1052-5).
According to Dr. Mease, controlled trials of DMARDs in PsA patients have yielded treatment effects that range from marginal in the joints to marginal or none at all in the skin. Data from the Methotrexate in Psoriatic Arthritis trial conducted in the United Kingdom and published in 2012 showed no evidence that methotrexate improves inflammatory synovitis in active PsA (Rheumatol. 2012;51:1368-77).
“There were issues with this trial, including the fact that it took 5 years to enroll patients, and many dropped out, so I don’t think it’s a very reliable study,” said Dr. Mease, who is also a professor of medicine in the division of rheumatology at the University of Washington, Seattle. “Currently, Amgen is in the process of starting a trial in which the goal is to enroll 840 subjects with early PsA who are being randomized to methotrexate alone, Enbrel alone, or Enbrel plus methotrexate. This, I think, is going to give us a better answer about the effectiveness of methotrexate. It will also teach us about whether there’s a value in combining an anti-TNF inhibitor with methotrexate. We still don’t know the answer to that question.”
The most recent data on methotrexate come from an open-label trial known as TICOPA, which used a tight control treatment paradigm through 48 weeks of treatment. A subanalysis of 188 patients treated with methotrexate through 12 weeks was presented at the 2015 meeting of the European League Against Rheumatism. It revealed that 41% of patients achieved an ACR 20, 22% achieved minimal disease activity, 62% experienced an improvement in dactylitis, and 25% experienced an improvement in enthesitis.
“So we have a few data suggesting that methotrexate may be modestly effective in treating PsA,” Dr. Mease said. “We often will start with methotrexate unless the patient has really aggressive disease activity. If they get some effect from the drug but not enough, we’ll often add a biologic agent but often keep some methotrexate in the background, even at 10 mg per week, in order to reduce immunogenicity from a biologic.”
Dr. Mease disclosed that he has received research grants, consultation fees, and/or speaker honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Crescendo Bioscience, Genentech, GlaxoSmithKline, Janssen, Eli Lilly, Merck, Novartis, Pfizer, and UCB.
PARK CITY, UTAH – Since most people with psoriatic arthritis (PsA) develop psoriasis before joint symptoms, it’s helpful to have a simple screening test for the condition.
One of Dr. Philip Mease’s favorite PsA screening tools is the Psoriasis Epidemiology Screening Test (PEST), which was first described at the 2009 annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) and consists of five simple questions. They are: Have you ever had a swollen joint (or joints)? Has a doctor ever told you that you have arthritis? Do your fingernails or toes have holes or pits? Have you had pain in your heel? Have you had a finger or toe that was completely swollen and painful for no apparent reason? (Clin Exp Rheumatol. 2009;27:469-74).
“Just these five simple questions, or trying to remember a few of them, can help you in your review of systems,” Dr. Mease, director of arthritis research at Swedish Medical Center, Seattle, said at the annual meeting of the Pacific Dermatologic Association. “I think patients appreciate it when you look beyond the skin in your questioning. These can pick up [PsA] with a high sensitivity and specificity” of 0.92 and 0.78, respectively.
He went on to discuss current PsA treatment approaches. According to an evidence review that he and his associates in GRAPPA published in 2009, biologics (anti–tumor necrosis factor inhibitors) as a group were found to be effective in all five domains of the disease: peripheral arthritis, skin and nail disease, axial disease, dactylitis, and enthesitis, while the oral disease-modifying antirheumatic drugs (DMARDs) were effective for peripheral arthritis and skin and nail disease.
Other treatments that were found effective are: psoralen and UVA/UVB for skin and nail disease, physiotherapy for axial disease, intra-articular steroids for peripheral arthritis, and NSAIDs for peripheral arthritis and axial disease (J Rheum. 2009;33:1417-21). “Patients with mild disease can be tried on NSAIDs, especially in a patient with monoarticular disease, but for the most part we need to move on to using systemic medication,” he said. Updated recommendations from GRAPPA include new data regarding ustekinumab, apremilast, and secukinumab, as well as data on comorbidities (J Rheumatol. 2015;42[6]:1052-5).
According to Dr. Mease, controlled trials of DMARDs in PsA patients have yielded treatment effects that range from marginal in the joints to marginal or none at all in the skin. Data from the Methotrexate in Psoriatic Arthritis trial conducted in the United Kingdom and published in 2012 showed no evidence that methotrexate improves inflammatory synovitis in active PsA (Rheumatol. 2012;51:1368-77).
“There were issues with this trial, including the fact that it took 5 years to enroll patients, and many dropped out, so I don’t think it’s a very reliable study,” said Dr. Mease, who is also a professor of medicine in the division of rheumatology at the University of Washington, Seattle. “Currently, Amgen is in the process of starting a trial in which the goal is to enroll 840 subjects with early PsA who are being randomized to methotrexate alone, Enbrel alone, or Enbrel plus methotrexate. This, I think, is going to give us a better answer about the effectiveness of methotrexate. It will also teach us about whether there’s a value in combining an anti-TNF inhibitor with methotrexate. We still don’t know the answer to that question.”
The most recent data on methotrexate come from an open-label trial known as TICOPA, which used a tight control treatment paradigm through 48 weeks of treatment. A subanalysis of 188 patients treated with methotrexate through 12 weeks was presented at the 2015 meeting of the European League Against Rheumatism. It revealed that 41% of patients achieved an ACR 20, 22% achieved minimal disease activity, 62% experienced an improvement in dactylitis, and 25% experienced an improvement in enthesitis.
“So we have a few data suggesting that methotrexate may be modestly effective in treating PsA,” Dr. Mease said. “We often will start with methotrexate unless the patient has really aggressive disease activity. If they get some effect from the drug but not enough, we’ll often add a biologic agent but often keep some methotrexate in the background, even at 10 mg per week, in order to reduce immunogenicity from a biologic.”
Dr. Mease disclosed that he has received research grants, consultation fees, and/or speaker honoraria from AbbVie, Amgen, Bristol-Myers Squibb, Celgene, Crescendo Bioscience, Genentech, GlaxoSmithKline, Janssen, Eli Lilly, Merck, Novartis, Pfizer, and UCB.
EXPERT ANALYSIS AT PDA 2015
‘Troubling’ trend in narcotic prescribing for ankylosing spondylitis
DENVER – Even as prescribing of tumor necrosis factor inhibitors for patients with ankylosing spondylitis climbed in the last half-dozen years, patients’ use of prescription narcotics increased as well.
This was the unexpected finding of a comprehensive analysis of trends in medication use over a 12-year period among 1,018 ankylosing spondylitis patients participating in the ongoing multicenter Prospective Study of Outcomes in Ankylosing Spondylitis (PSOAS), Dr. Mary C. Gibson reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
“Even with treatment with TNF inhibitors, patients are often requiring additional pain control with narcotic medications. This could reflect inadequate pain control from directed therapy or a trend toward change in pain control use in general practice,” observed Dr. Gibson of the University of Texas, Houston.
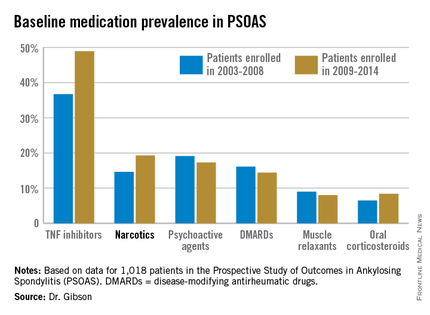
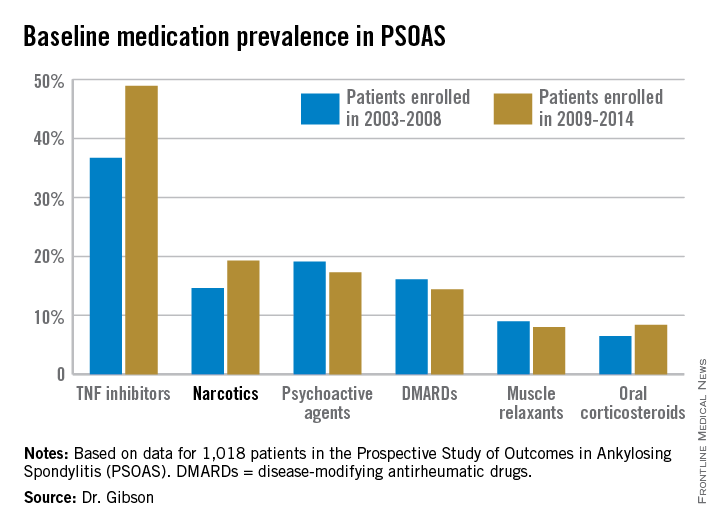
The patients enrolled in PSOAS during 2003-2014 are being followed longitudinally. They were recruited from community practice settings as well as academic centers. Dr. Gibson analyzed their medication use at the time of their baseline enrollment visit on a year-by-year basis, then stepped back to look at broader trends, comparing baseline medication use for patients enrolled in 2003-2008 to those enrolled during 2009-2014.
Not surprisingly, significantly more patients entered the study on a TNF inhibitor during the enrollment years 2009-2014 than during the first half of the study period: 49% compared with 37% during 2003-2008. What came as a surprise, however, was the time trend in narcotic use: 14.6% of ankylosing spondylitis patients in the 2003-2008 cohort were on narcotics, compared with 19.3% who enrolled since then.
“That’s a little bit troubling,” said Dr. John D. Reveille, professor and vice chair of rheumatology at the University of Texas, Houston, and head of PSOAS.
Oxycodone use nearly tripled between the first and second half of the study period: 1.1% were on the drug at enrollment in 2003-2008 compared with 3.1% in 2009-2014.
Of note, baseline use of oxycodone peaked in the enrollment class of 2012, 5.6% of whom were on the medication when they joined PSOAS. For narcotics overall, the peak year was 2008: over 27% of patients who enrolled in the study that year were on narcotic therapy at the time.
The use of NSAIDs – a guideline-recommended first-line therapy in ankylosing spondylitis – trended lower over time, albeit not significantly so. Among patients who enrolled in PSOAS during 2003-2008, over 70% were on NSAID therapy, compared with 66% of those who enrolled later.
There was no significant change over time in the use of DMARDs, psychoactive medications, muscle relaxants, or corticosteroids.
PSOAS is sponsored by the National Institutes of Health. Dr. Gibson reported having no financial conflicts.
DENVER – Even as prescribing of tumor necrosis factor inhibitors for patients with ankylosing spondylitis climbed in the last half-dozen years, patients’ use of prescription narcotics increased as well.
This was the unexpected finding of a comprehensive analysis of trends in medication use over a 12-year period among 1,018 ankylosing spondylitis patients participating in the ongoing multicenter Prospective Study of Outcomes in Ankylosing Spondylitis (PSOAS), Dr. Mary C. Gibson reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
“Even with treatment with TNF inhibitors, patients are often requiring additional pain control with narcotic medications. This could reflect inadequate pain control from directed therapy or a trend toward change in pain control use in general practice,” observed Dr. Gibson of the University of Texas, Houston.

The patients enrolled in PSOAS during 2003-2014 are being followed longitudinally. They were recruited from community practice settings as well as academic centers. Dr. Gibson analyzed their medication use at the time of their baseline enrollment visit on a year-by-year basis, then stepped back to look at broader trends, comparing baseline medication use for patients enrolled in 2003-2008 to those enrolled during 2009-2014.
Not surprisingly, significantly more patients entered the study on a TNF inhibitor during the enrollment years 2009-2014 than during the first half of the study period: 49% compared with 37% during 2003-2008. What came as a surprise, however, was the time trend in narcotic use: 14.6% of ankylosing spondylitis patients in the 2003-2008 cohort were on narcotics, compared with 19.3% who enrolled since then.
“That’s a little bit troubling,” said Dr. John D. Reveille, professor and vice chair of rheumatology at the University of Texas, Houston, and head of PSOAS.
Oxycodone use nearly tripled between the first and second half of the study period: 1.1% were on the drug at enrollment in 2003-2008 compared with 3.1% in 2009-2014.
Of note, baseline use of oxycodone peaked in the enrollment class of 2012, 5.6% of whom were on the medication when they joined PSOAS. For narcotics overall, the peak year was 2008: over 27% of patients who enrolled in the study that year were on narcotic therapy at the time.
The use of NSAIDs – a guideline-recommended first-line therapy in ankylosing spondylitis – trended lower over time, albeit not significantly so. Among patients who enrolled in PSOAS during 2003-2008, over 70% were on NSAID therapy, compared with 66% of those who enrolled later.
There was no significant change over time in the use of DMARDs, psychoactive medications, muscle relaxants, or corticosteroids.
PSOAS is sponsored by the National Institutes of Health. Dr. Gibson reported having no financial conflicts.
DENVER – Even as prescribing of tumor necrosis factor inhibitors for patients with ankylosing spondylitis climbed in the last half-dozen years, patients’ use of prescription narcotics increased as well.
This was the unexpected finding of a comprehensive analysis of trends in medication use over a 12-year period among 1,018 ankylosing spondylitis patients participating in the ongoing multicenter Prospective Study of Outcomes in Ankylosing Spondylitis (PSOAS), Dr. Mary C. Gibson reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
“Even with treatment with TNF inhibitors, patients are often requiring additional pain control with narcotic medications. This could reflect inadequate pain control from directed therapy or a trend toward change in pain control use in general practice,” observed Dr. Gibson of the University of Texas, Houston.

The patients enrolled in PSOAS during 2003-2014 are being followed longitudinally. They were recruited from community practice settings as well as academic centers. Dr. Gibson analyzed their medication use at the time of their baseline enrollment visit on a year-by-year basis, then stepped back to look at broader trends, comparing baseline medication use for patients enrolled in 2003-2008 to those enrolled during 2009-2014.
Not surprisingly, significantly more patients entered the study on a TNF inhibitor during the enrollment years 2009-2014 than during the first half of the study period: 49% compared with 37% during 2003-2008. What came as a surprise, however, was the time trend in narcotic use: 14.6% of ankylosing spondylitis patients in the 2003-2008 cohort were on narcotics, compared with 19.3% who enrolled since then.
“That’s a little bit troubling,” said Dr. John D. Reveille, professor and vice chair of rheumatology at the University of Texas, Houston, and head of PSOAS.
Oxycodone use nearly tripled between the first and second half of the study period: 1.1% were on the drug at enrollment in 2003-2008 compared with 3.1% in 2009-2014.
Of note, baseline use of oxycodone peaked in the enrollment class of 2012, 5.6% of whom were on the medication when they joined PSOAS. For narcotics overall, the peak year was 2008: over 27% of patients who enrolled in the study that year were on narcotic therapy at the time.
The use of NSAIDs – a guideline-recommended first-line therapy in ankylosing spondylitis – trended lower over time, albeit not significantly so. Among patients who enrolled in PSOAS during 2003-2008, over 70% were on NSAID therapy, compared with 66% of those who enrolled later.
There was no significant change over time in the use of DMARDs, psychoactive medications, muscle relaxants, or corticosteroids.
PSOAS is sponsored by the National Institutes of Health. Dr. Gibson reported having no financial conflicts.
AT THE 2015 SPARTAN ANNUAL MEETING
Key clinical point: The use of prescription narcotics by patients with ankylosing spondylitis has gone up during the past 12 years, even as their use of tumor necrosis factor inhibitor therapy has grown.
Major finding: Among a large cohort of patients with ankylosing spondylitis, 14.6% were on prescription narcotic therapy during 2003-2008, compared with 19.3% in 2009-2014.
Data source: Analysis of 1,018 patients with ankylosing spondylitis, from PSOAS – a prospective, multicenter, longitudinal cohort study.
Disclosures: The ongoing PSOAS study is funded by the National Institutes of Health. The presenter reported having no conflicts of interest.