User login
As psoriasis maintenance therapy, step-down matched full-dose etanercept for quality of life measures
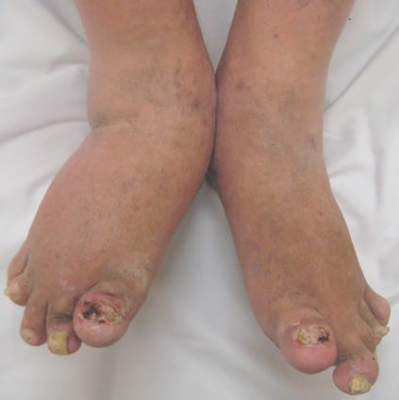
At 6 months, quality of life is largely the same whether psoriasis patients stay on their initial etanercept (Enbrel) dose of 50 mg twice weekly or step down to 50 mg once weekly with topical corticosteroids, according to a Canadian trial published in the Journal of The European Academy of Dermatology and Venereology.
“The opportunity to use a topical agent seemed to improve [patients’] overall satisfaction,” and weekly “etanercept and topical may be a less costly option compared with” etanercept twice weekly, said the investigators, led by Dr. Kim Papp, of Probity Medical Research in Waterloo, Ont.
Previously, the investigators showed that the step-down strategy works as well as full-dose maintenance in clearing the skin. They wanted to check if that also held true for quality of life, as assessed by the Dermatology Life Quality Index (DLQI) and the Treatment Satisfaction Questionnaire for Medication (TSQM). (J Eur Acad Dermatol Venereol. 2015 Aug;29(8):1555-61).
The 287 patients, all with moderate-to-severe plaque psoriasis for at least half a year, received etanercept 50 mg twice weekly for 12 weeks; 144 were then randomized to stay on that dose, and 143 others to drop to 50 mg weekly with the addition of topicals as required through week 24. The mean age of patients in the trial was 45 years; 88% of the subjects were white and 65% were men.
Topicals included hydrocortisone 2.5%, beta methasone valerate 0.1%, betamethasone dipropionate 0.05%, clobetasol 0.05%, calcitriol, or calcipotriol plus betamethasone dipropionate 0.05%; the topicals were selected by the researchers, who were allowed to change them as needed.
The mean change in DLQI from baseline to week 24 was 10.7 points in the etanercept group and 9.9 points in the etanercept-plus-topical group. Mean change in TSQM effectiveness, convenience, side-effects, and global satisfaction was 27.1, 14.8, -0.7 and 26.7 points in the etanercept group, and 32.5, 18.5, 1.3, and 28.4 points in the etanercept-plus-topical group. Healthcare visits, employment status, work productivity, and ability to perform daily activities were similar between the treatment arms.
The study was descriptive; the investigators didn’t run statistical analyses. Even so, measures of quality of life “were numerically similar in patients who stayed on etanercept 50 mg [twice weekly] and patients who received etanercept 50 mg [weekly] plus topical therapies. No notable differences between treatment arms ... were observed. Additionally, improvements in [Psoriasis Area and Severity Index] scores appeared to correlate with improvements in patient reported outcomes,” the investigators said.
“The duration of treatment with topical therapies was only 12 weeks; a longer duration may result in loss of efficacy as adherence to topical therapies may decrease with time,” they noted.
The work was funded by etanercept’s maker, Amgen. Dr. Papp reported consulting for the company. Two other investigators have been Amgen advisors and reported research grants, speakers fees, or other payments.
At 6 months, quality of life is largely the same whether psoriasis patients stay on their initial etanercept (Enbrel) dose of 50 mg twice weekly or step down to 50 mg once weekly with topical corticosteroids, according to a Canadian trial published in the Journal of The European Academy of Dermatology and Venereology.
“The opportunity to use a topical agent seemed to improve [patients’] overall satisfaction,” and weekly “etanercept and topical may be a less costly option compared with” etanercept twice weekly, said the investigators, led by Dr. Kim Papp, of Probity Medical Research in Waterloo, Ont.
Previously, the investigators showed that the step-down strategy works as well as full-dose maintenance in clearing the skin. They wanted to check if that also held true for quality of life, as assessed by the Dermatology Life Quality Index (DLQI) and the Treatment Satisfaction Questionnaire for Medication (TSQM). (J Eur Acad Dermatol Venereol. 2015 Aug;29(8):1555-61).
The 287 patients, all with moderate-to-severe plaque psoriasis for at least half a year, received etanercept 50 mg twice weekly for 12 weeks; 144 were then randomized to stay on that dose, and 143 others to drop to 50 mg weekly with the addition of topicals as required through week 24. The mean age of patients in the trial was 45 years; 88% of the subjects were white and 65% were men.
Topicals included hydrocortisone 2.5%, beta methasone valerate 0.1%, betamethasone dipropionate 0.05%, clobetasol 0.05%, calcitriol, or calcipotriol plus betamethasone dipropionate 0.05%; the topicals were selected by the researchers, who were allowed to change them as needed.
The mean change in DLQI from baseline to week 24 was 10.7 points in the etanercept group and 9.9 points in the etanercept-plus-topical group. Mean change in TSQM effectiveness, convenience, side-effects, and global satisfaction was 27.1, 14.8, -0.7 and 26.7 points in the etanercept group, and 32.5, 18.5, 1.3, and 28.4 points in the etanercept-plus-topical group. Healthcare visits, employment status, work productivity, and ability to perform daily activities were similar between the treatment arms.
The study was descriptive; the investigators didn’t run statistical analyses. Even so, measures of quality of life “were numerically similar in patients who stayed on etanercept 50 mg [twice weekly] and patients who received etanercept 50 mg [weekly] plus topical therapies. No notable differences between treatment arms ... were observed. Additionally, improvements in [Psoriasis Area and Severity Index] scores appeared to correlate with improvements in patient reported outcomes,” the investigators said.
“The duration of treatment with topical therapies was only 12 weeks; a longer duration may result in loss of efficacy as adherence to topical therapies may decrease with time,” they noted.
The work was funded by etanercept’s maker, Amgen. Dr. Papp reported consulting for the company. Two other investigators have been Amgen advisors and reported research grants, speakers fees, or other payments.
At 6 months, quality of life is largely the same whether psoriasis patients stay on their initial etanercept (Enbrel) dose of 50 mg twice weekly or step down to 50 mg once weekly with topical corticosteroids, according to a Canadian trial published in the Journal of The European Academy of Dermatology and Venereology.
“The opportunity to use a topical agent seemed to improve [patients’] overall satisfaction,” and weekly “etanercept and topical may be a less costly option compared with” etanercept twice weekly, said the investigators, led by Dr. Kim Papp, of Probity Medical Research in Waterloo, Ont.
Previously, the investigators showed that the step-down strategy works as well as full-dose maintenance in clearing the skin. They wanted to check if that also held true for quality of life, as assessed by the Dermatology Life Quality Index (DLQI) and the Treatment Satisfaction Questionnaire for Medication (TSQM). (J Eur Acad Dermatol Venereol. 2015 Aug;29(8):1555-61).
The 287 patients, all with moderate-to-severe plaque psoriasis for at least half a year, received etanercept 50 mg twice weekly for 12 weeks; 144 were then randomized to stay on that dose, and 143 others to drop to 50 mg weekly with the addition of topicals as required through week 24. The mean age of patients in the trial was 45 years; 88% of the subjects were white and 65% were men.
Topicals included hydrocortisone 2.5%, beta methasone valerate 0.1%, betamethasone dipropionate 0.05%, clobetasol 0.05%, calcitriol, or calcipotriol plus betamethasone dipropionate 0.05%; the topicals were selected by the researchers, who were allowed to change them as needed.
The mean change in DLQI from baseline to week 24 was 10.7 points in the etanercept group and 9.9 points in the etanercept-plus-topical group. Mean change in TSQM effectiveness, convenience, side-effects, and global satisfaction was 27.1, 14.8, -0.7 and 26.7 points in the etanercept group, and 32.5, 18.5, 1.3, and 28.4 points in the etanercept-plus-topical group. Healthcare visits, employment status, work productivity, and ability to perform daily activities were similar between the treatment arms.
The study was descriptive; the investigators didn’t run statistical analyses. Even so, measures of quality of life “were numerically similar in patients who stayed on etanercept 50 mg [twice weekly] and patients who received etanercept 50 mg [weekly] plus topical therapies. No notable differences between treatment arms ... were observed. Additionally, improvements in [Psoriasis Area and Severity Index] scores appeared to correlate with improvements in patient reported outcomes,” the investigators said.
“The duration of treatment with topical therapies was only 12 weeks; a longer duration may result in loss of efficacy as adherence to topical therapies may decrease with time,” they noted.
The work was funded by etanercept’s maker, Amgen. Dr. Papp reported consulting for the company. Two other investigators have been Amgen advisors and reported research grants, speakers fees, or other payments.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEROLOGY
Exercise boosts medication’s benefits in ankylosing spondylitis
DENVER – Recreational exercise and tumor necrosis factor–inhibitor therapy provide synergistic improvement in the functional status of patients with ankylosing spondylitis, according to an analysis from the multicenter Prospective Study of Outcomes in Ankylosing Spondylitis (PSOAS).
Among 683 PSOAS participants prospectively followed for up to 12 years, those who consistently engaged in moderate recreational exercise, defined as at least 120 minutes of exercise per week, had significantly lower, more favorable Bath Ankylosing Spondylitis Functional Index (BASFI) scores during years 1-5 than did those who exercised less, even after controlling for baseline function and other potential confounders in a multivariate analysis, Dr. Lianne S. Gensler said at the annual meeting of the Spondyloarthritis Research and Treatment Network.
Additionally, a significant interaction was observed between exercise time and anti-TNF therapy. BASFI scores improved through 5 years of follow-up in TNF inhibitor users regardless of their exercise status, but the impact on functional status was “strikingly” greater in those patients on a TNF inhibitor who engaged in at least 120 minutes of exercise per week, according to Dr. Gensler of the University of California, San Francisco.
“This suggests that it’s not simply taking a TNF inhibitor that’s going to promote long-term function. These patients should also be exercising to synergize and get the best out of their therapy,” she emphasized.
The PSOAS cohort included averaged 43 years of age at baseline, with a mean 19-year disease duration and an average of 2.1 comorbid conditions. In a multivariate longitudinal analysis, several factors emerged as significant predictors of more pronounced functional impairment over time: being age 40 or older at baseline was associated with a 25% increased risk, opiate use was associated with a 12% increase in risk, a baseline Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score of at least 40 conferred a 19% increase in risk, as did a modified Stoke Ankylosing Spondylitis Spinal Score of 4 or greater.
Dr. Gensler drew a sharp distinction between the effects of moderate recreational exercise and occupational physical activity in ankylosing spondylitis patients. In an earlier landmark study involving 397 members of the PSOAS cohort with at least a 20-year history of ankylosing spondylitis, investigators mapped the patients’ work histories, the physical activities their work entailed, and the time they spent at each activity. Jobs that involved repeated bending, stretching, twisting, and vibration exposure were associated with worse BASFI scores and greater radiographic disease severity (Arthritis Rheum. 2008 Jun 15;59[6]:822-32).
Taking a step back to summarize what’s known today about what affects outcomes in ankylosing spondylitis patients, Dr. Gensler listed in the ‘bad’ column smoking, ongoing inflammation, baseline damage to the spine, certain occupational activities involving biomechanical stress, adiposity, and low socioeconomic status.
What’s good for patients with ankylosing spondylitis is physical therapy and back exercises – both will receive favorable marks in soon-to-be-published American College of Rheumatology guidelines on the management of ankylosing spondylitis that Dr. Gensler worked on – as well as TNF-inhibitor therapy, NSAIDs, and recreational exercise, which wasn’t addressed in the former guidelines.
Dr. Gensler reported receiving research funds from or serving as a consultant to AbbVie, Amgen, Celgene, Janssen, and UCB.
DENVER – Recreational exercise and tumor necrosis factor–inhibitor therapy provide synergistic improvement in the functional status of patients with ankylosing spondylitis, according to an analysis from the multicenter Prospective Study of Outcomes in Ankylosing Spondylitis (PSOAS).
Among 683 PSOAS participants prospectively followed for up to 12 years, those who consistently engaged in moderate recreational exercise, defined as at least 120 minutes of exercise per week, had significantly lower, more favorable Bath Ankylosing Spondylitis Functional Index (BASFI) scores during years 1-5 than did those who exercised less, even after controlling for baseline function and other potential confounders in a multivariate analysis, Dr. Lianne S. Gensler said at the annual meeting of the Spondyloarthritis Research and Treatment Network.
Additionally, a significant interaction was observed between exercise time and anti-TNF therapy. BASFI scores improved through 5 years of follow-up in TNF inhibitor users regardless of their exercise status, but the impact on functional status was “strikingly” greater in those patients on a TNF inhibitor who engaged in at least 120 minutes of exercise per week, according to Dr. Gensler of the University of California, San Francisco.
“This suggests that it’s not simply taking a TNF inhibitor that’s going to promote long-term function. These patients should also be exercising to synergize and get the best out of their therapy,” she emphasized.
The PSOAS cohort included averaged 43 years of age at baseline, with a mean 19-year disease duration and an average of 2.1 comorbid conditions. In a multivariate longitudinal analysis, several factors emerged as significant predictors of more pronounced functional impairment over time: being age 40 or older at baseline was associated with a 25% increased risk, opiate use was associated with a 12% increase in risk, a baseline Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score of at least 40 conferred a 19% increase in risk, as did a modified Stoke Ankylosing Spondylitis Spinal Score of 4 or greater.
Dr. Gensler drew a sharp distinction between the effects of moderate recreational exercise and occupational physical activity in ankylosing spondylitis patients. In an earlier landmark study involving 397 members of the PSOAS cohort with at least a 20-year history of ankylosing spondylitis, investigators mapped the patients’ work histories, the physical activities their work entailed, and the time they spent at each activity. Jobs that involved repeated bending, stretching, twisting, and vibration exposure were associated with worse BASFI scores and greater radiographic disease severity (Arthritis Rheum. 2008 Jun 15;59[6]:822-32).
Taking a step back to summarize what’s known today about what affects outcomes in ankylosing spondylitis patients, Dr. Gensler listed in the ‘bad’ column smoking, ongoing inflammation, baseline damage to the spine, certain occupational activities involving biomechanical stress, adiposity, and low socioeconomic status.
What’s good for patients with ankylosing spondylitis is physical therapy and back exercises – both will receive favorable marks in soon-to-be-published American College of Rheumatology guidelines on the management of ankylosing spondylitis that Dr. Gensler worked on – as well as TNF-inhibitor therapy, NSAIDs, and recreational exercise, which wasn’t addressed in the former guidelines.
Dr. Gensler reported receiving research funds from or serving as a consultant to AbbVie, Amgen, Celgene, Janssen, and UCB.
DENVER – Recreational exercise and tumor necrosis factor–inhibitor therapy provide synergistic improvement in the functional status of patients with ankylosing spondylitis, according to an analysis from the multicenter Prospective Study of Outcomes in Ankylosing Spondylitis (PSOAS).
Among 683 PSOAS participants prospectively followed for up to 12 years, those who consistently engaged in moderate recreational exercise, defined as at least 120 minutes of exercise per week, had significantly lower, more favorable Bath Ankylosing Spondylitis Functional Index (BASFI) scores during years 1-5 than did those who exercised less, even after controlling for baseline function and other potential confounders in a multivariate analysis, Dr. Lianne S. Gensler said at the annual meeting of the Spondyloarthritis Research and Treatment Network.
Additionally, a significant interaction was observed between exercise time and anti-TNF therapy. BASFI scores improved through 5 years of follow-up in TNF inhibitor users regardless of their exercise status, but the impact on functional status was “strikingly” greater in those patients on a TNF inhibitor who engaged in at least 120 minutes of exercise per week, according to Dr. Gensler of the University of California, San Francisco.
“This suggests that it’s not simply taking a TNF inhibitor that’s going to promote long-term function. These patients should also be exercising to synergize and get the best out of their therapy,” she emphasized.
The PSOAS cohort included averaged 43 years of age at baseline, with a mean 19-year disease duration and an average of 2.1 comorbid conditions. In a multivariate longitudinal analysis, several factors emerged as significant predictors of more pronounced functional impairment over time: being age 40 or older at baseline was associated with a 25% increased risk, opiate use was associated with a 12% increase in risk, a baseline Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score of at least 40 conferred a 19% increase in risk, as did a modified Stoke Ankylosing Spondylitis Spinal Score of 4 or greater.
Dr. Gensler drew a sharp distinction between the effects of moderate recreational exercise and occupational physical activity in ankylosing spondylitis patients. In an earlier landmark study involving 397 members of the PSOAS cohort with at least a 20-year history of ankylosing spondylitis, investigators mapped the patients’ work histories, the physical activities their work entailed, and the time they spent at each activity. Jobs that involved repeated bending, stretching, twisting, and vibration exposure were associated with worse BASFI scores and greater radiographic disease severity (Arthritis Rheum. 2008 Jun 15;59[6]:822-32).
Taking a step back to summarize what’s known today about what affects outcomes in ankylosing spondylitis patients, Dr. Gensler listed in the ‘bad’ column smoking, ongoing inflammation, baseline damage to the spine, certain occupational activities involving biomechanical stress, adiposity, and low socioeconomic status.
What’s good for patients with ankylosing spondylitis is physical therapy and back exercises – both will receive favorable marks in soon-to-be-published American College of Rheumatology guidelines on the management of ankylosing spondylitis that Dr. Gensler worked on – as well as TNF-inhibitor therapy, NSAIDs, and recreational exercise, which wasn’t addressed in the former guidelines.
Dr. Gensler reported receiving research funds from or serving as a consultant to AbbVie, Amgen, Celgene, Janssen, and UCB.
EXPERT ANALYSIS FROM THE 2015 SPARTAN ANNUAL MEETING
New era ahead in spondyloarthritis therapy
DENVER – The emerging new era in the treatment of spondyloarthritis is combined blockade of tumor necrosis factor–alpha (TNF-alpha) and interleukin-17 using a bispecific antibody, Dr. Siba P. Raychaudhuri predicted at the annual meeting of the Spondyloarthritis Research and Treatment Network.
“This is the next generation of antibody therapies for spondyloarthritis,” said Dr. Raychaudhuri, a rheumatologist at the University of California, Davis.
This novel approach has a sound theoretical basis. Moreover, studies in mouse models, including models of rheumatoid arthritis (Arthritis Rheumatol. 2015 Jan;67[1]:51-62), have been positive. And proprietary bispecific anti-TNF-alpha/IL-17 antibodies are now in phase I and phase II clinical trials for various forms of spondyloarthritis, he noted.
The impetus for development of this new therapy is widespread agreement that while TNF inhibition has been a step forward in the treatment of axial spondyloarthritis, the efficacy is less than hoped for with these potent and costly agents.
“Things have been sort of gray for almost the last 10 years. All the evidence suggests we’re not getting full efficacy with anti-TNF therapy. We’re getting stuck at about a 60% ASAS 20 response,” he observed.
The explanation for this merely middling success rate appears to be that TNF inhibition induces a counterregulatory uptick in the IL-17 pathway. And in animal models, IL-17 is a key mediator of joint inflammation and destruction.
Dr. Raychaudhuri noted that brodalumab, an investigational human monoclonal antibody directed against IL-17 receptor A, has been shown “reasonably effective” in a phase II study in patients with psoriatic arthritis, which is part of the spondyloarthritis family of autoimmune diseases, achieving an ACR 20 response rate of 39% at week 12 in the high-dose arm (N Engl J Med. 2014 Jun 12;370[24]:2295-306).
The expectation is that by inhibiting both TNF-alpha and IL-17 using a bispecific protein composed of fragments of two different monoclonal antibodies, the result will be reduced growth and differentiation of proinflammatory T cells, decreased T cell migration, and synergistic clinical efficacy, compared with what’s attainable by addressing either target alone.
Dr. Raychaudhuri reported receiving research grants from or serving as a consultant to Abbott, Amgen, Celgene, UCB, and Janssen Biotech.
DENVER – The emerging new era in the treatment of spondyloarthritis is combined blockade of tumor necrosis factor–alpha (TNF-alpha) and interleukin-17 using a bispecific antibody, Dr. Siba P. Raychaudhuri predicted at the annual meeting of the Spondyloarthritis Research and Treatment Network.
“This is the next generation of antibody therapies for spondyloarthritis,” said Dr. Raychaudhuri, a rheumatologist at the University of California, Davis.
This novel approach has a sound theoretical basis. Moreover, studies in mouse models, including models of rheumatoid arthritis (Arthritis Rheumatol. 2015 Jan;67[1]:51-62), have been positive. And proprietary bispecific anti-TNF-alpha/IL-17 antibodies are now in phase I and phase II clinical trials for various forms of spondyloarthritis, he noted.
The impetus for development of this new therapy is widespread agreement that while TNF inhibition has been a step forward in the treatment of axial spondyloarthritis, the efficacy is less than hoped for with these potent and costly agents.
“Things have been sort of gray for almost the last 10 years. All the evidence suggests we’re not getting full efficacy with anti-TNF therapy. We’re getting stuck at about a 60% ASAS 20 response,” he observed.
The explanation for this merely middling success rate appears to be that TNF inhibition induces a counterregulatory uptick in the IL-17 pathway. And in animal models, IL-17 is a key mediator of joint inflammation and destruction.
Dr. Raychaudhuri noted that brodalumab, an investigational human monoclonal antibody directed against IL-17 receptor A, has been shown “reasonably effective” in a phase II study in patients with psoriatic arthritis, which is part of the spondyloarthritis family of autoimmune diseases, achieving an ACR 20 response rate of 39% at week 12 in the high-dose arm (N Engl J Med. 2014 Jun 12;370[24]:2295-306).
The expectation is that by inhibiting both TNF-alpha and IL-17 using a bispecific protein composed of fragments of two different monoclonal antibodies, the result will be reduced growth and differentiation of proinflammatory T cells, decreased T cell migration, and synergistic clinical efficacy, compared with what’s attainable by addressing either target alone.
Dr. Raychaudhuri reported receiving research grants from or serving as a consultant to Abbott, Amgen, Celgene, UCB, and Janssen Biotech.
DENVER – The emerging new era in the treatment of spondyloarthritis is combined blockade of tumor necrosis factor–alpha (TNF-alpha) and interleukin-17 using a bispecific antibody, Dr. Siba P. Raychaudhuri predicted at the annual meeting of the Spondyloarthritis Research and Treatment Network.
“This is the next generation of antibody therapies for spondyloarthritis,” said Dr. Raychaudhuri, a rheumatologist at the University of California, Davis.
This novel approach has a sound theoretical basis. Moreover, studies in mouse models, including models of rheumatoid arthritis (Arthritis Rheumatol. 2015 Jan;67[1]:51-62), have been positive. And proprietary bispecific anti-TNF-alpha/IL-17 antibodies are now in phase I and phase II clinical trials for various forms of spondyloarthritis, he noted.
The impetus for development of this new therapy is widespread agreement that while TNF inhibition has been a step forward in the treatment of axial spondyloarthritis, the efficacy is less than hoped for with these potent and costly agents.
“Things have been sort of gray for almost the last 10 years. All the evidence suggests we’re not getting full efficacy with anti-TNF therapy. We’re getting stuck at about a 60% ASAS 20 response,” he observed.
The explanation for this merely middling success rate appears to be that TNF inhibition induces a counterregulatory uptick in the IL-17 pathway. And in animal models, IL-17 is a key mediator of joint inflammation and destruction.
Dr. Raychaudhuri noted that brodalumab, an investigational human monoclonal antibody directed against IL-17 receptor A, has been shown “reasonably effective” in a phase II study in patients with psoriatic arthritis, which is part of the spondyloarthritis family of autoimmune diseases, achieving an ACR 20 response rate of 39% at week 12 in the high-dose arm (N Engl J Med. 2014 Jun 12;370[24]:2295-306).
The expectation is that by inhibiting both TNF-alpha and IL-17 using a bispecific protein composed of fragments of two different monoclonal antibodies, the result will be reduced growth and differentiation of proinflammatory T cells, decreased T cell migration, and synergistic clinical efficacy, compared with what’s attainable by addressing either target alone.
Dr. Raychaudhuri reported receiving research grants from or serving as a consultant to Abbott, Amgen, Celgene, UCB, and Janssen Biotech.
EXPERT ANALYSIS FROM THE 2015 SPARTAN ANNUAL MEETING
New spondyloarthritis criteria in the works
DENVER – The Spondyloarthritis Research and Treatment Network has voted decisively to create new classification criteria for axial spondyloarthritis, including both ankylosing spondylitis and nonradiographic axial spondyloarthritis.
Following the vote, in which 64% of SPARTAN members attending the group’s annual meeting voted that “this is the right time to proceed,” comoderator Dr. Liron Caplan declared, “This is a very strong push. We’ll need to begin work on our proposal immediately.”
The detailed proposal will contain the justification for new classification criteria as well as outline how SPARTAN will go about creating the criteria in an evidence-based manner with input and financing from industry and patient support groups. The proposal needs to be filed by next March in order to be considered for roughly $135,000 in joint funding from the American College of Rheumatology and the European League Against Rheumatism.
A major impetus for the ambitious project was a frank conversation SPARTAN officers had in the Spring of 2014 with officials at the Food and Drug Administration. The rheumatologists sought an explanation for the FDA’s seemingly counterintuitive rejection of adalimumab (Humira) and certolizumab pegol (Cimzia) for treatment of nonradiographic axial spondyloarthritis (nr-axSpA) on the basis of the very same randomized, controlled trials that earlier resulted in approval for that indication by the European Medicines Agency.
The FDA representatives deemed the Assessment of SpondyloArthritis Society (ASAS) classification criteria (Ann Rheum Dis. 2009;68:777-83) used in those and other SpA trials to be overly broad. They felt the ASAS criteria lack sufficient sensitivity and specificity to ensure that large numbers of patients with fibromyalgia – a far more common disorder – wouldn’t be misclassified as having SpA and treated inappropriately with biologics, which aren’t effective in fibromyalgia, explained Dr. John D. Reveille, professor and vice-chair of rheumatology at the University of Texas, Houston, who attended the meeting.
“The only physicians who can prescribe an approved anti-TNF drug or other biologic in Europe are rheumatologists. The reason the FDA was nervous about approving drugs using the ASAS criteria is because any physician in the U.S. can prescribe those drugs once they’re approved. So the authorities made a regulatory decision about who the people are who are educated or not educated in the diagnosis and management of patients with SpA,” according to Dr. Michael H. Weisman, chair and director of rheumatology at Cedars-Sinai Medical Center in Los Angeles.
Dr. Caplan of the University of Colorado, Denver, observed that developing new and improved criteria for SpA will be a multiyear endeavor. A top goal is to shed light on the natural history of nr-axSpA, about which almost nothing is known. This will probably entail developing a sizable cohort of patients with nr-axSpA, following them for 5 years or so to see who develops ankylosing spondylitis, then looking back to identify reliable predictors.
The SPARTAN vote was preceded by a formal debate on the merits of replacing the ASAS criteria. Dr. Muhammad Asim Khan, professor of medicine at Case Western Reserve University, Cleveland, who 30 years ago was a pioneer in the concept of spondylitic disease without x-ray evidence of sacroiliitis, argued that the ASAS criteria aren’t adequately doing what disease classification criteria are supposed to do: provide a simple, clear means of identifying a homogeneous population for clinical trials. By letting in substantial numbers of patients with fibromyalgia, the criteria water down the clinical outcomes of investigational therapies for SpA.
The opposing argument was provided by ASAS representative Dr. Filip Van den Bosch. He conceded the ASAS classification criteria “aren’t perfect,” but over time they have become familiar to rheumatologists worldwide.
“Modifying the criteria will lead to years of uncertainty,” argued Dr. Van den Bosch, professor of rheumatology at Ghent (Belgium) University.
If, as some have proposed, SPARTAN were to tighten the ASAS criteria by insisting that a patient lacking x-ray evidence of sacroiliitis must possess three or four SpA features instead of the two now required, the result would be a drastic reduction in the sensitivity of the criteria, he continued.
“The challenge of making a correct diagnosis of axSpA cannot be solved by modifying the ASAS criteria. I cannot think of a new SpA feature that’s been identified in the last 5 years that could be added on top of what’s already in there,” the rheumatologist said.
What’s needed, he added, is a concerted effort to better educate physicians about axSpA, including interpretation of spinal x-rays and MRIs, the disease’s signs and symptoms, and how to differentiate SpA from other disorders that cause back pain.
While nearly two-thirds of the SPARTAN membership voted in favor of developing new classification criteria, some prominent voices were raised in dissent.
“I agree with Filip,” said Dr. Weisman. “The problem is not with the criteria, it’s who’s using them.”
Dr. Robert D. Inman, professor of medicine at the University of Toronto, predicted “it’s an exercise in futility” to try to come up with a single set of classification criteria that will provide the high degree of sensitivity that patients and clinicians want so that people who present with early axSpA are not missed while at the same time providing the maximum specificity that payers and regulatory officials seek.
Dr. Sindhu Johnson, who as cochair of the ACR Classification Criteria Subcommittee has shepherded the development of new criteria for a number of rheumatologic diseases, offered the SPARTAN group some practical tips. Because European studies tend to be comprised of monoethnic cohorts, the ACR will look favorably upon a successful SPARTAN effort to incorporate African Americans, Asians, Native Americans, and other minorities in their study cohorts. And SPARTAN should be open and receptive to critical feedback from stakeholders during the criteria development process.
“Get buy in. There’s no point in developing criteria that are extremely rigorous scientifically but that the global community feels are useless, that use tests they don’t have access to. This is one of the criticisms around the Sjögren’s criteria: they require ophthalmologic testing and salivary gland biopsy, and for rheumatologists – even academic rheumatologists at recruiting centers for clinical trials – they don’t have access to this, or it takes too much time. You need to come up with something pragmatic to get buy in,” advised Dr. Johnson of the University of Toronto.
By the way, she added, if the project wins ACR and EULAR funding it can only be named an ACR/EULAR product. “No other organization can be named in the headline,” according to Dr. Johnson.
DENVER – The Spondyloarthritis Research and Treatment Network has voted decisively to create new classification criteria for axial spondyloarthritis, including both ankylosing spondylitis and nonradiographic axial spondyloarthritis.
Following the vote, in which 64% of SPARTAN members attending the group’s annual meeting voted that “this is the right time to proceed,” comoderator Dr. Liron Caplan declared, “This is a very strong push. We’ll need to begin work on our proposal immediately.”
The detailed proposal will contain the justification for new classification criteria as well as outline how SPARTAN will go about creating the criteria in an evidence-based manner with input and financing from industry and patient support groups. The proposal needs to be filed by next March in order to be considered for roughly $135,000 in joint funding from the American College of Rheumatology and the European League Against Rheumatism.
A major impetus for the ambitious project was a frank conversation SPARTAN officers had in the Spring of 2014 with officials at the Food and Drug Administration. The rheumatologists sought an explanation for the FDA’s seemingly counterintuitive rejection of adalimumab (Humira) and certolizumab pegol (Cimzia) for treatment of nonradiographic axial spondyloarthritis (nr-axSpA) on the basis of the very same randomized, controlled trials that earlier resulted in approval for that indication by the European Medicines Agency.
The FDA representatives deemed the Assessment of SpondyloArthritis Society (ASAS) classification criteria (Ann Rheum Dis. 2009;68:777-83) used in those and other SpA trials to be overly broad. They felt the ASAS criteria lack sufficient sensitivity and specificity to ensure that large numbers of patients with fibromyalgia – a far more common disorder – wouldn’t be misclassified as having SpA and treated inappropriately with biologics, which aren’t effective in fibromyalgia, explained Dr. John D. Reveille, professor and vice-chair of rheumatology at the University of Texas, Houston, who attended the meeting.
“The only physicians who can prescribe an approved anti-TNF drug or other biologic in Europe are rheumatologists. The reason the FDA was nervous about approving drugs using the ASAS criteria is because any physician in the U.S. can prescribe those drugs once they’re approved. So the authorities made a regulatory decision about who the people are who are educated or not educated in the diagnosis and management of patients with SpA,” according to Dr. Michael H. Weisman, chair and director of rheumatology at Cedars-Sinai Medical Center in Los Angeles.
Dr. Caplan of the University of Colorado, Denver, observed that developing new and improved criteria for SpA will be a multiyear endeavor. A top goal is to shed light on the natural history of nr-axSpA, about which almost nothing is known. This will probably entail developing a sizable cohort of patients with nr-axSpA, following them for 5 years or so to see who develops ankylosing spondylitis, then looking back to identify reliable predictors.
The SPARTAN vote was preceded by a formal debate on the merits of replacing the ASAS criteria. Dr. Muhammad Asim Khan, professor of medicine at Case Western Reserve University, Cleveland, who 30 years ago was a pioneer in the concept of spondylitic disease without x-ray evidence of sacroiliitis, argued that the ASAS criteria aren’t adequately doing what disease classification criteria are supposed to do: provide a simple, clear means of identifying a homogeneous population for clinical trials. By letting in substantial numbers of patients with fibromyalgia, the criteria water down the clinical outcomes of investigational therapies for SpA.
The opposing argument was provided by ASAS representative Dr. Filip Van den Bosch. He conceded the ASAS classification criteria “aren’t perfect,” but over time they have become familiar to rheumatologists worldwide.
“Modifying the criteria will lead to years of uncertainty,” argued Dr. Van den Bosch, professor of rheumatology at Ghent (Belgium) University.
If, as some have proposed, SPARTAN were to tighten the ASAS criteria by insisting that a patient lacking x-ray evidence of sacroiliitis must possess three or four SpA features instead of the two now required, the result would be a drastic reduction in the sensitivity of the criteria, he continued.
“The challenge of making a correct diagnosis of axSpA cannot be solved by modifying the ASAS criteria. I cannot think of a new SpA feature that’s been identified in the last 5 years that could be added on top of what’s already in there,” the rheumatologist said.
What’s needed, he added, is a concerted effort to better educate physicians about axSpA, including interpretation of spinal x-rays and MRIs, the disease’s signs and symptoms, and how to differentiate SpA from other disorders that cause back pain.
While nearly two-thirds of the SPARTAN membership voted in favor of developing new classification criteria, some prominent voices were raised in dissent.
“I agree with Filip,” said Dr. Weisman. “The problem is not with the criteria, it’s who’s using them.”
Dr. Robert D. Inman, professor of medicine at the University of Toronto, predicted “it’s an exercise in futility” to try to come up with a single set of classification criteria that will provide the high degree of sensitivity that patients and clinicians want so that people who present with early axSpA are not missed while at the same time providing the maximum specificity that payers and regulatory officials seek.
Dr. Sindhu Johnson, who as cochair of the ACR Classification Criteria Subcommittee has shepherded the development of new criteria for a number of rheumatologic diseases, offered the SPARTAN group some practical tips. Because European studies tend to be comprised of monoethnic cohorts, the ACR will look favorably upon a successful SPARTAN effort to incorporate African Americans, Asians, Native Americans, and other minorities in their study cohorts. And SPARTAN should be open and receptive to critical feedback from stakeholders during the criteria development process.
“Get buy in. There’s no point in developing criteria that are extremely rigorous scientifically but that the global community feels are useless, that use tests they don’t have access to. This is one of the criticisms around the Sjögren’s criteria: they require ophthalmologic testing and salivary gland biopsy, and for rheumatologists – even academic rheumatologists at recruiting centers for clinical trials – they don’t have access to this, or it takes too much time. You need to come up with something pragmatic to get buy in,” advised Dr. Johnson of the University of Toronto.
By the way, she added, if the project wins ACR and EULAR funding it can only be named an ACR/EULAR product. “No other organization can be named in the headline,” according to Dr. Johnson.
DENVER – The Spondyloarthritis Research and Treatment Network has voted decisively to create new classification criteria for axial spondyloarthritis, including both ankylosing spondylitis and nonradiographic axial spondyloarthritis.
Following the vote, in which 64% of SPARTAN members attending the group’s annual meeting voted that “this is the right time to proceed,” comoderator Dr. Liron Caplan declared, “This is a very strong push. We’ll need to begin work on our proposal immediately.”
The detailed proposal will contain the justification for new classification criteria as well as outline how SPARTAN will go about creating the criteria in an evidence-based manner with input and financing from industry and patient support groups. The proposal needs to be filed by next March in order to be considered for roughly $135,000 in joint funding from the American College of Rheumatology and the European League Against Rheumatism.
A major impetus for the ambitious project was a frank conversation SPARTAN officers had in the Spring of 2014 with officials at the Food and Drug Administration. The rheumatologists sought an explanation for the FDA’s seemingly counterintuitive rejection of adalimumab (Humira) and certolizumab pegol (Cimzia) for treatment of nonradiographic axial spondyloarthritis (nr-axSpA) on the basis of the very same randomized, controlled trials that earlier resulted in approval for that indication by the European Medicines Agency.
The FDA representatives deemed the Assessment of SpondyloArthritis Society (ASAS) classification criteria (Ann Rheum Dis. 2009;68:777-83) used in those and other SpA trials to be overly broad. They felt the ASAS criteria lack sufficient sensitivity and specificity to ensure that large numbers of patients with fibromyalgia – a far more common disorder – wouldn’t be misclassified as having SpA and treated inappropriately with biologics, which aren’t effective in fibromyalgia, explained Dr. John D. Reveille, professor and vice-chair of rheumatology at the University of Texas, Houston, who attended the meeting.
“The only physicians who can prescribe an approved anti-TNF drug or other biologic in Europe are rheumatologists. The reason the FDA was nervous about approving drugs using the ASAS criteria is because any physician in the U.S. can prescribe those drugs once they’re approved. So the authorities made a regulatory decision about who the people are who are educated or not educated in the diagnosis and management of patients with SpA,” according to Dr. Michael H. Weisman, chair and director of rheumatology at Cedars-Sinai Medical Center in Los Angeles.
Dr. Caplan of the University of Colorado, Denver, observed that developing new and improved criteria for SpA will be a multiyear endeavor. A top goal is to shed light on the natural history of nr-axSpA, about which almost nothing is known. This will probably entail developing a sizable cohort of patients with nr-axSpA, following them for 5 years or so to see who develops ankylosing spondylitis, then looking back to identify reliable predictors.
The SPARTAN vote was preceded by a formal debate on the merits of replacing the ASAS criteria. Dr. Muhammad Asim Khan, professor of medicine at Case Western Reserve University, Cleveland, who 30 years ago was a pioneer in the concept of spondylitic disease without x-ray evidence of sacroiliitis, argued that the ASAS criteria aren’t adequately doing what disease classification criteria are supposed to do: provide a simple, clear means of identifying a homogeneous population for clinical trials. By letting in substantial numbers of patients with fibromyalgia, the criteria water down the clinical outcomes of investigational therapies for SpA.
The opposing argument was provided by ASAS representative Dr. Filip Van den Bosch. He conceded the ASAS classification criteria “aren’t perfect,” but over time they have become familiar to rheumatologists worldwide.
“Modifying the criteria will lead to years of uncertainty,” argued Dr. Van den Bosch, professor of rheumatology at Ghent (Belgium) University.
If, as some have proposed, SPARTAN were to tighten the ASAS criteria by insisting that a patient lacking x-ray evidence of sacroiliitis must possess three or four SpA features instead of the two now required, the result would be a drastic reduction in the sensitivity of the criteria, he continued.
“The challenge of making a correct diagnosis of axSpA cannot be solved by modifying the ASAS criteria. I cannot think of a new SpA feature that’s been identified in the last 5 years that could be added on top of what’s already in there,” the rheumatologist said.
What’s needed, he added, is a concerted effort to better educate physicians about axSpA, including interpretation of spinal x-rays and MRIs, the disease’s signs and symptoms, and how to differentiate SpA from other disorders that cause back pain.
While nearly two-thirds of the SPARTAN membership voted in favor of developing new classification criteria, some prominent voices were raised in dissent.
“I agree with Filip,” said Dr. Weisman. “The problem is not with the criteria, it’s who’s using them.”
Dr. Robert D. Inman, professor of medicine at the University of Toronto, predicted “it’s an exercise in futility” to try to come up with a single set of classification criteria that will provide the high degree of sensitivity that patients and clinicians want so that people who present with early axSpA are not missed while at the same time providing the maximum specificity that payers and regulatory officials seek.
Dr. Sindhu Johnson, who as cochair of the ACR Classification Criteria Subcommittee has shepherded the development of new criteria for a number of rheumatologic diseases, offered the SPARTAN group some practical tips. Because European studies tend to be comprised of monoethnic cohorts, the ACR will look favorably upon a successful SPARTAN effort to incorporate African Americans, Asians, Native Americans, and other minorities in their study cohorts. And SPARTAN should be open and receptive to critical feedback from stakeholders during the criteria development process.
“Get buy in. There’s no point in developing criteria that are extremely rigorous scientifically but that the global community feels are useless, that use tests they don’t have access to. This is one of the criticisms around the Sjögren’s criteria: they require ophthalmologic testing and salivary gland biopsy, and for rheumatologists – even academic rheumatologists at recruiting centers for clinical trials – they don’t have access to this, or it takes too much time. You need to come up with something pragmatic to get buy in,” advised Dr. Johnson of the University of Toronto.
By the way, she added, if the project wins ACR and EULAR funding it can only be named an ACR/EULAR product. “No other organization can be named in the headline,” according to Dr. Johnson.
AT THE 2015 SPARTAN ANNUAL MEETING
Biologic treatment for psoriasis found to have CVD benefits
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
EXPERT ANALYSIS AT PDA 2015
Effective primary care screen identified for axial spondyloarthritis
DENVER – Spondyloarthritis experts have zeroed in on a simple and effective screening and referral strategy for nonrheumatologists to use in deciding which patients with chronic low back pain to send to rheumatologists for definitive diagnosis and treatment of axial spondyloarthritis.
The goal is to reduce the unacceptably long delay to diagnosis of axial spondyloarthritis – on average, 7 years – without flooding rheumatologists with enormous numbers of patients who have mechanical low back pain, a vastly more common yet generally self-limited condition, Dr. Abhijeet Danve explained at the annual meeting of the Spondyloarthritis Research and Treatment Network (SPARTAN).
He presented a systematic review of seven studies that examined various screening and referral strategies in the diagnosis of axial spondyloarthritis (axSpA). The studies totaled more than 3,300 patients with chronic low back pain.
Two key conclusions jumped out from this structured review: One, simple referral strategies and considerably more complex approaches work equally well, resulting in 35%-45% of referred patients ultimately being diagnosed by a rheumatologist as having axSpA.
And two, one strategy emerged on balance as the clear winner: “In our opinion, referring patients with chronic low back pain for more than 3 months, age of onset less than 45 years, and who meet one of three criteria – inflammatory back pain, HLA- [human leukocyte antigen] B27-positivity, or sacroiliitis on imaging – to a rheumatologist for assessment of possible axSpA would be convenient, practical, and widely acceptable,” according to Dr. Danve of Yale University, New Haven, Conn.
The more of these three criteria a patient possessed, the greater the rate of rheumatologist-diagnosed axSpa. If, for example, a patient under age 45 years with a history of low back pain for more than 3 months had classic inflammatory back pain but was HLA-B27-negative and didn’t show sacroiliitis on imaging, the rate of a confirmed diagnosis of axSpa was 25%. The axSpa diagnosis rate rose to 45% with two positive criteria and jumped to 75% in those who met all three criteria.
With appropriate application of any of the strategies evaluated in the systematic review, rheumatologists can expect to see two to three people with chronic low back pain in order to identify one patient with axSpA, he added.
SPARTAN is gearing up for a major educational initiative aimed at primary care physicians, orthopedists, physical therapists, and chiropractors. The goal is to improve early recognition of possible axSpA and bring affected patients to rheumatologists for definitive diagnosis and specialized care. Of note, Dr. Danve’s senior coinvestigator in the referral strategy study was Dr. Atul Deodhar, SPARTAN chair and professor of medicine and medical director of rheumatology clinics at Oregon Health and Science University, Portland.
DENVER – Spondyloarthritis experts have zeroed in on a simple and effective screening and referral strategy for nonrheumatologists to use in deciding which patients with chronic low back pain to send to rheumatologists for definitive diagnosis and treatment of axial spondyloarthritis.
The goal is to reduce the unacceptably long delay to diagnosis of axial spondyloarthritis – on average, 7 years – without flooding rheumatologists with enormous numbers of patients who have mechanical low back pain, a vastly more common yet generally self-limited condition, Dr. Abhijeet Danve explained at the annual meeting of the Spondyloarthritis Research and Treatment Network (SPARTAN).
He presented a systematic review of seven studies that examined various screening and referral strategies in the diagnosis of axial spondyloarthritis (axSpA). The studies totaled more than 3,300 patients with chronic low back pain.
Two key conclusions jumped out from this structured review: One, simple referral strategies and considerably more complex approaches work equally well, resulting in 35%-45% of referred patients ultimately being diagnosed by a rheumatologist as having axSpA.
And two, one strategy emerged on balance as the clear winner: “In our opinion, referring patients with chronic low back pain for more than 3 months, age of onset less than 45 years, and who meet one of three criteria – inflammatory back pain, HLA- [human leukocyte antigen] B27-positivity, or sacroiliitis on imaging – to a rheumatologist for assessment of possible axSpA would be convenient, practical, and widely acceptable,” according to Dr. Danve of Yale University, New Haven, Conn.
The more of these three criteria a patient possessed, the greater the rate of rheumatologist-diagnosed axSpa. If, for example, a patient under age 45 years with a history of low back pain for more than 3 months had classic inflammatory back pain but was HLA-B27-negative and didn’t show sacroiliitis on imaging, the rate of a confirmed diagnosis of axSpa was 25%. The axSpa diagnosis rate rose to 45% with two positive criteria and jumped to 75% in those who met all three criteria.
With appropriate application of any of the strategies evaluated in the systematic review, rheumatologists can expect to see two to three people with chronic low back pain in order to identify one patient with axSpA, he added.
SPARTAN is gearing up for a major educational initiative aimed at primary care physicians, orthopedists, physical therapists, and chiropractors. The goal is to improve early recognition of possible axSpA and bring affected patients to rheumatologists for definitive diagnosis and specialized care. Of note, Dr. Danve’s senior coinvestigator in the referral strategy study was Dr. Atul Deodhar, SPARTAN chair and professor of medicine and medical director of rheumatology clinics at Oregon Health and Science University, Portland.
DENVER – Spondyloarthritis experts have zeroed in on a simple and effective screening and referral strategy for nonrheumatologists to use in deciding which patients with chronic low back pain to send to rheumatologists for definitive diagnosis and treatment of axial spondyloarthritis.
The goal is to reduce the unacceptably long delay to diagnosis of axial spondyloarthritis – on average, 7 years – without flooding rheumatologists with enormous numbers of patients who have mechanical low back pain, a vastly more common yet generally self-limited condition, Dr. Abhijeet Danve explained at the annual meeting of the Spondyloarthritis Research and Treatment Network (SPARTAN).
He presented a systematic review of seven studies that examined various screening and referral strategies in the diagnosis of axial spondyloarthritis (axSpA). The studies totaled more than 3,300 patients with chronic low back pain.
Two key conclusions jumped out from this structured review: One, simple referral strategies and considerably more complex approaches work equally well, resulting in 35%-45% of referred patients ultimately being diagnosed by a rheumatologist as having axSpA.
And two, one strategy emerged on balance as the clear winner: “In our opinion, referring patients with chronic low back pain for more than 3 months, age of onset less than 45 years, and who meet one of three criteria – inflammatory back pain, HLA- [human leukocyte antigen] B27-positivity, or sacroiliitis on imaging – to a rheumatologist for assessment of possible axSpA would be convenient, practical, and widely acceptable,” according to Dr. Danve of Yale University, New Haven, Conn.
The more of these three criteria a patient possessed, the greater the rate of rheumatologist-diagnosed axSpa. If, for example, a patient under age 45 years with a history of low back pain for more than 3 months had classic inflammatory back pain but was HLA-B27-negative and didn’t show sacroiliitis on imaging, the rate of a confirmed diagnosis of axSpa was 25%. The axSpa diagnosis rate rose to 45% with two positive criteria and jumped to 75% in those who met all three criteria.
With appropriate application of any of the strategies evaluated in the systematic review, rheumatologists can expect to see two to three people with chronic low back pain in order to identify one patient with axSpA, he added.
SPARTAN is gearing up for a major educational initiative aimed at primary care physicians, orthopedists, physical therapists, and chiropractors. The goal is to improve early recognition of possible axSpA and bring affected patients to rheumatologists for definitive diagnosis and specialized care. Of note, Dr. Danve’s senior coinvestigator in the referral strategy study was Dr. Atul Deodhar, SPARTAN chair and professor of medicine and medical director of rheumatology clinics at Oregon Health and Science University, Portland.
EXPERT ANALYSIS FROM THE 2015 SPARTAN ANNUAL MEETING
Nocturnal back pain suggests systemic disorder
ESTES PARK, COLO. – When a patient presents with low back pain, the single most important question to ask is this: “Are you having nocturnal pain or pain that’s unrelieved by rest?
“I really, really like this question. Most low back pain is nonspecific. Less than 5% of patients (with low back pain) have serious systemic pathology, but that’s the 5% we can’t afford to miss initially. This question gets at a key red flag for systemic disease,” Dr. Robert W. Janson explained at a conference on internal medicine sponsored by the University of Colorado.
“If you have mechanical low back pain, when you rest you feel good, just like with osteoarthritis. So if you’re writhing all night or unable to find a comfortable position, then you’re usually in that 5%. It’s inflammatory, metastatic, infectious, a fracture, or acute nerve compression,” said Dr. Janson, chief of rheumatology at the Denver VA Medical Center.
Other red flags for systemic disease include unexplained weight loss, fevers, or sweats; age over 50 years; low back pain present for longer than 6 weeks; or pain that’s unresponsive to treatment. A history of cancer, osteoporosis, corticosteroid therapy, abdominal aortic aneurysm, bowel or bladder symptoms, intravenous drug abuse, skin or urinary tract infection, morning stiffness lasting more than an hour, or sensory or motor loss are additional red flags.
Dr. Janson noted that these are essentially the same criteria the American College of Radiology uses in its 2011 recommendations on the appropriateness of obtaining a lumbar MRI.
Differentiating between mechanical low pain and the far less common inflammatory back pain, or axial spondyloarthropathy, is crucial because the prognosis and treatment are so different.
Inflammatory back pain is typically insidious in onset, relieved by exercise, frequently associated with nocturnal pain, entails more than 60 minutes of morning stiffness, and begins prior to age 40. In contrast, mechanical back pain can begin at any age, is acute in onset, exacerbated by exercise, doesn’t involve nighttime pain, and typically features less than 30 minutes of morning stiffness.
Most mechanical low back pain is self-limited and resolves within 4 weeks. It is characterized by abnormal back flexion, whereas patients with inflammatory back pain lose back mobility in all planes. They also frequently have sacroiliac joint tenderness and reduced chest expansion; patients with mechanical low back pain do not. Neurologic deficits are unusual in patients with spondyloarthropathies, since they are typically young individuals without disc disease.
Dr. Janson reported having no financial conflicts regarding his presentation.
ESTES PARK, COLO. – When a patient presents with low back pain, the single most important question to ask is this: “Are you having nocturnal pain or pain that’s unrelieved by rest?
“I really, really like this question. Most low back pain is nonspecific. Less than 5% of patients (with low back pain) have serious systemic pathology, but that’s the 5% we can’t afford to miss initially. This question gets at a key red flag for systemic disease,” Dr. Robert W. Janson explained at a conference on internal medicine sponsored by the University of Colorado.
“If you have mechanical low back pain, when you rest you feel good, just like with osteoarthritis. So if you’re writhing all night or unable to find a comfortable position, then you’re usually in that 5%. It’s inflammatory, metastatic, infectious, a fracture, or acute nerve compression,” said Dr. Janson, chief of rheumatology at the Denver VA Medical Center.
Other red flags for systemic disease include unexplained weight loss, fevers, or sweats; age over 50 years; low back pain present for longer than 6 weeks; or pain that’s unresponsive to treatment. A history of cancer, osteoporosis, corticosteroid therapy, abdominal aortic aneurysm, bowel or bladder symptoms, intravenous drug abuse, skin or urinary tract infection, morning stiffness lasting more than an hour, or sensory or motor loss are additional red flags.
Dr. Janson noted that these are essentially the same criteria the American College of Radiology uses in its 2011 recommendations on the appropriateness of obtaining a lumbar MRI.
Differentiating between mechanical low pain and the far less common inflammatory back pain, or axial spondyloarthropathy, is crucial because the prognosis and treatment are so different.
Inflammatory back pain is typically insidious in onset, relieved by exercise, frequently associated with nocturnal pain, entails more than 60 minutes of morning stiffness, and begins prior to age 40. In contrast, mechanical back pain can begin at any age, is acute in onset, exacerbated by exercise, doesn’t involve nighttime pain, and typically features less than 30 minutes of morning stiffness.
Most mechanical low back pain is self-limited and resolves within 4 weeks. It is characterized by abnormal back flexion, whereas patients with inflammatory back pain lose back mobility in all planes. They also frequently have sacroiliac joint tenderness and reduced chest expansion; patients with mechanical low back pain do not. Neurologic deficits are unusual in patients with spondyloarthropathies, since they are typically young individuals without disc disease.
Dr. Janson reported having no financial conflicts regarding his presentation.
ESTES PARK, COLO. – When a patient presents with low back pain, the single most important question to ask is this: “Are you having nocturnal pain or pain that’s unrelieved by rest?
“I really, really like this question. Most low back pain is nonspecific. Less than 5% of patients (with low back pain) have serious systemic pathology, but that’s the 5% we can’t afford to miss initially. This question gets at a key red flag for systemic disease,” Dr. Robert W. Janson explained at a conference on internal medicine sponsored by the University of Colorado.
“If you have mechanical low back pain, when you rest you feel good, just like with osteoarthritis. So if you’re writhing all night or unable to find a comfortable position, then you’re usually in that 5%. It’s inflammatory, metastatic, infectious, a fracture, or acute nerve compression,” said Dr. Janson, chief of rheumatology at the Denver VA Medical Center.
Other red flags for systemic disease include unexplained weight loss, fevers, or sweats; age over 50 years; low back pain present for longer than 6 weeks; or pain that’s unresponsive to treatment. A history of cancer, osteoporosis, corticosteroid therapy, abdominal aortic aneurysm, bowel or bladder symptoms, intravenous drug abuse, skin or urinary tract infection, morning stiffness lasting more than an hour, or sensory or motor loss are additional red flags.
Dr. Janson noted that these are essentially the same criteria the American College of Radiology uses in its 2011 recommendations on the appropriateness of obtaining a lumbar MRI.
Differentiating between mechanical low pain and the far less common inflammatory back pain, or axial spondyloarthropathy, is crucial because the prognosis and treatment are so different.
Inflammatory back pain is typically insidious in onset, relieved by exercise, frequently associated with nocturnal pain, entails more than 60 minutes of morning stiffness, and begins prior to age 40. In contrast, mechanical back pain can begin at any age, is acute in onset, exacerbated by exercise, doesn’t involve nighttime pain, and typically features less than 30 minutes of morning stiffness.
Most mechanical low back pain is self-limited and resolves within 4 weeks. It is characterized by abnormal back flexion, whereas patients with inflammatory back pain lose back mobility in all planes. They also frequently have sacroiliac joint tenderness and reduced chest expansion; patients with mechanical low back pain do not. Neurologic deficits are unusual in patients with spondyloarthropathies, since they are typically young individuals without disc disease.
Dr. Janson reported having no financial conflicts regarding his presentation.
EXPERT ANALYSIS FROM THE ANNUAL INTERNAL MEDICINE PROGRAM
Strong evidence for tuberculosis screening before psoriasis, PsA biologic therapy
Tuberculosis is the only condition in patients with psoriasis or psoriatic arthritis who are being treated with systemic biologic agents for which there is strong evidence in favor of baseline screening, according to a literature review.
Analysis of 26 studies of systemic biologic treatments and screening tests – 13 of which included patients with hepatitis C, hepatitis B, or congestive heart failure – found the highest level evidence (grade B) in favor of tuberculin skin testing or interferon-gamma release assay, with the latter being preferred for its higher sensitivity and specificity.
“Based on the [U.S. Preventive Services Task Force] grading system, it is recommended this screen be provided because there is high certainty that the net benefit is moderate, or medium certainty that the net benefit is moderate to substantial,” wrote Dr. Christine S. Ahn of Wake Forest University, Winston-Salem, N.C., and coauthors in the Journal of the American Academy of Dermatology.
Screening for hepatitis B or C infection only was supported by grade-C evidence, and there was insufficient evidence for HIV screening, with the authors suggesting that selective screening should be performed based on professional judgment, clinical context, or patient preference (J Am Acad Dermatol. 2015 July 14 doi: 10.1016/j.jaad.2015.06.004).
Similarly, there was insufficient evidence to support complete blood cell count screening, and, given the low grade evidence on monitoring hepatic function, the authors suggested this should be performed at the clinician’s discretion, particularly among patients treated with infliximab.
Tuberculosis is the only condition in patients with psoriasis or psoriatic arthritis who are being treated with systemic biologic agents for which there is strong evidence in favor of baseline screening, according to a literature review.
Analysis of 26 studies of systemic biologic treatments and screening tests – 13 of which included patients with hepatitis C, hepatitis B, or congestive heart failure – found the highest level evidence (grade B) in favor of tuberculin skin testing or interferon-gamma release assay, with the latter being preferred for its higher sensitivity and specificity.
“Based on the [U.S. Preventive Services Task Force] grading system, it is recommended this screen be provided because there is high certainty that the net benefit is moderate, or medium certainty that the net benefit is moderate to substantial,” wrote Dr. Christine S. Ahn of Wake Forest University, Winston-Salem, N.C., and coauthors in the Journal of the American Academy of Dermatology.
Screening for hepatitis B or C infection only was supported by grade-C evidence, and there was insufficient evidence for HIV screening, with the authors suggesting that selective screening should be performed based on professional judgment, clinical context, or patient preference (J Am Acad Dermatol. 2015 July 14 doi: 10.1016/j.jaad.2015.06.004).
Similarly, there was insufficient evidence to support complete blood cell count screening, and, given the low grade evidence on monitoring hepatic function, the authors suggested this should be performed at the clinician’s discretion, particularly among patients treated with infliximab.
Tuberculosis is the only condition in patients with psoriasis or psoriatic arthritis who are being treated with systemic biologic agents for which there is strong evidence in favor of baseline screening, according to a literature review.
Analysis of 26 studies of systemic biologic treatments and screening tests – 13 of which included patients with hepatitis C, hepatitis B, or congestive heart failure – found the highest level evidence (grade B) in favor of tuberculin skin testing or interferon-gamma release assay, with the latter being preferred for its higher sensitivity and specificity.
“Based on the [U.S. Preventive Services Task Force] grading system, it is recommended this screen be provided because there is high certainty that the net benefit is moderate, or medium certainty that the net benefit is moderate to substantial,” wrote Dr. Christine S. Ahn of Wake Forest University, Winston-Salem, N.C., and coauthors in the Journal of the American Academy of Dermatology.
Screening for hepatitis B or C infection only was supported by grade-C evidence, and there was insufficient evidence for HIV screening, with the authors suggesting that selective screening should be performed based on professional judgment, clinical context, or patient preference (J Am Acad Dermatol. 2015 July 14 doi: 10.1016/j.jaad.2015.06.004).
Similarly, there was insufficient evidence to support complete blood cell count screening, and, given the low grade evidence on monitoring hepatic function, the authors suggested this should be performed at the clinician’s discretion, particularly among patients treated with infliximab.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point:Tuberculosis is the only condition with strong evidence in favor of a baseline screening in patients with psoriasis or psoriatic arthritis being treated with systemic biologic agents.
Major finding: There is grade B–level evidence in favor of baseline tuberculosis screening in patients undergoing systemic biologic agent therapy, but only grade-C evidence for hepatitis B or C screening.
Data source: Analysis of 26 studies of systemic biologic treatments and screening tests.
Disclosures: One author declared consultancies, speaking engagements, grants, and stock options from a range of pharmaceutical companies. There were no other conflicts of interest declared.
Reciprocal risks found between psoriatic disease and uveitis
Researchers have uncovered a bidirectional relationship between psoriatic disease – psoriasis and psoriatic arthritis – and uveitis, with either condition significantly increasing the risk of the other.
A Danish nationwide cohort study of 74,129 individuals with psoriasis, including 6,735 with psoriatic arthritis, showed that patients with mild psoriasis had a 38% increased risk of uveitis, while those with psoriatic arthritis had a 2.5-fold increase in risk and those with psoriatic spondylitis had a greater than eight-fold increased risk, according to a paper published online in JAMA Dermatology.
There was a nonsignificant increase in the risk of uveitis in patients with severe psoriasis, but this failed to reach significance because of the small number of patients.
Similarly, patients with uveitis had a 59% greater risk of mild psoriasis, a twofold greater risk of severe psoriasis, a nearly fourfold increase in the risk of psoriatic arthritis, and an eightfold increase in the risk of psoriatic spondylitis (JAMA Dermatol. 2015 July 29 doi: 10.1001/jamadermatol.2015.1986).
The authors suggested that an increased focus on eye symptoms in patients with psoriasis and psoriatic arthritis and on skin and joint symptoms in patients with uveitis may be appropriate.
“The bidirectional relationship between psoriasis and psoriatic arthritis and uveitis suggests a shared pathogenic pathway, and increased systemic inflammation may contribute to the observed relationship,” wrote Dr. Alexander Egeberg of Pfizer and his coauthors. Dr. Egeberg was at the University of Copenhagen, Hellerup, when the study was conducted.
The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is employed by Eli Lilly. No other conflicts of interest were declared.
Researchers have uncovered a bidirectional relationship between psoriatic disease – psoriasis and psoriatic arthritis – and uveitis, with either condition significantly increasing the risk of the other.
A Danish nationwide cohort study of 74,129 individuals with psoriasis, including 6,735 with psoriatic arthritis, showed that patients with mild psoriasis had a 38% increased risk of uveitis, while those with psoriatic arthritis had a 2.5-fold increase in risk and those with psoriatic spondylitis had a greater than eight-fold increased risk, according to a paper published online in JAMA Dermatology.
There was a nonsignificant increase in the risk of uveitis in patients with severe psoriasis, but this failed to reach significance because of the small number of patients.
Similarly, patients with uveitis had a 59% greater risk of mild psoriasis, a twofold greater risk of severe psoriasis, a nearly fourfold increase in the risk of psoriatic arthritis, and an eightfold increase in the risk of psoriatic spondylitis (JAMA Dermatol. 2015 July 29 doi: 10.1001/jamadermatol.2015.1986).
The authors suggested that an increased focus on eye symptoms in patients with psoriasis and psoriatic arthritis and on skin and joint symptoms in patients with uveitis may be appropriate.
“The bidirectional relationship between psoriasis and psoriatic arthritis and uveitis suggests a shared pathogenic pathway, and increased systemic inflammation may contribute to the observed relationship,” wrote Dr. Alexander Egeberg of Pfizer and his coauthors. Dr. Egeberg was at the University of Copenhagen, Hellerup, when the study was conducted.
The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is employed by Eli Lilly. No other conflicts of interest were declared.
Researchers have uncovered a bidirectional relationship between psoriatic disease – psoriasis and psoriatic arthritis – and uveitis, with either condition significantly increasing the risk of the other.
A Danish nationwide cohort study of 74,129 individuals with psoriasis, including 6,735 with psoriatic arthritis, showed that patients with mild psoriasis had a 38% increased risk of uveitis, while those with psoriatic arthritis had a 2.5-fold increase in risk and those with psoriatic spondylitis had a greater than eight-fold increased risk, according to a paper published online in JAMA Dermatology.
There was a nonsignificant increase in the risk of uveitis in patients with severe psoriasis, but this failed to reach significance because of the small number of patients.
Similarly, patients with uveitis had a 59% greater risk of mild psoriasis, a twofold greater risk of severe psoriasis, a nearly fourfold increase in the risk of psoriatic arthritis, and an eightfold increase in the risk of psoriatic spondylitis (JAMA Dermatol. 2015 July 29 doi: 10.1001/jamadermatol.2015.1986).
The authors suggested that an increased focus on eye symptoms in patients with psoriasis and psoriatic arthritis and on skin and joint symptoms in patients with uveitis may be appropriate.
“The bidirectional relationship between psoriasis and psoriatic arthritis and uveitis suggests a shared pathogenic pathway, and increased systemic inflammation may contribute to the observed relationship,” wrote Dr. Alexander Egeberg of Pfizer and his coauthors. Dr. Egeberg was at the University of Copenhagen, Hellerup, when the study was conducted.
The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is employed by Eli Lilly. No other conflicts of interest were declared.
FROM JAMA DERMATOLOGY
Key clinical point:Patients with psoriasis or uveitis are at significantly greater risk of the other condition.
Major finding: Psoriatic arthritis more than doubles the risk of uveitis, and uveitis is associated with a two-gold greater risk of severe psoriasis.
Data source: A Danish nationwide cohort study of 74,129 individuals with psoriasis.
Disclosures: The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is an employee of Pfizer and another is employed by Eli Lilly. No other conflicts of interest were declared.
PsA disease activity and treatment success defined
New criteria that define psoriatic arthritis disease activity and treatment response establish cutpoints and percentage changes on the Disease Activity Index for Psoriatic Arthritis scores, Dr. Monika M. Schoels of the Second Department of Internal Medicine at Hietzing Hospital, Vienna, and her coauthors reported.
Dr. Schoels and her colleagues collected 30 patient profiles from an observational data set and then surveyed 44 rheumatology experts (getting 33 responses) to assess the patients’ disease activity based on swollen joint count, tender joint count, global activity, C-reactive protein, and pain ratings. Based on the distribution of Disease Activity Index for Psoriatic Arthritis (DAPSA) scores in the patient population, the investigators then calculated the 25th and 75th percentiles to determine the thresholds for different disease states.
Based on the results, the investigators suggested cutoff values of 4 or less for remission, more than 4 and up to 14 for low disease activity, more than 14 and up to 28 for moderate disease activity, and greater than 28 for high disease activity.
They also found that percentage changes of 50%, 75%, and 85% on the DAPSA corresponded to minor, moderate, and major treatment responses based on American College of Rheumatology 20, 50, and 70 responses observed in three randomized trials.
The study findings provide a “definition of treatment targets … as well as the definition of inclusion criteria for clinical trials for patients in” moderate and high disease activity, Dr. Schoels and her colleagues wrote. “The new response levels will help assess treatment in many settings.”
Read the full study in Annals of the Rheumatic Diseases.
New criteria that define psoriatic arthritis disease activity and treatment response establish cutpoints and percentage changes on the Disease Activity Index for Psoriatic Arthritis scores, Dr. Monika M. Schoels of the Second Department of Internal Medicine at Hietzing Hospital, Vienna, and her coauthors reported.
Dr. Schoels and her colleagues collected 30 patient profiles from an observational data set and then surveyed 44 rheumatology experts (getting 33 responses) to assess the patients’ disease activity based on swollen joint count, tender joint count, global activity, C-reactive protein, and pain ratings. Based on the distribution of Disease Activity Index for Psoriatic Arthritis (DAPSA) scores in the patient population, the investigators then calculated the 25th and 75th percentiles to determine the thresholds for different disease states.
Based on the results, the investigators suggested cutoff values of 4 or less for remission, more than 4 and up to 14 for low disease activity, more than 14 and up to 28 for moderate disease activity, and greater than 28 for high disease activity.
They also found that percentage changes of 50%, 75%, and 85% on the DAPSA corresponded to minor, moderate, and major treatment responses based on American College of Rheumatology 20, 50, and 70 responses observed in three randomized trials.
The study findings provide a “definition of treatment targets … as well as the definition of inclusion criteria for clinical trials for patients in” moderate and high disease activity, Dr. Schoels and her colleagues wrote. “The new response levels will help assess treatment in many settings.”
Read the full study in Annals of the Rheumatic Diseases.
New criteria that define psoriatic arthritis disease activity and treatment response establish cutpoints and percentage changes on the Disease Activity Index for Psoriatic Arthritis scores, Dr. Monika M. Schoels of the Second Department of Internal Medicine at Hietzing Hospital, Vienna, and her coauthors reported.
Dr. Schoels and her colleagues collected 30 patient profiles from an observational data set and then surveyed 44 rheumatology experts (getting 33 responses) to assess the patients’ disease activity based on swollen joint count, tender joint count, global activity, C-reactive protein, and pain ratings. Based on the distribution of Disease Activity Index for Psoriatic Arthritis (DAPSA) scores in the patient population, the investigators then calculated the 25th and 75th percentiles to determine the thresholds for different disease states.
Based on the results, the investigators suggested cutoff values of 4 or less for remission, more than 4 and up to 14 for low disease activity, more than 14 and up to 28 for moderate disease activity, and greater than 28 for high disease activity.
They also found that percentage changes of 50%, 75%, and 85% on the DAPSA corresponded to minor, moderate, and major treatment responses based on American College of Rheumatology 20, 50, and 70 responses observed in three randomized trials.
The study findings provide a “definition of treatment targets … as well as the definition of inclusion criteria for clinical trials for patients in” moderate and high disease activity, Dr. Schoels and her colleagues wrote. “The new response levels will help assess treatment in many settings.”
Read the full study in Annals of the Rheumatic Diseases.