User login
Apremilast response durable at 52 weeks
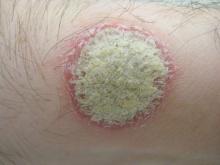
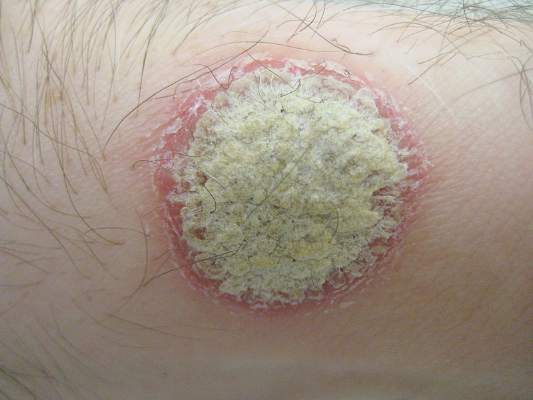
If apremilast (Otezla) works initially for psoriasis, it’s likely to keep on helping for at least a year, according to phase III results published by the drug’s maker, Celgene, in the Journal of the American Academy of Dermatology.
The trial randomized 562 patients with moderate to severe plaque psoriasis to apremilast 30 mg twice daily, and 282 to placebo (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
At week 16, 33% of apremilast patients, but only 5.3% of placebo patients, achieved a 75% or greater reduction from their baseline Psoriasis Area and Severity Index (PASI-75) scores (P less than .0001).
The placebo group was next switched to apremilast so that all the subjects were on the drug from weeks 16 to 32. By week 32, patients switched from placebo caught up with their apremilast peers on PASI-75 response rates and improved pruritus scores.
The study continued past week 32 with 154 patients who had been on apremilast since baseline and had reached PASI-75; half were rerandomized to placebo, half to the drug. At week 52, 47 (61%) of apremilast patients had maintained their PASI-75 response, versus 9 (12%) of placebo patients.
“PASI response was maintained over 52 weeks with continued apremilast treatment. In addition, apremilast demonstrated improvements in nail and scalp psoriasis, both difficult-to-treat forms of psoriasis. Most patients rerandomized to placebo who lost PASI-75 response regained it after apremilast reinitiation,” said the authors, led by Dr. Kim Papp of Probity Medical Research in Waterloo, Ont.
Apremilast, an oral phosphodiesterase 4 inhibitor, was approved by the Food and Drug Administration in 2014 for moderate to severe plaque psoriasis and psoriatic arthritis. The 16-week results are included in the drug’s label.
The label warns of weight loss and depression with apremilast. The new report doesn’t mention depression but does note a mean weight loss of 2.08 kg with the drug, and that 19% of patients lost more than 5% of their body weight. However, no one left the study because of it.
During the first 16 weeks of the trial, the most common side effects were diarrhea (7.1% placebo versus 19% apremilast); nausea (6.7% placebo versus 16% apremilast); upper respiratory tract infection (7.4% versus 10%); nasopharyngitis (8.2% versus 7.3%); and tension headache (4.3% versus 7.3%).
Side effects tended to present early with apremilast, and the incidence didn’t increase as treatment continued. To minimize side effects, the investigators titrated the drug in 10-mg increments over the first week of treatment. Discontinuations due to side effects were low.
Serious adverse events occurred in 2.8% of placebo and 2.1% of apremilast subjects. In the apremilast group, they included three cases of coronary artery disease and three cases of nephrolithiasis, plus two cases each of urinary tract infections, acute myocardial infarctions, and chronic obstructive pulmonary disease.
The investigators excluded patients with major uncontrolled comorbidities, significant infections, active or incompletely treated tuberculosis, biologic use within 12-24 months, use of active topical agents within 2 weeks, and prolonged sun or ultraviolet exposure. Most of the subjects were white, two-thirds were men, and the average age in the study was 45 years. Weak or low-potency topical corticosteroids, coal tar shampoo and salicylic acid for scalp lesions, and unmedicated moisturizers were allowed in the study.
Celgene Corporation, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial relationships with the company.
If apremilast (Otezla) works initially for psoriasis, it’s likely to keep on helping for at least a year, according to phase III results published by the drug’s maker, Celgene, in the Journal of the American Academy of Dermatology.
The trial randomized 562 patients with moderate to severe plaque psoriasis to apremilast 30 mg twice daily, and 282 to placebo (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
At week 16, 33% of apremilast patients, but only 5.3% of placebo patients, achieved a 75% or greater reduction from their baseline Psoriasis Area and Severity Index (PASI-75) scores (P less than .0001).
The placebo group was next switched to apremilast so that all the subjects were on the drug from weeks 16 to 32. By week 32, patients switched from placebo caught up with their apremilast peers on PASI-75 response rates and improved pruritus scores.
The study continued past week 32 with 154 patients who had been on apremilast since baseline and had reached PASI-75; half were rerandomized to placebo, half to the drug. At week 52, 47 (61%) of apremilast patients had maintained their PASI-75 response, versus 9 (12%) of placebo patients.
“PASI response was maintained over 52 weeks with continued apremilast treatment. In addition, apremilast demonstrated improvements in nail and scalp psoriasis, both difficult-to-treat forms of psoriasis. Most patients rerandomized to placebo who lost PASI-75 response regained it after apremilast reinitiation,” said the authors, led by Dr. Kim Papp of Probity Medical Research in Waterloo, Ont.
Apremilast, an oral phosphodiesterase 4 inhibitor, was approved by the Food and Drug Administration in 2014 for moderate to severe plaque psoriasis and psoriatic arthritis. The 16-week results are included in the drug’s label.
The label warns of weight loss and depression with apremilast. The new report doesn’t mention depression but does note a mean weight loss of 2.08 kg with the drug, and that 19% of patients lost more than 5% of their body weight. However, no one left the study because of it.
During the first 16 weeks of the trial, the most common side effects were diarrhea (7.1% placebo versus 19% apremilast); nausea (6.7% placebo versus 16% apremilast); upper respiratory tract infection (7.4% versus 10%); nasopharyngitis (8.2% versus 7.3%); and tension headache (4.3% versus 7.3%).
Side effects tended to present early with apremilast, and the incidence didn’t increase as treatment continued. To minimize side effects, the investigators titrated the drug in 10-mg increments over the first week of treatment. Discontinuations due to side effects were low.
Serious adverse events occurred in 2.8% of placebo and 2.1% of apremilast subjects. In the apremilast group, they included three cases of coronary artery disease and three cases of nephrolithiasis, plus two cases each of urinary tract infections, acute myocardial infarctions, and chronic obstructive pulmonary disease.
The investigators excluded patients with major uncontrolled comorbidities, significant infections, active or incompletely treated tuberculosis, biologic use within 12-24 months, use of active topical agents within 2 weeks, and prolonged sun or ultraviolet exposure. Most of the subjects were white, two-thirds were men, and the average age in the study was 45 years. Weak or low-potency topical corticosteroids, coal tar shampoo and salicylic acid for scalp lesions, and unmedicated moisturizers were allowed in the study.
Celgene Corporation, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial relationships with the company.
If apremilast (Otezla) works initially for psoriasis, it’s likely to keep on helping for at least a year, according to phase III results published by the drug’s maker, Celgene, in the Journal of the American Academy of Dermatology.
The trial randomized 562 patients with moderate to severe plaque psoriasis to apremilast 30 mg twice daily, and 282 to placebo (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
At week 16, 33% of apremilast patients, but only 5.3% of placebo patients, achieved a 75% or greater reduction from their baseline Psoriasis Area and Severity Index (PASI-75) scores (P less than .0001).
The placebo group was next switched to apremilast so that all the subjects were on the drug from weeks 16 to 32. By week 32, patients switched from placebo caught up with their apremilast peers on PASI-75 response rates and improved pruritus scores.
The study continued past week 32 with 154 patients who had been on apremilast since baseline and had reached PASI-75; half were rerandomized to placebo, half to the drug. At week 52, 47 (61%) of apremilast patients had maintained their PASI-75 response, versus 9 (12%) of placebo patients.
“PASI response was maintained over 52 weeks with continued apremilast treatment. In addition, apremilast demonstrated improvements in nail and scalp psoriasis, both difficult-to-treat forms of psoriasis. Most patients rerandomized to placebo who lost PASI-75 response regained it after apremilast reinitiation,” said the authors, led by Dr. Kim Papp of Probity Medical Research in Waterloo, Ont.
Apremilast, an oral phosphodiesterase 4 inhibitor, was approved by the Food and Drug Administration in 2014 for moderate to severe plaque psoriasis and psoriatic arthritis. The 16-week results are included in the drug’s label.
The label warns of weight loss and depression with apremilast. The new report doesn’t mention depression but does note a mean weight loss of 2.08 kg with the drug, and that 19% of patients lost more than 5% of their body weight. However, no one left the study because of it.
During the first 16 weeks of the trial, the most common side effects were diarrhea (7.1% placebo versus 19% apremilast); nausea (6.7% placebo versus 16% apremilast); upper respiratory tract infection (7.4% versus 10%); nasopharyngitis (8.2% versus 7.3%); and tension headache (4.3% versus 7.3%).
Side effects tended to present early with apremilast, and the incidence didn’t increase as treatment continued. To minimize side effects, the investigators titrated the drug in 10-mg increments over the first week of treatment. Discontinuations due to side effects were low.
Serious adverse events occurred in 2.8% of placebo and 2.1% of apremilast subjects. In the apremilast group, they included three cases of coronary artery disease and three cases of nephrolithiasis, plus two cases each of urinary tract infections, acute myocardial infarctions, and chronic obstructive pulmonary disease.
The investigators excluded patients with major uncontrolled comorbidities, significant infections, active or incompletely treated tuberculosis, biologic use within 12-24 months, use of active topical agents within 2 weeks, and prolonged sun or ultraviolet exposure. Most of the subjects were white, two-thirds were men, and the average age in the study was 45 years. Weak or low-potency topical corticosteroids, coal tar shampoo and salicylic acid for scalp lesions, and unmedicated moisturizers were allowed in the study.
Celgene Corporation, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial relationships with the company.
FROM JAAD
Key clinical point: Apremilast doesn’t seem to lose effect over time.
Major finding: At week 52, 61% of apremilast patients had maintained their PASI-75 response, versus 12% of patients switched to placebo at week 32.
Data source: ESTEEM 1, a phase III trial of patients with moderate to severe plaque psoriasis.
Disclosures: Celgene, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial ties with the company.
In psoriasis, low cholesterol efflux capacity may predict heart risk
Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, based on findings from a prospective cohort study.
High-density lipoprotein cholesterol efflux capacity (CEC) was inversely correlated with the noncalcified burden (NCB) of coronary atherosclerosis in baseline data from the first 101 patients enrolled in the 4-year study, Dr. Taufiq Salahuddin of the National Heart, Lung, and Blood Institute and colleagues reported online in the European Heart Journal.
The relationship between CEC and NCB “suggests that higher CEC may promote reverse cholesterol transport from earlier, more lipid-rich plaques. … Others have found this noncalcified plaque to be the culprit lesion in acute coronary syndromes. Therefore, in the context of our study, there is strong biological plausibility for observing a relationship between CEC and NCB; psoriasis is associated with both increased future cardiovascular events and impaired HDL function, and our findings suggest that this may be due to predisposition toward formation of noncalcified plaque,” the researchers wrote.
The relationship between CEC (quantified using a cell-based ex vivo assay), and NCB plaque indexes (assessed by quantitative coronary computed tomography angiography) persisted after adjustment for cardiovascular risk factors, high-density lipoprotein cholesterol levels, and apolipoprotein A1 levels.
Of note, the relationship between CEC and NCB was stronger in women than in men, with a statistically significant gender interaction, the investigators found (Eur Heart J. 2015 Jul 18. doi.org/10.1093/eurheartj/ehv339).
Cholesterol efflux capacity has been shown in prior studies to predict future cardiovascular events, and since psoriasis both increases cardiovascular risk and impairs CEC, the investigators sought to assess the cross-sectional relationship between coronary plaque burden and CEC.
Study subjects were adults over age 18 years with moderate skin disease severity (median psoriasis area severity index [PASI] score, 6.2), and a low 10-year pooled cohort equation risk score (median, 2.7%). Traditional lipid profiles were within normal limits, and the median CEC was 0.94.
In addition to the inverse correlation between CEC and NCB at baseline, significant relationships were seen between CEC and PASI score, body surface area affected by psoriasis, and HDL cholesterol level, and between noncalcified burden and CEC, psoriasis severity, body surface area affected by psoriasis, and HDL cholesterol.
“Stratified by the sample’s median CEC value, patients with low CEC had greater total burden of coronary plaque and greater NCB than did patients with high CEC (0.0131 and 0.0127 mm2 in low CEC vs. 0.0106 and 0.0103 mm2 with high CEC),” they wrote.
No differences were seen based on psoriasis treatment status or statin use, although systemic/biologic therapy was associated with a reduction in noncalcified burden.
The findings with respect to gender differences may suggest that greater CEC is more protective against noncalcified plaque in women than it is in men, the investigators added, concluding that “ongoing follow-up of these patients will inform whether aggressive treatment of psoriasis and lifestyle measures improve CEC and ultimately noncalcified burden within the coronary arteries in psoriasis.”
This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, based on findings from a prospective cohort study.
High-density lipoprotein cholesterol efflux capacity (CEC) was inversely correlated with the noncalcified burden (NCB) of coronary atherosclerosis in baseline data from the first 101 patients enrolled in the 4-year study, Dr. Taufiq Salahuddin of the National Heart, Lung, and Blood Institute and colleagues reported online in the European Heart Journal.
The relationship between CEC and NCB “suggests that higher CEC may promote reverse cholesterol transport from earlier, more lipid-rich plaques. … Others have found this noncalcified plaque to be the culprit lesion in acute coronary syndromes. Therefore, in the context of our study, there is strong biological plausibility for observing a relationship between CEC and NCB; psoriasis is associated with both increased future cardiovascular events and impaired HDL function, and our findings suggest that this may be due to predisposition toward formation of noncalcified plaque,” the researchers wrote.
The relationship between CEC (quantified using a cell-based ex vivo assay), and NCB plaque indexes (assessed by quantitative coronary computed tomography angiography) persisted after adjustment for cardiovascular risk factors, high-density lipoprotein cholesterol levels, and apolipoprotein A1 levels.
Of note, the relationship between CEC and NCB was stronger in women than in men, with a statistically significant gender interaction, the investigators found (Eur Heart J. 2015 Jul 18. doi.org/10.1093/eurheartj/ehv339).
Cholesterol efflux capacity has been shown in prior studies to predict future cardiovascular events, and since psoriasis both increases cardiovascular risk and impairs CEC, the investigators sought to assess the cross-sectional relationship between coronary plaque burden and CEC.
Study subjects were adults over age 18 years with moderate skin disease severity (median psoriasis area severity index [PASI] score, 6.2), and a low 10-year pooled cohort equation risk score (median, 2.7%). Traditional lipid profiles were within normal limits, and the median CEC was 0.94.
In addition to the inverse correlation between CEC and NCB at baseline, significant relationships were seen between CEC and PASI score, body surface area affected by psoriasis, and HDL cholesterol level, and between noncalcified burden and CEC, psoriasis severity, body surface area affected by psoriasis, and HDL cholesterol.
“Stratified by the sample’s median CEC value, patients with low CEC had greater total burden of coronary plaque and greater NCB than did patients with high CEC (0.0131 and 0.0127 mm2 in low CEC vs. 0.0106 and 0.0103 mm2 with high CEC),” they wrote.
No differences were seen based on psoriasis treatment status or statin use, although systemic/biologic therapy was associated with a reduction in noncalcified burden.
The findings with respect to gender differences may suggest that greater CEC is more protective against noncalcified plaque in women than it is in men, the investigators added, concluding that “ongoing follow-up of these patients will inform whether aggressive treatment of psoriasis and lifestyle measures improve CEC and ultimately noncalcified burden within the coronary arteries in psoriasis.”
This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, based on findings from a prospective cohort study.
High-density lipoprotein cholesterol efflux capacity (CEC) was inversely correlated with the noncalcified burden (NCB) of coronary atherosclerosis in baseline data from the first 101 patients enrolled in the 4-year study, Dr. Taufiq Salahuddin of the National Heart, Lung, and Blood Institute and colleagues reported online in the European Heart Journal.
The relationship between CEC and NCB “suggests that higher CEC may promote reverse cholesterol transport from earlier, more lipid-rich plaques. … Others have found this noncalcified plaque to be the culprit lesion in acute coronary syndromes. Therefore, in the context of our study, there is strong biological plausibility for observing a relationship between CEC and NCB; psoriasis is associated with both increased future cardiovascular events and impaired HDL function, and our findings suggest that this may be due to predisposition toward formation of noncalcified plaque,” the researchers wrote.
The relationship between CEC (quantified using a cell-based ex vivo assay), and NCB plaque indexes (assessed by quantitative coronary computed tomography angiography) persisted after adjustment for cardiovascular risk factors, high-density lipoprotein cholesterol levels, and apolipoprotein A1 levels.
Of note, the relationship between CEC and NCB was stronger in women than in men, with a statistically significant gender interaction, the investigators found (Eur Heart J. 2015 Jul 18. doi.org/10.1093/eurheartj/ehv339).
Cholesterol efflux capacity has been shown in prior studies to predict future cardiovascular events, and since psoriasis both increases cardiovascular risk and impairs CEC, the investigators sought to assess the cross-sectional relationship between coronary plaque burden and CEC.
Study subjects were adults over age 18 years with moderate skin disease severity (median psoriasis area severity index [PASI] score, 6.2), and a low 10-year pooled cohort equation risk score (median, 2.7%). Traditional lipid profiles were within normal limits, and the median CEC was 0.94.
In addition to the inverse correlation between CEC and NCB at baseline, significant relationships were seen between CEC and PASI score, body surface area affected by psoriasis, and HDL cholesterol level, and between noncalcified burden and CEC, psoriasis severity, body surface area affected by psoriasis, and HDL cholesterol.
“Stratified by the sample’s median CEC value, patients with low CEC had greater total burden of coronary plaque and greater NCB than did patients with high CEC (0.0131 and 0.0127 mm2 in low CEC vs. 0.0106 and 0.0103 mm2 with high CEC),” they wrote.
No differences were seen based on psoriasis treatment status or statin use, although systemic/biologic therapy was associated with a reduction in noncalcified burden.
The findings with respect to gender differences may suggest that greater CEC is more protective against noncalcified plaque in women than it is in men, the investigators added, concluding that “ongoing follow-up of these patients will inform whether aggressive treatment of psoriasis and lifestyle measures improve CEC and ultimately noncalcified burden within the coronary arteries in psoriasis.”
This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
FROM THE EUROPEAN HEART JOURNAL
Key clinical point: Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, according to findings from a prospective cohort study.
Major finding: Baseline data showed an inverse correlation between HDL cholesterol efflux capacity and noncalcified burden of coronary atherosclerosis (unadjusted beta-coefficient, –0.33).
Data source: The first 101 patients in a 4-year prospective cohort study.
Disclosures: This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
Uveitis prevalence, risk eyeballed in spondyloarthritis patients
DENVER – Anterior uveitis occurred in 13% of military veterans with spondyloarthritis followed longitudinally in the PULSAR registry, Dr. Maureen Dubreuil reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
One factor proved to be independently associated with the risk of developing uveitis in a multivariate logistic regression analysis: being positive for HLA-B27. This conferred an 11.3-fold increased risk of developing the serious ophthalmologic complication, according to Dr. Dubreuil of Boston University.
PULSAR (the Program to Understand Long-Term Outcomes in Spondyloarthritis) is an eight-site longitudinal registry funded by the Department of Veterans Affairs since 2007.
Thirty-five of 268 men (13.1%) with spondyloarthritis (SpA) were diagnosed with anterior uveitis a mean of 10.3 years following diagnosis of SpA. The diagnosis of uveitis was made by a participating PULSAR rheumatologist and confirmed by an ophthalmologist.
Sixty-two percent of subjects with uveitis experienced a mean of 5.7 recurrences, while the remainder had encountered a solitary episode at the time of the study. Two-thirds of the time, uveitis episodes occurred concurrently with periods of SpA activity. The uveitis occurred in an alternating unilateral pattern in nearly half of cases.
One-quarter of subjects with uveitis experienced complications, including cataracts, glaucoma, and blindness in two patients.
Uveitis is recognized as one of the most common extra-articular manifestations of radiographic axial SpA; however, the picture in patients with non-axial SpA has been far less clear. In the PULSAR analysis, the prevalence of uveitis was 24% among the 95 subjects with axial SpA. In a univariate analysis, this translated to a 2.45-fold increased likelihood of uveitis in individuals with axial SpA, compared with patients with non-axial SpA. Nonetheless, in a multivariate logistic regression analysis, axial SpA wasn’t associated with a significantly increased risk of uveitis. Neither were demographic variables, smoking status, SpA duration, or inflammatory markers, Dr. Dubreuil said.
She reported having no financial conflicts regarding the study, which was funded by the Department of Veterans Affairs.
DENVER – Anterior uveitis occurred in 13% of military veterans with spondyloarthritis followed longitudinally in the PULSAR registry, Dr. Maureen Dubreuil reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
One factor proved to be independently associated with the risk of developing uveitis in a multivariate logistic regression analysis: being positive for HLA-B27. This conferred an 11.3-fold increased risk of developing the serious ophthalmologic complication, according to Dr. Dubreuil of Boston University.
PULSAR (the Program to Understand Long-Term Outcomes in Spondyloarthritis) is an eight-site longitudinal registry funded by the Department of Veterans Affairs since 2007.
Thirty-five of 268 men (13.1%) with spondyloarthritis (SpA) were diagnosed with anterior uveitis a mean of 10.3 years following diagnosis of SpA. The diagnosis of uveitis was made by a participating PULSAR rheumatologist and confirmed by an ophthalmologist.
Sixty-two percent of subjects with uveitis experienced a mean of 5.7 recurrences, while the remainder had encountered a solitary episode at the time of the study. Two-thirds of the time, uveitis episodes occurred concurrently with periods of SpA activity. The uveitis occurred in an alternating unilateral pattern in nearly half of cases.
One-quarter of subjects with uveitis experienced complications, including cataracts, glaucoma, and blindness in two patients.
Uveitis is recognized as one of the most common extra-articular manifestations of radiographic axial SpA; however, the picture in patients with non-axial SpA has been far less clear. In the PULSAR analysis, the prevalence of uveitis was 24% among the 95 subjects with axial SpA. In a univariate analysis, this translated to a 2.45-fold increased likelihood of uveitis in individuals with axial SpA, compared with patients with non-axial SpA. Nonetheless, in a multivariate logistic regression analysis, axial SpA wasn’t associated with a significantly increased risk of uveitis. Neither were demographic variables, smoking status, SpA duration, or inflammatory markers, Dr. Dubreuil said.
She reported having no financial conflicts regarding the study, which was funded by the Department of Veterans Affairs.
DENVER – Anterior uveitis occurred in 13% of military veterans with spondyloarthritis followed longitudinally in the PULSAR registry, Dr. Maureen Dubreuil reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
One factor proved to be independently associated with the risk of developing uveitis in a multivariate logistic regression analysis: being positive for HLA-B27. This conferred an 11.3-fold increased risk of developing the serious ophthalmologic complication, according to Dr. Dubreuil of Boston University.
PULSAR (the Program to Understand Long-Term Outcomes in Spondyloarthritis) is an eight-site longitudinal registry funded by the Department of Veterans Affairs since 2007.
Thirty-five of 268 men (13.1%) with spondyloarthritis (SpA) were diagnosed with anterior uveitis a mean of 10.3 years following diagnosis of SpA. The diagnosis of uveitis was made by a participating PULSAR rheumatologist and confirmed by an ophthalmologist.
Sixty-two percent of subjects with uveitis experienced a mean of 5.7 recurrences, while the remainder had encountered a solitary episode at the time of the study. Two-thirds of the time, uveitis episodes occurred concurrently with periods of SpA activity. The uveitis occurred in an alternating unilateral pattern in nearly half of cases.
One-quarter of subjects with uveitis experienced complications, including cataracts, glaucoma, and blindness in two patients.
Uveitis is recognized as one of the most common extra-articular manifestations of radiographic axial SpA; however, the picture in patients with non-axial SpA has been far less clear. In the PULSAR analysis, the prevalence of uveitis was 24% among the 95 subjects with axial SpA. In a univariate analysis, this translated to a 2.45-fold increased likelihood of uveitis in individuals with axial SpA, compared with patients with non-axial SpA. Nonetheless, in a multivariate logistic regression analysis, axial SpA wasn’t associated with a significantly increased risk of uveitis. Neither were demographic variables, smoking status, SpA duration, or inflammatory markers, Dr. Dubreuil said.
She reported having no financial conflicts regarding the study, which was funded by the Department of Veterans Affairs.
AT THE 2015 SPARTAN ANNUAL MEETING
Key clinical point: The risk of anterior uveitis in patients with spondyloarthritis was 11.3-fold greater among those who were HLA-B27-positive.
Major finding: Thirteen percent of men with spondyloarthritis developed anterior uveitis a mean of 10.3 years following diagnosis of their rheumatologic disease.
Data source: An analysis of 268 male military veterans with spondyloarthritis in the Program to Understand Long-Term Outcomes in Spondyloarthritis (PULSAR) registry.
Disclosures: PULSAR is funded by the Department of Veterans Affairs. The presenter reported having no financial conflicts of interest.
Study yields insight into nonradiographic axial spondyloarthritis progression
DENVER – Only one-quarter of patients newly identified as having nonradiographic axial spondyloarthritis will progress to ankylosing spondylitis within 15 years, according to findings from the Rochester (Minn.) Epidemiology Project.
Disease progression occurred far more frequently and rapidly in patients who qualified for nonradiographic axial spondyloarthritis (nr-AxSpA) on the basis of positive pelvic MRI findings than in those who met only the clinical criteria for nr-AxSpA, Dr. Runsheng Wang reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
The Rochester Epidemiology Project is a unique health care research resource. Essentially all residents in Olmsted County, Minn., the home of the Mayo Clinic, have signed off on participation in the project. Dr. Weng’s study focused on 16- to 45-year-old county residents who reported new-onset chronic back pain during 1985-2010, none of whom showed the radiographic evidence of sacroiliitis required for a diagnosis of ankylosing spondylitis.
The study began with 1,142 patients who for a variety of reasons underwent a pelvic MRI. Eighteen of them ultimately met ASAS (Assessment of Spondyloarthritis International Society) imaging criteria for nr-AxSpA. They formed the imaging arm of the study.
Another 1,009 patients in the target age group presented with chronic back pain but no MRI findings. Sixty-five of them were entered into the clinical nr-AxSpA arm of the study on the basis of being positive for HLA-B27, explained Dr. Wang, a clinical scholar in rheumatology at the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Subjects were followed for a mean of 10.7 years. The overall rate of progression from nr-AxSpA to ankylosing spondylitis was 6.4% at 5 years, 17.3% at 10 years, and 26.4% at 15 years.
These data help fill an unmet need regarding understanding of the prognosis of nr-AxSpA. The two previous studies by other investigators have limitations: one German study showed an 11.2% rate of conversion to ankylosing spondylitis but was limited to 2 years of follow-up (Ann Rheum Dis. 2011 Aug;70[8]:1369-1374), while a Norwegian study comprising 20 patients showed a 20% progression rate over 8 years, Dr. Wang noted.
In her Minnesota study, patients who met criteria for nr-AxSpA based upon MRI criteria were 3.5-fold more likely to progress to ankylosing spondylitis with evidence of sacroiliitis on X-ray, compared with those in the clinical arm. There was a hint of a gender difference: Men were 1.58 times more likely to progress to ankylosing spondylitis than were women. However, this difference didn’t reach statistical significance.
Audience members were particularly interested in whether the Minnesota data demonstrated that treatment with NSAIDs and other agents had an impact upon the progression rate. Dr. Wang replied that the study size was simply too small to allow for a reasonable multivariate analysis examining this key question.
The National Institutes of Health funded the study. Dr. Wang reported having no financial conflicts of interest.
DENVER – Only one-quarter of patients newly identified as having nonradiographic axial spondyloarthritis will progress to ankylosing spondylitis within 15 years, according to findings from the Rochester (Minn.) Epidemiology Project.
Disease progression occurred far more frequently and rapidly in patients who qualified for nonradiographic axial spondyloarthritis (nr-AxSpA) on the basis of positive pelvic MRI findings than in those who met only the clinical criteria for nr-AxSpA, Dr. Runsheng Wang reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
The Rochester Epidemiology Project is a unique health care research resource. Essentially all residents in Olmsted County, Minn., the home of the Mayo Clinic, have signed off on participation in the project. Dr. Weng’s study focused on 16- to 45-year-old county residents who reported new-onset chronic back pain during 1985-2010, none of whom showed the radiographic evidence of sacroiliitis required for a diagnosis of ankylosing spondylitis.
The study began with 1,142 patients who for a variety of reasons underwent a pelvic MRI. Eighteen of them ultimately met ASAS (Assessment of Spondyloarthritis International Society) imaging criteria for nr-AxSpA. They formed the imaging arm of the study.
Another 1,009 patients in the target age group presented with chronic back pain but no MRI findings. Sixty-five of them were entered into the clinical nr-AxSpA arm of the study on the basis of being positive for HLA-B27, explained Dr. Wang, a clinical scholar in rheumatology at the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Subjects were followed for a mean of 10.7 years. The overall rate of progression from nr-AxSpA to ankylosing spondylitis was 6.4% at 5 years, 17.3% at 10 years, and 26.4% at 15 years.
These data help fill an unmet need regarding understanding of the prognosis of nr-AxSpA. The two previous studies by other investigators have limitations: one German study showed an 11.2% rate of conversion to ankylosing spondylitis but was limited to 2 years of follow-up (Ann Rheum Dis. 2011 Aug;70[8]:1369-1374), while a Norwegian study comprising 20 patients showed a 20% progression rate over 8 years, Dr. Wang noted.
In her Minnesota study, patients who met criteria for nr-AxSpA based upon MRI criteria were 3.5-fold more likely to progress to ankylosing spondylitis with evidence of sacroiliitis on X-ray, compared with those in the clinical arm. There was a hint of a gender difference: Men were 1.58 times more likely to progress to ankylosing spondylitis than were women. However, this difference didn’t reach statistical significance.
Audience members were particularly interested in whether the Minnesota data demonstrated that treatment with NSAIDs and other agents had an impact upon the progression rate. Dr. Wang replied that the study size was simply too small to allow for a reasonable multivariate analysis examining this key question.
The National Institutes of Health funded the study. Dr. Wang reported having no financial conflicts of interest.
DENVER – Only one-quarter of patients newly identified as having nonradiographic axial spondyloarthritis will progress to ankylosing spondylitis within 15 years, according to findings from the Rochester (Minn.) Epidemiology Project.
Disease progression occurred far more frequently and rapidly in patients who qualified for nonradiographic axial spondyloarthritis (nr-AxSpA) on the basis of positive pelvic MRI findings than in those who met only the clinical criteria for nr-AxSpA, Dr. Runsheng Wang reported at the annual meeting of the Spondyloarthritis Research and Treatment Network.
The Rochester Epidemiology Project is a unique health care research resource. Essentially all residents in Olmsted County, Minn., the home of the Mayo Clinic, have signed off on participation in the project. Dr. Weng’s study focused on 16- to 45-year-old county residents who reported new-onset chronic back pain during 1985-2010, none of whom showed the radiographic evidence of sacroiliitis required for a diagnosis of ankylosing spondylitis.
The study began with 1,142 patients who for a variety of reasons underwent a pelvic MRI. Eighteen of them ultimately met ASAS (Assessment of Spondyloarthritis International Society) imaging criteria for nr-AxSpA. They formed the imaging arm of the study.
Another 1,009 patients in the target age group presented with chronic back pain but no MRI findings. Sixty-five of them were entered into the clinical nr-AxSpA arm of the study on the basis of being positive for HLA-B27, explained Dr. Wang, a clinical scholar in rheumatology at the National Institute of Arthritis and Musculoskeletal and Skin Diseases.
Subjects were followed for a mean of 10.7 years. The overall rate of progression from nr-AxSpA to ankylosing spondylitis was 6.4% at 5 years, 17.3% at 10 years, and 26.4% at 15 years.
These data help fill an unmet need regarding understanding of the prognosis of nr-AxSpA. The two previous studies by other investigators have limitations: one German study showed an 11.2% rate of conversion to ankylosing spondylitis but was limited to 2 years of follow-up (Ann Rheum Dis. 2011 Aug;70[8]:1369-1374), while a Norwegian study comprising 20 patients showed a 20% progression rate over 8 years, Dr. Wang noted.
In her Minnesota study, patients who met criteria for nr-AxSpA based upon MRI criteria were 3.5-fold more likely to progress to ankylosing spondylitis with evidence of sacroiliitis on X-ray, compared with those in the clinical arm. There was a hint of a gender difference: Men were 1.58 times more likely to progress to ankylosing spondylitis than were women. However, this difference didn’t reach statistical significance.
Audience members were particularly interested in whether the Minnesota data demonstrated that treatment with NSAIDs and other agents had an impact upon the progression rate. Dr. Wang replied that the study size was simply too small to allow for a reasonable multivariate analysis examining this key question.
The National Institutes of Health funded the study. Dr. Wang reported having no financial conflicts of interest.
AT THE 2015 SPARTAN ANNUAL MEETING
Key clinical point: Radiographic evidence of sacroiliitis is s-l-o-w to develop in patients with nonradiographic axial spondyloarthritis.
Major finding: Fifteen years after patients were identified as having nonradiographic axial spondyloarthritis based upon MRI or clinical findings, 26% of them had progressed to ankylosing spondylitis as defined by sacroiliitis on x-ray.
Data source: The Rochester (Minn.) Epidemiology Project, a unique medical records database incorporating all residents of Olmsted County, Minn.
Disclosures: The National Institutes of Health funded the study. The presenter reported having no relevant financial disclosures.
GRAPPA initiatives continue to chart the way for psoriasis, psoriatic arthritis
The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting was held in Stockholm this year on July 7 and 8. This year’s meeting was packed with updates regarding numerous projects, including, but not limited to, educational initiatives, research work streams, inclusion of patients in the research process in psoriasis and psoriatic arthritis (PsA), and updating of the core set of outcome measures to be included in both psoriasis and PsA clinical trials through Outcome Measures in Rheumatology (OMERACT) and International Dermatology Outcome Measures (IDEOM) initiatives.
Among the most exciting initiatives was the review of the 2015 GRAPPA PsA Treatment Recommendations. The new treatment recommendations began 2 years ago and have included extensive literature reviews, small group discussions, and large group surveys. The final manuscript includes overarching principles and specific recommendations broken down by disease manifestations (for example, peripheral arthritis, axial disease, enthesitis, dactylitis, skin psoriasis, and nail disease). Changes from the 2009 GRAPPA Treatment Recommendations (Ann. Rheum. Dis. 2009;68:1387-94) include addition of new therapies (for example, phosphodiesterase-4 inhibitors, interleukin (IL) 12/23 inhibitors, IL-17 inhibitors) and recommendations for screening for comorbidities, as well as a table noting the influence of comorbidities on therapy selection. Each recommendation is supported as “weak” or “strong” depending on the level of evidence. Patient research partners were involved in all steps of the process. The final manuscript has been submitted for publication.
In addition to treating the disease, better defining the disease is also of critical importance. In particular, defining a “flare” of rheumatoid arthritis or PsA has been quite difficult, and there has been great interest in building an assessment tool to measure the occurrence of flare. During the meeting, Dr. Anna Moverley and Dr. Philip Helliwell, both from the NIHR Musculoskeletal Biomedical Research Unit, Leeds (England) Institute of Rheumatic & Musculoskeletal Medicine, presented their work to date on defining PsA flares. They began their work by conducting a qualitative analysis of 18 patient interviews to identify themes related to having a PsA flare. Domains (or themes) identified included physical, psychological, social withdrawal, fatigue, and loss of function. In addition, patients described flare triggers, timing of flares, preflare management, and flare management. The results of this study were recently published in a paper titled, “It’s not just the joints, it’s the whole thing: qualitative analysis of patients’ experience of flare in psoriatic arthritis” (Rheumatology 2015;54:1448-54). The team then used these themes to create a Delphi survey, which was sent to both patients and physicians. Working groups at GRAPPA were used to determine the next steps, in particular whether a physician or patient tool (or both) should be developed.
Care of the patient with PsA from a multispecialty perspective was discussed in the “combined clinics” presentation. Several academic centers across North America have developed combined dermatology-rheumatology clinics. The functioning of these clinics varies widely from monthly dual clinics in which the dermatologist and rheumatologist see the patient together for virtual combined clinics in which time is set aside for discussion of shared patients. The advantages of these dual clinics include rich educational opportunities for medical students, residents, and fellows; the ideal clinical care model for patients; enhanced patient satisfaction with their care; improved patient care and quicker transition to a disease-modifying agent when indicated; and physician satisfaction and learning opportunities through collaborative work. Institutions using these collaborative models have combined to form the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN). The group has had two meetings and has developed research initiatives that are moving forward. Another goal of the group is to assist others centers interested in creating similar collaborative clinics.
The GRAPPA PsA BioDam (Psoriatic Arthritis Biomarkers for Joint Damage) initiative was also discussed at the meeting. The goal of this initiative is to examine soluble biomarkers as predictors of structural damage in PsA. Blood samples are taken at enrollment and then patients are followed prospectively. At 24 months of follow-up, repeat radiographs are obtained to monitor the development of new erosions. In addition to plain film radiographs, three sites are now also including MRI to assess for erosions. The study team, led by Dr. Oliver FitzGerald, hopes to begin analyzing some of the results from the study in the next 1-2 years.
Dr. Ogdie-Beatty is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting was held in Stockholm this year on July 7 and 8. This year’s meeting was packed with updates regarding numerous projects, including, but not limited to, educational initiatives, research work streams, inclusion of patients in the research process in psoriasis and psoriatic arthritis (PsA), and updating of the core set of outcome measures to be included in both psoriasis and PsA clinical trials through Outcome Measures in Rheumatology (OMERACT) and International Dermatology Outcome Measures (IDEOM) initiatives.
Among the most exciting initiatives was the review of the 2015 GRAPPA PsA Treatment Recommendations. The new treatment recommendations began 2 years ago and have included extensive literature reviews, small group discussions, and large group surveys. The final manuscript includes overarching principles and specific recommendations broken down by disease manifestations (for example, peripheral arthritis, axial disease, enthesitis, dactylitis, skin psoriasis, and nail disease). Changes from the 2009 GRAPPA Treatment Recommendations (Ann. Rheum. Dis. 2009;68:1387-94) include addition of new therapies (for example, phosphodiesterase-4 inhibitors, interleukin (IL) 12/23 inhibitors, IL-17 inhibitors) and recommendations for screening for comorbidities, as well as a table noting the influence of comorbidities on therapy selection. Each recommendation is supported as “weak” or “strong” depending on the level of evidence. Patient research partners were involved in all steps of the process. The final manuscript has been submitted for publication.
In addition to treating the disease, better defining the disease is also of critical importance. In particular, defining a “flare” of rheumatoid arthritis or PsA has been quite difficult, and there has been great interest in building an assessment tool to measure the occurrence of flare. During the meeting, Dr. Anna Moverley and Dr. Philip Helliwell, both from the NIHR Musculoskeletal Biomedical Research Unit, Leeds (England) Institute of Rheumatic & Musculoskeletal Medicine, presented their work to date on defining PsA flares. They began their work by conducting a qualitative analysis of 18 patient interviews to identify themes related to having a PsA flare. Domains (or themes) identified included physical, psychological, social withdrawal, fatigue, and loss of function. In addition, patients described flare triggers, timing of flares, preflare management, and flare management. The results of this study were recently published in a paper titled, “It’s not just the joints, it’s the whole thing: qualitative analysis of patients’ experience of flare in psoriatic arthritis” (Rheumatology 2015;54:1448-54). The team then used these themes to create a Delphi survey, which was sent to both patients and physicians. Working groups at GRAPPA were used to determine the next steps, in particular whether a physician or patient tool (or both) should be developed.
Care of the patient with PsA from a multispecialty perspective was discussed in the “combined clinics” presentation. Several academic centers across North America have developed combined dermatology-rheumatology clinics. The functioning of these clinics varies widely from monthly dual clinics in which the dermatologist and rheumatologist see the patient together for virtual combined clinics in which time is set aside for discussion of shared patients. The advantages of these dual clinics include rich educational opportunities for medical students, residents, and fellows; the ideal clinical care model for patients; enhanced patient satisfaction with their care; improved patient care and quicker transition to a disease-modifying agent when indicated; and physician satisfaction and learning opportunities through collaborative work. Institutions using these collaborative models have combined to form the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN). The group has had two meetings and has developed research initiatives that are moving forward. Another goal of the group is to assist others centers interested in creating similar collaborative clinics.
The GRAPPA PsA BioDam (Psoriatic Arthritis Biomarkers for Joint Damage) initiative was also discussed at the meeting. The goal of this initiative is to examine soluble biomarkers as predictors of structural damage in PsA. Blood samples are taken at enrollment and then patients are followed prospectively. At 24 months of follow-up, repeat radiographs are obtained to monitor the development of new erosions. In addition to plain film radiographs, three sites are now also including MRI to assess for erosions. The study team, led by Dr. Oliver FitzGerald, hopes to begin analyzing some of the results from the study in the next 1-2 years.
Dr. Ogdie-Beatty is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting was held in Stockholm this year on July 7 and 8. This year’s meeting was packed with updates regarding numerous projects, including, but not limited to, educational initiatives, research work streams, inclusion of patients in the research process in psoriasis and psoriatic arthritis (PsA), and updating of the core set of outcome measures to be included in both psoriasis and PsA clinical trials through Outcome Measures in Rheumatology (OMERACT) and International Dermatology Outcome Measures (IDEOM) initiatives.
Among the most exciting initiatives was the review of the 2015 GRAPPA PsA Treatment Recommendations. The new treatment recommendations began 2 years ago and have included extensive literature reviews, small group discussions, and large group surveys. The final manuscript includes overarching principles and specific recommendations broken down by disease manifestations (for example, peripheral arthritis, axial disease, enthesitis, dactylitis, skin psoriasis, and nail disease). Changes from the 2009 GRAPPA Treatment Recommendations (Ann. Rheum. Dis. 2009;68:1387-94) include addition of new therapies (for example, phosphodiesterase-4 inhibitors, interleukin (IL) 12/23 inhibitors, IL-17 inhibitors) and recommendations for screening for comorbidities, as well as a table noting the influence of comorbidities on therapy selection. Each recommendation is supported as “weak” or “strong” depending on the level of evidence. Patient research partners were involved in all steps of the process. The final manuscript has been submitted for publication.
In addition to treating the disease, better defining the disease is also of critical importance. In particular, defining a “flare” of rheumatoid arthritis or PsA has been quite difficult, and there has been great interest in building an assessment tool to measure the occurrence of flare. During the meeting, Dr. Anna Moverley and Dr. Philip Helliwell, both from the NIHR Musculoskeletal Biomedical Research Unit, Leeds (England) Institute of Rheumatic & Musculoskeletal Medicine, presented their work to date on defining PsA flares. They began their work by conducting a qualitative analysis of 18 patient interviews to identify themes related to having a PsA flare. Domains (or themes) identified included physical, psychological, social withdrawal, fatigue, and loss of function. In addition, patients described flare triggers, timing of flares, preflare management, and flare management. The results of this study were recently published in a paper titled, “It’s not just the joints, it’s the whole thing: qualitative analysis of patients’ experience of flare in psoriatic arthritis” (Rheumatology 2015;54:1448-54). The team then used these themes to create a Delphi survey, which was sent to both patients and physicians. Working groups at GRAPPA were used to determine the next steps, in particular whether a physician or patient tool (or both) should be developed.
Care of the patient with PsA from a multispecialty perspective was discussed in the “combined clinics” presentation. Several academic centers across North America have developed combined dermatology-rheumatology clinics. The functioning of these clinics varies widely from monthly dual clinics in which the dermatologist and rheumatologist see the patient together for virtual combined clinics in which time is set aside for discussion of shared patients. The advantages of these dual clinics include rich educational opportunities for medical students, residents, and fellows; the ideal clinical care model for patients; enhanced patient satisfaction with their care; improved patient care and quicker transition to a disease-modifying agent when indicated; and physician satisfaction and learning opportunities through collaborative work. Institutions using these collaborative models have combined to form the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN). The group has had two meetings and has developed research initiatives that are moving forward. Another goal of the group is to assist others centers interested in creating similar collaborative clinics.
The GRAPPA PsA BioDam (Psoriatic Arthritis Biomarkers for Joint Damage) initiative was also discussed at the meeting. The goal of this initiative is to examine soluble biomarkers as predictors of structural damage in PsA. Blood samples are taken at enrollment and then patients are followed prospectively. At 24 months of follow-up, repeat radiographs are obtained to monitor the development of new erosions. In addition to plain film radiographs, three sites are now also including MRI to assess for erosions. The study team, led by Dr. Oliver FitzGerald, hopes to begin analyzing some of the results from the study in the next 1-2 years.
Dr. Ogdie-Beatty is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
Secukinumab proves successful against psoriatic arthritis in FUTURE 2 trial
Treatment of psoriatic arthritis over 24 weeks with the human anti-interleukin-17A monoclonal antibody secukinumab allowed a significantly higher percentage of patients who received 150-mg and 300-mg doses to achieve at least 20% improvement in the American College of Rheumatology response criteria than did placebo.
The results of the multicenter, double-blind, randomized, placebo-controlled FUTURE 2 trial hinted at the effectiveness of the 300-mg dosing regimen in treating patients who previously had not responded to treatment with tumor necrosis factor (TNF) inhibitors and did not show differences in effectiveness when used together with methotrexate. The drug did not have any particularly worrisome treatment-related adverse events, Dr. Iain McInnes of the University of Glasgow (Scotland) and his colleagues reported (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61134-5]).
At week 24, ACR20 responses occurred in 54 of 100 patients who received secukinumab 300 mg, 51 of 100 who took the 150-mg dosing regimen, 29 of 99 on the 75-mg dosing regimen, and 15 of 98 on placebo. The odds for achieving ACR20 at 24 weeks were nearly sevenfold higher for the 300-mg dose (odds ratio, 6.81; 95% confidence interval, 3.42-13.56; P < .0001) and 150-mg doses (OR, 6.52; 95% CI, 3.25-13.08; P < .0001) than with placebo.
In an editorial accompanying the report, Dr. Philip Helliwell and Dr. Laura Coates of the University of Leeds (England) said that it is important to note that responses at week 24 were similar to those observed at weeks 12 and 16 so that patients and physicians can “get an idea of response (or non-response) at an early timepoint” (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61170-9]).
Although radiographic progression wasn’t assessed in the FUTURE 2 trial, Dr. Helliwell and Dr. Coates noted that secukinumab inhibited radiographic damage in the unpublished FUTURE-1 trial, so “in view of its position in the cytokine hierarchy, interleukin-17 inhibition might prevent both of these features, and future studies with this class of drug should investigate this issue in detail.”
The lower response that seemed to have occurred in the secukinumab-treated patients who had previously used TNF inhibitors vs. those who had not “is in keeping with data for other treatments such as ustekinumab,” they wrote. The fact that there appeared to be a dose-response effect between the patients who received 150 mg secukinumab vs. 300 mg and had previously used TNF inhibitors suggests that the 300-mg dose might be more appropriate for patients who did not respond to TNF inhibitors and/or have moderate to severe psoriasis, for whom the 300-mg dose is already approved and recommended.
Read our meeting coverage of the FUTURE 2 trial at ACR 2014 here.
Treatment of psoriatic arthritis over 24 weeks with the human anti-interleukin-17A monoclonal antibody secukinumab allowed a significantly higher percentage of patients who received 150-mg and 300-mg doses to achieve at least 20% improvement in the American College of Rheumatology response criteria than did placebo.
The results of the multicenter, double-blind, randomized, placebo-controlled FUTURE 2 trial hinted at the effectiveness of the 300-mg dosing regimen in treating patients who previously had not responded to treatment with tumor necrosis factor (TNF) inhibitors and did not show differences in effectiveness when used together with methotrexate. The drug did not have any particularly worrisome treatment-related adverse events, Dr. Iain McInnes of the University of Glasgow (Scotland) and his colleagues reported (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61134-5]).
At week 24, ACR20 responses occurred in 54 of 100 patients who received secukinumab 300 mg, 51 of 100 who took the 150-mg dosing regimen, 29 of 99 on the 75-mg dosing regimen, and 15 of 98 on placebo. The odds for achieving ACR20 at 24 weeks were nearly sevenfold higher for the 300-mg dose (odds ratio, 6.81; 95% confidence interval, 3.42-13.56; P < .0001) and 150-mg doses (OR, 6.52; 95% CI, 3.25-13.08; P < .0001) than with placebo.
In an editorial accompanying the report, Dr. Philip Helliwell and Dr. Laura Coates of the University of Leeds (England) said that it is important to note that responses at week 24 were similar to those observed at weeks 12 and 16 so that patients and physicians can “get an idea of response (or non-response) at an early timepoint” (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61170-9]).
Although radiographic progression wasn’t assessed in the FUTURE 2 trial, Dr. Helliwell and Dr. Coates noted that secukinumab inhibited radiographic damage in the unpublished FUTURE-1 trial, so “in view of its position in the cytokine hierarchy, interleukin-17 inhibition might prevent both of these features, and future studies with this class of drug should investigate this issue in detail.”
The lower response that seemed to have occurred in the secukinumab-treated patients who had previously used TNF inhibitors vs. those who had not “is in keeping with data for other treatments such as ustekinumab,” they wrote. The fact that there appeared to be a dose-response effect between the patients who received 150 mg secukinumab vs. 300 mg and had previously used TNF inhibitors suggests that the 300-mg dose might be more appropriate for patients who did not respond to TNF inhibitors and/or have moderate to severe psoriasis, for whom the 300-mg dose is already approved and recommended.
Read our meeting coverage of the FUTURE 2 trial at ACR 2014 here.
Treatment of psoriatic arthritis over 24 weeks with the human anti-interleukin-17A monoclonal antibody secukinumab allowed a significantly higher percentage of patients who received 150-mg and 300-mg doses to achieve at least 20% improvement in the American College of Rheumatology response criteria than did placebo.
The results of the multicenter, double-blind, randomized, placebo-controlled FUTURE 2 trial hinted at the effectiveness of the 300-mg dosing regimen in treating patients who previously had not responded to treatment with tumor necrosis factor (TNF) inhibitors and did not show differences in effectiveness when used together with methotrexate. The drug did not have any particularly worrisome treatment-related adverse events, Dr. Iain McInnes of the University of Glasgow (Scotland) and his colleagues reported (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61134-5]).
At week 24, ACR20 responses occurred in 54 of 100 patients who received secukinumab 300 mg, 51 of 100 who took the 150-mg dosing regimen, 29 of 99 on the 75-mg dosing regimen, and 15 of 98 on placebo. The odds for achieving ACR20 at 24 weeks were nearly sevenfold higher for the 300-mg dose (odds ratio, 6.81; 95% confidence interval, 3.42-13.56; P < .0001) and 150-mg doses (OR, 6.52; 95% CI, 3.25-13.08; P < .0001) than with placebo.
In an editorial accompanying the report, Dr. Philip Helliwell and Dr. Laura Coates of the University of Leeds (England) said that it is important to note that responses at week 24 were similar to those observed at weeks 12 and 16 so that patients and physicians can “get an idea of response (or non-response) at an early timepoint” (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61170-9]).
Although radiographic progression wasn’t assessed in the FUTURE 2 trial, Dr. Helliwell and Dr. Coates noted that secukinumab inhibited radiographic damage in the unpublished FUTURE-1 trial, so “in view of its position in the cytokine hierarchy, interleukin-17 inhibition might prevent both of these features, and future studies with this class of drug should investigate this issue in detail.”
The lower response that seemed to have occurred in the secukinumab-treated patients who had previously used TNF inhibitors vs. those who had not “is in keeping with data for other treatments such as ustekinumab,” they wrote. The fact that there appeared to be a dose-response effect between the patients who received 150 mg secukinumab vs. 300 mg and had previously used TNF inhibitors suggests that the 300-mg dose might be more appropriate for patients who did not respond to TNF inhibitors and/or have moderate to severe psoriasis, for whom the 300-mg dose is already approved and recommended.
Read our meeting coverage of the FUTURE 2 trial at ACR 2014 here.
WCD: Touch avoidance is a new dimension of psoriatic impairment
VANCOUVER – Touch impairment is an important but largely unappreciated aspect of quality-of-life impairment in psoriasis, Dr. April Armstrong said at the World Congress of Dermatology.
“Touch avoidance is a new concept in dermatology, and I think it’s great because it really brings home a humanistic aspect to research,” observed Dr. Armstrong, director of the psoriasis program at the University of Colorado, Denver.
“Touch is a fundamental human gesture that conveys understanding, connection, validation, as well as being cared for or caring for someone,” she added.
She presented an online study of 1,109 psoriasis patients, 70% of whom were female. When asked whether within the past 2 weeks they had avoided hugging, shaking hands, or other touching of others, or of being touched, because of how their skin looks or feels, 48% responded yes. On a 0-10 scale, 27% of the overall patient population rated themselves as having touch avoidance at the level of 4-10, corresponding to moderate to severe.
In a multivariate analysis, the prevalence or severity of touch avoidance didn’t vary by gender. However, touch avoidance was reported most often by patients under age 25 years. And touch avoidance was strongly associated with disease severity: Those with mild psoriasis as defined by body surface area of involvement were only about one-third as likely to report touch avoidance, compared with those with severe disease. Also, psoriatic itching was an important correlate of touch avoidance: Those with itch were roughly fivefold more likely to report recent touch avoidance.
In addition, patients with psoriasis on exposed areas such as the face and neck or in sensitive areas such as the genitalia were more likely to report touch avoidance.
Touch avoidance had a strong negative impact on quality of life. Scores on the Dermatology Life Quality Index averaged nearly threefold higher – indicating worse quality of life – in participants who reported touch avoidance. They were at similarly increased risk for mild or greater depressive symptoms based upon their scores on the Quick Inventory of Depressive Symptoms.
Thus, touch avoidance is an important new patient-reported outcome, and these study results open up a new area of psoriasis research, according to Dr. Armstrong.
“Future research may reveal that therapies offering clearance of psoriasis, particularly in sensitive areas, could lead to large improvements in the initiation and acceptance of interpersonal touch by patients with psoriasis,” she noted.
This study was supported by the National Psoriasis Foundation. Dr. Armstrong reported receiving research grants from and/or serving as a consultant to more than half a dozen pharmaceutical companies as well as the National Institutes of Health.
VANCOUVER – Touch impairment is an important but largely unappreciated aspect of quality-of-life impairment in psoriasis, Dr. April Armstrong said at the World Congress of Dermatology.
“Touch avoidance is a new concept in dermatology, and I think it’s great because it really brings home a humanistic aspect to research,” observed Dr. Armstrong, director of the psoriasis program at the University of Colorado, Denver.
“Touch is a fundamental human gesture that conveys understanding, connection, validation, as well as being cared for or caring for someone,” she added.
She presented an online study of 1,109 psoriasis patients, 70% of whom were female. When asked whether within the past 2 weeks they had avoided hugging, shaking hands, or other touching of others, or of being touched, because of how their skin looks or feels, 48% responded yes. On a 0-10 scale, 27% of the overall patient population rated themselves as having touch avoidance at the level of 4-10, corresponding to moderate to severe.
In a multivariate analysis, the prevalence or severity of touch avoidance didn’t vary by gender. However, touch avoidance was reported most often by patients under age 25 years. And touch avoidance was strongly associated with disease severity: Those with mild psoriasis as defined by body surface area of involvement were only about one-third as likely to report touch avoidance, compared with those with severe disease. Also, psoriatic itching was an important correlate of touch avoidance: Those with itch were roughly fivefold more likely to report recent touch avoidance.
In addition, patients with psoriasis on exposed areas such as the face and neck or in sensitive areas such as the genitalia were more likely to report touch avoidance.
Touch avoidance had a strong negative impact on quality of life. Scores on the Dermatology Life Quality Index averaged nearly threefold higher – indicating worse quality of life – in participants who reported touch avoidance. They were at similarly increased risk for mild or greater depressive symptoms based upon their scores on the Quick Inventory of Depressive Symptoms.
Thus, touch avoidance is an important new patient-reported outcome, and these study results open up a new area of psoriasis research, according to Dr. Armstrong.
“Future research may reveal that therapies offering clearance of psoriasis, particularly in sensitive areas, could lead to large improvements in the initiation and acceptance of interpersonal touch by patients with psoriasis,” she noted.
This study was supported by the National Psoriasis Foundation. Dr. Armstrong reported receiving research grants from and/or serving as a consultant to more than half a dozen pharmaceutical companies as well as the National Institutes of Health.
VANCOUVER – Touch impairment is an important but largely unappreciated aspect of quality-of-life impairment in psoriasis, Dr. April Armstrong said at the World Congress of Dermatology.
“Touch avoidance is a new concept in dermatology, and I think it’s great because it really brings home a humanistic aspect to research,” observed Dr. Armstrong, director of the psoriasis program at the University of Colorado, Denver.
“Touch is a fundamental human gesture that conveys understanding, connection, validation, as well as being cared for or caring for someone,” she added.
She presented an online study of 1,109 psoriasis patients, 70% of whom were female. When asked whether within the past 2 weeks they had avoided hugging, shaking hands, or other touching of others, or of being touched, because of how their skin looks or feels, 48% responded yes. On a 0-10 scale, 27% of the overall patient population rated themselves as having touch avoidance at the level of 4-10, corresponding to moderate to severe.
In a multivariate analysis, the prevalence or severity of touch avoidance didn’t vary by gender. However, touch avoidance was reported most often by patients under age 25 years. And touch avoidance was strongly associated with disease severity: Those with mild psoriasis as defined by body surface area of involvement were only about one-third as likely to report touch avoidance, compared with those with severe disease. Also, psoriatic itching was an important correlate of touch avoidance: Those with itch were roughly fivefold more likely to report recent touch avoidance.
In addition, patients with psoriasis on exposed areas such as the face and neck or in sensitive areas such as the genitalia were more likely to report touch avoidance.
Touch avoidance had a strong negative impact on quality of life. Scores on the Dermatology Life Quality Index averaged nearly threefold higher – indicating worse quality of life – in participants who reported touch avoidance. They were at similarly increased risk for mild or greater depressive symptoms based upon their scores on the Quick Inventory of Depressive Symptoms.
Thus, touch avoidance is an important new patient-reported outcome, and these study results open up a new area of psoriasis research, according to Dr. Armstrong.
“Future research may reveal that therapies offering clearance of psoriasis, particularly in sensitive areas, could lead to large improvements in the initiation and acceptance of interpersonal touch by patients with psoriasis,” she noted.
This study was supported by the National Psoriasis Foundation. Dr. Armstrong reported receiving research grants from and/or serving as a consultant to more than half a dozen pharmaceutical companies as well as the National Institutes of Health.
AT WCD 2015
Key clinical point: Touch avoidance among psoriasis patients is an important new patient-reported outcome having a major negative quality-of-life impact.
Major finding: A total of 48 of a large group of psoriasis patients reported having avoided touching others, or having others touching them, within the prior 2 weeks because of how their skin looks or feels.
Data source: This was an online survey of 1,109 psoriasis patients.
Disclosures: The study was supported by the National Psoriasis Foundation. Dr. April Armstrong reported receiving research grants from and/or serving as a consultant to more than half a dozen pharmaceutical companies as well as the National Institutes of Health.
FDA will strengthen heart attack, stroke risk warnings for all NSAIDs
The Food and Drug Administration has taken new action to strengthen existing warning labels about the increased risk of heart attack or stroke with the use of prescription and over-the-counter nonaspirin nonsteroidal anti-inflammatory drugs.
In a July 9 drug safety communication, the agency did not provide the exact language that will be used on NSAID labels, but said that they “will be revised to reflect” information describing that:
• The risk of heart attack or stroke can occur as early as the first weeks of using an NSAID.
• The risk may increase with longer use and at higher doses of the NSAID.
• The drugs can increase the risk of heart attack or stroke even in patients without heart disease or risk factors for heart disease, but patients with heart disease or risk factors for it have a greater likelihood of heart attack or stroke following NSAID use.
• Treatment with NSAIDs following a first heart attack increases the risk of death in the first year after the heart attack, compared with patients who were not treated with NSAIDs after their first heart attack.
• NSAID use increases the risk of heart failure.
The new wording will also note that although newer information may suggest that the risk for heart attack or stroke is not the same for all NSAIDs, it “is not sufficient for us to determine that the risk of any particular NSAID is definitely higher or lower than that of any other particular NSAID.”
*The update to NSAID labels follows the recommendations given by panel members from a joint meeting of the FDA’s Arthritis Advisory Committee and the Drug Safety and Risk Management Advisory Committee in February 2014 in which there was a split vote (14-11) that was slightly in favor of rewording the warning labeling for NSAIDs in regard to the drug class’s current labeling, which implies that the cardiovascular thrombotic risk is not substantial with short treatment courses. At that meeting, the panelists also voted 16-9 that there were not enough data to suggest that naproxen presented a substantially lower risk of CV events than did either ibuprofen or selective NSAIDs, such as cyclooxygenase-2 inhibitors.
The FDA made its decision based on a comprehensive review of the data presented during that meeting.
*Correction, 7/16/2015: An earlier version of this story misstated the FDA panels’ recommendation for labeling changes.
The Food and Drug Administration has taken new action to strengthen existing warning labels about the increased risk of heart attack or stroke with the use of prescription and over-the-counter nonaspirin nonsteroidal anti-inflammatory drugs.
In a July 9 drug safety communication, the agency did not provide the exact language that will be used on NSAID labels, but said that they “will be revised to reflect” information describing that:
• The risk of heart attack or stroke can occur as early as the first weeks of using an NSAID.
• The risk may increase with longer use and at higher doses of the NSAID.
• The drugs can increase the risk of heart attack or stroke even in patients without heart disease or risk factors for heart disease, but patients with heart disease or risk factors for it have a greater likelihood of heart attack or stroke following NSAID use.
• Treatment with NSAIDs following a first heart attack increases the risk of death in the first year after the heart attack, compared with patients who were not treated with NSAIDs after their first heart attack.
• NSAID use increases the risk of heart failure.
The new wording will also note that although newer information may suggest that the risk for heart attack or stroke is not the same for all NSAIDs, it “is not sufficient for us to determine that the risk of any particular NSAID is definitely higher or lower than that of any other particular NSAID.”
*The update to NSAID labels follows the recommendations given by panel members from a joint meeting of the FDA’s Arthritis Advisory Committee and the Drug Safety and Risk Management Advisory Committee in February 2014 in which there was a split vote (14-11) that was slightly in favor of rewording the warning labeling for NSAIDs in regard to the drug class’s current labeling, which implies that the cardiovascular thrombotic risk is not substantial with short treatment courses. At that meeting, the panelists also voted 16-9 that there were not enough data to suggest that naproxen presented a substantially lower risk of CV events than did either ibuprofen or selective NSAIDs, such as cyclooxygenase-2 inhibitors.
The FDA made its decision based on a comprehensive review of the data presented during that meeting.
*Correction, 7/16/2015: An earlier version of this story misstated the FDA panels’ recommendation for labeling changes.
The Food and Drug Administration has taken new action to strengthen existing warning labels about the increased risk of heart attack or stroke with the use of prescription and over-the-counter nonaspirin nonsteroidal anti-inflammatory drugs.
In a July 9 drug safety communication, the agency did not provide the exact language that will be used on NSAID labels, but said that they “will be revised to reflect” information describing that:
• The risk of heart attack or stroke can occur as early as the first weeks of using an NSAID.
• The risk may increase with longer use and at higher doses of the NSAID.
• The drugs can increase the risk of heart attack or stroke even in patients without heart disease or risk factors for heart disease, but patients with heart disease or risk factors for it have a greater likelihood of heart attack or stroke following NSAID use.
• Treatment with NSAIDs following a first heart attack increases the risk of death in the first year after the heart attack, compared with patients who were not treated with NSAIDs after their first heart attack.
• NSAID use increases the risk of heart failure.
The new wording will also note that although newer information may suggest that the risk for heart attack or stroke is not the same for all NSAIDs, it “is not sufficient for us to determine that the risk of any particular NSAID is definitely higher or lower than that of any other particular NSAID.”
*The update to NSAID labels follows the recommendations given by panel members from a joint meeting of the FDA’s Arthritis Advisory Committee and the Drug Safety and Risk Management Advisory Committee in February 2014 in which there was a split vote (14-11) that was slightly in favor of rewording the warning labeling for NSAIDs in regard to the drug class’s current labeling, which implies that the cardiovascular thrombotic risk is not substantial with short treatment courses. At that meeting, the panelists also voted 16-9 that there were not enough data to suggest that naproxen presented a substantially lower risk of CV events than did either ibuprofen or selective NSAIDs, such as cyclooxygenase-2 inhibitors.
The FDA made its decision based on a comprehensive review of the data presented during that meeting.
*Correction, 7/16/2015: An earlier version of this story misstated the FDA panels’ recommendation for labeling changes.
Guselkumab prevails in phase II psoriasis trial
Guselkumab, a monoclonal antibody that specifically inhibits interleukin-23 signaling, was safe and more effective than placebo or adalimumab against plaque psoriasis in a phase II clinical trial, according to a report published online July 9 in the New England Journal of Medicine.
The 1-year randomized double-blind study, funded and administered by the maker of guselkumab, involved 293 adult patients treated at 31 sites in North America and 12 in Europe. The median duration of disease was 19 years. Study participants were randomly assigned to receive one of five subcutaneous doses of the agent (5 mg, 15 mg, 50 mg, 100 mg, or 200 mg) at two different dosing intervals (208 patients), matching placebo (42 patients), or adalimumab (43 patients) for 40 weeks and were followed for an additional 12 weeks. At 16 weeks, patients in the placebo group crossed over to receive 100 mg of guselkumab every 8 weeks, said Dr. Kenneth B. Gordon of Northwestern University, Chicago, and his associates.
The primary efficacy endpoint – the proportion of patients at week 16 with a Physician’s Global Assessment score of 0 or 1 – was significantly higher in every guselkumab dosing group (34%, 61%, 79%, 86%, and 83%, respectively) than in the placebo group (7%). In addition, the proportion of patients showing a 75%, 90%, or 100% improvement in Psoriasis Area and Severity Index score was significantly higher in each guselkumab group than in the placebo group. Scores on the Dermatology Life Quality Index also showed significantly greater improvement from baseline to week 16 in the guselkumab groups.
The findings were similar when patients taking the higher doses of guselkumab were compared with those taking adalimumab. At week 16, the proportion of patients with a PGA score of 0 or 1 was 58% with adalimumab, compared with 79%, 86%, and 83% at the highest doses of guselkumab (50 mg, 100 mg, or 200 mg). And at week 40, the proportion of patients with a PGA score of 0 or 1 was significantly higher in those taking 50 mg (71%), 100 mg (77%), or 200 mg (81%) guselkumab than in patients taking adalimumab (49%). The response to guselkumab was noted at the earliest patient assessment at week 4, peaked at week 20, and was maintained through week 40, Dr. Gordon and his associates said (N. Engl. J. Med. 2015 July 9 [doi:10.1056/NEJMoa1501646]).
The rates of adverse events, serious adverse events, and infections were similar across all the treatment groups. At week 16, infections were reported in 20% of patients taking guselkumab, 14% of those taking placebo, and 12% of those taking adalimumab. At final follow-up, infections were reported in 30% of patients taking guselkumab and 35% taking adalimumab. This study had a limited ability to assess uncommon adverse events; three patients taking guselkumab had major cardiovascular events during treatment, but “it is difficult to interpret the significance” of such events, the investigators said.
They added that larger and longer-term phase III trials are now underway to further assess the efficacy and safety of guselkumab for this indication. Moreover, their study results “validate the use of interleukin-23 as a therapeutic target by showing that therapy involving selective antagonism of this single cytokine in patients with moderate to severe plaque psoriasis is highly efficacious, as compared with adalimumab therapy,” Dr. Gordon and his associates said.
Guselkumab, a monoclonal antibody that specifically inhibits interleukin-23 signaling, was safe and more effective than placebo or adalimumab against plaque psoriasis in a phase II clinical trial, according to a report published online July 9 in the New England Journal of Medicine.
The 1-year randomized double-blind study, funded and administered by the maker of guselkumab, involved 293 adult patients treated at 31 sites in North America and 12 in Europe. The median duration of disease was 19 years. Study participants were randomly assigned to receive one of five subcutaneous doses of the agent (5 mg, 15 mg, 50 mg, 100 mg, or 200 mg) at two different dosing intervals (208 patients), matching placebo (42 patients), or adalimumab (43 patients) for 40 weeks and were followed for an additional 12 weeks. At 16 weeks, patients in the placebo group crossed over to receive 100 mg of guselkumab every 8 weeks, said Dr. Kenneth B. Gordon of Northwestern University, Chicago, and his associates.
The primary efficacy endpoint – the proportion of patients at week 16 with a Physician’s Global Assessment score of 0 or 1 – was significantly higher in every guselkumab dosing group (34%, 61%, 79%, 86%, and 83%, respectively) than in the placebo group (7%). In addition, the proportion of patients showing a 75%, 90%, or 100% improvement in Psoriasis Area and Severity Index score was significantly higher in each guselkumab group than in the placebo group. Scores on the Dermatology Life Quality Index also showed significantly greater improvement from baseline to week 16 in the guselkumab groups.
The findings were similar when patients taking the higher doses of guselkumab were compared with those taking adalimumab. At week 16, the proportion of patients with a PGA score of 0 or 1 was 58% with adalimumab, compared with 79%, 86%, and 83% at the highest doses of guselkumab (50 mg, 100 mg, or 200 mg). And at week 40, the proportion of patients with a PGA score of 0 or 1 was significantly higher in those taking 50 mg (71%), 100 mg (77%), or 200 mg (81%) guselkumab than in patients taking adalimumab (49%). The response to guselkumab was noted at the earliest patient assessment at week 4, peaked at week 20, and was maintained through week 40, Dr. Gordon and his associates said (N. Engl. J. Med. 2015 July 9 [doi:10.1056/NEJMoa1501646]).
The rates of adverse events, serious adverse events, and infections were similar across all the treatment groups. At week 16, infections were reported in 20% of patients taking guselkumab, 14% of those taking placebo, and 12% of those taking adalimumab. At final follow-up, infections were reported in 30% of patients taking guselkumab and 35% taking adalimumab. This study had a limited ability to assess uncommon adverse events; three patients taking guselkumab had major cardiovascular events during treatment, but “it is difficult to interpret the significance” of such events, the investigators said.
They added that larger and longer-term phase III trials are now underway to further assess the efficacy and safety of guselkumab for this indication. Moreover, their study results “validate the use of interleukin-23 as a therapeutic target by showing that therapy involving selective antagonism of this single cytokine in patients with moderate to severe plaque psoriasis is highly efficacious, as compared with adalimumab therapy,” Dr. Gordon and his associates said.
Guselkumab, a monoclonal antibody that specifically inhibits interleukin-23 signaling, was safe and more effective than placebo or adalimumab against plaque psoriasis in a phase II clinical trial, according to a report published online July 9 in the New England Journal of Medicine.
The 1-year randomized double-blind study, funded and administered by the maker of guselkumab, involved 293 adult patients treated at 31 sites in North America and 12 in Europe. The median duration of disease was 19 years. Study participants were randomly assigned to receive one of five subcutaneous doses of the agent (5 mg, 15 mg, 50 mg, 100 mg, or 200 mg) at two different dosing intervals (208 patients), matching placebo (42 patients), or adalimumab (43 patients) for 40 weeks and were followed for an additional 12 weeks. At 16 weeks, patients in the placebo group crossed over to receive 100 mg of guselkumab every 8 weeks, said Dr. Kenneth B. Gordon of Northwestern University, Chicago, and his associates.
The primary efficacy endpoint – the proportion of patients at week 16 with a Physician’s Global Assessment score of 0 or 1 – was significantly higher in every guselkumab dosing group (34%, 61%, 79%, 86%, and 83%, respectively) than in the placebo group (7%). In addition, the proportion of patients showing a 75%, 90%, or 100% improvement in Psoriasis Area and Severity Index score was significantly higher in each guselkumab group than in the placebo group. Scores on the Dermatology Life Quality Index also showed significantly greater improvement from baseline to week 16 in the guselkumab groups.
The findings were similar when patients taking the higher doses of guselkumab were compared with those taking adalimumab. At week 16, the proportion of patients with a PGA score of 0 or 1 was 58% with adalimumab, compared with 79%, 86%, and 83% at the highest doses of guselkumab (50 mg, 100 mg, or 200 mg). And at week 40, the proportion of patients with a PGA score of 0 or 1 was significantly higher in those taking 50 mg (71%), 100 mg (77%), or 200 mg (81%) guselkumab than in patients taking adalimumab (49%). The response to guselkumab was noted at the earliest patient assessment at week 4, peaked at week 20, and was maintained through week 40, Dr. Gordon and his associates said (N. Engl. J. Med. 2015 July 9 [doi:10.1056/NEJMoa1501646]).
The rates of adverse events, serious adverse events, and infections were similar across all the treatment groups. At week 16, infections were reported in 20% of patients taking guselkumab, 14% of those taking placebo, and 12% of those taking adalimumab. At final follow-up, infections were reported in 30% of patients taking guselkumab and 35% taking adalimumab. This study had a limited ability to assess uncommon adverse events; three patients taking guselkumab had major cardiovascular events during treatment, but “it is difficult to interpret the significance” of such events, the investigators said.
They added that larger and longer-term phase III trials are now underway to further assess the efficacy and safety of guselkumab for this indication. Moreover, their study results “validate the use of interleukin-23 as a therapeutic target by showing that therapy involving selective antagonism of this single cytokine in patients with moderate to severe plaque psoriasis is highly efficacious, as compared with adalimumab therapy,” Dr. Gordon and his associates said.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Guselkumab was safe and more effective than placebo and adalimumab against plaque psoriasis in a phase II industry-sponsored trial.
Major finding: The primary efficacy endpoint – the proportion of patients at week 16 with a Physician’s Global Assessment score of 0 or 1 – was significantly higher in every guselkumab group (34%, 61%, 79%, 86%, and 83%, respectively) than in the placebo group (7%).
Data source: A 1-year double-blind randomized phase II trial involving 293 adults at 43 sites in North America and Europe.
Disclosures: The trial was sponsored by Janssen, maker of guselkumab. Janssen supplied the study drugs, collected and analyzed the data, and helped write the report. Dr. Gordon reported receiving grants and personal fees from Janssen, AbbVie, Amgen, Celgene, Eli Lilly, Novartis, Pfizer, and Medac; his associates reported ties to numerous industry sources.
EULAR’s psoriatic arthritis recommendations gain new drugs
ROME – Tumor necrosis factor inhibitors remain the mainstay of treatment for patients with psoriatic arthritis who don’t fully respond to treatment with a nonsteroidal anti-inflammatory drug and at least one conventional synthetic disease-modifying antirheumatic drug in newly updated treatment recommendations written by a task force of the European League Against Rheumatism.
The revised recommendations for psoriatic arthritis (PsA) replace what EULAR had last released in 2011 and published in 2012 (Ann. Rheum. Dis. 2012;71:4-12) and also add three new drug options not mentioned in the 2011 version that the panel now endorsed as alternatives to a tumor necrosis factor (TNF) inhibitor when a TNF inhibitor is not appropriate. The three additions are ustekinumab (Stelara), which acts by inhibiting interleukin- (IL-)12 and IL-23, secukinumab (Cosentyx), which acts by inhibiting IL-17, and apremilast (Otezla), which inhibits phosphodiesterase-4. Ustekinumab and secukinumab are considered biological disease-modifying antirheumatic drugs (bDMARDs), like the TNF inhibitors, while apremilast occupies a new classification niche as a targeted synthetic DMARD, said Dr. Laure Gossec, convener of the PsA treatment task force, at the European Congress of Rheumatology.
Although ustekinumab, secukinumab, and apremilast all have a role to play in managing selected PsA patients, they all remain second line to TNF inhibitors, stressed Dr. Gossec, professor of rheumatology at Pierre and Marie Curie University in Paris. A TNF inhibitor is the first agent to turn to when treatment with an NSAID and a conventional synthetic DMARD fails to bring about remission or minimal disease activity essentially because of its much longer track record of safety and efficacy, she said. Specifically, TNF inhibitors have been studied long term and have demonstrated their safety in registries, they have not been surpassed for efficacy by any other agent using indirect comparisons, and now that biosimilar TNF inhibitors have entered the market they also could potentially offer a price advantage, compared with newer agents.
But for some patients, TNF inhibitors are not appropriate, such as a patient with certain comorbidities or a history of infections that would make a TNF inhibitor contraindicated, she said. For these patients, one of the other types of biologic DMARDS now available would be the top alternative. In other patients, a comorbidity profile or a history of infections might make any biologic DMARD contraindicated, in which case the targeted synthetic DMARD apremilast is an appropriate alternative, she said.
The revised recommendations contain several other new items:
• For patients with active enthesitis, dactylitis, or both and an insufficient response to an NSAID or local glucocorticoid injections, then a TNF inhibitor or alternatively an IL-12 and IL-23 inhibitor (currently ustekinumab) or an IL-17 inhibitor (currently secukinumab) may be considered. Conventional synthetic DMARDS are ineffective for these patients, while in contrast all of the biologic DMARDs are effective, but the TNF inhibitors have an edge because of greater experience with the class in this setting.
• For patients with predominantly axial disease that is active and insufficiently responsive to NSAID treatment, a biologic DMARD should be considered, specifically a TNF inhibitor. Conventional synthetic DMARDs also have no efficacy for these patients, and few data exist regarding the efficacy of IL-12 and IL-23 inhibitors or IL-17 inhibitors.
• When patients fail to respond adequately to one biologic DMARD, switching to another biologic DMARD should be considered. This could involve switching from one TNF inhibitor to another, as some evidence exists that this could be effective. Alternatively, it could involve switching to a IL-12 and IL-23 inhibitor, an IL-17 inhibitor, or possibly a targeted synthetic DMARD (currently apremilast). The task force did not endorse any particular order of these drugs when switching occurs.
Dr. Gossec has been a consultant to AbbVie, Bristol-Myers Squibb, Celgene, Chugai, Janssen, Novartis, Pfizer, Roche, and UCB.
On Twitter @mitchelzoler
ROME – Tumor necrosis factor inhibitors remain the mainstay of treatment for patients with psoriatic arthritis who don’t fully respond to treatment with a nonsteroidal anti-inflammatory drug and at least one conventional synthetic disease-modifying antirheumatic drug in newly updated treatment recommendations written by a task force of the European League Against Rheumatism.
The revised recommendations for psoriatic arthritis (PsA) replace what EULAR had last released in 2011 and published in 2012 (Ann. Rheum. Dis. 2012;71:4-12) and also add three new drug options not mentioned in the 2011 version that the panel now endorsed as alternatives to a tumor necrosis factor (TNF) inhibitor when a TNF inhibitor is not appropriate. The three additions are ustekinumab (Stelara), which acts by inhibiting interleukin- (IL-)12 and IL-23, secukinumab (Cosentyx), which acts by inhibiting IL-17, and apremilast (Otezla), which inhibits phosphodiesterase-4. Ustekinumab and secukinumab are considered biological disease-modifying antirheumatic drugs (bDMARDs), like the TNF inhibitors, while apremilast occupies a new classification niche as a targeted synthetic DMARD, said Dr. Laure Gossec, convener of the PsA treatment task force, at the European Congress of Rheumatology.
Although ustekinumab, secukinumab, and apremilast all have a role to play in managing selected PsA patients, they all remain second line to TNF inhibitors, stressed Dr. Gossec, professor of rheumatology at Pierre and Marie Curie University in Paris. A TNF inhibitor is the first agent to turn to when treatment with an NSAID and a conventional synthetic DMARD fails to bring about remission or minimal disease activity essentially because of its much longer track record of safety and efficacy, she said. Specifically, TNF inhibitors have been studied long term and have demonstrated their safety in registries, they have not been surpassed for efficacy by any other agent using indirect comparisons, and now that biosimilar TNF inhibitors have entered the market they also could potentially offer a price advantage, compared with newer agents.
But for some patients, TNF inhibitors are not appropriate, such as a patient with certain comorbidities or a history of infections that would make a TNF inhibitor contraindicated, she said. For these patients, one of the other types of biologic DMARDS now available would be the top alternative. In other patients, a comorbidity profile or a history of infections might make any biologic DMARD contraindicated, in which case the targeted synthetic DMARD apremilast is an appropriate alternative, she said.
The revised recommendations contain several other new items:
• For patients with active enthesitis, dactylitis, or both and an insufficient response to an NSAID or local glucocorticoid injections, then a TNF inhibitor or alternatively an IL-12 and IL-23 inhibitor (currently ustekinumab) or an IL-17 inhibitor (currently secukinumab) may be considered. Conventional synthetic DMARDS are ineffective for these patients, while in contrast all of the biologic DMARDs are effective, but the TNF inhibitors have an edge because of greater experience with the class in this setting.
• For patients with predominantly axial disease that is active and insufficiently responsive to NSAID treatment, a biologic DMARD should be considered, specifically a TNF inhibitor. Conventional synthetic DMARDs also have no efficacy for these patients, and few data exist regarding the efficacy of IL-12 and IL-23 inhibitors or IL-17 inhibitors.
• When patients fail to respond adequately to one biologic DMARD, switching to another biologic DMARD should be considered. This could involve switching from one TNF inhibitor to another, as some evidence exists that this could be effective. Alternatively, it could involve switching to a IL-12 and IL-23 inhibitor, an IL-17 inhibitor, or possibly a targeted synthetic DMARD (currently apremilast). The task force did not endorse any particular order of these drugs when switching occurs.
Dr. Gossec has been a consultant to AbbVie, Bristol-Myers Squibb, Celgene, Chugai, Janssen, Novartis, Pfizer, Roche, and UCB.
On Twitter @mitchelzoler
ROME – Tumor necrosis factor inhibitors remain the mainstay of treatment for patients with psoriatic arthritis who don’t fully respond to treatment with a nonsteroidal anti-inflammatory drug and at least one conventional synthetic disease-modifying antirheumatic drug in newly updated treatment recommendations written by a task force of the European League Against Rheumatism.
The revised recommendations for psoriatic arthritis (PsA) replace what EULAR had last released in 2011 and published in 2012 (Ann. Rheum. Dis. 2012;71:4-12) and also add three new drug options not mentioned in the 2011 version that the panel now endorsed as alternatives to a tumor necrosis factor (TNF) inhibitor when a TNF inhibitor is not appropriate. The three additions are ustekinumab (Stelara), which acts by inhibiting interleukin- (IL-)12 and IL-23, secukinumab (Cosentyx), which acts by inhibiting IL-17, and apremilast (Otezla), which inhibits phosphodiesterase-4. Ustekinumab and secukinumab are considered biological disease-modifying antirheumatic drugs (bDMARDs), like the TNF inhibitors, while apremilast occupies a new classification niche as a targeted synthetic DMARD, said Dr. Laure Gossec, convener of the PsA treatment task force, at the European Congress of Rheumatology.
Although ustekinumab, secukinumab, and apremilast all have a role to play in managing selected PsA patients, they all remain second line to TNF inhibitors, stressed Dr. Gossec, professor of rheumatology at Pierre and Marie Curie University in Paris. A TNF inhibitor is the first agent to turn to when treatment with an NSAID and a conventional synthetic DMARD fails to bring about remission or minimal disease activity essentially because of its much longer track record of safety and efficacy, she said. Specifically, TNF inhibitors have been studied long term and have demonstrated their safety in registries, they have not been surpassed for efficacy by any other agent using indirect comparisons, and now that biosimilar TNF inhibitors have entered the market they also could potentially offer a price advantage, compared with newer agents.
But for some patients, TNF inhibitors are not appropriate, such as a patient with certain comorbidities or a history of infections that would make a TNF inhibitor contraindicated, she said. For these patients, one of the other types of biologic DMARDS now available would be the top alternative. In other patients, a comorbidity profile or a history of infections might make any biologic DMARD contraindicated, in which case the targeted synthetic DMARD apremilast is an appropriate alternative, she said.
The revised recommendations contain several other new items:
• For patients with active enthesitis, dactylitis, or both and an insufficient response to an NSAID or local glucocorticoid injections, then a TNF inhibitor or alternatively an IL-12 and IL-23 inhibitor (currently ustekinumab) or an IL-17 inhibitor (currently secukinumab) may be considered. Conventional synthetic DMARDS are ineffective for these patients, while in contrast all of the biologic DMARDs are effective, but the TNF inhibitors have an edge because of greater experience with the class in this setting.
• For patients with predominantly axial disease that is active and insufficiently responsive to NSAID treatment, a biologic DMARD should be considered, specifically a TNF inhibitor. Conventional synthetic DMARDs also have no efficacy for these patients, and few data exist regarding the efficacy of IL-12 and IL-23 inhibitors or IL-17 inhibitors.
• When patients fail to respond adequately to one biologic DMARD, switching to another biologic DMARD should be considered. This could involve switching from one TNF inhibitor to another, as some evidence exists that this could be effective. Alternatively, it could involve switching to a IL-12 and IL-23 inhibitor, an IL-17 inhibitor, or possibly a targeted synthetic DMARD (currently apremilast). The task force did not endorse any particular order of these drugs when switching occurs.
Dr. Gossec has been a consultant to AbbVie, Bristol-Myers Squibb, Celgene, Chugai, Janssen, Novartis, Pfizer, Roche, and UCB.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE EULAR 2015 CONGRESS