User login
Conspiracy theory or delusion? 3 questions to tell them apart
Many psychiatrists conceptualize mental illnesses, including psychotic disorders, across a continuum where their borders can be ambiguous.1 The same can be said of individual symptoms such as delusions, where the line separating clear-cut pathology from nonpathological or subclinical “delusion-like beliefs” is often blurred.2,3 However, the categorical distinction between mental illness and normality is fundamental to diagnostic reliability and crucial to clinical decisions about whether and how to intervene.
Conspiracy theory beliefs are delusion-like beliefs that are commonly encountered within today’s political landscape. Surveys have consistently revealed that approximately one-half of the population believes in at least 1 conspiracy theory, highlighting the normality of such beliefs despite their potential outlandishness.4 Here are 3 questions you can ask to help differentiate conspiracy theory beliefs from delusions.
1. What is the evidence for the belief?
Drawing from Karl Jaspers’ conceptualization of delusions as “impossible” and “unshareable,” the DSM-5 distinguishes delusions from culturally-sanctioned shared beliefs such as religious creeds.3 Whereas delusions often arise out of anomalous subjective experiences, individuals who come to believe in conspiracytheories have typically sought explanations and found them from secondary sources, often on the internet.5 Despite the familiar term “conspiracy theorist,” most who believe in conspiracy theories aren’t so much theorizing as they are adopting counter-narratives based on assimilated information. Unlike delusions, conspiracy theory beliefs are learned, with the “evidence” to support them easily located online.
2. Is the belief self-referential?
The stereotypical unshareability of delusions often hinges upon their self-referential content. For example, while it is easy to find others who believe in the Second Coming, it would be much harder to convince others that you are the Second Coming. Unlike delusions, conspiracy theories are beliefs about the world and explanations of real-life events; their content is rarely, if ever, directly related to the believer.
Conspiracy theory beliefs involve a negation of authoritative accounts that is rooted in “epistemic mistrust” of authoritative sources of information.5 While conspiratorial mistrust has been compared with paranoia, with paranoia found to be associated with belief in conspiracy theories,6 epistemic mistrust encompasses a range of justified cultural mistrust, unwarranted mistrust based on racial prejudice, and subclinical paranoia typical of schizotypy. The more self-referential the underlying paranoia, the more likely an associated belief is to cross the boundary from conspiracy theory to delusion.7
3. Is there overlap?
Conspiracy theory beliefs and delusions are not mutually exclusive. “Gang stalking” offers a vexing example of paranoia that is part shared conspiracy theory, part idiosyncratic delusion.8 Reliably disentangling these components requires identifying the conspiracy theory component as a widely-shared belief about government surveillance, while carefully analyzing the self-referential component to determine credibility and potential delusionality.
1. Pierre JM. The borders of mental disorder in psychiatry and the DSM: past, present, and future. J Psychiatric Practice. 2010;16(6):375-386.
2. Pierre JM. Faith or delusion? At the crossroads of religion and psychosis. J Psychiatr Practice. 2001;7(3):163-172.
3. Pierre JM. Forensic psychiatry versus the varieties of delusion-like belief. J Am Acad Psychiatry Law. 2020;48(3):327-334.
4. Oliver JE, Wood, TJ. Conspiracy theories and the paranoid style(s) of mass opinion. Am J Pol Sci. 2014;58(5);952-966.
5. Pierre JM. Mistrust and misinformation: a two-component, socio-epistemic model of belief in conspiracy theories. J Soc Polit Psychol. 2020;8(2):617-641.
6. Dagnall N, Drinkwater K, Parker A, et al. Conspiracy theory and cognitive style: a worldview. Front Psychol. 2015;6:206.
7. Imhoff R, Lamberty P. How paranoid are conspiracy believers? Toward a more fine-grained understanding of the connect and disconnect between paranoia and belief in conspiracy theories. Eur J Soc Psychol. 2018;48(7):909-926.
8. Sheridan LP, James DV. Complaints of group-stalking (‘gang-stalking’): an exploratory study of their natures and impact on complainants. J Forens Psychiatry Psychol. 2015;26(5):601-623.
Many psychiatrists conceptualize mental illnesses, including psychotic disorders, across a continuum where their borders can be ambiguous.1 The same can be said of individual symptoms such as delusions, where the line separating clear-cut pathology from nonpathological or subclinical “delusion-like beliefs” is often blurred.2,3 However, the categorical distinction between mental illness and normality is fundamental to diagnostic reliability and crucial to clinical decisions about whether and how to intervene.
Conspiracy theory beliefs are delusion-like beliefs that are commonly encountered within today’s political landscape. Surveys have consistently revealed that approximately one-half of the population believes in at least 1 conspiracy theory, highlighting the normality of such beliefs despite their potential outlandishness.4 Here are 3 questions you can ask to help differentiate conspiracy theory beliefs from delusions.
1. What is the evidence for the belief?
Drawing from Karl Jaspers’ conceptualization of delusions as “impossible” and “unshareable,” the DSM-5 distinguishes delusions from culturally-sanctioned shared beliefs such as religious creeds.3 Whereas delusions often arise out of anomalous subjective experiences, individuals who come to believe in conspiracytheories have typically sought explanations and found them from secondary sources, often on the internet.5 Despite the familiar term “conspiracy theorist,” most who believe in conspiracy theories aren’t so much theorizing as they are adopting counter-narratives based on assimilated information. Unlike delusions, conspiracy theory beliefs are learned, with the “evidence” to support them easily located online.
2. Is the belief self-referential?
The stereotypical unshareability of delusions often hinges upon their self-referential content. For example, while it is easy to find others who believe in the Second Coming, it would be much harder to convince others that you are the Second Coming. Unlike delusions, conspiracy theories are beliefs about the world and explanations of real-life events; their content is rarely, if ever, directly related to the believer.
Conspiracy theory beliefs involve a negation of authoritative accounts that is rooted in “epistemic mistrust” of authoritative sources of information.5 While conspiratorial mistrust has been compared with paranoia, with paranoia found to be associated with belief in conspiracy theories,6 epistemic mistrust encompasses a range of justified cultural mistrust, unwarranted mistrust based on racial prejudice, and subclinical paranoia typical of schizotypy. The more self-referential the underlying paranoia, the more likely an associated belief is to cross the boundary from conspiracy theory to delusion.7
3. Is there overlap?
Conspiracy theory beliefs and delusions are not mutually exclusive. “Gang stalking” offers a vexing example of paranoia that is part shared conspiracy theory, part idiosyncratic delusion.8 Reliably disentangling these components requires identifying the conspiracy theory component as a widely-shared belief about government surveillance, while carefully analyzing the self-referential component to determine credibility and potential delusionality.
Many psychiatrists conceptualize mental illnesses, including psychotic disorders, across a continuum where their borders can be ambiguous.1 The same can be said of individual symptoms such as delusions, where the line separating clear-cut pathology from nonpathological or subclinical “delusion-like beliefs” is often blurred.2,3 However, the categorical distinction between mental illness and normality is fundamental to diagnostic reliability and crucial to clinical decisions about whether and how to intervene.
Conspiracy theory beliefs are delusion-like beliefs that are commonly encountered within today’s political landscape. Surveys have consistently revealed that approximately one-half of the population believes in at least 1 conspiracy theory, highlighting the normality of such beliefs despite their potential outlandishness.4 Here are 3 questions you can ask to help differentiate conspiracy theory beliefs from delusions.
1. What is the evidence for the belief?
Drawing from Karl Jaspers’ conceptualization of delusions as “impossible” and “unshareable,” the DSM-5 distinguishes delusions from culturally-sanctioned shared beliefs such as religious creeds.3 Whereas delusions often arise out of anomalous subjective experiences, individuals who come to believe in conspiracytheories have typically sought explanations and found them from secondary sources, often on the internet.5 Despite the familiar term “conspiracy theorist,” most who believe in conspiracy theories aren’t so much theorizing as they are adopting counter-narratives based on assimilated information. Unlike delusions, conspiracy theory beliefs are learned, with the “evidence” to support them easily located online.
2. Is the belief self-referential?
The stereotypical unshareability of delusions often hinges upon their self-referential content. For example, while it is easy to find others who believe in the Second Coming, it would be much harder to convince others that you are the Second Coming. Unlike delusions, conspiracy theories are beliefs about the world and explanations of real-life events; their content is rarely, if ever, directly related to the believer.
Conspiracy theory beliefs involve a negation of authoritative accounts that is rooted in “epistemic mistrust” of authoritative sources of information.5 While conspiratorial mistrust has been compared with paranoia, with paranoia found to be associated with belief in conspiracy theories,6 epistemic mistrust encompasses a range of justified cultural mistrust, unwarranted mistrust based on racial prejudice, and subclinical paranoia typical of schizotypy. The more self-referential the underlying paranoia, the more likely an associated belief is to cross the boundary from conspiracy theory to delusion.7
3. Is there overlap?
Conspiracy theory beliefs and delusions are not mutually exclusive. “Gang stalking” offers a vexing example of paranoia that is part shared conspiracy theory, part idiosyncratic delusion.8 Reliably disentangling these components requires identifying the conspiracy theory component as a widely-shared belief about government surveillance, while carefully analyzing the self-referential component to determine credibility and potential delusionality.
1. Pierre JM. The borders of mental disorder in psychiatry and the DSM: past, present, and future. J Psychiatric Practice. 2010;16(6):375-386.
2. Pierre JM. Faith or delusion? At the crossroads of religion and psychosis. J Psychiatr Practice. 2001;7(3):163-172.
3. Pierre JM. Forensic psychiatry versus the varieties of delusion-like belief. J Am Acad Psychiatry Law. 2020;48(3):327-334.
4. Oliver JE, Wood, TJ. Conspiracy theories and the paranoid style(s) of mass opinion. Am J Pol Sci. 2014;58(5);952-966.
5. Pierre JM. Mistrust and misinformation: a two-component, socio-epistemic model of belief in conspiracy theories. J Soc Polit Psychol. 2020;8(2):617-641.
6. Dagnall N, Drinkwater K, Parker A, et al. Conspiracy theory and cognitive style: a worldview. Front Psychol. 2015;6:206.
7. Imhoff R, Lamberty P. How paranoid are conspiracy believers? Toward a more fine-grained understanding of the connect and disconnect between paranoia and belief in conspiracy theories. Eur J Soc Psychol. 2018;48(7):909-926.
8. Sheridan LP, James DV. Complaints of group-stalking (‘gang-stalking’): an exploratory study of their natures and impact on complainants. J Forens Psychiatry Psychol. 2015;26(5):601-623.
1. Pierre JM. The borders of mental disorder in psychiatry and the DSM: past, present, and future. J Psychiatric Practice. 2010;16(6):375-386.
2. Pierre JM. Faith or delusion? At the crossroads of religion and psychosis. J Psychiatr Practice. 2001;7(3):163-172.
3. Pierre JM. Forensic psychiatry versus the varieties of delusion-like belief. J Am Acad Psychiatry Law. 2020;48(3):327-334.
4. Oliver JE, Wood, TJ. Conspiracy theories and the paranoid style(s) of mass opinion. Am J Pol Sci. 2014;58(5);952-966.
5. Pierre JM. Mistrust and misinformation: a two-component, socio-epistemic model of belief in conspiracy theories. J Soc Polit Psychol. 2020;8(2):617-641.
6. Dagnall N, Drinkwater K, Parker A, et al. Conspiracy theory and cognitive style: a worldview. Front Psychol. 2015;6:206.
7. Imhoff R, Lamberty P. How paranoid are conspiracy believers? Toward a more fine-grained understanding of the connect and disconnect between paranoia and belief in conspiracy theories. Eur J Soc Psychol. 2018;48(7):909-926.
8. Sheridan LP, James DV. Complaints of group-stalking (‘gang-stalking’): an exploratory study of their natures and impact on complainants. J Forens Psychiatry Psychol. 2015;26(5):601-623.
An unquenchable thirst
CASE Unresponsive after a presumed seizure
Mr. F, age 44, has schizophrenia. He is brought to the hospital by ambulance after he is found on the ground outside of his mother’s house following a presumed seizure and fall. On arrival to the emergency department, he is unresponsive. His laboratory values are significant for a sodium level of 110 mEq/L (reference range: 135 to 145 mEq/L), indicating hyponatremia.
HISTORY Fixated on purity
Mr. F’s mother reports that Mr. F had an unremarkable childhood. He was raised in a household with both parents and a younger sister. Mr. F did well academically and studied engineering and physics in college. There was no reported history of trauma or substance use.
During his senior year of college, Mr. F began experiencing paranoia, auditory hallucinations, and religious delusions. He required hospitalization and was diagnosed with schizophrenia. Following multiple hospitalizations over 5 years, he moved in with his mother, who was granted guardianship.
His mother said Mr. F’s religious delusions were of purity and cleansing the soul. He spent hours memorizing the Bible and would go for days without eating but would drink large amounts of water. She said she thought this was due to his desire to flush out imperfections.
In the past 3 years, Mr. F has been hospitalized several times for severe hyponatremia. At home, his mother attempted to restrict his water intake. However, Mr. F would still drink out of sinks and hoses. Mr. F’s mother reports that over the past month he had become more isolated. He would spend entire days reading the Bible, and his water intake had further increased.
Prior medication trials for Mr. F included haloperidol, up to 10 mg twice per day; aripiprazole, up to 20 mg/d; and risperidone, up to 6 mg nightly. These had been effective, but Mr. F had difficulty with adherence. He did not receive a long-acting injectable (LAI) antipsychotic initially due to lack of access at the rural clinic where he was treated, and later due to his mother’s preference for her son to receive oral medications. Prior to his current presentation, Mr. F’s medication regimen was olanzapine, 10 mg twice a day; perphenazine, 8 mg twice a day; and long-acting propranolol, 60 mg/d. Mr. F had no other chronic medical problems.
EVALUATION Hyponatremia, but why?
Mr. F is intubated and admitted to the surgical service for stabilization due to injuries from his fall. He has fractures of his right sinus and bilateral nasal bones, which are managed nonoperatively. He is delirious, with waxing and waning attention, memory disturbances, and disorientation. His psychotropic medications are held.
Continue to: Imaging of his head...
Imaging of his head does not reveal acute abnormalities suggesting a malignant or paraneoplastic process, and there are no concerns for ongoing seizures. An infection workup is negative. His urine toxicology is negative and blood alcohol level is 0. His sodium normalizes after 3 days of IV fluids and fluid restriction. Therefore, further tests to differentiate the causes of hyponatremia, such as urine electrolytes and urine osmolality, are not pursued.
[polldaddy:10910406]
The authors’ observations
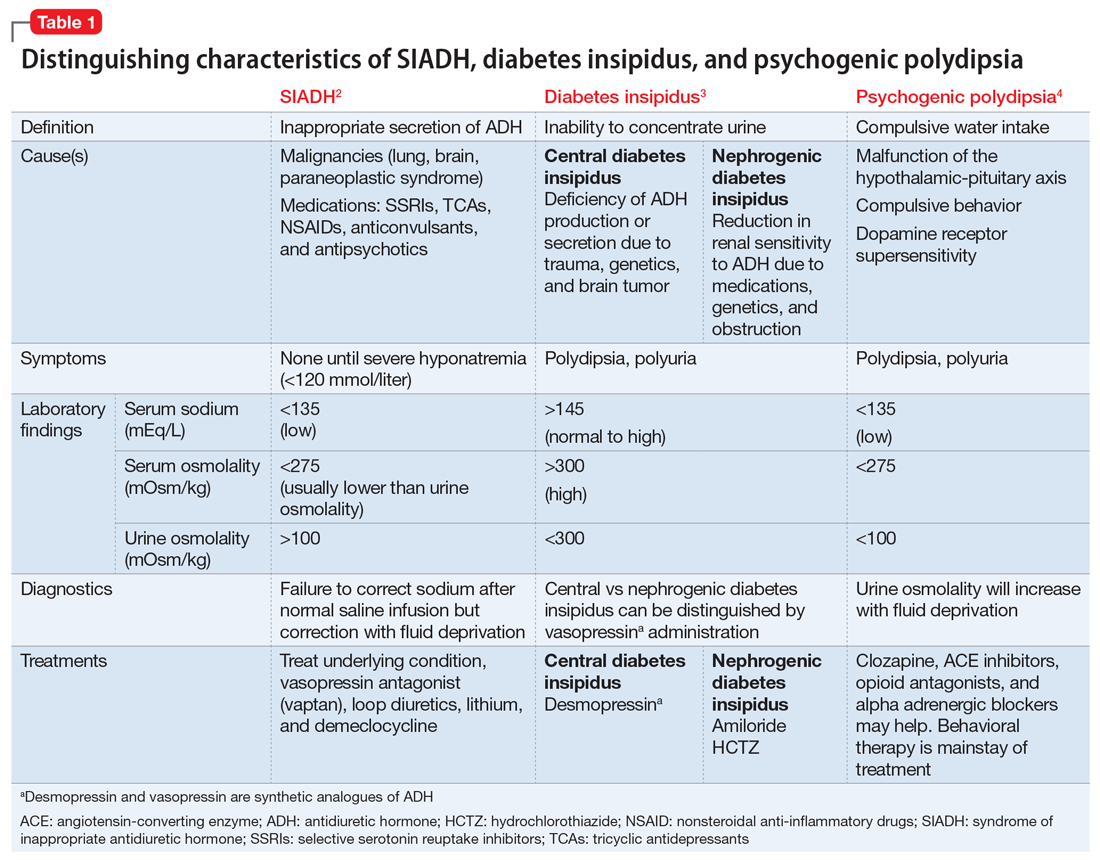
The differential diagnosis for hyponatremia is broad in the setting of psychiatric illness. Low sodium levels could be due to psychotropic medications, psychiatrically-driven behaviors, or an underlying medical problem. Our differential diagnosis for Mr. F included iatrogenic syndrome of inappropriate antidiuretic hormone (SIADH), diabetes insipidus, or psychogenic polydipsia, a form of primary polydipsia. Other causes of primary polydipsia are related to substances, such as heavy beer intakeor use of 3,4-methylenedioxymethamphetamine (MDMA, also known as “ecstasy”), or brain lesions,1 but these causes were less likely given Mr. F’s negative urine toxicology and head imaging.
While psychogenic polydipsia is due to increased water consumption, both SIADH and diabetes insipidus are due to alterations in fluid homeostasis.2,3 Table 12-4 outlines distinguishing characteristics of SIADH, diabetes insipidus, and psychogenic polydipsia. Urine studies were not pursued because Mr. F’s sodium resolved and acute concerns, such as malignancy or infection, were ruled out. Mr. F’s hyponatremia was presumed to be due to psychogenic polydipsia because of his increased fluid intake and normalization of sodium with hypertonic fluids and subsequent fluid restriction. During this time, he was managed on the surgical service; the plan was to pursue urine studies and possibly a fluid challenge if his hyponatremia persisted.
EVALUATION Delirium resolves, delusions persist
While Mr. F is on the surgical service, the treatment team focuses on stabilizing his sodium level and assessing for causes of altered mental status that led to his fall. Psychiatry is consulted for management of his agitation. Following the gradual correction of his sodium level and extubation, his sensorium improves. By hospital Day 5, Mr. F’s delirium resolves.
During this time, Mr. F’s disorganization and religious delusions become apparent. He spends much of his time reading his Bible. He has poor hygiene and limited engagement in activities of daily living. Due to his psychosis and inability to care for himself, Mr. F is transferred to the psychiatric unit with consent from his mother.
Continue to: TREATMENT Olanzapine and fluid restriction
TREATMENT Olanzapine and fluid restriction
In the psychiatric unit, Mr. F is restarted on olanzapine, but not on perphenazine due to anticholinergic effects and not on propranolol due to continued orthostatic hypotension. Five days later, he is at his baseline level of functioning with residual psychosis. His fluid intake is restricted to <1.5 L per day and he is easily compliant.
Mr. F’s mother is comfortable with his discharge home on a regimen of olanzapine, 25 mg/d, and the team discusses the fluid restrictions with her. The treatment team suggests initiating an LAI before Mr. F is discharged, but this is not pursued because his mother thinks he is doing well with the oral medication. She wants to monitor him with the medication changes in the clinic before pursuing an LAI; however, she is open to it in the future.
The authors’ observations
Approximately 20% of patients with schizophrenia may experience psychogenic polydipsia.4,5 The cause of psychogenic polydipsia in patients with serious mental illness is multifactorial. It may stem from malfunction of the hypothalamic-pituitary axis, which leads to alterations in antidiuretic hormone secretion and function.4-6
Mr. F’s case highlights several challenges associated with treating psychogenic polydipsia in patients with serious mental illness. Antipsychotics with high dopamine affinity, such as risperidone and haloperidol, may increase the risk of psychogenic polydipsia, while antipsychotics with lower dopamine affinity, such as clozapine, may decrease the occurrence.5 Antipsychotics block postsynaptic dopamine receptors, which can induce supersensitivity by increasing presynaptic dopamine release in the hypothalamic areas, where thirst regulation occurs. This increase in dopamine leads to increased thirst drive and fluid intake.3
Quetiapine or clozapine may have been a better antipsychotic choice because these agents have lower D2 receptor affinity, whereas olanzapine has intermediate binding to D2 receptors.6,7 However, quetiapine and clozapine are more strongly associated with orthostasis, which was a concern during Mr. F’s hospitalization. The weekly laboratory testing required with clozapine use would have been an unfeasible burden for Mr. F because he lived in a rural environment. Perphenazine was not continued due to higher D2 affinity and anticholinergic effects, which can increase thirst.6
Continue to: In addition to switching...
In addition to switching to an antipsychotic with looser D2 binding, other medications for treating polydipsia have been studied. It is hypothesized that the alpha-2 adrenergic system may play a role in thirst regulation. For example, mianserin, an alpha-2 antagonist, may decrease water intake. However, studies have been small and inconsistent.8,9 Propranolol,10 a beta adrenergic receptor blocker; irbesartan,11 an angiotensin-II receptor blocker; demeclocycline,12 a tetracycline that inhibits antidiuretic hormone action; and naltrexone,9 a mu opioid antagonist, have been studied with inconclusive results and a variety of adverse effects5,7,13 (Table 28-13).

Behavioral interventions for patients with psychogenic polydipsia include fluid restriction, twice-daily weight checks, cognitive-behavioral therapy, and reinforcement schedules, which may be useful but less realistic due to need for increased supervision.11,12 Patient and family education on the signs of hyponatremia are important to prevent serious complications, such as those Mr. F experienced.
OUTCOME Repeated hospitalizations
Mr. F is discharged with follow-up in our psychiatry clinic and attends 1 appointment. At that time, his mother reports that Mr. F is compliant with his medication and has limited fluid intake. However, over the next 2 months, he is admitted to our psychiatric unit twice with similar presentations. Each time, the treatment team has extensive discussions with Mr. F’s mother about strategies to limit his water intake and the possibility of residential placement due to his need for a higher level of care. Although she acknowledges that nursing home placement may be needed in the future, she is not ready to take this step.
Three months later, Mr. F returns to our hospital with severe abdominal pain and is found to have a perforated bowel obstruction. His sodium is within normal limits on presentation, and the psychiatry team is not involved during this hospitalization. Mr. F is treated for sepsis and undergoes 3 exploratory laparotomies with continued decline in his health. He dies during this hospitalization. The cause of Mr. F’s perforated bowel obstruction is not determined, and his family does not pursue an autopsy.
The authors’ observations
At Mr. F’s final hospital presentation, his sodium was normal. It is possible Mr. F and his mother had found an acceptable fluid restriction routine, and he may have been doing better from a psychiatric perspective, but this will remain unknown.
Continue to: This case highlights...
This case highlights the clinical and ethical complexity of treating patients with psychogenic polydipsia. Because Mr. F no longer had autonomy, we had to determine if his mother was acting in his best interest as his guardian. Guardianship requirements and expectations vary by state. In our state of Missouri, a guardian is appointed by the court to act in the best interest of the ward, and may be a family member (preferred) or state-appointed. The guardian is responsible for providing the ward’s care and is in charge of financial and medical decisions. In Missouri, the guardian must assure the ward resides in the “least restrictive setting reasonably available,” which is the minimum necessary to provide the ward safe care and housing.14 Full guardianship, as in Mr. F’s case, is different from limited guardianship, which is an option in states such as Missouri. In limited guardianship, the court decides the extent of the guardian’s role in decisions for the ward.14,15
Mr. F’s mother believed she was acting in her son’s best interest by having him home with his family. She believed by living at home, he would derive more enjoyment from life than living in a nursing home. By the time Mr. F presented to our hospital, he had been living with decompensated schizophrenia for years, so some level of psychosis was likely to persist, even with treatment. Given his increasingly frequent hospitalizations for hyponatremia due to increased water intake, more intense supervision may have been needed to maintain his safety, in line with nonmaleficence. The treatment team considered Mr. F’s best interest when discussing placement and worked to understand his mother’s preferences.
His mother continued to acknowledge the need for changes and adjustments at home. She was receptive to the need for fluid restriction and increased structure at home. Therefore, we felt she continued to be acting in his best interest, and his home would be the least restrictive setting for his care. If Mr. F had continued to require repeated hospitalizations and had not passed away, we would have pursued an ethics consult to discuss the need for nursing home placement and how to best approach this with Mr. F’s mother.
Bottom Line
Patients with serious mental illness who present with hyponatremia should be evaluated for psychogenic polydipsia by assessing their dietary and fluid intakes, along with collateral from family. The use of antipsychotics with high dopamine affinity may increase the risk of psychogenic polydipsia. Behavioral interventions include fluid restriction, weight checks, cognitive-behavioral therapy, and reinforcement schedules.
Related Resources
- Sharp CS, Wilson MP. Hyponatremia. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer International Publishing; 2018:115-119. doi:10.1007/ 978-3-319-58260-3_21
- Sailer C, Winzeler B, Christ-Crain M. Primary polydipsia in the medical and psychiatric patient: characteristics, complications and therapy. Swiss Med Wkly. 2017;147:w14514. doi:10.4414/ smw.2017.14514
Drug Brand Names
Amiloride • Midamor
Aripiprazole • Abilify
Clonidine • Catapres
Clozapine • Clozaril
Demeclocycline • Declomycin
Desmopressin • DDAVP
Haloperidol • Haldol
Irbesartan • Avapro
Lithium • Eskalith, Lithobid
Losartan • Cozaar
Mianserin • Tolvon
Naloxone • Narcan
Naltrexone • Revia
Olanzapine • Zyprexa
Perphenazine • Trilafon
Propranolol • Inderal LA
Quetiapine • Seroquel
Risperidone • Risperda
1. Sharp CS, Wilson MP. Hyponatremia. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer International Publishing; 2018:115-119. doi:10.1007/978-3-319-58260-3_21
2. Gross P. Clinical management of SIADH. Ther Adv Endocrinol Metab. 2012;3(2):61-73. doi:10.1177/2042018812437561
3. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primer. 2019;5(1):54. doi:10.1038/s41572-019-0103-2
4. Ahmadi L, Goldman MB. Primary polydipsia: update. Best Pract Res Clin Endocrinol Metab. 2020;34(5):101469. doi:10.1016/j.beem.2020.101469
5. Kirino S, Sakuma M, Misawa F, et al. Relationship between polydipsia and antipsychotics: a systematic review of clinical studies and case reports. Prog Neuropsychopharmacol Biol Psychiatry. 2020;96:109756. doi:10.1016/j.pnpbp.2019.109756
6. Siafis S, Tzachanis D, Samara M, et al. Antipsychotic drugs: from receptor-binding profiles to metabolic side effects. Curr Neuropharmacol. 2018;16(8):1210-1223. doi:10.2174/1570159X15666170630163616
7. Seeman P, Tallerico T. Antipsychotic drugs which elicit little or no parkinsonism bind more loosely than dopamine to brain D2 receptors, yet occupy high levels of these receptors. Mol Psychiatry. 1998;3(2):123-134. doi:10.1038/sj.mp.4000336
8. Hayashi T, Nishikawa T, Koga I, et al. Involvement of the α 2 -adrenergic system in polydipsia in schizophrenic patients: a pilot study. Psychopharmacology (Berl). 1997;130(4):382-386. doi:10.1007/s002130050254
9. Rizvi S, Gold J, Khan AM. Role of naltrexone in improving compulsive drinking in psychogenic polydipsia. Cureus. 2019;11(8):e5320. doi:10.7759/cureus.5320
10. Kishi Y, Kurosawa H, Endo S. Is propranolol effective in primary polydipsia? Int J Psychiatry Med. 1998;28(3):315-325. doi:10.2190/QPWL-14H7-HPGG-A29D
11. Kruse D, Pantelis C, Rudd R, et al. Treatment of psychogenic polydipsia: comparison of risperidone and olanzapine, and the effects of an adjunctive angiotensin-II receptor blocking drug (irbesartan). Aust N Z J Psychiatry. 2001;35(1):65-68. doi:10.1046/j.1440-1614.2001.00847.x
12. Alexander RC, Karp BI, Thompson S, et al. A double blind, placebo-controlled trial of demeclocycline treatment of polydipsia-hyponatremia in chronically psychotic patients. Biol Psychiatry. 1991;30(4):417-420. doi:10.1016/0006-3223(91)90300-B
13. Valente S, Fisher D. Recognizing and managing psychogenic polydipsia in mental health. J Nurse Pract. 2010;6(7):546-550. doi:10.1016/j.nurpra.2010.03.004
14. Barton R, Esq SL, Lockett LL. The use of conservatorships and adult guardianships and other options in the care of the mentally ill in the United States. World Guard Congr. Published May 29, 2014. Accessed June 18, 2021. http://www.guardianship.org/IRL/Resources/Handouts/Family%20Members%20as%20Guardians_Handout.pdf
15. ABA Commission on Law & Aging. Adult Guardianship Statutory Table of Authorities. ABA. Published January 2021. Accessed June 17, 2021. https://www.americanbar.org/content/dam/aba/administrative/law_aging/2019-adult-guardianship-statutory-table-of-authorities.pdf
CASE Unresponsive after a presumed seizure
Mr. F, age 44, has schizophrenia. He is brought to the hospital by ambulance after he is found on the ground outside of his mother’s house following a presumed seizure and fall. On arrival to the emergency department, he is unresponsive. His laboratory values are significant for a sodium level of 110 mEq/L (reference range: 135 to 145 mEq/L), indicating hyponatremia.
HISTORY Fixated on purity
Mr. F’s mother reports that Mr. F had an unremarkable childhood. He was raised in a household with both parents and a younger sister. Mr. F did well academically and studied engineering and physics in college. There was no reported history of trauma or substance use.
During his senior year of college, Mr. F began experiencing paranoia, auditory hallucinations, and religious delusions. He required hospitalization and was diagnosed with schizophrenia. Following multiple hospitalizations over 5 years, he moved in with his mother, who was granted guardianship.
His mother said Mr. F’s religious delusions were of purity and cleansing the soul. He spent hours memorizing the Bible and would go for days without eating but would drink large amounts of water. She said she thought this was due to his desire to flush out imperfections.
In the past 3 years, Mr. F has been hospitalized several times for severe hyponatremia. At home, his mother attempted to restrict his water intake. However, Mr. F would still drink out of sinks and hoses. Mr. F’s mother reports that over the past month he had become more isolated. He would spend entire days reading the Bible, and his water intake had further increased.
Prior medication trials for Mr. F included haloperidol, up to 10 mg twice per day; aripiprazole, up to 20 mg/d; and risperidone, up to 6 mg nightly. These had been effective, but Mr. F had difficulty with adherence. He did not receive a long-acting injectable (LAI) antipsychotic initially due to lack of access at the rural clinic where he was treated, and later due to his mother’s preference for her son to receive oral medications. Prior to his current presentation, Mr. F’s medication regimen was olanzapine, 10 mg twice a day; perphenazine, 8 mg twice a day; and long-acting propranolol, 60 mg/d. Mr. F had no other chronic medical problems.
EVALUATION Hyponatremia, but why?
Mr. F is intubated and admitted to the surgical service for stabilization due to injuries from his fall. He has fractures of his right sinus and bilateral nasal bones, which are managed nonoperatively. He is delirious, with waxing and waning attention, memory disturbances, and disorientation. His psychotropic medications are held.
Continue to: Imaging of his head...
Imaging of his head does not reveal acute abnormalities suggesting a malignant or paraneoplastic process, and there are no concerns for ongoing seizures. An infection workup is negative. His urine toxicology is negative and blood alcohol level is 0. His sodium normalizes after 3 days of IV fluids and fluid restriction. Therefore, further tests to differentiate the causes of hyponatremia, such as urine electrolytes and urine osmolality, are not pursued.
[polldaddy:10910406]
The authors’ observations
The differential diagnosis for hyponatremia is broad in the setting of psychiatric illness. Low sodium levels could be due to psychotropic medications, psychiatrically-driven behaviors, or an underlying medical problem. Our differential diagnosis for Mr. F included iatrogenic syndrome of inappropriate antidiuretic hormone (SIADH), diabetes insipidus, or psychogenic polydipsia, a form of primary polydipsia. Other causes of primary polydipsia are related to substances, such as heavy beer intakeor use of 3,4-methylenedioxymethamphetamine (MDMA, also known as “ecstasy”), or brain lesions,1 but these causes were less likely given Mr. F’s negative urine toxicology and head imaging.
While psychogenic polydipsia is due to increased water consumption, both SIADH and diabetes insipidus are due to alterations in fluid homeostasis.2,3 Table 12-4 outlines distinguishing characteristics of SIADH, diabetes insipidus, and psychogenic polydipsia. Urine studies were not pursued because Mr. F’s sodium resolved and acute concerns, such as malignancy or infection, were ruled out. Mr. F’s hyponatremia was presumed to be due to psychogenic polydipsia because of his increased fluid intake and normalization of sodium with hypertonic fluids and subsequent fluid restriction. During this time, he was managed on the surgical service; the plan was to pursue urine studies and possibly a fluid challenge if his hyponatremia persisted.

EVALUATION Delirium resolves, delusions persist
While Mr. F is on the surgical service, the treatment team focuses on stabilizing his sodium level and assessing for causes of altered mental status that led to his fall. Psychiatry is consulted for management of his agitation. Following the gradual correction of his sodium level and extubation, his sensorium improves. By hospital Day 5, Mr. F’s delirium resolves.
During this time, Mr. F’s disorganization and religious delusions become apparent. He spends much of his time reading his Bible. He has poor hygiene and limited engagement in activities of daily living. Due to his psychosis and inability to care for himself, Mr. F is transferred to the psychiatric unit with consent from his mother.
Continue to: TREATMENT Olanzapine and fluid restriction
TREATMENT Olanzapine and fluid restriction
In the psychiatric unit, Mr. F is restarted on olanzapine, but not on perphenazine due to anticholinergic effects and not on propranolol due to continued orthostatic hypotension. Five days later, he is at his baseline level of functioning with residual psychosis. His fluid intake is restricted to <1.5 L per day and he is easily compliant.
Mr. F’s mother is comfortable with his discharge home on a regimen of olanzapine, 25 mg/d, and the team discusses the fluid restrictions with her. The treatment team suggests initiating an LAI before Mr. F is discharged, but this is not pursued because his mother thinks he is doing well with the oral medication. She wants to monitor him with the medication changes in the clinic before pursuing an LAI; however, she is open to it in the future.
The authors’ observations
Approximately 20% of patients with schizophrenia may experience psychogenic polydipsia.4,5 The cause of psychogenic polydipsia in patients with serious mental illness is multifactorial. It may stem from malfunction of the hypothalamic-pituitary axis, which leads to alterations in antidiuretic hormone secretion and function.4-6
Mr. F’s case highlights several challenges associated with treating psychogenic polydipsia in patients with serious mental illness. Antipsychotics with high dopamine affinity, such as risperidone and haloperidol, may increase the risk of psychogenic polydipsia, while antipsychotics with lower dopamine affinity, such as clozapine, may decrease the occurrence.5 Antipsychotics block postsynaptic dopamine receptors, which can induce supersensitivity by increasing presynaptic dopamine release in the hypothalamic areas, where thirst regulation occurs. This increase in dopamine leads to increased thirst drive and fluid intake.3
Quetiapine or clozapine may have been a better antipsychotic choice because these agents have lower D2 receptor affinity, whereas olanzapine has intermediate binding to D2 receptors.6,7 However, quetiapine and clozapine are more strongly associated with orthostasis, which was a concern during Mr. F’s hospitalization. The weekly laboratory testing required with clozapine use would have been an unfeasible burden for Mr. F because he lived in a rural environment. Perphenazine was not continued due to higher D2 affinity and anticholinergic effects, which can increase thirst.6
Continue to: In addition to switching...
In addition to switching to an antipsychotic with looser D2 binding, other medications for treating polydipsia have been studied. It is hypothesized that the alpha-2 adrenergic system may play a role in thirst regulation. For example, mianserin, an alpha-2 antagonist, may decrease water intake. However, studies have been small and inconsistent.8,9 Propranolol,10 a beta adrenergic receptor blocker; irbesartan,11 an angiotensin-II receptor blocker; demeclocycline,12 a tetracycline that inhibits antidiuretic hormone action; and naltrexone,9 a mu opioid antagonist, have been studied with inconclusive results and a variety of adverse effects5,7,13 (Table 28-13).

Behavioral interventions for patients with psychogenic polydipsia include fluid restriction, twice-daily weight checks, cognitive-behavioral therapy, and reinforcement schedules, which may be useful but less realistic due to need for increased supervision.11,12 Patient and family education on the signs of hyponatremia are important to prevent serious complications, such as those Mr. F experienced.
OUTCOME Repeated hospitalizations
Mr. F is discharged with follow-up in our psychiatry clinic and attends 1 appointment. At that time, his mother reports that Mr. F is compliant with his medication and has limited fluid intake. However, over the next 2 months, he is admitted to our psychiatric unit twice with similar presentations. Each time, the treatment team has extensive discussions with Mr. F’s mother about strategies to limit his water intake and the possibility of residential placement due to his need for a higher level of care. Although she acknowledges that nursing home placement may be needed in the future, she is not ready to take this step.
Three months later, Mr. F returns to our hospital with severe abdominal pain and is found to have a perforated bowel obstruction. His sodium is within normal limits on presentation, and the psychiatry team is not involved during this hospitalization. Mr. F is treated for sepsis and undergoes 3 exploratory laparotomies with continued decline in his health. He dies during this hospitalization. The cause of Mr. F’s perforated bowel obstruction is not determined, and his family does not pursue an autopsy.
The authors’ observations
At Mr. F’s final hospital presentation, his sodium was normal. It is possible Mr. F and his mother had found an acceptable fluid restriction routine, and he may have been doing better from a psychiatric perspective, but this will remain unknown.
Continue to: This case highlights...
This case highlights the clinical and ethical complexity of treating patients with psychogenic polydipsia. Because Mr. F no longer had autonomy, we had to determine if his mother was acting in his best interest as his guardian. Guardianship requirements and expectations vary by state. In our state of Missouri, a guardian is appointed by the court to act in the best interest of the ward, and may be a family member (preferred) or state-appointed. The guardian is responsible for providing the ward’s care and is in charge of financial and medical decisions. In Missouri, the guardian must assure the ward resides in the “least restrictive setting reasonably available,” which is the minimum necessary to provide the ward safe care and housing.14 Full guardianship, as in Mr. F’s case, is different from limited guardianship, which is an option in states such as Missouri. In limited guardianship, the court decides the extent of the guardian’s role in decisions for the ward.14,15
Mr. F’s mother believed she was acting in her son’s best interest by having him home with his family. She believed by living at home, he would derive more enjoyment from life than living in a nursing home. By the time Mr. F presented to our hospital, he had been living with decompensated schizophrenia for years, so some level of psychosis was likely to persist, even with treatment. Given his increasingly frequent hospitalizations for hyponatremia due to increased water intake, more intense supervision may have been needed to maintain his safety, in line with nonmaleficence. The treatment team considered Mr. F’s best interest when discussing placement and worked to understand his mother’s preferences.
His mother continued to acknowledge the need for changes and adjustments at home. She was receptive to the need for fluid restriction and increased structure at home. Therefore, we felt she continued to be acting in his best interest, and his home would be the least restrictive setting for his care. If Mr. F had continued to require repeated hospitalizations and had not passed away, we would have pursued an ethics consult to discuss the need for nursing home placement and how to best approach this with Mr. F’s mother.
Bottom Line
Patients with serious mental illness who present with hyponatremia should be evaluated for psychogenic polydipsia by assessing their dietary and fluid intakes, along with collateral from family. The use of antipsychotics with high dopamine affinity may increase the risk of psychogenic polydipsia. Behavioral interventions include fluid restriction, weight checks, cognitive-behavioral therapy, and reinforcement schedules.
Related Resources
- Sharp CS, Wilson MP. Hyponatremia. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer International Publishing; 2018:115-119. doi:10.1007/ 978-3-319-58260-3_21
- Sailer C, Winzeler B, Christ-Crain M. Primary polydipsia in the medical and psychiatric patient: characteristics, complications and therapy. Swiss Med Wkly. 2017;147:w14514. doi:10.4414/ smw.2017.14514
Drug Brand Names
Amiloride • Midamor
Aripiprazole • Abilify
Clonidine • Catapres
Clozapine • Clozaril
Demeclocycline • Declomycin
Desmopressin • DDAVP
Haloperidol • Haldol
Irbesartan • Avapro
Lithium • Eskalith, Lithobid
Losartan • Cozaar
Mianserin • Tolvon
Naloxone • Narcan
Naltrexone • Revia
Olanzapine • Zyprexa
Perphenazine • Trilafon
Propranolol • Inderal LA
Quetiapine • Seroquel
Risperidone • Risperda
CASE Unresponsive after a presumed seizure
Mr. F, age 44, has schizophrenia. He is brought to the hospital by ambulance after he is found on the ground outside of his mother’s house following a presumed seizure and fall. On arrival to the emergency department, he is unresponsive. His laboratory values are significant for a sodium level of 110 mEq/L (reference range: 135 to 145 mEq/L), indicating hyponatremia.
HISTORY Fixated on purity
Mr. F’s mother reports that Mr. F had an unremarkable childhood. He was raised in a household with both parents and a younger sister. Mr. F did well academically and studied engineering and physics in college. There was no reported history of trauma or substance use.
During his senior year of college, Mr. F began experiencing paranoia, auditory hallucinations, and religious delusions. He required hospitalization and was diagnosed with schizophrenia. Following multiple hospitalizations over 5 years, he moved in with his mother, who was granted guardianship.
His mother said Mr. F’s religious delusions were of purity and cleansing the soul. He spent hours memorizing the Bible and would go for days without eating but would drink large amounts of water. She said she thought this was due to his desire to flush out imperfections.
In the past 3 years, Mr. F has been hospitalized several times for severe hyponatremia. At home, his mother attempted to restrict his water intake. However, Mr. F would still drink out of sinks and hoses. Mr. F’s mother reports that over the past month he had become more isolated. He would spend entire days reading the Bible, and his water intake had further increased.
Prior medication trials for Mr. F included haloperidol, up to 10 mg twice per day; aripiprazole, up to 20 mg/d; and risperidone, up to 6 mg nightly. These had been effective, but Mr. F had difficulty with adherence. He did not receive a long-acting injectable (LAI) antipsychotic initially due to lack of access at the rural clinic where he was treated, and later due to his mother’s preference for her son to receive oral medications. Prior to his current presentation, Mr. F’s medication regimen was olanzapine, 10 mg twice a day; perphenazine, 8 mg twice a day; and long-acting propranolol, 60 mg/d. Mr. F had no other chronic medical problems.
EVALUATION Hyponatremia, but why?
Mr. F is intubated and admitted to the surgical service for stabilization due to injuries from his fall. He has fractures of his right sinus and bilateral nasal bones, which are managed nonoperatively. He is delirious, with waxing and waning attention, memory disturbances, and disorientation. His psychotropic medications are held.
Continue to: Imaging of his head...
Imaging of his head does not reveal acute abnormalities suggesting a malignant or paraneoplastic process, and there are no concerns for ongoing seizures. An infection workup is negative. His urine toxicology is negative and blood alcohol level is 0. His sodium normalizes after 3 days of IV fluids and fluid restriction. Therefore, further tests to differentiate the causes of hyponatremia, such as urine electrolytes and urine osmolality, are not pursued.
[polldaddy:10910406]
The authors’ observations
The differential diagnosis for hyponatremia is broad in the setting of psychiatric illness. Low sodium levels could be due to psychotropic medications, psychiatrically-driven behaviors, or an underlying medical problem. Our differential diagnosis for Mr. F included iatrogenic syndrome of inappropriate antidiuretic hormone (SIADH), diabetes insipidus, or psychogenic polydipsia, a form of primary polydipsia. Other causes of primary polydipsia are related to substances, such as heavy beer intakeor use of 3,4-methylenedioxymethamphetamine (MDMA, also known as “ecstasy”), or brain lesions,1 but these causes were less likely given Mr. F’s negative urine toxicology and head imaging.
While psychogenic polydipsia is due to increased water consumption, both SIADH and diabetes insipidus are due to alterations in fluid homeostasis.2,3 Table 12-4 outlines distinguishing characteristics of SIADH, diabetes insipidus, and psychogenic polydipsia. Urine studies were not pursued because Mr. F’s sodium resolved and acute concerns, such as malignancy or infection, were ruled out. Mr. F’s hyponatremia was presumed to be due to psychogenic polydipsia because of his increased fluid intake and normalization of sodium with hypertonic fluids and subsequent fluid restriction. During this time, he was managed on the surgical service; the plan was to pursue urine studies and possibly a fluid challenge if his hyponatremia persisted.

EVALUATION Delirium resolves, delusions persist
While Mr. F is on the surgical service, the treatment team focuses on stabilizing his sodium level and assessing for causes of altered mental status that led to his fall. Psychiatry is consulted for management of his agitation. Following the gradual correction of his sodium level and extubation, his sensorium improves. By hospital Day 5, Mr. F’s delirium resolves.
During this time, Mr. F’s disorganization and religious delusions become apparent. He spends much of his time reading his Bible. He has poor hygiene and limited engagement in activities of daily living. Due to his psychosis and inability to care for himself, Mr. F is transferred to the psychiatric unit with consent from his mother.
Continue to: TREATMENT Olanzapine and fluid restriction
TREATMENT Olanzapine and fluid restriction
In the psychiatric unit, Mr. F is restarted on olanzapine, but not on perphenazine due to anticholinergic effects and not on propranolol due to continued orthostatic hypotension. Five days later, he is at his baseline level of functioning with residual psychosis. His fluid intake is restricted to <1.5 L per day and he is easily compliant.
Mr. F’s mother is comfortable with his discharge home on a regimen of olanzapine, 25 mg/d, and the team discusses the fluid restrictions with her. The treatment team suggests initiating an LAI before Mr. F is discharged, but this is not pursued because his mother thinks he is doing well with the oral medication. She wants to monitor him with the medication changes in the clinic before pursuing an LAI; however, she is open to it in the future.
The authors’ observations
Approximately 20% of patients with schizophrenia may experience psychogenic polydipsia.4,5 The cause of psychogenic polydipsia in patients with serious mental illness is multifactorial. It may stem from malfunction of the hypothalamic-pituitary axis, which leads to alterations in antidiuretic hormone secretion and function.4-6
Mr. F’s case highlights several challenges associated with treating psychogenic polydipsia in patients with serious mental illness. Antipsychotics with high dopamine affinity, such as risperidone and haloperidol, may increase the risk of psychogenic polydipsia, while antipsychotics with lower dopamine affinity, such as clozapine, may decrease the occurrence.5 Antipsychotics block postsynaptic dopamine receptors, which can induce supersensitivity by increasing presynaptic dopamine release in the hypothalamic areas, where thirst regulation occurs. This increase in dopamine leads to increased thirst drive and fluid intake.3
Quetiapine or clozapine may have been a better antipsychotic choice because these agents have lower D2 receptor affinity, whereas olanzapine has intermediate binding to D2 receptors.6,7 However, quetiapine and clozapine are more strongly associated with orthostasis, which was a concern during Mr. F’s hospitalization. The weekly laboratory testing required with clozapine use would have been an unfeasible burden for Mr. F because he lived in a rural environment. Perphenazine was not continued due to higher D2 affinity and anticholinergic effects, which can increase thirst.6
Continue to: In addition to switching...
In addition to switching to an antipsychotic with looser D2 binding, other medications for treating polydipsia have been studied. It is hypothesized that the alpha-2 adrenergic system may play a role in thirst regulation. For example, mianserin, an alpha-2 antagonist, may decrease water intake. However, studies have been small and inconsistent.8,9 Propranolol,10 a beta adrenergic receptor blocker; irbesartan,11 an angiotensin-II receptor blocker; demeclocycline,12 a tetracycline that inhibits antidiuretic hormone action; and naltrexone,9 a mu opioid antagonist, have been studied with inconclusive results and a variety of adverse effects5,7,13 (Table 28-13).

Behavioral interventions for patients with psychogenic polydipsia include fluid restriction, twice-daily weight checks, cognitive-behavioral therapy, and reinforcement schedules, which may be useful but less realistic due to need for increased supervision.11,12 Patient and family education on the signs of hyponatremia are important to prevent serious complications, such as those Mr. F experienced.
OUTCOME Repeated hospitalizations
Mr. F is discharged with follow-up in our psychiatry clinic and attends 1 appointment. At that time, his mother reports that Mr. F is compliant with his medication and has limited fluid intake. However, over the next 2 months, he is admitted to our psychiatric unit twice with similar presentations. Each time, the treatment team has extensive discussions with Mr. F’s mother about strategies to limit his water intake and the possibility of residential placement due to his need for a higher level of care. Although she acknowledges that nursing home placement may be needed in the future, she is not ready to take this step.
Three months later, Mr. F returns to our hospital with severe abdominal pain and is found to have a perforated bowel obstruction. His sodium is within normal limits on presentation, and the psychiatry team is not involved during this hospitalization. Mr. F is treated for sepsis and undergoes 3 exploratory laparotomies with continued decline in his health. He dies during this hospitalization. The cause of Mr. F’s perforated bowel obstruction is not determined, and his family does not pursue an autopsy.
The authors’ observations
At Mr. F’s final hospital presentation, his sodium was normal. It is possible Mr. F and his mother had found an acceptable fluid restriction routine, and he may have been doing better from a psychiatric perspective, but this will remain unknown.
Continue to: This case highlights...
This case highlights the clinical and ethical complexity of treating patients with psychogenic polydipsia. Because Mr. F no longer had autonomy, we had to determine if his mother was acting in his best interest as his guardian. Guardianship requirements and expectations vary by state. In our state of Missouri, a guardian is appointed by the court to act in the best interest of the ward, and may be a family member (preferred) or state-appointed. The guardian is responsible for providing the ward’s care and is in charge of financial and medical decisions. In Missouri, the guardian must assure the ward resides in the “least restrictive setting reasonably available,” which is the minimum necessary to provide the ward safe care and housing.14 Full guardianship, as in Mr. F’s case, is different from limited guardianship, which is an option in states such as Missouri. In limited guardianship, the court decides the extent of the guardian’s role in decisions for the ward.14,15
Mr. F’s mother believed she was acting in her son’s best interest by having him home with his family. She believed by living at home, he would derive more enjoyment from life than living in a nursing home. By the time Mr. F presented to our hospital, he had been living with decompensated schizophrenia for years, so some level of psychosis was likely to persist, even with treatment. Given his increasingly frequent hospitalizations for hyponatremia due to increased water intake, more intense supervision may have been needed to maintain his safety, in line with nonmaleficence. The treatment team considered Mr. F’s best interest when discussing placement and worked to understand his mother’s preferences.
His mother continued to acknowledge the need for changes and adjustments at home. She was receptive to the need for fluid restriction and increased structure at home. Therefore, we felt she continued to be acting in his best interest, and his home would be the least restrictive setting for his care. If Mr. F had continued to require repeated hospitalizations and had not passed away, we would have pursued an ethics consult to discuss the need for nursing home placement and how to best approach this with Mr. F’s mother.
Bottom Line
Patients with serious mental illness who present with hyponatremia should be evaluated for psychogenic polydipsia by assessing their dietary and fluid intakes, along with collateral from family. The use of antipsychotics with high dopamine affinity may increase the risk of psychogenic polydipsia. Behavioral interventions include fluid restriction, weight checks, cognitive-behavioral therapy, and reinforcement schedules.
Related Resources
- Sharp CS, Wilson MP. Hyponatremia. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer International Publishing; 2018:115-119. doi:10.1007/ 978-3-319-58260-3_21
- Sailer C, Winzeler B, Christ-Crain M. Primary polydipsia in the medical and psychiatric patient: characteristics, complications and therapy. Swiss Med Wkly. 2017;147:w14514. doi:10.4414/ smw.2017.14514
Drug Brand Names
Amiloride • Midamor
Aripiprazole • Abilify
Clonidine • Catapres
Clozapine • Clozaril
Demeclocycline • Declomycin
Desmopressin • DDAVP
Haloperidol • Haldol
Irbesartan • Avapro
Lithium • Eskalith, Lithobid
Losartan • Cozaar
Mianserin • Tolvon
Naloxone • Narcan
Naltrexone • Revia
Olanzapine • Zyprexa
Perphenazine • Trilafon
Propranolol • Inderal LA
Quetiapine • Seroquel
Risperidone • Risperda
1. Sharp CS, Wilson MP. Hyponatremia. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer International Publishing; 2018:115-119. doi:10.1007/978-3-319-58260-3_21
2. Gross P. Clinical management of SIADH. Ther Adv Endocrinol Metab. 2012;3(2):61-73. doi:10.1177/2042018812437561
3. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primer. 2019;5(1):54. doi:10.1038/s41572-019-0103-2
4. Ahmadi L, Goldman MB. Primary polydipsia: update. Best Pract Res Clin Endocrinol Metab. 2020;34(5):101469. doi:10.1016/j.beem.2020.101469
5. Kirino S, Sakuma M, Misawa F, et al. Relationship between polydipsia and antipsychotics: a systematic review of clinical studies and case reports. Prog Neuropsychopharmacol Biol Psychiatry. 2020;96:109756. doi:10.1016/j.pnpbp.2019.109756
6. Siafis S, Tzachanis D, Samara M, et al. Antipsychotic drugs: from receptor-binding profiles to metabolic side effects. Curr Neuropharmacol. 2018;16(8):1210-1223. doi:10.2174/1570159X15666170630163616
7. Seeman P, Tallerico T. Antipsychotic drugs which elicit little or no parkinsonism bind more loosely than dopamine to brain D2 receptors, yet occupy high levels of these receptors. Mol Psychiatry. 1998;3(2):123-134. doi:10.1038/sj.mp.4000336
8. Hayashi T, Nishikawa T, Koga I, et al. Involvement of the α 2 -adrenergic system in polydipsia in schizophrenic patients: a pilot study. Psychopharmacology (Berl). 1997;130(4):382-386. doi:10.1007/s002130050254
9. Rizvi S, Gold J, Khan AM. Role of naltrexone in improving compulsive drinking in psychogenic polydipsia. Cureus. 2019;11(8):e5320. doi:10.7759/cureus.5320
10. Kishi Y, Kurosawa H, Endo S. Is propranolol effective in primary polydipsia? Int J Psychiatry Med. 1998;28(3):315-325. doi:10.2190/QPWL-14H7-HPGG-A29D
11. Kruse D, Pantelis C, Rudd R, et al. Treatment of psychogenic polydipsia: comparison of risperidone and olanzapine, and the effects of an adjunctive angiotensin-II receptor blocking drug (irbesartan). Aust N Z J Psychiatry. 2001;35(1):65-68. doi:10.1046/j.1440-1614.2001.00847.x
12. Alexander RC, Karp BI, Thompson S, et al. A double blind, placebo-controlled trial of demeclocycline treatment of polydipsia-hyponatremia in chronically psychotic patients. Biol Psychiatry. 1991;30(4):417-420. doi:10.1016/0006-3223(91)90300-B
13. Valente S, Fisher D. Recognizing and managing psychogenic polydipsia in mental health. J Nurse Pract. 2010;6(7):546-550. doi:10.1016/j.nurpra.2010.03.004
14. Barton R, Esq SL, Lockett LL. The use of conservatorships and adult guardianships and other options in the care of the mentally ill in the United States. World Guard Congr. Published May 29, 2014. Accessed June 18, 2021. http://www.guardianship.org/IRL/Resources/Handouts/Family%20Members%20as%20Guardians_Handout.pdf
15. ABA Commission on Law & Aging. Adult Guardianship Statutory Table of Authorities. ABA. Published January 2021. Accessed June 17, 2021. https://www.americanbar.org/content/dam/aba/administrative/law_aging/2019-adult-guardianship-statutory-table-of-authorities.pdf
1. Sharp CS, Wilson MP. Hyponatremia. In: Nordstrom KD, Wilson MP, eds. Quick guide to psychiatric emergencies. Springer International Publishing; 2018:115-119. doi:10.1007/978-3-319-58260-3_21
2. Gross P. Clinical management of SIADH. Ther Adv Endocrinol Metab. 2012;3(2):61-73. doi:10.1177/2042018812437561
3. Christ-Crain M, Bichet DG, Fenske WK, et al. Diabetes insipidus. Nat Rev Dis Primer. 2019;5(1):54. doi:10.1038/s41572-019-0103-2
4. Ahmadi L, Goldman MB. Primary polydipsia: update. Best Pract Res Clin Endocrinol Metab. 2020;34(5):101469. doi:10.1016/j.beem.2020.101469
5. Kirino S, Sakuma M, Misawa F, et al. Relationship between polydipsia and antipsychotics: a systematic review of clinical studies and case reports. Prog Neuropsychopharmacol Biol Psychiatry. 2020;96:109756. doi:10.1016/j.pnpbp.2019.109756
6. Siafis S, Tzachanis D, Samara M, et al. Antipsychotic drugs: from receptor-binding profiles to metabolic side effects. Curr Neuropharmacol. 2018;16(8):1210-1223. doi:10.2174/1570159X15666170630163616
7. Seeman P, Tallerico T. Antipsychotic drugs which elicit little or no parkinsonism bind more loosely than dopamine to brain D2 receptors, yet occupy high levels of these receptors. Mol Psychiatry. 1998;3(2):123-134. doi:10.1038/sj.mp.4000336
8. Hayashi T, Nishikawa T, Koga I, et al. Involvement of the α 2 -adrenergic system in polydipsia in schizophrenic patients: a pilot study. Psychopharmacology (Berl). 1997;130(4):382-386. doi:10.1007/s002130050254
9. Rizvi S, Gold J, Khan AM. Role of naltrexone in improving compulsive drinking in psychogenic polydipsia. Cureus. 2019;11(8):e5320. doi:10.7759/cureus.5320
10. Kishi Y, Kurosawa H, Endo S. Is propranolol effective in primary polydipsia? Int J Psychiatry Med. 1998;28(3):315-325. doi:10.2190/QPWL-14H7-HPGG-A29D
11. Kruse D, Pantelis C, Rudd R, et al. Treatment of psychogenic polydipsia: comparison of risperidone and olanzapine, and the effects of an adjunctive angiotensin-II receptor blocking drug (irbesartan). Aust N Z J Psychiatry. 2001;35(1):65-68. doi:10.1046/j.1440-1614.2001.00847.x
12. Alexander RC, Karp BI, Thompson S, et al. A double blind, placebo-controlled trial of demeclocycline treatment of polydipsia-hyponatremia in chronically psychotic patients. Biol Psychiatry. 1991;30(4):417-420. doi:10.1016/0006-3223(91)90300-B
13. Valente S, Fisher D. Recognizing and managing psychogenic polydipsia in mental health. J Nurse Pract. 2010;6(7):546-550. doi:10.1016/j.nurpra.2010.03.004
14. Barton R, Esq SL, Lockett LL. The use of conservatorships and adult guardianships and other options in the care of the mentally ill in the United States. World Guard Congr. Published May 29, 2014. Accessed June 18, 2021. http://www.guardianship.org/IRL/Resources/Handouts/Family%20Members%20as%20Guardians_Handout.pdf
15. ABA Commission on Law & Aging. Adult Guardianship Statutory Table of Authorities. ABA. Published January 2021. Accessed June 17, 2021. https://www.americanbar.org/content/dam/aba/administrative/law_aging/2019-adult-guardianship-statutory-table-of-authorities.pdf
Neurodegenerative nature of schizophrenia makes case for LAIs
Schizophrenia is a complex disease caused by dysfunction in specific brain regions or circuits. In fact, schizophrenia is not a single disease but several hundred different diseases, according to Henry A. Nasrallah, MD, who spoke on the topic at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
The underlying causes of schizophrenia can be either genetic or environmental, but all involve changes in brain development in the fetus or newborn. Psychosis can occur in a range of disorders, including epilepsy, Parkinson’s disease, cerebral tumors, and narcolepsy, to name just a few. Although it starts out as a neurodevelopmental disorder, schizophrenia becomes neurodegenerative after onset, with each new psychotic episode leading to further damage, said Dr. Nasrallah, professor of psychiatry, neurology, and neuroscience at the University of Cincinnati. Further damage leaves patients with greater and greater disability over time, said Dr. Nasrallah at the meeting presented by MedscapeLive. MedscapeLive and this news organization are owned by the same parent company.
The course of illness in some ways resembles the cascading disability associated with strokes. Schizophrenia relapses lead to subcortical atrophy, ventricular enlargement, and further loss of white matter. The accumulating damage is a result of microglia activation, which leads to neuroinflammation and oxidative stress. Mitochondria may also produce insufficient amounts of the antioxidant glutathione.
“The main reason for relapse in schizophrenia is poor adherence to antipsychotic medications, due to anosognosia, memory impairment, avolition, and substance use. It is absolutely necessary to realize that, while oral antipsychotics are effective in the hospital due to enforced compliance by the nursing staff, patients should be switched to long-acting injectable antipsychotics (LAIs) upon discharge from the first episode, which astonishingly is rarely done by 99% of clinicians,” said Dr. Nasrallah in an interview.
That frequent failure leads to further neurodegeneration and increasing disability, which in turn can lead to high rates of homelessness, suicide, and as well as incarceration, because many state hospitals that used to provide medical care for relapsing individuals have been closed down. All of these consequences place great financial and emotional burdens on families and loved ones.
Reconceptualizing the illness
Dr. Nasrallah also advocated that schizophrenia should be classified as a neurologic disorder instead of a psychiatric disorder. He said that the neuropsychiatric mechanisms behind these related diseases support that classification, and neurologic disorders receive much more insurance coverage.
The neuroinflammatory mechanisms underlying schizophrenia suggest that therapies such as omega-3 fatty acids could provide benefit during the prodromal stages of illness. Antioxidants like N-acetyl cysteine could potentially be useful during psychotic episodes, since it boosts levels of glutathione to reduce damaging free radicals. Other approaches could prevent microglia activation, which appears to initiate neurodegeneration.
Another consequence of psychosis is programmed cell death, or apoptosis, in response to reduced levels of neurotropic agents. That could potentially be countered using agents to prevent apoptosis.
Dr. Nasrallah believes clinicians should not use first-generation antipsychotics such as haloperidol, because research has shown that those drugs, while effective, also destroy neurons. Second-generation antipsychotics (SGAs) are safer and avoid that neurotoxicity, and they also have a neuroprotective effect. The SGAs may owe their improved efficacy and safety to the fact that they don’t bind as strongly to dopamine receptors, and they are stronger 5-hydroxytryptamine2A antagonists, according to Dr. Nasrallah. A meta-analysis of 18 studies showed that patients on SGAs maintained gray matter volume, and may even achieve increases in the hippocampus and the prefrontal cortex.
In the Q&A session after the presentation, Dr. Nasrallah was asked whether treatment should be kept up for the rest of the patient’s life, or whether medication should be tapered – and perhaps stopped. He likened treatment of schizophrenia to diabetes or high blood pressure.
“It’s an illness. A lot of medical disorders require lifetime treatment, and there is no difference between psychiatry and the rest of medicine,” he said. “You have to continue the medication at the dose that worked in the acute episode, hopefully the lowest possible dose.”
Dr. Nasrallah did concede that it can be challenging to get patients to accept permanent treatment, and he shared his own strategy to achieve that outcome. “I don’t tell the patient, ‘You’re going to take this the rest of your life.’ It depresses them. So I say, ‘Let’s keep this on board for a year, and I’ll see you regularly, and I’ll monitor you, and we’ll see how it goes, and then we will make another decision at the end of the year.’ ”
During that year, Dr. Nasrallah educates the patient and develops a rapport. “I will show them a lot of data and information about the illness and the hazards of stopping [treatment]. And by the end of the year, most of my patients say: ‘Yeah, I agree. Let’s continue the good thing and let’s not fix something that’s not broken.’ ”
Dr. Nasrallah has consulted for Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Indivior, Intra-Cellular, Janssen, Neurocrine, Otsuka, Sunovion, and Teva. He has also served on a speaker’s bureau for most of those companies, in addition to that of Noven.
Schizophrenia is a complex disease caused by dysfunction in specific brain regions or circuits. In fact, schizophrenia is not a single disease but several hundred different diseases, according to Henry A. Nasrallah, MD, who spoke on the topic at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
The underlying causes of schizophrenia can be either genetic or environmental, but all involve changes in brain development in the fetus or newborn. Psychosis can occur in a range of disorders, including epilepsy, Parkinson’s disease, cerebral tumors, and narcolepsy, to name just a few. Although it starts out as a neurodevelopmental disorder, schizophrenia becomes neurodegenerative after onset, with each new psychotic episode leading to further damage, said Dr. Nasrallah, professor of psychiatry, neurology, and neuroscience at the University of Cincinnati. Further damage leaves patients with greater and greater disability over time, said Dr. Nasrallah at the meeting presented by MedscapeLive. MedscapeLive and this news organization are owned by the same parent company.
The course of illness in some ways resembles the cascading disability associated with strokes. Schizophrenia relapses lead to subcortical atrophy, ventricular enlargement, and further loss of white matter. The accumulating damage is a result of microglia activation, which leads to neuroinflammation and oxidative stress. Mitochondria may also produce insufficient amounts of the antioxidant glutathione.
“The main reason for relapse in schizophrenia is poor adherence to antipsychotic medications, due to anosognosia, memory impairment, avolition, and substance use. It is absolutely necessary to realize that, while oral antipsychotics are effective in the hospital due to enforced compliance by the nursing staff, patients should be switched to long-acting injectable antipsychotics (LAIs) upon discharge from the first episode, which astonishingly is rarely done by 99% of clinicians,” said Dr. Nasrallah in an interview.
That frequent failure leads to further neurodegeneration and increasing disability, which in turn can lead to high rates of homelessness, suicide, and as well as incarceration, because many state hospitals that used to provide medical care for relapsing individuals have been closed down. All of these consequences place great financial and emotional burdens on families and loved ones.
Reconceptualizing the illness
Dr. Nasrallah also advocated that schizophrenia should be classified as a neurologic disorder instead of a psychiatric disorder. He said that the neuropsychiatric mechanisms behind these related diseases support that classification, and neurologic disorders receive much more insurance coverage.
The neuroinflammatory mechanisms underlying schizophrenia suggest that therapies such as omega-3 fatty acids could provide benefit during the prodromal stages of illness. Antioxidants like N-acetyl cysteine could potentially be useful during psychotic episodes, since it boosts levels of glutathione to reduce damaging free radicals. Other approaches could prevent microglia activation, which appears to initiate neurodegeneration.
Another consequence of psychosis is programmed cell death, or apoptosis, in response to reduced levels of neurotropic agents. That could potentially be countered using agents to prevent apoptosis.
Dr. Nasrallah believes clinicians should not use first-generation antipsychotics such as haloperidol, because research has shown that those drugs, while effective, also destroy neurons. Second-generation antipsychotics (SGAs) are safer and avoid that neurotoxicity, and they also have a neuroprotective effect. The SGAs may owe their improved efficacy and safety to the fact that they don’t bind as strongly to dopamine receptors, and they are stronger 5-hydroxytryptamine2A antagonists, according to Dr. Nasrallah. A meta-analysis of 18 studies showed that patients on SGAs maintained gray matter volume, and may even achieve increases in the hippocampus and the prefrontal cortex.
In the Q&A session after the presentation, Dr. Nasrallah was asked whether treatment should be kept up for the rest of the patient’s life, or whether medication should be tapered – and perhaps stopped. He likened treatment of schizophrenia to diabetes or high blood pressure.
“It’s an illness. A lot of medical disorders require lifetime treatment, and there is no difference between psychiatry and the rest of medicine,” he said. “You have to continue the medication at the dose that worked in the acute episode, hopefully the lowest possible dose.”
Dr. Nasrallah did concede that it can be challenging to get patients to accept permanent treatment, and he shared his own strategy to achieve that outcome. “I don’t tell the patient, ‘You’re going to take this the rest of your life.’ It depresses them. So I say, ‘Let’s keep this on board for a year, and I’ll see you regularly, and I’ll monitor you, and we’ll see how it goes, and then we will make another decision at the end of the year.’ ”
During that year, Dr. Nasrallah educates the patient and develops a rapport. “I will show them a lot of data and information about the illness and the hazards of stopping [treatment]. And by the end of the year, most of my patients say: ‘Yeah, I agree. Let’s continue the good thing and let’s not fix something that’s not broken.’ ”
Dr. Nasrallah has consulted for Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Indivior, Intra-Cellular, Janssen, Neurocrine, Otsuka, Sunovion, and Teva. He has also served on a speaker’s bureau for most of those companies, in addition to that of Noven.
Schizophrenia is a complex disease caused by dysfunction in specific brain regions or circuits. In fact, schizophrenia is not a single disease but several hundred different diseases, according to Henry A. Nasrallah, MD, who spoke on the topic at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
The underlying causes of schizophrenia can be either genetic or environmental, but all involve changes in brain development in the fetus or newborn. Psychosis can occur in a range of disorders, including epilepsy, Parkinson’s disease, cerebral tumors, and narcolepsy, to name just a few. Although it starts out as a neurodevelopmental disorder, schizophrenia becomes neurodegenerative after onset, with each new psychotic episode leading to further damage, said Dr. Nasrallah, professor of psychiatry, neurology, and neuroscience at the University of Cincinnati. Further damage leaves patients with greater and greater disability over time, said Dr. Nasrallah at the meeting presented by MedscapeLive. MedscapeLive and this news organization are owned by the same parent company.
The course of illness in some ways resembles the cascading disability associated with strokes. Schizophrenia relapses lead to subcortical atrophy, ventricular enlargement, and further loss of white matter. The accumulating damage is a result of microglia activation, which leads to neuroinflammation and oxidative stress. Mitochondria may also produce insufficient amounts of the antioxidant glutathione.
“The main reason for relapse in schizophrenia is poor adherence to antipsychotic medications, due to anosognosia, memory impairment, avolition, and substance use. It is absolutely necessary to realize that, while oral antipsychotics are effective in the hospital due to enforced compliance by the nursing staff, patients should be switched to long-acting injectable antipsychotics (LAIs) upon discharge from the first episode, which astonishingly is rarely done by 99% of clinicians,” said Dr. Nasrallah in an interview.
That frequent failure leads to further neurodegeneration and increasing disability, which in turn can lead to high rates of homelessness, suicide, and as well as incarceration, because many state hospitals that used to provide medical care for relapsing individuals have been closed down. All of these consequences place great financial and emotional burdens on families and loved ones.
Reconceptualizing the illness
Dr. Nasrallah also advocated that schizophrenia should be classified as a neurologic disorder instead of a psychiatric disorder. He said that the neuropsychiatric mechanisms behind these related diseases support that classification, and neurologic disorders receive much more insurance coverage.
The neuroinflammatory mechanisms underlying schizophrenia suggest that therapies such as omega-3 fatty acids could provide benefit during the prodromal stages of illness. Antioxidants like N-acetyl cysteine could potentially be useful during psychotic episodes, since it boosts levels of glutathione to reduce damaging free radicals. Other approaches could prevent microglia activation, which appears to initiate neurodegeneration.
Another consequence of psychosis is programmed cell death, or apoptosis, in response to reduced levels of neurotropic agents. That could potentially be countered using agents to prevent apoptosis.
Dr. Nasrallah believes clinicians should not use first-generation antipsychotics such as haloperidol, because research has shown that those drugs, while effective, also destroy neurons. Second-generation antipsychotics (SGAs) are safer and avoid that neurotoxicity, and they also have a neuroprotective effect. The SGAs may owe their improved efficacy and safety to the fact that they don’t bind as strongly to dopamine receptors, and they are stronger 5-hydroxytryptamine2A antagonists, according to Dr. Nasrallah. A meta-analysis of 18 studies showed that patients on SGAs maintained gray matter volume, and may even achieve increases in the hippocampus and the prefrontal cortex.
In the Q&A session after the presentation, Dr. Nasrallah was asked whether treatment should be kept up for the rest of the patient’s life, or whether medication should be tapered – and perhaps stopped. He likened treatment of schizophrenia to diabetes or high blood pressure.
“It’s an illness. A lot of medical disorders require lifetime treatment, and there is no difference between psychiatry and the rest of medicine,” he said. “You have to continue the medication at the dose that worked in the acute episode, hopefully the lowest possible dose.”
Dr. Nasrallah did concede that it can be challenging to get patients to accept permanent treatment, and he shared his own strategy to achieve that outcome. “I don’t tell the patient, ‘You’re going to take this the rest of your life.’ It depresses them. So I say, ‘Let’s keep this on board for a year, and I’ll see you regularly, and I’ll monitor you, and we’ll see how it goes, and then we will make another decision at the end of the year.’ ”
During that year, Dr. Nasrallah educates the patient and develops a rapport. “I will show them a lot of data and information about the illness and the hazards of stopping [treatment]. And by the end of the year, most of my patients say: ‘Yeah, I agree. Let’s continue the good thing and let’s not fix something that’s not broken.’ ”
Dr. Nasrallah has consulted for Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Indivior, Intra-Cellular, Janssen, Neurocrine, Otsuka, Sunovion, and Teva. He has also served on a speaker’s bureau for most of those companies, in addition to that of Noven.
REPORTING FROM FOCUS ON NEUROPSYCHIATRY 2021
Nonmotor symptoms common in Parkinson’s
The hallmark of Parkinson’s disease is the accompanying motor symptoms, but the condition can bring other challenges. Among those are nonmotor symptoms, including depression, dementia, and even psychosis.
The culprit is Lewy bodies, which are also responsible for Lewy body dementia. “What we call Lewy body dementia and Parkinson’s disease are caused by the same pathological process – the formation of Lewy bodies in the brain,” Leslie Citrome, MD, MPH, said in an interview. Dr. Citrome discussed some of the psychiatric comorbidities associated with Parkinson’s disease at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
In fact, the association goes both ways. “Many people with Parkinson’s disease develop a dementia. Many people with Lewy body dementia develop motor symptoms that look just like Parkinson’s disease,” said Dr. Citrome, professor of psychiatry and behavioral sciences at New York Medical College, Valhalla, and president of the American Society for Clinical Psychopharmacology.
The motor symptoms of Parkinson’s disease are generally attributable to loss of striatal dopaminergic neurons, while nonmotor symptoms can be traced to loss of neurons in nondopaminergic regions. Nonmotor symptoms – often including sleep disorders, depression, cognitive changes, and psychosis – may occur before motor symptoms. Other problems may include autonomic dysfunction, such as constipation, sexual dysfunction, sweating, or urinary retention.
Patients might not be aware that nonmotor symptoms can occur with Parkinson’s disease and may not even consider mentioning mood changes or hallucinations to their neurologist. Family members may also be unaware.
Sleep problems are common in Parkinson’s disease, including rapid eye-movement sleep behavior disorders, vivid dreams, restless legs syndrome, insomnia, and daytime somnolence. Dopamine agonists may also cause unintended sleep.
Depression is extremely common, affecting up to 90% of Parkinson’s disease patients, and this may be related to dopaminergic losses. Antidepressant medications can worsen Parkinson’s disease symptoms: Tricyclic antidepressants increase risk of adverse events from anticholinergic drugs. Selective serotonin reuptake inhibitors (SSRIs) can exacerbate tremor and may increase risk of serotonin syndrome when combined with MAO‐B inhibitors.
Dr. Citrome was not aware of any antidepressant drugs that have been tested specifically in Parkinson’s disease patients, though “I’d be surprised if there wasn’t,” he said during the Q&A session. “There’s no one perfect antidepressant for people with depression associated with Parkinson’s disease. I would make sure to select one that they would tolerate and be willing to take and that doesn’t interfere with their treatment of their movement disorder, and (I would make sure) that there’s no drug-drug interaction,” he said.
This can include reduced working memory, learning, and planning, and generally does not manifest until at least 1 year after motor symptoms have begun. Rivastigmine is Food and Drug Administration–approved for treatment of cognitive impairment in Parkinson’s disease.
As many as 60% of Parkinson’s disease patients suffer from psychosis at some point, often visual hallucinations or delusions, which can include beliefs of spousal infidelity.
Many clinicians prescribe quetiapine off label, but there are not compelling data to support that it reduces intensity and frequency of hallucinations and delusions, according to Dr. Citrome. However, it is relatively easy to prescribe, requiring no preauthorizations, it is inexpensive, and it may improve sleep.
The FDA approved pimavanserin in 2016 for hallucinations and delusions in Parkinson’s disease, and it doesn’t worsen motor symptoms, Dr. Citrome said. That’s because pimavanserin is a highly selective antagonist of the 5-HT2A receptor, with no effect on dopaminergic, histaminergic, adrenergic, or muscarinic receptors.
The drug improves positive symptoms beginning at days 29 and 43, compared with placebo. An analysis by Dr. Citrome’s group found a number needed to treat (NNT) of 7 to gain a benefit over placebo if the metric is a ≥ 30% reduction in baseline symptom score. The drug had an NNT of 9 to achieve a ≥ 50% reduction, and an NNT of 5 to achieve a score of much improved or very much improved on the Clinical Global Impression–Improvement (CGI-I) scale. In general, an NNT less than 10 suggests that a drug is clinically useful.
In contrast, the number needed to harm (NNH) represents the number of patients who would need to receive a therapy to add one adverse event, compared with placebo. A number greater than 10 indicates that the therapy may be tolerable.
Using various measures, the NNH was well over 10 for pimavanserin. With respect to somnolence, the NNH over placebo was 138, and for a weight gain of 7% or more, the NNH was 594.
Overall, the study found that 4 patients would need to be treated to achieve a benefit over placebo with respect to a ≥ 3–point improvement in the Scale of Positive Symptoms–Parkinson’s Disease (SAPS-PD), while 21 would need to receive the drug to lead to one additional discontinuation because of an adverse event, compared to placebo.
When researchers compared pimavanserin to off-label use of quetiapine, olanzapine, and clozapine, they found a Cohen’s d value of 0.50, which was better than quetiapine and olanzapine, but lower than for clozapine. However, there is no requirement of blood monitoring, and clozapine can potentially worsen motor symptoms.
Dr. Citrome’s presentation should be a reminder to neurologists that psychiatric disorders are an important patient concern, said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience at the University of Cincinnati, who moderated the session.
“I think this serves as a model to recognize that many neurological disorders actually present with numerous psychiatric disorders,” Dr. Nasrallah said during the meeting, presented by MedscapeLive. MedscapeLive and this news organization are owned by the same parent company.
Dr. Citrome has consulted for AbbVie, Acadia, Alkermes, Allergan, Angelini, Astellas, Avanir, Axsome, BioXcel, Boehringer-Ingelheim, Cadent Therapeutics, Eisai, Impel, Intra-Cellular, Janssen, Karuna, Lundbeck, Lyndra, MedAvante-ProPhase, Merck, Neurocrine, Noven, Otsuka, Ovid, Relmada, Sage, Sunovion, and Teva. He has been a speaker for most of those companies, and he holds stock in Bristol Myers Squibb, Eli Lilly, J&J, Merck, and Pfizer.
Dr. Nasrallah has consulted for Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Indivior, Intra-Cellular, Janssen, Neurocrine, Otsuka, Sunovion, and Teva. He has served on a speakers bureau for most of those companies, in addition to that of Noven.
The hallmark of Parkinson’s disease is the accompanying motor symptoms, but the condition can bring other challenges. Among those are nonmotor symptoms, including depression, dementia, and even psychosis.
The culprit is Lewy bodies, which are also responsible for Lewy body dementia. “What we call Lewy body dementia and Parkinson’s disease are caused by the same pathological process – the formation of Lewy bodies in the brain,” Leslie Citrome, MD, MPH, said in an interview. Dr. Citrome discussed some of the psychiatric comorbidities associated with Parkinson’s disease at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
In fact, the association goes both ways. “Many people with Parkinson’s disease develop a dementia. Many people with Lewy body dementia develop motor symptoms that look just like Parkinson’s disease,” said Dr. Citrome, professor of psychiatry and behavioral sciences at New York Medical College, Valhalla, and president of the American Society for Clinical Psychopharmacology.
The motor symptoms of Parkinson’s disease are generally attributable to loss of striatal dopaminergic neurons, while nonmotor symptoms can be traced to loss of neurons in nondopaminergic regions. Nonmotor symptoms – often including sleep disorders, depression, cognitive changes, and psychosis – may occur before motor symptoms. Other problems may include autonomic dysfunction, such as constipation, sexual dysfunction, sweating, or urinary retention.
Patients might not be aware that nonmotor symptoms can occur with Parkinson’s disease and may not even consider mentioning mood changes or hallucinations to their neurologist. Family members may also be unaware.
Sleep problems are common in Parkinson’s disease, including rapid eye-movement sleep behavior disorders, vivid dreams, restless legs syndrome, insomnia, and daytime somnolence. Dopamine agonists may also cause unintended sleep.
Depression is extremely common, affecting up to 90% of Parkinson’s disease patients, and this may be related to dopaminergic losses. Antidepressant medications can worsen Parkinson’s disease symptoms: Tricyclic antidepressants increase risk of adverse events from anticholinergic drugs. Selective serotonin reuptake inhibitors (SSRIs) can exacerbate tremor and may increase risk of serotonin syndrome when combined with MAO‐B inhibitors.
Dr. Citrome was not aware of any antidepressant drugs that have been tested specifically in Parkinson’s disease patients, though “I’d be surprised if there wasn’t,” he said during the Q&A session. “There’s no one perfect antidepressant for people with depression associated with Parkinson’s disease. I would make sure to select one that they would tolerate and be willing to take and that doesn’t interfere with their treatment of their movement disorder, and (I would make sure) that there’s no drug-drug interaction,” he said.
This can include reduced working memory, learning, and planning, and generally does not manifest until at least 1 year after motor symptoms have begun. Rivastigmine is Food and Drug Administration–approved for treatment of cognitive impairment in Parkinson’s disease.
As many as 60% of Parkinson’s disease patients suffer from psychosis at some point, often visual hallucinations or delusions, which can include beliefs of spousal infidelity.
Many clinicians prescribe quetiapine off label, but there are not compelling data to support that it reduces intensity and frequency of hallucinations and delusions, according to Dr. Citrome. However, it is relatively easy to prescribe, requiring no preauthorizations, it is inexpensive, and it may improve sleep.
The FDA approved pimavanserin in 2016 for hallucinations and delusions in Parkinson’s disease, and it doesn’t worsen motor symptoms, Dr. Citrome said. That’s because pimavanserin is a highly selective antagonist of the 5-HT2A receptor, with no effect on dopaminergic, histaminergic, adrenergic, or muscarinic receptors.
The drug improves positive symptoms beginning at days 29 and 43, compared with placebo. An analysis by Dr. Citrome’s group found a number needed to treat (NNT) of 7 to gain a benefit over placebo if the metric is a ≥ 30% reduction in baseline symptom score. The drug had an NNT of 9 to achieve a ≥ 50% reduction, and an NNT of 5 to achieve a score of much improved or very much improved on the Clinical Global Impression–Improvement (CGI-I) scale. In general, an NNT less than 10 suggests that a drug is clinically useful.
In contrast, the number needed to harm (NNH) represents the number of patients who would need to receive a therapy to add one adverse event, compared with placebo. A number greater than 10 indicates that the therapy may be tolerable.
Using various measures, the NNH was well over 10 for pimavanserin. With respect to somnolence, the NNH over placebo was 138, and for a weight gain of 7% or more, the NNH was 594.
Overall, the study found that 4 patients would need to be treated to achieve a benefit over placebo with respect to a ≥ 3–point improvement in the Scale of Positive Symptoms–Parkinson’s Disease (SAPS-PD), while 21 would need to receive the drug to lead to one additional discontinuation because of an adverse event, compared to placebo.
When researchers compared pimavanserin to off-label use of quetiapine, olanzapine, and clozapine, they found a Cohen’s d value of 0.50, which was better than quetiapine and olanzapine, but lower than for clozapine. However, there is no requirement of blood monitoring, and clozapine can potentially worsen motor symptoms.
Dr. Citrome’s presentation should be a reminder to neurologists that psychiatric disorders are an important patient concern, said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience at the University of Cincinnati, who moderated the session.
“I think this serves as a model to recognize that many neurological disorders actually present with numerous psychiatric disorders,” Dr. Nasrallah said during the meeting, presented by MedscapeLive. MedscapeLive and this news organization are owned by the same parent company.
Dr. Citrome has consulted for AbbVie, Acadia, Alkermes, Allergan, Angelini, Astellas, Avanir, Axsome, BioXcel, Boehringer-Ingelheim, Cadent Therapeutics, Eisai, Impel, Intra-Cellular, Janssen, Karuna, Lundbeck, Lyndra, MedAvante-ProPhase, Merck, Neurocrine, Noven, Otsuka, Ovid, Relmada, Sage, Sunovion, and Teva. He has been a speaker for most of those companies, and he holds stock in Bristol Myers Squibb, Eli Lilly, J&J, Merck, and Pfizer.
Dr. Nasrallah has consulted for Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Indivior, Intra-Cellular, Janssen, Neurocrine, Otsuka, Sunovion, and Teva. He has served on a speakers bureau for most of those companies, in addition to that of Noven.
The hallmark of Parkinson’s disease is the accompanying motor symptoms, but the condition can bring other challenges. Among those are nonmotor symptoms, including depression, dementia, and even psychosis.
The culprit is Lewy bodies, which are also responsible for Lewy body dementia. “What we call Lewy body dementia and Parkinson’s disease are caused by the same pathological process – the formation of Lewy bodies in the brain,” Leslie Citrome, MD, MPH, said in an interview. Dr. Citrome discussed some of the psychiatric comorbidities associated with Parkinson’s disease at a virtual meeting presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
In fact, the association goes both ways. “Many people with Parkinson’s disease develop a dementia. Many people with Lewy body dementia develop motor symptoms that look just like Parkinson’s disease,” said Dr. Citrome, professor of psychiatry and behavioral sciences at New York Medical College, Valhalla, and president of the American Society for Clinical Psychopharmacology.
The motor symptoms of Parkinson’s disease are generally attributable to loss of striatal dopaminergic neurons, while nonmotor symptoms can be traced to loss of neurons in nondopaminergic regions. Nonmotor symptoms – often including sleep disorders, depression, cognitive changes, and psychosis – may occur before motor symptoms. Other problems may include autonomic dysfunction, such as constipation, sexual dysfunction, sweating, or urinary retention.
Patients might not be aware that nonmotor symptoms can occur with Parkinson’s disease and may not even consider mentioning mood changes or hallucinations to their neurologist. Family members may also be unaware.
Sleep problems are common in Parkinson’s disease, including rapid eye-movement sleep behavior disorders, vivid dreams, restless legs syndrome, insomnia, and daytime somnolence. Dopamine agonists may also cause unintended sleep.
Depression is extremely common, affecting up to 90% of Parkinson’s disease patients, and this may be related to dopaminergic losses. Antidepressant medications can worsen Parkinson’s disease symptoms: Tricyclic antidepressants increase risk of adverse events from anticholinergic drugs. Selective serotonin reuptake inhibitors (SSRIs) can exacerbate tremor and may increase risk of serotonin syndrome when combined with MAO‐B inhibitors.
Dr. Citrome was not aware of any antidepressant drugs that have been tested specifically in Parkinson’s disease patients, though “I’d be surprised if there wasn’t,” he said during the Q&A session. “There’s no one perfect antidepressant for people with depression associated with Parkinson’s disease. I would make sure to select one that they would tolerate and be willing to take and that doesn’t interfere with their treatment of their movement disorder, and (I would make sure) that there’s no drug-drug interaction,” he said.
This can include reduced working memory, learning, and planning, and generally does not manifest until at least 1 year after motor symptoms have begun. Rivastigmine is Food and Drug Administration–approved for treatment of cognitive impairment in Parkinson’s disease.
As many as 60% of Parkinson’s disease patients suffer from psychosis at some point, often visual hallucinations or delusions, which can include beliefs of spousal infidelity.
Many clinicians prescribe quetiapine off label, but there are not compelling data to support that it reduces intensity and frequency of hallucinations and delusions, according to Dr. Citrome. However, it is relatively easy to prescribe, requiring no preauthorizations, it is inexpensive, and it may improve sleep.
The FDA approved pimavanserin in 2016 for hallucinations and delusions in Parkinson’s disease, and it doesn’t worsen motor symptoms, Dr. Citrome said. That’s because pimavanserin is a highly selective antagonist of the 5-HT2A receptor, with no effect on dopaminergic, histaminergic, adrenergic, or muscarinic receptors.
The drug improves positive symptoms beginning at days 29 and 43, compared with placebo. An analysis by Dr. Citrome’s group found a number needed to treat (NNT) of 7 to gain a benefit over placebo if the metric is a ≥ 30% reduction in baseline symptom score. The drug had an NNT of 9 to achieve a ≥ 50% reduction, and an NNT of 5 to achieve a score of much improved or very much improved on the Clinical Global Impression–Improvement (CGI-I) scale. In general, an NNT less than 10 suggests that a drug is clinically useful.
In contrast, the number needed to harm (NNH) represents the number of patients who would need to receive a therapy to add one adverse event, compared with placebo. A number greater than 10 indicates that the therapy may be tolerable.
Using various measures, the NNH was well over 10 for pimavanserin. With respect to somnolence, the NNH over placebo was 138, and for a weight gain of 7% or more, the NNH was 594.
Overall, the study found that 4 patients would need to be treated to achieve a benefit over placebo with respect to a ≥ 3–point improvement in the Scale of Positive Symptoms–Parkinson’s Disease (SAPS-PD), while 21 would need to receive the drug to lead to one additional discontinuation because of an adverse event, compared to placebo.
When researchers compared pimavanserin to off-label use of quetiapine, olanzapine, and clozapine, they found a Cohen’s d value of 0.50, which was better than quetiapine and olanzapine, but lower than for clozapine. However, there is no requirement of blood monitoring, and clozapine can potentially worsen motor symptoms.
Dr. Citrome’s presentation should be a reminder to neurologists that psychiatric disorders are an important patient concern, said Henry A. Nasrallah, MD, professor of psychiatry, neurology, and neuroscience at the University of Cincinnati, who moderated the session.
“I think this serves as a model to recognize that many neurological disorders actually present with numerous psychiatric disorders,” Dr. Nasrallah said during the meeting, presented by MedscapeLive. MedscapeLive and this news organization are owned by the same parent company.
Dr. Citrome has consulted for AbbVie, Acadia, Alkermes, Allergan, Angelini, Astellas, Avanir, Axsome, BioXcel, Boehringer-Ingelheim, Cadent Therapeutics, Eisai, Impel, Intra-Cellular, Janssen, Karuna, Lundbeck, Lyndra, MedAvante-ProPhase, Merck, Neurocrine, Noven, Otsuka, Ovid, Relmada, Sage, Sunovion, and Teva. He has been a speaker for most of those companies, and he holds stock in Bristol Myers Squibb, Eli Lilly, J&J, Merck, and Pfizer.
Dr. Nasrallah has consulted for Acadia, Alkermes, Allergan, Boehringer-Ingelheim, Indivior, Intra-Cellular, Janssen, Neurocrine, Otsuka, Sunovion, and Teva. He has served on a speakers bureau for most of those companies, in addition to that of Noven.
FROM FOCUS ON NEUROPSYCHIATRY 2021
Psychotic features among older adults tied to Parkinson’s
Adults aged 65 years and older who develop psychotic manifestations are significantly more likely than those without such manifestations to develop prodromal Parkinson’s disease, data from 925 individuals suggest.
“The presence of perceptual abnormalities and/or delusional ideation among community-dwelling elderly individuals is more widespread than considered in the past,” wrote Ioanna Pachi, MD, of National and Kapodistrian University of Athens Medical School and colleagues. However, those psychoses and their potential impact on prodromal Parkinson’s disease (PD) have not been well studied in community-dwelling populations, they noted in the study, published in Parkinsonism and Related Disorders.
In the study, Dr. Pachi and colleagues reviewed data from 914 participants in the Hellenic Longitudinal Investigation of Aging and Diet study (HELIAD), a cross-sectional, population-based cohort study of older adults in Greece. The average age of the participants was 76 years, and 41% were men. Participants had no delusional features at baseline; delusional features were assessed using the Neuropsychiatric Inventory scale and the Columbia University Scale for Psychopathology in Alzheimer’s disease. The researchers calculated the probability of prodromal PD (pPD) for each participant based on the 2019 International Parkinson and Movement Disorders Society research criteria for prodromal PD.
Over a 3-year follow-up period, 20 participants developed psychotic manifestations and were 1.3 times more likely to have pPD, compared with those without psychoses (P = .006). Those with new-onset psychotic features were categorized together as the NPSY group, regardless of symptom severity or frequency; those with no symptoms at either baseline or during follow-up were categorized as unaffected (UPSY). Most of the NPSY participants showed isolated delusional features, although some expressed hallucinations. Most symptoms were mild.
New-onset psychosis was associated with a fivefold increased risk of both subthreshold parkinsonism and depression (adjusted odds ratios, 4.5 and 5.0, respectively) and with a threefold increased risk of constipation (aOR 2.6). Other factors, including nonsmoking, global cognitive deficit, and anxiety were not significantly associated with new-onset psychotic symptoms after adjusting for confounding factors.
Although the mechanism behind the association remains unclear,
The study findings were limited by several factors, including the administration of neuropsychiatric questionnaires by nonpsychiatrists, and lack of detailed psychiatric history, including complete information on medication use, the researchers noted. The small size of the NPSY group also prevented evaluation of the potential associations between pPD and different modalities of hallucinations, they said.
However, the results were strengthened by the overall large and population-based sample size, and the comprehensive evaluation of psychotic features, they wrote. More follow-up evaluations in the HELIAD cohort are planned to further explore the underlying mechanism of the association between late-life psychosis and pPD.
“Provided that these results are confirmed in other community cohorts of elderly subjects, psychotic features may be added to the list of manifestations of pPD,” they concluded.
The study was supported in part by grants from the Alzheimer’s Association, ARISTEIA, and the ESPA-EU program Excellence Grant. It was cofunded by the European Social Fund and Greek National resources, the Ministry for Health and Social Solidarity, Greece, and the Greek State Scholarships Foundation. Dr. Pachi had no disclosures.
Adults aged 65 years and older who develop psychotic manifestations are significantly more likely than those without such manifestations to develop prodromal Parkinson’s disease, data from 925 individuals suggest.
“The presence of perceptual abnormalities and/or delusional ideation among community-dwelling elderly individuals is more widespread than considered in the past,” wrote Ioanna Pachi, MD, of National and Kapodistrian University of Athens Medical School and colleagues. However, those psychoses and their potential impact on prodromal Parkinson’s disease (PD) have not been well studied in community-dwelling populations, they noted in the study, published in Parkinsonism and Related Disorders.
In the study, Dr. Pachi and colleagues reviewed data from 914 participants in the Hellenic Longitudinal Investigation of Aging and Diet study (HELIAD), a cross-sectional, population-based cohort study of older adults in Greece. The average age of the participants was 76 years, and 41% were men. Participants had no delusional features at baseline; delusional features were assessed using the Neuropsychiatric Inventory scale and the Columbia University Scale for Psychopathology in Alzheimer’s disease. The researchers calculated the probability of prodromal PD (pPD) for each participant based on the 2019 International Parkinson and Movement Disorders Society research criteria for prodromal PD.
Over a 3-year follow-up period, 20 participants developed psychotic manifestations and were 1.3 times more likely to have pPD, compared with those without psychoses (P = .006). Those with new-onset psychotic features were categorized together as the NPSY group, regardless of symptom severity or frequency; those with no symptoms at either baseline or during follow-up were categorized as unaffected (UPSY). Most of the NPSY participants showed isolated delusional features, although some expressed hallucinations. Most symptoms were mild.
New-onset psychosis was associated with a fivefold increased risk of both subthreshold parkinsonism and depression (adjusted odds ratios, 4.5 and 5.0, respectively) and with a threefold increased risk of constipation (aOR 2.6). Other factors, including nonsmoking, global cognitive deficit, and anxiety were not significantly associated with new-onset psychotic symptoms after adjusting for confounding factors.
Although the mechanism behind the association remains unclear,
The study findings were limited by several factors, including the administration of neuropsychiatric questionnaires by nonpsychiatrists, and lack of detailed psychiatric history, including complete information on medication use, the researchers noted. The small size of the NPSY group also prevented evaluation of the potential associations between pPD and different modalities of hallucinations, they said.
However, the results were strengthened by the overall large and population-based sample size, and the comprehensive evaluation of psychotic features, they wrote. More follow-up evaluations in the HELIAD cohort are planned to further explore the underlying mechanism of the association between late-life psychosis and pPD.
“Provided that these results are confirmed in other community cohorts of elderly subjects, psychotic features may be added to the list of manifestations of pPD,” they concluded.
The study was supported in part by grants from the Alzheimer’s Association, ARISTEIA, and the ESPA-EU program Excellence Grant. It was cofunded by the European Social Fund and Greek National resources, the Ministry for Health and Social Solidarity, Greece, and the Greek State Scholarships Foundation. Dr. Pachi had no disclosures.
Adults aged 65 years and older who develop psychotic manifestations are significantly more likely than those without such manifestations to develop prodromal Parkinson’s disease, data from 925 individuals suggest.
“The presence of perceptual abnormalities and/or delusional ideation among community-dwelling elderly individuals is more widespread than considered in the past,” wrote Ioanna Pachi, MD, of National and Kapodistrian University of Athens Medical School and colleagues. However, those psychoses and their potential impact on prodromal Parkinson’s disease (PD) have not been well studied in community-dwelling populations, they noted in the study, published in Parkinsonism and Related Disorders.
In the study, Dr. Pachi and colleagues reviewed data from 914 participants in the Hellenic Longitudinal Investigation of Aging and Diet study (HELIAD), a cross-sectional, population-based cohort study of older adults in Greece. The average age of the participants was 76 years, and 41% were men. Participants had no delusional features at baseline; delusional features were assessed using the Neuropsychiatric Inventory scale and the Columbia University Scale for Psychopathology in Alzheimer’s disease. The researchers calculated the probability of prodromal PD (pPD) for each participant based on the 2019 International Parkinson and Movement Disorders Society research criteria for prodromal PD.
Over a 3-year follow-up period, 20 participants developed psychotic manifestations and were 1.3 times more likely to have pPD, compared with those without psychoses (P = .006). Those with new-onset psychotic features were categorized together as the NPSY group, regardless of symptom severity or frequency; those with no symptoms at either baseline or during follow-up were categorized as unaffected (UPSY). Most of the NPSY participants showed isolated delusional features, although some expressed hallucinations. Most symptoms were mild.
New-onset psychosis was associated with a fivefold increased risk of both subthreshold parkinsonism and depression (adjusted odds ratios, 4.5 and 5.0, respectively) and with a threefold increased risk of constipation (aOR 2.6). Other factors, including nonsmoking, global cognitive deficit, and anxiety were not significantly associated with new-onset psychotic symptoms after adjusting for confounding factors.
Although the mechanism behind the association remains unclear,
The study findings were limited by several factors, including the administration of neuropsychiatric questionnaires by nonpsychiatrists, and lack of detailed psychiatric history, including complete information on medication use, the researchers noted. The small size of the NPSY group also prevented evaluation of the potential associations between pPD and different modalities of hallucinations, they said.
However, the results were strengthened by the overall large and population-based sample size, and the comprehensive evaluation of psychotic features, they wrote. More follow-up evaluations in the HELIAD cohort are planned to further explore the underlying mechanism of the association between late-life psychosis and pPD.
“Provided that these results are confirmed in other community cohorts of elderly subjects, psychotic features may be added to the list of manifestations of pPD,” they concluded.
The study was supported in part by grants from the Alzheimer’s Association, ARISTEIA, and the ESPA-EU program Excellence Grant. It was cofunded by the European Social Fund and Greek National resources, the Ministry for Health and Social Solidarity, Greece, and the Greek State Scholarships Foundation. Dr. Pachi had no disclosures.
FROM PARKINSONISM AND RELATED DISORDERS
Toward a clearer risk model for postpartum psychosis
Postpartum depression, in many respects, has become a household term. Over the last decade, there has been increasing awareness of the importance of screening for postpartum depression, with increased systematic screening across clinical settings where care is delivered to women during pregnancy and the postpartum period. There have also been greater efforts to identify women who are suffering postpartum depression and to support them with appropriate clinical interventions, whether through psychotherapy and/or pharmacologic therapy. Clinical interventions are supplemented by the increasing awareness of the value of community-based support groups for women who are suffering from postpartum mood and anxiety disorders.
Despite the growing, appropriate focus on recognition and acute treatment of postpartum depression as well as assessing clinical outcomes for those who suffer from postpartum mood and anxiety disorders, less attention has been given to postpartum psychosis, the most severe form of postpartum depression. It is indeed, the least common postpartum psychiatric syndrome (1 in 1,000 births) and comes to public attention when there has been a tragedy during the acute postpartum period such as maternal suicide or infanticide. In this sense, postpartum psychosis is ironically an underappreciated clinical entity across America given its severity, the effect it has on longer-term psychiatric morbidity, and its effect on children and families.
Our group at the Center for Women’s Mental Health has been interested in postpartum psychosis for years and started the Postpartum Psychosis Project in an effort to better understand the phenomenology, course, treatment, outcome, and genomic underpinnings of postpartum psychosis. Risk factors that are well established for postpartum psychosis have been described and overwhelmingly include patients with bipolar disorder. The risk for recurrent postpartum psychosis in women who have had a previous episode is as great as 75%-90% in the absence of prophylactic intervention. With that said, we are extremely interested in understanding the etiology of postpartum psychosis. Various studies over the last 5 years have looked at a whole host of psychosocial as well as neurobiologic variables that may contribute to risk for postpartum psychosis, including dysregulation of the stress axis, heightened inflammation as well as a history of child adversity and heightened experience of stress during the perinatal period.
There have also been anecdotal reports during Virtual Rounds at the Center for Women’s Mental Health of higher recent rates of postpartum psychosis manifesting during the postpartum period. This is a clinical observation and has not been systematically studied. However, one can wonder whether the experience of the pandemic has constituted a stressor for at-risk women, tipping the scales toward women becoming ill, or whether clinicians are seeing this finding more because of our ability to observe it more within the context of the pandemic.
Precise quantification of risk for postpartum psychosis is complicated; as noted, women with bipolar disorder have a predictably high risk for getting ill during the postpartum period and many go on to have clinical courses consistent with recurrent bipolar disorder. However, there are other women who have circumscribed episodes of psychotic illness in the postpartum period who recover and are totally well without any evidence of psychiatric disorder until they have another child, at which time the risk for recurrence of postpartum psychosis is very high. Interest in developing a model of risk that could reliably predict an illness as serious as postpartum psychosis is on the minds of researchers around the world.
One recent study that highlights the multiple factors involved in risk of postpartum psychosis involved a prospective longitudinal study of a group of women who were followed across the peripartum period from the third trimester until 4 weeks postpartum. In this group, 51 women were at increased risk for postpartum psychosis based on their diagnosis of bipolar disorder, schizoaffective disorder, or a previous episode of postpartum psychosis. These women were matched with a control group with no past or current diagnosis of psychiatric disorder or family history of postpartum psychosis. The findings suggested that women at risk for postpartum psychosis who experienced a psychiatric relapse during the first 4 weeks postpartum relative to women at risk who remained well had histories of severe childhood adversity as well as biomarkers consistent with a dysregulated stress axis (a statistically higher daily cortisol level). This is consistent with other data that have implicated the complex role between psychosocial variables as well as neurobiologic variables, such as a dysregulation in the hypothalamic pituitary adrenal axis and other studies that suggest that dysregulated inflammatory status may also drive risk for postpartum psychosis (Hazelgrove K et al. Psychoneuroendocrinology. 2021 Jun. doi: 10.1016/j.psyneuen.2021.105218).
At the end of the day, postpartum psychosis is a psychiatric and obstetrical emergency. In our center, it is rare for women not to be hospitalized with this condition to ensure the safety of the mother as well as her newborn, and to also get her recompensated and functioning as quickly and as significantly as possible. However, an interesting extrapolation of the findings noted by Hazelgrove and colleagues is that it raises the question of what effective treatments might be used to mitigate risk for those at greatest risk for postpartum psychosis. For example, are there other treatments over and above the few effective ones that have been studied as prophylactic pharmacologic interventions that might mitigate risk for recurrence of an illness as serious as postpartum psychosis?
The data suggesting dysregulation of the stress axis as a predictor variable for risk in women vulnerable to postpartum psychosis opens an array of opportunities that are nonpharmacologic, such as mindfulness-based cognitive therapy or other interventions that help to modulate the stress axis. This is a terrific opportunity to have pharmacologic intervention meet nonpharmacologic intervention to potentially mitigate risk for postpartum psychosis with its attendant serious sequelae.
In our own work, where we are evaluating genomic data in an extremely well-characterized group of women with known histories of postpartum psychosis, we are interested to see if we can enhance understanding of the model of risk for postpartum psychosis by factoring in genomic underpinning, history of diagnosis, and psychosocial variables to optimally craft interventions for this population of at-risk women. This brings us one step closer to the future in women’s mental health, to the practice of “precision reproductive psychiatry,” matching interventions to specific presentations across perinatal populations.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Postpartum depression, in many respects, has become a household term. Over the last decade, there has been increasing awareness of the importance of screening for postpartum depression, with increased systematic screening across clinical settings where care is delivered to women during pregnancy and the postpartum period. There have also been greater efforts to identify women who are suffering postpartum depression and to support them with appropriate clinical interventions, whether through psychotherapy and/or pharmacologic therapy. Clinical interventions are supplemented by the increasing awareness of the value of community-based support groups for women who are suffering from postpartum mood and anxiety disorders.
Despite the growing, appropriate focus on recognition and acute treatment of postpartum depression as well as assessing clinical outcomes for those who suffer from postpartum mood and anxiety disorders, less attention has been given to postpartum psychosis, the most severe form of postpartum depression. It is indeed, the least common postpartum psychiatric syndrome (1 in 1,000 births) and comes to public attention when there has been a tragedy during the acute postpartum period such as maternal suicide or infanticide. In this sense, postpartum psychosis is ironically an underappreciated clinical entity across America given its severity, the effect it has on longer-term psychiatric morbidity, and its effect on children and families.
Our group at the Center for Women’s Mental Health has been interested in postpartum psychosis for years and started the Postpartum Psychosis Project in an effort to better understand the phenomenology, course, treatment, outcome, and genomic underpinnings of postpartum psychosis. Risk factors that are well established for postpartum psychosis have been described and overwhelmingly include patients with bipolar disorder. The risk for recurrent postpartum psychosis in women who have had a previous episode is as great as 75%-90% in the absence of prophylactic intervention. With that said, we are extremely interested in understanding the etiology of postpartum psychosis. Various studies over the last 5 years have looked at a whole host of psychosocial as well as neurobiologic variables that may contribute to risk for postpartum psychosis, including dysregulation of the stress axis, heightened inflammation as well as a history of child adversity and heightened experience of stress during the perinatal period.
There have also been anecdotal reports during Virtual Rounds at the Center for Women’s Mental Health of higher recent rates of postpartum psychosis manifesting during the postpartum period. This is a clinical observation and has not been systematically studied. However, one can wonder whether the experience of the pandemic has constituted a stressor for at-risk women, tipping the scales toward women becoming ill, or whether clinicians are seeing this finding more because of our ability to observe it more within the context of the pandemic.
Precise quantification of risk for postpartum psychosis is complicated; as noted, women with bipolar disorder have a predictably high risk for getting ill during the postpartum period and many go on to have clinical courses consistent with recurrent bipolar disorder. However, there are other women who have circumscribed episodes of psychotic illness in the postpartum period who recover and are totally well without any evidence of psychiatric disorder until they have another child, at which time the risk for recurrence of postpartum psychosis is very high. Interest in developing a model of risk that could reliably predict an illness as serious as postpartum psychosis is on the minds of researchers around the world.
One recent study that highlights the multiple factors involved in risk of postpartum psychosis involved a prospective longitudinal study of a group of women who were followed across the peripartum period from the third trimester until 4 weeks postpartum. In this group, 51 women were at increased risk for postpartum psychosis based on their diagnosis of bipolar disorder, schizoaffective disorder, or a previous episode of postpartum psychosis. These women were matched with a control group with no past or current diagnosis of psychiatric disorder or family history of postpartum psychosis. The findings suggested that women at risk for postpartum psychosis who experienced a psychiatric relapse during the first 4 weeks postpartum relative to women at risk who remained well had histories of severe childhood adversity as well as biomarkers consistent with a dysregulated stress axis (a statistically higher daily cortisol level). This is consistent with other data that have implicated the complex role between psychosocial variables as well as neurobiologic variables, such as a dysregulation in the hypothalamic pituitary adrenal axis and other studies that suggest that dysregulated inflammatory status may also drive risk for postpartum psychosis (Hazelgrove K et al. Psychoneuroendocrinology. 2021 Jun. doi: 10.1016/j.psyneuen.2021.105218).
At the end of the day, postpartum psychosis is a psychiatric and obstetrical emergency. In our center, it is rare for women not to be hospitalized with this condition to ensure the safety of the mother as well as her newborn, and to also get her recompensated and functioning as quickly and as significantly as possible. However, an interesting extrapolation of the findings noted by Hazelgrove and colleagues is that it raises the question of what effective treatments might be used to mitigate risk for those at greatest risk for postpartum psychosis. For example, are there other treatments over and above the few effective ones that have been studied as prophylactic pharmacologic interventions that might mitigate risk for recurrence of an illness as serious as postpartum psychosis?
The data suggesting dysregulation of the stress axis as a predictor variable for risk in women vulnerable to postpartum psychosis opens an array of opportunities that are nonpharmacologic, such as mindfulness-based cognitive therapy or other interventions that help to modulate the stress axis. This is a terrific opportunity to have pharmacologic intervention meet nonpharmacologic intervention to potentially mitigate risk for postpartum psychosis with its attendant serious sequelae.
In our own work, where we are evaluating genomic data in an extremely well-characterized group of women with known histories of postpartum psychosis, we are interested to see if we can enhance understanding of the model of risk for postpartum psychosis by factoring in genomic underpinning, history of diagnosis, and psychosocial variables to optimally craft interventions for this population of at-risk women. This brings us one step closer to the future in women’s mental health, to the practice of “precision reproductive psychiatry,” matching interventions to specific presentations across perinatal populations.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
Postpartum depression, in many respects, has become a household term. Over the last decade, there has been increasing awareness of the importance of screening for postpartum depression, with increased systematic screening across clinical settings where care is delivered to women during pregnancy and the postpartum period. There have also been greater efforts to identify women who are suffering postpartum depression and to support them with appropriate clinical interventions, whether through psychotherapy and/or pharmacologic therapy. Clinical interventions are supplemented by the increasing awareness of the value of community-based support groups for women who are suffering from postpartum mood and anxiety disorders.
Despite the growing, appropriate focus on recognition and acute treatment of postpartum depression as well as assessing clinical outcomes for those who suffer from postpartum mood and anxiety disorders, less attention has been given to postpartum psychosis, the most severe form of postpartum depression. It is indeed, the least common postpartum psychiatric syndrome (1 in 1,000 births) and comes to public attention when there has been a tragedy during the acute postpartum period such as maternal suicide or infanticide. In this sense, postpartum psychosis is ironically an underappreciated clinical entity across America given its severity, the effect it has on longer-term psychiatric morbidity, and its effect on children and families.
Our group at the Center for Women’s Mental Health has been interested in postpartum psychosis for years and started the Postpartum Psychosis Project in an effort to better understand the phenomenology, course, treatment, outcome, and genomic underpinnings of postpartum psychosis. Risk factors that are well established for postpartum psychosis have been described and overwhelmingly include patients with bipolar disorder. The risk for recurrent postpartum psychosis in women who have had a previous episode is as great as 75%-90% in the absence of prophylactic intervention. With that said, we are extremely interested in understanding the etiology of postpartum psychosis. Various studies over the last 5 years have looked at a whole host of psychosocial as well as neurobiologic variables that may contribute to risk for postpartum psychosis, including dysregulation of the stress axis, heightened inflammation as well as a history of child adversity and heightened experience of stress during the perinatal period.
There have also been anecdotal reports during Virtual Rounds at the Center for Women’s Mental Health of higher recent rates of postpartum psychosis manifesting during the postpartum period. This is a clinical observation and has not been systematically studied. However, one can wonder whether the experience of the pandemic has constituted a stressor for at-risk women, tipping the scales toward women becoming ill, or whether clinicians are seeing this finding more because of our ability to observe it more within the context of the pandemic.
Precise quantification of risk for postpartum psychosis is complicated; as noted, women with bipolar disorder have a predictably high risk for getting ill during the postpartum period and many go on to have clinical courses consistent with recurrent bipolar disorder. However, there are other women who have circumscribed episodes of psychotic illness in the postpartum period who recover and are totally well without any evidence of psychiatric disorder until they have another child, at which time the risk for recurrence of postpartum psychosis is very high. Interest in developing a model of risk that could reliably predict an illness as serious as postpartum psychosis is on the minds of researchers around the world.
One recent study that highlights the multiple factors involved in risk of postpartum psychosis involved a prospective longitudinal study of a group of women who were followed across the peripartum period from the third trimester until 4 weeks postpartum. In this group, 51 women were at increased risk for postpartum psychosis based on their diagnosis of bipolar disorder, schizoaffective disorder, or a previous episode of postpartum psychosis. These women were matched with a control group with no past or current diagnosis of psychiatric disorder or family history of postpartum psychosis. The findings suggested that women at risk for postpartum psychosis who experienced a psychiatric relapse during the first 4 weeks postpartum relative to women at risk who remained well had histories of severe childhood adversity as well as biomarkers consistent with a dysregulated stress axis (a statistically higher daily cortisol level). This is consistent with other data that have implicated the complex role between psychosocial variables as well as neurobiologic variables, such as a dysregulation in the hypothalamic pituitary adrenal axis and other studies that suggest that dysregulated inflammatory status may also drive risk for postpartum psychosis (Hazelgrove K et al. Psychoneuroendocrinology. 2021 Jun. doi: 10.1016/j.psyneuen.2021.105218).
At the end of the day, postpartum psychosis is a psychiatric and obstetrical emergency. In our center, it is rare for women not to be hospitalized with this condition to ensure the safety of the mother as well as her newborn, and to also get her recompensated and functioning as quickly and as significantly as possible. However, an interesting extrapolation of the findings noted by Hazelgrove and colleagues is that it raises the question of what effective treatments might be used to mitigate risk for those at greatest risk for postpartum psychosis. For example, are there other treatments over and above the few effective ones that have been studied as prophylactic pharmacologic interventions that might mitigate risk for recurrence of an illness as serious as postpartum psychosis?
The data suggesting dysregulation of the stress axis as a predictor variable for risk in women vulnerable to postpartum psychosis opens an array of opportunities that are nonpharmacologic, such as mindfulness-based cognitive therapy or other interventions that help to modulate the stress axis. This is a terrific opportunity to have pharmacologic intervention meet nonpharmacologic intervention to potentially mitigate risk for postpartum psychosis with its attendant serious sequelae.
In our own work, where we are evaluating genomic data in an extremely well-characterized group of women with known histories of postpartum psychosis, we are interested to see if we can enhance understanding of the model of risk for postpartum psychosis by factoring in genomic underpinning, history of diagnosis, and psychosocial variables to optimally craft interventions for this population of at-risk women. This brings us one step closer to the future in women’s mental health, to the practice of “precision reproductive psychiatry,” matching interventions to specific presentations across perinatal populations.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Email Dr. Cohen at [email protected].
‘Reassuring’ findings for second-generation antipsychotics during pregnancy
Second-generation antipsychotics (SGAs) taken by pregnant women are linked to a low rate of adverse effects in their children, new research suggests.
Data from a large registry study of almost 2,000 women showed that 2.5% of the live births in a group that had been exposed to antipsychotics had confirmed major malformations compared with 2% of the live births in a non-exposed group. This translated into an estimated odds ratio of 1.5 for major malformations.
“The 2.5% absolute risk for major malformations is consistent with the estimates of the Centers for Disease Control and Prevention’s national baseline rate of major malformations in the general population,” lead author Adele Viguera, MD, MPH, director of research for women’s mental health, Cleveland Clinic Neurological Institute, told this news organization.
“Our results are reassuring and suggest that second-generation antipsychotics, as a class, do not substantially increase the risk of major malformations,” Dr. Viguera said.
The findings were published online August 3 in the Journal of Clinical Psychiatry.
Safety data scarce
Despite the increasing use of SGAs to treat a “spectrum of psychiatric disorders,” relatively little data are available on the reproductive safety of these agents, Dr. Viguera said.
The National Pregnancy Registry for Atypical Antipsychotics (NPRAA) was established in 2008 to determine risk for major malformation among infants exposed to these medications during the first trimester, relative to a comparison group of unexposed infants of mothers with histories of psychiatric morbidity.
The NPRAA follows pregnant women (aged 18 to 45 years) with psychiatric illness who are exposed or unexposed to SGAs during pregnancy. Participants are recruited through nationwide provider referral, self-referral, and advertisement through the Massachusetts General Hospital Center for Women’s Mental Health website.
Specific data collected are shown in the following table.
Since publication of the first results in 2015, the sample size for the trial has increased – and the absolute and relative risk for major malformations observed in the study population are “more precise,” the investigators note. The current study presented updated previous findings.
Demographic differences
Of the 1,906 women who enrolled as of April 2020, 1,311 (mean age, 32.6 years; 81.3% White) completed the study and were eligible for inclusion in the analysis.
Although the groups had a virtually identical mean age, fewer women in the exposure group were married compared with those in the non-exposure group (77% vs. 90%, respectively) and fewer had a college education (71.2% vs. 87.8%). There was also a higher percentage of first-trimester cigarette smokers in the exposure group (18.4% vs. 5.1%).
On the other hand, more women in the non-exposure group used alcohol than in the exposure group (28.6% vs. 21.4%, respectively).
The most frequent psychiatric disorder in the exposure group was bipolar disorder (63.9%), followed by major depression (12.9%), anxiety (5.8%), and schizophrenia (4.5%). Only 11.4% of women in the non-exposure group were diagnosed with bipolar disorder, whereas 34.1% were diagnosed with major depression, 31.3% with anxiety, and none with schizophrenia.
Notably, a large percentage of women in both groups had a history of postpartum depression and/or psychosis (41.4% and 35.5%, respectively).
The most frequently used SGAs in the exposure group were quetiapine (Seroquel), aripiprazole (Abilify), and lurasidone (Latuda).
Participants in the exposure group had a higher age at initial onset of primary psychiatric diagnosis and a lower proportion of lifetime illness compared with those in the non-exposure group.
Major clinical implication?
Among 640 live births in the exposure group, which included 17 twin pregnancies and 1 triplet pregnancy, 2.5% reported major malformations. Among 704 live births in the control group, which included 14 twin pregnancies, 1.99% reported major malformations.
The estimated OR for major malformations comparing exposed and unexposed infants was 1.48 (95% confidence interval, 0.625-3.517).
The authors note that their findings were consistent with one of the largest studies to date, which included a nationwide sample of more than 1 million women. Its results showed that, among infants exposed to SGAs versus those who were not exposed, the estimated risk ratio after adjusting for psychiatric conditions was 1.05 (95% CI, 0.96-1.16).
Additionally, “a hallmark of a teratogen is that it tends to cause a specific type or pattern of malformations, and we found no preponderance of one single type of major malformation or specific pattern of malformations among the exposed and unexposed groups,” Dr. Viguera said
“A major clinical implication of these findings is that for women with major mood and/or psychotic disorders, treatment with an atypical antipsychotic during pregnancy may be the most prudent clinical decision, much as continued treatment is recommended for pregnant women with other serious and chronic medical conditions, such as epilepsy,” she added.
The concept of ‘satisficing’
Commenting on the study, Vivien Burt, MD, PhD, founder and director/consultant of the Women’s Life Center at the Resnick University of California, Los Angeles (UCLA) Neuropsychiatric Hospital, called the findings “reassuring.”
The results “support the conclusion that in pregnant women with serious psychiatric illnesses, the use of SGAs is often a better option than avoiding these medications and exposing both the women and their offspring to the adverse consequences of maternal mental illness,” she said.
An accompanying editorial co-authored by Dr. Burt and colleague Sonya Rasminsky, MD, introduced the concept of “satisficing” – a term coined by Herbert Simon, a behavioral economist and Nobel Laureate. “Satisficing” is a “decision-making strategy that aims for a satisfactory (‘good enough’) outcome rather than a perfect one.”
The concept applies to decision-making beyond the field of economics “and is critical to how physicians help patients make decisions when they are faced with multiple treatment options,” said Dr. Burt, a professor emeritus of psychiatry at UCLA.
“The goal of ‘satisficing’ is to plan for the most satisfactory outcome, knowing that there are always unknowns, so in an uncertain world, clinicians should carefully help their patients make decisions that will allow them to achieve an outcome they can best live with,” she noted.
The investigators note that their findings may not be generalizable to the larger population of women taking SGAs, given that their participants were “overwhelmingly White, married, and well-educated women.”
They add that enrollment into the NPRAA registry is ongoing and larger sample sizes will “further narrow the confidence interval around the risk estimates and allow for adjustment of likely sources of confounding.”
The NPRAA is supported by Alkermes, Johnson & Johnson/Janssen Pharmaceuticals, Otsuka America Pharmaceutical, Sunovion Pharmaceuticals, SAGE Therapeutics, Teva Pharmaceuticals, and Aurobindo Pharma. Past sponsors of the NPRAA are listed in the original paper. Dr. Viguera receives research support from the NPRAA, Alkermes Biopharmaceuticals, Aurobindo Pharma, Janssen Pharmaceuticals, Otsuka Pharmaceutical, Sunovion Pharmaceuticals, Teva Pharmaceuticals, and SAGE Therapeutics and receives adviser/consulting fees from Up-to-Date. Dr. Burt has been a consultant/speaker for Sage Therapeutics. Dr. Rasminsky has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Second-generation antipsychotics (SGAs) taken by pregnant women are linked to a low rate of adverse effects in their children, new research suggests.
Data from a large registry study of almost 2,000 women showed that 2.5% of the live births in a group that had been exposed to antipsychotics had confirmed major malformations compared with 2% of the live births in a non-exposed group. This translated into an estimated odds ratio of 1.5 for major malformations.
“The 2.5% absolute risk for major malformations is consistent with the estimates of the Centers for Disease Control and Prevention’s national baseline rate of major malformations in the general population,” lead author Adele Viguera, MD, MPH, director of research for women’s mental health, Cleveland Clinic Neurological Institute, told this news organization.
“Our results are reassuring and suggest that second-generation antipsychotics, as a class, do not substantially increase the risk of major malformations,” Dr. Viguera said.
The findings were published online August 3 in the Journal of Clinical Psychiatry.
Safety data scarce
Despite the increasing use of SGAs to treat a “spectrum of psychiatric disorders,” relatively little data are available on the reproductive safety of these agents, Dr. Viguera said.
The National Pregnancy Registry for Atypical Antipsychotics (NPRAA) was established in 2008 to determine risk for major malformation among infants exposed to these medications during the first trimester, relative to a comparison group of unexposed infants of mothers with histories of psychiatric morbidity.
The NPRAA follows pregnant women (aged 18 to 45 years) with psychiatric illness who are exposed or unexposed to SGAs during pregnancy. Participants are recruited through nationwide provider referral, self-referral, and advertisement through the Massachusetts General Hospital Center for Women’s Mental Health website.
Specific data collected are shown in the following table.
Since publication of the first results in 2015, the sample size for the trial has increased – and the absolute and relative risk for major malformations observed in the study population are “more precise,” the investigators note. The current study presented updated previous findings.
Demographic differences
Of the 1,906 women who enrolled as of April 2020, 1,311 (mean age, 32.6 years; 81.3% White) completed the study and were eligible for inclusion in the analysis.
Although the groups had a virtually identical mean age, fewer women in the exposure group were married compared with those in the non-exposure group (77% vs. 90%, respectively) and fewer had a college education (71.2% vs. 87.8%). There was also a higher percentage of first-trimester cigarette smokers in the exposure group (18.4% vs. 5.1%).
On the other hand, more women in the non-exposure group used alcohol than in the exposure group (28.6% vs. 21.4%, respectively).
The most frequent psychiatric disorder in the exposure group was bipolar disorder (63.9%), followed by major depression (12.9%), anxiety (5.8%), and schizophrenia (4.5%). Only 11.4% of women in the non-exposure group were diagnosed with bipolar disorder, whereas 34.1% were diagnosed with major depression, 31.3% with anxiety, and none with schizophrenia.
Notably, a large percentage of women in both groups had a history of postpartum depression and/or psychosis (41.4% and 35.5%, respectively).
The most frequently used SGAs in the exposure group were quetiapine (Seroquel), aripiprazole (Abilify), and lurasidone (Latuda).
Participants in the exposure group had a higher age at initial onset of primary psychiatric diagnosis and a lower proportion of lifetime illness compared with those in the non-exposure group.
Major clinical implication?
Among 640 live births in the exposure group, which included 17 twin pregnancies and 1 triplet pregnancy, 2.5% reported major malformations. Among 704 live births in the control group, which included 14 twin pregnancies, 1.99% reported major malformations.
The estimated OR for major malformations comparing exposed and unexposed infants was 1.48 (95% confidence interval, 0.625-3.517).
The authors note that their findings were consistent with one of the largest studies to date, which included a nationwide sample of more than 1 million women. Its results showed that, among infants exposed to SGAs versus those who were not exposed, the estimated risk ratio after adjusting for psychiatric conditions was 1.05 (95% CI, 0.96-1.16).
Additionally, “a hallmark of a teratogen is that it tends to cause a specific type or pattern of malformations, and we found no preponderance of one single type of major malformation or specific pattern of malformations among the exposed and unexposed groups,” Dr. Viguera said
“A major clinical implication of these findings is that for women with major mood and/or psychotic disorders, treatment with an atypical antipsychotic during pregnancy may be the most prudent clinical decision, much as continued treatment is recommended for pregnant women with other serious and chronic medical conditions, such as epilepsy,” she added.
The concept of ‘satisficing’
Commenting on the study, Vivien Burt, MD, PhD, founder and director/consultant of the Women’s Life Center at the Resnick University of California, Los Angeles (UCLA) Neuropsychiatric Hospital, called the findings “reassuring.”
The results “support the conclusion that in pregnant women with serious psychiatric illnesses, the use of SGAs is often a better option than avoiding these medications and exposing both the women and their offspring to the adverse consequences of maternal mental illness,” she said.
An accompanying editorial co-authored by Dr. Burt and colleague Sonya Rasminsky, MD, introduced the concept of “satisficing” – a term coined by Herbert Simon, a behavioral economist and Nobel Laureate. “Satisficing” is a “decision-making strategy that aims for a satisfactory (‘good enough’) outcome rather than a perfect one.”
The concept applies to decision-making beyond the field of economics “and is critical to how physicians help patients make decisions when they are faced with multiple treatment options,” said Dr. Burt, a professor emeritus of psychiatry at UCLA.
“The goal of ‘satisficing’ is to plan for the most satisfactory outcome, knowing that there are always unknowns, so in an uncertain world, clinicians should carefully help their patients make decisions that will allow them to achieve an outcome they can best live with,” she noted.
The investigators note that their findings may not be generalizable to the larger population of women taking SGAs, given that their participants were “overwhelmingly White, married, and well-educated women.”
They add that enrollment into the NPRAA registry is ongoing and larger sample sizes will “further narrow the confidence interval around the risk estimates and allow for adjustment of likely sources of confounding.”
The NPRAA is supported by Alkermes, Johnson & Johnson/Janssen Pharmaceuticals, Otsuka America Pharmaceutical, Sunovion Pharmaceuticals, SAGE Therapeutics, Teva Pharmaceuticals, and Aurobindo Pharma. Past sponsors of the NPRAA are listed in the original paper. Dr. Viguera receives research support from the NPRAA, Alkermes Biopharmaceuticals, Aurobindo Pharma, Janssen Pharmaceuticals, Otsuka Pharmaceutical, Sunovion Pharmaceuticals, Teva Pharmaceuticals, and SAGE Therapeutics and receives adviser/consulting fees from Up-to-Date. Dr. Burt has been a consultant/speaker for Sage Therapeutics. Dr. Rasminsky has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Second-generation antipsychotics (SGAs) taken by pregnant women are linked to a low rate of adverse effects in their children, new research suggests.
Data from a large registry study of almost 2,000 women showed that 2.5% of the live births in a group that had been exposed to antipsychotics had confirmed major malformations compared with 2% of the live births in a non-exposed group. This translated into an estimated odds ratio of 1.5 for major malformations.
“The 2.5% absolute risk for major malformations is consistent with the estimates of the Centers for Disease Control and Prevention’s national baseline rate of major malformations in the general population,” lead author Adele Viguera, MD, MPH, director of research for women’s mental health, Cleveland Clinic Neurological Institute, told this news organization.
“Our results are reassuring and suggest that second-generation antipsychotics, as a class, do not substantially increase the risk of major malformations,” Dr. Viguera said.
The findings were published online August 3 in the Journal of Clinical Psychiatry.
Safety data scarce
Despite the increasing use of SGAs to treat a “spectrum of psychiatric disorders,” relatively little data are available on the reproductive safety of these agents, Dr. Viguera said.
The National Pregnancy Registry for Atypical Antipsychotics (NPRAA) was established in 2008 to determine risk for major malformation among infants exposed to these medications during the first trimester, relative to a comparison group of unexposed infants of mothers with histories of psychiatric morbidity.
The NPRAA follows pregnant women (aged 18 to 45 years) with psychiatric illness who are exposed or unexposed to SGAs during pregnancy. Participants are recruited through nationwide provider referral, self-referral, and advertisement through the Massachusetts General Hospital Center for Women’s Mental Health website.
Specific data collected are shown in the following table.
Since publication of the first results in 2015, the sample size for the trial has increased – and the absolute and relative risk for major malformations observed in the study population are “more precise,” the investigators note. The current study presented updated previous findings.
Demographic differences
Of the 1,906 women who enrolled as of April 2020, 1,311 (mean age, 32.6 years; 81.3% White) completed the study and were eligible for inclusion in the analysis.
Although the groups had a virtually identical mean age, fewer women in the exposure group were married compared with those in the non-exposure group (77% vs. 90%, respectively) and fewer had a college education (71.2% vs. 87.8%). There was also a higher percentage of first-trimester cigarette smokers in the exposure group (18.4% vs. 5.1%).
On the other hand, more women in the non-exposure group used alcohol than in the exposure group (28.6% vs. 21.4%, respectively).
The most frequent psychiatric disorder in the exposure group was bipolar disorder (63.9%), followed by major depression (12.9%), anxiety (5.8%), and schizophrenia (4.5%). Only 11.4% of women in the non-exposure group were diagnosed with bipolar disorder, whereas 34.1% were diagnosed with major depression, 31.3% with anxiety, and none with schizophrenia.
Notably, a large percentage of women in both groups had a history of postpartum depression and/or psychosis (41.4% and 35.5%, respectively).
The most frequently used SGAs in the exposure group were quetiapine (Seroquel), aripiprazole (Abilify), and lurasidone (Latuda).
Participants in the exposure group had a higher age at initial onset of primary psychiatric diagnosis and a lower proportion of lifetime illness compared with those in the non-exposure group.
Major clinical implication?
Among 640 live births in the exposure group, which included 17 twin pregnancies and 1 triplet pregnancy, 2.5% reported major malformations. Among 704 live births in the control group, which included 14 twin pregnancies, 1.99% reported major malformations.
The estimated OR for major malformations comparing exposed and unexposed infants was 1.48 (95% confidence interval, 0.625-3.517).
The authors note that their findings were consistent with one of the largest studies to date, which included a nationwide sample of more than 1 million women. Its results showed that, among infants exposed to SGAs versus those who were not exposed, the estimated risk ratio after adjusting for psychiatric conditions was 1.05 (95% CI, 0.96-1.16).
Additionally, “a hallmark of a teratogen is that it tends to cause a specific type or pattern of malformations, and we found no preponderance of one single type of major malformation or specific pattern of malformations among the exposed and unexposed groups,” Dr. Viguera said
“A major clinical implication of these findings is that for women with major mood and/or psychotic disorders, treatment with an atypical antipsychotic during pregnancy may be the most prudent clinical decision, much as continued treatment is recommended for pregnant women with other serious and chronic medical conditions, such as epilepsy,” she added.
The concept of ‘satisficing’
Commenting on the study, Vivien Burt, MD, PhD, founder and director/consultant of the Women’s Life Center at the Resnick University of California, Los Angeles (UCLA) Neuropsychiatric Hospital, called the findings “reassuring.”
The results “support the conclusion that in pregnant women with serious psychiatric illnesses, the use of SGAs is often a better option than avoiding these medications and exposing both the women and their offspring to the adverse consequences of maternal mental illness,” she said.
An accompanying editorial co-authored by Dr. Burt and colleague Sonya Rasminsky, MD, introduced the concept of “satisficing” – a term coined by Herbert Simon, a behavioral economist and Nobel Laureate. “Satisficing” is a “decision-making strategy that aims for a satisfactory (‘good enough’) outcome rather than a perfect one.”
The concept applies to decision-making beyond the field of economics “and is critical to how physicians help patients make decisions when they are faced with multiple treatment options,” said Dr. Burt, a professor emeritus of psychiatry at UCLA.
“The goal of ‘satisficing’ is to plan for the most satisfactory outcome, knowing that there are always unknowns, so in an uncertain world, clinicians should carefully help their patients make decisions that will allow them to achieve an outcome they can best live with,” she noted.
The investigators note that their findings may not be generalizable to the larger population of women taking SGAs, given that their participants were “overwhelmingly White, married, and well-educated women.”
They add that enrollment into the NPRAA registry is ongoing and larger sample sizes will “further narrow the confidence interval around the risk estimates and allow for adjustment of likely sources of confounding.”
The NPRAA is supported by Alkermes, Johnson & Johnson/Janssen Pharmaceuticals, Otsuka America Pharmaceutical, Sunovion Pharmaceuticals, SAGE Therapeutics, Teva Pharmaceuticals, and Aurobindo Pharma. Past sponsors of the NPRAA are listed in the original paper. Dr. Viguera receives research support from the NPRAA, Alkermes Biopharmaceuticals, Aurobindo Pharma, Janssen Pharmaceuticals, Otsuka Pharmaceutical, Sunovion Pharmaceuticals, Teva Pharmaceuticals, and SAGE Therapeutics and receives adviser/consulting fees from Up-to-Date. Dr. Burt has been a consultant/speaker for Sage Therapeutics. Dr. Rasminsky has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Mental illness admissions: 18-44 is the age of prevalence
More mental and/or substance use disorders are ranked among the top-five diagnoses for hospitalized men and women aged 18-44 years than for any other age group, according to a recent report from the Agency for Healthcare Research and Quality.
aged 18-44, while depressive disorders were the third-most common (195.0 stays per 100,000) and alcohol-related disorders were fifth at 153.2 per 100,000, Kimberly W. McDermott, PhD, and Marc Roemer, MS, said in an AHRQ statistical brief.
Prevalence was somewhat lower in women aged 18-44 years, with two mental illnesses appearing among the top five nonmaternal diagnoses: Depressive disorders were second at 222.5 stays per 100,000 and bipolar and related disorders were fourth at 142.0 per 100,000. The leading primary diagnosis in women in 2018 was septicemia, which was the most common cause overall in the age group at a rate of 279.3 per 100,000, the investigators reported.
There were no mental and/or substance use disorders in the top five primary diagnoses for any of the other adult age groups – 45-64, 65-74, and ≥75 – included in the report. Septicemia was the leading diagnosis for men in all three groups and for women in two of three (45-64 and ≥75), with osteoarthritis first among women aged 65-74 years, they said.
There was one mental illness among the top-five diagnoses for children under age 18 years, as depressive disorders were the most common reason for stays in girls (176.6 per 100,000 population) and the fifth most common for boys (74.0 per 100,000), said Dr. McDermott of IBM Watson Health and Mr. Roemer of AHRQ.
Septicemia was the leading nonmaternal, nonneonatal diagnosis for all inpatient stays and all ages in 2018 with a rate of 679.5 per 100,000, followed by heart failure (347.9), osteoarthritis (345.5), pneumonia not related to tuberculosis (226.8), and diabetes mellitus (207.8), based on data from the National Inpatient Sample.
Depressive disorders were most common mental health diagnosis in those admitted to hospitals and the 12th most common diagnosis overall; schizophrenia, in 16th place overall, was the only other mental illness among the top 20, the investigators said.
“This information can help establish national health priorities, initiatives, and action plans,” Dr. McDermott and Mr. Roemer wrote, and “at the hospital level, administrators can use diagnosis-related information to inform planning and resource allocation, such as optimizing subspecialty services or units for the care of high-priority conditions.”
More mental and/or substance use disorders are ranked among the top-five diagnoses for hospitalized men and women aged 18-44 years than for any other age group, according to a recent report from the Agency for Healthcare Research and Quality.

aged 18-44, while depressive disorders were the third-most common (195.0 stays per 100,000) and alcohol-related disorders were fifth at 153.2 per 100,000, Kimberly W. McDermott, PhD, and Marc Roemer, MS, said in an AHRQ statistical brief.
Prevalence was somewhat lower in women aged 18-44 years, with two mental illnesses appearing among the top five nonmaternal diagnoses: Depressive disorders were second at 222.5 stays per 100,000 and bipolar and related disorders were fourth at 142.0 per 100,000. The leading primary diagnosis in women in 2018 was septicemia, which was the most common cause overall in the age group at a rate of 279.3 per 100,000, the investigators reported.
There were no mental and/or substance use disorders in the top five primary diagnoses for any of the other adult age groups – 45-64, 65-74, and ≥75 – included in the report. Septicemia was the leading diagnosis for men in all three groups and for women in two of three (45-64 and ≥75), with osteoarthritis first among women aged 65-74 years, they said.
There was one mental illness among the top-five diagnoses for children under age 18 years, as depressive disorders were the most common reason for stays in girls (176.6 per 100,000 population) and the fifth most common for boys (74.0 per 100,000), said Dr. McDermott of IBM Watson Health and Mr. Roemer of AHRQ.
Septicemia was the leading nonmaternal, nonneonatal diagnosis for all inpatient stays and all ages in 2018 with a rate of 679.5 per 100,000, followed by heart failure (347.9), osteoarthritis (345.5), pneumonia not related to tuberculosis (226.8), and diabetes mellitus (207.8), based on data from the National Inpatient Sample.
Depressive disorders were most common mental health diagnosis in those admitted to hospitals and the 12th most common diagnosis overall; schizophrenia, in 16th place overall, was the only other mental illness among the top 20, the investigators said.
“This information can help establish national health priorities, initiatives, and action plans,” Dr. McDermott and Mr. Roemer wrote, and “at the hospital level, administrators can use diagnosis-related information to inform planning and resource allocation, such as optimizing subspecialty services or units for the care of high-priority conditions.”
More mental and/or substance use disorders are ranked among the top-five diagnoses for hospitalized men and women aged 18-44 years than for any other age group, according to a recent report from the Agency for Healthcare Research and Quality.

aged 18-44, while depressive disorders were the third-most common (195.0 stays per 100,000) and alcohol-related disorders were fifth at 153.2 per 100,000, Kimberly W. McDermott, PhD, and Marc Roemer, MS, said in an AHRQ statistical brief.
Prevalence was somewhat lower in women aged 18-44 years, with two mental illnesses appearing among the top five nonmaternal diagnoses: Depressive disorders were second at 222.5 stays per 100,000 and bipolar and related disorders were fourth at 142.0 per 100,000. The leading primary diagnosis in women in 2018 was septicemia, which was the most common cause overall in the age group at a rate of 279.3 per 100,000, the investigators reported.
There were no mental and/or substance use disorders in the top five primary diagnoses for any of the other adult age groups – 45-64, 65-74, and ≥75 – included in the report. Septicemia was the leading diagnosis for men in all three groups and for women in two of three (45-64 and ≥75), with osteoarthritis first among women aged 65-74 years, they said.
There was one mental illness among the top-five diagnoses for children under age 18 years, as depressive disorders were the most common reason for stays in girls (176.6 per 100,000 population) and the fifth most common for boys (74.0 per 100,000), said Dr. McDermott of IBM Watson Health and Mr. Roemer of AHRQ.
Septicemia was the leading nonmaternal, nonneonatal diagnosis for all inpatient stays and all ages in 2018 with a rate of 679.5 per 100,000, followed by heart failure (347.9), osteoarthritis (345.5), pneumonia not related to tuberculosis (226.8), and diabetes mellitus (207.8), based on data from the National Inpatient Sample.
Depressive disorders were most common mental health diagnosis in those admitted to hospitals and the 12th most common diagnosis overall; schizophrenia, in 16th place overall, was the only other mental illness among the top 20, the investigators said.
“This information can help establish national health priorities, initiatives, and action plans,” Dr. McDermott and Mr. Roemer wrote, and “at the hospital level, administrators can use diagnosis-related information to inform planning and resource allocation, such as optimizing subspecialty services or units for the care of high-priority conditions.”
Obesity treatment in mental illness: Is semaglutide a game changer?
It’s probably fair to say that most people would like to be thinner. More than 42% of Americans have obesity and another 30% are classified as being overweight, according to the latest statistics from the CDC.
Excess body weight is associated with many illnesses and plays a role in mental health; being heavy can take a toll on self-esteem. Many people worry that carrying excess weight makes them less attractive to potential romantic partners, and both physicians and employers treat those with obesity differently. Furthermore, in psychiatry, many of the medications we prescribe lead to weight gain.
In my clinical practice, I have listened as patients blamed themselves for their body habitus; many won’t consider biological treatments as they feel that would be “cheating” or taking an easy way out. They often point to periods in their life when they did lose weight and believe that they should be able to do it again, even if the weight loss took tremendous effort, was not sustained, and occurred decades ago.
That said, we psychiatrists often find ourselves in the position of managing obesity in our patients. I have been known to give patients who gain weight on antipsychotics either stimulants or metformin, or to add naltrexone to their Wellbutrin (bupropion) to effectively mimic a weight-loss medicine called Contrave.
Obesity a treatable medical condition
It wasn’t until 2013 that the American Medical Association recognized obesity as a medical condition.
In a New Yorker article that same year, “Diet Drugs Work: Why Won’t Doctors Prescribe Them?” Suzanne Koven wrote: “Several obesity experts told me they’ve encountered doctors who confide that they just didn’t like fat people and don’t enjoy taking care of them. Even doctors who treat obese patients feel stigmatized: ‘diet doctor’ is not a flattering term.”
Eat less, exercise more – with a blame-the-patient attitude – is still what people see as the “right” way to lose weight.
On June 4, 2021, the FDA approved semaglutide, a glucagonlike peptide–1 receptor agonist, previously used for the treatment of diabetes, for use as a weight loss agent for patients with obesity, or for those with a body mass index over 27 kg/m2 if they also have a weight-related comorbidity.
Semaglutide has three trade names, all manufactured by Novo Nordisk. The pill version is called Rybelsus and comes in 7-mg and 14-mg tablets. Ozempic is available in 0.5-mg and 1.0-mg doses and is administered weekly by subcutaneous injection for diabetes. The new, higher-dose preparation for weight loss, Wegovy, 2.4 mg, also comes as a weekly subcutaneous dose and is now available for the hefty price of $1,400 per month.
In STEP 1 trials, the higher-dose Wegovy was associated with an average 14.9% weight loss (15.3 kg) over 68 weeks, more than any other single-agent weight loss medication on the market.
GLP-1 receptor agonists work in the brain to decrease appetite, slow gastric emptying, increase insulin secretion, and stimulate brown adipose tissue thermogenesis.
Psych drugs lead to weight gain
Elaine Weiner, MD, is the medical director in the outpatient research program of the Maryland Psychiatric Research Center in Catonsville, where she treats patients with schizophrenia.
“Nearly all of our patients gain 20 pounds or more on the combinations of medications we use, mostly atypical antipsychotics,” she said. “Weight management is difficult for people who don’t have problems with motivation, but in our patients, lack of motivation is a core part of their illness, so asking them to adhere to diet and exercise regimens is of limited utility.
“Then, add to that the fact that they sometimes don’t have primary care doctors, and these issues of weight gain and metabolic syndrome come back to the psychiatrist. It is a really bad problem and we need more treatments.”
Fatima Cody Stanford, MD, MPH, MPA, is a fellowship-trained obesity medicine physician-scientist at the Massachusetts General Hospital Weight Center and Harvard Medical School, both in Boston. She has treated thousands of patients with obesity, speaks internationally on the topic of weight loss medicine, and has published over 100 peer-reviewed articles on obesity.
We spoke at length about recent changes in the field of obesity medicine and the introduction of the new GLP-1 receptor agonists.
“We as physicians have learned so little,” Dr. Stanford said. “This mantra of ‘calories in, calories out’ is not working; this is inaccurate and our focus on this has led to a rise in obesity. All calories are not created the same, and I think we are finally starting to see obesity medicine take off.”
Dr. Stanford is quick to note that obesity is a complex problem. Several different hormones are involved in regulating both appetite and satiety, processed foods promote weight gain, sleep is crucial to weight loss, and exercise helps maintain weight loss but is not usually effective in promoting it. “There are many contributors to energy storage,” she said.
The stimulant phentermine was approved in 1959. Addiction was a concern, and then in the 1990s, it was used in combination with fenfluramine to promote weight loss, a combination known as phen-fen. Fenfluramine was pulled from the market in 1997 when it was found to be associated with pulmonary hypertension and then heart valve abnormalities.
“This frightened quite a few physicians,” Dr. Stanford noted. Phentermine is still used for weight loss, either alone or together with topiramate, as a combination medication called Qsymia, nicknamed phen-top.
“Phen-top is the next best thing we have to semaglutide, and there is an average weight loss of 8%-9% of body weight. Semaglutide is going to be really significant for those people who are responders, and this has been quite well tolerated, the most common side effect being nausea,” she said.
However, she is quick to note that not everyone responds to every medication. “I use each patient’s clinical profile to determine what strategies and which medications to use.”
Cardiologists getting in the game
Michael Miller, MD, is a cardiologist at the University of Maryland, Baltimore, and author of “Heal Your Heart” (Emmaus, Pa.: Rodale, 2014). He is very enthusiastic about the approval of semaglutide.
“We are so excited because you finally can use these medicines without having to be diabetic,” Dr. Miller said. “We’re waiting on the results of the SELECT [Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity] trials looking at people who are not diabetic or who are prediabetic, to see the 5-year outcomes with regard to cardiac events.
“Usually endocrinologists prescribe these medications, but cardiologists have started to get into the game since GLP-1 receptor agonists reduce cardiovascular events.” Dr. Miller is hopeful that this medication may neutralize the weight gain caused by psychotropic medications.
Wegovy is administered via weekly injection and, like insulin, is a subcutaneous medication that patients self-administer. Will patients be amenable to injecting a medication for weight loss? Dr. Stanford said that roughly 20%-30% of her patients are hesitant when she suggests that they use liraglutide, another GLP-1 receptor agonist that is approved for weight loss, and some are very fearful of needles.
However, she also noted that during the COVID-19 pandemic, many more patients have sought treatment from obesity medicine physicians because of the association between obesity and mortality from COVID-19. Patients have been willing to consider treatments that they were not previously open to pursuing.
So if people are willing to take Wegovy and doctors are willing to prescribe it, will insurers pay for it? As of this writing, the medication is not yet available, but Ozempic, the lower-dose agent for diabetes, costs $850-$900 for a 4-week supply, according to the GoodRx website.
Liraglutide (Saxenda), the GLP-1 receptor agonist that is currently available for weight loss as a daily injectable, costs $1,300-$1,400 per month.
These medications are not covered by Medicare or Medicaid, and Dr. Stanford, who is well versed as to exactly which private insurers in Massachusetts will and will not reimburse specific medications, said her patients with insurance coverage have been known to delay retirement so that they can remain on the more expensive medications.
“For the past 8 years,” she said, “the Treat and Reduce Obesity Act has had bipartisan support in Congress but has not passed. We are still hopeful that insurers will be required to cover medical and behavioral treatments for obesity.”
As our society struggles to destigmatize so many disorders, obesity remains a highly stigmatized condition, one that our patients cannot hide and one that leads to so many other comorbid illnesses. As new treatments are approved, there will be more for physicians to offer. Semaglutide, if it becomes available to those who need it most, could be a game changer. For patients who have not had success with traditional weight-loss methods, it’s encouraging to have another option available, one that may be reasonable to try before resorting to bariatric surgery.
For decades, psychiatrists have been comfortable prescribing treatments that lead to weight gain. Now, maybe it’s time they also prescribe those that prevent it.
A version of this article first appeared on Medscape.com.
It’s probably fair to say that most people would like to be thinner. More than 42% of Americans have obesity and another 30% are classified as being overweight, according to the latest statistics from the CDC.
Excess body weight is associated with many illnesses and plays a role in mental health; being heavy can take a toll on self-esteem. Many people worry that carrying excess weight makes them less attractive to potential romantic partners, and both physicians and employers treat those with obesity differently. Furthermore, in psychiatry, many of the medications we prescribe lead to weight gain.
In my clinical practice, I have listened as patients blamed themselves for their body habitus; many won’t consider biological treatments as they feel that would be “cheating” or taking an easy way out. They often point to periods in their life when they did lose weight and believe that they should be able to do it again, even if the weight loss took tremendous effort, was not sustained, and occurred decades ago.
That said, we psychiatrists often find ourselves in the position of managing obesity in our patients. I have been known to give patients who gain weight on antipsychotics either stimulants or metformin, or to add naltrexone to their Wellbutrin (bupropion) to effectively mimic a weight-loss medicine called Contrave.
Obesity a treatable medical condition
It wasn’t until 2013 that the American Medical Association recognized obesity as a medical condition.
In a New Yorker article that same year, “Diet Drugs Work: Why Won’t Doctors Prescribe Them?” Suzanne Koven wrote: “Several obesity experts told me they’ve encountered doctors who confide that they just didn’t like fat people and don’t enjoy taking care of them. Even doctors who treat obese patients feel stigmatized: ‘diet doctor’ is not a flattering term.”
Eat less, exercise more – with a blame-the-patient attitude – is still what people see as the “right” way to lose weight.
On June 4, 2021, the FDA approved semaglutide, a glucagonlike peptide–1 receptor agonist, previously used for the treatment of diabetes, for use as a weight loss agent for patients with obesity, or for those with a body mass index over 27 kg/m2 if they also have a weight-related comorbidity.
Semaglutide has three trade names, all manufactured by Novo Nordisk. The pill version is called Rybelsus and comes in 7-mg and 14-mg tablets. Ozempic is available in 0.5-mg and 1.0-mg doses and is administered weekly by subcutaneous injection for diabetes. The new, higher-dose preparation for weight loss, Wegovy, 2.4 mg, also comes as a weekly subcutaneous dose and is now available for the hefty price of $1,400 per month.
In STEP 1 trials, the higher-dose Wegovy was associated with an average 14.9% weight loss (15.3 kg) over 68 weeks, more than any other single-agent weight loss medication on the market.
GLP-1 receptor agonists work in the brain to decrease appetite, slow gastric emptying, increase insulin secretion, and stimulate brown adipose tissue thermogenesis.
Psych drugs lead to weight gain
Elaine Weiner, MD, is the medical director in the outpatient research program of the Maryland Psychiatric Research Center in Catonsville, where she treats patients with schizophrenia.
“Nearly all of our patients gain 20 pounds or more on the combinations of medications we use, mostly atypical antipsychotics,” she said. “Weight management is difficult for people who don’t have problems with motivation, but in our patients, lack of motivation is a core part of their illness, so asking them to adhere to diet and exercise regimens is of limited utility.
“Then, add to that the fact that they sometimes don’t have primary care doctors, and these issues of weight gain and metabolic syndrome come back to the psychiatrist. It is a really bad problem and we need more treatments.”
Fatima Cody Stanford, MD, MPH, MPA, is a fellowship-trained obesity medicine physician-scientist at the Massachusetts General Hospital Weight Center and Harvard Medical School, both in Boston. She has treated thousands of patients with obesity, speaks internationally on the topic of weight loss medicine, and has published over 100 peer-reviewed articles on obesity.
We spoke at length about recent changes in the field of obesity medicine and the introduction of the new GLP-1 receptor agonists.
“We as physicians have learned so little,” Dr. Stanford said. “This mantra of ‘calories in, calories out’ is not working; this is inaccurate and our focus on this has led to a rise in obesity. All calories are not created the same, and I think we are finally starting to see obesity medicine take off.”
Dr. Stanford is quick to note that obesity is a complex problem. Several different hormones are involved in regulating both appetite and satiety, processed foods promote weight gain, sleep is crucial to weight loss, and exercise helps maintain weight loss but is not usually effective in promoting it. “There are many contributors to energy storage,” she said.
The stimulant phentermine was approved in 1959. Addiction was a concern, and then in the 1990s, it was used in combination with fenfluramine to promote weight loss, a combination known as phen-fen. Fenfluramine was pulled from the market in 1997 when it was found to be associated with pulmonary hypertension and then heart valve abnormalities.
“This frightened quite a few physicians,” Dr. Stanford noted. Phentermine is still used for weight loss, either alone or together with topiramate, as a combination medication called Qsymia, nicknamed phen-top.
“Phen-top is the next best thing we have to semaglutide, and there is an average weight loss of 8%-9% of body weight. Semaglutide is going to be really significant for those people who are responders, and this has been quite well tolerated, the most common side effect being nausea,” she said.
However, she is quick to note that not everyone responds to every medication. “I use each patient’s clinical profile to determine what strategies and which medications to use.”
Cardiologists getting in the game
Michael Miller, MD, is a cardiologist at the University of Maryland, Baltimore, and author of “Heal Your Heart” (Emmaus, Pa.: Rodale, 2014). He is very enthusiastic about the approval of semaglutide.
“We are so excited because you finally can use these medicines without having to be diabetic,” Dr. Miller said. “We’re waiting on the results of the SELECT [Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity] trials looking at people who are not diabetic or who are prediabetic, to see the 5-year outcomes with regard to cardiac events.
“Usually endocrinologists prescribe these medications, but cardiologists have started to get into the game since GLP-1 receptor agonists reduce cardiovascular events.” Dr. Miller is hopeful that this medication may neutralize the weight gain caused by psychotropic medications.
Wegovy is administered via weekly injection and, like insulin, is a subcutaneous medication that patients self-administer. Will patients be amenable to injecting a medication for weight loss? Dr. Stanford said that roughly 20%-30% of her patients are hesitant when she suggests that they use liraglutide, another GLP-1 receptor agonist that is approved for weight loss, and some are very fearful of needles.
However, she also noted that during the COVID-19 pandemic, many more patients have sought treatment from obesity medicine physicians because of the association between obesity and mortality from COVID-19. Patients have been willing to consider treatments that they were not previously open to pursuing.
So if people are willing to take Wegovy and doctors are willing to prescribe it, will insurers pay for it? As of this writing, the medication is not yet available, but Ozempic, the lower-dose agent for diabetes, costs $850-$900 for a 4-week supply, according to the GoodRx website.
Liraglutide (Saxenda), the GLP-1 receptor agonist that is currently available for weight loss as a daily injectable, costs $1,300-$1,400 per month.
These medications are not covered by Medicare or Medicaid, and Dr. Stanford, who is well versed as to exactly which private insurers in Massachusetts will and will not reimburse specific medications, said her patients with insurance coverage have been known to delay retirement so that they can remain on the more expensive medications.
“For the past 8 years,” she said, “the Treat and Reduce Obesity Act has had bipartisan support in Congress but has not passed. We are still hopeful that insurers will be required to cover medical and behavioral treatments for obesity.”
As our society struggles to destigmatize so many disorders, obesity remains a highly stigmatized condition, one that our patients cannot hide and one that leads to so many other comorbid illnesses. As new treatments are approved, there will be more for physicians to offer. Semaglutide, if it becomes available to those who need it most, could be a game changer. For patients who have not had success with traditional weight-loss methods, it’s encouraging to have another option available, one that may be reasonable to try before resorting to bariatric surgery.
For decades, psychiatrists have been comfortable prescribing treatments that lead to weight gain. Now, maybe it’s time they also prescribe those that prevent it.
A version of this article first appeared on Medscape.com.
It’s probably fair to say that most people would like to be thinner. More than 42% of Americans have obesity and another 30% are classified as being overweight, according to the latest statistics from the CDC.
Excess body weight is associated with many illnesses and plays a role in mental health; being heavy can take a toll on self-esteem. Many people worry that carrying excess weight makes them less attractive to potential romantic partners, and both physicians and employers treat those with obesity differently. Furthermore, in psychiatry, many of the medications we prescribe lead to weight gain.
In my clinical practice, I have listened as patients blamed themselves for their body habitus; many won’t consider biological treatments as they feel that would be “cheating” or taking an easy way out. They often point to periods in their life when they did lose weight and believe that they should be able to do it again, even if the weight loss took tremendous effort, was not sustained, and occurred decades ago.
That said, we psychiatrists often find ourselves in the position of managing obesity in our patients. I have been known to give patients who gain weight on antipsychotics either stimulants or metformin, or to add naltrexone to their Wellbutrin (bupropion) to effectively mimic a weight-loss medicine called Contrave.
Obesity a treatable medical condition
It wasn’t until 2013 that the American Medical Association recognized obesity as a medical condition.
In a New Yorker article that same year, “Diet Drugs Work: Why Won’t Doctors Prescribe Them?” Suzanne Koven wrote: “Several obesity experts told me they’ve encountered doctors who confide that they just didn’t like fat people and don’t enjoy taking care of them. Even doctors who treat obese patients feel stigmatized: ‘diet doctor’ is not a flattering term.”
Eat less, exercise more – with a blame-the-patient attitude – is still what people see as the “right” way to lose weight.
On June 4, 2021, the FDA approved semaglutide, a glucagonlike peptide–1 receptor agonist, previously used for the treatment of diabetes, for use as a weight loss agent for patients with obesity, or for those with a body mass index over 27 kg/m2 if they also have a weight-related comorbidity.
Semaglutide has three trade names, all manufactured by Novo Nordisk. The pill version is called Rybelsus and comes in 7-mg and 14-mg tablets. Ozempic is available in 0.5-mg and 1.0-mg doses and is administered weekly by subcutaneous injection for diabetes. The new, higher-dose preparation for weight loss, Wegovy, 2.4 mg, also comes as a weekly subcutaneous dose and is now available for the hefty price of $1,400 per month.
In STEP 1 trials, the higher-dose Wegovy was associated with an average 14.9% weight loss (15.3 kg) over 68 weeks, more than any other single-agent weight loss medication on the market.
GLP-1 receptor agonists work in the brain to decrease appetite, slow gastric emptying, increase insulin secretion, and stimulate brown adipose tissue thermogenesis.
Psych drugs lead to weight gain
Elaine Weiner, MD, is the medical director in the outpatient research program of the Maryland Psychiatric Research Center in Catonsville, where she treats patients with schizophrenia.
“Nearly all of our patients gain 20 pounds or more on the combinations of medications we use, mostly atypical antipsychotics,” she said. “Weight management is difficult for people who don’t have problems with motivation, but in our patients, lack of motivation is a core part of their illness, so asking them to adhere to diet and exercise regimens is of limited utility.
“Then, add to that the fact that they sometimes don’t have primary care doctors, and these issues of weight gain and metabolic syndrome come back to the psychiatrist. It is a really bad problem and we need more treatments.”
Fatima Cody Stanford, MD, MPH, MPA, is a fellowship-trained obesity medicine physician-scientist at the Massachusetts General Hospital Weight Center and Harvard Medical School, both in Boston. She has treated thousands of patients with obesity, speaks internationally on the topic of weight loss medicine, and has published over 100 peer-reviewed articles on obesity.
We spoke at length about recent changes in the field of obesity medicine and the introduction of the new GLP-1 receptor agonists.
“We as physicians have learned so little,” Dr. Stanford said. “This mantra of ‘calories in, calories out’ is not working; this is inaccurate and our focus on this has led to a rise in obesity. All calories are not created the same, and I think we are finally starting to see obesity medicine take off.”
Dr. Stanford is quick to note that obesity is a complex problem. Several different hormones are involved in regulating both appetite and satiety, processed foods promote weight gain, sleep is crucial to weight loss, and exercise helps maintain weight loss but is not usually effective in promoting it. “There are many contributors to energy storage,” she said.
The stimulant phentermine was approved in 1959. Addiction was a concern, and then in the 1990s, it was used in combination with fenfluramine to promote weight loss, a combination known as phen-fen. Fenfluramine was pulled from the market in 1997 when it was found to be associated with pulmonary hypertension and then heart valve abnormalities.
“This frightened quite a few physicians,” Dr. Stanford noted. Phentermine is still used for weight loss, either alone or together with topiramate, as a combination medication called Qsymia, nicknamed phen-top.
“Phen-top is the next best thing we have to semaglutide, and there is an average weight loss of 8%-9% of body weight. Semaglutide is going to be really significant for those people who are responders, and this has been quite well tolerated, the most common side effect being nausea,” she said.
However, she is quick to note that not everyone responds to every medication. “I use each patient’s clinical profile to determine what strategies and which medications to use.”
Cardiologists getting in the game
Michael Miller, MD, is a cardiologist at the University of Maryland, Baltimore, and author of “Heal Your Heart” (Emmaus, Pa.: Rodale, 2014). He is very enthusiastic about the approval of semaglutide.
“We are so excited because you finally can use these medicines without having to be diabetic,” Dr. Miller said. “We’re waiting on the results of the SELECT [Semaglutide Effects on Heart Disease and Stroke in Patients With Overweight or Obesity] trials looking at people who are not diabetic or who are prediabetic, to see the 5-year outcomes with regard to cardiac events.
“Usually endocrinologists prescribe these medications, but cardiologists have started to get into the game since GLP-1 receptor agonists reduce cardiovascular events.” Dr. Miller is hopeful that this medication may neutralize the weight gain caused by psychotropic medications.
Wegovy is administered via weekly injection and, like insulin, is a subcutaneous medication that patients self-administer. Will patients be amenable to injecting a medication for weight loss? Dr. Stanford said that roughly 20%-30% of her patients are hesitant when she suggests that they use liraglutide, another GLP-1 receptor agonist that is approved for weight loss, and some are very fearful of needles.
However, she also noted that during the COVID-19 pandemic, many more patients have sought treatment from obesity medicine physicians because of the association between obesity and mortality from COVID-19. Patients have been willing to consider treatments that they were not previously open to pursuing.
So if people are willing to take Wegovy and doctors are willing to prescribe it, will insurers pay for it? As of this writing, the medication is not yet available, but Ozempic, the lower-dose agent for diabetes, costs $850-$900 for a 4-week supply, according to the GoodRx website.
Liraglutide (Saxenda), the GLP-1 receptor agonist that is currently available for weight loss as a daily injectable, costs $1,300-$1,400 per month.
These medications are not covered by Medicare or Medicaid, and Dr. Stanford, who is well versed as to exactly which private insurers in Massachusetts will and will not reimburse specific medications, said her patients with insurance coverage have been known to delay retirement so that they can remain on the more expensive medications.
“For the past 8 years,” she said, “the Treat and Reduce Obesity Act has had bipartisan support in Congress but has not passed. We are still hopeful that insurers will be required to cover medical and behavioral treatments for obesity.”
As our society struggles to destigmatize so many disorders, obesity remains a highly stigmatized condition, one that our patients cannot hide and one that leads to so many other comorbid illnesses. As new treatments are approved, there will be more for physicians to offer. Semaglutide, if it becomes available to those who need it most, could be a game changer. For patients who have not had success with traditional weight-loss methods, it’s encouraging to have another option available, one that may be reasonable to try before resorting to bariatric surgery.
For decades, psychiatrists have been comfortable prescribing treatments that lead to weight gain. Now, maybe it’s time they also prescribe those that prevent it.
A version of this article first appeared on Medscape.com.
Let’s talk about race
“I feel like my aggression is being racialized.” “Of course I wouldn’t call the cops if I felt like hurting myself. I’m Black.”
Those statements represent the heightened trauma our Black and Brown patients with mental health issues have been experiencing. In the wake of increasingly publicized police brutality against Black and Brown communities, the role race plays in mental health decompensation is evident. At this moment in time, we must continue to improve our understanding of the role race plays in psychiatric disorders. We must also ask ourselves: At times, does psychiatry worsen the traumas of the communities we serve?
Some psychiatrists are afraid to speak about race. They may believe it to be too “political.” But avoiding these necessary conversations perpetuates the trauma of those we treat. It suggests that physicians are ignorant of an issue at the forefront of patients’ mental health. Psychiatry, today, is primarily focused on the biological aspects of disease. We must not forget that psychiatry is biopsychosocial. It is imperative that psychiatrists have conversations about race – and its significance to our patients and their care.
Only 10.4% of psychiatrists in the United States comprise those considered underrepresented in medicine (URM). Yet, those very groups make up 32.6% of the U.S. population and are overrepresented in psychiatric hospitals.1 Many studies have shown that concordant racial backgrounds between patient and physician lead to a more positive patient experience2 and arguably, the subsequent potential for better health outcomes. Our efforts in addressing this disparity often fall short. URM applicants may be hesitant to join an institution where diversity is lacking or where they may be the only minority.3 While there is no simple solution, I propose that psychiatrists promote the importance of mental health to Black and Brown students of all ages by collaborating with schools and community leaders.
It is important to acknowledge that the lack of diversity within psychiatry is reflective of that among all physicians. This in part stems from the barriers to medical education that Black and Brown communities face. Those who start off with more resources or have parents who are physicians are at an advantage when trying to get into medical school. In fact, one in five medical students have a parent who is a physician4 and about three-fourths of students come from families whose income falls among the top two quintiles.5 Impoverished communities, which have a disproportionate share of Black and Brown people, cannot afford to take MCAT preparatory classes or to accept unpaid “resume building” opportunities. Many medical schools continue to place more weight on test scores and research/medical experiences, despite a shift to a more holistic review process. Institutions that have tried a different approach and accepted students from more diverse backgrounds may often overlook the challenges that URM students face while in medical school and fail to provide appropriate support resources.
The result is a failure to retain such students. A study conducted at Stony Brook (N.Y.) University showed that those underrepresented in medicine were six times more likely to get dismissed from medical school, and three times more likely to both withdraw or graduate beyond 4 years, compared with their White counterparts.6 This is a serious issue that needs to change on a structural and systemic level.
Any discussion of race and psychiatry must acknowledge psychiatry’s history of racism against Black and Brown communities to engage in racially informed discussions with our patients. Only then can we play a better role advocating against racism within the field in the future. Dating back to the 18th century, psychiatry has promoted ideologies that promote racism. Benjamin Rush, considered the “father of American Psychiatry,” believed that Black skin was a disease derived from leprosy called “negritude.” In the late 19th century, this twisted ideology continued with the invention of the term “drapetomania,” which was used to describe enslaved people who ran away as having a mental disorder.7 Black prisoners were subjected to experimental treatment with substances such as LSD and bulbocapnine to subdue them.8 This idea that minorities were dangerous and needed to be subdued translated into a higher number of schizophrenia diagnoses, particularly among Black men, as it was used as a tool to vilify them in the 1970s. Although schizophrenia is equally prevalent among Whites and non-Whites, Black people are four times more likely to be diagnosed, compared with their White counterparts, while Hispanics are three times more likely. Studies have shown that Black and Brown men are also more likely to receive higher doses of antipsychotics.9
Given this history, it is not surprising that Black and Brown representation within the field is lacking and that patients may be hesitant to share their feelings about race with us. While we can’t change history, we can take a stance condemning the harmful behavior of the past. The American Psychiatric Association issued an apology earlier this year to Black, Indigenous, and People of Color for its support in structural racism.10 This is a step in the right direction, but we need more than statements or performative actions. We need to amplify the voices of Black and Brown psychiatrists and patients, as well as highlight their current and past contributions to the field. While my educational experiences focused on the work of prominent White scholars, medical curricula should showcase the work of people like Solomon Carter Fuller, MD, a Black psychiatrist who was essential to understanding Alzheimer’s, or Joseph White, PhD, sometimes referred to as the “godfather of Black psychology.”11
At times, I have found myself witness to situations where colleagues make statements that not only do not condemn racism, but in fact encourage it. I have unfortunately heard some use the all-too-familiar rhetoric of reverse racism, such as: “They just assume I am racist because I am a White male” or “They’re being racist against me” or statements like “Don’t you think it is far-fetched to believe she was just sitting on a college campus doing nothing when the police were called?” Rhetoric such as this is problematic to the field of psychiatry and medicine as a whole – and only serves to further invalidate the feelings of our Black and Brown patients. We must increase exposure and education regarding racism to address this, especially the meaning of microaggressions, a concept many fail to understand.
Attention to the subject of racism has increased within medical schools and residency training programs in the wake of George Floyd’s death. However, most programs often make these lectures optional or only have one to two limited sessions. Furthermore, many do not make it mandatory for faculty to attend; they are arguably the most in need of this training given that they set the precedent of how to practice psychiatry. Some institutions have incorporated comprehensive antiracist curriculums into medical training. One model that has been successful is the Social Justice and Health Equity program within Yale University’s psychiatry residency. The curriculum has four tracks:
- Structural competency, which focuses on the mental health impact of extraclinical structures, for example a patient’s neighborhood and associated barriers of access.
- Human experience, which explores the interaction of patients and providers and how biases play a role.
- Advocacy, which teaches residents the written and oral skills to lobby for patient interests on a community and legislative level.
- History of psychiatry, which focuses on understanding psychiatry’s prior role in racism.
In each track, there are group discussions, cases led by faculty, and meetings with community leaders. Through this curriculum, residents learn about power, privilege, and how to interact with and advocate for patients in a way that promotes equity, rather than racial disparity.12,13 This is a model that other psychiatric residency programs can promote, emulate, and benefit from.
Educating ourselves will hopefully lead to a deeper introspection of how we interact with patients and if we are promoting antiracism through our attitude and actions. Reflecting on my own personal practice, I have noted that the interplay of race, mental health, and provider decision-making becomes particularly complex when dealing with situations in which a patient exhibits increased aggression or agitation. As a second-year psychiatric resident immersed in the inpatient world, I have become familiar with patients at higher risk and greater need. The first attempt toward de-escalation involves verbal cues without any other more intrusive measures. If that fails, intramuscular (IM) medications are typically considered. If a patient has a history of aggressive behavior, the threshold to use IM medications can decrease dramatically. This is mainly to protect ourselves and our nursing staff and to prioritize safety. While I understand this rationale, I wonder about the patient’s experience. What constitutes “aggressive” behavior? For patients who have had violence used against them because of their race or who have suffered from police brutality, having police present or threatening IM medications will increasingly trigger them and escalate the situation. The aftermath can deepen the distrust of psychiatry by Black and Brown people.
How do we then handle such situations in a way that both protects our staff from physical harm and protects our patients from racial trauma? While I don’t have a great answer, I think we can benefit from standardizing what we consider aggressive behavior and have specific criteria that patients need to exhibit before administering an IM medication. In addition, discussions with the team, including residents, nurses, and attending physicians, about how to address an emergent situation before it actually happens are essential. Specifically discussing the patient’s history and race and how it may affect the situation is not something to be shied away from. Lastly, in the event that an IM medication is administered and police are present, debriefing with the patient afterward is necessary. The patient may not be willing or able to listen to you or trust you, but taking accountability and acknowledging what happened, justified or not, is a part of the process of rebuilding rapport.
Both in the purview of the individual psychiatrist and the field of psychiatry as a whole, we need to examine our behavior and not be afraid to make changes for the betterment of our patients. We must learn to talk about race with our patients and in the process, advocate for more representation of Black and Brown psychiatrists, understanding the barriers faced by these communities when pursuing the medical field. We must educate ourselves on psychiatry’s history, and equip ourselves with knowledge and tools to promote antiracism and shape psychiatry’s future. We can then apply these very tools to challenging situations we may encounter daily with the ultimate goal of improving the mental health of our patients. This is the only way we will progress and ensure that psychiatry is an equitable, antiracist field. As Ibram X. Kendi, PhD, has written, “The heartbeat of antiracism is self-reflection, recognition, admission, and fundamentally self-critique.”
Dr. Malik is a second-year psychiatry resident at the University of California, San Diego. She has a background in policy and grassroots organizing through her time working at the National Coalition for the Homeless and the Women’s Law Project. Dr. Malik has no disclosures.
References
1. Wyse R et al. Acad Psychiatry. 2020 Oct;44(5):523-30.
2. Cooper LA et al. Ann Intern Med. 2003;139:907-15.
3. Pierre JM et al. Acad Psychiatry. 2017;41:226-32.
4. Hartocollis A. “Getting into med school without hard sciences.” New York Times. 2010 Jul 29.
5. AAMC. An updated look at the economic diversity of U.S. medical students. Analysis in Brief. 2018 Oct;18(5).
6. Rainey ML. How do we retain minority health professions students. In: Smedley BD et al. The right thing to do, the smart thing to do: Enhancing diversity in the health professions: Summary of the Symposium on Diversity in Health Professions in Honor of Herbert W. Nickens, M.D. Institute of Medicine. National Academies Press. 2001.
7. Geller J. “Structural racism in American psychiatry and APA: Part 1.” Psychiatric News. 2020 Jun 23.
8. Mohr CL and Gordon JE. Tulane: The emergence of a modern university, 1945-1980. Louisiana State University Press, Baton Rouge. 2001.
9. Metzl JM. The protest psychosis: How schizophrenia became a Black disease. Beacon Press. 2010.
10. APA’s apology to Black, indigenous and people of color for its support of structural racism in psychiatry. American Psychiatric Association. 2021 Jan 18.
11. Black pioneers in mental health. Mental Health America. 2021.
12. Belli B. For Yale’s emerging psychiatrists, confronting racism is in the curriculum. Yale News. 2020 Jul 30.
13. Jordan A and Jackson D. Social justice and health equity curriculum. Yale School of Medicine. 2019 Sep 24.
“I feel like my aggression is being racialized.” “Of course I wouldn’t call the cops if I felt like hurting myself. I’m Black.”
Those statements represent the heightened trauma our Black and Brown patients with mental health issues have been experiencing. In the wake of increasingly publicized police brutality against Black and Brown communities, the role race plays in mental health decompensation is evident. At this moment in time, we must continue to improve our understanding of the role race plays in psychiatric disorders. We must also ask ourselves: At times, does psychiatry worsen the traumas of the communities we serve?
Some psychiatrists are afraid to speak about race. They may believe it to be too “political.” But avoiding these necessary conversations perpetuates the trauma of those we treat. It suggests that physicians are ignorant of an issue at the forefront of patients’ mental health. Psychiatry, today, is primarily focused on the biological aspects of disease. We must not forget that psychiatry is biopsychosocial. It is imperative that psychiatrists have conversations about race – and its significance to our patients and their care.
Only 10.4% of psychiatrists in the United States comprise those considered underrepresented in medicine (URM). Yet, those very groups make up 32.6% of the U.S. population and are overrepresented in psychiatric hospitals.1 Many studies have shown that concordant racial backgrounds between patient and physician lead to a more positive patient experience2 and arguably, the subsequent potential for better health outcomes. Our efforts in addressing this disparity often fall short. URM applicants may be hesitant to join an institution where diversity is lacking or where they may be the only minority.3 While there is no simple solution, I propose that psychiatrists promote the importance of mental health to Black and Brown students of all ages by collaborating with schools and community leaders.
It is important to acknowledge that the lack of diversity within psychiatry is reflective of that among all physicians. This in part stems from the barriers to medical education that Black and Brown communities face. Those who start off with more resources or have parents who are physicians are at an advantage when trying to get into medical school. In fact, one in five medical students have a parent who is a physician4 and about three-fourths of students come from families whose income falls among the top two quintiles.5 Impoverished communities, which have a disproportionate share of Black and Brown people, cannot afford to take MCAT preparatory classes or to accept unpaid “resume building” opportunities. Many medical schools continue to place more weight on test scores and research/medical experiences, despite a shift to a more holistic review process. Institutions that have tried a different approach and accepted students from more diverse backgrounds may often overlook the challenges that URM students face while in medical school and fail to provide appropriate support resources.
The result is a failure to retain such students. A study conducted at Stony Brook (N.Y.) University showed that those underrepresented in medicine were six times more likely to get dismissed from medical school, and three times more likely to both withdraw or graduate beyond 4 years, compared with their White counterparts.6 This is a serious issue that needs to change on a structural and systemic level.
Any discussion of race and psychiatry must acknowledge psychiatry’s history of racism against Black and Brown communities to engage in racially informed discussions with our patients. Only then can we play a better role advocating against racism within the field in the future. Dating back to the 18th century, psychiatry has promoted ideologies that promote racism. Benjamin Rush, considered the “father of American Psychiatry,” believed that Black skin was a disease derived from leprosy called “negritude.” In the late 19th century, this twisted ideology continued with the invention of the term “drapetomania,” which was used to describe enslaved people who ran away as having a mental disorder.7 Black prisoners were subjected to experimental treatment with substances such as LSD and bulbocapnine to subdue them.8 This idea that minorities were dangerous and needed to be subdued translated into a higher number of schizophrenia diagnoses, particularly among Black men, as it was used as a tool to vilify them in the 1970s. Although schizophrenia is equally prevalent among Whites and non-Whites, Black people are four times more likely to be diagnosed, compared with their White counterparts, while Hispanics are three times more likely. Studies have shown that Black and Brown men are also more likely to receive higher doses of antipsychotics.9
Given this history, it is not surprising that Black and Brown representation within the field is lacking and that patients may be hesitant to share their feelings about race with us. While we can’t change history, we can take a stance condemning the harmful behavior of the past. The American Psychiatric Association issued an apology earlier this year to Black, Indigenous, and People of Color for its support in structural racism.10 This is a step in the right direction, but we need more than statements or performative actions. We need to amplify the voices of Black and Brown psychiatrists and patients, as well as highlight their current and past contributions to the field. While my educational experiences focused on the work of prominent White scholars, medical curricula should showcase the work of people like Solomon Carter Fuller, MD, a Black psychiatrist who was essential to understanding Alzheimer’s, or Joseph White, PhD, sometimes referred to as the “godfather of Black psychology.”11
At times, I have found myself witness to situations where colleagues make statements that not only do not condemn racism, but in fact encourage it. I have unfortunately heard some use the all-too-familiar rhetoric of reverse racism, such as: “They just assume I am racist because I am a White male” or “They’re being racist against me” or statements like “Don’t you think it is far-fetched to believe she was just sitting on a college campus doing nothing when the police were called?” Rhetoric such as this is problematic to the field of psychiatry and medicine as a whole – and only serves to further invalidate the feelings of our Black and Brown patients. We must increase exposure and education regarding racism to address this, especially the meaning of microaggressions, a concept many fail to understand.
Attention to the subject of racism has increased within medical schools and residency training programs in the wake of George Floyd’s death. However, most programs often make these lectures optional or only have one to two limited sessions. Furthermore, many do not make it mandatory for faculty to attend; they are arguably the most in need of this training given that they set the precedent of how to practice psychiatry. Some institutions have incorporated comprehensive antiracist curriculums into medical training. One model that has been successful is the Social Justice and Health Equity program within Yale University’s psychiatry residency. The curriculum has four tracks:
- Structural competency, which focuses on the mental health impact of extraclinical structures, for example a patient’s neighborhood and associated barriers of access.
- Human experience, which explores the interaction of patients and providers and how biases play a role.
- Advocacy, which teaches residents the written and oral skills to lobby for patient interests on a community and legislative level.
- History of psychiatry, which focuses on understanding psychiatry’s prior role in racism.
In each track, there are group discussions, cases led by faculty, and meetings with community leaders. Through this curriculum, residents learn about power, privilege, and how to interact with and advocate for patients in a way that promotes equity, rather than racial disparity.12,13 This is a model that other psychiatric residency programs can promote, emulate, and benefit from.
Educating ourselves will hopefully lead to a deeper introspection of how we interact with patients and if we are promoting antiracism through our attitude and actions. Reflecting on my own personal practice, I have noted that the interplay of race, mental health, and provider decision-making becomes particularly complex when dealing with situations in which a patient exhibits increased aggression or agitation. As a second-year psychiatric resident immersed in the inpatient world, I have become familiar with patients at higher risk and greater need. The first attempt toward de-escalation involves verbal cues without any other more intrusive measures. If that fails, intramuscular (IM) medications are typically considered. If a patient has a history of aggressive behavior, the threshold to use IM medications can decrease dramatically. This is mainly to protect ourselves and our nursing staff and to prioritize safety. While I understand this rationale, I wonder about the patient’s experience. What constitutes “aggressive” behavior? For patients who have had violence used against them because of their race or who have suffered from police brutality, having police present or threatening IM medications will increasingly trigger them and escalate the situation. The aftermath can deepen the distrust of psychiatry by Black and Brown people.
How do we then handle such situations in a way that both protects our staff from physical harm and protects our patients from racial trauma? While I don’t have a great answer, I think we can benefit from standardizing what we consider aggressive behavior and have specific criteria that patients need to exhibit before administering an IM medication. In addition, discussions with the team, including residents, nurses, and attending physicians, about how to address an emergent situation before it actually happens are essential. Specifically discussing the patient’s history and race and how it may affect the situation is not something to be shied away from. Lastly, in the event that an IM medication is administered and police are present, debriefing with the patient afterward is necessary. The patient may not be willing or able to listen to you or trust you, but taking accountability and acknowledging what happened, justified or not, is a part of the process of rebuilding rapport.
Both in the purview of the individual psychiatrist and the field of psychiatry as a whole, we need to examine our behavior and not be afraid to make changes for the betterment of our patients. We must learn to talk about race with our patients and in the process, advocate for more representation of Black and Brown psychiatrists, understanding the barriers faced by these communities when pursuing the medical field. We must educate ourselves on psychiatry’s history, and equip ourselves with knowledge and tools to promote antiracism and shape psychiatry’s future. We can then apply these very tools to challenging situations we may encounter daily with the ultimate goal of improving the mental health of our patients. This is the only way we will progress and ensure that psychiatry is an equitable, antiracist field. As Ibram X. Kendi, PhD, has written, “The heartbeat of antiracism is self-reflection, recognition, admission, and fundamentally self-critique.”
Dr. Malik is a second-year psychiatry resident at the University of California, San Diego. She has a background in policy and grassroots organizing through her time working at the National Coalition for the Homeless and the Women’s Law Project. Dr. Malik has no disclosures.
References
1. Wyse R et al. Acad Psychiatry. 2020 Oct;44(5):523-30.
2. Cooper LA et al. Ann Intern Med. 2003;139:907-15.
3. Pierre JM et al. Acad Psychiatry. 2017;41:226-32.
4. Hartocollis A. “Getting into med school without hard sciences.” New York Times. 2010 Jul 29.
5. AAMC. An updated look at the economic diversity of U.S. medical students. Analysis in Brief. 2018 Oct;18(5).
6. Rainey ML. How do we retain minority health professions students. In: Smedley BD et al. The right thing to do, the smart thing to do: Enhancing diversity in the health professions: Summary of the Symposium on Diversity in Health Professions in Honor of Herbert W. Nickens, M.D. Institute of Medicine. National Academies Press. 2001.
7. Geller J. “Structural racism in American psychiatry and APA: Part 1.” Psychiatric News. 2020 Jun 23.
8. Mohr CL and Gordon JE. Tulane: The emergence of a modern university, 1945-1980. Louisiana State University Press, Baton Rouge. 2001.
9. Metzl JM. The protest psychosis: How schizophrenia became a Black disease. Beacon Press. 2010.
10. APA’s apology to Black, indigenous and people of color for its support of structural racism in psychiatry. American Psychiatric Association. 2021 Jan 18.
11. Black pioneers in mental health. Mental Health America. 2021.
12. Belli B. For Yale’s emerging psychiatrists, confronting racism is in the curriculum. Yale News. 2020 Jul 30.
13. Jordan A and Jackson D. Social justice and health equity curriculum. Yale School of Medicine. 2019 Sep 24.
“I feel like my aggression is being racialized.” “Of course I wouldn’t call the cops if I felt like hurting myself. I’m Black.”
Those statements represent the heightened trauma our Black and Brown patients with mental health issues have been experiencing. In the wake of increasingly publicized police brutality against Black and Brown communities, the role race plays in mental health decompensation is evident. At this moment in time, we must continue to improve our understanding of the role race plays in psychiatric disorders. We must also ask ourselves: At times, does psychiatry worsen the traumas of the communities we serve?
Some psychiatrists are afraid to speak about race. They may believe it to be too “political.” But avoiding these necessary conversations perpetuates the trauma of those we treat. It suggests that physicians are ignorant of an issue at the forefront of patients’ mental health. Psychiatry, today, is primarily focused on the biological aspects of disease. We must not forget that psychiatry is biopsychosocial. It is imperative that psychiatrists have conversations about race – and its significance to our patients and their care.
Only 10.4% of psychiatrists in the United States comprise those considered underrepresented in medicine (URM). Yet, those very groups make up 32.6% of the U.S. population and are overrepresented in psychiatric hospitals.1 Many studies have shown that concordant racial backgrounds between patient and physician lead to a more positive patient experience2 and arguably, the subsequent potential for better health outcomes. Our efforts in addressing this disparity often fall short. URM applicants may be hesitant to join an institution where diversity is lacking or where they may be the only minority.3 While there is no simple solution, I propose that psychiatrists promote the importance of mental health to Black and Brown students of all ages by collaborating with schools and community leaders.
It is important to acknowledge that the lack of diversity within psychiatry is reflective of that among all physicians. This in part stems from the barriers to medical education that Black and Brown communities face. Those who start off with more resources or have parents who are physicians are at an advantage when trying to get into medical school. In fact, one in five medical students have a parent who is a physician4 and about three-fourths of students come from families whose income falls among the top two quintiles.5 Impoverished communities, which have a disproportionate share of Black and Brown people, cannot afford to take MCAT preparatory classes or to accept unpaid “resume building” opportunities. Many medical schools continue to place more weight on test scores and research/medical experiences, despite a shift to a more holistic review process. Institutions that have tried a different approach and accepted students from more diverse backgrounds may often overlook the challenges that URM students face while in medical school and fail to provide appropriate support resources.
The result is a failure to retain such students. A study conducted at Stony Brook (N.Y.) University showed that those underrepresented in medicine were six times more likely to get dismissed from medical school, and three times more likely to both withdraw or graduate beyond 4 years, compared with their White counterparts.6 This is a serious issue that needs to change on a structural and systemic level.
Any discussion of race and psychiatry must acknowledge psychiatry’s history of racism against Black and Brown communities to engage in racially informed discussions with our patients. Only then can we play a better role advocating against racism within the field in the future. Dating back to the 18th century, psychiatry has promoted ideologies that promote racism. Benjamin Rush, considered the “father of American Psychiatry,” believed that Black skin was a disease derived from leprosy called “negritude.” In the late 19th century, this twisted ideology continued with the invention of the term “drapetomania,” which was used to describe enslaved people who ran away as having a mental disorder.7 Black prisoners were subjected to experimental treatment with substances such as LSD and bulbocapnine to subdue them.8 This idea that minorities were dangerous and needed to be subdued translated into a higher number of schizophrenia diagnoses, particularly among Black men, as it was used as a tool to vilify them in the 1970s. Although schizophrenia is equally prevalent among Whites and non-Whites, Black people are four times more likely to be diagnosed, compared with their White counterparts, while Hispanics are three times more likely. Studies have shown that Black and Brown men are also more likely to receive higher doses of antipsychotics.9
Given this history, it is not surprising that Black and Brown representation within the field is lacking and that patients may be hesitant to share their feelings about race with us. While we can’t change history, we can take a stance condemning the harmful behavior of the past. The American Psychiatric Association issued an apology earlier this year to Black, Indigenous, and People of Color for its support in structural racism.10 This is a step in the right direction, but we need more than statements or performative actions. We need to amplify the voices of Black and Brown psychiatrists and patients, as well as highlight their current and past contributions to the field. While my educational experiences focused on the work of prominent White scholars, medical curricula should showcase the work of people like Solomon Carter Fuller, MD, a Black psychiatrist who was essential to understanding Alzheimer’s, or Joseph White, PhD, sometimes referred to as the “godfather of Black psychology.”11
At times, I have found myself witness to situations where colleagues make statements that not only do not condemn racism, but in fact encourage it. I have unfortunately heard some use the all-too-familiar rhetoric of reverse racism, such as: “They just assume I am racist because I am a White male” or “They’re being racist against me” or statements like “Don’t you think it is far-fetched to believe she was just sitting on a college campus doing nothing when the police were called?” Rhetoric such as this is problematic to the field of psychiatry and medicine as a whole – and only serves to further invalidate the feelings of our Black and Brown patients. We must increase exposure and education regarding racism to address this, especially the meaning of microaggressions, a concept many fail to understand.
Attention to the subject of racism has increased within medical schools and residency training programs in the wake of George Floyd’s death. However, most programs often make these lectures optional or only have one to two limited sessions. Furthermore, many do not make it mandatory for faculty to attend; they are arguably the most in need of this training given that they set the precedent of how to practice psychiatry. Some institutions have incorporated comprehensive antiracist curriculums into medical training. One model that has been successful is the Social Justice and Health Equity program within Yale University’s psychiatry residency. The curriculum has four tracks:
- Structural competency, which focuses on the mental health impact of extraclinical structures, for example a patient’s neighborhood and associated barriers of access.
- Human experience, which explores the interaction of patients and providers and how biases play a role.
- Advocacy, which teaches residents the written and oral skills to lobby for patient interests on a community and legislative level.
- History of psychiatry, which focuses on understanding psychiatry’s prior role in racism.
In each track, there are group discussions, cases led by faculty, and meetings with community leaders. Through this curriculum, residents learn about power, privilege, and how to interact with and advocate for patients in a way that promotes equity, rather than racial disparity.12,13 This is a model that other psychiatric residency programs can promote, emulate, and benefit from.
Educating ourselves will hopefully lead to a deeper introspection of how we interact with patients and if we are promoting antiracism through our attitude and actions. Reflecting on my own personal practice, I have noted that the interplay of race, mental health, and provider decision-making becomes particularly complex when dealing with situations in which a patient exhibits increased aggression or agitation. As a second-year psychiatric resident immersed in the inpatient world, I have become familiar with patients at higher risk and greater need. The first attempt toward de-escalation involves verbal cues without any other more intrusive measures. If that fails, intramuscular (IM) medications are typically considered. If a patient has a history of aggressive behavior, the threshold to use IM medications can decrease dramatically. This is mainly to protect ourselves and our nursing staff and to prioritize safety. While I understand this rationale, I wonder about the patient’s experience. What constitutes “aggressive” behavior? For patients who have had violence used against them because of their race or who have suffered from police brutality, having police present or threatening IM medications will increasingly trigger them and escalate the situation. The aftermath can deepen the distrust of psychiatry by Black and Brown people.
How do we then handle such situations in a way that both protects our staff from physical harm and protects our patients from racial trauma? While I don’t have a great answer, I think we can benefit from standardizing what we consider aggressive behavior and have specific criteria that patients need to exhibit before administering an IM medication. In addition, discussions with the team, including residents, nurses, and attending physicians, about how to address an emergent situation before it actually happens are essential. Specifically discussing the patient’s history and race and how it may affect the situation is not something to be shied away from. Lastly, in the event that an IM medication is administered and police are present, debriefing with the patient afterward is necessary. The patient may not be willing or able to listen to you or trust you, but taking accountability and acknowledging what happened, justified or not, is a part of the process of rebuilding rapport.
Both in the purview of the individual psychiatrist and the field of psychiatry as a whole, we need to examine our behavior and not be afraid to make changes for the betterment of our patients. We must learn to talk about race with our patients and in the process, advocate for more representation of Black and Brown psychiatrists, understanding the barriers faced by these communities when pursuing the medical field. We must educate ourselves on psychiatry’s history, and equip ourselves with knowledge and tools to promote antiracism and shape psychiatry’s future. We can then apply these very tools to challenging situations we may encounter daily with the ultimate goal of improving the mental health of our patients. This is the only way we will progress and ensure that psychiatry is an equitable, antiracist field. As Ibram X. Kendi, PhD, has written, “The heartbeat of antiracism is self-reflection, recognition, admission, and fundamentally self-critique.”
Dr. Malik is a second-year psychiatry resident at the University of California, San Diego. She has a background in policy and grassroots organizing through her time working at the National Coalition for the Homeless and the Women’s Law Project. Dr. Malik has no disclosures.
References
1. Wyse R et al. Acad Psychiatry. 2020 Oct;44(5):523-30.
2. Cooper LA et al. Ann Intern Med. 2003;139:907-15.
3. Pierre JM et al. Acad Psychiatry. 2017;41:226-32.
4. Hartocollis A. “Getting into med school without hard sciences.” New York Times. 2010 Jul 29.
5. AAMC. An updated look at the economic diversity of U.S. medical students. Analysis in Brief. 2018 Oct;18(5).
6. Rainey ML. How do we retain minority health professions students. In: Smedley BD et al. The right thing to do, the smart thing to do: Enhancing diversity in the health professions: Summary of the Symposium on Diversity in Health Professions in Honor of Herbert W. Nickens, M.D. Institute of Medicine. National Academies Press. 2001.
7. Geller J. “Structural racism in American psychiatry and APA: Part 1.” Psychiatric News. 2020 Jun 23.
8. Mohr CL and Gordon JE. Tulane: The emergence of a modern university, 1945-1980. Louisiana State University Press, Baton Rouge. 2001.
9. Metzl JM. The protest psychosis: How schizophrenia became a Black disease. Beacon Press. 2010.
10. APA’s apology to Black, indigenous and people of color for its support of structural racism in psychiatry. American Psychiatric Association. 2021 Jan 18.
11. Black pioneers in mental health. Mental Health America. 2021.
12. Belli B. For Yale’s emerging psychiatrists, confronting racism is in the curriculum. Yale News. 2020 Jul 30.
13. Jordan A and Jackson D. Social justice and health equity curriculum. Yale School of Medicine. 2019 Sep 24.













