User login
Six healthy lifestyle habits linked to slowed memory decline
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.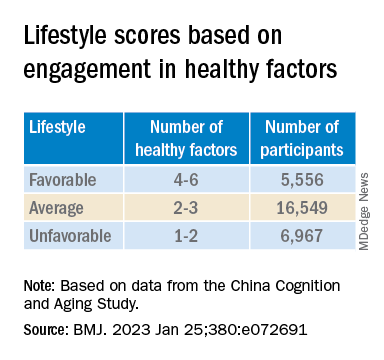
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.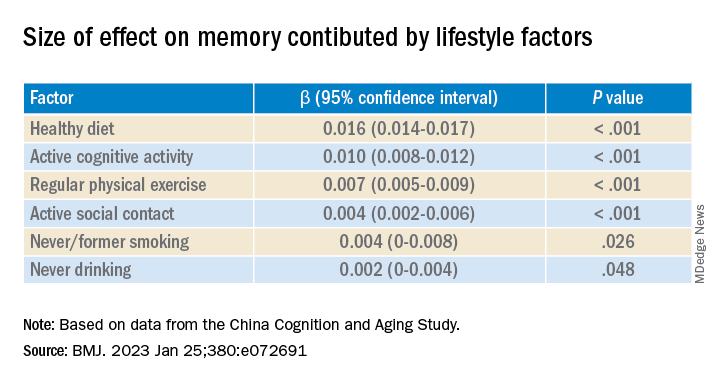
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
Investigators found that a healthy diet, cognitive activity, regular physical exercise, not smoking, and abstaining from alcohol were significantly linked to slowed cognitive decline irrespective of APOE4 status.
After adjusting for health and socioeconomic factors, investigators found that each individual healthy behavior was associated with a slower-than-average decline in memory over a decade. A healthy diet emerged as the strongest deterrent, followed by cognitive activity and physical exercise.
“A healthy lifestyle is associated with slower memory decline, even in the presence of the APOE4 allele,” study investigators led by Jianping Jia, MD, PhD, of the Innovation Center for Neurological Disorders and the department of neurology, Xuan Wu Hospital, Capital Medical University, Beijing, write.
“This study might offer important information to protect older adults against memory decline,” they add.
The study was published online in the BMJ.
Preventing memory decline
Memory “continuously declines as people age,” but age-related memory decline is not necessarily a prodrome of dementia and can “merely be senescent forgetfulness,” the investigators note. This can be “reversed or [can] become stable,” instead of progressing to a pathologic state.
Factors affecting memory include aging, APOE4 genotype, chronic diseases, and lifestyle patterns, with lifestyle “receiving increasing attention as a modifiable behavior.”
Nevertheless, few studies have focused on the impact of lifestyle on memory, and those that have are mostly cross-sectional and also “did not consider the interaction between a healthy lifestyle and genetic risk,” the researchers note.
To investigate, the researchers conducted a longitudinal study, known as the China Cognition and Aging Study, that considered genetic risk as well as lifestyle factors.
The study began in 2009 and concluded in 2019. Participants were evaluated and underwent neuropsychological testing in 2012, 2014, 2016, and at the study’s conclusion.
Participants (n = 29,072; mean [SD] age, 72.23 [6.61] years; 48.54% women; 20.43% APOE4 carriers) were required to have normal cognitive function at baseline. Data on those whose condition progressed to mild cognitive impairment (MCI) or dementia during the follow-up period were excluded after their diagnosis.
The Mini–Mental State Examination was used to assess global cognitive function. Memory function was assessed using the World Health Organization/University of California, Los Angeles Auditory Verbal Learning Test.
“Lifestyle” consisted of six modifiable factors: physical exercise (weekly frequency and total time), smoking (current, former, or never-smokers), alcohol consumption (never drank, drank occasionally, low to excess drinking, and heavy drinking), diet (daily intake of 12 food items: fruits, vegetables, fish, meat, dairy products, salt, oil, eggs, cereals, legumes, nuts, tea), cognitive activity (writing, reading, playing cards, mahjong, other games), and social contact (participating in meetings, attending parties, visiting friends/relatives, traveling, chatting online).
Participants’ lifestyles were scored on the basis of the number of healthy factors they engaged in.
Participants were also stratified by APOE genotype into APOE4 carriers and noncarriers.
Demographic and other items of health information, including the presence of medical illness, were used as covariates. The researchers also included the “learning effect of each participant as a covariate, due to repeated cognitive assessments.”
Important for public health
During the 10-year period, 7,164 participants died, and 3,567 stopped participating.
Participants in the favorable and average groups showed slower memory decline per increased year of age (0.007 [0.005-0.009], P < .001; and 0.002 [0 .000-0.003], P = .033 points higher, respectively), compared with those in the unfavorable group.
Healthy diet had the strongest protective effect on memory.
Memory decline occurred faster in APOE4 vesus non-APOE4 carriers (0.002 points/year [95% confidence interval, 0.001-0.003]; P = .007).
But APOE4 carriers with favorable and average lifestyles showed slower memory decline (0.027 [0.023-0.031] and 0.014 [0.010-0.019], respectively), compared with those with unfavorable lifestyles. Similar findings were obtained in non-APOE4 carriers.
Those with favorable or average lifestyle were respectively almost 90% and 30% less likely to develop dementia or MCI, compared with those with an unfavorable lifestyle.
The authors acknowledge the study’s limitations, including its observational design and the potential for measurement errors, owing to self-reporting of lifestyle factors. Additionally, some participants did not return for follow-up evaluations, leading to potential selection bias.
Nevertheless, the findings “might offer important information for public health to protect older [people] against memory decline,” they note – especially since the study “provides evidence that these effects also include individuals with the APOE4 allele.”
‘Important, encouraging’ research
In a comment, Severine Sabia, PhD, a senior researcher at the Université Paris Cité, INSERM Institut National de la Santé et de la Recherche Medicalé, France, called the findings “important and encouraging.”
However, said Dr. Sabia, who was not involved with the study, “there remain important research questions that need to be investigated in order to identify key behaviors: which combination, the cutoff of risk, and when to intervene.”
Future research on prevention “should examine a wider range of possible risk factors” and should also “identify specific exposures associated with the greatest risk, while also considering the risk threshold and age at exposure for each one.”
In an accompanying editorial, Dr. Sabia and co-author Archana Singh-Manoux, PhD, note that the risk of cognitive decline and dementia are probably determined by multiple factors.
They liken it to the “multifactorial risk paradigm introduced by the Framingham study,” which has “led to a substantial reduction in cardiovascular disease.” A similar approach could be used with dementia prevention, they suggest.
The authors received support from the Xuanwu Hospital of Capital Medical University for the submitted work. One of the authors received a grant from the French National Research Agency. The other authors have disclosed no relevant financial relationships. Dr. Sabia received grant funding from the French National Research Agency. Dr. Singh-Manoux received grants from the National Institute on Aging of the National Institutes of Health.
A version of this article first appeared on Medscape.com.
FROM THE BMJ
Novel nomogram distinguishes pneumonias
A model incorporating factors such as lymphocytes and lung lesions differentiated adenovirus pneumonias from Chlamydia psittaci (CPP) in a multicenter study of nearly 200 individuals.
Symptoms of pneumonia caused by CPP are often confused with other respiratory infections, particularly adenovirus pneumonia (AVP), which can delay correct diagnosis and impact treatment, Yi Li, MD, of Xiangya Hospital, Central South University, Changsha, China, and colleagues wrote. Detailed comparisons of the two conditions are lacking.
In a retrospective study published in the International Journal of Infectious Diseases, the researchers examined laboratory, clinical, and radiological differences and created a nomogram to distinguish CPP from AVP. The study population included 78 adults with CPP and 102 with AVP who were seen at a single center in China. The mean ages of the CPP and AVP patients were 61.0 years and 38.5 years, and 57.7% men and 91.2% men, respectively. Patients with CPP were significantly more likely to have hypertension and diabetes at baseline, compared with the AVP group.
The primary outcome was 30-day mortality after hospital admission, which was 10.3% and 14.7% for the CPP and AVP patients, respectively (P = 0.376). However, the incidence of cardiac injury was significantly higher in AVP patients versus those with CPP (48.0% vs. 11.5%; P < 0.001).
In a multivariate analysis, age, sex, nervous system symptoms, lymphocyte count, C-reactive protein level (CRP), and bilateral lung lesions were risk factors for CPP. The researchers combined these factors into a nomogram that showed a concordance value of 0.949 for differentiating between the CPP and AVP groups.
Overall, CPP patients were older, had more nervous system symptoms, and had higher CRP levels, compared with patients with AVP, who were more likely to be men and to have higher lymphocyte percentages and more bilateral lung lesions on chest imaging.
The current study is the first known to provide a way to distinguish CPP and AVP, the researchers wrote. “The antibiotic treatments, prognoses, and life support measures of CPP and AVP are considerably different. Therefore, differentiating the two diseases through early identification of specific clinical characteristics is vital.”
The findings were limited by several factors including the small sample size, retrospective design, and the use of mNGS to diagnose CPP in the absence of standard clinical diagnostic kits, which may have resulted in underestimated CPP incidence, the researchers noted.
However, “the nomogram we established combines patient data on age, sex, and readily available laboratory results to reasonably predict CPP, thus making rapid and direct diagnosis possible,” they said.
The study was supported by the Key R&D Program of Hunan Province, Project Program of National Clinical Research Center for Geriatric Disorders, National Natural Science Foundation of China, Hunan Natural Science Youth Foundation, and the national key clinical specialist construction programs of China. The researchers had no financial conflicts to disclose.
A model incorporating factors such as lymphocytes and lung lesions differentiated adenovirus pneumonias from Chlamydia psittaci (CPP) in a multicenter study of nearly 200 individuals.
Symptoms of pneumonia caused by CPP are often confused with other respiratory infections, particularly adenovirus pneumonia (AVP), which can delay correct diagnosis and impact treatment, Yi Li, MD, of Xiangya Hospital, Central South University, Changsha, China, and colleagues wrote. Detailed comparisons of the two conditions are lacking.
In a retrospective study published in the International Journal of Infectious Diseases, the researchers examined laboratory, clinical, and radiological differences and created a nomogram to distinguish CPP from AVP. The study population included 78 adults with CPP and 102 with AVP who were seen at a single center in China. The mean ages of the CPP and AVP patients were 61.0 years and 38.5 years, and 57.7% men and 91.2% men, respectively. Patients with CPP were significantly more likely to have hypertension and diabetes at baseline, compared with the AVP group.
The primary outcome was 30-day mortality after hospital admission, which was 10.3% and 14.7% for the CPP and AVP patients, respectively (P = 0.376). However, the incidence of cardiac injury was significantly higher in AVP patients versus those with CPP (48.0% vs. 11.5%; P < 0.001).
In a multivariate analysis, age, sex, nervous system symptoms, lymphocyte count, C-reactive protein level (CRP), and bilateral lung lesions were risk factors for CPP. The researchers combined these factors into a nomogram that showed a concordance value of 0.949 for differentiating between the CPP and AVP groups.
Overall, CPP patients were older, had more nervous system symptoms, and had higher CRP levels, compared with patients with AVP, who were more likely to be men and to have higher lymphocyte percentages and more bilateral lung lesions on chest imaging.
The current study is the first known to provide a way to distinguish CPP and AVP, the researchers wrote. “The antibiotic treatments, prognoses, and life support measures of CPP and AVP are considerably different. Therefore, differentiating the two diseases through early identification of specific clinical characteristics is vital.”
The findings were limited by several factors including the small sample size, retrospective design, and the use of mNGS to diagnose CPP in the absence of standard clinical diagnostic kits, which may have resulted in underestimated CPP incidence, the researchers noted.
However, “the nomogram we established combines patient data on age, sex, and readily available laboratory results to reasonably predict CPP, thus making rapid and direct diagnosis possible,” they said.
The study was supported by the Key R&D Program of Hunan Province, Project Program of National Clinical Research Center for Geriatric Disorders, National Natural Science Foundation of China, Hunan Natural Science Youth Foundation, and the national key clinical specialist construction programs of China. The researchers had no financial conflicts to disclose.
A model incorporating factors such as lymphocytes and lung lesions differentiated adenovirus pneumonias from Chlamydia psittaci (CPP) in a multicenter study of nearly 200 individuals.
Symptoms of pneumonia caused by CPP are often confused with other respiratory infections, particularly adenovirus pneumonia (AVP), which can delay correct diagnosis and impact treatment, Yi Li, MD, of Xiangya Hospital, Central South University, Changsha, China, and colleagues wrote. Detailed comparisons of the two conditions are lacking.
In a retrospective study published in the International Journal of Infectious Diseases, the researchers examined laboratory, clinical, and radiological differences and created a nomogram to distinguish CPP from AVP. The study population included 78 adults with CPP and 102 with AVP who were seen at a single center in China. The mean ages of the CPP and AVP patients were 61.0 years and 38.5 years, and 57.7% men and 91.2% men, respectively. Patients with CPP were significantly more likely to have hypertension and diabetes at baseline, compared with the AVP group.
The primary outcome was 30-day mortality after hospital admission, which was 10.3% and 14.7% for the CPP and AVP patients, respectively (P = 0.376). However, the incidence of cardiac injury was significantly higher in AVP patients versus those with CPP (48.0% vs. 11.5%; P < 0.001).
In a multivariate analysis, age, sex, nervous system symptoms, lymphocyte count, C-reactive protein level (CRP), and bilateral lung lesions were risk factors for CPP. The researchers combined these factors into a nomogram that showed a concordance value of 0.949 for differentiating between the CPP and AVP groups.
Overall, CPP patients were older, had more nervous system symptoms, and had higher CRP levels, compared with patients with AVP, who were more likely to be men and to have higher lymphocyte percentages and more bilateral lung lesions on chest imaging.
The current study is the first known to provide a way to distinguish CPP and AVP, the researchers wrote. “The antibiotic treatments, prognoses, and life support measures of CPP and AVP are considerably different. Therefore, differentiating the two diseases through early identification of specific clinical characteristics is vital.”
The findings were limited by several factors including the small sample size, retrospective design, and the use of mNGS to diagnose CPP in the absence of standard clinical diagnostic kits, which may have resulted in underestimated CPP incidence, the researchers noted.
However, “the nomogram we established combines patient data on age, sex, and readily available laboratory results to reasonably predict CPP, thus making rapid and direct diagnosis possible,” they said.
The study was supported by the Key R&D Program of Hunan Province, Project Program of National Clinical Research Center for Geriatric Disorders, National Natural Science Foundation of China, Hunan Natural Science Youth Foundation, and the national key clinical specialist construction programs of China. The researchers had no financial conflicts to disclose.
FROM THE INTERNATIONAL JOURNAL OF INFECTIOUS DISEASES
Long COVID affecting more than one-third of college students, faculty
With a median age of 23 years, the study is unique for evaluating mostly healthy, young adults and for its rare look at long COVID in a university community.
The more symptoms during a bout with COVID, the greater the risk for long COVID, the researchers found. That lines up with previous studies. Also, the more vaccinations and booster shots against SARS-CoV-2, the virus that causes COVID, the lower the long COVID risk.
Women were more likely than men to be affected. Current or prior smoking, seeking medical care for COVID, and receiving antibody treatment also were linked to higher chances for developing long COVID.
Lead author Megan Landry, DrPH, MPH, and colleagues were already assessing students, staff, and faculty at George Washington University, Washington, who tested positive for COVID. Then they started seeing symptoms that lasted 28 days or more after their 10-day isolation period.
“We were starting to recognize that individuals ... were still having symptoms longer than the typical isolation period,” said Dr. Landry. So they developed a questionnaire to figure out the how long these symptoms last and how many people are affected by them.
The list of potential symptoms was long and included trouble thinking, fatigue, loss of smell or taste, shortness of breath, and more.
The study was published online in Emerging Infectious Diseases. Results are based on records and responses from 1,388 students, faculty, and staff from July 2021 to March 2022.
People had a median of four long COVID symptoms, about 63% were women, and 56% were non-Hispanic White. About three-quarters were students and the remainder were faculty and staff.
The finding that 36% of people with a history of COVID reported long COVID symptoms did not surprise Dr. Landry.
“Based on the literature that’s currently out there, it ranges from a 10% to an 80% prevalence of long COVID,” she said. “We kind of figured that we would fall somewhere in there.”
In contrast, that figure seemed high to Eric Topol, MD.
“That’s really high,” said Dr. Topol, founder and director of the Scripps Research Translational Institute in La Jolla, Calif. He added most studies estimate that about 10% of people with a history of acute infection develop long COVID.
Even at 10%, which could be an underestimate, that’s a lot of affected people globally.
“At least 65 million individuals around the world have long COVID, based on a conservative estimated incidence of 10% of infected people and more than 651 million documented COVID-19 cases worldwide; the number is likely much higher due to many undocumented cases,” Dr. Topol and colleagues wrote in a long COVID review article published in Nature Reviews Microbiology.
About 30% of study participants were fully vaccinated with an initial vaccine series, 42% had received a booster dose, and 29% were not fully vaccinated at the time of their first positive test for COVID. Those who were not fully vaccinated were significantly more likely to report symptoms of long COVID.
“I know a lot of people wish they could put COVID on the back burner or brush it under the rug, but COVID is still a real thing. We need to continue supporting vaccines and boosters and make sure people are up to date. Not only for COVID, but for flu as well,” Dr. Topol said
Research continues
“Long COVID is still evolving and we continue to learn more about it every day,” Landry said. “It’s just so new and there are still a lot of unknowns. That’s why it’s important to get this information out.”
People with long COVID often have a hard time with occupational, educational, social, or personal activities, compared with before COVID, with effects that can last for more than 6 months, the authors noted.
“I think across the board, universities in general need to consider the possibility of folks on their campuses are having symptoms of long COVID,” Dr. Landry said.
Moving forward, Dr. Landry and colleagues would like to continue investigating long COVID. For example, in the current study, they did not ask about severity of symptoms or how the symptoms affected daily functioning.
“I would like to continue this and dive deeper into how disruptive their symptoms of long COVID are to their everyday studying, teaching, or their activities to keeping a university running,” Dr. Landry said.
A version of this article originally appeared on WebMD.com.
With a median age of 23 years, the study is unique for evaluating mostly healthy, young adults and for its rare look at long COVID in a university community.
The more symptoms during a bout with COVID, the greater the risk for long COVID, the researchers found. That lines up with previous studies. Also, the more vaccinations and booster shots against SARS-CoV-2, the virus that causes COVID, the lower the long COVID risk.
Women were more likely than men to be affected. Current or prior smoking, seeking medical care for COVID, and receiving antibody treatment also were linked to higher chances for developing long COVID.
Lead author Megan Landry, DrPH, MPH, and colleagues were already assessing students, staff, and faculty at George Washington University, Washington, who tested positive for COVID. Then they started seeing symptoms that lasted 28 days or more after their 10-day isolation period.
“We were starting to recognize that individuals ... were still having symptoms longer than the typical isolation period,” said Dr. Landry. So they developed a questionnaire to figure out the how long these symptoms last and how many people are affected by them.
The list of potential symptoms was long and included trouble thinking, fatigue, loss of smell or taste, shortness of breath, and more.
The study was published online in Emerging Infectious Diseases. Results are based on records and responses from 1,388 students, faculty, and staff from July 2021 to March 2022.
People had a median of four long COVID symptoms, about 63% were women, and 56% were non-Hispanic White. About three-quarters were students and the remainder were faculty and staff.
The finding that 36% of people with a history of COVID reported long COVID symptoms did not surprise Dr. Landry.
“Based on the literature that’s currently out there, it ranges from a 10% to an 80% prevalence of long COVID,” she said. “We kind of figured that we would fall somewhere in there.”
In contrast, that figure seemed high to Eric Topol, MD.
“That’s really high,” said Dr. Topol, founder and director of the Scripps Research Translational Institute in La Jolla, Calif. He added most studies estimate that about 10% of people with a history of acute infection develop long COVID.
Even at 10%, which could be an underestimate, that’s a lot of affected people globally.
“At least 65 million individuals around the world have long COVID, based on a conservative estimated incidence of 10% of infected people and more than 651 million documented COVID-19 cases worldwide; the number is likely much higher due to many undocumented cases,” Dr. Topol and colleagues wrote in a long COVID review article published in Nature Reviews Microbiology.
About 30% of study participants were fully vaccinated with an initial vaccine series, 42% had received a booster dose, and 29% were not fully vaccinated at the time of their first positive test for COVID. Those who were not fully vaccinated were significantly more likely to report symptoms of long COVID.
“I know a lot of people wish they could put COVID on the back burner or brush it under the rug, but COVID is still a real thing. We need to continue supporting vaccines and boosters and make sure people are up to date. Not only for COVID, but for flu as well,” Dr. Topol said
Research continues
“Long COVID is still evolving and we continue to learn more about it every day,” Landry said. “It’s just so new and there are still a lot of unknowns. That’s why it’s important to get this information out.”
People with long COVID often have a hard time with occupational, educational, social, or personal activities, compared with before COVID, with effects that can last for more than 6 months, the authors noted.
“I think across the board, universities in general need to consider the possibility of folks on their campuses are having symptoms of long COVID,” Dr. Landry said.
Moving forward, Dr. Landry and colleagues would like to continue investigating long COVID. For example, in the current study, they did not ask about severity of symptoms or how the symptoms affected daily functioning.
“I would like to continue this and dive deeper into how disruptive their symptoms of long COVID are to their everyday studying, teaching, or their activities to keeping a university running,” Dr. Landry said.
A version of this article originally appeared on WebMD.com.
With a median age of 23 years, the study is unique for evaluating mostly healthy, young adults and for its rare look at long COVID in a university community.
The more symptoms during a bout with COVID, the greater the risk for long COVID, the researchers found. That lines up with previous studies. Also, the more vaccinations and booster shots against SARS-CoV-2, the virus that causes COVID, the lower the long COVID risk.
Women were more likely than men to be affected. Current or prior smoking, seeking medical care for COVID, and receiving antibody treatment also were linked to higher chances for developing long COVID.
Lead author Megan Landry, DrPH, MPH, and colleagues were already assessing students, staff, and faculty at George Washington University, Washington, who tested positive for COVID. Then they started seeing symptoms that lasted 28 days or more after their 10-day isolation period.
“We were starting to recognize that individuals ... were still having symptoms longer than the typical isolation period,” said Dr. Landry. So they developed a questionnaire to figure out the how long these symptoms last and how many people are affected by them.
The list of potential symptoms was long and included trouble thinking, fatigue, loss of smell or taste, shortness of breath, and more.
The study was published online in Emerging Infectious Diseases. Results are based on records and responses from 1,388 students, faculty, and staff from July 2021 to March 2022.
People had a median of four long COVID symptoms, about 63% were women, and 56% were non-Hispanic White. About three-quarters were students and the remainder were faculty and staff.
The finding that 36% of people with a history of COVID reported long COVID symptoms did not surprise Dr. Landry.
“Based on the literature that’s currently out there, it ranges from a 10% to an 80% prevalence of long COVID,” she said. “We kind of figured that we would fall somewhere in there.”
In contrast, that figure seemed high to Eric Topol, MD.
“That’s really high,” said Dr. Topol, founder and director of the Scripps Research Translational Institute in La Jolla, Calif. He added most studies estimate that about 10% of people with a history of acute infection develop long COVID.
Even at 10%, which could be an underestimate, that’s a lot of affected people globally.
“At least 65 million individuals around the world have long COVID, based on a conservative estimated incidence of 10% of infected people and more than 651 million documented COVID-19 cases worldwide; the number is likely much higher due to many undocumented cases,” Dr. Topol and colleagues wrote in a long COVID review article published in Nature Reviews Microbiology.
About 30% of study participants were fully vaccinated with an initial vaccine series, 42% had received a booster dose, and 29% were not fully vaccinated at the time of their first positive test for COVID. Those who were not fully vaccinated were significantly more likely to report symptoms of long COVID.
“I know a lot of people wish they could put COVID on the back burner or brush it under the rug, but COVID is still a real thing. We need to continue supporting vaccines and boosters and make sure people are up to date. Not only for COVID, but for flu as well,” Dr. Topol said
Research continues
“Long COVID is still evolving and we continue to learn more about it every day,” Landry said. “It’s just so new and there are still a lot of unknowns. That’s why it’s important to get this information out.”
People with long COVID often have a hard time with occupational, educational, social, or personal activities, compared with before COVID, with effects that can last for more than 6 months, the authors noted.
“I think across the board, universities in general need to consider the possibility of folks on their campuses are having symptoms of long COVID,” Dr. Landry said.
Moving forward, Dr. Landry and colleagues would like to continue investigating long COVID. For example, in the current study, they did not ask about severity of symptoms or how the symptoms affected daily functioning.
“I would like to continue this and dive deeper into how disruptive their symptoms of long COVID are to their everyday studying, teaching, or their activities to keeping a university running,” Dr. Landry said.
A version of this article originally appeared on WebMD.com.
FROM EMERGING INFECTIOUS DISEASES
Novel resuscitation for patients with nonshockable rhythms in cardiac arrest
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr Robert Glatter, medical adviser for Medscape Emergency Medicine. with a remarkable increase in neurologically intact survival. Welcome, gentlemen.
Dr. Pepe, I’d like to start off by thanking you for taking time to join us to discuss this novel concept of head-up or what you now refer to as a neuroprotective cardiopulmonary resuscitation (CPR) bundle. Can you define what this entails and why it is referred to as a neuroprotective CPR bundle?
Paul E. Pepe, MD, MPH: CPR has been life saving for 60 years the way we’ve performed it, but probably only in a very small percentage of cases. That’s one of the problems. We have almost a thousand people a day who have sudden cardiac arrest out in the community alone and more in the hospital.
We know that early defibrillation and early CPR can contribute, but it’s still a small percentage of those. About 75%-85% of the cases that we go out to see will have nonshockable rhythms and flatlines. Some cases are what we call “pulseless electrical activity,” meaning that it looks like there is some kind of organized complex, but there is no pulse associated with it.
That’s why it’s a problem, because they don’t come back. Part of the reason why we see poor outcomes is not only that these cases tend to be people who, say, were in ventricular fibrillation and then just went on over time and were not witnessed or resuscitated or had a long response time. They basically either go into flatline or autoconvert into these bizarre rhythms.
The other issue is the way we perform CPR. CPR has been lifesaving, but it only generates about 20% and maybe 15% in some cases of normal blood flow, and particularly, cerebral perfusion pressure. We’ve looked at this nicely in the laboratory.
For example, during chest compressions, we’re hoping during the recoil phase to pull blood down and back into the right heart. The problem is that you’re not only setting a pressure rate up here to the arterial side but also, you’re setting back pressure wave on the venous side. Obviously, the arterial side always wins out, but it’s just not as efficient as it could be, at 20% or 30%.
What does this entail? It entails several independent mechanisms in terms of how they work, but they all do the same thing, which is they help to pull blood out of the brain and back into the right heart by basically manipulating intrathoracic pressure and creating more of a vacuum to get blood back there.
It’s so important that people do quality CPR. You have to have a good release and that helps us suck a little bit of blood and sucks the air in. As soon as the air rushes in, it neutralizes the pressure and there’s no more vacuum and nothing else is happening until the next squeeze.
What we have found is that we can cap the airway just for a second with a little pop-up valve. It acts like when you’re sucking a milkshake through a straw and it creates more of a vacuum in the chest. Just a little pop-up valve that pulls a little bit more blood out of the brain and the rest of the body and into the right heart.
We’ve shown in a human study that, for example, the systolic blood pressure almost doubles. It really goes from 40 mm Hg during standard CPR up to 80 mm Hg, and that would be sustained for 14-15 minutes. That was a nice little study that was done in Milwaukee a few years ago.
The other thing that happens is, if you add on something else, it’s like a toilet plunger. I think many people have seen it; it’s called “active compression-decompression.” It not only compresses, but it decompresses. Where it becomes even more effective is that if you had broken bones or stiff bones as you get older or whatever it may be, as you do the CPR, you’re still getting the push down and then you’re getting the pull out. It helps on several levels. More importantly, when you put the two together, they’re very synergistic.
We, have already done the clinical trial that is the proof of concept, and that was published in The Lancet about 10 years ago. In that study, we found that the combination of those two dramatically improved survival rates by 50%, with 1-year survival neurologically intact. That got us on the right track.
The interesting thing is that someone said, “Can we lift the head up a little bit?” We did a large amount of work in the laboratory over 10 years, fine tuning it. When do you first lift the head? How soon is too soon? It’s probably bad if you just go right to it.
We had to get the pump primed a little bit with these other things to get the flow going better, not only pulling blood out of the brain but now, you have a better flow this way. You have to prime at first for a couple of minutes, and we worked out the timing: Is it 3 or 4 minutes? It seems the timing is right at about 2 minutes, then you gradually elevate the head over about 2 minutes. We’re finding that seems to be the optimal way to do it. About 2 minutes of priming with those other two devices, the adjuncts, and then gradually elevate the head over 2 minutes.
When we do that in the laboratory, we’re getting normalized cerebral perfusion pressures. You’re normalizing the flow back again with that. We’re seeing profound differences in outcome as a result, even in these cases of the nonshockables.
Dr. Glatter: What you’re doing basically is resulting in an increase in cardiac output, essentially. That really is important, especially in these nonshockable rhythms, correct?
Dr. Pepe: Absolutely. As you’re doing this compression and you’re getting these intracranial pulse waves that are going up because they’re colliding up there. It could be even damaging in itself, but we’re seeing these intracranial raises. The intracranial pressure starts going up more and more over time. Also, peripherally in most people, you’re not getting good flow out there; then, your vasculature starts to relax. The arterials are starting to not get oxygen, so they don’t go out.
With this technique where we’re returning the pressure, we’re getting to 40% of normal now with the active compression-decompression CPR plus an impedance threshold device (ACD+ITD CPR) approach. Now, you add this, and you’re almost normalizing. In humans, even in these asystole patients, we’re seeing end-title CO2s which are generally in the 15-20 range with standard CPR are now up with ACD+ITD CPR in the 30%-40% range, where we’re getting through 30 or 40 end-tidal CO2s. Now, we’re seeing even the end-tidal CO2s moving up into the 40s and 50s. We know there’s a surrogate marker telling us that we are generating much better flows not only to the rest of the body, but most importantly, to the brain.
Dr. Glatter: Ryan, could you tell us about the approach in terms of on scene, what you’re doing and how you use the device itself? Maybe you could talk about the backpack that you developed with your fire department?
Ryan P. Quinn, BS, EMS: Our approach has always been to get to the patient quickly, like everybody’s approach on a cardiac arrest when you’re responding. We are an advanced life-support paramedic ambulance service through the fire department – we’re all cross-trained firefighter paramedics. Our first vehicle from the fire department is typically the ambulance. It’s smaller and a little quicker than the fire engine. Two paramedics are going to jump out with two backpacks. One has the automated compressive device (we use the Lucas), and the other one is the sequential patient lifting device, the EleGARD.
Our two paramedics are quick to the patient’s side, and once they make contact with the patient to verify pulseless cardiac arrest, they will unpack. One person will go right to compressions if there’s nobody on compressions already. Sometimes we have a first responder police officer with an automated external defibrillator (AED). We go right to the patient’s side, concentrate on compressions, and within 90 seconds to 2 minutes, we have our bags unpacked, we’ve got the devices turned on, patient lifted up, slid under the device, and we have a supraglottic airway that is placed within 15 seconds already premade with the ITD on top. We have a sealed airway that we can continue to compress with Dr. Pepe’s original discussion of building on what’s previously been shown to work.
Dr. Pepe: Let me make a comment about this. This is so important, what Ryan is saying, because it’s something we found during the study. It’s really a true pit-crew approach. You’re not only getting these materials, which you think you need a medical Sherpa for, but you don’t. They set it up and then when they open it up, it’s all laid out just exactly as you need it. It’s not just how fast you get there; it’s how fast you get this done.
When we look at all cases combined against high-performance systems that had some of the highest survival rates around, when we compare it to those, we found that overall, even if you looked at the ones that had over 20-minute responses, the odds ratios were still three to four times higher. It was impressive.
If you looked at it under 15 minutes, which is really reasonable for most systems that get there by the way, the average time that people start CPR in any system in these studies has been about 8 minutes if you actually start this thing, which takes about 2 minutes more for this new bundle of care with this triad, it’s almost 12-14 times higher in terms of the odds ratio. I’ve never seen anything like that where the higher end is over 100 in terms of your confidence intervals.
Ryan’s system did really well and is one of those with even higher levels of outcomes, mostly because they got it on quickly. It’s like the AED for nonshockables but better because you have a wider range of efficacy where it will work.
Dr. Glatter: When the elapsed time was less than 11 minutes, that seemed to be an inflection point in the study, is that correct? You saw that 11-fold higher incidence in terms of neurologically intact survival, is that correct?
Dr. Pepe: We picked that number because that was the median time to get it on board. Half the people were getting it within that time period. The fact that you have a larger window, we’re talking about 13- almost 14-fold improvements in outcome if it was under 15 minutes. It doesn’t matter about the 11 or the 12. It’s the faster you get it on board, the better off you are.
Dr. Glatter: What’s the next step in the process of doing trials and having implementation on a larger scale based on your Annals of Emergency Medicine study? Where do you go from here?
Dr. Pepe: I’ve come to find out there are many confounding variables. What was the quality of CPR? How did people ventilate? Did they give the breath and hold it? Did they give a large enough breath so that blood can go across the transpulmonary system? There are many confounding variables. That’s why I think, in the future, it’s going to be more of looking at things like propensity score matching because we know all the variables that change outcomes. I think that’s going to be a way for me.
The other thing is that we were looking at only 380 cases here. When this doubles up in numbers, as we accrue more cases around the country of people who are implementing this, these numbers I just quoted are going to go up much higher. Unwitnessed asystole is considered futile, and you just don’t get them back. To be able to get these folks back now, even if it’s a small percentage, and the fact that we know that we’re producing this better flow, is pretty striking.
I’m really impressed, and the main thing is to make sure people are educated about it. Number two is that they understand that it has to be done right. It cannot be done wrong or you’re not going to see the differences. Getting it done right is not only following the procedures, the sequence, and how you do it, but it also has to do with getting there quickly, including assigning the right people to put it on and having well-trained people who know what they’re doing.
Dr. Glatter: In general, the lay public obviously should not attempt this in the field lifting someone’s head up in the sense of trying to do chest compressions. I think that message is important that you just said. It’s not ready for prime time yet in any way. It has to be done right.
Dr. Pepe: Bystanders have to learn CPR – they will buy us time and we’ll have better outcomes when they do that. That’s number one. Number two is that as more and more systems adopt this, you’re going to see more people coming back. If you think about what we’re doing now, if we only get back 5% of these nonshockable vs. less than 1%, it’s 5% of 800 people a day because a thousand people a day die. Several dozens of lives can be saved on a daily basis, coming back neurologically intact. That’s the key thing.
Dr. Glatter: Ryan, can you comment about your experience in the field? Is there anything in terms of your current approach that you think would be ideal to change at this point?
Mr. Quinn: We’ve established that this is the approach that we want to take and we’re just fine tuning it to be more efficient. Using the choreography of which person is going to do which role, we have clearly defined roles and clearly defined command of the scene so we’re not missing anything. Training is extremely important.
Dr. Glatter: Paul, I want to ask you about your anecdotal experience of people waking up quickly and talking after elevating their heads and going through this process. Having people talk about it and waking up is really fascinating. Maybe you can comment further on this.
Dr. Pepe: That’s a great point that you bring up because a 40- to 50-year-old guy who got saved with this approach, when he came around, he said he was hearing what people were saying. When he came out of it, he found out he had been getting CPR for about 25 minutes because he had persistent recurring ventricular fibrillation. He said, “How could I have survived that that long?”
When we told him about the new approach, he added, “Well, that’s like neuroprotective.” He’s right, because in the laboratory, we showed it was neuroprotective and we’re also getting better flows back there. It goes along with everything else, and so we’ve adopted the name because it is.
These are really high-powered systems we are comparing against, and we have the same level of return of spontaneous circulation. The major difference was when you started talking about the neurointact survival. We don’t have enough numbers yet, but next go around, we’re going to look at cerebral performance category (CPC) – CPC1 vs. the CPC2 – which were both considered intact, but CPC1 is actually better. We’re seeing many more of those, anecdotally.
I also wanted to mention that people do bring this up and say, “Well, let’s do a trial.” As far as we’re concerned, the trial’s been done in terms of The Lancet study 10 years ago that showed that the active compression-decompression had tremendously better outcomes. We show in the laboratories that you augment that a little bit. These are all [Food and Drug Administration] approved. You can go out and buy it tomorrow and get it done. I have no conflicts of interest, by the way, with any of this.
To have this device that’s going to have the potential of saving so many more lives is really an exciting breakthrough. More importantly, we’re understanding more now about the physiology of CPR and why it works. It could work much better with the approaches that we’ve been developing over the last 20 years or so.
Dr. Glatter: Absolutely. I want to thank both of you gentlemen. It’s been really an incredible experience to learn more about an advance in resuscitation that could truly be lifesaving. Thank you again for taking time to join us.
Dr. Glatter is an attending physician in the department of emergency medicine, Lenox Hill Hospital, New York. Dr. Pepe is professor, department of management, policy, and community health, University of Texas Health Sciences Center, Houston. Mr. Quinn is EMS Chief, Edina (Minn.) Fire Department. No conflicts of interest were reported.
A version of this article first appeared Jan. 26 on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr Robert Glatter, medical adviser for Medscape Emergency Medicine. with a remarkable increase in neurologically intact survival. Welcome, gentlemen.
Dr. Pepe, I’d like to start off by thanking you for taking time to join us to discuss this novel concept of head-up or what you now refer to as a neuroprotective cardiopulmonary resuscitation (CPR) bundle. Can you define what this entails and why it is referred to as a neuroprotective CPR bundle?
Paul E. Pepe, MD, MPH: CPR has been life saving for 60 years the way we’ve performed it, but probably only in a very small percentage of cases. That’s one of the problems. We have almost a thousand people a day who have sudden cardiac arrest out in the community alone and more in the hospital.
We know that early defibrillation and early CPR can contribute, but it’s still a small percentage of those. About 75%-85% of the cases that we go out to see will have nonshockable rhythms and flatlines. Some cases are what we call “pulseless electrical activity,” meaning that it looks like there is some kind of organized complex, but there is no pulse associated with it.
That’s why it’s a problem, because they don’t come back. Part of the reason why we see poor outcomes is not only that these cases tend to be people who, say, were in ventricular fibrillation and then just went on over time and were not witnessed or resuscitated or had a long response time. They basically either go into flatline or autoconvert into these bizarre rhythms.
The other issue is the way we perform CPR. CPR has been lifesaving, but it only generates about 20% and maybe 15% in some cases of normal blood flow, and particularly, cerebral perfusion pressure. We’ve looked at this nicely in the laboratory.
For example, during chest compressions, we’re hoping during the recoil phase to pull blood down and back into the right heart. The problem is that you’re not only setting a pressure rate up here to the arterial side but also, you’re setting back pressure wave on the venous side. Obviously, the arterial side always wins out, but it’s just not as efficient as it could be, at 20% or 30%.
What does this entail? It entails several independent mechanisms in terms of how they work, but they all do the same thing, which is they help to pull blood out of the brain and back into the right heart by basically manipulating intrathoracic pressure and creating more of a vacuum to get blood back there.
It’s so important that people do quality CPR. You have to have a good release and that helps us suck a little bit of blood and sucks the air in. As soon as the air rushes in, it neutralizes the pressure and there’s no more vacuum and nothing else is happening until the next squeeze.
What we have found is that we can cap the airway just for a second with a little pop-up valve. It acts like when you’re sucking a milkshake through a straw and it creates more of a vacuum in the chest. Just a little pop-up valve that pulls a little bit more blood out of the brain and the rest of the body and into the right heart.
We’ve shown in a human study that, for example, the systolic blood pressure almost doubles. It really goes from 40 mm Hg during standard CPR up to 80 mm Hg, and that would be sustained for 14-15 minutes. That was a nice little study that was done in Milwaukee a few years ago.
The other thing that happens is, if you add on something else, it’s like a toilet plunger. I think many people have seen it; it’s called “active compression-decompression.” It not only compresses, but it decompresses. Where it becomes even more effective is that if you had broken bones or stiff bones as you get older or whatever it may be, as you do the CPR, you’re still getting the push down and then you’re getting the pull out. It helps on several levels. More importantly, when you put the two together, they’re very synergistic.
We, have already done the clinical trial that is the proof of concept, and that was published in The Lancet about 10 years ago. In that study, we found that the combination of those two dramatically improved survival rates by 50%, with 1-year survival neurologically intact. That got us on the right track.
The interesting thing is that someone said, “Can we lift the head up a little bit?” We did a large amount of work in the laboratory over 10 years, fine tuning it. When do you first lift the head? How soon is too soon? It’s probably bad if you just go right to it.
We had to get the pump primed a little bit with these other things to get the flow going better, not only pulling blood out of the brain but now, you have a better flow this way. You have to prime at first for a couple of minutes, and we worked out the timing: Is it 3 or 4 minutes? It seems the timing is right at about 2 minutes, then you gradually elevate the head over about 2 minutes. We’re finding that seems to be the optimal way to do it. About 2 minutes of priming with those other two devices, the adjuncts, and then gradually elevate the head over 2 minutes.
When we do that in the laboratory, we’re getting normalized cerebral perfusion pressures. You’re normalizing the flow back again with that. We’re seeing profound differences in outcome as a result, even in these cases of the nonshockables.
Dr. Glatter: What you’re doing basically is resulting in an increase in cardiac output, essentially. That really is important, especially in these nonshockable rhythms, correct?
Dr. Pepe: Absolutely. As you’re doing this compression and you’re getting these intracranial pulse waves that are going up because they’re colliding up there. It could be even damaging in itself, but we’re seeing these intracranial raises. The intracranial pressure starts going up more and more over time. Also, peripherally in most people, you’re not getting good flow out there; then, your vasculature starts to relax. The arterials are starting to not get oxygen, so they don’t go out.
With this technique where we’re returning the pressure, we’re getting to 40% of normal now with the active compression-decompression CPR plus an impedance threshold device (ACD+ITD CPR) approach. Now, you add this, and you’re almost normalizing. In humans, even in these asystole patients, we’re seeing end-title CO2s which are generally in the 15-20 range with standard CPR are now up with ACD+ITD CPR in the 30%-40% range, where we’re getting through 30 or 40 end-tidal CO2s. Now, we’re seeing even the end-tidal CO2s moving up into the 40s and 50s. We know there’s a surrogate marker telling us that we are generating much better flows not only to the rest of the body, but most importantly, to the brain.
Dr. Glatter: Ryan, could you tell us about the approach in terms of on scene, what you’re doing and how you use the device itself? Maybe you could talk about the backpack that you developed with your fire department?
Ryan P. Quinn, BS, EMS: Our approach has always been to get to the patient quickly, like everybody’s approach on a cardiac arrest when you’re responding. We are an advanced life-support paramedic ambulance service through the fire department – we’re all cross-trained firefighter paramedics. Our first vehicle from the fire department is typically the ambulance. It’s smaller and a little quicker than the fire engine. Two paramedics are going to jump out with two backpacks. One has the automated compressive device (we use the Lucas), and the other one is the sequential patient lifting device, the EleGARD.
Our two paramedics are quick to the patient’s side, and once they make contact with the patient to verify pulseless cardiac arrest, they will unpack. One person will go right to compressions if there’s nobody on compressions already. Sometimes we have a first responder police officer with an automated external defibrillator (AED). We go right to the patient’s side, concentrate on compressions, and within 90 seconds to 2 minutes, we have our bags unpacked, we’ve got the devices turned on, patient lifted up, slid under the device, and we have a supraglottic airway that is placed within 15 seconds already premade with the ITD on top. We have a sealed airway that we can continue to compress with Dr. Pepe’s original discussion of building on what’s previously been shown to work.
Dr. Pepe: Let me make a comment about this. This is so important, what Ryan is saying, because it’s something we found during the study. It’s really a true pit-crew approach. You’re not only getting these materials, which you think you need a medical Sherpa for, but you don’t. They set it up and then when they open it up, it’s all laid out just exactly as you need it. It’s not just how fast you get there; it’s how fast you get this done.
When we look at all cases combined against high-performance systems that had some of the highest survival rates around, when we compare it to those, we found that overall, even if you looked at the ones that had over 20-minute responses, the odds ratios were still three to four times higher. It was impressive.
If you looked at it under 15 minutes, which is really reasonable for most systems that get there by the way, the average time that people start CPR in any system in these studies has been about 8 minutes if you actually start this thing, which takes about 2 minutes more for this new bundle of care with this triad, it’s almost 12-14 times higher in terms of the odds ratio. I’ve never seen anything like that where the higher end is over 100 in terms of your confidence intervals.
Ryan’s system did really well and is one of those with even higher levels of outcomes, mostly because they got it on quickly. It’s like the AED for nonshockables but better because you have a wider range of efficacy where it will work.
Dr. Glatter: When the elapsed time was less than 11 minutes, that seemed to be an inflection point in the study, is that correct? You saw that 11-fold higher incidence in terms of neurologically intact survival, is that correct?
Dr. Pepe: We picked that number because that was the median time to get it on board. Half the people were getting it within that time period. The fact that you have a larger window, we’re talking about 13- almost 14-fold improvements in outcome if it was under 15 minutes. It doesn’t matter about the 11 or the 12. It’s the faster you get it on board, the better off you are.
Dr. Glatter: What’s the next step in the process of doing trials and having implementation on a larger scale based on your Annals of Emergency Medicine study? Where do you go from here?
Dr. Pepe: I’ve come to find out there are many confounding variables. What was the quality of CPR? How did people ventilate? Did they give the breath and hold it? Did they give a large enough breath so that blood can go across the transpulmonary system? There are many confounding variables. That’s why I think, in the future, it’s going to be more of looking at things like propensity score matching because we know all the variables that change outcomes. I think that’s going to be a way for me.
The other thing is that we were looking at only 380 cases here. When this doubles up in numbers, as we accrue more cases around the country of people who are implementing this, these numbers I just quoted are going to go up much higher. Unwitnessed asystole is considered futile, and you just don’t get them back. To be able to get these folks back now, even if it’s a small percentage, and the fact that we know that we’re producing this better flow, is pretty striking.
I’m really impressed, and the main thing is to make sure people are educated about it. Number two is that they understand that it has to be done right. It cannot be done wrong or you’re not going to see the differences. Getting it done right is not only following the procedures, the sequence, and how you do it, but it also has to do with getting there quickly, including assigning the right people to put it on and having well-trained people who know what they’re doing.
Dr. Glatter: In general, the lay public obviously should not attempt this in the field lifting someone’s head up in the sense of trying to do chest compressions. I think that message is important that you just said. It’s not ready for prime time yet in any way. It has to be done right.
Dr. Pepe: Bystanders have to learn CPR – they will buy us time and we’ll have better outcomes when they do that. That’s number one. Number two is that as more and more systems adopt this, you’re going to see more people coming back. If you think about what we’re doing now, if we only get back 5% of these nonshockable vs. less than 1%, it’s 5% of 800 people a day because a thousand people a day die. Several dozens of lives can be saved on a daily basis, coming back neurologically intact. That’s the key thing.
Dr. Glatter: Ryan, can you comment about your experience in the field? Is there anything in terms of your current approach that you think would be ideal to change at this point?
Mr. Quinn: We’ve established that this is the approach that we want to take and we’re just fine tuning it to be more efficient. Using the choreography of which person is going to do which role, we have clearly defined roles and clearly defined command of the scene so we’re not missing anything. Training is extremely important.
Dr. Glatter: Paul, I want to ask you about your anecdotal experience of people waking up quickly and talking after elevating their heads and going through this process. Having people talk about it and waking up is really fascinating. Maybe you can comment further on this.
Dr. Pepe: That’s a great point that you bring up because a 40- to 50-year-old guy who got saved with this approach, when he came around, he said he was hearing what people were saying. When he came out of it, he found out he had been getting CPR for about 25 minutes because he had persistent recurring ventricular fibrillation. He said, “How could I have survived that that long?”
When we told him about the new approach, he added, “Well, that’s like neuroprotective.” He’s right, because in the laboratory, we showed it was neuroprotective and we’re also getting better flows back there. It goes along with everything else, and so we’ve adopted the name because it is.
These are really high-powered systems we are comparing against, and we have the same level of return of spontaneous circulation. The major difference was when you started talking about the neurointact survival. We don’t have enough numbers yet, but next go around, we’re going to look at cerebral performance category (CPC) – CPC1 vs. the CPC2 – which were both considered intact, but CPC1 is actually better. We’re seeing many more of those, anecdotally.
I also wanted to mention that people do bring this up and say, “Well, let’s do a trial.” As far as we’re concerned, the trial’s been done in terms of The Lancet study 10 years ago that showed that the active compression-decompression had tremendously better outcomes. We show in the laboratories that you augment that a little bit. These are all [Food and Drug Administration] approved. You can go out and buy it tomorrow and get it done. I have no conflicts of interest, by the way, with any of this.
To have this device that’s going to have the potential of saving so many more lives is really an exciting breakthrough. More importantly, we’re understanding more now about the physiology of CPR and why it works. It could work much better with the approaches that we’ve been developing over the last 20 years or so.
Dr. Glatter: Absolutely. I want to thank both of you gentlemen. It’s been really an incredible experience to learn more about an advance in resuscitation that could truly be lifesaving. Thank you again for taking time to join us.
Dr. Glatter is an attending physician in the department of emergency medicine, Lenox Hill Hospital, New York. Dr. Pepe is professor, department of management, policy, and community health, University of Texas Health Sciences Center, Houston. Mr. Quinn is EMS Chief, Edina (Minn.) Fire Department. No conflicts of interest were reported.
A version of this article first appeared Jan. 26 on Medscape.com.
This transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome. I’m Dr Robert Glatter, medical adviser for Medscape Emergency Medicine. with a remarkable increase in neurologically intact survival. Welcome, gentlemen.
Dr. Pepe, I’d like to start off by thanking you for taking time to join us to discuss this novel concept of head-up or what you now refer to as a neuroprotective cardiopulmonary resuscitation (CPR) bundle. Can you define what this entails and why it is referred to as a neuroprotective CPR bundle?
Paul E. Pepe, MD, MPH: CPR has been life saving for 60 years the way we’ve performed it, but probably only in a very small percentage of cases. That’s one of the problems. We have almost a thousand people a day who have sudden cardiac arrest out in the community alone and more in the hospital.
We know that early defibrillation and early CPR can contribute, but it’s still a small percentage of those. About 75%-85% of the cases that we go out to see will have nonshockable rhythms and flatlines. Some cases are what we call “pulseless electrical activity,” meaning that it looks like there is some kind of organized complex, but there is no pulse associated with it.
That’s why it’s a problem, because they don’t come back. Part of the reason why we see poor outcomes is not only that these cases tend to be people who, say, were in ventricular fibrillation and then just went on over time and were not witnessed or resuscitated or had a long response time. They basically either go into flatline or autoconvert into these bizarre rhythms.
The other issue is the way we perform CPR. CPR has been lifesaving, but it only generates about 20% and maybe 15% in some cases of normal blood flow, and particularly, cerebral perfusion pressure. We’ve looked at this nicely in the laboratory.
For example, during chest compressions, we’re hoping during the recoil phase to pull blood down and back into the right heart. The problem is that you’re not only setting a pressure rate up here to the arterial side but also, you’re setting back pressure wave on the venous side. Obviously, the arterial side always wins out, but it’s just not as efficient as it could be, at 20% or 30%.
What does this entail? It entails several independent mechanisms in terms of how they work, but they all do the same thing, which is they help to pull blood out of the brain and back into the right heart by basically manipulating intrathoracic pressure and creating more of a vacuum to get blood back there.
It’s so important that people do quality CPR. You have to have a good release and that helps us suck a little bit of blood and sucks the air in. As soon as the air rushes in, it neutralizes the pressure and there’s no more vacuum and nothing else is happening until the next squeeze.
What we have found is that we can cap the airway just for a second with a little pop-up valve. It acts like when you’re sucking a milkshake through a straw and it creates more of a vacuum in the chest. Just a little pop-up valve that pulls a little bit more blood out of the brain and the rest of the body and into the right heart.
We’ve shown in a human study that, for example, the systolic blood pressure almost doubles. It really goes from 40 mm Hg during standard CPR up to 80 mm Hg, and that would be sustained for 14-15 minutes. That was a nice little study that was done in Milwaukee a few years ago.
The other thing that happens is, if you add on something else, it’s like a toilet plunger. I think many people have seen it; it’s called “active compression-decompression.” It not only compresses, but it decompresses. Where it becomes even more effective is that if you had broken bones or stiff bones as you get older or whatever it may be, as you do the CPR, you’re still getting the push down and then you’re getting the pull out. It helps on several levels. More importantly, when you put the two together, they’re very synergistic.
We, have already done the clinical trial that is the proof of concept, and that was published in The Lancet about 10 years ago. In that study, we found that the combination of those two dramatically improved survival rates by 50%, with 1-year survival neurologically intact. That got us on the right track.
The interesting thing is that someone said, “Can we lift the head up a little bit?” We did a large amount of work in the laboratory over 10 years, fine tuning it. When do you first lift the head? How soon is too soon? It’s probably bad if you just go right to it.
We had to get the pump primed a little bit with these other things to get the flow going better, not only pulling blood out of the brain but now, you have a better flow this way. You have to prime at first for a couple of minutes, and we worked out the timing: Is it 3 or 4 minutes? It seems the timing is right at about 2 minutes, then you gradually elevate the head over about 2 minutes. We’re finding that seems to be the optimal way to do it. About 2 minutes of priming with those other two devices, the adjuncts, and then gradually elevate the head over 2 minutes.
When we do that in the laboratory, we’re getting normalized cerebral perfusion pressures. You’re normalizing the flow back again with that. We’re seeing profound differences in outcome as a result, even in these cases of the nonshockables.
Dr. Glatter: What you’re doing basically is resulting in an increase in cardiac output, essentially. That really is important, especially in these nonshockable rhythms, correct?
Dr. Pepe: Absolutely. As you’re doing this compression and you’re getting these intracranial pulse waves that are going up because they’re colliding up there. It could be even damaging in itself, but we’re seeing these intracranial raises. The intracranial pressure starts going up more and more over time. Also, peripherally in most people, you’re not getting good flow out there; then, your vasculature starts to relax. The arterials are starting to not get oxygen, so they don’t go out.
With this technique where we’re returning the pressure, we’re getting to 40% of normal now with the active compression-decompression CPR plus an impedance threshold device (ACD+ITD CPR) approach. Now, you add this, and you’re almost normalizing. In humans, even in these asystole patients, we’re seeing end-title CO2s which are generally in the 15-20 range with standard CPR are now up with ACD+ITD CPR in the 30%-40% range, where we’re getting through 30 or 40 end-tidal CO2s. Now, we’re seeing even the end-tidal CO2s moving up into the 40s and 50s. We know there’s a surrogate marker telling us that we are generating much better flows not only to the rest of the body, but most importantly, to the brain.
Dr. Glatter: Ryan, could you tell us about the approach in terms of on scene, what you’re doing and how you use the device itself? Maybe you could talk about the backpack that you developed with your fire department?
Ryan P. Quinn, BS, EMS: Our approach has always been to get to the patient quickly, like everybody’s approach on a cardiac arrest when you’re responding. We are an advanced life-support paramedic ambulance service through the fire department – we’re all cross-trained firefighter paramedics. Our first vehicle from the fire department is typically the ambulance. It’s smaller and a little quicker than the fire engine. Two paramedics are going to jump out with two backpacks. One has the automated compressive device (we use the Lucas), and the other one is the sequential patient lifting device, the EleGARD.
Our two paramedics are quick to the patient’s side, and once they make contact with the patient to verify pulseless cardiac arrest, they will unpack. One person will go right to compressions if there’s nobody on compressions already. Sometimes we have a first responder police officer with an automated external defibrillator (AED). We go right to the patient’s side, concentrate on compressions, and within 90 seconds to 2 minutes, we have our bags unpacked, we’ve got the devices turned on, patient lifted up, slid under the device, and we have a supraglottic airway that is placed within 15 seconds already premade with the ITD on top. We have a sealed airway that we can continue to compress with Dr. Pepe’s original discussion of building on what’s previously been shown to work.
Dr. Pepe: Let me make a comment about this. This is so important, what Ryan is saying, because it’s something we found during the study. It’s really a true pit-crew approach. You’re not only getting these materials, which you think you need a medical Sherpa for, but you don’t. They set it up and then when they open it up, it’s all laid out just exactly as you need it. It’s not just how fast you get there; it’s how fast you get this done.
When we look at all cases combined against high-performance systems that had some of the highest survival rates around, when we compare it to those, we found that overall, even if you looked at the ones that had over 20-minute responses, the odds ratios were still three to four times higher. It was impressive.
If you looked at it under 15 minutes, which is really reasonable for most systems that get there by the way, the average time that people start CPR in any system in these studies has been about 8 minutes if you actually start this thing, which takes about 2 minutes more for this new bundle of care with this triad, it’s almost 12-14 times higher in terms of the odds ratio. I’ve never seen anything like that where the higher end is over 100 in terms of your confidence intervals.
Ryan’s system did really well and is one of those with even higher levels of outcomes, mostly because they got it on quickly. It’s like the AED for nonshockables but better because you have a wider range of efficacy where it will work.
Dr. Glatter: When the elapsed time was less than 11 minutes, that seemed to be an inflection point in the study, is that correct? You saw that 11-fold higher incidence in terms of neurologically intact survival, is that correct?
Dr. Pepe: We picked that number because that was the median time to get it on board. Half the people were getting it within that time period. The fact that you have a larger window, we’re talking about 13- almost 14-fold improvements in outcome if it was under 15 minutes. It doesn’t matter about the 11 or the 12. It’s the faster you get it on board, the better off you are.
Dr. Glatter: What’s the next step in the process of doing trials and having implementation on a larger scale based on your Annals of Emergency Medicine study? Where do you go from here?
Dr. Pepe: I’ve come to find out there are many confounding variables. What was the quality of CPR? How did people ventilate? Did they give the breath and hold it? Did they give a large enough breath so that blood can go across the transpulmonary system? There are many confounding variables. That’s why I think, in the future, it’s going to be more of looking at things like propensity score matching because we know all the variables that change outcomes. I think that’s going to be a way for me.
The other thing is that we were looking at only 380 cases here. When this doubles up in numbers, as we accrue more cases around the country of people who are implementing this, these numbers I just quoted are going to go up much higher. Unwitnessed asystole is considered futile, and you just don’t get them back. To be able to get these folks back now, even if it’s a small percentage, and the fact that we know that we’re producing this better flow, is pretty striking.
I’m really impressed, and the main thing is to make sure people are educated about it. Number two is that they understand that it has to be done right. It cannot be done wrong or you’re not going to see the differences. Getting it done right is not only following the procedures, the sequence, and how you do it, but it also has to do with getting there quickly, including assigning the right people to put it on and having well-trained people who know what they’re doing.
Dr. Glatter: In general, the lay public obviously should not attempt this in the field lifting someone’s head up in the sense of trying to do chest compressions. I think that message is important that you just said. It’s not ready for prime time yet in any way. It has to be done right.
Dr. Pepe: Bystanders have to learn CPR – they will buy us time and we’ll have better outcomes when they do that. That’s number one. Number two is that as more and more systems adopt this, you’re going to see more people coming back. If you think about what we’re doing now, if we only get back 5% of these nonshockable vs. less than 1%, it’s 5% of 800 people a day because a thousand people a day die. Several dozens of lives can be saved on a daily basis, coming back neurologically intact. That’s the key thing.
Dr. Glatter: Ryan, can you comment about your experience in the field? Is there anything in terms of your current approach that you think would be ideal to change at this point?
Mr. Quinn: We’ve established that this is the approach that we want to take and we’re just fine tuning it to be more efficient. Using the choreography of which person is going to do which role, we have clearly defined roles and clearly defined command of the scene so we’re not missing anything. Training is extremely important.
Dr. Glatter: Paul, I want to ask you about your anecdotal experience of people waking up quickly and talking after elevating their heads and going through this process. Having people talk about it and waking up is really fascinating. Maybe you can comment further on this.
Dr. Pepe: That’s a great point that you bring up because a 40- to 50-year-old guy who got saved with this approach, when he came around, he said he was hearing what people were saying. When he came out of it, he found out he had been getting CPR for about 25 minutes because he had persistent recurring ventricular fibrillation. He said, “How could I have survived that that long?”
When we told him about the new approach, he added, “Well, that’s like neuroprotective.” He’s right, because in the laboratory, we showed it was neuroprotective and we’re also getting better flows back there. It goes along with everything else, and so we’ve adopted the name because it is.
These are really high-powered systems we are comparing against, and we have the same level of return of spontaneous circulation. The major difference was when you started talking about the neurointact survival. We don’t have enough numbers yet, but next go around, we’re going to look at cerebral performance category (CPC) – CPC1 vs. the CPC2 – which were both considered intact, but CPC1 is actually better. We’re seeing many more of those, anecdotally.
I also wanted to mention that people do bring this up and say, “Well, let’s do a trial.” As far as we’re concerned, the trial’s been done in terms of The Lancet study 10 years ago that showed that the active compression-decompression had tremendously better outcomes. We show in the laboratories that you augment that a little bit. These are all [Food and Drug Administration] approved. You can go out and buy it tomorrow and get it done. I have no conflicts of interest, by the way, with any of this.
To have this device that’s going to have the potential of saving so many more lives is really an exciting breakthrough. More importantly, we’re understanding more now about the physiology of CPR and why it works. It could work much better with the approaches that we’ve been developing over the last 20 years or so.
Dr. Glatter: Absolutely. I want to thank both of you gentlemen. It’s been really an incredible experience to learn more about an advance in resuscitation that could truly be lifesaving. Thank you again for taking time to join us.
Dr. Glatter is an attending physician in the department of emergency medicine, Lenox Hill Hospital, New York. Dr. Pepe is professor, department of management, policy, and community health, University of Texas Health Sciences Center, Houston. Mr. Quinn is EMS Chief, Edina (Minn.) Fire Department. No conflicts of interest were reported.
A version of this article first appeared Jan. 26 on Medscape.com.
Severe health diagnoses drive suicide risk
Individuals diagnosed with a severe physical health condition were significantly more likely to commit suicide at 6 months and at 1 year later, based on data from more than 47 million individuals in a national database.
Previous smaller studies have shown a link between increased risk for suicide and a range of health conditions including cancer, coronary heart disease, neurologic conditions, diabetes, and osteoporosis, Vahé Nafilyan, PhD, of the Office for National Statistics, Newport, England, and colleagues wrote.
However, large-scale population-level studies of the association between specific diagnoses and suicide are lacking, they said.
In a study published in The Lancet Regional Health–Europe, the researchers reviewed a dataset that combined the 2011 Census, death registration records, and the Hospital Episode Statistics. The study population included 47,354,696 individuals aged 6 years and older living in England in 2017. The mean age of the study population was 39.6 years, and 52% were female. The researchers examined deaths that occurred between Jan. 1, 2017, and Dec. 31, 2021.
The health conditions included in the analysis were low-survival cancers, chronic ischemic heart disease, chronic obstructive pulmonary disease, and degenerative neurological disease.
The diagnosis of any of these conditions significantly increased the risk for suicide compared with controls. The highest risk appeared within 6 months of a diagnosis or first treatment, but the increased risk persisted at 1 year.
The suicide rate among low-survival cancer patients was 16.6 per 100,000 patients, compared with 5.7 per 100,000 controls; at 1 year, these rates were 21.6 and 9.5 per 100,000 patients and controls, respectively.
For COPD patients, the suicide rate at 6 months after diagnosis was 13.7 per 100,000 patients versus 5.6 per 100,000 matched controls; the suicide rates at 1 year were 22.4 per 100,000 patients and 10.6 per 100,000 matched controls.
The suicide rate at 6 months for individuals diagnosed with chronic ischemic heart disease was 11.0 per 100,000 patients and 4.2 per 100,000 matched controls; at 1 year, the suicide rates were 16.1 per 100,000 patients and 8.8 per 100,000 matched controls.
The 1-year suicide rate was especially high among patients with degenerative neurological conditions (114.5 per 100,000 patients); however, the estimate was considered imprecise because of the rarity of these diseases and subsequent low number of suicides, the researchers noted.
The results support data from previous studies showing links between increased risk of suicide and severe physical conditions, the researchers wrote. Patterns of suicide were similar between men and women and after adjusting for sociodemographic factors.
The findings were limited by the inability to fully control for a history of depression or self-harm, and by the imprecise estimates given the rare occurrence of suicide overall, the researchers noted. Other limitations included the late registration of deaths from external causes and the focus only on suicides that occurred in England and Wales, meaning that individuals who traveled abroad for assisted suicide were not captured in the dataset.
“Further research is needed to understand the mechanisms driving the elevated risk of suicide and help provide the best support to these patients,” the researchers concluded.
However, the current results enhance the literature with a large, population-based review of the elevated suicide risk among individuals newly diagnosed with severe health conditions, and reflect the need for better support for these patients to help with coping, they said.
The study was funded by the Office for National Statistics. The researchers reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Individuals diagnosed with a severe physical health condition were significantly more likely to commit suicide at 6 months and at 1 year later, based on data from more than 47 million individuals in a national database.
Previous smaller studies have shown a link between increased risk for suicide and a range of health conditions including cancer, coronary heart disease, neurologic conditions, diabetes, and osteoporosis, Vahé Nafilyan, PhD, of the Office for National Statistics, Newport, England, and colleagues wrote.
However, large-scale population-level studies of the association between specific diagnoses and suicide are lacking, they said.
In a study published in The Lancet Regional Health–Europe, the researchers reviewed a dataset that combined the 2011 Census, death registration records, and the Hospital Episode Statistics. The study population included 47,354,696 individuals aged 6 years and older living in England in 2017. The mean age of the study population was 39.6 years, and 52% were female. The researchers examined deaths that occurred between Jan. 1, 2017, and Dec. 31, 2021.
The health conditions included in the analysis were low-survival cancers, chronic ischemic heart disease, chronic obstructive pulmonary disease, and degenerative neurological disease.
The diagnosis of any of these conditions significantly increased the risk for suicide compared with controls. The highest risk appeared within 6 months of a diagnosis or first treatment, but the increased risk persisted at 1 year.
The suicide rate among low-survival cancer patients was 16.6 per 100,000 patients, compared with 5.7 per 100,000 controls; at 1 year, these rates were 21.6 and 9.5 per 100,000 patients and controls, respectively.
For COPD patients, the suicide rate at 6 months after diagnosis was 13.7 per 100,000 patients versus 5.6 per 100,000 matched controls; the suicide rates at 1 year were 22.4 per 100,000 patients and 10.6 per 100,000 matched controls.
The suicide rate at 6 months for individuals diagnosed with chronic ischemic heart disease was 11.0 per 100,000 patients and 4.2 per 100,000 matched controls; at 1 year, the suicide rates were 16.1 per 100,000 patients and 8.8 per 100,000 matched controls.
The 1-year suicide rate was especially high among patients with degenerative neurological conditions (114.5 per 100,000 patients); however, the estimate was considered imprecise because of the rarity of these diseases and subsequent low number of suicides, the researchers noted.
The results support data from previous studies showing links between increased risk of suicide and severe physical conditions, the researchers wrote. Patterns of suicide were similar between men and women and after adjusting for sociodemographic factors.
The findings were limited by the inability to fully control for a history of depression or self-harm, and by the imprecise estimates given the rare occurrence of suicide overall, the researchers noted. Other limitations included the late registration of deaths from external causes and the focus only on suicides that occurred in England and Wales, meaning that individuals who traveled abroad for assisted suicide were not captured in the dataset.
“Further research is needed to understand the mechanisms driving the elevated risk of suicide and help provide the best support to these patients,” the researchers concluded.
However, the current results enhance the literature with a large, population-based review of the elevated suicide risk among individuals newly diagnosed with severe health conditions, and reflect the need for better support for these patients to help with coping, they said.
The study was funded by the Office for National Statistics. The researchers reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Individuals diagnosed with a severe physical health condition were significantly more likely to commit suicide at 6 months and at 1 year later, based on data from more than 47 million individuals in a national database.
Previous smaller studies have shown a link between increased risk for suicide and a range of health conditions including cancer, coronary heart disease, neurologic conditions, diabetes, and osteoporosis, Vahé Nafilyan, PhD, of the Office for National Statistics, Newport, England, and colleagues wrote.
However, large-scale population-level studies of the association between specific diagnoses and suicide are lacking, they said.
In a study published in The Lancet Regional Health–Europe, the researchers reviewed a dataset that combined the 2011 Census, death registration records, and the Hospital Episode Statistics. The study population included 47,354,696 individuals aged 6 years and older living in England in 2017. The mean age of the study population was 39.6 years, and 52% were female. The researchers examined deaths that occurred between Jan. 1, 2017, and Dec. 31, 2021.
The health conditions included in the analysis were low-survival cancers, chronic ischemic heart disease, chronic obstructive pulmonary disease, and degenerative neurological disease.
The diagnosis of any of these conditions significantly increased the risk for suicide compared with controls. The highest risk appeared within 6 months of a diagnosis or first treatment, but the increased risk persisted at 1 year.
The suicide rate among low-survival cancer patients was 16.6 per 100,000 patients, compared with 5.7 per 100,000 controls; at 1 year, these rates were 21.6 and 9.5 per 100,000 patients and controls, respectively.
For COPD patients, the suicide rate at 6 months after diagnosis was 13.7 per 100,000 patients versus 5.6 per 100,000 matched controls; the suicide rates at 1 year were 22.4 per 100,000 patients and 10.6 per 100,000 matched controls.
The suicide rate at 6 months for individuals diagnosed with chronic ischemic heart disease was 11.0 per 100,000 patients and 4.2 per 100,000 matched controls; at 1 year, the suicide rates were 16.1 per 100,000 patients and 8.8 per 100,000 matched controls.
The 1-year suicide rate was especially high among patients with degenerative neurological conditions (114.5 per 100,000 patients); however, the estimate was considered imprecise because of the rarity of these diseases and subsequent low number of suicides, the researchers noted.
The results support data from previous studies showing links between increased risk of suicide and severe physical conditions, the researchers wrote. Patterns of suicide were similar between men and women and after adjusting for sociodemographic factors.
The findings were limited by the inability to fully control for a history of depression or self-harm, and by the imprecise estimates given the rare occurrence of suicide overall, the researchers noted. Other limitations included the late registration of deaths from external causes and the focus only on suicides that occurred in England and Wales, meaning that individuals who traveled abroad for assisted suicide were not captured in the dataset.
“Further research is needed to understand the mechanisms driving the elevated risk of suicide and help provide the best support to these patients,” the researchers concluded.
However, the current results enhance the literature with a large, population-based review of the elevated suicide risk among individuals newly diagnosed with severe health conditions, and reflect the need for better support for these patients to help with coping, they said.
The study was funded by the Office for National Statistics. The researchers reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM THE LANCET REGIONAL HEALTH–EUROPE
Patients with COPD at higher risk of death 1 year after surgery
Patients with chronic obstructive pulmonary disease (COPD) are more likely to die within a year of undergoing elective surgery and to incur higher health care costs than are similar patients without COPD, data suggest.
An analysis of close to a million patient records found that, after adjustment for sociodemographic factors, procedure type, and comorbidities, patients with COPD were 26% more likely to die in the year after surgery than were those without COPD. Moreover, COPD was associated with a 4.6% increase in health care costs.
Previous studies have evaluated outcomes for the first 30 days after surgery. Those data “may not adequately capture the overall burden of surgery and how long it may take patients to recover,” study author Ashwin Sankar, MD, a clinician-investigator at St. Michael’s Hospital and assistant professor of anesthesia at the University of Toronto, told this news organization.
“We found that COPD often coexists with other conditions, like diabetes, coronary artery disease, and frailty,” Dr. Sankar added.
The study was published online in the Canadian Medical Association Journal.
Additional recovery support
The authors analyzed data from 932,616 patients who underwent intermediate-risk to high-risk elective noncardiac surgeries from 2005 to 2019 in Ontario. Procedures included carotid endarterectomy, open or endovascular abdominal aortic aneurysm repair, peripheral arterial bypass, total hip replacement, total knee replacement, shoulder surgery, large-bowel surgery, partial liver resection, pancreaticoduodenectomy, gastrectomy, esophagectomy, nephrectomy, cystectomy, prostatectomy, and hysterectomy.
The researchers quantified the associations of COPD with survival and costs. Their analyses included partial adjustment for sociodemographic factors and procedure type and full adjustment, which included comorbidities.
The primary outcome was all-cause death in the year after surgery; the secondary outcome was total health care costs in that year.
The mean age of the population was 65 years, and 60% of patients were women. A total of 170,482 (18%) patients had COPD. Compared with those without COPD, the patients with COPD were older and were more likely to be male, to be in a lower income quintile, to be residents of long-term care facilities, and to have been admitted to the hospital before surgery. They were also more likely to have comorbidities, including coronary artery disease, heart failure, and lung cancer.
A larger proportion of patients with COPD had frailty and medium to high comorbidity. They also more frequently underwent orthopedic, open upper abdominal, and vascular surgery.
During the year after surgery, 52,021 (5.6%) patients died, including 18,007 (10.6%) with COPD and 34,014 (4.5%) without. Those with COPD were more likely to die within 30 days of surgery (3.4% vs 1.2%).
For patients with COPD, the partially adjusted hazard ratio (HR) was 1.61 for risk of death; the fully adjusted HR was 1.26. COPD also was associated with a partially adjusted relative increase of 13.1% in health care costs and an increase of 4.6% with full adjustment.
Frailty, cancer, and procedure type were factors that modified the association between COPD and outcomes. “Procedures such as open aortic and upper abdominal surgery are associated with higher postoperative risks irrespective of COPD status, whereas others, such as orthopedic and lower abdominal surgery, may be of significantly greater risk for patients with COPD,” the authors wrote. “Our results suggest that perioperative management of patients with COPD requires careful consideration of the multiple domains that contribute to their elevated perioperative risk.
“Our finding that patients with COPD are at risk beyond 30 days after surgery suggests that it may be worthwhile to additionally support these patients’ recovery well beyond the first month after the procedure,” said Dr. Sankar.
Shared decision-making
Commenting on the study, William Whalen, MD, a pulmonary critical care specialist at Weill Cornell Medicine in New York, said, “I echo the authors’ sentiments that these findings highlight how chronically ill COPD patients are, which may be playing a role in the elevated mortality seen in this study.”
One caveat is in regard to the interpretation of the interaction effects of the study, he said. “Clinicians are unlikely to send patients who are frail or have multiple comorbidities to overly complex surgeries. Therefore, these effects may be misestimated due to selection bias.”
Two questions remain after reading the study, he added. “The first is how the degree of obstruction (i.e., the severity of COPD) impacts long-term mortality. Previous observational studies in nonsurgical COPD patients have shown increased mortality as the severity of obstruction increases. The second is how much of the long-term mortality observed in this study is related to respiratory disease from COPD. Patients with COPD are complex, and many die from nonrespiratory-related causes.”
Dr. Whalen suggests that discussion be held with the surgical team about the long-term morbidity and mortality with and without surgical intervention. Such a discussion could inform a shared decision-making process with the patient.
“Some procedures may be necessary to reduce immediate mortality, such as aortic aneurysmal repair, so [the risk of] longer-term mortality may be more acceptable in this setting,” he said. “Less straightforward are procedures that may improve quality of life. Would a patient accept an increased long-term mortality [risk] if that meant living without orthopedic-related pain?”
The study was funded by the Government of Ontario. Dr. Sankar and Dr. Whalen have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with chronic obstructive pulmonary disease (COPD) are more likely to die within a year of undergoing elective surgery and to incur higher health care costs than are similar patients without COPD, data suggest.
An analysis of close to a million patient records found that, after adjustment for sociodemographic factors, procedure type, and comorbidities, patients with COPD were 26% more likely to die in the year after surgery than were those without COPD. Moreover, COPD was associated with a 4.6% increase in health care costs.
Previous studies have evaluated outcomes for the first 30 days after surgery. Those data “may not adequately capture the overall burden of surgery and how long it may take patients to recover,” study author Ashwin Sankar, MD, a clinician-investigator at St. Michael’s Hospital and assistant professor of anesthesia at the University of Toronto, told this news organization.
“We found that COPD often coexists with other conditions, like diabetes, coronary artery disease, and frailty,” Dr. Sankar added.
The study was published online in the Canadian Medical Association Journal.
Additional recovery support
The authors analyzed data from 932,616 patients who underwent intermediate-risk to high-risk elective noncardiac surgeries from 2005 to 2019 in Ontario. Procedures included carotid endarterectomy, open or endovascular abdominal aortic aneurysm repair, peripheral arterial bypass, total hip replacement, total knee replacement, shoulder surgery, large-bowel surgery, partial liver resection, pancreaticoduodenectomy, gastrectomy, esophagectomy, nephrectomy, cystectomy, prostatectomy, and hysterectomy.
The researchers quantified the associations of COPD with survival and costs. Their analyses included partial adjustment for sociodemographic factors and procedure type and full adjustment, which included comorbidities.
The primary outcome was all-cause death in the year after surgery; the secondary outcome was total health care costs in that year.
The mean age of the population was 65 years, and 60% of patients were women. A total of 170,482 (18%) patients had COPD. Compared with those without COPD, the patients with COPD were older and were more likely to be male, to be in a lower income quintile, to be residents of long-term care facilities, and to have been admitted to the hospital before surgery. They were also more likely to have comorbidities, including coronary artery disease, heart failure, and lung cancer.
A larger proportion of patients with COPD had frailty and medium to high comorbidity. They also more frequently underwent orthopedic, open upper abdominal, and vascular surgery.
During the year after surgery, 52,021 (5.6%) patients died, including 18,007 (10.6%) with COPD and 34,014 (4.5%) without. Those with COPD were more likely to die within 30 days of surgery (3.4% vs 1.2%).
For patients with COPD, the partially adjusted hazard ratio (HR) was 1.61 for risk of death; the fully adjusted HR was 1.26. COPD also was associated with a partially adjusted relative increase of 13.1% in health care costs and an increase of 4.6% with full adjustment.
Frailty, cancer, and procedure type were factors that modified the association between COPD and outcomes. “Procedures such as open aortic and upper abdominal surgery are associated with higher postoperative risks irrespective of COPD status, whereas others, such as orthopedic and lower abdominal surgery, may be of significantly greater risk for patients with COPD,” the authors wrote. “Our results suggest that perioperative management of patients with COPD requires careful consideration of the multiple domains that contribute to their elevated perioperative risk.
“Our finding that patients with COPD are at risk beyond 30 days after surgery suggests that it may be worthwhile to additionally support these patients’ recovery well beyond the first month after the procedure,” said Dr. Sankar.
Shared decision-making
Commenting on the study, William Whalen, MD, a pulmonary critical care specialist at Weill Cornell Medicine in New York, said, “I echo the authors’ sentiments that these findings highlight how chronically ill COPD patients are, which may be playing a role in the elevated mortality seen in this study.”
One caveat is in regard to the interpretation of the interaction effects of the study, he said. “Clinicians are unlikely to send patients who are frail or have multiple comorbidities to overly complex surgeries. Therefore, these effects may be misestimated due to selection bias.”
Two questions remain after reading the study, he added. “The first is how the degree of obstruction (i.e., the severity of COPD) impacts long-term mortality. Previous observational studies in nonsurgical COPD patients have shown increased mortality as the severity of obstruction increases. The second is how much of the long-term mortality observed in this study is related to respiratory disease from COPD. Patients with COPD are complex, and many die from nonrespiratory-related causes.”
Dr. Whalen suggests that discussion be held with the surgical team about the long-term morbidity and mortality with and without surgical intervention. Such a discussion could inform a shared decision-making process with the patient.
“Some procedures may be necessary to reduce immediate mortality, such as aortic aneurysmal repair, so [the risk of] longer-term mortality may be more acceptable in this setting,” he said. “Less straightforward are procedures that may improve quality of life. Would a patient accept an increased long-term mortality [risk] if that meant living without orthopedic-related pain?”
The study was funded by the Government of Ontario. Dr. Sankar and Dr. Whalen have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Patients with chronic obstructive pulmonary disease (COPD) are more likely to die within a year of undergoing elective surgery and to incur higher health care costs than are similar patients without COPD, data suggest.
An analysis of close to a million patient records found that, after adjustment for sociodemographic factors, procedure type, and comorbidities, patients with COPD were 26% more likely to die in the year after surgery than were those without COPD. Moreover, COPD was associated with a 4.6% increase in health care costs.
Previous studies have evaluated outcomes for the first 30 days after surgery. Those data “may not adequately capture the overall burden of surgery and how long it may take patients to recover,” study author Ashwin Sankar, MD, a clinician-investigator at St. Michael’s Hospital and assistant professor of anesthesia at the University of Toronto, told this news organization.
“We found that COPD often coexists with other conditions, like diabetes, coronary artery disease, and frailty,” Dr. Sankar added.
The study was published online in the Canadian Medical Association Journal.
Additional recovery support
The authors analyzed data from 932,616 patients who underwent intermediate-risk to high-risk elective noncardiac surgeries from 2005 to 2019 in Ontario. Procedures included carotid endarterectomy, open or endovascular abdominal aortic aneurysm repair, peripheral arterial bypass, total hip replacement, total knee replacement, shoulder surgery, large-bowel surgery, partial liver resection, pancreaticoduodenectomy, gastrectomy, esophagectomy, nephrectomy, cystectomy, prostatectomy, and hysterectomy.
The researchers quantified the associations of COPD with survival and costs. Their analyses included partial adjustment for sociodemographic factors and procedure type and full adjustment, which included comorbidities.
The primary outcome was all-cause death in the year after surgery; the secondary outcome was total health care costs in that year.
The mean age of the population was 65 years, and 60% of patients were women. A total of 170,482 (18%) patients had COPD. Compared with those without COPD, the patients with COPD were older and were more likely to be male, to be in a lower income quintile, to be residents of long-term care facilities, and to have been admitted to the hospital before surgery. They were also more likely to have comorbidities, including coronary artery disease, heart failure, and lung cancer.
A larger proportion of patients with COPD had frailty and medium to high comorbidity. They also more frequently underwent orthopedic, open upper abdominal, and vascular surgery.
During the year after surgery, 52,021 (5.6%) patients died, including 18,007 (10.6%) with COPD and 34,014 (4.5%) without. Those with COPD were more likely to die within 30 days of surgery (3.4% vs 1.2%).
For patients with COPD, the partially adjusted hazard ratio (HR) was 1.61 for risk of death; the fully adjusted HR was 1.26. COPD also was associated with a partially adjusted relative increase of 13.1% in health care costs and an increase of 4.6% with full adjustment.
Frailty, cancer, and procedure type were factors that modified the association between COPD and outcomes. “Procedures such as open aortic and upper abdominal surgery are associated with higher postoperative risks irrespective of COPD status, whereas others, such as orthopedic and lower abdominal surgery, may be of significantly greater risk for patients with COPD,” the authors wrote. “Our results suggest that perioperative management of patients with COPD requires careful consideration of the multiple domains that contribute to their elevated perioperative risk.
“Our finding that patients with COPD are at risk beyond 30 days after surgery suggests that it may be worthwhile to additionally support these patients’ recovery well beyond the first month after the procedure,” said Dr. Sankar.
Shared decision-making
Commenting on the study, William Whalen, MD, a pulmonary critical care specialist at Weill Cornell Medicine in New York, said, “I echo the authors’ sentiments that these findings highlight how chronically ill COPD patients are, which may be playing a role in the elevated mortality seen in this study.”
One caveat is in regard to the interpretation of the interaction effects of the study, he said. “Clinicians are unlikely to send patients who are frail or have multiple comorbidities to overly complex surgeries. Therefore, these effects may be misestimated due to selection bias.”
Two questions remain after reading the study, he added. “The first is how the degree of obstruction (i.e., the severity of COPD) impacts long-term mortality. Previous observational studies in nonsurgical COPD patients have shown increased mortality as the severity of obstruction increases. The second is how much of the long-term mortality observed in this study is related to respiratory disease from COPD. Patients with COPD are complex, and many die from nonrespiratory-related causes.”
Dr. Whalen suggests that discussion be held with the surgical team about the long-term morbidity and mortality with and without surgical intervention. Such a discussion could inform a shared decision-making process with the patient.
“Some procedures may be necessary to reduce immediate mortality, such as aortic aneurysmal repair, so [the risk of] longer-term mortality may be more acceptable in this setting,” he said. “Less straightforward are procedures that may improve quality of life. Would a patient accept an increased long-term mortality [risk] if that meant living without orthopedic-related pain?”
The study was funded by the Government of Ontario. Dr. Sankar and Dr. Whalen have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FDA wants annual COVID boosters, just like annual flu shots
The U.S. Food and Drug Administration is suggesting a single annual shot. The formulation would be selected in June targeting the most threatening COVID-19 strains, and then people could get a shot in the fall when people begin spending more time indoors and exposure increases.
Some people, such as those who are older or immunocompromised, may need more than one dose.
A national advisory committee is expected to vote on the proposal at a meeting Jan. 26.
People in the United States have been much less likely to get an updated COVID-19 booster shot, compared with widespread uptake of the primary vaccine series. In its proposal, the FDA indicated it hoped a single annual shot would overcome challenges created by the complexity of the process – both in messaging and administration – attributed to that low booster rate. Nine in 10 people age 12 or older got the primary vaccine series in the United States, but only 15% got the latest booster shot for COVID-19.
About half of children and adults in the U.S. get an annual flu shot, according to Centers for Disease Control and Prevention data.
The FDA also wants to move to a single COVID-19 vaccine formulation that would be used for primary vaccine series and for booster shots.
COVID-19 cases, hospitalizations, and deaths are trending downward, according to the data tracker from the New York Times. Cases are down 28%, with 47,290 tallied daily. Hospitalizations are down 22%, with 37,474 daily. Deaths are down 4%, with an average of 489 per day as of Jan. 22.
A version of this article originally appeared on WebMD.com.
The U.S. Food and Drug Administration is suggesting a single annual shot. The formulation would be selected in June targeting the most threatening COVID-19 strains, and then people could get a shot in the fall when people begin spending more time indoors and exposure increases.
Some people, such as those who are older or immunocompromised, may need more than one dose.
A national advisory committee is expected to vote on the proposal at a meeting Jan. 26.
People in the United States have been much less likely to get an updated COVID-19 booster shot, compared with widespread uptake of the primary vaccine series. In its proposal, the FDA indicated it hoped a single annual shot would overcome challenges created by the complexity of the process – both in messaging and administration – attributed to that low booster rate. Nine in 10 people age 12 or older got the primary vaccine series in the United States, but only 15% got the latest booster shot for COVID-19.
About half of children and adults in the U.S. get an annual flu shot, according to Centers for Disease Control and Prevention data.
The FDA also wants to move to a single COVID-19 vaccine formulation that would be used for primary vaccine series and for booster shots.
COVID-19 cases, hospitalizations, and deaths are trending downward, according to the data tracker from the New York Times. Cases are down 28%, with 47,290 tallied daily. Hospitalizations are down 22%, with 37,474 daily. Deaths are down 4%, with an average of 489 per day as of Jan. 22.
A version of this article originally appeared on WebMD.com.
The U.S. Food and Drug Administration is suggesting a single annual shot. The formulation would be selected in June targeting the most threatening COVID-19 strains, and then people could get a shot in the fall when people begin spending more time indoors and exposure increases.
Some people, such as those who are older or immunocompromised, may need more than one dose.
A national advisory committee is expected to vote on the proposal at a meeting Jan. 26.
People in the United States have been much less likely to get an updated COVID-19 booster shot, compared with widespread uptake of the primary vaccine series. In its proposal, the FDA indicated it hoped a single annual shot would overcome challenges created by the complexity of the process – both in messaging and administration – attributed to that low booster rate. Nine in 10 people age 12 or older got the primary vaccine series in the United States, but only 15% got the latest booster shot for COVID-19.
About half of children and adults in the U.S. get an annual flu shot, according to Centers for Disease Control and Prevention data.
The FDA also wants to move to a single COVID-19 vaccine formulation that would be used for primary vaccine series and for booster shots.
COVID-19 cases, hospitalizations, and deaths are trending downward, according to the data tracker from the New York Times. Cases are down 28%, with 47,290 tallied daily. Hospitalizations are down 22%, with 37,474 daily. Deaths are down 4%, with an average of 489 per day as of Jan. 22.
A version of this article originally appeared on WebMD.com.
How does SARS-CoV-2 affect other respiratory diseases?
In 2020, the rapid spread of the newly identified SARS-CoV-2 coronavirus led various global public health institutions to establish strategies to stop transmission and reduce mortality. Nonpharmacological measures – including social distancing, regular hand washing, and the use of face masks – contributed to reducing the impact of the COVID-19 pandemic on health systems in different regions of the world. However, because of the implementation of these measures, the transmission of other infectious agents also experienced a marked reduction.
Approximately 3 years after the start of the pandemic, , generating phenomena ranging from an immunity gap, which favors the increase in some diseases, to the apparent disappearance of an influenza virus lineage.
Understanding the phenomenon
In mid-2021, doctors and researchers around the world began to share their opinions about the side effect of the strict measures implemented to contain COVID-19.
In May 2021, along with some coresearchers, Emmanuel Grimprel, MD, of the Pediatric Infectious Pathology Group in Créteil, France, wrote for Infectious Disease Now, “The transmission of some pathogens is often similar to that of SARS-CoV-2, essentially large droplets, aerosols, and direct hand contact, often with lower transmissibility. The lack of immune system stimulation due to nonpharmaceutical measures induces an ‘immune debt’ that may have negative consequences when the pandemic is under control.” According to the authors, mathematical models evaluated up to that point were already suggesting that the respiratory syncytial virus (RSV) and influenza A epidemics would be more serious in subsequent years.
In July 2022, a commentary in The Lancet led by Kevin Messacar, MD, of the University of Colorado at Denver, Aurora, grew in relevance and gave prominence to the phenomenon. In the commentary, Dr. Messacar and a group of experts explained how the decrease in exposure to endemic viruses had given rise to an immunity gap.
“The immunity gap phenomenon that has been reported in articles such as The Lancet publication is mainly due to the isolation that took place to prevent SARS-CoV-2 infections. Although this distancing was a good response to combat infections, or at least delay them while coronavirus research advanced, what we are now experiencing is the increase in cases of respiratory diseases caused by other agents such as respiratory syncytial virus and influenza due to lack of exposure,” as explained to this news organization by Erandeni Martínez Jiménez, biomedicine graduate and member of the Medical Virology Laboratory of the Mexican Institute of Social Security, at the Zone No. 5 General Hospital in Metepec-Atlixco, Mexico.
“This phenomenon occurs in all age groups. However, it is more evident in children and babies, since at their age, they have been exposed to fewer pathogens and, when added to isolation, makes this immunity gap more evident. Many immunologists compare this to hygiene theory in which it is explained that a ‘sterile’ environment will cause children to avoid the everyday and common pathogens required to be able to develop an adequate immune system,” added Martínez Jimenez.
“In addition, due to the isolation, the vaccination rate in children decreased, since many parents did not risk their children going out. This causes the immunity gap to grow even further as these children are not protected against common pathogens. While a mother passes antibodies to the child through the uterus via her placenta, the mother will only pass on those antibodies to which she has been exposed and as expected due to the lockdown, exposure to other pathogens has been greatly reduced.”
On the other hand, Andreu Comas, MD, PhD, MHS, of the Center for Research in Health Sciences and Biomedicine of the Autonomous University of San Luis Potosí (Mexico), considered that there are other immunity gaps that are not limited to respiratory infections and that are related to the fall in vaccination coverage. “Children are going to experience several immunity gaps. In the middle of the previous 6-year term, we had a vaccination schedule coverage of around 70% for children. Now that vaccination coverage has fallen to 30%, today we have an immunity gap for measles, rubella, mumps, tetanus, diphtheria, whooping cough, and meningeal tuberculosis. We have a significant growth or risk for other diseases.”
Lineage extinction
Three types of influenza viruses – A, B, and C – cause infections in humans. Although influenza A virus is the main type associated with infections during seasonal periods, as of 2020, influenza B virus was considered the causative agent of about a quarter of annual influenza cases.
During the onset of the COVID-19 pandemic, cocirculation of the two distinct lineages of influenza B viruses, B/Victoria/2/1987 (B/Victoria) and B/Yamagata/16/1988 (B/Yamagata), decreased significantly. According to data from the FluNet tool, which is coordinated by the World Health Organization, since March 2020 the isolation or sequencing of viruses belonging to the Yamagata lineage was not conclusively carried out.
Specialists like John Paget, PhD, from the Netherlands Institute for Health Services Research (Nivel) in Utrecht, have indicated that determining the extinction of the B/Yamagata lineage is critical. There is the possibility of a reintroduction of the lineage, as has occurred in the past with the reemergence of influenza A (H1N1) in 1997, which could represent a risk in subsequent years.
“In the next few years, research related to viruses such as influenza B and the impact on population immunity will be important. Let’s remember that influenza changes every year due to its characteristics, so a lack of exposure will also have an impact on the development of the disease,” said Martínez Jiménez.
Vaccination is essential
According to Dr. Comas, the only way to overcome the immunity gap phenomenon is through vaccination campaigns. “There is no other way to overcome the phenomenon, and how fast it is done will depend on the effort,” he said.
“In the case of COVID-19, it is not planned to vaccinate children under 5 years of age, and if we do not vaccinate children under 5 years of age, that gap will exist. In addition, this winter season will be important to know whether we are already endemic or not. It will be the key point, and it will determine if we will have a peak or not in the summer.
“In the case of the rest of the diseases, we need to correct what has been deficient in different governments, and we are going to have the resurgence of other infectious diseases that had already been forgotten. We have the example of poliomyelitis, the increase in meningeal tuberculosis, and we will have an increase in whooping cough and pertussislike syndrome. In this sense, we are going back to the point where Mexico and the world were around the ‘60s and ‘70s, and we have to be very alert to detect, isolate, and revaccinate.”
Finally, Dr. Comas called for continuing precautionary measures before the arrival of the sixth wave. “At a national level, the sixth wave of COVID-19 has already begun, and an increase in cases is expected in January. Regarding vaccines, if you are over 18 years of age and have not had any vaccine dose, you can get Abdala, however, there are no studies on this vaccine as a booster, and it is not authorized by the Mexican government for this purpose. Therefore, it is necessary to continue with measures such as the use of face masks in crowded places or with poor ventilation, and in the event of having symptoms, avoid going out and encourage ventilation at work and schools. If we do this, at least in the case of diseases that are transmitted by the respiratory route, the impact will be minimal.”
Martínez Jiménez and Dr. Comas have disclosed no relevant financial relationships.
This article was translated from the Medscape Spanish Edition.
A version of this article first appeared on Medscape.com.
In 2020, the rapid spread of the newly identified SARS-CoV-2 coronavirus led various global public health institutions to establish strategies to stop transmission and reduce mortality. Nonpharmacological measures – including social distancing, regular hand washing, and the use of face masks – contributed to reducing the impact of the COVID-19 pandemic on health systems in different regions of the world. However, because of the implementation of these measures, the transmission of other infectious agents also experienced a marked reduction.
Approximately 3 years after the start of the pandemic, , generating phenomena ranging from an immunity gap, which favors the increase in some diseases, to the apparent disappearance of an influenza virus lineage.
Understanding the phenomenon
In mid-2021, doctors and researchers around the world began to share their opinions about the side effect of the strict measures implemented to contain COVID-19.
In May 2021, along with some coresearchers, Emmanuel Grimprel, MD, of the Pediatric Infectious Pathology Group in Créteil, France, wrote for Infectious Disease Now, “The transmission of some pathogens is often similar to that of SARS-CoV-2, essentially large droplets, aerosols, and direct hand contact, often with lower transmissibility. The lack of immune system stimulation due to nonpharmaceutical measures induces an ‘immune debt’ that may have negative consequences when the pandemic is under control.” According to the authors, mathematical models evaluated up to that point were already suggesting that the respiratory syncytial virus (RSV) and influenza A epidemics would be more serious in subsequent years.
In July 2022, a commentary in The Lancet led by Kevin Messacar, MD, of the University of Colorado at Denver, Aurora, grew in relevance and gave prominence to the phenomenon. In the commentary, Dr. Messacar and a group of experts explained how the decrease in exposure to endemic viruses had given rise to an immunity gap.
“The immunity gap phenomenon that has been reported in articles such as The Lancet publication is mainly due to the isolation that took place to prevent SARS-CoV-2 infections. Although this distancing was a good response to combat infections, or at least delay them while coronavirus research advanced, what we are now experiencing is the increase in cases of respiratory diseases caused by other agents such as respiratory syncytial virus and influenza due to lack of exposure,” as explained to this news organization by Erandeni Martínez Jiménez, biomedicine graduate and member of the Medical Virology Laboratory of the Mexican Institute of Social Security, at the Zone No. 5 General Hospital in Metepec-Atlixco, Mexico.
“This phenomenon occurs in all age groups. However, it is more evident in children and babies, since at their age, they have been exposed to fewer pathogens and, when added to isolation, makes this immunity gap more evident. Many immunologists compare this to hygiene theory in which it is explained that a ‘sterile’ environment will cause children to avoid the everyday and common pathogens required to be able to develop an adequate immune system,” added Martínez Jimenez.
“In addition, due to the isolation, the vaccination rate in children decreased, since many parents did not risk their children going out. This causes the immunity gap to grow even further as these children are not protected against common pathogens. While a mother passes antibodies to the child through the uterus via her placenta, the mother will only pass on those antibodies to which she has been exposed and as expected due to the lockdown, exposure to other pathogens has been greatly reduced.”
On the other hand, Andreu Comas, MD, PhD, MHS, of the Center for Research in Health Sciences and Biomedicine of the Autonomous University of San Luis Potosí (Mexico), considered that there are other immunity gaps that are not limited to respiratory infections and that are related to the fall in vaccination coverage. “Children are going to experience several immunity gaps. In the middle of the previous 6-year term, we had a vaccination schedule coverage of around 70% for children. Now that vaccination coverage has fallen to 30%, today we have an immunity gap for measles, rubella, mumps, tetanus, diphtheria, whooping cough, and meningeal tuberculosis. We have a significant growth or risk for other diseases.”
Lineage extinction
Three types of influenza viruses – A, B, and C – cause infections in humans. Although influenza A virus is the main type associated with infections during seasonal periods, as of 2020, influenza B virus was considered the causative agent of about a quarter of annual influenza cases.
During the onset of the COVID-19 pandemic, cocirculation of the two distinct lineages of influenza B viruses, B/Victoria/2/1987 (B/Victoria) and B/Yamagata/16/1988 (B/Yamagata), decreased significantly. According to data from the FluNet tool, which is coordinated by the World Health Organization, since March 2020 the isolation or sequencing of viruses belonging to the Yamagata lineage was not conclusively carried out.
Specialists like John Paget, PhD, from the Netherlands Institute for Health Services Research (Nivel) in Utrecht, have indicated that determining the extinction of the B/Yamagata lineage is critical. There is the possibility of a reintroduction of the lineage, as has occurred in the past with the reemergence of influenza A (H1N1) in 1997, which could represent a risk in subsequent years.
“In the next few years, research related to viruses such as influenza B and the impact on population immunity will be important. Let’s remember that influenza changes every year due to its characteristics, so a lack of exposure will also have an impact on the development of the disease,” said Martínez Jiménez.
Vaccination is essential
According to Dr. Comas, the only way to overcome the immunity gap phenomenon is through vaccination campaigns. “There is no other way to overcome the phenomenon, and how fast it is done will depend on the effort,” he said.
“In the case of COVID-19, it is not planned to vaccinate children under 5 years of age, and if we do not vaccinate children under 5 years of age, that gap will exist. In addition, this winter season will be important to know whether we are already endemic or not. It will be the key point, and it will determine if we will have a peak or not in the summer.
“In the case of the rest of the diseases, we need to correct what has been deficient in different governments, and we are going to have the resurgence of other infectious diseases that had already been forgotten. We have the example of poliomyelitis, the increase in meningeal tuberculosis, and we will have an increase in whooping cough and pertussislike syndrome. In this sense, we are going back to the point where Mexico and the world were around the ‘60s and ‘70s, and we have to be very alert to detect, isolate, and revaccinate.”
Finally, Dr. Comas called for continuing precautionary measures before the arrival of the sixth wave. “At a national level, the sixth wave of COVID-19 has already begun, and an increase in cases is expected in January. Regarding vaccines, if you are over 18 years of age and have not had any vaccine dose, you can get Abdala, however, there are no studies on this vaccine as a booster, and it is not authorized by the Mexican government for this purpose. Therefore, it is necessary to continue with measures such as the use of face masks in crowded places or with poor ventilation, and in the event of having symptoms, avoid going out and encourage ventilation at work and schools. If we do this, at least in the case of diseases that are transmitted by the respiratory route, the impact will be minimal.”
Martínez Jiménez and Dr. Comas have disclosed no relevant financial relationships.
This article was translated from the Medscape Spanish Edition.
A version of this article first appeared on Medscape.com.
In 2020, the rapid spread of the newly identified SARS-CoV-2 coronavirus led various global public health institutions to establish strategies to stop transmission and reduce mortality. Nonpharmacological measures – including social distancing, regular hand washing, and the use of face masks – contributed to reducing the impact of the COVID-19 pandemic on health systems in different regions of the world. However, because of the implementation of these measures, the transmission of other infectious agents also experienced a marked reduction.
Approximately 3 years after the start of the pandemic, , generating phenomena ranging from an immunity gap, which favors the increase in some diseases, to the apparent disappearance of an influenza virus lineage.
Understanding the phenomenon
In mid-2021, doctors and researchers around the world began to share their opinions about the side effect of the strict measures implemented to contain COVID-19.
In May 2021, along with some coresearchers, Emmanuel Grimprel, MD, of the Pediatric Infectious Pathology Group in Créteil, France, wrote for Infectious Disease Now, “The transmission of some pathogens is often similar to that of SARS-CoV-2, essentially large droplets, aerosols, and direct hand contact, often with lower transmissibility. The lack of immune system stimulation due to nonpharmaceutical measures induces an ‘immune debt’ that may have negative consequences when the pandemic is under control.” According to the authors, mathematical models evaluated up to that point were already suggesting that the respiratory syncytial virus (RSV) and influenza A epidemics would be more serious in subsequent years.
In July 2022, a commentary in The Lancet led by Kevin Messacar, MD, of the University of Colorado at Denver, Aurora, grew in relevance and gave prominence to the phenomenon. In the commentary, Dr. Messacar and a group of experts explained how the decrease in exposure to endemic viruses had given rise to an immunity gap.
“The immunity gap phenomenon that has been reported in articles such as The Lancet publication is mainly due to the isolation that took place to prevent SARS-CoV-2 infections. Although this distancing was a good response to combat infections, or at least delay them while coronavirus research advanced, what we are now experiencing is the increase in cases of respiratory diseases caused by other agents such as respiratory syncytial virus and influenza due to lack of exposure,” as explained to this news organization by Erandeni Martínez Jiménez, biomedicine graduate and member of the Medical Virology Laboratory of the Mexican Institute of Social Security, at the Zone No. 5 General Hospital in Metepec-Atlixco, Mexico.
“This phenomenon occurs in all age groups. However, it is more evident in children and babies, since at their age, they have been exposed to fewer pathogens and, when added to isolation, makes this immunity gap more evident. Many immunologists compare this to hygiene theory in which it is explained that a ‘sterile’ environment will cause children to avoid the everyday and common pathogens required to be able to develop an adequate immune system,” added Martínez Jimenez.
“In addition, due to the isolation, the vaccination rate in children decreased, since many parents did not risk their children going out. This causes the immunity gap to grow even further as these children are not protected against common pathogens. While a mother passes antibodies to the child through the uterus via her placenta, the mother will only pass on those antibodies to which she has been exposed and as expected due to the lockdown, exposure to other pathogens has been greatly reduced.”
On the other hand, Andreu Comas, MD, PhD, MHS, of the Center for Research in Health Sciences and Biomedicine of the Autonomous University of San Luis Potosí (Mexico), considered that there are other immunity gaps that are not limited to respiratory infections and that are related to the fall in vaccination coverage. “Children are going to experience several immunity gaps. In the middle of the previous 6-year term, we had a vaccination schedule coverage of around 70% for children. Now that vaccination coverage has fallen to 30%, today we have an immunity gap for measles, rubella, mumps, tetanus, diphtheria, whooping cough, and meningeal tuberculosis. We have a significant growth or risk for other diseases.”
Lineage extinction
Three types of influenza viruses – A, B, and C – cause infections in humans. Although influenza A virus is the main type associated with infections during seasonal periods, as of 2020, influenza B virus was considered the causative agent of about a quarter of annual influenza cases.
During the onset of the COVID-19 pandemic, cocirculation of the two distinct lineages of influenza B viruses, B/Victoria/2/1987 (B/Victoria) and B/Yamagata/16/1988 (B/Yamagata), decreased significantly. According to data from the FluNet tool, which is coordinated by the World Health Organization, since March 2020 the isolation or sequencing of viruses belonging to the Yamagata lineage was not conclusively carried out.
Specialists like John Paget, PhD, from the Netherlands Institute for Health Services Research (Nivel) in Utrecht, have indicated that determining the extinction of the B/Yamagata lineage is critical. There is the possibility of a reintroduction of the lineage, as has occurred in the past with the reemergence of influenza A (H1N1) in 1997, which could represent a risk in subsequent years.
“In the next few years, research related to viruses such as influenza B and the impact on population immunity will be important. Let’s remember that influenza changes every year due to its characteristics, so a lack of exposure will also have an impact on the development of the disease,” said Martínez Jiménez.
Vaccination is essential
According to Dr. Comas, the only way to overcome the immunity gap phenomenon is through vaccination campaigns. “There is no other way to overcome the phenomenon, and how fast it is done will depend on the effort,” he said.
“In the case of COVID-19, it is not planned to vaccinate children under 5 years of age, and if we do not vaccinate children under 5 years of age, that gap will exist. In addition, this winter season will be important to know whether we are already endemic or not. It will be the key point, and it will determine if we will have a peak or not in the summer.
“In the case of the rest of the diseases, we need to correct what has been deficient in different governments, and we are going to have the resurgence of other infectious diseases that had already been forgotten. We have the example of poliomyelitis, the increase in meningeal tuberculosis, and we will have an increase in whooping cough and pertussislike syndrome. In this sense, we are going back to the point where Mexico and the world were around the ‘60s and ‘70s, and we have to be very alert to detect, isolate, and revaccinate.”
Finally, Dr. Comas called for continuing precautionary measures before the arrival of the sixth wave. “At a national level, the sixth wave of COVID-19 has already begun, and an increase in cases is expected in January. Regarding vaccines, if you are over 18 years of age and have not had any vaccine dose, you can get Abdala, however, there are no studies on this vaccine as a booster, and it is not authorized by the Mexican government for this purpose. Therefore, it is necessary to continue with measures such as the use of face masks in crowded places or with poor ventilation, and in the event of having symptoms, avoid going out and encourage ventilation at work and schools. If we do this, at least in the case of diseases that are transmitted by the respiratory route, the impact will be minimal.”
Martínez Jiménez and Dr. Comas have disclosed no relevant financial relationships.
This article was translated from the Medscape Spanish Edition.
A version of this article first appeared on Medscape.com.
Is it time for yet another COVID booster? It’s complicated
For some people who have received a two-dose primary series and all the recommended boosters, that could mean a sixth shot since COVID-19 vaccines became available. But is even that enough (or too much)?
At this point, no one knows for sure, but new guidance may be on the docket.
On Jan. 26, the FDA’s Vaccines and Related Biological Products Advisory Committee is meeting. On the agenda is discussion about plans for future vaccinations for COVID-19.The committee, made up of external advisers, evaluates data on vaccines and other products for the agency.
According to the FDA announcement, after the meeting, “the FDA will consider whether to recommend adjustments to the current authorizations and approvals, and the FDA will consider the most efficient and transparent process to use for selection of strains for inclusion in the primary and booster vaccines.”
From there, the CDC will take up the issue and decide on recommendations.
The issue is important, as more than 550 Americans a day are still dying from COVID-19, as of the week ending Jan. 13, the CDC reported. That’s up from 346 a day for the week ending Dec. 28.
Yet, uptake of the newest vaccine, the bivalent booster, has been slow. As of Jan. 11, just 15.9% of the population 5 years and up has gotten it; for those most vulnerable to COVID19 – those 65 and up – the number is just 39%.
COVID vaccines, 2023 and beyond
Meanwhile, infectious disease experts have widely differing views on what the vaccination landscape of 2023 and beyond should look like. Among the areas of disagreement are how effective the bivalent vaccine is, which people most need another shot, and what type of vaccine is best.
“I think we probably will need another booster,” says Peter Hotez, MD, PhD, dean of the National School of Tropical Medicine at Baylor College of Medicine, and codirector of the Center for Vaccine Development at Texas Children’s Hospital in Houston. “The question is, what is it going to be? Is it going to be the same bivalent that we just got, or will it be a new bivalent or even a trivalent?”
The trivalent booster, he suggested, might include something more protective against XBB.1.5.
The bivalent booster gives “broadened immunity” that is improved from the original booster shots, says Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, WebMD’s sister site for health professionals.
In his publication Ground Truths, Dr. Topol on Jan. 11 explained how new data caused him to reverse his previously skeptical view of how the FDA authorized the bivalent vaccine in September without data on how it affected humans at the time.
Paul Offit, MD, director of the Vaccine Education Center and a professor of pediatrics at the Children’s Hospital of Philadelphia, is a member of the FDA advisory committee for vaccines. He still takes a dimmer view of more bivalent booster vaccines, at least as a blanket recommendation.
While he acknowledges that boosters can help some groups – such as older adults, people with multiple health conditions, and those with compromised immune systems – he opposes a recommendation that’s population-wide.
“People who fall into those three groups do benefit,” he says, “but the recommendation is everyone over 6 months get the bivalent, and what I’m asking is, ‘Where is the data that a healthy 12-year-old boy needs a booster to stay out of the hospital?’ ”
Evolving research
“We are trying to understand how to stay one step ahead rather than several steps behind [the virus],“ says Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota.
Among the key questions: How well can a vaccine work against a single subvariant, when no one can say for sure what the next predominant subvariant will be?
Much more research has become available recently about the bivalent vaccine and its effectiveness, Dr. Osterholm says. “The bivalent vaccine is working as well as we could have expected,” he says, especially in high-risk people and in those over age 65. “The challenge we have is, what does that mean going forward?”
In his review, Dr. Topol concludes: “There is now more than ample, highly consistent evidence via lab studies and clinical outcomes to support the bivalent’s benefit over the original booster.”
Among other evidence, he looked at eight studies, including four that used a live virus as part of the research. Six of the eight studies showed the bivalent booster is more effective against the BA.5 variant, compared with the original booster shots. Two others showed no real difference.
“The four live virus studies offer consistent evidence of broadened immunity for the BA.5 vaccine that is improved over the original booster shots,” Dr. Topol wrote. The evidence also found the bivalent antibody response superior against XBB, he wrote.
Dr. Topol also cited CDC data that supports the benefits of the bivalent shot on hospitalization in older adults. During November, hospitalization of adults 65 and above was 2.5 times higher for those vaccinated who did not get the booster, compared to those who got the updated bivalent booster.
Boosters do matter, Dr. Offit says. “But not for all.” In a perspective published Jan. 11 in the New England Journal of Medicine – the same issue that published the two studies finding few differences between the original and bivalent – Dr. Offit wrote that boosting is best reserved for vulnerable groups.
Chasing the variants with a bivalent vaccine, he says, “has not panned out. There remains no evidence that a bivalent vaccine is any better than what we had. Please, show me the data that one is better than the other.”
Dr. Offit believes the goal should not be to prevent all symptomatic infections in healthy, young people by boosting them “with vaccines containing mRNA from strains that might disappear a few months later.”
The CDC needs to parse the data by subgroups, Dr. Offit says. “The critical question is, ‘Who gets hospitalized and who is dying? Who are they?’ ”
That data should take into account age, ethnicity, vaccine history, and other factors, Dr. Offit says, because right now, there is no great data to say, “OK, everyone gets a boost.”
Future vaccine costs
Another debate – for not only current boosters but future ones, too – centers on cost. Without congressional action to fund more vaccines, vaccine makers have suggested their prices may reach $130 a dose, compared with the average $20-per-dose cost the federal government pays now, according to a Kaiser Family Foundation report.
The government has spent more than $30 billion on COVID-19 vaccines, including the bivalent, to provide them free of charge.
The suggested price increase infuriated many. On Jan. 10, Sen. Bernie Sanders (I-Vt.), incoming chair of the Senate Committee on Health, Education, Labor and Pensions, sent a letter to Moderna CEO Stéphane Bancel, urging him to reconsider and refrain from any price increase.
“The huge increase in price that you have proposed will have a significantly negative impact on the budgets of Medicaid, Medicare and other government programs that will continue covering the vaccine without cost-sharing for patients.”
He pointed out, too, the $19 billion in profits Moderna has made over the past 2 years.
While most people with health insurance would likely still get the vaccines and booster for free, according to the Kaiser analysis, will a higher price discourage people from keeping up with recommended vaccinations, including a possible new booster?
“I think so, yes,” Dr. Hotez says, noting that vaccine reluctance is high as it is, even with free vaccinations and easy access.
“The government is balking at paying for the boosters,” he says. “I think it’s very tone deaf from the pharmaceutical companies [to increase the price]. Given all the help they’ve gotten from the American people, I think they should not be gouging at this point.”
He noted that the federal government provided not just money to the companies for the vaccines, but a “glide path” through the FDA for the vaccine approvals.
Are new, variant-specific boosters coming?
Are Moderna, Pfizer-BioNTech, and others developing more variant-specific vaccines, boosters, or other advances?
Novavax, approved in July 2022 as a primary series and in some cases as a booster, is “also developing an Omicron-containing bivalent vaccine at the direction of public health agencies,” says spokesperson Alison Chartan.
Pfizer responded: “When and if we have something to share we will let you know.”
Moderna did not respond.
A version of this article first appeared on WebMD.com.
For some people who have received a two-dose primary series and all the recommended boosters, that could mean a sixth shot since COVID-19 vaccines became available. But is even that enough (or too much)?
At this point, no one knows for sure, but new guidance may be on the docket.
On Jan. 26, the FDA’s Vaccines and Related Biological Products Advisory Committee is meeting. On the agenda is discussion about plans for future vaccinations for COVID-19.The committee, made up of external advisers, evaluates data on vaccines and other products for the agency.
According to the FDA announcement, after the meeting, “the FDA will consider whether to recommend adjustments to the current authorizations and approvals, and the FDA will consider the most efficient and transparent process to use for selection of strains for inclusion in the primary and booster vaccines.”
From there, the CDC will take up the issue and decide on recommendations.
The issue is important, as more than 550 Americans a day are still dying from COVID-19, as of the week ending Jan. 13, the CDC reported. That’s up from 346 a day for the week ending Dec. 28.
Yet, uptake of the newest vaccine, the bivalent booster, has been slow. As of Jan. 11, just 15.9% of the population 5 years and up has gotten it; for those most vulnerable to COVID19 – those 65 and up – the number is just 39%.
COVID vaccines, 2023 and beyond
Meanwhile, infectious disease experts have widely differing views on what the vaccination landscape of 2023 and beyond should look like. Among the areas of disagreement are how effective the bivalent vaccine is, which people most need another shot, and what type of vaccine is best.
“I think we probably will need another booster,” says Peter Hotez, MD, PhD, dean of the National School of Tropical Medicine at Baylor College of Medicine, and codirector of the Center for Vaccine Development at Texas Children’s Hospital in Houston. “The question is, what is it going to be? Is it going to be the same bivalent that we just got, or will it be a new bivalent or even a trivalent?”
The trivalent booster, he suggested, might include something more protective against XBB.1.5.
The bivalent booster gives “broadened immunity” that is improved from the original booster shots, says Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, WebMD’s sister site for health professionals.
In his publication Ground Truths, Dr. Topol on Jan. 11 explained how new data caused him to reverse his previously skeptical view of how the FDA authorized the bivalent vaccine in September without data on how it affected humans at the time.
Paul Offit, MD, director of the Vaccine Education Center and a professor of pediatrics at the Children’s Hospital of Philadelphia, is a member of the FDA advisory committee for vaccines. He still takes a dimmer view of more bivalent booster vaccines, at least as a blanket recommendation.
While he acknowledges that boosters can help some groups – such as older adults, people with multiple health conditions, and those with compromised immune systems – he opposes a recommendation that’s population-wide.
“People who fall into those three groups do benefit,” he says, “but the recommendation is everyone over 6 months get the bivalent, and what I’m asking is, ‘Where is the data that a healthy 12-year-old boy needs a booster to stay out of the hospital?’ ”
Evolving research
“We are trying to understand how to stay one step ahead rather than several steps behind [the virus],“ says Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota.
Among the key questions: How well can a vaccine work against a single subvariant, when no one can say for sure what the next predominant subvariant will be?
Much more research has become available recently about the bivalent vaccine and its effectiveness, Dr. Osterholm says. “The bivalent vaccine is working as well as we could have expected,” he says, especially in high-risk people and in those over age 65. “The challenge we have is, what does that mean going forward?”
In his review, Dr. Topol concludes: “There is now more than ample, highly consistent evidence via lab studies and clinical outcomes to support the bivalent’s benefit over the original booster.”
Among other evidence, he looked at eight studies, including four that used a live virus as part of the research. Six of the eight studies showed the bivalent booster is more effective against the BA.5 variant, compared with the original booster shots. Two others showed no real difference.
“The four live virus studies offer consistent evidence of broadened immunity for the BA.5 vaccine that is improved over the original booster shots,” Dr. Topol wrote. The evidence also found the bivalent antibody response superior against XBB, he wrote.
Dr. Topol also cited CDC data that supports the benefits of the bivalent shot on hospitalization in older adults. During November, hospitalization of adults 65 and above was 2.5 times higher for those vaccinated who did not get the booster, compared to those who got the updated bivalent booster.
Boosters do matter, Dr. Offit says. “But not for all.” In a perspective published Jan. 11 in the New England Journal of Medicine – the same issue that published the two studies finding few differences between the original and bivalent – Dr. Offit wrote that boosting is best reserved for vulnerable groups.
Chasing the variants with a bivalent vaccine, he says, “has not panned out. There remains no evidence that a bivalent vaccine is any better than what we had. Please, show me the data that one is better than the other.”
Dr. Offit believes the goal should not be to prevent all symptomatic infections in healthy, young people by boosting them “with vaccines containing mRNA from strains that might disappear a few months later.”
The CDC needs to parse the data by subgroups, Dr. Offit says. “The critical question is, ‘Who gets hospitalized and who is dying? Who are they?’ ”
That data should take into account age, ethnicity, vaccine history, and other factors, Dr. Offit says, because right now, there is no great data to say, “OK, everyone gets a boost.”
Future vaccine costs
Another debate – for not only current boosters but future ones, too – centers on cost. Without congressional action to fund more vaccines, vaccine makers have suggested their prices may reach $130 a dose, compared with the average $20-per-dose cost the federal government pays now, according to a Kaiser Family Foundation report.
The government has spent more than $30 billion on COVID-19 vaccines, including the bivalent, to provide them free of charge.
The suggested price increase infuriated many. On Jan. 10, Sen. Bernie Sanders (I-Vt.), incoming chair of the Senate Committee on Health, Education, Labor and Pensions, sent a letter to Moderna CEO Stéphane Bancel, urging him to reconsider and refrain from any price increase.
“The huge increase in price that you have proposed will have a significantly negative impact on the budgets of Medicaid, Medicare and other government programs that will continue covering the vaccine without cost-sharing for patients.”
He pointed out, too, the $19 billion in profits Moderna has made over the past 2 years.
While most people with health insurance would likely still get the vaccines and booster for free, according to the Kaiser analysis, will a higher price discourage people from keeping up with recommended vaccinations, including a possible new booster?
“I think so, yes,” Dr. Hotez says, noting that vaccine reluctance is high as it is, even with free vaccinations and easy access.
“The government is balking at paying for the boosters,” he says. “I think it’s very tone deaf from the pharmaceutical companies [to increase the price]. Given all the help they’ve gotten from the American people, I think they should not be gouging at this point.”
He noted that the federal government provided not just money to the companies for the vaccines, but a “glide path” through the FDA for the vaccine approvals.
Are new, variant-specific boosters coming?
Are Moderna, Pfizer-BioNTech, and others developing more variant-specific vaccines, boosters, or other advances?
Novavax, approved in July 2022 as a primary series and in some cases as a booster, is “also developing an Omicron-containing bivalent vaccine at the direction of public health agencies,” says spokesperson Alison Chartan.
Pfizer responded: “When and if we have something to share we will let you know.”
Moderna did not respond.
A version of this article first appeared on WebMD.com.
For some people who have received a two-dose primary series and all the recommended boosters, that could mean a sixth shot since COVID-19 vaccines became available. But is even that enough (or too much)?
At this point, no one knows for sure, but new guidance may be on the docket.
On Jan. 26, the FDA’s Vaccines and Related Biological Products Advisory Committee is meeting. On the agenda is discussion about plans for future vaccinations for COVID-19.The committee, made up of external advisers, evaluates data on vaccines and other products for the agency.
According to the FDA announcement, after the meeting, “the FDA will consider whether to recommend adjustments to the current authorizations and approvals, and the FDA will consider the most efficient and transparent process to use for selection of strains for inclusion in the primary and booster vaccines.”
From there, the CDC will take up the issue and decide on recommendations.
The issue is important, as more than 550 Americans a day are still dying from COVID-19, as of the week ending Jan. 13, the CDC reported. That’s up from 346 a day for the week ending Dec. 28.
Yet, uptake of the newest vaccine, the bivalent booster, has been slow. As of Jan. 11, just 15.9% of the population 5 years and up has gotten it; for those most vulnerable to COVID19 – those 65 and up – the number is just 39%.
COVID vaccines, 2023 and beyond
Meanwhile, infectious disease experts have widely differing views on what the vaccination landscape of 2023 and beyond should look like. Among the areas of disagreement are how effective the bivalent vaccine is, which people most need another shot, and what type of vaccine is best.
“I think we probably will need another booster,” says Peter Hotez, MD, PhD, dean of the National School of Tropical Medicine at Baylor College of Medicine, and codirector of the Center for Vaccine Development at Texas Children’s Hospital in Houston. “The question is, what is it going to be? Is it going to be the same bivalent that we just got, or will it be a new bivalent or even a trivalent?”
The trivalent booster, he suggested, might include something more protective against XBB.1.5.
The bivalent booster gives “broadened immunity” that is improved from the original booster shots, says Eric Topol, MD, founder and director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape, WebMD’s sister site for health professionals.
In his publication Ground Truths, Dr. Topol on Jan. 11 explained how new data caused him to reverse his previously skeptical view of how the FDA authorized the bivalent vaccine in September without data on how it affected humans at the time.
Paul Offit, MD, director of the Vaccine Education Center and a professor of pediatrics at the Children’s Hospital of Philadelphia, is a member of the FDA advisory committee for vaccines. He still takes a dimmer view of more bivalent booster vaccines, at least as a blanket recommendation.
While he acknowledges that boosters can help some groups – such as older adults, people with multiple health conditions, and those with compromised immune systems – he opposes a recommendation that’s population-wide.
“People who fall into those three groups do benefit,” he says, “but the recommendation is everyone over 6 months get the bivalent, and what I’m asking is, ‘Where is the data that a healthy 12-year-old boy needs a booster to stay out of the hospital?’ ”
Evolving research
“We are trying to understand how to stay one step ahead rather than several steps behind [the virus],“ says Michael Osterholm, PhD, director of the Center for Infectious Disease Research and Policy at the University of Minnesota.
Among the key questions: How well can a vaccine work against a single subvariant, when no one can say for sure what the next predominant subvariant will be?
Much more research has become available recently about the bivalent vaccine and its effectiveness, Dr. Osterholm says. “The bivalent vaccine is working as well as we could have expected,” he says, especially in high-risk people and in those over age 65. “The challenge we have is, what does that mean going forward?”
In his review, Dr. Topol concludes: “There is now more than ample, highly consistent evidence via lab studies and clinical outcomes to support the bivalent’s benefit over the original booster.”
Among other evidence, he looked at eight studies, including four that used a live virus as part of the research. Six of the eight studies showed the bivalent booster is more effective against the BA.5 variant, compared with the original booster shots. Two others showed no real difference.
“The four live virus studies offer consistent evidence of broadened immunity for the BA.5 vaccine that is improved over the original booster shots,” Dr. Topol wrote. The evidence also found the bivalent antibody response superior against XBB, he wrote.
Dr. Topol also cited CDC data that supports the benefits of the bivalent shot on hospitalization in older adults. During November, hospitalization of adults 65 and above was 2.5 times higher for those vaccinated who did not get the booster, compared to those who got the updated bivalent booster.
Boosters do matter, Dr. Offit says. “But not for all.” In a perspective published Jan. 11 in the New England Journal of Medicine – the same issue that published the two studies finding few differences between the original and bivalent – Dr. Offit wrote that boosting is best reserved for vulnerable groups.
Chasing the variants with a bivalent vaccine, he says, “has not panned out. There remains no evidence that a bivalent vaccine is any better than what we had. Please, show me the data that one is better than the other.”
Dr. Offit believes the goal should not be to prevent all symptomatic infections in healthy, young people by boosting them “with vaccines containing mRNA from strains that might disappear a few months later.”
The CDC needs to parse the data by subgroups, Dr. Offit says. “The critical question is, ‘Who gets hospitalized and who is dying? Who are they?’ ”
That data should take into account age, ethnicity, vaccine history, and other factors, Dr. Offit says, because right now, there is no great data to say, “OK, everyone gets a boost.”
Future vaccine costs
Another debate – for not only current boosters but future ones, too – centers on cost. Without congressional action to fund more vaccines, vaccine makers have suggested their prices may reach $130 a dose, compared with the average $20-per-dose cost the federal government pays now, according to a Kaiser Family Foundation report.
The government has spent more than $30 billion on COVID-19 vaccines, including the bivalent, to provide them free of charge.
The suggested price increase infuriated many. On Jan. 10, Sen. Bernie Sanders (I-Vt.), incoming chair of the Senate Committee on Health, Education, Labor and Pensions, sent a letter to Moderna CEO Stéphane Bancel, urging him to reconsider and refrain from any price increase.
“The huge increase in price that you have proposed will have a significantly negative impact on the budgets of Medicaid, Medicare and other government programs that will continue covering the vaccine without cost-sharing for patients.”
He pointed out, too, the $19 billion in profits Moderna has made over the past 2 years.
While most people with health insurance would likely still get the vaccines and booster for free, according to the Kaiser analysis, will a higher price discourage people from keeping up with recommended vaccinations, including a possible new booster?
“I think so, yes,” Dr. Hotez says, noting that vaccine reluctance is high as it is, even with free vaccinations and easy access.
“The government is balking at paying for the boosters,” he says. “I think it’s very tone deaf from the pharmaceutical companies [to increase the price]. Given all the help they’ve gotten from the American people, I think they should not be gouging at this point.”
He noted that the federal government provided not just money to the companies for the vaccines, but a “glide path” through the FDA for the vaccine approvals.
Are new, variant-specific boosters coming?
Are Moderna, Pfizer-BioNTech, and others developing more variant-specific vaccines, boosters, or other advances?
Novavax, approved in July 2022 as a primary series and in some cases as a booster, is “also developing an Omicron-containing bivalent vaccine at the direction of public health agencies,” says spokesperson Alison Chartan.
Pfizer responded: “When and if we have something to share we will let you know.”
Moderna did not respond.
A version of this article first appeared on WebMD.com.
Will your smartphone be the next doctor’s office?
A fingertip pressed against a phone’s camera lens can measure a heart rate. The microphone, kept by the bedside, can screen for sleep apnea. Even the speaker is being tapped, to monitor breathing using sonar technology.
In the best of this new world, the data is conveyed remotely to a medical professional for the convenience and comfort of the patient or, in some cases, to support a clinician without the need for costly hardware.
But using smartphones as diagnostic tools is a work in progress, experts say. Although doctors and their patients have found some real-world success in deploying the phone as a medical device, the overall potential remains unfulfilled and uncertain.
Smartphones come packed with sensors capable of monitoring a patient’s vital signs. They can help assess people for concussions, watch for atrial fibrillation, and conduct mental health wellness checks, to name the uses of a few nascent applications.
Companies and researchers eager to find medical applications for smartphone technology are tapping into modern phones’ built-in cameras and light sensors; microphones; accelerometers, which detect body movements; gyroscopes; and even speakers. The apps then use artificial intelligence software to analyze the collected sights and sounds to create an easy connection between patients and physicians. Earning potential and marketability are evidenced by the more than 350,000 digital health products available in app stores, according to a Grand View Research report.
“It’s very hard to put devices into the patient home or in the hospital, but everybody is just walking around with a cellphone that has a network connection,” said Dr. Andrew Gostine, CEO of the sensor network company Artisight. Most Americans own a smartphone, including more than 60% of people 65 and over, an increase from just 13% a decade ago, according the Pew Research Center. The COVID-19 pandemic has also pushed people to become more comfortable with virtual care.
Some of these products have sought FDA clearance to be marketed as a medical device. That way, if patients must pay to use the software, health insurers are more likely to cover at least part of the cost. Other products are designated as exempt from this regulatory process, placed in the same clinical classification as a Band-Aid. But how the agency handles AI and machine learning–based medical devices is still being adjusted to reflect software’s adaptive nature.
Ensuring accuracy and clinical validation is crucial to securing buy-in from health care providers. And many tools still need fine-tuning, said Eugene Yang, MD, a professor of medicine at the University of Washington, Seattle. Currently, Dr. Yang is testing contactless measurement of blood pressure, heart rate, and oxygen saturation gleaned remotely via Zoom camera footage of a patient’s face.
Judging these new technologies is difficult because they rely on algorithms built by machine learning and artificial intelligence to collect data, rather than the physical tools typically used in hospitals. So researchers cannot “compare apples to apples” with medical industry standards, Dr. Yang said. Failure to build in such assurances undermines the technology’s ultimate goals of easing costs and access because a doctor still must verify results.
“False positives and false negatives lead to more testing and more cost to the health care system,” he said.
Big tech companies like Google have heavily invested in researching this kind of technology, catering to clinicians and in-home caregivers, as well as consumers. Currently, in the Google Fit app, users can check their heart rate by placing their finger on the rear-facing camera lens or track their breathing rate using the front-facing camera.
“If you took the sensor out of the phone and out of a clinical device, they are probably the same thing,” said Shwetak Patel, director of health technologies at Google and a professor of electrical and computer engineering at the University of Washington.
Google’s research uses machine learning and computer vision, a field within AI based on information from visual inputs like videos or images. So instead of using a blood pressure cuff, for example, the algorithm can interpret slight visual changes to the body that serve as proxies and biosignals for a patient’s blood pressure, Mr. Patel said.
Google is also investigating the effectiveness of the built-in microphone for detecting heartbeats and murmurs and using the camera to preserve eyesight by screening for diabetic eye disease, according to information the company published last year.
The tech giant recently purchased Sound Life Sciences, a Seattle startup with an FDA-cleared sonar technology app. It uses a smart device’s speaker to bounce inaudible pulses off a patient’s body to identify movement and monitor breathing.
Binah.ai, based in Israel, is another company using the smartphone camera to calculate vital signs. Its software looks at the region around the eyes, where the skin is a bit thinner, and analyzes the light reflecting off blood vessels back to the lens. The company is wrapping up a U.S. clinical trial and marketing its wellness app directly to insurers and other health companies, said company spokesperson Mona Popilian-Yona.
The applications even reach into disciplines such as optometry and mental health:
- With the microphone, Canary Speech uses the same underlying technology as Amazon’s Alexa to analyze patients’ voices for mental health conditions. The software can integrate with telemedicine appointments and allow clinicians to screen for anxiety and depression using a library of vocal biomarkers and predictive analytics, said Henry O’Connell, the company’s CEO.
- Australia-based ResApp Health last year for its iPhone app that screens for moderate to severe obstructive sleep apnea by listening to breathing and snoring. SleepCheckRx, which will require a prescription, is minimally invasive compared with sleep studies currently used to diagnose sleep apnea. Those can cost thousands of dollars and require an array of tests.
- Brightlamp’s Reflex app is a clinical decision support tool for helping manage concussions and vision rehabilitation, among other things. Using an iPad’s or iPhone’s camera, the mobile app measures how a person’s pupils react to changes in light. Through machine learning analysis, the imagery gives practitioners data points for evaluating patients. Brightlamp sells directly to health care providers and is being used in more than 230 clinics. Clinicians pay a $400 standard annual fee per account, which is currently not covered by insurance. The Department of Defense has an ongoing clinical trial using Reflex.
In some cases, such as with the Reflex app, the data is processed directly on the phone – rather than in the cloud, Brightlamp CEO Kurtis Sluss said. By processing everything on the device, the app avoids running into privacy issues, as streaming data elsewhere requires patient consent.
But algorithms need to be trained and tested by collecting reams of data, and that is an ongoing process.
Researchers, for example, have found that some computer vision applications, like heart rate or blood pressure monitoring, can be less accurate for darker skin. Studies are underway to find better solutions.
Small algorithm glitches can also produce false alarms and frighten patients enough to keep widespread adoption out of reach. For example, Apple’s new car-crash detection feature, available on both the latest iPhone and Apple Watch, was set off when people were riding roller coasters and automatically dialed 911.
“We’re not there yet,” Dr. Yang said. “That’s the bottom line.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
A fingertip pressed against a phone’s camera lens can measure a heart rate. The microphone, kept by the bedside, can screen for sleep apnea. Even the speaker is being tapped, to monitor breathing using sonar technology.
In the best of this new world, the data is conveyed remotely to a medical professional for the convenience and comfort of the patient or, in some cases, to support a clinician without the need for costly hardware.
But using smartphones as diagnostic tools is a work in progress, experts say. Although doctors and their patients have found some real-world success in deploying the phone as a medical device, the overall potential remains unfulfilled and uncertain.
Smartphones come packed with sensors capable of monitoring a patient’s vital signs. They can help assess people for concussions, watch for atrial fibrillation, and conduct mental health wellness checks, to name the uses of a few nascent applications.
Companies and researchers eager to find medical applications for smartphone technology are tapping into modern phones’ built-in cameras and light sensors; microphones; accelerometers, which detect body movements; gyroscopes; and even speakers. The apps then use artificial intelligence software to analyze the collected sights and sounds to create an easy connection between patients and physicians. Earning potential and marketability are evidenced by the more than 350,000 digital health products available in app stores, according to a Grand View Research report.
“It’s very hard to put devices into the patient home or in the hospital, but everybody is just walking around with a cellphone that has a network connection,” said Dr. Andrew Gostine, CEO of the sensor network company Artisight. Most Americans own a smartphone, including more than 60% of people 65 and over, an increase from just 13% a decade ago, according the Pew Research Center. The COVID-19 pandemic has also pushed people to become more comfortable with virtual care.
Some of these products have sought FDA clearance to be marketed as a medical device. That way, if patients must pay to use the software, health insurers are more likely to cover at least part of the cost. Other products are designated as exempt from this regulatory process, placed in the same clinical classification as a Band-Aid. But how the agency handles AI and machine learning–based medical devices is still being adjusted to reflect software’s adaptive nature.
Ensuring accuracy and clinical validation is crucial to securing buy-in from health care providers. And many tools still need fine-tuning, said Eugene Yang, MD, a professor of medicine at the University of Washington, Seattle. Currently, Dr. Yang is testing contactless measurement of blood pressure, heart rate, and oxygen saturation gleaned remotely via Zoom camera footage of a patient’s face.
Judging these new technologies is difficult because they rely on algorithms built by machine learning and artificial intelligence to collect data, rather than the physical tools typically used in hospitals. So researchers cannot “compare apples to apples” with medical industry standards, Dr. Yang said. Failure to build in such assurances undermines the technology’s ultimate goals of easing costs and access because a doctor still must verify results.
“False positives and false negatives lead to more testing and more cost to the health care system,” he said.
Big tech companies like Google have heavily invested in researching this kind of technology, catering to clinicians and in-home caregivers, as well as consumers. Currently, in the Google Fit app, users can check their heart rate by placing their finger on the rear-facing camera lens or track their breathing rate using the front-facing camera.
“If you took the sensor out of the phone and out of a clinical device, they are probably the same thing,” said Shwetak Patel, director of health technologies at Google and a professor of electrical and computer engineering at the University of Washington.
Google’s research uses machine learning and computer vision, a field within AI based on information from visual inputs like videos or images. So instead of using a blood pressure cuff, for example, the algorithm can interpret slight visual changes to the body that serve as proxies and biosignals for a patient’s blood pressure, Mr. Patel said.
Google is also investigating the effectiveness of the built-in microphone for detecting heartbeats and murmurs and using the camera to preserve eyesight by screening for diabetic eye disease, according to information the company published last year.
The tech giant recently purchased Sound Life Sciences, a Seattle startup with an FDA-cleared sonar technology app. It uses a smart device’s speaker to bounce inaudible pulses off a patient’s body to identify movement and monitor breathing.
Binah.ai, based in Israel, is another company using the smartphone camera to calculate vital signs. Its software looks at the region around the eyes, where the skin is a bit thinner, and analyzes the light reflecting off blood vessels back to the lens. The company is wrapping up a U.S. clinical trial and marketing its wellness app directly to insurers and other health companies, said company spokesperson Mona Popilian-Yona.
The applications even reach into disciplines such as optometry and mental health:
- With the microphone, Canary Speech uses the same underlying technology as Amazon’s Alexa to analyze patients’ voices for mental health conditions. The software can integrate with telemedicine appointments and allow clinicians to screen for anxiety and depression using a library of vocal biomarkers and predictive analytics, said Henry O’Connell, the company’s CEO.
- Australia-based ResApp Health last year for its iPhone app that screens for moderate to severe obstructive sleep apnea by listening to breathing and snoring. SleepCheckRx, which will require a prescription, is minimally invasive compared with sleep studies currently used to diagnose sleep apnea. Those can cost thousands of dollars and require an array of tests.
- Brightlamp’s Reflex app is a clinical decision support tool for helping manage concussions and vision rehabilitation, among other things. Using an iPad’s or iPhone’s camera, the mobile app measures how a person’s pupils react to changes in light. Through machine learning analysis, the imagery gives practitioners data points for evaluating patients. Brightlamp sells directly to health care providers and is being used in more than 230 clinics. Clinicians pay a $400 standard annual fee per account, which is currently not covered by insurance. The Department of Defense has an ongoing clinical trial using Reflex.
In some cases, such as with the Reflex app, the data is processed directly on the phone – rather than in the cloud, Brightlamp CEO Kurtis Sluss said. By processing everything on the device, the app avoids running into privacy issues, as streaming data elsewhere requires patient consent.
But algorithms need to be trained and tested by collecting reams of data, and that is an ongoing process.
Researchers, for example, have found that some computer vision applications, like heart rate or blood pressure monitoring, can be less accurate for darker skin. Studies are underway to find better solutions.
Small algorithm glitches can also produce false alarms and frighten patients enough to keep widespread adoption out of reach. For example, Apple’s new car-crash detection feature, available on both the latest iPhone and Apple Watch, was set off when people were riding roller coasters and automatically dialed 911.
“We’re not there yet,” Dr. Yang said. “That’s the bottom line.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
A fingertip pressed against a phone’s camera lens can measure a heart rate. The microphone, kept by the bedside, can screen for sleep apnea. Even the speaker is being tapped, to monitor breathing using sonar technology.
In the best of this new world, the data is conveyed remotely to a medical professional for the convenience and comfort of the patient or, in some cases, to support a clinician without the need for costly hardware.
But using smartphones as diagnostic tools is a work in progress, experts say. Although doctors and their patients have found some real-world success in deploying the phone as a medical device, the overall potential remains unfulfilled and uncertain.
Smartphones come packed with sensors capable of monitoring a patient’s vital signs. They can help assess people for concussions, watch for atrial fibrillation, and conduct mental health wellness checks, to name the uses of a few nascent applications.
Companies and researchers eager to find medical applications for smartphone technology are tapping into modern phones’ built-in cameras and light sensors; microphones; accelerometers, which detect body movements; gyroscopes; and even speakers. The apps then use artificial intelligence software to analyze the collected sights and sounds to create an easy connection between patients and physicians. Earning potential and marketability are evidenced by the more than 350,000 digital health products available in app stores, according to a Grand View Research report.
“It’s very hard to put devices into the patient home or in the hospital, but everybody is just walking around with a cellphone that has a network connection,” said Dr. Andrew Gostine, CEO of the sensor network company Artisight. Most Americans own a smartphone, including more than 60% of people 65 and over, an increase from just 13% a decade ago, according the Pew Research Center. The COVID-19 pandemic has also pushed people to become more comfortable with virtual care.
Some of these products have sought FDA clearance to be marketed as a medical device. That way, if patients must pay to use the software, health insurers are more likely to cover at least part of the cost. Other products are designated as exempt from this regulatory process, placed in the same clinical classification as a Band-Aid. But how the agency handles AI and machine learning–based medical devices is still being adjusted to reflect software’s adaptive nature.
Ensuring accuracy and clinical validation is crucial to securing buy-in from health care providers. And many tools still need fine-tuning, said Eugene Yang, MD, a professor of medicine at the University of Washington, Seattle. Currently, Dr. Yang is testing contactless measurement of blood pressure, heart rate, and oxygen saturation gleaned remotely via Zoom camera footage of a patient’s face.
Judging these new technologies is difficult because they rely on algorithms built by machine learning and artificial intelligence to collect data, rather than the physical tools typically used in hospitals. So researchers cannot “compare apples to apples” with medical industry standards, Dr. Yang said. Failure to build in such assurances undermines the technology’s ultimate goals of easing costs and access because a doctor still must verify results.
“False positives and false negatives lead to more testing and more cost to the health care system,” he said.
Big tech companies like Google have heavily invested in researching this kind of technology, catering to clinicians and in-home caregivers, as well as consumers. Currently, in the Google Fit app, users can check their heart rate by placing their finger on the rear-facing camera lens or track their breathing rate using the front-facing camera.
“If you took the sensor out of the phone and out of a clinical device, they are probably the same thing,” said Shwetak Patel, director of health technologies at Google and a professor of electrical and computer engineering at the University of Washington.
Google’s research uses machine learning and computer vision, a field within AI based on information from visual inputs like videos or images. So instead of using a blood pressure cuff, for example, the algorithm can interpret slight visual changes to the body that serve as proxies and biosignals for a patient’s blood pressure, Mr. Patel said.
Google is also investigating the effectiveness of the built-in microphone for detecting heartbeats and murmurs and using the camera to preserve eyesight by screening for diabetic eye disease, according to information the company published last year.
The tech giant recently purchased Sound Life Sciences, a Seattle startup with an FDA-cleared sonar technology app. It uses a smart device’s speaker to bounce inaudible pulses off a patient’s body to identify movement and monitor breathing.
Binah.ai, based in Israel, is another company using the smartphone camera to calculate vital signs. Its software looks at the region around the eyes, where the skin is a bit thinner, and analyzes the light reflecting off blood vessels back to the lens. The company is wrapping up a U.S. clinical trial and marketing its wellness app directly to insurers and other health companies, said company spokesperson Mona Popilian-Yona.
The applications even reach into disciplines such as optometry and mental health:
- With the microphone, Canary Speech uses the same underlying technology as Amazon’s Alexa to analyze patients’ voices for mental health conditions. The software can integrate with telemedicine appointments and allow clinicians to screen for anxiety and depression using a library of vocal biomarkers and predictive analytics, said Henry O’Connell, the company’s CEO.
- Australia-based ResApp Health last year for its iPhone app that screens for moderate to severe obstructive sleep apnea by listening to breathing and snoring. SleepCheckRx, which will require a prescription, is minimally invasive compared with sleep studies currently used to diagnose sleep apnea. Those can cost thousands of dollars and require an array of tests.
- Brightlamp’s Reflex app is a clinical decision support tool for helping manage concussions and vision rehabilitation, among other things. Using an iPad’s or iPhone’s camera, the mobile app measures how a person’s pupils react to changes in light. Through machine learning analysis, the imagery gives practitioners data points for evaluating patients. Brightlamp sells directly to health care providers and is being used in more than 230 clinics. Clinicians pay a $400 standard annual fee per account, which is currently not covered by insurance. The Department of Defense has an ongoing clinical trial using Reflex.
In some cases, such as with the Reflex app, the data is processed directly on the phone – rather than in the cloud, Brightlamp CEO Kurtis Sluss said. By processing everything on the device, the app avoids running into privacy issues, as streaming data elsewhere requires patient consent.
But algorithms need to be trained and tested by collecting reams of data, and that is an ongoing process.
Researchers, for example, have found that some computer vision applications, like heart rate or blood pressure monitoring, can be less accurate for darker skin. Studies are underway to find better solutions.
Small algorithm glitches can also produce false alarms and frighten patients enough to keep widespread adoption out of reach. For example, Apple’s new car-crash detection feature, available on both the latest iPhone and Apple Watch, was set off when people were riding roller coasters and automatically dialed 911.
“We’re not there yet,” Dr. Yang said. “That’s the bottom line.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.