User login
FREEDOM COVID: Full-dose anticoagulation cut mortality but missed primary endpoint
Study conducted in noncritically ill
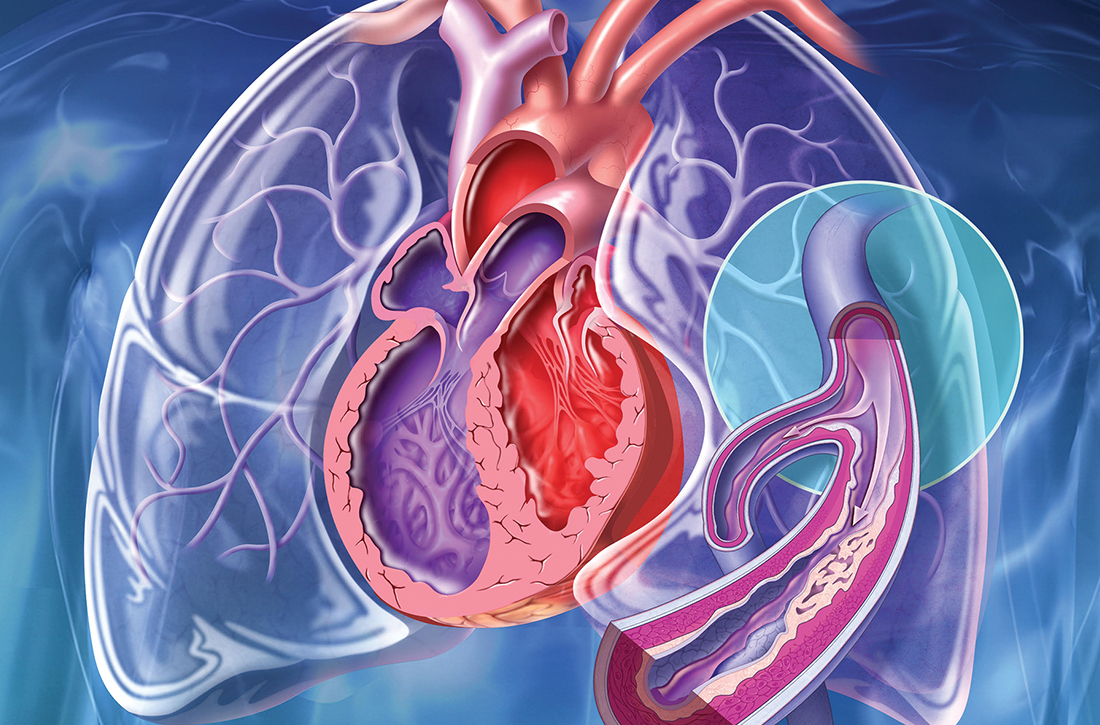
NEW ORLEANS – In the international FREEDOM COVID trial that randomized non–critically ill hospitalized patients, a therapeutic dose of anticoagulation relative to a prophylactic dose significantly reduced death from COVID-19 at 30 days, even as a larger composite primary endpoint was missed.
The mortality reduction suggests therapeutic-dose anticoagulation “may improve outcomes in non–critically ill patients hospitalized with COVID-19 who are at increased risk for adverse events but do not yet require ICU-level of care,” reported Valentin Fuster, MD, PhD, at the joint scientific sessions of the American College of Cardiology and the World Heart Federation.
These data provide a suggestion rather than a demonstration of benefit because the primary composite endpoint of all-cause mortality, intubation requiring mechanical ventilation, systemic thromboembolism or ischemic stroke at 30 days was not met. Although this 30-day outcome was lower on the therapeutic dose (11.3% vs. 13.2%), the difference was only a trend (hazard ratio, 0.85; P = .11), said Dr. Fuster, physician-in-chief, Mount Sinai Hospital, New York.
Missed primary endpoint blamed on low events
The declining severity of more recent COVID-19 variants (the trial was conducted from August 2022 to September 2022) might be one explanation that the primary endpoint was not met, but the more likely explanation is the relatively good health status – and therefore a low risk of events – among patients randomized in India, 1 of 10 participating countries.
India accounted for roughly 40% of the total number of 3,398 patients in the intention-to-treat population. In India, the rates of events were 0.7 and 1.3 in the prophylactic and therapeutic anticoagulation arms, respectively. In contrast, they were 17.5 and 9.5, respectively in the United States. In combined data from the other eight countries, the rates were 22.78 and 20.4, respectively.
“These results emphasize that varying country-specific thresholds for hospitalization may affect patient prognosis and the potential utility of advanced therapies” Dr. Fuster said.
In fact, the therapeutic anticoagulation was linked to a nonsignificant twofold increase in the risk of the primary outcome in India (HR, 2.01; 95% confidence interval, 0.57-7.13) when outcomes were stratified by country. In the United States, where there was a much higher incidence of events, therapeutic anticoagulation was associated with a nearly 50% reduction (HR, 0.53; 95% CI, 0.31-0.91).
In the remaining countries, which included those in Latin America and Europe as well as the city of Hong Kong, the primary outcome was reduced numerically but not statistically by therapeutic relative to prophylactic anticoagulation (HR, 0.89; 95% CI, 0.71-1.11).
Enoxaparin and apixaban are studied
In FREEDOM COVID, patients were randomized to a therapeutic dose of the low-molecular-weight heparin (LMWH) enoxaparin (1 mg/kg every 12 hours), a prophylactic dose of enoxaparin (40 mg once daily), or a therapeutic dose of the direct factor Xa inhibitor apixaban (5 mg every 12 hours). Lower doses of enoxaparin and apixaban were used for those with renal impairment, and lower doses of apixaban were employed for elderly patients (≥ 80 years) and those with low body weight (≤ 60 kg).
The major inclusion criteria were confirmed COVID-19 infection with symptomatic systemic involvement. The major exclusion criteria were need for ICU level of care or active bleeding.
The therapeutic anticoagulation arms performed similarly and were combined for comparison to the prophylactic arm. Despite the failure to show a difference in the primary outcome, the rate of 30-day mortality was substantially lower in the therapeutic arm (4.9% vs. 7.0%), translating into a 30% risk reduction (HR, 0.70; P = .01).
Therapeutic anticoagulation was also associated with a lower rate of intubation/mechanical ventilation (6.4% vs. 8.4%) that reached statistical significance (HR, 0.75; P = .03). The risk reduction was also significant for a combination of these endpoints (HR, 0.77; P = .03).
The lower proportion of patients who eventually required ICU-level of care (9.9% vs. 11.7%) showed a trend in favor of therapeutic anticoagulation (HR, 0.84; P = .11).
Bleeding rates did not differ between arms
Bleeding Academic Research Consortium major bleeding types 3 and 5 were slightly numerically higher in the group randomized to therapeutic enoxaparin (0.5%) than prophylactic enoxaparin (0.1%) and therapeutic apixaban (0.3%), but the differences between any groups were not significant.
Numerous anticoagulation trials in patients with COVID-19 have been published previously. One 2021 trial published in the New England Journal of Medicine also suggested benefit from a therapeutic relative to prophylactic anticoagulation. In that trial, which compared heparin to usual-care thromboprophylaxis, benefits were derived from a Bayesian analysis. Significant differences were not shown for death or other major outcome assessed individually.
Even though this more recent trial missed its primary endpoint, Gregg Stone, MD, a coauthor of this study and a colleague of Dr. Fuster at the Mount Sinai School of Medicine, New York, reiterated that these results support routine anticoagulation in hospitalized COVID-19 patients.
“These are robust reductions in mortality and intubation rates, which are the most serious outcomes,” said Dr. Stone, who is first author of the paper, which was published in the Journal of the American College of Cardiology immediately after Dr. Fuster’s presentation.
COVID-19 has proven to be a very thrombogenic virus, but the literature has not been wholly consistent on which anticoagulation treatment provides the best balance of benefits and risks, according to Julia Grapsa, MD, PhD, attending cardiologist, Guys and St. Thomas Hospital, London. She said that this randomized trial, despite its failure to meet the primary endpoint, is useful.
“This demonstrates that a therapeutic dose of enoxaparin is likely to improve outcomes over a prophylactic dose with a low risk of bleeding,” Dr. Grapsa said. On the basis of the randomized study, “I feel more confident with this approach.”
Dr. Fuster reported no potential conflicts of interest. Dr. Stone has financial relationships with more than 30 companies that make pharmaceuticals and medical devices. Dr. Grapsa reported no potential conflicts of interest.
Study conducted in noncritically ill
Study conducted in noncritically ill
NEW ORLEANS – In the international FREEDOM COVID trial that randomized non–critically ill hospitalized patients, a therapeutic dose of anticoagulation relative to a prophylactic dose significantly reduced death from COVID-19 at 30 days, even as a larger composite primary endpoint was missed.
The mortality reduction suggests therapeutic-dose anticoagulation “may improve outcomes in non–critically ill patients hospitalized with COVID-19 who are at increased risk for adverse events but do not yet require ICU-level of care,” reported Valentin Fuster, MD, PhD, at the joint scientific sessions of the American College of Cardiology and the World Heart Federation.
These data provide a suggestion rather than a demonstration of benefit because the primary composite endpoint of all-cause mortality, intubation requiring mechanical ventilation, systemic thromboembolism or ischemic stroke at 30 days was not met. Although this 30-day outcome was lower on the therapeutic dose (11.3% vs. 13.2%), the difference was only a trend (hazard ratio, 0.85; P = .11), said Dr. Fuster, physician-in-chief, Mount Sinai Hospital, New York.
Missed primary endpoint blamed on low events
The declining severity of more recent COVID-19 variants (the trial was conducted from August 2022 to September 2022) might be one explanation that the primary endpoint was not met, but the more likely explanation is the relatively good health status – and therefore a low risk of events – among patients randomized in India, 1 of 10 participating countries.
India accounted for roughly 40% of the total number of 3,398 patients in the intention-to-treat population. In India, the rates of events were 0.7 and 1.3 in the prophylactic and therapeutic anticoagulation arms, respectively. In contrast, they were 17.5 and 9.5, respectively in the United States. In combined data from the other eight countries, the rates were 22.78 and 20.4, respectively.
“These results emphasize that varying country-specific thresholds for hospitalization may affect patient prognosis and the potential utility of advanced therapies” Dr. Fuster said.
In fact, the therapeutic anticoagulation was linked to a nonsignificant twofold increase in the risk of the primary outcome in India (HR, 2.01; 95% confidence interval, 0.57-7.13) when outcomes were stratified by country. In the United States, where there was a much higher incidence of events, therapeutic anticoagulation was associated with a nearly 50% reduction (HR, 0.53; 95% CI, 0.31-0.91).
In the remaining countries, which included those in Latin America and Europe as well as the city of Hong Kong, the primary outcome was reduced numerically but not statistically by therapeutic relative to prophylactic anticoagulation (HR, 0.89; 95% CI, 0.71-1.11).
Enoxaparin and apixaban are studied
In FREEDOM COVID, patients were randomized to a therapeutic dose of the low-molecular-weight heparin (LMWH) enoxaparin (1 mg/kg every 12 hours), a prophylactic dose of enoxaparin (40 mg once daily), or a therapeutic dose of the direct factor Xa inhibitor apixaban (5 mg every 12 hours). Lower doses of enoxaparin and apixaban were used for those with renal impairment, and lower doses of apixaban were employed for elderly patients (≥ 80 years) and those with low body weight (≤ 60 kg).
The major inclusion criteria were confirmed COVID-19 infection with symptomatic systemic involvement. The major exclusion criteria were need for ICU level of care or active bleeding.
The therapeutic anticoagulation arms performed similarly and were combined for comparison to the prophylactic arm. Despite the failure to show a difference in the primary outcome, the rate of 30-day mortality was substantially lower in the therapeutic arm (4.9% vs. 7.0%), translating into a 30% risk reduction (HR, 0.70; P = .01).
Therapeutic anticoagulation was also associated with a lower rate of intubation/mechanical ventilation (6.4% vs. 8.4%) that reached statistical significance (HR, 0.75; P = .03). The risk reduction was also significant for a combination of these endpoints (HR, 0.77; P = .03).
The lower proportion of patients who eventually required ICU-level of care (9.9% vs. 11.7%) showed a trend in favor of therapeutic anticoagulation (HR, 0.84; P = .11).
Bleeding rates did not differ between arms
Bleeding Academic Research Consortium major bleeding types 3 and 5 were slightly numerically higher in the group randomized to therapeutic enoxaparin (0.5%) than prophylactic enoxaparin (0.1%) and therapeutic apixaban (0.3%), but the differences between any groups were not significant.
Numerous anticoagulation trials in patients with COVID-19 have been published previously. One 2021 trial published in the New England Journal of Medicine also suggested benefit from a therapeutic relative to prophylactic anticoagulation. In that trial, which compared heparin to usual-care thromboprophylaxis, benefits were derived from a Bayesian analysis. Significant differences were not shown for death or other major outcome assessed individually.
Even though this more recent trial missed its primary endpoint, Gregg Stone, MD, a coauthor of this study and a colleague of Dr. Fuster at the Mount Sinai School of Medicine, New York, reiterated that these results support routine anticoagulation in hospitalized COVID-19 patients.
“These are robust reductions in mortality and intubation rates, which are the most serious outcomes,” said Dr. Stone, who is first author of the paper, which was published in the Journal of the American College of Cardiology immediately after Dr. Fuster’s presentation.
COVID-19 has proven to be a very thrombogenic virus, but the literature has not been wholly consistent on which anticoagulation treatment provides the best balance of benefits and risks, according to Julia Grapsa, MD, PhD, attending cardiologist, Guys and St. Thomas Hospital, London. She said that this randomized trial, despite its failure to meet the primary endpoint, is useful.
“This demonstrates that a therapeutic dose of enoxaparin is likely to improve outcomes over a prophylactic dose with a low risk of bleeding,” Dr. Grapsa said. On the basis of the randomized study, “I feel more confident with this approach.”
Dr. Fuster reported no potential conflicts of interest. Dr. Stone has financial relationships with more than 30 companies that make pharmaceuticals and medical devices. Dr. Grapsa reported no potential conflicts of interest.
NEW ORLEANS – In the international FREEDOM COVID trial that randomized non–critically ill hospitalized patients, a therapeutic dose of anticoagulation relative to a prophylactic dose significantly reduced death from COVID-19 at 30 days, even as a larger composite primary endpoint was missed.
The mortality reduction suggests therapeutic-dose anticoagulation “may improve outcomes in non–critically ill patients hospitalized with COVID-19 who are at increased risk for adverse events but do not yet require ICU-level of care,” reported Valentin Fuster, MD, PhD, at the joint scientific sessions of the American College of Cardiology and the World Heart Federation.
These data provide a suggestion rather than a demonstration of benefit because the primary composite endpoint of all-cause mortality, intubation requiring mechanical ventilation, systemic thromboembolism or ischemic stroke at 30 days was not met. Although this 30-day outcome was lower on the therapeutic dose (11.3% vs. 13.2%), the difference was only a trend (hazard ratio, 0.85; P = .11), said Dr. Fuster, physician-in-chief, Mount Sinai Hospital, New York.
Missed primary endpoint blamed on low events
The declining severity of more recent COVID-19 variants (the trial was conducted from August 2022 to September 2022) might be one explanation that the primary endpoint was not met, but the more likely explanation is the relatively good health status – and therefore a low risk of events – among patients randomized in India, 1 of 10 participating countries.
India accounted for roughly 40% of the total number of 3,398 patients in the intention-to-treat population. In India, the rates of events were 0.7 and 1.3 in the prophylactic and therapeutic anticoagulation arms, respectively. In contrast, they were 17.5 and 9.5, respectively in the United States. In combined data from the other eight countries, the rates were 22.78 and 20.4, respectively.
“These results emphasize that varying country-specific thresholds for hospitalization may affect patient prognosis and the potential utility of advanced therapies” Dr. Fuster said.
In fact, the therapeutic anticoagulation was linked to a nonsignificant twofold increase in the risk of the primary outcome in India (HR, 2.01; 95% confidence interval, 0.57-7.13) when outcomes were stratified by country. In the United States, where there was a much higher incidence of events, therapeutic anticoagulation was associated with a nearly 50% reduction (HR, 0.53; 95% CI, 0.31-0.91).
In the remaining countries, which included those in Latin America and Europe as well as the city of Hong Kong, the primary outcome was reduced numerically but not statistically by therapeutic relative to prophylactic anticoagulation (HR, 0.89; 95% CI, 0.71-1.11).
Enoxaparin and apixaban are studied
In FREEDOM COVID, patients were randomized to a therapeutic dose of the low-molecular-weight heparin (LMWH) enoxaparin (1 mg/kg every 12 hours), a prophylactic dose of enoxaparin (40 mg once daily), or a therapeutic dose of the direct factor Xa inhibitor apixaban (5 mg every 12 hours). Lower doses of enoxaparin and apixaban were used for those with renal impairment, and lower doses of apixaban were employed for elderly patients (≥ 80 years) and those with low body weight (≤ 60 kg).
The major inclusion criteria were confirmed COVID-19 infection with symptomatic systemic involvement. The major exclusion criteria were need for ICU level of care or active bleeding.
The therapeutic anticoagulation arms performed similarly and were combined for comparison to the prophylactic arm. Despite the failure to show a difference in the primary outcome, the rate of 30-day mortality was substantially lower in the therapeutic arm (4.9% vs. 7.0%), translating into a 30% risk reduction (HR, 0.70; P = .01).
Therapeutic anticoagulation was also associated with a lower rate of intubation/mechanical ventilation (6.4% vs. 8.4%) that reached statistical significance (HR, 0.75; P = .03). The risk reduction was also significant for a combination of these endpoints (HR, 0.77; P = .03).
The lower proportion of patients who eventually required ICU-level of care (9.9% vs. 11.7%) showed a trend in favor of therapeutic anticoagulation (HR, 0.84; P = .11).
Bleeding rates did not differ between arms
Bleeding Academic Research Consortium major bleeding types 3 and 5 were slightly numerically higher in the group randomized to therapeutic enoxaparin (0.5%) than prophylactic enoxaparin (0.1%) and therapeutic apixaban (0.3%), but the differences between any groups were not significant.
Numerous anticoagulation trials in patients with COVID-19 have been published previously. One 2021 trial published in the New England Journal of Medicine also suggested benefit from a therapeutic relative to prophylactic anticoagulation. In that trial, which compared heparin to usual-care thromboprophylaxis, benefits were derived from a Bayesian analysis. Significant differences were not shown for death or other major outcome assessed individually.
Even though this more recent trial missed its primary endpoint, Gregg Stone, MD, a coauthor of this study and a colleague of Dr. Fuster at the Mount Sinai School of Medicine, New York, reiterated that these results support routine anticoagulation in hospitalized COVID-19 patients.
“These are robust reductions in mortality and intubation rates, which are the most serious outcomes,” said Dr. Stone, who is first author of the paper, which was published in the Journal of the American College of Cardiology immediately after Dr. Fuster’s presentation.
COVID-19 has proven to be a very thrombogenic virus, but the literature has not been wholly consistent on which anticoagulation treatment provides the best balance of benefits and risks, according to Julia Grapsa, MD, PhD, attending cardiologist, Guys and St. Thomas Hospital, London. She said that this randomized trial, despite its failure to meet the primary endpoint, is useful.
“This demonstrates that a therapeutic dose of enoxaparin is likely to improve outcomes over a prophylactic dose with a low risk of bleeding,” Dr. Grapsa said. On the basis of the randomized study, “I feel more confident with this approach.”
Dr. Fuster reported no potential conflicts of interest. Dr. Stone has financial relationships with more than 30 companies that make pharmaceuticals and medical devices. Dr. Grapsa reported no potential conflicts of interest.
AT ACC 2023
‘Breakthrough’ study: Diabetes drug helps prevent long COVID
with The Lancet on SSRN. The preprint hasn’t yet been peer-reviewed or published in a journal.
In particular, metformin led to a 42% drop in long COVID among people who had a mild to moderate COVID-19 infection.
“Long COVID affects millions of people, and preventing long COVID through a treatment like metformin could prevent significant disruptions in people’s lives,” said lead author Carolyn Bramante, MD, assistant professor of internal medicine and pediatrics at the University of Minnesota, Minneapolis.
Between January 2021 and February 2022, Dr. Bramante and colleagues tested three oral medications – metformin (typically used to treat type 2 diabetes), ivermectin (an antiparasitic), and fluvoxamine (an antidepressant) – in a clinical trial across the United States called COVID-OUT. The people being studied, investigators, care providers, and others involved in the study were blinded to the randomized treatments. The trial was decentralized, with no in-person contact with participants.
The researchers included patients who were aged 30-85 with overweight or obesity, had documentation of a confirmed COVID-19 infection, had fewer than 7 days of symptoms, had no known prior infection, and joined the study within 3 days of their positive test. The study included monthly follow-up for 300 days, and participants indicated whether they received a long COVID diagnosis from a medical doctor, which the researchers confirmed in medical records after participants gave consent.
The medications were prepackaged into pill boxes for fast delivery to participants and to ensure they took the correct number of each type of pill. The packages were sent via same-day courier or overnight shipping.
The metformin doses were doled out over 14 days, with 500 milligrams on the first day, 500 milligrams twice a day for the next 4 days, and then 500 milligrams in the morning and 1,000 milligrams in the evening for the remaining 9 days.
Among the 1,323 people studied, 1,125 agreed to do long-term follow-up for long COVID: 564 in the metformin group and 561 in the blinded placebo group. The average age was 45, and 56% were women, including 7% who were pregnant.
The average time from the start of symptoms to starting medication was 5 days, and 47% began taking the drug within 4 days or less. About 55% had received the primary COVID-19 vaccination series, including 5.1% who received an initial booster, before enrolling in the study.
Overall, 8.4% of participants reported that a medical provider diagnosed them with long COVID. Of those who took metformin, 6.3% developed long COVID, compared to 10.6% among those who took the identical-matched placebo.
The risk reduction for metformin was 42% versus the placebo, which was consistent across subgroups, including vaccination status and different COVID-19 variants.
When metformin was started less than 4 days after COVID-19 symptoms started, the effect was potentially even greater, with a 64% reduction, as compared with a 36% reduction among those who started metformin after 4 or more days after symptoms.
Neither ivermectin nor fluvoxamine showed any benefits for preventing long COVID.
At the same time, the study authors caution that more research is needed.
“The COVID-OUT trial does not indicate whether or not metformin would be effective at preventing long COVID if started at the time of emergency department visit or hospitalization for COVID-19, nor whether metformin would be effective as treatment in persons who already have long COVID,” they wrote. “With the burden of long COVID on society, confirmation is urgently needed in a trial that addresses our study’s limitations in order to translate these results into practice and policy.”
Several risk factors for long COVID emerged in the analysis. About 11.1% of the women had a long COVID diagnosis, compared with 4.9% of the men. Also, those who had received at least the primary vaccine series had a lower risk of developing long COVID, at 6.6%, as compared with 10.5% among the unvaccinated. Only 1 of the 57 people who received a booster shot developed long COVID.
Notably, pregnant and lactating people were included in this study, which is important given that pregnant people face higher risks for poor COVID-19 outcomes and are excluded from most nonobstetric clinical trials, the study authors wrote. In this study, they were randomized to metformin or placebo but not ivermectin or fluvoxamine due to limited research about the safety of those drugs during pregnancy and lactation.
The results are now under journal review but show findings consistent with those from other recent studies. Also, in August 2022, the authors published results from COVID-OUT that showed metformin led to a 42% reduction in hospital visits, emergency department visits, and deaths related to severe COVID-19.
“Given the lack of side effects and cost for a 2-week course, I think these data support use of metformin now,” said Eric Topol, MD, founder and director of the Scripps Research Translational Institute and editor-in-chief of Medscape, WebMD’s sister site for health care professionals.
Dr. Topol, who wasn’t involved with this study, has been a leading voice on COVID-19 research throughout the pandemic. He noted the need for more studies, including a factorial design trial to test metformin and Paxlovid, which has shown promise in preventing long COVID. Dr. Topol also wrote about the preprint in Ground Truths, his online newsletter.
“As I’ve written in the past, I don’t use the term ‘breakthrough’ lightly,” he wrote. “But to see such a pronounced benefit in the current randomized trial of metformin, in the context of its being so safe and low cost, I’d give it a breakthrough categorization.”
Another way to put it, Dr. Topol wrote, is that based on this study, he would take metformin if he became infected with COVID-19.
Jeremy Faust, MD, an emergency medicine doctor at Brigham and Women’s Hospital in Boston, also wrote about the study in his newsletter, Inside Medicine. He noted that the 42% reduction in long COVID means that 23 COVID-19 patients need to be treated with metformin to prevent one long COVID diagnosis, which is an “important reduction.”
“Bottom line: If a person who meets criteria for obesity or overweight status were to ask me if they should take metformin (for 2 weeks) starting as soon as they learn they have COVID-19, I would say yes in many if not most cases, based on this new data,” he wrote. “This is starting to look like a real win.”
A version of this article first appeared on WebMD.com.
with The Lancet on SSRN. The preprint hasn’t yet been peer-reviewed or published in a journal.
In particular, metformin led to a 42% drop in long COVID among people who had a mild to moderate COVID-19 infection.
“Long COVID affects millions of people, and preventing long COVID through a treatment like metformin could prevent significant disruptions in people’s lives,” said lead author Carolyn Bramante, MD, assistant professor of internal medicine and pediatrics at the University of Minnesota, Minneapolis.
Between January 2021 and February 2022, Dr. Bramante and colleagues tested three oral medications – metformin (typically used to treat type 2 diabetes), ivermectin (an antiparasitic), and fluvoxamine (an antidepressant) – in a clinical trial across the United States called COVID-OUT. The people being studied, investigators, care providers, and others involved in the study were blinded to the randomized treatments. The trial was decentralized, with no in-person contact with participants.
The researchers included patients who were aged 30-85 with overweight or obesity, had documentation of a confirmed COVID-19 infection, had fewer than 7 days of symptoms, had no known prior infection, and joined the study within 3 days of their positive test. The study included monthly follow-up for 300 days, and participants indicated whether they received a long COVID diagnosis from a medical doctor, which the researchers confirmed in medical records after participants gave consent.
The medications were prepackaged into pill boxes for fast delivery to participants and to ensure they took the correct number of each type of pill. The packages were sent via same-day courier or overnight shipping.
The metformin doses were doled out over 14 days, with 500 milligrams on the first day, 500 milligrams twice a day for the next 4 days, and then 500 milligrams in the morning and 1,000 milligrams in the evening for the remaining 9 days.
Among the 1,323 people studied, 1,125 agreed to do long-term follow-up for long COVID: 564 in the metformin group and 561 in the blinded placebo group. The average age was 45, and 56% were women, including 7% who were pregnant.
The average time from the start of symptoms to starting medication was 5 days, and 47% began taking the drug within 4 days or less. About 55% had received the primary COVID-19 vaccination series, including 5.1% who received an initial booster, before enrolling in the study.
Overall, 8.4% of participants reported that a medical provider diagnosed them with long COVID. Of those who took metformin, 6.3% developed long COVID, compared to 10.6% among those who took the identical-matched placebo.
The risk reduction for metformin was 42% versus the placebo, which was consistent across subgroups, including vaccination status and different COVID-19 variants.
When metformin was started less than 4 days after COVID-19 symptoms started, the effect was potentially even greater, with a 64% reduction, as compared with a 36% reduction among those who started metformin after 4 or more days after symptoms.
Neither ivermectin nor fluvoxamine showed any benefits for preventing long COVID.
At the same time, the study authors caution that more research is needed.
“The COVID-OUT trial does not indicate whether or not metformin would be effective at preventing long COVID if started at the time of emergency department visit or hospitalization for COVID-19, nor whether metformin would be effective as treatment in persons who already have long COVID,” they wrote. “With the burden of long COVID on society, confirmation is urgently needed in a trial that addresses our study’s limitations in order to translate these results into practice and policy.”
Several risk factors for long COVID emerged in the analysis. About 11.1% of the women had a long COVID diagnosis, compared with 4.9% of the men. Also, those who had received at least the primary vaccine series had a lower risk of developing long COVID, at 6.6%, as compared with 10.5% among the unvaccinated. Only 1 of the 57 people who received a booster shot developed long COVID.
Notably, pregnant and lactating people were included in this study, which is important given that pregnant people face higher risks for poor COVID-19 outcomes and are excluded from most nonobstetric clinical trials, the study authors wrote. In this study, they were randomized to metformin or placebo but not ivermectin or fluvoxamine due to limited research about the safety of those drugs during pregnancy and lactation.
The results are now under journal review but show findings consistent with those from other recent studies. Also, in August 2022, the authors published results from COVID-OUT that showed metformin led to a 42% reduction in hospital visits, emergency department visits, and deaths related to severe COVID-19.
“Given the lack of side effects and cost for a 2-week course, I think these data support use of metformin now,” said Eric Topol, MD, founder and director of the Scripps Research Translational Institute and editor-in-chief of Medscape, WebMD’s sister site for health care professionals.
Dr. Topol, who wasn’t involved with this study, has been a leading voice on COVID-19 research throughout the pandemic. He noted the need for more studies, including a factorial design trial to test metformin and Paxlovid, which has shown promise in preventing long COVID. Dr. Topol also wrote about the preprint in Ground Truths, his online newsletter.
“As I’ve written in the past, I don’t use the term ‘breakthrough’ lightly,” he wrote. “But to see such a pronounced benefit in the current randomized trial of metformin, in the context of its being so safe and low cost, I’d give it a breakthrough categorization.”
Another way to put it, Dr. Topol wrote, is that based on this study, he would take metformin if he became infected with COVID-19.
Jeremy Faust, MD, an emergency medicine doctor at Brigham and Women’s Hospital in Boston, also wrote about the study in his newsletter, Inside Medicine. He noted that the 42% reduction in long COVID means that 23 COVID-19 patients need to be treated with metformin to prevent one long COVID diagnosis, which is an “important reduction.”
“Bottom line: If a person who meets criteria for obesity or overweight status were to ask me if they should take metformin (for 2 weeks) starting as soon as they learn they have COVID-19, I would say yes in many if not most cases, based on this new data,” he wrote. “This is starting to look like a real win.”
A version of this article first appeared on WebMD.com.
with The Lancet on SSRN. The preprint hasn’t yet been peer-reviewed or published in a journal.
In particular, metformin led to a 42% drop in long COVID among people who had a mild to moderate COVID-19 infection.
“Long COVID affects millions of people, and preventing long COVID through a treatment like metformin could prevent significant disruptions in people’s lives,” said lead author Carolyn Bramante, MD, assistant professor of internal medicine and pediatrics at the University of Minnesota, Minneapolis.
Between January 2021 and February 2022, Dr. Bramante and colleagues tested three oral medications – metformin (typically used to treat type 2 diabetes), ivermectin (an antiparasitic), and fluvoxamine (an antidepressant) – in a clinical trial across the United States called COVID-OUT. The people being studied, investigators, care providers, and others involved in the study were blinded to the randomized treatments. The trial was decentralized, with no in-person contact with participants.
The researchers included patients who were aged 30-85 with overweight or obesity, had documentation of a confirmed COVID-19 infection, had fewer than 7 days of symptoms, had no known prior infection, and joined the study within 3 days of their positive test. The study included monthly follow-up for 300 days, and participants indicated whether they received a long COVID diagnosis from a medical doctor, which the researchers confirmed in medical records after participants gave consent.
The medications were prepackaged into pill boxes for fast delivery to participants and to ensure they took the correct number of each type of pill. The packages were sent via same-day courier or overnight shipping.
The metformin doses were doled out over 14 days, with 500 milligrams on the first day, 500 milligrams twice a day for the next 4 days, and then 500 milligrams in the morning and 1,000 milligrams in the evening for the remaining 9 days.
Among the 1,323 people studied, 1,125 agreed to do long-term follow-up for long COVID: 564 in the metformin group and 561 in the blinded placebo group. The average age was 45, and 56% were women, including 7% who were pregnant.
The average time from the start of symptoms to starting medication was 5 days, and 47% began taking the drug within 4 days or less. About 55% had received the primary COVID-19 vaccination series, including 5.1% who received an initial booster, before enrolling in the study.
Overall, 8.4% of participants reported that a medical provider diagnosed them with long COVID. Of those who took metformin, 6.3% developed long COVID, compared to 10.6% among those who took the identical-matched placebo.
The risk reduction for metformin was 42% versus the placebo, which was consistent across subgroups, including vaccination status and different COVID-19 variants.
When metformin was started less than 4 days after COVID-19 symptoms started, the effect was potentially even greater, with a 64% reduction, as compared with a 36% reduction among those who started metformin after 4 or more days after symptoms.
Neither ivermectin nor fluvoxamine showed any benefits for preventing long COVID.
At the same time, the study authors caution that more research is needed.
“The COVID-OUT trial does not indicate whether or not metformin would be effective at preventing long COVID if started at the time of emergency department visit or hospitalization for COVID-19, nor whether metformin would be effective as treatment in persons who already have long COVID,” they wrote. “With the burden of long COVID on society, confirmation is urgently needed in a trial that addresses our study’s limitations in order to translate these results into practice and policy.”
Several risk factors for long COVID emerged in the analysis. About 11.1% of the women had a long COVID diagnosis, compared with 4.9% of the men. Also, those who had received at least the primary vaccine series had a lower risk of developing long COVID, at 6.6%, as compared with 10.5% among the unvaccinated. Only 1 of the 57 people who received a booster shot developed long COVID.
Notably, pregnant and lactating people were included in this study, which is important given that pregnant people face higher risks for poor COVID-19 outcomes and are excluded from most nonobstetric clinical trials, the study authors wrote. In this study, they were randomized to metformin or placebo but not ivermectin or fluvoxamine due to limited research about the safety of those drugs during pregnancy and lactation.
The results are now under journal review but show findings consistent with those from other recent studies. Also, in August 2022, the authors published results from COVID-OUT that showed metformin led to a 42% reduction in hospital visits, emergency department visits, and deaths related to severe COVID-19.
“Given the lack of side effects and cost for a 2-week course, I think these data support use of metformin now,” said Eric Topol, MD, founder and director of the Scripps Research Translational Institute and editor-in-chief of Medscape, WebMD’s sister site for health care professionals.
Dr. Topol, who wasn’t involved with this study, has been a leading voice on COVID-19 research throughout the pandemic. He noted the need for more studies, including a factorial design trial to test metformin and Paxlovid, which has shown promise in preventing long COVID. Dr. Topol also wrote about the preprint in Ground Truths, his online newsletter.
“As I’ve written in the past, I don’t use the term ‘breakthrough’ lightly,” he wrote. “But to see such a pronounced benefit in the current randomized trial of metformin, in the context of its being so safe and low cost, I’d give it a breakthrough categorization.”
Another way to put it, Dr. Topol wrote, is that based on this study, he would take metformin if he became infected with COVID-19.
Jeremy Faust, MD, an emergency medicine doctor at Brigham and Women’s Hospital in Boston, also wrote about the study in his newsletter, Inside Medicine. He noted that the 42% reduction in long COVID means that 23 COVID-19 patients need to be treated with metformin to prevent one long COVID diagnosis, which is an “important reduction.”
“Bottom line: If a person who meets criteria for obesity or overweight status were to ask me if they should take metformin (for 2 weeks) starting as soon as they learn they have COVID-19, I would say yes in many if not most cases, based on this new data,” he wrote. “This is starting to look like a real win.”
A version of this article first appeared on WebMD.com.
In utero exposure to asthma medication not tied to risks of neurodevelopmental disorders
The drugs included in the study were leukotriene-receptor antagonists (LTRAs), which are often used to treat allergic airway diseases, including asthma and allergic rhinitis.
“Over the years, the U.S. Food and Drug Administration has monitored post-marketing data about the potential harm of neuropsychiatric events (NEs) associated with montelukast, the first type of LTRAs, and issued boxed warnings about serious mental health side effects for montelukast in 2020,” said corresponding author Tsung-Chieh Yao, MD, of Chang Gung Memorial Hospital, Taiwan, in an interview.
However, evidence of a link between NEs and LTRA use has been inconsistent, according to Dr. Yao and colleagues.
“To date, it remains totally unknown whether the exposure to LTRAs during pregnancy is associated with the risk of neuropsychiatric events in offspring,” said Dr. Yao.
To address this question, the researchers used data from National Health Insurance Research Database in Taiwan to identify pregnant women and their offspring from 2009 to 2019. The initial study population included 576,157 mother-offspring pairs, including 1,995 LTRA-exposed and 574,162 nonexposed children.
The women had a diagnosis of asthma or allergic rhinitis; multiple births and children with congenital malformations were excluded. LTRA exposure was defined as any dispensed prescription for LTRAs during pregnancy. Approximately two-thirds of the mothers were aged 30-40 years at the time of delivery.
The findings were published in a research letter in JAMA Network Open.
In the study population at large, the incidence of the three neurodevelopmental disorders ADHD, autism spectrum disorder (ASD), and Tourette syndrome was not significantly different between those children exposed to LTRAs and those not exposed to LTRAs in utero (1.25% vs. 1.32%; 3.31% vs. 4.36%; and 0.45% vs. 0.83%, respectively).
After propensity score matching, the study population included 1,988 LTRA-exposed children and 19,863 nonexposed children. In this group, no significant associations appeared between prenatal LTRA exposure and the risk of attention-deficit/hyperactivity disorder (adjusted hazard ratio, 1.03), autism spectrum disorder (AHR, 1.01), and Tourette syndrome (AHR, 0.63).
Neither duration nor cumulative dose of LTRA use during pregnancy showed an association with ADHD, ASD, or Tourette syndrome in offspring. Duration of LTRA use was categorized as shorter or longer periods of 1-4 weeks vs. more than 4 weeks; cumulative dose was categorized as 1-170 mg vs. 170 mg or higher.
The findings were limited by the lack of randomization, inability to detect long-term risk, and potential lack of generalizability to non-Asian populations, and more research is needed to replicate the results, the researchers noted. However, the current findings were strengthened by the large study population, and suggest that LTRA use in pregnancy does not present a significant risk for NEs in children, which should be reassuring to clinicians and patients, they concluded.
The current study is the first to use the whole of Taiwan population data and extends previous studies by examining the association between LTRA use during pregnancy and risk of neuropsychiatric events in offspring, Dr. Yao said in an interview. “The possibly surprising, but reassuring, finding is that prenatal LTRA exposure did not increase risk of ADHD, ASD, and Tourette syndrome in offspring,” he said.
“Clinicians prescribing LTRAs such as montelukast (Singulair and generics) to pregnant women with asthma or allergic rhinitis may be reassured by our findings,” Dr. Yao added. The results offer real-world evidence to help inform decision-making about the use of LTRAs during pregnancy, although additional research is needed to replicate the study findings in other populations, he said.
The study was supported by the National Health Research Institutes, Taiwan, the Ministry of Science and Technology of Taiwan, the National Science and Technology Council of Taiwan, and the Chang Gung Medical Foundation. The researchers had no financial conflicts to disclose.
The drugs included in the study were leukotriene-receptor antagonists (LTRAs), which are often used to treat allergic airway diseases, including asthma and allergic rhinitis.
“Over the years, the U.S. Food and Drug Administration has monitored post-marketing data about the potential harm of neuropsychiatric events (NEs) associated with montelukast, the first type of LTRAs, and issued boxed warnings about serious mental health side effects for montelukast in 2020,” said corresponding author Tsung-Chieh Yao, MD, of Chang Gung Memorial Hospital, Taiwan, in an interview.
However, evidence of a link between NEs and LTRA use has been inconsistent, according to Dr. Yao and colleagues.
“To date, it remains totally unknown whether the exposure to LTRAs during pregnancy is associated with the risk of neuropsychiatric events in offspring,” said Dr. Yao.
To address this question, the researchers used data from National Health Insurance Research Database in Taiwan to identify pregnant women and their offspring from 2009 to 2019. The initial study population included 576,157 mother-offspring pairs, including 1,995 LTRA-exposed and 574,162 nonexposed children.
The women had a diagnosis of asthma or allergic rhinitis; multiple births and children with congenital malformations were excluded. LTRA exposure was defined as any dispensed prescription for LTRAs during pregnancy. Approximately two-thirds of the mothers were aged 30-40 years at the time of delivery.
The findings were published in a research letter in JAMA Network Open.
In the study population at large, the incidence of the three neurodevelopmental disorders ADHD, autism spectrum disorder (ASD), and Tourette syndrome was not significantly different between those children exposed to LTRAs and those not exposed to LTRAs in utero (1.25% vs. 1.32%; 3.31% vs. 4.36%; and 0.45% vs. 0.83%, respectively).
After propensity score matching, the study population included 1,988 LTRA-exposed children and 19,863 nonexposed children. In this group, no significant associations appeared between prenatal LTRA exposure and the risk of attention-deficit/hyperactivity disorder (adjusted hazard ratio, 1.03), autism spectrum disorder (AHR, 1.01), and Tourette syndrome (AHR, 0.63).
Neither duration nor cumulative dose of LTRA use during pregnancy showed an association with ADHD, ASD, or Tourette syndrome in offspring. Duration of LTRA use was categorized as shorter or longer periods of 1-4 weeks vs. more than 4 weeks; cumulative dose was categorized as 1-170 mg vs. 170 mg or higher.
The findings were limited by the lack of randomization, inability to detect long-term risk, and potential lack of generalizability to non-Asian populations, and more research is needed to replicate the results, the researchers noted. However, the current findings were strengthened by the large study population, and suggest that LTRA use in pregnancy does not present a significant risk for NEs in children, which should be reassuring to clinicians and patients, they concluded.
The current study is the first to use the whole of Taiwan population data and extends previous studies by examining the association between LTRA use during pregnancy and risk of neuropsychiatric events in offspring, Dr. Yao said in an interview. “The possibly surprising, but reassuring, finding is that prenatal LTRA exposure did not increase risk of ADHD, ASD, and Tourette syndrome in offspring,” he said.
“Clinicians prescribing LTRAs such as montelukast (Singulair and generics) to pregnant women with asthma or allergic rhinitis may be reassured by our findings,” Dr. Yao added. The results offer real-world evidence to help inform decision-making about the use of LTRAs during pregnancy, although additional research is needed to replicate the study findings in other populations, he said.
The study was supported by the National Health Research Institutes, Taiwan, the Ministry of Science and Technology of Taiwan, the National Science and Technology Council of Taiwan, and the Chang Gung Medical Foundation. The researchers had no financial conflicts to disclose.
The drugs included in the study were leukotriene-receptor antagonists (LTRAs), which are often used to treat allergic airway diseases, including asthma and allergic rhinitis.
“Over the years, the U.S. Food and Drug Administration has monitored post-marketing data about the potential harm of neuropsychiatric events (NEs) associated with montelukast, the first type of LTRAs, and issued boxed warnings about serious mental health side effects for montelukast in 2020,” said corresponding author Tsung-Chieh Yao, MD, of Chang Gung Memorial Hospital, Taiwan, in an interview.
However, evidence of a link between NEs and LTRA use has been inconsistent, according to Dr. Yao and colleagues.
“To date, it remains totally unknown whether the exposure to LTRAs during pregnancy is associated with the risk of neuropsychiatric events in offspring,” said Dr. Yao.
To address this question, the researchers used data from National Health Insurance Research Database in Taiwan to identify pregnant women and their offspring from 2009 to 2019. The initial study population included 576,157 mother-offspring pairs, including 1,995 LTRA-exposed and 574,162 nonexposed children.
The women had a diagnosis of asthma or allergic rhinitis; multiple births and children with congenital malformations were excluded. LTRA exposure was defined as any dispensed prescription for LTRAs during pregnancy. Approximately two-thirds of the mothers were aged 30-40 years at the time of delivery.
The findings were published in a research letter in JAMA Network Open.
In the study population at large, the incidence of the three neurodevelopmental disorders ADHD, autism spectrum disorder (ASD), and Tourette syndrome was not significantly different between those children exposed to LTRAs and those not exposed to LTRAs in utero (1.25% vs. 1.32%; 3.31% vs. 4.36%; and 0.45% vs. 0.83%, respectively).
After propensity score matching, the study population included 1,988 LTRA-exposed children and 19,863 nonexposed children. In this group, no significant associations appeared between prenatal LTRA exposure and the risk of attention-deficit/hyperactivity disorder (adjusted hazard ratio, 1.03), autism spectrum disorder (AHR, 1.01), and Tourette syndrome (AHR, 0.63).
Neither duration nor cumulative dose of LTRA use during pregnancy showed an association with ADHD, ASD, or Tourette syndrome in offspring. Duration of LTRA use was categorized as shorter or longer periods of 1-4 weeks vs. more than 4 weeks; cumulative dose was categorized as 1-170 mg vs. 170 mg or higher.
The findings were limited by the lack of randomization, inability to detect long-term risk, and potential lack of generalizability to non-Asian populations, and more research is needed to replicate the results, the researchers noted. However, the current findings were strengthened by the large study population, and suggest that LTRA use in pregnancy does not present a significant risk for NEs in children, which should be reassuring to clinicians and patients, they concluded.
The current study is the first to use the whole of Taiwan population data and extends previous studies by examining the association between LTRA use during pregnancy and risk of neuropsychiatric events in offspring, Dr. Yao said in an interview. “The possibly surprising, but reassuring, finding is that prenatal LTRA exposure did not increase risk of ADHD, ASD, and Tourette syndrome in offspring,” he said.
“Clinicians prescribing LTRAs such as montelukast (Singulair and generics) to pregnant women with asthma or allergic rhinitis may be reassured by our findings,” Dr. Yao added. The results offer real-world evidence to help inform decision-making about the use of LTRAs during pregnancy, although additional research is needed to replicate the study findings in other populations, he said.
The study was supported by the National Health Research Institutes, Taiwan, the Ministry of Science and Technology of Taiwan, the National Science and Technology Council of Taiwan, and the Chang Gung Medical Foundation. The researchers had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Fixed-dose combo pill for PAH promises accelerated benefit: A DUE
Already commonly used in combination for the treatment of pulmonary arterial hypertension (PAH), macitentan and tadalafil are safe and effective in a fixed-dose combination even as first-line therapy, according to a randomized multicenter comparative trial.
The fixed-dose combination “led to a highly significant and marked improvement in pulmonary vascular resistance when compared to macitentan and tadalafil as monotherapies,” Kelly Chin, MD, reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation.
Guidelines encourage rapid PVR reductions
In practice, it is common to start treatment with either the endothelial receptor antagonist (ERA) macitentan, the phosphodiesterase-5 (PDE-5) inhibitor tadalafil, or other frequently used medications for PAH, and to then add additional treatments, according to Dr. Chin. She pointed out, however, that guidelines, including those issued jointly by the European Society of Cardiology and the European Respiratory Society, encourage rapid escalation of therapy to quickly lower pulmonary vascular resistance (PVR).
In general, both macitentan and tadalafil are well tolerated, but the advantage and the safety of rapidly reducing PVR when these are initiated together in a single pill had not been evaluated previously in a major trial. In this double-blind phase III trial, called A DUE, 187 patients in functional class II or III PAH were randomized. The three-arm study included both treatment naive patients and patients who had been on stable doses (> 3 months) of an ERA or a PDE5 inhibitor, explained Dr. Chin, director of pulmonary hypertension at the UT Southwestern, Dallas.
Treatment naive patients, representing about 53% of the study population, were randomized to 10 mg macitentan monotherapy, 40 mg tadalafil monotherapy, or a fixed-dose, single-pill combination containing both. If on a stable dose of an ERA at trial entry, patients were randomized to 10 macitentan as a monotherapy or to the fixed dose combination. Patients entering the trial already on a stable dose of a PDE5 inhibitor were randomized to 40 mg tadalafil or the combination.
PVR reduced twofold on combination therapy
Relative to macitentan monotherapy, the percentage change from baseline in PVR by ratio of geometric mean, which was the primary outcome, was about twice as high on the combination (45% vs. 23%) at the end of the 16-week trial. This translates into a 29% PVR reduction (hazard ratio, 0.71; P < .0001).
For combination therapy relative to tadalafil monotherapy, the advantage for the fixed dose combination (44% vs. 22%) was about the same, also providing a nearly 30% relative reduction (HR, 0.72; P < .0001).
The increases in 6-minute walk distance (6MWD) at 16 weeks, a secondary endpoint, numerically favored the combination pill over both macitentan monotherapy (52.9 vs. 39.5 meters; P = .38) and tadalafil (43.4 vs. 15.9 meters; P = .059), but only the improvement relative to tadalafil monotherapy was considered a trend.
The proportion of patients who experienced at least one serious adverse event was higher in the combination arm (14.0%) relative to single agent macitentan (8.6%) or single agent tadalafil (9.1%). The adverse events and serious adverse events more common on the combination included hypotension, fluid retention, and anemia. This latter side effect occurred in 18.7%, 2.9%, and 2.3% in the combination, macitentan monotherapy, and tadalafil arms, respectively.
Several of those invited by the ACC to discuss the paper, including Lee R. Goldberg, MD, section chief of advanced heart failure and cardiac transplant, University of Pennsylvania, Philadelphia, raised concern about the increased rate of anemia among those in the combination pill. Two of the patients (2%) treated with the combination developed a hemoglobin < 8 g/dL.
Overall, nine (8.4%) of those on the fixed-dose combination, two (4.5%) of those randomized to tadalafil monotherapy, and none of the patients randomized to macitentan discontinued therapy due to side effects.
Anemia risk unexpected
Based on “the unexpected signal of an anemia risk,” Biykem Bozkurt, MD, PhD, chair of cardiology at Baylor College of Medicine, Houston, said that a larger scale trial with a longer follow-up is needed. While the concept of front-loading two drugs is attractive “for the very challenging PAH population,” she called for further evaluation of this safety signal before clinicians switch from the current practice of starting with one PAH therapy before adding others.
In addition, Dr. Bozkurt said a more definitive study would be helpful in determining whether starting with a fixed-pill combination is better than sequential treatment to improve quality of life. Dr. Bozkurt said it is likely that the lack of significant benefit on 6MWD in this study was due to the relatively small sample size, but an improvement in this measure would be another reason to consider a front-line fixed-dose combination.
Dr. Chin, in an interview, did not agree. She agreed that a larger sample size might have yielded a significant improvement in 6MWD, but she noted this outcome was moving in the right direction and was not the primary endpoint. In her opinion, this phase 3 trial does confirm that fixed-dose combination is well tolerated, has acceptable safety, and markedly improves PVR, fulfilling the guideline goal of controlling PAH more quickly.
Dr. Chin reports financial relationships with Altavant, Arena, Gossamer Bio, Janssen, Merck, ShouTi, and United Therapeutics. Dr. Goldberg reports financial relationships with Abbott, Respicardia/Zoll, and Viscardia. Dr. Bozkurt reports financial relationships with Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Cardurion, LivaNova, Relypsa, Renovacor, Sanofi-Aventis, and Vifor.
Already commonly used in combination for the treatment of pulmonary arterial hypertension (PAH), macitentan and tadalafil are safe and effective in a fixed-dose combination even as first-line therapy, according to a randomized multicenter comparative trial.
The fixed-dose combination “led to a highly significant and marked improvement in pulmonary vascular resistance when compared to macitentan and tadalafil as monotherapies,” Kelly Chin, MD, reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation.
Guidelines encourage rapid PVR reductions
In practice, it is common to start treatment with either the endothelial receptor antagonist (ERA) macitentan, the phosphodiesterase-5 (PDE-5) inhibitor tadalafil, or other frequently used medications for PAH, and to then add additional treatments, according to Dr. Chin. She pointed out, however, that guidelines, including those issued jointly by the European Society of Cardiology and the European Respiratory Society, encourage rapid escalation of therapy to quickly lower pulmonary vascular resistance (PVR).
In general, both macitentan and tadalafil are well tolerated, but the advantage and the safety of rapidly reducing PVR when these are initiated together in a single pill had not been evaluated previously in a major trial. In this double-blind phase III trial, called A DUE, 187 patients in functional class II or III PAH were randomized. The three-arm study included both treatment naive patients and patients who had been on stable doses (> 3 months) of an ERA or a PDE5 inhibitor, explained Dr. Chin, director of pulmonary hypertension at the UT Southwestern, Dallas.
Treatment naive patients, representing about 53% of the study population, were randomized to 10 mg macitentan monotherapy, 40 mg tadalafil monotherapy, or a fixed-dose, single-pill combination containing both. If on a stable dose of an ERA at trial entry, patients were randomized to 10 macitentan as a monotherapy or to the fixed dose combination. Patients entering the trial already on a stable dose of a PDE5 inhibitor were randomized to 40 mg tadalafil or the combination.
PVR reduced twofold on combination therapy
Relative to macitentan monotherapy, the percentage change from baseline in PVR by ratio of geometric mean, which was the primary outcome, was about twice as high on the combination (45% vs. 23%) at the end of the 16-week trial. This translates into a 29% PVR reduction (hazard ratio, 0.71; P < .0001).
For combination therapy relative to tadalafil monotherapy, the advantage for the fixed dose combination (44% vs. 22%) was about the same, also providing a nearly 30% relative reduction (HR, 0.72; P < .0001).
The increases in 6-minute walk distance (6MWD) at 16 weeks, a secondary endpoint, numerically favored the combination pill over both macitentan monotherapy (52.9 vs. 39.5 meters; P = .38) and tadalafil (43.4 vs. 15.9 meters; P = .059), but only the improvement relative to tadalafil monotherapy was considered a trend.
The proportion of patients who experienced at least one serious adverse event was higher in the combination arm (14.0%) relative to single agent macitentan (8.6%) or single agent tadalafil (9.1%). The adverse events and serious adverse events more common on the combination included hypotension, fluid retention, and anemia. This latter side effect occurred in 18.7%, 2.9%, and 2.3% in the combination, macitentan monotherapy, and tadalafil arms, respectively.
Several of those invited by the ACC to discuss the paper, including Lee R. Goldberg, MD, section chief of advanced heart failure and cardiac transplant, University of Pennsylvania, Philadelphia, raised concern about the increased rate of anemia among those in the combination pill. Two of the patients (2%) treated with the combination developed a hemoglobin < 8 g/dL.
Overall, nine (8.4%) of those on the fixed-dose combination, two (4.5%) of those randomized to tadalafil monotherapy, and none of the patients randomized to macitentan discontinued therapy due to side effects.
Anemia risk unexpected
Based on “the unexpected signal of an anemia risk,” Biykem Bozkurt, MD, PhD, chair of cardiology at Baylor College of Medicine, Houston, said that a larger scale trial with a longer follow-up is needed. While the concept of front-loading two drugs is attractive “for the very challenging PAH population,” she called for further evaluation of this safety signal before clinicians switch from the current practice of starting with one PAH therapy before adding others.
In addition, Dr. Bozkurt said a more definitive study would be helpful in determining whether starting with a fixed-pill combination is better than sequential treatment to improve quality of life. Dr. Bozkurt said it is likely that the lack of significant benefit on 6MWD in this study was due to the relatively small sample size, but an improvement in this measure would be another reason to consider a front-line fixed-dose combination.
Dr. Chin, in an interview, did not agree. She agreed that a larger sample size might have yielded a significant improvement in 6MWD, but she noted this outcome was moving in the right direction and was not the primary endpoint. In her opinion, this phase 3 trial does confirm that fixed-dose combination is well tolerated, has acceptable safety, and markedly improves PVR, fulfilling the guideline goal of controlling PAH more quickly.
Dr. Chin reports financial relationships with Altavant, Arena, Gossamer Bio, Janssen, Merck, ShouTi, and United Therapeutics. Dr. Goldberg reports financial relationships with Abbott, Respicardia/Zoll, and Viscardia. Dr. Bozkurt reports financial relationships with Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Cardurion, LivaNova, Relypsa, Renovacor, Sanofi-Aventis, and Vifor.
Already commonly used in combination for the treatment of pulmonary arterial hypertension (PAH), macitentan and tadalafil are safe and effective in a fixed-dose combination even as first-line therapy, according to a randomized multicenter comparative trial.
The fixed-dose combination “led to a highly significant and marked improvement in pulmonary vascular resistance when compared to macitentan and tadalafil as monotherapies,” Kelly Chin, MD, reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation.
Guidelines encourage rapid PVR reductions
In practice, it is common to start treatment with either the endothelial receptor antagonist (ERA) macitentan, the phosphodiesterase-5 (PDE-5) inhibitor tadalafil, or other frequently used medications for PAH, and to then add additional treatments, according to Dr. Chin. She pointed out, however, that guidelines, including those issued jointly by the European Society of Cardiology and the European Respiratory Society, encourage rapid escalation of therapy to quickly lower pulmonary vascular resistance (PVR).
In general, both macitentan and tadalafil are well tolerated, but the advantage and the safety of rapidly reducing PVR when these are initiated together in a single pill had not been evaluated previously in a major trial. In this double-blind phase III trial, called A DUE, 187 patients in functional class II or III PAH were randomized. The three-arm study included both treatment naive patients and patients who had been on stable doses (> 3 months) of an ERA or a PDE5 inhibitor, explained Dr. Chin, director of pulmonary hypertension at the UT Southwestern, Dallas.
Treatment naive patients, representing about 53% of the study population, were randomized to 10 mg macitentan monotherapy, 40 mg tadalafil monotherapy, or a fixed-dose, single-pill combination containing both. If on a stable dose of an ERA at trial entry, patients were randomized to 10 macitentan as a monotherapy or to the fixed dose combination. Patients entering the trial already on a stable dose of a PDE5 inhibitor were randomized to 40 mg tadalafil or the combination.
PVR reduced twofold on combination therapy
Relative to macitentan monotherapy, the percentage change from baseline in PVR by ratio of geometric mean, which was the primary outcome, was about twice as high on the combination (45% vs. 23%) at the end of the 16-week trial. This translates into a 29% PVR reduction (hazard ratio, 0.71; P < .0001).
For combination therapy relative to tadalafil monotherapy, the advantage for the fixed dose combination (44% vs. 22%) was about the same, also providing a nearly 30% relative reduction (HR, 0.72; P < .0001).
The increases in 6-minute walk distance (6MWD) at 16 weeks, a secondary endpoint, numerically favored the combination pill over both macitentan monotherapy (52.9 vs. 39.5 meters; P = .38) and tadalafil (43.4 vs. 15.9 meters; P = .059), but only the improvement relative to tadalafil monotherapy was considered a trend.
The proportion of patients who experienced at least one serious adverse event was higher in the combination arm (14.0%) relative to single agent macitentan (8.6%) or single agent tadalafil (9.1%). The adverse events and serious adverse events more common on the combination included hypotension, fluid retention, and anemia. This latter side effect occurred in 18.7%, 2.9%, and 2.3% in the combination, macitentan monotherapy, and tadalafil arms, respectively.
Several of those invited by the ACC to discuss the paper, including Lee R. Goldberg, MD, section chief of advanced heart failure and cardiac transplant, University of Pennsylvania, Philadelphia, raised concern about the increased rate of anemia among those in the combination pill. Two of the patients (2%) treated with the combination developed a hemoglobin < 8 g/dL.
Overall, nine (8.4%) of those on the fixed-dose combination, two (4.5%) of those randomized to tadalafil monotherapy, and none of the patients randomized to macitentan discontinued therapy due to side effects.
Anemia risk unexpected
Based on “the unexpected signal of an anemia risk,” Biykem Bozkurt, MD, PhD, chair of cardiology at Baylor College of Medicine, Houston, said that a larger scale trial with a longer follow-up is needed. While the concept of front-loading two drugs is attractive “for the very challenging PAH population,” she called for further evaluation of this safety signal before clinicians switch from the current practice of starting with one PAH therapy before adding others.
In addition, Dr. Bozkurt said a more definitive study would be helpful in determining whether starting with a fixed-pill combination is better than sequential treatment to improve quality of life. Dr. Bozkurt said it is likely that the lack of significant benefit on 6MWD in this study was due to the relatively small sample size, but an improvement in this measure would be another reason to consider a front-line fixed-dose combination.
Dr. Chin, in an interview, did not agree. She agreed that a larger sample size might have yielded a significant improvement in 6MWD, but she noted this outcome was moving in the right direction and was not the primary endpoint. In her opinion, this phase 3 trial does confirm that fixed-dose combination is well tolerated, has acceptable safety, and markedly improves PVR, fulfilling the guideline goal of controlling PAH more quickly.
Dr. Chin reports financial relationships with Altavant, Arena, Gossamer Bio, Janssen, Merck, ShouTi, and United Therapeutics. Dr. Goldberg reports financial relationships with Abbott, Respicardia/Zoll, and Viscardia. Dr. Bozkurt reports financial relationships with Abbott, Amgen, AstraZeneca, Boehringer Ingelheim, Cardurion, LivaNova, Relypsa, Renovacor, Sanofi-Aventis, and Vifor.
AT ACC 2023
Pulmonary function may predict frailty
Pulmonary function was significantly associated with frailty in community-dwelling older adults over a 5-year period, as indicated by data from more than 1,000 individuals.
The pulmonary function test has been proposed as a predictive tool for clinical outcomes in geriatrics, including hospitalization, mortality, and frailty, but , write Walter Sepulveda-Loyola, MD, of Universidad de Las Americas, Santiago, Chile, and colleagues.
In an observational study published in Heart and Lung, the researchers reviewed data from adults older than 64 years who were participants in the Toledo Study for Healthy Aging.
The study population included 1,188 older adults (mean age, 74 years; 54% women). The prevalence of frailty at baseline ranged from 7% to 26%.
Frailty was defined using the frailty phenotype (FP) and the Frailty Trait Scale 5 (FTS5). Pulmonary function was determined on the basis of forced expiratory volume in the first second (FEV1) and forced vital capacity (FVC), using spirometry.
Overall, at the 5-year follow-up, FEV1 and FVC were inversely associated with prevalence and incidence of frailty in nonadjusted and adjusted models using FP and FTS5.
In adjusted models, FEV1 and FVC, as well as FEV1 and FVC percent predicted value, were significantly associated with the prevalence of frailty, with odds ratios ranging from 0.53 to 0.99. FEV1 and FVC were significantly associated with increased incidence of frailty, with odds ratios ranging from 0.49 to 0.50 (P < .05 for both).
Pulmonary function also was associated with prevalent and incident frailty, hospitalization, and mortality in regression models, including the whole sample and after respiratory diseases were excluded.
Pulmonary function measures below the cutoff points for FEV1 and FVC were significantly associated with frailty, as well as with hospitalization and mortality. The cutoff points for FEV1 were 1.805 L for men and 1.165 L for women; cutoff points for FVC were 2.385 L for men and 1.585 L for women.
“Pulmonary function should be evaluated not only in frail patients, with the aim of detecting patients with poor prognoses regardless of their comorbidity, but also in individuals who are not frail but have an increased risk of developing frailty, as well as other adverse events,” the researchers write.
The study findings were limited by lack of data on pulmonary function variables outside of spirometry and by the need for data from populations with different characteristics to assess whether the same cutoff points are predictive of frailty, the researchers note.
The results were strengthened by the large sample size and additional analysis that excluded other respiratory diseases. Future research should consider adding pulmonary function assessment to the frailty model, the authors write.
Given the relationship between pulmonary function and physical capacity, the current study supports more frequent evaluation of pulmonary function in clinical practice for older adults, including those with no pulmonary disease, they conclude.
The study was supported by the Spanish Ministry of Economy, Industry, and Competitiveness, financed by the European Regional Development Funds, and the Centro de Investigacion Biomedica en Red en Fragilidad y Envejecimiento Saludable and the Fundacion Francisco Soria Melguizo. Lead author Dr. Sepulveda-Loyola was supported by the Brazilian National Council for Scientific and Technological Development.
A version of this article first appeared on Medscape.com.
Pulmonary function was significantly associated with frailty in community-dwelling older adults over a 5-year period, as indicated by data from more than 1,000 individuals.
The pulmonary function test has been proposed as a predictive tool for clinical outcomes in geriatrics, including hospitalization, mortality, and frailty, but , write Walter Sepulveda-Loyola, MD, of Universidad de Las Americas, Santiago, Chile, and colleagues.
In an observational study published in Heart and Lung, the researchers reviewed data from adults older than 64 years who were participants in the Toledo Study for Healthy Aging.
The study population included 1,188 older adults (mean age, 74 years; 54% women). The prevalence of frailty at baseline ranged from 7% to 26%.
Frailty was defined using the frailty phenotype (FP) and the Frailty Trait Scale 5 (FTS5). Pulmonary function was determined on the basis of forced expiratory volume in the first second (FEV1) and forced vital capacity (FVC), using spirometry.
Overall, at the 5-year follow-up, FEV1 and FVC were inversely associated with prevalence and incidence of frailty in nonadjusted and adjusted models using FP and FTS5.
In adjusted models, FEV1 and FVC, as well as FEV1 and FVC percent predicted value, were significantly associated with the prevalence of frailty, with odds ratios ranging from 0.53 to 0.99. FEV1 and FVC were significantly associated with increased incidence of frailty, with odds ratios ranging from 0.49 to 0.50 (P < .05 for both).
Pulmonary function also was associated with prevalent and incident frailty, hospitalization, and mortality in regression models, including the whole sample and after respiratory diseases were excluded.
Pulmonary function measures below the cutoff points for FEV1 and FVC were significantly associated with frailty, as well as with hospitalization and mortality. The cutoff points for FEV1 were 1.805 L for men and 1.165 L for women; cutoff points for FVC were 2.385 L for men and 1.585 L for women.
“Pulmonary function should be evaluated not only in frail patients, with the aim of detecting patients with poor prognoses regardless of their comorbidity, but also in individuals who are not frail but have an increased risk of developing frailty, as well as other adverse events,” the researchers write.
The study findings were limited by lack of data on pulmonary function variables outside of spirometry and by the need for data from populations with different characteristics to assess whether the same cutoff points are predictive of frailty, the researchers note.
The results were strengthened by the large sample size and additional analysis that excluded other respiratory diseases. Future research should consider adding pulmonary function assessment to the frailty model, the authors write.
Given the relationship between pulmonary function and physical capacity, the current study supports more frequent evaluation of pulmonary function in clinical practice for older adults, including those with no pulmonary disease, they conclude.
The study was supported by the Spanish Ministry of Economy, Industry, and Competitiveness, financed by the European Regional Development Funds, and the Centro de Investigacion Biomedica en Red en Fragilidad y Envejecimiento Saludable and the Fundacion Francisco Soria Melguizo. Lead author Dr. Sepulveda-Loyola was supported by the Brazilian National Council for Scientific and Technological Development.
A version of this article first appeared on Medscape.com.
Pulmonary function was significantly associated with frailty in community-dwelling older adults over a 5-year period, as indicated by data from more than 1,000 individuals.
The pulmonary function test has been proposed as a predictive tool for clinical outcomes in geriatrics, including hospitalization, mortality, and frailty, but , write Walter Sepulveda-Loyola, MD, of Universidad de Las Americas, Santiago, Chile, and colleagues.
In an observational study published in Heart and Lung, the researchers reviewed data from adults older than 64 years who were participants in the Toledo Study for Healthy Aging.
The study population included 1,188 older adults (mean age, 74 years; 54% women). The prevalence of frailty at baseline ranged from 7% to 26%.
Frailty was defined using the frailty phenotype (FP) and the Frailty Trait Scale 5 (FTS5). Pulmonary function was determined on the basis of forced expiratory volume in the first second (FEV1) and forced vital capacity (FVC), using spirometry.
Overall, at the 5-year follow-up, FEV1 and FVC were inversely associated with prevalence and incidence of frailty in nonadjusted and adjusted models using FP and FTS5.
In adjusted models, FEV1 and FVC, as well as FEV1 and FVC percent predicted value, were significantly associated with the prevalence of frailty, with odds ratios ranging from 0.53 to 0.99. FEV1 and FVC were significantly associated with increased incidence of frailty, with odds ratios ranging from 0.49 to 0.50 (P < .05 for both).
Pulmonary function also was associated with prevalent and incident frailty, hospitalization, and mortality in regression models, including the whole sample and after respiratory diseases were excluded.
Pulmonary function measures below the cutoff points for FEV1 and FVC were significantly associated with frailty, as well as with hospitalization and mortality. The cutoff points for FEV1 were 1.805 L for men and 1.165 L for women; cutoff points for FVC were 2.385 L for men and 1.585 L for women.
“Pulmonary function should be evaluated not only in frail patients, with the aim of detecting patients with poor prognoses regardless of their comorbidity, but also in individuals who are not frail but have an increased risk of developing frailty, as well as other adverse events,” the researchers write.
The study findings were limited by lack of data on pulmonary function variables outside of spirometry and by the need for data from populations with different characteristics to assess whether the same cutoff points are predictive of frailty, the researchers note.
The results were strengthened by the large sample size and additional analysis that excluded other respiratory diseases. Future research should consider adding pulmonary function assessment to the frailty model, the authors write.
Given the relationship between pulmonary function and physical capacity, the current study supports more frequent evaluation of pulmonary function in clinical practice for older adults, including those with no pulmonary disease, they conclude.
The study was supported by the Spanish Ministry of Economy, Industry, and Competitiveness, financed by the European Regional Development Funds, and the Centro de Investigacion Biomedica en Red en Fragilidad y Envejecimiento Saludable and the Fundacion Francisco Soria Melguizo. Lead author Dr. Sepulveda-Loyola was supported by the Brazilian National Council for Scientific and Technological Development.
A version of this article first appeared on Medscape.com.
Even mild COVID is hard on the brain
early research suggests.
“Our results suggest a severe pattern of changes in how the brain communicates as well as its structure, mainly in people with anxiety and depression with long-COVID syndrome, which affects so many people,” study investigator Clarissa Yasuda, MD, PhD, from University of Campinas, São Paulo, said in a news release.
“The magnitude of these changes suggests that they could lead to problems with memory and thinking skills, so we need to be exploring holistic treatments even for people mildly affected by COVID-19,” Dr. Yasuda added.
The findings were released March 6 ahead of the study’s scheduled presentation at the annual meeting of the American Academy of Neurology.
Brain shrinkage
Some studies have shown a high prevalence of symptoms of anxiety and depression in COVID-19 survivors, but few have investigated the associated cerebral changes, Dr. Yasuda told this news organization.
The study included 254 adults (177 women, 77 men, median age 41 years) who had mild COVID-19 a median of 82 days earlier. A total of 102 had symptoms of both anxiety and depression, and 152 had no such symptoms.
On brain imaging, those with COVID-19 and anxiety and depression had atrophy in the limbic area of the brain, which plays a role in memory and emotional processing.
No shrinkage in this area was evident in people who had COVID-19 without anxiety and depression or in a healthy control group of individuals without COVID-19.
The researchers also observed a “severe” pattern of abnormal cerebral functional connectivity in those with COVID-19 and anxiety and depression.
In this functional connectivity analysis, individuals with COVID-19 and anxiety and depression had widespread functional changes in each of the 12 networks assessed, while those with COVID-19 but without symptoms of anxiety and depression showed changes in only 5 networks.
Mechanisms unclear
“Unfortunately, the underpinning mechanisms associated with brain changes and neuropsychiatric dysfunction after COVID-19 infection are unclear,” Dr. Yasuda told this news organization.
“Some studies have demonstrated an association between symptoms of anxiety and depression with inflammation. However, we hypothesize that these cerebral alterations may result from a more complex interaction of social, psychological, and systemic stressors, including inflammation. It is indeed intriguing that such alterations are present in individuals who presented mild acute infection,” Dr. Yasuda added.
“Symptoms of anxiety and depression are frequently observed after COVID-19 and are part of long-COVID syndrome for some individuals. These symptoms require adequate treatment to improve the quality of life, cognition, and work capacity,” she said.
Treating these symptoms may induce “brain plasticity, which may result in some degree of gray matter increase and eventually prevent further structural and functional damage,” Dr. Yasuda said.
A limitation of the study was that symptoms of anxiety and depression were self-reported, meaning people may have misjudged or misreported symptoms.
Commenting on the findings for this news organization, Cyrus Raji, MD, PhD, with the Mallinckrodt Institute of Radiology, Washington University, St. Louis, said the idea that COVID-19 is bad for the brain isn’t new. Dr. Raji was not involved with the study.
Early in the pandemic, Dr. Raji and colleagues published a paper detailing COVID-19’s effects on the brain, and Dr. Raji followed it up with a TED talk on the subject.
“Within the growing framework of what we already know about COVID-19 infection and its adverse effects on the brain, this work incrementally adds to this knowledge by identifying functional and structural neuroimaging abnormalities related to anxiety and depression in persons suffering from COVID-19 infection,” Dr. Raji said.
The study was supported by the São Paulo Research Foundation. The authors have no relevant disclosures. Raji is a consultant for Brainreader, Apollo Health, Pacific Neuroscience Foundation, and Neurevolution LLC.
early research suggests.
“Our results suggest a severe pattern of changes in how the brain communicates as well as its structure, mainly in people with anxiety and depression with long-COVID syndrome, which affects so many people,” study investigator Clarissa Yasuda, MD, PhD, from University of Campinas, São Paulo, said in a news release.
“The magnitude of these changes suggests that they could lead to problems with memory and thinking skills, so we need to be exploring holistic treatments even for people mildly affected by COVID-19,” Dr. Yasuda added.
The findings were released March 6 ahead of the study’s scheduled presentation at the annual meeting of the American Academy of Neurology.
Brain shrinkage
Some studies have shown a high prevalence of symptoms of anxiety and depression in COVID-19 survivors, but few have investigated the associated cerebral changes, Dr. Yasuda told this news organization.
The study included 254 adults (177 women, 77 men, median age 41 years) who had mild COVID-19 a median of 82 days earlier. A total of 102 had symptoms of both anxiety and depression, and 152 had no such symptoms.
On brain imaging, those with COVID-19 and anxiety and depression had atrophy in the limbic area of the brain, which plays a role in memory and emotional processing.
No shrinkage in this area was evident in people who had COVID-19 without anxiety and depression or in a healthy control group of individuals without COVID-19.
The researchers also observed a “severe” pattern of abnormal cerebral functional connectivity in those with COVID-19 and anxiety and depression.
In this functional connectivity analysis, individuals with COVID-19 and anxiety and depression had widespread functional changes in each of the 12 networks assessed, while those with COVID-19 but without symptoms of anxiety and depression showed changes in only 5 networks.
Mechanisms unclear
“Unfortunately, the underpinning mechanisms associated with brain changes and neuropsychiatric dysfunction after COVID-19 infection are unclear,” Dr. Yasuda told this news organization.
“Some studies have demonstrated an association between symptoms of anxiety and depression with inflammation. However, we hypothesize that these cerebral alterations may result from a more complex interaction of social, psychological, and systemic stressors, including inflammation. It is indeed intriguing that such alterations are present in individuals who presented mild acute infection,” Dr. Yasuda added.
“Symptoms of anxiety and depression are frequently observed after COVID-19 and are part of long-COVID syndrome for some individuals. These symptoms require adequate treatment to improve the quality of life, cognition, and work capacity,” she said.
Treating these symptoms may induce “brain plasticity, which may result in some degree of gray matter increase and eventually prevent further structural and functional damage,” Dr. Yasuda said.
A limitation of the study was that symptoms of anxiety and depression were self-reported, meaning people may have misjudged or misreported symptoms.
Commenting on the findings for this news organization, Cyrus Raji, MD, PhD, with the Mallinckrodt Institute of Radiology, Washington University, St. Louis, said the idea that COVID-19 is bad for the brain isn’t new. Dr. Raji was not involved with the study.
Early in the pandemic, Dr. Raji and colleagues published a paper detailing COVID-19’s effects on the brain, and Dr. Raji followed it up with a TED talk on the subject.
“Within the growing framework of what we already know about COVID-19 infection and its adverse effects on the brain, this work incrementally adds to this knowledge by identifying functional and structural neuroimaging abnormalities related to anxiety and depression in persons suffering from COVID-19 infection,” Dr. Raji said.
The study was supported by the São Paulo Research Foundation. The authors have no relevant disclosures. Raji is a consultant for Brainreader, Apollo Health, Pacific Neuroscience Foundation, and Neurevolution LLC.
early research suggests.
“Our results suggest a severe pattern of changes in how the brain communicates as well as its structure, mainly in people with anxiety and depression with long-COVID syndrome, which affects so many people,” study investigator Clarissa Yasuda, MD, PhD, from University of Campinas, São Paulo, said in a news release.
“The magnitude of these changes suggests that they could lead to problems with memory and thinking skills, so we need to be exploring holistic treatments even for people mildly affected by COVID-19,” Dr. Yasuda added.
The findings were released March 6 ahead of the study’s scheduled presentation at the annual meeting of the American Academy of Neurology.
Brain shrinkage
Some studies have shown a high prevalence of symptoms of anxiety and depression in COVID-19 survivors, but few have investigated the associated cerebral changes, Dr. Yasuda told this news organization.
The study included 254 adults (177 women, 77 men, median age 41 years) who had mild COVID-19 a median of 82 days earlier. A total of 102 had symptoms of both anxiety and depression, and 152 had no such symptoms.
On brain imaging, those with COVID-19 and anxiety and depression had atrophy in the limbic area of the brain, which plays a role in memory and emotional processing.
No shrinkage in this area was evident in people who had COVID-19 without anxiety and depression or in a healthy control group of individuals without COVID-19.
The researchers also observed a “severe” pattern of abnormal cerebral functional connectivity in those with COVID-19 and anxiety and depression.
In this functional connectivity analysis, individuals with COVID-19 and anxiety and depression had widespread functional changes in each of the 12 networks assessed, while those with COVID-19 but without symptoms of anxiety and depression showed changes in only 5 networks.
Mechanisms unclear
“Unfortunately, the underpinning mechanisms associated with brain changes and neuropsychiatric dysfunction after COVID-19 infection are unclear,” Dr. Yasuda told this news organization.
“Some studies have demonstrated an association between symptoms of anxiety and depression with inflammation. However, we hypothesize that these cerebral alterations may result from a more complex interaction of social, psychological, and systemic stressors, including inflammation. It is indeed intriguing that such alterations are present in individuals who presented mild acute infection,” Dr. Yasuda added.
“Symptoms of anxiety and depression are frequently observed after COVID-19 and are part of long-COVID syndrome for some individuals. These symptoms require adequate treatment to improve the quality of life, cognition, and work capacity,” she said.
Treating these symptoms may induce “brain plasticity, which may result in some degree of gray matter increase and eventually prevent further structural and functional damage,” Dr. Yasuda said.
A limitation of the study was that symptoms of anxiety and depression were self-reported, meaning people may have misjudged or misreported symptoms.
Commenting on the findings for this news organization, Cyrus Raji, MD, PhD, with the Mallinckrodt Institute of Radiology, Washington University, St. Louis, said the idea that COVID-19 is bad for the brain isn’t new. Dr. Raji was not involved with the study.
Early in the pandemic, Dr. Raji and colleagues published a paper detailing COVID-19’s effects on the brain, and Dr. Raji followed it up with a TED talk on the subject.
“Within the growing framework of what we already know about COVID-19 infection and its adverse effects on the brain, this work incrementally adds to this knowledge by identifying functional and structural neuroimaging abnormalities related to anxiety and depression in persons suffering from COVID-19 infection,” Dr. Raji said.
The study was supported by the São Paulo Research Foundation. The authors have no relevant disclosures. Raji is a consultant for Brainreader, Apollo Health, Pacific Neuroscience Foundation, and Neurevolution LLC.
OTC budesonide-formoterol for asthma could save lives, money
, according to a computer modeling study presented at the American Academy of Allergy, Asthma, and Immunology 2023 annual meeting in San Antonio.
Asthma affects 25 million people, about 1 in 13, in the United States. About 28% are uninsured or underinsured, and 70% have mild asthma. Many are using a $30 inhaled epinephrine product (Primatene Mist) – the only FDA-approved asthma inhaler available without a prescription, said Marcus Shaker, MD, MS, professor of pediatrics and medicine at Geisel School of Medicine at Dartmouth, and clinician at Dartmouth Health Children’s, N.H.
A new version of Primatene Mist was reintroduced on the market in 2018 after the product was pulled for containing chlorofluorocarbons in 2011, but it is not recommended by professional medical societies because of safety concerns over epinephrine’s adverse effects, such as increased heart rate and blood pressure.
Drugs in its class (bronchodilators) have long been associated with a higher risk for death or near-death.
Meanwhile, research more than 2 decades ago linked regular use of low-dose inhaled corticosteroids with reduced risk for asthma death.
More recently, two large studies (SYGMA 1 and SYGMA 2) compared maintenance therapy with a low-dose inhaled corticosteroid (budesonide) vs. on-demand treatment with an inhaler containing both a corticosteroid (budesonide) and a long-acting bronchodilator (formoterol).
“Using as-needed budesonide-formoterol led to outcomes that are almost as good as taking a maintenance budesonide dose every day,” said Dr. Shaker.
The Global Initiative for Asthma guidelines now recommend this approach – as-needed inhaled corticosteroids (ICS) plus long-acting bronchodilators – for adults with mild asthma. In the United States, however, the National Heart, Lung, and Blood Institute still suggests daily ICS plus quick-relief therapy as needed.
Dr. Shaker and colleagues used computer modeling to compare the cost-effectiveness of as-needed budesonide-formoterol vs. over-the-counter inhaled epinephrine in underinsured U.S. adults who were self-managing their mild asthma. The study randomly assigned these individuals into three groups: OTC inhaled epinephrine (current reality), OTC budesonide-formoterol (not yet available), or no OTC option. The model assumed that patients treated for an exacerbation were referred to a health care provider and started a regimen of ICS plus as-needed rescue therapy.
In this analysis, which has been submitted for publication, the OTC budesonide-formoterol strategy was associated with 12,495 fewer deaths, prevented nearly 14 million severe asthma exacerbations, and saved more than $68 billion. And “when we looked at OTC budesonide-formoterol vs. having no OTC option at all, budesonide-formoterol was similarly cost-effective,” said Dr. Shaker, who presented the results at an AAAAI oral abstract session.
The cost savings emerged even though in the United States asthma controller therapies (for example, fluticasone) cost about 10 times more than rescue therapies (for instance, salbutamol, OTC epinephrine).
Nevertheless, the results make sense. “If you’re using Primatene Mist, your health costs are predicted to be much greater because you’re going to be in the hospital more. Your asthma is not going to be well-controlled,” Thanai Pongdee, MD, an allergist-immunologist with the Mayo Clinic in Rochester, Minn., told this news organization. “It’s not only the cost of your ER visit but also the cost of loss of work or school, and loss of daily productivity. There are all these associated costs.”
The analysis “is certainly something policy makers could take a look at,” he said.
He noted that current use of budesonide-formoterol is stymied by difficulties with insurance coverage. The difficulties stem from a mismatch between the updated recommendation for as-needed use and the description printed on the brand-name product (Symbicort).
“On the product label, it says Symbicort should be used on a daily basis,” Dr. Pongdee said. “But if a prescription comes through and says you’re going to use this ‘as needed,’ the health plan may say that’s not appropriate because that’s not on the product label.”
Given these access challenges with the all-in-one inhaler, other researchers have developed a workaround – asking patients to continue their usual care (that is, using a rescue inhaler as needed) but to also administer a controller medication after each rescue. When tested in Black and Latino patients with moderate to severe asthma, this easy strategy (patient activated reliever-triggered inhaled corticosteroid, or PARTICS) reduced severe asthma exacerbations about as well as the all-in-one inhaler.
If the all-in-one budesonide-formoterol does become available OTC, Dr. Shaker stressed that it “would not be a substitute for seeing an allergist and getting appropriate medical care and an evaluation and all the rest. But it’s better than the status quo. It’s the sort of thing where the perfect is not the enemy of the good,” he said.
Dr. Shaker is the AAAAI cochair of the Joint Task Force on Practice Parameters and serves as an editorial board member of the Journal of Allergy and Clinical Immunology in Practice. He is also an associate editor of the Annals of Allergy, Asthma, and Immunology. Dr. Pongdee serves as an at-large director on the AAAAI board of directors. He receives grant funding from GlaxoSmithKline, and Mayo Clinic is a trial site for GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
, according to a computer modeling study presented at the American Academy of Allergy, Asthma, and Immunology 2023 annual meeting in San Antonio.
Asthma affects 25 million people, about 1 in 13, in the United States. About 28% are uninsured or underinsured, and 70% have mild asthma. Many are using a $30 inhaled epinephrine product (Primatene Mist) – the only FDA-approved asthma inhaler available without a prescription, said Marcus Shaker, MD, MS, professor of pediatrics and medicine at Geisel School of Medicine at Dartmouth, and clinician at Dartmouth Health Children’s, N.H.
A new version of Primatene Mist was reintroduced on the market in 2018 after the product was pulled for containing chlorofluorocarbons in 2011, but it is not recommended by professional medical societies because of safety concerns over epinephrine’s adverse effects, such as increased heart rate and blood pressure.
Drugs in its class (bronchodilators) have long been associated with a higher risk for death or near-death.
Meanwhile, research more than 2 decades ago linked regular use of low-dose inhaled corticosteroids with reduced risk for asthma death.
More recently, two large studies (SYGMA 1 and SYGMA 2) compared maintenance therapy with a low-dose inhaled corticosteroid (budesonide) vs. on-demand treatment with an inhaler containing both a corticosteroid (budesonide) and a long-acting bronchodilator (formoterol).
“Using as-needed budesonide-formoterol led to outcomes that are almost as good as taking a maintenance budesonide dose every day,” said Dr. Shaker.
The Global Initiative for Asthma guidelines now recommend this approach – as-needed inhaled corticosteroids (ICS) plus long-acting bronchodilators – for adults with mild asthma. In the United States, however, the National Heart, Lung, and Blood Institute still suggests daily ICS plus quick-relief therapy as needed.
Dr. Shaker and colleagues used computer modeling to compare the cost-effectiveness of as-needed budesonide-formoterol vs. over-the-counter inhaled epinephrine in underinsured U.S. adults who were self-managing their mild asthma. The study randomly assigned these individuals into three groups: OTC inhaled epinephrine (current reality), OTC budesonide-formoterol (not yet available), or no OTC option. The model assumed that patients treated for an exacerbation were referred to a health care provider and started a regimen of ICS plus as-needed rescue therapy.
In this analysis, which has been submitted for publication, the OTC budesonide-formoterol strategy was associated with 12,495 fewer deaths, prevented nearly 14 million severe asthma exacerbations, and saved more than $68 billion. And “when we looked at OTC budesonide-formoterol vs. having no OTC option at all, budesonide-formoterol was similarly cost-effective,” said Dr. Shaker, who presented the results at an AAAAI oral abstract session.
The cost savings emerged even though in the United States asthma controller therapies (for example, fluticasone) cost about 10 times more than rescue therapies (for instance, salbutamol, OTC epinephrine).
Nevertheless, the results make sense. “If you’re using Primatene Mist, your health costs are predicted to be much greater because you’re going to be in the hospital more. Your asthma is not going to be well-controlled,” Thanai Pongdee, MD, an allergist-immunologist with the Mayo Clinic in Rochester, Minn., told this news organization. “It’s not only the cost of your ER visit but also the cost of loss of work or school, and loss of daily productivity. There are all these associated costs.”
The analysis “is certainly something policy makers could take a look at,” he said.
He noted that current use of budesonide-formoterol is stymied by difficulties with insurance coverage. The difficulties stem from a mismatch between the updated recommendation for as-needed use and the description printed on the brand-name product (Symbicort).
“On the product label, it says Symbicort should be used on a daily basis,” Dr. Pongdee said. “But if a prescription comes through and says you’re going to use this ‘as needed,’ the health plan may say that’s not appropriate because that’s not on the product label.”
Given these access challenges with the all-in-one inhaler, other researchers have developed a workaround – asking patients to continue their usual care (that is, using a rescue inhaler as needed) but to also administer a controller medication after each rescue. When tested in Black and Latino patients with moderate to severe asthma, this easy strategy (patient activated reliever-triggered inhaled corticosteroid, or PARTICS) reduced severe asthma exacerbations about as well as the all-in-one inhaler.
If the all-in-one budesonide-formoterol does become available OTC, Dr. Shaker stressed that it “would not be a substitute for seeing an allergist and getting appropriate medical care and an evaluation and all the rest. But it’s better than the status quo. It’s the sort of thing where the perfect is not the enemy of the good,” he said.
Dr. Shaker is the AAAAI cochair of the Joint Task Force on Practice Parameters and serves as an editorial board member of the Journal of Allergy and Clinical Immunology in Practice. He is also an associate editor of the Annals of Allergy, Asthma, and Immunology. Dr. Pongdee serves as an at-large director on the AAAAI board of directors. He receives grant funding from GlaxoSmithKline, and Mayo Clinic is a trial site for GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
, according to a computer modeling study presented at the American Academy of Allergy, Asthma, and Immunology 2023 annual meeting in San Antonio.
Asthma affects 25 million people, about 1 in 13, in the United States. About 28% are uninsured or underinsured, and 70% have mild asthma. Many are using a $30 inhaled epinephrine product (Primatene Mist) – the only FDA-approved asthma inhaler available without a prescription, said Marcus Shaker, MD, MS, professor of pediatrics and medicine at Geisel School of Medicine at Dartmouth, and clinician at Dartmouth Health Children’s, N.H.
A new version of Primatene Mist was reintroduced on the market in 2018 after the product was pulled for containing chlorofluorocarbons in 2011, but it is not recommended by professional medical societies because of safety concerns over epinephrine’s adverse effects, such as increased heart rate and blood pressure.
Drugs in its class (bronchodilators) have long been associated with a higher risk for death or near-death.
Meanwhile, research more than 2 decades ago linked regular use of low-dose inhaled corticosteroids with reduced risk for asthma death.
More recently, two large studies (SYGMA 1 and SYGMA 2) compared maintenance therapy with a low-dose inhaled corticosteroid (budesonide) vs. on-demand treatment with an inhaler containing both a corticosteroid (budesonide) and a long-acting bronchodilator (formoterol).
“Using as-needed budesonide-formoterol led to outcomes that are almost as good as taking a maintenance budesonide dose every day,” said Dr. Shaker.
The Global Initiative for Asthma guidelines now recommend this approach – as-needed inhaled corticosteroids (ICS) plus long-acting bronchodilators – for adults with mild asthma. In the United States, however, the National Heart, Lung, and Blood Institute still suggests daily ICS plus quick-relief therapy as needed.
Dr. Shaker and colleagues used computer modeling to compare the cost-effectiveness of as-needed budesonide-formoterol vs. over-the-counter inhaled epinephrine in underinsured U.S. adults who were self-managing their mild asthma. The study randomly assigned these individuals into three groups: OTC inhaled epinephrine (current reality), OTC budesonide-formoterol (not yet available), or no OTC option. The model assumed that patients treated for an exacerbation were referred to a health care provider and started a regimen of ICS plus as-needed rescue therapy.
In this analysis, which has been submitted for publication, the OTC budesonide-formoterol strategy was associated with 12,495 fewer deaths, prevented nearly 14 million severe asthma exacerbations, and saved more than $68 billion. And “when we looked at OTC budesonide-formoterol vs. having no OTC option at all, budesonide-formoterol was similarly cost-effective,” said Dr. Shaker, who presented the results at an AAAAI oral abstract session.
The cost savings emerged even though in the United States asthma controller therapies (for example, fluticasone) cost about 10 times more than rescue therapies (for instance, salbutamol, OTC epinephrine).
Nevertheless, the results make sense. “If you’re using Primatene Mist, your health costs are predicted to be much greater because you’re going to be in the hospital more. Your asthma is not going to be well-controlled,” Thanai Pongdee, MD, an allergist-immunologist with the Mayo Clinic in Rochester, Minn., told this news organization. “It’s not only the cost of your ER visit but also the cost of loss of work or school, and loss of daily productivity. There are all these associated costs.”
The analysis “is certainly something policy makers could take a look at,” he said.
He noted that current use of budesonide-formoterol is stymied by difficulties with insurance coverage. The difficulties stem from a mismatch between the updated recommendation for as-needed use and the description printed on the brand-name product (Symbicort).
“On the product label, it says Symbicort should be used on a daily basis,” Dr. Pongdee said. “But if a prescription comes through and says you’re going to use this ‘as needed,’ the health plan may say that’s not appropriate because that’s not on the product label.”
Given these access challenges with the all-in-one inhaler, other researchers have developed a workaround – asking patients to continue their usual care (that is, using a rescue inhaler as needed) but to also administer a controller medication after each rescue. When tested in Black and Latino patients with moderate to severe asthma, this easy strategy (patient activated reliever-triggered inhaled corticosteroid, or PARTICS) reduced severe asthma exacerbations about as well as the all-in-one inhaler.
If the all-in-one budesonide-formoterol does become available OTC, Dr. Shaker stressed that it “would not be a substitute for seeing an allergist and getting appropriate medical care and an evaluation and all the rest. But it’s better than the status quo. It’s the sort of thing where the perfect is not the enemy of the good,” he said.
Dr. Shaker is the AAAAI cochair of the Joint Task Force on Practice Parameters and serves as an editorial board member of the Journal of Allergy and Clinical Immunology in Practice. He is also an associate editor of the Annals of Allergy, Asthma, and Immunology. Dr. Pongdee serves as an at-large director on the AAAAI board of directors. He receives grant funding from GlaxoSmithKline, and Mayo Clinic is a trial site for GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
FROM AAAAI 2023
Pulmonary hypertension: An update of Dx and Tx guidelines
New guidelines that redefine pulmonary hypertension (PH) by a lower mean pulmonary artery pressure (mPAP) have led to a reported increase in the number of patients given a diagnosis of PH. Although the evaluation and treatment of PH relies on the specialist, as we explain here, family physicians play a pivotal role in the diagnosis, reduction or elimination of risk factors for PH, and timely referral to a pulmonologist or cardiologist who has expertise in managing the disease. We also address the important finding that adult patients who have been evaluated, treated, and followed based on guidelines—updated just last year—have a longer life expectancy than patients who have not been treated properly or not treated at all.
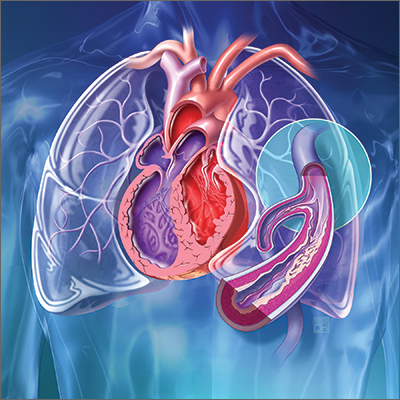
Last, we summarize the etiology, evaluation, and management of PH in the pediatric population.
What is pulmonary hypertension? A revised definition
Prior to 2018, PH was defined as mPAP (measured by right heart catheterization [RHC]) ≥ 25 mm Hg at rest. Now, based on guidelines developed at the 6th World Symposium on Pulmonary Hypertension (WSPH) in 2018, PH is defined as mPAP > 20 mm Hg.1,2 That change was based on studies in which researchers noted higher mortality in adults who had mPAP below the traditional threshold.3,4 There is no evidence, however, of increased mortality in the pediatric population in this lower mPAP range.5
PH is estimated to be present in approximately 1% of the population.6 PH due to other diseases—eg, cardiac disease, lung disease, or a chronic thromboembolic condition—reflects the prevalence of the causative disease.7
How is pulmonary hypertension classified?
Based on the work of a Task Force of the 6th WSPH, PH is classified by underlying pathophysiology, hemodynamics, and functional status. Clinical classification comprises 5 categories, or “groups,” based on underlying pathophysiology (TABLE 16).
Clinical classification
Group 1 PH includes patients with primary pulmonary hypertension, also referred to (including in this article) as pulmonary arterial hypertension (PAH). Hemodynamic criteria that define PAH include pulmonary vascular resistance (PVR) > 2 Woods unitsa and pulmonary capillary wedge pressure > 15 mm Hg. Idiopathic PAH is the most common diagnosis in this group.
The incidence of PAH is approximately 6 cases for every 1 million adults; prevalence is 48 to 55 cases for every 1 million adults. PAH is more common in women.6
Continue to: Less common causes...
Less common causes in Group 1 includ
Group 2 PH comprises patients whose disease results from left heart dysfunction, the most common cause of PH. This subgroup has an elevated pulmonary artery wedge pressure > 15 mm Hg.8 Patients have either isolated postcapillary PH or combined pre-capillary and postcapillary PH.
Group 3 PH comprises patients whose PH is secondary to chronic and hypoxic lung disease. Patients in this group have pre-capillary PH; even a modest elevation in mPAP (20-29 mm Hg) is associated with a poor prognosis. Group 3 patients have elevated PVR, even with mild PH.2 Exertional dyspnea disproportionate to the results of pulmonary function testing, low carbon monoxide diffusion capacity, and rapid decline of arterial oxygenation with exercise all point to severe PH in these patients.9
Group 4 PH encompasses patients with pulmonary artery obstruction, the most common cause of which is related to chronic thromboembolism. Other causes include obstruction of the pulmonary artery from an extrinsic source. Patients with chronic thromboembolic pulmonary hypertension (CTEPH) also have pre-capillary PH, resulting from elevated pulmonary pressures secondary to thromboembolic burden, as well as pulmonary remodeling in unobstructed small arterioles.
Group 5 PH is a miscellaneous group secondary to unclear or multiple causes, including chronic hematologic anemia (eg, sickle cell disease), systemic disorders (eg, sarcoidosis), and metabolic disorders (eg, glycogen storage disease). Patients in Group 5 can have both pre-capillary and postcapillary hypertension.
Classification by functional status
The World Health Organization (WHO) Functional Classification of Patients with Pulmonary Hypertension is divided into 4 classes.10 This system is used to guide treatment and for prognostic purposes:
Class I. Patients have no limitation of physical activity. Ordinary physical activity does not cause undue dyspnea or fatigue, chest pain, or near-syncope.
Continue to: Class II
Class II. Patients have slight limitation of physical activity. They are comfortable at rest but daily physical activity causes dyspnea, fatigue, chest pain, or near-syncope.
Class III. These patients have marked limitation of physical activity. They are comfortable at rest, but less-than-ordinary activity causes dyspnea, fatigue, chest pain, or near-syncope.
Class IV. Patients are unable to carry out any physical activity without symptoms. They manifest signs of right heart failure. Dyspnea or fatigue, or both, might be present even at rest.
How is the pathophysiology of PH described?
The term pulmonary hypertension refers to an elevation in PAP that can result from any number of causes. Pulmonary arterial hypertension is a subcategory of PH in which a rise in PAP is due to primary pathology in the arteries proper.
As noted, PH results from a variety of pathophysiologic mechanisms, reflected in the classification in TABLE 1.6
WSPH Group 1 patients are considered to have PAH; for most, disease is idiopathic. In small-caliber pulmonary arteries, hypertrophy of smooth muscle, endothelial cells, and adventitia leads to increased resistance. Production of nitric oxide and prostacyclins is also impaired in endothelial cells. Genetic mutation, environmental factors such as exposure to stimulant use, and collagen vascular disease have a role in different subtypes of PAH. Portopulmonary hypertension is a subtype of PAH in patients with portal hypertension.
WSPH Groups 2-5. Increased PVR can result from pulmonary vascular congestion due to left heart dysfunction; destruction of the alveolar capillary bed; chronic hypoxic vasoconstriction; and vascular occlusion from thromboembolism.
Continue to: Once approximately...
Once approximately 30% of the pulmonary vasculature is involved, pressure in the pulmonary circulation starts to rise. In all WSPH groups, this increase in PVR results in increased right ventricular afterload that, over time, leads to right ventricular dysfunction.7,11,12
How does PH manifest?
Patients who have PH usually present with dyspnea, fatigue, chest pain, near-syncope, syncope, or lower-extremity edema, or any combination of these symptoms. The nonspecificity of presenting symptoms can lead to a delay in diagnosis.
In addition, suspicion of PH should be raised when a patient:
- presents with skin discoloration (light or dark) or a telangiectatic rash
- presents with difficulty swallowing
- has a history of connective tissue disease or hemolytic anemia
- has risk factors for HIV infection or liver disease
- takes an appetite suppressant
- has been exposed to other toxins known to increase the risk of PH.
A detailed medical history—looking for chronic lung or heart disease, thromboembolism, sleep-disordered breathing, a thyroid disorder, chronic renal failure, or a metabolic disorder—should be obtained.
Common findings on the physical exam in PH include:
- an increased P2 heart sound (pulmonic closure)
- high-pitched holosystolic murmur from tricuspid regurgitation
- pulmonic insufficiency murmur
- jugular venous distension
- hepatojugular reflux
- peripheral edema.
These findings are not specific to PH but, again, their presence warrants consideration of PH.
How best to approach evaluation and diagnosis?
The work-up for PH is broad; FIGURE 113,14 provides an outline of how to proceed when there is a concern for PH. For the work-up of symptoms and signs listed earlier, chest radiography and electrocardiography are recommended.
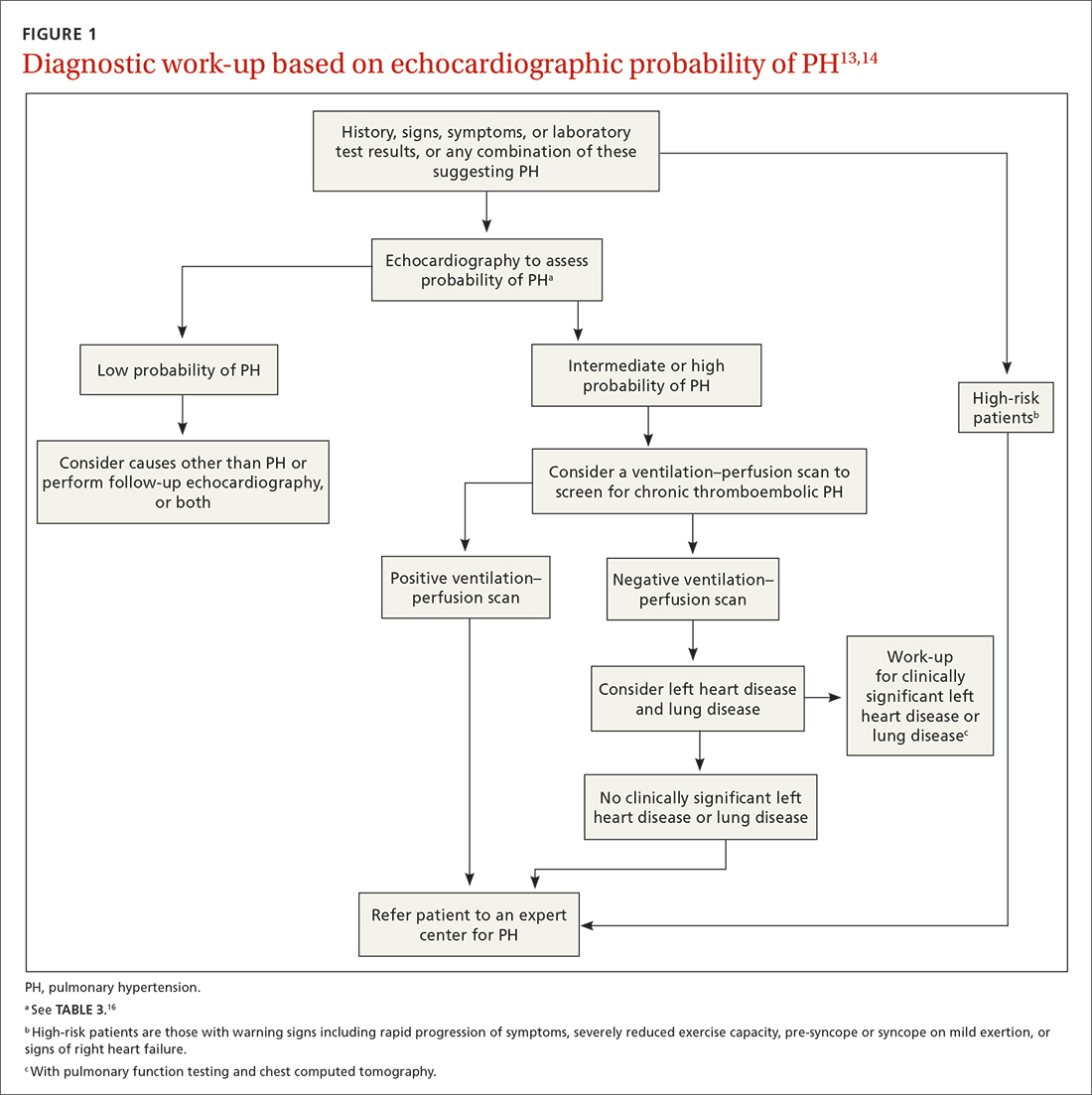
Continue to: Radiographic findings
Radiographic findings that suggest PH include enlargement of central pulmonary arteries and the right ventricle and dilation of the right atrium. Pulmonary vascular congestion might also be seen, secondary to left heart disease.7
Electrocardiographic findings of PH are demonstrated by signs of left ventricular hypertrophy, especially in Group 2 PH. Upright R waves in V1-V2 with deeper S waves in V5-V6 might represent right ventricular hypertrophy or right heart strain. Frequent premature atrial contractions and multifocal atrial tachycardia are also associated with PH.7
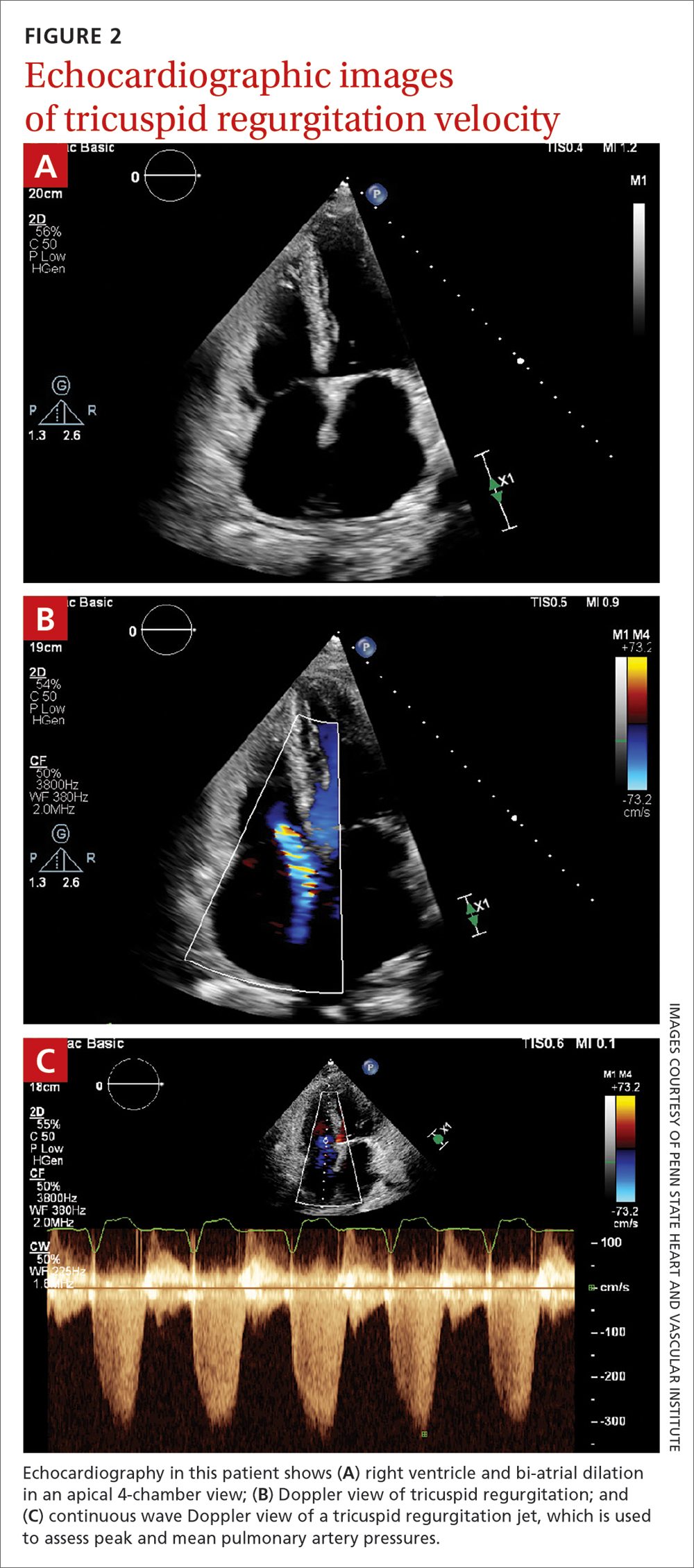
Brain natriuretic peptide (BNP) or N-terminal (NT) proBNP. The level of BNP might be elevated in PH, but its role in the diagnostic process has not been established. BNP can, however, be used to monitor treatment effectiveness and prognosis.15 A normal electrocardiogram in tandem with a normal level of BNP or NT-proBNP is associated with a low likelihood of PH.6
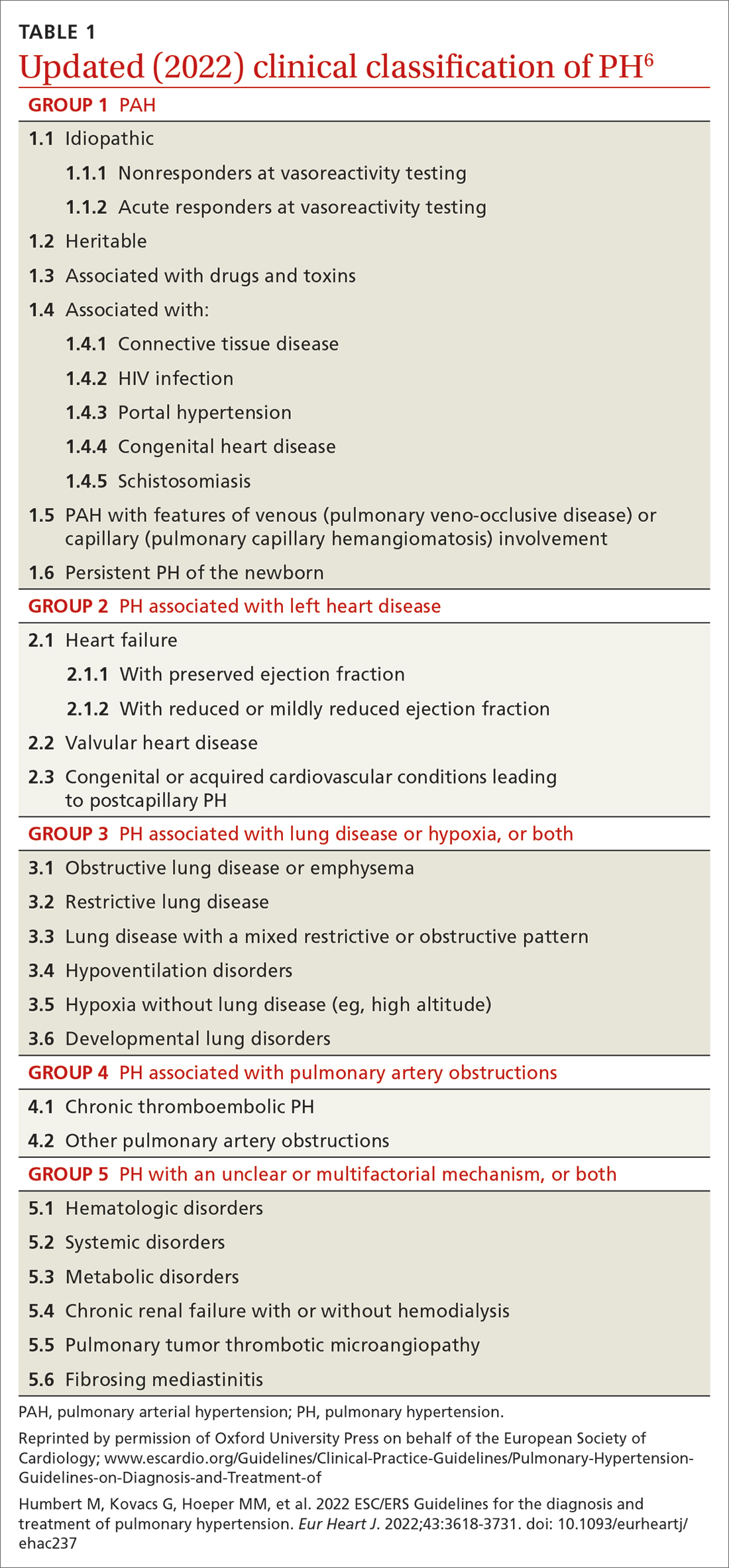
Transthoracic echocardiography (TTE) is the initial evaluation tool whenever PH is suspected. Echocardiographic findings suggestive of PH include a combination of tricuspid regurgitation velocity > 2.8 m/s (FIGURE 2); estimated pulmonary artery systolic pressure > 35 mm Hg in younger adults and > 40 mm Hg in older adults; right ventricular hypertrophy or strain; or a combination of these. Other TTE findings suggestive of PH are related to the ventricles, pulmonary artery, inferior vena cava, and right atrium (TABLE 26). The probability of PH based on TTE findings is categorized as low, intermediate, or high (see TABLE 26 and TABLE 316 for details).

Older guidelines, still used by some, rely on the estimated pulmonary artery systolic pressure (ePASP) reading on echocardiography.13,17 However, studies have reported poor correlation between ePASP readings and values obtained from RHC.18
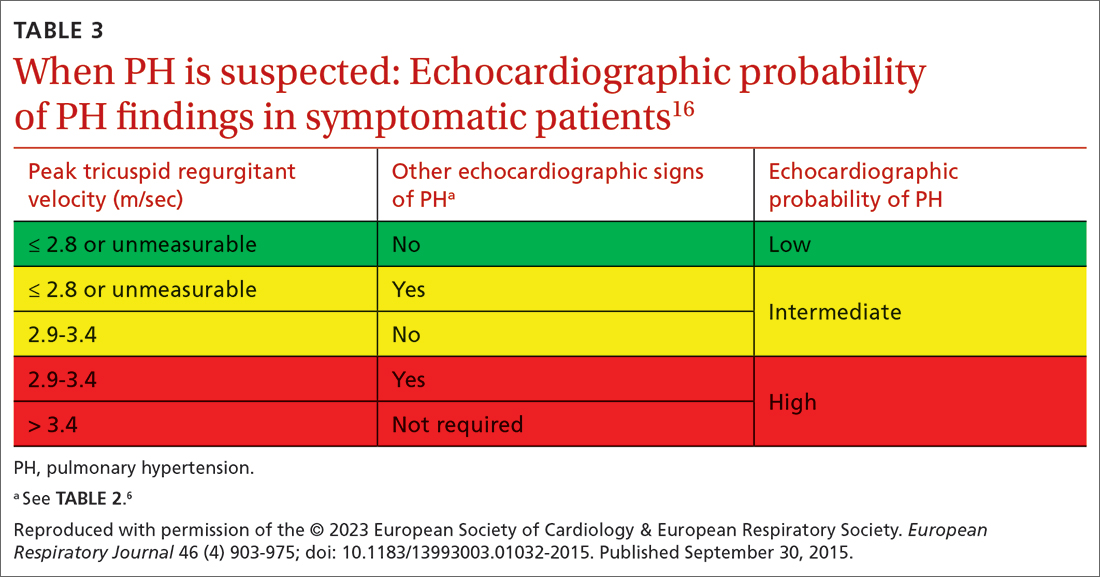
TTE also provides findings of left heart disease, such as left ventricular systolic and diastolic dysfunction and left-sided valvular pathology. Patients with suspected PH in whom evidence of left heart disease on TTE is insufficient for making the diagnosis should receive further evaluation for their possible status in Groups 3-5 PH.
Ventilation–perfusion (VQ) scan. If CTEPH is suspected, a VQ scan should be performed. The scan is highly sensitive for CTEPH; a normal VQ scan excludes CTEPH. Computed tomography (CT) of the chest is not helpful for identifying chronic thromboembolism.13
Continue to: Coagulation assays
Coagulation assays. When CTEPH is suspected, coagulopathy can be assessed by measuring anticardiolipin antibodies, lupus anticoagulant, and anti-b-2-glycoprotein antibodies.13
Chest CT will show radiographic findings in greater detail. An enlarged pulmonary artery (diameter ≥ 29 mm) or a ratio ≥ 1 of the diameter of the main pulmonary artery to the diameter of the ascending aorta is suggestive of PH.
Other tests. Overnight oximetry and testing for sleep-disordered breathing, performed in an appropriate setting, can be considered.13,14,19
Pulmonary function testing with diffusion capacity for carbon monoxide, high-resolution chest CT, and a 6-minute walk test (6MWT) can be considered in patients who have risk factors for chronic lung disease. Pulmonary function testing, including measurement of the diffusing capacity of the lungs for carbon monoxide, arterial blood gas analysis, and CT, is used to aid in interpreting echocardiographic findings in patients with lung disease in whom PH is suspected.
Testing for comorbidities. A given patient’s predisposing conditions for PH might already be known; if not, laboratory evaluation for conditions such as sickle cell disease, liver disease, thyroid dysfunction, connective tissue disorders (antibody tests of antinuclear antibody, rheumatoid factor, anticentromere, anti-topoisomerase, anti-RNA polymerase III, anti-double stranded DNA, anti-Ro, anti-La, and anti-U1-RNP), and vasculitis (anti-neutrophil cytoplasmic autoantibodies) should be undertaken.
Analysis of stool and urine for Schistosoma spp parasites can be considered in an appropriate clinical setting.13
Right heart catheterization. Once alternative diagnoses are excluded, RHC is recommended to make a definitive diagnosis and assess the contribution of left heart disease. Vasoreactivity—defined as a reduction in mPAP ≥ 10 mm Hg to reach an absolute value of mPAP ≤ 40 mm Hg with increased or unchanged cardiac output—is assessed during RHC by administering nitric oxide or another vasodilator. This definition of vasoreactivity helps guide medical management in patients with PAH.7,20
Continue to: 6MWT
6MWT. Once the diagnosis of PH is made, a 6MWT helps establish baseline functional performance and will help you to monitor disease progression.
Who can benefit from screening for PH?
Annual evaluation of the risk of PAH is recommended for patients with systemic sclerosis or portal hypertension13 and can be considered in patients who have connective tissue disease with overlap features of systemic sclerosis.
Assessment for CTEPH or chronic thromboembolic pulmonary disease is recommended for patients with persistent or new-onset dyspnea or exercise limitation after pulmonary embolism.
Screening echocardiography for PH is recommended for patients who have been referred for liver transplantation.6
How risk is stratified
Risk stratification is used to manage PH and assess prognosis.
At diagnosis. Application of a 3-strata model of risk assessment (low, intermediate, high) is recommended.6 Pertinent data to determine risk include signs of right heart failure, progression of symptoms and clinical manifestations, report of syncope, WHO functional class, 6MWT, cardiopulmonary exercise testing, biomarkers (BNP or NT-proBNP), echocardiography, presence of pericardial effusion, and cardiac magnetic resonance imaging.
At follow-up. Use of a 4-strata model (low, intermediate–low, intermediate–high, and high risk) is recommended. Data used are WHO functional class, 6MWT, and results of either BNP or NT-proBNP testing.6
Continue to: When to refer
When to refer
Specialty consultation21-23 is recommended for:
- all patients with PAH
- PH patients in clinical Groups 2 and 3 whose disease is disproportionate to the extent of their left heart disease or hypoxic lung disease
- patients in whom there is concern about CTEPH and who therefore require early referral to a specialist for definitive treatment
- patients in whom the cause of PH is unclear or multifactorial (ie, clinical Group 5).
What are the options for managing PH?
Management of PH is based on the cause and classification of the individual patient’s disease.
Treatment for WSPH Group 1
Patients require referral to a specialty clinic for diagnosis, treatment, and monitoring of progression.10
First, regrettably, none of the medications approved by the US Food and Drug Administration for treating PAH prevent progression.7
Patients with idiopathic, hereditary, or drug-induced PAH with positive vasoreactivity are treated with a calcium channel blocker (CCB). The dosage is titrated to optimize therapy for the individual patient.
The patient is then reassessed after 3 to 6 months of medical therapy. Current treatment is continued if the following goals have been met:
- WHO functional classification is I or II
- BNP < 50 ng/L or NT-proBNP < 300 ng/L
- hemodynamics are normal or near-normal (mPAP ≤ 30 mm Hg and PVR ≤ 4 WU).
If these goals have not been met, treatment is adjusted by following the algorithm described below.
Continue to: The treatment algorithm...
The treatment algorithm for idiopathic-, heritable-, drug-induced, and connective tissue disease–associated PAH highlights the importance of cardiopulmonary comorbidities and risk strata at the time treatment is initiated and then during follow-up.
Cardiopulmonary comorbidities are conditions associated with an increased risk of left ventricular diastolic dysfunction, including obesity, hypertension, diabetes, and coronary artery disease. Pulmonary comorbidities can include signs of mild parenchymal lung disease and are often associated with a low carbon monoxide diffusing capacity (< 45% of predicted value).
The management algorithm proceeds as follows:
- For patients without cardiopulmonary comorbidities and who are at low or intermediate risk, treatment of PAH with an endothelin receptor antagonist (ERA) plus a phosphodiesterase-5 (PDE5) inhibitor is recommended.
- For patients without cardiopulmonary comorbidities and who are at high risk, treatment with an ERA, a PDE5 inhibitor, and either an IV or subcutaneous prostacyclin analogue (PCA) can be considered.
- Patients in either of the preceding 2 categories should have regular follow-up assessment; at such follow-up, their risk should be stratified based on 4 strata (see “How risk is stratified”):
- Low risk: Continue initial therapy.
- Low-to-intermediate risk: Consider adding a prostacyclin receptor agonist to the initial regimen or switch to a PDE5 inhibitor or a soluble guanylate cyclase stimulator.
- Intermediate-to-high or high risk: Consider adding a PCA (IV epoprostenol or IV or subcutaneous treprostinil). In addition, or alternatively, have the patient evaluated for lung transplantation.
- For patients with cardiopulmonary comorbidity—in any risk category—consider oral monotherapy with a PDE5 inhibitor or an ERA. Provide regular follow-up and individualize therapy.6
Treatment for WSPH Groups 2 and 3
Treatment is focused on the underlying cause of PH:
- Patients who have left heart disease with either severe pre-capillary component PH or markers of right ventricular dysfunction, or both, should be referred to a PH center.
- Patients with combined pre-capillary and postcapillary PH in whom pre-capillary PH is severe should be considered for an individualized approach.
- Consider prescribing the ERA bosentan in specific scenarios (eg, the Eisenmenger syndrome of left-right shunting resulting from a congenital cardiac defect) to improve exercise capacity. If PAH persists after corrected adult congenital heart disease, follow the PAH treatment algorithm for Group 1 patients (described earlier).
- For patients in Group 3, those who have severe PH should be referred to a PH center.
- Consider prescribing inhaled treprostinil in PH with interstitial lung disease.
Treatment for WSPH Group 4
Patients with CTEPH are the only ones for whom pulmonary endarterectomy (PEA), the treatment of choice, might be curative. Balloon angioplasty can be considered for inoperable cases6; these patients should be placed on lifelong anticoagulant therapy.
Symptomatic patients who have inoperable CTEPH or persistent recurrent PH after PEA are medically managed; the agent of choice is riociguat. Patients who have undergone PEA or balloon angioplasty and those receiving pharmacotherapy should be followed long term.
Treatment for WSPH Group 5
Management of these patients focuses on associated conditions.
Continue to: Which medications for PAH?
Which medications for PAH?
CCBs. Four options in this class have shown utility, notably in patients who have had a positive vasoreactivity test (see “How best to approach evaluation and diagnosis?”):
- Nifedipine is started at 10 mg tid; target dosage is 20 to 60 mg, bid or tid.
- Diltiazem is started at 60 mg bid; target dosage is 120 to 360 mg bid.
- Amlodipine is started at 5 mg/d; target dosage is 15 to 30 mg/d.
- Felodipine is started at 5 mg/d; target dosage is 15 to 30 mg/d.
Felodipine and amlodipine have longer half-lives than other CCBs and are well tolerated.
ERA. Used as vasodilators are ambrinsentan (starting dosage, 5 mg/d; target dosage, 10 mg/d), macitentan (starting and target dosage, 10 mg/d), and bosentan (starting dosage, 62.5 mg bid; target dosage, 125 mg bid).
Nitric oxide–cyclic guanosine monophosphate enhancers. These are the PDE5 inhibitors sildenafil (starting and target dosages, 20 mg tid) and tadalafil (starting dosage, 20 or 40 mg/d; target dosage, 40 mg/d), and the guanylate cyclase stimulant riociguat (starting dosage, 1 mg tid; target dosage, 2.5 mg tid). All 3 agents enhance production of the potent vasodilator nitric oxide, production of which is impaired in PH.
Prostanoids. Several options are available:
- Beraprost sodium. For this oral prostacyclin analogue, starting dosage is 20 μg tid; target dosage is the maximum tolerated dosage (as high as 40 μg tid).
- Extended-release beraprost. Starting dosage is 60 μg bid; target dosage is the maximum tolerated dosage (as high as 180 μg bid).
- Oral treprostinil. Starting dosage is 0.25 mg bid or 0.125 mg tid; target dosage is the maximum tolerated dosage.
- Inhaled iloprost. Starting dosage of this prostacyclin analogue is 2.5 μg, 6 to 9 times per day; target dosage is 5 μg, 6 to 9 times per day.
- Inhaled treprostinil. Starting dosage is 18 μg qid; target dosage is 54 to 72 μg qid.
- Eproprostenol is administered by continuous IV infusion, at a starting dosage of 2 ng/kg/min; target dosage is determined by tolerability and effectiveness (typically, 30 ng/kg/min).
- IV treprostinil. Starting dosage 1.25 ng/kg/min; target dosage is determined by tolerability and effectiveness, with a typical dosage of 60 ng/kg/min.
Combination treatment with the agents listed above is often utilized.
Selexipag. This oral selective nonprostainoid prostacyclin receptor agonist is started at 200 μg bid; target dosage is the maximum tolerated, as high as 1600 μg bid.
Continue to: Supportive therapy
Supportive therapy
The need for oxygen should be addressed in patients with hypoxia in any setting—resting, exercise induced, and nocturnal.24 Patients with an arterial blood oxygen pressure < 60 mm Hg (SaO2 < 90 mm Hg) should be on long-term oxygen therapy.6
Diuretics are beneficial in patients with chronic fluid retention from PH that is related to right ventricular failure.24
Pulmonary rehabilitation and exercise. Contrary to common belief that exercise training is contraindicated in patients with PH, exercise training has emerged in the past decade as an effective tool to improve exercise capacity, ventilatory efficiency, and quality of life. While a patient is training, oxygen saturation, measured by pulse oximetry, should be maintained at > 90% throughout the exercise session to avoid hypoxic pulmonary artery vasoconstriction.25
A patient who does not qualify for pulmonary or cardiac rehabilitation should be referred for physical therapy.24
Ongoing follow-up in primary care
Instruct patients not to abruptly discontinue medications that have been prescribed for PH. Ongoing follow-up and monitoring involves assessing right heart function, exercise tolerance, and resting and ambulatory oximetry. Testing for the level of BNP provides prognostic information and allows assessment of treatment response.15 The frequency of 6MWT, echocardiography, and RHC is decided on a case-by-case basis.
Other considerations
Pregnancy. PAH often affects patients of childbearing age. Because PAH-associated maternal mortality and the risk to the fetus during pregnancy are high, pregnancy is not recommended for patients with PAH. After a diagnosis of PAH in a patient of childbearing age, counseling should be offered at an expert center. Advice on effective contraception methods should be given early on.10,26-29
Surgery. Every patient with clinically significant PH is at increased risk of perioperative morbidity and death.30,31 Guidelines recommend that these patients avoid nonessential surgery; if surgery is necessary, care should be provided at a PH expert center.10
Continue to: Patients with severe PH...
Patients with severe PH should consider surgery for any indication carefully, discussing with the care team their risk and exploring nonsurgical options. Cardiothoracic surgical and liver transplantation services might have highly specific criteria for treating patients with PH, but other essential and nonessential surgeries require individualized risk stratification. Surgery for patients with severe PH and right ventricular dysfunction should be performed at a center equipped to handle high-risk patients.
Other preventive measures. Patients with PAH should6,10:
- remain current with immunization against influenza virus, SARS-CoV-2, and pneumococcal pneumonia
- avoid high altitudes
- use supplemental oxygen during air travel to keep arterial oxygen saturation > 91%.
Lung transplantation. Patients eligible for transplantation who (1) are at intermediate-to-high risk or high risk or (2) have a REVEAL (Registry to EValuate Early And Long-term pulmonary arterial hypertension disease management) risk score > 7, and who have had an inadequate response to oral combination therapy, should be referred for evaluation for lung transplantation. Placement on the list for lung transplantation is also recommended for patients at high risk of death and who have a REVEAL risk score ≥ 10 despite medical therapy, including a subcutaneous or IV prostacyclin analogue.6
PH in infants and children
The Pediatric Task Force of the 6th WSPH has applied the new definition proposed for adult PH (> 20 mm Hg mPAP) to children and infants > 3 months of age (see “Pulmonary hypertension in the pediatric population,” at left32-36).
SIDEBAR
Pulmonary hypertension in the pediatric population
The onset of pulmonary hypertension (PH) in children can occur at any age and be of quite different causes than in adults. In newborns, pulmonary pressure drops rapidly during the week after delivery; in some cases, however, pressures remain elevated (> 20 mm Hg) despite healthy lungs. These asymptomatic newborns require close monitoring.32
Etiology. Pediatric PH can be persistent or transient. Prominent causes of persistent or progressive PH in children are pulmonary arterial hypertension (PAH) associated with congenital heart disease and developmental lung disease, such as bronchopulmonary dysplasia and idiopathic PAH. Major categories of congenital heart disease that cause PH are shunting lesions and left heart disease associated with elevated atrial pressure. Other causes are rare.33
Persistent PH of the newborn (PPHN) and PH due to diaphragmatic hernia are common causes of transient PH.34 In PPHN, pulmonary vascular resistance remains abnormally high after birth, resulting in right-to-left shunting of the circulation that, in turn, leads to hypoxemia unresponsive to usual measures. In most cases, signs of respiratory distress and hypoxia are noted within the first 24 hours of life. The most common cause of PPHN is infection.35
Evaluation. The typical diagnostic work-up of suspected pediatric PH is similar to what is undertaken in the adult population—varying, however, according to the specific suspected cause. As in adults, right heart catheterization remains the gold standard of diagnosis, and should be conducted at a pediatric PH expert center. As with adult patients, infants and children with PH should be managed by a multidisciplinary expert team.
Management. PAH-targeted medications (see “What are the options for managing PH?”) are used to treat PAH in children.36
CORRESPONDENCE
Madhavi Singh, MD, 1850 East Park Ave., Suite 207, State College, PA 16803; [email protected]
1. Galiè N, McLaughlin VV, Rubin LJ, et al. An overview of the 6th World Symposium on Pulmonary Hypertension. Eur Respir J. 2019;53:1802148. doi: 10.1183/13993003.02148-2018
2. Simonneau G, Montani D, Celermajer DS, et al. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J. 2019;53:1801913. doi: 10.1183/13993003.01913-2018
3. Kolte D, Lakshmanan S, Jankowich MD, et al. Mild pulmonary hypertension is associated with increased mortality: a systematic review and meta-analysis. J Am Heart Assoc. 2018;7:e009729. doi: 10.1161/JAHA.118.009729
4. Douschan P, Kovacs G, Avian A, et al. Mild elevation of pulmonary arterial pressure as a predictor of mortality. Am J Respir Crit Care Med. 2018;197:509-516. doi: 10.1164/rccm.201706-1215OC
5. Lammers AE, Apitz C. Update from the World Symposium on Pulmonary Hypertension 2018: does the new hemodynamic definition of pediatric pulmonary hypertension have an impact on treatment strategies? Cardiovasc Diagn Ther. 2021;11:1048-1051. doi: 10.21037/cdt-20-412
6. Humbert M, Kovacs G, Hoeper MM, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2022;43:3618-3731. doi: 10.1093/eurheartj/ehac237
7. Oldroyd SH, Manek G, Bhardwaj A. Pulmonary hypertension. In: StatPearls [Internet]. StatPearls Publishing. Updated July 20, 2022. Accessed November 27, 2022. www.ncbi.nlm.nih.gov/books/NBK482463/?report=classic
8. Vachiéry JL, Tedford RJ, Rosenkranz S, et al. Pulmonary hypertension due to left heart disease. Eur Respir J. 2019;53:1801897. doi: 10.1183/13993003.01897-2018
9. Seeger W, Adir Y, Barberà JA, et al. Pulmonary hypertension in chronic lung diseases. J Am Coll Cardiol. 2013;62(25 suppl):D109-D116. doi: 10.1016/j.jacc.2013.10.036
10. Taichman DB, Ornelas J, Chung L, et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST guideline and expert panel report. Chest. 2014;146:449-475. doi: 10.1378/chest.14-0793
11. Krowl L, Anjum F, Kaul P. Pulmonary idiopathic hypertension. In: StatPearls [Internet]. StatPearls Publishing. Updated August 8, 2022. Accessed November 27, 2022. www.ncbi.nlm.nih.gov/books/NBK519041/#_NBK519041_pubdet_
12. Bartolome SD. Portopulmonary hypertension: diagnosis, clinical features, and medical therapy. Clin Liver Dis (Hoboken). 2014;4:42-45. doi: 10.1002/cld.401
13. Frost A, Badesch D, Gibbs JSR, et al. Diagnosis of pulmonary hypertension. Eur Respir J. 2019;53:1801904. doi: 10.1183/ 13993003.01904-2018
14. Yaghi S, Novikov A, Trandafirescu T. Clinical update on pulmonary hypertension. J Investig Med. 2020;68:821-827. doi: 10.1136/jim-2020-001291
15. Chin KM, Rubin LJ, Channick R, et al. Association of N-terminal pro brain natriuretic peptide and long-term outcome in patients with pulmonary arterial hypertension. Circulation. 2019;139:2440-2450. doi: 10.1161/CIRCULATIONAHA.118.039360
16. Galiè N, Humbert M, Vachiery J-L, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J. 2015;46:903-975. doi: 10.1183/13993003.01032-2015
17. N,, , et al; Task Force for Diagnosis and Treatment of Pulmonary Hypertension of European Society of Cardiology (ESC); European Respiratory Society (ERS); International Society of Heart and Lung Transplantation (ISHLT). Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Respir J. 2009;34:1219-1263. doi: 10.1183/09031936.00139009
18. Rich JD, Shah SJ, Swamy RS, et al. Inaccuracy of Doppler echocardiographic estimates of pulmonary artery pressures in patients with pulmonary hypertension: implications for clinical practice. Chest. 2011;139:988-993. doi: 10.1378/chest.10-1269
19. Janda S, Shahidi N, Gin K, et al. Diagnostic accuracy of echocardiography for pulmonary hypertension: a systematic review and meta-analysis. Heart. 2011;97:612-622. doi: 10.1136/hrt.2010.212084
20. Farber HW, Foreman AJ, Miller DP, et al. REVEAL Registry: correlation of right heart catheterization and echocardiography in patients with pulmonary arterial hypertension. Congest Heart Fail. 2011;17:56-63. doi: 10.1111/j.1751-7133.2010.00202.x
21. Suntharalingam J, Ross RM, Easaw J, et al. Who should be referred to a specialist pulmonary hypertension centre—a referrer’s guide. Clin Med (Lond). 2016;16:135-141. doi: 10.7861/clinmedicine.16-2-135
22. Deaño RC, Glassner-Kolmin C, Rubenfire M, et al. Referral of patients with pulmonary hypertension diagnoses to tertiary pulmonary hypertension centers: the multicenter RePHerral Study. JAMA Intern Med. 2013;173:887-893. doi: 10.1001/jamainternmed.2013.319
23. Guidelines for referring patients with pulmonary hypertension. Royal Papworth Hospital, NHS Foundation Trust. Updated February 2019. Accessed November 27, 2022. https://royalpapworth.nhs.uk/application/files/9015/5014/6935/PVDU-Referral-guidelines-2019.pdf
24. Yuan P, Yuan X-T, Sun X-Y, et al. Exercise training for pulmonary hypertension: a systematic review and meta-analysis. Int J Cardiol. 2015;178:142-146. doi: 10.1016/j.ijcard.2014.10.161
25. Spruit MA, Singh SJ, Garvey C, et al; . An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188:e13-e64. doi: 10.1164/rccm.201309-1634ST
26. Olsson KM, Channick R. Pregnancy in pulmonary arterial hypertension. Eur Respir Rev. 2016;25:431-437. doi: 10.1183/ 16000617.0079-2016
27. Weiss BM, Zemp L, Swifert B, et al. Outcome of pulmonary vascular disease in pregnancy: a systematic overview from 1978 through 1996; J Am Coll Cardiol. 1998;31:1650-1657. doi: 10.1016/s0735-1097(98)00162-4
28. Qiangqiang Li, Dimopoulos K, Liu T, et al, Peripartum outcomes in a large population of women with pulmonary arterial hypertension associated with congenital heart disease, Euro J Prev Cardiol. 2019;26:1067-1076. doi: 10.1177/2047487318821246
29. Olsson KM, Jaïs X. Birth control and pregnancy management in pulmonary hypertension. Semin Respir Crit Care Med. 2013;34:681-688. doi: 10.1055/s-0033-1355438
30. Price LC, Montani D, Jaïs X, et al. Noncardiothoracic nonobstetric surgery in mild-to-moderate pulmonary hypertension. Eur Respir J. 2010;35:1294-1302. doi: 10.1183/09031936.00113009
31. Memtsoudis SG, Ma Y, Chiu YL, et al. Perioperative mortality in patients with pulmonary hypertension undergoing major joint replacement. Anesth Analg. 2010;111:1110-1116. doi: 10.1213/ANE.0b013e3181f43149
32. Rosenzweig EB, Abman SH, Adatia I, et al. Paediatric pulmonary arterial hypertension: updates on definition, classification, diagnostics and management. Eur Respir J. 2019;53:1801916. doi: 10.1183/13993003.01916-2018
33. Berger RMF, Beghetti M, Humpl T, et al. Clinical features of paediatric pulmonary hypertension: a registry study. Lancet. 2012;379:537-546. doi: 10.1016/S0140-6736(11)61621-8
34. van Loon RL, Roofthooft MTR, Hillege HL, et al. Pediatric pulmonary hypertension in the Netherlands: epidemiology and characterization during the period 1991 to 2005. Circulation. 2011;124:1755-1764. doi: 10.1161/CIRCULATIONAHA.110.969584
35. Steurer MA, Jelliffe-Pawlowski LL, Baer RJ, et al. Persistent pulmonary hypertension of the newborn in late preterm and term infants in California. Pediatrics. 2017;139:e20161165. doi: 10.1542/peds.2016-1165
36. Hansmann G, Koestenberger M, Alastalo TP, et al. 2019 updated consensus statement on the diagnosis and treatment of pediatric pulmonary hypertension: the European Pediatric Pulmonary Vascular Disease Network (EPPVDN), endorsed by AEPC, ESPR and ISHLT. J Heart Lung Transplant. 2019;38:879-901. doi: 10.1016/j.healun.2019.06.022
New guidelines that redefine pulmonary hypertension (PH) by a lower mean pulmonary artery pressure (mPAP) have led to a reported increase in the number of patients given a diagnosis of PH. Although the evaluation and treatment of PH relies on the specialist, as we explain here, family physicians play a pivotal role in the diagnosis, reduction or elimination of risk factors for PH, and timely referral to a pulmonologist or cardiologist who has expertise in managing the disease. We also address the important finding that adult patients who have been evaluated, treated, and followed based on guidelines—updated just last year—have a longer life expectancy than patients who have not been treated properly or not treated at all.

Last, we summarize the etiology, evaluation, and management of PH in the pediatric population.
What is pulmonary hypertension? A revised definition
Prior to 2018, PH was defined as mPAP (measured by right heart catheterization [RHC]) ≥ 25 mm Hg at rest. Now, based on guidelines developed at the 6th World Symposium on Pulmonary Hypertension (WSPH) in 2018, PH is defined as mPAP > 20 mm Hg.1,2 That change was based on studies in which researchers noted higher mortality in adults who had mPAP below the traditional threshold.3,4 There is no evidence, however, of increased mortality in the pediatric population in this lower mPAP range.5
PH is estimated to be present in approximately 1% of the population.6 PH due to other diseases—eg, cardiac disease, lung disease, or a chronic thromboembolic condition—reflects the prevalence of the causative disease.7
How is pulmonary hypertension classified?
Based on the work of a Task Force of the 6th WSPH, PH is classified by underlying pathophysiology, hemodynamics, and functional status. Clinical classification comprises 5 categories, or “groups,” based on underlying pathophysiology (TABLE 16).
Clinical classification
Group 1 PH includes patients with primary pulmonary hypertension, also referred to (including in this article) as pulmonary arterial hypertension (PAH). Hemodynamic criteria that define PAH include pulmonary vascular resistance (PVR) > 2 Woods unitsa and pulmonary capillary wedge pressure > 15 mm Hg. Idiopathic PAH is the most common diagnosis in this group.
The incidence of PAH is approximately 6 cases for every 1 million adults; prevalence is 48 to 55 cases for every 1 million adults. PAH is more common in women.6
Continue to: Less common causes...
Less common causes in Group 1 includ
Group 2 PH comprises patients whose disease results from left heart dysfunction, the most common cause of PH. This subgroup has an elevated pulmonary artery wedge pressure > 15 mm Hg.8 Patients have either isolated postcapillary PH or combined pre-capillary and postcapillary PH.
Group 3 PH comprises patients whose PH is secondary to chronic and hypoxic lung disease. Patients in this group have pre-capillary PH; even a modest elevation in mPAP (20-29 mm Hg) is associated with a poor prognosis. Group 3 patients have elevated PVR, even with mild PH.2 Exertional dyspnea disproportionate to the results of pulmonary function testing, low carbon monoxide diffusion capacity, and rapid decline of arterial oxygenation with exercise all point to severe PH in these patients.9
Group 4 PH encompasses patients with pulmonary artery obstruction, the most common cause of which is related to chronic thromboembolism. Other causes include obstruction of the pulmonary artery from an extrinsic source. Patients with chronic thromboembolic pulmonary hypertension (CTEPH) also have pre-capillary PH, resulting from elevated pulmonary pressures secondary to thromboembolic burden, as well as pulmonary remodeling in unobstructed small arterioles.
Group 5 PH is a miscellaneous group secondary to unclear or multiple causes, including chronic hematologic anemia (eg, sickle cell disease), systemic disorders (eg, sarcoidosis), and metabolic disorders (eg, glycogen storage disease). Patients in Group 5 can have both pre-capillary and postcapillary hypertension.
Classification by functional status
The World Health Organization (WHO) Functional Classification of Patients with Pulmonary Hypertension is divided into 4 classes.10 This system is used to guide treatment and for prognostic purposes:
Class I. Patients have no limitation of physical activity. Ordinary physical activity does not cause undue dyspnea or fatigue, chest pain, or near-syncope.
Continue to: Class II
Class II. Patients have slight limitation of physical activity. They are comfortable at rest but daily physical activity causes dyspnea, fatigue, chest pain, or near-syncope.
Class III. These patients have marked limitation of physical activity. They are comfortable at rest, but less-than-ordinary activity causes dyspnea, fatigue, chest pain, or near-syncope.
Class IV. Patients are unable to carry out any physical activity without symptoms. They manifest signs of right heart failure. Dyspnea or fatigue, or both, might be present even at rest.
How is the pathophysiology of PH described?
The term pulmonary hypertension refers to an elevation in PAP that can result from any number of causes. Pulmonary arterial hypertension is a subcategory of PH in which a rise in PAP is due to primary pathology in the arteries proper.
As noted, PH results from a variety of pathophysiologic mechanisms, reflected in the classification in TABLE 1.6
WSPH Group 1 patients are considered to have PAH; for most, disease is idiopathic. In small-caliber pulmonary arteries, hypertrophy of smooth muscle, endothelial cells, and adventitia leads to increased resistance. Production of nitric oxide and prostacyclins is also impaired in endothelial cells. Genetic mutation, environmental factors such as exposure to stimulant use, and collagen vascular disease have a role in different subtypes of PAH. Portopulmonary hypertension is a subtype of PAH in patients with portal hypertension.
WSPH Groups 2-5. Increased PVR can result from pulmonary vascular congestion due to left heart dysfunction; destruction of the alveolar capillary bed; chronic hypoxic vasoconstriction; and vascular occlusion from thromboembolism.
Continue to: Once approximately...
Once approximately 30% of the pulmonary vasculature is involved, pressure in the pulmonary circulation starts to rise. In all WSPH groups, this increase in PVR results in increased right ventricular afterload that, over time, leads to right ventricular dysfunction.7,11,12
How does PH manifest?
Patients who have PH usually present with dyspnea, fatigue, chest pain, near-syncope, syncope, or lower-extremity edema, or any combination of these symptoms. The nonspecificity of presenting symptoms can lead to a delay in diagnosis.
In addition, suspicion of PH should be raised when a patient:
- presents with skin discoloration (light or dark) or a telangiectatic rash
- presents with difficulty swallowing
- has a history of connective tissue disease or hemolytic anemia
- has risk factors for HIV infection or liver disease
- takes an appetite suppressant
- has been exposed to other toxins known to increase the risk of PH.
A detailed medical history—looking for chronic lung or heart disease, thromboembolism, sleep-disordered breathing, a thyroid disorder, chronic renal failure, or a metabolic disorder—should be obtained.
Common findings on the physical exam in PH include:
- an increased P2 heart sound (pulmonic closure)
- high-pitched holosystolic murmur from tricuspid regurgitation
- pulmonic insufficiency murmur
- jugular venous distension
- hepatojugular reflux
- peripheral edema.
These findings are not specific to PH but, again, their presence warrants consideration of PH.
How best to approach evaluation and diagnosis?
The work-up for PH is broad; FIGURE 113,14 provides an outline of how to proceed when there is a concern for PH. For the work-up of symptoms and signs listed earlier, chest radiography and electrocardiography are recommended.

Continue to: Radiographic findings
Radiographic findings that suggest PH include enlargement of central pulmonary arteries and the right ventricle and dilation of the right atrium. Pulmonary vascular congestion might also be seen, secondary to left heart disease.7
Electrocardiographic findings of PH are demonstrated by signs of left ventricular hypertrophy, especially in Group 2 PH. Upright R waves in V1-V2 with deeper S waves in V5-V6 might represent right ventricular hypertrophy or right heart strain. Frequent premature atrial contractions and multifocal atrial tachycardia are also associated with PH.7

Brain natriuretic peptide (BNP) or N-terminal (NT) proBNP. The level of BNP might be elevated in PH, but its role in the diagnostic process has not been established. BNP can, however, be used to monitor treatment effectiveness and prognosis.15 A normal electrocardiogram in tandem with a normal level of BNP or NT-proBNP is associated with a low likelihood of PH.6

Transthoracic echocardiography (TTE) is the initial evaluation tool whenever PH is suspected. Echocardiographic findings suggestive of PH include a combination of tricuspid regurgitation velocity > 2.8 m/s (FIGURE 2); estimated pulmonary artery systolic pressure > 35 mm Hg in younger adults and > 40 mm Hg in older adults; right ventricular hypertrophy or strain; or a combination of these. Other TTE findings suggestive of PH are related to the ventricles, pulmonary artery, inferior vena cava, and right atrium (TABLE 26). The probability of PH based on TTE findings is categorized as low, intermediate, or high (see TABLE 26 and TABLE 316 for details).

Older guidelines, still used by some, rely on the estimated pulmonary artery systolic pressure (ePASP) reading on echocardiography.13,17 However, studies have reported poor correlation between ePASP readings and values obtained from RHC.18

TTE also provides findings of left heart disease, such as left ventricular systolic and diastolic dysfunction and left-sided valvular pathology. Patients with suspected PH in whom evidence of left heart disease on TTE is insufficient for making the diagnosis should receive further evaluation for their possible status in Groups 3-5 PH.
Ventilation–perfusion (VQ) scan. If CTEPH is suspected, a VQ scan should be performed. The scan is highly sensitive for CTEPH; a normal VQ scan excludes CTEPH. Computed tomography (CT) of the chest is not helpful for identifying chronic thromboembolism.13
Continue to: Coagulation assays
Coagulation assays. When CTEPH is suspected, coagulopathy can be assessed by measuring anticardiolipin antibodies, lupus anticoagulant, and anti-b-2-glycoprotein antibodies.13
Chest CT will show radiographic findings in greater detail. An enlarged pulmonary artery (diameter ≥ 29 mm) or a ratio ≥ 1 of the diameter of the main pulmonary artery to the diameter of the ascending aorta is suggestive of PH.
Other tests. Overnight oximetry and testing for sleep-disordered breathing, performed in an appropriate setting, can be considered.13,14,19
Pulmonary function testing with diffusion capacity for carbon monoxide, high-resolution chest CT, and a 6-minute walk test (6MWT) can be considered in patients who have risk factors for chronic lung disease. Pulmonary function testing, including measurement of the diffusing capacity of the lungs for carbon monoxide, arterial blood gas analysis, and CT, is used to aid in interpreting echocardiographic findings in patients with lung disease in whom PH is suspected.
Testing for comorbidities. A given patient’s predisposing conditions for PH might already be known; if not, laboratory evaluation for conditions such as sickle cell disease, liver disease, thyroid dysfunction, connective tissue disorders (antibody tests of antinuclear antibody, rheumatoid factor, anticentromere, anti-topoisomerase, anti-RNA polymerase III, anti-double stranded DNA, anti-Ro, anti-La, and anti-U1-RNP), and vasculitis (anti-neutrophil cytoplasmic autoantibodies) should be undertaken.
Analysis of stool and urine for Schistosoma spp parasites can be considered in an appropriate clinical setting.13
Right heart catheterization. Once alternative diagnoses are excluded, RHC is recommended to make a definitive diagnosis and assess the contribution of left heart disease. Vasoreactivity—defined as a reduction in mPAP ≥ 10 mm Hg to reach an absolute value of mPAP ≤ 40 mm Hg with increased or unchanged cardiac output—is assessed during RHC by administering nitric oxide or another vasodilator. This definition of vasoreactivity helps guide medical management in patients with PAH.7,20
Continue to: 6MWT
6MWT. Once the diagnosis of PH is made, a 6MWT helps establish baseline functional performance and will help you to monitor disease progression.
Who can benefit from screening for PH?
Annual evaluation of the risk of PAH is recommended for patients with systemic sclerosis or portal hypertension13 and can be considered in patients who have connective tissue disease with overlap features of systemic sclerosis.
Assessment for CTEPH or chronic thromboembolic pulmonary disease is recommended for patients with persistent or new-onset dyspnea or exercise limitation after pulmonary embolism.
Screening echocardiography for PH is recommended for patients who have been referred for liver transplantation.6
How risk is stratified
Risk stratification is used to manage PH and assess prognosis.
At diagnosis. Application of a 3-strata model of risk assessment (low, intermediate, high) is recommended.6 Pertinent data to determine risk include signs of right heart failure, progression of symptoms and clinical manifestations, report of syncope, WHO functional class, 6MWT, cardiopulmonary exercise testing, biomarkers (BNP or NT-proBNP), echocardiography, presence of pericardial effusion, and cardiac magnetic resonance imaging.
At follow-up. Use of a 4-strata model (low, intermediate–low, intermediate–high, and high risk) is recommended. Data used are WHO functional class, 6MWT, and results of either BNP or NT-proBNP testing.6
Continue to: When to refer
When to refer
Specialty consultation21-23 is recommended for:
- all patients with PAH
- PH patients in clinical Groups 2 and 3 whose disease is disproportionate to the extent of their left heart disease or hypoxic lung disease
- patients in whom there is concern about CTEPH and who therefore require early referral to a specialist for definitive treatment
- patients in whom the cause of PH is unclear or multifactorial (ie, clinical Group 5).
What are the options for managing PH?
Management of PH is based on the cause and classification of the individual patient’s disease.
Treatment for WSPH Group 1
Patients require referral to a specialty clinic for diagnosis, treatment, and monitoring of progression.10
First, regrettably, none of the medications approved by the US Food and Drug Administration for treating PAH prevent progression.7
Patients with idiopathic, hereditary, or drug-induced PAH with positive vasoreactivity are treated with a calcium channel blocker (CCB). The dosage is titrated to optimize therapy for the individual patient.
The patient is then reassessed after 3 to 6 months of medical therapy. Current treatment is continued if the following goals have been met:
- WHO functional classification is I or II
- BNP < 50 ng/L or NT-proBNP < 300 ng/L
- hemodynamics are normal or near-normal (mPAP ≤ 30 mm Hg and PVR ≤ 4 WU).
If these goals have not been met, treatment is adjusted by following the algorithm described below.
Continue to: The treatment algorithm...
The treatment algorithm for idiopathic-, heritable-, drug-induced, and connective tissue disease–associated PAH highlights the importance of cardiopulmonary comorbidities and risk strata at the time treatment is initiated and then during follow-up.
Cardiopulmonary comorbidities are conditions associated with an increased risk of left ventricular diastolic dysfunction, including obesity, hypertension, diabetes, and coronary artery disease. Pulmonary comorbidities can include signs of mild parenchymal lung disease and are often associated with a low carbon monoxide diffusing capacity (< 45% of predicted value).
The management algorithm proceeds as follows:
- For patients without cardiopulmonary comorbidities and who are at low or intermediate risk, treatment of PAH with an endothelin receptor antagonist (ERA) plus a phosphodiesterase-5 (PDE5) inhibitor is recommended.
- For patients without cardiopulmonary comorbidities and who are at high risk, treatment with an ERA, a PDE5 inhibitor, and either an IV or subcutaneous prostacyclin analogue (PCA) can be considered.
- Patients in either of the preceding 2 categories should have regular follow-up assessment; at such follow-up, their risk should be stratified based on 4 strata (see “How risk is stratified”):
- Low risk: Continue initial therapy.
- Low-to-intermediate risk: Consider adding a prostacyclin receptor agonist to the initial regimen or switch to a PDE5 inhibitor or a soluble guanylate cyclase stimulator.
- Intermediate-to-high or high risk: Consider adding a PCA (IV epoprostenol or IV or subcutaneous treprostinil). In addition, or alternatively, have the patient evaluated for lung transplantation.
- For patients with cardiopulmonary comorbidity—in any risk category—consider oral monotherapy with a PDE5 inhibitor or an ERA. Provide regular follow-up and individualize therapy.6
Treatment for WSPH Groups 2 and 3
Treatment is focused on the underlying cause of PH:
- Patients who have left heart disease with either severe pre-capillary component PH or markers of right ventricular dysfunction, or both, should be referred to a PH center.
- Patients with combined pre-capillary and postcapillary PH in whom pre-capillary PH is severe should be considered for an individualized approach.
- Consider prescribing the ERA bosentan in specific scenarios (eg, the Eisenmenger syndrome of left-right shunting resulting from a congenital cardiac defect) to improve exercise capacity. If PAH persists after corrected adult congenital heart disease, follow the PAH treatment algorithm for Group 1 patients (described earlier).
- For patients in Group 3, those who have severe PH should be referred to a PH center.
- Consider prescribing inhaled treprostinil in PH with interstitial lung disease.
Treatment for WSPH Group 4
Patients with CTEPH are the only ones for whom pulmonary endarterectomy (PEA), the treatment of choice, might be curative. Balloon angioplasty can be considered for inoperable cases6; these patients should be placed on lifelong anticoagulant therapy.
Symptomatic patients who have inoperable CTEPH or persistent recurrent PH after PEA are medically managed; the agent of choice is riociguat. Patients who have undergone PEA or balloon angioplasty and those receiving pharmacotherapy should be followed long term.
Treatment for WSPH Group 5
Management of these patients focuses on associated conditions.
Continue to: Which medications for PAH?
Which medications for PAH?
CCBs. Four options in this class have shown utility, notably in patients who have had a positive vasoreactivity test (see “How best to approach evaluation and diagnosis?”):
- Nifedipine is started at 10 mg tid; target dosage is 20 to 60 mg, bid or tid.
- Diltiazem is started at 60 mg bid; target dosage is 120 to 360 mg bid.
- Amlodipine is started at 5 mg/d; target dosage is 15 to 30 mg/d.
- Felodipine is started at 5 mg/d; target dosage is 15 to 30 mg/d.
Felodipine and amlodipine have longer half-lives than other CCBs and are well tolerated.
ERA. Used as vasodilators are ambrinsentan (starting dosage, 5 mg/d; target dosage, 10 mg/d), macitentan (starting and target dosage, 10 mg/d), and bosentan (starting dosage, 62.5 mg bid; target dosage, 125 mg bid).
Nitric oxide–cyclic guanosine monophosphate enhancers. These are the PDE5 inhibitors sildenafil (starting and target dosages, 20 mg tid) and tadalafil (starting dosage, 20 or 40 mg/d; target dosage, 40 mg/d), and the guanylate cyclase stimulant riociguat (starting dosage, 1 mg tid; target dosage, 2.5 mg tid). All 3 agents enhance production of the potent vasodilator nitric oxide, production of which is impaired in PH.
Prostanoids. Several options are available:
- Beraprost sodium. For this oral prostacyclin analogue, starting dosage is 20 μg tid; target dosage is the maximum tolerated dosage (as high as 40 μg tid).
- Extended-release beraprost. Starting dosage is 60 μg bid; target dosage is the maximum tolerated dosage (as high as 180 μg bid).
- Oral treprostinil. Starting dosage is 0.25 mg bid or 0.125 mg tid; target dosage is the maximum tolerated dosage.
- Inhaled iloprost. Starting dosage of this prostacyclin analogue is 2.5 μg, 6 to 9 times per day; target dosage is 5 μg, 6 to 9 times per day.
- Inhaled treprostinil. Starting dosage is 18 μg qid; target dosage is 54 to 72 μg qid.
- Eproprostenol is administered by continuous IV infusion, at a starting dosage of 2 ng/kg/min; target dosage is determined by tolerability and effectiveness (typically, 30 ng/kg/min).
- IV treprostinil. Starting dosage 1.25 ng/kg/min; target dosage is determined by tolerability and effectiveness, with a typical dosage of 60 ng/kg/min.
Combination treatment with the agents listed above is often utilized.
Selexipag. This oral selective nonprostainoid prostacyclin receptor agonist is started at 200 μg bid; target dosage is the maximum tolerated, as high as 1600 μg bid.
Continue to: Supportive therapy
Supportive therapy
The need for oxygen should be addressed in patients with hypoxia in any setting—resting, exercise induced, and nocturnal.24 Patients with an arterial blood oxygen pressure < 60 mm Hg (SaO2 < 90 mm Hg) should be on long-term oxygen therapy.6
Diuretics are beneficial in patients with chronic fluid retention from PH that is related to right ventricular failure.24
Pulmonary rehabilitation and exercise. Contrary to common belief that exercise training is contraindicated in patients with PH, exercise training has emerged in the past decade as an effective tool to improve exercise capacity, ventilatory efficiency, and quality of life. While a patient is training, oxygen saturation, measured by pulse oximetry, should be maintained at > 90% throughout the exercise session to avoid hypoxic pulmonary artery vasoconstriction.25
A patient who does not qualify for pulmonary or cardiac rehabilitation should be referred for physical therapy.24
Ongoing follow-up in primary care
Instruct patients not to abruptly discontinue medications that have been prescribed for PH. Ongoing follow-up and monitoring involves assessing right heart function, exercise tolerance, and resting and ambulatory oximetry. Testing for the level of BNP provides prognostic information and allows assessment of treatment response.15 The frequency of 6MWT, echocardiography, and RHC is decided on a case-by-case basis.
Other considerations
Pregnancy. PAH often affects patients of childbearing age. Because PAH-associated maternal mortality and the risk to the fetus during pregnancy are high, pregnancy is not recommended for patients with PAH. After a diagnosis of PAH in a patient of childbearing age, counseling should be offered at an expert center. Advice on effective contraception methods should be given early on.10,26-29
Surgery. Every patient with clinically significant PH is at increased risk of perioperative morbidity and death.30,31 Guidelines recommend that these patients avoid nonessential surgery; if surgery is necessary, care should be provided at a PH expert center.10
Continue to: Patients with severe PH...
Patients with severe PH should consider surgery for any indication carefully, discussing with the care team their risk and exploring nonsurgical options. Cardiothoracic surgical and liver transplantation services might have highly specific criteria for treating patients with PH, but other essential and nonessential surgeries require individualized risk stratification. Surgery for patients with severe PH and right ventricular dysfunction should be performed at a center equipped to handle high-risk patients.
Other preventive measures. Patients with PAH should6,10:
- remain current with immunization against influenza virus, SARS-CoV-2, and pneumococcal pneumonia
- avoid high altitudes
- use supplemental oxygen during air travel to keep arterial oxygen saturation > 91%.
Lung transplantation. Patients eligible for transplantation who (1) are at intermediate-to-high risk or high risk or (2) have a REVEAL (Registry to EValuate Early And Long-term pulmonary arterial hypertension disease management) risk score > 7, and who have had an inadequate response to oral combination therapy, should be referred for evaluation for lung transplantation. Placement on the list for lung transplantation is also recommended for patients at high risk of death and who have a REVEAL risk score ≥ 10 despite medical therapy, including a subcutaneous or IV prostacyclin analogue.6
PH in infants and children
The Pediatric Task Force of the 6th WSPH has applied the new definition proposed for adult PH (> 20 mm Hg mPAP) to children and infants > 3 months of age (see “Pulmonary hypertension in the pediatric population,” at left32-36).
SIDEBAR
Pulmonary hypertension in the pediatric population
The onset of pulmonary hypertension (PH) in children can occur at any age and be of quite different causes than in adults. In newborns, pulmonary pressure drops rapidly during the week after delivery; in some cases, however, pressures remain elevated (> 20 mm Hg) despite healthy lungs. These asymptomatic newborns require close monitoring.32
Etiology. Pediatric PH can be persistent or transient. Prominent causes of persistent or progressive PH in children are pulmonary arterial hypertension (PAH) associated with congenital heart disease and developmental lung disease, such as bronchopulmonary dysplasia and idiopathic PAH. Major categories of congenital heart disease that cause PH are shunting lesions and left heart disease associated with elevated atrial pressure. Other causes are rare.33
Persistent PH of the newborn (PPHN) and PH due to diaphragmatic hernia are common causes of transient PH.34 In PPHN, pulmonary vascular resistance remains abnormally high after birth, resulting in right-to-left shunting of the circulation that, in turn, leads to hypoxemia unresponsive to usual measures. In most cases, signs of respiratory distress and hypoxia are noted within the first 24 hours of life. The most common cause of PPHN is infection.35
Evaluation. The typical diagnostic work-up of suspected pediatric PH is similar to what is undertaken in the adult population—varying, however, according to the specific suspected cause. As in adults, right heart catheterization remains the gold standard of diagnosis, and should be conducted at a pediatric PH expert center. As with adult patients, infants and children with PH should be managed by a multidisciplinary expert team.
Management. PAH-targeted medications (see “What are the options for managing PH?”) are used to treat PAH in children.36
CORRESPONDENCE
Madhavi Singh, MD, 1850 East Park Ave., Suite 207, State College, PA 16803; [email protected]
New guidelines that redefine pulmonary hypertension (PH) by a lower mean pulmonary artery pressure (mPAP) have led to a reported increase in the number of patients given a diagnosis of PH. Although the evaluation and treatment of PH relies on the specialist, as we explain here, family physicians play a pivotal role in the diagnosis, reduction or elimination of risk factors for PH, and timely referral to a pulmonologist or cardiologist who has expertise in managing the disease. We also address the important finding that adult patients who have been evaluated, treated, and followed based on guidelines—updated just last year—have a longer life expectancy than patients who have not been treated properly or not treated at all.

Last, we summarize the etiology, evaluation, and management of PH in the pediatric population.
What is pulmonary hypertension? A revised definition
Prior to 2018, PH was defined as mPAP (measured by right heart catheterization [RHC]) ≥ 25 mm Hg at rest. Now, based on guidelines developed at the 6th World Symposium on Pulmonary Hypertension (WSPH) in 2018, PH is defined as mPAP > 20 mm Hg.1,2 That change was based on studies in which researchers noted higher mortality in adults who had mPAP below the traditional threshold.3,4 There is no evidence, however, of increased mortality in the pediatric population in this lower mPAP range.5
PH is estimated to be present in approximately 1% of the population.6 PH due to other diseases—eg, cardiac disease, lung disease, or a chronic thromboembolic condition—reflects the prevalence of the causative disease.7
How is pulmonary hypertension classified?
Based on the work of a Task Force of the 6th WSPH, PH is classified by underlying pathophysiology, hemodynamics, and functional status. Clinical classification comprises 5 categories, or “groups,” based on underlying pathophysiology (TABLE 16).
Clinical classification
Group 1 PH includes patients with primary pulmonary hypertension, also referred to (including in this article) as pulmonary arterial hypertension (PAH). Hemodynamic criteria that define PAH include pulmonary vascular resistance (PVR) > 2 Woods unitsa and pulmonary capillary wedge pressure > 15 mm Hg. Idiopathic PAH is the most common diagnosis in this group.
The incidence of PAH is approximately 6 cases for every 1 million adults; prevalence is 48 to 55 cases for every 1 million adults. PAH is more common in women.6
Continue to: Less common causes...
Less common causes in Group 1 includ
Group 2 PH comprises patients whose disease results from left heart dysfunction, the most common cause of PH. This subgroup has an elevated pulmonary artery wedge pressure > 15 mm Hg.8 Patients have either isolated postcapillary PH or combined pre-capillary and postcapillary PH.
Group 3 PH comprises patients whose PH is secondary to chronic and hypoxic lung disease. Patients in this group have pre-capillary PH; even a modest elevation in mPAP (20-29 mm Hg) is associated with a poor prognosis. Group 3 patients have elevated PVR, even with mild PH.2 Exertional dyspnea disproportionate to the results of pulmonary function testing, low carbon monoxide diffusion capacity, and rapid decline of arterial oxygenation with exercise all point to severe PH in these patients.9
Group 4 PH encompasses patients with pulmonary artery obstruction, the most common cause of which is related to chronic thromboembolism. Other causes include obstruction of the pulmonary artery from an extrinsic source. Patients with chronic thromboembolic pulmonary hypertension (CTEPH) also have pre-capillary PH, resulting from elevated pulmonary pressures secondary to thromboembolic burden, as well as pulmonary remodeling in unobstructed small arterioles.
Group 5 PH is a miscellaneous group secondary to unclear or multiple causes, including chronic hematologic anemia (eg, sickle cell disease), systemic disorders (eg, sarcoidosis), and metabolic disorders (eg, glycogen storage disease). Patients in Group 5 can have both pre-capillary and postcapillary hypertension.
Classification by functional status
The World Health Organization (WHO) Functional Classification of Patients with Pulmonary Hypertension is divided into 4 classes.10 This system is used to guide treatment and for prognostic purposes:
Class I. Patients have no limitation of physical activity. Ordinary physical activity does not cause undue dyspnea or fatigue, chest pain, or near-syncope.
Continue to: Class II
Class II. Patients have slight limitation of physical activity. They are comfortable at rest but daily physical activity causes dyspnea, fatigue, chest pain, or near-syncope.
Class III. These patients have marked limitation of physical activity. They are comfortable at rest, but less-than-ordinary activity causes dyspnea, fatigue, chest pain, or near-syncope.
Class IV. Patients are unable to carry out any physical activity without symptoms. They manifest signs of right heart failure. Dyspnea or fatigue, or both, might be present even at rest.
How is the pathophysiology of PH described?
The term pulmonary hypertension refers to an elevation in PAP that can result from any number of causes. Pulmonary arterial hypertension is a subcategory of PH in which a rise in PAP is due to primary pathology in the arteries proper.
As noted, PH results from a variety of pathophysiologic mechanisms, reflected in the classification in TABLE 1.6
WSPH Group 1 patients are considered to have PAH; for most, disease is idiopathic. In small-caliber pulmonary arteries, hypertrophy of smooth muscle, endothelial cells, and adventitia leads to increased resistance. Production of nitric oxide and prostacyclins is also impaired in endothelial cells. Genetic mutation, environmental factors such as exposure to stimulant use, and collagen vascular disease have a role in different subtypes of PAH. Portopulmonary hypertension is a subtype of PAH in patients with portal hypertension.
WSPH Groups 2-5. Increased PVR can result from pulmonary vascular congestion due to left heart dysfunction; destruction of the alveolar capillary bed; chronic hypoxic vasoconstriction; and vascular occlusion from thromboembolism.
Continue to: Once approximately...
Once approximately 30% of the pulmonary vasculature is involved, pressure in the pulmonary circulation starts to rise. In all WSPH groups, this increase in PVR results in increased right ventricular afterload that, over time, leads to right ventricular dysfunction.7,11,12
How does PH manifest?
Patients who have PH usually present with dyspnea, fatigue, chest pain, near-syncope, syncope, or lower-extremity edema, or any combination of these symptoms. The nonspecificity of presenting symptoms can lead to a delay in diagnosis.
In addition, suspicion of PH should be raised when a patient:
- presents with skin discoloration (light or dark) or a telangiectatic rash
- presents with difficulty swallowing
- has a history of connective tissue disease or hemolytic anemia
- has risk factors for HIV infection or liver disease
- takes an appetite suppressant
- has been exposed to other toxins known to increase the risk of PH.
A detailed medical history—looking for chronic lung or heart disease, thromboembolism, sleep-disordered breathing, a thyroid disorder, chronic renal failure, or a metabolic disorder—should be obtained.
Common findings on the physical exam in PH include:
- an increased P2 heart sound (pulmonic closure)
- high-pitched holosystolic murmur from tricuspid regurgitation
- pulmonic insufficiency murmur
- jugular venous distension
- hepatojugular reflux
- peripheral edema.
These findings are not specific to PH but, again, their presence warrants consideration of PH.
How best to approach evaluation and diagnosis?
The work-up for PH is broad; FIGURE 113,14 provides an outline of how to proceed when there is a concern for PH. For the work-up of symptoms and signs listed earlier, chest radiography and electrocardiography are recommended.

Continue to: Radiographic findings
Radiographic findings that suggest PH include enlargement of central pulmonary arteries and the right ventricle and dilation of the right atrium. Pulmonary vascular congestion might also be seen, secondary to left heart disease.7
Electrocardiographic findings of PH are demonstrated by signs of left ventricular hypertrophy, especially in Group 2 PH. Upright R waves in V1-V2 with deeper S waves in V5-V6 might represent right ventricular hypertrophy or right heart strain. Frequent premature atrial contractions and multifocal atrial tachycardia are also associated with PH.7

Brain natriuretic peptide (BNP) or N-terminal (NT) proBNP. The level of BNP might be elevated in PH, but its role in the diagnostic process has not been established. BNP can, however, be used to monitor treatment effectiveness and prognosis.15 A normal electrocardiogram in tandem with a normal level of BNP or NT-proBNP is associated with a low likelihood of PH.6

Transthoracic echocardiography (TTE) is the initial evaluation tool whenever PH is suspected. Echocardiographic findings suggestive of PH include a combination of tricuspid regurgitation velocity > 2.8 m/s (FIGURE 2); estimated pulmonary artery systolic pressure > 35 mm Hg in younger adults and > 40 mm Hg in older adults; right ventricular hypertrophy or strain; or a combination of these. Other TTE findings suggestive of PH are related to the ventricles, pulmonary artery, inferior vena cava, and right atrium (TABLE 26). The probability of PH based on TTE findings is categorized as low, intermediate, or high (see TABLE 26 and TABLE 316 for details).

Older guidelines, still used by some, rely on the estimated pulmonary artery systolic pressure (ePASP) reading on echocardiography.13,17 However, studies have reported poor correlation between ePASP readings and values obtained from RHC.18

TTE also provides findings of left heart disease, such as left ventricular systolic and diastolic dysfunction and left-sided valvular pathology. Patients with suspected PH in whom evidence of left heart disease on TTE is insufficient for making the diagnosis should receive further evaluation for their possible status in Groups 3-5 PH.
Ventilation–perfusion (VQ) scan. If CTEPH is suspected, a VQ scan should be performed. The scan is highly sensitive for CTEPH; a normal VQ scan excludes CTEPH. Computed tomography (CT) of the chest is not helpful for identifying chronic thromboembolism.13
Continue to: Coagulation assays
Coagulation assays. When CTEPH is suspected, coagulopathy can be assessed by measuring anticardiolipin antibodies, lupus anticoagulant, and anti-b-2-glycoprotein antibodies.13
Chest CT will show radiographic findings in greater detail. An enlarged pulmonary artery (diameter ≥ 29 mm) or a ratio ≥ 1 of the diameter of the main pulmonary artery to the diameter of the ascending aorta is suggestive of PH.
Other tests. Overnight oximetry and testing for sleep-disordered breathing, performed in an appropriate setting, can be considered.13,14,19
Pulmonary function testing with diffusion capacity for carbon monoxide, high-resolution chest CT, and a 6-minute walk test (6MWT) can be considered in patients who have risk factors for chronic lung disease. Pulmonary function testing, including measurement of the diffusing capacity of the lungs for carbon monoxide, arterial blood gas analysis, and CT, is used to aid in interpreting echocardiographic findings in patients with lung disease in whom PH is suspected.
Testing for comorbidities. A given patient’s predisposing conditions for PH might already be known; if not, laboratory evaluation for conditions such as sickle cell disease, liver disease, thyroid dysfunction, connective tissue disorders (antibody tests of antinuclear antibody, rheumatoid factor, anticentromere, anti-topoisomerase, anti-RNA polymerase III, anti-double stranded DNA, anti-Ro, anti-La, and anti-U1-RNP), and vasculitis (anti-neutrophil cytoplasmic autoantibodies) should be undertaken.
Analysis of stool and urine for Schistosoma spp parasites can be considered in an appropriate clinical setting.13
Right heart catheterization. Once alternative diagnoses are excluded, RHC is recommended to make a definitive diagnosis and assess the contribution of left heart disease. Vasoreactivity—defined as a reduction in mPAP ≥ 10 mm Hg to reach an absolute value of mPAP ≤ 40 mm Hg with increased or unchanged cardiac output—is assessed during RHC by administering nitric oxide or another vasodilator. This definition of vasoreactivity helps guide medical management in patients with PAH.7,20
Continue to: 6MWT
6MWT. Once the diagnosis of PH is made, a 6MWT helps establish baseline functional performance and will help you to monitor disease progression.
Who can benefit from screening for PH?
Annual evaluation of the risk of PAH is recommended for patients with systemic sclerosis or portal hypertension13 and can be considered in patients who have connective tissue disease with overlap features of systemic sclerosis.
Assessment for CTEPH or chronic thromboembolic pulmonary disease is recommended for patients with persistent or new-onset dyspnea or exercise limitation after pulmonary embolism.
Screening echocardiography for PH is recommended for patients who have been referred for liver transplantation.6
How risk is stratified
Risk stratification is used to manage PH and assess prognosis.
At diagnosis. Application of a 3-strata model of risk assessment (low, intermediate, high) is recommended.6 Pertinent data to determine risk include signs of right heart failure, progression of symptoms and clinical manifestations, report of syncope, WHO functional class, 6MWT, cardiopulmonary exercise testing, biomarkers (BNP or NT-proBNP), echocardiography, presence of pericardial effusion, and cardiac magnetic resonance imaging.
At follow-up. Use of a 4-strata model (low, intermediate–low, intermediate–high, and high risk) is recommended. Data used are WHO functional class, 6MWT, and results of either BNP or NT-proBNP testing.6
Continue to: When to refer
When to refer
Specialty consultation21-23 is recommended for:
- all patients with PAH
- PH patients in clinical Groups 2 and 3 whose disease is disproportionate to the extent of their left heart disease or hypoxic lung disease
- patients in whom there is concern about CTEPH and who therefore require early referral to a specialist for definitive treatment
- patients in whom the cause of PH is unclear or multifactorial (ie, clinical Group 5).
What are the options for managing PH?
Management of PH is based on the cause and classification of the individual patient’s disease.
Treatment for WSPH Group 1
Patients require referral to a specialty clinic for diagnosis, treatment, and monitoring of progression.10
First, regrettably, none of the medications approved by the US Food and Drug Administration for treating PAH prevent progression.7
Patients with idiopathic, hereditary, or drug-induced PAH with positive vasoreactivity are treated with a calcium channel blocker (CCB). The dosage is titrated to optimize therapy for the individual patient.
The patient is then reassessed after 3 to 6 months of medical therapy. Current treatment is continued if the following goals have been met:
- WHO functional classification is I or II
- BNP < 50 ng/L or NT-proBNP < 300 ng/L
- hemodynamics are normal or near-normal (mPAP ≤ 30 mm Hg and PVR ≤ 4 WU).
If these goals have not been met, treatment is adjusted by following the algorithm described below.
Continue to: The treatment algorithm...
The treatment algorithm for idiopathic-, heritable-, drug-induced, and connective tissue disease–associated PAH highlights the importance of cardiopulmonary comorbidities and risk strata at the time treatment is initiated and then during follow-up.
Cardiopulmonary comorbidities are conditions associated with an increased risk of left ventricular diastolic dysfunction, including obesity, hypertension, diabetes, and coronary artery disease. Pulmonary comorbidities can include signs of mild parenchymal lung disease and are often associated with a low carbon monoxide diffusing capacity (< 45% of predicted value).
The management algorithm proceeds as follows:
- For patients without cardiopulmonary comorbidities and who are at low or intermediate risk, treatment of PAH with an endothelin receptor antagonist (ERA) plus a phosphodiesterase-5 (PDE5) inhibitor is recommended.
- For patients without cardiopulmonary comorbidities and who are at high risk, treatment with an ERA, a PDE5 inhibitor, and either an IV or subcutaneous prostacyclin analogue (PCA) can be considered.
- Patients in either of the preceding 2 categories should have regular follow-up assessment; at such follow-up, their risk should be stratified based on 4 strata (see “How risk is stratified”):
- Low risk: Continue initial therapy.
- Low-to-intermediate risk: Consider adding a prostacyclin receptor agonist to the initial regimen or switch to a PDE5 inhibitor or a soluble guanylate cyclase stimulator.
- Intermediate-to-high or high risk: Consider adding a PCA (IV epoprostenol or IV or subcutaneous treprostinil). In addition, or alternatively, have the patient evaluated for lung transplantation.
- For patients with cardiopulmonary comorbidity—in any risk category—consider oral monotherapy with a PDE5 inhibitor or an ERA. Provide regular follow-up and individualize therapy.6
Treatment for WSPH Groups 2 and 3
Treatment is focused on the underlying cause of PH:
- Patients who have left heart disease with either severe pre-capillary component PH or markers of right ventricular dysfunction, or both, should be referred to a PH center.
- Patients with combined pre-capillary and postcapillary PH in whom pre-capillary PH is severe should be considered for an individualized approach.
- Consider prescribing the ERA bosentan in specific scenarios (eg, the Eisenmenger syndrome of left-right shunting resulting from a congenital cardiac defect) to improve exercise capacity. If PAH persists after corrected adult congenital heart disease, follow the PAH treatment algorithm for Group 1 patients (described earlier).
- For patients in Group 3, those who have severe PH should be referred to a PH center.
- Consider prescribing inhaled treprostinil in PH with interstitial lung disease.
Treatment for WSPH Group 4
Patients with CTEPH are the only ones for whom pulmonary endarterectomy (PEA), the treatment of choice, might be curative. Balloon angioplasty can be considered for inoperable cases6; these patients should be placed on lifelong anticoagulant therapy.
Symptomatic patients who have inoperable CTEPH or persistent recurrent PH after PEA are medically managed; the agent of choice is riociguat. Patients who have undergone PEA or balloon angioplasty and those receiving pharmacotherapy should be followed long term.
Treatment for WSPH Group 5
Management of these patients focuses on associated conditions.
Continue to: Which medications for PAH?
Which medications for PAH?
CCBs. Four options in this class have shown utility, notably in patients who have had a positive vasoreactivity test (see “How best to approach evaluation and diagnosis?”):
- Nifedipine is started at 10 mg tid; target dosage is 20 to 60 mg, bid or tid.
- Diltiazem is started at 60 mg bid; target dosage is 120 to 360 mg bid.
- Amlodipine is started at 5 mg/d; target dosage is 15 to 30 mg/d.
- Felodipine is started at 5 mg/d; target dosage is 15 to 30 mg/d.
Felodipine and amlodipine have longer half-lives than other CCBs and are well tolerated.
ERA. Used as vasodilators are ambrinsentan (starting dosage, 5 mg/d; target dosage, 10 mg/d), macitentan (starting and target dosage, 10 mg/d), and bosentan (starting dosage, 62.5 mg bid; target dosage, 125 mg bid).
Nitric oxide–cyclic guanosine monophosphate enhancers. These are the PDE5 inhibitors sildenafil (starting and target dosages, 20 mg tid) and tadalafil (starting dosage, 20 or 40 mg/d; target dosage, 40 mg/d), and the guanylate cyclase stimulant riociguat (starting dosage, 1 mg tid; target dosage, 2.5 mg tid). All 3 agents enhance production of the potent vasodilator nitric oxide, production of which is impaired in PH.
Prostanoids. Several options are available:
- Beraprost sodium. For this oral prostacyclin analogue, starting dosage is 20 μg tid; target dosage is the maximum tolerated dosage (as high as 40 μg tid).
- Extended-release beraprost. Starting dosage is 60 μg bid; target dosage is the maximum tolerated dosage (as high as 180 μg bid).
- Oral treprostinil. Starting dosage is 0.25 mg bid or 0.125 mg tid; target dosage is the maximum tolerated dosage.
- Inhaled iloprost. Starting dosage of this prostacyclin analogue is 2.5 μg, 6 to 9 times per day; target dosage is 5 μg, 6 to 9 times per day.
- Inhaled treprostinil. Starting dosage is 18 μg qid; target dosage is 54 to 72 μg qid.
- Eproprostenol is administered by continuous IV infusion, at a starting dosage of 2 ng/kg/min; target dosage is determined by tolerability and effectiveness (typically, 30 ng/kg/min).
- IV treprostinil. Starting dosage 1.25 ng/kg/min; target dosage is determined by tolerability and effectiveness, with a typical dosage of 60 ng/kg/min.
Combination treatment with the agents listed above is often utilized.
Selexipag. This oral selective nonprostainoid prostacyclin receptor agonist is started at 200 μg bid; target dosage is the maximum tolerated, as high as 1600 μg bid.
Continue to: Supportive therapy
Supportive therapy
The need for oxygen should be addressed in patients with hypoxia in any setting—resting, exercise induced, and nocturnal.24 Patients with an arterial blood oxygen pressure < 60 mm Hg (SaO2 < 90 mm Hg) should be on long-term oxygen therapy.6
Diuretics are beneficial in patients with chronic fluid retention from PH that is related to right ventricular failure.24
Pulmonary rehabilitation and exercise. Contrary to common belief that exercise training is contraindicated in patients with PH, exercise training has emerged in the past decade as an effective tool to improve exercise capacity, ventilatory efficiency, and quality of life. While a patient is training, oxygen saturation, measured by pulse oximetry, should be maintained at > 90% throughout the exercise session to avoid hypoxic pulmonary artery vasoconstriction.25
A patient who does not qualify for pulmonary or cardiac rehabilitation should be referred for physical therapy.24
Ongoing follow-up in primary care
Instruct patients not to abruptly discontinue medications that have been prescribed for PH. Ongoing follow-up and monitoring involves assessing right heart function, exercise tolerance, and resting and ambulatory oximetry. Testing for the level of BNP provides prognostic information and allows assessment of treatment response.15 The frequency of 6MWT, echocardiography, and RHC is decided on a case-by-case basis.
Other considerations
Pregnancy. PAH often affects patients of childbearing age. Because PAH-associated maternal mortality and the risk to the fetus during pregnancy are high, pregnancy is not recommended for patients with PAH. After a diagnosis of PAH in a patient of childbearing age, counseling should be offered at an expert center. Advice on effective contraception methods should be given early on.10,26-29
Surgery. Every patient with clinically significant PH is at increased risk of perioperative morbidity and death.30,31 Guidelines recommend that these patients avoid nonessential surgery; if surgery is necessary, care should be provided at a PH expert center.10
Continue to: Patients with severe PH...
Patients with severe PH should consider surgery for any indication carefully, discussing with the care team their risk and exploring nonsurgical options. Cardiothoracic surgical and liver transplantation services might have highly specific criteria for treating patients with PH, but other essential and nonessential surgeries require individualized risk stratification. Surgery for patients with severe PH and right ventricular dysfunction should be performed at a center equipped to handle high-risk patients.
Other preventive measures. Patients with PAH should6,10:
- remain current with immunization against influenza virus, SARS-CoV-2, and pneumococcal pneumonia
- avoid high altitudes
- use supplemental oxygen during air travel to keep arterial oxygen saturation > 91%.
Lung transplantation. Patients eligible for transplantation who (1) are at intermediate-to-high risk or high risk or (2) have a REVEAL (Registry to EValuate Early And Long-term pulmonary arterial hypertension disease management) risk score > 7, and who have had an inadequate response to oral combination therapy, should be referred for evaluation for lung transplantation. Placement on the list for lung transplantation is also recommended for patients at high risk of death and who have a REVEAL risk score ≥ 10 despite medical therapy, including a subcutaneous or IV prostacyclin analogue.6
PH in infants and children
The Pediatric Task Force of the 6th WSPH has applied the new definition proposed for adult PH (> 20 mm Hg mPAP) to children and infants > 3 months of age (see “Pulmonary hypertension in the pediatric population,” at left32-36).
SIDEBAR
Pulmonary hypertension in the pediatric population
The onset of pulmonary hypertension (PH) in children can occur at any age and be of quite different causes than in adults. In newborns, pulmonary pressure drops rapidly during the week after delivery; in some cases, however, pressures remain elevated (> 20 mm Hg) despite healthy lungs. These asymptomatic newborns require close monitoring.32
Etiology. Pediatric PH can be persistent or transient. Prominent causes of persistent or progressive PH in children are pulmonary arterial hypertension (PAH) associated with congenital heart disease and developmental lung disease, such as bronchopulmonary dysplasia and idiopathic PAH. Major categories of congenital heart disease that cause PH are shunting lesions and left heart disease associated with elevated atrial pressure. Other causes are rare.33
Persistent PH of the newborn (PPHN) and PH due to diaphragmatic hernia are common causes of transient PH.34 In PPHN, pulmonary vascular resistance remains abnormally high after birth, resulting in right-to-left shunting of the circulation that, in turn, leads to hypoxemia unresponsive to usual measures. In most cases, signs of respiratory distress and hypoxia are noted within the first 24 hours of life. The most common cause of PPHN is infection.35
Evaluation. The typical diagnostic work-up of suspected pediatric PH is similar to what is undertaken in the adult population—varying, however, according to the specific suspected cause. As in adults, right heart catheterization remains the gold standard of diagnosis, and should be conducted at a pediatric PH expert center. As with adult patients, infants and children with PH should be managed by a multidisciplinary expert team.
Management. PAH-targeted medications (see “What are the options for managing PH?”) are used to treat PAH in children.36
CORRESPONDENCE
Madhavi Singh, MD, 1850 East Park Ave., Suite 207, State College, PA 16803; [email protected]
1. Galiè N, McLaughlin VV, Rubin LJ, et al. An overview of the 6th World Symposium on Pulmonary Hypertension. Eur Respir J. 2019;53:1802148. doi: 10.1183/13993003.02148-2018
2. Simonneau G, Montani D, Celermajer DS, et al. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J. 2019;53:1801913. doi: 10.1183/13993003.01913-2018
3. Kolte D, Lakshmanan S, Jankowich MD, et al. Mild pulmonary hypertension is associated with increased mortality: a systematic review and meta-analysis. J Am Heart Assoc. 2018;7:e009729. doi: 10.1161/JAHA.118.009729
4. Douschan P, Kovacs G, Avian A, et al. Mild elevation of pulmonary arterial pressure as a predictor of mortality. Am J Respir Crit Care Med. 2018;197:509-516. doi: 10.1164/rccm.201706-1215OC
5. Lammers AE, Apitz C. Update from the World Symposium on Pulmonary Hypertension 2018: does the new hemodynamic definition of pediatric pulmonary hypertension have an impact on treatment strategies? Cardiovasc Diagn Ther. 2021;11:1048-1051. doi: 10.21037/cdt-20-412
6. Humbert M, Kovacs G, Hoeper MM, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2022;43:3618-3731. doi: 10.1093/eurheartj/ehac237
7. Oldroyd SH, Manek G, Bhardwaj A. Pulmonary hypertension. In: StatPearls [Internet]. StatPearls Publishing. Updated July 20, 2022. Accessed November 27, 2022. www.ncbi.nlm.nih.gov/books/NBK482463/?report=classic
8. Vachiéry JL, Tedford RJ, Rosenkranz S, et al. Pulmonary hypertension due to left heart disease. Eur Respir J. 2019;53:1801897. doi: 10.1183/13993003.01897-2018
9. Seeger W, Adir Y, Barberà JA, et al. Pulmonary hypertension in chronic lung diseases. J Am Coll Cardiol. 2013;62(25 suppl):D109-D116. doi: 10.1016/j.jacc.2013.10.036
10. Taichman DB, Ornelas J, Chung L, et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST guideline and expert panel report. Chest. 2014;146:449-475. doi: 10.1378/chest.14-0793
11. Krowl L, Anjum F, Kaul P. Pulmonary idiopathic hypertension. In: StatPearls [Internet]. StatPearls Publishing. Updated August 8, 2022. Accessed November 27, 2022. www.ncbi.nlm.nih.gov/books/NBK519041/#_NBK519041_pubdet_
12. Bartolome SD. Portopulmonary hypertension: diagnosis, clinical features, and medical therapy. Clin Liver Dis (Hoboken). 2014;4:42-45. doi: 10.1002/cld.401
13. Frost A, Badesch D, Gibbs JSR, et al. Diagnosis of pulmonary hypertension. Eur Respir J. 2019;53:1801904. doi: 10.1183/ 13993003.01904-2018
14. Yaghi S, Novikov A, Trandafirescu T. Clinical update on pulmonary hypertension. J Investig Med. 2020;68:821-827. doi: 10.1136/jim-2020-001291
15. Chin KM, Rubin LJ, Channick R, et al. Association of N-terminal pro brain natriuretic peptide and long-term outcome in patients with pulmonary arterial hypertension. Circulation. 2019;139:2440-2450. doi: 10.1161/CIRCULATIONAHA.118.039360
16. Galiè N, Humbert M, Vachiery J-L, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J. 2015;46:903-975. doi: 10.1183/13993003.01032-2015
17. N,, , et al; Task Force for Diagnosis and Treatment of Pulmonary Hypertension of European Society of Cardiology (ESC); European Respiratory Society (ERS); International Society of Heart and Lung Transplantation (ISHLT). Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Respir J. 2009;34:1219-1263. doi: 10.1183/09031936.00139009
18. Rich JD, Shah SJ, Swamy RS, et al. Inaccuracy of Doppler echocardiographic estimates of pulmonary artery pressures in patients with pulmonary hypertension: implications for clinical practice. Chest. 2011;139:988-993. doi: 10.1378/chest.10-1269
19. Janda S, Shahidi N, Gin K, et al. Diagnostic accuracy of echocardiography for pulmonary hypertension: a systematic review and meta-analysis. Heart. 2011;97:612-622. doi: 10.1136/hrt.2010.212084
20. Farber HW, Foreman AJ, Miller DP, et al. REVEAL Registry: correlation of right heart catheterization and echocardiography in patients with pulmonary arterial hypertension. Congest Heart Fail. 2011;17:56-63. doi: 10.1111/j.1751-7133.2010.00202.x
21. Suntharalingam J, Ross RM, Easaw J, et al. Who should be referred to a specialist pulmonary hypertension centre—a referrer’s guide. Clin Med (Lond). 2016;16:135-141. doi: 10.7861/clinmedicine.16-2-135
22. Deaño RC, Glassner-Kolmin C, Rubenfire M, et al. Referral of patients with pulmonary hypertension diagnoses to tertiary pulmonary hypertension centers: the multicenter RePHerral Study. JAMA Intern Med. 2013;173:887-893. doi: 10.1001/jamainternmed.2013.319
23. Guidelines for referring patients with pulmonary hypertension. Royal Papworth Hospital, NHS Foundation Trust. Updated February 2019. Accessed November 27, 2022. https://royalpapworth.nhs.uk/application/files/9015/5014/6935/PVDU-Referral-guidelines-2019.pdf
24. Yuan P, Yuan X-T, Sun X-Y, et al. Exercise training for pulmonary hypertension: a systematic review and meta-analysis. Int J Cardiol. 2015;178:142-146. doi: 10.1016/j.ijcard.2014.10.161
25. Spruit MA, Singh SJ, Garvey C, et al; . An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188:e13-e64. doi: 10.1164/rccm.201309-1634ST
26. Olsson KM, Channick R. Pregnancy in pulmonary arterial hypertension. Eur Respir Rev. 2016;25:431-437. doi: 10.1183/ 16000617.0079-2016
27. Weiss BM, Zemp L, Swifert B, et al. Outcome of pulmonary vascular disease in pregnancy: a systematic overview from 1978 through 1996; J Am Coll Cardiol. 1998;31:1650-1657. doi: 10.1016/s0735-1097(98)00162-4
28. Qiangqiang Li, Dimopoulos K, Liu T, et al, Peripartum outcomes in a large population of women with pulmonary arterial hypertension associated with congenital heart disease, Euro J Prev Cardiol. 2019;26:1067-1076. doi: 10.1177/2047487318821246
29. Olsson KM, Jaïs X. Birth control and pregnancy management in pulmonary hypertension. Semin Respir Crit Care Med. 2013;34:681-688. doi: 10.1055/s-0033-1355438
30. Price LC, Montani D, Jaïs X, et al. Noncardiothoracic nonobstetric surgery in mild-to-moderate pulmonary hypertension. Eur Respir J. 2010;35:1294-1302. doi: 10.1183/09031936.00113009
31. Memtsoudis SG, Ma Y, Chiu YL, et al. Perioperative mortality in patients with pulmonary hypertension undergoing major joint replacement. Anesth Analg. 2010;111:1110-1116. doi: 10.1213/ANE.0b013e3181f43149
32. Rosenzweig EB, Abman SH, Adatia I, et al. Paediatric pulmonary arterial hypertension: updates on definition, classification, diagnostics and management. Eur Respir J. 2019;53:1801916. doi: 10.1183/13993003.01916-2018
33. Berger RMF, Beghetti M, Humpl T, et al. Clinical features of paediatric pulmonary hypertension: a registry study. Lancet. 2012;379:537-546. doi: 10.1016/S0140-6736(11)61621-8
34. van Loon RL, Roofthooft MTR, Hillege HL, et al. Pediatric pulmonary hypertension in the Netherlands: epidemiology and characterization during the period 1991 to 2005. Circulation. 2011;124:1755-1764. doi: 10.1161/CIRCULATIONAHA.110.969584
35. Steurer MA, Jelliffe-Pawlowski LL, Baer RJ, et al. Persistent pulmonary hypertension of the newborn in late preterm and term infants in California. Pediatrics. 2017;139:e20161165. doi: 10.1542/peds.2016-1165
36. Hansmann G, Koestenberger M, Alastalo TP, et al. 2019 updated consensus statement on the diagnosis and treatment of pediatric pulmonary hypertension: the European Pediatric Pulmonary Vascular Disease Network (EPPVDN), endorsed by AEPC, ESPR and ISHLT. J Heart Lung Transplant. 2019;38:879-901. doi: 10.1016/j.healun.2019.06.022
1. Galiè N, McLaughlin VV, Rubin LJ, et al. An overview of the 6th World Symposium on Pulmonary Hypertension. Eur Respir J. 2019;53:1802148. doi: 10.1183/13993003.02148-2018
2. Simonneau G, Montani D, Celermajer DS, et al. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J. 2019;53:1801913. doi: 10.1183/13993003.01913-2018
3. Kolte D, Lakshmanan S, Jankowich MD, et al. Mild pulmonary hypertension is associated with increased mortality: a systematic review and meta-analysis. J Am Heart Assoc. 2018;7:e009729. doi: 10.1161/JAHA.118.009729
4. Douschan P, Kovacs G, Avian A, et al. Mild elevation of pulmonary arterial pressure as a predictor of mortality. Am J Respir Crit Care Med. 2018;197:509-516. doi: 10.1164/rccm.201706-1215OC
5. Lammers AE, Apitz C. Update from the World Symposium on Pulmonary Hypertension 2018: does the new hemodynamic definition of pediatric pulmonary hypertension have an impact on treatment strategies? Cardiovasc Diagn Ther. 2021;11:1048-1051. doi: 10.21037/cdt-20-412
6. Humbert M, Kovacs G, Hoeper MM, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2022;43:3618-3731. doi: 10.1093/eurheartj/ehac237
7. Oldroyd SH, Manek G, Bhardwaj A. Pulmonary hypertension. In: StatPearls [Internet]. StatPearls Publishing. Updated July 20, 2022. Accessed November 27, 2022. www.ncbi.nlm.nih.gov/books/NBK482463/?report=classic
8. Vachiéry JL, Tedford RJ, Rosenkranz S, et al. Pulmonary hypertension due to left heart disease. Eur Respir J. 2019;53:1801897. doi: 10.1183/13993003.01897-2018
9. Seeger W, Adir Y, Barberà JA, et al. Pulmonary hypertension in chronic lung diseases. J Am Coll Cardiol. 2013;62(25 suppl):D109-D116. doi: 10.1016/j.jacc.2013.10.036
10. Taichman DB, Ornelas J, Chung L, et al. Pharmacologic therapy for pulmonary arterial hypertension in adults: CHEST guideline and expert panel report. Chest. 2014;146:449-475. doi: 10.1378/chest.14-0793
11. Krowl L, Anjum F, Kaul P. Pulmonary idiopathic hypertension. In: StatPearls [Internet]. StatPearls Publishing. Updated August 8, 2022. Accessed November 27, 2022. www.ncbi.nlm.nih.gov/books/NBK519041/#_NBK519041_pubdet_
12. Bartolome SD. Portopulmonary hypertension: diagnosis, clinical features, and medical therapy. Clin Liver Dis (Hoboken). 2014;4:42-45. doi: 10.1002/cld.401
13. Frost A, Badesch D, Gibbs JSR, et al. Diagnosis of pulmonary hypertension. Eur Respir J. 2019;53:1801904. doi: 10.1183/ 13993003.01904-2018
14. Yaghi S, Novikov A, Trandafirescu T. Clinical update on pulmonary hypertension. J Investig Med. 2020;68:821-827. doi: 10.1136/jim-2020-001291
15. Chin KM, Rubin LJ, Channick R, et al. Association of N-terminal pro brain natriuretic peptide and long-term outcome in patients with pulmonary arterial hypertension. Circulation. 2019;139:2440-2450. doi: 10.1161/CIRCULATIONAHA.118.039360
16. Galiè N, Humbert M, Vachiery J-L, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J. 2015;46:903-975. doi: 10.1183/13993003.01032-2015
17. N,, , et al; Task Force for Diagnosis and Treatment of Pulmonary Hypertension of European Society of Cardiology (ESC); European Respiratory Society (ERS); International Society of Heart and Lung Transplantation (ISHLT). Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Respir J. 2009;34:1219-1263. doi: 10.1183/09031936.00139009
18. Rich JD, Shah SJ, Swamy RS, et al. Inaccuracy of Doppler echocardiographic estimates of pulmonary artery pressures in patients with pulmonary hypertension: implications for clinical practice. Chest. 2011;139:988-993. doi: 10.1378/chest.10-1269
19. Janda S, Shahidi N, Gin K, et al. Diagnostic accuracy of echocardiography for pulmonary hypertension: a systematic review and meta-analysis. Heart. 2011;97:612-622. doi: 10.1136/hrt.2010.212084
20. Farber HW, Foreman AJ, Miller DP, et al. REVEAL Registry: correlation of right heart catheterization and echocardiography in patients with pulmonary arterial hypertension. Congest Heart Fail. 2011;17:56-63. doi: 10.1111/j.1751-7133.2010.00202.x
21. Suntharalingam J, Ross RM, Easaw J, et al. Who should be referred to a specialist pulmonary hypertension centre—a referrer’s guide. Clin Med (Lond). 2016;16:135-141. doi: 10.7861/clinmedicine.16-2-135
22. Deaño RC, Glassner-Kolmin C, Rubenfire M, et al. Referral of patients with pulmonary hypertension diagnoses to tertiary pulmonary hypertension centers: the multicenter RePHerral Study. JAMA Intern Med. 2013;173:887-893. doi: 10.1001/jamainternmed.2013.319
23. Guidelines for referring patients with pulmonary hypertension. Royal Papworth Hospital, NHS Foundation Trust. Updated February 2019. Accessed November 27, 2022. https://royalpapworth.nhs.uk/application/files/9015/5014/6935/PVDU-Referral-guidelines-2019.pdf
24. Yuan P, Yuan X-T, Sun X-Y, et al. Exercise training for pulmonary hypertension: a systematic review and meta-analysis. Int J Cardiol. 2015;178:142-146. doi: 10.1016/j.ijcard.2014.10.161
25. Spruit MA, Singh SJ, Garvey C, et al; . An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013;188:e13-e64. doi: 10.1164/rccm.201309-1634ST
26. Olsson KM, Channick R. Pregnancy in pulmonary arterial hypertension. Eur Respir Rev. 2016;25:431-437. doi: 10.1183/ 16000617.0079-2016
27. Weiss BM, Zemp L, Swifert B, et al. Outcome of pulmonary vascular disease in pregnancy: a systematic overview from 1978 through 1996; J Am Coll Cardiol. 1998;31:1650-1657. doi: 10.1016/s0735-1097(98)00162-4
28. Qiangqiang Li, Dimopoulos K, Liu T, et al, Peripartum outcomes in a large population of women with pulmonary arterial hypertension associated with congenital heart disease, Euro J Prev Cardiol. 2019;26:1067-1076. doi: 10.1177/2047487318821246
29. Olsson KM, Jaïs X. Birth control and pregnancy management in pulmonary hypertension. Semin Respir Crit Care Med. 2013;34:681-688. doi: 10.1055/s-0033-1355438
30. Price LC, Montani D, Jaïs X, et al. Noncardiothoracic nonobstetric surgery in mild-to-moderate pulmonary hypertension. Eur Respir J. 2010;35:1294-1302. doi: 10.1183/09031936.00113009
31. Memtsoudis SG, Ma Y, Chiu YL, et al. Perioperative mortality in patients with pulmonary hypertension undergoing major joint replacement. Anesth Analg. 2010;111:1110-1116. doi: 10.1213/ANE.0b013e3181f43149
32. Rosenzweig EB, Abman SH, Adatia I, et al. Paediatric pulmonary arterial hypertension: updates on definition, classification, diagnostics and management. Eur Respir J. 2019;53:1801916. doi: 10.1183/13993003.01916-2018
33. Berger RMF, Beghetti M, Humpl T, et al. Clinical features of paediatric pulmonary hypertension: a registry study. Lancet. 2012;379:537-546. doi: 10.1016/S0140-6736(11)61621-8
34. van Loon RL, Roofthooft MTR, Hillege HL, et al. Pediatric pulmonary hypertension in the Netherlands: epidemiology and characterization during the period 1991 to 2005. Circulation. 2011;124:1755-1764. doi: 10.1161/CIRCULATIONAHA.110.969584
35. Steurer MA, Jelliffe-Pawlowski LL, Baer RJ, et al. Persistent pulmonary hypertension of the newborn in late preterm and term infants in California. Pediatrics. 2017;139:e20161165. doi: 10.1542/peds.2016-1165
36. Hansmann G, Koestenberger M, Alastalo TP, et al. 2019 updated consensus statement on the diagnosis and treatment of pediatric pulmonary hypertension: the European Pediatric Pulmonary Vascular Disease Network (EPPVDN), endorsed by AEPC, ESPR and ISHLT. J Heart Lung Transplant. 2019;38:879-901. doi: 10.1016/j.healun.2019.06.022
PRACTICE RECOMMENDATIONS
› Employ echocardiography as the first-line diagnostic test when pulmonary hypertension (PH) is suspected. C
› Order a ventilation– perfusion scan in patients with unexplained PH to exclude chronic thromboembolic PH. C
› Order lung function testing with diffusion capacity for carbon monoxide as part of the initial evaluation of PH. C
› Use right heart catheterization to confirm the diagnosis of pulmonary arterial hypertension. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Call it preclinical or subclinical, ILD in RA needs to be tracked
More clinical guidance is needed for monitoring interstitial lung disease (ILD) in patients with rheumatoid arthritis, according to a new commentary.
Though ILD is a leading cause of death among patients with RA, these patients are not routinely screened for ILD, the authors say, and there are currently no guidelines on how to monitor ILD progression in patients with RA.
“ILD associated with rheumatoid arthritis is a disease for which there’s been very little research done, so it’s an area of rheumatology where there are many unknowns,” lead author Elizabeth R. Volkmann, MD, who codirects the connective tissue disease–related interstitial lung disease (CTD-ILD) program at University of California, Los Angeles, told this news organization.
The commentary was published in The Lancet Rheumatology.
Defining disease
One of the major unknowns is how to define the disease, she said. RA patients sometimes undergo imaging for other medical reasons, and interstitial lung abnormalities are incidentally detected. These patients can be classified as having “preclinical” or “subclinical” ILD, as they do not yet have symptoms; however, there is no consensus as to what these terms mean, the commentary authors write. “The other problem that we have with these terms is that it sometimes creates the perception that this is a nonworrisome feature of rheumatoid arthritis,” Dr. Volkmann said, although the condition should be followed closely.
“We know we can detect imaging features of ILD in people who may not yet have symptoms, and we need to know when to define a clinically important informality that requires follow-up or treatment,” added John M. Davis III, MD, a rheumatologist at the Mayo Clinic, Rochester, Minn. He was not involved with the work.
Dr. Volkmann proposed eliminating the prefixes “pre” and “sub” when referring to ILD. “In other connective tissue diseases, like systemic sclerosis, for example, we can use the term ‘limited’ or ‘extensive’ ILD, based on the extent of involvement of the ILD on high-resolution computed tomography (HRCT) imaging,” she said. “This could potentially be something that is applied to how we classify patients with RA-ILD.”
Tracking ILD progression
Once ILD is identified, monitoring its progression poses challenges, as respiratory symptoms may be difficult to detect. RA patients may already be avoiding exercise because of joint pain, so they may not notice shortness of breath during physical activity, noted Jessica K. Gordon, MD, of the Hospital for Special Surgery, New York, in an interview with this news organization. She was not involved with the commentary. Cough is a potential symptom of ILD, but cough can also be the result of allergies, postnasal drip, or reflux, she said. Making the distinction between “preclinical” and symptomatic disease can be “complicated,” she added; “you may have to really dig.”
Additionally, there has been little research on the outcomes of patients with preclinical or subclinical ILD and clinical ILD, the commentary authors write. “It is therefore conceivable that some patients with rheumatoid arthritis diagnosed with preclinical or subclinical ILD could potentially have worse outcomes if both the rheumatoid arthritis and ILD are not monitored closely,” they note.
To better track RA-associated ILD for patients with and those without symptoms, the authors advocate for monitoring patients using pulmonary testing and CT scanning, as well as evaluating symptoms. How often these assessments should be conducted depends on the individual, they note. In her own practice, Dr. Volkmann sees patients every 3 months to evaluate their symptoms and conduct pulmonary function tests (PFTs). For patients early in the course of ILD, she orders HRCT imaging once per year.
For Dr. Davis, the frequency of follow-up depends on the severity of ILD. “For minimally symptomatic patients without compromised lung function, we would generally follow annually. For patients with symptomatic ILD on stable therapy, we may monitor every 6 months. For patients with active/progressive ILD, we would generally be following at least every 1-3 months,” he said.
Screening and future research
While there is no evidence to recommend screening patients for ILD using CT, there are certain risk factors for ILD in RA patients, including a history of smoking, male sex, and high RA disease activity despite antirheumatic treatment, Dr. Volkmann said. In both of their practices, Dr. Davis and Dr. Volkmann screen with RA via HRCT and PFTs for ILD for patients with known risk factors that predispose them to the lung condition and/or for patients who report respiratory symptoms.
“We still don’t have an algorithm [for screening patients], and that is a desperate need in this field,” added Joshua J. Solomon, MD, a pulmonologist at National Jewish Health, Denver, whose research focuses on RA-associated ILD. While recommendations state that all patients with scleroderma should be screened with CT, ILD incidence is lower among patients with RA, and thus these screening recommendations need to be narrowed, he said. But more research is needed to better fine tune recommendations, he said; “The only thing you can do is give some expert consensus until there are good data.”
Dr. Volkmann has received consulting and speaking fees from Boehringer Ingelheim and institutional support for performing studies on systemic sclerosis for Kadmon, Forbius, Boehringer Ingelheim, Horizon, and Prometheus. Dr. Gordon, Dr. Davis, and Dr. Solomon report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
More clinical guidance is needed for monitoring interstitial lung disease (ILD) in patients with rheumatoid arthritis, according to a new commentary.
Though ILD is a leading cause of death among patients with RA, these patients are not routinely screened for ILD, the authors say, and there are currently no guidelines on how to monitor ILD progression in patients with RA.
“ILD associated with rheumatoid arthritis is a disease for which there’s been very little research done, so it’s an area of rheumatology where there are many unknowns,” lead author Elizabeth R. Volkmann, MD, who codirects the connective tissue disease–related interstitial lung disease (CTD-ILD) program at University of California, Los Angeles, told this news organization.
The commentary was published in The Lancet Rheumatology.
Defining disease
One of the major unknowns is how to define the disease, she said. RA patients sometimes undergo imaging for other medical reasons, and interstitial lung abnormalities are incidentally detected. These patients can be classified as having “preclinical” or “subclinical” ILD, as they do not yet have symptoms; however, there is no consensus as to what these terms mean, the commentary authors write. “The other problem that we have with these terms is that it sometimes creates the perception that this is a nonworrisome feature of rheumatoid arthritis,” Dr. Volkmann said, although the condition should be followed closely.
“We know we can detect imaging features of ILD in people who may not yet have symptoms, and we need to know when to define a clinically important informality that requires follow-up or treatment,” added John M. Davis III, MD, a rheumatologist at the Mayo Clinic, Rochester, Minn. He was not involved with the work.
Dr. Volkmann proposed eliminating the prefixes “pre” and “sub” when referring to ILD. “In other connective tissue diseases, like systemic sclerosis, for example, we can use the term ‘limited’ or ‘extensive’ ILD, based on the extent of involvement of the ILD on high-resolution computed tomography (HRCT) imaging,” she said. “This could potentially be something that is applied to how we classify patients with RA-ILD.”
Tracking ILD progression
Once ILD is identified, monitoring its progression poses challenges, as respiratory symptoms may be difficult to detect. RA patients may already be avoiding exercise because of joint pain, so they may not notice shortness of breath during physical activity, noted Jessica K. Gordon, MD, of the Hospital for Special Surgery, New York, in an interview with this news organization. She was not involved with the commentary. Cough is a potential symptom of ILD, but cough can also be the result of allergies, postnasal drip, or reflux, she said. Making the distinction between “preclinical” and symptomatic disease can be “complicated,” she added; “you may have to really dig.”
Additionally, there has been little research on the outcomes of patients with preclinical or subclinical ILD and clinical ILD, the commentary authors write. “It is therefore conceivable that some patients with rheumatoid arthritis diagnosed with preclinical or subclinical ILD could potentially have worse outcomes if both the rheumatoid arthritis and ILD are not monitored closely,” they note.
To better track RA-associated ILD for patients with and those without symptoms, the authors advocate for monitoring patients using pulmonary testing and CT scanning, as well as evaluating symptoms. How often these assessments should be conducted depends on the individual, they note. In her own practice, Dr. Volkmann sees patients every 3 months to evaluate their symptoms and conduct pulmonary function tests (PFTs). For patients early in the course of ILD, she orders HRCT imaging once per year.
For Dr. Davis, the frequency of follow-up depends on the severity of ILD. “For minimally symptomatic patients without compromised lung function, we would generally follow annually. For patients with symptomatic ILD on stable therapy, we may monitor every 6 months. For patients with active/progressive ILD, we would generally be following at least every 1-3 months,” he said.
Screening and future research
While there is no evidence to recommend screening patients for ILD using CT, there are certain risk factors for ILD in RA patients, including a history of smoking, male sex, and high RA disease activity despite antirheumatic treatment, Dr. Volkmann said. In both of their practices, Dr. Davis and Dr. Volkmann screen with RA via HRCT and PFTs for ILD for patients with known risk factors that predispose them to the lung condition and/or for patients who report respiratory symptoms.
“We still don’t have an algorithm [for screening patients], and that is a desperate need in this field,” added Joshua J. Solomon, MD, a pulmonologist at National Jewish Health, Denver, whose research focuses on RA-associated ILD. While recommendations state that all patients with scleroderma should be screened with CT, ILD incidence is lower among patients with RA, and thus these screening recommendations need to be narrowed, he said. But more research is needed to better fine tune recommendations, he said; “The only thing you can do is give some expert consensus until there are good data.”
Dr. Volkmann has received consulting and speaking fees from Boehringer Ingelheim and institutional support for performing studies on systemic sclerosis for Kadmon, Forbius, Boehringer Ingelheim, Horizon, and Prometheus. Dr. Gordon, Dr. Davis, and Dr. Solomon report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
More clinical guidance is needed for monitoring interstitial lung disease (ILD) in patients with rheumatoid arthritis, according to a new commentary.
Though ILD is a leading cause of death among patients with RA, these patients are not routinely screened for ILD, the authors say, and there are currently no guidelines on how to monitor ILD progression in patients with RA.
“ILD associated with rheumatoid arthritis is a disease for which there’s been very little research done, so it’s an area of rheumatology where there are many unknowns,” lead author Elizabeth R. Volkmann, MD, who codirects the connective tissue disease–related interstitial lung disease (CTD-ILD) program at University of California, Los Angeles, told this news organization.
The commentary was published in The Lancet Rheumatology.
Defining disease
One of the major unknowns is how to define the disease, she said. RA patients sometimes undergo imaging for other medical reasons, and interstitial lung abnormalities are incidentally detected. These patients can be classified as having “preclinical” or “subclinical” ILD, as they do not yet have symptoms; however, there is no consensus as to what these terms mean, the commentary authors write. “The other problem that we have with these terms is that it sometimes creates the perception that this is a nonworrisome feature of rheumatoid arthritis,” Dr. Volkmann said, although the condition should be followed closely.
“We know we can detect imaging features of ILD in people who may not yet have symptoms, and we need to know when to define a clinically important informality that requires follow-up or treatment,” added John M. Davis III, MD, a rheumatologist at the Mayo Clinic, Rochester, Minn. He was not involved with the work.
Dr. Volkmann proposed eliminating the prefixes “pre” and “sub” when referring to ILD. “In other connective tissue diseases, like systemic sclerosis, for example, we can use the term ‘limited’ or ‘extensive’ ILD, based on the extent of involvement of the ILD on high-resolution computed tomography (HRCT) imaging,” she said. “This could potentially be something that is applied to how we classify patients with RA-ILD.”
Tracking ILD progression
Once ILD is identified, monitoring its progression poses challenges, as respiratory symptoms may be difficult to detect. RA patients may already be avoiding exercise because of joint pain, so they may not notice shortness of breath during physical activity, noted Jessica K. Gordon, MD, of the Hospital for Special Surgery, New York, in an interview with this news organization. She was not involved with the commentary. Cough is a potential symptom of ILD, but cough can also be the result of allergies, postnasal drip, or reflux, she said. Making the distinction between “preclinical” and symptomatic disease can be “complicated,” she added; “you may have to really dig.”
Additionally, there has been little research on the outcomes of patients with preclinical or subclinical ILD and clinical ILD, the commentary authors write. “It is therefore conceivable that some patients with rheumatoid arthritis diagnosed with preclinical or subclinical ILD could potentially have worse outcomes if both the rheumatoid arthritis and ILD are not monitored closely,” they note.
To better track RA-associated ILD for patients with and those without symptoms, the authors advocate for monitoring patients using pulmonary testing and CT scanning, as well as evaluating symptoms. How often these assessments should be conducted depends on the individual, they note. In her own practice, Dr. Volkmann sees patients every 3 months to evaluate their symptoms and conduct pulmonary function tests (PFTs). For patients early in the course of ILD, she orders HRCT imaging once per year.
For Dr. Davis, the frequency of follow-up depends on the severity of ILD. “For minimally symptomatic patients without compromised lung function, we would generally follow annually. For patients with symptomatic ILD on stable therapy, we may monitor every 6 months. For patients with active/progressive ILD, we would generally be following at least every 1-3 months,” he said.
Screening and future research
While there is no evidence to recommend screening patients for ILD using CT, there are certain risk factors for ILD in RA patients, including a history of smoking, male sex, and high RA disease activity despite antirheumatic treatment, Dr. Volkmann said. In both of their practices, Dr. Davis and Dr. Volkmann screen with RA via HRCT and PFTs for ILD for patients with known risk factors that predispose them to the lung condition and/or for patients who report respiratory symptoms.
“We still don’t have an algorithm [for screening patients], and that is a desperate need in this field,” added Joshua J. Solomon, MD, a pulmonologist at National Jewish Health, Denver, whose research focuses on RA-associated ILD. While recommendations state that all patients with scleroderma should be screened with CT, ILD incidence is lower among patients with RA, and thus these screening recommendations need to be narrowed, he said. But more research is needed to better fine tune recommendations, he said; “The only thing you can do is give some expert consensus until there are good data.”
Dr. Volkmann has received consulting and speaking fees from Boehringer Ingelheim and institutional support for performing studies on systemic sclerosis for Kadmon, Forbius, Boehringer Ingelheim, Horizon, and Prometheus. Dr. Gordon, Dr. Davis, and Dr. Solomon report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Beware risk of sedatives for respiratory patients
Both asthma and chronic obstructive pulmonary disease can be challenging to diagnose, and medication-driven episodes of sedation or hypoventilation are often overlooked as causes of acute exacerbations in these conditions, according to a letter published in The Lancet Respiratory Medicine.
write Christos V. Chalitsios, PhD, of the University of Nottingham, England, and colleagues.
The authors note that exacerbations are the main complications of both asthma and COPD, and stress the importance of identifying causes and preventive strategies.
Sedatives such as opioids have been shown to depress respiratory drive, reduce muscle tone, and increase the risk of pneumonia, they write. The authors also propose that the risk of sedative-induced aspiration or hypoventilation would be associated with medications including pregabalin, gabapentin, and amitriptyline.
Other mechanisms may be involved in the association between sedatives and exacerbations in asthma and COPD. For example, sedative medications can suppress coughing, which may promote airway mucous compaction and possible infection, the authors write.
Most research involving prevention of asthma and COPD exacerbations has not addressed the potential impact of sedatives taken for reasons outside of obstructive lung disease, the authors say.
“Although the risk of sedation and hypoventilation events are known to be increased by opioids and antipsychotic drugs, there has not been a systematic assessment of commonly prescribed medications with potential respiratory side-effects, including gabapentin, amitriptyline, and pregabalin,” they write.
Polypharmacy is increasingly common and results in many patients with asthma or COPD presenting for treatment of acute exacerbations while on a combination of gabapentin, pregabalin, amitriptyline, and opioids, the authors note; “however, there is little data or disease-specific guidance on how best to manage this problem, which often starts with a prescription in primary care,” they write. Simply stopping sedatives is not an option for many patients given the addictive nature of these drugs and the unlikely resolution of the condition for which the drugs were prescribed, the authors say. However, “cautious dose reduction” of sedatives is possible once patients understand the reason, they add.
Clinicians may be able to suggest reduced doses and alternative treatments to patients with asthma and COPD while highlighting the risk of respiratory depression and polypharmacy – “potentially reducing the number of exacerbations of obstructive lung disease,” the authors conclude.
The study received no outside funding. The authors have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Both asthma and chronic obstructive pulmonary disease can be challenging to diagnose, and medication-driven episodes of sedation or hypoventilation are often overlooked as causes of acute exacerbations in these conditions, according to a letter published in The Lancet Respiratory Medicine.
write Christos V. Chalitsios, PhD, of the University of Nottingham, England, and colleagues.
The authors note that exacerbations are the main complications of both asthma and COPD, and stress the importance of identifying causes and preventive strategies.
Sedatives such as opioids have been shown to depress respiratory drive, reduce muscle tone, and increase the risk of pneumonia, they write. The authors also propose that the risk of sedative-induced aspiration or hypoventilation would be associated with medications including pregabalin, gabapentin, and amitriptyline.
Other mechanisms may be involved in the association between sedatives and exacerbations in asthma and COPD. For example, sedative medications can suppress coughing, which may promote airway mucous compaction and possible infection, the authors write.
Most research involving prevention of asthma and COPD exacerbations has not addressed the potential impact of sedatives taken for reasons outside of obstructive lung disease, the authors say.
“Although the risk of sedation and hypoventilation events are known to be increased by opioids and antipsychotic drugs, there has not been a systematic assessment of commonly prescribed medications with potential respiratory side-effects, including gabapentin, amitriptyline, and pregabalin,” they write.
Polypharmacy is increasingly common and results in many patients with asthma or COPD presenting for treatment of acute exacerbations while on a combination of gabapentin, pregabalin, amitriptyline, and opioids, the authors note; “however, there is little data or disease-specific guidance on how best to manage this problem, which often starts with a prescription in primary care,” they write. Simply stopping sedatives is not an option for many patients given the addictive nature of these drugs and the unlikely resolution of the condition for which the drugs were prescribed, the authors say. However, “cautious dose reduction” of sedatives is possible once patients understand the reason, they add.
Clinicians may be able to suggest reduced doses and alternative treatments to patients with asthma and COPD while highlighting the risk of respiratory depression and polypharmacy – “potentially reducing the number of exacerbations of obstructive lung disease,” the authors conclude.
The study received no outside funding. The authors have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Both asthma and chronic obstructive pulmonary disease can be challenging to diagnose, and medication-driven episodes of sedation or hypoventilation are often overlooked as causes of acute exacerbations in these conditions, according to a letter published in The Lancet Respiratory Medicine.
write Christos V. Chalitsios, PhD, of the University of Nottingham, England, and colleagues.
The authors note that exacerbations are the main complications of both asthma and COPD, and stress the importance of identifying causes and preventive strategies.
Sedatives such as opioids have been shown to depress respiratory drive, reduce muscle tone, and increase the risk of pneumonia, they write. The authors also propose that the risk of sedative-induced aspiration or hypoventilation would be associated with medications including pregabalin, gabapentin, and amitriptyline.
Other mechanisms may be involved in the association between sedatives and exacerbations in asthma and COPD. For example, sedative medications can suppress coughing, which may promote airway mucous compaction and possible infection, the authors write.
Most research involving prevention of asthma and COPD exacerbations has not addressed the potential impact of sedatives taken for reasons outside of obstructive lung disease, the authors say.
“Although the risk of sedation and hypoventilation events are known to be increased by opioids and antipsychotic drugs, there has not been a systematic assessment of commonly prescribed medications with potential respiratory side-effects, including gabapentin, amitriptyline, and pregabalin,” they write.
Polypharmacy is increasingly common and results in many patients with asthma or COPD presenting for treatment of acute exacerbations while on a combination of gabapentin, pregabalin, amitriptyline, and opioids, the authors note; “however, there is little data or disease-specific guidance on how best to manage this problem, which often starts with a prescription in primary care,” they write. Simply stopping sedatives is not an option for many patients given the addictive nature of these drugs and the unlikely resolution of the condition for which the drugs were prescribed, the authors say. However, “cautious dose reduction” of sedatives is possible once patients understand the reason, they add.
Clinicians may be able to suggest reduced doses and alternative treatments to patients with asthma and COPD while highlighting the risk of respiratory depression and polypharmacy – “potentially reducing the number of exacerbations of obstructive lung disease,” the authors conclude.
The study received no outside funding. The authors have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.