User login
Sexual health assessment and counseling: oncology nurses’ perceptions, practices, and perceived barriers
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
Cancer clinical trial enrollment of diverse and underserved patients within an urban safety net hospital
Background Enrollment rates onto cancer clinical trials are low and reflect a small subset of the population of which even fewer participants come from populations of racial or ethnic diversity or low socioeconomic status. There is a need to increase enrollment onto cancer clinical trials with a focus on recruitment of a diverse, underrepresented patient population.
Objective To use the electronic medical record (EMR) to understand the eligibility and enrollment rates for all available cancer trials in the ambulatory care setting at an urban safety net hospital to identify specific strategies for enhanced accrual onto cancer clinical trials of diverse and underserved patients.
Methods A clinical trial screening note was created for the EMR by the clinical trials office at an urban safety net hospital. 847 cancer clinical trial screening notes were extracted from the EMR between January 1, 2010 and December 31, 2010. During that time, 99 cancer trials were registered for accrual, including clinical treatment, survey, data repository, imaging, and symptom management trials. Data on eligibility, enrollment status, and relationship to sociodemographic status were compared.
Limitations This is a single-institution and retrospective study.
Conclusion The findings demonstrate that a formal process of tracking cancer clinical trial screens using an EMR can document baseline rates of institution-specific accrual patterns and identify targeted strategies for increasing cancer clinical trial enrollment among a vulnerable patient population. Offering nontreatment trials may be an important and strategic method of engaging this vulnerable population in clinical research.
Funding/sponsorship Boston Medical Center Minority-Based Community Clinical Oncology Program (NCI 1U-10CA129519- 01A1), Boston Me
Click on the PDF icon at the top of this introduction to read the full article.
Background Enrollment rates onto cancer clinical trials are low and reflect a small subset of the population of which even fewer participants come from populations of racial or ethnic diversity or low socioeconomic status. There is a need to increase enrollment onto cancer clinical trials with a focus on recruitment of a diverse, underrepresented patient population.
Objective To use the electronic medical record (EMR) to understand the eligibility and enrollment rates for all available cancer trials in the ambulatory care setting at an urban safety net hospital to identify specific strategies for enhanced accrual onto cancer clinical trials of diverse and underserved patients.
Methods A clinical trial screening note was created for the EMR by the clinical trials office at an urban safety net hospital. 847 cancer clinical trial screening notes were extracted from the EMR between January 1, 2010 and December 31, 2010. During that time, 99 cancer trials were registered for accrual, including clinical treatment, survey, data repository, imaging, and symptom management trials. Data on eligibility, enrollment status, and relationship to sociodemographic status were compared.
Limitations This is a single-institution and retrospective study.
Conclusion The findings demonstrate that a formal process of tracking cancer clinical trial screens using an EMR can document baseline rates of institution-specific accrual patterns and identify targeted strategies for increasing cancer clinical trial enrollment among a vulnerable patient population. Offering nontreatment trials may be an important and strategic method of engaging this vulnerable population in clinical research.
Funding/sponsorship Boston Medical Center Minority-Based Community Clinical Oncology Program (NCI 1U-10CA129519- 01A1), Boston Me
Click on the PDF icon at the top of this introduction to read the full article.
Background Enrollment rates onto cancer clinical trials are low and reflect a small subset of the population of which even fewer participants come from populations of racial or ethnic diversity or low socioeconomic status. There is a need to increase enrollment onto cancer clinical trials with a focus on recruitment of a diverse, underrepresented patient population.
Objective To use the electronic medical record (EMR) to understand the eligibility and enrollment rates for all available cancer trials in the ambulatory care setting at an urban safety net hospital to identify specific strategies for enhanced accrual onto cancer clinical trials of diverse and underserved patients.
Methods A clinical trial screening note was created for the EMR by the clinical trials office at an urban safety net hospital. 847 cancer clinical trial screening notes were extracted from the EMR between January 1, 2010 and December 31, 2010. During that time, 99 cancer trials were registered for accrual, including clinical treatment, survey, data repository, imaging, and symptom management trials. Data on eligibility, enrollment status, and relationship to sociodemographic status were compared.
Limitations This is a single-institution and retrospective study.
Conclusion The findings demonstrate that a formal process of tracking cancer clinical trial screens using an EMR can document baseline rates of institution-specific accrual patterns and identify targeted strategies for increasing cancer clinical trial enrollment among a vulnerable patient population. Offering nontreatment trials may be an important and strategic method of engaging this vulnerable population in clinical research.
Funding/sponsorship Boston Medical Center Minority-Based Community Clinical Oncology Program (NCI 1U-10CA129519- 01A1), Boston Me
Click on the PDF icon at the top of this introduction to read the full article.
Implementation of distress screening in an oncology setting
The recommendations of numerous groups, such as the Institute of Medicine and the National Comprehensive Cancer Network, have resulted in the first regulatory standard on distress screening in oncology implemented in 2015 by the American College of Surgeons Commission on Cancer. This practice-changing standard promises to result in better quality cancer care, but presents unique challenges to many centers struggling to provide high-quality practical assessment and management of distress. The current paper reviews the history behind the CoC standard, identifies the most prevalent symptoms underlying distress, and discusses the importance of distress screening. We also review some commonly used instruments for assessing distress, and address barriers to implementation of screening and management.
Click on the PDF icon at the top of this introduction to read the full article.
The recommendations of numerous groups, such as the Institute of Medicine and the National Comprehensive Cancer Network, have resulted in the first regulatory standard on distress screening in oncology implemented in 2015 by the American College of Surgeons Commission on Cancer. This practice-changing standard promises to result in better quality cancer care, but presents unique challenges to many centers struggling to provide high-quality practical assessment and management of distress. The current paper reviews the history behind the CoC standard, identifies the most prevalent symptoms underlying distress, and discusses the importance of distress screening. We also review some commonly used instruments for assessing distress, and address barriers to implementation of screening and management.
Click on the PDF icon at the top of this introduction to read the full article.
The recommendations of numerous groups, such as the Institute of Medicine and the National Comprehensive Cancer Network, have resulted in the first regulatory standard on distress screening in oncology implemented in 2015 by the American College of Surgeons Commission on Cancer. This practice-changing standard promises to result in better quality cancer care, but presents unique challenges to many centers struggling to provide high-quality practical assessment and management of distress. The current paper reviews the history behind the CoC standard, identifies the most prevalent symptoms underlying distress, and discusses the importance of distress screening. We also review some commonly used instruments for assessing distress, and address barriers to implementation of screening and management.
Click on the PDF icon at the top of this introduction to read the full article.
Filgrastim-sndz debuts as the first biosimilar approved in United States
Pregnancy did not increase Hodgkin lymphoma relapse rate
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
Women who become pregnant while in remission from Hodgkin lymphoma were not at increased risk for cancer relapse, according to an analysis of data from Swedish health care registries combined with medical records.
Of 449 women who were diagnosed with Hodgkin lymphoma between 1992 and 2009, 144 (32%) became pregnant during follow-up, which started 6 months after diagnosis, when the disease was assumed to be in remission. Only one of these women experienced a pregnancy-associated relapse, which was defined as a relapse occurring during pregnancy or within 5 years of delivery. Of the women who did not become pregnant, 46 had a relapse.
The effect of pregnancy on relapse has been a concern of patients and clinicians, but “our findings suggest that the risk of pregnancy-associated relapse does not need to be taken into account in family planning for women whose Hodgkin lymphoma is in remission,” said Caroline E. Weibull of Karolinska Institutet in Stockholm, and her associates.
The researchers used the nationwide “Swedish Cancer Register” to identify all cases of Hodgkin lymphoma (reporting is mandatory) and merged this data with clinical information from other registries and medical records.
The pregnancy rates were similar among women who had limited- and advanced-stage disease and among women with and without B symptoms at diagnosis – a finding that negates consideration of a so-called “healthy mother effect” in protecting against relapse, they wrote (J Clin Onc. 2015 Dec. 14 [doi:10.1200/JCO.2015.63.3446]).
The researchers also found that the absolute risk for relapse was highest in the first 2-3 years after diagnosis, which suggests that women should be advised, “if possible, to wait 2 years after cessation of treatment before becoming pregnant.” Additionally, the relapse rate more than doubled in women aged 30 years or older at diagnosis, compared with women aged 18-24 years at diagnosis – a finding consistent with previous research, they noted.
Women in the study were aged 18-40 at diagnosis. Follow-up ended on the date of relapse, the date of death, or at the end of 2010, whichever came first.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Pregnancy did not increase the risk of relapse of Hodgkin lymphoma in a population-based study.
Major finding: Of 144 women who became pregnant 6 months or longer after diagnosis of Hodgkin lymphoma, 1 experienced a pregnancy-associated relapse.
Data source: Population-based study utilizing Swedish health care registries and medical records, in which 449 women with Hodgkin lymphoma diagnoses, and 47 relapses, were identified.
Disclosures: The study was supported by the Swedish Cancer Society, the Strategic Research Program in Epidemiology at Karolinska Institutet, the Swedish Society for Medicine, and the Swedish Society for Medical Research.
Early infection-related hospitalization portends poor breast cancer prognosis
SAN ANTONIO – Hospitalization for an infection within the first year following diagnosis of primary nonmetastatic breast cancer is a red flag for increased risk of subsequent development of distant metastases or breast cancer–related death, Judith S. Brand, Ph.D., reported at the San Antonio Breast Cancer Symposium.
This increased risk for future adverse breast cancer outcomes was statistically significant and clinically meaningful for women hospitalized for respiratory tract or skin infections or sepsis. Those are the patients for whom particularly close monitoring for recurrence of breast cancer is warranted in the next 5 years, said Dr. Brand of the Karolinska Institute, Stockholm.
In contrast, hospitalization for a gastrointestinal or urinary tract infection didn’t achieve significance as an independent predictor of increased risk of adverse breast cancer outcomes.
Dr. Brand presented a prospective population-based study of 8,338 women diagnosed with stage I-III breast cancer in the Stockholm area during 2001-2008. During a median 4.9 years of follow-up after diagnosis, 720 women had an infection-related hospitalization, with the great majority of these events occurring during the first year.
The incidence of hospitalization for sepsis among breast cancer patients in their first year post diagnosis was 14-fold greater than in the general Swedish female population matched for age and year. Respiratory infections resulting in hospitalization were fourfold more frequent than in the general population, skin infections were eightfold more common, and GI infections were twice as common.
Infection-related hospitalizations had a strong and independent association with breast cancer mortality during follow-up, as seen in a multivariate analysis adjusted for age at diagnosis, comorbid conditions, infectious disease history, type of breast cancer therapy, and tumor characteristics including size, grade, hormone receptor status, and lymph node involvement.
Moreover, the risk of developing distant metastases was 50%-78% greater in breast cancer patients hospitalized for respiratory infection, sepsis, or skin infection, compared with breast cancer patients who didn’t have an infection-related hospitalization.
“We think the sepsis results are the most interesting findings,” Dr. Brand said in an interview. “Sepsis could be an expression of an immunosuppressed state. And sepsis itself can induce immunosuppression for a long time, which could trigger tumor growth. Animal studies have shown that in postseptic mice, tumor grows faster.”
Infection-related hospitalizations didn’t increase the future risk of locoregional recurrences.
Independent predictors of infection-related hospitalization included older age, comorbidities, markers indicative of greater tumor aggressiveness, and treatment with chemotherapy or axillary radiotherapy.
“This shows that the risk of infection-related hospitalizations is not only due to immunosuppression caused by chemotherapy, but that the characteristics of the tumor itself play a role, as do patient characteristics. This is the first epidemiologic study to show with very extensive data that all three elements contribute to the risk,” Dr. Brand said.
SAN ANTONIO – Hospitalization for an infection within the first year following diagnosis of primary nonmetastatic breast cancer is a red flag for increased risk of subsequent development of distant metastases or breast cancer–related death, Judith S. Brand, Ph.D., reported at the San Antonio Breast Cancer Symposium.
This increased risk for future adverse breast cancer outcomes was statistically significant and clinically meaningful for women hospitalized for respiratory tract or skin infections or sepsis. Those are the patients for whom particularly close monitoring for recurrence of breast cancer is warranted in the next 5 years, said Dr. Brand of the Karolinska Institute, Stockholm.
In contrast, hospitalization for a gastrointestinal or urinary tract infection didn’t achieve significance as an independent predictor of increased risk of adverse breast cancer outcomes.
Dr. Brand presented a prospective population-based study of 8,338 women diagnosed with stage I-III breast cancer in the Stockholm area during 2001-2008. During a median 4.9 years of follow-up after diagnosis, 720 women had an infection-related hospitalization, with the great majority of these events occurring during the first year.
The incidence of hospitalization for sepsis among breast cancer patients in their first year post diagnosis was 14-fold greater than in the general Swedish female population matched for age and year. Respiratory infections resulting in hospitalization were fourfold more frequent than in the general population, skin infections were eightfold more common, and GI infections were twice as common.
Infection-related hospitalizations had a strong and independent association with breast cancer mortality during follow-up, as seen in a multivariate analysis adjusted for age at diagnosis, comorbid conditions, infectious disease history, type of breast cancer therapy, and tumor characteristics including size, grade, hormone receptor status, and lymph node involvement.
Moreover, the risk of developing distant metastases was 50%-78% greater in breast cancer patients hospitalized for respiratory infection, sepsis, or skin infection, compared with breast cancer patients who didn’t have an infection-related hospitalization.
“We think the sepsis results are the most interesting findings,” Dr. Brand said in an interview. “Sepsis could be an expression of an immunosuppressed state. And sepsis itself can induce immunosuppression for a long time, which could trigger tumor growth. Animal studies have shown that in postseptic mice, tumor grows faster.”
Infection-related hospitalizations didn’t increase the future risk of locoregional recurrences.
Independent predictors of infection-related hospitalization included older age, comorbidities, markers indicative of greater tumor aggressiveness, and treatment with chemotherapy or axillary radiotherapy.
“This shows that the risk of infection-related hospitalizations is not only due to immunosuppression caused by chemotherapy, but that the characteristics of the tumor itself play a role, as do patient characteristics. This is the first epidemiologic study to show with very extensive data that all three elements contribute to the risk,” Dr. Brand said.
SAN ANTONIO – Hospitalization for an infection within the first year following diagnosis of primary nonmetastatic breast cancer is a red flag for increased risk of subsequent development of distant metastases or breast cancer–related death, Judith S. Brand, Ph.D., reported at the San Antonio Breast Cancer Symposium.
This increased risk for future adverse breast cancer outcomes was statistically significant and clinically meaningful for women hospitalized for respiratory tract or skin infections or sepsis. Those are the patients for whom particularly close monitoring for recurrence of breast cancer is warranted in the next 5 years, said Dr. Brand of the Karolinska Institute, Stockholm.
In contrast, hospitalization for a gastrointestinal or urinary tract infection didn’t achieve significance as an independent predictor of increased risk of adverse breast cancer outcomes.
Dr. Brand presented a prospective population-based study of 8,338 women diagnosed with stage I-III breast cancer in the Stockholm area during 2001-2008. During a median 4.9 years of follow-up after diagnosis, 720 women had an infection-related hospitalization, with the great majority of these events occurring during the first year.
The incidence of hospitalization for sepsis among breast cancer patients in their first year post diagnosis was 14-fold greater than in the general Swedish female population matched for age and year. Respiratory infections resulting in hospitalization were fourfold more frequent than in the general population, skin infections were eightfold more common, and GI infections were twice as common.
Infection-related hospitalizations had a strong and independent association with breast cancer mortality during follow-up, as seen in a multivariate analysis adjusted for age at diagnosis, comorbid conditions, infectious disease history, type of breast cancer therapy, and tumor characteristics including size, grade, hormone receptor status, and lymph node involvement.
Moreover, the risk of developing distant metastases was 50%-78% greater in breast cancer patients hospitalized for respiratory infection, sepsis, or skin infection, compared with breast cancer patients who didn’t have an infection-related hospitalization.
“We think the sepsis results are the most interesting findings,” Dr. Brand said in an interview. “Sepsis could be an expression of an immunosuppressed state. And sepsis itself can induce immunosuppression for a long time, which could trigger tumor growth. Animal studies have shown that in postseptic mice, tumor grows faster.”
Infection-related hospitalizations didn’t increase the future risk of locoregional recurrences.
Independent predictors of infection-related hospitalization included older age, comorbidities, markers indicative of greater tumor aggressiveness, and treatment with chemotherapy or axillary radiotherapy.
“This shows that the risk of infection-related hospitalizations is not only due to immunosuppression caused by chemotherapy, but that the characteristics of the tumor itself play a role, as do patient characteristics. This is the first epidemiologic study to show with very extensive data that all three elements contribute to the risk,” Dr. Brand said.
AT SABCS 2015
Key clinical point: Particularly close monitoring for adverse breast cancer outcomes is warranted for women with an infection-related hospitalization during the first year after breast cancer diagnosis.
Major finding: Women hospitalized for sepsis or a respiratory or skin infection during the first year after being diagnosed with stage I-III breast cancer were up to 4.8 times more likely to subsequently die of breast cancer than those without an infection-related hospitalization.
Data source: This was a prospective observational study of 8,338 Stockholm-area breast cancer patients followed for a median of 4.9 years post diagnosis.
Disclosures: The study presenter reported having no financial conflicts regarding this university-funded study.
Oncology 2015: new therapies and new transitions toward value-based cancer care
The past year has been an exciting one for new oncology and hematology drug approvals and the continued evolution of our oncology delivery system toward high quality and value. In all, at press time in mid-November, the US Food and Drug Administration (FDA) had approved or granted expanded indications for 24 drugs, compared with 19 in the 2 preceding years. Of those 24 approvals, 7 were accelerated and 6 were expanded approvals, and 3 alone were for the immunotherapeutic drug, nivolumab – 2 for non-small-cell lung cancer (NSCLC) and 1 for metastatic melanoma.
Click on the PDF icon at the top of this introduction to read the full article.
The past year has been an exciting one for new oncology and hematology drug approvals and the continued evolution of our oncology delivery system toward high quality and value. In all, at press time in mid-November, the US Food and Drug Administration (FDA) had approved or granted expanded indications for 24 drugs, compared with 19 in the 2 preceding years. Of those 24 approvals, 7 were accelerated and 6 were expanded approvals, and 3 alone were for the immunotherapeutic drug, nivolumab – 2 for non-small-cell lung cancer (NSCLC) and 1 for metastatic melanoma.
Click on the PDF icon at the top of this introduction to read the full article.
The past year has been an exciting one for new oncology and hematology drug approvals and the continued evolution of our oncology delivery system toward high quality and value. In all, at press time in mid-November, the US Food and Drug Administration (FDA) had approved or granted expanded indications for 24 drugs, compared with 19 in the 2 preceding years. Of those 24 approvals, 7 were accelerated and 6 were expanded approvals, and 3 alone were for the immunotherapeutic drug, nivolumab – 2 for non-small-cell lung cancer (NSCLC) and 1 for metastatic melanoma.
Click on the PDF icon at the top of this introduction to read the full article.
Review: Opioid prescriptions are the work of many physicians
A “broad swath” of Medicare providers wrote scripts for opioids in 2013, contradicting the idea that the overdose epidemic is mainly the work of “small groups of prolific prescribers and corrupt pill mills,” investigators wrote online in JAMA Internal Medicine.
“Contrary to the California workers’ compensation data showing a small subset of prescribers accounting for a disproportionately large percentage of opioid prescribing, Medicare opioid prescribing is distributed across many prescribers and is, if anything, less skewed than all drug prescribing,” said Dr. Jonathan H. Chen of the Veterans Affairs Palo Alto (Calif.) Health Care System, and his associates.
Their study included 808,020 prescribers and almost 1.2 billion Medicare Part D claims worth nearly $81 billion dollars. They focused on schedule II opioid prescriptions containing oxycodone, fentanyl, hydrocodone, morphine, methadone, hydromorphone, oxymorphone, meperidine, codeine, opium, or levorphanol (JAMA Intern Med. 2015 Dec 14. doi: 10.1001/jamainternmed.2015.6662).
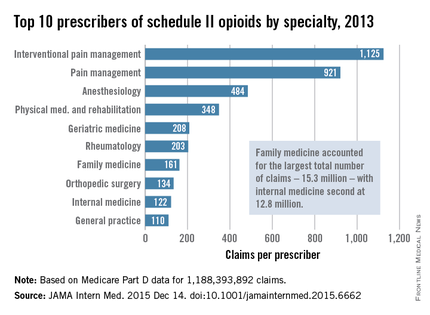
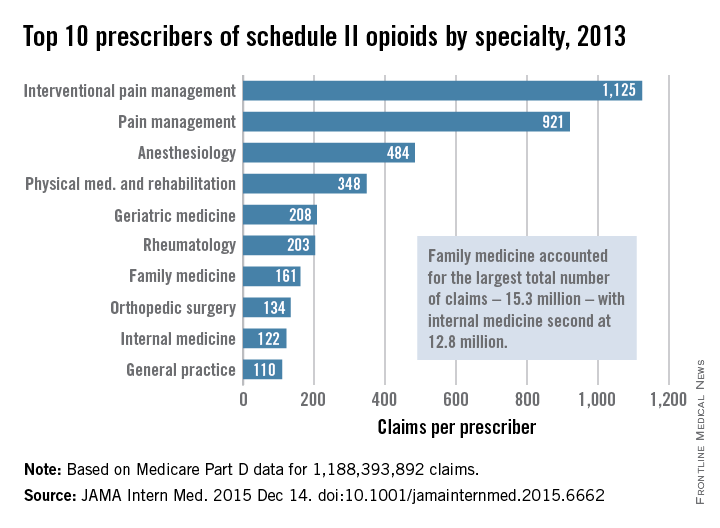
Not surprisingly, specialists in pain management, anesthesia, and physical medicine wrote the most prescriptions per provider. But family practitioners, internists, nurse practitioners, and physician assistants wrote 35,268,234 prescriptions – more than all other specialties combined. “The trends hold up across state lines, with negligible geographic variability,” the researchers said.
The findings contradict an analysis of California workers’ compensation data, in which 1% of prescribers accounted for a third of schedule II opioid prescriptions, and 10% of prescribers accounted for 80% of prescriptions, the investigators noted. Nonetheless, 10% of Medicare prescribers in Dr. Chen’s study accounted for 78% of the total cost of opioids, possibly because they were prescribing pricier formulations or higher doses.
Overall, the findings suggest that opioid prescribing is “widespread” and “relatively indifferent to individual physicians, specialty, or region” – and that efforts to stem the tide must be equally broad, the researchers concluded.
Their study was supported by the VA Office of Academic Affiliations, the VA Health Services Research and Development Service, the National Institute of General Medical Sciences, and the Peter F. McManus Charitable Trust. The researchers had no disclosures.
A “broad swath” of Medicare providers wrote scripts for opioids in 2013, contradicting the idea that the overdose epidemic is mainly the work of “small groups of prolific prescribers and corrupt pill mills,” investigators wrote online in JAMA Internal Medicine.
“Contrary to the California workers’ compensation data showing a small subset of prescribers accounting for a disproportionately large percentage of opioid prescribing, Medicare opioid prescribing is distributed across many prescribers and is, if anything, less skewed than all drug prescribing,” said Dr. Jonathan H. Chen of the Veterans Affairs Palo Alto (Calif.) Health Care System, and his associates.
Their study included 808,020 prescribers and almost 1.2 billion Medicare Part D claims worth nearly $81 billion dollars. They focused on schedule II opioid prescriptions containing oxycodone, fentanyl, hydrocodone, morphine, methadone, hydromorphone, oxymorphone, meperidine, codeine, opium, or levorphanol (JAMA Intern Med. 2015 Dec 14. doi: 10.1001/jamainternmed.2015.6662).

Not surprisingly, specialists in pain management, anesthesia, and physical medicine wrote the most prescriptions per provider. But family practitioners, internists, nurse practitioners, and physician assistants wrote 35,268,234 prescriptions – more than all other specialties combined. “The trends hold up across state lines, with negligible geographic variability,” the researchers said.
The findings contradict an analysis of California workers’ compensation data, in which 1% of prescribers accounted for a third of schedule II opioid prescriptions, and 10% of prescribers accounted for 80% of prescriptions, the investigators noted. Nonetheless, 10% of Medicare prescribers in Dr. Chen’s study accounted for 78% of the total cost of opioids, possibly because they were prescribing pricier formulations or higher doses.
Overall, the findings suggest that opioid prescribing is “widespread” and “relatively indifferent to individual physicians, specialty, or region” – and that efforts to stem the tide must be equally broad, the researchers concluded.
Their study was supported by the VA Office of Academic Affiliations, the VA Health Services Research and Development Service, the National Institute of General Medical Sciences, and the Peter F. McManus Charitable Trust. The researchers had no disclosures.
A “broad swath” of Medicare providers wrote scripts for opioids in 2013, contradicting the idea that the overdose epidemic is mainly the work of “small groups of prolific prescribers and corrupt pill mills,” investigators wrote online in JAMA Internal Medicine.
“Contrary to the California workers’ compensation data showing a small subset of prescribers accounting for a disproportionately large percentage of opioid prescribing, Medicare opioid prescribing is distributed across many prescribers and is, if anything, less skewed than all drug prescribing,” said Dr. Jonathan H. Chen of the Veterans Affairs Palo Alto (Calif.) Health Care System, and his associates.
Their study included 808,020 prescribers and almost 1.2 billion Medicare Part D claims worth nearly $81 billion dollars. They focused on schedule II opioid prescriptions containing oxycodone, fentanyl, hydrocodone, morphine, methadone, hydromorphone, oxymorphone, meperidine, codeine, opium, or levorphanol (JAMA Intern Med. 2015 Dec 14. doi: 10.1001/jamainternmed.2015.6662).

Not surprisingly, specialists in pain management, anesthesia, and physical medicine wrote the most prescriptions per provider. But family practitioners, internists, nurse practitioners, and physician assistants wrote 35,268,234 prescriptions – more than all other specialties combined. “The trends hold up across state lines, with negligible geographic variability,” the researchers said.
The findings contradict an analysis of California workers’ compensation data, in which 1% of prescribers accounted for a third of schedule II opioid prescriptions, and 10% of prescribers accounted for 80% of prescriptions, the investigators noted. Nonetheless, 10% of Medicare prescribers in Dr. Chen’s study accounted for 78% of the total cost of opioids, possibly because they were prescribing pricier formulations or higher doses.
Overall, the findings suggest that opioid prescribing is “widespread” and “relatively indifferent to individual physicians, specialty, or region” – and that efforts to stem the tide must be equally broad, the researchers concluded.
Their study was supported by the VA Office of Academic Affiliations, the VA Health Services Research and Development Service, the National Institute of General Medical Sciences, and the Peter F. McManus Charitable Trust. The researchers had no disclosures.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Many different types of general practitioners and specialists often prescribe opioids to Medicare beneficiaries.
Major finding: Family practitioners, internists, nurse practitioners, and physician assistants wrote 35,268,234 prescriptions – more than all other specialties combined.
Data source: An analysis of nearly 1.2 billion Medicare part D claims from 2013.
Disclosures: The study was supported by the VA Office of Academic Affiliations, the VA Health Services Research and Development Service, the National Institute of General Medical Sciences, and the Peter F. McManus Charitable Trust. The researchers had no disclosures.
AHA: Older breast cancer patients more likely to die of heart disease than malignancy
ORLANDO – Women diagnosed with localized breast cancer while in their 70s have a higher mortality from cardiovascular disease than from their breast cancer, according to new data from the Women’s Health Initiative.
“Identification and treatment of cardiovascular disease risk factors among older women with breast cancer will likely improve survivorship and should be a high priority, especially for older women with incident localized breast cancer,” Na-Jin Park, Ph.D., said at the American Heart Association scientific sessions.
She presented an analysis that included 101,916 women who were free of cardiovascular disease and breast cancer upon enrollment in the Women’s Health Initiative (WHI) at age 50-79 years during 1993-1998. During follow-up in this prospective cohort study, 4,340 of them developed invasive breast cancer. The diagnosis occurred an average of 5 years into the study, and patients were followed for 10 years afterwards. “Forty-one percent of women with breast cancer already had cardiovascular risk factors at baseline, way before their breast cancer diagnosis,” noted Dr. Park of the University of Pittsburgh.
Among women diagnosed with breast cancer in their 50s, 40% of all deaths were from breast cancer and 15% were caused by cardiovascular disease. In contrast, the cumulative impact of atherosclerosis was far more prominent in women diagnosed with breast cancer at a more advanced age. Indeed, among women diagnosed with breast cancer in their 70s, about 15% of deaths were caused by breast cancer, while 25% resulted from cardiovascular disease.
The number of baseline cardiovascular risk factors present at enrollment in the WHI turned out to be a powerful determinant of the likelihood of acute MI or death as a result of coronary heart disease in participants who developed breast cancer.
Of the 4,340 women who later developed invasive breast cancer, 2,562 were free of hypertension, diabetes, and hypercholesterolemia and were nonsmokers upon enrollment. In an age-adjusted analysis in which this risk factor–free group served as the reference population, the risk of MI after breast cancer diagnosis was increased 1.65-fold in those with a single baseline risk factor, 3.2-fold in those with two, and 5.8-fold in women with three cardiovascular risk factors. Similarly, the breast cancer patients’ risk of CHD death climbed stepwise by 1.78-, 2.28-, and 3.6-fold as the number of baseline cardiovascular risk factors increased from one to three.
The greatest risk was seen in breast cancer patients who at WHI enrollment were current smokers with an additional cardiovascular risk factor. They had a 9.6-fold greater risk of an acute MI after developing breast cancer, compared with breast cancer patients with none of the baseline cardiovascular risk factors. They also had a 7.7-fold increased risk of CHD death.
The WHI was funded by the National Heart, Lung, and Blood Institute. Dr. Park reported having no financial conflicts of interest.
ORLANDO – Women diagnosed with localized breast cancer while in their 70s have a higher mortality from cardiovascular disease than from their breast cancer, according to new data from the Women’s Health Initiative.
“Identification and treatment of cardiovascular disease risk factors among older women with breast cancer will likely improve survivorship and should be a high priority, especially for older women with incident localized breast cancer,” Na-Jin Park, Ph.D., said at the American Heart Association scientific sessions.
She presented an analysis that included 101,916 women who were free of cardiovascular disease and breast cancer upon enrollment in the Women’s Health Initiative (WHI) at age 50-79 years during 1993-1998. During follow-up in this prospective cohort study, 4,340 of them developed invasive breast cancer. The diagnosis occurred an average of 5 years into the study, and patients were followed for 10 years afterwards. “Forty-one percent of women with breast cancer already had cardiovascular risk factors at baseline, way before their breast cancer diagnosis,” noted Dr. Park of the University of Pittsburgh.
Among women diagnosed with breast cancer in their 50s, 40% of all deaths were from breast cancer and 15% were caused by cardiovascular disease. In contrast, the cumulative impact of atherosclerosis was far more prominent in women diagnosed with breast cancer at a more advanced age. Indeed, among women diagnosed with breast cancer in their 70s, about 15% of deaths were caused by breast cancer, while 25% resulted from cardiovascular disease.
The number of baseline cardiovascular risk factors present at enrollment in the WHI turned out to be a powerful determinant of the likelihood of acute MI or death as a result of coronary heart disease in participants who developed breast cancer.
Of the 4,340 women who later developed invasive breast cancer, 2,562 were free of hypertension, diabetes, and hypercholesterolemia and were nonsmokers upon enrollment. In an age-adjusted analysis in which this risk factor–free group served as the reference population, the risk of MI after breast cancer diagnosis was increased 1.65-fold in those with a single baseline risk factor, 3.2-fold in those with two, and 5.8-fold in women with three cardiovascular risk factors. Similarly, the breast cancer patients’ risk of CHD death climbed stepwise by 1.78-, 2.28-, and 3.6-fold as the number of baseline cardiovascular risk factors increased from one to three.
The greatest risk was seen in breast cancer patients who at WHI enrollment were current smokers with an additional cardiovascular risk factor. They had a 9.6-fold greater risk of an acute MI after developing breast cancer, compared with breast cancer patients with none of the baseline cardiovascular risk factors. They also had a 7.7-fold increased risk of CHD death.
The WHI was funded by the National Heart, Lung, and Blood Institute. Dr. Park reported having no financial conflicts of interest.
ORLANDO – Women diagnosed with localized breast cancer while in their 70s have a higher mortality from cardiovascular disease than from their breast cancer, according to new data from the Women’s Health Initiative.
“Identification and treatment of cardiovascular disease risk factors among older women with breast cancer will likely improve survivorship and should be a high priority, especially for older women with incident localized breast cancer,” Na-Jin Park, Ph.D., said at the American Heart Association scientific sessions.
She presented an analysis that included 101,916 women who were free of cardiovascular disease and breast cancer upon enrollment in the Women’s Health Initiative (WHI) at age 50-79 years during 1993-1998. During follow-up in this prospective cohort study, 4,340 of them developed invasive breast cancer. The diagnosis occurred an average of 5 years into the study, and patients were followed for 10 years afterwards. “Forty-one percent of women with breast cancer already had cardiovascular risk factors at baseline, way before their breast cancer diagnosis,” noted Dr. Park of the University of Pittsburgh.
Among women diagnosed with breast cancer in their 50s, 40% of all deaths were from breast cancer and 15% were caused by cardiovascular disease. In contrast, the cumulative impact of atherosclerosis was far more prominent in women diagnosed with breast cancer at a more advanced age. Indeed, among women diagnosed with breast cancer in their 70s, about 15% of deaths were caused by breast cancer, while 25% resulted from cardiovascular disease.
The number of baseline cardiovascular risk factors present at enrollment in the WHI turned out to be a powerful determinant of the likelihood of acute MI or death as a result of coronary heart disease in participants who developed breast cancer.
Of the 4,340 women who later developed invasive breast cancer, 2,562 were free of hypertension, diabetes, and hypercholesterolemia and were nonsmokers upon enrollment. In an age-adjusted analysis in which this risk factor–free group served as the reference population, the risk of MI after breast cancer diagnosis was increased 1.65-fold in those with a single baseline risk factor, 3.2-fold in those with two, and 5.8-fold in women with three cardiovascular risk factors. Similarly, the breast cancer patients’ risk of CHD death climbed stepwise by 1.78-, 2.28-, and 3.6-fold as the number of baseline cardiovascular risk factors increased from one to three.
The greatest risk was seen in breast cancer patients who at WHI enrollment were current smokers with an additional cardiovascular risk factor. They had a 9.6-fold greater risk of an acute MI after developing breast cancer, compared with breast cancer patients with none of the baseline cardiovascular risk factors. They also had a 7.7-fold increased risk of CHD death.
The WHI was funded by the National Heart, Lung, and Blood Institute. Dr. Park reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Among women diagnosed with breast cancer in their 70s, more will die from cardiovascular disease than from their malignancy.
Major finding: Among women diagnosed with breast cancer in their 70s, only about 15% of deaths were caused by breast cancer, while 25% resulted from cardiovascular disease.
Data source: This prospective cohort analysis from the Women’s Health Initiative included 101,916 women free of cardiovascular disease and breast cancer upon enrollment, of whom 4,340 later developed invasive breast cancer.
Disclosures: The Women’s Health Initiative was funded by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts of interest.
Hold the checkpoint inhibitors when pneumonitis symptoms occur
BOSTON – Treatment of pneumonitis associated with the PD-1 axis checkpoint inhibitors requires close monitoring of patients and rapid clinical response, an oncologist advised.
“We need to be vigilant when we treat our patients. If a patient has cough or shortness of breath, you take it seriously, even if [it is] a patient who has lung cancer and is a smoker,” said Dr. Scott Gettinger of Yale Cancer Center, New Haven, Conn.
He recommended that before starting patients on a programmed death-1 (PD-1) axis inhibitor such as pembrolizumab (Keytruda) or nivolumab (Opdivo), clinicians get baseline oxygen saturation levels to obtain an objective measure for following patients during therapy.
“When you suspect pneumonitis you have to start steroids right away, or patients can spiral down,” he said at the AACR/NCI/EORTC International Conference on Molecular Targets and Cancer Therapeutics.
Pneumonitis – characterized by cough, dyspnea, and hypoxia – is a common adverse event associated with PD-1 and PD-ligand 1 (PD-L1) inhibitors, but is rarely seen in patients treated with CTLA-4 inhibitors such as ipilimumab (Yervoy). Current evidence suggests that pneumonitis is more prevalent in patients with non–small-cell lung cancer, occurring in approximately 4%-6% of patients cases, than in melanoma (1%). The difference is probably due to a history of smoking common to the majority of patients with NSCLC, Dr. Gettinger said.
“The other theme that we’re beginning to see is that maybe pneumonitis is a bit more common with anti-PD-1 vs. anti-PD-L1 antibodies,” he said.
It’s theorized that PD-1 inhibitors may block binding of PD-L2 to one of its binding partners, thereby interfering with respiratory tolerance, he said.
Evidence from the pivotal clinical trial of nivolumab in lung cancer (Checkmate 057) suggests that the time to onset of pneumonitis was a median of 31.1 weeks (range 11.7 to 56.9 weeks). The pneumonitis resolved in about 5-7 weeks.
Investigators at Memorial Sloan Kettering Cancer Center in New York City reported at the European Cancer Congress 2015 on pneumonitis in 36 of 653 patients treated with an anti PD-1/PD-L1 monoclonal antibody from 2009 through 2014, 33 of whom had received a PD-1 inhibitor, and 3 of whom received a PD-L1 inhibitor.
They found that pneumonitis in patients with lung cancer tended to resemble chronic obstructive pneumonia, whereas patients with melanoma were more likely to have ground-glass opacifications on radiography. There was a trend, falling just short of significance, toward association of COP-like pneumonitis with development of grade 3 or greater, and with a requirement for more than one type of immunosuppression, compared to other subtypes.
Algorithms for management
Dr. Gettinger briefly outlined an algorithm offered by Bristol-Myers Squibb for management of suspected pulmonary toxicity with nivolumab. For management of patients with grade 1 toxicities (asymptomatic, with radiographic changes only), it recommends that clinicians consider delay of immuno-oncologic (I-O) therapy, monitor for symptoms every 2-3 days, and consider consultations with pulmonary and infectious disease specialists.
For grade 2 pneumonitis, marked by mild to moderate new symptoms, the algorithm calls for clinicians to delay I-O therapy per protocol, consult with pulmonary and ID specialists, monitor symptoms daily and consider hospitalization, start the patient on steroids with 1.0 mg/kg per day methylprednisolone IV or the oral equivalent, and consider bronchoscopy and lung biopsy.
For patients with grade 3 or 4 toxicities, marked by severe new symptoms, new or worsening hypoxia, or other life-threatening symptoms, the algorithm states that clinicians should discontinue I-O therapy, hospitalize the patient, consult with pulmonary and ID, give 2-4 mg/kg per day methylprednisolone IV or the oral equivalent, add prophylactic antibiotics for opportunistic infections, and consider bronchoscopy and lung biopsy.
At Yale, Dr. Gettinger and colleagues, when presented with a symptomatic patient on a PD-axis inhibitor, will first rule out other etiologies such as infection, chronic obstructive pulmonary disease exacerbation, or cancer progression), typically with bronchoscopy.
For patients with moderate pneumonitis, they may treat with prednisone for 1 or 2 weeks, with a 4-6 week taper begun as symptoms start to resolve.
“In a patient who has profound hypoxia and shortness of breath, we may want to go higher: We might give them 2 mg/kg twice a day of [methylprednisolone] in the hospital, wait until they get better, and then slowly taper them, whether it be over 4 or 6 weeks or longer. Occasionally our patients need to get even higher doses of steroids and there’s really nothing to guide us. Who knows if 1 gram of [methylprednisolone] may be better than 60 g of prednisone? But we do it, and patients do get better with the higher doses,” he said.
In rare instances, patients may required other immunosuppressive agents, such as infliximab (Remicade), cyclophosphamide, or mycophenolate mofetil.
Patients who require additional immunosuppressive agents to resolve severe pneumonitis tend to have poor outcomes, Dr. Gettinger said.
Re-challenge of patients with a PD-1 or PD-L1 inhibitor following resolution of pneumonitis appears to be inadvisable for all patients except possibly those with grade 1 (asymptomatic) toxicity, due to the high risk of recurrence, he said.
BOSTON – Treatment of pneumonitis associated with the PD-1 axis checkpoint inhibitors requires close monitoring of patients and rapid clinical response, an oncologist advised.
“We need to be vigilant when we treat our patients. If a patient has cough or shortness of breath, you take it seriously, even if [it is] a patient who has lung cancer and is a smoker,” said Dr. Scott Gettinger of Yale Cancer Center, New Haven, Conn.
He recommended that before starting patients on a programmed death-1 (PD-1) axis inhibitor such as pembrolizumab (Keytruda) or nivolumab (Opdivo), clinicians get baseline oxygen saturation levels to obtain an objective measure for following patients during therapy.
“When you suspect pneumonitis you have to start steroids right away, or patients can spiral down,” he said at the AACR/NCI/EORTC International Conference on Molecular Targets and Cancer Therapeutics.
Pneumonitis – characterized by cough, dyspnea, and hypoxia – is a common adverse event associated with PD-1 and PD-ligand 1 (PD-L1) inhibitors, but is rarely seen in patients treated with CTLA-4 inhibitors such as ipilimumab (Yervoy). Current evidence suggests that pneumonitis is more prevalent in patients with non–small-cell lung cancer, occurring in approximately 4%-6% of patients cases, than in melanoma (1%). The difference is probably due to a history of smoking common to the majority of patients with NSCLC, Dr. Gettinger said.
“The other theme that we’re beginning to see is that maybe pneumonitis is a bit more common with anti-PD-1 vs. anti-PD-L1 antibodies,” he said.
It’s theorized that PD-1 inhibitors may block binding of PD-L2 to one of its binding partners, thereby interfering with respiratory tolerance, he said.
Evidence from the pivotal clinical trial of nivolumab in lung cancer (Checkmate 057) suggests that the time to onset of pneumonitis was a median of 31.1 weeks (range 11.7 to 56.9 weeks). The pneumonitis resolved in about 5-7 weeks.
Investigators at Memorial Sloan Kettering Cancer Center in New York City reported at the European Cancer Congress 2015 on pneumonitis in 36 of 653 patients treated with an anti PD-1/PD-L1 monoclonal antibody from 2009 through 2014, 33 of whom had received a PD-1 inhibitor, and 3 of whom received a PD-L1 inhibitor.
They found that pneumonitis in patients with lung cancer tended to resemble chronic obstructive pneumonia, whereas patients with melanoma were more likely to have ground-glass opacifications on radiography. There was a trend, falling just short of significance, toward association of COP-like pneumonitis with development of grade 3 or greater, and with a requirement for more than one type of immunosuppression, compared to other subtypes.
Algorithms for management
Dr. Gettinger briefly outlined an algorithm offered by Bristol-Myers Squibb for management of suspected pulmonary toxicity with nivolumab. For management of patients with grade 1 toxicities (asymptomatic, with radiographic changes only), it recommends that clinicians consider delay of immuno-oncologic (I-O) therapy, monitor for symptoms every 2-3 days, and consider consultations with pulmonary and infectious disease specialists.
For grade 2 pneumonitis, marked by mild to moderate new symptoms, the algorithm calls for clinicians to delay I-O therapy per protocol, consult with pulmonary and ID specialists, monitor symptoms daily and consider hospitalization, start the patient on steroids with 1.0 mg/kg per day methylprednisolone IV or the oral equivalent, and consider bronchoscopy and lung biopsy.
For patients with grade 3 or 4 toxicities, marked by severe new symptoms, new or worsening hypoxia, or other life-threatening symptoms, the algorithm states that clinicians should discontinue I-O therapy, hospitalize the patient, consult with pulmonary and ID, give 2-4 mg/kg per day methylprednisolone IV or the oral equivalent, add prophylactic antibiotics for opportunistic infections, and consider bronchoscopy and lung biopsy.
At Yale, Dr. Gettinger and colleagues, when presented with a symptomatic patient on a PD-axis inhibitor, will first rule out other etiologies such as infection, chronic obstructive pulmonary disease exacerbation, or cancer progression), typically with bronchoscopy.
For patients with moderate pneumonitis, they may treat with prednisone for 1 or 2 weeks, with a 4-6 week taper begun as symptoms start to resolve.
“In a patient who has profound hypoxia and shortness of breath, we may want to go higher: We might give them 2 mg/kg twice a day of [methylprednisolone] in the hospital, wait until they get better, and then slowly taper them, whether it be over 4 or 6 weeks or longer. Occasionally our patients need to get even higher doses of steroids and there’s really nothing to guide us. Who knows if 1 gram of [methylprednisolone] may be better than 60 g of prednisone? But we do it, and patients do get better with the higher doses,” he said.
In rare instances, patients may required other immunosuppressive agents, such as infliximab (Remicade), cyclophosphamide, or mycophenolate mofetil.
Patients who require additional immunosuppressive agents to resolve severe pneumonitis tend to have poor outcomes, Dr. Gettinger said.
Re-challenge of patients with a PD-1 or PD-L1 inhibitor following resolution of pneumonitis appears to be inadvisable for all patients except possibly those with grade 1 (asymptomatic) toxicity, due to the high risk of recurrence, he said.
BOSTON – Treatment of pneumonitis associated with the PD-1 axis checkpoint inhibitors requires close monitoring of patients and rapid clinical response, an oncologist advised.
“We need to be vigilant when we treat our patients. If a patient has cough or shortness of breath, you take it seriously, even if [it is] a patient who has lung cancer and is a smoker,” said Dr. Scott Gettinger of Yale Cancer Center, New Haven, Conn.
He recommended that before starting patients on a programmed death-1 (PD-1) axis inhibitor such as pembrolizumab (Keytruda) or nivolumab (Opdivo), clinicians get baseline oxygen saturation levels to obtain an objective measure for following patients during therapy.
“When you suspect pneumonitis you have to start steroids right away, or patients can spiral down,” he said at the AACR/NCI/EORTC International Conference on Molecular Targets and Cancer Therapeutics.
Pneumonitis – characterized by cough, dyspnea, and hypoxia – is a common adverse event associated with PD-1 and PD-ligand 1 (PD-L1) inhibitors, but is rarely seen in patients treated with CTLA-4 inhibitors such as ipilimumab (Yervoy). Current evidence suggests that pneumonitis is more prevalent in patients with non–small-cell lung cancer, occurring in approximately 4%-6% of patients cases, than in melanoma (1%). The difference is probably due to a history of smoking common to the majority of patients with NSCLC, Dr. Gettinger said.
“The other theme that we’re beginning to see is that maybe pneumonitis is a bit more common with anti-PD-1 vs. anti-PD-L1 antibodies,” he said.
It’s theorized that PD-1 inhibitors may block binding of PD-L2 to one of its binding partners, thereby interfering with respiratory tolerance, he said.
Evidence from the pivotal clinical trial of nivolumab in lung cancer (Checkmate 057) suggests that the time to onset of pneumonitis was a median of 31.1 weeks (range 11.7 to 56.9 weeks). The pneumonitis resolved in about 5-7 weeks.
Investigators at Memorial Sloan Kettering Cancer Center in New York City reported at the European Cancer Congress 2015 on pneumonitis in 36 of 653 patients treated with an anti PD-1/PD-L1 monoclonal antibody from 2009 through 2014, 33 of whom had received a PD-1 inhibitor, and 3 of whom received a PD-L1 inhibitor.
They found that pneumonitis in patients with lung cancer tended to resemble chronic obstructive pneumonia, whereas patients with melanoma were more likely to have ground-glass opacifications on radiography. There was a trend, falling just short of significance, toward association of COP-like pneumonitis with development of grade 3 or greater, and with a requirement for more than one type of immunosuppression, compared to other subtypes.
Algorithms for management
Dr. Gettinger briefly outlined an algorithm offered by Bristol-Myers Squibb for management of suspected pulmonary toxicity with nivolumab. For management of patients with grade 1 toxicities (asymptomatic, with radiographic changes only), it recommends that clinicians consider delay of immuno-oncologic (I-O) therapy, monitor for symptoms every 2-3 days, and consider consultations with pulmonary and infectious disease specialists.
For grade 2 pneumonitis, marked by mild to moderate new symptoms, the algorithm calls for clinicians to delay I-O therapy per protocol, consult with pulmonary and ID specialists, monitor symptoms daily and consider hospitalization, start the patient on steroids with 1.0 mg/kg per day methylprednisolone IV or the oral equivalent, and consider bronchoscopy and lung biopsy.
For patients with grade 3 or 4 toxicities, marked by severe new symptoms, new or worsening hypoxia, or other life-threatening symptoms, the algorithm states that clinicians should discontinue I-O therapy, hospitalize the patient, consult with pulmonary and ID, give 2-4 mg/kg per day methylprednisolone IV or the oral equivalent, add prophylactic antibiotics for opportunistic infections, and consider bronchoscopy and lung biopsy.
At Yale, Dr. Gettinger and colleagues, when presented with a symptomatic patient on a PD-axis inhibitor, will first rule out other etiologies such as infection, chronic obstructive pulmonary disease exacerbation, or cancer progression), typically with bronchoscopy.
For patients with moderate pneumonitis, they may treat with prednisone for 1 or 2 weeks, with a 4-6 week taper begun as symptoms start to resolve.
“In a patient who has profound hypoxia and shortness of breath, we may want to go higher: We might give them 2 mg/kg twice a day of [methylprednisolone] in the hospital, wait until they get better, and then slowly taper them, whether it be over 4 or 6 weeks or longer. Occasionally our patients need to get even higher doses of steroids and there’s really nothing to guide us. Who knows if 1 gram of [methylprednisolone] may be better than 60 g of prednisone? But we do it, and patients do get better with the higher doses,” he said.
In rare instances, patients may required other immunosuppressive agents, such as infliximab (Remicade), cyclophosphamide, or mycophenolate mofetil.
Patients who require additional immunosuppressive agents to resolve severe pneumonitis tend to have poor outcomes, Dr. Gettinger said.
Re-challenge of patients with a PD-1 or PD-L1 inhibitor following resolution of pneumonitis appears to be inadvisable for all patients except possibly those with grade 1 (asymptomatic) toxicity, due to the high risk of recurrence, he said.
AT AACR–NCI–EORTC
Key clinical point: Pneumonitis is an uncommon but potentially serious adverse event associated with PD-1/PD-L1 checkpoint inhibitor therapy.
Major finding: Pneumonitis occurs in about 4%-6% of patients with non–small-cell lung cancer treated with a PD-1 axis inhibitor.
Data source: Review of current understanding of pneumonitis associated with anti-PD-1/PD-L1 therapeutic agents.
Disclosures: Dr. Gettinger disclosed serving as a consultant for BMS.