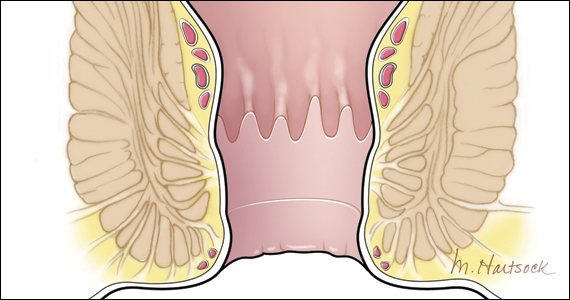
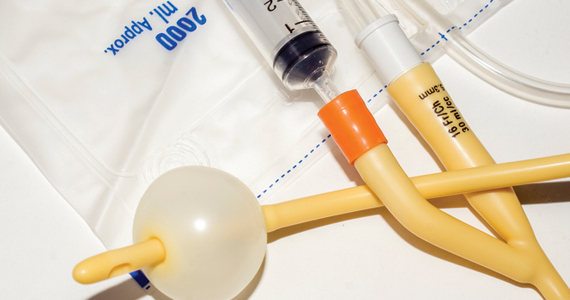
User login
‘Reassuring’ data on COVID-19 vaccines in pregnancy
Pregnant women can safely get vaccinated with the Pfizer-BioNTech and Moderna vaccines for COVID-19, surveillance data from the Centers for Disease Control and Prevention suggest.
More than 30,000 women who received these vaccines have reported pregnancies through the CDC’s V-Safe voluntary reporting system, and their rates of complications are not significantly different from those of unvaccinated pregnant women, said Tom Shimabukuro, MD, MPH, MBA, deputy director of the CDC Immunization Safety Office.
“Overall, the data are reassuring with respect to vaccine safety in pregnant women,” he told this news organization.
Dr. Shimabukuro presented the data during a March 1 meeting of the Advisory Committee on Immunization Practices, a group of health experts selected by the Secretary of the U.S. Department of Health & Human Services.
The CDC has included pregnancy along with other underlying conditions that qualify people to be offered vaccines in the third priority tier (Phase 1c).
“There is evidence that pregnant women who get COVID-19 are at increased risk of severe illness and complications from severe illness,” Dr. Shimabukuro explained. “And there is also evidence that pregnant persons who get COVID-19 may be at increased risk for adverse pregnancy outcomes.”
The American College of Obstetrics and Gynecology recommends that “COVID-19 vaccines should not be withheld from pregnant individuals.”
By contrast, the World Health Organization recommends the vaccines only for those pregnant women who are “at high risk of exposure to SARS-CoV-2 (for example, health workers) or who have comorbidities which add to their risk of severe disease.”
Not enough information was available from the pivotal trials of the Moderna and Pfizer vaccines to assess risk in pregnant women, according to these manufacturers. Pfizer has announced a follow-up trial of its vaccine in healthy pregnant women.
Analyzing surveillance data
To better assess whether the Pfizer or Moderna vaccines cause problems in pregnancy or childbirth, Dr. Shimabukuro and colleagues analyzed data from V-Safe and the Vaccine Adverse Event Reporting System (VAERS).
The CDC encourages providers to inform people they vaccinate about the V-Safe program. Participants can voluntarily enter their data through a website, and may receive follow-up text messages and phone calls from the CDC asking for additional information at various times after vaccination. It is not a systematic survey, and the sample is not necessarily representative of everyone who gets the vaccine, Dr. Shimabukuro noted.
At the time of the study, V-Safe recorded 55,220,364 reports from people who received at least one dose of the Pfizer or Moderna vaccine through Feb. 16. These included 30,494 pregnancies, of which 16,039 were in women who received the Pfizer vaccine and 14,455 in women who received the Moderna vaccine.
Analyzing data collected through Jan. 13, 2021, the researchers found that both local and systemic reactions were similar between pregnant and nonpregnant women aged 16-54 years.
Most women reported pain, and some reported swelling, redness, and itching at the injection site. Of systemic reactions, fatigue was the most common, followed by headache, myalgia, chills, nausea, and fever. The systemic reactions were more common with the second Pfizer dose; fatigue affected a majority of both pregnant and nonpregnant women. Data on the second Moderna dose were not available.
The CDC enrolled 1,815 pregnant women for additional follow-up, among whom there were 275 completed pregnancies and 232 live births.
Rates of outcomes “of interest” were no higher among these women than in the general population. 
In contrast to V-Safe, data from VAERS, comanaged by the CDC and U.S. Food and Drug Administration, are from spontaneous reports of adverse events. The sources for those reports are varied. “That could be the health care provider,” Dr. Shimabukuro said. “That could be the patient themselves. It could be a caregiver for children.”
Just 154 VAERS reports through Feb. 16 concerned pregnant women, and of these, only 42 (27%) were for pregnancy-specific conditions, with the other 73% representing the types of adverse events reported for the general population of vaccinated people, such as headache and fatigue.
Of the 42 pregnancy-related events, there were 29 spontaneous abortions or miscarriages, with the remainder divided among 10 other pregnancy and neonatal conditions.
“When we looked at those outcomes and we compared the reporting rates, based on known background rates of these conditions, we did not see anything unexpected or concerning with respect to pregnancy or neonatal-specific conditions,” Dr. Shimabukuro said about the VAERS data.
The CDC did not collect data on fertility. “We’ve done a lot of work with other vaccines,” said Dr. Shimabukuro. “And just from a biological basis, we don’t have any evidence that vaccination, just in general, causes fertility problems.”
Also, Dr. Shimabukuro noted that the COVID-19 vaccine made by Janssen/Johnson & Johnson did not receive emergency authorization from the FDA in time to be included in the current report, but is being tracked for future reports.
Vaccination could benefit infants
In addition to the new safety data, experts continue to remind clinicians and the public that vaccination during pregnancy could benefit offspring. The unborn babies of pregnant women who receive the COVID-19 vaccine could be protected from the virus for the first several months of their lives, said White House COVID-19 czar Anthony Fauci, MD, at a briefing on March 10.
“We’ve seen this with many other vaccines,” Dr. Fauci said. “That’s a very good way you can get protection for the mother during pregnancy and also a transfer of protection for the infant, which will last a few months following the birth.”
Dr. Fauci also noted that the same vaccine platform used in Johnson & Johnson’s COVID-19 vaccine was successfully used for Ebola in pregnant women in Africa.
Dr. Shimabukuro has reported no relevant financial relationships.
Lindsay Kalter contributed to the reporting for this story.
A version of this article first appeared on Medscape.com.
Pregnant women can safely get vaccinated with the Pfizer-BioNTech and Moderna vaccines for COVID-19, surveillance data from the Centers for Disease Control and Prevention suggest.
More than 30,000 women who received these vaccines have reported pregnancies through the CDC’s V-Safe voluntary reporting system, and their rates of complications are not significantly different from those of unvaccinated pregnant women, said Tom Shimabukuro, MD, MPH, MBA, deputy director of the CDC Immunization Safety Office.
“Overall, the data are reassuring with respect to vaccine safety in pregnant women,” he told this news organization.
Dr. Shimabukuro presented the data during a March 1 meeting of the Advisory Committee on Immunization Practices, a group of health experts selected by the Secretary of the U.S. Department of Health & Human Services.
The CDC has included pregnancy along with other underlying conditions that qualify people to be offered vaccines in the third priority tier (Phase 1c).
“There is evidence that pregnant women who get COVID-19 are at increased risk of severe illness and complications from severe illness,” Dr. Shimabukuro explained. “And there is also evidence that pregnant persons who get COVID-19 may be at increased risk for adverse pregnancy outcomes.”
The American College of Obstetrics and Gynecology recommends that “COVID-19 vaccines should not be withheld from pregnant individuals.”
By contrast, the World Health Organization recommends the vaccines only for those pregnant women who are “at high risk of exposure to SARS-CoV-2 (for example, health workers) or who have comorbidities which add to their risk of severe disease.”
Not enough information was available from the pivotal trials of the Moderna and Pfizer vaccines to assess risk in pregnant women, according to these manufacturers. Pfizer has announced a follow-up trial of its vaccine in healthy pregnant women.
Analyzing surveillance data
To better assess whether the Pfizer or Moderna vaccines cause problems in pregnancy or childbirth, Dr. Shimabukuro and colleagues analyzed data from V-Safe and the Vaccine Adverse Event Reporting System (VAERS).
The CDC encourages providers to inform people they vaccinate about the V-Safe program. Participants can voluntarily enter their data through a website, and may receive follow-up text messages and phone calls from the CDC asking for additional information at various times after vaccination. It is not a systematic survey, and the sample is not necessarily representative of everyone who gets the vaccine, Dr. Shimabukuro noted.
At the time of the study, V-Safe recorded 55,220,364 reports from people who received at least one dose of the Pfizer or Moderna vaccine through Feb. 16. These included 30,494 pregnancies, of which 16,039 were in women who received the Pfizer vaccine and 14,455 in women who received the Moderna vaccine.
Analyzing data collected through Jan. 13, 2021, the researchers found that both local and systemic reactions were similar between pregnant and nonpregnant women aged 16-54 years.
Most women reported pain, and some reported swelling, redness, and itching at the injection site. Of systemic reactions, fatigue was the most common, followed by headache, myalgia, chills, nausea, and fever. The systemic reactions were more common with the second Pfizer dose; fatigue affected a majority of both pregnant and nonpregnant women. Data on the second Moderna dose were not available.
The CDC enrolled 1,815 pregnant women for additional follow-up, among whom there were 275 completed pregnancies and 232 live births.
Rates of outcomes “of interest” were no higher among these women than in the general population. 
In contrast to V-Safe, data from VAERS, comanaged by the CDC and U.S. Food and Drug Administration, are from spontaneous reports of adverse events. The sources for those reports are varied. “That could be the health care provider,” Dr. Shimabukuro said. “That could be the patient themselves. It could be a caregiver for children.”
Just 154 VAERS reports through Feb. 16 concerned pregnant women, and of these, only 42 (27%) were for pregnancy-specific conditions, with the other 73% representing the types of adverse events reported for the general population of vaccinated people, such as headache and fatigue.
Of the 42 pregnancy-related events, there were 29 spontaneous abortions or miscarriages, with the remainder divided among 10 other pregnancy and neonatal conditions.
“When we looked at those outcomes and we compared the reporting rates, based on known background rates of these conditions, we did not see anything unexpected or concerning with respect to pregnancy or neonatal-specific conditions,” Dr. Shimabukuro said about the VAERS data.
The CDC did not collect data on fertility. “We’ve done a lot of work with other vaccines,” said Dr. Shimabukuro. “And just from a biological basis, we don’t have any evidence that vaccination, just in general, causes fertility problems.”
Also, Dr. Shimabukuro noted that the COVID-19 vaccine made by Janssen/Johnson & Johnson did not receive emergency authorization from the FDA in time to be included in the current report, but is being tracked for future reports.
Vaccination could benefit infants
In addition to the new safety data, experts continue to remind clinicians and the public that vaccination during pregnancy could benefit offspring. The unborn babies of pregnant women who receive the COVID-19 vaccine could be protected from the virus for the first several months of their lives, said White House COVID-19 czar Anthony Fauci, MD, at a briefing on March 10.
“We’ve seen this with many other vaccines,” Dr. Fauci said. “That’s a very good way you can get protection for the mother during pregnancy and also a transfer of protection for the infant, which will last a few months following the birth.”
Dr. Fauci also noted that the same vaccine platform used in Johnson & Johnson’s COVID-19 vaccine was successfully used for Ebola in pregnant women in Africa.
Dr. Shimabukuro has reported no relevant financial relationships.
Lindsay Kalter contributed to the reporting for this story.
A version of this article first appeared on Medscape.com.
Pregnant women can safely get vaccinated with the Pfizer-BioNTech and Moderna vaccines for COVID-19, surveillance data from the Centers for Disease Control and Prevention suggest.
More than 30,000 women who received these vaccines have reported pregnancies through the CDC’s V-Safe voluntary reporting system, and their rates of complications are not significantly different from those of unvaccinated pregnant women, said Tom Shimabukuro, MD, MPH, MBA, deputy director of the CDC Immunization Safety Office.
“Overall, the data are reassuring with respect to vaccine safety in pregnant women,” he told this news organization.
Dr. Shimabukuro presented the data during a March 1 meeting of the Advisory Committee on Immunization Practices, a group of health experts selected by the Secretary of the U.S. Department of Health & Human Services.
The CDC has included pregnancy along with other underlying conditions that qualify people to be offered vaccines in the third priority tier (Phase 1c).
“There is evidence that pregnant women who get COVID-19 are at increased risk of severe illness and complications from severe illness,” Dr. Shimabukuro explained. “And there is also evidence that pregnant persons who get COVID-19 may be at increased risk for adverse pregnancy outcomes.”
The American College of Obstetrics and Gynecology recommends that “COVID-19 vaccines should not be withheld from pregnant individuals.”
By contrast, the World Health Organization recommends the vaccines only for those pregnant women who are “at high risk of exposure to SARS-CoV-2 (for example, health workers) or who have comorbidities which add to their risk of severe disease.”
Not enough information was available from the pivotal trials of the Moderna and Pfizer vaccines to assess risk in pregnant women, according to these manufacturers. Pfizer has announced a follow-up trial of its vaccine in healthy pregnant women.
Analyzing surveillance data
To better assess whether the Pfizer or Moderna vaccines cause problems in pregnancy or childbirth, Dr. Shimabukuro and colleagues analyzed data from V-Safe and the Vaccine Adverse Event Reporting System (VAERS).
The CDC encourages providers to inform people they vaccinate about the V-Safe program. Participants can voluntarily enter their data through a website, and may receive follow-up text messages and phone calls from the CDC asking for additional information at various times after vaccination. It is not a systematic survey, and the sample is not necessarily representative of everyone who gets the vaccine, Dr. Shimabukuro noted.
At the time of the study, V-Safe recorded 55,220,364 reports from people who received at least one dose of the Pfizer or Moderna vaccine through Feb. 16. These included 30,494 pregnancies, of which 16,039 were in women who received the Pfizer vaccine and 14,455 in women who received the Moderna vaccine.
Analyzing data collected through Jan. 13, 2021, the researchers found that both local and systemic reactions were similar between pregnant and nonpregnant women aged 16-54 years.
Most women reported pain, and some reported swelling, redness, and itching at the injection site. Of systemic reactions, fatigue was the most common, followed by headache, myalgia, chills, nausea, and fever. The systemic reactions were more common with the second Pfizer dose; fatigue affected a majority of both pregnant and nonpregnant women. Data on the second Moderna dose were not available.
The CDC enrolled 1,815 pregnant women for additional follow-up, among whom there were 275 completed pregnancies and 232 live births.
Rates of outcomes “of interest” were no higher among these women than in the general population. 
In contrast to V-Safe, data from VAERS, comanaged by the CDC and U.S. Food and Drug Administration, are from spontaneous reports of adverse events. The sources for those reports are varied. “That could be the health care provider,” Dr. Shimabukuro said. “That could be the patient themselves. It could be a caregiver for children.”
Just 154 VAERS reports through Feb. 16 concerned pregnant women, and of these, only 42 (27%) were for pregnancy-specific conditions, with the other 73% representing the types of adverse events reported for the general population of vaccinated people, such as headache and fatigue.
Of the 42 pregnancy-related events, there were 29 spontaneous abortions or miscarriages, with the remainder divided among 10 other pregnancy and neonatal conditions.
“When we looked at those outcomes and we compared the reporting rates, based on known background rates of these conditions, we did not see anything unexpected or concerning with respect to pregnancy or neonatal-specific conditions,” Dr. Shimabukuro said about the VAERS data.
The CDC did not collect data on fertility. “We’ve done a lot of work with other vaccines,” said Dr. Shimabukuro. “And just from a biological basis, we don’t have any evidence that vaccination, just in general, causes fertility problems.”
Also, Dr. Shimabukuro noted that the COVID-19 vaccine made by Janssen/Johnson & Johnson did not receive emergency authorization from the FDA in time to be included in the current report, but is being tracked for future reports.
Vaccination could benefit infants
In addition to the new safety data, experts continue to remind clinicians and the public that vaccination during pregnancy could benefit offspring. The unborn babies of pregnant women who receive the COVID-19 vaccine could be protected from the virus for the first several months of their lives, said White House COVID-19 czar Anthony Fauci, MD, at a briefing on March 10.
“We’ve seen this with many other vaccines,” Dr. Fauci said. “That’s a very good way you can get protection for the mother during pregnancy and also a transfer of protection for the infant, which will last a few months following the birth.”
Dr. Fauci also noted that the same vaccine platform used in Johnson & Johnson’s COVID-19 vaccine was successfully used for Ebola in pregnant women in Africa.
Dr. Shimabukuro has reported no relevant financial relationships.
Lindsay Kalter contributed to the reporting for this story.
A version of this article first appeared on Medscape.com.
Baby born to partially vaccinated mom has COVID-19 antibodies
A baby girl who was born 3 weeks after her mom got the first dose of the Moderna COVID-19 vaccine has antibodies against the coronavirus, according to a preprint paper published on the medRxiv server Feb. 5. The paper hasn’t yet been peer reviewed.
The mom, a health care worker in Florida, developed COVID-19 antibodies after she received the shot. Testing showed that the antibodies passed through the placenta to the baby.
“Maternal vaccination for influenza and TDaP have been well studied in terms of safety and efficacy for protection of the newborn by placental passage of antibodies,” Paul Gilbert, MD, and Chad Rudnick, MD, pediatricians and researchers at Florida Atlantic University, wrote in the paper.
Previous research has indicated that moms who have recovered from COVID-19 can deliver babies with antibodies, according to Insider, but this may be the first report that shows how vaccination during pregnancy can provide antibodies as well.
Dr. Gilbert and Dr. Rudnick said they were fortunate to connect with the mom in Boca Raton. She hadn’t contracted COVID-19 and was able to get the vaccine at the end of her pregnancy in January. When the baby was born, they were able to test the cord blood to look for antibodies specifically from the vaccine.
“We were very excited to see, once the test result came back, that the antibodies from the mom’s vaccine did in fact pass through the placenta to the newborn,” Dr. Rudnick told WPTV, an NBC affiliate in West Palm Beach.
“We knew that we were going to be potentially one of the first in the world to report it, and that opportunity probably only comes once in a career,” Dr. Gilbert told WPTV.
In the preprint, Dr. Gilbert and Dr. Rudnick said a “vigorous, healthy, full-term” baby was born, and the mom received the second dose of the Moderna vaccine during the postpartum period. The newborn received a normal “well-infant” evaluation and was breastfeeding.
The two doctors called for a “significant and urgent need” to research the safety and efficacy of COVID-19 vaccines during pregnancy. They also encouraged other researchers to create pregnancy and breastfeeding registries to study COVID-19 vaccines in pregnant and breastfeeding moms and newborns.
Dr. Gilbert and Dr. Rudnick are now preparing their research for publication and hope future studies will investigate the amount and length of antibody response in newborns.
“Total antibody measurements may be used to determine how long protection is expected, which may help to determine when the best time would be to begin vaccination,” they wrote.
A version of this article first appeared on Medscape.com.
A baby girl who was born 3 weeks after her mom got the first dose of the Moderna COVID-19 vaccine has antibodies against the coronavirus, according to a preprint paper published on the medRxiv server Feb. 5. The paper hasn’t yet been peer reviewed.
The mom, a health care worker in Florida, developed COVID-19 antibodies after she received the shot. Testing showed that the antibodies passed through the placenta to the baby.
“Maternal vaccination for influenza and TDaP have been well studied in terms of safety and efficacy for protection of the newborn by placental passage of antibodies,” Paul Gilbert, MD, and Chad Rudnick, MD, pediatricians and researchers at Florida Atlantic University, wrote in the paper.
Previous research has indicated that moms who have recovered from COVID-19 can deliver babies with antibodies, according to Insider, but this may be the first report that shows how vaccination during pregnancy can provide antibodies as well.
Dr. Gilbert and Dr. Rudnick said they were fortunate to connect with the mom in Boca Raton. She hadn’t contracted COVID-19 and was able to get the vaccine at the end of her pregnancy in January. When the baby was born, they were able to test the cord blood to look for antibodies specifically from the vaccine.
“We were very excited to see, once the test result came back, that the antibodies from the mom’s vaccine did in fact pass through the placenta to the newborn,” Dr. Rudnick told WPTV, an NBC affiliate in West Palm Beach.
“We knew that we were going to be potentially one of the first in the world to report it, and that opportunity probably only comes once in a career,” Dr. Gilbert told WPTV.
In the preprint, Dr. Gilbert and Dr. Rudnick said a “vigorous, healthy, full-term” baby was born, and the mom received the second dose of the Moderna vaccine during the postpartum period. The newborn received a normal “well-infant” evaluation and was breastfeeding.
The two doctors called for a “significant and urgent need” to research the safety and efficacy of COVID-19 vaccines during pregnancy. They also encouraged other researchers to create pregnancy and breastfeeding registries to study COVID-19 vaccines in pregnant and breastfeeding moms and newborns.
Dr. Gilbert and Dr. Rudnick are now preparing their research for publication and hope future studies will investigate the amount and length of antibody response in newborns.
“Total antibody measurements may be used to determine how long protection is expected, which may help to determine when the best time would be to begin vaccination,” they wrote.
A version of this article first appeared on Medscape.com.
A baby girl who was born 3 weeks after her mom got the first dose of the Moderna COVID-19 vaccine has antibodies against the coronavirus, according to a preprint paper published on the medRxiv server Feb. 5. The paper hasn’t yet been peer reviewed.
The mom, a health care worker in Florida, developed COVID-19 antibodies after she received the shot. Testing showed that the antibodies passed through the placenta to the baby.
“Maternal vaccination for influenza and TDaP have been well studied in terms of safety and efficacy for protection of the newborn by placental passage of antibodies,” Paul Gilbert, MD, and Chad Rudnick, MD, pediatricians and researchers at Florida Atlantic University, wrote in the paper.
Previous research has indicated that moms who have recovered from COVID-19 can deliver babies with antibodies, according to Insider, but this may be the first report that shows how vaccination during pregnancy can provide antibodies as well.
Dr. Gilbert and Dr. Rudnick said they were fortunate to connect with the mom in Boca Raton. She hadn’t contracted COVID-19 and was able to get the vaccine at the end of her pregnancy in January. When the baby was born, they were able to test the cord blood to look for antibodies specifically from the vaccine.
“We were very excited to see, once the test result came back, that the antibodies from the mom’s vaccine did in fact pass through the placenta to the newborn,” Dr. Rudnick told WPTV, an NBC affiliate in West Palm Beach.
“We knew that we were going to be potentially one of the first in the world to report it, and that opportunity probably only comes once in a career,” Dr. Gilbert told WPTV.
In the preprint, Dr. Gilbert and Dr. Rudnick said a “vigorous, healthy, full-term” baby was born, and the mom received the second dose of the Moderna vaccine during the postpartum period. The newborn received a normal “well-infant” evaluation and was breastfeeding.
The two doctors called for a “significant and urgent need” to research the safety and efficacy of COVID-19 vaccines during pregnancy. They also encouraged other researchers to create pregnancy and breastfeeding registries to study COVID-19 vaccines in pregnant and breastfeeding moms and newborns.
Dr. Gilbert and Dr. Rudnick are now preparing their research for publication and hope future studies will investigate the amount and length of antibody response in newborns.
“Total antibody measurements may be used to determine how long protection is expected, which may help to determine when the best time would be to begin vaccination,” they wrote.
A version of this article first appeared on Medscape.com.
Pregnant patients with severe COVID-19 disease at increased risk of complications
Pregnant patients with COVID-19 infections were more likely to experience severe disease if they had preexisting comorbidities, such as chronic hypertension, asthma, or pregestational diabetes, according to findings from a new study presented at the meeting sponsored by the Society for Maternal-Fetal Medicine.
The study included outcomes for the largest multistate cohort of pregnant patients with COVID-19 outside of what the Centers for Disease Control and Prevention is tracking. Its findings also mirrored those of a multicenter, retrospective study in Washington state, published in the American Journal of Obstetrics & Gynecology. That study also found that pregnant patients hospitalized for COVID-19 were more likely to have comorbidities, and both studies found an increased likelihood of preterm birth among pregnant patients with severe or critical disease.
Disease severity linked to risk of perinatal complications
In the abstract presented at the SMFM meeting, more severe disease was associated with older age and a higher median body mass index, as seen in the general population, but the researchers found no differences in disease severity occurred by race or ethnicity, Torri D. Metz, MD, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, told attendees of the conference. The researchers also found that perinatal complications were more prevalent in those with severe or critical COVID-19 disease but not in those with mild or moderate disease. Vertical COVID-19 transmission from mother to child was rare.
The observational study included all patients who had a singleton pregnancy, had a positive SARS-CoV-2 test, and delivered between March 1 and July 31, 2020, at one of the 33 U.S. hospitals in the NICHD Maternal-Fetal Medicine Units Network, spread across 14 states. The researchers used electronic medical records to determine incidence of cesarean delivery, postpartum hemorrhage, hypertensive disorders of pregnancy, preterm birth (less than 37 weeks), maternal death, infant death, and positive infant COVID-19 test. They tracked mothers through 6 weeks post partum and newborns through delivery hospitalization.
Of 1,291 patients in the cohort, 1,219 received their first positive COVID-19 test during pregnancy. The others tested positive while in the hospital for delivery or within a month and a half after discharge. Limiting their analysis to those who developed COVID-19 while pregnant prior to delivery, nearly half (47%) were asymptomatic.
The disease was mild in 27%, moderate in 14%, severe in 8%, and critical in 4%. The researchers used the National Institutes of Health classifications for severity and included deaths in the critical group. The most common symptom was a cough, reported by a third of the patients (34%). Four of six maternal deaths that occurred were caused by COVID-19.
Compared with an average age of 28 in those without symptoms, the mean age was 29 in those with mild/moderate disease and 30 in those with severe/critical disease (P = .006). Similarly, the mean BMI was 28.3 in asymptomatic patients, 29 in those with mild/moderate disease, and 32.3 in those with severe/critical disease (P < .001). Despite a diverse cohort – 53% Hispanic, 23% Black, and 15% White – the researches found no racial/ethnic trends in disease severity.
Patients who had asthma, chronic obstructive pulmonary disorder, pregestational diabetes, chronic hypertension, chronic liver disease, or a seizure disorder were all significantly more likely to have critical/severe disease than mild/moderate disease, and more likely to have mild/moderate disease than asymptomatic (P values ranged from < .001 to .02).
The mothers with critical or severe illness were 1.6 times more likely to have cesarean births and to have hypertensive disorders of pregnancy, and they were twice as likely to have postpartum hemorrhage (P < .001; P = .007). Those with mild or moderate disease, however, had no increased risks for perinatal complications over asymptomatic patients.
Critical or severe illness was also associated with more than triple the risk of preterm birth (adjusted risk ratio, 3.6; P < .001). Newborns of mothers with critical or severe illness also had three times greater risk of neonatal ICU admission (ARR, 3.1; P <. 001) and weighed an average 385 g less than newborns of asymptomatic mothers. COVID-19 rate among infants was only 1% during delivery hospitalization.
Since the study cutoff was July 30 and COVID infections only became prevalent in March, the researchers were unable to evaluate women for outcomes resulting from COVID infections in early pregnancy, such as congenital anomalies or early miscarriage, Dr. Metz said. In addition, since many of the sites are urban centers, the data may not be generalizable to rural areas.
Peter S. Bernstein, MD, MPH, of Montefiore Medical Center, New York, asked whether the increased cesarean deliveries and preterm births in the group of women with severe disease were caused by usual obstetric causes or the treatment of COVID-19 infection. Dr. Metz said the vast majority of preterm deliveries were indicated, but only a small proportion were induced for COVID-19 alone. “A lot had hypertensive disorders of pregnancies or PPROM, so it’s partly driven by the infection itself but also partly driven by some of those perinatal complications,” she said.
Similar findings in Washington
In the Washington study, among 240 pregnant patients with confirmed COVID-19 infection between March 1 and July 30, 2020, 1 in 11 developed severe or critical disease, and 1 in 10 were hospitalized. The pregnant patients had more than triple the risk of hospitalization compared with adults of similar ages in the general population (10% vs. 2.8%; rate ratio, 3.5). Similar to the multistate NICHD study, women were more likely to be hospitalized if they had asthma, hypertension, type 2 diabetes, autoimmune disease, or class III obesity.
Three mothers died of COVID-19, resulting in a case fatality rate 13.6 times greater than nonpregnant patients with COVID-19 in the general population. The absolute difference in the rate was 1.2%. As seen in the NICHD study, preterm birth was more common in mothers with severe or critical COVID-19. Nearly half (45.4%) of mothers with severe or critical COVID-19 delivered preterm compared to 5.2% in those with mild COVID-19 (P < .001).
“Our finding that deaths in pregnant patients contributed disproportionately to deaths from COVID-19 among 20- to 39-year-olds in Washington state is similar to what was observed during the influenza A virus H1N1 2009 pandemic,” Erica M. Lokken, PhD, MS, of the departments of global health and ob.gyn. at the University of Washington, Seattle, and colleagues wrote in the Washington study. But they noted that it took 8 months into the pandemic before pregnant patients were identified as a high-risk group for COVID-19.
“Given the similarity in clinical course between COVID-19 and IAV H1N1 2009 with an increased risk for mortality during pregnancy and the postpartum period, we strongly recommend that pregnant patients should be considered a high-risk population to novel highly pathogenic respiratory viruses until proven otherwise by population-based studies with good ascertainment of pregnancy status,” they wrote.
Judette Louis, MD, MPH, associate professor of ob.gyn. and department chair at the University of South Florida, Tampa, said in an interview that the findings in these studies were fairly expected, but it’s important to have data from such a large cohort as the one presented at SMFM.
“It confirmed that those who had severe disease were more likely to have chronic medical conditions, mirroring what we saw in the general population who isn’t pregnant,” Dr. Louis said. “I thought this was very crucial because as pregnant women are trying to decide whether they should get the COVID vaccine, this provides support to say that if you’re pregnant, you’re more likely to have severe disease [if you have] other chronic medical conditions.”
The findings also confirm the importance of pregnant people taking precautions to avoid infection.
“Even though these individuals are, as a group, in an age cohort that mostly has asymptomatic disease, for some of them, it results in severe disease and even maternal death,” she said. “They should still take it seriously if they’re pregnant.”
The SMFM abstract study was funded by the NICHD. The Washington study was funded by the University of Washington Population Health Initiative, the National Institutes of Health, and philanthropic gift funds. One coauthor of the Washington study is on a Pfizer and GlaxoSmithKline advisory board for immunizations. No other authors or individuals interviewed reported any disclosures.
Pregnant patients with COVID-19 infections were more likely to experience severe disease if they had preexisting comorbidities, such as chronic hypertension, asthma, or pregestational diabetes, according to findings from a new study presented at the meeting sponsored by the Society for Maternal-Fetal Medicine.
The study included outcomes for the largest multistate cohort of pregnant patients with COVID-19 outside of what the Centers for Disease Control and Prevention is tracking. Its findings also mirrored those of a multicenter, retrospective study in Washington state, published in the American Journal of Obstetrics & Gynecology. That study also found that pregnant patients hospitalized for COVID-19 were more likely to have comorbidities, and both studies found an increased likelihood of preterm birth among pregnant patients with severe or critical disease.
Disease severity linked to risk of perinatal complications
In the abstract presented at the SMFM meeting, more severe disease was associated with older age and a higher median body mass index, as seen in the general population, but the researchers found no differences in disease severity occurred by race or ethnicity, Torri D. Metz, MD, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, told attendees of the conference. The researchers also found that perinatal complications were more prevalent in those with severe or critical COVID-19 disease but not in those with mild or moderate disease. Vertical COVID-19 transmission from mother to child was rare.
The observational study included all patients who had a singleton pregnancy, had a positive SARS-CoV-2 test, and delivered between March 1 and July 31, 2020, at one of the 33 U.S. hospitals in the NICHD Maternal-Fetal Medicine Units Network, spread across 14 states. The researchers used electronic medical records to determine incidence of cesarean delivery, postpartum hemorrhage, hypertensive disorders of pregnancy, preterm birth (less than 37 weeks), maternal death, infant death, and positive infant COVID-19 test. They tracked mothers through 6 weeks post partum and newborns through delivery hospitalization.
Of 1,291 patients in the cohort, 1,219 received their first positive COVID-19 test during pregnancy. The others tested positive while in the hospital for delivery or within a month and a half after discharge. Limiting their analysis to those who developed COVID-19 while pregnant prior to delivery, nearly half (47%) were asymptomatic.
The disease was mild in 27%, moderate in 14%, severe in 8%, and critical in 4%. The researchers used the National Institutes of Health classifications for severity and included deaths in the critical group. The most common symptom was a cough, reported by a third of the patients (34%). Four of six maternal deaths that occurred were caused by COVID-19.
Compared with an average age of 28 in those without symptoms, the mean age was 29 in those with mild/moderate disease and 30 in those with severe/critical disease (P = .006). Similarly, the mean BMI was 28.3 in asymptomatic patients, 29 in those with mild/moderate disease, and 32.3 in those with severe/critical disease (P < .001). Despite a diverse cohort – 53% Hispanic, 23% Black, and 15% White – the researches found no racial/ethnic trends in disease severity.
Patients who had asthma, chronic obstructive pulmonary disorder, pregestational diabetes, chronic hypertension, chronic liver disease, or a seizure disorder were all significantly more likely to have critical/severe disease than mild/moderate disease, and more likely to have mild/moderate disease than asymptomatic (P values ranged from < .001 to .02).
The mothers with critical or severe illness were 1.6 times more likely to have cesarean births and to have hypertensive disorders of pregnancy, and they were twice as likely to have postpartum hemorrhage (P < .001; P = .007). Those with mild or moderate disease, however, had no increased risks for perinatal complications over asymptomatic patients.
Critical or severe illness was also associated with more than triple the risk of preterm birth (adjusted risk ratio, 3.6; P < .001). Newborns of mothers with critical or severe illness also had three times greater risk of neonatal ICU admission (ARR, 3.1; P <. 001) and weighed an average 385 g less than newborns of asymptomatic mothers. COVID-19 rate among infants was only 1% during delivery hospitalization.
Since the study cutoff was July 30 and COVID infections only became prevalent in March, the researchers were unable to evaluate women for outcomes resulting from COVID infections in early pregnancy, such as congenital anomalies or early miscarriage, Dr. Metz said. In addition, since many of the sites are urban centers, the data may not be generalizable to rural areas.
Peter S. Bernstein, MD, MPH, of Montefiore Medical Center, New York, asked whether the increased cesarean deliveries and preterm births in the group of women with severe disease were caused by usual obstetric causes or the treatment of COVID-19 infection. Dr. Metz said the vast majority of preterm deliveries were indicated, but only a small proportion were induced for COVID-19 alone. “A lot had hypertensive disorders of pregnancies or PPROM, so it’s partly driven by the infection itself but also partly driven by some of those perinatal complications,” she said.
Similar findings in Washington
In the Washington study, among 240 pregnant patients with confirmed COVID-19 infection between March 1 and July 30, 2020, 1 in 11 developed severe or critical disease, and 1 in 10 were hospitalized. The pregnant patients had more than triple the risk of hospitalization compared with adults of similar ages in the general population (10% vs. 2.8%; rate ratio, 3.5). Similar to the multistate NICHD study, women were more likely to be hospitalized if they had asthma, hypertension, type 2 diabetes, autoimmune disease, or class III obesity.
Three mothers died of COVID-19, resulting in a case fatality rate 13.6 times greater than nonpregnant patients with COVID-19 in the general population. The absolute difference in the rate was 1.2%. As seen in the NICHD study, preterm birth was more common in mothers with severe or critical COVID-19. Nearly half (45.4%) of mothers with severe or critical COVID-19 delivered preterm compared to 5.2% in those with mild COVID-19 (P < .001).
“Our finding that deaths in pregnant patients contributed disproportionately to deaths from COVID-19 among 20- to 39-year-olds in Washington state is similar to what was observed during the influenza A virus H1N1 2009 pandemic,” Erica M. Lokken, PhD, MS, of the departments of global health and ob.gyn. at the University of Washington, Seattle, and colleagues wrote in the Washington study. But they noted that it took 8 months into the pandemic before pregnant patients were identified as a high-risk group for COVID-19.
“Given the similarity in clinical course between COVID-19 and IAV H1N1 2009 with an increased risk for mortality during pregnancy and the postpartum period, we strongly recommend that pregnant patients should be considered a high-risk population to novel highly pathogenic respiratory viruses until proven otherwise by population-based studies with good ascertainment of pregnancy status,” they wrote.
Judette Louis, MD, MPH, associate professor of ob.gyn. and department chair at the University of South Florida, Tampa, said in an interview that the findings in these studies were fairly expected, but it’s important to have data from such a large cohort as the one presented at SMFM.
“It confirmed that those who had severe disease were more likely to have chronic medical conditions, mirroring what we saw in the general population who isn’t pregnant,” Dr. Louis said. “I thought this was very crucial because as pregnant women are trying to decide whether they should get the COVID vaccine, this provides support to say that if you’re pregnant, you’re more likely to have severe disease [if you have] other chronic medical conditions.”
The findings also confirm the importance of pregnant people taking precautions to avoid infection.
“Even though these individuals are, as a group, in an age cohort that mostly has asymptomatic disease, for some of them, it results in severe disease and even maternal death,” she said. “They should still take it seriously if they’re pregnant.”
The SMFM abstract study was funded by the NICHD. The Washington study was funded by the University of Washington Population Health Initiative, the National Institutes of Health, and philanthropic gift funds. One coauthor of the Washington study is on a Pfizer and GlaxoSmithKline advisory board for immunizations. No other authors or individuals interviewed reported any disclosures.
Pregnant patients with COVID-19 infections were more likely to experience severe disease if they had preexisting comorbidities, such as chronic hypertension, asthma, or pregestational diabetes, according to findings from a new study presented at the meeting sponsored by the Society for Maternal-Fetal Medicine.
The study included outcomes for the largest multistate cohort of pregnant patients with COVID-19 outside of what the Centers for Disease Control and Prevention is tracking. Its findings also mirrored those of a multicenter, retrospective study in Washington state, published in the American Journal of Obstetrics & Gynecology. That study also found that pregnant patients hospitalized for COVID-19 were more likely to have comorbidities, and both studies found an increased likelihood of preterm birth among pregnant patients with severe or critical disease.
Disease severity linked to risk of perinatal complications
In the abstract presented at the SMFM meeting, more severe disease was associated with older age and a higher median body mass index, as seen in the general population, but the researchers found no differences in disease severity occurred by race or ethnicity, Torri D. Metz, MD, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network, told attendees of the conference. The researchers also found that perinatal complications were more prevalent in those with severe or critical COVID-19 disease but not in those with mild or moderate disease. Vertical COVID-19 transmission from mother to child was rare.
The observational study included all patients who had a singleton pregnancy, had a positive SARS-CoV-2 test, and delivered between March 1 and July 31, 2020, at one of the 33 U.S. hospitals in the NICHD Maternal-Fetal Medicine Units Network, spread across 14 states. The researchers used electronic medical records to determine incidence of cesarean delivery, postpartum hemorrhage, hypertensive disorders of pregnancy, preterm birth (less than 37 weeks), maternal death, infant death, and positive infant COVID-19 test. They tracked mothers through 6 weeks post partum and newborns through delivery hospitalization.
Of 1,291 patients in the cohort, 1,219 received their first positive COVID-19 test during pregnancy. The others tested positive while in the hospital for delivery or within a month and a half after discharge. Limiting their analysis to those who developed COVID-19 while pregnant prior to delivery, nearly half (47%) were asymptomatic.
The disease was mild in 27%, moderate in 14%, severe in 8%, and critical in 4%. The researchers used the National Institutes of Health classifications for severity and included deaths in the critical group. The most common symptom was a cough, reported by a third of the patients (34%). Four of six maternal deaths that occurred were caused by COVID-19.
Compared with an average age of 28 in those without symptoms, the mean age was 29 in those with mild/moderate disease and 30 in those with severe/critical disease (P = .006). Similarly, the mean BMI was 28.3 in asymptomatic patients, 29 in those with mild/moderate disease, and 32.3 in those with severe/critical disease (P < .001). Despite a diverse cohort – 53% Hispanic, 23% Black, and 15% White – the researches found no racial/ethnic trends in disease severity.
Patients who had asthma, chronic obstructive pulmonary disorder, pregestational diabetes, chronic hypertension, chronic liver disease, or a seizure disorder were all significantly more likely to have critical/severe disease than mild/moderate disease, and more likely to have mild/moderate disease than asymptomatic (P values ranged from < .001 to .02).
The mothers with critical or severe illness were 1.6 times more likely to have cesarean births and to have hypertensive disorders of pregnancy, and they were twice as likely to have postpartum hemorrhage (P < .001; P = .007). Those with mild or moderate disease, however, had no increased risks for perinatal complications over asymptomatic patients.
Critical or severe illness was also associated with more than triple the risk of preterm birth (adjusted risk ratio, 3.6; P < .001). Newborns of mothers with critical or severe illness also had three times greater risk of neonatal ICU admission (ARR, 3.1; P <. 001) and weighed an average 385 g less than newborns of asymptomatic mothers. COVID-19 rate among infants was only 1% during delivery hospitalization.
Since the study cutoff was July 30 and COVID infections only became prevalent in March, the researchers were unable to evaluate women for outcomes resulting from COVID infections in early pregnancy, such as congenital anomalies or early miscarriage, Dr. Metz said. In addition, since many of the sites are urban centers, the data may not be generalizable to rural areas.
Peter S. Bernstein, MD, MPH, of Montefiore Medical Center, New York, asked whether the increased cesarean deliveries and preterm births in the group of women with severe disease were caused by usual obstetric causes or the treatment of COVID-19 infection. Dr. Metz said the vast majority of preterm deliveries were indicated, but only a small proportion were induced for COVID-19 alone. “A lot had hypertensive disorders of pregnancies or PPROM, so it’s partly driven by the infection itself but also partly driven by some of those perinatal complications,” she said.
Similar findings in Washington
In the Washington study, among 240 pregnant patients with confirmed COVID-19 infection between March 1 and July 30, 2020, 1 in 11 developed severe or critical disease, and 1 in 10 were hospitalized. The pregnant patients had more than triple the risk of hospitalization compared with adults of similar ages in the general population (10% vs. 2.8%; rate ratio, 3.5). Similar to the multistate NICHD study, women were more likely to be hospitalized if they had asthma, hypertension, type 2 diabetes, autoimmune disease, or class III obesity.
Three mothers died of COVID-19, resulting in a case fatality rate 13.6 times greater than nonpregnant patients with COVID-19 in the general population. The absolute difference in the rate was 1.2%. As seen in the NICHD study, preterm birth was more common in mothers with severe or critical COVID-19. Nearly half (45.4%) of mothers with severe or critical COVID-19 delivered preterm compared to 5.2% in those with mild COVID-19 (P < .001).
“Our finding that deaths in pregnant patients contributed disproportionately to deaths from COVID-19 among 20- to 39-year-olds in Washington state is similar to what was observed during the influenza A virus H1N1 2009 pandemic,” Erica M. Lokken, PhD, MS, of the departments of global health and ob.gyn. at the University of Washington, Seattle, and colleagues wrote in the Washington study. But they noted that it took 8 months into the pandemic before pregnant patients were identified as a high-risk group for COVID-19.
“Given the similarity in clinical course between COVID-19 and IAV H1N1 2009 with an increased risk for mortality during pregnancy and the postpartum period, we strongly recommend that pregnant patients should be considered a high-risk population to novel highly pathogenic respiratory viruses until proven otherwise by population-based studies with good ascertainment of pregnancy status,” they wrote.
Judette Louis, MD, MPH, associate professor of ob.gyn. and department chair at the University of South Florida, Tampa, said in an interview that the findings in these studies were fairly expected, but it’s important to have data from such a large cohort as the one presented at SMFM.
“It confirmed that those who had severe disease were more likely to have chronic medical conditions, mirroring what we saw in the general population who isn’t pregnant,” Dr. Louis said. “I thought this was very crucial because as pregnant women are trying to decide whether they should get the COVID vaccine, this provides support to say that if you’re pregnant, you’re more likely to have severe disease [if you have] other chronic medical conditions.”
The findings also confirm the importance of pregnant people taking precautions to avoid infection.
“Even though these individuals are, as a group, in an age cohort that mostly has asymptomatic disease, for some of them, it results in severe disease and even maternal death,” she said. “They should still take it seriously if they’re pregnant.”
The SMFM abstract study was funded by the NICHD. The Washington study was funded by the University of Washington Population Health Initiative, the National Institutes of Health, and philanthropic gift funds. One coauthor of the Washington study is on a Pfizer and GlaxoSmithKline advisory board for immunizations. No other authors or individuals interviewed reported any disclosures.
FROM THE PREGNANCY MEETING
Obesity: A ‘double hit’ in pregnant women with heart disease
Being obese and pregnant raises the risk for cardiac complications in women with preexisting heart disease, new research suggests, highlighting the need for earlier interventions in this high-risk population.
The analysis of 790 pregnancies revealed that 23% of women with obesity, defined as body mass index greater than 30 kg/m2, had a cardiac event during pregnancy versus 14% of women with normal body weight (P = .006).
The difference was driven largely by an increase in heart failure (8% vs. 3%; P = .02), although arrhythmias also trended higher in obese women (14% vs. 10%; P = .19).
Nearly half of the women with obesity and a cardiac event presented in the postpartum period (47%).
In multivariate analysis, both obesity and Canadian Cardiac Disease in Pregnancy Study (CARPREG) II risk score were independent predictors of cardiac events (odds ratios for both, 1.7), the investigators, led by Birgit Pfaller, MD, University of Toronto, reported in the Journal of the American College of Cardiology.
Although obesity has been linked to worse pregnancy outcomes and higher cardiovascular risk after delivery in the general population, the authors noted that this is the first study to examine its effect on outcomes in women with heart disease.
“We wanted to look at this high-risk group of women that had preexisting heart disease, but in addition had obesity, to try and find out if there was a kind of double hit for these women – and that, in the end, is what we found. It’s not just simply having heart disease, not simply having obesity, but the combination that’s problematic,” senior author and cardiologist Candice Silversides, MD, University of Toronto, said in an interview.
The findings are concerning given the rising prevalence of obesity worldwide. National data from 2018 show that slightly more than half of women who gave birth in the United States were significantly overweight or obese before becoming pregnant.
Similarly, in the present analysis of 600 women in the CARPREG study who gave birth from 2004 to 2014, nearly 1 in 5 pregnancies (19%) occurred in women with obesity and 25% were in overweight women.
Obese women were significantly more likely than those without obesity to have coronary artery disease (6% vs. 2%), cardiomyopathies (19% vs. 8%) and left ventricular dysfunction (19% vs. 12%) and to be hypertensive or have a hypertensive disorder of pregnancy (13% vs. 3%).
Preeclampsia developed in 32 women during the index pregnancy and 69% of these women were obese or overweight. Cardiac event rates were similar in women with or without preeclampsia but trended higher in women with preeclampsia with versus without obesity (36% vs. 14%; P = .20).
The ill effects of obesity were also reflected in fetal and neonatal events. Overall, 43% of women with obesity and 33% of normal-weight women had at least one fetal event (P = .02), with higher rates of preterm birth (19% vs. 10%; P = .005) and respiratory distress syndrome (8% vs. 3%; P = .02) in women with obesity. Congenital cardiac malformations were present in 6% of women in both groups.
Taken together, the composite of cardiac events, preeclampsia, or fetal events was significantly more common in women with obesity than in normal-weight women (56% vs. 41%; P = .002).
“We’ve spent the last number of years trying to research and understand what the drivers of these adverse outcomes are in this high-risk pregnant cohort, but on a bigger picture the real issue is how do we start intervening in a meaningful way,” Dr. Silversides said.
Like many in the burgeoning field of cardio-obstetrics, the team proposed a multidisciplinary approach that stresses preconception counseling, educating pregnant women with heart disease and obesity about their risks, ensuring that dietary advice, weight-gain recommendations, and comorbidities are addressed as part of routine care, and providing postpartum surveillance.
Preconception screening “has been the recommendation for a long, long time; it’s just that it doesn’t always happen in reality,” she said. “Many pregnancies aren’t planned and not all women are filtered into preconception counseling. So sometimes you’ll do it at the first antenatal visit and try to ensure women are educated but optimally you want to do it well in advance of pregnancy.”
Part of that preconception counseling “should also include giving them appropriate advice for contraception, if what they want to do is avoid pregnancy,” added Dr. Silversides.
Garima Sharma, MD, Ciccarone Center for the Prevention of Cardiovascular Disease, Johns Hopkins University, Baltimore, and colleagues wrote in an accompanying editorial that the adverse events observed in this high-risk cohort have “important implications for cardio-obstetricians and should be incorporated in routine prepregnancy and antenatal counseling, monitoring, and risk stratification for women with existing cardiovascular disease.”
They pointed to a paucity of data incorporating maternal prepregnancy obesity and gestational weight gain in risk prediction and called for larger population-based studies on the additive impact of obesity severity on predicting adverse cardiac events in women with existing cardiovascular disease.
Randomized trials are also urgently needed to evaluate the effect of nutritional and behavioral interventions in pregnancy on short- and long-term outcomes in mother and child.
“As the obesity epidemic continues to grow and public health interventions promoting lifestyle changes for obesity management remain a major challenge, maternal obesity may prove to be the ‘Achilles’ heel’ of sustainable national efforts to reduce maternal mortality and improve health equity. This is a call to action,” Dr. Sharma and colleagues concluded.
The investigators noted that the study was conducted at a single center and used self-reported pregnancy weight collected at the first antenatal visit, which may have underestimated obesity rates. Other limitations are that weight changes over the course of pregnancy were not studied and there was a limited number of women with a body mass index of 40 or higher.
The study was supported by a grant from the Allan E. Tiffin Trust, Toronto General and Western Hospital Foundation, and by a donation from Mrs. Josephine Rogers, Toronto General Hospital. Dr. Silversides is supported by the Miles Nadal Chair in Pregnancy and Heart Disease. Dr. Sharma and colleagues disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Being obese and pregnant raises the risk for cardiac complications in women with preexisting heart disease, new research suggests, highlighting the need for earlier interventions in this high-risk population.
The analysis of 790 pregnancies revealed that 23% of women with obesity, defined as body mass index greater than 30 kg/m2, had a cardiac event during pregnancy versus 14% of women with normal body weight (P = .006).
The difference was driven largely by an increase in heart failure (8% vs. 3%; P = .02), although arrhythmias also trended higher in obese women (14% vs. 10%; P = .19).
Nearly half of the women with obesity and a cardiac event presented in the postpartum period (47%).
In multivariate analysis, both obesity and Canadian Cardiac Disease in Pregnancy Study (CARPREG) II risk score were independent predictors of cardiac events (odds ratios for both, 1.7), the investigators, led by Birgit Pfaller, MD, University of Toronto, reported in the Journal of the American College of Cardiology.
Although obesity has been linked to worse pregnancy outcomes and higher cardiovascular risk after delivery in the general population, the authors noted that this is the first study to examine its effect on outcomes in women with heart disease.
“We wanted to look at this high-risk group of women that had preexisting heart disease, but in addition had obesity, to try and find out if there was a kind of double hit for these women – and that, in the end, is what we found. It’s not just simply having heart disease, not simply having obesity, but the combination that’s problematic,” senior author and cardiologist Candice Silversides, MD, University of Toronto, said in an interview.
The findings are concerning given the rising prevalence of obesity worldwide. National data from 2018 show that slightly more than half of women who gave birth in the United States were significantly overweight or obese before becoming pregnant.
Similarly, in the present analysis of 600 women in the CARPREG study who gave birth from 2004 to 2014, nearly 1 in 5 pregnancies (19%) occurred in women with obesity and 25% were in overweight women.
Obese women were significantly more likely than those without obesity to have coronary artery disease (6% vs. 2%), cardiomyopathies (19% vs. 8%) and left ventricular dysfunction (19% vs. 12%) and to be hypertensive or have a hypertensive disorder of pregnancy (13% vs. 3%).
Preeclampsia developed in 32 women during the index pregnancy and 69% of these women were obese or overweight. Cardiac event rates were similar in women with or without preeclampsia but trended higher in women with preeclampsia with versus without obesity (36% vs. 14%; P = .20).
The ill effects of obesity were also reflected in fetal and neonatal events. Overall, 43% of women with obesity and 33% of normal-weight women had at least one fetal event (P = .02), with higher rates of preterm birth (19% vs. 10%; P = .005) and respiratory distress syndrome (8% vs. 3%; P = .02) in women with obesity. Congenital cardiac malformations were present in 6% of women in both groups.
Taken together, the composite of cardiac events, preeclampsia, or fetal events was significantly more common in women with obesity than in normal-weight women (56% vs. 41%; P = .002).
“We’ve spent the last number of years trying to research and understand what the drivers of these adverse outcomes are in this high-risk pregnant cohort, but on a bigger picture the real issue is how do we start intervening in a meaningful way,” Dr. Silversides said.
Like many in the burgeoning field of cardio-obstetrics, the team proposed a multidisciplinary approach that stresses preconception counseling, educating pregnant women with heart disease and obesity about their risks, ensuring that dietary advice, weight-gain recommendations, and comorbidities are addressed as part of routine care, and providing postpartum surveillance.
Preconception screening “has been the recommendation for a long, long time; it’s just that it doesn’t always happen in reality,” she said. “Many pregnancies aren’t planned and not all women are filtered into preconception counseling. So sometimes you’ll do it at the first antenatal visit and try to ensure women are educated but optimally you want to do it well in advance of pregnancy.”
Part of that preconception counseling “should also include giving them appropriate advice for contraception, if what they want to do is avoid pregnancy,” added Dr. Silversides.
Garima Sharma, MD, Ciccarone Center for the Prevention of Cardiovascular Disease, Johns Hopkins University, Baltimore, and colleagues wrote in an accompanying editorial that the adverse events observed in this high-risk cohort have “important implications for cardio-obstetricians and should be incorporated in routine prepregnancy and antenatal counseling, monitoring, and risk stratification for women with existing cardiovascular disease.”
They pointed to a paucity of data incorporating maternal prepregnancy obesity and gestational weight gain in risk prediction and called for larger population-based studies on the additive impact of obesity severity on predicting adverse cardiac events in women with existing cardiovascular disease.
Randomized trials are also urgently needed to evaluate the effect of nutritional and behavioral interventions in pregnancy on short- and long-term outcomes in mother and child.
“As the obesity epidemic continues to grow and public health interventions promoting lifestyle changes for obesity management remain a major challenge, maternal obesity may prove to be the ‘Achilles’ heel’ of sustainable national efforts to reduce maternal mortality and improve health equity. This is a call to action,” Dr. Sharma and colleagues concluded.
The investigators noted that the study was conducted at a single center and used self-reported pregnancy weight collected at the first antenatal visit, which may have underestimated obesity rates. Other limitations are that weight changes over the course of pregnancy were not studied and there was a limited number of women with a body mass index of 40 or higher.
The study was supported by a grant from the Allan E. Tiffin Trust, Toronto General and Western Hospital Foundation, and by a donation from Mrs. Josephine Rogers, Toronto General Hospital. Dr. Silversides is supported by the Miles Nadal Chair in Pregnancy and Heart Disease. Dr. Sharma and colleagues disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Being obese and pregnant raises the risk for cardiac complications in women with preexisting heart disease, new research suggests, highlighting the need for earlier interventions in this high-risk population.
The analysis of 790 pregnancies revealed that 23% of women with obesity, defined as body mass index greater than 30 kg/m2, had a cardiac event during pregnancy versus 14% of women with normal body weight (P = .006).
The difference was driven largely by an increase in heart failure (8% vs. 3%; P = .02), although arrhythmias also trended higher in obese women (14% vs. 10%; P = .19).
Nearly half of the women with obesity and a cardiac event presented in the postpartum period (47%).
In multivariate analysis, both obesity and Canadian Cardiac Disease in Pregnancy Study (CARPREG) II risk score were independent predictors of cardiac events (odds ratios for both, 1.7), the investigators, led by Birgit Pfaller, MD, University of Toronto, reported in the Journal of the American College of Cardiology.
Although obesity has been linked to worse pregnancy outcomes and higher cardiovascular risk after delivery in the general population, the authors noted that this is the first study to examine its effect on outcomes in women with heart disease.
“We wanted to look at this high-risk group of women that had preexisting heart disease, but in addition had obesity, to try and find out if there was a kind of double hit for these women – and that, in the end, is what we found. It’s not just simply having heart disease, not simply having obesity, but the combination that’s problematic,” senior author and cardiologist Candice Silversides, MD, University of Toronto, said in an interview.
The findings are concerning given the rising prevalence of obesity worldwide. National data from 2018 show that slightly more than half of women who gave birth in the United States were significantly overweight or obese before becoming pregnant.
Similarly, in the present analysis of 600 women in the CARPREG study who gave birth from 2004 to 2014, nearly 1 in 5 pregnancies (19%) occurred in women with obesity and 25% were in overweight women.
Obese women were significantly more likely than those without obesity to have coronary artery disease (6% vs. 2%), cardiomyopathies (19% vs. 8%) and left ventricular dysfunction (19% vs. 12%) and to be hypertensive or have a hypertensive disorder of pregnancy (13% vs. 3%).
Preeclampsia developed in 32 women during the index pregnancy and 69% of these women were obese or overweight. Cardiac event rates were similar in women with or without preeclampsia but trended higher in women with preeclampsia with versus without obesity (36% vs. 14%; P = .20).
The ill effects of obesity were also reflected in fetal and neonatal events. Overall, 43% of women with obesity and 33% of normal-weight women had at least one fetal event (P = .02), with higher rates of preterm birth (19% vs. 10%; P = .005) and respiratory distress syndrome (8% vs. 3%; P = .02) in women with obesity. Congenital cardiac malformations were present in 6% of women in both groups.
Taken together, the composite of cardiac events, preeclampsia, or fetal events was significantly more common in women with obesity than in normal-weight women (56% vs. 41%; P = .002).
“We’ve spent the last number of years trying to research and understand what the drivers of these adverse outcomes are in this high-risk pregnant cohort, but on a bigger picture the real issue is how do we start intervening in a meaningful way,” Dr. Silversides said.
Like many in the burgeoning field of cardio-obstetrics, the team proposed a multidisciplinary approach that stresses preconception counseling, educating pregnant women with heart disease and obesity about their risks, ensuring that dietary advice, weight-gain recommendations, and comorbidities are addressed as part of routine care, and providing postpartum surveillance.
Preconception screening “has been the recommendation for a long, long time; it’s just that it doesn’t always happen in reality,” she said. “Many pregnancies aren’t planned and not all women are filtered into preconception counseling. So sometimes you’ll do it at the first antenatal visit and try to ensure women are educated but optimally you want to do it well in advance of pregnancy.”
Part of that preconception counseling “should also include giving them appropriate advice for contraception, if what they want to do is avoid pregnancy,” added Dr. Silversides.
Garima Sharma, MD, Ciccarone Center for the Prevention of Cardiovascular Disease, Johns Hopkins University, Baltimore, and colleagues wrote in an accompanying editorial that the adverse events observed in this high-risk cohort have “important implications for cardio-obstetricians and should be incorporated in routine prepregnancy and antenatal counseling, monitoring, and risk stratification for women with existing cardiovascular disease.”
They pointed to a paucity of data incorporating maternal prepregnancy obesity and gestational weight gain in risk prediction and called for larger population-based studies on the additive impact of obesity severity on predicting adverse cardiac events in women with existing cardiovascular disease.
Randomized trials are also urgently needed to evaluate the effect of nutritional and behavioral interventions in pregnancy on short- and long-term outcomes in mother and child.
“As the obesity epidemic continues to grow and public health interventions promoting lifestyle changes for obesity management remain a major challenge, maternal obesity may prove to be the ‘Achilles’ heel’ of sustainable national efforts to reduce maternal mortality and improve health equity. This is a call to action,” Dr. Sharma and colleagues concluded.
The investigators noted that the study was conducted at a single center and used self-reported pregnancy weight collected at the first antenatal visit, which may have underestimated obesity rates. Other limitations are that weight changes over the course of pregnancy were not studied and there was a limited number of women with a body mass index of 40 or higher.
The study was supported by a grant from the Allan E. Tiffin Trust, Toronto General and Western Hospital Foundation, and by a donation from Mrs. Josephine Rogers, Toronto General Hospital. Dr. Silversides is supported by the Miles Nadal Chair in Pregnancy and Heart Disease. Dr. Sharma and colleagues disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Two popular screening tests for gestational diabetes clinically equivalent
Broadening the diagnosis of gestational diabetes mellitus (GDM) with a one-step screening approach does not lead to significant differences in maternal or perinatal outcomes, compared with a two-step approach. Investigators reported these findings in the New England Journal of Medicine after testing the two screening methods in more than 23,000 pregnant women.
GDM affects 6%-25% of pregnant women, increasing the risk of neonatal death and stillborn births. It can also lead to serious complications such as fetal overgrowth. Clinical guidelines recommend GDM screening between 24 and 28 weeks’ gestation to improve outcomes in mothers and infants. However, the scientific community has struggled to reach a consensus on testing approach.
For decades, clinicians used a two-step screening approach: a nonfasting 1-hour glucose challenge test and a longer 3-hour fasting oral glucose tolerance test to diagnose GDM; roughly 20% who test positive on this glucose challenge test require the second step. Results of a large study led to new diagnostic criteria on a one-step 75-g oral glucose tolerance test. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study “found a linear relationship with hyperglycemia and outcomes – the higher the glucose, the worse the outcomes,” said Teresa Hillier, MD, MS, an endocrinologist and investigator with Kaiser Permanente Center for Health Research Northwest and CHR-Hawaii. The International Association of the Diabetes and Pregnancy Study Groups (IADPSG) made a clinical recommendation on the one-step approach, now a common screening tool in the United States.
A focus on rare GDM outcomes
The IADPSG fasting one-step criteria typically identifies women with milder symptoms as having gestational diabetes, a factor expected to increase diagnosis rates by two- or threefold, said Dr. Hillier. “The unknown question was whether diagnosing and treating more women would be associated with any differences in any of the multiple GDM-associated outcomes for mother and baby.”
She and her colleagues conducted a large-scale randomized trial at two Kaiser sites to assess multiple maternal and perinatal outcomes including rare but important GDM-associated outcomes such as stillbirth and neonatal death between the two screening methods.
They randomized 23,792 pregnant women 1:1 to the one- or two-step gestational diabetes test at their first prenatal visit. Primary outcomes included diagnosis of gestational diabetes; large-for-gestational-age infants; primary cesarean section, and gestational hypertension or preeclampsia; and a composite perinatal outcome of any stillbirth, neonatal death, shoulder dystocia, bone fracture, or arm or hand nerve palsy related to birth injury.
Most participants (94%) completed screening, although there was lower adherence to screening in the one-step approach. The reasons for this aren’t entirely clear, said Dr. Hillier. Convenience may be a factor; patients have to fast for several hours to complete the one-step test, whereas the first test of the two-step screening approach can be done at any time of day, and most patients pass this test.
Corroborating HAPO’s results, twice as many women in the one-step group (16.5%) received a GDM diagnosis, compared with 8.5% in the two-step group (unadjusted relative risk, 1.94; 97.5% confidence interval, 1.79-2.11). However, for the other primary outcomes, investigators found no significant differences in incidences or unadjusted risks. Perinatal composite outcomes for the one- and two-step groups were 3.1% and 3.0%, respectively, and primary cesarean section outcomes were 24.0% and 24.6%.
In the one-step group, 8.9% experienced large-for-gestational-age infants outcomes, compared with 9.2% in the two-step group (RR, 0.95; 97.5% CI, 0.87-1.05). Among those diagnosed with gestational diabetes, similar percentages of women in the one- and two-step groups received insulin or hypoglycemic medication (42.6% and 45.6%, respectively).
Dr. Hillier and colleagues also reported comparable results among the two groups on safety outcomes and secondary outcomes such as macrosomia incidence, small-for-gestational-age infants, and factors such as neonatal hypoglycemia and respiratory distress.
“Although we did not find increased harms associated with the diagnosis and treatment of gestational diabetes in many more women with the one-step approach, some retrospective observational cohort studies have shown higher incidences of primary cesarean delivery and neonatal hypoglycemia with one-step screening after conversion from two-step protocols, with no substantive improvement in outcomes,” Dr. Hillier and colleagues noted.
The trial had several limitations. Adjustments made to address lower adherence to the one-step approach might not have accounted for all nonadherence differences. Another issue is the two sites didn’t use identical thresholds for the glucose challenge test in the two-step cohort. Demographically, the study lacked Black and American Indian representation.
“Moreover, the potential long-term benefits of increased diagnoses of gestational diabetes – such as the identification of more women at high risk for subsequent diabetes who might benefit from risk-reduction strategies – were not addressed by the trial,” Brian Casey, MD, wrote in a related editorial. Based on the study’s findings, “the perinatal benefits of the diagnosis of gestational diabetes with the use of the IADPSG single-step approach appear to be insufficient to justify the associated patient and health care costs of broadening the diagnosis” of GDM, added Dr. Casey, a professor with the department of obstetrics and gynecology at the University of Alabama at Birmingham.
U.S. doctors unlikely to change behaviors
Most U.S. physicians favor the two-step method. This has been a huge controversy worldwide, with other countries pushing the United States to use the one-step method, Vincenzo Berghella, MD, a professor with Thomas Jefferson University, Philadelphia, said in an interview. “I expect this study will increase the divide between the U.S. and the rest of the world,” since U.S. physicians will see no benefit to the one-step method, and continue to use the two-step method.
It’s not surprising that GDM diagnosis incidence went up to 16.5% with the inclusion of the one-step test, compared with 8.5% with the two-step test, Dr. Berghella continued. What’s less clear, are the details of treatment among the 8% diagnosed to have GDM with the one-step test, but not the two-step test.
These women were likely to have milder degrees of insulin resistance or GDM. Dr. Berghella, who has advocated in the past for the one-step approach, said it would be important to find out if these women, who test positive at the one-step test but would test negative at the two-step test, were treated properly with diet, exercise, and possibly insulin or other hypoglycemic agents for their mild degree of insulin resistance. The researchers concluded that expanding the definition of GDM through the one-step test didn’t make a difference. However, “it’s not just the test that will make the difference in maternal and baby outcomes, but the aggressive management of diabetes with diet, exercise, and medications as needed once that test comes back abnormal,” he said.
The randomized trial was a massive undertaking, said Dr. Hillier.
“We are still evaluating our future plans,” she added. Forthcoming subgroup analyses from the trial could further help inform clinical practice guidelines.
Dr. Hillier received a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development to support this study. The investigators reported no potential conflict of interest relevant to this article.
Broadening the diagnosis of gestational diabetes mellitus (GDM) with a one-step screening approach does not lead to significant differences in maternal or perinatal outcomes, compared with a two-step approach. Investigators reported these findings in the New England Journal of Medicine after testing the two screening methods in more than 23,000 pregnant women.
GDM affects 6%-25% of pregnant women, increasing the risk of neonatal death and stillborn births. It can also lead to serious complications such as fetal overgrowth. Clinical guidelines recommend GDM screening between 24 and 28 weeks’ gestation to improve outcomes in mothers and infants. However, the scientific community has struggled to reach a consensus on testing approach.
For decades, clinicians used a two-step screening approach: a nonfasting 1-hour glucose challenge test and a longer 3-hour fasting oral glucose tolerance test to diagnose GDM; roughly 20% who test positive on this glucose challenge test require the second step. Results of a large study led to new diagnostic criteria on a one-step 75-g oral glucose tolerance test. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study “found a linear relationship with hyperglycemia and outcomes – the higher the glucose, the worse the outcomes,” said Teresa Hillier, MD, MS, an endocrinologist and investigator with Kaiser Permanente Center for Health Research Northwest and CHR-Hawaii. The International Association of the Diabetes and Pregnancy Study Groups (IADPSG) made a clinical recommendation on the one-step approach, now a common screening tool in the United States.
A focus on rare GDM outcomes
The IADPSG fasting one-step criteria typically identifies women with milder symptoms as having gestational diabetes, a factor expected to increase diagnosis rates by two- or threefold, said Dr. Hillier. “The unknown question was whether diagnosing and treating more women would be associated with any differences in any of the multiple GDM-associated outcomes for mother and baby.”
She and her colleagues conducted a large-scale randomized trial at two Kaiser sites to assess multiple maternal and perinatal outcomes including rare but important GDM-associated outcomes such as stillbirth and neonatal death between the two screening methods.
They randomized 23,792 pregnant women 1:1 to the one- or two-step gestational diabetes test at their first prenatal visit. Primary outcomes included diagnosis of gestational diabetes; large-for-gestational-age infants; primary cesarean section, and gestational hypertension or preeclampsia; and a composite perinatal outcome of any stillbirth, neonatal death, shoulder dystocia, bone fracture, or arm or hand nerve palsy related to birth injury.
Most participants (94%) completed screening, although there was lower adherence to screening in the one-step approach. The reasons for this aren’t entirely clear, said Dr. Hillier. Convenience may be a factor; patients have to fast for several hours to complete the one-step test, whereas the first test of the two-step screening approach can be done at any time of day, and most patients pass this test.
Corroborating HAPO’s results, twice as many women in the one-step group (16.5%) received a GDM diagnosis, compared with 8.5% in the two-step group (unadjusted relative risk, 1.94; 97.5% confidence interval, 1.79-2.11). However, for the other primary outcomes, investigators found no significant differences in incidences or unadjusted risks. Perinatal composite outcomes for the one- and two-step groups were 3.1% and 3.0%, respectively, and primary cesarean section outcomes were 24.0% and 24.6%.
In the one-step group, 8.9% experienced large-for-gestational-age infants outcomes, compared with 9.2% in the two-step group (RR, 0.95; 97.5% CI, 0.87-1.05). Among those diagnosed with gestational diabetes, similar percentages of women in the one- and two-step groups received insulin or hypoglycemic medication (42.6% and 45.6%, respectively).
Dr. Hillier and colleagues also reported comparable results among the two groups on safety outcomes and secondary outcomes such as macrosomia incidence, small-for-gestational-age infants, and factors such as neonatal hypoglycemia and respiratory distress.
“Although we did not find increased harms associated with the diagnosis and treatment of gestational diabetes in many more women with the one-step approach, some retrospective observational cohort studies have shown higher incidences of primary cesarean delivery and neonatal hypoglycemia with one-step screening after conversion from two-step protocols, with no substantive improvement in outcomes,” Dr. Hillier and colleagues noted.
The trial had several limitations. Adjustments made to address lower adherence to the one-step approach might not have accounted for all nonadherence differences. Another issue is the two sites didn’t use identical thresholds for the glucose challenge test in the two-step cohort. Demographically, the study lacked Black and American Indian representation.
“Moreover, the potential long-term benefits of increased diagnoses of gestational diabetes – such as the identification of more women at high risk for subsequent diabetes who might benefit from risk-reduction strategies – were not addressed by the trial,” Brian Casey, MD, wrote in a related editorial. Based on the study’s findings, “the perinatal benefits of the diagnosis of gestational diabetes with the use of the IADPSG single-step approach appear to be insufficient to justify the associated patient and health care costs of broadening the diagnosis” of GDM, added Dr. Casey, a professor with the department of obstetrics and gynecology at the University of Alabama at Birmingham.
U.S. doctors unlikely to change behaviors
Most U.S. physicians favor the two-step method. This has been a huge controversy worldwide, with other countries pushing the United States to use the one-step method, Vincenzo Berghella, MD, a professor with Thomas Jefferson University, Philadelphia, said in an interview. “I expect this study will increase the divide between the U.S. and the rest of the world,” since U.S. physicians will see no benefit to the one-step method, and continue to use the two-step method.
It’s not surprising that GDM diagnosis incidence went up to 16.5% with the inclusion of the one-step test, compared with 8.5% with the two-step test, Dr. Berghella continued. What’s less clear, are the details of treatment among the 8% diagnosed to have GDM with the one-step test, but not the two-step test.
These women were likely to have milder degrees of insulin resistance or GDM. Dr. Berghella, who has advocated in the past for the one-step approach, said it would be important to find out if these women, who test positive at the one-step test but would test negative at the two-step test, were treated properly with diet, exercise, and possibly insulin or other hypoglycemic agents for their mild degree of insulin resistance. The researchers concluded that expanding the definition of GDM through the one-step test didn’t make a difference. However, “it’s not just the test that will make the difference in maternal and baby outcomes, but the aggressive management of diabetes with diet, exercise, and medications as needed once that test comes back abnormal,” he said.
The randomized trial was a massive undertaking, said Dr. Hillier.
“We are still evaluating our future plans,” she added. Forthcoming subgroup analyses from the trial could further help inform clinical practice guidelines.
Dr. Hillier received a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development to support this study. The investigators reported no potential conflict of interest relevant to this article.
Broadening the diagnosis of gestational diabetes mellitus (GDM) with a one-step screening approach does not lead to significant differences in maternal or perinatal outcomes, compared with a two-step approach. Investigators reported these findings in the New England Journal of Medicine after testing the two screening methods in more than 23,000 pregnant women.
GDM affects 6%-25% of pregnant women, increasing the risk of neonatal death and stillborn births. It can also lead to serious complications such as fetal overgrowth. Clinical guidelines recommend GDM screening between 24 and 28 weeks’ gestation to improve outcomes in mothers and infants. However, the scientific community has struggled to reach a consensus on testing approach.
For decades, clinicians used a two-step screening approach: a nonfasting 1-hour glucose challenge test and a longer 3-hour fasting oral glucose tolerance test to diagnose GDM; roughly 20% who test positive on this glucose challenge test require the second step. Results of a large study led to new diagnostic criteria on a one-step 75-g oral glucose tolerance test. The Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study “found a linear relationship with hyperglycemia and outcomes – the higher the glucose, the worse the outcomes,” said Teresa Hillier, MD, MS, an endocrinologist and investigator with Kaiser Permanente Center for Health Research Northwest and CHR-Hawaii. The International Association of the Diabetes and Pregnancy Study Groups (IADPSG) made a clinical recommendation on the one-step approach, now a common screening tool in the United States.
A focus on rare GDM outcomes
The IADPSG fasting one-step criteria typically identifies women with milder symptoms as having gestational diabetes, a factor expected to increase diagnosis rates by two- or threefold, said Dr. Hillier. “The unknown question was whether diagnosing and treating more women would be associated with any differences in any of the multiple GDM-associated outcomes for mother and baby.”
She and her colleagues conducted a large-scale randomized trial at two Kaiser sites to assess multiple maternal and perinatal outcomes including rare but important GDM-associated outcomes such as stillbirth and neonatal death between the two screening methods.
They randomized 23,792 pregnant women 1:1 to the one- or two-step gestational diabetes test at their first prenatal visit. Primary outcomes included diagnosis of gestational diabetes; large-for-gestational-age infants; primary cesarean section, and gestational hypertension or preeclampsia; and a composite perinatal outcome of any stillbirth, neonatal death, shoulder dystocia, bone fracture, or arm or hand nerve palsy related to birth injury.
Most participants (94%) completed screening, although there was lower adherence to screening in the one-step approach. The reasons for this aren’t entirely clear, said Dr. Hillier. Convenience may be a factor; patients have to fast for several hours to complete the one-step test, whereas the first test of the two-step screening approach can be done at any time of day, and most patients pass this test.
Corroborating HAPO’s results, twice as many women in the one-step group (16.5%) received a GDM diagnosis, compared with 8.5% in the two-step group (unadjusted relative risk, 1.94; 97.5% confidence interval, 1.79-2.11). However, for the other primary outcomes, investigators found no significant differences in incidences or unadjusted risks. Perinatal composite outcomes for the one- and two-step groups were 3.1% and 3.0%, respectively, and primary cesarean section outcomes were 24.0% and 24.6%.
In the one-step group, 8.9% experienced large-for-gestational-age infants outcomes, compared with 9.2% in the two-step group (RR, 0.95; 97.5% CI, 0.87-1.05). Among those diagnosed with gestational diabetes, similar percentages of women in the one- and two-step groups received insulin or hypoglycemic medication (42.6% and 45.6%, respectively).
Dr. Hillier and colleagues also reported comparable results among the two groups on safety outcomes and secondary outcomes such as macrosomia incidence, small-for-gestational-age infants, and factors such as neonatal hypoglycemia and respiratory distress.
“Although we did not find increased harms associated with the diagnosis and treatment of gestational diabetes in many more women with the one-step approach, some retrospective observational cohort studies have shown higher incidences of primary cesarean delivery and neonatal hypoglycemia with one-step screening after conversion from two-step protocols, with no substantive improvement in outcomes,” Dr. Hillier and colleagues noted.
The trial had several limitations. Adjustments made to address lower adherence to the one-step approach might not have accounted for all nonadherence differences. Another issue is the two sites didn’t use identical thresholds for the glucose challenge test in the two-step cohort. Demographically, the study lacked Black and American Indian representation.
“Moreover, the potential long-term benefits of increased diagnoses of gestational diabetes – such as the identification of more women at high risk for subsequent diabetes who might benefit from risk-reduction strategies – were not addressed by the trial,” Brian Casey, MD, wrote in a related editorial. Based on the study’s findings, “the perinatal benefits of the diagnosis of gestational diabetes with the use of the IADPSG single-step approach appear to be insufficient to justify the associated patient and health care costs of broadening the diagnosis” of GDM, added Dr. Casey, a professor with the department of obstetrics and gynecology at the University of Alabama at Birmingham.
U.S. doctors unlikely to change behaviors
Most U.S. physicians favor the two-step method. This has been a huge controversy worldwide, with other countries pushing the United States to use the one-step method, Vincenzo Berghella, MD, a professor with Thomas Jefferson University, Philadelphia, said in an interview. “I expect this study will increase the divide between the U.S. and the rest of the world,” since U.S. physicians will see no benefit to the one-step method, and continue to use the two-step method.
It’s not surprising that GDM diagnosis incidence went up to 16.5% with the inclusion of the one-step test, compared with 8.5% with the two-step test, Dr. Berghella continued. What’s less clear, are the details of treatment among the 8% diagnosed to have GDM with the one-step test, but not the two-step test.
These women were likely to have milder degrees of insulin resistance or GDM. Dr. Berghella, who has advocated in the past for the one-step approach, said it would be important to find out if these women, who test positive at the one-step test but would test negative at the two-step test, were treated properly with diet, exercise, and possibly insulin or other hypoglycemic agents for their mild degree of insulin resistance. The researchers concluded that expanding the definition of GDM through the one-step test didn’t make a difference. However, “it’s not just the test that will make the difference in maternal and baby outcomes, but the aggressive management of diabetes with diet, exercise, and medications as needed once that test comes back abnormal,” he said.
The randomized trial was a massive undertaking, said Dr. Hillier.
“We are still evaluating our future plans,” she added. Forthcoming subgroup analyses from the trial could further help inform clinical practice guidelines.
Dr. Hillier received a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development to support this study. The investigators reported no potential conflict of interest relevant to this article.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Caring for women with pelvic floor disorders during pregnancy and postpartum: Expert guidance
Pelvic floor disorders (PFDs) affect many pregnant and newly postpartum women. These conditions, including urinary incontinence, anal incontinence, and pelvic organ prolapse (POP), can be overshadowed by common pregnancy and postpartum concerns (TABLE 1).1 With the use of a few quick screening questions, however, PFDs easily can be identified in this at-risk population. Active management need not be delayed until after delivery for women experiencing bother, as options exist for women with PFDs during pregnancy as well as postpartum.
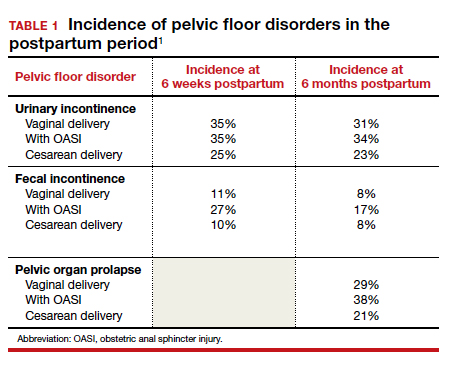
In this article, we discuss the common PFDs that obstetric clinicians face in the context of case scenarios and review how you can be better equipped to care for affected individuals.
CASE 1 Screening
A 30-year-old woman (G1P1) presents for her routine postpartum visit after an operative vaginal delivery with a second-degree laceration.
How would you screen this patient for PFDs?
Why screening for PFDs matters
While there are no validated PFD screening tools for this patient population, clinicians can ask a series of brief open-ended questions as part of the review of systems to efficiently evaluate for the common PFDs in peripartum patients (see “Screening questions to evaluate patients for peripartum pelvic floor disorders” below).
Pelvic floor disorders in the peripartum period can have a significant negative impact. In pregnancy, nearly half of women report psychological strain due to the presence of bowel, bladder, prolapse, or sexual dysfunction symptoms.2 Postpartum, PFDs have negative effects on overall health, well-being, and self-esteem, with significantly increased rates of postpartum depression in women who experience urinary incontinence.3,4 Proactively inquiring about PFD symptoms, providing anticipatory guidance, and recommending treatment options can positively impact a patient in multiple domains.
Sometimes during pregnancy or after having a baby, a woman experiences pelvic floor symptoms. Do you have any of the following?
- leakage with coughing, laughing, sneezing, or physical activity
- urgency to urinate or leakage due to urgency
- bulging or pressure within the vagina
- pain with intercourse
- accidental bowel leakage of stool or flatus
CASE 2 Stress urinary incontinence
A 27-year-old woman (G1P1) presents 2 months following spontaneous vaginal delivery with symptoms of urine leakage with laughing and running. Her urinary incontinence has been improving since delivery, but it continues to be bothersome.
What would you recommend for this patient?
Conservative SUI management strategies in pregnancy
Urinary tract symptoms are common in pregnancy, with up to 41.8% of women reporting urinary symptom distress in the third trimester.5 During pregnancy, estrogen and progesterone decrease urethral pressure that, together with increased intra-abdominal pressure from the gravid uterus, can cause or worsen stress urinary incontinence (SUI).6
During pregnancy, women should be offered conservative therapies for SUI. For women who can perform a pelvic floor contraction (a Kegel exercise), self-guided pelvic floor muscle exercises (PFMEs) may be helpful (see “Pelvic floor muscle exercises” below). We recommend that women start with 1 to 2 sets of 10 Kegel exercises per day and that they hold the squeeze for 2 to 3 seconds, working up to holding for 10 seconds. The goal is to strengthen and improve muscle control so that the Kegel squeeze can be paired with activities that cause SUI.
For women who are unable to perform a Kegel exercise or are not improving with a home PFME regimen, referral to pelvic floor physical therapy (PFPT) can be considered. While data support the efficacy of PFPT for SUI treatment in nonpregnant women,7 data are lacking on PFME in pregnancy.
In women without urinary incontinence, PFME in early pregnancy can prevent the onset of incontinence in late pregnancy and the postpartum period.8 By contrast, the same 2020 Cochrane Review found no evidence that antenatal pelvic floor muscle therapy in incontinent women decreases incontinence in mid- or late-pregnancy or in the postpartum period.8 As the quality of this evidence is very low and there is no evidence of harm with PFME, we continue to recommend it for women with bothersome SUI.
Incontinence pessaries or vaginal inserts (such as Poise Impressa bladder supports) can be helpful for SUI treatment. An incontinence pessary can be fitted in the office, and fitting kits are available for both. Pessaries can safely be used in pregnancy, but there are no data on the efficacy of pessaries for treating SUI in pregnancy. In nonpregnant women, evidence demonstrates 63% satisfaction 3 months post–pessary placement for SUI.7
We do not recommend invasive procedures for the treatment of SUI during pregnancy or in the first 6 months following delivery. There is no evidence that elective cesarean delivery prevents persistent SUI postpartum.9
To identify and engage the proper pelvic floor muscles:
- Insert a finger in the vagina and squeeze the vaginal muscles around your finger.
- Imagine you are sitting on a marble and have to pick it up with the vaginal muscles.
- Squeeze the muscles you would use to stop the flow of urine or hold back flatulence.
Perform sets of 10, 2 to 3 times per day as follows:
- Squeeze: Engage the pelvic floor muscles as described above; avoid performing Kegels while voiding.
- Hold: For 2 to 10 seconds; increase the duration to 10 seconds as able.
- Relax: Completely relax muscles before initating the next squeeze.
Reference
1. UpToDate. Patient education: pelvic muscle (Kegel) exercises (the basics). 2018. https://uptodatefree.ir/topic.htm?path=pelvic-muscle-kegel-exercises-the-basics. Accessed February 24, 2021.
Continue to: Managing SUI in the postpartum period...
Managing SUI in the postpartum period
After the first 6 months postpartum and exhaustion of conservative measures, we offer surgical interventions for women with persistent, bothersome incontinence. Surgery for SUI typically is not recommended until childbearing is complete, but it can be considered if the patient’s bother is significant.
For women with bothersome SUI who still desire future pregnancy, management options include periurethral bulking, a retropubic urethropexy (Burch procedure), or a midurethral sling procedure. Women who undergo an anti-incontinence procedure have an increased risk for urinary retention during a subsequent pregnancy.10 Most women with a midurethral sling will continue to be continent following an obstetric delivery.
Anticipatory guidance
At 3 months postpartum, the incidence of urinary incontinence is 6% to 40%, depending on parity and delivery type. Postpartum urinary incontinence is most common after instrumented vaginal delivery (32%) followed by spontaneous vaginal delivery (28%) and cesarean delivery (15%). The mean prevalence of any type of urinary incontinence is 33% at 3 months postpartum, and only small changes in the rate of urinary incontinence occur over the first postpartum year.11 While urinary incontinence is common postpartum, it should not be considered normal. We counsel that symptoms may improve spontaneously, but treatment can be initiated if the patient experiences significant bother.
A longitudinal cohort study that followed women from 3 months to 12 years postpartum found that, of women with urinary incontinence at 3 months postpartum, 76% continued to report incontinence at 12 years postpartum.12 We recommend that women be counseled that, even when symptoms resolve, they remain at increased risk for urinary incontinence in the future. Invasive therapies should be used to treat bothersome urinary incontinence, not to prevent future incontinence.
CASE 3 Fecal incontinence
A 24-year-old woman (G1P1) presents 3 weeks postpartum following a forceps-assisted vaginal delivery complicated by a 3c laceration. She reports fecal urgency, inability to control flatus, and once-daily fecal incontinence.
How would you evaluate these symptoms?
Steps in evaluation
The initial evaluation should include an inquiry regarding the patient’s stool consistency and bowel regimen. The Bristol stool form scale can be used to help patients describe their typical bowel movements (TABLE 2).13 During healing, the goal is to achieve a Bristol type 4 stool, both to avoid straining and to improve continence, as loose stool is the most difficult to control.

A physical examination can evaluate healing and sphincter integrity; it should include inspection of the distal vagina and perineal body and a digital rectal exam. Anal canal resting tone and squeeze strength should be evaluated, and the digital rectal examination scoring system (DRESS) can be useful for quantification (TABLE 3).14 Lack of tone at rest in the anterolateral portion of the sphincter complex can indicate an internal anal sphincter defect, as 80% of the resting tone comes from this muscle (FIGURE).15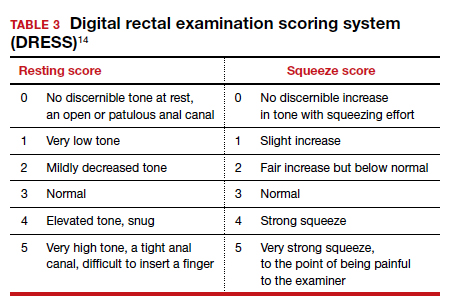
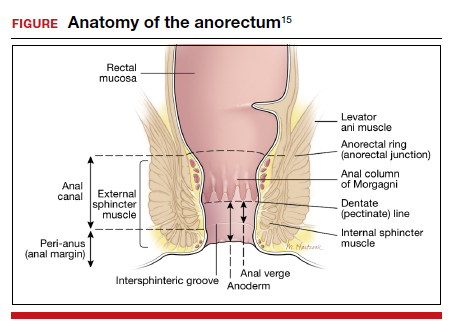
The rectovaginal septum should be assessed given the increased risk of rectovaginal fistula in women with obstetric anal sphincter injury (OASI). The patient should be instructed to contract the anal sphincter, allowing evaluation of muscular contraction. Lack of contraction anteriolaterally may indicate external anal sphincter separation.
Continue to: Conservative options for improving fecal incontinence symptoms...
Conservative options for improving fecal incontinence symptoms
The patient can be counseled regarding stool bulking, first with insoluble fiber supplementation and cessation of stool softeners if she is incontinent of liquid stool. If these measures are not effective, use of a constipating agent, such as loperamide, can improve stool consistency and thereby decrease incontinence episodes. PFPT with biofeedback can be offered as well. While typically we do not recommend initiating PFPT before 6 weeks postpartum, so the initial phases of healing can occur, early referral enables the patient to avoid a delay in access to care.
The patient also can be counseled about a referral to a pelvic floor specialist for further evaluation. A variety of peripartum pelvic floor disorder clinics are being established by Female Pelvic Medicine and Reconstructive Surgery (FPMRS) physicians. These clinics provide the benefit of comprehensive care for pelvic floor disorders in this unique population.
When conservative measures fail. If a patient has persistent bowel control issues despite conservative measures, a referral to an FPMRS physician should be initiated.
Delivery route in future pregnancies
The risk of a subsequent OASI is low. While this means that many women can safely pursue a future vaginal delivery, a scheduled cesarean delivery is indicated for women with persistent bowel control issues, wound healing complications, and those who experienced psychological trauma from their delivery.16 We recommend a shared-decision making approach, reviewing modifiable and nonmodifiable risk factors to help determine whether or not a future vaginal birth is appropriate. It is important to highlight that a cesarean delivery does not protect against fecal incontinence in women with a history of OASI; however, there is benefit in preventing worsening of anal incontinence, if present.17
CASE 4 Uterovaginal prolapse
A 36-year-old woman (G3P3) presents for her routine postpartum visit at 6 weeks after a spontaneous vaginal delivery without lacerations. She reports a persistent feeling of vaginal pressure and fullness. She thinks she felt a bulge with wiping after a bowel movement.
What options are available for this patient?
Prolapse in the peripartum population
Previous studies have revealed an increased prevalence of POP in pregnant women on examination compared with their nulligravid counterparts (47.6% vs 0%).18 With the changes in the hormonal milieu in pregnancy, as well as the weight of the gravid uterus on the pelvic floor, it is not surprising that pregnancy may be the inciting event to expose even transient defects in pelvic organ support.19
It is well established that increasing parity and, to a lesser extent, larger babies are associated with increased risk for future POP and surgery for prolapse. In the first year postpartum, nearly one-third of women have stage 2 or greater prolapse on exam, with studies demonstrating an increased prevalence of postpartum POP in women who delivered vaginally compared with those who delivered by cesarean.20,21
Initial evaluation
Diagnosis can be made during a routine pelvic exam by having the patient perform a Valsalva maneuver while in the lithotomy position. Using half of a speculum permits evaluation of the anterior and posterior vaginal walls separately, and Valsalva during a bimanual exam can aid in evaluating descensus of the uterus and cervix.
Excellent free patient education resources available online through the American Urogynecologic Society and the International Urogynecological Association can be used to direct counseling.
Continue to: Treatments you can offer for POP...
Treatments you can offer for POP
For pregnant or postpartum patients with bothersome prolapse, initial management options include pessary fitting and/or PFPT referral. In pregnancy, women often can be successfully fitted with a pessary for POP; however, as expulsion is a common issue, selection of a stiffer or space-occupying device may be more efficacious.
Often, early onset POP in pregnancy resolves as the gravid uterus lifts out of the pelvis in the second trimester, at which time the pessary can be discontinued. In the postpartum period, a pessary fitting can be undertaken similarly to that in nonpregnant patients. While data are lacking in the peripartum population, evidence supports the positive impact of PFPT on improving POP symptom bother.22 Additionally, for postpartum women who experience OASI, PFPT can produce significant improvement in subjective POP and associated bother.23
Impact of future childbearing wishes on treatment
The desire for future childbearing does not preclude treatment of patients experiencing bother from POP after conservative management options have failed. Both vaginal native tissue and mesh-augmented uterine-sparing repairs are performed by many FPMRS specialists and are associated with good outcomes. As with SUI, we do not recommend invasive treatment for POP during pregnancy or before 6 months postpartum.
In conclusion
Obstetric specialists play an essential role in caring for women with PFDs in the peripartum period. Basic evaluation, counseling, and management can be initiated using many of the resources already available in an obstetric ambulatory practice. Important adjunctive resources include those available for both providers and patients through the American Urogynecologic Society and the International Urogynecological Association. In addition, clinicians can partner with pelvic floor specialists through the growing number of FPMRS-run peripartum pelvic floor disorder clinics across the country and pelvic floor physical therapists.
If these specialty clinics and therapists are not available in your area, FPMRS specialists, urologists, gastroenterologists, and/or colorectal surgeons can aid in patient diagnosis and management to reach the ultimate goal of improving PFDs in this at-risk population. ●
- Madsen AM, Hickman LC, Propst K. Recognition and management of pelvic floor disorders in pregnancy and the postpartum period. Obstet Gynecol Clin North Am. Forthcoming 2021.
- Bodner-Adler B, Kimberger O, Laml T, et al. Prevalence and risk factors for pelvic floor disorders during early and late pregnancy in a cohort of Austrian women. Arch Gynecol Obstet. 2019;300:1325-1330.
- Swenson CW, DePorre JA, Haefner JK, et al. Postpartum depression screening and pelvic floor symptoms among women referred to a specialty postpartum perineal clinic. Am J Obstet Gynecol. 2018;218:335.e1-335.e6.
- Skinner EM, Dietz HP. Psychological and somatic sequelae of traumatic vaginal delivery: a literature review. Aust N Z J Obstet Gynaecol. 2015;55:309-314.
- Yohay D, Weintraub AY, Mauer-Perry N, et al. Prevalence and trends of pelvic floor disorders in late pregnancy and after delivery in a cohort of Israeli women using the PFDI-20. Eur J Obstet Gynecol Reprod Biol. 2016;200:35-39.
- Gregory WT, Sibai BM. Obstetrics and pelvic floor disorders. In: Walters M, Karram M, eds. Urogynecology and Reconstructive Pelvic Surgery. 4th ed. Philadelphia, PA: Saunders; 2015:224-237.
- Richter HE, Burgio KL, Brubaker L, et al; Pelvic Floor Disorders Network. Continence pessary compared with behavioral therapy or combined therapy for stress incontinence: a randomized controlled trial. Obstet Gynecol. 2010;115:609-617.
- Woodley SJ, Lawrenson P, Boyle R, et al. Pelvic floor muscle training for preventing and treating urinary and faecal incontinence in antenatal and postnatal women. Cochrane Database Syst Rev. 2020;6:CD007471.
- Foldspang A, Hvidman L, Mommsen S, et al. Risk of postpartum urinary incontinence associated with pregnancy and mode of delivery. Acta Obstet Gynecol Scand. 2004;83:923-927.
- Wieslander CK, Weinstein MM, Handa V, et al. Pregnancy in women with prior treatments for pelvic floor disorders. Female Pelvic Med Reconstr Surg. 2020;26:299-305.
- Thom DH, Rortveit G. Prevalence of postpartum urinary incontinence: a systematic review. Acta Obstet Gynecol Scand. 2010;89:1511-1522.
- MacArthur C, Wilson D, Herbison P, et al; Prolong Study Group. Urinary incontinence persisting after childbirth: extent, delivery history, and effects in a 12-year longitudinal cohort study. BJOG. 2016;123:1022-1029.
- Blake MR, Raker JM, Whelan K. Validity and reliability of the Bristol Stool Form Scale in healthy adults and patients with diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2016;44:693-703
- Orkin BA, Sinykin SB, Lloyd PC. The digital rectal examination scoring system (DRESS). Dis Colon Rectum. 2010;53:1656-1660.
- UpToDate. Repair of episiotomy and perineal lacerations associated with childbirth. 2020. https://www-uptodate-com .ccmain.ohionet.org/contents/repair-of-perineal-and-other -lacerations-associated-with-childbirth?search=repair%20 episiotomy&source=search_result&selectedTitle=1~150&usa ge_type=default&display_rank=1. Accessed February 28, 2021.
- Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 198: prevention and management of obstetric lacerations at vaginal delivery. Obstet Gynecol. 2018;132:e87-e102.
- Jangö H, Langhoff-Roos J, Rosthøj S, et al. Long-term anal incontinence after obstetric anal sphincter injury—does grade of tear matter? Am J Obstet Gynecol. 2018;218:232.e1-232.e10.
- O’Boyle AL, Woodman PJ, O’Boyle JD, et al. Pelvic organ support in nulliparous pregnant and nonpregnant women: a case control study. Am J Obstet Gynecol. 2002;187:99-102.
- Handa VL, Blomquist JL, McDermott KC, et al. Pelvic floor disorders after vaginal birth. Obstet Gynecol. 2012;119 (2, pt 1):233-239.
- Handa VL, Nygaard I, Kenton K, et al; Pelvic Floor Disorders Network. Pelvic organ support among primiparous women in the first year after childbirth. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20:1407-1411.
- O’Boyle AL, O’Boyle JD, Calhoun B, et al. Pelvic organ support in pregnancy and postpartum. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:69-72.
- Hagen S, Stark D, Glazener C, et al; POPPY Trial Collaborators. Individualised pelvic floor muscle training in women with pelvic organ prolapse (POPPY): a multicentre randomised controlled trial. Lancet. 2014;383:796-806.
- Von Bargen E, Haviland MJ, Chang OH, et al. Evaluation of postpartum pelvic floor physical therapy on obstetrical anal sphincter injury: a randomized controlled trial. Female Pelvic Med Reconstr Surg. 2020. doi: 10.1097/SPV.0000000000000849.
Pelvic floor disorders (PFDs) affect many pregnant and newly postpartum women. These conditions, including urinary incontinence, anal incontinence, and pelvic organ prolapse (POP), can be overshadowed by common pregnancy and postpartum concerns (TABLE 1).1 With the use of a few quick screening questions, however, PFDs easily can be identified in this at-risk population. Active management need not be delayed until after delivery for women experiencing bother, as options exist for women with PFDs during pregnancy as well as postpartum.

In this article, we discuss the common PFDs that obstetric clinicians face in the context of case scenarios and review how you can be better equipped to care for affected individuals.
CASE 1 Screening
A 30-year-old woman (G1P1) presents for her routine postpartum visit after an operative vaginal delivery with a second-degree laceration.
How would you screen this patient for PFDs?
Why screening for PFDs matters
While there are no validated PFD screening tools for this patient population, clinicians can ask a series of brief open-ended questions as part of the review of systems to efficiently evaluate for the common PFDs in peripartum patients (see “Screening questions to evaluate patients for peripartum pelvic floor disorders” below).
Pelvic floor disorders in the peripartum period can have a significant negative impact. In pregnancy, nearly half of women report psychological strain due to the presence of bowel, bladder, prolapse, or sexual dysfunction symptoms.2 Postpartum, PFDs have negative effects on overall health, well-being, and self-esteem, with significantly increased rates of postpartum depression in women who experience urinary incontinence.3,4 Proactively inquiring about PFD symptoms, providing anticipatory guidance, and recommending treatment options can positively impact a patient in multiple domains.
Sometimes during pregnancy or after having a baby, a woman experiences pelvic floor symptoms. Do you have any of the following?
- leakage with coughing, laughing, sneezing, or physical activity
- urgency to urinate or leakage due to urgency
- bulging or pressure within the vagina
- pain with intercourse
- accidental bowel leakage of stool or flatus
CASE 2 Stress urinary incontinence
A 27-year-old woman (G1P1) presents 2 months following spontaneous vaginal delivery with symptoms of urine leakage with laughing and running. Her urinary incontinence has been improving since delivery, but it continues to be bothersome.
What would you recommend for this patient?
Conservative SUI management strategies in pregnancy
Urinary tract symptoms are common in pregnancy, with up to 41.8% of women reporting urinary symptom distress in the third trimester.5 During pregnancy, estrogen and progesterone decrease urethral pressure that, together with increased intra-abdominal pressure from the gravid uterus, can cause or worsen stress urinary incontinence (SUI).6
During pregnancy, women should be offered conservative therapies for SUI. For women who can perform a pelvic floor contraction (a Kegel exercise), self-guided pelvic floor muscle exercises (PFMEs) may be helpful (see “Pelvic floor muscle exercises” below). We recommend that women start with 1 to 2 sets of 10 Kegel exercises per day and that they hold the squeeze for 2 to 3 seconds, working up to holding for 10 seconds. The goal is to strengthen and improve muscle control so that the Kegel squeeze can be paired with activities that cause SUI.
For women who are unable to perform a Kegel exercise or are not improving with a home PFME regimen, referral to pelvic floor physical therapy (PFPT) can be considered. While data support the efficacy of PFPT for SUI treatment in nonpregnant women,7 data are lacking on PFME in pregnancy.
In women without urinary incontinence, PFME in early pregnancy can prevent the onset of incontinence in late pregnancy and the postpartum period.8 By contrast, the same 2020 Cochrane Review found no evidence that antenatal pelvic floor muscle therapy in incontinent women decreases incontinence in mid- or late-pregnancy or in the postpartum period.8 As the quality of this evidence is very low and there is no evidence of harm with PFME, we continue to recommend it for women with bothersome SUI.
Incontinence pessaries or vaginal inserts (such as Poise Impressa bladder supports) can be helpful for SUI treatment. An incontinence pessary can be fitted in the office, and fitting kits are available for both. Pessaries can safely be used in pregnancy, but there are no data on the efficacy of pessaries for treating SUI in pregnancy. In nonpregnant women, evidence demonstrates 63% satisfaction 3 months post–pessary placement for SUI.7
We do not recommend invasive procedures for the treatment of SUI during pregnancy or in the first 6 months following delivery. There is no evidence that elective cesarean delivery prevents persistent SUI postpartum.9
To identify and engage the proper pelvic floor muscles:
- Insert a finger in the vagina and squeeze the vaginal muscles around your finger.
- Imagine you are sitting on a marble and have to pick it up with the vaginal muscles.
- Squeeze the muscles you would use to stop the flow of urine or hold back flatulence.
Perform sets of 10, 2 to 3 times per day as follows:
- Squeeze: Engage the pelvic floor muscles as described above; avoid performing Kegels while voiding.
- Hold: For 2 to 10 seconds; increase the duration to 10 seconds as able.
- Relax: Completely relax muscles before initating the next squeeze.
Reference
1. UpToDate. Patient education: pelvic muscle (Kegel) exercises (the basics). 2018. https://uptodatefree.ir/topic.htm?path=pelvic-muscle-kegel-exercises-the-basics. Accessed February 24, 2021.
Continue to: Managing SUI in the postpartum period...
Managing SUI in the postpartum period
After the first 6 months postpartum and exhaustion of conservative measures, we offer surgical interventions for women with persistent, bothersome incontinence. Surgery for SUI typically is not recommended until childbearing is complete, but it can be considered if the patient’s bother is significant.
For women with bothersome SUI who still desire future pregnancy, management options include periurethral bulking, a retropubic urethropexy (Burch procedure), or a midurethral sling procedure. Women who undergo an anti-incontinence procedure have an increased risk for urinary retention during a subsequent pregnancy.10 Most women with a midurethral sling will continue to be continent following an obstetric delivery.
Anticipatory guidance
At 3 months postpartum, the incidence of urinary incontinence is 6% to 40%, depending on parity and delivery type. Postpartum urinary incontinence is most common after instrumented vaginal delivery (32%) followed by spontaneous vaginal delivery (28%) and cesarean delivery (15%). The mean prevalence of any type of urinary incontinence is 33% at 3 months postpartum, and only small changes in the rate of urinary incontinence occur over the first postpartum year.11 While urinary incontinence is common postpartum, it should not be considered normal. We counsel that symptoms may improve spontaneously, but treatment can be initiated if the patient experiences significant bother.
A longitudinal cohort study that followed women from 3 months to 12 years postpartum found that, of women with urinary incontinence at 3 months postpartum, 76% continued to report incontinence at 12 years postpartum.12 We recommend that women be counseled that, even when symptoms resolve, they remain at increased risk for urinary incontinence in the future. Invasive therapies should be used to treat bothersome urinary incontinence, not to prevent future incontinence.
CASE 3 Fecal incontinence
A 24-year-old woman (G1P1) presents 3 weeks postpartum following a forceps-assisted vaginal delivery complicated by a 3c laceration. She reports fecal urgency, inability to control flatus, and once-daily fecal incontinence.
How would you evaluate these symptoms?
Steps in evaluation
The initial evaluation should include an inquiry regarding the patient’s stool consistency and bowel regimen. The Bristol stool form scale can be used to help patients describe their typical bowel movements (TABLE 2).13 During healing, the goal is to achieve a Bristol type 4 stool, both to avoid straining and to improve continence, as loose stool is the most difficult to control.

A physical examination can evaluate healing and sphincter integrity; it should include inspection of the distal vagina and perineal body and a digital rectal exam. Anal canal resting tone and squeeze strength should be evaluated, and the digital rectal examination scoring system (DRESS) can be useful for quantification (TABLE 3).14 Lack of tone at rest in the anterolateral portion of the sphincter complex can indicate an internal anal sphincter defect, as 80% of the resting tone comes from this muscle (FIGURE).15

The rectovaginal septum should be assessed given the increased risk of rectovaginal fistula in women with obstetric anal sphincter injury (OASI). The patient should be instructed to contract the anal sphincter, allowing evaluation of muscular contraction. Lack of contraction anteriolaterally may indicate external anal sphincter separation.
Continue to: Conservative options for improving fecal incontinence symptoms...
Conservative options for improving fecal incontinence symptoms
The patient can be counseled regarding stool bulking, first with insoluble fiber supplementation and cessation of stool softeners if she is incontinent of liquid stool. If these measures are not effective, use of a constipating agent, such as loperamide, can improve stool consistency and thereby decrease incontinence episodes. PFPT with biofeedback can be offered as well. While typically we do not recommend initiating PFPT before 6 weeks postpartum, so the initial phases of healing can occur, early referral enables the patient to avoid a delay in access to care.
The patient also can be counseled about a referral to a pelvic floor specialist for further evaluation. A variety of peripartum pelvic floor disorder clinics are being established by Female Pelvic Medicine and Reconstructive Surgery (FPMRS) physicians. These clinics provide the benefit of comprehensive care for pelvic floor disorders in this unique population.
When conservative measures fail. If a patient has persistent bowel control issues despite conservative measures, a referral to an FPMRS physician should be initiated.
Delivery route in future pregnancies
The risk of a subsequent OASI is low. While this means that many women can safely pursue a future vaginal delivery, a scheduled cesarean delivery is indicated for women with persistent bowel control issues, wound healing complications, and those who experienced psychological trauma from their delivery.16 We recommend a shared-decision making approach, reviewing modifiable and nonmodifiable risk factors to help determine whether or not a future vaginal birth is appropriate. It is important to highlight that a cesarean delivery does not protect against fecal incontinence in women with a history of OASI; however, there is benefit in preventing worsening of anal incontinence, if present.17
CASE 4 Uterovaginal prolapse
A 36-year-old woman (G3P3) presents for her routine postpartum visit at 6 weeks after a spontaneous vaginal delivery without lacerations. She reports a persistent feeling of vaginal pressure and fullness. She thinks she felt a bulge with wiping after a bowel movement.
What options are available for this patient?
Prolapse in the peripartum population
Previous studies have revealed an increased prevalence of POP in pregnant women on examination compared with their nulligravid counterparts (47.6% vs 0%).18 With the changes in the hormonal milieu in pregnancy, as well as the weight of the gravid uterus on the pelvic floor, it is not surprising that pregnancy may be the inciting event to expose even transient defects in pelvic organ support.19
It is well established that increasing parity and, to a lesser extent, larger babies are associated with increased risk for future POP and surgery for prolapse. In the first year postpartum, nearly one-third of women have stage 2 or greater prolapse on exam, with studies demonstrating an increased prevalence of postpartum POP in women who delivered vaginally compared with those who delivered by cesarean.20,21
Initial evaluation
Diagnosis can be made during a routine pelvic exam by having the patient perform a Valsalva maneuver while in the lithotomy position. Using half of a speculum permits evaluation of the anterior and posterior vaginal walls separately, and Valsalva during a bimanual exam can aid in evaluating descensus of the uterus and cervix.
Excellent free patient education resources available online through the American Urogynecologic Society and the International Urogynecological Association can be used to direct counseling.
Continue to: Treatments you can offer for POP...
Treatments you can offer for POP
For pregnant or postpartum patients with bothersome prolapse, initial management options include pessary fitting and/or PFPT referral. In pregnancy, women often can be successfully fitted with a pessary for POP; however, as expulsion is a common issue, selection of a stiffer or space-occupying device may be more efficacious.
Often, early onset POP in pregnancy resolves as the gravid uterus lifts out of the pelvis in the second trimester, at which time the pessary can be discontinued. In the postpartum period, a pessary fitting can be undertaken similarly to that in nonpregnant patients. While data are lacking in the peripartum population, evidence supports the positive impact of PFPT on improving POP symptom bother.22 Additionally, for postpartum women who experience OASI, PFPT can produce significant improvement in subjective POP and associated bother.23
Impact of future childbearing wishes on treatment
The desire for future childbearing does not preclude treatment of patients experiencing bother from POP after conservative management options have failed. Both vaginal native tissue and mesh-augmented uterine-sparing repairs are performed by many FPMRS specialists and are associated with good outcomes. As with SUI, we do not recommend invasive treatment for POP during pregnancy or before 6 months postpartum.
In conclusion
Obstetric specialists play an essential role in caring for women with PFDs in the peripartum period. Basic evaluation, counseling, and management can be initiated using many of the resources already available in an obstetric ambulatory practice. Important adjunctive resources include those available for both providers and patients through the American Urogynecologic Society and the International Urogynecological Association. In addition, clinicians can partner with pelvic floor specialists through the growing number of FPMRS-run peripartum pelvic floor disorder clinics across the country and pelvic floor physical therapists.
If these specialty clinics and therapists are not available in your area, FPMRS specialists, urologists, gastroenterologists, and/or colorectal surgeons can aid in patient diagnosis and management to reach the ultimate goal of improving PFDs in this at-risk population. ●
Pelvic floor disorders (PFDs) affect many pregnant and newly postpartum women. These conditions, including urinary incontinence, anal incontinence, and pelvic organ prolapse (POP), can be overshadowed by common pregnancy and postpartum concerns (TABLE 1).1 With the use of a few quick screening questions, however, PFDs easily can be identified in this at-risk population. Active management need not be delayed until after delivery for women experiencing bother, as options exist for women with PFDs during pregnancy as well as postpartum.

In this article, we discuss the common PFDs that obstetric clinicians face in the context of case scenarios and review how you can be better equipped to care for affected individuals.
CASE 1 Screening
A 30-year-old woman (G1P1) presents for her routine postpartum visit after an operative vaginal delivery with a second-degree laceration.
How would you screen this patient for PFDs?
Why screening for PFDs matters
While there are no validated PFD screening tools for this patient population, clinicians can ask a series of brief open-ended questions as part of the review of systems to efficiently evaluate for the common PFDs in peripartum patients (see “Screening questions to evaluate patients for peripartum pelvic floor disorders” below).
Pelvic floor disorders in the peripartum period can have a significant negative impact. In pregnancy, nearly half of women report psychological strain due to the presence of bowel, bladder, prolapse, or sexual dysfunction symptoms.2 Postpartum, PFDs have negative effects on overall health, well-being, and self-esteem, with significantly increased rates of postpartum depression in women who experience urinary incontinence.3,4 Proactively inquiring about PFD symptoms, providing anticipatory guidance, and recommending treatment options can positively impact a patient in multiple domains.
Sometimes during pregnancy or after having a baby, a woman experiences pelvic floor symptoms. Do you have any of the following?
- leakage with coughing, laughing, sneezing, or physical activity
- urgency to urinate or leakage due to urgency
- bulging or pressure within the vagina
- pain with intercourse
- accidental bowel leakage of stool or flatus
CASE 2 Stress urinary incontinence
A 27-year-old woman (G1P1) presents 2 months following spontaneous vaginal delivery with symptoms of urine leakage with laughing and running. Her urinary incontinence has been improving since delivery, but it continues to be bothersome.
What would you recommend for this patient?
Conservative SUI management strategies in pregnancy
Urinary tract symptoms are common in pregnancy, with up to 41.8% of women reporting urinary symptom distress in the third trimester.5 During pregnancy, estrogen and progesterone decrease urethral pressure that, together with increased intra-abdominal pressure from the gravid uterus, can cause or worsen stress urinary incontinence (SUI).6
During pregnancy, women should be offered conservative therapies for SUI. For women who can perform a pelvic floor contraction (a Kegel exercise), self-guided pelvic floor muscle exercises (PFMEs) may be helpful (see “Pelvic floor muscle exercises” below). We recommend that women start with 1 to 2 sets of 10 Kegel exercises per day and that they hold the squeeze for 2 to 3 seconds, working up to holding for 10 seconds. The goal is to strengthen and improve muscle control so that the Kegel squeeze can be paired with activities that cause SUI.
For women who are unable to perform a Kegel exercise or are not improving with a home PFME regimen, referral to pelvic floor physical therapy (PFPT) can be considered. While data support the efficacy of PFPT for SUI treatment in nonpregnant women,7 data are lacking on PFME in pregnancy.
In women without urinary incontinence, PFME in early pregnancy can prevent the onset of incontinence in late pregnancy and the postpartum period.8 By contrast, the same 2020 Cochrane Review found no evidence that antenatal pelvic floor muscle therapy in incontinent women decreases incontinence in mid- or late-pregnancy or in the postpartum period.8 As the quality of this evidence is very low and there is no evidence of harm with PFME, we continue to recommend it for women with bothersome SUI.
Incontinence pessaries or vaginal inserts (such as Poise Impressa bladder supports) can be helpful for SUI treatment. An incontinence pessary can be fitted in the office, and fitting kits are available for both. Pessaries can safely be used in pregnancy, but there are no data on the efficacy of pessaries for treating SUI in pregnancy. In nonpregnant women, evidence demonstrates 63% satisfaction 3 months post–pessary placement for SUI.7
We do not recommend invasive procedures for the treatment of SUI during pregnancy or in the first 6 months following delivery. There is no evidence that elective cesarean delivery prevents persistent SUI postpartum.9
To identify and engage the proper pelvic floor muscles:
- Insert a finger in the vagina and squeeze the vaginal muscles around your finger.
- Imagine you are sitting on a marble and have to pick it up with the vaginal muscles.
- Squeeze the muscles you would use to stop the flow of urine or hold back flatulence.
Perform sets of 10, 2 to 3 times per day as follows:
- Squeeze: Engage the pelvic floor muscles as described above; avoid performing Kegels while voiding.
- Hold: For 2 to 10 seconds; increase the duration to 10 seconds as able.
- Relax: Completely relax muscles before initating the next squeeze.
Reference
1. UpToDate. Patient education: pelvic muscle (Kegel) exercises (the basics). 2018. https://uptodatefree.ir/topic.htm?path=pelvic-muscle-kegel-exercises-the-basics. Accessed February 24, 2021.
Continue to: Managing SUI in the postpartum period...
Managing SUI in the postpartum period
After the first 6 months postpartum and exhaustion of conservative measures, we offer surgical interventions for women with persistent, bothersome incontinence. Surgery for SUI typically is not recommended until childbearing is complete, but it can be considered if the patient’s bother is significant.
For women with bothersome SUI who still desire future pregnancy, management options include periurethral bulking, a retropubic urethropexy (Burch procedure), or a midurethral sling procedure. Women who undergo an anti-incontinence procedure have an increased risk for urinary retention during a subsequent pregnancy.10 Most women with a midurethral sling will continue to be continent following an obstetric delivery.
Anticipatory guidance
At 3 months postpartum, the incidence of urinary incontinence is 6% to 40%, depending on parity and delivery type. Postpartum urinary incontinence is most common after instrumented vaginal delivery (32%) followed by spontaneous vaginal delivery (28%) and cesarean delivery (15%). The mean prevalence of any type of urinary incontinence is 33% at 3 months postpartum, and only small changes in the rate of urinary incontinence occur over the first postpartum year.11 While urinary incontinence is common postpartum, it should not be considered normal. We counsel that symptoms may improve spontaneously, but treatment can be initiated if the patient experiences significant bother.
A longitudinal cohort study that followed women from 3 months to 12 years postpartum found that, of women with urinary incontinence at 3 months postpartum, 76% continued to report incontinence at 12 years postpartum.12 We recommend that women be counseled that, even when symptoms resolve, they remain at increased risk for urinary incontinence in the future. Invasive therapies should be used to treat bothersome urinary incontinence, not to prevent future incontinence.
CASE 3 Fecal incontinence
A 24-year-old woman (G1P1) presents 3 weeks postpartum following a forceps-assisted vaginal delivery complicated by a 3c laceration. She reports fecal urgency, inability to control flatus, and once-daily fecal incontinence.
How would you evaluate these symptoms?
Steps in evaluation
The initial evaluation should include an inquiry regarding the patient’s stool consistency and bowel regimen. The Bristol stool form scale can be used to help patients describe their typical bowel movements (TABLE 2).13 During healing, the goal is to achieve a Bristol type 4 stool, both to avoid straining and to improve continence, as loose stool is the most difficult to control.

A physical examination can evaluate healing and sphincter integrity; it should include inspection of the distal vagina and perineal body and a digital rectal exam. Anal canal resting tone and squeeze strength should be evaluated, and the digital rectal examination scoring system (DRESS) can be useful for quantification (TABLE 3).14 Lack of tone at rest in the anterolateral portion of the sphincter complex can indicate an internal anal sphincter defect, as 80% of the resting tone comes from this muscle (FIGURE).15

The rectovaginal septum should be assessed given the increased risk of rectovaginal fistula in women with obstetric anal sphincter injury (OASI). The patient should be instructed to contract the anal sphincter, allowing evaluation of muscular contraction. Lack of contraction anteriolaterally may indicate external anal sphincter separation.
Continue to: Conservative options for improving fecal incontinence symptoms...
Conservative options for improving fecal incontinence symptoms
The patient can be counseled regarding stool bulking, first with insoluble fiber supplementation and cessation of stool softeners if she is incontinent of liquid stool. If these measures are not effective, use of a constipating agent, such as loperamide, can improve stool consistency and thereby decrease incontinence episodes. PFPT with biofeedback can be offered as well. While typically we do not recommend initiating PFPT before 6 weeks postpartum, so the initial phases of healing can occur, early referral enables the patient to avoid a delay in access to care.
The patient also can be counseled about a referral to a pelvic floor specialist for further evaluation. A variety of peripartum pelvic floor disorder clinics are being established by Female Pelvic Medicine and Reconstructive Surgery (FPMRS) physicians. These clinics provide the benefit of comprehensive care for pelvic floor disorders in this unique population.
When conservative measures fail. If a patient has persistent bowel control issues despite conservative measures, a referral to an FPMRS physician should be initiated.
Delivery route in future pregnancies
The risk of a subsequent OASI is low. While this means that many women can safely pursue a future vaginal delivery, a scheduled cesarean delivery is indicated for women with persistent bowel control issues, wound healing complications, and those who experienced psychological trauma from their delivery.16 We recommend a shared-decision making approach, reviewing modifiable and nonmodifiable risk factors to help determine whether or not a future vaginal birth is appropriate. It is important to highlight that a cesarean delivery does not protect against fecal incontinence in women with a history of OASI; however, there is benefit in preventing worsening of anal incontinence, if present.17
CASE 4 Uterovaginal prolapse
A 36-year-old woman (G3P3) presents for her routine postpartum visit at 6 weeks after a spontaneous vaginal delivery without lacerations. She reports a persistent feeling of vaginal pressure and fullness. She thinks she felt a bulge with wiping after a bowel movement.
What options are available for this patient?
Prolapse in the peripartum population
Previous studies have revealed an increased prevalence of POP in pregnant women on examination compared with their nulligravid counterparts (47.6% vs 0%).18 With the changes in the hormonal milieu in pregnancy, as well as the weight of the gravid uterus on the pelvic floor, it is not surprising that pregnancy may be the inciting event to expose even transient defects in pelvic organ support.19
It is well established that increasing parity and, to a lesser extent, larger babies are associated with increased risk for future POP and surgery for prolapse. In the first year postpartum, nearly one-third of women have stage 2 or greater prolapse on exam, with studies demonstrating an increased prevalence of postpartum POP in women who delivered vaginally compared with those who delivered by cesarean.20,21
Initial evaluation
Diagnosis can be made during a routine pelvic exam by having the patient perform a Valsalva maneuver while in the lithotomy position. Using half of a speculum permits evaluation of the anterior and posterior vaginal walls separately, and Valsalva during a bimanual exam can aid in evaluating descensus of the uterus and cervix.
Excellent free patient education resources available online through the American Urogynecologic Society and the International Urogynecological Association can be used to direct counseling.
Continue to: Treatments you can offer for POP...
Treatments you can offer for POP
For pregnant or postpartum patients with bothersome prolapse, initial management options include pessary fitting and/or PFPT referral. In pregnancy, women often can be successfully fitted with a pessary for POP; however, as expulsion is a common issue, selection of a stiffer or space-occupying device may be more efficacious.
Often, early onset POP in pregnancy resolves as the gravid uterus lifts out of the pelvis in the second trimester, at which time the pessary can be discontinued. In the postpartum period, a pessary fitting can be undertaken similarly to that in nonpregnant patients. While data are lacking in the peripartum population, evidence supports the positive impact of PFPT on improving POP symptom bother.22 Additionally, for postpartum women who experience OASI, PFPT can produce significant improvement in subjective POP and associated bother.23
Impact of future childbearing wishes on treatment
The desire for future childbearing does not preclude treatment of patients experiencing bother from POP after conservative management options have failed. Both vaginal native tissue and mesh-augmented uterine-sparing repairs are performed by many FPMRS specialists and are associated with good outcomes. As with SUI, we do not recommend invasive treatment for POP during pregnancy or before 6 months postpartum.
In conclusion
Obstetric specialists play an essential role in caring for women with PFDs in the peripartum period. Basic evaluation, counseling, and management can be initiated using many of the resources already available in an obstetric ambulatory practice. Important adjunctive resources include those available for both providers and patients through the American Urogynecologic Society and the International Urogynecological Association. In addition, clinicians can partner with pelvic floor specialists through the growing number of FPMRS-run peripartum pelvic floor disorder clinics across the country and pelvic floor physical therapists.
If these specialty clinics and therapists are not available in your area, FPMRS specialists, urologists, gastroenterologists, and/or colorectal surgeons can aid in patient diagnosis and management to reach the ultimate goal of improving PFDs in this at-risk population. ●
- Madsen AM, Hickman LC, Propst K. Recognition and management of pelvic floor disorders in pregnancy and the postpartum period. Obstet Gynecol Clin North Am. Forthcoming 2021.
- Bodner-Adler B, Kimberger O, Laml T, et al. Prevalence and risk factors for pelvic floor disorders during early and late pregnancy in a cohort of Austrian women. Arch Gynecol Obstet. 2019;300:1325-1330.
- Swenson CW, DePorre JA, Haefner JK, et al. Postpartum depression screening and pelvic floor symptoms among women referred to a specialty postpartum perineal clinic. Am J Obstet Gynecol. 2018;218:335.e1-335.e6.
- Skinner EM, Dietz HP. Psychological and somatic sequelae of traumatic vaginal delivery: a literature review. Aust N Z J Obstet Gynaecol. 2015;55:309-314.
- Yohay D, Weintraub AY, Mauer-Perry N, et al. Prevalence and trends of pelvic floor disorders in late pregnancy and after delivery in a cohort of Israeli women using the PFDI-20. Eur J Obstet Gynecol Reprod Biol. 2016;200:35-39.
- Gregory WT, Sibai BM. Obstetrics and pelvic floor disorders. In: Walters M, Karram M, eds. Urogynecology and Reconstructive Pelvic Surgery. 4th ed. Philadelphia, PA: Saunders; 2015:224-237.
- Richter HE, Burgio KL, Brubaker L, et al; Pelvic Floor Disorders Network. Continence pessary compared with behavioral therapy or combined therapy for stress incontinence: a randomized controlled trial. Obstet Gynecol. 2010;115:609-617.
- Woodley SJ, Lawrenson P, Boyle R, et al. Pelvic floor muscle training for preventing and treating urinary and faecal incontinence in antenatal and postnatal women. Cochrane Database Syst Rev. 2020;6:CD007471.
- Foldspang A, Hvidman L, Mommsen S, et al. Risk of postpartum urinary incontinence associated with pregnancy and mode of delivery. Acta Obstet Gynecol Scand. 2004;83:923-927.
- Wieslander CK, Weinstein MM, Handa V, et al. Pregnancy in women with prior treatments for pelvic floor disorders. Female Pelvic Med Reconstr Surg. 2020;26:299-305.
- Thom DH, Rortveit G. Prevalence of postpartum urinary incontinence: a systematic review. Acta Obstet Gynecol Scand. 2010;89:1511-1522.
- MacArthur C, Wilson D, Herbison P, et al; Prolong Study Group. Urinary incontinence persisting after childbirth: extent, delivery history, and effects in a 12-year longitudinal cohort study. BJOG. 2016;123:1022-1029.
- Blake MR, Raker JM, Whelan K. Validity and reliability of the Bristol Stool Form Scale in healthy adults and patients with diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2016;44:693-703
- Orkin BA, Sinykin SB, Lloyd PC. The digital rectal examination scoring system (DRESS). Dis Colon Rectum. 2010;53:1656-1660.
- UpToDate. Repair of episiotomy and perineal lacerations associated with childbirth. 2020. https://www-uptodate-com .ccmain.ohionet.org/contents/repair-of-perineal-and-other -lacerations-associated-with-childbirth?search=repair%20 episiotomy&source=search_result&selectedTitle=1~150&usa ge_type=default&display_rank=1. Accessed February 28, 2021.
- Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 198: prevention and management of obstetric lacerations at vaginal delivery. Obstet Gynecol. 2018;132:e87-e102.
- Jangö H, Langhoff-Roos J, Rosthøj S, et al. Long-term anal incontinence after obstetric anal sphincter injury—does grade of tear matter? Am J Obstet Gynecol. 2018;218:232.e1-232.e10.
- O’Boyle AL, Woodman PJ, O’Boyle JD, et al. Pelvic organ support in nulliparous pregnant and nonpregnant women: a case control study. Am J Obstet Gynecol. 2002;187:99-102.
- Handa VL, Blomquist JL, McDermott KC, et al. Pelvic floor disorders after vaginal birth. Obstet Gynecol. 2012;119 (2, pt 1):233-239.
- Handa VL, Nygaard I, Kenton K, et al; Pelvic Floor Disorders Network. Pelvic organ support among primiparous women in the first year after childbirth. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20:1407-1411.
- O’Boyle AL, O’Boyle JD, Calhoun B, et al. Pelvic organ support in pregnancy and postpartum. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:69-72.
- Hagen S, Stark D, Glazener C, et al; POPPY Trial Collaborators. Individualised pelvic floor muscle training in women with pelvic organ prolapse (POPPY): a multicentre randomised controlled trial. Lancet. 2014;383:796-806.
- Von Bargen E, Haviland MJ, Chang OH, et al. Evaluation of postpartum pelvic floor physical therapy on obstetrical anal sphincter injury: a randomized controlled trial. Female Pelvic Med Reconstr Surg. 2020. doi: 10.1097/SPV.0000000000000849.
- Madsen AM, Hickman LC, Propst K. Recognition and management of pelvic floor disorders in pregnancy and the postpartum period. Obstet Gynecol Clin North Am. Forthcoming 2021.
- Bodner-Adler B, Kimberger O, Laml T, et al. Prevalence and risk factors for pelvic floor disorders during early and late pregnancy in a cohort of Austrian women. Arch Gynecol Obstet. 2019;300:1325-1330.
- Swenson CW, DePorre JA, Haefner JK, et al. Postpartum depression screening and pelvic floor symptoms among women referred to a specialty postpartum perineal clinic. Am J Obstet Gynecol. 2018;218:335.e1-335.e6.
- Skinner EM, Dietz HP. Psychological and somatic sequelae of traumatic vaginal delivery: a literature review. Aust N Z J Obstet Gynaecol. 2015;55:309-314.
- Yohay D, Weintraub AY, Mauer-Perry N, et al. Prevalence and trends of pelvic floor disorders in late pregnancy and after delivery in a cohort of Israeli women using the PFDI-20. Eur J Obstet Gynecol Reprod Biol. 2016;200:35-39.
- Gregory WT, Sibai BM. Obstetrics and pelvic floor disorders. In: Walters M, Karram M, eds. Urogynecology and Reconstructive Pelvic Surgery. 4th ed. Philadelphia, PA: Saunders; 2015:224-237.
- Richter HE, Burgio KL, Brubaker L, et al; Pelvic Floor Disorders Network. Continence pessary compared with behavioral therapy or combined therapy for stress incontinence: a randomized controlled trial. Obstet Gynecol. 2010;115:609-617.
- Woodley SJ, Lawrenson P, Boyle R, et al. Pelvic floor muscle training for preventing and treating urinary and faecal incontinence in antenatal and postnatal women. Cochrane Database Syst Rev. 2020;6:CD007471.
- Foldspang A, Hvidman L, Mommsen S, et al. Risk of postpartum urinary incontinence associated with pregnancy and mode of delivery. Acta Obstet Gynecol Scand. 2004;83:923-927.
- Wieslander CK, Weinstein MM, Handa V, et al. Pregnancy in women with prior treatments for pelvic floor disorders. Female Pelvic Med Reconstr Surg. 2020;26:299-305.
- Thom DH, Rortveit G. Prevalence of postpartum urinary incontinence: a systematic review. Acta Obstet Gynecol Scand. 2010;89:1511-1522.
- MacArthur C, Wilson D, Herbison P, et al; Prolong Study Group. Urinary incontinence persisting after childbirth: extent, delivery history, and effects in a 12-year longitudinal cohort study. BJOG. 2016;123:1022-1029.
- Blake MR, Raker JM, Whelan K. Validity and reliability of the Bristol Stool Form Scale in healthy adults and patients with diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2016;44:693-703
- Orkin BA, Sinykin SB, Lloyd PC. The digital rectal examination scoring system (DRESS). Dis Colon Rectum. 2010;53:1656-1660.
- UpToDate. Repair of episiotomy and perineal lacerations associated with childbirth. 2020. https://www-uptodate-com .ccmain.ohionet.org/contents/repair-of-perineal-and-other -lacerations-associated-with-childbirth?search=repair%20 episiotomy&source=search_result&selectedTitle=1~150&usa ge_type=default&display_rank=1. Accessed February 28, 2021.
- Committee on Practice Bulletins–Obstetrics. ACOG practice bulletin no. 198: prevention and management of obstetric lacerations at vaginal delivery. Obstet Gynecol. 2018;132:e87-e102.
- Jangö H, Langhoff-Roos J, Rosthøj S, et al. Long-term anal incontinence after obstetric anal sphincter injury—does grade of tear matter? Am J Obstet Gynecol. 2018;218:232.e1-232.e10.
- O’Boyle AL, Woodman PJ, O’Boyle JD, et al. Pelvic organ support in nulliparous pregnant and nonpregnant women: a case control study. Am J Obstet Gynecol. 2002;187:99-102.
- Handa VL, Blomquist JL, McDermott KC, et al. Pelvic floor disorders after vaginal birth. Obstet Gynecol. 2012;119 (2, pt 1):233-239.
- Handa VL, Nygaard I, Kenton K, et al; Pelvic Floor Disorders Network. Pelvic organ support among primiparous women in the first year after childbirth. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20:1407-1411.
- O’Boyle AL, O’Boyle JD, Calhoun B, et al. Pelvic organ support in pregnancy and postpartum. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:69-72.
- Hagen S, Stark D, Glazener C, et al; POPPY Trial Collaborators. Individualised pelvic floor muscle training in women with pelvic organ prolapse (POPPY): a multicentre randomised controlled trial. Lancet. 2014;383:796-806.
- Von Bargen E, Haviland MJ, Chang OH, et al. Evaluation of postpartum pelvic floor physical therapy on obstetrical anal sphincter injury: a randomized controlled trial. Female Pelvic Med Reconstr Surg. 2020. doi: 10.1097/SPV.0000000000000849.
For which patients is maternal oxygen supplementation of value?
Raghuraman N, Temming LA, Doering MM, et al. Maternal oxygen supplementation compared with room air for intrauterine resuscitation: a systematic review and meta-analysis. JAMA Pediatr. January 4, 2021. doi:10.1001/jamapediatrics.2020.5351.
EXPERT COMMENTARY
Maternal oxygen supplementation is widely used in labor for nonreassuring fetal heart rate (FHR) tracings, although its efficacy is uncertain for preventing fetal acidosis, operative intervention, or sequelae of neonatal encephalopathy. Recently, Raghuraman and colleagues reported the results of a systematic review and meta-analysis that included 16 randomized controlled trials. A total of 1,078 women were included in the oxygen group and 974 in the room air group. The primary outcome was umbilical artery pH; 14 trials reported on this outcome.
After analyzing the pooled and stratified results of the effect of maternal oxygen supplementation versus room air on umbilical artery gas measures, the authors concluded that peripartum oxygen supplementation is not associated with clinically relevant improvement in umbilical artery pH. They acknowledged, however, that the published studies were heterogeneous, lacked data on the association of oxygen supplementation with neonatal outcome, and did not assess oxygen use for abnormal FHR tracings, except for one trial with category II FHR tracings.
Effects of O2 supplementation
As maternal arterial hemoglobin is nearly saturated under normal conditions, maternal hyperoxia produces only modest increases in umbilical vein P
Fetal hypoxemia and acidosis can result from an interruption or an impairment of the mother-to-placenta-to-fetus oxygen pathway. With some interruptions of the oxygen pathway, such as placental abruption and complete cord occlusion–induced bradycardia, there would be less impact of maternal hyperoxia. By contrast, with other oxygen pathway impairments, such as reduced oxygen transfer with placental insufficiency, maternal hyperoxia can be of greater value by increasing maternal uterine artery and vein P
Continue to: Circumstances that may benefit from O2 supplementation...
Circumstances that may benefit from O2 supplementation
Late FHR decelerations reflect impairment of oxygen transfer and thus represent the heart rate pattern that is most likely to benefit from maternal hyperoxia. However, recurrent late decelerations occur in less than 2% of low-risk patients in labor,3 and severe levels of acidosis (umbilical artery pH <7.0 or base deficit [BD] ≥12 mmol/L) occur in only 1% to 2% of near-term or term deliveries.4,5
Variable decelerations also reflect fetal hypoxia and are much more common than late decelerations, so they also may benefit from O2 supplementation. Regardless, O2 supplementation should be seen only as a temporizing strategy while other resuscitative actions are initiated, including preparation for operative delivery, if indicated.
In a prior study by Raghuraman and colleagues (1 of only 4 studies that met selection criteria of oxygen supplementation for patients in labor), newborns of patients not receiving oxygen demonstrated 95% confidence limits of umbilical artery pH (7.24–7.28) and BD (2.9–4.3) well within the normal range.6 Thus, the low prevalence of cases in which a benefit might be anticipated and the low incidence of severe acidosis challenges the design of prospective studies to detect statistically and clinically significant changes in blood gas measures and newborn outcomes.
The normal mild fetal acidosis that develops during labor is likely a result of recurrent interruption of uterine placental blood flow during uterine contractions7 and is unlikely to benefit from maternal hyperoxia. Similarly, as placental oxygen transfer is predominantly flow rather than diffusion limited,8 oxygen supplementation is unlikely to improve severe variable FHR decelerations. Thus, a randomized study of hyperoxia in unselected laboring patients is unlikely to have a measurable effect on clinically significant acidosis.
Oxygen transport pathway guides treatment
For the present, an understanding of oxygen transport can guide clinical oxygen use. Thus, mothers with relative hypoxemia will unquestionably benefit with supplemental oxygen administration. Similarly, fetuses at risk for placental dysfunction (for example, growth restriction, postterm) and particularly those manifesting evidence of impaired oxygen transport (that is, late decelerations) may be most likely to benefit from the increased O2 gradient. For patients with reduced maternal uterine perfusion (such as hypotension or hypovolemia), pressors and/or fluid volume are likely to be more effective, while amnioinfusion is of greater value for umbilical cord compression patterns. A reduction in uterine activity may be of benefit to all fetuses exhibiting compromise. Due to the modest impact on fetal oxygen content, maternal hyperoxia does not produce significant fetal oxidative stress as measured by fetal malondialdehyde levels.
In view of the lack of demonstrated adverse effects of maternal supplemental oxygen, clinicians should not hesitate to use it. However, clinicians should recognize that supplemental oxygen is likely to be of value only in patients with significant impairment in the oxygen pathway, and they should choose additional intrauterine resuscitative measures focused on the etiology.
MICHAEL G. ROSS, MD, MPH,
AND BRYAN S. RICHARDSON, MD
- McNanley T, Woods J. Placental physiology. Glob Libr Women’s Med. (ISSN: 1756-2228). 2008. doi: 10.3843 /GLOWM.10195.
- Richardson BS. Fetal adaptive responses to asphyxia. Clin Perinatol. 1989;16:595-611.
- Sameshima H, Ikenoue T. Predictive value of late decelerations of fetal acidemia in unselective low-risk pregnancies. Am J Perinatol. 2005;22:19-23.
- Yeh P, Emary K, Impey L. The relationship between umbilical cord arterial pH and serious adverse neonatal outcome: analysis of 51,519 consecutive validated samples. BJOG. 2012;119:824-831.
- Kelly R, Ramaiah SM, Sheridan H, et al. Dose-dependent relationship between acidosis at birth and likelihood of death or cerebral palsy. Arch Dis Child Fetal Neonatal Ed. 2018;103:F567-F572.
- Raghuraman N, Wan L, Temming LA, et al. Effect of oxygen vs room air on intrauterine fetal resuscitation: a randomized noninferiority clinical trial. JAMA Pediatr. 2018;172:818-823.
- Ramsey EM, Corner JW Jr, Donner MW. Serial and cineradioangiographic visualization of maternal circulation in the primate (hemochorial) placenta. Am J Obstet Gynecol. 1963;86:213-225.
- Nye GA, Ingram E, Johnstone ED, et al. Human placental oxygenation in late gestation: experimental and theoretical approaches. J Physiol. 2018;596:5523-5534.
Raghuraman N, Temming LA, Doering MM, et al. Maternal oxygen supplementation compared with room air for intrauterine resuscitation: a systematic review and meta-analysis. JAMA Pediatr. January 4, 2021. doi:10.1001/jamapediatrics.2020.5351.
EXPERT COMMENTARY
Maternal oxygen supplementation is widely used in labor for nonreassuring fetal heart rate (FHR) tracings, although its efficacy is uncertain for preventing fetal acidosis, operative intervention, or sequelae of neonatal encephalopathy. Recently, Raghuraman and colleagues reported the results of a systematic review and meta-analysis that included 16 randomized controlled trials. A total of 1,078 women were included in the oxygen group and 974 in the room air group. The primary outcome was umbilical artery pH; 14 trials reported on this outcome.
After analyzing the pooled and stratified results of the effect of maternal oxygen supplementation versus room air on umbilical artery gas measures, the authors concluded that peripartum oxygen supplementation is not associated with clinically relevant improvement in umbilical artery pH. They acknowledged, however, that the published studies were heterogeneous, lacked data on the association of oxygen supplementation with neonatal outcome, and did not assess oxygen use for abnormal FHR tracings, except for one trial with category II FHR tracings.
Effects of O2 supplementation
As maternal arterial hemoglobin is nearly saturated under normal conditions, maternal hyperoxia produces only modest increases in umbilical vein P

Fetal hypoxemia and acidosis can result from an interruption or an impairment of the mother-to-placenta-to-fetus oxygen pathway. With some interruptions of the oxygen pathway, such as placental abruption and complete cord occlusion–induced bradycardia, there would be less impact of maternal hyperoxia. By contrast, with other oxygen pathway impairments, such as reduced oxygen transfer with placental insufficiency, maternal hyperoxia can be of greater value by increasing maternal uterine artery and vein P
Continue to: Circumstances that may benefit from O2 supplementation...
Circumstances that may benefit from O2 supplementation
Late FHR decelerations reflect impairment of oxygen transfer and thus represent the heart rate pattern that is most likely to benefit from maternal hyperoxia. However, recurrent late decelerations occur in less than 2% of low-risk patients in labor,3 and severe levels of acidosis (umbilical artery pH <7.0 or base deficit [BD] ≥12 mmol/L) occur in only 1% to 2% of near-term or term deliveries.4,5
Variable decelerations also reflect fetal hypoxia and are much more common than late decelerations, so they also may benefit from O2 supplementation. Regardless, O2 supplementation should be seen only as a temporizing strategy while other resuscitative actions are initiated, including preparation for operative delivery, if indicated.
In a prior study by Raghuraman and colleagues (1 of only 4 studies that met selection criteria of oxygen supplementation for patients in labor), newborns of patients not receiving oxygen demonstrated 95% confidence limits of umbilical artery pH (7.24–7.28) and BD (2.9–4.3) well within the normal range.6 Thus, the low prevalence of cases in which a benefit might be anticipated and the low incidence of severe acidosis challenges the design of prospective studies to detect statistically and clinically significant changes in blood gas measures and newborn outcomes.
The normal mild fetal acidosis that develops during labor is likely a result of recurrent interruption of uterine placental blood flow during uterine contractions7 and is unlikely to benefit from maternal hyperoxia. Similarly, as placental oxygen transfer is predominantly flow rather than diffusion limited,8 oxygen supplementation is unlikely to improve severe variable FHR decelerations. Thus, a randomized study of hyperoxia in unselected laboring patients is unlikely to have a measurable effect on clinically significant acidosis.
Oxygen transport pathway guides treatment
For the present, an understanding of oxygen transport can guide clinical oxygen use. Thus, mothers with relative hypoxemia will unquestionably benefit with supplemental oxygen administration. Similarly, fetuses at risk for placental dysfunction (for example, growth restriction, postterm) and particularly those manifesting evidence of impaired oxygen transport (that is, late decelerations) may be most likely to benefit from the increased O2 gradient. For patients with reduced maternal uterine perfusion (such as hypotension or hypovolemia), pressors and/or fluid volume are likely to be more effective, while amnioinfusion is of greater value for umbilical cord compression patterns. A reduction in uterine activity may be of benefit to all fetuses exhibiting compromise. Due to the modest impact on fetal oxygen content, maternal hyperoxia does not produce significant fetal oxidative stress as measured by fetal malondialdehyde levels.
In view of the lack of demonstrated adverse effects of maternal supplemental oxygen, clinicians should not hesitate to use it. However, clinicians should recognize that supplemental oxygen is likely to be of value only in patients with significant impairment in the oxygen pathway, and they should choose additional intrauterine resuscitative measures focused on the etiology.
MICHAEL G. ROSS, MD, MPH,
AND BRYAN S. RICHARDSON, MD
Raghuraman N, Temming LA, Doering MM, et al. Maternal oxygen supplementation compared with room air for intrauterine resuscitation: a systematic review and meta-analysis. JAMA Pediatr. January 4, 2021. doi:10.1001/jamapediatrics.2020.5351.
EXPERT COMMENTARY
Maternal oxygen supplementation is widely used in labor for nonreassuring fetal heart rate (FHR) tracings, although its efficacy is uncertain for preventing fetal acidosis, operative intervention, or sequelae of neonatal encephalopathy. Recently, Raghuraman and colleagues reported the results of a systematic review and meta-analysis that included 16 randomized controlled trials. A total of 1,078 women were included in the oxygen group and 974 in the room air group. The primary outcome was umbilical artery pH; 14 trials reported on this outcome.
After analyzing the pooled and stratified results of the effect of maternal oxygen supplementation versus room air on umbilical artery gas measures, the authors concluded that peripartum oxygen supplementation is not associated with clinically relevant improvement in umbilical artery pH. They acknowledged, however, that the published studies were heterogeneous, lacked data on the association of oxygen supplementation with neonatal outcome, and did not assess oxygen use for abnormal FHR tracings, except for one trial with category II FHR tracings.
Effects of O2 supplementation
As maternal arterial hemoglobin is nearly saturated under normal conditions, maternal hyperoxia produces only modest increases in umbilical vein P

Fetal hypoxemia and acidosis can result from an interruption or an impairment of the mother-to-placenta-to-fetus oxygen pathway. With some interruptions of the oxygen pathway, such as placental abruption and complete cord occlusion–induced bradycardia, there would be less impact of maternal hyperoxia. By contrast, with other oxygen pathway impairments, such as reduced oxygen transfer with placental insufficiency, maternal hyperoxia can be of greater value by increasing maternal uterine artery and vein P
Continue to: Circumstances that may benefit from O2 supplementation...
Circumstances that may benefit from O2 supplementation
Late FHR decelerations reflect impairment of oxygen transfer and thus represent the heart rate pattern that is most likely to benefit from maternal hyperoxia. However, recurrent late decelerations occur in less than 2% of low-risk patients in labor,3 and severe levels of acidosis (umbilical artery pH <7.0 or base deficit [BD] ≥12 mmol/L) occur in only 1% to 2% of near-term or term deliveries.4,5
Variable decelerations also reflect fetal hypoxia and are much more common than late decelerations, so they also may benefit from O2 supplementation. Regardless, O2 supplementation should be seen only as a temporizing strategy while other resuscitative actions are initiated, including preparation for operative delivery, if indicated.
In a prior study by Raghuraman and colleagues (1 of only 4 studies that met selection criteria of oxygen supplementation for patients in labor), newborns of patients not receiving oxygen demonstrated 95% confidence limits of umbilical artery pH (7.24–7.28) and BD (2.9–4.3) well within the normal range.6 Thus, the low prevalence of cases in which a benefit might be anticipated and the low incidence of severe acidosis challenges the design of prospective studies to detect statistically and clinically significant changes in blood gas measures and newborn outcomes.
The normal mild fetal acidosis that develops during labor is likely a result of recurrent interruption of uterine placental blood flow during uterine contractions7 and is unlikely to benefit from maternal hyperoxia. Similarly, as placental oxygen transfer is predominantly flow rather than diffusion limited,8 oxygen supplementation is unlikely to improve severe variable FHR decelerations. Thus, a randomized study of hyperoxia in unselected laboring patients is unlikely to have a measurable effect on clinically significant acidosis.
Oxygen transport pathway guides treatment
For the present, an understanding of oxygen transport can guide clinical oxygen use. Thus, mothers with relative hypoxemia will unquestionably benefit with supplemental oxygen administration. Similarly, fetuses at risk for placental dysfunction (for example, growth restriction, postterm) and particularly those manifesting evidence of impaired oxygen transport (that is, late decelerations) may be most likely to benefit from the increased O2 gradient. For patients with reduced maternal uterine perfusion (such as hypotension or hypovolemia), pressors and/or fluid volume are likely to be more effective, while amnioinfusion is of greater value for umbilical cord compression patterns. A reduction in uterine activity may be of benefit to all fetuses exhibiting compromise. Due to the modest impact on fetal oxygen content, maternal hyperoxia does not produce significant fetal oxidative stress as measured by fetal malondialdehyde levels.
In view of the lack of demonstrated adverse effects of maternal supplemental oxygen, clinicians should not hesitate to use it. However, clinicians should recognize that supplemental oxygen is likely to be of value only in patients with significant impairment in the oxygen pathway, and they should choose additional intrauterine resuscitative measures focused on the etiology.
MICHAEL G. ROSS, MD, MPH,
AND BRYAN S. RICHARDSON, MD
- McNanley T, Woods J. Placental physiology. Glob Libr Women’s Med. (ISSN: 1756-2228). 2008. doi: 10.3843 /GLOWM.10195.
- Richardson BS. Fetal adaptive responses to asphyxia. Clin Perinatol. 1989;16:595-611.
- Sameshima H, Ikenoue T. Predictive value of late decelerations of fetal acidemia in unselective low-risk pregnancies. Am J Perinatol. 2005;22:19-23.
- Yeh P, Emary K, Impey L. The relationship between umbilical cord arterial pH and serious adverse neonatal outcome: analysis of 51,519 consecutive validated samples. BJOG. 2012;119:824-831.
- Kelly R, Ramaiah SM, Sheridan H, et al. Dose-dependent relationship between acidosis at birth and likelihood of death or cerebral palsy. Arch Dis Child Fetal Neonatal Ed. 2018;103:F567-F572.
- Raghuraman N, Wan L, Temming LA, et al. Effect of oxygen vs room air on intrauterine fetal resuscitation: a randomized noninferiority clinical trial. JAMA Pediatr. 2018;172:818-823.
- Ramsey EM, Corner JW Jr, Donner MW. Serial and cineradioangiographic visualization of maternal circulation in the primate (hemochorial) placenta. Am J Obstet Gynecol. 1963;86:213-225.
- Nye GA, Ingram E, Johnstone ED, et al. Human placental oxygenation in late gestation: experimental and theoretical approaches. J Physiol. 2018;596:5523-5534.
- McNanley T, Woods J. Placental physiology. Glob Libr Women’s Med. (ISSN: 1756-2228). 2008. doi: 10.3843 /GLOWM.10195.
- Richardson BS. Fetal adaptive responses to asphyxia. Clin Perinatol. 1989;16:595-611.
- Sameshima H, Ikenoue T. Predictive value of late decelerations of fetal acidemia in unselective low-risk pregnancies. Am J Perinatol. 2005;22:19-23.
- Yeh P, Emary K, Impey L. The relationship between umbilical cord arterial pH and serious adverse neonatal outcome: analysis of 51,519 consecutive validated samples. BJOG. 2012;119:824-831.
- Kelly R, Ramaiah SM, Sheridan H, et al. Dose-dependent relationship between acidosis at birth and likelihood of death or cerebral palsy. Arch Dis Child Fetal Neonatal Ed. 2018;103:F567-F572.
- Raghuraman N, Wan L, Temming LA, et al. Effect of oxygen vs room air on intrauterine fetal resuscitation: a randomized noninferiority clinical trial. JAMA Pediatr. 2018;172:818-823.
- Ramsey EM, Corner JW Jr, Donner MW. Serial and cineradioangiographic visualization of maternal circulation in the primate (hemochorial) placenta. Am J Obstet Gynecol. 1963;86:213-225.
- Nye GA, Ingram E, Johnstone ED, et al. Human placental oxygenation in late gestation: experimental and theoretical approaches. J Physiol. 2018;596:5523-5534.
Office-based ambulatory cervical ripening prior to inpatient induction of labor
For women with a Bishop score ≤6, CR is an important first step in planned induction of labor (IOL). CR is believed to reduce the length of labor induction and increase the probability of a vaginal delivery. Historically, CR has been undertaken on a labor unit. However, with an increased rate of labor induction, the resources of the modern labor unit are incredibly stressed. Compounding the problem is the nursing shortage caused by the COVID-19 pandemic, which has resulted in staff being unavailable as they recover from a respiratory infection or are quarantined after an exposure. The COVID-19 pandemic also has motivated many patients to avoid the hospital as much as possible.
Office-based ambulatory CR is an alternative to inpatient CR and has the potential to reduce the use of labor unit resources. When CR is initiated in the office, the patient either is sent home overnight to return to the labor unit for IOL in the morning or is sent home in the morning to return for IOL in the evening or at night. A secondary benefit of office- and home-based CR is that it may increase patient satisfaction with the process of CR. This editorial summarizes the literature supporting office-based ambulatory CR.
Mechanical methods of CR
Contemporary mechanical methods of CR include the transcervical insertion of a Foley catheter, Cook double-balloon CR catheter, Dilapan-S, or laminaria. There are many publications reporting the feasibility of office-based ambulatory CR with transcervical balloon catheters and very few publications reporting on the use of Dilapan-S or laminaria for ambulatory CR.
Foley catheter
Many studies have investigated the effectiveness of transcervical Foley catheter for ambulatory CR. Policiano and colleagues compared the effectiveness of ambulatory versus inpatient Foley catheter CR.1 A total of 130 women with a Bishop score <6 at ≥41 weeks’ gestation were randomly assigned to outpatient or inpatient CR with a transcervical Foley catheter (Covidian Dover Silicon coated latex Foley catheter 16 Fr/5.3 mm diameter). The Foley catheter bulb was distended with 40 mL of a sterile saline solution. The end of the Foley was taped to the patient’s inner thigh. Manual traction was gently applied to the catheter every 6 hours. If the catheter was extruded, the Bishop score was assessed. For a Bishop score <6, the patient was given additional inpatient misoprostol (25 µg vaginally every 4 hours for up to 5 doses). For a Bishop score ≥6, intravenous oxytocin IOL was initiated. At 24 hours if the Foley catheter was still in situ, it was removed. Women were excluded from the study for the following factors: noncephalic presentation, spontaneous labor, hydramnios, nonreassuring cardiotocography (CTG), multiple pregnancy, ruptured membranes, active vaginal bleeding, Streptococcus group B infection, and HIV infection. Prostaglandin CR was not used if the woman had a previous cesarean delivery. No prophylactic antibiotics were administered. After placement of the Foley catheter, reassuring CTG was documented prior to sending the patient home.
Outpatient, compared with inpatient, CR resulted in a mean reduction of 10 hours in the time from admission to delivery. The time from insertion of the Foley catheter to delivery in the outpatient group was 38.2 hours, and 44.9 hours for the inpatient group (P<.01). The cesarean delivery rates were similar in both groups—28% and 38%, respectively. Three cases of chorioamnionitis occurred in each group. These study results support the feasibility of office-based ambulatory CR with a transcervical Foley.
Ausbeck and colleagues randomly assigned 126 nulliparous women with a Bishop score <5, at a gestational age ranging from 39 weeks and 0 days through 41 weeks and 6 days, to outpatient overnight CR or inpatient CR with a transcervical Foley catheter.2 Breech presentation and multiple gestation pregnancies were excluded from the study. The investigators utilized a 16 French Foley catheter and filled the balloon with 30 mL of sterile water. The Foley was taped to the woman’s inner thigh on slight tension. After placement of the Foley catheter at least 20 minutes of CTG monitoring was performed. The women in the outpatient group were given the contact number for the labor unit and advised that they could take acetaminophen for pain. They were advised that they could stay at home if the Foley catheter was expelled. They were admitted to the labor unit at the time scheduled for their IOL.
The mean time from admission to delivery was reduced by 4.3 hours in the outpatient compared with the inpatient CR group (17.4 vs 21.7 hours; P<.01). In the outpatient CR group, 22% of the women were admitted to labor before the time of the scheduled IOL. The cesarean delivery rates were similar in the outpatient and inpatient CR groups (24% vs 33%, P = .32). In the outpatient and inpatient groups, chorioamnionitis was diagnosed in 22% and 13% (P = .16) of the women. The authors concluded that outpatient CR with a transcervical Foley catheter reduced the time from admission to delivery.
Other research groups also have confirmed the feasibility of outpatient CR with a transcervical Foley catheter.3-5
Placement of the Foley catheter can be performed digitally without direct visualization of the cervix or by direct visualization using a vaginal speculum. After placement of the speculum, the cervix is cleansed with a povidone-iodine solution and a sterile ring forceps is used to grasp the catheter and guide it through the cervical os. In one small study, self-reported pain was similar for both digital and direct visualization methods for placement of the balloon catheter.6 When using Foley catheter CR, filling the standard Foley catheter balloon with 60 mL of fluid, rather than 30 to 40 mL of fluid, is rarely associated with balloon rupture and may result in more effective CR.6,7
Continue to: Double-balloon catheter...
Double-balloon catheter
The Cook double-balloon catheter for CR is meant to create pressure on both sides of the cervix, facilitating CR. Studies have reported that the Cook double-balloon catheter can be used for outpatient CR. In one study, 48 women with a low-risk pregnancy, at 37 to 42 weeks’ gestation and a Bishop score <7 were randomly assigned to outpatient or inpatient double-balloon CR.8 Both balloons were filled with 70 to 80 mL of sterile water. CTG monitoring was performed for 20 minutes before and after balloon placement. The women in the outpatient CR group were instructed to return to the labor unit the next day at 8 AM for IOL or earlier if they had regular uterine contractions, rupture of membranes, or vaginal bleeding. Seven percent of the women in the outpatient group returned to the labor unit before 8 AM. After removal of the balloon catheter, women in the outpatient and inpatient groups needed additional misoprostol CR in 12% and 13% of cases, respectively. Outcomes were similar in the two groups, but the study was not powered to identify small differences between the groups.
In another study of outpatient CR with the Cook double-balloon catheter, 695 women with a Bishop score <7, at ≥37 weeks’ gestation, were randomly assigned to outpatient CR with a double-balloon catheter or inpatient CR with dinoprostone (PGE2) (2 mg dinoprostone vaginal gel [Prostin] or dinoprostone 10 mg controlled-release tape (Cervidil).9 Women assigned to dinoprostone CR had CTG monitoring prior to commencing PGE2 CR and at least 30 min of CTG monitoring after insertion of the vaginal PGE2. Women assigned to balloon CR were not admitted to the hospital. CTG was performed prior to insertion of the balloon. After insertion, the two balloons on the catheter were each filled with 80 mL of saline. After catheter insertion CTG monitoring was not routinely performed. The women in the double-balloon catheter group returned to the labor unit 12 hours after insertion to initiate IOL. The primary outcome was composite neonatal morbidity and mortality, including admission to a neonatal intensive care unit (NICU), intubation, cardiac compressions, acidemia, hypoxic ischemic encephalopathy, seizure, infection, pulmonary hypertension, stillbirth, or death.
There was no significant difference in the rate of the primary outcome in the catheter versus the PGE2 group (18.6% and 25.8%; P = .07). Admission to the NICU occurred at rates of 12.6% and 15.5% in the catheter and PGE2 groups. Umbilical cord arterial pH <7.00 at birth occurred at a rate of 3.5% in the catheter group and 9.2% in the PGE2 group. The cesarean delivery rates in the catheter and PGE groups were 32.6% and 25.8%, respectively (P = .24). The investigators concluded that outpatient CR using a double-balloon catheter is safe and feasible for nulliparous women.
Two systematic reviews and meta-analyses reported that outcomes were similar when using the Foley or double-balloon catheter for CR.10,11 The Cook double-balloon CR kit includes a stylet, which can facilitate passing the catheter through the cervix.
Continue to: Dilapan-S and laminaria...
Dilapan-S and laminaria
There are many published studies using Dilapan-S and laminaria for cervical preparation prior to uterine evacuation.12 There are few published studies using Dilapan-S or laminaria for CR prior to IOL. In a pilot study, 21 patients were randomly assigned to outpatient versus inpatient Dilapan-S for CR the night prior to scheduled oxytocin IOL.13 The length of time from initiation of oxytocin to delivery in the outpatient and inpatient groups was similar (11 vs 14 hours, respectively). The outpatient compared with the inpatient group had a shorter length of hospitalization until delivery (51 vs 70 hours).
In other studies of Dilapan-S for CR, the patients remained in the hospital once the dilators were inserted. In one small trial, 41 women were randomized to CR with Dilapan-S or laminaria. As many dilators as could be comfortably tolerated by the patient were inserted.14 The mean numbers of Dilapan-S and laminaria dilators inserted were 4.3 and 9.7, respectively. The morning after the insertion of the dilators, oxytocin IOL was initiated. The times from initiation of oxytocin to delivery for the women in the Dilapan-S and laminaria groups were 11.6 and 15.5 hours, respectively.
An observational study reported on outcomes with Dilapan-S for CR on inpatients.15 In the study 444 women scheduled for IOL at 37 to 40 weeks’ gestation, with a mean baseline Bishop score of 2.9, had Dilapan-S placed for approximately 15 hours prior to oxytocin IOL. The mean number of Dilapan-S dilators that were inserted was 3.8. The study protocol prohibited placing more than 5 cervical dilator devices. The mean Bishop score after removal of the dilators was 6.5. The most common adverse effects of Dilapan-S CR were bleeding (2.7%) and pain (0.2%). The cesarean delivery rate in the cohort was 30.1%. An Apgar score <7 at 5 minutes was recorded for 3 newborns. An umbilical artery pH of <7.10 was observed in 8 newborns.
In a randomized trial performed on inpatients, 419 women undergoing CR were assigned to a Foley balloon or Dilapan-S.16 The vaginal delivery rates were similar in the groups—76% for Foley and 81% for Dilapan-S. Maternal and neonatal adverse effects were similar between the two groups. Compared with Foley catheter, women assigned to Dilapan-S reported greater satisfaction with their CR experience, more sleep, and more ability to perform daily activities.
Misoprostol and dinoprostone
Both misoprostol and dinoprostone are effective for outpatient CR. However, a Cochrane systematic review and meta-analysis concluded that balloon CR, compared with prostaglandin CR, is probably associated with a lower risk of uterine hyperstimulation with concerning fetal heart rate changes.17 Because misoprostol and dinoprostone occasionally can cause uterine hyperstimulation with fetal heart changes, many experts recommend CTG monitoring both before and after administration of misoprostol or dinoprostone for CR.
In a trial of outpatient versus inpatient vaginal PGE2 CR, 425 women at 37 to 42 weeks’ gestation were assigned randomly to outpatient or inpatient CR.18 All women had CTG monitoring for 20 minutes before and after vaginal placement of the PGE2 gel. The PGE2 dose was 2 mg for nulliparous and 1 mg for parous women. The cesarean delivery rates were similar in the outpatient and inpatient groups—22.3% and 22.9%, respectively. Among the women randomized to outpatient CR, 27 women (13%) could not be discharged home after administration of the vaginal PGE2 because of frequent uterine contractions or an abnormal fetal heart rate pattern. In addition, 64 women (30%) in the outpatient group returned to the hospital before scheduled induction because of frequent contractions. Maternal and neonatal complications were similar in the two groups. The investigators concluded that, at the dose and route of prostaglandin utilized in this study, the resultant rates of abnormal fetal heart rate pattern and frequent contractions might reduce the clinical utility of outpatient vaginal prostaglandin CR.
Another study also reported a greater rate of uterine tachysystole with vaginal PGE2 compared with a Foley catheter for CR (9% vs 0%).19 In a Cochrane systematic review of vaginal prostaglandin for CR, compared with placebo, vaginal prostaglandins were associated with a significantly greater rate of uterine hyperstimulation with fetal heart rate changes (4.8% vs 1.0%).20 Other studies also reported the feasibility of outpatient CR with vaginal prostaglandin.21,22
Both oral and vaginal misoprostol have been utilized for outpatient CR. In one study, 87 women with singleton pregnancy at 40 to 42 weeks’ gestation with a Bishop score <6 were randomized to outpatient CR with oral misoprostol (100 µg) or placebo.23 Following administration of the oral misoprostol, the women had 2 hours of CTG monitoring. The treatment was repeated daily for up to 3 days if there was no change in the cervix. If labor occurred, the patient was admitted to the labor unit for oxytocin IOL. The times from first dose of misoprostol or placebo to delivery were 46 and 84 hours (P<.001), respectively.
In another study, 49 women ≥40 weeks’ gestation with a Bishop score <5 were randomly assigned to receive outpatient oral misoprostol 25 µg or 50 µg.24 The dose could be repeated every 3 days over 9 days if ripening or labor had not been achieved. The women had CTG before administration of oral misoprostol. After the misoprostol dose, they had 2 hours of CTG monitoring. The number of doses received by the women assigned to the 50 µg group were 83%, 13%, and 4% for 1, 2, and 3 doses, respectively. The number of doses received by the women assigned to the 25 µg group were 58%, 26%, and 16% for 1, 2, and 3 doses, respectively. The mean intervals from initiation of CR to delivery in the 25 µg and the 50 µg groups were 3.9 and 2.5 days, respectively. The investigators reported no maternal or newborn adverse events, although the study was not powered to detect infrequent events.
Many studies have reported on the feasibility of outpatient CR with vaginal misoprostol.25-30 In one study, 77 women at 40 weeks’ gestation and a Bishop score ≤8 were randomized to a single dose of vaginal misoprostol 25 µg or gentle cervical examination (control).25 The women had 1 hour of CTG monitoring after the intervention. If they had regular contractions they were admitted to the birthing unit. If they had no regular contractions they were discharged home. For nulliparous women, the time from intervention to delivery in the misoprostol group was 4.9 days, and 8.1 days in the control group. For parous women, the times from intervention to delivery in the two groups were 3.8 and 6.9 days, respectively.
Continue to: Inclusion and exclusion criteria for outpatient CR...
Inclusion and exclusion criteria for outpatient CR
Outpatient CR should be limited to low-risk women with a singleton gestation, who have reliable access to transportation from home to the labor unit and have a clear understanding of the instructions for outpatient CR. Patient characteristics that may be utilized to offer office-based CR include:
- singleton pregnancy at 39 weeks’ and 0 days’ gestation through 40 weeks’ and 6 days’ gestation
- cephalic presentation
- Bishop score ≤6.
Women who should be excluded from outpatient CR include those with:
- contraindications to vaginal delivery
- fetal growth restriction
- abnormal umbilical artery Doppler results
- oligo- or polyhydramnios
- multiple gestation
- major fetal anomaly
- recent nonreactive fetal heart rate tracing
- maternal report of decreased fetal movement
- abnormal biophysical profile
- prior cesarean delivery
- recent vaginal bleeding
- gestational diabetes requiring medication treatment
- significant hypertension.
Practices should establish their own inclusion and exclusion criteria for ambulatory CR.
Safety of office-based ambulatory CR among low-risk women
Safety is a complex concept with experts often disagreeing on what level of safety is required to accept a new medical procedure. Establishing the safety of office-based ambulatory CR among low-risk women would require a very large cohort or randomized studies with at least a thousand participants. Only a few large studies focused on the safety of CR have been reported. Sciscione and colleagues reported a large observational study of inpatient transcervical Foley catheter for CR involving 1,905 women.31 They reported no adverse outcomes among term, singleton, uncomplicated pregnancies. They calculated that the 95% confidence interval (CI) for an adverse event was between 0.0% and 0.2%. In a meta-analysis of 26 studies including 5,563 women, the risk of chorioamnionitis during IOL was equivalent with pre-IOL Foley catheter CR (7.2%) or prostaglandin CR (7.2%) (relative risk, 0.96; 95% CI, 0.66–1.38).32
Two systematic reviews have reported that, compared with balloon CR, misoprostol CR is associated with an increased risk of uterine tachysystole.33-34 In a large retrospective study, compared with inpatient CR, outpatient CR with dinoprostone vaginal insert was not associated with an increased risk of newborn admission to the neonatal intensive care unit or a low Apgar score at 5 minutes after birth.35
Will you consider office-based CR in your obstetric practice?
As reviewed in this editorial, evolving data suggest that it is feasible to initiate CR in the office ambulatory setting prior to admission to the labor unit for additional CR or IOL. Many women prefer to complete CR at home after initiation in the office, rather than have CR in a labor unit or hospital setting.36 The transcervical balloon catheter has the most published data supporting the feasibility of ambulatory CR. Compared with misoprostol, the transcervical balloon catheter is associated with a low rate of uterine tachysystole. It may be a preferred method for outpatient CR. If placement of a transcervical balloon catheter is challenging, for example when the patient has a tightly closed cervix, oral misoprostol ambulatory CR may be an option if CTG monitoring is available in the office.
During the COVID pandemic, many in-person office visits have transitioned to virtual visits with the patient in their home. Historically, most cases of CR have been performed on labor and delivery units. It may be time for your practice to consider office-based ambulatory CR for low-risk women planning an IOL. Office-based ambulatory CR is a win for labor nurses who generally prefer to manage laboring patients rather than patients undergoing prolonged in-hospital CR. Outpatient CR is also a win for low-risk patients who prefer to be at home rather than in a labor unit. ●
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Repro Biol. 2017;210:1-6.
- Ausbeck EB, Jauk VC, Xue Y, et al. Outpatient Foley catheter for induction of labor in nulliparous women. Obstet Gynecol. 2020;136:597-606.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98:751-756.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Kuhlmann MJ, Spencer N, Garcia-Jasso C, et al. Foley bulb insertion by blind placement compared with direct visualization. Obstet Gynecol. 2021;137:139-145.
- Delaney S, Shaffer BL, Chen YW, et al. Labor induction with a Foley balloon inflated to 30 mL compared with 60 mL. Obstet Gynecol. 2015;115:1239-1245.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Beckmann M, Gibbons K, Flenady V, et al. Induction of labor using prostaglandin E2 as an inpatient versus balloon catheter as an outpatient: a multicenter randomised controlled trial. BJOG. 2020;127:571-579.
- Liu X, Wang Y, Zhange F, et al. Double- versus single-balloon catheters for labour induction and cervical ripening: a meta-analysis. BMC Pregnancy Childbirth. 2019;19:358.
- Yang F, Huan S, Long Y, et al. Double-balloon versus single-balloon catheter for cervical ripening and labor induction: a systematic review and meta-analysis. J Obstet Gynaecol Res. 2018;44: 27-34.
- Goldberg AB, Fortin JA, Drey EA, et al. Cervical preparation before dilation and evacuation using adjunctive misoprostol and mifepristone compared with overnight osmotic dilators alone: a randomized controlled trial. Obstet Gynecol. 2015;126:599-609.
- Upadhyaya NB, Childs KD, Neiger R, et al. Ambulatory cervical ripening in term pregnancy. J Reprod Med. 1999;44:363-366.
- Blumenthal PD, Rmanauskas R. Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor. Obstet Gynecol. 1990;75:365-368.
- Gupta J, Chodankar R, Baev O, et al. Synthetic osmotic dilators in the induction of labour—an international multicenter observational study. Eur J Obstet Gynecol Repro Biol. 2018;229:70-75.
- Saad AF, Villarreal J, Eid J, et al. A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial). Am J Obstet Gynecol. 2019;220:275.e1-e9.
- de Vaan MD, Eikleder MLT, Jozwiak M, et al. Mechanical methods for induction of labour. Cochrane Database Syst Rev. 2019;CD001233.
- Wilkinson C, Bryce R, Adelson P, et al. A randomized controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E2 (OPRA study). BJOG. 2015;122:94-104.
- Blair R, Harvey MA, Pudwell J, et al. Retrospective comparison of PGE2 vaginal insert and Foley catheter for outpatient cervical ripening. J Obstet Gynaecol Can. 2020;42:1103-1110.
- Thomas J, Fairclough A, Kavanagh J, et al. Vaginal prostaglandin (PGE2 or PGF2alpha) for induction of labour at term. Cochrane Database Syst Rev. 2014;CD003101.
- O’Brien JM, Mercer BM, Cleary NT, et al. Efficacy of outpatient induction with low-dose intravaginal prostaglandin E2: a randomized, doubleblind, placebo controlled trial. Am J Obstet Gynecol. 1995;173:1855-1859.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26: 673-677.
- Kipikasa JH, Adair CD, Williamson J, et al. Use of misoprostol on an outpatient basis for postdate pregnancy. Int J Gynaecol Obstet. 2005;88:108-111.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening without subsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96:684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labor. Arch Gynecol Obstet. 2017;295:33-38.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs inpatient administration. Obstet Gynecol Surv. 2006;61:167-168.
- Meyer M, Pflum J, Howard D. Outpatient misoprostol compared with dinoprostone gel for preinduction cervical ripening: a randomized controlled trial. Obstet Gynecol. 2005;105:466-472.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- McMaster K, Sanchez-Ramos L, Kaunitz AM. Evaluation of a transcervical Foley catheter as a source of infection. Obstet Gynecol. 2015;126:539-551.
- Fox NS, Saltzman DH, Roman AS, et al. Intravaginal misoprostol versus Foley catheter for labour induction: a meta-analysis. BJOG. 2011;118: 647-654.
- Hofmeyr GJ, Gulmezoglu AM, Pileggi C. Vaginal misoprostol for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2010:CD000941.
- Salvador SC, Simpson ML, Cundiff GW. Dinoprostone vaginal insert for labour induction: a comparison of outpatient and inpatient settings. J Obstet Gynaecol Can. 2009;31:1028-1034.
- Sutton C, Harding J, Griffin C. Patient attitudes towards outpatient cervical ripening prior to induction of labour at an Australian tertiary hospital. J Obstet Gynaecol. 2016;36:921-928.
For women with a Bishop score ≤6, CR is an important first step in planned induction of labor (IOL). CR is believed to reduce the length of labor induction and increase the probability of a vaginal delivery. Historically, CR has been undertaken on a labor unit. However, with an increased rate of labor induction, the resources of the modern labor unit are incredibly stressed. Compounding the problem is the nursing shortage caused by the COVID-19 pandemic, which has resulted in staff being unavailable as they recover from a respiratory infection or are quarantined after an exposure. The COVID-19 pandemic also has motivated many patients to avoid the hospital as much as possible.
Office-based ambulatory CR is an alternative to inpatient CR and has the potential to reduce the use of labor unit resources. When CR is initiated in the office, the patient either is sent home overnight to return to the labor unit for IOL in the morning or is sent home in the morning to return for IOL in the evening or at night. A secondary benefit of office- and home-based CR is that it may increase patient satisfaction with the process of CR. This editorial summarizes the literature supporting office-based ambulatory CR.
Mechanical methods of CR
Contemporary mechanical methods of CR include the transcervical insertion of a Foley catheter, Cook double-balloon CR catheter, Dilapan-S, or laminaria. There are many publications reporting the feasibility of office-based ambulatory CR with transcervical balloon catheters and very few publications reporting on the use of Dilapan-S or laminaria for ambulatory CR.
Foley catheter
Many studies have investigated the effectiveness of transcervical Foley catheter for ambulatory CR. Policiano and colleagues compared the effectiveness of ambulatory versus inpatient Foley catheter CR.1 A total of 130 women with a Bishop score <6 at ≥41 weeks’ gestation were randomly assigned to outpatient or inpatient CR with a transcervical Foley catheter (Covidian Dover Silicon coated latex Foley catheter 16 Fr/5.3 mm diameter). The Foley catheter bulb was distended with 40 mL of a sterile saline solution. The end of the Foley was taped to the patient’s inner thigh. Manual traction was gently applied to the catheter every 6 hours. If the catheter was extruded, the Bishop score was assessed. For a Bishop score <6, the patient was given additional inpatient misoprostol (25 µg vaginally every 4 hours for up to 5 doses). For a Bishop score ≥6, intravenous oxytocin IOL was initiated. At 24 hours if the Foley catheter was still in situ, it was removed. Women were excluded from the study for the following factors: noncephalic presentation, spontaneous labor, hydramnios, nonreassuring cardiotocography (CTG), multiple pregnancy, ruptured membranes, active vaginal bleeding, Streptococcus group B infection, and HIV infection. Prostaglandin CR was not used if the woman had a previous cesarean delivery. No prophylactic antibiotics were administered. After placement of the Foley catheter, reassuring CTG was documented prior to sending the patient home.
Outpatient, compared with inpatient, CR resulted in a mean reduction of 10 hours in the time from admission to delivery. The time from insertion of the Foley catheter to delivery in the outpatient group was 38.2 hours, and 44.9 hours for the inpatient group (P<.01). The cesarean delivery rates were similar in both groups—28% and 38%, respectively. Three cases of chorioamnionitis occurred in each group. These study results support the feasibility of office-based ambulatory CR with a transcervical Foley.
Ausbeck and colleagues randomly assigned 126 nulliparous women with a Bishop score <5, at a gestational age ranging from 39 weeks and 0 days through 41 weeks and 6 days, to outpatient overnight CR or inpatient CR with a transcervical Foley catheter.2 Breech presentation and multiple gestation pregnancies were excluded from the study. The investigators utilized a 16 French Foley catheter and filled the balloon with 30 mL of sterile water. The Foley was taped to the woman’s inner thigh on slight tension. After placement of the Foley catheter at least 20 minutes of CTG monitoring was performed. The women in the outpatient group were given the contact number for the labor unit and advised that they could take acetaminophen for pain. They were advised that they could stay at home if the Foley catheter was expelled. They were admitted to the labor unit at the time scheduled for their IOL.
The mean time from admission to delivery was reduced by 4.3 hours in the outpatient compared with the inpatient CR group (17.4 vs 21.7 hours; P<.01). In the outpatient CR group, 22% of the women were admitted to labor before the time of the scheduled IOL. The cesarean delivery rates were similar in the outpatient and inpatient CR groups (24% vs 33%, P = .32). In the outpatient and inpatient groups, chorioamnionitis was diagnosed in 22% and 13% (P = .16) of the women. The authors concluded that outpatient CR with a transcervical Foley catheter reduced the time from admission to delivery.
Other research groups also have confirmed the feasibility of outpatient CR with a transcervical Foley catheter.3-5
Placement of the Foley catheter can be performed digitally without direct visualization of the cervix or by direct visualization using a vaginal speculum. After placement of the speculum, the cervix is cleansed with a povidone-iodine solution and a sterile ring forceps is used to grasp the catheter and guide it through the cervical os. In one small study, self-reported pain was similar for both digital and direct visualization methods for placement of the balloon catheter.6 When using Foley catheter CR, filling the standard Foley catheter balloon with 60 mL of fluid, rather than 30 to 40 mL of fluid, is rarely associated with balloon rupture and may result in more effective CR.6,7
Continue to: Double-balloon catheter...
Double-balloon catheter
The Cook double-balloon catheter for CR is meant to create pressure on both sides of the cervix, facilitating CR. Studies have reported that the Cook double-balloon catheter can be used for outpatient CR. In one study, 48 women with a low-risk pregnancy, at 37 to 42 weeks’ gestation and a Bishop score <7 were randomly assigned to outpatient or inpatient double-balloon CR.8 Both balloons were filled with 70 to 80 mL of sterile water. CTG monitoring was performed for 20 minutes before and after balloon placement. The women in the outpatient CR group were instructed to return to the labor unit the next day at 8 AM for IOL or earlier if they had regular uterine contractions, rupture of membranes, or vaginal bleeding. Seven percent of the women in the outpatient group returned to the labor unit before 8 AM. After removal of the balloon catheter, women in the outpatient and inpatient groups needed additional misoprostol CR in 12% and 13% of cases, respectively. Outcomes were similar in the two groups, but the study was not powered to identify small differences between the groups.
In another study of outpatient CR with the Cook double-balloon catheter, 695 women with a Bishop score <7, at ≥37 weeks’ gestation, were randomly assigned to outpatient CR with a double-balloon catheter or inpatient CR with dinoprostone (PGE2) (2 mg dinoprostone vaginal gel [Prostin] or dinoprostone 10 mg controlled-release tape (Cervidil).9 Women assigned to dinoprostone CR had CTG monitoring prior to commencing PGE2 CR and at least 30 min of CTG monitoring after insertion of the vaginal PGE2. Women assigned to balloon CR were not admitted to the hospital. CTG was performed prior to insertion of the balloon. After insertion, the two balloons on the catheter were each filled with 80 mL of saline. After catheter insertion CTG monitoring was not routinely performed. The women in the double-balloon catheter group returned to the labor unit 12 hours after insertion to initiate IOL. The primary outcome was composite neonatal morbidity and mortality, including admission to a neonatal intensive care unit (NICU), intubation, cardiac compressions, acidemia, hypoxic ischemic encephalopathy, seizure, infection, pulmonary hypertension, stillbirth, or death.
There was no significant difference in the rate of the primary outcome in the catheter versus the PGE2 group (18.6% and 25.8%; P = .07). Admission to the NICU occurred at rates of 12.6% and 15.5% in the catheter and PGE2 groups. Umbilical cord arterial pH <7.00 at birth occurred at a rate of 3.5% in the catheter group and 9.2% in the PGE2 group. The cesarean delivery rates in the catheter and PGE groups were 32.6% and 25.8%, respectively (P = .24). The investigators concluded that outpatient CR using a double-balloon catheter is safe and feasible for nulliparous women.
Two systematic reviews and meta-analyses reported that outcomes were similar when using the Foley or double-balloon catheter for CR.10,11 The Cook double-balloon CR kit includes a stylet, which can facilitate passing the catheter through the cervix.
Continue to: Dilapan-S and laminaria...
Dilapan-S and laminaria
There are many published studies using Dilapan-S and laminaria for cervical preparation prior to uterine evacuation.12 There are few published studies using Dilapan-S or laminaria for CR prior to IOL. In a pilot study, 21 patients were randomly assigned to outpatient versus inpatient Dilapan-S for CR the night prior to scheduled oxytocin IOL.13 The length of time from initiation of oxytocin to delivery in the outpatient and inpatient groups was similar (11 vs 14 hours, respectively). The outpatient compared with the inpatient group had a shorter length of hospitalization until delivery (51 vs 70 hours).
In other studies of Dilapan-S for CR, the patients remained in the hospital once the dilators were inserted. In one small trial, 41 women were randomized to CR with Dilapan-S or laminaria. As many dilators as could be comfortably tolerated by the patient were inserted.14 The mean numbers of Dilapan-S and laminaria dilators inserted were 4.3 and 9.7, respectively. The morning after the insertion of the dilators, oxytocin IOL was initiated. The times from initiation of oxytocin to delivery for the women in the Dilapan-S and laminaria groups were 11.6 and 15.5 hours, respectively.
An observational study reported on outcomes with Dilapan-S for CR on inpatients.15 In the study 444 women scheduled for IOL at 37 to 40 weeks’ gestation, with a mean baseline Bishop score of 2.9, had Dilapan-S placed for approximately 15 hours prior to oxytocin IOL. The mean number of Dilapan-S dilators that were inserted was 3.8. The study protocol prohibited placing more than 5 cervical dilator devices. The mean Bishop score after removal of the dilators was 6.5. The most common adverse effects of Dilapan-S CR were bleeding (2.7%) and pain (0.2%). The cesarean delivery rate in the cohort was 30.1%. An Apgar score <7 at 5 minutes was recorded for 3 newborns. An umbilical artery pH of <7.10 was observed in 8 newborns.
In a randomized trial performed on inpatients, 419 women undergoing CR were assigned to a Foley balloon or Dilapan-S.16 The vaginal delivery rates were similar in the groups—76% for Foley and 81% for Dilapan-S. Maternal and neonatal adverse effects were similar between the two groups. Compared with Foley catheter, women assigned to Dilapan-S reported greater satisfaction with their CR experience, more sleep, and more ability to perform daily activities.
Misoprostol and dinoprostone
Both misoprostol and dinoprostone are effective for outpatient CR. However, a Cochrane systematic review and meta-analysis concluded that balloon CR, compared with prostaglandin CR, is probably associated with a lower risk of uterine hyperstimulation with concerning fetal heart rate changes.17 Because misoprostol and dinoprostone occasionally can cause uterine hyperstimulation with fetal heart changes, many experts recommend CTG monitoring both before and after administration of misoprostol or dinoprostone for CR.
In a trial of outpatient versus inpatient vaginal PGE2 CR, 425 women at 37 to 42 weeks’ gestation were assigned randomly to outpatient or inpatient CR.18 All women had CTG monitoring for 20 minutes before and after vaginal placement of the PGE2 gel. The PGE2 dose was 2 mg for nulliparous and 1 mg for parous women. The cesarean delivery rates were similar in the outpatient and inpatient groups—22.3% and 22.9%, respectively. Among the women randomized to outpatient CR, 27 women (13%) could not be discharged home after administration of the vaginal PGE2 because of frequent uterine contractions or an abnormal fetal heart rate pattern. In addition, 64 women (30%) in the outpatient group returned to the hospital before scheduled induction because of frequent contractions. Maternal and neonatal complications were similar in the two groups. The investigators concluded that, at the dose and route of prostaglandin utilized in this study, the resultant rates of abnormal fetal heart rate pattern and frequent contractions might reduce the clinical utility of outpatient vaginal prostaglandin CR.
Another study also reported a greater rate of uterine tachysystole with vaginal PGE2 compared with a Foley catheter for CR (9% vs 0%).19 In a Cochrane systematic review of vaginal prostaglandin for CR, compared with placebo, vaginal prostaglandins were associated with a significantly greater rate of uterine hyperstimulation with fetal heart rate changes (4.8% vs 1.0%).20 Other studies also reported the feasibility of outpatient CR with vaginal prostaglandin.21,22
Both oral and vaginal misoprostol have been utilized for outpatient CR. In one study, 87 women with singleton pregnancy at 40 to 42 weeks’ gestation with a Bishop score <6 were randomized to outpatient CR with oral misoprostol (100 µg) or placebo.23 Following administration of the oral misoprostol, the women had 2 hours of CTG monitoring. The treatment was repeated daily for up to 3 days if there was no change in the cervix. If labor occurred, the patient was admitted to the labor unit for oxytocin IOL. The times from first dose of misoprostol or placebo to delivery were 46 and 84 hours (P<.001), respectively.
In another study, 49 women ≥40 weeks’ gestation with a Bishop score <5 were randomly assigned to receive outpatient oral misoprostol 25 µg or 50 µg.24 The dose could be repeated every 3 days over 9 days if ripening or labor had not been achieved. The women had CTG before administration of oral misoprostol. After the misoprostol dose, they had 2 hours of CTG monitoring. The number of doses received by the women assigned to the 50 µg group were 83%, 13%, and 4% for 1, 2, and 3 doses, respectively. The number of doses received by the women assigned to the 25 µg group were 58%, 26%, and 16% for 1, 2, and 3 doses, respectively. The mean intervals from initiation of CR to delivery in the 25 µg and the 50 µg groups were 3.9 and 2.5 days, respectively. The investigators reported no maternal or newborn adverse events, although the study was not powered to detect infrequent events.
Many studies have reported on the feasibility of outpatient CR with vaginal misoprostol.25-30 In one study, 77 women at 40 weeks’ gestation and a Bishop score ≤8 were randomized to a single dose of vaginal misoprostol 25 µg or gentle cervical examination (control).25 The women had 1 hour of CTG monitoring after the intervention. If they had regular contractions they were admitted to the birthing unit. If they had no regular contractions they were discharged home. For nulliparous women, the time from intervention to delivery in the misoprostol group was 4.9 days, and 8.1 days in the control group. For parous women, the times from intervention to delivery in the two groups were 3.8 and 6.9 days, respectively.
Continue to: Inclusion and exclusion criteria for outpatient CR...
Inclusion and exclusion criteria for outpatient CR
Outpatient CR should be limited to low-risk women with a singleton gestation, who have reliable access to transportation from home to the labor unit and have a clear understanding of the instructions for outpatient CR. Patient characteristics that may be utilized to offer office-based CR include:
- singleton pregnancy at 39 weeks’ and 0 days’ gestation through 40 weeks’ and 6 days’ gestation
- cephalic presentation
- Bishop score ≤6.
Women who should be excluded from outpatient CR include those with:
- contraindications to vaginal delivery
- fetal growth restriction
- abnormal umbilical artery Doppler results
- oligo- or polyhydramnios
- multiple gestation
- major fetal anomaly
- recent nonreactive fetal heart rate tracing
- maternal report of decreased fetal movement
- abnormal biophysical profile
- prior cesarean delivery
- recent vaginal bleeding
- gestational diabetes requiring medication treatment
- significant hypertension.
Practices should establish their own inclusion and exclusion criteria for ambulatory CR.
Safety of office-based ambulatory CR among low-risk women
Safety is a complex concept with experts often disagreeing on what level of safety is required to accept a new medical procedure. Establishing the safety of office-based ambulatory CR among low-risk women would require a very large cohort or randomized studies with at least a thousand participants. Only a few large studies focused on the safety of CR have been reported. Sciscione and colleagues reported a large observational study of inpatient transcervical Foley catheter for CR involving 1,905 women.31 They reported no adverse outcomes among term, singleton, uncomplicated pregnancies. They calculated that the 95% confidence interval (CI) for an adverse event was between 0.0% and 0.2%. In a meta-analysis of 26 studies including 5,563 women, the risk of chorioamnionitis during IOL was equivalent with pre-IOL Foley catheter CR (7.2%) or prostaglandin CR (7.2%) (relative risk, 0.96; 95% CI, 0.66–1.38).32
Two systematic reviews have reported that, compared with balloon CR, misoprostol CR is associated with an increased risk of uterine tachysystole.33-34 In a large retrospective study, compared with inpatient CR, outpatient CR with dinoprostone vaginal insert was not associated with an increased risk of newborn admission to the neonatal intensive care unit or a low Apgar score at 5 minutes after birth.35
Will you consider office-based CR in your obstetric practice?
As reviewed in this editorial, evolving data suggest that it is feasible to initiate CR in the office ambulatory setting prior to admission to the labor unit for additional CR or IOL. Many women prefer to complete CR at home after initiation in the office, rather than have CR in a labor unit or hospital setting.36 The transcervical balloon catheter has the most published data supporting the feasibility of ambulatory CR. Compared with misoprostol, the transcervical balloon catheter is associated with a low rate of uterine tachysystole. It may be a preferred method for outpatient CR. If placement of a transcervical balloon catheter is challenging, for example when the patient has a tightly closed cervix, oral misoprostol ambulatory CR may be an option if CTG monitoring is available in the office.
During the COVID pandemic, many in-person office visits have transitioned to virtual visits with the patient in their home. Historically, most cases of CR have been performed on labor and delivery units. It may be time for your practice to consider office-based ambulatory CR for low-risk women planning an IOL. Office-based ambulatory CR is a win for labor nurses who generally prefer to manage laboring patients rather than patients undergoing prolonged in-hospital CR. Outpatient CR is also a win for low-risk patients who prefer to be at home rather than in a labor unit. ●
For women with a Bishop score ≤6, CR is an important first step in planned induction of labor (IOL). CR is believed to reduce the length of labor induction and increase the probability of a vaginal delivery. Historically, CR has been undertaken on a labor unit. However, with an increased rate of labor induction, the resources of the modern labor unit are incredibly stressed. Compounding the problem is the nursing shortage caused by the COVID-19 pandemic, which has resulted in staff being unavailable as they recover from a respiratory infection or are quarantined after an exposure. The COVID-19 pandemic also has motivated many patients to avoid the hospital as much as possible.
Office-based ambulatory CR is an alternative to inpatient CR and has the potential to reduce the use of labor unit resources. When CR is initiated in the office, the patient either is sent home overnight to return to the labor unit for IOL in the morning or is sent home in the morning to return for IOL in the evening or at night. A secondary benefit of office- and home-based CR is that it may increase patient satisfaction with the process of CR. This editorial summarizes the literature supporting office-based ambulatory CR.
Mechanical methods of CR
Contemporary mechanical methods of CR include the transcervical insertion of a Foley catheter, Cook double-balloon CR catheter, Dilapan-S, or laminaria. There are many publications reporting the feasibility of office-based ambulatory CR with transcervical balloon catheters and very few publications reporting on the use of Dilapan-S or laminaria for ambulatory CR.
Foley catheter
Many studies have investigated the effectiveness of transcervical Foley catheter for ambulatory CR. Policiano and colleagues compared the effectiveness of ambulatory versus inpatient Foley catheter CR.1 A total of 130 women with a Bishop score <6 at ≥41 weeks’ gestation were randomly assigned to outpatient or inpatient CR with a transcervical Foley catheter (Covidian Dover Silicon coated latex Foley catheter 16 Fr/5.3 mm diameter). The Foley catheter bulb was distended with 40 mL of a sterile saline solution. The end of the Foley was taped to the patient’s inner thigh. Manual traction was gently applied to the catheter every 6 hours. If the catheter was extruded, the Bishop score was assessed. For a Bishop score <6, the patient was given additional inpatient misoprostol (25 µg vaginally every 4 hours for up to 5 doses). For a Bishop score ≥6, intravenous oxytocin IOL was initiated. At 24 hours if the Foley catheter was still in situ, it was removed. Women were excluded from the study for the following factors: noncephalic presentation, spontaneous labor, hydramnios, nonreassuring cardiotocography (CTG), multiple pregnancy, ruptured membranes, active vaginal bleeding, Streptococcus group B infection, and HIV infection. Prostaglandin CR was not used if the woman had a previous cesarean delivery. No prophylactic antibiotics were administered. After placement of the Foley catheter, reassuring CTG was documented prior to sending the patient home.
Outpatient, compared with inpatient, CR resulted in a mean reduction of 10 hours in the time from admission to delivery. The time from insertion of the Foley catheter to delivery in the outpatient group was 38.2 hours, and 44.9 hours for the inpatient group (P<.01). The cesarean delivery rates were similar in both groups—28% and 38%, respectively. Three cases of chorioamnionitis occurred in each group. These study results support the feasibility of office-based ambulatory CR with a transcervical Foley.
Ausbeck and colleagues randomly assigned 126 nulliparous women with a Bishop score <5, at a gestational age ranging from 39 weeks and 0 days through 41 weeks and 6 days, to outpatient overnight CR or inpatient CR with a transcervical Foley catheter.2 Breech presentation and multiple gestation pregnancies were excluded from the study. The investigators utilized a 16 French Foley catheter and filled the balloon with 30 mL of sterile water. The Foley was taped to the woman’s inner thigh on slight tension. After placement of the Foley catheter at least 20 minutes of CTG monitoring was performed. The women in the outpatient group were given the contact number for the labor unit and advised that they could take acetaminophen for pain. They were advised that they could stay at home if the Foley catheter was expelled. They were admitted to the labor unit at the time scheduled for their IOL.
The mean time from admission to delivery was reduced by 4.3 hours in the outpatient compared with the inpatient CR group (17.4 vs 21.7 hours; P<.01). In the outpatient CR group, 22% of the women were admitted to labor before the time of the scheduled IOL. The cesarean delivery rates were similar in the outpatient and inpatient CR groups (24% vs 33%, P = .32). In the outpatient and inpatient groups, chorioamnionitis was diagnosed in 22% and 13% (P = .16) of the women. The authors concluded that outpatient CR with a transcervical Foley catheter reduced the time from admission to delivery.
Other research groups also have confirmed the feasibility of outpatient CR with a transcervical Foley catheter.3-5
Placement of the Foley catheter can be performed digitally without direct visualization of the cervix or by direct visualization using a vaginal speculum. After placement of the speculum, the cervix is cleansed with a povidone-iodine solution and a sterile ring forceps is used to grasp the catheter and guide it through the cervical os. In one small study, self-reported pain was similar for both digital and direct visualization methods for placement of the balloon catheter.6 When using Foley catheter CR, filling the standard Foley catheter balloon with 60 mL of fluid, rather than 30 to 40 mL of fluid, is rarely associated with balloon rupture and may result in more effective CR.6,7
Continue to: Double-balloon catheter...
Double-balloon catheter
The Cook double-balloon catheter for CR is meant to create pressure on both sides of the cervix, facilitating CR. Studies have reported that the Cook double-balloon catheter can be used for outpatient CR. In one study, 48 women with a low-risk pregnancy, at 37 to 42 weeks’ gestation and a Bishop score <7 were randomly assigned to outpatient or inpatient double-balloon CR.8 Both balloons were filled with 70 to 80 mL of sterile water. CTG monitoring was performed for 20 minutes before and after balloon placement. The women in the outpatient CR group were instructed to return to the labor unit the next day at 8 AM for IOL or earlier if they had regular uterine contractions, rupture of membranes, or vaginal bleeding. Seven percent of the women in the outpatient group returned to the labor unit before 8 AM. After removal of the balloon catheter, women in the outpatient and inpatient groups needed additional misoprostol CR in 12% and 13% of cases, respectively. Outcomes were similar in the two groups, but the study was not powered to identify small differences between the groups.
In another study of outpatient CR with the Cook double-balloon catheter, 695 women with a Bishop score <7, at ≥37 weeks’ gestation, were randomly assigned to outpatient CR with a double-balloon catheter or inpatient CR with dinoprostone (PGE2) (2 mg dinoprostone vaginal gel [Prostin] or dinoprostone 10 mg controlled-release tape (Cervidil).9 Women assigned to dinoprostone CR had CTG monitoring prior to commencing PGE2 CR and at least 30 min of CTG monitoring after insertion of the vaginal PGE2. Women assigned to balloon CR were not admitted to the hospital. CTG was performed prior to insertion of the balloon. After insertion, the two balloons on the catheter were each filled with 80 mL of saline. After catheter insertion CTG monitoring was not routinely performed. The women in the double-balloon catheter group returned to the labor unit 12 hours after insertion to initiate IOL. The primary outcome was composite neonatal morbidity and mortality, including admission to a neonatal intensive care unit (NICU), intubation, cardiac compressions, acidemia, hypoxic ischemic encephalopathy, seizure, infection, pulmonary hypertension, stillbirth, or death.
There was no significant difference in the rate of the primary outcome in the catheter versus the PGE2 group (18.6% and 25.8%; P = .07). Admission to the NICU occurred at rates of 12.6% and 15.5% in the catheter and PGE2 groups. Umbilical cord arterial pH <7.00 at birth occurred at a rate of 3.5% in the catheter group and 9.2% in the PGE2 group. The cesarean delivery rates in the catheter and PGE groups were 32.6% and 25.8%, respectively (P = .24). The investigators concluded that outpatient CR using a double-balloon catheter is safe and feasible for nulliparous women.
Two systematic reviews and meta-analyses reported that outcomes were similar when using the Foley or double-balloon catheter for CR.10,11 The Cook double-balloon CR kit includes a stylet, which can facilitate passing the catheter through the cervix.
Continue to: Dilapan-S and laminaria...
Dilapan-S and laminaria
There are many published studies using Dilapan-S and laminaria for cervical preparation prior to uterine evacuation.12 There are few published studies using Dilapan-S or laminaria for CR prior to IOL. In a pilot study, 21 patients were randomly assigned to outpatient versus inpatient Dilapan-S for CR the night prior to scheduled oxytocin IOL.13 The length of time from initiation of oxytocin to delivery in the outpatient and inpatient groups was similar (11 vs 14 hours, respectively). The outpatient compared with the inpatient group had a shorter length of hospitalization until delivery (51 vs 70 hours).
In other studies of Dilapan-S for CR, the patients remained in the hospital once the dilators were inserted. In one small trial, 41 women were randomized to CR with Dilapan-S or laminaria. As many dilators as could be comfortably tolerated by the patient were inserted.14 The mean numbers of Dilapan-S and laminaria dilators inserted were 4.3 and 9.7, respectively. The morning after the insertion of the dilators, oxytocin IOL was initiated. The times from initiation of oxytocin to delivery for the women in the Dilapan-S and laminaria groups were 11.6 and 15.5 hours, respectively.
An observational study reported on outcomes with Dilapan-S for CR on inpatients.15 In the study 444 women scheduled for IOL at 37 to 40 weeks’ gestation, with a mean baseline Bishop score of 2.9, had Dilapan-S placed for approximately 15 hours prior to oxytocin IOL. The mean number of Dilapan-S dilators that were inserted was 3.8. The study protocol prohibited placing more than 5 cervical dilator devices. The mean Bishop score after removal of the dilators was 6.5. The most common adverse effects of Dilapan-S CR were bleeding (2.7%) and pain (0.2%). The cesarean delivery rate in the cohort was 30.1%. An Apgar score <7 at 5 minutes was recorded for 3 newborns. An umbilical artery pH of <7.10 was observed in 8 newborns.
In a randomized trial performed on inpatients, 419 women undergoing CR were assigned to a Foley balloon or Dilapan-S.16 The vaginal delivery rates were similar in the groups—76% for Foley and 81% for Dilapan-S. Maternal and neonatal adverse effects were similar between the two groups. Compared with Foley catheter, women assigned to Dilapan-S reported greater satisfaction with their CR experience, more sleep, and more ability to perform daily activities.
Misoprostol and dinoprostone
Both misoprostol and dinoprostone are effective for outpatient CR. However, a Cochrane systematic review and meta-analysis concluded that balloon CR, compared with prostaglandin CR, is probably associated with a lower risk of uterine hyperstimulation with concerning fetal heart rate changes.17 Because misoprostol and dinoprostone occasionally can cause uterine hyperstimulation with fetal heart changes, many experts recommend CTG monitoring both before and after administration of misoprostol or dinoprostone for CR.
In a trial of outpatient versus inpatient vaginal PGE2 CR, 425 women at 37 to 42 weeks’ gestation were assigned randomly to outpatient or inpatient CR.18 All women had CTG monitoring for 20 minutes before and after vaginal placement of the PGE2 gel. The PGE2 dose was 2 mg for nulliparous and 1 mg for parous women. The cesarean delivery rates were similar in the outpatient and inpatient groups—22.3% and 22.9%, respectively. Among the women randomized to outpatient CR, 27 women (13%) could not be discharged home after administration of the vaginal PGE2 because of frequent uterine contractions or an abnormal fetal heart rate pattern. In addition, 64 women (30%) in the outpatient group returned to the hospital before scheduled induction because of frequent contractions. Maternal and neonatal complications were similar in the two groups. The investigators concluded that, at the dose and route of prostaglandin utilized in this study, the resultant rates of abnormal fetal heart rate pattern and frequent contractions might reduce the clinical utility of outpatient vaginal prostaglandin CR.
Another study also reported a greater rate of uterine tachysystole with vaginal PGE2 compared with a Foley catheter for CR (9% vs 0%).19 In a Cochrane systematic review of vaginal prostaglandin for CR, compared with placebo, vaginal prostaglandins were associated with a significantly greater rate of uterine hyperstimulation with fetal heart rate changes (4.8% vs 1.0%).20 Other studies also reported the feasibility of outpatient CR with vaginal prostaglandin.21,22
Both oral and vaginal misoprostol have been utilized for outpatient CR. In one study, 87 women with singleton pregnancy at 40 to 42 weeks’ gestation with a Bishop score <6 were randomized to outpatient CR with oral misoprostol (100 µg) or placebo.23 Following administration of the oral misoprostol, the women had 2 hours of CTG monitoring. The treatment was repeated daily for up to 3 days if there was no change in the cervix. If labor occurred, the patient was admitted to the labor unit for oxytocin IOL. The times from first dose of misoprostol or placebo to delivery were 46 and 84 hours (P<.001), respectively.
In another study, 49 women ≥40 weeks’ gestation with a Bishop score <5 were randomly assigned to receive outpatient oral misoprostol 25 µg or 50 µg.24 The dose could be repeated every 3 days over 9 days if ripening or labor had not been achieved. The women had CTG before administration of oral misoprostol. After the misoprostol dose, they had 2 hours of CTG monitoring. The number of doses received by the women assigned to the 50 µg group were 83%, 13%, and 4% for 1, 2, and 3 doses, respectively. The number of doses received by the women assigned to the 25 µg group were 58%, 26%, and 16% for 1, 2, and 3 doses, respectively. The mean intervals from initiation of CR to delivery in the 25 µg and the 50 µg groups were 3.9 and 2.5 days, respectively. The investigators reported no maternal or newborn adverse events, although the study was not powered to detect infrequent events.
Many studies have reported on the feasibility of outpatient CR with vaginal misoprostol.25-30 In one study, 77 women at 40 weeks’ gestation and a Bishop score ≤8 were randomized to a single dose of vaginal misoprostol 25 µg or gentle cervical examination (control).25 The women had 1 hour of CTG monitoring after the intervention. If they had regular contractions they were admitted to the birthing unit. If they had no regular contractions they were discharged home. For nulliparous women, the time from intervention to delivery in the misoprostol group was 4.9 days, and 8.1 days in the control group. For parous women, the times from intervention to delivery in the two groups were 3.8 and 6.9 days, respectively.
Continue to: Inclusion and exclusion criteria for outpatient CR...
Inclusion and exclusion criteria for outpatient CR
Outpatient CR should be limited to low-risk women with a singleton gestation, who have reliable access to transportation from home to the labor unit and have a clear understanding of the instructions for outpatient CR. Patient characteristics that may be utilized to offer office-based CR include:
- singleton pregnancy at 39 weeks’ and 0 days’ gestation through 40 weeks’ and 6 days’ gestation
- cephalic presentation
- Bishop score ≤6.
Women who should be excluded from outpatient CR include those with:
- contraindications to vaginal delivery
- fetal growth restriction
- abnormal umbilical artery Doppler results
- oligo- or polyhydramnios
- multiple gestation
- major fetal anomaly
- recent nonreactive fetal heart rate tracing
- maternal report of decreased fetal movement
- abnormal biophysical profile
- prior cesarean delivery
- recent vaginal bleeding
- gestational diabetes requiring medication treatment
- significant hypertension.
Practices should establish their own inclusion and exclusion criteria for ambulatory CR.
Safety of office-based ambulatory CR among low-risk women
Safety is a complex concept with experts often disagreeing on what level of safety is required to accept a new medical procedure. Establishing the safety of office-based ambulatory CR among low-risk women would require a very large cohort or randomized studies with at least a thousand participants. Only a few large studies focused on the safety of CR have been reported. Sciscione and colleagues reported a large observational study of inpatient transcervical Foley catheter for CR involving 1,905 women.31 They reported no adverse outcomes among term, singleton, uncomplicated pregnancies. They calculated that the 95% confidence interval (CI) for an adverse event was between 0.0% and 0.2%. In a meta-analysis of 26 studies including 5,563 women, the risk of chorioamnionitis during IOL was equivalent with pre-IOL Foley catheter CR (7.2%) or prostaglandin CR (7.2%) (relative risk, 0.96; 95% CI, 0.66–1.38).32
Two systematic reviews have reported that, compared with balloon CR, misoprostol CR is associated with an increased risk of uterine tachysystole.33-34 In a large retrospective study, compared with inpatient CR, outpatient CR with dinoprostone vaginal insert was not associated with an increased risk of newborn admission to the neonatal intensive care unit or a low Apgar score at 5 minutes after birth.35
Will you consider office-based CR in your obstetric practice?
As reviewed in this editorial, evolving data suggest that it is feasible to initiate CR in the office ambulatory setting prior to admission to the labor unit for additional CR or IOL. Many women prefer to complete CR at home after initiation in the office, rather than have CR in a labor unit or hospital setting.36 The transcervical balloon catheter has the most published data supporting the feasibility of ambulatory CR. Compared with misoprostol, the transcervical balloon catheter is associated with a low rate of uterine tachysystole. It may be a preferred method for outpatient CR. If placement of a transcervical balloon catheter is challenging, for example when the patient has a tightly closed cervix, oral misoprostol ambulatory CR may be an option if CTG monitoring is available in the office.
During the COVID pandemic, many in-person office visits have transitioned to virtual visits with the patient in their home. Historically, most cases of CR have been performed on labor and delivery units. It may be time for your practice to consider office-based ambulatory CR for low-risk women planning an IOL. Office-based ambulatory CR is a win for labor nurses who generally prefer to manage laboring patients rather than patients undergoing prolonged in-hospital CR. Outpatient CR is also a win for low-risk patients who prefer to be at home rather than in a labor unit. ●
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Repro Biol. 2017;210:1-6.
- Ausbeck EB, Jauk VC, Xue Y, et al. Outpatient Foley catheter for induction of labor in nulliparous women. Obstet Gynecol. 2020;136:597-606.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98:751-756.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Kuhlmann MJ, Spencer N, Garcia-Jasso C, et al. Foley bulb insertion by blind placement compared with direct visualization. Obstet Gynecol. 2021;137:139-145.
- Delaney S, Shaffer BL, Chen YW, et al. Labor induction with a Foley balloon inflated to 30 mL compared with 60 mL. Obstet Gynecol. 2015;115:1239-1245.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Beckmann M, Gibbons K, Flenady V, et al. Induction of labor using prostaglandin E2 as an inpatient versus balloon catheter as an outpatient: a multicenter randomised controlled trial. BJOG. 2020;127:571-579.
- Liu X, Wang Y, Zhange F, et al. Double- versus single-balloon catheters for labour induction and cervical ripening: a meta-analysis. BMC Pregnancy Childbirth. 2019;19:358.
- Yang F, Huan S, Long Y, et al. Double-balloon versus single-balloon catheter for cervical ripening and labor induction: a systematic review and meta-analysis. J Obstet Gynaecol Res. 2018;44: 27-34.
- Goldberg AB, Fortin JA, Drey EA, et al. Cervical preparation before dilation and evacuation using adjunctive misoprostol and mifepristone compared with overnight osmotic dilators alone: a randomized controlled trial. Obstet Gynecol. 2015;126:599-609.
- Upadhyaya NB, Childs KD, Neiger R, et al. Ambulatory cervical ripening in term pregnancy. J Reprod Med. 1999;44:363-366.
- Blumenthal PD, Rmanauskas R. Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor. Obstet Gynecol. 1990;75:365-368.
- Gupta J, Chodankar R, Baev O, et al. Synthetic osmotic dilators in the induction of labour—an international multicenter observational study. Eur J Obstet Gynecol Repro Biol. 2018;229:70-75.
- Saad AF, Villarreal J, Eid J, et al. A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial). Am J Obstet Gynecol. 2019;220:275.e1-e9.
- de Vaan MD, Eikleder MLT, Jozwiak M, et al. Mechanical methods for induction of labour. Cochrane Database Syst Rev. 2019;CD001233.
- Wilkinson C, Bryce R, Adelson P, et al. A randomized controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E2 (OPRA study). BJOG. 2015;122:94-104.
- Blair R, Harvey MA, Pudwell J, et al. Retrospective comparison of PGE2 vaginal insert and Foley catheter for outpatient cervical ripening. J Obstet Gynaecol Can. 2020;42:1103-1110.
- Thomas J, Fairclough A, Kavanagh J, et al. Vaginal prostaglandin (PGE2 or PGF2alpha) for induction of labour at term. Cochrane Database Syst Rev. 2014;CD003101.
- O’Brien JM, Mercer BM, Cleary NT, et al. Efficacy of outpatient induction with low-dose intravaginal prostaglandin E2: a randomized, doubleblind, placebo controlled trial. Am J Obstet Gynecol. 1995;173:1855-1859.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26: 673-677.
- Kipikasa JH, Adair CD, Williamson J, et al. Use of misoprostol on an outpatient basis for postdate pregnancy. Int J Gynaecol Obstet. 2005;88:108-111.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening without subsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96:684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labor. Arch Gynecol Obstet. 2017;295:33-38.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs inpatient administration. Obstet Gynecol Surv. 2006;61:167-168.
- Meyer M, Pflum J, Howard D. Outpatient misoprostol compared with dinoprostone gel for preinduction cervical ripening: a randomized controlled trial. Obstet Gynecol. 2005;105:466-472.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- McMaster K, Sanchez-Ramos L, Kaunitz AM. Evaluation of a transcervical Foley catheter as a source of infection. Obstet Gynecol. 2015;126:539-551.
- Fox NS, Saltzman DH, Roman AS, et al. Intravaginal misoprostol versus Foley catheter for labour induction: a meta-analysis. BJOG. 2011;118: 647-654.
- Hofmeyr GJ, Gulmezoglu AM, Pileggi C. Vaginal misoprostol for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2010:CD000941.
- Salvador SC, Simpson ML, Cundiff GW. Dinoprostone vaginal insert for labour induction: a comparison of outpatient and inpatient settings. J Obstet Gynaecol Can. 2009;31:1028-1034.
- Sutton C, Harding J, Griffin C. Patient attitudes towards outpatient cervical ripening prior to induction of labour at an Australian tertiary hospital. J Obstet Gynaecol. 2016;36:921-928.
- Policiano C, Pimenta M, Martins D, et al. Outpatient versus inpatient cervix priming with Foley catheter: a randomized trial. Eur J Obstet Gynecol Repro Biol. 2017;210:1-6.
- Ausbeck EB, Jauk VC, Xue Y, et al. Outpatient Foley catheter for induction of labor in nulliparous women. Obstet Gynecol. 2020;136:597-606.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Sciscione AC, Muench M, Pollock M, et al. Transcervical Foley catheter for preinduction cervical ripening in an outpatient versus inpatient setting. Obstet Gynecol. 2001;98:751-756.
- Henry A, Madan A, Reid R, et al. Outpatient Foley catheter versus inpatient prostaglandin E2 gel for induction of labour: a randomised trial. BMC Pregnancy Childbirth. 2013;13:25.
- Kuhlmann MJ, Spencer N, Garcia-Jasso C, et al. Foley bulb insertion by blind placement compared with direct visualization. Obstet Gynecol. 2021;137:139-145.
- Delaney S, Shaffer BL, Chen YW, et al. Labor induction with a Foley balloon inflated to 30 mL compared with 60 mL. Obstet Gynecol. 2015;115:1239-1245.
- Wilkinson C, Adelson P, Turnbull D. A comparison of inpatient with outpatient balloon catheter cervical ripening: a pilot randomized controlled trial. BMC Pregnancy Childbirth. 2015;15:126.
- Beckmann M, Gibbons K, Flenady V, et al. Induction of labor using prostaglandin E2 as an inpatient versus balloon catheter as an outpatient: a multicenter randomised controlled trial. BJOG. 2020;127:571-579.
- Liu X, Wang Y, Zhange F, et al. Double- versus single-balloon catheters for labour induction and cervical ripening: a meta-analysis. BMC Pregnancy Childbirth. 2019;19:358.
- Yang F, Huan S, Long Y, et al. Double-balloon versus single-balloon catheter for cervical ripening and labor induction: a systematic review and meta-analysis. J Obstet Gynaecol Res. 2018;44: 27-34.
- Goldberg AB, Fortin JA, Drey EA, et al. Cervical preparation before dilation and evacuation using adjunctive misoprostol and mifepristone compared with overnight osmotic dilators alone: a randomized controlled trial. Obstet Gynecol. 2015;126:599-609.
- Upadhyaya NB, Childs KD, Neiger R, et al. Ambulatory cervical ripening in term pregnancy. J Reprod Med. 1999;44:363-366.
- Blumenthal PD, Rmanauskas R. Randomized trial of Dilapan and Laminaria as cervical ripening agents before induction of labor. Obstet Gynecol. 1990;75:365-368.
- Gupta J, Chodankar R, Baev O, et al. Synthetic osmotic dilators in the induction of labour—an international multicenter observational study. Eur J Obstet Gynecol Repro Biol. 2018;229:70-75.
- Saad AF, Villarreal J, Eid J, et al. A randomized controlled trial of Dilapan-S vs Foley balloon for preinduction cervical ripening (DILAFOL trial). Am J Obstet Gynecol. 2019;220:275.e1-e9.
- de Vaan MD, Eikleder MLT, Jozwiak M, et al. Mechanical methods for induction of labour. Cochrane Database Syst Rev. 2019;CD001233.
- Wilkinson C, Bryce R, Adelson P, et al. A randomized controlled trial of outpatient compared with inpatient cervical ripening with prostaglandin E2 (OPRA study). BJOG. 2015;122:94-104.
- Blair R, Harvey MA, Pudwell J, et al. Retrospective comparison of PGE2 vaginal insert and Foley catheter for outpatient cervical ripening. J Obstet Gynaecol Can. 2020;42:1103-1110.
- Thomas J, Fairclough A, Kavanagh J, et al. Vaginal prostaglandin (PGE2 or PGF2alpha) for induction of labour at term. Cochrane Database Syst Rev. 2014;CD003101.
- O’Brien JM, Mercer BM, Cleary NT, et al. Efficacy of outpatient induction with low-dose intravaginal prostaglandin E2: a randomized, doubleblind, placebo controlled trial. Am J Obstet Gynecol. 1995;173:1855-1859.
- Biem SR, Turnell RW, Olatunbosun O, et al. A randomized controlled trial of outpatient versus inpatient labour induction with vaginal controlled-release prostaglandin-E2: effectiveness and satisfaction. J Obstet Gynaecol Can. 2003;25:23-31.
- Gaffaney CA, Saul LL, Rumney PJ, et al. Outpatient oral misoprostol for prolonged pregnancies: a pilot investigation. Am J Perinatol. 2009;26: 673-677.
- Kipikasa JH, Adair CD, Williamson J, et al. Use of misoprostol on an outpatient basis for postdate pregnancy. Int J Gynaecol Obstet. 2005;88:108-111.
- Oboro VO, Tabowei TO. Outpatient misoprostol cervical ripening without subsequent induction of labor to prevent post-term pregnancy. Acta Obstet Gynecol Scand. 2005;84:628-631.
- Stitely ML, Browning J, Fowler M, et al. Outpatient cervical ripening with intravaginal misoprostol. Obstet Gynecol. 2000;96:684-688.
- McKenna DS, Ester JB, Proffitt M, et al. Misoprostol outpatient cervical ripening without subsequent induction of labor: a randomized trial. Obstet Gynecol. 2004;104:579-584.
- PonMalar J, Benjamin SJ, Abraham A, et al. Randomized double-blind placebo controlled study of preinduction cervical priming with 25 µg of misoprostol in the outpatient setting to prevent formal induction of labor. Arch Gynecol Obstet. 2017;295:33-38.
- Chang DW, Velazquez MD, Colyer M, et al. Vaginal misoprostol for cervical ripening at term: comparison of outpatient vs inpatient administration. Obstet Gynecol Surv. 2006;61:167-168.
- Meyer M, Pflum J, Howard D. Outpatient misoprostol compared with dinoprostone gel for preinduction cervical ripening: a randomized controlled trial. Obstet Gynecol. 2005;105:466-472.
- Sciscione AC, Bedder CL, Hoffman MK, et al. The timing of adverse events with Foley catheter preinduction cervical ripening; implications for outpatient use. Am J Perinatol. 2014;31:781-786.
- McMaster K, Sanchez-Ramos L, Kaunitz AM. Evaluation of a transcervical Foley catheter as a source of infection. Obstet Gynecol. 2015;126:539-551.
- Fox NS, Saltzman DH, Roman AS, et al. Intravaginal misoprostol versus Foley catheter for labour induction: a meta-analysis. BJOG. 2011;118: 647-654.
- Hofmeyr GJ, Gulmezoglu AM, Pileggi C. Vaginal misoprostol for cervical ripening and induction of labour. Cochrane Database Syst Rev. 2010:CD000941.
- Salvador SC, Simpson ML, Cundiff GW. Dinoprostone vaginal insert for labour induction: a comparison of outpatient and inpatient settings. J Obstet Gynaecol Can. 2009;31:1028-1034.
- Sutton C, Harding J, Griffin C. Patient attitudes towards outpatient cervical ripening prior to induction of labour at an Australian tertiary hospital. J Obstet Gynaecol. 2016;36:921-928.
Maternal chronic conditions predict cerebral palsy in offspring
Several maternal chronic conditions increase the risk of giving birth to a child with cerebral palsy, based on data from more than 1.3 million Norwegian children.
Mothers with autoimmune disorders, such as diabetes and lupus, had the greatest risks, reported lead author Marianne S. Strøm, MD, of the University of Bergen (Norway) and colleagues.
“The etiologies of cerebral palsy are complex, and only a few prenatal risk factors have been identified,” the investigators wrote in Pediatrics. “Among these possible risk factors are maternal chronic conditions, although studies are typically underpowered and limited to one or two conditions.”
According to Dr. Strøm and colleagues, several components of maternal chronic conditions have been linked with cerebral palsy, including placental abnormalities, altered thrombotic state, and inflammation. Furthermore, mothers with chronic conditions are more likely to give birth prematurely and have children with congenital malformations, both of which have also been associated with cerebral palsy.
To date, however, “there has been no systematic description of maternal chronic conditions and risk of cerebral palsy in offspring,” the investigators noted.
The present, prospective cohort study aimed to meet this need with a population of 1,360,149 children born in Norway from 1990 to 2012, among whom 3,575 had cerebral palsy. Case data were extracted from the Norwegian Patient Registry and the National Insurance Scheme. Information about maternal chronic conditions was extracted from the Medical Birth Registry of Norway and the Norwegian Patient Registry, with the latter also providing information about paternal chronic conditions.
Using log binomial regression models, the investigators determined relative risks of having children with cerebral palsy among parents with chronic conditions versus parents from the general population. This revealed that chronic conditions in fathers had no correlation with cerebral palsy. In contrast, mothers with chronic conditions had a 30% increased risk (relative risk, 1.3; 95% confidence interval, 1.2-1.5), which could be further stratified by number of chronic conditions; mothers with one chronic condition, for instance, had a 20% increased risk (RR, 1.2; 95% CI, 1.1-1.4), while those with two chronic conditions had a 60% increased risk (RR, 1.6; 95% CI, 1.1-2.2), and those with more than two chronic conditions had triple the risk (RR, 3.1; 95% CI, 1.4-6.8)
“The lack of associations between the father’s chronic illness and cerebral palsy risk supports the interpretation that cerebral palsy risk in offspring is the direct result of the mother’s condition and not genetic predisposition or unmeasured situational factors,” the investigators wrote.
Maternal autoimmune conditions were particularly relevant, as they were associated with a 40% increased risk of cerebral palsy (RR, 1.4; 95% CI, 1.1-1.7), a rate that climbed dramatically, to 270%, among mothers with more than one autoimmune condition (RR, 2.7; 95% CI, 1.1-6.6).
“The role of autoimmune diseases in cerebral palsy risk (and maternal inflammation specifically) deserves closer attention,” the investigators wrote. “Using studies with larger sample sizes and a more clinical focus, including measures of placental structure and perinatal blood assays, researchers may be able to explore these possible connections between maternal autoimmune diseases and fetal neurodevelopment.”
Specifically, cerebral palsy in offspring was most strongly associated with maternal Crohn’s disease (RR, 2.1; 95% CI, 1.0-4.1), type 1 diabetes (RR, 2.2; 95% CI, 1.4-3.4), lupus erythematosus (RR, 2.7; 95% CI, 0.9-8.3), and type 2 diabetes (RR, 3.2; 95% CI, 1.8-5.4). Associations were also found for migraine (RR, 1.6; 95% CI, 1.2-2.2), multiple sclerosis (RR, 1.8; 95% CI, 0.8-4.4), and rheumatoid arthritis (RR, 2.0; 95% CI, 1.3-2.9). Several “weaker and less convincing associations” were detected for ulcerative colitis, thyroid disorder, epilepsy, asthma, anemia, and hypertension. Adjusting for parental education level, age, smoking status, and single-mother status did not significantly alter findings. Poisson and logistic regression models generated similar results.
In an accompanying editorial, Sandra Julsen Hollung, PhD, of the Cerebral Palsy Registry of Norway, Vestfold Hospital Trust, Tønsberg, and colleagues, advised that clinicians maintain perspective when discussing these findings with the general public.
“As the authors state, the absolute risk of cerebral palsy associated with at least one chronic maternal condition is low,” wrote Dr. Hollung and colleagues. “Among 1,000 pregnant women with any chronic and/or autoimmune disorder, more than 990 will deliver an infant who will not be diagnosed with cerebral palsy.”
They went on to emphasize that the study findings should not be viewed as firm evidence of causal relationships.
“Thus, the study cannot give clues to any specific preventive treatment,” wrote Dr. Hollung and colleagues. “However, if these disorders are part of a causal pathway, optimal treatment might reduce the risk of cerebral palsy.”
Although Dr. Hollung and colleagues advised that such efforts “would hardly affect the birth prevalence of cerebral palsy,” they also cited the Royal College of Obstetricians and Gynaecologists in the United Kingdom, noting that “each baby counts.”
Emeritus Professor Alastair MacLennan, AO, MB ChB, FRCOG, FRANZCOG, head of the Australian Collaborative Cerebral Palsy Research Group at the University of Adelaide (Australia) suggested that the findings may guide future research.
“An increasing proportion of cerebral palsy cases are being diagnosed by genome sequencing and other genetic techniques to have causative genetic variations,” Dr. MacLennan said. “The possibility of epigenetic interactions are also likely and are still to be investigated. Maternal disorders such as diabetes, lupus, or Crohn’s disease are possible epigenetic factors and this study helps to target these in future genetic and environmental studies of cerebral palsy causation. The days of attributing cerebral palsy to ‘birth asphyxia’ are over.”
The study was supported by the National Institutes of Health and the Western Norwegian Regional Health Authorities. The investigators reported no conflicts of interest.
Several maternal chronic conditions increase the risk of giving birth to a child with cerebral palsy, based on data from more than 1.3 million Norwegian children.
Mothers with autoimmune disorders, such as diabetes and lupus, had the greatest risks, reported lead author Marianne S. Strøm, MD, of the University of Bergen (Norway) and colleagues.
“The etiologies of cerebral palsy are complex, and only a few prenatal risk factors have been identified,” the investigators wrote in Pediatrics. “Among these possible risk factors are maternal chronic conditions, although studies are typically underpowered and limited to one or two conditions.”
According to Dr. Strøm and colleagues, several components of maternal chronic conditions have been linked with cerebral palsy, including placental abnormalities, altered thrombotic state, and inflammation. Furthermore, mothers with chronic conditions are more likely to give birth prematurely and have children with congenital malformations, both of which have also been associated with cerebral palsy.
To date, however, “there has been no systematic description of maternal chronic conditions and risk of cerebral palsy in offspring,” the investigators noted.
The present, prospective cohort study aimed to meet this need with a population of 1,360,149 children born in Norway from 1990 to 2012, among whom 3,575 had cerebral palsy. Case data were extracted from the Norwegian Patient Registry and the National Insurance Scheme. Information about maternal chronic conditions was extracted from the Medical Birth Registry of Norway and the Norwegian Patient Registry, with the latter also providing information about paternal chronic conditions.
Using log binomial regression models, the investigators determined relative risks of having children with cerebral palsy among parents with chronic conditions versus parents from the general population. This revealed that chronic conditions in fathers had no correlation with cerebral palsy. In contrast, mothers with chronic conditions had a 30% increased risk (relative risk, 1.3; 95% confidence interval, 1.2-1.5), which could be further stratified by number of chronic conditions; mothers with one chronic condition, for instance, had a 20% increased risk (RR, 1.2; 95% CI, 1.1-1.4), while those with two chronic conditions had a 60% increased risk (RR, 1.6; 95% CI, 1.1-2.2), and those with more than two chronic conditions had triple the risk (RR, 3.1; 95% CI, 1.4-6.8)
“The lack of associations between the father’s chronic illness and cerebral palsy risk supports the interpretation that cerebral palsy risk in offspring is the direct result of the mother’s condition and not genetic predisposition or unmeasured situational factors,” the investigators wrote.
Maternal autoimmune conditions were particularly relevant, as they were associated with a 40% increased risk of cerebral palsy (RR, 1.4; 95% CI, 1.1-1.7), a rate that climbed dramatically, to 270%, among mothers with more than one autoimmune condition (RR, 2.7; 95% CI, 1.1-6.6).
“The role of autoimmune diseases in cerebral palsy risk (and maternal inflammation specifically) deserves closer attention,” the investigators wrote. “Using studies with larger sample sizes and a more clinical focus, including measures of placental structure and perinatal blood assays, researchers may be able to explore these possible connections between maternal autoimmune diseases and fetal neurodevelopment.”
Specifically, cerebral palsy in offspring was most strongly associated with maternal Crohn’s disease (RR, 2.1; 95% CI, 1.0-4.1), type 1 diabetes (RR, 2.2; 95% CI, 1.4-3.4), lupus erythematosus (RR, 2.7; 95% CI, 0.9-8.3), and type 2 diabetes (RR, 3.2; 95% CI, 1.8-5.4). Associations were also found for migraine (RR, 1.6; 95% CI, 1.2-2.2), multiple sclerosis (RR, 1.8; 95% CI, 0.8-4.4), and rheumatoid arthritis (RR, 2.0; 95% CI, 1.3-2.9). Several “weaker and less convincing associations” were detected for ulcerative colitis, thyroid disorder, epilepsy, asthma, anemia, and hypertension. Adjusting for parental education level, age, smoking status, and single-mother status did not significantly alter findings. Poisson and logistic regression models generated similar results.
In an accompanying editorial, Sandra Julsen Hollung, PhD, of the Cerebral Palsy Registry of Norway, Vestfold Hospital Trust, Tønsberg, and colleagues, advised that clinicians maintain perspective when discussing these findings with the general public.
“As the authors state, the absolute risk of cerebral palsy associated with at least one chronic maternal condition is low,” wrote Dr. Hollung and colleagues. “Among 1,000 pregnant women with any chronic and/or autoimmune disorder, more than 990 will deliver an infant who will not be diagnosed with cerebral palsy.”
They went on to emphasize that the study findings should not be viewed as firm evidence of causal relationships.
“Thus, the study cannot give clues to any specific preventive treatment,” wrote Dr. Hollung and colleagues. “However, if these disorders are part of a causal pathway, optimal treatment might reduce the risk of cerebral palsy.”
Although Dr. Hollung and colleagues advised that such efforts “would hardly affect the birth prevalence of cerebral palsy,” they also cited the Royal College of Obstetricians and Gynaecologists in the United Kingdom, noting that “each baby counts.”
Emeritus Professor Alastair MacLennan, AO, MB ChB, FRCOG, FRANZCOG, head of the Australian Collaborative Cerebral Palsy Research Group at the University of Adelaide (Australia) suggested that the findings may guide future research.
“An increasing proportion of cerebral palsy cases are being diagnosed by genome sequencing and other genetic techniques to have causative genetic variations,” Dr. MacLennan said. “The possibility of epigenetic interactions are also likely and are still to be investigated. Maternal disorders such as diabetes, lupus, or Crohn’s disease are possible epigenetic factors and this study helps to target these in future genetic and environmental studies of cerebral palsy causation. The days of attributing cerebral palsy to ‘birth asphyxia’ are over.”
The study was supported by the National Institutes of Health and the Western Norwegian Regional Health Authorities. The investigators reported no conflicts of interest.
Several maternal chronic conditions increase the risk of giving birth to a child with cerebral palsy, based on data from more than 1.3 million Norwegian children.
Mothers with autoimmune disorders, such as diabetes and lupus, had the greatest risks, reported lead author Marianne S. Strøm, MD, of the University of Bergen (Norway) and colleagues.
“The etiologies of cerebral palsy are complex, and only a few prenatal risk factors have been identified,” the investigators wrote in Pediatrics. “Among these possible risk factors are maternal chronic conditions, although studies are typically underpowered and limited to one or two conditions.”
According to Dr. Strøm and colleagues, several components of maternal chronic conditions have been linked with cerebral palsy, including placental abnormalities, altered thrombotic state, and inflammation. Furthermore, mothers with chronic conditions are more likely to give birth prematurely and have children with congenital malformations, both of which have also been associated with cerebral palsy.
To date, however, “there has been no systematic description of maternal chronic conditions and risk of cerebral palsy in offspring,” the investigators noted.
The present, prospective cohort study aimed to meet this need with a population of 1,360,149 children born in Norway from 1990 to 2012, among whom 3,575 had cerebral palsy. Case data were extracted from the Norwegian Patient Registry and the National Insurance Scheme. Information about maternal chronic conditions was extracted from the Medical Birth Registry of Norway and the Norwegian Patient Registry, with the latter also providing information about paternal chronic conditions.
Using log binomial regression models, the investigators determined relative risks of having children with cerebral palsy among parents with chronic conditions versus parents from the general population. This revealed that chronic conditions in fathers had no correlation with cerebral palsy. In contrast, mothers with chronic conditions had a 30% increased risk (relative risk, 1.3; 95% confidence interval, 1.2-1.5), which could be further stratified by number of chronic conditions; mothers with one chronic condition, for instance, had a 20% increased risk (RR, 1.2; 95% CI, 1.1-1.4), while those with two chronic conditions had a 60% increased risk (RR, 1.6; 95% CI, 1.1-2.2), and those with more than two chronic conditions had triple the risk (RR, 3.1; 95% CI, 1.4-6.8)
“The lack of associations between the father’s chronic illness and cerebral palsy risk supports the interpretation that cerebral palsy risk in offspring is the direct result of the mother’s condition and not genetic predisposition or unmeasured situational factors,” the investigators wrote.
Maternal autoimmune conditions were particularly relevant, as they were associated with a 40% increased risk of cerebral palsy (RR, 1.4; 95% CI, 1.1-1.7), a rate that climbed dramatically, to 270%, among mothers with more than one autoimmune condition (RR, 2.7; 95% CI, 1.1-6.6).
“The role of autoimmune diseases in cerebral palsy risk (and maternal inflammation specifically) deserves closer attention,” the investigators wrote. “Using studies with larger sample sizes and a more clinical focus, including measures of placental structure and perinatal blood assays, researchers may be able to explore these possible connections between maternal autoimmune diseases and fetal neurodevelopment.”
Specifically, cerebral palsy in offspring was most strongly associated with maternal Crohn’s disease (RR, 2.1; 95% CI, 1.0-4.1), type 1 diabetes (RR, 2.2; 95% CI, 1.4-3.4), lupus erythematosus (RR, 2.7; 95% CI, 0.9-8.3), and type 2 diabetes (RR, 3.2; 95% CI, 1.8-5.4). Associations were also found for migraine (RR, 1.6; 95% CI, 1.2-2.2), multiple sclerosis (RR, 1.8; 95% CI, 0.8-4.4), and rheumatoid arthritis (RR, 2.0; 95% CI, 1.3-2.9). Several “weaker and less convincing associations” were detected for ulcerative colitis, thyroid disorder, epilepsy, asthma, anemia, and hypertension. Adjusting for parental education level, age, smoking status, and single-mother status did not significantly alter findings. Poisson and logistic regression models generated similar results.
In an accompanying editorial, Sandra Julsen Hollung, PhD, of the Cerebral Palsy Registry of Norway, Vestfold Hospital Trust, Tønsberg, and colleagues, advised that clinicians maintain perspective when discussing these findings with the general public.
“As the authors state, the absolute risk of cerebral palsy associated with at least one chronic maternal condition is low,” wrote Dr. Hollung and colleagues. “Among 1,000 pregnant women with any chronic and/or autoimmune disorder, more than 990 will deliver an infant who will not be diagnosed with cerebral palsy.”
They went on to emphasize that the study findings should not be viewed as firm evidence of causal relationships.
“Thus, the study cannot give clues to any specific preventive treatment,” wrote Dr. Hollung and colleagues. “However, if these disorders are part of a causal pathway, optimal treatment might reduce the risk of cerebral palsy.”
Although Dr. Hollung and colleagues advised that such efforts “would hardly affect the birth prevalence of cerebral palsy,” they also cited the Royal College of Obstetricians and Gynaecologists in the United Kingdom, noting that “each baby counts.”
Emeritus Professor Alastair MacLennan, AO, MB ChB, FRCOG, FRANZCOG, head of the Australian Collaborative Cerebral Palsy Research Group at the University of Adelaide (Australia) suggested that the findings may guide future research.
“An increasing proportion of cerebral palsy cases are being diagnosed by genome sequencing and other genetic techniques to have causative genetic variations,” Dr. MacLennan said. “The possibility of epigenetic interactions are also likely and are still to be investigated. Maternal disorders such as diabetes, lupus, or Crohn’s disease are possible epigenetic factors and this study helps to target these in future genetic and environmental studies of cerebral palsy causation. The days of attributing cerebral palsy to ‘birth asphyxia’ are over.”
The study was supported by the National Institutes of Health and the Western Norwegian Regional Health Authorities. The investigators reported no conflicts of interest.
FROM PEDIATRICS
Consideration of herbal products in pregnancy and lactation
In recent decades, natural products have had increased consumer attention in industrialized nations. One of the challenges is that “natural” can be more of a perception than a standard. “Herbal products” is a more frequently used and perhaps a more apt term. Herbal products come in many forms, including herbs used in food preparation, teas, infusions, caplets, dried extracts, essential oils, and tinctures.
Multiple prescription medications have pharmacologically active compounds that originated from herbal products, both historically and currently. Examples include the cardiac stimulant digoxin (foxglove plant), the antimalarial quinine (Cinchona bark), and antihypertensives (Rauwolfia serpentina). Indeed, the first pharmacologically active compound, morphine, was extracted from the seed pods of opium poppies approximately 200 years ago. This demonstrated that medications could be purified from plants and that a precise dose could be determined for administration. However, herbal products are grown and harvested in varying seasonal conditions and soil types, which, over time and geography, may contribute to variability in the levels of active compound in the final products.
The importance of active compound purification and consistent precise dosage in herbal products brings up the topic of regulation. Herbal products are considered dietary supplements and as such are Food and Drug Administration regulated as a food under the 1994 Dietary Supplement Health Education Act. Regulation as a food product does not involve the same level of scrutiny as a medication. There is no requirement that manufacturers check for purity and consistency of their product’s active compound(s). Manufacturers must ensure that the claims they make about herbal products are not false or misleading. They must also support their claims with evidence. However, there is no requirement for the manufacturers to submit this evidence to the FDA. This can translate into a discrepancy between the claim on the product label and scientific evidence that the product does what it claims to do. In other words, the product may not be effective.
With uncertain efficacy, the safety of herbal products comes into focus. Very few herbal products (or their specific active compounds) have been scientifically studied for safety in pregnancy and lactation. Further, herbal products may contain contaminants. Metals such as lead and mercury occur naturally. Yet, because of human activities, both may have collected in areas where herbal products are grown. From a safety perspective, both can be concerning in pregnancy or lactation. Lead and mercury are two examples of metal contaminants. Other contaminants may include pesticides, chemicals, and bacteria or other microorganisms. Some liquid herbal products such as tinctures contain alcohol, which should be avoided in pregnancy. An additional consideration would be the potential for herbal products, including any of their known or unknown product contents, to interact with prescribed medications or anesthesia.
Select examples of herbal products
Astragalus is the root of an herb and it is used for reasons of boosting immunity, energy, and other functions. These and its purported promotion of breast milk flow (galactagogue) are unsupported. Safety concerns include irregular heartbeat and dizziness, rendering it unsafe for use in pregnancy and of unknown efficacy and safety in lactation.
Kombucha is an herbal product made from leaves (tea), sugar, a culture, and other varying products. Like many herbal products, it is both manufactured and home brewed. It is used for probiotic and antioxidant reasons. As a fermented product, kombucha may contain 0.2%-0.5% alcohol. There is no known safe level of alcohol and no known safe type of alcohol for use in pregnancy. Alcohol exposure in pregnancy can result in fetal alcohol spectrum disorders, involving a range of birth defects and life-long intellectual, learning and behavioral disorders. Alcohol found in breast milk approximates the level of alcohol found in the maternal bloodstream. Alcohol-containing products should be avoided in pregnancy and lactation.
Nux vomica is an herbal product and is used for reasons of reducing nausea or vomiting in pregnancy. It comes from the raw seeds (toxic) of an evergreen tree. It has serious safety concerns and yet it is still in use. It contains strychnine, which can harm both the pregnant individual and the developing fetus. It is not recommended in lactation.
Red raspberry leaf is a leaf, brewed and ingested as a tea. It is used for reasons of preventing miscarriage, relieving nausea and stomach discomfort, toning the uterus, reducing labor pain, increasing breast milk production, and other functions. In low doses, it appears to be safe. In high doses, it can induce smooth muscle relaxation. Efficacy has not been demonstrated with labor and delivery or in increasing breast milk production.
Tabacum is an herbal product and is used for reasons of reducing nausea or vomiting in pregnancy. Its full name is Nicotiana tabacum (tobacco) and it contains 2%-8% nicotine, which should be avoided in pregnancy. Nicotine is a health danger for the pregnant individual and can damage a developing fetus’ brain and lungs.
Unless otherwise scientifically demonstrated, herbal products should be considered medications with pharmacologic activity, potential adverse effects, and potential toxicity in pregnancy and lactation. It’s easy for a patient to forget about reporting any nonprescription medications during a patient-provider visit. As a provider, purposefully asking about all over-the-counter and herbal products during each visit can prompt the patient to provide this important information. Further, it may facilitate discussion about the continuation/discontinuation of products of unknown safety and unknown benefit, culminating in the serious reflection: “Is it really worth the risk?”
For further information about the safety of herbal products, consult local Poison Control Centers, MothertoBaby, MothertoBaby affiliates, and the National Institutes of Health Drugs and Lactation Database, LactMed.
Dr. Hardy is a consultant on global maternal-child health and pharmacoepidemiology, and represents the Society for Birth Defects Research and Prevention and the Organization of Teratology Information Specialists at PRGLAC meetings. Dr. Hardy has worked with multiple pharmaceutical manufacturers regarding studies of medication safety in pregnancy, most recently Biohaven Pharmaceuticals, New Haven, CT.
.
In recent decades, natural products have had increased consumer attention in industrialized nations. One of the challenges is that “natural” can be more of a perception than a standard. “Herbal products” is a more frequently used and perhaps a more apt term. Herbal products come in many forms, including herbs used in food preparation, teas, infusions, caplets, dried extracts, essential oils, and tinctures.
Multiple prescription medications have pharmacologically active compounds that originated from herbal products, both historically and currently. Examples include the cardiac stimulant digoxin (foxglove plant), the antimalarial quinine (Cinchona bark), and antihypertensives (Rauwolfia serpentina). Indeed, the first pharmacologically active compound, morphine, was extracted from the seed pods of opium poppies approximately 200 years ago. This demonstrated that medications could be purified from plants and that a precise dose could be determined for administration. However, herbal products are grown and harvested in varying seasonal conditions and soil types, which, over time and geography, may contribute to variability in the levels of active compound in the final products.
The importance of active compound purification and consistent precise dosage in herbal products brings up the topic of regulation. Herbal products are considered dietary supplements and as such are Food and Drug Administration regulated as a food under the 1994 Dietary Supplement Health Education Act. Regulation as a food product does not involve the same level of scrutiny as a medication. There is no requirement that manufacturers check for purity and consistency of their product’s active compound(s). Manufacturers must ensure that the claims they make about herbal products are not false or misleading. They must also support their claims with evidence. However, there is no requirement for the manufacturers to submit this evidence to the FDA. This can translate into a discrepancy between the claim on the product label and scientific evidence that the product does what it claims to do. In other words, the product may not be effective.
With uncertain efficacy, the safety of herbal products comes into focus. Very few herbal products (or their specific active compounds) have been scientifically studied for safety in pregnancy and lactation. Further, herbal products may contain contaminants. Metals such as lead and mercury occur naturally. Yet, because of human activities, both may have collected in areas where herbal products are grown. From a safety perspective, both can be concerning in pregnancy or lactation. Lead and mercury are two examples of metal contaminants. Other contaminants may include pesticides, chemicals, and bacteria or other microorganisms. Some liquid herbal products such as tinctures contain alcohol, which should be avoided in pregnancy. An additional consideration would be the potential for herbal products, including any of their known or unknown product contents, to interact with prescribed medications or anesthesia.
Select examples of herbal products
Astragalus is the root of an herb and it is used for reasons of boosting immunity, energy, and other functions. These and its purported promotion of breast milk flow (galactagogue) are unsupported. Safety concerns include irregular heartbeat and dizziness, rendering it unsafe for use in pregnancy and of unknown efficacy and safety in lactation.
Kombucha is an herbal product made from leaves (tea), sugar, a culture, and other varying products. Like many herbal products, it is both manufactured and home brewed. It is used for probiotic and antioxidant reasons. As a fermented product, kombucha may contain 0.2%-0.5% alcohol. There is no known safe level of alcohol and no known safe type of alcohol for use in pregnancy. Alcohol exposure in pregnancy can result in fetal alcohol spectrum disorders, involving a range of birth defects and life-long intellectual, learning and behavioral disorders. Alcohol found in breast milk approximates the level of alcohol found in the maternal bloodstream. Alcohol-containing products should be avoided in pregnancy and lactation.
Nux vomica is an herbal product and is used for reasons of reducing nausea or vomiting in pregnancy. It comes from the raw seeds (toxic) of an evergreen tree. It has serious safety concerns and yet it is still in use. It contains strychnine, which can harm both the pregnant individual and the developing fetus. It is not recommended in lactation.
Red raspberry leaf is a leaf, brewed and ingested as a tea. It is used for reasons of preventing miscarriage, relieving nausea and stomach discomfort, toning the uterus, reducing labor pain, increasing breast milk production, and other functions. In low doses, it appears to be safe. In high doses, it can induce smooth muscle relaxation. Efficacy has not been demonstrated with labor and delivery or in increasing breast milk production.
Tabacum is an herbal product and is used for reasons of reducing nausea or vomiting in pregnancy. Its full name is Nicotiana tabacum (tobacco) and it contains 2%-8% nicotine, which should be avoided in pregnancy. Nicotine is a health danger for the pregnant individual and can damage a developing fetus’ brain and lungs.
Unless otherwise scientifically demonstrated, herbal products should be considered medications with pharmacologic activity, potential adverse effects, and potential toxicity in pregnancy and lactation. It’s easy for a patient to forget about reporting any nonprescription medications during a patient-provider visit. As a provider, purposefully asking about all over-the-counter and herbal products during each visit can prompt the patient to provide this important information. Further, it may facilitate discussion about the continuation/discontinuation of products of unknown safety and unknown benefit, culminating in the serious reflection: “Is it really worth the risk?”
For further information about the safety of herbal products, consult local Poison Control Centers, MothertoBaby, MothertoBaby affiliates, and the National Institutes of Health Drugs and Lactation Database, LactMed.
Dr. Hardy is a consultant on global maternal-child health and pharmacoepidemiology, and represents the Society for Birth Defects Research and Prevention and the Organization of Teratology Information Specialists at PRGLAC meetings. Dr. Hardy has worked with multiple pharmaceutical manufacturers regarding studies of medication safety in pregnancy, most recently Biohaven Pharmaceuticals, New Haven, CT.
.
In recent decades, natural products have had increased consumer attention in industrialized nations. One of the challenges is that “natural” can be more of a perception than a standard. “Herbal products” is a more frequently used and perhaps a more apt term. Herbal products come in many forms, including herbs used in food preparation, teas, infusions, caplets, dried extracts, essential oils, and tinctures.
Multiple prescription medications have pharmacologically active compounds that originated from herbal products, both historically and currently. Examples include the cardiac stimulant digoxin (foxglove plant), the antimalarial quinine (Cinchona bark), and antihypertensives (Rauwolfia serpentina). Indeed, the first pharmacologically active compound, morphine, was extracted from the seed pods of opium poppies approximately 200 years ago. This demonstrated that medications could be purified from plants and that a precise dose could be determined for administration. However, herbal products are grown and harvested in varying seasonal conditions and soil types, which, over time and geography, may contribute to variability in the levels of active compound in the final products.
The importance of active compound purification and consistent precise dosage in herbal products brings up the topic of regulation. Herbal products are considered dietary supplements and as such are Food and Drug Administration regulated as a food under the 1994 Dietary Supplement Health Education Act. Regulation as a food product does not involve the same level of scrutiny as a medication. There is no requirement that manufacturers check for purity and consistency of their product’s active compound(s). Manufacturers must ensure that the claims they make about herbal products are not false or misleading. They must also support their claims with evidence. However, there is no requirement for the manufacturers to submit this evidence to the FDA. This can translate into a discrepancy between the claim on the product label and scientific evidence that the product does what it claims to do. In other words, the product may not be effective.
With uncertain efficacy, the safety of herbal products comes into focus. Very few herbal products (or their specific active compounds) have been scientifically studied for safety in pregnancy and lactation. Further, herbal products may contain contaminants. Metals such as lead and mercury occur naturally. Yet, because of human activities, both may have collected in areas where herbal products are grown. From a safety perspective, both can be concerning in pregnancy or lactation. Lead and mercury are two examples of metal contaminants. Other contaminants may include pesticides, chemicals, and bacteria or other microorganisms. Some liquid herbal products such as tinctures contain alcohol, which should be avoided in pregnancy. An additional consideration would be the potential for herbal products, including any of their known or unknown product contents, to interact with prescribed medications or anesthesia.
Select examples of herbal products
Astragalus is the root of an herb and it is used for reasons of boosting immunity, energy, and other functions. These and its purported promotion of breast milk flow (galactagogue) are unsupported. Safety concerns include irregular heartbeat and dizziness, rendering it unsafe for use in pregnancy and of unknown efficacy and safety in lactation.
Kombucha is an herbal product made from leaves (tea), sugar, a culture, and other varying products. Like many herbal products, it is both manufactured and home brewed. It is used for probiotic and antioxidant reasons. As a fermented product, kombucha may contain 0.2%-0.5% alcohol. There is no known safe level of alcohol and no known safe type of alcohol for use in pregnancy. Alcohol exposure in pregnancy can result in fetal alcohol spectrum disorders, involving a range of birth defects and life-long intellectual, learning and behavioral disorders. Alcohol found in breast milk approximates the level of alcohol found in the maternal bloodstream. Alcohol-containing products should be avoided in pregnancy and lactation.
Nux vomica is an herbal product and is used for reasons of reducing nausea or vomiting in pregnancy. It comes from the raw seeds (toxic) of an evergreen tree. It has serious safety concerns and yet it is still in use. It contains strychnine, which can harm both the pregnant individual and the developing fetus. It is not recommended in lactation.
Red raspberry leaf is a leaf, brewed and ingested as a tea. It is used for reasons of preventing miscarriage, relieving nausea and stomach discomfort, toning the uterus, reducing labor pain, increasing breast milk production, and other functions. In low doses, it appears to be safe. In high doses, it can induce smooth muscle relaxation. Efficacy has not been demonstrated with labor and delivery or in increasing breast milk production.
Tabacum is an herbal product and is used for reasons of reducing nausea or vomiting in pregnancy. Its full name is Nicotiana tabacum (tobacco) and it contains 2%-8% nicotine, which should be avoided in pregnancy. Nicotine is a health danger for the pregnant individual and can damage a developing fetus’ brain and lungs.
Unless otherwise scientifically demonstrated, herbal products should be considered medications with pharmacologic activity, potential adverse effects, and potential toxicity in pregnancy and lactation. It’s easy for a patient to forget about reporting any nonprescription medications during a patient-provider visit. As a provider, purposefully asking about all over-the-counter and herbal products during each visit can prompt the patient to provide this important information. Further, it may facilitate discussion about the continuation/discontinuation of products of unknown safety and unknown benefit, culminating in the serious reflection: “Is it really worth the risk?”
For further information about the safety of herbal products, consult local Poison Control Centers, MothertoBaby, MothertoBaby affiliates, and the National Institutes of Health Drugs and Lactation Database, LactMed.
Dr. Hardy is a consultant on global maternal-child health and pharmacoepidemiology, and represents the Society for Birth Defects Research and Prevention and the Organization of Teratology Information Specialists at PRGLAC meetings. Dr. Hardy has worked with multiple pharmaceutical manufacturers regarding studies of medication safety in pregnancy, most recently Biohaven Pharmaceuticals, New Haven, CT.
.