User login
For MD-IQ use only
Sharing Cancer Care Information Across VA Health Care Systems (FULL)
A telementoring program based on the Specialty Care Access Network Extension for Community Healthcare Outcomes model shared information about cancer care across VA health Care systems.
In 2016, the Cancer Care Coordinator at the US Department of Veterans Affairs (VA) Connecticut Healthcare System (VACT) in West Haven partnered with the VA New England Healthcare System to use its telementoring program. The VA Specialty Care Access Network Extension for Community Healthcare Outcomes (VA ECHO) was used to present a series of educational conferences on cancer care. This article describes our experience implementing the program and reviews participant feedback gathered from voluntary surveys.
Background
In 2011, the Veterans Health Administration (VHA) Office of Healthcare Transformation launched VA ECHO, a telementoring program for primary care providers (PCPs) and patient-aligned care team staff. VACT was selected as 1 of 7 hub sites across the US. The VA ECHO system uses video and online technology to provide PCPs with case-based specialist consultation and didactic education. The system enables providers at any VA location to participate in online and telephone conferences in real time. The presentations are recorded and made available online to VA providers through a secure site.
VA ECHO is based on the highly successful Project ECHO model established by Sanjeev Arora and the University of New Mexico in 2007.1 The rationale for Project ECHO was that patient care could be improved by increasing the competence of PCPs in the management of complex diseases by providing access to disease specialists through a case-based learning approach that used technology, which it termed knowledge networks, to connect the PCPs to specialists.
The original model addressed management of hepatitis C in a medically underserved area where half of the population was widely geographically dispersed, making the provision of specialty care challenging. Developers identified 6 characteristics that make a disease appropriate for treatment using the Project ECHO knowledge network model:
- The disease is common;
- Management of the disease is complex;
- Treatment for the disease is evolving;
- The disease has a high societal impact;
- There are serious outcomes if the disease is not treated; and
- Disease management improves outcomes.1
VA ECHO conferences are available to all VA personnel. Staff can subscribe to an e-mail group list to be alerted to conference times and topics. Participants can connect directly to the conference using Microsoft Outlook Lync or Skype (Redmond, WA) and see the slides in real time on their computer as they listen to the presentation. The presentations are recorded, and the slides with audio can be accessed easily on the VA ECHO SharePoint site for download, enabling VA staff to listen to conferences at their convenience (Figure).
VA Cancer ECHO
The impetus to create a series of talks related to cancer care using VA ECHO was the frequent and often time-consuming requests we received from colleagues at other VA sites for information about areas of cancer care, such as survivorship and cancer care coordination. It was felt that presenting cancer care information as a VA ECHO series would make this information available to a large group of providers at one time, making the method more time effective than sharing the information via one-on-one conversations.
The cancer care coordinator originally conceived this as a 3-part, 1-time series to present work done at VACT in the areas of survivorship, psychosocial distress monitoring, and coordination of cancer care using the VA Cancer Care Tracking System, an online tracking tool. Information about the series was disseminated via VA group e-mail lists for oncology providers and via the existing VA ECHO subscriber invitation process. The 3-presentation series garnered positive feedback and had attendance that ranged from 49 to 75 participants (mean, 60). Participants expressed enthusiasm for the format via e-mail and phone feedback directly to the West Haven staff.
Expansion
The success of this original 3-part series led to a trial of an ongoing Cancer Care Conference series (Conference) using VA ECHO. This was a novel use of VA ECHO and was outside its traditional format, which is geared to discussion of individual cases and clinical knowledge. Nevertheless, this new style of communication has been embraced by a wide range of VA cancer care professionals.
One reason we considered expanding the program was that oncology fit the framework of the original Project ECHO knowledge network model. Cancer is common at the VA, which cares for 175,000 patients with cancer annually.2 The management of cancer is complex involving many disciplines working together, and treatments are constantly changing. In addition, cancer has a high societal impact; there are serious outcomes both in terms of patient survival and patient symptom burden. And lastly, outcomes are improved with proactive disease management that is informed by the most current, evidence-based medicine.
The Conference was conceived as a forum for providers across disciplines to share best practices and discuss common challenges in caring for veterans with cancer. We invited participants to submit proposals for presentations related to cancer care initiatives at their VA sites. Potential speakers across all areas of care for veterans with cancer were invited to submit possible topics for the conference. The submissions were reviewed by the moderators in an effort to create a series of talks on a variety of topics across all aspects of care for oncology patients in the VA. This process of effectively crowd-sourcing educational content inspires providers to think more creatively about their practice and quality improvement projects and has sparked an ongoing dialogue about quality initiatives among VA oncology providers across disciplines and geographic locations. As a result, this approach also has enabled participants to learn from colleagues who work at a wide range of rural and urban VA locations throughout the country and to network with colleagues who are working on similar quality initiatives and challenges related to caring for veterans with cancer.
Program
The first Conference talk was in October 2016. It encompassed ten 1-hour talks during the 2016 to 2017 academic year. Speakers were recruited from the VACT West Haven campus and from several other VA sites nationwide. Topics included survivorship, psychosocial distress, palliative care, cancer navigation, and establishing a clinical trials program.
In its first year, the Conference series had 260 unique attendees representing such disciplines as medicine, nursing, social work, pharmacy, psychology, and clinic administration and representing all 21 Veterans Integrated Services Networks (VISNs). Speakers including oncologists, hepatologists, cancer care coordinators, health psychologists, and a research coordinator gave presentations on psychosocial distress screening and issues, cognitive behavioral therapy for cancer pain, cancer navigation, cancer case tracking, VISN-based liver cancer tumor tracker and liver tumor board, starting a VA-based clinical trial, palliative care, and survivorship.
The Conference accounted for 508 continuing medical education (CME) hours, which accounted for one-third of the total CME hours generated by the VACT West Haven VA ECHO program. Highlights of the talks were presented at the 2017 Association of VA Hematology/Oncology annual meeting in Denver, Colorado.
During the second year of the Conference, speakers were recruited to address new American College of Surgeons Commission on Cancer (CoC) requirements regarding survivorship treatment summaries for a subset of cancer survivors.3 The focus on survivorship was driven by ongoing feedback from participants who were working on initiatives to implement this process at their VA sites and wanted to learn from peers involved in this process throughout the VA system. Several speakers gave talks on implementing survivorship care at their VA and specifically on the use of computerized patient record system templates to create survivorship treatment summaries for veterans in accordance with CoC standards.
Since the first Conference in 2016, the number of unique attendees grew by 20% to 327 in 2018. During its first 2 years, participants have earned a total of 1,095 CME credits through Yale University CME. Conferences are usually broadcast at noon eastern time so that providers can take advantage of sessions during lunch breaks.
Participant Surveys
Attendees were invited to participate in voluntary, anonymous surveys to obtain feedback on and to receive input on topics of interest for future talks. Participants also were asked to comment on resources that they utilized to be updated on practice changes (Table 1).
The Conference has led to increased awareness of other continuing education opportunities available through VA ECHO-Connecticut. Of survey participants, 20% reported that they had attended other VA ECHO conferences.
The survey samples are self-selecting and may not necessarily be representative of the Conference participants or of the VA oncology interdisciplinary team as a whole; however, the relatively large number of survey participants provides some confidence that these survey results can help inform future planning for this and other continuing education opportunities for VA oncology providers.
An additional online survey was designed to elucidate whether participants were incorporating knowledge gained from the Conference in their cancer care practice. Half of the 32 participants strongly agreed with the following statement: “Participation in the VA Cancer Care Conference has added to my knowledge of information relevant to my practice,” and 13 more agreed with the statement for a total of 90.6% of those surveyed responding affirmatively. Only 3 participants neither agreed nor disagreed, and none disagreed with the statement. More than half of the participants reported that they made changes to their practice or plan to make changes as a result of the Conference.
Conculsion
The VA ECHO program established at the VACT West Haven campus in 2012 now offers regular monthly or bimonthly conferences in 9 specialties: pain, liver/hepatitis C, neurology, nephrology, cardiology, diabetes/endocrinology, mental health and addiction, nursing grand rounds, and cancer care. The VACT ECHO program is led by a medical director, and each specialty has a clinical director who conducts sessions and recruits other specialists from their department.
Teleconferencing can provide opportunities for colleagues living in distant locations to connect; share best practices, common goals, and challenges; and initiate ongoing and lasting relationships. The Conference draws the most diverse audience by discipline of all the VA ECHO conferences hosted at VACT (Table 2).
Traditionally, the national VA ECHO program has been a forum for specialists to discuss clinical case presentations for the benefit of primary care providers and to deliver didactics about chronic clinical conditions. Our Cancer Care Management VA ECHO has explored new ground by discussing material that has helped sites set up and enhance cancer care clinics and disseminate best practices for cancer survivorship and other aspects of cancer care. As a result, this conference has attracted and provided a forum for the most diverse audience of staff among VA ECHO clinics, with participation from clinic administrators to social workers to primary care providers to tumor registrars.
Through the creation of the Conference, > 300 individuals who care for veterans with cancer have been provided with a regular forum at which to connect with colleagues, receive updates on new treatment options for their patients, and learn about and share best practices specific to VA oncology patients. The VA ECHO technology creates a resource that can be accessed by all VA staff from their desktop computer. The VA ECHO SharePoint saves the slides of the Conference presentations both with and without audio to enable staff who can’t participate in real time to access the information at their convenience.
The Conference has facilitated networking among VA oncology providers who have common interests. Conference participants also have participated in other VA ECHO conferences in disciplines beyond oncology. Participants in the Conference also are encouraged to participate as speakers by presenting quality improvement initiatives at their VA site. This novel approach to generating content for this educational series has led to a dynamic interchange of ideas and increased networking among VA providers related to their practice and quality improvement initiatives at their VA sites. The Conference provides a regular forum for VA staff across a wide range of disciplines to share personal experiences, successes, and frustrations and to get feedback from colleagues.
The Conference combines a structured approach to presenting VA-specific educational content related to cancer care and multiple mechanisms that encourage staff to participate in an ongoing dialogue related to quality initiatives both on the phone during the Conference, online using Outlook LYNC or Skype to ask questions during the Conference, and during conversations on group e-mail. The Conference promotes staff engagement at little or no extra cost to the VA. For more information about the VA ECHO Cancer Care Conference or to submit a presentation for consideration for a future session, please contact [email protected] or [email protected].
1. Arora S, Geppert CM, Kalishman S, et al. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Acad Med. 2007;82(2):154-160.
2. Hematology and oncology federal health care data trends. Fed Pract. 2017;33(suppl 5):S12-S15.
3. American College of Surgeons Commission on Cancer. Cancer Program Standards: Ensuring Patient Centered Care, 2016 Edition. https://www.facs.org/quality-programs/cancer/coc/standards. Accessed March 14, 2018.
A telementoring program based on the Specialty Care Access Network Extension for Community Healthcare Outcomes model shared information about cancer care across VA health Care systems.
A telementoring program based on the Specialty Care Access Network Extension for Community Healthcare Outcomes model shared information about cancer care across VA health Care systems.
In 2016, the Cancer Care Coordinator at the US Department of Veterans Affairs (VA) Connecticut Healthcare System (VACT) in West Haven partnered with the VA New England Healthcare System to use its telementoring program. The VA Specialty Care Access Network Extension for Community Healthcare Outcomes (VA ECHO) was used to present a series of educational conferences on cancer care. This article describes our experience implementing the program and reviews participant feedback gathered from voluntary surveys.
Background
In 2011, the Veterans Health Administration (VHA) Office of Healthcare Transformation launched VA ECHO, a telementoring program for primary care providers (PCPs) and patient-aligned care team staff. VACT was selected as 1 of 7 hub sites across the US. The VA ECHO system uses video and online technology to provide PCPs with case-based specialist consultation and didactic education. The system enables providers at any VA location to participate in online and telephone conferences in real time. The presentations are recorded and made available online to VA providers through a secure site.
VA ECHO is based on the highly successful Project ECHO model established by Sanjeev Arora and the University of New Mexico in 2007.1 The rationale for Project ECHO was that patient care could be improved by increasing the competence of PCPs in the management of complex diseases by providing access to disease specialists through a case-based learning approach that used technology, which it termed knowledge networks, to connect the PCPs to specialists.
The original model addressed management of hepatitis C in a medically underserved area where half of the population was widely geographically dispersed, making the provision of specialty care challenging. Developers identified 6 characteristics that make a disease appropriate for treatment using the Project ECHO knowledge network model:
- The disease is common;
- Management of the disease is complex;
- Treatment for the disease is evolving;
- The disease has a high societal impact;
- There are serious outcomes if the disease is not treated; and
- Disease management improves outcomes.1
VA ECHO conferences are available to all VA personnel. Staff can subscribe to an e-mail group list to be alerted to conference times and topics. Participants can connect directly to the conference using Microsoft Outlook Lync or Skype (Redmond, WA) and see the slides in real time on their computer as they listen to the presentation. The presentations are recorded, and the slides with audio can be accessed easily on the VA ECHO SharePoint site for download, enabling VA staff to listen to conferences at their convenience (Figure).
VA Cancer ECHO
The impetus to create a series of talks related to cancer care using VA ECHO was the frequent and often time-consuming requests we received from colleagues at other VA sites for information about areas of cancer care, such as survivorship and cancer care coordination. It was felt that presenting cancer care information as a VA ECHO series would make this information available to a large group of providers at one time, making the method more time effective than sharing the information via one-on-one conversations.
The cancer care coordinator originally conceived this as a 3-part, 1-time series to present work done at VACT in the areas of survivorship, psychosocial distress monitoring, and coordination of cancer care using the VA Cancer Care Tracking System, an online tracking tool. Information about the series was disseminated via VA group e-mail lists for oncology providers and via the existing VA ECHO subscriber invitation process. The 3-presentation series garnered positive feedback and had attendance that ranged from 49 to 75 participants (mean, 60). Participants expressed enthusiasm for the format via e-mail and phone feedback directly to the West Haven staff.
Expansion
The success of this original 3-part series led to a trial of an ongoing Cancer Care Conference series (Conference) using VA ECHO. This was a novel use of VA ECHO and was outside its traditional format, which is geared to discussion of individual cases and clinical knowledge. Nevertheless, this new style of communication has been embraced by a wide range of VA cancer care professionals.
One reason we considered expanding the program was that oncology fit the framework of the original Project ECHO knowledge network model. Cancer is common at the VA, which cares for 175,000 patients with cancer annually.2 The management of cancer is complex involving many disciplines working together, and treatments are constantly changing. In addition, cancer has a high societal impact; there are serious outcomes both in terms of patient survival and patient symptom burden. And lastly, outcomes are improved with proactive disease management that is informed by the most current, evidence-based medicine.
The Conference was conceived as a forum for providers across disciplines to share best practices and discuss common challenges in caring for veterans with cancer. We invited participants to submit proposals for presentations related to cancer care initiatives at their VA sites. Potential speakers across all areas of care for veterans with cancer were invited to submit possible topics for the conference. The submissions were reviewed by the moderators in an effort to create a series of talks on a variety of topics across all aspects of care for oncology patients in the VA. This process of effectively crowd-sourcing educational content inspires providers to think more creatively about their practice and quality improvement projects and has sparked an ongoing dialogue about quality initiatives among VA oncology providers across disciplines and geographic locations. As a result, this approach also has enabled participants to learn from colleagues who work at a wide range of rural and urban VA locations throughout the country and to network with colleagues who are working on similar quality initiatives and challenges related to caring for veterans with cancer.
Program
The first Conference talk was in October 2016. It encompassed ten 1-hour talks during the 2016 to 2017 academic year. Speakers were recruited from the VACT West Haven campus and from several other VA sites nationwide. Topics included survivorship, psychosocial distress, palliative care, cancer navigation, and establishing a clinical trials program.
In its first year, the Conference series had 260 unique attendees representing such disciplines as medicine, nursing, social work, pharmacy, psychology, and clinic administration and representing all 21 Veterans Integrated Services Networks (VISNs). Speakers including oncologists, hepatologists, cancer care coordinators, health psychologists, and a research coordinator gave presentations on psychosocial distress screening and issues, cognitive behavioral therapy for cancer pain, cancer navigation, cancer case tracking, VISN-based liver cancer tumor tracker and liver tumor board, starting a VA-based clinical trial, palliative care, and survivorship.
The Conference accounted for 508 continuing medical education (CME) hours, which accounted for one-third of the total CME hours generated by the VACT West Haven VA ECHO program. Highlights of the talks were presented at the 2017 Association of VA Hematology/Oncology annual meeting in Denver, Colorado.
During the second year of the Conference, speakers were recruited to address new American College of Surgeons Commission on Cancer (CoC) requirements regarding survivorship treatment summaries for a subset of cancer survivors.3 The focus on survivorship was driven by ongoing feedback from participants who were working on initiatives to implement this process at their VA sites and wanted to learn from peers involved in this process throughout the VA system. Several speakers gave talks on implementing survivorship care at their VA and specifically on the use of computerized patient record system templates to create survivorship treatment summaries for veterans in accordance with CoC standards.
Since the first Conference in 2016, the number of unique attendees grew by 20% to 327 in 2018. During its first 2 years, participants have earned a total of 1,095 CME credits through Yale University CME. Conferences are usually broadcast at noon eastern time so that providers can take advantage of sessions during lunch breaks.
Participant Surveys
Attendees were invited to participate in voluntary, anonymous surveys to obtain feedback on and to receive input on topics of interest for future talks. Participants also were asked to comment on resources that they utilized to be updated on practice changes (Table 1).
The Conference has led to increased awareness of other continuing education opportunities available through VA ECHO-Connecticut. Of survey participants, 20% reported that they had attended other VA ECHO conferences.
The survey samples are self-selecting and may not necessarily be representative of the Conference participants or of the VA oncology interdisciplinary team as a whole; however, the relatively large number of survey participants provides some confidence that these survey results can help inform future planning for this and other continuing education opportunities for VA oncology providers.
An additional online survey was designed to elucidate whether participants were incorporating knowledge gained from the Conference in their cancer care practice. Half of the 32 participants strongly agreed with the following statement: “Participation in the VA Cancer Care Conference has added to my knowledge of information relevant to my practice,” and 13 more agreed with the statement for a total of 90.6% of those surveyed responding affirmatively. Only 3 participants neither agreed nor disagreed, and none disagreed with the statement. More than half of the participants reported that they made changes to their practice or plan to make changes as a result of the Conference.
Conculsion
The VA ECHO program established at the VACT West Haven campus in 2012 now offers regular monthly or bimonthly conferences in 9 specialties: pain, liver/hepatitis C, neurology, nephrology, cardiology, diabetes/endocrinology, mental health and addiction, nursing grand rounds, and cancer care. The VACT ECHO program is led by a medical director, and each specialty has a clinical director who conducts sessions and recruits other specialists from their department.
Teleconferencing can provide opportunities for colleagues living in distant locations to connect; share best practices, common goals, and challenges; and initiate ongoing and lasting relationships. The Conference draws the most diverse audience by discipline of all the VA ECHO conferences hosted at VACT (Table 2).
Traditionally, the national VA ECHO program has been a forum for specialists to discuss clinical case presentations for the benefit of primary care providers and to deliver didactics about chronic clinical conditions. Our Cancer Care Management VA ECHO has explored new ground by discussing material that has helped sites set up and enhance cancer care clinics and disseminate best practices for cancer survivorship and other aspects of cancer care. As a result, this conference has attracted and provided a forum for the most diverse audience of staff among VA ECHO clinics, with participation from clinic administrators to social workers to primary care providers to tumor registrars.
Through the creation of the Conference, > 300 individuals who care for veterans with cancer have been provided with a regular forum at which to connect with colleagues, receive updates on new treatment options for their patients, and learn about and share best practices specific to VA oncology patients. The VA ECHO technology creates a resource that can be accessed by all VA staff from their desktop computer. The VA ECHO SharePoint saves the slides of the Conference presentations both with and without audio to enable staff who can’t participate in real time to access the information at their convenience.
The Conference has facilitated networking among VA oncology providers who have common interests. Conference participants also have participated in other VA ECHO conferences in disciplines beyond oncology. Participants in the Conference also are encouraged to participate as speakers by presenting quality improvement initiatives at their VA site. This novel approach to generating content for this educational series has led to a dynamic interchange of ideas and increased networking among VA providers related to their practice and quality improvement initiatives at their VA sites. The Conference provides a regular forum for VA staff across a wide range of disciplines to share personal experiences, successes, and frustrations and to get feedback from colleagues.
The Conference combines a structured approach to presenting VA-specific educational content related to cancer care and multiple mechanisms that encourage staff to participate in an ongoing dialogue related to quality initiatives both on the phone during the Conference, online using Outlook LYNC or Skype to ask questions during the Conference, and during conversations on group e-mail. The Conference promotes staff engagement at little or no extra cost to the VA. For more information about the VA ECHO Cancer Care Conference or to submit a presentation for consideration for a future session, please contact [email protected] or [email protected].
In 2016, the Cancer Care Coordinator at the US Department of Veterans Affairs (VA) Connecticut Healthcare System (VACT) in West Haven partnered with the VA New England Healthcare System to use its telementoring program. The VA Specialty Care Access Network Extension for Community Healthcare Outcomes (VA ECHO) was used to present a series of educational conferences on cancer care. This article describes our experience implementing the program and reviews participant feedback gathered from voluntary surveys.
Background
In 2011, the Veterans Health Administration (VHA) Office of Healthcare Transformation launched VA ECHO, a telementoring program for primary care providers (PCPs) and patient-aligned care team staff. VACT was selected as 1 of 7 hub sites across the US. The VA ECHO system uses video and online technology to provide PCPs with case-based specialist consultation and didactic education. The system enables providers at any VA location to participate in online and telephone conferences in real time. The presentations are recorded and made available online to VA providers through a secure site.
VA ECHO is based on the highly successful Project ECHO model established by Sanjeev Arora and the University of New Mexico in 2007.1 The rationale for Project ECHO was that patient care could be improved by increasing the competence of PCPs in the management of complex diseases by providing access to disease specialists through a case-based learning approach that used technology, which it termed knowledge networks, to connect the PCPs to specialists.
The original model addressed management of hepatitis C in a medically underserved area where half of the population was widely geographically dispersed, making the provision of specialty care challenging. Developers identified 6 characteristics that make a disease appropriate for treatment using the Project ECHO knowledge network model:
- The disease is common;
- Management of the disease is complex;
- Treatment for the disease is evolving;
- The disease has a high societal impact;
- There are serious outcomes if the disease is not treated; and
- Disease management improves outcomes.1
VA ECHO conferences are available to all VA personnel. Staff can subscribe to an e-mail group list to be alerted to conference times and topics. Participants can connect directly to the conference using Microsoft Outlook Lync or Skype (Redmond, WA) and see the slides in real time on their computer as they listen to the presentation. The presentations are recorded, and the slides with audio can be accessed easily on the VA ECHO SharePoint site for download, enabling VA staff to listen to conferences at their convenience (Figure).
VA Cancer ECHO
The impetus to create a series of talks related to cancer care using VA ECHO was the frequent and often time-consuming requests we received from colleagues at other VA sites for information about areas of cancer care, such as survivorship and cancer care coordination. It was felt that presenting cancer care information as a VA ECHO series would make this information available to a large group of providers at one time, making the method more time effective than sharing the information via one-on-one conversations.
The cancer care coordinator originally conceived this as a 3-part, 1-time series to present work done at VACT in the areas of survivorship, psychosocial distress monitoring, and coordination of cancer care using the VA Cancer Care Tracking System, an online tracking tool. Information about the series was disseminated via VA group e-mail lists for oncology providers and via the existing VA ECHO subscriber invitation process. The 3-presentation series garnered positive feedback and had attendance that ranged from 49 to 75 participants (mean, 60). Participants expressed enthusiasm for the format via e-mail and phone feedback directly to the West Haven staff.
Expansion
The success of this original 3-part series led to a trial of an ongoing Cancer Care Conference series (Conference) using VA ECHO. This was a novel use of VA ECHO and was outside its traditional format, which is geared to discussion of individual cases and clinical knowledge. Nevertheless, this new style of communication has been embraced by a wide range of VA cancer care professionals.
One reason we considered expanding the program was that oncology fit the framework of the original Project ECHO knowledge network model. Cancer is common at the VA, which cares for 175,000 patients with cancer annually.2 The management of cancer is complex involving many disciplines working together, and treatments are constantly changing. In addition, cancer has a high societal impact; there are serious outcomes both in terms of patient survival and patient symptom burden. And lastly, outcomes are improved with proactive disease management that is informed by the most current, evidence-based medicine.
The Conference was conceived as a forum for providers across disciplines to share best practices and discuss common challenges in caring for veterans with cancer. We invited participants to submit proposals for presentations related to cancer care initiatives at their VA sites. Potential speakers across all areas of care for veterans with cancer were invited to submit possible topics for the conference. The submissions were reviewed by the moderators in an effort to create a series of talks on a variety of topics across all aspects of care for oncology patients in the VA. This process of effectively crowd-sourcing educational content inspires providers to think more creatively about their practice and quality improvement projects and has sparked an ongoing dialogue about quality initiatives among VA oncology providers across disciplines and geographic locations. As a result, this approach also has enabled participants to learn from colleagues who work at a wide range of rural and urban VA locations throughout the country and to network with colleagues who are working on similar quality initiatives and challenges related to caring for veterans with cancer.
Program
The first Conference talk was in October 2016. It encompassed ten 1-hour talks during the 2016 to 2017 academic year. Speakers were recruited from the VACT West Haven campus and from several other VA sites nationwide. Topics included survivorship, psychosocial distress, palliative care, cancer navigation, and establishing a clinical trials program.
In its first year, the Conference series had 260 unique attendees representing such disciplines as medicine, nursing, social work, pharmacy, psychology, and clinic administration and representing all 21 Veterans Integrated Services Networks (VISNs). Speakers including oncologists, hepatologists, cancer care coordinators, health psychologists, and a research coordinator gave presentations on psychosocial distress screening and issues, cognitive behavioral therapy for cancer pain, cancer navigation, cancer case tracking, VISN-based liver cancer tumor tracker and liver tumor board, starting a VA-based clinical trial, palliative care, and survivorship.
The Conference accounted for 508 continuing medical education (CME) hours, which accounted for one-third of the total CME hours generated by the VACT West Haven VA ECHO program. Highlights of the talks were presented at the 2017 Association of VA Hematology/Oncology annual meeting in Denver, Colorado.
During the second year of the Conference, speakers were recruited to address new American College of Surgeons Commission on Cancer (CoC) requirements regarding survivorship treatment summaries for a subset of cancer survivors.3 The focus on survivorship was driven by ongoing feedback from participants who were working on initiatives to implement this process at their VA sites and wanted to learn from peers involved in this process throughout the VA system. Several speakers gave talks on implementing survivorship care at their VA and specifically on the use of computerized patient record system templates to create survivorship treatment summaries for veterans in accordance with CoC standards.
Since the first Conference in 2016, the number of unique attendees grew by 20% to 327 in 2018. During its first 2 years, participants have earned a total of 1,095 CME credits through Yale University CME. Conferences are usually broadcast at noon eastern time so that providers can take advantage of sessions during lunch breaks.
Participant Surveys
Attendees were invited to participate in voluntary, anonymous surveys to obtain feedback on and to receive input on topics of interest for future talks. Participants also were asked to comment on resources that they utilized to be updated on practice changes (Table 1).
The Conference has led to increased awareness of other continuing education opportunities available through VA ECHO-Connecticut. Of survey participants, 20% reported that they had attended other VA ECHO conferences.
The survey samples are self-selecting and may not necessarily be representative of the Conference participants or of the VA oncology interdisciplinary team as a whole; however, the relatively large number of survey participants provides some confidence that these survey results can help inform future planning for this and other continuing education opportunities for VA oncology providers.
An additional online survey was designed to elucidate whether participants were incorporating knowledge gained from the Conference in their cancer care practice. Half of the 32 participants strongly agreed with the following statement: “Participation in the VA Cancer Care Conference has added to my knowledge of information relevant to my practice,” and 13 more agreed with the statement for a total of 90.6% of those surveyed responding affirmatively. Only 3 participants neither agreed nor disagreed, and none disagreed with the statement. More than half of the participants reported that they made changes to their practice or plan to make changes as a result of the Conference.
Conculsion
The VA ECHO program established at the VACT West Haven campus in 2012 now offers regular monthly or bimonthly conferences in 9 specialties: pain, liver/hepatitis C, neurology, nephrology, cardiology, diabetes/endocrinology, mental health and addiction, nursing grand rounds, and cancer care. The VACT ECHO program is led by a medical director, and each specialty has a clinical director who conducts sessions and recruits other specialists from their department.
Teleconferencing can provide opportunities for colleagues living in distant locations to connect; share best practices, common goals, and challenges; and initiate ongoing and lasting relationships. The Conference draws the most diverse audience by discipline of all the VA ECHO conferences hosted at VACT (Table 2).
Traditionally, the national VA ECHO program has been a forum for specialists to discuss clinical case presentations for the benefit of primary care providers and to deliver didactics about chronic clinical conditions. Our Cancer Care Management VA ECHO has explored new ground by discussing material that has helped sites set up and enhance cancer care clinics and disseminate best practices for cancer survivorship and other aspects of cancer care. As a result, this conference has attracted and provided a forum for the most diverse audience of staff among VA ECHO clinics, with participation from clinic administrators to social workers to primary care providers to tumor registrars.
Through the creation of the Conference, > 300 individuals who care for veterans with cancer have been provided with a regular forum at which to connect with colleagues, receive updates on new treatment options for their patients, and learn about and share best practices specific to VA oncology patients. The VA ECHO technology creates a resource that can be accessed by all VA staff from their desktop computer. The VA ECHO SharePoint saves the slides of the Conference presentations both with and without audio to enable staff who can’t participate in real time to access the information at their convenience.
The Conference has facilitated networking among VA oncology providers who have common interests. Conference participants also have participated in other VA ECHO conferences in disciplines beyond oncology. Participants in the Conference also are encouraged to participate as speakers by presenting quality improvement initiatives at their VA site. This novel approach to generating content for this educational series has led to a dynamic interchange of ideas and increased networking among VA providers related to their practice and quality improvement initiatives at their VA sites. The Conference provides a regular forum for VA staff across a wide range of disciplines to share personal experiences, successes, and frustrations and to get feedback from colleagues.
The Conference combines a structured approach to presenting VA-specific educational content related to cancer care and multiple mechanisms that encourage staff to participate in an ongoing dialogue related to quality initiatives both on the phone during the Conference, online using Outlook LYNC or Skype to ask questions during the Conference, and during conversations on group e-mail. The Conference promotes staff engagement at little or no extra cost to the VA. For more information about the VA ECHO Cancer Care Conference or to submit a presentation for consideration for a future session, please contact [email protected] or [email protected].
1. Arora S, Geppert CM, Kalishman S, et al. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Acad Med. 2007;82(2):154-160.
2. Hematology and oncology federal health care data trends. Fed Pract. 2017;33(suppl 5):S12-S15.
3. American College of Surgeons Commission on Cancer. Cancer Program Standards: Ensuring Patient Centered Care, 2016 Edition. https://www.facs.org/quality-programs/cancer/coc/standards. Accessed March 14, 2018.
1. Arora S, Geppert CM, Kalishman S, et al. Academic health center management of chronic diseases through knowledge networks: Project ECHO. Acad Med. 2007;82(2):154-160.
2. Hematology and oncology federal health care data trends. Fed Pract. 2017;33(suppl 5):S12-S15.
3. American College of Surgeons Commission on Cancer. Cancer Program Standards: Ensuring Patient Centered Care, 2016 Edition. https://www.facs.org/quality-programs/cancer/coc/standards. Accessed March 14, 2018.
Liver Imaging Reporting and Data System in Patients at High Risk for Hepatocellular Carcinoma in the Memphis Veterans Affairs Population (FULL)
Although hepatocellular carcinoma can be difficult to detect, use of the LI-RADS algorithm could lead to earlier identification in at-risk patients.
Hepatocellular carcinoma (HCC) is the third most common cause of death from cancer worldwide.1 Liver cancer is the fifth most common cancer in men and the seventh in women.2 The highest incidence rates are in sub-Saharan Africa and Southeast Asia where hepatitis B virus is endemic. The incidence of HCC in western countries is increasing, particularly due to the rise of hepatitis C virus (HCV) as well as alcoholic liver disease and nonalcoholic fatty liver disease. The incidence of HCC has tripled in the US in the past 2 decades.1-3
HCC can be diagnosed by radiographic images without the need for biopsy if the typical imaging features are present.3 The European Association for the Study of Liver Disease (EASL) and the American Association for the Study of Liver Diseases (AASLD) recommend screening abdominal ultrasonography at 6-month intervals for high-risk patients.3,4 High-risk patients include patients with cirrhosis, especially those with hepatitis B or C.3
If screening ultrasonography detects a nodule, size determines whether a follow-up ultrasound is needed vs obtaining a contrast-enhanced dynamic computed tomography (CT) scan or a magnetic resonance image (MRI).3 If ultrasonography detects a nodule > 1 cm in diameter, then a dynamic CT or MRI is performed. Characteristic hyperenhancement during later arterial phase and washout during the venous or delayed phase is associated with a nearly 100% specificity for HCC diagnosis.5 Arterial-enhancing contrast is required when using CT and MRI because HCC is a hypervascular lesion.6 The portal venous blood dilutes the majority of the liver’s arterial blood; therefore, the liver does not enhance during the arterial phase, while HCC will show maximum enhancement.7 Furthermore, HCC should demonstrate a “washout” of contrast during the venous phase on CT and MRI.4 Standard imaging protocol dictates that 4 phases are needed to properly diagnose HCC including unenhanced, arterial, venous, and delayed.4
Regular surveillance increases the likelihood of detecting HCC before the presentation of clinical symptoms and facilitates receipt of curative therapy.8-10 Patients with viral hepatitis and cirrhosis with HCC found on screening are more likely to have earlier-stage disease and survive longer from the time of diagnosis.11 Furthermore, it has been observed that HCC detected by surveillance is significantly more likely to undergo curative therapy compared with incidental or symptomatic detection of HCC.9
Technical improvements in imaging techniques include advancement in contrast agents, multidetector row helical CT, and the flexibility/range of pulse sequences available in MRI.7 Even with technical improvements in all modalities used in HCC imaging, detecting HCC remains difficult, especially when detecting the small (< 2 cm) lesions in a cirrhotic liver.7 Interpretation of imaging also remains a challenge as HCC does not always fit strict criteria: lack of “washout” in a hypervascular lesion, determining small HCC lesions from benign nodules, and hypovascular/isovascular HCC.5 Radiologic differentials in the diagnosis of HCC include transient hepatic intensity difference (THID)/transient hepatic attenuation difference (THAD), arterio-portal shunt, and regenerative nodules.12 In the common clinical setting, patients undergo multiple imaging studies that are interpreted by multiple radiologists, which can add to the difficulty in the diagnosis of HCC.13
The radiology community recognized the inconsistencies and complexities of HCC imaging. Therefore, the American College of Radiology endorsed the Liver Imaging Reporting and Data System (LI-RADS), which had the goal of reducing variability in lesion interpretation through standardization and improving communication with clinicians.14 LI-RADS uses a diagnostic algorithm for CT and MRI that categorizes observed liver findings in high-risk individuals based on the probability or relative risk of HCC without assigning a formal diagnosis.14 LI-RADS takes into account arterial phase enhancement, tumor size, washout appearance, the presence and nature of a capsule, and threshold growth.15 LI-RADS categorizes an observed liver finding on a scale of 1 to 5, with 1 corresponding to a definitely benign finding and 5 with definitive HCC.14 Furthermore, LI-RADS sought to limit the technical variabilities among institutions.
LI-RADS was launched in 2011 and has been utilized by many clinical practices while continuing to be expanded and updated.16 Recent studies examined the specificity of LI-RADS as well as interreader variability.17,18 For nodules viewed on MRI, both LI-RADS categories 4 and 5 had high specificity for HCC.17 When looking at interreader repeatability, LI-RADS showed moderate agreement among experts using the diagnostic algorithm.19 Further studies have compared LI-RADS with the AASLD guidelines and the Organ Procurement and Transplantation Network (OPTN) guidelines.16 When compared with other guidelines, LI-RADS expands the definition of indeterminate findings into probably benign, intermediate probability of HCC, and probably HCC, which corresponds to LI-RADS categories 2, 3, and 4.16
We looked retrospectively at a group of patients previously diagnosed with HCC to see whether utilizing the LI-RADS scoring system within our screening system might have allowed an earlier prediction of HCC and a timelier intervention. Prior to this investigation the LI-RADS system was not used for HCC screening at our US Department of Veterans Affairs (VA) facility. We examined screened patients at the Memphis VA Medical Center (MVAMC) in Tennessee who were subsequently diagnosed with HCC to see which LI-RADS category the last surveillance CT prior to diagnosis would fall into, 6 months to a year prior to the diagnosis of HCC. Our control population was a group of patients screened with CT for their liver nodules who were found not to have HCC.
Methods
Patients at MVAMC with cirrhosis and patients with chronic hepatitis B are routinely screened with ultrasound, CT, or MRI in accordance with the AASLD, EASL, and VA guidelines. Of 303 patients with HCV and cirrhosis under care in 2015, 242 (81%) received imaging to screen for HCC according to the VA National Hepatitis C Registry 2015 (Personal Communication, Population Health Service, Office of Patient Care Services).The LI-RADS scoring system was not applied as a standard screening methodology.
Under an institutional review board-approved protocol, we reviewed the charts of all patients diagnosed with HCC at MVAMC from 2009 to 2014, utilizing ICD-9 code of 155.0 for HCC. We identified within these charts patients who had a surveillance CT image performed within a 6- to 13-month period prior to the CTs that diagnosed HCC (prediagnostic HCC CT). Furthermore, we reviewed the charts of all patients diagnosed with benign liver nodules at MVAMC from 2009 to 2014, utilizing the ICD-9 code of 573.8 for other specified disorders of the liver.
Within these charts, we found patients who had a surveillance CT image performed and who were followed after that image with additional imaging for ≥ 2 years or who had a liver biopsy negative for HCC (benign surveillance CT). We compared these 2 sets of CTs utilizing LI-RADS criteria. Once these patients were identified, a list of the CTs to be examined were given to 2 MVAMC radiologists who specialize in CT.
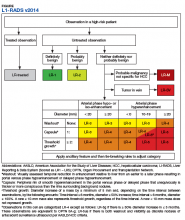
No identifying information of the patients was included, and a 13-digit number unique to each CT exam identified the CTs to be reviewed. Radiologist 1 and 2 examined the CTs on the MVAMC Picture Archiving and Communication System (PACS). Both radiologists were asked to give each nodule a score according to LI-RADS v2014 diagnostic algorithm (Figure).
We hypothesized that the prediagnostic CT images of patients eventually determined to have HCC would have a LI-RADS score of 4 (LR4) or LR5. Furthermore, we hypothesized that the CT images of the benign liver nodule patients would have a score ≤ LR3. If there was a disagreement between the radiologists in terms of a malignant score (LR4 or LR5) vs a benign score (≤ LR3), then a third radiologist (radiologist 3) provided a score for these nodules. The third, tiebreaker radiologist was given the scores of both prior radiologists and asked to choose which score was correct.
Statistical analysis was then applied to the data to determine the sensitivity, specificity, and diagnostic accuracy in diagnosing eventual HCC, as well as the false-negative and false-positive rates of radiologists 1 and 2. Raw data also were used to determine the agreement between raters by calculating the κ statistic with a 95% CI.
Results
A total of 70 nodules were examined by radiologists 1 and 2 with 42 of the nodules in the prediagnostic HCC CTs and 28 of the nodules in the benign surveillance CTs.
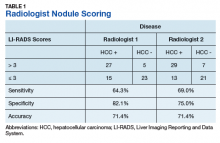
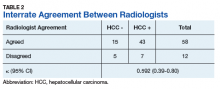
Radiologist 1 identified 11 patients with LR4 and 21 patients with LR5. His scores showed a sensitivity of 64.3% and specificity of 82.1% with accuracy of 71.4% for LI-RADS in identifying eventual HCC. The false-negative rate of the LI-RADS diagnostic algorithm for radiologist 1 was 35.7% and the false-positive rate was 17.9%. Radiologist 2 identified 17 patients LR4 and 19 patients with LR5. Radiologist 2’s scores showed a sensitivity of 69.0% and specificity of 75.0% with accuracy of 71.4% for LI-RADS in identifying eventual HCC.The false-negative rate of the LI-RADS diagnostic algorithm for radiologist 2 was 31.0% and false-positive rate of 25.0%. The κ statistic was calculated to determine the interrater agreement. The radiologists agreed on 58 of 70 samples; 15 without HCC and 43 with HCC. The κ statistic was 0.592, which indicates moderate agreement (Table 2).
Discussion
If HCC is diagnosed late in the disease process based on symptomatology and not on surveillance imaging, the likelihood of receiving early and potential curative therapy greatly declines as was shown in a systemic literature review.9 Surveillance imaging and lesion interpretation by various radiologists has been difficult to standardize as new technologic advances continue to occur in the imaging of HCC.14 LI-RADS was initiated to help standardize CT and MRI interpretation and reporting of hepatic nodules. As a dynamic algorithm, it continues to adjust with new advances in imaging techniques with the most recent updates being made to the algorithm in 2014.14,19 LI-RADS applies to patients at high risk for HCC most often who are already enrolled in a surveillance program.19 The MVAMC has a high incidence of patients with cirrhosis who are at risk for HCC, which is why we chose it as our study population.
LI-RADS can be applied to both MRI and CT imaging. Much of the recent literature have looked at LI-RADS in terms of MRI. A group in China looked at 100 pathologically confirmed patients and assigned a LI-RADS score to the MRI at the time of diagnosis and showed that MRI LI-RADS scoring was highly sensitive and specific in the diagnosis of HCC.20 This study did note a numeric difference in the specificity of LI-RADS algorithm depending on how LR3 scores were viewed. If a LR3 score was considered negative rather than positive for HCC, then the specificity increased by almost 20%.20
Another study looked at patients with liver nodules ≤ 20 mm found on ultrasound and obtained MRIs and biopsies on these patients, assigning the MRI a LI-RADs score.17 Darnell and colleagues found that MRI LR4 and LR5 have a high specificity for HCC. However, 29 of the 42 LR3 lesions examined were found to be HCC.17 Furthermore, Choi and colleagues retrospectively looked at patients in a HCC surveillance program who had undergone MRI as part of the program and assigned LI-RADS scores to these MRIs.21 Their study showed that LR5 criteria on gadoxetate disodium-enhanced MRI has excellent positive predictive value (PPV) for diagnosing HCC, and LR4 showed good PPV.21
In our study, we chose to look at LI-RADS in terms of surveillance CT scans 6 to 13 months prior to the diagnosis of HCC to see whether this method would allow us to intervene earlier with more aggressive diagnostics or therapy in those suspected of having HCC. Although Choi and colleagues looked retrospectively at MRI surveillance imaging, most of the prior studies have looked at LI-RADS scoring in imaging at the time of diagnosis.17,20,21 By looking at surveillance CT scans, we sought to determine LI-RADS sensitivity, specificity, and diagnostic accuracy as a screening tool compared with CT evaluations without LI-RADS scoring.
We also chose to look at CT scans since most of the prior studies have looked at the more detailed and often more expensive MRIs. For both radiologists 1 and 2, the sensitivity was > 60% and specificity was > 70% with a diagnostic accuracy of 71.4% in predicting a diagnosis of HCC in future scans. Although there was high false negative of > 30% for both radiologists, we did consider LR3 as negative for HCC. As Darnell and colleagues’ study of MRI LI-RADS shows, LR3 may need to be revised in the future as its ambiguity can lead to false-negatives.17 Our results also showed moderate interreader agreement, which has been seen in previous studies with LI-RADS.18
Some studies have compared MRI with CT imaging in terms of LI-RADs classification of hepatic nodules to find out whether concordance was seen.22,23 Both studies found that there was substantial discordance between MRI and CT with CT often underscoring hepatic nodules.22,23 In Zhang and colleagues, interclass agreement between CT and MRI varied the most in terms of arterial enhancement with CT producing false-negative findings.22 CT also underestimated LI-RADS score by 16.9% for LR3, 37.3% for LR4, and 8.5% for LR5 in this study.22 Furthermore, Corwin and colleagues found a significant upgrade in terms of LI-RADS categorization with MRI for 42.5% of observations.23 In this study, upgraded LI-RADS scores on MRI included 2 upgraded to LR5V (Figure), 15 upgraded to LR5, and 12 upgraded to LR4.23
Our study shows that the LI-RADS algorithm has a good sensitivity, specificity, and diagnostic accuracy as a screening tool, predicting HCC in scans earlier than standard CT evaluation. In our study, the patients with HCC were shown to have higher LI-RADS scores on prediagnostic imaging, while the benign liver nodule patients were shown to have lower LI-RADS scores. This data would suggest that a LI-RADS score given to surveillance CT of LR4 or higher should recommend either a biopsy or follow-up imaging after a short interval. If LI-RADS is applied to surveillance CTs in patients at risk for HCC, a diagnosis of HCC may be arrived at earlier as compared with not using the LI-RADS algorithm. Earlier detection may lead to earlier intervention and improved treatment outcomes.
Limitations
Limitations to our study occurred because radiologist 3 did not review all of the images nor score them. Radiologist 3 was limited to 12 images where there was disagreement and was limited to 2 scores to choose from for each image. Further limitations include that this study was performed at a single center. Our study focused on one imaging modality and did not include ultrasounds or MRIs. We did not compare the demographics of our patients with those of other VA hospitals. The radiologists interpreted the images individually, and their subjectivity was another limitation.
Conclusion
In the MVAMC population, LI-RADS showed a good sensitivity, specificity, and diagnostic accuracy for CT surveillance scans in patient at high risk for HCC at an earlier time point than did standard evaluation by very experienced CT radiologists. Higher LI-RADS scores on surveillance CTs had good diagnostic accuracy for the probable future diagnosis of HCC, whereas lower LI-RADS scores had a good diagnostic accuracy for probable benign nodules. Utilizing the LI-RADS algorithm on all surveillance CTs in patients at high risk for HCC may lead to obtaining MRIs or follow-up CTs sooner for suspicious nodules, leading to an earlier diagnosis of HCC and possible earlier and more effective intervention.
1. El–Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557-2576.
2. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-1127.
3. Bruix J, Sherman M; American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53(3):1020-1022.
4. Selvapatt N, House H, Brown A. Hepatocellular carcinoma surveillance: are we utilizing it? J Clin Gastroenterol. 2016;50(1):e8-e12.
5. Lee JM, Yoon JH, Joo I, Woo HS. Recent advances in CT and MR imaging for evaluation of hepatocellular carcinoma. Liver Cancer. 2012;1(1):22-40.
6. Chou R, Cuevas C, Fu R, et al. Imaging techniques for the diagnosis of hepatocellular carcinoma: a systemic review and meta-analysis. Ann Intern Med. 2015;162(10):697-711.
7. Ariff B, Lloyd CR, Khan S, et al. Imaging of liver cancer. World J Gastroenterol. 2009;15(11):1289-1300.
8. Yuen MF, Cheng CC, Lauder IJ, Lam SK, Ooi CG, Lai CL. Early detection of hepatocellular carcinoma increases the chance of treatment: Hong Kong experience. Hepatology. 2000;31(2):330-335.
9. Singal AG, Pillai A, Tiro J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS Med. 2014;11(4):e1001624.
10. Nusbaum, JD, Smirniotopoulos J, Wright HC, et al. The effect of hepatocellular carcinoma surveillance in an urban population with liver cirrhosis. J Clin Gastroenterol. 2015;49(10):e91-e95.
11. Kansagara D, Papak J, Pasha AS, et al. Screening for hepatocellular carcinoma in chronic liver disease: a systemic review. Ann Intern Med. 2014;161(4):261-269.
12. Shah S, Shukla A, Paunipagar B. Radiological features of hepatocellular carcinoma. J Clin Exp Hepatol. 2014;4(suppl 3):S63-S66.
13. You MW, Kim SY, Kim KW, et al. Recent advances in the imaging of hepatocellular carcinoma. Clin Mol Hepatol. 2015;21(1):95-103.
14. American College of Radiology. Liver reporting and data system (LI-RADS). https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS. Accessed April 10, 2018.
15. Anis M. Imaging of hepatocellular carcinoma: new approaches to diagnosis. Clin Liver Dis. 2015;19(2):325-340.
16. Mitchell D, Bruix J, Sherman M, Sirlin CB. LI-RADS (Liver Imaging Reporting and Data System): summary, discussion, and consensus of the LI-RADS Management Working Group and future directions. Hepatology. 2015;61(3):1056-1065.
17. Darnell A, Forner A, Rimola J, et al. Liver imaging reporting and data system with MR imaging: evaluation in nodules 20 mm or smaller detected in cirrhosis at screening US. Radiology. 2015; 275(3):698-707.
18. Davenport MS, Khalatbari S, Liu PS, et al. Repeatability of diagnostic features and scoring systems for hepatocellular carcinoma by using MR imaging. Radiology. 2014;272(1):132-142.
19. An C, Rakhmonova G, Choi JY, Kim MJ. Liver imaging reporting and data system (LI-RADS) version 2014: understanding and application of the diagnostic algorithm. Clin Mol Hepatol. 2016;22(2):296-307.
20. Zhao W, Li W, Yi X, et al. [Diagnostic value of liver imaging reporting and data system on primary hepatocellular carcinoma] [in Chinese]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016;41(4):380-387.
21. Choi SH, Byun JH, Kim SY, et al. Liver imaging reporting and data system v2014 with gadoxetate disodium-enhanced magnetic resonance imaging: validation of LIRADS category 4 and 5 criteria. Invest Radiol. 2016;51(8):483-490.
22. Zhang YD, Zhu FP, Xu X, et al. Liver imaging reporting and data system: substantial discordance between CT and MR for imaging classification of hepatic nodules. Acad Radiol. 2016;23(3):344-352.
23. Corwin MT, Fananapazir G, Jin M, Lamba R, Bashir MR. Difference in liver imaging and reporting data system categorization between MRI and CT. Am J Roentgenol. 2016;206(2):307-312.
Although hepatocellular carcinoma can be difficult to detect, use of the LI-RADS algorithm could lead to earlier identification in at-risk patients.
Although hepatocellular carcinoma can be difficult to detect, use of the LI-RADS algorithm could lead to earlier identification in at-risk patients.
Hepatocellular carcinoma (HCC) is the third most common cause of death from cancer worldwide.1 Liver cancer is the fifth most common cancer in men and the seventh in women.2 The highest incidence rates are in sub-Saharan Africa and Southeast Asia where hepatitis B virus is endemic. The incidence of HCC in western countries is increasing, particularly due to the rise of hepatitis C virus (HCV) as well as alcoholic liver disease and nonalcoholic fatty liver disease. The incidence of HCC has tripled in the US in the past 2 decades.1-3
HCC can be diagnosed by radiographic images without the need for biopsy if the typical imaging features are present.3 The European Association for the Study of Liver Disease (EASL) and the American Association for the Study of Liver Diseases (AASLD) recommend screening abdominal ultrasonography at 6-month intervals for high-risk patients.3,4 High-risk patients include patients with cirrhosis, especially those with hepatitis B or C.3
If screening ultrasonography detects a nodule, size determines whether a follow-up ultrasound is needed vs obtaining a contrast-enhanced dynamic computed tomography (CT) scan or a magnetic resonance image (MRI).3 If ultrasonography detects a nodule > 1 cm in diameter, then a dynamic CT or MRI is performed. Characteristic hyperenhancement during later arterial phase and washout during the venous or delayed phase is associated with a nearly 100% specificity for HCC diagnosis.5 Arterial-enhancing contrast is required when using CT and MRI because HCC is a hypervascular lesion.6 The portal venous blood dilutes the majority of the liver’s arterial blood; therefore, the liver does not enhance during the arterial phase, while HCC will show maximum enhancement.7 Furthermore, HCC should demonstrate a “washout” of contrast during the venous phase on CT and MRI.4 Standard imaging protocol dictates that 4 phases are needed to properly diagnose HCC including unenhanced, arterial, venous, and delayed.4
Regular surveillance increases the likelihood of detecting HCC before the presentation of clinical symptoms and facilitates receipt of curative therapy.8-10 Patients with viral hepatitis and cirrhosis with HCC found on screening are more likely to have earlier-stage disease and survive longer from the time of diagnosis.11 Furthermore, it has been observed that HCC detected by surveillance is significantly more likely to undergo curative therapy compared with incidental or symptomatic detection of HCC.9
Technical improvements in imaging techniques include advancement in contrast agents, multidetector row helical CT, and the flexibility/range of pulse sequences available in MRI.7 Even with technical improvements in all modalities used in HCC imaging, detecting HCC remains difficult, especially when detecting the small (< 2 cm) lesions in a cirrhotic liver.7 Interpretation of imaging also remains a challenge as HCC does not always fit strict criteria: lack of “washout” in a hypervascular lesion, determining small HCC lesions from benign nodules, and hypovascular/isovascular HCC.5 Radiologic differentials in the diagnosis of HCC include transient hepatic intensity difference (THID)/transient hepatic attenuation difference (THAD), arterio-portal shunt, and regenerative nodules.12 In the common clinical setting, patients undergo multiple imaging studies that are interpreted by multiple radiologists, which can add to the difficulty in the diagnosis of HCC.13
The radiology community recognized the inconsistencies and complexities of HCC imaging. Therefore, the American College of Radiology endorsed the Liver Imaging Reporting and Data System (LI-RADS), which had the goal of reducing variability in lesion interpretation through standardization and improving communication with clinicians.14 LI-RADS uses a diagnostic algorithm for CT and MRI that categorizes observed liver findings in high-risk individuals based on the probability or relative risk of HCC without assigning a formal diagnosis.14 LI-RADS takes into account arterial phase enhancement, tumor size, washout appearance, the presence and nature of a capsule, and threshold growth.15 LI-RADS categorizes an observed liver finding on a scale of 1 to 5, with 1 corresponding to a definitely benign finding and 5 with definitive HCC.14 Furthermore, LI-RADS sought to limit the technical variabilities among institutions.
LI-RADS was launched in 2011 and has been utilized by many clinical practices while continuing to be expanded and updated.16 Recent studies examined the specificity of LI-RADS as well as interreader variability.17,18 For nodules viewed on MRI, both LI-RADS categories 4 and 5 had high specificity for HCC.17 When looking at interreader repeatability, LI-RADS showed moderate agreement among experts using the diagnostic algorithm.19 Further studies have compared LI-RADS with the AASLD guidelines and the Organ Procurement and Transplantation Network (OPTN) guidelines.16 When compared with other guidelines, LI-RADS expands the definition of indeterminate findings into probably benign, intermediate probability of HCC, and probably HCC, which corresponds to LI-RADS categories 2, 3, and 4.16
We looked retrospectively at a group of patients previously diagnosed with HCC to see whether utilizing the LI-RADS scoring system within our screening system might have allowed an earlier prediction of HCC and a timelier intervention. Prior to this investigation the LI-RADS system was not used for HCC screening at our US Department of Veterans Affairs (VA) facility. We examined screened patients at the Memphis VA Medical Center (MVAMC) in Tennessee who were subsequently diagnosed with HCC to see which LI-RADS category the last surveillance CT prior to diagnosis would fall into, 6 months to a year prior to the diagnosis of HCC. Our control population was a group of patients screened with CT for their liver nodules who were found not to have HCC.
Methods
Patients at MVAMC with cirrhosis and patients with chronic hepatitis B are routinely screened with ultrasound, CT, or MRI in accordance with the AASLD, EASL, and VA guidelines. Of 303 patients with HCV and cirrhosis under care in 2015, 242 (81%) received imaging to screen for HCC according to the VA National Hepatitis C Registry 2015 (Personal Communication, Population Health Service, Office of Patient Care Services).The LI-RADS scoring system was not applied as a standard screening methodology.
Under an institutional review board-approved protocol, we reviewed the charts of all patients diagnosed with HCC at MVAMC from 2009 to 2014, utilizing ICD-9 code of 155.0 for HCC. We identified within these charts patients who had a surveillance CT image performed within a 6- to 13-month period prior to the CTs that diagnosed HCC (prediagnostic HCC CT). Furthermore, we reviewed the charts of all patients diagnosed with benign liver nodules at MVAMC from 2009 to 2014, utilizing the ICD-9 code of 573.8 for other specified disorders of the liver.
Within these charts, we found patients who had a surveillance CT image performed and who were followed after that image with additional imaging for ≥ 2 years or who had a liver biopsy negative for HCC (benign surveillance CT). We compared these 2 sets of CTs utilizing LI-RADS criteria. Once these patients were identified, a list of the CTs to be examined were given to 2 MVAMC radiologists who specialize in CT.
No identifying information of the patients was included, and a 13-digit number unique to each CT exam identified the CTs to be reviewed. Radiologist 1 and 2 examined the CTs on the MVAMC Picture Archiving and Communication System (PACS). Both radiologists were asked to give each nodule a score according to LI-RADS v2014 diagnostic algorithm (Figure).
We hypothesized that the prediagnostic CT images of patients eventually determined to have HCC would have a LI-RADS score of 4 (LR4) or LR5. Furthermore, we hypothesized that the CT images of the benign liver nodule patients would have a score ≤ LR3. If there was a disagreement between the radiologists in terms of a malignant score (LR4 or LR5) vs a benign score (≤ LR3), then a third radiologist (radiologist 3) provided a score for these nodules. The third, tiebreaker radiologist was given the scores of both prior radiologists and asked to choose which score was correct.
Statistical analysis was then applied to the data to determine the sensitivity, specificity, and diagnostic accuracy in diagnosing eventual HCC, as well as the false-negative and false-positive rates of radiologists 1 and 2. Raw data also were used to determine the agreement between raters by calculating the κ statistic with a 95% CI.
Results
A total of 70 nodules were examined by radiologists 1 and 2 with 42 of the nodules in the prediagnostic HCC CTs and 28 of the nodules in the benign surveillance CTs.
Radiologist 1 identified 11 patients with LR4 and 21 patients with LR5. His scores showed a sensitivity of 64.3% and specificity of 82.1% with accuracy of 71.4% for LI-RADS in identifying eventual HCC. The false-negative rate of the LI-RADS diagnostic algorithm for radiologist 1 was 35.7% and the false-positive rate was 17.9%. Radiologist 2 identified 17 patients LR4 and 19 patients with LR5. Radiologist 2’s scores showed a sensitivity of 69.0% and specificity of 75.0% with accuracy of 71.4% for LI-RADS in identifying eventual HCC.The false-negative rate of the LI-RADS diagnostic algorithm for radiologist 2 was 31.0% and false-positive rate of 25.0%. The κ statistic was calculated to determine the interrater agreement. The radiologists agreed on 58 of 70 samples; 15 without HCC and 43 with HCC. The κ statistic was 0.592, which indicates moderate agreement (Table 2).
Discussion
If HCC is diagnosed late in the disease process based on symptomatology and not on surveillance imaging, the likelihood of receiving early and potential curative therapy greatly declines as was shown in a systemic literature review.9 Surveillance imaging and lesion interpretation by various radiologists has been difficult to standardize as new technologic advances continue to occur in the imaging of HCC.14 LI-RADS was initiated to help standardize CT and MRI interpretation and reporting of hepatic nodules. As a dynamic algorithm, it continues to adjust with new advances in imaging techniques with the most recent updates being made to the algorithm in 2014.14,19 LI-RADS applies to patients at high risk for HCC most often who are already enrolled in a surveillance program.19 The MVAMC has a high incidence of patients with cirrhosis who are at risk for HCC, which is why we chose it as our study population.
LI-RADS can be applied to both MRI and CT imaging. Much of the recent literature have looked at LI-RADS in terms of MRI. A group in China looked at 100 pathologically confirmed patients and assigned a LI-RADS score to the MRI at the time of diagnosis and showed that MRI LI-RADS scoring was highly sensitive and specific in the diagnosis of HCC.20 This study did note a numeric difference in the specificity of LI-RADS algorithm depending on how LR3 scores were viewed. If a LR3 score was considered negative rather than positive for HCC, then the specificity increased by almost 20%.20
Another study looked at patients with liver nodules ≤ 20 mm found on ultrasound and obtained MRIs and biopsies on these patients, assigning the MRI a LI-RADs score.17 Darnell and colleagues found that MRI LR4 and LR5 have a high specificity for HCC. However, 29 of the 42 LR3 lesions examined were found to be HCC.17 Furthermore, Choi and colleagues retrospectively looked at patients in a HCC surveillance program who had undergone MRI as part of the program and assigned LI-RADS scores to these MRIs.21 Their study showed that LR5 criteria on gadoxetate disodium-enhanced MRI has excellent positive predictive value (PPV) for diagnosing HCC, and LR4 showed good PPV.21
In our study, we chose to look at LI-RADS in terms of surveillance CT scans 6 to 13 months prior to the diagnosis of HCC to see whether this method would allow us to intervene earlier with more aggressive diagnostics or therapy in those suspected of having HCC. Although Choi and colleagues looked retrospectively at MRI surveillance imaging, most of the prior studies have looked at LI-RADS scoring in imaging at the time of diagnosis.17,20,21 By looking at surveillance CT scans, we sought to determine LI-RADS sensitivity, specificity, and diagnostic accuracy as a screening tool compared with CT evaluations without LI-RADS scoring.
We also chose to look at CT scans since most of the prior studies have looked at the more detailed and often more expensive MRIs. For both radiologists 1 and 2, the sensitivity was > 60% and specificity was > 70% with a diagnostic accuracy of 71.4% in predicting a diagnosis of HCC in future scans. Although there was high false negative of > 30% for both radiologists, we did consider LR3 as negative for HCC. As Darnell and colleagues’ study of MRI LI-RADS shows, LR3 may need to be revised in the future as its ambiguity can lead to false-negatives.17 Our results also showed moderate interreader agreement, which has been seen in previous studies with LI-RADS.18
Some studies have compared MRI with CT imaging in terms of LI-RADs classification of hepatic nodules to find out whether concordance was seen.22,23 Both studies found that there was substantial discordance between MRI and CT with CT often underscoring hepatic nodules.22,23 In Zhang and colleagues, interclass agreement between CT and MRI varied the most in terms of arterial enhancement with CT producing false-negative findings.22 CT also underestimated LI-RADS score by 16.9% for LR3, 37.3% for LR4, and 8.5% for LR5 in this study.22 Furthermore, Corwin and colleagues found a significant upgrade in terms of LI-RADS categorization with MRI for 42.5% of observations.23 In this study, upgraded LI-RADS scores on MRI included 2 upgraded to LR5V (Figure), 15 upgraded to LR5, and 12 upgraded to LR4.23
Our study shows that the LI-RADS algorithm has a good sensitivity, specificity, and diagnostic accuracy as a screening tool, predicting HCC in scans earlier than standard CT evaluation. In our study, the patients with HCC were shown to have higher LI-RADS scores on prediagnostic imaging, while the benign liver nodule patients were shown to have lower LI-RADS scores. This data would suggest that a LI-RADS score given to surveillance CT of LR4 or higher should recommend either a biopsy or follow-up imaging after a short interval. If LI-RADS is applied to surveillance CTs in patients at risk for HCC, a diagnosis of HCC may be arrived at earlier as compared with not using the LI-RADS algorithm. Earlier detection may lead to earlier intervention and improved treatment outcomes.
Limitations
Limitations to our study occurred because radiologist 3 did not review all of the images nor score them. Radiologist 3 was limited to 12 images where there was disagreement and was limited to 2 scores to choose from for each image. Further limitations include that this study was performed at a single center. Our study focused on one imaging modality and did not include ultrasounds or MRIs. We did not compare the demographics of our patients with those of other VA hospitals. The radiologists interpreted the images individually, and their subjectivity was another limitation.
Conclusion
In the MVAMC population, LI-RADS showed a good sensitivity, specificity, and diagnostic accuracy for CT surveillance scans in patient at high risk for HCC at an earlier time point than did standard evaluation by very experienced CT radiologists. Higher LI-RADS scores on surveillance CTs had good diagnostic accuracy for the probable future diagnosis of HCC, whereas lower LI-RADS scores had a good diagnostic accuracy for probable benign nodules. Utilizing the LI-RADS algorithm on all surveillance CTs in patients at high risk for HCC may lead to obtaining MRIs or follow-up CTs sooner for suspicious nodules, leading to an earlier diagnosis of HCC and possible earlier and more effective intervention.
Hepatocellular carcinoma (HCC) is the third most common cause of death from cancer worldwide.1 Liver cancer is the fifth most common cancer in men and the seventh in women.2 The highest incidence rates are in sub-Saharan Africa and Southeast Asia where hepatitis B virus is endemic. The incidence of HCC in western countries is increasing, particularly due to the rise of hepatitis C virus (HCV) as well as alcoholic liver disease and nonalcoholic fatty liver disease. The incidence of HCC has tripled in the US in the past 2 decades.1-3
HCC can be diagnosed by radiographic images without the need for biopsy if the typical imaging features are present.3 The European Association for the Study of Liver Disease (EASL) and the American Association for the Study of Liver Diseases (AASLD) recommend screening abdominal ultrasonography at 6-month intervals for high-risk patients.3,4 High-risk patients include patients with cirrhosis, especially those with hepatitis B or C.3
If screening ultrasonography detects a nodule, size determines whether a follow-up ultrasound is needed vs obtaining a contrast-enhanced dynamic computed tomography (CT) scan or a magnetic resonance image (MRI).3 If ultrasonography detects a nodule > 1 cm in diameter, then a dynamic CT or MRI is performed. Characteristic hyperenhancement during later arterial phase and washout during the venous or delayed phase is associated with a nearly 100% specificity for HCC diagnosis.5 Arterial-enhancing contrast is required when using CT and MRI because HCC is a hypervascular lesion.6 The portal venous blood dilutes the majority of the liver’s arterial blood; therefore, the liver does not enhance during the arterial phase, while HCC will show maximum enhancement.7 Furthermore, HCC should demonstrate a “washout” of contrast during the venous phase on CT and MRI.4 Standard imaging protocol dictates that 4 phases are needed to properly diagnose HCC including unenhanced, arterial, venous, and delayed.4
Regular surveillance increases the likelihood of detecting HCC before the presentation of clinical symptoms and facilitates receipt of curative therapy.8-10 Patients with viral hepatitis and cirrhosis with HCC found on screening are more likely to have earlier-stage disease and survive longer from the time of diagnosis.11 Furthermore, it has been observed that HCC detected by surveillance is significantly more likely to undergo curative therapy compared with incidental or symptomatic detection of HCC.9
Technical improvements in imaging techniques include advancement in contrast agents, multidetector row helical CT, and the flexibility/range of pulse sequences available in MRI.7 Even with technical improvements in all modalities used in HCC imaging, detecting HCC remains difficult, especially when detecting the small (< 2 cm) lesions in a cirrhotic liver.7 Interpretation of imaging also remains a challenge as HCC does not always fit strict criteria: lack of “washout” in a hypervascular lesion, determining small HCC lesions from benign nodules, and hypovascular/isovascular HCC.5 Radiologic differentials in the diagnosis of HCC include transient hepatic intensity difference (THID)/transient hepatic attenuation difference (THAD), arterio-portal shunt, and regenerative nodules.12 In the common clinical setting, patients undergo multiple imaging studies that are interpreted by multiple radiologists, which can add to the difficulty in the diagnosis of HCC.13
The radiology community recognized the inconsistencies and complexities of HCC imaging. Therefore, the American College of Radiology endorsed the Liver Imaging Reporting and Data System (LI-RADS), which had the goal of reducing variability in lesion interpretation through standardization and improving communication with clinicians.14 LI-RADS uses a diagnostic algorithm for CT and MRI that categorizes observed liver findings in high-risk individuals based on the probability or relative risk of HCC without assigning a formal diagnosis.14 LI-RADS takes into account arterial phase enhancement, tumor size, washout appearance, the presence and nature of a capsule, and threshold growth.15 LI-RADS categorizes an observed liver finding on a scale of 1 to 5, with 1 corresponding to a definitely benign finding and 5 with definitive HCC.14 Furthermore, LI-RADS sought to limit the technical variabilities among institutions.
LI-RADS was launched in 2011 and has been utilized by many clinical practices while continuing to be expanded and updated.16 Recent studies examined the specificity of LI-RADS as well as interreader variability.17,18 For nodules viewed on MRI, both LI-RADS categories 4 and 5 had high specificity for HCC.17 When looking at interreader repeatability, LI-RADS showed moderate agreement among experts using the diagnostic algorithm.19 Further studies have compared LI-RADS with the AASLD guidelines and the Organ Procurement and Transplantation Network (OPTN) guidelines.16 When compared with other guidelines, LI-RADS expands the definition of indeterminate findings into probably benign, intermediate probability of HCC, and probably HCC, which corresponds to LI-RADS categories 2, 3, and 4.16
We looked retrospectively at a group of patients previously diagnosed with HCC to see whether utilizing the LI-RADS scoring system within our screening system might have allowed an earlier prediction of HCC and a timelier intervention. Prior to this investigation the LI-RADS system was not used for HCC screening at our US Department of Veterans Affairs (VA) facility. We examined screened patients at the Memphis VA Medical Center (MVAMC) in Tennessee who were subsequently diagnosed with HCC to see which LI-RADS category the last surveillance CT prior to diagnosis would fall into, 6 months to a year prior to the diagnosis of HCC. Our control population was a group of patients screened with CT for their liver nodules who were found not to have HCC.
Methods
Patients at MVAMC with cirrhosis and patients with chronic hepatitis B are routinely screened with ultrasound, CT, or MRI in accordance with the AASLD, EASL, and VA guidelines. Of 303 patients with HCV and cirrhosis under care in 2015, 242 (81%) received imaging to screen for HCC according to the VA National Hepatitis C Registry 2015 (Personal Communication, Population Health Service, Office of Patient Care Services).The LI-RADS scoring system was not applied as a standard screening methodology.
Under an institutional review board-approved protocol, we reviewed the charts of all patients diagnosed with HCC at MVAMC from 2009 to 2014, utilizing ICD-9 code of 155.0 for HCC. We identified within these charts patients who had a surveillance CT image performed within a 6- to 13-month period prior to the CTs that diagnosed HCC (prediagnostic HCC CT). Furthermore, we reviewed the charts of all patients diagnosed with benign liver nodules at MVAMC from 2009 to 2014, utilizing the ICD-9 code of 573.8 for other specified disorders of the liver.
Within these charts, we found patients who had a surveillance CT image performed and who were followed after that image with additional imaging for ≥ 2 years or who had a liver biopsy negative for HCC (benign surveillance CT). We compared these 2 sets of CTs utilizing LI-RADS criteria. Once these patients were identified, a list of the CTs to be examined were given to 2 MVAMC radiologists who specialize in CT.
No identifying information of the patients was included, and a 13-digit number unique to each CT exam identified the CTs to be reviewed. Radiologist 1 and 2 examined the CTs on the MVAMC Picture Archiving and Communication System (PACS). Both radiologists were asked to give each nodule a score according to LI-RADS v2014 diagnostic algorithm (Figure).
We hypothesized that the prediagnostic CT images of patients eventually determined to have HCC would have a LI-RADS score of 4 (LR4) or LR5. Furthermore, we hypothesized that the CT images of the benign liver nodule patients would have a score ≤ LR3. If there was a disagreement between the radiologists in terms of a malignant score (LR4 or LR5) vs a benign score (≤ LR3), then a third radiologist (radiologist 3) provided a score for these nodules. The third, tiebreaker radiologist was given the scores of both prior radiologists and asked to choose which score was correct.
Statistical analysis was then applied to the data to determine the sensitivity, specificity, and diagnostic accuracy in diagnosing eventual HCC, as well as the false-negative and false-positive rates of radiologists 1 and 2. Raw data also were used to determine the agreement between raters by calculating the κ statistic with a 95% CI.
Results
A total of 70 nodules were examined by radiologists 1 and 2 with 42 of the nodules in the prediagnostic HCC CTs and 28 of the nodules in the benign surveillance CTs.
Radiologist 1 identified 11 patients with LR4 and 21 patients with LR5. His scores showed a sensitivity of 64.3% and specificity of 82.1% with accuracy of 71.4% for LI-RADS in identifying eventual HCC. The false-negative rate of the LI-RADS diagnostic algorithm for radiologist 1 was 35.7% and the false-positive rate was 17.9%. Radiologist 2 identified 17 patients LR4 and 19 patients with LR5. Radiologist 2’s scores showed a sensitivity of 69.0% and specificity of 75.0% with accuracy of 71.4% for LI-RADS in identifying eventual HCC.The false-negative rate of the LI-RADS diagnostic algorithm for radiologist 2 was 31.0% and false-positive rate of 25.0%. The κ statistic was calculated to determine the interrater agreement. The radiologists agreed on 58 of 70 samples; 15 without HCC and 43 with HCC. The κ statistic was 0.592, which indicates moderate agreement (Table 2).
Discussion
If HCC is diagnosed late in the disease process based on symptomatology and not on surveillance imaging, the likelihood of receiving early and potential curative therapy greatly declines as was shown in a systemic literature review.9 Surveillance imaging and lesion interpretation by various radiologists has been difficult to standardize as new technologic advances continue to occur in the imaging of HCC.14 LI-RADS was initiated to help standardize CT and MRI interpretation and reporting of hepatic nodules. As a dynamic algorithm, it continues to adjust with new advances in imaging techniques with the most recent updates being made to the algorithm in 2014.14,19 LI-RADS applies to patients at high risk for HCC most often who are already enrolled in a surveillance program.19 The MVAMC has a high incidence of patients with cirrhosis who are at risk for HCC, which is why we chose it as our study population.
LI-RADS can be applied to both MRI and CT imaging. Much of the recent literature have looked at LI-RADS in terms of MRI. A group in China looked at 100 pathologically confirmed patients and assigned a LI-RADS score to the MRI at the time of diagnosis and showed that MRI LI-RADS scoring was highly sensitive and specific in the diagnosis of HCC.20 This study did note a numeric difference in the specificity of LI-RADS algorithm depending on how LR3 scores were viewed. If a LR3 score was considered negative rather than positive for HCC, then the specificity increased by almost 20%.20
Another study looked at patients with liver nodules ≤ 20 mm found on ultrasound and obtained MRIs and biopsies on these patients, assigning the MRI a LI-RADs score.17 Darnell and colleagues found that MRI LR4 and LR5 have a high specificity for HCC. However, 29 of the 42 LR3 lesions examined were found to be HCC.17 Furthermore, Choi and colleagues retrospectively looked at patients in a HCC surveillance program who had undergone MRI as part of the program and assigned LI-RADS scores to these MRIs.21 Their study showed that LR5 criteria on gadoxetate disodium-enhanced MRI has excellent positive predictive value (PPV) for diagnosing HCC, and LR4 showed good PPV.21
In our study, we chose to look at LI-RADS in terms of surveillance CT scans 6 to 13 months prior to the diagnosis of HCC to see whether this method would allow us to intervene earlier with more aggressive diagnostics or therapy in those suspected of having HCC. Although Choi and colleagues looked retrospectively at MRI surveillance imaging, most of the prior studies have looked at LI-RADS scoring in imaging at the time of diagnosis.17,20,21 By looking at surveillance CT scans, we sought to determine LI-RADS sensitivity, specificity, and diagnostic accuracy as a screening tool compared with CT evaluations without LI-RADS scoring.
We also chose to look at CT scans since most of the prior studies have looked at the more detailed and often more expensive MRIs. For both radiologists 1 and 2, the sensitivity was > 60% and specificity was > 70% with a diagnostic accuracy of 71.4% in predicting a diagnosis of HCC in future scans. Although there was high false negative of > 30% for both radiologists, we did consider LR3 as negative for HCC. As Darnell and colleagues’ study of MRI LI-RADS shows, LR3 may need to be revised in the future as its ambiguity can lead to false-negatives.17 Our results also showed moderate interreader agreement, which has been seen in previous studies with LI-RADS.18
Some studies have compared MRI with CT imaging in terms of LI-RADs classification of hepatic nodules to find out whether concordance was seen.22,23 Both studies found that there was substantial discordance between MRI and CT with CT often underscoring hepatic nodules.22,23 In Zhang and colleagues, interclass agreement between CT and MRI varied the most in terms of arterial enhancement with CT producing false-negative findings.22 CT also underestimated LI-RADS score by 16.9% for LR3, 37.3% for LR4, and 8.5% for LR5 in this study.22 Furthermore, Corwin and colleagues found a significant upgrade in terms of LI-RADS categorization with MRI for 42.5% of observations.23 In this study, upgraded LI-RADS scores on MRI included 2 upgraded to LR5V (Figure), 15 upgraded to LR5, and 12 upgraded to LR4.23
Our study shows that the LI-RADS algorithm has a good sensitivity, specificity, and diagnostic accuracy as a screening tool, predicting HCC in scans earlier than standard CT evaluation. In our study, the patients with HCC were shown to have higher LI-RADS scores on prediagnostic imaging, while the benign liver nodule patients were shown to have lower LI-RADS scores. This data would suggest that a LI-RADS score given to surveillance CT of LR4 or higher should recommend either a biopsy or follow-up imaging after a short interval. If LI-RADS is applied to surveillance CTs in patients at risk for HCC, a diagnosis of HCC may be arrived at earlier as compared with not using the LI-RADS algorithm. Earlier detection may lead to earlier intervention and improved treatment outcomes.
Limitations
Limitations to our study occurred because radiologist 3 did not review all of the images nor score them. Radiologist 3 was limited to 12 images where there was disagreement and was limited to 2 scores to choose from for each image. Further limitations include that this study was performed at a single center. Our study focused on one imaging modality and did not include ultrasounds or MRIs. We did not compare the demographics of our patients with those of other VA hospitals. The radiologists interpreted the images individually, and their subjectivity was another limitation.
Conclusion
In the MVAMC population, LI-RADS showed a good sensitivity, specificity, and diagnostic accuracy for CT surveillance scans in patient at high risk for HCC at an earlier time point than did standard evaluation by very experienced CT radiologists. Higher LI-RADS scores on surveillance CTs had good diagnostic accuracy for the probable future diagnosis of HCC, whereas lower LI-RADS scores had a good diagnostic accuracy for probable benign nodules. Utilizing the LI-RADS algorithm on all surveillance CTs in patients at high risk for HCC may lead to obtaining MRIs or follow-up CTs sooner for suspicious nodules, leading to an earlier diagnosis of HCC and possible earlier and more effective intervention.
1. El–Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557-2576.
2. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-1127.
3. Bruix J, Sherman M; American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53(3):1020-1022.
4. Selvapatt N, House H, Brown A. Hepatocellular carcinoma surveillance: are we utilizing it? J Clin Gastroenterol. 2016;50(1):e8-e12.
5. Lee JM, Yoon JH, Joo I, Woo HS. Recent advances in CT and MR imaging for evaluation of hepatocellular carcinoma. Liver Cancer. 2012;1(1):22-40.
6. Chou R, Cuevas C, Fu R, et al. Imaging techniques for the diagnosis of hepatocellular carcinoma: a systemic review and meta-analysis. Ann Intern Med. 2015;162(10):697-711.
7. Ariff B, Lloyd CR, Khan S, et al. Imaging of liver cancer. World J Gastroenterol. 2009;15(11):1289-1300.
8. Yuen MF, Cheng CC, Lauder IJ, Lam SK, Ooi CG, Lai CL. Early detection of hepatocellular carcinoma increases the chance of treatment: Hong Kong experience. Hepatology. 2000;31(2):330-335.
9. Singal AG, Pillai A, Tiro J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS Med. 2014;11(4):e1001624.
10. Nusbaum, JD, Smirniotopoulos J, Wright HC, et al. The effect of hepatocellular carcinoma surveillance in an urban population with liver cirrhosis. J Clin Gastroenterol. 2015;49(10):e91-e95.
11. Kansagara D, Papak J, Pasha AS, et al. Screening for hepatocellular carcinoma in chronic liver disease: a systemic review. Ann Intern Med. 2014;161(4):261-269.
12. Shah S, Shukla A, Paunipagar B. Radiological features of hepatocellular carcinoma. J Clin Exp Hepatol. 2014;4(suppl 3):S63-S66.
13. You MW, Kim SY, Kim KW, et al. Recent advances in the imaging of hepatocellular carcinoma. Clin Mol Hepatol. 2015;21(1):95-103.
14. American College of Radiology. Liver reporting and data system (LI-RADS). https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS. Accessed April 10, 2018.
15. Anis M. Imaging of hepatocellular carcinoma: new approaches to diagnosis. Clin Liver Dis. 2015;19(2):325-340.
16. Mitchell D, Bruix J, Sherman M, Sirlin CB. LI-RADS (Liver Imaging Reporting and Data System): summary, discussion, and consensus of the LI-RADS Management Working Group and future directions. Hepatology. 2015;61(3):1056-1065.
17. Darnell A, Forner A, Rimola J, et al. Liver imaging reporting and data system with MR imaging: evaluation in nodules 20 mm or smaller detected in cirrhosis at screening US. Radiology. 2015; 275(3):698-707.
18. Davenport MS, Khalatbari S, Liu PS, et al. Repeatability of diagnostic features and scoring systems for hepatocellular carcinoma by using MR imaging. Radiology. 2014;272(1):132-142.
19. An C, Rakhmonova G, Choi JY, Kim MJ. Liver imaging reporting and data system (LI-RADS) version 2014: understanding and application of the diagnostic algorithm. Clin Mol Hepatol. 2016;22(2):296-307.
20. Zhao W, Li W, Yi X, et al. [Diagnostic value of liver imaging reporting and data system on primary hepatocellular carcinoma] [in Chinese]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016;41(4):380-387.
21. Choi SH, Byun JH, Kim SY, et al. Liver imaging reporting and data system v2014 with gadoxetate disodium-enhanced magnetic resonance imaging: validation of LIRADS category 4 and 5 criteria. Invest Radiol. 2016;51(8):483-490.
22. Zhang YD, Zhu FP, Xu X, et al. Liver imaging reporting and data system: substantial discordance between CT and MR for imaging classification of hepatic nodules. Acad Radiol. 2016;23(3):344-352.
23. Corwin MT, Fananapazir G, Jin M, Lamba R, Bashir MR. Difference in liver imaging and reporting data system categorization between MRI and CT. Am J Roentgenol. 2016;206(2):307-312.
1. El–Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132(7):2557-2576.
2. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011;365(12):1118-1127.
3. Bruix J, Sherman M; American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53(3):1020-1022.
4. Selvapatt N, House H, Brown A. Hepatocellular carcinoma surveillance: are we utilizing it? J Clin Gastroenterol. 2016;50(1):e8-e12.
5. Lee JM, Yoon JH, Joo I, Woo HS. Recent advances in CT and MR imaging for evaluation of hepatocellular carcinoma. Liver Cancer. 2012;1(1):22-40.
6. Chou R, Cuevas C, Fu R, et al. Imaging techniques for the diagnosis of hepatocellular carcinoma: a systemic review and meta-analysis. Ann Intern Med. 2015;162(10):697-711.
7. Ariff B, Lloyd CR, Khan S, et al. Imaging of liver cancer. World J Gastroenterol. 2009;15(11):1289-1300.
8. Yuen MF, Cheng CC, Lauder IJ, Lam SK, Ooi CG, Lai CL. Early detection of hepatocellular carcinoma increases the chance of treatment: Hong Kong experience. Hepatology. 2000;31(2):330-335.
9. Singal AG, Pillai A, Tiro J. Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS Med. 2014;11(4):e1001624.
10. Nusbaum, JD, Smirniotopoulos J, Wright HC, et al. The effect of hepatocellular carcinoma surveillance in an urban population with liver cirrhosis. J Clin Gastroenterol. 2015;49(10):e91-e95.
11. Kansagara D, Papak J, Pasha AS, et al. Screening for hepatocellular carcinoma in chronic liver disease: a systemic review. Ann Intern Med. 2014;161(4):261-269.
12. Shah S, Shukla A, Paunipagar B. Radiological features of hepatocellular carcinoma. J Clin Exp Hepatol. 2014;4(suppl 3):S63-S66.
13. You MW, Kim SY, Kim KW, et al. Recent advances in the imaging of hepatocellular carcinoma. Clin Mol Hepatol. 2015;21(1):95-103.
14. American College of Radiology. Liver reporting and data system (LI-RADS). https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS. Accessed April 10, 2018.
15. Anis M. Imaging of hepatocellular carcinoma: new approaches to diagnosis. Clin Liver Dis. 2015;19(2):325-340.
16. Mitchell D, Bruix J, Sherman M, Sirlin CB. LI-RADS (Liver Imaging Reporting and Data System): summary, discussion, and consensus of the LI-RADS Management Working Group and future directions. Hepatology. 2015;61(3):1056-1065.
17. Darnell A, Forner A, Rimola J, et al. Liver imaging reporting and data system with MR imaging: evaluation in nodules 20 mm or smaller detected in cirrhosis at screening US. Radiology. 2015; 275(3):698-707.
18. Davenport MS, Khalatbari S, Liu PS, et al. Repeatability of diagnostic features and scoring systems for hepatocellular carcinoma by using MR imaging. Radiology. 2014;272(1):132-142.
19. An C, Rakhmonova G, Choi JY, Kim MJ. Liver imaging reporting and data system (LI-RADS) version 2014: understanding and application of the diagnostic algorithm. Clin Mol Hepatol. 2016;22(2):296-307.
20. Zhao W, Li W, Yi X, et al. [Diagnostic value of liver imaging reporting and data system on primary hepatocellular carcinoma] [in Chinese]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016;41(4):380-387.
21. Choi SH, Byun JH, Kim SY, et al. Liver imaging reporting and data system v2014 with gadoxetate disodium-enhanced magnetic resonance imaging: validation of LIRADS category 4 and 5 criteria. Invest Radiol. 2016;51(8):483-490.
22. Zhang YD, Zhu FP, Xu X, et al. Liver imaging reporting and data system: substantial discordance between CT and MR for imaging classification of hepatic nodules. Acad Radiol. 2016;23(3):344-352.
23. Corwin MT, Fananapazir G, Jin M, Lamba R, Bashir MR. Difference in liver imaging and reporting data system categorization between MRI and CT. Am J Roentgenol. 2016;206(2):307-312.
Out-of-hospital cardiac arrests soar during COVID-19 in Italy
Out-of-hospital cardiac arrests increased 58% during the peak of the COVID-19 outbreak in the hard-hit region of Lombardy, Italy, compared with the same period last year, a new analysis shows.
During the first 40 days of the outbreak beginning Feb. 21, four provinces in northern Italy reported 362 cases of out-of-hospital cardiac arrest compared with 229 during the same period in 2019.
The increases in these provinces varied in magnitude from 18% in Mantua, where there were 1,688 confirmed COVID-19 cases, to 187% in Lodi, which had 2,116 COVID-19 cases. The Cremona province, which had the highest number of COVID-19 cases at 3,869, saw a 143% increase in out-of-hospital cardiac arrests.
The mortality rate in the field was 14.9 percentage points higher in 2020 than in 2019 among patients in whom resuscitation was attempted by emergency medical services (EMS), Enrico Baldi, MD, University of Pavia, Italy, and colleagues reported in a letter April 29 in the New England Journal of Medicine.
“The sex and age of the patients were similar in the 2020 and 2019 periods, but in 2020, the incidence of out-of-hospital cardiac arrest due to a medical cause was 6.5 percentage points higher, the incidence of out-of-hospital cardiac arrest at home was 7.3 percentage points higher, and the incidence of unwitnessed cardiac arrest was 11.3 percentage points higher,” the authors wrote.
Patients were also less likely to receive cardiopulmonary resuscitation from bystanders in 2020 vs 2019 (–15.6 percentage points) and were more likely to die before reaching the hospital when resuscitation was attempted by EMS (+14.9 percentage points).
Among all patients, the death rate in the field increased 11.4 percentage points during the outbreak, from 77.3% in 2019 to 88.7% in 2020.
The cumulative incidence of out-of-hospital cardiac arrest in 2020 was “strongly associated” with the cumulative incidence of COVID-19 (Spearman rank correlation coefficient, 0.87; 95% confidence interval, 0.83-0.91) and the spike in cases “followed the time course of the COVID-19 outbreak,” the researchers noted.
A total of 103 patients, who arrested out of hospital and were diagnosed with or suspected of having COVID-19, “account for 77.4% of the increase in cases of out-of-hospital cardiac arrest observed in these provinces in 2020,” the investigators noted.
As the pandemic has taken hold, hospitals and physicians across the United States are also voicing concerns about the drop in the number of patients presenting with myocardial infarction (MI) or stroke.
Nearly one-third of Americans (29%) report having delayed or avoided medical care because of concerns of catching COVID-19, according to a new poll released April 28 from the American College of Emergency Physicians (ACEP) and Morning Consult, a global data research firm.
Despite many emergency departments reporting a decline in patient volume, 74% of respondents said they were worried about hospital wait times and overcrowding. Another 59% expressed concerns about being turned away from the hospital or doctor’s office.
At the same time, the survey found strong support for emergency physicians and 73% of respondents said they were concerned about overstressing the health care system.
The drop-off in Americans seeking care for MI and strokes nationally prompted eight professional societies – including ACEP, the American Heart Association, and the Association of Black Cardiologists – to issue a joint statement urging those experiencing symptoms to call 911 and seek care for these life-threatening events.
The authors have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Out-of-hospital cardiac arrests increased 58% during the peak of the COVID-19 outbreak in the hard-hit region of Lombardy, Italy, compared with the same period last year, a new analysis shows.
During the first 40 days of the outbreak beginning Feb. 21, four provinces in northern Italy reported 362 cases of out-of-hospital cardiac arrest compared with 229 during the same period in 2019.
The increases in these provinces varied in magnitude from 18% in Mantua, where there were 1,688 confirmed COVID-19 cases, to 187% in Lodi, which had 2,116 COVID-19 cases. The Cremona province, which had the highest number of COVID-19 cases at 3,869, saw a 143% increase in out-of-hospital cardiac arrests.
The mortality rate in the field was 14.9 percentage points higher in 2020 than in 2019 among patients in whom resuscitation was attempted by emergency medical services (EMS), Enrico Baldi, MD, University of Pavia, Italy, and colleagues reported in a letter April 29 in the New England Journal of Medicine.
“The sex and age of the patients were similar in the 2020 and 2019 periods, but in 2020, the incidence of out-of-hospital cardiac arrest due to a medical cause was 6.5 percentage points higher, the incidence of out-of-hospital cardiac arrest at home was 7.3 percentage points higher, and the incidence of unwitnessed cardiac arrest was 11.3 percentage points higher,” the authors wrote.
Patients were also less likely to receive cardiopulmonary resuscitation from bystanders in 2020 vs 2019 (–15.6 percentage points) and were more likely to die before reaching the hospital when resuscitation was attempted by EMS (+14.9 percentage points).
Among all patients, the death rate in the field increased 11.4 percentage points during the outbreak, from 77.3% in 2019 to 88.7% in 2020.
The cumulative incidence of out-of-hospital cardiac arrest in 2020 was “strongly associated” with the cumulative incidence of COVID-19 (Spearman rank correlation coefficient, 0.87; 95% confidence interval, 0.83-0.91) and the spike in cases “followed the time course of the COVID-19 outbreak,” the researchers noted.
A total of 103 patients, who arrested out of hospital and were diagnosed with or suspected of having COVID-19, “account for 77.4% of the increase in cases of out-of-hospital cardiac arrest observed in these provinces in 2020,” the investigators noted.
As the pandemic has taken hold, hospitals and physicians across the United States are also voicing concerns about the drop in the number of patients presenting with myocardial infarction (MI) or stroke.
Nearly one-third of Americans (29%) report having delayed or avoided medical care because of concerns of catching COVID-19, according to a new poll released April 28 from the American College of Emergency Physicians (ACEP) and Morning Consult, a global data research firm.
Despite many emergency departments reporting a decline in patient volume, 74% of respondents said they were worried about hospital wait times and overcrowding. Another 59% expressed concerns about being turned away from the hospital or doctor’s office.
At the same time, the survey found strong support for emergency physicians and 73% of respondents said they were concerned about overstressing the health care system.
The drop-off in Americans seeking care for MI and strokes nationally prompted eight professional societies – including ACEP, the American Heart Association, and the Association of Black Cardiologists – to issue a joint statement urging those experiencing symptoms to call 911 and seek care for these life-threatening events.
The authors have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Out-of-hospital cardiac arrests increased 58% during the peak of the COVID-19 outbreak in the hard-hit region of Lombardy, Italy, compared with the same period last year, a new analysis shows.
During the first 40 days of the outbreak beginning Feb. 21, four provinces in northern Italy reported 362 cases of out-of-hospital cardiac arrest compared with 229 during the same period in 2019.
The increases in these provinces varied in magnitude from 18% in Mantua, where there were 1,688 confirmed COVID-19 cases, to 187% in Lodi, which had 2,116 COVID-19 cases. The Cremona province, which had the highest number of COVID-19 cases at 3,869, saw a 143% increase in out-of-hospital cardiac arrests.
The mortality rate in the field was 14.9 percentage points higher in 2020 than in 2019 among patients in whom resuscitation was attempted by emergency medical services (EMS), Enrico Baldi, MD, University of Pavia, Italy, and colleagues reported in a letter April 29 in the New England Journal of Medicine.
“The sex and age of the patients were similar in the 2020 and 2019 periods, but in 2020, the incidence of out-of-hospital cardiac arrest due to a medical cause was 6.5 percentage points higher, the incidence of out-of-hospital cardiac arrest at home was 7.3 percentage points higher, and the incidence of unwitnessed cardiac arrest was 11.3 percentage points higher,” the authors wrote.
Patients were also less likely to receive cardiopulmonary resuscitation from bystanders in 2020 vs 2019 (–15.6 percentage points) and were more likely to die before reaching the hospital when resuscitation was attempted by EMS (+14.9 percentage points).
Among all patients, the death rate in the field increased 11.4 percentage points during the outbreak, from 77.3% in 2019 to 88.7% in 2020.
The cumulative incidence of out-of-hospital cardiac arrest in 2020 was “strongly associated” with the cumulative incidence of COVID-19 (Spearman rank correlation coefficient, 0.87; 95% confidence interval, 0.83-0.91) and the spike in cases “followed the time course of the COVID-19 outbreak,” the researchers noted.
A total of 103 patients, who arrested out of hospital and were diagnosed with or suspected of having COVID-19, “account for 77.4% of the increase in cases of out-of-hospital cardiac arrest observed in these provinces in 2020,” the investigators noted.
As the pandemic has taken hold, hospitals and physicians across the United States are also voicing concerns about the drop in the number of patients presenting with myocardial infarction (MI) or stroke.
Nearly one-third of Americans (29%) report having delayed or avoided medical care because of concerns of catching COVID-19, according to a new poll released April 28 from the American College of Emergency Physicians (ACEP) and Morning Consult, a global data research firm.
Despite many emergency departments reporting a decline in patient volume, 74% of respondents said they were worried about hospital wait times and overcrowding. Another 59% expressed concerns about being turned away from the hospital or doctor’s office.
At the same time, the survey found strong support for emergency physicians and 73% of respondents said they were concerned about overstressing the health care system.
The drop-off in Americans seeking care for MI and strokes nationally prompted eight professional societies – including ACEP, the American Heart Association, and the Association of Black Cardiologists – to issue a joint statement urging those experiencing symptoms to call 911 and seek care for these life-threatening events.
The authors have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Continuity rules
Simple operational adjustments your team can make
Although there are many benefits to the hospital medicine model of inpatient care, there is perhaps no greater Achilles heel than the discontinuity inherent to the care model. The trust and familiarity garnered from longitudinal patient-provider relationships is sacrificed for the benefits of focused practice, efficiency, and enhanced availability.
Any system involves competing priorities, and some degree of discontinuity is inevitable. Would it make sense for a hospitalist to stay on service until every panel patient is discharged? For obvious economic, lifestyle, and other reasons, of course not. Our charge then is not to make the perfect the enemy of the good, but to ensure thoughtful and consistent continuity for the good of the patient, the provider, and the hospital. The following tips should help your team achieve the best possible balance.
Avoid orphan rounding shifts
An “orphan” rounding shift refers to a single shift untethered to a stretch. For admitting or administrative duties, this generally poses no problem, but for a rounding shift it is undesirable. No matter how talented or industrious the provider, it is very difficult for them to effectively provide seamless care for a single day; such care is often disconcerting for patients, families, case managers, and consultants. In situations such as significant census spikes, this may be a necessary evil, but avoid this if you can.
Orphan shift duties
If you can’t avoid an orphan rounding shift, be creative regarding which patients get assigned. Can that provider cover observation or simple short stay patients who may be discharged, or consult follow ups that may be signed off? Can they see stable long-stay patients where the plan isn’t changing and the patient isn’t going anywhere? (Think guardianships, chronic ventilated patients awaiting a facility, stable patients with a history of intravenous drug abuse who may not be safely discharged with a line, etc.) Can they do lab, culture, or path report follow-up calls? Getting creative in responsibilities for an orphan shift can benefit all involved.
Rounding shifts following admitting shifts
Dedicated admitting and rounding shifts are the norm these days. But rather than a pure stretch of one or the other, consider a few days admitting followed by the rest of the stretch rounding. Particularly in a small- to mid-sized hospital, multiple admits done over a few days (and especially if also cross-covering floor calls) will mean many familiar cases when rounding thereafter.
Standard sign-out that travels with patients
The hospital is a dynamic environment. Patients, providers and staff move around a lot. Given this reality, the importance of a complete standardized and accessible sign-out is paramount.
Imagine a rounder starting their last day with 15 patients. By the end of the shift, some have been discharged, transferred to telemetry or the ICU, or left against medical advice, leaving seven patients to sign out. By the next day, there are eight new faces, including fresh admits or consults from the prior day, swing, and night providers as well as existing patients transferred from telemetry/ICU to the general medical ward. A practical solution incorporates an asynchronous sign-out that travels with the patient regardless of geographic location or which provider(s) are following them. Billing software or census reports can typically achieve this. Of course, allow for additional verbal communication as necessary and appropriate.
Geographic rounds, with exceptions
Geographic rounds make a lot of sense most of the time. Less transit time and phone tag and more frequent interactions with the care team make for a more efficient day. But sometimes it’s best to bend this rule.
A patient that you’ve seen for 5 days and was transferred off your telemetry floor to go home tomorrow might best be served by you trekking up a flight of stairs to do the discharge. Similarly, complicated medical, psychosocial, or other circumstances may argue for keeping the patient on your list despite a change in location.
The above rules are foundational elements for good continuity. Two bonus considerations include:
Wind up, wind down
It’s difficult to walk into a full panel of patients especially when many have been in house for a while. Consider overlapping providers coming onto and going off a shared service.
In a buddy arrangement the oncoming provider starting would take new patients from the outgoing provider finishing. The provider finishing discharges patients with long length of stays and continues to round on more-complicated patients with whom they are familiar. Opportunities for face-to-face verbal handover, and even bedside introduction to the provider starting, can improve care coordination and safety and enhance the patient experience.
Reconsider split rounding and admitting
Most physicians would attest that the second time seeing a patient is much easier than the first, the third easier than the second, and so on. This holds true even more so when the first encounter is the history and physical, and the provider subsequently rounds on the patient for the duration of the hospitalization.
You know what the plan is because you made it; you are confident that the patient’s leg with cellulitis looks better or the patient with congested lungs sounds clearer because the baseline against which you’re comparing is your own. It can be a challenge to interrupt a busy day of clinical rounds, discharges, and interdisciplinary meetings to admit a patient. But the upstream investment pays rich downstream dividends and is well worth consideration.
Hospital medicine outcomes as measured by cost, quality, and patient and provider experience are often hampered by suboptimal continuity of care. With recognition of the problem and some simple operational adjustments as outlined above, your team can minimize negative impacts.
Dr. Krisa is a former regional medical director for a national hospitalist group and currently serves as a physician advisor for St. Peter’s Health Partners, a large integrated health system in Albany, N.Y. You can contact him at [email protected].
Simple operational adjustments your team can make
Simple operational adjustments your team can make
Although there are many benefits to the hospital medicine model of inpatient care, there is perhaps no greater Achilles heel than the discontinuity inherent to the care model. The trust and familiarity garnered from longitudinal patient-provider relationships is sacrificed for the benefits of focused practice, efficiency, and enhanced availability.
Any system involves competing priorities, and some degree of discontinuity is inevitable. Would it make sense for a hospitalist to stay on service until every panel patient is discharged? For obvious economic, lifestyle, and other reasons, of course not. Our charge then is not to make the perfect the enemy of the good, but to ensure thoughtful and consistent continuity for the good of the patient, the provider, and the hospital. The following tips should help your team achieve the best possible balance.
Avoid orphan rounding shifts
An “orphan” rounding shift refers to a single shift untethered to a stretch. For admitting or administrative duties, this generally poses no problem, but for a rounding shift it is undesirable. No matter how talented or industrious the provider, it is very difficult for them to effectively provide seamless care for a single day; such care is often disconcerting for patients, families, case managers, and consultants. In situations such as significant census spikes, this may be a necessary evil, but avoid this if you can.
Orphan shift duties
If you can’t avoid an orphan rounding shift, be creative regarding which patients get assigned. Can that provider cover observation or simple short stay patients who may be discharged, or consult follow ups that may be signed off? Can they see stable long-stay patients where the plan isn’t changing and the patient isn’t going anywhere? (Think guardianships, chronic ventilated patients awaiting a facility, stable patients with a history of intravenous drug abuse who may not be safely discharged with a line, etc.) Can they do lab, culture, or path report follow-up calls? Getting creative in responsibilities for an orphan shift can benefit all involved.
Rounding shifts following admitting shifts
Dedicated admitting and rounding shifts are the norm these days. But rather than a pure stretch of one or the other, consider a few days admitting followed by the rest of the stretch rounding. Particularly in a small- to mid-sized hospital, multiple admits done over a few days (and especially if also cross-covering floor calls) will mean many familiar cases when rounding thereafter.
Standard sign-out that travels with patients
The hospital is a dynamic environment. Patients, providers and staff move around a lot. Given this reality, the importance of a complete standardized and accessible sign-out is paramount.
Imagine a rounder starting their last day with 15 patients. By the end of the shift, some have been discharged, transferred to telemetry or the ICU, or left against medical advice, leaving seven patients to sign out. By the next day, there are eight new faces, including fresh admits or consults from the prior day, swing, and night providers as well as existing patients transferred from telemetry/ICU to the general medical ward. A practical solution incorporates an asynchronous sign-out that travels with the patient regardless of geographic location or which provider(s) are following them. Billing software or census reports can typically achieve this. Of course, allow for additional verbal communication as necessary and appropriate.
Geographic rounds, with exceptions
Geographic rounds make a lot of sense most of the time. Less transit time and phone tag and more frequent interactions with the care team make for a more efficient day. But sometimes it’s best to bend this rule.
A patient that you’ve seen for 5 days and was transferred off your telemetry floor to go home tomorrow might best be served by you trekking up a flight of stairs to do the discharge. Similarly, complicated medical, psychosocial, or other circumstances may argue for keeping the patient on your list despite a change in location.
The above rules are foundational elements for good continuity. Two bonus considerations include:
Wind up, wind down
It’s difficult to walk into a full panel of patients especially when many have been in house for a while. Consider overlapping providers coming onto and going off a shared service.
In a buddy arrangement the oncoming provider starting would take new patients from the outgoing provider finishing. The provider finishing discharges patients with long length of stays and continues to round on more-complicated patients with whom they are familiar. Opportunities for face-to-face verbal handover, and even bedside introduction to the provider starting, can improve care coordination and safety and enhance the patient experience.
Reconsider split rounding and admitting
Most physicians would attest that the second time seeing a patient is much easier than the first, the third easier than the second, and so on. This holds true even more so when the first encounter is the history and physical, and the provider subsequently rounds on the patient for the duration of the hospitalization.
You know what the plan is because you made it; you are confident that the patient’s leg with cellulitis looks better or the patient with congested lungs sounds clearer because the baseline against which you’re comparing is your own. It can be a challenge to interrupt a busy day of clinical rounds, discharges, and interdisciplinary meetings to admit a patient. But the upstream investment pays rich downstream dividends and is well worth consideration.
Hospital medicine outcomes as measured by cost, quality, and patient and provider experience are often hampered by suboptimal continuity of care. With recognition of the problem and some simple operational adjustments as outlined above, your team can minimize negative impacts.
Dr. Krisa is a former regional medical director for a national hospitalist group and currently serves as a physician advisor for St. Peter’s Health Partners, a large integrated health system in Albany, N.Y. You can contact him at [email protected].
Although there are many benefits to the hospital medicine model of inpatient care, there is perhaps no greater Achilles heel than the discontinuity inherent to the care model. The trust and familiarity garnered from longitudinal patient-provider relationships is sacrificed for the benefits of focused practice, efficiency, and enhanced availability.
Any system involves competing priorities, and some degree of discontinuity is inevitable. Would it make sense for a hospitalist to stay on service until every panel patient is discharged? For obvious economic, lifestyle, and other reasons, of course not. Our charge then is not to make the perfect the enemy of the good, but to ensure thoughtful and consistent continuity for the good of the patient, the provider, and the hospital. The following tips should help your team achieve the best possible balance.
Avoid orphan rounding shifts
An “orphan” rounding shift refers to a single shift untethered to a stretch. For admitting or administrative duties, this generally poses no problem, but for a rounding shift it is undesirable. No matter how talented or industrious the provider, it is very difficult for them to effectively provide seamless care for a single day; such care is often disconcerting for patients, families, case managers, and consultants. In situations such as significant census spikes, this may be a necessary evil, but avoid this if you can.
Orphan shift duties
If you can’t avoid an orphan rounding shift, be creative regarding which patients get assigned. Can that provider cover observation or simple short stay patients who may be discharged, or consult follow ups that may be signed off? Can they see stable long-stay patients where the plan isn’t changing and the patient isn’t going anywhere? (Think guardianships, chronic ventilated patients awaiting a facility, stable patients with a history of intravenous drug abuse who may not be safely discharged with a line, etc.) Can they do lab, culture, or path report follow-up calls? Getting creative in responsibilities for an orphan shift can benefit all involved.
Rounding shifts following admitting shifts
Dedicated admitting and rounding shifts are the norm these days. But rather than a pure stretch of one or the other, consider a few days admitting followed by the rest of the stretch rounding. Particularly in a small- to mid-sized hospital, multiple admits done over a few days (and especially if also cross-covering floor calls) will mean many familiar cases when rounding thereafter.
Standard sign-out that travels with patients
The hospital is a dynamic environment. Patients, providers and staff move around a lot. Given this reality, the importance of a complete standardized and accessible sign-out is paramount.
Imagine a rounder starting their last day with 15 patients. By the end of the shift, some have been discharged, transferred to telemetry or the ICU, or left against medical advice, leaving seven patients to sign out. By the next day, there are eight new faces, including fresh admits or consults from the prior day, swing, and night providers as well as existing patients transferred from telemetry/ICU to the general medical ward. A practical solution incorporates an asynchronous sign-out that travels with the patient regardless of geographic location or which provider(s) are following them. Billing software or census reports can typically achieve this. Of course, allow for additional verbal communication as necessary and appropriate.
Geographic rounds, with exceptions
Geographic rounds make a lot of sense most of the time. Less transit time and phone tag and more frequent interactions with the care team make for a more efficient day. But sometimes it’s best to bend this rule.
A patient that you’ve seen for 5 days and was transferred off your telemetry floor to go home tomorrow might best be served by you trekking up a flight of stairs to do the discharge. Similarly, complicated medical, psychosocial, or other circumstances may argue for keeping the patient on your list despite a change in location.
The above rules are foundational elements for good continuity. Two bonus considerations include:
Wind up, wind down
It’s difficult to walk into a full panel of patients especially when many have been in house for a while. Consider overlapping providers coming onto and going off a shared service.
In a buddy arrangement the oncoming provider starting would take new patients from the outgoing provider finishing. The provider finishing discharges patients with long length of stays and continues to round on more-complicated patients with whom they are familiar. Opportunities for face-to-face verbal handover, and even bedside introduction to the provider starting, can improve care coordination and safety and enhance the patient experience.
Reconsider split rounding and admitting
Most physicians would attest that the second time seeing a patient is much easier than the first, the third easier than the second, and so on. This holds true even more so when the first encounter is the history and physical, and the provider subsequently rounds on the patient for the duration of the hospitalization.
You know what the plan is because you made it; you are confident that the patient’s leg with cellulitis looks better or the patient with congested lungs sounds clearer because the baseline against which you’re comparing is your own. It can be a challenge to interrupt a busy day of clinical rounds, discharges, and interdisciplinary meetings to admit a patient. But the upstream investment pays rich downstream dividends and is well worth consideration.
Hospital medicine outcomes as measured by cost, quality, and patient and provider experience are often hampered by suboptimal continuity of care. With recognition of the problem and some simple operational adjustments as outlined above, your team can minimize negative impacts.
Dr. Krisa is a former regional medical director for a national hospitalist group and currently serves as a physician advisor for St. Peter’s Health Partners, a large integrated health system in Albany, N.Y. You can contact him at [email protected].
COVID-19: A ‘marathon, not a sprint’ for psychiatry
The tragic death by suicide of an emergency department physician who had been caring for COVID-19 patients in New York City underscores the huge psychological impact of the pandemic – which will linger long after the virus is gone, experts say.
“For frontline responders, the trauma of witnessing so much illness and death will have lasting effects for many,” Bruce Schwartz, MD, president of the American Psychiatric Association (APA), said during the opening session of the annual meeting of the American Psychiatric Association, which was held as a virtual live event, replacing the organization’s canceled annual meeting.
“We will need the full workforce to cope with the psychiatric effects” of the pandemic, added Dr. Schwartz, deputy chairman and professor, department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York.
Joshua Morganstein, MD, chair of the APA’s Committee on the Psychiatric Dimensions of Disaster, led an afternoon session at the virtual meeting on “healthcare worker and organizational sustainment during COVID-19.”
The crisis is shaping up to be “a marathon, not a sprint; and self-care will remain a critical and ongoing issue. We are in this together,” he said.
Once the pandemic passes, Dr. Morganstein added.
Psychological first aid
It is important to realize that the psychological and behavioral effects of disasters are experienced by “more people, over a greater geography, across a much longer period of time than all other medical effects combined. This is important for disaster resource planning,” Dr. Morganstein told meeting attendees.
At times of crisis, many people will experience distress reactions and engage in behaviors that put their health at risk. Insomnia, increased alcohol and substance use, and family conflict are common and have a negative impact on functioning, he said.
In addition, pandemics result in unique responses. Protracted fear and uncertainty, elements of isolation, anger, misinformation, and faltering confidence in government/institutions may alter perceptions of risk.
“It’s the perception of risk, not the actual risk, that will ultimately determine how people behave,” Dr. Morganstein said.
“The ability to influence risk perception will alter the degree to which any group, community, or population ultimately chooses to engage in or reject recommended health behaviors,” he added.
In times of crisis, it’s also helpful to keep in mind and act upon the five essential elements of “psychological first aid,” he noted. These are safety, calming, self/community efficacy, social connectedness, and hope/optimism.
Psychological first aid is an evidence-based framework of supporting resilience in individuals, communities, and organizations, Dr. Morganstein said.
Individuals have a wide range of needs during times of crisis, and support should be tailored accordingly, he noted. As with many crises, instrumental support needs are significant and may be the primary need for many people. These include the need for food, clothing, rent/mortgage, financial relief, and child care.
Providing emotional support – empathy, validation, self-actualization, encouragement, and insight – will help individuals engage with instrumental supports.
“The reality is that it’s often difficult to talk about being sad when you feel hungry or worried you can’t pay the rent,” said Dr. Morganstein.
He also emphasized the importance of appropriate messaging and language during a crisis. These can have a profound impact on community well-being and the willingness of the public to engage in recommended health behaviors.
“As psychiatrists, we understand [that] the words we choose when we discuss this pandemic will have power. Communication is not only a means by which we deliver interventions, but it is, in and of itself, a behavioral health intervention. Good communication can serve to normalize experiences and function as an antidote to distress during times of uncertainty,” Dr. Morganstein said.
Importantly, “we need to remind people that eventually this will end and the vast majority of people, including those who have difficulties along the way, will ultimately be okay.”
The APA has provided a COVID-19 resource page on its website.
Dr. Morganstein and Dr. Schwartz have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The tragic death by suicide of an emergency department physician who had been caring for COVID-19 patients in New York City underscores the huge psychological impact of the pandemic – which will linger long after the virus is gone, experts say.
“For frontline responders, the trauma of witnessing so much illness and death will have lasting effects for many,” Bruce Schwartz, MD, president of the American Psychiatric Association (APA), said during the opening session of the annual meeting of the American Psychiatric Association, which was held as a virtual live event, replacing the organization’s canceled annual meeting.
“We will need the full workforce to cope with the psychiatric effects” of the pandemic, added Dr. Schwartz, deputy chairman and professor, department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York.
Joshua Morganstein, MD, chair of the APA’s Committee on the Psychiatric Dimensions of Disaster, led an afternoon session at the virtual meeting on “healthcare worker and organizational sustainment during COVID-19.”
The crisis is shaping up to be “a marathon, not a sprint; and self-care will remain a critical and ongoing issue. We are in this together,” he said.
Once the pandemic passes, Dr. Morganstein added.
Psychological first aid
It is important to realize that the psychological and behavioral effects of disasters are experienced by “more people, over a greater geography, across a much longer period of time than all other medical effects combined. This is important for disaster resource planning,” Dr. Morganstein told meeting attendees.
At times of crisis, many people will experience distress reactions and engage in behaviors that put their health at risk. Insomnia, increased alcohol and substance use, and family conflict are common and have a negative impact on functioning, he said.
In addition, pandemics result in unique responses. Protracted fear and uncertainty, elements of isolation, anger, misinformation, and faltering confidence in government/institutions may alter perceptions of risk.
“It’s the perception of risk, not the actual risk, that will ultimately determine how people behave,” Dr. Morganstein said.
“The ability to influence risk perception will alter the degree to which any group, community, or population ultimately chooses to engage in or reject recommended health behaviors,” he added.
In times of crisis, it’s also helpful to keep in mind and act upon the five essential elements of “psychological first aid,” he noted. These are safety, calming, self/community efficacy, social connectedness, and hope/optimism.
Psychological first aid is an evidence-based framework of supporting resilience in individuals, communities, and organizations, Dr. Morganstein said.
Individuals have a wide range of needs during times of crisis, and support should be tailored accordingly, he noted. As with many crises, instrumental support needs are significant and may be the primary need for many people. These include the need for food, clothing, rent/mortgage, financial relief, and child care.
Providing emotional support – empathy, validation, self-actualization, encouragement, and insight – will help individuals engage with instrumental supports.
“The reality is that it’s often difficult to talk about being sad when you feel hungry or worried you can’t pay the rent,” said Dr. Morganstein.
He also emphasized the importance of appropriate messaging and language during a crisis. These can have a profound impact on community well-being and the willingness of the public to engage in recommended health behaviors.
“As psychiatrists, we understand [that] the words we choose when we discuss this pandemic will have power. Communication is not only a means by which we deliver interventions, but it is, in and of itself, a behavioral health intervention. Good communication can serve to normalize experiences and function as an antidote to distress during times of uncertainty,” Dr. Morganstein said.
Importantly, “we need to remind people that eventually this will end and the vast majority of people, including those who have difficulties along the way, will ultimately be okay.”
The APA has provided a COVID-19 resource page on its website.
Dr. Morganstein and Dr. Schwartz have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The tragic death by suicide of an emergency department physician who had been caring for COVID-19 patients in New York City underscores the huge psychological impact of the pandemic – which will linger long after the virus is gone, experts say.
“For frontline responders, the trauma of witnessing so much illness and death will have lasting effects for many,” Bruce Schwartz, MD, president of the American Psychiatric Association (APA), said during the opening session of the annual meeting of the American Psychiatric Association, which was held as a virtual live event, replacing the organization’s canceled annual meeting.
“We will need the full workforce to cope with the psychiatric effects” of the pandemic, added Dr. Schwartz, deputy chairman and professor, department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York.
Joshua Morganstein, MD, chair of the APA’s Committee on the Psychiatric Dimensions of Disaster, led an afternoon session at the virtual meeting on “healthcare worker and organizational sustainment during COVID-19.”
The crisis is shaping up to be “a marathon, not a sprint; and self-care will remain a critical and ongoing issue. We are in this together,” he said.
Once the pandemic passes, Dr. Morganstein added.
Psychological first aid
It is important to realize that the psychological and behavioral effects of disasters are experienced by “more people, over a greater geography, across a much longer period of time than all other medical effects combined. This is important for disaster resource planning,” Dr. Morganstein told meeting attendees.
At times of crisis, many people will experience distress reactions and engage in behaviors that put their health at risk. Insomnia, increased alcohol and substance use, and family conflict are common and have a negative impact on functioning, he said.
In addition, pandemics result in unique responses. Protracted fear and uncertainty, elements of isolation, anger, misinformation, and faltering confidence in government/institutions may alter perceptions of risk.
“It’s the perception of risk, not the actual risk, that will ultimately determine how people behave,” Dr. Morganstein said.
“The ability to influence risk perception will alter the degree to which any group, community, or population ultimately chooses to engage in or reject recommended health behaviors,” he added.
In times of crisis, it’s also helpful to keep in mind and act upon the five essential elements of “psychological first aid,” he noted. These are safety, calming, self/community efficacy, social connectedness, and hope/optimism.
Psychological first aid is an evidence-based framework of supporting resilience in individuals, communities, and organizations, Dr. Morganstein said.
Individuals have a wide range of needs during times of crisis, and support should be tailored accordingly, he noted. As with many crises, instrumental support needs are significant and may be the primary need for many people. These include the need for food, clothing, rent/mortgage, financial relief, and child care.
Providing emotional support – empathy, validation, self-actualization, encouragement, and insight – will help individuals engage with instrumental supports.
“The reality is that it’s often difficult to talk about being sad when you feel hungry or worried you can’t pay the rent,” said Dr. Morganstein.
He also emphasized the importance of appropriate messaging and language during a crisis. These can have a profound impact on community well-being and the willingness of the public to engage in recommended health behaviors.
“As psychiatrists, we understand [that] the words we choose when we discuss this pandemic will have power. Communication is not only a means by which we deliver interventions, but it is, in and of itself, a behavioral health intervention. Good communication can serve to normalize experiences and function as an antidote to distress during times of uncertainty,” Dr. Morganstein said.
Importantly, “we need to remind people that eventually this will end and the vast majority of people, including those who have difficulties along the way, will ultimately be okay.”
The APA has provided a COVID-19 resource page on its website.
Dr. Morganstein and Dr. Schwartz have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM APA 2020
New study of diabetes drug for COVID-19 raises eyebrows
A just-launched study of the type 2 diabetes agent dapagliflozin (Farxiga, AstraZeneca) in patients with mild to moderate COVID-19 is raising eyebrows, given that several expert groups have advised that drugs in this class – the sodium-glucose cotransporter 2 (SGLT2) inhibitors – be stopped in all patients hospitalized with COVID-19 because of the increased risk for diabetic ketoacidosis (DKA).
The randomized, double-blind, placebo-controlled, phase 3 Dapagliflozin in Respiratory Failure in Patients With COVID-19 (DARE-19) study is sponsored by AstraZeneca and Saint Luke’s Mid America Heart Institute.
The trial will assess whether dapagliflozin reduces the risks of disease progression, clinical complications, and death because of COVID-19 in patients with type 2 diabetes, cardiovascular disease, and/or mild to moderate chronic kidney disease (CKD).
“Dapagliflozin has demonstrated cardio- and renal-protective benefits and improved outcomes in high-risk patients with type 2 diabetes, heart failure with reduced ejection fraction, and CKD,” said the principal investigator of DARE-19, Mikhail N. Kosiborod, MD, a cardiologist at Saint Luke’s Mid America Heart Institute, Kansas City, Mo.
And “patients with COVID-19 and underlying cardiometabolic disease appear to be at the highest risk of morbid complications,” he explained in an AstraZeneca statement.
“Through DARE-19, we hope to decrease the severity of illness, and prevent cardiovascular, respiratory, and kidney decompensation, which are common in patients with COVID-19,” Dr. Kosiborod continued.
However, advice to stop SGLT2 inhibitors in patients hospitalized with COVID-19 because of its associated DKA risk has come from several channels.
These include initial guidance from Diabetes UK; experts who spoke during an American Diabetes Association webinar; and most recently, an international panel of diabetes experts.
Some clinicians went so far as to say that they view the trial as potentially dangerous, while others said they could see some logic to it, as long as it is carefully managed.
“A dangerous proposition – a DARE I would not take”
Partha Kar, MD, of Portsmouth Hospitals NHS Trust and national clinical director of diabetes at NHS England, said in an interview: “It’s interesting to see [AstraZeneca] embark on a study with a particular class of drug whereby ... [in] the UK we have said that if you get sent to hospital with COVID-19 you should stop [SGLT2 inhibitors] immediately.”
It “sounds like a risky proposition to go ahead with, [and it] definitely made me raise an eyebrow,” he added.
Nephrologist Bruce R. Leslie, MD, of Seventh Doctor Consulting in Princeton, N.J., agreed with Dr. Kar.
“Giving SGLT2 inhibitors to patients in the DARE-19 study is a dangerous proposition because these drugs can induce ketoacidosis during the stress of acute illness such as COVID-19. ... Moreover, ketoacidosis is associated with hypercoagulability which could be especially dangerous in COVID-19, given that it has been causing thrombophilia with large-vessel occlusive strokes in young patients,” he said in an interview.
“One wonders how these risks were assessed by the authorities that approved the DARE-19 study,” said Dr. Leslie, who formerly worked for Bristol-Myers Squibb.
“How does the sponsor intend to secure informed consent given the risks? This is a DARE I would not take,” he said.
Asked to address these concerns, Dr. Kosiborod said in an interview that “the DARE-19 trial will assess both the efficacy and the safety of dapagliflozin in this patient population in a closely monitored environment of a rigorously designed randomized clinical trial. The trial protocol excludes patients with type 1 diabetes or at high risk for DKA.
“Furthermore, the protocol includes detailed specific instructions to ensure careful monitoring for DKA, including frequent assessments of acid-base status in the hospital setting. The safety data will be closely monitored by an independent data-monitoring committee,” he continued.
Dr. Kosiborod also pointed out that there is “no systematically collected information on the use of dapagliflozin or any other SGLT2 inhibitor in patients being treated for COVID-19, including the associated potential benefits, possible risks such as DKA, and the balance of these potential benefits and risks.”
DARE-19 design: Several outcomes will be examined
The DARE-19 trial is designed to enroll 900 adults with confirmed SARS-CoV-2 infection and oxygen saturation of 94% or greater.
Inclusion criteria include a medical history of hypertension, type 2 diabetes, atherosclerotic cardiovascular disease, heart failure, and/or stage 3-4 CKD. Exclusion criteria include current SGLT2 inhibitor treatment, type 1 diabetes, severe CKD, and severe COVID-19.
Dapagliflozin is approved in the EU for use in some patients with type 1 diabetes; this is not the case in the United States, although SGLT2 inhibitors in general are sometimes used off label in these patients.
Patients in DARE-19 will be randomized to 10 mg/day dapagliflozin or placebo for 30 days, in addition to standard care, in participating hospital. Primary outcomes are time to first occurrence of either death or new or worsened organ dysfunction, including respiratory decompensation, new or worsening heart failure, requirement for vasopressor therapy, ventricular tachycardia, and renal failure.
Secondary outcomes include a composite of time to death from any cause, time to new/worsened organ dysfunction, clinical status at day 30, and time to hospital discharge.
Rationale for the study
Irl B. Hirsch, MD, professor and diabetes treatment and teaching chair at the University of Washington, Seattle, said in an interview that he does see some logic to the trial.
Admitting that he doesn’t know much about “COVID-19 cardiomyopathy” – which would be one of the targets of dapagliflozin – other than it is quite common, he said that this, along with the potential renal benefits of dapagliflozin in the setting of COVID-19, make the study “intriguing.”
“Perhaps there is some rationale to it,” he said. However, “my concern is these sick COVID-19 patients are often acidemic, and besides the very complex acid-base challenges we see with intubated patients, these patients likely have combination lactic and ketoacidemia, the latter at least some from starvation.
“Still, if enough dextrose and insulin are provided to prevent ketoacid accumulation, my guess is it would do at least as well as hydroxychloroquine,” he said.
And Simon Heller, MD, professor of clinical diabetes at the University of Sheffield (England), said in an interview: “I think it is quite a brave study, mainly because of the increased risk of DKA.
“However, on the basis that these patients will be carefully monitored, the risk of DKA shouldn’t be great. I think it is important that patients with type 2 diabetes can participate whenever possible in such trials,” he said.
The estimated completion date for DARE-19 is December 2020.
Dr. Kosiborod has reported receiving grant support, honoraria, and/or research support from AstraZeneca, Boehringer Ingelheim, Sanofi, Amgen, Novo Nordisk, Merck, Eisai, Janssen, Bayer, GlaxoSmithKline, Glytec, Intarcia Therapeutics, Novartis, Applied Therapeutics, Amarin, and Eli Lilly. Dr. Leslie has reported owning stock in Bristol-Myers Squibb, Pfizer, and Lilly. Dr. Hirsch has reported consulting for Abbott Diabetes Care, Roche, and Bigfoot Biomedical, conducting research for Medtronic, and is a diabetes editor for UpToDate. Dr. Heller has received advisory or consultation fees from Lilly, Novo Nordisk, Takeda, MSD, and Becton Dickinson; has served as a speaker for AstraZeneca, Lilly, Novo Nordisk, Boehringer Ingelheim, and Takeda; and has received research support from Medtronic UK. He is on the advisory board for Medscape. Dr. Kar has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A just-launched study of the type 2 diabetes agent dapagliflozin (Farxiga, AstraZeneca) in patients with mild to moderate COVID-19 is raising eyebrows, given that several expert groups have advised that drugs in this class – the sodium-glucose cotransporter 2 (SGLT2) inhibitors – be stopped in all patients hospitalized with COVID-19 because of the increased risk for diabetic ketoacidosis (DKA).
The randomized, double-blind, placebo-controlled, phase 3 Dapagliflozin in Respiratory Failure in Patients With COVID-19 (DARE-19) study is sponsored by AstraZeneca and Saint Luke’s Mid America Heart Institute.
The trial will assess whether dapagliflozin reduces the risks of disease progression, clinical complications, and death because of COVID-19 in patients with type 2 diabetes, cardiovascular disease, and/or mild to moderate chronic kidney disease (CKD).
“Dapagliflozin has demonstrated cardio- and renal-protective benefits and improved outcomes in high-risk patients with type 2 diabetes, heart failure with reduced ejection fraction, and CKD,” said the principal investigator of DARE-19, Mikhail N. Kosiborod, MD, a cardiologist at Saint Luke’s Mid America Heart Institute, Kansas City, Mo.
And “patients with COVID-19 and underlying cardiometabolic disease appear to be at the highest risk of morbid complications,” he explained in an AstraZeneca statement.
“Through DARE-19, we hope to decrease the severity of illness, and prevent cardiovascular, respiratory, and kidney decompensation, which are common in patients with COVID-19,” Dr. Kosiborod continued.
However, advice to stop SGLT2 inhibitors in patients hospitalized with COVID-19 because of its associated DKA risk has come from several channels.
These include initial guidance from Diabetes UK; experts who spoke during an American Diabetes Association webinar; and most recently, an international panel of diabetes experts.
Some clinicians went so far as to say that they view the trial as potentially dangerous, while others said they could see some logic to it, as long as it is carefully managed.
“A dangerous proposition – a DARE I would not take”
Partha Kar, MD, of Portsmouth Hospitals NHS Trust and national clinical director of diabetes at NHS England, said in an interview: “It’s interesting to see [AstraZeneca] embark on a study with a particular class of drug whereby ... [in] the UK we have said that if you get sent to hospital with COVID-19 you should stop [SGLT2 inhibitors] immediately.”
It “sounds like a risky proposition to go ahead with, [and it] definitely made me raise an eyebrow,” he added.
Nephrologist Bruce R. Leslie, MD, of Seventh Doctor Consulting in Princeton, N.J., agreed with Dr. Kar.
“Giving SGLT2 inhibitors to patients in the DARE-19 study is a dangerous proposition because these drugs can induce ketoacidosis during the stress of acute illness such as COVID-19. ... Moreover, ketoacidosis is associated with hypercoagulability which could be especially dangerous in COVID-19, given that it has been causing thrombophilia with large-vessel occlusive strokes in young patients,” he said in an interview.
“One wonders how these risks were assessed by the authorities that approved the DARE-19 study,” said Dr. Leslie, who formerly worked for Bristol-Myers Squibb.
“How does the sponsor intend to secure informed consent given the risks? This is a DARE I would not take,” he said.
Asked to address these concerns, Dr. Kosiborod said in an interview that “the DARE-19 trial will assess both the efficacy and the safety of dapagliflozin in this patient population in a closely monitored environment of a rigorously designed randomized clinical trial. The trial protocol excludes patients with type 1 diabetes or at high risk for DKA.
“Furthermore, the protocol includes detailed specific instructions to ensure careful monitoring for DKA, including frequent assessments of acid-base status in the hospital setting. The safety data will be closely monitored by an independent data-monitoring committee,” he continued.
Dr. Kosiborod also pointed out that there is “no systematically collected information on the use of dapagliflozin or any other SGLT2 inhibitor in patients being treated for COVID-19, including the associated potential benefits, possible risks such as DKA, and the balance of these potential benefits and risks.”
DARE-19 design: Several outcomes will be examined
The DARE-19 trial is designed to enroll 900 adults with confirmed SARS-CoV-2 infection and oxygen saturation of 94% or greater.
Inclusion criteria include a medical history of hypertension, type 2 diabetes, atherosclerotic cardiovascular disease, heart failure, and/or stage 3-4 CKD. Exclusion criteria include current SGLT2 inhibitor treatment, type 1 diabetes, severe CKD, and severe COVID-19.
Dapagliflozin is approved in the EU for use in some patients with type 1 diabetes; this is not the case in the United States, although SGLT2 inhibitors in general are sometimes used off label in these patients.
Patients in DARE-19 will be randomized to 10 mg/day dapagliflozin or placebo for 30 days, in addition to standard care, in participating hospital. Primary outcomes are time to first occurrence of either death or new or worsened organ dysfunction, including respiratory decompensation, new or worsening heart failure, requirement for vasopressor therapy, ventricular tachycardia, and renal failure.
Secondary outcomes include a composite of time to death from any cause, time to new/worsened organ dysfunction, clinical status at day 30, and time to hospital discharge.
Rationale for the study
Irl B. Hirsch, MD, professor and diabetes treatment and teaching chair at the University of Washington, Seattle, said in an interview that he does see some logic to the trial.
Admitting that he doesn’t know much about “COVID-19 cardiomyopathy” – which would be one of the targets of dapagliflozin – other than it is quite common, he said that this, along with the potential renal benefits of dapagliflozin in the setting of COVID-19, make the study “intriguing.”
“Perhaps there is some rationale to it,” he said. However, “my concern is these sick COVID-19 patients are often acidemic, and besides the very complex acid-base challenges we see with intubated patients, these patients likely have combination lactic and ketoacidemia, the latter at least some from starvation.
“Still, if enough dextrose and insulin are provided to prevent ketoacid accumulation, my guess is it would do at least as well as hydroxychloroquine,” he said.
And Simon Heller, MD, professor of clinical diabetes at the University of Sheffield (England), said in an interview: “I think it is quite a brave study, mainly because of the increased risk of DKA.
“However, on the basis that these patients will be carefully monitored, the risk of DKA shouldn’t be great. I think it is important that patients with type 2 diabetes can participate whenever possible in such trials,” he said.
The estimated completion date for DARE-19 is December 2020.
Dr. Kosiborod has reported receiving grant support, honoraria, and/or research support from AstraZeneca, Boehringer Ingelheim, Sanofi, Amgen, Novo Nordisk, Merck, Eisai, Janssen, Bayer, GlaxoSmithKline, Glytec, Intarcia Therapeutics, Novartis, Applied Therapeutics, Amarin, and Eli Lilly. Dr. Leslie has reported owning stock in Bristol-Myers Squibb, Pfizer, and Lilly. Dr. Hirsch has reported consulting for Abbott Diabetes Care, Roche, and Bigfoot Biomedical, conducting research for Medtronic, and is a diabetes editor for UpToDate. Dr. Heller has received advisory or consultation fees from Lilly, Novo Nordisk, Takeda, MSD, and Becton Dickinson; has served as a speaker for AstraZeneca, Lilly, Novo Nordisk, Boehringer Ingelheim, and Takeda; and has received research support from Medtronic UK. He is on the advisory board for Medscape. Dr. Kar has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A just-launched study of the type 2 diabetes agent dapagliflozin (Farxiga, AstraZeneca) in patients with mild to moderate COVID-19 is raising eyebrows, given that several expert groups have advised that drugs in this class – the sodium-glucose cotransporter 2 (SGLT2) inhibitors – be stopped in all patients hospitalized with COVID-19 because of the increased risk for diabetic ketoacidosis (DKA).
The randomized, double-blind, placebo-controlled, phase 3 Dapagliflozin in Respiratory Failure in Patients With COVID-19 (DARE-19) study is sponsored by AstraZeneca and Saint Luke’s Mid America Heart Institute.
The trial will assess whether dapagliflozin reduces the risks of disease progression, clinical complications, and death because of COVID-19 in patients with type 2 diabetes, cardiovascular disease, and/or mild to moderate chronic kidney disease (CKD).
“Dapagliflozin has demonstrated cardio- and renal-protective benefits and improved outcomes in high-risk patients with type 2 diabetes, heart failure with reduced ejection fraction, and CKD,” said the principal investigator of DARE-19, Mikhail N. Kosiborod, MD, a cardiologist at Saint Luke’s Mid America Heart Institute, Kansas City, Mo.
And “patients with COVID-19 and underlying cardiometabolic disease appear to be at the highest risk of morbid complications,” he explained in an AstraZeneca statement.
“Through DARE-19, we hope to decrease the severity of illness, and prevent cardiovascular, respiratory, and kidney decompensation, which are common in patients with COVID-19,” Dr. Kosiborod continued.
However, advice to stop SGLT2 inhibitors in patients hospitalized with COVID-19 because of its associated DKA risk has come from several channels.
These include initial guidance from Diabetes UK; experts who spoke during an American Diabetes Association webinar; and most recently, an international panel of diabetes experts.
Some clinicians went so far as to say that they view the trial as potentially dangerous, while others said they could see some logic to it, as long as it is carefully managed.
“A dangerous proposition – a DARE I would not take”
Partha Kar, MD, of Portsmouth Hospitals NHS Trust and national clinical director of diabetes at NHS England, said in an interview: “It’s interesting to see [AstraZeneca] embark on a study with a particular class of drug whereby ... [in] the UK we have said that if you get sent to hospital with COVID-19 you should stop [SGLT2 inhibitors] immediately.”
It “sounds like a risky proposition to go ahead with, [and it] definitely made me raise an eyebrow,” he added.
Nephrologist Bruce R. Leslie, MD, of Seventh Doctor Consulting in Princeton, N.J., agreed with Dr. Kar.
“Giving SGLT2 inhibitors to patients in the DARE-19 study is a dangerous proposition because these drugs can induce ketoacidosis during the stress of acute illness such as COVID-19. ... Moreover, ketoacidosis is associated with hypercoagulability which could be especially dangerous in COVID-19, given that it has been causing thrombophilia with large-vessel occlusive strokes in young patients,” he said in an interview.
“One wonders how these risks were assessed by the authorities that approved the DARE-19 study,” said Dr. Leslie, who formerly worked for Bristol-Myers Squibb.
“How does the sponsor intend to secure informed consent given the risks? This is a DARE I would not take,” he said.
Asked to address these concerns, Dr. Kosiborod said in an interview that “the DARE-19 trial will assess both the efficacy and the safety of dapagliflozin in this patient population in a closely monitored environment of a rigorously designed randomized clinical trial. The trial protocol excludes patients with type 1 diabetes or at high risk for DKA.
“Furthermore, the protocol includes detailed specific instructions to ensure careful monitoring for DKA, including frequent assessments of acid-base status in the hospital setting. The safety data will be closely monitored by an independent data-monitoring committee,” he continued.
Dr. Kosiborod also pointed out that there is “no systematically collected information on the use of dapagliflozin or any other SGLT2 inhibitor in patients being treated for COVID-19, including the associated potential benefits, possible risks such as DKA, and the balance of these potential benefits and risks.”
DARE-19 design: Several outcomes will be examined
The DARE-19 trial is designed to enroll 900 adults with confirmed SARS-CoV-2 infection and oxygen saturation of 94% or greater.
Inclusion criteria include a medical history of hypertension, type 2 diabetes, atherosclerotic cardiovascular disease, heart failure, and/or stage 3-4 CKD. Exclusion criteria include current SGLT2 inhibitor treatment, type 1 diabetes, severe CKD, and severe COVID-19.
Dapagliflozin is approved in the EU for use in some patients with type 1 diabetes; this is not the case in the United States, although SGLT2 inhibitors in general are sometimes used off label in these patients.
Patients in DARE-19 will be randomized to 10 mg/day dapagliflozin or placebo for 30 days, in addition to standard care, in participating hospital. Primary outcomes are time to first occurrence of either death or new or worsened organ dysfunction, including respiratory decompensation, new or worsening heart failure, requirement for vasopressor therapy, ventricular tachycardia, and renal failure.
Secondary outcomes include a composite of time to death from any cause, time to new/worsened organ dysfunction, clinical status at day 30, and time to hospital discharge.
Rationale for the study
Irl B. Hirsch, MD, professor and diabetes treatment and teaching chair at the University of Washington, Seattle, said in an interview that he does see some logic to the trial.
Admitting that he doesn’t know much about “COVID-19 cardiomyopathy” – which would be one of the targets of dapagliflozin – other than it is quite common, he said that this, along with the potential renal benefits of dapagliflozin in the setting of COVID-19, make the study “intriguing.”
“Perhaps there is some rationale to it,” he said. However, “my concern is these sick COVID-19 patients are often acidemic, and besides the very complex acid-base challenges we see with intubated patients, these patients likely have combination lactic and ketoacidemia, the latter at least some from starvation.
“Still, if enough dextrose and insulin are provided to prevent ketoacid accumulation, my guess is it would do at least as well as hydroxychloroquine,” he said.
And Simon Heller, MD, professor of clinical diabetes at the University of Sheffield (England), said in an interview: “I think it is quite a brave study, mainly because of the increased risk of DKA.
“However, on the basis that these patients will be carefully monitored, the risk of DKA shouldn’t be great. I think it is important that patients with type 2 diabetes can participate whenever possible in such trials,” he said.
The estimated completion date for DARE-19 is December 2020.
Dr. Kosiborod has reported receiving grant support, honoraria, and/or research support from AstraZeneca, Boehringer Ingelheim, Sanofi, Amgen, Novo Nordisk, Merck, Eisai, Janssen, Bayer, GlaxoSmithKline, Glytec, Intarcia Therapeutics, Novartis, Applied Therapeutics, Amarin, and Eli Lilly. Dr. Leslie has reported owning stock in Bristol-Myers Squibb, Pfizer, and Lilly. Dr. Hirsch has reported consulting for Abbott Diabetes Care, Roche, and Bigfoot Biomedical, conducting research for Medtronic, and is a diabetes editor for UpToDate. Dr. Heller has received advisory or consultation fees from Lilly, Novo Nordisk, Takeda, MSD, and Becton Dickinson; has served as a speaker for AstraZeneca, Lilly, Novo Nordisk, Boehringer Ingelheim, and Takeda; and has received research support from Medtronic UK. He is on the advisory board for Medscape. Dr. Kar has reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Let’s learn from our experience
COVID-19 has revealed the worst and best of our country. Some have used it to validate their racism (my Korean-born son keeps me apprised of the Asian prejudice he has experienced) and a few leaders have made policy decisions based on ideology and not science, with disastrous consequences. That said, no world threat since those 13 days in October 1962 has demonstrated so decisively our interconnectedness. The best of our country has been demonstrated by our frontline health care workers, grocery clerks, people who deliver our packages, and volunteers who help feed our fellow citizens.
We are witnessing consequences of long-term health disparities that America continues to condone. Current hotspots are clustered in cities with high population density where people (usually minorities) lack ready access to health care and live with barriers to preventive care (poor nutritional options and a lack of sufficient primary care). We have underfunded our public health system and allowed politicians to ignore science. When testing was not prioritized initially, we lost the ability to isolate and trace index cases. If we want to honor those people who have died, let’s learn from our experience and change our priorities.
Private practices and health systems alike are being financially devastated. We are seeing massive numbers of people furloughed or laid off, as practices see drastic revenue loss. The transition to virtual health (video visits, remote patient monitoring) has been breath-taking with real implications about future needs for bricks and mortar. These changes in care delivery will be sustained in the future. Practice acquisitions have stopped, planned private equity exits are on hold, and the job market for graduating fellows will be challenging for the next 2 years. Now is a time for our GI societies to come together and find solutions for these problems so that our specialty can remain viable.
John I. Allen, MD, MBA, AGAF
Editor in Chief
COVID-19 has revealed the worst and best of our country. Some have used it to validate their racism (my Korean-born son keeps me apprised of the Asian prejudice he has experienced) and a few leaders have made policy decisions based on ideology and not science, with disastrous consequences. That said, no world threat since those 13 days in October 1962 has demonstrated so decisively our interconnectedness. The best of our country has been demonstrated by our frontline health care workers, grocery clerks, people who deliver our packages, and volunteers who help feed our fellow citizens.
We are witnessing consequences of long-term health disparities that America continues to condone. Current hotspots are clustered in cities with high population density where people (usually minorities) lack ready access to health care and live with barriers to preventive care (poor nutritional options and a lack of sufficient primary care). We have underfunded our public health system and allowed politicians to ignore science. When testing was not prioritized initially, we lost the ability to isolate and trace index cases. If we want to honor those people who have died, let’s learn from our experience and change our priorities.
Private practices and health systems alike are being financially devastated. We are seeing massive numbers of people furloughed or laid off, as practices see drastic revenue loss. The transition to virtual health (video visits, remote patient monitoring) has been breath-taking with real implications about future needs for bricks and mortar. These changes in care delivery will be sustained in the future. Practice acquisitions have stopped, planned private equity exits are on hold, and the job market for graduating fellows will be challenging for the next 2 years. Now is a time for our GI societies to come together and find solutions for these problems so that our specialty can remain viable.
John I. Allen, MD, MBA, AGAF
Editor in Chief
COVID-19 has revealed the worst and best of our country. Some have used it to validate their racism (my Korean-born son keeps me apprised of the Asian prejudice he has experienced) and a few leaders have made policy decisions based on ideology and not science, with disastrous consequences. That said, no world threat since those 13 days in October 1962 has demonstrated so decisively our interconnectedness. The best of our country has been demonstrated by our frontline health care workers, grocery clerks, people who deliver our packages, and volunteers who help feed our fellow citizens.
We are witnessing consequences of long-term health disparities that America continues to condone. Current hotspots are clustered in cities with high population density where people (usually minorities) lack ready access to health care and live with barriers to preventive care (poor nutritional options and a lack of sufficient primary care). We have underfunded our public health system and allowed politicians to ignore science. When testing was not prioritized initially, we lost the ability to isolate and trace index cases. If we want to honor those people who have died, let’s learn from our experience and change our priorities.
Private practices and health systems alike are being financially devastated. We are seeing massive numbers of people furloughed or laid off, as practices see drastic revenue loss. The transition to virtual health (video visits, remote patient monitoring) has been breath-taking with real implications about future needs for bricks and mortar. These changes in care delivery will be sustained in the future. Practice acquisitions have stopped, planned private equity exits are on hold, and the job market for graduating fellows will be challenging for the next 2 years. Now is a time for our GI societies to come together and find solutions for these problems so that our specialty can remain viable.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Remaining connected
Dear colleagues,
We bring you the spring edition of The New Gastroenterologist amid a backdrop of uncertainty in the setting of the novel coronavirus disease 2019 (COVID-19) pandemic. As physicians, we are poised to view this unprecedented situation in modern medicine through a unique lens. At the time of this writing, we are experiencing significant interruptions to our work as gastroenterologists coupled with the possibility of reassignments in order to care for COVID-19 patients to meet the demand of the precipitous rise in cases. Weighing these responsibilities, along with the heightened concern about the threat of exposure to ourselves and our families, is a formidable challenge, but one that we can navigate together.
My sincere hope is that this quarter’s newsletter can provide, at the very least, a brief reprieve from some of these constant stressors. It is during times like this that remaining connected to our colleagues through digital platforms and publications such as The New Gastroenterologist remains of utmost importance.
That being said, I felt it was prudent to first address some common concerns regarding the COVID-19 pandemic, specifically, its implications within gastroenterology. In conjunction with Krishna Rao (University of Michigan), a specialist in infectious diseases, we attempt to shed some light on what is a rapidly evolving situation. For more resources from the American Gastroenterological Association (AGA) on up-to-date clinical guidance and research, you can also visit https://www.gastro.org/practice-guidance/practice-updates/covid-19.
Moving on to our “In Focus” feature, Thangam Ventakesan and Harrison Mooers (Medical College of Wisconsin) provide a comprehensive overview of cyclic vomiting syndrome. This is a valuable read as cyclic vomiting syndrome has been gaining increased recognition among adults, and Dr. Ventakesan and Dr. Mooers elucidate a thorough approach to the diagnosis and treatment of this disorder.
A facet of endoscopy that is extremely important, but frequently overlooked, is ergonomics. Manish Singla and Jared Magee (Walter Reed National Military Medical Center) compile a high-yield list of recommendations on the best practices to preserve our own safety and health as endoscopists.
We continue our medical ethics series with Jennifer Wang and Andrew Aronsohn (University of Chicago) who offer a thought-provoking discussion on the role of early liver transplantation for alcoholic hepatitis, including an analysis of the medical, psychosocial, and ethical considerations.
Also in this issue, Animesh Jain (University of North Carolina) gives us some excellent financial advice on student loan management, outlining a basic strategy of repayment with clear explanations of the available options including refinancing, public service loan forgiveness, and income-driven repayment.
Dilhana Badurdeen (Johns Hopkins), Aline Charabaty Pishvaian (Sibley Memorial Hospital), Miguel Malespin (University of South Florida), Ibironke Oduyebo (Midatlantic Permanente Medical Group), and Sandra Quezada (University of Maryland) give us an in-depth summary of the efforts of the AGA’s Diversity Committee, including publications, events, and future initiatives.
This quarter’s DHPA Private Practice Perspectives series features Paul Berggreen (Arizona Digestive Health), who reviews the advantages and disadvantages of pathology lab ownership as a gastroenterologist. Lastly, Sarah Ordway, Dawn Torres, Manish Singla, and Adam Tritsch (Walter Reed National Military Medical Center) broach the issue of fellowship burnout by providing guidance on how to identify signs and those at risk in addition to providing tangible solutions that any fellowship can incorporate.
Although the cancellation of the upcoming DDW meetings in Chicago is a disappointment, I hope that we can all take this time to prioritize the well-being of ourselves and our communities until we meet again.
As always, if you have interest in contributing or have ideas for future TNG topics, please contact me ([email protected]), or Ryan Farrell ([email protected]), managing editor of TNG.
Best wishes to stay safe and healthy.
Vijaya L. Rao, MD
Editor in Chief
Dr. Rao is assistant professor of medicine, University of Chicago, section of gastroenterology, hepatology & nutrition.
Dear colleagues,
We bring you the spring edition of The New Gastroenterologist amid a backdrop of uncertainty in the setting of the novel coronavirus disease 2019 (COVID-19) pandemic. As physicians, we are poised to view this unprecedented situation in modern medicine through a unique lens. At the time of this writing, we are experiencing significant interruptions to our work as gastroenterologists coupled with the possibility of reassignments in order to care for COVID-19 patients to meet the demand of the precipitous rise in cases. Weighing these responsibilities, along with the heightened concern about the threat of exposure to ourselves and our families, is a formidable challenge, but one that we can navigate together.
My sincere hope is that this quarter’s newsletter can provide, at the very least, a brief reprieve from some of these constant stressors. It is during times like this that remaining connected to our colleagues through digital platforms and publications such as The New Gastroenterologist remains of utmost importance.
That being said, I felt it was prudent to first address some common concerns regarding the COVID-19 pandemic, specifically, its implications within gastroenterology. In conjunction with Krishna Rao (University of Michigan), a specialist in infectious diseases, we attempt to shed some light on what is a rapidly evolving situation. For more resources from the American Gastroenterological Association (AGA) on up-to-date clinical guidance and research, you can also visit https://www.gastro.org/practice-guidance/practice-updates/covid-19.
Moving on to our “In Focus” feature, Thangam Ventakesan and Harrison Mooers (Medical College of Wisconsin) provide a comprehensive overview of cyclic vomiting syndrome. This is a valuable read as cyclic vomiting syndrome has been gaining increased recognition among adults, and Dr. Ventakesan and Dr. Mooers elucidate a thorough approach to the diagnosis and treatment of this disorder.
A facet of endoscopy that is extremely important, but frequently overlooked, is ergonomics. Manish Singla and Jared Magee (Walter Reed National Military Medical Center) compile a high-yield list of recommendations on the best practices to preserve our own safety and health as endoscopists.
We continue our medical ethics series with Jennifer Wang and Andrew Aronsohn (University of Chicago) who offer a thought-provoking discussion on the role of early liver transplantation for alcoholic hepatitis, including an analysis of the medical, psychosocial, and ethical considerations.
Also in this issue, Animesh Jain (University of North Carolina) gives us some excellent financial advice on student loan management, outlining a basic strategy of repayment with clear explanations of the available options including refinancing, public service loan forgiveness, and income-driven repayment.
Dilhana Badurdeen (Johns Hopkins), Aline Charabaty Pishvaian (Sibley Memorial Hospital), Miguel Malespin (University of South Florida), Ibironke Oduyebo (Midatlantic Permanente Medical Group), and Sandra Quezada (University of Maryland) give us an in-depth summary of the efforts of the AGA’s Diversity Committee, including publications, events, and future initiatives.
This quarter’s DHPA Private Practice Perspectives series features Paul Berggreen (Arizona Digestive Health), who reviews the advantages and disadvantages of pathology lab ownership as a gastroenterologist. Lastly, Sarah Ordway, Dawn Torres, Manish Singla, and Adam Tritsch (Walter Reed National Military Medical Center) broach the issue of fellowship burnout by providing guidance on how to identify signs and those at risk in addition to providing tangible solutions that any fellowship can incorporate.
Although the cancellation of the upcoming DDW meetings in Chicago is a disappointment, I hope that we can all take this time to prioritize the well-being of ourselves and our communities until we meet again.
As always, if you have interest in contributing or have ideas for future TNG topics, please contact me ([email protected]), or Ryan Farrell ([email protected]), managing editor of TNG.
Best wishes to stay safe and healthy.
Vijaya L. Rao, MD
Editor in Chief
Dr. Rao is assistant professor of medicine, University of Chicago, section of gastroenterology, hepatology & nutrition.
Dear colleagues,
We bring you the spring edition of The New Gastroenterologist amid a backdrop of uncertainty in the setting of the novel coronavirus disease 2019 (COVID-19) pandemic. As physicians, we are poised to view this unprecedented situation in modern medicine through a unique lens. At the time of this writing, we are experiencing significant interruptions to our work as gastroenterologists coupled with the possibility of reassignments in order to care for COVID-19 patients to meet the demand of the precipitous rise in cases. Weighing these responsibilities, along with the heightened concern about the threat of exposure to ourselves and our families, is a formidable challenge, but one that we can navigate together.
My sincere hope is that this quarter’s newsletter can provide, at the very least, a brief reprieve from some of these constant stressors. It is during times like this that remaining connected to our colleagues through digital platforms and publications such as The New Gastroenterologist remains of utmost importance.
That being said, I felt it was prudent to first address some common concerns regarding the COVID-19 pandemic, specifically, its implications within gastroenterology. In conjunction with Krishna Rao (University of Michigan), a specialist in infectious diseases, we attempt to shed some light on what is a rapidly evolving situation. For more resources from the American Gastroenterological Association (AGA) on up-to-date clinical guidance and research, you can also visit https://www.gastro.org/practice-guidance/practice-updates/covid-19.
Moving on to our “In Focus” feature, Thangam Ventakesan and Harrison Mooers (Medical College of Wisconsin) provide a comprehensive overview of cyclic vomiting syndrome. This is a valuable read as cyclic vomiting syndrome has been gaining increased recognition among adults, and Dr. Ventakesan and Dr. Mooers elucidate a thorough approach to the diagnosis and treatment of this disorder.
A facet of endoscopy that is extremely important, but frequently overlooked, is ergonomics. Manish Singla and Jared Magee (Walter Reed National Military Medical Center) compile a high-yield list of recommendations on the best practices to preserve our own safety and health as endoscopists.
We continue our medical ethics series with Jennifer Wang and Andrew Aronsohn (University of Chicago) who offer a thought-provoking discussion on the role of early liver transplantation for alcoholic hepatitis, including an analysis of the medical, psychosocial, and ethical considerations.
Also in this issue, Animesh Jain (University of North Carolina) gives us some excellent financial advice on student loan management, outlining a basic strategy of repayment with clear explanations of the available options including refinancing, public service loan forgiveness, and income-driven repayment.
Dilhana Badurdeen (Johns Hopkins), Aline Charabaty Pishvaian (Sibley Memorial Hospital), Miguel Malespin (University of South Florida), Ibironke Oduyebo (Midatlantic Permanente Medical Group), and Sandra Quezada (University of Maryland) give us an in-depth summary of the efforts of the AGA’s Diversity Committee, including publications, events, and future initiatives.
This quarter’s DHPA Private Practice Perspectives series features Paul Berggreen (Arizona Digestive Health), who reviews the advantages and disadvantages of pathology lab ownership as a gastroenterologist. Lastly, Sarah Ordway, Dawn Torres, Manish Singla, and Adam Tritsch (Walter Reed National Military Medical Center) broach the issue of fellowship burnout by providing guidance on how to identify signs and those at risk in addition to providing tangible solutions that any fellowship can incorporate.
Although the cancellation of the upcoming DDW meetings in Chicago is a disappointment, I hope that we can all take this time to prioritize the well-being of ourselves and our communities until we meet again.
As always, if you have interest in contributing or have ideas for future TNG topics, please contact me ([email protected]), or Ryan Farrell ([email protected]), managing editor of TNG.
Best wishes to stay safe and healthy.
Vijaya L. Rao, MD
Editor in Chief
Dr. Rao is assistant professor of medicine, University of Chicago, section of gastroenterology, hepatology & nutrition.
Cyclic vomiting syndrome: A GI primer
Introduction
Cyclic vomiting syndrome (CVS) is a chronic disorder of gut-brain interaction (DGBI) and is characterized by recurrent episodes of severe nausea, vomiting, and often, abdominal pain. Patients are usually asymptomatic in between episodes.1 CVS was considered a pediatric disease but is now known to be as common in adults. The prevalence of CVS in adults was 2% in a recent population-based study.2 Patients are predominantly white. Both males and females are affected with some studies showing a female preponderance. The mean age of onset is 5 years in children and 35 years in adults.3
The etiology of CVS is not known, but various hypotheses have been proposed. Zaki et al. showed that two mitochondrial DNA polymorphisms 16519T and 3010A were associated with a 17-fold increased odds of having CVS in children.4 These polymorphisms were not associated with CVS in adults.5 Alterations in the brain-gut axis also have been shown in CVS. Functional neuroimaging studies demonstrate that patients with CVS displayed increased connectivity between insula and salience networks with concomitant decrease in connectivity to somatosensory networks.6 Recent data also indicate that the endocannabinoid system (ECS) and the hypothalamic-pituitary-adrenal axis are implicated in CVS with an increase in serum endocannabinoid concentration during an episode.7 The same study also showed a significant increase in salivary cortisol in CVS patients who used cannabis. Further, single nucleotide polymorphisms (SNPs) in the gene that encodes for the cannabinoid receptor type 1 (CB1R) are implicated in CVS.8 The CB1R is part of the ECS and is densely expressed in brain areas involved in emesis, such as the dorsal vagal complex consisting of the area postrema (AP), nucleus of the solitary tract (NTS), and also the dorsal motor nucleus of the vagus.9 Wasilewski et al. showed an increased risk of CVS among individuals with AG and GG genotypes of CNR1 rs806380 (P less than .01), whereas the CC genotype of CNR1 rs806368 was associated with a decreased risk of CVS (P less than .05).8 CB1R agonists – endocannabinoids and tetrahydrocannabinol (THC) – have acute antiemetic and anxiolytic effects.9-11 The apparent paradoxical effects of cannabis in this patient population are yet to be explained and need further study.
Diagnosis and clinical features of CVS
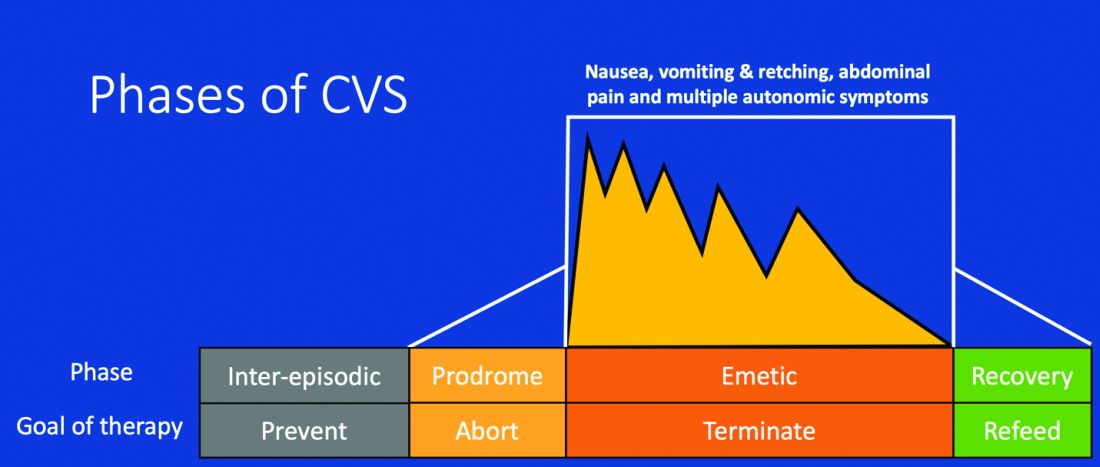
Figure 1: Phases of Cyclic Vomiting Syndrome12
Adapted from Fleisher DR, Gornowicz B, Adams K, Burch R, Feldman EJ. Cyclic Vomiting Syndrome in 41 adults: The illness, the patients, and problems of management. BMC Med 2005;3:20. This work is licensed under the Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use, distribution, modification, and reproduction in any medium.
CVS consists of four phases which include the a) prodromal phase, b) the episodic phase, c) recovery phase, and d) the interepisodic phase; and was first described by David Fleisher.12 The phases of CVS are important for clinicians and patients alike as they have therapeutic implications. The administration of abortive medications during a prodrome can terminate an episode. The phases of CVS are shown above.
Most patients (~ 93%) have a prodromal phase. Symptoms during this phase can include nausea, abdominal pain, diaphoresis, fatigue, weakness, hot flashes, chills, shivering, increased thirst, loss of appetite, burping, lightheadedness, and paresthesia.13 Some patients report a sense of impending doom and many have symptoms consistent with panic. If untreated, this progresses to the emetic phase and patients have unrelenting nausea, retching, vomiting, and other symptoms. During an episode, patients may vomit up to 20 times per hour and the episode may last several hours to days. During this phase, patients are sometimes described as being in a “conscious coma” and exhibit lethargy, listlessness, withdrawal, and sometimes disorientation.14,15 The emetic phase is followed by the recovery phase, during which symptoms subside and patients are able to resume oral intake. Patients are usually asymptomatic between episodes but ~ 30% can have interepisodic nausea and dyspepsia. In some patients, episodes become progressively longer and the interepisodic phase is considerably shortened and patients have a “coalescence of symptoms.”12 It is important to elicit a thorough history in all patients with vomiting in order to make an accurate diagnosis of CVS since coalescence of symptoms only occurs over a period of time. Episodes often are triggered by psychological stress, both positive and negative. Common triggers can include positive events such as birthdays, holidays, and negative ones like examinations, the death of a loved one, etc. Sleep deprivation and physical exhaustion also can trigger an episode.12
CVS remains a clinical diagnosis since there are no biomarkers. While there is a lack of data on the optimal work-up in these patients, experts recommend an upper endoscopy or upper GI series in order to rule out alternative gastric and intestinal pathology (e.g., malrotation with volvulus).16 Of note, a gastric-emptying study is not recommended as part of the routine work-up as per recent guidelines because of the poor specificity of this test in establishing a diagnosis of CVS.16 Biochemical testing including a complete blood count, serum electrolytes, serum glucose, liver panel, and urinalysis is also warranted. Any additional testing is indicated when clinical features suggest an alternative diagnosis. For instance, neurologic symptoms might warrant a cranial MRI to exclude an intracerebral tumor or other lesions of the brain.

The severity and unpredictable nature of symptoms makes it difficult for some patients to attend school or work; one study found that 32% of patients with CVS were completely disabled.12 Despite increasing awareness of this disorder, patients often are misdiagnosed. The prevalence of CVS in an outpatient gastroenterology clinic in the United Kingdom was 11% and was markedly underdiagnosed in the community.17 Only 5% of patients who were subsequently diagnosed with CVS were initially diagnosed accurately by their referring physician despite meeting criteria for the disorder.17 A subset of patients with CVS even undergo futile surgeries.13 Fleisher et al. noted that 30% of a 41-patient cohort underwent cholecystectomy for CVS symptoms without any improvement in disease.12 Prompt diagnosis and appropriate therapy is essential to improve patient outcomes and improve quality of life.
CVS is associated with various comorbidities such as migraine, anxiety, depression and dysautonomia, which can further impair quality of life.18,19 Approximately 70% of CVS patients report a personal or family history of migraine. Anxiety and depression affects nearly half of patients with CVS.13 Cannabis use is significantly more prevalent among patients with CVS than patients without CVS.20
Role of cannabis in CVS
The role of cannabis in the pathogenesis of symptoms in CVS is controversial. While cannabis has antiemetic properties, there is a strong link between its use and CVS. The use of cannabis has increased over the past decade with increasing legalization.21 Several studies have shown that 40%-80% of patients with CVS use cannabis.22,23 Following this, cannabinoid hyperemesis syndrome (CHS) was coined as a separate entity based on this statistical association, though there are no data to support the notion that cannabis causes vomiting.24,25 CHS has clinical features that are indistinguishable from CVS except for the chronic heavy cannabis use. A peculiar bathing behavior called “compulsive hot-water bathing” has been described and was thought to be pathognomonic of cannabis use.26 During an episode, patients will take multiple hot showers/baths, which temporarily alleviate their symptoms. Many patients even report running out of hot water and sometimes check into a hotel for a continuous supply of hot water. A small number of patients may sustain burns from the hot-water bathing. However, studies show that this hot-water bathing behavior also is seen in about 50% of patents with CVS who do not use cannabis.22

CHS is now defined by Rome IV criteria, which include episodes of nausea and vomiting similar to CVS preceded by chronic, heavy cannabis use. Patients must have complete resolution of symptoms following cessation.1 A recent systematic review of 376 cases of purported CHS showed that only 59 (15.7%) met Rome IV criteria for this disorder.27 This is because of considerable heterogeneity in how the diagnosis of CHS was made and the lack of standard diagnostic criteria at the time. Some cases of CHS were diagnosed merely based on an association of vomiting, hot-water bathing, and cannabis use.28 Only a minority of patients (71,19%) had a duration of follow-up more than 4 weeks, which would make it impossible to establish a diagnosis of CHS. A period of at least a year or a duration of time that spans at least three episodes is generally recommended to determine if abstinence from cannabis causes a true resolution of symptoms.27 Whether CHS is a separate entity or a subtype of CVS remains to be determined. The paradoxical effects of cannabis may happen because of the use of highly potent cannabis products that are currently in use. A complete discussion of the role of cannabis in CVS is beyond the scope of this article, and the reader is referred to a recent systematic review and discussion.27
Treatment
CVS should be treated based on a biopsychosocial model with a multidisciplinary team that includes a gastroenterologist with knowledge of CVS, primary care physician, psychologist, psychiatrist, and sleep specialist if needed.16 Initiating prophylactic treatment is based on the severity of disease. An algorithm for the treatment of CVS based on severity of symptoms is shown below.
Figure 2. Adapted and reprinted by permission from the Licensor: Springer Nature, Current Treatment Options in Gastroenterology, Bhandari S, Venkatesan T. Novel Treatments for Cyclic Vomiting Syndrome: Beyond Ondansetron and Amitriptyline, 14:495-506, Copyright 2016.
Patients who have mild disease (defined as fewer than four episodes/year, episodes lasting up to 2 days, quick recovery from episodes, or episodes not requiring ED care or hospitalization) are usually prescribed abortive medications.16 These medications are best administered during the prodromal phase and can prevent progression to the emetic phase. Medications used for aborting episodes include sumatriptan (20 mg intranasal or 6 mg subcutaneous), ondansetron (8 mg sublingual), and diphenhydramine (25-50 mg).30,31 This combination can help abort symptoms and potentially avoid ED visits or hospitalizations. Patients with moderate-to-severe CVS are offered prophylactic therapy in addition to abortive therapy.16
Recent guidelines recommend tricyclic antidepressants (TCAs) as the first-line agent in the prophylaxis of CVS episodes. Data from 14 studies determined that 70% (413/600) of patients responded partially or completely to TCAs.16 An open-label study of 46 patients by Hejazi et al. noted a decline in the number of CVS episodes from 17 to 3, in the duration of a CVS episode from 6 to 2 days, and in the number of ED visits/ hospitalizations from 15 to 3.3.32Amitriptyline should be started at 25 mg at night and titrated up by 10-25 mg each week to minimize emergence of side effects. The mean effective dose is 75-100 mg or 1.0-1.5 mg/kg. An EKG should be checked at baseline and during titration to monitor the QT interval. Unfortunately, side effects from TCAs are quite common and include cognitive impairment, drowsiness, dryness of mouth, weight gain, constipation, and mood changes, which may warrant dose reduction or discontinuation. Antiepileptics such as topiramate, mitochondrial supplements such as Coenzyme Q10 and riboflavin are alternative prophylactic agents in CVS.33 Aprepitant, a newer NK1 receptor antagonist has been found to be effective in refractory CVS.34 In addition to pharmacotherapy, addressing comorbid conditions such as anxiety and depression and counseling patients to abstain from heavy cannabis use is also important to achieve good health care outcomes.
In summary, CVS is a common, chronic functional GI disorder with episodic nausea, vomiting, and often, abdominal pain. Symptoms can be disabling, and prompt diagnosis and therapy is important. CVS is associated with multiple comorbid conditions such as migraine, anxiety and depression, and a biopsychosocial model of care is essential. Medications such as amitriptyline are effective in the prophylaxis of CVS, but side effects hamper their use. Recent recommendations for management of CVS have been published.16 Cannabis is frequently used by patients for symptom relief but use of high potency products may cause worsening of symptoms or unmask symptoms in genetically predisposed individuals.23 Studies to elucidate the pathophysiology of CVS should help in the development of better therapies.
Dr. Mooers is PGY-2, an internal medicine resident in the department of medicine, Medical College of Wisconsin, Milwaukee; Dr. Venkatesan is professor of medicine, division of gastroenterology and hepatology, department of medicine, Medical College of Wisconsin, Milwaukee. The authors have no conflicts to disclose.
References
1. Stanghellini V et al. Gastroenterology. 2016;150:1380-92.
2. Aziz I et al. Clin Gastroenterol Hepatol. 2019 Apr;17(5):878-86.
3. Kovacic K et al. Curr Gastroenterol Rep. 2018;20(10):46.
4. Zaki EA et al. Cephalalgia. 2009;29:719-28.
5. Venkatesan T et al. BMC Gastroenterol. 2014;14:181.
6. Ellingsen DM et al. Neurogastroenterol Motil. 2017;29 (6)e13004 9.
7. Venkatesan T et al. Neurogastroenterol Motil. 2016;28:1409-18.
8. Wasilewski A et al. Am J Gastroenterol. 2017;112:933-9.
9. van Sickle MD et al. Am J Physiol Gastrointest Liver Physiol 2003;285:G566-76.
10. Parker LA et al. Br J Pharmacol. 2011;163:1411-22.
11. van Sickle MD et al. Gastroenterology. 2001;121:767-74.
12. Fleisher DR et al. BMC Med. 2005;3:20.
13. Kumar N et al. BMC Gastroenterol. 2012;12:52.
14. Li BU et al. J Pediatr Gastroenterol Nutr. 2008;47:379-93.
15. Bhandari S et al. Clin Auton Res. 2018 Apr;28(2):203-9.
16. Venkatesan T et al. Neurogastroenterol Motil. 2019;31 Suppl 2:e13604. doi: 10.1111/nmo.13604.
17. Sagar RC et al. Neurogastroenterol Motil. 2018;30. doi: 10.1111/nmo.13174.
18. Taranukha T et al. Neurogastroenterol Motil. 2018 Apr;30(4):e13245. doi: 10.1111/nmo.13245.
19. Bhandari S and Venkatesan T. Dig Dis Sci. 2017;62:2035-44.
20. Choung RS et al. Neurogastroenterol Motil. 2012;24:20-6, e21. doi: 10.1111/j.1365-2982.2011.01791.x.
21. Bhandari S et al. Intern Med J. 2019 May;49(5):649-55.
22. Venkatesan T et al. Exp Brain Res. 2014; 232:2563-70.
23. Venkatesan T et al. Clin Gastroenterol Hepatol. 2019 Jul 25. doi: 10.1016/j.cgh.2019.07.039.
24. Simonetto DA et al. Mayo Clin Proc. 2012;87:114-9.
25. Wallace EA et al. South Med J. 2011;104:659-64.
26. Allen JH et al. Gut. 2004;53:1566-70.
27. Venkatesan T et al. Neurogastroenterol Motil. 2019;31 Suppl 2:e13606. doi: 10.1111/nmo.13606.
28. Habboushe J et al. Basic Clin Pharmacol Toxicol. 2018;122:660-2.
29. Bhandari S and Venkatesan T. Curr Treat Options Gastroenterol. 2016;14:495-506.
30. Hikita T et al. Cephalalgia. 2011;31:504-7.
31. Fuseau E et al. Clin Pharmacokinet 2002;41:801-11.
32. Hejazi RA et al. J Clin Gastroenterol. 2010;44:18-21.
33. Sezer OB and Sezer T. J Neurogastroenterol Motil. 2016;22:656-60.
34. Cristofori F et al. Aliment Pharmacol Ther. 2014;40:309-17.
Introduction
Cyclic vomiting syndrome (CVS) is a chronic disorder of gut-brain interaction (DGBI) and is characterized by recurrent episodes of severe nausea, vomiting, and often, abdominal pain. Patients are usually asymptomatic in between episodes.1 CVS was considered a pediatric disease but is now known to be as common in adults. The prevalence of CVS in adults was 2% in a recent population-based study.2 Patients are predominantly white. Both males and females are affected with some studies showing a female preponderance. The mean age of onset is 5 years in children and 35 years in adults.3
The etiology of CVS is not known, but various hypotheses have been proposed. Zaki et al. showed that two mitochondrial DNA polymorphisms 16519T and 3010A were associated with a 17-fold increased odds of having CVS in children.4 These polymorphisms were not associated with CVS in adults.5 Alterations in the brain-gut axis also have been shown in CVS. Functional neuroimaging studies demonstrate that patients with CVS displayed increased connectivity between insula and salience networks with concomitant decrease in connectivity to somatosensory networks.6 Recent data also indicate that the endocannabinoid system (ECS) and the hypothalamic-pituitary-adrenal axis are implicated in CVS with an increase in serum endocannabinoid concentration during an episode.7 The same study also showed a significant increase in salivary cortisol in CVS patients who used cannabis. Further, single nucleotide polymorphisms (SNPs) in the gene that encodes for the cannabinoid receptor type 1 (CB1R) are implicated in CVS.8 The CB1R is part of the ECS and is densely expressed in brain areas involved in emesis, such as the dorsal vagal complex consisting of the area postrema (AP), nucleus of the solitary tract (NTS), and also the dorsal motor nucleus of the vagus.9 Wasilewski et al. showed an increased risk of CVS among individuals with AG and GG genotypes of CNR1 rs806380 (P less than .01), whereas the CC genotype of CNR1 rs806368 was associated with a decreased risk of CVS (P less than .05).8 CB1R agonists – endocannabinoids and tetrahydrocannabinol (THC) – have acute antiemetic and anxiolytic effects.9-11 The apparent paradoxical effects of cannabis in this patient population are yet to be explained and need further study.
Diagnosis and clinical features of CVS

Figure 1: Phases of Cyclic Vomiting Syndrome12
Adapted from Fleisher DR, Gornowicz B, Adams K, Burch R, Feldman EJ. Cyclic Vomiting Syndrome in 41 adults: The illness, the patients, and problems of management. BMC Med 2005;3:20. This work is licensed under the Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use, distribution, modification, and reproduction in any medium.
CVS consists of four phases which include the a) prodromal phase, b) the episodic phase, c) recovery phase, and d) the interepisodic phase; and was first described by David Fleisher.12 The phases of CVS are important for clinicians and patients alike as they have therapeutic implications. The administration of abortive medications during a prodrome can terminate an episode. The phases of CVS are shown above.
Most patients (~ 93%) have a prodromal phase. Symptoms during this phase can include nausea, abdominal pain, diaphoresis, fatigue, weakness, hot flashes, chills, shivering, increased thirst, loss of appetite, burping, lightheadedness, and paresthesia.13 Some patients report a sense of impending doom and many have symptoms consistent with panic. If untreated, this progresses to the emetic phase and patients have unrelenting nausea, retching, vomiting, and other symptoms. During an episode, patients may vomit up to 20 times per hour and the episode may last several hours to days. During this phase, patients are sometimes described as being in a “conscious coma” and exhibit lethargy, listlessness, withdrawal, and sometimes disorientation.14,15 The emetic phase is followed by the recovery phase, during which symptoms subside and patients are able to resume oral intake. Patients are usually asymptomatic between episodes but ~ 30% can have interepisodic nausea and dyspepsia. In some patients, episodes become progressively longer and the interepisodic phase is considerably shortened and patients have a “coalescence of symptoms.”12 It is important to elicit a thorough history in all patients with vomiting in order to make an accurate diagnosis of CVS since coalescence of symptoms only occurs over a period of time. Episodes often are triggered by psychological stress, both positive and negative. Common triggers can include positive events such as birthdays, holidays, and negative ones like examinations, the death of a loved one, etc. Sleep deprivation and physical exhaustion also can trigger an episode.12
CVS remains a clinical diagnosis since there are no biomarkers. While there is a lack of data on the optimal work-up in these patients, experts recommend an upper endoscopy or upper GI series in order to rule out alternative gastric and intestinal pathology (e.g., malrotation with volvulus).16 Of note, a gastric-emptying study is not recommended as part of the routine work-up as per recent guidelines because of the poor specificity of this test in establishing a diagnosis of CVS.16 Biochemical testing including a complete blood count, serum electrolytes, serum glucose, liver panel, and urinalysis is also warranted. Any additional testing is indicated when clinical features suggest an alternative diagnosis. For instance, neurologic symptoms might warrant a cranial MRI to exclude an intracerebral tumor or other lesions of the brain.

The severity and unpredictable nature of symptoms makes it difficult for some patients to attend school or work; one study found that 32% of patients with CVS were completely disabled.12 Despite increasing awareness of this disorder, patients often are misdiagnosed. The prevalence of CVS in an outpatient gastroenterology clinic in the United Kingdom was 11% and was markedly underdiagnosed in the community.17 Only 5% of patients who were subsequently diagnosed with CVS were initially diagnosed accurately by their referring physician despite meeting criteria for the disorder.17 A subset of patients with CVS even undergo futile surgeries.13 Fleisher et al. noted that 30% of a 41-patient cohort underwent cholecystectomy for CVS symptoms without any improvement in disease.12 Prompt diagnosis and appropriate therapy is essential to improve patient outcomes and improve quality of life.
CVS is associated with various comorbidities such as migraine, anxiety, depression and dysautonomia, which can further impair quality of life.18,19 Approximately 70% of CVS patients report a personal or family history of migraine. Anxiety and depression affects nearly half of patients with CVS.13 Cannabis use is significantly more prevalent among patients with CVS than patients without CVS.20
Role of cannabis in CVS
The role of cannabis in the pathogenesis of symptoms in CVS is controversial. While cannabis has antiemetic properties, there is a strong link between its use and CVS. The use of cannabis has increased over the past decade with increasing legalization.21 Several studies have shown that 40%-80% of patients with CVS use cannabis.22,23 Following this, cannabinoid hyperemesis syndrome (CHS) was coined as a separate entity based on this statistical association, though there are no data to support the notion that cannabis causes vomiting.24,25 CHS has clinical features that are indistinguishable from CVS except for the chronic heavy cannabis use. A peculiar bathing behavior called “compulsive hot-water bathing” has been described and was thought to be pathognomonic of cannabis use.26 During an episode, patients will take multiple hot showers/baths, which temporarily alleviate their symptoms. Many patients even report running out of hot water and sometimes check into a hotel for a continuous supply of hot water. A small number of patients may sustain burns from the hot-water bathing. However, studies show that this hot-water bathing behavior also is seen in about 50% of patents with CVS who do not use cannabis.22

CHS is now defined by Rome IV criteria, which include episodes of nausea and vomiting similar to CVS preceded by chronic, heavy cannabis use. Patients must have complete resolution of symptoms following cessation.1 A recent systematic review of 376 cases of purported CHS showed that only 59 (15.7%) met Rome IV criteria for this disorder.27 This is because of considerable heterogeneity in how the diagnosis of CHS was made and the lack of standard diagnostic criteria at the time. Some cases of CHS were diagnosed merely based on an association of vomiting, hot-water bathing, and cannabis use.28 Only a minority of patients (71,19%) had a duration of follow-up more than 4 weeks, which would make it impossible to establish a diagnosis of CHS. A period of at least a year or a duration of time that spans at least three episodes is generally recommended to determine if abstinence from cannabis causes a true resolution of symptoms.27 Whether CHS is a separate entity or a subtype of CVS remains to be determined. The paradoxical effects of cannabis may happen because of the use of highly potent cannabis products that are currently in use. A complete discussion of the role of cannabis in CVS is beyond the scope of this article, and the reader is referred to a recent systematic review and discussion.27
Treatment
CVS should be treated based on a biopsychosocial model with a multidisciplinary team that includes a gastroenterologist with knowledge of CVS, primary care physician, psychologist, psychiatrist, and sleep specialist if needed.16 Initiating prophylactic treatment is based on the severity of disease. An algorithm for the treatment of CVS based on severity of symptoms is shown below.
Figure 2. Adapted and reprinted by permission from the Licensor: Springer Nature, Current Treatment Options in Gastroenterology, Bhandari S, Venkatesan T. Novel Treatments for Cyclic Vomiting Syndrome: Beyond Ondansetron and Amitriptyline, 14:495-506, Copyright 2016.
Patients who have mild disease (defined as fewer than four episodes/year, episodes lasting up to 2 days, quick recovery from episodes, or episodes not requiring ED care or hospitalization) are usually prescribed abortive medications.16 These medications are best administered during the prodromal phase and can prevent progression to the emetic phase. Medications used for aborting episodes include sumatriptan (20 mg intranasal or 6 mg subcutaneous), ondansetron (8 mg sublingual), and diphenhydramine (25-50 mg).30,31 This combination can help abort symptoms and potentially avoid ED visits or hospitalizations. Patients with moderate-to-severe CVS are offered prophylactic therapy in addition to abortive therapy.16
Recent guidelines recommend tricyclic antidepressants (TCAs) as the first-line agent in the prophylaxis of CVS episodes. Data from 14 studies determined that 70% (413/600) of patients responded partially or completely to TCAs.16 An open-label study of 46 patients by Hejazi et al. noted a decline in the number of CVS episodes from 17 to 3, in the duration of a CVS episode from 6 to 2 days, and in the number of ED visits/ hospitalizations from 15 to 3.3.32Amitriptyline should be started at 25 mg at night and titrated up by 10-25 mg each week to minimize emergence of side effects. The mean effective dose is 75-100 mg or 1.0-1.5 mg/kg. An EKG should be checked at baseline and during titration to monitor the QT interval. Unfortunately, side effects from TCAs are quite common and include cognitive impairment, drowsiness, dryness of mouth, weight gain, constipation, and mood changes, which may warrant dose reduction or discontinuation. Antiepileptics such as topiramate, mitochondrial supplements such as Coenzyme Q10 and riboflavin are alternative prophylactic agents in CVS.33 Aprepitant, a newer NK1 receptor antagonist has been found to be effective in refractory CVS.34 In addition to pharmacotherapy, addressing comorbid conditions such as anxiety and depression and counseling patients to abstain from heavy cannabis use is also important to achieve good health care outcomes.
In summary, CVS is a common, chronic functional GI disorder with episodic nausea, vomiting, and often, abdominal pain. Symptoms can be disabling, and prompt diagnosis and therapy is important. CVS is associated with multiple comorbid conditions such as migraine, anxiety and depression, and a biopsychosocial model of care is essential. Medications such as amitriptyline are effective in the prophylaxis of CVS, but side effects hamper their use. Recent recommendations for management of CVS have been published.16 Cannabis is frequently used by patients for symptom relief but use of high potency products may cause worsening of symptoms or unmask symptoms in genetically predisposed individuals.23 Studies to elucidate the pathophysiology of CVS should help in the development of better therapies.
Dr. Mooers is PGY-2, an internal medicine resident in the department of medicine, Medical College of Wisconsin, Milwaukee; Dr. Venkatesan is professor of medicine, division of gastroenterology and hepatology, department of medicine, Medical College of Wisconsin, Milwaukee. The authors have no conflicts to disclose.
References
1. Stanghellini V et al. Gastroenterology. 2016;150:1380-92.
2. Aziz I et al. Clin Gastroenterol Hepatol. 2019 Apr;17(5):878-86.
3. Kovacic K et al. Curr Gastroenterol Rep. 2018;20(10):46.
4. Zaki EA et al. Cephalalgia. 2009;29:719-28.
5. Venkatesan T et al. BMC Gastroenterol. 2014;14:181.
6. Ellingsen DM et al. Neurogastroenterol Motil. 2017;29 (6)e13004 9.
7. Venkatesan T et al. Neurogastroenterol Motil. 2016;28:1409-18.
8. Wasilewski A et al. Am J Gastroenterol. 2017;112:933-9.
9. van Sickle MD et al. Am J Physiol Gastrointest Liver Physiol 2003;285:G566-76.
10. Parker LA et al. Br J Pharmacol. 2011;163:1411-22.
11. van Sickle MD et al. Gastroenterology. 2001;121:767-74.
12. Fleisher DR et al. BMC Med. 2005;3:20.
13. Kumar N et al. BMC Gastroenterol. 2012;12:52.
14. Li BU et al. J Pediatr Gastroenterol Nutr. 2008;47:379-93.
15. Bhandari S et al. Clin Auton Res. 2018 Apr;28(2):203-9.
16. Venkatesan T et al. Neurogastroenterol Motil. 2019;31 Suppl 2:e13604. doi: 10.1111/nmo.13604.
17. Sagar RC et al. Neurogastroenterol Motil. 2018;30. doi: 10.1111/nmo.13174.
18. Taranukha T et al. Neurogastroenterol Motil. 2018 Apr;30(4):e13245. doi: 10.1111/nmo.13245.
19. Bhandari S and Venkatesan T. Dig Dis Sci. 2017;62:2035-44.
20. Choung RS et al. Neurogastroenterol Motil. 2012;24:20-6, e21. doi: 10.1111/j.1365-2982.2011.01791.x.
21. Bhandari S et al. Intern Med J. 2019 May;49(5):649-55.
22. Venkatesan T et al. Exp Brain Res. 2014; 232:2563-70.
23. Venkatesan T et al. Clin Gastroenterol Hepatol. 2019 Jul 25. doi: 10.1016/j.cgh.2019.07.039.
24. Simonetto DA et al. Mayo Clin Proc. 2012;87:114-9.
25. Wallace EA et al. South Med J. 2011;104:659-64.
26. Allen JH et al. Gut. 2004;53:1566-70.
27. Venkatesan T et al. Neurogastroenterol Motil. 2019;31 Suppl 2:e13606. doi: 10.1111/nmo.13606.
28. Habboushe J et al. Basic Clin Pharmacol Toxicol. 2018;122:660-2.
29. Bhandari S and Venkatesan T. Curr Treat Options Gastroenterol. 2016;14:495-506.
30. Hikita T et al. Cephalalgia. 2011;31:504-7.
31. Fuseau E et al. Clin Pharmacokinet 2002;41:801-11.
32. Hejazi RA et al. J Clin Gastroenterol. 2010;44:18-21.
33. Sezer OB and Sezer T. J Neurogastroenterol Motil. 2016;22:656-60.
34. Cristofori F et al. Aliment Pharmacol Ther. 2014;40:309-17.
Introduction
Cyclic vomiting syndrome (CVS) is a chronic disorder of gut-brain interaction (DGBI) and is characterized by recurrent episodes of severe nausea, vomiting, and often, abdominal pain. Patients are usually asymptomatic in between episodes.1 CVS was considered a pediatric disease but is now known to be as common in adults. The prevalence of CVS in adults was 2% in a recent population-based study.2 Patients are predominantly white. Both males and females are affected with some studies showing a female preponderance. The mean age of onset is 5 years in children and 35 years in adults.3
The etiology of CVS is not known, but various hypotheses have been proposed. Zaki et al. showed that two mitochondrial DNA polymorphisms 16519T and 3010A were associated with a 17-fold increased odds of having CVS in children.4 These polymorphisms were not associated with CVS in adults.5 Alterations in the brain-gut axis also have been shown in CVS. Functional neuroimaging studies demonstrate that patients with CVS displayed increased connectivity between insula and salience networks with concomitant decrease in connectivity to somatosensory networks.6 Recent data also indicate that the endocannabinoid system (ECS) and the hypothalamic-pituitary-adrenal axis are implicated in CVS with an increase in serum endocannabinoid concentration during an episode.7 The same study also showed a significant increase in salivary cortisol in CVS patients who used cannabis. Further, single nucleotide polymorphisms (SNPs) in the gene that encodes for the cannabinoid receptor type 1 (CB1R) are implicated in CVS.8 The CB1R is part of the ECS and is densely expressed in brain areas involved in emesis, such as the dorsal vagal complex consisting of the area postrema (AP), nucleus of the solitary tract (NTS), and also the dorsal motor nucleus of the vagus.9 Wasilewski et al. showed an increased risk of CVS among individuals with AG and GG genotypes of CNR1 rs806380 (P less than .01), whereas the CC genotype of CNR1 rs806368 was associated with a decreased risk of CVS (P less than .05).8 CB1R agonists – endocannabinoids and tetrahydrocannabinol (THC) – have acute antiemetic and anxiolytic effects.9-11 The apparent paradoxical effects of cannabis in this patient population are yet to be explained and need further study.
Diagnosis and clinical features of CVS

Figure 1: Phases of Cyclic Vomiting Syndrome12
Adapted from Fleisher DR, Gornowicz B, Adams K, Burch R, Feldman EJ. Cyclic Vomiting Syndrome in 41 adults: The illness, the patients, and problems of management. BMC Med 2005;3:20. This work is licensed under the Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/, which permits unrestricted use, distribution, modification, and reproduction in any medium.
CVS consists of four phases which include the a) prodromal phase, b) the episodic phase, c) recovery phase, and d) the interepisodic phase; and was first described by David Fleisher.12 The phases of CVS are important for clinicians and patients alike as they have therapeutic implications. The administration of abortive medications during a prodrome can terminate an episode. The phases of CVS are shown above.
Most patients (~ 93%) have a prodromal phase. Symptoms during this phase can include nausea, abdominal pain, diaphoresis, fatigue, weakness, hot flashes, chills, shivering, increased thirst, loss of appetite, burping, lightheadedness, and paresthesia.13 Some patients report a sense of impending doom and many have symptoms consistent with panic. If untreated, this progresses to the emetic phase and patients have unrelenting nausea, retching, vomiting, and other symptoms. During an episode, patients may vomit up to 20 times per hour and the episode may last several hours to days. During this phase, patients are sometimes described as being in a “conscious coma” and exhibit lethargy, listlessness, withdrawal, and sometimes disorientation.14,15 The emetic phase is followed by the recovery phase, during which symptoms subside and patients are able to resume oral intake. Patients are usually asymptomatic between episodes but ~ 30% can have interepisodic nausea and dyspepsia. In some patients, episodes become progressively longer and the interepisodic phase is considerably shortened and patients have a “coalescence of symptoms.”12 It is important to elicit a thorough history in all patients with vomiting in order to make an accurate diagnosis of CVS since coalescence of symptoms only occurs over a period of time. Episodes often are triggered by psychological stress, both positive and negative. Common triggers can include positive events such as birthdays, holidays, and negative ones like examinations, the death of a loved one, etc. Sleep deprivation and physical exhaustion also can trigger an episode.12
CVS remains a clinical diagnosis since there are no biomarkers. While there is a lack of data on the optimal work-up in these patients, experts recommend an upper endoscopy or upper GI series in order to rule out alternative gastric and intestinal pathology (e.g., malrotation with volvulus).16 Of note, a gastric-emptying study is not recommended as part of the routine work-up as per recent guidelines because of the poor specificity of this test in establishing a diagnosis of CVS.16 Biochemical testing including a complete blood count, serum electrolytes, serum glucose, liver panel, and urinalysis is also warranted. Any additional testing is indicated when clinical features suggest an alternative diagnosis. For instance, neurologic symptoms might warrant a cranial MRI to exclude an intracerebral tumor or other lesions of the brain.

The severity and unpredictable nature of symptoms makes it difficult for some patients to attend school or work; one study found that 32% of patients with CVS were completely disabled.12 Despite increasing awareness of this disorder, patients often are misdiagnosed. The prevalence of CVS in an outpatient gastroenterology clinic in the United Kingdom was 11% and was markedly underdiagnosed in the community.17 Only 5% of patients who were subsequently diagnosed with CVS were initially diagnosed accurately by their referring physician despite meeting criteria for the disorder.17 A subset of patients with CVS even undergo futile surgeries.13 Fleisher et al. noted that 30% of a 41-patient cohort underwent cholecystectomy for CVS symptoms without any improvement in disease.12 Prompt diagnosis and appropriate therapy is essential to improve patient outcomes and improve quality of life.
CVS is associated with various comorbidities such as migraine, anxiety, depression and dysautonomia, which can further impair quality of life.18,19 Approximately 70% of CVS patients report a personal or family history of migraine. Anxiety and depression affects nearly half of patients with CVS.13 Cannabis use is significantly more prevalent among patients with CVS than patients without CVS.20
Role of cannabis in CVS
The role of cannabis in the pathogenesis of symptoms in CVS is controversial. While cannabis has antiemetic properties, there is a strong link between its use and CVS. The use of cannabis has increased over the past decade with increasing legalization.21 Several studies have shown that 40%-80% of patients with CVS use cannabis.22,23 Following this, cannabinoid hyperemesis syndrome (CHS) was coined as a separate entity based on this statistical association, though there are no data to support the notion that cannabis causes vomiting.24,25 CHS has clinical features that are indistinguishable from CVS except for the chronic heavy cannabis use. A peculiar bathing behavior called “compulsive hot-water bathing” has been described and was thought to be pathognomonic of cannabis use.26 During an episode, patients will take multiple hot showers/baths, which temporarily alleviate their symptoms. Many patients even report running out of hot water and sometimes check into a hotel for a continuous supply of hot water. A small number of patients may sustain burns from the hot-water bathing. However, studies show that this hot-water bathing behavior also is seen in about 50% of patents with CVS who do not use cannabis.22

CHS is now defined by Rome IV criteria, which include episodes of nausea and vomiting similar to CVS preceded by chronic, heavy cannabis use. Patients must have complete resolution of symptoms following cessation.1 A recent systematic review of 376 cases of purported CHS showed that only 59 (15.7%) met Rome IV criteria for this disorder.27 This is because of considerable heterogeneity in how the diagnosis of CHS was made and the lack of standard diagnostic criteria at the time. Some cases of CHS were diagnosed merely based on an association of vomiting, hot-water bathing, and cannabis use.28 Only a minority of patients (71,19%) had a duration of follow-up more than 4 weeks, which would make it impossible to establish a diagnosis of CHS. A period of at least a year or a duration of time that spans at least three episodes is generally recommended to determine if abstinence from cannabis causes a true resolution of symptoms.27 Whether CHS is a separate entity or a subtype of CVS remains to be determined. The paradoxical effects of cannabis may happen because of the use of highly potent cannabis products that are currently in use. A complete discussion of the role of cannabis in CVS is beyond the scope of this article, and the reader is referred to a recent systematic review and discussion.27
Treatment
CVS should be treated based on a biopsychosocial model with a multidisciplinary team that includes a gastroenterologist with knowledge of CVS, primary care physician, psychologist, psychiatrist, and sleep specialist if needed.16 Initiating prophylactic treatment is based on the severity of disease. An algorithm for the treatment of CVS based on severity of symptoms is shown below.
Figure 2. Adapted and reprinted by permission from the Licensor: Springer Nature, Current Treatment Options in Gastroenterology, Bhandari S, Venkatesan T. Novel Treatments for Cyclic Vomiting Syndrome: Beyond Ondansetron and Amitriptyline, 14:495-506, Copyright 2016.
Patients who have mild disease (defined as fewer than four episodes/year, episodes lasting up to 2 days, quick recovery from episodes, or episodes not requiring ED care or hospitalization) are usually prescribed abortive medications.16 These medications are best administered during the prodromal phase and can prevent progression to the emetic phase. Medications used for aborting episodes include sumatriptan (20 mg intranasal or 6 mg subcutaneous), ondansetron (8 mg sublingual), and diphenhydramine (25-50 mg).30,31 This combination can help abort symptoms and potentially avoid ED visits or hospitalizations. Patients with moderate-to-severe CVS are offered prophylactic therapy in addition to abortive therapy.16
Recent guidelines recommend tricyclic antidepressants (TCAs) as the first-line agent in the prophylaxis of CVS episodes. Data from 14 studies determined that 70% (413/600) of patients responded partially or completely to TCAs.16 An open-label study of 46 patients by Hejazi et al. noted a decline in the number of CVS episodes from 17 to 3, in the duration of a CVS episode from 6 to 2 days, and in the number of ED visits/ hospitalizations from 15 to 3.3.32Amitriptyline should be started at 25 mg at night and titrated up by 10-25 mg each week to minimize emergence of side effects. The mean effective dose is 75-100 mg or 1.0-1.5 mg/kg. An EKG should be checked at baseline and during titration to monitor the QT interval. Unfortunately, side effects from TCAs are quite common and include cognitive impairment, drowsiness, dryness of mouth, weight gain, constipation, and mood changes, which may warrant dose reduction or discontinuation. Antiepileptics such as topiramate, mitochondrial supplements such as Coenzyme Q10 and riboflavin are alternative prophylactic agents in CVS.33 Aprepitant, a newer NK1 receptor antagonist has been found to be effective in refractory CVS.34 In addition to pharmacotherapy, addressing comorbid conditions such as anxiety and depression and counseling patients to abstain from heavy cannabis use is also important to achieve good health care outcomes.
In summary, CVS is a common, chronic functional GI disorder with episodic nausea, vomiting, and often, abdominal pain. Symptoms can be disabling, and prompt diagnosis and therapy is important. CVS is associated with multiple comorbid conditions such as migraine, anxiety and depression, and a biopsychosocial model of care is essential. Medications such as amitriptyline are effective in the prophylaxis of CVS, but side effects hamper their use. Recent recommendations for management of CVS have been published.16 Cannabis is frequently used by patients for symptom relief but use of high potency products may cause worsening of symptoms or unmask symptoms in genetically predisposed individuals.23 Studies to elucidate the pathophysiology of CVS should help in the development of better therapies.
Dr. Mooers is PGY-2, an internal medicine resident in the department of medicine, Medical College of Wisconsin, Milwaukee; Dr. Venkatesan is professor of medicine, division of gastroenterology and hepatology, department of medicine, Medical College of Wisconsin, Milwaukee. The authors have no conflicts to disclose.
References
1. Stanghellini V et al. Gastroenterology. 2016;150:1380-92.
2. Aziz I et al. Clin Gastroenterol Hepatol. 2019 Apr;17(5):878-86.
3. Kovacic K et al. Curr Gastroenterol Rep. 2018;20(10):46.
4. Zaki EA et al. Cephalalgia. 2009;29:719-28.
5. Venkatesan T et al. BMC Gastroenterol. 2014;14:181.
6. Ellingsen DM et al. Neurogastroenterol Motil. 2017;29 (6)e13004 9.
7. Venkatesan T et al. Neurogastroenterol Motil. 2016;28:1409-18.
8. Wasilewski A et al. Am J Gastroenterol. 2017;112:933-9.
9. van Sickle MD et al. Am J Physiol Gastrointest Liver Physiol 2003;285:G566-76.
10. Parker LA et al. Br J Pharmacol. 2011;163:1411-22.
11. van Sickle MD et al. Gastroenterology. 2001;121:767-74.
12. Fleisher DR et al. BMC Med. 2005;3:20.
13. Kumar N et al. BMC Gastroenterol. 2012;12:52.
14. Li BU et al. J Pediatr Gastroenterol Nutr. 2008;47:379-93.
15. Bhandari S et al. Clin Auton Res. 2018 Apr;28(2):203-9.
16. Venkatesan T et al. Neurogastroenterol Motil. 2019;31 Suppl 2:e13604. doi: 10.1111/nmo.13604.
17. Sagar RC et al. Neurogastroenterol Motil. 2018;30. doi: 10.1111/nmo.13174.
18. Taranukha T et al. Neurogastroenterol Motil. 2018 Apr;30(4):e13245. doi: 10.1111/nmo.13245.
19. Bhandari S and Venkatesan T. Dig Dis Sci. 2017;62:2035-44.
20. Choung RS et al. Neurogastroenterol Motil. 2012;24:20-6, e21. doi: 10.1111/j.1365-2982.2011.01791.x.
21. Bhandari S et al. Intern Med J. 2019 May;49(5):649-55.
22. Venkatesan T et al. Exp Brain Res. 2014; 232:2563-70.
23. Venkatesan T et al. Clin Gastroenterol Hepatol. 2019 Jul 25. doi: 10.1016/j.cgh.2019.07.039.
24. Simonetto DA et al. Mayo Clin Proc. 2012;87:114-9.
25. Wallace EA et al. South Med J. 2011;104:659-64.
26. Allen JH et al. Gut. 2004;53:1566-70.
27. Venkatesan T et al. Neurogastroenterol Motil. 2019;31 Suppl 2:e13606. doi: 10.1111/nmo.13606.
28. Habboushe J et al. Basic Clin Pharmacol Toxicol. 2018;122:660-2.
29. Bhandari S and Venkatesan T. Curr Treat Options Gastroenterol. 2016;14:495-506.
30. Hikita T et al. Cephalalgia. 2011;31:504-7.
31. Fuseau E et al. Clin Pharmacokinet 2002;41:801-11.
32. Hejazi RA et al. J Clin Gastroenterol. 2010;44:18-21.
33. Sezer OB and Sezer T. J Neurogastroenterol Motil. 2016;22:656-60.
34. Cristofori F et al. Aliment Pharmacol Ther. 2014;40:309-17.
Survey: Hydroxychloroquine use fairly common in COVID-19
One of five physicians in front-line treatment roles has prescribed hydroxychloroquine for COVID-19, according to a new survey from health care market research company InCrowd.
The most common treatments were acetaminophen, prescribed to 82% of patients, antibiotics (41%), and bronchodilators (40%), InCrowd said after surveying 203 primary care physicians, pediatricians, and emergency medicine or critical care physicians who are treating at least 20 patients with flulike symptoms.
On April 24, the Food and Drug Administration warned against the use of hydroxychloroquine or chloroquine outside of hospitals and clinical trials.
The InCrowd survey, which took place April 14-15 and is the fourth in a series investigating COVID-19’s impact on physicians, showed that access to testing was up to 82% in mid-April, compared with 67% in March and 20% in late February. The April respondents also were twice as likely (59% vs. 24% in March) to say that their facilities were prepared to treat patients, InCrowd reported.
“U.S. physicians report sluggish optimism around preparedness, safety, and institutional efforts, while many worry about the future, including a second outbreak and job security,” the company said in a separate written statement.
The average estimate for a return to normal was just over 6 months among respondents, and only 28% believed that their facility was prepared for a second outbreak later in the year, InCrowd noted.
On a personal level, 45% of the respondents were concerned about the safety of their job. An emergency/critical care physician from Tennessee said, “We’ve been cutting back on staff due to overall revenue reductions, but have increased acuity and complexity which requires more staffing. This puts even more of a burden on those of us still here.”
Support for institutional responses to slow the pandemic was strongest for state governments, which gained approval from 54% of front-line physicians, up from 33% in March. Actions taken by the federal government were supported by 21% of respondents, compared with 38% for the World Health Organization and 46% for governments outside the United States, InCrowd reported.
Suggestions for further actions by state and local authorities included this comment from an emergency/critical care physician in Florida: “Continued, broad and properly enforced stay at home and social distancing measures MUST remain in place to keep citizens and healthcare workers safe, and the latter alive and in adequate supply.”
One of five physicians in front-line treatment roles has prescribed hydroxychloroquine for COVID-19, according to a new survey from health care market research company InCrowd.

The most common treatments were acetaminophen, prescribed to 82% of patients, antibiotics (41%), and bronchodilators (40%), InCrowd said after surveying 203 primary care physicians, pediatricians, and emergency medicine or critical care physicians who are treating at least 20 patients with flulike symptoms.
On April 24, the Food and Drug Administration warned against the use of hydroxychloroquine or chloroquine outside of hospitals and clinical trials.
The InCrowd survey, which took place April 14-15 and is the fourth in a series investigating COVID-19’s impact on physicians, showed that access to testing was up to 82% in mid-April, compared with 67% in March and 20% in late February. The April respondents also were twice as likely (59% vs. 24% in March) to say that their facilities were prepared to treat patients, InCrowd reported.
“U.S. physicians report sluggish optimism around preparedness, safety, and institutional efforts, while many worry about the future, including a second outbreak and job security,” the company said in a separate written statement.
The average estimate for a return to normal was just over 6 months among respondents, and only 28% believed that their facility was prepared for a second outbreak later in the year, InCrowd noted.
On a personal level, 45% of the respondents were concerned about the safety of their job. An emergency/critical care physician from Tennessee said, “We’ve been cutting back on staff due to overall revenue reductions, but have increased acuity and complexity which requires more staffing. This puts even more of a burden on those of us still here.”
Support for institutional responses to slow the pandemic was strongest for state governments, which gained approval from 54% of front-line physicians, up from 33% in March. Actions taken by the federal government were supported by 21% of respondents, compared with 38% for the World Health Organization and 46% for governments outside the United States, InCrowd reported.
Suggestions for further actions by state and local authorities included this comment from an emergency/critical care physician in Florida: “Continued, broad and properly enforced stay at home and social distancing measures MUST remain in place to keep citizens and healthcare workers safe, and the latter alive and in adequate supply.”
One of five physicians in front-line treatment roles has prescribed hydroxychloroquine for COVID-19, according to a new survey from health care market research company InCrowd.

The most common treatments were acetaminophen, prescribed to 82% of patients, antibiotics (41%), and bronchodilators (40%), InCrowd said after surveying 203 primary care physicians, pediatricians, and emergency medicine or critical care physicians who are treating at least 20 patients with flulike symptoms.
On April 24, the Food and Drug Administration warned against the use of hydroxychloroquine or chloroquine outside of hospitals and clinical trials.
The InCrowd survey, which took place April 14-15 and is the fourth in a series investigating COVID-19’s impact on physicians, showed that access to testing was up to 82% in mid-April, compared with 67% in March and 20% in late February. The April respondents also were twice as likely (59% vs. 24% in March) to say that their facilities were prepared to treat patients, InCrowd reported.
“U.S. physicians report sluggish optimism around preparedness, safety, and institutional efforts, while many worry about the future, including a second outbreak and job security,” the company said in a separate written statement.
The average estimate for a return to normal was just over 6 months among respondents, and only 28% believed that their facility was prepared for a second outbreak later in the year, InCrowd noted.
On a personal level, 45% of the respondents were concerned about the safety of their job. An emergency/critical care physician from Tennessee said, “We’ve been cutting back on staff due to overall revenue reductions, but have increased acuity and complexity which requires more staffing. This puts even more of a burden on those of us still here.”
Support for institutional responses to slow the pandemic was strongest for state governments, which gained approval from 54% of front-line physicians, up from 33% in March. Actions taken by the federal government were supported by 21% of respondents, compared with 38% for the World Health Organization and 46% for governments outside the United States, InCrowd reported.
Suggestions for further actions by state and local authorities included this comment from an emergency/critical care physician in Florida: “Continued, broad and properly enforced stay at home and social distancing measures MUST remain in place to keep citizens and healthcare workers safe, and the latter alive and in adequate supply.”