User login
For MD-IQ use only
FDA warns of potential mechanical concerns with MAGEC devices
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
‘Gold cards’ allow Texas docs to skip prior authorizations
The law was passed in June and will take effect in September. It excuses physicians from having to obtain prior authorization if, during the previous 6 months, 90% of their treatments met medical necessity criteria by the health insurer. Through this law, doctors in the state will spend less time getting approvals for treatments for their patients.
Automatic approval of authorizations for treatments – or what the Texas Medical Association (TMA) calls a “gold card” – “allows patients to get the care they need in a more timely fashion,” says Debra Patt, MD, an Austin, Tex.–based oncologist and former chair of the council on legislation for the TMA.
Eighty-seven percent of Texas physicians reported a “drastic increase over the past five years in the burden of prior authorization on their patients and their practices,” per a 2020 survey by the TMA. Nearly half (48%) of Texas physicians have hired staff whose work focuses on processing requests for prior authorization, according to the survey.
Jack Resneck Jr., MD, a San Francisco–based dermatologist and president-elect of the American Medical Association (AMA), said other states have investigated ways to ease the impact of prior authorizations on physicians, but no other state has passed such a law.
Administrative burdens plague physicians around the country. The Medscape Physician Compensation Report 2021 found that physicians spend on average 15.6 hours per week on paperwork and administrative duties.
Better outcomes, less anxiety for patients
Dr. Patt, who testified in support of the law’s passage in the Texas legislature, says automatic approval of authorizations “is better for patients because it reduces their anxiety about whether they’re able to get the treatments they need now, and they will have better outcomes if they’re able to receive more timely care.”
Recently, a chemotherapy treatment Dr. Patt prescribed for one of her patients was not authorized by an insurer. The result is “a lot of anxiety and potentially health problems” for the patient, said Dr. Patt.
She expects that automatic approval for treatments will be based on prescribing patterns during the preceding 6 months. “It means that when I order a test today, the [health insurer] looks back at my record 6 months previously,” she said. Still, Dr. Patt awaits guidance from the Texas Department of Insurance, which regulates health insurers in the state, regarding the law.
Dr. Resneck said the pharmacy counter is where most patients encounter prior authorization delays. “That’s when the pharmacist looks at them and says, ‘Actually, this isn’t covered by your health insurer’s formulary,’ or it isn’t covered fully on their formulary.”
One of Dr. Resneck’s patients had a life-altering case of eczema that lasted many years. Because of the condition, the patient couldn’t work or maintain meaningful bonds with family members. A biologic treatment transformed his patient’s life. The patient was able to return to work and to re-engage with family, said Dr. Resneck. But a year after his patient started the treatment, the health insurer wouldn’t authorize the treatment because the patient wasn’t experiencing the same symptoms.
The patient didn’t have the same symptoms because the biologic treatment worked, said Dr. Resneck.
Kristine Grow, a spokesperson for America’s Health Insurance Plans, a national association for health insurers, said, “The use of prior authorization is relatively small – typically, less than 15% – and can help ensure safer opioid prescribing, help prevent dangerous drug interactions, and help protect patients from unnecessary exposure to potentially harmful radiation for inappropriate diagnostic imaging. Numerous studies show that Americans frequently receive inappropriate care, and 25% of unnecessary treatments are associated with complications or adverse events.”
Medical management tools, such as prior authorization, are an “an important way” to deliver “safe, high-quality care” to patients, she added.
State and federal efforts to curb prior authorization
In addition to efforts to curb prior authorization in other states, the AMA supports the Improving Seniors’ Timely Access to Care Act (HR 3173). The act includes a provision related to “gold-carding,” said Robert Mills, an AMA spokesperson.
The bill establishes requirements and standards for prior authorization processes related to Medicare Advantage (MA) plans. The requirements and standards for MA plans include the following:
- Establishing an electronic prior authorization program that meets specific standards, such as the ability to provide real-time decisions in response to requests for items and services that are routinely approved.
- Publishing on an annual basis specific prior authorization information, including the percentage of requests approved and the average response time.
- Meeting standards set by the Centers for Medicare & Medicaid Services related to the quality and timeliness of prior authorization determinations.
The act was introduced to the U.S. House of Representatives in May, after which it was referred to two committees for consideration.
A version of this article first appeared on Medscape.com.
The law was passed in June and will take effect in September. It excuses physicians from having to obtain prior authorization if, during the previous 6 months, 90% of their treatments met medical necessity criteria by the health insurer. Through this law, doctors in the state will spend less time getting approvals for treatments for their patients.
Automatic approval of authorizations for treatments – or what the Texas Medical Association (TMA) calls a “gold card” – “allows patients to get the care they need in a more timely fashion,” says Debra Patt, MD, an Austin, Tex.–based oncologist and former chair of the council on legislation for the TMA.
Eighty-seven percent of Texas physicians reported a “drastic increase over the past five years in the burden of prior authorization on their patients and their practices,” per a 2020 survey by the TMA. Nearly half (48%) of Texas physicians have hired staff whose work focuses on processing requests for prior authorization, according to the survey.
Jack Resneck Jr., MD, a San Francisco–based dermatologist and president-elect of the American Medical Association (AMA), said other states have investigated ways to ease the impact of prior authorizations on physicians, but no other state has passed such a law.
Administrative burdens plague physicians around the country. The Medscape Physician Compensation Report 2021 found that physicians spend on average 15.6 hours per week on paperwork and administrative duties.
Better outcomes, less anxiety for patients
Dr. Patt, who testified in support of the law’s passage in the Texas legislature, says automatic approval of authorizations “is better for patients because it reduces their anxiety about whether they’re able to get the treatments they need now, and they will have better outcomes if they’re able to receive more timely care.”
Recently, a chemotherapy treatment Dr. Patt prescribed for one of her patients was not authorized by an insurer. The result is “a lot of anxiety and potentially health problems” for the patient, said Dr. Patt.
She expects that automatic approval for treatments will be based on prescribing patterns during the preceding 6 months. “It means that when I order a test today, the [health insurer] looks back at my record 6 months previously,” she said. Still, Dr. Patt awaits guidance from the Texas Department of Insurance, which regulates health insurers in the state, regarding the law.
Dr. Resneck said the pharmacy counter is where most patients encounter prior authorization delays. “That’s when the pharmacist looks at them and says, ‘Actually, this isn’t covered by your health insurer’s formulary,’ or it isn’t covered fully on their formulary.”
One of Dr. Resneck’s patients had a life-altering case of eczema that lasted many years. Because of the condition, the patient couldn’t work or maintain meaningful bonds with family members. A biologic treatment transformed his patient’s life. The patient was able to return to work and to re-engage with family, said Dr. Resneck. But a year after his patient started the treatment, the health insurer wouldn’t authorize the treatment because the patient wasn’t experiencing the same symptoms.
The patient didn’t have the same symptoms because the biologic treatment worked, said Dr. Resneck.
Kristine Grow, a spokesperson for America’s Health Insurance Plans, a national association for health insurers, said, “The use of prior authorization is relatively small – typically, less than 15% – and can help ensure safer opioid prescribing, help prevent dangerous drug interactions, and help protect patients from unnecessary exposure to potentially harmful radiation for inappropriate diagnostic imaging. Numerous studies show that Americans frequently receive inappropriate care, and 25% of unnecessary treatments are associated with complications or adverse events.”
Medical management tools, such as prior authorization, are an “an important way” to deliver “safe, high-quality care” to patients, she added.
State and federal efforts to curb prior authorization
In addition to efforts to curb prior authorization in other states, the AMA supports the Improving Seniors’ Timely Access to Care Act (HR 3173). The act includes a provision related to “gold-carding,” said Robert Mills, an AMA spokesperson.
The bill establishes requirements and standards for prior authorization processes related to Medicare Advantage (MA) plans. The requirements and standards for MA plans include the following:
- Establishing an electronic prior authorization program that meets specific standards, such as the ability to provide real-time decisions in response to requests for items and services that are routinely approved.
- Publishing on an annual basis specific prior authorization information, including the percentage of requests approved and the average response time.
- Meeting standards set by the Centers for Medicare & Medicaid Services related to the quality and timeliness of prior authorization determinations.
The act was introduced to the U.S. House of Representatives in May, after which it was referred to two committees for consideration.
A version of this article first appeared on Medscape.com.
The law was passed in June and will take effect in September. It excuses physicians from having to obtain prior authorization if, during the previous 6 months, 90% of their treatments met medical necessity criteria by the health insurer. Through this law, doctors in the state will spend less time getting approvals for treatments for their patients.
Automatic approval of authorizations for treatments – or what the Texas Medical Association (TMA) calls a “gold card” – “allows patients to get the care they need in a more timely fashion,” says Debra Patt, MD, an Austin, Tex.–based oncologist and former chair of the council on legislation for the TMA.
Eighty-seven percent of Texas physicians reported a “drastic increase over the past five years in the burden of prior authorization on their patients and their practices,” per a 2020 survey by the TMA. Nearly half (48%) of Texas physicians have hired staff whose work focuses on processing requests for prior authorization, according to the survey.
Jack Resneck Jr., MD, a San Francisco–based dermatologist and president-elect of the American Medical Association (AMA), said other states have investigated ways to ease the impact of prior authorizations on physicians, but no other state has passed such a law.
Administrative burdens plague physicians around the country. The Medscape Physician Compensation Report 2021 found that physicians spend on average 15.6 hours per week on paperwork and administrative duties.
Better outcomes, less anxiety for patients
Dr. Patt, who testified in support of the law’s passage in the Texas legislature, says automatic approval of authorizations “is better for patients because it reduces their anxiety about whether they’re able to get the treatments they need now, and they will have better outcomes if they’re able to receive more timely care.”
Recently, a chemotherapy treatment Dr. Patt prescribed for one of her patients was not authorized by an insurer. The result is “a lot of anxiety and potentially health problems” for the patient, said Dr. Patt.
She expects that automatic approval for treatments will be based on prescribing patterns during the preceding 6 months. “It means that when I order a test today, the [health insurer] looks back at my record 6 months previously,” she said. Still, Dr. Patt awaits guidance from the Texas Department of Insurance, which regulates health insurers in the state, regarding the law.
Dr. Resneck said the pharmacy counter is where most patients encounter prior authorization delays. “That’s when the pharmacist looks at them and says, ‘Actually, this isn’t covered by your health insurer’s formulary,’ or it isn’t covered fully on their formulary.”
One of Dr. Resneck’s patients had a life-altering case of eczema that lasted many years. Because of the condition, the patient couldn’t work or maintain meaningful bonds with family members. A biologic treatment transformed his patient’s life. The patient was able to return to work and to re-engage with family, said Dr. Resneck. But a year after his patient started the treatment, the health insurer wouldn’t authorize the treatment because the patient wasn’t experiencing the same symptoms.
The patient didn’t have the same symptoms because the biologic treatment worked, said Dr. Resneck.
Kristine Grow, a spokesperson for America’s Health Insurance Plans, a national association for health insurers, said, “The use of prior authorization is relatively small – typically, less than 15% – and can help ensure safer opioid prescribing, help prevent dangerous drug interactions, and help protect patients from unnecessary exposure to potentially harmful radiation for inappropriate diagnostic imaging. Numerous studies show that Americans frequently receive inappropriate care, and 25% of unnecessary treatments are associated with complications or adverse events.”
Medical management tools, such as prior authorization, are an “an important way” to deliver “safe, high-quality care” to patients, she added.
State and federal efforts to curb prior authorization
In addition to efforts to curb prior authorization in other states, the AMA supports the Improving Seniors’ Timely Access to Care Act (HR 3173). The act includes a provision related to “gold-carding,” said Robert Mills, an AMA spokesperson.
The bill establishes requirements and standards for prior authorization processes related to Medicare Advantage (MA) plans. The requirements and standards for MA plans include the following:
- Establishing an electronic prior authorization program that meets specific standards, such as the ability to provide real-time decisions in response to requests for items and services that are routinely approved.
- Publishing on an annual basis specific prior authorization information, including the percentage of requests approved and the average response time.
- Meeting standards set by the Centers for Medicare & Medicaid Services related to the quality and timeliness of prior authorization determinations.
The act was introduced to the U.S. House of Representatives in May, after which it was referred to two committees for consideration.
A version of this article first appeared on Medscape.com.
Hospital medicine and the future of smart care
People often overestimate what will happen in the next two years and underestimate what will happen in ten. – Bill Gates
The COVID-19 pandemic set in motion a series of innovations catalyzing the digital transformation of the health care landscape.
Telemedicine use exploded over the last 12 months to the point that it has almost become ubiquitous. With that, we saw a rapid proliferation of wearables and remote patient monitoring devices. Thanks to virtual care, care delivery is no longer strictly dependent on having onsite specialists, and care itself is not confined to the boundaries of hospitals or doctors’ offices anymore.
We saw the formation of the digital front door and the emergence of new virtual care sites like virtual urgent care, virtual home health, virtual office visits, virtual hospital at home that allowed clinical care to be delivered safely outside the boundaries of hospitals. Nonclinical public places like gyms, schools, and community centers were being transformed into virtual health care portals that brought care closer to the people.
Inside the hospital, we saw a fusion of traditional inpatient care and virtual care. Onsite hospital teams embraced telemedicine during the pandemic for various reasons; to conserve personal protective equipment (PPE), limit exposure, boost care capacity, improve access to specialists at distant sites, and bring family memberse to “webside” who cannot be at a patient’s bedside.
In clinical trials as well, virtual care is a welcome change. According to one survey1, most trial participants favored the use of telehealth services for clinical trials, as these helped them stay engaged, compliant, monitored, and on track while remaining at home. Furthermore, we are seeing the integration of artificial intelligence (AI) into telehealth, whether it is to aid physicians in clinical decision-making or to generate reminders to help patients with chronic disease management. However, this integration is only beginning to scratch the surface of the combination of two technologies’ real potential.
What’s next?
Based on these trends, it should be no surprise that digital health will become a vital sign for health care organizations.
The next 12 to 24 months will set new standards for digital health and play a significant role in defining the next generation of virtual care. There are projections that global health care industry revenues will exceed $2.6 trillion by 2025, with AI and telehealth playing a prominent role in this growth.2 According to estimates, telehealth itself will be a $175 billion market by 2026 and approximately one in three patient encounters will go virtual.3,4 Moreover, virtual care will continue to make exciting transformations, helping to make quality care accessible to everyone in innovative ways. For example, the University of Cincinnati has recently developed a pilot project using a drone equipped with video technology, artificial intelligence, sensors, and first aid kits to go to hard-to-reach areas to deliver care via telemedicine.5
Smart hospitals
In coming years, we can expect the integration of AI, augmented reality (AR), and virtual reality (VR) into telemedicine at lightning speed – and at a much larger scale – that will enable surgeons from different parts of the globe to perform procedures remotely and more precisely.
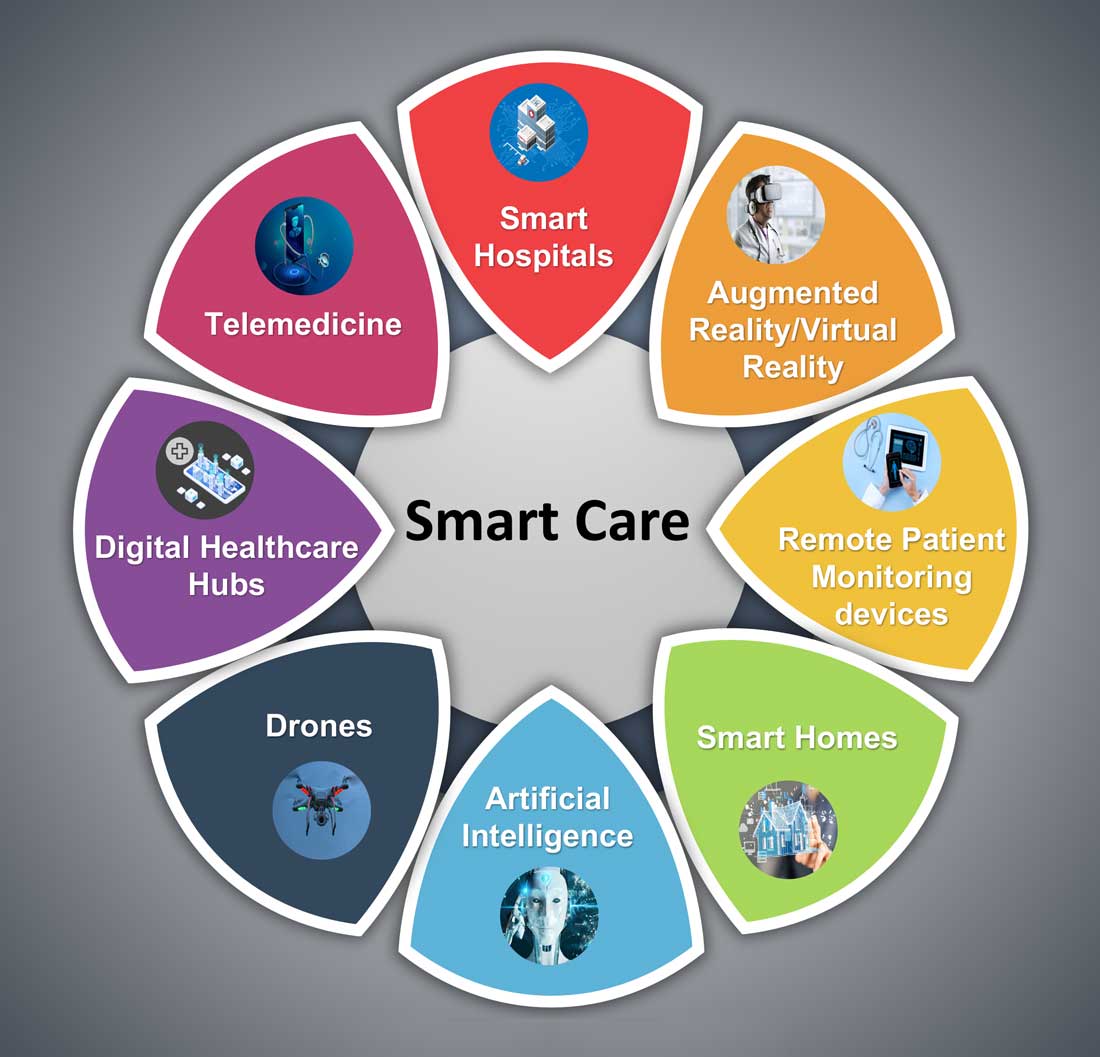
AI is already gaining traction in different fields within health care – whether it’s predicting length of stay in the ICU, or assisting in triage decisions, or reading radiological images, to name just a few. The Mayo Clinic is using AI and computer-aided decision-making tools to predict the risk of surgery and potential post-op complications, which could allow even better collaboration between medical and surgical teams. We hear about the “X-ray” vision offered to proceduralists using HoloLens – mixed reality smartglasses – a technology that enables them to perform procedures more precisely. Others project that there will be more sensors and voice recognition tools in the OR that will be used to gather data to develop intelligent algorithms, and to build a safety net for interventionalists that can notify them of potential hazards or accidental sterile field breaches. The insights gained will be used to create best practices and even allow some procedures to be performed outside the traditional OR setting.
Additionally, we are seeing the development of “smart” patient rooms. For example, one health system in Florida is working on deploying Amazon Alexa in 2,500 patient rooms to allow patients to connect more easily to their care team members. In the not-so-distant future, smart hospitals with smart patient rooms and smart ORs equipped with telemedicine, AI, AR, mixed reality, and computer-aided decision-making tools will no longer be an exception.
Smart homes for smart care
Smart homes with technologies like gas detectors, movement sensors, and sleep sensors will continue to evolve. According to one estimate, the global smart home health care market was $8.7 billion in 2019, and is expected to be $96.2 billion by 2030.6
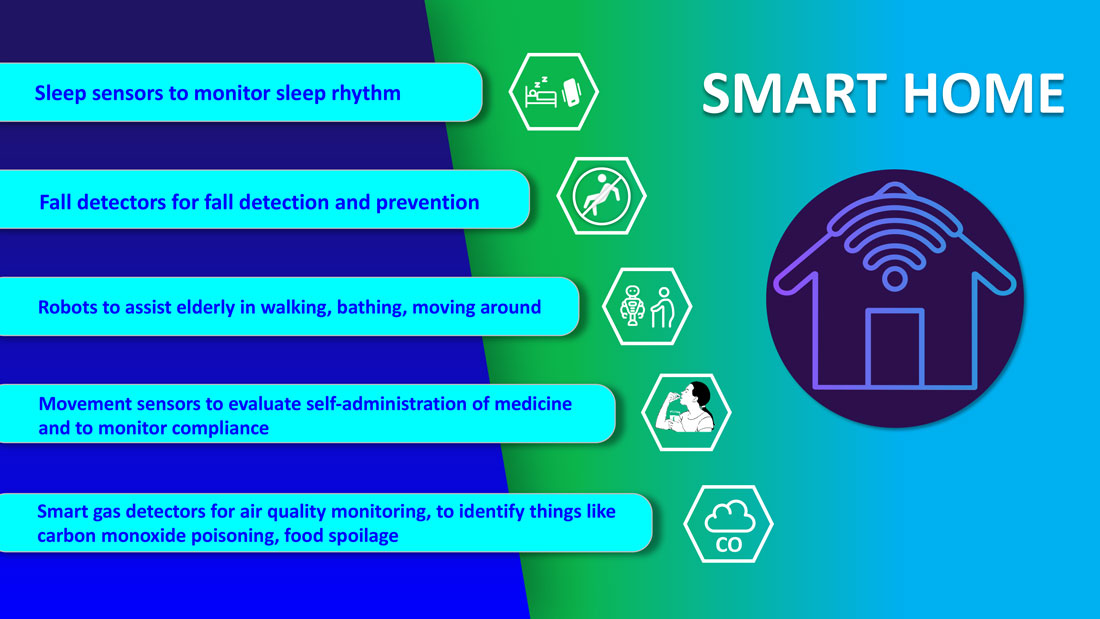
Smart technologies will have applications in fall detection and prevention, evaluation of self-administration of medicine, sleep rhythm monitoring, air quality monitoring for the detection of abnormal gas levels, and identification of things like carbon monoxide poisoning or food spoilage. In coming years, expect to see more virtual medical homes and digital health care complexes. Patients, from the convenience of their homes, might be able to connect to a suite of caregivers, all working collaboratively to provide more coordinated, effective care. The “hospital at home” model that started with six hospitals has already grown to over 100 hospitals across 29 states. The shift from onsite specialists to onscreen specialists will continue, providing greater access to specialized services.
With these emerging trends, it can be anticipated that much acute care will be provided to patients outside the hospital – either under the hospital at home model, via drone technology using telemedicine, through smart devices in smart homes, or via wearables and artificial intelligence. Hence, hospitals’ configuration in the future will be much different and more compact than currently, and many hospitals will be reserved for trauma patients, casualties of natural disasters, higher acuity diseases requiring complex procedures, and other emergencies.
The role of hospitalists has evolved over the years and is still evolving. It should be no surprise if, in the future, we work alongside a digital hospitalist twin to provide better and more personalized care to our patients. Change is uncomfortable but it is inevitable. When COVID hit, we were forced to find innovative ways to deliver care to our patients. One thing is for certain: post-pandemic (AD, or After Disease) we are not going back to a Before COVID (BC) state in terms of virtual care. With the new dawn of digital era, the crucial questions to address will be: What will the future role of a hospitalist look like? How can we leverage technology and embrace our flexibility to adapt to these trends? How can we apply the lessons learned during the pandemic to propel hospital medicine into the future? And is it time to rethink our role and even reclassify ourselves – from hospitalists to Acute Care Experts (ACE) or Primary Acute Care Physicians?
Dr. Zia is a hospitalist, physician advisor, and founder of Virtual Hospitalist - a telemedicine company with a 360-degree care model for hospital patients.
References
1. www.subjectwell.com/news/data-shows-a-majority-of-patients-remain-interested-in-clinical-trials-during-the-coronavirus-pandemic/
2. ww2.frost.com/news/press-releases/technology-innovations-and-virtual-consultations-drive-healthcare-2025/
3. www.gminsights.com/industry-analysis/telemedicine-market
4. www.healthcareitnews.com/blog/frost-sullivans-top-10-predictions-healthcare-2021
5. www.uc.edu/news/articles/2021/03/virtual-medicine--new-uc-telehealth-drone-makes-house-calls.html
6. www.psmarketresearch.com/market-analysis/smart-home-healthcare-market
People often overestimate what will happen in the next two years and underestimate what will happen in ten. – Bill Gates
The COVID-19 pandemic set in motion a series of innovations catalyzing the digital transformation of the health care landscape.
Telemedicine use exploded over the last 12 months to the point that it has almost become ubiquitous. With that, we saw a rapid proliferation of wearables and remote patient monitoring devices. Thanks to virtual care, care delivery is no longer strictly dependent on having onsite specialists, and care itself is not confined to the boundaries of hospitals or doctors’ offices anymore.
We saw the formation of the digital front door and the emergence of new virtual care sites like virtual urgent care, virtual home health, virtual office visits, virtual hospital at home that allowed clinical care to be delivered safely outside the boundaries of hospitals. Nonclinical public places like gyms, schools, and community centers were being transformed into virtual health care portals that brought care closer to the people.
Inside the hospital, we saw a fusion of traditional inpatient care and virtual care. Onsite hospital teams embraced telemedicine during the pandemic for various reasons; to conserve personal protective equipment (PPE), limit exposure, boost care capacity, improve access to specialists at distant sites, and bring family memberse to “webside” who cannot be at a patient’s bedside.
In clinical trials as well, virtual care is a welcome change. According to one survey1, most trial participants favored the use of telehealth services for clinical trials, as these helped them stay engaged, compliant, monitored, and on track while remaining at home. Furthermore, we are seeing the integration of artificial intelligence (AI) into telehealth, whether it is to aid physicians in clinical decision-making or to generate reminders to help patients with chronic disease management. However, this integration is only beginning to scratch the surface of the combination of two technologies’ real potential.
What’s next?
Based on these trends, it should be no surprise that digital health will become a vital sign for health care organizations.
The next 12 to 24 months will set new standards for digital health and play a significant role in defining the next generation of virtual care. There are projections that global health care industry revenues will exceed $2.6 trillion by 2025, with AI and telehealth playing a prominent role in this growth.2 According to estimates, telehealth itself will be a $175 billion market by 2026 and approximately one in three patient encounters will go virtual.3,4 Moreover, virtual care will continue to make exciting transformations, helping to make quality care accessible to everyone in innovative ways. For example, the University of Cincinnati has recently developed a pilot project using a drone equipped with video technology, artificial intelligence, sensors, and first aid kits to go to hard-to-reach areas to deliver care via telemedicine.5
Smart hospitals
In coming years, we can expect the integration of AI, augmented reality (AR), and virtual reality (VR) into telemedicine at lightning speed – and at a much larger scale – that will enable surgeons from different parts of the globe to perform procedures remotely and more precisely.

AI is already gaining traction in different fields within health care – whether it’s predicting length of stay in the ICU, or assisting in triage decisions, or reading radiological images, to name just a few. The Mayo Clinic is using AI and computer-aided decision-making tools to predict the risk of surgery and potential post-op complications, which could allow even better collaboration between medical and surgical teams. We hear about the “X-ray” vision offered to proceduralists using HoloLens – mixed reality smartglasses – a technology that enables them to perform procedures more precisely. Others project that there will be more sensors and voice recognition tools in the OR that will be used to gather data to develop intelligent algorithms, and to build a safety net for interventionalists that can notify them of potential hazards or accidental sterile field breaches. The insights gained will be used to create best practices and even allow some procedures to be performed outside the traditional OR setting.
Additionally, we are seeing the development of “smart” patient rooms. For example, one health system in Florida is working on deploying Amazon Alexa in 2,500 patient rooms to allow patients to connect more easily to their care team members. In the not-so-distant future, smart hospitals with smart patient rooms and smart ORs equipped with telemedicine, AI, AR, mixed reality, and computer-aided decision-making tools will no longer be an exception.
Smart homes for smart care
Smart homes with technologies like gas detectors, movement sensors, and sleep sensors will continue to evolve. According to one estimate, the global smart home health care market was $8.7 billion in 2019, and is expected to be $96.2 billion by 2030.6

Smart technologies will have applications in fall detection and prevention, evaluation of self-administration of medicine, sleep rhythm monitoring, air quality monitoring for the detection of abnormal gas levels, and identification of things like carbon monoxide poisoning or food spoilage. In coming years, expect to see more virtual medical homes and digital health care complexes. Patients, from the convenience of their homes, might be able to connect to a suite of caregivers, all working collaboratively to provide more coordinated, effective care. The “hospital at home” model that started with six hospitals has already grown to over 100 hospitals across 29 states. The shift from onsite specialists to onscreen specialists will continue, providing greater access to specialized services.
With these emerging trends, it can be anticipated that much acute care will be provided to patients outside the hospital – either under the hospital at home model, via drone technology using telemedicine, through smart devices in smart homes, or via wearables and artificial intelligence. Hence, hospitals’ configuration in the future will be much different and more compact than currently, and many hospitals will be reserved for trauma patients, casualties of natural disasters, higher acuity diseases requiring complex procedures, and other emergencies.
The role of hospitalists has evolved over the years and is still evolving. It should be no surprise if, in the future, we work alongside a digital hospitalist twin to provide better and more personalized care to our patients. Change is uncomfortable but it is inevitable. When COVID hit, we were forced to find innovative ways to deliver care to our patients. One thing is for certain: post-pandemic (AD, or After Disease) we are not going back to a Before COVID (BC) state in terms of virtual care. With the new dawn of digital era, the crucial questions to address will be: What will the future role of a hospitalist look like? How can we leverage technology and embrace our flexibility to adapt to these trends? How can we apply the lessons learned during the pandemic to propel hospital medicine into the future? And is it time to rethink our role and even reclassify ourselves – from hospitalists to Acute Care Experts (ACE) or Primary Acute Care Physicians?
Dr. Zia is a hospitalist, physician advisor, and founder of Virtual Hospitalist - a telemedicine company with a 360-degree care model for hospital patients.
References
1. www.subjectwell.com/news/data-shows-a-majority-of-patients-remain-interested-in-clinical-trials-during-the-coronavirus-pandemic/
2. ww2.frost.com/news/press-releases/technology-innovations-and-virtual-consultations-drive-healthcare-2025/
3. www.gminsights.com/industry-analysis/telemedicine-market
4. www.healthcareitnews.com/blog/frost-sullivans-top-10-predictions-healthcare-2021
5. www.uc.edu/news/articles/2021/03/virtual-medicine--new-uc-telehealth-drone-makes-house-calls.html
6. www.psmarketresearch.com/market-analysis/smart-home-healthcare-market
People often overestimate what will happen in the next two years and underestimate what will happen in ten. – Bill Gates
The COVID-19 pandemic set in motion a series of innovations catalyzing the digital transformation of the health care landscape.
Telemedicine use exploded over the last 12 months to the point that it has almost become ubiquitous. With that, we saw a rapid proliferation of wearables and remote patient monitoring devices. Thanks to virtual care, care delivery is no longer strictly dependent on having onsite specialists, and care itself is not confined to the boundaries of hospitals or doctors’ offices anymore.
We saw the formation of the digital front door and the emergence of new virtual care sites like virtual urgent care, virtual home health, virtual office visits, virtual hospital at home that allowed clinical care to be delivered safely outside the boundaries of hospitals. Nonclinical public places like gyms, schools, and community centers were being transformed into virtual health care portals that brought care closer to the people.
Inside the hospital, we saw a fusion of traditional inpatient care and virtual care. Onsite hospital teams embraced telemedicine during the pandemic for various reasons; to conserve personal protective equipment (PPE), limit exposure, boost care capacity, improve access to specialists at distant sites, and bring family memberse to “webside” who cannot be at a patient’s bedside.
In clinical trials as well, virtual care is a welcome change. According to one survey1, most trial participants favored the use of telehealth services for clinical trials, as these helped them stay engaged, compliant, monitored, and on track while remaining at home. Furthermore, we are seeing the integration of artificial intelligence (AI) into telehealth, whether it is to aid physicians in clinical decision-making or to generate reminders to help patients with chronic disease management. However, this integration is only beginning to scratch the surface of the combination of two technologies’ real potential.
What’s next?
Based on these trends, it should be no surprise that digital health will become a vital sign for health care organizations.
The next 12 to 24 months will set new standards for digital health and play a significant role in defining the next generation of virtual care. There are projections that global health care industry revenues will exceed $2.6 trillion by 2025, with AI and telehealth playing a prominent role in this growth.2 According to estimates, telehealth itself will be a $175 billion market by 2026 and approximately one in three patient encounters will go virtual.3,4 Moreover, virtual care will continue to make exciting transformations, helping to make quality care accessible to everyone in innovative ways. For example, the University of Cincinnati has recently developed a pilot project using a drone equipped with video technology, artificial intelligence, sensors, and first aid kits to go to hard-to-reach areas to deliver care via telemedicine.5
Smart hospitals
In coming years, we can expect the integration of AI, augmented reality (AR), and virtual reality (VR) into telemedicine at lightning speed – and at a much larger scale – that will enable surgeons from different parts of the globe to perform procedures remotely and more precisely.

AI is already gaining traction in different fields within health care – whether it’s predicting length of stay in the ICU, or assisting in triage decisions, or reading radiological images, to name just a few. The Mayo Clinic is using AI and computer-aided decision-making tools to predict the risk of surgery and potential post-op complications, which could allow even better collaboration between medical and surgical teams. We hear about the “X-ray” vision offered to proceduralists using HoloLens – mixed reality smartglasses – a technology that enables them to perform procedures more precisely. Others project that there will be more sensors and voice recognition tools in the OR that will be used to gather data to develop intelligent algorithms, and to build a safety net for interventionalists that can notify them of potential hazards or accidental sterile field breaches. The insights gained will be used to create best practices and even allow some procedures to be performed outside the traditional OR setting.
Additionally, we are seeing the development of “smart” patient rooms. For example, one health system in Florida is working on deploying Amazon Alexa in 2,500 patient rooms to allow patients to connect more easily to their care team members. In the not-so-distant future, smart hospitals with smart patient rooms and smart ORs equipped with telemedicine, AI, AR, mixed reality, and computer-aided decision-making tools will no longer be an exception.
Smart homes for smart care
Smart homes with technologies like gas detectors, movement sensors, and sleep sensors will continue to evolve. According to one estimate, the global smart home health care market was $8.7 billion in 2019, and is expected to be $96.2 billion by 2030.6

Smart technologies will have applications in fall detection and prevention, evaluation of self-administration of medicine, sleep rhythm monitoring, air quality monitoring for the detection of abnormal gas levels, and identification of things like carbon monoxide poisoning or food spoilage. In coming years, expect to see more virtual medical homes and digital health care complexes. Patients, from the convenience of their homes, might be able to connect to a suite of caregivers, all working collaboratively to provide more coordinated, effective care. The “hospital at home” model that started with six hospitals has already grown to over 100 hospitals across 29 states. The shift from onsite specialists to onscreen specialists will continue, providing greater access to specialized services.
With these emerging trends, it can be anticipated that much acute care will be provided to patients outside the hospital – either under the hospital at home model, via drone technology using telemedicine, through smart devices in smart homes, or via wearables and artificial intelligence. Hence, hospitals’ configuration in the future will be much different and more compact than currently, and many hospitals will be reserved for trauma patients, casualties of natural disasters, higher acuity diseases requiring complex procedures, and other emergencies.
The role of hospitalists has evolved over the years and is still evolving. It should be no surprise if, in the future, we work alongside a digital hospitalist twin to provide better and more personalized care to our patients. Change is uncomfortable but it is inevitable. When COVID hit, we were forced to find innovative ways to deliver care to our patients. One thing is for certain: post-pandemic (AD, or After Disease) we are not going back to a Before COVID (BC) state in terms of virtual care. With the new dawn of digital era, the crucial questions to address will be: What will the future role of a hospitalist look like? How can we leverage technology and embrace our flexibility to adapt to these trends? How can we apply the lessons learned during the pandemic to propel hospital medicine into the future? And is it time to rethink our role and even reclassify ourselves – from hospitalists to Acute Care Experts (ACE) or Primary Acute Care Physicians?
Dr. Zia is a hospitalist, physician advisor, and founder of Virtual Hospitalist - a telemedicine company with a 360-degree care model for hospital patients.
References
1. www.subjectwell.com/news/data-shows-a-majority-of-patients-remain-interested-in-clinical-trials-during-the-coronavirus-pandemic/
2. ww2.frost.com/news/press-releases/technology-innovations-and-virtual-consultations-drive-healthcare-2025/
3. www.gminsights.com/industry-analysis/telemedicine-market
4. www.healthcareitnews.com/blog/frost-sullivans-top-10-predictions-healthcare-2021
5. www.uc.edu/news/articles/2021/03/virtual-medicine--new-uc-telehealth-drone-makes-house-calls.html
6. www.psmarketresearch.com/market-analysis/smart-home-healthcare-market
Stop using Neutrogena and Aveeno spray sunscreen, J&J warns
Benzene is not an ingredient of sunscreen, and should not be present in these products. The levels detected were low and would not be expected to have an adverse effect on health, but the company says it is recalling the products anyway “out of an abundance of caution.”
The sunscreen products that have been recalled are:
- NEUTROGENA® Beach Defense® aerosol sunscreen.
- NEUTROGENA® Cool Dry Sport aerosol sunscreen.
- NEUTROGENA® Invisible Daily™ defense aerosol sunscreen.
- NEUTROGENA® Ultra Sheer® aerosol sunscreen.
- AVEENO® Protect + Refresh aerosol sunscreen.
These products were distributed nationwide through a variety of retail stores. Consumers should stop using these products and throw them away, the company said.
At the same time, it emphasized the importance of using alternative sunscreen products to protect the skin from excessive sun exposure, which can lead to skin cancer including melanoma.
Johnson & Johnson has launched an investigation into how benzene got into these products.
One of the company’s other spray sunscreen products, Neutrogena Wet Skin, was not included in the recall.
Recently, benzene was found in 78 widely-used sunscreen products in tests conducted by the online pharmacy and laboratory Valisure. Most of the products were aerosol sprays, and the company called on the Food and Drug Administration to recall them all.
That petition suggested that the finding of benzene was the result of contamination somewhere in the manufacturing process.
“This isn’t a sunscreen issue, it’s a manufacturing issue,” said Adam Friedman, MD, professor and chief of dermatology at George Washington University. “We don’t want those things to be blurred.”
There is a risk that people take away the wrong message from these findings.
“People already have ambivalence about sunscreen, and this is just going to make that worse,” Dr. Friedman said in an interview.
He pointed out that benzene is present in car exhaust, second-hand smoke, and elsewhere. Inhalation exposure has been the primary focus of toxicology investigations, as has exposure from things such as contaminated drinking water – not via topical application. “We don’t know how effectively [benzene] gets through the skin, if it gets absorbed systemically, and how that then behaves downstream,” he noted.
On the other hand, ultraviolet radiation is a well-established carcinogen. Avoiding an effective preventive measure such as sunscreen could prove more harmful than exposure to trace amounts of benzene, he said.
A version of this article first appeared on WebMD.com.
Benzene is not an ingredient of sunscreen, and should not be present in these products. The levels detected were low and would not be expected to have an adverse effect on health, but the company says it is recalling the products anyway “out of an abundance of caution.”
The sunscreen products that have been recalled are:
- NEUTROGENA® Beach Defense® aerosol sunscreen.
- NEUTROGENA® Cool Dry Sport aerosol sunscreen.
- NEUTROGENA® Invisible Daily™ defense aerosol sunscreen.
- NEUTROGENA® Ultra Sheer® aerosol sunscreen.
- AVEENO® Protect + Refresh aerosol sunscreen.
These products were distributed nationwide through a variety of retail stores. Consumers should stop using these products and throw them away, the company said.
At the same time, it emphasized the importance of using alternative sunscreen products to protect the skin from excessive sun exposure, which can lead to skin cancer including melanoma.
Johnson & Johnson has launched an investigation into how benzene got into these products.
One of the company’s other spray sunscreen products, Neutrogena Wet Skin, was not included in the recall.
Recently, benzene was found in 78 widely-used sunscreen products in tests conducted by the online pharmacy and laboratory Valisure. Most of the products were aerosol sprays, and the company called on the Food and Drug Administration to recall them all.
That petition suggested that the finding of benzene was the result of contamination somewhere in the manufacturing process.
“This isn’t a sunscreen issue, it’s a manufacturing issue,” said Adam Friedman, MD, professor and chief of dermatology at George Washington University. “We don’t want those things to be blurred.”
There is a risk that people take away the wrong message from these findings.
“People already have ambivalence about sunscreen, and this is just going to make that worse,” Dr. Friedman said in an interview.
He pointed out that benzene is present in car exhaust, second-hand smoke, and elsewhere. Inhalation exposure has been the primary focus of toxicology investigations, as has exposure from things such as contaminated drinking water – not via topical application. “We don’t know how effectively [benzene] gets through the skin, if it gets absorbed systemically, and how that then behaves downstream,” he noted.
On the other hand, ultraviolet radiation is a well-established carcinogen. Avoiding an effective preventive measure such as sunscreen could prove more harmful than exposure to trace amounts of benzene, he said.
A version of this article first appeared on WebMD.com.
Benzene is not an ingredient of sunscreen, and should not be present in these products. The levels detected were low and would not be expected to have an adverse effect on health, but the company says it is recalling the products anyway “out of an abundance of caution.”
The sunscreen products that have been recalled are:
- NEUTROGENA® Beach Defense® aerosol sunscreen.
- NEUTROGENA® Cool Dry Sport aerosol sunscreen.
- NEUTROGENA® Invisible Daily™ defense aerosol sunscreen.
- NEUTROGENA® Ultra Sheer® aerosol sunscreen.
- AVEENO® Protect + Refresh aerosol sunscreen.
These products were distributed nationwide through a variety of retail stores. Consumers should stop using these products and throw them away, the company said.
At the same time, it emphasized the importance of using alternative sunscreen products to protect the skin from excessive sun exposure, which can lead to skin cancer including melanoma.
Johnson & Johnson has launched an investigation into how benzene got into these products.
One of the company’s other spray sunscreen products, Neutrogena Wet Skin, was not included in the recall.
Recently, benzene was found in 78 widely-used sunscreen products in tests conducted by the online pharmacy and laboratory Valisure. Most of the products were aerosol sprays, and the company called on the Food and Drug Administration to recall them all.
That petition suggested that the finding of benzene was the result of contamination somewhere in the manufacturing process.
“This isn’t a sunscreen issue, it’s a manufacturing issue,” said Adam Friedman, MD, professor and chief of dermatology at George Washington University. “We don’t want those things to be blurred.”
There is a risk that people take away the wrong message from these findings.
“People already have ambivalence about sunscreen, and this is just going to make that worse,” Dr. Friedman said in an interview.
He pointed out that benzene is present in car exhaust, second-hand smoke, and elsewhere. Inhalation exposure has been the primary focus of toxicology investigations, as has exposure from things such as contaminated drinking water – not via topical application. “We don’t know how effectively [benzene] gets through the skin, if it gets absorbed systemically, and how that then behaves downstream,” he noted.
On the other hand, ultraviolet radiation is a well-established carcinogen. Avoiding an effective preventive measure such as sunscreen could prove more harmful than exposure to trace amounts of benzene, he said.
A version of this article first appeared on WebMD.com.
Florida-based doctor arrested in Haiti president’s assassination
About two dozen people have been arrested as suspects, the newspaper reported, though police believe Christian Emmanuel Sanon, 63, was plotting to become president.
“He arrived by private plane in June with political objectives and contacted a private security firm to recruit the people who committed this act,” Léon Charles, Haiti’s national police chief, said during a news conference on July 11.
The firm, called CTU Security, is a Venezuelan company based in Miami, Mr. Charles said. During a raid at Mr. Sanon’s home in Port-au-Prince, police found six rifles, 20 boxes of bullets, 24 unused shooting targets, pistol holsters, and a hat with a U.S. Drug Enforcement Agency logo.
“This initial mission that was given to these assailants was to protect the individual named Emmanuel Sanon, but afterwards, the mission changed,” Mr. Charles said.
The new “mission” was to arrest President Moïse and install Mr. Sanon as president, The New York Times reported, though Mr. Charles didn’t explain when the mission changed to assassination or how Mr. Sanon could have taken control of the government.
President Moïse was shot to death on July 7 at his home in Port-au-Prince by a “team of commandos,” according to The Washington Post. On July 9, Haiti asked the U.S. to send troops to the country to protect its airport and key infrastructure.
The announcement of Mr. Sanon’s arrest came hours after FBI and Department of Homeland Security officials arrived in Haiti on July 11 to discuss how the U.S. can offer assistance, the newspaper reported.
Mr. Sanon has a YouTube channel with three political campaign videos from 2011, which include discussions about Haitian politics, according to Forbes. In one of the videos, titled “Dr. Christian Sanon – Leadership for Haiti,” Mr. Sanon talks about corruption in the country and presents himself as a potential leader.
Mr. Sanon lived in Florida for more than 20 years, ranging from the Tampa Bay area to South Florida, according to the Miami Herald. Public records show that he had more than a dozen businesses registered in the state, including medical services and real estate, though most are inactive.
Mr. Sanon is the third person with links to the U.S. who has been arrested in connection with the assassination, the Miami Herald reported. Two Haitian-Americans from southern Florida – James Solages, 35, and Joseph G. Vincent, 55 – were arrested by local police. They claimed they were working as translators for the assassins.
The first lady, Martine Moïse, was wounded in the attack and is now receiving treatment at a hospital in Miami, the newspaper reported.
A version of this article first appeared on WebMD.com.
About two dozen people have been arrested as suspects, the newspaper reported, though police believe Christian Emmanuel Sanon, 63, was plotting to become president.
“He arrived by private plane in June with political objectives and contacted a private security firm to recruit the people who committed this act,” Léon Charles, Haiti’s national police chief, said during a news conference on July 11.
The firm, called CTU Security, is a Venezuelan company based in Miami, Mr. Charles said. During a raid at Mr. Sanon’s home in Port-au-Prince, police found six rifles, 20 boxes of bullets, 24 unused shooting targets, pistol holsters, and a hat with a U.S. Drug Enforcement Agency logo.
“This initial mission that was given to these assailants was to protect the individual named Emmanuel Sanon, but afterwards, the mission changed,” Mr. Charles said.
The new “mission” was to arrest President Moïse and install Mr. Sanon as president, The New York Times reported, though Mr. Charles didn’t explain when the mission changed to assassination or how Mr. Sanon could have taken control of the government.
President Moïse was shot to death on July 7 at his home in Port-au-Prince by a “team of commandos,” according to The Washington Post. On July 9, Haiti asked the U.S. to send troops to the country to protect its airport and key infrastructure.
The announcement of Mr. Sanon’s arrest came hours after FBI and Department of Homeland Security officials arrived in Haiti on July 11 to discuss how the U.S. can offer assistance, the newspaper reported.
Mr. Sanon has a YouTube channel with three political campaign videos from 2011, which include discussions about Haitian politics, according to Forbes. In one of the videos, titled “Dr. Christian Sanon – Leadership for Haiti,” Mr. Sanon talks about corruption in the country and presents himself as a potential leader.
Mr. Sanon lived in Florida for more than 20 years, ranging from the Tampa Bay area to South Florida, according to the Miami Herald. Public records show that he had more than a dozen businesses registered in the state, including medical services and real estate, though most are inactive.
Mr. Sanon is the third person with links to the U.S. who has been arrested in connection with the assassination, the Miami Herald reported. Two Haitian-Americans from southern Florida – James Solages, 35, and Joseph G. Vincent, 55 – were arrested by local police. They claimed they were working as translators for the assassins.
The first lady, Martine Moïse, was wounded in the attack and is now receiving treatment at a hospital in Miami, the newspaper reported.
A version of this article first appeared on WebMD.com.
About two dozen people have been arrested as suspects, the newspaper reported, though police believe Christian Emmanuel Sanon, 63, was plotting to become president.
“He arrived by private plane in June with political objectives and contacted a private security firm to recruit the people who committed this act,” Léon Charles, Haiti’s national police chief, said during a news conference on July 11.
The firm, called CTU Security, is a Venezuelan company based in Miami, Mr. Charles said. During a raid at Mr. Sanon’s home in Port-au-Prince, police found six rifles, 20 boxes of bullets, 24 unused shooting targets, pistol holsters, and a hat with a U.S. Drug Enforcement Agency logo.
“This initial mission that was given to these assailants was to protect the individual named Emmanuel Sanon, but afterwards, the mission changed,” Mr. Charles said.
The new “mission” was to arrest President Moïse and install Mr. Sanon as president, The New York Times reported, though Mr. Charles didn’t explain when the mission changed to assassination or how Mr. Sanon could have taken control of the government.
President Moïse was shot to death on July 7 at his home in Port-au-Prince by a “team of commandos,” according to The Washington Post. On July 9, Haiti asked the U.S. to send troops to the country to protect its airport and key infrastructure.
The announcement of Mr. Sanon’s arrest came hours after FBI and Department of Homeland Security officials arrived in Haiti on July 11 to discuss how the U.S. can offer assistance, the newspaper reported.
Mr. Sanon has a YouTube channel with three political campaign videos from 2011, which include discussions about Haitian politics, according to Forbes. In one of the videos, titled “Dr. Christian Sanon – Leadership for Haiti,” Mr. Sanon talks about corruption in the country and presents himself as a potential leader.
Mr. Sanon lived in Florida for more than 20 years, ranging from the Tampa Bay area to South Florida, according to the Miami Herald. Public records show that he had more than a dozen businesses registered in the state, including medical services and real estate, though most are inactive.
Mr. Sanon is the third person with links to the U.S. who has been arrested in connection with the assassination, the Miami Herald reported. Two Haitian-Americans from southern Florida – James Solages, 35, and Joseph G. Vincent, 55 – were arrested by local police. They claimed they were working as translators for the assassins.
The first lady, Martine Moïse, was wounded in the attack and is now receiving treatment at a hospital in Miami, the newspaper reported.
A version of this article first appeared on WebMD.com.
Metformin use may curb BCC risk
in Iceland.
“In addition to general anticarcinogenic effects, metformin has also been shown to directly inhibit the sonic hedgehog pathway, a key pathway in basal cell carcinoma (BCC) pathogenesis,” Jonas A. Adalsteinsson, MD, of the University of Iceland, Reykjavik, and colleagues wrote. “The relationship between metformin and keratinocyte carcinoma has not been well-characterized but is of importance considering that metformin is a commonly prescribed medication.”
They added that the hedgehog pathway inhibitors vismodegib (Erivedge) and sonidegib (Odomzo), approved for treating BCC, “are highly effective for BCC prevention, but their broad use for BCC prophylaxis is limited due to numerous side effects.”
In the study, published in the Journal of the American Academy of Dermatology, the researchers identified 6,880 first-time cancer patients with BCC, squamous cell carcinoma in situ (SCCis), or invasive SCC, and 69,620 population controls using data from the Icelandic Cancer Registry and the Icelandic Prescription Medicine Register between 2003 and 2017. Metformin exposure was defined as having filled at least one prescription of metformin more than 2 years prior to cancer diagnosis. They used grams and daily dose units of metformin in their analysis; one DDU of metformin, “or its average daily maintenance dose when used for its primary indication, is 2 grams,” they noted.
Overall, metformin use was associated with a significantly lower risk of developing BCC, compared with nonuse (adjusted odds ratio, 0.71; 95% confidence interval, 0.61-0.83).
The reduced risk occurred similarly across age and gender subgroups, with the exception of individuals younger than 60 years, the researchers said. “This might signify that metformin has less of a protective effect in younger individuals, but we might also have lacked power in this category.” The association with reduced BCC risk remained significant at all three cumulative dose levels measured: 1-500 DDUs, 501-1,500 DDUs, and more than 1,500 DDUs.
Metformin use was not significantly associated with reduced risk of invasive SCC (aOR, 1.01) and in most cases of SCCis. However, the 501-1,500 DDU dose category was associated with a slight increase in risk of SCCis (aOR, 1.40; 95% CI, 1.00-1.96), “showing a possible increased risk of SCCis,” the authors wrote.
The decrease in BCC risk was seen across all metformin dosing levels, but the reason for this remains unclear, and might be related to a confounding factor that was not considered in this study, the researchers said. “It could also be that metformin’s BCC risk-lowering effect is immediate, with only a low dose being needed to see a clinical benefit.”
The study findings were limited by several factors, including the retrospective design and the inability to adjust for factors including ultraviolet exposure, Fitzpatrick skin type, and comorbidities. The frequent use of metformin by people with type 2 diabetes suggests diabetes itself or other diabetes medications could be possible confounding factors, the researchers wrote.
However, the results were strengthened by the large study population, and the data suggest an association between reduced risk of first-time BCC and metformin use, they added.
“Randomized, prospective trials are required to fully understand the effect metformin has on BCC and SCC risk,” the researchers concluded.
“There is a dire need to reduce incidence of skin cancers in general, and consequently a need for new non-surgical treatment options for keratinocytic nonmelanoma skin cancers,” Amor Khachemoune, MD, a dermatologist at the State University of New York, Brooklyn, and the department of dermatology of the Veteran Affairs NY Harbor Healthcare System, also in Brooklyn, said in an interview.
Dr. Khachemoune, who was not involved with the study, said that he was not surprised by the findings. “Like other well-studied sonic hedgehog inhibitors, vismodegib and sonidegib, metformin has a demonstrated effect on this pathway. The medical community outside of dermatology has extensive experience with the use of metformin for a host of other indications, including its role as anticarcinogenic, so it seemed natural that one would consider widening its use to quell the ever-expanding cases of basal cell carcinomas.”
However, complications from long-term use, though likely rare, could be a limitation in using metformin as a chemoprotective agent, Dr. Khachemoune said. Metformin-associated lactic acidosis is one example of a rare, but potentially life-threatening adverse event.
“Finding the right dosage and having an algorithm for follow up monitoring of side effects would certainly need to be put in place in a standardized way,” he emphasized. “As stated by the authors of this study, more inclusive research involving other groups with nonkeratinocytic malignancies in larger cohorts is needed.”
The study received no outside funding. The researchers and Dr. Khachemoune had no financial conflicts to disclose.
in Iceland.
“In addition to general anticarcinogenic effects, metformin has also been shown to directly inhibit the sonic hedgehog pathway, a key pathway in basal cell carcinoma (BCC) pathogenesis,” Jonas A. Adalsteinsson, MD, of the University of Iceland, Reykjavik, and colleagues wrote. “The relationship between metformin and keratinocyte carcinoma has not been well-characterized but is of importance considering that metformin is a commonly prescribed medication.”
They added that the hedgehog pathway inhibitors vismodegib (Erivedge) and sonidegib (Odomzo), approved for treating BCC, “are highly effective for BCC prevention, but their broad use for BCC prophylaxis is limited due to numerous side effects.”
In the study, published in the Journal of the American Academy of Dermatology, the researchers identified 6,880 first-time cancer patients with BCC, squamous cell carcinoma in situ (SCCis), or invasive SCC, and 69,620 population controls using data from the Icelandic Cancer Registry and the Icelandic Prescription Medicine Register between 2003 and 2017. Metformin exposure was defined as having filled at least one prescription of metformin more than 2 years prior to cancer diagnosis. They used grams and daily dose units of metformin in their analysis; one DDU of metformin, “or its average daily maintenance dose when used for its primary indication, is 2 grams,” they noted.
Overall, metformin use was associated with a significantly lower risk of developing BCC, compared with nonuse (adjusted odds ratio, 0.71; 95% confidence interval, 0.61-0.83).
The reduced risk occurred similarly across age and gender subgroups, with the exception of individuals younger than 60 years, the researchers said. “This might signify that metformin has less of a protective effect in younger individuals, but we might also have lacked power in this category.” The association with reduced BCC risk remained significant at all three cumulative dose levels measured: 1-500 DDUs, 501-1,500 DDUs, and more than 1,500 DDUs.
Metformin use was not significantly associated with reduced risk of invasive SCC (aOR, 1.01) and in most cases of SCCis. However, the 501-1,500 DDU dose category was associated with a slight increase in risk of SCCis (aOR, 1.40; 95% CI, 1.00-1.96), “showing a possible increased risk of SCCis,” the authors wrote.
The decrease in BCC risk was seen across all metformin dosing levels, but the reason for this remains unclear, and might be related to a confounding factor that was not considered in this study, the researchers said. “It could also be that metformin’s BCC risk-lowering effect is immediate, with only a low dose being needed to see a clinical benefit.”
The study findings were limited by several factors, including the retrospective design and the inability to adjust for factors including ultraviolet exposure, Fitzpatrick skin type, and comorbidities. The frequent use of metformin by people with type 2 diabetes suggests diabetes itself or other diabetes medications could be possible confounding factors, the researchers wrote.
However, the results were strengthened by the large study population, and the data suggest an association between reduced risk of first-time BCC and metformin use, they added.
“Randomized, prospective trials are required to fully understand the effect metformin has on BCC and SCC risk,” the researchers concluded.
“There is a dire need to reduce incidence of skin cancers in general, and consequently a need for new non-surgical treatment options for keratinocytic nonmelanoma skin cancers,” Amor Khachemoune, MD, a dermatologist at the State University of New York, Brooklyn, and the department of dermatology of the Veteran Affairs NY Harbor Healthcare System, also in Brooklyn, said in an interview.
Dr. Khachemoune, who was not involved with the study, said that he was not surprised by the findings. “Like other well-studied sonic hedgehog inhibitors, vismodegib and sonidegib, metformin has a demonstrated effect on this pathway. The medical community outside of dermatology has extensive experience with the use of metformin for a host of other indications, including its role as anticarcinogenic, so it seemed natural that one would consider widening its use to quell the ever-expanding cases of basal cell carcinomas.”
However, complications from long-term use, though likely rare, could be a limitation in using metformin as a chemoprotective agent, Dr. Khachemoune said. Metformin-associated lactic acidosis is one example of a rare, but potentially life-threatening adverse event.
“Finding the right dosage and having an algorithm for follow up monitoring of side effects would certainly need to be put in place in a standardized way,” he emphasized. “As stated by the authors of this study, more inclusive research involving other groups with nonkeratinocytic malignancies in larger cohorts is needed.”
The study received no outside funding. The researchers and Dr. Khachemoune had no financial conflicts to disclose.
in Iceland.
“In addition to general anticarcinogenic effects, metformin has also been shown to directly inhibit the sonic hedgehog pathway, a key pathway in basal cell carcinoma (BCC) pathogenesis,” Jonas A. Adalsteinsson, MD, of the University of Iceland, Reykjavik, and colleagues wrote. “The relationship between metformin and keratinocyte carcinoma has not been well-characterized but is of importance considering that metformin is a commonly prescribed medication.”
They added that the hedgehog pathway inhibitors vismodegib (Erivedge) and sonidegib (Odomzo), approved for treating BCC, “are highly effective for BCC prevention, but their broad use for BCC prophylaxis is limited due to numerous side effects.”
In the study, published in the Journal of the American Academy of Dermatology, the researchers identified 6,880 first-time cancer patients with BCC, squamous cell carcinoma in situ (SCCis), or invasive SCC, and 69,620 population controls using data from the Icelandic Cancer Registry and the Icelandic Prescription Medicine Register between 2003 and 2017. Metformin exposure was defined as having filled at least one prescription of metformin more than 2 years prior to cancer diagnosis. They used grams and daily dose units of metformin in their analysis; one DDU of metformin, “or its average daily maintenance dose when used for its primary indication, is 2 grams,” they noted.
Overall, metformin use was associated with a significantly lower risk of developing BCC, compared with nonuse (adjusted odds ratio, 0.71; 95% confidence interval, 0.61-0.83).
The reduced risk occurred similarly across age and gender subgroups, with the exception of individuals younger than 60 years, the researchers said. “This might signify that metformin has less of a protective effect in younger individuals, but we might also have lacked power in this category.” The association with reduced BCC risk remained significant at all three cumulative dose levels measured: 1-500 DDUs, 501-1,500 DDUs, and more than 1,500 DDUs.
Metformin use was not significantly associated with reduced risk of invasive SCC (aOR, 1.01) and in most cases of SCCis. However, the 501-1,500 DDU dose category was associated with a slight increase in risk of SCCis (aOR, 1.40; 95% CI, 1.00-1.96), “showing a possible increased risk of SCCis,” the authors wrote.
The decrease in BCC risk was seen across all metformin dosing levels, but the reason for this remains unclear, and might be related to a confounding factor that was not considered in this study, the researchers said. “It could also be that metformin’s BCC risk-lowering effect is immediate, with only a low dose being needed to see a clinical benefit.”
The study findings were limited by several factors, including the retrospective design and the inability to adjust for factors including ultraviolet exposure, Fitzpatrick skin type, and comorbidities. The frequent use of metformin by people with type 2 diabetes suggests diabetes itself or other diabetes medications could be possible confounding factors, the researchers wrote.
However, the results were strengthened by the large study population, and the data suggest an association between reduced risk of first-time BCC and metformin use, they added.
“Randomized, prospective trials are required to fully understand the effect metformin has on BCC and SCC risk,” the researchers concluded.
“There is a dire need to reduce incidence of skin cancers in general, and consequently a need for new non-surgical treatment options for keratinocytic nonmelanoma skin cancers,” Amor Khachemoune, MD, a dermatologist at the State University of New York, Brooklyn, and the department of dermatology of the Veteran Affairs NY Harbor Healthcare System, also in Brooklyn, said in an interview.
Dr. Khachemoune, who was not involved with the study, said that he was not surprised by the findings. “Like other well-studied sonic hedgehog inhibitors, vismodegib and sonidegib, metformin has a demonstrated effect on this pathway. The medical community outside of dermatology has extensive experience with the use of metformin for a host of other indications, including its role as anticarcinogenic, so it seemed natural that one would consider widening its use to quell the ever-expanding cases of basal cell carcinomas.”
However, complications from long-term use, though likely rare, could be a limitation in using metformin as a chemoprotective agent, Dr. Khachemoune said. Metformin-associated lactic acidosis is one example of a rare, but potentially life-threatening adverse event.
“Finding the right dosage and having an algorithm for follow up monitoring of side effects would certainly need to be put in place in a standardized way,” he emphasized. “As stated by the authors of this study, more inclusive research involving other groups with nonkeratinocytic malignancies in larger cohorts is needed.”
The study received no outside funding. The researchers and Dr. Khachemoune had no financial conflicts to disclose.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Most U.S. adults age 50+ report good health: Survey
a nonprofit hospice/advanced illness care organization based in Virginia.
Among the respondents, 41% said their health was very good or excellent.
However, the ratings differed largely by race, employment status, and income.
Employment status was also associated with a significant difference in the way people viewed their health at the top tier and bottom tier.
The middle tier (“good” health) was reported similarly (from 33% to 37%) whether a person was employed, retired, or not employed. However, employed respondents were much more likely to report they had “excellent” or “very good” health (51% vs. 44% for retirees and 21% for the not employed).
Conversely, those who were not employed were far more likely to report “fair” or “poor” health (45%) than those who were employed (13%) or retired (20%).
Similarly, respondents with incomes of less than $50,000 were three times more likely to report their health as “fair” or “poor” than were those with incomes of more than $100,000 (36% vs. 12%).
WebMD/CCH surveyed 3,464 U.S. residents ages 50 and older between Aug. 13 and Nov. 9, 2020. WebMD.com readers were randomly invited to take a 10-minute online survey.
Aging at home a priority
The survey also highlighted a strong preference for aging in place, says Steve Cone, chief of communications and philanthropy at CCH.
“More now than ever before, thanks to the COVID experience, baby boomers and their children really believe that’s the holy grail,” he says.
Mr. Cone notes that the quick spread of COVID-19 through some nursing homes early in the pandemic likely has strengthened people’s resolve to live out their lives in their own homes.
The survey indicated that 85% of people aged 50+ who are living in their own home, a family member’s home, or a loved one’s home responded that it is “very important” or “important” to stay in their home as they age.
When asked what services they would need to continue their living situation, the most common responses were housekeeping, home repair services, and transportation (listed as needs by 35% to 45% of respondents). Regarding changes they would have to make to feel safe in their home as they age, installing grab bars and/or safety rails in the bath/shower was the most popular answer (50%).
Use of telemedicine
Respondents were also asked about their acceptance of telemedicine, and 62% said they would be likely or very likely to engage in virtual visits with a doctor it in the future.
However, the likelihood varied by income level. Specifically, respondents with incomes over $100,000 were significantly more likely to say they would use telemedicine in the future than were those with incomes below $50,000 (74% vs. 60%). They were also more likely to already have used telemedicine.
Although respondents generally embraced telemedicine, they are less confident about some types of monitoring, according to Mr. Cone.
Emergency response (64%) was the leading type of remote monitoring respondents ages 50 and older would allow. Only a minority of respondents would allow the other types of monitoring asked about in the survey.
Close to one-quarter of respondents would not allow any type of monitoring.
Fewer than one-third would allow tracking of medication compliance, refrigerator use, sleep habits, or bathroom use.
People see monitoring of some movements as “Orwellian,” Mr. Cone says.
Knowledge of hospice
The survey findings support the need for more widespread use of hospice so people can stay in their homes as they age, Mr. Cone says.
When illness gets severe, “There’s no reason you have to get rushed to the emergency room or wind up in a hospital,” Mr. Cone says.
He notes that hospice and palliative care can come to patients wherever they reside – in their home, an assisted living center, a nursing home, or even a hospital room.
“That doesn’t mean the physician isn’t involved,” he says. “But working as a team, we can keep them in their homes and their lifestyle intact.”
Patients whose doctors attest that they are likely to live a maximum 6 months are eligible for hospice. But most families wait too long to long to start hospice or palliative care for a patient, Mr. Cone says, and may not be aware of what these services typically cover, including meal preparation and pet care.
In the survey, nearly one-third of respondents said they did not know that palliative care is something that “can be given at any stage of a serious illness” or “provides non-medical services (e.g., patient/family communication, help with insurance issues, scheduling appointments, arranging transportation).”
He notes palliative care and hospice are covered by Medicare and Medicaid and also by most private insurance plans or by individual companies providing the service.
However, health care providers may have to overcome a general reluctance to discuss hospice when sharing options for those severely ill.
The survey showed that while 51% of those 50 and older are at least “slightly interested” in learning more about hospice, a nearly equal number say they are “not at all interested” (49%).
Most using hospice are White
More than 90% of those surveyed reported that aspects of hospice care, including “comfort and relief from pain at the end of patients’ lives,” providing a dedicated care team, and an alternative to other care settings, are “very important” or “important.”
However, national hospice use rates are extremely low for minorities and the LGBTQ community, according to Mr. Cone. Among Medicare hospice recipients, 82% were white, 8.2% Black, 6.7% Hispanic, and 1.8% Asian or Pacific Islander, according to the National Hospice and Palliative Care Organization.
Those numbers signal a need for outreach to those communities with information on what services are available and how to access them, he says.
Health costs top concern
The survey also asked about level of concern regarding matters including family, health, financials, and end-of-life directives and found adults aged 50 and older expressed the greatest amount of concern for health care costs that are not covered by insurance.
More than half (56%) said they were concerned or very concerned about those costs, which was higher than the percentage concerned about losing a spouse (49%).
Respondents were less concerned (“slightly concerned” or “not at all concerned”) about their children living far away, planning end-of life-directives, and falling or having reduced mobility.
A version of this article first appeared on WebMD.com.
a nonprofit hospice/advanced illness care organization based in Virginia.
Among the respondents, 41% said their health was very good or excellent.
However, the ratings differed largely by race, employment status, and income.
Employment status was also associated with a significant difference in the way people viewed their health at the top tier and bottom tier.
The middle tier (“good” health) was reported similarly (from 33% to 37%) whether a person was employed, retired, or not employed. However, employed respondents were much more likely to report they had “excellent” or “very good” health (51% vs. 44% for retirees and 21% for the not employed).
Conversely, those who were not employed were far more likely to report “fair” or “poor” health (45%) than those who were employed (13%) or retired (20%).
Similarly, respondents with incomes of less than $50,000 were three times more likely to report their health as “fair” or “poor” than were those with incomes of more than $100,000 (36% vs. 12%).
WebMD/CCH surveyed 3,464 U.S. residents ages 50 and older between Aug. 13 and Nov. 9, 2020. WebMD.com readers were randomly invited to take a 10-minute online survey.
Aging at home a priority
The survey also highlighted a strong preference for aging in place, says Steve Cone, chief of communications and philanthropy at CCH.
“More now than ever before, thanks to the COVID experience, baby boomers and their children really believe that’s the holy grail,” he says.
Mr. Cone notes that the quick spread of COVID-19 through some nursing homes early in the pandemic likely has strengthened people’s resolve to live out their lives in their own homes.
The survey indicated that 85% of people aged 50+ who are living in their own home, a family member’s home, or a loved one’s home responded that it is “very important” or “important” to stay in their home as they age.
When asked what services they would need to continue their living situation, the most common responses were housekeeping, home repair services, and transportation (listed as needs by 35% to 45% of respondents). Regarding changes they would have to make to feel safe in their home as they age, installing grab bars and/or safety rails in the bath/shower was the most popular answer (50%).
Use of telemedicine
Respondents were also asked about their acceptance of telemedicine, and 62% said they would be likely or very likely to engage in virtual visits with a doctor it in the future.
However, the likelihood varied by income level. Specifically, respondents with incomes over $100,000 were significantly more likely to say they would use telemedicine in the future than were those with incomes below $50,000 (74% vs. 60%). They were also more likely to already have used telemedicine.
Although respondents generally embraced telemedicine, they are less confident about some types of monitoring, according to Mr. Cone.
Emergency response (64%) was the leading type of remote monitoring respondents ages 50 and older would allow. Only a minority of respondents would allow the other types of monitoring asked about in the survey.
Close to one-quarter of respondents would not allow any type of monitoring.
Fewer than one-third would allow tracking of medication compliance, refrigerator use, sleep habits, or bathroom use.
People see monitoring of some movements as “Orwellian,” Mr. Cone says.
Knowledge of hospice
The survey findings support the need for more widespread use of hospice so people can stay in their homes as they age, Mr. Cone says.
When illness gets severe, “There’s no reason you have to get rushed to the emergency room or wind up in a hospital,” Mr. Cone says.
He notes that hospice and palliative care can come to patients wherever they reside – in their home, an assisted living center, a nursing home, or even a hospital room.
“That doesn’t mean the physician isn’t involved,” he says. “But working as a team, we can keep them in their homes and their lifestyle intact.”
Patients whose doctors attest that they are likely to live a maximum 6 months are eligible for hospice. But most families wait too long to long to start hospice or palliative care for a patient, Mr. Cone says, and may not be aware of what these services typically cover, including meal preparation and pet care.
In the survey, nearly one-third of respondents said they did not know that palliative care is something that “can be given at any stage of a serious illness” or “provides non-medical services (e.g., patient/family communication, help with insurance issues, scheduling appointments, arranging transportation).”
He notes palliative care and hospice are covered by Medicare and Medicaid and also by most private insurance plans or by individual companies providing the service.
However, health care providers may have to overcome a general reluctance to discuss hospice when sharing options for those severely ill.
The survey showed that while 51% of those 50 and older are at least “slightly interested” in learning more about hospice, a nearly equal number say they are “not at all interested” (49%).
Most using hospice are White
More than 90% of those surveyed reported that aspects of hospice care, including “comfort and relief from pain at the end of patients’ lives,” providing a dedicated care team, and an alternative to other care settings, are “very important” or “important.”
However, national hospice use rates are extremely low for minorities and the LGBTQ community, according to Mr. Cone. Among Medicare hospice recipients, 82% were white, 8.2% Black, 6.7% Hispanic, and 1.8% Asian or Pacific Islander, according to the National Hospice and Palliative Care Organization.
Those numbers signal a need for outreach to those communities with information on what services are available and how to access them, he says.
Health costs top concern
The survey also asked about level of concern regarding matters including family, health, financials, and end-of-life directives and found adults aged 50 and older expressed the greatest amount of concern for health care costs that are not covered by insurance.
More than half (56%) said they were concerned or very concerned about those costs, which was higher than the percentage concerned about losing a spouse (49%).
Respondents were less concerned (“slightly concerned” or “not at all concerned”) about their children living far away, planning end-of life-directives, and falling or having reduced mobility.
A version of this article first appeared on WebMD.com.
a nonprofit hospice/advanced illness care organization based in Virginia.
Among the respondents, 41% said their health was very good or excellent.
However, the ratings differed largely by race, employment status, and income.
Employment status was also associated with a significant difference in the way people viewed their health at the top tier and bottom tier.
The middle tier (“good” health) was reported similarly (from 33% to 37%) whether a person was employed, retired, or not employed. However, employed respondents were much more likely to report they had “excellent” or “very good” health (51% vs. 44% for retirees and 21% for the not employed).
Conversely, those who were not employed were far more likely to report “fair” or “poor” health (45%) than those who were employed (13%) or retired (20%).
Similarly, respondents with incomes of less than $50,000 were three times more likely to report their health as “fair” or “poor” than were those with incomes of more than $100,000 (36% vs. 12%).
WebMD/CCH surveyed 3,464 U.S. residents ages 50 and older between Aug. 13 and Nov. 9, 2020. WebMD.com readers were randomly invited to take a 10-minute online survey.
Aging at home a priority
The survey also highlighted a strong preference for aging in place, says Steve Cone, chief of communications and philanthropy at CCH.
“More now than ever before, thanks to the COVID experience, baby boomers and their children really believe that’s the holy grail,” he says.
Mr. Cone notes that the quick spread of COVID-19 through some nursing homes early in the pandemic likely has strengthened people’s resolve to live out their lives in their own homes.
The survey indicated that 85% of people aged 50+ who are living in their own home, a family member’s home, or a loved one’s home responded that it is “very important” or “important” to stay in their home as they age.
When asked what services they would need to continue their living situation, the most common responses were housekeeping, home repair services, and transportation (listed as needs by 35% to 45% of respondents). Regarding changes they would have to make to feel safe in their home as they age, installing grab bars and/or safety rails in the bath/shower was the most popular answer (50%).
Use of telemedicine
Respondents were also asked about their acceptance of telemedicine, and 62% said they would be likely or very likely to engage in virtual visits with a doctor it in the future.
However, the likelihood varied by income level. Specifically, respondents with incomes over $100,000 were significantly more likely to say they would use telemedicine in the future than were those with incomes below $50,000 (74% vs. 60%). They were also more likely to already have used telemedicine.
Although respondents generally embraced telemedicine, they are less confident about some types of monitoring, according to Mr. Cone.
Emergency response (64%) was the leading type of remote monitoring respondents ages 50 and older would allow. Only a minority of respondents would allow the other types of monitoring asked about in the survey.
Close to one-quarter of respondents would not allow any type of monitoring.
Fewer than one-third would allow tracking of medication compliance, refrigerator use, sleep habits, or bathroom use.
People see monitoring of some movements as “Orwellian,” Mr. Cone says.
Knowledge of hospice
The survey findings support the need for more widespread use of hospice so people can stay in their homes as they age, Mr. Cone says.
When illness gets severe, “There’s no reason you have to get rushed to the emergency room or wind up in a hospital,” Mr. Cone says.
He notes that hospice and palliative care can come to patients wherever they reside – in their home, an assisted living center, a nursing home, or even a hospital room.
“That doesn’t mean the physician isn’t involved,” he says. “But working as a team, we can keep them in their homes and their lifestyle intact.”
Patients whose doctors attest that they are likely to live a maximum 6 months are eligible for hospice. But most families wait too long to long to start hospice or palliative care for a patient, Mr. Cone says, and may not be aware of what these services typically cover, including meal preparation and pet care.
In the survey, nearly one-third of respondents said they did not know that palliative care is something that “can be given at any stage of a serious illness” or “provides non-medical services (e.g., patient/family communication, help with insurance issues, scheduling appointments, arranging transportation).”
He notes palliative care and hospice are covered by Medicare and Medicaid and also by most private insurance plans or by individual companies providing the service.
However, health care providers may have to overcome a general reluctance to discuss hospice when sharing options for those severely ill.
The survey showed that while 51% of those 50 and older are at least “slightly interested” in learning more about hospice, a nearly equal number say they are “not at all interested” (49%).
Most using hospice are White
More than 90% of those surveyed reported that aspects of hospice care, including “comfort and relief from pain at the end of patients’ lives,” providing a dedicated care team, and an alternative to other care settings, are “very important” or “important.”
However, national hospice use rates are extremely low for minorities and the LGBTQ community, according to Mr. Cone. Among Medicare hospice recipients, 82% were white, 8.2% Black, 6.7% Hispanic, and 1.8% Asian or Pacific Islander, according to the National Hospice and Palliative Care Organization.
Those numbers signal a need for outreach to those communities with information on what services are available and how to access them, he says.
Health costs top concern
The survey also asked about level of concern regarding matters including family, health, financials, and end-of-life directives and found adults aged 50 and older expressed the greatest amount of concern for health care costs that are not covered by insurance.
More than half (56%) said they were concerned or very concerned about those costs, which was higher than the percentage concerned about losing a spouse (49%).
Respondents were less concerned (“slightly concerned” or “not at all concerned”) about their children living far away, planning end-of life-directives, and falling or having reduced mobility.
A version of this article first appeared on WebMD.com.
The Peer Review Process During the COVID-19 Pandemic
The COVID-19 pandemic put unparalleled strain on US health care systems and individual health care providers (HCPs), which has been well documented. Like all other medical peer reviewed journals, Federal Practitioner relies heavily on the generosity and dedication of federal HCPs. As the pandemic unfolded, we questioned whether HCPs would have the time and energy to write new articles, complete research projects, and review the work of their peers. To assess the impact of COVID-19 on the journal, we compared data from a full year during the COVID-19 pandemic with that of the previous year to determine whether and how the pandemic reshaped the peer review and publication process.
For the purposes of this review, we will compare a full year of COVID-19 journal performance with the prior year. Since COVID-19 infections spiked at different times in different places, there is no clear starting point for the pandemic. Similarly, states varied widely in their vaccination rates and opening procedures. Nevertheless, the period from May 1, 2020 to April 30, 2021, most of the country experienced COVID-19 restrictions, and the number of cases rose dramatically.
From May 1, 2020 to April 30, 2021, Federal Practitioner received 208 submissions, 110% increase over the previous year (189 submissions from May 1, 2019 to April 30, 2020) and a 28% increase over a 2-year period. After submission, it took an average of 9.0 days to the first reviewer invitation compared with 10.3 days in the previous year and 4.7 days 2 years prior. Time from the initial submission to the first decision (ie, accept, reject, or revise) took 72.8 days in the COVID-19 year compared with 91.1 days in the previous year and 69.6 days 2 years prior. In both periods it took reviewers a mean 9.5 days to complete a review from the date invited, and the rate of late reviews was unchanged as well.
During the COVID-19 pandemic year, 1481 reviewer invitations were sent to potential reviewers and 498 reviews were completed (33.6%) by 195 individual reviewers: an average of 2.4 reviews per manuscript. Most reviewers recommended to accept the manuscript, and just 14.7% of reviewers recommended to reject the manuscript (Table). The previous year 1295 invitations were sent to potential reviewers and 460 reviews were completed (38.1%) by 181 individual reviewers for an average of 2.4 reviews per manuscript.
For the original submissions, the journal accepted just 26 (12.7%) articles, recommended revisions for 105 (51.2%) submissions, and rejected 74 (36.1%) submissions from May 1, 2020 to April 30, 3021. One hundred seven manuscripts were revised once, and 75.7% were accepted, and 2.8% were rejected. Twenty-two articles had a second revision and 1 had a third revision and all were published. In the year before the pandemic, just 16 (9.5%) manuscripts were accepted in their original form and 59 (39.1%) were rejected.
Federal Practitioner published 113 articles from May 2020 to April 2021. These articles included 44 (38.9%) original studies, 25 (22.1%) case studies, 20 (17.7%) program profiles, 16 (14.2%) commentaries/editorials, and 8 (7.1%) review articles; 19 (16.8%) articles were focused on COVID-19. The prior year saw Federal Practitioner publish 106 articles in 18 issues. Of these articles 36.8% were original studies, 22.6% were program profiles, 18.9% were case studies, 13.2% were commentaries/editorials, and 8.5% were review articles.
Despite the impact of COVID-19, federal HCPs continued to contribute to this journal without significant interruption. The journal saw a 10% increase in submissions during the pandemic year compared with the previous year but that was in keeping with prior increases in submissions. Similarly, the journal saw more individual reviewers submit more total reviews from May 2020 to April 2021 compared with the previous year. The broad spectrum of reviewers involved in the process and the growing volume of both reviews and submissions suggest that our reviewers remained available and committed to the peer review process despite the impact of a pandemic.
Reducing the time to first decision remains an important priority for the journal. Although the time was shortened during the pandemic, it still took longer to inform authors of the first decision compared with 2 years before. There is no indication that COVID-19 had an impact on the speed of decision making. Reviewers were as timely during the pandemic as they were the year before.
Similarly, there was little difference in the types of articles that were published, other than the obvious increase in COVID-19 submissions. Most of the articles on COVID-19 were editorials and columns, though the journal also published case studies, program profiles, and review articles on treatment. During the pandemic, a higher percentage of articles were original studies and case reports, and fewer were program profiles compared with the types the year before. It is unclear if these differences resulted from random fluctuations in unsolicited manuscripts or are part of a larger trend. The journal managed to publish slightly more articles from May 2020 to April 2021 compared with May 2019 to April 2020 despite fewer issues. This is likely due to increased submissions and articles published online.
For the original submissions, the journal accepted just 26 (12.7%) articles, recommended revisions for 105 (51.2%) submissions and rejected 74 (36.1%) submissions from May 2020 to April 3021. One hundred seven manuscripts were revised once and 75.7% were accepted and 2.8% were rejected. Twenty-two articles had a second revision and 1 had a third revision and all were published. In the year prior to the pandemic, just 16 (9.5%) manuscripts were accepted in their original form, and 59 (39.1%) were rejected.
Although Federal Practitioner improved the efficiency of its decision making, there is still significant room for improvement. We are committed to providing our authors with more rapid decisions and reducing the time to the first decision. Seventy-two days is still too long for authors to wait to hear about the initial decision on their article. Future reviews of the publication process should focus not only on the types of articles that are included, but their subjects as well. Given the great diversity of clinical care practiced across the US Department of Veterans Affairs, US Department of Defense, and the US Public Health Service, the journal must ensure that its articles reflect its diverse audience. We would like to see articles come from authors associated with all 3 major branches of our audience, as well as small portions of the readership (eg, Federal Bureau of Prisons, National Institutes of Health) and ask our readers to help us promote the journal to potential authors in all Federal Health Care organizations. We are especially interested in submissions on or from underserved populations.
Despite the significant burdens on HCPs and federal health care systems, Federal Practitioner managed to increase the speed of publication and the number of articles between May 2020 and April 2021 thanks to the work of all the authors and reviewers who contributed their time and energy to the publication during this challenging period. Their efforts are impressive and greatly appreciated. We pledge to continue to improve our process to reduce the time to publication and to continue to provide regular updates on our process and performance.
The COVID-19 pandemic put unparalleled strain on US health care systems and individual health care providers (HCPs), which has been well documented. Like all other medical peer reviewed journals, Federal Practitioner relies heavily on the generosity and dedication of federal HCPs. As the pandemic unfolded, we questioned whether HCPs would have the time and energy to write new articles, complete research projects, and review the work of their peers. To assess the impact of COVID-19 on the journal, we compared data from a full year during the COVID-19 pandemic with that of the previous year to determine whether and how the pandemic reshaped the peer review and publication process.
For the purposes of this review, we will compare a full year of COVID-19 journal performance with the prior year. Since COVID-19 infections spiked at different times in different places, there is no clear starting point for the pandemic. Similarly, states varied widely in their vaccination rates and opening procedures. Nevertheless, the period from May 1, 2020 to April 30, 2021, most of the country experienced COVID-19 restrictions, and the number of cases rose dramatically.
From May 1, 2020 to April 30, 2021, Federal Practitioner received 208 submissions, 110% increase over the previous year (189 submissions from May 1, 2019 to April 30, 2020) and a 28% increase over a 2-year period. After submission, it took an average of 9.0 days to the first reviewer invitation compared with 10.3 days in the previous year and 4.7 days 2 years prior. Time from the initial submission to the first decision (ie, accept, reject, or revise) took 72.8 days in the COVID-19 year compared with 91.1 days in the previous year and 69.6 days 2 years prior. In both periods it took reviewers a mean 9.5 days to complete a review from the date invited, and the rate of late reviews was unchanged as well.
During the COVID-19 pandemic year, 1481 reviewer invitations were sent to potential reviewers and 498 reviews were completed (33.6%) by 195 individual reviewers: an average of 2.4 reviews per manuscript. Most reviewers recommended to accept the manuscript, and just 14.7% of reviewers recommended to reject the manuscript (Table). The previous year 1295 invitations were sent to potential reviewers and 460 reviews were completed (38.1%) by 181 individual reviewers for an average of 2.4 reviews per manuscript.
For the original submissions, the journal accepted just 26 (12.7%) articles, recommended revisions for 105 (51.2%) submissions, and rejected 74 (36.1%) submissions from May 1, 2020 to April 30, 3021. One hundred seven manuscripts were revised once, and 75.7% were accepted, and 2.8% were rejected. Twenty-two articles had a second revision and 1 had a third revision and all were published. In the year before the pandemic, just 16 (9.5%) manuscripts were accepted in their original form and 59 (39.1%) were rejected.
Federal Practitioner published 113 articles from May 2020 to April 2021. These articles included 44 (38.9%) original studies, 25 (22.1%) case studies, 20 (17.7%) program profiles, 16 (14.2%) commentaries/editorials, and 8 (7.1%) review articles; 19 (16.8%) articles were focused on COVID-19. The prior year saw Federal Practitioner publish 106 articles in 18 issues. Of these articles 36.8% were original studies, 22.6% were program profiles, 18.9% were case studies, 13.2% were commentaries/editorials, and 8.5% were review articles.
Despite the impact of COVID-19, federal HCPs continued to contribute to this journal without significant interruption. The journal saw a 10% increase in submissions during the pandemic year compared with the previous year but that was in keeping with prior increases in submissions. Similarly, the journal saw more individual reviewers submit more total reviews from May 2020 to April 2021 compared with the previous year. The broad spectrum of reviewers involved in the process and the growing volume of both reviews and submissions suggest that our reviewers remained available and committed to the peer review process despite the impact of a pandemic.
Reducing the time to first decision remains an important priority for the journal. Although the time was shortened during the pandemic, it still took longer to inform authors of the first decision compared with 2 years before. There is no indication that COVID-19 had an impact on the speed of decision making. Reviewers were as timely during the pandemic as they were the year before.
Similarly, there was little difference in the types of articles that were published, other than the obvious increase in COVID-19 submissions. Most of the articles on COVID-19 were editorials and columns, though the journal also published case studies, program profiles, and review articles on treatment. During the pandemic, a higher percentage of articles were original studies and case reports, and fewer were program profiles compared with the types the year before. It is unclear if these differences resulted from random fluctuations in unsolicited manuscripts or are part of a larger trend. The journal managed to publish slightly more articles from May 2020 to April 2021 compared with May 2019 to April 2020 despite fewer issues. This is likely due to increased submissions and articles published online.
For the original submissions, the journal accepted just 26 (12.7%) articles, recommended revisions for 105 (51.2%) submissions and rejected 74 (36.1%) submissions from May 2020 to April 3021. One hundred seven manuscripts were revised once and 75.7% were accepted and 2.8% were rejected. Twenty-two articles had a second revision and 1 had a third revision and all were published. In the year prior to the pandemic, just 16 (9.5%) manuscripts were accepted in their original form, and 59 (39.1%) were rejected.
Although Federal Practitioner improved the efficiency of its decision making, there is still significant room for improvement. We are committed to providing our authors with more rapid decisions and reducing the time to the first decision. Seventy-two days is still too long for authors to wait to hear about the initial decision on their article. Future reviews of the publication process should focus not only on the types of articles that are included, but their subjects as well. Given the great diversity of clinical care practiced across the US Department of Veterans Affairs, US Department of Defense, and the US Public Health Service, the journal must ensure that its articles reflect its diverse audience. We would like to see articles come from authors associated with all 3 major branches of our audience, as well as small portions of the readership (eg, Federal Bureau of Prisons, National Institutes of Health) and ask our readers to help us promote the journal to potential authors in all Federal Health Care organizations. We are especially interested in submissions on or from underserved populations.
Despite the significant burdens on HCPs and federal health care systems, Federal Practitioner managed to increase the speed of publication and the number of articles between May 2020 and April 2021 thanks to the work of all the authors and reviewers who contributed their time and energy to the publication during this challenging period. Their efforts are impressive and greatly appreciated. We pledge to continue to improve our process to reduce the time to publication and to continue to provide regular updates on our process and performance.
The COVID-19 pandemic put unparalleled strain on US health care systems and individual health care providers (HCPs), which has been well documented. Like all other medical peer reviewed journals, Federal Practitioner relies heavily on the generosity and dedication of federal HCPs. As the pandemic unfolded, we questioned whether HCPs would have the time and energy to write new articles, complete research projects, and review the work of their peers. To assess the impact of COVID-19 on the journal, we compared data from a full year during the COVID-19 pandemic with that of the previous year to determine whether and how the pandemic reshaped the peer review and publication process.
For the purposes of this review, we will compare a full year of COVID-19 journal performance with the prior year. Since COVID-19 infections spiked at different times in different places, there is no clear starting point for the pandemic. Similarly, states varied widely in their vaccination rates and opening procedures. Nevertheless, the period from May 1, 2020 to April 30, 2021, most of the country experienced COVID-19 restrictions, and the number of cases rose dramatically.
From May 1, 2020 to April 30, 2021, Federal Practitioner received 208 submissions, 110% increase over the previous year (189 submissions from May 1, 2019 to April 30, 2020) and a 28% increase over a 2-year period. After submission, it took an average of 9.0 days to the first reviewer invitation compared with 10.3 days in the previous year and 4.7 days 2 years prior. Time from the initial submission to the first decision (ie, accept, reject, or revise) took 72.8 days in the COVID-19 year compared with 91.1 days in the previous year and 69.6 days 2 years prior. In both periods it took reviewers a mean 9.5 days to complete a review from the date invited, and the rate of late reviews was unchanged as well.
During the COVID-19 pandemic year, 1481 reviewer invitations were sent to potential reviewers and 498 reviews were completed (33.6%) by 195 individual reviewers: an average of 2.4 reviews per manuscript. Most reviewers recommended to accept the manuscript, and just 14.7% of reviewers recommended to reject the manuscript (Table). The previous year 1295 invitations were sent to potential reviewers and 460 reviews were completed (38.1%) by 181 individual reviewers for an average of 2.4 reviews per manuscript.
For the original submissions, the journal accepted just 26 (12.7%) articles, recommended revisions for 105 (51.2%) submissions, and rejected 74 (36.1%) submissions from May 1, 2020 to April 30, 3021. One hundred seven manuscripts were revised once, and 75.7% were accepted, and 2.8% were rejected. Twenty-two articles had a second revision and 1 had a third revision and all were published. In the year before the pandemic, just 16 (9.5%) manuscripts were accepted in their original form and 59 (39.1%) were rejected.
Federal Practitioner published 113 articles from May 2020 to April 2021. These articles included 44 (38.9%) original studies, 25 (22.1%) case studies, 20 (17.7%) program profiles, 16 (14.2%) commentaries/editorials, and 8 (7.1%) review articles; 19 (16.8%) articles were focused on COVID-19. The prior year saw Federal Practitioner publish 106 articles in 18 issues. Of these articles 36.8% were original studies, 22.6% were program profiles, 18.9% were case studies, 13.2% were commentaries/editorials, and 8.5% were review articles.
Despite the impact of COVID-19, federal HCPs continued to contribute to this journal without significant interruption. The journal saw a 10% increase in submissions during the pandemic year compared with the previous year but that was in keeping with prior increases in submissions. Similarly, the journal saw more individual reviewers submit more total reviews from May 2020 to April 2021 compared with the previous year. The broad spectrum of reviewers involved in the process and the growing volume of both reviews and submissions suggest that our reviewers remained available and committed to the peer review process despite the impact of a pandemic.
Reducing the time to first decision remains an important priority for the journal. Although the time was shortened during the pandemic, it still took longer to inform authors of the first decision compared with 2 years before. There is no indication that COVID-19 had an impact on the speed of decision making. Reviewers were as timely during the pandemic as they were the year before.
Similarly, there was little difference in the types of articles that were published, other than the obvious increase in COVID-19 submissions. Most of the articles on COVID-19 were editorials and columns, though the journal also published case studies, program profiles, and review articles on treatment. During the pandemic, a higher percentage of articles were original studies and case reports, and fewer were program profiles compared with the types the year before. It is unclear if these differences resulted from random fluctuations in unsolicited manuscripts or are part of a larger trend. The journal managed to publish slightly more articles from May 2020 to April 2021 compared with May 2019 to April 2020 despite fewer issues. This is likely due to increased submissions and articles published online.
For the original submissions, the journal accepted just 26 (12.7%) articles, recommended revisions for 105 (51.2%) submissions and rejected 74 (36.1%) submissions from May 2020 to April 3021. One hundred seven manuscripts were revised once and 75.7% were accepted and 2.8% were rejected. Twenty-two articles had a second revision and 1 had a third revision and all were published. In the year prior to the pandemic, just 16 (9.5%) manuscripts were accepted in their original form, and 59 (39.1%) were rejected.
Although Federal Practitioner improved the efficiency of its decision making, there is still significant room for improvement. We are committed to providing our authors with more rapid decisions and reducing the time to the first decision. Seventy-two days is still too long for authors to wait to hear about the initial decision on their article. Future reviews of the publication process should focus not only on the types of articles that are included, but their subjects as well. Given the great diversity of clinical care practiced across the US Department of Veterans Affairs, US Department of Defense, and the US Public Health Service, the journal must ensure that its articles reflect its diverse audience. We would like to see articles come from authors associated with all 3 major branches of our audience, as well as small portions of the readership (eg, Federal Bureau of Prisons, National Institutes of Health) and ask our readers to help us promote the journal to potential authors in all Federal Health Care organizations. We are especially interested in submissions on or from underserved populations.
Despite the significant burdens on HCPs and federal health care systems, Federal Practitioner managed to increase the speed of publication and the number of articles between May 2020 and April 2021 thanks to the work of all the authors and reviewers who contributed their time and energy to the publication during this challenging period. Their efforts are impressive and greatly appreciated. We pledge to continue to improve our process to reduce the time to publication and to continue to provide regular updates on our process and performance.
Home Modifications for Rural Veterans With Disabilities
The US Department of Veterans Affairs (VA) created the Home Improvements and Structural Alterations (HISA) program to help provide necessary home modifications (HMs) to veterans with disabilities (VWDs) that will facilitate the provision of medical services at home and improve home accessibility and functional independence. The Veterans Health Administration (VHA) has more than 9 million veteran enrollees; of those, 2.7 million are classified as rural or highly rural.1 Rural veterans (RVs) possess higher rate of disability compared with that of urban veterans.2-5 RVs have unequal access to screening of ambulatory care sensitive conditions (eg, hypertension, diabetes mellitus).6 Furthermore, RVs are at risk of poor medical outcomes due to distance from health care facilities and specialist care, which can be a barrier to emergency care when issues arise. These barriers, among others, are associated with compromised health quality of life and health outcomes for RVs.3,6 The HISA program may be key to decreasing falls and other serious mishaps in the home. Therefore, understanding use of the HISA program by RVs is important. However, to date little information has been available regarding use of HISA benefits by RVs or characteristics of RVs who receive HISA benefits.
HISA Alterations Program
HISA was initially developed by VA to improve veterans’ transition from acute medical care to home.7,8 However, to obtain HISA grants currently, there is an average 3 to 6 months application process.7 Through the HISA program, VWDs can be prescribed the following HMs, including (but not limited to): flooring replacement, permanent ramps, roll-in showers, installation of central air-conditioning systems, improved lighting, kitchen/bathroom modifications, and home inspections. The HMs prescribed depend on an assessment of medical need by health care providers (HCPs).8
As time passed and the veteran population aged, the program now primarily helps ensure the ability to enter into essential areas and safety in the home.5 The amount of a HISA payment is based on whether a veteran’s health condition is related to military service as defined by the VHA service connection medical evaluation process. Barriers to obtaining a HISA HM can include difficulty in navigating the evaluation process and difficulty in finding a qualified contractor or builder to do the HM.7
This article aims to: (1) Detail the sociodemographic and clinical characteristics of rural HISA users (RHUs); (2) report on HISA usage patterns in number, types, and cost of HMs; (3) compare use amid the diverse VA medical centers (VAMCs) and related complexity levels and Veterans Integrated Service Networks (VISNs); and (4) examine the relationship between travel time/distance and HISA utilization. The long-term goal is to provide accurate information to researchers, HM administrators, health care providers and policy makers on HISA program utilization by rural VWDs, which may help improve its use and bring awareness of its users. This study was approved by the affiliate University of Florida Institutional Review Board and VA research and development committee at the North Florida/South Georgia Veterans Health System.
Methods
Data were obtained from 3 VA sources: the National Prosthetics Patient Database (NPPD), the VHA Medical Inpatient Dataset, and the VHA Outpatient Dataset.7 The NPPD is a national administrative database that contains information on prosthetic-associated products ordered by HCPs for patients, such as portable ramps, handrails, home oxygen equipment, and orthotic and prosthetic apparatus. Data obtained from the NPPD included cost of HMs, clinical characteristics, VISN, and VAMC. VA facilities are categorized into complexity levels 1a, 1b, 1c, 2, and 3. Complexity level 1a to 1c VAMCs address medical cases that entail “heightening involvedness,” meaning a larger number of patients presented with medical concerns needing medical specialists. Complexity levels 2 and 3 have fewer resources, lower patient numbers, and less medically complex patients. Finally, the VHA Medical Inpatient and Outpatient Datasets administrated by VA Informatics and Computing Infrastructure, consist of in-depth health services national data on inpatient and outpatient encounters and procedures.
The study cohort was divided into those with service-connected conditions (Class 1) or those with conditions not related to military service (Class 2). If veterans were identified in both classes, they were assigned to Class 1. The cost variable is determined by using the veterans’ classification. Class 1 veterans receive a lifetime limit of $6800, and Class 2 veterans receive a lifetime limit of $2000. A Class 2 veteran with ≥ 50% disability rating is eligible for a HISA lifetime limit of $6800. Whenever a value exceeds allowed limit of $6800 or $2000, due to data entry error or other reasons, the study team reassigned the cost value to the maximum allowed value.
Travel distance and time were derived by loading patient zip codes and HISA facility locations into the geographical information system program and using the nearest facility and find-route tools. These tools used a road network that simulates real-world driving conditions to calculate distance.
Study Variables
VWDs of any age, gender, and race/ethnicity who qualified for HISA and received HMs from fiscal year ( FY) 2015 through FY 2018 were identified (N = 30,823). Most VWDs were nonrural subjects (n = 19,970), and 43 had no Federal Information Processing System data. The final study cohort consisted of 10,810 HISA recipients. The NPPD, inpatient and outpatient data were merged by scrambled social security numbers to retrieve the following data: age, gender, race, ethnicity, marital status, Class (1 or 2), mean and total number of inpatient days, and type of HMs prescribed.
We also recorded rurality using the VA Rural-Urban Commuting Areas (RUCA) system, but we combined the rural and highly rural designation.1 Census tracts with a RUCA score of 10.0 are deemed highly rural, the remainder are considered rural except those with a RUCA score of 1.0 or 1.1. Travel time and distance from a veteran’s home to the VA facility that provided the HISA prescription were determined from zip codes. The current study focuses on VAMCs prescribing stations (affiliated sites of administrative parent medical facilities) where the HISA users obtained the HM, not the parent station (administrative parent medical facilities).
HISA Utilization
To characterize HISA utilization geographically and over time, the number of users were mapped by county. Areas where users were increasing (hot spots) or decreasing (cold spots) also were mapped. The maps were created using Environmental Systems Research Institute ArcGIS Pro 2.2.1 software. We chose to use natural breaks (Jenks) data classification method in a choropleth to symbolize the change over time map. We then used the Getis Ord GI* optimized hot spot analysis tool in the ArcGIS Pro spatial statistics tool set to generate the hot/cold spot maps. This tool identifies clusters of high values (hot spots) and low values (cold spots) creating a new output layer, RHUs by county, with a Z score, P value, and CI for each county. The Gi Bin field classifies statistically significant hot and cold spots. Counties sorted into the ± 3 category (bin) have a clustering characteristic (eg, with neighboring counties) that is statistically significant with a 99% CI; the ± 2 bin indicates a 95% CI for those county clustering sorted therein; ± 1 reflects a 90% CI; and 0 bin contains county features that have no statistical significant clustering with neighboring counties.
Data Analysis
Data were cleaned and analyzed using SAS 9.4 and R 3.5.3. Descriptive statistics are provided for sociodemographic characteristics, clinical characteristics, and class. ANOVA and t tests were used to compare continuous variables between groups, while χ2 and Fisher exact tests were used for dichotomous and categorical outcome variables. The threshold for statistical significance for these tests was set at α = .001.
Results
There were 10,810 RHUs from FY 2015 through FY 2018 and HISA utilization increased each year (Figure 1). Although some years may show usage decreases relative to previous fiscal years, the cumulative trends showed an increase relative to FY 2015 for both Classes of RVs (Figure 2). There was a 45.4% increase from FY 2015 to FY 2018 with a mean 13.6% yearly increase. Class 1 increased 21.0% and Class 2 increased 39.5% from FY 2015 to FY 2016 (Figure 3).
Most RHUs were male, White, and married. Class 1 and Class 2 RHUs differed significantly by age, race, marital status, and disability conditions: Class 1 RHUs were aged 6.6 years younger with a mean age of 69.1 years compared with 75.7 years for Class 2 users. For Class 1 RHUs, a plurality (29.4%) were aged 65 to 69 years; while a plurality (41.4%) of Class 2 users were aged ≥ 80 years. Musculoskeletal was the most common identified type of condition for all RHUs (Table 1).
To better understand HISA utilization patterns and net RHUs per county, we used a map to detail RHUs by county and change over time (Figure 4). Additionally, we compared US counties by RHUs from FY 2015 to FY 2018 and determined how clusters of high numbers of RHUs (hot spots) and low numbers of RHUs (cold spots) shifted over this period (Figure 5). While HISA utilization grew over the study period, the net count of RHUs per county varied by 9 to 20 persons/county. The population of RHUs increased over time in the Southwest, Southeast, and over much of the East/Northeast, while in the Central and Midwest regions, number of RHUs seems to decrease in population and/or use of the system. The cold spots in the Midwest and South Central US seem to increase with a significant relationship to neighboring counties having a low number of RHUs.
There were 11,166 HM prescribed to RHUs (Table 2). Bathroom HMs also were the dominant HM type for all facilities regardless of complexity levels (Table 3). The San Antonio, Texas, VAMC demonstrated the highest Class 1 vs Class 2 difference in HISA use (Class 1: 87.7% and Class 2: 12.3%). Except for the Des Moines VAMC, all other VAMCs showed HISA use > 60% by Class 1.
Cost Data
Air-conditioning installation ($5007) was the costliest HM overall (Table 4), closely followed by bathroom ($4978) and kitchen modifications ($4305). Bathroom renovations were the costliest HM type for both Class 1 and Class 2, closely followed by electrical repair and air-conditioning installation for Class 1 and driveway reconstruction and wooden ramp construction for Class 2.
The mean award received for HM was $4687 (Table 5). While the number of RHUs increased from FY 2015 to FY 2016, the average cost decreased, both overall ($280) and for Class 1 ($195) and Class 2 ($153). Except for a small decline in the number of Class 2 HISA recipients from FY 2017 to FY 2018, overall, the number of RHUs continuously grew from FY 2015 to FY 2018: 977 for the overall cohort, 678 for Class 1 and 299 for Class 2. Despite the obvious gain in the number of RHUs, the average costs did not notably change over time. VISN 21 had the highest mean cost, followed by VISNs 17, 6, 22, and 20.
Travel
Travel time and distance to the HISA prescribing facility differed significantly between Class 1 and Class 2 HISA users. RHUs had to travel about 95 minutes from their place of residence to access the HISA benefits program. There were no statistically significant differences between Class 1 and 2 users with respect to travel time and distance traveled (Table 6).
The majority of Class 1 and Class 2 veterans accessed the HISA from their nearest facility. However, nearly one-quarter of both Class 1 and 2 RHUs (24% each) did not. Among the 2598 who accessed the nonnearest facility, 97 (3.7%) accessed a facility that is ≤ 40 miles. Many (44%) users traveled 40 to 100 miles, and another 43.2% traveled 100 to 200 miles from their residence to access a HM prescription. Some 2598 users (1.1%) traveled > 500 miles to access a facility.
Discussion
Although utilization of the HISA program has steadily increased, overall participation by subpopulations such as RHUs can still be improved significantly. Veterans aged ≤ 46 years who have a disability that is common to those receiving HISA benefits have low HISA utilization. Similarly, veterans with sensory disabilities also have low use. These subpopulations are among those in great need of attention and services.
A study by Lucas and Zelaya, using the 2016 National Health Interview Survey data with an aim to measure degree of vision problems, dual sensory impairment, and hearing trouble in male veterans aged ≥ 18 years, found that veterans were more likely to report dual sensory impairment and balance difficulties when compared with nonveterans.9 The number of female veterans is growing but had very low representation in this study.10 This emerging VHA population requires information and education on their HM benefits.
Home Modifications
The most common HM prescribed for RHUs was for the bathroom. Further investigation is warranted as to why, given the diversity of HM types that the grant covers, low prescription rates exist across most of the HM types. There may be a lack of knowledge by providers and VWD as to the range of HMs that can be awarded under the grant. It is important that HCPs and veterans receive education on HISA HM options.
Semeah and colleagues pointed out the need for an assessment of the HISA HM ordering system to ensure that multiple HMs items (eg, kitchen, air conditioning, fees, driveway, and plumbing) are listed among the forced choices shown to clinicians to select from.7 Poor housing in rural America is widespread: 63% of rural dwellings need renovations and/or repairs to be accessible to individuals with disabilities, with > 6.7 million rural homes having no or faulty plumbing or kitchens; yet in this study, prescriptions for these HMs accounted for < 1%.11,12
VISN 6 had the most HISA awards with 1364, while VISN 21 had the fewest (245). Across all VISNs, Class 1 RHUs received more prescriptions than did Class 2 RHUs. Future research may seek to examine whether prescribers are fully aware of the eligibility of HM prescription to Class 2 veterans. VISN 21 ($5354); VISN 17 ($5302); and VISN 6 ($5301) had the highest mean HM expenditures. The national mean cost for HISA HMs were $4978 for bathrooms and $4305 for kitchens; for non-HISA HMs in FY 2017, the mean costs were $6362 and $12,255, respectively. A noteworthy concern is whether the maximum grant limit awards are sufficient to perform more expensive and complex HMs, such as the kitchen or major bathroom alternations.13
Facilities categorized as 1a, 1b, or 1c provided
North Florida/Sough Georgia was the highest-prescribing VAMC with 39% more HM prescriptions than the second highest prescribing facility (Durham, NC). Unfortunately, the data presented here cannot establish causality for the large variance difference between the top facilities, and the skewed distribution of total RHUs across VAMCs.
Travel-Related Variables
HISA beneficiaries face significant travel-related challenges. Just 3.6% of RHUs could access a facility within 40 miles of their home and 43.2% traveled 100 to 200 miles from their home to access a HM prescription. Further exploration is warranted to understand how travel patterns impact access to or the uptake of HISA.
RVs already have problems with accessing care because of long travel time.14,15 The choice or necessity to travel to a farther facility for HISA prescription is problematic for RVs, especially when transportation is often reported in the literature as a barrier to resources for people living in rural communities.15-17 When patients have travel barriers, they wait longer to obtain medical services and often wait for their conditions to worsen before seeking services.15,18 Once HM is completed, telerehabilitation is an effective delivery method used for delivering health care services to people in remote places.18,19 Considering that HISA use has the potential to improve quality of life, afford comfort, facilitate the accomplishment of activities of daily living for RVs, it is important that future studies examine how existing telehealth technologies can be used to improve HISA access.
Future Directions
County-level analyses is warranted in future studies exploring potential variables associated with HISA use; for example, county-level rates of primary care physicians and other HCPs. Future research should explore how long distance travel impacts the HISA application process and HM implementation. Further research also should focus on the HISA application structure and process to identify causes of delays. The HISA application process takes a mean 6 months to complete, yet the duration of hospital stays is 1 to 3 weeks, thus it is impossible to connect HISA to hospital discharge, which was the original intent of the program. Future research can examine how telehealth services can expedite HISA obtainment and coordination of the application process. Future research also may study the possible causes of the wide variations in HM prescriptions per facility. It is also important that educational programs provide information on the array of HM items that veterans can obtain.
Conclusions
In our previous study of the HISA cohort (2011-2017), we documented that an increase in utilization of the HISA program was warranted based on the low national budgetary appropriation and identification of significant low participation by vulnerable subpopulations, including veterans residing in rural areas or having returned from recent conflicts.7 The present study documents national utilization patterns, demographic profiles, and clinical characteristics of RHUs from FY 2015 through FY 2018, data that may be useful to policy makers and HISA administrators in predicting future use and users. It is important to note that the data and information presented in this article identify trends. The work in no way establishes a gold standard or any targeted goal of utilization. Future research could focus on conceptualizing or theorizing what steps are necessary to set such a gold standard of utilization rate and steps toward achievement.
Acknowledgments
This research was supported by grant 15521 from the US Department of Veterans Affairs, Office of Rural Health . Furthermore, the research was supported in part by grant K12 HD055929 from the National Institutes of Health.
1. US Department of Veterans Affairs, Veteran Health Administration, Office of Rural Health. Rural veteran health care challenges. Updated February 9, 2021. Accessed June 11, 2021. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp
2. Holder, K.A. Veterans in rural America, 2011–2015. Published January 2017. Accessed June 11, 2021. https://www.census.gov/content/dam/Census/library/publications/2017/acs/acs-36.pdf
3. Pezzin LE, Bogner HR, Kurichi JE, et al. Preventable hospitalizations, barriers to care, and disability. Medicine (Baltimore). 2018;97(19):e0691. doi:10.1097/MD.0000000000010691
4. Rosenbach ML. Access and satisfaction within the disabled Medicare population. Health Care Financ Rev. 1995;17(2):147-167.
5. Semeah LM, Ganesh SP, Wang X, et al. Home modification and health services utilization in rural and urban veterans with disabilities. Housing Policy Debate. 2021. Published online: March 4, 2021. doi:10.1080/10511482.2020.1858923
6. Spoont M, Greer N, Su J, Fitzgerald P, Rutks I, and Wilt TJ. Rural vs. urban ambulatory health care: A Systematic Review. Published May 2011. Accessed June 11, 2021. https://www.hsrd.research.va.gov/publications/esp/ambulatory.pdf
7. Semeah LM, Wang X, Cowper Ripley DC, et al. Improving health through a home modification service for veterans. In: Fiedler BA, ed. Three Facets of Public Health and Paths to Improvements. Academic Press; 2020:381-416.
8. Semeah LM, Ahrentzen S, Jia H, Cowper-Ripley DC, Levy CE, Mann WC. The home improvements and structural alterations benefits program: veterans with disabilities and home accessibility. J Disability Policy Studies. 2017;28(1):43-51. doi:10.1177/1044207317696275
9. Lucas, JW, Zelaya, CE. Hearing difficulty, vision trouble, and balance problems among male veterans and nonveterans. Published June 12, 2020. Accessed June 11, 2021. https://www.cdc.gov/nchs/data/nhsr/nhsr142-508.pdf
10. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Women veterans report: the past, present, and future of women veterans. Published February 2017. Accessed June 11, 2021. https://www.va.gov/vetdata/docs/SpecialReports/Women_Veterans_2015_Final.pdf
11. US Department of Housing and Urban Development, Office of Policy Development and Research. Housing challenges of rural seniors. Published 2017. Accessed June 11, 2021. https://www.huduser.gov/portal/periodicals/em/summer17/highlight1.html
12. Pendall R, Goodman L, Zhu J, Gold A. The future of rural housing. Published October 2016. Accessed June 11, 202.1 https://www.urban.org/sites/default/files/publication/85101/2000972-the-future-of-rural-housing_6.pdf
13. Joint Center for Housing Studies at Harvard University. Improving America’s housing 2019. Published 2019. Accessed June 11, 2021. https://www.jchs.harvard.edu/sites/default/files/reports/files/Harvard_JCHS_Improving_Americas_Housing_2019.pdf
14. Schooley BL, Horan TA, Lee PW, West PA. Rural veteran access to healthcare services: investigating the role of information and communication technologies in overcoming spatial barriers. Perspect Health Inf Manag. 2010;7(Spring):1f. Published 2010 Apr 1.
15. Ripley DC, Kwong PL, Vogel WB, Kurichi JE, Bates BE, Davenport C. How does geographic access affect in-hospital mortality for veterans with acute ischemic stroke?. Med Care. 2015;53(6):501-509. doi:10.1097/MLR.0000000000000366
16. Cowper-Ripley DC, Reker DM, Hayes J, et al. Geographic access to VHA rehabilitation services for traumatically injured veterans. Fed Pract. 2009;26(10):28-39.
17. Smith M, Towne S, Herrera-Venson A, Cameron K, Horel S, Ory M, et al. Delivery of fall prevention interventions for at-risk older adults in rural areas: Findings from a national dissemination. International journal of environmental research and public health. 2018;15:2798. doi: 10.3390/ijerph15122798
18. Hale-Gallardo JL, Kreider CM, Jia H, et al. Telerehabilitation for Rural Veterans: A Qualitative Assessment of Barriers and Facilitators to Implementation. J Multidiscip Healthc. 2020;13:559-570. doi:10.2147/JMDH.S247267
19. Sarfo FS, Akassi J, Kyem G, et al. Long-Term Outcomes of Stroke in a Ghanaian Outpatient Clinic. J Stroke Cerebrovasc Dis. 2018;27(4):1090-1099. doi:10.1016/j.jstrokecerebrovasdis.2017.11.017
The US Department of Veterans Affairs (VA) created the Home Improvements and Structural Alterations (HISA) program to help provide necessary home modifications (HMs) to veterans with disabilities (VWDs) that will facilitate the provision of medical services at home and improve home accessibility and functional independence. The Veterans Health Administration (VHA) has more than 9 million veteran enrollees; of those, 2.7 million are classified as rural or highly rural.1 Rural veterans (RVs) possess higher rate of disability compared with that of urban veterans.2-5 RVs have unequal access to screening of ambulatory care sensitive conditions (eg, hypertension, diabetes mellitus).6 Furthermore, RVs are at risk of poor medical outcomes due to distance from health care facilities and specialist care, which can be a barrier to emergency care when issues arise. These barriers, among others, are associated with compromised health quality of life and health outcomes for RVs.3,6 The HISA program may be key to decreasing falls and other serious mishaps in the home. Therefore, understanding use of the HISA program by RVs is important. However, to date little information has been available regarding use of HISA benefits by RVs or characteristics of RVs who receive HISA benefits.
HISA Alterations Program
HISA was initially developed by VA to improve veterans’ transition from acute medical care to home.7,8 However, to obtain HISA grants currently, there is an average 3 to 6 months application process.7 Through the HISA program, VWDs can be prescribed the following HMs, including (but not limited to): flooring replacement, permanent ramps, roll-in showers, installation of central air-conditioning systems, improved lighting, kitchen/bathroom modifications, and home inspections. The HMs prescribed depend on an assessment of medical need by health care providers (HCPs).8
As time passed and the veteran population aged, the program now primarily helps ensure the ability to enter into essential areas and safety in the home.5 The amount of a HISA payment is based on whether a veteran’s health condition is related to military service as defined by the VHA service connection medical evaluation process. Barriers to obtaining a HISA HM can include difficulty in navigating the evaluation process and difficulty in finding a qualified contractor or builder to do the HM.7
This article aims to: (1) Detail the sociodemographic and clinical characteristics of rural HISA users (RHUs); (2) report on HISA usage patterns in number, types, and cost of HMs; (3) compare use amid the diverse VA medical centers (VAMCs) and related complexity levels and Veterans Integrated Service Networks (VISNs); and (4) examine the relationship between travel time/distance and HISA utilization. The long-term goal is to provide accurate information to researchers, HM administrators, health care providers and policy makers on HISA program utilization by rural VWDs, which may help improve its use and bring awareness of its users. This study was approved by the affiliate University of Florida Institutional Review Board and VA research and development committee at the North Florida/South Georgia Veterans Health System.
Methods
Data were obtained from 3 VA sources: the National Prosthetics Patient Database (NPPD), the VHA Medical Inpatient Dataset, and the VHA Outpatient Dataset.7 The NPPD is a national administrative database that contains information on prosthetic-associated products ordered by HCPs for patients, such as portable ramps, handrails, home oxygen equipment, and orthotic and prosthetic apparatus. Data obtained from the NPPD included cost of HMs, clinical characteristics, VISN, and VAMC. VA facilities are categorized into complexity levels 1a, 1b, 1c, 2, and 3. Complexity level 1a to 1c VAMCs address medical cases that entail “heightening involvedness,” meaning a larger number of patients presented with medical concerns needing medical specialists. Complexity levels 2 and 3 have fewer resources, lower patient numbers, and less medically complex patients. Finally, the VHA Medical Inpatient and Outpatient Datasets administrated by VA Informatics and Computing Infrastructure, consist of in-depth health services national data on inpatient and outpatient encounters and procedures.
The study cohort was divided into those with service-connected conditions (Class 1) or those with conditions not related to military service (Class 2). If veterans were identified in both classes, they were assigned to Class 1. The cost variable is determined by using the veterans’ classification. Class 1 veterans receive a lifetime limit of $6800, and Class 2 veterans receive a lifetime limit of $2000. A Class 2 veteran with ≥ 50% disability rating is eligible for a HISA lifetime limit of $6800. Whenever a value exceeds allowed limit of $6800 or $2000, due to data entry error or other reasons, the study team reassigned the cost value to the maximum allowed value.
Travel distance and time were derived by loading patient zip codes and HISA facility locations into the geographical information system program and using the nearest facility and find-route tools. These tools used a road network that simulates real-world driving conditions to calculate distance.
Study Variables
VWDs of any age, gender, and race/ethnicity who qualified for HISA and received HMs from fiscal year ( FY) 2015 through FY 2018 were identified (N = 30,823). Most VWDs were nonrural subjects (n = 19,970), and 43 had no Federal Information Processing System data. The final study cohort consisted of 10,810 HISA recipients. The NPPD, inpatient and outpatient data were merged by scrambled social security numbers to retrieve the following data: age, gender, race, ethnicity, marital status, Class (1 or 2), mean and total number of inpatient days, and type of HMs prescribed.
We also recorded rurality using the VA Rural-Urban Commuting Areas (RUCA) system, but we combined the rural and highly rural designation.1 Census tracts with a RUCA score of 10.0 are deemed highly rural, the remainder are considered rural except those with a RUCA score of 1.0 or 1.1. Travel time and distance from a veteran’s home to the VA facility that provided the HISA prescription were determined from zip codes. The current study focuses on VAMCs prescribing stations (affiliated sites of administrative parent medical facilities) where the HISA users obtained the HM, not the parent station (administrative parent medical facilities).
HISA Utilization
To characterize HISA utilization geographically and over time, the number of users were mapped by county. Areas where users were increasing (hot spots) or decreasing (cold spots) also were mapped. The maps were created using Environmental Systems Research Institute ArcGIS Pro 2.2.1 software. We chose to use natural breaks (Jenks) data classification method in a choropleth to symbolize the change over time map. We then used the Getis Ord GI* optimized hot spot analysis tool in the ArcGIS Pro spatial statistics tool set to generate the hot/cold spot maps. This tool identifies clusters of high values (hot spots) and low values (cold spots) creating a new output layer, RHUs by county, with a Z score, P value, and CI for each county. The Gi Bin field classifies statistically significant hot and cold spots. Counties sorted into the ± 3 category (bin) have a clustering characteristic (eg, with neighboring counties) that is statistically significant with a 99% CI; the ± 2 bin indicates a 95% CI for those county clustering sorted therein; ± 1 reflects a 90% CI; and 0 bin contains county features that have no statistical significant clustering with neighboring counties.
Data Analysis
Data were cleaned and analyzed using SAS 9.4 and R 3.5.3. Descriptive statistics are provided for sociodemographic characteristics, clinical characteristics, and class. ANOVA and t tests were used to compare continuous variables between groups, while χ2 and Fisher exact tests were used for dichotomous and categorical outcome variables. The threshold for statistical significance for these tests was set at α = .001.
Results
There were 10,810 RHUs from FY 2015 through FY 2018 and HISA utilization increased each year (Figure 1). Although some years may show usage decreases relative to previous fiscal years, the cumulative trends showed an increase relative to FY 2015 for both Classes of RVs (Figure 2). There was a 45.4% increase from FY 2015 to FY 2018 with a mean 13.6% yearly increase. Class 1 increased 21.0% and Class 2 increased 39.5% from FY 2015 to FY 2016 (Figure 3).
Most RHUs were male, White, and married. Class 1 and Class 2 RHUs differed significantly by age, race, marital status, and disability conditions: Class 1 RHUs were aged 6.6 years younger with a mean age of 69.1 years compared with 75.7 years for Class 2 users. For Class 1 RHUs, a plurality (29.4%) were aged 65 to 69 years; while a plurality (41.4%) of Class 2 users were aged ≥ 80 years. Musculoskeletal was the most common identified type of condition for all RHUs (Table 1).
To better understand HISA utilization patterns and net RHUs per county, we used a map to detail RHUs by county and change over time (Figure 4). Additionally, we compared US counties by RHUs from FY 2015 to FY 2018 and determined how clusters of high numbers of RHUs (hot spots) and low numbers of RHUs (cold spots) shifted over this period (Figure 5). While HISA utilization grew over the study period, the net count of RHUs per county varied by 9 to 20 persons/county. The population of RHUs increased over time in the Southwest, Southeast, and over much of the East/Northeast, while in the Central and Midwest regions, number of RHUs seems to decrease in population and/or use of the system. The cold spots in the Midwest and South Central US seem to increase with a significant relationship to neighboring counties having a low number of RHUs.
There were 11,166 HM prescribed to RHUs (Table 2). Bathroom HMs also were the dominant HM type for all facilities regardless of complexity levels (Table 3). The San Antonio, Texas, VAMC demonstrated the highest Class 1 vs Class 2 difference in HISA use (Class 1: 87.7% and Class 2: 12.3%). Except for the Des Moines VAMC, all other VAMCs showed HISA use > 60% by Class 1.
Cost Data
Air-conditioning installation ($5007) was the costliest HM overall (Table 4), closely followed by bathroom ($4978) and kitchen modifications ($4305). Bathroom renovations were the costliest HM type for both Class 1 and Class 2, closely followed by electrical repair and air-conditioning installation for Class 1 and driveway reconstruction and wooden ramp construction for Class 2.
The mean award received for HM was $4687 (Table 5). While the number of RHUs increased from FY 2015 to FY 2016, the average cost decreased, both overall ($280) and for Class 1 ($195) and Class 2 ($153). Except for a small decline in the number of Class 2 HISA recipients from FY 2017 to FY 2018, overall, the number of RHUs continuously grew from FY 2015 to FY 2018: 977 for the overall cohort, 678 for Class 1 and 299 for Class 2. Despite the obvious gain in the number of RHUs, the average costs did not notably change over time. VISN 21 had the highest mean cost, followed by VISNs 17, 6, 22, and 20.
Travel
Travel time and distance to the HISA prescribing facility differed significantly between Class 1 and Class 2 HISA users. RHUs had to travel about 95 minutes from their place of residence to access the HISA benefits program. There were no statistically significant differences between Class 1 and 2 users with respect to travel time and distance traveled (Table 6).
The majority of Class 1 and Class 2 veterans accessed the HISA from their nearest facility. However, nearly one-quarter of both Class 1 and 2 RHUs (24% each) did not. Among the 2598 who accessed the nonnearest facility, 97 (3.7%) accessed a facility that is ≤ 40 miles. Many (44%) users traveled 40 to 100 miles, and another 43.2% traveled 100 to 200 miles from their residence to access a HM prescription. Some 2598 users (1.1%) traveled > 500 miles to access a facility.
Discussion
Although utilization of the HISA program has steadily increased, overall participation by subpopulations such as RHUs can still be improved significantly. Veterans aged ≤ 46 years who have a disability that is common to those receiving HISA benefits have low HISA utilization. Similarly, veterans with sensory disabilities also have low use. These subpopulations are among those in great need of attention and services.
A study by Lucas and Zelaya, using the 2016 National Health Interview Survey data with an aim to measure degree of vision problems, dual sensory impairment, and hearing trouble in male veterans aged ≥ 18 years, found that veterans were more likely to report dual sensory impairment and balance difficulties when compared with nonveterans.9 The number of female veterans is growing but had very low representation in this study.10 This emerging VHA population requires information and education on their HM benefits.
Home Modifications
The most common HM prescribed for RHUs was for the bathroom. Further investigation is warranted as to why, given the diversity of HM types that the grant covers, low prescription rates exist across most of the HM types. There may be a lack of knowledge by providers and VWD as to the range of HMs that can be awarded under the grant. It is important that HCPs and veterans receive education on HISA HM options.
Semeah and colleagues pointed out the need for an assessment of the HISA HM ordering system to ensure that multiple HMs items (eg, kitchen, air conditioning, fees, driveway, and plumbing) are listed among the forced choices shown to clinicians to select from.7 Poor housing in rural America is widespread: 63% of rural dwellings need renovations and/or repairs to be accessible to individuals with disabilities, with > 6.7 million rural homes having no or faulty plumbing or kitchens; yet in this study, prescriptions for these HMs accounted for < 1%.11,12
VISN 6 had the most HISA awards with 1364, while VISN 21 had the fewest (245). Across all VISNs, Class 1 RHUs received more prescriptions than did Class 2 RHUs. Future research may seek to examine whether prescribers are fully aware of the eligibility of HM prescription to Class 2 veterans. VISN 21 ($5354); VISN 17 ($5302); and VISN 6 ($5301) had the highest mean HM expenditures. The national mean cost for HISA HMs were $4978 for bathrooms and $4305 for kitchens; for non-HISA HMs in FY 2017, the mean costs were $6362 and $12,255, respectively. A noteworthy concern is whether the maximum grant limit awards are sufficient to perform more expensive and complex HMs, such as the kitchen or major bathroom alternations.13
Facilities categorized as 1a, 1b, or 1c provided
North Florida/Sough Georgia was the highest-prescribing VAMC with 39% more HM prescriptions than the second highest prescribing facility (Durham, NC). Unfortunately, the data presented here cannot establish causality for the large variance difference between the top facilities, and the skewed distribution of total RHUs across VAMCs.
Travel-Related Variables
HISA beneficiaries face significant travel-related challenges. Just 3.6% of RHUs could access a facility within 40 miles of their home and 43.2% traveled 100 to 200 miles from their home to access a HM prescription. Further exploration is warranted to understand how travel patterns impact access to or the uptake of HISA.
RVs already have problems with accessing care because of long travel time.14,15 The choice or necessity to travel to a farther facility for HISA prescription is problematic for RVs, especially when transportation is often reported in the literature as a barrier to resources for people living in rural communities.15-17 When patients have travel barriers, they wait longer to obtain medical services and often wait for their conditions to worsen before seeking services.15,18 Once HM is completed, telerehabilitation is an effective delivery method used for delivering health care services to people in remote places.18,19 Considering that HISA use has the potential to improve quality of life, afford comfort, facilitate the accomplishment of activities of daily living for RVs, it is important that future studies examine how existing telehealth technologies can be used to improve HISA access.
Future Directions
County-level analyses is warranted in future studies exploring potential variables associated with HISA use; for example, county-level rates of primary care physicians and other HCPs. Future research should explore how long distance travel impacts the HISA application process and HM implementation. Further research also should focus on the HISA application structure and process to identify causes of delays. The HISA application process takes a mean 6 months to complete, yet the duration of hospital stays is 1 to 3 weeks, thus it is impossible to connect HISA to hospital discharge, which was the original intent of the program. Future research can examine how telehealth services can expedite HISA obtainment and coordination of the application process. Future research also may study the possible causes of the wide variations in HM prescriptions per facility. It is also important that educational programs provide information on the array of HM items that veterans can obtain.
Conclusions
In our previous study of the HISA cohort (2011-2017), we documented that an increase in utilization of the HISA program was warranted based on the low national budgetary appropriation and identification of significant low participation by vulnerable subpopulations, including veterans residing in rural areas or having returned from recent conflicts.7 The present study documents national utilization patterns, demographic profiles, and clinical characteristics of RHUs from FY 2015 through FY 2018, data that may be useful to policy makers and HISA administrators in predicting future use and users. It is important to note that the data and information presented in this article identify trends. The work in no way establishes a gold standard or any targeted goal of utilization. Future research could focus on conceptualizing or theorizing what steps are necessary to set such a gold standard of utilization rate and steps toward achievement.
Acknowledgments
This research was supported by grant 15521 from the US Department of Veterans Affairs, Office of Rural Health . Furthermore, the research was supported in part by grant K12 HD055929 from the National Institutes of Health.
The US Department of Veterans Affairs (VA) created the Home Improvements and Structural Alterations (HISA) program to help provide necessary home modifications (HMs) to veterans with disabilities (VWDs) that will facilitate the provision of medical services at home and improve home accessibility and functional independence. The Veterans Health Administration (VHA) has more than 9 million veteran enrollees; of those, 2.7 million are classified as rural or highly rural.1 Rural veterans (RVs) possess higher rate of disability compared with that of urban veterans.2-5 RVs have unequal access to screening of ambulatory care sensitive conditions (eg, hypertension, diabetes mellitus).6 Furthermore, RVs are at risk of poor medical outcomes due to distance from health care facilities and specialist care, which can be a barrier to emergency care when issues arise. These barriers, among others, are associated with compromised health quality of life and health outcomes for RVs.3,6 The HISA program may be key to decreasing falls and other serious mishaps in the home. Therefore, understanding use of the HISA program by RVs is important. However, to date little information has been available regarding use of HISA benefits by RVs or characteristics of RVs who receive HISA benefits.
HISA Alterations Program
HISA was initially developed by VA to improve veterans’ transition from acute medical care to home.7,8 However, to obtain HISA grants currently, there is an average 3 to 6 months application process.7 Through the HISA program, VWDs can be prescribed the following HMs, including (but not limited to): flooring replacement, permanent ramps, roll-in showers, installation of central air-conditioning systems, improved lighting, kitchen/bathroom modifications, and home inspections. The HMs prescribed depend on an assessment of medical need by health care providers (HCPs).8
As time passed and the veteran population aged, the program now primarily helps ensure the ability to enter into essential areas and safety in the home.5 The amount of a HISA payment is based on whether a veteran’s health condition is related to military service as defined by the VHA service connection medical evaluation process. Barriers to obtaining a HISA HM can include difficulty in navigating the evaluation process and difficulty in finding a qualified contractor or builder to do the HM.7
This article aims to: (1) Detail the sociodemographic and clinical characteristics of rural HISA users (RHUs); (2) report on HISA usage patterns in number, types, and cost of HMs; (3) compare use amid the diverse VA medical centers (VAMCs) and related complexity levels and Veterans Integrated Service Networks (VISNs); and (4) examine the relationship between travel time/distance and HISA utilization. The long-term goal is to provide accurate information to researchers, HM administrators, health care providers and policy makers on HISA program utilization by rural VWDs, which may help improve its use and bring awareness of its users. This study was approved by the affiliate University of Florida Institutional Review Board and VA research and development committee at the North Florida/South Georgia Veterans Health System.
Methods
Data were obtained from 3 VA sources: the National Prosthetics Patient Database (NPPD), the VHA Medical Inpatient Dataset, and the VHA Outpatient Dataset.7 The NPPD is a national administrative database that contains information on prosthetic-associated products ordered by HCPs for patients, such as portable ramps, handrails, home oxygen equipment, and orthotic and prosthetic apparatus. Data obtained from the NPPD included cost of HMs, clinical characteristics, VISN, and VAMC. VA facilities are categorized into complexity levels 1a, 1b, 1c, 2, and 3. Complexity level 1a to 1c VAMCs address medical cases that entail “heightening involvedness,” meaning a larger number of patients presented with medical concerns needing medical specialists. Complexity levels 2 and 3 have fewer resources, lower patient numbers, and less medically complex patients. Finally, the VHA Medical Inpatient and Outpatient Datasets administrated by VA Informatics and Computing Infrastructure, consist of in-depth health services national data on inpatient and outpatient encounters and procedures.
The study cohort was divided into those with service-connected conditions (Class 1) or those with conditions not related to military service (Class 2). If veterans were identified in both classes, they were assigned to Class 1. The cost variable is determined by using the veterans’ classification. Class 1 veterans receive a lifetime limit of $6800, and Class 2 veterans receive a lifetime limit of $2000. A Class 2 veteran with ≥ 50% disability rating is eligible for a HISA lifetime limit of $6800. Whenever a value exceeds allowed limit of $6800 or $2000, due to data entry error or other reasons, the study team reassigned the cost value to the maximum allowed value.
Travel distance and time were derived by loading patient zip codes and HISA facility locations into the geographical information system program and using the nearest facility and find-route tools. These tools used a road network that simulates real-world driving conditions to calculate distance.
Study Variables
VWDs of any age, gender, and race/ethnicity who qualified for HISA and received HMs from fiscal year ( FY) 2015 through FY 2018 were identified (N = 30,823). Most VWDs were nonrural subjects (n = 19,970), and 43 had no Federal Information Processing System data. The final study cohort consisted of 10,810 HISA recipients. The NPPD, inpatient and outpatient data were merged by scrambled social security numbers to retrieve the following data: age, gender, race, ethnicity, marital status, Class (1 or 2), mean and total number of inpatient days, and type of HMs prescribed.
We also recorded rurality using the VA Rural-Urban Commuting Areas (RUCA) system, but we combined the rural and highly rural designation.1 Census tracts with a RUCA score of 10.0 are deemed highly rural, the remainder are considered rural except those with a RUCA score of 1.0 or 1.1. Travel time and distance from a veteran’s home to the VA facility that provided the HISA prescription were determined from zip codes. The current study focuses on VAMCs prescribing stations (affiliated sites of administrative parent medical facilities) where the HISA users obtained the HM, not the parent station (administrative parent medical facilities).
HISA Utilization
To characterize HISA utilization geographically and over time, the number of users were mapped by county. Areas where users were increasing (hot spots) or decreasing (cold spots) also were mapped. The maps were created using Environmental Systems Research Institute ArcGIS Pro 2.2.1 software. We chose to use natural breaks (Jenks) data classification method in a choropleth to symbolize the change over time map. We then used the Getis Ord GI* optimized hot spot analysis tool in the ArcGIS Pro spatial statistics tool set to generate the hot/cold spot maps. This tool identifies clusters of high values (hot spots) and low values (cold spots) creating a new output layer, RHUs by county, with a Z score, P value, and CI for each county. The Gi Bin field classifies statistically significant hot and cold spots. Counties sorted into the ± 3 category (bin) have a clustering characteristic (eg, with neighboring counties) that is statistically significant with a 99% CI; the ± 2 bin indicates a 95% CI for those county clustering sorted therein; ± 1 reflects a 90% CI; and 0 bin contains county features that have no statistical significant clustering with neighboring counties.
Data Analysis
Data were cleaned and analyzed using SAS 9.4 and R 3.5.3. Descriptive statistics are provided for sociodemographic characteristics, clinical characteristics, and class. ANOVA and t tests were used to compare continuous variables between groups, while χ2 and Fisher exact tests were used for dichotomous and categorical outcome variables. The threshold for statistical significance for these tests was set at α = .001.
Results
There were 10,810 RHUs from FY 2015 through FY 2018 and HISA utilization increased each year (Figure 1). Although some years may show usage decreases relative to previous fiscal years, the cumulative trends showed an increase relative to FY 2015 for both Classes of RVs (Figure 2). There was a 45.4% increase from FY 2015 to FY 2018 with a mean 13.6% yearly increase. Class 1 increased 21.0% and Class 2 increased 39.5% from FY 2015 to FY 2016 (Figure 3).
Most RHUs were male, White, and married. Class 1 and Class 2 RHUs differed significantly by age, race, marital status, and disability conditions: Class 1 RHUs were aged 6.6 years younger with a mean age of 69.1 years compared with 75.7 years for Class 2 users. For Class 1 RHUs, a plurality (29.4%) were aged 65 to 69 years; while a plurality (41.4%) of Class 2 users were aged ≥ 80 years. Musculoskeletal was the most common identified type of condition for all RHUs (Table 1).
To better understand HISA utilization patterns and net RHUs per county, we used a map to detail RHUs by county and change over time (Figure 4). Additionally, we compared US counties by RHUs from FY 2015 to FY 2018 and determined how clusters of high numbers of RHUs (hot spots) and low numbers of RHUs (cold spots) shifted over this period (Figure 5). While HISA utilization grew over the study period, the net count of RHUs per county varied by 9 to 20 persons/county. The population of RHUs increased over time in the Southwest, Southeast, and over much of the East/Northeast, while in the Central and Midwest regions, number of RHUs seems to decrease in population and/or use of the system. The cold spots in the Midwest and South Central US seem to increase with a significant relationship to neighboring counties having a low number of RHUs.
There were 11,166 HM prescribed to RHUs (Table 2). Bathroom HMs also were the dominant HM type for all facilities regardless of complexity levels (Table 3). The San Antonio, Texas, VAMC demonstrated the highest Class 1 vs Class 2 difference in HISA use (Class 1: 87.7% and Class 2: 12.3%). Except for the Des Moines VAMC, all other VAMCs showed HISA use > 60% by Class 1.
Cost Data
Air-conditioning installation ($5007) was the costliest HM overall (Table 4), closely followed by bathroom ($4978) and kitchen modifications ($4305). Bathroom renovations were the costliest HM type for both Class 1 and Class 2, closely followed by electrical repair and air-conditioning installation for Class 1 and driveway reconstruction and wooden ramp construction for Class 2.
The mean award received for HM was $4687 (Table 5). While the number of RHUs increased from FY 2015 to FY 2016, the average cost decreased, both overall ($280) and for Class 1 ($195) and Class 2 ($153). Except for a small decline in the number of Class 2 HISA recipients from FY 2017 to FY 2018, overall, the number of RHUs continuously grew from FY 2015 to FY 2018: 977 for the overall cohort, 678 for Class 1 and 299 for Class 2. Despite the obvious gain in the number of RHUs, the average costs did not notably change over time. VISN 21 had the highest mean cost, followed by VISNs 17, 6, 22, and 20.
Travel
Travel time and distance to the HISA prescribing facility differed significantly between Class 1 and Class 2 HISA users. RHUs had to travel about 95 minutes from their place of residence to access the HISA benefits program. There were no statistically significant differences between Class 1 and 2 users with respect to travel time and distance traveled (Table 6).
The majority of Class 1 and Class 2 veterans accessed the HISA from their nearest facility. However, nearly one-quarter of both Class 1 and 2 RHUs (24% each) did not. Among the 2598 who accessed the nonnearest facility, 97 (3.7%) accessed a facility that is ≤ 40 miles. Many (44%) users traveled 40 to 100 miles, and another 43.2% traveled 100 to 200 miles from their residence to access a HM prescription. Some 2598 users (1.1%) traveled > 500 miles to access a facility.
Discussion
Although utilization of the HISA program has steadily increased, overall participation by subpopulations such as RHUs can still be improved significantly. Veterans aged ≤ 46 years who have a disability that is common to those receiving HISA benefits have low HISA utilization. Similarly, veterans with sensory disabilities also have low use. These subpopulations are among those in great need of attention and services.
A study by Lucas and Zelaya, using the 2016 National Health Interview Survey data with an aim to measure degree of vision problems, dual sensory impairment, and hearing trouble in male veterans aged ≥ 18 years, found that veterans were more likely to report dual sensory impairment and balance difficulties when compared with nonveterans.9 The number of female veterans is growing but had very low representation in this study.10 This emerging VHA population requires information and education on their HM benefits.
Home Modifications
The most common HM prescribed for RHUs was for the bathroom. Further investigation is warranted as to why, given the diversity of HM types that the grant covers, low prescription rates exist across most of the HM types. There may be a lack of knowledge by providers and VWD as to the range of HMs that can be awarded under the grant. It is important that HCPs and veterans receive education on HISA HM options.
Semeah and colleagues pointed out the need for an assessment of the HISA HM ordering system to ensure that multiple HMs items (eg, kitchen, air conditioning, fees, driveway, and plumbing) are listed among the forced choices shown to clinicians to select from.7 Poor housing in rural America is widespread: 63% of rural dwellings need renovations and/or repairs to be accessible to individuals with disabilities, with > 6.7 million rural homes having no or faulty plumbing or kitchens; yet in this study, prescriptions for these HMs accounted for < 1%.11,12
VISN 6 had the most HISA awards with 1364, while VISN 21 had the fewest (245). Across all VISNs, Class 1 RHUs received more prescriptions than did Class 2 RHUs. Future research may seek to examine whether prescribers are fully aware of the eligibility of HM prescription to Class 2 veterans. VISN 21 ($5354); VISN 17 ($5302); and VISN 6 ($5301) had the highest mean HM expenditures. The national mean cost for HISA HMs were $4978 for bathrooms and $4305 for kitchens; for non-HISA HMs in FY 2017, the mean costs were $6362 and $12,255, respectively. A noteworthy concern is whether the maximum grant limit awards are sufficient to perform more expensive and complex HMs, such as the kitchen or major bathroom alternations.13
Facilities categorized as 1a, 1b, or 1c provided
North Florida/Sough Georgia was the highest-prescribing VAMC with 39% more HM prescriptions than the second highest prescribing facility (Durham, NC). Unfortunately, the data presented here cannot establish causality for the large variance difference between the top facilities, and the skewed distribution of total RHUs across VAMCs.
Travel-Related Variables
HISA beneficiaries face significant travel-related challenges. Just 3.6% of RHUs could access a facility within 40 miles of their home and 43.2% traveled 100 to 200 miles from their home to access a HM prescription. Further exploration is warranted to understand how travel patterns impact access to or the uptake of HISA.
RVs already have problems with accessing care because of long travel time.14,15 The choice or necessity to travel to a farther facility for HISA prescription is problematic for RVs, especially when transportation is often reported in the literature as a barrier to resources for people living in rural communities.15-17 When patients have travel barriers, they wait longer to obtain medical services and often wait for their conditions to worsen before seeking services.15,18 Once HM is completed, telerehabilitation is an effective delivery method used for delivering health care services to people in remote places.18,19 Considering that HISA use has the potential to improve quality of life, afford comfort, facilitate the accomplishment of activities of daily living for RVs, it is important that future studies examine how existing telehealth technologies can be used to improve HISA access.
Future Directions
County-level analyses is warranted in future studies exploring potential variables associated with HISA use; for example, county-level rates of primary care physicians and other HCPs. Future research should explore how long distance travel impacts the HISA application process and HM implementation. Further research also should focus on the HISA application structure and process to identify causes of delays. The HISA application process takes a mean 6 months to complete, yet the duration of hospital stays is 1 to 3 weeks, thus it is impossible to connect HISA to hospital discharge, which was the original intent of the program. Future research can examine how telehealth services can expedite HISA obtainment and coordination of the application process. Future research also may study the possible causes of the wide variations in HM prescriptions per facility. It is also important that educational programs provide information on the array of HM items that veterans can obtain.
Conclusions
In our previous study of the HISA cohort (2011-2017), we documented that an increase in utilization of the HISA program was warranted based on the low national budgetary appropriation and identification of significant low participation by vulnerable subpopulations, including veterans residing in rural areas or having returned from recent conflicts.7 The present study documents national utilization patterns, demographic profiles, and clinical characteristics of RHUs from FY 2015 through FY 2018, data that may be useful to policy makers and HISA administrators in predicting future use and users. It is important to note that the data and information presented in this article identify trends. The work in no way establishes a gold standard or any targeted goal of utilization. Future research could focus on conceptualizing or theorizing what steps are necessary to set such a gold standard of utilization rate and steps toward achievement.
Acknowledgments
This research was supported by grant 15521 from the US Department of Veterans Affairs, Office of Rural Health . Furthermore, the research was supported in part by grant K12 HD055929 from the National Institutes of Health.
1. US Department of Veterans Affairs, Veteran Health Administration, Office of Rural Health. Rural veteran health care challenges. Updated February 9, 2021. Accessed June 11, 2021. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp
2. Holder, K.A. Veterans in rural America, 2011–2015. Published January 2017. Accessed June 11, 2021. https://www.census.gov/content/dam/Census/library/publications/2017/acs/acs-36.pdf
3. Pezzin LE, Bogner HR, Kurichi JE, et al. Preventable hospitalizations, barriers to care, and disability. Medicine (Baltimore). 2018;97(19):e0691. doi:10.1097/MD.0000000000010691
4. Rosenbach ML. Access and satisfaction within the disabled Medicare population. Health Care Financ Rev. 1995;17(2):147-167.
5. Semeah LM, Ganesh SP, Wang X, et al. Home modification and health services utilization in rural and urban veterans with disabilities. Housing Policy Debate. 2021. Published online: March 4, 2021. doi:10.1080/10511482.2020.1858923
6. Spoont M, Greer N, Su J, Fitzgerald P, Rutks I, and Wilt TJ. Rural vs. urban ambulatory health care: A Systematic Review. Published May 2011. Accessed June 11, 2021. https://www.hsrd.research.va.gov/publications/esp/ambulatory.pdf
7. Semeah LM, Wang X, Cowper Ripley DC, et al. Improving health through a home modification service for veterans. In: Fiedler BA, ed. Three Facets of Public Health and Paths to Improvements. Academic Press; 2020:381-416.
8. Semeah LM, Ahrentzen S, Jia H, Cowper-Ripley DC, Levy CE, Mann WC. The home improvements and structural alterations benefits program: veterans with disabilities and home accessibility. J Disability Policy Studies. 2017;28(1):43-51. doi:10.1177/1044207317696275
9. Lucas, JW, Zelaya, CE. Hearing difficulty, vision trouble, and balance problems among male veterans and nonveterans. Published June 12, 2020. Accessed June 11, 2021. https://www.cdc.gov/nchs/data/nhsr/nhsr142-508.pdf
10. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Women veterans report: the past, present, and future of women veterans. Published February 2017. Accessed June 11, 2021. https://www.va.gov/vetdata/docs/SpecialReports/Women_Veterans_2015_Final.pdf
11. US Department of Housing and Urban Development, Office of Policy Development and Research. Housing challenges of rural seniors. Published 2017. Accessed June 11, 2021. https://www.huduser.gov/portal/periodicals/em/summer17/highlight1.html
12. Pendall R, Goodman L, Zhu J, Gold A. The future of rural housing. Published October 2016. Accessed June 11, 202.1 https://www.urban.org/sites/default/files/publication/85101/2000972-the-future-of-rural-housing_6.pdf
13. Joint Center for Housing Studies at Harvard University. Improving America’s housing 2019. Published 2019. Accessed June 11, 2021. https://www.jchs.harvard.edu/sites/default/files/reports/files/Harvard_JCHS_Improving_Americas_Housing_2019.pdf
14. Schooley BL, Horan TA, Lee PW, West PA. Rural veteran access to healthcare services: investigating the role of information and communication technologies in overcoming spatial barriers. Perspect Health Inf Manag. 2010;7(Spring):1f. Published 2010 Apr 1.
15. Ripley DC, Kwong PL, Vogel WB, Kurichi JE, Bates BE, Davenport C. How does geographic access affect in-hospital mortality for veterans with acute ischemic stroke?. Med Care. 2015;53(6):501-509. doi:10.1097/MLR.0000000000000366
16. Cowper-Ripley DC, Reker DM, Hayes J, et al. Geographic access to VHA rehabilitation services for traumatically injured veterans. Fed Pract. 2009;26(10):28-39.
17. Smith M, Towne S, Herrera-Venson A, Cameron K, Horel S, Ory M, et al. Delivery of fall prevention interventions for at-risk older adults in rural areas: Findings from a national dissemination. International journal of environmental research and public health. 2018;15:2798. doi: 10.3390/ijerph15122798
18. Hale-Gallardo JL, Kreider CM, Jia H, et al. Telerehabilitation for Rural Veterans: A Qualitative Assessment of Barriers and Facilitators to Implementation. J Multidiscip Healthc. 2020;13:559-570. doi:10.2147/JMDH.S247267
19. Sarfo FS, Akassi J, Kyem G, et al. Long-Term Outcomes of Stroke in a Ghanaian Outpatient Clinic. J Stroke Cerebrovasc Dis. 2018;27(4):1090-1099. doi:10.1016/j.jstrokecerebrovasdis.2017.11.017
1. US Department of Veterans Affairs, Veteran Health Administration, Office of Rural Health. Rural veteran health care challenges. Updated February 9, 2021. Accessed June 11, 2021. https://www.ruralhealth.va.gov/aboutus/ruralvets.asp
2. Holder, K.A. Veterans in rural America, 2011–2015. Published January 2017. Accessed June 11, 2021. https://www.census.gov/content/dam/Census/library/publications/2017/acs/acs-36.pdf
3. Pezzin LE, Bogner HR, Kurichi JE, et al. Preventable hospitalizations, barriers to care, and disability. Medicine (Baltimore). 2018;97(19):e0691. doi:10.1097/MD.0000000000010691
4. Rosenbach ML. Access and satisfaction within the disabled Medicare population. Health Care Financ Rev. 1995;17(2):147-167.
5. Semeah LM, Ganesh SP, Wang X, et al. Home modification and health services utilization in rural and urban veterans with disabilities. Housing Policy Debate. 2021. Published online: March 4, 2021. doi:10.1080/10511482.2020.1858923
6. Spoont M, Greer N, Su J, Fitzgerald P, Rutks I, and Wilt TJ. Rural vs. urban ambulatory health care: A Systematic Review. Published May 2011. Accessed June 11, 2021. https://www.hsrd.research.va.gov/publications/esp/ambulatory.pdf
7. Semeah LM, Wang X, Cowper Ripley DC, et al. Improving health through a home modification service for veterans. In: Fiedler BA, ed. Three Facets of Public Health and Paths to Improvements. Academic Press; 2020:381-416.
8. Semeah LM, Ahrentzen S, Jia H, Cowper-Ripley DC, Levy CE, Mann WC. The home improvements and structural alterations benefits program: veterans with disabilities and home accessibility. J Disability Policy Studies. 2017;28(1):43-51. doi:10.1177/1044207317696275
9. Lucas, JW, Zelaya, CE. Hearing difficulty, vision trouble, and balance problems among male veterans and nonveterans. Published June 12, 2020. Accessed June 11, 2021. https://www.cdc.gov/nchs/data/nhsr/nhsr142-508.pdf
10. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Women veterans report: the past, present, and future of women veterans. Published February 2017. Accessed June 11, 2021. https://www.va.gov/vetdata/docs/SpecialReports/Women_Veterans_2015_Final.pdf
11. US Department of Housing and Urban Development, Office of Policy Development and Research. Housing challenges of rural seniors. Published 2017. Accessed June 11, 2021. https://www.huduser.gov/portal/periodicals/em/summer17/highlight1.html
12. Pendall R, Goodman L, Zhu J, Gold A. The future of rural housing. Published October 2016. Accessed June 11, 202.1 https://www.urban.org/sites/default/files/publication/85101/2000972-the-future-of-rural-housing_6.pdf
13. Joint Center for Housing Studies at Harvard University. Improving America’s housing 2019. Published 2019. Accessed June 11, 2021. https://www.jchs.harvard.edu/sites/default/files/reports/files/Harvard_JCHS_Improving_Americas_Housing_2019.pdf
14. Schooley BL, Horan TA, Lee PW, West PA. Rural veteran access to healthcare services: investigating the role of information and communication technologies in overcoming spatial barriers. Perspect Health Inf Manag. 2010;7(Spring):1f. Published 2010 Apr 1.
15. Ripley DC, Kwong PL, Vogel WB, Kurichi JE, Bates BE, Davenport C. How does geographic access affect in-hospital mortality for veterans with acute ischemic stroke?. Med Care. 2015;53(6):501-509. doi:10.1097/MLR.0000000000000366
16. Cowper-Ripley DC, Reker DM, Hayes J, et al. Geographic access to VHA rehabilitation services for traumatically injured veterans. Fed Pract. 2009;26(10):28-39.
17. Smith M, Towne S, Herrera-Venson A, Cameron K, Horel S, Ory M, et al. Delivery of fall prevention interventions for at-risk older adults in rural areas: Findings from a national dissemination. International journal of environmental research and public health. 2018;15:2798. doi: 10.3390/ijerph15122798
18. Hale-Gallardo JL, Kreider CM, Jia H, et al. Telerehabilitation for Rural Veterans: A Qualitative Assessment of Barriers and Facilitators to Implementation. J Multidiscip Healthc. 2020;13:559-570. doi:10.2147/JMDH.S247267
19. Sarfo FS, Akassi J, Kyem G, et al. Long-Term Outcomes of Stroke in a Ghanaian Outpatient Clinic. J Stroke Cerebrovasc Dis. 2018;27(4):1090-1099. doi:10.1016/j.jstrokecerebrovasdis.2017.11.017
Assessment of a Medication Deprescribing Tool on Polypharmacy and Cost Avoidance
According to the Centers for Disease Control and Prevention National Center for Health Statistics (NCHS), the use of prescription drugs has increased in the past half century. Although prescription drugs have played an important role in preventing, controlling, and delaying onset or progression of disease, their growth in use also has posed many risks.1 One ramification of this growth is the occurrence of polypharmacy, which does not have a universal, clear definition. In general, it can be described as the concurrent use of multiple medications by a single patient to treat one or more medical ailments. Five or more medications taken simultaneously is the most common definition to date, but this is just one of many acceptable definitions and that varies from one health care facility to another.1,2
Regardless of the cutoffs established to indicate polypharmacy, its incidence can result in poor and potentially harmful health outcomes. Polypharmacy increases the risk of experiencing adverse drug events (ADEs), drug-drug interactions (DDIs), geriatric-related syndromes, falls, hospitalization, and mortality. Issues with adherence may begin to unfold secondary to increased pill burden. Both the patient and the health care system may encounter financial strain, as polypharmacy can lead to unnecessary and essentially preventable costs of care. When evaluating the likelihood of polypharmacy based on age group, NCHS found that 47.5% of patients taking ≥ 5 medications were aged ≥ 65 years.1-5 This indicates that polypharmacy is of great concern in the geriatric population, which also represents a large proportion of individuals accessing Veterans Health Administration (VHA) care.
Deprescibing
Deprescribing is the act of withdrawing or discontinuing potentially inappropriate medications (PIM), or medications used by older patients harboring ADEs that generally outweigh the clinical benefits of the drug. Deprescribing is an effective tool for managing or reducing polypharmacy. A variety of tools have been created whose sole purpose is to simplify deprescribing. Some tools explicitly identify PIM and are widely familiar in medical practice. Examples are the Beers Criteria developed in 1991 or Screening Tool to Alert Right Treatment/Screening Tool of Older Persons Prescriptions (START/STOPP) criteria created in 2003. Other tools that are less commonplace but equally as resourceful are MedStopper and Deprescribing.org. The former was launched in 2015 and is a Canadian online system that provides risk assessments for medications with guidance for tapering or stopping medications if continuation of the drug presents higher risk than benefit.5-7 The latter is a full-fledged website developed by a physician, a pharmacist, and their research teams that serves as an exchange hub for deprescribing information.
In 2016, the VIONE (Vital, Important, Optional, Not indicated/treatment complete, and Every medication has an indication) deprescribing tool was developed by Saraswathy Battar, MD, at Central Arkansas Veterans Healthcare System (CAVHS) in Little Rock, as a system that could go beyond medication reconciliation (Table 1). Health care providers (HCPs) and pharmacists evaluate each medication that a patient has been prescribed and places each medication in a VIONE category. Prescribers may then take the opportunity to deprescribe or discontinue medications if deemed appropriate based on their clinical assessments and shared decision making.8 Traditionally, medication reconciliation involves the process of obtaining a complete and accurate list of medications as reported by a patient or caregiver to a HCP. VIONE encourages HCPs and pharmacists not only to ensure medication lists are accurate, but also that each medication reported is appropriate for continued use. In other words, VIONE is meant to help implement deprescribing at opportune times. More than 14,000 medications have been deprescribed using the VIONE method, resulting in more than $2,000,000 of annualized cost avoidance after just 1 year of implementation at CAVHS.9
VIONE consists of 2 major components in the Computerized Patient Record System (CPRS): a template and a dropdown discontinuation menu. The template captured patient allergies, pertinent laboratory data, the patient’s active problem list and applicable diagnoses, and active medication list. Patient aligned care team (PACT) pharmacists used the information captured in the template to conduct medication reconciliations and polypharmacy reviews. Each medication is categorized in VIONE using data collected during reviews. A menu delineates reasons for discontinuation: optional, dose decrease, no diagnosis, not indicated/treatment complete, discontinue alternate medication prescribed, and patient reported no longer taking. The discontinuation menu allowed PACT pharmacists and physicians to choose 1 VIONE option per medication to clarify the reason for discontinuation. VIONE-based discontinuations are recorded in CPRS and identified as deprescribed.
At the time of this project, > 30 US Department of Veterans Affairs (VA) facilities had adopted VIONE. Use of VIONE at VA Southern Nevada Healthcare System (VASNHS) in North Las Vegas has been incorporated in the everyday practices of home-based primary care pharmacists and physicians but has yet to be implemented in other areas of the facility. The purpose of this project was to determine the impact of the VIONE tool on polypharmacy and cost avoidance at VASNHS when used by primary care physicians (PCPs) and PACT primary care clinics.
Methods
Veterans receiving care at VASNHS aged ≥ 65 years with ≥ 10 active medications noted in CPRS were included in this project. PACT pharmacists and physicians were educated on the proper use of the VIONE tool prior to its implementation. Education included a 15-minute slide presentation followed by dissemination of a 1-page VIONE tool handout during a PACT all-staff clinic meeting.
Data were collected for 3 months before and after the intervention. Data were made available for assessment by the Automated Data Processing Application Coordinator (ADPAC) at VASNHS. The ADPAC created and generated an Excel spreadsheet report, which listed all medications deprescribed using the VIONE method. The primary endpoint was the total number of medications discontinued using the VIONE template and/or discontinuation menu. For the purpose of this project, appropriate discontinuation was considered any prescription deprescribed, excluding medical supplies, by pharmacists and PCPs who received VIONE education.
The secondary endpoint was the estimated annualized cost avoidance for the facility (Figure). The calculation does not include medications discontinued due to the prescription of an alternative medication or dose decreases since these VIONE selections imply that a new prescription or order was placed and the original prescription was not deprescribed. Annualized cost avoidance was determined with use of the VIONE dashboard, a database that retrospectively gathers information regarding patients at risk of polypharmacy, polypharmacy-related ADEs, and cost. Manual adjustments were made to various parameters on the Veterans Integrated Service Network 15 VIONE dashboard by the author in order to obtain data specific to this project. These parameters allowed selection of service sections, specific staff members or the option to include or exclude chronic or nonchronic medications. The annualized cost avoidance figure was then compared to raw data pulled by a VIONE dashboard correspondent to ensure the manual calculation was accurate. Finally, the 5 most common classes of medications deprescribed were identified for information purposes and to provide a better postulation on the types of medications being discontinued using the VIONE method.
Results
A total of 2,442 veterans met inclusion criteria, and the VIONE method was applied to 598 between late October 2018 and January 2019. The 13 PACT pharmacists contacted at least 10 veterans each, thus at least 130 were randomly selected for telephone calls to perform polypharmacy reviews using the VIONE note template. The discontinuation menu was used if a medication qualified to be deprescribed. After 3 months, 1986 prescriptions were deprescribed using VIONE; however, 1060 prescriptions were considered appropriately deprescribed (Table 2). The 13 PACT pharmacists deprescribed 361 medications, and the 29 PACT physicians deprescribed 699 medications. These prescriptions were then separated into medication categories to determine the most common discontinued classes. Vitamins and supplements were the medication class most frequently deprescribed (19.4%), followed by pain medications (15.5%), antimicrobial agents (9.6%), antihypertensive medications (9.2%), and diabetes medications (6.4%) (Table 3). The top 5 medication categories accounted for 60% of all medications appropriately deprescribed.
The estimated annualized cost avoidance for all medications deprescribed in the 3-month project period was $84,030.46. To provide the most appropriate and accurate calculation, medication classes excluded from this figure were acute or short-term prescriptions and antimicrobial agents. Medications prescribed short-term typically are not suitable to continue for an extended period, and antimicrobial agents were excluded since they are normally associated with higher costs, and may overestimate the cost avoidance calculation for the facility.
Discussion
The outcomes for the primary and secondary endpoints of this project illustrate that using VIONE in PACT primary care clinics had a notable impact on polypharmacy and cost avoidance over a short period. This outcome can be attributed to 2 significant effects of using the deprescribing tool. VIONE’s simplicity in application allowed clinicians to incorporate daily use of the tool with minimal effort. Education was all that was required to fully enable clinicians to work together successfully and exercise collaborative practice to promote deprescribing. VIONE also elicited a cascade of favorable effects that improve patient safety and health outcomes. The tool aided in identification of PIM, which helped reduce polypharmacy and medication burden. The risk for DDIs and ADEs may decrease; therefore, the incidence of falls, need for emergency department visits or inpatient care related to polypharmacy may decline. Less complex medication regimens may alleviate issues with adherence and avoid the various consequences of polypharmacy in theory. Simplified regimens can potentially improve disease management and quality of life for patients. Further studies are needed to substantiate deprescribing and its true effect on patient adherence and better health outcomes at this time.10
Reducing polypharmacy can lead to cost savings. Based on the results of this 3-month study, we expect that VASNHS would save more than $84,000 by reducing polypharmacy among its patients. Those savings can be funneled back into the health care system, and allotted to necessary patient care, prescriptions, and health care facility needs.
Limitations
There are some important limitations to this study. Definitions of polypharmacy may vary from one health care facility to another. The cutoffs for polypharmacy may differ, causing the prevalence of polypharmacy and potential costs savings to vary. Use of VIONE may be inconsistent among users if not previously educated or properly trained. For instance, VIONE selections are listed in the same menu as the standard CPRS discontinuation options, which may lead to discontinuation of medical supplies or laboratory orders instead of prescriptions.
The method of data analysis and project design used in this study may have been subject to error. For example, the list of PCPs may have been inaccurate or outdated, which would result in an over- or underrepresentation of those who contributed to data collection. Furthermore, there is some volatility in calculating the total cost avoidance. For example, medications for chronic conditions that were only taken on an as needed basis may have overestimated savings. Either under- or overestimations could occur when parameters are adjusted on the VIONE discontinuation dashboard without appropriate guidance. With the ability to manually adjust the dashboard parameters, dissimilarities in calculations may follow.
Conclusions
The VIONE tool may be useful in improving patient safety through deprescribing and discontinuing PIM. Decreasing the number of medications being taken concomitantly by a patient and continuing only those that are imperative in their medical treatment is the first step to reducing the incidence of polypharmacy. Consequently, chances of ADEs or DDIs are lessened, especially among older individuals who are considered high risk for experiencing the detrimental effects that may ensue. These effects include geriatric-related syndromes, increased risk of fall, hospital visits or admissions, or death. Use of VIONE easily promotes collaboration among clinicians to evaluate medications eligible for discontinuation more regularly. If this deprescribing tool is continuously used, costs avoided can likely be maximized within VA health care systems.
The results of this project should serve as an incentive to push for better prescribing practices and increase deprescribing efforts. It should provoke the need for change in regimens and the subsequent discontinuation of prescriptions that are not considered vital to continue. Finally, the result of this project should substantiate the positive impact a deprescribing tool can possess to avert the issues commonly associated with polypharmacy.
1. Centers for Disease Control and Prevention, National Center for Health Statistics. Health, United States, 2013: with special feature on prescription drugs. Published May 2014. Accessed May 13, 2021. https://www.cdc.gov/nchs/data/hus/hus13.pdf
2. Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE. What is polypharmacy? A systematic review of definitions. BMC Geriatr. 2017;17(1):230. Published 2017 Oct 10. doi:10.1186/s12877-017-0621-2
3. Parulekar MS, Rogers CK. Polypharmacy and mobility. In: Cifu DX, Lew HL, Oh-Park M., eds Geriatric Rehabilitation. Elsevier; 2018. doi:10.1016/B978-0-323-54454-2.12001-1
4. Rieckert A, Trampisch US, Klaaßen-Mielke R, et al. Polypharmacy in older patients with chronic diseases: a cross-sectional analysis of factors associated with excessive polypharmacy. BMC Fam Pract. 2018;19(1):113. Published 2018 Jul 18. doi:10.1186/s12875-018-0795-5
5. Thompson CA. New medication review method cuts veterans’ Rx load, saves millions. Am J Health Syst Pharm. 2018;75(8):502-503. doi:10.2146/news180023
6. Reeve E. Deprescribing tools: a review of the types of tools available to aid deprescribing in clinical practice. J Pharm Pract Res. 2020;50(1):98-107. doi:10.1002/jppr.1626
7. Fried TR, Niehoff KM, Street RL, et al. Effect of the Tool to Reduce Inappropriate Medications on Medication Communication and Deprescribing. J Am Geriatr Soc. 2017;65(10):2265-2271. doi:10.1111/jgs.15042
8. Battar S, Dickerson KR, Sedgwick C, et al. Understanding principles of high reliability organizations through the eyes of VIONE, a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564-568.
9. Battar S, Cmelik T, Dickerson K, Scott, M. Experience better health with VIONE a safe medication deprescribing tool [Nonpublic source, not verified]
10. Ulley J, Harrop D, Ali A, et al. Desprescribing interventions and their impact on medication adherence in community-dwelling older adults with polypharmacy: a systematic review. BMC Geriatr. 2019;19(15):1-13.
According to the Centers for Disease Control and Prevention National Center for Health Statistics (NCHS), the use of prescription drugs has increased in the past half century. Although prescription drugs have played an important role in preventing, controlling, and delaying onset or progression of disease, their growth in use also has posed many risks.1 One ramification of this growth is the occurrence of polypharmacy, which does not have a universal, clear definition. In general, it can be described as the concurrent use of multiple medications by a single patient to treat one or more medical ailments. Five or more medications taken simultaneously is the most common definition to date, but this is just one of many acceptable definitions and that varies from one health care facility to another.1,2
Regardless of the cutoffs established to indicate polypharmacy, its incidence can result in poor and potentially harmful health outcomes. Polypharmacy increases the risk of experiencing adverse drug events (ADEs), drug-drug interactions (DDIs), geriatric-related syndromes, falls, hospitalization, and mortality. Issues with adherence may begin to unfold secondary to increased pill burden. Both the patient and the health care system may encounter financial strain, as polypharmacy can lead to unnecessary and essentially preventable costs of care. When evaluating the likelihood of polypharmacy based on age group, NCHS found that 47.5% of patients taking ≥ 5 medications were aged ≥ 65 years.1-5 This indicates that polypharmacy is of great concern in the geriatric population, which also represents a large proportion of individuals accessing Veterans Health Administration (VHA) care.
Deprescibing
Deprescribing is the act of withdrawing or discontinuing potentially inappropriate medications (PIM), or medications used by older patients harboring ADEs that generally outweigh the clinical benefits of the drug. Deprescribing is an effective tool for managing or reducing polypharmacy. A variety of tools have been created whose sole purpose is to simplify deprescribing. Some tools explicitly identify PIM and are widely familiar in medical practice. Examples are the Beers Criteria developed in 1991 or Screening Tool to Alert Right Treatment/Screening Tool of Older Persons Prescriptions (START/STOPP) criteria created in 2003. Other tools that are less commonplace but equally as resourceful are MedStopper and Deprescribing.org. The former was launched in 2015 and is a Canadian online system that provides risk assessments for medications with guidance for tapering or stopping medications if continuation of the drug presents higher risk than benefit.5-7 The latter is a full-fledged website developed by a physician, a pharmacist, and their research teams that serves as an exchange hub for deprescribing information.
In 2016, the VIONE (Vital, Important, Optional, Not indicated/treatment complete, and Every medication has an indication) deprescribing tool was developed by Saraswathy Battar, MD, at Central Arkansas Veterans Healthcare System (CAVHS) in Little Rock, as a system that could go beyond medication reconciliation (Table 1). Health care providers (HCPs) and pharmacists evaluate each medication that a patient has been prescribed and places each medication in a VIONE category. Prescribers may then take the opportunity to deprescribe or discontinue medications if deemed appropriate based on their clinical assessments and shared decision making.8 Traditionally, medication reconciliation involves the process of obtaining a complete and accurate list of medications as reported by a patient or caregiver to a HCP. VIONE encourages HCPs and pharmacists not only to ensure medication lists are accurate, but also that each medication reported is appropriate for continued use. In other words, VIONE is meant to help implement deprescribing at opportune times. More than 14,000 medications have been deprescribed using the VIONE method, resulting in more than $2,000,000 of annualized cost avoidance after just 1 year of implementation at CAVHS.9
VIONE consists of 2 major components in the Computerized Patient Record System (CPRS): a template and a dropdown discontinuation menu. The template captured patient allergies, pertinent laboratory data, the patient’s active problem list and applicable diagnoses, and active medication list. Patient aligned care team (PACT) pharmacists used the information captured in the template to conduct medication reconciliations and polypharmacy reviews. Each medication is categorized in VIONE using data collected during reviews. A menu delineates reasons for discontinuation: optional, dose decrease, no diagnosis, not indicated/treatment complete, discontinue alternate medication prescribed, and patient reported no longer taking. The discontinuation menu allowed PACT pharmacists and physicians to choose 1 VIONE option per medication to clarify the reason for discontinuation. VIONE-based discontinuations are recorded in CPRS and identified as deprescribed.
At the time of this project, > 30 US Department of Veterans Affairs (VA) facilities had adopted VIONE. Use of VIONE at VA Southern Nevada Healthcare System (VASNHS) in North Las Vegas has been incorporated in the everyday practices of home-based primary care pharmacists and physicians but has yet to be implemented in other areas of the facility. The purpose of this project was to determine the impact of the VIONE tool on polypharmacy and cost avoidance at VASNHS when used by primary care physicians (PCPs) and PACT primary care clinics.
Methods
Veterans receiving care at VASNHS aged ≥ 65 years with ≥ 10 active medications noted in CPRS were included in this project. PACT pharmacists and physicians were educated on the proper use of the VIONE tool prior to its implementation. Education included a 15-minute slide presentation followed by dissemination of a 1-page VIONE tool handout during a PACT all-staff clinic meeting.
Data were collected for 3 months before and after the intervention. Data were made available for assessment by the Automated Data Processing Application Coordinator (ADPAC) at VASNHS. The ADPAC created and generated an Excel spreadsheet report, which listed all medications deprescribed using the VIONE method. The primary endpoint was the total number of medications discontinued using the VIONE template and/or discontinuation menu. For the purpose of this project, appropriate discontinuation was considered any prescription deprescribed, excluding medical supplies, by pharmacists and PCPs who received VIONE education.
The secondary endpoint was the estimated annualized cost avoidance for the facility (Figure). The calculation does not include medications discontinued due to the prescription of an alternative medication or dose decreases since these VIONE selections imply that a new prescription or order was placed and the original prescription was not deprescribed. Annualized cost avoidance was determined with use of the VIONE dashboard, a database that retrospectively gathers information regarding patients at risk of polypharmacy, polypharmacy-related ADEs, and cost. Manual adjustments were made to various parameters on the Veterans Integrated Service Network 15 VIONE dashboard by the author in order to obtain data specific to this project. These parameters allowed selection of service sections, specific staff members or the option to include or exclude chronic or nonchronic medications. The annualized cost avoidance figure was then compared to raw data pulled by a VIONE dashboard correspondent to ensure the manual calculation was accurate. Finally, the 5 most common classes of medications deprescribed were identified for information purposes and to provide a better postulation on the types of medications being discontinued using the VIONE method.
Results
A total of 2,442 veterans met inclusion criteria, and the VIONE method was applied to 598 between late October 2018 and January 2019. The 13 PACT pharmacists contacted at least 10 veterans each, thus at least 130 were randomly selected for telephone calls to perform polypharmacy reviews using the VIONE note template. The discontinuation menu was used if a medication qualified to be deprescribed. After 3 months, 1986 prescriptions were deprescribed using VIONE; however, 1060 prescriptions were considered appropriately deprescribed (Table 2). The 13 PACT pharmacists deprescribed 361 medications, and the 29 PACT physicians deprescribed 699 medications. These prescriptions were then separated into medication categories to determine the most common discontinued classes. Vitamins and supplements were the medication class most frequently deprescribed (19.4%), followed by pain medications (15.5%), antimicrobial agents (9.6%), antihypertensive medications (9.2%), and diabetes medications (6.4%) (Table 3). The top 5 medication categories accounted for 60% of all medications appropriately deprescribed.
The estimated annualized cost avoidance for all medications deprescribed in the 3-month project period was $84,030.46. To provide the most appropriate and accurate calculation, medication classes excluded from this figure were acute or short-term prescriptions and antimicrobial agents. Medications prescribed short-term typically are not suitable to continue for an extended period, and antimicrobial agents were excluded since they are normally associated with higher costs, and may overestimate the cost avoidance calculation for the facility.
Discussion
The outcomes for the primary and secondary endpoints of this project illustrate that using VIONE in PACT primary care clinics had a notable impact on polypharmacy and cost avoidance over a short period. This outcome can be attributed to 2 significant effects of using the deprescribing tool. VIONE’s simplicity in application allowed clinicians to incorporate daily use of the tool with minimal effort. Education was all that was required to fully enable clinicians to work together successfully and exercise collaborative practice to promote deprescribing. VIONE also elicited a cascade of favorable effects that improve patient safety and health outcomes. The tool aided in identification of PIM, which helped reduce polypharmacy and medication burden. The risk for DDIs and ADEs may decrease; therefore, the incidence of falls, need for emergency department visits or inpatient care related to polypharmacy may decline. Less complex medication regimens may alleviate issues with adherence and avoid the various consequences of polypharmacy in theory. Simplified regimens can potentially improve disease management and quality of life for patients. Further studies are needed to substantiate deprescribing and its true effect on patient adherence and better health outcomes at this time.10
Reducing polypharmacy can lead to cost savings. Based on the results of this 3-month study, we expect that VASNHS would save more than $84,000 by reducing polypharmacy among its patients. Those savings can be funneled back into the health care system, and allotted to necessary patient care, prescriptions, and health care facility needs.
Limitations
There are some important limitations to this study. Definitions of polypharmacy may vary from one health care facility to another. The cutoffs for polypharmacy may differ, causing the prevalence of polypharmacy and potential costs savings to vary. Use of VIONE may be inconsistent among users if not previously educated or properly trained. For instance, VIONE selections are listed in the same menu as the standard CPRS discontinuation options, which may lead to discontinuation of medical supplies or laboratory orders instead of prescriptions.
The method of data analysis and project design used in this study may have been subject to error. For example, the list of PCPs may have been inaccurate or outdated, which would result in an over- or underrepresentation of those who contributed to data collection. Furthermore, there is some volatility in calculating the total cost avoidance. For example, medications for chronic conditions that were only taken on an as needed basis may have overestimated savings. Either under- or overestimations could occur when parameters are adjusted on the VIONE discontinuation dashboard without appropriate guidance. With the ability to manually adjust the dashboard parameters, dissimilarities in calculations may follow.
Conclusions
The VIONE tool may be useful in improving patient safety through deprescribing and discontinuing PIM. Decreasing the number of medications being taken concomitantly by a patient and continuing only those that are imperative in their medical treatment is the first step to reducing the incidence of polypharmacy. Consequently, chances of ADEs or DDIs are lessened, especially among older individuals who are considered high risk for experiencing the detrimental effects that may ensue. These effects include geriatric-related syndromes, increased risk of fall, hospital visits or admissions, or death. Use of VIONE easily promotes collaboration among clinicians to evaluate medications eligible for discontinuation more regularly. If this deprescribing tool is continuously used, costs avoided can likely be maximized within VA health care systems.
The results of this project should serve as an incentive to push for better prescribing practices and increase deprescribing efforts. It should provoke the need for change in regimens and the subsequent discontinuation of prescriptions that are not considered vital to continue. Finally, the result of this project should substantiate the positive impact a deprescribing tool can possess to avert the issues commonly associated with polypharmacy.
According to the Centers for Disease Control and Prevention National Center for Health Statistics (NCHS), the use of prescription drugs has increased in the past half century. Although prescription drugs have played an important role in preventing, controlling, and delaying onset or progression of disease, their growth in use also has posed many risks.1 One ramification of this growth is the occurrence of polypharmacy, which does not have a universal, clear definition. In general, it can be described as the concurrent use of multiple medications by a single patient to treat one or more medical ailments. Five or more medications taken simultaneously is the most common definition to date, but this is just one of many acceptable definitions and that varies from one health care facility to another.1,2
Regardless of the cutoffs established to indicate polypharmacy, its incidence can result in poor and potentially harmful health outcomes. Polypharmacy increases the risk of experiencing adverse drug events (ADEs), drug-drug interactions (DDIs), geriatric-related syndromes, falls, hospitalization, and mortality. Issues with adherence may begin to unfold secondary to increased pill burden. Both the patient and the health care system may encounter financial strain, as polypharmacy can lead to unnecessary and essentially preventable costs of care. When evaluating the likelihood of polypharmacy based on age group, NCHS found that 47.5% of patients taking ≥ 5 medications were aged ≥ 65 years.1-5 This indicates that polypharmacy is of great concern in the geriatric population, which also represents a large proportion of individuals accessing Veterans Health Administration (VHA) care.
Deprescibing
Deprescribing is the act of withdrawing or discontinuing potentially inappropriate medications (PIM), or medications used by older patients harboring ADEs that generally outweigh the clinical benefits of the drug. Deprescribing is an effective tool for managing or reducing polypharmacy. A variety of tools have been created whose sole purpose is to simplify deprescribing. Some tools explicitly identify PIM and are widely familiar in medical practice. Examples are the Beers Criteria developed in 1991 or Screening Tool to Alert Right Treatment/Screening Tool of Older Persons Prescriptions (START/STOPP) criteria created in 2003. Other tools that are less commonplace but equally as resourceful are MedStopper and Deprescribing.org. The former was launched in 2015 and is a Canadian online system that provides risk assessments for medications with guidance for tapering or stopping medications if continuation of the drug presents higher risk than benefit.5-7 The latter is a full-fledged website developed by a physician, a pharmacist, and their research teams that serves as an exchange hub for deprescribing information.
In 2016, the VIONE (Vital, Important, Optional, Not indicated/treatment complete, and Every medication has an indication) deprescribing tool was developed by Saraswathy Battar, MD, at Central Arkansas Veterans Healthcare System (CAVHS) in Little Rock, as a system that could go beyond medication reconciliation (Table 1). Health care providers (HCPs) and pharmacists evaluate each medication that a patient has been prescribed and places each medication in a VIONE category. Prescribers may then take the opportunity to deprescribe or discontinue medications if deemed appropriate based on their clinical assessments and shared decision making.8 Traditionally, medication reconciliation involves the process of obtaining a complete and accurate list of medications as reported by a patient or caregiver to a HCP. VIONE encourages HCPs and pharmacists not only to ensure medication lists are accurate, but also that each medication reported is appropriate for continued use. In other words, VIONE is meant to help implement deprescribing at opportune times. More than 14,000 medications have been deprescribed using the VIONE method, resulting in more than $2,000,000 of annualized cost avoidance after just 1 year of implementation at CAVHS.9
VIONE consists of 2 major components in the Computerized Patient Record System (CPRS): a template and a dropdown discontinuation menu. The template captured patient allergies, pertinent laboratory data, the patient’s active problem list and applicable diagnoses, and active medication list. Patient aligned care team (PACT) pharmacists used the information captured in the template to conduct medication reconciliations and polypharmacy reviews. Each medication is categorized in VIONE using data collected during reviews. A menu delineates reasons for discontinuation: optional, dose decrease, no diagnosis, not indicated/treatment complete, discontinue alternate medication prescribed, and patient reported no longer taking. The discontinuation menu allowed PACT pharmacists and physicians to choose 1 VIONE option per medication to clarify the reason for discontinuation. VIONE-based discontinuations are recorded in CPRS and identified as deprescribed.
At the time of this project, > 30 US Department of Veterans Affairs (VA) facilities had adopted VIONE. Use of VIONE at VA Southern Nevada Healthcare System (VASNHS) in North Las Vegas has been incorporated in the everyday practices of home-based primary care pharmacists and physicians but has yet to be implemented in other areas of the facility. The purpose of this project was to determine the impact of the VIONE tool on polypharmacy and cost avoidance at VASNHS when used by primary care physicians (PCPs) and PACT primary care clinics.
Methods
Veterans receiving care at VASNHS aged ≥ 65 years with ≥ 10 active medications noted in CPRS were included in this project. PACT pharmacists and physicians were educated on the proper use of the VIONE tool prior to its implementation. Education included a 15-minute slide presentation followed by dissemination of a 1-page VIONE tool handout during a PACT all-staff clinic meeting.
Data were collected for 3 months before and after the intervention. Data were made available for assessment by the Automated Data Processing Application Coordinator (ADPAC) at VASNHS. The ADPAC created and generated an Excel spreadsheet report, which listed all medications deprescribed using the VIONE method. The primary endpoint was the total number of medications discontinued using the VIONE template and/or discontinuation menu. For the purpose of this project, appropriate discontinuation was considered any prescription deprescribed, excluding medical supplies, by pharmacists and PCPs who received VIONE education.
The secondary endpoint was the estimated annualized cost avoidance for the facility (Figure). The calculation does not include medications discontinued due to the prescription of an alternative medication or dose decreases since these VIONE selections imply that a new prescription or order was placed and the original prescription was not deprescribed. Annualized cost avoidance was determined with use of the VIONE dashboard, a database that retrospectively gathers information regarding patients at risk of polypharmacy, polypharmacy-related ADEs, and cost. Manual adjustments were made to various parameters on the Veterans Integrated Service Network 15 VIONE dashboard by the author in order to obtain data specific to this project. These parameters allowed selection of service sections, specific staff members or the option to include or exclude chronic or nonchronic medications. The annualized cost avoidance figure was then compared to raw data pulled by a VIONE dashboard correspondent to ensure the manual calculation was accurate. Finally, the 5 most common classes of medications deprescribed were identified for information purposes and to provide a better postulation on the types of medications being discontinued using the VIONE method.
Results
A total of 2,442 veterans met inclusion criteria, and the VIONE method was applied to 598 between late October 2018 and January 2019. The 13 PACT pharmacists contacted at least 10 veterans each, thus at least 130 were randomly selected for telephone calls to perform polypharmacy reviews using the VIONE note template. The discontinuation menu was used if a medication qualified to be deprescribed. After 3 months, 1986 prescriptions were deprescribed using VIONE; however, 1060 prescriptions were considered appropriately deprescribed (Table 2). The 13 PACT pharmacists deprescribed 361 medications, and the 29 PACT physicians deprescribed 699 medications. These prescriptions were then separated into medication categories to determine the most common discontinued classes. Vitamins and supplements were the medication class most frequently deprescribed (19.4%), followed by pain medications (15.5%), antimicrobial agents (9.6%), antihypertensive medications (9.2%), and diabetes medications (6.4%) (Table 3). The top 5 medication categories accounted for 60% of all medications appropriately deprescribed.
The estimated annualized cost avoidance for all medications deprescribed in the 3-month project period was $84,030.46. To provide the most appropriate and accurate calculation, medication classes excluded from this figure were acute or short-term prescriptions and antimicrobial agents. Medications prescribed short-term typically are not suitable to continue for an extended period, and antimicrobial agents were excluded since they are normally associated with higher costs, and may overestimate the cost avoidance calculation for the facility.
Discussion
The outcomes for the primary and secondary endpoints of this project illustrate that using VIONE in PACT primary care clinics had a notable impact on polypharmacy and cost avoidance over a short period. This outcome can be attributed to 2 significant effects of using the deprescribing tool. VIONE’s simplicity in application allowed clinicians to incorporate daily use of the tool with minimal effort. Education was all that was required to fully enable clinicians to work together successfully and exercise collaborative practice to promote deprescribing. VIONE also elicited a cascade of favorable effects that improve patient safety and health outcomes. The tool aided in identification of PIM, which helped reduce polypharmacy and medication burden. The risk for DDIs and ADEs may decrease; therefore, the incidence of falls, need for emergency department visits or inpatient care related to polypharmacy may decline. Less complex medication regimens may alleviate issues with adherence and avoid the various consequences of polypharmacy in theory. Simplified regimens can potentially improve disease management and quality of life for patients. Further studies are needed to substantiate deprescribing and its true effect on patient adherence and better health outcomes at this time.10
Reducing polypharmacy can lead to cost savings. Based on the results of this 3-month study, we expect that VASNHS would save more than $84,000 by reducing polypharmacy among its patients. Those savings can be funneled back into the health care system, and allotted to necessary patient care, prescriptions, and health care facility needs.
Limitations
There are some important limitations to this study. Definitions of polypharmacy may vary from one health care facility to another. The cutoffs for polypharmacy may differ, causing the prevalence of polypharmacy and potential costs savings to vary. Use of VIONE may be inconsistent among users if not previously educated or properly trained. For instance, VIONE selections are listed in the same menu as the standard CPRS discontinuation options, which may lead to discontinuation of medical supplies or laboratory orders instead of prescriptions.
The method of data analysis and project design used in this study may have been subject to error. For example, the list of PCPs may have been inaccurate or outdated, which would result in an over- or underrepresentation of those who contributed to data collection. Furthermore, there is some volatility in calculating the total cost avoidance. For example, medications for chronic conditions that were only taken on an as needed basis may have overestimated savings. Either under- or overestimations could occur when parameters are adjusted on the VIONE discontinuation dashboard without appropriate guidance. With the ability to manually adjust the dashboard parameters, dissimilarities in calculations may follow.
Conclusions
The VIONE tool may be useful in improving patient safety through deprescribing and discontinuing PIM. Decreasing the number of medications being taken concomitantly by a patient and continuing only those that are imperative in their medical treatment is the first step to reducing the incidence of polypharmacy. Consequently, chances of ADEs or DDIs are lessened, especially among older individuals who are considered high risk for experiencing the detrimental effects that may ensue. These effects include geriatric-related syndromes, increased risk of fall, hospital visits or admissions, or death. Use of VIONE easily promotes collaboration among clinicians to evaluate medications eligible for discontinuation more regularly. If this deprescribing tool is continuously used, costs avoided can likely be maximized within VA health care systems.
The results of this project should serve as an incentive to push for better prescribing practices and increase deprescribing efforts. It should provoke the need for change in regimens and the subsequent discontinuation of prescriptions that are not considered vital to continue. Finally, the result of this project should substantiate the positive impact a deprescribing tool can possess to avert the issues commonly associated with polypharmacy.
1. Centers for Disease Control and Prevention, National Center for Health Statistics. Health, United States, 2013: with special feature on prescription drugs. Published May 2014. Accessed May 13, 2021. https://www.cdc.gov/nchs/data/hus/hus13.pdf
2. Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE. What is polypharmacy? A systematic review of definitions. BMC Geriatr. 2017;17(1):230. Published 2017 Oct 10. doi:10.1186/s12877-017-0621-2
3. Parulekar MS, Rogers CK. Polypharmacy and mobility. In: Cifu DX, Lew HL, Oh-Park M., eds Geriatric Rehabilitation. Elsevier; 2018. doi:10.1016/B978-0-323-54454-2.12001-1
4. Rieckert A, Trampisch US, Klaaßen-Mielke R, et al. Polypharmacy in older patients with chronic diseases: a cross-sectional analysis of factors associated with excessive polypharmacy. BMC Fam Pract. 2018;19(1):113. Published 2018 Jul 18. doi:10.1186/s12875-018-0795-5
5. Thompson CA. New medication review method cuts veterans’ Rx load, saves millions. Am J Health Syst Pharm. 2018;75(8):502-503. doi:10.2146/news180023
6. Reeve E. Deprescribing tools: a review of the types of tools available to aid deprescribing in clinical practice. J Pharm Pract Res. 2020;50(1):98-107. doi:10.1002/jppr.1626
7. Fried TR, Niehoff KM, Street RL, et al. Effect of the Tool to Reduce Inappropriate Medications on Medication Communication and Deprescribing. J Am Geriatr Soc. 2017;65(10):2265-2271. doi:10.1111/jgs.15042
8. Battar S, Dickerson KR, Sedgwick C, et al. Understanding principles of high reliability organizations through the eyes of VIONE, a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564-568.
9. Battar S, Cmelik T, Dickerson K, Scott, M. Experience better health with VIONE a safe medication deprescribing tool [Nonpublic source, not verified]
10. Ulley J, Harrop D, Ali A, et al. Desprescribing interventions and their impact on medication adherence in community-dwelling older adults with polypharmacy: a systematic review. BMC Geriatr. 2019;19(15):1-13.
1. Centers for Disease Control and Prevention, National Center for Health Statistics. Health, United States, 2013: with special feature on prescription drugs. Published May 2014. Accessed May 13, 2021. https://www.cdc.gov/nchs/data/hus/hus13.pdf
2. Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE. What is polypharmacy? A systematic review of definitions. BMC Geriatr. 2017;17(1):230. Published 2017 Oct 10. doi:10.1186/s12877-017-0621-2
3. Parulekar MS, Rogers CK. Polypharmacy and mobility. In: Cifu DX, Lew HL, Oh-Park M., eds Geriatric Rehabilitation. Elsevier; 2018. doi:10.1016/B978-0-323-54454-2.12001-1
4. Rieckert A, Trampisch US, Klaaßen-Mielke R, et al. Polypharmacy in older patients with chronic diseases: a cross-sectional analysis of factors associated with excessive polypharmacy. BMC Fam Pract. 2018;19(1):113. Published 2018 Jul 18. doi:10.1186/s12875-018-0795-5
5. Thompson CA. New medication review method cuts veterans’ Rx load, saves millions. Am J Health Syst Pharm. 2018;75(8):502-503. doi:10.2146/news180023
6. Reeve E. Deprescribing tools: a review of the types of tools available to aid deprescribing in clinical practice. J Pharm Pract Res. 2020;50(1):98-107. doi:10.1002/jppr.1626
7. Fried TR, Niehoff KM, Street RL, et al. Effect of the Tool to Reduce Inappropriate Medications on Medication Communication and Deprescribing. J Am Geriatr Soc. 2017;65(10):2265-2271. doi:10.1111/jgs.15042
8. Battar S, Dickerson KR, Sedgwick C, et al. Understanding principles of high reliability organizations through the eyes of VIONE, a clinical program to improve patient safety by deprescribing potentially inappropriate medications and reducing polypharmacy. Fed Pract. 2019;36(12):564-568.
9. Battar S, Cmelik T, Dickerson K, Scott, M. Experience better health with VIONE a safe medication deprescribing tool [Nonpublic source, not verified]
10. Ulley J, Harrop D, Ali A, et al. Desprescribing interventions and their impact on medication adherence in community-dwelling older adults with polypharmacy: a systematic review. BMC Geriatr. 2019;19(15):1-13.