User login
For MD-IQ use only
Five reasons to update your will
You have a will, so you can rest easy, right? Not necessarily. If your will is outdated, it can actually cause more harm than good. Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Here are five reasons to update your will.
Keep it current
When life changes, so should your will. Ensure that this important document matches your current wishes by reviewing it every few years.
Take a look at what has changed
Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
- Family changes. If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
- Relocating to a new state. The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
- Changes in your estate’s value. When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
- Tax law changes. Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
- You want to support a favorite cause. If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
Get the help you need
To make sure your will accomplishes all you intend, seek the help of an attorney who specializes in estate planning. Already finalized your charitable distribution to the AGA Research Foundation? Send us your letter of intent at [email protected].
You have a will, so you can rest easy, right? Not necessarily. If your will is outdated, it can actually cause more harm than good. Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Here are five reasons to update your will.
Keep it current
When life changes, so should your will. Ensure that this important document matches your current wishes by reviewing it every few years.
Take a look at what has changed
Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
- Family changes. If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
- Relocating to a new state. The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
- Changes in your estate’s value. When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
- Tax law changes. Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
- You want to support a favorite cause. If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
Get the help you need
To make sure your will accomplishes all you intend, seek the help of an attorney who specializes in estate planning. Already finalized your charitable distribution to the AGA Research Foundation? Send us your letter of intent at [email protected].
You have a will, so you can rest easy, right? Not necessarily. If your will is outdated, it can actually cause more harm than good. Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Here are five reasons to update your will.
Keep it current
When life changes, so should your will. Ensure that this important document matches your current wishes by reviewing it every few years.
Take a look at what has changed
Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
- Family changes. If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
- Relocating to a new state. The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
- Changes in your estate’s value. When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
- Tax law changes. Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
- You want to support a favorite cause. If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
Get the help you need
To make sure your will accomplishes all you intend, seek the help of an attorney who specializes in estate planning. Already finalized your charitable distribution to the AGA Research Foundation? Send us your letter of intent at [email protected].
Top cases
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community ( https://community.gastro.org ) to seek advice from colleagues about therapy and disease management options, best practices, and diagnoses. Here’s a preview of a recent popular clinical discussion:
From Brock Doubledee, DO: Xeljanz for Crohn’s
“I have a 20-year-old female with moderately active Crohn’s disease who has now failed Humira, Remicade, Entyvio and Stelara. The only option I know of for her at this time is Xeljanz, however her insurance will not approve this medication given its lack of FDA approval. I would be interested to know if anyone has any other recommended options or has had success with getting insurance approval. If you have had success I would appreciate any articles or guidance you have utilized to gain this approval.”
See how AGA members responded and join the discussion: https://community.gastro.org/posts/24445.
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community ( https://community.gastro.org ) to seek advice from colleagues about therapy and disease management options, best practices, and diagnoses. Here’s a preview of a recent popular clinical discussion:
From Brock Doubledee, DO: Xeljanz for Crohn’s
“I have a 20-year-old female with moderately active Crohn’s disease who has now failed Humira, Remicade, Entyvio and Stelara. The only option I know of for her at this time is Xeljanz, however her insurance will not approve this medication given its lack of FDA approval. I would be interested to know if anyone has any other recommended options or has had success with getting insurance approval. If you have had success I would appreciate any articles or guidance you have utilized to gain this approval.”
See how AGA members responded and join the discussion: https://community.gastro.org/posts/24445.
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community ( https://community.gastro.org ) to seek advice from colleagues about therapy and disease management options, best practices, and diagnoses. Here’s a preview of a recent popular clinical discussion:
From Brock Doubledee, DO: Xeljanz for Crohn’s
“I have a 20-year-old female with moderately active Crohn’s disease who has now failed Humira, Remicade, Entyvio and Stelara. The only option I know of for her at this time is Xeljanz, however her insurance will not approve this medication given its lack of FDA approval. I would be interested to know if anyone has any other recommended options or has had success with getting insurance approval. If you have had success I would appreciate any articles or guidance you have utilized to gain this approval.”
See how AGA members responded and join the discussion: https://community.gastro.org/posts/24445.
The first signs of elusive dysautonomia may appear on the skin
During the annual meeting of the Society for Pediatric Dermatology, Adelaide A. Hebert, MD, defined dysautonomia as an umbrella term describing conditions that result in a malfunction of the autonomic nervous system. “This encompasses both the sympathetic and the parasympathetic components of the nervous system,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “Clinical findings may be neurometabolic, developmental, and/or degenerative,” representing a “whole constellation of issues” that physicians may encounter in practice, she noted. Of particular interest is postural orthostatic tachycardia syndrome (POTS), which affects between 1 million and 3 million people in the United States. Typical symptoms include lightheadedness, fainting, and a rapid increase in heartbeat after standing up from a seated position. Other conditions associated with dysautonomia include neurocardiogenic syncope and multiple system atrophy.
Dysautonomia can impact the brain, heart, mouth, blood vessels, eyes, immune cells, and bladder, as well as the skin. Patient presentations vary with symptoms that can range from mild to debilitating. The average time from symptom onset to diagnosis of dysautonomia is 7 years. “It is very difficult to put together these mysterious symptoms that patients have unless one really thinks about dysautonomia as a possible diagnosis,” Dr. Hebert said.
One of the common symptoms that she has seen in her clinical practice is joint hypermobility. “There is a known association between dysautonomia and hypermobile-type Ehlers-Danlos syndrome (EDS), and these patients often have hyperhidrosis,” she said. “So, keep in mind that you could see hypermobility, especially in those with EDS, with associated hyperhidrosis and dysautonomia.” Two key references that she recommends to clinicians when evaluating patients with possible dysautonomia are a study on postural tachycardia in hypermobile EDS, and an article on cardiovascular autonomic dysfunction in hypermobile EDS.
The Beighton Scoring System, which measures joint mobility on a 9-point scale, involves assessment of the joint mobility of the knuckle of both pinky fingers, the base of both thumbs, the elbows, knees, and spine. An instructional video on how to perform a joint hypermobility assessment is available on the Ehler-Danlos Society website.
Literature review
In March 2021, Dr. Hebert and colleagues from other medical specialties published a summary of the literature on cutaneous manifestations in dysautonomia, with an emphasis on syndromes of orthostatic intolerance. “We had neurology, cardiology, along with dermatology involved in contributing the findings they had seen in the UTHealth McGovern Dysautonomia Center of Excellence as there was a dearth of literature that taught us about the cutaneous manifestations of orthostatic intolerance syndromes,” Dr. Hebert said.
One study included in the review showed that 23 out of 26 patients with POTS had at least one of the following cutaneous manifestations: flushing, Raynaud’s phenomenon, evanescent hyperemia, livedo reticularis, erythromelalgia, and hypo- or hyperhidrosis. “If you see a patient with any of these findings, you want to think about the possibility of dysautonomia,” she said, adding that urticaria can also be a finding.
To screen for dysautonomia, she advised, “ask patients if they have difficulty sitting or standing upright, if they have indigestion or other gastric symptoms, abnormal blood vessel functioning such as low or high blood pressure, increased or decreased sweating, changes in urinary frequency or urinary incontinence, or challenges with vision.”
If the patient answers yes to two or more of these questions, she said, consider a referral to neurology and/or cardiology or a center of excellence for further evaluation with tilt-table testing and other screening tools. She also recommended a review published in 2015 that describes the dermatological manifestations of postural tachycardia syndrome and includes illustrated cases.
One of Dr. Hebert’s future dermatology residents assembled a composite of data from the Dysautonomia Center of Excellence, and in the study, found that, compared with males, females with dysautonomia suffer more from excessive sweating, paleness of the face, pale extremities, swelling, cyanosis, cold intolerance, flushing, and hot flashes.
Dr. Hebert disclosed that she has been a consultant to and an adviser for several pharmaceutical companies.
During the annual meeting of the Society for Pediatric Dermatology, Adelaide A. Hebert, MD, defined dysautonomia as an umbrella term describing conditions that result in a malfunction of the autonomic nervous system. “This encompasses both the sympathetic and the parasympathetic components of the nervous system,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “Clinical findings may be neurometabolic, developmental, and/or degenerative,” representing a “whole constellation of issues” that physicians may encounter in practice, she noted. Of particular interest is postural orthostatic tachycardia syndrome (POTS), which affects between 1 million and 3 million people in the United States. Typical symptoms include lightheadedness, fainting, and a rapid increase in heartbeat after standing up from a seated position. Other conditions associated with dysautonomia include neurocardiogenic syncope and multiple system atrophy.
Dysautonomia can impact the brain, heart, mouth, blood vessels, eyes, immune cells, and bladder, as well as the skin. Patient presentations vary with symptoms that can range from mild to debilitating. The average time from symptom onset to diagnosis of dysautonomia is 7 years. “It is very difficult to put together these mysterious symptoms that patients have unless one really thinks about dysautonomia as a possible diagnosis,” Dr. Hebert said.
One of the common symptoms that she has seen in her clinical practice is joint hypermobility. “There is a known association between dysautonomia and hypermobile-type Ehlers-Danlos syndrome (EDS), and these patients often have hyperhidrosis,” she said. “So, keep in mind that you could see hypermobility, especially in those with EDS, with associated hyperhidrosis and dysautonomia.” Two key references that she recommends to clinicians when evaluating patients with possible dysautonomia are a study on postural tachycardia in hypermobile EDS, and an article on cardiovascular autonomic dysfunction in hypermobile EDS.
The Beighton Scoring System, which measures joint mobility on a 9-point scale, involves assessment of the joint mobility of the knuckle of both pinky fingers, the base of both thumbs, the elbows, knees, and spine. An instructional video on how to perform a joint hypermobility assessment is available on the Ehler-Danlos Society website.
Literature review
In March 2021, Dr. Hebert and colleagues from other medical specialties published a summary of the literature on cutaneous manifestations in dysautonomia, with an emphasis on syndromes of orthostatic intolerance. “We had neurology, cardiology, along with dermatology involved in contributing the findings they had seen in the UTHealth McGovern Dysautonomia Center of Excellence as there was a dearth of literature that taught us about the cutaneous manifestations of orthostatic intolerance syndromes,” Dr. Hebert said.
One study included in the review showed that 23 out of 26 patients with POTS had at least one of the following cutaneous manifestations: flushing, Raynaud’s phenomenon, evanescent hyperemia, livedo reticularis, erythromelalgia, and hypo- or hyperhidrosis. “If you see a patient with any of these findings, you want to think about the possibility of dysautonomia,” she said, adding that urticaria can also be a finding.
To screen for dysautonomia, she advised, “ask patients if they have difficulty sitting or standing upright, if they have indigestion or other gastric symptoms, abnormal blood vessel functioning such as low or high blood pressure, increased or decreased sweating, changes in urinary frequency or urinary incontinence, or challenges with vision.”
If the patient answers yes to two or more of these questions, she said, consider a referral to neurology and/or cardiology or a center of excellence for further evaluation with tilt-table testing and other screening tools. She also recommended a review published in 2015 that describes the dermatological manifestations of postural tachycardia syndrome and includes illustrated cases.
One of Dr. Hebert’s future dermatology residents assembled a composite of data from the Dysautonomia Center of Excellence, and in the study, found that, compared with males, females with dysautonomia suffer more from excessive sweating, paleness of the face, pale extremities, swelling, cyanosis, cold intolerance, flushing, and hot flashes.
Dr. Hebert disclosed that she has been a consultant to and an adviser for several pharmaceutical companies.
During the annual meeting of the Society for Pediatric Dermatology, Adelaide A. Hebert, MD, defined dysautonomia as an umbrella term describing conditions that result in a malfunction of the autonomic nervous system. “This encompasses both the sympathetic and the parasympathetic components of the nervous system,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “Clinical findings may be neurometabolic, developmental, and/or degenerative,” representing a “whole constellation of issues” that physicians may encounter in practice, she noted. Of particular interest is postural orthostatic tachycardia syndrome (POTS), which affects between 1 million and 3 million people in the United States. Typical symptoms include lightheadedness, fainting, and a rapid increase in heartbeat after standing up from a seated position. Other conditions associated with dysautonomia include neurocardiogenic syncope and multiple system atrophy.
Dysautonomia can impact the brain, heart, mouth, blood vessels, eyes, immune cells, and bladder, as well as the skin. Patient presentations vary with symptoms that can range from mild to debilitating. The average time from symptom onset to diagnosis of dysautonomia is 7 years. “It is very difficult to put together these mysterious symptoms that patients have unless one really thinks about dysautonomia as a possible diagnosis,” Dr. Hebert said.
One of the common symptoms that she has seen in her clinical practice is joint hypermobility. “There is a known association between dysautonomia and hypermobile-type Ehlers-Danlos syndrome (EDS), and these patients often have hyperhidrosis,” she said. “So, keep in mind that you could see hypermobility, especially in those with EDS, with associated hyperhidrosis and dysautonomia.” Two key references that she recommends to clinicians when evaluating patients with possible dysautonomia are a study on postural tachycardia in hypermobile EDS, and an article on cardiovascular autonomic dysfunction in hypermobile EDS.
The Beighton Scoring System, which measures joint mobility on a 9-point scale, involves assessment of the joint mobility of the knuckle of both pinky fingers, the base of both thumbs, the elbows, knees, and spine. An instructional video on how to perform a joint hypermobility assessment is available on the Ehler-Danlos Society website.
Literature review
In March 2021, Dr. Hebert and colleagues from other medical specialties published a summary of the literature on cutaneous manifestations in dysautonomia, with an emphasis on syndromes of orthostatic intolerance. “We had neurology, cardiology, along with dermatology involved in contributing the findings they had seen in the UTHealth McGovern Dysautonomia Center of Excellence as there was a dearth of literature that taught us about the cutaneous manifestations of orthostatic intolerance syndromes,” Dr. Hebert said.
One study included in the review showed that 23 out of 26 patients with POTS had at least one of the following cutaneous manifestations: flushing, Raynaud’s phenomenon, evanescent hyperemia, livedo reticularis, erythromelalgia, and hypo- or hyperhidrosis. “If you see a patient with any of these findings, you want to think about the possibility of dysautonomia,” she said, adding that urticaria can also be a finding.
To screen for dysautonomia, she advised, “ask patients if they have difficulty sitting or standing upright, if they have indigestion or other gastric symptoms, abnormal blood vessel functioning such as low or high blood pressure, increased or decreased sweating, changes in urinary frequency or urinary incontinence, or challenges with vision.”
If the patient answers yes to two or more of these questions, she said, consider a referral to neurology and/or cardiology or a center of excellence for further evaluation with tilt-table testing and other screening tools. She also recommended a review published in 2015 that describes the dermatological manifestations of postural tachycardia syndrome and includes illustrated cases.
One of Dr. Hebert’s future dermatology residents assembled a composite of data from the Dysautonomia Center of Excellence, and in the study, found that, compared with males, females with dysautonomia suffer more from excessive sweating, paleness of the face, pale extremities, swelling, cyanosis, cold intolerance, flushing, and hot flashes.
Dr. Hebert disclosed that she has been a consultant to and an adviser for several pharmaceutical companies.
FROM SPD 2021
Rising meth-related heart failure admissions a ‘crisis,’ costly for society
Rates of heart failure (HF) caused by methamphetamine abuse are climbing quickly in the western United States, at great financial and societal cost, suggests an analysis that documents the trends in California over a recent decade.
In the new study, methamphetamine-associated HF (meth-HF) admissions in the state rose by 585% between 2008 and 2018, and charges related those hospitalizations jumped 840%. Cases of HF unrelated to meth fell by 6% during the same period.
The recent explosion in meth-HF hospitalizations has also been costly for society in general, because most cases are younger adults in their most productive, prime earning years, Susan X. Zhao, MD, Santa Clara Valley Medical Center, San Jose, Calif., said in an interview.
“Over the past 11 years, especially since 2018, it has really started to take off, with a pretty dramatic rise. And it happened without much attention, because when we think about drugs, we think about acute overdose and not so much about the chronic, smoldering, long-term effects,” said Dr. Zhao, who is lead author on the study published July 13, 2021, in Circulation: Cardiovascular Quality and Outcomes.
“It’s really affecting a section of the population that is not supposed to be having heart failure problems. I think it is going to continue for the next decade until we put a stop to the parent problem, which is methamphetamine,” Dr. Zhao said. “We’re at the beginning, even though the rise has been pretty dramatic. The worst is yet to come.”
Under the radar
Methamphetamine-associated HF has been a growing problem for many years but has largely been “flying under the radar” because HF hospitalization data focus on Medicare-age patients, not the overwhelmingly younger meth-HF population, the report notes.
“We have to get this message out. Many of my patients with meth heart failure had no idea this would happen to them. They didn’t know,” Dr. Zhao said. “Once I tell them that this is what methamphetamines will do to you after years and years of use, they say they wish someone had told them.”
Dr. Zhao and colleagues looked at HF admission data collected by California’s Health and Human Services Agency to assess meth-HF trends and disease burden. They identified 1,033,076 HF hospitalizations during the decade, of which 42,565 (4.12%) were for meth-HF.
Patients hospitalized with meth-HF had a mean age of 49.6 years, compared with 72.2 for the other patients admitted with HF (P < .001). Virtually all of the patients hospitalized for meth-HF were younger than 65 years: 94.5%, compared with 30% for the other HF patients (P < .001).
Hospitalized patients with meth-HF were mostly men, their prevalence of 80% contrasting with 52.4% for patients with non–meth-related HF (P < .001).
Rates of hospitalization for meth-HF steadily increased during the study period. The age-adjusted rate of meth-HF hospitalization per 100,000 rose from 4.1 in 2008 to 28.1 in 2018. The rate of hospitalization for HF unrelated to meth actually declined, going from 342.3 in 2008 to 321.6 in 2018.
Charges for hospitalizations related to meth-HF shot up more than eight times, from $41.5 million in 2008 to $390.2 million in 2018. In contrast, charges for other HF hospitalizations rose by only 82%, from $3.5 billion to $6.3 billion.
Multiple layers of prevention
Dr. Zhao proposed ways that clinicians can communicate with their patients who are using or considering to use meth. “There are multiple layers of prevention. For people who are thinking of using meth, they need to get the message that something really bad can happen to them years down the road. They’re not going to die from it overnight, but it will damage the heart slowly,” she said.
The next layer of prevention can potentially help meth users who have not yet developed heart problems, Dr. Zhao said. “This would be the time to say, ‘you’re so lucky, your heart is still good. It’s time to stop because people like you, a few years from now are going to die prematurely from a very horrible, very suffering kind of death’.”
Importantly, in meth users who have already developed HF, even then it may not be too late to reverse the cardiomyopathy and symptoms. For up to a third of people with established meth-HF, “if they stop using meth, if they take good cardiac medications, and if the heart failure is in an early enough course, their heart can entirely revert to normal,” Dr. Zhao said, citing an earlier work from her and her colleagues.
Currently, methamphetamine abuse has taken especially strong root in rural areas in California and the Midwest. But Dr. Zhao predicts it will soon become prevalent throughout the United States.
Spotlight on an ‘epidemic’
The rapid growth of the methamphetamine “epidemic” has been well-documented in the United States and around the world, observed an accompanying editorial from Pavan Reddy, MD, Icahn School of Medicine at Mount Sinai Morningside, New York, and Uri Elkayam, MD, University of Southern California, Los Angeles.
They contend that more attention has been given to opioid overdose deaths; meth abuse does not seem to command the same attention, likely because meth is not as strongly associated with acute overdose.
But meth, wrote Dr. Reddy and Dr. Elkayam, “is a different drug with its own M.O., equally dangerous and costly to society but more insidious in nature, its effects potentially causing decades of mental and physical debilitation before ending in premature death.”
The current study “has turned a spotlight on a public health crisis that has grown unfettered for over 2 decades,” and is a call for the “medical community to recognize and manage cases of meth-HF with a comprehensive approach that addresses both mental and physical illness,” they concluded. “Only then can we hope to properly help these patients and with that, reduce the socioeconomic burden of meth-HF.”
A quietly building crisis
The sharp rise in meth-HF hospitalizations is an expected reflection of the methamphetamine crisis, which has been quietly building over the last few years, addiction psychiatrist Corneliu N. Stanciu, MD, Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said in an interview.
“This new version of methamphetamines looks like ice and is more potent and toxic than former versions traditionally made in home-built labs,” he said. Lately the vast majority of methamphetamines in the United States have come from Mexico, are less expensive with higher purity, “and can be manufactured in greater quantities.”
Some patients with opioid use disorder (OUD) also inject methamphetamines, which can make OUD treatment clinics good places to screen for meth abuse and educate about its cardiovascular implications, Dr. Stanciu said.
“Just as addiction treatment centers present an opportunity to implement cardiac screening and referrals,” he said, “cardiology visits and hospitalizations such as those for meth-HF also present a golden opportunity for involvement of substance use disorder interventions and referrals to get patients into treatment and prevent further damage through ongoing use.”
Dr. Zhao, Dr. Reddy, Dr. Eklayam, and Dr. Stanciu report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rates of heart failure (HF) caused by methamphetamine abuse are climbing quickly in the western United States, at great financial and societal cost, suggests an analysis that documents the trends in California over a recent decade.
In the new study, methamphetamine-associated HF (meth-HF) admissions in the state rose by 585% between 2008 and 2018, and charges related those hospitalizations jumped 840%. Cases of HF unrelated to meth fell by 6% during the same period.
The recent explosion in meth-HF hospitalizations has also been costly for society in general, because most cases are younger adults in their most productive, prime earning years, Susan X. Zhao, MD, Santa Clara Valley Medical Center, San Jose, Calif., said in an interview.
“Over the past 11 years, especially since 2018, it has really started to take off, with a pretty dramatic rise. And it happened without much attention, because when we think about drugs, we think about acute overdose and not so much about the chronic, smoldering, long-term effects,” said Dr. Zhao, who is lead author on the study published July 13, 2021, in Circulation: Cardiovascular Quality and Outcomes.
“It’s really affecting a section of the population that is not supposed to be having heart failure problems. I think it is going to continue for the next decade until we put a stop to the parent problem, which is methamphetamine,” Dr. Zhao said. “We’re at the beginning, even though the rise has been pretty dramatic. The worst is yet to come.”
Under the radar
Methamphetamine-associated HF has been a growing problem for many years but has largely been “flying under the radar” because HF hospitalization data focus on Medicare-age patients, not the overwhelmingly younger meth-HF population, the report notes.
“We have to get this message out. Many of my patients with meth heart failure had no idea this would happen to them. They didn’t know,” Dr. Zhao said. “Once I tell them that this is what methamphetamines will do to you after years and years of use, they say they wish someone had told them.”
Dr. Zhao and colleagues looked at HF admission data collected by California’s Health and Human Services Agency to assess meth-HF trends and disease burden. They identified 1,033,076 HF hospitalizations during the decade, of which 42,565 (4.12%) were for meth-HF.
Patients hospitalized with meth-HF had a mean age of 49.6 years, compared with 72.2 for the other patients admitted with HF (P < .001). Virtually all of the patients hospitalized for meth-HF were younger than 65 years: 94.5%, compared with 30% for the other HF patients (P < .001).
Hospitalized patients with meth-HF were mostly men, their prevalence of 80% contrasting with 52.4% for patients with non–meth-related HF (P < .001).
Rates of hospitalization for meth-HF steadily increased during the study period. The age-adjusted rate of meth-HF hospitalization per 100,000 rose from 4.1 in 2008 to 28.1 in 2018. The rate of hospitalization for HF unrelated to meth actually declined, going from 342.3 in 2008 to 321.6 in 2018.
Charges for hospitalizations related to meth-HF shot up more than eight times, from $41.5 million in 2008 to $390.2 million in 2018. In contrast, charges for other HF hospitalizations rose by only 82%, from $3.5 billion to $6.3 billion.
Multiple layers of prevention
Dr. Zhao proposed ways that clinicians can communicate with their patients who are using or considering to use meth. “There are multiple layers of prevention. For people who are thinking of using meth, they need to get the message that something really bad can happen to them years down the road. They’re not going to die from it overnight, but it will damage the heart slowly,” she said.
The next layer of prevention can potentially help meth users who have not yet developed heart problems, Dr. Zhao said. “This would be the time to say, ‘you’re so lucky, your heart is still good. It’s time to stop because people like you, a few years from now are going to die prematurely from a very horrible, very suffering kind of death’.”
Importantly, in meth users who have already developed HF, even then it may not be too late to reverse the cardiomyopathy and symptoms. For up to a third of people with established meth-HF, “if they stop using meth, if they take good cardiac medications, and if the heart failure is in an early enough course, their heart can entirely revert to normal,” Dr. Zhao said, citing an earlier work from her and her colleagues.
Currently, methamphetamine abuse has taken especially strong root in rural areas in California and the Midwest. But Dr. Zhao predicts it will soon become prevalent throughout the United States.
Spotlight on an ‘epidemic’
The rapid growth of the methamphetamine “epidemic” has been well-documented in the United States and around the world, observed an accompanying editorial from Pavan Reddy, MD, Icahn School of Medicine at Mount Sinai Morningside, New York, and Uri Elkayam, MD, University of Southern California, Los Angeles.
They contend that more attention has been given to opioid overdose deaths; meth abuse does not seem to command the same attention, likely because meth is not as strongly associated with acute overdose.
But meth, wrote Dr. Reddy and Dr. Elkayam, “is a different drug with its own M.O., equally dangerous and costly to society but more insidious in nature, its effects potentially causing decades of mental and physical debilitation before ending in premature death.”
The current study “has turned a spotlight on a public health crisis that has grown unfettered for over 2 decades,” and is a call for the “medical community to recognize and manage cases of meth-HF with a comprehensive approach that addresses both mental and physical illness,” they concluded. “Only then can we hope to properly help these patients and with that, reduce the socioeconomic burden of meth-HF.”
A quietly building crisis
The sharp rise in meth-HF hospitalizations is an expected reflection of the methamphetamine crisis, which has been quietly building over the last few years, addiction psychiatrist Corneliu N. Stanciu, MD, Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said in an interview.
“This new version of methamphetamines looks like ice and is more potent and toxic than former versions traditionally made in home-built labs,” he said. Lately the vast majority of methamphetamines in the United States have come from Mexico, are less expensive with higher purity, “and can be manufactured in greater quantities.”
Some patients with opioid use disorder (OUD) also inject methamphetamines, which can make OUD treatment clinics good places to screen for meth abuse and educate about its cardiovascular implications, Dr. Stanciu said.
“Just as addiction treatment centers present an opportunity to implement cardiac screening and referrals,” he said, “cardiology visits and hospitalizations such as those for meth-HF also present a golden opportunity for involvement of substance use disorder interventions and referrals to get patients into treatment and prevent further damage through ongoing use.”
Dr. Zhao, Dr. Reddy, Dr. Eklayam, and Dr. Stanciu report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rates of heart failure (HF) caused by methamphetamine abuse are climbing quickly in the western United States, at great financial and societal cost, suggests an analysis that documents the trends in California over a recent decade.
In the new study, methamphetamine-associated HF (meth-HF) admissions in the state rose by 585% between 2008 and 2018, and charges related those hospitalizations jumped 840%. Cases of HF unrelated to meth fell by 6% during the same period.
The recent explosion in meth-HF hospitalizations has also been costly for society in general, because most cases are younger adults in their most productive, prime earning years, Susan X. Zhao, MD, Santa Clara Valley Medical Center, San Jose, Calif., said in an interview.
“Over the past 11 years, especially since 2018, it has really started to take off, with a pretty dramatic rise. And it happened without much attention, because when we think about drugs, we think about acute overdose and not so much about the chronic, smoldering, long-term effects,” said Dr. Zhao, who is lead author on the study published July 13, 2021, in Circulation: Cardiovascular Quality and Outcomes.
“It’s really affecting a section of the population that is not supposed to be having heart failure problems. I think it is going to continue for the next decade until we put a stop to the parent problem, which is methamphetamine,” Dr. Zhao said. “We’re at the beginning, even though the rise has been pretty dramatic. The worst is yet to come.”
Under the radar
Methamphetamine-associated HF has been a growing problem for many years but has largely been “flying under the radar” because HF hospitalization data focus on Medicare-age patients, not the overwhelmingly younger meth-HF population, the report notes.
“We have to get this message out. Many of my patients with meth heart failure had no idea this would happen to them. They didn’t know,” Dr. Zhao said. “Once I tell them that this is what methamphetamines will do to you after years and years of use, they say they wish someone had told them.”
Dr. Zhao and colleagues looked at HF admission data collected by California’s Health and Human Services Agency to assess meth-HF trends and disease burden. They identified 1,033,076 HF hospitalizations during the decade, of which 42,565 (4.12%) were for meth-HF.
Patients hospitalized with meth-HF had a mean age of 49.6 years, compared with 72.2 for the other patients admitted with HF (P < .001). Virtually all of the patients hospitalized for meth-HF were younger than 65 years: 94.5%, compared with 30% for the other HF patients (P < .001).
Hospitalized patients with meth-HF were mostly men, their prevalence of 80% contrasting with 52.4% for patients with non–meth-related HF (P < .001).
Rates of hospitalization for meth-HF steadily increased during the study period. The age-adjusted rate of meth-HF hospitalization per 100,000 rose from 4.1 in 2008 to 28.1 in 2018. The rate of hospitalization for HF unrelated to meth actually declined, going from 342.3 in 2008 to 321.6 in 2018.
Charges for hospitalizations related to meth-HF shot up more than eight times, from $41.5 million in 2008 to $390.2 million in 2018. In contrast, charges for other HF hospitalizations rose by only 82%, from $3.5 billion to $6.3 billion.
Multiple layers of prevention
Dr. Zhao proposed ways that clinicians can communicate with their patients who are using or considering to use meth. “There are multiple layers of prevention. For people who are thinking of using meth, they need to get the message that something really bad can happen to them years down the road. They’re not going to die from it overnight, but it will damage the heart slowly,” she said.
The next layer of prevention can potentially help meth users who have not yet developed heart problems, Dr. Zhao said. “This would be the time to say, ‘you’re so lucky, your heart is still good. It’s time to stop because people like you, a few years from now are going to die prematurely from a very horrible, very suffering kind of death’.”
Importantly, in meth users who have already developed HF, even then it may not be too late to reverse the cardiomyopathy and symptoms. For up to a third of people with established meth-HF, “if they stop using meth, if they take good cardiac medications, and if the heart failure is in an early enough course, their heart can entirely revert to normal,” Dr. Zhao said, citing an earlier work from her and her colleagues.
Currently, methamphetamine abuse has taken especially strong root in rural areas in California and the Midwest. But Dr. Zhao predicts it will soon become prevalent throughout the United States.
Spotlight on an ‘epidemic’
The rapid growth of the methamphetamine “epidemic” has been well-documented in the United States and around the world, observed an accompanying editorial from Pavan Reddy, MD, Icahn School of Medicine at Mount Sinai Morningside, New York, and Uri Elkayam, MD, University of Southern California, Los Angeles.
They contend that more attention has been given to opioid overdose deaths; meth abuse does not seem to command the same attention, likely because meth is not as strongly associated with acute overdose.
But meth, wrote Dr. Reddy and Dr. Elkayam, “is a different drug with its own M.O., equally dangerous and costly to society but more insidious in nature, its effects potentially causing decades of mental and physical debilitation before ending in premature death.”
The current study “has turned a spotlight on a public health crisis that has grown unfettered for over 2 decades,” and is a call for the “medical community to recognize and manage cases of meth-HF with a comprehensive approach that addresses both mental and physical illness,” they concluded. “Only then can we hope to properly help these patients and with that, reduce the socioeconomic burden of meth-HF.”
A quietly building crisis
The sharp rise in meth-HF hospitalizations is an expected reflection of the methamphetamine crisis, which has been quietly building over the last few years, addiction psychiatrist Corneliu N. Stanciu, MD, Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said in an interview.
“This new version of methamphetamines looks like ice and is more potent and toxic than former versions traditionally made in home-built labs,” he said. Lately the vast majority of methamphetamines in the United States have come from Mexico, are less expensive with higher purity, “and can be manufactured in greater quantities.”
Some patients with opioid use disorder (OUD) also inject methamphetamines, which can make OUD treatment clinics good places to screen for meth abuse and educate about its cardiovascular implications, Dr. Stanciu said.
“Just as addiction treatment centers present an opportunity to implement cardiac screening and referrals,” he said, “cardiology visits and hospitalizations such as those for meth-HF also present a golden opportunity for involvement of substance use disorder interventions and referrals to get patients into treatment and prevent further damage through ongoing use.”
Dr. Zhao, Dr. Reddy, Dr. Eklayam, and Dr. Stanciu report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PHM virtual conference promises practical pearls, plus Dr. Fauci
The Pediatric Hospital Medicine annual conference, though virtual in 2021, promises to retain its role as the premier educational event for pediatric hospitalists and other clinicians involved in treating pediatric patients.
The “can’t-miss” session, on August 5, at 6:30 p.m. ET, is a one-on-one discussion between Anthony S. Fauci, MD, and Lee Savio Beers, MD, president of the American Academic of Pediatrics, according to members of the meeting planning committee.
In addition to the conversation between Dr. Beers and Dr. Fauci, this year’s meeting offers a mix of workshops with pointers and pearls to improve practice, keynote and plenary sessions to inform and inspire, and abstract presentations of new research. Three members of the PHM Planning Committee shared their insights on the hot topics, advice for new clinicians, and tips for making the most of this year’s meeting.
Workshops worth watching
“The keynote plenary sessions by Julie Silver, MD, on ‘Accelerating Patient Care and Healthcare Workforce Diversity and Inclusion,’ and by Ilan Alhadeff, MD, on ‘Leading through Adversity’ should inspire even the least enthusiastic among us,” Mirna Giordano, MD, FHM, of Columbia University Medical Center, New York, said in an interview. A talk by Nathan T. Chomilo, MD, “will likely prompt reflection on how George Floyd’s death changed us, and how we practice medicine forever.” In addition, “PHM Stories are not to be missed, they are voices that speak loud and move mountains.”
The PHM Stories are concise, narrative talks with minimal use of slides; each PHM Stories session includes three distinct talks and a 15-minute question and answer session. PHM Stories sessions are scheduled for each day of the conference, and topics include “Practicing Medicine While Human: The Secrets Physicians Keep,” by Uchenna Ewulonu, MD; “Finding the Power of the Imposter: How I Learned to Be Exactly the Color I Am, Everywhere I Go,” by Alexandra Coria, MD; and “Purple Butterflies: A Reflection on Why I’m a Pediatric Hospitalist,” by Joanne Mendoza, MD.
“The PHM community has been through a lot in the aftermath of the pandemic,” said Dr. Giordano. “The mini-plenary session on the mental health needs of our patients, and clinical quick-hit sessions on verbal deescalation of the agitated patients and cardiac effects of COVID-19 will likely be not only very popular, but also useful in clinical endeavors. The workshop on how to navigate the adult issues in hospitalized patients will provide the Med-Peds pearls we all wish we heard earlier.”
Although a 75-minute workshop session may seem long, “the workshop choices will offer something for everyone’s taste: education, research, clinical topics, diversity, and advocacy,” Dr. Giordano said. “I suggest that attendees check in advance which sessions will be available after the meeting, so that they prioritize highly interactive sessions like workshops, and that they experience, even if virtual, small group/room gatherings and networking.” There will be time for fun, too, she emphasized, with social sessions “that we hope will break the screen monotony and bring smiles to everyone’s faces.”
For younger clinicians relatively new to practice, Dr. Giordano recommended several workshops for a wealth of advice and guidance, including “New Kids on the Block: Thriving in your First Faculty Position,” “Channeling Your Inner Coach: Techniques to Enhance Clinical Teaching & Feedback,” “Palliative Care Pearls for the Pediatric Hospitalist,” “Perioperative Medicine for Medically Complex Children: Case Studies in Programmatic Approaches,” “The Bare Necessities: Social Determinant of Health Screening for the Hospitalist,” and “Mentorship, Autonomy, and Supervising a PHM Fellow.”
Classic topics and new concepts
“We are so excited to be able to offer a full spectrum of offerings at this year’s virtual meeting,” Yemisi Jones, MD, FHM, of Cincinnati Children’s Hospital, said in an interview. “We are covering some classic topics that we can’t do without at PHM, such as clinical updates in the management of sick and well newborns; workshops on best practices for educators; as well as the latest in PHM scholarship.” Sessions include “timely topics such as equity for women in medicine with one of our plenary speakers, Julie Silver, MD, and new febrile infant guidelines,” she added.
In particular, the COVID-19 and mental health session will help address clinicians’ evolving understanding of the COVID-19 pandemic and its effects on hospitalized children, said Dr. Jones. “Attendees can expect practical, timely updates on the current state of the science and ways to improve their practice to provide the best care for our patients.”
Attendees will be able to maximize the virtual conference format by accessing archived recordings, including clinical quick hits, mini-plenaries, and PHM Stories, which can be viewed during the scheduled meeting time or after, Dr. Jones said. “Workshops and abstract presentations will involve real-time interaction with presenters, so would be highest yield to attend during the live meeting. We also encourage all participants to take full advantage of the platform and the various networking opportunities to engage with others in our PHM community.”
For residents and new fellows, Dr. Jones advised making the workshop, “A Whole New World: Tips and Tools to Soar Into Your First Year of Fellowship,” a priority. “For early-career faculty, the ‘New Kids on the Block: Thriving in your First Faculty Position workshop will be a valuable resource.”
Make the meeting content a priority
This year’s conference has an exceptional slate of plenary speakers, Michelle Marks, DO, SFHM, of the Cleveland Clinic said in an interview. In addition to the much-anticipated session on vaccinations, school guidelines, and other topics with Dr. Fauci and Dr. Beers, the sessions on leading through adversity and workforce diversity and inclusion are “important topics to the PHM community and to our greater communities as a whole.”
Dr. Marks also highlighted the value of the COVID-19 and mental health session, as the long-term impact of COVID-19 on mental health of children and adults continues to grab headlines. “From this session specifically, I hope the attendees will gain awareness of the special mental health needs for child during a global disaster like a pandemic, which can be generalized to other situations and gain skills and resources to help meet and advocate for children’s mental health needs.”
For clinicians attending the virtual conference, “The most important strategy is to schedule time off of clinical work for the virtual meeting if you can so you can focus on the content,” said Dr. Marks. “For the longer sessions, it would be very important to block time in your day to fully attend the session, attend in a private space if possible since there will be breakouts with discussion, have your camera on, and engage with the workshop group as much as possible. The virtual format can be challenging because of all the external distractions, so intentional focus is necessary,” to get the most out of the experience.
The mini-plenary session on “The New AAP Clinical Practice Guideline on the Evaluation and Management of Febrile Infants 8-60 Days Old,” is an important session for all attendees, Dr. Marks said. She also recommended the Clinical Quick Hits sessions for anyone seeking “a diverse array of practical knowledge which can be easily applied to everyday practice.” The Clinical Quick Hits are designed as 35-minute, rapid-fire presentations focused on clinical knowledge. Each of these presentations will focus on the latest updates or evolutions in clinical practice in one area. Some key topics include counseling parents when a child has an abnormal exam finding, assessing pelvic pain in adolescent girls, and preventing venous thromboembolism in the inpatient setting.
“I would also recommend that younger clinicians take in at least one or two workshops or sessions on nonclinical topics to see the breath of content at the meeting and to develop a niche interest for themselves outside of clinical work,” Dr. Marks noted.
Nonclinical sessions at PHM 2021 include workshops on a pilot for a comprehensive LGBTQ+ curriculum, using media tools for public health messaging, and practicing health literacy.
To register for the Pediatric Hospital Medicine 2021 virtual conference, visit https://apaevents.regfox.com/phm21-virtual-conference.
Dr. Giordano, Dr. Jones, and Dr. Marks are members of the PHM conference planning committee and had no relevant financial conflicts to disclose.
The Pediatric Hospital Medicine annual conference, though virtual in 2021, promises to retain its role as the premier educational event for pediatric hospitalists and other clinicians involved in treating pediatric patients.
The “can’t-miss” session, on August 5, at 6:30 p.m. ET, is a one-on-one discussion between Anthony S. Fauci, MD, and Lee Savio Beers, MD, president of the American Academic of Pediatrics, according to members of the meeting planning committee.
In addition to the conversation between Dr. Beers and Dr. Fauci, this year’s meeting offers a mix of workshops with pointers and pearls to improve practice, keynote and plenary sessions to inform and inspire, and abstract presentations of new research. Three members of the PHM Planning Committee shared their insights on the hot topics, advice for new clinicians, and tips for making the most of this year’s meeting.
Workshops worth watching
“The keynote plenary sessions by Julie Silver, MD, on ‘Accelerating Patient Care and Healthcare Workforce Diversity and Inclusion,’ and by Ilan Alhadeff, MD, on ‘Leading through Adversity’ should inspire even the least enthusiastic among us,” Mirna Giordano, MD, FHM, of Columbia University Medical Center, New York, said in an interview. A talk by Nathan T. Chomilo, MD, “will likely prompt reflection on how George Floyd’s death changed us, and how we practice medicine forever.” In addition, “PHM Stories are not to be missed, they are voices that speak loud and move mountains.”
The PHM Stories are concise, narrative talks with minimal use of slides; each PHM Stories session includes three distinct talks and a 15-minute question and answer session. PHM Stories sessions are scheduled for each day of the conference, and topics include “Practicing Medicine While Human: The Secrets Physicians Keep,” by Uchenna Ewulonu, MD; “Finding the Power of the Imposter: How I Learned to Be Exactly the Color I Am, Everywhere I Go,” by Alexandra Coria, MD; and “Purple Butterflies: A Reflection on Why I’m a Pediatric Hospitalist,” by Joanne Mendoza, MD.
“The PHM community has been through a lot in the aftermath of the pandemic,” said Dr. Giordano. “The mini-plenary session on the mental health needs of our patients, and clinical quick-hit sessions on verbal deescalation of the agitated patients and cardiac effects of COVID-19 will likely be not only very popular, but also useful in clinical endeavors. The workshop on how to navigate the adult issues in hospitalized patients will provide the Med-Peds pearls we all wish we heard earlier.”
Although a 75-minute workshop session may seem long, “the workshop choices will offer something for everyone’s taste: education, research, clinical topics, diversity, and advocacy,” Dr. Giordano said. “I suggest that attendees check in advance which sessions will be available after the meeting, so that they prioritize highly interactive sessions like workshops, and that they experience, even if virtual, small group/room gatherings and networking.” There will be time for fun, too, she emphasized, with social sessions “that we hope will break the screen monotony and bring smiles to everyone’s faces.”
For younger clinicians relatively new to practice, Dr. Giordano recommended several workshops for a wealth of advice and guidance, including “New Kids on the Block: Thriving in your First Faculty Position,” “Channeling Your Inner Coach: Techniques to Enhance Clinical Teaching & Feedback,” “Palliative Care Pearls for the Pediatric Hospitalist,” “Perioperative Medicine for Medically Complex Children: Case Studies in Programmatic Approaches,” “The Bare Necessities: Social Determinant of Health Screening for the Hospitalist,” and “Mentorship, Autonomy, and Supervising a PHM Fellow.”
Classic topics and new concepts
“We are so excited to be able to offer a full spectrum of offerings at this year’s virtual meeting,” Yemisi Jones, MD, FHM, of Cincinnati Children’s Hospital, said in an interview. “We are covering some classic topics that we can’t do without at PHM, such as clinical updates in the management of sick and well newborns; workshops on best practices for educators; as well as the latest in PHM scholarship.” Sessions include “timely topics such as equity for women in medicine with one of our plenary speakers, Julie Silver, MD, and new febrile infant guidelines,” she added.
In particular, the COVID-19 and mental health session will help address clinicians’ evolving understanding of the COVID-19 pandemic and its effects on hospitalized children, said Dr. Jones. “Attendees can expect practical, timely updates on the current state of the science and ways to improve their practice to provide the best care for our patients.”
Attendees will be able to maximize the virtual conference format by accessing archived recordings, including clinical quick hits, mini-plenaries, and PHM Stories, which can be viewed during the scheduled meeting time or after, Dr. Jones said. “Workshops and abstract presentations will involve real-time interaction with presenters, so would be highest yield to attend during the live meeting. We also encourage all participants to take full advantage of the platform and the various networking opportunities to engage with others in our PHM community.”
For residents and new fellows, Dr. Jones advised making the workshop, “A Whole New World: Tips and Tools to Soar Into Your First Year of Fellowship,” a priority. “For early-career faculty, the ‘New Kids on the Block: Thriving in your First Faculty Position workshop will be a valuable resource.”
Make the meeting content a priority
This year’s conference has an exceptional slate of plenary speakers, Michelle Marks, DO, SFHM, of the Cleveland Clinic said in an interview. In addition to the much-anticipated session on vaccinations, school guidelines, and other topics with Dr. Fauci and Dr. Beers, the sessions on leading through adversity and workforce diversity and inclusion are “important topics to the PHM community and to our greater communities as a whole.”
Dr. Marks also highlighted the value of the COVID-19 and mental health session, as the long-term impact of COVID-19 on mental health of children and adults continues to grab headlines. “From this session specifically, I hope the attendees will gain awareness of the special mental health needs for child during a global disaster like a pandemic, which can be generalized to other situations and gain skills and resources to help meet and advocate for children’s mental health needs.”
For clinicians attending the virtual conference, “The most important strategy is to schedule time off of clinical work for the virtual meeting if you can so you can focus on the content,” said Dr. Marks. “For the longer sessions, it would be very important to block time in your day to fully attend the session, attend in a private space if possible since there will be breakouts with discussion, have your camera on, and engage with the workshop group as much as possible. The virtual format can be challenging because of all the external distractions, so intentional focus is necessary,” to get the most out of the experience.
The mini-plenary session on “The New AAP Clinical Practice Guideline on the Evaluation and Management of Febrile Infants 8-60 Days Old,” is an important session for all attendees, Dr. Marks said. She also recommended the Clinical Quick Hits sessions for anyone seeking “a diverse array of practical knowledge which can be easily applied to everyday practice.” The Clinical Quick Hits are designed as 35-minute, rapid-fire presentations focused on clinical knowledge. Each of these presentations will focus on the latest updates or evolutions in clinical practice in one area. Some key topics include counseling parents when a child has an abnormal exam finding, assessing pelvic pain in adolescent girls, and preventing venous thromboembolism in the inpatient setting.
“I would also recommend that younger clinicians take in at least one or two workshops or sessions on nonclinical topics to see the breath of content at the meeting and to develop a niche interest for themselves outside of clinical work,” Dr. Marks noted.
Nonclinical sessions at PHM 2021 include workshops on a pilot for a comprehensive LGBTQ+ curriculum, using media tools for public health messaging, and practicing health literacy.
To register for the Pediatric Hospital Medicine 2021 virtual conference, visit https://apaevents.regfox.com/phm21-virtual-conference.
Dr. Giordano, Dr. Jones, and Dr. Marks are members of the PHM conference planning committee and had no relevant financial conflicts to disclose.
The Pediatric Hospital Medicine annual conference, though virtual in 2021, promises to retain its role as the premier educational event for pediatric hospitalists and other clinicians involved in treating pediatric patients.
The “can’t-miss” session, on August 5, at 6:30 p.m. ET, is a one-on-one discussion between Anthony S. Fauci, MD, and Lee Savio Beers, MD, president of the American Academic of Pediatrics, according to members of the meeting planning committee.
In addition to the conversation between Dr. Beers and Dr. Fauci, this year’s meeting offers a mix of workshops with pointers and pearls to improve practice, keynote and plenary sessions to inform and inspire, and abstract presentations of new research. Three members of the PHM Planning Committee shared their insights on the hot topics, advice for new clinicians, and tips for making the most of this year’s meeting.
Workshops worth watching
“The keynote plenary sessions by Julie Silver, MD, on ‘Accelerating Patient Care and Healthcare Workforce Diversity and Inclusion,’ and by Ilan Alhadeff, MD, on ‘Leading through Adversity’ should inspire even the least enthusiastic among us,” Mirna Giordano, MD, FHM, of Columbia University Medical Center, New York, said in an interview. A talk by Nathan T. Chomilo, MD, “will likely prompt reflection on how George Floyd’s death changed us, and how we practice medicine forever.” In addition, “PHM Stories are not to be missed, they are voices that speak loud and move mountains.”
The PHM Stories are concise, narrative talks with minimal use of slides; each PHM Stories session includes three distinct talks and a 15-minute question and answer session. PHM Stories sessions are scheduled for each day of the conference, and topics include “Practicing Medicine While Human: The Secrets Physicians Keep,” by Uchenna Ewulonu, MD; “Finding the Power of the Imposter: How I Learned to Be Exactly the Color I Am, Everywhere I Go,” by Alexandra Coria, MD; and “Purple Butterflies: A Reflection on Why I’m a Pediatric Hospitalist,” by Joanne Mendoza, MD.
“The PHM community has been through a lot in the aftermath of the pandemic,” said Dr. Giordano. “The mini-plenary session on the mental health needs of our patients, and clinical quick-hit sessions on verbal deescalation of the agitated patients and cardiac effects of COVID-19 will likely be not only very popular, but also useful in clinical endeavors. The workshop on how to navigate the adult issues in hospitalized patients will provide the Med-Peds pearls we all wish we heard earlier.”
Although a 75-minute workshop session may seem long, “the workshop choices will offer something for everyone’s taste: education, research, clinical topics, diversity, and advocacy,” Dr. Giordano said. “I suggest that attendees check in advance which sessions will be available after the meeting, so that they prioritize highly interactive sessions like workshops, and that they experience, even if virtual, small group/room gatherings and networking.” There will be time for fun, too, she emphasized, with social sessions “that we hope will break the screen monotony and bring smiles to everyone’s faces.”
For younger clinicians relatively new to practice, Dr. Giordano recommended several workshops for a wealth of advice and guidance, including “New Kids on the Block: Thriving in your First Faculty Position,” “Channeling Your Inner Coach: Techniques to Enhance Clinical Teaching & Feedback,” “Palliative Care Pearls for the Pediatric Hospitalist,” “Perioperative Medicine for Medically Complex Children: Case Studies in Programmatic Approaches,” “The Bare Necessities: Social Determinant of Health Screening for the Hospitalist,” and “Mentorship, Autonomy, and Supervising a PHM Fellow.”
Classic topics and new concepts
“We are so excited to be able to offer a full spectrum of offerings at this year’s virtual meeting,” Yemisi Jones, MD, FHM, of Cincinnati Children’s Hospital, said in an interview. “We are covering some classic topics that we can’t do without at PHM, such as clinical updates in the management of sick and well newborns; workshops on best practices for educators; as well as the latest in PHM scholarship.” Sessions include “timely topics such as equity for women in medicine with one of our plenary speakers, Julie Silver, MD, and new febrile infant guidelines,” she added.
In particular, the COVID-19 and mental health session will help address clinicians’ evolving understanding of the COVID-19 pandemic and its effects on hospitalized children, said Dr. Jones. “Attendees can expect practical, timely updates on the current state of the science and ways to improve their practice to provide the best care for our patients.”
Attendees will be able to maximize the virtual conference format by accessing archived recordings, including clinical quick hits, mini-plenaries, and PHM Stories, which can be viewed during the scheduled meeting time or after, Dr. Jones said. “Workshops and abstract presentations will involve real-time interaction with presenters, so would be highest yield to attend during the live meeting. We also encourage all participants to take full advantage of the platform and the various networking opportunities to engage with others in our PHM community.”
For residents and new fellows, Dr. Jones advised making the workshop, “A Whole New World: Tips and Tools to Soar Into Your First Year of Fellowship,” a priority. “For early-career faculty, the ‘New Kids on the Block: Thriving in your First Faculty Position workshop will be a valuable resource.”
Make the meeting content a priority
This year’s conference has an exceptional slate of plenary speakers, Michelle Marks, DO, SFHM, of the Cleveland Clinic said in an interview. In addition to the much-anticipated session on vaccinations, school guidelines, and other topics with Dr. Fauci and Dr. Beers, the sessions on leading through adversity and workforce diversity and inclusion are “important topics to the PHM community and to our greater communities as a whole.”
Dr. Marks also highlighted the value of the COVID-19 and mental health session, as the long-term impact of COVID-19 on mental health of children and adults continues to grab headlines. “From this session specifically, I hope the attendees will gain awareness of the special mental health needs for child during a global disaster like a pandemic, which can be generalized to other situations and gain skills and resources to help meet and advocate for children’s mental health needs.”
For clinicians attending the virtual conference, “The most important strategy is to schedule time off of clinical work for the virtual meeting if you can so you can focus on the content,” said Dr. Marks. “For the longer sessions, it would be very important to block time in your day to fully attend the session, attend in a private space if possible since there will be breakouts with discussion, have your camera on, and engage with the workshop group as much as possible. The virtual format can be challenging because of all the external distractions, so intentional focus is necessary,” to get the most out of the experience.
The mini-plenary session on “The New AAP Clinical Practice Guideline on the Evaluation and Management of Febrile Infants 8-60 Days Old,” is an important session for all attendees, Dr. Marks said. She also recommended the Clinical Quick Hits sessions for anyone seeking “a diverse array of practical knowledge which can be easily applied to everyday practice.” The Clinical Quick Hits are designed as 35-minute, rapid-fire presentations focused on clinical knowledge. Each of these presentations will focus on the latest updates or evolutions in clinical practice in one area. Some key topics include counseling parents when a child has an abnormal exam finding, assessing pelvic pain in adolescent girls, and preventing venous thromboembolism in the inpatient setting.
“I would also recommend that younger clinicians take in at least one or two workshops or sessions on nonclinical topics to see the breath of content at the meeting and to develop a niche interest for themselves outside of clinical work,” Dr. Marks noted.
Nonclinical sessions at PHM 2021 include workshops on a pilot for a comprehensive LGBTQ+ curriculum, using media tools for public health messaging, and practicing health literacy.
To register for the Pediatric Hospital Medicine 2021 virtual conference, visit https://apaevents.regfox.com/phm21-virtual-conference.
Dr. Giordano, Dr. Jones, and Dr. Marks are members of the PHM conference planning committee and had no relevant financial conflicts to disclose.
CDC revamps STI treatment guidelines
On July 22, the Centers for Disease Control and Prevention released updated sexually transmitted infection treatment guidelines to reflect current screening, testing, and treatment recommendations. The guidelines were last updated in 2015.
The new recommendations come at a pivotal moment in the field’s history, Kimberly Workowski, MD, a medical officer at the CDC’s Division of STD Prevention, told this news organization in an email. “The COVID-19 pandemic has caused decreased clinic capacity and drug and diagnostic test kit shortages,” she says. Many of these shortages have been resolved, she added, and it is important that health care professionals use the most current evidence-based recommendations for screening and management of STIs.
Updates to these guidelines were necessary to reflect “continued advances in research in the prevention of STIs, new interventions in terms of STI prevention, and thirdly, changing epidemiology,” Jeffrey Klausner, MD, MPH, an STI specialist with the Keck School of Medicine at the University of Southern California, Los Angeles, said in an interview. “There’s been increased concern about antimicrobial resistance, and that’s really driven some of the key changes in these new STI treatment guidelines.”
Notable updates to the guidelines include the following:
- Updated treatment recommendations for gonorrhea, chlamydia, , and
- Two-step testing for diagnosing genital virus
- Expanded risk factors for testing in pregnant women
- Information on FDA-cleared rectal and oral tests to diagnose chlamydia and gonorrhea
- A recommendation that universal screening be conducted at least once in a lifetime for adults aged 18 years and older
Dr. Workowski emphasized updates to gonorrhea treatment that built on the recommendation published in December 2020 in Morbidity and Mortality Weekly Report. The CDC now recommends that gonorrhea be treated with a single 500-mg injection of ceftriaxone, and if chlamydial infection is not ruled out, treating with a regimen of 100 mg of oral doxycycline taken twice daily for 7 days. Other gonorrhea treatment recommendations include retesting patients 3 months after treatment and that a test of cure be conducted for people with pharyngeal gonorrhea 1 to 2 weeks after treatment, using either culture or nucleic-acid amplification tests.
“Effectively treating gonorrhea remains a public health priority,” Dr. Workowski said. “Gonorrhea can rapidly develop antibiotic resistance and is the second most commonly reported bacterial STI in the U.S., increasing 56% from 2015 to 2019.”
The updates to syphilis screening for pregnant women are also important, added Dr. Klausner. “We’ve seen a dramatic and shameful rise in congenital syphilis,” he said. In addition to screening all pregnant women at the first prenatal visit, the CDC recommends retesting for syphilis at 28 weeks’ gestation and at delivery if the mother lives in an area where the prevalence of syphilis is high or if she is at risk of acquiring syphilis during pregnancy. An expectant mother is at higher risk if she has multiple sex partners, has an STI during pregnancy, has a partner with an STI, has a new sex partner, or misuses drugs, the recommendations state.
Dr. Klausner also noted that the updates provide more robust guidelines for treating transgender individuals and incarcerated people.
The treatment guidelines are available online along with a wall chart and a pocket guide that summarizes these updates. The mobile app with the 2015 guidelines will be retired at the end of July 2021, Dr. Workowski said. An app with these updated treatment recommendations is in development and will be available later this year.
A version of this article first appeared on Medscape.com.
On July 22, the Centers for Disease Control and Prevention released updated sexually transmitted infection treatment guidelines to reflect current screening, testing, and treatment recommendations. The guidelines were last updated in 2015.
The new recommendations come at a pivotal moment in the field’s history, Kimberly Workowski, MD, a medical officer at the CDC’s Division of STD Prevention, told this news organization in an email. “The COVID-19 pandemic has caused decreased clinic capacity and drug and diagnostic test kit shortages,” she says. Many of these shortages have been resolved, she added, and it is important that health care professionals use the most current evidence-based recommendations for screening and management of STIs.
Updates to these guidelines were necessary to reflect “continued advances in research in the prevention of STIs, new interventions in terms of STI prevention, and thirdly, changing epidemiology,” Jeffrey Klausner, MD, MPH, an STI specialist with the Keck School of Medicine at the University of Southern California, Los Angeles, said in an interview. “There’s been increased concern about antimicrobial resistance, and that’s really driven some of the key changes in these new STI treatment guidelines.”
Notable updates to the guidelines include the following:
- Updated treatment recommendations for gonorrhea, chlamydia, , and
- Two-step testing for diagnosing genital virus
- Expanded risk factors for testing in pregnant women
- Information on FDA-cleared rectal and oral tests to diagnose chlamydia and gonorrhea
- A recommendation that universal screening be conducted at least once in a lifetime for adults aged 18 years and older
Dr. Workowski emphasized updates to gonorrhea treatment that built on the recommendation published in December 2020 in Morbidity and Mortality Weekly Report. The CDC now recommends that gonorrhea be treated with a single 500-mg injection of ceftriaxone, and if chlamydial infection is not ruled out, treating with a regimen of 100 mg of oral doxycycline taken twice daily for 7 days. Other gonorrhea treatment recommendations include retesting patients 3 months after treatment and that a test of cure be conducted for people with pharyngeal gonorrhea 1 to 2 weeks after treatment, using either culture or nucleic-acid amplification tests.
“Effectively treating gonorrhea remains a public health priority,” Dr. Workowski said. “Gonorrhea can rapidly develop antibiotic resistance and is the second most commonly reported bacterial STI in the U.S., increasing 56% from 2015 to 2019.”
The updates to syphilis screening for pregnant women are also important, added Dr. Klausner. “We’ve seen a dramatic and shameful rise in congenital syphilis,” he said. In addition to screening all pregnant women at the first prenatal visit, the CDC recommends retesting for syphilis at 28 weeks’ gestation and at delivery if the mother lives in an area where the prevalence of syphilis is high or if she is at risk of acquiring syphilis during pregnancy. An expectant mother is at higher risk if she has multiple sex partners, has an STI during pregnancy, has a partner with an STI, has a new sex partner, or misuses drugs, the recommendations state.
Dr. Klausner also noted that the updates provide more robust guidelines for treating transgender individuals and incarcerated people.
The treatment guidelines are available online along with a wall chart and a pocket guide that summarizes these updates. The mobile app with the 2015 guidelines will be retired at the end of July 2021, Dr. Workowski said. An app with these updated treatment recommendations is in development and will be available later this year.
A version of this article first appeared on Medscape.com.
On July 22, the Centers for Disease Control and Prevention released updated sexually transmitted infection treatment guidelines to reflect current screening, testing, and treatment recommendations. The guidelines were last updated in 2015.
The new recommendations come at a pivotal moment in the field’s history, Kimberly Workowski, MD, a medical officer at the CDC’s Division of STD Prevention, told this news organization in an email. “The COVID-19 pandemic has caused decreased clinic capacity and drug and diagnostic test kit shortages,” she says. Many of these shortages have been resolved, she added, and it is important that health care professionals use the most current evidence-based recommendations for screening and management of STIs.
Updates to these guidelines were necessary to reflect “continued advances in research in the prevention of STIs, new interventions in terms of STI prevention, and thirdly, changing epidemiology,” Jeffrey Klausner, MD, MPH, an STI specialist with the Keck School of Medicine at the University of Southern California, Los Angeles, said in an interview. “There’s been increased concern about antimicrobial resistance, and that’s really driven some of the key changes in these new STI treatment guidelines.”
Notable updates to the guidelines include the following:
- Updated treatment recommendations for gonorrhea, chlamydia, , and
- Two-step testing for diagnosing genital virus
- Expanded risk factors for testing in pregnant women
- Information on FDA-cleared rectal and oral tests to diagnose chlamydia and gonorrhea
- A recommendation that universal screening be conducted at least once in a lifetime for adults aged 18 years and older
Dr. Workowski emphasized updates to gonorrhea treatment that built on the recommendation published in December 2020 in Morbidity and Mortality Weekly Report. The CDC now recommends that gonorrhea be treated with a single 500-mg injection of ceftriaxone, and if chlamydial infection is not ruled out, treating with a regimen of 100 mg of oral doxycycline taken twice daily for 7 days. Other gonorrhea treatment recommendations include retesting patients 3 months after treatment and that a test of cure be conducted for people with pharyngeal gonorrhea 1 to 2 weeks after treatment, using either culture or nucleic-acid amplification tests.
“Effectively treating gonorrhea remains a public health priority,” Dr. Workowski said. “Gonorrhea can rapidly develop antibiotic resistance and is the second most commonly reported bacterial STI in the U.S., increasing 56% from 2015 to 2019.”
The updates to syphilis screening for pregnant women are also important, added Dr. Klausner. “We’ve seen a dramatic and shameful rise in congenital syphilis,” he said. In addition to screening all pregnant women at the first prenatal visit, the CDC recommends retesting for syphilis at 28 weeks’ gestation and at delivery if the mother lives in an area where the prevalence of syphilis is high or if she is at risk of acquiring syphilis during pregnancy. An expectant mother is at higher risk if she has multiple sex partners, has an STI during pregnancy, has a partner with an STI, has a new sex partner, or misuses drugs, the recommendations state.
Dr. Klausner also noted that the updates provide more robust guidelines for treating transgender individuals and incarcerated people.
The treatment guidelines are available online along with a wall chart and a pocket guide that summarizes these updates. The mobile app with the 2015 guidelines will be retired at the end of July 2021, Dr. Workowski said. An app with these updated treatment recommendations is in development and will be available later this year.
A version of this article first appeared on Medscape.com.
HIV-associated cryptococcal meningitis: Single-dose regimen found non-inferior
A single high dose of the antifungal agent liposomal amphotericin B (L-AmB; AmBisome, Gilead Sciences), with a background regimen of flucytosine and fluconazole, is non-inferior and significantly safer in preventing mortality in HIV-associated cryptococcal meningitis than a conventional seven-day regimen that is the current standard of care, according to a new study.
“The results of this phase 3 [AMBITION-cm] trial make it clear that this approach is just as good as the current World Health Organization-recommended first-line treatment in preventing death,” first author David S. Lawrence, MD, AMBITION study lead clinician, of the London School of Hygiene and Tropical Medicine, United Kingdom, said in an interview.
“The fact that this was the largest ever trial [on HIV-associated cryptococcal meningitis] conducted to date… gives us a high level of confidence in these results,” he said of the study, which was presented at the virtual annual meeting of the International AIDS Society.
“We believe that this should become the WHO-recommended first-line regimen,” he emphasized.
In commenting on the study, Meg Doherty, MD, PhD, director of global HIV, hepatitis, and STI programs at WHO, agreed that a shorter regimen could be vital, particularly in settings with limited resources.
“The results from the AMBITION trial are important for low- and middle-income countries, where the cost and complexity of implementing the current standard seven-day course of L-ambisome or cryptococcal meningitis treatment can put this out of reach for many,” she told this news organization.
“Simplification that maintains the highest quality of care is an important component of the public health approach for HIV treatment and care,” she added.
Dr. Doherty could not comment on any possible changes to WHO recommendations, which are formulated by an independent guideline development group, but a spokesperson said that “WHO is preparing an updated review of the evidence for treating cryptococcal disease as a first step towards updating guidance.”
Conventional treatment toxicities
Cryptococcal meningitis is a leading cause of death in HIV, and the conventional treatment of amphotericin B deoxycholate, though less expensive than L-AmB, is more toxic, causing anemia, renal impairment, and electrolyte abnormalities, Dr. Lawrence explained
Having previously shown a single 10 mg/kg dose of L-AmB to be as effective as the longer regimen of 14 daily doses in terms of clearing cryptococcus from the cerebrospinal fluid, Dr. Lawrence and his colleagues conducted the phase 3 AMBITION-Cm trial to evaluate the effect on mortality, enrolling 844 patients in Botswana, Malawi, South Africa, Uganda, and Zimbabwe who were HIV-positive and had a first episode of cryptococcal meningitis.
Participants in the study were randomized to treatment either with single, high-dose L-AmB (10 mg/kg), combined with 14 days of flucytosine 100 mg/kg/day and fluconazole 1,200 mg/day or to a control group receiving 7 daily doses of AmB deoxycholate (1 mg/kg) plus 7 days of flucytosine 100 mg/kg/day, followed by 7 days of fluconazole 1,200 mg/day.
All patients were also provided with consolidation therapy of fluconazole 800 mg/day for eight weeks. Of the patients, 60.2% were male, their median age was 37, and their median CD4 count was 27 cells/mm3.
For the primary endpoint in the intention-to-treat analysis of 814 patients, the 10-week mortality rate in the single-dose L-AmB group was 24.82% (101 of 407) and 28.75% (117 of 407) in the control arm, for a difference (-3.93%) that was well within the pre-specified non-inferiority margin of 10%.
As expected, the safety measures were significantly improved with the single-dose of L-AmB: Rates of grade 3 or 4 adverse events within the initial 21 days of treatment in the single-dose L-AmB group were 50% versus 62.3% in the control group, and severe anemia occurred in just 13% of single-dose L-AmB participants, compared with 41% in the AmB deoxycholate control arm (both P < .001), Dr. Lawrence reported.
Furthermore, the average decline in hemoglobin over the first week was 0.3 g/dL in the single-dose L-AmB arm and 1.9 g/dL in the control arm, resulting in the need for more blood transfusions in the control arm (P < .001).
The impact on kidney function was also worse in the higher dose arm, with an average increase in creatinine over the first week of 20.2% in the L-AmB group versus 49.7% in the control group, while hypokalemia and thrombophlebitis were also more common with the higher dose group, Dr. Lawrence noted.
In the adjusted analysis, the single-dose L-AmB measures were in fact superior after adjusting for factors including research site, age, sex, baseline Glasgow Coma Scale, CD4 count, CSF cryptococcal colony-forming units/mL, antiretroviral therapy status, hemoglobin, and CSF opening pressure.
Mortality rate still high – but significantly reduced
The mortality rate of about 25% in the study after the treatment is still significantly higher than typically seen in high-income countries such as the United States, where HIV-associated cryptococcal meningitis is less common and associated with a mortality of roughly 10-15%, Dr. Lawrence noted.
The rate is nevertheless among the lowest mortality rates ever reported within a clinical trial conducted in resource-limited settings, he explained.
“These results are a step in the right direction and a significant improvement on the rates of 40% to 45% reported with two-week L-AmB-based regimens in African settings,” Dr. Lawrence underscored.
Higher cost — but potentially more cost-effective
With a higher cost than AmB deoxycholate, L-AmB’s utilization in resource-limited settings has been a challenge: A single vial of L-AmB ranges from $80 to $200, according to some reports, and while 14-day dosing requires as many as 42 vials of L-AmB, even a 7-day regimen still requires 21 vials.
In comparison, the single-dose L-AmB regimen only requires an average of 10 to 11 vials per patient, but the regimen’s higher safety could translate to far greater cost savings, Dr. Lawrence explained.
“While the AmBisome regimen is technically more expensive in terms of drugs, we expect it to be cost-effective or possibly cost-saving when taking into account that there is less toxicity, fewer blood tests, less transfusions, etc., and possibly shorter duration of hospital admission,” he said.
Cost, supply controversy: ‘Black fungus’-related demand
The drug’s cost — as well as supply issues — have meanwhile become even more of a problem as L-AmB has unexpectedly also become urgently needed in the treatment of mucormycosis in India and Nepal, where the otherwise rare fungal disease, commonly known as “black fungus,” has been increasingly affecting COVID-19 patients and survivors.
Gilead had previously announced in 2018 its intention to make L-AmB more widely available at a price of $16.25 per vial, but “implementation of this has been slow,” Dr. Lawrence said.
As a result, Gilead is facing heightened pressure to implement the lower prices – and also improve substantial supply issues, with Médecins Sans Frontières (Doctors Without Borders) and dozens of other global organizations issuing an open letter to Gilead and partner Viatris in June calling for immediate action to implement the lower price and improve supply of L-AmB.
In a company statement, Gilead responded, detailing its “commit[ment] to the non-profit pricing for the treatment of cryptococcal meningitis” and to efforts to improve the public health crisis in India.
For their part, Dr. Lawrence and his colleagues are working on producing more research on the issue.
“We hope that the conclusive results of the AMBITION trial will give a much needed push to implement this program,” he said.
“We are also currently completing the cost-effectiveness analysis of the study, which we hope will provide additional evidence to support widespread implementation of this regimen and highlight further the urgent need to broaden access to AmBisome and flucytosine,” he said.
The trial was supported by a grant through the European Developing Countries Clinical Trials Partnership (EDCTP), the Swedish International Development Cooperation Agency (SIDA) (TRIA2015-1092), and the Wellcome Trust / Medical Research Council (UK)/UKAID Joint Global Health Trials (MR/P006922/1. The AmBisome was donated by Gilead Sciences. Dr. Lawrence had no disclosures to report.
A single high dose of the antifungal agent liposomal amphotericin B (L-AmB; AmBisome, Gilead Sciences), with a background regimen of flucytosine and fluconazole, is non-inferior and significantly safer in preventing mortality in HIV-associated cryptococcal meningitis than a conventional seven-day regimen that is the current standard of care, according to a new study.
“The results of this phase 3 [AMBITION-cm] trial make it clear that this approach is just as good as the current World Health Organization-recommended first-line treatment in preventing death,” first author David S. Lawrence, MD, AMBITION study lead clinician, of the London School of Hygiene and Tropical Medicine, United Kingdom, said in an interview.
“The fact that this was the largest ever trial [on HIV-associated cryptococcal meningitis] conducted to date… gives us a high level of confidence in these results,” he said of the study, which was presented at the virtual annual meeting of the International AIDS Society.
“We believe that this should become the WHO-recommended first-line regimen,” he emphasized.
In commenting on the study, Meg Doherty, MD, PhD, director of global HIV, hepatitis, and STI programs at WHO, agreed that a shorter regimen could be vital, particularly in settings with limited resources.
“The results from the AMBITION trial are important for low- and middle-income countries, where the cost and complexity of implementing the current standard seven-day course of L-ambisome or cryptococcal meningitis treatment can put this out of reach for many,” she told this news organization.
“Simplification that maintains the highest quality of care is an important component of the public health approach for HIV treatment and care,” she added.
Dr. Doherty could not comment on any possible changes to WHO recommendations, which are formulated by an independent guideline development group, but a spokesperson said that “WHO is preparing an updated review of the evidence for treating cryptococcal disease as a first step towards updating guidance.”
Conventional treatment toxicities
Cryptococcal meningitis is a leading cause of death in HIV, and the conventional treatment of amphotericin B deoxycholate, though less expensive than L-AmB, is more toxic, causing anemia, renal impairment, and electrolyte abnormalities, Dr. Lawrence explained
Having previously shown a single 10 mg/kg dose of L-AmB to be as effective as the longer regimen of 14 daily doses in terms of clearing cryptococcus from the cerebrospinal fluid, Dr. Lawrence and his colleagues conducted the phase 3 AMBITION-Cm trial to evaluate the effect on mortality, enrolling 844 patients in Botswana, Malawi, South Africa, Uganda, and Zimbabwe who were HIV-positive and had a first episode of cryptococcal meningitis.
Participants in the study were randomized to treatment either with single, high-dose L-AmB (10 mg/kg), combined with 14 days of flucytosine 100 mg/kg/day and fluconazole 1,200 mg/day or to a control group receiving 7 daily doses of AmB deoxycholate (1 mg/kg) plus 7 days of flucytosine 100 mg/kg/day, followed by 7 days of fluconazole 1,200 mg/day.
All patients were also provided with consolidation therapy of fluconazole 800 mg/day for eight weeks. Of the patients, 60.2% were male, their median age was 37, and their median CD4 count was 27 cells/mm3.
For the primary endpoint in the intention-to-treat analysis of 814 patients, the 10-week mortality rate in the single-dose L-AmB group was 24.82% (101 of 407) and 28.75% (117 of 407) in the control arm, for a difference (-3.93%) that was well within the pre-specified non-inferiority margin of 10%.
As expected, the safety measures were significantly improved with the single-dose of L-AmB: Rates of grade 3 or 4 adverse events within the initial 21 days of treatment in the single-dose L-AmB group were 50% versus 62.3% in the control group, and severe anemia occurred in just 13% of single-dose L-AmB participants, compared with 41% in the AmB deoxycholate control arm (both P < .001), Dr. Lawrence reported.
Furthermore, the average decline in hemoglobin over the first week was 0.3 g/dL in the single-dose L-AmB arm and 1.9 g/dL in the control arm, resulting in the need for more blood transfusions in the control arm (P < .001).
The impact on kidney function was also worse in the higher dose arm, with an average increase in creatinine over the first week of 20.2% in the L-AmB group versus 49.7% in the control group, while hypokalemia and thrombophlebitis were also more common with the higher dose group, Dr. Lawrence noted.
In the adjusted analysis, the single-dose L-AmB measures were in fact superior after adjusting for factors including research site, age, sex, baseline Glasgow Coma Scale, CD4 count, CSF cryptococcal colony-forming units/mL, antiretroviral therapy status, hemoglobin, and CSF opening pressure.
Mortality rate still high – but significantly reduced
The mortality rate of about 25% in the study after the treatment is still significantly higher than typically seen in high-income countries such as the United States, where HIV-associated cryptococcal meningitis is less common and associated with a mortality of roughly 10-15%, Dr. Lawrence noted.
The rate is nevertheless among the lowest mortality rates ever reported within a clinical trial conducted in resource-limited settings, he explained.
“These results are a step in the right direction and a significant improvement on the rates of 40% to 45% reported with two-week L-AmB-based regimens in African settings,” Dr. Lawrence underscored.
Higher cost — but potentially more cost-effective
With a higher cost than AmB deoxycholate, L-AmB’s utilization in resource-limited settings has been a challenge: A single vial of L-AmB ranges from $80 to $200, according to some reports, and while 14-day dosing requires as many as 42 vials of L-AmB, even a 7-day regimen still requires 21 vials.
In comparison, the single-dose L-AmB regimen only requires an average of 10 to 11 vials per patient, but the regimen’s higher safety could translate to far greater cost savings, Dr. Lawrence explained.
“While the AmBisome regimen is technically more expensive in terms of drugs, we expect it to be cost-effective or possibly cost-saving when taking into account that there is less toxicity, fewer blood tests, less transfusions, etc., and possibly shorter duration of hospital admission,” he said.
Cost, supply controversy: ‘Black fungus’-related demand
The drug’s cost — as well as supply issues — have meanwhile become even more of a problem as L-AmB has unexpectedly also become urgently needed in the treatment of mucormycosis in India and Nepal, where the otherwise rare fungal disease, commonly known as “black fungus,” has been increasingly affecting COVID-19 patients and survivors.
Gilead had previously announced in 2018 its intention to make L-AmB more widely available at a price of $16.25 per vial, but “implementation of this has been slow,” Dr. Lawrence said.
As a result, Gilead is facing heightened pressure to implement the lower prices – and also improve substantial supply issues, with Médecins Sans Frontières (Doctors Without Borders) and dozens of other global organizations issuing an open letter to Gilead and partner Viatris in June calling for immediate action to implement the lower price and improve supply of L-AmB.
In a company statement, Gilead responded, detailing its “commit[ment] to the non-profit pricing for the treatment of cryptococcal meningitis” and to efforts to improve the public health crisis in India.
For their part, Dr. Lawrence and his colleagues are working on producing more research on the issue.
“We hope that the conclusive results of the AMBITION trial will give a much needed push to implement this program,” he said.
“We are also currently completing the cost-effectiveness analysis of the study, which we hope will provide additional evidence to support widespread implementation of this regimen and highlight further the urgent need to broaden access to AmBisome and flucytosine,” he said.
The trial was supported by a grant through the European Developing Countries Clinical Trials Partnership (EDCTP), the Swedish International Development Cooperation Agency (SIDA) (TRIA2015-1092), and the Wellcome Trust / Medical Research Council (UK)/UKAID Joint Global Health Trials (MR/P006922/1. The AmBisome was donated by Gilead Sciences. Dr. Lawrence had no disclosures to report.
A single high dose of the antifungal agent liposomal amphotericin B (L-AmB; AmBisome, Gilead Sciences), with a background regimen of flucytosine and fluconazole, is non-inferior and significantly safer in preventing mortality in HIV-associated cryptococcal meningitis than a conventional seven-day regimen that is the current standard of care, according to a new study.
“The results of this phase 3 [AMBITION-cm] trial make it clear that this approach is just as good as the current World Health Organization-recommended first-line treatment in preventing death,” first author David S. Lawrence, MD, AMBITION study lead clinician, of the London School of Hygiene and Tropical Medicine, United Kingdom, said in an interview.
“The fact that this was the largest ever trial [on HIV-associated cryptococcal meningitis] conducted to date… gives us a high level of confidence in these results,” he said of the study, which was presented at the virtual annual meeting of the International AIDS Society.
“We believe that this should become the WHO-recommended first-line regimen,” he emphasized.
In commenting on the study, Meg Doherty, MD, PhD, director of global HIV, hepatitis, and STI programs at WHO, agreed that a shorter regimen could be vital, particularly in settings with limited resources.
“The results from the AMBITION trial are important for low- and middle-income countries, where the cost and complexity of implementing the current standard seven-day course of L-ambisome or cryptococcal meningitis treatment can put this out of reach for many,” she told this news organization.
“Simplification that maintains the highest quality of care is an important component of the public health approach for HIV treatment and care,” she added.
Dr. Doherty could not comment on any possible changes to WHO recommendations, which are formulated by an independent guideline development group, but a spokesperson said that “WHO is preparing an updated review of the evidence for treating cryptococcal disease as a first step towards updating guidance.”
Conventional treatment toxicities
Cryptococcal meningitis is a leading cause of death in HIV, and the conventional treatment of amphotericin B deoxycholate, though less expensive than L-AmB, is more toxic, causing anemia, renal impairment, and electrolyte abnormalities, Dr. Lawrence explained
Having previously shown a single 10 mg/kg dose of L-AmB to be as effective as the longer regimen of 14 daily doses in terms of clearing cryptococcus from the cerebrospinal fluid, Dr. Lawrence and his colleagues conducted the phase 3 AMBITION-Cm trial to evaluate the effect on mortality, enrolling 844 patients in Botswana, Malawi, South Africa, Uganda, and Zimbabwe who were HIV-positive and had a first episode of cryptococcal meningitis.
Participants in the study were randomized to treatment either with single, high-dose L-AmB (10 mg/kg), combined with 14 days of flucytosine 100 mg/kg/day and fluconazole 1,200 mg/day or to a control group receiving 7 daily doses of AmB deoxycholate (1 mg/kg) plus 7 days of flucytosine 100 mg/kg/day, followed by 7 days of fluconazole 1,200 mg/day.
All patients were also provided with consolidation therapy of fluconazole 800 mg/day for eight weeks. Of the patients, 60.2% were male, their median age was 37, and their median CD4 count was 27 cells/mm3.
For the primary endpoint in the intention-to-treat analysis of 814 patients, the 10-week mortality rate in the single-dose L-AmB group was 24.82% (101 of 407) and 28.75% (117 of 407) in the control arm, for a difference (-3.93%) that was well within the pre-specified non-inferiority margin of 10%.
As expected, the safety measures were significantly improved with the single-dose of L-AmB: Rates of grade 3 or 4 adverse events within the initial 21 days of treatment in the single-dose L-AmB group were 50% versus 62.3% in the control group, and severe anemia occurred in just 13% of single-dose L-AmB participants, compared with 41% in the AmB deoxycholate control arm (both P < .001), Dr. Lawrence reported.
Furthermore, the average decline in hemoglobin over the first week was 0.3 g/dL in the single-dose L-AmB arm and 1.9 g/dL in the control arm, resulting in the need for more blood transfusions in the control arm (P < .001).
The impact on kidney function was also worse in the higher dose arm, with an average increase in creatinine over the first week of 20.2% in the L-AmB group versus 49.7% in the control group, while hypokalemia and thrombophlebitis were also more common with the higher dose group, Dr. Lawrence noted.
In the adjusted analysis, the single-dose L-AmB measures were in fact superior after adjusting for factors including research site, age, sex, baseline Glasgow Coma Scale, CD4 count, CSF cryptococcal colony-forming units/mL, antiretroviral therapy status, hemoglobin, and CSF opening pressure.
Mortality rate still high – but significantly reduced
The mortality rate of about 25% in the study after the treatment is still significantly higher than typically seen in high-income countries such as the United States, where HIV-associated cryptococcal meningitis is less common and associated with a mortality of roughly 10-15%, Dr. Lawrence noted.
The rate is nevertheless among the lowest mortality rates ever reported within a clinical trial conducted in resource-limited settings, he explained.
“These results are a step in the right direction and a significant improvement on the rates of 40% to 45% reported with two-week L-AmB-based regimens in African settings,” Dr. Lawrence underscored.
Higher cost — but potentially more cost-effective
With a higher cost than AmB deoxycholate, L-AmB’s utilization in resource-limited settings has been a challenge: A single vial of L-AmB ranges from $80 to $200, according to some reports, and while 14-day dosing requires as many as 42 vials of L-AmB, even a 7-day regimen still requires 21 vials.
In comparison, the single-dose L-AmB regimen only requires an average of 10 to 11 vials per patient, but the regimen’s higher safety could translate to far greater cost savings, Dr. Lawrence explained.
“While the AmBisome regimen is technically more expensive in terms of drugs, we expect it to be cost-effective or possibly cost-saving when taking into account that there is less toxicity, fewer blood tests, less transfusions, etc., and possibly shorter duration of hospital admission,” he said.
Cost, supply controversy: ‘Black fungus’-related demand
The drug’s cost — as well as supply issues — have meanwhile become even more of a problem as L-AmB has unexpectedly also become urgently needed in the treatment of mucormycosis in India and Nepal, where the otherwise rare fungal disease, commonly known as “black fungus,” has been increasingly affecting COVID-19 patients and survivors.
Gilead had previously announced in 2018 its intention to make L-AmB more widely available at a price of $16.25 per vial, but “implementation of this has been slow,” Dr. Lawrence said.
As a result, Gilead is facing heightened pressure to implement the lower prices – and also improve substantial supply issues, with Médecins Sans Frontières (Doctors Without Borders) and dozens of other global organizations issuing an open letter to Gilead and partner Viatris in June calling for immediate action to implement the lower price and improve supply of L-AmB.
In a company statement, Gilead responded, detailing its “commit[ment] to the non-profit pricing for the treatment of cryptococcal meningitis” and to efforts to improve the public health crisis in India.
For their part, Dr. Lawrence and his colleagues are working on producing more research on the issue.
“We hope that the conclusive results of the AMBITION trial will give a much needed push to implement this program,” he said.
“We are also currently completing the cost-effectiveness analysis of the study, which we hope will provide additional evidence to support widespread implementation of this regimen and highlight further the urgent need to broaden access to AmBisome and flucytosine,” he said.
The trial was supported by a grant through the European Developing Countries Clinical Trials Partnership (EDCTP), the Swedish International Development Cooperation Agency (SIDA) (TRIA2015-1092), and the Wellcome Trust / Medical Research Council (UK)/UKAID Joint Global Health Trials (MR/P006922/1. The AmBisome was donated by Gilead Sciences. Dr. Lawrence had no disclosures to report.
REPORTING FROM IAS 2021
FDA warns of potential mechanical concerns with MAGEC devices
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
‘Gold cards’ allow Texas docs to skip prior authorizations
The law was passed in June and will take effect in September. It excuses physicians from having to obtain prior authorization if, during the previous 6 months, 90% of their treatments met medical necessity criteria by the health insurer. Through this law, doctors in the state will spend less time getting approvals for treatments for their patients.
Automatic approval of authorizations for treatments – or what the Texas Medical Association (TMA) calls a “gold card” – “allows patients to get the care they need in a more timely fashion,” says Debra Patt, MD, an Austin, Tex.–based oncologist and former chair of the council on legislation for the TMA.
Eighty-seven percent of Texas physicians reported a “drastic increase over the past five years in the burden of prior authorization on their patients and their practices,” per a 2020 survey by the TMA. Nearly half (48%) of Texas physicians have hired staff whose work focuses on processing requests for prior authorization, according to the survey.
Jack Resneck Jr., MD, a San Francisco–based dermatologist and president-elect of the American Medical Association (AMA), said other states have investigated ways to ease the impact of prior authorizations on physicians, but no other state has passed such a law.
Administrative burdens plague physicians around the country. The Medscape Physician Compensation Report 2021 found that physicians spend on average 15.6 hours per week on paperwork and administrative duties.
Better outcomes, less anxiety for patients
Dr. Patt, who testified in support of the law’s passage in the Texas legislature, says automatic approval of authorizations “is better for patients because it reduces their anxiety about whether they’re able to get the treatments they need now, and they will have better outcomes if they’re able to receive more timely care.”
Recently, a chemotherapy treatment Dr. Patt prescribed for one of her patients was not authorized by an insurer. The result is “a lot of anxiety and potentially health problems” for the patient, said Dr. Patt.
She expects that automatic approval for treatments will be based on prescribing patterns during the preceding 6 months. “It means that when I order a test today, the [health insurer] looks back at my record 6 months previously,” she said. Still, Dr. Patt awaits guidance from the Texas Department of Insurance, which regulates health insurers in the state, regarding the law.
Dr. Resneck said the pharmacy counter is where most patients encounter prior authorization delays. “That’s when the pharmacist looks at them and says, ‘Actually, this isn’t covered by your health insurer’s formulary,’ or it isn’t covered fully on their formulary.”
One of Dr. Resneck’s patients had a life-altering case of eczema that lasted many years. Because of the condition, the patient couldn’t work or maintain meaningful bonds with family members. A biologic treatment transformed his patient’s life. The patient was able to return to work and to re-engage with family, said Dr. Resneck. But a year after his patient started the treatment, the health insurer wouldn’t authorize the treatment because the patient wasn’t experiencing the same symptoms.
The patient didn’t have the same symptoms because the biologic treatment worked, said Dr. Resneck.
Kristine Grow, a spokesperson for America’s Health Insurance Plans, a national association for health insurers, said, “The use of prior authorization is relatively small – typically, less than 15% – and can help ensure safer opioid prescribing, help prevent dangerous drug interactions, and help protect patients from unnecessary exposure to potentially harmful radiation for inappropriate diagnostic imaging. Numerous studies show that Americans frequently receive inappropriate care, and 25% of unnecessary treatments are associated with complications or adverse events.”
Medical management tools, such as prior authorization, are an “an important way” to deliver “safe, high-quality care” to patients, she added.
State and federal efforts to curb prior authorization
In addition to efforts to curb prior authorization in other states, the AMA supports the Improving Seniors’ Timely Access to Care Act (HR 3173). The act includes a provision related to “gold-carding,” said Robert Mills, an AMA spokesperson.
The bill establishes requirements and standards for prior authorization processes related to Medicare Advantage (MA) plans. The requirements and standards for MA plans include the following:
- Establishing an electronic prior authorization program that meets specific standards, such as the ability to provide real-time decisions in response to requests for items and services that are routinely approved.
- Publishing on an annual basis specific prior authorization information, including the percentage of requests approved and the average response time.
- Meeting standards set by the Centers for Medicare & Medicaid Services related to the quality and timeliness of prior authorization determinations.
The act was introduced to the U.S. House of Representatives in May, after which it was referred to two committees for consideration.
A version of this article first appeared on Medscape.com.
The law was passed in June and will take effect in September. It excuses physicians from having to obtain prior authorization if, during the previous 6 months, 90% of their treatments met medical necessity criteria by the health insurer. Through this law, doctors in the state will spend less time getting approvals for treatments for their patients.
Automatic approval of authorizations for treatments – or what the Texas Medical Association (TMA) calls a “gold card” – “allows patients to get the care they need in a more timely fashion,” says Debra Patt, MD, an Austin, Tex.–based oncologist and former chair of the council on legislation for the TMA.
Eighty-seven percent of Texas physicians reported a “drastic increase over the past five years in the burden of prior authorization on their patients and their practices,” per a 2020 survey by the TMA. Nearly half (48%) of Texas physicians have hired staff whose work focuses on processing requests for prior authorization, according to the survey.
Jack Resneck Jr., MD, a San Francisco–based dermatologist and president-elect of the American Medical Association (AMA), said other states have investigated ways to ease the impact of prior authorizations on physicians, but no other state has passed such a law.
Administrative burdens plague physicians around the country. The Medscape Physician Compensation Report 2021 found that physicians spend on average 15.6 hours per week on paperwork and administrative duties.
Better outcomes, less anxiety for patients
Dr. Patt, who testified in support of the law’s passage in the Texas legislature, says automatic approval of authorizations “is better for patients because it reduces their anxiety about whether they’re able to get the treatments they need now, and they will have better outcomes if they’re able to receive more timely care.”
Recently, a chemotherapy treatment Dr. Patt prescribed for one of her patients was not authorized by an insurer. The result is “a lot of anxiety and potentially health problems” for the patient, said Dr. Patt.
She expects that automatic approval for treatments will be based on prescribing patterns during the preceding 6 months. “It means that when I order a test today, the [health insurer] looks back at my record 6 months previously,” she said. Still, Dr. Patt awaits guidance from the Texas Department of Insurance, which regulates health insurers in the state, regarding the law.
Dr. Resneck said the pharmacy counter is where most patients encounter prior authorization delays. “That’s when the pharmacist looks at them and says, ‘Actually, this isn’t covered by your health insurer’s formulary,’ or it isn’t covered fully on their formulary.”
One of Dr. Resneck’s patients had a life-altering case of eczema that lasted many years. Because of the condition, the patient couldn’t work or maintain meaningful bonds with family members. A biologic treatment transformed his patient’s life. The patient was able to return to work and to re-engage with family, said Dr. Resneck. But a year after his patient started the treatment, the health insurer wouldn’t authorize the treatment because the patient wasn’t experiencing the same symptoms.
The patient didn’t have the same symptoms because the biologic treatment worked, said Dr. Resneck.
Kristine Grow, a spokesperson for America’s Health Insurance Plans, a national association for health insurers, said, “The use of prior authorization is relatively small – typically, less than 15% – and can help ensure safer opioid prescribing, help prevent dangerous drug interactions, and help protect patients from unnecessary exposure to potentially harmful radiation for inappropriate diagnostic imaging. Numerous studies show that Americans frequently receive inappropriate care, and 25% of unnecessary treatments are associated with complications or adverse events.”
Medical management tools, such as prior authorization, are an “an important way” to deliver “safe, high-quality care” to patients, she added.
State and federal efforts to curb prior authorization
In addition to efforts to curb prior authorization in other states, the AMA supports the Improving Seniors’ Timely Access to Care Act (HR 3173). The act includes a provision related to “gold-carding,” said Robert Mills, an AMA spokesperson.
The bill establishes requirements and standards for prior authorization processes related to Medicare Advantage (MA) plans. The requirements and standards for MA plans include the following:
- Establishing an electronic prior authorization program that meets specific standards, such as the ability to provide real-time decisions in response to requests for items and services that are routinely approved.
- Publishing on an annual basis specific prior authorization information, including the percentage of requests approved and the average response time.
- Meeting standards set by the Centers for Medicare & Medicaid Services related to the quality and timeliness of prior authorization determinations.
The act was introduced to the U.S. House of Representatives in May, after which it was referred to two committees for consideration.
A version of this article first appeared on Medscape.com.
The law was passed in June and will take effect in September. It excuses physicians from having to obtain prior authorization if, during the previous 6 months, 90% of their treatments met medical necessity criteria by the health insurer. Through this law, doctors in the state will spend less time getting approvals for treatments for their patients.
Automatic approval of authorizations for treatments – or what the Texas Medical Association (TMA) calls a “gold card” – “allows patients to get the care they need in a more timely fashion,” says Debra Patt, MD, an Austin, Tex.–based oncologist and former chair of the council on legislation for the TMA.
Eighty-seven percent of Texas physicians reported a “drastic increase over the past five years in the burden of prior authorization on their patients and their practices,” per a 2020 survey by the TMA. Nearly half (48%) of Texas physicians have hired staff whose work focuses on processing requests for prior authorization, according to the survey.
Jack Resneck Jr., MD, a San Francisco–based dermatologist and president-elect of the American Medical Association (AMA), said other states have investigated ways to ease the impact of prior authorizations on physicians, but no other state has passed such a law.
Administrative burdens plague physicians around the country. The Medscape Physician Compensation Report 2021 found that physicians spend on average 15.6 hours per week on paperwork and administrative duties.
Better outcomes, less anxiety for patients
Dr. Patt, who testified in support of the law’s passage in the Texas legislature, says automatic approval of authorizations “is better for patients because it reduces their anxiety about whether they’re able to get the treatments they need now, and they will have better outcomes if they’re able to receive more timely care.”
Recently, a chemotherapy treatment Dr. Patt prescribed for one of her patients was not authorized by an insurer. The result is “a lot of anxiety and potentially health problems” for the patient, said Dr. Patt.
She expects that automatic approval for treatments will be based on prescribing patterns during the preceding 6 months. “It means that when I order a test today, the [health insurer] looks back at my record 6 months previously,” she said. Still, Dr. Patt awaits guidance from the Texas Department of Insurance, which regulates health insurers in the state, regarding the law.
Dr. Resneck said the pharmacy counter is where most patients encounter prior authorization delays. “That’s when the pharmacist looks at them and says, ‘Actually, this isn’t covered by your health insurer’s formulary,’ or it isn’t covered fully on their formulary.”
One of Dr. Resneck’s patients had a life-altering case of eczema that lasted many years. Because of the condition, the patient couldn’t work or maintain meaningful bonds with family members. A biologic treatment transformed his patient’s life. The patient was able to return to work and to re-engage with family, said Dr. Resneck. But a year after his patient started the treatment, the health insurer wouldn’t authorize the treatment because the patient wasn’t experiencing the same symptoms.
The patient didn’t have the same symptoms because the biologic treatment worked, said Dr. Resneck.
Kristine Grow, a spokesperson for America’s Health Insurance Plans, a national association for health insurers, said, “The use of prior authorization is relatively small – typically, less than 15% – and can help ensure safer opioid prescribing, help prevent dangerous drug interactions, and help protect patients from unnecessary exposure to potentially harmful radiation for inappropriate diagnostic imaging. Numerous studies show that Americans frequently receive inappropriate care, and 25% of unnecessary treatments are associated with complications or adverse events.”
Medical management tools, such as prior authorization, are an “an important way” to deliver “safe, high-quality care” to patients, she added.
State and federal efforts to curb prior authorization
In addition to efforts to curb prior authorization in other states, the AMA supports the Improving Seniors’ Timely Access to Care Act (HR 3173). The act includes a provision related to “gold-carding,” said Robert Mills, an AMA spokesperson.
The bill establishes requirements and standards for prior authorization processes related to Medicare Advantage (MA) plans. The requirements and standards for MA plans include the following:
- Establishing an electronic prior authorization program that meets specific standards, such as the ability to provide real-time decisions in response to requests for items and services that are routinely approved.
- Publishing on an annual basis specific prior authorization information, including the percentage of requests approved and the average response time.
- Meeting standards set by the Centers for Medicare & Medicaid Services related to the quality and timeliness of prior authorization determinations.
The act was introduced to the U.S. House of Representatives in May, after which it was referred to two committees for consideration.
A version of this article first appeared on Medscape.com.
Hospital medicine and the future of smart care
People often overestimate what will happen in the next two years and underestimate what will happen in ten. – Bill Gates
The COVID-19 pandemic set in motion a series of innovations catalyzing the digital transformation of the health care landscape.
Telemedicine use exploded over the last 12 months to the point that it has almost become ubiquitous. With that, we saw a rapid proliferation of wearables and remote patient monitoring devices. Thanks to virtual care, care delivery is no longer strictly dependent on having onsite specialists, and care itself is not confined to the boundaries of hospitals or doctors’ offices anymore.
We saw the formation of the digital front door and the emergence of new virtual care sites like virtual urgent care, virtual home health, virtual office visits, virtual hospital at home that allowed clinical care to be delivered safely outside the boundaries of hospitals. Nonclinical public places like gyms, schools, and community centers were being transformed into virtual health care portals that brought care closer to the people.
Inside the hospital, we saw a fusion of traditional inpatient care and virtual care. Onsite hospital teams embraced telemedicine during the pandemic for various reasons; to conserve personal protective equipment (PPE), limit exposure, boost care capacity, improve access to specialists at distant sites, and bring family memberse to “webside” who cannot be at a patient’s bedside.
In clinical trials as well, virtual care is a welcome change. According to one survey1, most trial participants favored the use of telehealth services for clinical trials, as these helped them stay engaged, compliant, monitored, and on track while remaining at home. Furthermore, we are seeing the integration of artificial intelligence (AI) into telehealth, whether it is to aid physicians in clinical decision-making or to generate reminders to help patients with chronic disease management. However, this integration is only beginning to scratch the surface of the combination of two technologies’ real potential.
What’s next?
Based on these trends, it should be no surprise that digital health will become a vital sign for health care organizations.
The next 12 to 24 months will set new standards for digital health and play a significant role in defining the next generation of virtual care. There are projections that global health care industry revenues will exceed $2.6 trillion by 2025, with AI and telehealth playing a prominent role in this growth.2 According to estimates, telehealth itself will be a $175 billion market by 2026 and approximately one in three patient encounters will go virtual.3,4 Moreover, virtual care will continue to make exciting transformations, helping to make quality care accessible to everyone in innovative ways. For example, the University of Cincinnati has recently developed a pilot project using a drone equipped with video technology, artificial intelligence, sensors, and first aid kits to go to hard-to-reach areas to deliver care via telemedicine.5
Smart hospitals
In coming years, we can expect the integration of AI, augmented reality (AR), and virtual reality (VR) into telemedicine at lightning speed – and at a much larger scale – that will enable surgeons from different parts of the globe to perform procedures remotely and more precisely.
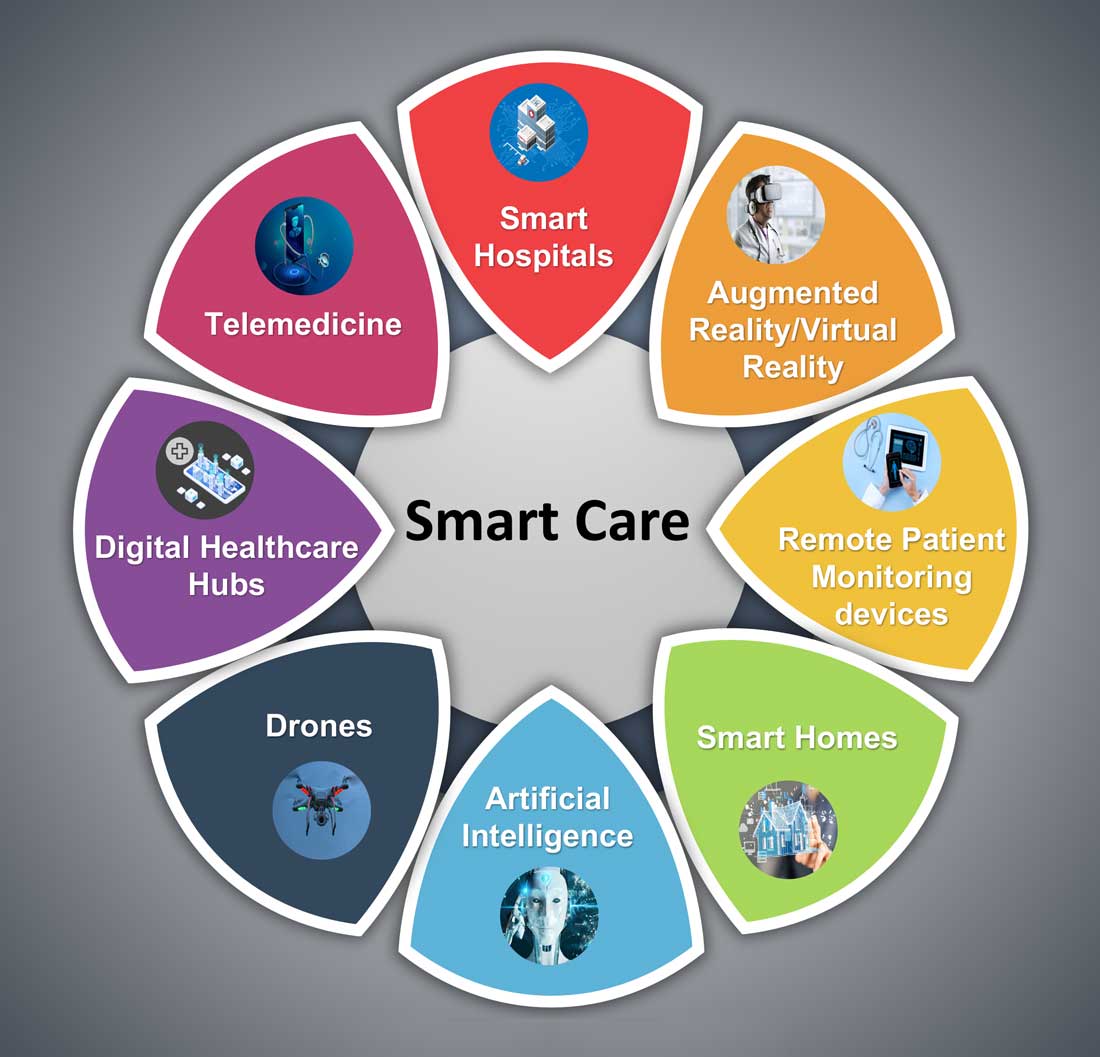
AI is already gaining traction in different fields within health care – whether it’s predicting length of stay in the ICU, or assisting in triage decisions, or reading radiological images, to name just a few. The Mayo Clinic is using AI and computer-aided decision-making tools to predict the risk of surgery and potential post-op complications, which could allow even better collaboration between medical and surgical teams. We hear about the “X-ray” vision offered to proceduralists using HoloLens – mixed reality smartglasses – a technology that enables them to perform procedures more precisely. Others project that there will be more sensors and voice recognition tools in the OR that will be used to gather data to develop intelligent algorithms, and to build a safety net for interventionalists that can notify them of potential hazards or accidental sterile field breaches. The insights gained will be used to create best practices and even allow some procedures to be performed outside the traditional OR setting.
Additionally, we are seeing the development of “smart” patient rooms. For example, one health system in Florida is working on deploying Amazon Alexa in 2,500 patient rooms to allow patients to connect more easily to their care team members. In the not-so-distant future, smart hospitals with smart patient rooms and smart ORs equipped with telemedicine, AI, AR, mixed reality, and computer-aided decision-making tools will no longer be an exception.
Smart homes for smart care
Smart homes with technologies like gas detectors, movement sensors, and sleep sensors will continue to evolve. According to one estimate, the global smart home health care market was $8.7 billion in 2019, and is expected to be $96.2 billion by 2030.6
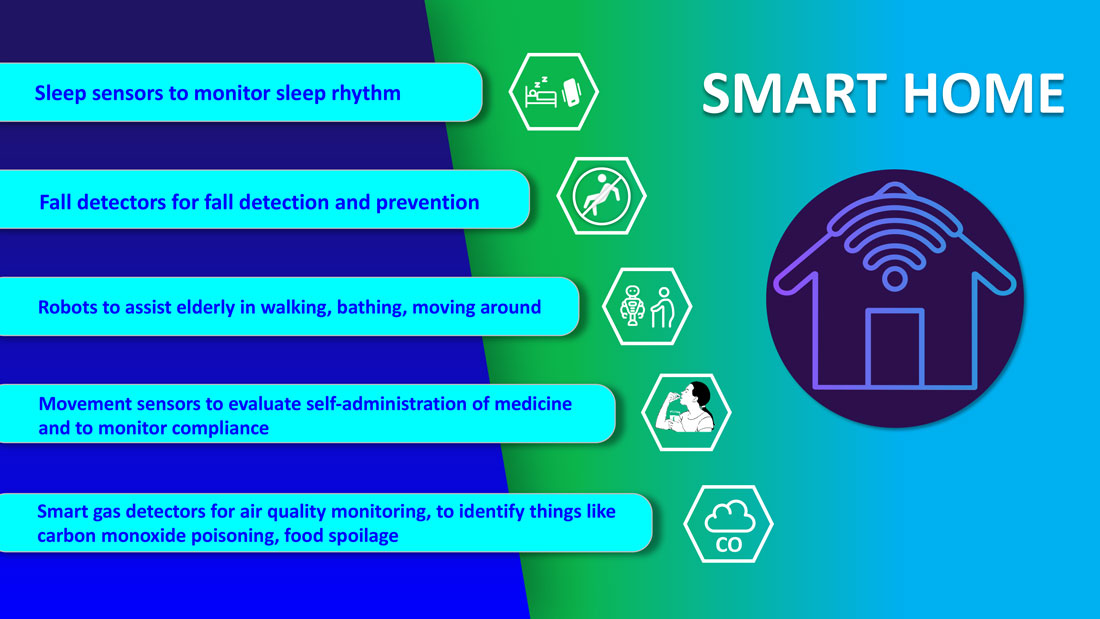
Smart technologies will have applications in fall detection and prevention, evaluation of self-administration of medicine, sleep rhythm monitoring, air quality monitoring for the detection of abnormal gas levels, and identification of things like carbon monoxide poisoning or food spoilage. In coming years, expect to see more virtual medical homes and digital health care complexes. Patients, from the convenience of their homes, might be able to connect to a suite of caregivers, all working collaboratively to provide more coordinated, effective care. The “hospital at home” model that started with six hospitals has already grown to over 100 hospitals across 29 states. The shift from onsite specialists to onscreen specialists will continue, providing greater access to specialized services.
With these emerging trends, it can be anticipated that much acute care will be provided to patients outside the hospital – either under the hospital at home model, via drone technology using telemedicine, through smart devices in smart homes, or via wearables and artificial intelligence. Hence, hospitals’ configuration in the future will be much different and more compact than currently, and many hospitals will be reserved for trauma patients, casualties of natural disasters, higher acuity diseases requiring complex procedures, and other emergencies.
The role of hospitalists has evolved over the years and is still evolving. It should be no surprise if, in the future, we work alongside a digital hospitalist twin to provide better and more personalized care to our patients. Change is uncomfortable but it is inevitable. When COVID hit, we were forced to find innovative ways to deliver care to our patients. One thing is for certain: post-pandemic (AD, or After Disease) we are not going back to a Before COVID (BC) state in terms of virtual care. With the new dawn of digital era, the crucial questions to address will be: What will the future role of a hospitalist look like? How can we leverage technology and embrace our flexibility to adapt to these trends? How can we apply the lessons learned during the pandemic to propel hospital medicine into the future? And is it time to rethink our role and even reclassify ourselves – from hospitalists to Acute Care Experts (ACE) or Primary Acute Care Physicians?
Dr. Zia is a hospitalist, physician advisor, and founder of Virtual Hospitalist - a telemedicine company with a 360-degree care model for hospital patients.
References
1. www.subjectwell.com/news/data-shows-a-majority-of-patients-remain-interested-in-clinical-trials-during-the-coronavirus-pandemic/
2. ww2.frost.com/news/press-releases/technology-innovations-and-virtual-consultations-drive-healthcare-2025/
3. www.gminsights.com/industry-analysis/telemedicine-market
4. www.healthcareitnews.com/blog/frost-sullivans-top-10-predictions-healthcare-2021
5. www.uc.edu/news/articles/2021/03/virtual-medicine--new-uc-telehealth-drone-makes-house-calls.html
6. www.psmarketresearch.com/market-analysis/smart-home-healthcare-market
People often overestimate what will happen in the next two years and underestimate what will happen in ten. – Bill Gates
The COVID-19 pandemic set in motion a series of innovations catalyzing the digital transformation of the health care landscape.
Telemedicine use exploded over the last 12 months to the point that it has almost become ubiquitous. With that, we saw a rapid proliferation of wearables and remote patient monitoring devices. Thanks to virtual care, care delivery is no longer strictly dependent on having onsite specialists, and care itself is not confined to the boundaries of hospitals or doctors’ offices anymore.
We saw the formation of the digital front door and the emergence of new virtual care sites like virtual urgent care, virtual home health, virtual office visits, virtual hospital at home that allowed clinical care to be delivered safely outside the boundaries of hospitals. Nonclinical public places like gyms, schools, and community centers were being transformed into virtual health care portals that brought care closer to the people.
Inside the hospital, we saw a fusion of traditional inpatient care and virtual care. Onsite hospital teams embraced telemedicine during the pandemic for various reasons; to conserve personal protective equipment (PPE), limit exposure, boost care capacity, improve access to specialists at distant sites, and bring family memberse to “webside” who cannot be at a patient’s bedside.
In clinical trials as well, virtual care is a welcome change. According to one survey1, most trial participants favored the use of telehealth services for clinical trials, as these helped them stay engaged, compliant, monitored, and on track while remaining at home. Furthermore, we are seeing the integration of artificial intelligence (AI) into telehealth, whether it is to aid physicians in clinical decision-making or to generate reminders to help patients with chronic disease management. However, this integration is only beginning to scratch the surface of the combination of two technologies’ real potential.
What’s next?
Based on these trends, it should be no surprise that digital health will become a vital sign for health care organizations.
The next 12 to 24 months will set new standards for digital health and play a significant role in defining the next generation of virtual care. There are projections that global health care industry revenues will exceed $2.6 trillion by 2025, with AI and telehealth playing a prominent role in this growth.2 According to estimates, telehealth itself will be a $175 billion market by 2026 and approximately one in three patient encounters will go virtual.3,4 Moreover, virtual care will continue to make exciting transformations, helping to make quality care accessible to everyone in innovative ways. For example, the University of Cincinnati has recently developed a pilot project using a drone equipped with video technology, artificial intelligence, sensors, and first aid kits to go to hard-to-reach areas to deliver care via telemedicine.5
Smart hospitals
In coming years, we can expect the integration of AI, augmented reality (AR), and virtual reality (VR) into telemedicine at lightning speed – and at a much larger scale – that will enable surgeons from different parts of the globe to perform procedures remotely and more precisely.

AI is already gaining traction in different fields within health care – whether it’s predicting length of stay in the ICU, or assisting in triage decisions, or reading radiological images, to name just a few. The Mayo Clinic is using AI and computer-aided decision-making tools to predict the risk of surgery and potential post-op complications, which could allow even better collaboration between medical and surgical teams. We hear about the “X-ray” vision offered to proceduralists using HoloLens – mixed reality smartglasses – a technology that enables them to perform procedures more precisely. Others project that there will be more sensors and voice recognition tools in the OR that will be used to gather data to develop intelligent algorithms, and to build a safety net for interventionalists that can notify them of potential hazards or accidental sterile field breaches. The insights gained will be used to create best practices and even allow some procedures to be performed outside the traditional OR setting.
Additionally, we are seeing the development of “smart” patient rooms. For example, one health system in Florida is working on deploying Amazon Alexa in 2,500 patient rooms to allow patients to connect more easily to their care team members. In the not-so-distant future, smart hospitals with smart patient rooms and smart ORs equipped with telemedicine, AI, AR, mixed reality, and computer-aided decision-making tools will no longer be an exception.
Smart homes for smart care
Smart homes with technologies like gas detectors, movement sensors, and sleep sensors will continue to evolve. According to one estimate, the global smart home health care market was $8.7 billion in 2019, and is expected to be $96.2 billion by 2030.6

Smart technologies will have applications in fall detection and prevention, evaluation of self-administration of medicine, sleep rhythm monitoring, air quality monitoring for the detection of abnormal gas levels, and identification of things like carbon monoxide poisoning or food spoilage. In coming years, expect to see more virtual medical homes and digital health care complexes. Patients, from the convenience of their homes, might be able to connect to a suite of caregivers, all working collaboratively to provide more coordinated, effective care. The “hospital at home” model that started with six hospitals has already grown to over 100 hospitals across 29 states. The shift from onsite specialists to onscreen specialists will continue, providing greater access to specialized services.
With these emerging trends, it can be anticipated that much acute care will be provided to patients outside the hospital – either under the hospital at home model, via drone technology using telemedicine, through smart devices in smart homes, or via wearables and artificial intelligence. Hence, hospitals’ configuration in the future will be much different and more compact than currently, and many hospitals will be reserved for trauma patients, casualties of natural disasters, higher acuity diseases requiring complex procedures, and other emergencies.
The role of hospitalists has evolved over the years and is still evolving. It should be no surprise if, in the future, we work alongside a digital hospitalist twin to provide better and more personalized care to our patients. Change is uncomfortable but it is inevitable. When COVID hit, we were forced to find innovative ways to deliver care to our patients. One thing is for certain: post-pandemic (AD, or After Disease) we are not going back to a Before COVID (BC) state in terms of virtual care. With the new dawn of digital era, the crucial questions to address will be: What will the future role of a hospitalist look like? How can we leverage technology and embrace our flexibility to adapt to these trends? How can we apply the lessons learned during the pandemic to propel hospital medicine into the future? And is it time to rethink our role and even reclassify ourselves – from hospitalists to Acute Care Experts (ACE) or Primary Acute Care Physicians?
Dr. Zia is a hospitalist, physician advisor, and founder of Virtual Hospitalist - a telemedicine company with a 360-degree care model for hospital patients.
References
1. www.subjectwell.com/news/data-shows-a-majority-of-patients-remain-interested-in-clinical-trials-during-the-coronavirus-pandemic/
2. ww2.frost.com/news/press-releases/technology-innovations-and-virtual-consultations-drive-healthcare-2025/
3. www.gminsights.com/industry-analysis/telemedicine-market
4. www.healthcareitnews.com/blog/frost-sullivans-top-10-predictions-healthcare-2021
5. www.uc.edu/news/articles/2021/03/virtual-medicine--new-uc-telehealth-drone-makes-house-calls.html
6. www.psmarketresearch.com/market-analysis/smart-home-healthcare-market
People often overestimate what will happen in the next two years and underestimate what will happen in ten. – Bill Gates
The COVID-19 pandemic set in motion a series of innovations catalyzing the digital transformation of the health care landscape.
Telemedicine use exploded over the last 12 months to the point that it has almost become ubiquitous. With that, we saw a rapid proliferation of wearables and remote patient monitoring devices. Thanks to virtual care, care delivery is no longer strictly dependent on having onsite specialists, and care itself is not confined to the boundaries of hospitals or doctors’ offices anymore.
We saw the formation of the digital front door and the emergence of new virtual care sites like virtual urgent care, virtual home health, virtual office visits, virtual hospital at home that allowed clinical care to be delivered safely outside the boundaries of hospitals. Nonclinical public places like gyms, schools, and community centers were being transformed into virtual health care portals that brought care closer to the people.
Inside the hospital, we saw a fusion of traditional inpatient care and virtual care. Onsite hospital teams embraced telemedicine during the pandemic for various reasons; to conserve personal protective equipment (PPE), limit exposure, boost care capacity, improve access to specialists at distant sites, and bring family memberse to “webside” who cannot be at a patient’s bedside.
In clinical trials as well, virtual care is a welcome change. According to one survey1, most trial participants favored the use of telehealth services for clinical trials, as these helped them stay engaged, compliant, monitored, and on track while remaining at home. Furthermore, we are seeing the integration of artificial intelligence (AI) into telehealth, whether it is to aid physicians in clinical decision-making or to generate reminders to help patients with chronic disease management. However, this integration is only beginning to scratch the surface of the combination of two technologies’ real potential.
What’s next?
Based on these trends, it should be no surprise that digital health will become a vital sign for health care organizations.
The next 12 to 24 months will set new standards for digital health and play a significant role in defining the next generation of virtual care. There are projections that global health care industry revenues will exceed $2.6 trillion by 2025, with AI and telehealth playing a prominent role in this growth.2 According to estimates, telehealth itself will be a $175 billion market by 2026 and approximately one in three patient encounters will go virtual.3,4 Moreover, virtual care will continue to make exciting transformations, helping to make quality care accessible to everyone in innovative ways. For example, the University of Cincinnati has recently developed a pilot project using a drone equipped with video technology, artificial intelligence, sensors, and first aid kits to go to hard-to-reach areas to deliver care via telemedicine.5
Smart hospitals
In coming years, we can expect the integration of AI, augmented reality (AR), and virtual reality (VR) into telemedicine at lightning speed – and at a much larger scale – that will enable surgeons from different parts of the globe to perform procedures remotely and more precisely.

AI is already gaining traction in different fields within health care – whether it’s predicting length of stay in the ICU, or assisting in triage decisions, or reading radiological images, to name just a few. The Mayo Clinic is using AI and computer-aided decision-making tools to predict the risk of surgery and potential post-op complications, which could allow even better collaboration between medical and surgical teams. We hear about the “X-ray” vision offered to proceduralists using HoloLens – mixed reality smartglasses – a technology that enables them to perform procedures more precisely. Others project that there will be more sensors and voice recognition tools in the OR that will be used to gather data to develop intelligent algorithms, and to build a safety net for interventionalists that can notify them of potential hazards or accidental sterile field breaches. The insights gained will be used to create best practices and even allow some procedures to be performed outside the traditional OR setting.
Additionally, we are seeing the development of “smart” patient rooms. For example, one health system in Florida is working on deploying Amazon Alexa in 2,500 patient rooms to allow patients to connect more easily to their care team members. In the not-so-distant future, smart hospitals with smart patient rooms and smart ORs equipped with telemedicine, AI, AR, mixed reality, and computer-aided decision-making tools will no longer be an exception.
Smart homes for smart care
Smart homes with technologies like gas detectors, movement sensors, and sleep sensors will continue to evolve. According to one estimate, the global smart home health care market was $8.7 billion in 2019, and is expected to be $96.2 billion by 2030.6

Smart technologies will have applications in fall detection and prevention, evaluation of self-administration of medicine, sleep rhythm monitoring, air quality monitoring for the detection of abnormal gas levels, and identification of things like carbon monoxide poisoning or food spoilage. In coming years, expect to see more virtual medical homes and digital health care complexes. Patients, from the convenience of their homes, might be able to connect to a suite of caregivers, all working collaboratively to provide more coordinated, effective care. The “hospital at home” model that started with six hospitals has already grown to over 100 hospitals across 29 states. The shift from onsite specialists to onscreen specialists will continue, providing greater access to specialized services.
With these emerging trends, it can be anticipated that much acute care will be provided to patients outside the hospital – either under the hospital at home model, via drone technology using telemedicine, through smart devices in smart homes, or via wearables and artificial intelligence. Hence, hospitals’ configuration in the future will be much different and more compact than currently, and many hospitals will be reserved for trauma patients, casualties of natural disasters, higher acuity diseases requiring complex procedures, and other emergencies.
The role of hospitalists has evolved over the years and is still evolving. It should be no surprise if, in the future, we work alongside a digital hospitalist twin to provide better and more personalized care to our patients. Change is uncomfortable but it is inevitable. When COVID hit, we were forced to find innovative ways to deliver care to our patients. One thing is for certain: post-pandemic (AD, or After Disease) we are not going back to a Before COVID (BC) state in terms of virtual care. With the new dawn of digital era, the crucial questions to address will be: What will the future role of a hospitalist look like? How can we leverage technology and embrace our flexibility to adapt to these trends? How can we apply the lessons learned during the pandemic to propel hospital medicine into the future? And is it time to rethink our role and even reclassify ourselves – from hospitalists to Acute Care Experts (ACE) or Primary Acute Care Physicians?
Dr. Zia is a hospitalist, physician advisor, and founder of Virtual Hospitalist - a telemedicine company with a 360-degree care model for hospital patients.
References
1. www.subjectwell.com/news/data-shows-a-majority-of-patients-remain-interested-in-clinical-trials-during-the-coronavirus-pandemic/
2. ww2.frost.com/news/press-releases/technology-innovations-and-virtual-consultations-drive-healthcare-2025/
3. www.gminsights.com/industry-analysis/telemedicine-market
4. www.healthcareitnews.com/blog/frost-sullivans-top-10-predictions-healthcare-2021
5. www.uc.edu/news/articles/2021/03/virtual-medicine--new-uc-telehealth-drone-makes-house-calls.html
6. www.psmarketresearch.com/market-analysis/smart-home-healthcare-market








