User login
HPV-positive women who undergo IVF don’t have worse outcomes
A new study provides more evidence that HPV infection doesn’t raise the risk of poor outcomes in women who undergo fertility treatment via in vitro fertilization with fresh embryos. In fact, HPV-positive women were somewhat more likely than HPV-negative women to become pregnant (relative risk, 1.20; 95% confidence interval, 1.03-1.39) and have live births (RR, 1.39; 95% CI, 1.13-1.70), researchers reported Oct. 24 at the American Society for Reproductive Medicine’s 2022 meeting .
“This evidence should reassure women that being HPV positive will not affect live birth rates after a fresh embryo transfer cycle,” said study coauthor and ob.gyn. Nina Vyas, MD, a clinical fellow at Weill Cornell Medicine, New York, in an interview.
According to Dr. Vyas, previous studies have offered conflicting results about whether HPV affects pregnancy outcomes. In 2006, for example, her group performed a pilot study (Fertil Steril. Jun 16. doi: 10.1016/j.fertnstert.2006.01.051) that linked lower pregnancy rates to HPV-positive tests on the day of egg retrieval.
“We sought to reevaluate this finding in a retrospective manner,” Dr. Vyas said. “You’re taking eggs out of their home, injecting with sperm, and putting them back. There’s so much that we don’t know, and we want to make sure there’s no extra risk.”
Also, she added, “prior studies had a relatively low sample size. We sought to use our patient volume to address this question on a larger scale. Our current study benefits from a large sample size and using the clinically meaningful endpoint of live birth as our primary outcome.”
For the new study, researchers retrospectively analyzed 1,333 patients (of 2,209 screened) who received first fresh embryo transfers from 2017 to 2019. All had cytology or HPV status documented per cervical cancer screening guidelines within 6 months before embryos were transferred.
The researchers looked at only fresh embryo transfers “so we could account for pregnancy outcomes closest to the documented HPV status at the time of egg retrieval,” Dr. Vyas said.
Ten percent (133) of patients were HPV positive. Of those, 60.1% became pregnant, and 43.6% of them had live births. Of the HPV-negative women (90% of subjects, n = 1,200), 52.2% became pregnant and 33.5% had live births. The researchers didn’t calculate P values, but Dr. Vyas said an analysis determined that the differences between HPV-positive and HPV-negative women were statistically significant.
The study size doesn’t allow researchers to determine whether HPV actually has a protective effect on pregnancy/live birth rates in IVF, Dr. Vyas said. Even if it did, the virus is dangerous.
What else could explain the discrepancy? “Some elements driving this could the smaller sample size of the HPV-positive group, differences in HPV prevalence between the general population and our population,” she said, “or other confounding factors we were not able to appreciate due to the limitations of the retrospective study.”
Researchers also reported that they found “no significant difference in biochemical or spontaneous abortion rates” between HPV-positive and HPV-negative women.
What is the message of the study? “Women with HPV can rest assured that they won’t have worse outcomes than their non-HPV [infected] counterparts after a fresh embryo transfer cycle,” Dr. Vyas said.
In an interview, McGill University, Montreal, epidemiologist Helen Trottier, PhD, MSc, noted that she recently coauthored a study that linked persistent HPV infection in pregnancy to premature births. The findings appear convincing, she said: “I think we can say that HPV is associated with preterm birth.”
She praised the new study but noted “the relative risks that are reported need to be adjusted for race and possibly other factors.”
Dr. Vyas said that kind of adjustment will occur in a future study that’s in progress. “We are now prospectively enrolling patients and collecting cytology data to understand whether there might be a difference for women with higher malignancy potential/different types of HPV genotypes.”
The study authors have no disclosures. Disclosure information for Dr. Trottier was unavailable.
A new study provides more evidence that HPV infection doesn’t raise the risk of poor outcomes in women who undergo fertility treatment via in vitro fertilization with fresh embryos. In fact, HPV-positive women were somewhat more likely than HPV-negative women to become pregnant (relative risk, 1.20; 95% confidence interval, 1.03-1.39) and have live births (RR, 1.39; 95% CI, 1.13-1.70), researchers reported Oct. 24 at the American Society for Reproductive Medicine’s 2022 meeting .
“This evidence should reassure women that being HPV positive will not affect live birth rates after a fresh embryo transfer cycle,” said study coauthor and ob.gyn. Nina Vyas, MD, a clinical fellow at Weill Cornell Medicine, New York, in an interview.
According to Dr. Vyas, previous studies have offered conflicting results about whether HPV affects pregnancy outcomes. In 2006, for example, her group performed a pilot study (Fertil Steril. Jun 16. doi: 10.1016/j.fertnstert.2006.01.051) that linked lower pregnancy rates to HPV-positive tests on the day of egg retrieval.
“We sought to reevaluate this finding in a retrospective manner,” Dr. Vyas said. “You’re taking eggs out of their home, injecting with sperm, and putting them back. There’s so much that we don’t know, and we want to make sure there’s no extra risk.”
Also, she added, “prior studies had a relatively low sample size. We sought to use our patient volume to address this question on a larger scale. Our current study benefits from a large sample size and using the clinically meaningful endpoint of live birth as our primary outcome.”
For the new study, researchers retrospectively analyzed 1,333 patients (of 2,209 screened) who received first fresh embryo transfers from 2017 to 2019. All had cytology or HPV status documented per cervical cancer screening guidelines within 6 months before embryos were transferred.
The researchers looked at only fresh embryo transfers “so we could account for pregnancy outcomes closest to the documented HPV status at the time of egg retrieval,” Dr. Vyas said.
Ten percent (133) of patients were HPV positive. Of those, 60.1% became pregnant, and 43.6% of them had live births. Of the HPV-negative women (90% of subjects, n = 1,200), 52.2% became pregnant and 33.5% had live births. The researchers didn’t calculate P values, but Dr. Vyas said an analysis determined that the differences between HPV-positive and HPV-negative women were statistically significant.
The study size doesn’t allow researchers to determine whether HPV actually has a protective effect on pregnancy/live birth rates in IVF, Dr. Vyas said. Even if it did, the virus is dangerous.
What else could explain the discrepancy? “Some elements driving this could the smaller sample size of the HPV-positive group, differences in HPV prevalence between the general population and our population,” she said, “or other confounding factors we were not able to appreciate due to the limitations of the retrospective study.”
Researchers also reported that they found “no significant difference in biochemical or spontaneous abortion rates” between HPV-positive and HPV-negative women.
What is the message of the study? “Women with HPV can rest assured that they won’t have worse outcomes than their non-HPV [infected] counterparts after a fresh embryo transfer cycle,” Dr. Vyas said.
In an interview, McGill University, Montreal, epidemiologist Helen Trottier, PhD, MSc, noted that she recently coauthored a study that linked persistent HPV infection in pregnancy to premature births. The findings appear convincing, she said: “I think we can say that HPV is associated with preterm birth.”
She praised the new study but noted “the relative risks that are reported need to be adjusted for race and possibly other factors.”
Dr. Vyas said that kind of adjustment will occur in a future study that’s in progress. “We are now prospectively enrolling patients and collecting cytology data to understand whether there might be a difference for women with higher malignancy potential/different types of HPV genotypes.”
The study authors have no disclosures. Disclosure information for Dr. Trottier was unavailable.
A new study provides more evidence that HPV infection doesn’t raise the risk of poor outcomes in women who undergo fertility treatment via in vitro fertilization with fresh embryos. In fact, HPV-positive women were somewhat more likely than HPV-negative women to become pregnant (relative risk, 1.20; 95% confidence interval, 1.03-1.39) and have live births (RR, 1.39; 95% CI, 1.13-1.70), researchers reported Oct. 24 at the American Society for Reproductive Medicine’s 2022 meeting .
“This evidence should reassure women that being HPV positive will not affect live birth rates after a fresh embryo transfer cycle,” said study coauthor and ob.gyn. Nina Vyas, MD, a clinical fellow at Weill Cornell Medicine, New York, in an interview.
According to Dr. Vyas, previous studies have offered conflicting results about whether HPV affects pregnancy outcomes. In 2006, for example, her group performed a pilot study (Fertil Steril. Jun 16. doi: 10.1016/j.fertnstert.2006.01.051) that linked lower pregnancy rates to HPV-positive tests on the day of egg retrieval.
“We sought to reevaluate this finding in a retrospective manner,” Dr. Vyas said. “You’re taking eggs out of their home, injecting with sperm, and putting them back. There’s so much that we don’t know, and we want to make sure there’s no extra risk.”
Also, she added, “prior studies had a relatively low sample size. We sought to use our patient volume to address this question on a larger scale. Our current study benefits from a large sample size and using the clinically meaningful endpoint of live birth as our primary outcome.”
For the new study, researchers retrospectively analyzed 1,333 patients (of 2,209 screened) who received first fresh embryo transfers from 2017 to 2019. All had cytology or HPV status documented per cervical cancer screening guidelines within 6 months before embryos were transferred.
The researchers looked at only fresh embryo transfers “so we could account for pregnancy outcomes closest to the documented HPV status at the time of egg retrieval,” Dr. Vyas said.
Ten percent (133) of patients were HPV positive. Of those, 60.1% became pregnant, and 43.6% of them had live births. Of the HPV-negative women (90% of subjects, n = 1,200), 52.2% became pregnant and 33.5% had live births. The researchers didn’t calculate P values, but Dr. Vyas said an analysis determined that the differences between HPV-positive and HPV-negative women were statistically significant.
The study size doesn’t allow researchers to determine whether HPV actually has a protective effect on pregnancy/live birth rates in IVF, Dr. Vyas said. Even if it did, the virus is dangerous.
What else could explain the discrepancy? “Some elements driving this could the smaller sample size of the HPV-positive group, differences in HPV prevalence between the general population and our population,” she said, “or other confounding factors we were not able to appreciate due to the limitations of the retrospective study.”
Researchers also reported that they found “no significant difference in biochemical or spontaneous abortion rates” between HPV-positive and HPV-negative women.
What is the message of the study? “Women with HPV can rest assured that they won’t have worse outcomes than their non-HPV [infected] counterparts after a fresh embryo transfer cycle,” Dr. Vyas said.
In an interview, McGill University, Montreal, epidemiologist Helen Trottier, PhD, MSc, noted that she recently coauthored a study that linked persistent HPV infection in pregnancy to premature births. The findings appear convincing, she said: “I think we can say that HPV is associated with preterm birth.”
She praised the new study but noted “the relative risks that are reported need to be adjusted for race and possibly other factors.”
Dr. Vyas said that kind of adjustment will occur in a future study that’s in progress. “We are now prospectively enrolling patients and collecting cytology data to understand whether there might be a difference for women with higher malignancy potential/different types of HPV genotypes.”
The study authors have no disclosures. Disclosure information for Dr. Trottier was unavailable.
FROM ASRM 2022
IgA Vasculitis in the Setting of Biologic Therapy for Psoriasis and Recurrent Cutaneous Methicillin-Resistant Staphylococcus aureus Colonization
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
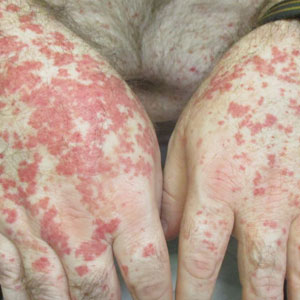
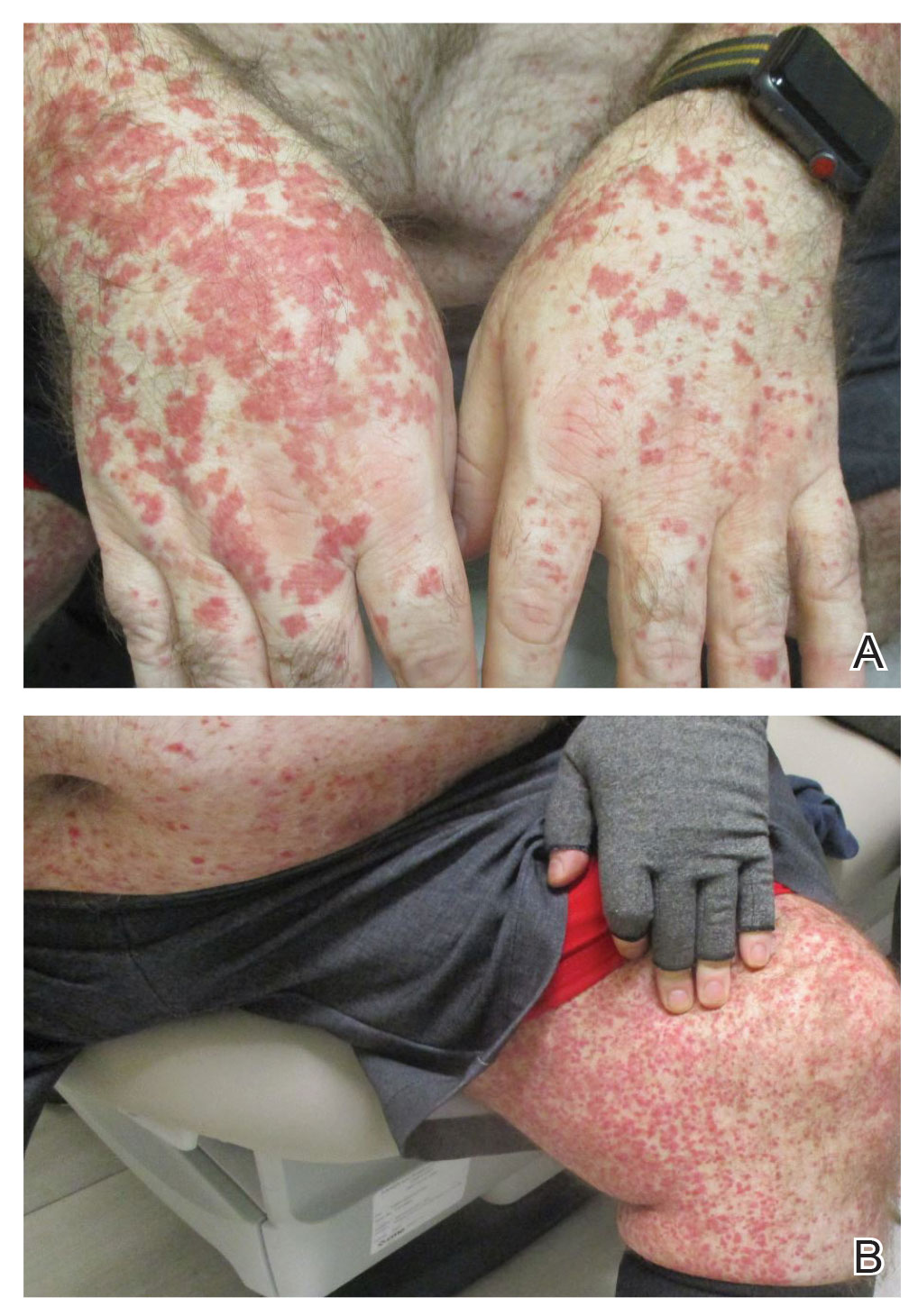
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.
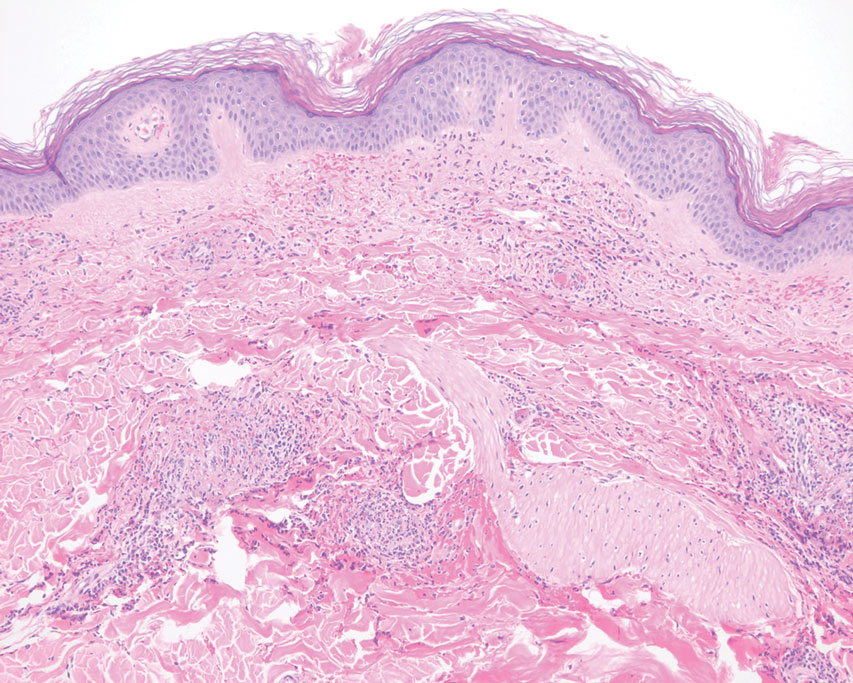
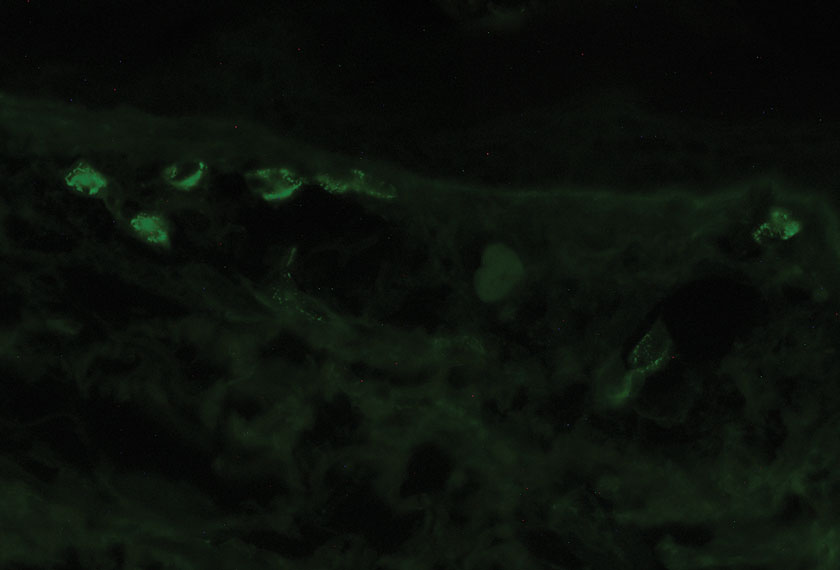
Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.
Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.
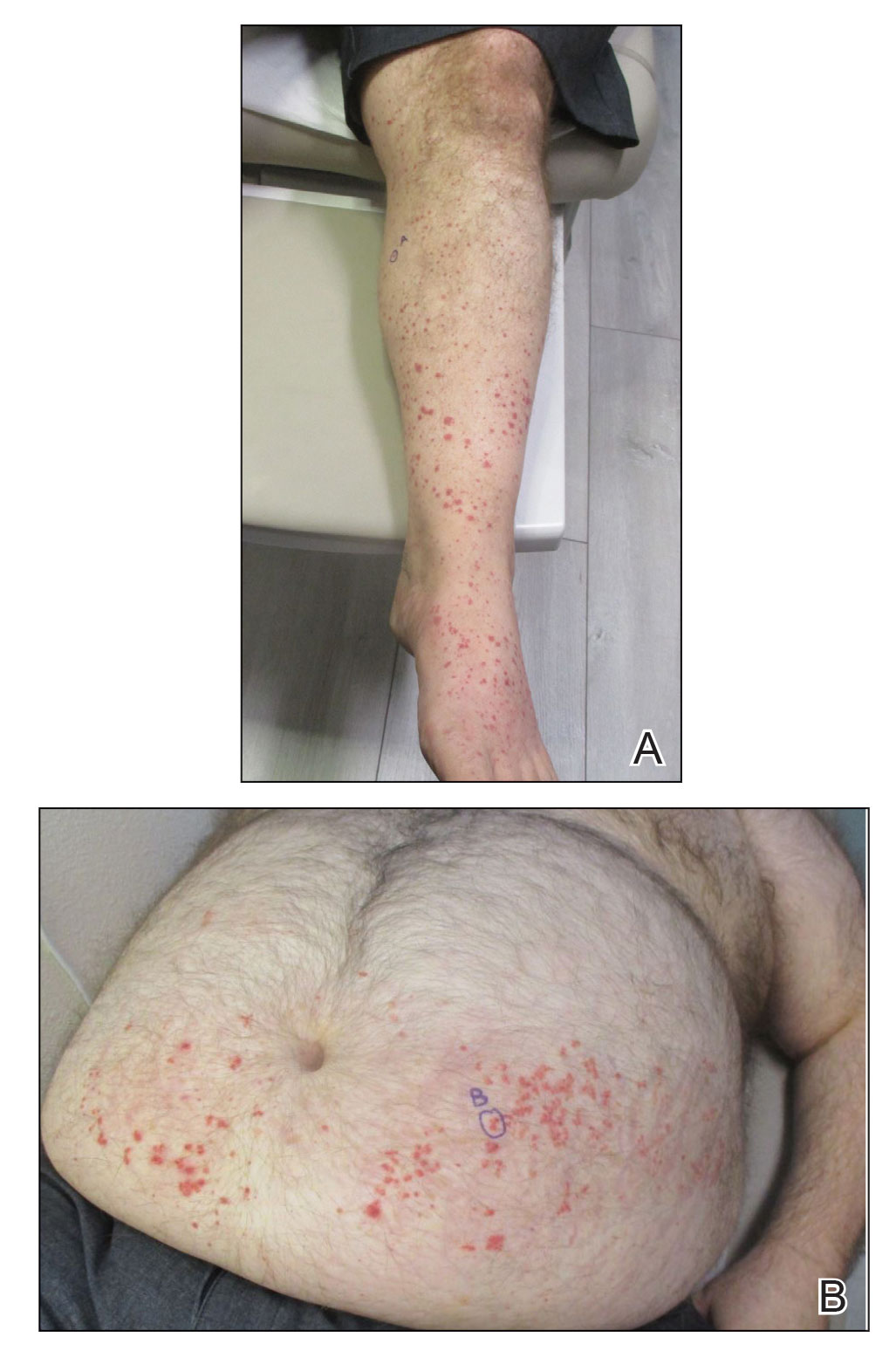
Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.
Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
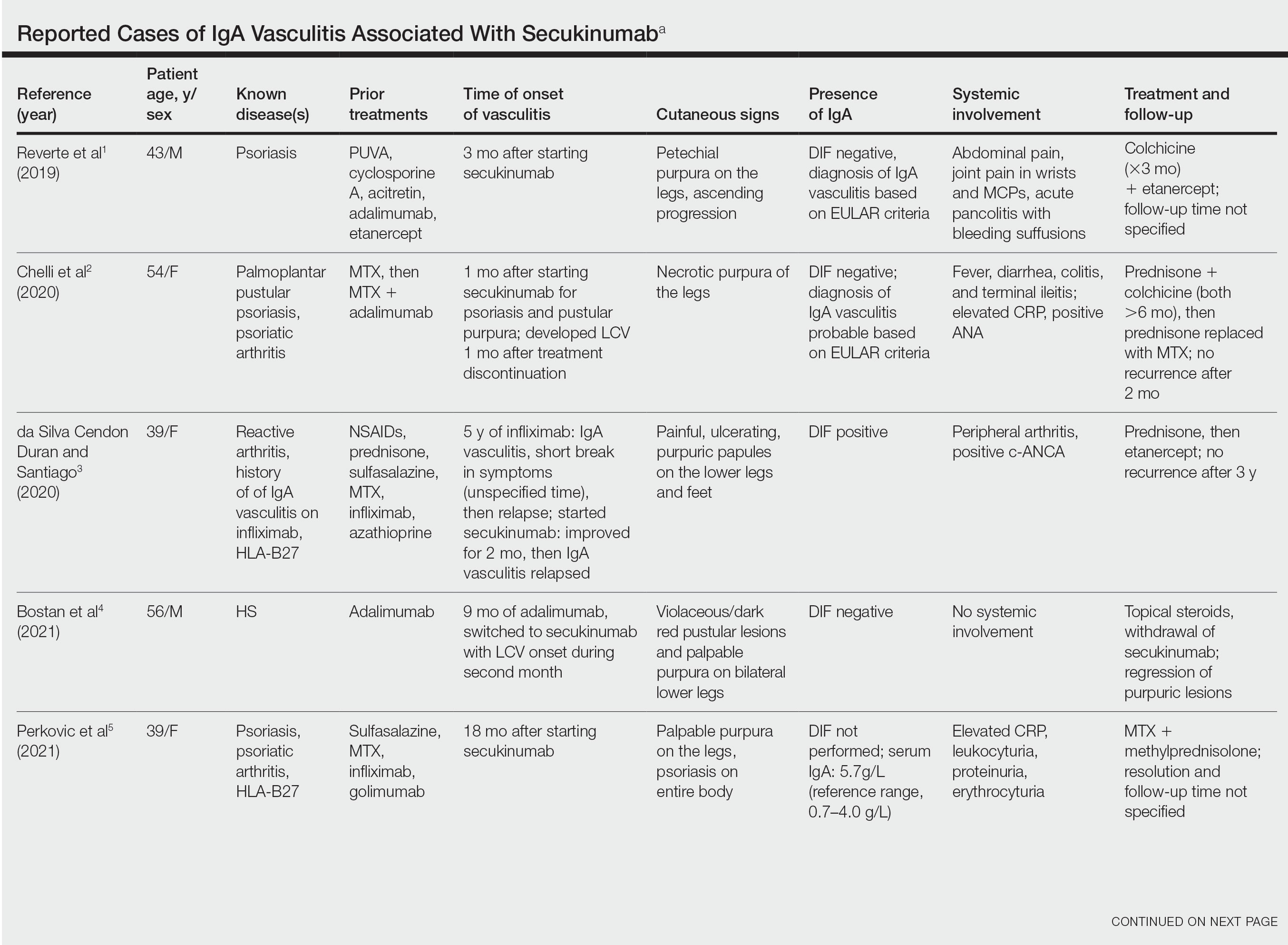
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7
The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10
There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.

Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.

Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.

Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.

Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7

The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10

There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
Case Report
A 47-year-old man presented with a sudden-onset rash consisting of red bumps on the abdomen and legs that had been ongoing for several days. He had known psoriasis and psoriatic arthritis that had been well controlled with adalimumab for the last 18 months. He reported concurrent onset of nausea but denied fevers, chills, night sweats, unintentional weight loss, abdominal pain, and pruritus. He endorsed prior cutaneous infections of methicillin-resistant Staphylococcus aureus (MRSA). His medical history also included diabetes mellitus, hypertension, and obesity. His other medications included oral losartan-hydrochlorothiazide, amlodipine, naproxen, and atorvastatin.
Physical examination revealed numerous thin purpuric papules—some with adherent scale—distributed on the lower legs, extensor forearms, and abdomen. Abdominal lesions were confined to weight-related striae (Figure 1). The palms, soles, oral mucosa, and face were spared. Three punch biopsies were performed, including 1 for direct immunofluorescence (DIF), and the patient was instructed to apply clobetasol to the affected areas twice daily until further notice.

Pathology showed perivascular extravasation of erythrocytes, neutrophils, eosinophils, and leukocytoclasis surrounding blood vessels associated with fibrin (Figure 2). Direct immunofluorescence showed granular deposition of IgA, complement component 3, and fibrinogen in a superficial dermal vascular pattern (Figure 3). These results were consistent with IgA small-vessel vasculitis. One specimen was consistent with the patient’s known psoriasis.

Urinalysis revealed moderate hemoglobinuria, and urine microscopy showed 174 red blood cells per high-power field. Creatinine was high at 1.87 mg/dL (reference range, <1.34 mg/dL; patient’s baseline, 0.81 mg/dL) and glomerular filtration rate was low (42 mL/min, patient’s baseline, >60 mL/min [reference range, 90–120 mL/min]). Erythrocyte sedimentation rate (21 mm/h [reference range, 0–22 mm/h]) and C-reactive protein were elevated (2.2 mg/dL [reference range, 0.3–1.0 mg/dL]). Given his history of cutaneous MRSA infections, a bacterial culture swab was collected from the skin surface to check for colonization, which showed moderate growth of MRSA. Naproxen was discontinued over concern of worsening the patient’s renal status. The patient was instructed to rest at home with his legs elevated, wear compression socks when ambulatory, use chlorhexidine antiseptic daily as a body wash when showering, and apply mupirocin three times daily to the biopsy sites. He was referred to urology for his microhematuria, where cystoscopy revealed no abnormalities.A month passed with no improvement of the patient’s cutaneous vasculitis, and his psoriatic arthritis worsened without his usual use of naproxen. He developed abdominal pain and loss of appetite. A prednisone taper was ordered starting at 40 mg/d (28.8 mg/kg), which provided relief of the skin and joint symptoms only until the course was completed 12 days later.

Five weeks after the initial presentation, the patient returned with a more severe eruption consisting of innumerable purpuric papules that coalesced in plaques on the abdomen, arms, and legs. He also had erythematous facial pustules and mild palmar petechiae (Figure 4). Three biopsies were performed, including 1 for DIF and 1 from a pustule on the forehead. Histology and DIF were again consistent with IgA small-vessel vasculitis. The forehead biopsy was compatible with steroid acne (attributed to recent prednisone use) and psoriasis.

Rheumatology was consulted, and adalimumab was discontinued 6 weeks after the initial presentation out of concern for drug-induced cutaneous vasculitis. Vasculitis work-up was unremarkable, including antineutrophil cytoplasmic antibodies, rheumatoid factor, cyclic citrullinated peptide, and serum protein electrophoresis. Oral dapsone was started at 100 mg/d, with the tentative plan of starting secukinumab if cutaneous symptoms improved. For 3 weeks, the patient’s cutaneous symptoms steadily improved.
Nine weeks after initial presentation to dermatology (3 weeks after discontinuing adalimumab) the patient self-administered his first dose of secukinumab at home. Several hours later, he reported sudden reappearance of vasculitis. He denied diarrhea, abdominal pain, bowel movement urgency, fevers, fatigue, and unintentional weight loss. Antistreptolysin O and hepatitis A antibodies were negative. He was instructed to hold secukinumab indefinitely.
Four weeks after his only secukinumab injection, the patient reported another episode of acute worsening cutaneous symptoms. A 4-week prednisone taper starting at 40 mg/d was ordered. Computed tomography of the chest, abdomen, and pelvis to rule out internal malignancy was unremarkable. Around this time, the patient reported major emotional distress related to an unexpected death in his family, which added to a gradual increase in his stress level related to the COVID-19 pandemic.
Three weeks later, dapsone was increased to 100 mg twice daily on account of the patient’s adiposity and lack of cutaneous improvement on the lower dose. Subsequently, the vasculitis rapidly improved for 2 weeks. The patient then reported symptoms of headache, dizziness, and chills. He was tested for COVID-19 and was negative. Six weeks after increasing the dapsone dose (5 months after initial presentation), the skin was normalizing, showing only faintly hyperpigmented macules confined to areas of resolved vasculitis (forearms, abdomen, legs).
The patient had been on dapsone 100 mg twice daily for 3 months when he was started on ustekinumab (90 mg at weeks 0 and 4, with planned doses every 12 weeks) for psoriatic arthritis in hopes of withdrawing dapsone. His cutaneous symptoms have remained well controlled on this regimen for 18 months. Lowering of dapsone below 100 mg daily has resulted in recurrent mild vasculitis symptoms; he now maintains the once-daily dosing without negative side effects.
Comment
IgA vasculitis is a form of cutaneous small-vessel leukocytoclastic vasculitis (LCV) characterized by episodes of palpable purpura on the extensor surfaces of the arms and legs that may be associated with arthritis, abdominal pain, and/or hematuria. Although vasculitis is a known potential adverse effect of anti–tumor necrosis factor (TNF) α therapy, cases of adalimumab-induced IgA vasculitis are uncommon. As use of more targeted therapies for psoriasis and psoriatic arthritis, such as the IL-17 inhibitor secukinumab, increases so do reports of associated adverse events. Of 6 previously reported cases of secukinumab-associated vasculitis, at least 4 were IgA vasculitis (Table).1-6 Another case described one patient with rheumatoid arthritis undergoing secukinumab treatment who experienced necrotizing glomerulonephritis; however, the authors concluded secukinumab likely was not causative in that case, as serologies and urinalyses suggested gradual onset of the process prior to initiating the medication.7

The exact pathogenesis of IgA vasculitis is unclear, but a prevailing theory involves the dysregulation of IgA synthesis and metabolism. Other than increased serum levels of transforming growth factor β, which is a major stimulating factor for IgA production, it also has been hypothesized that the presence of aberrantly hypoglycosylated IgA exposes an autoepitope for recognition by other pathogenic IgG and IgA, leading to the formation of large immune complexes that can readily deposit in postcapillary venules. The deposition of IgA immune complexes in postcapillary venules and the subsequent activation of the complement system causes direct damage to the endothelial cells of vessel walls. This complement activation is evidenced by vascular complement component 3 deposition on DIF (a nonspecific feature of LCV). Chemotaxis of neutrophils ensues, followed by their firm adherence and transendothelial migration (mediated by monocyte chemoattractant protein 1 [MCP-1]). Neutrophil degranulation releases reactive oxygen species and cytokines, which in turn recruit additional leukocytes to the area of inflammation, subsequently undergoing degeneration (leukocytoclasis). Microvascular permeability also is enhanced by MCP-1, allowing exudation of serum, erythrocytes, and fibrin. In the setting of elevated circulating TNF and IL-1, endothelium is stimulated to activate the intrinsic and extrinsic coagulation pathways. This decreases endothelial fibrinolytic activity, leading to thrombosis. The high venous pressure and low fibrinolytic activity in the lower legs explains why vasculitic lesions often are confined to or begin in this distribution.1,8-10

There also are noteworthy roles for cytokines in LCV. Circulating transforming growth factor β and IL-6—which are necessary for development of T helper 17 (TH17) cells and production of IL-17—are higher in patients with LCV compared to controls. Peripheral blood monocytes in patients with LCV demonstrate higher production of IL-17. Once TH17 cells develop, their survival and phenotype are maintained by IL-23 (considered the master regulator of TH17 differentiation). IL-17 is a potent chemoattractant of IL-8 (CXCL8) and MCP-1, both of which promote neutrophil-mediated perivascular inflammation. The IL-23 and IL-17 pathways implicated in the pathogenesis of psoriasis also cause neutrophil activation and upregulate transcription of proinflammatory cytokines (IL-1, IL-6, IL-8, and TNF-α), which overlap with those implicated in LCV. Autoimmune disease generally entails some positive feedback loop of progressively severe self-recognition and tissue destruction by the immune system. These shared cytokinetic processes may explain how the internal environment of psoriasis could perpetuate IgA vasculitis.1,2,8,10-12
The mechanisms underlying vasculitis associated with adalimumab are unclear, but hypotheses involve direct toxicity on vessels, capillary deposition of anti-TNF/TNF immune complexes, or an inflammatory process resulting in autoantibodies. Similar hypotheses are posited for secukinumab-associated vasculitis, including deposition of secukinumab–IL-17 complexes. Anti–TNF-α medications may increase TH17 cell numbers, leading to increased production of IL-22 and a resultant immunologic microenvironment conducive to vasculitis. All 6 published cases of secukinumab-associated vasculitis that we found had received prior treatment with a TNF-α blocker, but only 1 had occurrence of vasculitis during that treatment.1-6,10
In the 6 cases we reviewed, the time from starting secukinumab to onset of vasculitis ranged from 1 to 18 months. Our patient’s same-day re-emergence of vasculitis after his first secukinumab dose was so acute that we were skeptical of secukinumab as a potential trigger; this may simply have been coincident to the natural waxing and waning of the vasculitis (although onset of IgA vasculitis within 1 day of starting anti–TNF-α therapy has been reported).1-6,13
Specific associations of IgA vasculitis are many and can include bacterial organisms such as Helicobacter pylori, streptococci, and staphylococci. Although internal mucous membrane infections are considered more linked because of the surveillance role of IgA predominantly in mucosal tissues, it is possible that our patient with cutaneous MRSA harbored the same within the nasal mucosa. Our patient also received multiple vaccinations outside our department throughout his clinical course (2 hepatitis B and 1 pneumococcal conjugate), which are known potential triggers for vasculitis. Psychological stress is a known trigger for psoriasis, and given the cytokinetic relationship of psoriasis to vasculitis described previously, it may have indirectly contributed to vasculitis in our case. The anxiety associated with being immunosuppressed during the COVID-19 pandemic and bereavement of losing a family member may have contributed to the refractory nature of our patient’s condition. Renal involvement is relatively common in adults with IgA vasculitis and so should be ruled out, as should occult internal malignancy.8,10,14
It is unclear which of the above factors was causative in our case, but a multifactorial process is likely. Treatment of monoclonal antibody–associated vasculitis entails investigating for triggers and systemic involvement, removing the most likely culprit, quelling the vasculitis acutely, avoiding known potential exacerbators, and introducing an alternative long-term immunomodulant. In all 6 reported similar cases, discontinuation of secukinumab and initiation of prednisone or colchicine led to resolution.1-6 Dapsone also is acceptable for acute control of IgA vasculitis, although this medication is highly lipid soluble and penetrates well into various tissues.15 Thus, lower doses may prove ineffective for obese patients, as was demonstrated in our case. Given the known potential of vaccinations, infections, and other factors (eg, alcohol, penicillin) to trigger IgA vasculitis, these should be avoided.10
Blockade of IL-23 with ustekinumab has been suggested by other authors encountering secukinumab-associated vasculitis, as IL-23 is the main driver and sustainer of TH17 cell differentiation.8 Although 6 previously reported cases of secukinumab-associated vasculitis achieved resolution without long-term recurrence, none did so using an IL-23 inhibitor (nor had any of the described patients received IL-23 inhibitors previously).1-6 Given the established safety of IL-23 inhibitors and that they theoretically are well suited for this unique circumstance (by ceasing the main causative cytokine cascades “upstream”) and were efficacious in quickly resolving our patient’s vasculitis, we suggest that ustekinumab may represent
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
- Reverte M, Etienne M, Fouchard M, et al. Occurrence of Henoch-Schönlein purpura in a patient treated with secukinumab. J Eur Acad Dermatol Venereol. 2019;33:E455-E457.
- Chelli C, Loget J, Vanhaecke C, et al. Cutaneous vasculitis with gut involvement during secukinumab treatment for psoriatic arthritis. Acta Derm Venereol. 2020;100:adv00077.
- da Silva Cendon Duran C, Santiago MB. Cutaneous vasculitis during secukinumab treatment. Eur J Case Rep Intern Med. 2020;7:001815.
- Bostan E, Gulseren D, Yalici-Armagan B, et al. Vasculitis during certolizumab pegol and secukinumab treatment: report of two cases. Dermatol Ther. 2021;34:E15007.
- Perkovic D, Simac P, Katic J. IgA vasculitis during secukinumab therapy. Clin Rheumatol. 2021;40:2071-2073.
- Villani A, DE Fata Salvatores G, Nappa P, et al. Cutaneous leucocytoclastic vasculitis during secukinumab treatment. Ital J Dermatol Venerol. 2021;156(suppl 1 to no. 6):9-10.
- Góis M, Messias A, Carvalho D, et al. MPO-ANCA-associated necrotizing glomerulonephritis in rheumatoid arthritis; a case report and review of literature. J Nephropathol. 2017;6:58-62.
- Jen HY, Chuang YH, Lin SC, et al. Increased serum interleukin-17 and peripheral Th17 cells in children with acute Henoch-Schönlein purpura. Pediatr Allergy Immunol. 2011;22:862-868.
- Hetland LE, Susrud KS, Lindahl KH, et al. Henoch-Schönlein purpura: a literature review. Acta Derm Venereol 2017;97:1160-1166.
- Weedon D. The vasculopathic reaction pattern. In: Houston M, Davie B, eds. Weedon’s Skin Pathology. 3rd ed. Elsevier Limited; 2010:207-211.
- Puig L. Paradoxical reactions: anti-TNFα ants, ustekinumab, secukinumab, ixekizumab, and others. Curr Probl Dermatol. 2018;53:49-63.
- Nestle F, Kaplan D, Barker J. Psoriasis. N Engl J Med. 2009;361:496-509.
- Pinheiro RR, Lencastre A. Henoch-Schönlein purpura during anti-TNFα therapy: a fortuitous event or an indication to stop therapy? Eur J Dermatol. 2017;27:304-305.
- Hello CL, Cohen P, Bousser MG, et al. Suspected hepatitis B vaccination related vasculitis. J Rheumatol. 1999;26:191-194.
- Wolverton SE. Dapsone. In: Wolverton SE, Wu JJ, eds. Comprehensive Dermatologic Drug Therapy. 4th ed. Elsevier, Inc; 2021:222-231.
Practice Points
- Biologic medications including adalimumab and more rarely secukinumab may be associated with leukocytoclastic vasculitis; a smaller subset of patients may experience IgA vasculitis.
- The IL-23 blocker ustekinumab may represent an ideal therapeutic agent when secukinumabassociated vasculitis is suspected. Because IL-23 is the main driver and sustainer of TH17 cell differentiation, it may cease the main causative cytokine cascades “upstream.”
Side effects from COVID vaccine show its effectiveness
It means your body had a greater antibody response than people who had just a little pain or rash at the injection site, or no reaction at all.
That’s according to new research published in the journal JAMA Network Open .
“These findings support reframing postvaccination symptoms as signals of vaccine effectiveness and reinforce guidelines for vaccine boosters in older adults,” researchers from Columbia University in New York, the University of Vermont, and Boston University wrote.
The vaccines provided strong protection regardless of the level of reaction, researchers said. Almost all the study’s 928 adult participants had a positive antibody response after receiving two doses of vaccine.
“I don’t want a patient to tell me that, ‘Golly, I didn’t get any reaction, my arm wasn’t sore, I didn’t have fever. The vaccine didn’t work.’ I don’t want that conclusion to be out there,” William Schaffner, MD, a professor in the division of infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn., told CNN.
“This is more to reassure people who have had a reaction that that’s their immune system responding, actually in a rather good way, to the vaccine, even though it has caused them some discomfort,” said Dr. Schaffner, who was not involved in the study.
A version of this article first appeared on WebMD.com.
It means your body had a greater antibody response than people who had just a little pain or rash at the injection site, or no reaction at all.
That’s according to new research published in the journal JAMA Network Open .
“These findings support reframing postvaccination symptoms as signals of vaccine effectiveness and reinforce guidelines for vaccine boosters in older adults,” researchers from Columbia University in New York, the University of Vermont, and Boston University wrote.
The vaccines provided strong protection regardless of the level of reaction, researchers said. Almost all the study’s 928 adult participants had a positive antibody response after receiving two doses of vaccine.
“I don’t want a patient to tell me that, ‘Golly, I didn’t get any reaction, my arm wasn’t sore, I didn’t have fever. The vaccine didn’t work.’ I don’t want that conclusion to be out there,” William Schaffner, MD, a professor in the division of infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn., told CNN.
“This is more to reassure people who have had a reaction that that’s their immune system responding, actually in a rather good way, to the vaccine, even though it has caused them some discomfort,” said Dr. Schaffner, who was not involved in the study.
A version of this article first appeared on WebMD.com.
It means your body had a greater antibody response than people who had just a little pain or rash at the injection site, or no reaction at all.
That’s according to new research published in the journal JAMA Network Open .
“These findings support reframing postvaccination symptoms as signals of vaccine effectiveness and reinforce guidelines for vaccine boosters in older adults,” researchers from Columbia University in New York, the University of Vermont, and Boston University wrote.
The vaccines provided strong protection regardless of the level of reaction, researchers said. Almost all the study’s 928 adult participants had a positive antibody response after receiving two doses of vaccine.
“I don’t want a patient to tell me that, ‘Golly, I didn’t get any reaction, my arm wasn’t sore, I didn’t have fever. The vaccine didn’t work.’ I don’t want that conclusion to be out there,” William Schaffner, MD, a professor in the division of infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn., told CNN.
“This is more to reassure people who have had a reaction that that’s their immune system responding, actually in a rather good way, to the vaccine, even though it has caused them some discomfort,” said Dr. Schaffner, who was not involved in the study.
A version of this article first appeared on WebMD.com.
FROM JAMA NETWORK OPEN
Children and COVID: Weekly cases fall to lowest level in over a year
With the third autumn of the COVID era now upon us, the discussion has turned again to a possible influenza/COVID twindemic, as well as the new-for-2022 influenza/COVID/respiratory syncytial virus tripledemic. It appears, however, that COVID may have missed the memo.
For the sixth time in the last 7 weeks, the number of new COVID cases in children fell, with just under 23,000 reported during the week of Oct. 14-20, according to the American Academy of Pediatrics and the Children’s Hospital Association. That is the lowest weekly count so far this year, and the lowest since early July of 2021, just as the Delta surge was starting. New pediatric cases had dipped to 8,500, the lowest for any week during the pandemic, a couple of weeks before that, the AAP/CHA data show.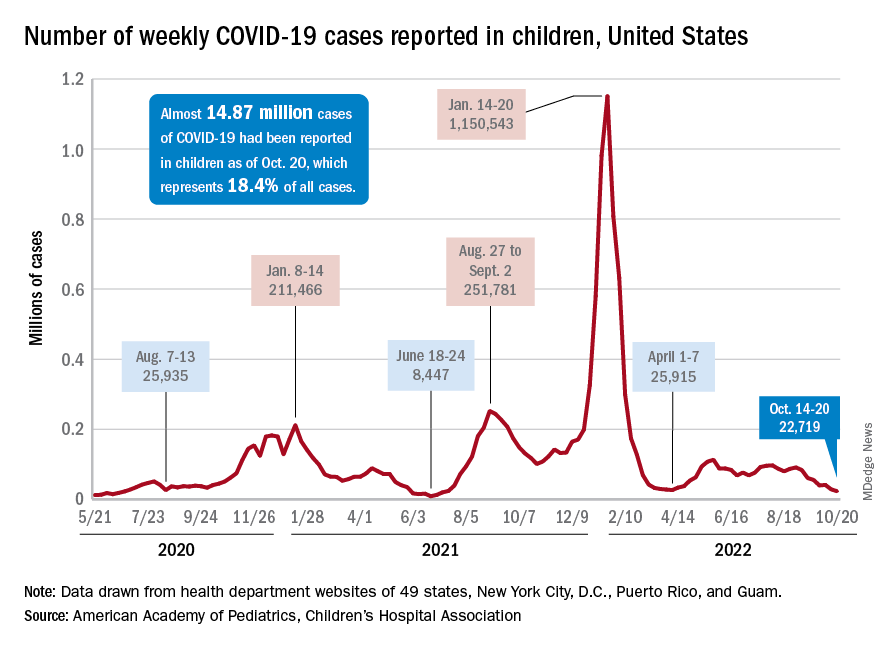
Weekly cases have fallen by almost 75% since over 90,000 were reported for the week of Aug. 26 to Sept. 1, even as children have returned to school and vaccine uptake remains slow in the youngest age groups. Rates of emergency department visits with diagnosed COVID also have continued to drop, as have new admissions, and both are nearing their 2021 lows, according to the Centers for Disease Control and Prevention.
New vaccinations in children under age 5 years were up slightly for the most recent week (Oct. 13-19), but total uptake for that age group is only 7.1% for an initial dose and 2.9% for full vaccination. Among children aged 5-11 years, 38.7% have received at least one dose and 31.6% have completed the primary series, with corresponding figures of 71.2% and 60.9% for those aged 12-17, the CDC said on its COVID Data Tracker.
Despite the low overall numbers, though, the youngest children are, in one respect, punching above their weight when it comes to vaccinations. In the 2 weeks from Oct. 6 to Oct. 19, children under 5 years of age, who represent 5.9% of the U.S. population, received 9.2% of the initial vaccine doses administered. Children aged 5-11 years, who represent 8.7% of the total population, got just 4.2% of all first doses over those same 2 weeks, while 12- to 17-year-olds, who make up 7.6% of the population, got 3.4% of the vaccine doses, the CDC reported.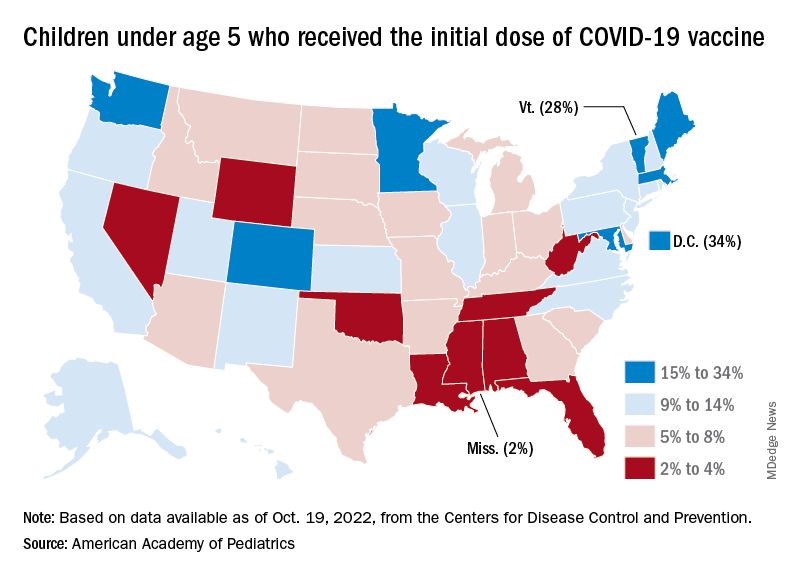
On the vaccine-approval front, the Food and Drug Administration recently announced that the new bivalent COVID-19 vaccines are now included in the emergency use authorizations for children who have completed primary or booster vaccination. The Moderna vaccine is authorized as a single-dose booster for children as young as 6 years and the Pfizer-BioNTech vaccine can be given as a single booster dose in children as young as 5 years, the FDA said.
“These bivalent COVID-19 vaccines include an mRNA component of the original strain to provide an immune response that is broadly protective against COVID-19 and an mRNA component in common between the omicron variant BA.4 and BA.5 lineages,” the FDA said.
With the third autumn of the COVID era now upon us, the discussion has turned again to a possible influenza/COVID twindemic, as well as the new-for-2022 influenza/COVID/respiratory syncytial virus tripledemic. It appears, however, that COVID may have missed the memo.
For the sixth time in the last 7 weeks, the number of new COVID cases in children fell, with just under 23,000 reported during the week of Oct. 14-20, according to the American Academy of Pediatrics and the Children’s Hospital Association. That is the lowest weekly count so far this year, and the lowest since early July of 2021, just as the Delta surge was starting. New pediatric cases had dipped to 8,500, the lowest for any week during the pandemic, a couple of weeks before that, the AAP/CHA data show.
Weekly cases have fallen by almost 75% since over 90,000 were reported for the week of Aug. 26 to Sept. 1, even as children have returned to school and vaccine uptake remains slow in the youngest age groups. Rates of emergency department visits with diagnosed COVID also have continued to drop, as have new admissions, and both are nearing their 2021 lows, according to the Centers for Disease Control and Prevention.
New vaccinations in children under age 5 years were up slightly for the most recent week (Oct. 13-19), but total uptake for that age group is only 7.1% for an initial dose and 2.9% for full vaccination. Among children aged 5-11 years, 38.7% have received at least one dose and 31.6% have completed the primary series, with corresponding figures of 71.2% and 60.9% for those aged 12-17, the CDC said on its COVID Data Tracker.
Despite the low overall numbers, though, the youngest children are, in one respect, punching above their weight when it comes to vaccinations. In the 2 weeks from Oct. 6 to Oct. 19, children under 5 years of age, who represent 5.9% of the U.S. population, received 9.2% of the initial vaccine doses administered. Children aged 5-11 years, who represent 8.7% of the total population, got just 4.2% of all first doses over those same 2 weeks, while 12- to 17-year-olds, who make up 7.6% of the population, got 3.4% of the vaccine doses, the CDC reported.
On the vaccine-approval front, the Food and Drug Administration recently announced that the new bivalent COVID-19 vaccines are now included in the emergency use authorizations for children who have completed primary or booster vaccination. The Moderna vaccine is authorized as a single-dose booster for children as young as 6 years and the Pfizer-BioNTech vaccine can be given as a single booster dose in children as young as 5 years, the FDA said.
“These bivalent COVID-19 vaccines include an mRNA component of the original strain to provide an immune response that is broadly protective against COVID-19 and an mRNA component in common between the omicron variant BA.4 and BA.5 lineages,” the FDA said.
With the third autumn of the COVID era now upon us, the discussion has turned again to a possible influenza/COVID twindemic, as well as the new-for-2022 influenza/COVID/respiratory syncytial virus tripledemic. It appears, however, that COVID may have missed the memo.
For the sixth time in the last 7 weeks, the number of new COVID cases in children fell, with just under 23,000 reported during the week of Oct. 14-20, according to the American Academy of Pediatrics and the Children’s Hospital Association. That is the lowest weekly count so far this year, and the lowest since early July of 2021, just as the Delta surge was starting. New pediatric cases had dipped to 8,500, the lowest for any week during the pandemic, a couple of weeks before that, the AAP/CHA data show.
Weekly cases have fallen by almost 75% since over 90,000 were reported for the week of Aug. 26 to Sept. 1, even as children have returned to school and vaccine uptake remains slow in the youngest age groups. Rates of emergency department visits with diagnosed COVID also have continued to drop, as have new admissions, and both are nearing their 2021 lows, according to the Centers for Disease Control and Prevention.
New vaccinations in children under age 5 years were up slightly for the most recent week (Oct. 13-19), but total uptake for that age group is only 7.1% for an initial dose and 2.9% for full vaccination. Among children aged 5-11 years, 38.7% have received at least one dose and 31.6% have completed the primary series, with corresponding figures of 71.2% and 60.9% for those aged 12-17, the CDC said on its COVID Data Tracker.
Despite the low overall numbers, though, the youngest children are, in one respect, punching above their weight when it comes to vaccinations. In the 2 weeks from Oct. 6 to Oct. 19, children under 5 years of age, who represent 5.9% of the U.S. population, received 9.2% of the initial vaccine doses administered. Children aged 5-11 years, who represent 8.7% of the total population, got just 4.2% of all first doses over those same 2 weeks, while 12- to 17-year-olds, who make up 7.6% of the population, got 3.4% of the vaccine doses, the CDC reported.
On the vaccine-approval front, the Food and Drug Administration recently announced that the new bivalent COVID-19 vaccines are now included in the emergency use authorizations for children who have completed primary or booster vaccination. The Moderna vaccine is authorized as a single-dose booster for children as young as 6 years and the Pfizer-BioNTech vaccine can be given as a single booster dose in children as young as 5 years, the FDA said.
“These bivalent COVID-19 vaccines include an mRNA component of the original strain to provide an immune response that is broadly protective against COVID-19 and an mRNA component in common between the omicron variant BA.4 and BA.5 lineages,” the FDA said.
Time to ditch clarithromycin for H. pylori?
Rates of resistance to clarithromycin among Helicobacter pylori isolates in the United States and Europe are high enough to warrant discontinuation of empiric use of proton pump inhibitor (PPI)–based triple therapy that includes the antibiotic in these regions, a new study has found.
Overall, 22.2% of participants were resistant to clarithromycin – a rate that is above the currently recommended threshold of 15% or higher for avoidance of PPI-based triple therapy that includes clarithromycin.
, study investigator William Chey, MD, professor and chief, Division of Gastroenterology and Hepatology, Michigan Medicine, Ann Arbor, said in an interview.
Judith Kim, MD, a gastroenterologist at NYU Langone Health and clinical instructor of medicine at NYU Grossman School of Medicine, who wasn’t involved in the study, agrees.
“The use of PPI-based triple therapy is still common practice despite recent recommendations to avoid clarithromycin in areas with high resistance rates,” Dr. Kim told this news organization.
“This study shows that multiple parts of the United States and Europe have high resistance rates,” rendering clarithromycin-based regimens “more likely to ineffectively eradicate H pylori,” Dr. Kim said.
The study was published online in The American Journal of Gastroenterology.
Better options now available
Guidelines advise against the use of PPI-based triple regimens with clarithromycin for H. pylori infection in areas where resistance is 15% or higher or for patients who have previously received macrolides. However, up-to-date information on H. pylori antimicrobial resistance patterns is limited, especially in the United States.
Dr. Chey and colleagues assessed resistance rates to antibiotics commonly used to treat H. pylori in isolates from 907 adults with the infection in the United States and Europe. They included four U.S. subregions and five participating European countries.
In all U.S. subregions and European countries, clarithromycin resistance rates were above 15% except possibly in the United Kingdom, where the sample size was too small to provide a reliable estimate.
Three-quarters of the clarithromycin-resistant isolates were also resistant to metronidazole.
The study also found that, overall, 1.2% of patients had isolates that were resistant to amoxicillin, and 69.2% had isolates resistant to metronidazole. Resistance patterns were similar in the United States and Europe; metronidazole resistance was the most common (50%-79% of isolates), and amoxicillin was the least common (≤ 5%).
“Overall, these data provide robust evidence to support a shift away from the default empiric prescription of triple combinations containing a PPI and clarithromycin for H. pylori infection in the United States and Europe,” the study team writes.
The high prevalence of resistance, including dual resistance, highlights the need for antibiotic stewardship and resistance surveillance, as well as novel treatment strategies for H. pylori infection, they add.
Last spring, as previously reported, the United States Food and Drug Administration approved two vonoprazan-based treatments for H. pylori: Voquezna Triple Pak (vonoprazan, amoxicillin, clarithromycin) and Voquezna Dual Pak (vonoprazan, amoxicillin), both from Phathom Pharmaceuticals.
“Vonoprazan-based treatment may be superior to standard PPI triple therapy for clarithromycin-resistant infections based on prior studies and is a potential good option,” Dr. Kim said.
Still, she added, she “would most likely first recommend regimens that do not have clarithromycin, such as bismuth quadruple therapy.”
Study’s importance
Because the study drew upon the largest dataset to date on U.S. resistance rates, it should be used to more precisely guide first-line therapy decisions, said Richard Peek, Jr., MD, professor of medicine and director of gastroenterology at Vanderbilt University Medical Center, Nashville, Tenn.
“To date, there has been a dearth of information in the United States regarding H. pylori resistance rates, which has often led to the use of ineffective empiric therapies and inappropriate exposure to antibiotics,” Dr. Peek, who wasn’t involved in the study, told this news organization.
“These data are particularly exciting when viewed within the context of new genomic sequencing tests that can determine H. pylori resistance patterns using DNA isolated from the stomach or the stool,” he said.
Dr. Peek agreed that the recent approval of vonoprazan-based therapies “adds another regimen to the therapeutic armamentarium available for eradicating H. pylori, and its value seems to be particularly beneficial for eradication failures.”
The research was funded by Phathom Pharmaceuticals. Dr. Chey is a board member of the American College of Gastroenterology, GI on Demand, the International Foundation of Functional GI Disorders, and the Rome Foundation. He has received compensation as a consultant from AbbVie, Alfasigma, Allakos, Alnylam, Bayer, BioAmerica, Cosmo, Intrinsic Medicine, Ironwood Pharmaceuticals, QOL Medical, Nestle, Phathom Pharmaceuticals, RedHill Biopharma, Salix/Valeant, Takeda, Urovant, and Vibrant; grant/research support from BioAmerica, Commonwealth Diagnostics International, QOL Medical, Salix, and Vibrant; owns stock/stock options in GI on Demand and Modify Health; and owns patents relating to methods and kits for identifying food sensitivities and intolerances, digital manometry, and a rectal expulsion device. Dr. Peek and Dr. Kim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rates of resistance to clarithromycin among Helicobacter pylori isolates in the United States and Europe are high enough to warrant discontinuation of empiric use of proton pump inhibitor (PPI)–based triple therapy that includes the antibiotic in these regions, a new study has found.
Overall, 22.2% of participants were resistant to clarithromycin – a rate that is above the currently recommended threshold of 15% or higher for avoidance of PPI-based triple therapy that includes clarithromycin.
, study investigator William Chey, MD, professor and chief, Division of Gastroenterology and Hepatology, Michigan Medicine, Ann Arbor, said in an interview.
Judith Kim, MD, a gastroenterologist at NYU Langone Health and clinical instructor of medicine at NYU Grossman School of Medicine, who wasn’t involved in the study, agrees.
“The use of PPI-based triple therapy is still common practice despite recent recommendations to avoid clarithromycin in areas with high resistance rates,” Dr. Kim told this news organization.
“This study shows that multiple parts of the United States and Europe have high resistance rates,” rendering clarithromycin-based regimens “more likely to ineffectively eradicate H pylori,” Dr. Kim said.
The study was published online in The American Journal of Gastroenterology.
Better options now available
Guidelines advise against the use of PPI-based triple regimens with clarithromycin for H. pylori infection in areas where resistance is 15% or higher or for patients who have previously received macrolides. However, up-to-date information on H. pylori antimicrobial resistance patterns is limited, especially in the United States.
Dr. Chey and colleagues assessed resistance rates to antibiotics commonly used to treat H. pylori in isolates from 907 adults with the infection in the United States and Europe. They included four U.S. subregions and five participating European countries.
In all U.S. subregions and European countries, clarithromycin resistance rates were above 15% except possibly in the United Kingdom, where the sample size was too small to provide a reliable estimate.
Three-quarters of the clarithromycin-resistant isolates were also resistant to metronidazole.
The study also found that, overall, 1.2% of patients had isolates that were resistant to amoxicillin, and 69.2% had isolates resistant to metronidazole. Resistance patterns were similar in the United States and Europe; metronidazole resistance was the most common (50%-79% of isolates), and amoxicillin was the least common (≤ 5%).
“Overall, these data provide robust evidence to support a shift away from the default empiric prescription of triple combinations containing a PPI and clarithromycin for H. pylori infection in the United States and Europe,” the study team writes.
The high prevalence of resistance, including dual resistance, highlights the need for antibiotic stewardship and resistance surveillance, as well as novel treatment strategies for H. pylori infection, they add.
Last spring, as previously reported, the United States Food and Drug Administration approved two vonoprazan-based treatments for H. pylori: Voquezna Triple Pak (vonoprazan, amoxicillin, clarithromycin) and Voquezna Dual Pak (vonoprazan, amoxicillin), both from Phathom Pharmaceuticals.
“Vonoprazan-based treatment may be superior to standard PPI triple therapy for clarithromycin-resistant infections based on prior studies and is a potential good option,” Dr. Kim said.
Still, she added, she “would most likely first recommend regimens that do not have clarithromycin, such as bismuth quadruple therapy.”
Study’s importance
Because the study drew upon the largest dataset to date on U.S. resistance rates, it should be used to more precisely guide first-line therapy decisions, said Richard Peek, Jr., MD, professor of medicine and director of gastroenterology at Vanderbilt University Medical Center, Nashville, Tenn.
“To date, there has been a dearth of information in the United States regarding H. pylori resistance rates, which has often led to the use of ineffective empiric therapies and inappropriate exposure to antibiotics,” Dr. Peek, who wasn’t involved in the study, told this news organization.
“These data are particularly exciting when viewed within the context of new genomic sequencing tests that can determine H. pylori resistance patterns using DNA isolated from the stomach or the stool,” he said.
Dr. Peek agreed that the recent approval of vonoprazan-based therapies “adds another regimen to the therapeutic armamentarium available for eradicating H. pylori, and its value seems to be particularly beneficial for eradication failures.”
The research was funded by Phathom Pharmaceuticals. Dr. Chey is a board member of the American College of Gastroenterology, GI on Demand, the International Foundation of Functional GI Disorders, and the Rome Foundation. He has received compensation as a consultant from AbbVie, Alfasigma, Allakos, Alnylam, Bayer, BioAmerica, Cosmo, Intrinsic Medicine, Ironwood Pharmaceuticals, QOL Medical, Nestle, Phathom Pharmaceuticals, RedHill Biopharma, Salix/Valeant, Takeda, Urovant, and Vibrant; grant/research support from BioAmerica, Commonwealth Diagnostics International, QOL Medical, Salix, and Vibrant; owns stock/stock options in GI on Demand and Modify Health; and owns patents relating to methods and kits for identifying food sensitivities and intolerances, digital manometry, and a rectal expulsion device. Dr. Peek and Dr. Kim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Rates of resistance to clarithromycin among Helicobacter pylori isolates in the United States and Europe are high enough to warrant discontinuation of empiric use of proton pump inhibitor (PPI)–based triple therapy that includes the antibiotic in these regions, a new study has found.
Overall, 22.2% of participants were resistant to clarithromycin – a rate that is above the currently recommended threshold of 15% or higher for avoidance of PPI-based triple therapy that includes clarithromycin.
, study investigator William Chey, MD, professor and chief, Division of Gastroenterology and Hepatology, Michigan Medicine, Ann Arbor, said in an interview.
Judith Kim, MD, a gastroenterologist at NYU Langone Health and clinical instructor of medicine at NYU Grossman School of Medicine, who wasn’t involved in the study, agrees.
“The use of PPI-based triple therapy is still common practice despite recent recommendations to avoid clarithromycin in areas with high resistance rates,” Dr. Kim told this news organization.
“This study shows that multiple parts of the United States and Europe have high resistance rates,” rendering clarithromycin-based regimens “more likely to ineffectively eradicate H pylori,” Dr. Kim said.
The study was published online in The American Journal of Gastroenterology.
Better options now available
Guidelines advise against the use of PPI-based triple regimens with clarithromycin for H. pylori infection in areas where resistance is 15% or higher or for patients who have previously received macrolides. However, up-to-date information on H. pylori antimicrobial resistance patterns is limited, especially in the United States.
Dr. Chey and colleagues assessed resistance rates to antibiotics commonly used to treat H. pylori in isolates from 907 adults with the infection in the United States and Europe. They included four U.S. subregions and five participating European countries.
In all U.S. subregions and European countries, clarithromycin resistance rates were above 15% except possibly in the United Kingdom, where the sample size was too small to provide a reliable estimate.
Three-quarters of the clarithromycin-resistant isolates were also resistant to metronidazole.
The study also found that, overall, 1.2% of patients had isolates that were resistant to amoxicillin, and 69.2% had isolates resistant to metronidazole. Resistance patterns were similar in the United States and Europe; metronidazole resistance was the most common (50%-79% of isolates), and amoxicillin was the least common (≤ 5%).
“Overall, these data provide robust evidence to support a shift away from the default empiric prescription of triple combinations containing a PPI and clarithromycin for H. pylori infection in the United States and Europe,” the study team writes.
The high prevalence of resistance, including dual resistance, highlights the need for antibiotic stewardship and resistance surveillance, as well as novel treatment strategies for H. pylori infection, they add.
Last spring, as previously reported, the United States Food and Drug Administration approved two vonoprazan-based treatments for H. pylori: Voquezna Triple Pak (vonoprazan, amoxicillin, clarithromycin) and Voquezna Dual Pak (vonoprazan, amoxicillin), both from Phathom Pharmaceuticals.
“Vonoprazan-based treatment may be superior to standard PPI triple therapy for clarithromycin-resistant infections based on prior studies and is a potential good option,” Dr. Kim said.
Still, she added, she “would most likely first recommend regimens that do not have clarithromycin, such as bismuth quadruple therapy.”
Study’s importance
Because the study drew upon the largest dataset to date on U.S. resistance rates, it should be used to more precisely guide first-line therapy decisions, said Richard Peek, Jr., MD, professor of medicine and director of gastroenterology at Vanderbilt University Medical Center, Nashville, Tenn.
“To date, there has been a dearth of information in the United States regarding H. pylori resistance rates, which has often led to the use of ineffective empiric therapies and inappropriate exposure to antibiotics,” Dr. Peek, who wasn’t involved in the study, told this news organization.
“These data are particularly exciting when viewed within the context of new genomic sequencing tests that can determine H. pylori resistance patterns using DNA isolated from the stomach or the stool,” he said.
Dr. Peek agreed that the recent approval of vonoprazan-based therapies “adds another regimen to the therapeutic armamentarium available for eradicating H. pylori, and its value seems to be particularly beneficial for eradication failures.”
The research was funded by Phathom Pharmaceuticals. Dr. Chey is a board member of the American College of Gastroenterology, GI on Demand, the International Foundation of Functional GI Disorders, and the Rome Foundation. He has received compensation as a consultant from AbbVie, Alfasigma, Allakos, Alnylam, Bayer, BioAmerica, Cosmo, Intrinsic Medicine, Ironwood Pharmaceuticals, QOL Medical, Nestle, Phathom Pharmaceuticals, RedHill Biopharma, Salix/Valeant, Takeda, Urovant, and Vibrant; grant/research support from BioAmerica, Commonwealth Diagnostics International, QOL Medical, Salix, and Vibrant; owns stock/stock options in GI on Demand and Modify Health; and owns patents relating to methods and kits for identifying food sensitivities and intolerances, digital manometry, and a rectal expulsion device. Dr. Peek and Dr. Kim report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Life expectancy 6.3 years shorter for Black MSM with HIV
WASHINGTON –
Lead author Katherine Rich, MPH, a student at Harvard Medical School, Boston, presented the data during an annual scientific meeting on infectious diseases.
“Substantial disparities in care exist between Black and White MSM here in the United States,” she said. “The 2030 goals of the EHE [Ending the HIV Epidemic in the U.S. initiative] won’t be met until HIV-related disparities are reduced.”
Using modeling, the team was able to measure both the gaps and the potential of interventions to address those gaps.
- The team found that improving engagement in care had the largest benefit in narrowing the gap. Improving engagement and retention in care, they write, would result in a gain of 1.4 life-years for Black MSM and 1 year for White MSM.
- Annual testing would add 0.6 life-years for Black MSM and 0.3 life-years for White MSM, compared with standard care.
- In simulating viral suppression, the model-predicted gain would be 1.1 years for Black MSM and 0.3 for White.
Furthermore, a combination of annual testing, 95% engagement in care, and 95% virologic suppression would add 3.4 years for Black MSM (more than double the increase in life-years for any one intervention) and 1.6 years for their White counterparts, the research suggests.
The researchers projected life expectancy from age 15 to be 52.2 years for Black MSM (or 67.2 years old) and 58.5 years from age 15 for White MSM (or 73.5 years old), a difference of 6.3 years.
Kathleen McManus, MD, assistant professor of medicine in infectious diseases and international health at the University of Virginia, Charlottesville, said in an interview that the projected gap in years of life should be a call to action. Dr. McManus was not involved with the study.
Life expectancy gap ‘alarming’
“It is alarming that with current usual HIV care Black MSM with HIV have 6.3 fewer years of life expectancy than White MSM,” she said. “Black MSM having lower retention in care and a lower rate of viral suppression than White MSM demonstrates that there is a problem with our current health care delivery to Black MSM.”
With qualitative, community-engaged research, she said, “we need to ask the Black MSM community what care innovations they need – and then we need clinics and organizations to make the identified changes.”
Researchers used the validated CEPAC (Cost-Effectiveness of Preventing AIDS Complications) microsimulation HIV model to project life expectancy. Using data from the United States Centers for Disease Control and Prevention, they estimated the average age at HIV infection to be 26.8 years for Black MSM and 35 years for White MSM.
They estimated the proportion of time that MSM with diagnosed HIV are retained in care to be 75.2% for Black MSM and 80.6% for White MSM. They calculated the proportion who achieve virologic suppression to be 82% for Black MSM and 91.2% for White MSM.
Senior author Emily P. Hyle, MD, associate professor of medicine at Harvard and infectious diseases physician at Massachusetts General Hospital, both in Boston, said in a press conference before the presentation that strategies to narrow the gap will look different by region.
“Our study highlights that if you can find effective interventions, the effect can be incredibly large. These are very large differences in life-years and life expectancies,” she said.
Ms. Rich gave an example of promising interventions by pointing to work by study coauthor Aima Ahonkhai, MD, MPH, assistant professor of medicine at Vanderbilt University, Nashville, Tenn., who has received federal funding to pursue research on whether preventive care outreach in barbershops can improve prevention for Black men with HIV.
Ms. Rich noted that the modeling has limitations in that it focused on health outcomes and did not simulate transmissions. Results also reflect national data and not local HIV care continuums, which, she acknowledged, differ substantially.
Dr. McManus and the study authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON –
Lead author Katherine Rich, MPH, a student at Harvard Medical School, Boston, presented the data during an annual scientific meeting on infectious diseases.
“Substantial disparities in care exist between Black and White MSM here in the United States,” she said. “The 2030 goals of the EHE [Ending the HIV Epidemic in the U.S. initiative] won’t be met until HIV-related disparities are reduced.”
Using modeling, the team was able to measure both the gaps and the potential of interventions to address those gaps.
- The team found that improving engagement in care had the largest benefit in narrowing the gap. Improving engagement and retention in care, they write, would result in a gain of 1.4 life-years for Black MSM and 1 year for White MSM.
- Annual testing would add 0.6 life-years for Black MSM and 0.3 life-years for White MSM, compared with standard care.
- In simulating viral suppression, the model-predicted gain would be 1.1 years for Black MSM and 0.3 for White.
Furthermore, a combination of annual testing, 95% engagement in care, and 95% virologic suppression would add 3.4 years for Black MSM (more than double the increase in life-years for any one intervention) and 1.6 years for their White counterparts, the research suggests.
The researchers projected life expectancy from age 15 to be 52.2 years for Black MSM (or 67.2 years old) and 58.5 years from age 15 for White MSM (or 73.5 years old), a difference of 6.3 years.
Kathleen McManus, MD, assistant professor of medicine in infectious diseases and international health at the University of Virginia, Charlottesville, said in an interview that the projected gap in years of life should be a call to action. Dr. McManus was not involved with the study.
Life expectancy gap ‘alarming’
“It is alarming that with current usual HIV care Black MSM with HIV have 6.3 fewer years of life expectancy than White MSM,” she said. “Black MSM having lower retention in care and a lower rate of viral suppression than White MSM demonstrates that there is a problem with our current health care delivery to Black MSM.”
With qualitative, community-engaged research, she said, “we need to ask the Black MSM community what care innovations they need – and then we need clinics and organizations to make the identified changes.”
Researchers used the validated CEPAC (Cost-Effectiveness of Preventing AIDS Complications) microsimulation HIV model to project life expectancy. Using data from the United States Centers for Disease Control and Prevention, they estimated the average age at HIV infection to be 26.8 years for Black MSM and 35 years for White MSM.
They estimated the proportion of time that MSM with diagnosed HIV are retained in care to be 75.2% for Black MSM and 80.6% for White MSM. They calculated the proportion who achieve virologic suppression to be 82% for Black MSM and 91.2% for White MSM.
Senior author Emily P. Hyle, MD, associate professor of medicine at Harvard and infectious diseases physician at Massachusetts General Hospital, both in Boston, said in a press conference before the presentation that strategies to narrow the gap will look different by region.
“Our study highlights that if you can find effective interventions, the effect can be incredibly large. These are very large differences in life-years and life expectancies,” she said.
Ms. Rich gave an example of promising interventions by pointing to work by study coauthor Aima Ahonkhai, MD, MPH, assistant professor of medicine at Vanderbilt University, Nashville, Tenn., who has received federal funding to pursue research on whether preventive care outreach in barbershops can improve prevention for Black men with HIV.
Ms. Rich noted that the modeling has limitations in that it focused on health outcomes and did not simulate transmissions. Results also reflect national data and not local HIV care continuums, which, she acknowledged, differ substantially.
Dr. McManus and the study authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON –
Lead author Katherine Rich, MPH, a student at Harvard Medical School, Boston, presented the data during an annual scientific meeting on infectious diseases.
“Substantial disparities in care exist between Black and White MSM here in the United States,” she said. “The 2030 goals of the EHE [Ending the HIV Epidemic in the U.S. initiative] won’t be met until HIV-related disparities are reduced.”
Using modeling, the team was able to measure both the gaps and the potential of interventions to address those gaps.
- The team found that improving engagement in care had the largest benefit in narrowing the gap. Improving engagement and retention in care, they write, would result in a gain of 1.4 life-years for Black MSM and 1 year for White MSM.
- Annual testing would add 0.6 life-years for Black MSM and 0.3 life-years for White MSM, compared with standard care.
- In simulating viral suppression, the model-predicted gain would be 1.1 years for Black MSM and 0.3 for White.
Furthermore, a combination of annual testing, 95% engagement in care, and 95% virologic suppression would add 3.4 years for Black MSM (more than double the increase in life-years for any one intervention) and 1.6 years for their White counterparts, the research suggests.
The researchers projected life expectancy from age 15 to be 52.2 years for Black MSM (or 67.2 years old) and 58.5 years from age 15 for White MSM (or 73.5 years old), a difference of 6.3 years.
Kathleen McManus, MD, assistant professor of medicine in infectious diseases and international health at the University of Virginia, Charlottesville, said in an interview that the projected gap in years of life should be a call to action. Dr. McManus was not involved with the study.
Life expectancy gap ‘alarming’
“It is alarming that with current usual HIV care Black MSM with HIV have 6.3 fewer years of life expectancy than White MSM,” she said. “Black MSM having lower retention in care and a lower rate of viral suppression than White MSM demonstrates that there is a problem with our current health care delivery to Black MSM.”
With qualitative, community-engaged research, she said, “we need to ask the Black MSM community what care innovations they need – and then we need clinics and organizations to make the identified changes.”
Researchers used the validated CEPAC (Cost-Effectiveness of Preventing AIDS Complications) microsimulation HIV model to project life expectancy. Using data from the United States Centers for Disease Control and Prevention, they estimated the average age at HIV infection to be 26.8 years for Black MSM and 35 years for White MSM.
They estimated the proportion of time that MSM with diagnosed HIV are retained in care to be 75.2% for Black MSM and 80.6% for White MSM. They calculated the proportion who achieve virologic suppression to be 82% for Black MSM and 91.2% for White MSM.
Senior author Emily P. Hyle, MD, associate professor of medicine at Harvard and infectious diseases physician at Massachusetts General Hospital, both in Boston, said in a press conference before the presentation that strategies to narrow the gap will look different by region.
“Our study highlights that if you can find effective interventions, the effect can be incredibly large. These are very large differences in life-years and life expectancies,” she said.
Ms. Rich gave an example of promising interventions by pointing to work by study coauthor Aima Ahonkhai, MD, MPH, assistant professor of medicine at Vanderbilt University, Nashville, Tenn., who has received federal funding to pursue research on whether preventive care outreach in barbershops can improve prevention for Black men with HIV.
Ms. Rich noted that the modeling has limitations in that it focused on health outcomes and did not simulate transmissions. Results also reflect national data and not local HIV care continuums, which, she acknowledged, differ substantially.
Dr. McManus and the study authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT IDWEEK 2022
Worse COVID outcomes seen with gout, particularly in women
People with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status, researchers suggest.
“We found that the risks of SARS-CoV-2 infection, 30-day hospitalization, and 30-day death among individuals with gout were higher than the general population irrespective of the vaccination status,” lead study author Dongxing Xie, MD, PhD, Xiangya Hospital, Central South University, Changsha, China, and his colleagues write in their large population study. “This finding informs individuals with gout, especially women, that additional measures, even after vaccination, should be considered in order to mitigate the risk of SARS-CoV-2 infection and its severe sequelae.”
People with gout, the most common inflammatory arthritis, often have other conditions that are linked to higher risk for SARS-CoV-2 infection and poor outcomes as well, including obesity, cardiovascular disease, and chronic kidney disease, the authors write. And elevated serum urate may contribute to inflammation and possible COVID-19 complications. But unlike in the case of diseases such as lupus and rheumatoid arthritis, little is known about SARS-CoV-2 infection risk among patients with gout.
As reported in Arthritis & Rheumatology, Dr. Xie and his research team used the Health Improvement Network ([THIN], now called IQVIA Medical Research Database) repository of medical conditions, demographics, and other details of around 17 million people in the United Kingdom to estimate the risk for SARS-CoV-2 infection, hospitalization, and death in people with gout. They compared those outcomes with outcomes of people without gout and compared outcomes of vaccinated vs. nonvaccinated participants.
From December 2020 through October 2021, the researchers investigated the risk for SARS-CoV-2 breakthrough infection in vaccinated people between age 18 and 90 years who had gout and were hospitalized within 30 days after the infection diagnosis or who died within 30 days after the diagnosis. They compared these outcomes with the outcomes of people in the general population without gout after COVID-19 vaccination. They also compared the risk for SARS-CoV-2 infection and its severe outcomes between individuals with gout and the general population among unvaccinated people.
They weighted these comparisons on the basis of age, sex, body mass index, socioeconomic deprivation index score, region, and number of previous COVID-19 tests in one model. A more fully adjusted model also weighted the comparisons for lifestyle factors, comorbidities, medications, and healthcare utilization.
The vaccinated cohort consisted of 54,576 people with gout and 1,336,377 without gout from the general population. The unvaccinated cohort included 61,111 individuals with gout and 1,697,168 individuals without gout from the general population.
Women more likely to be hospitalized and die
The risk for breakthrough infection in the vaccinated cohort was significantly higher among people with gout than among those without gout in the general population, particularly for men, who had hazard ratios (HRs) ranging from 1.22 with a fully adjusted exposure score to 1.30 with a partially adjusted score, but this was not seen in women. The overall incidence of breakthrough infection per 1,000 person-months for these groups was 4.68 with gout vs. 3.76 without gout.
The researchers showed a similar pattern of a higher rate of hospitalizations for people with gout vs. without (0.42/1,000 person-months vs. 0.28); in this case, women had higher risks than did men, with HRs for women ranging from 1.55 with a fully adjusted exposure score to 1.91 with a partially adjusted score, compared with 1.22 and 1.43 for men, respectively.
People with gout had significantly higher mortality than did those without (0.06/1,000 person-months vs. 0.04), but the risk for death was only higher for women, with HRs calculated to be 2.23 in fully adjusted exposure scores and 3.01 in partially adjusted scores.
These same comparisons in the unvaccinated cohort all went in the same direction as did those in the vaccinated cohort but showed higher rates for infection (8.69/1,000 person-months vs. 6.89), hospitalization (2.57/1,000 person-months vs. 1.71), and death (0.65/1,000 person-months vs. 0.53). Similar sex-specific links between gout and risks for SARS-CoV-2 infection, hospitalization, and death were seen in the unvaccinated cohort.
Patients with gout and COVID-19 need close monitoring
Four experts who were not involved in the study encourage greater attention to the needs of patients with gout.
Pamela B. Davis, MD, PhD, research professor at Case Western Reserve University, Cleveland, told this news organization, “This study brings to attention yet another potentially vulnerable group for physicians to monitor closely if they are infected with SARS-CoV-2.
“It is not clear why women with gout are more vulnerable, but fewer women than men were in the cohort with gout, and the confidence intervals for the results in women were, in general, larger,” she said.
“The authors suggest that women with gout tend to be older and have more comorbidities than men with gout,” Dr. Davis added. “The excess risk diminishes when the model is fully adjusted for comorbidities, such as obesity, hypertension, or heart disease, suggesting that already-known antecedents of infection severity account for a great deal of the excess risk.”
Kevin D. Deane, MD, PhD, associate professor of medicine and chair in rheumatology research at the University of Colorado at Denver, Aurora, advises physicians to keep in mind other conditions linked with increased risk for severe COVID-19, including advanced age; heart, lung, or kidney problems; and autoimmune diseases.
“I would be very cautious about the finding that there was not a difference in outcomes in individuals with gout based on vaccination status,” he cautioned, urging clinicians to “still strongly recommend vaccines according to guidelines.”
Sarah E. Waldman, MD, associate clinical professor of infectious diseases at UC Davis Health in Sacramento, Calif., called the study interesting but not surprising.
“The reason for increased risk for COVID-19 infection among those with gout may have to do with their underlying inflammatory state. Additional research needs to be done on this topic.
“Retrospective population-based cohort studies can be difficult to interpret due to biases,” she added. Associations identified in this type of study do not determine causation.
“As the researchers noted, those with gout tend to have additional comorbidities as well as advanced age,” she said. “They may also seek medical care more often and be tested for SARS-CoV-2 more frequently.”
Dr. Waldman advises clinicians to counsel patients with gout about their potential increased infection risk and ways they can protect themselves, including COVID-19 vaccinations.
“The strong association between gout and COVID-19 infection could involve coexisting conditions such as diabetes, hypertension, cardiovascular disease, and chronic kidney disease,” Dr. Aung added.
Earlier studies show links between gout and severe COVID-19 outcomes
Lead author Kanon Jatuworapruk, MD, PhD, of Thammasat University in Pathumthani, Thailand, and his colleagues investigated characteristics and outcomes of people with gout who were hospitalized for COVID-19 between March 2020 and October 2021, using data from the COVID-19 Global Rheumatology Alliance registry.
“This cohort of people with gout and COVID-19 who were hospitalized had high frequencies of ventilatory support and death,” the authors write in ACR Open Rheumatology . “This suggests that patients with gout who were hospitalized for COVID-19 may be at risk of poor outcomes, perhaps related to known risk factors for poor outcomes, such as age and presence of comorbidity.”
In their study, the average age of the 163 patients was 63 years, and 85% were men. Most lived in the Western Pacific Region and North America, and 46% had two or more comorbidities, most commonly hypertension, cardiovascular disease, diabetes, chronic kidney disease, and obesity. The researchers found that:
- Sixty-eight percent of the cohort required supplemental oxygen or ventilatory support during hospitalization.
- Sixteen percent of deaths were related to COVID-19, with 73% of deaths occurring in people with two or more comorbidities.
Ruth K. Topless, assistant research fellow in the department of biochemistry at the University of Otago in Dunedin, New Zealand, is the lead author on a study she and her colleagues are conducting using the UK Biobank databases of 459,837 participants in the United Kingdom, including 15,871 people with gout, through April 6, 2021, to investigate whether gout is a risk factor for diagnosis of COVID-19 and COVID-19–related death.
“Gout is a risk factor for COVID-19-related death in the UK Biobank cohort, with an increased risk in women with gout, which was driven by risk factors independent of the metabolic comorbidities of gout,” the researchers conclude in The Lancet Rheumatology.
In their study, gout was linked with COVID-19 diagnosis (odds ratio, 1.20; 95% confidence interval, 1.11-1.29) but not with risk for COVID-19–related death in the group of patients with COVID-19 (OR, 1.20; 95% CI, 0.96-1.51). In the entire cohort, gout was linked with COVID-19–related death (OR, 1.29; 95% CI, 1.06-1.56); women with gout were at increased risk for COVID-19–related death (OR, 1.98; 95% CI, 1.34-2.94), but men with gout were not (OR, 1.16; 95% CI, 0.93-1.45). The risk for COVID-19 diagnosis was significant in the nonvaccinated group (OR, 1.21; 95% CI, 1.11-1.30) but not in the vaccinated group (OR, 1.09; 95% CI, 0.65-1.85).
Editorial authors join in recommending further related research
In a commentary in The Lancet Rheumatology about the UK Biobank and other related research, Christoffer B. Nissen, MD, of University Hospital of Southern Denmark in Sonderborg, and his co-authors call the Topless and colleagues study “an elegantly conducted analysis of data from the UK Biobank supporting the hypothesis that gout needs attention in patients with COVID-19.”
Further studies are needed to investigate to what degree a diagnosis of gout is a risk factor for COVID-19 and whether treatment modifies the risk of a severe disease course,” they write. “However, in the interim, the results of this study could be considered when risk stratifying patients with gout in view of vaccination recommendations and early treatment interventions.”
Each of the three studies received grant funding. Several of the authors of the studies report financial involvements with pharmaceutical companies. All outside experts commented by email and report no relevant financial involvements.
A version of this article first appeared on Medscape.com.
People with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status, researchers suggest.
“We found that the risks of SARS-CoV-2 infection, 30-day hospitalization, and 30-day death among individuals with gout were higher than the general population irrespective of the vaccination status,” lead study author Dongxing Xie, MD, PhD, Xiangya Hospital, Central South University, Changsha, China, and his colleagues write in their large population study. “This finding informs individuals with gout, especially women, that additional measures, even after vaccination, should be considered in order to mitigate the risk of SARS-CoV-2 infection and its severe sequelae.”
People with gout, the most common inflammatory arthritis, often have other conditions that are linked to higher risk for SARS-CoV-2 infection and poor outcomes as well, including obesity, cardiovascular disease, and chronic kidney disease, the authors write. And elevated serum urate may contribute to inflammation and possible COVID-19 complications. But unlike in the case of diseases such as lupus and rheumatoid arthritis, little is known about SARS-CoV-2 infection risk among patients with gout.
As reported in Arthritis & Rheumatology, Dr. Xie and his research team used the Health Improvement Network ([THIN], now called IQVIA Medical Research Database) repository of medical conditions, demographics, and other details of around 17 million people in the United Kingdom to estimate the risk for SARS-CoV-2 infection, hospitalization, and death in people with gout. They compared those outcomes with outcomes of people without gout and compared outcomes of vaccinated vs. nonvaccinated participants.
From December 2020 through October 2021, the researchers investigated the risk for SARS-CoV-2 breakthrough infection in vaccinated people between age 18 and 90 years who had gout and were hospitalized within 30 days after the infection diagnosis or who died within 30 days after the diagnosis. They compared these outcomes with the outcomes of people in the general population without gout after COVID-19 vaccination. They also compared the risk for SARS-CoV-2 infection and its severe outcomes between individuals with gout and the general population among unvaccinated people.
They weighted these comparisons on the basis of age, sex, body mass index, socioeconomic deprivation index score, region, and number of previous COVID-19 tests in one model. A more fully adjusted model also weighted the comparisons for lifestyle factors, comorbidities, medications, and healthcare utilization.
The vaccinated cohort consisted of 54,576 people with gout and 1,336,377 without gout from the general population. The unvaccinated cohort included 61,111 individuals with gout and 1,697,168 individuals without gout from the general population.
Women more likely to be hospitalized and die
The risk for breakthrough infection in the vaccinated cohort was significantly higher among people with gout than among those without gout in the general population, particularly for men, who had hazard ratios (HRs) ranging from 1.22 with a fully adjusted exposure score to 1.30 with a partially adjusted score, but this was not seen in women. The overall incidence of breakthrough infection per 1,000 person-months for these groups was 4.68 with gout vs. 3.76 without gout.
The researchers showed a similar pattern of a higher rate of hospitalizations for people with gout vs. without (0.42/1,000 person-months vs. 0.28); in this case, women had higher risks than did men, with HRs for women ranging from 1.55 with a fully adjusted exposure score to 1.91 with a partially adjusted score, compared with 1.22 and 1.43 for men, respectively.
People with gout had significantly higher mortality than did those without (0.06/1,000 person-months vs. 0.04), but the risk for death was only higher for women, with HRs calculated to be 2.23 in fully adjusted exposure scores and 3.01 in partially adjusted scores.
These same comparisons in the unvaccinated cohort all went in the same direction as did those in the vaccinated cohort but showed higher rates for infection (8.69/1,000 person-months vs. 6.89), hospitalization (2.57/1,000 person-months vs. 1.71), and death (0.65/1,000 person-months vs. 0.53). Similar sex-specific links between gout and risks for SARS-CoV-2 infection, hospitalization, and death were seen in the unvaccinated cohort.
Patients with gout and COVID-19 need close monitoring
Four experts who were not involved in the study encourage greater attention to the needs of patients with gout.
Pamela B. Davis, MD, PhD, research professor at Case Western Reserve University, Cleveland, told this news organization, “This study brings to attention yet another potentially vulnerable group for physicians to monitor closely if they are infected with SARS-CoV-2.
“It is not clear why women with gout are more vulnerable, but fewer women than men were in the cohort with gout, and the confidence intervals for the results in women were, in general, larger,” she said.
“The authors suggest that women with gout tend to be older and have more comorbidities than men with gout,” Dr. Davis added. “The excess risk diminishes when the model is fully adjusted for comorbidities, such as obesity, hypertension, or heart disease, suggesting that already-known antecedents of infection severity account for a great deal of the excess risk.”
Kevin D. Deane, MD, PhD, associate professor of medicine and chair in rheumatology research at the University of Colorado at Denver, Aurora, advises physicians to keep in mind other conditions linked with increased risk for severe COVID-19, including advanced age; heart, lung, or kidney problems; and autoimmune diseases.
“I would be very cautious about the finding that there was not a difference in outcomes in individuals with gout based on vaccination status,” he cautioned, urging clinicians to “still strongly recommend vaccines according to guidelines.”
Sarah E. Waldman, MD, associate clinical professor of infectious diseases at UC Davis Health in Sacramento, Calif., called the study interesting but not surprising.
“The reason for increased risk for COVID-19 infection among those with gout may have to do with their underlying inflammatory state. Additional research needs to be done on this topic.
“Retrospective population-based cohort studies can be difficult to interpret due to biases,” she added. Associations identified in this type of study do not determine causation.
“As the researchers noted, those with gout tend to have additional comorbidities as well as advanced age,” she said. “They may also seek medical care more often and be tested for SARS-CoV-2 more frequently.”
Dr. Waldman advises clinicians to counsel patients with gout about their potential increased infection risk and ways they can protect themselves, including COVID-19 vaccinations.
“The strong association between gout and COVID-19 infection could involve coexisting conditions such as diabetes, hypertension, cardiovascular disease, and chronic kidney disease,” Dr. Aung added.
Earlier studies show links between gout and severe COVID-19 outcomes
Lead author Kanon Jatuworapruk, MD, PhD, of Thammasat University in Pathumthani, Thailand, and his colleagues investigated characteristics and outcomes of people with gout who were hospitalized for COVID-19 between March 2020 and October 2021, using data from the COVID-19 Global Rheumatology Alliance registry.
“This cohort of people with gout and COVID-19 who were hospitalized had high frequencies of ventilatory support and death,” the authors write in ACR Open Rheumatology . “This suggests that patients with gout who were hospitalized for COVID-19 may be at risk of poor outcomes, perhaps related to known risk factors for poor outcomes, such as age and presence of comorbidity.”
In their study, the average age of the 163 patients was 63 years, and 85% were men. Most lived in the Western Pacific Region and North America, and 46% had two or more comorbidities, most commonly hypertension, cardiovascular disease, diabetes, chronic kidney disease, and obesity. The researchers found that:
- Sixty-eight percent of the cohort required supplemental oxygen or ventilatory support during hospitalization.
- Sixteen percent of deaths were related to COVID-19, with 73% of deaths occurring in people with two or more comorbidities.
Ruth K. Topless, assistant research fellow in the department of biochemistry at the University of Otago in Dunedin, New Zealand, is the lead author on a study she and her colleagues are conducting using the UK Biobank databases of 459,837 participants in the United Kingdom, including 15,871 people with gout, through April 6, 2021, to investigate whether gout is a risk factor for diagnosis of COVID-19 and COVID-19–related death.
“Gout is a risk factor for COVID-19-related death in the UK Biobank cohort, with an increased risk in women with gout, which was driven by risk factors independent of the metabolic comorbidities of gout,” the researchers conclude in The Lancet Rheumatology.
In their study, gout was linked with COVID-19 diagnosis (odds ratio, 1.20; 95% confidence interval, 1.11-1.29) but not with risk for COVID-19–related death in the group of patients with COVID-19 (OR, 1.20; 95% CI, 0.96-1.51). In the entire cohort, gout was linked with COVID-19–related death (OR, 1.29; 95% CI, 1.06-1.56); women with gout were at increased risk for COVID-19–related death (OR, 1.98; 95% CI, 1.34-2.94), but men with gout were not (OR, 1.16; 95% CI, 0.93-1.45). The risk for COVID-19 diagnosis was significant in the nonvaccinated group (OR, 1.21; 95% CI, 1.11-1.30) but not in the vaccinated group (OR, 1.09; 95% CI, 0.65-1.85).
Editorial authors join in recommending further related research
In a commentary in The Lancet Rheumatology about the UK Biobank and other related research, Christoffer B. Nissen, MD, of University Hospital of Southern Denmark in Sonderborg, and his co-authors call the Topless and colleagues study “an elegantly conducted analysis of data from the UK Biobank supporting the hypothesis that gout needs attention in patients with COVID-19.”
Further studies are needed to investigate to what degree a diagnosis of gout is a risk factor for COVID-19 and whether treatment modifies the risk of a severe disease course,” they write. “However, in the interim, the results of this study could be considered when risk stratifying patients with gout in view of vaccination recommendations and early treatment interventions.”
Each of the three studies received grant funding. Several of the authors of the studies report financial involvements with pharmaceutical companies. All outside experts commented by email and report no relevant financial involvements.
A version of this article first appeared on Medscape.com.
People with gout, especially women, appear to be at higher risk for poor COVID-19 outcomes, including hospitalization and death, regardless of COVID-19 vaccination status, researchers suggest.
“We found that the risks of SARS-CoV-2 infection, 30-day hospitalization, and 30-day death among individuals with gout were higher than the general population irrespective of the vaccination status,” lead study author Dongxing Xie, MD, PhD, Xiangya Hospital, Central South University, Changsha, China, and his colleagues write in their large population study. “This finding informs individuals with gout, especially women, that additional measures, even after vaccination, should be considered in order to mitigate the risk of SARS-CoV-2 infection and its severe sequelae.”
People with gout, the most common inflammatory arthritis, often have other conditions that are linked to higher risk for SARS-CoV-2 infection and poor outcomes as well, including obesity, cardiovascular disease, and chronic kidney disease, the authors write. And elevated serum urate may contribute to inflammation and possible COVID-19 complications. But unlike in the case of diseases such as lupus and rheumatoid arthritis, little is known about SARS-CoV-2 infection risk among patients with gout.
As reported in Arthritis & Rheumatology, Dr. Xie and his research team used the Health Improvement Network ([THIN], now called IQVIA Medical Research Database) repository of medical conditions, demographics, and other details of around 17 million people in the United Kingdom to estimate the risk for SARS-CoV-2 infection, hospitalization, and death in people with gout. They compared those outcomes with outcomes of people without gout and compared outcomes of vaccinated vs. nonvaccinated participants.
From December 2020 through October 2021, the researchers investigated the risk for SARS-CoV-2 breakthrough infection in vaccinated people between age 18 and 90 years who had gout and were hospitalized within 30 days after the infection diagnosis or who died within 30 days after the diagnosis. They compared these outcomes with the outcomes of people in the general population without gout after COVID-19 vaccination. They also compared the risk for SARS-CoV-2 infection and its severe outcomes between individuals with gout and the general population among unvaccinated people.
They weighted these comparisons on the basis of age, sex, body mass index, socioeconomic deprivation index score, region, and number of previous COVID-19 tests in one model. A more fully adjusted model also weighted the comparisons for lifestyle factors, comorbidities, medications, and healthcare utilization.
The vaccinated cohort consisted of 54,576 people with gout and 1,336,377 without gout from the general population. The unvaccinated cohort included 61,111 individuals with gout and 1,697,168 individuals without gout from the general population.
Women more likely to be hospitalized and die
The risk for breakthrough infection in the vaccinated cohort was significantly higher among people with gout than among those without gout in the general population, particularly for men, who had hazard ratios (HRs) ranging from 1.22 with a fully adjusted exposure score to 1.30 with a partially adjusted score, but this was not seen in women. The overall incidence of breakthrough infection per 1,000 person-months for these groups was 4.68 with gout vs. 3.76 without gout.
The researchers showed a similar pattern of a higher rate of hospitalizations for people with gout vs. without (0.42/1,000 person-months vs. 0.28); in this case, women had higher risks than did men, with HRs for women ranging from 1.55 with a fully adjusted exposure score to 1.91 with a partially adjusted score, compared with 1.22 and 1.43 for men, respectively.
People with gout had significantly higher mortality than did those without (0.06/1,000 person-months vs. 0.04), but the risk for death was only higher for women, with HRs calculated to be 2.23 in fully adjusted exposure scores and 3.01 in partially adjusted scores.
These same comparisons in the unvaccinated cohort all went in the same direction as did those in the vaccinated cohort but showed higher rates for infection (8.69/1,000 person-months vs. 6.89), hospitalization (2.57/1,000 person-months vs. 1.71), and death (0.65/1,000 person-months vs. 0.53). Similar sex-specific links between gout and risks for SARS-CoV-2 infection, hospitalization, and death were seen in the unvaccinated cohort.
Patients with gout and COVID-19 need close monitoring
Four experts who were not involved in the study encourage greater attention to the needs of patients with gout.
Pamela B. Davis, MD, PhD, research professor at Case Western Reserve University, Cleveland, told this news organization, “This study brings to attention yet another potentially vulnerable group for physicians to monitor closely if they are infected with SARS-CoV-2.
“It is not clear why women with gout are more vulnerable, but fewer women than men were in the cohort with gout, and the confidence intervals for the results in women were, in general, larger,” she said.
“The authors suggest that women with gout tend to be older and have more comorbidities than men with gout,” Dr. Davis added. “The excess risk diminishes when the model is fully adjusted for comorbidities, such as obesity, hypertension, or heart disease, suggesting that already-known antecedents of infection severity account for a great deal of the excess risk.”
Kevin D. Deane, MD, PhD, associate professor of medicine and chair in rheumatology research at the University of Colorado at Denver, Aurora, advises physicians to keep in mind other conditions linked with increased risk for severe COVID-19, including advanced age; heart, lung, or kidney problems; and autoimmune diseases.
“I would be very cautious about the finding that there was not a difference in outcomes in individuals with gout based on vaccination status,” he cautioned, urging clinicians to “still strongly recommend vaccines according to guidelines.”
Sarah E. Waldman, MD, associate clinical professor of infectious diseases at UC Davis Health in Sacramento, Calif., called the study interesting but not surprising.
“The reason for increased risk for COVID-19 infection among those with gout may have to do with their underlying inflammatory state. Additional research needs to be done on this topic.
“Retrospective population-based cohort studies can be difficult to interpret due to biases,” she added. Associations identified in this type of study do not determine causation.
“As the researchers noted, those with gout tend to have additional comorbidities as well as advanced age,” she said. “They may also seek medical care more often and be tested for SARS-CoV-2 more frequently.”
Dr. Waldman advises clinicians to counsel patients with gout about their potential increased infection risk and ways they can protect themselves, including COVID-19 vaccinations.
“The strong association between gout and COVID-19 infection could involve coexisting conditions such as diabetes, hypertension, cardiovascular disease, and chronic kidney disease,” Dr. Aung added.
Earlier studies show links between gout and severe COVID-19 outcomes
Lead author Kanon Jatuworapruk, MD, PhD, of Thammasat University in Pathumthani, Thailand, and his colleagues investigated characteristics and outcomes of people with gout who were hospitalized for COVID-19 between March 2020 and October 2021, using data from the COVID-19 Global Rheumatology Alliance registry.
“This cohort of people with gout and COVID-19 who were hospitalized had high frequencies of ventilatory support and death,” the authors write in ACR Open Rheumatology . “This suggests that patients with gout who were hospitalized for COVID-19 may be at risk of poor outcomes, perhaps related to known risk factors for poor outcomes, such as age and presence of comorbidity.”
In their study, the average age of the 163 patients was 63 years, and 85% were men. Most lived in the Western Pacific Region and North America, and 46% had two or more comorbidities, most commonly hypertension, cardiovascular disease, diabetes, chronic kidney disease, and obesity. The researchers found that:
- Sixty-eight percent of the cohort required supplemental oxygen or ventilatory support during hospitalization.
- Sixteen percent of deaths were related to COVID-19, with 73% of deaths occurring in people with two or more comorbidities.
Ruth K. Topless, assistant research fellow in the department of biochemistry at the University of Otago in Dunedin, New Zealand, is the lead author on a study she and her colleagues are conducting using the UK Biobank databases of 459,837 participants in the United Kingdom, including 15,871 people with gout, through April 6, 2021, to investigate whether gout is a risk factor for diagnosis of COVID-19 and COVID-19–related death.
“Gout is a risk factor for COVID-19-related death in the UK Biobank cohort, with an increased risk in women with gout, which was driven by risk factors independent of the metabolic comorbidities of gout,” the researchers conclude in The Lancet Rheumatology.
In their study, gout was linked with COVID-19 diagnosis (odds ratio, 1.20; 95% confidence interval, 1.11-1.29) but not with risk for COVID-19–related death in the group of patients with COVID-19 (OR, 1.20; 95% CI, 0.96-1.51). In the entire cohort, gout was linked with COVID-19–related death (OR, 1.29; 95% CI, 1.06-1.56); women with gout were at increased risk for COVID-19–related death (OR, 1.98; 95% CI, 1.34-2.94), but men with gout were not (OR, 1.16; 95% CI, 0.93-1.45). The risk for COVID-19 diagnosis was significant in the nonvaccinated group (OR, 1.21; 95% CI, 1.11-1.30) but not in the vaccinated group (OR, 1.09; 95% CI, 0.65-1.85).
Editorial authors join in recommending further related research
In a commentary in The Lancet Rheumatology about the UK Biobank and other related research, Christoffer B. Nissen, MD, of University Hospital of Southern Denmark in Sonderborg, and his co-authors call the Topless and colleagues study “an elegantly conducted analysis of data from the UK Biobank supporting the hypothesis that gout needs attention in patients with COVID-19.”
Further studies are needed to investigate to what degree a diagnosis of gout is a risk factor for COVID-19 and whether treatment modifies the risk of a severe disease course,” they write. “However, in the interim, the results of this study could be considered when risk stratifying patients with gout in view of vaccination recommendations and early treatment interventions.”
Each of the three studies received grant funding. Several of the authors of the studies report financial involvements with pharmaceutical companies. All outside experts commented by email and report no relevant financial involvements.
A version of this article first appeared on Medscape.com.
Florida sees spike in deadly bacterial infections after Hurricane Ian
At least 4 people have died and 29 have been infected in Lee County after the hurricane, Florida health officials said in a news release.
Vibrio vulnificus bacteria is found in warm, brackish seawater, according to the Florida Department of Health. Anyone with open wounds or cuts should avoid standing water, floodwater, or seawater in the area, health officials said.
“Sewage spills in coastal waters, like those caused by Hurricane Ian, may increase bacteria levels,” the department advised in a news release. “People with open wounds, cuts, or scratches can be exposed to Vibrio vulnificus through direct contact with sea water or brackish water … Vibrio vulnificus can also cause disease in those who eat raw or undercooked oysters and shellfish.”
Infection can cause severe illness or death. Symptoms include fever, chills, decreased blood pressure, and blistering skin lesions. The bacteria does not spread person to person.
“If someone is concerned that they may have been exposed to Vibrio vulnificus and are experiencing the above symptoms, they should seek medical attention immediately,” officials said in the statement. “Individuals with wound infections should also seek care promptly.”
A version of this article first appeared on WebMD.com.
At least 4 people have died and 29 have been infected in Lee County after the hurricane, Florida health officials said in a news release.
Vibrio vulnificus bacteria is found in warm, brackish seawater, according to the Florida Department of Health. Anyone with open wounds or cuts should avoid standing water, floodwater, or seawater in the area, health officials said.
“Sewage spills in coastal waters, like those caused by Hurricane Ian, may increase bacteria levels,” the department advised in a news release. “People with open wounds, cuts, or scratches can be exposed to Vibrio vulnificus through direct contact with sea water or brackish water … Vibrio vulnificus can also cause disease in those who eat raw or undercooked oysters and shellfish.”
Infection can cause severe illness or death. Symptoms include fever, chills, decreased blood pressure, and blistering skin lesions. The bacteria does not spread person to person.
“If someone is concerned that they may have been exposed to Vibrio vulnificus and are experiencing the above symptoms, they should seek medical attention immediately,” officials said in the statement. “Individuals with wound infections should also seek care promptly.”
A version of this article first appeared on WebMD.com.
At least 4 people have died and 29 have been infected in Lee County after the hurricane, Florida health officials said in a news release.
Vibrio vulnificus bacteria is found in warm, brackish seawater, according to the Florida Department of Health. Anyone with open wounds or cuts should avoid standing water, floodwater, or seawater in the area, health officials said.
“Sewage spills in coastal waters, like those caused by Hurricane Ian, may increase bacteria levels,” the department advised in a news release. “People with open wounds, cuts, or scratches can be exposed to Vibrio vulnificus through direct contact with sea water or brackish water … Vibrio vulnificus can also cause disease in those who eat raw or undercooked oysters and shellfish.”
Infection can cause severe illness or death. Symptoms include fever, chills, decreased blood pressure, and blistering skin lesions. The bacteria does not spread person to person.
“If someone is concerned that they may have been exposed to Vibrio vulnificus and are experiencing the above symptoms, they should seek medical attention immediately,” officials said in the statement. “Individuals with wound infections should also seek care promptly.”
A version of this article first appeared on WebMD.com.
25 years of chickenpox vaccine: 91 million cases prevented
WASHINGTON – In the 25 years since the United States first launched its universal vaccinations program to protect children against chickenpox (varicella), the program has seen dramatic results, a data analysis indicates.
Results from 1995 – when universal vaccinations began – through 2019 were presented an annual scientific meeting on infectious diseases by Mona Marin, MD, a medical epidemiologist at the Centers for Disease Control and Prevention. Researchers analyzed published data and surveillance data reported to the CDC.
Deaths in under-20 group all but eliminated
Immunocompromised people or pregnant women and infants too young to be vaccinated also benefited from the children’s immunizations.
Each year, about 3.8 million cases, 10,500 hospitalizations, and 100 deaths from chickenpox are prevented in the United States thanks to the vaccination program, Dr. Marin said.
Over 25 years, 91 million cases, 238,000 hospitalizations, and 1,933 – 2,446 deaths have been prevented.
However, chickenpox is still widespread in most of the world.
U.S. first with universal program
The disease was thought to be of little consequence, Dr. Marin said, until the mid-1950s after the first cases of fatal varicella in immunocompromised children revealed the virus’ lethal potential.
The United States was the first country to introduce a universal vaccination program, Dr. Marin said. At the time, it was a one-dose vaccine. Within the first 10 years of the one-dose program, declines in chickenpox cases, hospitalization, and death rates went from 71% to 90% in comparison with previous years. But health care leaders wanted to close the remaining gap and target transmission in schools.
“It was a burden the United States considered unacceptable,” Dr. Marin said.
The leaders had seen the control of measles and polio and wanted the same for chickenpox.
Two-dose vaccines started in 2007
In 2007, the current two-dose policy was introduced. Administration of the first dose is recommended at age 12–15 months, and the second at age 4–6 years. Vaccination is required before the children enter kindergarten.
Coverage was high – at least 90% – the study authors reported; the two-dose program further reduced the number, size, and duration of outbreaks. Over the 25 years, the proportion of outbreaks with fewer than 10 cases increased from 28% to 73%.
By 2019, incidence had dropped by 97%, hospitalizations were down by 94%, and deaths had dropped by 97%.
The biggest decline was seen in those younger than 20, who were born during the vaccination program. That group saw declines of 97% to 99% in cases, hospitalizations, and incidence compared with rates before vaccinations.
Dr. Marin says one dose of the vaccine is moderately effective in preventing all varicella (82%) and is highly effective in preventing severe varicella (more than 97%).
“The second dose adds 10% or more improved protection against all varicella,” she said.
But there have been gains beyond medical advances.
Researchers calculated the economic benefit and found a net savings of $23 billion in medical costs (which also factored in lost wages from parents staying home to care for sick children).
Jaw-dropping results
Jeanne Marrazzo, MD, director of the division of infectious diseases at the University of Alabama at Birmingham, said in an interview that “as someone who is not a vaccinologist, the declines in deaths, let alone hospitalizations, were jaw-dropping. I hadn’t really seen a synthesis of the impact of one and two doses.”
She said the declines in zoster among young people were interesting. The big question, she said, is what impact this may have for shingles infections in middle-aged adults over time, since chickenpox and shingles are caused by the same virus.
Dr. Marrazzo also noted the economic savings calculations.
“It’s such a cheap intervention. It’s one of the best examples of how a simple vaccine can affect a cascade of events that are a result of chronic viral infection,” she said.
There are also messages for the current debates over COVID-19 vaccinations.
“For me, it is further evidence of the profound population-level effect safe vaccines can have,” Dr. Marrazzo said.
The authors and Dr. Marrazzo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON – In the 25 years since the United States first launched its universal vaccinations program to protect children against chickenpox (varicella), the program has seen dramatic results, a data analysis indicates.
Results from 1995 – when universal vaccinations began – through 2019 were presented an annual scientific meeting on infectious diseases by Mona Marin, MD, a medical epidemiologist at the Centers for Disease Control and Prevention. Researchers analyzed published data and surveillance data reported to the CDC.
Deaths in under-20 group all but eliminated
Immunocompromised people or pregnant women and infants too young to be vaccinated also benefited from the children’s immunizations.
Each year, about 3.8 million cases, 10,500 hospitalizations, and 100 deaths from chickenpox are prevented in the United States thanks to the vaccination program, Dr. Marin said.
Over 25 years, 91 million cases, 238,000 hospitalizations, and 1,933 – 2,446 deaths have been prevented.
However, chickenpox is still widespread in most of the world.
U.S. first with universal program
The disease was thought to be of little consequence, Dr. Marin said, until the mid-1950s after the first cases of fatal varicella in immunocompromised children revealed the virus’ lethal potential.
The United States was the first country to introduce a universal vaccination program, Dr. Marin said. At the time, it was a one-dose vaccine. Within the first 10 years of the one-dose program, declines in chickenpox cases, hospitalization, and death rates went from 71% to 90% in comparison with previous years. But health care leaders wanted to close the remaining gap and target transmission in schools.
“It was a burden the United States considered unacceptable,” Dr. Marin said.
The leaders had seen the control of measles and polio and wanted the same for chickenpox.
Two-dose vaccines started in 2007
In 2007, the current two-dose policy was introduced. Administration of the first dose is recommended at age 12–15 months, and the second at age 4–6 years. Vaccination is required before the children enter kindergarten.
Coverage was high – at least 90% – the study authors reported; the two-dose program further reduced the number, size, and duration of outbreaks. Over the 25 years, the proportion of outbreaks with fewer than 10 cases increased from 28% to 73%.
By 2019, incidence had dropped by 97%, hospitalizations were down by 94%, and deaths had dropped by 97%.
The biggest decline was seen in those younger than 20, who were born during the vaccination program. That group saw declines of 97% to 99% in cases, hospitalizations, and incidence compared with rates before vaccinations.
Dr. Marin says one dose of the vaccine is moderately effective in preventing all varicella (82%) and is highly effective in preventing severe varicella (more than 97%).
“The second dose adds 10% or more improved protection against all varicella,” she said.
But there have been gains beyond medical advances.
Researchers calculated the economic benefit and found a net savings of $23 billion in medical costs (which also factored in lost wages from parents staying home to care for sick children).
Jaw-dropping results
Jeanne Marrazzo, MD, director of the division of infectious diseases at the University of Alabama at Birmingham, said in an interview that “as someone who is not a vaccinologist, the declines in deaths, let alone hospitalizations, were jaw-dropping. I hadn’t really seen a synthesis of the impact of one and two doses.”
She said the declines in zoster among young people were interesting. The big question, she said, is what impact this may have for shingles infections in middle-aged adults over time, since chickenpox and shingles are caused by the same virus.
Dr. Marrazzo also noted the economic savings calculations.
“It’s such a cheap intervention. It’s one of the best examples of how a simple vaccine can affect a cascade of events that are a result of chronic viral infection,” she said.
There are also messages for the current debates over COVID-19 vaccinations.
“For me, it is further evidence of the profound population-level effect safe vaccines can have,” Dr. Marrazzo said.
The authors and Dr. Marrazzo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
WASHINGTON – In the 25 years since the United States first launched its universal vaccinations program to protect children against chickenpox (varicella), the program has seen dramatic results, a data analysis indicates.
Results from 1995 – when universal vaccinations began – through 2019 were presented an annual scientific meeting on infectious diseases by Mona Marin, MD, a medical epidemiologist at the Centers for Disease Control and Prevention. Researchers analyzed published data and surveillance data reported to the CDC.
Deaths in under-20 group all but eliminated
Immunocompromised people or pregnant women and infants too young to be vaccinated also benefited from the children’s immunizations.
Each year, about 3.8 million cases, 10,500 hospitalizations, and 100 deaths from chickenpox are prevented in the United States thanks to the vaccination program, Dr. Marin said.
Over 25 years, 91 million cases, 238,000 hospitalizations, and 1,933 – 2,446 deaths have been prevented.
However, chickenpox is still widespread in most of the world.
U.S. first with universal program
The disease was thought to be of little consequence, Dr. Marin said, until the mid-1950s after the first cases of fatal varicella in immunocompromised children revealed the virus’ lethal potential.
The United States was the first country to introduce a universal vaccination program, Dr. Marin said. At the time, it was a one-dose vaccine. Within the first 10 years of the one-dose program, declines in chickenpox cases, hospitalization, and death rates went from 71% to 90% in comparison with previous years. But health care leaders wanted to close the remaining gap and target transmission in schools.
“It was a burden the United States considered unacceptable,” Dr. Marin said.
The leaders had seen the control of measles and polio and wanted the same for chickenpox.
Two-dose vaccines started in 2007
In 2007, the current two-dose policy was introduced. Administration of the first dose is recommended at age 12–15 months, and the second at age 4–6 years. Vaccination is required before the children enter kindergarten.
Coverage was high – at least 90% – the study authors reported; the two-dose program further reduced the number, size, and duration of outbreaks. Over the 25 years, the proportion of outbreaks with fewer than 10 cases increased from 28% to 73%.
By 2019, incidence had dropped by 97%, hospitalizations were down by 94%, and deaths had dropped by 97%.
The biggest decline was seen in those younger than 20, who were born during the vaccination program. That group saw declines of 97% to 99% in cases, hospitalizations, and incidence compared with rates before vaccinations.
Dr. Marin says one dose of the vaccine is moderately effective in preventing all varicella (82%) and is highly effective in preventing severe varicella (more than 97%).
“The second dose adds 10% or more improved protection against all varicella,” she said.
But there have been gains beyond medical advances.
Researchers calculated the economic benefit and found a net savings of $23 billion in medical costs (which also factored in lost wages from parents staying home to care for sick children).
Jaw-dropping results
Jeanne Marrazzo, MD, director of the division of infectious diseases at the University of Alabama at Birmingham, said in an interview that “as someone who is not a vaccinologist, the declines in deaths, let alone hospitalizations, were jaw-dropping. I hadn’t really seen a synthesis of the impact of one and two doses.”
She said the declines in zoster among young people were interesting. The big question, she said, is what impact this may have for shingles infections in middle-aged adults over time, since chickenpox and shingles are caused by the same virus.
Dr. Marrazzo also noted the economic savings calculations.
“It’s such a cheap intervention. It’s one of the best examples of how a simple vaccine can affect a cascade of events that are a result of chronic viral infection,” she said.
There are also messages for the current debates over COVID-19 vaccinations.
“For me, it is further evidence of the profound population-level effect safe vaccines can have,” Dr. Marrazzo said.
The authors and Dr. Marrazzo report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT IDWEEK 2022
Monkeypox presentations, prevention strategies shifting
New areas of concern include transmissions among people experiencing homelessness and severe cases in immunocompromised persons.
Agam K. Rao, MD, with the Poxvirus and Rabies Branch of the Centers for Disease Control and Prevention, updated the global picture during an annual scientific meeting on infectious diseases: As of Oct. 14, the confirmed worldwide cases number 73,288, with more than one-third of them (27,317) in the United States. Case counts in the United States, however, have been decreasing since early August.
Cases have been most commonly found in men who have sex with men (MSM), though monkeypox has also been diagnosed in cisgender and transgender women, children, and men who do not report recent sex with other men.
Shift away from White men
Dr. Rao described a demographic shift in infections from White, non-Hispanic men early on to non-Hispanic Black and Hispanic men.
“There’s a lot of emphasis right now at CDC to try to understand these spreads, whether they are household [transmission] or another contact. We know that some of the women have had sexual contact with men who were diagnosed with monkeypox,” Dr. Rao said.
In children under age 12, direct skin-to-skin contact with household members seems to be the source, she said. In adolescents, as in adults, the main source seems to be male-male consensual sex.
“And just as in adults, Black and Hispanic children have been disproportionately affected,” she said.
No sustained spread outside MSM
Dr. Rao said that so far there has been no sustained spread detected beyond the MSM community. A CDC study of inmates in Cook County Jail in Chicago at the end of September, she noted, found no secondary cases.
However, health care workers are another group that was suspected to be at higher risk given close contact with patients, although there have been only three confirmed exposures. Sharps injuries from unroofed lesions are tied to some of those confirmed or suspected cases.
“We do not recommend unroofing lesions,” she said. “We’re getting very good samples from just rigorous swabbing of the lesions.”
She said that the CDC is also monitoring “a few hundred” cases, some of them severe, among people experiencing homelessness.
“We are working to try to understand the exposures that have occurred to those individuals and whether transmission has occurred person-to-person,” Dr. Rao said.
Severe cases among immunocompromised
Also of concern are people with compromised immune systems owing to advanced HIV or organ or stem cell transplants.
Among immunocompromised persons, Dr. Rao said, “we’re seeing large necrotic lesions affecting a large percentage of body surface, lesions that continue to develop over weeks.”
Boghuma Titanji, MD, PhD, MSc, a physician-scientist at Emory University in Atlanta, and an emerging-disease specialist, addressed the difference in presentations between immunocompromised and immunocompetent patients.
She said the main distinction is the extent of the lesions. Patients with AIDS and very low CD4 counts, for instance, are presenting with more lesions and have a longer course of illness.
Dr. Rao said in an interview, “It’s really important to understand someone’s immune status and understand whether they are severely immunocompromised. If there is a concern that a person has monkeypox, also testing for HIV concurrently may be important. It could be a missed opportunity to evaluate for it, especially given the fact that these can occur together.”
Assessing the size and appearance of the lesions is important to understanding whether patients could develop severe infection, she said.
Differences from past epidemics
Dr. Titanji said the current outbreak has some differences from historic outbreaks.
The incubation period, for instance, has tended to be shorter than in previous outbreaks – now 7-10 days, with a range of 5-14 days instead of a range of up to 21 days in previous outbreaks.
There are also more cases of presentations with only single lesions, which were infrequent in past epidemics, she said.
The scope of suspected cases has also broadened, with changing clinical features.
“We have expanded the clinical descriptions to include presentations that involve isolated rectal presentation – individuals presenting solely with rectal pain as the primary manifestation of monkeypox – or presenting with a sore throat as the only manifestation,” she said.
Expanding the case definition will help identify who should be tested.
“Monkeypox is an incredible clinical mimic,” Dr. Titanji said. “The rash can really take the form of a lot of the things we encounter on a regular basis in ID. It’s important to always have a low index of suspicion to test patients when they fit the right epidemiological profile.”
Vaccine strategy has evolved
Brett Petersen, MD, MPH, captain of the U.S. Public Health Service with the CDC, said that Jynneos, licensed by the U.S. Food and Drug Administration, continues to be the primary vaccine for monkeypox. However, the strategy has changed.
Whereas the initial vaccine strategy was to administer the vaccine after known exposure, the guidance now includes vaccinating after “both known and presumed exposures, as described in the eligible populations.”
It’s now been expanded even further to include preexposure inoculations for a wide group of people at greater risk, he explained.
Early data from the CDC indicate that the Jynneos vaccine is effective.
In a report updated in September, the CDC found that among 32 U.S. jurisdictions, monkeypox incidence was much higher among at-risk, unvaccinated people for whom vaccination is recommended than among those who got the Jynneos vaccine.
“Unvaccinated people had 14 times the risk of monkeypox disease compared to people who were vaccinated,” the CDC reported.
Asked about the end goal for monkeypox, Dr. Petersen said, “Our goal should be elimination. I think that is an achievable goal, but it will depend on a lot of factors and a lot of continued public health efforts.”
Dr. Rao, Dr. Titanji, and Dr. Petersen declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New areas of concern include transmissions among people experiencing homelessness and severe cases in immunocompromised persons.
Agam K. Rao, MD, with the Poxvirus and Rabies Branch of the Centers for Disease Control and Prevention, updated the global picture during an annual scientific meeting on infectious diseases: As of Oct. 14, the confirmed worldwide cases number 73,288, with more than one-third of them (27,317) in the United States. Case counts in the United States, however, have been decreasing since early August.
Cases have been most commonly found in men who have sex with men (MSM), though monkeypox has also been diagnosed in cisgender and transgender women, children, and men who do not report recent sex with other men.
Shift away from White men
Dr. Rao described a demographic shift in infections from White, non-Hispanic men early on to non-Hispanic Black and Hispanic men.
“There’s a lot of emphasis right now at CDC to try to understand these spreads, whether they are household [transmission] or another contact. We know that some of the women have had sexual contact with men who were diagnosed with monkeypox,” Dr. Rao said.
In children under age 12, direct skin-to-skin contact with household members seems to be the source, she said. In adolescents, as in adults, the main source seems to be male-male consensual sex.
“And just as in adults, Black and Hispanic children have been disproportionately affected,” she said.
No sustained spread outside MSM
Dr. Rao said that so far there has been no sustained spread detected beyond the MSM community. A CDC study of inmates in Cook County Jail in Chicago at the end of September, she noted, found no secondary cases.
However, health care workers are another group that was suspected to be at higher risk given close contact with patients, although there have been only three confirmed exposures. Sharps injuries from unroofed lesions are tied to some of those confirmed or suspected cases.
“We do not recommend unroofing lesions,” she said. “We’re getting very good samples from just rigorous swabbing of the lesions.”
She said that the CDC is also monitoring “a few hundred” cases, some of them severe, among people experiencing homelessness.
“We are working to try to understand the exposures that have occurred to those individuals and whether transmission has occurred person-to-person,” Dr. Rao said.
Severe cases among immunocompromised
Also of concern are people with compromised immune systems owing to advanced HIV or organ or stem cell transplants.
Among immunocompromised persons, Dr. Rao said, “we’re seeing large necrotic lesions affecting a large percentage of body surface, lesions that continue to develop over weeks.”
Boghuma Titanji, MD, PhD, MSc, a physician-scientist at Emory University in Atlanta, and an emerging-disease specialist, addressed the difference in presentations between immunocompromised and immunocompetent patients.
She said the main distinction is the extent of the lesions. Patients with AIDS and very low CD4 counts, for instance, are presenting with more lesions and have a longer course of illness.
Dr. Rao said in an interview, “It’s really important to understand someone’s immune status and understand whether they are severely immunocompromised. If there is a concern that a person has monkeypox, also testing for HIV concurrently may be important. It could be a missed opportunity to evaluate for it, especially given the fact that these can occur together.”
Assessing the size and appearance of the lesions is important to understanding whether patients could develop severe infection, she said.
Differences from past epidemics
Dr. Titanji said the current outbreak has some differences from historic outbreaks.
The incubation period, for instance, has tended to be shorter than in previous outbreaks – now 7-10 days, with a range of 5-14 days instead of a range of up to 21 days in previous outbreaks.
There are also more cases of presentations with only single lesions, which were infrequent in past epidemics, she said.
The scope of suspected cases has also broadened, with changing clinical features.
“We have expanded the clinical descriptions to include presentations that involve isolated rectal presentation – individuals presenting solely with rectal pain as the primary manifestation of monkeypox – or presenting with a sore throat as the only manifestation,” she said.
Expanding the case definition will help identify who should be tested.
“Monkeypox is an incredible clinical mimic,” Dr. Titanji said. “The rash can really take the form of a lot of the things we encounter on a regular basis in ID. It’s important to always have a low index of suspicion to test patients when they fit the right epidemiological profile.”
Vaccine strategy has evolved
Brett Petersen, MD, MPH, captain of the U.S. Public Health Service with the CDC, said that Jynneos, licensed by the U.S. Food and Drug Administration, continues to be the primary vaccine for monkeypox. However, the strategy has changed.
Whereas the initial vaccine strategy was to administer the vaccine after known exposure, the guidance now includes vaccinating after “both known and presumed exposures, as described in the eligible populations.”
It’s now been expanded even further to include preexposure inoculations for a wide group of people at greater risk, he explained.
Early data from the CDC indicate that the Jynneos vaccine is effective.
In a report updated in September, the CDC found that among 32 U.S. jurisdictions, monkeypox incidence was much higher among at-risk, unvaccinated people for whom vaccination is recommended than among those who got the Jynneos vaccine.
“Unvaccinated people had 14 times the risk of monkeypox disease compared to people who were vaccinated,” the CDC reported.
Asked about the end goal for monkeypox, Dr. Petersen said, “Our goal should be elimination. I think that is an achievable goal, but it will depend on a lot of factors and a lot of continued public health efforts.”
Dr. Rao, Dr. Titanji, and Dr. Petersen declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New areas of concern include transmissions among people experiencing homelessness and severe cases in immunocompromised persons.
Agam K. Rao, MD, with the Poxvirus and Rabies Branch of the Centers for Disease Control and Prevention, updated the global picture during an annual scientific meeting on infectious diseases: As of Oct. 14, the confirmed worldwide cases number 73,288, with more than one-third of them (27,317) in the United States. Case counts in the United States, however, have been decreasing since early August.
Cases have been most commonly found in men who have sex with men (MSM), though monkeypox has also been diagnosed in cisgender and transgender women, children, and men who do not report recent sex with other men.
Shift away from White men
Dr. Rao described a demographic shift in infections from White, non-Hispanic men early on to non-Hispanic Black and Hispanic men.
“There’s a lot of emphasis right now at CDC to try to understand these spreads, whether they are household [transmission] or another contact. We know that some of the women have had sexual contact with men who were diagnosed with monkeypox,” Dr. Rao said.
In children under age 12, direct skin-to-skin contact with household members seems to be the source, she said. In adolescents, as in adults, the main source seems to be male-male consensual sex.
“And just as in adults, Black and Hispanic children have been disproportionately affected,” she said.
No sustained spread outside MSM
Dr. Rao said that so far there has been no sustained spread detected beyond the MSM community. A CDC study of inmates in Cook County Jail in Chicago at the end of September, she noted, found no secondary cases.
However, health care workers are another group that was suspected to be at higher risk given close contact with patients, although there have been only three confirmed exposures. Sharps injuries from unroofed lesions are tied to some of those confirmed or suspected cases.
“We do not recommend unroofing lesions,” she said. “We’re getting very good samples from just rigorous swabbing of the lesions.”
She said that the CDC is also monitoring “a few hundred” cases, some of them severe, among people experiencing homelessness.
“We are working to try to understand the exposures that have occurred to those individuals and whether transmission has occurred person-to-person,” Dr. Rao said.
Severe cases among immunocompromised
Also of concern are people with compromised immune systems owing to advanced HIV or organ or stem cell transplants.
Among immunocompromised persons, Dr. Rao said, “we’re seeing large necrotic lesions affecting a large percentage of body surface, lesions that continue to develop over weeks.”
Boghuma Titanji, MD, PhD, MSc, a physician-scientist at Emory University in Atlanta, and an emerging-disease specialist, addressed the difference in presentations between immunocompromised and immunocompetent patients.
She said the main distinction is the extent of the lesions. Patients with AIDS and very low CD4 counts, for instance, are presenting with more lesions and have a longer course of illness.
Dr. Rao said in an interview, “It’s really important to understand someone’s immune status and understand whether they are severely immunocompromised. If there is a concern that a person has monkeypox, also testing for HIV concurrently may be important. It could be a missed opportunity to evaluate for it, especially given the fact that these can occur together.”
Assessing the size and appearance of the lesions is important to understanding whether patients could develop severe infection, she said.
Differences from past epidemics
Dr. Titanji said the current outbreak has some differences from historic outbreaks.
The incubation period, for instance, has tended to be shorter than in previous outbreaks – now 7-10 days, with a range of 5-14 days instead of a range of up to 21 days in previous outbreaks.
There are also more cases of presentations with only single lesions, which were infrequent in past epidemics, she said.
The scope of suspected cases has also broadened, with changing clinical features.
“We have expanded the clinical descriptions to include presentations that involve isolated rectal presentation – individuals presenting solely with rectal pain as the primary manifestation of monkeypox – or presenting with a sore throat as the only manifestation,” she said.
Expanding the case definition will help identify who should be tested.
“Monkeypox is an incredible clinical mimic,” Dr. Titanji said. “The rash can really take the form of a lot of the things we encounter on a regular basis in ID. It’s important to always have a low index of suspicion to test patients when they fit the right epidemiological profile.”
Vaccine strategy has evolved
Brett Petersen, MD, MPH, captain of the U.S. Public Health Service with the CDC, said that Jynneos, licensed by the U.S. Food and Drug Administration, continues to be the primary vaccine for monkeypox. However, the strategy has changed.
Whereas the initial vaccine strategy was to administer the vaccine after known exposure, the guidance now includes vaccinating after “both known and presumed exposures, as described in the eligible populations.”
It’s now been expanded even further to include preexposure inoculations for a wide group of people at greater risk, he explained.
Early data from the CDC indicate that the Jynneos vaccine is effective.
In a report updated in September, the CDC found that among 32 U.S. jurisdictions, monkeypox incidence was much higher among at-risk, unvaccinated people for whom vaccination is recommended than among those who got the Jynneos vaccine.
“Unvaccinated people had 14 times the risk of monkeypox disease compared to people who were vaccinated,” the CDC reported.
Asked about the end goal for monkeypox, Dr. Petersen said, “Our goal should be elimination. I think that is an achievable goal, but it will depend on a lot of factors and a lot of continued public health efforts.”
Dr. Rao, Dr. Titanji, and Dr. Petersen declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM IDWEEK 2022