User login
Inside Mercy’s mission to care for non-COVID patients in Los Angeles
When the hospital ship USNS Mercy departed San Diego’s Naval Station North Island on March 23, 2020, to support the Department of Defense efforts in Los Angeles during the coronavirus outbreak, Commander Erin Blevins remembers the crew’s excitement was palpable.
“We normally do partnerships abroad and respond to tsunamis and earthquakes,” said Cdr. Blevins, MD, a pediatric hematologist-oncologist who served as director of medical services for the mission. “This was a slight change in situation, but still disaster relief in the form of a pandemic. We switched our mindset to putting together the best experts for an infectious disease pandemic versus an earthquake disaster relief.”
A new mission
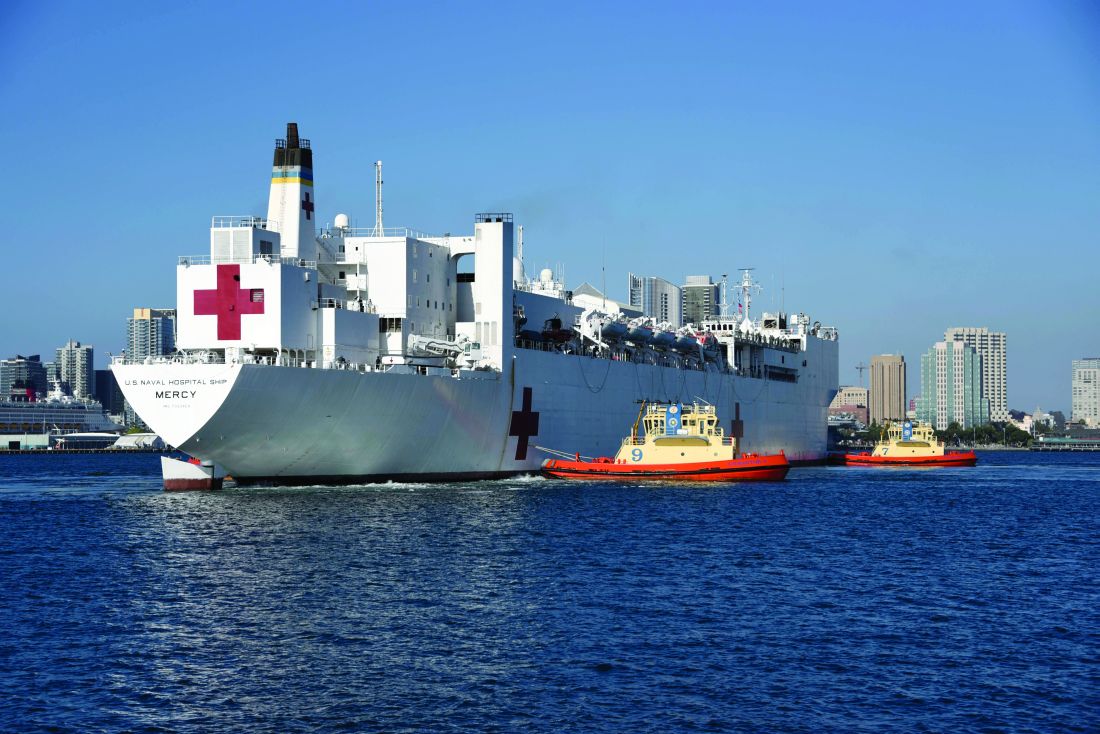
The 1,000-bed Mercy ship – a converted San Clemente–class oil tanker that was delivered in 1986 – spent nearly 50 days pier side in Los Angeles as a referral hospital for non–COVID-19 patients, so that clinicians at Los Angeles area hospitals could care for an anticipated surge of COVID-19 patients. “We went into it with expectations of, ‘We’ll treat as many patients as you need us to take,” Cdr. Blevins recalled. “I don’t even think Los Angeles [health officials] knew exactly where they were going to peak and what the need was going to be.”
Between March 29 and May 15, about 1,071 medical personnel aboard the Mercy cared for 77 patients with an average age of 53 years who were referred from 11 Los Angeles area hospitals. The physicians, nurses, and other medical support personnel were drawn from military treatment facilities across the country. “We had additional people join us as we scoped the mission to be more medically heavy and surgically light,” said Captain John Rotruck, MD, an anesthesiologist who is commanding officer of Mercy’s medical treatment facility. “We did adjust to make sure that we had the right staffing mix to meet the parameters that we were assigned. That was the crux of the change: a change in flavors of staffing to ensure that we focused on ICU and ward medical care as opposed to very heavy surgical care in support of a combat operation.”
About 10% of the team consisted of reservists who volunteered for the mission. “There’s no way you could have walked around the ship and known who was active duty and who was reservist,” said Capt. Rotruck, who was formerly chief of staff at Walter Reed National Military Medical Center, Bethesda, Md. “They worked together so well, and I think that marriage of active duty who are used to working in a military medical treatment facility – in our case, a Navy medical treatment facility – together with our reservist physician colleagues who work in civilian facilities around the country, was beneficial. It was a synergistic relationship. I think both sides walked away learning quite a bit from each other.”
Start with screening
All crew members underwent a temperature check and completed a health screening questionnaire: once before departing their home of record and again before boarding Mercy. Based on those results, crew members and medical staff were screened for COVID-19 and tested as needed in order to minimize the risk of an outbreak aboard the ship.
Fewer than 1% of crew members developed COVID-19 or tested positive for the virus during the mission, according to Capt. Rotruck. Affected individuals were isolated and quarantined. “All staff have recovered and are doing well,” he said.
Mercy personnel worked with local health officials to ensure that all patients transferred to the ship tested negative for COVID-19. Physicians aboard the Mercy then worked directly with the patients’ civilian physician to ensure a safe and thorough turnover process before the patients were transferred.
From basic medical to trauma care
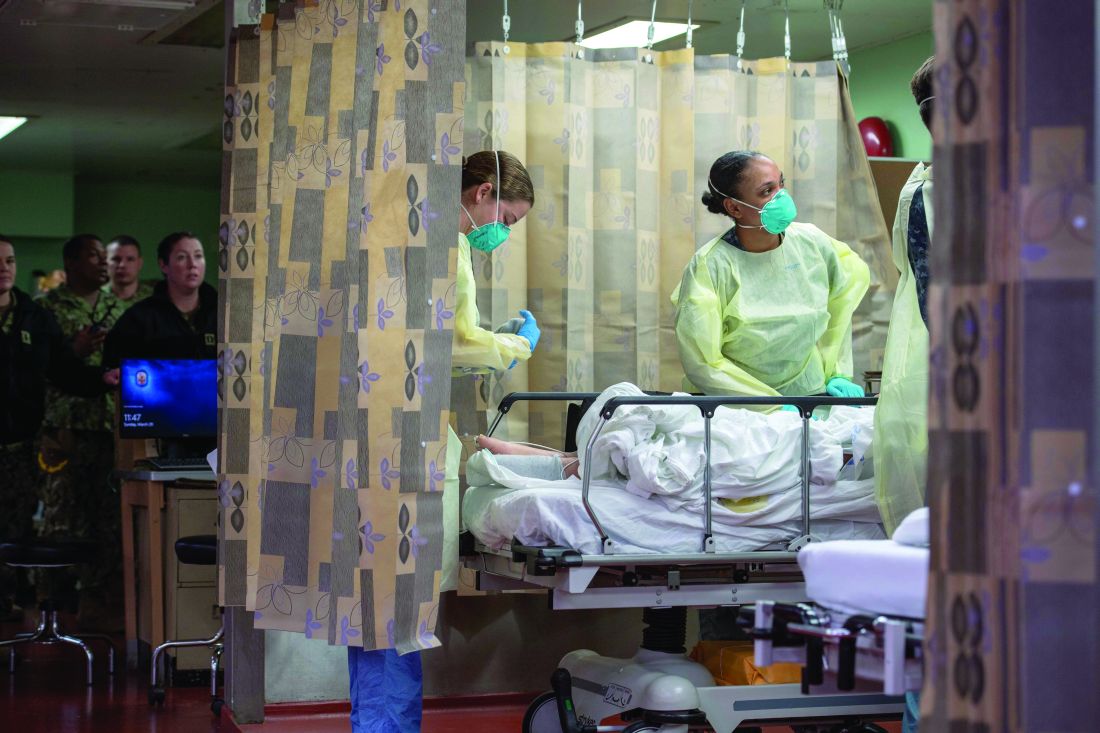
Care aboard the ship, which consists of open-bay medical wards, ranged from basic medical and surgical care to critical care and trauma. The most common procedures were cholecystectomies and orthopedic procedures, and the average length of stay was 4-5 days, according to Cdr. Blevins. Over the course of the mission, the medical professionals conducted 36 surgeries, 77 x-ray exams, 26 CT scans, and administered hundreds of ancillary studies ranging from routine labs to high-end x-rays and blood transfusion support.
“Within our ICU, we did have some end-of-life patients who ended up dying on our ship in comfort care,” Cdr. Blevins said. “Fortunately, we had a wonderful ICU team who had a great deal of experience with end-of-life care and were able to take care of these patients very comfortably and ensure good communication with family and loved ones during that time. In most instances we tried to make sure that people got to FaceTime or video chat with their loved one before they passed away.”
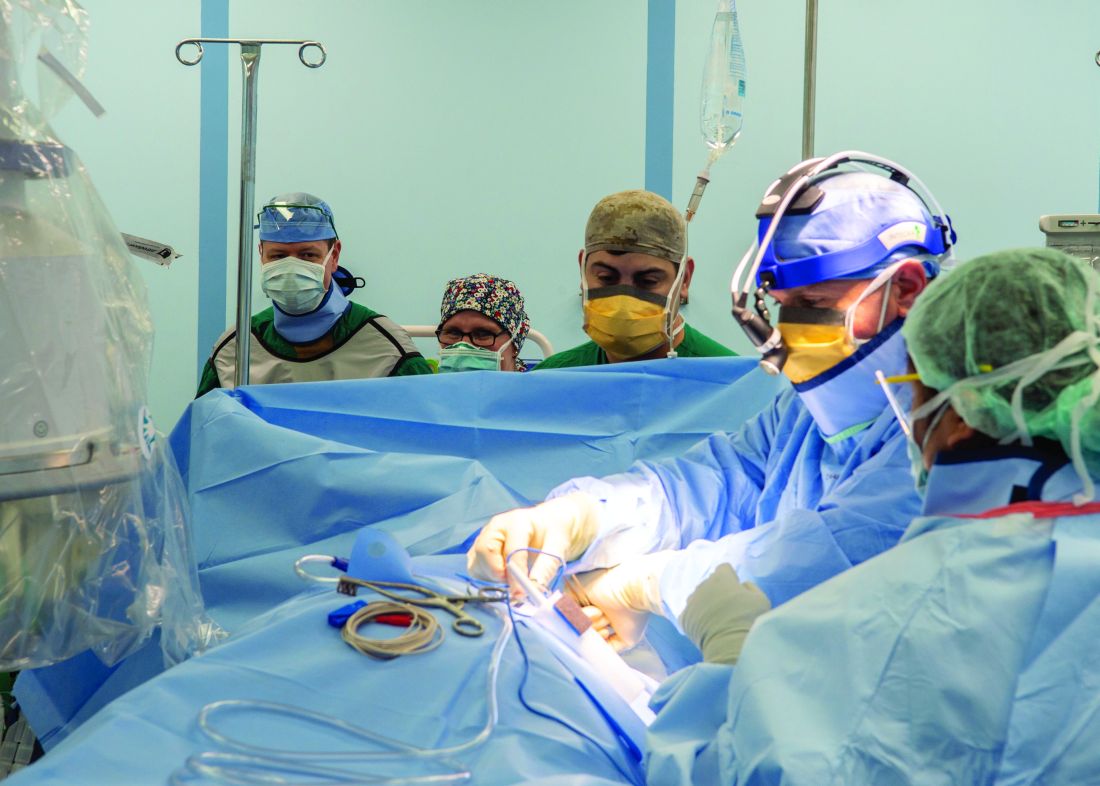
The Mercy, which includes 12 operating rooms, four x-ray units, and one CAT-scan unit, was not equipped to deliver pediatric or obstetrical care. Other unavailable services included psychiatry, oncology, cardiac and thoracic surgery, nuclear medicine, MRI, mammography, electrophysiology, cardiac catheterization, negative-pressure isolation, speech therapy, and occupational therapy.
Not your typical hospital experience
But for patients who did receive medical care aboard the Mercy – which made three 150-day deployments in recent years for the military-led humanitarian response known as Pacific Partnership in 2015, 2016, and 2018 – it was an experience that they are unlikely to forget.
“Every time a patient left the ship, our team on the ground surveyed them to see how their experience was and see what we could do to improve,” Cdr. Blevins said. “Across the board, they were all very appreciative of the medical care. We had a couple of veterans on board. They got [USNS Mercy] hats on their way out and seemed to very much enjoy a slightly different experience than they would get at a regular hospital.”
Capt. Rotruck added that the enthusiasm crew members had for supporting fellow Americans “really energized our team and really saturated that caring aspect of the people who interacted directly with patients,” he said. “It wasn’t just the physicians and nurses, but it was the staff delivering the food and coming to take blood samples and every other interaction that the patients had with our team. I think they really felt that enthusiasm for being there and supporting our neighbors in LA [Los Angeles].”
Crew life aboard the Mercy
Just as with any hospital on shore, personnel aboard the Mercy practiced preventive hygiene measures recommended by the Centers for Disease Control and Prevention to help prevent the spread of COVID-19, such as wearing cloth face masks, spacing out tables in the dining hall, closing indoor gyms, and devising creative ways to stay physically fit. Popular options included jogging around the perimeter of the ship and practicing yoga and calisthenics on the deck, “making sure you were physically distanced appropriately, and when you were done, putting your mask back on,” Cdr. Blevins said. Others supplemented their workouts with a pull-up bar on the deck. “In addition, we have a series of ramps that run on the starboard side of the ship that we can use for patient movement with litters on wheels or patient beds,” Capt. Rotruck said. “The uphill portion of those ramps represents a good workout opportunity as well.”
Downtime in an era of physical distancing also afforded crew members the opportunity to call or FaceTime with loved ones, watch streamed TV shows and movies, and work on their own professional development. Some continued with coursework for online degree programs offered by colleges and universities they were enrolled in, while some enlisted personnel used the time to complete the Navy Enlisted Warfare Qualification Programs Instruction, which issues the basic overarching requirements for the qualification and designation of all enlisted warfare programs.
“As you can imagine, people spend a lot of time learning how the ship works and how it integrates into larger naval forces and so forth,” Capt. Rotruck said. “Not just our ship but also other ships: their weapons systems and defense mechanisms and navigation systems. We had people spending a significant amount of time working on that. We had people complete their Enlisted Surface Warfare qualification while we were on the mission.”
End of the mission
Mercy returned to its home base in San Diego on May 15, but about 60 medical personnel stayed behind in Los Angeles to support Federal Emergency Management Agency (FEMA), state, and local health care professionals. Some worked at a site where clinicians provided care for COVID-19–positive patients who had been transferred from area skilled nursing facilities.
In addition, a team consisting of one nurse and five corpsmen “would go out to individual skilled nursing facilities and mainly conduct assessments and training, such as training in donning proper PPE [personal protective equipment] and determining what needs they had,” Capt. Rotruck said. “They met those needs if possible or [communicated with California officials] and let them know what the requirements were and what the needs were in that facility.” The assignment for those who stayed behind ended on May 31.
On the opposite coast, Mercy’s sister ship, USNS Comfort, arrived in New York Harbor from Norfolk, Va., on March 30 and spent 3½ weeks assisting area hospitals in the COVID-19 pandemic fight. A few days into the mission, Comfort’s internal spaces were reconfigured to create separate COVID-negative and COVID-positive sections. Medical teams aboard the ship cared for a total of 182 patients during the assignment.
Looking back on Mercy’s mission, Cdr. Blevins marveled at the sense of teamwork that unfolded. “We have quarterly training exercises with a core set of personnel, [and] we train getting ready for activation in 5 days,” she said. “All of that training kicks in and it comes to fruition in a mission like this. It was terrific to see a group of very disparate subject matter experts from all over the country come together with one purpose: which was to serve our own country during the pandemic.”
Capt. Rotruck pointed out that the experience enabled enlisted and nonenlisted physicians to maintain their skill sets during a time when military and civilian hospitals had stopped doing elective procedures and routine appointments. “The fact that those people were able to come on board the ship and continue to conduct their medical practice and maintain their skills and competencies in an environment that they weren’t quite used to is great,” he said. “Otherwise, some of those medical personnel would have been sitting idle, wherever they were from. This is the power of Navy medicine on behalf of our country.”
When the hospital ship USNS Mercy departed San Diego’s Naval Station North Island on March 23, 2020, to support the Department of Defense efforts in Los Angeles during the coronavirus outbreak, Commander Erin Blevins remembers the crew’s excitement was palpable.
“We normally do partnerships abroad and respond to tsunamis and earthquakes,” said Cdr. Blevins, MD, a pediatric hematologist-oncologist who served as director of medical services for the mission. “This was a slight change in situation, but still disaster relief in the form of a pandemic. We switched our mindset to putting together the best experts for an infectious disease pandemic versus an earthquake disaster relief.”
A new mission

The 1,000-bed Mercy ship – a converted San Clemente–class oil tanker that was delivered in 1986 – spent nearly 50 days pier side in Los Angeles as a referral hospital for non–COVID-19 patients, so that clinicians at Los Angeles area hospitals could care for an anticipated surge of COVID-19 patients. “We went into it with expectations of, ‘We’ll treat as many patients as you need us to take,” Cdr. Blevins recalled. “I don’t even think Los Angeles [health officials] knew exactly where they were going to peak and what the need was going to be.”
Between March 29 and May 15, about 1,071 medical personnel aboard the Mercy cared for 77 patients with an average age of 53 years who were referred from 11 Los Angeles area hospitals. The physicians, nurses, and other medical support personnel were drawn from military treatment facilities across the country. “We had additional people join us as we scoped the mission to be more medically heavy and surgically light,” said Captain John Rotruck, MD, an anesthesiologist who is commanding officer of Mercy’s medical treatment facility. “We did adjust to make sure that we had the right staffing mix to meet the parameters that we were assigned. That was the crux of the change: a change in flavors of staffing to ensure that we focused on ICU and ward medical care as opposed to very heavy surgical care in support of a combat operation.”
About 10% of the team consisted of reservists who volunteered for the mission. “There’s no way you could have walked around the ship and known who was active duty and who was reservist,” said Capt. Rotruck, who was formerly chief of staff at Walter Reed National Military Medical Center, Bethesda, Md. “They worked together so well, and I think that marriage of active duty who are used to working in a military medical treatment facility – in our case, a Navy medical treatment facility – together with our reservist physician colleagues who work in civilian facilities around the country, was beneficial. It was a synergistic relationship. I think both sides walked away learning quite a bit from each other.”
Start with screening
All crew members underwent a temperature check and completed a health screening questionnaire: once before departing their home of record and again before boarding Mercy. Based on those results, crew members and medical staff were screened for COVID-19 and tested as needed in order to minimize the risk of an outbreak aboard the ship.
Fewer than 1% of crew members developed COVID-19 or tested positive for the virus during the mission, according to Capt. Rotruck. Affected individuals were isolated and quarantined. “All staff have recovered and are doing well,” he said.
Mercy personnel worked with local health officials to ensure that all patients transferred to the ship tested negative for COVID-19. Physicians aboard the Mercy then worked directly with the patients’ civilian physician to ensure a safe and thorough turnover process before the patients were transferred.
From basic medical to trauma care

Care aboard the ship, which consists of open-bay medical wards, ranged from basic medical and surgical care to critical care and trauma. The most common procedures were cholecystectomies and orthopedic procedures, and the average length of stay was 4-5 days, according to Cdr. Blevins. Over the course of the mission, the medical professionals conducted 36 surgeries, 77 x-ray exams, 26 CT scans, and administered hundreds of ancillary studies ranging from routine labs to high-end x-rays and blood transfusion support.
“Within our ICU, we did have some end-of-life patients who ended up dying on our ship in comfort care,” Cdr. Blevins said. “Fortunately, we had a wonderful ICU team who had a great deal of experience with end-of-life care and were able to take care of these patients very comfortably and ensure good communication with family and loved ones during that time. In most instances we tried to make sure that people got to FaceTime or video chat with their loved one before they passed away.”

The Mercy, which includes 12 operating rooms, four x-ray units, and one CAT-scan unit, was not equipped to deliver pediatric or obstetrical care. Other unavailable services included psychiatry, oncology, cardiac and thoracic surgery, nuclear medicine, MRI, mammography, electrophysiology, cardiac catheterization, negative-pressure isolation, speech therapy, and occupational therapy.
Not your typical hospital experience
But for patients who did receive medical care aboard the Mercy – which made three 150-day deployments in recent years for the military-led humanitarian response known as Pacific Partnership in 2015, 2016, and 2018 – it was an experience that they are unlikely to forget.
“Every time a patient left the ship, our team on the ground surveyed them to see how their experience was and see what we could do to improve,” Cdr. Blevins said. “Across the board, they were all very appreciative of the medical care. We had a couple of veterans on board. They got [USNS Mercy] hats on their way out and seemed to very much enjoy a slightly different experience than they would get at a regular hospital.”
Capt. Rotruck added that the enthusiasm crew members had for supporting fellow Americans “really energized our team and really saturated that caring aspect of the people who interacted directly with patients,” he said. “It wasn’t just the physicians and nurses, but it was the staff delivering the food and coming to take blood samples and every other interaction that the patients had with our team. I think they really felt that enthusiasm for being there and supporting our neighbors in LA [Los Angeles].”
Crew life aboard the Mercy
Just as with any hospital on shore, personnel aboard the Mercy practiced preventive hygiene measures recommended by the Centers for Disease Control and Prevention to help prevent the spread of COVID-19, such as wearing cloth face masks, spacing out tables in the dining hall, closing indoor gyms, and devising creative ways to stay physically fit. Popular options included jogging around the perimeter of the ship and practicing yoga and calisthenics on the deck, “making sure you were physically distanced appropriately, and when you were done, putting your mask back on,” Cdr. Blevins said. Others supplemented their workouts with a pull-up bar on the deck. “In addition, we have a series of ramps that run on the starboard side of the ship that we can use for patient movement with litters on wheels or patient beds,” Capt. Rotruck said. “The uphill portion of those ramps represents a good workout opportunity as well.”
Downtime in an era of physical distancing also afforded crew members the opportunity to call or FaceTime with loved ones, watch streamed TV shows and movies, and work on their own professional development. Some continued with coursework for online degree programs offered by colleges and universities they were enrolled in, while some enlisted personnel used the time to complete the Navy Enlisted Warfare Qualification Programs Instruction, which issues the basic overarching requirements for the qualification and designation of all enlisted warfare programs.
“As you can imagine, people spend a lot of time learning how the ship works and how it integrates into larger naval forces and so forth,” Capt. Rotruck said. “Not just our ship but also other ships: their weapons systems and defense mechanisms and navigation systems. We had people spending a significant amount of time working on that. We had people complete their Enlisted Surface Warfare qualification while we were on the mission.”
End of the mission
Mercy returned to its home base in San Diego on May 15, but about 60 medical personnel stayed behind in Los Angeles to support Federal Emergency Management Agency (FEMA), state, and local health care professionals. Some worked at a site where clinicians provided care for COVID-19–positive patients who had been transferred from area skilled nursing facilities.
In addition, a team consisting of one nurse and five corpsmen “would go out to individual skilled nursing facilities and mainly conduct assessments and training, such as training in donning proper PPE [personal protective equipment] and determining what needs they had,” Capt. Rotruck said. “They met those needs if possible or [communicated with California officials] and let them know what the requirements were and what the needs were in that facility.” The assignment for those who stayed behind ended on May 31.
On the opposite coast, Mercy’s sister ship, USNS Comfort, arrived in New York Harbor from Norfolk, Va., on March 30 and spent 3½ weeks assisting area hospitals in the COVID-19 pandemic fight. A few days into the mission, Comfort’s internal spaces were reconfigured to create separate COVID-negative and COVID-positive sections. Medical teams aboard the ship cared for a total of 182 patients during the assignment.
Looking back on Mercy’s mission, Cdr. Blevins marveled at the sense of teamwork that unfolded. “We have quarterly training exercises with a core set of personnel, [and] we train getting ready for activation in 5 days,” she said. “All of that training kicks in and it comes to fruition in a mission like this. It was terrific to see a group of very disparate subject matter experts from all over the country come together with one purpose: which was to serve our own country during the pandemic.”
Capt. Rotruck pointed out that the experience enabled enlisted and nonenlisted physicians to maintain their skill sets during a time when military and civilian hospitals had stopped doing elective procedures and routine appointments. “The fact that those people were able to come on board the ship and continue to conduct their medical practice and maintain their skills and competencies in an environment that they weren’t quite used to is great,” he said. “Otherwise, some of those medical personnel would have been sitting idle, wherever they were from. This is the power of Navy medicine on behalf of our country.”
When the hospital ship USNS Mercy departed San Diego’s Naval Station North Island on March 23, 2020, to support the Department of Defense efforts in Los Angeles during the coronavirus outbreak, Commander Erin Blevins remembers the crew’s excitement was palpable.
“We normally do partnerships abroad and respond to tsunamis and earthquakes,” said Cdr. Blevins, MD, a pediatric hematologist-oncologist who served as director of medical services for the mission. “This was a slight change in situation, but still disaster relief in the form of a pandemic. We switched our mindset to putting together the best experts for an infectious disease pandemic versus an earthquake disaster relief.”
A new mission

The 1,000-bed Mercy ship – a converted San Clemente–class oil tanker that was delivered in 1986 – spent nearly 50 days pier side in Los Angeles as a referral hospital for non–COVID-19 patients, so that clinicians at Los Angeles area hospitals could care for an anticipated surge of COVID-19 patients. “We went into it with expectations of, ‘We’ll treat as many patients as you need us to take,” Cdr. Blevins recalled. “I don’t even think Los Angeles [health officials] knew exactly where they were going to peak and what the need was going to be.”
Between March 29 and May 15, about 1,071 medical personnel aboard the Mercy cared for 77 patients with an average age of 53 years who were referred from 11 Los Angeles area hospitals. The physicians, nurses, and other medical support personnel were drawn from military treatment facilities across the country. “We had additional people join us as we scoped the mission to be more medically heavy and surgically light,” said Captain John Rotruck, MD, an anesthesiologist who is commanding officer of Mercy’s medical treatment facility. “We did adjust to make sure that we had the right staffing mix to meet the parameters that we were assigned. That was the crux of the change: a change in flavors of staffing to ensure that we focused on ICU and ward medical care as opposed to very heavy surgical care in support of a combat operation.”
About 10% of the team consisted of reservists who volunteered for the mission. “There’s no way you could have walked around the ship and known who was active duty and who was reservist,” said Capt. Rotruck, who was formerly chief of staff at Walter Reed National Military Medical Center, Bethesda, Md. “They worked together so well, and I think that marriage of active duty who are used to working in a military medical treatment facility – in our case, a Navy medical treatment facility – together with our reservist physician colleagues who work in civilian facilities around the country, was beneficial. It was a synergistic relationship. I think both sides walked away learning quite a bit from each other.”
Start with screening
All crew members underwent a temperature check and completed a health screening questionnaire: once before departing their home of record and again before boarding Mercy. Based on those results, crew members and medical staff were screened for COVID-19 and tested as needed in order to minimize the risk of an outbreak aboard the ship.
Fewer than 1% of crew members developed COVID-19 or tested positive for the virus during the mission, according to Capt. Rotruck. Affected individuals were isolated and quarantined. “All staff have recovered and are doing well,” he said.
Mercy personnel worked with local health officials to ensure that all patients transferred to the ship tested negative for COVID-19. Physicians aboard the Mercy then worked directly with the patients’ civilian physician to ensure a safe and thorough turnover process before the patients were transferred.
From basic medical to trauma care

Care aboard the ship, which consists of open-bay medical wards, ranged from basic medical and surgical care to critical care and trauma. The most common procedures were cholecystectomies and orthopedic procedures, and the average length of stay was 4-5 days, according to Cdr. Blevins. Over the course of the mission, the medical professionals conducted 36 surgeries, 77 x-ray exams, 26 CT scans, and administered hundreds of ancillary studies ranging from routine labs to high-end x-rays and blood transfusion support.
“Within our ICU, we did have some end-of-life patients who ended up dying on our ship in comfort care,” Cdr. Blevins said. “Fortunately, we had a wonderful ICU team who had a great deal of experience with end-of-life care and were able to take care of these patients very comfortably and ensure good communication with family and loved ones during that time. In most instances we tried to make sure that people got to FaceTime or video chat with their loved one before they passed away.”

The Mercy, which includes 12 operating rooms, four x-ray units, and one CAT-scan unit, was not equipped to deliver pediatric or obstetrical care. Other unavailable services included psychiatry, oncology, cardiac and thoracic surgery, nuclear medicine, MRI, mammography, electrophysiology, cardiac catheterization, negative-pressure isolation, speech therapy, and occupational therapy.
Not your typical hospital experience
But for patients who did receive medical care aboard the Mercy – which made three 150-day deployments in recent years for the military-led humanitarian response known as Pacific Partnership in 2015, 2016, and 2018 – it was an experience that they are unlikely to forget.
“Every time a patient left the ship, our team on the ground surveyed them to see how their experience was and see what we could do to improve,” Cdr. Blevins said. “Across the board, they were all very appreciative of the medical care. We had a couple of veterans on board. They got [USNS Mercy] hats on their way out and seemed to very much enjoy a slightly different experience than they would get at a regular hospital.”
Capt. Rotruck added that the enthusiasm crew members had for supporting fellow Americans “really energized our team and really saturated that caring aspect of the people who interacted directly with patients,” he said. “It wasn’t just the physicians and nurses, but it was the staff delivering the food and coming to take blood samples and every other interaction that the patients had with our team. I think they really felt that enthusiasm for being there and supporting our neighbors in LA [Los Angeles].”
Crew life aboard the Mercy
Just as with any hospital on shore, personnel aboard the Mercy practiced preventive hygiene measures recommended by the Centers for Disease Control and Prevention to help prevent the spread of COVID-19, such as wearing cloth face masks, spacing out tables in the dining hall, closing indoor gyms, and devising creative ways to stay physically fit. Popular options included jogging around the perimeter of the ship and practicing yoga and calisthenics on the deck, “making sure you were physically distanced appropriately, and when you were done, putting your mask back on,” Cdr. Blevins said. Others supplemented their workouts with a pull-up bar on the deck. “In addition, we have a series of ramps that run on the starboard side of the ship that we can use for patient movement with litters on wheels or patient beds,” Capt. Rotruck said. “The uphill portion of those ramps represents a good workout opportunity as well.”
Downtime in an era of physical distancing also afforded crew members the opportunity to call or FaceTime with loved ones, watch streamed TV shows and movies, and work on their own professional development. Some continued with coursework for online degree programs offered by colleges and universities they were enrolled in, while some enlisted personnel used the time to complete the Navy Enlisted Warfare Qualification Programs Instruction, which issues the basic overarching requirements for the qualification and designation of all enlisted warfare programs.
“As you can imagine, people spend a lot of time learning how the ship works and how it integrates into larger naval forces and so forth,” Capt. Rotruck said. “Not just our ship but also other ships: their weapons systems and defense mechanisms and navigation systems. We had people spending a significant amount of time working on that. We had people complete their Enlisted Surface Warfare qualification while we were on the mission.”
End of the mission
Mercy returned to its home base in San Diego on May 15, but about 60 medical personnel stayed behind in Los Angeles to support Federal Emergency Management Agency (FEMA), state, and local health care professionals. Some worked at a site where clinicians provided care for COVID-19–positive patients who had been transferred from area skilled nursing facilities.
In addition, a team consisting of one nurse and five corpsmen “would go out to individual skilled nursing facilities and mainly conduct assessments and training, such as training in donning proper PPE [personal protective equipment] and determining what needs they had,” Capt. Rotruck said. “They met those needs if possible or [communicated with California officials] and let them know what the requirements were and what the needs were in that facility.” The assignment for those who stayed behind ended on May 31.
On the opposite coast, Mercy’s sister ship, USNS Comfort, arrived in New York Harbor from Norfolk, Va., on March 30 and spent 3½ weeks assisting area hospitals in the COVID-19 pandemic fight. A few days into the mission, Comfort’s internal spaces were reconfigured to create separate COVID-negative and COVID-positive sections. Medical teams aboard the ship cared for a total of 182 patients during the assignment.
Looking back on Mercy’s mission, Cdr. Blevins marveled at the sense of teamwork that unfolded. “We have quarterly training exercises with a core set of personnel, [and] we train getting ready for activation in 5 days,” she said. “All of that training kicks in and it comes to fruition in a mission like this. It was terrific to see a group of very disparate subject matter experts from all over the country come together with one purpose: which was to serve our own country during the pandemic.”
Capt. Rotruck pointed out that the experience enabled enlisted and nonenlisted physicians to maintain their skill sets during a time when military and civilian hospitals had stopped doing elective procedures and routine appointments. “The fact that those people were able to come on board the ship and continue to conduct their medical practice and maintain their skills and competencies in an environment that they weren’t quite used to is great,” he said. “Otherwise, some of those medical personnel would have been sitting idle, wherever they were from. This is the power of Navy medicine on behalf of our country.”
Guidance on infection prevention for health care personnel
As we reopen our offices we are faced with the challenge of determining the best way to do it safely – protecting ourselves, our staff, and our patients.
In this column we will focus on selected details of the recommendations from IDSA and the CDC that may be helpful in primary care offices.
Face masks
Many clinicians have asked whether a physician should use a mask while seeing patients without COVID-19 in the office, and if yes, which type. The IDSA guideline states that mask usage is imperative for reducing the risk of health care workers contracting COVID-19.1 The evidence is derived from a number of sources, including a retrospective study from Wuhan (China) University that examined two groups of health care workers during the outbreak. The first group wore N95 masks and washed their hands frequently, while the second group did not wear masks and washed their hands less frequently. In the group that took greater actions to protect themselves, none of the 493 staff members contracted COVID-19, compared with 10 of 213 staff members in the other group. The decrease in infection rate occurred in the group that wore masks despite the fact that this group had 733% more exposure to COVID-19 patients.2 Further evidence came from a case-control study done in hospitals in Hong Kong during the 2003 SARS-CoV outbreak.3 This study showed that mask wearing was the most significant intervention for reducing infection, followed by gowning, and then handwashing. These findings make it clear that mask usage is a must for all health care providers who may be caring for patients who could have COVID-19.
The guideline also reviews evidence about the use of surgical masks versus N95 masks. On reviewing indirect evidence from the SARS-CoV epidemic, IDSA found that wearing any mask – surgical or N95 – led to a large reduction in the risk of developing an infection. In this systematic review of five observational studies in health care personnel, for those wearing surgical masks, the odds ratio for developing an infection was 0.13 (95% CI, 0.03-0.62), and for those wearing N95 masks, the odds ratio was 0.12 (95% CI, 0.06-0.26). There was not a significant difference between risk reductions for those who wore surgical masks and N95 masks, respectively.1,4 The IDSA guideline panel recommended “that health care personnel caring for patients with suspected or known COVID-19 use either a surgical mask or N95 respirator ... as part of appropriate PPE.” Since there is not a significant difference in outcomes between those who use surgical masks and those who use N95 respirators, and the IDSA guideline states either type of mask is considered appropriate when taking care of patients with suspected or known COVID-19, in our opinion, use of surgical masks rather than N95s is sufficient when performing low-risk activities. Such activities include seeing patients who do not have a high likelihood of COVID-19 in the office setting.
The IDSA recommendation also discusses universal masking, defined as both patients and clinicians wearing masks. The recommendation is supported by the findings of a study in which universal mask usage was used to prevent the spread of H1N 1 during the 2009 outbreak. In this study of staff members and patients exposed to H1N1 who all wore masks, only 0.48% of 836 acquired infection. In the same study, not wearing a mask by either the provider or patient increased the risk of infection.5 Also, in a prospective study of hematopoietic stem cell transplant patients, universal masking caused infection rates to drop from 10.3% to 4.4%.6
The IDSA guideline states the following: “There may be some, albeit uncertain, benefit to universal masking in the absence of resource constraints. However, the benefits of universal masking with surgical masks should be weighed against the risk of increasing the PPE burn rate and contextualized to the background COVID-19 prevalence rate for asymptomatic or minimally symptomatic HCPs [health care providers] and visitors.”1
The CDC’s guidance statement says the following: “Continued community transmission has increased the number of individuals potentially exposed to and infectious with SARS-CoV-2. Fever and symptom screening have proven to be relatively ineffective in identifying all infected individuals, including HCPs. Symptom screening also will not identify individuals who are infected but otherwise asymptomatic or pre-symptomatic; additional interventions are needed to limit the unrecognized introduction of SARS-CoV-2 into healthcare settings by these individuals. As part of aggressive source control measures, healthcare facilities should consider implementing policies requiring everyone entering the facility to wear a cloth face covering (if tolerated) while in the building, regardless of symptoms.”7
It is our opinion, based on the CDC and IDSA recommendations, that both clinicians and patients should be required to wear masks when patients are seen in the office if possible. Many offices have instituted a policy that says, if a patient refuses to wear a mask during an office visit, then the patient will not be seen.
Eye protection
Many clinicians are uncertain about whether eye protection needs to be used when seeing asymptomatic patients. The IDSA acknowledges that there are not studies that have looked critically at eye protection, but the society also acknowledges “appropriate personal protective equipment includes, in addition to a mask or respirator, eye protection, gown and gloves.”1 In addition, the CDC recommends that, for healthcare workers located in areas with moderate or higher prevalence of COVID-19, HCPs should wear eye protection in addition to facemasks since they may encounter asymptomatic individuals with COVID-19.
Gowns and gloves
Gowns and gloves are recommended as a part of personal protective gear when caring for patients who have COVID-19. The IDSA guideline is clear in its recommendations, but does not cite evidence for having no gloves versus having gloves. Furthermore, they state that the evidence is insufficient to recommend double gloves, with the top glove used to take off a personal protective gown, and the inner glove discarded after the gown is removed. The CDC do not make recommendations for routine use of gloves in the care of patients who do not have COVID-19, even in areas where there may be asymptomatic COVID-19, and recommends standard precautions, specifically practicing hand hygiene before and after patient contact.8
The Bottom Line
When seeing patients with COVID-19, N-95 masks, goggles or face shields, gowns, and gloves should be used, with hand hygiene routinely practiced before and after seeing patients. For offices seeing patients not suspected of having COVID-19, the IDSA guideline clarifies that there is not a statistical difference in acquisition of infection with the use of surgical face masks vs N95 respirators. According to the CDC recommendations, eye protection in addition to facemasks should be used by the health care provider, and masks should be worn by patients. Hand hygiene should be used routinely before and after all patient contact. With use of these approaches, it should be safe for offices to reopen and see patients.
Neil Skolnik, MD, is professor of family and community medicine at the Thomas Jefferson University, Philadelphia, and associate director of the Family Medicine Residency Program at Abington (Pa.) Jefferson Health. Jeffrey Matthews, DO, is a second-year resident in the Family Medicine Residency at Abington Jefferson Health. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. Lynch JB, Davitkov P, Anderson DJ, et al. COVID-19 Guideline, Part 2: Infection Prevention. IDSA Home. https://www.idsociety.org/practice-guideline/covid-19-guideline-infection-prevention/. April 27, 2020. Accessed June 10, 2020.
2. J Hosp Infect. 2020 May;105(1):104-5.
3. Lancet. 2003;361(9368):1519-20.
4. Influenza Other Respir Viruses. 2020 Apr 4. doi: 2020;10.1111/irv.12745.
5. J Hosp Infect. 2010;74(3):271-7.
6. Clin Infect Dis. 2016;63(8):999-1006.
7. Centers for Disease Control and Prevention. Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed Jun 16, 2020.
8. Centers for Disease Control and Prevention. Healthcare Infection Prevention and Control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html. Accessed June 15, 2020.
As we reopen our offices we are faced with the challenge of determining the best way to do it safely – protecting ourselves, our staff, and our patients.
In this column we will focus on selected details of the recommendations from IDSA and the CDC that may be helpful in primary care offices.
Face masks
Many clinicians have asked whether a physician should use a mask while seeing patients without COVID-19 in the office, and if yes, which type. The IDSA guideline states that mask usage is imperative for reducing the risk of health care workers contracting COVID-19.1 The evidence is derived from a number of sources, including a retrospective study from Wuhan (China) University that examined two groups of health care workers during the outbreak. The first group wore N95 masks and washed their hands frequently, while the second group did not wear masks and washed their hands less frequently. In the group that took greater actions to protect themselves, none of the 493 staff members contracted COVID-19, compared with 10 of 213 staff members in the other group. The decrease in infection rate occurred in the group that wore masks despite the fact that this group had 733% more exposure to COVID-19 patients.2 Further evidence came from a case-control study done in hospitals in Hong Kong during the 2003 SARS-CoV outbreak.3 This study showed that mask wearing was the most significant intervention for reducing infection, followed by gowning, and then handwashing. These findings make it clear that mask usage is a must for all health care providers who may be caring for patients who could have COVID-19.
The guideline also reviews evidence about the use of surgical masks versus N95 masks. On reviewing indirect evidence from the SARS-CoV epidemic, IDSA found that wearing any mask – surgical or N95 – led to a large reduction in the risk of developing an infection. In this systematic review of five observational studies in health care personnel, for those wearing surgical masks, the odds ratio for developing an infection was 0.13 (95% CI, 0.03-0.62), and for those wearing N95 masks, the odds ratio was 0.12 (95% CI, 0.06-0.26). There was not a significant difference between risk reductions for those who wore surgical masks and N95 masks, respectively.1,4 The IDSA guideline panel recommended “that health care personnel caring for patients with suspected or known COVID-19 use either a surgical mask or N95 respirator ... as part of appropriate PPE.” Since there is not a significant difference in outcomes between those who use surgical masks and those who use N95 respirators, and the IDSA guideline states either type of mask is considered appropriate when taking care of patients with suspected or known COVID-19, in our opinion, use of surgical masks rather than N95s is sufficient when performing low-risk activities. Such activities include seeing patients who do not have a high likelihood of COVID-19 in the office setting.
The IDSA recommendation also discusses universal masking, defined as both patients and clinicians wearing masks. The recommendation is supported by the findings of a study in which universal mask usage was used to prevent the spread of H1N 1 during the 2009 outbreak. In this study of staff members and patients exposed to H1N1 who all wore masks, only 0.48% of 836 acquired infection. In the same study, not wearing a mask by either the provider or patient increased the risk of infection.5 Also, in a prospective study of hematopoietic stem cell transplant patients, universal masking caused infection rates to drop from 10.3% to 4.4%.6
The IDSA guideline states the following: “There may be some, albeit uncertain, benefit to universal masking in the absence of resource constraints. However, the benefits of universal masking with surgical masks should be weighed against the risk of increasing the PPE burn rate and contextualized to the background COVID-19 prevalence rate for asymptomatic or minimally symptomatic HCPs [health care providers] and visitors.”1
The CDC’s guidance statement says the following: “Continued community transmission has increased the number of individuals potentially exposed to and infectious with SARS-CoV-2. Fever and symptom screening have proven to be relatively ineffective in identifying all infected individuals, including HCPs. Symptom screening also will not identify individuals who are infected but otherwise asymptomatic or pre-symptomatic; additional interventions are needed to limit the unrecognized introduction of SARS-CoV-2 into healthcare settings by these individuals. As part of aggressive source control measures, healthcare facilities should consider implementing policies requiring everyone entering the facility to wear a cloth face covering (if tolerated) while in the building, regardless of symptoms.”7
It is our opinion, based on the CDC and IDSA recommendations, that both clinicians and patients should be required to wear masks when patients are seen in the office if possible. Many offices have instituted a policy that says, if a patient refuses to wear a mask during an office visit, then the patient will not be seen.
Eye protection
Many clinicians are uncertain about whether eye protection needs to be used when seeing asymptomatic patients. The IDSA acknowledges that there are not studies that have looked critically at eye protection, but the society also acknowledges “appropriate personal protective equipment includes, in addition to a mask or respirator, eye protection, gown and gloves.”1 In addition, the CDC recommends that, for healthcare workers located in areas with moderate or higher prevalence of COVID-19, HCPs should wear eye protection in addition to facemasks since they may encounter asymptomatic individuals with COVID-19.
Gowns and gloves
Gowns and gloves are recommended as a part of personal protective gear when caring for patients who have COVID-19. The IDSA guideline is clear in its recommendations, but does not cite evidence for having no gloves versus having gloves. Furthermore, they state that the evidence is insufficient to recommend double gloves, with the top glove used to take off a personal protective gown, and the inner glove discarded after the gown is removed. The CDC do not make recommendations for routine use of gloves in the care of patients who do not have COVID-19, even in areas where there may be asymptomatic COVID-19, and recommends standard precautions, specifically practicing hand hygiene before and after patient contact.8
The Bottom Line
When seeing patients with COVID-19, N-95 masks, goggles or face shields, gowns, and gloves should be used, with hand hygiene routinely practiced before and after seeing patients. For offices seeing patients not suspected of having COVID-19, the IDSA guideline clarifies that there is not a statistical difference in acquisition of infection with the use of surgical face masks vs N95 respirators. According to the CDC recommendations, eye protection in addition to facemasks should be used by the health care provider, and masks should be worn by patients. Hand hygiene should be used routinely before and after all patient contact. With use of these approaches, it should be safe for offices to reopen and see patients.
Neil Skolnik, MD, is professor of family and community medicine at the Thomas Jefferson University, Philadelphia, and associate director of the Family Medicine Residency Program at Abington (Pa.) Jefferson Health. Jeffrey Matthews, DO, is a second-year resident in the Family Medicine Residency at Abington Jefferson Health. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. Lynch JB, Davitkov P, Anderson DJ, et al. COVID-19 Guideline, Part 2: Infection Prevention. IDSA Home. https://www.idsociety.org/practice-guideline/covid-19-guideline-infection-prevention/. April 27, 2020. Accessed June 10, 2020.
2. J Hosp Infect. 2020 May;105(1):104-5.
3. Lancet. 2003;361(9368):1519-20.
4. Influenza Other Respir Viruses. 2020 Apr 4. doi: 2020;10.1111/irv.12745.
5. J Hosp Infect. 2010;74(3):271-7.
6. Clin Infect Dis. 2016;63(8):999-1006.
7. Centers for Disease Control and Prevention. Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed Jun 16, 2020.
8. Centers for Disease Control and Prevention. Healthcare Infection Prevention and Control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html. Accessed June 15, 2020.
As we reopen our offices we are faced with the challenge of determining the best way to do it safely – protecting ourselves, our staff, and our patients.
In this column we will focus on selected details of the recommendations from IDSA and the CDC that may be helpful in primary care offices.
Face masks
Many clinicians have asked whether a physician should use a mask while seeing patients without COVID-19 in the office, and if yes, which type. The IDSA guideline states that mask usage is imperative for reducing the risk of health care workers contracting COVID-19.1 The evidence is derived from a number of sources, including a retrospective study from Wuhan (China) University that examined two groups of health care workers during the outbreak. The first group wore N95 masks and washed their hands frequently, while the second group did not wear masks and washed their hands less frequently. In the group that took greater actions to protect themselves, none of the 493 staff members contracted COVID-19, compared with 10 of 213 staff members in the other group. The decrease in infection rate occurred in the group that wore masks despite the fact that this group had 733% more exposure to COVID-19 patients.2 Further evidence came from a case-control study done in hospitals in Hong Kong during the 2003 SARS-CoV outbreak.3 This study showed that mask wearing was the most significant intervention for reducing infection, followed by gowning, and then handwashing. These findings make it clear that mask usage is a must for all health care providers who may be caring for patients who could have COVID-19.
The guideline also reviews evidence about the use of surgical masks versus N95 masks. On reviewing indirect evidence from the SARS-CoV epidemic, IDSA found that wearing any mask – surgical or N95 – led to a large reduction in the risk of developing an infection. In this systematic review of five observational studies in health care personnel, for those wearing surgical masks, the odds ratio for developing an infection was 0.13 (95% CI, 0.03-0.62), and for those wearing N95 masks, the odds ratio was 0.12 (95% CI, 0.06-0.26). There was not a significant difference between risk reductions for those who wore surgical masks and N95 masks, respectively.1,4 The IDSA guideline panel recommended “that health care personnel caring for patients with suspected or known COVID-19 use either a surgical mask or N95 respirator ... as part of appropriate PPE.” Since there is not a significant difference in outcomes between those who use surgical masks and those who use N95 respirators, and the IDSA guideline states either type of mask is considered appropriate when taking care of patients with suspected or known COVID-19, in our opinion, use of surgical masks rather than N95s is sufficient when performing low-risk activities. Such activities include seeing patients who do not have a high likelihood of COVID-19 in the office setting.
The IDSA recommendation also discusses universal masking, defined as both patients and clinicians wearing masks. The recommendation is supported by the findings of a study in which universal mask usage was used to prevent the spread of H1N 1 during the 2009 outbreak. In this study of staff members and patients exposed to H1N1 who all wore masks, only 0.48% of 836 acquired infection. In the same study, not wearing a mask by either the provider or patient increased the risk of infection.5 Also, in a prospective study of hematopoietic stem cell transplant patients, universal masking caused infection rates to drop from 10.3% to 4.4%.6
The IDSA guideline states the following: “There may be some, albeit uncertain, benefit to universal masking in the absence of resource constraints. However, the benefits of universal masking with surgical masks should be weighed against the risk of increasing the PPE burn rate and contextualized to the background COVID-19 prevalence rate for asymptomatic or minimally symptomatic HCPs [health care providers] and visitors.”1
The CDC’s guidance statement says the following: “Continued community transmission has increased the number of individuals potentially exposed to and infectious with SARS-CoV-2. Fever and symptom screening have proven to be relatively ineffective in identifying all infected individuals, including HCPs. Symptom screening also will not identify individuals who are infected but otherwise asymptomatic or pre-symptomatic; additional interventions are needed to limit the unrecognized introduction of SARS-CoV-2 into healthcare settings by these individuals. As part of aggressive source control measures, healthcare facilities should consider implementing policies requiring everyone entering the facility to wear a cloth face covering (if tolerated) while in the building, regardless of symptoms.”7
It is our opinion, based on the CDC and IDSA recommendations, that both clinicians and patients should be required to wear masks when patients are seen in the office if possible. Many offices have instituted a policy that says, if a patient refuses to wear a mask during an office visit, then the patient will not be seen.
Eye protection
Many clinicians are uncertain about whether eye protection needs to be used when seeing asymptomatic patients. The IDSA acknowledges that there are not studies that have looked critically at eye protection, but the society also acknowledges “appropriate personal protective equipment includes, in addition to a mask or respirator, eye protection, gown and gloves.”1 In addition, the CDC recommends that, for healthcare workers located in areas with moderate or higher prevalence of COVID-19, HCPs should wear eye protection in addition to facemasks since they may encounter asymptomatic individuals with COVID-19.
Gowns and gloves
Gowns and gloves are recommended as a part of personal protective gear when caring for patients who have COVID-19. The IDSA guideline is clear in its recommendations, but does not cite evidence for having no gloves versus having gloves. Furthermore, they state that the evidence is insufficient to recommend double gloves, with the top glove used to take off a personal protective gown, and the inner glove discarded after the gown is removed. The CDC do not make recommendations for routine use of gloves in the care of patients who do not have COVID-19, even in areas where there may be asymptomatic COVID-19, and recommends standard precautions, specifically practicing hand hygiene before and after patient contact.8
The Bottom Line
When seeing patients with COVID-19, N-95 masks, goggles or face shields, gowns, and gloves should be used, with hand hygiene routinely practiced before and after seeing patients. For offices seeing patients not suspected of having COVID-19, the IDSA guideline clarifies that there is not a statistical difference in acquisition of infection with the use of surgical face masks vs N95 respirators. According to the CDC recommendations, eye protection in addition to facemasks should be used by the health care provider, and masks should be worn by patients. Hand hygiene should be used routinely before and after all patient contact. With use of these approaches, it should be safe for offices to reopen and see patients.
Neil Skolnik, MD, is professor of family and community medicine at the Thomas Jefferson University, Philadelphia, and associate director of the Family Medicine Residency Program at Abington (Pa.) Jefferson Health. Jeffrey Matthews, DO, is a second-year resident in the Family Medicine Residency at Abington Jefferson Health. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. Lynch JB, Davitkov P, Anderson DJ, et al. COVID-19 Guideline, Part 2: Infection Prevention. IDSA Home. https://www.idsociety.org/practice-guideline/covid-19-guideline-infection-prevention/. April 27, 2020. Accessed June 10, 2020.
2. J Hosp Infect. 2020 May;105(1):104-5.
3. Lancet. 2003;361(9368):1519-20.
4. Influenza Other Respir Viruses. 2020 Apr 4. doi: 2020;10.1111/irv.12745.
5. J Hosp Infect. 2010;74(3):271-7.
6. Clin Infect Dis. 2016;63(8):999-1006.
7. Centers for Disease Control and Prevention. Interim Infection Prevention and Control Recommendations for Patients with Suspected or Confirmed Coronavirus Disease 2019 (COVID-19) in Healthcare Settings. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html. Accessed Jun 16, 2020.
8. Centers for Disease Control and Prevention. Healthcare Infection Prevention and Control FAQs for COVID-19. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-faq.html. Accessed June 15, 2020.
‘COVID-sorting’: How we decide whom to get close to and whom to avoid
I was recently interviewed, as a gay psychiatrist treating gay patients who lived through the AIDS epidemic, about my perspectives on living through a COVID pandemic: Were there parallels and contrasts between the two? A month later, listening to patients remotely via teletherapy, I’m experiencing an unsettling similarity to serosorting, a phenomenon that emerged during the AIDS epidemic.
Serosorting is the practice of choosing a sexual partner based on their HIV serostatus. Sorting out who was positive from who was negative allowed people to give themselves permission to have unprotected sex without risk of getting HIV. However, it was not uncommon to make those decisions without really knowing a potential partner’s actual serostatus. In fact, a lot of people serosorted by guessing.
Why not just ask a potential partner, “What’s your serostatus?” Apparently, for some, introducing the subject of HIV was deemed a sexual buzzkill. Instead, assumptions were made based on outer appearances.
Did someone look healthy? Were they well built? Were they overweight, meaning not emaciated from AIDS? If so, they were presumed negative and safe to have risky, unprotected sex with them.
Some imagined age correlated with serostatus. Since anyone older than some arbitrary age – like 30, to pull a number out of a hat – was expected to be more likely to have HIV than someone under 30, they would use that guideline in choosing sexual partners. However, these decisions were made without factual knowledge, like a blood test, but using some internal reasoning process.
Which brings us to what might be called “COVID-sorting.”
Some of my patients believe they had COVID-19, although they’d not been tested to either confirm or disprove that belief. Others had positive COVID-19 antibody tests, which they believe provides immunity. Among that group, some had symptoms, others did not.
Yet regardless of what they actually know or don’t know, patients are making calculations about managing physical distancing using their own internal formulas. They make risk calculations having little to do with actual knowledge of public health precautions on preventing COVID’s spread.
For example, one patient was planning a Memorial Day weekend in a shared Fire Island house with five friends and acquaintances. All six live alone and, as far as he knows, all are physically distancing. Consequently, my patient doesn’t think house-sharing is anything to worry about, even though he doesn’t know how scrupulously others have followed distancing guidelines.
Another patient, recovering at home after being ill with COVID-19, felt safe inviting someone over for sex who had also been ill and recovered. He didn’t think they could infect each other, presuming, not altogether unreasonably, they were both immune.
Finally, there are those who don’t know whether they had COVID-19, but think they did because they experienced influenza-like symptoms. They are giving themselves permission to meet up with others who feel the same.
Yet a Mount Sinai study, which has not yet been peer-reviewed, raises fascinating issues about immunity. The study included 719 people who suspected they had COVID-19 based on some respiratory symptoms. The majority, 62%, had no antibodies. Researchers believe they mistook influenza, another viral infection, or allergies for COVID-19 (medRxiv. 2020 May 5. doi: 10.1101/2020.04.04.2008516).
The study also included 624 people who tested positive for the virus and recovered. All but three developed antibodies. Many assume those who are antibody-positive are now immune. They may be right. However, we don’t know definitively that they are, and if they are, we do not yet know how long immunity may last. Further, as reported in the New York Times, just because you test positive for antibodies, doesn’t mean you have them.
It should be underscored that COVID-sorting is not limited to gay men or psychiatric patients. And as many states have begun opening up restrictions on social gatherings, we are seeing an all-too-human psychological mindset with wider implications – rising numbers of cases. As we move forward, all of us will have to decide for ourselves, and not only in sexual situations, how to get on with our lives in a post–COVID-19 era.
Given how much is still unknown, it is likely each of us will come up with our own algorithm of risk assessment. It is likely that the formulas used will not necessarily be based on scientific facts, although that would be ideal. If past epidemic and recent pandemic behaviors are any indicators, people’s actions will reflect some combination of their own needs and desires, their own comfort level with risk-taking, and their relative understanding of complex subjects like virology, immunology, epidemiology, and public health. The challenge faced by public health officials today is to translate complex scientific and medical issues into messages average people can understand.
What exactly can be done? I’m not exactly sure, but I hope that improved education and communication can help. In the first 2 decades of the AIDS epidemic, efforts were made to change and tailor HIV-prevention messages to specific, at-risk demographic groups. Today, public health messages aimed at preventing COVID-19’s spread that resonate with older people can fall on a younger person’s deaf ears. One message size does not fit all. Hopefully, public health officials and government leaders will act on this sooner rather than later.
Dr. Drescher, a psychoanalyst, is clinical professor of psychiatry at Columbia University, and training and supervising analyst at the William A. White Institute, both in New York. He also is emeritus editor of the Journal of Gay & Lesbian Mental Health. Dr. Drescher has no other disclosures.
I was recently interviewed, as a gay psychiatrist treating gay patients who lived through the AIDS epidemic, about my perspectives on living through a COVID pandemic: Were there parallels and contrasts between the two? A month later, listening to patients remotely via teletherapy, I’m experiencing an unsettling similarity to serosorting, a phenomenon that emerged during the AIDS epidemic.
Serosorting is the practice of choosing a sexual partner based on their HIV serostatus. Sorting out who was positive from who was negative allowed people to give themselves permission to have unprotected sex without risk of getting HIV. However, it was not uncommon to make those decisions without really knowing a potential partner’s actual serostatus. In fact, a lot of people serosorted by guessing.
Why not just ask a potential partner, “What’s your serostatus?” Apparently, for some, introducing the subject of HIV was deemed a sexual buzzkill. Instead, assumptions were made based on outer appearances.
Did someone look healthy? Were they well built? Were they overweight, meaning not emaciated from AIDS? If so, they were presumed negative and safe to have risky, unprotected sex with them.
Some imagined age correlated with serostatus. Since anyone older than some arbitrary age – like 30, to pull a number out of a hat – was expected to be more likely to have HIV than someone under 30, they would use that guideline in choosing sexual partners. However, these decisions were made without factual knowledge, like a blood test, but using some internal reasoning process.
Which brings us to what might be called “COVID-sorting.”
Some of my patients believe they had COVID-19, although they’d not been tested to either confirm or disprove that belief. Others had positive COVID-19 antibody tests, which they believe provides immunity. Among that group, some had symptoms, others did not.
Yet regardless of what they actually know or don’t know, patients are making calculations about managing physical distancing using their own internal formulas. They make risk calculations having little to do with actual knowledge of public health precautions on preventing COVID’s spread.
For example, one patient was planning a Memorial Day weekend in a shared Fire Island house with five friends and acquaintances. All six live alone and, as far as he knows, all are physically distancing. Consequently, my patient doesn’t think house-sharing is anything to worry about, even though he doesn’t know how scrupulously others have followed distancing guidelines.
Another patient, recovering at home after being ill with COVID-19, felt safe inviting someone over for sex who had also been ill and recovered. He didn’t think they could infect each other, presuming, not altogether unreasonably, they were both immune.
Finally, there are those who don’t know whether they had COVID-19, but think they did because they experienced influenza-like symptoms. They are giving themselves permission to meet up with others who feel the same.
Yet a Mount Sinai study, which has not yet been peer-reviewed, raises fascinating issues about immunity. The study included 719 people who suspected they had COVID-19 based on some respiratory symptoms. The majority, 62%, had no antibodies. Researchers believe they mistook influenza, another viral infection, or allergies for COVID-19 (medRxiv. 2020 May 5. doi: 10.1101/2020.04.04.2008516).
The study also included 624 people who tested positive for the virus and recovered. All but three developed antibodies. Many assume those who are antibody-positive are now immune. They may be right. However, we don’t know definitively that they are, and if they are, we do not yet know how long immunity may last. Further, as reported in the New York Times, just because you test positive for antibodies, doesn’t mean you have them.
It should be underscored that COVID-sorting is not limited to gay men or psychiatric patients. And as many states have begun opening up restrictions on social gatherings, we are seeing an all-too-human psychological mindset with wider implications – rising numbers of cases. As we move forward, all of us will have to decide for ourselves, and not only in sexual situations, how to get on with our lives in a post–COVID-19 era.
Given how much is still unknown, it is likely each of us will come up with our own algorithm of risk assessment. It is likely that the formulas used will not necessarily be based on scientific facts, although that would be ideal. If past epidemic and recent pandemic behaviors are any indicators, people’s actions will reflect some combination of their own needs and desires, their own comfort level with risk-taking, and their relative understanding of complex subjects like virology, immunology, epidemiology, and public health. The challenge faced by public health officials today is to translate complex scientific and medical issues into messages average people can understand.
What exactly can be done? I’m not exactly sure, but I hope that improved education and communication can help. In the first 2 decades of the AIDS epidemic, efforts were made to change and tailor HIV-prevention messages to specific, at-risk demographic groups. Today, public health messages aimed at preventing COVID-19’s spread that resonate with older people can fall on a younger person’s deaf ears. One message size does not fit all. Hopefully, public health officials and government leaders will act on this sooner rather than later.
Dr. Drescher, a psychoanalyst, is clinical professor of psychiatry at Columbia University, and training and supervising analyst at the William A. White Institute, both in New York. He also is emeritus editor of the Journal of Gay & Lesbian Mental Health. Dr. Drescher has no other disclosures.
I was recently interviewed, as a gay psychiatrist treating gay patients who lived through the AIDS epidemic, about my perspectives on living through a COVID pandemic: Were there parallels and contrasts between the two? A month later, listening to patients remotely via teletherapy, I’m experiencing an unsettling similarity to serosorting, a phenomenon that emerged during the AIDS epidemic.
Serosorting is the practice of choosing a sexual partner based on their HIV serostatus. Sorting out who was positive from who was negative allowed people to give themselves permission to have unprotected sex without risk of getting HIV. However, it was not uncommon to make those decisions without really knowing a potential partner’s actual serostatus. In fact, a lot of people serosorted by guessing.
Why not just ask a potential partner, “What’s your serostatus?” Apparently, for some, introducing the subject of HIV was deemed a sexual buzzkill. Instead, assumptions were made based on outer appearances.
Did someone look healthy? Were they well built? Were they overweight, meaning not emaciated from AIDS? If so, they were presumed negative and safe to have risky, unprotected sex with them.
Some imagined age correlated with serostatus. Since anyone older than some arbitrary age – like 30, to pull a number out of a hat – was expected to be more likely to have HIV than someone under 30, they would use that guideline in choosing sexual partners. However, these decisions were made without factual knowledge, like a blood test, but using some internal reasoning process.
Which brings us to what might be called “COVID-sorting.”
Some of my patients believe they had COVID-19, although they’d not been tested to either confirm or disprove that belief. Others had positive COVID-19 antibody tests, which they believe provides immunity. Among that group, some had symptoms, others did not.
Yet regardless of what they actually know or don’t know, patients are making calculations about managing physical distancing using their own internal formulas. They make risk calculations having little to do with actual knowledge of public health precautions on preventing COVID’s spread.
For example, one patient was planning a Memorial Day weekend in a shared Fire Island house with five friends and acquaintances. All six live alone and, as far as he knows, all are physically distancing. Consequently, my patient doesn’t think house-sharing is anything to worry about, even though he doesn’t know how scrupulously others have followed distancing guidelines.
Another patient, recovering at home after being ill with COVID-19, felt safe inviting someone over for sex who had also been ill and recovered. He didn’t think they could infect each other, presuming, not altogether unreasonably, they were both immune.
Finally, there are those who don’t know whether they had COVID-19, but think they did because they experienced influenza-like symptoms. They are giving themselves permission to meet up with others who feel the same.
Yet a Mount Sinai study, which has not yet been peer-reviewed, raises fascinating issues about immunity. The study included 719 people who suspected they had COVID-19 based on some respiratory symptoms. The majority, 62%, had no antibodies. Researchers believe they mistook influenza, another viral infection, or allergies for COVID-19 (medRxiv. 2020 May 5. doi: 10.1101/2020.04.04.2008516).
The study also included 624 people who tested positive for the virus and recovered. All but three developed antibodies. Many assume those who are antibody-positive are now immune. They may be right. However, we don’t know definitively that they are, and if they are, we do not yet know how long immunity may last. Further, as reported in the New York Times, just because you test positive for antibodies, doesn’t mean you have them.
It should be underscored that COVID-sorting is not limited to gay men or psychiatric patients. And as many states have begun opening up restrictions on social gatherings, we are seeing an all-too-human psychological mindset with wider implications – rising numbers of cases. As we move forward, all of us will have to decide for ourselves, and not only in sexual situations, how to get on with our lives in a post–COVID-19 era.
Given how much is still unknown, it is likely each of us will come up with our own algorithm of risk assessment. It is likely that the formulas used will not necessarily be based on scientific facts, although that would be ideal. If past epidemic and recent pandemic behaviors are any indicators, people’s actions will reflect some combination of their own needs and desires, their own comfort level with risk-taking, and their relative understanding of complex subjects like virology, immunology, epidemiology, and public health. The challenge faced by public health officials today is to translate complex scientific and medical issues into messages average people can understand.
What exactly can be done? I’m not exactly sure, but I hope that improved education and communication can help. In the first 2 decades of the AIDS epidemic, efforts were made to change and tailor HIV-prevention messages to specific, at-risk demographic groups. Today, public health messages aimed at preventing COVID-19’s spread that resonate with older people can fall on a younger person’s deaf ears. One message size does not fit all. Hopefully, public health officials and government leaders will act on this sooner rather than later.
Dr. Drescher, a psychoanalyst, is clinical professor of psychiatry at Columbia University, and training and supervising analyst at the William A. White Institute, both in New York. He also is emeritus editor of the Journal of Gay & Lesbian Mental Health. Dr. Drescher has no other disclosures.
The evolution of “COVIDists”
Adapting to the demands placed on hospital resources by COVID-19
The challenges posed by COVID-19 have crippled health care systems around the globe. By February 2020, the first outbreak in the United States had been set off in Washington State. We quickly became the world’s epicenter of the epidemic, with over 1.8 million patients and over 110,000 deaths.1 The rapidity of spread and the severity of the disease created a tremendous strain on resources. It blindsided policymakers and hospital administrators, which left little time to react to the challenges placed on hospital operations all over the country.
The necessity of a new care model
Although health systems in the United States are adept in managing complications of common seasonal viral respiratory illnesses, COVID-19 presented an entirely different challenge with its significantly higher mortality rate. A respiratory disease turning into a multiorgan disease that causes debilitating cardiac, renal, neurological, hematological, and psychosocial complications2 was not something we had experience managing effectively. Additional challenges included a massive surge of COVID-19 patients, a limited supply of personal protective equipment (PPE), an inadequate number of intensivists for managing the anticipated ventilated patients, and most importantly, the potential of losing some of our workforce if they became infected.
Based on the experiences in China and Italy, and various predictive models, the division of hospital medicine at Baystate Health quickly realized the necessity of a new model of care for COVID-19 patients. We came up with an elaborate plan to manage the disease burden and the strain on resources effectively. The measures we put in place could be broadly divided into three categories following the timeline of the disease: the preparatory phase, the execution phase, and the maintenance phase.
The preparatory phase: From “Hospitalists” to “COVIDists”
As in most hospitals around the country, hospitalists are the backbone of inpatient clinical operations at our health system. A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them.
COVIDists were trained in various treatment protocols and ongoing clinical trials. They were given refresher training in Advanced Cardiac Life Support (ACLS) and Fundamental Critical Care Support (FCCS) courses and were taught in critical care/ventilator management by the intensivists through rapid indoctrination in the ICU. All of them had their N-95 mask fitting updated and were trained in the safe donning and doffing of all kinds of PPE by PPE coaches. The palliative care team trained them in conducting end-of-life/code status discussions with a focus on being unable to speak with family members at the bedside. COVIDists were also assigned as Code Blue leaders for any “COVID code blue” in the hospital.
In addition to the rapid training course, COVID-related updates were disseminated daily using three different modalities: brief huddles at the start of the day with the COVIDists; a COVID-19 newsletter summarizing daily updates, new treatments, strategies, and policies; and a WhatsApp group for instantly broadcasting information to the COVIDists (Table 1).
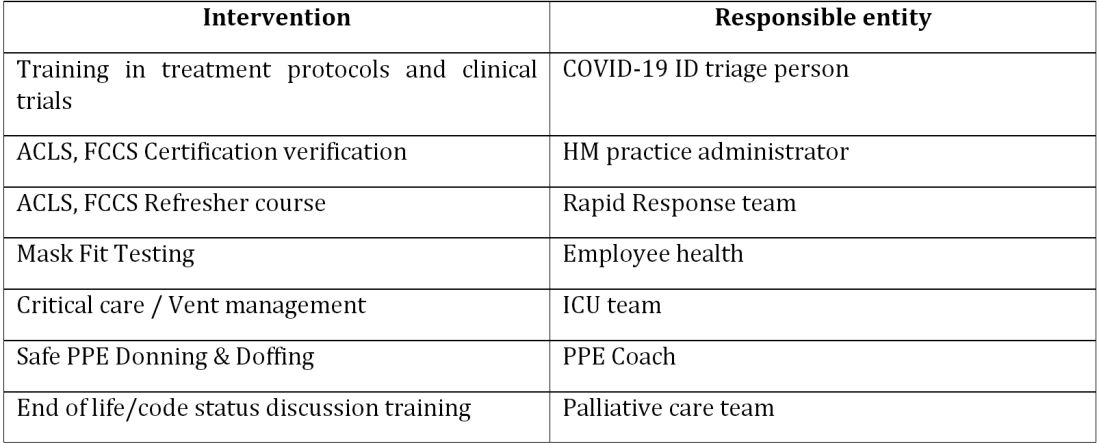
The execution phase
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. The COVIDists were also undertaking the most challenging part of the care – talking to families about end-of-life issues and the futility of aggressive care in certain patients with preexisting conditions.
Some COVIDists were deployed to the ICU to work alongside the intensivists and became an invaluable resource in ICU management when the ICU census skyrocketed during the initial phase of the outbreak. This helped in tiding the health system over during the initial crisis. Within a short time, we shifted away from an early intubation strategy, and most of the ICU patients were managed in the intermediate care units on high flow oxygen along with the awake-proning protocol. The COVIDists exclusively managed these units. They led multidisciplinary rounds two times a day with the ICU, rapid response team (RRT), the palliative care team, and the nursing team. This step drastically decreased the number of intubations, RRT activations, reduced ICU census,3 and helped with hospital capacity and patient flow (Tables 2 and 3).
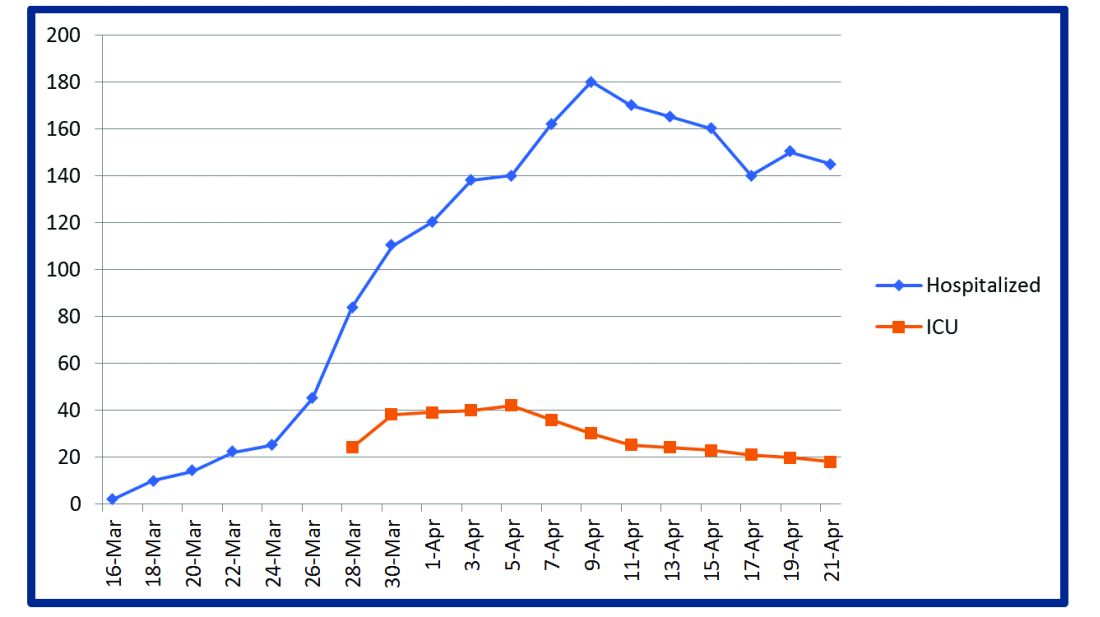
This strategy also helped build solidarity and camaraderie between all these groups, making the COVIDists feel that they were never alone and that the whole hospital supported them. We are currently evaluating clinical outcomes and attempting to identify effects on mortality, length of stay, days on the ventilator, and days in ICU.

The maintenance phase
It is already 2 months since the first devising COVIDists. There is no difference in sick callouts between COVIDists and non-COVIDists. One COVIDist and one non-COVIDist contracted the disease, but none of them required hospitalization. Although we initially thought that COVIDists would be needed for only a short period of time, the evolution of the disease is showing signs that it might be prolonged over the next several months. Hence, we are planning to continue COVIDist service for at least the next 6 months and reevaluate the need.
Hospital medicine leadership checked on COVIDists daily in regard to their physical health and, more importantly, their mental well-being. They were offered the chance to be taken off the schedule if they felt burned out, but no one wanted to come off their scheduled service before finishing their shifts. BlueCross MA recognized one of the COVIDists, Raghuveer Rakasi, MD, as a “hero on the front line.”4 In Dr. Rakasi’s words, “We took a nosedive into something without knowing its depth, and aware that we could have fatalities among ourselves. We took up new roles, faced new challenges, learned new things every day, evolving every step of the way. We had to change the way we practice medicine, finding new ways to treat patients, and protecting the workforce by limiting patient exposure, prioritizing investigations.” He added that “we have to adapt to a new normal; we should be prepared for this to come in waves. Putting aside our political views, we should stand united 6 feet apart, with a mask covering our brave faces, frequently washing our helping hands to overcome these uncertain times.”
Conclusion
The creation of a focused group of hospitalists called COVIDists and providing them with structured and rapid training (in various aspects of clinical care of COVID-19 patients, critical care/ventilator management, efficient and safe use of PPE) and daily information dissemination allowed our health system to prepare for the large volume of COVID-19 patients. It also helped in preserving the larger hospital workforce for a possible future surge.
The rapid development and implementation of the COVIDist strategy succeeded because of the intrinsic motivation of the providers to improve the outcomes of this high-risk patient population and the close collaboration of the stakeholders. Our institution remains successful in managing the pandemic in Western Massachusetts, with reserve capacity remaining even during the peak of the epidemic. A large part of this was because of creating and training a pool of COVIDists.
Dr. Medarametla is medical director, clinical operations, in the division of hospital medicine at Baystate Health, and assistant professor at University of Massachusetts, Worcester. Readers can contact him at [email protected]. Dr. Prabhakaran is unit medical director, geriatrics unit, in the division of hospital medicine at Baystate Health and assistant professor at University of Massachusetts. Dr. Bryson is associate program director of the Internal Medicine Residency at Baystate Health and assistant professor at University of Massachusetts. Dr. Umar is medical director, clinical operations, in the division of hospital medicine at Baystate Health. Dr. Natanasabapathy is division chief of hospital medicine at Baystate Health and assistant professor at University of Massachusetts.
References
1. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Updated Jun 10, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
2. Zhou F et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020 Mar 28;395(10229):1054-62.
3. Westafer LM et al. A transdisciplinary COVID-19 early respiratory intervention protocol: An implementation story. J Hosp Med. 2020 May 21;15(6):372-374.
4. Miller J. “Heroes on the front line: Dr. Raghuveer Rakasi.” Coverage. May 18, 2020. https://coverage.bluecrossma.com/article/heroes-front-line-dr-raghuveer-rakasi
Adapting to the demands placed on hospital resources by COVID-19
Adapting to the demands placed on hospital resources by COVID-19
The challenges posed by COVID-19 have crippled health care systems around the globe. By February 2020, the first outbreak in the United States had been set off in Washington State. We quickly became the world’s epicenter of the epidemic, with over 1.8 million patients and over 110,000 deaths.1 The rapidity of spread and the severity of the disease created a tremendous strain on resources. It blindsided policymakers and hospital administrators, which left little time to react to the challenges placed on hospital operations all over the country.
The necessity of a new care model
Although health systems in the United States are adept in managing complications of common seasonal viral respiratory illnesses, COVID-19 presented an entirely different challenge with its significantly higher mortality rate. A respiratory disease turning into a multiorgan disease that causes debilitating cardiac, renal, neurological, hematological, and psychosocial complications2 was not something we had experience managing effectively. Additional challenges included a massive surge of COVID-19 patients, a limited supply of personal protective equipment (PPE), an inadequate number of intensivists for managing the anticipated ventilated patients, and most importantly, the potential of losing some of our workforce if they became infected.
Based on the experiences in China and Italy, and various predictive models, the division of hospital medicine at Baystate Health quickly realized the necessity of a new model of care for COVID-19 patients. We came up with an elaborate plan to manage the disease burden and the strain on resources effectively. The measures we put in place could be broadly divided into three categories following the timeline of the disease: the preparatory phase, the execution phase, and the maintenance phase.
The preparatory phase: From “Hospitalists” to “COVIDists”
As in most hospitals around the country, hospitalists are the backbone of inpatient clinical operations at our health system. A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them.
COVIDists were trained in various treatment protocols and ongoing clinical trials. They were given refresher training in Advanced Cardiac Life Support (ACLS) and Fundamental Critical Care Support (FCCS) courses and were taught in critical care/ventilator management by the intensivists through rapid indoctrination in the ICU. All of them had their N-95 mask fitting updated and were trained in the safe donning and doffing of all kinds of PPE by PPE coaches. The palliative care team trained them in conducting end-of-life/code status discussions with a focus on being unable to speak with family members at the bedside. COVIDists were also assigned as Code Blue leaders for any “COVID code blue” in the hospital.
In addition to the rapid training course, COVID-related updates were disseminated daily using three different modalities: brief huddles at the start of the day with the COVIDists; a COVID-19 newsletter summarizing daily updates, new treatments, strategies, and policies; and a WhatsApp group for instantly broadcasting information to the COVIDists (Table 1).

The execution phase
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. The COVIDists were also undertaking the most challenging part of the care – talking to families about end-of-life issues and the futility of aggressive care in certain patients with preexisting conditions.
Some COVIDists were deployed to the ICU to work alongside the intensivists and became an invaluable resource in ICU management when the ICU census skyrocketed during the initial phase of the outbreak. This helped in tiding the health system over during the initial crisis. Within a short time, we shifted away from an early intubation strategy, and most of the ICU patients were managed in the intermediate care units on high flow oxygen along with the awake-proning protocol. The COVIDists exclusively managed these units. They led multidisciplinary rounds two times a day with the ICU, rapid response team (RRT), the palliative care team, and the nursing team. This step drastically decreased the number of intubations, RRT activations, reduced ICU census,3 and helped with hospital capacity and patient flow (Tables 2 and 3).

This strategy also helped build solidarity and camaraderie between all these groups, making the COVIDists feel that they were never alone and that the whole hospital supported them. We are currently evaluating clinical outcomes and attempting to identify effects on mortality, length of stay, days on the ventilator, and days in ICU.

The maintenance phase
It is already 2 months since the first devising COVIDists. There is no difference in sick callouts between COVIDists and non-COVIDists. One COVIDist and one non-COVIDist contracted the disease, but none of them required hospitalization. Although we initially thought that COVIDists would be needed for only a short period of time, the evolution of the disease is showing signs that it might be prolonged over the next several months. Hence, we are planning to continue COVIDist service for at least the next 6 months and reevaluate the need.
Hospital medicine leadership checked on COVIDists daily in regard to their physical health and, more importantly, their mental well-being. They were offered the chance to be taken off the schedule if they felt burned out, but no one wanted to come off their scheduled service before finishing their shifts. BlueCross MA recognized one of the COVIDists, Raghuveer Rakasi, MD, as a “hero on the front line.”4 In Dr. Rakasi’s words, “We took a nosedive into something without knowing its depth, and aware that we could have fatalities among ourselves. We took up new roles, faced new challenges, learned new things every day, evolving every step of the way. We had to change the way we practice medicine, finding new ways to treat patients, and protecting the workforce by limiting patient exposure, prioritizing investigations.” He added that “we have to adapt to a new normal; we should be prepared for this to come in waves. Putting aside our political views, we should stand united 6 feet apart, with a mask covering our brave faces, frequently washing our helping hands to overcome these uncertain times.”
Conclusion
The creation of a focused group of hospitalists called COVIDists and providing them with structured and rapid training (in various aspects of clinical care of COVID-19 patients, critical care/ventilator management, efficient and safe use of PPE) and daily information dissemination allowed our health system to prepare for the large volume of COVID-19 patients. It also helped in preserving the larger hospital workforce for a possible future surge.
The rapid development and implementation of the COVIDist strategy succeeded because of the intrinsic motivation of the providers to improve the outcomes of this high-risk patient population and the close collaboration of the stakeholders. Our institution remains successful in managing the pandemic in Western Massachusetts, with reserve capacity remaining even during the peak of the epidemic. A large part of this was because of creating and training a pool of COVIDists.
Dr. Medarametla is medical director, clinical operations, in the division of hospital medicine at Baystate Health, and assistant professor at University of Massachusetts, Worcester. Readers can contact him at [email protected]. Dr. Prabhakaran is unit medical director, geriatrics unit, in the division of hospital medicine at Baystate Health and assistant professor at University of Massachusetts. Dr. Bryson is associate program director of the Internal Medicine Residency at Baystate Health and assistant professor at University of Massachusetts. Dr. Umar is medical director, clinical operations, in the division of hospital medicine at Baystate Health. Dr. Natanasabapathy is division chief of hospital medicine at Baystate Health and assistant professor at University of Massachusetts.
References
1. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Updated Jun 10, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
2. Zhou F et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020 Mar 28;395(10229):1054-62.
3. Westafer LM et al. A transdisciplinary COVID-19 early respiratory intervention protocol: An implementation story. J Hosp Med. 2020 May 21;15(6):372-374.
4. Miller J. “Heroes on the front line: Dr. Raghuveer Rakasi.” Coverage. May 18, 2020. https://coverage.bluecrossma.com/article/heroes-front-line-dr-raghuveer-rakasi
The challenges posed by COVID-19 have crippled health care systems around the globe. By February 2020, the first outbreak in the United States had been set off in Washington State. We quickly became the world’s epicenter of the epidemic, with over 1.8 million patients and over 110,000 deaths.1 The rapidity of spread and the severity of the disease created a tremendous strain on resources. It blindsided policymakers and hospital administrators, which left little time to react to the challenges placed on hospital operations all over the country.
The necessity of a new care model
Although health systems in the United States are adept in managing complications of common seasonal viral respiratory illnesses, COVID-19 presented an entirely different challenge with its significantly higher mortality rate. A respiratory disease turning into a multiorgan disease that causes debilitating cardiac, renal, neurological, hematological, and psychosocial complications2 was not something we had experience managing effectively. Additional challenges included a massive surge of COVID-19 patients, a limited supply of personal protective equipment (PPE), an inadequate number of intensivists for managing the anticipated ventilated patients, and most importantly, the potential of losing some of our workforce if they became infected.
Based on the experiences in China and Italy, and various predictive models, the division of hospital medicine at Baystate Health quickly realized the necessity of a new model of care for COVID-19 patients. We came up with an elaborate plan to manage the disease burden and the strain on resources effectively. The measures we put in place could be broadly divided into three categories following the timeline of the disease: the preparatory phase, the execution phase, and the maintenance phase.
The preparatory phase: From “Hospitalists” to “COVIDists”
As in most hospitals around the country, hospitalists are the backbone of inpatient clinical operations at our health system. A focused group of 10 hospitalists who volunteered to take care of COVID-19 patients with a particular interest in the pandemic and experience in critical care were selected, and the term “COVIDists” was coined to refer to them.
COVIDists were trained in various treatment protocols and ongoing clinical trials. They were given refresher training in Advanced Cardiac Life Support (ACLS) and Fundamental Critical Care Support (FCCS) courses and were taught in critical care/ventilator management by the intensivists through rapid indoctrination in the ICU. All of them had their N-95 mask fitting updated and were trained in the safe donning and doffing of all kinds of PPE by PPE coaches. The palliative care team trained them in conducting end-of-life/code status discussions with a focus on being unable to speak with family members at the bedside. COVIDists were also assigned as Code Blue leaders for any “COVID code blue” in the hospital.
In addition to the rapid training course, COVID-related updates were disseminated daily using three different modalities: brief huddles at the start of the day with the COVIDists; a COVID-19 newsletter summarizing daily updates, new treatments, strategies, and policies; and a WhatsApp group for instantly broadcasting information to the COVIDists (Table 1).

The execution phase
All the hospitalized COVID-19 patients were grouped together to COVID units, and the COVIDists were deployed to those units geographically. COVIDists were given lighter than usual patient loads to deal with the extra time needed for donning and doffing of PPE and for coordination with specialists. COVIDists were almost the only clinicians physically visiting the patients in most cases, and they became the “eyes and ears” of specialists since the specialists were advised to minimize exposure and pursue telemedicine consults. The COVIDists were also undertaking the most challenging part of the care – talking to families about end-of-life issues and the futility of aggressive care in certain patients with preexisting conditions.
Some COVIDists were deployed to the ICU to work alongside the intensivists and became an invaluable resource in ICU management when the ICU census skyrocketed during the initial phase of the outbreak. This helped in tiding the health system over during the initial crisis. Within a short time, we shifted away from an early intubation strategy, and most of the ICU patients were managed in the intermediate care units on high flow oxygen along with the awake-proning protocol. The COVIDists exclusively managed these units. They led multidisciplinary rounds two times a day with the ICU, rapid response team (RRT), the palliative care team, and the nursing team. This step drastically decreased the number of intubations, RRT activations, reduced ICU census,3 and helped with hospital capacity and patient flow (Tables 2 and 3).

This strategy also helped build solidarity and camaraderie between all these groups, making the COVIDists feel that they were never alone and that the whole hospital supported them. We are currently evaluating clinical outcomes and attempting to identify effects on mortality, length of stay, days on the ventilator, and days in ICU.

The maintenance phase
It is already 2 months since the first devising COVIDists. There is no difference in sick callouts between COVIDists and non-COVIDists. One COVIDist and one non-COVIDist contracted the disease, but none of them required hospitalization. Although we initially thought that COVIDists would be needed for only a short period of time, the evolution of the disease is showing signs that it might be prolonged over the next several months. Hence, we are planning to continue COVIDist service for at least the next 6 months and reevaluate the need.
Hospital medicine leadership checked on COVIDists daily in regard to their physical health and, more importantly, their mental well-being. They were offered the chance to be taken off the schedule if they felt burned out, but no one wanted to come off their scheduled service before finishing their shifts. BlueCross MA recognized one of the COVIDists, Raghuveer Rakasi, MD, as a “hero on the front line.”4 In Dr. Rakasi’s words, “We took a nosedive into something without knowing its depth, and aware that we could have fatalities among ourselves. We took up new roles, faced new challenges, learned new things every day, evolving every step of the way. We had to change the way we practice medicine, finding new ways to treat patients, and protecting the workforce by limiting patient exposure, prioritizing investigations.” He added that “we have to adapt to a new normal; we should be prepared for this to come in waves. Putting aside our political views, we should stand united 6 feet apart, with a mask covering our brave faces, frequently washing our helping hands to overcome these uncertain times.”
Conclusion
The creation of a focused group of hospitalists called COVIDists and providing them with structured and rapid training (in various aspects of clinical care of COVID-19 patients, critical care/ventilator management, efficient and safe use of PPE) and daily information dissemination allowed our health system to prepare for the large volume of COVID-19 patients. It also helped in preserving the larger hospital workforce for a possible future surge.
The rapid development and implementation of the COVIDist strategy succeeded because of the intrinsic motivation of the providers to improve the outcomes of this high-risk patient population and the close collaboration of the stakeholders. Our institution remains successful in managing the pandemic in Western Massachusetts, with reserve capacity remaining even during the peak of the epidemic. A large part of this was because of creating and training a pool of COVIDists.
Dr. Medarametla is medical director, clinical operations, in the division of hospital medicine at Baystate Health, and assistant professor at University of Massachusetts, Worcester. Readers can contact him at [email protected]. Dr. Prabhakaran is unit medical director, geriatrics unit, in the division of hospital medicine at Baystate Health and assistant professor at University of Massachusetts. Dr. Bryson is associate program director of the Internal Medicine Residency at Baystate Health and assistant professor at University of Massachusetts. Dr. Umar is medical director, clinical operations, in the division of hospital medicine at Baystate Health. Dr. Natanasabapathy is division chief of hospital medicine at Baystate Health and assistant professor at University of Massachusetts.
References
1. Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19). Updated Jun 10, 2020. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html.
2. Zhou F et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020 Mar 28;395(10229):1054-62.
3. Westafer LM et al. A transdisciplinary COVID-19 early respiratory intervention protocol: An implementation story. J Hosp Med. 2020 May 21;15(6):372-374.
4. Miller J. “Heroes on the front line: Dr. Raghuveer Rakasi.” Coverage. May 18, 2020. https://coverage.bluecrossma.com/article/heroes-front-line-dr-raghuveer-rakasi
Daily Recap: Lung ultrasound helps diagnose COVID-19 in kids, first treatment approved for adult-onset Still’s disease
Here are the stories our MDedge editors across specialties think you need to know about today:
Lung ultrasound works well in children with COVID-19
Lung ultrasound has “high concordance” with radiologic findings in children with COVID-19 and offers benefits over other imaging techniques, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” wrote Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy. “Secondly, when performed at the bedside, [lung ultrasound] allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].” The findings of the small, observational study were published in Pediatrics. Read more.
New hypertension definitions reveal preclampsia risk
Using the new clinical definitions of hypertension, pregnant women with even modest elevations in blood pressure are at increased risk for preeclampsia, according to results from a large retrospective cohort study. Elizabeth F. Sutton, PhD, of the University of Pittsburgh and colleagues looked at records from 18,162 women who had given birth to a single baby. The authors found preeclampsia risk increased with increasing blood pressure elevation. Among women with normal blood pressure before 20 weeks’ gestation, 5% had preeclampsia, while 7% of those with elevated blood pressure did, as did 12% of women with stage 1 hypertension and 30% of women with stage 2 hypertension. The increase in risk of preeclampsia was because of preterm preeclampsia in the women with elevated blood pressure. Preeclampsia researcher Mark Santillan, MD, PhD, of the University of Iowa in Iowa City, said in an interview that the results “open the door to considering these new blood pressure categories as a prognosticator” for preeclampsia. “This paper furthers the field by applying these new categories to hypertensive diseases in pregnancy, which are not well studied” in comparison to nonpregnant hypertensive states. Read more.
Face mask type matters when sterilizing
When sterilizing face masks, the type of mask and the method of sterilization have a bearing on subsequent filtration efficiency, according to new research published in JAMA Network Open. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks. With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%. Read more.
FDA approves first treatment for adult-onset Still’s disease
The Food and Drug Administration has expanded the indications for canakinumab (Ilaris) to include all patients with active Still’s disease older than 2 years, adding adult-onset Still’s disease (AOSD) to a previous approval for juvenile-onset Still’s disease, also known as systemic juvenile idiopathic arthritis (sJIA). That makes Ilaris the first approved treatment for AOSD. The results from a randomized, double-blind, placebo-controlled study of 36 patients with AOSD aged 22-70 years showed that the efficacy and safety data in AOSD were generally consistent with the results of a pooled analysis of sJIA patients, according to Novartis, which markets canakinumab. Read more.
Intranasal DHE shows promise in migraine
An intranasal form of dihydroergotamine (DHE) targeting the upper nasal region is safe and effective for the treatment of migraine, according to results from a phase 3 clinical trial. The new formulation could offer patients an at-home alternative to intramuscular infusions or intravenous injections currently used to deliver DHE. The STOP 301 phase 3 open-label safety and tolerability trial treated over 5,650 migraine attacks in 354 patients who self-administered INP104 for up to 52 weeks. They were provided up to three doses per week (1.45 mg in a dose of two puffs, one per nostril). A total of 66.3% of participants reported pain relief by 2 hours following a dose, and 38% had freedom from pain. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Lung ultrasound works well in children with COVID-19
Lung ultrasound has “high concordance” with radiologic findings in children with COVID-19 and offers benefits over other imaging techniques, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” wrote Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy. “Secondly, when performed at the bedside, [lung ultrasound] allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].” The findings of the small, observational study were published in Pediatrics. Read more.
New hypertension definitions reveal preclampsia risk
Using the new clinical definitions of hypertension, pregnant women with even modest elevations in blood pressure are at increased risk for preeclampsia, according to results from a large retrospective cohort study. Elizabeth F. Sutton, PhD, of the University of Pittsburgh and colleagues looked at records from 18,162 women who had given birth to a single baby. The authors found preeclampsia risk increased with increasing blood pressure elevation. Among women with normal blood pressure before 20 weeks’ gestation, 5% had preeclampsia, while 7% of those with elevated blood pressure did, as did 12% of women with stage 1 hypertension and 30% of women with stage 2 hypertension. The increase in risk of preeclampsia was because of preterm preeclampsia in the women with elevated blood pressure. Preeclampsia researcher Mark Santillan, MD, PhD, of the University of Iowa in Iowa City, said in an interview that the results “open the door to considering these new blood pressure categories as a prognosticator” for preeclampsia. “This paper furthers the field by applying these new categories to hypertensive diseases in pregnancy, which are not well studied” in comparison to nonpregnant hypertensive states. Read more.
Face mask type matters when sterilizing
When sterilizing face masks, the type of mask and the method of sterilization have a bearing on subsequent filtration efficiency, according to new research published in JAMA Network Open. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks. With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%. Read more.
FDA approves first treatment for adult-onset Still’s disease
The Food and Drug Administration has expanded the indications for canakinumab (Ilaris) to include all patients with active Still’s disease older than 2 years, adding adult-onset Still’s disease (AOSD) to a previous approval for juvenile-onset Still’s disease, also known as systemic juvenile idiopathic arthritis (sJIA). That makes Ilaris the first approved treatment for AOSD. The results from a randomized, double-blind, placebo-controlled study of 36 patients with AOSD aged 22-70 years showed that the efficacy and safety data in AOSD were generally consistent with the results of a pooled analysis of sJIA patients, according to Novartis, which markets canakinumab. Read more.
Intranasal DHE shows promise in migraine
An intranasal form of dihydroergotamine (DHE) targeting the upper nasal region is safe and effective for the treatment of migraine, according to results from a phase 3 clinical trial. The new formulation could offer patients an at-home alternative to intramuscular infusions or intravenous injections currently used to deliver DHE. The STOP 301 phase 3 open-label safety and tolerability trial treated over 5,650 migraine attacks in 354 patients who self-administered INP104 for up to 52 weeks. They were provided up to three doses per week (1.45 mg in a dose of two puffs, one per nostril). A total of 66.3% of participants reported pain relief by 2 hours following a dose, and 38% had freedom from pain. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Lung ultrasound works well in children with COVID-19
Lung ultrasound has “high concordance” with radiologic findings in children with COVID-19 and offers benefits over other imaging techniques, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” wrote Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy. “Secondly, when performed at the bedside, [lung ultrasound] allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].” The findings of the small, observational study were published in Pediatrics. Read more.
New hypertension definitions reveal preclampsia risk
Using the new clinical definitions of hypertension, pregnant women with even modest elevations in blood pressure are at increased risk for preeclampsia, according to results from a large retrospective cohort study. Elizabeth F. Sutton, PhD, of the University of Pittsburgh and colleagues looked at records from 18,162 women who had given birth to a single baby. The authors found preeclampsia risk increased with increasing blood pressure elevation. Among women with normal blood pressure before 20 weeks’ gestation, 5% had preeclampsia, while 7% of those with elevated blood pressure did, as did 12% of women with stage 1 hypertension and 30% of women with stage 2 hypertension. The increase in risk of preeclampsia was because of preterm preeclampsia in the women with elevated blood pressure. Preeclampsia researcher Mark Santillan, MD, PhD, of the University of Iowa in Iowa City, said in an interview that the results “open the door to considering these new blood pressure categories as a prognosticator” for preeclampsia. “This paper furthers the field by applying these new categories to hypertensive diseases in pregnancy, which are not well studied” in comparison to nonpregnant hypertensive states. Read more.
Face mask type matters when sterilizing
When sterilizing face masks, the type of mask and the method of sterilization have a bearing on subsequent filtration efficiency, according to new research published in JAMA Network Open. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks. With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%. Read more.
FDA approves first treatment for adult-onset Still’s disease
The Food and Drug Administration has expanded the indications for canakinumab (Ilaris) to include all patients with active Still’s disease older than 2 years, adding adult-onset Still’s disease (AOSD) to a previous approval for juvenile-onset Still’s disease, also known as systemic juvenile idiopathic arthritis (sJIA). That makes Ilaris the first approved treatment for AOSD. The results from a randomized, double-blind, placebo-controlled study of 36 patients with AOSD aged 22-70 years showed that the efficacy and safety data in AOSD were generally consistent with the results of a pooled analysis of sJIA patients, according to Novartis, which markets canakinumab. Read more.
Intranasal DHE shows promise in migraine
An intranasal form of dihydroergotamine (DHE) targeting the upper nasal region is safe and effective for the treatment of migraine, according to results from a phase 3 clinical trial. The new formulation could offer patients an at-home alternative to intramuscular infusions or intravenous injections currently used to deliver DHE. The STOP 301 phase 3 open-label safety and tolerability trial treated over 5,650 migraine attacks in 354 patients who self-administered INP104 for up to 52 weeks. They were provided up to three doses per week (1.45 mg in a dose of two puffs, one per nostril). A total of 66.3% of participants reported pain relief by 2 hours following a dose, and 38% had freedom from pain. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Lung ultrasound works well in children with COVID-19
researchers wrote in Pediatrics.
They also noted the benefits that modality provides over other imaging techniques.
Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy, performed an observational study of eight children aged 0-17 years who were admitted to the hospital for COVID-19 between March 8 and 26, 2020. In seven of eight patients, the findings were concordant between imaging modalities; in the remaining patient, lung ultrasound (LUS) found an interstitial B-lines pattern that was not seen on radiography. In seven patients with pathologic ultrasound findings at baseline, the improvement or resolution of the subpleural consolidations or interstitial patterns was consistent with concomitant radiologic findings.
The authors cited the benefits of using point-of-care ultrasound instead of other modalities, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” they wrote. “Secondly, when performed at the bedside, LUS allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].”
One limitation of the study is the small sample size; however, the researchers felt the high concordance still suggests LUS is a reasonable method for COVID-19 patients.
There was no external funding for this study and the investigators had no relevant financial disclosures.
SOURCE: Denina M et al. Pediatrics. 2020 Jun. doi: 10.1542/peds.2020-1157.
researchers wrote in Pediatrics.
They also noted the benefits that modality provides over other imaging techniques.
Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy, performed an observational study of eight children aged 0-17 years who were admitted to the hospital for COVID-19 between March 8 and 26, 2020. In seven of eight patients, the findings were concordant between imaging modalities; in the remaining patient, lung ultrasound (LUS) found an interstitial B-lines pattern that was not seen on radiography. In seven patients with pathologic ultrasound findings at baseline, the improvement or resolution of the subpleural consolidations or interstitial patterns was consistent with concomitant radiologic findings.
The authors cited the benefits of using point-of-care ultrasound instead of other modalities, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” they wrote. “Secondly, when performed at the bedside, LUS allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].”
One limitation of the study is the small sample size; however, the researchers felt the high concordance still suggests LUS is a reasonable method for COVID-19 patients.
There was no external funding for this study and the investigators had no relevant financial disclosures.
SOURCE: Denina M et al. Pediatrics. 2020 Jun. doi: 10.1542/peds.2020-1157.
researchers wrote in Pediatrics.
They also noted the benefits that modality provides over other imaging techniques.
Marco Denina, MD, and colleagues from the pediatric infectious diseases unit at Regina Margherita Children’s Hospital in Turin, Italy, performed an observational study of eight children aged 0-17 years who were admitted to the hospital for COVID-19 between March 8 and 26, 2020. In seven of eight patients, the findings were concordant between imaging modalities; in the remaining patient, lung ultrasound (LUS) found an interstitial B-lines pattern that was not seen on radiography. In seven patients with pathologic ultrasound findings at baseline, the improvement or resolution of the subpleural consolidations or interstitial patterns was consistent with concomitant radiologic findings.
The authors cited the benefits of using point-of-care ultrasound instead of other modalities, such as CT. “First, it may reduce the number of radiologic examinations, lowering the radiation exposure of the patients,” they wrote. “Secondly, when performed at the bedside, LUS allows for the reduction of the patient’s movement within the hospital; thus, it lowers the number of health care workers and medical devices exposed to [SARS-CoV-2].”
One limitation of the study is the small sample size; however, the researchers felt the high concordance still suggests LUS is a reasonable method for COVID-19 patients.
There was no external funding for this study and the investigators had no relevant financial disclosures.
SOURCE: Denina M et al. Pediatrics. 2020 Jun. doi: 10.1542/peds.2020-1157.
FROM PEDIATRICS
ID dermatology: Advancements, but new challenges, over 50 years
When Stephen Tyring, MD, PhD, an infectious disease dermatologist, started his career in the early 1980s, he said “we were diagnosing Kaposi’s sarcoma right and left. We would see a new case every day or two.”
It was the early days of the HIV/AIDS epidemic, and dermatologists were at the forefront because HIV/AIDS often presented with skin manifestations. Dr. Tyring, clinical professor in the departments of dermatology, microbiology & molecular genetics and internal medicine at the University of Texas Health Science Center, Houston, and his colleagues referred Kaposi’s patients for chemotherapy and radiation, but the outlook was often grim, especially if lesions developed in the lungs.
Dermatologist don’t see much Kaposi’s anymore because of highly effective treatments for HIV.
Members of the original editorial advisory board saw it coming. In a feature in which board members provided their prediction for the 1970s that appeared in the first issue, New York dermatologist Norman Orentreich, MD, counted the “probable introduction of virucidal agents” as one of the “significant advances or changes that I foresee in the next 10 years.” J. Lamar Callaway, MD, professor of dermatology at Duke University, Durham, N.C., predicted that “the next 10 years should develop effective anti-viral agents for warts, herpes simplex, and herpes zoster.”
To celebrate the 50th anniversary of Dermatology News, we are looking back at how the field has changed since that first issue. The focus this month is infectious disease. There’s a lot to be grateful for but there are also challenges like antibiotic resistance that weren’t on the radar screens of Dr. Orentreich, Dr. Callaway, and their peers in 1970.
All in all, “the only thing I wish we did the old way is sit at the bedside and talk to patients more. We rely so much on technology now that we sometimes lose the art of medicine, which is comforting to the patient,” said Theodore Rosen, MD, an ID dermatologist and professor of dermatology at Baylor College of Medicine, Houston, who’s been in practice for 42 years.
“A lot of advancements against herpes viruses”
One of the biggest wins for ID dermatology over the last 5 decades has been the management of herpes, both herpes simplex virus 1 and 2, as well as herpes zoster virus. It started with the approval of acyclovir in 1981. Before then, “we had no direct therapy for genital herpes, herpes zoster, or disseminated herpes in immunosuppressed or cancer patients,” Dr. Rosen said.
“I can remember doing an interview with Good Morning America when I gave the first IV dose of acyclovir in the city of Houston for really bad disseminated herpes” in an HIV patient, he said, and it worked.
Two derivatives, valacyclovir and famciclovir, became available in the mid-1990s, so today “we have three drugs and some others at the periphery that are all highly effective not only” against herpes, but also for preventing outbreaks; valacyclovir can even prevent asymptomatic shedding, therefore possibly preventing new infections. “That’s a concept we didn’t even have 40 years ago,” Dr. Rosen said.
Cidofovir has also made a difference. The IV formulation was approved for AIDS-associated cytomegalovirus retinitis in 1996 but discontinued a few years later amid concerns of severe renal toxicity. It’s found a new home in dermatology since then, explained ID dermatologist Carrie Kovarik, MD, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
Dermatologists see acyclovir-resistant herpes “heaped up on the genitals in HIV patients,” and there weren’t many options in the past. A few years ago, “we [tried] injecting cidofovir directly into the skin lesions, and it’s been remarkably successful. It is a good way to treat these lesions” if dermatologists can get it compounded, she said.
Shingles vaccines, first the live attenuated zoster vaccine (Zostavax) approved by the Food and Drug Administration in 2006 and the more effective recombinant zoster vaccine (Shingrix) approved in 2017, have also had a significant impact.
Dr. Rosen remembers what it was like when he first started practicing over 40 years ago. Not uncommonly, “we saw horrible cases of shingles,” including one in his uncle, who was left with permanent hand pain long after the rash subsided.
Today, “I see much less shingles, and when I do see it, it’s in a much-attenuated form. [Shingrix], even if it doesn’t prevent the disease, often prevents postherpetic neuralgia,” he said.
Also, with pediatric vaccinations against chicken pox, “we’re probably going to see a whole new generation without shingles, which is huge. We’ve made a lot of advancements against herpes viruses,” Dr. Kovarik said.
“We finally found something that helps”
“We’ve [also] come a really long way with genital wart treatment,” Dr. Kovarik said.
It started with approval of topical imiquimod in 1997. “Before that, we were just killing one wart here and one wart there” but they would often come back and pop up in other areas. Injectable interferon was an option at the time, but people didn’t like all the needles.
With imiquimod, “we finally [had] a way to target HPV [human papillomavirus] and not just scrape” or freeze one wart at a time, and “we were able to generate an inflammatory response in the whole area to clear the virus.” Working with HIV patients, “I see sheets and sheets of confluent warts throughout the whole genital area; to try to freeze that is impossible. Now I have a way to get rid of [genital] warts and keep them away even if you have a big cluster,” she said.
“Sometimes, we’ll do both liquid nitrogen and imiquimod. That’s a good way to tackle people who have a high burden of warts,” Dr. Kovarik noted. Other effective treatments have come out as well, including an ointment formulation of sinecatechins, extracted from green tea, “but you have to put it on several times a day, and insurance companies don’t cover it often,” she said.
Intralesional cidofovir is also proving to be boon for potentially malignant refractory warts in HIV and transplant patients. “It’s an incredible treatment. We can inject that antiviral into warts and get rid of them. We finally found something that helps” these people, Dr. Kovarik said.
The HPV vaccine Gardasil is making a difference, as well. In addition to cervical dysplasia and anogenital cancers, it protects against two condyloma strains. Dr. Rosen said he’s seeing fewer cases of genital warts now than when he started practicing, likely because of the vaccine.
“Organisms that weren’t pathogens are now pathogens”
Antibiotic resistance probably tops the list for what’s changed in a bad way in ID dermatology since 1970. Dr. Rosen remembers at the start of his career that “we never worried about antibiotic resistance. We’d put people on antibiotics for acne, rosacea, and we’d keep them on them for 3 years, 6 years”; resistance wasn’t on the radar screen and was not mentioned once in the first issue of Dermatology News, which was packed with articles and ran 24 pages.
The situation is different now. Driven by decades of overuse in agriculture and the medical system, antibiotic resistance is a concern throughout medicine, and unfortunately, “we have not come nearly as far as fast with antibiotics,” at least the ones dermatologists use, “as we have with antivirals,” Dr. Tyring said.
For instance, methicillin-resistant Staphylococcus aureus (MRSA), first described in the United States in 1968, is “no longer the exception to the rule, but the rule” itself, he said, with carbuncles, furuncles, and abscesses not infrequently growing out MRSA. There are also new drug-resistant forms of old problems like gonorrhea and tuberculosis, among other developments, and impetigo has shifted since 1970 from mostly a Streptococcus infection easily treated with penicillin to often a Staphylococcus disease that’s resistant to it. There’s also been a steady march of new pathogens, including the latest one, SARS-CoV-2, the virus that causes COVID-19, which has been recognized as having a variety of skin manifestations.
“No matter how smart we think we are, nature has a way of putting us back in our place,” Dr. Rosen said.
The bright spot is that “we’ve become very adept at identifying and characterizing” microbes “based on techniques we didn’t even have when I started practicing,” such as polymerase chain reaction. “It has taken a lot of guess work out of treating infectious diseases,” he said.
The widespread use of immunosuppressives such as cyclophosphamide, mycophenolate, azathioprine, rituximab, and other agents used in conjunction with solid organ transplantation, has also been a challenge. “We are seeing infections with really odd organisms. Just recently, I had a patient with fusarium in the skin; it’s a fungus that lives in the dirt. I saw a patient with a species of algae” that normally lives in stagnant water, he commented. “We used to get [things like that] back on reports, and we’d throw them away. You can’t do that anymore. Organisms that weren’t pathogens in the past are now pathogens,” particularly in immunosuppressed people, Dr. Rosen said.
Venereologists no more
There’s been another big change in the field. “Back in the not too distant past, dermatologists in the U.S. were referred to as ‘dermatologist-venereologists.’ ” It goes back to the time when syphilis wasn’t diagnosed and treated early, so patients often presented with secondary skin complications and went to dermatologists for help. As a result, “dermatologists became the most experienced at treating it,” Dr. Tyring said.
That’s faded from practice. Part of the reason is that as late as 2000, syphilis seemed to be on the way out; the Centers for Disease and Control and Prevention even raised the possibility of elimination. Dermatologists turned their attention to other areas.
It might have been short-sighted, Dr. Rosen said. Syphilis has made a strong comeback, and drug-resistant gonorrhea has also emerged globally and in at least a few states. No other medical field has stepped in to take up the slack. “Ob.gyns. are busy delivering babies, ID [physicians are] concerned about HIV, and urologists are worried about kidney stones and cancer.” Other than herpes and genital warts, “we have not done well” with management of sexually transmitted diseases, he said.
“I could sense” his frustration
The first issue of Dermatology News carried an article and photospread about scabies that could run today, except that topical permethrin and oral ivermectin have largely replaced benzyl benzoate and sulfur ointments for treatment in the United States. In the article, Scottish dermatologist J. O’D. Alexander, MD, called scabies “the scourge of mankind” and blamed it’s prevalence on “an offhand attitude to the disease which makes control very difficult.”
“I could sense this man’s frustration that people were not recognizing scabies,” Dr. Kovarik said, and it’s no closer to being eradicated than it was in 1970. “It’s still around, and we see it in our clinics. It’s a horrible disease in kids we see in dermatology not infrequently,” and treatment has only advanced a bit.
The article highlights what hasn’t changed much in ID dermatology over the years. Common warts are another one. “With all the evolution in medicine, we don’t have any better treatments approved for common warts than we ever had.” Injecting cidofovir “works great,” but access is a problem, Dr. Tyring said.
Onychomycosis has also proven a tough nut to crack. Readers back in 1970 counted the introduction of the antifungal, griseofulvin, as a major advancement in the 1960s; it’s still a go-to for tinea capitis, but it didn’t work very well for toenail fungus. Terbinafine (Lamisil), approved in 1993, and subsequent developments have helped, but the field still awaits more effective options; a few potential new agents are in the pipeline.
Although there have been major advancements for serious systemic fungal infections, “we’ve mainly seen small steps forward” in ID dermatology, Dr. Tyring said.
Dr. Tyring, Dr. Kovarik, and Dr. Rosen said they had no relevant disclosures.
When Stephen Tyring, MD, PhD, an infectious disease dermatologist, started his career in the early 1980s, he said “we were diagnosing Kaposi’s sarcoma right and left. We would see a new case every day or two.”
It was the early days of the HIV/AIDS epidemic, and dermatologists were at the forefront because HIV/AIDS often presented with skin manifestations. Dr. Tyring, clinical professor in the departments of dermatology, microbiology & molecular genetics and internal medicine at the University of Texas Health Science Center, Houston, and his colleagues referred Kaposi’s patients for chemotherapy and radiation, but the outlook was often grim, especially if lesions developed in the lungs.
Dermatologist don’t see much Kaposi’s anymore because of highly effective treatments for HIV.
Members of the original editorial advisory board saw it coming. In a feature in which board members provided their prediction for the 1970s that appeared in the first issue, New York dermatologist Norman Orentreich, MD, counted the “probable introduction of virucidal agents” as one of the “significant advances or changes that I foresee in the next 10 years.” J. Lamar Callaway, MD, professor of dermatology at Duke University, Durham, N.C., predicted that “the next 10 years should develop effective anti-viral agents for warts, herpes simplex, and herpes zoster.”
To celebrate the 50th anniversary of Dermatology News, we are looking back at how the field has changed since that first issue. The focus this month is infectious disease. There’s a lot to be grateful for but there are also challenges like antibiotic resistance that weren’t on the radar screens of Dr. Orentreich, Dr. Callaway, and their peers in 1970.
All in all, “the only thing I wish we did the old way is sit at the bedside and talk to patients more. We rely so much on technology now that we sometimes lose the art of medicine, which is comforting to the patient,” said Theodore Rosen, MD, an ID dermatologist and professor of dermatology at Baylor College of Medicine, Houston, who’s been in practice for 42 years.
“A lot of advancements against herpes viruses”
One of the biggest wins for ID dermatology over the last 5 decades has been the management of herpes, both herpes simplex virus 1 and 2, as well as herpes zoster virus. It started with the approval of acyclovir in 1981. Before then, “we had no direct therapy for genital herpes, herpes zoster, or disseminated herpes in immunosuppressed or cancer patients,” Dr. Rosen said.
“I can remember doing an interview with Good Morning America when I gave the first IV dose of acyclovir in the city of Houston for really bad disseminated herpes” in an HIV patient, he said, and it worked.
Two derivatives, valacyclovir and famciclovir, became available in the mid-1990s, so today “we have three drugs and some others at the periphery that are all highly effective not only” against herpes, but also for preventing outbreaks; valacyclovir can even prevent asymptomatic shedding, therefore possibly preventing new infections. “That’s a concept we didn’t even have 40 years ago,” Dr. Rosen said.
Cidofovir has also made a difference. The IV formulation was approved for AIDS-associated cytomegalovirus retinitis in 1996 but discontinued a few years later amid concerns of severe renal toxicity. It’s found a new home in dermatology since then, explained ID dermatologist Carrie Kovarik, MD, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
Dermatologists see acyclovir-resistant herpes “heaped up on the genitals in HIV patients,” and there weren’t many options in the past. A few years ago, “we [tried] injecting cidofovir directly into the skin lesions, and it’s been remarkably successful. It is a good way to treat these lesions” if dermatologists can get it compounded, she said.
Shingles vaccines, first the live attenuated zoster vaccine (Zostavax) approved by the Food and Drug Administration in 2006 and the more effective recombinant zoster vaccine (Shingrix) approved in 2017, have also had a significant impact.
Dr. Rosen remembers what it was like when he first started practicing over 40 years ago. Not uncommonly, “we saw horrible cases of shingles,” including one in his uncle, who was left with permanent hand pain long after the rash subsided.
Today, “I see much less shingles, and when I do see it, it’s in a much-attenuated form. [Shingrix], even if it doesn’t prevent the disease, often prevents postherpetic neuralgia,” he said.
Also, with pediatric vaccinations against chicken pox, “we’re probably going to see a whole new generation without shingles, which is huge. We’ve made a lot of advancements against herpes viruses,” Dr. Kovarik said.
“We finally found something that helps”
“We’ve [also] come a really long way with genital wart treatment,” Dr. Kovarik said.
It started with approval of topical imiquimod in 1997. “Before that, we were just killing one wart here and one wart there” but they would often come back and pop up in other areas. Injectable interferon was an option at the time, but people didn’t like all the needles.
With imiquimod, “we finally [had] a way to target HPV [human papillomavirus] and not just scrape” or freeze one wart at a time, and “we were able to generate an inflammatory response in the whole area to clear the virus.” Working with HIV patients, “I see sheets and sheets of confluent warts throughout the whole genital area; to try to freeze that is impossible. Now I have a way to get rid of [genital] warts and keep them away even if you have a big cluster,” she said.
“Sometimes, we’ll do both liquid nitrogen and imiquimod. That’s a good way to tackle people who have a high burden of warts,” Dr. Kovarik noted. Other effective treatments have come out as well, including an ointment formulation of sinecatechins, extracted from green tea, “but you have to put it on several times a day, and insurance companies don’t cover it often,” she said.
Intralesional cidofovir is also proving to be boon for potentially malignant refractory warts in HIV and transplant patients. “It’s an incredible treatment. We can inject that antiviral into warts and get rid of them. We finally found something that helps” these people, Dr. Kovarik said.
The HPV vaccine Gardasil is making a difference, as well. In addition to cervical dysplasia and anogenital cancers, it protects against two condyloma strains. Dr. Rosen said he’s seeing fewer cases of genital warts now than when he started practicing, likely because of the vaccine.
“Organisms that weren’t pathogens are now pathogens”
Antibiotic resistance probably tops the list for what’s changed in a bad way in ID dermatology since 1970. Dr. Rosen remembers at the start of his career that “we never worried about antibiotic resistance. We’d put people on antibiotics for acne, rosacea, and we’d keep them on them for 3 years, 6 years”; resistance wasn’t on the radar screen and was not mentioned once in the first issue of Dermatology News, which was packed with articles and ran 24 pages.
The situation is different now. Driven by decades of overuse in agriculture and the medical system, antibiotic resistance is a concern throughout medicine, and unfortunately, “we have not come nearly as far as fast with antibiotics,” at least the ones dermatologists use, “as we have with antivirals,” Dr. Tyring said.
For instance, methicillin-resistant Staphylococcus aureus (MRSA), first described in the United States in 1968, is “no longer the exception to the rule, but the rule” itself, he said, with carbuncles, furuncles, and abscesses not infrequently growing out MRSA. There are also new drug-resistant forms of old problems like gonorrhea and tuberculosis, among other developments, and impetigo has shifted since 1970 from mostly a Streptococcus infection easily treated with penicillin to often a Staphylococcus disease that’s resistant to it. There’s also been a steady march of new pathogens, including the latest one, SARS-CoV-2, the virus that causes COVID-19, which has been recognized as having a variety of skin manifestations.
“No matter how smart we think we are, nature has a way of putting us back in our place,” Dr. Rosen said.
The bright spot is that “we’ve become very adept at identifying and characterizing” microbes “based on techniques we didn’t even have when I started practicing,” such as polymerase chain reaction. “It has taken a lot of guess work out of treating infectious diseases,” he said.
The widespread use of immunosuppressives such as cyclophosphamide, mycophenolate, azathioprine, rituximab, and other agents used in conjunction with solid organ transplantation, has also been a challenge. “We are seeing infections with really odd organisms. Just recently, I had a patient with fusarium in the skin; it’s a fungus that lives in the dirt. I saw a patient with a species of algae” that normally lives in stagnant water, he commented. “We used to get [things like that] back on reports, and we’d throw them away. You can’t do that anymore. Organisms that weren’t pathogens in the past are now pathogens,” particularly in immunosuppressed people, Dr. Rosen said.
Venereologists no more
There’s been another big change in the field. “Back in the not too distant past, dermatologists in the U.S. were referred to as ‘dermatologist-venereologists.’ ” It goes back to the time when syphilis wasn’t diagnosed and treated early, so patients often presented with secondary skin complications and went to dermatologists for help. As a result, “dermatologists became the most experienced at treating it,” Dr. Tyring said.
That’s faded from practice. Part of the reason is that as late as 2000, syphilis seemed to be on the way out; the Centers for Disease and Control and Prevention even raised the possibility of elimination. Dermatologists turned their attention to other areas.
It might have been short-sighted, Dr. Rosen said. Syphilis has made a strong comeback, and drug-resistant gonorrhea has also emerged globally and in at least a few states. No other medical field has stepped in to take up the slack. “Ob.gyns. are busy delivering babies, ID [physicians are] concerned about HIV, and urologists are worried about kidney stones and cancer.” Other than herpes and genital warts, “we have not done well” with management of sexually transmitted diseases, he said.
“I could sense” his frustration
The first issue of Dermatology News carried an article and photospread about scabies that could run today, except that topical permethrin and oral ivermectin have largely replaced benzyl benzoate and sulfur ointments for treatment in the United States. In the article, Scottish dermatologist J. O’D. Alexander, MD, called scabies “the scourge of mankind” and blamed it’s prevalence on “an offhand attitude to the disease which makes control very difficult.”
“I could sense this man’s frustration that people were not recognizing scabies,” Dr. Kovarik said, and it’s no closer to being eradicated than it was in 1970. “It’s still around, and we see it in our clinics. It’s a horrible disease in kids we see in dermatology not infrequently,” and treatment has only advanced a bit.
The article highlights what hasn’t changed much in ID dermatology over the years. Common warts are another one. “With all the evolution in medicine, we don’t have any better treatments approved for common warts than we ever had.” Injecting cidofovir “works great,” but access is a problem, Dr. Tyring said.
Onychomycosis has also proven a tough nut to crack. Readers back in 1970 counted the introduction of the antifungal, griseofulvin, as a major advancement in the 1960s; it’s still a go-to for tinea capitis, but it didn’t work very well for toenail fungus. Terbinafine (Lamisil), approved in 1993, and subsequent developments have helped, but the field still awaits more effective options; a few potential new agents are in the pipeline.
Although there have been major advancements for serious systemic fungal infections, “we’ve mainly seen small steps forward” in ID dermatology, Dr. Tyring said.
Dr. Tyring, Dr. Kovarik, and Dr. Rosen said they had no relevant disclosures.
When Stephen Tyring, MD, PhD, an infectious disease dermatologist, started his career in the early 1980s, he said “we were diagnosing Kaposi’s sarcoma right and left. We would see a new case every day or two.”
It was the early days of the HIV/AIDS epidemic, and dermatologists were at the forefront because HIV/AIDS often presented with skin manifestations. Dr. Tyring, clinical professor in the departments of dermatology, microbiology & molecular genetics and internal medicine at the University of Texas Health Science Center, Houston, and his colleagues referred Kaposi’s patients for chemotherapy and radiation, but the outlook was often grim, especially if lesions developed in the lungs.
Dermatologist don’t see much Kaposi’s anymore because of highly effective treatments for HIV.
Members of the original editorial advisory board saw it coming. In a feature in which board members provided their prediction for the 1970s that appeared in the first issue, New York dermatologist Norman Orentreich, MD, counted the “probable introduction of virucidal agents” as one of the “significant advances or changes that I foresee in the next 10 years.” J. Lamar Callaway, MD, professor of dermatology at Duke University, Durham, N.C., predicted that “the next 10 years should develop effective anti-viral agents for warts, herpes simplex, and herpes zoster.”
To celebrate the 50th anniversary of Dermatology News, we are looking back at how the field has changed since that first issue. The focus this month is infectious disease. There’s a lot to be grateful for but there are also challenges like antibiotic resistance that weren’t on the radar screens of Dr. Orentreich, Dr. Callaway, and their peers in 1970.
All in all, “the only thing I wish we did the old way is sit at the bedside and talk to patients more. We rely so much on technology now that we sometimes lose the art of medicine, which is comforting to the patient,” said Theodore Rosen, MD, an ID dermatologist and professor of dermatology at Baylor College of Medicine, Houston, who’s been in practice for 42 years.
“A lot of advancements against herpes viruses”
One of the biggest wins for ID dermatology over the last 5 decades has been the management of herpes, both herpes simplex virus 1 and 2, as well as herpes zoster virus. It started with the approval of acyclovir in 1981. Before then, “we had no direct therapy for genital herpes, herpes zoster, or disseminated herpes in immunosuppressed or cancer patients,” Dr. Rosen said.
“I can remember doing an interview with Good Morning America when I gave the first IV dose of acyclovir in the city of Houston for really bad disseminated herpes” in an HIV patient, he said, and it worked.
Two derivatives, valacyclovir and famciclovir, became available in the mid-1990s, so today “we have three drugs and some others at the periphery that are all highly effective not only” against herpes, but also for preventing outbreaks; valacyclovir can even prevent asymptomatic shedding, therefore possibly preventing new infections. “That’s a concept we didn’t even have 40 years ago,” Dr. Rosen said.
Cidofovir has also made a difference. The IV formulation was approved for AIDS-associated cytomegalovirus retinitis in 1996 but discontinued a few years later amid concerns of severe renal toxicity. It’s found a new home in dermatology since then, explained ID dermatologist Carrie Kovarik, MD, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
Dermatologists see acyclovir-resistant herpes “heaped up on the genitals in HIV patients,” and there weren’t many options in the past. A few years ago, “we [tried] injecting cidofovir directly into the skin lesions, and it’s been remarkably successful. It is a good way to treat these lesions” if dermatologists can get it compounded, she said.
Shingles vaccines, first the live attenuated zoster vaccine (Zostavax) approved by the Food and Drug Administration in 2006 and the more effective recombinant zoster vaccine (Shingrix) approved in 2017, have also had a significant impact.
Dr. Rosen remembers what it was like when he first started practicing over 40 years ago. Not uncommonly, “we saw horrible cases of shingles,” including one in his uncle, who was left with permanent hand pain long after the rash subsided.
Today, “I see much less shingles, and when I do see it, it’s in a much-attenuated form. [Shingrix], even if it doesn’t prevent the disease, often prevents postherpetic neuralgia,” he said.
Also, with pediatric vaccinations against chicken pox, “we’re probably going to see a whole new generation without shingles, which is huge. We’ve made a lot of advancements against herpes viruses,” Dr. Kovarik said.
“We finally found something that helps”
“We’ve [also] come a really long way with genital wart treatment,” Dr. Kovarik said.
It started with approval of topical imiquimod in 1997. “Before that, we were just killing one wart here and one wart there” but they would often come back and pop up in other areas. Injectable interferon was an option at the time, but people didn’t like all the needles.
With imiquimod, “we finally [had] a way to target HPV [human papillomavirus] and not just scrape” or freeze one wart at a time, and “we were able to generate an inflammatory response in the whole area to clear the virus.” Working with HIV patients, “I see sheets and sheets of confluent warts throughout the whole genital area; to try to freeze that is impossible. Now I have a way to get rid of [genital] warts and keep them away even if you have a big cluster,” she said.
“Sometimes, we’ll do both liquid nitrogen and imiquimod. That’s a good way to tackle people who have a high burden of warts,” Dr. Kovarik noted. Other effective treatments have come out as well, including an ointment formulation of sinecatechins, extracted from green tea, “but you have to put it on several times a day, and insurance companies don’t cover it often,” she said.
Intralesional cidofovir is also proving to be boon for potentially malignant refractory warts in HIV and transplant patients. “It’s an incredible treatment. We can inject that antiviral into warts and get rid of them. We finally found something that helps” these people, Dr. Kovarik said.
The HPV vaccine Gardasil is making a difference, as well. In addition to cervical dysplasia and anogenital cancers, it protects against two condyloma strains. Dr. Rosen said he’s seeing fewer cases of genital warts now than when he started practicing, likely because of the vaccine.
“Organisms that weren’t pathogens are now pathogens”
Antibiotic resistance probably tops the list for what’s changed in a bad way in ID dermatology since 1970. Dr. Rosen remembers at the start of his career that “we never worried about antibiotic resistance. We’d put people on antibiotics for acne, rosacea, and we’d keep them on them for 3 years, 6 years”; resistance wasn’t on the radar screen and was not mentioned once in the first issue of Dermatology News, which was packed with articles and ran 24 pages.
The situation is different now. Driven by decades of overuse in agriculture and the medical system, antibiotic resistance is a concern throughout medicine, and unfortunately, “we have not come nearly as far as fast with antibiotics,” at least the ones dermatologists use, “as we have with antivirals,” Dr. Tyring said.
For instance, methicillin-resistant Staphylococcus aureus (MRSA), first described in the United States in 1968, is “no longer the exception to the rule, but the rule” itself, he said, with carbuncles, furuncles, and abscesses not infrequently growing out MRSA. There are also new drug-resistant forms of old problems like gonorrhea and tuberculosis, among other developments, and impetigo has shifted since 1970 from mostly a Streptococcus infection easily treated with penicillin to often a Staphylococcus disease that’s resistant to it. There’s also been a steady march of new pathogens, including the latest one, SARS-CoV-2, the virus that causes COVID-19, which has been recognized as having a variety of skin manifestations.
“No matter how smart we think we are, nature has a way of putting us back in our place,” Dr. Rosen said.
The bright spot is that “we’ve become very adept at identifying and characterizing” microbes “based on techniques we didn’t even have when I started practicing,” such as polymerase chain reaction. “It has taken a lot of guess work out of treating infectious diseases,” he said.
The widespread use of immunosuppressives such as cyclophosphamide, mycophenolate, azathioprine, rituximab, and other agents used in conjunction with solid organ transplantation, has also been a challenge. “We are seeing infections with really odd organisms. Just recently, I had a patient with fusarium in the skin; it’s a fungus that lives in the dirt. I saw a patient with a species of algae” that normally lives in stagnant water, he commented. “We used to get [things like that] back on reports, and we’d throw them away. You can’t do that anymore. Organisms that weren’t pathogens in the past are now pathogens,” particularly in immunosuppressed people, Dr. Rosen said.
Venereologists no more
There’s been another big change in the field. “Back in the not too distant past, dermatologists in the U.S. were referred to as ‘dermatologist-venereologists.’ ” It goes back to the time when syphilis wasn’t diagnosed and treated early, so patients often presented with secondary skin complications and went to dermatologists for help. As a result, “dermatologists became the most experienced at treating it,” Dr. Tyring said.
That’s faded from practice. Part of the reason is that as late as 2000, syphilis seemed to be on the way out; the Centers for Disease and Control and Prevention even raised the possibility of elimination. Dermatologists turned their attention to other areas.
It might have been short-sighted, Dr. Rosen said. Syphilis has made a strong comeback, and drug-resistant gonorrhea has also emerged globally and in at least a few states. No other medical field has stepped in to take up the slack. “Ob.gyns. are busy delivering babies, ID [physicians are] concerned about HIV, and urologists are worried about kidney stones and cancer.” Other than herpes and genital warts, “we have not done well” with management of sexually transmitted diseases, he said.
“I could sense” his frustration
The first issue of Dermatology News carried an article and photospread about scabies that could run today, except that topical permethrin and oral ivermectin have largely replaced benzyl benzoate and sulfur ointments for treatment in the United States. In the article, Scottish dermatologist J. O’D. Alexander, MD, called scabies “the scourge of mankind” and blamed it’s prevalence on “an offhand attitude to the disease which makes control very difficult.”
“I could sense this man’s frustration that people were not recognizing scabies,” Dr. Kovarik said, and it’s no closer to being eradicated than it was in 1970. “It’s still around, and we see it in our clinics. It’s a horrible disease in kids we see in dermatology not infrequently,” and treatment has only advanced a bit.
The article highlights what hasn’t changed much in ID dermatology over the years. Common warts are another one. “With all the evolution in medicine, we don’t have any better treatments approved for common warts than we ever had.” Injecting cidofovir “works great,” but access is a problem, Dr. Tyring said.
Onychomycosis has also proven a tough nut to crack. Readers back in 1970 counted the introduction of the antifungal, griseofulvin, as a major advancement in the 1960s; it’s still a go-to for tinea capitis, but it didn’t work very well for toenail fungus. Terbinafine (Lamisil), approved in 1993, and subsequent developments have helped, but the field still awaits more effective options; a few potential new agents are in the pipeline.
Although there have been major advancements for serious systemic fungal infections, “we’ve mainly seen small steps forward” in ID dermatology, Dr. Tyring said.
Dr. Tyring, Dr. Kovarik, and Dr. Rosen said they had no relevant disclosures.
Face mask type matters when sterilizing, study finds
according to researchers. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks.
With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%.
In a research letter published online June 15 in JAMA Network Open, researchers from the University of Oklahoma Health Sciences Center, Oklahoma City, report the results of a study of the two sterilization techniques on the pressure drop and filtration efficiency of N95, KN95, and surgical face masks.
“The H2O2 treatment showed a small effect on the overall filtration efficiency of the tested masks, but the ClO2 treatment showed marked reduction in the overall filtration efficiency of the KN95s and surgical face masks. All pressure drop changes were within the acceptable range,” the researchers write.
The study did not evaluate the effect of repeated sterilizations on face masks.
Five masks of each type were sterilized with either H2O2 or ClO2. Masks were then placed in a test chamber, and a salt aerosol was nebulized to assess both upstream and downstream filtration as well as pressure drop. The researchers used a mobility particle sizer to measure particle number concentration from 16.8 nm to 514 nm. An acceptable pressure drop was defined as a drop of less than 1.38 inches of water (35 mm) for inhalation.
Although pressure drop changes were within the acceptable range for all three mask types following sterilization with either method, H2O2 sterilization yielded the least reduction in filtration efficacy in all cases. After sterilization with H2O2, filtration efficiencies were 96.6%, 97.1%, and 91.6% for the N95s, KN95s, and the surgical face masks, respectively. In contrast, filtration efficiencies after ClO2 sterilization were 95.1%, 76.2%, and 77.9%, respectively.
The researchers note that, although overall filtration efficiency was maintained with ClO2 sterilization, there was a significant drop in efficiency with respect to particles of approximately 300 nm (0.3 microns) in size. For particles of that size, mean filtration efficiency decreased to 86.2% for N95s, 40.8% for KN95s, and 47.1% for surgical face masks.
The testing described in the report is “quite affordable at $350 per mask type, so it is hard to imagine any health care provider cannot set aside a small budget to conduct such an important test,” author Evan Floyd, PhD, told Medscape Medical News.
Given the high demand for effective face masks and the current risk for counterfeit products, Floyd suggested that individual facilities test all masks intended for use by healthcare workers before and after sterilization procedures.
“However, if for some reason testing is not an option, we would recommend sticking to established brands and suppliers, perhaps reach out to your state health department or a local representative of the strategic stockpile of PPE,” he noted.
The authors acknowledge that further studies using a larger sample size and a greater variety of masks, as well as studies to evaluate different sterilization techniques, are required. Further, “measuring the respirator’s filtration efficiency by aerosol size instead of only measuring the overall filtration efficiency” should also be considered. Such an approach would enable researchers to evaluate the degree to which masks protect against specific infectious agents.
This article first appeared on Medscape.com.
according to researchers. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks.
With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%.
In a research letter published online June 15 in JAMA Network Open, researchers from the University of Oklahoma Health Sciences Center, Oklahoma City, report the results of a study of the two sterilization techniques on the pressure drop and filtration efficiency of N95, KN95, and surgical face masks.
“The H2O2 treatment showed a small effect on the overall filtration efficiency of the tested masks, but the ClO2 treatment showed marked reduction in the overall filtration efficiency of the KN95s and surgical face masks. All pressure drop changes were within the acceptable range,” the researchers write.
The study did not evaluate the effect of repeated sterilizations on face masks.
Five masks of each type were sterilized with either H2O2 or ClO2. Masks were then placed in a test chamber, and a salt aerosol was nebulized to assess both upstream and downstream filtration as well as pressure drop. The researchers used a mobility particle sizer to measure particle number concentration from 16.8 nm to 514 nm. An acceptable pressure drop was defined as a drop of less than 1.38 inches of water (35 mm) for inhalation.
Although pressure drop changes were within the acceptable range for all three mask types following sterilization with either method, H2O2 sterilization yielded the least reduction in filtration efficacy in all cases. After sterilization with H2O2, filtration efficiencies were 96.6%, 97.1%, and 91.6% for the N95s, KN95s, and the surgical face masks, respectively. In contrast, filtration efficiencies after ClO2 sterilization were 95.1%, 76.2%, and 77.9%, respectively.
The researchers note that, although overall filtration efficiency was maintained with ClO2 sterilization, there was a significant drop in efficiency with respect to particles of approximately 300 nm (0.3 microns) in size. For particles of that size, mean filtration efficiency decreased to 86.2% for N95s, 40.8% for KN95s, and 47.1% for surgical face masks.
The testing described in the report is “quite affordable at $350 per mask type, so it is hard to imagine any health care provider cannot set aside a small budget to conduct such an important test,” author Evan Floyd, PhD, told Medscape Medical News.
Given the high demand for effective face masks and the current risk for counterfeit products, Floyd suggested that individual facilities test all masks intended for use by healthcare workers before and after sterilization procedures.
“However, if for some reason testing is not an option, we would recommend sticking to established brands and suppliers, perhaps reach out to your state health department or a local representative of the strategic stockpile of PPE,” he noted.
The authors acknowledge that further studies using a larger sample size and a greater variety of masks, as well as studies to evaluate different sterilization techniques, are required. Further, “measuring the respirator’s filtration efficiency by aerosol size instead of only measuring the overall filtration efficiency” should also be considered. Such an approach would enable researchers to evaluate the degree to which masks protect against specific infectious agents.
This article first appeared on Medscape.com.
according to researchers. The greatest reduction in filtration efficiency after sterilization occurred with surgical face masks.
With plasma vapor hydrogen peroxide (H2O2) sterilization, filtration efficiency of N95 and KN95 masks was maintained at more than 95%, but for surgical face masks, filtration efficiency was reduced to less than 95%. With chlorine dioxide (ClO2) sterilization, on the other hand, filtration efficiency was maintained at above 95% for N95 masks, but for KN95 and surgical face masks, filtration efficiency was reduced to less than 80%.
In a research letter published online June 15 in JAMA Network Open, researchers from the University of Oklahoma Health Sciences Center, Oklahoma City, report the results of a study of the two sterilization techniques on the pressure drop and filtration efficiency of N95, KN95, and surgical face masks.
“The H2O2 treatment showed a small effect on the overall filtration efficiency of the tested masks, but the ClO2 treatment showed marked reduction in the overall filtration efficiency of the KN95s and surgical face masks. All pressure drop changes were within the acceptable range,” the researchers write.
The study did not evaluate the effect of repeated sterilizations on face masks.
Five masks of each type were sterilized with either H2O2 or ClO2. Masks were then placed in a test chamber, and a salt aerosol was nebulized to assess both upstream and downstream filtration as well as pressure drop. The researchers used a mobility particle sizer to measure particle number concentration from 16.8 nm to 514 nm. An acceptable pressure drop was defined as a drop of less than 1.38 inches of water (35 mm) for inhalation.
Although pressure drop changes were within the acceptable range for all three mask types following sterilization with either method, H2O2 sterilization yielded the least reduction in filtration efficacy in all cases. After sterilization with H2O2, filtration efficiencies were 96.6%, 97.1%, and 91.6% for the N95s, KN95s, and the surgical face masks, respectively. In contrast, filtration efficiencies after ClO2 sterilization were 95.1%, 76.2%, and 77.9%, respectively.
The researchers note that, although overall filtration efficiency was maintained with ClO2 sterilization, there was a significant drop in efficiency with respect to particles of approximately 300 nm (0.3 microns) in size. For particles of that size, mean filtration efficiency decreased to 86.2% for N95s, 40.8% for KN95s, and 47.1% for surgical face masks.
The testing described in the report is “quite affordable at $350 per mask type, so it is hard to imagine any health care provider cannot set aside a small budget to conduct such an important test,” author Evan Floyd, PhD, told Medscape Medical News.
Given the high demand for effective face masks and the current risk for counterfeit products, Floyd suggested that individual facilities test all masks intended for use by healthcare workers before and after sterilization procedures.
“However, if for some reason testing is not an option, we would recommend sticking to established brands and suppliers, perhaps reach out to your state health department or a local representative of the strategic stockpile of PPE,” he noted.
The authors acknowledge that further studies using a larger sample size and a greater variety of masks, as well as studies to evaluate different sterilization techniques, are required. Further, “measuring the respirator’s filtration efficiency by aerosol size instead of only measuring the overall filtration efficiency” should also be considered. Such an approach would enable researchers to evaluate the degree to which masks protect against specific infectious agents.
This article first appeared on Medscape.com.
Study compares pulse vs. continuous therapy for dermatophyte toenail onychomycosis
There appear to be results from a systematic review and network meta-analysis showed.
“Previous meta-analyses of pulse and continuous therapies have generated ambiguous results,” study authors led by Aditya K. Gupta, MD, PhD, wrote in a poster abstract presented at the virtual annual meeting of the American Academy of Dermatology. “There are few head-to-head clinical studies and no meta-analyses comparing regimens of terbinafine to regimens of itraconazole.”
In what is believed to be the first study of its kind, Dr. Gupta, professor of dermatology at the University of Toronto, and colleagues used network meta-analysis to compare pulse and continuous systemic therapies for toenail onychomycosis. They used PubMed to search for randomized, controlled trials of oral antifungal treatments for the condition in patients aged 18 years and older that included data on mycologic cure, complete cure, adverse events, and dropout rates. Treatment effects were based on intention-to-treat cure rates, and the researchers excluded studies of ketoconazole and griseofulvin since they are no longer indicated for the condition.
For their network meta-analysis, Dr. Gupta and colleagues evaluated 22 studies from 20 publications that included 4,205 randomized patients. Data on complete cure were excluded because of a lack of studies. When the researchers compared all treatments to placebo, the likelihood of mycologic cure did not differ significantly between continuous and pulse regimens for terbinafine and itraconazole. Compared with placebo, the most successful treatments were continuous terbinafine 250 mg daily for 24 weeks (risk ratio of achieving mycologic cure, 11.0) and continuous terbinafine 250 mg daily for 16 weeks (RR, 8.90). The researchers also observed no significant differences in the likelihood of adverse events between any continuous and pulse regimens of terbinafine, itraconazole, and fluconazole.
“Although continuous terbinafine 250 mg for 24 weeks was significantly more likely to produce mycologic cure than continuous itraconazole 200 mg for 12 weeks and weekly fluconazole (150-450 mg), it is not significantly different from the other included treatments,” Dr. Gupta and colleagues wrote in the abstract. “Considering the fungal life cycle, pulse therapy should theoretically be as effective as, or more effective than, continuous therapies: the sudden high concentration of an antifungal drug eliminates hyphae, sparing already-present spores. During the ‘off’ portion, these spores may germinate and be eliminated during the next pulse. Continuous therapy spares the spores, allowing them to germinate once treatment ends.”
They went on to note that, in clinical practice, “neither continuous nor pulse therapy is necessarily better. It is possible that the drug concentration in the nail is maintained during the ‘off’ period of pulse therapy. In both therapies, it may be that residual spores that have not been eliminated by the end of therapy are left to germinate, possibly contributing to the recalcitrant nature of onychomycosis.”
The study was awarded fourth place in the AAD’s 2020 poster awards. Dr. Gupta disclosed that he is a clinical trials investigator for Moberg Pharma and Bausch Health Canada and a speaker for Bausch Health Canada.
SOURCE: Gupta A et al. AAD 20, Abstract 16014.
There appear to be results from a systematic review and network meta-analysis showed.
“Previous meta-analyses of pulse and continuous therapies have generated ambiguous results,” study authors led by Aditya K. Gupta, MD, PhD, wrote in a poster abstract presented at the virtual annual meeting of the American Academy of Dermatology. “There are few head-to-head clinical studies and no meta-analyses comparing regimens of terbinafine to regimens of itraconazole.”
In what is believed to be the first study of its kind, Dr. Gupta, professor of dermatology at the University of Toronto, and colleagues used network meta-analysis to compare pulse and continuous systemic therapies for toenail onychomycosis. They used PubMed to search for randomized, controlled trials of oral antifungal treatments for the condition in patients aged 18 years and older that included data on mycologic cure, complete cure, adverse events, and dropout rates. Treatment effects were based on intention-to-treat cure rates, and the researchers excluded studies of ketoconazole and griseofulvin since they are no longer indicated for the condition.
For their network meta-analysis, Dr. Gupta and colleagues evaluated 22 studies from 20 publications that included 4,205 randomized patients. Data on complete cure were excluded because of a lack of studies. When the researchers compared all treatments to placebo, the likelihood of mycologic cure did not differ significantly between continuous and pulse regimens for terbinafine and itraconazole. Compared with placebo, the most successful treatments were continuous terbinafine 250 mg daily for 24 weeks (risk ratio of achieving mycologic cure, 11.0) and continuous terbinafine 250 mg daily for 16 weeks (RR, 8.90). The researchers also observed no significant differences in the likelihood of adverse events between any continuous and pulse regimens of terbinafine, itraconazole, and fluconazole.
“Although continuous terbinafine 250 mg for 24 weeks was significantly more likely to produce mycologic cure than continuous itraconazole 200 mg for 12 weeks and weekly fluconazole (150-450 mg), it is not significantly different from the other included treatments,” Dr. Gupta and colleagues wrote in the abstract. “Considering the fungal life cycle, pulse therapy should theoretically be as effective as, or more effective than, continuous therapies: the sudden high concentration of an antifungal drug eliminates hyphae, sparing already-present spores. During the ‘off’ portion, these spores may germinate and be eliminated during the next pulse. Continuous therapy spares the spores, allowing them to germinate once treatment ends.”
They went on to note that, in clinical practice, “neither continuous nor pulse therapy is necessarily better. It is possible that the drug concentration in the nail is maintained during the ‘off’ period of pulse therapy. In both therapies, it may be that residual spores that have not been eliminated by the end of therapy are left to germinate, possibly contributing to the recalcitrant nature of onychomycosis.”
The study was awarded fourth place in the AAD’s 2020 poster awards. Dr. Gupta disclosed that he is a clinical trials investigator for Moberg Pharma and Bausch Health Canada and a speaker for Bausch Health Canada.
SOURCE: Gupta A et al. AAD 20, Abstract 16014.
There appear to be results from a systematic review and network meta-analysis showed.
“Previous meta-analyses of pulse and continuous therapies have generated ambiguous results,” study authors led by Aditya K. Gupta, MD, PhD, wrote in a poster abstract presented at the virtual annual meeting of the American Academy of Dermatology. “There are few head-to-head clinical studies and no meta-analyses comparing regimens of terbinafine to regimens of itraconazole.”
In what is believed to be the first study of its kind, Dr. Gupta, professor of dermatology at the University of Toronto, and colleagues used network meta-analysis to compare pulse and continuous systemic therapies for toenail onychomycosis. They used PubMed to search for randomized, controlled trials of oral antifungal treatments for the condition in patients aged 18 years and older that included data on mycologic cure, complete cure, adverse events, and dropout rates. Treatment effects were based on intention-to-treat cure rates, and the researchers excluded studies of ketoconazole and griseofulvin since they are no longer indicated for the condition.
For their network meta-analysis, Dr. Gupta and colleagues evaluated 22 studies from 20 publications that included 4,205 randomized patients. Data on complete cure were excluded because of a lack of studies. When the researchers compared all treatments to placebo, the likelihood of mycologic cure did not differ significantly between continuous and pulse regimens for terbinafine and itraconazole. Compared with placebo, the most successful treatments were continuous terbinafine 250 mg daily for 24 weeks (risk ratio of achieving mycologic cure, 11.0) and continuous terbinafine 250 mg daily for 16 weeks (RR, 8.90). The researchers also observed no significant differences in the likelihood of adverse events between any continuous and pulse regimens of terbinafine, itraconazole, and fluconazole.
“Although continuous terbinafine 250 mg for 24 weeks was significantly more likely to produce mycologic cure than continuous itraconazole 200 mg for 12 weeks and weekly fluconazole (150-450 mg), it is not significantly different from the other included treatments,” Dr. Gupta and colleagues wrote in the abstract. “Considering the fungal life cycle, pulse therapy should theoretically be as effective as, or more effective than, continuous therapies: the sudden high concentration of an antifungal drug eliminates hyphae, sparing already-present spores. During the ‘off’ portion, these spores may germinate and be eliminated during the next pulse. Continuous therapy spares the spores, allowing them to germinate once treatment ends.”
They went on to note that, in clinical practice, “neither continuous nor pulse therapy is necessarily better. It is possible that the drug concentration in the nail is maintained during the ‘off’ period of pulse therapy. In both therapies, it may be that residual spores that have not been eliminated by the end of therapy are left to germinate, possibly contributing to the recalcitrant nature of onychomycosis.”
The study was awarded fourth place in the AAD’s 2020 poster awards. Dr. Gupta disclosed that he is a clinical trials investigator for Moberg Pharma and Bausch Health Canada and a speaker for Bausch Health Canada.
SOURCE: Gupta A et al. AAD 20, Abstract 16014.
FROM AAD 20
Survey: 26% of parents hesitant about influenza vaccine
according to a nationally representative survey.
Influenza vaccination hesitancy may be driven by concerns about vaccine effectiveness, researchers wrote in Pediatrics. These findings “underscore the importance of better communicating to providers and parents the effectiveness of influenza vaccines in reducing severity and morbidity from influenza, even in years when the vaccine has relatively low effectiveness,” noted Allison Kempe, MD, MPH, professor of pediatrics and director of the Adult and Child Consortium for Health Outcomes Research and Delivery Science at the University of Colorado at Denver, Aurora, and colleagues.
The World Health Organization considers vaccine hesitancy a leading threat to global health, but national data about vaccine hesitancy in the United States are limited. To assess hesitancy about routine childhood and influenza vaccinations and related factors, Dr. Kempe and colleagues surveyed more than 2,000 parents in February 2019.
The investigators used an online panel to survey a nationally representative sample of families with children aged between 6 months and 18 years. Parents completed a modified version of the Vaccine Hesitancy Scale, which measures confidence in and concerns about vaccines. Parents with an average score greater than 3 on the scale were considered hesitant.
Factors associated with vaccine hesitancy
Of 4,445 parents sampled, 2,176 completed the survey and 2,052 were eligible respondents. For routine childhood vaccines, the average score on the modified Vaccine Hesitancy Scale was 2 and the percentage of hesitant parents was 6%. For influenza vaccine, the average score was 2 and the percentage of hesitant parents was 26%.
Among hesitant parents, 68% had deferred or refused routine childhood vaccination, compared with 9% of nonhesitant parents (risk ratio, 8.0). For the influenza vaccine, 70% of hesitant parents had deferred or refused influenza vaccination for their child versus 10% of nonhesitant parents (RR, 7.0). Parents were more likely to strongly agree that routine childhood vaccines are effective, compared with the influenza vaccine (70% vs. 26%). “Hesitancy about influenza vaccination is largely driven by concerns about low vaccine effectiveness,” Dr. Kempe and associates wrote.
Although concern about serious side effects was the factor most associated with hesitancy, the percentage of parents who were strongly (12%) or somewhat (27%) concerned about serious side effects was the same for routine childhood vaccines and influenza vaccines. Other factors associated with hesitancy for both routine childhood vaccines and influenza vaccines included lower educational level and household income less than 400% of the federal poverty level.
The survey data may be subject to reporting bias based on social desirability, the authors noted. In addition, the exclusion of infants younger than 6 months may have resulted in an underestimate of hesitancy.
“Although influenza vaccine could be included as a ‘routine’ vaccine, in that it is recommended yearly, we hypothesized that parents view it differently from other childhood vaccines because each year it needs to be given again, its content and effectiveness vary, and it addresses a disease that is often perceived as minor, compared with other childhood diseases,” Dr. Kempe and colleagues wrote. Interventions to counter hesitancy have “a surprising lack of evidence,” and “more work needs to be done to develop methods that are practical and effective for convincing vaccine-hesitant parents to vaccinate.”
Logical next step
“From the pragmatic standpoint of improving immunization rates and disease control, determining the correct evidence-based messaging to counter these perceptions is the next logical step,” Annabelle de St. Maurice, MD, MPH, an assistant professor of pediatrics in the division of infectious diseases at University of California, Los Angeles, and Kathryn Edwards, MD, a professor of pediatrics and director of the vaccine research program at Vanderbilt University, Nashville, wrote in an accompanying editorial.
“Communications should be focused on the burden of influenza in children, rebranding influenza vaccine as a ‘routine’ childhood immunization, reassurance on influenza vaccine safety, and discussion of the efficacy of influenza vaccine in preventing severe disease,” they wrote. “Even in the years when there is a poor match, the vaccine is impactful.”
The research was supported by the National Institutes of Health. Two study authors disclosed financial ties to Sanofi Pasteur, with one also disclosing financial ties to Merck, for work related to vaccinations. The remaining investigators had no relevant financial disclosures. Dr. de St. Maurice indicated that she had no relevant financial disclosures. Dr. Edwards disclosed grants from the Centers for Disease Control and Prevention and the NIH; consulting for Merck, Bionet, and IBM; and serving on data safety and monitoring boards for Sanofi, X4 Pharmaceuticals, Seqirus, Moderna, and Pfizer.
SOURCE: Kempe A et al. Pediatrics. 2020 Jun 15. doi: 10.1542/peds.2019-3852.
according to a nationally representative survey.
Influenza vaccination hesitancy may be driven by concerns about vaccine effectiveness, researchers wrote in Pediatrics. These findings “underscore the importance of better communicating to providers and parents the effectiveness of influenza vaccines in reducing severity and morbidity from influenza, even in years when the vaccine has relatively low effectiveness,” noted Allison Kempe, MD, MPH, professor of pediatrics and director of the Adult and Child Consortium for Health Outcomes Research and Delivery Science at the University of Colorado at Denver, Aurora, and colleagues.
The World Health Organization considers vaccine hesitancy a leading threat to global health, but national data about vaccine hesitancy in the United States are limited. To assess hesitancy about routine childhood and influenza vaccinations and related factors, Dr. Kempe and colleagues surveyed more than 2,000 parents in February 2019.
The investigators used an online panel to survey a nationally representative sample of families with children aged between 6 months and 18 years. Parents completed a modified version of the Vaccine Hesitancy Scale, which measures confidence in and concerns about vaccines. Parents with an average score greater than 3 on the scale were considered hesitant.
Factors associated with vaccine hesitancy
Of 4,445 parents sampled, 2,176 completed the survey and 2,052 were eligible respondents. For routine childhood vaccines, the average score on the modified Vaccine Hesitancy Scale was 2 and the percentage of hesitant parents was 6%. For influenza vaccine, the average score was 2 and the percentage of hesitant parents was 26%.
Among hesitant parents, 68% had deferred or refused routine childhood vaccination, compared with 9% of nonhesitant parents (risk ratio, 8.0). For the influenza vaccine, 70% of hesitant parents had deferred or refused influenza vaccination for their child versus 10% of nonhesitant parents (RR, 7.0). Parents were more likely to strongly agree that routine childhood vaccines are effective, compared with the influenza vaccine (70% vs. 26%). “Hesitancy about influenza vaccination is largely driven by concerns about low vaccine effectiveness,” Dr. Kempe and associates wrote.
Although concern about serious side effects was the factor most associated with hesitancy, the percentage of parents who were strongly (12%) or somewhat (27%) concerned about serious side effects was the same for routine childhood vaccines and influenza vaccines. Other factors associated with hesitancy for both routine childhood vaccines and influenza vaccines included lower educational level and household income less than 400% of the federal poverty level.
The survey data may be subject to reporting bias based on social desirability, the authors noted. In addition, the exclusion of infants younger than 6 months may have resulted in an underestimate of hesitancy.
“Although influenza vaccine could be included as a ‘routine’ vaccine, in that it is recommended yearly, we hypothesized that parents view it differently from other childhood vaccines because each year it needs to be given again, its content and effectiveness vary, and it addresses a disease that is often perceived as minor, compared with other childhood diseases,” Dr. Kempe and colleagues wrote. Interventions to counter hesitancy have “a surprising lack of evidence,” and “more work needs to be done to develop methods that are practical and effective for convincing vaccine-hesitant parents to vaccinate.”
Logical next step
“From the pragmatic standpoint of improving immunization rates and disease control, determining the correct evidence-based messaging to counter these perceptions is the next logical step,” Annabelle de St. Maurice, MD, MPH, an assistant professor of pediatrics in the division of infectious diseases at University of California, Los Angeles, and Kathryn Edwards, MD, a professor of pediatrics and director of the vaccine research program at Vanderbilt University, Nashville, wrote in an accompanying editorial.
“Communications should be focused on the burden of influenza in children, rebranding influenza vaccine as a ‘routine’ childhood immunization, reassurance on influenza vaccine safety, and discussion of the efficacy of influenza vaccine in preventing severe disease,” they wrote. “Even in the years when there is a poor match, the vaccine is impactful.”
The research was supported by the National Institutes of Health. Two study authors disclosed financial ties to Sanofi Pasteur, with one also disclosing financial ties to Merck, for work related to vaccinations. The remaining investigators had no relevant financial disclosures. Dr. de St. Maurice indicated that she had no relevant financial disclosures. Dr. Edwards disclosed grants from the Centers for Disease Control and Prevention and the NIH; consulting for Merck, Bionet, and IBM; and serving on data safety and monitoring boards for Sanofi, X4 Pharmaceuticals, Seqirus, Moderna, and Pfizer.
SOURCE: Kempe A et al. Pediatrics. 2020 Jun 15. doi: 10.1542/peds.2019-3852.
according to a nationally representative survey.
Influenza vaccination hesitancy may be driven by concerns about vaccine effectiveness, researchers wrote in Pediatrics. These findings “underscore the importance of better communicating to providers and parents the effectiveness of influenza vaccines in reducing severity and morbidity from influenza, even in years when the vaccine has relatively low effectiveness,” noted Allison Kempe, MD, MPH, professor of pediatrics and director of the Adult and Child Consortium for Health Outcomes Research and Delivery Science at the University of Colorado at Denver, Aurora, and colleagues.
The World Health Organization considers vaccine hesitancy a leading threat to global health, but national data about vaccine hesitancy in the United States are limited. To assess hesitancy about routine childhood and influenza vaccinations and related factors, Dr. Kempe and colleagues surveyed more than 2,000 parents in February 2019.
The investigators used an online panel to survey a nationally representative sample of families with children aged between 6 months and 18 years. Parents completed a modified version of the Vaccine Hesitancy Scale, which measures confidence in and concerns about vaccines. Parents with an average score greater than 3 on the scale were considered hesitant.
Factors associated with vaccine hesitancy
Of 4,445 parents sampled, 2,176 completed the survey and 2,052 were eligible respondents. For routine childhood vaccines, the average score on the modified Vaccine Hesitancy Scale was 2 and the percentage of hesitant parents was 6%. For influenza vaccine, the average score was 2 and the percentage of hesitant parents was 26%.
Among hesitant parents, 68% had deferred or refused routine childhood vaccination, compared with 9% of nonhesitant parents (risk ratio, 8.0). For the influenza vaccine, 70% of hesitant parents had deferred or refused influenza vaccination for their child versus 10% of nonhesitant parents (RR, 7.0). Parents were more likely to strongly agree that routine childhood vaccines are effective, compared with the influenza vaccine (70% vs. 26%). “Hesitancy about influenza vaccination is largely driven by concerns about low vaccine effectiveness,” Dr. Kempe and associates wrote.
Although concern about serious side effects was the factor most associated with hesitancy, the percentage of parents who were strongly (12%) or somewhat (27%) concerned about serious side effects was the same for routine childhood vaccines and influenza vaccines. Other factors associated with hesitancy for both routine childhood vaccines and influenza vaccines included lower educational level and household income less than 400% of the federal poverty level.
The survey data may be subject to reporting bias based on social desirability, the authors noted. In addition, the exclusion of infants younger than 6 months may have resulted in an underestimate of hesitancy.
“Although influenza vaccine could be included as a ‘routine’ vaccine, in that it is recommended yearly, we hypothesized that parents view it differently from other childhood vaccines because each year it needs to be given again, its content and effectiveness vary, and it addresses a disease that is often perceived as minor, compared with other childhood diseases,” Dr. Kempe and colleagues wrote. Interventions to counter hesitancy have “a surprising lack of evidence,” and “more work needs to be done to develop methods that are practical and effective for convincing vaccine-hesitant parents to vaccinate.”
Logical next step
“From the pragmatic standpoint of improving immunization rates and disease control, determining the correct evidence-based messaging to counter these perceptions is the next logical step,” Annabelle de St. Maurice, MD, MPH, an assistant professor of pediatrics in the division of infectious diseases at University of California, Los Angeles, and Kathryn Edwards, MD, a professor of pediatrics and director of the vaccine research program at Vanderbilt University, Nashville, wrote in an accompanying editorial.
“Communications should be focused on the burden of influenza in children, rebranding influenza vaccine as a ‘routine’ childhood immunization, reassurance on influenza vaccine safety, and discussion of the efficacy of influenza vaccine in preventing severe disease,” they wrote. “Even in the years when there is a poor match, the vaccine is impactful.”
The research was supported by the National Institutes of Health. Two study authors disclosed financial ties to Sanofi Pasteur, with one also disclosing financial ties to Merck, for work related to vaccinations. The remaining investigators had no relevant financial disclosures. Dr. de St. Maurice indicated that she had no relevant financial disclosures. Dr. Edwards disclosed grants from the Centers for Disease Control and Prevention and the NIH; consulting for Merck, Bionet, and IBM; and serving on data safety and monitoring boards for Sanofi, X4 Pharmaceuticals, Seqirus, Moderna, and Pfizer.
SOURCE: Kempe A et al. Pediatrics. 2020 Jun 15. doi: 10.1542/peds.2019-3852.
FROM PEDIATRICS