User login
Hypothermia Could Prove Harmful in Adults with Severe Meningitis
Inducing hypothermia in patients with severe bacterial meningitis offers no clinical benefit and might, in fact, be harmful, according to a clinical trial conducted in France.
Investigators had planned to enroll up to 318 patients in a randomized trial comparing hypothermia treatment to standard care, conducted at 49 intensive care units in France between February 2009 and November 2011. They halted the trial, however, after enrolling the first 98 patients because of concerns by the data and safety monitoring board about excess mortality among those randomized to receive hypothermia treatment, which consisted of a loading dose of 4°C/39°F cold saline and cooling the patient to 32°C/90°F to 34°C/93°F for 48 hours, then passive warming. The trial was led by Dr. Bruno Mourvillier of the Groupe Hospitalier Bichat-Claude Bernard in Paris.
Twenty-five of 49 patients (51%) in the hypothermia group died, compared with 15 of 49 patients (31%) receiving standard care (relative risk, 1.99). Pneumococcal meningitis was diagnosed in 77% of patients. At 3 months, 42 of 49 patients (86%) in the hypothermia group and 36 of 49 patients (74%) in the control group had an unfavorable outcome (RR, 2.17), as gauged by the Glasgow Outcome Scale.
After adjustment for age, scores on the Glasgow Coma Scale at the point of study inclusion, and the presence of septic shock at study inclusion, mortality remained higher in the hypothermia group, but not significantly (hazard ratio, 1.76). However, a post hoc analysis showed a low probability to reach statistical significance in favor of hypothermia by the end of the three original planned stages of the trial.
The study, published online (JAMA 2013 Oct. 8 [doi:10.1001/jama.2013.280506]), was released at the European Society of Intensive Care Medicine’s annual congress in Paris.
Potential mechanisms behind the mortality difference "remain unclear," the authors wrote, noting that they found no difference in nosocomial infections, hemorrhage, cardiovascular effects, or hyperglycemia between the treatment groups. In addition, no significant differences were found in baseline characteristics. All patients received mechanical ventilation and were severely ill, with an average Glasgow Coma Scale rating of 7.
In animal model studies of meningitis, moderate hypothermia has shown favorable effects, such as lowering intracranial pressure and reducing cerebral injury, Dr. Mourvillier and associates noted. They hypothesized that hypothermia would improve functional outcome at 3 months.
"Our trial does not support the use of hypothermia in adults with severe meningitis," they concluded. "Moderate hypothermia did not improve outcome in patients with severe bacterial meningitis and may even be harmful. Our results may have important implications for future trials on hypothermia in patients presenting with septic shock or stroke."
Careful evaluation of safety issues in ongoing trials is needed, they said.
The study was supported by the French Ministry of Health, IST Cardiology, and Covidien. The authors reported no conflicts of interest.
Inducing hypothermia in patients with severe bacterial meningitis offers no clinical benefit and might, in fact, be harmful, according to a clinical trial conducted in France.
Investigators had planned to enroll up to 318 patients in a randomized trial comparing hypothermia treatment to standard care, conducted at 49 intensive care units in France between February 2009 and November 2011. They halted the trial, however, after enrolling the first 98 patients because of concerns by the data and safety monitoring board about excess mortality among those randomized to receive hypothermia treatment, which consisted of a loading dose of 4°C/39°F cold saline and cooling the patient to 32°C/90°F to 34°C/93°F for 48 hours, then passive warming. The trial was led by Dr. Bruno Mourvillier of the Groupe Hospitalier Bichat-Claude Bernard in Paris.
Twenty-five of 49 patients (51%) in the hypothermia group died, compared with 15 of 49 patients (31%) receiving standard care (relative risk, 1.99). Pneumococcal meningitis was diagnosed in 77% of patients. At 3 months, 42 of 49 patients (86%) in the hypothermia group and 36 of 49 patients (74%) in the control group had an unfavorable outcome (RR, 2.17), as gauged by the Glasgow Outcome Scale.
After adjustment for age, scores on the Glasgow Coma Scale at the point of study inclusion, and the presence of septic shock at study inclusion, mortality remained higher in the hypothermia group, but not significantly (hazard ratio, 1.76). However, a post hoc analysis showed a low probability to reach statistical significance in favor of hypothermia by the end of the three original planned stages of the trial.
The study, published online (JAMA 2013 Oct. 8 [doi:10.1001/jama.2013.280506]), was released at the European Society of Intensive Care Medicine’s annual congress in Paris.
Potential mechanisms behind the mortality difference "remain unclear," the authors wrote, noting that they found no difference in nosocomial infections, hemorrhage, cardiovascular effects, or hyperglycemia between the treatment groups. In addition, no significant differences were found in baseline characteristics. All patients received mechanical ventilation and were severely ill, with an average Glasgow Coma Scale rating of 7.
In animal model studies of meningitis, moderate hypothermia has shown favorable effects, such as lowering intracranial pressure and reducing cerebral injury, Dr. Mourvillier and associates noted. They hypothesized that hypothermia would improve functional outcome at 3 months.
"Our trial does not support the use of hypothermia in adults with severe meningitis," they concluded. "Moderate hypothermia did not improve outcome in patients with severe bacterial meningitis and may even be harmful. Our results may have important implications for future trials on hypothermia in patients presenting with septic shock or stroke."
Careful evaluation of safety issues in ongoing trials is needed, they said.
The study was supported by the French Ministry of Health, IST Cardiology, and Covidien. The authors reported no conflicts of interest.
Inducing hypothermia in patients with severe bacterial meningitis offers no clinical benefit and might, in fact, be harmful, according to a clinical trial conducted in France.
Investigators had planned to enroll up to 318 patients in a randomized trial comparing hypothermia treatment to standard care, conducted at 49 intensive care units in France between February 2009 and November 2011. They halted the trial, however, after enrolling the first 98 patients because of concerns by the data and safety monitoring board about excess mortality among those randomized to receive hypothermia treatment, which consisted of a loading dose of 4°C/39°F cold saline and cooling the patient to 32°C/90°F to 34°C/93°F for 48 hours, then passive warming. The trial was led by Dr. Bruno Mourvillier of the Groupe Hospitalier Bichat-Claude Bernard in Paris.
Twenty-five of 49 patients (51%) in the hypothermia group died, compared with 15 of 49 patients (31%) receiving standard care (relative risk, 1.99). Pneumococcal meningitis was diagnosed in 77% of patients. At 3 months, 42 of 49 patients (86%) in the hypothermia group and 36 of 49 patients (74%) in the control group had an unfavorable outcome (RR, 2.17), as gauged by the Glasgow Outcome Scale.
After adjustment for age, scores on the Glasgow Coma Scale at the point of study inclusion, and the presence of septic shock at study inclusion, mortality remained higher in the hypothermia group, but not significantly (hazard ratio, 1.76). However, a post hoc analysis showed a low probability to reach statistical significance in favor of hypothermia by the end of the three original planned stages of the trial.
The study, published online (JAMA 2013 Oct. 8 [doi:10.1001/jama.2013.280506]), was released at the European Society of Intensive Care Medicine’s annual congress in Paris.
Potential mechanisms behind the mortality difference "remain unclear," the authors wrote, noting that they found no difference in nosocomial infections, hemorrhage, cardiovascular effects, or hyperglycemia between the treatment groups. In addition, no significant differences were found in baseline characteristics. All patients received mechanical ventilation and were severely ill, with an average Glasgow Coma Scale rating of 7.
In animal model studies of meningitis, moderate hypothermia has shown favorable effects, such as lowering intracranial pressure and reducing cerebral injury, Dr. Mourvillier and associates noted. They hypothesized that hypothermia would improve functional outcome at 3 months.
"Our trial does not support the use of hypothermia in adults with severe meningitis," they concluded. "Moderate hypothermia did not improve outcome in patients with severe bacterial meningitis and may even be harmful. Our results may have important implications for future trials on hypothermia in patients presenting with septic shock or stroke."
Careful evaluation of safety issues in ongoing trials is needed, they said.
The study was supported by the French Ministry of Health, IST Cardiology, and Covidien. The authors reported no conflicts of interest.
FROM JAMA
Varicella Vaccine: Two Doses are Better Than One
A two-dose varicella vaccination program, begun in 2006, has been shown to be more effective in promoting population immunity than the single-dose regimen introduced more than a decade earlier, in addition to further reducing disease severity and incidence.
Although the single-dose regimen, implemented in 1995, was associated with dramatic declines in varicella-related illness and deaths of 90% and 88%, respectively (Pediatrics 2011;128:214-20), a new study, published online Oct. 7 in Pediatrics showed that implementation of the two-dose scheme not only slashed cases further, but conferred protection even among unvaccinated infants and adults (Pediatrics 2013;132:1-7 [doi: 10.1542/peds.2013-0863]).
For their research, Dr. Stephanie R. Bialek of the National Center for Immunization and Respiratory Diseases, Atlanta, and her colleagues analyzed incidence rates and disease characteristics in two metropolitan centers totaling 650,000 in population between 1995 and 2010; one was a suburb of Los Angeles and the other was an inner-city area of Philadelphia. The study period covered the rollout of the single-dose vaccine and the two-dose scheme.
In 2010, the California surveillance area showed an incidence of 0.3 cases per 1,000 population, a decline of 76% since 2006 and a 97% decline from 1995. The Pennsylvania site, with 0.1 cases per 1,000, saw a 67% decline since 2006 and a 97% decline since 1995. From 2006 to 2010, 61.7% of case patients in both surveillance areas had been vaccinated with a single dose and 7.5% with two doses. Hospitalizations declined by half in both areas between 2006 and 2010.
Approximately 15%-20% of children do not adequately respond to a single dose of vaccine, Dr. Bialek and her colleagues noted, and the two surveillance areas continued to see outbreaks even after the single-dose scheme was in effect. About 65% of outbreak cases after 2007 in California had received one dose, and a larger proportion had milder disease (50 lesions or less) than in outbreaks earlier in the study period. During the two-dose period, the California surveillance area saw a fourfold decrease in outbreaks while the Pennsylvania area reported no outbreaks at all.
"The substantial declines in varicella incidence and outbreaks we report on from these two active surveillance areas during the first 5 years of the two-dose varicella vaccination program provide additional evidence of the program’s sustained impact," Dr. Bialek and her colleagues wrote in their analysis. "With full implementation of the two-dose varicella vaccination program, it may be possible to eliminate the most severe outcomes of varicella."
Dr. Bialek and her colleagues noted as limitations to their study the fact that not all cases reported in the study were laboratory confirmed, allowing for potential overreporting of cases and an underestimation of declines. They also acknowledged that some varicella cases may not have been reported, leading to overestimation of declines, and that their data sources for estimating two-dose coverage levels were limited.
The research was publicly funded, and none of the investigators reported conflicts of interest.
Dr. Bialek and her colleagues’ results definitively show that the current two-dose schedule is superior to the one-dose schedule, with decreased varicella disease among two-dose vaccine recipients, but perhaps even more encouraging, with decreased rates of varicella disease among adults and among groups who cannot receive the varicella vaccine (e.g., infants). This evidence of herd immunity is very promising. Prior to the varicella immunization program in the United States, 125 children each year died of chickenpox, and thousands of survivors were left with serious sequelae. We now can prevent that, and the work of Dr. Bialek and her colleagues clearly demonstrates this benefit.
Dr. David W. Kimberlin is codirector of the division of pediatric infectious diseases at the University of Alabama at Birmingham. He responded to a request to comment on Dr. Bialek and her colleagues’ article.
Dr. Bialek and her colleagues’ results definitively show that the current two-dose schedule is superior to the one-dose schedule, with decreased varicella disease among two-dose vaccine recipients, but perhaps even more encouraging, with decreased rates of varicella disease among adults and among groups who cannot receive the varicella vaccine (e.g., infants). This evidence of herd immunity is very promising. Prior to the varicella immunization program in the United States, 125 children each year died of chickenpox, and thousands of survivors were left with serious sequelae. We now can prevent that, and the work of Dr. Bialek and her colleagues clearly demonstrates this benefit.
Dr. David W. Kimberlin is codirector of the division of pediatric infectious diseases at the University of Alabama at Birmingham. He responded to a request to comment on Dr. Bialek and her colleagues’ article.
Dr. Bialek and her colleagues’ results definitively show that the current two-dose schedule is superior to the one-dose schedule, with decreased varicella disease among two-dose vaccine recipients, but perhaps even more encouraging, with decreased rates of varicella disease among adults and among groups who cannot receive the varicella vaccine (e.g., infants). This evidence of herd immunity is very promising. Prior to the varicella immunization program in the United States, 125 children each year died of chickenpox, and thousands of survivors were left with serious sequelae. We now can prevent that, and the work of Dr. Bialek and her colleagues clearly demonstrates this benefit.
Dr. David W. Kimberlin is codirector of the division of pediatric infectious diseases at the University of Alabama at Birmingham. He responded to a request to comment on Dr. Bialek and her colleagues’ article.
A two-dose varicella vaccination program, begun in 2006, has been shown to be more effective in promoting population immunity than the single-dose regimen introduced more than a decade earlier, in addition to further reducing disease severity and incidence.
Although the single-dose regimen, implemented in 1995, was associated with dramatic declines in varicella-related illness and deaths of 90% and 88%, respectively (Pediatrics 2011;128:214-20), a new study, published online Oct. 7 in Pediatrics showed that implementation of the two-dose scheme not only slashed cases further, but conferred protection even among unvaccinated infants and adults (Pediatrics 2013;132:1-7 [doi: 10.1542/peds.2013-0863]).
For their research, Dr. Stephanie R. Bialek of the National Center for Immunization and Respiratory Diseases, Atlanta, and her colleagues analyzed incidence rates and disease characteristics in two metropolitan centers totaling 650,000 in population between 1995 and 2010; one was a suburb of Los Angeles and the other was an inner-city area of Philadelphia. The study period covered the rollout of the single-dose vaccine and the two-dose scheme.
In 2010, the California surveillance area showed an incidence of 0.3 cases per 1,000 population, a decline of 76% since 2006 and a 97% decline from 1995. The Pennsylvania site, with 0.1 cases per 1,000, saw a 67% decline since 2006 and a 97% decline since 1995. From 2006 to 2010, 61.7% of case patients in both surveillance areas had been vaccinated with a single dose and 7.5% with two doses. Hospitalizations declined by half in both areas between 2006 and 2010.
Approximately 15%-20% of children do not adequately respond to a single dose of vaccine, Dr. Bialek and her colleagues noted, and the two surveillance areas continued to see outbreaks even after the single-dose scheme was in effect. About 65% of outbreak cases after 2007 in California had received one dose, and a larger proportion had milder disease (50 lesions or less) than in outbreaks earlier in the study period. During the two-dose period, the California surveillance area saw a fourfold decrease in outbreaks while the Pennsylvania area reported no outbreaks at all.
"The substantial declines in varicella incidence and outbreaks we report on from these two active surveillance areas during the first 5 years of the two-dose varicella vaccination program provide additional evidence of the program’s sustained impact," Dr. Bialek and her colleagues wrote in their analysis. "With full implementation of the two-dose varicella vaccination program, it may be possible to eliminate the most severe outcomes of varicella."
Dr. Bialek and her colleagues noted as limitations to their study the fact that not all cases reported in the study were laboratory confirmed, allowing for potential overreporting of cases and an underestimation of declines. They also acknowledged that some varicella cases may not have been reported, leading to overestimation of declines, and that their data sources for estimating two-dose coverage levels were limited.
The research was publicly funded, and none of the investigators reported conflicts of interest.
A two-dose varicella vaccination program, begun in 2006, has been shown to be more effective in promoting population immunity than the single-dose regimen introduced more than a decade earlier, in addition to further reducing disease severity and incidence.
Although the single-dose regimen, implemented in 1995, was associated with dramatic declines in varicella-related illness and deaths of 90% and 88%, respectively (Pediatrics 2011;128:214-20), a new study, published online Oct. 7 in Pediatrics showed that implementation of the two-dose scheme not only slashed cases further, but conferred protection even among unvaccinated infants and adults (Pediatrics 2013;132:1-7 [doi: 10.1542/peds.2013-0863]).
For their research, Dr. Stephanie R. Bialek of the National Center for Immunization and Respiratory Diseases, Atlanta, and her colleagues analyzed incidence rates and disease characteristics in two metropolitan centers totaling 650,000 in population between 1995 and 2010; one was a suburb of Los Angeles and the other was an inner-city area of Philadelphia. The study period covered the rollout of the single-dose vaccine and the two-dose scheme.
In 2010, the California surveillance area showed an incidence of 0.3 cases per 1,000 population, a decline of 76% since 2006 and a 97% decline from 1995. The Pennsylvania site, with 0.1 cases per 1,000, saw a 67% decline since 2006 and a 97% decline since 1995. From 2006 to 2010, 61.7% of case patients in both surveillance areas had been vaccinated with a single dose and 7.5% with two doses. Hospitalizations declined by half in both areas between 2006 and 2010.
Approximately 15%-20% of children do not adequately respond to a single dose of vaccine, Dr. Bialek and her colleagues noted, and the two surveillance areas continued to see outbreaks even after the single-dose scheme was in effect. About 65% of outbreak cases after 2007 in California had received one dose, and a larger proportion had milder disease (50 lesions or less) than in outbreaks earlier in the study period. During the two-dose period, the California surveillance area saw a fourfold decrease in outbreaks while the Pennsylvania area reported no outbreaks at all.
"The substantial declines in varicella incidence and outbreaks we report on from these two active surveillance areas during the first 5 years of the two-dose varicella vaccination program provide additional evidence of the program’s sustained impact," Dr. Bialek and her colleagues wrote in their analysis. "With full implementation of the two-dose varicella vaccination program, it may be possible to eliminate the most severe outcomes of varicella."
Dr. Bialek and her colleagues noted as limitations to their study the fact that not all cases reported in the study were laboratory confirmed, allowing for potential overreporting of cases and an underestimation of declines. They also acknowledged that some varicella cases may not have been reported, leading to overestimation of declines, and that their data sources for estimating two-dose coverage levels were limited.
The research was publicly funded, and none of the investigators reported conflicts of interest.
FROM PEDIATRICS
Investigational Norovirus Vaccine Reduces GI Symptoms
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
SAN FRANCISCO – An investigational norovirus vaccine safely reduced the vomiting and diarrhea associated with norovirus genotype GII.4, the most common strain of the disease, in a randomized, double-blind, placebo-controlled trial.
Study subjects were randomized to receive either placebo or the bivalent vaccine, which also targets norovirus genotype GI.1 (the Norwalk strain), and they subsequently drank water containing a significant amount of the GII.4 strain of the virus. Infection with the challenge virus occurred in 52% of 56 subjects in the vaccine group and 60.4% of 53 subjects in the placebo group. Significantly fewer patients with infection in the vaccine group, compared with the placebo group, reported severe vomiting and/or diarrhea (0% vs. 8.3%), moderate or severe diarrhea or vomiting (6.0% vs. 18.8%), and vomiting and/or diarrhea of any severity (20.0% vs. 41.7%), Dr. David I. Bernstein reported during a press conference at an annual scientific meeting on infectious diseases.
Also, fewer subjects in the vaccine group shed norovirus at day 10 after the challenge (22.4% vs. 36.2%), according to Dr. Bernstein of Cincinnati Children’s Hospital Medical Center and the University of Cincinnati.
Participants in this multicenter trial were adults aged 18-50 years who were injected twice, 28 days apart, with either placebo or the vaccine – a virus-like particle vaccine adjuvanted with monophosphoryl lipid A (MPL) and alum. The virus challenge included 4,000 real-time polymerase chain reaction genome equivalents of a heterologous GII.4 norovirus. Subjects were isolated for 4 days as inpatients following the challenge, during which time they were monitored for infection.
"We are excited about the results," Dr. Bernstein said, noting that the findings with respect to the effect on severe symptoms are particularly encouraging because it is severe disease that is of the most concern.
Larger trials in a real-world setting are planned, he said.
Norovirus is the leading cause of acute gastroenteritis among both adults and children, and it is highly contagious. Significant outbreaks occur in many settings where people are in close quarters, including health care facilities, child care centers, cruise ships, and in the military, he said.
In fact, 19-21 million Americans are infected each year, and as many as 800 die from the infection. Children and older adults are particularly vulnerable to developing more serious illness.
"Until recently we accepted [norovirus] as a part of life, but this research gives us a glimmer as to a very different future," said Dr. Andrew T. Pavia of the University of Utah, Salt Lake City, the press conference moderator.
Indeed, one could envision a scenario in which this vaccine, if ultimately approved, could be used to help prevent norovirus among nursing home residents, members of the military, cruise ship passengers, and children in school settings, Dr. Bernstein said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
"This [research] is a good start," he said, adding that there is still a long way to go.
If this vaccine proves as safe and effective in the real world as in the challenge setting used in this trial, it would be a minimum of 5 years before a commercial vaccine was available, he estimated.
Dr. Bernstein reporting serving as an investigator for and receiving research support from LigoCyte Inc., the maker of the investigational vaccine. He also receives royalties for a different norovirus vaccine.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
SAN FRANCISCO – An investigational norovirus vaccine safely reduced the vomiting and diarrhea associated with norovirus genotype GII.4, the most common strain of the disease, in a randomized, double-blind, placebo-controlled trial.
Study subjects were randomized to receive either placebo or the bivalent vaccine, which also targets norovirus genotype GI.1 (the Norwalk strain), and they subsequently drank water containing a significant amount of the GII.4 strain of the virus. Infection with the challenge virus occurred in 52% of 56 subjects in the vaccine group and 60.4% of 53 subjects in the placebo group. Significantly fewer patients with infection in the vaccine group, compared with the placebo group, reported severe vomiting and/or diarrhea (0% vs. 8.3%), moderate or severe diarrhea or vomiting (6.0% vs. 18.8%), and vomiting and/or diarrhea of any severity (20.0% vs. 41.7%), Dr. David I. Bernstein reported during a press conference at an annual scientific meeting on infectious diseases.
Also, fewer subjects in the vaccine group shed norovirus at day 10 after the challenge (22.4% vs. 36.2%), according to Dr. Bernstein of Cincinnati Children’s Hospital Medical Center and the University of Cincinnati.
Participants in this multicenter trial were adults aged 18-50 years who were injected twice, 28 days apart, with either placebo or the vaccine – a virus-like particle vaccine adjuvanted with monophosphoryl lipid A (MPL) and alum. The virus challenge included 4,000 real-time polymerase chain reaction genome equivalents of a heterologous GII.4 norovirus. Subjects were isolated for 4 days as inpatients following the challenge, during which time they were monitored for infection.
"We are excited about the results," Dr. Bernstein said, noting that the findings with respect to the effect on severe symptoms are particularly encouraging because it is severe disease that is of the most concern.
Larger trials in a real-world setting are planned, he said.
Norovirus is the leading cause of acute gastroenteritis among both adults and children, and it is highly contagious. Significant outbreaks occur in many settings where people are in close quarters, including health care facilities, child care centers, cruise ships, and in the military, he said.
In fact, 19-21 million Americans are infected each year, and as many as 800 die from the infection. Children and older adults are particularly vulnerable to developing more serious illness.
"Until recently we accepted [norovirus] as a part of life, but this research gives us a glimmer as to a very different future," said Dr. Andrew T. Pavia of the University of Utah, Salt Lake City, the press conference moderator.
Indeed, one could envision a scenario in which this vaccine, if ultimately approved, could be used to help prevent norovirus among nursing home residents, members of the military, cruise ship passengers, and children in school settings, Dr. Bernstein said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
"This [research] is a good start," he said, adding that there is still a long way to go.
If this vaccine proves as safe and effective in the real world as in the challenge setting used in this trial, it would be a minimum of 5 years before a commercial vaccine was available, he estimated.
Dr. Bernstein reporting serving as an investigator for and receiving research support from LigoCyte Inc., the maker of the investigational vaccine. He also receives royalties for a different norovirus vaccine.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
SAN FRANCISCO – An investigational norovirus vaccine safely reduced the vomiting and diarrhea associated with norovirus genotype GII.4, the most common strain of the disease, in a randomized, double-blind, placebo-controlled trial.
Study subjects were randomized to receive either placebo or the bivalent vaccine, which also targets norovirus genotype GI.1 (the Norwalk strain), and they subsequently drank water containing a significant amount of the GII.4 strain of the virus. Infection with the challenge virus occurred in 52% of 56 subjects in the vaccine group and 60.4% of 53 subjects in the placebo group. Significantly fewer patients with infection in the vaccine group, compared with the placebo group, reported severe vomiting and/or diarrhea (0% vs. 8.3%), moderate or severe diarrhea or vomiting (6.0% vs. 18.8%), and vomiting and/or diarrhea of any severity (20.0% vs. 41.7%), Dr. David I. Bernstein reported during a press conference at an annual scientific meeting on infectious diseases.
Also, fewer subjects in the vaccine group shed norovirus at day 10 after the challenge (22.4% vs. 36.2%), according to Dr. Bernstein of Cincinnati Children’s Hospital Medical Center and the University of Cincinnati.
Participants in this multicenter trial were adults aged 18-50 years who were injected twice, 28 days apart, with either placebo or the vaccine – a virus-like particle vaccine adjuvanted with monophosphoryl lipid A (MPL) and alum. The virus challenge included 4,000 real-time polymerase chain reaction genome equivalents of a heterologous GII.4 norovirus. Subjects were isolated for 4 days as inpatients following the challenge, during which time they were monitored for infection.
"We are excited about the results," Dr. Bernstein said, noting that the findings with respect to the effect on severe symptoms are particularly encouraging because it is severe disease that is of the most concern.
Larger trials in a real-world setting are planned, he said.
Norovirus is the leading cause of acute gastroenteritis among both adults and children, and it is highly contagious. Significant outbreaks occur in many settings where people are in close quarters, including health care facilities, child care centers, cruise ships, and in the military, he said.
In fact, 19-21 million Americans are infected each year, and as many as 800 die from the infection. Children and older adults are particularly vulnerable to developing more serious illness.
"Until recently we accepted [norovirus] as a part of life, but this research gives us a glimmer as to a very different future," said Dr. Andrew T. Pavia of the University of Utah, Salt Lake City, the press conference moderator.
Indeed, one could envision a scenario in which this vaccine, if ultimately approved, could be used to help prevent norovirus among nursing home residents, members of the military, cruise ship passengers, and children in school settings, Dr. Bernstein said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
"This [research] is a good start," he said, adding that there is still a long way to go.
If this vaccine proves as safe and effective in the real world as in the challenge setting used in this trial, it would be a minimum of 5 years before a commercial vaccine was available, he estimated.
Dr. Bernstein reporting serving as an investigator for and receiving research support from LigoCyte Inc., the maker of the investigational vaccine. He also receives royalties for a different norovirus vaccine.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
AT IDWEEK 2013
Be Assertive When Tackling Smoking, Obesity, Etc
Primary care providers must intervene more assertively to get patients to adopt healthier lifestyles, directly targeting smoking, obesity, poor diet, and physical inactivity, according to an American Heart Association science advisory published online Oct. 7 in Circulation.
The investigators termed this science advisory "a call to action" for clinicians, citing their vital role in fostering healthier behaviors. They added that system-wide changes also are necessary "to shift the majority of the public toward the next level of improved cardiovascular health," and so also called on "the health care system, insurance companies, employers, and educational institutions" to do so.
The science advisory urged physicians to follow "the 5 As" – a comprehensive, validated treatment algorithm of counseling steps to facilitate patient behavior change that can be completed within the constraints of the typical medical visit. These include Assessing the risk behavior; Advising change, Agreeing on goals and an action plan; Assisting with treatment; and Arranging follow-up.
Most clinicians easily follow the first A, assessing and tracking health behaviors such as smoking habits, weight gain, diet, and exercise over time.
However, "many providers say they omit the last three As because they perceive them as time consuming," and they also feel they lack the necessary counseling skills.
But even taking a single step toward that goal can be extremely helpful to patients. Simple use of patient-centered communication is key: that is, avoiding the use of "commanding language" and instead asking open-ended questions and expressing empathy signals that the physician takes an active interest in the patient’s perspective. It also reveals what actions a patient is willing to take, helping him or her to develop a behavior change plan.
Physicians also can enlist the help of many allied health professionals to take this step, including clinical psychologists, dieticians, health educators, and kinesiologists. They also should be prepared to connect patients to community resources such as park or community-center programs, biking trials, and farmers’ markets.
Direct physician intervention "will undoubtedly also take the form of answering patients’ questions about which of an armory of computer programs, applications, sensors, and online communities they should use to support healthy lifestyle changes," said Bonnie Spring, Ph.D., and Judith K. Ockene, Ph.D., cochairs of the AHA committee that issued the report. (Circulation 2013 Oct. 7 [doi:10.1161/01.cir.0000435173.25936.e1]).
On the population level, physicians should advocate for policies and strategies that support a large-scale shift toward healthier behaviors. Chief among these is the reimbursement for the intensive behavioral counseling and the multidisciplinary provider teams that are required for patients whose poor health habits put them at cardiovascular risk, Dr. Spring and Dr. Ockene said.
Copies of "Better Population Health Through Behavior Change in Adults: A Call to Action" are available at my.americanheart.org/statements.
This science advisory was issued on behalf of the AHA’s behavior change committee of the Council on Epidemiology and Prevention, the Council on Lifestyle and Cardiometabolic Health, the Council for High Blood Pressure Research, and the Council on Cardiovascular and Stroke Nursing. The writing panel’s disclosure questionnaires are available from the AHA.
Primary care providers must intervene more assertively to get patients to adopt healthier lifestyles, directly targeting smoking, obesity, poor diet, and physical inactivity, according to an American Heart Association science advisory published online Oct. 7 in Circulation.
The investigators termed this science advisory "a call to action" for clinicians, citing their vital role in fostering healthier behaviors. They added that system-wide changes also are necessary "to shift the majority of the public toward the next level of improved cardiovascular health," and so also called on "the health care system, insurance companies, employers, and educational institutions" to do so.
The science advisory urged physicians to follow "the 5 As" – a comprehensive, validated treatment algorithm of counseling steps to facilitate patient behavior change that can be completed within the constraints of the typical medical visit. These include Assessing the risk behavior; Advising change, Agreeing on goals and an action plan; Assisting with treatment; and Arranging follow-up.
Most clinicians easily follow the first A, assessing and tracking health behaviors such as smoking habits, weight gain, diet, and exercise over time.
However, "many providers say they omit the last three As because they perceive them as time consuming," and they also feel they lack the necessary counseling skills.
But even taking a single step toward that goal can be extremely helpful to patients. Simple use of patient-centered communication is key: that is, avoiding the use of "commanding language" and instead asking open-ended questions and expressing empathy signals that the physician takes an active interest in the patient’s perspective. It also reveals what actions a patient is willing to take, helping him or her to develop a behavior change plan.
Physicians also can enlist the help of many allied health professionals to take this step, including clinical psychologists, dieticians, health educators, and kinesiologists. They also should be prepared to connect patients to community resources such as park or community-center programs, biking trials, and farmers’ markets.
Direct physician intervention "will undoubtedly also take the form of answering patients’ questions about which of an armory of computer programs, applications, sensors, and online communities they should use to support healthy lifestyle changes," said Bonnie Spring, Ph.D., and Judith K. Ockene, Ph.D., cochairs of the AHA committee that issued the report. (Circulation 2013 Oct. 7 [doi:10.1161/01.cir.0000435173.25936.e1]).
On the population level, physicians should advocate for policies and strategies that support a large-scale shift toward healthier behaviors. Chief among these is the reimbursement for the intensive behavioral counseling and the multidisciplinary provider teams that are required for patients whose poor health habits put them at cardiovascular risk, Dr. Spring and Dr. Ockene said.
Copies of "Better Population Health Through Behavior Change in Adults: A Call to Action" are available at my.americanheart.org/statements.
This science advisory was issued on behalf of the AHA’s behavior change committee of the Council on Epidemiology and Prevention, the Council on Lifestyle and Cardiometabolic Health, the Council for High Blood Pressure Research, and the Council on Cardiovascular and Stroke Nursing. The writing panel’s disclosure questionnaires are available from the AHA.
Primary care providers must intervene more assertively to get patients to adopt healthier lifestyles, directly targeting smoking, obesity, poor diet, and physical inactivity, according to an American Heart Association science advisory published online Oct. 7 in Circulation.
The investigators termed this science advisory "a call to action" for clinicians, citing their vital role in fostering healthier behaviors. They added that system-wide changes also are necessary "to shift the majority of the public toward the next level of improved cardiovascular health," and so also called on "the health care system, insurance companies, employers, and educational institutions" to do so.
The science advisory urged physicians to follow "the 5 As" – a comprehensive, validated treatment algorithm of counseling steps to facilitate patient behavior change that can be completed within the constraints of the typical medical visit. These include Assessing the risk behavior; Advising change, Agreeing on goals and an action plan; Assisting with treatment; and Arranging follow-up.
Most clinicians easily follow the first A, assessing and tracking health behaviors such as smoking habits, weight gain, diet, and exercise over time.
However, "many providers say they omit the last three As because they perceive them as time consuming," and they also feel they lack the necessary counseling skills.
But even taking a single step toward that goal can be extremely helpful to patients. Simple use of patient-centered communication is key: that is, avoiding the use of "commanding language" and instead asking open-ended questions and expressing empathy signals that the physician takes an active interest in the patient’s perspective. It also reveals what actions a patient is willing to take, helping him or her to develop a behavior change plan.
Physicians also can enlist the help of many allied health professionals to take this step, including clinical psychologists, dieticians, health educators, and kinesiologists. They also should be prepared to connect patients to community resources such as park or community-center programs, biking trials, and farmers’ markets.
Direct physician intervention "will undoubtedly also take the form of answering patients’ questions about which of an armory of computer programs, applications, sensors, and online communities they should use to support healthy lifestyle changes," said Bonnie Spring, Ph.D., and Judith K. Ockene, Ph.D., cochairs of the AHA committee that issued the report. (Circulation 2013 Oct. 7 [doi:10.1161/01.cir.0000435173.25936.e1]).
On the population level, physicians should advocate for policies and strategies that support a large-scale shift toward healthier behaviors. Chief among these is the reimbursement for the intensive behavioral counseling and the multidisciplinary provider teams that are required for patients whose poor health habits put them at cardiovascular risk, Dr. Spring and Dr. Ockene said.
Copies of "Better Population Health Through Behavior Change in Adults: A Call to Action" are available at my.americanheart.org/statements.
This science advisory was issued on behalf of the AHA’s behavior change committee of the Council on Epidemiology and Prevention, the Council on Lifestyle and Cardiometabolic Health, the Council for High Blood Pressure Research, and the Council on Cardiovascular and Stroke Nursing. The writing panel’s disclosure questionnaires are available from the AHA.
FROM CIRCULATION
Influenza: Update for the 2013-2014 Season
Each year in late summer, the CDC publishes its recommendations for the prevention of influenza for the upcoming season. The severity of each influenza season varies and is difficult to predict—underscoring the need to provide maximal vaccine coverage for at-risk patient populations.
Hoping for the best, planning for the worst.
Over the past several decades, the annual number of influenza-related hospitalizations has varied from approximately 55,000 to 431,000,1 and the number of deaths from influenza has been as low as 3,349 and as high as 48,614.2 Infection rates are usually highest in children.
Complications, hospitalizations, and deaths are highest in adults ≥ 65, children < 2 years, and patients with medical conditions known to increase risk for influenza complications. Those at high risk of complications appear in Table 1.3
The main recommendations for this coming year are the same as those for last year, including vaccinating everyone ≥ 6 months of age without a contraindication, starting vaccinations as soon as vaccine is available, and continuing throughout the influenza season for those who need it.
What’s new this year
An increasing number of influenza vaccine products are available; although to date, their effectiveness (which was determined to be 56% for all vaccines used last influenza season)4 remains below what we would hope for. The CDC’s recommendations address these new types of vaccines, including ones that have four antigens instead of three, and use new terminology to describe the vaccines.3
New terminology reflects changing vaccine formulations.
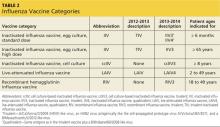
Last influenza season there were two major categories of influenza vaccines: live-attenuated influenza vaccine (LAIV) and trivalent inactivated influenza vaccine (TIV). All products were produced using egg-culture methods and contained two influenza A antigen subtypes and oneB subtype.
Several products this year include four antigens (two A subtypes and two B subtypes), and some are now produced with non–egg-culture methods. This has led to a new system of classification, with the term inactivated influenza vaccine (IIV) replacing TIV. Table 2 lists the influenza vaccine categories and abbreviations. Table 33 lists the contraindications for the different vaccine types.
The new products include Flumist Quadrivalent (MedImmune), a quadrivalent LAIV (LAIV4); Fluarix Quadrivalent (GlaxoSmithKline), a quadrivalent IIV (IIV4); Flucelvax (Novartis Vaccines and Diagnostics), a cell culture-based trivalent IIV (ccIIV3); and FluBlok (Protein Sciences), a trivalent recombinant hemagglutinin influenza vaccine (RIV3). Fluzone (Sanofi Pasteur), introduced last season in a trivalent formulation, is also available this season as a quadrivalent IIV (IIV4).
As a group, influenza vaccine products now offer three routes of administration: intramuscular, subcutaneous, and intranasal. There is currently no evidence that any route offers an advantage over another, and the CDC states no preference for any particular product or route of administration.
Mercury content is not a problem
Even though there is no scientific controversy over the safety of the mercury-containing preservative thimerosal, some patients still have doubts and may ask for a thimerosal-free product. The only influenza products that contain any thimerosal are those that come in multidose vials. A description of each influenza vaccine product, including thimerosal content, indicated ages, and routes of administration, can be found on the CDC’s Web site (www.cdc.gov/flu/professionals/acip/2013-summary-recommendations.htm).3
Options for those with egg allergy
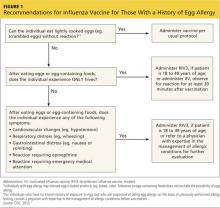
There is now a product, RIV3 (FluBlok), that is manufactured without the use of eggs. It can be used in those 18 to 49 years of age with a history of egg allergy of any severity. Since 2011, the Advisory Committee on Immunization Practices (ACIP) has recommended that individuals with a history of mild egg allergy (those who experience only hives after egg exposure) may receive IIV, with additional safety precautions. Do not delay vaccination for these individuals if RIV is unavailable. Because of a lack of data demonstrating safety of LAIV for individuals with egg allergy, those allergic to eggs should receive RIV or IIV rather than LAIV.
Though the new ccIIV product, Flucelvax, is manufactured without the use of eggs, the seed viruses used to create the vaccine have been processed in eggs. The egg protein content in the vaccine is extremely low (< 50 femtograms [5 × 10-14 g] per 0.5-mL dose), but the CDC does not consider it egg free. Figure 1 (see page 32) depicts the recommendations for those with a history of egg allergy.3
Other interventions for influenza prevention
Vaccination is only one tool available to prevent morbidity and mortality from influenza. Antiviral chemoprevention and treatment and infection control practices can also be effective.
Antiviral chemoprevention is available for both pre- and post-exposure administration. In the past few years, the CDC has de-emphasized such use of antivirals for these indications out of concern for the supply of these agents and for the possibility that their use might lead to increased rates of viral resistance. Consider antiviral chemoprevention for those who have conditions that place them at risk for complications, and for those who are unvaccinated if they are at high risk for exposure to influenza (pre-exposure prophylaxis) or have been exposed (postexposure prophylaxis), if the medication can be started within 48 hours of exposure.
Another option for unvaccinated high-risk patients is vigilant symptom monitoring with early treatment for influenza symptoms. Chemoprophylaxis is recommended in addition to vaccination to control influenza outbreaks at institutions that house patients at high risk for complications of influenza. Details on recommended antivirals including doses and duration of treatment can be found in a 2011 issue of Morbidity and Mortality Weekly Report.5
Antiviral treatment. The CDC recommends antiviral treatment for anyone with suspected or confirmed influenza who has progressive, severe, or complicated illness or is hospitalized for his or her illness.5 Treatment is also recommended for outpatients with suspected or confirmed influenza who are at higher risk for influenza complications. This latter group includes those in Table 1, particularly children 6 to 59 months and adults ≥ 50. Start antiviral treatment within 48 hours of the first symptoms. For hospitalized patients, however, begin treatment at any point, regardless of duration of illness.
Infection control practices can prevent the spread of influenza in the health care setting and in the homes of those with influenza. These practices are also described on the CDC influenza Web site.6
References
1. Thompson WW, Shay DK, Weintraub E. Influenza-associated hospitalizations in the United States. JAMA. 2004;292:1333-1340.
2. CDC. Estimates of deaths associated with seasonal influenza–United States, 1976-2007. MMWR Morb Mortal Wkly Rep. 2010;59:1057-1062.
3. CDC. Summary* recommendations: prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—(ACIP)—United States, 2013-14. www.cdc.gov/flu/professionals/acip/2013-summary-recommendations.htm. Accessed August 9, 2013.
4. CDC. Interim adjusted estimates of seasonal influenza vaccine effectiveness–United States, February 2013. MMWR Morb Mortal Wkly Rep. 2013;62:119-123.
5. CDC. Antiviral agents for the treatment and chemoprophylaxis of influenza. MMWR Recomm Rep. 2011;60(RR01):1-24. www.cdc.gov/mmwr/preview/mmwrhtml/rr6001a1.htm. Accessed July 2, 2013.
6. CDC. Infection control in health care facilities. www.cdc.gov/flu/professionals/infectioncontrol/index.htm. Accessed July 2, 2013.
Each year in late summer, the CDC publishes its recommendations for the prevention of influenza for the upcoming season. The severity of each influenza season varies and is difficult to predict—underscoring the need to provide maximal vaccine coverage for at-risk patient populations.
Hoping for the best, planning for the worst.
Over the past several decades, the annual number of influenza-related hospitalizations has varied from approximately 55,000 to 431,000,1 and the number of deaths from influenza has been as low as 3,349 and as high as 48,614.2 Infection rates are usually highest in children.
Complications, hospitalizations, and deaths are highest in adults ≥ 65, children < 2 years, and patients with medical conditions known to increase risk for influenza complications. Those at high risk of complications appear in Table 1.3
The main recommendations for this coming year are the same as those for last year, including vaccinating everyone ≥ 6 months of age without a contraindication, starting vaccinations as soon as vaccine is available, and continuing throughout the influenza season for those who need it.
What’s new this year
An increasing number of influenza vaccine products are available; although to date, their effectiveness (which was determined to be 56% for all vaccines used last influenza season)4 remains below what we would hope for. The CDC’s recommendations address these new types of vaccines, including ones that have four antigens instead of three, and use new terminology to describe the vaccines.3
New terminology reflects changing vaccine formulations.
Last influenza season there were two major categories of influenza vaccines: live-attenuated influenza vaccine (LAIV) and trivalent inactivated influenza vaccine (TIV). All products were produced using egg-culture methods and contained two influenza A antigen subtypes and oneB subtype.
Several products this year include four antigens (two A subtypes and two B subtypes), and some are now produced with non–egg-culture methods. This has led to a new system of classification, with the term inactivated influenza vaccine (IIV) replacing TIV. Table 2 lists the influenza vaccine categories and abbreviations. Table 33 lists the contraindications for the different vaccine types.
The new products include Flumist Quadrivalent (MedImmune), a quadrivalent LAIV (LAIV4); Fluarix Quadrivalent (GlaxoSmithKline), a quadrivalent IIV (IIV4); Flucelvax (Novartis Vaccines and Diagnostics), a cell culture-based trivalent IIV (ccIIV3); and FluBlok (Protein Sciences), a trivalent recombinant hemagglutinin influenza vaccine (RIV3). Fluzone (Sanofi Pasteur), introduced last season in a trivalent formulation, is also available this season as a quadrivalent IIV (IIV4).
As a group, influenza vaccine products now offer three routes of administration: intramuscular, subcutaneous, and intranasal. There is currently no evidence that any route offers an advantage over another, and the CDC states no preference for any particular product or route of administration.
Mercury content is not a problem
Even though there is no scientific controversy over the safety of the mercury-containing preservative thimerosal, some patients still have doubts and may ask for a thimerosal-free product. The only influenza products that contain any thimerosal are those that come in multidose vials. A description of each influenza vaccine product, including thimerosal content, indicated ages, and routes of administration, can be found on the CDC’s Web site (www.cdc.gov/flu/professionals/acip/2013-summary-recommendations.htm).3
Options for those with egg allergy
There is now a product, RIV3 (FluBlok), that is manufactured without the use of eggs. It can be used in those 18 to 49 years of age with a history of egg allergy of any severity. Since 2011, the Advisory Committee on Immunization Practices (ACIP) has recommended that individuals with a history of mild egg allergy (those who experience only hives after egg exposure) may receive IIV, with additional safety precautions. Do not delay vaccination for these individuals if RIV is unavailable. Because of a lack of data demonstrating safety of LAIV for individuals with egg allergy, those allergic to eggs should receive RIV or IIV rather than LAIV.
Though the new ccIIV product, Flucelvax, is manufactured without the use of eggs, the seed viruses used to create the vaccine have been processed in eggs. The egg protein content in the vaccine is extremely low (< 50 femtograms [5 × 10-14 g] per 0.5-mL dose), but the CDC does not consider it egg free. Figure 1 (see page 32) depicts the recommendations for those with a history of egg allergy.3
Other interventions for influenza prevention
Vaccination is only one tool available to prevent morbidity and mortality from influenza. Antiviral chemoprevention and treatment and infection control practices can also be effective.
Antiviral chemoprevention is available for both pre- and post-exposure administration. In the past few years, the CDC has de-emphasized such use of antivirals for these indications out of concern for the supply of these agents and for the possibility that their use might lead to increased rates of viral resistance. Consider antiviral chemoprevention for those who have conditions that place them at risk for complications, and for those who are unvaccinated if they are at high risk for exposure to influenza (pre-exposure prophylaxis) or have been exposed (postexposure prophylaxis), if the medication can be started within 48 hours of exposure.
Another option for unvaccinated high-risk patients is vigilant symptom monitoring with early treatment for influenza symptoms. Chemoprophylaxis is recommended in addition to vaccination to control influenza outbreaks at institutions that house patients at high risk for complications of influenza. Details on recommended antivirals including doses and duration of treatment can be found in a 2011 issue of Morbidity and Mortality Weekly Report.5
Antiviral treatment. The CDC recommends antiviral treatment for anyone with suspected or confirmed influenza who has progressive, severe, or complicated illness or is hospitalized for his or her illness.5 Treatment is also recommended for outpatients with suspected or confirmed influenza who are at higher risk for influenza complications. This latter group includes those in Table 1, particularly children 6 to 59 months and adults ≥ 50. Start antiviral treatment within 48 hours of the first symptoms. For hospitalized patients, however, begin treatment at any point, regardless of duration of illness.
Infection control practices can prevent the spread of influenza in the health care setting and in the homes of those with influenza. These practices are also described on the CDC influenza Web site.6
References
1. Thompson WW, Shay DK, Weintraub E. Influenza-associated hospitalizations in the United States. JAMA. 2004;292:1333-1340.
2. CDC. Estimates of deaths associated with seasonal influenza–United States, 1976-2007. MMWR Morb Mortal Wkly Rep. 2010;59:1057-1062.
3. CDC. Summary* recommendations: prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—(ACIP)—United States, 2013-14. www.cdc.gov/flu/professionals/acip/2013-summary-recommendations.htm. Accessed August 9, 2013.
4. CDC. Interim adjusted estimates of seasonal influenza vaccine effectiveness–United States, February 2013. MMWR Morb Mortal Wkly Rep. 2013;62:119-123.
5. CDC. Antiviral agents for the treatment and chemoprophylaxis of influenza. MMWR Recomm Rep. 2011;60(RR01):1-24. www.cdc.gov/mmwr/preview/mmwrhtml/rr6001a1.htm. Accessed July 2, 2013.
6. CDC. Infection control in health care facilities. www.cdc.gov/flu/professionals/infectioncontrol/index.htm. Accessed July 2, 2013.
Each year in late summer, the CDC publishes its recommendations for the prevention of influenza for the upcoming season. The severity of each influenza season varies and is difficult to predict—underscoring the need to provide maximal vaccine coverage for at-risk patient populations.
Hoping for the best, planning for the worst.
Over the past several decades, the annual number of influenza-related hospitalizations has varied from approximately 55,000 to 431,000,1 and the number of deaths from influenza has been as low as 3,349 and as high as 48,614.2 Infection rates are usually highest in children.
Complications, hospitalizations, and deaths are highest in adults ≥ 65, children < 2 years, and patients with medical conditions known to increase risk for influenza complications. Those at high risk of complications appear in Table 1.3
The main recommendations for this coming year are the same as those for last year, including vaccinating everyone ≥ 6 months of age without a contraindication, starting vaccinations as soon as vaccine is available, and continuing throughout the influenza season for those who need it.
What’s new this year
An increasing number of influenza vaccine products are available; although to date, their effectiveness (which was determined to be 56% for all vaccines used last influenza season)4 remains below what we would hope for. The CDC’s recommendations address these new types of vaccines, including ones that have four antigens instead of three, and use new terminology to describe the vaccines.3
New terminology reflects changing vaccine formulations.
Last influenza season there were two major categories of influenza vaccines: live-attenuated influenza vaccine (LAIV) and trivalent inactivated influenza vaccine (TIV). All products were produced using egg-culture methods and contained two influenza A antigen subtypes and oneB subtype.
Several products this year include four antigens (two A subtypes and two B subtypes), and some are now produced with non–egg-culture methods. This has led to a new system of classification, with the term inactivated influenza vaccine (IIV) replacing TIV. Table 2 lists the influenza vaccine categories and abbreviations. Table 33 lists the contraindications for the different vaccine types.
The new products include Flumist Quadrivalent (MedImmune), a quadrivalent LAIV (LAIV4); Fluarix Quadrivalent (GlaxoSmithKline), a quadrivalent IIV (IIV4); Flucelvax (Novartis Vaccines and Diagnostics), a cell culture-based trivalent IIV (ccIIV3); and FluBlok (Protein Sciences), a trivalent recombinant hemagglutinin influenza vaccine (RIV3). Fluzone (Sanofi Pasteur), introduced last season in a trivalent formulation, is also available this season as a quadrivalent IIV (IIV4).
As a group, influenza vaccine products now offer three routes of administration: intramuscular, subcutaneous, and intranasal. There is currently no evidence that any route offers an advantage over another, and the CDC states no preference for any particular product or route of administration.
Mercury content is not a problem
Even though there is no scientific controversy over the safety of the mercury-containing preservative thimerosal, some patients still have doubts and may ask for a thimerosal-free product. The only influenza products that contain any thimerosal are those that come in multidose vials. A description of each influenza vaccine product, including thimerosal content, indicated ages, and routes of administration, can be found on the CDC’s Web site (www.cdc.gov/flu/professionals/acip/2013-summary-recommendations.htm).3
Options for those with egg allergy
There is now a product, RIV3 (FluBlok), that is manufactured without the use of eggs. It can be used in those 18 to 49 years of age with a history of egg allergy of any severity. Since 2011, the Advisory Committee on Immunization Practices (ACIP) has recommended that individuals with a history of mild egg allergy (those who experience only hives after egg exposure) may receive IIV, with additional safety precautions. Do not delay vaccination for these individuals if RIV is unavailable. Because of a lack of data demonstrating safety of LAIV for individuals with egg allergy, those allergic to eggs should receive RIV or IIV rather than LAIV.
Though the new ccIIV product, Flucelvax, is manufactured without the use of eggs, the seed viruses used to create the vaccine have been processed in eggs. The egg protein content in the vaccine is extremely low (< 50 femtograms [5 × 10-14 g] per 0.5-mL dose), but the CDC does not consider it egg free. Figure 1 (see page 32) depicts the recommendations for those with a history of egg allergy.3
Other interventions for influenza prevention
Vaccination is only one tool available to prevent morbidity and mortality from influenza. Antiviral chemoprevention and treatment and infection control practices can also be effective.
Antiviral chemoprevention is available for both pre- and post-exposure administration. In the past few years, the CDC has de-emphasized such use of antivirals for these indications out of concern for the supply of these agents and for the possibility that their use might lead to increased rates of viral resistance. Consider antiviral chemoprevention for those who have conditions that place them at risk for complications, and for those who are unvaccinated if they are at high risk for exposure to influenza (pre-exposure prophylaxis) or have been exposed (postexposure prophylaxis), if the medication can be started within 48 hours of exposure.
Another option for unvaccinated high-risk patients is vigilant symptom monitoring with early treatment for influenza symptoms. Chemoprophylaxis is recommended in addition to vaccination to control influenza outbreaks at institutions that house patients at high risk for complications of influenza. Details on recommended antivirals including doses and duration of treatment can be found in a 2011 issue of Morbidity and Mortality Weekly Report.5
Antiviral treatment. The CDC recommends antiviral treatment for anyone with suspected or confirmed influenza who has progressive, severe, or complicated illness or is hospitalized for his or her illness.5 Treatment is also recommended for outpatients with suspected or confirmed influenza who are at higher risk for influenza complications. This latter group includes those in Table 1, particularly children 6 to 59 months and adults ≥ 50. Start antiviral treatment within 48 hours of the first symptoms. For hospitalized patients, however, begin treatment at any point, regardless of duration of illness.
Infection control practices can prevent the spread of influenza in the health care setting and in the homes of those with influenza. These practices are also described on the CDC influenza Web site.6
References
1. Thompson WW, Shay DK, Weintraub E. Influenza-associated hospitalizations in the United States. JAMA. 2004;292:1333-1340.
2. CDC. Estimates of deaths associated with seasonal influenza–United States, 1976-2007. MMWR Morb Mortal Wkly Rep. 2010;59:1057-1062.
3. CDC. Summary* recommendations: prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices—(ACIP)—United States, 2013-14. www.cdc.gov/flu/professionals/acip/2013-summary-recommendations.htm. Accessed August 9, 2013.
4. CDC. Interim adjusted estimates of seasonal influenza vaccine effectiveness–United States, February 2013. MMWR Morb Mortal Wkly Rep. 2013;62:119-123.
5. CDC. Antiviral agents for the treatment and chemoprophylaxis of influenza. MMWR Recomm Rep. 2011;60(RR01):1-24. www.cdc.gov/mmwr/preview/mmwrhtml/rr6001a1.htm. Accessed July 2, 2013.
6. CDC. Infection control in health care facilities. www.cdc.gov/flu/professionals/infectioncontrol/index.htm. Accessed July 2, 2013.
Dexamethasone improves outcomes for infants with bronchiolitis, atopy history
A 5-day course of dexamethasone significantly shortened hospital stays for infants with bronchiolitis who had eczema or close relatives with asthma.
The randomized, placebo-controlled study suggests that a family history of atopy could identify a subset of babies who would benefit from the addition of a corticosteroid to the usual salbutamol therapy for acute bronchiolitis, according to Dr. Khalid Alansari and colleagues. The report was published in the Sept. 16 issue of Pediatrics.
The researchers examined 7-day outcomes in 200 infants with acute bronchiolitis who were at a high risk of asthma, as determined by having at least one first-degree relative with either asthma or eczema. All of the children (mean age 3.5 months) were admitted to a pediatric hospital for treatment, wrote Dr. Alansari of Weill Cornell Medical College, Doha, Qatar, and coauthors. Infants who received dexamethasone were discharged 8 hours earlier than were those receiving standard treatment. The mean duration of symptoms was 4.5 days (Pediatrics 2013 Sept. 13 [doi: 10.1542/peds.2012-3746]).
The study’s primary outcome was time until discharge. Secondary outcomes included the number of patients who needed epinephrine treatment, readmission for a shorter stay in an infirmary site, and revisiting the emergency department or another clinic for the same illness. A study nurse made daily calls to assess the patients after discharge.
Infants in the dexamethasone group were discharged at a mean of 18.6 hours – significantly sooner than those in the control group (27 hours). Epinephrine was necessary for 19 infants in the dexamethasone group and 31 in the placebo group – again a significant difference.
Similar numbers in each group needed readmission and additional outpatient visits in the week after discharge. During the follow-up week, 22% of the dexamethasone group needed infirmary care and the mean stay was 17 hours, compared with 21% of the placebo group with a mean stay of 18 hours.
Nineteen in the dexamethasone group and 11 in the placebo group made a clinic visit (18.6% vs. 11%); this difference was not significant.
The chest radiograph was normal in about 37% of infants studied. About half showed lesser infiltrates; 15% had a lobar collapse or consolidation.
More than 70% had a full sibling with asthma. About 20% had a parent with the disease; in 5%, both parents had it. About 20% of patients had both eczema and first-degree relative with asthma.
All of the infants received 2.5 mg salbutamol nebulization at baseline and at 30, 60, and 120 minutes, and then every 2 hours until discharge. Nebulized epinephrine (0.5 mL/kg with a maximum dose of 5 mL) was available if needed. In addition, they were randomized to either placebo or to a 5-day course of dexamethasone 1 mg/mL, at a rate of 1 mL/kg on day 1, reduced to 0.6 mL/kg for days 2-5.
The study was sponsored by Hamad Medical Corporation. The authors reported no financial conflicts.
A 5-day course of dexamethasone significantly shortened hospital stays for infants with bronchiolitis who had eczema or close relatives with asthma.
The randomized, placebo-controlled study suggests that a family history of atopy could identify a subset of babies who would benefit from the addition of a corticosteroid to the usual salbutamol therapy for acute bronchiolitis, according to Dr. Khalid Alansari and colleagues. The report was published in the Sept. 16 issue of Pediatrics.
The researchers examined 7-day outcomes in 200 infants with acute bronchiolitis who were at a high risk of asthma, as determined by having at least one first-degree relative with either asthma or eczema. All of the children (mean age 3.5 months) were admitted to a pediatric hospital for treatment, wrote Dr. Alansari of Weill Cornell Medical College, Doha, Qatar, and coauthors. Infants who received dexamethasone were discharged 8 hours earlier than were those receiving standard treatment. The mean duration of symptoms was 4.5 days (Pediatrics 2013 Sept. 13 [doi: 10.1542/peds.2012-3746]).
The study’s primary outcome was time until discharge. Secondary outcomes included the number of patients who needed epinephrine treatment, readmission for a shorter stay in an infirmary site, and revisiting the emergency department or another clinic for the same illness. A study nurse made daily calls to assess the patients after discharge.
Infants in the dexamethasone group were discharged at a mean of 18.6 hours – significantly sooner than those in the control group (27 hours). Epinephrine was necessary for 19 infants in the dexamethasone group and 31 in the placebo group – again a significant difference.
Similar numbers in each group needed readmission and additional outpatient visits in the week after discharge. During the follow-up week, 22% of the dexamethasone group needed infirmary care and the mean stay was 17 hours, compared with 21% of the placebo group with a mean stay of 18 hours.
Nineteen in the dexamethasone group and 11 in the placebo group made a clinic visit (18.6% vs. 11%); this difference was not significant.
The chest radiograph was normal in about 37% of infants studied. About half showed lesser infiltrates; 15% had a lobar collapse or consolidation.
More than 70% had a full sibling with asthma. About 20% had a parent with the disease; in 5%, both parents had it. About 20% of patients had both eczema and first-degree relative with asthma.
All of the infants received 2.5 mg salbutamol nebulization at baseline and at 30, 60, and 120 minutes, and then every 2 hours until discharge. Nebulized epinephrine (0.5 mL/kg with a maximum dose of 5 mL) was available if needed. In addition, they were randomized to either placebo or to a 5-day course of dexamethasone 1 mg/mL, at a rate of 1 mL/kg on day 1, reduced to 0.6 mL/kg for days 2-5.
The study was sponsored by Hamad Medical Corporation. The authors reported no financial conflicts.
A 5-day course of dexamethasone significantly shortened hospital stays for infants with bronchiolitis who had eczema or close relatives with asthma.
The randomized, placebo-controlled study suggests that a family history of atopy could identify a subset of babies who would benefit from the addition of a corticosteroid to the usual salbutamol therapy for acute bronchiolitis, according to Dr. Khalid Alansari and colleagues. The report was published in the Sept. 16 issue of Pediatrics.
The researchers examined 7-day outcomes in 200 infants with acute bronchiolitis who were at a high risk of asthma, as determined by having at least one first-degree relative with either asthma or eczema. All of the children (mean age 3.5 months) were admitted to a pediatric hospital for treatment, wrote Dr. Alansari of Weill Cornell Medical College, Doha, Qatar, and coauthors. Infants who received dexamethasone were discharged 8 hours earlier than were those receiving standard treatment. The mean duration of symptoms was 4.5 days (Pediatrics 2013 Sept. 13 [doi: 10.1542/peds.2012-3746]).
The study’s primary outcome was time until discharge. Secondary outcomes included the number of patients who needed epinephrine treatment, readmission for a shorter stay in an infirmary site, and revisiting the emergency department or another clinic for the same illness. A study nurse made daily calls to assess the patients after discharge.
Infants in the dexamethasone group were discharged at a mean of 18.6 hours – significantly sooner than those in the control group (27 hours). Epinephrine was necessary for 19 infants in the dexamethasone group and 31 in the placebo group – again a significant difference.
Similar numbers in each group needed readmission and additional outpatient visits in the week after discharge. During the follow-up week, 22% of the dexamethasone group needed infirmary care and the mean stay was 17 hours, compared with 21% of the placebo group with a mean stay of 18 hours.
Nineteen in the dexamethasone group and 11 in the placebo group made a clinic visit (18.6% vs. 11%); this difference was not significant.
The chest radiograph was normal in about 37% of infants studied. About half showed lesser infiltrates; 15% had a lobar collapse or consolidation.
More than 70% had a full sibling with asthma. About 20% had a parent with the disease; in 5%, both parents had it. About 20% of patients had both eczema and first-degree relative with asthma.
All of the infants received 2.5 mg salbutamol nebulization at baseline and at 30, 60, and 120 minutes, and then every 2 hours until discharge. Nebulized epinephrine (0.5 mL/kg with a maximum dose of 5 mL) was available if needed. In addition, they were randomized to either placebo or to a 5-day course of dexamethasone 1 mg/mL, at a rate of 1 mL/kg on day 1, reduced to 0.6 mL/kg for days 2-5.
The study was sponsored by Hamad Medical Corporation. The authors reported no financial conflicts.
Low, high dose vancomycin equally effective in C. difficile
DENVER – Oral vancomycin at a dose of 125 mg four times daily is just as effective as is a dose of 250 mg or higher given at the same frequency in the treatment of diarrhea associated with Clostridium difficile infection, judging from the results from a retrospective study.
Use of the lower dosing regimen has the potential to decrease treatment costs without worsening outcomes, Philip Chung, Pharm.D., said in an interview before the annual Interscience Conference on Antimicrobial Agents and Chemotherapy, where the study was presented.
According to current recommendations, oral vancomycin 125 mg q.i.d. is the treatment of choice for severe uncomplicated C. difficile infection. To date, no studies have shown the use of oral vancomycin doses higher than 125 mg q.i.d. to be more efficacious than the recommended 125 mg q.i.d. in this setting, said Dr. Chung, clinical pharmacy manager of infectious diseases at Montefiore Medical Center, New York.
"Prior to 2008, prescribers at our institution frequently requested vancomycin doses higher than the recommended 125 mg q.i.d. for treatment of [C. difficile infection] despite the absence of data showing added benefits with the higher dosing regimens," he said. "This practice not only increases medication and/or preparation costs, but it may also increase the potential for untoward effects in patients being treated with the higher doses (e.g., increased risks for vancomycin-resistant enterococci colonization or higher likelihood to further alter the GI flora)."
Since the inception of the Antimicrobial Stewardship Program at Montefiore Medical Center in 2008, Dr. Chung and his associates observed a shift in oral vancomycin prescribing practices from a higher dosing regimen to a lower dosing regimen. "Because of this change in practice, we wished to evaluate the efficacy of the different oral vancomycin dosing regimen in order to ensure treatment outcomes remained unchanged at our institution," he said. To do so, the researchers retrospectively reviewed clinical outcomes of 300 adult patients treated with oral vancomycin at the medical center between 2006 and 2010. They looked at clinical parameters, concomitant antibiotics, in-hospital mortality, and 30-day readmission.
The primary endpoint was clinical improvement at 72 hours of oral vancomycin. Secondary endpoints included clinical improvement at end of therapy or discharge, length of stay, in-hospital mortality, and 30-day readmission rate.
Of the 300 patients, 197 were prescribed oral vancomycin 125 mg q.i.d. (low-dose group) and 103 patients were prescribed 250 mg or higher q.i.d. (high-dose group). Dr. Chung and his associates reported that clinical improvement assessed 72 hours after starting oral vancomycin therapy was observed in 85% and 86% of patients in the low- and high-dose groups, respectively (P less than 0.05). Rates of clinical improvement at end of therapy or time of hospital discharge between the two groups were also found to be similar (93% vs. 96%), as were total length of hospital stay (20 days vs. 19 days), in-hospital mortality (15% vs. 23%), and rates of 30-day readmission (34% vs. 24%).
"The finding that oral vancomycin 125 mg q.i.d. works as well as higher doses for severe uncomplicated C. difficile infection for the most part is not a surprise to us, as some evidence already exists in the literature," said Dr. Chung, also of Albert Einstein College of Medicine in New York. "Our finding only confirmed what is already known."
Limitations include the fact that this is a single-center study, and that it is subject to selection bias because of its retrospective design, Dr. Chung said. "However, we took steps to ensure that all patients treated with oral vancomycin who had laboratory-confirmed [C. difficile infection] and symptoms consistent with [C. difficile infection] were included in the study," he said.
Dr. Chung said that he had no relevant financial conflicts to disclose.
DENVER – Oral vancomycin at a dose of 125 mg four times daily is just as effective as is a dose of 250 mg or higher given at the same frequency in the treatment of diarrhea associated with Clostridium difficile infection, judging from the results from a retrospective study.
Use of the lower dosing regimen has the potential to decrease treatment costs without worsening outcomes, Philip Chung, Pharm.D., said in an interview before the annual Interscience Conference on Antimicrobial Agents and Chemotherapy, where the study was presented.
According to current recommendations, oral vancomycin 125 mg q.i.d. is the treatment of choice for severe uncomplicated C. difficile infection. To date, no studies have shown the use of oral vancomycin doses higher than 125 mg q.i.d. to be more efficacious than the recommended 125 mg q.i.d. in this setting, said Dr. Chung, clinical pharmacy manager of infectious diseases at Montefiore Medical Center, New York.
"Prior to 2008, prescribers at our institution frequently requested vancomycin doses higher than the recommended 125 mg q.i.d. for treatment of [C. difficile infection] despite the absence of data showing added benefits with the higher dosing regimens," he said. "This practice not only increases medication and/or preparation costs, but it may also increase the potential for untoward effects in patients being treated with the higher doses (e.g., increased risks for vancomycin-resistant enterococci colonization or higher likelihood to further alter the GI flora)."
Since the inception of the Antimicrobial Stewardship Program at Montefiore Medical Center in 2008, Dr. Chung and his associates observed a shift in oral vancomycin prescribing practices from a higher dosing regimen to a lower dosing regimen. "Because of this change in practice, we wished to evaluate the efficacy of the different oral vancomycin dosing regimen in order to ensure treatment outcomes remained unchanged at our institution," he said. To do so, the researchers retrospectively reviewed clinical outcomes of 300 adult patients treated with oral vancomycin at the medical center between 2006 and 2010. They looked at clinical parameters, concomitant antibiotics, in-hospital mortality, and 30-day readmission.
The primary endpoint was clinical improvement at 72 hours of oral vancomycin. Secondary endpoints included clinical improvement at end of therapy or discharge, length of stay, in-hospital mortality, and 30-day readmission rate.
Of the 300 patients, 197 were prescribed oral vancomycin 125 mg q.i.d. (low-dose group) and 103 patients were prescribed 250 mg or higher q.i.d. (high-dose group). Dr. Chung and his associates reported that clinical improvement assessed 72 hours after starting oral vancomycin therapy was observed in 85% and 86% of patients in the low- and high-dose groups, respectively (P less than 0.05). Rates of clinical improvement at end of therapy or time of hospital discharge between the two groups were also found to be similar (93% vs. 96%), as were total length of hospital stay (20 days vs. 19 days), in-hospital mortality (15% vs. 23%), and rates of 30-day readmission (34% vs. 24%).
"The finding that oral vancomycin 125 mg q.i.d. works as well as higher doses for severe uncomplicated C. difficile infection for the most part is not a surprise to us, as some evidence already exists in the literature," said Dr. Chung, also of Albert Einstein College of Medicine in New York. "Our finding only confirmed what is already known."
Limitations include the fact that this is a single-center study, and that it is subject to selection bias because of its retrospective design, Dr. Chung said. "However, we took steps to ensure that all patients treated with oral vancomycin who had laboratory-confirmed [C. difficile infection] and symptoms consistent with [C. difficile infection] were included in the study," he said.
Dr. Chung said that he had no relevant financial conflicts to disclose.
DENVER – Oral vancomycin at a dose of 125 mg four times daily is just as effective as is a dose of 250 mg or higher given at the same frequency in the treatment of diarrhea associated with Clostridium difficile infection, judging from the results from a retrospective study.
Use of the lower dosing regimen has the potential to decrease treatment costs without worsening outcomes, Philip Chung, Pharm.D., said in an interview before the annual Interscience Conference on Antimicrobial Agents and Chemotherapy, where the study was presented.
According to current recommendations, oral vancomycin 125 mg q.i.d. is the treatment of choice for severe uncomplicated C. difficile infection. To date, no studies have shown the use of oral vancomycin doses higher than 125 mg q.i.d. to be more efficacious than the recommended 125 mg q.i.d. in this setting, said Dr. Chung, clinical pharmacy manager of infectious diseases at Montefiore Medical Center, New York.
"Prior to 2008, prescribers at our institution frequently requested vancomycin doses higher than the recommended 125 mg q.i.d. for treatment of [C. difficile infection] despite the absence of data showing added benefits with the higher dosing regimens," he said. "This practice not only increases medication and/or preparation costs, but it may also increase the potential for untoward effects in patients being treated with the higher doses (e.g., increased risks for vancomycin-resistant enterococci colonization or higher likelihood to further alter the GI flora)."
Since the inception of the Antimicrobial Stewardship Program at Montefiore Medical Center in 2008, Dr. Chung and his associates observed a shift in oral vancomycin prescribing practices from a higher dosing regimen to a lower dosing regimen. "Because of this change in practice, we wished to evaluate the efficacy of the different oral vancomycin dosing regimen in order to ensure treatment outcomes remained unchanged at our institution," he said. To do so, the researchers retrospectively reviewed clinical outcomes of 300 adult patients treated with oral vancomycin at the medical center between 2006 and 2010. They looked at clinical parameters, concomitant antibiotics, in-hospital mortality, and 30-day readmission.
The primary endpoint was clinical improvement at 72 hours of oral vancomycin. Secondary endpoints included clinical improvement at end of therapy or discharge, length of stay, in-hospital mortality, and 30-day readmission rate.
Of the 300 patients, 197 were prescribed oral vancomycin 125 mg q.i.d. (low-dose group) and 103 patients were prescribed 250 mg or higher q.i.d. (high-dose group). Dr. Chung and his associates reported that clinical improvement assessed 72 hours after starting oral vancomycin therapy was observed in 85% and 86% of patients in the low- and high-dose groups, respectively (P less than 0.05). Rates of clinical improvement at end of therapy or time of hospital discharge between the two groups were also found to be similar (93% vs. 96%), as were total length of hospital stay (20 days vs. 19 days), in-hospital mortality (15% vs. 23%), and rates of 30-day readmission (34% vs. 24%).
"The finding that oral vancomycin 125 mg q.i.d. works as well as higher doses for severe uncomplicated C. difficile infection for the most part is not a surprise to us, as some evidence already exists in the literature," said Dr. Chung, also of Albert Einstein College of Medicine in New York. "Our finding only confirmed what is already known."
Limitations include the fact that this is a single-center study, and that it is subject to selection bias because of its retrospective design, Dr. Chung said. "However, we took steps to ensure that all patients treated with oral vancomycin who had laboratory-confirmed [C. difficile infection] and symptoms consistent with [C. difficile infection] were included in the study," he said.
Dr. Chung said that he had no relevant financial conflicts to disclose.
AT ICAAC 2013
Pearls in clinical diagnosis of pertussis
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
VAIL, COLO. – One of the most useful signs that a young infant with an afebrile coughing illness has pertussis is the combination of an elevated white blood cell count of 20,000 cells/mcL or more plus a lymphocyte count of at least 10,000 cells/mcL.
"This is a pediatric pearl. It’s really a poor man’s way of diagnosing pertussis. It’s an effect of the pertussis toxin spreading throughout the neonate’s body, causing a very high white cell count with absolute lymphocytosis," Dr. Ann-Christine Nyquist said at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
This clinical pearl is part of a highly useful algorithm put forth by the Global Pertussis Initiative in an effort to update and standardize the case definitions of pertussis. Existing case definitions were developed more than 40 years ago and have numerous shortcomings.
The group developed a three-pronged, age-based algorithm, reflecting an understanding that the key manifestations of pertussis are different in infants aged 0-3 months, children aged 4 months to 9 years, and adolescents or adults.
According to the algorithm, the presence of an elevated WBC count with absolute lymphocytosis in an infant up to 3 months old with an afebrile illness and a cough of less than 3 weeks’ duration that’s increasing in frequency and severity is "virtually diagnostic" of pertussis (Clin. Infect. Dis. 2012;54:1756-64).
Another key feature of pertussis – and this one applies across the age spectrum, from infants to adults – is that the coryza remains watery and doesn’t become purulent, unlike in most viral respiratory infections.
"That green snotty nose doesn’t usually happen when kids have pertussis," explained Dr. Nyquist, professor of pediatrics at the University of Colorado, Denver.
Similarly, in patients of all ages the pertussis cough, even as it worsens, does not become truly productive.
To help nail down the diagnosis of pertussis in infants, the key question to ask parents is, "Is there an adult or adolescent in your family who’s had the most severe cough in their life?" Most infants with pertussis will have had close exposure to an older family member with a prolonged afebrile coughing illness, Dr. Nyquist noted.
In the 4-month to 9-year-old age group, the cough becomes more paroxysmal. The key indicators of pertussis in this age group, according to the Global Pertussis Initiative algorithm, are worsening paroxysmal nonproductive cough of at least 7 days’ duration in an afebrile child with nonpurulent coryza.
This same triad – worsening paroxysmal nonproductive cough, afebrile illness, and nonpurulent coryza – also has high sensitivity and good specificity for the clinical diagnosis of pertussis in adolescents and adults. In addition, the algorithm highlights another useful clue to the diagnosis in patients in this age range: the occurrence of sweating episodes between coughing paroxysms.
In terms of laboratory diagnostics, real-time PCR and culture of nasopharyngeal mucus are most useful in the first 3 weeks after illness onset. Serology is a challenge because the results are influenced by the effects of vaccination; it shouldn’t be used to diagnose pertussis within 1 year following inoculation with any pertussis vaccine because it’s impossible to tell if a positive result represents a response to the vaccine or to infection.
Also, serology can’t distinguish between Bordetella pertussis and B. parapertussis infection. Most PCR tests can. It’s an important distinction because B. parapertussis turns out to be the pathogen in roughly 15% of cases of coughing illnesses similar to pertussis. B. parapertussis infection isn’t vaccine preventable, and its treatment hasn’t been well studied.
The expert consensus is that direct fluorescent antibody testing should be discouraged as a tool to diagnose pertussis because of its unreliable sensitivity and specificity. IgG anti–pertussis toxin ELISA testing is superior to IgA anti–pertussis toxin ELISA, which has a high false-negative rate, Dr. Nyquist observed.
She reported having no financial relationships with any commercial interests.
The push is on for universal influenza vaccines
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
VAIL, COLO. – A universal influenza vaccine is not a pipe dream.
"There is a really big push for this now. It’s a major goal," Dr. Wayne Sullender observed at a conference on pediatric infectious diseases sponsored by the Children’s Hospital Colorado.
The impetus for development of a universal influenza vaccine is that influenza still poses a major public health threat despite the widespread availability of current vaccines. Worldwide, roughly 1.4 million children die of pneumonia each year, more than from malaria, AIDS, and measles combined. It has been estimated that each year up to 112,000 children under age 5 die of influenza-associated acute lower respiratory tract infection, with 99% of the deaths occurring in developing countries.
A universal influenza vaccine could render obsolete the current costly, time-consuming, and uncertainty-ridden process of reformulating flu vaccines from year to year based upon expert consensus as to what the epidemic strains are most likely to be in the next flu season. This is a guessing game, and vaccine efficacy is reduced in seasons where the match isn’t good.
Also, a universal vaccine could conceivably protect against highly pathogenic pandemic influenza viruses, such as the swine flu H3N2 or the even more lethal avian H7N9 influenza virus. And even if a universal influenza vaccine wasn’t fully protective against threatening pandemic strains, it could perhaps prime vaccine recipients so they are no longer immunologically naïve, explained Dr. Sullender, an infectious diseases expert who is a visiting professor of pediatrics at the University of Colorado, Denver.
All of the universal flu vaccines in clinical development employ various highly conserved regions of influenza virus target antigens. In focusing on these targets shared by different influenza virus subtypes, the goal is to develop vaccines that protect against seasonal influenza, even as the viruses engage in their relentless antigenic shift and drift, as well as to provide immunity against emerging pandemic strains having the potential for rapid spread and high mortality throughout the world.
Among the novel strategies for development of a universal influenza vaccine being pursued in laboratories around the world, one of the most promising in Dr. Sullender’s view involves stimulation of anti-M2e antibodies. M2 is a proton-selective ion channel that plays a key role in virus assembly. M2 is found on the surface of virus-infected cells. Its advantage as an antigen is that its sequence is virtually the same in every influenza virus isolated since the 1930s. Natural infection doesn’t stimulate much of an antibody response to M2. Yet even though M2e antibodies are not virus-neutralizing, it appears they are able to kill influenza virus by other mechanisms.
Another active area involves antibody responses to highly conserved epitopes on hemagglutinin. A region of vulnerability has been identified in the stem region of hemagglutinin, the viral spike. If the amino acids in this stem antibody binding site prove to be so important to the structure of hemagglutinin that the virus can’t tolerate change there, then the virus wouldn’t be able to adapt to and mutate away from a vaccine targeting this site via stimulation of neutralizing antibodies. Such a vaccine could very well be a universal influenza vaccine.
In addition, a novel epitope has been identified on the globular head of the H1N1 influenza virus hemagglutinin. Investigators have isolated a human monoclonal antibody that recognizes this epitope and neutralizes many different H1N1 strains. This could eventually lead to production of vaccines that incorporate protection against the severe H1N1 flu.
With regard to the avian-origin H7N9 influenza A virus that emerged last winter in China, Dr. Sullender commented, "This one is pretty scary." First estimates are that one-third of people hospitalized with the infection died. However, less severe cases were probably underrecognized, and it’s unlikely the death rate will remain this high.
The human-to-human transmission rate of H7N9 is low. Still, there are several reasons for concern about this virus. Although the pathogenicity in birds is low, the virus appears to have enhanced replication and virulence in humans. And H7N9 is already resistant to amantadine. Moreover, cases of resistance to oseltamivir and zanamivir have been reported.
The potential for mayhem due to H7N9 is such that vaccine development efforts are already underway. Among infectious respiratory disease experts, all eyes are on the coming flu season in Asia and what role H7N9 will play.
"Time will tell whether this will be just another story that comes and goes with influenza, or it becomes a more long-lasting problem," he said.
Experts all agree that it’s not a matter of "if’" another worldwide, high-mortality flu pandemic such as the one that occurred after the end of World War I will happen, it’s simply a question of "when."
"It might occur in 5 years, or it might not happen during our lifetime," according to Dr. Sullender.
He reported receiving research funding from the Centers for Disease Control and Prevention and has no relevant financial relationships.
EXPERT OPINION FROM THE ANNUAL PEDIATRIC INFECTIOUS DISEASES CONFERENCE
New urticaria guidelines stress simplicity
NEW YORK – New guidelines for the diagnosis and treatment of urticaria have been endorsed by 15 professional organizations so far and are now being prepared for publication, according to a consensus meeting participant who summarized key points at the American Academy of Dermatology summer meeting.
The guidelines, developed at an earlier conference held in Berlin attended by experts from 39 countries, are straightforward, relatively simple, "and truly developed for global application," according to Dr. Kiran Godse of Patil Medical College and Hospital, Navi Mumbai, India. The guidelines represent a joint initiative of the Dermatology Section of the European Academy of Allergology and Clinical Immunology (EAACI), the Global Allergy and Asthma European Network (GA2LEN), the European Dermatology Forum (EDF), the American Academy of Allergy, Asthma and Immunology (AAAAI), and the World Allergy Organization (WAO).
The simplicity of the guidelines starts with the definition of urticaria. It consists of three characteristics: "wheals, angioedema, or both." While the definition goes on to specify that these conditions should be differentiated from autoinflammatory syndromes, hereditary angioedema, and other diseases that produce hives or swelling, the new guidelines abandon the term "idiopathic."
"Our understanding of the etiology and pathogenesis has advanced to the point that we can identify the causes in most cases," said Dr. Godse, indicating that classifying cases as "idiopathic" without further investigation is unhelpful when the goal is to find and avoid triggers.
A number of subclassifications, such as spontaneous urticaria, inducible urticaria, acute urticaria, and chronic urticaria, are defined and employed to guide clinical management. In patients with acute urticaria, diagnostic testing beyond a careful history is not recommended, except when avoidance strategies fail and recurrences are common.
Even in chronic urticaria, which is defined as symptoms persisting for at least 6 months, Dr. Godse said that the guidelines recommend "limited" initial diagnostic studies.
By relying on careful patient history rather than clinical tests to differentiate the major forms of this disease, such as cold urticaria, heat urticaria, delayed pressure urticaria, solar urticaria, and symptomatic dermographism, the guidelines in effect propose that underlying etiologies do not usually require an extensive workup. However, the guidelines do advise more extensive tests in individuals with persistent and significant disease, which can be measured with the Chronic Urticaria Quality of Life (CU-QoL) and the Angioedema Quality of Life (Ae-QoL) instruments. Both are strongly recommended for baseline assessment of symptom burden.
The treatment goal of the stepwise management is clear: complete absence of symptoms. "Treat the disease until it is gone," said Dr. Godse, summarizing this recommendation.
If symptoms cannot be eliminated simply by avoiding causes and aggravating factors, the guidelines identify second-generation, nonsedating H1 antihistamines as the first-line pharmacotherapy. Dr. Godse said that the guidelines specifically recommend continuous rather than on-demand regimens at the lowest effective dose. However, if symptoms persist after 1-4 weeks of therapy, the dosing frequency should be increased before moving to adjunctive use of additional therapies. Adjunctive therapies listed in the guidelines include omalizumab, cyclosporine A, and montelukast. The first two of these options received strong recommendations on the basis of a high level of evidence, but the third was given a weak recommendation on the basis of a low level of evidence.
In those who fail these therapies, the list of alternatives is lengthy and includes a short course of corticosteroids, immunomodulating therapies such as methotrexate, and intravenous immunoglobulins. While any one of these may be useful in an individual patient, the overall evidence of benefit was considered to be of relatively low quality.
Ultimately, the guidelines attempt to define an approach that is uniformly applicable across diverse populations, a full range of possible etiologies, and within different systems of medical care, according to Dr. Godse.
Asked for their opinion after hearing the guidelines explained at the meeting, Dr. Paul Schneiderman and Dr. Aaron Warshawsky said they were favorably impressed. Both thought the guidelines were clear, reasonable, and potentially helpful in clinical practice. Dr. Schneiderman, an associate clinical professor of dermatology at Yale University, New Haven, Conn., who maintains a private practice in Syosset, N.Y., reported that he will be able to better judge the clinical applicability of the new guidelines when he sees the full publication, but both he and Dr. Warshawsky, a dermatologist in private practice in Poughkeepsie, N.Y., agreed that advances in urticaria justify updated guidelines.
Dr. Godse reported no financial disclosures relevant to his presentation.
NEW YORK – New guidelines for the diagnosis and treatment of urticaria have been endorsed by 15 professional organizations so far and are now being prepared for publication, according to a consensus meeting participant who summarized key points at the American Academy of Dermatology summer meeting.
The guidelines, developed at an earlier conference held in Berlin attended by experts from 39 countries, are straightforward, relatively simple, "and truly developed for global application," according to Dr. Kiran Godse of Patil Medical College and Hospital, Navi Mumbai, India. The guidelines represent a joint initiative of the Dermatology Section of the European Academy of Allergology and Clinical Immunology (EAACI), the Global Allergy and Asthma European Network (GA2LEN), the European Dermatology Forum (EDF), the American Academy of Allergy, Asthma and Immunology (AAAAI), and the World Allergy Organization (WAO).
The simplicity of the guidelines starts with the definition of urticaria. It consists of three characteristics: "wheals, angioedema, or both." While the definition goes on to specify that these conditions should be differentiated from autoinflammatory syndromes, hereditary angioedema, and other diseases that produce hives or swelling, the new guidelines abandon the term "idiopathic."
"Our understanding of the etiology and pathogenesis has advanced to the point that we can identify the causes in most cases," said Dr. Godse, indicating that classifying cases as "idiopathic" without further investigation is unhelpful when the goal is to find and avoid triggers.
A number of subclassifications, such as spontaneous urticaria, inducible urticaria, acute urticaria, and chronic urticaria, are defined and employed to guide clinical management. In patients with acute urticaria, diagnostic testing beyond a careful history is not recommended, except when avoidance strategies fail and recurrences are common.
Even in chronic urticaria, which is defined as symptoms persisting for at least 6 months, Dr. Godse said that the guidelines recommend "limited" initial diagnostic studies.
By relying on careful patient history rather than clinical tests to differentiate the major forms of this disease, such as cold urticaria, heat urticaria, delayed pressure urticaria, solar urticaria, and symptomatic dermographism, the guidelines in effect propose that underlying etiologies do not usually require an extensive workup. However, the guidelines do advise more extensive tests in individuals with persistent and significant disease, which can be measured with the Chronic Urticaria Quality of Life (CU-QoL) and the Angioedema Quality of Life (Ae-QoL) instruments. Both are strongly recommended for baseline assessment of symptom burden.
The treatment goal of the stepwise management is clear: complete absence of symptoms. "Treat the disease until it is gone," said Dr. Godse, summarizing this recommendation.
If symptoms cannot be eliminated simply by avoiding causes and aggravating factors, the guidelines identify second-generation, nonsedating H1 antihistamines as the first-line pharmacotherapy. Dr. Godse said that the guidelines specifically recommend continuous rather than on-demand regimens at the lowest effective dose. However, if symptoms persist after 1-4 weeks of therapy, the dosing frequency should be increased before moving to adjunctive use of additional therapies. Adjunctive therapies listed in the guidelines include omalizumab, cyclosporine A, and montelukast. The first two of these options received strong recommendations on the basis of a high level of evidence, but the third was given a weak recommendation on the basis of a low level of evidence.
In those who fail these therapies, the list of alternatives is lengthy and includes a short course of corticosteroids, immunomodulating therapies such as methotrexate, and intravenous immunoglobulins. While any one of these may be useful in an individual patient, the overall evidence of benefit was considered to be of relatively low quality.
Ultimately, the guidelines attempt to define an approach that is uniformly applicable across diverse populations, a full range of possible etiologies, and within different systems of medical care, according to Dr. Godse.
Asked for their opinion after hearing the guidelines explained at the meeting, Dr. Paul Schneiderman and Dr. Aaron Warshawsky said they were favorably impressed. Both thought the guidelines were clear, reasonable, and potentially helpful in clinical practice. Dr. Schneiderman, an associate clinical professor of dermatology at Yale University, New Haven, Conn., who maintains a private practice in Syosset, N.Y., reported that he will be able to better judge the clinical applicability of the new guidelines when he sees the full publication, but both he and Dr. Warshawsky, a dermatologist in private practice in Poughkeepsie, N.Y., agreed that advances in urticaria justify updated guidelines.
Dr. Godse reported no financial disclosures relevant to his presentation.
NEW YORK – New guidelines for the diagnosis and treatment of urticaria have been endorsed by 15 professional organizations so far and are now being prepared for publication, according to a consensus meeting participant who summarized key points at the American Academy of Dermatology summer meeting.
The guidelines, developed at an earlier conference held in Berlin attended by experts from 39 countries, are straightforward, relatively simple, "and truly developed for global application," according to Dr. Kiran Godse of Patil Medical College and Hospital, Navi Mumbai, India. The guidelines represent a joint initiative of the Dermatology Section of the European Academy of Allergology and Clinical Immunology (EAACI), the Global Allergy and Asthma European Network (GA2LEN), the European Dermatology Forum (EDF), the American Academy of Allergy, Asthma and Immunology (AAAAI), and the World Allergy Organization (WAO).
The simplicity of the guidelines starts with the definition of urticaria. It consists of three characteristics: "wheals, angioedema, or both." While the definition goes on to specify that these conditions should be differentiated from autoinflammatory syndromes, hereditary angioedema, and other diseases that produce hives or swelling, the new guidelines abandon the term "idiopathic."
"Our understanding of the etiology and pathogenesis has advanced to the point that we can identify the causes in most cases," said Dr. Godse, indicating that classifying cases as "idiopathic" without further investigation is unhelpful when the goal is to find and avoid triggers.
A number of subclassifications, such as spontaneous urticaria, inducible urticaria, acute urticaria, and chronic urticaria, are defined and employed to guide clinical management. In patients with acute urticaria, diagnostic testing beyond a careful history is not recommended, except when avoidance strategies fail and recurrences are common.
Even in chronic urticaria, which is defined as symptoms persisting for at least 6 months, Dr. Godse said that the guidelines recommend "limited" initial diagnostic studies.
By relying on careful patient history rather than clinical tests to differentiate the major forms of this disease, such as cold urticaria, heat urticaria, delayed pressure urticaria, solar urticaria, and symptomatic dermographism, the guidelines in effect propose that underlying etiologies do not usually require an extensive workup. However, the guidelines do advise more extensive tests in individuals with persistent and significant disease, which can be measured with the Chronic Urticaria Quality of Life (CU-QoL) and the Angioedema Quality of Life (Ae-QoL) instruments. Both are strongly recommended for baseline assessment of symptom burden.
The treatment goal of the stepwise management is clear: complete absence of symptoms. "Treat the disease until it is gone," said Dr. Godse, summarizing this recommendation.
If symptoms cannot be eliminated simply by avoiding causes and aggravating factors, the guidelines identify second-generation, nonsedating H1 antihistamines as the first-line pharmacotherapy. Dr. Godse said that the guidelines specifically recommend continuous rather than on-demand regimens at the lowest effective dose. However, if symptoms persist after 1-4 weeks of therapy, the dosing frequency should be increased before moving to adjunctive use of additional therapies. Adjunctive therapies listed in the guidelines include omalizumab, cyclosporine A, and montelukast. The first two of these options received strong recommendations on the basis of a high level of evidence, but the third was given a weak recommendation on the basis of a low level of evidence.
In those who fail these therapies, the list of alternatives is lengthy and includes a short course of corticosteroids, immunomodulating therapies such as methotrexate, and intravenous immunoglobulins. While any one of these may be useful in an individual patient, the overall evidence of benefit was considered to be of relatively low quality.
Ultimately, the guidelines attempt to define an approach that is uniformly applicable across diverse populations, a full range of possible etiologies, and within different systems of medical care, according to Dr. Godse.
Asked for their opinion after hearing the guidelines explained at the meeting, Dr. Paul Schneiderman and Dr. Aaron Warshawsky said they were favorably impressed. Both thought the guidelines were clear, reasonable, and potentially helpful in clinical practice. Dr. Schneiderman, an associate clinical professor of dermatology at Yale University, New Haven, Conn., who maintains a private practice in Syosset, N.Y., reported that he will be able to better judge the clinical applicability of the new guidelines when he sees the full publication, but both he and Dr. Warshawsky, a dermatologist in private practice in Poughkeepsie, N.Y., agreed that advances in urticaria justify updated guidelines.
Dr. Godse reported no financial disclosures relevant to his presentation.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2013