User login
Even COVID-19 can’t stop a true optimist
Squeezing a little lemonade out of COVID-19
We like to think of ourselves as optimists here at LOTME. A glass is half full, the sky is partly sunny, and our motto is “Always look on the bright side of insanity.” Then again, our motto before that was “LOTME: Where science meets stupid,” so what do we know?
Anyway, it’s that upbeat, can-do attitude that allows us to say something positive – two somethings, actually – about the insanity that is COVID-19.
Our journey to the bright side begins, oddly enough, in the courtroom. Seems that our old friend, the face mask, is something of a lie-detector aid for juries. The authors of a recent literature review of studies on deception “found that facial expressions and other forms of nonverbal behaviour are an unreliable indicator of deceit,” according to a statement from the University of Portsmouth, where the analysis was conducted.
The one study that directly examined the role of face coverings in court proceedings showed that, “by taking away the distraction of nonverbal behaviours, observers had to rely on speech content, which turned out to be better for detecting lies,” the university said.
The second stage of our positivity trek brings us to the National Trends in Disability Employment monthly update, where we see a fourth consecutive month of gains for people with disabilities despite the larger trend of declines among those without disabilities.
Here are some numbers from the Kessler Foundation and the University of New Hampshire’s Institute on Disability to tell the story: From October to November, the employment-to-population ratio increased 4.2% for working-age people with disabilities, compared with 0.4% for people without disabilities. At the same time, the labor force participation rate rose 2.4% for working-age people with disabilities and just 0.1% for working-age people without disabilities.
Both indicators surpassed their historic highs, Andrew Houtenville, PhD, director of the Institute on Disability, said in the update. “These gains suggest that the restructuring resulting from the pandemic may be benefiting people with disabilities. Ironically, it may have taken a pandemic to shake the labor market loose for people with disabilities.”
And that is how a world-class optimist turns one gigantic lemon into lemonade.
Cut the cheese for better sleep
So, we’ve already talked about the TikTok lettuce tea hack that’s supposed to help us sleep better. Well, there’s another food that could have the opposite effect.
According to an article from the BBC, cheese has something of a reputation. Ever since the 1960s, when a researcher noted that one patient’s nightmares stopped after he quit eating an ounce or two of cheddar each night, there’s been speculation that cheese gives you weird dreams. Another study in 2005 suggested certain types of cheese cause certain types of dreams. Blue cheese for vivid dreams and cheddar cheese for celebrity cameos.
But is there any truth to it at all?
Regardless of what we eat, going to bed hungry could cause vivid dreams, according to research by Tore Nielsen, director of the University of Montreal’s dream and nightmare lab. The 2015 study showed that high lactose could have an effect on dreams.
In that study, 17% of participants said their dreams were influenced by what they ate, but the kicker was that dairy products were the foods most reported as causing the weird dreams, the BBC noted.
“It’s likely an indirect effect in that lactose produces symptoms like gas, bloating and diarrhoea and influences dreams, as dreams draw on somatic sources like this. And if you have certain kinds of intolerances, you still may be likely to eat those foods sometimes,” Mr. Nielsen told the BBC.
There’s also the theory that it’s all in the timing of consumption. Are you the type of person to sneak a slice of cheese from the fridge late at night? (Nods.) Same.
“One reason cheese and nightmares come about is that eating later before bed is more likely to disrupt sleep, and cheese can be hard to digest,” said Charlotte Gupta, a research fellow at Central Queensland University in Australia and a coauthor of a 2020 review on how diet affects our sleep.
So as tempting as it is, maybe skip sprinkling Parmesan cheese shreds into your mouth at the open fridge before bed.
Teeing up against Parkinson’s
For the nearly 1 million people in the United States with Parkinson’s disease, tai chi is one of the best ways to alleviate the symptoms. The average Parkinson’s patient, however, is going to be on the older side and more likely to view the martial art as some sort of communist plot. And would you participate in a communist plot? We don’t think so.
One group of researchers saw that patients weren’t keeping up with their therapy and decided to try a different activity, something that older people would be more likely to stick with. Something a bit more stereotypical. No, not shuffleboard. They tried golf.
“Golf is popular – the most popular sport for people over the age of 55 – which might encourage people to try it and stick with it,” study author Anne-Marie A. Wills, MD, of Massachusetts General Hospital, Boston, said in a Study Finds report.
In a small study, the investigators had a group of patients with Parkinson’s regularly go to a driving range for 10 weeks to hit golf balls (all expenses paid too, and that’s a big deal for golf), while another group continued with their tai chi.
At the end of the study, the 8 patients who went to the driving range had significantly better results in a Parkinson’s mobility test than those of the 12 patients in the tai chi group. In addition, the golf-group participants said they were more likely to continue with their therapy than were those who did tai chi.
Despite the small size of the study, the research team said the results certainly warrant further research. After all, the best sort of therapy is the kind that actually gets done. And golf just gets in your head. The eternal quest to add distance, to straighten out that annoying slice, to stop thinning half your chips, to make those annoying 4-footers. ... Maybe that’s just us.
Squeezing a little lemonade out of COVID-19
We like to think of ourselves as optimists here at LOTME. A glass is half full, the sky is partly sunny, and our motto is “Always look on the bright side of insanity.” Then again, our motto before that was “LOTME: Where science meets stupid,” so what do we know?
Anyway, it’s that upbeat, can-do attitude that allows us to say something positive – two somethings, actually – about the insanity that is COVID-19.
Our journey to the bright side begins, oddly enough, in the courtroom. Seems that our old friend, the face mask, is something of a lie-detector aid for juries. The authors of a recent literature review of studies on deception “found that facial expressions and other forms of nonverbal behaviour are an unreliable indicator of deceit,” according to a statement from the University of Portsmouth, where the analysis was conducted.
The one study that directly examined the role of face coverings in court proceedings showed that, “by taking away the distraction of nonverbal behaviours, observers had to rely on speech content, which turned out to be better for detecting lies,” the university said.
The second stage of our positivity trek brings us to the National Trends in Disability Employment monthly update, where we see a fourth consecutive month of gains for people with disabilities despite the larger trend of declines among those without disabilities.
Here are some numbers from the Kessler Foundation and the University of New Hampshire’s Institute on Disability to tell the story: From October to November, the employment-to-population ratio increased 4.2% for working-age people with disabilities, compared with 0.4% for people without disabilities. At the same time, the labor force participation rate rose 2.4% for working-age people with disabilities and just 0.1% for working-age people without disabilities.
Both indicators surpassed their historic highs, Andrew Houtenville, PhD, director of the Institute on Disability, said in the update. “These gains suggest that the restructuring resulting from the pandemic may be benefiting people with disabilities. Ironically, it may have taken a pandemic to shake the labor market loose for people with disabilities.”
And that is how a world-class optimist turns one gigantic lemon into lemonade.
Cut the cheese for better sleep
So, we’ve already talked about the TikTok lettuce tea hack that’s supposed to help us sleep better. Well, there’s another food that could have the opposite effect.
According to an article from the BBC, cheese has something of a reputation. Ever since the 1960s, when a researcher noted that one patient’s nightmares stopped after he quit eating an ounce or two of cheddar each night, there’s been speculation that cheese gives you weird dreams. Another study in 2005 suggested certain types of cheese cause certain types of dreams. Blue cheese for vivid dreams and cheddar cheese for celebrity cameos.
But is there any truth to it at all?
Regardless of what we eat, going to bed hungry could cause vivid dreams, according to research by Tore Nielsen, director of the University of Montreal’s dream and nightmare lab. The 2015 study showed that high lactose could have an effect on dreams.
In that study, 17% of participants said their dreams were influenced by what they ate, but the kicker was that dairy products were the foods most reported as causing the weird dreams, the BBC noted.
“It’s likely an indirect effect in that lactose produces symptoms like gas, bloating and diarrhoea and influences dreams, as dreams draw on somatic sources like this. And if you have certain kinds of intolerances, you still may be likely to eat those foods sometimes,” Mr. Nielsen told the BBC.
There’s also the theory that it’s all in the timing of consumption. Are you the type of person to sneak a slice of cheese from the fridge late at night? (Nods.) Same.
“One reason cheese and nightmares come about is that eating later before bed is more likely to disrupt sleep, and cheese can be hard to digest,” said Charlotte Gupta, a research fellow at Central Queensland University in Australia and a coauthor of a 2020 review on how diet affects our sleep.
So as tempting as it is, maybe skip sprinkling Parmesan cheese shreds into your mouth at the open fridge before bed.
Teeing up against Parkinson’s
For the nearly 1 million people in the United States with Parkinson’s disease, tai chi is one of the best ways to alleviate the symptoms. The average Parkinson’s patient, however, is going to be on the older side and more likely to view the martial art as some sort of communist plot. And would you participate in a communist plot? We don’t think so.
One group of researchers saw that patients weren’t keeping up with their therapy and decided to try a different activity, something that older people would be more likely to stick with. Something a bit more stereotypical. No, not shuffleboard. They tried golf.
“Golf is popular – the most popular sport for people over the age of 55 – which might encourage people to try it and stick with it,” study author Anne-Marie A. Wills, MD, of Massachusetts General Hospital, Boston, said in a Study Finds report.
In a small study, the investigators had a group of patients with Parkinson’s regularly go to a driving range for 10 weeks to hit golf balls (all expenses paid too, and that’s a big deal for golf), while another group continued with their tai chi.
At the end of the study, the 8 patients who went to the driving range had significantly better results in a Parkinson’s mobility test than those of the 12 patients in the tai chi group. In addition, the golf-group participants said they were more likely to continue with their therapy than were those who did tai chi.
Despite the small size of the study, the research team said the results certainly warrant further research. After all, the best sort of therapy is the kind that actually gets done. And golf just gets in your head. The eternal quest to add distance, to straighten out that annoying slice, to stop thinning half your chips, to make those annoying 4-footers. ... Maybe that’s just us.
Squeezing a little lemonade out of COVID-19
We like to think of ourselves as optimists here at LOTME. A glass is half full, the sky is partly sunny, and our motto is “Always look on the bright side of insanity.” Then again, our motto before that was “LOTME: Where science meets stupid,” so what do we know?
Anyway, it’s that upbeat, can-do attitude that allows us to say something positive – two somethings, actually – about the insanity that is COVID-19.
Our journey to the bright side begins, oddly enough, in the courtroom. Seems that our old friend, the face mask, is something of a lie-detector aid for juries. The authors of a recent literature review of studies on deception “found that facial expressions and other forms of nonverbal behaviour are an unreliable indicator of deceit,” according to a statement from the University of Portsmouth, where the analysis was conducted.
The one study that directly examined the role of face coverings in court proceedings showed that, “by taking away the distraction of nonverbal behaviours, observers had to rely on speech content, which turned out to be better for detecting lies,” the university said.
The second stage of our positivity trek brings us to the National Trends in Disability Employment monthly update, where we see a fourth consecutive month of gains for people with disabilities despite the larger trend of declines among those without disabilities.
Here are some numbers from the Kessler Foundation and the University of New Hampshire’s Institute on Disability to tell the story: From October to November, the employment-to-population ratio increased 4.2% for working-age people with disabilities, compared with 0.4% for people without disabilities. At the same time, the labor force participation rate rose 2.4% for working-age people with disabilities and just 0.1% for working-age people without disabilities.
Both indicators surpassed their historic highs, Andrew Houtenville, PhD, director of the Institute on Disability, said in the update. “These gains suggest that the restructuring resulting from the pandemic may be benefiting people with disabilities. Ironically, it may have taken a pandemic to shake the labor market loose for people with disabilities.”
And that is how a world-class optimist turns one gigantic lemon into lemonade.
Cut the cheese for better sleep
So, we’ve already talked about the TikTok lettuce tea hack that’s supposed to help us sleep better. Well, there’s another food that could have the opposite effect.
According to an article from the BBC, cheese has something of a reputation. Ever since the 1960s, when a researcher noted that one patient’s nightmares stopped after he quit eating an ounce or two of cheddar each night, there’s been speculation that cheese gives you weird dreams. Another study in 2005 suggested certain types of cheese cause certain types of dreams. Blue cheese for vivid dreams and cheddar cheese for celebrity cameos.
But is there any truth to it at all?
Regardless of what we eat, going to bed hungry could cause vivid dreams, according to research by Tore Nielsen, director of the University of Montreal’s dream and nightmare lab. The 2015 study showed that high lactose could have an effect on dreams.
In that study, 17% of participants said their dreams were influenced by what they ate, but the kicker was that dairy products were the foods most reported as causing the weird dreams, the BBC noted.
“It’s likely an indirect effect in that lactose produces symptoms like gas, bloating and diarrhoea and influences dreams, as dreams draw on somatic sources like this. And if you have certain kinds of intolerances, you still may be likely to eat those foods sometimes,” Mr. Nielsen told the BBC.
There’s also the theory that it’s all in the timing of consumption. Are you the type of person to sneak a slice of cheese from the fridge late at night? (Nods.) Same.
“One reason cheese and nightmares come about is that eating later before bed is more likely to disrupt sleep, and cheese can be hard to digest,” said Charlotte Gupta, a research fellow at Central Queensland University in Australia and a coauthor of a 2020 review on how diet affects our sleep.
So as tempting as it is, maybe skip sprinkling Parmesan cheese shreds into your mouth at the open fridge before bed.
Teeing up against Parkinson’s
For the nearly 1 million people in the United States with Parkinson’s disease, tai chi is one of the best ways to alleviate the symptoms. The average Parkinson’s patient, however, is going to be on the older side and more likely to view the martial art as some sort of communist plot. And would you participate in a communist plot? We don’t think so.
One group of researchers saw that patients weren’t keeping up with their therapy and decided to try a different activity, something that older people would be more likely to stick with. Something a bit more stereotypical. No, not shuffleboard. They tried golf.
“Golf is popular – the most popular sport for people over the age of 55 – which might encourage people to try it and stick with it,” study author Anne-Marie A. Wills, MD, of Massachusetts General Hospital, Boston, said in a Study Finds report.
In a small study, the investigators had a group of patients with Parkinson’s regularly go to a driving range for 10 weeks to hit golf balls (all expenses paid too, and that’s a big deal for golf), while another group continued with their tai chi.
At the end of the study, the 8 patients who went to the driving range had significantly better results in a Parkinson’s mobility test than those of the 12 patients in the tai chi group. In addition, the golf-group participants said they were more likely to continue with their therapy than were those who did tai chi.
Despite the small size of the study, the research team said the results certainly warrant further research. After all, the best sort of therapy is the kind that actually gets done. And golf just gets in your head. The eternal quest to add distance, to straighten out that annoying slice, to stop thinning half your chips, to make those annoying 4-footers. ... Maybe that’s just us.
COVID-19 asymptomatic infection rate remains high
Based on data from a meta-analysis of 95 studies that included nearly 30,000,000 individuals, the pooled percentage of asymptomatic COVID-19 infections was 0.25% in the tested population and 40.5% among confirmed cases.
, wrote Qiuyue Ma, PhD, and colleagues of Peking University, Beijing.
In a study published in JAMA Network Open the researchers identified 44 cross-sectional studies, 41 cohort studies, seven case series, and three case series on transmission studies. A total of 74 studies were conducted in developed countries, including those in Europe, North America, and Asia. Approximately one-third (37) of the studies were conducted among health care workers or in-hospital patients, 17 among nursing home staff or residents, and 14 among community residents. In addition, 13 studies involved pregnant women, eight involved air or cruise ship travelers, and six involved close contacts of individuals with confirmed infections.
The meta-analysis included 29,776,306 tested individuals; 11,516 of them had asymptomatic infections.
Overall, the pooled percentage of asymptomatic infections among the tested population was 0.25%. In an analysis of different study populations, the percentage was higher in nursing home residents or staff (4.52%), air or cruise ship travelers (2.02%), and pregnant women (2.34%), compared against the pooled percentage.
The pooled percentage of asymptomatic infections among the confirmed population was 40.50%, and this percentage was higher in pregnant women (54.11%), air or cruise ship travelers (52.91%), and nursing home residents or staff (47.53%).
The pooled percentage in the tested population was higher than the overall percentage when the mean age of the study population was 60 years or older (3.69%). By contrast, in the confirmed population, the pooled percentage was higher than the overall percentage when the study population was younger than 20 years (60.2%) or aged 20 to 39 years (49.5%).
The researchers noted in their discussion that the varying percentage of asymptomatic individuals according to community prevalence might impact the heterogeneity of the included studies. They also noted the high number of studies conducted in nursing home populations, groups in which asymptomatic individuals were more likely to be tested.
The study findings were limited by several factors, including the potential for missed studies that were not published at the time of the meta-analysis, as well as the exclusion of studies written in Chinese, the researchers noted. Other limitations included lack of follow-up on presymptomatic and covert infections, and the focus on specific populations, factors that may limit the degree to which the results can be generalized.
However, the results highlight the need to screen for asymptomatic infections, especially in countries where COVID-19 has been better controlled, the researchers said. Management strategies for asymptomatic infections, when identified, should include isolation and contact tracing similar to strategies used with confirmed cases, they added.
More testing needed to catch cases early
“During the initial phase of [the] COVID-19 pandemic, testing was not widely available in the United States or the rest of the world,” Setu Patolia, MD, of Saint Louis University School of Medicine, Missouri, said in an interview. Much of the world still lacks access to COVID-19 testing, and early in the pandemic only severely symptomatic patients were tested, he said. “With new variants, particularly the Omicron variant, which may have mild or minimally symptomatic disease, asymptomatic carriers play an important role in propagation of the pandemic,” he explained. “It is important to know the asymptomatic carrier rate among the general population for the future control of [the] pandemic,” he added.
Dr. Patolia said he was surprised by the study finding that one in 400 people in the general population could be asymptomatic carriers of COVID-19.
“Also, nursing home patients are more at risk of complications of COVID, and I expected that they would have a higher rate of symptomatic disease as compared to [the] general population,” said Dr. Patolia. He was also surprised by the high rate of asymptomatic infections in travelers.
“Physicians should be more aware about the asymptomatic carrier rate, particularly in travelers and nursing home patients,” he noted. “Travelers carry high risk of transferring infection from one region to another region of the world, and physicians should advise them to get tested despite the absence of symptoms,” Dr. Patolia emphasized. “Similarly, once any nursing home patient has been diagnosed with COVID-19, physicians should be more careful with the rest of the nursing home patients and test them despite the absence of the symptoms,” he added.
Dr. Patolia also recommended that pregnant women wear masks to help prevent disease transmission when visiting a doctor’s office or labor unit.
Looking ahead, there is a need for cheaper at-home testing kits so that all vulnerable populations can be tested fast and frequently, Dr. Patolia said.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose. Dr. Patolia has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Based on data from a meta-analysis of 95 studies that included nearly 30,000,000 individuals, the pooled percentage of asymptomatic COVID-19 infections was 0.25% in the tested population and 40.5% among confirmed cases.
, wrote Qiuyue Ma, PhD, and colleagues of Peking University, Beijing.
In a study published in JAMA Network Open the researchers identified 44 cross-sectional studies, 41 cohort studies, seven case series, and three case series on transmission studies. A total of 74 studies were conducted in developed countries, including those in Europe, North America, and Asia. Approximately one-third (37) of the studies were conducted among health care workers or in-hospital patients, 17 among nursing home staff or residents, and 14 among community residents. In addition, 13 studies involved pregnant women, eight involved air or cruise ship travelers, and six involved close contacts of individuals with confirmed infections.
The meta-analysis included 29,776,306 tested individuals; 11,516 of them had asymptomatic infections.
Overall, the pooled percentage of asymptomatic infections among the tested population was 0.25%. In an analysis of different study populations, the percentage was higher in nursing home residents or staff (4.52%), air or cruise ship travelers (2.02%), and pregnant women (2.34%), compared against the pooled percentage.
The pooled percentage of asymptomatic infections among the confirmed population was 40.50%, and this percentage was higher in pregnant women (54.11%), air or cruise ship travelers (52.91%), and nursing home residents or staff (47.53%).
The pooled percentage in the tested population was higher than the overall percentage when the mean age of the study population was 60 years or older (3.69%). By contrast, in the confirmed population, the pooled percentage was higher than the overall percentage when the study population was younger than 20 years (60.2%) or aged 20 to 39 years (49.5%).
The researchers noted in their discussion that the varying percentage of asymptomatic individuals according to community prevalence might impact the heterogeneity of the included studies. They also noted the high number of studies conducted in nursing home populations, groups in which asymptomatic individuals were more likely to be tested.
The study findings were limited by several factors, including the potential for missed studies that were not published at the time of the meta-analysis, as well as the exclusion of studies written in Chinese, the researchers noted. Other limitations included lack of follow-up on presymptomatic and covert infections, and the focus on specific populations, factors that may limit the degree to which the results can be generalized.
However, the results highlight the need to screen for asymptomatic infections, especially in countries where COVID-19 has been better controlled, the researchers said. Management strategies for asymptomatic infections, when identified, should include isolation and contact tracing similar to strategies used with confirmed cases, they added.
More testing needed to catch cases early
“During the initial phase of [the] COVID-19 pandemic, testing was not widely available in the United States or the rest of the world,” Setu Patolia, MD, of Saint Louis University School of Medicine, Missouri, said in an interview. Much of the world still lacks access to COVID-19 testing, and early in the pandemic only severely symptomatic patients were tested, he said. “With new variants, particularly the Omicron variant, which may have mild or minimally symptomatic disease, asymptomatic carriers play an important role in propagation of the pandemic,” he explained. “It is important to know the asymptomatic carrier rate among the general population for the future control of [the] pandemic,” he added.
Dr. Patolia said he was surprised by the study finding that one in 400 people in the general population could be asymptomatic carriers of COVID-19.
“Also, nursing home patients are more at risk of complications of COVID, and I expected that they would have a higher rate of symptomatic disease as compared to [the] general population,” said Dr. Patolia. He was also surprised by the high rate of asymptomatic infections in travelers.
“Physicians should be more aware about the asymptomatic carrier rate, particularly in travelers and nursing home patients,” he noted. “Travelers carry high risk of transferring infection from one region to another region of the world, and physicians should advise them to get tested despite the absence of symptoms,” Dr. Patolia emphasized. “Similarly, once any nursing home patient has been diagnosed with COVID-19, physicians should be more careful with the rest of the nursing home patients and test them despite the absence of the symptoms,” he added.
Dr. Patolia also recommended that pregnant women wear masks to help prevent disease transmission when visiting a doctor’s office or labor unit.
Looking ahead, there is a need for cheaper at-home testing kits so that all vulnerable populations can be tested fast and frequently, Dr. Patolia said.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose. Dr. Patolia has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Based on data from a meta-analysis of 95 studies that included nearly 30,000,000 individuals, the pooled percentage of asymptomatic COVID-19 infections was 0.25% in the tested population and 40.5% among confirmed cases.
, wrote Qiuyue Ma, PhD, and colleagues of Peking University, Beijing.
In a study published in JAMA Network Open the researchers identified 44 cross-sectional studies, 41 cohort studies, seven case series, and three case series on transmission studies. A total of 74 studies were conducted in developed countries, including those in Europe, North America, and Asia. Approximately one-third (37) of the studies were conducted among health care workers or in-hospital patients, 17 among nursing home staff or residents, and 14 among community residents. In addition, 13 studies involved pregnant women, eight involved air or cruise ship travelers, and six involved close contacts of individuals with confirmed infections.
The meta-analysis included 29,776,306 tested individuals; 11,516 of them had asymptomatic infections.
Overall, the pooled percentage of asymptomatic infections among the tested population was 0.25%. In an analysis of different study populations, the percentage was higher in nursing home residents or staff (4.52%), air or cruise ship travelers (2.02%), and pregnant women (2.34%), compared against the pooled percentage.
The pooled percentage of asymptomatic infections among the confirmed population was 40.50%, and this percentage was higher in pregnant women (54.11%), air or cruise ship travelers (52.91%), and nursing home residents or staff (47.53%).
The pooled percentage in the tested population was higher than the overall percentage when the mean age of the study population was 60 years or older (3.69%). By contrast, in the confirmed population, the pooled percentage was higher than the overall percentage when the study population was younger than 20 years (60.2%) or aged 20 to 39 years (49.5%).
The researchers noted in their discussion that the varying percentage of asymptomatic individuals according to community prevalence might impact the heterogeneity of the included studies. They also noted the high number of studies conducted in nursing home populations, groups in which asymptomatic individuals were more likely to be tested.
The study findings were limited by several factors, including the potential for missed studies that were not published at the time of the meta-analysis, as well as the exclusion of studies written in Chinese, the researchers noted. Other limitations included lack of follow-up on presymptomatic and covert infections, and the focus on specific populations, factors that may limit the degree to which the results can be generalized.
However, the results highlight the need to screen for asymptomatic infections, especially in countries where COVID-19 has been better controlled, the researchers said. Management strategies for asymptomatic infections, when identified, should include isolation and contact tracing similar to strategies used with confirmed cases, they added.
More testing needed to catch cases early
“During the initial phase of [the] COVID-19 pandemic, testing was not widely available in the United States or the rest of the world,” Setu Patolia, MD, of Saint Louis University School of Medicine, Missouri, said in an interview. Much of the world still lacks access to COVID-19 testing, and early in the pandemic only severely symptomatic patients were tested, he said. “With new variants, particularly the Omicron variant, which may have mild or minimally symptomatic disease, asymptomatic carriers play an important role in propagation of the pandemic,” he explained. “It is important to know the asymptomatic carrier rate among the general population for the future control of [the] pandemic,” he added.
Dr. Patolia said he was surprised by the study finding that one in 400 people in the general population could be asymptomatic carriers of COVID-19.
“Also, nursing home patients are more at risk of complications of COVID, and I expected that they would have a higher rate of symptomatic disease as compared to [the] general population,” said Dr. Patolia. He was also surprised by the high rate of asymptomatic infections in travelers.
“Physicians should be more aware about the asymptomatic carrier rate, particularly in travelers and nursing home patients,” he noted. “Travelers carry high risk of transferring infection from one region to another region of the world, and physicians should advise them to get tested despite the absence of symptoms,” Dr. Patolia emphasized. “Similarly, once any nursing home patient has been diagnosed with COVID-19, physicians should be more careful with the rest of the nursing home patients and test them despite the absence of the symptoms,” he added.
Dr. Patolia also recommended that pregnant women wear masks to help prevent disease transmission when visiting a doctor’s office or labor unit.
Looking ahead, there is a need for cheaper at-home testing kits so that all vulnerable populations can be tested fast and frequently, Dr. Patolia said.
The study was supported by the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose. Dr. Patolia has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
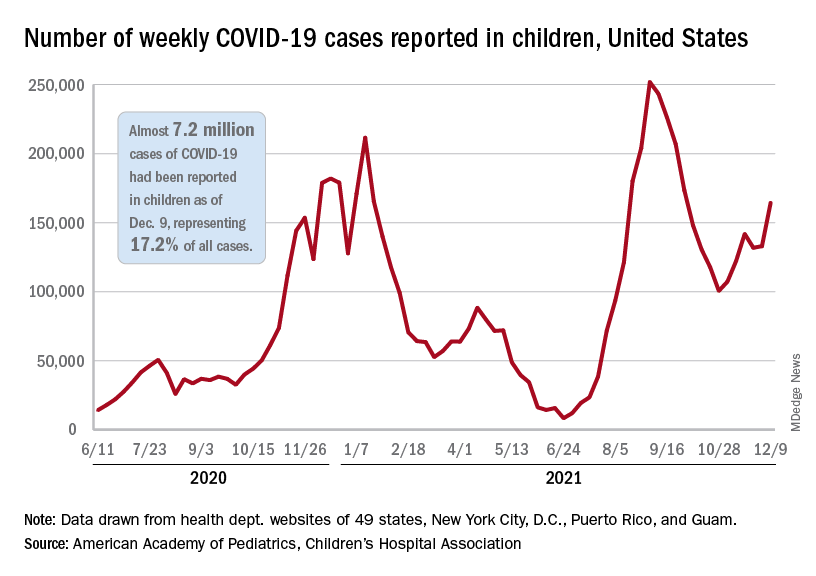
Children and COVID: Weekly cases resume their climb
After a brief lull in activity, weekly COVID-19 cases in children returned to the upward trend that began in early November, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.
according to the Centers for Disease Control and Prevention.
New COVID-19 cases were up by 23.5% for the week of Dec. 3-9, after a 2-week period that saw a drop and then just a slight increase, the AAP and CHA said in their latest weekly COVID report. There were 164,000 new cases from Dec. 3 to Dec. 9 in 46 states (Alabama, Nebraska, and Texas stopped reporting over the summer of 2021 and New York has never reported by age), the District of Columbia, New York City, Puerto Rico, and Guam.
The increase occurred across all four regions of the country, but the largest share came in the Midwest, with over 65,000 new cases, followed by the West (just over 35,000), the Northeast (just under 35,000), and the South (close to 28,000), the AAP/CHA data show.
The 7.2 million cumulative cases in children as of Dec. 9 represent 17.2% of all cases reported in the United States since the start of the pandemic, with available state reports showing that proportion ranges from 12.3% in Florida to 26.1% in Vermont. Alaska has the highest incidence of COVID at 19,000 cases per 100,000 children, and Hawaii has the lowest (5,300 per 100,000) among the states currently reporting, the AAP and CHA said.
State reporting on vaccinations shows that 37% of children aged 5-11 years in Massachusetts have received at least one dose, the highest of any state, while West Virginia is lowest at just 4%. The highest vaccination rate for children aged 12-17 goes to Massachusetts at 84%, with Wyoming lowest at 37%, the AAP said in a separate report.
Nationally, new vaccinations fell by a third during the week of Dec. 7-13, compared with the previous week, with the largest decline (34.7%) coming from the 5- to 11-year-olds, who still represented the majority (almost 84%) of the 430,000 new child vaccinations received, according to the CDC’s COVID Data Tracker. Corresponding declines for the last week were 27.5% for 12- to 15-year-olds and 22.7% for those aged 16-17.
Altogether, 21.2 million children aged 5-17 had received at least one dose and 16.0 million were fully vaccinated as of Dec. 13. By age group, 19.2% of children aged 5-11 years have gotten at least one dose and 9.6% are fully vaccinated, compared with 62.1% and 52.3%, respectively, among children aged 12-17, the CDC said.
After a brief lull in activity, weekly COVID-19 cases in children returned to the upward trend that began in early November, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

according to the Centers for Disease Control and Prevention.
New COVID-19 cases were up by 23.5% for the week of Dec. 3-9, after a 2-week period that saw a drop and then just a slight increase, the AAP and CHA said in their latest weekly COVID report. There were 164,000 new cases from Dec. 3 to Dec. 9 in 46 states (Alabama, Nebraska, and Texas stopped reporting over the summer of 2021 and New York has never reported by age), the District of Columbia, New York City, Puerto Rico, and Guam.
The increase occurred across all four regions of the country, but the largest share came in the Midwest, with over 65,000 new cases, followed by the West (just over 35,000), the Northeast (just under 35,000), and the South (close to 28,000), the AAP/CHA data show.
The 7.2 million cumulative cases in children as of Dec. 9 represent 17.2% of all cases reported in the United States since the start of the pandemic, with available state reports showing that proportion ranges from 12.3% in Florida to 26.1% in Vermont. Alaska has the highest incidence of COVID at 19,000 cases per 100,000 children, and Hawaii has the lowest (5,300 per 100,000) among the states currently reporting, the AAP and CHA said.
State reporting on vaccinations shows that 37% of children aged 5-11 years in Massachusetts have received at least one dose, the highest of any state, while West Virginia is lowest at just 4%. The highest vaccination rate for children aged 12-17 goes to Massachusetts at 84%, with Wyoming lowest at 37%, the AAP said in a separate report.
Nationally, new vaccinations fell by a third during the week of Dec. 7-13, compared with the previous week, with the largest decline (34.7%) coming from the 5- to 11-year-olds, who still represented the majority (almost 84%) of the 430,000 new child vaccinations received, according to the CDC’s COVID Data Tracker. Corresponding declines for the last week were 27.5% for 12- to 15-year-olds and 22.7% for those aged 16-17.
Altogether, 21.2 million children aged 5-17 had received at least one dose and 16.0 million were fully vaccinated as of Dec. 13. By age group, 19.2% of children aged 5-11 years have gotten at least one dose and 9.6% are fully vaccinated, compared with 62.1% and 52.3%, respectively, among children aged 12-17, the CDC said.
After a brief lull in activity, weekly COVID-19 cases in children returned to the upward trend that began in early November, based on data from the American Academy of Pediatrics and the Children’s Hospital Association.

according to the Centers for Disease Control and Prevention.
New COVID-19 cases were up by 23.5% for the week of Dec. 3-9, after a 2-week period that saw a drop and then just a slight increase, the AAP and CHA said in their latest weekly COVID report. There were 164,000 new cases from Dec. 3 to Dec. 9 in 46 states (Alabama, Nebraska, and Texas stopped reporting over the summer of 2021 and New York has never reported by age), the District of Columbia, New York City, Puerto Rico, and Guam.
The increase occurred across all four regions of the country, but the largest share came in the Midwest, with over 65,000 new cases, followed by the West (just over 35,000), the Northeast (just under 35,000), and the South (close to 28,000), the AAP/CHA data show.
The 7.2 million cumulative cases in children as of Dec. 9 represent 17.2% of all cases reported in the United States since the start of the pandemic, with available state reports showing that proportion ranges from 12.3% in Florida to 26.1% in Vermont. Alaska has the highest incidence of COVID at 19,000 cases per 100,000 children, and Hawaii has the lowest (5,300 per 100,000) among the states currently reporting, the AAP and CHA said.
State reporting on vaccinations shows that 37% of children aged 5-11 years in Massachusetts have received at least one dose, the highest of any state, while West Virginia is lowest at just 4%. The highest vaccination rate for children aged 12-17 goes to Massachusetts at 84%, with Wyoming lowest at 37%, the AAP said in a separate report.
Nationally, new vaccinations fell by a third during the week of Dec. 7-13, compared with the previous week, with the largest decline (34.7%) coming from the 5- to 11-year-olds, who still represented the majority (almost 84%) of the 430,000 new child vaccinations received, according to the CDC’s COVID Data Tracker. Corresponding declines for the last week were 27.5% for 12- to 15-year-olds and 22.7% for those aged 16-17.
Altogether, 21.2 million children aged 5-17 had received at least one dose and 16.0 million were fully vaccinated as of Dec. 13. By age group, 19.2% of children aged 5-11 years have gotten at least one dose and 9.6% are fully vaccinated, compared with 62.1% and 52.3%, respectively, among children aged 12-17, the CDC said.
Booster recommendations for pregnant women, teens, and other groups explained
These recommendations have been widened because of the continued emergence of new variants of the virus and the wane of protection over time for both vaccinations and previous disease.
The new recommendations take away some of the questions surrounding eligibility for booster vaccinations while potentially leaving some additional questions. All in all, they provide flexibility for individuals to help protect themselves against the COVID-19 virus, as many are considering celebrating the holidays with friends and family.
The first item that has become clear is that all individuals over 18 are now not only eligible for a booster vaccination a certain time after they have completed their series, but have a recommendation for one.1
But what about a fourth dose? There is a possibility that some patients should be receiving one. For those who require a three-dose series due to a condition that makes them immunocompromised, they should receive their booster vaccination six months after completion of the three-dose series. This distinction may cause confusion for some, but is important for those immunocompromised.
Boosters in women who are pregnant
The recommendations also include specific comments about individuals who are pregnant. Although initial studies did not include pregnant individuals, there has been increasing real world data that vaccination against COVID, including booster vaccinations, is safe and recommended. As pregnancy increases the risk of severe disease if infected by COVID-19, both the CDC and the American College of Obstetricians and Gynecologists,2 along with other specialty organizations, such as the Royal College of Obstetricians and Gynaecologists, recommend vaccinations for pregnant individuals.
The CDC goes on to describe that there is no evidence of vaccination increasing the risk of infertility. The vaccine protects the pregnant individual and also provides protection to the baby once born. The same is true of breastfeeding individuals.3
I hope that this information allows physicians to feel comfortable recommending vaccinations and boosters to those who are pregnant and breast feeding.
Expanded recommendations for those aged 16-17 years
Recently, the CDC also expanded booster recommendations to include those aged 16-17 years, 6 months after completing their vaccine series.
Those under 18 are currently only able to receive the Pfizer-BioNtech vaccine. This new guidance has left some parents wondering if there will also be approval for booster vaccinations soon for those aged 12-16 who are approaching or have reached six months past the initial vaccine.1
Booster brand for those over 18 years?
Although the recommendation has been simplified for all over age 18 years, there is still a decision to be made about which vaccine to use as the booster.
The recommendations allow individuals to decide which brand of vaccine they would like to have as a booster. They may choose to be vaccinated with the same vaccine they originally received or with a different vaccine. This vaccine flexibility may cause confusion, but ultimately is a good thing as it allows individuals to receive whatever vaccine is available and most convenient. This also allows individuals who have been vaccinated outside of the United States by a different brand of vaccine to also receive a booster vaccination with one of the options available here.
Take home message
Overall, the expansion of booster recommendations will help everyone avoid severe disease from COVID-19 infections. Physicians now have more clarity on who should be receiving these vaccines. Along with testing, masking, and appropriate distancing, these recommendations should help prevent severe disease and death from COVID-19.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program, also in Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
1. COVID-19 Vaccine Booster Shots. Centers for Disease Control and Prevention. 2021 Dec 9.
2. COVID-19 Vaccines and Pregnancy: Conversation Guide. American College of Obstetricians and Gynecologists. 2021 November.
3. COVID-19 Vaccines While Pregnant or Breastfeeding. Centers for Disease Control and Prevention. 2021 Dec 6.
These recommendations have been widened because of the continued emergence of new variants of the virus and the wane of protection over time for both vaccinations and previous disease.
The new recommendations take away some of the questions surrounding eligibility for booster vaccinations while potentially leaving some additional questions. All in all, they provide flexibility for individuals to help protect themselves against the COVID-19 virus, as many are considering celebrating the holidays with friends and family.
The first item that has become clear is that all individuals over 18 are now not only eligible for a booster vaccination a certain time after they have completed their series, but have a recommendation for one.1
But what about a fourth dose? There is a possibility that some patients should be receiving one. For those who require a three-dose series due to a condition that makes them immunocompromised, they should receive their booster vaccination six months after completion of the three-dose series. This distinction may cause confusion for some, but is important for those immunocompromised.
Boosters in women who are pregnant
The recommendations also include specific comments about individuals who are pregnant. Although initial studies did not include pregnant individuals, there has been increasing real world data that vaccination against COVID, including booster vaccinations, is safe and recommended. As pregnancy increases the risk of severe disease if infected by COVID-19, both the CDC and the American College of Obstetricians and Gynecologists,2 along with other specialty organizations, such as the Royal College of Obstetricians and Gynaecologists, recommend vaccinations for pregnant individuals.
The CDC goes on to describe that there is no evidence of vaccination increasing the risk of infertility. The vaccine protects the pregnant individual and also provides protection to the baby once born. The same is true of breastfeeding individuals.3
I hope that this information allows physicians to feel comfortable recommending vaccinations and boosters to those who are pregnant and breast feeding.
Expanded recommendations for those aged 16-17 years
Recently, the CDC also expanded booster recommendations to include those aged 16-17 years, 6 months after completing their vaccine series.
Those under 18 are currently only able to receive the Pfizer-BioNtech vaccine. This new guidance has left some parents wondering if there will also be approval for booster vaccinations soon for those aged 12-16 who are approaching or have reached six months past the initial vaccine.1
Booster brand for those over 18 years?
Although the recommendation has been simplified for all over age 18 years, there is still a decision to be made about which vaccine to use as the booster.
The recommendations allow individuals to decide which brand of vaccine they would like to have as a booster. They may choose to be vaccinated with the same vaccine they originally received or with a different vaccine. This vaccine flexibility may cause confusion, but ultimately is a good thing as it allows individuals to receive whatever vaccine is available and most convenient. This also allows individuals who have been vaccinated outside of the United States by a different brand of vaccine to also receive a booster vaccination with one of the options available here.
Take home message
Overall, the expansion of booster recommendations will help everyone avoid severe disease from COVID-19 infections. Physicians now have more clarity on who should be receiving these vaccines. Along with testing, masking, and appropriate distancing, these recommendations should help prevent severe disease and death from COVID-19.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program, also in Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
1. COVID-19 Vaccine Booster Shots. Centers for Disease Control and Prevention. 2021 Dec 9.
2. COVID-19 Vaccines and Pregnancy: Conversation Guide. American College of Obstetricians and Gynecologists. 2021 November.
3. COVID-19 Vaccines While Pregnant or Breastfeeding. Centers for Disease Control and Prevention. 2021 Dec 6.
These recommendations have been widened because of the continued emergence of new variants of the virus and the wane of protection over time for both vaccinations and previous disease.
The new recommendations take away some of the questions surrounding eligibility for booster vaccinations while potentially leaving some additional questions. All in all, they provide flexibility for individuals to help protect themselves against the COVID-19 virus, as many are considering celebrating the holidays with friends and family.
The first item that has become clear is that all individuals over 18 are now not only eligible for a booster vaccination a certain time after they have completed their series, but have a recommendation for one.1
But what about a fourth dose? There is a possibility that some patients should be receiving one. For those who require a three-dose series due to a condition that makes them immunocompromised, they should receive their booster vaccination six months after completion of the three-dose series. This distinction may cause confusion for some, but is important for those immunocompromised.
Boosters in women who are pregnant
The recommendations also include specific comments about individuals who are pregnant. Although initial studies did not include pregnant individuals, there has been increasing real world data that vaccination against COVID, including booster vaccinations, is safe and recommended. As pregnancy increases the risk of severe disease if infected by COVID-19, both the CDC and the American College of Obstetricians and Gynecologists,2 along with other specialty organizations, such as the Royal College of Obstetricians and Gynaecologists, recommend vaccinations for pregnant individuals.
The CDC goes on to describe that there is no evidence of vaccination increasing the risk of infertility. The vaccine protects the pregnant individual and also provides protection to the baby once born. The same is true of breastfeeding individuals.3
I hope that this information allows physicians to feel comfortable recommending vaccinations and boosters to those who are pregnant and breast feeding.
Expanded recommendations for those aged 16-17 years
Recently, the CDC also expanded booster recommendations to include those aged 16-17 years, 6 months after completing their vaccine series.
Those under 18 are currently only able to receive the Pfizer-BioNtech vaccine. This new guidance has left some parents wondering if there will also be approval for booster vaccinations soon for those aged 12-16 who are approaching or have reached six months past the initial vaccine.1
Booster brand for those over 18 years?
Although the recommendation has been simplified for all over age 18 years, there is still a decision to be made about which vaccine to use as the booster.
The recommendations allow individuals to decide which brand of vaccine they would like to have as a booster. They may choose to be vaccinated with the same vaccine they originally received or with a different vaccine. This vaccine flexibility may cause confusion, but ultimately is a good thing as it allows individuals to receive whatever vaccine is available and most convenient. This also allows individuals who have been vaccinated outside of the United States by a different brand of vaccine to also receive a booster vaccination with one of the options available here.
Take home message
Overall, the expansion of booster recommendations will help everyone avoid severe disease from COVID-19 infections. Physicians now have more clarity on who should be receiving these vaccines. Along with testing, masking, and appropriate distancing, these recommendations should help prevent severe disease and death from COVID-19.
Dr. Wheat is a family physician at Erie Family Health Center in Chicago. She is program director of Northwestern’s McGaw Family Medicine residency program, also in Chicago. Dr. Wheat serves on the editorial advisory board of Family Practice News. You can contact her at [email protected].
References
1. COVID-19 Vaccine Booster Shots. Centers for Disease Control and Prevention. 2021 Dec 9.
2. COVID-19 Vaccines and Pregnancy: Conversation Guide. American College of Obstetricians and Gynecologists. 2021 November.
3. COVID-19 Vaccines While Pregnant or Breastfeeding. Centers for Disease Control and Prevention. 2021 Dec 6.
12 state boards have disciplined docs for COVID misinformation
, according to a new survey from the Federation of State Medical Boards (FSMB).
The FSMB reports that in its 2021 annual survey two-thirds of its 71 member boards (which includes the United States, its territories, and Washington, DC) reported an increase in complaints about doctors spreading false or misleading information.
“The staggering number of state medical boards that have seen an increase in COVID-19 disinformation complaints is a sign of how widespread the issue has become,” said Humayun J. Chaudhry, DO, MACP, president and CEO of the FSMB, in a statement.
The FSMB board of directors warned physicians in July that they risked disciplinary action if they spread COVID-19 vaccine misinformation or disinformation.
The organization said 15 state boards have now adopted similar statements.
Dr. Chaudhry said the FSMB was “encouraged by the number of boards that have already taken action to combat COVID-19 disinformation by disciplining physicians who engage in that behavior and by reminding all physicians that their words and actions matter, and they should think twice before spreading disinformation that may harm patients.”
This news organization asked the FSMB for further comment on why more physicians have not been disciplined, but did not receive a response before publication.
Misinformation policies a new battleground
The FSMB and member board policies on COVID-19 around the country have become a new front in the war against mandates and restrictions.
The Tennessee Board of Medical Examiners voted just recently to remove its statement of policy against the spread of misinformation from its website after a Republican lawmaker allegedly threatened to dissolve the board.
The vote came just a few months after the board had approved the policy. The board did not rescind the policy, however, according to a report by the Associated Press.
In California, the president of the state’s medical board tweeted on December 8 about what she said was an incident of harassment by a group that has promoted “fake COVID-19 treatments.”Ms. Kristina Lawson said she observed four men sitting in front of her house in a truck. They flew a drone over her residence, and then followed her to work, parking nose-to-nose with her vehicle.
Ms. Lawson claimed that when she went to drive home the four men ambushed her in what was by then a dark parking garage. She said her “concern turned to terror” as they jumped out, cameras and recording equipment in hand.
The men told law enforcement called to the scene that they were just trying to interview her, according to a statement emailed by Ms. Lawson.
They had not made such a request to the California Medical Board.
Ms. Lawson tweeted that she would continue to volunteer for the board. “That means protecting Californians from bad doctors, and ensuring disinformation and misinformation do not detract from our work to protect patients and consumers,” she wrote.
The men who ambushed Ms. Larson allegedly identified themselves and were wearing clothing emblazoned with the logo of “America’s Frontline Doctors,” an organization that has trafficked in COVID-19 conspiracy theories and promoted unproven treatments like hydroxychloroquine and ivermectin, according to Time. It is led by Simone Gold, MD, who was arrested for breaching the U.S. Capitol on January 6.
Despite her activities, on November 30, the California Medical Board renewed Ms. Gold’s 2-year license to practice.
Who’s being disciplined, who’s not
Dr. Gold is not alone. An investigation by NPRin September found that 15 of 16 physicians who have spread false information in a high-profile manner have medical licenses in good standing.
Sherri Tenpenny, DO, who has claimed that COVID-19 vaccines magnetize people and “interface” with 5G cell phone towers, was able to renew her license with the Ohio State Medical Board on October 1, according to the Cincinnati Enquirer.
Some boards have acted. The Oregon Medical Board revoked the license of Steven LaTulippe, MD, and fined him $10,000 for spreading misinformation about masks and overprescribing opioids.
In August, Rhode Island’s Board of Medical Licensure suspended Mark Brody’s license for 5 years after finding that the doctor spread falsehoods about COVID-19 vaccines, according to board documents.
Maine physician Paul Gosselin, DO, is on temporary suspension until a February hearing, while the osteopathic board investigates his issuance of vaccine exemption letters and the promotion of unproven COVID-19 therapies.
The board found that Gosselin had “engaged in conduct that constitutes fraud or deceit,” according to official documents.
The Washington State Medical Board has opened an investigation into Ryan N. Cole, MD, a physician who has claimed that COVID vaccines are “fake,” and was appointed to a regional health board in Idaho in September, according to the Washington Post.
The Idaho Capital Sun reported that Dr. Cole claims he is licensed in 11 states, including Washington. The Idaho Medical Association has also filed a complaint about Dr. Cole with the Idaho Board of Medicine, according to the paper.
New FSMB guidance coming
The FSMB said it expects more disciplinary actions as investigations continue to unfold.
The organization is drafting a new policy document that will include further guidelines and recommendations for state medical boards “to help address the spread of disinformation,” it said. The final document would be released in April 2022.
In the meantime, some states, like Tennessee and others, are trying to find ways to counter the current policy — a development the FSMB called “troubling.”
“The FSMB strongly opposes any effort to restrict a board’s authority to evaluate the standard of care and assess risk for patient harm,” the organization said in its statement.
A version of this article was first published on Medscape.com.
, according to a new survey from the Federation of State Medical Boards (FSMB).
The FSMB reports that in its 2021 annual survey two-thirds of its 71 member boards (which includes the United States, its territories, and Washington, DC) reported an increase in complaints about doctors spreading false or misleading information.
“The staggering number of state medical boards that have seen an increase in COVID-19 disinformation complaints is a sign of how widespread the issue has become,” said Humayun J. Chaudhry, DO, MACP, president and CEO of the FSMB, in a statement.
The FSMB board of directors warned physicians in July that they risked disciplinary action if they spread COVID-19 vaccine misinformation or disinformation.
The organization said 15 state boards have now adopted similar statements.
Dr. Chaudhry said the FSMB was “encouraged by the number of boards that have already taken action to combat COVID-19 disinformation by disciplining physicians who engage in that behavior and by reminding all physicians that their words and actions matter, and they should think twice before spreading disinformation that may harm patients.”
This news organization asked the FSMB for further comment on why more physicians have not been disciplined, but did not receive a response before publication.
Misinformation policies a new battleground
The FSMB and member board policies on COVID-19 around the country have become a new front in the war against mandates and restrictions.
The Tennessee Board of Medical Examiners voted just recently to remove its statement of policy against the spread of misinformation from its website after a Republican lawmaker allegedly threatened to dissolve the board.
The vote came just a few months after the board had approved the policy. The board did not rescind the policy, however, according to a report by the Associated Press.
In California, the president of the state’s medical board tweeted on December 8 about what she said was an incident of harassment by a group that has promoted “fake COVID-19 treatments.”Ms. Kristina Lawson said she observed four men sitting in front of her house in a truck. They flew a drone over her residence, and then followed her to work, parking nose-to-nose with her vehicle.
Ms. Lawson claimed that when she went to drive home the four men ambushed her in what was by then a dark parking garage. She said her “concern turned to terror” as they jumped out, cameras and recording equipment in hand.
The men told law enforcement called to the scene that they were just trying to interview her, according to a statement emailed by Ms. Lawson.
They had not made such a request to the California Medical Board.
Ms. Lawson tweeted that she would continue to volunteer for the board. “That means protecting Californians from bad doctors, and ensuring disinformation and misinformation do not detract from our work to protect patients and consumers,” she wrote.
The men who ambushed Ms. Larson allegedly identified themselves and were wearing clothing emblazoned with the logo of “America’s Frontline Doctors,” an organization that has trafficked in COVID-19 conspiracy theories and promoted unproven treatments like hydroxychloroquine and ivermectin, according to Time. It is led by Simone Gold, MD, who was arrested for breaching the U.S. Capitol on January 6.
Despite her activities, on November 30, the California Medical Board renewed Ms. Gold’s 2-year license to practice.
Who’s being disciplined, who’s not
Dr. Gold is not alone. An investigation by NPRin September found that 15 of 16 physicians who have spread false information in a high-profile manner have medical licenses in good standing.
Sherri Tenpenny, DO, who has claimed that COVID-19 vaccines magnetize people and “interface” with 5G cell phone towers, was able to renew her license with the Ohio State Medical Board on October 1, according to the Cincinnati Enquirer.
Some boards have acted. The Oregon Medical Board revoked the license of Steven LaTulippe, MD, and fined him $10,000 for spreading misinformation about masks and overprescribing opioids.
In August, Rhode Island’s Board of Medical Licensure suspended Mark Brody’s license for 5 years after finding that the doctor spread falsehoods about COVID-19 vaccines, according to board documents.
Maine physician Paul Gosselin, DO, is on temporary suspension until a February hearing, while the osteopathic board investigates his issuance of vaccine exemption letters and the promotion of unproven COVID-19 therapies.
The board found that Gosselin had “engaged in conduct that constitutes fraud or deceit,” according to official documents.
The Washington State Medical Board has opened an investigation into Ryan N. Cole, MD, a physician who has claimed that COVID vaccines are “fake,” and was appointed to a regional health board in Idaho in September, according to the Washington Post.
The Idaho Capital Sun reported that Dr. Cole claims he is licensed in 11 states, including Washington. The Idaho Medical Association has also filed a complaint about Dr. Cole with the Idaho Board of Medicine, according to the paper.
New FSMB guidance coming
The FSMB said it expects more disciplinary actions as investigations continue to unfold.
The organization is drafting a new policy document that will include further guidelines and recommendations for state medical boards “to help address the spread of disinformation,” it said. The final document would be released in April 2022.
In the meantime, some states, like Tennessee and others, are trying to find ways to counter the current policy — a development the FSMB called “troubling.”
“The FSMB strongly opposes any effort to restrict a board’s authority to evaluate the standard of care and assess risk for patient harm,” the organization said in its statement.
A version of this article was first published on Medscape.com.
, according to a new survey from the Federation of State Medical Boards (FSMB).
The FSMB reports that in its 2021 annual survey two-thirds of its 71 member boards (which includes the United States, its territories, and Washington, DC) reported an increase in complaints about doctors spreading false or misleading information.
“The staggering number of state medical boards that have seen an increase in COVID-19 disinformation complaints is a sign of how widespread the issue has become,” said Humayun J. Chaudhry, DO, MACP, president and CEO of the FSMB, in a statement.
The FSMB board of directors warned physicians in July that they risked disciplinary action if they spread COVID-19 vaccine misinformation or disinformation.
The organization said 15 state boards have now adopted similar statements.
Dr. Chaudhry said the FSMB was “encouraged by the number of boards that have already taken action to combat COVID-19 disinformation by disciplining physicians who engage in that behavior and by reminding all physicians that their words and actions matter, and they should think twice before spreading disinformation that may harm patients.”
This news organization asked the FSMB for further comment on why more physicians have not been disciplined, but did not receive a response before publication.
Misinformation policies a new battleground
The FSMB and member board policies on COVID-19 around the country have become a new front in the war against mandates and restrictions.
The Tennessee Board of Medical Examiners voted just recently to remove its statement of policy against the spread of misinformation from its website after a Republican lawmaker allegedly threatened to dissolve the board.
The vote came just a few months after the board had approved the policy. The board did not rescind the policy, however, according to a report by the Associated Press.
In California, the president of the state’s medical board tweeted on December 8 about what she said was an incident of harassment by a group that has promoted “fake COVID-19 treatments.”Ms. Kristina Lawson said she observed four men sitting in front of her house in a truck. They flew a drone over her residence, and then followed her to work, parking nose-to-nose with her vehicle.
Ms. Lawson claimed that when she went to drive home the four men ambushed her in what was by then a dark parking garage. She said her “concern turned to terror” as they jumped out, cameras and recording equipment in hand.
The men told law enforcement called to the scene that they were just trying to interview her, according to a statement emailed by Ms. Lawson.
They had not made such a request to the California Medical Board.
Ms. Lawson tweeted that she would continue to volunteer for the board. “That means protecting Californians from bad doctors, and ensuring disinformation and misinformation do not detract from our work to protect patients and consumers,” she wrote.
The men who ambushed Ms. Larson allegedly identified themselves and were wearing clothing emblazoned with the logo of “America’s Frontline Doctors,” an organization that has trafficked in COVID-19 conspiracy theories and promoted unproven treatments like hydroxychloroquine and ivermectin, according to Time. It is led by Simone Gold, MD, who was arrested for breaching the U.S. Capitol on January 6.
Despite her activities, on November 30, the California Medical Board renewed Ms. Gold’s 2-year license to practice.
Who’s being disciplined, who’s not
Dr. Gold is not alone. An investigation by NPRin September found that 15 of 16 physicians who have spread false information in a high-profile manner have medical licenses in good standing.
Sherri Tenpenny, DO, who has claimed that COVID-19 vaccines magnetize people and “interface” with 5G cell phone towers, was able to renew her license with the Ohio State Medical Board on October 1, according to the Cincinnati Enquirer.
Some boards have acted. The Oregon Medical Board revoked the license of Steven LaTulippe, MD, and fined him $10,000 for spreading misinformation about masks and overprescribing opioids.
In August, Rhode Island’s Board of Medical Licensure suspended Mark Brody’s license for 5 years after finding that the doctor spread falsehoods about COVID-19 vaccines, according to board documents.
Maine physician Paul Gosselin, DO, is on temporary suspension until a February hearing, while the osteopathic board investigates his issuance of vaccine exemption letters and the promotion of unproven COVID-19 therapies.
The board found that Gosselin had “engaged in conduct that constitutes fraud or deceit,” according to official documents.
The Washington State Medical Board has opened an investigation into Ryan N. Cole, MD, a physician who has claimed that COVID vaccines are “fake,” and was appointed to a regional health board in Idaho in September, according to the Washington Post.
The Idaho Capital Sun reported that Dr. Cole claims he is licensed in 11 states, including Washington. The Idaho Medical Association has also filed a complaint about Dr. Cole with the Idaho Board of Medicine, according to the paper.
New FSMB guidance coming
The FSMB said it expects more disciplinary actions as investigations continue to unfold.
The organization is drafting a new policy document that will include further guidelines and recommendations for state medical boards “to help address the spread of disinformation,” it said. The final document would be released in April 2022.
In the meantime, some states, like Tennessee and others, are trying to find ways to counter the current policy — a development the FSMB called “troubling.”
“The FSMB strongly opposes any effort to restrict a board’s authority to evaluate the standard of care and assess risk for patient harm,” the organization said in its statement.
A version of this article was first published on Medscape.com.
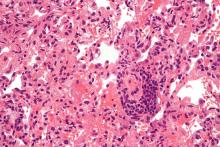
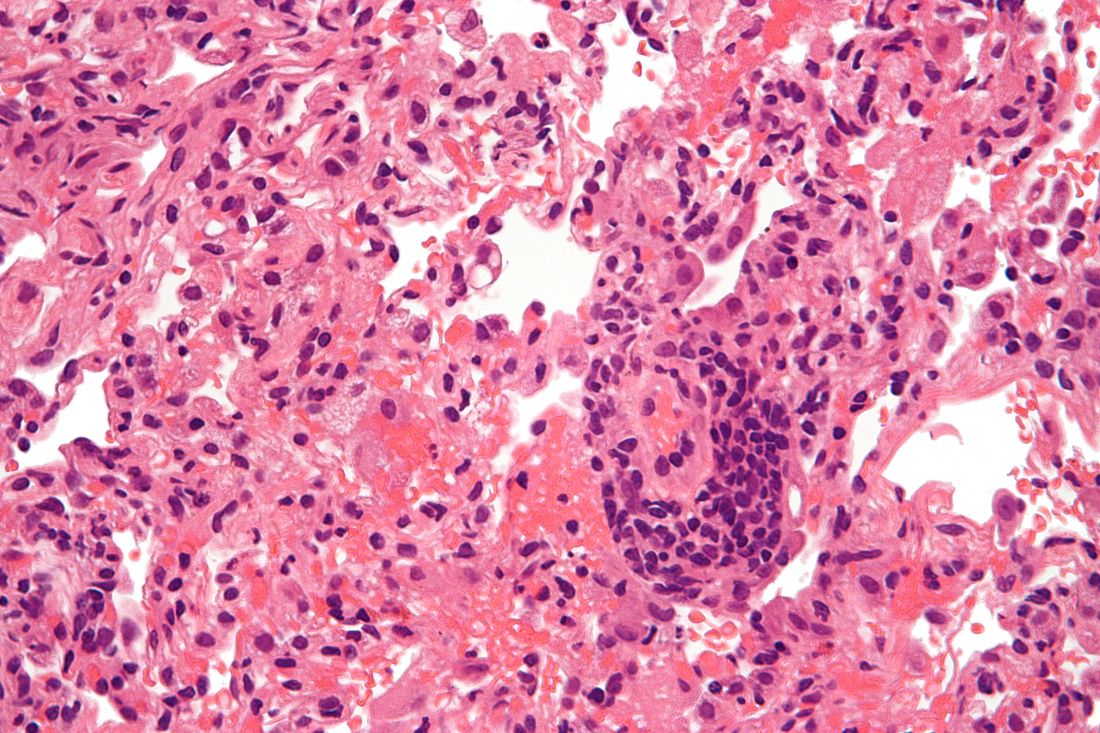
Lung transplantation in the era of COVID-19: New issues and paradigms
Data is sparse thus far, but there is concern in lung transplant medicine about the long-term risk of chronic lung allograft dysfunction (CLAD) and a potentially shortened longevity of transplanted lungs in recipients who become ill with COVID-19.
“My fear is that we’re potentially sitting on this iceberg worth of people who, come 6 months or a year from [the acute phase of] their COVID illness, will in fact have earlier and progressive, chronic rejection,” said Cameron R. Wolfe, MBBS, MPH, associate professor of medicine in transplant infectious disease at Duke University, Durham, N.C.
Lower respiratory viral infections have long been concerning for lung transplant recipients given their propensity to cause scarring, a decline in lung function, and a heightened risk of allograft rejection. Time will tell whether lung transplant recipients who survive COVID-19 follow a similar path, or one that is worse, he said.
Short-term data
Outcomes beyond hospitalization and acute illness for lung transplant recipients affected by COVID-19 have been reported in the literature by only a few lung transplant programs. These reports – as well as anecdotal experiences being informally shared among transplant programs – have raised the specter of more severe dysfunction following the acute phase and more early CLAD, said Tathagat Narula, MD, assistant professor of medicine at the Mayo Medical School, Rochester, Minn., and a consultant in lung transplantation at the Mayo Clinic’s Jacksonville program.
“The available data cover only 3-6 months out. We don’t know what will happen in the next 6 months and beyond,” Dr. Narula said in an interview.
The risks of COVID-19 in already-transplanted patients and issues relating to the inadequate antibody responses to vaccination are just some of the challenges of lung transplant medicine in the era of SARS-CoV-2. “COVID-19,” said Dr. Narula, “has completely changed the way we practice lung transplant medicine – the way we’re looking both at our recipients and our donors.”
Potential donors are being evaluated with lower respiratory SARS-CoV-2 testing and an abundance of caution. And patients with severe COVID-19 affecting their own lungs are roundly expected to drive up lung transplant volume in the near future. “The whole paradigm has changed,” Dr. Narula said.
Post-acute trajectories
A chart review study published in October by the lung transplant team at the University of Texas Southwestern Medical Center, Dallas, covered 44 consecutive survivors at a median follow-up of 4.5 months from hospital discharge or acute illness (the survival rate was 83.3%). Patients had significantly impaired functional status, and 18 of the 44 (40.9%) had a significant and persistent loss of forced vital capacity or forced expiratory volume in 1 second (>10% from pre–COVID-19 baseline).
Three patients met the criteria for new CLAD after COVID-19 infection, with all three classified as restrictive allograft syndrome (RAS) phenotype.
Moreover, the majority of COVID-19 survivors who had CT chest scans (22 of 28) showed persistent parenchymal opacities – a finding that, regardless of symptomatology, suggests persistent allograft injury, said Amit Banga, MD, associate professor of medicine and medical director of the ex vivo lung perfusion program in UT Southwestern’s lung transplant program.
“The implication is that there may be long-term consequences of COVID-19, perhaps related to some degree of ongoing inflammation and damage,” said Dr. Banga, a coauthor of the postinfection outcomes paper.
The UT Southwestern lung transplant program, which normally performs 60-80 transplants a year, began routine CT scanning 4-5 months into the pandemic, after “stumbling into a few patients who had no symptoms indicative of COVID pneumonia and no changes on an x-ray but significant involvement on a CT,” he said.
Without routine scanning in the general population of COVID-19 patients, Dr. Banga noted, “we’re limited in convincingly saying that COVID is uniquely doing this to lung transplant recipients.” Nor can they conclude that SARS-CoV-2 is unique from other respiratory viruses such as respiratory syncytial virus (RSV) in this regard. (The program has added CT scanning to its protocol for lung transplant recipients afflicted with other respiratory viruses to learn more.)
However, in the big picture, COVID-19 has proven to be far worse for lung transplant recipients than illness with other respiratory viruses, including RSV. “Patients have more frequent and greater loss of lung function, and worse debility from the acute illness,” Dr. Banga said.
“The cornerstones of treatment of both these viruses are very similar, but both the in-hospital course and the postdischarge outcomes are significantly different.”
In an initial paper published in September 2021, Dr. Banga and colleagues compared their first 25 lung transplant patients testing positive for SARS-CoV-2 with a historical cohort of 36 patients with RSV treated during 2016-2018.
Patients with COVID-19 had significantly worse morbidity and mortality, including worse postinfection lung function loss, functional decline, and 3-month survival.
More time, he said, will shed light on the risks of CLAD and the long-term potential for recovery of lung function. Currently, at UT Southwestern, it appears that patients who survive acute illness and the “first 3-6 months after COVID-19, when we’re seeing all the postinfection morbidity, may [enter] a period of stability,” Dr. Banga said.
Overall, he said, patients in their initial cohort are “holding steady” without unusual morbidity, readmissions, or “other setbacks to their allografts.”
At the Mayo Clinic in Jacksonville, which normally performs 40-50 lung transplants a year, transplant physicians have similarly observed significant declines in lung function beyond the acute phase of COVID-19. “Anecdotally, we’re seeing that some patients are beginning to recover some of their lung function, while others have not,” said Dr. Narula. “And we don’t have predictors as to who will progress to CLAD. It’s a big knowledge gap.”
Dr. Narula noted that patients with restrictive allograft syndrome, such as those reported by the UT Southwestern team, “have scarring of the lung and a much worse prognosis than the obstructive type of chronic rejection.” Whether there’s a role for antifibrotic therapy is a question worthy of research.
In UT Southwestern’s analysis, persistently lower absolute lymphocyte counts (< 600/dL) and higher ferritin levels (>150 ng/mL) at the time of hospital discharge were independently associated with significant lung function loss. This finding, reported in their October paper, has helped guide their management practices, Dr. Banga said.
“Persistently elevated ferritin may indicate ongoing inflammation at the allograft level,” he said. “We now send [such patients] home on a longer course of oral corticosteroids.”
At the front end of care for infected lung transplant recipients, Dr. Banga said that his team and physicians at other lung transplant programs are holding the cell-cycle inhibitor component of patients’ maintenance immunosuppression therapy (commonly mycophenolate or azathioprine) once infection is diagnosed to maximize chances of a better outcome.
“There may be variation on how long [the regimens are adjusted],” he said. “We changed our duration from 4 weeks to 2 due to patients developing a rebound worsening in the third and fourth week of acute illness.”
There is significant variation from institution to institution in how viral infections are managed in lung transplant recipients, he and Dr. Narula said. “Our numbers are so small in lung transplant, and we don’t have standardized protocols – it’s one of the biggest challenges in our field,” said Dr. Narula.
Vaccination issues, evaluation of donors
Whether or not immunosuppression regimens should be adjusted prior to vaccination is a controversial question, but is “an absolutely valid one” and is currently being studied in at least one National Institutes of Health–funded trial involving solid organ transplant recipients, said Dr. Wolfe.
“Some have jumped to the conclusion [based on some earlier data] that they should reduce immunosuppression regimens for everyone at the time of vaccination ... but I don’t know the answer yet,” he said. “Balancing staying rejection free with potentially gaining more immune response is complicated ... and it may depend on where the pandemic is going in your area and other factors.”
Reductions aside, Dr. Wolfe tells lung transplant recipients that, based on his approximation of a number of different studies in solid organ transplant recipients, approximately 40%-50% of patients who are immunized with two doses of the COVID-19 mRNA vaccines will develop meaningful antibody levels – and that this rises to 50%-60% after a third dose.
It is difficult to glean from available studies the level of vaccine response for lung transplant recipients specifically. But given that their level of maintenance immunosuppression is higher than for recipients of other donor organs, “as a broad sweep, lung transplant recipients tend to be lower in the pecking order of response,” he said.
Still, “there’s a lot to gain,” he said, pointing to a recent study from the Morbidity and Mortality Weekly Report (2021 Nov 5. doi: 10.15585/mmwr.mm7044e3) showing that effectiveness of mRNA vaccination against COVID-19–associated hospitalization was 77% among immunocompromised adults (compared with 90% in immunocompetent adults).
“This is good vindication to keep vaccinating,” he said, “and perhaps speaks to how difficult it is to assess the vaccine response [through measurement of antibody only].”
Neither Duke University’s transplant program, which performed 100-120 lung transplants a year pre-COVID, nor the programs at UT Southwestern or the Mayo Clinic in Jacksonville require that solid organ transplant candidates be vaccinated against SARS-CoV-2 in order to receive transplants, as some other transplant programs have done. (When asked about the issue, Dr. Banga and Dr. Narula each said that they have had no or little trouble convincing patients awaiting lung transplants of the need for COVID-19 vaccination.)
In an August statement, the American Society of Transplantation recommended vaccination for all solid organ transplant recipients, preferably prior to transplantation, and said that it “support[s] the development of institutional policies regarding pretransplant vaccination.”
The Society is not tracking centers’ vaccination policies. But Kaiser Health News reported in October that a growing number of transplant programs, such as UCHealth in Denver and UW Medicine in Seattle, have decided to either bar patients who refuse to be vaccinated from receiving transplants or give them lower priority on waitlists.
Potential lung donors, meanwhile, must be evaluated with lower respiratory COVID-19 testing, with results available prior to transplantation, according to policy developed by the Organ Procurement and Transplantation Network and effective in May 2021. The policy followed three published cases of donor-derived COVID-19 in lung transplant recipients, said Dr. Wolfe, who wrote about use of COVID-positive donors in an editorial published in October.
In each case, the donor had a negative COVID-19 nasopharyngeal swab at the time of organ procurement but was later found to have the virus on bronchoalveolar lavage, he said.
(The use of other organs from COVID-positive donors is appearing thus far to be safe, Dr. Wolfe noted. In the editorial, he references 13 cases of solid organ transplantation from SARS-CoV-2–infected donors into noninfected recipients; none of the 13 transplant recipients developed COVID-19).
Some questions remain, such as how many lower respiratory tests should be run, and how donors should be evaluated in cases of discordant results. Dr. Banga shared the case of a donor with one positive lower respiratory test result followed by two negative results. After internal debate, and consideration of potential false positives and other issues, the team at UT Southwestern decided to decline the donor, Dr. Banga said.
Other programs are likely making similar, appropriately cautious decisions, said Dr. Wolfe. “There’s no way in real-time donor evaluation to know whether the positive test is active virus that could infect the recipient and replicate ... or whether it’s [picking up] inactive or dead fragments of virus that was there several weeks ago. Our tests don’t differentiate that.”
Transplants in COVID-19 patients
Decision-making about lung transplant candidacy among patients with COVID-19 acute respiratory distress syndrome is complex and in need of a new paradigm.
“Some of these patients have the potential to recover, and they’re going to recover way later than what we’re used to,” said Dr. Banga. “We can’t extrapolate for COVID ARDS what we’ve learned for any other virus-related ARDS.”
Dr. Narula also has recently seen at least one COVID-19 patient on ECMO and under evaluation for transplantation recover. “We do not want to transplant too early,” he said, noting that there is consensus that lung transplant should be pursued only when the damage is deemed irreversible clinically and radiologically in the best judgment of the team. Still, “for many of these patients the only exit route will be lung transplants. For the next 12-24 months, a significant proportion of our lung transplant patients will have had post-COVID–related lung damage.”
As of October 2021, 233 lung transplants had been performed in the United States in recipients whose primary diagnosis was reported as COVID related, said Anne Paschke, media relations specialist with the United Network for Organ Sharing.
Dr. Banga, Dr. Wolfe, and Dr. Narula reported that they have no relevant disclosures.
Data is sparse thus far, but there is concern in lung transplant medicine about the long-term risk of chronic lung allograft dysfunction (CLAD) and a potentially shortened longevity of transplanted lungs in recipients who become ill with COVID-19.
“My fear is that we’re potentially sitting on this iceberg worth of people who, come 6 months or a year from [the acute phase of] their COVID illness, will in fact have earlier and progressive, chronic rejection,” said Cameron R. Wolfe, MBBS, MPH, associate professor of medicine in transplant infectious disease at Duke University, Durham, N.C.
Lower respiratory viral infections have long been concerning for lung transplant recipients given their propensity to cause scarring, a decline in lung function, and a heightened risk of allograft rejection. Time will tell whether lung transplant recipients who survive COVID-19 follow a similar path, or one that is worse, he said.
Short-term data
Outcomes beyond hospitalization and acute illness for lung transplant recipients affected by COVID-19 have been reported in the literature by only a few lung transplant programs. These reports – as well as anecdotal experiences being informally shared among transplant programs – have raised the specter of more severe dysfunction following the acute phase and more early CLAD, said Tathagat Narula, MD, assistant professor of medicine at the Mayo Medical School, Rochester, Minn., and a consultant in lung transplantation at the Mayo Clinic’s Jacksonville program.
“The available data cover only 3-6 months out. We don’t know what will happen in the next 6 months and beyond,” Dr. Narula said in an interview.
The risks of COVID-19 in already-transplanted patients and issues relating to the inadequate antibody responses to vaccination are just some of the challenges of lung transplant medicine in the era of SARS-CoV-2. “COVID-19,” said Dr. Narula, “has completely changed the way we practice lung transplant medicine – the way we’re looking both at our recipients and our donors.”
Potential donors are being evaluated with lower respiratory SARS-CoV-2 testing and an abundance of caution. And patients with severe COVID-19 affecting their own lungs are roundly expected to drive up lung transplant volume in the near future. “The whole paradigm has changed,” Dr. Narula said.
Post-acute trajectories
A chart review study published in October by the lung transplant team at the University of Texas Southwestern Medical Center, Dallas, covered 44 consecutive survivors at a median follow-up of 4.5 months from hospital discharge or acute illness (the survival rate was 83.3%). Patients had significantly impaired functional status, and 18 of the 44 (40.9%) had a significant and persistent loss of forced vital capacity or forced expiratory volume in 1 second (>10% from pre–COVID-19 baseline).
Three patients met the criteria for new CLAD after COVID-19 infection, with all three classified as restrictive allograft syndrome (RAS) phenotype.
Moreover, the majority of COVID-19 survivors who had CT chest scans (22 of 28) showed persistent parenchymal opacities – a finding that, regardless of symptomatology, suggests persistent allograft injury, said Amit Banga, MD, associate professor of medicine and medical director of the ex vivo lung perfusion program in UT Southwestern’s lung transplant program.
“The implication is that there may be long-term consequences of COVID-19, perhaps related to some degree of ongoing inflammation and damage,” said Dr. Banga, a coauthor of the postinfection outcomes paper.
The UT Southwestern lung transplant program, which normally performs 60-80 transplants a year, began routine CT scanning 4-5 months into the pandemic, after “stumbling into a few patients who had no symptoms indicative of COVID pneumonia and no changes on an x-ray but significant involvement on a CT,” he said.
Without routine scanning in the general population of COVID-19 patients, Dr. Banga noted, “we’re limited in convincingly saying that COVID is uniquely doing this to lung transplant recipients.” Nor can they conclude that SARS-CoV-2 is unique from other respiratory viruses such as respiratory syncytial virus (RSV) in this regard. (The program has added CT scanning to its protocol for lung transplant recipients afflicted with other respiratory viruses to learn more.)
However, in the big picture, COVID-19 has proven to be far worse for lung transplant recipients than illness with other respiratory viruses, including RSV. “Patients have more frequent and greater loss of lung function, and worse debility from the acute illness,” Dr. Banga said.
“The cornerstones of treatment of both these viruses are very similar, but both the in-hospital course and the postdischarge outcomes are significantly different.”
In an initial paper published in September 2021, Dr. Banga and colleagues compared their first 25 lung transplant patients testing positive for SARS-CoV-2 with a historical cohort of 36 patients with RSV treated during 2016-2018.
Patients with COVID-19 had significantly worse morbidity and mortality, including worse postinfection lung function loss, functional decline, and 3-month survival.
More time, he said, will shed light on the risks of CLAD and the long-term potential for recovery of lung function. Currently, at UT Southwestern, it appears that patients who survive acute illness and the “first 3-6 months after COVID-19, when we’re seeing all the postinfection morbidity, may [enter] a period of stability,” Dr. Banga said.
Overall, he said, patients in their initial cohort are “holding steady” without unusual morbidity, readmissions, or “other setbacks to their allografts.”
At the Mayo Clinic in Jacksonville, which normally performs 40-50 lung transplants a year, transplant physicians have similarly observed significant declines in lung function beyond the acute phase of COVID-19. “Anecdotally, we’re seeing that some patients are beginning to recover some of their lung function, while others have not,” said Dr. Narula. “And we don’t have predictors as to who will progress to CLAD. It’s a big knowledge gap.”
Dr. Narula noted that patients with restrictive allograft syndrome, such as those reported by the UT Southwestern team, “have scarring of the lung and a much worse prognosis than the obstructive type of chronic rejection.” Whether there’s a role for antifibrotic therapy is a question worthy of research.
In UT Southwestern’s analysis, persistently lower absolute lymphocyte counts (< 600/dL) and higher ferritin levels (>150 ng/mL) at the time of hospital discharge were independently associated with significant lung function loss. This finding, reported in their October paper, has helped guide their management practices, Dr. Banga said.
“Persistently elevated ferritin may indicate ongoing inflammation at the allograft level,” he said. “We now send [such patients] home on a longer course of oral corticosteroids.”
At the front end of care for infected lung transplant recipients, Dr. Banga said that his team and physicians at other lung transplant programs are holding the cell-cycle inhibitor component of patients’ maintenance immunosuppression therapy (commonly mycophenolate or azathioprine) once infection is diagnosed to maximize chances of a better outcome.
“There may be variation on how long [the regimens are adjusted],” he said. “We changed our duration from 4 weeks to 2 due to patients developing a rebound worsening in the third and fourth week of acute illness.”
There is significant variation from institution to institution in how viral infections are managed in lung transplant recipients, he and Dr. Narula said. “Our numbers are so small in lung transplant, and we don’t have standardized protocols – it’s one of the biggest challenges in our field,” said Dr. Narula.
Vaccination issues, evaluation of donors
Whether or not immunosuppression regimens should be adjusted prior to vaccination is a controversial question, but is “an absolutely valid one” and is currently being studied in at least one National Institutes of Health–funded trial involving solid organ transplant recipients, said Dr. Wolfe.
“Some have jumped to the conclusion [based on some earlier data] that they should reduce immunosuppression regimens for everyone at the time of vaccination ... but I don’t know the answer yet,” he said. “Balancing staying rejection free with potentially gaining more immune response is complicated ... and it may depend on where the pandemic is going in your area and other factors.”
Reductions aside, Dr. Wolfe tells lung transplant recipients that, based on his approximation of a number of different studies in solid organ transplant recipients, approximately 40%-50% of patients who are immunized with two doses of the COVID-19 mRNA vaccines will develop meaningful antibody levels – and that this rises to 50%-60% after a third dose.
It is difficult to glean from available studies the level of vaccine response for lung transplant recipients specifically. But given that their level of maintenance immunosuppression is higher than for recipients of other donor organs, “as a broad sweep, lung transplant recipients tend to be lower in the pecking order of response,” he said.
Still, “there’s a lot to gain,” he said, pointing to a recent study from the Morbidity and Mortality Weekly Report (2021 Nov 5. doi: 10.15585/mmwr.mm7044e3) showing that effectiveness of mRNA vaccination against COVID-19–associated hospitalization was 77% among immunocompromised adults (compared with 90% in immunocompetent adults).
“This is good vindication to keep vaccinating,” he said, “and perhaps speaks to how difficult it is to assess the vaccine response [through measurement of antibody only].”
Neither Duke University’s transplant program, which performed 100-120 lung transplants a year pre-COVID, nor the programs at UT Southwestern or the Mayo Clinic in Jacksonville require that solid organ transplant candidates be vaccinated against SARS-CoV-2 in order to receive transplants, as some other transplant programs have done. (When asked about the issue, Dr. Banga and Dr. Narula each said that they have had no or little trouble convincing patients awaiting lung transplants of the need for COVID-19 vaccination.)
In an August statement, the American Society of Transplantation recommended vaccination for all solid organ transplant recipients, preferably prior to transplantation, and said that it “support[s] the development of institutional policies regarding pretransplant vaccination.”
The Society is not tracking centers’ vaccination policies. But Kaiser Health News reported in October that a growing number of transplant programs, such as UCHealth in Denver and UW Medicine in Seattle, have decided to either bar patients who refuse to be vaccinated from receiving transplants or give them lower priority on waitlists.
Potential lung donors, meanwhile, must be evaluated with lower respiratory COVID-19 testing, with results available prior to transplantation, according to policy developed by the Organ Procurement and Transplantation Network and effective in May 2021. The policy followed three published cases of donor-derived COVID-19 in lung transplant recipients, said Dr. Wolfe, who wrote about use of COVID-positive donors in an editorial published in October.
In each case, the donor had a negative COVID-19 nasopharyngeal swab at the time of organ procurement but was later found to have the virus on bronchoalveolar lavage, he said.
(The use of other organs from COVID-positive donors is appearing thus far to be safe, Dr. Wolfe noted. In the editorial, he references 13 cases of solid organ transplantation from SARS-CoV-2–infected donors into noninfected recipients; none of the 13 transplant recipients developed COVID-19).
Some questions remain, such as how many lower respiratory tests should be run, and how donors should be evaluated in cases of discordant results. Dr. Banga shared the case of a donor with one positive lower respiratory test result followed by two negative results. After internal debate, and consideration of potential false positives and other issues, the team at UT Southwestern decided to decline the donor, Dr. Banga said.
Other programs are likely making similar, appropriately cautious decisions, said Dr. Wolfe. “There’s no way in real-time donor evaluation to know whether the positive test is active virus that could infect the recipient and replicate ... or whether it’s [picking up] inactive or dead fragments of virus that was there several weeks ago. Our tests don’t differentiate that.”
Transplants in COVID-19 patients
Decision-making about lung transplant candidacy among patients with COVID-19 acute respiratory distress syndrome is complex and in need of a new paradigm.
“Some of these patients have the potential to recover, and they’re going to recover way later than what we’re used to,” said Dr. Banga. “We can’t extrapolate for COVID ARDS what we’ve learned for any other virus-related ARDS.”
Dr. Narula also has recently seen at least one COVID-19 patient on ECMO and under evaluation for transplantation recover. “We do not want to transplant too early,” he said, noting that there is consensus that lung transplant should be pursued only when the damage is deemed irreversible clinically and radiologically in the best judgment of the team. Still, “for many of these patients the only exit route will be lung transplants. For the next 12-24 months, a significant proportion of our lung transplant patients will have had post-COVID–related lung damage.”
As of October 2021, 233 lung transplants had been performed in the United States in recipients whose primary diagnosis was reported as COVID related, said Anne Paschke, media relations specialist with the United Network for Organ Sharing.
Dr. Banga, Dr. Wolfe, and Dr. Narula reported that they have no relevant disclosures.
Data is sparse thus far, but there is concern in lung transplant medicine about the long-term risk of chronic lung allograft dysfunction (CLAD) and a potentially shortened longevity of transplanted lungs in recipients who become ill with COVID-19.
“My fear is that we’re potentially sitting on this iceberg worth of people who, come 6 months or a year from [the acute phase of] their COVID illness, will in fact have earlier and progressive, chronic rejection,” said Cameron R. Wolfe, MBBS, MPH, associate professor of medicine in transplant infectious disease at Duke University, Durham, N.C.
Lower respiratory viral infections have long been concerning for lung transplant recipients given their propensity to cause scarring, a decline in lung function, and a heightened risk of allograft rejection. Time will tell whether lung transplant recipients who survive COVID-19 follow a similar path, or one that is worse, he said.
Short-term data
Outcomes beyond hospitalization and acute illness for lung transplant recipients affected by COVID-19 have been reported in the literature by only a few lung transplant programs. These reports – as well as anecdotal experiences being informally shared among transplant programs – have raised the specter of more severe dysfunction following the acute phase and more early CLAD, said Tathagat Narula, MD, assistant professor of medicine at the Mayo Medical School, Rochester, Minn., and a consultant in lung transplantation at the Mayo Clinic’s Jacksonville program.
“The available data cover only 3-6 months out. We don’t know what will happen in the next 6 months and beyond,” Dr. Narula said in an interview.
The risks of COVID-19 in already-transplanted patients and issues relating to the inadequate antibody responses to vaccination are just some of the challenges of lung transplant medicine in the era of SARS-CoV-2. “COVID-19,” said Dr. Narula, “has completely changed the way we practice lung transplant medicine – the way we’re looking both at our recipients and our donors.”
Potential donors are being evaluated with lower respiratory SARS-CoV-2 testing and an abundance of caution. And patients with severe COVID-19 affecting their own lungs are roundly expected to drive up lung transplant volume in the near future. “The whole paradigm has changed,” Dr. Narula said.
Post-acute trajectories
A chart review study published in October by the lung transplant team at the University of Texas Southwestern Medical Center, Dallas, covered 44 consecutive survivors at a median follow-up of 4.5 months from hospital discharge or acute illness (the survival rate was 83.3%). Patients had significantly impaired functional status, and 18 of the 44 (40.9%) had a significant and persistent loss of forced vital capacity or forced expiratory volume in 1 second (>10% from pre–COVID-19 baseline).
Three patients met the criteria for new CLAD after COVID-19 infection, with all three classified as restrictive allograft syndrome (RAS) phenotype.
Moreover, the majority of COVID-19 survivors who had CT chest scans (22 of 28) showed persistent parenchymal opacities – a finding that, regardless of symptomatology, suggests persistent allograft injury, said Amit Banga, MD, associate professor of medicine and medical director of the ex vivo lung perfusion program in UT Southwestern’s lung transplant program.
“The implication is that there may be long-term consequences of COVID-19, perhaps related to some degree of ongoing inflammation and damage,” said Dr. Banga, a coauthor of the postinfection outcomes paper.
The UT Southwestern lung transplant program, which normally performs 60-80 transplants a year, began routine CT scanning 4-5 months into the pandemic, after “stumbling into a few patients who had no symptoms indicative of COVID pneumonia and no changes on an x-ray but significant involvement on a CT,” he said.
Without routine scanning in the general population of COVID-19 patients, Dr. Banga noted, “we’re limited in convincingly saying that COVID is uniquely doing this to lung transplant recipients.” Nor can they conclude that SARS-CoV-2 is unique from other respiratory viruses such as respiratory syncytial virus (RSV) in this regard. (The program has added CT scanning to its protocol for lung transplant recipients afflicted with other respiratory viruses to learn more.)
However, in the big picture, COVID-19 has proven to be far worse for lung transplant recipients than illness with other respiratory viruses, including RSV. “Patients have more frequent and greater loss of lung function, and worse debility from the acute illness,” Dr. Banga said.
“The cornerstones of treatment of both these viruses are very similar, but both the in-hospital course and the postdischarge outcomes are significantly different.”
In an initial paper published in September 2021, Dr. Banga and colleagues compared their first 25 lung transplant patients testing positive for SARS-CoV-2 with a historical cohort of 36 patients with RSV treated during 2016-2018.
Patients with COVID-19 had significantly worse morbidity and mortality, including worse postinfection lung function loss, functional decline, and 3-month survival.
More time, he said, will shed light on the risks of CLAD and the long-term potential for recovery of lung function. Currently, at UT Southwestern, it appears that patients who survive acute illness and the “first 3-6 months after COVID-19, when we’re seeing all the postinfection morbidity, may [enter] a period of stability,” Dr. Banga said.
Overall, he said, patients in their initial cohort are “holding steady” without unusual morbidity, readmissions, or “other setbacks to their allografts.”
At the Mayo Clinic in Jacksonville, which normally performs 40-50 lung transplants a year, transplant physicians have similarly observed significant declines in lung function beyond the acute phase of COVID-19. “Anecdotally, we’re seeing that some patients are beginning to recover some of their lung function, while others have not,” said Dr. Narula. “And we don’t have predictors as to who will progress to CLAD. It’s a big knowledge gap.”
Dr. Narula noted that patients with restrictive allograft syndrome, such as those reported by the UT Southwestern team, “have scarring of the lung and a much worse prognosis than the obstructive type of chronic rejection.” Whether there’s a role for antifibrotic therapy is a question worthy of research.
In UT Southwestern’s analysis, persistently lower absolute lymphocyte counts (< 600/dL) and higher ferritin levels (>150 ng/mL) at the time of hospital discharge were independently associated with significant lung function loss. This finding, reported in their October paper, has helped guide their management practices, Dr. Banga said.
“Persistently elevated ferritin may indicate ongoing inflammation at the allograft level,” he said. “We now send [such patients] home on a longer course of oral corticosteroids.”
At the front end of care for infected lung transplant recipients, Dr. Banga said that his team and physicians at other lung transplant programs are holding the cell-cycle inhibitor component of patients’ maintenance immunosuppression therapy (commonly mycophenolate or azathioprine) once infection is diagnosed to maximize chances of a better outcome.
“There may be variation on how long [the regimens are adjusted],” he said. “We changed our duration from 4 weeks to 2 due to patients developing a rebound worsening in the third and fourth week of acute illness.”
There is significant variation from institution to institution in how viral infections are managed in lung transplant recipients, he and Dr. Narula said. “Our numbers are so small in lung transplant, and we don’t have standardized protocols – it’s one of the biggest challenges in our field,” said Dr. Narula.
Vaccination issues, evaluation of donors
Whether or not immunosuppression regimens should be adjusted prior to vaccination is a controversial question, but is “an absolutely valid one” and is currently being studied in at least one National Institutes of Health–funded trial involving solid organ transplant recipients, said Dr. Wolfe.
“Some have jumped to the conclusion [based on some earlier data] that they should reduce immunosuppression regimens for everyone at the time of vaccination ... but I don’t know the answer yet,” he said. “Balancing staying rejection free with potentially gaining more immune response is complicated ... and it may depend on where the pandemic is going in your area and other factors.”
Reductions aside, Dr. Wolfe tells lung transplant recipients that, based on his approximation of a number of different studies in solid organ transplant recipients, approximately 40%-50% of patients who are immunized with two doses of the COVID-19 mRNA vaccines will develop meaningful antibody levels – and that this rises to 50%-60% after a third dose.
It is difficult to glean from available studies the level of vaccine response for lung transplant recipients specifically. But given that their level of maintenance immunosuppression is higher than for recipients of other donor organs, “as a broad sweep, lung transplant recipients tend to be lower in the pecking order of response,” he said.
Still, “there’s a lot to gain,” he said, pointing to a recent study from the Morbidity and Mortality Weekly Report (2021 Nov 5. doi: 10.15585/mmwr.mm7044e3) showing that effectiveness of mRNA vaccination against COVID-19–associated hospitalization was 77% among immunocompromised adults (compared with 90% in immunocompetent adults).
“This is good vindication to keep vaccinating,” he said, “and perhaps speaks to how difficult it is to assess the vaccine response [through measurement of antibody only].”
Neither Duke University’s transplant program, which performed 100-120 lung transplants a year pre-COVID, nor the programs at UT Southwestern or the Mayo Clinic in Jacksonville require that solid organ transplant candidates be vaccinated against SARS-CoV-2 in order to receive transplants, as some other transplant programs have done. (When asked about the issue, Dr. Banga and Dr. Narula each said that they have had no or little trouble convincing patients awaiting lung transplants of the need for COVID-19 vaccination.)
In an August statement, the American Society of Transplantation recommended vaccination for all solid organ transplant recipients, preferably prior to transplantation, and said that it “support[s] the development of institutional policies regarding pretransplant vaccination.”
The Society is not tracking centers’ vaccination policies. But Kaiser Health News reported in October that a growing number of transplant programs, such as UCHealth in Denver and UW Medicine in Seattle, have decided to either bar patients who refuse to be vaccinated from receiving transplants or give them lower priority on waitlists.
Potential lung donors, meanwhile, must be evaluated with lower respiratory COVID-19 testing, with results available prior to transplantation, according to policy developed by the Organ Procurement and Transplantation Network and effective in May 2021. The policy followed three published cases of donor-derived COVID-19 in lung transplant recipients, said Dr. Wolfe, who wrote about use of COVID-positive donors in an editorial published in October.
In each case, the donor had a negative COVID-19 nasopharyngeal swab at the time of organ procurement but was later found to have the virus on bronchoalveolar lavage, he said.
(The use of other organs from COVID-positive donors is appearing thus far to be safe, Dr. Wolfe noted. In the editorial, he references 13 cases of solid organ transplantation from SARS-CoV-2–infected donors into noninfected recipients; none of the 13 transplant recipients developed COVID-19).
Some questions remain, such as how many lower respiratory tests should be run, and how donors should be evaluated in cases of discordant results. Dr. Banga shared the case of a donor with one positive lower respiratory test result followed by two negative results. After internal debate, and consideration of potential false positives and other issues, the team at UT Southwestern decided to decline the donor, Dr. Banga said.
Other programs are likely making similar, appropriately cautious decisions, said Dr. Wolfe. “There’s no way in real-time donor evaluation to know whether the positive test is active virus that could infect the recipient and replicate ... or whether it’s [picking up] inactive or dead fragments of virus that was there several weeks ago. Our tests don’t differentiate that.”
Transplants in COVID-19 patients
Decision-making about lung transplant candidacy among patients with COVID-19 acute respiratory distress syndrome is complex and in need of a new paradigm.
“Some of these patients have the potential to recover, and they’re going to recover way later than what we’re used to,” said Dr. Banga. “We can’t extrapolate for COVID ARDS what we’ve learned for any other virus-related ARDS.”
Dr. Narula also has recently seen at least one COVID-19 patient on ECMO and under evaluation for transplantation recover. “We do not want to transplant too early,” he said, noting that there is consensus that lung transplant should be pursued only when the damage is deemed irreversible clinically and radiologically in the best judgment of the team. Still, “for many of these patients the only exit route will be lung transplants. For the next 12-24 months, a significant proportion of our lung transplant patients will have had post-COVID–related lung damage.”
As of October 2021, 233 lung transplants had been performed in the United States in recipients whose primary diagnosis was reported as COVID related, said Anne Paschke, media relations specialist with the United Network for Organ Sharing.
Dr. Banga, Dr. Wolfe, and Dr. Narula reported that they have no relevant disclosures.
Major COVID-19 case growth expected in coming weeks
by the PolicyLab at Children’s Hospital of Philadelphia.
Large metropolitan areas, especially those in the Northeast, are already seeing a major increase in cases following Thanksgiving, and that trend is expected to continue.
“Why? Simply stated, the large amount of Thanksgiving travel and gatherings undermined the nation’s pandemic footing and has elevated disease burden in areas of the country that were fortunate to have lower case rates before the holidays,” the forecasters wrote.
Case numbers in New York City are expected to double throughout December, the forecasters said. Similar growth could happen across Boston, Philadelphia, and Baltimore.
Overall, COVID-19 cases, hospitalizations, and deaths are rising across the United States but remain below levels seen during the summer and last winter’s surges, according to the New York Times. The increase is still being driven by the Delta variant, though it remains unclear how the Omicron variant, which has been detected in 27 states, could affect the trends in the coming weeks.
During the past week, the United States has reported an average of more than 120,000 new cases each day, the newspaper reported, which is an increase of 38% from two weeks ago.
The daily average of COVID-19 hospitalizations is around 64,000, which marks an increase of 22% from two weeks ago. More than 1,300 deaths are being reported each day, which is up 26%.
Numerous states are reporting double the cases from two weeks ago, stretching across the country from states in the Northeast such as Connecticut and Rhode Island to southern states such as North Carolina and Texas and western states such as California.
The Great Lakes region and the Northeast are seeing some of the most severe increases, the newspaper reported. New Hampshire leads the United States in recent cases per capita, and Maine has reported more cases in the past week than in any other seven-day period during the pandemic.
Michigan has the country’s highest hospitalization rate, and federal medical teams have been sent to the state to help with the surge in patients, according to The Detroit News. Michigan’s top public health officials described the surge as a “critical” and “deeply concerning” situation on Dec. 10, and they requested 200 more ventilators from the Strategic National Stockpile.
Indiana, Maine, and New York have also requested aid from the National Guard, according to USA Today. Health officials in those states urged residents to get vaccines or booster shots and wear masks in indoor public settings.
The Omicron variant can evade some vaccine protection, but booster shots can increase efficacy and offer more coverage, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said Dec. 12.
“If you want to be optimally protected, absolutely get a booster,” he said on ABC’s “This Week.”
In addition, New York Gov. Kathy Hochul has announced a statewide mask mandate, which will take effect Dec. 13. Masks will be required in all indoor public spaces and businesses, unless the location implements a vaccine requirement instead, the news outlet reported.
A version of this article first appeared on WebMD.com.
by the PolicyLab at Children’s Hospital of Philadelphia.
Large metropolitan areas, especially those in the Northeast, are already seeing a major increase in cases following Thanksgiving, and that trend is expected to continue.
“Why? Simply stated, the large amount of Thanksgiving travel and gatherings undermined the nation’s pandemic footing and has elevated disease burden in areas of the country that were fortunate to have lower case rates before the holidays,” the forecasters wrote.
Case numbers in New York City are expected to double throughout December, the forecasters said. Similar growth could happen across Boston, Philadelphia, and Baltimore.
Overall, COVID-19 cases, hospitalizations, and deaths are rising across the United States but remain below levels seen during the summer and last winter’s surges, according to the New York Times. The increase is still being driven by the Delta variant, though it remains unclear how the Omicron variant, which has been detected in 27 states, could affect the trends in the coming weeks.
During the past week, the United States has reported an average of more than 120,000 new cases each day, the newspaper reported, which is an increase of 38% from two weeks ago.
The daily average of COVID-19 hospitalizations is around 64,000, which marks an increase of 22% from two weeks ago. More than 1,300 deaths are being reported each day, which is up 26%.
Numerous states are reporting double the cases from two weeks ago, stretching across the country from states in the Northeast such as Connecticut and Rhode Island to southern states such as North Carolina and Texas and western states such as California.
The Great Lakes region and the Northeast are seeing some of the most severe increases, the newspaper reported. New Hampshire leads the United States in recent cases per capita, and Maine has reported more cases in the past week than in any other seven-day period during the pandemic.
Michigan has the country’s highest hospitalization rate, and federal medical teams have been sent to the state to help with the surge in patients, according to The Detroit News. Michigan’s top public health officials described the surge as a “critical” and “deeply concerning” situation on Dec. 10, and they requested 200 more ventilators from the Strategic National Stockpile.
Indiana, Maine, and New York have also requested aid from the National Guard, according to USA Today. Health officials in those states urged residents to get vaccines or booster shots and wear masks in indoor public settings.
The Omicron variant can evade some vaccine protection, but booster shots can increase efficacy and offer more coverage, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said Dec. 12.
“If you want to be optimally protected, absolutely get a booster,” he said on ABC’s “This Week.”
In addition, New York Gov. Kathy Hochul has announced a statewide mask mandate, which will take effect Dec. 13. Masks will be required in all indoor public spaces and businesses, unless the location implements a vaccine requirement instead, the news outlet reported.
A version of this article first appeared on WebMD.com.
by the PolicyLab at Children’s Hospital of Philadelphia.
Large metropolitan areas, especially those in the Northeast, are already seeing a major increase in cases following Thanksgiving, and that trend is expected to continue.
“Why? Simply stated, the large amount of Thanksgiving travel and gatherings undermined the nation’s pandemic footing and has elevated disease burden in areas of the country that were fortunate to have lower case rates before the holidays,” the forecasters wrote.
Case numbers in New York City are expected to double throughout December, the forecasters said. Similar growth could happen across Boston, Philadelphia, and Baltimore.
Overall, COVID-19 cases, hospitalizations, and deaths are rising across the United States but remain below levels seen during the summer and last winter’s surges, according to the New York Times. The increase is still being driven by the Delta variant, though it remains unclear how the Omicron variant, which has been detected in 27 states, could affect the trends in the coming weeks.
During the past week, the United States has reported an average of more than 120,000 new cases each day, the newspaper reported, which is an increase of 38% from two weeks ago.
The daily average of COVID-19 hospitalizations is around 64,000, which marks an increase of 22% from two weeks ago. More than 1,300 deaths are being reported each day, which is up 26%.
Numerous states are reporting double the cases from two weeks ago, stretching across the country from states in the Northeast such as Connecticut and Rhode Island to southern states such as North Carolina and Texas and western states such as California.
The Great Lakes region and the Northeast are seeing some of the most severe increases, the newspaper reported. New Hampshire leads the United States in recent cases per capita, and Maine has reported more cases in the past week than in any other seven-day period during the pandemic.
Michigan has the country’s highest hospitalization rate, and federal medical teams have been sent to the state to help with the surge in patients, according to The Detroit News. Michigan’s top public health officials described the surge as a “critical” and “deeply concerning” situation on Dec. 10, and they requested 200 more ventilators from the Strategic National Stockpile.
Indiana, Maine, and New York have also requested aid from the National Guard, according to USA Today. Health officials in those states urged residents to get vaccines or booster shots and wear masks in indoor public settings.
The Omicron variant can evade some vaccine protection, but booster shots can increase efficacy and offer more coverage, Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said Dec. 12.
“If you want to be optimally protected, absolutely get a booster,” he said on ABC’s “This Week.”
In addition, New York Gov. Kathy Hochul has announced a statewide mask mandate, which will take effect Dec. 13. Masks will be required in all indoor public spaces and businesses, unless the location implements a vaccine requirement instead, the news outlet reported.
A version of this article first appeared on WebMD.com.
Coping with a shattered immune system: COVID and beyond
The co-opting and weakening of the immune system by hematologic malignancies and many of their treatments – and the blunting of the immune system’s response to vaccines – may be more salient during the COVID-19 pandemic than ever before.
Hematologic malignancies have been associated in large cancer-and-COVID-19 registries with more severe COVID-19 outcomes than solid tumors, and COVID-19 mRNA vaccines have yielded suboptimal responses across multiple studies. Clinicians and researchers have no shortage of questions, like what is the optimal timing of vaccines relative to cancer-directed therapy? What is the durability and impact of the immune response? What is the status of the immune system in patients who do not produce antispike antibodies after COVID-19 vaccination?
Moreover, will there be novel nonvaccine strategies – such as antibody cocktails or convalescent plasma – to ensure protection against COVID-19 and other future viral threats? And what really defines immunocompromise today and moving forward?
“We don’t know what we don’t know,” said Jeremy L. Warner, MD, associate professor of medicine (hematology/oncology) and biomedical informatics at Vanderbilt University, Nashville, Tenn., and cofounder of the international COVID-19 and Cancer Consortium. “The immune system is incredibly complex and there are numerous defenses, in addition to the humoral response that we routinely measure.”
Another of the pressing pandemic-time questions for infectious disease specialists working in cancer centers concerns a different infectious threat: measles. “There is a lot of concern in this space about the reported drop in childhood vaccinations and the possibility of measles outbreaks as a follow-up to COVID-19,” said Steven A. Pergam, MD, MPH, associate professor in the vaccine and infectious disease division and the clinical research division of the Fred Hutchinson Cancer Research Center, Seattle Cancer Care Alliance.
Whether recipients of hematopoietic cell transplantation (HCT) and cellular therapy should be revaccinated earlier than 2 years post treatment is a question worthy of preemptive discussion, he said.
What about timing?
“A silver lining of the pandemic is that it’s improving our understanding of response to vaccinations and outcomes with respiratory viruses in patients with hematologic malignancies,” said Samuel Rubinstein, MD, of the division of hematology at the University of North Carolina at Chapel Hill. “We’re going to learn a lot more about how to ensure that our patients are optimally protected from respiratory viruses.”
Dr. Rubinstein focuses on plasma cell disorders, mostly multiple myeloma, and routinely explains to patients consenting to use daratumumab, an anti-CD38 monoclonal antibody, or a BCMA-directed therapy, that these therapies “in particular probably do impair vaccine immune response.”
He has handled the timing of the COVID-19 vaccines – currently boosters, in most cases – as he has with influenza and other immunizations such as the pneumococcal vaccine, administering the vaccines agnostic to therapy unless the patient is about to start daratumumab or a BCMA-directed therapy. In this case, he considers vaccinating and waiting 2 weeks (for an immune response to occur) before starting therapy.
However, “if I have any concern that a delay will result in suboptimal cancer control, then I don’t wait,” Dr. Rubinstein said. Poor control of a primary malignancy has been consistently associated with worse COVID-19–specific outcomes in cancer–COVID-19 studies, he said, including an analysis of almost 5,000 patients recorded to the COVID-19 and Cancer Consortium .1
(The analysis also documented that patients with a hematologic malignancy had an odds ratio of higher COVID-19 severity of 1.7, compared with patients with a solid tumor, and an odds ratio of 30-day mortality of 1.44.)
Ideally, said Dr. Warner, patients will get vaccinated with the COVID-19 vaccines or others, “before starting on any cytotoxic chemotherapy and when they do not have low blood counts or perhaps autoimmune complications of immunotherapy.” However, “perfect being the enemy of good, it’s better to get vaccinated than to wait for the exact ideal time.”
Peter Paul Yu, MD, physician-in-chief at Hartford (Conn.) Healthcare Cancer Institute, said that for most patients, there’s no evidence to support an optimal timing of vaccine administration during the chemotherapy cycle. “We looked into that [to guide administration of the COVID-19 vaccines], thinking there might be some data about influenza vaccination,” he said. “But there isn’t much. … And if we make things more complicated than the evidence suggests, we may have fewer people getting vaccinations.”
The National Comprehensive Cancer Network offered several timing recommendations in its August 2021 COVID-19 vaccination guidance – mainly that patients receiving intensive cytotoxic chemotherapy (such as those on cytarabine/anthracycline-based induction regimens for acute myeloid leukemia) delay COVID-19 vaccination until absolute neutrophil count recovery, and that patients on long-term maintenance therapy (for instance, targeted agents for chronic lymphocytic leukemia or myeloproliferative neoplasms) be vaccinated as soon as possible.
Vaccination should be delayed for at least 3 months, the NCCN noted, following HCT or engineered cell therapy (for example, chimeric antigen receptor [CAR] T cells) “in order to maximize vaccine efficacy.”
More known unknowns
The tempered efficacy of the COVID-19 vaccines in patients with hematologic malignancies “has been shown in multiple studies of multiple myeloma, chronic lymphocytic leukemia (CLL), and other malignancies, and we know it’s true in transplant,” said Dr. Pergam.
In a study of 67 patients with hematologic malignancies at the University of Pittsburgh Medical Center Hillman Cancer Center, for instance, 46.3% did not generate IgG antibodies against the SARS-CoV-2 spike protein receptor–binding domain after completing their two-dose mRNA vaccine series. Patients with B-cell CLL were especially unlikely to develop antibodies.2A much larger study of more than 1,400 patients from investigators at the Mayo Clinics in Rochester, Minn., and Jacksonville, Fla., found that approximately 25% of all patients with hematologic malignancies did not produce antispike IgG antibodies, and that those with the most common B-cell malignancies had the lowest rate of seropositivity (44%-79%).3There’s a clear but challenging delineation between antibody testing in the research space and in clinical practice, however. Various national and cancer societies recommended earlier this year against routine postvaccine serological monitoring outside of clinical trials, and the sources interviewed for this story all emphasized that antibody titer measurements should not guide decisions about boosters or about the precautions advised for patients.
Titers checked at a single point in time do not capture the kinetics, multidimensional nature, or durability of an immune response, Dr. Warner said. “There are papers out there that say zero patients with CCL seroconverted … but they do still have some immunity, and maybe even a lot of immunity.”
Antibody testing can create a false sense of security, or a false sense of dread, he said. Yet in practice, the use of serological monitoring “has been all over the place [with] no consistency … and decisions probably being made at the individual clinic level or health system level,” he said.
To a lesser degree, so have definitions of what composes significant immunocompromise in the context of COVID-19 vaccine eligibility. “The question comes up, what does immunocompromised really mean?” said Dr. Yu, whose institution is a member of the Memorial Sloan Kettering (MSK) Cancer Alliance.
As of September, the MSK Cancer Center had taken a more granular approach to describing moderate to severe immunocompromise than did the Centers for Disease Control and Prevention. The CDC said this level of immunocompromise includes people receiving active cancer treatment for tumors or cancers of the blood, and those who’ve received a stem cell transplant within the past 2 years. MSK extended the recommendation, as it concerns hematologic malignancies, to patients who are within 12 months after treatment with B-cell depleting drugs, patients who have been treated for blood cancers within the last 6 months, and patients who received CAR T therapy within the past 2 years.
Dr. Yu, who was not involved in creating the MSK recommendations for third COVID-19 vaccines, said that he has been thinking more broadly during the pandemic about the notion of immunocompetence. “It’s my opinion that patients with hematologic malignancies, even if they’re not on treatment, are not fully immune competent,” he said. This includes patients with CLL stage 0 and patients with plasma cell dyscrasias who don’t yet meet the criteria for multiple myeloma but have a monoclonal gammopathy, and those with lower-risk myelodysplastic syndromes, he said.
“We’re seeing [variable] recommendations based on expert opinion, and I think that’s justifiable in such a dynamic situation,” Dr. Yu said. “I would [even] argue it’s desirable so we can learn from different approaches” and collect more rigorous observational data.
Immunocompetence needs to be “viewed in the context of the threat,” he added. “COVID changes the equation. … What’s immunocompromised in my mind has changed [from prepandemic times].”
Preparing for measles
Measles lit up on Dr. Pergam’s radar screen in 2019, when an outbreak occurred in nearby Clark County, Wash. This and other outbreaks in New York, California, and other states highlighted declines in measles herd immunity in the United States and prompted him to investigate the seroprevalence of measles antibodies in the Fred Hutchinson Cancer Research Center’s outpatient population.
Of 959 consecutive patients seen at the center, they found, 25% lacked protective antibodies for measles. For patients with hematologic malignancies and those with a history of HCT, seroprevalence was worse: 37% and 54%, respectively, were without the IgG antibodies.4 Measles “is the most contagious human virus we have at the moment,” he said, and “revaccinating people is hard when it comes to cancer because it is a live virus vaccine.”
Vaccine hesitancy, a rise in nonmedical exemptions, and other factors were threatening herd immunity before the pandemic began. Now, with declines in routine childhood medical visits and other vaccination opportunities and resources here and in other countries – and declining immunization rates documented by the CDC in May 2021 – the pandemic has made measles outbreaks more likely, he said. (Measles outbreaks in West Africa on the tail end of the Ebola outbreak in 2014-2015 caused more deaths in children than Ebola, he noted.)
The first priority is vaccination “cocooning,” a strategy that has long been important for patients with hematologic malignancies. But it also possible, Dr. Pergam said, that in the setting of any future community transmission, revaccination for HCT recipients could occur earlier than the standard 2-year post-transplantation recommendation.
In a 2019 position statement endorsed by the American Society for Transplantation and Cellular Therapy, Dr. Pergam and other infectious disease physicians and oncologists provide criteria for considering early revaccination on a case-by-case basis for patients on minimal immunosuppressive therapy who are at least 1-year post transplantation.5
“Our thinking was that there may be lower-risk patients to whom we could offer the vaccine” – patients for whom the risk of developing measles might outweigh the risk of potential vaccine-related complications, he said.
And if there were community cases, he added, there might be a place for testing antibody levels in post-transplant patients, however imperfect the window to immunity may be. “We’re thinking through potential scenarios,” he said. “Oncologists should think about measles again and have it on their back burner.”
References
1. Grivas P et al. Ann Oncol. 2021 Jun;32(6):787-800.
2. Agha ME et al. Open Forum Infect Dis. 2021 July;8(7):ofab353.
3. Greenberger LM et al. Cancer Cell. 2021 Aug 9;39(8):1031-3.
4. Marquis SR et al. JAMA Netw Open. 2021 July;4(7):e2118508.
5. Pergam SA et al. Biol Blood Marrow Transplant. 2019 Nov;25:e321-30.
The co-opting and weakening of the immune system by hematologic malignancies and many of their treatments – and the blunting of the immune system’s response to vaccines – may be more salient during the COVID-19 pandemic than ever before.
Hematologic malignancies have been associated in large cancer-and-COVID-19 registries with more severe COVID-19 outcomes than solid tumors, and COVID-19 mRNA vaccines have yielded suboptimal responses across multiple studies. Clinicians and researchers have no shortage of questions, like what is the optimal timing of vaccines relative to cancer-directed therapy? What is the durability and impact of the immune response? What is the status of the immune system in patients who do not produce antispike antibodies after COVID-19 vaccination?
Moreover, will there be novel nonvaccine strategies – such as antibody cocktails or convalescent plasma – to ensure protection against COVID-19 and other future viral threats? And what really defines immunocompromise today and moving forward?
“We don’t know what we don’t know,” said Jeremy L. Warner, MD, associate professor of medicine (hematology/oncology) and biomedical informatics at Vanderbilt University, Nashville, Tenn., and cofounder of the international COVID-19 and Cancer Consortium. “The immune system is incredibly complex and there are numerous defenses, in addition to the humoral response that we routinely measure.”
Another of the pressing pandemic-time questions for infectious disease specialists working in cancer centers concerns a different infectious threat: measles. “There is a lot of concern in this space about the reported drop in childhood vaccinations and the possibility of measles outbreaks as a follow-up to COVID-19,” said Steven A. Pergam, MD, MPH, associate professor in the vaccine and infectious disease division and the clinical research division of the Fred Hutchinson Cancer Research Center, Seattle Cancer Care Alliance.
Whether recipients of hematopoietic cell transplantation (HCT) and cellular therapy should be revaccinated earlier than 2 years post treatment is a question worthy of preemptive discussion, he said.
What about timing?
“A silver lining of the pandemic is that it’s improving our understanding of response to vaccinations and outcomes with respiratory viruses in patients with hematologic malignancies,” said Samuel Rubinstein, MD, of the division of hematology at the University of North Carolina at Chapel Hill. “We’re going to learn a lot more about how to ensure that our patients are optimally protected from respiratory viruses.”
Dr. Rubinstein focuses on plasma cell disorders, mostly multiple myeloma, and routinely explains to patients consenting to use daratumumab, an anti-CD38 monoclonal antibody, or a BCMA-directed therapy, that these therapies “in particular probably do impair vaccine immune response.”
He has handled the timing of the COVID-19 vaccines – currently boosters, in most cases – as he has with influenza and other immunizations such as the pneumococcal vaccine, administering the vaccines agnostic to therapy unless the patient is about to start daratumumab or a BCMA-directed therapy. In this case, he considers vaccinating and waiting 2 weeks (for an immune response to occur) before starting therapy.
However, “if I have any concern that a delay will result in suboptimal cancer control, then I don’t wait,” Dr. Rubinstein said. Poor control of a primary malignancy has been consistently associated with worse COVID-19–specific outcomes in cancer–COVID-19 studies, he said, including an analysis of almost 5,000 patients recorded to the COVID-19 and Cancer Consortium .1
(The analysis also documented that patients with a hematologic malignancy had an odds ratio of higher COVID-19 severity of 1.7, compared with patients with a solid tumor, and an odds ratio of 30-day mortality of 1.44.)
Ideally, said Dr. Warner, patients will get vaccinated with the COVID-19 vaccines or others, “before starting on any cytotoxic chemotherapy and when they do not have low blood counts or perhaps autoimmune complications of immunotherapy.” However, “perfect being the enemy of good, it’s better to get vaccinated than to wait for the exact ideal time.”
Peter Paul Yu, MD, physician-in-chief at Hartford (Conn.) Healthcare Cancer Institute, said that for most patients, there’s no evidence to support an optimal timing of vaccine administration during the chemotherapy cycle. “We looked into that [to guide administration of the COVID-19 vaccines], thinking there might be some data about influenza vaccination,” he said. “But there isn’t much. … And if we make things more complicated than the evidence suggests, we may have fewer people getting vaccinations.”
The National Comprehensive Cancer Network offered several timing recommendations in its August 2021 COVID-19 vaccination guidance – mainly that patients receiving intensive cytotoxic chemotherapy (such as those on cytarabine/anthracycline-based induction regimens for acute myeloid leukemia) delay COVID-19 vaccination until absolute neutrophil count recovery, and that patients on long-term maintenance therapy (for instance, targeted agents for chronic lymphocytic leukemia or myeloproliferative neoplasms) be vaccinated as soon as possible.
Vaccination should be delayed for at least 3 months, the NCCN noted, following HCT or engineered cell therapy (for example, chimeric antigen receptor [CAR] T cells) “in order to maximize vaccine efficacy.”
More known unknowns
The tempered efficacy of the COVID-19 vaccines in patients with hematologic malignancies “has been shown in multiple studies of multiple myeloma, chronic lymphocytic leukemia (CLL), and other malignancies, and we know it’s true in transplant,” said Dr. Pergam.
In a study of 67 patients with hematologic malignancies at the University of Pittsburgh Medical Center Hillman Cancer Center, for instance, 46.3% did not generate IgG antibodies against the SARS-CoV-2 spike protein receptor–binding domain after completing their two-dose mRNA vaccine series. Patients with B-cell CLL were especially unlikely to develop antibodies.2A much larger study of more than 1,400 patients from investigators at the Mayo Clinics in Rochester, Minn., and Jacksonville, Fla., found that approximately 25% of all patients with hematologic malignancies did not produce antispike IgG antibodies, and that those with the most common B-cell malignancies had the lowest rate of seropositivity (44%-79%).3There’s a clear but challenging delineation between antibody testing in the research space and in clinical practice, however. Various national and cancer societies recommended earlier this year against routine postvaccine serological monitoring outside of clinical trials, and the sources interviewed for this story all emphasized that antibody titer measurements should not guide decisions about boosters or about the precautions advised for patients.
Titers checked at a single point in time do not capture the kinetics, multidimensional nature, or durability of an immune response, Dr. Warner said. “There are papers out there that say zero patients with CCL seroconverted … but they do still have some immunity, and maybe even a lot of immunity.”
Antibody testing can create a false sense of security, or a false sense of dread, he said. Yet in practice, the use of serological monitoring “has been all over the place [with] no consistency … and decisions probably being made at the individual clinic level or health system level,” he said.
To a lesser degree, so have definitions of what composes significant immunocompromise in the context of COVID-19 vaccine eligibility. “The question comes up, what does immunocompromised really mean?” said Dr. Yu, whose institution is a member of the Memorial Sloan Kettering (MSK) Cancer Alliance.
As of September, the MSK Cancer Center had taken a more granular approach to describing moderate to severe immunocompromise than did the Centers for Disease Control and Prevention. The CDC said this level of immunocompromise includes people receiving active cancer treatment for tumors or cancers of the blood, and those who’ve received a stem cell transplant within the past 2 years. MSK extended the recommendation, as it concerns hematologic malignancies, to patients who are within 12 months after treatment with B-cell depleting drugs, patients who have been treated for blood cancers within the last 6 months, and patients who received CAR T therapy within the past 2 years.
Dr. Yu, who was not involved in creating the MSK recommendations for third COVID-19 vaccines, said that he has been thinking more broadly during the pandemic about the notion of immunocompetence. “It’s my opinion that patients with hematologic malignancies, even if they’re not on treatment, are not fully immune competent,” he said. This includes patients with CLL stage 0 and patients with plasma cell dyscrasias who don’t yet meet the criteria for multiple myeloma but have a monoclonal gammopathy, and those with lower-risk myelodysplastic syndromes, he said.
“We’re seeing [variable] recommendations based on expert opinion, and I think that’s justifiable in such a dynamic situation,” Dr. Yu said. “I would [even] argue it’s desirable so we can learn from different approaches” and collect more rigorous observational data.
Immunocompetence needs to be “viewed in the context of the threat,” he added. “COVID changes the equation. … What’s immunocompromised in my mind has changed [from prepandemic times].”
Preparing for measles
Measles lit up on Dr. Pergam’s radar screen in 2019, when an outbreak occurred in nearby Clark County, Wash. This and other outbreaks in New York, California, and other states highlighted declines in measles herd immunity in the United States and prompted him to investigate the seroprevalence of measles antibodies in the Fred Hutchinson Cancer Research Center’s outpatient population.
Of 959 consecutive patients seen at the center, they found, 25% lacked protective antibodies for measles. For patients with hematologic malignancies and those with a history of HCT, seroprevalence was worse: 37% and 54%, respectively, were without the IgG antibodies.4 Measles “is the most contagious human virus we have at the moment,” he said, and “revaccinating people is hard when it comes to cancer because it is a live virus vaccine.”
Vaccine hesitancy, a rise in nonmedical exemptions, and other factors were threatening herd immunity before the pandemic began. Now, with declines in routine childhood medical visits and other vaccination opportunities and resources here and in other countries – and declining immunization rates documented by the CDC in May 2021 – the pandemic has made measles outbreaks more likely, he said. (Measles outbreaks in West Africa on the tail end of the Ebola outbreak in 2014-2015 caused more deaths in children than Ebola, he noted.)
The first priority is vaccination “cocooning,” a strategy that has long been important for patients with hematologic malignancies. But it also possible, Dr. Pergam said, that in the setting of any future community transmission, revaccination for HCT recipients could occur earlier than the standard 2-year post-transplantation recommendation.
In a 2019 position statement endorsed by the American Society for Transplantation and Cellular Therapy, Dr. Pergam and other infectious disease physicians and oncologists provide criteria for considering early revaccination on a case-by-case basis for patients on minimal immunosuppressive therapy who are at least 1-year post transplantation.5
“Our thinking was that there may be lower-risk patients to whom we could offer the vaccine” – patients for whom the risk of developing measles might outweigh the risk of potential vaccine-related complications, he said.
And if there were community cases, he added, there might be a place for testing antibody levels in post-transplant patients, however imperfect the window to immunity may be. “We’re thinking through potential scenarios,” he said. “Oncologists should think about measles again and have it on their back burner.”
References
1. Grivas P et al. Ann Oncol. 2021 Jun;32(6):787-800.
2. Agha ME et al. Open Forum Infect Dis. 2021 July;8(7):ofab353.
3. Greenberger LM et al. Cancer Cell. 2021 Aug 9;39(8):1031-3.
4. Marquis SR et al. JAMA Netw Open. 2021 July;4(7):e2118508.
5. Pergam SA et al. Biol Blood Marrow Transplant. 2019 Nov;25:e321-30.
The co-opting and weakening of the immune system by hematologic malignancies and many of their treatments – and the blunting of the immune system’s response to vaccines – may be more salient during the COVID-19 pandemic than ever before.
Hematologic malignancies have been associated in large cancer-and-COVID-19 registries with more severe COVID-19 outcomes than solid tumors, and COVID-19 mRNA vaccines have yielded suboptimal responses across multiple studies. Clinicians and researchers have no shortage of questions, like what is the optimal timing of vaccines relative to cancer-directed therapy? What is the durability and impact of the immune response? What is the status of the immune system in patients who do not produce antispike antibodies after COVID-19 vaccination?
Moreover, will there be novel nonvaccine strategies – such as antibody cocktails or convalescent plasma – to ensure protection against COVID-19 and other future viral threats? And what really defines immunocompromise today and moving forward?
“We don’t know what we don’t know,” said Jeremy L. Warner, MD, associate professor of medicine (hematology/oncology) and biomedical informatics at Vanderbilt University, Nashville, Tenn., and cofounder of the international COVID-19 and Cancer Consortium. “The immune system is incredibly complex and there are numerous defenses, in addition to the humoral response that we routinely measure.”
Another of the pressing pandemic-time questions for infectious disease specialists working in cancer centers concerns a different infectious threat: measles. “There is a lot of concern in this space about the reported drop in childhood vaccinations and the possibility of measles outbreaks as a follow-up to COVID-19,” said Steven A. Pergam, MD, MPH, associate professor in the vaccine and infectious disease division and the clinical research division of the Fred Hutchinson Cancer Research Center, Seattle Cancer Care Alliance.
Whether recipients of hematopoietic cell transplantation (HCT) and cellular therapy should be revaccinated earlier than 2 years post treatment is a question worthy of preemptive discussion, he said.
What about timing?
“A silver lining of the pandemic is that it’s improving our understanding of response to vaccinations and outcomes with respiratory viruses in patients with hematologic malignancies,” said Samuel Rubinstein, MD, of the division of hematology at the University of North Carolina at Chapel Hill. “We’re going to learn a lot more about how to ensure that our patients are optimally protected from respiratory viruses.”
Dr. Rubinstein focuses on plasma cell disorders, mostly multiple myeloma, and routinely explains to patients consenting to use daratumumab, an anti-CD38 monoclonal antibody, or a BCMA-directed therapy, that these therapies “in particular probably do impair vaccine immune response.”
He has handled the timing of the COVID-19 vaccines – currently boosters, in most cases – as he has with influenza and other immunizations such as the pneumococcal vaccine, administering the vaccines agnostic to therapy unless the patient is about to start daratumumab or a BCMA-directed therapy. In this case, he considers vaccinating and waiting 2 weeks (for an immune response to occur) before starting therapy.
However, “if I have any concern that a delay will result in suboptimal cancer control, then I don’t wait,” Dr. Rubinstein said. Poor control of a primary malignancy has been consistently associated with worse COVID-19–specific outcomes in cancer–COVID-19 studies, he said, including an analysis of almost 5,000 patients recorded to the COVID-19 and Cancer Consortium .1
(The analysis also documented that patients with a hematologic malignancy had an odds ratio of higher COVID-19 severity of 1.7, compared with patients with a solid tumor, and an odds ratio of 30-day mortality of 1.44.)
Ideally, said Dr. Warner, patients will get vaccinated with the COVID-19 vaccines or others, “before starting on any cytotoxic chemotherapy and when they do not have low blood counts or perhaps autoimmune complications of immunotherapy.” However, “perfect being the enemy of good, it’s better to get vaccinated than to wait for the exact ideal time.”
Peter Paul Yu, MD, physician-in-chief at Hartford (Conn.) Healthcare Cancer Institute, said that for most patients, there’s no evidence to support an optimal timing of vaccine administration during the chemotherapy cycle. “We looked into that [to guide administration of the COVID-19 vaccines], thinking there might be some data about influenza vaccination,” he said. “But there isn’t much. … And if we make things more complicated than the evidence suggests, we may have fewer people getting vaccinations.”
The National Comprehensive Cancer Network offered several timing recommendations in its August 2021 COVID-19 vaccination guidance – mainly that patients receiving intensive cytotoxic chemotherapy (such as those on cytarabine/anthracycline-based induction regimens for acute myeloid leukemia) delay COVID-19 vaccination until absolute neutrophil count recovery, and that patients on long-term maintenance therapy (for instance, targeted agents for chronic lymphocytic leukemia or myeloproliferative neoplasms) be vaccinated as soon as possible.
Vaccination should be delayed for at least 3 months, the NCCN noted, following HCT or engineered cell therapy (for example, chimeric antigen receptor [CAR] T cells) “in order to maximize vaccine efficacy.”
More known unknowns
The tempered efficacy of the COVID-19 vaccines in patients with hematologic malignancies “has been shown in multiple studies of multiple myeloma, chronic lymphocytic leukemia (CLL), and other malignancies, and we know it’s true in transplant,” said Dr. Pergam.
In a study of 67 patients with hematologic malignancies at the University of Pittsburgh Medical Center Hillman Cancer Center, for instance, 46.3% did not generate IgG antibodies against the SARS-CoV-2 spike protein receptor–binding domain after completing their two-dose mRNA vaccine series. Patients with B-cell CLL were especially unlikely to develop antibodies.2A much larger study of more than 1,400 patients from investigators at the Mayo Clinics in Rochester, Minn., and Jacksonville, Fla., found that approximately 25% of all patients with hematologic malignancies did not produce antispike IgG antibodies, and that those with the most common B-cell malignancies had the lowest rate of seropositivity (44%-79%).3There’s a clear but challenging delineation between antibody testing in the research space and in clinical practice, however. Various national and cancer societies recommended earlier this year against routine postvaccine serological monitoring outside of clinical trials, and the sources interviewed for this story all emphasized that antibody titer measurements should not guide decisions about boosters or about the precautions advised for patients.
Titers checked at a single point in time do not capture the kinetics, multidimensional nature, or durability of an immune response, Dr. Warner said. “There are papers out there that say zero patients with CCL seroconverted … but they do still have some immunity, and maybe even a lot of immunity.”
Antibody testing can create a false sense of security, or a false sense of dread, he said. Yet in practice, the use of serological monitoring “has been all over the place [with] no consistency … and decisions probably being made at the individual clinic level or health system level,” he said.
To a lesser degree, so have definitions of what composes significant immunocompromise in the context of COVID-19 vaccine eligibility. “The question comes up, what does immunocompromised really mean?” said Dr. Yu, whose institution is a member of the Memorial Sloan Kettering (MSK) Cancer Alliance.
As of September, the MSK Cancer Center had taken a more granular approach to describing moderate to severe immunocompromise than did the Centers for Disease Control and Prevention. The CDC said this level of immunocompromise includes people receiving active cancer treatment for tumors or cancers of the blood, and those who’ve received a stem cell transplant within the past 2 years. MSK extended the recommendation, as it concerns hematologic malignancies, to patients who are within 12 months after treatment with B-cell depleting drugs, patients who have been treated for blood cancers within the last 6 months, and patients who received CAR T therapy within the past 2 years.
Dr. Yu, who was not involved in creating the MSK recommendations for third COVID-19 vaccines, said that he has been thinking more broadly during the pandemic about the notion of immunocompetence. “It’s my opinion that patients with hematologic malignancies, even if they’re not on treatment, are not fully immune competent,” he said. This includes patients with CLL stage 0 and patients with plasma cell dyscrasias who don’t yet meet the criteria for multiple myeloma but have a monoclonal gammopathy, and those with lower-risk myelodysplastic syndromes, he said.
“We’re seeing [variable] recommendations based on expert opinion, and I think that’s justifiable in such a dynamic situation,” Dr. Yu said. “I would [even] argue it’s desirable so we can learn from different approaches” and collect more rigorous observational data.
Immunocompetence needs to be “viewed in the context of the threat,” he added. “COVID changes the equation. … What’s immunocompromised in my mind has changed [from prepandemic times].”
Preparing for measles
Measles lit up on Dr. Pergam’s radar screen in 2019, when an outbreak occurred in nearby Clark County, Wash. This and other outbreaks in New York, California, and other states highlighted declines in measles herd immunity in the United States and prompted him to investigate the seroprevalence of measles antibodies in the Fred Hutchinson Cancer Research Center’s outpatient population.
Of 959 consecutive patients seen at the center, they found, 25% lacked protective antibodies for measles. For patients with hematologic malignancies and those with a history of HCT, seroprevalence was worse: 37% and 54%, respectively, were without the IgG antibodies.4 Measles “is the most contagious human virus we have at the moment,” he said, and “revaccinating people is hard when it comes to cancer because it is a live virus vaccine.”
Vaccine hesitancy, a rise in nonmedical exemptions, and other factors were threatening herd immunity before the pandemic began. Now, with declines in routine childhood medical visits and other vaccination opportunities and resources here and in other countries – and declining immunization rates documented by the CDC in May 2021 – the pandemic has made measles outbreaks more likely, he said. (Measles outbreaks in West Africa on the tail end of the Ebola outbreak in 2014-2015 caused more deaths in children than Ebola, he noted.)
The first priority is vaccination “cocooning,” a strategy that has long been important for patients with hematologic malignancies. But it also possible, Dr. Pergam said, that in the setting of any future community transmission, revaccination for HCT recipients could occur earlier than the standard 2-year post-transplantation recommendation.
In a 2019 position statement endorsed by the American Society for Transplantation and Cellular Therapy, Dr. Pergam and other infectious disease physicians and oncologists provide criteria for considering early revaccination on a case-by-case basis for patients on minimal immunosuppressive therapy who are at least 1-year post transplantation.5
“Our thinking was that there may be lower-risk patients to whom we could offer the vaccine” – patients for whom the risk of developing measles might outweigh the risk of potential vaccine-related complications, he said.
And if there were community cases, he added, there might be a place for testing antibody levels in post-transplant patients, however imperfect the window to immunity may be. “We’re thinking through potential scenarios,” he said. “Oncologists should think about measles again and have it on their back burner.”
References
1. Grivas P et al. Ann Oncol. 2021 Jun;32(6):787-800.
2. Agha ME et al. Open Forum Infect Dis. 2021 July;8(7):ofab353.
3. Greenberger LM et al. Cancer Cell. 2021 Aug 9;39(8):1031-3.
4. Marquis SR et al. JAMA Netw Open. 2021 July;4(7):e2118508.
5. Pergam SA et al. Biol Blood Marrow Transplant. 2019 Nov;25:e321-30.
Myeloid patients respond robustly to Moderna COVID vaccine
Factors including age, gender, race, disease status, lower-intensity active treatment, baseline neutrophil and lymphocyte counts, and past history of stem cell transplant had no effects on seroconversion in the study, which, despite its small numbers, is one of the largest series to date among patients with myeloid cancers. The findings were reported at the annual meeting of the American Society of Hematology.
COVID vaccination “appears to induce a strong antibody response” in patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), unlike with B-cell malignancies. “It indicates we should be aggressive about vaccinating such patients,” said senior investigator Jeffrey Lancet, MD, a blood cancer specialist at Moffitt, when he presented the findings at the meeting.
Presentation moderator Laura Michaelis, MD, a hematologist-oncologist at the Medical College of Wisconsin, Milwaukee, agreed.
The “strong antibody response in this group,” coupled with its high risk for severe COVID, “confirm the importance of these patients getting vaccinated,” she said.
Thirty patients with AML and 16 with MDS were included in the review. Most patients were in remission at the time of vaccination, but a third were in active treatment, including six on hypomethylating agents, six on targeted therapies, two on luspatercept, and one on lenalidomide. Thirty-two patients (69.6%) were a median of 17 months past allogeneic stem cell transplant.
Overall, 69.6% of patients developed IgG against spike proteins after the first shot and 95.7% of patients after the second dose, with a large increase in titer levels from the first to the second dose, from a mean of 315 AU/mL to 3,806.5 AU/mL following the second dose.
“Lab and clinical variables did not affect the antibody positivity rate after the second dose,” but patients on steroids and other immunosuppressants seemed less likely to respond to the first shot, Dr. Lancet said.
The study, conducted in early 2021, did not include acutely ill patients or those undergoing cheomotherapy induction and other aggressive treatments, because such patients were not being vaccinated at Moffitt during the study period.
The investigators measured anti-spike IgG by ELISA at baseline, then again about a month after the first shot and a month after the second shot.
Side effects were common and typically mild, including injection site pain, fatigue, headache, and arm swelling. Two patients with AML relapsed after vaccination.
Patients were a median of 68 years old when they were vaccinated; 58.7% were men; and almost all of the subjects were White. The median time from diagnosis to the first shot was 2 years.
The next step in the project is to study the timing of vaccination and response to it among patients on aggressive treatment and to perform neutralizing antibody assays to correlate IgG response with protection from COVID.
No funding was reported for the study. Investigators had numerous industry ties, including Dr. Lancet, a consultant for Celgene/BMS, Millenium Pharma/Takeda, AbbVie, and other firms. Dr. Michaelis didn’t have any disclosures.
[email protected]
Factors including age, gender, race, disease status, lower-intensity active treatment, baseline neutrophil and lymphocyte counts, and past history of stem cell transplant had no effects on seroconversion in the study, which, despite its small numbers, is one of the largest series to date among patients with myeloid cancers. The findings were reported at the annual meeting of the American Society of Hematology.
COVID vaccination “appears to induce a strong antibody response” in patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), unlike with B-cell malignancies. “It indicates we should be aggressive about vaccinating such patients,” said senior investigator Jeffrey Lancet, MD, a blood cancer specialist at Moffitt, when he presented the findings at the meeting.
Presentation moderator Laura Michaelis, MD, a hematologist-oncologist at the Medical College of Wisconsin, Milwaukee, agreed.
The “strong antibody response in this group,” coupled with its high risk for severe COVID, “confirm the importance of these patients getting vaccinated,” she said.
Thirty patients with AML and 16 with MDS were included in the review. Most patients were in remission at the time of vaccination, but a third were in active treatment, including six on hypomethylating agents, six on targeted therapies, two on luspatercept, and one on lenalidomide. Thirty-two patients (69.6%) were a median of 17 months past allogeneic stem cell transplant.
Overall, 69.6% of patients developed IgG against spike proteins after the first shot and 95.7% of patients after the second dose, with a large increase in titer levels from the first to the second dose, from a mean of 315 AU/mL to 3,806.5 AU/mL following the second dose.
“Lab and clinical variables did not affect the antibody positivity rate after the second dose,” but patients on steroids and other immunosuppressants seemed less likely to respond to the first shot, Dr. Lancet said.
The study, conducted in early 2021, did not include acutely ill patients or those undergoing cheomotherapy induction and other aggressive treatments, because such patients were not being vaccinated at Moffitt during the study period.
The investigators measured anti-spike IgG by ELISA at baseline, then again about a month after the first shot and a month after the second shot.
Side effects were common and typically mild, including injection site pain, fatigue, headache, and arm swelling. Two patients with AML relapsed after vaccination.
Patients were a median of 68 years old when they were vaccinated; 58.7% were men; and almost all of the subjects were White. The median time from diagnosis to the first shot was 2 years.
The next step in the project is to study the timing of vaccination and response to it among patients on aggressive treatment and to perform neutralizing antibody assays to correlate IgG response with protection from COVID.
No funding was reported for the study. Investigators had numerous industry ties, including Dr. Lancet, a consultant for Celgene/BMS, Millenium Pharma/Takeda, AbbVie, and other firms. Dr. Michaelis didn’t have any disclosures.
[email protected]
Factors including age, gender, race, disease status, lower-intensity active treatment, baseline neutrophil and lymphocyte counts, and past history of stem cell transplant had no effects on seroconversion in the study, which, despite its small numbers, is one of the largest series to date among patients with myeloid cancers. The findings were reported at the annual meeting of the American Society of Hematology.
COVID vaccination “appears to induce a strong antibody response” in patients with acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), unlike with B-cell malignancies. “It indicates we should be aggressive about vaccinating such patients,” said senior investigator Jeffrey Lancet, MD, a blood cancer specialist at Moffitt, when he presented the findings at the meeting.
Presentation moderator Laura Michaelis, MD, a hematologist-oncologist at the Medical College of Wisconsin, Milwaukee, agreed.
The “strong antibody response in this group,” coupled with its high risk for severe COVID, “confirm the importance of these patients getting vaccinated,” she said.
Thirty patients with AML and 16 with MDS were included in the review. Most patients were in remission at the time of vaccination, but a third were in active treatment, including six on hypomethylating agents, six on targeted therapies, two on luspatercept, and one on lenalidomide. Thirty-two patients (69.6%) were a median of 17 months past allogeneic stem cell transplant.
Overall, 69.6% of patients developed IgG against spike proteins after the first shot and 95.7% of patients after the second dose, with a large increase in titer levels from the first to the second dose, from a mean of 315 AU/mL to 3,806.5 AU/mL following the second dose.
“Lab and clinical variables did not affect the antibody positivity rate after the second dose,” but patients on steroids and other immunosuppressants seemed less likely to respond to the first shot, Dr. Lancet said.
The study, conducted in early 2021, did not include acutely ill patients or those undergoing cheomotherapy induction and other aggressive treatments, because such patients were not being vaccinated at Moffitt during the study period.
The investigators measured anti-spike IgG by ELISA at baseline, then again about a month after the first shot and a month after the second shot.
Side effects were common and typically mild, including injection site pain, fatigue, headache, and arm swelling. Two patients with AML relapsed after vaccination.
Patients were a median of 68 years old when they were vaccinated; 58.7% were men; and almost all of the subjects were White. The median time from diagnosis to the first shot was 2 years.
The next step in the project is to study the timing of vaccination and response to it among patients on aggressive treatment and to perform neutralizing antibody assays to correlate IgG response with protection from COVID.
No funding was reported for the study. Investigators had numerous industry ties, including Dr. Lancet, a consultant for Celgene/BMS, Millenium Pharma/Takeda, AbbVie, and other firms. Dr. Michaelis didn’t have any disclosures.
[email protected]
FROM ASH 2021
For leukemias, COVID-19 death risks tied to poor prognoses, ICU deferrals
, results of an American Society of Hematology (ASH) COVID-19 registry study suggest.
Rates of severe COVID-19 were significantly higher among patients who had active disease or neutropenia at the time of their COVID-19 diagnosis. Mortality related to COVID-19 was linked to neutropenia, primary disease prognosis of less than 6 months, and deferral of recommended ICU care, study results show.
By contrast, mortality was not associated with active primary disease or its treatment, according to researcher Pinkal Desai, MD, MPH.
Taken together, these findings provide preliminary evidence to support the use of aggressive supportive treatment of COVID-19 in patients with acute leukemias and myelodysplastic syndromes, said Dr. Desai, a hematologist-oncologist with Weill Cornell Medicine and NewYork-Presbyterian in New York.
“If desired by patients, aggressive support for hospitalized patients with COVID-19 is appropriate, regardless of remission status, given the results of our study,” Dr. Desai said in a press conference during the annual meeting of the American Society of Hematology.
In non-cancer patient populations, advanced age and cytopenias have been associated with mortality related to COVID-19, Dr. Desai said. Likewise, patients with acute leukemias and myelodysplastic syndrome are generally older and have disease- or treatment-related cytopenias, which might affect the severity of and mortality from COVID-19, she added.
With that concern in mind, Dr. Desai and co-investigators looked at predictors of severe COVID-19 disease and death among patients in the ASH Research Collaborative (ASH RC) COVID-19 Registry for Hematology.
This registry was started in the early days of the pandemic to provide real-time observational COVID-19 data to clinicians, according to an ASH news release.
The analysis by Dr. Desai and co-authors included 257 patients with COVID-19 as determined by their physician, including 135 with a primary diagnosis of acute myeloid leukemia, 82 with acute lymphocytic leukemia, and 40 with myelodysplastic syndromes. Sixty percent of the patients were hospitalized due to COVID-19.
At the time of COVID-19 diagnosis, 46% of patients were in remission, and 44% had active disease, according to the report.
Both neutropenia and active disease status at COVID-19 diagnosis were linked to severe COVID-19, defined as ICU admission due to a COVID-19-related reason, according to results of multivariable analysis. Among patients with severe COVID-19, 67% had active disease, meaning just 33% were in remission, Dr. Desai noted.
In multivariable analysis, two factors were significantly associated with mortality, she added: having an estimated pre-COVID-19 prognosis from the primary disease of less than 6 months, and deferral of ICU care when it was recommended to the patient.
Mortality was 21% overall, higher than would be expected in a non-cancer population, Dr. Desai said. For patients with COVID-19 requiring hospitalization, the mortality rate was 34% and for those patients who did go to the ICU, the mortality rate was 68%.
By contrast, there was no significant association between mortality and active disease as compared to disease in remission, Dr. Desai noted in her presentation. Likewise, mortality was not associated with active treatment at the time of COVID-19 diagnosis as compared to no treatment.
Gwen Nichols, MD, executive vice president and chief medical officer of the Leukemia & Lymphoma Society, New York, said those are reassuring data for patients with acute leukemias and myelodysplastic syndromes and their healthcare providers.
“From our point of view, it helps us say, ‘do not stop your treatment because of worries about COVID-19—it’s more important that you treat your cancer,” Dr. Nichols said in an interview. “We now know we can help people through COVID-19, and I think this is just really important data to back that up,” she added.
Dr. Desai provided disclosures related to Agios, Kura Oncology, and Bristol Myers Squibb (consultancy), and to Janssen R&D and Astex (research funding).
, results of an American Society of Hematology (ASH) COVID-19 registry study suggest.
Rates of severe COVID-19 were significantly higher among patients who had active disease or neutropenia at the time of their COVID-19 diagnosis. Mortality related to COVID-19 was linked to neutropenia, primary disease prognosis of less than 6 months, and deferral of recommended ICU care, study results show.
By contrast, mortality was not associated with active primary disease or its treatment, according to researcher Pinkal Desai, MD, MPH.
Taken together, these findings provide preliminary evidence to support the use of aggressive supportive treatment of COVID-19 in patients with acute leukemias and myelodysplastic syndromes, said Dr. Desai, a hematologist-oncologist with Weill Cornell Medicine and NewYork-Presbyterian in New York.
“If desired by patients, aggressive support for hospitalized patients with COVID-19 is appropriate, regardless of remission status, given the results of our study,” Dr. Desai said in a press conference during the annual meeting of the American Society of Hematology.
In non-cancer patient populations, advanced age and cytopenias have been associated with mortality related to COVID-19, Dr. Desai said. Likewise, patients with acute leukemias and myelodysplastic syndrome are generally older and have disease- or treatment-related cytopenias, which might affect the severity of and mortality from COVID-19, she added.
With that concern in mind, Dr. Desai and co-investigators looked at predictors of severe COVID-19 disease and death among patients in the ASH Research Collaborative (ASH RC) COVID-19 Registry for Hematology.
This registry was started in the early days of the pandemic to provide real-time observational COVID-19 data to clinicians, according to an ASH news release.
The analysis by Dr. Desai and co-authors included 257 patients with COVID-19 as determined by their physician, including 135 with a primary diagnosis of acute myeloid leukemia, 82 with acute lymphocytic leukemia, and 40 with myelodysplastic syndromes. Sixty percent of the patients were hospitalized due to COVID-19.
At the time of COVID-19 diagnosis, 46% of patients were in remission, and 44% had active disease, according to the report.
Both neutropenia and active disease status at COVID-19 diagnosis were linked to severe COVID-19, defined as ICU admission due to a COVID-19-related reason, according to results of multivariable analysis. Among patients with severe COVID-19, 67% had active disease, meaning just 33% were in remission, Dr. Desai noted.
In multivariable analysis, two factors were significantly associated with mortality, she added: having an estimated pre-COVID-19 prognosis from the primary disease of less than 6 months, and deferral of ICU care when it was recommended to the patient.
Mortality was 21% overall, higher than would be expected in a non-cancer population, Dr. Desai said. For patients with COVID-19 requiring hospitalization, the mortality rate was 34% and for those patients who did go to the ICU, the mortality rate was 68%.
By contrast, there was no significant association between mortality and active disease as compared to disease in remission, Dr. Desai noted in her presentation. Likewise, mortality was not associated with active treatment at the time of COVID-19 diagnosis as compared to no treatment.
Gwen Nichols, MD, executive vice president and chief medical officer of the Leukemia & Lymphoma Society, New York, said those are reassuring data for patients with acute leukemias and myelodysplastic syndromes and their healthcare providers.
“From our point of view, it helps us say, ‘do not stop your treatment because of worries about COVID-19—it’s more important that you treat your cancer,” Dr. Nichols said in an interview. “We now know we can help people through COVID-19, and I think this is just really important data to back that up,” she added.
Dr. Desai provided disclosures related to Agios, Kura Oncology, and Bristol Myers Squibb (consultancy), and to Janssen R&D and Astex (research funding).
, results of an American Society of Hematology (ASH) COVID-19 registry study suggest.
Rates of severe COVID-19 were significantly higher among patients who had active disease or neutropenia at the time of their COVID-19 diagnosis. Mortality related to COVID-19 was linked to neutropenia, primary disease prognosis of less than 6 months, and deferral of recommended ICU care, study results show.
By contrast, mortality was not associated with active primary disease or its treatment, according to researcher Pinkal Desai, MD, MPH.
Taken together, these findings provide preliminary evidence to support the use of aggressive supportive treatment of COVID-19 in patients with acute leukemias and myelodysplastic syndromes, said Dr. Desai, a hematologist-oncologist with Weill Cornell Medicine and NewYork-Presbyterian in New York.
“If desired by patients, aggressive support for hospitalized patients with COVID-19 is appropriate, regardless of remission status, given the results of our study,” Dr. Desai said in a press conference during the annual meeting of the American Society of Hematology.
In non-cancer patient populations, advanced age and cytopenias have been associated with mortality related to COVID-19, Dr. Desai said. Likewise, patients with acute leukemias and myelodysplastic syndrome are generally older and have disease- or treatment-related cytopenias, which might affect the severity of and mortality from COVID-19, she added.
With that concern in mind, Dr. Desai and co-investigators looked at predictors of severe COVID-19 disease and death among patients in the ASH Research Collaborative (ASH RC) COVID-19 Registry for Hematology.
This registry was started in the early days of the pandemic to provide real-time observational COVID-19 data to clinicians, according to an ASH news release.
The analysis by Dr. Desai and co-authors included 257 patients with COVID-19 as determined by their physician, including 135 with a primary diagnosis of acute myeloid leukemia, 82 with acute lymphocytic leukemia, and 40 with myelodysplastic syndromes. Sixty percent of the patients were hospitalized due to COVID-19.
At the time of COVID-19 diagnosis, 46% of patients were in remission, and 44% had active disease, according to the report.
Both neutropenia and active disease status at COVID-19 diagnosis were linked to severe COVID-19, defined as ICU admission due to a COVID-19-related reason, according to results of multivariable analysis. Among patients with severe COVID-19, 67% had active disease, meaning just 33% were in remission, Dr. Desai noted.
In multivariable analysis, two factors were significantly associated with mortality, she added: having an estimated pre-COVID-19 prognosis from the primary disease of less than 6 months, and deferral of ICU care when it was recommended to the patient.
Mortality was 21% overall, higher than would be expected in a non-cancer population, Dr. Desai said. For patients with COVID-19 requiring hospitalization, the mortality rate was 34% and for those patients who did go to the ICU, the mortality rate was 68%.
By contrast, there was no significant association between mortality and active disease as compared to disease in remission, Dr. Desai noted in her presentation. Likewise, mortality was not associated with active treatment at the time of COVID-19 diagnosis as compared to no treatment.
Gwen Nichols, MD, executive vice president and chief medical officer of the Leukemia & Lymphoma Society, New York, said those are reassuring data for patients with acute leukemias and myelodysplastic syndromes and their healthcare providers.
“From our point of view, it helps us say, ‘do not stop your treatment because of worries about COVID-19—it’s more important that you treat your cancer,” Dr. Nichols said in an interview. “We now know we can help people through COVID-19, and I think this is just really important data to back that up,” she added.
Dr. Desai provided disclosures related to Agios, Kura Oncology, and Bristol Myers Squibb (consultancy), and to Janssen R&D and Astex (research funding).
FROM ASH 2021