User login
New urticaria guidelines stress simplicity
NEW YORK – New guidelines for the diagnosis and treatment of urticaria have been endorsed by 15 professional organizations so far and are now being prepared for publication, according to a consensus meeting participant who summarized key points at the American Academy of Dermatology summer meeting.
The guidelines, developed at an earlier conference held in Berlin attended by experts from 39 countries, are straightforward, relatively simple, "and truly developed for global application," according to Dr. Kiran Godse of Patil Medical College and Hospital, Navi Mumbai, India. The guidelines represent a joint initiative of the Dermatology Section of the European Academy of Allergology and Clinical Immunology (EAACI), the Global Allergy and Asthma European Network (GA2LEN), the European Dermatology Forum (EDF), the American Academy of Allergy, Asthma and Immunology (AAAAI), and the World Allergy Organization (WAO).
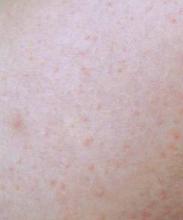
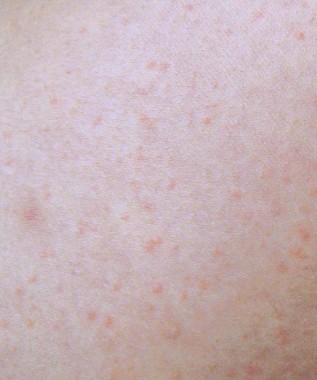
The simplicity of the guidelines starts with the definition of urticaria. It consists of three characteristics: "wheals, angioedema, or both." While the definition goes on to specify that these conditions should be differentiated from autoinflammatory syndromes, hereditary angioedema, and other diseases that produce hives or swelling, the new guidelines abandon the term "idiopathic."
"Our understanding of the etiology and pathogenesis has advanced to the point that we can identify the causes in most cases," said Dr. Godse, indicating that classifying cases as "idiopathic" without further investigation is unhelpful when the goal is to find and avoid triggers.
A number of subclassifications, such as spontaneous urticaria, inducible urticaria, acute urticaria, and chronic urticaria, are defined and employed to guide clinical management. In patients with acute urticaria, diagnostic testing beyond a careful history is not recommended, except when avoidance strategies fail and recurrences are common.
Even in chronic urticaria, which is defined as symptoms persisting for at least 6 months, Dr. Godse said that the guidelines recommend "limited" initial diagnostic studies.
By relying on careful patient history rather than clinical tests to differentiate the major forms of this disease, such as cold urticaria, heat urticaria, delayed pressure urticaria, solar urticaria, and symptomatic dermographism, the guidelines in effect propose that underlying etiologies do not usually require an extensive workup. However, the guidelines do advise more extensive tests in individuals with persistent and significant disease, which can be measured with the Chronic Urticaria Quality of Life (CU-QoL) and the Angioedema Quality of Life (Ae-QoL) instruments. Both are strongly recommended for baseline assessment of symptom burden.
The treatment goal of the stepwise management is clear: complete absence of symptoms. "Treat the disease until it is gone," said Dr. Godse, summarizing this recommendation.
If symptoms cannot be eliminated simply by avoiding causes and aggravating factors, the guidelines identify second-generation, nonsedating H1 antihistamines as the first-line pharmacotherapy. Dr. Godse said that the guidelines specifically recommend continuous rather than on-demand regimens at the lowest effective dose. However, if symptoms persist after 1-4 weeks of therapy, the dosing frequency should be increased before moving to adjunctive use of additional therapies. Adjunctive therapies listed in the guidelines include omalizumab, cyclosporine A, and montelukast. The first two of these options received strong recommendations on the basis of a high level of evidence, but the third was given a weak recommendation on the basis of a low level of evidence.
In those who fail these therapies, the list of alternatives is lengthy and includes a short course of corticosteroids, immunomodulating therapies such as methotrexate, and intravenous immunoglobulins. While any one of these may be useful in an individual patient, the overall evidence of benefit was considered to be of relatively low quality.
Ultimately, the guidelines attempt to define an approach that is uniformly applicable across diverse populations, a full range of possible etiologies, and within different systems of medical care, according to Dr. Godse.
Asked for their opinion after hearing the guidelines explained at the meeting, Dr. Paul Schneiderman and Dr. Aaron Warshawsky said they were favorably impressed. Both thought the guidelines were clear, reasonable, and potentially helpful in clinical practice. Dr. Schneiderman, an associate clinical professor of dermatology at Yale University, New Haven, Conn., who maintains a private practice in Syosset, N.Y., reported that he will be able to better judge the clinical applicability of the new guidelines when he sees the full publication, but both he and Dr. Warshawsky, a dermatologist in private practice in Poughkeepsie, N.Y., agreed that advances in urticaria justify updated guidelines.
Dr. Godse reported no financial disclosures relevant to his presentation.
NEW YORK – New guidelines for the diagnosis and treatment of urticaria have been endorsed by 15 professional organizations so far and are now being prepared for publication, according to a consensus meeting participant who summarized key points at the American Academy of Dermatology summer meeting.
The guidelines, developed at an earlier conference held in Berlin attended by experts from 39 countries, are straightforward, relatively simple, "and truly developed for global application," according to Dr. Kiran Godse of Patil Medical College and Hospital, Navi Mumbai, India. The guidelines represent a joint initiative of the Dermatology Section of the European Academy of Allergology and Clinical Immunology (EAACI), the Global Allergy and Asthma European Network (GA2LEN), the European Dermatology Forum (EDF), the American Academy of Allergy, Asthma and Immunology (AAAAI), and the World Allergy Organization (WAO).
The simplicity of the guidelines starts with the definition of urticaria. It consists of three characteristics: "wheals, angioedema, or both." While the definition goes on to specify that these conditions should be differentiated from autoinflammatory syndromes, hereditary angioedema, and other diseases that produce hives or swelling, the new guidelines abandon the term "idiopathic."
"Our understanding of the etiology and pathogenesis has advanced to the point that we can identify the causes in most cases," said Dr. Godse, indicating that classifying cases as "idiopathic" without further investigation is unhelpful when the goal is to find and avoid triggers.
A number of subclassifications, such as spontaneous urticaria, inducible urticaria, acute urticaria, and chronic urticaria, are defined and employed to guide clinical management. In patients with acute urticaria, diagnostic testing beyond a careful history is not recommended, except when avoidance strategies fail and recurrences are common.
Even in chronic urticaria, which is defined as symptoms persisting for at least 6 months, Dr. Godse said that the guidelines recommend "limited" initial diagnostic studies.
By relying on careful patient history rather than clinical tests to differentiate the major forms of this disease, such as cold urticaria, heat urticaria, delayed pressure urticaria, solar urticaria, and symptomatic dermographism, the guidelines in effect propose that underlying etiologies do not usually require an extensive workup. However, the guidelines do advise more extensive tests in individuals with persistent and significant disease, which can be measured with the Chronic Urticaria Quality of Life (CU-QoL) and the Angioedema Quality of Life (Ae-QoL) instruments. Both are strongly recommended for baseline assessment of symptom burden.
The treatment goal of the stepwise management is clear: complete absence of symptoms. "Treat the disease until it is gone," said Dr. Godse, summarizing this recommendation.
If symptoms cannot be eliminated simply by avoiding causes and aggravating factors, the guidelines identify second-generation, nonsedating H1 antihistamines as the first-line pharmacotherapy. Dr. Godse said that the guidelines specifically recommend continuous rather than on-demand regimens at the lowest effective dose. However, if symptoms persist after 1-4 weeks of therapy, the dosing frequency should be increased before moving to adjunctive use of additional therapies. Adjunctive therapies listed in the guidelines include omalizumab, cyclosporine A, and montelukast. The first two of these options received strong recommendations on the basis of a high level of evidence, but the third was given a weak recommendation on the basis of a low level of evidence.
In those who fail these therapies, the list of alternatives is lengthy and includes a short course of corticosteroids, immunomodulating therapies such as methotrexate, and intravenous immunoglobulins. While any one of these may be useful in an individual patient, the overall evidence of benefit was considered to be of relatively low quality.
Ultimately, the guidelines attempt to define an approach that is uniformly applicable across diverse populations, a full range of possible etiologies, and within different systems of medical care, according to Dr. Godse.
Asked for their opinion after hearing the guidelines explained at the meeting, Dr. Paul Schneiderman and Dr. Aaron Warshawsky said they were favorably impressed. Both thought the guidelines were clear, reasonable, and potentially helpful in clinical practice. Dr. Schneiderman, an associate clinical professor of dermatology at Yale University, New Haven, Conn., who maintains a private practice in Syosset, N.Y., reported that he will be able to better judge the clinical applicability of the new guidelines when he sees the full publication, but both he and Dr. Warshawsky, a dermatologist in private practice in Poughkeepsie, N.Y., agreed that advances in urticaria justify updated guidelines.
Dr. Godse reported no financial disclosures relevant to his presentation.
NEW YORK – New guidelines for the diagnosis and treatment of urticaria have been endorsed by 15 professional organizations so far and are now being prepared for publication, according to a consensus meeting participant who summarized key points at the American Academy of Dermatology summer meeting.
The guidelines, developed at an earlier conference held in Berlin attended by experts from 39 countries, are straightforward, relatively simple, "and truly developed for global application," according to Dr. Kiran Godse of Patil Medical College and Hospital, Navi Mumbai, India. The guidelines represent a joint initiative of the Dermatology Section of the European Academy of Allergology and Clinical Immunology (EAACI), the Global Allergy and Asthma European Network (GA2LEN), the European Dermatology Forum (EDF), the American Academy of Allergy, Asthma and Immunology (AAAAI), and the World Allergy Organization (WAO).
The simplicity of the guidelines starts with the definition of urticaria. It consists of three characteristics: "wheals, angioedema, or both." While the definition goes on to specify that these conditions should be differentiated from autoinflammatory syndromes, hereditary angioedema, and other diseases that produce hives or swelling, the new guidelines abandon the term "idiopathic."
"Our understanding of the etiology and pathogenesis has advanced to the point that we can identify the causes in most cases," said Dr. Godse, indicating that classifying cases as "idiopathic" without further investigation is unhelpful when the goal is to find and avoid triggers.
A number of subclassifications, such as spontaneous urticaria, inducible urticaria, acute urticaria, and chronic urticaria, are defined and employed to guide clinical management. In patients with acute urticaria, diagnostic testing beyond a careful history is not recommended, except when avoidance strategies fail and recurrences are common.
Even in chronic urticaria, which is defined as symptoms persisting for at least 6 months, Dr. Godse said that the guidelines recommend "limited" initial diagnostic studies.
By relying on careful patient history rather than clinical tests to differentiate the major forms of this disease, such as cold urticaria, heat urticaria, delayed pressure urticaria, solar urticaria, and symptomatic dermographism, the guidelines in effect propose that underlying etiologies do not usually require an extensive workup. However, the guidelines do advise more extensive tests in individuals with persistent and significant disease, which can be measured with the Chronic Urticaria Quality of Life (CU-QoL) and the Angioedema Quality of Life (Ae-QoL) instruments. Both are strongly recommended for baseline assessment of symptom burden.
The treatment goal of the stepwise management is clear: complete absence of symptoms. "Treat the disease until it is gone," said Dr. Godse, summarizing this recommendation.
If symptoms cannot be eliminated simply by avoiding causes and aggravating factors, the guidelines identify second-generation, nonsedating H1 antihistamines as the first-line pharmacotherapy. Dr. Godse said that the guidelines specifically recommend continuous rather than on-demand regimens at the lowest effective dose. However, if symptoms persist after 1-4 weeks of therapy, the dosing frequency should be increased before moving to adjunctive use of additional therapies. Adjunctive therapies listed in the guidelines include omalizumab, cyclosporine A, and montelukast. The first two of these options received strong recommendations on the basis of a high level of evidence, but the third was given a weak recommendation on the basis of a low level of evidence.
In those who fail these therapies, the list of alternatives is lengthy and includes a short course of corticosteroids, immunomodulating therapies such as methotrexate, and intravenous immunoglobulins. While any one of these may be useful in an individual patient, the overall evidence of benefit was considered to be of relatively low quality.
Ultimately, the guidelines attempt to define an approach that is uniformly applicable across diverse populations, a full range of possible etiologies, and within different systems of medical care, according to Dr. Godse.
Asked for their opinion after hearing the guidelines explained at the meeting, Dr. Paul Schneiderman and Dr. Aaron Warshawsky said they were favorably impressed. Both thought the guidelines were clear, reasonable, and potentially helpful in clinical practice. Dr. Schneiderman, an associate clinical professor of dermatology at Yale University, New Haven, Conn., who maintains a private practice in Syosset, N.Y., reported that he will be able to better judge the clinical applicability of the new guidelines when he sees the full publication, but both he and Dr. Warshawsky, a dermatologist in private practice in Poughkeepsie, N.Y., agreed that advances in urticaria justify updated guidelines.
Dr. Godse reported no financial disclosures relevant to his presentation.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2013
Epidermolysis bullosa patients rate itching worse than pain
MILWAUKEE – Itching is more problematic than is pain for patients with epidermolysis bullosa, according to an online survey of 146 patients.
Symptoms reported in conjunction with itching included pain, stinging, burning, and a sensation of ants crawling on the skin, as well as a feeling that the itch was too deep to scratch, Christina Danial and her associates reported at the annual meeting of the Society for Pediatric Dermatology.
Although data from a prior study of 11 children with epidermolysis bullosa (EB) hinted that pruritus is more bothersome than pain is (Acta. Derm. Venereol. 2008;88:143-50), the results are nonetheless startling given that EB is a mutilating disease of the skin and mucosa, Ms. Danial said. Patients with this rare condition cope with blistering of the skin and/or epithelial lining of the organs, GI complications, anemia, and shortened survival due to infection or cancer.
In this study, 216 EB patients and/or caregivers registered in the Epidermolysis Bullosa Clinical Research Consortium received a 42-item online questionnaire, and 146 completed the survey. Responses were based on a 5-point Likert scale in which 1 was "never" and 5 was "always."
Itchiness was rated the most bothersome item (mean 3.3), followed by acute pain (mean 2.9), chronic pain (mean 2.7), problems eating (mean 2.7), stomach problems (mean 2.6), dental problems (mean 2.6), movement (2.5), and surgical procedures (mean 2.4), reported Ms. Danial, a medical student at Stanford (Calif.) University.
Patients with the more severe recessive dystrophic EB subtype had significantly more pruritus than did those with EB simplex (mean 3.9 vs. 3.1; P = .01).
The frequency of itching increased as the day progressed, with bedtime having the highest frequency (mean 3.8). Not surprisingly, itching was found to interfere with sleep (mean 3.1), she noted.
Sweating (mean 3.9) and stress (mean 4.0) increased itching, as did dryness (mean 4.0), heat (mean 3.8), and humidity (mean 3.5).
The 146 respondents comprised 90 patients, 36 caregivers, and 20 patient/caregivers. The average age of the respondents was 22 years; 73 were male and 73 were female.
EB patients reported that pruritus makes them feel frustrated, agitated, and as if they cannot control the itch, although activity was noted to relieve the itching, Ms. Danial reported.
Ms. Danial suggested that further investigation is necessary to determine which treatments are most effective against itching in EB, but recommended that patients avoid dryness, heat, and humidity, and consider nighttime treatment. Treatments to control pain also may help alleviate pruritus, since patients may experience pain while itching, she added.
In all, 66% of patients had dystrophic EB, 21% had EB simplex, 10% had junctional EB, and the EB subtype of 3% was unknown. Severe disease was reported in 38% of those with recessive dystrophic EB, 14% with dominant dystrophic EB, 17% with unknown dystrophic subtype, 3% with EB simplex, 36% with junctional EB, and none with unknown EB.
Ms. Danial and her coauthors reported having no financial disclosures.
MILWAUKEE – Itching is more problematic than is pain for patients with epidermolysis bullosa, according to an online survey of 146 patients.
Symptoms reported in conjunction with itching included pain, stinging, burning, and a sensation of ants crawling on the skin, as well as a feeling that the itch was too deep to scratch, Christina Danial and her associates reported at the annual meeting of the Society for Pediatric Dermatology.
Although data from a prior study of 11 children with epidermolysis bullosa (EB) hinted that pruritus is more bothersome than pain is (Acta. Derm. Venereol. 2008;88:143-50), the results are nonetheless startling given that EB is a mutilating disease of the skin and mucosa, Ms. Danial said. Patients with this rare condition cope with blistering of the skin and/or epithelial lining of the organs, GI complications, anemia, and shortened survival due to infection or cancer.
In this study, 216 EB patients and/or caregivers registered in the Epidermolysis Bullosa Clinical Research Consortium received a 42-item online questionnaire, and 146 completed the survey. Responses were based on a 5-point Likert scale in which 1 was "never" and 5 was "always."
Itchiness was rated the most bothersome item (mean 3.3), followed by acute pain (mean 2.9), chronic pain (mean 2.7), problems eating (mean 2.7), stomach problems (mean 2.6), dental problems (mean 2.6), movement (2.5), and surgical procedures (mean 2.4), reported Ms. Danial, a medical student at Stanford (Calif.) University.
Patients with the more severe recessive dystrophic EB subtype had significantly more pruritus than did those with EB simplex (mean 3.9 vs. 3.1; P = .01).
The frequency of itching increased as the day progressed, with bedtime having the highest frequency (mean 3.8). Not surprisingly, itching was found to interfere with sleep (mean 3.1), she noted.
Sweating (mean 3.9) and stress (mean 4.0) increased itching, as did dryness (mean 4.0), heat (mean 3.8), and humidity (mean 3.5).
The 146 respondents comprised 90 patients, 36 caregivers, and 20 patient/caregivers. The average age of the respondents was 22 years; 73 were male and 73 were female.
EB patients reported that pruritus makes them feel frustrated, agitated, and as if they cannot control the itch, although activity was noted to relieve the itching, Ms. Danial reported.
Ms. Danial suggested that further investigation is necessary to determine which treatments are most effective against itching in EB, but recommended that patients avoid dryness, heat, and humidity, and consider nighttime treatment. Treatments to control pain also may help alleviate pruritus, since patients may experience pain while itching, she added.
In all, 66% of patients had dystrophic EB, 21% had EB simplex, 10% had junctional EB, and the EB subtype of 3% was unknown. Severe disease was reported in 38% of those with recessive dystrophic EB, 14% with dominant dystrophic EB, 17% with unknown dystrophic subtype, 3% with EB simplex, 36% with junctional EB, and none with unknown EB.
Ms. Danial and her coauthors reported having no financial disclosures.
MILWAUKEE – Itching is more problematic than is pain for patients with epidermolysis bullosa, according to an online survey of 146 patients.
Symptoms reported in conjunction with itching included pain, stinging, burning, and a sensation of ants crawling on the skin, as well as a feeling that the itch was too deep to scratch, Christina Danial and her associates reported at the annual meeting of the Society for Pediatric Dermatology.
Although data from a prior study of 11 children with epidermolysis bullosa (EB) hinted that pruritus is more bothersome than pain is (Acta. Derm. Venereol. 2008;88:143-50), the results are nonetheless startling given that EB is a mutilating disease of the skin and mucosa, Ms. Danial said. Patients with this rare condition cope with blistering of the skin and/or epithelial lining of the organs, GI complications, anemia, and shortened survival due to infection or cancer.
In this study, 216 EB patients and/or caregivers registered in the Epidermolysis Bullosa Clinical Research Consortium received a 42-item online questionnaire, and 146 completed the survey. Responses were based on a 5-point Likert scale in which 1 was "never" and 5 was "always."
Itchiness was rated the most bothersome item (mean 3.3), followed by acute pain (mean 2.9), chronic pain (mean 2.7), problems eating (mean 2.7), stomach problems (mean 2.6), dental problems (mean 2.6), movement (2.5), and surgical procedures (mean 2.4), reported Ms. Danial, a medical student at Stanford (Calif.) University.
Patients with the more severe recessive dystrophic EB subtype had significantly more pruritus than did those with EB simplex (mean 3.9 vs. 3.1; P = .01).
The frequency of itching increased as the day progressed, with bedtime having the highest frequency (mean 3.8). Not surprisingly, itching was found to interfere with sleep (mean 3.1), she noted.
Sweating (mean 3.9) and stress (mean 4.0) increased itching, as did dryness (mean 4.0), heat (mean 3.8), and humidity (mean 3.5).
The 146 respondents comprised 90 patients, 36 caregivers, and 20 patient/caregivers. The average age of the respondents was 22 years; 73 were male and 73 were female.
EB patients reported that pruritus makes them feel frustrated, agitated, and as if they cannot control the itch, although activity was noted to relieve the itching, Ms. Danial reported.
Ms. Danial suggested that further investigation is necessary to determine which treatments are most effective against itching in EB, but recommended that patients avoid dryness, heat, and humidity, and consider nighttime treatment. Treatments to control pain also may help alleviate pruritus, since patients may experience pain while itching, she added.
In all, 66% of patients had dystrophic EB, 21% had EB simplex, 10% had junctional EB, and the EB subtype of 3% was unknown. Severe disease was reported in 38% of those with recessive dystrophic EB, 14% with dominant dystrophic EB, 17% with unknown dystrophic subtype, 3% with EB simplex, 36% with junctional EB, and none with unknown EB.
Ms. Danial and her coauthors reported having no financial disclosures.
AT THE SPD ANNUAL MEETING
Major finding: Itchiness was rated the most bothersome symptom (mean 3.3 on a 5-point scale), followed by acute pain (mean 2.9), chronic pain (mean 2.7), and problems eating (mean 2.7).
Data source: Online survey of 146 patients with epidermolysis bullosa and/or their caregivers.
Disclosures: Ms. Danial and her coauthors reported having no financial disclosures.
Methotrexate assay useful in severe pediatric dermatitis, psoriasis
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
MILWAUKEE – In children with severe atopic dermatitis or psoriasis, the methotrexate polyglutamate assay appears useful in guiding dose modification in those failing to respond to methotrexate at 12 weeks, a retrospective study has shown.
The methotrexate polyglutamate 3 assay (MTX PG3) measures concentrations of methotrexate polyglutamates, the active metabolites of methotrexate, Syed Rahman said at the annual meeting of the Society for Pediatric Dermatology.
Methotrexate, a prodrug, requires enzymatic conversion via sequential addition of glutamic acid residues. Those residues form a glutamate "tail" that enhances intracellular methotrexate retention. The 3-tail metabolite, MTX PG3, is the predominant species in most patients, explained Mr. Rahman of St. Louis University.
"This study supports the usefulness of a commercially available tool for customizing optimal methotrexate dosing, coauthor Dr. Elaine Siegfried, professor of pediatrics and dermatology at the university, said in an interview.
The MTX PG3 assay has been validated in adults with rheumatoid arthritis where patients with a therapeutic methotrexate polyglutamate level of more than 60 nmol/L were fivefold more likely to have a good response to methotrexate than those with levels below this threshold, according to poster results presented at the 2008 American College of Rheumatology annual meeting (cited on the company website). However, the test was not predictive of response in 36% of patients whose arthritis improved with treatment despite subtherapeutic levels.
For the current analysis, the investigators reviewed the charts of 46 children with severe atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap, who were treated with a starting dose of oral methotrexate 0.5 mg/kg per week (maximum, 15 mg) and assessed by MTX PG3 assay at week 12. In a subset of children who failed to respond, the dose was modified and the level rechecked at 8 weeks. Disease severity was rated using a 5-point physician global assessment scale, with a good to excellent response defined by a 2-point improvement.
In all, 83% of children achieved a good to excellent response, with 27 patients doing so within 12 weeks and 11 responding after dose adjustment involving either a higher oral dose or switching to subcutaneous administration, Mr. Rahman reported.
Response rates to methotrexate were slightly higher in children with psoriasis or overlap (the two groups were combined) than those with atopic dermatitis (94% vs. 77%; no P value given). Overall, 8 children failed to respond.
The mean MTX PG3 level significantly correlated with efficacy in patients with atopic dermatitis (responders, 34.8 nmol/L, vs. 17.8 nmol/L, nonresponders; P = .018), but not in those with psoriasis–atopic dermatitis overlap (responders, 26.5 nmol/L, vs. 20.7, nonresponders; P = .724), Mr. Rahman reported.
The data suggest that compared with the therapeutic level of 60 nmol/L identified in adults, "pediatric patients have a lower therapeutic value," he said.
A subset analysis of MTX PG3 levels based on response identified a significantly higher mean maximum MTX PG3 level for responders than nonresponders (31.5 vs. 18.1 nmol/L; P = .035).
More importantly, late responders had the highest methotrexate levels. All late responders achieved levels greater than 30 nmol/L, and mean levels were significantly higher than for early responders (41.9 vs. 27.3 nmol/L; P = .024), suggesting that the test is most valuable for the subset of children requiring dose adjustment, Mr. Rahman said.
"Patients who respond won’t even need the test, but for those at the 12-week juncture who do not respond and their levels are less than 30 nmol/L, this [study] suggests that if we increase the PO [dose] or change to subcutaneous [administration], we will eventually have a response," he said.
During a discussion of the results, an audience member questioned whether the test, with its added expense, really enhances clinical judgment because most clinicians would already adjust the dose in their patients failing to respond at 12 weeks.
Mr. Rahman replied that not all patients will respond to continued treatment with methotrexate, and that the test could differentiate between responders and nonresponders, thereby directing the patient to alternative therapy earlier.
Session moderator Dr. Amy Paller, chair of dermatology and professor of pediatrics and dermatology at Northwestern University in Chicago, agreed that the test is most valuable for the subset of children who are nonresponders at 12 weeks.
"In these children, this test can guide dose modification," she said in an interview. "Pharmacogenetic variation can require relatively higher oral doses or subcutaneous administration."
In contrast to a therapeutic level of more than 60 nmol/L established for adults, a level of 30 nmol/L may be sufficient in children, Dr. Paller said.
"A MTX PG3 less than 30 nmol/L in a child who has failed to respond within 12 weeks suggests that an increase in oral dose, or even a change to subcutaneous [administration], will boost efficacy," she commented.
Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE SOCIETY FOR PEDIATRIC DERMATOLOGY
Major finding: Late responders had the highest methotrexate levels (41.9 nmol/L vs. 27.3 nmol/L in early responders; P = .024) on the MTX PG3 assay.
Data source: A retrospective study of 46 children on methotrexate for atopic dermatitis, psoriasis, or atopic dermatitis–psoriasis overlap who were assessed by MTX PG3 assay.
Disclosures: Mr. Rahman, Dr. Siegfried, and Dr. Paller reported having no relevant financial disclosures.
Modern Moisturizer Myths, Misconceptions, and Truths
Reduced Burning and Stinging Associated With Topical Application of Lactic Acid 10% With Strontium Versus Ammonium Lactate 12%
Dupilumab cuts moderate, severe asthma exacerbations
Dupilumab reduced exacerbations of moderate to severe asthma by 87% in adults with poorly controlled disease, and induced rapid and sustained improvements in numerous other measures of asthma severity in an industry-sponsored phase II study published online May 21 in the New England Journal of Medicine.
Dupilumab, a monoclonal antibody that inhibits interleukin-4 and interleukin-13 signaling, "showed substantial efficacy with regard to both objective and patient-reported end points" when it was used concomitantly with inhaled glucocorticoids and long-acting beta-agonists (LABAs), as well as when those background therapies were withdrawn, reported Dr. Sally Wenzel of the University of Pittsburgh and her associates.
"The magnitude and breadth of efficacy that we observed exceed those in other studies of Th2 [type 2 helper T-cell] cytokine inhibition," the researchers noted in the online report, which was presented simultaneously at the annual meeting of the American Thoracic Society.
Dupilumab currently is being assessed for the treatment of several diseases mediated by Th2 pathways. The goal of this phase II trial was to evaluate its safety and efficacy in adults with persistent moderate to severe asthma and elevated eosinophil levels whose symptoms were not well controlled with medium- to high-dose inhaled glucocorticoids plus LABAs (usually fluticasone and salmeterol).
The 104 participants were treated at 28 sites across the United States for 12 weeks, and then followed for another 8 weeks. During the intervention phase of the study, approximately half were randomized to receive once-weekly subcutaneous injections of dupilumab (300 mg) and half to receive matching placebo injections, in addition to the background asthma medications.
At week 4, the study subjects discontinued LABAs, and at weeks 6-9 they tapered off inhaled glucocorticoids. "This approach enabled us to observe the effects of dupilumab when added to background therapy, after LABA discontinuation, during the tapering of inhaled glucocorticoids, and as monotherapy," the researchers said.
The primary endpoint of the study was an asthma exacerbation during the 12-week intervention period. Exacerbations occurred in 3 patients receiving dupilumab (6%), compared with 23 receiving placebo (44%), a highly significant difference.
In addition, the time to an asthma exacerbation was significantly longer with dupilumab than placebo. And forced expiratory volume in 1 second (FEV1) "improved by more than 200 mL when dupilumab, as compared with placebo, was added to inhaled glucocorticoids and LABAs, an increase sustained during their tapering and discontinuation," the researchers noted (N. Engl. J. Med. 2013 May 21 [doi: 10.1056/NEJMoa1304048]).
Several other secondary endpoints also favored dupilumab over placebo, including morning peak expiratory flow values, scores on the Asthma Control Questionnaire (ACQ5), morning and evening asthma symptom scores, and the number of albuterol or levalbuterol inhalations needed per day. These measures improved at the beginning of the intervention in both study groups, then quickly returned to baseline levels in the placebo group while remaining constant in the dupilumab group.
The percentage of patients reporting adverse events was similar between the dupilumab and placebo groups (81% vs. 77%). These events tended to be nonspecific and mild, and included nasopharyngitis, nausea, and headache. One patient developed a progressive papular rash, urticaria, and edema after his ninth injection of dupilumab, which responded to nonurgent treatment of the symptoms and did not recur once the drug was withdrawn.
No serious adverse events were attributed to the study drug, and no patient showed clinically significant changes in vital signs, findings on physical examination, laboratory tests, or ECGs.
"Further studies are needed to confirm these observations and better define the target population, dosing regimen, and long-term efficacy and safety," Dr. Wenzel noted.
However, the study findings support the theory that the Th2 cytokines interleukin-4 and interleukin-13 play a pathogenic role in persistent, moderate-to-severe asthma, she added.
This study was funded by Sanofi and Regeneron Pharmaceuticals. Dr. Wenzel reported ties to Sanofi, Regeneron, Amgen, Merck, and other companies. Her associates reported ties to numerous industry sources.
Dupilumab reduced exacerbations of moderate to severe asthma by 87% in adults with poorly controlled disease, and induced rapid and sustained improvements in numerous other measures of asthma severity in an industry-sponsored phase II study published online May 21 in the New England Journal of Medicine.
Dupilumab, a monoclonal antibody that inhibits interleukin-4 and interleukin-13 signaling, "showed substantial efficacy with regard to both objective and patient-reported end points" when it was used concomitantly with inhaled glucocorticoids and long-acting beta-agonists (LABAs), as well as when those background therapies were withdrawn, reported Dr. Sally Wenzel of the University of Pittsburgh and her associates.
"The magnitude and breadth of efficacy that we observed exceed those in other studies of Th2 [type 2 helper T-cell] cytokine inhibition," the researchers noted in the online report, which was presented simultaneously at the annual meeting of the American Thoracic Society.
Dupilumab currently is being assessed for the treatment of several diseases mediated by Th2 pathways. The goal of this phase II trial was to evaluate its safety and efficacy in adults with persistent moderate to severe asthma and elevated eosinophil levels whose symptoms were not well controlled with medium- to high-dose inhaled glucocorticoids plus LABAs (usually fluticasone and salmeterol).
The 104 participants were treated at 28 sites across the United States for 12 weeks, and then followed for another 8 weeks. During the intervention phase of the study, approximately half were randomized to receive once-weekly subcutaneous injections of dupilumab (300 mg) and half to receive matching placebo injections, in addition to the background asthma medications.
At week 4, the study subjects discontinued LABAs, and at weeks 6-9 they tapered off inhaled glucocorticoids. "This approach enabled us to observe the effects of dupilumab when added to background therapy, after LABA discontinuation, during the tapering of inhaled glucocorticoids, and as monotherapy," the researchers said.
The primary endpoint of the study was an asthma exacerbation during the 12-week intervention period. Exacerbations occurred in 3 patients receiving dupilumab (6%), compared with 23 receiving placebo (44%), a highly significant difference.
In addition, the time to an asthma exacerbation was significantly longer with dupilumab than placebo. And forced expiratory volume in 1 second (FEV1) "improved by more than 200 mL when dupilumab, as compared with placebo, was added to inhaled glucocorticoids and LABAs, an increase sustained during their tapering and discontinuation," the researchers noted (N. Engl. J. Med. 2013 May 21 [doi: 10.1056/NEJMoa1304048]).
Several other secondary endpoints also favored dupilumab over placebo, including morning peak expiratory flow values, scores on the Asthma Control Questionnaire (ACQ5), morning and evening asthma symptom scores, and the number of albuterol or levalbuterol inhalations needed per day. These measures improved at the beginning of the intervention in both study groups, then quickly returned to baseline levels in the placebo group while remaining constant in the dupilumab group.
The percentage of patients reporting adverse events was similar between the dupilumab and placebo groups (81% vs. 77%). These events tended to be nonspecific and mild, and included nasopharyngitis, nausea, and headache. One patient developed a progressive papular rash, urticaria, and edema after his ninth injection of dupilumab, which responded to nonurgent treatment of the symptoms and did not recur once the drug was withdrawn.
No serious adverse events were attributed to the study drug, and no patient showed clinically significant changes in vital signs, findings on physical examination, laboratory tests, or ECGs.
"Further studies are needed to confirm these observations and better define the target population, dosing regimen, and long-term efficacy and safety," Dr. Wenzel noted.
However, the study findings support the theory that the Th2 cytokines interleukin-4 and interleukin-13 play a pathogenic role in persistent, moderate-to-severe asthma, she added.
This study was funded by Sanofi and Regeneron Pharmaceuticals. Dr. Wenzel reported ties to Sanofi, Regeneron, Amgen, Merck, and other companies. Her associates reported ties to numerous industry sources.
Dupilumab reduced exacerbations of moderate to severe asthma by 87% in adults with poorly controlled disease, and induced rapid and sustained improvements in numerous other measures of asthma severity in an industry-sponsored phase II study published online May 21 in the New England Journal of Medicine.
Dupilumab, a monoclonal antibody that inhibits interleukin-4 and interleukin-13 signaling, "showed substantial efficacy with regard to both objective and patient-reported end points" when it was used concomitantly with inhaled glucocorticoids and long-acting beta-agonists (LABAs), as well as when those background therapies were withdrawn, reported Dr. Sally Wenzel of the University of Pittsburgh and her associates.
"The magnitude and breadth of efficacy that we observed exceed those in other studies of Th2 [type 2 helper T-cell] cytokine inhibition," the researchers noted in the online report, which was presented simultaneously at the annual meeting of the American Thoracic Society.
Dupilumab currently is being assessed for the treatment of several diseases mediated by Th2 pathways. The goal of this phase II trial was to evaluate its safety and efficacy in adults with persistent moderate to severe asthma and elevated eosinophil levels whose symptoms were not well controlled with medium- to high-dose inhaled glucocorticoids plus LABAs (usually fluticasone and salmeterol).
The 104 participants were treated at 28 sites across the United States for 12 weeks, and then followed for another 8 weeks. During the intervention phase of the study, approximately half were randomized to receive once-weekly subcutaneous injections of dupilumab (300 mg) and half to receive matching placebo injections, in addition to the background asthma medications.
At week 4, the study subjects discontinued LABAs, and at weeks 6-9 they tapered off inhaled glucocorticoids. "This approach enabled us to observe the effects of dupilumab when added to background therapy, after LABA discontinuation, during the tapering of inhaled glucocorticoids, and as monotherapy," the researchers said.
The primary endpoint of the study was an asthma exacerbation during the 12-week intervention period. Exacerbations occurred in 3 patients receiving dupilumab (6%), compared with 23 receiving placebo (44%), a highly significant difference.
In addition, the time to an asthma exacerbation was significantly longer with dupilumab than placebo. And forced expiratory volume in 1 second (FEV1) "improved by more than 200 mL when dupilumab, as compared with placebo, was added to inhaled glucocorticoids and LABAs, an increase sustained during their tapering and discontinuation," the researchers noted (N. Engl. J. Med. 2013 May 21 [doi: 10.1056/NEJMoa1304048]).
Several other secondary endpoints also favored dupilumab over placebo, including morning peak expiratory flow values, scores on the Asthma Control Questionnaire (ACQ5), morning and evening asthma symptom scores, and the number of albuterol or levalbuterol inhalations needed per day. These measures improved at the beginning of the intervention in both study groups, then quickly returned to baseline levels in the placebo group while remaining constant in the dupilumab group.
The percentage of patients reporting adverse events was similar between the dupilumab and placebo groups (81% vs. 77%). These events tended to be nonspecific and mild, and included nasopharyngitis, nausea, and headache. One patient developed a progressive papular rash, urticaria, and edema after his ninth injection of dupilumab, which responded to nonurgent treatment of the symptoms and did not recur once the drug was withdrawn.
No serious adverse events were attributed to the study drug, and no patient showed clinically significant changes in vital signs, findings on physical examination, laboratory tests, or ECGs.
"Further studies are needed to confirm these observations and better define the target population, dosing regimen, and long-term efficacy and safety," Dr. Wenzel noted.
However, the study findings support the theory that the Th2 cytokines interleukin-4 and interleukin-13 play a pathogenic role in persistent, moderate-to-severe asthma, she added.
This study was funded by Sanofi and Regeneron Pharmaceuticals. Dr. Wenzel reported ties to Sanofi, Regeneron, Amgen, Merck, and other companies. Her associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Dupilumab was associated with an 87% reduction in asthma exacerbation; three patients receiving dupilumab (6%) had an exacerbation during the study, compared with 23 receiving placebo (44%).
Data Source: A randomized, double-blind, phase II clinical trial including 104 adults with persistent moderate to severe asthma.
Disclosures: This study was funded by Sanofi and Regeneron Pharmaceuticals. Dr. Wenzel reported ties to Sanofi, Regeneron, Amgen, Merck, and other companies. Her associates reported ties to numerous industry sources.
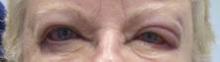
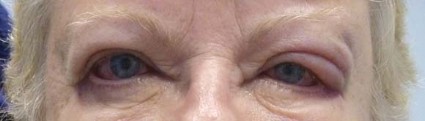
Eczema of the eyelids? Think chemical allergy
LAS VEGAS – If a patient presents with eczema of the eyelids, or swollen eyelids that don’t respond to topical steroids, think about sending them for chemical testing, advised Dr. Janet M. Neigel.
"The eyelids are red and scaly, a little swollen, and it just never goes away," she said in an interview at the annual meeting of the American Academy of Cosmetic Surgery.
Dr. Neigel, a cosmetic surgeon in West Orange, N.J., said that over the past 5 years, she has seen increasing numbers of patients present with eczema localized to the eyelids or eyeball area that recurs like pesky crabgrass.
"I treat them with topical steroids," Dr. Neigel said. "It will get better, but it always comes back. Some of this is seasonal. It may only happen in the winter, when the air is drier and their skin tends to get drier. In others it can be all year long," she said. "It seems to be more common in women, but I see men with this condition, too. In men, it tends to present as a reddish eye and tearing," she noted.
In the majority of cases, the culprit turns out to be an allergy to chemicals including gold, nickel, tin, rubber, preservatives in shampoos and laundry detergent, and formaldehyde resin, which is used in nail polish. "There was one patient who was allergic to the preservative in eyedrops," Dr. Neigel recalled. "She was on several different eyedrops trying to treat the swollen eye area, and it was just making the condition worse."
Another patient’s eczema cleared only after she removed her wedding ring, Dr. Neigel said. "So she couldn’t wear any gold jewelry. In somebody else it was tin and nickel, so she couldn’t wear any cheap jewelry."
Ointments commonly used for cosmetic procedures also can cause trouble. "There is cross-reactivity between neomycin, tobramycin, and Neosporin," Dr. Neigel said. "One patient was applying Neosporin every time she bumped herself on different parts of her body, and her eyelids were the only things flaring up."
Dr. Neigel speculated that the reaction in such cases is localized to the eyelid because "it’s the thinnest skin in the body. It’s the most sensitive, and for some reason, the patients I’m seeing only have reactions there," she noted. So, for patients with allergic conjunctivitis or tearing for a contact dermatitis–type presentation around the eyeball or the eyelids, send them for chemical testing, she advised. "There’s a good chance you might clear things up and figure out what they’re truly reacting to – get to the source instead of just treating the problem symptomatically," she said.
Dr. Neigel said she had no relevant financial disclosures.
LAS VEGAS – If a patient presents with eczema of the eyelids, or swollen eyelids that don’t respond to topical steroids, think about sending them for chemical testing, advised Dr. Janet M. Neigel.
"The eyelids are red and scaly, a little swollen, and it just never goes away," she said in an interview at the annual meeting of the American Academy of Cosmetic Surgery.
Dr. Neigel, a cosmetic surgeon in West Orange, N.J., said that over the past 5 years, she has seen increasing numbers of patients present with eczema localized to the eyelids or eyeball area that recurs like pesky crabgrass.
"I treat them with topical steroids," Dr. Neigel said. "It will get better, but it always comes back. Some of this is seasonal. It may only happen in the winter, when the air is drier and their skin tends to get drier. In others it can be all year long," she said. "It seems to be more common in women, but I see men with this condition, too. In men, it tends to present as a reddish eye and tearing," she noted.
In the majority of cases, the culprit turns out to be an allergy to chemicals including gold, nickel, tin, rubber, preservatives in shampoos and laundry detergent, and formaldehyde resin, which is used in nail polish. "There was one patient who was allergic to the preservative in eyedrops," Dr. Neigel recalled. "She was on several different eyedrops trying to treat the swollen eye area, and it was just making the condition worse."
Another patient’s eczema cleared only after she removed her wedding ring, Dr. Neigel said. "So she couldn’t wear any gold jewelry. In somebody else it was tin and nickel, so she couldn’t wear any cheap jewelry."
Ointments commonly used for cosmetic procedures also can cause trouble. "There is cross-reactivity between neomycin, tobramycin, and Neosporin," Dr. Neigel said. "One patient was applying Neosporin every time she bumped herself on different parts of her body, and her eyelids were the only things flaring up."
Dr. Neigel speculated that the reaction in such cases is localized to the eyelid because "it’s the thinnest skin in the body. It’s the most sensitive, and for some reason, the patients I’m seeing only have reactions there," she noted. So, for patients with allergic conjunctivitis or tearing for a contact dermatitis–type presentation around the eyeball or the eyelids, send them for chemical testing, she advised. "There’s a good chance you might clear things up and figure out what they’re truly reacting to – get to the source instead of just treating the problem symptomatically," she said.
Dr. Neigel said she had no relevant financial disclosures.
LAS VEGAS – If a patient presents with eczema of the eyelids, or swollen eyelids that don’t respond to topical steroids, think about sending them for chemical testing, advised Dr. Janet M. Neigel.
"The eyelids are red and scaly, a little swollen, and it just never goes away," she said in an interview at the annual meeting of the American Academy of Cosmetic Surgery.
Dr. Neigel, a cosmetic surgeon in West Orange, N.J., said that over the past 5 years, she has seen increasing numbers of patients present with eczema localized to the eyelids or eyeball area that recurs like pesky crabgrass.
"I treat them with topical steroids," Dr. Neigel said. "It will get better, but it always comes back. Some of this is seasonal. It may only happen in the winter, when the air is drier and their skin tends to get drier. In others it can be all year long," she said. "It seems to be more common in women, but I see men with this condition, too. In men, it tends to present as a reddish eye and tearing," she noted.
In the majority of cases, the culprit turns out to be an allergy to chemicals including gold, nickel, tin, rubber, preservatives in shampoos and laundry detergent, and formaldehyde resin, which is used in nail polish. "There was one patient who was allergic to the preservative in eyedrops," Dr. Neigel recalled. "She was on several different eyedrops trying to treat the swollen eye area, and it was just making the condition worse."
Another patient’s eczema cleared only after she removed her wedding ring, Dr. Neigel said. "So she couldn’t wear any gold jewelry. In somebody else it was tin and nickel, so she couldn’t wear any cheap jewelry."
Ointments commonly used for cosmetic procedures also can cause trouble. "There is cross-reactivity between neomycin, tobramycin, and Neosporin," Dr. Neigel said. "One patient was applying Neosporin every time she bumped herself on different parts of her body, and her eyelids were the only things flaring up."
Dr. Neigel speculated that the reaction in such cases is localized to the eyelid because "it’s the thinnest skin in the body. It’s the most sensitive, and for some reason, the patients I’m seeing only have reactions there," she noted. So, for patients with allergic conjunctivitis or tearing for a contact dermatitis–type presentation around the eyeball or the eyelids, send them for chemical testing, she advised. "There’s a good chance you might clear things up and figure out what they’re truly reacting to – get to the source instead of just treating the problem symptomatically," she said.
Dr. Neigel said she had no relevant financial disclosures.
AT THE AACS ANNUAL MEETING
Nickel, cobalt sensitivity increases with number of body piercings
MIAMI BEACH – The risk of nickel and cobalt sensitivity increases in tandem with the number of body piercings, according to findings from a study involving nearly 9,400 patch-tested patients.
Overall, 3,907 (41.6%) of the 9,388 subjects had no piercings, 131 (1.3%) had one piercing, 3,987 (42.5%) had two piercings, 934 (9.9%) had three to four piercings, and 429 (4.6%) had five or more piercings, Jaime L. Loso reported at the annual meeting of the American Contact Dermatitis Society.
Nickel and cobalt sensitivity both were significantly associated with piercing (relative risks of 2.52 and 1.63, respectively); chromate sensitivity had an inverse relationship with piercing (relative risk, 0.60), said Ms. Loso, a third-year medical student at the University of Minnesota, Minneapolis.
Overall, 1,651 patients (17.7%) had nickel sensitivity and 685 (7.3%) had cobalt sensitivity. The sensitivity rates increased with the number of piercings. The rates of sensitivity among those with zero, one, two, three to four, and five or more piercings were 9.4%, 16%, 22.6%, 25.1%, and 32.4%, respectively, for nickel and 5.3%, 7.6%, 8.2%, 9.5%, and 11.7% for cobalt.
Chromate sensitivity was less common, occurring in only 306 patients (3.26%). Less than 4.3% of patients in all piercing groups had chromate sensitivity, Ms. Loso noted.
"Nickel is the most common allergen for patch-tested patients, and body piercing has been directly correlated with the development of nickel allergy," she said, adding that cobalt content is often associated with nickel content in consumer products, although recent studies suggest that only a small amount of jewelry – mainly earrings – releases cobalt.
In the current study, which involved patients identified using North American Contact Dermatitis Group data from 2007 to 2010, younger patients were affected more often than older patients, females were affected more often than males, and – surprisingly – piercing was associated with allergy in males more often than in females, she reported.
The findings, though limited by the fact that the referral population may not be representative of the general population and by a lack of information regarding age at the time of piercing and the body sites pierced, help characterize metal allergy associated with body piercing, Ms. Loso said.
She noted that nickel allergy has been well studied in Europe – resulting in regulation of the nickel content in consumer goods and a subsequent significant decline in nickel sensitivity among young females there. The same is not true for the United States, where nickel sensitivity remains an important problem.
Ms. Loso reported having no disclosures.
MIAMI BEACH – The risk of nickel and cobalt sensitivity increases in tandem with the number of body piercings, according to findings from a study involving nearly 9,400 patch-tested patients.
Overall, 3,907 (41.6%) of the 9,388 subjects had no piercings, 131 (1.3%) had one piercing, 3,987 (42.5%) had two piercings, 934 (9.9%) had three to four piercings, and 429 (4.6%) had five or more piercings, Jaime L. Loso reported at the annual meeting of the American Contact Dermatitis Society.
Nickel and cobalt sensitivity both were significantly associated with piercing (relative risks of 2.52 and 1.63, respectively); chromate sensitivity had an inverse relationship with piercing (relative risk, 0.60), said Ms. Loso, a third-year medical student at the University of Minnesota, Minneapolis.
Overall, 1,651 patients (17.7%) had nickel sensitivity and 685 (7.3%) had cobalt sensitivity. The sensitivity rates increased with the number of piercings. The rates of sensitivity among those with zero, one, two, three to four, and five or more piercings were 9.4%, 16%, 22.6%, 25.1%, and 32.4%, respectively, for nickel and 5.3%, 7.6%, 8.2%, 9.5%, and 11.7% for cobalt.
Chromate sensitivity was less common, occurring in only 306 patients (3.26%). Less than 4.3% of patients in all piercing groups had chromate sensitivity, Ms. Loso noted.
"Nickel is the most common allergen for patch-tested patients, and body piercing has been directly correlated with the development of nickel allergy," she said, adding that cobalt content is often associated with nickel content in consumer products, although recent studies suggest that only a small amount of jewelry – mainly earrings – releases cobalt.
In the current study, which involved patients identified using North American Contact Dermatitis Group data from 2007 to 2010, younger patients were affected more often than older patients, females were affected more often than males, and – surprisingly – piercing was associated with allergy in males more often than in females, she reported.
The findings, though limited by the fact that the referral population may not be representative of the general population and by a lack of information regarding age at the time of piercing and the body sites pierced, help characterize metal allergy associated with body piercing, Ms. Loso said.
She noted that nickel allergy has been well studied in Europe – resulting in regulation of the nickel content in consumer goods and a subsequent significant decline in nickel sensitivity among young females there. The same is not true for the United States, where nickel sensitivity remains an important problem.
Ms. Loso reported having no disclosures.
MIAMI BEACH – The risk of nickel and cobalt sensitivity increases in tandem with the number of body piercings, according to findings from a study involving nearly 9,400 patch-tested patients.
Overall, 3,907 (41.6%) of the 9,388 subjects had no piercings, 131 (1.3%) had one piercing, 3,987 (42.5%) had two piercings, 934 (9.9%) had three to four piercings, and 429 (4.6%) had five or more piercings, Jaime L. Loso reported at the annual meeting of the American Contact Dermatitis Society.
Nickel and cobalt sensitivity both were significantly associated with piercing (relative risks of 2.52 and 1.63, respectively); chromate sensitivity had an inverse relationship with piercing (relative risk, 0.60), said Ms. Loso, a third-year medical student at the University of Minnesota, Minneapolis.
Overall, 1,651 patients (17.7%) had nickel sensitivity and 685 (7.3%) had cobalt sensitivity. The sensitivity rates increased with the number of piercings. The rates of sensitivity among those with zero, one, two, three to four, and five or more piercings were 9.4%, 16%, 22.6%, 25.1%, and 32.4%, respectively, for nickel and 5.3%, 7.6%, 8.2%, 9.5%, and 11.7% for cobalt.
Chromate sensitivity was less common, occurring in only 306 patients (3.26%). Less than 4.3% of patients in all piercing groups had chromate sensitivity, Ms. Loso noted.
"Nickel is the most common allergen for patch-tested patients, and body piercing has been directly correlated with the development of nickel allergy," she said, adding that cobalt content is often associated with nickel content in consumer products, although recent studies suggest that only a small amount of jewelry – mainly earrings – releases cobalt.
In the current study, which involved patients identified using North American Contact Dermatitis Group data from 2007 to 2010, younger patients were affected more often than older patients, females were affected more often than males, and – surprisingly – piercing was associated with allergy in males more often than in females, she reported.
The findings, though limited by the fact that the referral population may not be representative of the general population and by a lack of information regarding age at the time of piercing and the body sites pierced, help characterize metal allergy associated with body piercing, Ms. Loso said.
She noted that nickel allergy has been well studied in Europe – resulting in regulation of the nickel content in consumer goods and a subsequent significant decline in nickel sensitivity among young females there. The same is not true for the United States, where nickel sensitivity remains an important problem.
Ms. Loso reported having no disclosures.
AT THE ACDS ANNUAL MEETING
Major finding: Sensitivity to nickel and cobalt (relative risk of 2.52, 1.63, respectively) was significantly associated with body piercing.
Data source: A retrospective cross-sectional analysis.
Disclosures: Dr. Loso reported having no disclosures.
Cat allergy vaccine effects persist at 2 years
A short course of treatment with an investigational synthetic cat-peptide antigen desensitizing vaccine, or Cat-PAD, results in a substantial and persistent reduction in cat allergy symptom scores, according to two-year findings from a randomized, placebo-controlled phase II clinical study involving 202 adult patients.
Study participants were initially randomized to receive either eight 3-nmol intradermal doses at 2-week intervals, four 6 nmol-doses at 4-week intervals, or placebo. At 1-year follow-up, the improvement in Total Rhinoconjunctivitis Symptom Score (TRSS) was significantly greater in the patients who received four doses of Cat-PAD (ToleroMune Cat, Circassia Limited, Oxford, England), compared with those who received placebo (–7.1 points vs. –2.99 points), according to findings published online in the Journal of Allergy and Clinical Immunology (doi:10.1016/j.jaci.2012.12.1185).
Data from a 2-year follow-up study were reported by Rod P. Hafner, Ph.D., and his colleagues in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Of 89 patients enrolled into the follow-up study, 50 returned at 2-years after the start of treatment, having received no additional treatment, for an environmental exposure chamber challenge. The magnitude of difference from baseline in TRSS seen at 1 year in those who received 4 doses and those who received placebo was maintained at 2 years (–5.87 vs. –2.02) among those who initially received the 4-dose regimen.
"Cat-PAD is the first in a new class of synthetic peptide immune-regulatory epitopes. The results from this study provide the first evidence that four doses of 6 nmol Cat-PAD over a 12-week period has a disease-modifying effect with subjects showing sustained improvement at 2 years," the investigators wrote.
Study participants were adults aged 18-65 years with cat allergy who underwent a baseline environmental exposure chamber (EEC) challenge, as well as follow-up EEC challenges at 18-22 weeks and at 100-104 weeks.
"Cat allergen was dispersed into the EEC to achieve a consistent mean level of approx. 50 ng Fel d1/m3, using a validated method," they explained, noting that TRSS was calculated at each EEC challenge based on self-scoring of four nasal symptoms (running nose, sneezing, blocked nose, itchy nose), and four ocular symptoms (itchy eyes, watery eyes, red eyes, sore eyes) on a scale of 0-3, every 30 minutes during the challenge.
Cat-PAD is a "potentially exciting new approach to cat allergy immunotherapy," the investigators said, noting that improvements in the TRSS seen in the initial phase II study and follow-up study represent a substantial improvement over numerous therapies investigated in the past, in some cases reaching a threefold improvement in symptom reduction.
"For example, studies of similar design in an EEC reported TRSS changes of approximately –1.5 units after 16 weeks treatment with a sublingual cat allergy tablet, while a single 180-mg dose of the antihistamine fexofenadine achieved a mean difference in TRSS of –1.3 in a cat allergen study. Moreover, the change in TRSS of –3.8 units reported here was observed after four administrations of Cat-PAD over a 12-week period and persisted 21 months after the end of treatment," they concluded.
In late 2012 the investigators began enrolling a phase III study, which will include individuals aged 12-65 years.
This study was funded by Circassia Limited. Dr. Hafner is employed by Circassia.
A short course of treatment with an investigational synthetic cat-peptide antigen desensitizing vaccine, or Cat-PAD, results in a substantial and persistent reduction in cat allergy symptom scores, according to two-year findings from a randomized, placebo-controlled phase II clinical study involving 202 adult patients.
Study participants were initially randomized to receive either eight 3-nmol intradermal doses at 2-week intervals, four 6 nmol-doses at 4-week intervals, or placebo. At 1-year follow-up, the improvement in Total Rhinoconjunctivitis Symptom Score (TRSS) was significantly greater in the patients who received four doses of Cat-PAD (ToleroMune Cat, Circassia Limited, Oxford, England), compared with those who received placebo (–7.1 points vs. –2.99 points), according to findings published online in the Journal of Allergy and Clinical Immunology (doi:10.1016/j.jaci.2012.12.1185).
Data from a 2-year follow-up study were reported by Rod P. Hafner, Ph.D., and his colleagues in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Of 89 patients enrolled into the follow-up study, 50 returned at 2-years after the start of treatment, having received no additional treatment, for an environmental exposure chamber challenge. The magnitude of difference from baseline in TRSS seen at 1 year in those who received 4 doses and those who received placebo was maintained at 2 years (–5.87 vs. –2.02) among those who initially received the 4-dose regimen.
"Cat-PAD is the first in a new class of synthetic peptide immune-regulatory epitopes. The results from this study provide the first evidence that four doses of 6 nmol Cat-PAD over a 12-week period has a disease-modifying effect with subjects showing sustained improvement at 2 years," the investigators wrote.
Study participants were adults aged 18-65 years with cat allergy who underwent a baseline environmental exposure chamber (EEC) challenge, as well as follow-up EEC challenges at 18-22 weeks and at 100-104 weeks.
"Cat allergen was dispersed into the EEC to achieve a consistent mean level of approx. 50 ng Fel d1/m3, using a validated method," they explained, noting that TRSS was calculated at each EEC challenge based on self-scoring of four nasal symptoms (running nose, sneezing, blocked nose, itchy nose), and four ocular symptoms (itchy eyes, watery eyes, red eyes, sore eyes) on a scale of 0-3, every 30 minutes during the challenge.
Cat-PAD is a "potentially exciting new approach to cat allergy immunotherapy," the investigators said, noting that improvements in the TRSS seen in the initial phase II study and follow-up study represent a substantial improvement over numerous therapies investigated in the past, in some cases reaching a threefold improvement in symptom reduction.
"For example, studies of similar design in an EEC reported TRSS changes of approximately –1.5 units after 16 weeks treatment with a sublingual cat allergy tablet, while a single 180-mg dose of the antihistamine fexofenadine achieved a mean difference in TRSS of –1.3 in a cat allergen study. Moreover, the change in TRSS of –3.8 units reported here was observed after four administrations of Cat-PAD over a 12-week period and persisted 21 months after the end of treatment," they concluded.
In late 2012 the investigators began enrolling a phase III study, which will include individuals aged 12-65 years.
This study was funded by Circassia Limited. Dr. Hafner is employed by Circassia.
A short course of treatment with an investigational synthetic cat-peptide antigen desensitizing vaccine, or Cat-PAD, results in a substantial and persistent reduction in cat allergy symptom scores, according to two-year findings from a randomized, placebo-controlled phase II clinical study involving 202 adult patients.
Study participants were initially randomized to receive either eight 3-nmol intradermal doses at 2-week intervals, four 6 nmol-doses at 4-week intervals, or placebo. At 1-year follow-up, the improvement in Total Rhinoconjunctivitis Symptom Score (TRSS) was significantly greater in the patients who received four doses of Cat-PAD (ToleroMune Cat, Circassia Limited, Oxford, England), compared with those who received placebo (–7.1 points vs. –2.99 points), according to findings published online in the Journal of Allergy and Clinical Immunology (doi:10.1016/j.jaci.2012.12.1185).
Data from a 2-year follow-up study were reported by Rod P. Hafner, Ph.D., and his colleagues in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Of 89 patients enrolled into the follow-up study, 50 returned at 2-years after the start of treatment, having received no additional treatment, for an environmental exposure chamber challenge. The magnitude of difference from baseline in TRSS seen at 1 year in those who received 4 doses and those who received placebo was maintained at 2 years (–5.87 vs. –2.02) among those who initially received the 4-dose regimen.
"Cat-PAD is the first in a new class of synthetic peptide immune-regulatory epitopes. The results from this study provide the first evidence that four doses of 6 nmol Cat-PAD over a 12-week period has a disease-modifying effect with subjects showing sustained improvement at 2 years," the investigators wrote.
Study participants were adults aged 18-65 years with cat allergy who underwent a baseline environmental exposure chamber (EEC) challenge, as well as follow-up EEC challenges at 18-22 weeks and at 100-104 weeks.
"Cat allergen was dispersed into the EEC to achieve a consistent mean level of approx. 50 ng Fel d1/m3, using a validated method," they explained, noting that TRSS was calculated at each EEC challenge based on self-scoring of four nasal symptoms (running nose, sneezing, blocked nose, itchy nose), and four ocular symptoms (itchy eyes, watery eyes, red eyes, sore eyes) on a scale of 0-3, every 30 minutes during the challenge.
Cat-PAD is a "potentially exciting new approach to cat allergy immunotherapy," the investigators said, noting that improvements in the TRSS seen in the initial phase II study and follow-up study represent a substantial improvement over numerous therapies investigated in the past, in some cases reaching a threefold improvement in symptom reduction.
"For example, studies of similar design in an EEC reported TRSS changes of approximately –1.5 units after 16 weeks treatment with a sublingual cat allergy tablet, while a single 180-mg dose of the antihistamine fexofenadine achieved a mean difference in TRSS of –1.3 in a cat allergen study. Moreover, the change in TRSS of –3.8 units reported here was observed after four administrations of Cat-PAD over a 12-week period and persisted 21 months after the end of treatment," they concluded.
In late 2012 the investigators began enrolling a phase III study, which will include individuals aged 12-65 years.
This study was funded by Circassia Limited. Dr. Hafner is employed by Circassia.
AT THE AAAAI ANNUAL MEETING
Major finding: Improvements in TRSS in treated vs. placebo patients were similar at 1 year (–7.1 points vs. –2.99 points), and 2 years (–5.87 vs. –2.02) follow-up.
Data source: A randomized placebo-controlled study and follow-up study involving 202 adults.
Disclosures: This study was funded by Circassia Limited. Dr. Hafner is employed by Circassia.
Atopic dermatitis linked to psychiatric comorbidities
Emerging data strongly suggest that children with atopic dermatitis are at increased risk for attention-deficit/hyperactivity disorder.
"The first paper was actually a really good paper, but it was just one study and so we did not quite believe it. But now there’s a confirmatory study by a separate group with really big data," Dr. Lawrence K. Eichenfield said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
In addition, the second study pointed to a possible higher risk of autism in children with atopic dermatitis, and it also confirmed other reports of increased rates of depression and anxiety, noted Dr. Eichenfield, professor of clinical pediatrics and medicine at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital, also in San Diego.
Atopic dermatitis is the most common chronic inflammatory condition in childhood. Attention-deficit/hyperactivity disorder is the most common psychiatric disorder. The first study to report an association between the two was a German, case-control, population-based study involving 1,436 atopic dermatitis patients aged 6-17 years and an equal number of age- and gender-matched controls without the skin disease.
The prevalence of diagnosed ADHD was 5.2% in the atopic dermatitis patients and 3.4% in controls (P = 0.02). In a multivariate logistic regression analysis, atopic dermatitis was an independent predictor of prevalent ADHD, with an associated 47% increased risk of the psychiatric disorder compared with youths without atopic dermatitis. Moreover, there was a suggestion that the more severe a patient’s atopic dermatitis, the greater the likelihood of comorbid ADHD, since the likelihood of ADHD increased with each additional physician visit for atopic dermatitis (JAMA 2009;301:724-6).
More recently, in a study presented at a major meeting but not yet published, Dr. Eric L. Simpson and his coinvestigators at Oregon Health and Science University, Portland, confirmed the initial German finding that pediatric atopic dermatitis is associated with ADHD, this time in a database of 90,000 U.S. children. Once again, the more severe the skin disease, the greater the risk of ADHD. In this study, atopic dermatitis was also associated with increased risks of depression, anxiety, and autism.
Possible mechanisms underlying the observed association between atopic dermatitis and comorbid psychiatric conditions include sleep disturbance – a prominent feature in atopic dermatitis – and systemic inflammation, according to Dr. Eichenfield.
The well-established atopic comorbidities associated with atopic dermatitis are asthma, allergic rhinitis, and food allergy. Dr. Eichenfield said that he welcomes the evolving evidence of psychologic comorbidities for the opportunity it provides to encourage treatment adherence. Like other treating physicians, he finds topical steroid phobia to be a huge problem.
"I actually use this information in my clinical practice as a way to get my families to adequately treat atopic dermatitis," he said. "If I have families that are very steroid phobic, I’ll say, ‘You know, you really want to treat your kid with appropriate topical therapy to minimize the disease impact: to minimize the sleep disturbance and potentially minimize the neurobehavioral impact of atopic dermatitis.’ I use that as a tool to stay with our regimens of care."
Steroid phobia is a problem worldwide, not just in the United States. For example, Dr. Eichenfield noted that when French dermatologists recently surveyed 208 consecutive atopic dermatitis patients or their parents using a detailed 69-item questionnaire, 81% reported having fears about topical corticosteroids – the mainstay of atopic dermatitis treatment – and 36% admitted to treatment nonadherence as a result of their worries. Fears did not correlate with disease severity (Br. J. Dermatol. 2011;165:808-14).
"I think that in all of dermatology there’s probably no greater chasm between where our families are at in terms of concerns about a medication and where we are as physicians in terms of not having concerns than in the area of topical steroids for atopic dermatitis," Dr. Eichenfield said.
He noted that he makes extensive use of parental educational handouts and written action plans in addition to face-to-face discussion of safety concerns at the time he prescribes topical steroids. One strategy that he’s recently realized makes a big difference in terms of adherence is to explain carefully that the midpotency topical steroids used in treating atopic dermatitis – as distinct from potent and superpotent topical steroids – do not have the side effects and systemic absorption issues steroid-phobic parents are worried about.
Another effective strategy is to be very specific about the quantity of topical medication he wants the patient to use for flare control.
"Instead of saying, ‘Twice a day application of a midstrength topical steroid for 2 weeks,’ now I’ll say, "I would like you to use up this 80-g tube in the next 2 weeks and then I’ll see you back – and it’s safe to use this amount for this particular length of time.’ I find that makes a huge difference in terms of outcomes if I explain it that way," Dr. Eichenfield said.
He reported serving as a consultant to Bayer, Galderma, GlaxoSmithKline, and Valeant.
SDEF and this news organization are owned by the same parent company.
Emerging data strongly suggest that children with atopic dermatitis are at increased risk for attention-deficit/hyperactivity disorder.
"The first paper was actually a really good paper, but it was just one study and so we did not quite believe it. But now there’s a confirmatory study by a separate group with really big data," Dr. Lawrence K. Eichenfield said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
In addition, the second study pointed to a possible higher risk of autism in children with atopic dermatitis, and it also confirmed other reports of increased rates of depression and anxiety, noted Dr. Eichenfield, professor of clinical pediatrics and medicine at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital, also in San Diego.
Atopic dermatitis is the most common chronic inflammatory condition in childhood. Attention-deficit/hyperactivity disorder is the most common psychiatric disorder. The first study to report an association between the two was a German, case-control, population-based study involving 1,436 atopic dermatitis patients aged 6-17 years and an equal number of age- and gender-matched controls without the skin disease.
The prevalence of diagnosed ADHD was 5.2% in the atopic dermatitis patients and 3.4% in controls (P = 0.02). In a multivariate logistic regression analysis, atopic dermatitis was an independent predictor of prevalent ADHD, with an associated 47% increased risk of the psychiatric disorder compared with youths without atopic dermatitis. Moreover, there was a suggestion that the more severe a patient’s atopic dermatitis, the greater the likelihood of comorbid ADHD, since the likelihood of ADHD increased with each additional physician visit for atopic dermatitis (JAMA 2009;301:724-6).
More recently, in a study presented at a major meeting but not yet published, Dr. Eric L. Simpson and his coinvestigators at Oregon Health and Science University, Portland, confirmed the initial German finding that pediatric atopic dermatitis is associated with ADHD, this time in a database of 90,000 U.S. children. Once again, the more severe the skin disease, the greater the risk of ADHD. In this study, atopic dermatitis was also associated with increased risks of depression, anxiety, and autism.
Possible mechanisms underlying the observed association between atopic dermatitis and comorbid psychiatric conditions include sleep disturbance – a prominent feature in atopic dermatitis – and systemic inflammation, according to Dr. Eichenfield.
The well-established atopic comorbidities associated with atopic dermatitis are asthma, allergic rhinitis, and food allergy. Dr. Eichenfield said that he welcomes the evolving evidence of psychologic comorbidities for the opportunity it provides to encourage treatment adherence. Like other treating physicians, he finds topical steroid phobia to be a huge problem.
"I actually use this information in my clinical practice as a way to get my families to adequately treat atopic dermatitis," he said. "If I have families that are very steroid phobic, I’ll say, ‘You know, you really want to treat your kid with appropriate topical therapy to minimize the disease impact: to minimize the sleep disturbance and potentially minimize the neurobehavioral impact of atopic dermatitis.’ I use that as a tool to stay with our regimens of care."
Steroid phobia is a problem worldwide, not just in the United States. For example, Dr. Eichenfield noted that when French dermatologists recently surveyed 208 consecutive atopic dermatitis patients or their parents using a detailed 69-item questionnaire, 81% reported having fears about topical corticosteroids – the mainstay of atopic dermatitis treatment – and 36% admitted to treatment nonadherence as a result of their worries. Fears did not correlate with disease severity (Br. J. Dermatol. 2011;165:808-14).
"I think that in all of dermatology there’s probably no greater chasm between where our families are at in terms of concerns about a medication and where we are as physicians in terms of not having concerns than in the area of topical steroids for atopic dermatitis," Dr. Eichenfield said.
He noted that he makes extensive use of parental educational handouts and written action plans in addition to face-to-face discussion of safety concerns at the time he prescribes topical steroids. One strategy that he’s recently realized makes a big difference in terms of adherence is to explain carefully that the midpotency topical steroids used in treating atopic dermatitis – as distinct from potent and superpotent topical steroids – do not have the side effects and systemic absorption issues steroid-phobic parents are worried about.
Another effective strategy is to be very specific about the quantity of topical medication he wants the patient to use for flare control.
"Instead of saying, ‘Twice a day application of a midstrength topical steroid for 2 weeks,’ now I’ll say, "I would like you to use up this 80-g tube in the next 2 weeks and then I’ll see you back – and it’s safe to use this amount for this particular length of time.’ I find that makes a huge difference in terms of outcomes if I explain it that way," Dr. Eichenfield said.
He reported serving as a consultant to Bayer, Galderma, GlaxoSmithKline, and Valeant.
SDEF and this news organization are owned by the same parent company.
Emerging data strongly suggest that children with atopic dermatitis are at increased risk for attention-deficit/hyperactivity disorder.
"The first paper was actually a really good paper, but it was just one study and so we did not quite believe it. But now there’s a confirmatory study by a separate group with really big data," Dr. Lawrence K. Eichenfield said at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
In addition, the second study pointed to a possible higher risk of autism in children with atopic dermatitis, and it also confirmed other reports of increased rates of depression and anxiety, noted Dr. Eichenfield, professor of clinical pediatrics and medicine at the University of California, San Diego, and chief of pediatric and adolescent dermatology at Rady Children’s Hospital, also in San Diego.
Atopic dermatitis is the most common chronic inflammatory condition in childhood. Attention-deficit/hyperactivity disorder is the most common psychiatric disorder. The first study to report an association between the two was a German, case-control, population-based study involving 1,436 atopic dermatitis patients aged 6-17 years and an equal number of age- and gender-matched controls without the skin disease.
The prevalence of diagnosed ADHD was 5.2% in the atopic dermatitis patients and 3.4% in controls (P = 0.02). In a multivariate logistic regression analysis, atopic dermatitis was an independent predictor of prevalent ADHD, with an associated 47% increased risk of the psychiatric disorder compared with youths without atopic dermatitis. Moreover, there was a suggestion that the more severe a patient’s atopic dermatitis, the greater the likelihood of comorbid ADHD, since the likelihood of ADHD increased with each additional physician visit for atopic dermatitis (JAMA 2009;301:724-6).
More recently, in a study presented at a major meeting but not yet published, Dr. Eric L. Simpson and his coinvestigators at Oregon Health and Science University, Portland, confirmed the initial German finding that pediatric atopic dermatitis is associated with ADHD, this time in a database of 90,000 U.S. children. Once again, the more severe the skin disease, the greater the risk of ADHD. In this study, atopic dermatitis was also associated with increased risks of depression, anxiety, and autism.
Possible mechanisms underlying the observed association between atopic dermatitis and comorbid psychiatric conditions include sleep disturbance – a prominent feature in atopic dermatitis – and systemic inflammation, according to Dr. Eichenfield.
The well-established atopic comorbidities associated with atopic dermatitis are asthma, allergic rhinitis, and food allergy. Dr. Eichenfield said that he welcomes the evolving evidence of psychologic comorbidities for the opportunity it provides to encourage treatment adherence. Like other treating physicians, he finds topical steroid phobia to be a huge problem.
"I actually use this information in my clinical practice as a way to get my families to adequately treat atopic dermatitis," he said. "If I have families that are very steroid phobic, I’ll say, ‘You know, you really want to treat your kid with appropriate topical therapy to minimize the disease impact: to minimize the sleep disturbance and potentially minimize the neurobehavioral impact of atopic dermatitis.’ I use that as a tool to stay with our regimens of care."
Steroid phobia is a problem worldwide, not just in the United States. For example, Dr. Eichenfield noted that when French dermatologists recently surveyed 208 consecutive atopic dermatitis patients or their parents using a detailed 69-item questionnaire, 81% reported having fears about topical corticosteroids – the mainstay of atopic dermatitis treatment – and 36% admitted to treatment nonadherence as a result of their worries. Fears did not correlate with disease severity (Br. J. Dermatol. 2011;165:808-14).
"I think that in all of dermatology there’s probably no greater chasm between where our families are at in terms of concerns about a medication and where we are as physicians in terms of not having concerns than in the area of topical steroids for atopic dermatitis," Dr. Eichenfield said.
He noted that he makes extensive use of parental educational handouts and written action plans in addition to face-to-face discussion of safety concerns at the time he prescribes topical steroids. One strategy that he’s recently realized makes a big difference in terms of adherence is to explain carefully that the midpotency topical steroids used in treating atopic dermatitis – as distinct from potent and superpotent topical steroids – do not have the side effects and systemic absorption issues steroid-phobic parents are worried about.
Another effective strategy is to be very specific about the quantity of topical medication he wants the patient to use for flare control.
"Instead of saying, ‘Twice a day application of a midstrength topical steroid for 2 weeks,’ now I’ll say, "I would like you to use up this 80-g tube in the next 2 weeks and then I’ll see you back – and it’s safe to use this amount for this particular length of time.’ I find that makes a huge difference in terms of outcomes if I explain it that way," Dr. Eichenfield said.
He reported serving as a consultant to Bayer, Galderma, GlaxoSmithKline, and Valeant.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR