User login
Current Concepts in Lip Augmentation
Historically, a variety of tools have been used to alter one’s appearance for cultural or religious purposes or to conform to standards of beauty. As a defining feature of the face, the lips provide a unique opportunity for facial aesthetic enhancement. There has been a paradigm shift in medicine favoring preventative health and a desire to slow and even reverse the aging process.1 Acknowledging that product technology, skill sets, and cultural ideals continually evolve, this article highlights perioral anatomy, explains aging of the lower face, and reviews techniques to achieve perioral rejuvenation through volume restoration and muscle control.
Perioral Anatomy
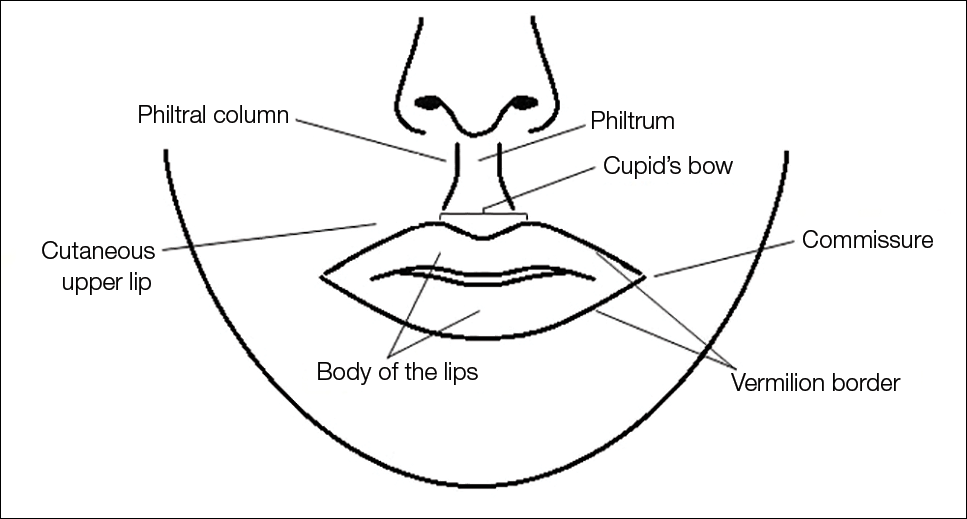
The layers of the lips include the epidermis, subcutaneous tissue, orbicularis oris muscle fibers, and mucosa. The upper lip extends from the base of the nose to the mucosa inferiorly and to the nasolabial folds laterally. The curvilinear lower lip extends from the mucosa to the mandible inferiorly and to the oral commissures laterally.2 Circumferential at the vermilion-cutaneous junction, a raised area of pale skin known as the white roll accentuates the vermilion border and provides an important landmark during lip augmentation.3 At the upper lip, this elevation of the vermilion joins at a V-shaped depression centrally to form the Cupid’s bow. The cutaneous upper lip has 2 raised vertical pillars known as the philtral columns, which are formed from decussating fibers of the orbicularis oris muscle.2 The resultant midline depression is the philtrum. These defining features of the upper lip are to be preserved during augmentation procedures (Figure 1).4
The superior and inferior labial arteries, both branches of the facial artery, supply the upper and lower lip, respectively. The anastomotic arch of the superior labial artery is susceptible to injury from deep injection of the upper lip between the muscle layer and mucosa; therefore, caution must be exercised in this area.5 Injections into the vermilion and lower lip can be safely performed with less concern for vascular compromise. The vermilion derives its red color from the translucency of capillaries in the superficial papillae.2 The capillary plexus at the papillae and rich sensory nerve network render the lip a highly vascular and sensitive structure.
Aging of the Lower Face
Subcutaneous fat atrophy, loss of elasticity, gravitational forces, and remodeling of the skeletal foundation all contribute to aging of the lower face. Starting as early as the third decade of life, intrinsic factors including hormonal changes and genetically determined processes produce alterations in skin quality and structure. Similarly, extrinsic aging through environmental influences, namely exposure to UV radiation and smoking, accelerate the loss of skin integrity.6
The decreased laxity of the skin in combination with repeated contraction of the orbicularis oris muscle results in perioral rhytides.7 For women in particular, vertically oriented perioral rhytides develop above the vermilion; terminal hair follicles, thicker skin, and a greater density of subcutaneous fat are presumptive protective factors for males.8 With time, the cutaneous portion of the upper lip lengthens and there is redistribution of volume with effacement of the upper lip vermilion.9 Additionally, the demarcation of the vermilion becomes blurred secondary to pallor, flattening of the philtral columns, and loss of projection of the Cupid’s bow.10
Downturning of the oral commissures is observed secondary to a combination of gravity, bone resorption, and soft tissue volume loss. Hyperactivity of the depressor anguli oris muscle exacerbates the mesolabial folds, producing marionette lines and a saddened expression.7 With ongoing volume loss and ligament laxity, tissue redistributes near the jaws and chin, giving rise to jowls. Similarly, perioral volume loss and descent of the malar fat-pad deepen the nasolabial folds in the aging midface.6
The main objective of perioral rejuvenation is to reinstate a harmonious refreshed look to the lower face; however, aesthetic analysis should occur within the context of the face as a whole, as the lips should complement the surrounding perioral cosmetic unit and overall skeletal foundation of the face. To accomplish this goal, the dermatologist’s armamentarium contains a broad variety of approaches including restriction of muscle movement, volume restoration, and surface contouring.
Volume Restoration
Treatment Options
In 2015, hyaluronic acid (HA) fillers constituted 80% of all injectable soft-tissue fillers, an 8% increase from 2014.11 Hyaluronic acid has achieved immense popularity as a temporary dermal filler given its biocompatibility, longevity, and reversibility via hyaluronidase.12
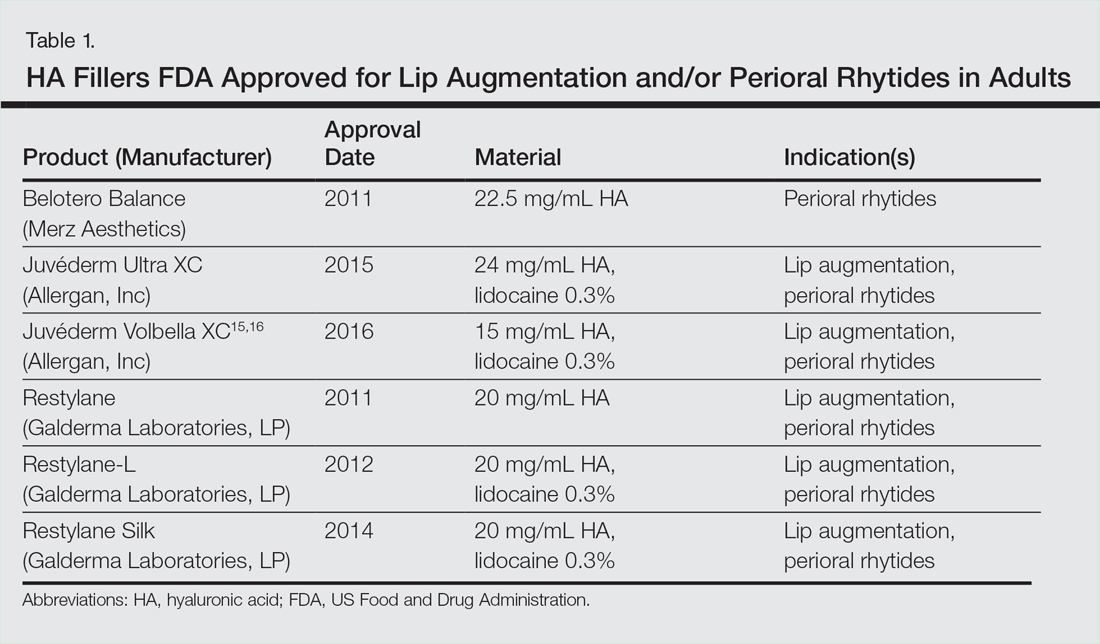
Hyaluronic acid is a naturally occurring glycosaminoglycan that comprises the connective tissue matrix. The molecular composition affords HA its hydrophilic property, which augments dermal volume.7 Endogenous HA has a short half-life, and chemical modification by a cross-linking process extends longevity by 6 to 12 months. The various HA fillers are distinguished by method of purification, size of molecules, concentration and degree of cross-linking, and viscosity.7,13,14 These differences dictate overall clinical performance such as flow properties, longevity, and stability. As a general rule, a high-viscosity product is more appropriate for deeper augmentation; fillers with low viscosity are more appropriate for correction of shallow defects.1 Table 1 lists the HA fillers that are currently approved by the US Food and Drug Administration for lip augmentation and/or perioral rhytides in adults 21 years and older.15-17
Randomized controlled trials comparing the efficacy, longevity, and tolerability of different HA products are lacking in the literature and, where present, have strong industry influence.18,19 The advent of assessment scales has provided an objective evaluation of perioral and lip augmentation, facilitating comparisons between products in both clinical research and practice.20
Semipermanent biostimulatory dermal fillers such as calcium hydroxylapatite and poly-L-lactic acid are not recommended for lip augmentation due to an increased incidence of submucosal nodule formation.6,14,21 Likewise, permanent fillers are not recommended given their irreversibility and risk of nodule formation around the lips.14,22 Nonetheless, liquid silicone (purified polydimethylsiloxane) administered via a microdroplet technique (0.01 mL of silicone at a time, no more than 1 cc per lip per session) has been used off label as a permanent filling agent for lip augmentation with limited complications.23 Regardless, trepidations about its use with respect to reported risks continue to limit its application.22
Similarly, surgical lip implants such as expanded polytetrafluoroethylene is an option for a subset of patients desiring permanent enhancement but are less commonly utilized given the side-effect profile, irreversibility, and relatively invasive nature of the procedure.22 Lastly, autologous fat transfer has been used in correction of the nasolabial and mesolabial folds as well as in lip augmentation; however, irregular surface contours and unpredictable longevity secondary to postinjection resorption (20%–90%) has limited its popularity.3,14,21
HA Injection Technique
With respect to HA fillers in the perioral area, numerous approaches have been described.10,22 The techniques in Table 2 provide a foundation for lip rejuvenation.

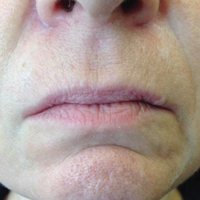
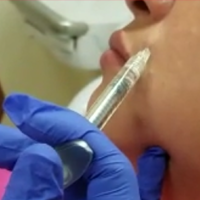
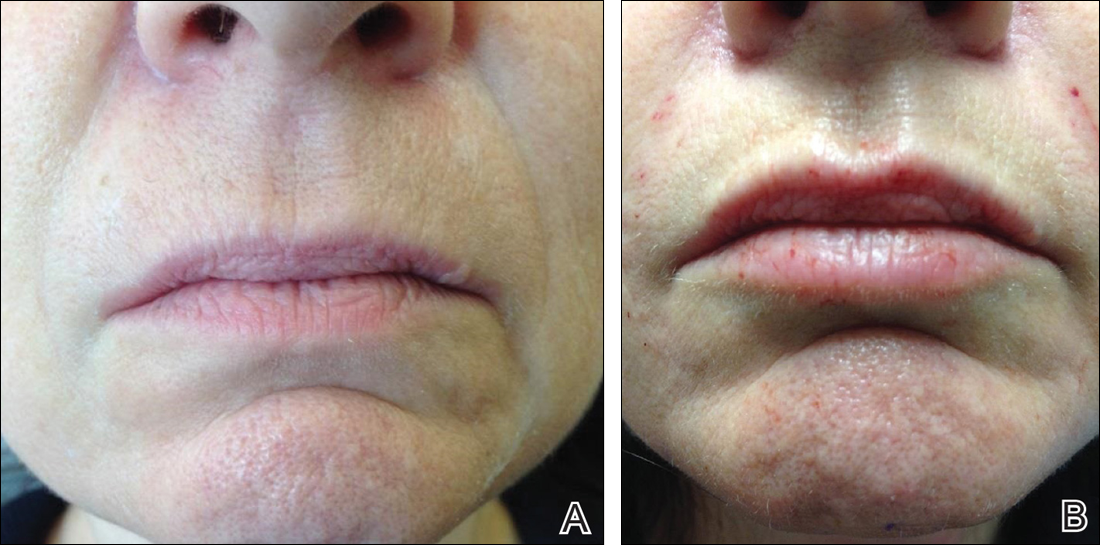
Several injection techniques exist, including serial puncture, linear threading, cross-hatching, and fanning in a retrograde or anterograde manner.24 A blunt microcannula (27 gauge, 38 mm) may be used in place of sharp needles and offers the benefit of increased patient comfort, reduced edema and ecchymosis, and shortened recovery period.25,26 Gentle massage of the product after injection can assist with an even contour. Lastly, a key determinant of successful outcomes is using an adequate volume of HA filler (1–2 mL for shaping the vermilion border and volumizing the lips).27 Figure 2 highlights a clinical example of HA filler for lip augmentation.
Fortunately, most complications encountered with HA lip augmentation are mild and transient. The most commonly observed side effects include injection-site reactions such as pain, erythema, and edema. Similarly, most adverse effects are related to injection technique. All HA fillers are prone to the Tyndall effect, a consequence of too superficial an injection plane. Patients with history of recurrent herpes simplex virus infections should receive prophylactic antiviral therapy.12
Muscle Control
An emerging concept in rejuvenation of the lower face recognizes not only restoration of volume but also control of muscle movement. Local injection of botulinum toxin type A induces relaxation of hyperfunctional facial muscles through temporary inhibition of neurotransmitter release.6 The potential for paralysis of the oral cavity may limit the application of botulinum toxin type A in that region.7 Nonetheless, the off-label potential of botulinum toxin type A has expanded to include several targets in the lower face. The orbicularis oris muscle is targeted to soften perioral rhytides. Conservative dosing (1–2 U per lip quadrant or approximately 5 U total) and superficial injection is emphasized in this area.27 Similarly, the depressor anguli oris muscle is targeted by injection of 4 U bilaterally to soften the marionette lines. In the chin area, the mentalis muscle can be targeted by injection of 2 U deep into each belly of the muscle to reduce the mental crease and dimpling.28 Combination treatment with dermal filler and neurotoxin demonstrates effects that last longer than either modality alone without additional adverse events.29 With combination therapy, guidelines suggest treating with filler first.27
Conclusion
A greater understanding of the extrinsic and intrinsic factors that contribute to the structural and surface changes of the aging face coupled with a preference for minimally invasive procedures has revolutionized the dermatologist’s approach to perioral rejuvenation. Serving as a focal point of the face, the lips and perioral skin are well poised to benefit from this paradigm shift. A multifaceted approach utilizing dermal fillers and neurotoxins may be most appropriate and has demonstrated optimal outcomes in facial aesthetics.
- Buck DW, Alam M, Kim JYS. Injectable fillers for facial rejuvenation: a review. J Plast Reconstr Aesthet Surg. 2009;62:11-18.
- Guareschi M, Stella E. Lips. In: Goisis M, ed. Injections in Aesthetic Medicine. Milan, Italy: Springer; 2014:125-136.
- Byrne PJ, Hilger PA. Lip augmentation. Facial Plast Surg. 2004;20:31-38.
- Niamtu J. Rejuvenation of the lip and perioral areas. In: Bell WH, Guerroro CA, eds. Distraction Osteogenesis of the Facial Skeleton. Ontario, Canada: BC Decker Inc; 2007:38-48.
- Tansatit T, Apinuntrum P, Phetudom T. A typical pattern of the labial arteries with implication for lip augmentation with injectable fillers. Aesthet Plast Surg. 2014;38:1083-1089.
- Sadick NS, Karcher C, Palmisano L. Cosmetic dermatology of the aging face. Clin Dermatol. 2009;27(suppl):S3-S12.
- Ali MJ, Ende K, Mass CS. Perioral rejuvenation and lip augmentation. Facial Plast Surg Clin N Am. 2007;15:491-500.
- Chien AL, Qi J, Cheng N, et al. Perioral wrinkles are associated with female gender, aging, and smoking: development of a gender-specific photonumeric scale. J Am Acad Dermatol. 2016;74:924-930.
- Iblher N, Stark GB, Penna V. The aging perioral region—do we really know what is happening? J Nutr Health Aging. 2012;16:581-585.
- Sarnoff DS, Gotkin RH. Six steps to the “perfect” lip. J Drugs Dermatol. 2012;11:1081-1088.
- American Society of Plastic Surgeons. 2015 Cosmetic plastic surgery statistics. https://d2wirczt3b6wjm.cloudfront.net/News/Statistics/2015/cosmetic-procedure-trends-2015.pdf. Published February 26, 2015. Accessed October 5, 2016.
- Abduljabbar MH, Basendwh MA. Complications of hyaluronic acid fillers and their managements. J Dermatol Surg. 2016;20:1-7.
- Luebberding S, Alexiades-Armenakas M. Facial volume augmentation in 2014: overview of different filler options. J Drugs Dermatol. 2013;12:1339-1344.
- Huang Attenello N, Mass CS. Injectable fillers: review of material and properties. Facial Plast Surg. 2015;31:29-34.
- Eccleston D, Murphy DK. Juvéderm Volbella in the perioral area: a 12-month perspective, multicenter, open-label study. Clin Cosmet Investig Dermatol. 2012;5:167-172.
- Raspaldo H, Chantrey J, Belhaouari L, et al. Lip and perioral enhancement: a 12-month prospective, randomized, controlled study. J Drugs Dermatol. 2015;14:1444-1452.
- Soft tissue fillers approved by the center for devices and radiological health. US Food and Drug Administration website. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed October 5, 2016.
- Butterwick K, Marmur E, Narurkar V, et al. HYC-24L demonstrates greater effectiveness with less pain than CPM-22.5 for treatment of perioral lines in a randomized controlled trial. Dermatol Surg. 2015;41:1351-1360.
- San Miguel Moragas J, Reddy RR, Hernández Alfaro F, et al. Systematic review of “filling” procedures for lip augmentation regarding types of material, outcomes and complications. J Craniomaxillofac Surg. 2015;43:883-906.
- Cohen JL, Thomas J, Paradkar D, et al. An interrater and intrarater reliability study of 3 photographic scales for the classification of perioral aesthetic features. Dermatol Surg. 2014;40:663-670.
- Broder KW, Cohen SR. An overview of permanent and semipermanent fillers. Plast Reconstr Surg. 2006;118(3 suppl):7S-14S.
- Sarnoff DS, Saini R, Gotkin RH. Comparison of filling agents for lip augmentation. Aesthet Surg J. 2008;28:556-563.
- Moscona RA, Fodor L. A retrospective study on liquid injectable silicone for lip augmentation: long-term results and patient satisfaction. J Plast Reconstr Aesthet Surg. 2010;63:1694-1698.
- Bertucci V, Lynde CB. Current concepts in the use of small-particle hyaluronic acid. Plast Reconstr Surg. 2015;136(5 suppl):132S-138S.
- Wilson AJ, Taglienti AJ, Chang CS, et al. Current applications of facial volumization with fillers. Plast Reconstr Surg. 2016;137:E872-E889.
- Dewandre L, Caperton C, Fulton J. Filler injections with the blunt-tip microcannula compared to the sharp hypodermic needle. J Drugs Dermatol. 2012;11:1098-1103.
- Carruthers JD, Glogau RG, Blitzer A; Facial Aesthetics Consensus Group Faculty. Advances in facial rejuvenation: botulinum toxin type A, hyaluronic acid dermal fillers, and combination therapies-consensus recommendations. Plast Reconstr Surg. 2008;121(5 suppl):5S-30S.
- Wu DC, Fabi SG, Goldman MP. Neurotoxins: current concepts in cosmetic use on the face and neck-lower face. Plast Reconstr Surg. 2015;136(5 suppl):76S-79S.
- Carruthers A, Carruthers J, Monheit GD, et al. Multicenter, randomized, parallel-group study of the safety and effectiveness of onabotulinumtoxin A and hyaluronic acid dermal fillers (24-mg/mL smooth, cohesive gel) alone and in combination for lower facial rejuvenation. Dermatol Surg. 2010;36:2121-2134.
Historically, a variety of tools have been used to alter one’s appearance for cultural or religious purposes or to conform to standards of beauty. As a defining feature of the face, the lips provide a unique opportunity for facial aesthetic enhancement. There has been a paradigm shift in medicine favoring preventative health and a desire to slow and even reverse the aging process.1 Acknowledging that product technology, skill sets, and cultural ideals continually evolve, this article highlights perioral anatomy, explains aging of the lower face, and reviews techniques to achieve perioral rejuvenation through volume restoration and muscle control.
Perioral Anatomy
The layers of the lips include the epidermis, subcutaneous tissue, orbicularis oris muscle fibers, and mucosa. The upper lip extends from the base of the nose to the mucosa inferiorly and to the nasolabial folds laterally. The curvilinear lower lip extends from the mucosa to the mandible inferiorly and to the oral commissures laterally.2 Circumferential at the vermilion-cutaneous junction, a raised area of pale skin known as the white roll accentuates the vermilion border and provides an important landmark during lip augmentation.3 At the upper lip, this elevation of the vermilion joins at a V-shaped depression centrally to form the Cupid’s bow. The cutaneous upper lip has 2 raised vertical pillars known as the philtral columns, which are formed from decussating fibers of the orbicularis oris muscle.2 The resultant midline depression is the philtrum. These defining features of the upper lip are to be preserved during augmentation procedures (Figure 1).4

The superior and inferior labial arteries, both branches of the facial artery, supply the upper and lower lip, respectively. The anastomotic arch of the superior labial artery is susceptible to injury from deep injection of the upper lip between the muscle layer and mucosa; therefore, caution must be exercised in this area.5 Injections into the vermilion and lower lip can be safely performed with less concern for vascular compromise. The vermilion derives its red color from the translucency of capillaries in the superficial papillae.2 The capillary plexus at the papillae and rich sensory nerve network render the lip a highly vascular and sensitive structure.
Aging of the Lower Face
Subcutaneous fat atrophy, loss of elasticity, gravitational forces, and remodeling of the skeletal foundation all contribute to aging of the lower face. Starting as early as the third decade of life, intrinsic factors including hormonal changes and genetically determined processes produce alterations in skin quality and structure. Similarly, extrinsic aging through environmental influences, namely exposure to UV radiation and smoking, accelerate the loss of skin integrity.6
The decreased laxity of the skin in combination with repeated contraction of the orbicularis oris muscle results in perioral rhytides.7 For women in particular, vertically oriented perioral rhytides develop above the vermilion; terminal hair follicles, thicker skin, and a greater density of subcutaneous fat are presumptive protective factors for males.8 With time, the cutaneous portion of the upper lip lengthens and there is redistribution of volume with effacement of the upper lip vermilion.9 Additionally, the demarcation of the vermilion becomes blurred secondary to pallor, flattening of the philtral columns, and loss of projection of the Cupid’s bow.10
Downturning of the oral commissures is observed secondary to a combination of gravity, bone resorption, and soft tissue volume loss. Hyperactivity of the depressor anguli oris muscle exacerbates the mesolabial folds, producing marionette lines and a saddened expression.7 With ongoing volume loss and ligament laxity, tissue redistributes near the jaws and chin, giving rise to jowls. Similarly, perioral volume loss and descent of the malar fat-pad deepen the nasolabial folds in the aging midface.6
The main objective of perioral rejuvenation is to reinstate a harmonious refreshed look to the lower face; however, aesthetic analysis should occur within the context of the face as a whole, as the lips should complement the surrounding perioral cosmetic unit and overall skeletal foundation of the face. To accomplish this goal, the dermatologist’s armamentarium contains a broad variety of approaches including restriction of muscle movement, volume restoration, and surface contouring.
Volume Restoration
Treatment Options
In 2015, hyaluronic acid (HA) fillers constituted 80% of all injectable soft-tissue fillers, an 8% increase from 2014.11 Hyaluronic acid has achieved immense popularity as a temporary dermal filler given its biocompatibility, longevity, and reversibility via hyaluronidase.12
Hyaluronic acid is a naturally occurring glycosaminoglycan that comprises the connective tissue matrix. The molecular composition affords HA its hydrophilic property, which augments dermal volume.7 Endogenous HA has a short half-life, and chemical modification by a cross-linking process extends longevity by 6 to 12 months. The various HA fillers are distinguished by method of purification, size of molecules, concentration and degree of cross-linking, and viscosity.7,13,14 These differences dictate overall clinical performance such as flow properties, longevity, and stability. As a general rule, a high-viscosity product is more appropriate for deeper augmentation; fillers with low viscosity are more appropriate for correction of shallow defects.1 Table 1 lists the HA fillers that are currently approved by the US Food and Drug Administration for lip augmentation and/or perioral rhytides in adults 21 years and older.15-17
Randomized controlled trials comparing the efficacy, longevity, and tolerability of different HA products are lacking in the literature and, where present, have strong industry influence.18,19 The advent of assessment scales has provided an objective evaluation of perioral and lip augmentation, facilitating comparisons between products in both clinical research and practice.20

Semipermanent biostimulatory dermal fillers such as calcium hydroxylapatite and poly-L-lactic acid are not recommended for lip augmentation due to an increased incidence of submucosal nodule formation.6,14,21 Likewise, permanent fillers are not recommended given their irreversibility and risk of nodule formation around the lips.14,22 Nonetheless, liquid silicone (purified polydimethylsiloxane) administered via a microdroplet technique (0.01 mL of silicone at a time, no more than 1 cc per lip per session) has been used off label as a permanent filling agent for lip augmentation with limited complications.23 Regardless, trepidations about its use with respect to reported risks continue to limit its application.22
Similarly, surgical lip implants such as expanded polytetrafluoroethylene is an option for a subset of patients desiring permanent enhancement but are less commonly utilized given the side-effect profile, irreversibility, and relatively invasive nature of the procedure.22 Lastly, autologous fat transfer has been used in correction of the nasolabial and mesolabial folds as well as in lip augmentation; however, irregular surface contours and unpredictable longevity secondary to postinjection resorption (20%–90%) has limited its popularity.3,14,21
HA Injection Technique
With respect to HA fillers in the perioral area, numerous approaches have been described.10,22 The techniques in Table 2 provide a foundation for lip rejuvenation.

Several injection techniques exist, including serial puncture, linear threading, cross-hatching, and fanning in a retrograde or anterograde manner.24 A blunt microcannula (27 gauge, 38 mm) may be used in place of sharp needles and offers the benefit of increased patient comfort, reduced edema and ecchymosis, and shortened recovery period.25,26 Gentle massage of the product after injection can assist with an even contour. Lastly, a key determinant of successful outcomes is using an adequate volume of HA filler (1–2 mL for shaping the vermilion border and volumizing the lips).27 Figure 2 highlights a clinical example of HA filler for lip augmentation.

Fortunately, most complications encountered with HA lip augmentation are mild and transient. The most commonly observed side effects include injection-site reactions such as pain, erythema, and edema. Similarly, most adverse effects are related to injection technique. All HA fillers are prone to the Tyndall effect, a consequence of too superficial an injection plane. Patients with history of recurrent herpes simplex virus infections should receive prophylactic antiviral therapy.12
Muscle Control
An emerging concept in rejuvenation of the lower face recognizes not only restoration of volume but also control of muscle movement. Local injection of botulinum toxin type A induces relaxation of hyperfunctional facial muscles through temporary inhibition of neurotransmitter release.6 The potential for paralysis of the oral cavity may limit the application of botulinum toxin type A in that region.7 Nonetheless, the off-label potential of botulinum toxin type A has expanded to include several targets in the lower face. The orbicularis oris muscle is targeted to soften perioral rhytides. Conservative dosing (1–2 U per lip quadrant or approximately 5 U total) and superficial injection is emphasized in this area.27 Similarly, the depressor anguli oris muscle is targeted by injection of 4 U bilaterally to soften the marionette lines. In the chin area, the mentalis muscle can be targeted by injection of 2 U deep into each belly of the muscle to reduce the mental crease and dimpling.28 Combination treatment with dermal filler and neurotoxin demonstrates effects that last longer than either modality alone without additional adverse events.29 With combination therapy, guidelines suggest treating with filler first.27
Conclusion
A greater understanding of the extrinsic and intrinsic factors that contribute to the structural and surface changes of the aging face coupled with a preference for minimally invasive procedures has revolutionized the dermatologist’s approach to perioral rejuvenation. Serving as a focal point of the face, the lips and perioral skin are well poised to benefit from this paradigm shift. A multifaceted approach utilizing dermal fillers and neurotoxins may be most appropriate and has demonstrated optimal outcomes in facial aesthetics.
Historically, a variety of tools have been used to alter one’s appearance for cultural or religious purposes or to conform to standards of beauty. As a defining feature of the face, the lips provide a unique opportunity for facial aesthetic enhancement. There has been a paradigm shift in medicine favoring preventative health and a desire to slow and even reverse the aging process.1 Acknowledging that product technology, skill sets, and cultural ideals continually evolve, this article highlights perioral anatomy, explains aging of the lower face, and reviews techniques to achieve perioral rejuvenation through volume restoration and muscle control.
Perioral Anatomy
The layers of the lips include the epidermis, subcutaneous tissue, orbicularis oris muscle fibers, and mucosa. The upper lip extends from the base of the nose to the mucosa inferiorly and to the nasolabial folds laterally. The curvilinear lower lip extends from the mucosa to the mandible inferiorly and to the oral commissures laterally.2 Circumferential at the vermilion-cutaneous junction, a raised area of pale skin known as the white roll accentuates the vermilion border and provides an important landmark during lip augmentation.3 At the upper lip, this elevation of the vermilion joins at a V-shaped depression centrally to form the Cupid’s bow. The cutaneous upper lip has 2 raised vertical pillars known as the philtral columns, which are formed from decussating fibers of the orbicularis oris muscle.2 The resultant midline depression is the philtrum. These defining features of the upper lip are to be preserved during augmentation procedures (Figure 1).4

The superior and inferior labial arteries, both branches of the facial artery, supply the upper and lower lip, respectively. The anastomotic arch of the superior labial artery is susceptible to injury from deep injection of the upper lip between the muscle layer and mucosa; therefore, caution must be exercised in this area.5 Injections into the vermilion and lower lip can be safely performed with less concern for vascular compromise. The vermilion derives its red color from the translucency of capillaries in the superficial papillae.2 The capillary plexus at the papillae and rich sensory nerve network render the lip a highly vascular and sensitive structure.
Aging of the Lower Face
Subcutaneous fat atrophy, loss of elasticity, gravitational forces, and remodeling of the skeletal foundation all contribute to aging of the lower face. Starting as early as the third decade of life, intrinsic factors including hormonal changes and genetically determined processes produce alterations in skin quality and structure. Similarly, extrinsic aging through environmental influences, namely exposure to UV radiation and smoking, accelerate the loss of skin integrity.6
The decreased laxity of the skin in combination with repeated contraction of the orbicularis oris muscle results in perioral rhytides.7 For women in particular, vertically oriented perioral rhytides develop above the vermilion; terminal hair follicles, thicker skin, and a greater density of subcutaneous fat are presumptive protective factors for males.8 With time, the cutaneous portion of the upper lip lengthens and there is redistribution of volume with effacement of the upper lip vermilion.9 Additionally, the demarcation of the vermilion becomes blurred secondary to pallor, flattening of the philtral columns, and loss of projection of the Cupid’s bow.10
Downturning of the oral commissures is observed secondary to a combination of gravity, bone resorption, and soft tissue volume loss. Hyperactivity of the depressor anguli oris muscle exacerbates the mesolabial folds, producing marionette lines and a saddened expression.7 With ongoing volume loss and ligament laxity, tissue redistributes near the jaws and chin, giving rise to jowls. Similarly, perioral volume loss and descent of the malar fat-pad deepen the nasolabial folds in the aging midface.6
The main objective of perioral rejuvenation is to reinstate a harmonious refreshed look to the lower face; however, aesthetic analysis should occur within the context of the face as a whole, as the lips should complement the surrounding perioral cosmetic unit and overall skeletal foundation of the face. To accomplish this goal, the dermatologist’s armamentarium contains a broad variety of approaches including restriction of muscle movement, volume restoration, and surface contouring.
Volume Restoration
Treatment Options
In 2015, hyaluronic acid (HA) fillers constituted 80% of all injectable soft-tissue fillers, an 8% increase from 2014.11 Hyaluronic acid has achieved immense popularity as a temporary dermal filler given its biocompatibility, longevity, and reversibility via hyaluronidase.12
Hyaluronic acid is a naturally occurring glycosaminoglycan that comprises the connective tissue matrix. The molecular composition affords HA its hydrophilic property, which augments dermal volume.7 Endogenous HA has a short half-life, and chemical modification by a cross-linking process extends longevity by 6 to 12 months. The various HA fillers are distinguished by method of purification, size of molecules, concentration and degree of cross-linking, and viscosity.7,13,14 These differences dictate overall clinical performance such as flow properties, longevity, and stability. As a general rule, a high-viscosity product is more appropriate for deeper augmentation; fillers with low viscosity are more appropriate for correction of shallow defects.1 Table 1 lists the HA fillers that are currently approved by the US Food and Drug Administration for lip augmentation and/or perioral rhytides in adults 21 years and older.15-17
Randomized controlled trials comparing the efficacy, longevity, and tolerability of different HA products are lacking in the literature and, where present, have strong industry influence.18,19 The advent of assessment scales has provided an objective evaluation of perioral and lip augmentation, facilitating comparisons between products in both clinical research and practice.20

Semipermanent biostimulatory dermal fillers such as calcium hydroxylapatite and poly-L-lactic acid are not recommended for lip augmentation due to an increased incidence of submucosal nodule formation.6,14,21 Likewise, permanent fillers are not recommended given their irreversibility and risk of nodule formation around the lips.14,22 Nonetheless, liquid silicone (purified polydimethylsiloxane) administered via a microdroplet technique (0.01 mL of silicone at a time, no more than 1 cc per lip per session) has been used off label as a permanent filling agent for lip augmentation with limited complications.23 Regardless, trepidations about its use with respect to reported risks continue to limit its application.22
Similarly, surgical lip implants such as expanded polytetrafluoroethylene is an option for a subset of patients desiring permanent enhancement but are less commonly utilized given the side-effect profile, irreversibility, and relatively invasive nature of the procedure.22 Lastly, autologous fat transfer has been used in correction of the nasolabial and mesolabial folds as well as in lip augmentation; however, irregular surface contours and unpredictable longevity secondary to postinjection resorption (20%–90%) has limited its popularity.3,14,21
HA Injection Technique
With respect to HA fillers in the perioral area, numerous approaches have been described.10,22 The techniques in Table 2 provide a foundation for lip rejuvenation.

Several injection techniques exist, including serial puncture, linear threading, cross-hatching, and fanning in a retrograde or anterograde manner.24 A blunt microcannula (27 gauge, 38 mm) may be used in place of sharp needles and offers the benefit of increased patient comfort, reduced edema and ecchymosis, and shortened recovery period.25,26 Gentle massage of the product after injection can assist with an even contour. Lastly, a key determinant of successful outcomes is using an adequate volume of HA filler (1–2 mL for shaping the vermilion border and volumizing the lips).27 Figure 2 highlights a clinical example of HA filler for lip augmentation.

Fortunately, most complications encountered with HA lip augmentation are mild and transient. The most commonly observed side effects include injection-site reactions such as pain, erythema, and edema. Similarly, most adverse effects are related to injection technique. All HA fillers are prone to the Tyndall effect, a consequence of too superficial an injection plane. Patients with history of recurrent herpes simplex virus infections should receive prophylactic antiviral therapy.12
Muscle Control
An emerging concept in rejuvenation of the lower face recognizes not only restoration of volume but also control of muscle movement. Local injection of botulinum toxin type A induces relaxation of hyperfunctional facial muscles through temporary inhibition of neurotransmitter release.6 The potential for paralysis of the oral cavity may limit the application of botulinum toxin type A in that region.7 Nonetheless, the off-label potential of botulinum toxin type A has expanded to include several targets in the lower face. The orbicularis oris muscle is targeted to soften perioral rhytides. Conservative dosing (1–2 U per lip quadrant or approximately 5 U total) and superficial injection is emphasized in this area.27 Similarly, the depressor anguli oris muscle is targeted by injection of 4 U bilaterally to soften the marionette lines. In the chin area, the mentalis muscle can be targeted by injection of 2 U deep into each belly of the muscle to reduce the mental crease and dimpling.28 Combination treatment with dermal filler and neurotoxin demonstrates effects that last longer than either modality alone without additional adverse events.29 With combination therapy, guidelines suggest treating with filler first.27
Conclusion
A greater understanding of the extrinsic and intrinsic factors that contribute to the structural and surface changes of the aging face coupled with a preference for minimally invasive procedures has revolutionized the dermatologist’s approach to perioral rejuvenation. Serving as a focal point of the face, the lips and perioral skin are well poised to benefit from this paradigm shift. A multifaceted approach utilizing dermal fillers and neurotoxins may be most appropriate and has demonstrated optimal outcomes in facial aesthetics.
- Buck DW, Alam M, Kim JYS. Injectable fillers for facial rejuvenation: a review. J Plast Reconstr Aesthet Surg. 2009;62:11-18.
- Guareschi M, Stella E. Lips. In: Goisis M, ed. Injections in Aesthetic Medicine. Milan, Italy: Springer; 2014:125-136.
- Byrne PJ, Hilger PA. Lip augmentation. Facial Plast Surg. 2004;20:31-38.
- Niamtu J. Rejuvenation of the lip and perioral areas. In: Bell WH, Guerroro CA, eds. Distraction Osteogenesis of the Facial Skeleton. Ontario, Canada: BC Decker Inc; 2007:38-48.
- Tansatit T, Apinuntrum P, Phetudom T. A typical pattern of the labial arteries with implication for lip augmentation with injectable fillers. Aesthet Plast Surg. 2014;38:1083-1089.
- Sadick NS, Karcher C, Palmisano L. Cosmetic dermatology of the aging face. Clin Dermatol. 2009;27(suppl):S3-S12.
- Ali MJ, Ende K, Mass CS. Perioral rejuvenation and lip augmentation. Facial Plast Surg Clin N Am. 2007;15:491-500.
- Chien AL, Qi J, Cheng N, et al. Perioral wrinkles are associated with female gender, aging, and smoking: development of a gender-specific photonumeric scale. J Am Acad Dermatol. 2016;74:924-930.
- Iblher N, Stark GB, Penna V. The aging perioral region—do we really know what is happening? J Nutr Health Aging. 2012;16:581-585.
- Sarnoff DS, Gotkin RH. Six steps to the “perfect” lip. J Drugs Dermatol. 2012;11:1081-1088.
- American Society of Plastic Surgeons. 2015 Cosmetic plastic surgery statistics. https://d2wirczt3b6wjm.cloudfront.net/News/Statistics/2015/cosmetic-procedure-trends-2015.pdf. Published February 26, 2015. Accessed October 5, 2016.
- Abduljabbar MH, Basendwh MA. Complications of hyaluronic acid fillers and their managements. J Dermatol Surg. 2016;20:1-7.
- Luebberding S, Alexiades-Armenakas M. Facial volume augmentation in 2014: overview of different filler options. J Drugs Dermatol. 2013;12:1339-1344.
- Huang Attenello N, Mass CS. Injectable fillers: review of material and properties. Facial Plast Surg. 2015;31:29-34.
- Eccleston D, Murphy DK. Juvéderm Volbella in the perioral area: a 12-month perspective, multicenter, open-label study. Clin Cosmet Investig Dermatol. 2012;5:167-172.
- Raspaldo H, Chantrey J, Belhaouari L, et al. Lip and perioral enhancement: a 12-month prospective, randomized, controlled study. J Drugs Dermatol. 2015;14:1444-1452.
- Soft tissue fillers approved by the center for devices and radiological health. US Food and Drug Administration website. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed October 5, 2016.
- Butterwick K, Marmur E, Narurkar V, et al. HYC-24L demonstrates greater effectiveness with less pain than CPM-22.5 for treatment of perioral lines in a randomized controlled trial. Dermatol Surg. 2015;41:1351-1360.
- San Miguel Moragas J, Reddy RR, Hernández Alfaro F, et al. Systematic review of “filling” procedures for lip augmentation regarding types of material, outcomes and complications. J Craniomaxillofac Surg. 2015;43:883-906.
- Cohen JL, Thomas J, Paradkar D, et al. An interrater and intrarater reliability study of 3 photographic scales for the classification of perioral aesthetic features. Dermatol Surg. 2014;40:663-670.
- Broder KW, Cohen SR. An overview of permanent and semipermanent fillers. Plast Reconstr Surg. 2006;118(3 suppl):7S-14S.
- Sarnoff DS, Saini R, Gotkin RH. Comparison of filling agents for lip augmentation. Aesthet Surg J. 2008;28:556-563.
- Moscona RA, Fodor L. A retrospective study on liquid injectable silicone for lip augmentation: long-term results and patient satisfaction. J Plast Reconstr Aesthet Surg. 2010;63:1694-1698.
- Bertucci V, Lynde CB. Current concepts in the use of small-particle hyaluronic acid. Plast Reconstr Surg. 2015;136(5 suppl):132S-138S.
- Wilson AJ, Taglienti AJ, Chang CS, et al. Current applications of facial volumization with fillers. Plast Reconstr Surg. 2016;137:E872-E889.
- Dewandre L, Caperton C, Fulton J. Filler injections with the blunt-tip microcannula compared to the sharp hypodermic needle. J Drugs Dermatol. 2012;11:1098-1103.
- Carruthers JD, Glogau RG, Blitzer A; Facial Aesthetics Consensus Group Faculty. Advances in facial rejuvenation: botulinum toxin type A, hyaluronic acid dermal fillers, and combination therapies-consensus recommendations. Plast Reconstr Surg. 2008;121(5 suppl):5S-30S.
- Wu DC, Fabi SG, Goldman MP. Neurotoxins: current concepts in cosmetic use on the face and neck-lower face. Plast Reconstr Surg. 2015;136(5 suppl):76S-79S.
- Carruthers A, Carruthers J, Monheit GD, et al. Multicenter, randomized, parallel-group study of the safety and effectiveness of onabotulinumtoxin A and hyaluronic acid dermal fillers (24-mg/mL smooth, cohesive gel) alone and in combination for lower facial rejuvenation. Dermatol Surg. 2010;36:2121-2134.
- Buck DW, Alam M, Kim JYS. Injectable fillers for facial rejuvenation: a review. J Plast Reconstr Aesthet Surg. 2009;62:11-18.
- Guareschi M, Stella E. Lips. In: Goisis M, ed. Injections in Aesthetic Medicine. Milan, Italy: Springer; 2014:125-136.
- Byrne PJ, Hilger PA. Lip augmentation. Facial Plast Surg. 2004;20:31-38.
- Niamtu J. Rejuvenation of the lip and perioral areas. In: Bell WH, Guerroro CA, eds. Distraction Osteogenesis of the Facial Skeleton. Ontario, Canada: BC Decker Inc; 2007:38-48.
- Tansatit T, Apinuntrum P, Phetudom T. A typical pattern of the labial arteries with implication for lip augmentation with injectable fillers. Aesthet Plast Surg. 2014;38:1083-1089.
- Sadick NS, Karcher C, Palmisano L. Cosmetic dermatology of the aging face. Clin Dermatol. 2009;27(suppl):S3-S12.
- Ali MJ, Ende K, Mass CS. Perioral rejuvenation and lip augmentation. Facial Plast Surg Clin N Am. 2007;15:491-500.
- Chien AL, Qi J, Cheng N, et al. Perioral wrinkles are associated with female gender, aging, and smoking: development of a gender-specific photonumeric scale. J Am Acad Dermatol. 2016;74:924-930.
- Iblher N, Stark GB, Penna V. The aging perioral region—do we really know what is happening? J Nutr Health Aging. 2012;16:581-585.
- Sarnoff DS, Gotkin RH. Six steps to the “perfect” lip. J Drugs Dermatol. 2012;11:1081-1088.
- American Society of Plastic Surgeons. 2015 Cosmetic plastic surgery statistics. https://d2wirczt3b6wjm.cloudfront.net/News/Statistics/2015/cosmetic-procedure-trends-2015.pdf. Published February 26, 2015. Accessed October 5, 2016.
- Abduljabbar MH, Basendwh MA. Complications of hyaluronic acid fillers and their managements. J Dermatol Surg. 2016;20:1-7.
- Luebberding S, Alexiades-Armenakas M. Facial volume augmentation in 2014: overview of different filler options. J Drugs Dermatol. 2013;12:1339-1344.
- Huang Attenello N, Mass CS. Injectable fillers: review of material and properties. Facial Plast Surg. 2015;31:29-34.
- Eccleston D, Murphy DK. Juvéderm Volbella in the perioral area: a 12-month perspective, multicenter, open-label study. Clin Cosmet Investig Dermatol. 2012;5:167-172.
- Raspaldo H, Chantrey J, Belhaouari L, et al. Lip and perioral enhancement: a 12-month prospective, randomized, controlled study. J Drugs Dermatol. 2015;14:1444-1452.
- Soft tissue fillers approved by the center for devices and radiological health. US Food and Drug Administration website. http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CosmeticDevices/WrinkleFillers/ucm227749.htm. Updated July 27, 2015. Accessed October 5, 2016.
- Butterwick K, Marmur E, Narurkar V, et al. HYC-24L demonstrates greater effectiveness with less pain than CPM-22.5 for treatment of perioral lines in a randomized controlled trial. Dermatol Surg. 2015;41:1351-1360.
- San Miguel Moragas J, Reddy RR, Hernández Alfaro F, et al. Systematic review of “filling” procedures for lip augmentation regarding types of material, outcomes and complications. J Craniomaxillofac Surg. 2015;43:883-906.
- Cohen JL, Thomas J, Paradkar D, et al. An interrater and intrarater reliability study of 3 photographic scales for the classification of perioral aesthetic features. Dermatol Surg. 2014;40:663-670.
- Broder KW, Cohen SR. An overview of permanent and semipermanent fillers. Plast Reconstr Surg. 2006;118(3 suppl):7S-14S.
- Sarnoff DS, Saini R, Gotkin RH. Comparison of filling agents for lip augmentation. Aesthet Surg J. 2008;28:556-563.
- Moscona RA, Fodor L. A retrospective study on liquid injectable silicone for lip augmentation: long-term results and patient satisfaction. J Plast Reconstr Aesthet Surg. 2010;63:1694-1698.
- Bertucci V, Lynde CB. Current concepts in the use of small-particle hyaluronic acid. Plast Reconstr Surg. 2015;136(5 suppl):132S-138S.
- Wilson AJ, Taglienti AJ, Chang CS, et al. Current applications of facial volumization with fillers. Plast Reconstr Surg. 2016;137:E872-E889.
- Dewandre L, Caperton C, Fulton J. Filler injections with the blunt-tip microcannula compared to the sharp hypodermic needle. J Drugs Dermatol. 2012;11:1098-1103.
- Carruthers JD, Glogau RG, Blitzer A; Facial Aesthetics Consensus Group Faculty. Advances in facial rejuvenation: botulinum toxin type A, hyaluronic acid dermal fillers, and combination therapies-consensus recommendations. Plast Reconstr Surg. 2008;121(5 suppl):5S-30S.
- Wu DC, Fabi SG, Goldman MP. Neurotoxins: current concepts in cosmetic use on the face and neck-lower face. Plast Reconstr Surg. 2015;136(5 suppl):76S-79S.
- Carruthers A, Carruthers J, Monheit GD, et al. Multicenter, randomized, parallel-group study of the safety and effectiveness of onabotulinumtoxin A and hyaluronic acid dermal fillers (24-mg/mL smooth, cohesive gel) alone and in combination for lower facial rejuvenation. Dermatol Surg. 2010;36:2121-2134.
Practice Points
- Hyaluronic acid (HA) fillers are approved by the US Food and Drug Administration for lip augmentation and/or treatment of perioral rhytides in adults 21 years and older.
- Most complications encountered with HA lip augmentation are mild and transient and can include injection-site reactions such as pain, erythema, and edema.
- Combination treatment with dermal fillers and neurotoxins (off label) may demonstrate effects that last longer than either modality alone without additional adverse events.
Reasons Behind the Ink
Tattoos have been viewed as one of the most exotic forms of art for thousands of years. In ancient times, tattoos were used mainly for therapeutic and status purposes. According to British archeologist Joann Fletcher, the oldest evidence of tattoo use was found on the famous “Iceman,” a 5200-year-old frozen mummy that was discovered more than 20 years ago.1 Tattoos were thought to be a form of therapy used to decrease joint pain. On the other hand, the ancient Egyptians used tattoos as symbols of wealth and high status; surprisingly, only women were tattooed. Fletcher also reported that tattoos were used as a form of therapy during pregnancy in upper-class women.1
Tattoos have served different purposes in the last few centuries, making their way to the United States at the start of the 20th century.2 New York City became the tattoo capital of the country. During this early period, male artists often would tattoo their wives so that they could advertise their work. After the Prohibition era, tattoos became widely used within the US Military, becoming a way to show pride and patriotism.2
Due to the permanent nature of tattoos, we sought to understand the reasons for obtaining this particular genre of body art. The purpose of this study was to provide a greater understanding of the current demographics of individuals who get tattoos, looking at specific trends in age and level of education of those who get tattoos as well as the motivation for tattoo placement. As dermatologists, it is essential to understand this patient population to be able to provide services (ie, tattoo removal) in the safe setting of a physician’s office.
Methods
The study was conducted at a private dermatology clinic in the Chicago (Illinois) metropolitan area with no institutional review board approval. Between January 2011 and December 2012, local patients with at least 1 tattoo were asked, with assumed consent, to fill out an investigator-developed survey containing 18 multiple-choice questions regarding age, educational and family background, and other factors. The race and gender of the respondents as well as the number of patients who declined to complete the survey were not recorded.
Results
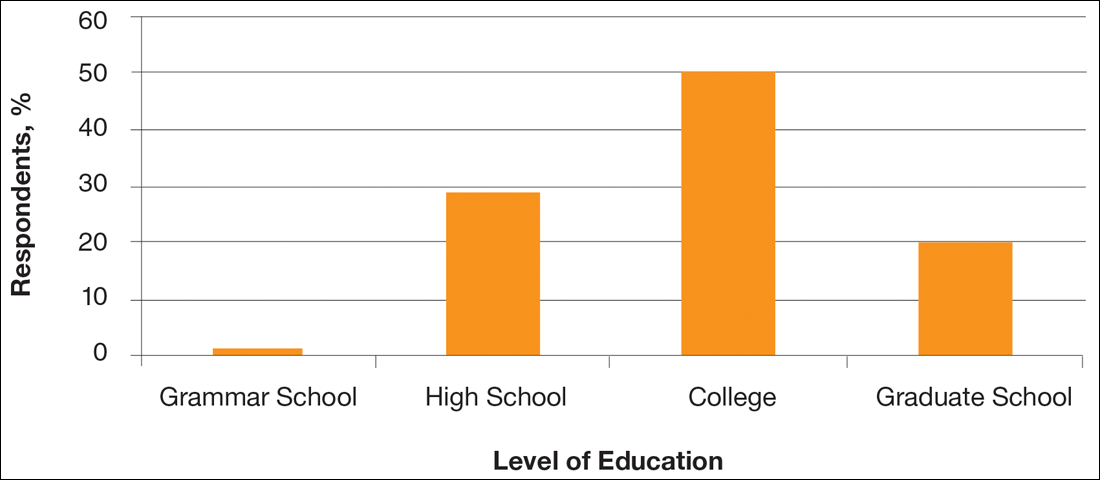
A total of 363 patients completed the in-person survey. Responses were tabulated and converted into percentages for comparison (N=363). Data analysis was divided into 3 parameters: education level, health concerns, and motivation for getting a tattoo. Figure 1 shows that 70% of respondents had obtained a college degree or higher.
With regard to health concerns associated with tattoos, the majority of respondents (71%) claimed they were not concerned with the health risks (eg, infection with human immunodeficiency virus or hepatitis C virus) associated with getting a tattoo. Also, only 6% of respondents admitted to being under the influence of drugs or alcohol at the time of getting a tattoo. Of 21 respondents who claimed drugs and/or alcohol were part of their tattoo experience, the highest level of education was high school in 7 respondents and 2 got their first tattoo when they were younger than 14 years.
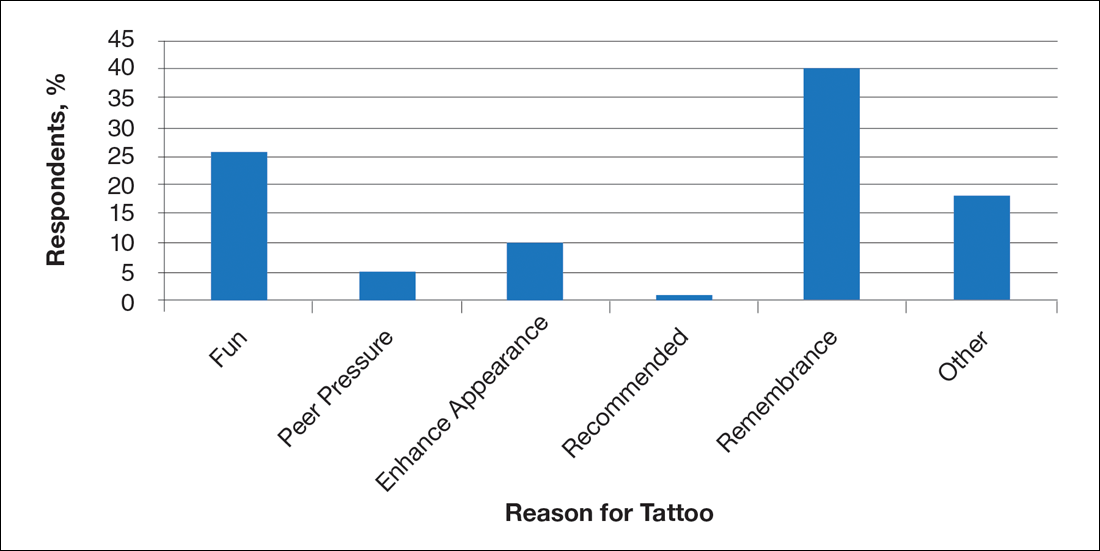
Survey results revealed that the majority of respondents got a tattoo as an act of rememberance (Figure 2). For example, one respondent reported getting a tattoo for religious purposes, while another got a tattoo to celebrate and mark each level of completed education (ie, high school, college, graduate school). However, a high percentage of respondents (26%) got a tattoo for fun.
Comment
Although ancient tattoos were used for therapeutic purposes, this study revealed that tattoos are now obtained by individuals with higher levels of education to remember a loved one or purely for enjoyment. The potential health risks associated with getting a tattoo did not deter the respondents in this study. Converse to the popular belief that individuals are under the influence of drugs and/or alcohol when getting a tattoo, our study found that only 6% of respondents were under the influence. A comparable trend was found among US military service members in a similar study.3 The majority of respondents did not regret their tattoos and did not report taking a mind-altering substance. Tattoos serve as a symbol of one’s proud individualism.3 However, a 2001 study found a correlation between greater use of alcohol and marijuana among college students with tattoos and piecings.4 These circumstances may lead patients to seek consultation from a dermatologist for tattoo removal. Therefore, it is important to have a better understanding of this particular patient population to facilitate care in an efficient manner.
Evaluation of the gender and race of survey respondents would be useful in the future. Financial status of respondents also may be explored, as wealth and status were used by the ancient Egyptians to determine who could get a tattoo. A follow-up analysis on removal of tattoos also will be explored in the future.
- Lineberry C. Tattoos: the ancient and mysterious history. Smithsonian. http://www.smithsonianmag.com/history-archaeology/tattoo.html. Published January 1, 2007. Accessed April 29, 2016.
- Bickerstaff L. Tattoos: fad, fashion, or folly? Odyssey. 2005;14:34-36.
- Lande RG, Bahroo BA, Soumoff A. United States military service members and their tattoos: a descriptive study. Mil Med. 2013;178:921-925.
- Forbes GB. College students with tattoos and piercings: motives, family experiences, personality factors, and perception by others. Psychol Rep. 2001;89:774-786.
Tattoos have been viewed as one of the most exotic forms of art for thousands of years. In ancient times, tattoos were used mainly for therapeutic and status purposes. According to British archeologist Joann Fletcher, the oldest evidence of tattoo use was found on the famous “Iceman,” a 5200-year-old frozen mummy that was discovered more than 20 years ago.1 Tattoos were thought to be a form of therapy used to decrease joint pain. On the other hand, the ancient Egyptians used tattoos as symbols of wealth and high status; surprisingly, only women were tattooed. Fletcher also reported that tattoos were used as a form of therapy during pregnancy in upper-class women.1
Tattoos have served different purposes in the last few centuries, making their way to the United States at the start of the 20th century.2 New York City became the tattoo capital of the country. During this early period, male artists often would tattoo their wives so that they could advertise their work. After the Prohibition era, tattoos became widely used within the US Military, becoming a way to show pride and patriotism.2
Due to the permanent nature of tattoos, we sought to understand the reasons for obtaining this particular genre of body art. The purpose of this study was to provide a greater understanding of the current demographics of individuals who get tattoos, looking at specific trends in age and level of education of those who get tattoos as well as the motivation for tattoo placement. As dermatologists, it is essential to understand this patient population to be able to provide services (ie, tattoo removal) in the safe setting of a physician’s office.
Methods
The study was conducted at a private dermatology clinic in the Chicago (Illinois) metropolitan area with no institutional review board approval. Between January 2011 and December 2012, local patients with at least 1 tattoo were asked, with assumed consent, to fill out an investigator-developed survey containing 18 multiple-choice questions regarding age, educational and family background, and other factors. The race and gender of the respondents as well as the number of patients who declined to complete the survey were not recorded.
Results
A total of 363 patients completed the in-person survey. Responses were tabulated and converted into percentages for comparison (N=363). Data analysis was divided into 3 parameters: education level, health concerns, and motivation for getting a tattoo. Figure 1 shows that 70% of respondents had obtained a college degree or higher.

With regard to health concerns associated with tattoos, the majority of respondents (71%) claimed they were not concerned with the health risks (eg, infection with human immunodeficiency virus or hepatitis C virus) associated with getting a tattoo. Also, only 6% of respondents admitted to being under the influence of drugs or alcohol at the time of getting a tattoo. Of 21 respondents who claimed drugs and/or alcohol were part of their tattoo experience, the highest level of education was high school in 7 respondents and 2 got their first tattoo when they were younger than 14 years.
Survey results revealed that the majority of respondents got a tattoo as an act of rememberance (Figure 2). For example, one respondent reported getting a tattoo for religious purposes, while another got a tattoo to celebrate and mark each level of completed education (ie, high school, college, graduate school). However, a high percentage of respondents (26%) got a tattoo for fun.

Comment
Although ancient tattoos were used for therapeutic purposes, this study revealed that tattoos are now obtained by individuals with higher levels of education to remember a loved one or purely for enjoyment. The potential health risks associated with getting a tattoo did not deter the respondents in this study. Converse to the popular belief that individuals are under the influence of drugs and/or alcohol when getting a tattoo, our study found that only 6% of respondents were under the influence. A comparable trend was found among US military service members in a similar study.3 The majority of respondents did not regret their tattoos and did not report taking a mind-altering substance. Tattoos serve as a symbol of one’s proud individualism.3 However, a 2001 study found a correlation between greater use of alcohol and marijuana among college students with tattoos and piecings.4 These circumstances may lead patients to seek consultation from a dermatologist for tattoo removal. Therefore, it is important to have a better understanding of this particular patient population to facilitate care in an efficient manner.
Evaluation of the gender and race of survey respondents would be useful in the future. Financial status of respondents also may be explored, as wealth and status were used by the ancient Egyptians to determine who could get a tattoo. A follow-up analysis on removal of tattoos also will be explored in the future.
Tattoos have been viewed as one of the most exotic forms of art for thousands of years. In ancient times, tattoos were used mainly for therapeutic and status purposes. According to British archeologist Joann Fletcher, the oldest evidence of tattoo use was found on the famous “Iceman,” a 5200-year-old frozen mummy that was discovered more than 20 years ago.1 Tattoos were thought to be a form of therapy used to decrease joint pain. On the other hand, the ancient Egyptians used tattoos as symbols of wealth and high status; surprisingly, only women were tattooed. Fletcher also reported that tattoos were used as a form of therapy during pregnancy in upper-class women.1
Tattoos have served different purposes in the last few centuries, making their way to the United States at the start of the 20th century.2 New York City became the tattoo capital of the country. During this early period, male artists often would tattoo their wives so that they could advertise their work. After the Prohibition era, tattoos became widely used within the US Military, becoming a way to show pride and patriotism.2
Due to the permanent nature of tattoos, we sought to understand the reasons for obtaining this particular genre of body art. The purpose of this study was to provide a greater understanding of the current demographics of individuals who get tattoos, looking at specific trends in age and level of education of those who get tattoos as well as the motivation for tattoo placement. As dermatologists, it is essential to understand this patient population to be able to provide services (ie, tattoo removal) in the safe setting of a physician’s office.
Methods
The study was conducted at a private dermatology clinic in the Chicago (Illinois) metropolitan area with no institutional review board approval. Between January 2011 and December 2012, local patients with at least 1 tattoo were asked, with assumed consent, to fill out an investigator-developed survey containing 18 multiple-choice questions regarding age, educational and family background, and other factors. The race and gender of the respondents as well as the number of patients who declined to complete the survey were not recorded.
Results
A total of 363 patients completed the in-person survey. Responses were tabulated and converted into percentages for comparison (N=363). Data analysis was divided into 3 parameters: education level, health concerns, and motivation for getting a tattoo. Figure 1 shows that 70% of respondents had obtained a college degree or higher.

With regard to health concerns associated with tattoos, the majority of respondents (71%) claimed they were not concerned with the health risks (eg, infection with human immunodeficiency virus or hepatitis C virus) associated with getting a tattoo. Also, only 6% of respondents admitted to being under the influence of drugs or alcohol at the time of getting a tattoo. Of 21 respondents who claimed drugs and/or alcohol were part of their tattoo experience, the highest level of education was high school in 7 respondents and 2 got their first tattoo when they were younger than 14 years.
Survey results revealed that the majority of respondents got a tattoo as an act of rememberance (Figure 2). For example, one respondent reported getting a tattoo for religious purposes, while another got a tattoo to celebrate and mark each level of completed education (ie, high school, college, graduate school). However, a high percentage of respondents (26%) got a tattoo for fun.

Comment
Although ancient tattoos were used for therapeutic purposes, this study revealed that tattoos are now obtained by individuals with higher levels of education to remember a loved one or purely for enjoyment. The potential health risks associated with getting a tattoo did not deter the respondents in this study. Converse to the popular belief that individuals are under the influence of drugs and/or alcohol when getting a tattoo, our study found that only 6% of respondents were under the influence. A comparable trend was found among US military service members in a similar study.3 The majority of respondents did not regret their tattoos and did not report taking a mind-altering substance. Tattoos serve as a symbol of one’s proud individualism.3 However, a 2001 study found a correlation between greater use of alcohol and marijuana among college students with tattoos and piecings.4 These circumstances may lead patients to seek consultation from a dermatologist for tattoo removal. Therefore, it is important to have a better understanding of this particular patient population to facilitate care in an efficient manner.
Evaluation of the gender and race of survey respondents would be useful in the future. Financial status of respondents also may be explored, as wealth and status were used by the ancient Egyptians to determine who could get a tattoo. A follow-up analysis on removal of tattoos also will be explored in the future.
- Lineberry C. Tattoos: the ancient and mysterious history. Smithsonian. http://www.smithsonianmag.com/history-archaeology/tattoo.html. Published January 1, 2007. Accessed April 29, 2016.
- Bickerstaff L. Tattoos: fad, fashion, or folly? Odyssey. 2005;14:34-36.
- Lande RG, Bahroo BA, Soumoff A. United States military service members and their tattoos: a descriptive study. Mil Med. 2013;178:921-925.
- Forbes GB. College students with tattoos and piercings: motives, family experiences, personality factors, and perception by others. Psychol Rep. 2001;89:774-786.
- Lineberry C. Tattoos: the ancient and mysterious history. Smithsonian. http://www.smithsonianmag.com/history-archaeology/tattoo.html. Published January 1, 2007. Accessed April 29, 2016.
- Bickerstaff L. Tattoos: fad, fashion, or folly? Odyssey. 2005;14:34-36.
- Lande RG, Bahroo BA, Soumoff A. United States military service members and their tattoos: a descriptive study. Mil Med. 2013;178:921-925.
- Forbes GB. College students with tattoos and piercings: motives, family experiences, personality factors, and perception by others. Psychol Rep. 2001;89:774-786.
Practice Points
- Individuals who get tattoos often are more educated and well informed than previously thought, more likely leading them to seek removal if desired.
- Our results indicate that tattoos are not regretted as often as previously speculated.
Renewal in Cosmetic Dermatology
It is an exciting time for dermatologists. In the 16 years that I have been in practice our knowledge of disease pathogenesis has increased and has shaped treatments that can now offer life-changing improvement for patients with extensive dermatologic disease. The everyday practice of cosmetic dermatology also has advanced. Sixteen years ago the only cosmetic options we had for our patients were bovine or human collagen injections, traditional CO2 laser resurfacing, and the older generations of pulsed dye lasers. Neuromodulators were just being introduced. We quickly learned the limitations and complications associated with these modalities. Collagen injections were directed at fine perioral lines and lasted 3 to 4 months. The worst complication would be a bruise or an allergic reaction to the bovine collagen. If we really needed to restore volume beyond the perioral regions, our only option was autologous fat transfer. CO2 laser resurfacing was used to firm skin, erase deep wrinkles, and improve sun damage, but its use was limited to older, fair-skinned individuals due to the inherent risk for hypopigmentation and depigmentation. Pulsed dye lasers similarly had no means of cooling the skin, thus they were limited to lighter-skinned individuals and had notable risk for blisters, burns, and hypopigmentation. Since then, our understanding of skin healing, laser-tissue interaction, and facial aging has driven the field to new heights of technological advances and safety. Just as I tell my patients and residents, there is no better time to be in this field than at this moment. Dermatologists have driven the advances behind many of the technologies that are now in widespread use among physicians in a variety of specialties.
Our understanding of facial aging has evolved to include the complex interplay of skeletal change, fat atrophy, and skin aging, which must all be considered when improving a patient’s appearance. Fillers have evolved in physical characteristics to give us the ability to choose between lift, spread, neocollagenesis, or water absorption. Thus, we can select the proper filler for the specific anatomic area we are rejuvenating.
Our understanding of photoaging and laser-tissue physics has allowed for the development of a newer generation of lasers ranging from fractionated lasers to noninvasive modalities that can safely be used in a variety of ethnic skin types to address acne scars, wrinkles, and inflammatory processes such as acne and rosacea. Similarly, the desire to tighten redundant skin has continued to drive ultrasound and radiofrequency technology and sparked growth in a newer field of cryolipolysis and chemical lipolysis agents.
Our dialogue with patients also has evolved to include the 4 R’s of antiaging: resurfacing the skin (eg, lasers, peels), refilling the lost volume (eg, fillers, fat), redraping the excess skin (eg, radiofrequency, ultrasound, laser, surgery), and relaxing dynamic lines (eg, neuromodulators). I propose an additional R: renewal! We must focus on the need for constant renewal so that patients maintain the results we have achieved. I apply the analogy of exercising to get into shape. One must go to the gym regularly to get to the desired level of fitness, but you do not stop exercising, otherwise you will quickly relapse to your former lack of fitness. Similarly, patients should receive the appropriate treatments to bring them to the desired level of rejuvenation, but then some form of constant renewal process is needed to maintain them at that level. Without the stimulation of the skin, the aging process continues and the cycle begins all over again.
In my practice, renewal is achieved by driving the skin to maintain the glow and smoothness that enhances the results of the fillers, neuromodulators, lasers, and peels that we have used. The skin is the first thing people notice. Without the glow, the patient will look good but not great. I tell patients that this part of the process is their responsibility. They must adhere to the skin care regimen specifically designed to address their needs. By incorporating the patient in the rejuvenation process, he/she is empowered to take control over the aging process and has grown more confident in you as a physician.
These are exciting times and there is still so much in the pipeline. By continually learning, reading, and attending workshops and meetings, you can make sure our specialty continue to be the leader in the antiaging field.
It is an exciting time for dermatologists. In the 16 years that I have been in practice our knowledge of disease pathogenesis has increased and has shaped treatments that can now offer life-changing improvement for patients with extensive dermatologic disease. The everyday practice of cosmetic dermatology also has advanced. Sixteen years ago the only cosmetic options we had for our patients were bovine or human collagen injections, traditional CO2 laser resurfacing, and the older generations of pulsed dye lasers. Neuromodulators were just being introduced. We quickly learned the limitations and complications associated with these modalities. Collagen injections were directed at fine perioral lines and lasted 3 to 4 months. The worst complication would be a bruise or an allergic reaction to the bovine collagen. If we really needed to restore volume beyond the perioral regions, our only option was autologous fat transfer. CO2 laser resurfacing was used to firm skin, erase deep wrinkles, and improve sun damage, but its use was limited to older, fair-skinned individuals due to the inherent risk for hypopigmentation and depigmentation. Pulsed dye lasers similarly had no means of cooling the skin, thus they were limited to lighter-skinned individuals and had notable risk for blisters, burns, and hypopigmentation. Since then, our understanding of skin healing, laser-tissue interaction, and facial aging has driven the field to new heights of technological advances and safety. Just as I tell my patients and residents, there is no better time to be in this field than at this moment. Dermatologists have driven the advances behind many of the technologies that are now in widespread use among physicians in a variety of specialties.
Our understanding of facial aging has evolved to include the complex interplay of skeletal change, fat atrophy, and skin aging, which must all be considered when improving a patient’s appearance. Fillers have evolved in physical characteristics to give us the ability to choose between lift, spread, neocollagenesis, or water absorption. Thus, we can select the proper filler for the specific anatomic area we are rejuvenating.
Our understanding of photoaging and laser-tissue physics has allowed for the development of a newer generation of lasers ranging from fractionated lasers to noninvasive modalities that can safely be used in a variety of ethnic skin types to address acne scars, wrinkles, and inflammatory processes such as acne and rosacea. Similarly, the desire to tighten redundant skin has continued to drive ultrasound and radiofrequency technology and sparked growth in a newer field of cryolipolysis and chemical lipolysis agents.
Our dialogue with patients also has evolved to include the 4 R’s of antiaging: resurfacing the skin (eg, lasers, peels), refilling the lost volume (eg, fillers, fat), redraping the excess skin (eg, radiofrequency, ultrasound, laser, surgery), and relaxing dynamic lines (eg, neuromodulators). I propose an additional R: renewal! We must focus on the need for constant renewal so that patients maintain the results we have achieved. I apply the analogy of exercising to get into shape. One must go to the gym regularly to get to the desired level of fitness, but you do not stop exercising, otherwise you will quickly relapse to your former lack of fitness. Similarly, patients should receive the appropriate treatments to bring them to the desired level of rejuvenation, but then some form of constant renewal process is needed to maintain them at that level. Without the stimulation of the skin, the aging process continues and the cycle begins all over again.
In my practice, renewal is achieved by driving the skin to maintain the glow and smoothness that enhances the results of the fillers, neuromodulators, lasers, and peels that we have used. The skin is the first thing people notice. Without the glow, the patient will look good but not great. I tell patients that this part of the process is their responsibility. They must adhere to the skin care regimen specifically designed to address their needs. By incorporating the patient in the rejuvenation process, he/she is empowered to take control over the aging process and has grown more confident in you as a physician.
These are exciting times and there is still so much in the pipeline. By continually learning, reading, and attending workshops and meetings, you can make sure our specialty continue to be the leader in the antiaging field.
It is an exciting time for dermatologists. In the 16 years that I have been in practice our knowledge of disease pathogenesis has increased and has shaped treatments that can now offer life-changing improvement for patients with extensive dermatologic disease. The everyday practice of cosmetic dermatology also has advanced. Sixteen years ago the only cosmetic options we had for our patients were bovine or human collagen injections, traditional CO2 laser resurfacing, and the older generations of pulsed dye lasers. Neuromodulators were just being introduced. We quickly learned the limitations and complications associated with these modalities. Collagen injections were directed at fine perioral lines and lasted 3 to 4 months. The worst complication would be a bruise or an allergic reaction to the bovine collagen. If we really needed to restore volume beyond the perioral regions, our only option was autologous fat transfer. CO2 laser resurfacing was used to firm skin, erase deep wrinkles, and improve sun damage, but its use was limited to older, fair-skinned individuals due to the inherent risk for hypopigmentation and depigmentation. Pulsed dye lasers similarly had no means of cooling the skin, thus they were limited to lighter-skinned individuals and had notable risk for blisters, burns, and hypopigmentation. Since then, our understanding of skin healing, laser-tissue interaction, and facial aging has driven the field to new heights of technological advances and safety. Just as I tell my patients and residents, there is no better time to be in this field than at this moment. Dermatologists have driven the advances behind many of the technologies that are now in widespread use among physicians in a variety of specialties.
Our understanding of facial aging has evolved to include the complex interplay of skeletal change, fat atrophy, and skin aging, which must all be considered when improving a patient’s appearance. Fillers have evolved in physical characteristics to give us the ability to choose between lift, spread, neocollagenesis, or water absorption. Thus, we can select the proper filler for the specific anatomic area we are rejuvenating.
Our understanding of photoaging and laser-tissue physics has allowed for the development of a newer generation of lasers ranging from fractionated lasers to noninvasive modalities that can safely be used in a variety of ethnic skin types to address acne scars, wrinkles, and inflammatory processes such as acne and rosacea. Similarly, the desire to tighten redundant skin has continued to drive ultrasound and radiofrequency technology and sparked growth in a newer field of cryolipolysis and chemical lipolysis agents.
Our dialogue with patients also has evolved to include the 4 R’s of antiaging: resurfacing the skin (eg, lasers, peels), refilling the lost volume (eg, fillers, fat), redraping the excess skin (eg, radiofrequency, ultrasound, laser, surgery), and relaxing dynamic lines (eg, neuromodulators). I propose an additional R: renewal! We must focus on the need for constant renewal so that patients maintain the results we have achieved. I apply the analogy of exercising to get into shape. One must go to the gym regularly to get to the desired level of fitness, but you do not stop exercising, otherwise you will quickly relapse to your former lack of fitness. Similarly, patients should receive the appropriate treatments to bring them to the desired level of rejuvenation, but then some form of constant renewal process is needed to maintain them at that level. Without the stimulation of the skin, the aging process continues and the cycle begins all over again.
In my practice, renewal is achieved by driving the skin to maintain the glow and smoothness that enhances the results of the fillers, neuromodulators, lasers, and peels that we have used. The skin is the first thing people notice. Without the glow, the patient will look good but not great. I tell patients that this part of the process is their responsibility. They must adhere to the skin care regimen specifically designed to address their needs. By incorporating the patient in the rejuvenation process, he/she is empowered to take control over the aging process and has grown more confident in you as a physician.
These are exciting times and there is still so much in the pipeline. By continually learning, reading, and attending workshops and meetings, you can make sure our specialty continue to be the leader in the antiaging field.
Chemical Peels
Review the PDF of the fact sheet on Chemical Peels with board-relevant, easy-to-review material. This fact sheet will review the use of chemical peels for dermatologic indications.
Practice Questions
1. Which peel requires neutralization?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
2. Which peel contains resorcinol?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
3. Which peel would be the best treatment of severe actinic photodamage?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
4. Which peel would not be indicated for treatment of melasma in a patient with Fitzpatrick skin type IV?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
5. Which peel is a β-hydroxy acid?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
Answers to practice questions provided on next page
Practice Question Answers
1. Which peel requires neutralization?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
2. Which peel contains resorcinol?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
3. Which peel would be the best treatment of severe actinic photodamage?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
4. Which peel would not be indicated for treatment of melasma in a patient with Fitzpatrick skin type IV?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
5. Which peel is a β-hydroxy acid?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
Review the PDF of the fact sheet on Chemical Peels with board-relevant, easy-to-review material. This fact sheet will review the use of chemical peels for dermatologic indications.
Practice Questions
1. Which peel requires neutralization?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
2. Which peel contains resorcinol?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
3. Which peel would be the best treatment of severe actinic photodamage?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
4. Which peel would not be indicated for treatment of melasma in a patient with Fitzpatrick skin type IV?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
5. Which peel is a β-hydroxy acid?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
Answers to practice questions provided on next page
Practice Question Answers
1. Which peel requires neutralization?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
2. Which peel contains resorcinol?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
3. Which peel would be the best treatment of severe actinic photodamage?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
4. Which peel would not be indicated for treatment of melasma in a patient with Fitzpatrick skin type IV?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
5. Which peel is a β-hydroxy acid?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
Review the PDF of the fact sheet on Chemical Peels with board-relevant, easy-to-review material. This fact sheet will review the use of chemical peels for dermatologic indications.
Practice Questions
1. Which peel requires neutralization?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
2. Which peel contains resorcinol?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
3. Which peel would be the best treatment of severe actinic photodamage?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
4. Which peel would not be indicated for treatment of melasma in a patient with Fitzpatrick skin type IV?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
5. Which peel is a β-hydroxy acid?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
Answers to practice questions provided on next page
Practice Question Answers
1. Which peel requires neutralization?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
2. Which peel contains resorcinol?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
3. Which peel would be the best treatment of severe actinic photodamage?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
4. Which peel would not be indicated for treatment of melasma in a patient with Fitzpatrick skin type IV?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
5. Which peel is a β-hydroxy acid?
a. Baker-Gordon
b. glycolic acid
c. Jessner
d. salicylic acid
e. trichloroacetic acid
Lip Augmentation With Juvéderm Ultra XC



Postinflammatory erythema
The troubling, frustrating part of acne: the persistent acne scars that are often a prolonged battle for most of our patients. We have many techniques to deal with postinflammatory hyperpigmentation (PIH). However, postinflammatory erythema (PIE), the erythematous scars often seen in acne and other inflammatory skin conditions, is not well understood. And despite its pervasive nature, very little data exist that identify its etiology and effective treatment options.
Inflammatory acne scars are not all the same. PIH, often seen with Fitzpatrick skin types III-VI, is related to brown spots, not red spots. Hyperpigmentation is caused by an excess production of melanin. There are treatments for PIH in our armamentarium – such as microdermabrasion, chemical peels, hydroquinone, and vitamin C – that inhibit melanogenesis and blend the skin discoloration.
In contrast, PIE is characterized by pink, red, and sometimes purple-appearing vascular neogenesis seen most often with skin types I-III after an inflammatory skin condition resolves, and is often seen in cystic acne.
The term postinflammatory erythema was initially introduced in the dermatology literature in 2013 by Bae-Harboe et al. to describe erythema often seen after the resolution of inflammatory acne or other inflammatory skin conditions.1 It is not to be confused with the erythema and telangiectasias seen in erythematotelangiectatic rosacea, which is a separate entity.
In my practice, microneedling has also been effective in reducing PIE. Although this may seem counterintuitive because of the bleeding associated with the microneedling process, microneedling-induced skin tissue injury and neocollagenesis have been clinically shown to improve the abnormal vascular proliferation that occurs in PIE. Similar techniques can be used with fractional resurfacing lasers. However, no studies have specifically evaluated the erythematous component of acne scars treated with fractionated lasers.
Topical preparations containing brimonidine (Mirvaso), azelaic acid, and green tea, as well as oral nicotinamide, can have a temporary effect on reducing skin erythema.
However, very little data or clinical studies are available on treatments for PIE, and there are no well-studied preparations with long-term efficacy data. Studies are needed to provide better clinical guidelines for treatment methods and alternatives to treatments, including topical and systemic medications.
References
1. J Clin Aesthet Dermatol. 2013 Sep;6(9):46-7.
2. J Am Acad Dermatol. 2009 May;60(5):801-7.
Dr. Talakoub and Dr. Wesley are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected].
The troubling, frustrating part of acne: the persistent acne scars that are often a prolonged battle for most of our patients. We have many techniques to deal with postinflammatory hyperpigmentation (PIH). However, postinflammatory erythema (PIE), the erythematous scars often seen in acne and other inflammatory skin conditions, is not well understood. And despite its pervasive nature, very little data exist that identify its etiology and effective treatment options.
Inflammatory acne scars are not all the same. PIH, often seen with Fitzpatrick skin types III-VI, is related to brown spots, not red spots. Hyperpigmentation is caused by an excess production of melanin. There are treatments for PIH in our armamentarium – such as microdermabrasion, chemical peels, hydroquinone, and vitamin C – that inhibit melanogenesis and blend the skin discoloration.
In contrast, PIE is characterized by pink, red, and sometimes purple-appearing vascular neogenesis seen most often with skin types I-III after an inflammatory skin condition resolves, and is often seen in cystic acne.
The term postinflammatory erythema was initially introduced in the dermatology literature in 2013 by Bae-Harboe et al. to describe erythema often seen after the resolution of inflammatory acne or other inflammatory skin conditions.1 It is not to be confused with the erythema and telangiectasias seen in erythematotelangiectatic rosacea, which is a separate entity.
In my practice, microneedling has also been effective in reducing PIE. Although this may seem counterintuitive because of the bleeding associated with the microneedling process, microneedling-induced skin tissue injury and neocollagenesis have been clinically shown to improve the abnormal vascular proliferation that occurs in PIE. Similar techniques can be used with fractional resurfacing lasers. However, no studies have specifically evaluated the erythematous component of acne scars treated with fractionated lasers.
Topical preparations containing brimonidine (Mirvaso), azelaic acid, and green tea, as well as oral nicotinamide, can have a temporary effect on reducing skin erythema.
However, very little data or clinical studies are available on treatments for PIE, and there are no well-studied preparations with long-term efficacy data. Studies are needed to provide better clinical guidelines for treatment methods and alternatives to treatments, including topical and systemic medications.
References
1. J Clin Aesthet Dermatol. 2013 Sep;6(9):46-7.
2. J Am Acad Dermatol. 2009 May;60(5):801-7.
Dr. Talakoub and Dr. Wesley are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected].
The troubling, frustrating part of acne: the persistent acne scars that are often a prolonged battle for most of our patients. We have many techniques to deal with postinflammatory hyperpigmentation (PIH). However, postinflammatory erythema (PIE), the erythematous scars often seen in acne and other inflammatory skin conditions, is not well understood. And despite its pervasive nature, very little data exist that identify its etiology and effective treatment options.
Inflammatory acne scars are not all the same. PIH, often seen with Fitzpatrick skin types III-VI, is related to brown spots, not red spots. Hyperpigmentation is caused by an excess production of melanin. There are treatments for PIH in our armamentarium – such as microdermabrasion, chemical peels, hydroquinone, and vitamin C – that inhibit melanogenesis and blend the skin discoloration.
In contrast, PIE is characterized by pink, red, and sometimes purple-appearing vascular neogenesis seen most often with skin types I-III after an inflammatory skin condition resolves, and is often seen in cystic acne.
The term postinflammatory erythema was initially introduced in the dermatology literature in 2013 by Bae-Harboe et al. to describe erythema often seen after the resolution of inflammatory acne or other inflammatory skin conditions.1 It is not to be confused with the erythema and telangiectasias seen in erythematotelangiectatic rosacea, which is a separate entity.
In my practice, microneedling has also been effective in reducing PIE. Although this may seem counterintuitive because of the bleeding associated with the microneedling process, microneedling-induced skin tissue injury and neocollagenesis have been clinically shown to improve the abnormal vascular proliferation that occurs in PIE. Similar techniques can be used with fractional resurfacing lasers. However, no studies have specifically evaluated the erythematous component of acne scars treated with fractionated lasers.
Topical preparations containing brimonidine (Mirvaso), azelaic acid, and green tea, as well as oral nicotinamide, can have a temporary effect on reducing skin erythema.
However, very little data or clinical studies are available on treatments for PIE, and there are no well-studied preparations with long-term efficacy data. Studies are needed to provide better clinical guidelines for treatment methods and alternatives to treatments, including topical and systemic medications.
References
1. J Clin Aesthet Dermatol. 2013 Sep;6(9):46-7.
2. J Am Acad Dermatol. 2009 May;60(5):801-7.
Dr. Talakoub and Dr. Wesley are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected].
Topical anticholinergic for axillary hyperhidrosis hits marks in phase III
VIENNA – An investigational topical cholinergic receptor antagonist known as DRM04 achieved its efficacy and safety endpoints for the treatment of primary axillary hyperhidrosis in the pivotal phase III ATMOS-1 and ATMOS-2 trials.
“We haven’t had any good new treatment options for patients with hyperhidrosis for a long time,” Dr. David M. Pariser said, in presenting the pivotal trial outcomes data at the annual congress of the European Academy of Dermatology and Venereology.
ATMOS-1 and ATMOS-2 were identically designed 4-week, double-blind studies involving 697 patients with excessive underarm sweating who were randomized 2:1 to once daily use of DRM04 3.75% wipes or a vehicle control. The patients averaged 33 years of age, although as Dr. Pariser noted, primary axillary hyperhidrosis often begins in adolescence and patients as young as age 9 were enrolled. The majority of participants were female. Roughly two-thirds of subjects were grade 3 on the 4-point Hyperhidrosis Disease Severity Scale. The rest were grade 4.
The coprimary endpoints in ATMOS-1 and ATMOS-2 were a 4-point or greater improvement on the Axillary Sweating Daily Diary (ASDD) between baseline and week 4, and the absolute change from baseline in axillary sweat production measured gravimetrically.
The ASDD is a new patient-reported outcome measure developed specifically for the ATMOS trials. At baseline, participants scored a mean of 7.2 points on the 0-10 scale. A 4-point or greater improvement was seen at week 4 in 52.8% of ATMOS-1 participants on DRM04, compared with 28.3% of controls. In ATMOS-2, the spread was 66.1% vs. 26.9%.
Mean baseline sweat production was roughly 175 mg/5 min per armpit, a prodigious rate given that 50 mg/5 min is considered excessive. In ATMOS-1, the rate dropped by an adjusted average of 96.2 mg/5 min per armpit with active therapy, compared with 90.6 mg/5 min in the control group. In ATMOS-2, the DRM04 users had a mean drop in sweat production of 110.3 mg/5 min, compared with a reduction of 92.2 mg/5 min in the control group. Both of these differences were statistically significant and clinically meaningful, Dr. Pariser said.
The secondary endpoint in the studies was change in the Dermatology Life Quality Index from baseline to week 4. The improvement in DRM04 users averaged 8.1 points in ATMOS-1 and 8.6 points in ATMOS-2, both significantly greater than the 4.3- and 5.0-point improvements in the control arms.
In ATMOS-1, 9.2% of patients in the DRM04 arm dropped out of the study, in many cases because of anticholinergic side effects, the most common of which included dry mouth, dry eyes, urinary hesitation or less frequently retention, and constipation. These were mostly mild to moderate in nature and were generally responsive to temporary treatment discontinuation, which the study protocol allowed. The dropout rate in the DRM04 arm of ATMOS-2 was 6.8%.
Seven percent of subjects in the DRM04 study arms experienced mydriasis, most often unilaterally. Dr. Pariser said this might be due to patients touching an eye while they still had DRM04 on their hands, a problem that can be readily addressed in the medication use instructions.
The dropout rates in the control groups were 2.6% and 5%.
More than 80% of ATMOS-1 and ATMOS-2 participants enrolled in ARIDO, the 48-week, open label, phase III extension study of DRM04. Dermira, which is developing DRM04, has announced it plans to file for marketing approval by the Food and Drug Administration in the second half of 2017.
Dr. Pariser reported serving as an investigator for and consultant to Dermira, Brickell Biotech, and TheraVida.
VIENNA – An investigational topical cholinergic receptor antagonist known as DRM04 achieved its efficacy and safety endpoints for the treatment of primary axillary hyperhidrosis in the pivotal phase III ATMOS-1 and ATMOS-2 trials.
“We haven’t had any good new treatment options for patients with hyperhidrosis for a long time,” Dr. David M. Pariser said, in presenting the pivotal trial outcomes data at the annual congress of the European Academy of Dermatology and Venereology.
ATMOS-1 and ATMOS-2 were identically designed 4-week, double-blind studies involving 697 patients with excessive underarm sweating who were randomized 2:1 to once daily use of DRM04 3.75% wipes or a vehicle control. The patients averaged 33 years of age, although as Dr. Pariser noted, primary axillary hyperhidrosis often begins in adolescence and patients as young as age 9 were enrolled. The majority of participants were female. Roughly two-thirds of subjects were grade 3 on the 4-point Hyperhidrosis Disease Severity Scale. The rest were grade 4.
The coprimary endpoints in ATMOS-1 and ATMOS-2 were a 4-point or greater improvement on the Axillary Sweating Daily Diary (ASDD) between baseline and week 4, and the absolute change from baseline in axillary sweat production measured gravimetrically.
The ASDD is a new patient-reported outcome measure developed specifically for the ATMOS trials. At baseline, participants scored a mean of 7.2 points on the 0-10 scale. A 4-point or greater improvement was seen at week 4 in 52.8% of ATMOS-1 participants on DRM04, compared with 28.3% of controls. In ATMOS-2, the spread was 66.1% vs. 26.9%.
Mean baseline sweat production was roughly 175 mg/5 min per armpit, a prodigious rate given that 50 mg/5 min is considered excessive. In ATMOS-1, the rate dropped by an adjusted average of 96.2 mg/5 min per armpit with active therapy, compared with 90.6 mg/5 min in the control group. In ATMOS-2, the DRM04 users had a mean drop in sweat production of 110.3 mg/5 min, compared with a reduction of 92.2 mg/5 min in the control group. Both of these differences were statistically significant and clinically meaningful, Dr. Pariser said.
The secondary endpoint in the studies was change in the Dermatology Life Quality Index from baseline to week 4. The improvement in DRM04 users averaged 8.1 points in ATMOS-1 and 8.6 points in ATMOS-2, both significantly greater than the 4.3- and 5.0-point improvements in the control arms.
In ATMOS-1, 9.2% of patients in the DRM04 arm dropped out of the study, in many cases because of anticholinergic side effects, the most common of which included dry mouth, dry eyes, urinary hesitation or less frequently retention, and constipation. These were mostly mild to moderate in nature and were generally responsive to temporary treatment discontinuation, which the study protocol allowed. The dropout rate in the DRM04 arm of ATMOS-2 was 6.8%.
Seven percent of subjects in the DRM04 study arms experienced mydriasis, most often unilaterally. Dr. Pariser said this might be due to patients touching an eye while they still had DRM04 on their hands, a problem that can be readily addressed in the medication use instructions.
The dropout rates in the control groups were 2.6% and 5%.
More than 80% of ATMOS-1 and ATMOS-2 participants enrolled in ARIDO, the 48-week, open label, phase III extension study of DRM04. Dermira, which is developing DRM04, has announced it plans to file for marketing approval by the Food and Drug Administration in the second half of 2017.
Dr. Pariser reported serving as an investigator for and consultant to Dermira, Brickell Biotech, and TheraVida.
VIENNA – An investigational topical cholinergic receptor antagonist known as DRM04 achieved its efficacy and safety endpoints for the treatment of primary axillary hyperhidrosis in the pivotal phase III ATMOS-1 and ATMOS-2 trials.
“We haven’t had any good new treatment options for patients with hyperhidrosis for a long time,” Dr. David M. Pariser said, in presenting the pivotal trial outcomes data at the annual congress of the European Academy of Dermatology and Venereology.
ATMOS-1 and ATMOS-2 were identically designed 4-week, double-blind studies involving 697 patients with excessive underarm sweating who were randomized 2:1 to once daily use of DRM04 3.75% wipes or a vehicle control. The patients averaged 33 years of age, although as Dr. Pariser noted, primary axillary hyperhidrosis often begins in adolescence and patients as young as age 9 were enrolled. The majority of participants were female. Roughly two-thirds of subjects were grade 3 on the 4-point Hyperhidrosis Disease Severity Scale. The rest were grade 4.
The coprimary endpoints in ATMOS-1 and ATMOS-2 were a 4-point or greater improvement on the Axillary Sweating Daily Diary (ASDD) between baseline and week 4, and the absolute change from baseline in axillary sweat production measured gravimetrically.
The ASDD is a new patient-reported outcome measure developed specifically for the ATMOS trials. At baseline, participants scored a mean of 7.2 points on the 0-10 scale. A 4-point or greater improvement was seen at week 4 in 52.8% of ATMOS-1 participants on DRM04, compared with 28.3% of controls. In ATMOS-2, the spread was 66.1% vs. 26.9%.
Mean baseline sweat production was roughly 175 mg/5 min per armpit, a prodigious rate given that 50 mg/5 min is considered excessive. In ATMOS-1, the rate dropped by an adjusted average of 96.2 mg/5 min per armpit with active therapy, compared with 90.6 mg/5 min in the control group. In ATMOS-2, the DRM04 users had a mean drop in sweat production of 110.3 mg/5 min, compared with a reduction of 92.2 mg/5 min in the control group. Both of these differences were statistically significant and clinically meaningful, Dr. Pariser said.
The secondary endpoint in the studies was change in the Dermatology Life Quality Index from baseline to week 4. The improvement in DRM04 users averaged 8.1 points in ATMOS-1 and 8.6 points in ATMOS-2, both significantly greater than the 4.3- and 5.0-point improvements in the control arms.
In ATMOS-1, 9.2% of patients in the DRM04 arm dropped out of the study, in many cases because of anticholinergic side effects, the most common of which included dry mouth, dry eyes, urinary hesitation or less frequently retention, and constipation. These were mostly mild to moderate in nature and were generally responsive to temporary treatment discontinuation, which the study protocol allowed. The dropout rate in the DRM04 arm of ATMOS-2 was 6.8%.
Seven percent of subjects in the DRM04 study arms experienced mydriasis, most often unilaterally. Dr. Pariser said this might be due to patients touching an eye while they still had DRM04 on their hands, a problem that can be readily addressed in the medication use instructions.
The dropout rates in the control groups were 2.6% and 5%.
More than 80% of ATMOS-1 and ATMOS-2 participants enrolled in ARIDO, the 48-week, open label, phase III extension study of DRM04. Dermira, which is developing DRM04, has announced it plans to file for marketing approval by the Food and Drug Administration in the second half of 2017.
Dr. Pariser reported serving as an investigator for and consultant to Dermira, Brickell Biotech, and TheraVida.
AT THE EADV CONGRESS
Key clinical point: A once-daily topical anticholinergic agent called DRM04 achieved positive outcomes for the treatment of primary axillary hyperhidrosis in two pivotal phase III trials.
Major finding: 53% and 66% of subjects on DRM04 in two large studies achieved clinically meaningful improvement in axillary sweating, compared with 28% and 27%, respectively, of controls.
Data source: Based on findings from ATMOS-1 and ATMOS-2, identically designed, 4-week, double-blind, vehicle-controlled clinical trials including a total of 687 patients with primary axillary hyperhidrosis.
Disclosures: The studies were funded by Dermira. The presenter reported serving as an investigator for and consultant to the company.
Study finds picosecond laser, with diffractive lens array, effective wrinkle treatment
An industry-funded study suggests that picosecond lasers, touted for their effectiveness at tattoo removal, can safely and effectively treat perioral and periocular wrinkles.
Six months after treatment with picosecond 755-nm alexandrite laser with a diffractive lens array, subjects reported high levels of satisfaction and blinded physicians rated the treated wrinkles as improved or much improved over the same time period (Lasers Surg Med. 2016 Sep 29. doi: 10.1002/lsm.22577).
The laser “is very useful for treating fine lines and other visible signs of premature photoaging,” said study coauthor David H. McDaniel, MD, a dermatologist in Virginia Beach, Va., and codirector of the Hampton University Skin of Color Research Institute. “These types of lasers have great potential benefit for patients and minimal risk of any significant adverse events.”
According to Dr. McDaniel, picosecond 755-nm alexandrite lasers are commonly used to treat wrinkles, acne scars, and pigment dyschromia, while picosecond 532- and 1,064-nm lasers are used to remove tattoos and treat some pigment dyschromia.
He and his coinvestigators sought to “better define the parameters that are uniquely contributing to wrinkle reduction and dermal matrix remodeling and also define the actual clinical benefits and treatment protocol,” Dr. McDaniel said in an interview.
At four 1-month intervals, they used a picosecond 755 nm alexandrite laser with diffractive lens array to treat the full faces of 40 women with wrinkles caused by photodamage. The subjects were all healthy white nonsmokers, whose average age was 58 years.
At 6 months following treatment, the average Fitzpatrick wrinkle score improved, dropping from 5.48 to 3.47 (P less than .05). Also at 6 months, blinded physician evaluators rated the average degree of improvement from baseline as “moderate” (for fine lines) “less than mild” (for erythema), “high moderate” (for dyschromia) and “mid moderate” (for global improvement).
The evaluators successfully identified posttreatment photos in 82% of cases. As for patients, 36.8% said they were extremely satisfied and 57.9% said they were satisfied at 6 months. Adverse effects were reported as mild: One patient reported 2 days of erythema, another reported 4 days of edema, and one experienced bruising. Serial punch biopsies obtained at 6 months after the last treatment in six patients revealed increases in dermal collagen and thicker, denser elastin fibers.
The picosecond 755- nm alexandrite laser produces “very little thermal effect, which reduces both treatment discomfort as well as the risk of adverse events,” Dr. McDaniel said in an interview. “It typically leads to shorter recovery time socially, as erythema is very mild and quite transient.”
He noted that the laser can be used in conjunction with other lasers. “Some of the other gold standard fractional lasers are still used in our practice, and they still deliver good results when properly indicated,” he said. “For example, we may use – or even combine with the picosecond laser – a fractional erbium laser to reach deeper into the dermis for severe acne scarring or for deep wrinkles. Or we may use a fractional thulium laser for people with early actinic keratosis or also combine it with the picosecond laser.
Cynosure, a maker of picosecond lasers, funded the study and provided a discounted price for the laser. Dr. McDaniel and another author report serving as consultants, researchers and speakers for Cynosure.
An industry-funded study suggests that picosecond lasers, touted for their effectiveness at tattoo removal, can safely and effectively treat perioral and periocular wrinkles.
Six months after treatment with picosecond 755-nm alexandrite laser with a diffractive lens array, subjects reported high levels of satisfaction and blinded physicians rated the treated wrinkles as improved or much improved over the same time period (Lasers Surg Med. 2016 Sep 29. doi: 10.1002/lsm.22577).
The laser “is very useful for treating fine lines and other visible signs of premature photoaging,” said study coauthor David H. McDaniel, MD, a dermatologist in Virginia Beach, Va., and codirector of the Hampton University Skin of Color Research Institute. “These types of lasers have great potential benefit for patients and minimal risk of any significant adverse events.”
According to Dr. McDaniel, picosecond 755-nm alexandrite lasers are commonly used to treat wrinkles, acne scars, and pigment dyschromia, while picosecond 532- and 1,064-nm lasers are used to remove tattoos and treat some pigment dyschromia.
He and his coinvestigators sought to “better define the parameters that are uniquely contributing to wrinkle reduction and dermal matrix remodeling and also define the actual clinical benefits and treatment protocol,” Dr. McDaniel said in an interview.
At four 1-month intervals, they used a picosecond 755 nm alexandrite laser with diffractive lens array to treat the full faces of 40 women with wrinkles caused by photodamage. The subjects were all healthy white nonsmokers, whose average age was 58 years.
At 6 months following treatment, the average Fitzpatrick wrinkle score improved, dropping from 5.48 to 3.47 (P less than .05). Also at 6 months, blinded physician evaluators rated the average degree of improvement from baseline as “moderate” (for fine lines) “less than mild” (for erythema), “high moderate” (for dyschromia) and “mid moderate” (for global improvement).
The evaluators successfully identified posttreatment photos in 82% of cases. As for patients, 36.8% said they were extremely satisfied and 57.9% said they were satisfied at 6 months. Adverse effects were reported as mild: One patient reported 2 days of erythema, another reported 4 days of edema, and one experienced bruising. Serial punch biopsies obtained at 6 months after the last treatment in six patients revealed increases in dermal collagen and thicker, denser elastin fibers.
The picosecond 755- nm alexandrite laser produces “very little thermal effect, which reduces both treatment discomfort as well as the risk of adverse events,” Dr. McDaniel said in an interview. “It typically leads to shorter recovery time socially, as erythema is very mild and quite transient.”
He noted that the laser can be used in conjunction with other lasers. “Some of the other gold standard fractional lasers are still used in our practice, and they still deliver good results when properly indicated,” he said. “For example, we may use – or even combine with the picosecond laser – a fractional erbium laser to reach deeper into the dermis for severe acne scarring or for deep wrinkles. Or we may use a fractional thulium laser for people with early actinic keratosis or also combine it with the picosecond laser.
Cynosure, a maker of picosecond lasers, funded the study and provided a discounted price for the laser. Dr. McDaniel and another author report serving as consultants, researchers and speakers for Cynosure.
An industry-funded study suggests that picosecond lasers, touted for their effectiveness at tattoo removal, can safely and effectively treat perioral and periocular wrinkles.
Six months after treatment with picosecond 755-nm alexandrite laser with a diffractive lens array, subjects reported high levels of satisfaction and blinded physicians rated the treated wrinkles as improved or much improved over the same time period (Lasers Surg Med. 2016 Sep 29. doi: 10.1002/lsm.22577).
The laser “is very useful for treating fine lines and other visible signs of premature photoaging,” said study coauthor David H. McDaniel, MD, a dermatologist in Virginia Beach, Va., and codirector of the Hampton University Skin of Color Research Institute. “These types of lasers have great potential benefit for patients and minimal risk of any significant adverse events.”
According to Dr. McDaniel, picosecond 755-nm alexandrite lasers are commonly used to treat wrinkles, acne scars, and pigment dyschromia, while picosecond 532- and 1,064-nm lasers are used to remove tattoos and treat some pigment dyschromia.
He and his coinvestigators sought to “better define the parameters that are uniquely contributing to wrinkle reduction and dermal matrix remodeling and also define the actual clinical benefits and treatment protocol,” Dr. McDaniel said in an interview.
At four 1-month intervals, they used a picosecond 755 nm alexandrite laser with diffractive lens array to treat the full faces of 40 women with wrinkles caused by photodamage. The subjects were all healthy white nonsmokers, whose average age was 58 years.
At 6 months following treatment, the average Fitzpatrick wrinkle score improved, dropping from 5.48 to 3.47 (P less than .05). Also at 6 months, blinded physician evaluators rated the average degree of improvement from baseline as “moderate” (for fine lines) “less than mild” (for erythema), “high moderate” (for dyschromia) and “mid moderate” (for global improvement).
The evaluators successfully identified posttreatment photos in 82% of cases. As for patients, 36.8% said they were extremely satisfied and 57.9% said they were satisfied at 6 months. Adverse effects were reported as mild: One patient reported 2 days of erythema, another reported 4 days of edema, and one experienced bruising. Serial punch biopsies obtained at 6 months after the last treatment in six patients revealed increases in dermal collagen and thicker, denser elastin fibers.
The picosecond 755- nm alexandrite laser produces “very little thermal effect, which reduces both treatment discomfort as well as the risk of adverse events,” Dr. McDaniel said in an interview. “It typically leads to shorter recovery time socially, as erythema is very mild and quite transient.”
He noted that the laser can be used in conjunction with other lasers. “Some of the other gold standard fractional lasers are still used in our practice, and they still deliver good results when properly indicated,” he said. “For example, we may use – or even combine with the picosecond laser – a fractional erbium laser to reach deeper into the dermis for severe acne scarring or for deep wrinkles. Or we may use a fractional thulium laser for people with early actinic keratosis or also combine it with the picosecond laser.
Cynosure, a maker of picosecond lasers, funded the study and provided a discounted price for the laser. Dr. McDaniel and another author report serving as consultants, researchers and speakers for Cynosure.
FROM LASERS IN SURGERY AND MEDICINE
Key clinical point: Wrinkle treatment via picosecond 755-nm alexandrite laser with a diffractive lens array appears to be safe and effective.
Major finding: Six months after the last treatment, 36.8% of patients were extremely satisfied and 57.9% were satisfied with the results, with minor, transient adverse effects. Blinded physician evaluators reported “mid moderate” global improvement.
Data source: A prospective, blinded study of 40 healthy white women, nonsmokers, average age 58 (range: 47-64), who underwent four full-face treatments via laser at 1-month intervals.
Disclosures: Cynosure, a maker of picosecond lasers, funded the study and provided a discounted price for the laser. Dr. McDaniel and another author report serving as consultants, researchers, and speakers for Cynosure.
Update on argan oil
• Used traditionally in Northwest Africa for its cosmetic, bactericidal, and fungicidal activity.
• Rich in vitamin E, oleic acid, and linoleic acid, which are believed to contribute to the perceived cutaneous benefits of this vegetable oil.
• Reputed to impart antiacne, antisebum, antiaging, moisturizing, and wound-healing activity, but clinical evidence is sparse.
• In a small study, the nightly topical application of argan oil resulted in a moisturizing effect, and in statistically significant decreases in transepidermal water loss and increases in the water content of the epidermis.
For more than 800 years, native Moroccans and explorers in the region have cited the health benefits of the topical use or consumption of argan oil.1 The oil, derived from the fruit of Argania spinosa, is a slow-growing tree native to the arid climate of Southwestern Morocco2-4 as well as the Algerian province of Tindouf in the Western Mediterranean area.5 For many years, it was primarily the populations of the Essaouira and Souss-Massa-Draa regions of Morocco that benefited from the production and use of argan oil.6 Largely through the efforts of the Moroccan government, as well as cooperating nongovernmental organizations and private entities, argan oil is now also a well-established ingredient on the edible oil as well as cosmetic oil markets throughout the world.6
The vitamin E–rich oil has a reputation for imparting antiaging, hydrating, and antioxidant activity to the skin and ameliorating conditions such as acne, eczema, psoriasis, wrinkles, and xerosis,12 and, in fact, has been used to treat these conditions as well as dry hair,3,13 hair loss, skin inflammation, and joint pain.3 This column will focus on the topical uses of this botanical that has been dubbed “liquid gold.”12
Chemistry
Oleic acid, an omega-9 monounsaturated fatty acid, is abundant in argan oil (43%-49%) and has been found to act as a penetration enhancer by disturbing the skin barrier.14,15 Linoleic acid, an omega-6 polyunsaturated fatty acid, found in concentrations of 29%-36% in the oil, is integral in the biosynthesis of inflammatory prostaglandins through the arachidonic acid pathway.4,16 The presence of linoleic acid may help prevent or mitigate inflammation. Linoleic acid is also a component of ceramide 1 linoleate, which is diminished in dry skin. Topical application of linoleic acid can raise ceramide 1 linoleate levels in the skin, thus reducing xerosis.17 Argan oil also contains the saturated fatty acids palmitic acid (11%-15%) and stearic acid (4%-7%).2
Though argan oil is mainly composed of unsaturated fatty acids (80%),1,18,19 the unsaponifiable fraction (1%) is replete with antioxidants, including sterols, saponins, and polyphenols.4,19 The polyphenolic constituents, primarily gamma-tocopherol, which is considered the most efficient among the tocopherols at scavenging free radicals, are thought to account for the antioxidant effects of argan oil.1,2,18,20,21
Topical uses
Unroasted kernels are used to produce cosmetic-grade argan oil, which is used in moisturizing creams, body lotions, and shampoos.2 Although argan oil contains components that have antioxidant and anti-inflammatory features and there are many patents on the use of argan oil in skin care, there is a dearth of published research studies looking at the effect of argan oil–containing skin care products on aging, inflamed, or dry skin. A study by Dobrev evaluated the efficacy of a sebum control cream composed of saw palmetto extract, sesame seeds, and argan oil applied twice daily to the face over a period of 4 weeks in 20 healthy volunteers, 16 with oily skin and 4 with combination skin. All volunteers tolerated the product. A visible sebum-regulating or antisebum efficacy was observed in 95% of the subjects. Clinical evaluation scores and casual sebum levels decreased significantly after 1 month of treatment. Dobrev concluded that this argan oil-containing formulation was efficacious in lessening the greasiness and improving the appearance of oily facial skin.22
Early in 2015, Boucetta et al. reported on their study of the effects on skin elasticity of the daily application or consumption of argan oil in 60 postmenopausal women. During a 60-day period, the treatment group of 30 subjects consumed dietary argan oil; the 30 members in the control group received olive oil. Both groups also applied topical argan oil to the left volar forearm. Skin parameters, including gross skin elasticity, net elasticity, and biologic elasticity, improved significantly with both oral and topical treatments. The researchers concluded that argan oil use confers an antiaging effect to the skin through enhanced elasticity.24 Boucetta and another team previously showed that daily consumption or topical application of argan oil in postmenopausal women yielded significant reductions in transepidermal water loss and significant increases in epidermal water content, suggesting that the botanical agent ameliorates skin hydration by reviving barrier function and preserving the water-holding capacity.25 The same team also demonstrated in 30 healthy postmenopausal women that the nightly topical application of argan oil over a 2-month period yielded a moisturizing effect, with statistically significant reductions in transepidermal water loss and statistically significant increases in the water content of the epidermis observed.26
As a cosmetic agent, argan oil, which is popular in France, Japan, and North America, is touted for hydrating and revitalizing the skin, treating acne, and imparting shine to the hair. The therapeutic activities of topical argan oil are reputed to be antiacne, antisebum, antiaging, moisturizing, and wound healing, but such claims are based on traditional uses with only a small body of supportive clinical evidence.2,27
Generally, argan oil prices are as high as $40/100 mL in the European, Japanese, and American markets.27 Topical argan oil has been characterized as having a brief shelf-life of approximately 3-4 months.2,28 A 2014 report on a 1-year study of the oxidative stability of cosmetic argan oil by Gharby et al. found that argan oil quality remains satisfactory when stored at 25° C and protected from sunlight, but storage should not exceed 6 months to meet industrial standards. A rapid loss of quality was seen when argan oil was stored at 40° C.29
Conclusion
Although clinical research data on argan oil are limited, its traditional uses and inclusion in novel cosmetic products suggest that further study is warranted. Randomized controlled trials are needed to elucidate cutaneous benefits, if any, from this rare botanical.
1. J Pharm Pharmacol. 2010 Dec;62(12):1669-75.
2. Altern Med Rev. 2011 Sep;16(3):275-9.
3. J Ethnopharmacol. 1999 Oct;67(1):7-14.
4. Pharmacol Res. 2006 Jul;54(1):1-5.
5. Nutr Rev. 2012 May;70(5):266-79.
6. Eur J Lipid Sci Technol. 2014 Oct;116(10):1316-21.
7. Ann Nutr Metab. 2005 May-Jun;49(3):196-201.
8. Nutr Metab Cardiovasc Dis. 2005 Oct;15(5):352-60.
9. Evid Based Complement Alternat Med. 2006 Sep;3(3):317-27.
10. Cancer Invest. 2006 Oct;24(6):588-92.
11. Cancer Detect Prev. 2007;31(1):64-9.
12. “Liquid Gold in Morocco,” by Amy Larocca, The New York Times, Nov. 18, 2007.
13. Phytomedicine. 2010 Feb;17(2):157-60.
14. J Control Release. 1995;37(3):299-306.
15. Thermochimica Acta. 1997 Jun;293:77-85.
16. Prostaglandins Leukot Essent Fatty Acids. 1995 Jun;52(6):387-91.
17. Int J Cosmet Sci. 1996 Feb;18(1):1-12.
18. Crit Rev Food Sci Nutr. 2010 May;50(5):473-7.
19. Eur J Cancer Prev. 2003 Feb;12(1):67-75.
20. Fitoterapia. 2008 Jul;79(5):337-44.
21. Am J Clin Nutr. 2001 Dec;74(6):714-22.
22. J Cosmet Dermatol. 2007;6(2):113-8.
23. Int J Nanomedicine. 2014 Aug 11;9:3855-64.
24. Clin Interv Aging. 2015 Jan 30;10:339-49.
25. Prz Menopauzalny. 2014 Oct;13(5):280-8.
26. Skin Res Technol. 2013;19:356-7.
27. Inflamm Allergy Drug Targets. 2014;13(3):168-76.
28. Nat Prod Commun. 2010 Nov;5(11):1799-802.
29. J Cosmet Sci. 2014 Mar-Apr;65(2):81-7.
Dr. Baumann is the CEO of Baumann Cosmetic & Research Institute in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. She is the author of “Cosmetic Dermatology: Principles and Practice” (New York: McGraw Hill, 2002), and a book for consumers, “The Skin Type Solution,” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients” (McGraw Hill) was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also develops and owns the Baumann Skin Type Solution skin typing solutions and related products.
• Used traditionally in Northwest Africa for its cosmetic, bactericidal, and fungicidal activity.
• Rich in vitamin E, oleic acid, and linoleic acid, which are believed to contribute to the perceived cutaneous benefits of this vegetable oil.
• Reputed to impart antiacne, antisebum, antiaging, moisturizing, and wound-healing activity, but clinical evidence is sparse.
• In a small study, the nightly topical application of argan oil resulted in a moisturizing effect, and in statistically significant decreases in transepidermal water loss and increases in the water content of the epidermis.
For more than 800 years, native Moroccans and explorers in the region have cited the health benefits of the topical use or consumption of argan oil.1 The oil, derived from the fruit of Argania spinosa, is a slow-growing tree native to the arid climate of Southwestern Morocco2-4 as well as the Algerian province of Tindouf in the Western Mediterranean area.5 For many years, it was primarily the populations of the Essaouira and Souss-Massa-Draa regions of Morocco that benefited from the production and use of argan oil.6 Largely through the efforts of the Moroccan government, as well as cooperating nongovernmental organizations and private entities, argan oil is now also a well-established ingredient on the edible oil as well as cosmetic oil markets throughout the world.6
The vitamin E–rich oil has a reputation for imparting antiaging, hydrating, and antioxidant activity to the skin and ameliorating conditions such as acne, eczema, psoriasis, wrinkles, and xerosis,12 and, in fact, has been used to treat these conditions as well as dry hair,3,13 hair loss, skin inflammation, and joint pain.3 This column will focus on the topical uses of this botanical that has been dubbed “liquid gold.”12
Chemistry
Oleic acid, an omega-9 monounsaturated fatty acid, is abundant in argan oil (43%-49%) and has been found to act as a penetration enhancer by disturbing the skin barrier.14,15 Linoleic acid, an omega-6 polyunsaturated fatty acid, found in concentrations of 29%-36% in the oil, is integral in the biosynthesis of inflammatory prostaglandins through the arachidonic acid pathway.4,16 The presence of linoleic acid may help prevent or mitigate inflammation. Linoleic acid is also a component of ceramide 1 linoleate, which is diminished in dry skin. Topical application of linoleic acid can raise ceramide 1 linoleate levels in the skin, thus reducing xerosis.17 Argan oil also contains the saturated fatty acids palmitic acid (11%-15%) and stearic acid (4%-7%).2
Though argan oil is mainly composed of unsaturated fatty acids (80%),1,18,19 the unsaponifiable fraction (1%) is replete with antioxidants, including sterols, saponins, and polyphenols.4,19 The polyphenolic constituents, primarily gamma-tocopherol, which is considered the most efficient among the tocopherols at scavenging free radicals, are thought to account for the antioxidant effects of argan oil.1,2,18,20,21
Topical uses
Unroasted kernels are used to produce cosmetic-grade argan oil, which is used in moisturizing creams, body lotions, and shampoos.2 Although argan oil contains components that have antioxidant and anti-inflammatory features and there are many patents on the use of argan oil in skin care, there is a dearth of published research studies looking at the effect of argan oil–containing skin care products on aging, inflamed, or dry skin. A study by Dobrev evaluated the efficacy of a sebum control cream composed of saw palmetto extract, sesame seeds, and argan oil applied twice daily to the face over a period of 4 weeks in 20 healthy volunteers, 16 with oily skin and 4 with combination skin. All volunteers tolerated the product. A visible sebum-regulating or antisebum efficacy was observed in 95% of the subjects. Clinical evaluation scores and casual sebum levels decreased significantly after 1 month of treatment. Dobrev concluded that this argan oil-containing formulation was efficacious in lessening the greasiness and improving the appearance of oily facial skin.22
Early in 2015, Boucetta et al. reported on their study of the effects on skin elasticity of the daily application or consumption of argan oil in 60 postmenopausal women. During a 60-day period, the treatment group of 30 subjects consumed dietary argan oil; the 30 members in the control group received olive oil. Both groups also applied topical argan oil to the left volar forearm. Skin parameters, including gross skin elasticity, net elasticity, and biologic elasticity, improved significantly with both oral and topical treatments. The researchers concluded that argan oil use confers an antiaging effect to the skin through enhanced elasticity.24 Boucetta and another team previously showed that daily consumption or topical application of argan oil in postmenopausal women yielded significant reductions in transepidermal water loss and significant increases in epidermal water content, suggesting that the botanical agent ameliorates skin hydration by reviving barrier function and preserving the water-holding capacity.25 The same team also demonstrated in 30 healthy postmenopausal women that the nightly topical application of argan oil over a 2-month period yielded a moisturizing effect, with statistically significant reductions in transepidermal water loss and statistically significant increases in the water content of the epidermis observed.26
As a cosmetic agent, argan oil, which is popular in France, Japan, and North America, is touted for hydrating and revitalizing the skin, treating acne, and imparting shine to the hair. The therapeutic activities of topical argan oil are reputed to be antiacne, antisebum, antiaging, moisturizing, and wound healing, but such claims are based on traditional uses with only a small body of supportive clinical evidence.2,27
Generally, argan oil prices are as high as $40/100 mL in the European, Japanese, and American markets.27 Topical argan oil has been characterized as having a brief shelf-life of approximately 3-4 months.2,28 A 2014 report on a 1-year study of the oxidative stability of cosmetic argan oil by Gharby et al. found that argan oil quality remains satisfactory when stored at 25° C and protected from sunlight, but storage should not exceed 6 months to meet industrial standards. A rapid loss of quality was seen when argan oil was stored at 40° C.29
Conclusion
Although clinical research data on argan oil are limited, its traditional uses and inclusion in novel cosmetic products suggest that further study is warranted. Randomized controlled trials are needed to elucidate cutaneous benefits, if any, from this rare botanical.
1. J Pharm Pharmacol. 2010 Dec;62(12):1669-75.
2. Altern Med Rev. 2011 Sep;16(3):275-9.
3. J Ethnopharmacol. 1999 Oct;67(1):7-14.
4. Pharmacol Res. 2006 Jul;54(1):1-5.
5. Nutr Rev. 2012 May;70(5):266-79.
6. Eur J Lipid Sci Technol. 2014 Oct;116(10):1316-21.
7. Ann Nutr Metab. 2005 May-Jun;49(3):196-201.
8. Nutr Metab Cardiovasc Dis. 2005 Oct;15(5):352-60.
9. Evid Based Complement Alternat Med. 2006 Sep;3(3):317-27.
10. Cancer Invest. 2006 Oct;24(6):588-92.
11. Cancer Detect Prev. 2007;31(1):64-9.
12. “Liquid Gold in Morocco,” by Amy Larocca, The New York Times, Nov. 18, 2007.
13. Phytomedicine. 2010 Feb;17(2):157-60.
14. J Control Release. 1995;37(3):299-306.
15. Thermochimica Acta. 1997 Jun;293:77-85.
16. Prostaglandins Leukot Essent Fatty Acids. 1995 Jun;52(6):387-91.
17. Int J Cosmet Sci. 1996 Feb;18(1):1-12.
18. Crit Rev Food Sci Nutr. 2010 May;50(5):473-7.
19. Eur J Cancer Prev. 2003 Feb;12(1):67-75.
20. Fitoterapia. 2008 Jul;79(5):337-44.
21. Am J Clin Nutr. 2001 Dec;74(6):714-22.
22. J Cosmet Dermatol. 2007;6(2):113-8.
23. Int J Nanomedicine. 2014 Aug 11;9:3855-64.
24. Clin Interv Aging. 2015 Jan 30;10:339-49.
25. Prz Menopauzalny. 2014 Oct;13(5):280-8.
26. Skin Res Technol. 2013;19:356-7.
27. Inflamm Allergy Drug Targets. 2014;13(3):168-76.
28. Nat Prod Commun. 2010 Nov;5(11):1799-802.
29. J Cosmet Sci. 2014 Mar-Apr;65(2):81-7.
Dr. Baumann is the CEO of Baumann Cosmetic & Research Institute in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. She is the author of “Cosmetic Dermatology: Principles and Practice” (New York: McGraw Hill, 2002), and a book for consumers, “The Skin Type Solution,” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients” (McGraw Hill) was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also develops and owns the Baumann Skin Type Solution skin typing solutions and related products.
• Used traditionally in Northwest Africa for its cosmetic, bactericidal, and fungicidal activity.
• Rich in vitamin E, oleic acid, and linoleic acid, which are believed to contribute to the perceived cutaneous benefits of this vegetable oil.
• Reputed to impart antiacne, antisebum, antiaging, moisturizing, and wound-healing activity, but clinical evidence is sparse.
• In a small study, the nightly topical application of argan oil resulted in a moisturizing effect, and in statistically significant decreases in transepidermal water loss and increases in the water content of the epidermis.
For more than 800 years, native Moroccans and explorers in the region have cited the health benefits of the topical use or consumption of argan oil.1 The oil, derived from the fruit of Argania spinosa, is a slow-growing tree native to the arid climate of Southwestern Morocco2-4 as well as the Algerian province of Tindouf in the Western Mediterranean area.5 For many years, it was primarily the populations of the Essaouira and Souss-Massa-Draa regions of Morocco that benefited from the production and use of argan oil.6 Largely through the efforts of the Moroccan government, as well as cooperating nongovernmental organizations and private entities, argan oil is now also a well-established ingredient on the edible oil as well as cosmetic oil markets throughout the world.6
The vitamin E–rich oil has a reputation for imparting antiaging, hydrating, and antioxidant activity to the skin and ameliorating conditions such as acne, eczema, psoriasis, wrinkles, and xerosis,12 and, in fact, has been used to treat these conditions as well as dry hair,3,13 hair loss, skin inflammation, and joint pain.3 This column will focus on the topical uses of this botanical that has been dubbed “liquid gold.”12
Chemistry
Oleic acid, an omega-9 monounsaturated fatty acid, is abundant in argan oil (43%-49%) and has been found to act as a penetration enhancer by disturbing the skin barrier.14,15 Linoleic acid, an omega-6 polyunsaturated fatty acid, found in concentrations of 29%-36% in the oil, is integral in the biosynthesis of inflammatory prostaglandins through the arachidonic acid pathway.4,16 The presence of linoleic acid may help prevent or mitigate inflammation. Linoleic acid is also a component of ceramide 1 linoleate, which is diminished in dry skin. Topical application of linoleic acid can raise ceramide 1 linoleate levels in the skin, thus reducing xerosis.17 Argan oil also contains the saturated fatty acids palmitic acid (11%-15%) and stearic acid (4%-7%).2
Though argan oil is mainly composed of unsaturated fatty acids (80%),1,18,19 the unsaponifiable fraction (1%) is replete with antioxidants, including sterols, saponins, and polyphenols.4,19 The polyphenolic constituents, primarily gamma-tocopherol, which is considered the most efficient among the tocopherols at scavenging free radicals, are thought to account for the antioxidant effects of argan oil.1,2,18,20,21
Topical uses
Unroasted kernels are used to produce cosmetic-grade argan oil, which is used in moisturizing creams, body lotions, and shampoos.2 Although argan oil contains components that have antioxidant and anti-inflammatory features and there are many patents on the use of argan oil in skin care, there is a dearth of published research studies looking at the effect of argan oil–containing skin care products on aging, inflamed, or dry skin. A study by Dobrev evaluated the efficacy of a sebum control cream composed of saw palmetto extract, sesame seeds, and argan oil applied twice daily to the face over a period of 4 weeks in 20 healthy volunteers, 16 with oily skin and 4 with combination skin. All volunteers tolerated the product. A visible sebum-regulating or antisebum efficacy was observed in 95% of the subjects. Clinical evaluation scores and casual sebum levels decreased significantly after 1 month of treatment. Dobrev concluded that this argan oil-containing formulation was efficacious in lessening the greasiness and improving the appearance of oily facial skin.22
Early in 2015, Boucetta et al. reported on their study of the effects on skin elasticity of the daily application or consumption of argan oil in 60 postmenopausal women. During a 60-day period, the treatment group of 30 subjects consumed dietary argan oil; the 30 members in the control group received olive oil. Both groups also applied topical argan oil to the left volar forearm. Skin parameters, including gross skin elasticity, net elasticity, and biologic elasticity, improved significantly with both oral and topical treatments. The researchers concluded that argan oil use confers an antiaging effect to the skin through enhanced elasticity.24 Boucetta and another team previously showed that daily consumption or topical application of argan oil in postmenopausal women yielded significant reductions in transepidermal water loss and significant increases in epidermal water content, suggesting that the botanical agent ameliorates skin hydration by reviving barrier function and preserving the water-holding capacity.25 The same team also demonstrated in 30 healthy postmenopausal women that the nightly topical application of argan oil over a 2-month period yielded a moisturizing effect, with statistically significant reductions in transepidermal water loss and statistically significant increases in the water content of the epidermis observed.26
As a cosmetic agent, argan oil, which is popular in France, Japan, and North America, is touted for hydrating and revitalizing the skin, treating acne, and imparting shine to the hair. The therapeutic activities of topical argan oil are reputed to be antiacne, antisebum, antiaging, moisturizing, and wound healing, but such claims are based on traditional uses with only a small body of supportive clinical evidence.2,27
Generally, argan oil prices are as high as $40/100 mL in the European, Japanese, and American markets.27 Topical argan oil has been characterized as having a brief shelf-life of approximately 3-4 months.2,28 A 2014 report on a 1-year study of the oxidative stability of cosmetic argan oil by Gharby et al. found that argan oil quality remains satisfactory when stored at 25° C and protected from sunlight, but storage should not exceed 6 months to meet industrial standards. A rapid loss of quality was seen when argan oil was stored at 40° C.29
Conclusion
Although clinical research data on argan oil are limited, its traditional uses and inclusion in novel cosmetic products suggest that further study is warranted. Randomized controlled trials are needed to elucidate cutaneous benefits, if any, from this rare botanical.
1. J Pharm Pharmacol. 2010 Dec;62(12):1669-75.
2. Altern Med Rev. 2011 Sep;16(3):275-9.
3. J Ethnopharmacol. 1999 Oct;67(1):7-14.
4. Pharmacol Res. 2006 Jul;54(1):1-5.
5. Nutr Rev. 2012 May;70(5):266-79.
6. Eur J Lipid Sci Technol. 2014 Oct;116(10):1316-21.
7. Ann Nutr Metab. 2005 May-Jun;49(3):196-201.
8. Nutr Metab Cardiovasc Dis. 2005 Oct;15(5):352-60.
9. Evid Based Complement Alternat Med. 2006 Sep;3(3):317-27.
10. Cancer Invest. 2006 Oct;24(6):588-92.
11. Cancer Detect Prev. 2007;31(1):64-9.
12. “Liquid Gold in Morocco,” by Amy Larocca, The New York Times, Nov. 18, 2007.
13. Phytomedicine. 2010 Feb;17(2):157-60.
14. J Control Release. 1995;37(3):299-306.
15. Thermochimica Acta. 1997 Jun;293:77-85.
16. Prostaglandins Leukot Essent Fatty Acids. 1995 Jun;52(6):387-91.
17. Int J Cosmet Sci. 1996 Feb;18(1):1-12.
18. Crit Rev Food Sci Nutr. 2010 May;50(5):473-7.
19. Eur J Cancer Prev. 2003 Feb;12(1):67-75.
20. Fitoterapia. 2008 Jul;79(5):337-44.
21. Am J Clin Nutr. 2001 Dec;74(6):714-22.
22. J Cosmet Dermatol. 2007;6(2):113-8.
23. Int J Nanomedicine. 2014 Aug 11;9:3855-64.
24. Clin Interv Aging. 2015 Jan 30;10:339-49.
25. Prz Menopauzalny. 2014 Oct;13(5):280-8.
26. Skin Res Technol. 2013;19:356-7.
27. Inflamm Allergy Drug Targets. 2014;13(3):168-76.
28. Nat Prod Commun. 2010 Nov;5(11):1799-802.
29. J Cosmet Sci. 2014 Mar-Apr;65(2):81-7.
Dr. Baumann is the CEO of Baumann Cosmetic & Research Institute in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. She is the author of “Cosmetic Dermatology: Principles and Practice” (New York: McGraw Hill, 2002), and a book for consumers, “The Skin Type Solution,” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients” (McGraw Hill) was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also develops and owns the Baumann Skin Type Solution skin typing solutions and related products.
Cosmetic Corner: Dermatologists Weigh in on Acne Scar Treatments
To improve patient care and outcomes, leading dermatologists offered their recommendations on acne scar treatments. Consideration must be given to:
- Effaclar BB Blur
La Roche-Posay Laboratoire Dermatologique
“Unfortunately, acne scars are permanent, but by applying cosmetics over the skin, you can minimize their appearance. Combining ingredients that give cosmetic coverage, minimize pores, and absorb oil, this product gives the skin a brighter, more even complexion.”
— Joshua Zeichner, MD, New York, New York
- Fraxel
Valeant Pharmaceuticals International, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Glytone Rejuvenating Mini Peel Gel
Pierre Fabre Dermo Cosmetique USA
“This gel product contains a low concentration of glycolic acid. It can be applied at home, left on the skin for 10 minutes, and neutralized with water.”
—Cherise M. Levi, DO, New York, New York
- SkinMedica Retinol Complex 0.25
Allergan
“Retinol will help to minimize acne scarring.”
— Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices and self-tanners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on acne scar treatments. Consideration must be given to:
- Effaclar BB Blur
La Roche-Posay Laboratoire Dermatologique
“Unfortunately, acne scars are permanent, but by applying cosmetics over the skin, you can minimize their appearance. Combining ingredients that give cosmetic coverage, minimize pores, and absorb oil, this product gives the skin a brighter, more even complexion.”
— Joshua Zeichner, MD, New York, New York
- Fraxel
Valeant Pharmaceuticals International, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Glytone Rejuvenating Mini Peel Gel
Pierre Fabre Dermo Cosmetique USA
“This gel product contains a low concentration of glycolic acid. It can be applied at home, left on the skin for 10 minutes, and neutralized with water.”
—Cherise M. Levi, DO, New York, New York
- SkinMedica Retinol Complex 0.25
Allergan
“Retinol will help to minimize acne scarring.”
— Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices and self-tanners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on acne scar treatments. Consideration must be given to:
- Effaclar BB Blur
La Roche-Posay Laboratoire Dermatologique
“Unfortunately, acne scars are permanent, but by applying cosmetics over the skin, you can minimize their appearance. Combining ingredients that give cosmetic coverage, minimize pores, and absorb oil, this product gives the skin a brighter, more even complexion.”
— Joshua Zeichner, MD, New York, New York
- Fraxel
Valeant Pharmaceuticals International, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Glytone Rejuvenating Mini Peel Gel
Pierre Fabre Dermo Cosmetique USA
“This gel product contains a low concentration of glycolic acid. It can be applied at home, left on the skin for 10 minutes, and neutralized with water.”
—Cherise M. Levi, DO, New York, New York
- SkinMedica Retinol Complex 0.25
Allergan
“Retinol will help to minimize acne scarring.”
— Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices and self-tanners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.