User login
A Noninvasive Mechanical Treatment to Reduce the Visible Appearance of Cellulite
Cellulite is a cosmetic problem, not a disease process. It affects 85% to 90% of all women worldwide and was described nearly 100 years ago.1 Causes may be genetic, hormonal, or vascular in nature and may be related to the septa configuration in the subdermal tissue. Fibrosis at the dermal-subcutaneous junction as well as decreased vascular and lymphatic circulation also may be causative factors.
Cellulite has a multifactorial etiology. Khan et al2 noted that there are specific classic patterns of cellulite that affect women exclusively. White women tend to have somewhat higher rates of cellulite than Asian women. The authors also stated that lifestyle factors such as high carbohydrate diets may lead to an increase in total body fat content, which enhances the appearance of cellulite.2
The subdermal anatomy affects the appearance of cellulite. Utilizing in vivo magnetic resonance imaging, Querleux et al3 showed that women with visible cellulite have dermal septa that are thinner and generally more perpendicular to the skin’s surface than women without cellulite. In women without cellulite, the orientation of the septa is more angled into a crisscross pattern. In women with a high percentage of perpendicular septa, the perpendicular septa allow for fat herniation with dimpling of the skin compared to the crisscross septa pattern.2 Other investigators have discussed the reduction of blood flow in specific areas of the body in women, particularly in cellulite-prone areas such as the buttocks and thighs, as another causative factor.2,4,5 Rossi and Vergnanini6 showed that the blood flow was 35% lower in affected cellulite regions than in nonaffected regions without cellulite, which can cause congestion of blood and lymphatic flow and increased subdermal pressure, thus increasing the appearance of cellulite.
Although there is some controversy regarding the effects of weight loss on the appearance of cellulite,2,7 it appears that the subdermal septa and morphology have more of an effect on the appearance of cellulite.2,3,8
Rossi and Vergnanini6 proposed a 4-grade system for evaluating the appearance of cellulite (grade I, no cellulite; grade II, skin that is smooth and without any pronounced dimpling upon standing or lying down but may show some dimpling upon pinching and strong muscle contraction; grade III, cellulite is present in upright positions but not when the patient is in a supine position; grade IV, cellulite can be seen when the patient is standing and in a supine position). Both grades III and IV can be exacerbated by maximal voluntary contraction and strong pinching of the skin because these actions cause the subcutaneous fat to move toward the surface of the skin between the septa. This grading system aligns with categories I through III described by Mirrashed et al.9
There are many cellulite treatments available but few actually create a reduction in the visible appearance of cellulite. A number of these treatments were reviewed by Khan et al,10 including massage; a noninvasive suction-assisted massage technique; and topical agents such as xanthine, retinols, and other botanicals.4,11-14 Liposuction has not been shown to be effective in the treatment of cellulite and in fact may increase the appearance of cellulite.9,15 Mesotherapy, a modality that entails injecting substances into the subcutaneous fat layer, is another treatment of cellulite. Two of the most common agents purported to dissolve fat include phosphatidylcholine and sodium deoxycholate. The efficacy and safety of mesotherapy remains controversial and unproven. A July 2008 position statement from the American Society of Plastic Surgeons stated that “low levels of validity and quality of the literature does not allow [American Society of Plastic Surgeons] to support a recommendation for the use of mesotherapy/injection lipolysis for fat reduction.”16 Other modalities such as noninvasive dual-wavelength laser/suction devices; low-energy diode laser, contact cooling, suction, and massage devices; and infrared, bipolar radiofrequency, and suction with mechanical massage devices are available and show some small improvements in the visible appearance of cellulite, but no rating scales were used in any of these studies.17,18 DiBernardo19 utilized a 1440-nm pulsed laser to treat cellulite. It is an invasive treatment that works by breaking down some of the connective tissue septa responsible for the majority and greater severity of the dermal dimpling seen in cellulite, increasing the thickness of the dermis as well as its elasticity, reducing subcutaneous fat, and improving circulation and reducing general lymphatic congestion.19 The system showed promise but was an invasive treatment, and one session could cost $5000 to $7000 for bilateral areas and another $2500 for each additional area.20 Burns21 expressed that the short-term results showed promise in reducing the appearance of cellulite. Noninvasive ultrasound22,23 as well as extracorporeal shock wave therapy24,25 also has shown some improvement in the firmness of collagen but generally not in the appearance of cellulite.
We sought to evaluate the efficacy and safety of a noninvasive mechanical treatment of cellulite.
Methods
This study was conducted in accordance with the guidelines set forth by the US Department of Health and Human Services’ Policy for Protection of Human Research Subjects and the World Medical Association’s Declaration of Helsinki. Participants were recruited through local area medical facilities in southeastern Michigan. Written informed consent was obtained from all participants prior to beginning the study.
Patients with grades II to IV cellulite, according to the Rossi and Vergnanini6 grading system, were allowed to participate. All participants in the study were asked not to make lifestyle changes (eg, exercise habits, diet) or use any other treatments for cellulite that might be available to them during the study period. Exclusion criteria included history of deep vein thrombosis, cancer diagnosed within the last year, pregnancy, hemophilia, severe lymphedema, presence of a pacemaker, epilepsy, seizure disorder, or current use of anticoagulants. History of partial or total joint replacements, acute hernia, nonunited fractures, advanced arthritis, or detached retina also excluded participation in the study.
Participants completed an 8-week, twice-weekly treatment protocol with a noninvasive mechanical device performed in clinic. The device consisted of a 10.16-cm belt with a layer of nonslip material wrapped around the belt. The belt was attached to a mechanical oscillator. We adjusted the stroke length to approximately 2 cm and moved the dermis at that length at approximately 1000 strokes per minute.
Each participant was treated for a total treatment time of 18 to 24 minutes. The total treatment area included the top of the iliac crest to just above the top of the popliteal space. The width of the belt (10.16 cm) was equal to 1 individual treatment area. Each individual treatment area was treated for 2 minutes. First the buttocks and bilateral thighs were treated, followed by the right lateral thigh and the left lateral thigh. The belt was moved progressively down the total treatment area until all individual treatment areas were addressed. The average participant had 3 to 4 bilateral thigh and buttocks treatment areas and 3 to 4 lateral treatment areas on both the left and right sides of the body.
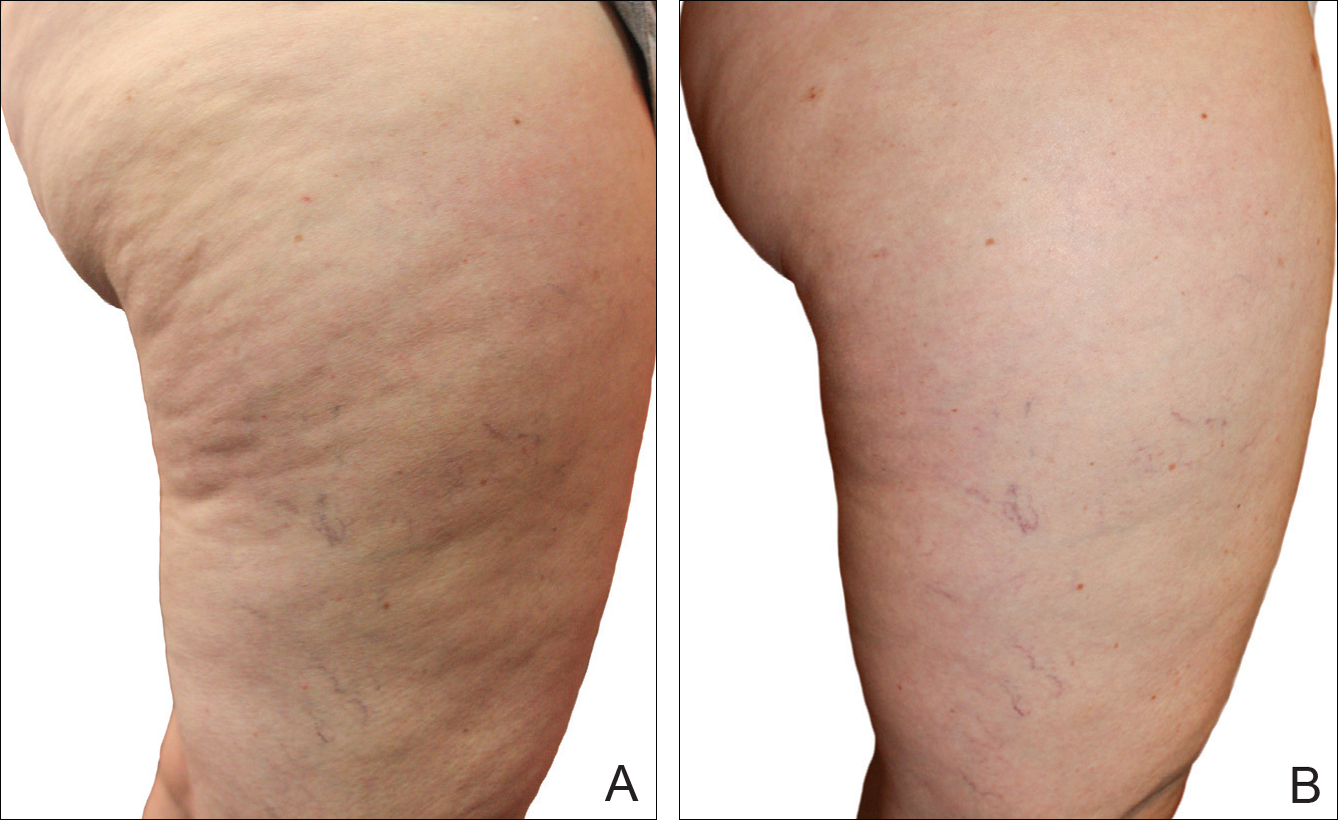
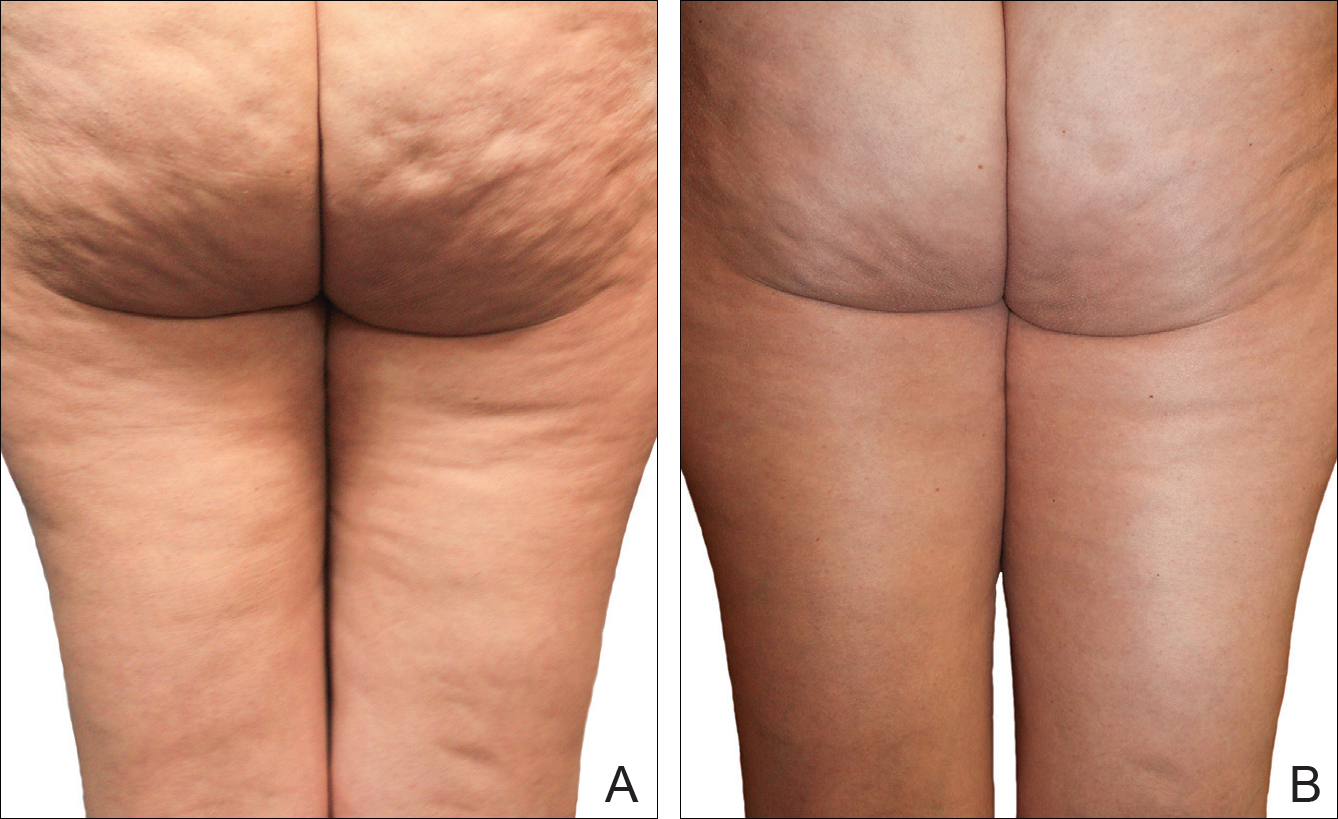
Digital photographs were taken with standardized lighting for all participants. Photographs were taken before the first treatment on the lateral and posterior aspects of the participant and were taken again at the end of the treatment program immediately before the last treatment. Participants were asked to contract the gluteal musculature for all photographs.
Two board-certified plastic surgeons were asked to rate the before/after photographs in a blinded manner. They graded each photograph on a rating scale of 0 to 10 (0=no cellulite; 10=worst possible cellulite). These data were analyzed using a Wilcoxon signed rank test. These data were compared to the participants self-evaluation of the appearance of cellulite in the photographs from the initial and final treatments using a rating scale of 0 to 10 (0=no cellulite; 10=worst possible cellulite).
The circumference of the widest part of the gluteal area was measured before and after treatment (+/–0.5 cm). The data were analyzed using a paired t test.
Results
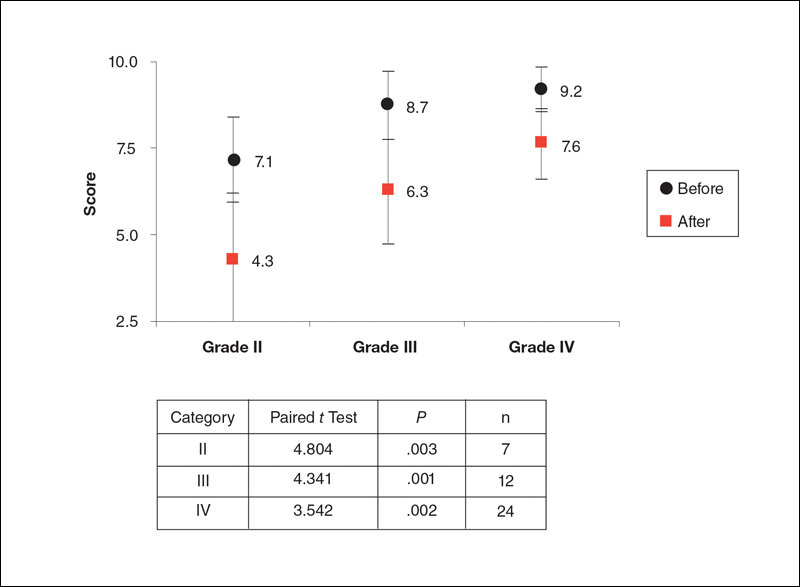
The study included 43 participants (age range, 21–67 years; mean age, 37.6 years; weight range, 51–97 kg; mean weight, 64.95 kg) who resided in the Midwestern United States, were interested in reducing their cellulite, and were willing to commit to treatment 2 times weekly for the duration of the 8-week study. Fourteen percent (6/43) of participants were smokers. Participant self-assessments were divided into 3 categories based on the Rossi and Vergnanini6 grading system: category II, n=7; category III, n=12; and category IV, n=24. Although all the categories in our analysis showed statistically significant improvements, we found that there was more improvement in category II participants versus category III, and then again more improvement in category III versus category IV. The data for each treatment were analyzed separately using a paired t test, as we were not interested in comparing categories, only the effect of the treatment. We were testing to see if the difference was greater than 0, and the paired t values were statistically significant in all cases (category II, P=.003; category III, P=.001; category IV, P=.002)(Figure 1).
Using a correlation analysis, we found that age, body weight, or body mass index were not significantly correlated with the difference between the before and after physician rating. The difference between before and after treatment also was independent of whether or not the participant exercised or had an adverse reaction to the belt. Adverse reactions to the belt were characterized by redness and/or minor raising of the skin immediately following the treatment. These reactions all dissipated within 12 hours. It also appeared that the rating scales correlated well with the participants self-perception of their cellulite and the improvements seen in the photographs (Figures 2 and 3).
The mean circumference of the widest part of the gluteal area before treatment was 100.2 cm and the standard deviation was 8.14 cm. The mean circumference after treatment was 98.3 cm and the standard deviation was 8.02 (t=–2.81; P<.05). Many of the women commented that they felt more “toned,” which probably accounted for the slight difference in circumference rather than weight loss.
Of the 2 blinded board-certified plastic surgeons, one physician rated all participants in category III as significantly improved (P<.05) and rated the other categories as marginally insignificantly improved; the second physician rated all categories as marginally insignificantly improved.
Comment
Although there are a large number of treatment protocols that have been introduced and studied for the reduction of the appearance of cellulite,4,9,11-18 many have not shown promising long-term results. Some treatments have shown improvement in the firmness of collagen and the dermis but not in the appearance of cellulite.22-25 One of the only treatments that has shown some promise is an expensive invasive treatment.20
The system used in this study was shown to be safe in all study participants. No significant adverse reactions were noted, and each participant successfully completed the protocol. Figures 2 and 3 show the strong correlation between the treatment and the reduction in the visible appearance of cellulite in this study population, which was supported by statistical analysis, particularly the participant self-reported ratings. The participants and the blinded physicians were not in agreement on the improvement of cellulite. Although the participants knew the changes that occurred to their bodies, the physicians only had photographs from which to make their decisions. The participants clearly observed noticeable differences to their bodies, while the physicians either saw no change or some improvement.
The physicians were asked to evaluate only the cellulite, but the process we employed changed more than the cellulite. The first step in the process was a toning of the legs and buttocks, which was readily observable by the patients but was outside the scope of the physicians’ assessment. After the body toning, the cellulite began to improve. It is possible that the participants were responding to the entire process, which clearly was positive, while the physicians were responding only to the cellulite end point.
Our treatment regimen accomplished reduction of the visible appearance of cellulite by breaking down connective tissue septa as well as increasing the thickness of the dermis and its elasticity. It also helped reduce subcutaneous fat, improve circulation, and reduce general lymphatic congestion. The parallel motions of the unit could be adjusted, but we kept them at a mid-level range of motion. The motion at this frequency would have a tendency to not only heat the epidermis and dermal layer that we were attempting to affect but would also help accomplish breaking down the septa and improving the elasticity of the dermis. Also, the rapid motion over a period of time of pulling the dermis parallel to the subdermal tissue and fascia most likely helped improve the circulation and lymphatic flow in treated areas as well as possibly broke down the subcutaneous fat. All of these factors appear to have led to an improvement in the appearance of cellulite in our study participants.
A maintenance-type program, if continued, would likely demonstrate improved results by further breaking down the septa and improving the other factors that reduce the appearance of cellulite. We believe that the participants would eventually be able to discontinue the use of the unit or reduce its use substantially once the desired results were obtained.
When utilizing the device, the participants were in a standing posture and leaning into the belt with a moderate force, which seemed to secondarily improve the tone of the gluteal and thigh musculature that was being treated. It may be that the oscillatory motion and the standing posture caused the muscles to isometrically co-contract, adding a secondary exerciselike effect.26-29
Proving our suggested mechanisms of action would require tissue biopsies and/or magnetic resonance imaging studies that were beyond the scope of this study. However, regardless of the mechanism of action, we do believe that this treatment has been shown to be effective, convenient, and most importantly safe.
Conclusion
The unique device that was utilized in our study is a safe and cost-effective method of reducing the appearance of cellulite for home use and would allow for a noninvasive, low-risk procedure.
- Scherwitz C, Braun-Falco O. So-called cellulite. J Dermatol Surg Oncol. 1978;4:230-234.
- Khan MH, Victor F, Rao B, et al. Treatment of cellulite: part I. pathophysiology. J Am Acad Dermatol. 2010;62:361-370, quiz 371-372.
- Querleux B, Cornillon C, Jolivet O, et al. Anatomy and physiology of subcutaneous adipose tissue by in vivo magnetic resonance imaging and spectroscopy: relationships with sex and presence of cellulite. Skin Res Technol. 2002;8:118-124.
- Rawlings A. Cellulite and its treatment. Int J Cos Sci. 2006;28:175-190.
- Rosenbaum M, Prieto V, Hellmer J, et al. An exploratory investigation of the morphology and biochemistry of cellulite. Plast Reconstr Surg. 1998;101:1934-1939.
- Rossi AB, Vergnanini AL. Cellulite: a review. J Eur Acad Dermatol Venereol. 2000;14:251-262.
- Smalls LK, Hicks M, Passeretti D, et al. Effect of weight loss on cellulite: gynoid lypodystrophy. Plast Reconstr Surg. 2006;118:510-516.
- Nürnberger F, Müller G. So-called cellulite: an invented disease. J Dermatol Surg Oncol. 1978;4:221-229.
- Mirrashed F, Sharp JC, Krause V, et al. Pilot study of dermal and subcutaneous fat structures by MRI in individuals who differ in gender, BMI, and cellulite grading. Skin Res Technol. 2004;10:161-168.
- Khan M, Victor F, Rao B, et al. Treatment of cellulite, part II. advances and controversies. J Am Acad Dermatol. 2010;62:373-384.
- Collis N, Elliot L, Sharp C, et al. Cellulite treatment: a myth or reality: a prospective randomized, controlled trial of two therapies, endermologie and aminophylline cream. Plast Reconstr Surg. 1999;104:1110-1114.
- Adcock D, Paulsen S, Jabour K, et al. Analysis of the effects of deep mechanical massage in the porcine model. Plast Reconstr Surg. 2000;108:233-240.
- Güleç AT. Treatment of cellulite with LPG endermologie. Int J Dermatol. 2009;48:265-270.
- Piérard-Franchimont C, Piérard GE, Henry F, et al. A randomized, placebo-controlled trial of tropical retinol in the treatment of cellulite. Am J Clin Dermatol. 2000;1:369-374.
- Coleman WP. Liposuction. In: Coleman WP, Hanke CW, Alt TH, eds. Cosmetic Surgery of the Skin: Principles and Practice. Philadelphia, PA: BC Decker; 1991:213-238.
- ASPS guiding principles for mesotherapy/injection lipolysis. American Society of Plastic Surgeons website. http://www.plasticsurgery.org/Documents/medical-professionals/health-policy/guiding-principles/ASPS-Guiding-Principles-for-Mesotherapy-Injection-Lipolysis-7-08.pdf. Published July 2008. Accessed February 17, 2016.
- Kulick MI. Evaluation of a noninvasive, dual-wavelength laser-suction and massage device for the regional treatment of cellulite. Plast Reconstr Surg. 2010;125:1788-1796.
- Nootheti PK, Magpantay A, Yosowitz G, et al. A single center, randomized, comparative, prospective clinical study to determine the efficacy of the VelaSmooth system versus the TriActive system for the treatment of cellulite. Lasers Surg Med. 2006;38:908-912.
- DiBernardo BE. Treatment of cellulite using a 1440-nm pulsed laser with one-year follow up. Aesthet Surg J. 2011;31:328-341.
- Johannes L. New laser aims to zap cellulite at the source. Wall Street Journal. July 3, 2012. http://www.wsj.com/articles/SB10001424052702303649504577496981754619546. Accessed November 21, 2016.
- Burns AJ. Commentary on: treatment of cellulite using a 1440-nm pulsed laser with one-year follow up: preliminary report. Aesthet Surg J. 2011;31:342-343.
- Teitelbaum SA, Burns JL, Kubota J, et al. Noninvasive body contouring by focused ultrasound: safety efficacy of the contour I device in a multicenter, controlled, clinical study. Plast Reconstr Surg. 2007;120:779-789.
- Brown SA, Greenbaum L, Shtukmaster S, et al. Characterization of nonthermal focused ultrasound for noninvasive selective fat cell disruption (lysis): technical and preclinical assessment. Plast Reconstr Surg. 2009;124:92-101.
- Angehrn F, Kuhn C, Voss A. Can cellulite be treated with low energy extracorporeal shock wave therapy? Clin Interv Aging. 2007;2:623-630.
- Christ C, Brenke R, Sattler G, et al. Improvement in skin elasticity in the treatment of cellulite and connective tissue weakness by means of extracorporeal pulse activation therapy. Aesthet Surg J. 2008;28:538-544.
- Bosco C, Colli R, Introini E, et al. Adaptive responses of human skeletal muscle to vibration exposure. Clin Physiol. 1999;19:183-187.
- Luo J, McNamara B, Moran K. The use of vibration training to enhance muscle strength and power. Sports Med. 2005;35:23-41.
- Annino G, Padua E, Castagna C, et al. Effect of whole body vibration training on lower limb performance in selected high-level ballet students. J Strength Cond Res. 2007;21:1072-1076.
- Verschueren SM, Roelants M, Delecluse C, et al. Effect of 6-month whole body vibration training on hip density, muscle strength, and postural control in postmenopausal women: a randomized controlled pilot study [published online December 22, 2003]. J Bone Miner Res. 2004;19:352-359.
Cellulite is a cosmetic problem, not a disease process. It affects 85% to 90% of all women worldwide and was described nearly 100 years ago.1 Causes may be genetic, hormonal, or vascular in nature and may be related to the septa configuration in the subdermal tissue. Fibrosis at the dermal-subcutaneous junction as well as decreased vascular and lymphatic circulation also may be causative factors.
Cellulite has a multifactorial etiology. Khan et al2 noted that there are specific classic patterns of cellulite that affect women exclusively. White women tend to have somewhat higher rates of cellulite than Asian women. The authors also stated that lifestyle factors such as high carbohydrate diets may lead to an increase in total body fat content, which enhances the appearance of cellulite.2
The subdermal anatomy affects the appearance of cellulite. Utilizing in vivo magnetic resonance imaging, Querleux et al3 showed that women with visible cellulite have dermal septa that are thinner and generally more perpendicular to the skin’s surface than women without cellulite. In women without cellulite, the orientation of the septa is more angled into a crisscross pattern. In women with a high percentage of perpendicular septa, the perpendicular septa allow for fat herniation with dimpling of the skin compared to the crisscross septa pattern.2 Other investigators have discussed the reduction of blood flow in specific areas of the body in women, particularly in cellulite-prone areas such as the buttocks and thighs, as another causative factor.2,4,5 Rossi and Vergnanini6 showed that the blood flow was 35% lower in affected cellulite regions than in nonaffected regions without cellulite, which can cause congestion of blood and lymphatic flow and increased subdermal pressure, thus increasing the appearance of cellulite.
Although there is some controversy regarding the effects of weight loss on the appearance of cellulite,2,7 it appears that the subdermal septa and morphology have more of an effect on the appearance of cellulite.2,3,8
Rossi and Vergnanini6 proposed a 4-grade system for evaluating the appearance of cellulite (grade I, no cellulite; grade II, skin that is smooth and without any pronounced dimpling upon standing or lying down but may show some dimpling upon pinching and strong muscle contraction; grade III, cellulite is present in upright positions but not when the patient is in a supine position; grade IV, cellulite can be seen when the patient is standing and in a supine position). Both grades III and IV can be exacerbated by maximal voluntary contraction and strong pinching of the skin because these actions cause the subcutaneous fat to move toward the surface of the skin between the septa. This grading system aligns with categories I through III described by Mirrashed et al.9
There are many cellulite treatments available but few actually create a reduction in the visible appearance of cellulite. A number of these treatments were reviewed by Khan et al,10 including massage; a noninvasive suction-assisted massage technique; and topical agents such as xanthine, retinols, and other botanicals.4,11-14 Liposuction has not been shown to be effective in the treatment of cellulite and in fact may increase the appearance of cellulite.9,15 Mesotherapy, a modality that entails injecting substances into the subcutaneous fat layer, is another treatment of cellulite. Two of the most common agents purported to dissolve fat include phosphatidylcholine and sodium deoxycholate. The efficacy and safety of mesotherapy remains controversial and unproven. A July 2008 position statement from the American Society of Plastic Surgeons stated that “low levels of validity and quality of the literature does not allow [American Society of Plastic Surgeons] to support a recommendation for the use of mesotherapy/injection lipolysis for fat reduction.”16 Other modalities such as noninvasive dual-wavelength laser/suction devices; low-energy diode laser, contact cooling, suction, and massage devices; and infrared, bipolar radiofrequency, and suction with mechanical massage devices are available and show some small improvements in the visible appearance of cellulite, but no rating scales were used in any of these studies.17,18 DiBernardo19 utilized a 1440-nm pulsed laser to treat cellulite. It is an invasive treatment that works by breaking down some of the connective tissue septa responsible for the majority and greater severity of the dermal dimpling seen in cellulite, increasing the thickness of the dermis as well as its elasticity, reducing subcutaneous fat, and improving circulation and reducing general lymphatic congestion.19 The system showed promise but was an invasive treatment, and one session could cost $5000 to $7000 for bilateral areas and another $2500 for each additional area.20 Burns21 expressed that the short-term results showed promise in reducing the appearance of cellulite. Noninvasive ultrasound22,23 as well as extracorporeal shock wave therapy24,25 also has shown some improvement in the firmness of collagen but generally not in the appearance of cellulite.
We sought to evaluate the efficacy and safety of a noninvasive mechanical treatment of cellulite.
Methods
This study was conducted in accordance with the guidelines set forth by the US Department of Health and Human Services’ Policy for Protection of Human Research Subjects and the World Medical Association’s Declaration of Helsinki. Participants were recruited through local area medical facilities in southeastern Michigan. Written informed consent was obtained from all participants prior to beginning the study.
Patients with grades II to IV cellulite, according to the Rossi and Vergnanini6 grading system, were allowed to participate. All participants in the study were asked not to make lifestyle changes (eg, exercise habits, diet) or use any other treatments for cellulite that might be available to them during the study period. Exclusion criteria included history of deep vein thrombosis, cancer diagnosed within the last year, pregnancy, hemophilia, severe lymphedema, presence of a pacemaker, epilepsy, seizure disorder, or current use of anticoagulants. History of partial or total joint replacements, acute hernia, nonunited fractures, advanced arthritis, or detached retina also excluded participation in the study.
Participants completed an 8-week, twice-weekly treatment protocol with a noninvasive mechanical device performed in clinic. The device consisted of a 10.16-cm belt with a layer of nonslip material wrapped around the belt. The belt was attached to a mechanical oscillator. We adjusted the stroke length to approximately 2 cm and moved the dermis at that length at approximately 1000 strokes per minute.
Each participant was treated for a total treatment time of 18 to 24 minutes. The total treatment area included the top of the iliac crest to just above the top of the popliteal space. The width of the belt (10.16 cm) was equal to 1 individual treatment area. Each individual treatment area was treated for 2 minutes. First the buttocks and bilateral thighs were treated, followed by the right lateral thigh and the left lateral thigh. The belt was moved progressively down the total treatment area until all individual treatment areas were addressed. The average participant had 3 to 4 bilateral thigh and buttocks treatment areas and 3 to 4 lateral treatment areas on both the left and right sides of the body.
Digital photographs were taken with standardized lighting for all participants. Photographs were taken before the first treatment on the lateral and posterior aspects of the participant and were taken again at the end of the treatment program immediately before the last treatment. Participants were asked to contract the gluteal musculature for all photographs.
Two board-certified plastic surgeons were asked to rate the before/after photographs in a blinded manner. They graded each photograph on a rating scale of 0 to 10 (0=no cellulite; 10=worst possible cellulite). These data were analyzed using a Wilcoxon signed rank test. These data were compared to the participants self-evaluation of the appearance of cellulite in the photographs from the initial and final treatments using a rating scale of 0 to 10 (0=no cellulite; 10=worst possible cellulite).
The circumference of the widest part of the gluteal area was measured before and after treatment (+/–0.5 cm). The data were analyzed using a paired t test.
Results
The study included 43 participants (age range, 21–67 years; mean age, 37.6 years; weight range, 51–97 kg; mean weight, 64.95 kg) who resided in the Midwestern United States, were interested in reducing their cellulite, and were willing to commit to treatment 2 times weekly for the duration of the 8-week study. Fourteen percent (6/43) of participants were smokers. Participant self-assessments were divided into 3 categories based on the Rossi and Vergnanini6 grading system: category II, n=7; category III, n=12; and category IV, n=24. Although all the categories in our analysis showed statistically significant improvements, we found that there was more improvement in category II participants versus category III, and then again more improvement in category III versus category IV. The data for each treatment were analyzed separately using a paired t test, as we were not interested in comparing categories, only the effect of the treatment. We were testing to see if the difference was greater than 0, and the paired t values were statistically significant in all cases (category II, P=.003; category III, P=.001; category IV, P=.002)(Figure 1).

Using a correlation analysis, we found that age, body weight, or body mass index were not significantly correlated with the difference between the before and after physician rating. The difference between before and after treatment also was independent of whether or not the participant exercised or had an adverse reaction to the belt. Adverse reactions to the belt were characterized by redness and/or minor raising of the skin immediately following the treatment. These reactions all dissipated within 12 hours. It also appeared that the rating scales correlated well with the participants self-perception of their cellulite and the improvements seen in the photographs (Figures 2 and 3).


The mean circumference of the widest part of the gluteal area before treatment was 100.2 cm and the standard deviation was 8.14 cm. The mean circumference after treatment was 98.3 cm and the standard deviation was 8.02 (t=–2.81; P<.05). Many of the women commented that they felt more “toned,” which probably accounted for the slight difference in circumference rather than weight loss.
Of the 2 blinded board-certified plastic surgeons, one physician rated all participants in category III as significantly improved (P<.05) and rated the other categories as marginally insignificantly improved; the second physician rated all categories as marginally insignificantly improved.
Comment
Although there are a large number of treatment protocols that have been introduced and studied for the reduction of the appearance of cellulite,4,9,11-18 many have not shown promising long-term results. Some treatments have shown improvement in the firmness of collagen and the dermis but not in the appearance of cellulite.22-25 One of the only treatments that has shown some promise is an expensive invasive treatment.20
The system used in this study was shown to be safe in all study participants. No significant adverse reactions were noted, and each participant successfully completed the protocol. Figures 2 and 3 show the strong correlation between the treatment and the reduction in the visible appearance of cellulite in this study population, which was supported by statistical analysis, particularly the participant self-reported ratings. The participants and the blinded physicians were not in agreement on the improvement of cellulite. Although the participants knew the changes that occurred to their bodies, the physicians only had photographs from which to make their decisions. The participants clearly observed noticeable differences to their bodies, while the physicians either saw no change or some improvement.
The physicians were asked to evaluate only the cellulite, but the process we employed changed more than the cellulite. The first step in the process was a toning of the legs and buttocks, which was readily observable by the patients but was outside the scope of the physicians’ assessment. After the body toning, the cellulite began to improve. It is possible that the participants were responding to the entire process, which clearly was positive, while the physicians were responding only to the cellulite end point.
Our treatment regimen accomplished reduction of the visible appearance of cellulite by breaking down connective tissue septa as well as increasing the thickness of the dermis and its elasticity. It also helped reduce subcutaneous fat, improve circulation, and reduce general lymphatic congestion. The parallel motions of the unit could be adjusted, but we kept them at a mid-level range of motion. The motion at this frequency would have a tendency to not only heat the epidermis and dermal layer that we were attempting to affect but would also help accomplish breaking down the septa and improving the elasticity of the dermis. Also, the rapid motion over a period of time of pulling the dermis parallel to the subdermal tissue and fascia most likely helped improve the circulation and lymphatic flow in treated areas as well as possibly broke down the subcutaneous fat. All of these factors appear to have led to an improvement in the appearance of cellulite in our study participants.
A maintenance-type program, if continued, would likely demonstrate improved results by further breaking down the septa and improving the other factors that reduce the appearance of cellulite. We believe that the participants would eventually be able to discontinue the use of the unit or reduce its use substantially once the desired results were obtained.
When utilizing the device, the participants were in a standing posture and leaning into the belt with a moderate force, which seemed to secondarily improve the tone of the gluteal and thigh musculature that was being treated. It may be that the oscillatory motion and the standing posture caused the muscles to isometrically co-contract, adding a secondary exerciselike effect.26-29
Proving our suggested mechanisms of action would require tissue biopsies and/or magnetic resonance imaging studies that were beyond the scope of this study. However, regardless of the mechanism of action, we do believe that this treatment has been shown to be effective, convenient, and most importantly safe.
Conclusion
The unique device that was utilized in our study is a safe and cost-effective method of reducing the appearance of cellulite for home use and would allow for a noninvasive, low-risk procedure.
Cellulite is a cosmetic problem, not a disease process. It affects 85% to 90% of all women worldwide and was described nearly 100 years ago.1 Causes may be genetic, hormonal, or vascular in nature and may be related to the septa configuration in the subdermal tissue. Fibrosis at the dermal-subcutaneous junction as well as decreased vascular and lymphatic circulation also may be causative factors.
Cellulite has a multifactorial etiology. Khan et al2 noted that there are specific classic patterns of cellulite that affect women exclusively. White women tend to have somewhat higher rates of cellulite than Asian women. The authors also stated that lifestyle factors such as high carbohydrate diets may lead to an increase in total body fat content, which enhances the appearance of cellulite.2
The subdermal anatomy affects the appearance of cellulite. Utilizing in vivo magnetic resonance imaging, Querleux et al3 showed that women with visible cellulite have dermal septa that are thinner and generally more perpendicular to the skin’s surface than women without cellulite. In women without cellulite, the orientation of the septa is more angled into a crisscross pattern. In women with a high percentage of perpendicular septa, the perpendicular septa allow for fat herniation with dimpling of the skin compared to the crisscross septa pattern.2 Other investigators have discussed the reduction of blood flow in specific areas of the body in women, particularly in cellulite-prone areas such as the buttocks and thighs, as another causative factor.2,4,5 Rossi and Vergnanini6 showed that the blood flow was 35% lower in affected cellulite regions than in nonaffected regions without cellulite, which can cause congestion of blood and lymphatic flow and increased subdermal pressure, thus increasing the appearance of cellulite.
Although there is some controversy regarding the effects of weight loss on the appearance of cellulite,2,7 it appears that the subdermal septa and morphology have more of an effect on the appearance of cellulite.2,3,8
Rossi and Vergnanini6 proposed a 4-grade system for evaluating the appearance of cellulite (grade I, no cellulite; grade II, skin that is smooth and without any pronounced dimpling upon standing or lying down but may show some dimpling upon pinching and strong muscle contraction; grade III, cellulite is present in upright positions but not when the patient is in a supine position; grade IV, cellulite can be seen when the patient is standing and in a supine position). Both grades III and IV can be exacerbated by maximal voluntary contraction and strong pinching of the skin because these actions cause the subcutaneous fat to move toward the surface of the skin between the septa. This grading system aligns with categories I through III described by Mirrashed et al.9
There are many cellulite treatments available but few actually create a reduction in the visible appearance of cellulite. A number of these treatments were reviewed by Khan et al,10 including massage; a noninvasive suction-assisted massage technique; and topical agents such as xanthine, retinols, and other botanicals.4,11-14 Liposuction has not been shown to be effective in the treatment of cellulite and in fact may increase the appearance of cellulite.9,15 Mesotherapy, a modality that entails injecting substances into the subcutaneous fat layer, is another treatment of cellulite. Two of the most common agents purported to dissolve fat include phosphatidylcholine and sodium deoxycholate. The efficacy and safety of mesotherapy remains controversial and unproven. A July 2008 position statement from the American Society of Plastic Surgeons stated that “low levels of validity and quality of the literature does not allow [American Society of Plastic Surgeons] to support a recommendation for the use of mesotherapy/injection lipolysis for fat reduction.”16 Other modalities such as noninvasive dual-wavelength laser/suction devices; low-energy diode laser, contact cooling, suction, and massage devices; and infrared, bipolar radiofrequency, and suction with mechanical massage devices are available and show some small improvements in the visible appearance of cellulite, but no rating scales were used in any of these studies.17,18 DiBernardo19 utilized a 1440-nm pulsed laser to treat cellulite. It is an invasive treatment that works by breaking down some of the connective tissue septa responsible for the majority and greater severity of the dermal dimpling seen in cellulite, increasing the thickness of the dermis as well as its elasticity, reducing subcutaneous fat, and improving circulation and reducing general lymphatic congestion.19 The system showed promise but was an invasive treatment, and one session could cost $5000 to $7000 for bilateral areas and another $2500 for each additional area.20 Burns21 expressed that the short-term results showed promise in reducing the appearance of cellulite. Noninvasive ultrasound22,23 as well as extracorporeal shock wave therapy24,25 also has shown some improvement in the firmness of collagen but generally not in the appearance of cellulite.
We sought to evaluate the efficacy and safety of a noninvasive mechanical treatment of cellulite.
Methods
This study was conducted in accordance with the guidelines set forth by the US Department of Health and Human Services’ Policy for Protection of Human Research Subjects and the World Medical Association’s Declaration of Helsinki. Participants were recruited through local area medical facilities in southeastern Michigan. Written informed consent was obtained from all participants prior to beginning the study.
Patients with grades II to IV cellulite, according to the Rossi and Vergnanini6 grading system, were allowed to participate. All participants in the study were asked not to make lifestyle changes (eg, exercise habits, diet) or use any other treatments for cellulite that might be available to them during the study period. Exclusion criteria included history of deep vein thrombosis, cancer diagnosed within the last year, pregnancy, hemophilia, severe lymphedema, presence of a pacemaker, epilepsy, seizure disorder, or current use of anticoagulants. History of partial or total joint replacements, acute hernia, nonunited fractures, advanced arthritis, or detached retina also excluded participation in the study.
Participants completed an 8-week, twice-weekly treatment protocol with a noninvasive mechanical device performed in clinic. The device consisted of a 10.16-cm belt with a layer of nonslip material wrapped around the belt. The belt was attached to a mechanical oscillator. We adjusted the stroke length to approximately 2 cm and moved the dermis at that length at approximately 1000 strokes per minute.
Each participant was treated for a total treatment time of 18 to 24 minutes. The total treatment area included the top of the iliac crest to just above the top of the popliteal space. The width of the belt (10.16 cm) was equal to 1 individual treatment area. Each individual treatment area was treated for 2 minutes. First the buttocks and bilateral thighs were treated, followed by the right lateral thigh and the left lateral thigh. The belt was moved progressively down the total treatment area until all individual treatment areas were addressed. The average participant had 3 to 4 bilateral thigh and buttocks treatment areas and 3 to 4 lateral treatment areas on both the left and right sides of the body.
Digital photographs were taken with standardized lighting for all participants. Photographs were taken before the first treatment on the lateral and posterior aspects of the participant and were taken again at the end of the treatment program immediately before the last treatment. Participants were asked to contract the gluteal musculature for all photographs.
Two board-certified plastic surgeons were asked to rate the before/after photographs in a blinded manner. They graded each photograph on a rating scale of 0 to 10 (0=no cellulite; 10=worst possible cellulite). These data were analyzed using a Wilcoxon signed rank test. These data were compared to the participants self-evaluation of the appearance of cellulite in the photographs from the initial and final treatments using a rating scale of 0 to 10 (0=no cellulite; 10=worst possible cellulite).
The circumference of the widest part of the gluteal area was measured before and after treatment (+/–0.5 cm). The data were analyzed using a paired t test.
Results
The study included 43 participants (age range, 21–67 years; mean age, 37.6 years; weight range, 51–97 kg; mean weight, 64.95 kg) who resided in the Midwestern United States, were interested in reducing their cellulite, and were willing to commit to treatment 2 times weekly for the duration of the 8-week study. Fourteen percent (6/43) of participants were smokers. Participant self-assessments were divided into 3 categories based on the Rossi and Vergnanini6 grading system: category II, n=7; category III, n=12; and category IV, n=24. Although all the categories in our analysis showed statistically significant improvements, we found that there was more improvement in category II participants versus category III, and then again more improvement in category III versus category IV. The data for each treatment were analyzed separately using a paired t test, as we were not interested in comparing categories, only the effect of the treatment. We were testing to see if the difference was greater than 0, and the paired t values were statistically significant in all cases (category II, P=.003; category III, P=.001; category IV, P=.002)(Figure 1).

Using a correlation analysis, we found that age, body weight, or body mass index were not significantly correlated with the difference between the before and after physician rating. The difference between before and after treatment also was independent of whether or not the participant exercised or had an adverse reaction to the belt. Adverse reactions to the belt were characterized by redness and/or minor raising of the skin immediately following the treatment. These reactions all dissipated within 12 hours. It also appeared that the rating scales correlated well with the participants self-perception of their cellulite and the improvements seen in the photographs (Figures 2 and 3).


The mean circumference of the widest part of the gluteal area before treatment was 100.2 cm and the standard deviation was 8.14 cm. The mean circumference after treatment was 98.3 cm and the standard deviation was 8.02 (t=–2.81; P<.05). Many of the women commented that they felt more “toned,” which probably accounted for the slight difference in circumference rather than weight loss.
Of the 2 blinded board-certified plastic surgeons, one physician rated all participants in category III as significantly improved (P<.05) and rated the other categories as marginally insignificantly improved; the second physician rated all categories as marginally insignificantly improved.
Comment
Although there are a large number of treatment protocols that have been introduced and studied for the reduction of the appearance of cellulite,4,9,11-18 many have not shown promising long-term results. Some treatments have shown improvement in the firmness of collagen and the dermis but not in the appearance of cellulite.22-25 One of the only treatments that has shown some promise is an expensive invasive treatment.20
The system used in this study was shown to be safe in all study participants. No significant adverse reactions were noted, and each participant successfully completed the protocol. Figures 2 and 3 show the strong correlation between the treatment and the reduction in the visible appearance of cellulite in this study population, which was supported by statistical analysis, particularly the participant self-reported ratings. The participants and the blinded physicians were not in agreement on the improvement of cellulite. Although the participants knew the changes that occurred to their bodies, the physicians only had photographs from which to make their decisions. The participants clearly observed noticeable differences to their bodies, while the physicians either saw no change or some improvement.
The physicians were asked to evaluate only the cellulite, but the process we employed changed more than the cellulite. The first step in the process was a toning of the legs and buttocks, which was readily observable by the patients but was outside the scope of the physicians’ assessment. After the body toning, the cellulite began to improve. It is possible that the participants were responding to the entire process, which clearly was positive, while the physicians were responding only to the cellulite end point.
Our treatment regimen accomplished reduction of the visible appearance of cellulite by breaking down connective tissue septa as well as increasing the thickness of the dermis and its elasticity. It also helped reduce subcutaneous fat, improve circulation, and reduce general lymphatic congestion. The parallel motions of the unit could be adjusted, but we kept them at a mid-level range of motion. The motion at this frequency would have a tendency to not only heat the epidermis and dermal layer that we were attempting to affect but would also help accomplish breaking down the septa and improving the elasticity of the dermis. Also, the rapid motion over a period of time of pulling the dermis parallel to the subdermal tissue and fascia most likely helped improve the circulation and lymphatic flow in treated areas as well as possibly broke down the subcutaneous fat. All of these factors appear to have led to an improvement in the appearance of cellulite in our study participants.
A maintenance-type program, if continued, would likely demonstrate improved results by further breaking down the septa and improving the other factors that reduce the appearance of cellulite. We believe that the participants would eventually be able to discontinue the use of the unit or reduce its use substantially once the desired results were obtained.
When utilizing the device, the participants were in a standing posture and leaning into the belt with a moderate force, which seemed to secondarily improve the tone of the gluteal and thigh musculature that was being treated. It may be that the oscillatory motion and the standing posture caused the muscles to isometrically co-contract, adding a secondary exerciselike effect.26-29
Proving our suggested mechanisms of action would require tissue biopsies and/or magnetic resonance imaging studies that were beyond the scope of this study. However, regardless of the mechanism of action, we do believe that this treatment has been shown to be effective, convenient, and most importantly safe.
Conclusion
The unique device that was utilized in our study is a safe and cost-effective method of reducing the appearance of cellulite for home use and would allow for a noninvasive, low-risk procedure.
- Scherwitz C, Braun-Falco O. So-called cellulite. J Dermatol Surg Oncol. 1978;4:230-234.
- Khan MH, Victor F, Rao B, et al. Treatment of cellulite: part I. pathophysiology. J Am Acad Dermatol. 2010;62:361-370, quiz 371-372.
- Querleux B, Cornillon C, Jolivet O, et al. Anatomy and physiology of subcutaneous adipose tissue by in vivo magnetic resonance imaging and spectroscopy: relationships with sex and presence of cellulite. Skin Res Technol. 2002;8:118-124.
- Rawlings A. Cellulite and its treatment. Int J Cos Sci. 2006;28:175-190.
- Rosenbaum M, Prieto V, Hellmer J, et al. An exploratory investigation of the morphology and biochemistry of cellulite. Plast Reconstr Surg. 1998;101:1934-1939.
- Rossi AB, Vergnanini AL. Cellulite: a review. J Eur Acad Dermatol Venereol. 2000;14:251-262.
- Smalls LK, Hicks M, Passeretti D, et al. Effect of weight loss on cellulite: gynoid lypodystrophy. Plast Reconstr Surg. 2006;118:510-516.
- Nürnberger F, Müller G. So-called cellulite: an invented disease. J Dermatol Surg Oncol. 1978;4:221-229.
- Mirrashed F, Sharp JC, Krause V, et al. Pilot study of dermal and subcutaneous fat structures by MRI in individuals who differ in gender, BMI, and cellulite grading. Skin Res Technol. 2004;10:161-168.
- Khan M, Victor F, Rao B, et al. Treatment of cellulite, part II. advances and controversies. J Am Acad Dermatol. 2010;62:373-384.
- Collis N, Elliot L, Sharp C, et al. Cellulite treatment: a myth or reality: a prospective randomized, controlled trial of two therapies, endermologie and aminophylline cream. Plast Reconstr Surg. 1999;104:1110-1114.
- Adcock D, Paulsen S, Jabour K, et al. Analysis of the effects of deep mechanical massage in the porcine model. Plast Reconstr Surg. 2000;108:233-240.
- Güleç AT. Treatment of cellulite with LPG endermologie. Int J Dermatol. 2009;48:265-270.
- Piérard-Franchimont C, Piérard GE, Henry F, et al. A randomized, placebo-controlled trial of tropical retinol in the treatment of cellulite. Am J Clin Dermatol. 2000;1:369-374.
- Coleman WP. Liposuction. In: Coleman WP, Hanke CW, Alt TH, eds. Cosmetic Surgery of the Skin: Principles and Practice. Philadelphia, PA: BC Decker; 1991:213-238.
- ASPS guiding principles for mesotherapy/injection lipolysis. American Society of Plastic Surgeons website. http://www.plasticsurgery.org/Documents/medical-professionals/health-policy/guiding-principles/ASPS-Guiding-Principles-for-Mesotherapy-Injection-Lipolysis-7-08.pdf. Published July 2008. Accessed February 17, 2016.
- Kulick MI. Evaluation of a noninvasive, dual-wavelength laser-suction and massage device for the regional treatment of cellulite. Plast Reconstr Surg. 2010;125:1788-1796.
- Nootheti PK, Magpantay A, Yosowitz G, et al. A single center, randomized, comparative, prospective clinical study to determine the efficacy of the VelaSmooth system versus the TriActive system for the treatment of cellulite. Lasers Surg Med. 2006;38:908-912.
- DiBernardo BE. Treatment of cellulite using a 1440-nm pulsed laser with one-year follow up. Aesthet Surg J. 2011;31:328-341.
- Johannes L. New laser aims to zap cellulite at the source. Wall Street Journal. July 3, 2012. http://www.wsj.com/articles/SB10001424052702303649504577496981754619546. Accessed November 21, 2016.
- Burns AJ. Commentary on: treatment of cellulite using a 1440-nm pulsed laser with one-year follow up: preliminary report. Aesthet Surg J. 2011;31:342-343.
- Teitelbaum SA, Burns JL, Kubota J, et al. Noninvasive body contouring by focused ultrasound: safety efficacy of the contour I device in a multicenter, controlled, clinical study. Plast Reconstr Surg. 2007;120:779-789.
- Brown SA, Greenbaum L, Shtukmaster S, et al. Characterization of nonthermal focused ultrasound for noninvasive selective fat cell disruption (lysis): technical and preclinical assessment. Plast Reconstr Surg. 2009;124:92-101.
- Angehrn F, Kuhn C, Voss A. Can cellulite be treated with low energy extracorporeal shock wave therapy? Clin Interv Aging. 2007;2:623-630.
- Christ C, Brenke R, Sattler G, et al. Improvement in skin elasticity in the treatment of cellulite and connective tissue weakness by means of extracorporeal pulse activation therapy. Aesthet Surg J. 2008;28:538-544.
- Bosco C, Colli R, Introini E, et al. Adaptive responses of human skeletal muscle to vibration exposure. Clin Physiol. 1999;19:183-187.
- Luo J, McNamara B, Moran K. The use of vibration training to enhance muscle strength and power. Sports Med. 2005;35:23-41.
- Annino G, Padua E, Castagna C, et al. Effect of whole body vibration training on lower limb performance in selected high-level ballet students. J Strength Cond Res. 2007;21:1072-1076.
- Verschueren SM, Roelants M, Delecluse C, et al. Effect of 6-month whole body vibration training on hip density, muscle strength, and postural control in postmenopausal women: a randomized controlled pilot study [published online December 22, 2003]. J Bone Miner Res. 2004;19:352-359.
- Scherwitz C, Braun-Falco O. So-called cellulite. J Dermatol Surg Oncol. 1978;4:230-234.
- Khan MH, Victor F, Rao B, et al. Treatment of cellulite: part I. pathophysiology. J Am Acad Dermatol. 2010;62:361-370, quiz 371-372.
- Querleux B, Cornillon C, Jolivet O, et al. Anatomy and physiology of subcutaneous adipose tissue by in vivo magnetic resonance imaging and spectroscopy: relationships with sex and presence of cellulite. Skin Res Technol. 2002;8:118-124.
- Rawlings A. Cellulite and its treatment. Int J Cos Sci. 2006;28:175-190.
- Rosenbaum M, Prieto V, Hellmer J, et al. An exploratory investigation of the morphology and biochemistry of cellulite. Plast Reconstr Surg. 1998;101:1934-1939.
- Rossi AB, Vergnanini AL. Cellulite: a review. J Eur Acad Dermatol Venereol. 2000;14:251-262.
- Smalls LK, Hicks M, Passeretti D, et al. Effect of weight loss on cellulite: gynoid lypodystrophy. Plast Reconstr Surg. 2006;118:510-516.
- Nürnberger F, Müller G. So-called cellulite: an invented disease. J Dermatol Surg Oncol. 1978;4:221-229.
- Mirrashed F, Sharp JC, Krause V, et al. Pilot study of dermal and subcutaneous fat structures by MRI in individuals who differ in gender, BMI, and cellulite grading. Skin Res Technol. 2004;10:161-168.
- Khan M, Victor F, Rao B, et al. Treatment of cellulite, part II. advances and controversies. J Am Acad Dermatol. 2010;62:373-384.
- Collis N, Elliot L, Sharp C, et al. Cellulite treatment: a myth or reality: a prospective randomized, controlled trial of two therapies, endermologie and aminophylline cream. Plast Reconstr Surg. 1999;104:1110-1114.
- Adcock D, Paulsen S, Jabour K, et al. Analysis of the effects of deep mechanical massage in the porcine model. Plast Reconstr Surg. 2000;108:233-240.
- Güleç AT. Treatment of cellulite with LPG endermologie. Int J Dermatol. 2009;48:265-270.
- Piérard-Franchimont C, Piérard GE, Henry F, et al. A randomized, placebo-controlled trial of tropical retinol in the treatment of cellulite. Am J Clin Dermatol. 2000;1:369-374.
- Coleman WP. Liposuction. In: Coleman WP, Hanke CW, Alt TH, eds. Cosmetic Surgery of the Skin: Principles and Practice. Philadelphia, PA: BC Decker; 1991:213-238.
- ASPS guiding principles for mesotherapy/injection lipolysis. American Society of Plastic Surgeons website. http://www.plasticsurgery.org/Documents/medical-professionals/health-policy/guiding-principles/ASPS-Guiding-Principles-for-Mesotherapy-Injection-Lipolysis-7-08.pdf. Published July 2008. Accessed February 17, 2016.
- Kulick MI. Evaluation of a noninvasive, dual-wavelength laser-suction and massage device for the regional treatment of cellulite. Plast Reconstr Surg. 2010;125:1788-1796.
- Nootheti PK, Magpantay A, Yosowitz G, et al. A single center, randomized, comparative, prospective clinical study to determine the efficacy of the VelaSmooth system versus the TriActive system for the treatment of cellulite. Lasers Surg Med. 2006;38:908-912.
- DiBernardo BE. Treatment of cellulite using a 1440-nm pulsed laser with one-year follow up. Aesthet Surg J. 2011;31:328-341.
- Johannes L. New laser aims to zap cellulite at the source. Wall Street Journal. July 3, 2012. http://www.wsj.com/articles/SB10001424052702303649504577496981754619546. Accessed November 21, 2016.
- Burns AJ. Commentary on: treatment of cellulite using a 1440-nm pulsed laser with one-year follow up: preliminary report. Aesthet Surg J. 2011;31:342-343.
- Teitelbaum SA, Burns JL, Kubota J, et al. Noninvasive body contouring by focused ultrasound: safety efficacy of the contour I device in a multicenter, controlled, clinical study. Plast Reconstr Surg. 2007;120:779-789.
- Brown SA, Greenbaum L, Shtukmaster S, et al. Characterization of nonthermal focused ultrasound for noninvasive selective fat cell disruption (lysis): technical and preclinical assessment. Plast Reconstr Surg. 2009;124:92-101.
- Angehrn F, Kuhn C, Voss A. Can cellulite be treated with low energy extracorporeal shock wave therapy? Clin Interv Aging. 2007;2:623-630.
- Christ C, Brenke R, Sattler G, et al. Improvement in skin elasticity in the treatment of cellulite and connective tissue weakness by means of extracorporeal pulse activation therapy. Aesthet Surg J. 2008;28:538-544.
- Bosco C, Colli R, Introini E, et al. Adaptive responses of human skeletal muscle to vibration exposure. Clin Physiol. 1999;19:183-187.
- Luo J, McNamara B, Moran K. The use of vibration training to enhance muscle strength and power. Sports Med. 2005;35:23-41.
- Annino G, Padua E, Castagna C, et al. Effect of whole body vibration training on lower limb performance in selected high-level ballet students. J Strength Cond Res. 2007;21:1072-1076.
- Verschueren SM, Roelants M, Delecluse C, et al. Effect of 6-month whole body vibration training on hip density, muscle strength, and postural control in postmenopausal women: a randomized controlled pilot study [published online December 22, 2003]. J Bone Miner Res. 2004;19:352-359.
Practice Points
- Several cellulite treatments have shown improvement in the firmness of collagen and the dermis but not in the appearance of cellulite.
- The noninvasive mechanical treatment for women with cellulite evaluated in this study showed a strong correlation between the treatment and the reduction in the visible appearance of cellulite in this study population.
Update on New Drugs in Dermatology
CenterWatch (http://www.centerwatch.com/) is an online resource that provides directories, analysis, and market research of medications that are either under clinical evaluation or available for use in patients. A list of currently approved drugs by the US Food and Drug Administration (FDA) also is available by specialty. It is important for dermatologists in-training to know about recently approved drugs and those that are in the pipeline, as these treatments may benefit patients who are unresponsive to other previously used medications. New drugs also may be useful for physicians who have a difficult time getting insurance to cover prescriptions for their patients, as most new medications have built-in patient assistance.
New Drugs in Dermatology
Actinic Keratosis
Ameluz (aminolevulinic acid hydrochloride)(Biofrontera AG) is a new drug that was approved in May 2016 for treatment of mild to moderate actinic keratosis on the face and scalp.1 It is only intended for in-office use on patients who may not be candidates for other treatment options for actinic keratosis. The product is a gel formulation that should be applied to cover the lesions and approximately 5 mm of the surrounding area with a film of approximately 1-mm thickness. The entire treatment area is then illuminated with a red light source, either with a narrow spectrum around 630 nm with a light dose of approximately 37 J/cm2 or a broader and continuous spectrum in the range of 570 to 670 nm with a light dose between 75 and 200 J/cm2.1 Similar to the previously used aminolevulinic acid treatment method for actinic keratosis, the patient may experience a burning stinging sensation throughout the treatment and the skin will then proceed to peel.
Psoriasis and Psoriatic Arthritis
Taltz (ixekizumab)(Eli Lilly and Company) was approved by the FDA in March 2016 for the treatment of moderate to severe plaque psoriasis.2 It is a humanized IL-17A antagonist that works when IgG4 monoclonal antibodies selectively bind with IL-17A cytokines and inhibit their interaction with the IL-17 receptor. Although this injectable medication is approved for the treatment of psoriasis, it also can potentially be used off label for the treatment of psoriatic arthritis and rheumatoid arthritis. The approved dosage is 160 mg (two 80-mg injections) at week 0, followed by 80 mg at weeks 2, 4, 6, 8, 10, and 12, then 80 mg every 4 weeks.2 Injectable immunomodulatory medications such as ixekizumab are ideal for patients in whom topical treatments and light therapy failed and they continue to have serious psoriatic discomfort as well as for those who have substantial body surface area coverage.
In January 2015, Cosentyx (secukinumab)(Novartis Corporation) was approved by the FDA.3 Similar to ixekizumab, this injectable is an IgG1 monoclonal antibody that selectively binds to the IL-17A cytokine and inhibits its interaction with the IL-17 receptor. It is approved for the treatment of moderate to severe plaque psoriasis and psoriatic arthritis. The approved dosage for plaque psoriasis is 300 mg (two 150-mg subcutaneous injections) at weeks 0 through 4 followed by 300 mg every 4 weeks as needed until clearance.3 Similar to ixekizumab, secukinumab may be used for the treatment of recalcitrant psoriasis or psoriasis with substantial body surface area involvement.
Melanoma
Cotellic (cobimetinib)(Genentech USA, Inc) was FDA approved in November 2015.4 Cobimetinib is a reversible inhibitor of mitogen-activated protein kinase (MAPK)/extracellular signal regulated kinase 1. Mitogen-activated protein kinase MEK1 and MEK2 are regulators of the extracellular signal-related kinase pathway, which promotes cellular proliferation. This pathway is key, as melanomas that have a BRAF V600E and kinase mutation continue to proliferate due to the constitutive activation of MEK1 and MEK2, further promoting cellular proliferation. Cobimetinib is approved for the treatment of melanoma in patients with unresectable or metastatic melanoma with a BRAF V600E or V600K mutation, in conjunction with vemurafenib. Zelboraf (vemurafenib)(Genentech USA, Inc), another inhibitor of BRAF V600E, also is used for the treatment of unresectable melanomas and was initially approved in 2011.5
BRAF is a serine/threonine protein kinase. When unregulated, it results in the deregulation of cell proliferation. According to Ascierto et al,6 50% of melanomas have a BRAF mutation, with nearly 90% of them with a V600E mutation. Hence, since the advent of direct chemotherapeutic agents such as BRAF inhibitors, clinical trials have shown notable reduction in mortality and morbidity of melanoma patients with BRAF mutations.6
Imlygic (talimogene laherparepvec)(Amgen, Inc) is a modified oncolytic viral therapy.7 This treatment was approved by the FDA in 2015 and replicates within tumors to produce granulocyte-macrophage colony-stimulating factor protein, which promotes an antitumor immune response within unresectable cutaneous, subcutaneous, and nodal melanoma lesions. Although it is not a gene-directed therapy, the melanoma does not require a specific mutation for treatment. Again, this medication is better served in conjunction with other melanoma chemotherapeutic and surgical interventions.
Submental Fat
Kybella (deoxycholic acid)(Allergan) is a nonhuman, nonanimal, synthetically created compound that is naturally found within the human body for the breakdown and absorption of dietary fat.8 This drug was FDA approved in 2015 for the improvement of the appearance of moderate subcutaneous fat under the chin. Patients are evaluated in clinic to determine if the submental fat would be responsive to an injectable or require more radical surgical intervention based on desired outcomes. The treatment is administered as 0.2-mL injections (up to a total of 10 mL) spaced 1-cm apart and ideally is repeated at regular intervals to evaluate for efficacy.
Basal Cell Carcinoma
Odomzo (sonidegib)(Novartis Corporation) was FDA approved in 2015 for locally advanced basal cell carcinoma.9 Odomzo is a smoothened antagonist that inhibits the hedgehog signaling pathway. Smoothened is a transmembrane protein that allows for signal transduction of hedgehog proteins.10 Protein patched homolog 1 binds to smoothened protein and prevents the signal transduction through the cell for Gli family zinc factor 1 to continue protein translation; however, when PTCH is mutated and can no longer bind to smoothened, tumor formation results, specifically basal cell carcinoma. Hence, sonidegib is for the treatment of basal cell carcinomas that have persisted despite radiation treatment and/or surgery as well as for patients who have multiple basal cell carcinomas that can no longer be treated with surgery or radiation.
Final Thoughts
Overall, although there are several medications that can be used in conjunction for treatment of dermatological conditions, it always is recommended to know what is in the pipeline as FDA-approved medications for dermatology.
- Ameluz [package insert]. Leverkusen, Germany: Biofrontera Bioscience GmbH; 2016.
- Taltz [package insert]. Indianapolis, IN: Eli Lilly and Company; 2016.
- Cosentyx [package insert]. East Hanover, NJ: Novartis Corporation; 2015.
- Cotellic [package insert]. San Francisco, CA: Genentech, Inc; 2016.
- Zelboraf [package insert]. San Francisco, CA: Genentech, Inc; 2016.
- Ascierto PA, Kirkwood JM, Grob JJ, et al. The role of BRAF V600 mutation in melanoma. J Transl Med. 2012;10:85.
- Imlygic (talimogene laherparepvec). Thousand Oaks, CA: Amgen Inc; 2015.
- Kybella [package insert]. West Lake Village, CA: Kythera Biopharmaceuticals, Inc; 2015.
- Odomzo [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corporation; 2015.
- Villavicencio EH, Walterhouse DO, Iannaccone PM. The sonic hedgehog-patched-gli pathway in human development and disease. Am J Hum Genet. 2000;67:1047-1054.
CenterWatch (http://www.centerwatch.com/) is an online resource that provides directories, analysis, and market research of medications that are either under clinical evaluation or available for use in patients. A list of currently approved drugs by the US Food and Drug Administration (FDA) also is available by specialty. It is important for dermatologists in-training to know about recently approved drugs and those that are in the pipeline, as these treatments may benefit patients who are unresponsive to other previously used medications. New drugs also may be useful for physicians who have a difficult time getting insurance to cover prescriptions for their patients, as most new medications have built-in patient assistance.
New Drugs in Dermatology
Actinic Keratosis
Ameluz (aminolevulinic acid hydrochloride)(Biofrontera AG) is a new drug that was approved in May 2016 for treatment of mild to moderate actinic keratosis on the face and scalp.1 It is only intended for in-office use on patients who may not be candidates for other treatment options for actinic keratosis. The product is a gel formulation that should be applied to cover the lesions and approximately 5 mm of the surrounding area with a film of approximately 1-mm thickness. The entire treatment area is then illuminated with a red light source, either with a narrow spectrum around 630 nm with a light dose of approximately 37 J/cm2 or a broader and continuous spectrum in the range of 570 to 670 nm with a light dose between 75 and 200 J/cm2.1 Similar to the previously used aminolevulinic acid treatment method for actinic keratosis, the patient may experience a burning stinging sensation throughout the treatment and the skin will then proceed to peel.
Psoriasis and Psoriatic Arthritis
Taltz (ixekizumab)(Eli Lilly and Company) was approved by the FDA in March 2016 for the treatment of moderate to severe plaque psoriasis.2 It is a humanized IL-17A antagonist that works when IgG4 monoclonal antibodies selectively bind with IL-17A cytokines and inhibit their interaction with the IL-17 receptor. Although this injectable medication is approved for the treatment of psoriasis, it also can potentially be used off label for the treatment of psoriatic arthritis and rheumatoid arthritis. The approved dosage is 160 mg (two 80-mg injections) at week 0, followed by 80 mg at weeks 2, 4, 6, 8, 10, and 12, then 80 mg every 4 weeks.2 Injectable immunomodulatory medications such as ixekizumab are ideal for patients in whom topical treatments and light therapy failed and they continue to have serious psoriatic discomfort as well as for those who have substantial body surface area coverage.
In January 2015, Cosentyx (secukinumab)(Novartis Corporation) was approved by the FDA.3 Similar to ixekizumab, this injectable is an IgG1 monoclonal antibody that selectively binds to the IL-17A cytokine and inhibits its interaction with the IL-17 receptor. It is approved for the treatment of moderate to severe plaque psoriasis and psoriatic arthritis. The approved dosage for plaque psoriasis is 300 mg (two 150-mg subcutaneous injections) at weeks 0 through 4 followed by 300 mg every 4 weeks as needed until clearance.3 Similar to ixekizumab, secukinumab may be used for the treatment of recalcitrant psoriasis or psoriasis with substantial body surface area involvement.
Melanoma
Cotellic (cobimetinib)(Genentech USA, Inc) was FDA approved in November 2015.4 Cobimetinib is a reversible inhibitor of mitogen-activated protein kinase (MAPK)/extracellular signal regulated kinase 1. Mitogen-activated protein kinase MEK1 and MEK2 are regulators of the extracellular signal-related kinase pathway, which promotes cellular proliferation. This pathway is key, as melanomas that have a BRAF V600E and kinase mutation continue to proliferate due to the constitutive activation of MEK1 and MEK2, further promoting cellular proliferation. Cobimetinib is approved for the treatment of melanoma in patients with unresectable or metastatic melanoma with a BRAF V600E or V600K mutation, in conjunction with vemurafenib. Zelboraf (vemurafenib)(Genentech USA, Inc), another inhibitor of BRAF V600E, also is used for the treatment of unresectable melanomas and was initially approved in 2011.5
BRAF is a serine/threonine protein kinase. When unregulated, it results in the deregulation of cell proliferation. According to Ascierto et al,6 50% of melanomas have a BRAF mutation, with nearly 90% of them with a V600E mutation. Hence, since the advent of direct chemotherapeutic agents such as BRAF inhibitors, clinical trials have shown notable reduction in mortality and morbidity of melanoma patients with BRAF mutations.6
Imlygic (talimogene laherparepvec)(Amgen, Inc) is a modified oncolytic viral therapy.7 This treatment was approved by the FDA in 2015 and replicates within tumors to produce granulocyte-macrophage colony-stimulating factor protein, which promotes an antitumor immune response within unresectable cutaneous, subcutaneous, and nodal melanoma lesions. Although it is not a gene-directed therapy, the melanoma does not require a specific mutation for treatment. Again, this medication is better served in conjunction with other melanoma chemotherapeutic and surgical interventions.
Submental Fat
Kybella (deoxycholic acid)(Allergan) is a nonhuman, nonanimal, synthetically created compound that is naturally found within the human body for the breakdown and absorption of dietary fat.8 This drug was FDA approved in 2015 for the improvement of the appearance of moderate subcutaneous fat under the chin. Patients are evaluated in clinic to determine if the submental fat would be responsive to an injectable or require more radical surgical intervention based on desired outcomes. The treatment is administered as 0.2-mL injections (up to a total of 10 mL) spaced 1-cm apart and ideally is repeated at regular intervals to evaluate for efficacy.
Basal Cell Carcinoma
Odomzo (sonidegib)(Novartis Corporation) was FDA approved in 2015 for locally advanced basal cell carcinoma.9 Odomzo is a smoothened antagonist that inhibits the hedgehog signaling pathway. Smoothened is a transmembrane protein that allows for signal transduction of hedgehog proteins.10 Protein patched homolog 1 binds to smoothened protein and prevents the signal transduction through the cell for Gli family zinc factor 1 to continue protein translation; however, when PTCH is mutated and can no longer bind to smoothened, tumor formation results, specifically basal cell carcinoma. Hence, sonidegib is for the treatment of basal cell carcinomas that have persisted despite radiation treatment and/or surgery as well as for patients who have multiple basal cell carcinomas that can no longer be treated with surgery or radiation.
Final Thoughts
Overall, although there are several medications that can be used in conjunction for treatment of dermatological conditions, it always is recommended to know what is in the pipeline as FDA-approved medications for dermatology.
CenterWatch (http://www.centerwatch.com/) is an online resource that provides directories, analysis, and market research of medications that are either under clinical evaluation or available for use in patients. A list of currently approved drugs by the US Food and Drug Administration (FDA) also is available by specialty. It is important for dermatologists in-training to know about recently approved drugs and those that are in the pipeline, as these treatments may benefit patients who are unresponsive to other previously used medications. New drugs also may be useful for physicians who have a difficult time getting insurance to cover prescriptions for their patients, as most new medications have built-in patient assistance.
New Drugs in Dermatology
Actinic Keratosis
Ameluz (aminolevulinic acid hydrochloride)(Biofrontera AG) is a new drug that was approved in May 2016 for treatment of mild to moderate actinic keratosis on the face and scalp.1 It is only intended for in-office use on patients who may not be candidates for other treatment options for actinic keratosis. The product is a gel formulation that should be applied to cover the lesions and approximately 5 mm of the surrounding area with a film of approximately 1-mm thickness. The entire treatment area is then illuminated with a red light source, either with a narrow spectrum around 630 nm with a light dose of approximately 37 J/cm2 or a broader and continuous spectrum in the range of 570 to 670 nm with a light dose between 75 and 200 J/cm2.1 Similar to the previously used aminolevulinic acid treatment method for actinic keratosis, the patient may experience a burning stinging sensation throughout the treatment and the skin will then proceed to peel.
Psoriasis and Psoriatic Arthritis
Taltz (ixekizumab)(Eli Lilly and Company) was approved by the FDA in March 2016 for the treatment of moderate to severe plaque psoriasis.2 It is a humanized IL-17A antagonist that works when IgG4 monoclonal antibodies selectively bind with IL-17A cytokines and inhibit their interaction with the IL-17 receptor. Although this injectable medication is approved for the treatment of psoriasis, it also can potentially be used off label for the treatment of psoriatic arthritis and rheumatoid arthritis. The approved dosage is 160 mg (two 80-mg injections) at week 0, followed by 80 mg at weeks 2, 4, 6, 8, 10, and 12, then 80 mg every 4 weeks.2 Injectable immunomodulatory medications such as ixekizumab are ideal for patients in whom topical treatments and light therapy failed and they continue to have serious psoriatic discomfort as well as for those who have substantial body surface area coverage.
In January 2015, Cosentyx (secukinumab)(Novartis Corporation) was approved by the FDA.3 Similar to ixekizumab, this injectable is an IgG1 monoclonal antibody that selectively binds to the IL-17A cytokine and inhibits its interaction with the IL-17 receptor. It is approved for the treatment of moderate to severe plaque psoriasis and psoriatic arthritis. The approved dosage for plaque psoriasis is 300 mg (two 150-mg subcutaneous injections) at weeks 0 through 4 followed by 300 mg every 4 weeks as needed until clearance.3 Similar to ixekizumab, secukinumab may be used for the treatment of recalcitrant psoriasis or psoriasis with substantial body surface area involvement.
Melanoma
Cotellic (cobimetinib)(Genentech USA, Inc) was FDA approved in November 2015.4 Cobimetinib is a reversible inhibitor of mitogen-activated protein kinase (MAPK)/extracellular signal regulated kinase 1. Mitogen-activated protein kinase MEK1 and MEK2 are regulators of the extracellular signal-related kinase pathway, which promotes cellular proliferation. This pathway is key, as melanomas that have a BRAF V600E and kinase mutation continue to proliferate due to the constitutive activation of MEK1 and MEK2, further promoting cellular proliferation. Cobimetinib is approved for the treatment of melanoma in patients with unresectable or metastatic melanoma with a BRAF V600E or V600K mutation, in conjunction with vemurafenib. Zelboraf (vemurafenib)(Genentech USA, Inc), another inhibitor of BRAF V600E, also is used for the treatment of unresectable melanomas and was initially approved in 2011.5
BRAF is a serine/threonine protein kinase. When unregulated, it results in the deregulation of cell proliferation. According to Ascierto et al,6 50% of melanomas have a BRAF mutation, with nearly 90% of them with a V600E mutation. Hence, since the advent of direct chemotherapeutic agents such as BRAF inhibitors, clinical trials have shown notable reduction in mortality and morbidity of melanoma patients with BRAF mutations.6
Imlygic (talimogene laherparepvec)(Amgen, Inc) is a modified oncolytic viral therapy.7 This treatment was approved by the FDA in 2015 and replicates within tumors to produce granulocyte-macrophage colony-stimulating factor protein, which promotes an antitumor immune response within unresectable cutaneous, subcutaneous, and nodal melanoma lesions. Although it is not a gene-directed therapy, the melanoma does not require a specific mutation for treatment. Again, this medication is better served in conjunction with other melanoma chemotherapeutic and surgical interventions.
Submental Fat
Kybella (deoxycholic acid)(Allergan) is a nonhuman, nonanimal, synthetically created compound that is naturally found within the human body for the breakdown and absorption of dietary fat.8 This drug was FDA approved in 2015 for the improvement of the appearance of moderate subcutaneous fat under the chin. Patients are evaluated in clinic to determine if the submental fat would be responsive to an injectable or require more radical surgical intervention based on desired outcomes. The treatment is administered as 0.2-mL injections (up to a total of 10 mL) spaced 1-cm apart and ideally is repeated at regular intervals to evaluate for efficacy.
Basal Cell Carcinoma
Odomzo (sonidegib)(Novartis Corporation) was FDA approved in 2015 for locally advanced basal cell carcinoma.9 Odomzo is a smoothened antagonist that inhibits the hedgehog signaling pathway. Smoothened is a transmembrane protein that allows for signal transduction of hedgehog proteins.10 Protein patched homolog 1 binds to smoothened protein and prevents the signal transduction through the cell for Gli family zinc factor 1 to continue protein translation; however, when PTCH is mutated and can no longer bind to smoothened, tumor formation results, specifically basal cell carcinoma. Hence, sonidegib is for the treatment of basal cell carcinomas that have persisted despite radiation treatment and/or surgery as well as for patients who have multiple basal cell carcinomas that can no longer be treated with surgery or radiation.
Final Thoughts
Overall, although there are several medications that can be used in conjunction for treatment of dermatological conditions, it always is recommended to know what is in the pipeline as FDA-approved medications for dermatology.
- Ameluz [package insert]. Leverkusen, Germany: Biofrontera Bioscience GmbH; 2016.
- Taltz [package insert]. Indianapolis, IN: Eli Lilly and Company; 2016.
- Cosentyx [package insert]. East Hanover, NJ: Novartis Corporation; 2015.
- Cotellic [package insert]. San Francisco, CA: Genentech, Inc; 2016.
- Zelboraf [package insert]. San Francisco, CA: Genentech, Inc; 2016.
- Ascierto PA, Kirkwood JM, Grob JJ, et al. The role of BRAF V600 mutation in melanoma. J Transl Med. 2012;10:85.
- Imlygic (talimogene laherparepvec). Thousand Oaks, CA: Amgen Inc; 2015.
- Kybella [package insert]. West Lake Village, CA: Kythera Biopharmaceuticals, Inc; 2015.
- Odomzo [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corporation; 2015.
- Villavicencio EH, Walterhouse DO, Iannaccone PM. The sonic hedgehog-patched-gli pathway in human development and disease. Am J Hum Genet. 2000;67:1047-1054.
- Ameluz [package insert]. Leverkusen, Germany: Biofrontera Bioscience GmbH; 2016.
- Taltz [package insert]. Indianapolis, IN: Eli Lilly and Company; 2016.
- Cosentyx [package insert]. East Hanover, NJ: Novartis Corporation; 2015.
- Cotellic [package insert]. San Francisco, CA: Genentech, Inc; 2016.
- Zelboraf [package insert]. San Francisco, CA: Genentech, Inc; 2016.
- Ascierto PA, Kirkwood JM, Grob JJ, et al. The role of BRAF V600 mutation in melanoma. J Transl Med. 2012;10:85.
- Imlygic (talimogene laherparepvec). Thousand Oaks, CA: Amgen Inc; 2015.
- Kybella [package insert]. West Lake Village, CA: Kythera Biopharmaceuticals, Inc; 2015.
- Odomzo [package insert]. East Hanover, NJ: Novartis Pharmaceuticals Corporation; 2015.
- Villavicencio EH, Walterhouse DO, Iannaccone PM. The sonic hedgehog-patched-gli pathway in human development and disease. Am J Hum Genet. 2000;67:1047-1054.
VIDEO: For facial resurfacing, revisit CO2 lasers
LAS VEGAS – Patients can enjoy positive results from facial resurfacing with fractionated lasers, but they don’t always yield the same benefits as the traditional CO2 laser, Christopher Zachary, M.D., said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
“A lot of us are going back to using traditional laser resurfacing” for the patients who need it, such as those with many wrinkles, crepey skin, and extensive sun damage, Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine, said in a video interview.
“Those patients are not going to have an optimal result, even with the most aggressive of fractionated ablative lasers, as compared to the traditional laser resurfacing,” he added.
Dr. Zachary disclosed relationships with multiple companies, including Solta, Zeltiq, Scion, Amway, and Candela. SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Patients can enjoy positive results from facial resurfacing with fractionated lasers, but they don’t always yield the same benefits as the traditional CO2 laser, Christopher Zachary, M.D., said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
“A lot of us are going back to using traditional laser resurfacing” for the patients who need it, such as those with many wrinkles, crepey skin, and extensive sun damage, Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine, said in a video interview.
“Those patients are not going to have an optimal result, even with the most aggressive of fractionated ablative lasers, as compared to the traditional laser resurfacing,” he added.
Dr. Zachary disclosed relationships with multiple companies, including Solta, Zeltiq, Scion, Amway, and Candela. SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Patients can enjoy positive results from facial resurfacing with fractionated lasers, but they don’t always yield the same benefits as the traditional CO2 laser, Christopher Zachary, M.D., said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
“A lot of us are going back to using traditional laser resurfacing” for the patients who need it, such as those with many wrinkles, crepey skin, and extensive sun damage, Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine, said in a video interview.
“Those patients are not going to have an optimal result, even with the most aggressive of fractionated ablative lasers, as compared to the traditional laser resurfacing,” he added.
Dr. Zachary disclosed relationships with multiple companies, including Solta, Zeltiq, Scion, Amway, and Candela. SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
VIDEO: Bulk matters in body sculpting
LAS VEGAS – Both heating and cooling techniques can provide effective results for patients seeking to improve their appearance with body sculpting, Christopher Zachary, MD, said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Whether the clinician chooses devices that use radiofrequency, laser, or cryolipolysis to target fat, the key is bulk treatment, Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine, said in a video interview.
When cooling or heating the fat, “it has to been done in bulk; it has to be done for a certain length of time,” he said, noting that treatment times vary with devices, from 5 to 60 minutes. “I can’t stress enough the importance of bulk cooling or bulk heating,” which induce a chronic reaction “that results in localized fat reduction,” he added.
Dr. Zachary disclosed relationships with multiple companies, including Solta, Zeltiq, Scion, Amway, and Candela. SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Both heating and cooling techniques can provide effective results for patients seeking to improve their appearance with body sculpting, Christopher Zachary, MD, said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Whether the clinician chooses devices that use radiofrequency, laser, or cryolipolysis to target fat, the key is bulk treatment, Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine, said in a video interview.
When cooling or heating the fat, “it has to been done in bulk; it has to be done for a certain length of time,” he said, noting that treatment times vary with devices, from 5 to 60 minutes. “I can’t stress enough the importance of bulk cooling or bulk heating,” which induce a chronic reaction “that results in localized fat reduction,” he added.
Dr. Zachary disclosed relationships with multiple companies, including Solta, Zeltiq, Scion, Amway, and Candela. SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Both heating and cooling techniques can provide effective results for patients seeking to improve their appearance with body sculpting, Christopher Zachary, MD, said at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
Whether the clinician chooses devices that use radiofrequency, laser, or cryolipolysis to target fat, the key is bulk treatment, Dr. Zachary, professor and chair of the department of dermatology at the University of California, Irvine, said in a video interview.
When cooling or heating the fat, “it has to been done in bulk; it has to be done for a certain length of time,” he said, noting that treatment times vary with devices, from 5 to 60 minutes. “I can’t stress enough the importance of bulk cooling or bulk heating,” which induce a chronic reaction “that results in localized fat reduction,” he added.
Dr. Zachary disclosed relationships with multiple companies, including Solta, Zeltiq, Scion, Amway, and Candela. SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SDEF LAS VEGAS DERMATOLOGY SEMINAR
Highlights from the ASDS Annual Conference 2016
At this year’s American Society for Dermatologic Surgery (ASDS) annual meeting, many of the hot topics pertained to tightening and smoothness of the skin. More naturally derived skin care products were discussed, as well as skin-tightening devices, techniques for improving fat, microneedling, devices for improving cellulite, and some of the newer injectable fillers on the market.
Of all of these, the use of microneedling with and without radiofrequency energy to improve acne scars, rhytids, and pore size was the most prominent emerging trend. Microneedling devices with radiofrequency also are being used for skin tightening, in addition to improving skin texture.
There were also presentations on improvements in acne scars with a number of techniques, including fractional ablative and nonablative lasers, subcision, microneedling, and fillers.
Controversies addressed at the meeting included whether or not dermatologists should use the same lasers with different Food and Drug Administration–approved hand pieces for vaginal rejuvenation. In addition, controversies over whether Mohs surgeons should undergo board examination were discussed.
Dr. Talakoub and Dr. Wesley are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
At this year’s American Society for Dermatologic Surgery (ASDS) annual meeting, many of the hot topics pertained to tightening and smoothness of the skin. More naturally derived skin care products were discussed, as well as skin-tightening devices, techniques for improving fat, microneedling, devices for improving cellulite, and some of the newer injectable fillers on the market.
Of all of these, the use of microneedling with and without radiofrequency energy to improve acne scars, rhytids, and pore size was the most prominent emerging trend. Microneedling devices with radiofrequency also are being used for skin tightening, in addition to improving skin texture.
There were also presentations on improvements in acne scars with a number of techniques, including fractional ablative and nonablative lasers, subcision, microneedling, and fillers.
Controversies addressed at the meeting included whether or not dermatologists should use the same lasers with different Food and Drug Administration–approved hand pieces for vaginal rejuvenation. In addition, controversies over whether Mohs surgeons should undergo board examination were discussed.
Dr. Talakoub and Dr. Wesley are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
At this year’s American Society for Dermatologic Surgery (ASDS) annual meeting, many of the hot topics pertained to tightening and smoothness of the skin. More naturally derived skin care products were discussed, as well as skin-tightening devices, techniques for improving fat, microneedling, devices for improving cellulite, and some of the newer injectable fillers on the market.
Of all of these, the use of microneedling with and without radiofrequency energy to improve acne scars, rhytids, and pore size was the most prominent emerging trend. Microneedling devices with radiofrequency also are being used for skin tightening, in addition to improving skin texture.
There were also presentations on improvements in acne scars with a number of techniques, including fractional ablative and nonablative lasers, subcision, microneedling, and fillers.
Controversies addressed at the meeting included whether or not dermatologists should use the same lasers with different Food and Drug Administration–approved hand pieces for vaginal rejuvenation. In addition, controversies over whether Mohs surgeons should undergo board examination were discussed.
Dr. Talakoub and Dr. Wesley are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Wesley. Write to them at [email protected].
Shea butter
Indigenous to Africa, Vitellaria paradoxa, better known as the shea or shi tree, is a member of the Sapotaceae family. It has long been used in traditional medicine in sub-Saharan West Africa (as far west as Mali) as well as parts of East Africa (as far east as Uganda and Ethiopia) for its anti-inflammatory and analgesic properties.1,2
Some indications in traditional Nigerian medicine include nasal congestion, scabies, and ulcers.2 In addition, anecdotal success in treating keloids has been reported in association with traditional African remedies, including shea butter and boa constrictor oil.3 Antioxidant activities have also been linked to V. paradoxa.4 Given such purported properties, it is not surprising that demand for shea kernels and butter has steadily increased in recent years for various purposes, including use as food (particularly as a cocoa butter additive in chocolate) and in medical and cosmetic products.2,4 The use of shea butter in skin care is attributed to its hydrating qualities and reputed effectiveness in softening scars.3
Constituents
Shea butter contains fatty acids that have been shown to improve the skin barrier. These include palmitic, stearic, and linoleic acid. It also contains the fatty acids oleic and arachidic. Shea butter also has phenolic components that function as antioxidants.
Anti-inflammatory effects
In 2010, Akihisa et al. evaluated the inhibitory effects of four triterpene acetates and four triterpene cinnamates isolated from the kernel fat of V. paradoxa against 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced inflammation in mice. All of the tested compounds showed considerable anti-inflammatory activity (ID50 values ranged from 0.15 to 0.75 micromol/ear). Lupeol cinnamate displayed the greatest anti-inflammatory activity, on carrageenan-induced edema on rat hind paws. All eight substances also exhibited moderate inhibitory effects on Epstein-Barr virus early antigen (EBV-EA) in Raji cells as a primary screening test for tumor promoter inhibitors. Using 7,12-dimethylbenz[a]anthracene (DMBA) as an initiator and TPA as a promoter in a two-stage carcinogenesis model in mice, the investigators also found that lupeol cinnamate inhibited skin tumor promotion. They concluded that the triterpenes and triterpene esters found in shea nuts and shea butter are significant anti-inflammatory and antitumor-promoting agents.2
The next year, Akihisa et al. determined the triacylglycerol and triterpene ester fraction composition of the kernel fats of the shea tree from 36 samples from Cote d’Ivoire, Ghana, Nigeria, Cameroon, Chad, Sudan, and Uganda. There were no significant differences in the composition of the triterpene ester fractions between West African and East African plants. Generally, though, West African shea kernel fats contained higher levels of high-melting triacylglycerols (e.g., stearic-oleic-stearic) and triterpene esters.6
Also that year, Olaitan et al. found that shea butter (as well as boa constrictor oil) was effective in suppressing the in vitro growth of normal and keloid fibroblasts.3
In 2012, Verma et al. used the lipopolysaccharide (LPS)-induced murine macrophage cell line J774 to investigate the anti-inflammatory properties of the methanolic extract of shea butter. They found that shea butter extract dose-dependently reduced, to a significant degree, the levels of nitric oxide, tumor necrosis factor (TNF)–alpha, as well as interleukin (IL)-1beta and IL-12 in the culture supernatants. In addition, the botanical extract suppressed IkappaB phosphorylation and NF-kappaB nuclear translocation as well as the expression of pro-inflammatory enzymes, inducible nitric oxide synthase (iNOS), and cyclooxygenase (COX)-2. The investigators attributed the anti-inflammatory activity of the extract to its inhibitory impact on LPS-induced iNOS, COX-2, TNF-alpha, IL-1beta, and IL-12 mRNA expression.1
In 2014, Honfo et al. conducted a literature review indicating that shea pulp is laden with vitamin C and the kernels contain copious fat (butter), which is used in food, drugs, and cosmetics.4
Notably, shea butter is also an ingredient in the topical nonsteroidal anti-inflammatory drug (NSAID) atopiclair, which has shown efficacy in alleviating pruritus in adults with mild to-moderate atopic dermatitis.7
Conclusion
Shea butter has long been incorporated into traditional medical practice in West and East Africa based on observed anti-inflammatory and analgesic characteristics. Such uses are compelling and often the basis for systematic scientific investigation. That said, there remains a dearth of experimental and clinical research on the potential cutaneous benefits of topically applied shea butter. Current data and traditional applications provide ample reason for continued research into this popular botanical agent.
References
1. J Complement Integr Med. 2012;9:Article 4.
2. J Oleo Sci. 2010;59[6]:273-80)
3. Wounds. 2011;23[4]:97-106.
4. Crit Rev Food Sci Nutr. 2014;54[5]:673-86.
5. J Agric Food Chem. 2003;51[21]:6268-73.
6. J Oleo Sci. 2011;60[8]:385-91.
7. J Drugs Dermatol. 2009 Jun;8[6]:537-9.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Indigenous to Africa, Vitellaria paradoxa, better known as the shea or shi tree, is a member of the Sapotaceae family. It has long been used in traditional medicine in sub-Saharan West Africa (as far west as Mali) as well as parts of East Africa (as far east as Uganda and Ethiopia) for its anti-inflammatory and analgesic properties.1,2
Some indications in traditional Nigerian medicine include nasal congestion, scabies, and ulcers.2 In addition, anecdotal success in treating keloids has been reported in association with traditional African remedies, including shea butter and boa constrictor oil.3 Antioxidant activities have also been linked to V. paradoxa.4 Given such purported properties, it is not surprising that demand for shea kernels and butter has steadily increased in recent years for various purposes, including use as food (particularly as a cocoa butter additive in chocolate) and in medical and cosmetic products.2,4 The use of shea butter in skin care is attributed to its hydrating qualities and reputed effectiveness in softening scars.3
Constituents
Shea butter contains fatty acids that have been shown to improve the skin barrier. These include palmitic, stearic, and linoleic acid. It also contains the fatty acids oleic and arachidic. Shea butter also has phenolic components that function as antioxidants.
Anti-inflammatory effects
In 2010, Akihisa et al. evaluated the inhibitory effects of four triterpene acetates and four triterpene cinnamates isolated from the kernel fat of V. paradoxa against 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced inflammation in mice. All of the tested compounds showed considerable anti-inflammatory activity (ID50 values ranged from 0.15 to 0.75 micromol/ear). Lupeol cinnamate displayed the greatest anti-inflammatory activity, on carrageenan-induced edema on rat hind paws. All eight substances also exhibited moderate inhibitory effects on Epstein-Barr virus early antigen (EBV-EA) in Raji cells as a primary screening test for tumor promoter inhibitors. Using 7,12-dimethylbenz[a]anthracene (DMBA) as an initiator and TPA as a promoter in a two-stage carcinogenesis model in mice, the investigators also found that lupeol cinnamate inhibited skin tumor promotion. They concluded that the triterpenes and triterpene esters found in shea nuts and shea butter are significant anti-inflammatory and antitumor-promoting agents.2
The next year, Akihisa et al. determined the triacylglycerol and triterpene ester fraction composition of the kernel fats of the shea tree from 36 samples from Cote d’Ivoire, Ghana, Nigeria, Cameroon, Chad, Sudan, and Uganda. There were no significant differences in the composition of the triterpene ester fractions between West African and East African plants. Generally, though, West African shea kernel fats contained higher levels of high-melting triacylglycerols (e.g., stearic-oleic-stearic) and triterpene esters.6
Also that year, Olaitan et al. found that shea butter (as well as boa constrictor oil) was effective in suppressing the in vitro growth of normal and keloid fibroblasts.3
In 2012, Verma et al. used the lipopolysaccharide (LPS)-induced murine macrophage cell line J774 to investigate the anti-inflammatory properties of the methanolic extract of shea butter. They found that shea butter extract dose-dependently reduced, to a significant degree, the levels of nitric oxide, tumor necrosis factor (TNF)–alpha, as well as interleukin (IL)-1beta and IL-12 in the culture supernatants. In addition, the botanical extract suppressed IkappaB phosphorylation and NF-kappaB nuclear translocation as well as the expression of pro-inflammatory enzymes, inducible nitric oxide synthase (iNOS), and cyclooxygenase (COX)-2. The investigators attributed the anti-inflammatory activity of the extract to its inhibitory impact on LPS-induced iNOS, COX-2, TNF-alpha, IL-1beta, and IL-12 mRNA expression.1
In 2014, Honfo et al. conducted a literature review indicating that shea pulp is laden with vitamin C and the kernels contain copious fat (butter), which is used in food, drugs, and cosmetics.4
Notably, shea butter is also an ingredient in the topical nonsteroidal anti-inflammatory drug (NSAID) atopiclair, which has shown efficacy in alleviating pruritus in adults with mild to-moderate atopic dermatitis.7
Conclusion
Shea butter has long been incorporated into traditional medical practice in West and East Africa based on observed anti-inflammatory and analgesic characteristics. Such uses are compelling and often the basis for systematic scientific investigation. That said, there remains a dearth of experimental and clinical research on the potential cutaneous benefits of topically applied shea butter. Current data and traditional applications provide ample reason for continued research into this popular botanical agent.
References
1. J Complement Integr Med. 2012;9:Article 4.
2. J Oleo Sci. 2010;59[6]:273-80)
3. Wounds. 2011;23[4]:97-106.
4. Crit Rev Food Sci Nutr. 2014;54[5]:673-86.
5. J Agric Food Chem. 2003;51[21]:6268-73.
6. J Oleo Sci. 2011;60[8]:385-91.
7. J Drugs Dermatol. 2009 Jun;8[6]:537-9.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Indigenous to Africa, Vitellaria paradoxa, better known as the shea or shi tree, is a member of the Sapotaceae family. It has long been used in traditional medicine in sub-Saharan West Africa (as far west as Mali) as well as parts of East Africa (as far east as Uganda and Ethiopia) for its anti-inflammatory and analgesic properties.1,2
Some indications in traditional Nigerian medicine include nasal congestion, scabies, and ulcers.2 In addition, anecdotal success in treating keloids has been reported in association with traditional African remedies, including shea butter and boa constrictor oil.3 Antioxidant activities have also been linked to V. paradoxa.4 Given such purported properties, it is not surprising that demand for shea kernels and butter has steadily increased in recent years for various purposes, including use as food (particularly as a cocoa butter additive in chocolate) and in medical and cosmetic products.2,4 The use of shea butter in skin care is attributed to its hydrating qualities and reputed effectiveness in softening scars.3
Constituents
Shea butter contains fatty acids that have been shown to improve the skin barrier. These include palmitic, stearic, and linoleic acid. It also contains the fatty acids oleic and arachidic. Shea butter also has phenolic components that function as antioxidants.
Anti-inflammatory effects
In 2010, Akihisa et al. evaluated the inhibitory effects of four triterpene acetates and four triterpene cinnamates isolated from the kernel fat of V. paradoxa against 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced inflammation in mice. All of the tested compounds showed considerable anti-inflammatory activity (ID50 values ranged from 0.15 to 0.75 micromol/ear). Lupeol cinnamate displayed the greatest anti-inflammatory activity, on carrageenan-induced edema on rat hind paws. All eight substances also exhibited moderate inhibitory effects on Epstein-Barr virus early antigen (EBV-EA) in Raji cells as a primary screening test for tumor promoter inhibitors. Using 7,12-dimethylbenz[a]anthracene (DMBA) as an initiator and TPA as a promoter in a two-stage carcinogenesis model in mice, the investigators also found that lupeol cinnamate inhibited skin tumor promotion. They concluded that the triterpenes and triterpene esters found in shea nuts and shea butter are significant anti-inflammatory and antitumor-promoting agents.2
The next year, Akihisa et al. determined the triacylglycerol and triterpene ester fraction composition of the kernel fats of the shea tree from 36 samples from Cote d’Ivoire, Ghana, Nigeria, Cameroon, Chad, Sudan, and Uganda. There were no significant differences in the composition of the triterpene ester fractions between West African and East African plants. Generally, though, West African shea kernel fats contained higher levels of high-melting triacylglycerols (e.g., stearic-oleic-stearic) and triterpene esters.6
Also that year, Olaitan et al. found that shea butter (as well as boa constrictor oil) was effective in suppressing the in vitro growth of normal and keloid fibroblasts.3
In 2012, Verma et al. used the lipopolysaccharide (LPS)-induced murine macrophage cell line J774 to investigate the anti-inflammatory properties of the methanolic extract of shea butter. They found that shea butter extract dose-dependently reduced, to a significant degree, the levels of nitric oxide, tumor necrosis factor (TNF)–alpha, as well as interleukin (IL)-1beta and IL-12 in the culture supernatants. In addition, the botanical extract suppressed IkappaB phosphorylation and NF-kappaB nuclear translocation as well as the expression of pro-inflammatory enzymes, inducible nitric oxide synthase (iNOS), and cyclooxygenase (COX)-2. The investigators attributed the anti-inflammatory activity of the extract to its inhibitory impact on LPS-induced iNOS, COX-2, TNF-alpha, IL-1beta, and IL-12 mRNA expression.1
In 2014, Honfo et al. conducted a literature review indicating that shea pulp is laden with vitamin C and the kernels contain copious fat (butter), which is used in food, drugs, and cosmetics.4
Notably, shea butter is also an ingredient in the topical nonsteroidal anti-inflammatory drug (NSAID) atopiclair, which has shown efficacy in alleviating pruritus in adults with mild to-moderate atopic dermatitis.7
Conclusion
Shea butter has long been incorporated into traditional medical practice in West and East Africa based on observed anti-inflammatory and analgesic characteristics. Such uses are compelling and often the basis for systematic scientific investigation. That said, there remains a dearth of experimental and clinical research on the potential cutaneous benefits of topically applied shea butter. Current data and traditional applications provide ample reason for continued research into this popular botanical agent.
References
1. J Complement Integr Med. 2012;9:Article 4.
2. J Oleo Sci. 2010;59[6]:273-80)
3. Wounds. 2011;23[4]:97-106.
4. Crit Rev Food Sci Nutr. 2014;54[5]:673-86.
5. J Agric Food Chem. 2003;51[21]:6268-73.
6. J Oleo Sci. 2011;60[8]:385-91.
7. J Drugs Dermatol. 2009 Jun;8[6]:537-9.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Acne and Antiaging: Is There a Connection?
As a chronic inflammatory skin disease well known for its poor cosmesis including scarring, residual macular erythema, and postinflammatory pigment alteration, acne vulgaris may, according to recent research, confer some antiaging benefits to affected patients. In a research letter published online on September 27 in the Journal of Investigative Dermatology, Ribero et al analyzed white blood cells and found that women who said they had acne had longer telomeres (the "caps" at the end of chromosomes that protect them from deteriorating following repeated cell replication). Telomere length, or rather shortening, has been correlated with age-related degenerative change, according to Saum et al (Exp Gerontol. 2014;58:250-255), and therefore the thinking is that in women with acne, something is going on that maintains the length of the cellular guardians. Let's clarify a couple things to help us all understand the why and what.
The impetus of this study, according to Ribero et al, was the observation that women with acne show signs of aging later than those who have never had acne. I personally have not witnessed this finding in my patients, and given that acne in its essence is a disease of chronic inflammation resulting from, for example, persistent activation of toll-like receptor 2 (TLR2) and NOD-like receptor family pyrin domain containing 3 (NLRP3) pattern recognition receptors, one would think the skin damage accrued would make these individuals look older, right? Last I checked, pitted scarring does not make one immediately think of the fountain of youth.
The results from the study show that there is a link between acne and longer telomeres, but the study did not show that telomere length is a cause of acne, that women with longer telomeres had fewer signs of skin aging, or that women with acne lived longer.
Given these points, Ribero et al concluded that "delayed skin aging may be due to reduced senescence," which means that skin aging may be delayed because the longer telomeres in the cells protect them from deterioration. They did find that the expression of one gene in particular was reduced in women with acne--the regulatory gene zinc finger protein 420, ZNF420--suggesting that those without acne may produce more of a particular protein linked to that gene, though the significance is unclear.
What's the issue?
This study is interesting, but it is important not to make any broad conclusions, such as those who get acne will live longer or look younger longer regardless of other factors such as acne treatment, comorbidities, or even environmental factors. This study may give more support for the genetic contribution of acne, but much more work is needed to determine the clinical relevance. For starters, what about men?
Would you assure your acne patients that their disease may be for their own cosmetic good?
As a chronic inflammatory skin disease well known for its poor cosmesis including scarring, residual macular erythema, and postinflammatory pigment alteration, acne vulgaris may, according to recent research, confer some antiaging benefits to affected patients. In a research letter published online on September 27 in the Journal of Investigative Dermatology, Ribero et al analyzed white blood cells and found that women who said they had acne had longer telomeres (the "caps" at the end of chromosomes that protect them from deteriorating following repeated cell replication). Telomere length, or rather shortening, has been correlated with age-related degenerative change, according to Saum et al (Exp Gerontol. 2014;58:250-255), and therefore the thinking is that in women with acne, something is going on that maintains the length of the cellular guardians. Let's clarify a couple things to help us all understand the why and what.
The impetus of this study, according to Ribero et al, was the observation that women with acne show signs of aging later than those who have never had acne. I personally have not witnessed this finding in my patients, and given that acne in its essence is a disease of chronic inflammation resulting from, for example, persistent activation of toll-like receptor 2 (TLR2) and NOD-like receptor family pyrin domain containing 3 (NLRP3) pattern recognition receptors, one would think the skin damage accrued would make these individuals look older, right? Last I checked, pitted scarring does not make one immediately think of the fountain of youth.
The results from the study show that there is a link between acne and longer telomeres, but the study did not show that telomere length is a cause of acne, that women with longer telomeres had fewer signs of skin aging, or that women with acne lived longer.
Given these points, Ribero et al concluded that "delayed skin aging may be due to reduced senescence," which means that skin aging may be delayed because the longer telomeres in the cells protect them from deterioration. They did find that the expression of one gene in particular was reduced in women with acne--the regulatory gene zinc finger protein 420, ZNF420--suggesting that those without acne may produce more of a particular protein linked to that gene, though the significance is unclear.
What's the issue?
This study is interesting, but it is important not to make any broad conclusions, such as those who get acne will live longer or look younger longer regardless of other factors such as acne treatment, comorbidities, or even environmental factors. This study may give more support for the genetic contribution of acne, but much more work is needed to determine the clinical relevance. For starters, what about men?
Would you assure your acne patients that their disease may be for their own cosmetic good?
As a chronic inflammatory skin disease well known for its poor cosmesis including scarring, residual macular erythema, and postinflammatory pigment alteration, acne vulgaris may, according to recent research, confer some antiaging benefits to affected patients. In a research letter published online on September 27 in the Journal of Investigative Dermatology, Ribero et al analyzed white blood cells and found that women who said they had acne had longer telomeres (the "caps" at the end of chromosomes that protect them from deteriorating following repeated cell replication). Telomere length, or rather shortening, has been correlated with age-related degenerative change, according to Saum et al (Exp Gerontol. 2014;58:250-255), and therefore the thinking is that in women with acne, something is going on that maintains the length of the cellular guardians. Let's clarify a couple things to help us all understand the why and what.
The impetus of this study, according to Ribero et al, was the observation that women with acne show signs of aging later than those who have never had acne. I personally have not witnessed this finding in my patients, and given that acne in its essence is a disease of chronic inflammation resulting from, for example, persistent activation of toll-like receptor 2 (TLR2) and NOD-like receptor family pyrin domain containing 3 (NLRP3) pattern recognition receptors, one would think the skin damage accrued would make these individuals look older, right? Last I checked, pitted scarring does not make one immediately think of the fountain of youth.
The results from the study show that there is a link between acne and longer telomeres, but the study did not show that telomere length is a cause of acne, that women with longer telomeres had fewer signs of skin aging, or that women with acne lived longer.
Given these points, Ribero et al concluded that "delayed skin aging may be due to reduced senescence," which means that skin aging may be delayed because the longer telomeres in the cells protect them from deterioration. They did find that the expression of one gene in particular was reduced in women with acne--the regulatory gene zinc finger protein 420, ZNF420--suggesting that those without acne may produce more of a particular protein linked to that gene, though the significance is unclear.
What's the issue?
This study is interesting, but it is important not to make any broad conclusions, such as those who get acne will live longer or look younger longer regardless of other factors such as acne treatment, comorbidities, or even environmental factors. This study may give more support for the genetic contribution of acne, but much more work is needed to determine the clinical relevance. For starters, what about men?
Would you assure your acne patients that their disease may be for their own cosmetic good?
Cosmetic Corner: Dermatologists Weigh in on OTC Rosacea Treatments
To improve patient care and outcomes, leading dermatologists offered their recommendations on OTC rosacea treatments. Consideration must be given to:
- Eucerin Redness Relief
Beiersdorf Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Rosaliac CC Cream
La Roche-Posay Laboratoire Dermatologique
“This product provides hydration and an even tint to correct and reduce the erythema of rosacea. It is made with both titanium dioxide and organic UV filters to give broad-spectrum coverage with SPF 30.”—Cherise Mizrahi-Levi, DO, New York, New York
- Vanicream
Pharmaceutical Specialties, Inc.
“I like Vanicream products since they are mild and hypoallergenic.”—Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, skin-lightening agents, and self-tanners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on OTC rosacea treatments. Consideration must be given to:
- Eucerin Redness Relief
Beiersdorf Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Rosaliac CC Cream
La Roche-Posay Laboratoire Dermatologique
“This product provides hydration and an even tint to correct and reduce the erythema of rosacea. It is made with both titanium dioxide and organic UV filters to give broad-spectrum coverage with SPF 30.”—Cherise Mizrahi-Levi, DO, New York, New York
- Vanicream
Pharmaceutical Specialties, Inc.
“I like Vanicream products since they are mild and hypoallergenic.”—Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, skin-lightening agents, and self-tanners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on OTC rosacea treatments. Consideration must be given to:
- Eucerin Redness Relief
Beiersdorf Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Rosaliac CC Cream
La Roche-Posay Laboratoire Dermatologique
“This product provides hydration and an even tint to correct and reduce the erythema of rosacea. It is made with both titanium dioxide and organic UV filters to give broad-spectrum coverage with SPF 30.”—Cherise Mizrahi-Levi, DO, New York, New York
- Vanicream
Pharmaceutical Specialties, Inc.
“I like Vanicream products since they are mild and hypoallergenic.”—Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, skin-lightening agents, and self-tanners will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Efficacy and Safety of New Dermal Fillers
Facial aging is the result of the interplay between loss of skin elasticity, changes in subcutaneous fat and other soft-tissue layers, and skeletal remodeling with chronological age.1 Dermal fillers are effective for the treatment of rhytides, facial scars, and lipoatrophy, as well as facial contouring and augmentation. Given that multiple filler options exist, updated reviews are necessary to inform clinicians of the choices that are available. We provide a detailed review of the clinical efficacy and safety of the dermal fillers with the most recent approvals by the US Food and Drug Administration (FDA).
Polymethylmethacrylate
Polymethylmethacrylate (PMMA) microspheres suspended in bovine collagen and lidocaine 0.3% were approved in 2006 for use in nasolabial folds (NLFs) and in 2014 for acne scars. Now branded as Bellafill (Suneva Medical, Inc), it is the only permanent injectable filler currently available. Once injected, the particles are not reabsorbed and can only be removed by procedural extraction (eg, liposuction of the surrounding fat); however, the permanence of PMMA does not extend to facial rejuvenation, which can last up to 5 years. Prior to use, skin testing for bovine collagen reaction is necessary. In a clinical trial of 147 patients with moderate to severe acne scarring, patients were randomized to receive PMMA in collagen (n=97) or saline (n=50).2 Injections were administered using a linear threading or serial puncture technique, and patients were reevaluated after 4 weeks for touch-up injections. After 6 months, 64% of patients treated with PMMA in collagen achieved improvement in acne scars by 2 points or more on the acne scar rating scale versus 33% of the control group (P=.0005).2
Treatment-related adverse events (AEs) include injection-site pain, bruising, swelling, erythema, and more rarely pruritus and lumps/granulomas.3 A 5-year longitudinal safety investigation of 871 patients initially treated with PMMA in collagen for NLF correction revealed that 17 patients (2.0%) had biopsy-confirmed granulomas with half of these retained at study end.4 Fifteen of these patients were treated with intralesional corticosteroids alone or in combination with intralesional 5-fluorouracil, oral antibiotics, or topical calcineurin inhibitors; 1 patient was untreated and another used topical corticosteroids. The authors noted no correlation between treatment method and granuloma response.4 Polymethylmethacrylate in collagen is contraindicated in patients with lidocaine or bovine collagen sensitivity and is not indicated for use in lip augmentation due to high rates of nodule formation.3
Hyaluronic Acid
Hyaluronic acid (HA) is a naturally occurring glycosaminoglycan polymer found in the extracellular matrix of the dermis. Hyaluronic acid fillers are bacteria derived and come in gel form. A useful advantage of HA fillers compared to other dermal fillers is the commercial availability of hyaluronidase to correct injections. Preinjection skin testing is not necessary.5
This category of nonpermanent dermal fillers has the most robust market choices. Older HA dermal fillers with reliable and proven efficacy are Restylane (Galderma Laboratories, LP)(facial rhytides, lip augmentation), Juvéderm (Ultra/Ultra XC/Ultra Plus/Ultra Plus XC [Allergan, Inc])(facial rhytides, lip augmentation), Hydrelle (Anika Therapeutics, Inc)(facial rhytides), and Prevelle Silk (Mentor Corporation)(facial rhytides); they will not be reviewed here. Newer agents include Belotero Balance (Merz Aesthetics), Juvéderm Voluma XC (Allergan, Inc), Restylane Silk (Galderma Laboratories, LP), and Restylane Lyft (Galderma Laboratories, LP).
Belotero Balance
Belotero Balance is used to treat fine lines and wrinkles, especially NLFs.6 The initial pivotal studies that led to FDA approval in 2011 demonstrated noninferiority and superiority to bovine collagen for use in the treatment of NLFs.7,8 One hundred eighteen patients with bilateral NLFs that were rated as 2 (moderate) or 3 (severe) on the wrinkle severity rating scale (WSRS) were randomized to split-face injection of Belotero Balance in one NLF and bovine collagen in the contralateral NLF.7 An additional injection at week 2 was allowed for optimal correction. Belotero Balance was noninferior to bovine collagen at week 2, with mean improvement in WSRS of 1.52 versus 1.57 (P=.50). Belotero Balance was superior to bovine collagen in mean WSRS improvement at weeks 12 (1.25 vs 0.26; P<.001), 16 (1.09 vs 0.66; P<.001), and 24 (1.08 vs 0.50; P<.001).7 In a subsequent open-label extension study, which included 95 of 118 patients who received Belotero Balance injections in both NLFs at week 24, 80.2% of patients showed sustained improvement in WSRS from baseline for 48 weeks without further injection.8
The first comparative study of Belotero Balance with other established HA fillers at the time—Restylane and Juvéderm Ultra 3/Ultra Plus XC—to treat NLFs demonstrated noninferiority.9 Forty patients with bilateral, moderate to severe NLFs (rated 3 or 4 on the Merz severity scale) were randomized to split-face groups of Belotero Balance versus Restylane or Belotero Balance versus Juvéderm. At 12 months, NLF severity improved from 2.3 to 1.5 in the Restylane group and from 2.3 to 1.6 in the Juvéderm group.9
Belotero Balance has been compared to Juvéderm Ultra XC for use in perioral lines.10 The study included 136 patients with moderate to severe perioral lines, according to the perioral lines severity scale, who were randomized (1:1 ratio) to receive injections of Belotero Balance or Juvéderm Ultra XC to correct upper and lower perioral lines, with assessment at week 2 for optimization. After 6 months, 87% of Juvéderm-treated patients compared to 72% of Belotero Balance–treated patients had 1-point improvement in perioral lines (P<.04). Juvéderm-treated patients also reported significantly less pain than Belotero Balance–treated patients (P<.001).10
Treatment-related AEs are described in the Table, with the majority occurring at lower rates compared to a collagen control group and self-resolving within 2 weeks.7
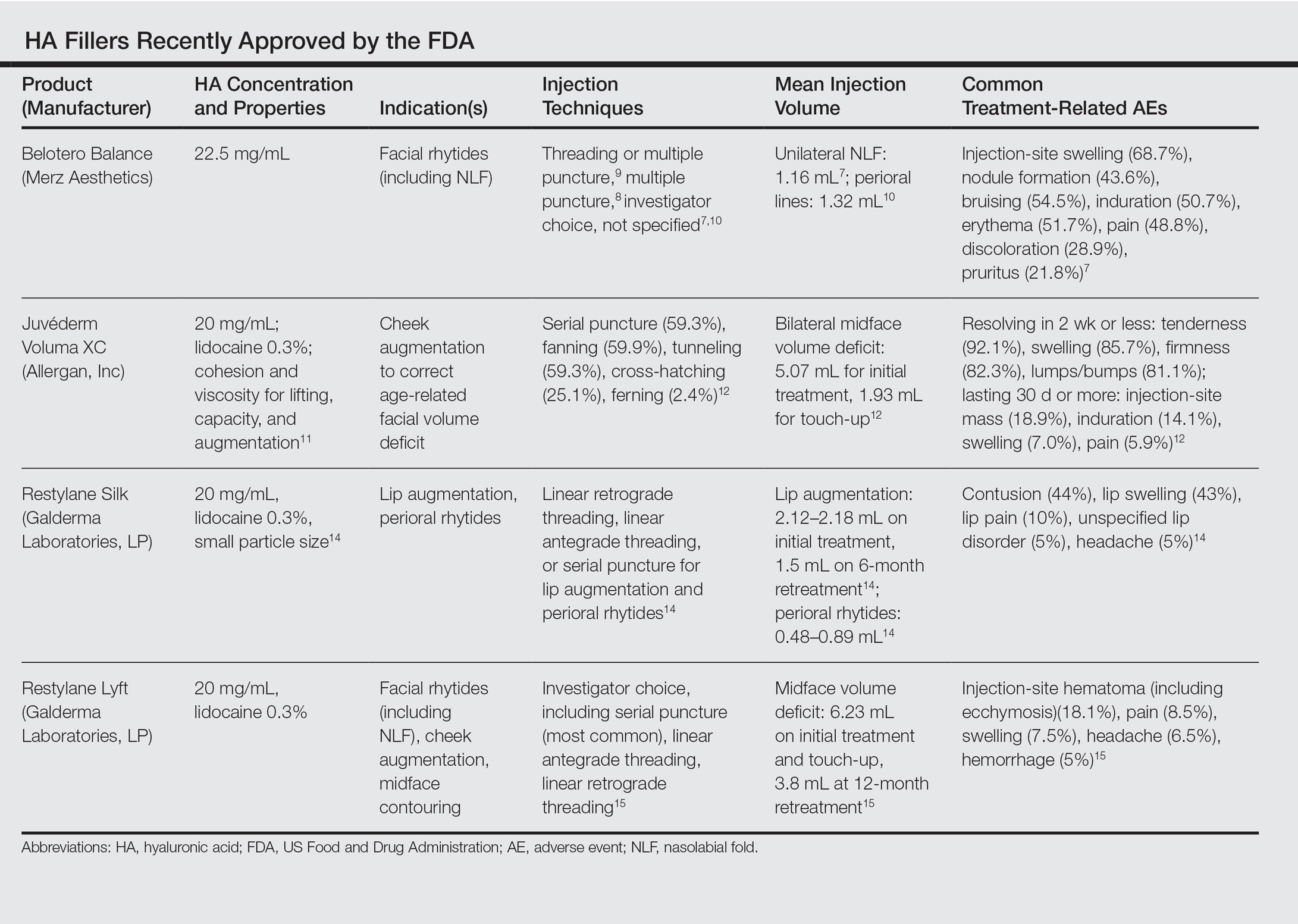
Juvéderm Voluma XC
Juvéderm Voluma XC was FDA approved in 2013 for cheek augmentation to correct age-related volume deficit restoration by subcutaneous or subperiosteal injections. In its landmark multicenter investigation, 282 patients with moderate to severe midface (eg, zygomaticomalar, anteromedial cheek, submalar regions) volume deficit measured on a validated midface volume deficit scale (MFVDS) were treated with Juvéderm Voluma XC (n=235) or control (n=47).11 Patients were reevaluated at 30 days and 81.9% received touch-up injections. At a 6-month primary evaluation, 86% of the Juvéderm-treated patients versus 39% of the control patients showed 1-point improvement on the MFVDS (P<.001). At 24-months’ follow-up, 44.6% of patients sustained efficacy.11 Of these aforementioned patients, 167 received repeat treatment due to lost correction or patient request and 91.1% improved by 1 point or more on the MFVDS on evaluation 12 months after repeat treatment.12 For this same population of patients, a 2-year extended follow-up of patient-reported outcomes revealed that 49% of patients felt fulfilled in their treatment goals 2 years after treatment and 79% of patients rated improvement from baseline based on the global aesthetic improvement scale.13 Efficacy studies involving Juvéderm Voluma XC are currently ongoing for facial temporal aging (registered at www.clinicaltrials.gov with the identifier NCT02437903) and recruiting for mandibular hypoplasia (NCT02330016).
Common treatment-related AEs are detailed in the Table. Two patients required treatment with hyaluronidase for chronic lumpiness and nodularity following non–treatment-related cellulitis.11 The product is contraindicated in patients with allergy to lidocaine.
Restylane Silk
Restylane Silk was approved in 2014 for lip augmentation and perioral rhytides. Efficacy and safety was demonstrated in a large multicenter randomized investigation in which 221 patients seeking lip augmentation received either Restylane Silk (n=177) injected submucosally for treatment of the upper and lower lips and/or intradermally for perioral rhytides or no treatment (n=44).14 Restylane treatment group patients optionally received touch-up at 2 weeks for optimization. All patients, including the control group, received injections at 6 months. At the 2-month primary end point, 80.2% of the treatment group exhibited at least 1-point improvement in upper lip fullness on the Medicis lip fullness scale compared to 11.9% (P<.001) of the control group; response rates for the lower lips were 84.2% versus 18.4% (P<.001). Patients in the treatment group receiving injections for perioral rhytides showed significant improvement in perioral rhytides through week 24 compared to patients treated for lip augmentation only (P<.001).14 Restylane Silk currently is undergoing investigation for cheek rejuvenation (NCT02636894, NCT02679924) and treatment of hand photoaging (NCT02780258).
The most common AEs are listed in the Table. No lip disorders were considered clinically concerning on evaluation. Concomitant lip augmentation and treatment of perioral rhytides yielded similar rates of AEs.14 Restylane Silk is not to be used in patients with known lidocaine allergy.
Restylane Lyft
Restylane Lyft (formerly known as Perlane-L) was approved in 2010 for use in facial rhytides, including NLFs, and gained approval in 2015 for use in cheek augmentation and midface contouring. Only its efficacy and safety for the more recent indication will be reviewed here.
In an evaluator-blinded investigation of 200 patients with mild to substantial bilateral midface deficiency based on the Medicis midface volume scale (MMVS), patients were randomized to receive supraperiosteal and subcutaneous treatment with Restylane Lyft (n=150) or no treatment (n=50).15 Touch-up injections at week 2 or month 12 were available to treatment group patients and all patients were given either an initial treatment or retreatment at 12 months. Primary end point evaluation at week 8 showed that 89% of treatment group patients had at least 1 grade MMVS improvement compared to 16% of the control group (P<.001). Although the percentage of these MMVS responders in the treatment group decreased with each follow-up period to 54.3% at month 12, retreatment was effective in reproducing a similar MMVS response rate as with initial treatment.15 Restylane Lyft is under ongoing investigation for dorsal hand rejuvenation (NCT02650921).
In addition to the common treatment-related AEs listed in the Table, 2 patients reported serious AEs, including bilateral implant-site inflammation and unilateral implant-site hematoma and infection (organism not described), all of which resolved with unspecified treatment.15 Lidocaine allergies are contraindications for use.
Conclusion
Several new options in dermal fillers have been approved in recent years and have demonstrated efficacy and acceptable safety in various cosmetic rejuvenation applications. Restylane Silk and Restylane Lyft are undergoing further studies to evaluate use in hand rejuvenation, an area that currently has few cosmetic filler treatment options. As technology continues to progress and new formulations of dermal fillers with varied properties and benefits are available, clinicians should expect multiple options for use in rhytides, volume deficits, and contouring.
ADDENDUM
After the manuscript was accepted for publication, Juvéderm Volbella XC (Allergan, Inc) was approved by the FDA for use in lip augmentation and thus is not included in this review.
- Fitzgerald R, Graivier MH, Kane M, et al. Update on facial aging. Aesthet Surg J. 2010;30(suppl):S11-S24.
- Karnik J, Baumann L, Bruce S, et al. A double-blind, randomized, multicenter, controlled trial of suspended polymethylmethacrylate microspheres for the correction of atrophic facial acne scars. J Am Acad Dermatol. 2014;71:77-83.
- Bellafill [package insert]. San Diego, CA: Suneva Medical, Inc; 2015.
- Cohen S, Dover J, Monheit G, et al. Five-year safety and satisfaction study of PMMA-collagen in the correction of nasolabial folds. Dermatol Surg. 2015;41(suppl 1):S302-S313.
- Greene JJ, Sidle DM. The hyaluronic acid fillers: current understanding of the tissue device interface. Facial Plast Surg Clin North Am. 2015;23:423-432.
- Lorenc ZP, Fagien S, Flynn TC, et al. Review of key Belotero Balance safety and efficacy trials. Plast Reconstr Surg. 2013;132(4, suppl 2):33S-40S.
- Narins RS, Coleman W, Donofrio L, et al. Nonanimal sourced hyaluronic acid–based dermal filler using a cohesive polydensified matrix technology is superior to bovine collagen in the correction of moderate to severe nasolabial folds: results from a 6-month, randomized, blinded, controlled, multicenter study. Dermatol Surg. 2010;36(suppl 1):730-740.
- Narins RS, Coleman WP 3rd, Donofrio LM, et al. Improvement in nasolabial folds with a hyaluronic acid filler using a cohesive polydensified matrix technology: results from an 18-month open-label extension trial. Dermatol Surg. 2010;36(suppl 3):1800-1808.
- Prager W, Wissmueller E, Havermann I, et al. A prospective, split-face, randomized, comparative study of safety and 12-month longevity of three formulations of hyaluronic acid dermal filler for treatment of nasolabial folds. Dermatol Surg. 2012;38(7, pt 2):1143-1150.
- Butterwick K, Marmur E, Narurkar V, et al. HYC-24L demonstrates greater effectiveness with less pain than CPM-22.5 for treatment of perioral lines in a randomized controlled trial. Dermatol Surg. 2015;41:1351-1360.
- Jones D, Murphy DK. Volumizing hyaluronic acid filler for midface volume deficit: 2-year results from a pivotal single-blind randomized controlled study. Dermatol Surg. 2013;39:1602-1612.
- Baumann L, Narins RS, Beer K, et al. Volumizing hyaluronic acid filler for midface volume deficit: results after repeat treatment. Dermatol Surg. 2015;41(suppl 1):S284-S292.
- Few J, Cox SE, Paradkar-Mitragotri D, et al. A multicenter, single-blind randomized, controlled study of a volumizing hyaluronic acid filler for midface volume deficit: patient-reported outcomes at 2 years. Aesthet Surg J. 2015;35:589-599.
- Beer K, Glogau RG, Dover JS, et al. A randomized, evaluator-blinded, controlled study of effectiveness and safety of small particle hyaluronic acid plus lidocaine for lip augmentation and perioral rhytides. Dermatol Surg. 2015;41(suppl 1):S127-S136.
- Weiss RA, Moradi A, Bank D, et al. Effectiveness and safety of large gel particle hyaluronic acid with lidocaine for correction of midface volume deficit or contour deficiency. Dermatol Surg. 2016;42:699-709.
Facial aging is the result of the interplay between loss of skin elasticity, changes in subcutaneous fat and other soft-tissue layers, and skeletal remodeling with chronological age.1 Dermal fillers are effective for the treatment of rhytides, facial scars, and lipoatrophy, as well as facial contouring and augmentation. Given that multiple filler options exist, updated reviews are necessary to inform clinicians of the choices that are available. We provide a detailed review of the clinical efficacy and safety of the dermal fillers with the most recent approvals by the US Food and Drug Administration (FDA).
Polymethylmethacrylate
Polymethylmethacrylate (PMMA) microspheres suspended in bovine collagen and lidocaine 0.3% were approved in 2006 for use in nasolabial folds (NLFs) and in 2014 for acne scars. Now branded as Bellafill (Suneva Medical, Inc), it is the only permanent injectable filler currently available. Once injected, the particles are not reabsorbed and can only be removed by procedural extraction (eg, liposuction of the surrounding fat); however, the permanence of PMMA does not extend to facial rejuvenation, which can last up to 5 years. Prior to use, skin testing for bovine collagen reaction is necessary. In a clinical trial of 147 patients with moderate to severe acne scarring, patients were randomized to receive PMMA in collagen (n=97) or saline (n=50).2 Injections were administered using a linear threading or serial puncture technique, and patients were reevaluated after 4 weeks for touch-up injections. After 6 months, 64% of patients treated with PMMA in collagen achieved improvement in acne scars by 2 points or more on the acne scar rating scale versus 33% of the control group (P=.0005).2
Treatment-related adverse events (AEs) include injection-site pain, bruising, swelling, erythema, and more rarely pruritus and lumps/granulomas.3 A 5-year longitudinal safety investigation of 871 patients initially treated with PMMA in collagen for NLF correction revealed that 17 patients (2.0%) had biopsy-confirmed granulomas with half of these retained at study end.4 Fifteen of these patients were treated with intralesional corticosteroids alone or in combination with intralesional 5-fluorouracil, oral antibiotics, or topical calcineurin inhibitors; 1 patient was untreated and another used topical corticosteroids. The authors noted no correlation between treatment method and granuloma response.4 Polymethylmethacrylate in collagen is contraindicated in patients with lidocaine or bovine collagen sensitivity and is not indicated for use in lip augmentation due to high rates of nodule formation.3
Hyaluronic Acid
Hyaluronic acid (HA) is a naturally occurring glycosaminoglycan polymer found in the extracellular matrix of the dermis. Hyaluronic acid fillers are bacteria derived and come in gel form. A useful advantage of HA fillers compared to other dermal fillers is the commercial availability of hyaluronidase to correct injections. Preinjection skin testing is not necessary.5
This category of nonpermanent dermal fillers has the most robust market choices. Older HA dermal fillers with reliable and proven efficacy are Restylane (Galderma Laboratories, LP)(facial rhytides, lip augmentation), Juvéderm (Ultra/Ultra XC/Ultra Plus/Ultra Plus XC [Allergan, Inc])(facial rhytides, lip augmentation), Hydrelle (Anika Therapeutics, Inc)(facial rhytides), and Prevelle Silk (Mentor Corporation)(facial rhytides); they will not be reviewed here. Newer agents include Belotero Balance (Merz Aesthetics), Juvéderm Voluma XC (Allergan, Inc), Restylane Silk (Galderma Laboratories, LP), and Restylane Lyft (Galderma Laboratories, LP).
Belotero Balance
Belotero Balance is used to treat fine lines and wrinkles, especially NLFs.6 The initial pivotal studies that led to FDA approval in 2011 demonstrated noninferiority and superiority to bovine collagen for use in the treatment of NLFs.7,8 One hundred eighteen patients with bilateral NLFs that were rated as 2 (moderate) or 3 (severe) on the wrinkle severity rating scale (WSRS) were randomized to split-face injection of Belotero Balance in one NLF and bovine collagen in the contralateral NLF.7 An additional injection at week 2 was allowed for optimal correction. Belotero Balance was noninferior to bovine collagen at week 2, with mean improvement in WSRS of 1.52 versus 1.57 (P=.50). Belotero Balance was superior to bovine collagen in mean WSRS improvement at weeks 12 (1.25 vs 0.26; P<.001), 16 (1.09 vs 0.66; P<.001), and 24 (1.08 vs 0.50; P<.001).7 In a subsequent open-label extension study, which included 95 of 118 patients who received Belotero Balance injections in both NLFs at week 24, 80.2% of patients showed sustained improvement in WSRS from baseline for 48 weeks without further injection.8
The first comparative study of Belotero Balance with other established HA fillers at the time—Restylane and Juvéderm Ultra 3/Ultra Plus XC—to treat NLFs demonstrated noninferiority.9 Forty patients with bilateral, moderate to severe NLFs (rated 3 or 4 on the Merz severity scale) were randomized to split-face groups of Belotero Balance versus Restylane or Belotero Balance versus Juvéderm. At 12 months, NLF severity improved from 2.3 to 1.5 in the Restylane group and from 2.3 to 1.6 in the Juvéderm group.9
Belotero Balance has been compared to Juvéderm Ultra XC for use in perioral lines.10 The study included 136 patients with moderate to severe perioral lines, according to the perioral lines severity scale, who were randomized (1:1 ratio) to receive injections of Belotero Balance or Juvéderm Ultra XC to correct upper and lower perioral lines, with assessment at week 2 for optimization. After 6 months, 87% of Juvéderm-treated patients compared to 72% of Belotero Balance–treated patients had 1-point improvement in perioral lines (P<.04). Juvéderm-treated patients also reported significantly less pain than Belotero Balance–treated patients (P<.001).10
Treatment-related AEs are described in the Table, with the majority occurring at lower rates compared to a collagen control group and self-resolving within 2 weeks.7

Juvéderm Voluma XC
Juvéderm Voluma XC was FDA approved in 2013 for cheek augmentation to correct age-related volume deficit restoration by subcutaneous or subperiosteal injections. In its landmark multicenter investigation, 282 patients with moderate to severe midface (eg, zygomaticomalar, anteromedial cheek, submalar regions) volume deficit measured on a validated midface volume deficit scale (MFVDS) were treated with Juvéderm Voluma XC (n=235) or control (n=47).11 Patients were reevaluated at 30 days and 81.9% received touch-up injections. At a 6-month primary evaluation, 86% of the Juvéderm-treated patients versus 39% of the control patients showed 1-point improvement on the MFVDS (P<.001). At 24-months’ follow-up, 44.6% of patients sustained efficacy.11 Of these aforementioned patients, 167 received repeat treatment due to lost correction or patient request and 91.1% improved by 1 point or more on the MFVDS on evaluation 12 months after repeat treatment.12 For this same population of patients, a 2-year extended follow-up of patient-reported outcomes revealed that 49% of patients felt fulfilled in their treatment goals 2 years after treatment and 79% of patients rated improvement from baseline based on the global aesthetic improvement scale.13 Efficacy studies involving Juvéderm Voluma XC are currently ongoing for facial temporal aging (registered at www.clinicaltrials.gov with the identifier NCT02437903) and recruiting for mandibular hypoplasia (NCT02330016).
Common treatment-related AEs are detailed in the Table. Two patients required treatment with hyaluronidase for chronic lumpiness and nodularity following non–treatment-related cellulitis.11 The product is contraindicated in patients with allergy to lidocaine.
Restylane Silk
Restylane Silk was approved in 2014 for lip augmentation and perioral rhytides. Efficacy and safety was demonstrated in a large multicenter randomized investigation in which 221 patients seeking lip augmentation received either Restylane Silk (n=177) injected submucosally for treatment of the upper and lower lips and/or intradermally for perioral rhytides or no treatment (n=44).14 Restylane treatment group patients optionally received touch-up at 2 weeks for optimization. All patients, including the control group, received injections at 6 months. At the 2-month primary end point, 80.2% of the treatment group exhibited at least 1-point improvement in upper lip fullness on the Medicis lip fullness scale compared to 11.9% (P<.001) of the control group; response rates for the lower lips were 84.2% versus 18.4% (P<.001). Patients in the treatment group receiving injections for perioral rhytides showed significant improvement in perioral rhytides through week 24 compared to patients treated for lip augmentation only (P<.001).14 Restylane Silk currently is undergoing investigation for cheek rejuvenation (NCT02636894, NCT02679924) and treatment of hand photoaging (NCT02780258).
The most common AEs are listed in the Table. No lip disorders were considered clinically concerning on evaluation. Concomitant lip augmentation and treatment of perioral rhytides yielded similar rates of AEs.14 Restylane Silk is not to be used in patients with known lidocaine allergy.
Restylane Lyft
Restylane Lyft (formerly known as Perlane-L) was approved in 2010 for use in facial rhytides, including NLFs, and gained approval in 2015 for use in cheek augmentation and midface contouring. Only its efficacy and safety for the more recent indication will be reviewed here.
In an evaluator-blinded investigation of 200 patients with mild to substantial bilateral midface deficiency based on the Medicis midface volume scale (MMVS), patients were randomized to receive supraperiosteal and subcutaneous treatment with Restylane Lyft (n=150) or no treatment (n=50).15 Touch-up injections at week 2 or month 12 were available to treatment group patients and all patients were given either an initial treatment or retreatment at 12 months. Primary end point evaluation at week 8 showed that 89% of treatment group patients had at least 1 grade MMVS improvement compared to 16% of the control group (P<.001). Although the percentage of these MMVS responders in the treatment group decreased with each follow-up period to 54.3% at month 12, retreatment was effective in reproducing a similar MMVS response rate as with initial treatment.15 Restylane Lyft is under ongoing investigation for dorsal hand rejuvenation (NCT02650921).
In addition to the common treatment-related AEs listed in the Table, 2 patients reported serious AEs, including bilateral implant-site inflammation and unilateral implant-site hematoma and infection (organism not described), all of which resolved with unspecified treatment.15 Lidocaine allergies are contraindications for use.
Conclusion
Several new options in dermal fillers have been approved in recent years and have demonstrated efficacy and acceptable safety in various cosmetic rejuvenation applications. Restylane Silk and Restylane Lyft are undergoing further studies to evaluate use in hand rejuvenation, an area that currently has few cosmetic filler treatment options. As technology continues to progress and new formulations of dermal fillers with varied properties and benefits are available, clinicians should expect multiple options for use in rhytides, volume deficits, and contouring.
ADDENDUM
After the manuscript was accepted for publication, Juvéderm Volbella XC (Allergan, Inc) was approved by the FDA for use in lip augmentation and thus is not included in this review.
Facial aging is the result of the interplay between loss of skin elasticity, changes in subcutaneous fat and other soft-tissue layers, and skeletal remodeling with chronological age.1 Dermal fillers are effective for the treatment of rhytides, facial scars, and lipoatrophy, as well as facial contouring and augmentation. Given that multiple filler options exist, updated reviews are necessary to inform clinicians of the choices that are available. We provide a detailed review of the clinical efficacy and safety of the dermal fillers with the most recent approvals by the US Food and Drug Administration (FDA).
Polymethylmethacrylate
Polymethylmethacrylate (PMMA) microspheres suspended in bovine collagen and lidocaine 0.3% were approved in 2006 for use in nasolabial folds (NLFs) and in 2014 for acne scars. Now branded as Bellafill (Suneva Medical, Inc), it is the only permanent injectable filler currently available. Once injected, the particles are not reabsorbed and can only be removed by procedural extraction (eg, liposuction of the surrounding fat); however, the permanence of PMMA does not extend to facial rejuvenation, which can last up to 5 years. Prior to use, skin testing for bovine collagen reaction is necessary. In a clinical trial of 147 patients with moderate to severe acne scarring, patients were randomized to receive PMMA in collagen (n=97) or saline (n=50).2 Injections were administered using a linear threading or serial puncture technique, and patients were reevaluated after 4 weeks for touch-up injections. After 6 months, 64% of patients treated with PMMA in collagen achieved improvement in acne scars by 2 points or more on the acne scar rating scale versus 33% of the control group (P=.0005).2
Treatment-related adverse events (AEs) include injection-site pain, bruising, swelling, erythema, and more rarely pruritus and lumps/granulomas.3 A 5-year longitudinal safety investigation of 871 patients initially treated with PMMA in collagen for NLF correction revealed that 17 patients (2.0%) had biopsy-confirmed granulomas with half of these retained at study end.4 Fifteen of these patients were treated with intralesional corticosteroids alone or in combination with intralesional 5-fluorouracil, oral antibiotics, or topical calcineurin inhibitors; 1 patient was untreated and another used topical corticosteroids. The authors noted no correlation between treatment method and granuloma response.4 Polymethylmethacrylate in collagen is contraindicated in patients with lidocaine or bovine collagen sensitivity and is not indicated for use in lip augmentation due to high rates of nodule formation.3
Hyaluronic Acid
Hyaluronic acid (HA) is a naturally occurring glycosaminoglycan polymer found in the extracellular matrix of the dermis. Hyaluronic acid fillers are bacteria derived and come in gel form. A useful advantage of HA fillers compared to other dermal fillers is the commercial availability of hyaluronidase to correct injections. Preinjection skin testing is not necessary.5
This category of nonpermanent dermal fillers has the most robust market choices. Older HA dermal fillers with reliable and proven efficacy are Restylane (Galderma Laboratories, LP)(facial rhytides, lip augmentation), Juvéderm (Ultra/Ultra XC/Ultra Plus/Ultra Plus XC [Allergan, Inc])(facial rhytides, lip augmentation), Hydrelle (Anika Therapeutics, Inc)(facial rhytides), and Prevelle Silk (Mentor Corporation)(facial rhytides); they will not be reviewed here. Newer agents include Belotero Balance (Merz Aesthetics), Juvéderm Voluma XC (Allergan, Inc), Restylane Silk (Galderma Laboratories, LP), and Restylane Lyft (Galderma Laboratories, LP).
Belotero Balance
Belotero Balance is used to treat fine lines and wrinkles, especially NLFs.6 The initial pivotal studies that led to FDA approval in 2011 demonstrated noninferiority and superiority to bovine collagen for use in the treatment of NLFs.7,8 One hundred eighteen patients with bilateral NLFs that were rated as 2 (moderate) or 3 (severe) on the wrinkle severity rating scale (WSRS) were randomized to split-face injection of Belotero Balance in one NLF and bovine collagen in the contralateral NLF.7 An additional injection at week 2 was allowed for optimal correction. Belotero Balance was noninferior to bovine collagen at week 2, with mean improvement in WSRS of 1.52 versus 1.57 (P=.50). Belotero Balance was superior to bovine collagen in mean WSRS improvement at weeks 12 (1.25 vs 0.26; P<.001), 16 (1.09 vs 0.66; P<.001), and 24 (1.08 vs 0.50; P<.001).7 In a subsequent open-label extension study, which included 95 of 118 patients who received Belotero Balance injections in both NLFs at week 24, 80.2% of patients showed sustained improvement in WSRS from baseline for 48 weeks without further injection.8
The first comparative study of Belotero Balance with other established HA fillers at the time—Restylane and Juvéderm Ultra 3/Ultra Plus XC—to treat NLFs demonstrated noninferiority.9 Forty patients with bilateral, moderate to severe NLFs (rated 3 or 4 on the Merz severity scale) were randomized to split-face groups of Belotero Balance versus Restylane or Belotero Balance versus Juvéderm. At 12 months, NLF severity improved from 2.3 to 1.5 in the Restylane group and from 2.3 to 1.6 in the Juvéderm group.9
Belotero Balance has been compared to Juvéderm Ultra XC for use in perioral lines.10 The study included 136 patients with moderate to severe perioral lines, according to the perioral lines severity scale, who were randomized (1:1 ratio) to receive injections of Belotero Balance or Juvéderm Ultra XC to correct upper and lower perioral lines, with assessment at week 2 for optimization. After 6 months, 87% of Juvéderm-treated patients compared to 72% of Belotero Balance–treated patients had 1-point improvement in perioral lines (P<.04). Juvéderm-treated patients also reported significantly less pain than Belotero Balance–treated patients (P<.001).10
Treatment-related AEs are described in the Table, with the majority occurring at lower rates compared to a collagen control group and self-resolving within 2 weeks.7

Juvéderm Voluma XC
Juvéderm Voluma XC was FDA approved in 2013 for cheek augmentation to correct age-related volume deficit restoration by subcutaneous or subperiosteal injections. In its landmark multicenter investigation, 282 patients with moderate to severe midface (eg, zygomaticomalar, anteromedial cheek, submalar regions) volume deficit measured on a validated midface volume deficit scale (MFVDS) were treated with Juvéderm Voluma XC (n=235) or control (n=47).11 Patients were reevaluated at 30 days and 81.9% received touch-up injections. At a 6-month primary evaluation, 86% of the Juvéderm-treated patients versus 39% of the control patients showed 1-point improvement on the MFVDS (P<.001). At 24-months’ follow-up, 44.6% of patients sustained efficacy.11 Of these aforementioned patients, 167 received repeat treatment due to lost correction or patient request and 91.1% improved by 1 point or more on the MFVDS on evaluation 12 months after repeat treatment.12 For this same population of patients, a 2-year extended follow-up of patient-reported outcomes revealed that 49% of patients felt fulfilled in their treatment goals 2 years after treatment and 79% of patients rated improvement from baseline based on the global aesthetic improvement scale.13 Efficacy studies involving Juvéderm Voluma XC are currently ongoing for facial temporal aging (registered at www.clinicaltrials.gov with the identifier NCT02437903) and recruiting for mandibular hypoplasia (NCT02330016).
Common treatment-related AEs are detailed in the Table. Two patients required treatment with hyaluronidase for chronic lumpiness and nodularity following non–treatment-related cellulitis.11 The product is contraindicated in patients with allergy to lidocaine.
Restylane Silk
Restylane Silk was approved in 2014 for lip augmentation and perioral rhytides. Efficacy and safety was demonstrated in a large multicenter randomized investigation in which 221 patients seeking lip augmentation received either Restylane Silk (n=177) injected submucosally for treatment of the upper and lower lips and/or intradermally for perioral rhytides or no treatment (n=44).14 Restylane treatment group patients optionally received touch-up at 2 weeks for optimization. All patients, including the control group, received injections at 6 months. At the 2-month primary end point, 80.2% of the treatment group exhibited at least 1-point improvement in upper lip fullness on the Medicis lip fullness scale compared to 11.9% (P<.001) of the control group; response rates for the lower lips were 84.2% versus 18.4% (P<.001). Patients in the treatment group receiving injections for perioral rhytides showed significant improvement in perioral rhytides through week 24 compared to patients treated for lip augmentation only (P<.001).14 Restylane Silk currently is undergoing investigation for cheek rejuvenation (NCT02636894, NCT02679924) and treatment of hand photoaging (NCT02780258).
The most common AEs are listed in the Table. No lip disorders were considered clinically concerning on evaluation. Concomitant lip augmentation and treatment of perioral rhytides yielded similar rates of AEs.14 Restylane Silk is not to be used in patients with known lidocaine allergy.
Restylane Lyft
Restylane Lyft (formerly known as Perlane-L) was approved in 2010 for use in facial rhytides, including NLFs, and gained approval in 2015 for use in cheek augmentation and midface contouring. Only its efficacy and safety for the more recent indication will be reviewed here.
In an evaluator-blinded investigation of 200 patients with mild to substantial bilateral midface deficiency based on the Medicis midface volume scale (MMVS), patients were randomized to receive supraperiosteal and subcutaneous treatment with Restylane Lyft (n=150) or no treatment (n=50).15 Touch-up injections at week 2 or month 12 were available to treatment group patients and all patients were given either an initial treatment or retreatment at 12 months. Primary end point evaluation at week 8 showed that 89% of treatment group patients had at least 1 grade MMVS improvement compared to 16% of the control group (P<.001). Although the percentage of these MMVS responders in the treatment group decreased with each follow-up period to 54.3% at month 12, retreatment was effective in reproducing a similar MMVS response rate as with initial treatment.15 Restylane Lyft is under ongoing investigation for dorsal hand rejuvenation (NCT02650921).
In addition to the common treatment-related AEs listed in the Table, 2 patients reported serious AEs, including bilateral implant-site inflammation and unilateral implant-site hematoma and infection (organism not described), all of which resolved with unspecified treatment.15 Lidocaine allergies are contraindications for use.
Conclusion
Several new options in dermal fillers have been approved in recent years and have demonstrated efficacy and acceptable safety in various cosmetic rejuvenation applications. Restylane Silk and Restylane Lyft are undergoing further studies to evaluate use in hand rejuvenation, an area that currently has few cosmetic filler treatment options. As technology continues to progress and new formulations of dermal fillers with varied properties and benefits are available, clinicians should expect multiple options for use in rhytides, volume deficits, and contouring.
ADDENDUM
After the manuscript was accepted for publication, Juvéderm Volbella XC (Allergan, Inc) was approved by the FDA for use in lip augmentation and thus is not included in this review.
- Fitzgerald R, Graivier MH, Kane M, et al. Update on facial aging. Aesthet Surg J. 2010;30(suppl):S11-S24.
- Karnik J, Baumann L, Bruce S, et al. A double-blind, randomized, multicenter, controlled trial of suspended polymethylmethacrylate microspheres for the correction of atrophic facial acne scars. J Am Acad Dermatol. 2014;71:77-83.
- Bellafill [package insert]. San Diego, CA: Suneva Medical, Inc; 2015.
- Cohen S, Dover J, Monheit G, et al. Five-year safety and satisfaction study of PMMA-collagen in the correction of nasolabial folds. Dermatol Surg. 2015;41(suppl 1):S302-S313.
- Greene JJ, Sidle DM. The hyaluronic acid fillers: current understanding of the tissue device interface. Facial Plast Surg Clin North Am. 2015;23:423-432.
- Lorenc ZP, Fagien S, Flynn TC, et al. Review of key Belotero Balance safety and efficacy trials. Plast Reconstr Surg. 2013;132(4, suppl 2):33S-40S.
- Narins RS, Coleman W, Donofrio L, et al. Nonanimal sourced hyaluronic acid–based dermal filler using a cohesive polydensified matrix technology is superior to bovine collagen in the correction of moderate to severe nasolabial folds: results from a 6-month, randomized, blinded, controlled, multicenter study. Dermatol Surg. 2010;36(suppl 1):730-740.
- Narins RS, Coleman WP 3rd, Donofrio LM, et al. Improvement in nasolabial folds with a hyaluronic acid filler using a cohesive polydensified matrix technology: results from an 18-month open-label extension trial. Dermatol Surg. 2010;36(suppl 3):1800-1808.
- Prager W, Wissmueller E, Havermann I, et al. A prospective, split-face, randomized, comparative study of safety and 12-month longevity of three formulations of hyaluronic acid dermal filler for treatment of nasolabial folds. Dermatol Surg. 2012;38(7, pt 2):1143-1150.
- Butterwick K, Marmur E, Narurkar V, et al. HYC-24L demonstrates greater effectiveness with less pain than CPM-22.5 for treatment of perioral lines in a randomized controlled trial. Dermatol Surg. 2015;41:1351-1360.
- Jones D, Murphy DK. Volumizing hyaluronic acid filler for midface volume deficit: 2-year results from a pivotal single-blind randomized controlled study. Dermatol Surg. 2013;39:1602-1612.
- Baumann L, Narins RS, Beer K, et al. Volumizing hyaluronic acid filler for midface volume deficit: results after repeat treatment. Dermatol Surg. 2015;41(suppl 1):S284-S292.
- Few J, Cox SE, Paradkar-Mitragotri D, et al. A multicenter, single-blind randomized, controlled study of a volumizing hyaluronic acid filler for midface volume deficit: patient-reported outcomes at 2 years. Aesthet Surg J. 2015;35:589-599.
- Beer K, Glogau RG, Dover JS, et al. A randomized, evaluator-blinded, controlled study of effectiveness and safety of small particle hyaluronic acid plus lidocaine for lip augmentation and perioral rhytides. Dermatol Surg. 2015;41(suppl 1):S127-S136.
- Weiss RA, Moradi A, Bank D, et al. Effectiveness and safety of large gel particle hyaluronic acid with lidocaine for correction of midface volume deficit or contour deficiency. Dermatol Surg. 2016;42:699-709.
- Fitzgerald R, Graivier MH, Kane M, et al. Update on facial aging. Aesthet Surg J. 2010;30(suppl):S11-S24.
- Karnik J, Baumann L, Bruce S, et al. A double-blind, randomized, multicenter, controlled trial of suspended polymethylmethacrylate microspheres for the correction of atrophic facial acne scars. J Am Acad Dermatol. 2014;71:77-83.
- Bellafill [package insert]. San Diego, CA: Suneva Medical, Inc; 2015.
- Cohen S, Dover J, Monheit G, et al. Five-year safety and satisfaction study of PMMA-collagen in the correction of nasolabial folds. Dermatol Surg. 2015;41(suppl 1):S302-S313.
- Greene JJ, Sidle DM. The hyaluronic acid fillers: current understanding of the tissue device interface. Facial Plast Surg Clin North Am. 2015;23:423-432.
- Lorenc ZP, Fagien S, Flynn TC, et al. Review of key Belotero Balance safety and efficacy trials. Plast Reconstr Surg. 2013;132(4, suppl 2):33S-40S.
- Narins RS, Coleman W, Donofrio L, et al. Nonanimal sourced hyaluronic acid–based dermal filler using a cohesive polydensified matrix technology is superior to bovine collagen in the correction of moderate to severe nasolabial folds: results from a 6-month, randomized, blinded, controlled, multicenter study. Dermatol Surg. 2010;36(suppl 1):730-740.
- Narins RS, Coleman WP 3rd, Donofrio LM, et al. Improvement in nasolabial folds with a hyaluronic acid filler using a cohesive polydensified matrix technology: results from an 18-month open-label extension trial. Dermatol Surg. 2010;36(suppl 3):1800-1808.
- Prager W, Wissmueller E, Havermann I, et al. A prospective, split-face, randomized, comparative study of safety and 12-month longevity of three formulations of hyaluronic acid dermal filler for treatment of nasolabial folds. Dermatol Surg. 2012;38(7, pt 2):1143-1150.
- Butterwick K, Marmur E, Narurkar V, et al. HYC-24L demonstrates greater effectiveness with less pain than CPM-22.5 for treatment of perioral lines in a randomized controlled trial. Dermatol Surg. 2015;41:1351-1360.
- Jones D, Murphy DK. Volumizing hyaluronic acid filler for midface volume deficit: 2-year results from a pivotal single-blind randomized controlled study. Dermatol Surg. 2013;39:1602-1612.
- Baumann L, Narins RS, Beer K, et al. Volumizing hyaluronic acid filler for midface volume deficit: results after repeat treatment. Dermatol Surg. 2015;41(suppl 1):S284-S292.
- Few J, Cox SE, Paradkar-Mitragotri D, et al. A multicenter, single-blind randomized, controlled study of a volumizing hyaluronic acid filler for midface volume deficit: patient-reported outcomes at 2 years. Aesthet Surg J. 2015;35:589-599.
- Beer K, Glogau RG, Dover JS, et al. A randomized, evaluator-blinded, controlled study of effectiveness and safety of small particle hyaluronic acid plus lidocaine for lip augmentation and perioral rhytides. Dermatol Surg. 2015;41(suppl 1):S127-S136.
- Weiss RA, Moradi A, Bank D, et al. Effectiveness and safety of large gel particle hyaluronic acid with lidocaine for correction of midface volume deficit or contour deficiency. Dermatol Surg. 2016;42:699-709.
Practice Points
- The merits of new dermal fillers approved by the US Food and Drug Administration should be weighed with an understanding of aesthetic indications of use, duration of efficacy, and common adverse effects, in line with patient preference.
- The most common adverse effects are injection-site contusion, swelling, and pain, usually self-resolving within days to 2 weeks. Patient quality of care can be improved with forewarning and emphasis on alleviating symptoms.
Periocular Fillers and Related Anatomy
Rejuvenation of the periocular area is in high demand among patients who want to look and feel their best. Physicians should understand the complicated anatomy surrounding the eyes before attempting to inject this area with facial fillers, both to understand the aging process and to minimize treatment complications.
Basic Oculoplastic and Orbital Anatomy
The injector should understand the anatomy of the periocular muscles, the orbital osteology, and the secretory and lacrimal system, in addition to the fat, ligaments, and vascular anatomy in this area.1
The eyes are surrounded by fat compartments that provide glide planes for the motion of the eyelids and globe. There are 2 upper eyelid fat-pads—nasal and central [preaponeurotic])—in the upper lid, leaving room for the lacrimal gland laterally. There are 3 fat compartments—nasal, central, and lateral—in the lower eyelid. The nasal and central compartments are separated by the inferior oblique muscle, which elevates and extorts the eye. The orbital septum holds the fat-pads in place in the orbit. The brow fat-pad is the retro-orbicularis oculi fat-pad (ROOF). There are fat compartments that lie in the subcutaneous space along the entire forehead and in the temple. The suborbicularis oculi fat-pad (SOOF) lies over the malar eminence. Superficial and deep submuscular fat compartments of the face have been described.2 Deep fat compartments also have been examined on computed tomography.3
Orbital circulation comes from the internal carotid artery and anastomoses with the supply from the external carotid artery to supply the orbit. The first branch off of the carotid artery is the ophthalmic artery, and the first branch off of the ophthalmic artery is the central retinal artery that enters the optic nerve sheath 1 cm behind the globe to supply the retina. The supraorbital and supratrochlear arteries branch off of the ophthalmic artery and supply the forehead. The supraorbital artery runs through the supraorbital notch (foramen in 8%)1 and can usually be palpated with one’s finger. There are 15 to 20 short posterior ciliary arteries leading to the choroid, 2 long posterior ciliary arteries to the iris circle, and 7 anterior ciliary arteries to the extraocular muscles. The superior and inferior venous systems drain into the cavernous sinus.4
The ligaments are important to signs of facial aging because tissue atrophy occurs along them. The main orbital ligaments are the lateral orbital thickening (known as the LOT) that adheres the eyelids to the lateral orbital rim and the orbitomalar ligament (orbicularis retaining ligament), which is a condensation fibrous tissue that attaches the skin to the inferior orbital rim and orbital septum along the arcus marginalis and defines the superior edge of the SOOF.5 The zygomatic ligament not only suspends the zygomaticus major and zygomaticus minor muscles to the malar eminence but there are osseocutaneous attachments that connect the skin over the zygoma’s malar eminence and demarcate the inferior edge of the SOOF.6
Periocular Aging
The skin, fat, muscles, and bones change and rotate with aging, and not all orbits age in the same manner. Older patients with dermatochalasis (excess skin fat and muscle) often undergo rejuvenation with blepharoplasty, a brow-lift, and a midface-lift, but many atrophic changes can be improved with facial fillers.7,8
As adults age, the soft tissue along the ligaments begins to show atrophy, prime signs of aging that are often improved with fillers. Atrophy along the orbitomalar ligament along the infraorbital rim creates a depressed tear trough, which is an early sign of aging. A 3-point grading system reported by Hirmand8 describes the severity of progressive hallowing. There also is atrophy along the zygomatic cutaneous ligament that creates the malar hollow. The SOOF appears to be more prominent when these areas above and below show atrophy, which creates the look of an unwanted bag known as a festoon. Additionally, there is atrophy along the superior orbital notch where the ophthalmic branch of the trigeminal nerve (V1) and the supraorbital artery traverse. Soft-tissue atrophy along the supraorbital notch resembles the peak at the top of the letter A, giving the slang term A-frame deformity.
Periocular fat can atrophy, hypertrophy, herniate forward as the septum weakens, or become ptotic. Some patients develop hypertrophy and herniation of the superior and inferior orbital fat-pads, while others develop unwanted atrophy leaving a hollow superior orbit and loss of support to the levator muscle that contributes to eyelid ptosis. The frontalis fat deflates, leaving veins, arteries, and the hypertrophied corrugators unwantedly visible. Loss of subcutaneous fat in the glabella contributes to the formation of frown lines between the brows (also called number 11’s). The ROOF deflates in some patients adding to brow ptosis. Loss of the facial frame occurs when temple fat atrophies.
Skeletal rotation also occurs. Throughout a patient’s life, the skeleton remodels itself via activity of osteoclasts and osteoblasts. Pessa et al9,10 has described the expansion of the anterior orbital aperture superomedially and inferolaterally as well as maxillary retrusion that results in angular changes of the midface in relation to the orbital rim. Lambros’ algorithm describes the rotational changes of the cranium where the superior orbit protrudes as the maxilla retreats posteriorly.9-11 The equator of the globe does not change its distance from the ROOF of the orbit, presumably because of its suspension in the orbit by the optic nerve after it passes through the optic canal and trochlea via the superior oblique muscle, but the distance of the inferior equator of the globe to the floor of the orbit increases as the floor of the orbit descends.12
Dermal Fillers for Periocular Rejuvenation
Hyaluronic acid (HA) was first pioneered for use in humans in the late 1970s by ophthalmologists for anterior segment surgery.13-15 Biocompatibility for orthopedic and dermal applications was explored in the early 1990s.16
At this time, no dermal filler is approved by the US Food and Drug Administration for use in the periorbital area. Some fillers are approved for subdermal areas extending to the preperiosteal plane and can be used in the midface such as HA fillers (eg, Restylane Lyft [Galderma Laboratories, LP]), Juvéderm Voluma XC [Allergan, Inc]), poly-L-lactic acid (PLLA), and calcium hydroxylapatite (CaHA). No dermal fillers are approved for use in the forehead, glabella, or temples. Their use is becoming increasingly popular but is considered off label. In addition, cannulas are not approved for use in these areas. Cannulas may be beneficial in that they are thought to create less bruising and have less chance of entering a vessel than needles, but some injectors prefer needles because they are stiffer and therefore more precise.
The ideal filler for the tear trough, superior sulcus, ROOF, over the orbitomalar ligament, forehead, and glabella is one that is somewhat moldable but does not migrate, is not hydrophilic, is smooth to inject, and is reversible should there be any complications. No single filler fits this ideal description, but HAs typically are the first choice.
In vitro studies to determine the stiffness (G') and the ability to flow (viscosity) have been performed.17,18 Calcium hydroxylapatite has the most stiffness, while Belotero Balance (Merz Aesthetics) and Juvéderm Ultra XC (Allergan, Inc) are more soft17 (Table). These guidelines are important but may not correlate directly with how the fillers behave in vivo as demonstrated in animal models.18
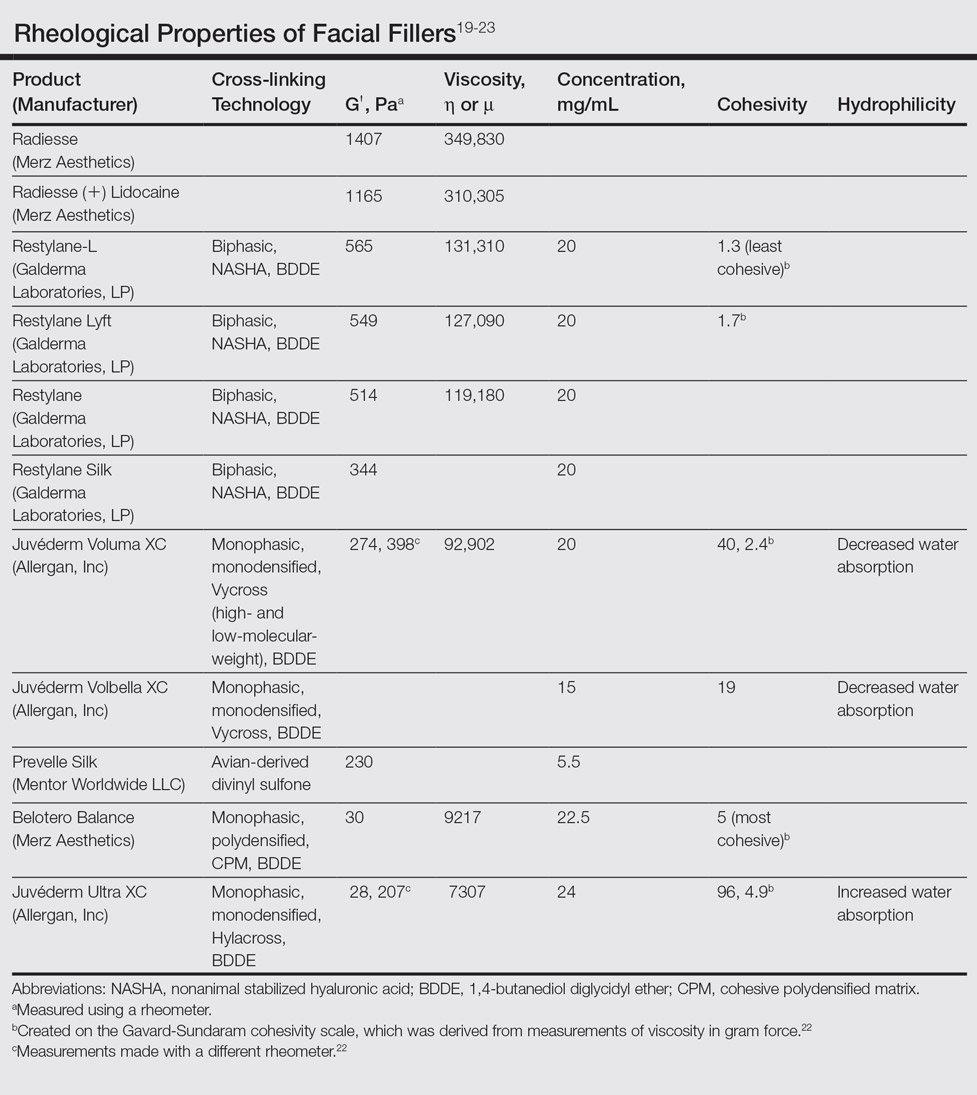
Hyaluronic acid fillers are produced by different technologies to create their cross-link patterns with 1,4-butanediol diglycidyl ether, which determines, to some degree, their behavior in human tissue. Fillers are either monophasic; monodensified; formed by Hylacross (Juvéderm), Vycross (Juvéderm Voluma XC, Juvéderm Volbella XC), or cohesive polydensified matrix technology (Belotero Balance), or biphasic, formed by nonanimal stabilized HA sieving technology (Restylane family). Biopsy has demonstrated that monophasic fillers tend to percolate through and integrate into the tissue, while biphasic fillers dissect tissue to the sides to create a potential space for the filler to reside (Table).24
Periocular Injection Considerations
An experienced injector is one who has developed not only an artistic eye for the face and excellent sense of anatomy but also has a sensitive ability to predict the filler-tissue interaction based on tactile feedback dependent on 3 main qualities: (1) stiffness and viscosity of the filler, (2) gauge of the needle or cannula, and (3) depth of the needle in the tissue. Periocular injections of dermal fillers can be delivered with needles or cannulas, diluted or undiluted. Smaller-gauge needles require more force than larger-gauge needles and cannulas that flow more freely. A needle in the dense dermis will require more force than one placed in the loose subcutaneous space.
The tear trough is generally preferable to fill with a mid-level G' HA filler that is less apt to migrate. A neutral gaze during the injection is preferred because closing or moving the eyes can distort the position of the inferior orbital fat-pads (Figure 1). A needle or cannula can be used, diluted or undiluted. The tear trough can be filled with the injection directed horizontally or vertically via a fanning technique. If needles are used, the skin should be stretched to view the 3 to 5 vertical veins and then the needle should be advanced beneath them to avoid bruising. Avoidance of hydrophilic fillers in the tear trough is important to avoid edema. The superior sulcus can be filled both anteriorly and posteriorly to the septum, which is a highly advanced injection for experienced injectors because of the proximity to the supratrochlear and supraorbital arteries as well as the superior ophthalmic vein (Figure 2). Sharp creases such as deep lateral periocular rhytides known as crow’s-feet are nicely filled with intradermal HAs with a low G'.
Adding volume to the midface is important because it is the continuum of the lower eyelid. Fillers can be injected into multiple levels in this area: deep (to act as pillars to lift the malar eminence and replace bone that has rotated and soft tissue that has become atrophic or descended) and subcutaneous (to efface soft tissue along the zygomatic cutaneous ligament). Higher G' HA fillers and CaHA often are used in the midface along with PLLA. Facial framing of the temples, lateral cheeks, and preauricular area is often accomplished with PLLA but also can be done with mid to high G' HA fillers or CaHA. A cannula may be used to undermine and break apart the zygomatic cutaneous ligament’s cutaneous attachments prior to delivery of the filler in the subcutaneous plane.26 If not done, filler may track away from the hollow area where the ligament is attached and instead move to adjacent areas that will accentuate the hollow and make it look worse.
The temples and lateral face often are filled with PLLA for framing. Mid or high G' HA fillers and CaHA also are used in the temples both beneath the temporalis muscle and also above the deep temporalis fascia or sometimes in the subcutaneous plane.27
Prevention and Management of Periocular Complications
Blindness is the most devastating periocular complication of facial fillers, which is caused by retrograde arterial embolization followed by anterograde flow into the ophthalmic then central retinal arteries. Injectables that have caused blindness include (in descending order of frequency) fat, HA, collagen, paraffin, polymethyl methacrylate, silicone, PLLA, CaHA, polyacrylamide hydrogel, and micronized acellular dermal matrix. Of the 98 cases of blindness from periocular complications from dermal fillers reported in the world literature, the order of affected sites include the glabella (38 cases), nose (25), nasolabial folds (13), superior forehead (12), infraorbital rim (6), temples (1), malar area (1), lip (1), and chin (1). Prevention includes avoidance of danger zone arteries including the supratrochlear, supraorbital, dorsal nasal, angular, infraorbital, zygomaticofacial and zygomaticotemporal arteries.28
Avoiding the average critical volume of 0.84 in any single aliquot dispensed is key to avoid filling of these periocular arteries to the critical bifurcation point that can result in anterograde flow into the eye (Freudenthal Nicolau syndrome). The smallest supratrochlear artery’s volume in this study was 0.04 cc, so aliquots that do not exceed 0.03 cc are ideal.29,30
The injector should always be thinking about the anatomy of the danger zones (eg, infratrochlear and supratrochlear arteries, supraorbital artery, frontal branch of the superficial temporal artery, lacrimal artery, dorsal nasal artery, infraorbital artery, angular artery, zygomaticofacial artery, zygomaticotemporal artery)(Figure 3).
Hyaluronidase can be used off label to hydrolyze unwanted HA. It was first used to aid transcutaneous hydration and was used by ophthalmologists in the 1960s and 1970s to promote the spread of anesthetics by retrobulbar injection.31,32 It can penetrate through soft tissues and blood vessels.33 It is therefore hypothesized that a retrobulbar injection of hyaluronidase could aid in a case of impending blindness34 but has not been successfully accomplished to date. If vision is confirmed to be poor or there is no light perception, a retrobulbar injection of 300 U of hyaluronidase should be given immediately and then repeated in approximately 30 to 45 minutes. The retina begins to show permanent loss of function after being deprived of blood flow for just 97 minutes,35 so there may not be time for an immediate ophthalmology consultation, though such a consultation would be ideal.
Aside from common complications such as bruising and swelling, granulomas and biofilms are well documented in the literature. There are a variety of algorithms to treat such complications, which can happen many weeks after the injection of a dermal filler or years after the injection of a semipermanent filler.36 Postinjection periocular edema can occur years after the initial injection.37,38 Other periocular complications of dermal fillers include nonischemic (eg, bluish hue, filler migration, infection, inflammation, lumps) and ischemic (eg, blindness, necrosis, ophthalmoplegia, ptosis) disturbances.
Conclusion
In summary, periocular injections of facial fillers are useful tools for rejuvenation of the upper face when used with great caution and respect for anatomy.
- Foster J, ed. Orbit, Eyelids, and Lacrimal System. San Francisco, CA: American Academy of Ophthalmology; 2016. 2016-2017 Basic and Clinical Science Course; section 7.
- Rohrich RJ, Pessa JE. The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007;119:2219-2227; discussion 2228-2231.
- Gierloff M, Stöhring C, Buder T, et al. Aging changes of the midfacial fat compartments: a computed tomographic study. Plast Reconst Surg. 2012;129:263-273.
- Zide BM, Jelks GW. Surgical Anatomy of the Orbit: The System of Zones. Philadelphia, PA: Lippincott Williams & Wilkins; 2006.
- Kikkawa DO, Lemke BN, Dortzbach RK. Relations of the superficial musculoaponeurotic system to the orbit and characterization of the oribitomalar ligament. Ophthal Plast Reconstr Surg. 1996;12:77-88.
- Furnas DW. The retaining ligaments of the cheek. Plast Reconstr Surg. 1989;83:11-16.
- Morley AM, Taban M, Malhotra R, et al. Use of hyaluronic acid gel for upper eyelid filling and contouring. Ophthal Plast Reconstr Surg. 2009;25:440-444.
- Hirmand H. Anatomy and nonsurgical correction of the tear trough deformity. Plast Reconstr Surg. 2010;125:699-708.
- Pessa JE, Zadoo VP, Mutimer KL, et al. Relative maxillary retrusion as a natural consequence of aging. Plast Reconstr Surg. 1998;102:205-212.
- Pessa JE, Desvigne LD, Lambros VS, et al. Changes in ocular globe-to-orbital rim position with age: implications for aesthetic blepharoplasty of the lower eyelids. Aesthet Plast Surg. 1999;23:337-345.
- Goldberg RA, Relan A, Hoenig J. Relationship of the eye to the bony orbit, with clinical correlations. Aust N Z J Ophthalmol. 1999;27:398-403.
- Richard MJ, Morris C, Deen BF, et al. Analysis of the anatomic changes of the aging facial skeleton using computer-assisted tomography. Ophthal Plast Reconstr Surg. 2009;25:382-386.
- Miller D, O’Connor P, Williams J. Use of Na-hyaluronate during intraocular lens implantation in rabbits. Ophthalmic Surg. 1977;8:58-61.
- Miller D, Stegmann R. Use of Na-hyaluronate in anterior segment eye surgery. J Am Intraocul Implant Soc. 1980;6:13-15.
- Pape LG, Balazs EA. The use of sodium hyaluronate (Healon) in human anterior segment surgery. Ophthalmology. 1980;87:699-705.
- Larsen NE, Pollak CT, Reiner K, et al. Hylan gel biomaterial: dermal and immunologic compatibility. J Biomed Mater Res. 1993;27:1129-1134.
- Sundaram H, Cassuto D. Biophysical characteristics of hyaluronic acid soft-tissue fillers and their relevance to aesthetic applications. Plast Reconstr Surg. 2013;132(4, suppl 2):5S-21S.
- Hee CK, Shumate GT, Narurkar V, et al. Rheological properties and in vivo performance characteristics of soft tissue fillers. Dermatol Surg. 2015;41(suppl 1):S373-S381.
- Sundaram H, Voigts B, Beer K, et al. Comparison of the rheological properties of viscosity and elasticity in two categories of soft tissue fillers: calcium hydroxylapatite and hyaluronic acid. Dermatol Surg. 2010;36(suppl 3):1859-1865.
- Sundaram H. The new face of fillers: a multi-specialty CME initiative: supplement part II of II. J Drugs Dermatol. 2012;11(suppl 8):S8.
- Stocks D, Sundaram H, Michaels J, et al. Rheological evaluation of the physical properties of hyaluronic acid dermal fillers. J Drugs Dermatol. 2011;10:974-980.
- Goodman GJ, Swift A, Remington BK. Current concepts in the use of Voluma, Volift, and Volbella. Plast Reconstr Surg. 2015;136(suppl 5):139S-148S.
- Sundaram H, Rohrich RJ, Liew S, et al. Cohesivity of hyaluronic acid fillers: development and clinical implications of a novel assay, pilot validation with a five-point grading scale and evaluation of six U.S. Food and Drug Administration–approved fillers. Plast Reconstr Surg. 2015;136:678-686.
- Flynn TC, Sarazin D, Bezzola A, et al. Comparative histology of intradermal implantation of mono and biphasic hyaluronic acid fillers. Dermatol Surg. 2011;37:637-643.
- Woodward JA, Langelier N. Filler enhancement of the superior periocular area [published online Jun 23, 2016]. JAMA Facial Plast Surg. doi:10.1001/jamafacial.2016.0636.
- Cotofana S, Schenck TL, Trevidic P, et al. Midface: clinical anatomy and regional approaches with injectable fillers. Plast Reconstr Surg. 2015;136(suppl 5):219S-234S.
- Buckingham ED, Glasgold R, Kontis T, et al. Volume rejuvenation of the facial upper third. Facial Plast Surg. 2015;31:43-54.
- Beleznay K, Carruthers JD, Humphrey S, et al. Avoiding and treating blindness from fillers: a review of the world literature. Dermatol Surg. 2015;41:1097-1117.
- Coleman SR. Avoidance of arterial occlusion from injection of soft tissue fillers. Aesthet Surg J. 2002;22:555-557.
- Khan T, Colon-Acevedo B, Mettu P, et al. An anatomical analysis of the supratrochlear artery: considerations in facial filler injections and preventing vision loss [published online August 16, 2016]. Aesthet Surg J. pii: sjw132.
- Iserle J, Kumstat Z. Retrobulbar injections of hyaluronidase as a method of increasing safety in cataract surgery [in Czech]. Cesk Oftalmol. 1960;15:126-130.
- Wojtowicz S. Effect of retrobulbar injections of novocaine and lignocaine with adrenalin and hyaluronidase for the immobilization of the eye in electromyography [in Polish]. Klin Oczna. 1964;34:285-296.
- Delorenzi C. Transarterial degradation of hyaluronic acid filler by hyaluronidase. Dermatol Surg. 2014;40:832-841.
- Carruthers J, Fagien S, Dolman P. Retro or peribulbar injections techniques to reverse visual loss after filler injections. 2015;41(suppl 1):S354-S357.
- Hayreh SS, Zimmerman MB, Kimura A, et al. Central retinal artery occlusion. retinal survival time. Exp Eye Res. 2004;78:723-736.
- Woodward J, Khan T, Martin J. Facial filler complications. Facial Plast Surg Clin North Am. 2015;23:447-458.
- Khan TT, Woodward JA. Retained dermal filler in the upper eyelid masquerading as periorbital edema. Dermatol Surg. 2015;41:1182-1184.
- Chang JR, Baharestani S, Salek SS, et al. Delayed superficial migration of retained hyaluronic acid years following periocular injection [published online April 20, 2015]. Ophthal Plast Reconstr Surg. doi:10.1097/IOP.0000000000000434.
Rejuvenation of the periocular area is in high demand among patients who want to look and feel their best. Physicians should understand the complicated anatomy surrounding the eyes before attempting to inject this area with facial fillers, both to understand the aging process and to minimize treatment complications.
Basic Oculoplastic and Orbital Anatomy
The injector should understand the anatomy of the periocular muscles, the orbital osteology, and the secretory and lacrimal system, in addition to the fat, ligaments, and vascular anatomy in this area.1
The eyes are surrounded by fat compartments that provide glide planes for the motion of the eyelids and globe. There are 2 upper eyelid fat-pads—nasal and central [preaponeurotic])—in the upper lid, leaving room for the lacrimal gland laterally. There are 3 fat compartments—nasal, central, and lateral—in the lower eyelid. The nasal and central compartments are separated by the inferior oblique muscle, which elevates and extorts the eye. The orbital septum holds the fat-pads in place in the orbit. The brow fat-pad is the retro-orbicularis oculi fat-pad (ROOF). There are fat compartments that lie in the subcutaneous space along the entire forehead and in the temple. The suborbicularis oculi fat-pad (SOOF) lies over the malar eminence. Superficial and deep submuscular fat compartments of the face have been described.2 Deep fat compartments also have been examined on computed tomography.3
Orbital circulation comes from the internal carotid artery and anastomoses with the supply from the external carotid artery to supply the orbit. The first branch off of the carotid artery is the ophthalmic artery, and the first branch off of the ophthalmic artery is the central retinal artery that enters the optic nerve sheath 1 cm behind the globe to supply the retina. The supraorbital and supratrochlear arteries branch off of the ophthalmic artery and supply the forehead. The supraorbital artery runs through the supraorbital notch (foramen in 8%)1 and can usually be palpated with one’s finger. There are 15 to 20 short posterior ciliary arteries leading to the choroid, 2 long posterior ciliary arteries to the iris circle, and 7 anterior ciliary arteries to the extraocular muscles. The superior and inferior venous systems drain into the cavernous sinus.4
The ligaments are important to signs of facial aging because tissue atrophy occurs along them. The main orbital ligaments are the lateral orbital thickening (known as the LOT) that adheres the eyelids to the lateral orbital rim and the orbitomalar ligament (orbicularis retaining ligament), which is a condensation fibrous tissue that attaches the skin to the inferior orbital rim and orbital septum along the arcus marginalis and defines the superior edge of the SOOF.5 The zygomatic ligament not only suspends the zygomaticus major and zygomaticus minor muscles to the malar eminence but there are osseocutaneous attachments that connect the skin over the zygoma’s malar eminence and demarcate the inferior edge of the SOOF.6
Periocular Aging
The skin, fat, muscles, and bones change and rotate with aging, and not all orbits age in the same manner. Older patients with dermatochalasis (excess skin fat and muscle) often undergo rejuvenation with blepharoplasty, a brow-lift, and a midface-lift, but many atrophic changes can be improved with facial fillers.7,8
As adults age, the soft tissue along the ligaments begins to show atrophy, prime signs of aging that are often improved with fillers. Atrophy along the orbitomalar ligament along the infraorbital rim creates a depressed tear trough, which is an early sign of aging. A 3-point grading system reported by Hirmand8 describes the severity of progressive hallowing. There also is atrophy along the zygomatic cutaneous ligament that creates the malar hollow. The SOOF appears to be more prominent when these areas above and below show atrophy, which creates the look of an unwanted bag known as a festoon. Additionally, there is atrophy along the superior orbital notch where the ophthalmic branch of the trigeminal nerve (V1) and the supraorbital artery traverse. Soft-tissue atrophy along the supraorbital notch resembles the peak at the top of the letter A, giving the slang term A-frame deformity.
Periocular fat can atrophy, hypertrophy, herniate forward as the septum weakens, or become ptotic. Some patients develop hypertrophy and herniation of the superior and inferior orbital fat-pads, while others develop unwanted atrophy leaving a hollow superior orbit and loss of support to the levator muscle that contributes to eyelid ptosis. The frontalis fat deflates, leaving veins, arteries, and the hypertrophied corrugators unwantedly visible. Loss of subcutaneous fat in the glabella contributes to the formation of frown lines between the brows (also called number 11’s). The ROOF deflates in some patients adding to brow ptosis. Loss of the facial frame occurs when temple fat atrophies.
Skeletal rotation also occurs. Throughout a patient’s life, the skeleton remodels itself via activity of osteoclasts and osteoblasts. Pessa et al9,10 has described the expansion of the anterior orbital aperture superomedially and inferolaterally as well as maxillary retrusion that results in angular changes of the midface in relation to the orbital rim. Lambros’ algorithm describes the rotational changes of the cranium where the superior orbit protrudes as the maxilla retreats posteriorly.9-11 The equator of the globe does not change its distance from the ROOF of the orbit, presumably because of its suspension in the orbit by the optic nerve after it passes through the optic canal and trochlea via the superior oblique muscle, but the distance of the inferior equator of the globe to the floor of the orbit increases as the floor of the orbit descends.12
Dermal Fillers for Periocular Rejuvenation
Hyaluronic acid (HA) was first pioneered for use in humans in the late 1970s by ophthalmologists for anterior segment surgery.13-15 Biocompatibility for orthopedic and dermal applications was explored in the early 1990s.16
At this time, no dermal filler is approved by the US Food and Drug Administration for use in the periorbital area. Some fillers are approved for subdermal areas extending to the preperiosteal plane and can be used in the midface such as HA fillers (eg, Restylane Lyft [Galderma Laboratories, LP]), Juvéderm Voluma XC [Allergan, Inc]), poly-L-lactic acid (PLLA), and calcium hydroxylapatite (CaHA). No dermal fillers are approved for use in the forehead, glabella, or temples. Their use is becoming increasingly popular but is considered off label. In addition, cannulas are not approved for use in these areas. Cannulas may be beneficial in that they are thought to create less bruising and have less chance of entering a vessel than needles, but some injectors prefer needles because they are stiffer and therefore more precise.
The ideal filler for the tear trough, superior sulcus, ROOF, over the orbitomalar ligament, forehead, and glabella is one that is somewhat moldable but does not migrate, is not hydrophilic, is smooth to inject, and is reversible should there be any complications. No single filler fits this ideal description, but HAs typically are the first choice.
In vitro studies to determine the stiffness (G') and the ability to flow (viscosity) have been performed.17,18 Calcium hydroxylapatite has the most stiffness, while Belotero Balance (Merz Aesthetics) and Juvéderm Ultra XC (Allergan, Inc) are more soft17 (Table). These guidelines are important but may not correlate directly with how the fillers behave in vivo as demonstrated in animal models.18

Hyaluronic acid fillers are produced by different technologies to create their cross-link patterns with 1,4-butanediol diglycidyl ether, which determines, to some degree, their behavior in human tissue. Fillers are either monophasic; monodensified; formed by Hylacross (Juvéderm), Vycross (Juvéderm Voluma XC, Juvéderm Volbella XC), or cohesive polydensified matrix technology (Belotero Balance), or biphasic, formed by nonanimal stabilized HA sieving technology (Restylane family). Biopsy has demonstrated that monophasic fillers tend to percolate through and integrate into the tissue, while biphasic fillers dissect tissue to the sides to create a potential space for the filler to reside (Table).24
Periocular Injection Considerations
An experienced injector is one who has developed not only an artistic eye for the face and excellent sense of anatomy but also has a sensitive ability to predict the filler-tissue interaction based on tactile feedback dependent on 3 main qualities: (1) stiffness and viscosity of the filler, (2) gauge of the needle or cannula, and (3) depth of the needle in the tissue. Periocular injections of dermal fillers can be delivered with needles or cannulas, diluted or undiluted. Smaller-gauge needles require more force than larger-gauge needles and cannulas that flow more freely. A needle in the dense dermis will require more force than one placed in the loose subcutaneous space.
The tear trough is generally preferable to fill with a mid-level G' HA filler that is less apt to migrate. A neutral gaze during the injection is preferred because closing or moving the eyes can distort the position of the inferior orbital fat-pads (Figure 1). A needle or cannula can be used, diluted or undiluted. The tear trough can be filled with the injection directed horizontally or vertically via a fanning technique. If needles are used, the skin should be stretched to view the 3 to 5 vertical veins and then the needle should be advanced beneath them to avoid bruising. Avoidance of hydrophilic fillers in the tear trough is important to avoid edema. The superior sulcus can be filled both anteriorly and posteriorly to the septum, which is a highly advanced injection for experienced injectors because of the proximity to the supratrochlear and supraorbital arteries as well as the superior ophthalmic vein (Figure 2). Sharp creases such as deep lateral periocular rhytides known as crow’s-feet are nicely filled with intradermal HAs with a low G'.


Adding volume to the midface is important because it is the continuum of the lower eyelid. Fillers can be injected into multiple levels in this area: deep (to act as pillars to lift the malar eminence and replace bone that has rotated and soft tissue that has become atrophic or descended) and subcutaneous (to efface soft tissue along the zygomatic cutaneous ligament). Higher G' HA fillers and CaHA often are used in the midface along with PLLA. Facial framing of the temples, lateral cheeks, and preauricular area is often accomplished with PLLA but also can be done with mid to high G' HA fillers or CaHA. A cannula may be used to undermine and break apart the zygomatic cutaneous ligament’s cutaneous attachments prior to delivery of the filler in the subcutaneous plane.26 If not done, filler may track away from the hollow area where the ligament is attached and instead move to adjacent areas that will accentuate the hollow and make it look worse.
The temples and lateral face often are filled with PLLA for framing. Mid or high G' HA fillers and CaHA also are used in the temples both beneath the temporalis muscle and also above the deep temporalis fascia or sometimes in the subcutaneous plane.27
Prevention and Management of Periocular Complications
Blindness is the most devastating periocular complication of facial fillers, which is caused by retrograde arterial embolization followed by anterograde flow into the ophthalmic then central retinal arteries. Injectables that have caused blindness include (in descending order of frequency) fat, HA, collagen, paraffin, polymethyl methacrylate, silicone, PLLA, CaHA, polyacrylamide hydrogel, and micronized acellular dermal matrix. Of the 98 cases of blindness from periocular complications from dermal fillers reported in the world literature, the order of affected sites include the glabella (38 cases), nose (25), nasolabial folds (13), superior forehead (12), infraorbital rim (6), temples (1), malar area (1), lip (1), and chin (1). Prevention includes avoidance of danger zone arteries including the supratrochlear, supraorbital, dorsal nasal, angular, infraorbital, zygomaticofacial and zygomaticotemporal arteries.28
Avoiding the average critical volume of 0.84 in any single aliquot dispensed is key to avoid filling of these periocular arteries to the critical bifurcation point that can result in anterograde flow into the eye (Freudenthal Nicolau syndrome). The smallest supratrochlear artery’s volume in this study was 0.04 cc, so aliquots that do not exceed 0.03 cc are ideal.29,30
The injector should always be thinking about the anatomy of the danger zones (eg, infratrochlear and supratrochlear arteries, supraorbital artery, frontal branch of the superficial temporal artery, lacrimal artery, dorsal nasal artery, infraorbital artery, angular artery, zygomaticofacial artery, zygomaticotemporal artery)(Figure 3).

Hyaluronidase can be used off label to hydrolyze unwanted HA. It was first used to aid transcutaneous hydration and was used by ophthalmologists in the 1960s and 1970s to promote the spread of anesthetics by retrobulbar injection.31,32 It can penetrate through soft tissues and blood vessels.33 It is therefore hypothesized that a retrobulbar injection of hyaluronidase could aid in a case of impending blindness34 but has not been successfully accomplished to date. If vision is confirmed to be poor or there is no light perception, a retrobulbar injection of 300 U of hyaluronidase should be given immediately and then repeated in approximately 30 to 45 minutes. The retina begins to show permanent loss of function after being deprived of blood flow for just 97 minutes,35 so there may not be time for an immediate ophthalmology consultation, though such a consultation would be ideal.
Aside from common complications such as bruising and swelling, granulomas and biofilms are well documented in the literature. There are a variety of algorithms to treat such complications, which can happen many weeks after the injection of a dermal filler or years after the injection of a semipermanent filler.36 Postinjection periocular edema can occur years after the initial injection.37,38 Other periocular complications of dermal fillers include nonischemic (eg, bluish hue, filler migration, infection, inflammation, lumps) and ischemic (eg, blindness, necrosis, ophthalmoplegia, ptosis) disturbances.
Conclusion
In summary, periocular injections of facial fillers are useful tools for rejuvenation of the upper face when used with great caution and respect for anatomy.
Rejuvenation of the periocular area is in high demand among patients who want to look and feel their best. Physicians should understand the complicated anatomy surrounding the eyes before attempting to inject this area with facial fillers, both to understand the aging process and to minimize treatment complications.
Basic Oculoplastic and Orbital Anatomy
The injector should understand the anatomy of the periocular muscles, the orbital osteology, and the secretory and lacrimal system, in addition to the fat, ligaments, and vascular anatomy in this area.1
The eyes are surrounded by fat compartments that provide glide planes for the motion of the eyelids and globe. There are 2 upper eyelid fat-pads—nasal and central [preaponeurotic])—in the upper lid, leaving room for the lacrimal gland laterally. There are 3 fat compartments—nasal, central, and lateral—in the lower eyelid. The nasal and central compartments are separated by the inferior oblique muscle, which elevates and extorts the eye. The orbital septum holds the fat-pads in place in the orbit. The brow fat-pad is the retro-orbicularis oculi fat-pad (ROOF). There are fat compartments that lie in the subcutaneous space along the entire forehead and in the temple. The suborbicularis oculi fat-pad (SOOF) lies over the malar eminence. Superficial and deep submuscular fat compartments of the face have been described.2 Deep fat compartments also have been examined on computed tomography.3
Orbital circulation comes from the internal carotid artery and anastomoses with the supply from the external carotid artery to supply the orbit. The first branch off of the carotid artery is the ophthalmic artery, and the first branch off of the ophthalmic artery is the central retinal artery that enters the optic nerve sheath 1 cm behind the globe to supply the retina. The supraorbital and supratrochlear arteries branch off of the ophthalmic artery and supply the forehead. The supraorbital artery runs through the supraorbital notch (foramen in 8%)1 and can usually be palpated with one’s finger. There are 15 to 20 short posterior ciliary arteries leading to the choroid, 2 long posterior ciliary arteries to the iris circle, and 7 anterior ciliary arteries to the extraocular muscles. The superior and inferior venous systems drain into the cavernous sinus.4
The ligaments are important to signs of facial aging because tissue atrophy occurs along them. The main orbital ligaments are the lateral orbital thickening (known as the LOT) that adheres the eyelids to the lateral orbital rim and the orbitomalar ligament (orbicularis retaining ligament), which is a condensation fibrous tissue that attaches the skin to the inferior orbital rim and orbital septum along the arcus marginalis and defines the superior edge of the SOOF.5 The zygomatic ligament not only suspends the zygomaticus major and zygomaticus minor muscles to the malar eminence but there are osseocutaneous attachments that connect the skin over the zygoma’s malar eminence and demarcate the inferior edge of the SOOF.6
Periocular Aging
The skin, fat, muscles, and bones change and rotate with aging, and not all orbits age in the same manner. Older patients with dermatochalasis (excess skin fat and muscle) often undergo rejuvenation with blepharoplasty, a brow-lift, and a midface-lift, but many atrophic changes can be improved with facial fillers.7,8
As adults age, the soft tissue along the ligaments begins to show atrophy, prime signs of aging that are often improved with fillers. Atrophy along the orbitomalar ligament along the infraorbital rim creates a depressed tear trough, which is an early sign of aging. A 3-point grading system reported by Hirmand8 describes the severity of progressive hallowing. There also is atrophy along the zygomatic cutaneous ligament that creates the malar hollow. The SOOF appears to be more prominent when these areas above and below show atrophy, which creates the look of an unwanted bag known as a festoon. Additionally, there is atrophy along the superior orbital notch where the ophthalmic branch of the trigeminal nerve (V1) and the supraorbital artery traverse. Soft-tissue atrophy along the supraorbital notch resembles the peak at the top of the letter A, giving the slang term A-frame deformity.
Periocular fat can atrophy, hypertrophy, herniate forward as the septum weakens, or become ptotic. Some patients develop hypertrophy and herniation of the superior and inferior orbital fat-pads, while others develop unwanted atrophy leaving a hollow superior orbit and loss of support to the levator muscle that contributes to eyelid ptosis. The frontalis fat deflates, leaving veins, arteries, and the hypertrophied corrugators unwantedly visible. Loss of subcutaneous fat in the glabella contributes to the formation of frown lines between the brows (also called number 11’s). The ROOF deflates in some patients adding to brow ptosis. Loss of the facial frame occurs when temple fat atrophies.
Skeletal rotation also occurs. Throughout a patient’s life, the skeleton remodels itself via activity of osteoclasts and osteoblasts. Pessa et al9,10 has described the expansion of the anterior orbital aperture superomedially and inferolaterally as well as maxillary retrusion that results in angular changes of the midface in relation to the orbital rim. Lambros’ algorithm describes the rotational changes of the cranium where the superior orbit protrudes as the maxilla retreats posteriorly.9-11 The equator of the globe does not change its distance from the ROOF of the orbit, presumably because of its suspension in the orbit by the optic nerve after it passes through the optic canal and trochlea via the superior oblique muscle, but the distance of the inferior equator of the globe to the floor of the orbit increases as the floor of the orbit descends.12
Dermal Fillers for Periocular Rejuvenation
Hyaluronic acid (HA) was first pioneered for use in humans in the late 1970s by ophthalmologists for anterior segment surgery.13-15 Biocompatibility for orthopedic and dermal applications was explored in the early 1990s.16
At this time, no dermal filler is approved by the US Food and Drug Administration for use in the periorbital area. Some fillers are approved for subdermal areas extending to the preperiosteal plane and can be used in the midface such as HA fillers (eg, Restylane Lyft [Galderma Laboratories, LP]), Juvéderm Voluma XC [Allergan, Inc]), poly-L-lactic acid (PLLA), and calcium hydroxylapatite (CaHA). No dermal fillers are approved for use in the forehead, glabella, or temples. Their use is becoming increasingly popular but is considered off label. In addition, cannulas are not approved for use in these areas. Cannulas may be beneficial in that they are thought to create less bruising and have less chance of entering a vessel than needles, but some injectors prefer needles because they are stiffer and therefore more precise.
The ideal filler for the tear trough, superior sulcus, ROOF, over the orbitomalar ligament, forehead, and glabella is one that is somewhat moldable but does not migrate, is not hydrophilic, is smooth to inject, and is reversible should there be any complications. No single filler fits this ideal description, but HAs typically are the first choice.
In vitro studies to determine the stiffness (G') and the ability to flow (viscosity) have been performed.17,18 Calcium hydroxylapatite has the most stiffness, while Belotero Balance (Merz Aesthetics) and Juvéderm Ultra XC (Allergan, Inc) are more soft17 (Table). These guidelines are important but may not correlate directly with how the fillers behave in vivo as demonstrated in animal models.18

Hyaluronic acid fillers are produced by different technologies to create their cross-link patterns with 1,4-butanediol diglycidyl ether, which determines, to some degree, their behavior in human tissue. Fillers are either monophasic; monodensified; formed by Hylacross (Juvéderm), Vycross (Juvéderm Voluma XC, Juvéderm Volbella XC), or cohesive polydensified matrix technology (Belotero Balance), or biphasic, formed by nonanimal stabilized HA sieving technology (Restylane family). Biopsy has demonstrated that monophasic fillers tend to percolate through and integrate into the tissue, while biphasic fillers dissect tissue to the sides to create a potential space for the filler to reside (Table).24
Periocular Injection Considerations
An experienced injector is one who has developed not only an artistic eye for the face and excellent sense of anatomy but also has a sensitive ability to predict the filler-tissue interaction based on tactile feedback dependent on 3 main qualities: (1) stiffness and viscosity of the filler, (2) gauge of the needle or cannula, and (3) depth of the needle in the tissue. Periocular injections of dermal fillers can be delivered with needles or cannulas, diluted or undiluted. Smaller-gauge needles require more force than larger-gauge needles and cannulas that flow more freely. A needle in the dense dermis will require more force than one placed in the loose subcutaneous space.
The tear trough is generally preferable to fill with a mid-level G' HA filler that is less apt to migrate. A neutral gaze during the injection is preferred because closing or moving the eyes can distort the position of the inferior orbital fat-pads (Figure 1). A needle or cannula can be used, diluted or undiluted. The tear trough can be filled with the injection directed horizontally or vertically via a fanning technique. If needles are used, the skin should be stretched to view the 3 to 5 vertical veins and then the needle should be advanced beneath them to avoid bruising. Avoidance of hydrophilic fillers in the tear trough is important to avoid edema. The superior sulcus can be filled both anteriorly and posteriorly to the septum, which is a highly advanced injection for experienced injectors because of the proximity to the supratrochlear and supraorbital arteries as well as the superior ophthalmic vein (Figure 2). Sharp creases such as deep lateral periocular rhytides known as crow’s-feet are nicely filled with intradermal HAs with a low G'.


Adding volume to the midface is important because it is the continuum of the lower eyelid. Fillers can be injected into multiple levels in this area: deep (to act as pillars to lift the malar eminence and replace bone that has rotated and soft tissue that has become atrophic or descended) and subcutaneous (to efface soft tissue along the zygomatic cutaneous ligament). Higher G' HA fillers and CaHA often are used in the midface along with PLLA. Facial framing of the temples, lateral cheeks, and preauricular area is often accomplished with PLLA but also can be done with mid to high G' HA fillers or CaHA. A cannula may be used to undermine and break apart the zygomatic cutaneous ligament’s cutaneous attachments prior to delivery of the filler in the subcutaneous plane.26 If not done, filler may track away from the hollow area where the ligament is attached and instead move to adjacent areas that will accentuate the hollow and make it look worse.
The temples and lateral face often are filled with PLLA for framing. Mid or high G' HA fillers and CaHA also are used in the temples both beneath the temporalis muscle and also above the deep temporalis fascia or sometimes in the subcutaneous plane.27
Prevention and Management of Periocular Complications
Blindness is the most devastating periocular complication of facial fillers, which is caused by retrograde arterial embolization followed by anterograde flow into the ophthalmic then central retinal arteries. Injectables that have caused blindness include (in descending order of frequency) fat, HA, collagen, paraffin, polymethyl methacrylate, silicone, PLLA, CaHA, polyacrylamide hydrogel, and micronized acellular dermal matrix. Of the 98 cases of blindness from periocular complications from dermal fillers reported in the world literature, the order of affected sites include the glabella (38 cases), nose (25), nasolabial folds (13), superior forehead (12), infraorbital rim (6), temples (1), malar area (1), lip (1), and chin (1). Prevention includes avoidance of danger zone arteries including the supratrochlear, supraorbital, dorsal nasal, angular, infraorbital, zygomaticofacial and zygomaticotemporal arteries.28
Avoiding the average critical volume of 0.84 in any single aliquot dispensed is key to avoid filling of these periocular arteries to the critical bifurcation point that can result in anterograde flow into the eye (Freudenthal Nicolau syndrome). The smallest supratrochlear artery’s volume in this study was 0.04 cc, so aliquots that do not exceed 0.03 cc are ideal.29,30
The injector should always be thinking about the anatomy of the danger zones (eg, infratrochlear and supratrochlear arteries, supraorbital artery, frontal branch of the superficial temporal artery, lacrimal artery, dorsal nasal artery, infraorbital artery, angular artery, zygomaticofacial artery, zygomaticotemporal artery)(Figure 3).

Hyaluronidase can be used off label to hydrolyze unwanted HA. It was first used to aid transcutaneous hydration and was used by ophthalmologists in the 1960s and 1970s to promote the spread of anesthetics by retrobulbar injection.31,32 It can penetrate through soft tissues and blood vessels.33 It is therefore hypothesized that a retrobulbar injection of hyaluronidase could aid in a case of impending blindness34 but has not been successfully accomplished to date. If vision is confirmed to be poor or there is no light perception, a retrobulbar injection of 300 U of hyaluronidase should be given immediately and then repeated in approximately 30 to 45 minutes. The retina begins to show permanent loss of function after being deprived of blood flow for just 97 minutes,35 so there may not be time for an immediate ophthalmology consultation, though such a consultation would be ideal.
Aside from common complications such as bruising and swelling, granulomas and biofilms are well documented in the literature. There are a variety of algorithms to treat such complications, which can happen many weeks after the injection of a dermal filler or years after the injection of a semipermanent filler.36 Postinjection periocular edema can occur years after the initial injection.37,38 Other periocular complications of dermal fillers include nonischemic (eg, bluish hue, filler migration, infection, inflammation, lumps) and ischemic (eg, blindness, necrosis, ophthalmoplegia, ptosis) disturbances.
Conclusion
In summary, periocular injections of facial fillers are useful tools for rejuvenation of the upper face when used with great caution and respect for anatomy.
- Foster J, ed. Orbit, Eyelids, and Lacrimal System. San Francisco, CA: American Academy of Ophthalmology; 2016. 2016-2017 Basic and Clinical Science Course; section 7.
- Rohrich RJ, Pessa JE. The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007;119:2219-2227; discussion 2228-2231.
- Gierloff M, Stöhring C, Buder T, et al. Aging changes of the midfacial fat compartments: a computed tomographic study. Plast Reconst Surg. 2012;129:263-273.
- Zide BM, Jelks GW. Surgical Anatomy of the Orbit: The System of Zones. Philadelphia, PA: Lippincott Williams & Wilkins; 2006.
- Kikkawa DO, Lemke BN, Dortzbach RK. Relations of the superficial musculoaponeurotic system to the orbit and characterization of the oribitomalar ligament. Ophthal Plast Reconstr Surg. 1996;12:77-88.
- Furnas DW. The retaining ligaments of the cheek. Plast Reconstr Surg. 1989;83:11-16.
- Morley AM, Taban M, Malhotra R, et al. Use of hyaluronic acid gel for upper eyelid filling and contouring. Ophthal Plast Reconstr Surg. 2009;25:440-444.
- Hirmand H. Anatomy and nonsurgical correction of the tear trough deformity. Plast Reconstr Surg. 2010;125:699-708.
- Pessa JE, Zadoo VP, Mutimer KL, et al. Relative maxillary retrusion as a natural consequence of aging. Plast Reconstr Surg. 1998;102:205-212.
- Pessa JE, Desvigne LD, Lambros VS, et al. Changes in ocular globe-to-orbital rim position with age: implications for aesthetic blepharoplasty of the lower eyelids. Aesthet Plast Surg. 1999;23:337-345.
- Goldberg RA, Relan A, Hoenig J. Relationship of the eye to the bony orbit, with clinical correlations. Aust N Z J Ophthalmol. 1999;27:398-403.
- Richard MJ, Morris C, Deen BF, et al. Analysis of the anatomic changes of the aging facial skeleton using computer-assisted tomography. Ophthal Plast Reconstr Surg. 2009;25:382-386.
- Miller D, O’Connor P, Williams J. Use of Na-hyaluronate during intraocular lens implantation in rabbits. Ophthalmic Surg. 1977;8:58-61.
- Miller D, Stegmann R. Use of Na-hyaluronate in anterior segment eye surgery. J Am Intraocul Implant Soc. 1980;6:13-15.
- Pape LG, Balazs EA. The use of sodium hyaluronate (Healon) in human anterior segment surgery. Ophthalmology. 1980;87:699-705.
- Larsen NE, Pollak CT, Reiner K, et al. Hylan gel biomaterial: dermal and immunologic compatibility. J Biomed Mater Res. 1993;27:1129-1134.
- Sundaram H, Cassuto D. Biophysical characteristics of hyaluronic acid soft-tissue fillers and their relevance to aesthetic applications. Plast Reconstr Surg. 2013;132(4, suppl 2):5S-21S.
- Hee CK, Shumate GT, Narurkar V, et al. Rheological properties and in vivo performance characteristics of soft tissue fillers. Dermatol Surg. 2015;41(suppl 1):S373-S381.
- Sundaram H, Voigts B, Beer K, et al. Comparison of the rheological properties of viscosity and elasticity in two categories of soft tissue fillers: calcium hydroxylapatite and hyaluronic acid. Dermatol Surg. 2010;36(suppl 3):1859-1865.
- Sundaram H. The new face of fillers: a multi-specialty CME initiative: supplement part II of II. J Drugs Dermatol. 2012;11(suppl 8):S8.
- Stocks D, Sundaram H, Michaels J, et al. Rheological evaluation of the physical properties of hyaluronic acid dermal fillers. J Drugs Dermatol. 2011;10:974-980.
- Goodman GJ, Swift A, Remington BK. Current concepts in the use of Voluma, Volift, and Volbella. Plast Reconstr Surg. 2015;136(suppl 5):139S-148S.
- Sundaram H, Rohrich RJ, Liew S, et al. Cohesivity of hyaluronic acid fillers: development and clinical implications of a novel assay, pilot validation with a five-point grading scale and evaluation of six U.S. Food and Drug Administration–approved fillers. Plast Reconstr Surg. 2015;136:678-686.
- Flynn TC, Sarazin D, Bezzola A, et al. Comparative histology of intradermal implantation of mono and biphasic hyaluronic acid fillers. Dermatol Surg. 2011;37:637-643.
- Woodward JA, Langelier N. Filler enhancement of the superior periocular area [published online Jun 23, 2016]. JAMA Facial Plast Surg. doi:10.1001/jamafacial.2016.0636.
- Cotofana S, Schenck TL, Trevidic P, et al. Midface: clinical anatomy and regional approaches with injectable fillers. Plast Reconstr Surg. 2015;136(suppl 5):219S-234S.
- Buckingham ED, Glasgold R, Kontis T, et al. Volume rejuvenation of the facial upper third. Facial Plast Surg. 2015;31:43-54.
- Beleznay K, Carruthers JD, Humphrey S, et al. Avoiding and treating blindness from fillers: a review of the world literature. Dermatol Surg. 2015;41:1097-1117.
- Coleman SR. Avoidance of arterial occlusion from injection of soft tissue fillers. Aesthet Surg J. 2002;22:555-557.
- Khan T, Colon-Acevedo B, Mettu P, et al. An anatomical analysis of the supratrochlear artery: considerations in facial filler injections and preventing vision loss [published online August 16, 2016]. Aesthet Surg J. pii: sjw132.
- Iserle J, Kumstat Z. Retrobulbar injections of hyaluronidase as a method of increasing safety in cataract surgery [in Czech]. Cesk Oftalmol. 1960;15:126-130.
- Wojtowicz S. Effect of retrobulbar injections of novocaine and lignocaine with adrenalin and hyaluronidase for the immobilization of the eye in electromyography [in Polish]. Klin Oczna. 1964;34:285-296.
- Delorenzi C. Transarterial degradation of hyaluronic acid filler by hyaluronidase. Dermatol Surg. 2014;40:832-841.
- Carruthers J, Fagien S, Dolman P. Retro or peribulbar injections techniques to reverse visual loss after filler injections. 2015;41(suppl 1):S354-S357.
- Hayreh SS, Zimmerman MB, Kimura A, et al. Central retinal artery occlusion. retinal survival time. Exp Eye Res. 2004;78:723-736.
- Woodward J, Khan T, Martin J. Facial filler complications. Facial Plast Surg Clin North Am. 2015;23:447-458.
- Khan TT, Woodward JA. Retained dermal filler in the upper eyelid masquerading as periorbital edema. Dermatol Surg. 2015;41:1182-1184.
- Chang JR, Baharestani S, Salek SS, et al. Delayed superficial migration of retained hyaluronic acid years following periocular injection [published online April 20, 2015]. Ophthal Plast Reconstr Surg. doi:10.1097/IOP.0000000000000434.
- Foster J, ed. Orbit, Eyelids, and Lacrimal System. San Francisco, CA: American Academy of Ophthalmology; 2016. 2016-2017 Basic and Clinical Science Course; section 7.
- Rohrich RJ, Pessa JE. The fat compartments of the face: anatomy and clinical implications for cosmetic surgery. Plast Reconstr Surg. 2007;119:2219-2227; discussion 2228-2231.
- Gierloff M, Stöhring C, Buder T, et al. Aging changes of the midfacial fat compartments: a computed tomographic study. Plast Reconst Surg. 2012;129:263-273.
- Zide BM, Jelks GW. Surgical Anatomy of the Orbit: The System of Zones. Philadelphia, PA: Lippincott Williams & Wilkins; 2006.
- Kikkawa DO, Lemke BN, Dortzbach RK. Relations of the superficial musculoaponeurotic system to the orbit and characterization of the oribitomalar ligament. Ophthal Plast Reconstr Surg. 1996;12:77-88.
- Furnas DW. The retaining ligaments of the cheek. Plast Reconstr Surg. 1989;83:11-16.
- Morley AM, Taban M, Malhotra R, et al. Use of hyaluronic acid gel for upper eyelid filling and contouring. Ophthal Plast Reconstr Surg. 2009;25:440-444.
- Hirmand H. Anatomy and nonsurgical correction of the tear trough deformity. Plast Reconstr Surg. 2010;125:699-708.
- Pessa JE, Zadoo VP, Mutimer KL, et al. Relative maxillary retrusion as a natural consequence of aging. Plast Reconstr Surg. 1998;102:205-212.
- Pessa JE, Desvigne LD, Lambros VS, et al. Changes in ocular globe-to-orbital rim position with age: implications for aesthetic blepharoplasty of the lower eyelids. Aesthet Plast Surg. 1999;23:337-345.
- Goldberg RA, Relan A, Hoenig J. Relationship of the eye to the bony orbit, with clinical correlations. Aust N Z J Ophthalmol. 1999;27:398-403.
- Richard MJ, Morris C, Deen BF, et al. Analysis of the anatomic changes of the aging facial skeleton using computer-assisted tomography. Ophthal Plast Reconstr Surg. 2009;25:382-386.
- Miller D, O’Connor P, Williams J. Use of Na-hyaluronate during intraocular lens implantation in rabbits. Ophthalmic Surg. 1977;8:58-61.
- Miller D, Stegmann R. Use of Na-hyaluronate in anterior segment eye surgery. J Am Intraocul Implant Soc. 1980;6:13-15.
- Pape LG, Balazs EA. The use of sodium hyaluronate (Healon) in human anterior segment surgery. Ophthalmology. 1980;87:699-705.
- Larsen NE, Pollak CT, Reiner K, et al. Hylan gel biomaterial: dermal and immunologic compatibility. J Biomed Mater Res. 1993;27:1129-1134.
- Sundaram H, Cassuto D. Biophysical characteristics of hyaluronic acid soft-tissue fillers and their relevance to aesthetic applications. Plast Reconstr Surg. 2013;132(4, suppl 2):5S-21S.
- Hee CK, Shumate GT, Narurkar V, et al. Rheological properties and in vivo performance characteristics of soft tissue fillers. Dermatol Surg. 2015;41(suppl 1):S373-S381.
- Sundaram H, Voigts B, Beer K, et al. Comparison of the rheological properties of viscosity and elasticity in two categories of soft tissue fillers: calcium hydroxylapatite and hyaluronic acid. Dermatol Surg. 2010;36(suppl 3):1859-1865.
- Sundaram H. The new face of fillers: a multi-specialty CME initiative: supplement part II of II. J Drugs Dermatol. 2012;11(suppl 8):S8.
- Stocks D, Sundaram H, Michaels J, et al. Rheological evaluation of the physical properties of hyaluronic acid dermal fillers. J Drugs Dermatol. 2011;10:974-980.
- Goodman GJ, Swift A, Remington BK. Current concepts in the use of Voluma, Volift, and Volbella. Plast Reconstr Surg. 2015;136(suppl 5):139S-148S.
- Sundaram H, Rohrich RJ, Liew S, et al. Cohesivity of hyaluronic acid fillers: development and clinical implications of a novel assay, pilot validation with a five-point grading scale and evaluation of six U.S. Food and Drug Administration–approved fillers. Plast Reconstr Surg. 2015;136:678-686.
- Flynn TC, Sarazin D, Bezzola A, et al. Comparative histology of intradermal implantation of mono and biphasic hyaluronic acid fillers. Dermatol Surg. 2011;37:637-643.
- Woodward JA, Langelier N. Filler enhancement of the superior periocular area [published online Jun 23, 2016]. JAMA Facial Plast Surg. doi:10.1001/jamafacial.2016.0636.
- Cotofana S, Schenck TL, Trevidic P, et al. Midface: clinical anatomy and regional approaches with injectable fillers. Plast Reconstr Surg. 2015;136(suppl 5):219S-234S.
- Buckingham ED, Glasgold R, Kontis T, et al. Volume rejuvenation of the facial upper third. Facial Plast Surg. 2015;31:43-54.
- Beleznay K, Carruthers JD, Humphrey S, et al. Avoiding and treating blindness from fillers: a review of the world literature. Dermatol Surg. 2015;41:1097-1117.
- Coleman SR. Avoidance of arterial occlusion from injection of soft tissue fillers. Aesthet Surg J. 2002;22:555-557.
- Khan T, Colon-Acevedo B, Mettu P, et al. An anatomical analysis of the supratrochlear artery: considerations in facial filler injections and preventing vision loss [published online August 16, 2016]. Aesthet Surg J. pii: sjw132.
- Iserle J, Kumstat Z. Retrobulbar injections of hyaluronidase as a method of increasing safety in cataract surgery [in Czech]. Cesk Oftalmol. 1960;15:126-130.
- Wojtowicz S. Effect of retrobulbar injections of novocaine and lignocaine with adrenalin and hyaluronidase for the immobilization of the eye in electromyography [in Polish]. Klin Oczna. 1964;34:285-296.
- Delorenzi C. Transarterial degradation of hyaluronic acid filler by hyaluronidase. Dermatol Surg. 2014;40:832-841.
- Carruthers J, Fagien S, Dolman P. Retro or peribulbar injections techniques to reverse visual loss after filler injections. 2015;41(suppl 1):S354-S357.
- Hayreh SS, Zimmerman MB, Kimura A, et al. Central retinal artery occlusion. retinal survival time. Exp Eye Res. 2004;78:723-736.
- Woodward J, Khan T, Martin J. Facial filler complications. Facial Plast Surg Clin North Am. 2015;23:447-458.
- Khan TT, Woodward JA. Retained dermal filler in the upper eyelid masquerading as periorbital edema. Dermatol Surg. 2015;41:1182-1184.
- Chang JR, Baharestani S, Salek SS, et al. Delayed superficial migration of retained hyaluronic acid years following periocular injection [published online April 20, 2015]. Ophthal Plast Reconstr Surg. doi:10.1097/IOP.0000000000000434.
Practice Points
- When performing periocular dermal injections, physicians should understand the complicated anatomy surrounding the eyes and related changes with upper face aging.
- The different rheological properties of facial fillers impact product selection for various areas of the upper face.
- Physicians should be aware of the anatomical danger zones to avoid intravascular embolization.