User login
Understand form and function to make the most of fillers
LAS VEGAS – Combining the right products and anatomical knowledge translates patient concerns into better outcomes when using fillers, according to Burt Steffes, MD, a dermatologist in Fond du Lac, Wisc.
“If you understand differences in products, you’ll get better results and minimize complications” for different areas, Dr. Steffes said in a presentation at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
The physical properties of different fillers impact the results, he pointed out. For example, G prime, a measure of elasticity, varies among fillers; a filler with a higher G prime is more contour stable and is designed to lift tissue, while a product with a lower G prime is designed to spread through tissues and move more naturally with the face.
However, “lift capacity using G prime is only useful across products in the entire family and make up,” Dr. Steffes noted. A recent Allergan-sponsored study emphasized that “product performance is a complex interaction of various properties and tissue interactions not just G prime,” he said (Dermatol Surg, 2015;41:S373-81).
Determining the most effective filler to use depends on the location and area to be corrected.
• Tear troughs: Dr. Steffes recommends avoiding hydrophilic products; he prefers Belotero, Restylane, or Restylane Silk.
• Lips: More flexible products such as Juvederm, Restylane, or Belotero can provide patients with the desired result, he noted.
• Nasolabial fold: Juvederm, Restylane, or Radiesse are appropriate choices likely to produce favorable outcomes, Dr. Steffes said.
• Lateral and midface atrophy: For greater lift and cohesivity in these patients, Dr. Steffes considers Perlane, Voluma, Radiesse, or Sculptra as options.
“Fillers are a great way to help people look and feel better,” but complications occur, so be prepared, he said. “You have to know how to recognize and manage complications.”
Proper management starts with staff education. Provide instruction and training to staff (including reception) on how to recognize impending problems. “Have a written protocol available, and keep nitroglycerin paste, hyaluronidase, and aspirin on hand,” he advised.
Cases of swelling may require hyaluronidase to correct, as may lumps and bumps, Dr. Steffes said. Bruising may be managed with ice packs after the fact, but can often be avoided or minimized by using blunt tip cannulas, slow injections, small aliquots, and small-gauge needles, he added.
In cases of extreme complications such as vascular occlusion, start by massaging the area with nitroglycerin paste, and administer hyaluronidase if necessary, he said. However, be sure to proceed carefully if using hyaluronidase in patients with an allergy to bee stings; use it only in an emergency in these patients and treat anaphylaxis if necessary, he added.
Dr. Steffes had no relevant financial conflicts to disclose.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Combining the right products and anatomical knowledge translates patient concerns into better outcomes when using fillers, according to Burt Steffes, MD, a dermatologist in Fond du Lac, Wisc.
“If you understand differences in products, you’ll get better results and minimize complications” for different areas, Dr. Steffes said in a presentation at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
The physical properties of different fillers impact the results, he pointed out. For example, G prime, a measure of elasticity, varies among fillers; a filler with a higher G prime is more contour stable and is designed to lift tissue, while a product with a lower G prime is designed to spread through tissues and move more naturally with the face.
However, “lift capacity using G prime is only useful across products in the entire family and make up,” Dr. Steffes noted. A recent Allergan-sponsored study emphasized that “product performance is a complex interaction of various properties and tissue interactions not just G prime,” he said (Dermatol Surg, 2015;41:S373-81).
Determining the most effective filler to use depends on the location and area to be corrected.
• Tear troughs: Dr. Steffes recommends avoiding hydrophilic products; he prefers Belotero, Restylane, or Restylane Silk.
• Lips: More flexible products such as Juvederm, Restylane, or Belotero can provide patients with the desired result, he noted.
• Nasolabial fold: Juvederm, Restylane, or Radiesse are appropriate choices likely to produce favorable outcomes, Dr. Steffes said.
• Lateral and midface atrophy: For greater lift and cohesivity in these patients, Dr. Steffes considers Perlane, Voluma, Radiesse, or Sculptra as options.
“Fillers are a great way to help people look and feel better,” but complications occur, so be prepared, he said. “You have to know how to recognize and manage complications.”
Proper management starts with staff education. Provide instruction and training to staff (including reception) on how to recognize impending problems. “Have a written protocol available, and keep nitroglycerin paste, hyaluronidase, and aspirin on hand,” he advised.
Cases of swelling may require hyaluronidase to correct, as may lumps and bumps, Dr. Steffes said. Bruising may be managed with ice packs after the fact, but can often be avoided or minimized by using blunt tip cannulas, slow injections, small aliquots, and small-gauge needles, he added.
In cases of extreme complications such as vascular occlusion, start by massaging the area with nitroglycerin paste, and administer hyaluronidase if necessary, he said. However, be sure to proceed carefully if using hyaluronidase in patients with an allergy to bee stings; use it only in an emergency in these patients and treat anaphylaxis if necessary, he added.
Dr. Steffes had no relevant financial conflicts to disclose.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Combining the right products and anatomical knowledge translates patient concerns into better outcomes when using fillers, according to Burt Steffes, MD, a dermatologist in Fond du Lac, Wisc.
“If you understand differences in products, you’ll get better results and minimize complications” for different areas, Dr. Steffes said in a presentation at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
The physical properties of different fillers impact the results, he pointed out. For example, G prime, a measure of elasticity, varies among fillers; a filler with a higher G prime is more contour stable and is designed to lift tissue, while a product with a lower G prime is designed to spread through tissues and move more naturally with the face.
However, “lift capacity using G prime is only useful across products in the entire family and make up,” Dr. Steffes noted. A recent Allergan-sponsored study emphasized that “product performance is a complex interaction of various properties and tissue interactions not just G prime,” he said (Dermatol Surg, 2015;41:S373-81).
Determining the most effective filler to use depends on the location and area to be corrected.
• Tear troughs: Dr. Steffes recommends avoiding hydrophilic products; he prefers Belotero, Restylane, or Restylane Silk.
• Lips: More flexible products such as Juvederm, Restylane, or Belotero can provide patients with the desired result, he noted.
• Nasolabial fold: Juvederm, Restylane, or Radiesse are appropriate choices likely to produce favorable outcomes, Dr. Steffes said.
• Lateral and midface atrophy: For greater lift and cohesivity in these patients, Dr. Steffes considers Perlane, Voluma, Radiesse, or Sculptra as options.
“Fillers are a great way to help people look and feel better,” but complications occur, so be prepared, he said. “You have to know how to recognize and manage complications.”
Proper management starts with staff education. Provide instruction and training to staff (including reception) on how to recognize impending problems. “Have a written protocol available, and keep nitroglycerin paste, hyaluronidase, and aspirin on hand,” he advised.
Cases of swelling may require hyaluronidase to correct, as may lumps and bumps, Dr. Steffes said. Bruising may be managed with ice packs after the fact, but can often be avoided or minimized by using blunt tip cannulas, slow injections, small aliquots, and small-gauge needles, he added.
In cases of extreme complications such as vascular occlusion, start by massaging the area with nitroglycerin paste, and administer hyaluronidase if necessary, he said. However, be sure to proceed carefully if using hyaluronidase in patients with an allergy to bee stings; use it only in an emergency in these patients and treat anaphylaxis if necessary, he added.
Dr. Steffes had no relevant financial conflicts to disclose.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Simple tips optimize toxin treatments
LAS VEGAS – Listening to patients is the “first and most important step” toward using neuromodulators successfully, according to Burt Steffes, MD.
Clinicians armed with a thorough knowledge of anatomy and the available products can use neuromodulators successfully for a range of aging concerns including the glabellar complex, crow’s feet, marionette lines, smoker’s lines, a dimpled chin, and platysmal bands, Dr. Steffes, a dermatologist in Fond du Lac, Wisc., said in a presentation at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
When diluting toxins, be consistent for best results, he advised. “Pick a concentration you like and stick with it,” he said. Establishing realistic expectations and taking preprocedure photos are essential for optimizing results, he added.
Dr. Steffes shared the following tips for successful toxin treatments:
• Horizontal forehead rhytids. Check the brow position and stay approximately 2 cm above the eyebrows. Be conservative with the amount of product used to avoid the frozen look. In cases of upper eyelid ptosis, “improvement can be achieved with apraclonidine 0.5% drops,” he said.
• Glabellar complex. Dr. Steffes said he usually uses 10-30 units. Avoid blood vessels to avoid bruising, he said, “and ask patients to scowl” to identify the exact location for injections.
• Crow’s feet. Treatment of crow’s feet means managing an area “of great variability,” Dr. Steffes said. He advised using superficial injections to minimize bruising, while staying 2 cm from the orbital rim and adjusting to match the patient’s rhytid pattern. He recommends 1-2 units placed 1 cm below the lower eyelid margin at the mid-pupillary line to decrease the bunching of the lower eyelid.
• Platysmal bands. Managing volume is the key to successful treatment of platysmal bands, said Dr. Steffes. “Injecting too much product into the neck can lead to asymmetry” he cautioned. Pinch the platysmal band between the fingers and inject intramuscularly, using 2 units per injection. “Use conservative amounts and adjust in two weeks if needed,” he said.
Be sure that all patients make follow-up appointments before they leave the office after a procedure, and schedule new patients for follow-up in 2 weeks, said Dr. Steffes. Instruct all patients to contact your office immediately for any concerns including bruising that may be managed within the first 24-48 hours with a low-fluence 595-nm, 532-nm, or 1,064-nm laser, he added.
Dr. Steffes had no relevant financial conflicts to disclose.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Listening to patients is the “first and most important step” toward using neuromodulators successfully, according to Burt Steffes, MD.
Clinicians armed with a thorough knowledge of anatomy and the available products can use neuromodulators successfully for a range of aging concerns including the glabellar complex, crow’s feet, marionette lines, smoker’s lines, a dimpled chin, and platysmal bands, Dr. Steffes, a dermatologist in Fond du Lac, Wisc., said in a presentation at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
When diluting toxins, be consistent for best results, he advised. “Pick a concentration you like and stick with it,” he said. Establishing realistic expectations and taking preprocedure photos are essential for optimizing results, he added.
Dr. Steffes shared the following tips for successful toxin treatments:
• Horizontal forehead rhytids. Check the brow position and stay approximately 2 cm above the eyebrows. Be conservative with the amount of product used to avoid the frozen look. In cases of upper eyelid ptosis, “improvement can be achieved with apraclonidine 0.5% drops,” he said.
• Glabellar complex. Dr. Steffes said he usually uses 10-30 units. Avoid blood vessels to avoid bruising, he said, “and ask patients to scowl” to identify the exact location for injections.
• Crow’s feet. Treatment of crow’s feet means managing an area “of great variability,” Dr. Steffes said. He advised using superficial injections to minimize bruising, while staying 2 cm from the orbital rim and adjusting to match the patient’s rhytid pattern. He recommends 1-2 units placed 1 cm below the lower eyelid margin at the mid-pupillary line to decrease the bunching of the lower eyelid.
• Platysmal bands. Managing volume is the key to successful treatment of platysmal bands, said Dr. Steffes. “Injecting too much product into the neck can lead to asymmetry” he cautioned. Pinch the platysmal band between the fingers and inject intramuscularly, using 2 units per injection. “Use conservative amounts and adjust in two weeks if needed,” he said.
Be sure that all patients make follow-up appointments before they leave the office after a procedure, and schedule new patients for follow-up in 2 weeks, said Dr. Steffes. Instruct all patients to contact your office immediately for any concerns including bruising that may be managed within the first 24-48 hours with a low-fluence 595-nm, 532-nm, or 1,064-nm laser, he added.
Dr. Steffes had no relevant financial conflicts to disclose.
SDEF and this news organization are owned by the same parent company.
LAS VEGAS – Listening to patients is the “first and most important step” toward using neuromodulators successfully, according to Burt Steffes, MD.
Clinicians armed with a thorough knowledge of anatomy and the available products can use neuromodulators successfully for a range of aging concerns including the glabellar complex, crow’s feet, marionette lines, smoker’s lines, a dimpled chin, and platysmal bands, Dr. Steffes, a dermatologist in Fond du Lac, Wisc., said in a presentation at Skin Disease Education Foundation’s annual Las Vegas dermatology seminar.
When diluting toxins, be consistent for best results, he advised. “Pick a concentration you like and stick with it,” he said. Establishing realistic expectations and taking preprocedure photos are essential for optimizing results, he added.
Dr. Steffes shared the following tips for successful toxin treatments:
• Horizontal forehead rhytids. Check the brow position and stay approximately 2 cm above the eyebrows. Be conservative with the amount of product used to avoid the frozen look. In cases of upper eyelid ptosis, “improvement can be achieved with apraclonidine 0.5% drops,” he said.
• Glabellar complex. Dr. Steffes said he usually uses 10-30 units. Avoid blood vessels to avoid bruising, he said, “and ask patients to scowl” to identify the exact location for injections.
• Crow’s feet. Treatment of crow’s feet means managing an area “of great variability,” Dr. Steffes said. He advised using superficial injections to minimize bruising, while staying 2 cm from the orbital rim and adjusting to match the patient’s rhytid pattern. He recommends 1-2 units placed 1 cm below the lower eyelid margin at the mid-pupillary line to decrease the bunching of the lower eyelid.
• Platysmal bands. Managing volume is the key to successful treatment of platysmal bands, said Dr. Steffes. “Injecting too much product into the neck can lead to asymmetry” he cautioned. Pinch the platysmal band between the fingers and inject intramuscularly, using 2 units per injection. “Use conservative amounts and adjust in two weeks if needed,” he said.
Be sure that all patients make follow-up appointments before they leave the office after a procedure, and schedule new patients for follow-up in 2 weeks, said Dr. Steffes. Instruct all patients to contact your office immediately for any concerns including bruising that may be managed within the first 24-48 hours with a low-fluence 595-nm, 532-nm, or 1,064-nm laser, he added.
Dr. Steffes had no relevant financial conflicts to disclose.
SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Advances in Minimally Invasive and Noninvasive Treatments for Submental Fat
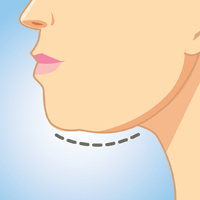
Submental fat (SMF) accumulation within the subcutaneous (preplatysmal) or subplatysmal fat compartment of the cervical anatomy results in an obtuse cervicomental angle and loss of mandibular and cervical contours. It is a common cosmetic concern due to its aesthetic association with weight gain and aging.1 Minimally invasive or noninvasive submental lipolytic agents and techniques are sought for patients who are not candidates for surgery or prefer more conservative cosmetic treatments. These methods typically are only effective in addressing preplatysmal SMF, as subplatysmal SMF requires more surgical methods due to its less-accessible location. The pathology of SMF should initially be assessed by clinical examination or ultrasonography. In this article, we review the most relevant clinical and safety data on minimally invasive and noninvasive treatments for SMF, including laser-assisted lipolysis (LAL), radiofrequency (RF)–assisted lipolysis, deoxycholic acid (DCA), and cryolipolysis.
MINIMALLY INVASIVE MODALITIES
Traditional, or tumescent, liposuction is still widely considered the most effective method for removal of large masses of adiposity. Laser- and RF-assisted adjuncts have been more recently developed to improve patient side effects and recovery time and reduce the manual effort of surgeons. Of note, these adjuncts, with some exceptions, still require the same invasiveness as traditional liposuction, involving submental stab incisions of up to 2.4 mm.
Laser-Assisted Lipolysis
Laser-assisted lipolysis produces a similar effect as suction-assisted lipoplasty by focusing pulses of laser energy through a 1-mm wide fiber optic cannula and inducing thermally mediated adipolysis. The directed laser results in adipocyte rupturing with added benefits of skin retraction and small vessel coagulation, thus lessening intraoperative blood loss.2 This technique typically requires smaller incisions than traditional liposuction. The most common laser lipolysis systems used in cosmetic dermatology are the 920- to 980-nm diode lasers and 1064- to 1440-nm Nd:YAG lasers. The 924-nm diode, 1064-nm Nd:YAG, and 1064/1320-nm Nd:YAG have been best characterized in clinical trials, as reviewed by Fakhouri et al,3 with demonstrated efficacy in reducing SMF density.
The first randomized prospective trial comparing LAL (using 1064-nm Nd:YAG) and traditional liposuction in various anatomical areas on 25 patients showed no difference in cosmetic results, ecchymoses, edema, or retraction, and significantly lower postoperative pain ratings (P<.0001) in LAL.4 A more recent prospective randomized comparison of LAL (980-nm diode laser; 6–8 W) and traditional liposuction of the submental area in 40 female patients showed greater reduction in SMF thickness in the LAL group compared to the liposuction group at 2-month follow-up (6.2 vs 8.22 unspecified units; P<.001) with significant improvement from baseline in both groups (P<.001).5 However, the cosmetic benefit of LAL over traditional liposuction remains controversial and has not been unequivocally established in the literature.
Common adverse events (AEs) are postoperative swelling, ecchymoses, and pain, and complications of interest are nodularity, skin infections, burns, and nerve damage.6 In one retrospective investigation (N=537), these complications occurred at a rate of less than 1% (4 burns and 1 skin infection).6 Patients treated with LAL may report fewer AEs, especially pain and bleeding, compared to liposuction-treated patients.3
RF-Assisted Lipolysis
Radiofrequency-assisted lipolysis is one of the newest technologies in lipocontouring. NeckTite (Invasix Aesthetic Solutions) is effective for treatment of preplatysmal adiposity and cervicomental lipocontouring; a 2.4-mm bipolar probe that is inserted into the subdermal space and connected with an external electrode emits RF energy and simultaneously coagulates and aspirates adipose tissue. NeckTite also may be used in conjunction with FaceTite (Invasix Aesthetic Solutions), which promotes fibroseptal network remodeling and dermal contraction.2
In the first published investigation of the efficacy and safety of NeckTite, 47 of 55 patients received treatment of slight to moderate SMF (average body mass index [BMI], 25 kg/m2) with NeckTite and FaceTite or NeckTite alone.7 At 6-month follow-up, 87% (48/55) of patients subjectively rated treatment efficacy as satisfactory, and 2 independent physicians rated the improvement between before-and-after frontal and lateral photographs of the submental area as moderate to excellent in 95% (52/55) of all cases. Reported complications in this study were full-thickness burns resulting in minor scarring (2/55 [4%]), neck tissue hardness that resolved with daily massage after 3 months (5/55 [9%]), and transient facial nerve paresis of the mandibular branch that resolved after 2 months (1/55 [2%]).7
NONINVASIVE MODALITIES
RF-Assisted Contouring
Another exciting development in RF technology is truSculpt (Cutera), a noninvasive contouring device that is placed over the epidermis and emits RF energy that preferentially heats fat more than other tissue types. In a single-center prospective trial of efficacy and safety in the treatment of SMF, 17 patients received 2 treatments with truSculpt administered 1 month apart.8 At 1- and 6-month follow-up, 82.3% (14/17) and 52.9% (9/17) of patients showed improvement on physician assessment. Submental circumference and ultrasonographic fat thickness reductions at 1-month follow-up were 1.4 cm (5.7% of pretreatment circumference [P<.001]) and 5.4 mm (9.7% of pretreatment fat thickness [P=.005]), respectively. At further longer-term follow-up to 6 months, submental circumference was 0.9 cm (3.8% of pretreatment circumference [P<.001]) and ultrasonographic fat reduction was 6.8 mm (10.5% of pretreatment fat thickness [P=.006]). Commonly reported AEs were pain (rate not given), erythema (8/17 [47%]), edema (1/17 [6%]), and vesicle formation (1/17 [6%]); all were self-resolving. Erythema usually subsided within 6 hours posttreatment. No other AEs or complications were reported.8
Deoxycholic Acid
Deoxycholic acid (DCA)(formerly ATX-101) is an injectable liquid formulation of synthetic DCA that was approved by the US Food and Drug Administration (FDA) in 2015 for moderate to severe SMF. Deoxycholic acid exists endogenously as a bile salt emulsifier and has been shown to cause dose-dependent adipocyte lysis, necrosis, disruption and dissolution of fat architecture, and inflammatory targeting of adipocytes by immune cells.9,10 Thus, DCA causes targeted adipocytolysis and is a novel medical agent in the treatment of SMF. Supplied in 2-mL vials, clinicians may inject 10 mL at each treatment for up to 6 treatments administered 1 month apart.11
Efficacy
REFINE-1, a pivotal North American–based phase 3 trial, investigated the efficacy and safety of DCA.12 A total of 506 participants with scores of 2 (moderate) or 3 (severe) on the Clinician-Reported Submental Fat Rating Scale (CR-SMFRS) and a mean BMI of 29 kg/m2 were randomized to receive preplatysmal fat injections of 2 mg/cm2 of DCA (n=256) or placebo (n=250). Participants received up to 10 mL of product (mean total of 25 mL of DCA across all visits) at each treatment session for up to 6 sessions depending on individual efficacy, with approximately 28 days between sessions. Sixty-four percent of the treatment group received all 6 treatments. At 12-week follow-up after the last treatment session, 70% of DCA-treated participants versus 18.6% of placebo-treated participants (P<.001) improved by 1 grade or more on the CR-SMFRS and 13.4% versus 0% (P<.001) improved by 2 grades or more. Skin laxity was unchanged or improved in 92.7% of the DCA group and 87.6% of the placebo group.12
REFINE-2, the second of the North American phase 3 trials, had parallel inclusionary criteria and study design and established efficacy of 2 mg/cm2 DCA over placebo in 516 participants (randomized 1:1).13 At 12 weeks posttreatment, 66.5% of DCA-treated participants versus 22.2% of placebo-treated participants improved by 1 grade or more according to the CR-SMFRS (P<.001) and 18.6% versus 3% improved by 2 grades or more in SMF (P<.001). Magnetic resonance imaging analysis of participants in the DCA (n=113) and placebo groups (n=112) showed that 40.2% versus 5.2% (P<.001) exhibited 10% or more reduction in submental volume, with similar comparative rates of SMF thickness reduction via caliper measurements.13
Safety
Safety data from REFINE-1 showed higher rates of treatment-related AEs in DCA-treated participants compared to placebo, including hematoma (70% vs 67.3%), anesthesia (66.9% vs 4.4%), pain (65.4% vs 23.4%), edema (52.9% vs 21.8%), induration (18.3% vs 1.6%), paresthesia (12.8% vs 3.2%), nodule formation (12.5% vs 0.8%), and pruritus (8.6% vs 3.6%).12 In this trial, 11 of 258 cases (4.3%) of marginal mandibular nerve paresis and asymmetric smile occurred, all in DCA-treated participants and with a median duration of 31 days. Dysphagia resolving in a median duration of 4 days occurred in 1.6% (4/258) of DCA-treated participants.12 REFINE-2 exhibited similar rates of common AEs. Complications of note were 14 cases of marginal mandibular nerve paresis (11 in DCA group, 3 in placebo group) attributed to injection technique, 1 case of skin ulceration possibly related to accidental injection into dermis, and 6 cases of dysphagia in DCA participants attributed to higher volume treatment sessions and postinjection swelling. Dysphagia lasted a median of 2.5 days and resolved without sequelae.13
Overall, DCA demonstrated high rates of minor injection-site AEs that resolved without sequelae and could be mitigated by comfort therapies (eg, lidocaine, nonsteroidal anti-inflammatory drugs) as well as understanding the anatomy of the submental region. Adverse effects of particular interest included marginal mandibular nerve palsy, skin ulceration, and dysphagia.12,13
Cryolipolysis
Cryolipolysis is an advancement that utilizes the application of noninvasive cooling temperatures to the skin’s surface to destroy underlying adipocytes based on the concept that lipid-filled cells are more susceptible to cold-induced injury than water-filled cells. Thus, cryolipolysis selectively targets adipose tissue, leading to cell death without harm to surrounding cells and without the need for surgery or injections.14
Cryolipolysis typically is delivered via a vacuum applicator (CoolMini, Zeltiq Aesthetics Inc), which applies temperatures of –10°C (14°F) to the skin in cycles of 60 minutes each. Initially approved by the FDA for treatment of flank adiposity in 2010, cryolipolysis has since been approved for treatment of the abdomen, thighs, and submental area.14 An advantage of cryolipolysis is that it does not require frequent treatment sessions for maximal efficacy.
Efficacy
The efficacy of cryolipolysis in the treatment of SMF was established in a multicenter device investigation resulting in its FDA approval for the submental region.15 Sixty participants with a mean BMI of 31.8 kg/m2 received 1 (1/60) or 2 (59/60) treatment sessions of the submental area administered 6 weeks apart. Primary efficacy assessments included analysis by 3 blinded reviewers who viewed photographs of each participant at baseline, immediately posttreatment, 6 weeks posttreatment, and 12 weeks posttreatment; ultrasonographic measurements of SMF thickness; and a 12-point patient satisfaction questionnaire. Blinded reviewers correctly identified baseline images in 91.4% (55/60) of cases. Ultrasonography confirmed a mean reduction in SMF of 2 mm (P<.0001) or 20% of fat thickness at 12 weeks posttreatment. On subjective patient satisfaction surveys, 83% (50/60) of participants were satisfied with the procedure and 77% (46/60) reported a visible reduction in fat and perceived an improvement in appearance.15
Safety
The most common immediate posttreatment AEs were erythema/purpura (100%), numbness (90%), edema (62%), tingling (30%), blanching (25%), and bruising (3%) at the site of cryolipolysis with resolution within 1 week posttreatment, except for numbness.15 At 6-week follow-up, all AEs had resolved, except continued numbness in 4 participants that resolved by 12-week follow-up. A further event of note was fullness in the throat in 1 participant that was attributed to swelling and resolved at 40 days posttreatment without incident. No serious AEs were reported in this trial.15
A particularly concerning but rare complication that is increasing in awareness is paradoxical adipose hyperplasia following cryolipolysis. Patients may develop firm painless areas of soft tissue enlargements in the area of cryolipolysis typically 3 to 6 months posttreatment.16 The largest published report recorded an incidence rate of 0.46% (n=2, all males) at a single-center institution of 422 cryolipolysis treatments.16 Other incidence rates reported are 0.0051% and 0.78%.17 Causes and associations are not known, though male gender is speculated to increase risk.
Conclusion
This article highlights the available information on advances in minimally invasive and noninvasive treatments for SMF accumulation. The efficacy and safety trials varied in quality and in different methods of end point analysis of SMF reduction. Further, few trials have featured head-to-head comparisons of treatments.
Although liposuction and adjuncts remain the gold standard in large-mass lipid removal, these procedures are invasive and exhibit typical risks of surgery. Given its sensitive location, the submental area may require the use of more delicate therapeutic methods, including completely noninvasive devices such as truSculpt and cryolipolysis. Regardless of the chosen treatment, the most important factors in yielding patient satisfaction and SMF improvement are proper patient selection and an understanding of the anatomical source of adiposity to be addressed with the therapeutic modalities.
[polldaddy:9711250]
- Hatef DA, Koshy JC, Sandoval SE, et al. The submental fat compartment of the neck. Semin Plast Surg. 2009;23:288-291.
- Mulholland RS. Nonexcisional, minimally invasive rejuvenation of the neck. Clin Plast Surg. 2014;41:11-31.
- Fakhouri TM, El Tal AK, Abrou AE, et al. Laser-assisted lipolysis: a review. Dermatol Surg. 2012;38:155-169.
- Prado A, Andrades P, Danilla S, et al. A prospective, randomized, double-blind, controlled clinical trial comparing laser-assisted lipoplasty with suction-assisted lipoplasty. Plast Reconstr Surg. 2006;118:1032-1045.
- Valizadeh N, Jalaly NY, Zarghampour M, et al. Evaluation of safety and efficacy of 980-nm diode laser-assisted lipolysis versus traditional liposuction for submental rejuvenation: a randomized clinical trial. J Cosmet Laser Ther. 2016;18:41-45.
- Katz B, McBean J. Laser-assisted lipolysis: a report on complications. J Cosmet Laser Ther. 2008;10:231-233.
- Keramidas E, Rodopoulou S. Radiofrequency-assisted liposuction for neck and lower face adipodermal remodeling and contouring. Plast Reconstr Surg Glob Open. 2016;4:e850.
- Park JH, Kim JI, Park HJ, et al. Evaluation of safety and efficacy of noninvasive radiofrequency technology for submental rejuvenation [published online July 12, 2016]. Lasers Med Sci. 2016;31:1599-1605.
- Yagima Odo ME, Cucé LC, Odo LM, et al. Action of sodium deoxycholate on subcutaneous human tissue: local and systemic effects. Dermatol Surg. 2007;33:178-188; discussion 188-189.
- Rotunda AM, Suzuki H, Moy RL, et al. Detergent effects of sodium deoxycholate are a major feature of an injectable phosphatidylcholine formulation used for localized fat dissolution. Dermatol Surg. 2004;30:1001-1008.
- Kybella [package insert]. Westlake Village, CA: Kythera Biopharmaceuticals, Inc; 2015.
- Jones DH, Carruthers J, Joseph JH, et al. REFINE-1, a multicenter, randomized, double-blind, placebo-controlled, phase 3 trial with ATX-101, an injectable drug for submental fat reduction. Dermatol Surg. 2016;42:38-49.
- Humphrey S, Sykes J, Kantor J, et al. ATX-101 for reduction of submental fat: a phase III randomized controlled trial [published online July 16, 2016]. J Am Acad Dermatol. 2016;75:788-797.e7.
- Manstein D, Laubach H, Watanabe K, et al. Selective cryolysis: a novel method of non-invasive fat removal. Lasers Surg Med. 2008;40:595-604.
- Kilmer SL, Burns AJ, Zelickson BD. Safety and efficacy of cryolipolysis for non-invasive reduction of submental fat. Lasers Surg Med. 2016;48:3-13.
- Singh SM, Geddes ER, Boutrous SG, et al. Paradoxical adipose hyperplasia secondary to cryolipolysis: an underreported entity? Lasers Surg Med. 2015;47:476-478.
- Kelly E, Rodriguez-Feliz J, Kelly ME. Paradoxical adipose hyperplasia after cryolipolysis: a report on incidence and common factors identified in 510 patients. Plast Reconst Surg. 2016;137:639e-640e.
Submental fat (SMF) accumulation within the subcutaneous (preplatysmal) or subplatysmal fat compartment of the cervical anatomy results in an obtuse cervicomental angle and loss of mandibular and cervical contours. It is a common cosmetic concern due to its aesthetic association with weight gain and aging.1 Minimally invasive or noninvasive submental lipolytic agents and techniques are sought for patients who are not candidates for surgery or prefer more conservative cosmetic treatments. These methods typically are only effective in addressing preplatysmal SMF, as subplatysmal SMF requires more surgical methods due to its less-accessible location. The pathology of SMF should initially be assessed by clinical examination or ultrasonography. In this article, we review the most relevant clinical and safety data on minimally invasive and noninvasive treatments for SMF, including laser-assisted lipolysis (LAL), radiofrequency (RF)–assisted lipolysis, deoxycholic acid (DCA), and cryolipolysis.
MINIMALLY INVASIVE MODALITIES
Traditional, or tumescent, liposuction is still widely considered the most effective method for removal of large masses of adiposity. Laser- and RF-assisted adjuncts have been more recently developed to improve patient side effects and recovery time and reduce the manual effort of surgeons. Of note, these adjuncts, with some exceptions, still require the same invasiveness as traditional liposuction, involving submental stab incisions of up to 2.4 mm.
Laser-Assisted Lipolysis
Laser-assisted lipolysis produces a similar effect as suction-assisted lipoplasty by focusing pulses of laser energy through a 1-mm wide fiber optic cannula and inducing thermally mediated adipolysis. The directed laser results in adipocyte rupturing with added benefits of skin retraction and small vessel coagulation, thus lessening intraoperative blood loss.2 This technique typically requires smaller incisions than traditional liposuction. The most common laser lipolysis systems used in cosmetic dermatology are the 920- to 980-nm diode lasers and 1064- to 1440-nm Nd:YAG lasers. The 924-nm diode, 1064-nm Nd:YAG, and 1064/1320-nm Nd:YAG have been best characterized in clinical trials, as reviewed by Fakhouri et al,3 with demonstrated efficacy in reducing SMF density.
The first randomized prospective trial comparing LAL (using 1064-nm Nd:YAG) and traditional liposuction in various anatomical areas on 25 patients showed no difference in cosmetic results, ecchymoses, edema, or retraction, and significantly lower postoperative pain ratings (P<.0001) in LAL.4 A more recent prospective randomized comparison of LAL (980-nm diode laser; 6–8 W) and traditional liposuction of the submental area in 40 female patients showed greater reduction in SMF thickness in the LAL group compared to the liposuction group at 2-month follow-up (6.2 vs 8.22 unspecified units; P<.001) with significant improvement from baseline in both groups (P<.001).5 However, the cosmetic benefit of LAL over traditional liposuction remains controversial and has not been unequivocally established in the literature.
Common adverse events (AEs) are postoperative swelling, ecchymoses, and pain, and complications of interest are nodularity, skin infections, burns, and nerve damage.6 In one retrospective investigation (N=537), these complications occurred at a rate of less than 1% (4 burns and 1 skin infection).6 Patients treated with LAL may report fewer AEs, especially pain and bleeding, compared to liposuction-treated patients.3
RF-Assisted Lipolysis
Radiofrequency-assisted lipolysis is one of the newest technologies in lipocontouring. NeckTite (Invasix Aesthetic Solutions) is effective for treatment of preplatysmal adiposity and cervicomental lipocontouring; a 2.4-mm bipolar probe that is inserted into the subdermal space and connected with an external electrode emits RF energy and simultaneously coagulates and aspirates adipose tissue. NeckTite also may be used in conjunction with FaceTite (Invasix Aesthetic Solutions), which promotes fibroseptal network remodeling and dermal contraction.2
In the first published investigation of the efficacy and safety of NeckTite, 47 of 55 patients received treatment of slight to moderate SMF (average body mass index [BMI], 25 kg/m2) with NeckTite and FaceTite or NeckTite alone.7 At 6-month follow-up, 87% (48/55) of patients subjectively rated treatment efficacy as satisfactory, and 2 independent physicians rated the improvement between before-and-after frontal and lateral photographs of the submental area as moderate to excellent in 95% (52/55) of all cases. Reported complications in this study were full-thickness burns resulting in minor scarring (2/55 [4%]), neck tissue hardness that resolved with daily massage after 3 months (5/55 [9%]), and transient facial nerve paresis of the mandibular branch that resolved after 2 months (1/55 [2%]).7
NONINVASIVE MODALITIES
RF-Assisted Contouring
Another exciting development in RF technology is truSculpt (Cutera), a noninvasive contouring device that is placed over the epidermis and emits RF energy that preferentially heats fat more than other tissue types. In a single-center prospective trial of efficacy and safety in the treatment of SMF, 17 patients received 2 treatments with truSculpt administered 1 month apart.8 At 1- and 6-month follow-up, 82.3% (14/17) and 52.9% (9/17) of patients showed improvement on physician assessment. Submental circumference and ultrasonographic fat thickness reductions at 1-month follow-up were 1.4 cm (5.7% of pretreatment circumference [P<.001]) and 5.4 mm (9.7% of pretreatment fat thickness [P=.005]), respectively. At further longer-term follow-up to 6 months, submental circumference was 0.9 cm (3.8% of pretreatment circumference [P<.001]) and ultrasonographic fat reduction was 6.8 mm (10.5% of pretreatment fat thickness [P=.006]). Commonly reported AEs were pain (rate not given), erythema (8/17 [47%]), edema (1/17 [6%]), and vesicle formation (1/17 [6%]); all were self-resolving. Erythema usually subsided within 6 hours posttreatment. No other AEs or complications were reported.8
Deoxycholic Acid
Deoxycholic acid (DCA)(formerly ATX-101) is an injectable liquid formulation of synthetic DCA that was approved by the US Food and Drug Administration (FDA) in 2015 for moderate to severe SMF. Deoxycholic acid exists endogenously as a bile salt emulsifier and has been shown to cause dose-dependent adipocyte lysis, necrosis, disruption and dissolution of fat architecture, and inflammatory targeting of adipocytes by immune cells.9,10 Thus, DCA causes targeted adipocytolysis and is a novel medical agent in the treatment of SMF. Supplied in 2-mL vials, clinicians may inject 10 mL at each treatment for up to 6 treatments administered 1 month apart.11
Efficacy
REFINE-1, a pivotal North American–based phase 3 trial, investigated the efficacy and safety of DCA.12 A total of 506 participants with scores of 2 (moderate) or 3 (severe) on the Clinician-Reported Submental Fat Rating Scale (CR-SMFRS) and a mean BMI of 29 kg/m2 were randomized to receive preplatysmal fat injections of 2 mg/cm2 of DCA (n=256) or placebo (n=250). Participants received up to 10 mL of product (mean total of 25 mL of DCA across all visits) at each treatment session for up to 6 sessions depending on individual efficacy, with approximately 28 days between sessions. Sixty-four percent of the treatment group received all 6 treatments. At 12-week follow-up after the last treatment session, 70% of DCA-treated participants versus 18.6% of placebo-treated participants (P<.001) improved by 1 grade or more on the CR-SMFRS and 13.4% versus 0% (P<.001) improved by 2 grades or more. Skin laxity was unchanged or improved in 92.7% of the DCA group and 87.6% of the placebo group.12
REFINE-2, the second of the North American phase 3 trials, had parallel inclusionary criteria and study design and established efficacy of 2 mg/cm2 DCA over placebo in 516 participants (randomized 1:1).13 At 12 weeks posttreatment, 66.5% of DCA-treated participants versus 22.2% of placebo-treated participants improved by 1 grade or more according to the CR-SMFRS (P<.001) and 18.6% versus 3% improved by 2 grades or more in SMF (P<.001). Magnetic resonance imaging analysis of participants in the DCA (n=113) and placebo groups (n=112) showed that 40.2% versus 5.2% (P<.001) exhibited 10% or more reduction in submental volume, with similar comparative rates of SMF thickness reduction via caliper measurements.13
Safety
Safety data from REFINE-1 showed higher rates of treatment-related AEs in DCA-treated participants compared to placebo, including hematoma (70% vs 67.3%), anesthesia (66.9% vs 4.4%), pain (65.4% vs 23.4%), edema (52.9% vs 21.8%), induration (18.3% vs 1.6%), paresthesia (12.8% vs 3.2%), nodule formation (12.5% vs 0.8%), and pruritus (8.6% vs 3.6%).12 In this trial, 11 of 258 cases (4.3%) of marginal mandibular nerve paresis and asymmetric smile occurred, all in DCA-treated participants and with a median duration of 31 days. Dysphagia resolving in a median duration of 4 days occurred in 1.6% (4/258) of DCA-treated participants.12 REFINE-2 exhibited similar rates of common AEs. Complications of note were 14 cases of marginal mandibular nerve paresis (11 in DCA group, 3 in placebo group) attributed to injection technique, 1 case of skin ulceration possibly related to accidental injection into dermis, and 6 cases of dysphagia in DCA participants attributed to higher volume treatment sessions and postinjection swelling. Dysphagia lasted a median of 2.5 days and resolved without sequelae.13
Overall, DCA demonstrated high rates of minor injection-site AEs that resolved without sequelae and could be mitigated by comfort therapies (eg, lidocaine, nonsteroidal anti-inflammatory drugs) as well as understanding the anatomy of the submental region. Adverse effects of particular interest included marginal mandibular nerve palsy, skin ulceration, and dysphagia.12,13
Cryolipolysis
Cryolipolysis is an advancement that utilizes the application of noninvasive cooling temperatures to the skin’s surface to destroy underlying adipocytes based on the concept that lipid-filled cells are more susceptible to cold-induced injury than water-filled cells. Thus, cryolipolysis selectively targets adipose tissue, leading to cell death without harm to surrounding cells and without the need for surgery or injections.14
Cryolipolysis typically is delivered via a vacuum applicator (CoolMini, Zeltiq Aesthetics Inc), which applies temperatures of –10°C (14°F) to the skin in cycles of 60 minutes each. Initially approved by the FDA for treatment of flank adiposity in 2010, cryolipolysis has since been approved for treatment of the abdomen, thighs, and submental area.14 An advantage of cryolipolysis is that it does not require frequent treatment sessions for maximal efficacy.
Efficacy
The efficacy of cryolipolysis in the treatment of SMF was established in a multicenter device investigation resulting in its FDA approval for the submental region.15 Sixty participants with a mean BMI of 31.8 kg/m2 received 1 (1/60) or 2 (59/60) treatment sessions of the submental area administered 6 weeks apart. Primary efficacy assessments included analysis by 3 blinded reviewers who viewed photographs of each participant at baseline, immediately posttreatment, 6 weeks posttreatment, and 12 weeks posttreatment; ultrasonographic measurements of SMF thickness; and a 12-point patient satisfaction questionnaire. Blinded reviewers correctly identified baseline images in 91.4% (55/60) of cases. Ultrasonography confirmed a mean reduction in SMF of 2 mm (P<.0001) or 20% of fat thickness at 12 weeks posttreatment. On subjective patient satisfaction surveys, 83% (50/60) of participants were satisfied with the procedure and 77% (46/60) reported a visible reduction in fat and perceived an improvement in appearance.15
Safety
The most common immediate posttreatment AEs were erythema/purpura (100%), numbness (90%), edema (62%), tingling (30%), blanching (25%), and bruising (3%) at the site of cryolipolysis with resolution within 1 week posttreatment, except for numbness.15 At 6-week follow-up, all AEs had resolved, except continued numbness in 4 participants that resolved by 12-week follow-up. A further event of note was fullness in the throat in 1 participant that was attributed to swelling and resolved at 40 days posttreatment without incident. No serious AEs were reported in this trial.15
A particularly concerning but rare complication that is increasing in awareness is paradoxical adipose hyperplasia following cryolipolysis. Patients may develop firm painless areas of soft tissue enlargements in the area of cryolipolysis typically 3 to 6 months posttreatment.16 The largest published report recorded an incidence rate of 0.46% (n=2, all males) at a single-center institution of 422 cryolipolysis treatments.16 Other incidence rates reported are 0.0051% and 0.78%.17 Causes and associations are not known, though male gender is speculated to increase risk.
Conclusion
This article highlights the available information on advances in minimally invasive and noninvasive treatments for SMF accumulation. The efficacy and safety trials varied in quality and in different methods of end point analysis of SMF reduction. Further, few trials have featured head-to-head comparisons of treatments.
Although liposuction and adjuncts remain the gold standard in large-mass lipid removal, these procedures are invasive and exhibit typical risks of surgery. Given its sensitive location, the submental area may require the use of more delicate therapeutic methods, including completely noninvasive devices such as truSculpt and cryolipolysis. Regardless of the chosen treatment, the most important factors in yielding patient satisfaction and SMF improvement are proper patient selection and an understanding of the anatomical source of adiposity to be addressed with the therapeutic modalities.
[polldaddy:9711250]
Submental fat (SMF) accumulation within the subcutaneous (preplatysmal) or subplatysmal fat compartment of the cervical anatomy results in an obtuse cervicomental angle and loss of mandibular and cervical contours. It is a common cosmetic concern due to its aesthetic association with weight gain and aging.1 Minimally invasive or noninvasive submental lipolytic agents and techniques are sought for patients who are not candidates for surgery or prefer more conservative cosmetic treatments. These methods typically are only effective in addressing preplatysmal SMF, as subplatysmal SMF requires more surgical methods due to its less-accessible location. The pathology of SMF should initially be assessed by clinical examination or ultrasonography. In this article, we review the most relevant clinical and safety data on minimally invasive and noninvasive treatments for SMF, including laser-assisted lipolysis (LAL), radiofrequency (RF)–assisted lipolysis, deoxycholic acid (DCA), and cryolipolysis.
MINIMALLY INVASIVE MODALITIES
Traditional, or tumescent, liposuction is still widely considered the most effective method for removal of large masses of adiposity. Laser- and RF-assisted adjuncts have been more recently developed to improve patient side effects and recovery time and reduce the manual effort of surgeons. Of note, these adjuncts, with some exceptions, still require the same invasiveness as traditional liposuction, involving submental stab incisions of up to 2.4 mm.
Laser-Assisted Lipolysis
Laser-assisted lipolysis produces a similar effect as suction-assisted lipoplasty by focusing pulses of laser energy through a 1-mm wide fiber optic cannula and inducing thermally mediated adipolysis. The directed laser results in adipocyte rupturing with added benefits of skin retraction and small vessel coagulation, thus lessening intraoperative blood loss.2 This technique typically requires smaller incisions than traditional liposuction. The most common laser lipolysis systems used in cosmetic dermatology are the 920- to 980-nm diode lasers and 1064- to 1440-nm Nd:YAG lasers. The 924-nm diode, 1064-nm Nd:YAG, and 1064/1320-nm Nd:YAG have been best characterized in clinical trials, as reviewed by Fakhouri et al,3 with demonstrated efficacy in reducing SMF density.
The first randomized prospective trial comparing LAL (using 1064-nm Nd:YAG) and traditional liposuction in various anatomical areas on 25 patients showed no difference in cosmetic results, ecchymoses, edema, or retraction, and significantly lower postoperative pain ratings (P<.0001) in LAL.4 A more recent prospective randomized comparison of LAL (980-nm diode laser; 6–8 W) and traditional liposuction of the submental area in 40 female patients showed greater reduction in SMF thickness in the LAL group compared to the liposuction group at 2-month follow-up (6.2 vs 8.22 unspecified units; P<.001) with significant improvement from baseline in both groups (P<.001).5 However, the cosmetic benefit of LAL over traditional liposuction remains controversial and has not been unequivocally established in the literature.
Common adverse events (AEs) are postoperative swelling, ecchymoses, and pain, and complications of interest are nodularity, skin infections, burns, and nerve damage.6 In one retrospective investigation (N=537), these complications occurred at a rate of less than 1% (4 burns and 1 skin infection).6 Patients treated with LAL may report fewer AEs, especially pain and bleeding, compared to liposuction-treated patients.3
RF-Assisted Lipolysis
Radiofrequency-assisted lipolysis is one of the newest technologies in lipocontouring. NeckTite (Invasix Aesthetic Solutions) is effective for treatment of preplatysmal adiposity and cervicomental lipocontouring; a 2.4-mm bipolar probe that is inserted into the subdermal space and connected with an external electrode emits RF energy and simultaneously coagulates and aspirates adipose tissue. NeckTite also may be used in conjunction with FaceTite (Invasix Aesthetic Solutions), which promotes fibroseptal network remodeling and dermal contraction.2
In the first published investigation of the efficacy and safety of NeckTite, 47 of 55 patients received treatment of slight to moderate SMF (average body mass index [BMI], 25 kg/m2) with NeckTite and FaceTite or NeckTite alone.7 At 6-month follow-up, 87% (48/55) of patients subjectively rated treatment efficacy as satisfactory, and 2 independent physicians rated the improvement between before-and-after frontal and lateral photographs of the submental area as moderate to excellent in 95% (52/55) of all cases. Reported complications in this study were full-thickness burns resulting in minor scarring (2/55 [4%]), neck tissue hardness that resolved with daily massage after 3 months (5/55 [9%]), and transient facial nerve paresis of the mandibular branch that resolved after 2 months (1/55 [2%]).7
NONINVASIVE MODALITIES
RF-Assisted Contouring
Another exciting development in RF technology is truSculpt (Cutera), a noninvasive contouring device that is placed over the epidermis and emits RF energy that preferentially heats fat more than other tissue types. In a single-center prospective trial of efficacy and safety in the treatment of SMF, 17 patients received 2 treatments with truSculpt administered 1 month apart.8 At 1- and 6-month follow-up, 82.3% (14/17) and 52.9% (9/17) of patients showed improvement on physician assessment. Submental circumference and ultrasonographic fat thickness reductions at 1-month follow-up were 1.4 cm (5.7% of pretreatment circumference [P<.001]) and 5.4 mm (9.7% of pretreatment fat thickness [P=.005]), respectively. At further longer-term follow-up to 6 months, submental circumference was 0.9 cm (3.8% of pretreatment circumference [P<.001]) and ultrasonographic fat reduction was 6.8 mm (10.5% of pretreatment fat thickness [P=.006]). Commonly reported AEs were pain (rate not given), erythema (8/17 [47%]), edema (1/17 [6%]), and vesicle formation (1/17 [6%]); all were self-resolving. Erythema usually subsided within 6 hours posttreatment. No other AEs or complications were reported.8
Deoxycholic Acid
Deoxycholic acid (DCA)(formerly ATX-101) is an injectable liquid formulation of synthetic DCA that was approved by the US Food and Drug Administration (FDA) in 2015 for moderate to severe SMF. Deoxycholic acid exists endogenously as a bile salt emulsifier and has been shown to cause dose-dependent adipocyte lysis, necrosis, disruption and dissolution of fat architecture, and inflammatory targeting of adipocytes by immune cells.9,10 Thus, DCA causes targeted adipocytolysis and is a novel medical agent in the treatment of SMF. Supplied in 2-mL vials, clinicians may inject 10 mL at each treatment for up to 6 treatments administered 1 month apart.11
Efficacy
REFINE-1, a pivotal North American–based phase 3 trial, investigated the efficacy and safety of DCA.12 A total of 506 participants with scores of 2 (moderate) or 3 (severe) on the Clinician-Reported Submental Fat Rating Scale (CR-SMFRS) and a mean BMI of 29 kg/m2 were randomized to receive preplatysmal fat injections of 2 mg/cm2 of DCA (n=256) or placebo (n=250). Participants received up to 10 mL of product (mean total of 25 mL of DCA across all visits) at each treatment session for up to 6 sessions depending on individual efficacy, with approximately 28 days between sessions. Sixty-four percent of the treatment group received all 6 treatments. At 12-week follow-up after the last treatment session, 70% of DCA-treated participants versus 18.6% of placebo-treated participants (P<.001) improved by 1 grade or more on the CR-SMFRS and 13.4% versus 0% (P<.001) improved by 2 grades or more. Skin laxity was unchanged or improved in 92.7% of the DCA group and 87.6% of the placebo group.12
REFINE-2, the second of the North American phase 3 trials, had parallel inclusionary criteria and study design and established efficacy of 2 mg/cm2 DCA over placebo in 516 participants (randomized 1:1).13 At 12 weeks posttreatment, 66.5% of DCA-treated participants versus 22.2% of placebo-treated participants improved by 1 grade or more according to the CR-SMFRS (P<.001) and 18.6% versus 3% improved by 2 grades or more in SMF (P<.001). Magnetic resonance imaging analysis of participants in the DCA (n=113) and placebo groups (n=112) showed that 40.2% versus 5.2% (P<.001) exhibited 10% or more reduction in submental volume, with similar comparative rates of SMF thickness reduction via caliper measurements.13
Safety
Safety data from REFINE-1 showed higher rates of treatment-related AEs in DCA-treated participants compared to placebo, including hematoma (70% vs 67.3%), anesthesia (66.9% vs 4.4%), pain (65.4% vs 23.4%), edema (52.9% vs 21.8%), induration (18.3% vs 1.6%), paresthesia (12.8% vs 3.2%), nodule formation (12.5% vs 0.8%), and pruritus (8.6% vs 3.6%).12 In this trial, 11 of 258 cases (4.3%) of marginal mandibular nerve paresis and asymmetric smile occurred, all in DCA-treated participants and with a median duration of 31 days. Dysphagia resolving in a median duration of 4 days occurred in 1.6% (4/258) of DCA-treated participants.12 REFINE-2 exhibited similar rates of common AEs. Complications of note were 14 cases of marginal mandibular nerve paresis (11 in DCA group, 3 in placebo group) attributed to injection technique, 1 case of skin ulceration possibly related to accidental injection into dermis, and 6 cases of dysphagia in DCA participants attributed to higher volume treatment sessions and postinjection swelling. Dysphagia lasted a median of 2.5 days and resolved without sequelae.13
Overall, DCA demonstrated high rates of minor injection-site AEs that resolved without sequelae and could be mitigated by comfort therapies (eg, lidocaine, nonsteroidal anti-inflammatory drugs) as well as understanding the anatomy of the submental region. Adverse effects of particular interest included marginal mandibular nerve palsy, skin ulceration, and dysphagia.12,13
Cryolipolysis
Cryolipolysis is an advancement that utilizes the application of noninvasive cooling temperatures to the skin’s surface to destroy underlying adipocytes based on the concept that lipid-filled cells are more susceptible to cold-induced injury than water-filled cells. Thus, cryolipolysis selectively targets adipose tissue, leading to cell death without harm to surrounding cells and without the need for surgery or injections.14
Cryolipolysis typically is delivered via a vacuum applicator (CoolMini, Zeltiq Aesthetics Inc), which applies temperatures of –10°C (14°F) to the skin in cycles of 60 minutes each. Initially approved by the FDA for treatment of flank adiposity in 2010, cryolipolysis has since been approved for treatment of the abdomen, thighs, and submental area.14 An advantage of cryolipolysis is that it does not require frequent treatment sessions for maximal efficacy.
Efficacy
The efficacy of cryolipolysis in the treatment of SMF was established in a multicenter device investigation resulting in its FDA approval for the submental region.15 Sixty participants with a mean BMI of 31.8 kg/m2 received 1 (1/60) or 2 (59/60) treatment sessions of the submental area administered 6 weeks apart. Primary efficacy assessments included analysis by 3 blinded reviewers who viewed photographs of each participant at baseline, immediately posttreatment, 6 weeks posttreatment, and 12 weeks posttreatment; ultrasonographic measurements of SMF thickness; and a 12-point patient satisfaction questionnaire. Blinded reviewers correctly identified baseline images in 91.4% (55/60) of cases. Ultrasonography confirmed a mean reduction in SMF of 2 mm (P<.0001) or 20% of fat thickness at 12 weeks posttreatment. On subjective patient satisfaction surveys, 83% (50/60) of participants were satisfied with the procedure and 77% (46/60) reported a visible reduction in fat and perceived an improvement in appearance.15
Safety
The most common immediate posttreatment AEs were erythema/purpura (100%), numbness (90%), edema (62%), tingling (30%), blanching (25%), and bruising (3%) at the site of cryolipolysis with resolution within 1 week posttreatment, except for numbness.15 At 6-week follow-up, all AEs had resolved, except continued numbness in 4 participants that resolved by 12-week follow-up. A further event of note was fullness in the throat in 1 participant that was attributed to swelling and resolved at 40 days posttreatment without incident. No serious AEs were reported in this trial.15
A particularly concerning but rare complication that is increasing in awareness is paradoxical adipose hyperplasia following cryolipolysis. Patients may develop firm painless areas of soft tissue enlargements in the area of cryolipolysis typically 3 to 6 months posttreatment.16 The largest published report recorded an incidence rate of 0.46% (n=2, all males) at a single-center institution of 422 cryolipolysis treatments.16 Other incidence rates reported are 0.0051% and 0.78%.17 Causes and associations are not known, though male gender is speculated to increase risk.
Conclusion
This article highlights the available information on advances in minimally invasive and noninvasive treatments for SMF accumulation. The efficacy and safety trials varied in quality and in different methods of end point analysis of SMF reduction. Further, few trials have featured head-to-head comparisons of treatments.
Although liposuction and adjuncts remain the gold standard in large-mass lipid removal, these procedures are invasive and exhibit typical risks of surgery. Given its sensitive location, the submental area may require the use of more delicate therapeutic methods, including completely noninvasive devices such as truSculpt and cryolipolysis. Regardless of the chosen treatment, the most important factors in yielding patient satisfaction and SMF improvement are proper patient selection and an understanding of the anatomical source of adiposity to be addressed with the therapeutic modalities.
[polldaddy:9711250]
- Hatef DA, Koshy JC, Sandoval SE, et al. The submental fat compartment of the neck. Semin Plast Surg. 2009;23:288-291.
- Mulholland RS. Nonexcisional, minimally invasive rejuvenation of the neck. Clin Plast Surg. 2014;41:11-31.
- Fakhouri TM, El Tal AK, Abrou AE, et al. Laser-assisted lipolysis: a review. Dermatol Surg. 2012;38:155-169.
- Prado A, Andrades P, Danilla S, et al. A prospective, randomized, double-blind, controlled clinical trial comparing laser-assisted lipoplasty with suction-assisted lipoplasty. Plast Reconstr Surg. 2006;118:1032-1045.
- Valizadeh N, Jalaly NY, Zarghampour M, et al. Evaluation of safety and efficacy of 980-nm diode laser-assisted lipolysis versus traditional liposuction for submental rejuvenation: a randomized clinical trial. J Cosmet Laser Ther. 2016;18:41-45.
- Katz B, McBean J. Laser-assisted lipolysis: a report on complications. J Cosmet Laser Ther. 2008;10:231-233.
- Keramidas E, Rodopoulou S. Radiofrequency-assisted liposuction for neck and lower face adipodermal remodeling and contouring. Plast Reconstr Surg Glob Open. 2016;4:e850.
- Park JH, Kim JI, Park HJ, et al. Evaluation of safety and efficacy of noninvasive radiofrequency technology for submental rejuvenation [published online July 12, 2016]. Lasers Med Sci. 2016;31:1599-1605.
- Yagima Odo ME, Cucé LC, Odo LM, et al. Action of sodium deoxycholate on subcutaneous human tissue: local and systemic effects. Dermatol Surg. 2007;33:178-188; discussion 188-189.
- Rotunda AM, Suzuki H, Moy RL, et al. Detergent effects of sodium deoxycholate are a major feature of an injectable phosphatidylcholine formulation used for localized fat dissolution. Dermatol Surg. 2004;30:1001-1008.
- Kybella [package insert]. Westlake Village, CA: Kythera Biopharmaceuticals, Inc; 2015.
- Jones DH, Carruthers J, Joseph JH, et al. REFINE-1, a multicenter, randomized, double-blind, placebo-controlled, phase 3 trial with ATX-101, an injectable drug for submental fat reduction. Dermatol Surg. 2016;42:38-49.
- Humphrey S, Sykes J, Kantor J, et al. ATX-101 for reduction of submental fat: a phase III randomized controlled trial [published online July 16, 2016]. J Am Acad Dermatol. 2016;75:788-797.e7.
- Manstein D, Laubach H, Watanabe K, et al. Selective cryolysis: a novel method of non-invasive fat removal. Lasers Surg Med. 2008;40:595-604.
- Kilmer SL, Burns AJ, Zelickson BD. Safety and efficacy of cryolipolysis for non-invasive reduction of submental fat. Lasers Surg Med. 2016;48:3-13.
- Singh SM, Geddes ER, Boutrous SG, et al. Paradoxical adipose hyperplasia secondary to cryolipolysis: an underreported entity? Lasers Surg Med. 2015;47:476-478.
- Kelly E, Rodriguez-Feliz J, Kelly ME. Paradoxical adipose hyperplasia after cryolipolysis: a report on incidence and common factors identified in 510 patients. Plast Reconst Surg. 2016;137:639e-640e.
- Hatef DA, Koshy JC, Sandoval SE, et al. The submental fat compartment of the neck. Semin Plast Surg. 2009;23:288-291.
- Mulholland RS. Nonexcisional, minimally invasive rejuvenation of the neck. Clin Plast Surg. 2014;41:11-31.
- Fakhouri TM, El Tal AK, Abrou AE, et al. Laser-assisted lipolysis: a review. Dermatol Surg. 2012;38:155-169.
- Prado A, Andrades P, Danilla S, et al. A prospective, randomized, double-blind, controlled clinical trial comparing laser-assisted lipoplasty with suction-assisted lipoplasty. Plast Reconstr Surg. 2006;118:1032-1045.
- Valizadeh N, Jalaly NY, Zarghampour M, et al. Evaluation of safety and efficacy of 980-nm diode laser-assisted lipolysis versus traditional liposuction for submental rejuvenation: a randomized clinical trial. J Cosmet Laser Ther. 2016;18:41-45.
- Katz B, McBean J. Laser-assisted lipolysis: a report on complications. J Cosmet Laser Ther. 2008;10:231-233.
- Keramidas E, Rodopoulou S. Radiofrequency-assisted liposuction for neck and lower face adipodermal remodeling and contouring. Plast Reconstr Surg Glob Open. 2016;4:e850.
- Park JH, Kim JI, Park HJ, et al. Evaluation of safety and efficacy of noninvasive radiofrequency technology for submental rejuvenation [published online July 12, 2016]. Lasers Med Sci. 2016;31:1599-1605.
- Yagima Odo ME, Cucé LC, Odo LM, et al. Action of sodium deoxycholate on subcutaneous human tissue: local and systemic effects. Dermatol Surg. 2007;33:178-188; discussion 188-189.
- Rotunda AM, Suzuki H, Moy RL, et al. Detergent effects of sodium deoxycholate are a major feature of an injectable phosphatidylcholine formulation used for localized fat dissolution. Dermatol Surg. 2004;30:1001-1008.
- Kybella [package insert]. Westlake Village, CA: Kythera Biopharmaceuticals, Inc; 2015.
- Jones DH, Carruthers J, Joseph JH, et al. REFINE-1, a multicenter, randomized, double-blind, placebo-controlled, phase 3 trial with ATX-101, an injectable drug for submental fat reduction. Dermatol Surg. 2016;42:38-49.
- Humphrey S, Sykes J, Kantor J, et al. ATX-101 for reduction of submental fat: a phase III randomized controlled trial [published online July 16, 2016]. J Am Acad Dermatol. 2016;75:788-797.e7.
- Manstein D, Laubach H, Watanabe K, et al. Selective cryolysis: a novel method of non-invasive fat removal. Lasers Surg Med. 2008;40:595-604.
- Kilmer SL, Burns AJ, Zelickson BD. Safety and efficacy of cryolipolysis for non-invasive reduction of submental fat. Lasers Surg Med. 2016;48:3-13.
- Singh SM, Geddes ER, Boutrous SG, et al. Paradoxical adipose hyperplasia secondary to cryolipolysis: an underreported entity? Lasers Surg Med. 2015;47:476-478.
- Kelly E, Rodriguez-Feliz J, Kelly ME. Paradoxical adipose hyperplasia after cryolipolysis: a report on incidence and common factors identified in 510 patients. Plast Reconst Surg. 2016;137:639e-640e.
Practice Points
- New developments in minimally invasive techniques for treating submental adiposity include laser-assisted and radiofrequency-assisted lipoplasty with demonstrated clinical benefit and acceptable safety.
- Noninvasive treatments for submental adiposity include radiofrequency-assisted contouring devices, deoxycholic acid, and cryolipolysis, which offer an alternative to more invasive procedures such as lipoplasty.
- There are no comparative studies to date to suggest noninferiority of these noninvasive treatments compared to lipoplasty.
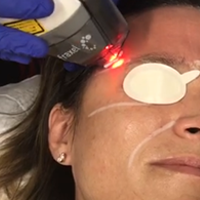
Facial Rejuvenation With Fractional Laser Resurfacing



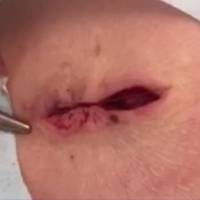
Pulley Suture for Wound Closure



Cosmetic Corner: Dermatologists Weigh in on Self-tanners
To improve patient care and outcomes, leading dermatologists offered their recommendations on self-tanners. Consideration must be given to:
- Anthelios 50 Mineral Tinted
La Roche-Posay Laboratoire Dermatologique
Recommended by Gary Goldenberg, MD, New York, New York
- St. Tropez Self Tan products
PZ Cussons Beauty LLP
“It helps to produce an even and natural-looking skin tone.”—Anthony M. Rossi, MD, New York, New York
- Sun-Free Self-Tanning Formula
Kiehl’s
Recommended by Gary Goldenberg, MD, New York, New York
- Sunless Tanning Towelette
Sun Bum
“This product is easy to use. Make sure to use it in conjunction with a broad-spectrum sunscreen.”—Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, skin-lightening products, and athlete’s foot treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on self-tanners. Consideration must be given to:
- Anthelios 50 Mineral Tinted
La Roche-Posay Laboratoire Dermatologique
Recommended by Gary Goldenberg, MD, New York, New York
- St. Tropez Self Tan products
PZ Cussons Beauty LLP
“It helps to produce an even and natural-looking skin tone.”—Anthony M. Rossi, MD, New York, New York
- Sun-Free Self-Tanning Formula
Kiehl’s
Recommended by Gary Goldenberg, MD, New York, New York
- Sunless Tanning Towelette
Sun Bum
“This product is easy to use. Make sure to use it in conjunction with a broad-spectrum sunscreen.”—Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, skin-lightening products, and athlete’s foot treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on self-tanners. Consideration must be given to:
- Anthelios 50 Mineral Tinted
La Roche-Posay Laboratoire Dermatologique
Recommended by Gary Goldenberg, MD, New York, New York
- St. Tropez Self Tan products
PZ Cussons Beauty LLP
“It helps to produce an even and natural-looking skin tone.”—Anthony M. Rossi, MD, New York, New York
- Sun-Free Self-Tanning Formula
Kiehl’s
Recommended by Gary Goldenberg, MD, New York, New York
- Sunless Tanning Towelette
Sun Bum
“This product is easy to use. Make sure to use it in conjunction with a broad-spectrum sunscreen.”—Shari Lipner, MD, PhD, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, skin-lightening products, and athlete’s foot treatments will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
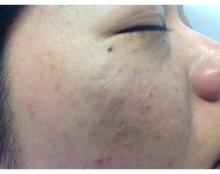
The hidden vascular component to melasma
So lets revisit this topic. We hate melasma. It is recalcitrant, resistant, and often recurs. But how many of us dermatologists look at melasma with a dermatoscope? A minority? And probably even fewer of us biopsy melasma. Are we missing the many, many cases of vascular or erythematotelangiectatic melasma and just treating melasma as a pigment problem instead of treating it as a vascular problem?
We know that melasma develops because of increased melanin production, not an increased number of melanocytes, but the underlying cause of increased melanogenesis is not fully understood. Several recent studies suggest that the increased vascularity in melasma skin is the underlying etiology. Understanding the way endogeneous and exogeneous stimuli such as sex hormones, oral contraceptives, ultraviolet irradiation, and visible light stimulate inflammation in the dermis, leading to the release of various mediators that stimulate angiogenesis and the activation of melanocytes, will help us improve the treatment of this relentless disease.
Similarly, elevated estrogen and progesterone in pregnancy or with oral contraceptive use is known to stimulate melasma. Melanocytes express estrogen receptors and estradiol increases the level of TRP-2, which stimulates melanocytes to produce melanin. Recent literature has shown that the number of blood vessels, vessel size, and vessel density also are greater in lesional melasma skin than in perilesional skin. In addition, immunohistochemical staining has shown an increased level of factor VIIIa-related antigen in blood vessels in melasma skin, compared with perilesional normal skin.
Unfortunately, the treatment of vascular melasma is very difficult. Lasers such as the pulsed dye laser that help skin vascularity can trigger worsening melanogenesis through dermal inflammation. The melanin cap overlying the melanocyte nucleus also can mask the underlying vascularity and make laser treatments more difficult. The isolated treatment of epidermal pigment also may be ineffective and transient.
By targeting the vessels in addition to the pigment, we will get improved clinical results and fewer relapses. We suggest that melasma be treated conservatively, not aggressively. It should be treated as an inflammatory process. Patients with melasma also have a slightly abnormal skin barrier, so we should be hesitant in using deep lasers, radio frequency, and aggressive chemical peels. Topical preparations – particularly triple-combination bleaching agents, retinoids, and nonhydroquinone skin lighteners – should be used sparingly and always in combination with treatments targeting skin vascularity.
References
J Dermatol Sci. 2007 May;46(2):111-6.
Exp Dermatol. 2005 Aug;14(8):625-33.
Clin Exp Dermatol. 2008;33(3):305-8.
J Eur Acad Dermatol Venereol. 2009 Nov;23(11):1254-62.
Ann Dermatol. 2010 Nov;22(4):373-8.
J Eur Acad Dermatol Venereol. 2013 Jan;27 Suppl 1:5-6.
Am J Clin Dermatol. 2013 Oct;14(5):359-76.
J Am Acad Dermatol. 2014 Feb;70(2):369-73.
Dr. Talakoub and Dr. Wesley and are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected].
So lets revisit this topic. We hate melasma. It is recalcitrant, resistant, and often recurs. But how many of us dermatologists look at melasma with a dermatoscope? A minority? And probably even fewer of us biopsy melasma. Are we missing the many, many cases of vascular or erythematotelangiectatic melasma and just treating melasma as a pigment problem instead of treating it as a vascular problem?
We know that melasma develops because of increased melanin production, not an increased number of melanocytes, but the underlying cause of increased melanogenesis is not fully understood. Several recent studies suggest that the increased vascularity in melasma skin is the underlying etiology. Understanding the way endogeneous and exogeneous stimuli such as sex hormones, oral contraceptives, ultraviolet irradiation, and visible light stimulate inflammation in the dermis, leading to the release of various mediators that stimulate angiogenesis and the activation of melanocytes, will help us improve the treatment of this relentless disease.
Similarly, elevated estrogen and progesterone in pregnancy or with oral contraceptive use is known to stimulate melasma. Melanocytes express estrogen receptors and estradiol increases the level of TRP-2, which stimulates melanocytes to produce melanin. Recent literature has shown that the number of blood vessels, vessel size, and vessel density also are greater in lesional melasma skin than in perilesional skin. In addition, immunohistochemical staining has shown an increased level of factor VIIIa-related antigen in blood vessels in melasma skin, compared with perilesional normal skin.
Unfortunately, the treatment of vascular melasma is very difficult. Lasers such as the pulsed dye laser that help skin vascularity can trigger worsening melanogenesis through dermal inflammation. The melanin cap overlying the melanocyte nucleus also can mask the underlying vascularity and make laser treatments more difficult. The isolated treatment of epidermal pigment also may be ineffective and transient.
By targeting the vessels in addition to the pigment, we will get improved clinical results and fewer relapses. We suggest that melasma be treated conservatively, not aggressively. It should be treated as an inflammatory process. Patients with melasma also have a slightly abnormal skin barrier, so we should be hesitant in using deep lasers, radio frequency, and aggressive chemical peels. Topical preparations – particularly triple-combination bleaching agents, retinoids, and nonhydroquinone skin lighteners – should be used sparingly and always in combination with treatments targeting skin vascularity.
References
J Dermatol Sci. 2007 May;46(2):111-6.
Exp Dermatol. 2005 Aug;14(8):625-33.
Clin Exp Dermatol. 2008;33(3):305-8.
J Eur Acad Dermatol Venereol. 2009 Nov;23(11):1254-62.
Ann Dermatol. 2010 Nov;22(4):373-8.
J Eur Acad Dermatol Venereol. 2013 Jan;27 Suppl 1:5-6.
Am J Clin Dermatol. 2013 Oct;14(5):359-76.
J Am Acad Dermatol. 2014 Feb;70(2):369-73.
Dr. Talakoub and Dr. Wesley and are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected].
So lets revisit this topic. We hate melasma. It is recalcitrant, resistant, and often recurs. But how many of us dermatologists look at melasma with a dermatoscope? A minority? And probably even fewer of us biopsy melasma. Are we missing the many, many cases of vascular or erythematotelangiectatic melasma and just treating melasma as a pigment problem instead of treating it as a vascular problem?
We know that melasma develops because of increased melanin production, not an increased number of melanocytes, but the underlying cause of increased melanogenesis is not fully understood. Several recent studies suggest that the increased vascularity in melasma skin is the underlying etiology. Understanding the way endogeneous and exogeneous stimuli such as sex hormones, oral contraceptives, ultraviolet irradiation, and visible light stimulate inflammation in the dermis, leading to the release of various mediators that stimulate angiogenesis and the activation of melanocytes, will help us improve the treatment of this relentless disease.
Similarly, elevated estrogen and progesterone in pregnancy or with oral contraceptive use is known to stimulate melasma. Melanocytes express estrogen receptors and estradiol increases the level of TRP-2, which stimulates melanocytes to produce melanin. Recent literature has shown that the number of blood vessels, vessel size, and vessel density also are greater in lesional melasma skin than in perilesional skin. In addition, immunohistochemical staining has shown an increased level of factor VIIIa-related antigen in blood vessels in melasma skin, compared with perilesional normal skin.
Unfortunately, the treatment of vascular melasma is very difficult. Lasers such as the pulsed dye laser that help skin vascularity can trigger worsening melanogenesis through dermal inflammation. The melanin cap overlying the melanocyte nucleus also can mask the underlying vascularity and make laser treatments more difficult. The isolated treatment of epidermal pigment also may be ineffective and transient.
By targeting the vessels in addition to the pigment, we will get improved clinical results and fewer relapses. We suggest that melasma be treated conservatively, not aggressively. It should be treated as an inflammatory process. Patients with melasma also have a slightly abnormal skin barrier, so we should be hesitant in using deep lasers, radio frequency, and aggressive chemical peels. Topical preparations – particularly triple-combination bleaching agents, retinoids, and nonhydroquinone skin lighteners – should be used sparingly and always in combination with treatments targeting skin vascularity.
References
J Dermatol Sci. 2007 May;46(2):111-6.
Exp Dermatol. 2005 Aug;14(8):625-33.
Clin Exp Dermatol. 2008;33(3):305-8.
J Eur Acad Dermatol Venereol. 2009 Nov;23(11):1254-62.
Ann Dermatol. 2010 Nov;22(4):373-8.
J Eur Acad Dermatol Venereol. 2013 Jan;27 Suppl 1:5-6.
Am J Clin Dermatol. 2013 Oct;14(5):359-76.
J Am Acad Dermatol. 2014 Feb;70(2):369-73.
Dr. Talakoub and Dr. Wesley and are co-contributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected].
Rosacea improved with fractional microneedling radiofrequency therapy
Fractional microneedling radiofrequency (FMR) therapy resulted in modest but clinically significant improvements in the appearance and inflammation of rosacea in a small study of patients with mild to moderate rosacea.
The treatment delivers bipolar radiofrequency energy to the dermis via an array of microneedles, without damaging the epidermis, noted Seon Yong Park, MD, and colleagues from the department of dermatology at Seoul (South Korea) National University. It has previously been associated with clinical and histological improvements in acne-associated postinflammatory erythema and is used in the treatment of cutaneous wrinkles. The authors said that, as far as they know, this is the first study to evaluate the use of FMR in patients with rosacea.
In the prospective, single-blind, randomized, split-face clinical study, 21 patients (20 females, 1 male) with mild to moderate rosacea were treated with two FMR sessions, 4 weeks apart, then assessed 4, 8, and 12 weeks after the second session. The mean age of the patients was 43 years; they had Fitzpatrick skin type III (13 patients) or IV (8 patients), and rosacea was considered mild in 12 patients and moderate in 9 patients at baseline.
Researchers saw clinical improvements in 17 (81%) of the patients on the treated side; these patients had a mean improvement in the Investigator’s Global Assessment (IGA) score of 2.47 by week 12, representing about a 20% improvement (Dermatol Surg. 2016 Dec;42[12]:1362-9).
In the group overall, mean IGA scores at weeks 4, 8, and 12 were 1.05, 1.57, and 2.00 for the treated side, compared with 0.29, 0.38, and 0.38, respectively, for the untreated side, which, the authors wrote, indicated that “there was modest but statistically significant improvements in the treated side.”
Photometric measurements of redness showed significant reductions on the treated side, compared with the untreated side and baseline, with reductions in the erythema index of 11.9%, 10.7%, and 13.6% at week 4, 8, and 12, respectively.
Histological assessment showed reduced dermal inflammation, significant reductions in average mast cell count, and an overall decrease in immunohistochemical intensity in the treated skin 8 weeks after treatment. Similarly, there were significant decreases in markers of angiogenesis, inflammation, innate immunity, and neuroimmunity on the treated side, compared with baseline.
“Fractional microneedling radiofrequency was slightly more effective in reducing erythema in patients with PPR [papulopustular rosacea] than in those with ETR [erythematotelangiectatic rosacea], suggesting that inflammatory lesions, such as papules and pustules, could be more effectively treated with this device,” the authors wrote. “This result agreed with reports showing that FMR is effective in treating inflammatory acne.”
No serious adverse effects were reported, although 19 patients (90.5%) experienced mild pain during the procedure and 17 (81%) had mild erythema that lasted for up to 5 days. Patients also reported less itching, heat, burning, or pricking on the treated side, which showed that the treatment was effective in controlling the symptoms of rosacea, Dr. Park and associates said.
The study was supported by the SNUH Research Fund and National Research Foundation of Korea. The authors, who are also in the acne and rosacea research laboratory, Seoul National University Hospital, had no conflicts to disclose.
Fractional microneedling radiofrequency (FMR) therapy resulted in modest but clinically significant improvements in the appearance and inflammation of rosacea in a small study of patients with mild to moderate rosacea.
The treatment delivers bipolar radiofrequency energy to the dermis via an array of microneedles, without damaging the epidermis, noted Seon Yong Park, MD, and colleagues from the department of dermatology at Seoul (South Korea) National University. It has previously been associated with clinical and histological improvements in acne-associated postinflammatory erythema and is used in the treatment of cutaneous wrinkles. The authors said that, as far as they know, this is the first study to evaluate the use of FMR in patients with rosacea.
In the prospective, single-blind, randomized, split-face clinical study, 21 patients (20 females, 1 male) with mild to moderate rosacea were treated with two FMR sessions, 4 weeks apart, then assessed 4, 8, and 12 weeks after the second session. The mean age of the patients was 43 years; they had Fitzpatrick skin type III (13 patients) or IV (8 patients), and rosacea was considered mild in 12 patients and moderate in 9 patients at baseline.
Researchers saw clinical improvements in 17 (81%) of the patients on the treated side; these patients had a mean improvement in the Investigator’s Global Assessment (IGA) score of 2.47 by week 12, representing about a 20% improvement (Dermatol Surg. 2016 Dec;42[12]:1362-9).
In the group overall, mean IGA scores at weeks 4, 8, and 12 were 1.05, 1.57, and 2.00 for the treated side, compared with 0.29, 0.38, and 0.38, respectively, for the untreated side, which, the authors wrote, indicated that “there was modest but statistically significant improvements in the treated side.”
Photometric measurements of redness showed significant reductions on the treated side, compared with the untreated side and baseline, with reductions in the erythema index of 11.9%, 10.7%, and 13.6% at week 4, 8, and 12, respectively.
Histological assessment showed reduced dermal inflammation, significant reductions in average mast cell count, and an overall decrease in immunohistochemical intensity in the treated skin 8 weeks after treatment. Similarly, there were significant decreases in markers of angiogenesis, inflammation, innate immunity, and neuroimmunity on the treated side, compared with baseline.
“Fractional microneedling radiofrequency was slightly more effective in reducing erythema in patients with PPR [papulopustular rosacea] than in those with ETR [erythematotelangiectatic rosacea], suggesting that inflammatory lesions, such as papules and pustules, could be more effectively treated with this device,” the authors wrote. “This result agreed with reports showing that FMR is effective in treating inflammatory acne.”
No serious adverse effects were reported, although 19 patients (90.5%) experienced mild pain during the procedure and 17 (81%) had mild erythema that lasted for up to 5 days. Patients also reported less itching, heat, burning, or pricking on the treated side, which showed that the treatment was effective in controlling the symptoms of rosacea, Dr. Park and associates said.
The study was supported by the SNUH Research Fund and National Research Foundation of Korea. The authors, who are also in the acne and rosacea research laboratory, Seoul National University Hospital, had no conflicts to disclose.
Fractional microneedling radiofrequency (FMR) therapy resulted in modest but clinically significant improvements in the appearance and inflammation of rosacea in a small study of patients with mild to moderate rosacea.
The treatment delivers bipolar radiofrequency energy to the dermis via an array of microneedles, without damaging the epidermis, noted Seon Yong Park, MD, and colleagues from the department of dermatology at Seoul (South Korea) National University. It has previously been associated with clinical and histological improvements in acne-associated postinflammatory erythema and is used in the treatment of cutaneous wrinkles. The authors said that, as far as they know, this is the first study to evaluate the use of FMR in patients with rosacea.
In the prospective, single-blind, randomized, split-face clinical study, 21 patients (20 females, 1 male) with mild to moderate rosacea were treated with two FMR sessions, 4 weeks apart, then assessed 4, 8, and 12 weeks after the second session. The mean age of the patients was 43 years; they had Fitzpatrick skin type III (13 patients) or IV (8 patients), and rosacea was considered mild in 12 patients and moderate in 9 patients at baseline.
Researchers saw clinical improvements in 17 (81%) of the patients on the treated side; these patients had a mean improvement in the Investigator’s Global Assessment (IGA) score of 2.47 by week 12, representing about a 20% improvement (Dermatol Surg. 2016 Dec;42[12]:1362-9).
In the group overall, mean IGA scores at weeks 4, 8, and 12 were 1.05, 1.57, and 2.00 for the treated side, compared with 0.29, 0.38, and 0.38, respectively, for the untreated side, which, the authors wrote, indicated that “there was modest but statistically significant improvements in the treated side.”
Photometric measurements of redness showed significant reductions on the treated side, compared with the untreated side and baseline, with reductions in the erythema index of 11.9%, 10.7%, and 13.6% at week 4, 8, and 12, respectively.
Histological assessment showed reduced dermal inflammation, significant reductions in average mast cell count, and an overall decrease in immunohistochemical intensity in the treated skin 8 weeks after treatment. Similarly, there were significant decreases in markers of angiogenesis, inflammation, innate immunity, and neuroimmunity on the treated side, compared with baseline.
“Fractional microneedling radiofrequency was slightly more effective in reducing erythema in patients with PPR [papulopustular rosacea] than in those with ETR [erythematotelangiectatic rosacea], suggesting that inflammatory lesions, such as papules and pustules, could be more effectively treated with this device,” the authors wrote. “This result agreed with reports showing that FMR is effective in treating inflammatory acne.”
No serious adverse effects were reported, although 19 patients (90.5%) experienced mild pain during the procedure and 17 (81%) had mild erythema that lasted for up to 5 days. Patients also reported less itching, heat, burning, or pricking on the treated side, which showed that the treatment was effective in controlling the symptoms of rosacea, Dr. Park and associates said.
The study was supported by the SNUH Research Fund and National Research Foundation of Korea. The authors, who are also in the acne and rosacea research laboratory, Seoul National University Hospital, had no conflicts to disclose.
FROM DERMATOLOGIC SURGERY
Key clinical point: Fractional microneedling radiofrequency therapy may achieve modest but clinically significant improvements in the appearance and inflammation of rosacea.
Major finding: Fractional microneedling radiofrequency therapy was associated with clinical improvements in 81% of treated patients.
Data source: A prospective, single-blind, randomized, split-face clinical trial in 21 patients with mild to moderate rosacea.
Disclosures: The study was supported by the SNUH Research Fund and National Research Foundation of Korea. No conflicts of interest were declared.
Thyme
Native to the western Mediterranean, Thymus vulgaris (one of approximately 300 Thymus species) is a small bush used for centuries as a spice and in medicine, particularly to treat bronchitis.1Thymus species are among the wild and cultivated species used in traditional medicine in Bosnia and Herzegovina for various indications, including skin disorders.2 Thyme essential oil is a natural compound generally recognized as safe by the Food and Drug Administration, with demonstrated antibacterial, antifungal, and antispasmodic activities.3,4 Several other biologic activities have been associated with the polyphenol-rich herb, many of which have dermatologic implications. Notably, the essential oil of thyme and thymol, a key constituent of thyme, are known to act as skin sensitizers and allergens.5
Photoprotective activity
Recently, Sun et al. showed that UVB-induced skin damage was attenuated by treating hairless mice (HR-1) with T. vulgaris, as indicated by reduced matrix metalloproteinases and elevated collagen synthesis. In cultured normal human dermal fibroblasts, the investigators found that T. vulgaris blocked UVB-induced reactive oxygen species and lactate dehydrogenase, and dose-dependently yielded increases in glutathione, NAD(P)H: quinone oxidoreductase 1, and heme oxygenase-1. Further, the botanical significantly reduced UVB-induced phosphorylation of mitogen-activated protein kinases. The investigators concluded that T. vulgaris has potential for use in preventing skin damage caused by UV radiation–induced oxidative stress.6
Thyme also was demonstrated by Cornaghi et al. in 2016 to exert a protective effect on normal human skin explants obtained from seven young healthy women that were treated 1 hour before UVB irradiation.7
In 2015, Calò et al. evaluated the protective effects of a dry extract from T. vulgaris and its primary synthetic constituent thymol against UVA- and UVB-induced oxidative and genotoxic damage in the keratinocyte cell line NCTC 2544. Both thymol and T. vulgaris suppressed reactive oxygen species production in UVA- and UVB-treated cells, but lowered malondialdehyde synthesis only in cells treated with UVA.8
Antioxidant activity
In 2007, Wei and Shibamoto reported that thyme essential oil mixed with clove oil exhibited over a 90% inhibitory effect against the formation of malondialdehyde. They speculated that the presence of thymol and eugenol might account for the strong antioxidant activity displayed by the thyme/clove leaf combination.9 The investigators previously observed antioxidant activities exhibited by volatile extracts isolated from thyme (as well as various other herbs and spices) using aldehyde/carboxylic acid as well as conjugated diene assays.10 The antioxidant activity of thyme also was demonstrated by Miura et al. using the oil stability index method.11
Antimicrobial activity
In 2011, Sienkiewicz et al. reported that the oil of T. vulgaris displayed potent activity against clinical bacterial strains of Staphylococcus, Enterococcus, Escherichia, and Pseudomonas genera. In addition, thyme essential oil exhibited efficacy against tested antibiotic-resistant strains of bacteria.12 The following year, Sienkiewicz et al. assessed the antimicrobial activity of thyme essential oil against clinical multidrug-resistant strains of Staphylococcus, Enterococcus, Escherichia, and Pseudomonas, finding that it potently suppressed the growth of each.13
Potential cutaneous indications: atopic dermatitis, leishmaniasis, eczema, hair growth
In 2015, Seo and Jeong showed that lavender oil, thyme oil, and a blend of the two were all effective in reducing the symptoms of atopic dermatitis in mice. The researchers suggested that developing treatments with these oils for human patients with atopic dermatitis is warranted.14
Nilforoushzadeh et al. found in 2008 that herbal extracts of T. vulgaris and Achillea millefolium (yarrow), as well as propolis hydroalcoholic extracts, were effective in treating cutaneous leishmaniasis in mice and recommended the study of these extracts alone or in combination in human trials.15
A two-arm, randomized, double-blind, placebo-controlled trial conducted by Shimelis et al. in 2012 evaluated the efficacy of a 3% thyme essential oil antifungal cream and a 10% chamomile extract cream in the treatment of eczemalike lesions. Complete healing was achieved in 10 patients (66.5%) treated with the thyme cream, compared with four patients (28.5%) in the placebo group. Although no significant differences were observed between the active chamomile group and placebo, an appreciable number of subjects improved or healed. The investigators concluded that their findings from this small study suggest that, while more research is needed, a 3% thyme essential oil cream appears to be an inexpensive and readily available option to treat mild to moderate cutaneous conditions, including fungal infections, pityriasis alba, and eczema.16
In 2013, Rastegar et al. found that the combination of herbal extracts (including thyme) and platelet-rich plasma induced significant proliferation of human dermal papilla cells by regulating extracellular signal-regulated kinase (ERK) and Akt (protein kinase B). They concluded that their findings suggest the potential for developing combination therapies intended to improve hair growth.17
Insect repellent activity
In a 2016 study by Gutiérrez et al., the essential oil of T. vulgaris was found to be effective against Pediculus humanus capitis (head lice) adults and eggs. The researchers concluded that T. vulgaris achieves a strong knockdown and mortality rate in adult head lice and toxicity in the eggs after 21 minutes of application at a low concentration.18
In a small 1999 study by Barnard of the repellency to Aedes aegypti and Anopheles albimanus of various concentrations and combinations of five essential oils (Bourbon geranium, cedarwood, clove, peppermint, and thyme) applied to human skin, thyme and clove oils were found to be the most effective mosquito repellents. The author noted that thyme oil (as well as clove and peppermint oils) can irritate the skin and the odor of thyme and clove oils, at concentrations of 25%, or more were deemed unacceptable by the two participants in the study.19
Three years later, Choi et al. found that the essential oil of T. vulgaris also repelled adult mosquitoes (Culex pipiens pallens) on hairless mice and displayed potent repellent activity.20
Melanoma
In 2005, Carrera et al. reported a case of long-term complete remission of cutaneous melanoma metastases in a 73-year-old white woman who consumed a dried thyme herbal tea and thyme topical applications in compresses.4 An association between thyme and melanoma has not been reported in the subsequent literature.
Conclusion
Thyme has a long history of culinary and medical uses. Its antimicrobial and antioxidant activity are well documented. While there is reason to consider the potential applications of thyme for dermatologic conditions, much more research is necessary to determine its viability for such purposes.
References
1. An Illustrated Guide to 101 Medicinal Herbs: Their History, Use, Recommended Dosages, and Cautions (Loveland, Colo.: Interweave Press, 1998, pp. 198-9).
2. J Ethnopharmacol. 2010 Aug 19;131(1):33-55.
3. J Food Sci. 2014 May;79(5):M903-10.
4. J Am Acad Dermatol. 2005 Apr;52(4):713-5.
5. Nat Neurosci. 2006 May;9(5):628-35.
6. J Cell Mol Med. 2016 Sep 19. doi: 10.1111/jcmm.12968. [Epub ahead of print]
7. Cells Tissues Organs. 2016;201(3):180-92.
8. Mutat Res Genet Toxicol Environ Mutagen. 2015 Sep;791:30-7.
9. Cutan Ocul Toxicol. 2007;26(3):227-33.
10. J Agric Food Chem. 2002 Aug 14;50(17):4947-52.
11. J Agric Food Chem. 2002 Mar 27;50(7):1845-51.
12. Med Chem. 2011 Nov;7(6):674-89.
13. Microb Drug Resist. 2012 Apr;18(2):137-48.
14. J Korean Acad Nurs. 2015 Jun;45(3):367-77.
15. J Vector Borne Dis. 2008 Dec;45(4):301-6.
16. Int J Dermatol. 2012 Jul;51(7):790-5.
17. J Cosmet Dermatol. 2013 Jun;12(2):116-22.
18. Parasitol Res. 2016 Feb;115(2):633-41.
19. J Med Entomol. 1999 Sep;36(5):625-9.
20. J Am Mosq Control Assoc. 2002 Dec;18(4):348-51.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Native to the western Mediterranean, Thymus vulgaris (one of approximately 300 Thymus species) is a small bush used for centuries as a spice and in medicine, particularly to treat bronchitis.1Thymus species are among the wild and cultivated species used in traditional medicine in Bosnia and Herzegovina for various indications, including skin disorders.2 Thyme essential oil is a natural compound generally recognized as safe by the Food and Drug Administration, with demonstrated antibacterial, antifungal, and antispasmodic activities.3,4 Several other biologic activities have been associated with the polyphenol-rich herb, many of which have dermatologic implications. Notably, the essential oil of thyme and thymol, a key constituent of thyme, are known to act as skin sensitizers and allergens.5
Photoprotective activity
Recently, Sun et al. showed that UVB-induced skin damage was attenuated by treating hairless mice (HR-1) with T. vulgaris, as indicated by reduced matrix metalloproteinases and elevated collagen synthesis. In cultured normal human dermal fibroblasts, the investigators found that T. vulgaris blocked UVB-induced reactive oxygen species and lactate dehydrogenase, and dose-dependently yielded increases in glutathione, NAD(P)H: quinone oxidoreductase 1, and heme oxygenase-1. Further, the botanical significantly reduced UVB-induced phosphorylation of mitogen-activated protein kinases. The investigators concluded that T. vulgaris has potential for use in preventing skin damage caused by UV radiation–induced oxidative stress.6
Thyme also was demonstrated by Cornaghi et al. in 2016 to exert a protective effect on normal human skin explants obtained from seven young healthy women that were treated 1 hour before UVB irradiation.7
In 2015, Calò et al. evaluated the protective effects of a dry extract from T. vulgaris and its primary synthetic constituent thymol against UVA- and UVB-induced oxidative and genotoxic damage in the keratinocyte cell line NCTC 2544. Both thymol and T. vulgaris suppressed reactive oxygen species production in UVA- and UVB-treated cells, but lowered malondialdehyde synthesis only in cells treated with UVA.8
Antioxidant activity
In 2007, Wei and Shibamoto reported that thyme essential oil mixed with clove oil exhibited over a 90% inhibitory effect against the formation of malondialdehyde. They speculated that the presence of thymol and eugenol might account for the strong antioxidant activity displayed by the thyme/clove leaf combination.9 The investigators previously observed antioxidant activities exhibited by volatile extracts isolated from thyme (as well as various other herbs and spices) using aldehyde/carboxylic acid as well as conjugated diene assays.10 The antioxidant activity of thyme also was demonstrated by Miura et al. using the oil stability index method.11
Antimicrobial activity
In 2011, Sienkiewicz et al. reported that the oil of T. vulgaris displayed potent activity against clinical bacterial strains of Staphylococcus, Enterococcus, Escherichia, and Pseudomonas genera. In addition, thyme essential oil exhibited efficacy against tested antibiotic-resistant strains of bacteria.12 The following year, Sienkiewicz et al. assessed the antimicrobial activity of thyme essential oil against clinical multidrug-resistant strains of Staphylococcus, Enterococcus, Escherichia, and Pseudomonas, finding that it potently suppressed the growth of each.13
Potential cutaneous indications: atopic dermatitis, leishmaniasis, eczema, hair growth
In 2015, Seo and Jeong showed that lavender oil, thyme oil, and a blend of the two were all effective in reducing the symptoms of atopic dermatitis in mice. The researchers suggested that developing treatments with these oils for human patients with atopic dermatitis is warranted.14
Nilforoushzadeh et al. found in 2008 that herbal extracts of T. vulgaris and Achillea millefolium (yarrow), as well as propolis hydroalcoholic extracts, were effective in treating cutaneous leishmaniasis in mice and recommended the study of these extracts alone or in combination in human trials.15
A two-arm, randomized, double-blind, placebo-controlled trial conducted by Shimelis et al. in 2012 evaluated the efficacy of a 3% thyme essential oil antifungal cream and a 10% chamomile extract cream in the treatment of eczemalike lesions. Complete healing was achieved in 10 patients (66.5%) treated with the thyme cream, compared with four patients (28.5%) in the placebo group. Although no significant differences were observed between the active chamomile group and placebo, an appreciable number of subjects improved or healed. The investigators concluded that their findings from this small study suggest that, while more research is needed, a 3% thyme essential oil cream appears to be an inexpensive and readily available option to treat mild to moderate cutaneous conditions, including fungal infections, pityriasis alba, and eczema.16
In 2013, Rastegar et al. found that the combination of herbal extracts (including thyme) and platelet-rich plasma induced significant proliferation of human dermal papilla cells by regulating extracellular signal-regulated kinase (ERK) and Akt (protein kinase B). They concluded that their findings suggest the potential for developing combination therapies intended to improve hair growth.17
Insect repellent activity
In a 2016 study by Gutiérrez et al., the essential oil of T. vulgaris was found to be effective against Pediculus humanus capitis (head lice) adults and eggs. The researchers concluded that T. vulgaris achieves a strong knockdown and mortality rate in adult head lice and toxicity in the eggs after 21 minutes of application at a low concentration.18
In a small 1999 study by Barnard of the repellency to Aedes aegypti and Anopheles albimanus of various concentrations and combinations of five essential oils (Bourbon geranium, cedarwood, clove, peppermint, and thyme) applied to human skin, thyme and clove oils were found to be the most effective mosquito repellents. The author noted that thyme oil (as well as clove and peppermint oils) can irritate the skin and the odor of thyme and clove oils, at concentrations of 25%, or more were deemed unacceptable by the two participants in the study.19
Three years later, Choi et al. found that the essential oil of T. vulgaris also repelled adult mosquitoes (Culex pipiens pallens) on hairless mice and displayed potent repellent activity.20
Melanoma
In 2005, Carrera et al. reported a case of long-term complete remission of cutaneous melanoma metastases in a 73-year-old white woman who consumed a dried thyme herbal tea and thyme topical applications in compresses.4 An association between thyme and melanoma has not been reported in the subsequent literature.
Conclusion
Thyme has a long history of culinary and medical uses. Its antimicrobial and antioxidant activity are well documented. While there is reason to consider the potential applications of thyme for dermatologic conditions, much more research is necessary to determine its viability for such purposes.
References
1. An Illustrated Guide to 101 Medicinal Herbs: Their History, Use, Recommended Dosages, and Cautions (Loveland, Colo.: Interweave Press, 1998, pp. 198-9).
2. J Ethnopharmacol. 2010 Aug 19;131(1):33-55.
3. J Food Sci. 2014 May;79(5):M903-10.
4. J Am Acad Dermatol. 2005 Apr;52(4):713-5.
5. Nat Neurosci. 2006 May;9(5):628-35.
6. J Cell Mol Med. 2016 Sep 19. doi: 10.1111/jcmm.12968. [Epub ahead of print]
7. Cells Tissues Organs. 2016;201(3):180-92.
8. Mutat Res Genet Toxicol Environ Mutagen. 2015 Sep;791:30-7.
9. Cutan Ocul Toxicol. 2007;26(3):227-33.
10. J Agric Food Chem. 2002 Aug 14;50(17):4947-52.
11. J Agric Food Chem. 2002 Mar 27;50(7):1845-51.
12. Med Chem. 2011 Nov;7(6):674-89.
13. Microb Drug Resist. 2012 Apr;18(2):137-48.
14. J Korean Acad Nurs. 2015 Jun;45(3):367-77.
15. J Vector Borne Dis. 2008 Dec;45(4):301-6.
16. Int J Dermatol. 2012 Jul;51(7):790-5.
17. J Cosmet Dermatol. 2013 Jun;12(2):116-22.
18. Parasitol Res. 2016 Feb;115(2):633-41.
19. J Med Entomol. 1999 Sep;36(5):625-9.
20. J Am Mosq Control Assoc. 2002 Dec;18(4):348-51.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Native to the western Mediterranean, Thymus vulgaris (one of approximately 300 Thymus species) is a small bush used for centuries as a spice and in medicine, particularly to treat bronchitis.1Thymus species are among the wild and cultivated species used in traditional medicine in Bosnia and Herzegovina for various indications, including skin disorders.2 Thyme essential oil is a natural compound generally recognized as safe by the Food and Drug Administration, with demonstrated antibacterial, antifungal, and antispasmodic activities.3,4 Several other biologic activities have been associated with the polyphenol-rich herb, many of which have dermatologic implications. Notably, the essential oil of thyme and thymol, a key constituent of thyme, are known to act as skin sensitizers and allergens.5
Photoprotective activity
Recently, Sun et al. showed that UVB-induced skin damage was attenuated by treating hairless mice (HR-1) with T. vulgaris, as indicated by reduced matrix metalloproteinases and elevated collagen synthesis. In cultured normal human dermal fibroblasts, the investigators found that T. vulgaris blocked UVB-induced reactive oxygen species and lactate dehydrogenase, and dose-dependently yielded increases in glutathione, NAD(P)H: quinone oxidoreductase 1, and heme oxygenase-1. Further, the botanical significantly reduced UVB-induced phosphorylation of mitogen-activated protein kinases. The investigators concluded that T. vulgaris has potential for use in preventing skin damage caused by UV radiation–induced oxidative stress.6
Thyme also was demonstrated by Cornaghi et al. in 2016 to exert a protective effect on normal human skin explants obtained from seven young healthy women that were treated 1 hour before UVB irradiation.7
In 2015, Calò et al. evaluated the protective effects of a dry extract from T. vulgaris and its primary synthetic constituent thymol against UVA- and UVB-induced oxidative and genotoxic damage in the keratinocyte cell line NCTC 2544. Both thymol and T. vulgaris suppressed reactive oxygen species production in UVA- and UVB-treated cells, but lowered malondialdehyde synthesis only in cells treated with UVA.8
Antioxidant activity
In 2007, Wei and Shibamoto reported that thyme essential oil mixed with clove oil exhibited over a 90% inhibitory effect against the formation of malondialdehyde. They speculated that the presence of thymol and eugenol might account for the strong antioxidant activity displayed by the thyme/clove leaf combination.9 The investigators previously observed antioxidant activities exhibited by volatile extracts isolated from thyme (as well as various other herbs and spices) using aldehyde/carboxylic acid as well as conjugated diene assays.10 The antioxidant activity of thyme also was demonstrated by Miura et al. using the oil stability index method.11
Antimicrobial activity
In 2011, Sienkiewicz et al. reported that the oil of T. vulgaris displayed potent activity against clinical bacterial strains of Staphylococcus, Enterococcus, Escherichia, and Pseudomonas genera. In addition, thyme essential oil exhibited efficacy against tested antibiotic-resistant strains of bacteria.12 The following year, Sienkiewicz et al. assessed the antimicrobial activity of thyme essential oil against clinical multidrug-resistant strains of Staphylococcus, Enterococcus, Escherichia, and Pseudomonas, finding that it potently suppressed the growth of each.13
Potential cutaneous indications: atopic dermatitis, leishmaniasis, eczema, hair growth
In 2015, Seo and Jeong showed that lavender oil, thyme oil, and a blend of the two were all effective in reducing the symptoms of atopic dermatitis in mice. The researchers suggested that developing treatments with these oils for human patients with atopic dermatitis is warranted.14
Nilforoushzadeh et al. found in 2008 that herbal extracts of T. vulgaris and Achillea millefolium (yarrow), as well as propolis hydroalcoholic extracts, were effective in treating cutaneous leishmaniasis in mice and recommended the study of these extracts alone or in combination in human trials.15
A two-arm, randomized, double-blind, placebo-controlled trial conducted by Shimelis et al. in 2012 evaluated the efficacy of a 3% thyme essential oil antifungal cream and a 10% chamomile extract cream in the treatment of eczemalike lesions. Complete healing was achieved in 10 patients (66.5%) treated with the thyme cream, compared with four patients (28.5%) in the placebo group. Although no significant differences were observed between the active chamomile group and placebo, an appreciable number of subjects improved or healed. The investigators concluded that their findings from this small study suggest that, while more research is needed, a 3% thyme essential oil cream appears to be an inexpensive and readily available option to treat mild to moderate cutaneous conditions, including fungal infections, pityriasis alba, and eczema.16
In 2013, Rastegar et al. found that the combination of herbal extracts (including thyme) and platelet-rich plasma induced significant proliferation of human dermal papilla cells by regulating extracellular signal-regulated kinase (ERK) and Akt (protein kinase B). They concluded that their findings suggest the potential for developing combination therapies intended to improve hair growth.17
Insect repellent activity
In a 2016 study by Gutiérrez et al., the essential oil of T. vulgaris was found to be effective against Pediculus humanus capitis (head lice) adults and eggs. The researchers concluded that T. vulgaris achieves a strong knockdown and mortality rate in adult head lice and toxicity in the eggs after 21 minutes of application at a low concentration.18
In a small 1999 study by Barnard of the repellency to Aedes aegypti and Anopheles albimanus of various concentrations and combinations of five essential oils (Bourbon geranium, cedarwood, clove, peppermint, and thyme) applied to human skin, thyme and clove oils were found to be the most effective mosquito repellents. The author noted that thyme oil (as well as clove and peppermint oils) can irritate the skin and the odor of thyme and clove oils, at concentrations of 25%, or more were deemed unacceptable by the two participants in the study.19
Three years later, Choi et al. found that the essential oil of T. vulgaris also repelled adult mosquitoes (Culex pipiens pallens) on hairless mice and displayed potent repellent activity.20
Melanoma
In 2005, Carrera et al. reported a case of long-term complete remission of cutaneous melanoma metastases in a 73-year-old white woman who consumed a dried thyme herbal tea and thyme topical applications in compresses.4 An association between thyme and melanoma has not been reported in the subsequent literature.
Conclusion
Thyme has a long history of culinary and medical uses. Its antimicrobial and antioxidant activity are well documented. While there is reason to consider the potential applications of thyme for dermatologic conditions, much more research is necessary to determine its viability for such purposes.
References
1. An Illustrated Guide to 101 Medicinal Herbs: Their History, Use, Recommended Dosages, and Cautions (Loveland, Colo.: Interweave Press, 1998, pp. 198-9).
2. J Ethnopharmacol. 2010 Aug 19;131(1):33-55.
3. J Food Sci. 2014 May;79(5):M903-10.
4. J Am Acad Dermatol. 2005 Apr;52(4):713-5.
5. Nat Neurosci. 2006 May;9(5):628-35.
6. J Cell Mol Med. 2016 Sep 19. doi: 10.1111/jcmm.12968. [Epub ahead of print]
7. Cells Tissues Organs. 2016;201(3):180-92.
8. Mutat Res Genet Toxicol Environ Mutagen. 2015 Sep;791:30-7.
9. Cutan Ocul Toxicol. 2007;26(3):227-33.
10. J Agric Food Chem. 2002 Aug 14;50(17):4947-52.
11. J Agric Food Chem. 2002 Mar 27;50(7):1845-51.
12. Med Chem. 2011 Nov;7(6):674-89.
13. Microb Drug Resist. 2012 Apr;18(2):137-48.
14. J Korean Acad Nurs. 2015 Jun;45(3):367-77.
15. J Vector Borne Dis. 2008 Dec;45(4):301-6.
16. Int J Dermatol. 2012 Jul;51(7):790-5.
17. J Cosmet Dermatol. 2013 Jun;12(2):116-22.
18. Parasitol Res. 2016 Feb;115(2):633-41.
19. J Med Entomol. 1999 Sep;36(5):625-9.
20. J Am Mosq Control Assoc. 2002 Dec;18(4):348-51.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Using the Blanch Sign to Differentiate Weathering Nodules From Auricular Tophaceous Gout
To the Editor:
We commend the recent report by Smith et al (Cutis. 2016;97:166, 175-176) that described multiple white nodules on the bilateral helical rims of the ears in a 40-year-old man, which was determined to be bilateral auricular tophaceous gout. Furthermore, we appreciate the inclusion of weathering nodules in the differential diagnosis and wish to share our experience with these lesions.
Auricular tophaceous gout and weathering nodules are clinically similar. Weathering nodules may appear as single or multiple, 2 to 3 mm in diameter and 1 to 2 mm in height, white to flesh-colored papules usually found on the helical rim of the ear (Figure 1).1 We recently described 10 patients with weathering nodules and their associated risk factors.2 We observed that the weathering nodules will blanch upon the application of pressure to the adjacent helical rim; a positive “blanch sign” may be used to differentiate weathering nodules from auricular tophaceous gout and other lesions of the ear (Figure 2). Furthermore, patients with weathering nodules typically exhibit a history of sun exposure and often have other cutaneous findings such as actinic keratoses. The pathogenesis of weathering nodules was previously thought to rely solely on actinic damage; however, we reported a pediatric case of weathering nodules that presented following radiotherapy to the ears.2
In summary, weathering nodules should be included in the differential diagnosis of auricular tophaceous gout. In addition, a positive blanch sign may be a useful clinical tool in differentiating weathering nodules from other ear lesions.
- Kavanagh GM, Bradfield JW, Collins CM, et al. Weathering nodules of the ear: a clinicopathological study. Br J Dermatol. 1996;135:550-554.
- Udkoff J, Cohen PR. Weathering nodules: a report of ten individuals with weathering nodules and review of the literature. Indian J Dermatol. 2016;61:433-436.
To the Editor:
We commend the recent report by Smith et al (Cutis. 2016;97:166, 175-176) that described multiple white nodules on the bilateral helical rims of the ears in a 40-year-old man, which was determined to be bilateral auricular tophaceous gout. Furthermore, we appreciate the inclusion of weathering nodules in the differential diagnosis and wish to share our experience with these lesions.
Auricular tophaceous gout and weathering nodules are clinically similar. Weathering nodules may appear as single or multiple, 2 to 3 mm in diameter and 1 to 2 mm in height, white to flesh-colored papules usually found on the helical rim of the ear (Figure 1).1 We recently described 10 patients with weathering nodules and their associated risk factors.2 We observed that the weathering nodules will blanch upon the application of pressure to the adjacent helical rim; a positive “blanch sign” may be used to differentiate weathering nodules from auricular tophaceous gout and other lesions of the ear (Figure 2). Furthermore, patients with weathering nodules typically exhibit a history of sun exposure and often have other cutaneous findings such as actinic keratoses. The pathogenesis of weathering nodules was previously thought to rely solely on actinic damage; however, we reported a pediatric case of weathering nodules that presented following radiotherapy to the ears.2


In summary, weathering nodules should be included in the differential diagnosis of auricular tophaceous gout. In addition, a positive blanch sign may be a useful clinical tool in differentiating weathering nodules from other ear lesions.
To the Editor:
We commend the recent report by Smith et al (Cutis. 2016;97:166, 175-176) that described multiple white nodules on the bilateral helical rims of the ears in a 40-year-old man, which was determined to be bilateral auricular tophaceous gout. Furthermore, we appreciate the inclusion of weathering nodules in the differential diagnosis and wish to share our experience with these lesions.
Auricular tophaceous gout and weathering nodules are clinically similar. Weathering nodules may appear as single or multiple, 2 to 3 mm in diameter and 1 to 2 mm in height, white to flesh-colored papules usually found on the helical rim of the ear (Figure 1).1 We recently described 10 patients with weathering nodules and their associated risk factors.2 We observed that the weathering nodules will blanch upon the application of pressure to the adjacent helical rim; a positive “blanch sign” may be used to differentiate weathering nodules from auricular tophaceous gout and other lesions of the ear (Figure 2). Furthermore, patients with weathering nodules typically exhibit a history of sun exposure and often have other cutaneous findings such as actinic keratoses. The pathogenesis of weathering nodules was previously thought to rely solely on actinic damage; however, we reported a pediatric case of weathering nodules that presented following radiotherapy to the ears.2


In summary, weathering nodules should be included in the differential diagnosis of auricular tophaceous gout. In addition, a positive blanch sign may be a useful clinical tool in differentiating weathering nodules from other ear lesions.
- Kavanagh GM, Bradfield JW, Collins CM, et al. Weathering nodules of the ear: a clinicopathological study. Br J Dermatol. 1996;135:550-554.
- Udkoff J, Cohen PR. Weathering nodules: a report of ten individuals with weathering nodules and review of the literature. Indian J Dermatol. 2016;61:433-436.
- Kavanagh GM, Bradfield JW, Collins CM, et al. Weathering nodules of the ear: a clinicopathological study. Br J Dermatol. 1996;135:550-554.
- Udkoff J, Cohen PR. Weathering nodules: a report of ten individuals with weathering nodules and review of the literature. Indian J Dermatol. 2016;61:433-436.