User login
VIDEO: New dermal fillers add flexibility
WAILEA, HAWAII – Two hyaluronic acid products now available in the United States are “much more stretchable and flexible” than other fillers, according to Nowell Solish, MD, of the University of Toronto.
The dermal fillers, Restylane Defyne and Restylane Refyne, have been available in Canada, and Dr. Solish was involved in a Canadian study of the fillers in patients in motion. With the new fillers, “animation looks more natural after than before the fillers,” he said at the Hawaii Dermatology Seminar, provided by Global Academy for Medical Education/Skin Disease Education Foundation. In addition to providing a more natural look, the new fillers may also help prevent the development of lines in certain areas, such as around the mouth, he noted.
In a video interview at the meeting, Dr. Solish explained that when he treats a patient, he looks for where there is “too much activity,” such as frequent pursing of the lips, and puts filler in to balance the activity.
He disclosed relationships with Allergan, Galderma (the manufacturer of Restylane products), and Revance.
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WAILEA, HAWAII – Two hyaluronic acid products now available in the United States are “much more stretchable and flexible” than other fillers, according to Nowell Solish, MD, of the University of Toronto.
The dermal fillers, Restylane Defyne and Restylane Refyne, have been available in Canada, and Dr. Solish was involved in a Canadian study of the fillers in patients in motion. With the new fillers, “animation looks more natural after than before the fillers,” he said at the Hawaii Dermatology Seminar, provided by Global Academy for Medical Education/Skin Disease Education Foundation. In addition to providing a more natural look, the new fillers may also help prevent the development of lines in certain areas, such as around the mouth, he noted.
In a video interview at the meeting, Dr. Solish explained that when he treats a patient, he looks for where there is “too much activity,” such as frequent pursing of the lips, and puts filler in to balance the activity.
He disclosed relationships with Allergan, Galderma (the manufacturer of Restylane products), and Revance.
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WAILEA, HAWAII – Two hyaluronic acid products now available in the United States are “much more stretchable and flexible” than other fillers, according to Nowell Solish, MD, of the University of Toronto.
The dermal fillers, Restylane Defyne and Restylane Refyne, have been available in Canada, and Dr. Solish was involved in a Canadian study of the fillers in patients in motion. With the new fillers, “animation looks more natural after than before the fillers,” he said at the Hawaii Dermatology Seminar, provided by Global Academy for Medical Education/Skin Disease Education Foundation. In addition to providing a more natural look, the new fillers may also help prevent the development of lines in certain areas, such as around the mouth, he noted.
In a video interview at the meeting, Dr. Solish explained that when he treats a patient, he looks for where there is “too much activity,” such as frequent pursing of the lips, and puts filler in to balance the activity.
He disclosed relationships with Allergan, Galderma (the manufacturer of Restylane products), and Revance.
SDEF and this news organization are owned by the same parent company.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT SDEF HAWAII DERMATOLOGY SEMINAR
Evidence supports efficacy of topical skin products for photoaging reversal
MIAMI – Topical skin care products are evolving along with evidence in the literature supporting their efficacy; these products include serums, lotions, and cleansers with DNA repair enzymes or epidermal growth factor as active ingredients, according to Ron Moy, MD.
Importantly, some of these formulations not only show efficacy to reverse the signs of photodamage to skin and to promote rejuvenation, but may have a role in skin cancer prevention as well, said Dr. Moy, a dermatologist and facial plastic surgeon in private practice in Beverly Hills, Calif.
More investigators are assessing the mechanisms and potential advantages of DNA repair, Dr. Moy said at the Orlando Dermatology Aesthetic and Clinical Conference. In 2015, the Nobel Prize in Chemistry was awarded to scientists who discovered key mechanisms underlying DNA repair. “Since then, DNA repair has gotten a lot more attention,” he said.
Evidence suggests a person’s DNA repair capability can modulate colon cancer risk. Also, smokers with a low level of DNA repair enzymes carry a higher risk for lung cancer, and DNA repair genes can predict ovarian cancer and lung cancer survival, Dr. Moy said.
“It is still not entirely understood how everything ties together,” he said. “But the work on DNA repair is convincing, more convincing than work on antioxidants or retinoids.” Although antioxidants look favorable in experimental and animal studies, for example, “generally the published work on antioxidants does not give you good clinical results,” he said.
DNA repair enzymes have a beneficial effect on the proto-oncogene hyperexpression in human skin and ultraviolet light telomere shortening, according to an experimental pilot study in the Journal of Drugs in Dermatology (2013 Sep;12[9]:1017-21).
Also, a DNA repair enzyme derived from bacteria, T4 endonuclease V, showed promise in an older study of 30 patients with xeroderma pigmentosum (Lancet. 2001 Mar 24;357[9260]:926-9). Those affected carry a higher risk overall for any skin cancer, compared with the general population. The DNA repair enzyme group had fewer new actinic keratoses and fewer new basal cell carcinoma, squamous cell carcinomas, and melanoma lesions at 1 year, compared with a vehicle-only group at 1 year.
Another class of DNA repair molecules, photolyases, is newer than T4 endonuclease V and “might work even better,” Dr. Moy said.
“There is evidence that DNA enzymes are very effective and helpful in preventing skin cancer,” he pointed out. One mechanism is the ability of exogenous DNA repair enzymes to bolster intrinsic DNA in the fight against carcinogenesis, according to a review article Dr. Moy and his colleagues published in the Journal of Drugs in Dermatology (2015 Mar;14[3]:297-303).
The role of human epidermal growth factor for improving skin appearance and tightening is another area of active research and promise.
“It basically thickens and tightens skin,” Dr. Moy explained. “It works better on thinner skin, including the eyelids and neck.” Ultrasound objectively demonstrates gains in skin thickness. Studies also show improvement in acne scars following application of epidermal growth factor.
In addition, epidermal growth factor can enhance the appearance of solar purpura. In fact, this is the dermatologic condition with the most convincing evidence supporting its use so far, Dr. Moy said. Clinical studies have shown that epidermal growth factor along with other active ingredients also can improve acne scars and eye bags.
During the question and answer session at the ODAC conference, moderator Susan Weinkle, MD, a dermatologist and Mohs surgeon in private practice in Bradenton, Fla., asked Dr. Moy which epidermal growth factor product he recommends.
“There are a lot of different growth factors. Epidermal growth factors are not all the same,” said Dr. Moy, who is the founder of DNA EGF Renewal, a company that manufactures skin care products containing epidermal growth factor and DNA repair enzymes.
When pressed for the name of his growth factor product by Dr. Weinkle and an uproar from the audience, Dr. Moy chose to stay noncommercial, pointing attendees to his website instead: www.dnaegfrenewal.com.
Dr. Moy is founder of DNA EGF Renewal in Beverly Hills, Calif.
MIAMI – Topical skin care products are evolving along with evidence in the literature supporting their efficacy; these products include serums, lotions, and cleansers with DNA repair enzymes or epidermal growth factor as active ingredients, according to Ron Moy, MD.
Importantly, some of these formulations not only show efficacy to reverse the signs of photodamage to skin and to promote rejuvenation, but may have a role in skin cancer prevention as well, said Dr. Moy, a dermatologist and facial plastic surgeon in private practice in Beverly Hills, Calif.
More investigators are assessing the mechanisms and potential advantages of DNA repair, Dr. Moy said at the Orlando Dermatology Aesthetic and Clinical Conference. In 2015, the Nobel Prize in Chemistry was awarded to scientists who discovered key mechanisms underlying DNA repair. “Since then, DNA repair has gotten a lot more attention,” he said.
Evidence suggests a person’s DNA repair capability can modulate colon cancer risk. Also, smokers with a low level of DNA repair enzymes carry a higher risk for lung cancer, and DNA repair genes can predict ovarian cancer and lung cancer survival, Dr. Moy said.
“It is still not entirely understood how everything ties together,” he said. “But the work on DNA repair is convincing, more convincing than work on antioxidants or retinoids.” Although antioxidants look favorable in experimental and animal studies, for example, “generally the published work on antioxidants does not give you good clinical results,” he said.
DNA repair enzymes have a beneficial effect on the proto-oncogene hyperexpression in human skin and ultraviolet light telomere shortening, according to an experimental pilot study in the Journal of Drugs in Dermatology (2013 Sep;12[9]:1017-21).
Also, a DNA repair enzyme derived from bacteria, T4 endonuclease V, showed promise in an older study of 30 patients with xeroderma pigmentosum (Lancet. 2001 Mar 24;357[9260]:926-9). Those affected carry a higher risk overall for any skin cancer, compared with the general population. The DNA repair enzyme group had fewer new actinic keratoses and fewer new basal cell carcinoma, squamous cell carcinomas, and melanoma lesions at 1 year, compared with a vehicle-only group at 1 year.
Another class of DNA repair molecules, photolyases, is newer than T4 endonuclease V and “might work even better,” Dr. Moy said.
“There is evidence that DNA enzymes are very effective and helpful in preventing skin cancer,” he pointed out. One mechanism is the ability of exogenous DNA repair enzymes to bolster intrinsic DNA in the fight against carcinogenesis, according to a review article Dr. Moy and his colleagues published in the Journal of Drugs in Dermatology (2015 Mar;14[3]:297-303).
The role of human epidermal growth factor for improving skin appearance and tightening is another area of active research and promise.
“It basically thickens and tightens skin,” Dr. Moy explained. “It works better on thinner skin, including the eyelids and neck.” Ultrasound objectively demonstrates gains in skin thickness. Studies also show improvement in acne scars following application of epidermal growth factor.
In addition, epidermal growth factor can enhance the appearance of solar purpura. In fact, this is the dermatologic condition with the most convincing evidence supporting its use so far, Dr. Moy said. Clinical studies have shown that epidermal growth factor along with other active ingredients also can improve acne scars and eye bags.
During the question and answer session at the ODAC conference, moderator Susan Weinkle, MD, a dermatologist and Mohs surgeon in private practice in Bradenton, Fla., asked Dr. Moy which epidermal growth factor product he recommends.
“There are a lot of different growth factors. Epidermal growth factors are not all the same,” said Dr. Moy, who is the founder of DNA EGF Renewal, a company that manufactures skin care products containing epidermal growth factor and DNA repair enzymes.
When pressed for the name of his growth factor product by Dr. Weinkle and an uproar from the audience, Dr. Moy chose to stay noncommercial, pointing attendees to his website instead: www.dnaegfrenewal.com.
Dr. Moy is founder of DNA EGF Renewal in Beverly Hills, Calif.
MIAMI – Topical skin care products are evolving along with evidence in the literature supporting their efficacy; these products include serums, lotions, and cleansers with DNA repair enzymes or epidermal growth factor as active ingredients, according to Ron Moy, MD.
Importantly, some of these formulations not only show efficacy to reverse the signs of photodamage to skin and to promote rejuvenation, but may have a role in skin cancer prevention as well, said Dr. Moy, a dermatologist and facial plastic surgeon in private practice in Beverly Hills, Calif.
More investigators are assessing the mechanisms and potential advantages of DNA repair, Dr. Moy said at the Orlando Dermatology Aesthetic and Clinical Conference. In 2015, the Nobel Prize in Chemistry was awarded to scientists who discovered key mechanisms underlying DNA repair. “Since then, DNA repair has gotten a lot more attention,” he said.
Evidence suggests a person’s DNA repair capability can modulate colon cancer risk. Also, smokers with a low level of DNA repair enzymes carry a higher risk for lung cancer, and DNA repair genes can predict ovarian cancer and lung cancer survival, Dr. Moy said.
“It is still not entirely understood how everything ties together,” he said. “But the work on DNA repair is convincing, more convincing than work on antioxidants or retinoids.” Although antioxidants look favorable in experimental and animal studies, for example, “generally the published work on antioxidants does not give you good clinical results,” he said.
DNA repair enzymes have a beneficial effect on the proto-oncogene hyperexpression in human skin and ultraviolet light telomere shortening, according to an experimental pilot study in the Journal of Drugs in Dermatology (2013 Sep;12[9]:1017-21).
Also, a DNA repair enzyme derived from bacteria, T4 endonuclease V, showed promise in an older study of 30 patients with xeroderma pigmentosum (Lancet. 2001 Mar 24;357[9260]:926-9). Those affected carry a higher risk overall for any skin cancer, compared with the general population. The DNA repair enzyme group had fewer new actinic keratoses and fewer new basal cell carcinoma, squamous cell carcinomas, and melanoma lesions at 1 year, compared with a vehicle-only group at 1 year.
Another class of DNA repair molecules, photolyases, is newer than T4 endonuclease V and “might work even better,” Dr. Moy said.
“There is evidence that DNA enzymes are very effective and helpful in preventing skin cancer,” he pointed out. One mechanism is the ability of exogenous DNA repair enzymes to bolster intrinsic DNA in the fight against carcinogenesis, according to a review article Dr. Moy and his colleagues published in the Journal of Drugs in Dermatology (2015 Mar;14[3]:297-303).
The role of human epidermal growth factor for improving skin appearance and tightening is another area of active research and promise.
“It basically thickens and tightens skin,” Dr. Moy explained. “It works better on thinner skin, including the eyelids and neck.” Ultrasound objectively demonstrates gains in skin thickness. Studies also show improvement in acne scars following application of epidermal growth factor.
In addition, epidermal growth factor can enhance the appearance of solar purpura. In fact, this is the dermatologic condition with the most convincing evidence supporting its use so far, Dr. Moy said. Clinical studies have shown that epidermal growth factor along with other active ingredients also can improve acne scars and eye bags.
During the question and answer session at the ODAC conference, moderator Susan Weinkle, MD, a dermatologist and Mohs surgeon in private practice in Bradenton, Fla., asked Dr. Moy which epidermal growth factor product he recommends.
“There are a lot of different growth factors. Epidermal growth factors are not all the same,” said Dr. Moy, who is the founder of DNA EGF Renewal, a company that manufactures skin care products containing epidermal growth factor and DNA repair enzymes.
When pressed for the name of his growth factor product by Dr. Weinkle and an uproar from the audience, Dr. Moy chose to stay noncommercial, pointing attendees to his website instead: www.dnaegfrenewal.com.
Dr. Moy is founder of DNA EGF Renewal in Beverly Hills, Calif.
EXPERT ANALYSIS FROM ODAC 2017
Recognize, treat, and teach others to spot hidradenitis suppurativa
MIAMI – Clinicians have many options to treat and help people manage hidradenitis suppurativa, but for most patients, an early and accurate diagnosis remains elusive.
“The problem here is because it has so many mimickers, the diagnosis is often delayed, patients can be [treated for an incorrect diagnosis] and in many ways that treatment can be harmful,” said Adam Friedman, MD, of the George Washington University in Washington. Missed or ignored diagnoses can lead to more pain, impaired function, and wasted time and money.
“There are – no question – gaps in clinical care. Patients seek care outside of dermatology and go to urgent care centers, emergency rooms, [and other settings]. That’s why it’s not only important for us to recognize this, but to teach everyone else as well,” Dr. Friedman said at the Orlando Dermatology Aesthetic and Clinical Conference.
Tips for early detection
Look for chronicity in the disease’s presentation, Dr. Friedman said. “Chronicity is key, but the morphology will change, and lesions will look different over time.” Therefore, “the clinical presentation can be challenging, depending on when you catch the patient.”
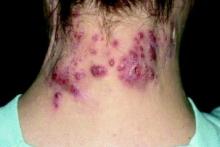
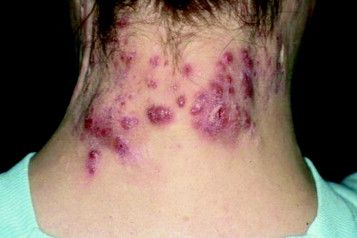
Hidradenitis suppurativa is characterized by very purulent, indurated, abscesslike structures often on the underarms and groin. Ask patients where and how often they see lesions. Lesions in certain locations can be very disabling for patients not only because of pain, but also from a psychosocial impact. The groin and chest are prime examples. Also, there is a genetic predisposition so it is important to ask patients about family history as well.
Use combination therapy to hit disease ‘from all angles’
It is imperative to treat patients even when they present at a time of mild disease, Dr. Friedman said. “This is a chronic, snowballing disease that will get worse over time, because inflammation begets inflammation. Even if it’s mild disease, you still want to treat. Combinations are king, and we dermatologists are the synergy masters.”
Effective treatment strategies include medications that curtail inflammation and block hormonal influences; dietary changes (a minimal-dairy, low-carbohydrate diet helps some patients, for example); environmental changes and/or eliminating the invasive proliferative gelatinous mass (IPGM). “This is not step therapy,” Dr. Friedman emphasized. “You want to hit all these angles at once.”
In terms of nutritional support, “I usually put my patients on a combination of zinc and vitamin C, both anti-inflammatories, but also good for wound healing,” Dr. Friedman said. “I also get them on board with V-8, which can be a tough sell sometimes.” He recommends patients drink three small cans per week, adding that he has no financial disclosure related to the vegetable juice.
Patient education, smoking cessation, and keeping affected areas dry and cool are other important management strategies. Instruct patients that stress, friction, and obesity can each worsen the condition “I think obesity is an independent risk factor here, like it is in psoriasis, where obesity alone has been shown to increase the risk of psoriasis later on,” he said.
Ease the inflammation
Hidradenitis suppurativa is a disease of “inappropriate inflammation,” Dr. Friedman said, which explains why anti-inflammatory agents remain the mainstay of treatment. These include antibiotics, classic corticosteroids, and biologics, which all can have a role in therapy. He highlighted the potential role of the cutaneous microbiota and possible dysbiosis associated with disease activity. “I also use a lot chlorhexidine washes to wipe the microbial slate clean in high-risk areas; just be careful to avoid the face.”
Intralesional Kenalog (triamcinolone acetonide), in particular, is useful for its rapid results. “This is such a great and easy trick, and it really works quickly,” Dr. Friedman said. He added that a recent case series provides evidence for its efficacy as well (J Amer Acad Dermatol. 2016 Dec;75:1151-5).
Three take-home treatment pointers
Dr. Friedman shared these three take-aways for treatment of hidradenitis suppurativa with antibiotics:
- The combination of oral clindamycin 300 mg twice daily and oral rifampin 300 mg twice daily carries the most evidence for efficacy and safety, with no evidence of resistance.
- Rifampin also acts against Clostridium difficile infections, which decreases the risk of associated colitis.
- Do not give a tetracycline antibiotic as monotherapy.
In terms of retinoids, “I’ve been pretty disappointed. I find them effective [in other conditions], but for hidradenitis suppurativa, I’m just not impressed, unfortunately,” Dr. Friedman said. “Antihormonal therapies such as oral contraceptives and spironolactone for women and finasteride for men have been a useful adjuncts in my practice, with evidence supporting their use in the literature, he added. Biologics are among the new treatment options, but there are cost and insurance coverage issues, Dr. Friedman said. There are small case series evaluating biologics such as infliximab, which are very supportive – and he himself has had good responses – although the Food and Drug Administration indication has been the hurdle.
One exception is the recent FDA approval of adalimumab (Humira) for hidradenitis suppurativa. “Make sure you realize that the dosing is different, more like a ‘whopping’ Crohn’s disease dose. This is a very important medication in our armamentarium – the issue is when you start it,” Dr. Friedman said. He also cautioned, “if a patient has sinus tracts and scarring, it’s not going to be enough. You still need to address that. Adalimumab is only going to get rid of the inflammation. This is why early diagnosis is so important!”
When it comes to surgery, “my philosophy is it’s all or none. If you’re going to do surgery, do surgery. You better cut these things out. Do a wide global excision,” Dr. Friedman emphasized.
Dr. Friedman is a member of the Dermatology News editorial advisory board.
Dr. Friedman had no relevant financial disclosures.
MIAMI – Clinicians have many options to treat and help people manage hidradenitis suppurativa, but for most patients, an early and accurate diagnosis remains elusive.
“The problem here is because it has so many mimickers, the diagnosis is often delayed, patients can be [treated for an incorrect diagnosis] and in many ways that treatment can be harmful,” said Adam Friedman, MD, of the George Washington University in Washington. Missed or ignored diagnoses can lead to more pain, impaired function, and wasted time and money.
“There are – no question – gaps in clinical care. Patients seek care outside of dermatology and go to urgent care centers, emergency rooms, [and other settings]. That’s why it’s not only important for us to recognize this, but to teach everyone else as well,” Dr. Friedman said at the Orlando Dermatology Aesthetic and Clinical Conference.
Tips for early detection
Look for chronicity in the disease’s presentation, Dr. Friedman said. “Chronicity is key, but the morphology will change, and lesions will look different over time.” Therefore, “the clinical presentation can be challenging, depending on when you catch the patient.”
Hidradenitis suppurativa is characterized by very purulent, indurated, abscesslike structures often on the underarms and groin. Ask patients where and how often they see lesions. Lesions in certain locations can be very disabling for patients not only because of pain, but also from a psychosocial impact. The groin and chest are prime examples. Also, there is a genetic predisposition so it is important to ask patients about family history as well.
Use combination therapy to hit disease ‘from all angles’
It is imperative to treat patients even when they present at a time of mild disease, Dr. Friedman said. “This is a chronic, snowballing disease that will get worse over time, because inflammation begets inflammation. Even if it’s mild disease, you still want to treat. Combinations are king, and we dermatologists are the synergy masters.”
Effective treatment strategies include medications that curtail inflammation and block hormonal influences; dietary changes (a minimal-dairy, low-carbohydrate diet helps some patients, for example); environmental changes and/or eliminating the invasive proliferative gelatinous mass (IPGM). “This is not step therapy,” Dr. Friedman emphasized. “You want to hit all these angles at once.”
In terms of nutritional support, “I usually put my patients on a combination of zinc and vitamin C, both anti-inflammatories, but also good for wound healing,” Dr. Friedman said. “I also get them on board with V-8, which can be a tough sell sometimes.” He recommends patients drink three small cans per week, adding that he has no financial disclosure related to the vegetable juice.
Patient education, smoking cessation, and keeping affected areas dry and cool are other important management strategies. Instruct patients that stress, friction, and obesity can each worsen the condition “I think obesity is an independent risk factor here, like it is in psoriasis, where obesity alone has been shown to increase the risk of psoriasis later on,” he said.
Ease the inflammation
Hidradenitis suppurativa is a disease of “inappropriate inflammation,” Dr. Friedman said, which explains why anti-inflammatory agents remain the mainstay of treatment. These include antibiotics, classic corticosteroids, and biologics, which all can have a role in therapy. He highlighted the potential role of the cutaneous microbiota and possible dysbiosis associated with disease activity. “I also use a lot chlorhexidine washes to wipe the microbial slate clean in high-risk areas; just be careful to avoid the face.”
Intralesional Kenalog (triamcinolone acetonide), in particular, is useful for its rapid results. “This is such a great and easy trick, and it really works quickly,” Dr. Friedman said. He added that a recent case series provides evidence for its efficacy as well (J Amer Acad Dermatol. 2016 Dec;75:1151-5).
Three take-home treatment pointers
Dr. Friedman shared these three take-aways for treatment of hidradenitis suppurativa with antibiotics:
- The combination of oral clindamycin 300 mg twice daily and oral rifampin 300 mg twice daily carries the most evidence for efficacy and safety, with no evidence of resistance.
- Rifampin also acts against Clostridium difficile infections, which decreases the risk of associated colitis.
- Do not give a tetracycline antibiotic as monotherapy.
In terms of retinoids, “I’ve been pretty disappointed. I find them effective [in other conditions], but for hidradenitis suppurativa, I’m just not impressed, unfortunately,” Dr. Friedman said. “Antihormonal therapies such as oral contraceptives and spironolactone for women and finasteride for men have been a useful adjuncts in my practice, with evidence supporting their use in the literature, he added. Biologics are among the new treatment options, but there are cost and insurance coverage issues, Dr. Friedman said. There are small case series evaluating biologics such as infliximab, which are very supportive – and he himself has had good responses – although the Food and Drug Administration indication has been the hurdle.
One exception is the recent FDA approval of adalimumab (Humira) for hidradenitis suppurativa. “Make sure you realize that the dosing is different, more like a ‘whopping’ Crohn’s disease dose. This is a very important medication in our armamentarium – the issue is when you start it,” Dr. Friedman said. He also cautioned, “if a patient has sinus tracts and scarring, it’s not going to be enough. You still need to address that. Adalimumab is only going to get rid of the inflammation. This is why early diagnosis is so important!”
When it comes to surgery, “my philosophy is it’s all or none. If you’re going to do surgery, do surgery. You better cut these things out. Do a wide global excision,” Dr. Friedman emphasized.
Dr. Friedman is a member of the Dermatology News editorial advisory board.
Dr. Friedman had no relevant financial disclosures.
MIAMI – Clinicians have many options to treat and help people manage hidradenitis suppurativa, but for most patients, an early and accurate diagnosis remains elusive.
“The problem here is because it has so many mimickers, the diagnosis is often delayed, patients can be [treated for an incorrect diagnosis] and in many ways that treatment can be harmful,” said Adam Friedman, MD, of the George Washington University in Washington. Missed or ignored diagnoses can lead to more pain, impaired function, and wasted time and money.
“There are – no question – gaps in clinical care. Patients seek care outside of dermatology and go to urgent care centers, emergency rooms, [and other settings]. That’s why it’s not only important for us to recognize this, but to teach everyone else as well,” Dr. Friedman said at the Orlando Dermatology Aesthetic and Clinical Conference.
Tips for early detection
Look for chronicity in the disease’s presentation, Dr. Friedman said. “Chronicity is key, but the morphology will change, and lesions will look different over time.” Therefore, “the clinical presentation can be challenging, depending on when you catch the patient.”
Hidradenitis suppurativa is characterized by very purulent, indurated, abscesslike structures often on the underarms and groin. Ask patients where and how often they see lesions. Lesions in certain locations can be very disabling for patients not only because of pain, but also from a psychosocial impact. The groin and chest are prime examples. Also, there is a genetic predisposition so it is important to ask patients about family history as well.
Use combination therapy to hit disease ‘from all angles’
It is imperative to treat patients even when they present at a time of mild disease, Dr. Friedman said. “This is a chronic, snowballing disease that will get worse over time, because inflammation begets inflammation. Even if it’s mild disease, you still want to treat. Combinations are king, and we dermatologists are the synergy masters.”
Effective treatment strategies include medications that curtail inflammation and block hormonal influences; dietary changes (a minimal-dairy, low-carbohydrate diet helps some patients, for example); environmental changes and/or eliminating the invasive proliferative gelatinous mass (IPGM). “This is not step therapy,” Dr. Friedman emphasized. “You want to hit all these angles at once.”
In terms of nutritional support, “I usually put my patients on a combination of zinc and vitamin C, both anti-inflammatories, but also good for wound healing,” Dr. Friedman said. “I also get them on board with V-8, which can be a tough sell sometimes.” He recommends patients drink three small cans per week, adding that he has no financial disclosure related to the vegetable juice.
Patient education, smoking cessation, and keeping affected areas dry and cool are other important management strategies. Instruct patients that stress, friction, and obesity can each worsen the condition “I think obesity is an independent risk factor here, like it is in psoriasis, where obesity alone has been shown to increase the risk of psoriasis later on,” he said.
Ease the inflammation
Hidradenitis suppurativa is a disease of “inappropriate inflammation,” Dr. Friedman said, which explains why anti-inflammatory agents remain the mainstay of treatment. These include antibiotics, classic corticosteroids, and biologics, which all can have a role in therapy. He highlighted the potential role of the cutaneous microbiota and possible dysbiosis associated with disease activity. “I also use a lot chlorhexidine washes to wipe the microbial slate clean in high-risk areas; just be careful to avoid the face.”
Intralesional Kenalog (triamcinolone acetonide), in particular, is useful for its rapid results. “This is such a great and easy trick, and it really works quickly,” Dr. Friedman said. He added that a recent case series provides evidence for its efficacy as well (J Amer Acad Dermatol. 2016 Dec;75:1151-5).
Three take-home treatment pointers
Dr. Friedman shared these three take-aways for treatment of hidradenitis suppurativa with antibiotics:
- The combination of oral clindamycin 300 mg twice daily and oral rifampin 300 mg twice daily carries the most evidence for efficacy and safety, with no evidence of resistance.
- Rifampin also acts against Clostridium difficile infections, which decreases the risk of associated colitis.
- Do not give a tetracycline antibiotic as monotherapy.
In terms of retinoids, “I’ve been pretty disappointed. I find them effective [in other conditions], but for hidradenitis suppurativa, I’m just not impressed, unfortunately,” Dr. Friedman said. “Antihormonal therapies such as oral contraceptives and spironolactone for women and finasteride for men have been a useful adjuncts in my practice, with evidence supporting their use in the literature, he added. Biologics are among the new treatment options, but there are cost and insurance coverage issues, Dr. Friedman said. There are small case series evaluating biologics such as infliximab, which are very supportive – and he himself has had good responses – although the Food and Drug Administration indication has been the hurdle.
One exception is the recent FDA approval of adalimumab (Humira) for hidradenitis suppurativa. “Make sure you realize that the dosing is different, more like a ‘whopping’ Crohn’s disease dose. This is a very important medication in our armamentarium – the issue is when you start it,” Dr. Friedman said. He also cautioned, “if a patient has sinus tracts and scarring, it’s not going to be enough. You still need to address that. Adalimumab is only going to get rid of the inflammation. This is why early diagnosis is so important!”
When it comes to surgery, “my philosophy is it’s all or none. If you’re going to do surgery, do surgery. You better cut these things out. Do a wide global excision,” Dr. Friedman emphasized.
Dr. Friedman is a member of the Dermatology News editorial advisory board.
Dr. Friedman had no relevant financial disclosures.
EXPERT ANALYSIS FROM ODAC 2017
Heparan sulfate
The discussion this month focuses on an exciting new ingredient that is showing great promise as a topical antiaging agent. For years we have known that aging skin needs more collagen, elastin, and hyaluronic acid. It is time to add a new player to the antiaging team – heparan sulfate (HS). New studies have shown that lower levels of HS are associated with aged skin. This is what you need to know.
Significance of HS
Endogenous HS, an essential glycosaminoglycan (GAG), contributes to skin development and homeostasis, and thus actively promotes skin health.1 GAGs such as HS and hyaluronic acid are well known as endogenous superhydrators that bind and retain water, thus contributing to skin hydration as well as preserving the structural integrity of collagen and elastin fibers. Specifically, HS and its protein-bound forms (HS proteoglycans such as syndecan, glypican, and perlecan) – the most common constituents on the cell surface and in the extracellular matrix (ECM) – play an important role in cutaneous cell proliferation, migration, communication, and activation as well as collagen fiber development, basement membrane regeneration, granulation tissue formation, and cell adhesion related to wound healing.1 This results from their capacity to bind, store, present, degrade, and amplify growth factors and cytokines.
Accordingly, mature skin would be potently activated by its endogenous growth factors (at lower concentration) as cell signal is amplified (lower threshold of activation) by HS analogs. When used topically, HS analogs appear to promote the formation of healthy and functional ECM, resulting in firmer, more elastic, and stronger skin. Augmenting the amount of HS in the skin may yield a rejuvenating effect by expanding the skin’s ability to hold water and restore cutaneous homeostasis. Studies with an HS analog have demonstrated that the formulation penetrates into skin, enhancing hydration, reducing transepidermal water loss, and improving the appearance of wrinkles and skin tone.1 The use of HS analogs or mimics, known as matrix therapy, has been shown to be effective clinically in cutaneous and corneal healing formulations, exhibiting proof of concept in humans, according to Barritault et al.3
Matrix therapy also has been employed successfully in plastic and aesthetic procedures. Zakine et al. assessed the impact of using ReGeneraTing Agents (RGTA) – biodegradable polymers designed to mimic HS in the ECM of damaged tissue – in 17 patients with breast hypertrophy who underwent mammoplasty. Patients received topical treatment on one breast 1, 4, 8, and 11 days after surgery. The investigators also evaluated a different group of 50 patients after centrofacial lifting. These patients received RGTA drops bilaterally in the treatment area after surgery. Inflammation, hypertrophic scars, and pruritus were noted less frequently for the breasts treated with RGTA. Similarly, in patients receiving a centrofacial lift, scar inflammation, edema, and bruising were significantly less frequent in the treated group (10%), compared with the untreated group (90%).4
In 2015, Gallo et al. reported that 15 patients using a new HS analog formulation in an 8-week study displayed improvement in various skin metrics, including hydration, firmness, elasticity, barrier function, and the appearance of fine lines and wrinkles. The investigators concluded that photodamaged skin may benefit from the use of novel topically applied products containing low-molecular-weight HS.1
The synthetic heparan sulfate Cacipliq20 was reported in 2015 to have been used successfully to treat a chronic lower-extremity ulcer in a 22-year-old male patient with Stewart-Bluefarb syndrome,5 and in 2016 to treat a refractory sickle cell ulcer, with the encouraging outcome demonstrated by complete wound coverage and significant improvement in pain score.6 In 2012, Polieri et al. showed that HS 1% cream was comparable or more effective than glycosaminoglycan-polysulphate gel in alleviating the signs and symptoms of hematomas or subcutaneous hematic extravasations in a study with 96 white men and women.7
Conclusion
Heparan sulfate does appear to be a novel viable antiaging option. Endogenous heparan sulfate is involved in skin defense against pathogens, dehydrated/dried skin, redness, and wound healing. Theoretically, then, HS analogs should be able to modulate these processes. More research is necessary to determine if this is the case, however. In the meantime, HS analogs are extremely well tolerated by all patients and especially those with sensitive skin, which is often the case with aging skin. Further, HS analogs promote skin hydration, providing resistance to compressive forces as well as keeping skin looking healthy. Anecdotally, I can report that I have been using the Senté Dermal Repair Cream in my rosacea patients without any problems. I think HS analogs represent a good option for patients who cannot tolerate retinoids.
Resources
1. J Drugs Dermatol. 2015 Jul;14(7):669-74.
2. Proteoglycans in Skin Aging, in “Textbook of Aging Skin” (Heidelberg, Berlin: Springer-Verlag, pp. 109-120).
3. Joint Bone Spine. 2016 Sep 20. doi: 10.1016/j.jbspin.2016.06.012.
4. Ann Chir Plast Esthet. 2010 Oct;55(5):421-8.
5. Int Wound J. 2015 Apr;12(2):169-72.
6. Int Wound J. 2016 Feb;13(1):35-8.
7. ISRN Orthop. 2012 Jan 17. doi: 10.5402/2012/504151.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
The discussion this month focuses on an exciting new ingredient that is showing great promise as a topical antiaging agent. For years we have known that aging skin needs more collagen, elastin, and hyaluronic acid. It is time to add a new player to the antiaging team – heparan sulfate (HS). New studies have shown that lower levels of HS are associated with aged skin. This is what you need to know.
Significance of HS
Endogenous HS, an essential glycosaminoglycan (GAG), contributes to skin development and homeostasis, and thus actively promotes skin health.1 GAGs such as HS and hyaluronic acid are well known as endogenous superhydrators that bind and retain water, thus contributing to skin hydration as well as preserving the structural integrity of collagen and elastin fibers. Specifically, HS and its protein-bound forms (HS proteoglycans such as syndecan, glypican, and perlecan) – the most common constituents on the cell surface and in the extracellular matrix (ECM) – play an important role in cutaneous cell proliferation, migration, communication, and activation as well as collagen fiber development, basement membrane regeneration, granulation tissue formation, and cell adhesion related to wound healing.1 This results from their capacity to bind, store, present, degrade, and amplify growth factors and cytokines.
Accordingly, mature skin would be potently activated by its endogenous growth factors (at lower concentration) as cell signal is amplified (lower threshold of activation) by HS analogs. When used topically, HS analogs appear to promote the formation of healthy and functional ECM, resulting in firmer, more elastic, and stronger skin. Augmenting the amount of HS in the skin may yield a rejuvenating effect by expanding the skin’s ability to hold water and restore cutaneous homeostasis. Studies with an HS analog have demonstrated that the formulation penetrates into skin, enhancing hydration, reducing transepidermal water loss, and improving the appearance of wrinkles and skin tone.1 The use of HS analogs or mimics, known as matrix therapy, has been shown to be effective clinically in cutaneous and corneal healing formulations, exhibiting proof of concept in humans, according to Barritault et al.3
Matrix therapy also has been employed successfully in plastic and aesthetic procedures. Zakine et al. assessed the impact of using ReGeneraTing Agents (RGTA) – biodegradable polymers designed to mimic HS in the ECM of damaged tissue – in 17 patients with breast hypertrophy who underwent mammoplasty. Patients received topical treatment on one breast 1, 4, 8, and 11 days after surgery. The investigators also evaluated a different group of 50 patients after centrofacial lifting. These patients received RGTA drops bilaterally in the treatment area after surgery. Inflammation, hypertrophic scars, and pruritus were noted less frequently for the breasts treated with RGTA. Similarly, in patients receiving a centrofacial lift, scar inflammation, edema, and bruising were significantly less frequent in the treated group (10%), compared with the untreated group (90%).4
In 2015, Gallo et al. reported that 15 patients using a new HS analog formulation in an 8-week study displayed improvement in various skin metrics, including hydration, firmness, elasticity, barrier function, and the appearance of fine lines and wrinkles. The investigators concluded that photodamaged skin may benefit from the use of novel topically applied products containing low-molecular-weight HS.1
The synthetic heparan sulfate Cacipliq20 was reported in 2015 to have been used successfully to treat a chronic lower-extremity ulcer in a 22-year-old male patient with Stewart-Bluefarb syndrome,5 and in 2016 to treat a refractory sickle cell ulcer, with the encouraging outcome demonstrated by complete wound coverage and significant improvement in pain score.6 In 2012, Polieri et al. showed that HS 1% cream was comparable or more effective than glycosaminoglycan-polysulphate gel in alleviating the signs and symptoms of hematomas or subcutaneous hematic extravasations in a study with 96 white men and women.7
Conclusion
Heparan sulfate does appear to be a novel viable antiaging option. Endogenous heparan sulfate is involved in skin defense against pathogens, dehydrated/dried skin, redness, and wound healing. Theoretically, then, HS analogs should be able to modulate these processes. More research is necessary to determine if this is the case, however. In the meantime, HS analogs are extremely well tolerated by all patients and especially those with sensitive skin, which is often the case with aging skin. Further, HS analogs promote skin hydration, providing resistance to compressive forces as well as keeping skin looking healthy. Anecdotally, I can report that I have been using the Senté Dermal Repair Cream in my rosacea patients without any problems. I think HS analogs represent a good option for patients who cannot tolerate retinoids.
Resources
1. J Drugs Dermatol. 2015 Jul;14(7):669-74.
2. Proteoglycans in Skin Aging, in “Textbook of Aging Skin” (Heidelberg, Berlin: Springer-Verlag, pp. 109-120).
3. Joint Bone Spine. 2016 Sep 20. doi: 10.1016/j.jbspin.2016.06.012.
4. Ann Chir Plast Esthet. 2010 Oct;55(5):421-8.
5. Int Wound J. 2015 Apr;12(2):169-72.
6. Int Wound J. 2016 Feb;13(1):35-8.
7. ISRN Orthop. 2012 Jan 17. doi: 10.5402/2012/504151.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
The discussion this month focuses on an exciting new ingredient that is showing great promise as a topical antiaging agent. For years we have known that aging skin needs more collagen, elastin, and hyaluronic acid. It is time to add a new player to the antiaging team – heparan sulfate (HS). New studies have shown that lower levels of HS are associated with aged skin. This is what you need to know.
Significance of HS
Endogenous HS, an essential glycosaminoglycan (GAG), contributes to skin development and homeostasis, and thus actively promotes skin health.1 GAGs such as HS and hyaluronic acid are well known as endogenous superhydrators that bind and retain water, thus contributing to skin hydration as well as preserving the structural integrity of collagen and elastin fibers. Specifically, HS and its protein-bound forms (HS proteoglycans such as syndecan, glypican, and perlecan) – the most common constituents on the cell surface and in the extracellular matrix (ECM) – play an important role in cutaneous cell proliferation, migration, communication, and activation as well as collagen fiber development, basement membrane regeneration, granulation tissue formation, and cell adhesion related to wound healing.1 This results from their capacity to bind, store, present, degrade, and amplify growth factors and cytokines.
Accordingly, mature skin would be potently activated by its endogenous growth factors (at lower concentration) as cell signal is amplified (lower threshold of activation) by HS analogs. When used topically, HS analogs appear to promote the formation of healthy and functional ECM, resulting in firmer, more elastic, and stronger skin. Augmenting the amount of HS in the skin may yield a rejuvenating effect by expanding the skin’s ability to hold water and restore cutaneous homeostasis. Studies with an HS analog have demonstrated that the formulation penetrates into skin, enhancing hydration, reducing transepidermal water loss, and improving the appearance of wrinkles and skin tone.1 The use of HS analogs or mimics, known as matrix therapy, has been shown to be effective clinically in cutaneous and corneal healing formulations, exhibiting proof of concept in humans, according to Barritault et al.3
Matrix therapy also has been employed successfully in plastic and aesthetic procedures. Zakine et al. assessed the impact of using ReGeneraTing Agents (RGTA) – biodegradable polymers designed to mimic HS in the ECM of damaged tissue – in 17 patients with breast hypertrophy who underwent mammoplasty. Patients received topical treatment on one breast 1, 4, 8, and 11 days after surgery. The investigators also evaluated a different group of 50 patients after centrofacial lifting. These patients received RGTA drops bilaterally in the treatment area after surgery. Inflammation, hypertrophic scars, and pruritus were noted less frequently for the breasts treated with RGTA. Similarly, in patients receiving a centrofacial lift, scar inflammation, edema, and bruising were significantly less frequent in the treated group (10%), compared with the untreated group (90%).4
In 2015, Gallo et al. reported that 15 patients using a new HS analog formulation in an 8-week study displayed improvement in various skin metrics, including hydration, firmness, elasticity, barrier function, and the appearance of fine lines and wrinkles. The investigators concluded that photodamaged skin may benefit from the use of novel topically applied products containing low-molecular-weight HS.1
The synthetic heparan sulfate Cacipliq20 was reported in 2015 to have been used successfully to treat a chronic lower-extremity ulcer in a 22-year-old male patient with Stewart-Bluefarb syndrome,5 and in 2016 to treat a refractory sickle cell ulcer, with the encouraging outcome demonstrated by complete wound coverage and significant improvement in pain score.6 In 2012, Polieri et al. showed that HS 1% cream was comparable or more effective than glycosaminoglycan-polysulphate gel in alleviating the signs and symptoms of hematomas or subcutaneous hematic extravasations in a study with 96 white men and women.7
Conclusion
Heparan sulfate does appear to be a novel viable antiaging option. Endogenous heparan sulfate is involved in skin defense against pathogens, dehydrated/dried skin, redness, and wound healing. Theoretically, then, HS analogs should be able to modulate these processes. More research is necessary to determine if this is the case, however. In the meantime, HS analogs are extremely well tolerated by all patients and especially those with sensitive skin, which is often the case with aging skin. Further, HS analogs promote skin hydration, providing resistance to compressive forces as well as keeping skin looking healthy. Anecdotally, I can report that I have been using the Senté Dermal Repair Cream in my rosacea patients without any problems. I think HS analogs represent a good option for patients who cannot tolerate retinoids.
Resources
1. J Drugs Dermatol. 2015 Jul;14(7):669-74.
2. Proteoglycans in Skin Aging, in “Textbook of Aging Skin” (Heidelberg, Berlin: Springer-Verlag, pp. 109-120).
3. Joint Bone Spine. 2016 Sep 20. doi: 10.1016/j.jbspin.2016.06.012.
4. Ann Chir Plast Esthet. 2010 Oct;55(5):421-8.
5. Int Wound J. 2015 Apr;12(2):169-72.
6. Int Wound J. 2016 Feb;13(1):35-8.
7. ISRN Orthop. 2012 Jan 17. doi: 10.5402/2012/504151.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
An insider’s guide to aesthetic dermatology
When aesthetic dermatology takes the stage at SDEF’s Annual Hawaii Dermatology Seminar, experts will address the latest tips for successful outcomes with filler and toxins, offer guidance on choosing devices, and explain how to make the most of cosmeceuticals in a dermatology practice.
Christopher B. Zachary, MD, meeting codirector and professor and chair of the department of dermatology at the University of California, Irvine, cochairs a pair of aesthetic and procedural dermatology sessions with Michael S. Kaminer, MD, of Yale University, New Haven, Conn., and SkinCare Physicians, Chestnut Hill, Mass.
Last year, Dr. Zachary and a panel of expert aesthetic dermatologists addressed the enduring value of topical retinoids for facial rejuvenation. Read their comments here.
Stay tuned for the latest tips and techniques in aesthetic dermatology from the 2017 SDEF Hawaii Dermatology Seminar.
When aesthetic dermatology takes the stage at SDEF’s Annual Hawaii Dermatology Seminar, experts will address the latest tips for successful outcomes with filler and toxins, offer guidance on choosing devices, and explain how to make the most of cosmeceuticals in a dermatology practice.
Christopher B. Zachary, MD, meeting codirector and professor and chair of the department of dermatology at the University of California, Irvine, cochairs a pair of aesthetic and procedural dermatology sessions with Michael S. Kaminer, MD, of Yale University, New Haven, Conn., and SkinCare Physicians, Chestnut Hill, Mass.
Last year, Dr. Zachary and a panel of expert aesthetic dermatologists addressed the enduring value of topical retinoids for facial rejuvenation. Read their comments here.
Stay tuned for the latest tips and techniques in aesthetic dermatology from the 2017 SDEF Hawaii Dermatology Seminar.
When aesthetic dermatology takes the stage at SDEF’s Annual Hawaii Dermatology Seminar, experts will address the latest tips for successful outcomes with filler and toxins, offer guidance on choosing devices, and explain how to make the most of cosmeceuticals in a dermatology practice.
Christopher B. Zachary, MD, meeting codirector and professor and chair of the department of dermatology at the University of California, Irvine, cochairs a pair of aesthetic and procedural dermatology sessions with Michael S. Kaminer, MD, of Yale University, New Haven, Conn., and SkinCare Physicians, Chestnut Hill, Mass.
Last year, Dr. Zachary and a panel of expert aesthetic dermatologists addressed the enduring value of topical retinoids for facial rejuvenation. Read their comments here.
Stay tuned for the latest tips and techniques in aesthetic dermatology from the 2017 SDEF Hawaii Dermatology Seminar.
Laser vaginal rejuvenation procedures
VIENNA – Vaginal rejuvenation is a major practice growth opportunity for dermatologists who have expertise with lasers, Peter Bjerring, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
The rejuvenation procedures involve heating the connective tissue of the vaginal wall to 40-42 C in order to stimulate tissue remodeling with formation of new collagen and elastic fibers. The evidence base for vaginal rejuvenation using a variety of noninvasive energy-based devices developed for vaginal use isn’t nearly as extensive as it is for skin rejuvenation using lasers. Research is beginning to increase for this indication, but current results are primarily limited to small single-arm studies based on self-reported improvements. Further, studies don’t compare outcomes vs another treatment, such as estrogen cream.
Despite the minimal evidence base for laser procedures, “feminine rejuvenation is becoming very popular. These are (women) who might present to a dermatologist or to a gynecologist,” observed Dr. Bjerring, medical director and head of the Skin and Laser Center at Malholm (Denmark) Hospital.
Minimally ablative or nonablative fractional laser therapy for vaginal rejuvenation requires no anesthesia and no downtime. The lasers being used for this purpose are similar to those already used most often for skin resurfacing and rejuvenation of the face and neck: fractional CO2 lasers at the 10,600-nm wavelength, such as the MonaLisa Touch or FemTouch, and 2,940-nm nonablative erbium:yttrium-aluminum-garnet (Er:YAG) lasers such as the IntimaLase. A course of treatment with these devices typically consists of three, 15-minute sessions at 4- to 6-week intervals.
Dr. Bjerring noted that this mechanism of benefit has been demonstrated by Italian investigators who conducted a histologic study of the effects of microablative fractional CO2 laser therapy on ex vivo specimens of atrophic vaginal tissue obtained from women with vulvovaginal atrophy who underwent major surgery for pelvic organ prolapse.
The investigators treated one side of the atrophic vaginal wall specimen with the microablative fractional CO2 laser and left the contralateral area untreated as a control. They documented that laser therapy restored the vaginal squamous stratified epithelium, with enhanced storage of glycogen in the epithelial cells and shedding of glycogen-rich cells at the epithelial surface. In the connective tissue, activated fibroblasts synthesized new collagen-laden extracellular matrix. All this was accomplished without damage to adjacent untreated tissue (Menopause 2015 Aug;22(8):845-9).
In a single-arm study performed by many of the same Italian investigators, 77 postmenopausal women underwent a course of fractional microablative CO2 laser therapy because they experienced painful sexual intercourse due to vulvovaginal atrophy. At 12 weeks of followup, the group reported significant improvement in sexual function and satisfaction with their sexual life as measured by the Short Form-12 and the Female Sexual Function Index. Self-rated scores of vaginal burning, dryness, and itching improved significantly, as did complaints of pain during intercourse or urination. At baseline, 20 of the 77 women were not sexually active due to the severity of their vulvovaginal atrophy; at followup, 17 of the 20 had reestablished sexual activity (Climacteric. 2015 Apr;18(2):219-25).
Dr. Bjerring also highlighted an American study, again single-arm, in which 27 women with genitourinary syndrome of menopause were examined at baseline and again 3 months after their third and final treatment with a fractional CO2 laser. At follow up, 26 of the 27 pronounced themselves satisfied or extremely satisfied with the results, with significant improvement in the same outcome measures used in the Italian study. At follow up, 25 of the women had an increase in comfortable dilator size (Menopause 2016 Oct;23(10):1102-7).
Dr. Bjerring reported having no financial conflicts of interest regarding his presentation.
VIENNA – Vaginal rejuvenation is a major practice growth opportunity for dermatologists who have expertise with lasers, Peter Bjerring, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
The rejuvenation procedures involve heating the connective tissue of the vaginal wall to 40-42 C in order to stimulate tissue remodeling with formation of new collagen and elastic fibers. The evidence base for vaginal rejuvenation using a variety of noninvasive energy-based devices developed for vaginal use isn’t nearly as extensive as it is for skin rejuvenation using lasers. Research is beginning to increase for this indication, but current results are primarily limited to small single-arm studies based on self-reported improvements. Further, studies don’t compare outcomes vs another treatment, such as estrogen cream.
Despite the minimal evidence base for laser procedures, “feminine rejuvenation is becoming very popular. These are (women) who might present to a dermatologist or to a gynecologist,” observed Dr. Bjerring, medical director and head of the Skin and Laser Center at Malholm (Denmark) Hospital.
Minimally ablative or nonablative fractional laser therapy for vaginal rejuvenation requires no anesthesia and no downtime. The lasers being used for this purpose are similar to those already used most often for skin resurfacing and rejuvenation of the face and neck: fractional CO2 lasers at the 10,600-nm wavelength, such as the MonaLisa Touch or FemTouch, and 2,940-nm nonablative erbium:yttrium-aluminum-garnet (Er:YAG) lasers such as the IntimaLase. A course of treatment with these devices typically consists of three, 15-minute sessions at 4- to 6-week intervals.
Dr. Bjerring noted that this mechanism of benefit has been demonstrated by Italian investigators who conducted a histologic study of the effects of microablative fractional CO2 laser therapy on ex vivo specimens of atrophic vaginal tissue obtained from women with vulvovaginal atrophy who underwent major surgery for pelvic organ prolapse.
The investigators treated one side of the atrophic vaginal wall specimen with the microablative fractional CO2 laser and left the contralateral area untreated as a control. They documented that laser therapy restored the vaginal squamous stratified epithelium, with enhanced storage of glycogen in the epithelial cells and shedding of glycogen-rich cells at the epithelial surface. In the connective tissue, activated fibroblasts synthesized new collagen-laden extracellular matrix. All this was accomplished without damage to adjacent untreated tissue (Menopause 2015 Aug;22(8):845-9).
In a single-arm study performed by many of the same Italian investigators, 77 postmenopausal women underwent a course of fractional microablative CO2 laser therapy because they experienced painful sexual intercourse due to vulvovaginal atrophy. At 12 weeks of followup, the group reported significant improvement in sexual function and satisfaction with their sexual life as measured by the Short Form-12 and the Female Sexual Function Index. Self-rated scores of vaginal burning, dryness, and itching improved significantly, as did complaints of pain during intercourse or urination. At baseline, 20 of the 77 women were not sexually active due to the severity of their vulvovaginal atrophy; at followup, 17 of the 20 had reestablished sexual activity (Climacteric. 2015 Apr;18(2):219-25).
Dr. Bjerring also highlighted an American study, again single-arm, in which 27 women with genitourinary syndrome of menopause were examined at baseline and again 3 months after their third and final treatment with a fractional CO2 laser. At follow up, 26 of the 27 pronounced themselves satisfied or extremely satisfied with the results, with significant improvement in the same outcome measures used in the Italian study. At follow up, 25 of the women had an increase in comfortable dilator size (Menopause 2016 Oct;23(10):1102-7).
Dr. Bjerring reported having no financial conflicts of interest regarding his presentation.
VIENNA – Vaginal rejuvenation is a major practice growth opportunity for dermatologists who have expertise with lasers, Peter Bjerring, MD, said at the annual congress of the European Academy of Dermatology and Venereology.
The rejuvenation procedures involve heating the connective tissue of the vaginal wall to 40-42 C in order to stimulate tissue remodeling with formation of new collagen and elastic fibers. The evidence base for vaginal rejuvenation using a variety of noninvasive energy-based devices developed for vaginal use isn’t nearly as extensive as it is for skin rejuvenation using lasers. Research is beginning to increase for this indication, but current results are primarily limited to small single-arm studies based on self-reported improvements. Further, studies don’t compare outcomes vs another treatment, such as estrogen cream.
Despite the minimal evidence base for laser procedures, “feminine rejuvenation is becoming very popular. These are (women) who might present to a dermatologist or to a gynecologist,” observed Dr. Bjerring, medical director and head of the Skin and Laser Center at Malholm (Denmark) Hospital.
Minimally ablative or nonablative fractional laser therapy for vaginal rejuvenation requires no anesthesia and no downtime. The lasers being used for this purpose are similar to those already used most often for skin resurfacing and rejuvenation of the face and neck: fractional CO2 lasers at the 10,600-nm wavelength, such as the MonaLisa Touch or FemTouch, and 2,940-nm nonablative erbium:yttrium-aluminum-garnet (Er:YAG) lasers such as the IntimaLase. A course of treatment with these devices typically consists of three, 15-minute sessions at 4- to 6-week intervals.
Dr. Bjerring noted that this mechanism of benefit has been demonstrated by Italian investigators who conducted a histologic study of the effects of microablative fractional CO2 laser therapy on ex vivo specimens of atrophic vaginal tissue obtained from women with vulvovaginal atrophy who underwent major surgery for pelvic organ prolapse.
The investigators treated one side of the atrophic vaginal wall specimen with the microablative fractional CO2 laser and left the contralateral area untreated as a control. They documented that laser therapy restored the vaginal squamous stratified epithelium, with enhanced storage of glycogen in the epithelial cells and shedding of glycogen-rich cells at the epithelial surface. In the connective tissue, activated fibroblasts synthesized new collagen-laden extracellular matrix. All this was accomplished without damage to adjacent untreated tissue (Menopause 2015 Aug;22(8):845-9).
In a single-arm study performed by many of the same Italian investigators, 77 postmenopausal women underwent a course of fractional microablative CO2 laser therapy because they experienced painful sexual intercourse due to vulvovaginal atrophy. At 12 weeks of followup, the group reported significant improvement in sexual function and satisfaction with their sexual life as measured by the Short Form-12 and the Female Sexual Function Index. Self-rated scores of vaginal burning, dryness, and itching improved significantly, as did complaints of pain during intercourse or urination. At baseline, 20 of the 77 women were not sexually active due to the severity of their vulvovaginal atrophy; at followup, 17 of the 20 had reestablished sexual activity (Climacteric. 2015 Apr;18(2):219-25).
Dr. Bjerring also highlighted an American study, again single-arm, in which 27 women with genitourinary syndrome of menopause were examined at baseline and again 3 months after their third and final treatment with a fractional CO2 laser. At follow up, 26 of the 27 pronounced themselves satisfied or extremely satisfied with the results, with significant improvement in the same outcome measures used in the Italian study. At follow up, 25 of the women had an increase in comfortable dilator size (Menopause 2016 Oct;23(10):1102-7).
Dr. Bjerring reported having no financial conflicts of interest regarding his presentation.
EXPERT ANALYSIS FROM THE EADV CONGRESS
Cosmetic Corner: Dermatologists Weigh in on Skin-Lightening Agents
To improve patient care and outcomes, leading dermatologists offered their recommendations on skin-lightening agents. Consideration must be given to:
- Even Better Clinical Dark Spot Corrector
Clinique Laboratories, LLC
Recommended by Gary Goldenberg, MD, New York, New York
- Lytera Skin Brightening Complex
SkinMedica, an Allergan compan
“It contains vitamin C, niacinamide, retinol, and licorice root extract to help lighten the skin and improve texture without hydroquinone.”—Anthony M. Rossi, MD, New York, New York
- Meladerm
Civant Skin Care
“This is an excellent hydroquinone-free cream for treating postinflammatory hyperpigmentation and melasma. It contains a combination of ingredients known to inhibit various steps along the melanogenesis pathway, such as retinyl palmitate, licorice extract, and arbutin, as well as lactic, kojic, and ascorbic acids.”—Cherise M. Levi, DO, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, dry shampoos, athlete’s foot treatments, and face scrubs will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
To improve patient care and outcomes, leading dermatologists offered their recommendations on skin-lightening agents. Consideration must be given to:
- Even Better Clinical Dark Spot Corrector
Clinique Laboratories, LLC
Recommended by Gary Goldenberg, MD, New York, New York
- Lytera Skin Brightening Complex
SkinMedica, an Allergan compan
“It contains vitamin C, niacinamide, retinol, and licorice root extract to help lighten the skin and improve texture without hydroquinone.”—Anthony M. Rossi, MD, New York, New York
- Meladerm
Civant Skin Care
“This is an excellent hydroquinone-free cream for treating postinflammatory hyperpigmentation and melasma. It contains a combination of ingredients known to inhibit various steps along the melanogenesis pathway, such as retinyl palmitate, licorice extract, and arbutin, as well as lactic, kojic, and ascorbic acids.”—Cherise M. Levi, DO, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, dry shampoos, athlete’s foot treatments, and face scrubs will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
To improve patient care and outcomes, leading dermatologists offered their recommendations on skin-lightening agents. Consideration must be given to:
- Even Better Clinical Dark Spot Corrector
Clinique Laboratories, LLC
Recommended by Gary Goldenberg, MD, New York, New York
- Lytera Skin Brightening Complex
SkinMedica, an Allergan compan
“It contains vitamin C, niacinamide, retinol, and licorice root extract to help lighten the skin and improve texture without hydroquinone.”—Anthony M. Rossi, MD, New York, New York
- Meladerm
Civant Skin Care
“This is an excellent hydroquinone-free cream for treating postinflammatory hyperpigmentation and melasma. It contains a combination of ingredients known to inhibit various steps along the melanogenesis pathway, such as retinyl palmitate, licorice extract, and arbutin, as well as lactic, kojic, and ascorbic acids.”—Cherise M. Levi, DO, New York, New York
Cutis invites readers to send us their recommendations. Cleansing devices, dry shampoos, athlete’s foot treatments, and face scrubs will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
Wheat in skin care
Gluten-free food products have inundated the marketplace in recent years as the food industry has responded to greater awareness of celiac sprue disease and wheat sensitivity. Gluten is the primary form of wheat protein.
Wheat is a versatile and globally popular member of the Poaceae or Gramineae family known as grasses; it is of the Triticum species with Triticum aestivum and Triticum vulgare being particularly pervasive.1 In traditional Iranian medicine, the topical application of wheat germ oil has been used to treat psoriasis.2
Moisturization
In 2008, N. Akhtar and Y. Yazan investigated the effects of a stable emulsion containing two ingredients included to exert anti-aging effects: vitamin C and wheat protein. The antioxidant vitamin C was entrapped in the inner aqueous phase of the water-in-oil-in-water emulsion while wheat protein was incorporated in the oily phase. The investigators prepared and applied stable emulsions to the cheeks of 11 volunteers over 4 weeks, finding that the formulation increased skin moisture.7
Melanoma and wheat supplementation
Demidov et al. reported in 2008 on a randomized, pilot, phase II clinical trial to assess the impact of the adjuvant use of a fermented wheat germ extract nutraceutical (Avemar) in high-risk cutaneous melanoma patients. Investigators compared the efficacy of dacarbazine-based adjuvant chemotherapy on survival parameters of melanoma patients to that of the identical treatment supplemented with a 1-year administration of fermented wheat germ extract nutraceutical (FWGE), which is generally recognized as safe. They reported that after a 7-year follow-up, significant differences favoring the nutraceutical group were observed in progression-free and overall survival. Including nutraceutical as an adjuvant treatment for such patients was recommended by the authors.8
Telekes et al. noted that nutraceutical is registered as a special nutriment for cancer patients in Hungary that has exhibited potent anticancer activity on cell lines and immunomodulatory activity in vivo.9 In 2005, Boros et al. reported that orally administered FWGE suppressed metastatic tumor dissemination and proliferation during and after chemotherapy, surgery, or radiation, with benefits seen in some human cancers and cultured cells as well as some autoimmune disorders and in chemical carcinogenesis prevention.10
Hypersensitivity and allergic reactions
The risks of sensitization to topical wheat proteins are thought to be higher in patients with atopic dermatitis, who have an impaired skin barrier.1 Indeed, Codreanu et al. have suggested that topical products containing food proteins of known allergenicity (including wheat) are contraindicated for neonates and infants with atopic dermatitis.11
In 2015, Bonciolini et al. studied 17 patients (13 females and 4 males, median age 36 years) with nonceliac gluten sensitivity presenting with nonspecific skin lesions. The eczema-, psoriasis-, or dermatitis herpetiformis-like lesions on the extensor surfaces of the upper and lower limbs, especially, were confirmed histologically, but immunopathological evaluations revealed pervasive C3 deposits along the dermoepidermal junction in a microgranular/granular pattern (82%). Notably, all of the patients improved markedly after initiating a gluten-free diet.12
In 2014, Fukutomi et al. conducted a case-control study of Japanese women aged 20-54 years (157 cases) who self-reported wheat allergy to ascertain the epidemiologic relationship between food allergy to wheat after exposure to facial soaps containing hydrolyzed wheat protein. There were 449 age-matched controls without wheat allergy. Participants answered a Web-based questionnaire about their use of skin and hair care products. The investigators found that current use of the facial soap Cha no Shizuku (Drop of Tea), which contains hydrolyzed wheat protein, was significantly linked to a greater risk of wheat allergy, with use of the soap more frequent in consumers whose wheat allergy had newly emerged (11% vs. 6% in controls).13
Cha no Shizuku had earlier been implicated in provoking hundreds of cases of allergic reactions between 2009 and June 2013. R. Teshima noted that the soap contains acid-hydrolyzed wheat protein produced from gluten after partial hydrolysis with hydrogen chloride at 95 ° C for 40 minutes.14
It is worth noting that case reports of allergic reactions to facial soap containing hydrolyzed wheat protein continue to appear. Iseki et al. described in 2014 a 38-year-old woman who experienced irregular headaches, sleepiness, and an episode of facial rash eruption after daily use for about 1 year of a facial soap with hydrolyzed wheat proteins (Glupearl 19s, which is also used in Cha no Shizuku). The investigators added that the patient’s serum contained wheat-specific IgE antibodies. Symptoms disappeared after the patient abstained from wheat.15
In 2012, Tammaro studied cutaneous hypersensitivity to gluten in 14 female patients (aged 12-60 years) with celiac disease who presented with eczema on the face, neck, and arms, after topical application of gluten-containing emollient cream, bath or face powder, or contact with foods containing wheat and durum. Five of the patients tested positive for wheat and durum wheat, while none of the 14 control patients tested positive. Improvement in cutaneous lesions, with no relapses during a 6-month follow-up, resulted when these patients used gluten-free cream and bath powder, and wore gloves before handling wheat-containing food.16
In 2011, Celakovská et al. studied the impact of wheat allergy in 179 adults with atopic eczema (128 females, 51 males; average age 26 years), using open exposure and double-blind, placebo-controlled food challenge tests, as well as specific serum IgE, skin prick, and atopy patch tests. The double-blind, placebo-controlled food challenge test showed that the course of atopic eczema was exacerbated by wheat allergy in eight patients (4.5%). A positive trend revealing that the course of atopic eczema was impacted by wheat allergy emerged during follow-up (at 3, 6, 9, and 12 months).17
Contact urticaria also has been reported to have been induced by hydrolyzed wheat proteins in cosmetics and is notable for the potential to precede food allergies.2,3 A wide variety of protein hydrolysates found in hair products have been associated with inducing contact urticaria, particularly in patients with atopic dermatitis.4
In 2006, Laurière et al. studied nine female patients without common wheat allergy who presented with contact urticaria to cosmetics containing hydrolyzed wheat proteins; six also had experienced generalized urticaria or anaphylaxis in response to foods containing such wheat proteins. Analyses revealed the importance of hydrolysis in augmenting the allergenicity of wheat proteins through contact or consumption.18 Immediate contact urticaria in reaction to hydrolyzed wheat protein in topical products also has been reported in a child.19
Conclusion
Can the presence of wheat hydrolysates in personal care products adversely affect a patient with celiac sprue or wheat sensitivity? The short answer appears to be “yes.” Given the use of hydrolyzed wheat protein in various skin care products, it is important that consumers who have celiac disease or sensitivity to wheat be advised to avoid skin care formulations with such active ingredients. On the positive side of the wheat ledger, there are some indications (albeit in very limited research) that the plant protein may impart beneficial health effects. Much more research is necessary to delineate the full impact of wheat on skin health.
I thank my dermatologist colleague Sharon E. Jacob, MD, at the University of Miami, for suggesting this topic.
References
1. Dermatitis. 2013 Nov-Dec;24(6):291-5.
2. Iran J Med Sci. 2016 May;41(3 Suppl):S54.
3. Contact Dermatitis. 2007 Feb;56(2):119-20.
4. Ann Dermatol Venereol. 2010 Apr;137(4):281-4.
5. Allergy. 1998 Nov;53(11):1078-82.
6. J Drugs Dermatol. 2013 Sep;12(9 Suppl):s133-6.
7. Pak J Pharm Sci. 2008 Jan;21(1):45-50.
8. Cancer Biother Radiopharm. 2008 Aug;23(4):477-82.
9. Nutr Cancer. 2009;61(6):891-9.
10. Ann N Y Acad Sci. 2005 Jun;1051:529-42.
11. Eur Ann Allergy Clin Immunol. 2006 Apr;38(4):126-30.
12. Nutrients. 2015 Sep 15;7(9):7798-805.
13. Allergy. 2014 Oct;69(10):1405-11.
14. Yakugaku Zasshi. 2014;134(1):33-8.
15. Intern Med. 2014;53(2):151-4.
16. Dermatitis. 2012 Sep-Oct;23(5):220-1.
17. Acta Medica (Hradec Kralove). 2011;54(4):157-62.
18. Contact Dermatitis. 2006 May;54(5):283-9.
19. Contact Dermatitis. 2013 Jun;68(6):379-80.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Gluten-free food products have inundated the marketplace in recent years as the food industry has responded to greater awareness of celiac sprue disease and wheat sensitivity. Gluten is the primary form of wheat protein.
Wheat is a versatile and globally popular member of the Poaceae or Gramineae family known as grasses; it is of the Triticum species with Triticum aestivum and Triticum vulgare being particularly pervasive.1 In traditional Iranian medicine, the topical application of wheat germ oil has been used to treat psoriasis.2
Moisturization
In 2008, N. Akhtar and Y. Yazan investigated the effects of a stable emulsion containing two ingredients included to exert anti-aging effects: vitamin C and wheat protein. The antioxidant vitamin C was entrapped in the inner aqueous phase of the water-in-oil-in-water emulsion while wheat protein was incorporated in the oily phase. The investigators prepared and applied stable emulsions to the cheeks of 11 volunteers over 4 weeks, finding that the formulation increased skin moisture.7
Melanoma and wheat supplementation
Demidov et al. reported in 2008 on a randomized, pilot, phase II clinical trial to assess the impact of the adjuvant use of a fermented wheat germ extract nutraceutical (Avemar) in high-risk cutaneous melanoma patients. Investigators compared the efficacy of dacarbazine-based adjuvant chemotherapy on survival parameters of melanoma patients to that of the identical treatment supplemented with a 1-year administration of fermented wheat germ extract nutraceutical (FWGE), which is generally recognized as safe. They reported that after a 7-year follow-up, significant differences favoring the nutraceutical group were observed in progression-free and overall survival. Including nutraceutical as an adjuvant treatment for such patients was recommended by the authors.8
Telekes et al. noted that nutraceutical is registered as a special nutriment for cancer patients in Hungary that has exhibited potent anticancer activity on cell lines and immunomodulatory activity in vivo.9 In 2005, Boros et al. reported that orally administered FWGE suppressed metastatic tumor dissemination and proliferation during and after chemotherapy, surgery, or radiation, with benefits seen in some human cancers and cultured cells as well as some autoimmune disorders and in chemical carcinogenesis prevention.10
Hypersensitivity and allergic reactions
The risks of sensitization to topical wheat proteins are thought to be higher in patients with atopic dermatitis, who have an impaired skin barrier.1 Indeed, Codreanu et al. have suggested that topical products containing food proteins of known allergenicity (including wheat) are contraindicated for neonates and infants with atopic dermatitis.11
In 2015, Bonciolini et al. studied 17 patients (13 females and 4 males, median age 36 years) with nonceliac gluten sensitivity presenting with nonspecific skin lesions. The eczema-, psoriasis-, or dermatitis herpetiformis-like lesions on the extensor surfaces of the upper and lower limbs, especially, were confirmed histologically, but immunopathological evaluations revealed pervasive C3 deposits along the dermoepidermal junction in a microgranular/granular pattern (82%). Notably, all of the patients improved markedly after initiating a gluten-free diet.12
In 2014, Fukutomi et al. conducted a case-control study of Japanese women aged 20-54 years (157 cases) who self-reported wheat allergy to ascertain the epidemiologic relationship between food allergy to wheat after exposure to facial soaps containing hydrolyzed wheat protein. There were 449 age-matched controls without wheat allergy. Participants answered a Web-based questionnaire about their use of skin and hair care products. The investigators found that current use of the facial soap Cha no Shizuku (Drop of Tea), which contains hydrolyzed wheat protein, was significantly linked to a greater risk of wheat allergy, with use of the soap more frequent in consumers whose wheat allergy had newly emerged (11% vs. 6% in controls).13
Cha no Shizuku had earlier been implicated in provoking hundreds of cases of allergic reactions between 2009 and June 2013. R. Teshima noted that the soap contains acid-hydrolyzed wheat protein produced from gluten after partial hydrolysis with hydrogen chloride at 95 ° C for 40 minutes.14
It is worth noting that case reports of allergic reactions to facial soap containing hydrolyzed wheat protein continue to appear. Iseki et al. described in 2014 a 38-year-old woman who experienced irregular headaches, sleepiness, and an episode of facial rash eruption after daily use for about 1 year of a facial soap with hydrolyzed wheat proteins (Glupearl 19s, which is also used in Cha no Shizuku). The investigators added that the patient’s serum contained wheat-specific IgE antibodies. Symptoms disappeared after the patient abstained from wheat.15
In 2012, Tammaro studied cutaneous hypersensitivity to gluten in 14 female patients (aged 12-60 years) with celiac disease who presented with eczema on the face, neck, and arms, after topical application of gluten-containing emollient cream, bath or face powder, or contact with foods containing wheat and durum. Five of the patients tested positive for wheat and durum wheat, while none of the 14 control patients tested positive. Improvement in cutaneous lesions, with no relapses during a 6-month follow-up, resulted when these patients used gluten-free cream and bath powder, and wore gloves before handling wheat-containing food.16
In 2011, Celakovská et al. studied the impact of wheat allergy in 179 adults with atopic eczema (128 females, 51 males; average age 26 years), using open exposure and double-blind, placebo-controlled food challenge tests, as well as specific serum IgE, skin prick, and atopy patch tests. The double-blind, placebo-controlled food challenge test showed that the course of atopic eczema was exacerbated by wheat allergy in eight patients (4.5%). A positive trend revealing that the course of atopic eczema was impacted by wheat allergy emerged during follow-up (at 3, 6, 9, and 12 months).17
Contact urticaria also has been reported to have been induced by hydrolyzed wheat proteins in cosmetics and is notable for the potential to precede food allergies.2,3 A wide variety of protein hydrolysates found in hair products have been associated with inducing contact urticaria, particularly in patients with atopic dermatitis.4
In 2006, Laurière et al. studied nine female patients without common wheat allergy who presented with contact urticaria to cosmetics containing hydrolyzed wheat proteins; six also had experienced generalized urticaria or anaphylaxis in response to foods containing such wheat proteins. Analyses revealed the importance of hydrolysis in augmenting the allergenicity of wheat proteins through contact or consumption.18 Immediate contact urticaria in reaction to hydrolyzed wheat protein in topical products also has been reported in a child.19
Conclusion
Can the presence of wheat hydrolysates in personal care products adversely affect a patient with celiac sprue or wheat sensitivity? The short answer appears to be “yes.” Given the use of hydrolyzed wheat protein in various skin care products, it is important that consumers who have celiac disease or sensitivity to wheat be advised to avoid skin care formulations with such active ingredients. On the positive side of the wheat ledger, there are some indications (albeit in very limited research) that the plant protein may impart beneficial health effects. Much more research is necessary to delineate the full impact of wheat on skin health.
I thank my dermatologist colleague Sharon E. Jacob, MD, at the University of Miami, for suggesting this topic.
References
1. Dermatitis. 2013 Nov-Dec;24(6):291-5.
2. Iran J Med Sci. 2016 May;41(3 Suppl):S54.
3. Contact Dermatitis. 2007 Feb;56(2):119-20.
4. Ann Dermatol Venereol. 2010 Apr;137(4):281-4.
5. Allergy. 1998 Nov;53(11):1078-82.
6. J Drugs Dermatol. 2013 Sep;12(9 Suppl):s133-6.
7. Pak J Pharm Sci. 2008 Jan;21(1):45-50.
8. Cancer Biother Radiopharm. 2008 Aug;23(4):477-82.
9. Nutr Cancer. 2009;61(6):891-9.
10. Ann N Y Acad Sci. 2005 Jun;1051:529-42.
11. Eur Ann Allergy Clin Immunol. 2006 Apr;38(4):126-30.
12. Nutrients. 2015 Sep 15;7(9):7798-805.
13. Allergy. 2014 Oct;69(10):1405-11.
14. Yakugaku Zasshi. 2014;134(1):33-8.
15. Intern Med. 2014;53(2):151-4.
16. Dermatitis. 2012 Sep-Oct;23(5):220-1.
17. Acta Medica (Hradec Kralove). 2011;54(4):157-62.
18. Contact Dermatitis. 2006 May;54(5):283-9.
19. Contact Dermatitis. 2013 Jun;68(6):379-80.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Gluten-free food products have inundated the marketplace in recent years as the food industry has responded to greater awareness of celiac sprue disease and wheat sensitivity. Gluten is the primary form of wheat protein.
Wheat is a versatile and globally popular member of the Poaceae or Gramineae family known as grasses; it is of the Triticum species with Triticum aestivum and Triticum vulgare being particularly pervasive.1 In traditional Iranian medicine, the topical application of wheat germ oil has been used to treat psoriasis.2
Moisturization
In 2008, N. Akhtar and Y. Yazan investigated the effects of a stable emulsion containing two ingredients included to exert anti-aging effects: vitamin C and wheat protein. The antioxidant vitamin C was entrapped in the inner aqueous phase of the water-in-oil-in-water emulsion while wheat protein was incorporated in the oily phase. The investigators prepared and applied stable emulsions to the cheeks of 11 volunteers over 4 weeks, finding that the formulation increased skin moisture.7
Melanoma and wheat supplementation
Demidov et al. reported in 2008 on a randomized, pilot, phase II clinical trial to assess the impact of the adjuvant use of a fermented wheat germ extract nutraceutical (Avemar) in high-risk cutaneous melanoma patients. Investigators compared the efficacy of dacarbazine-based adjuvant chemotherapy on survival parameters of melanoma patients to that of the identical treatment supplemented with a 1-year administration of fermented wheat germ extract nutraceutical (FWGE), which is generally recognized as safe. They reported that after a 7-year follow-up, significant differences favoring the nutraceutical group were observed in progression-free and overall survival. Including nutraceutical as an adjuvant treatment for such patients was recommended by the authors.8
Telekes et al. noted that nutraceutical is registered as a special nutriment for cancer patients in Hungary that has exhibited potent anticancer activity on cell lines and immunomodulatory activity in vivo.9 In 2005, Boros et al. reported that orally administered FWGE suppressed metastatic tumor dissemination and proliferation during and after chemotherapy, surgery, or radiation, with benefits seen in some human cancers and cultured cells as well as some autoimmune disorders and in chemical carcinogenesis prevention.10
Hypersensitivity and allergic reactions
The risks of sensitization to topical wheat proteins are thought to be higher in patients with atopic dermatitis, who have an impaired skin barrier.1 Indeed, Codreanu et al. have suggested that topical products containing food proteins of known allergenicity (including wheat) are contraindicated for neonates and infants with atopic dermatitis.11
In 2015, Bonciolini et al. studied 17 patients (13 females and 4 males, median age 36 years) with nonceliac gluten sensitivity presenting with nonspecific skin lesions. The eczema-, psoriasis-, or dermatitis herpetiformis-like lesions on the extensor surfaces of the upper and lower limbs, especially, were confirmed histologically, but immunopathological evaluations revealed pervasive C3 deposits along the dermoepidermal junction in a microgranular/granular pattern (82%). Notably, all of the patients improved markedly after initiating a gluten-free diet.12
In 2014, Fukutomi et al. conducted a case-control study of Japanese women aged 20-54 years (157 cases) who self-reported wheat allergy to ascertain the epidemiologic relationship between food allergy to wheat after exposure to facial soaps containing hydrolyzed wheat protein. There were 449 age-matched controls without wheat allergy. Participants answered a Web-based questionnaire about their use of skin and hair care products. The investigators found that current use of the facial soap Cha no Shizuku (Drop of Tea), which contains hydrolyzed wheat protein, was significantly linked to a greater risk of wheat allergy, with use of the soap more frequent in consumers whose wheat allergy had newly emerged (11% vs. 6% in controls).13
Cha no Shizuku had earlier been implicated in provoking hundreds of cases of allergic reactions between 2009 and June 2013. R. Teshima noted that the soap contains acid-hydrolyzed wheat protein produced from gluten after partial hydrolysis with hydrogen chloride at 95 ° C for 40 minutes.14
It is worth noting that case reports of allergic reactions to facial soap containing hydrolyzed wheat protein continue to appear. Iseki et al. described in 2014 a 38-year-old woman who experienced irregular headaches, sleepiness, and an episode of facial rash eruption after daily use for about 1 year of a facial soap with hydrolyzed wheat proteins (Glupearl 19s, which is also used in Cha no Shizuku). The investigators added that the patient’s serum contained wheat-specific IgE antibodies. Symptoms disappeared after the patient abstained from wheat.15
In 2012, Tammaro studied cutaneous hypersensitivity to gluten in 14 female patients (aged 12-60 years) with celiac disease who presented with eczema on the face, neck, and arms, after topical application of gluten-containing emollient cream, bath or face powder, or contact with foods containing wheat and durum. Five of the patients tested positive for wheat and durum wheat, while none of the 14 control patients tested positive. Improvement in cutaneous lesions, with no relapses during a 6-month follow-up, resulted when these patients used gluten-free cream and bath powder, and wore gloves before handling wheat-containing food.16
In 2011, Celakovská et al. studied the impact of wheat allergy in 179 adults with atopic eczema (128 females, 51 males; average age 26 years), using open exposure and double-blind, placebo-controlled food challenge tests, as well as specific serum IgE, skin prick, and atopy patch tests. The double-blind, placebo-controlled food challenge test showed that the course of atopic eczema was exacerbated by wheat allergy in eight patients (4.5%). A positive trend revealing that the course of atopic eczema was impacted by wheat allergy emerged during follow-up (at 3, 6, 9, and 12 months).17
Contact urticaria also has been reported to have been induced by hydrolyzed wheat proteins in cosmetics and is notable for the potential to precede food allergies.2,3 A wide variety of protein hydrolysates found in hair products have been associated with inducing contact urticaria, particularly in patients with atopic dermatitis.4
In 2006, Laurière et al. studied nine female patients without common wheat allergy who presented with contact urticaria to cosmetics containing hydrolyzed wheat proteins; six also had experienced generalized urticaria or anaphylaxis in response to foods containing such wheat proteins. Analyses revealed the importance of hydrolysis in augmenting the allergenicity of wheat proteins through contact or consumption.18 Immediate contact urticaria in reaction to hydrolyzed wheat protein in topical products also has been reported in a child.19
Conclusion
Can the presence of wheat hydrolysates in personal care products adversely affect a patient with celiac sprue or wheat sensitivity? The short answer appears to be “yes.” Given the use of hydrolyzed wheat protein in various skin care products, it is important that consumers who have celiac disease or sensitivity to wheat be advised to avoid skin care formulations with such active ingredients. On the positive side of the wheat ledger, there are some indications (albeit in very limited research) that the plant protein may impart beneficial health effects. Much more research is necessary to delineate the full impact of wheat on skin health.
I thank my dermatologist colleague Sharon E. Jacob, MD, at the University of Miami, for suggesting this topic.
References
1. Dermatitis. 2013 Nov-Dec;24(6):291-5.
2. Iran J Med Sci. 2016 May;41(3 Suppl):S54.
3. Contact Dermatitis. 2007 Feb;56(2):119-20.
4. Ann Dermatol Venereol. 2010 Apr;137(4):281-4.
5. Allergy. 1998 Nov;53(11):1078-82.
6. J Drugs Dermatol. 2013 Sep;12(9 Suppl):s133-6.
7. Pak J Pharm Sci. 2008 Jan;21(1):45-50.
8. Cancer Biother Radiopharm. 2008 Aug;23(4):477-82.
9. Nutr Cancer. 2009;61(6):891-9.
10. Ann N Y Acad Sci. 2005 Jun;1051:529-42.
11. Eur Ann Allergy Clin Immunol. 2006 Apr;38(4):126-30.
12. Nutrients. 2015 Sep 15;7(9):7798-805.
13. Allergy. 2014 Oct;69(10):1405-11.
14. Yakugaku Zasshi. 2014;134(1):33-8.
15. Intern Med. 2014;53(2):151-4.
16. Dermatitis. 2012 Sep-Oct;23(5):220-1.
17. Acta Medica (Hradec Kralove). 2011;54(4):157-62.
18. Contact Dermatitis. 2006 May;54(5):283-9.
19. Contact Dermatitis. 2013 Jun;68(6):379-80.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Cannulas versus needles for soft tissue filler injection
With loss of deep fat pad compartments and bony resorption with normal aging, soft tissue fillers have become a mainstay in minimally invasive aesthetic treatments. Preference of injecting with a needle versus a cannula is often user and training dependent. Blunt-tipped cannulas may provide a lower risk of bruising, as well as potentially devastating complications such as intravascular occlusion that can lead to skin necrosis and blindness. Even for advanced injectors, however, cannula use may portend a learning curve if the clinicians are used to injecting with needles.
A recently published observational study using cadaver heads looked at precision in supraperiosteal placement with a sharp needle compared with a blunt tipped cannula.1 The investigators injected dye material with soft-tissue fillers at different aesthetic facial sites on the supraperiosteum, then observed the placement of dye and filler after dissection. In this study, the placement of product was more precise with the cannulas. The filler was injected on the periosteum with a needle. Some of the filler then migrated along the trajectory of the needle path back toward the epidermis, ending up in multiple tissue layers. So there was more extrusion of the filler in the superficial layers with a needle without a retrograde injection technique. Even with the needle tip on the periosteum and no movement of the needle, the needle technique showed a higher risk of intra-arterial injections. This study is limited by the fact that in vivo circumstances could potentially alter the outcome, as could user injection technique.
Cannulas should be highly considered in any deep tissue compartment, but especially in more advanced injection technique areas, such as the nasal dorsum. Another cadaver study from Thailand showed that the anatomy of the dorsal nasal artery is not consistent.2 It is injection into this artery that can lead to blindness via flow to the ophthalmic artery. The study showed that both the diameter of the artery and the presence of a single or bilateral dorsal nasal artery varied. The dorsal nasal artery travels in the subcutaneous tissue layer of the nasal dorsum on the transverse nasalis muscle and its midline nasal aponeurosis, which connects the muscles on both sides. Bilateral dorsal nasal arteries were present in 34% of the specimens. A single and large dorsal nasal artery was present in 28%.
Needles are still useful in some places where precise small aliquot touch-up of filler placement is needed or where it may be difficult to reach with the cannula without making an additional portal of entry. More viscous fillers such as calcium hydroxylapatite and poly-L-lactic acid can be difficult to inject through a cannula and require a needle for injection. More superficially, small 30- or 32-gauge needles are also required for the injection of certain hyaluronic acid fillers in the superficial dermis for more etched lines.
The risk of arterial wall perforation and emboli with cannulas is lower, but these complications can still occur. The risk increases with a perpendicular angle between the artery and the cannula, thus slow small aliquot injection technique along with knowledge of anatomy is essential.3 While both needles and cannulas are useful in practice and achieve excellent cosmetic results, cannula use in the deeper compartments among practitioners is encouraged to minimize complications.
References
1. Aesthet Surg J. 2016 Dec 16. pii: sjw220.
2. Aesthetic Plast Surg. 2016 Dec 28. doi: 10.1007/s00266-016-0756-0.
3. Aesthetic Plast Surg. 2016 Dec 23. doi: 10.1007/s00266-016-0725-7.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
With loss of deep fat pad compartments and bony resorption with normal aging, soft tissue fillers have become a mainstay in minimally invasive aesthetic treatments. Preference of injecting with a needle versus a cannula is often user and training dependent. Blunt-tipped cannulas may provide a lower risk of bruising, as well as potentially devastating complications such as intravascular occlusion that can lead to skin necrosis and blindness. Even for advanced injectors, however, cannula use may portend a learning curve if the clinicians are used to injecting with needles.
A recently published observational study using cadaver heads looked at precision in supraperiosteal placement with a sharp needle compared with a blunt tipped cannula.1 The investigators injected dye material with soft-tissue fillers at different aesthetic facial sites on the supraperiosteum, then observed the placement of dye and filler after dissection. In this study, the placement of product was more precise with the cannulas. The filler was injected on the periosteum with a needle. Some of the filler then migrated along the trajectory of the needle path back toward the epidermis, ending up in multiple tissue layers. So there was more extrusion of the filler in the superficial layers with a needle without a retrograde injection technique. Even with the needle tip on the periosteum and no movement of the needle, the needle technique showed a higher risk of intra-arterial injections. This study is limited by the fact that in vivo circumstances could potentially alter the outcome, as could user injection technique.
Cannulas should be highly considered in any deep tissue compartment, but especially in more advanced injection technique areas, such as the nasal dorsum. Another cadaver study from Thailand showed that the anatomy of the dorsal nasal artery is not consistent.2 It is injection into this artery that can lead to blindness via flow to the ophthalmic artery. The study showed that both the diameter of the artery and the presence of a single or bilateral dorsal nasal artery varied. The dorsal nasal artery travels in the subcutaneous tissue layer of the nasal dorsum on the transverse nasalis muscle and its midline nasal aponeurosis, which connects the muscles on both sides. Bilateral dorsal nasal arteries were present in 34% of the specimens. A single and large dorsal nasal artery was present in 28%.
Needles are still useful in some places where precise small aliquot touch-up of filler placement is needed or where it may be difficult to reach with the cannula without making an additional portal of entry. More viscous fillers such as calcium hydroxylapatite and poly-L-lactic acid can be difficult to inject through a cannula and require a needle for injection. More superficially, small 30- or 32-gauge needles are also required for the injection of certain hyaluronic acid fillers in the superficial dermis for more etched lines.
The risk of arterial wall perforation and emboli with cannulas is lower, but these complications can still occur. The risk increases with a perpendicular angle between the artery and the cannula, thus slow small aliquot injection technique along with knowledge of anatomy is essential.3 While both needles and cannulas are useful in practice and achieve excellent cosmetic results, cannula use in the deeper compartments among practitioners is encouraged to minimize complications.
References
1. Aesthet Surg J. 2016 Dec 16. pii: sjw220.
2. Aesthetic Plast Surg. 2016 Dec 28. doi: 10.1007/s00266-016-0756-0.
3. Aesthetic Plast Surg. 2016 Dec 23. doi: 10.1007/s00266-016-0725-7.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
With loss of deep fat pad compartments and bony resorption with normal aging, soft tissue fillers have become a mainstay in minimally invasive aesthetic treatments. Preference of injecting with a needle versus a cannula is often user and training dependent. Blunt-tipped cannulas may provide a lower risk of bruising, as well as potentially devastating complications such as intravascular occlusion that can lead to skin necrosis and blindness. Even for advanced injectors, however, cannula use may portend a learning curve if the clinicians are used to injecting with needles.
A recently published observational study using cadaver heads looked at precision in supraperiosteal placement with a sharp needle compared with a blunt tipped cannula.1 The investigators injected dye material with soft-tissue fillers at different aesthetic facial sites on the supraperiosteum, then observed the placement of dye and filler after dissection. In this study, the placement of product was more precise with the cannulas. The filler was injected on the periosteum with a needle. Some of the filler then migrated along the trajectory of the needle path back toward the epidermis, ending up in multiple tissue layers. So there was more extrusion of the filler in the superficial layers with a needle without a retrograde injection technique. Even with the needle tip on the periosteum and no movement of the needle, the needle technique showed a higher risk of intra-arterial injections. This study is limited by the fact that in vivo circumstances could potentially alter the outcome, as could user injection technique.
Cannulas should be highly considered in any deep tissue compartment, but especially in more advanced injection technique areas, such as the nasal dorsum. Another cadaver study from Thailand showed that the anatomy of the dorsal nasal artery is not consistent.2 It is injection into this artery that can lead to blindness via flow to the ophthalmic artery. The study showed that both the diameter of the artery and the presence of a single or bilateral dorsal nasal artery varied. The dorsal nasal artery travels in the subcutaneous tissue layer of the nasal dorsum on the transverse nasalis muscle and its midline nasal aponeurosis, which connects the muscles on both sides. Bilateral dorsal nasal arteries were present in 34% of the specimens. A single and large dorsal nasal artery was present in 28%.
Needles are still useful in some places where precise small aliquot touch-up of filler placement is needed or where it may be difficult to reach with the cannula without making an additional portal of entry. More viscous fillers such as calcium hydroxylapatite and poly-L-lactic acid can be difficult to inject through a cannula and require a needle for injection. More superficially, small 30- or 32-gauge needles are also required for the injection of certain hyaluronic acid fillers in the superficial dermis for more etched lines.
The risk of arterial wall perforation and emboli with cannulas is lower, but these complications can still occur. The risk increases with a perpendicular angle between the artery and the cannula, thus slow small aliquot injection technique along with knowledge of anatomy is essential.3 While both needles and cannulas are useful in practice and achieve excellent cosmetic results, cannula use in the deeper compartments among practitioners is encouraged to minimize complications.
References
1. Aesthet Surg J. 2016 Dec 16. pii: sjw220.
2. Aesthetic Plast Surg. 2016 Dec 28. doi: 10.1007/s00266-016-0756-0.
3. Aesthetic Plast Surg. 2016 Dec 23. doi: 10.1007/s00266-016-0725-7.
Dr. Wesley and Dr. Talakoub are co-contributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
Silver
Silver is a naturally occurring chemical element (number 47 on the periodic table) with the chemical symbol Ag, which is derived from the Latin word argentum (árgyros in Greek), from the Indo-European root “arg-,” meaning “shining,” “white,” or “gray.” It has been used for medical purposes since ancient times, with Hippocrates (circa 460-370 BCE) noting the beneficial healing and disease-altering activity of the element.1
In modern times, silver compounds – in metallic, nanocrystalline, and ionic formulations – have exhibited broad antibacterial activity and attracted interest for topical antiseptic use in wound dressings.2 Nanocrystalline silver dressings were introduced commercially as antimicrobial dressings in 1998.3 Silver is now used in dressings, catheters, cleansers, ophthalmic ointments, and myriad other medical products. In 2010 alone, an estimated 15 metric tons of silver were incorporated into medical products worldwide.4 It also is included in personal care products, textiles, and water purification devices. In topical skin preparations, the noble metal is included as colloidal silver (suspension of silver particles in an aqueous base) or nanosilver (as nanoparticles ranging from 1 nm to 100 nm in at least one dimension).5 Silver has been used to treat burns and wounds, but this discussion will be limited to acne, atopic dermatitis, and the anti-inflammatory response.
Anti-inflammatory uses
For several decades, noble metals including silver have been known to exert anti-inflammatory activity.7-11 In the case of silver, its anti-inflammatory properties appear to be mediated by its influence on the cytokine system. Silver nanoparticles inhibit the activity of interleukin-6 (IL-6), IL-12, IL-1beta, and tumor necrosis factor–alpha (TNF-alpha). This impact on the cytokine system is responsible for the impact of silver in demonstrably alleviating symptoms of rheumatoid arthritis.3
In 2004, Bhol et al. used dinitrochlorobenzene (DNCB) to induce allergic contact dermatitis in a guinea pig model, finding that topical nanocrystalline silver cream dose-dependently decreased erythema and as effectively as topical steroids and immunosuppressants.12 The next year, Bhol and Schechter showed that nanocrystalline silver suppressed allergic contact dermatitis in mice, inhibited TNF-alpha and IL-12 expression, and induced inflammatory cell apoptosis.13
In 2008, Nadworny et al. used a porcine contact dermatitis model to investigate the anti-inflammatory activity of nanocrystalline silver. They found that nanocrystalline silver treatments reduced DNCB-induced erythema and edema, promoted apoptosis in dermal cells, and diminished matrix metalloproteinase (MMP) and proinflammatory cytokine expression.3 The investigators speculated that the lower TNF-alpha observed in the silver-treated animals occurred due to apoptosis of the inflammatory cells.
Acne
Silver acts as a bactericidal and anti-inflammatory agent, without generating free radicals, as seen with benzoyl peroxide. Therefore, it is a compelling option for responding to the presence of Propionibacterium acnes. However, silver has not been approved by the Food and Drug Administration for this use. Even though formal acne studies have not been performed with silver sulfadiazine, it has long been used “off-label” for this purpose. As suggested above, the use of silver sulfadiazine for acne is limited by the risk of sulfa allergy. Cosmetic appearance and ease of use also are limiting factors, as silver sulfadiazine preparations are characterized by a thick, white pasty consistency. Other options for use of silver to treat acne include silver-containing cleansers and textiles.
Atopic dermatitis
A 2006 study in patients with atopic dermatitis demonstrated that silver-coated textiles could significantly diminish Staphylococcus aureus density after 2 days of wearing, with the effect enduring through the end of 7 days of treatment and then 1 week after removal of the textiles.14 Within 2 weeks, objective and subjective symptoms of atopic dermatitis were significantly enhanced in association with the silver-coated textiles, compared with cotton, without measurable adverse effects. A technology called Padycare incorporates silver into micromesh material (82% polyamide, 18% Lycra) used in clothing and bedding.15 As compared with topical formulations applied directly to the skin, textiles confer certain advantages such as preventing scratching and protecting against irritating substances and allergens. Washing of silver-infused textiles is a possible disadvantage, though, as the amount of silver lost from textiles can range from a 100% loss after four washings to less than a 1% loss.16 It also is important to note that there are concerns regarding the potential of silver to leak from textiles into the water supply, and eradicating the beneficial bacteria used to treat the water.
Conclusion
Despite centuries of medical use, silver has not been approved by the FDA for any medical applications. Further study, particularly in terms of safety and efficacy, is necessary. Nevertheless, it is used off-label before and after minimally invasive dermatologic procedures (for example, dermal filling, botulinum toxin injections, chemical peeling) because of its antimicrobial and anti-inflammatory activities as well as soothing qualities for facial skin and the skin barrier. Silver appears to be particularly suitable for use as an acne therapy option due to the low risk of bacterial resistance, lack of irritation, and its preservation of the skin barrier unlike harsher options such as retinoids, antibiotics, and benzoyl peroxide.
References
1. Adv Skin Wound Care. 2006 Nov-Dec;19(9):472-4.
2. Clin Infect Dis. 2009 Nov 15;49(10):1541-9.
3. Nanomedicine. 2008 Sep;4(3):241-51.
4. J Antimicrob Chemother. 2013 Jan;68(1):131-8.
5. Nanocrystalline Silver: Use in wound care, in Current Advances in the Medical Application of Nanotechnology (Manchester, England: Bentham Books, 2012, pp. 25-31).
6. Nanomedicine. 2013 Jan;9(1):39-54.
7. Jpn J Pharmacol. 1965 Jun;15(2):131-4.
8. J Allergy Clin Immunol. 1995 Aug;96(2):251-6.
9. Inflamm Res. 2003 Dec;52(12):487-501.
10. J Nutr Environ Med. 1997;7(4):295-305.
11. Clin Exp Pharmacol Physiol. 2000 Mar;27(3):139-44.
12. Clin Exp Dermatol. 2004 May;29(3):282-7.
13. Br J Dermatol. 2005 Jun;152(6):1235-42.
14. Curr Probl Dermatol. 2006;33:152-64.
15. J Eur Acad Dermatol Venereol. 2006 May;20(5):534-41.
16. Environ Sci Technol. 2008 Jun 1;42(11):4133-9.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis, Neutrogena, Philosophy, Topix, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Silver is a naturally occurring chemical element (number 47 on the periodic table) with the chemical symbol Ag, which is derived from the Latin word argentum (árgyros in Greek), from the Indo-European root “arg-,” meaning “shining,” “white,” or “gray.” It has been used for medical purposes since ancient times, with Hippocrates (circa 460-370 BCE) noting the beneficial healing and disease-altering activity of the element.1
In modern times, silver compounds – in metallic, nanocrystalline, and ionic formulations – have exhibited broad antibacterial activity and attracted interest for topical antiseptic use in wound dressings.2 Nanocrystalline silver dressings were introduced commercially as antimicrobial dressings in 1998.3 Silver is now used in dressings, catheters, cleansers, ophthalmic ointments, and myriad other medical products. In 2010 alone, an estimated 15 metric tons of silver were incorporated into medical products worldwide.4 It also is included in personal care products, textiles, and water purification devices. In topical skin preparations, the noble metal is included as colloidal silver (suspension of silver particles in an aqueous base) or nanosilver (as nanoparticles ranging from 1 nm to 100 nm in at least one dimension).5 Silver has been used to treat burns and wounds, but this discussion will be limited to acne, atopic dermatitis, and the anti-inflammatory response.
Anti-inflammatory uses
For several decades, noble metals including silver have been known to exert anti-inflammatory activity.7-11 In the case of silver, its anti-inflammatory properties appear to be mediated by its influence on the cytokine system. Silver nanoparticles inhibit the activity of interleukin-6 (IL-6), IL-12, IL-1beta, and tumor necrosis factor–alpha (TNF-alpha). This impact on the cytokine system is responsible for the impact of silver in demonstrably alleviating symptoms of rheumatoid arthritis.3
In 2004, Bhol et al. used dinitrochlorobenzene (DNCB) to induce allergic contact dermatitis in a guinea pig model, finding that topical nanocrystalline silver cream dose-dependently decreased erythema and as effectively as topical steroids and immunosuppressants.12 The next year, Bhol and Schechter showed that nanocrystalline silver suppressed allergic contact dermatitis in mice, inhibited TNF-alpha and IL-12 expression, and induced inflammatory cell apoptosis.13
In 2008, Nadworny et al. used a porcine contact dermatitis model to investigate the anti-inflammatory activity of nanocrystalline silver. They found that nanocrystalline silver treatments reduced DNCB-induced erythema and edema, promoted apoptosis in dermal cells, and diminished matrix metalloproteinase (MMP) and proinflammatory cytokine expression.3 The investigators speculated that the lower TNF-alpha observed in the silver-treated animals occurred due to apoptosis of the inflammatory cells.
Acne
Silver acts as a bactericidal and anti-inflammatory agent, without generating free radicals, as seen with benzoyl peroxide. Therefore, it is a compelling option for responding to the presence of Propionibacterium acnes. However, silver has not been approved by the Food and Drug Administration for this use. Even though formal acne studies have not been performed with silver sulfadiazine, it has long been used “off-label” for this purpose. As suggested above, the use of silver sulfadiazine for acne is limited by the risk of sulfa allergy. Cosmetic appearance and ease of use also are limiting factors, as silver sulfadiazine preparations are characterized by a thick, white pasty consistency. Other options for use of silver to treat acne include silver-containing cleansers and textiles.
Atopic dermatitis
A 2006 study in patients with atopic dermatitis demonstrated that silver-coated textiles could significantly diminish Staphylococcus aureus density after 2 days of wearing, with the effect enduring through the end of 7 days of treatment and then 1 week after removal of the textiles.14 Within 2 weeks, objective and subjective symptoms of atopic dermatitis were significantly enhanced in association with the silver-coated textiles, compared with cotton, without measurable adverse effects. A technology called Padycare incorporates silver into micromesh material (82% polyamide, 18% Lycra) used in clothing and bedding.15 As compared with topical formulations applied directly to the skin, textiles confer certain advantages such as preventing scratching and protecting against irritating substances and allergens. Washing of silver-infused textiles is a possible disadvantage, though, as the amount of silver lost from textiles can range from a 100% loss after four washings to less than a 1% loss.16 It also is important to note that there are concerns regarding the potential of silver to leak from textiles into the water supply, and eradicating the beneficial bacteria used to treat the water.
Conclusion
Despite centuries of medical use, silver has not been approved by the FDA for any medical applications. Further study, particularly in terms of safety and efficacy, is necessary. Nevertheless, it is used off-label before and after minimally invasive dermatologic procedures (for example, dermal filling, botulinum toxin injections, chemical peeling) because of its antimicrobial and anti-inflammatory activities as well as soothing qualities for facial skin and the skin barrier. Silver appears to be particularly suitable for use as an acne therapy option due to the low risk of bacterial resistance, lack of irritation, and its preservation of the skin barrier unlike harsher options such as retinoids, antibiotics, and benzoyl peroxide.
References
1. Adv Skin Wound Care. 2006 Nov-Dec;19(9):472-4.
2. Clin Infect Dis. 2009 Nov 15;49(10):1541-9.
3. Nanomedicine. 2008 Sep;4(3):241-51.
4. J Antimicrob Chemother. 2013 Jan;68(1):131-8.
5. Nanocrystalline Silver: Use in wound care, in Current Advances in the Medical Application of Nanotechnology (Manchester, England: Bentham Books, 2012, pp. 25-31).
6. Nanomedicine. 2013 Jan;9(1):39-54.
7. Jpn J Pharmacol. 1965 Jun;15(2):131-4.
8. J Allergy Clin Immunol. 1995 Aug;96(2):251-6.
9. Inflamm Res. 2003 Dec;52(12):487-501.
10. J Nutr Environ Med. 1997;7(4):295-305.
11. Clin Exp Pharmacol Physiol. 2000 Mar;27(3):139-44.
12. Clin Exp Dermatol. 2004 May;29(3):282-7.
13. Br J Dermatol. 2005 Jun;152(6):1235-42.
14. Curr Probl Dermatol. 2006;33:152-64.
15. J Eur Acad Dermatol Venereol. 2006 May;20(5):534-41.
16. Environ Sci Technol. 2008 Jun 1;42(11):4133-9.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis, Neutrogena, Philosophy, Topix, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Silver is a naturally occurring chemical element (number 47 on the periodic table) with the chemical symbol Ag, which is derived from the Latin word argentum (árgyros in Greek), from the Indo-European root “arg-,” meaning “shining,” “white,” or “gray.” It has been used for medical purposes since ancient times, with Hippocrates (circa 460-370 BCE) noting the beneficial healing and disease-altering activity of the element.1
In modern times, silver compounds – in metallic, nanocrystalline, and ionic formulations – have exhibited broad antibacterial activity and attracted interest for topical antiseptic use in wound dressings.2 Nanocrystalline silver dressings were introduced commercially as antimicrobial dressings in 1998.3 Silver is now used in dressings, catheters, cleansers, ophthalmic ointments, and myriad other medical products. In 2010 alone, an estimated 15 metric tons of silver were incorporated into medical products worldwide.4 It also is included in personal care products, textiles, and water purification devices. In topical skin preparations, the noble metal is included as colloidal silver (suspension of silver particles in an aqueous base) or nanosilver (as nanoparticles ranging from 1 nm to 100 nm in at least one dimension).5 Silver has been used to treat burns and wounds, but this discussion will be limited to acne, atopic dermatitis, and the anti-inflammatory response.
Anti-inflammatory uses
For several decades, noble metals including silver have been known to exert anti-inflammatory activity.7-11 In the case of silver, its anti-inflammatory properties appear to be mediated by its influence on the cytokine system. Silver nanoparticles inhibit the activity of interleukin-6 (IL-6), IL-12, IL-1beta, and tumor necrosis factor–alpha (TNF-alpha). This impact on the cytokine system is responsible for the impact of silver in demonstrably alleviating symptoms of rheumatoid arthritis.3
In 2004, Bhol et al. used dinitrochlorobenzene (DNCB) to induce allergic contact dermatitis in a guinea pig model, finding that topical nanocrystalline silver cream dose-dependently decreased erythema and as effectively as topical steroids and immunosuppressants.12 The next year, Bhol and Schechter showed that nanocrystalline silver suppressed allergic contact dermatitis in mice, inhibited TNF-alpha and IL-12 expression, and induced inflammatory cell apoptosis.13
In 2008, Nadworny et al. used a porcine contact dermatitis model to investigate the anti-inflammatory activity of nanocrystalline silver. They found that nanocrystalline silver treatments reduced DNCB-induced erythema and edema, promoted apoptosis in dermal cells, and diminished matrix metalloproteinase (MMP) and proinflammatory cytokine expression.3 The investigators speculated that the lower TNF-alpha observed in the silver-treated animals occurred due to apoptosis of the inflammatory cells.
Acne
Silver acts as a bactericidal and anti-inflammatory agent, without generating free radicals, as seen with benzoyl peroxide. Therefore, it is a compelling option for responding to the presence of Propionibacterium acnes. However, silver has not been approved by the Food and Drug Administration for this use. Even though formal acne studies have not been performed with silver sulfadiazine, it has long been used “off-label” for this purpose. As suggested above, the use of silver sulfadiazine for acne is limited by the risk of sulfa allergy. Cosmetic appearance and ease of use also are limiting factors, as silver sulfadiazine preparations are characterized by a thick, white pasty consistency. Other options for use of silver to treat acne include silver-containing cleansers and textiles.
Atopic dermatitis
A 2006 study in patients with atopic dermatitis demonstrated that silver-coated textiles could significantly diminish Staphylococcus aureus density after 2 days of wearing, with the effect enduring through the end of 7 days of treatment and then 1 week after removal of the textiles.14 Within 2 weeks, objective and subjective symptoms of atopic dermatitis were significantly enhanced in association with the silver-coated textiles, compared with cotton, without measurable adverse effects. A technology called Padycare incorporates silver into micromesh material (82% polyamide, 18% Lycra) used in clothing and bedding.15 As compared with topical formulations applied directly to the skin, textiles confer certain advantages such as preventing scratching and protecting against irritating substances and allergens. Washing of silver-infused textiles is a possible disadvantage, though, as the amount of silver lost from textiles can range from a 100% loss after four washings to less than a 1% loss.16 It also is important to note that there are concerns regarding the potential of silver to leak from textiles into the water supply, and eradicating the beneficial bacteria used to treat the water.
Conclusion
Despite centuries of medical use, silver has not been approved by the FDA for any medical applications. Further study, particularly in terms of safety and efficacy, is necessary. Nevertheless, it is used off-label before and after minimally invasive dermatologic procedures (for example, dermal filling, botulinum toxin injections, chemical peeling) because of its antimicrobial and anti-inflammatory activities as well as soothing qualities for facial skin and the skin barrier. Silver appears to be particularly suitable for use as an acne therapy option due to the low risk of bacterial resistance, lack of irritation, and its preservation of the skin barrier unlike harsher options such as retinoids, antibiotics, and benzoyl peroxide.
References
1. Adv Skin Wound Care. 2006 Nov-Dec;19(9):472-4.
2. Clin Infect Dis. 2009 Nov 15;49(10):1541-9.
3. Nanomedicine. 2008 Sep;4(3):241-51.
4. J Antimicrob Chemother. 2013 Jan;68(1):131-8.
5. Nanocrystalline Silver: Use in wound care, in Current Advances in the Medical Application of Nanotechnology (Manchester, England: Bentham Books, 2012, pp. 25-31).
6. Nanomedicine. 2013 Jan;9(1):39-54.
7. Jpn J Pharmacol. 1965 Jun;15(2):131-4.
8. J Allergy Clin Immunol. 1995 Aug;96(2):251-6.
9. Inflamm Res. 2003 Dec;52(12):487-501.
10. J Nutr Environ Med. 1997;7(4):295-305.
11. Clin Exp Pharmacol Physiol. 2000 Mar;27(3):139-44.
12. Clin Exp Dermatol. 2004 May;29(3):282-7.
13. Br J Dermatol. 2005 Jun;152(6):1235-42.
14. Curr Probl Dermatol. 2006;33:152-64.
15. J Eur Acad Dermatol Venereol. 2006 May;20(5):534-41.
16. Environ Sci Technol. 2008 Jun 1;42(11):4133-9.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis, Neutrogena, Philosophy, Topix, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.