User login
Filler agents may improve QOL in HIV facial lipoatrophy
Patients with HIV facial lipoatrophy (FLA) had improved quality of life scores after treatment with hyaluronic acid (HA) filler, report Derek Ho of the Sacramento VA Medical Center, and coauthors.
A prospective, open-label, phase I and II study assessed 20 patients with an HIV FLA Carruthers Lipoatrophy Severity Scale (CLSS) grade of 2 or higher, who had not received treatment for HIV FLA in the past year. Volumizing of the cheeks and temples was performed using a 20 mg/mL HA filler, with an optional touch-up at 2 weeks follow-up. Patients were given a vision exam before treatment, immediately after treatment, and 15 minutes after treatment.
Quality of life was assessed before treatment and at 12 months follow-up using the Dermatology Life Quality Index (DLQI). Satisfaction was evaluated using a subject satisfaction questionnaire at 12 months follow-up, Mr. Ho and his colleagues reported.
DLQI scores were 1.6±3.0 (range = 0-11) and 0.5±1.2 (range = 0-5) at baseline and follow-up, respectively, the authors said. Additionally, 100% of patients reported high satisfaction as indicated by answers on the subject satisfaction questionnaire, they added.
The investigators warned, however, that they would not recommend the DLQI in the future as a measure of quality of life, as this scale focuses on disability, and fails to account for mental health issues.
Still, the findings highlight “the importance of educating and offering aesthetic and corrective treatment to all patients with HIV FLA,” the authors concluded.
Read the full article in the Journal of Drugs in Dermatology.
Patients with HIV facial lipoatrophy (FLA) had improved quality of life scores after treatment with hyaluronic acid (HA) filler, report Derek Ho of the Sacramento VA Medical Center, and coauthors.
A prospective, open-label, phase I and II study assessed 20 patients with an HIV FLA Carruthers Lipoatrophy Severity Scale (CLSS) grade of 2 or higher, who had not received treatment for HIV FLA in the past year. Volumizing of the cheeks and temples was performed using a 20 mg/mL HA filler, with an optional touch-up at 2 weeks follow-up. Patients were given a vision exam before treatment, immediately after treatment, and 15 minutes after treatment.
Quality of life was assessed before treatment and at 12 months follow-up using the Dermatology Life Quality Index (DLQI). Satisfaction was evaluated using a subject satisfaction questionnaire at 12 months follow-up, Mr. Ho and his colleagues reported.
DLQI scores were 1.6±3.0 (range = 0-11) and 0.5±1.2 (range = 0-5) at baseline and follow-up, respectively, the authors said. Additionally, 100% of patients reported high satisfaction as indicated by answers on the subject satisfaction questionnaire, they added.
The investigators warned, however, that they would not recommend the DLQI in the future as a measure of quality of life, as this scale focuses on disability, and fails to account for mental health issues.
Still, the findings highlight “the importance of educating and offering aesthetic and corrective treatment to all patients with HIV FLA,” the authors concluded.
Read the full article in the Journal of Drugs in Dermatology.
Patients with HIV facial lipoatrophy (FLA) had improved quality of life scores after treatment with hyaluronic acid (HA) filler, report Derek Ho of the Sacramento VA Medical Center, and coauthors.
A prospective, open-label, phase I and II study assessed 20 patients with an HIV FLA Carruthers Lipoatrophy Severity Scale (CLSS) grade of 2 or higher, who had not received treatment for HIV FLA in the past year. Volumizing of the cheeks and temples was performed using a 20 mg/mL HA filler, with an optional touch-up at 2 weeks follow-up. Patients were given a vision exam before treatment, immediately after treatment, and 15 minutes after treatment.
Quality of life was assessed before treatment and at 12 months follow-up using the Dermatology Life Quality Index (DLQI). Satisfaction was evaluated using a subject satisfaction questionnaire at 12 months follow-up, Mr. Ho and his colleagues reported.
DLQI scores were 1.6±3.0 (range = 0-11) and 0.5±1.2 (range = 0-5) at baseline and follow-up, respectively, the authors said. Additionally, 100% of patients reported high satisfaction as indicated by answers on the subject satisfaction questionnaire, they added.
The investigators warned, however, that they would not recommend the DLQI in the future as a measure of quality of life, as this scale focuses on disability, and fails to account for mental health issues.
Still, the findings highlight “the importance of educating and offering aesthetic and corrective treatment to all patients with HIV FLA,” the authors concluded.
Read the full article in the Journal of Drugs in Dermatology.
Mucous Membrane Pemphigoid Involving the Trachea and Bronchi: An Extremely Rare and Life-Threatening Presentation
To the Editor:
Mucous membrane pemphigoid (MMP) is an autoimmune blistering disorder that causes subepithelial damage and scarring of mucosal surfaces with or without skin involvement.1 The clinical presentation is highly variable. The oropharynx is the most common site of initial presentation, followed by ocular, nasopharyngeal, anogenital, skin, laryngeal, and esophageal involvement.2 Patients often present to a variety of specialists depending on initial symptoms, and due to the diverse clinical manifestations, MMP often is misdiagnosed. Our patient presented an even greater challenge because the disease progressed to tracheal and bronchial involvement.
A 37-year-old man presented to his primary care physician with a chief concern of a sore throat and oral ulcers. The patient was treated with a course of antibiotics followed by a nystatin oral solution. He continued to develop ulcerative lesions on the soft palate, posterior pharynx, and nasal mucosae. He sought treatment from 2 otolaryngologists (ENTs) and a gastroenterologist, and continued to be treated with multiple oral antibiotics, fluconazole, and topical nystatin. Despite treatment, the patient developed pansinusitis and laryngitis and presented to the ENT department at our institution with severe hoarseness and dyspnea on exertion. Examination by the ENT department revealed ulcerative lesions of the nares with stenosis and ulcers along the soft palate. Videolaryngostroboscopy showed remarkable supraglottic edema with thick endolaryngeal mucus. The patient worked as a funeral director and had notable formaldehyde exposure. He also hunted wild game and performed taxidermy regularly.
The patient was admitted and treated with intravenous dexamethasone for a compromised airway. Subsequently, he was taken to the operating room and had biopsies performed of the posterior pharynx. Given his exposure history, the infectious disease department was consulted and he was evaluated for multiple viral, bacterial, and fungal suspects including leishmania and tularemia. Age-appropriate screening, physical examination, and review of systems were negative for an underlying neoplasm. Histopathologic examination revealed a subepithelial vesicular mucositis with a mixed infiltrate of lymphocytes and histiocytes. Direct immunofluorescence microscopy demonstrated strong linear fluorescence along the epithelial-subepithelial junction with IgG and C3. Based on these findings, the diagnosis of MMP was made.
Further testing for bullous pemphigoid antigen 1 (BP230) and bullous pemphigoid antigen 2 (BP180) were negative. On one occasion the patient tested positive for anti-BP230 IgG, but it was at a level judged to be insignificant (7.5 [reference range, <9]). The patient also was negative for autoantibodies against desmoglein 1 and 3. Indirect immunofluorescence using rat bladder epithelium was not performed.
The patient was started on methotrexate and oral prednisone by the rheumatology department, but after 1 week, he presented in respiratory distress and was taken for an emergency tracheostomy. The patient eventually was referred to the dermatology department where methotrexate was discontinued and the patient was started on titrating doses of prednisone and mycophenolate mofetil. Eight weeks later, the patient became completely aphonic and was taken by ENT for dilation of the supraglottic, glottic, and subglottic stenosis with mucosal triamcinolone injections. Doxycycline 100 mg twice daily and nicotinamide 500 mg twice daily was initiated in addition to mycophenolate mofetil 3 g and prednisone 80 mg, but again the patient developed near-complete tracheal stenosis just proximal to the tracheostomy entry site. At 16 weeks, balloon dilation was repeated with dexamethasone injections and topical mitomycin C. Subsequently, the patient regained some use of his voice. Although the next several laryngoscopes showed improvement in the patient’s epiglottis and glottis, the trachea continued to require debridement and dilation.
Despite maximal medical therapy and surgical interventions, the patient had little improvement in his voice and large clots of blood obstructed his tracheostomy daily. He was unable to sleep in his preferred position on the stomach (prone) due to dyspnea but had less distress sleeping on his back (supine). The patient was referred to the pulmonology department for an endotracheobronchoscopy to further evaluate the airway. It was discovered that the mucosa of the trachea from the level of the tracheostomy to the carina was friable with active erosions and thick bloody secretions (Figure 1). Lesions extended as far as the scope was able to visualize to the left upper lobe takeoff and the right mainstem bronchus (Figure 2). Biopsies of the carinal mucosa showed 3+/3+ linear fluorescence with IgG along the dermoepidermal junction. Salt-split studies were performed, but because the specimen was fragmented, it was not possible to assess if the fluorescence was present at the floor or at the roof of the split.
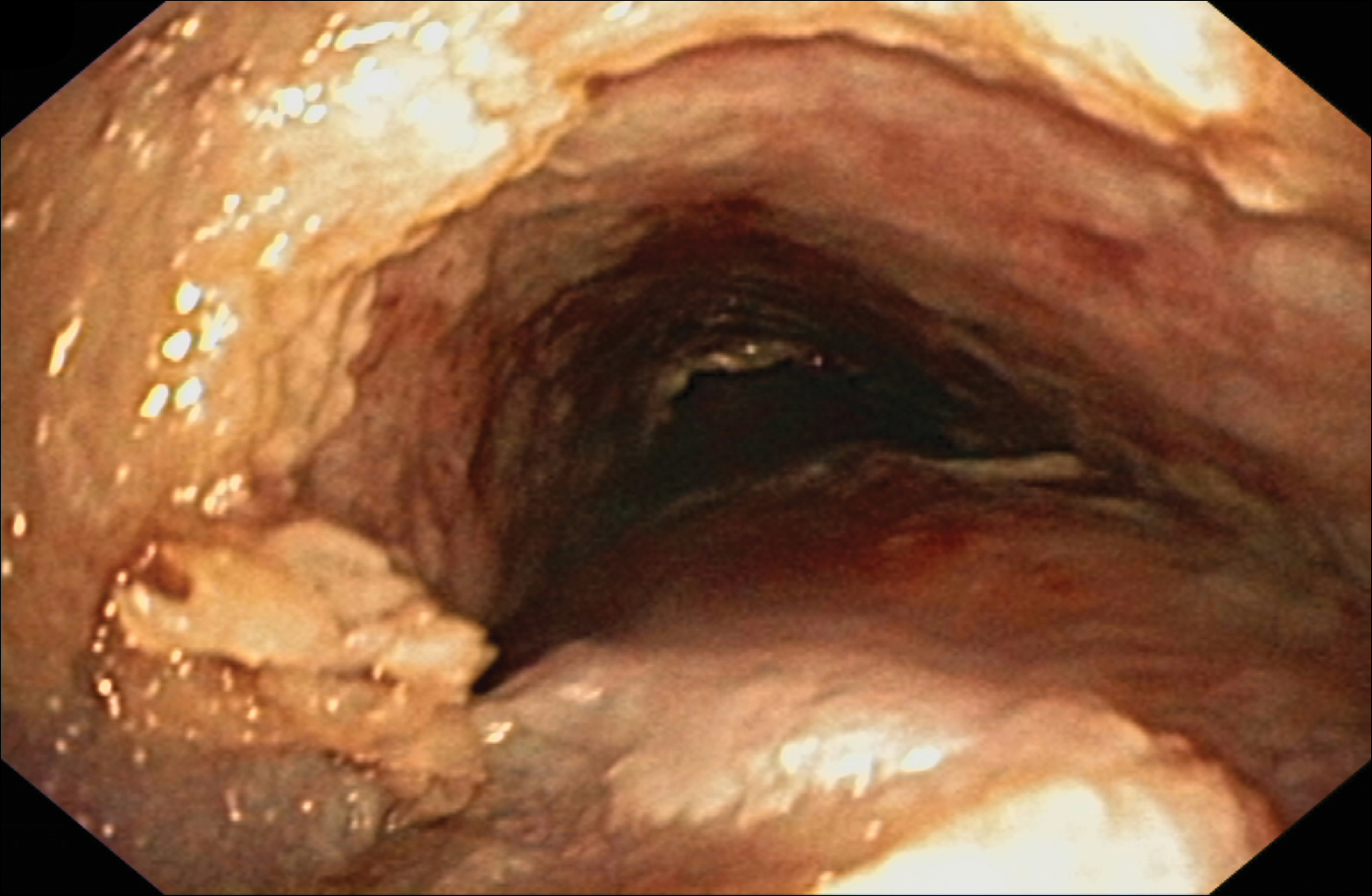

Given the severity of disease and failure to respond to other aggressive immunosuppressive therapies as well as having been with a tracheostomy for 22 months, the patient was started on 2 doses of intravenous rituximab 1 g 2 weeks apart along with trimethoprim-sulfamethoxazole (3 times weekly) for pneumocystis pneumonia prophylaxis. No complications were observed during infusions. After 2 rituximab infusions, he was weaned off of prednisone and a repeat bronchoscopy showed no airway ulcers beyond the distal trachea or endobronchial obstruction. However, the subglottic space and area above the tracheostomy showed remarkable stenosis with a cobblestone pattern and granulation tissue with continued narrowing of the subglottic area. The ENT performed further dilation and after 34 months, the tracheostomy was removed and a T-tube was placed. The patient required cleaning out of the T-tube approximately every 3 months, and after 2 years the original T-tube was replaced with a new one. At the time of this report, the ENT recommended removing the T-tube, but the patient was reluctant to do so; therefore, a second T-tube replacement is planned. He continues to do well without relapse and has been off all medical therapy for nearly 4 years.
Mucous membrane pemphigoid is an acquired autoimmune subepithelial blistering disease that predominantly affects mucous membranes with or without skin involvement. This condition has been referred to as cicatricial pemphigoid, oral pemphigoid, and ocular cicatricial pemphigoid, among other names. It is characterized by linear deposition of IgG, IgA, or C3 along the epithelial basement membrane zone. According to the international consensus on MMP, the target antigens identified in the epithelial basement membrane zone include bullous pemphigoid antigen 1 (BP230), bullous pemphigoid antigen 2 (BP180), laminin 5 (α3, β3, γ2 chains), laminin 6 (α3 chain), type VII collagen, and integrin β4 subunit.3 Not all patients with MMP will have circulating autoantibodies to the above components, and although our patient did have detectable anti-BP230 IgG, it was not considered clinically significant. Furthermore, the type of autoantibody does not impact decisions regarding therapy selection.3
Although rare, MMP is well-known to dermatologists and ophthalmologists who manage a large majority of MMP patients depending on which mucosa is involved. Mucous membrane pemphigoid is extremely rare in the lower respiratory tract, and when these lesions are discovered, it often is in the face of life-threatening respiratory distress. Mucous membrane pemphigoid is a challenging disease to treat, even more so when the primary specialty physician is unable to visualize the affected areas. Our patient’s disease was limited primarily to the pharynx, larynx, trachea, and bronchi with few oral lesions. According to a PubMed search of articles indexed for MEDLINE using the terms mucous membrane pemphigus, cicatricial pemphigoid, trachea, bronchus, and fatal, 8 reports (7 case reports and 1 prospective study) of MMP involving the lower respiratory tract have been published.4-11 Of the case reports, each patient also presented with involvement of the eyes or skin.4,5,7-11 Four of these cases were fatal secondary to cardiopulmonary arrest.5,7,9,10 In the prospective study, 110 consecutive patients with clinical, histologic, and immunologic criteria of MMP were examined with a flexible nasopharyngolaryngoscope.6 Thirty-eight patients had nose or throat symptoms but only 10 had laryngeal involvement and 5 had acute dyspnea. The nasal valves, choanae, pharynx, and/or larynx were severely scarred in 7 patients, which was fatal in 3.6
Medical treatment should be based on the following factors of the patient’s disease: site, severity, and rapidity of progression.3 High-risk patients can be defined as those who have lesions at any of the following sites: ocular, genital, nasopharyngeal, esophageal, and laryngeal mucosae. As our patient had involvement at several high-risk sites, in particular sites only visualized by various scoping procedures, a team of physicians including dermatologists, ENT physicians, pulmonologists, and oncologists was necessary to facilitate his care. Scarring is the hallmark of MMP and prevention of scarring is the most important aspect of treatment of MMP. Surgical repair of the previously involved mucosa is difficult, as the tissue is prone to re-scarring and difficult to heal. Over the last several years, there has been increasing evidence for the use of rituximab in autoimmune bullous skin diseases including pemphigus vulgaris, epidermolysis bullosa acquisita, and MMP.12-14 After 2 infusions of rituximab, our patient had clearance of his disease and currently is doing well with a T-tube.
Acknowledgments
We thank Kim Yancey, MD (Dallas, Texas), for providing access to the patient’s diagnostic laboratory immunology and reviewing biopsy specimens; Luis Angel, MD (San Antonio, Texas), for providing bronchoscopy photographs; and C. Blake Simpson, MD (San Antonio, Texas), for co-managing this challenging case.
- James WD, Berger TG, Elston D. Chronic blistering diseases. In: James WD, Berger TG, Elston D. Andrews’ Diseases of the Skin: Clinical Dermatology. 11th ed. Philadelphia, PA: Sanders Elsevier; 2010:448-467.
- Neff AG, Turner M, Mutasim DF. Treatment strategies in mucous membrane pemphigoid. Ther Clin Risk Manag. 2008;4:617-626.
- Chan LS, Ahmed AR, Anhalt GJ, et al. The first international consensus on mucous membrane pemphigoid: definition, diagnostic criteria, pathogenic factors, medical treatment, and prognostic indicators. Arch Dermatol. 2002;138:370-379.
- Kato K, Moriyama Y, Saito H, et al. A case of mucous membrane pemphigoid involving the trachea and bronchus with autoantibodies to β3 subunit of laminin-332. Acta Derm Venereol. 2014;94:237-238.
- Gamm DM, Harris A, Mehran RJ, et al. Mucous membrane pemphigoid with fatal bronchial involvement in a seventeen-year-old girl. Cornea. 2006;25:474-478.
- Alexandre M, Brette MD, Pascal F, et al. A prospective study of upper aerodigestive tract manifestations of mucous membrane pemphigoid. Medicine (Baltimore). 2006;85:239-252.
- de Carvalho CR, Amato MB, Da Silva LM, et al. Obstructive respiratory failure in cicatricial pemphigoid. Thorax. 1989;44:601-602.
- Müller LC, Salzer GM. Stenosis of left mainstem bronchus in a case of cicatricial pemphigoid. Eur J Cardiothorac Surg. 1988;2:284-286.
- Camisa C, Allen CM. Death from CP in a young woman with oral, laryngeal, and bronchial involvement. Cutis. 1987;40:426-429.
- Derbes VJ, Pitot HC, Chernosky ME. Fatal cicatricial mucous membrane pemphigoid of the trachea. Dermatol Trop Ecol Geogr. 1962;1:114-117.
- Wieme N, Lambert J, Moerman M, et al. Epidermolysis bullosa acquisita with combined features of bullous pemphigoid and cicatricial pemphigoid. Dermatology. 1999;198:310-313.
- Taylor J, McMillan R, Shephard M, et al. World Workshop on Oral Medicine VI: a systematic review of the treatment of mucous membrane pemphigoid [published online March 11, 2015]. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;120:161.e20-171.e20.
- Sobolewska B, Deuter C, Zierhut M. Current medical treatment of ocular mucous membrane pemphigoid [published online July 9, 2013]. Ocul Surf. 2013;11:259-266.
- Maley A, Warren M, Haberman I, et al. Rituximab combined with conventional therapy versus conventional therapy alone for the treatment of mucous membrane pemphigoid (MMP) [published online February 28, 2016]. J Am Acad Dermatol. 2016;74:835-840.
To the Editor:
Mucous membrane pemphigoid (MMP) is an autoimmune blistering disorder that causes subepithelial damage and scarring of mucosal surfaces with or without skin involvement.1 The clinical presentation is highly variable. The oropharynx is the most common site of initial presentation, followed by ocular, nasopharyngeal, anogenital, skin, laryngeal, and esophageal involvement.2 Patients often present to a variety of specialists depending on initial symptoms, and due to the diverse clinical manifestations, MMP often is misdiagnosed. Our patient presented an even greater challenge because the disease progressed to tracheal and bronchial involvement.
A 37-year-old man presented to his primary care physician with a chief concern of a sore throat and oral ulcers. The patient was treated with a course of antibiotics followed by a nystatin oral solution. He continued to develop ulcerative lesions on the soft palate, posterior pharynx, and nasal mucosae. He sought treatment from 2 otolaryngologists (ENTs) and a gastroenterologist, and continued to be treated with multiple oral antibiotics, fluconazole, and topical nystatin. Despite treatment, the patient developed pansinusitis and laryngitis and presented to the ENT department at our institution with severe hoarseness and dyspnea on exertion. Examination by the ENT department revealed ulcerative lesions of the nares with stenosis and ulcers along the soft palate. Videolaryngostroboscopy showed remarkable supraglottic edema with thick endolaryngeal mucus. The patient worked as a funeral director and had notable formaldehyde exposure. He also hunted wild game and performed taxidermy regularly.
The patient was admitted and treated with intravenous dexamethasone for a compromised airway. Subsequently, he was taken to the operating room and had biopsies performed of the posterior pharynx. Given his exposure history, the infectious disease department was consulted and he was evaluated for multiple viral, bacterial, and fungal suspects including leishmania and tularemia. Age-appropriate screening, physical examination, and review of systems were negative for an underlying neoplasm. Histopathologic examination revealed a subepithelial vesicular mucositis with a mixed infiltrate of lymphocytes and histiocytes. Direct immunofluorescence microscopy demonstrated strong linear fluorescence along the epithelial-subepithelial junction with IgG and C3. Based on these findings, the diagnosis of MMP was made.
Further testing for bullous pemphigoid antigen 1 (BP230) and bullous pemphigoid antigen 2 (BP180) were negative. On one occasion the patient tested positive for anti-BP230 IgG, but it was at a level judged to be insignificant (7.5 [reference range, <9]). The patient also was negative for autoantibodies against desmoglein 1 and 3. Indirect immunofluorescence using rat bladder epithelium was not performed.
The patient was started on methotrexate and oral prednisone by the rheumatology department, but after 1 week, he presented in respiratory distress and was taken for an emergency tracheostomy. The patient eventually was referred to the dermatology department where methotrexate was discontinued and the patient was started on titrating doses of prednisone and mycophenolate mofetil. Eight weeks later, the patient became completely aphonic and was taken by ENT for dilation of the supraglottic, glottic, and subglottic stenosis with mucosal triamcinolone injections. Doxycycline 100 mg twice daily and nicotinamide 500 mg twice daily was initiated in addition to mycophenolate mofetil 3 g and prednisone 80 mg, but again the patient developed near-complete tracheal stenosis just proximal to the tracheostomy entry site. At 16 weeks, balloon dilation was repeated with dexamethasone injections and topical mitomycin C. Subsequently, the patient regained some use of his voice. Although the next several laryngoscopes showed improvement in the patient’s epiglottis and glottis, the trachea continued to require debridement and dilation.
Despite maximal medical therapy and surgical interventions, the patient had little improvement in his voice and large clots of blood obstructed his tracheostomy daily. He was unable to sleep in his preferred position on the stomach (prone) due to dyspnea but had less distress sleeping on his back (supine). The patient was referred to the pulmonology department for an endotracheobronchoscopy to further evaluate the airway. It was discovered that the mucosa of the trachea from the level of the tracheostomy to the carina was friable with active erosions and thick bloody secretions (Figure 1). Lesions extended as far as the scope was able to visualize to the left upper lobe takeoff and the right mainstem bronchus (Figure 2). Biopsies of the carinal mucosa showed 3+/3+ linear fluorescence with IgG along the dermoepidermal junction. Salt-split studies were performed, but because the specimen was fragmented, it was not possible to assess if the fluorescence was present at the floor or at the roof of the split.


Given the severity of disease and failure to respond to other aggressive immunosuppressive therapies as well as having been with a tracheostomy for 22 months, the patient was started on 2 doses of intravenous rituximab 1 g 2 weeks apart along with trimethoprim-sulfamethoxazole (3 times weekly) for pneumocystis pneumonia prophylaxis. No complications were observed during infusions. After 2 rituximab infusions, he was weaned off of prednisone and a repeat bronchoscopy showed no airway ulcers beyond the distal trachea or endobronchial obstruction. However, the subglottic space and area above the tracheostomy showed remarkable stenosis with a cobblestone pattern and granulation tissue with continued narrowing of the subglottic area. The ENT performed further dilation and after 34 months, the tracheostomy was removed and a T-tube was placed. The patient required cleaning out of the T-tube approximately every 3 months, and after 2 years the original T-tube was replaced with a new one. At the time of this report, the ENT recommended removing the T-tube, but the patient was reluctant to do so; therefore, a second T-tube replacement is planned. He continues to do well without relapse and has been off all medical therapy for nearly 4 years.
Mucous membrane pemphigoid is an acquired autoimmune subepithelial blistering disease that predominantly affects mucous membranes with or without skin involvement. This condition has been referred to as cicatricial pemphigoid, oral pemphigoid, and ocular cicatricial pemphigoid, among other names. It is characterized by linear deposition of IgG, IgA, or C3 along the epithelial basement membrane zone. According to the international consensus on MMP, the target antigens identified in the epithelial basement membrane zone include bullous pemphigoid antigen 1 (BP230), bullous pemphigoid antigen 2 (BP180), laminin 5 (α3, β3, γ2 chains), laminin 6 (α3 chain), type VII collagen, and integrin β4 subunit.3 Not all patients with MMP will have circulating autoantibodies to the above components, and although our patient did have detectable anti-BP230 IgG, it was not considered clinically significant. Furthermore, the type of autoantibody does not impact decisions regarding therapy selection.3
Although rare, MMP is well-known to dermatologists and ophthalmologists who manage a large majority of MMP patients depending on which mucosa is involved. Mucous membrane pemphigoid is extremely rare in the lower respiratory tract, and when these lesions are discovered, it often is in the face of life-threatening respiratory distress. Mucous membrane pemphigoid is a challenging disease to treat, even more so when the primary specialty physician is unable to visualize the affected areas. Our patient’s disease was limited primarily to the pharynx, larynx, trachea, and bronchi with few oral lesions. According to a PubMed search of articles indexed for MEDLINE using the terms mucous membrane pemphigus, cicatricial pemphigoid, trachea, bronchus, and fatal, 8 reports (7 case reports and 1 prospective study) of MMP involving the lower respiratory tract have been published.4-11 Of the case reports, each patient also presented with involvement of the eyes or skin.4,5,7-11 Four of these cases were fatal secondary to cardiopulmonary arrest.5,7,9,10 In the prospective study, 110 consecutive patients with clinical, histologic, and immunologic criteria of MMP were examined with a flexible nasopharyngolaryngoscope.6 Thirty-eight patients had nose or throat symptoms but only 10 had laryngeal involvement and 5 had acute dyspnea. The nasal valves, choanae, pharynx, and/or larynx were severely scarred in 7 patients, which was fatal in 3.6
Medical treatment should be based on the following factors of the patient’s disease: site, severity, and rapidity of progression.3 High-risk patients can be defined as those who have lesions at any of the following sites: ocular, genital, nasopharyngeal, esophageal, and laryngeal mucosae. As our patient had involvement at several high-risk sites, in particular sites only visualized by various scoping procedures, a team of physicians including dermatologists, ENT physicians, pulmonologists, and oncologists was necessary to facilitate his care. Scarring is the hallmark of MMP and prevention of scarring is the most important aspect of treatment of MMP. Surgical repair of the previously involved mucosa is difficult, as the tissue is prone to re-scarring and difficult to heal. Over the last several years, there has been increasing evidence for the use of rituximab in autoimmune bullous skin diseases including pemphigus vulgaris, epidermolysis bullosa acquisita, and MMP.12-14 After 2 infusions of rituximab, our patient had clearance of his disease and currently is doing well with a T-tube.
Acknowledgments
We thank Kim Yancey, MD (Dallas, Texas), for providing access to the patient’s diagnostic laboratory immunology and reviewing biopsy specimens; Luis Angel, MD (San Antonio, Texas), for providing bronchoscopy photographs; and C. Blake Simpson, MD (San Antonio, Texas), for co-managing this challenging case.
To the Editor:
Mucous membrane pemphigoid (MMP) is an autoimmune blistering disorder that causes subepithelial damage and scarring of mucosal surfaces with or without skin involvement.1 The clinical presentation is highly variable. The oropharynx is the most common site of initial presentation, followed by ocular, nasopharyngeal, anogenital, skin, laryngeal, and esophageal involvement.2 Patients often present to a variety of specialists depending on initial symptoms, and due to the diverse clinical manifestations, MMP often is misdiagnosed. Our patient presented an even greater challenge because the disease progressed to tracheal and bronchial involvement.
A 37-year-old man presented to his primary care physician with a chief concern of a sore throat and oral ulcers. The patient was treated with a course of antibiotics followed by a nystatin oral solution. He continued to develop ulcerative lesions on the soft palate, posterior pharynx, and nasal mucosae. He sought treatment from 2 otolaryngologists (ENTs) and a gastroenterologist, and continued to be treated with multiple oral antibiotics, fluconazole, and topical nystatin. Despite treatment, the patient developed pansinusitis and laryngitis and presented to the ENT department at our institution with severe hoarseness and dyspnea on exertion. Examination by the ENT department revealed ulcerative lesions of the nares with stenosis and ulcers along the soft palate. Videolaryngostroboscopy showed remarkable supraglottic edema with thick endolaryngeal mucus. The patient worked as a funeral director and had notable formaldehyde exposure. He also hunted wild game and performed taxidermy regularly.
The patient was admitted and treated with intravenous dexamethasone for a compromised airway. Subsequently, he was taken to the operating room and had biopsies performed of the posterior pharynx. Given his exposure history, the infectious disease department was consulted and he was evaluated for multiple viral, bacterial, and fungal suspects including leishmania and tularemia. Age-appropriate screening, physical examination, and review of systems were negative for an underlying neoplasm. Histopathologic examination revealed a subepithelial vesicular mucositis with a mixed infiltrate of lymphocytes and histiocytes. Direct immunofluorescence microscopy demonstrated strong linear fluorescence along the epithelial-subepithelial junction with IgG and C3. Based on these findings, the diagnosis of MMP was made.
Further testing for bullous pemphigoid antigen 1 (BP230) and bullous pemphigoid antigen 2 (BP180) were negative. On one occasion the patient tested positive for anti-BP230 IgG, but it was at a level judged to be insignificant (7.5 [reference range, <9]). The patient also was negative for autoantibodies against desmoglein 1 and 3. Indirect immunofluorescence using rat bladder epithelium was not performed.
The patient was started on methotrexate and oral prednisone by the rheumatology department, but after 1 week, he presented in respiratory distress and was taken for an emergency tracheostomy. The patient eventually was referred to the dermatology department where methotrexate was discontinued and the patient was started on titrating doses of prednisone and mycophenolate mofetil. Eight weeks later, the patient became completely aphonic and was taken by ENT for dilation of the supraglottic, glottic, and subglottic stenosis with mucosal triamcinolone injections. Doxycycline 100 mg twice daily and nicotinamide 500 mg twice daily was initiated in addition to mycophenolate mofetil 3 g and prednisone 80 mg, but again the patient developed near-complete tracheal stenosis just proximal to the tracheostomy entry site. At 16 weeks, balloon dilation was repeated with dexamethasone injections and topical mitomycin C. Subsequently, the patient regained some use of his voice. Although the next several laryngoscopes showed improvement in the patient’s epiglottis and glottis, the trachea continued to require debridement and dilation.
Despite maximal medical therapy and surgical interventions, the patient had little improvement in his voice and large clots of blood obstructed his tracheostomy daily. He was unable to sleep in his preferred position on the stomach (prone) due to dyspnea but had less distress sleeping on his back (supine). The patient was referred to the pulmonology department for an endotracheobronchoscopy to further evaluate the airway. It was discovered that the mucosa of the trachea from the level of the tracheostomy to the carina was friable with active erosions and thick bloody secretions (Figure 1). Lesions extended as far as the scope was able to visualize to the left upper lobe takeoff and the right mainstem bronchus (Figure 2). Biopsies of the carinal mucosa showed 3+/3+ linear fluorescence with IgG along the dermoepidermal junction. Salt-split studies were performed, but because the specimen was fragmented, it was not possible to assess if the fluorescence was present at the floor or at the roof of the split.


Given the severity of disease and failure to respond to other aggressive immunosuppressive therapies as well as having been with a tracheostomy for 22 months, the patient was started on 2 doses of intravenous rituximab 1 g 2 weeks apart along with trimethoprim-sulfamethoxazole (3 times weekly) for pneumocystis pneumonia prophylaxis. No complications were observed during infusions. After 2 rituximab infusions, he was weaned off of prednisone and a repeat bronchoscopy showed no airway ulcers beyond the distal trachea or endobronchial obstruction. However, the subglottic space and area above the tracheostomy showed remarkable stenosis with a cobblestone pattern and granulation tissue with continued narrowing of the subglottic area. The ENT performed further dilation and after 34 months, the tracheostomy was removed and a T-tube was placed. The patient required cleaning out of the T-tube approximately every 3 months, and after 2 years the original T-tube was replaced with a new one. At the time of this report, the ENT recommended removing the T-tube, but the patient was reluctant to do so; therefore, a second T-tube replacement is planned. He continues to do well without relapse and has been off all medical therapy for nearly 4 years.
Mucous membrane pemphigoid is an acquired autoimmune subepithelial blistering disease that predominantly affects mucous membranes with or without skin involvement. This condition has been referred to as cicatricial pemphigoid, oral pemphigoid, and ocular cicatricial pemphigoid, among other names. It is characterized by linear deposition of IgG, IgA, or C3 along the epithelial basement membrane zone. According to the international consensus on MMP, the target antigens identified in the epithelial basement membrane zone include bullous pemphigoid antigen 1 (BP230), bullous pemphigoid antigen 2 (BP180), laminin 5 (α3, β3, γ2 chains), laminin 6 (α3 chain), type VII collagen, and integrin β4 subunit.3 Not all patients with MMP will have circulating autoantibodies to the above components, and although our patient did have detectable anti-BP230 IgG, it was not considered clinically significant. Furthermore, the type of autoantibody does not impact decisions regarding therapy selection.3
Although rare, MMP is well-known to dermatologists and ophthalmologists who manage a large majority of MMP patients depending on which mucosa is involved. Mucous membrane pemphigoid is extremely rare in the lower respiratory tract, and when these lesions are discovered, it often is in the face of life-threatening respiratory distress. Mucous membrane pemphigoid is a challenging disease to treat, even more so when the primary specialty physician is unable to visualize the affected areas. Our patient’s disease was limited primarily to the pharynx, larynx, trachea, and bronchi with few oral lesions. According to a PubMed search of articles indexed for MEDLINE using the terms mucous membrane pemphigus, cicatricial pemphigoid, trachea, bronchus, and fatal, 8 reports (7 case reports and 1 prospective study) of MMP involving the lower respiratory tract have been published.4-11 Of the case reports, each patient also presented with involvement of the eyes or skin.4,5,7-11 Four of these cases were fatal secondary to cardiopulmonary arrest.5,7,9,10 In the prospective study, 110 consecutive patients with clinical, histologic, and immunologic criteria of MMP were examined with a flexible nasopharyngolaryngoscope.6 Thirty-eight patients had nose or throat symptoms but only 10 had laryngeal involvement and 5 had acute dyspnea. The nasal valves, choanae, pharynx, and/or larynx were severely scarred in 7 patients, which was fatal in 3.6
Medical treatment should be based on the following factors of the patient’s disease: site, severity, and rapidity of progression.3 High-risk patients can be defined as those who have lesions at any of the following sites: ocular, genital, nasopharyngeal, esophageal, and laryngeal mucosae. As our patient had involvement at several high-risk sites, in particular sites only visualized by various scoping procedures, a team of physicians including dermatologists, ENT physicians, pulmonologists, and oncologists was necessary to facilitate his care. Scarring is the hallmark of MMP and prevention of scarring is the most important aspect of treatment of MMP. Surgical repair of the previously involved mucosa is difficult, as the tissue is prone to re-scarring and difficult to heal. Over the last several years, there has been increasing evidence for the use of rituximab in autoimmune bullous skin diseases including pemphigus vulgaris, epidermolysis bullosa acquisita, and MMP.12-14 After 2 infusions of rituximab, our patient had clearance of his disease and currently is doing well with a T-tube.
Acknowledgments
We thank Kim Yancey, MD (Dallas, Texas), for providing access to the patient’s diagnostic laboratory immunology and reviewing biopsy specimens; Luis Angel, MD (San Antonio, Texas), for providing bronchoscopy photographs; and C. Blake Simpson, MD (San Antonio, Texas), for co-managing this challenging case.
- James WD, Berger TG, Elston D. Chronic blistering diseases. In: James WD, Berger TG, Elston D. Andrews’ Diseases of the Skin: Clinical Dermatology. 11th ed. Philadelphia, PA: Sanders Elsevier; 2010:448-467.
- Neff AG, Turner M, Mutasim DF. Treatment strategies in mucous membrane pemphigoid. Ther Clin Risk Manag. 2008;4:617-626.
- Chan LS, Ahmed AR, Anhalt GJ, et al. The first international consensus on mucous membrane pemphigoid: definition, diagnostic criteria, pathogenic factors, medical treatment, and prognostic indicators. Arch Dermatol. 2002;138:370-379.
- Kato K, Moriyama Y, Saito H, et al. A case of mucous membrane pemphigoid involving the trachea and bronchus with autoantibodies to β3 subunit of laminin-332. Acta Derm Venereol. 2014;94:237-238.
- Gamm DM, Harris A, Mehran RJ, et al. Mucous membrane pemphigoid with fatal bronchial involvement in a seventeen-year-old girl. Cornea. 2006;25:474-478.
- Alexandre M, Brette MD, Pascal F, et al. A prospective study of upper aerodigestive tract manifestations of mucous membrane pemphigoid. Medicine (Baltimore). 2006;85:239-252.
- de Carvalho CR, Amato MB, Da Silva LM, et al. Obstructive respiratory failure in cicatricial pemphigoid. Thorax. 1989;44:601-602.
- Müller LC, Salzer GM. Stenosis of left mainstem bronchus in a case of cicatricial pemphigoid. Eur J Cardiothorac Surg. 1988;2:284-286.
- Camisa C, Allen CM. Death from CP in a young woman with oral, laryngeal, and bronchial involvement. Cutis. 1987;40:426-429.
- Derbes VJ, Pitot HC, Chernosky ME. Fatal cicatricial mucous membrane pemphigoid of the trachea. Dermatol Trop Ecol Geogr. 1962;1:114-117.
- Wieme N, Lambert J, Moerman M, et al. Epidermolysis bullosa acquisita with combined features of bullous pemphigoid and cicatricial pemphigoid. Dermatology. 1999;198:310-313.
- Taylor J, McMillan R, Shephard M, et al. World Workshop on Oral Medicine VI: a systematic review of the treatment of mucous membrane pemphigoid [published online March 11, 2015]. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;120:161.e20-171.e20.
- Sobolewska B, Deuter C, Zierhut M. Current medical treatment of ocular mucous membrane pemphigoid [published online July 9, 2013]. Ocul Surf. 2013;11:259-266.
- Maley A, Warren M, Haberman I, et al. Rituximab combined with conventional therapy versus conventional therapy alone for the treatment of mucous membrane pemphigoid (MMP) [published online February 28, 2016]. J Am Acad Dermatol. 2016;74:835-840.
- James WD, Berger TG, Elston D. Chronic blistering diseases. In: James WD, Berger TG, Elston D. Andrews’ Diseases of the Skin: Clinical Dermatology. 11th ed. Philadelphia, PA: Sanders Elsevier; 2010:448-467.
- Neff AG, Turner M, Mutasim DF. Treatment strategies in mucous membrane pemphigoid. Ther Clin Risk Manag. 2008;4:617-626.
- Chan LS, Ahmed AR, Anhalt GJ, et al. The first international consensus on mucous membrane pemphigoid: definition, diagnostic criteria, pathogenic factors, medical treatment, and prognostic indicators. Arch Dermatol. 2002;138:370-379.
- Kato K, Moriyama Y, Saito H, et al. A case of mucous membrane pemphigoid involving the trachea and bronchus with autoantibodies to β3 subunit of laminin-332. Acta Derm Venereol. 2014;94:237-238.
- Gamm DM, Harris A, Mehran RJ, et al. Mucous membrane pemphigoid with fatal bronchial involvement in a seventeen-year-old girl. Cornea. 2006;25:474-478.
- Alexandre M, Brette MD, Pascal F, et al. A prospective study of upper aerodigestive tract manifestations of mucous membrane pemphigoid. Medicine (Baltimore). 2006;85:239-252.
- de Carvalho CR, Amato MB, Da Silva LM, et al. Obstructive respiratory failure in cicatricial pemphigoid. Thorax. 1989;44:601-602.
- Müller LC, Salzer GM. Stenosis of left mainstem bronchus in a case of cicatricial pemphigoid. Eur J Cardiothorac Surg. 1988;2:284-286.
- Camisa C, Allen CM. Death from CP in a young woman with oral, laryngeal, and bronchial involvement. Cutis. 1987;40:426-429.
- Derbes VJ, Pitot HC, Chernosky ME. Fatal cicatricial mucous membrane pemphigoid of the trachea. Dermatol Trop Ecol Geogr. 1962;1:114-117.
- Wieme N, Lambert J, Moerman M, et al. Epidermolysis bullosa acquisita with combined features of bullous pemphigoid and cicatricial pemphigoid. Dermatology. 1999;198:310-313.
- Taylor J, McMillan R, Shephard M, et al. World Workshop on Oral Medicine VI: a systematic review of the treatment of mucous membrane pemphigoid [published online March 11, 2015]. Oral Surg Oral Med Oral Pathol Oral Radiol. 2015;120:161.e20-171.e20.
- Sobolewska B, Deuter C, Zierhut M. Current medical treatment of ocular mucous membrane pemphigoid [published online July 9, 2013]. Ocul Surf. 2013;11:259-266.
- Maley A, Warren M, Haberman I, et al. Rituximab combined with conventional therapy versus conventional therapy alone for the treatment of mucous membrane pemphigoid (MMP) [published online February 28, 2016]. J Am Acad Dermatol. 2016;74:835-840.
Practice Points
- Mucous membrane pemphigoid (MMP) can present with diverse clinical manifestations, making the diagnosis challenging for many clinicians, including experienced dermatologists.
- If not treated early and aggressively, MMP can lead to scarring and is a potentially life-threatening disease.
Aim for modulation with neurotoxin injections
BOSTON – When it comes to chemical denervation, it is best to aim for modulation rather than paralysis, according to Anthony Rossi, MD.
Showing an image of a woman making an exaggerated facial expression, he noted that her treatment had left her overparalyzed in certain areas while other areas were moving. “It’s hard to figure out what she’s trying to emote and what she’s trying to express,” Dr. Rossi of Memorial Sloan Kettering Cancer Center, New York, said at the American Academy of Dermatology summer meeting.
“When we think about facial expressions and when we think about muscles, they work in tandem together, so if things are overparalyzed then some muscles won’t work while others will take over and it will create a perplexed look,” he explained.
A computer analysis of college students demonstrating various facial expressions that represent different emotions identified 21 unique expressions that used a combination of muscles that were different from all other expressions. “There were 6 basic expressions and 15 compound expressions. So as humans, we just need to know we’re using all these facial muscles to convey our message to the world,” Dr. Rossi said.
This finding has particular implications when treating younger versus older patients, he noted.
Younger patients are coming in for cosmetic procedures in increasing numbers, and use of chemical denervation in these patients, compared with in older patients, may be more likely to change facial expressions and appearance. “In the older patient, it may be appropriate to do more cosmetic procedures, but in the younger patient, even the tiniest amount of change can really have a drastic effect on their look,” he said.
As with any surgical or cosmetic case, good patient selection is important for improving patient satisfaction, as is management of patient expectations, he added.
Dr. Rossi compared photos of two women, both in their 40s. One was toxin naive, but the other – despite having scleroderma and undergoing chemotherapy for breast cancer – looked much more “refreshed.” The difference was that the second patient received regular filler and neurotoxin injections, he explained.
To achieve a comparable cosmetic result in the toxin-naive patient, multiple procedures are needed, and procedures should be staged over time, he said.
He noted that it is important to take photos to show patients; photos look different from a mirror image. In addition, static and dynamic rhytids will respond differently to treatment, he pointed out. Static, etched-in rhytids may take longer to correct and may require repeat injections, whereas dynamic rhytids will respond well to neurotoxins, he added.
Combination treatments, such as those incorporating fillers and/or laser resurfacing, may also be needed. “I like to stage my cosmetic procedures,” he said. Staging may not always be possible, but he prefers to allow 2 weeks for a neurotoxin to take effect before adding more neurotoxin or filler.
Among other “pearls” that Dr. Rossi offered for improving outcomes and patient satisfaction were warning patients in advance that static rhytids may require filler in addition to neurotoxin injection, checking for and photographing baseline asymmetry (such as a crooked smile) prior to treatment to avoid blaming the treatment after the fact, and tailoring injections to the patient.
For example, for a particularly expressive individual, it is important to maintain natural movement. For this type of patient, “a standard injection pattern may not work. You still want to give her some natural movement,” he said, explaining that the injection pattern in such a patient would “be more unique, more spread out.
“I usually use multiple rows of injections, but I tend to keep the units that I’m using at a lower dosage so there is some movement, and we don’t drop her brow too much,” he said.
A possible aesthetic trend toward more natural movement – “less frozen, less paralysis” – was suggested by a 2015 retrospective chart review, led by Alastair Carruthers, MD, of the University of British Columbia, Vancouver, showing a decreasing number of units of onabotulinumtoxinA being used over time. In 5,112 treatment sessions in 194 patients over an average of 9 years, the mean dose for forehead lines steadily decreased over time (Dermatol Surg. 2015 Jun;41[6]:693-701).
Dr. Rossi disclosed that he has served on an advisory board for Allergan.
BOSTON – When it comes to chemical denervation, it is best to aim for modulation rather than paralysis, according to Anthony Rossi, MD.
Showing an image of a woman making an exaggerated facial expression, he noted that her treatment had left her overparalyzed in certain areas while other areas were moving. “It’s hard to figure out what she’s trying to emote and what she’s trying to express,” Dr. Rossi of Memorial Sloan Kettering Cancer Center, New York, said at the American Academy of Dermatology summer meeting.
“When we think about facial expressions and when we think about muscles, they work in tandem together, so if things are overparalyzed then some muscles won’t work while others will take over and it will create a perplexed look,” he explained.
A computer analysis of college students demonstrating various facial expressions that represent different emotions identified 21 unique expressions that used a combination of muscles that were different from all other expressions. “There were 6 basic expressions and 15 compound expressions. So as humans, we just need to know we’re using all these facial muscles to convey our message to the world,” Dr. Rossi said.
This finding has particular implications when treating younger versus older patients, he noted.
Younger patients are coming in for cosmetic procedures in increasing numbers, and use of chemical denervation in these patients, compared with in older patients, may be more likely to change facial expressions and appearance. “In the older patient, it may be appropriate to do more cosmetic procedures, but in the younger patient, even the tiniest amount of change can really have a drastic effect on their look,” he said.
As with any surgical or cosmetic case, good patient selection is important for improving patient satisfaction, as is management of patient expectations, he added.
Dr. Rossi compared photos of two women, both in their 40s. One was toxin naive, but the other – despite having scleroderma and undergoing chemotherapy for breast cancer – looked much more “refreshed.” The difference was that the second patient received regular filler and neurotoxin injections, he explained.
To achieve a comparable cosmetic result in the toxin-naive patient, multiple procedures are needed, and procedures should be staged over time, he said.
He noted that it is important to take photos to show patients; photos look different from a mirror image. In addition, static and dynamic rhytids will respond differently to treatment, he pointed out. Static, etched-in rhytids may take longer to correct and may require repeat injections, whereas dynamic rhytids will respond well to neurotoxins, he added.
Combination treatments, such as those incorporating fillers and/or laser resurfacing, may also be needed. “I like to stage my cosmetic procedures,” he said. Staging may not always be possible, but he prefers to allow 2 weeks for a neurotoxin to take effect before adding more neurotoxin or filler.
Among other “pearls” that Dr. Rossi offered for improving outcomes and patient satisfaction were warning patients in advance that static rhytids may require filler in addition to neurotoxin injection, checking for and photographing baseline asymmetry (such as a crooked smile) prior to treatment to avoid blaming the treatment after the fact, and tailoring injections to the patient.
For example, for a particularly expressive individual, it is important to maintain natural movement. For this type of patient, “a standard injection pattern may not work. You still want to give her some natural movement,” he said, explaining that the injection pattern in such a patient would “be more unique, more spread out.
“I usually use multiple rows of injections, but I tend to keep the units that I’m using at a lower dosage so there is some movement, and we don’t drop her brow too much,” he said.
A possible aesthetic trend toward more natural movement – “less frozen, less paralysis” – was suggested by a 2015 retrospective chart review, led by Alastair Carruthers, MD, of the University of British Columbia, Vancouver, showing a decreasing number of units of onabotulinumtoxinA being used over time. In 5,112 treatment sessions in 194 patients over an average of 9 years, the mean dose for forehead lines steadily decreased over time (Dermatol Surg. 2015 Jun;41[6]:693-701).
Dr. Rossi disclosed that he has served on an advisory board for Allergan.
BOSTON – When it comes to chemical denervation, it is best to aim for modulation rather than paralysis, according to Anthony Rossi, MD.
Showing an image of a woman making an exaggerated facial expression, he noted that her treatment had left her overparalyzed in certain areas while other areas were moving. “It’s hard to figure out what she’s trying to emote and what she’s trying to express,” Dr. Rossi of Memorial Sloan Kettering Cancer Center, New York, said at the American Academy of Dermatology summer meeting.
“When we think about facial expressions and when we think about muscles, they work in tandem together, so if things are overparalyzed then some muscles won’t work while others will take over and it will create a perplexed look,” he explained.
A computer analysis of college students demonstrating various facial expressions that represent different emotions identified 21 unique expressions that used a combination of muscles that were different from all other expressions. “There were 6 basic expressions and 15 compound expressions. So as humans, we just need to know we’re using all these facial muscles to convey our message to the world,” Dr. Rossi said.
This finding has particular implications when treating younger versus older patients, he noted.
Younger patients are coming in for cosmetic procedures in increasing numbers, and use of chemical denervation in these patients, compared with in older patients, may be more likely to change facial expressions and appearance. “In the older patient, it may be appropriate to do more cosmetic procedures, but in the younger patient, even the tiniest amount of change can really have a drastic effect on their look,” he said.
As with any surgical or cosmetic case, good patient selection is important for improving patient satisfaction, as is management of patient expectations, he added.
Dr. Rossi compared photos of two women, both in their 40s. One was toxin naive, but the other – despite having scleroderma and undergoing chemotherapy for breast cancer – looked much more “refreshed.” The difference was that the second patient received regular filler and neurotoxin injections, he explained.
To achieve a comparable cosmetic result in the toxin-naive patient, multiple procedures are needed, and procedures should be staged over time, he said.
He noted that it is important to take photos to show patients; photos look different from a mirror image. In addition, static and dynamic rhytids will respond differently to treatment, he pointed out. Static, etched-in rhytids may take longer to correct and may require repeat injections, whereas dynamic rhytids will respond well to neurotoxins, he added.
Combination treatments, such as those incorporating fillers and/or laser resurfacing, may also be needed. “I like to stage my cosmetic procedures,” he said. Staging may not always be possible, but he prefers to allow 2 weeks for a neurotoxin to take effect before adding more neurotoxin or filler.
Among other “pearls” that Dr. Rossi offered for improving outcomes and patient satisfaction were warning patients in advance that static rhytids may require filler in addition to neurotoxin injection, checking for and photographing baseline asymmetry (such as a crooked smile) prior to treatment to avoid blaming the treatment after the fact, and tailoring injections to the patient.
For example, for a particularly expressive individual, it is important to maintain natural movement. For this type of patient, “a standard injection pattern may not work. You still want to give her some natural movement,” he said, explaining that the injection pattern in such a patient would “be more unique, more spread out.
“I usually use multiple rows of injections, but I tend to keep the units that I’m using at a lower dosage so there is some movement, and we don’t drop her brow too much,” he said.
A possible aesthetic trend toward more natural movement – “less frozen, less paralysis” – was suggested by a 2015 retrospective chart review, led by Alastair Carruthers, MD, of the University of British Columbia, Vancouver, showing a decreasing number of units of onabotulinumtoxinA being used over time. In 5,112 treatment sessions in 194 patients over an average of 9 years, the mean dose for forehead lines steadily decreased over time (Dermatol Surg. 2015 Jun;41[6]:693-701).
Dr. Rossi disclosed that he has served on an advisory board for Allergan.
EXPERT ANALYSIS FROM AAD SUMMER ACADEMY 2016
Update on resveratrol
• Found abundantly in nature, this polyphenolic phytoalexin is believed to exhibit a wide range of biologic activity.
• Potent antioxidant, anti-inflammatory, and antiproliferative properties.
• Highly studied polyphenolic substance also considered a chemopreventive agent against skin cancer.
• In small studies, has contributed to antiaging, antiacne, antierythema, and skin-lightening results.
Resveratrol (trans-3,5,4’-trihydroxystilbene), a polyphenolic phytoalexin synthesized in nearly 70 plant species, is found to be particularly abundant in Vitis vinifera (grape vine) and its derivatives (e.g., red wine, purple grape juice), various berries, peanuts, jackfruit, pomegranate, eucalyptus, the roots of Polygonum cuspidatum (Japanese knotweed, which is used in traditional Chinese and Japanese medicine to treat dermatitis, among other conditions)1, Scots pine, spruce, corn lily, gnetum, and butterfly orchid.2-6 Several studies have demonstrated that resveratrol possesses potent antioxidant, anticarcinogenic, anti-inflammatory, as well as antimicrobial characteristics.7-11 Specifically, in vitro and in vivo studies have shown that resveratrol exerts chemopreventive and antiproliferative activity against various cancers, including skin cancer, by suppressing cellular events associated with tumor initiation, promotion, and progression, and triggering apoptosis in such tumor cells.12-14 It also is reputed to impart antiaging benefits.15 This column will focus on recent research findings pertaining to effects on the skin as well as topical uses of this botanical agent, the main source of which, V. vinifera, has been used since antiquity.
Resveratrol was first identified from the roots of Veratrum grandiflorum (white hellebore) in 1940,16-19 but research on the compound did not take root until after a 1997 report in Science suggested chemopreventive properties. In that study, purified resveratrol was found to exhibit major inhibitory activity against cancer initiation, promotion, and progression.20 Since then, copious research on this botanical compound has yielded a reputation as a strong antioxidant, anti-inflammatory, and antiproliferative agent.7,21,22 Most importantly, resveratrol is considered to act as a chemopreventive agent against skin cancer and antiproliferative influence on oral squamous, breast, colon, and prostate cancer cells.12,14 It is also one of the most studied polyphenolic compounds.
Skin cancer and photoprotection
In 2012, Osmond et al. conducted in vitro and in vivo experiments to assess the potential of resveratrol as a chemotherapy adjunct for melanoma treatment. Resveratrol significantly reduced melanoma cell viability in both melanoma cell lines tested, and selectively spared cells in the nonmalignant fibroblast lines, compared with its cytotoxic impact on melanoma cells. Further, cytotoxicity to malignant cells was greatly enhanced by 72 hours of treatment with resveratrol and temozolomide, compared with temozolomide treatment alone. No significant differences were seen in vivo. The researchers concluded that the in vitro antitumor activity of resveratrol suggested its potential as a therapeutic agent in melanoma management.23
Resveratrol has been shown to protect against UVB-mediated cutaneous damage in SKH-1 hairless mice. Afaq et al. demonstrated that UVB-induced skin edema was significantly suppressed by the topical application of resveratrol to SKH-1 hairless mice.7
In a different study by the same team, topically applied resveratrol significantly inhibited UVB-mediated increases in bifold skin thickness and edema and greatly diminished UVB-induced lipid peroxidation, cyclo-oxygenase and ornithine decarboxylase (ODC) activities, as well as protein expression of the ODC enzyme in SKH-1 hairless mice.24 In an experiment by some of the same researchers, resveratrol was topically applied to SKH-1 hairless mice 30 minutes before exposure to UVB; 24 hours later, significant decreases were observed in bifold skin thickness, hyperplasia, and leukocyte infiltration. Critical cell cycle regulatory proteins, the target of the investigation, were substantially down-regulated because of the resveratrol. The investigators concluded that resveratrol might have the potential to play a significant role in preventing UVB-mediated photodamage and carcinogenesis.25
More recently, in 2015, Sirerol et al. found that the topical treatment with pterostilbene, a natural dimethoxy analog of resveratrol, effectively shielded SKH-1 hairless mice from UVB-induced photodamage and carcinogenesis.26
Interestingly, recent in vitro studies by Sticozzi and colleagues have shown that topical resveratrol dose-dependently protected human keratinocytes from cigarette smoke–induced reduction of scavenger receptor B1 protein expression27 and can lower cigarette smoke–induced reactive oxygen species and carbonyl formation in human keratinocytes.28
Antiaging activity
In 2010, Giardina et al. performed an in vitro study to evaluate the tonic-trophic characteristics of resveratrol alone and resveratrol plus N-acetyl-cysteine on cultured skin fibroblasts. Both formulations dose-dependently increased cell proliferation and inhibition of collagenase activity.29
In 2012, Wu et al. investigated the protective effects of resveratrate, a stable derivative of resveratrol, against damage to human skin caused by repetitive solar simulator UV radiation (ssUVR) in 15 healthy human volunteers. Six sites on nonexposed dorsal skin of each participant were assessed, with four sites exposed to ssUVR and the remaining sites serving as positive control (ssUVR only) and baseline control (no treatment or exposure). The researchers noted minimal erythema on areas treated with resveratrate and the resveratrol derivative significantly inhibited sunburn cell formation. They concluded that resveratrate protects the skin against sunburn and suntan caused by repetitive ssUVR.4
Also that year, Buonocore et al. conducted a placebo-controlled, double-blind study in 50 subjects that revealed the antiaging efficacy of a dietary nutraceutical blend of resveratrol and procyanidin. Specifically, skin moisturization and elasticity improved while wrinkle depth and skin roughness lessened after 60 days of treatment.30
In a 2013 in vitro study of the skin permeation kinetics of polyphenols using diffusion cells via ex vivo pig skin and a cellulose membrane, Zillich et al. showed that several polyphenols, including resveratrol, epigallocatechin gallate, quercetin, rutin, and protocatechuic acid formulated in oil-in-water emulsions could permeate the stratum corneum and were identified in the epidermis and dermis. The team concluded that their findings validate the use of polyphenols as active ingredients in antiaging products.31
In 2014, Farris et al. found that the topical application of resveratrol in a proprietary blend (1% resveratrol, 0.5% baicalin, 1% vitamin E) yielded a statistically significant amelioration of fine lines and wrinkles, hyperpigmentation, radiance, as well as skin roughness, firmness, elasticity, and laxity in a small study over 12 weeks.32
Skin lightening
Resveratrol has also been used as a promising topical treatment for hyperpigmentation disorders.33 The compound has been shown to work synergistically with 4-n-butylresorcinol (a derivative of resorcinol, one of the main phenols found in argan oil) to reduce tyrosinase levels and significantly diminish melanin synthesis, more effectively than either compound alone.34 In 2012, Franco et al. observed that resveratrol can inhibit tyrosinase but it does not sufficiently suppress melanin production to justify its use as a lone skin-whitening agent in pharmaceutical formulations, but warrants attention as a coadjuvant for treating hyperpigmentation.35 The skin-lightening capacity of resveratrol supported by a 2013 study in which 52 medicinal plants grown in Korea were tested for human tyrosinase activity and the dried stems of the grape tree V. vinifera were found to potently suppress human tyrosinase, and more effectively than arbutin.36 It is worth noting that resveratrol, through its antioxidant activity and possible inhibitory effect on cytochrome P450 2E1 expression, has been shown to protect mouse primary hepatocytes from hydroquinone-induced cytotoxicity.37 Also, J.M. Galgut and S.A. Ali, noted the gathering of pigment cells and resultant skin lightening from the effects of topical ethanolic extract of Arachis hypogaea (peanuts, which contain half the resveratrol of red wine) on the tail melanophores of tadpole Bufo melanostictus, concluding that resveratrol merits attention for potential clinical use as a nontoxic melanolytic agent to treat hyperpigmentation.38,34
Acne
Resveratrol is considered an emerging agent in the topical armamentarium for treating acne.39 In a single-blind pilot study in 2011, Fabbrocini et al. investigated the potential therapeutic impact of resveratrol on 20 patients with acne. A resveratrol-containing hydrogel was applied daily on the right side of the face for 60 days, with the left side receiving the hydrogel vehicle as control. No adverse effects were reported and all patients were satisfied with the treatment. Researchers reported a 53.75 % mean reduction in the Global Acne Grading System score on the resveratrol-treated sides and a 6.10 % decrease on the control sides. Histologic analysis revealed a statistically significant reduction of lesions in areas treated with resveratrol (66.7 % mean reduction in the average area of microcomedones on the resveratrol-treated sides vs. 9.7% reduction on the control sides).40
A 2015 comprehensive literature review of randomized clinical trials and controlled trials by Dall’oglio et al. found that cosmetics with antimicrobial and anti-inflammatory ingredients, including resveratrol, may accelerate acne resolution.41
Significantly, resveratrol is one of the novel acne treatments considered to have little potential for susceptibility to antibiotic resistance to Propionibacterium acnes.42
Erythema
In a small 2013 study by Ferzli et al., 16 subjects with erythema applied a formulation containing resveratrol, green tea polyphenols, and caffeine twice daily to the whole face. Clinical photographs and spectrally enhanced images taken before treatment and every 2 weeks through 3 months were assessed. The investigators reported that improvement was seen in 16 of 16 clinical images and 13 of 16 spectrally enhanced images. Erythema reduction was observable by 6 weeks of treatment, and no adverse effects were observed.43
Conclusion
Resveratrol is emerging as a compound with the potential to deliver significant health benefits, particularly in terms of photoprotective, cancer preventive, and cardioprotective activity. While there are 2 strong decades of research, more is necessary to elucidate the full potential of resveratrol as a first-line dermatologic therapy. Preclinical data do support the use of resveratrol in various product types (e.g., emollients, patches, sunscreens, and additional skin care products) intended to prevent skin cancer or to prevent or treat other conditions caused or exacerbated by solar exposure, such as photoaging. I look forward to seeing clinical evidence of the efficacy of topically applied resveratrol.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin-typing systems and related products.
References
1. Toxicol Appl Pharmacol. 2003 Jan 1;186(1):28-37.
2. Science. 1997 Jan 10;275(5297):218-20.
3. Cancer Res. 2001 Feb 15;61(4):1604-10.
4. J Eur Acad Dermatol Venereol. 2013 Mar;27(3):345-50.
5. Photochem Photobiol. 2008 Mar-Apr;84(2):415-21.
6. Dose Response. 2010 Mar 18;8(4):478-500.
7. Toxicol Appl Pharmacol. 2003 Jan 1;186(1):28-37.
8. Neoplasia. 2003 Jan-Feb;5(1):74-82.
9. An Illustrated Guide to 101 Medicinal Herbs: Their History, Use, Recommended Dosages, and Cautions (Loveland, Colo.: Interweave Press, 1998, pp. 108-9.)
11. Eur J Pharm Sci. 2015 Oct 12;78:204-13.
12. Pancreas. 2002 Nov;25(4):e71-6.
13. Toxicol Appl Pharmacol. 2007 Nov 1;224(3):274-83.
14. J Biol Chem. 2003 Oct 17;278(42):41482-90.
15. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
16. Fitoterapia. 2013 Apr;86:84-91.
17. Ann N Y Acad Sci. 2011 Jan;1215:60-71.
18. Recent Pat Cardiovasc Drug Discov. 2007 Jun;2(2):133-8.
19. Anticancer Res. 2004 Sep-Oct;24(5A):2783-840.
20. Science. 1997 Jan 10;275(5297):218-20.
21. Biochem Pharmacol. 2002 Jan 15:63(2):99-104.
22. Biomed Pap Med Fac univ Palacky Olomouc Czech Repub. 2003 Dec;147(2):137-45.
23. J Surg Res. 2012 Jan;172(1):109-15.
24. Front Biosci. 2002 Apr 1;7:d784-92.
25. Oncogene. 2004 Jul 1;23(30):5151-60.
26. Free Radic Biol Med. 2015 Aug;85:1-11.
27. Free Radic Biol Med. 2014 Apr;69:50-7.
28. Food Funct. 2014 Sep;5(9):2348-56.
29. Minerva Ginecol. 2010 Jun;62(3):195-201.
30. Clin Cosmet Investig Dermatol. 2012;5:159-65.
31. Int J Cosmet Sci. 2013 Oct;35(5):491-501.
32. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
33. Semin Cutan Med Surg. 2012 Jun;31(2):133-9.
34. Pharmazie. 2012 Jun;67(6):542-6.
35. Molecules. 2012 Oct 9;17(10):11816-25.
36. Evid Based Complement Alternat Med. 2013;2013:645257.
37. Int J Environ Res Public Health. 2012 Sep 19;9(9):3354-64.
38. J Recept Signal Transduct Res. 2011 Oct;31(5):374-80.
39. Am J Clin Dermatol. 2012 Dec 1;13(6):357-64.
40. Am J Clin Dermatol. 2011 Apr 1;12(2):133-41.
41. G Ital Dermatol Venereol. 2015 Feb;150(1):1-11.
• Found abundantly in nature, this polyphenolic phytoalexin is believed to exhibit a wide range of biologic activity.
• Potent antioxidant, anti-inflammatory, and antiproliferative properties.
• Highly studied polyphenolic substance also considered a chemopreventive agent against skin cancer.
• In small studies, has contributed to antiaging, antiacne, antierythema, and skin-lightening results.
Resveratrol (trans-3,5,4’-trihydroxystilbene), a polyphenolic phytoalexin synthesized in nearly 70 plant species, is found to be particularly abundant in Vitis vinifera (grape vine) and its derivatives (e.g., red wine, purple grape juice), various berries, peanuts, jackfruit, pomegranate, eucalyptus, the roots of Polygonum cuspidatum (Japanese knotweed, which is used in traditional Chinese and Japanese medicine to treat dermatitis, among other conditions)1, Scots pine, spruce, corn lily, gnetum, and butterfly orchid.2-6 Several studies have demonstrated that resveratrol possesses potent antioxidant, anticarcinogenic, anti-inflammatory, as well as antimicrobial characteristics.7-11 Specifically, in vitro and in vivo studies have shown that resveratrol exerts chemopreventive and antiproliferative activity against various cancers, including skin cancer, by suppressing cellular events associated with tumor initiation, promotion, and progression, and triggering apoptosis in such tumor cells.12-14 It also is reputed to impart antiaging benefits.15 This column will focus on recent research findings pertaining to effects on the skin as well as topical uses of this botanical agent, the main source of which, V. vinifera, has been used since antiquity.
Resveratrol was first identified from the roots of Veratrum grandiflorum (white hellebore) in 1940,16-19 but research on the compound did not take root until after a 1997 report in Science suggested chemopreventive properties. In that study, purified resveratrol was found to exhibit major inhibitory activity against cancer initiation, promotion, and progression.20 Since then, copious research on this botanical compound has yielded a reputation as a strong antioxidant, anti-inflammatory, and antiproliferative agent.7,21,22 Most importantly, resveratrol is considered to act as a chemopreventive agent against skin cancer and antiproliferative influence on oral squamous, breast, colon, and prostate cancer cells.12,14 It is also one of the most studied polyphenolic compounds.
Skin cancer and photoprotection
In 2012, Osmond et al. conducted in vitro and in vivo experiments to assess the potential of resveratrol as a chemotherapy adjunct for melanoma treatment. Resveratrol significantly reduced melanoma cell viability in both melanoma cell lines tested, and selectively spared cells in the nonmalignant fibroblast lines, compared with its cytotoxic impact on melanoma cells. Further, cytotoxicity to malignant cells was greatly enhanced by 72 hours of treatment with resveratrol and temozolomide, compared with temozolomide treatment alone. No significant differences were seen in vivo. The researchers concluded that the in vitro antitumor activity of resveratrol suggested its potential as a therapeutic agent in melanoma management.23
Resveratrol has been shown to protect against UVB-mediated cutaneous damage in SKH-1 hairless mice. Afaq et al. demonstrated that UVB-induced skin edema was significantly suppressed by the topical application of resveratrol to SKH-1 hairless mice.7
In a different study by the same team, topically applied resveratrol significantly inhibited UVB-mediated increases in bifold skin thickness and edema and greatly diminished UVB-induced lipid peroxidation, cyclo-oxygenase and ornithine decarboxylase (ODC) activities, as well as protein expression of the ODC enzyme in SKH-1 hairless mice.24 In an experiment by some of the same researchers, resveratrol was topically applied to SKH-1 hairless mice 30 minutes before exposure to UVB; 24 hours later, significant decreases were observed in bifold skin thickness, hyperplasia, and leukocyte infiltration. Critical cell cycle regulatory proteins, the target of the investigation, were substantially down-regulated because of the resveratrol. The investigators concluded that resveratrol might have the potential to play a significant role in preventing UVB-mediated photodamage and carcinogenesis.25
More recently, in 2015, Sirerol et al. found that the topical treatment with pterostilbene, a natural dimethoxy analog of resveratrol, effectively shielded SKH-1 hairless mice from UVB-induced photodamage and carcinogenesis.26
Interestingly, recent in vitro studies by Sticozzi and colleagues have shown that topical resveratrol dose-dependently protected human keratinocytes from cigarette smoke–induced reduction of scavenger receptor B1 protein expression27 and can lower cigarette smoke–induced reactive oxygen species and carbonyl formation in human keratinocytes.28
Antiaging activity
In 2010, Giardina et al. performed an in vitro study to evaluate the tonic-trophic characteristics of resveratrol alone and resveratrol plus N-acetyl-cysteine on cultured skin fibroblasts. Both formulations dose-dependently increased cell proliferation and inhibition of collagenase activity.29
In 2012, Wu et al. investigated the protective effects of resveratrate, a stable derivative of resveratrol, against damage to human skin caused by repetitive solar simulator UV radiation (ssUVR) in 15 healthy human volunteers. Six sites on nonexposed dorsal skin of each participant were assessed, with four sites exposed to ssUVR and the remaining sites serving as positive control (ssUVR only) and baseline control (no treatment or exposure). The researchers noted minimal erythema on areas treated with resveratrate and the resveratrol derivative significantly inhibited sunburn cell formation. They concluded that resveratrate protects the skin against sunburn and suntan caused by repetitive ssUVR.4
Also that year, Buonocore et al. conducted a placebo-controlled, double-blind study in 50 subjects that revealed the antiaging efficacy of a dietary nutraceutical blend of resveratrol and procyanidin. Specifically, skin moisturization and elasticity improved while wrinkle depth and skin roughness lessened after 60 days of treatment.30
In a 2013 in vitro study of the skin permeation kinetics of polyphenols using diffusion cells via ex vivo pig skin and a cellulose membrane, Zillich et al. showed that several polyphenols, including resveratrol, epigallocatechin gallate, quercetin, rutin, and protocatechuic acid formulated in oil-in-water emulsions could permeate the stratum corneum and were identified in the epidermis and dermis. The team concluded that their findings validate the use of polyphenols as active ingredients in antiaging products.31
In 2014, Farris et al. found that the topical application of resveratrol in a proprietary blend (1% resveratrol, 0.5% baicalin, 1% vitamin E) yielded a statistically significant amelioration of fine lines and wrinkles, hyperpigmentation, radiance, as well as skin roughness, firmness, elasticity, and laxity in a small study over 12 weeks.32
Skin lightening
Resveratrol has also been used as a promising topical treatment for hyperpigmentation disorders.33 The compound has been shown to work synergistically with 4-n-butylresorcinol (a derivative of resorcinol, one of the main phenols found in argan oil) to reduce tyrosinase levels and significantly diminish melanin synthesis, more effectively than either compound alone.34 In 2012, Franco et al. observed that resveratrol can inhibit tyrosinase but it does not sufficiently suppress melanin production to justify its use as a lone skin-whitening agent in pharmaceutical formulations, but warrants attention as a coadjuvant for treating hyperpigmentation.35 The skin-lightening capacity of resveratrol supported by a 2013 study in which 52 medicinal plants grown in Korea were tested for human tyrosinase activity and the dried stems of the grape tree V. vinifera were found to potently suppress human tyrosinase, and more effectively than arbutin.36 It is worth noting that resveratrol, through its antioxidant activity and possible inhibitory effect on cytochrome P450 2E1 expression, has been shown to protect mouse primary hepatocytes from hydroquinone-induced cytotoxicity.37 Also, J.M. Galgut and S.A. Ali, noted the gathering of pigment cells and resultant skin lightening from the effects of topical ethanolic extract of Arachis hypogaea (peanuts, which contain half the resveratrol of red wine) on the tail melanophores of tadpole Bufo melanostictus, concluding that resveratrol merits attention for potential clinical use as a nontoxic melanolytic agent to treat hyperpigmentation.38,34
Acne
Resveratrol is considered an emerging agent in the topical armamentarium for treating acne.39 In a single-blind pilot study in 2011, Fabbrocini et al. investigated the potential therapeutic impact of resveratrol on 20 patients with acne. A resveratrol-containing hydrogel was applied daily on the right side of the face for 60 days, with the left side receiving the hydrogel vehicle as control. No adverse effects were reported and all patients were satisfied with the treatment. Researchers reported a 53.75 % mean reduction in the Global Acne Grading System score on the resveratrol-treated sides and a 6.10 % decrease on the control sides. Histologic analysis revealed a statistically significant reduction of lesions in areas treated with resveratrol (66.7 % mean reduction in the average area of microcomedones on the resveratrol-treated sides vs. 9.7% reduction on the control sides).40
A 2015 comprehensive literature review of randomized clinical trials and controlled trials by Dall’oglio et al. found that cosmetics with antimicrobial and anti-inflammatory ingredients, including resveratrol, may accelerate acne resolution.41
Significantly, resveratrol is one of the novel acne treatments considered to have little potential for susceptibility to antibiotic resistance to Propionibacterium acnes.42
Erythema
In a small 2013 study by Ferzli et al., 16 subjects with erythema applied a formulation containing resveratrol, green tea polyphenols, and caffeine twice daily to the whole face. Clinical photographs and spectrally enhanced images taken before treatment and every 2 weeks through 3 months were assessed. The investigators reported that improvement was seen in 16 of 16 clinical images and 13 of 16 spectrally enhanced images. Erythema reduction was observable by 6 weeks of treatment, and no adverse effects were observed.43
Conclusion
Resveratrol is emerging as a compound with the potential to deliver significant health benefits, particularly in terms of photoprotective, cancer preventive, and cardioprotective activity. While there are 2 strong decades of research, more is necessary to elucidate the full potential of resveratrol as a first-line dermatologic therapy. Preclinical data do support the use of resveratrol in various product types (e.g., emollients, patches, sunscreens, and additional skin care products) intended to prevent skin cancer or to prevent or treat other conditions caused or exacerbated by solar exposure, such as photoaging. I look forward to seeing clinical evidence of the efficacy of topically applied resveratrol.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin-typing systems and related products.
References
1. Toxicol Appl Pharmacol. 2003 Jan 1;186(1):28-37.
2. Science. 1997 Jan 10;275(5297):218-20.
3. Cancer Res. 2001 Feb 15;61(4):1604-10.
4. J Eur Acad Dermatol Venereol. 2013 Mar;27(3):345-50.
5. Photochem Photobiol. 2008 Mar-Apr;84(2):415-21.
6. Dose Response. 2010 Mar 18;8(4):478-500.
7. Toxicol Appl Pharmacol. 2003 Jan 1;186(1):28-37.
8. Neoplasia. 2003 Jan-Feb;5(1):74-82.
9. An Illustrated Guide to 101 Medicinal Herbs: Their History, Use, Recommended Dosages, and Cautions (Loveland, Colo.: Interweave Press, 1998, pp. 108-9.)
11. Eur J Pharm Sci. 2015 Oct 12;78:204-13.
12. Pancreas. 2002 Nov;25(4):e71-6.
13. Toxicol Appl Pharmacol. 2007 Nov 1;224(3):274-83.
14. J Biol Chem. 2003 Oct 17;278(42):41482-90.
15. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
16. Fitoterapia. 2013 Apr;86:84-91.
17. Ann N Y Acad Sci. 2011 Jan;1215:60-71.
18. Recent Pat Cardiovasc Drug Discov. 2007 Jun;2(2):133-8.
19. Anticancer Res. 2004 Sep-Oct;24(5A):2783-840.
20. Science. 1997 Jan 10;275(5297):218-20.
21. Biochem Pharmacol. 2002 Jan 15:63(2):99-104.
22. Biomed Pap Med Fac univ Palacky Olomouc Czech Repub. 2003 Dec;147(2):137-45.
23. J Surg Res. 2012 Jan;172(1):109-15.
24. Front Biosci. 2002 Apr 1;7:d784-92.
25. Oncogene. 2004 Jul 1;23(30):5151-60.
26. Free Radic Biol Med. 2015 Aug;85:1-11.
27. Free Radic Biol Med. 2014 Apr;69:50-7.
28. Food Funct. 2014 Sep;5(9):2348-56.
29. Minerva Ginecol. 2010 Jun;62(3):195-201.
30. Clin Cosmet Investig Dermatol. 2012;5:159-65.
31. Int J Cosmet Sci. 2013 Oct;35(5):491-501.
32. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
33. Semin Cutan Med Surg. 2012 Jun;31(2):133-9.
34. Pharmazie. 2012 Jun;67(6):542-6.
35. Molecules. 2012 Oct 9;17(10):11816-25.
36. Evid Based Complement Alternat Med. 2013;2013:645257.
37. Int J Environ Res Public Health. 2012 Sep 19;9(9):3354-64.
38. J Recept Signal Transduct Res. 2011 Oct;31(5):374-80.
39. Am J Clin Dermatol. 2012 Dec 1;13(6):357-64.
40. Am J Clin Dermatol. 2011 Apr 1;12(2):133-41.
41. G Ital Dermatol Venereol. 2015 Feb;150(1):1-11.
• Found abundantly in nature, this polyphenolic phytoalexin is believed to exhibit a wide range of biologic activity.
• Potent antioxidant, anti-inflammatory, and antiproliferative properties.
• Highly studied polyphenolic substance also considered a chemopreventive agent against skin cancer.
• In small studies, has contributed to antiaging, antiacne, antierythema, and skin-lightening results.
Resveratrol (trans-3,5,4’-trihydroxystilbene), a polyphenolic phytoalexin synthesized in nearly 70 plant species, is found to be particularly abundant in Vitis vinifera (grape vine) and its derivatives (e.g., red wine, purple grape juice), various berries, peanuts, jackfruit, pomegranate, eucalyptus, the roots of Polygonum cuspidatum (Japanese knotweed, which is used in traditional Chinese and Japanese medicine to treat dermatitis, among other conditions)1, Scots pine, spruce, corn lily, gnetum, and butterfly orchid.2-6 Several studies have demonstrated that resveratrol possesses potent antioxidant, anticarcinogenic, anti-inflammatory, as well as antimicrobial characteristics.7-11 Specifically, in vitro and in vivo studies have shown that resveratrol exerts chemopreventive and antiproliferative activity against various cancers, including skin cancer, by suppressing cellular events associated with tumor initiation, promotion, and progression, and triggering apoptosis in such tumor cells.12-14 It also is reputed to impart antiaging benefits.15 This column will focus on recent research findings pertaining to effects on the skin as well as topical uses of this botanical agent, the main source of which, V. vinifera, has been used since antiquity.
Resveratrol was first identified from the roots of Veratrum grandiflorum (white hellebore) in 1940,16-19 but research on the compound did not take root until after a 1997 report in Science suggested chemopreventive properties. In that study, purified resveratrol was found to exhibit major inhibitory activity against cancer initiation, promotion, and progression.20 Since then, copious research on this botanical compound has yielded a reputation as a strong antioxidant, anti-inflammatory, and antiproliferative agent.7,21,22 Most importantly, resveratrol is considered to act as a chemopreventive agent against skin cancer and antiproliferative influence on oral squamous, breast, colon, and prostate cancer cells.12,14 It is also one of the most studied polyphenolic compounds.
Skin cancer and photoprotection
In 2012, Osmond et al. conducted in vitro and in vivo experiments to assess the potential of resveratrol as a chemotherapy adjunct for melanoma treatment. Resveratrol significantly reduced melanoma cell viability in both melanoma cell lines tested, and selectively spared cells in the nonmalignant fibroblast lines, compared with its cytotoxic impact on melanoma cells. Further, cytotoxicity to malignant cells was greatly enhanced by 72 hours of treatment with resveratrol and temozolomide, compared with temozolomide treatment alone. No significant differences were seen in vivo. The researchers concluded that the in vitro antitumor activity of resveratrol suggested its potential as a therapeutic agent in melanoma management.23
Resveratrol has been shown to protect against UVB-mediated cutaneous damage in SKH-1 hairless mice. Afaq et al. demonstrated that UVB-induced skin edema was significantly suppressed by the topical application of resveratrol to SKH-1 hairless mice.7
In a different study by the same team, topically applied resveratrol significantly inhibited UVB-mediated increases in bifold skin thickness and edema and greatly diminished UVB-induced lipid peroxidation, cyclo-oxygenase and ornithine decarboxylase (ODC) activities, as well as protein expression of the ODC enzyme in SKH-1 hairless mice.24 In an experiment by some of the same researchers, resveratrol was topically applied to SKH-1 hairless mice 30 minutes before exposure to UVB; 24 hours later, significant decreases were observed in bifold skin thickness, hyperplasia, and leukocyte infiltration. Critical cell cycle regulatory proteins, the target of the investigation, were substantially down-regulated because of the resveratrol. The investigators concluded that resveratrol might have the potential to play a significant role in preventing UVB-mediated photodamage and carcinogenesis.25
More recently, in 2015, Sirerol et al. found that the topical treatment with pterostilbene, a natural dimethoxy analog of resveratrol, effectively shielded SKH-1 hairless mice from UVB-induced photodamage and carcinogenesis.26
Interestingly, recent in vitro studies by Sticozzi and colleagues have shown that topical resveratrol dose-dependently protected human keratinocytes from cigarette smoke–induced reduction of scavenger receptor B1 protein expression27 and can lower cigarette smoke–induced reactive oxygen species and carbonyl formation in human keratinocytes.28
Antiaging activity
In 2010, Giardina et al. performed an in vitro study to evaluate the tonic-trophic characteristics of resveratrol alone and resveratrol plus N-acetyl-cysteine on cultured skin fibroblasts. Both formulations dose-dependently increased cell proliferation and inhibition of collagenase activity.29
In 2012, Wu et al. investigated the protective effects of resveratrate, a stable derivative of resveratrol, against damage to human skin caused by repetitive solar simulator UV radiation (ssUVR) in 15 healthy human volunteers. Six sites on nonexposed dorsal skin of each participant were assessed, with four sites exposed to ssUVR and the remaining sites serving as positive control (ssUVR only) and baseline control (no treatment or exposure). The researchers noted minimal erythema on areas treated with resveratrate and the resveratrol derivative significantly inhibited sunburn cell formation. They concluded that resveratrate protects the skin against sunburn and suntan caused by repetitive ssUVR.4
Also that year, Buonocore et al. conducted a placebo-controlled, double-blind study in 50 subjects that revealed the antiaging efficacy of a dietary nutraceutical blend of resveratrol and procyanidin. Specifically, skin moisturization and elasticity improved while wrinkle depth and skin roughness lessened after 60 days of treatment.30
In a 2013 in vitro study of the skin permeation kinetics of polyphenols using diffusion cells via ex vivo pig skin and a cellulose membrane, Zillich et al. showed that several polyphenols, including resveratrol, epigallocatechin gallate, quercetin, rutin, and protocatechuic acid formulated in oil-in-water emulsions could permeate the stratum corneum and were identified in the epidermis and dermis. The team concluded that their findings validate the use of polyphenols as active ingredients in antiaging products.31
In 2014, Farris et al. found that the topical application of resveratrol in a proprietary blend (1% resveratrol, 0.5% baicalin, 1% vitamin E) yielded a statistically significant amelioration of fine lines and wrinkles, hyperpigmentation, radiance, as well as skin roughness, firmness, elasticity, and laxity in a small study over 12 weeks.32
Skin lightening
Resveratrol has also been used as a promising topical treatment for hyperpigmentation disorders.33 The compound has been shown to work synergistically with 4-n-butylresorcinol (a derivative of resorcinol, one of the main phenols found in argan oil) to reduce tyrosinase levels and significantly diminish melanin synthesis, more effectively than either compound alone.34 In 2012, Franco et al. observed that resveratrol can inhibit tyrosinase but it does not sufficiently suppress melanin production to justify its use as a lone skin-whitening agent in pharmaceutical formulations, but warrants attention as a coadjuvant for treating hyperpigmentation.35 The skin-lightening capacity of resveratrol supported by a 2013 study in which 52 medicinal plants grown in Korea were tested for human tyrosinase activity and the dried stems of the grape tree V. vinifera were found to potently suppress human tyrosinase, and more effectively than arbutin.36 It is worth noting that resveratrol, through its antioxidant activity and possible inhibitory effect on cytochrome P450 2E1 expression, has been shown to protect mouse primary hepatocytes from hydroquinone-induced cytotoxicity.37 Also, J.M. Galgut and S.A. Ali, noted the gathering of pigment cells and resultant skin lightening from the effects of topical ethanolic extract of Arachis hypogaea (peanuts, which contain half the resveratrol of red wine) on the tail melanophores of tadpole Bufo melanostictus, concluding that resveratrol merits attention for potential clinical use as a nontoxic melanolytic agent to treat hyperpigmentation.38,34
Acne
Resveratrol is considered an emerging agent in the topical armamentarium for treating acne.39 In a single-blind pilot study in 2011, Fabbrocini et al. investigated the potential therapeutic impact of resveratrol on 20 patients with acne. A resveratrol-containing hydrogel was applied daily on the right side of the face for 60 days, with the left side receiving the hydrogel vehicle as control. No adverse effects were reported and all patients were satisfied with the treatment. Researchers reported a 53.75 % mean reduction in the Global Acne Grading System score on the resveratrol-treated sides and a 6.10 % decrease on the control sides. Histologic analysis revealed a statistically significant reduction of lesions in areas treated with resveratrol (66.7 % mean reduction in the average area of microcomedones on the resveratrol-treated sides vs. 9.7% reduction on the control sides).40
A 2015 comprehensive literature review of randomized clinical trials and controlled trials by Dall’oglio et al. found that cosmetics with antimicrobial and anti-inflammatory ingredients, including resveratrol, may accelerate acne resolution.41
Significantly, resveratrol is one of the novel acne treatments considered to have little potential for susceptibility to antibiotic resistance to Propionibacterium acnes.42
Erythema
In a small 2013 study by Ferzli et al., 16 subjects with erythema applied a formulation containing resveratrol, green tea polyphenols, and caffeine twice daily to the whole face. Clinical photographs and spectrally enhanced images taken before treatment and every 2 weeks through 3 months were assessed. The investigators reported that improvement was seen in 16 of 16 clinical images and 13 of 16 spectrally enhanced images. Erythema reduction was observable by 6 weeks of treatment, and no adverse effects were observed.43
Conclusion
Resveratrol is emerging as a compound with the potential to deliver significant health benefits, particularly in terms of photoprotective, cancer preventive, and cardioprotective activity. While there are 2 strong decades of research, more is necessary to elucidate the full potential of resveratrol as a first-line dermatologic therapy. Preclinical data do support the use of resveratrol in various product types (e.g., emollients, patches, sunscreens, and additional skin care products) intended to prevent skin cancer or to prevent or treat other conditions caused or exacerbated by solar exposure, such as photoaging. I look forward to seeing clinical evidence of the efficacy of topically applied resveratrol.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin-typing systems and related products.
References
1. Toxicol Appl Pharmacol. 2003 Jan 1;186(1):28-37.
2. Science. 1997 Jan 10;275(5297):218-20.
3. Cancer Res. 2001 Feb 15;61(4):1604-10.
4. J Eur Acad Dermatol Venereol. 2013 Mar;27(3):345-50.
5. Photochem Photobiol. 2008 Mar-Apr;84(2):415-21.
6. Dose Response. 2010 Mar 18;8(4):478-500.
7. Toxicol Appl Pharmacol. 2003 Jan 1;186(1):28-37.
8. Neoplasia. 2003 Jan-Feb;5(1):74-82.
9. An Illustrated Guide to 101 Medicinal Herbs: Their History, Use, Recommended Dosages, and Cautions (Loveland, Colo.: Interweave Press, 1998, pp. 108-9.)
11. Eur J Pharm Sci. 2015 Oct 12;78:204-13.
12. Pancreas. 2002 Nov;25(4):e71-6.
13. Toxicol Appl Pharmacol. 2007 Nov 1;224(3):274-83.
14. J Biol Chem. 2003 Oct 17;278(42):41482-90.
15. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
16. Fitoterapia. 2013 Apr;86:84-91.
17. Ann N Y Acad Sci. 2011 Jan;1215:60-71.
18. Recent Pat Cardiovasc Drug Discov. 2007 Jun;2(2):133-8.
19. Anticancer Res. 2004 Sep-Oct;24(5A):2783-840.
20. Science. 1997 Jan 10;275(5297):218-20.
21. Biochem Pharmacol. 2002 Jan 15:63(2):99-104.
22. Biomed Pap Med Fac univ Palacky Olomouc Czech Repub. 2003 Dec;147(2):137-45.
23. J Surg Res. 2012 Jan;172(1):109-15.
24. Front Biosci. 2002 Apr 1;7:d784-92.
25. Oncogene. 2004 Jul 1;23(30):5151-60.
26. Free Radic Biol Med. 2015 Aug;85:1-11.
27. Free Radic Biol Med. 2014 Apr;69:50-7.
28. Food Funct. 2014 Sep;5(9):2348-56.
29. Minerva Ginecol. 2010 Jun;62(3):195-201.
30. Clin Cosmet Investig Dermatol. 2012;5:159-65.
31. Int J Cosmet Sci. 2013 Oct;35(5):491-501.
32. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
33. Semin Cutan Med Surg. 2012 Jun;31(2):133-9.
34. Pharmazie. 2012 Jun;67(6):542-6.
35. Molecules. 2012 Oct 9;17(10):11816-25.
36. Evid Based Complement Alternat Med. 2013;2013:645257.
37. Int J Environ Res Public Health. 2012 Sep 19;9(9):3354-64.
38. J Recept Signal Transduct Res. 2011 Oct;31(5):374-80.
39. Am J Clin Dermatol. 2012 Dec 1;13(6):357-64.
40. Am J Clin Dermatol. 2011 Apr 1;12(2):133-41.
41. G Ital Dermatol Venereol. 2015 Feb;150(1):1-11.
Aesthetic dermatology: What do we know so far about Volbella?
On May 31, 2016, the Food and Drug Administration approved Juvéderm Volbella XC for use in the lips for lip augmentation and for correction of perioral rhytids, in adults over the age of 21. In the U.S. pivotal clinical trial of 168 patients, Juvederm Volbella XC was found to increase lip fullness and soften the appearance of perioral rhytids in two-thirds through 1 year.
Like its previously approved predecessor Juvéderm Voluma XC, approved for the mid-face/cheek area, Juvéderm Volbella XC is composed of hyaluronic acid (HA) using Vycross technology. Vycross technology blends different molecular weights of hyaluronic acid together, allowing for longer duration of the product. Unlike Juvéderm Voluma XC (20 mg/mL), Juvéderm Volbella XC is a lower-concentration HA (15 mg/mL) and has a lower G prime and cohesivity, allowing more horizontal spread as opposed to a lift, which makes it more appropriate for injection into the lips and superficial perioral lines.1
Juvéderm Volbella XC will not be available for use in the United States until October 2016. However, the product was first approved in Europe in 2011, and subsequently in Latin America, the Middle East, Asia Pacific, and Canada. What has been the experience of our neighboring colleagues?
In a randomized, prospective, 12-month controlled European study of 280 patients comparing Volbella to a nonanimal stabilized hyaluronic acid (NASHA), Volbella was found to be noninferior to NASHA at 3 months.2 Improvements in lip fullness, perioral lines, and oral commissures for Volbella were statistically significant at 6 and 12 months. Acute swelling was also noted to be less.
In a German 62-patient study with primary endpoints of satisfaction with improvement and look and feel of the lips, a high degree of subject satisfaction with aesthetic improvement in the lips, as well as with their natural look and feel, was noted among 83.6% and 75%-93%, respectively.3
In a retrospective chart review of 400 patients in Israel, where Volbella was injected into tear troughs (disclaimer: not approved for tear troughs in the United States) and/or lips, 17 patients (4.25%) developed prolonged inflammatory cutaneous reactions, lasting up to 11 months, which recurred (with an average number of 3.17 episodes) and occurred late (with an average onset of 8.41 weeks after the injection).4 The reactions were treated with antibiotics and hyaluronidase injections. The finding in this published report is higher than the 0.02% of delayed-onset inflammatory reaction previously reported with HA fillers.
With a full armamentarium of HA and non-HA fillers on the market, we are lucky to have many options to select from to treat aesthetic concerns of patients. For lip and perioral enhancement in the United States, Restylane, Juvéderm, Belotero, and now Volbella are all excellent options. As the dermal filler portfolio available internationally is larger than that in the United States, much can be learned from the experience of our international colleagues.
References
1. Plast Reconstr Surg. 2015 Nov;136(5 Suppl):139S-48S.
2. J Drugs Dermatol. 2015 Dec;14(12):1444-52.
3. J Cosmet Dermatol. 2014 Jun;13(2):125-34.
4. Dermatol Surg. 2016 Jan;42(1):31-7.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. Dr. Wesley has no disclosures with regard to Volbella; she participated in clinical trials for Juvéderm Voluma. Dr. Talakoub is a national trainer for Juvéderm manufacturer Allergan for all its injectables.
On May 31, 2016, the Food and Drug Administration approved Juvéderm Volbella XC for use in the lips for lip augmentation and for correction of perioral rhytids, in adults over the age of 21. In the U.S. pivotal clinical trial of 168 patients, Juvederm Volbella XC was found to increase lip fullness and soften the appearance of perioral rhytids in two-thirds through 1 year.
Like its previously approved predecessor Juvéderm Voluma XC, approved for the mid-face/cheek area, Juvéderm Volbella XC is composed of hyaluronic acid (HA) using Vycross technology. Vycross technology blends different molecular weights of hyaluronic acid together, allowing for longer duration of the product. Unlike Juvéderm Voluma XC (20 mg/mL), Juvéderm Volbella XC is a lower-concentration HA (15 mg/mL) and has a lower G prime and cohesivity, allowing more horizontal spread as opposed to a lift, which makes it more appropriate for injection into the lips and superficial perioral lines.1
Juvéderm Volbella XC will not be available for use in the United States until October 2016. However, the product was first approved in Europe in 2011, and subsequently in Latin America, the Middle East, Asia Pacific, and Canada. What has been the experience of our neighboring colleagues?
In a randomized, prospective, 12-month controlled European study of 280 patients comparing Volbella to a nonanimal stabilized hyaluronic acid (NASHA), Volbella was found to be noninferior to NASHA at 3 months.2 Improvements in lip fullness, perioral lines, and oral commissures for Volbella were statistically significant at 6 and 12 months. Acute swelling was also noted to be less.
In a German 62-patient study with primary endpoints of satisfaction with improvement and look and feel of the lips, a high degree of subject satisfaction with aesthetic improvement in the lips, as well as with their natural look and feel, was noted among 83.6% and 75%-93%, respectively.3
In a retrospective chart review of 400 patients in Israel, where Volbella was injected into tear troughs (disclaimer: not approved for tear troughs in the United States) and/or lips, 17 patients (4.25%) developed prolonged inflammatory cutaneous reactions, lasting up to 11 months, which recurred (with an average number of 3.17 episodes) and occurred late (with an average onset of 8.41 weeks after the injection).4 The reactions were treated with antibiotics and hyaluronidase injections. The finding in this published report is higher than the 0.02% of delayed-onset inflammatory reaction previously reported with HA fillers.
With a full armamentarium of HA and non-HA fillers on the market, we are lucky to have many options to select from to treat aesthetic concerns of patients. For lip and perioral enhancement in the United States, Restylane, Juvéderm, Belotero, and now Volbella are all excellent options. As the dermal filler portfolio available internationally is larger than that in the United States, much can be learned from the experience of our international colleagues.
References
1. Plast Reconstr Surg. 2015 Nov;136(5 Suppl):139S-48S.
2. J Drugs Dermatol. 2015 Dec;14(12):1444-52.
3. J Cosmet Dermatol. 2014 Jun;13(2):125-34.
4. Dermatol Surg. 2016 Jan;42(1):31-7.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. Dr. Wesley has no disclosures with regard to Volbella; she participated in clinical trials for Juvéderm Voluma. Dr. Talakoub is a national trainer for Juvéderm manufacturer Allergan for all its injectables.
On May 31, 2016, the Food and Drug Administration approved Juvéderm Volbella XC for use in the lips for lip augmentation and for correction of perioral rhytids, in adults over the age of 21. In the U.S. pivotal clinical trial of 168 patients, Juvederm Volbella XC was found to increase lip fullness and soften the appearance of perioral rhytids in two-thirds through 1 year.
Like its previously approved predecessor Juvéderm Voluma XC, approved for the mid-face/cheek area, Juvéderm Volbella XC is composed of hyaluronic acid (HA) using Vycross technology. Vycross technology blends different molecular weights of hyaluronic acid together, allowing for longer duration of the product. Unlike Juvéderm Voluma XC (20 mg/mL), Juvéderm Volbella XC is a lower-concentration HA (15 mg/mL) and has a lower G prime and cohesivity, allowing more horizontal spread as opposed to a lift, which makes it more appropriate for injection into the lips and superficial perioral lines.1
Juvéderm Volbella XC will not be available for use in the United States until October 2016. However, the product was first approved in Europe in 2011, and subsequently in Latin America, the Middle East, Asia Pacific, and Canada. What has been the experience of our neighboring colleagues?
In a randomized, prospective, 12-month controlled European study of 280 patients comparing Volbella to a nonanimal stabilized hyaluronic acid (NASHA), Volbella was found to be noninferior to NASHA at 3 months.2 Improvements in lip fullness, perioral lines, and oral commissures for Volbella were statistically significant at 6 and 12 months. Acute swelling was also noted to be less.
In a German 62-patient study with primary endpoints of satisfaction with improvement and look and feel of the lips, a high degree of subject satisfaction with aesthetic improvement in the lips, as well as with their natural look and feel, was noted among 83.6% and 75%-93%, respectively.3
In a retrospective chart review of 400 patients in Israel, where Volbella was injected into tear troughs (disclaimer: not approved for tear troughs in the United States) and/or lips, 17 patients (4.25%) developed prolonged inflammatory cutaneous reactions, lasting up to 11 months, which recurred (with an average number of 3.17 episodes) and occurred late (with an average onset of 8.41 weeks after the injection).4 The reactions were treated with antibiotics and hyaluronidase injections. The finding in this published report is higher than the 0.02% of delayed-onset inflammatory reaction previously reported with HA fillers.
With a full armamentarium of HA and non-HA fillers on the market, we are lucky to have many options to select from to treat aesthetic concerns of patients. For lip and perioral enhancement in the United States, Restylane, Juvéderm, Belotero, and now Volbella are all excellent options. As the dermal filler portfolio available internationally is larger than that in the United States, much can be learned from the experience of our international colleagues.
References
1. Plast Reconstr Surg. 2015 Nov;136(5 Suppl):139S-48S.
2. J Drugs Dermatol. 2015 Dec;14(12):1444-52.
3. J Cosmet Dermatol. 2014 Jun;13(2):125-34.
4. Dermatol Surg. 2016 Jan;42(1):31-7.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. Dr. Wesley has no disclosures with regard to Volbella; she participated in clinical trials for Juvéderm Voluma. Dr. Talakoub is a national trainer for Juvéderm manufacturer Allergan for all its injectables.
Cosmetic Corner: Dermatologists Weigh in on Nail Care Products
To improve patient care and outcomes, leading dermatologists offered their recommendations on nail care products. Consideration must be given to:
- Aquaphor Healing Ointment
Beiersdorf, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Biotin Oral Supplements
Manufacturers vary
“Biotin is a helpful supplement for brittle nails. It may take 6 months to see improvement in the nails.”—Shari Lipner, MD, PhD, New York, New York
Recommended by Gary Goldenberg, MD, New York, New York
- Deep Comfort Hand and Cuticle Cream
Clinique
“It has good hydration for cuticles with sodium hyaluronate and squalene. It also is fragrance free.”—Anthony M. Rossi, MD, New York, New York
- Genadur
Medimetriks Pharmaceuticals, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Lanolin-Rich Nail Conditioner
Elon
“It’s great for moisturizing and nail hardening.”—Marta Rendon, MD, Boca Raton, Florida
- Nail Renewal System
Dr. Dana
“Developed by dermatologist Dr. Dana Stern, the system combines glycolic acid to improve discoloration and ridging, along with hydrating and strengthening botanicals to improve the look, feel, and overall health of the nails.”— Joshua Zeichner, MD, New York, New York
Cutis invites readers to send us their recommendations. Acne scar treatments, self-tanners, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on nail care products. Consideration must be given to:
- Aquaphor Healing Ointment
Beiersdorf, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Biotin Oral Supplements
Manufacturers vary
“Biotin is a helpful supplement for brittle nails. It may take 6 months to see improvement in the nails.”—Shari Lipner, MD, PhD, New York, New York
Recommended by Gary Goldenberg, MD, New York, New York
- Deep Comfort Hand and Cuticle Cream
Clinique
“It has good hydration for cuticles with sodium hyaluronate and squalene. It also is fragrance free.”—Anthony M. Rossi, MD, New York, New York
- Genadur
Medimetriks Pharmaceuticals, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Lanolin-Rich Nail Conditioner
Elon
“It’s great for moisturizing and nail hardening.”—Marta Rendon, MD, Boca Raton, Florida
- Nail Renewal System
Dr. Dana
“Developed by dermatologist Dr. Dana Stern, the system combines glycolic acid to improve discoloration and ridging, along with hydrating and strengthening botanicals to improve the look, feel, and overall health of the nails.”— Joshua Zeichner, MD, New York, New York
Cutis invites readers to send us their recommendations. Acne scar treatments, self-tanners, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on nail care products. Consideration must be given to:
- Aquaphor Healing Ointment
Beiersdorf, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Biotin Oral Supplements
Manufacturers vary
“Biotin is a helpful supplement for brittle nails. It may take 6 months to see improvement in the nails.”—Shari Lipner, MD, PhD, New York, New York
Recommended by Gary Goldenberg, MD, New York, New York
- Deep Comfort Hand and Cuticle Cream
Clinique
“It has good hydration for cuticles with sodium hyaluronate and squalene. It also is fragrance free.”—Anthony M. Rossi, MD, New York, New York
- Genadur
Medimetriks Pharmaceuticals, Inc
Recommended by Gary Goldenberg, MD, New York, New York
- Lanolin-Rich Nail Conditioner
Elon
“It’s great for moisturizing and nail hardening.”—Marta Rendon, MD, Boca Raton, Florida
- Nail Renewal System
Dr. Dana
“Developed by dermatologist Dr. Dana Stern, the system combines glycolic acid to improve discoloration and ridging, along with hydrating and strengthening botanicals to improve the look, feel, and overall health of the nails.”— Joshua Zeichner, MD, New York, New York
Cutis invites readers to send us their recommendations. Acne scar treatments, self-tanners, and cleansing devices will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
An Update on Neurotoxin Products and Administration Methods
The first botulinum neurotoxin (BoNT) approved by the US Food and Drug Administration (FDA) was onabotulinumtoxinA in 1989 for the treatment of strabismus and blepharospasm. It was not until 1992, however, that the aesthetic benefits of BoNT were first reported in the medical literature by Carruthers and Carruthers,1 and a cosmetic indication was not approved by the FDA until 2002. Since that time, the popularity of BoNT products has grown rapidly with a nearly 6500% increase in popularity from 1997 to 2015 in addition to the introduction of a variety of new BoNT formulations to the market.2 It is estimated by the American Society for Aesthetic Plastic Surgery that there were at least 4,000,000 BoNT injections performed in 2015 alone, making it the most popular nonsurgical aesthetic procedure available.2 As the demand for minimally invasive cosmetic procedures continues to increase, we will continue to see the introduction of additional formulations of BoNT products as well as novel administration techniques and delivery devices. In this article, we provide an update on current and upcoming BoNT products and also review the literature on novel administration methods based on studies published from January 1, 2014, to December 31, 2015.
Current Products
To date, there are only 4 FDA-approved formulations of BoNT available for clinical use (eg, cervical dystonia, strabismus, blepharospasm, headache, urinary incontinence) in the United States: abobotulinumtoxinA, incobotulinumtoxinA, onabotulinumtoxinA, and rimabotulinumtoxinB.The FDA-approved dermatologic indications (eg, moderate to severe glabellar or canthal lines, severe axillary hyperhidrosis) for these products are provided in the Table. On a global scale, there are several other commonly utilized formulations of BoNT, including a Korean serotype resembling onabotulinumtoxinA and a Chinese botulinum toxin type A.3 Although there is some evidence to demonstrate comparable efficacy and safety of these latter products, the literature is relatively lacking in comparison to the FDA-approved products.4,5
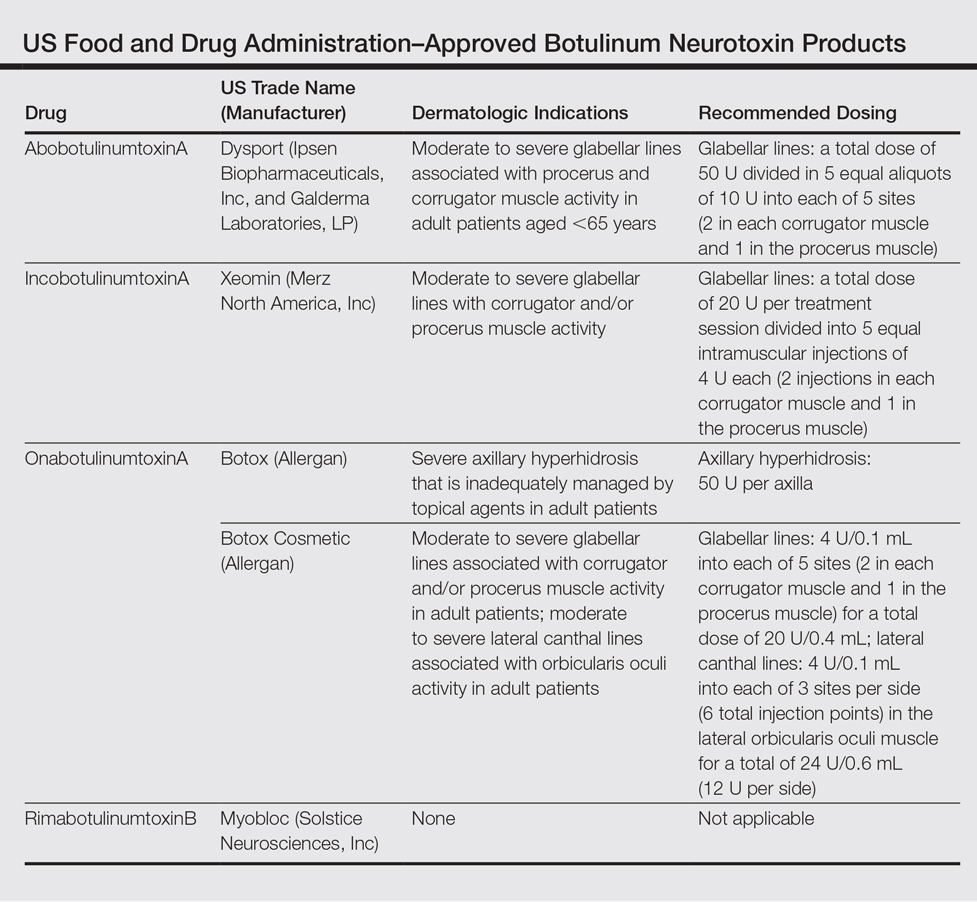
Upcoming Products
Currently, there are several new BoNT formulations being studied for clinical use. RT 002 (Revance Therapeutics, Inc) is a novel injectable formulation of onabotulinumtoxinA that consists of the purified neurotoxin in combination with patented TransMTS peptides that have been shown to provide high-binding avidity for the neurotoxin, and thus the product is designed to reduce diffusion to adjacent muscles and diminish unwanted effects. With a reduced level of neurotoxin dissemination, it is theorized that a higher administration of targeted doses can be injected, which may lead to a longer duration of desired effects.6 A clinical pilot study done to establish the safety and efficacy of RT 002 for treatment of moderate to severe glabellar lines evaluated 4 equally sized cohorts of 12 participants, each receiving single-dose administration of RT 002 ranging in potency equivalent to 25 U, 50 U, 75 U, and 100 U of abobotulinumtoxinA as determined by the gelatin phosphate method.6 It was concluded that RT 002 is both safe and efficacious with an extended duration of action, with a median duration of effect of 7 months observed in the highest dose group (dose equivalent to 100 U of abobotulinumtoxinA). Notably, 80% of all 48 participants maintained a minimum 1-point improvement in investigator-determined glabellar line severity scores at the 6-month time point and 60% achieved wrinkle scores of none or mild at 6 months posttreatment.6
DWP 450 (Daewoong Pharmaceutical Co, Ltd) is derived from the wild-type Clostridium botulinum and is reported to be of higher purity than standard onabotulinumtoxinA. An initial 16-week pilot study demonstrated that 20 U of DWP 450 is noninferior and of comparable efficacy and safety to 20 U of onabotulinumtoxinA in the treatment of glabellar lines.7
NTC (Botulax [Hugel, Inc]) is the name of the toxin derived from the C botulinum strain CBFC26, which has already been approved in many Asian, European, and Latin American countries for the treatment of blepharospasm. This formulation has demonstrated noninferiority to onabotulinumtoxinA at equivalent 20-U doses for the treatment of moderate to severe glabellar lines in a double-blind, randomized, multicenter, phase 3 trial of 272 participants with a 16-week follow-up.8
MT 10109L (Medytox Inc) is a unique product in that it is distributed as a liquid type A botulinum toxin rather than the standard freeze-dried formulation; thus, a major advantage of this product is its convenience, as it does not need reconstitution or dilution prior to administration. In a double-blind, randomized, active drug–controlled, phase 3 study of 168 participants, it was determined that MT 10109L (20 U) is comparable in efficacy to onabotulinumtoxinA (20 U) for the treatment of moderate to severe glabellar lines. No significant difference was seen between the 2 treatment groups when glabellar lines were assessed at rest at 4 and 16 weeks after treatment, but a significantly greater improvement in glabellar lines was seen at maximum frown in the MT 10109L group at the 16-week follow-up (P=.0064).9
Administration Techniques
With regard to safe and effective BoNT product administration techniques, a variety of studies have provided insight into optimal practice methods. A 2015 expert consensus statement formed by an American Society for Dermatologic Surgery task force reviewed data from 42 papers and unanimously determined that for all current type A BoNT products available in the United States, a vial of BoNT reconstituted appropriately for the purpose of facial injections can be reconstituted at least 4 weeks prior to administration without contamination risk or decrease in efficacy and that multiple patients can be treated with the same vial.Although the statement was not explicit on whether or not preserved or unpreserved saline is to be used, it is considered routine practice to use preservative-containing saline to reconstitute BoNT, as it has been shown to reduce patient discomfort and is not associated with adverse effects.10
Pain Minimization
With respect to minimizing the pain associated with BoNT injections, several studies have assessed administration techniques to minimize patient discomfort. A split-face, double-blind study of 20 participants demonstrated that the use of a 32-gauge needle has a significantly greater chance of reducing clinically significant levels of pain as compared to a 30-gauge needle when performing facial injections (P=.04). Overall, however, injections of the face and arms were on average only nominally and not significantly more painful with 30-gauge needles compared to 32-gauge needles.11
Another technique that has been found to reduce patient discomfort is the application of cold packs prior to injection. A study of patients with chronic facial palsy observed a significant reduction in pain with the administration of a cold (3°C–5°C) gel pack for 1 minute compared to a room temperature (20°C) gel pack prior to the administration of onabotulinumtoxinA into the platysma (P<.001).12 In the matter of injection with rimabotulinumtoxinB, which has been shown to be considerably more painful to receive than its more popularly administered counterpart onabotulinumtoxinA, a split-face pilot study examined the effect of increasing the pH of rimabotulinumtoxinB to 7.5 with sodium bicarbonate from the usual pH of 5.6.13,14 Pain was reported to be considerably less in the higher pH group and no reduction of efficacy was seen over the 10-week follow-up period.14
Delivery Methods
Several preliminary studies also have examined novel delivery techniques to identify minimally painful yet effective methods for administering BoNT. It has been reported that standard BoNT formulations are not effective as topical agents in a comparison study in which onabotulinumtoxinA injection was compared to topically applied onabotulinumtoxinA.15 However, a follow-up prospective study by the same authors has demonstrated efficacy of topical onabotulinumtoxinA following pretreatment with a fractional ablative CO2 laser for treatment of crow’s-feet. In this randomized, split-face, controlled trial (N=10), participants were first pretreated with topical lidocaine 30% before receiving a single pass of fractional ablative CO2 laser with no overlap and a pulse energy of 100 mJ. Within 60 seconds of laser treatment, participants then received either 100 U of abobotulinumtoxinA diluted in 0.1 mL of saline or simple normal saline applied topically. A clinically significant improvement in periorbital wrinkles was seen both at 1-week and 1-month posttreatment in the laser and onabotulinumtoxinA–treated group compared to the laser and topical saline–treated group (P<.02).15
Another unique administration method studied, albeit with less successful results, involves the use of iontophoresis to deliver BoNT painlessly in a transdermal fashion with the assistance of an electrical current.16 This prospective, randomized, assessor-blinded, split-axilla, controlled trial of 11 participants compared the effectiveness of administering onabotulinumtoxinA via iontophoresis to traditional injection with onabotulinumtoxinA (250 U). Iontophoresis was accomplished with a single electrode pad soaked with 250 U of onabotulinumtoxinA applied directly to the axilla and a second electrode pad soaked in 0.9% saline applied to the hand to complete the circuit. An alternating electrical current was slowly increased for 30 minutes to a maximum current of 15 mA with a voltage of 12 V. Among the 11 participants recruited, the side treated with traditional injection showed a significantly greater percentage reduction in baseline sweating at the 1-week, 1-month, and 6-month posttreatment evaluations compared to iontophoresis (84%, 76%, and 50%, respectively vs 73%, 22%, and 32%, respectively)(P<.05). Despite being less efficacious than standard injection therapy, participants reported that iontophoresis delivery was significantly less painful (P<.05).16
A high-pressure oxygen delivery device, which utilizes a powerful jet of microdroplets containing water, the drug, air, and oxygen to deliver medication onto the skin surface, also has been studied as a means of delivery of BoNT in a minimally painful manner. In this study, the device was used to assess the efficacy of transdermal delivery of BoNT via jet nebulization in the treatment of primary palmar, plantar, and axillary hyperhidrosis.17 The 20 participants included in the study were randomized to receive either a combination of lidocaine and onabotulinumtoxinA (50 U) administered through the device or lidocaine delivered through the device followed by multiple transcutaneous injections of onabotulinumtoxinA (100 U). Both treatments significantly reduced sweating compared to baseline as measured by a visual analogue scale at 3-month follow-up (P<.001), but the combination delivery of lidocaine and onabotulinumtoxinA via the device resulted in significantly less procedure-related pain and sweating (P<.001) as well as significantly greater patient satisfaction (P<.001).17
Optimizing Aesthetic Outcomes
A frequent concern of patients receiving BoNT for cosmetic purposes is a desire to avoid a “frozen” or expressionless look. As such, many clinicians have attempted a variety of techniques to achieve more natural aesthetic results. One such method is known as the multipoint and multilevel injection technique, which consists of utilizing multiple injection sites at varying depths (intramuscular, subcutaneous, or intradermal) and doses (2–6 U) depending on the degree of contractility of the targeted muscle. In a preliminary study of 223 participants using this technique with a total dose of 125 U of abobotulinumtoxinA, good and natural results were reported with perseveration of facial emotion in all participants in addition to a mean overall satisfaction rate of 6.4 of 7 on the Facial Line Treatment Satisfaction Questionnaire with the maximum satisfaction rating possible reported in 66% of cases.18 It also has been postulated that injection depth of BoNT can affect brow elevation whereupon deeper injection depths can result in inactivation of the brow depressors and allow for increased elevation of the eyebrows. This technique has been applied in attempts to correct brow height asymmetry. However, a prospective, split-face study of 23 women suggested that this method is not effective.19 Participants received 64 U of onabotulinumtoxinA via 16 injection sites in the glabella, forehead, and lateral canthal area with either all deep or all shallow injections depending on the side treated and whether brow-lift was desired. Results at 4 weeks posttreatment showed no significant difference in brow height, and it was concluded that eyebrow depressor muscles cannot be accurately targeted with deep injection into the muscle belly for correction of eyebrow height discrepancies.19 Conversely, a 5-year retrospective, nonrandomized study of 227 patients with 563 treatments utilizing a “microdroplet” technique reported success at selectively targeting the eyebrow depressors while leaving the brow elevators unaffected.20 Here, a total dose of 33 U of onabotulinumtoxinA was administered via microdroplets of 10 to 20 μL, each with more than 60 to 100 injections into the brow, glabella, and crow’s-feet area. This method of injection resulted in a statistically significant improvement of forehead lines and brow ptosis and furrowing at follow-up between 10 and 45 days after treatment (P<.0001). Additionally, average brow height was significantly increased from 24.6 mm to 25 mm after treatment (P=.02).20
Conclusion
The use of BoNT products for both on- and off-label cosmetic and medical indications has rapidly grown over the past 2 decades. As demonstrated in this review, a variety of promising new products and delivery techniques are being developed. Given the rise in popularity of BoNT products among both physicians and consumers, clinicians should be aware of the current data and ongoing research.
- Carruthers JD, Carruthers JA. Treatment of glabellar frown lines with C. botulinum-A exotoxin. J Dermatol Surg Oncol. 1992;18:17-21.
- American Society for Aesthetic Plastic Surgery. Cosmetic Surgery National Data Bank statistics. American Society for Aesthetic Plastic Surgery website. http://www.surgery.org/sites/default/files/ASAPS-Stats2015.pdf. Accessed June 12, 2016.
- Walker TJ, Dayan SH. Comparison and overview of currently available neurotoxins. J Clin Aesthet Dermatol. 2014;7:31-39.
- Feng Z, Sun Q, He L, et al. Optimal dosage of botulinum toxin type A for treatment of glabellar frown lines: efficacy and safety in a clinical trial. Dermatol Surg. 2015;41(suppl 1):S56-S63.
- Jiang HY, Chen S, Zhou J, et al. Diffusion of two botulinum toxins type A on the forehead: double-blinded, randomized, controlled study. Dermatol Surg. 2014;40:184-192.
- Garcia-Murray E, Velasco Villasenor ML, Acevedo B, et al. Safety and efficacy of RT002, an injectable botulinum toxin type A, for treating glabellar lines: results of a phase 1/2, open-label, sequential dose-escalation study. Dermatol Surg. 2015;41(suppl 1):S47-S55.
- Won CH, Kim HK, Kim BJ, et al. Comparative trial of a novel botulinum neurotoxin type A versus onabotulinumtoxinA in the treatment of glabellar lines: a multicenter, randomized, double-blind, active-controlled study. Int J Dermatol. 2015;54:227-234.
- Kim BJ, Kwon HH, Park SY, et al. Double-blind, randomized non-inferiority trial of a novel botulinum toxin A processed from the strain CBFC26, compared with onabotulinumtoxin A in the treatment of glabellar lines. J Eur Acad Dermatol Venereol. 2014;28:1761-1767.
- Kim JE, Song EJ, Choi GS, et al. The efficacy and safety of liquid-type botulinum toxin type A for the management of moderate to severe glabellar frown lines. Plast Reconstr Surg. 2015;135:732-741.
- Alam M, Bolotin D, Carruthers J, et al. Consensus statement regarding storage and reuse of previously reconstituted neuromodulators. Dermatol Surg. 2015;41:321-326.
- Alam M, Geisler A, Sadhwani D, et al. Effect of needle size on pain perception in patients treated with botulinum toxin type A injections: a randomized clinical trial. JAMA Dermatol. 2015;151:1194-1199.
- Pucks N, Thomas A, Hallam MJ, et al. Cutaneous cooling to manage botulinum toxin injection-associated pain in patients with facial palsy: a randomised controlled trial. J Plast Reconstr Aesthet Surg. 2015;68:1701-1705.
- Kranz G, Sycha T, Voller B, et al. Pain sensation during intradermal injections of three different botulinum toxin preparations in different doses and dilutions. Dermatol Surg. 2006;32:886-890.
- Lowe PL, Lowe NJ. Botulinum toxin type B: pH change reduces injection pain, retains efficacy. Dermatol Surg. 2014;40:1328-1333.
- Mahmoud BH, Burnett C, Ozog D. Prospective randomized controlled study to determine the effect of topical application of botulinum toxin A for crow’s feet after treatment with ablative fractional CO2 laser. Dermatol Surg. 2015;41(suppl 1):S75-S81.
- Montaser-Kouhsari L, Zartab H, Fanian F, et al. Comparison of intradermal injection with iontophoresis of abo-botulinum toxin A for the treatment of primary axillary hyperhidrosis: a randomized, controlled trial. J Dermatolog Treat. 2014;25:337-341.
- Iannitti T, Palmieri B, Aspiro A, et al. A preliminary study of painless and effective transdermal botulinum toxin A delivery by jet nebulization for treatment of primary hyperhidrosis. Drug Des Devel Ther. 2014;8:931-935.
- Iozzo I, Tengattini V, Antonucci VA. Multipoint and multilevel injection technique of botulinum toxin A in facial aesthetics. J Cosmet Dermatol. 2014;13:135-142.
- Sneath J, Humphrey S, Carruthers A, et al. Injecting botulinum toxin at different depths is not effective for the correction of eyebrow asymmetry. Dermatol Surg. 2015;41(suppl 1):S82-S87.
- Steinsapir KD, Rootman D, Wulc A, et al. Cosmetic microdroplet botulinum toxin A forehead lift: a new treatment paradigm. Ophthal Plast Reconstr Surg. 2015;31:263-268.
The first botulinum neurotoxin (BoNT) approved by the US Food and Drug Administration (FDA) was onabotulinumtoxinA in 1989 for the treatment of strabismus and blepharospasm. It was not until 1992, however, that the aesthetic benefits of BoNT were first reported in the medical literature by Carruthers and Carruthers,1 and a cosmetic indication was not approved by the FDA until 2002. Since that time, the popularity of BoNT products has grown rapidly with a nearly 6500% increase in popularity from 1997 to 2015 in addition to the introduction of a variety of new BoNT formulations to the market.2 It is estimated by the American Society for Aesthetic Plastic Surgery that there were at least 4,000,000 BoNT injections performed in 2015 alone, making it the most popular nonsurgical aesthetic procedure available.2 As the demand for minimally invasive cosmetic procedures continues to increase, we will continue to see the introduction of additional formulations of BoNT products as well as novel administration techniques and delivery devices. In this article, we provide an update on current and upcoming BoNT products and also review the literature on novel administration methods based on studies published from January 1, 2014, to December 31, 2015.
Current Products
To date, there are only 4 FDA-approved formulations of BoNT available for clinical use (eg, cervical dystonia, strabismus, blepharospasm, headache, urinary incontinence) in the United States: abobotulinumtoxinA, incobotulinumtoxinA, onabotulinumtoxinA, and rimabotulinumtoxinB.The FDA-approved dermatologic indications (eg, moderate to severe glabellar or canthal lines, severe axillary hyperhidrosis) for these products are provided in the Table. On a global scale, there are several other commonly utilized formulations of BoNT, including a Korean serotype resembling onabotulinumtoxinA and a Chinese botulinum toxin type A.3 Although there is some evidence to demonstrate comparable efficacy and safety of these latter products, the literature is relatively lacking in comparison to the FDA-approved products.4,5

Upcoming Products
Currently, there are several new BoNT formulations being studied for clinical use. RT 002 (Revance Therapeutics, Inc) is a novel injectable formulation of onabotulinumtoxinA that consists of the purified neurotoxin in combination with patented TransMTS peptides that have been shown to provide high-binding avidity for the neurotoxin, and thus the product is designed to reduce diffusion to adjacent muscles and diminish unwanted effects. With a reduced level of neurotoxin dissemination, it is theorized that a higher administration of targeted doses can be injected, which may lead to a longer duration of desired effects.6 A clinical pilot study done to establish the safety and efficacy of RT 002 for treatment of moderate to severe glabellar lines evaluated 4 equally sized cohorts of 12 participants, each receiving single-dose administration of RT 002 ranging in potency equivalent to 25 U, 50 U, 75 U, and 100 U of abobotulinumtoxinA as determined by the gelatin phosphate method.6 It was concluded that RT 002 is both safe and efficacious with an extended duration of action, with a median duration of effect of 7 months observed in the highest dose group (dose equivalent to 100 U of abobotulinumtoxinA). Notably, 80% of all 48 participants maintained a minimum 1-point improvement in investigator-determined glabellar line severity scores at the 6-month time point and 60% achieved wrinkle scores of none or mild at 6 months posttreatment.6
DWP 450 (Daewoong Pharmaceutical Co, Ltd) is derived from the wild-type Clostridium botulinum and is reported to be of higher purity than standard onabotulinumtoxinA. An initial 16-week pilot study demonstrated that 20 U of DWP 450 is noninferior and of comparable efficacy and safety to 20 U of onabotulinumtoxinA in the treatment of glabellar lines.7
NTC (Botulax [Hugel, Inc]) is the name of the toxin derived from the C botulinum strain CBFC26, which has already been approved in many Asian, European, and Latin American countries for the treatment of blepharospasm. This formulation has demonstrated noninferiority to onabotulinumtoxinA at equivalent 20-U doses for the treatment of moderate to severe glabellar lines in a double-blind, randomized, multicenter, phase 3 trial of 272 participants with a 16-week follow-up.8
MT 10109L (Medytox Inc) is a unique product in that it is distributed as a liquid type A botulinum toxin rather than the standard freeze-dried formulation; thus, a major advantage of this product is its convenience, as it does not need reconstitution or dilution prior to administration. In a double-blind, randomized, active drug–controlled, phase 3 study of 168 participants, it was determined that MT 10109L (20 U) is comparable in efficacy to onabotulinumtoxinA (20 U) for the treatment of moderate to severe glabellar lines. No significant difference was seen between the 2 treatment groups when glabellar lines were assessed at rest at 4 and 16 weeks after treatment, but a significantly greater improvement in glabellar lines was seen at maximum frown in the MT 10109L group at the 16-week follow-up (P=.0064).9
Administration Techniques
With regard to safe and effective BoNT product administration techniques, a variety of studies have provided insight into optimal practice methods. A 2015 expert consensus statement formed by an American Society for Dermatologic Surgery task force reviewed data from 42 papers and unanimously determined that for all current type A BoNT products available in the United States, a vial of BoNT reconstituted appropriately for the purpose of facial injections can be reconstituted at least 4 weeks prior to administration without contamination risk or decrease in efficacy and that multiple patients can be treated with the same vial.Although the statement was not explicit on whether or not preserved or unpreserved saline is to be used, it is considered routine practice to use preservative-containing saline to reconstitute BoNT, as it has been shown to reduce patient discomfort and is not associated with adverse effects.10
Pain Minimization
With respect to minimizing the pain associated with BoNT injections, several studies have assessed administration techniques to minimize patient discomfort. A split-face, double-blind study of 20 participants demonstrated that the use of a 32-gauge needle has a significantly greater chance of reducing clinically significant levels of pain as compared to a 30-gauge needle when performing facial injections (P=.04). Overall, however, injections of the face and arms were on average only nominally and not significantly more painful with 30-gauge needles compared to 32-gauge needles.11
Another technique that has been found to reduce patient discomfort is the application of cold packs prior to injection. A study of patients with chronic facial palsy observed a significant reduction in pain with the administration of a cold (3°C–5°C) gel pack for 1 minute compared to a room temperature (20°C) gel pack prior to the administration of onabotulinumtoxinA into the platysma (P<.001).12 In the matter of injection with rimabotulinumtoxinB, which has been shown to be considerably more painful to receive than its more popularly administered counterpart onabotulinumtoxinA, a split-face pilot study examined the effect of increasing the pH of rimabotulinumtoxinB to 7.5 with sodium bicarbonate from the usual pH of 5.6.13,14 Pain was reported to be considerably less in the higher pH group and no reduction of efficacy was seen over the 10-week follow-up period.14
Delivery Methods
Several preliminary studies also have examined novel delivery techniques to identify minimally painful yet effective methods for administering BoNT. It has been reported that standard BoNT formulations are not effective as topical agents in a comparison study in which onabotulinumtoxinA injection was compared to topically applied onabotulinumtoxinA.15 However, a follow-up prospective study by the same authors has demonstrated efficacy of topical onabotulinumtoxinA following pretreatment with a fractional ablative CO2 laser for treatment of crow’s-feet. In this randomized, split-face, controlled trial (N=10), participants were first pretreated with topical lidocaine 30% before receiving a single pass of fractional ablative CO2 laser with no overlap and a pulse energy of 100 mJ. Within 60 seconds of laser treatment, participants then received either 100 U of abobotulinumtoxinA diluted in 0.1 mL of saline or simple normal saline applied topically. A clinically significant improvement in periorbital wrinkles was seen both at 1-week and 1-month posttreatment in the laser and onabotulinumtoxinA–treated group compared to the laser and topical saline–treated group (P<.02).15
Another unique administration method studied, albeit with less successful results, involves the use of iontophoresis to deliver BoNT painlessly in a transdermal fashion with the assistance of an electrical current.16 This prospective, randomized, assessor-blinded, split-axilla, controlled trial of 11 participants compared the effectiveness of administering onabotulinumtoxinA via iontophoresis to traditional injection with onabotulinumtoxinA (250 U). Iontophoresis was accomplished with a single electrode pad soaked with 250 U of onabotulinumtoxinA applied directly to the axilla and a second electrode pad soaked in 0.9% saline applied to the hand to complete the circuit. An alternating electrical current was slowly increased for 30 minutes to a maximum current of 15 mA with a voltage of 12 V. Among the 11 participants recruited, the side treated with traditional injection showed a significantly greater percentage reduction in baseline sweating at the 1-week, 1-month, and 6-month posttreatment evaluations compared to iontophoresis (84%, 76%, and 50%, respectively vs 73%, 22%, and 32%, respectively)(P<.05). Despite being less efficacious than standard injection therapy, participants reported that iontophoresis delivery was significantly less painful (P<.05).16
A high-pressure oxygen delivery device, which utilizes a powerful jet of microdroplets containing water, the drug, air, and oxygen to deliver medication onto the skin surface, also has been studied as a means of delivery of BoNT in a minimally painful manner. In this study, the device was used to assess the efficacy of transdermal delivery of BoNT via jet nebulization in the treatment of primary palmar, plantar, and axillary hyperhidrosis.17 The 20 participants included in the study were randomized to receive either a combination of lidocaine and onabotulinumtoxinA (50 U) administered through the device or lidocaine delivered through the device followed by multiple transcutaneous injections of onabotulinumtoxinA (100 U). Both treatments significantly reduced sweating compared to baseline as measured by a visual analogue scale at 3-month follow-up (P<.001), but the combination delivery of lidocaine and onabotulinumtoxinA via the device resulted in significantly less procedure-related pain and sweating (P<.001) as well as significantly greater patient satisfaction (P<.001).17
Optimizing Aesthetic Outcomes
A frequent concern of patients receiving BoNT for cosmetic purposes is a desire to avoid a “frozen” or expressionless look. As such, many clinicians have attempted a variety of techniques to achieve more natural aesthetic results. One such method is known as the multipoint and multilevel injection technique, which consists of utilizing multiple injection sites at varying depths (intramuscular, subcutaneous, or intradermal) and doses (2–6 U) depending on the degree of contractility of the targeted muscle. In a preliminary study of 223 participants using this technique with a total dose of 125 U of abobotulinumtoxinA, good and natural results were reported with perseveration of facial emotion in all participants in addition to a mean overall satisfaction rate of 6.4 of 7 on the Facial Line Treatment Satisfaction Questionnaire with the maximum satisfaction rating possible reported in 66% of cases.18 It also has been postulated that injection depth of BoNT can affect brow elevation whereupon deeper injection depths can result in inactivation of the brow depressors and allow for increased elevation of the eyebrows. This technique has been applied in attempts to correct brow height asymmetry. However, a prospective, split-face study of 23 women suggested that this method is not effective.19 Participants received 64 U of onabotulinumtoxinA via 16 injection sites in the glabella, forehead, and lateral canthal area with either all deep or all shallow injections depending on the side treated and whether brow-lift was desired. Results at 4 weeks posttreatment showed no significant difference in brow height, and it was concluded that eyebrow depressor muscles cannot be accurately targeted with deep injection into the muscle belly for correction of eyebrow height discrepancies.19 Conversely, a 5-year retrospective, nonrandomized study of 227 patients with 563 treatments utilizing a “microdroplet” technique reported success at selectively targeting the eyebrow depressors while leaving the brow elevators unaffected.20 Here, a total dose of 33 U of onabotulinumtoxinA was administered via microdroplets of 10 to 20 μL, each with more than 60 to 100 injections into the brow, glabella, and crow’s-feet area. This method of injection resulted in a statistically significant improvement of forehead lines and brow ptosis and furrowing at follow-up between 10 and 45 days after treatment (P<.0001). Additionally, average brow height was significantly increased from 24.6 mm to 25 mm after treatment (P=.02).20
Conclusion
The use of BoNT products for both on- and off-label cosmetic and medical indications has rapidly grown over the past 2 decades. As demonstrated in this review, a variety of promising new products and delivery techniques are being developed. Given the rise in popularity of BoNT products among both physicians and consumers, clinicians should be aware of the current data and ongoing research.
The first botulinum neurotoxin (BoNT) approved by the US Food and Drug Administration (FDA) was onabotulinumtoxinA in 1989 for the treatment of strabismus and blepharospasm. It was not until 1992, however, that the aesthetic benefits of BoNT were first reported in the medical literature by Carruthers and Carruthers,1 and a cosmetic indication was not approved by the FDA until 2002. Since that time, the popularity of BoNT products has grown rapidly with a nearly 6500% increase in popularity from 1997 to 2015 in addition to the introduction of a variety of new BoNT formulations to the market.2 It is estimated by the American Society for Aesthetic Plastic Surgery that there were at least 4,000,000 BoNT injections performed in 2015 alone, making it the most popular nonsurgical aesthetic procedure available.2 As the demand for minimally invasive cosmetic procedures continues to increase, we will continue to see the introduction of additional formulations of BoNT products as well as novel administration techniques and delivery devices. In this article, we provide an update on current and upcoming BoNT products and also review the literature on novel administration methods based on studies published from January 1, 2014, to December 31, 2015.
Current Products
To date, there are only 4 FDA-approved formulations of BoNT available for clinical use (eg, cervical dystonia, strabismus, blepharospasm, headache, urinary incontinence) in the United States: abobotulinumtoxinA, incobotulinumtoxinA, onabotulinumtoxinA, and rimabotulinumtoxinB.The FDA-approved dermatologic indications (eg, moderate to severe glabellar or canthal lines, severe axillary hyperhidrosis) for these products are provided in the Table. On a global scale, there are several other commonly utilized formulations of BoNT, including a Korean serotype resembling onabotulinumtoxinA and a Chinese botulinum toxin type A.3 Although there is some evidence to demonstrate comparable efficacy and safety of these latter products, the literature is relatively lacking in comparison to the FDA-approved products.4,5

Upcoming Products
Currently, there are several new BoNT formulations being studied for clinical use. RT 002 (Revance Therapeutics, Inc) is a novel injectable formulation of onabotulinumtoxinA that consists of the purified neurotoxin in combination with patented TransMTS peptides that have been shown to provide high-binding avidity for the neurotoxin, and thus the product is designed to reduce diffusion to adjacent muscles and diminish unwanted effects. With a reduced level of neurotoxin dissemination, it is theorized that a higher administration of targeted doses can be injected, which may lead to a longer duration of desired effects.6 A clinical pilot study done to establish the safety and efficacy of RT 002 for treatment of moderate to severe glabellar lines evaluated 4 equally sized cohorts of 12 participants, each receiving single-dose administration of RT 002 ranging in potency equivalent to 25 U, 50 U, 75 U, and 100 U of abobotulinumtoxinA as determined by the gelatin phosphate method.6 It was concluded that RT 002 is both safe and efficacious with an extended duration of action, with a median duration of effect of 7 months observed in the highest dose group (dose equivalent to 100 U of abobotulinumtoxinA). Notably, 80% of all 48 participants maintained a minimum 1-point improvement in investigator-determined glabellar line severity scores at the 6-month time point and 60% achieved wrinkle scores of none or mild at 6 months posttreatment.6
DWP 450 (Daewoong Pharmaceutical Co, Ltd) is derived from the wild-type Clostridium botulinum and is reported to be of higher purity than standard onabotulinumtoxinA. An initial 16-week pilot study demonstrated that 20 U of DWP 450 is noninferior and of comparable efficacy and safety to 20 U of onabotulinumtoxinA in the treatment of glabellar lines.7
NTC (Botulax [Hugel, Inc]) is the name of the toxin derived from the C botulinum strain CBFC26, which has already been approved in many Asian, European, and Latin American countries for the treatment of blepharospasm. This formulation has demonstrated noninferiority to onabotulinumtoxinA at equivalent 20-U doses for the treatment of moderate to severe glabellar lines in a double-blind, randomized, multicenter, phase 3 trial of 272 participants with a 16-week follow-up.8
MT 10109L (Medytox Inc) is a unique product in that it is distributed as a liquid type A botulinum toxin rather than the standard freeze-dried formulation; thus, a major advantage of this product is its convenience, as it does not need reconstitution or dilution prior to administration. In a double-blind, randomized, active drug–controlled, phase 3 study of 168 participants, it was determined that MT 10109L (20 U) is comparable in efficacy to onabotulinumtoxinA (20 U) for the treatment of moderate to severe glabellar lines. No significant difference was seen between the 2 treatment groups when glabellar lines were assessed at rest at 4 and 16 weeks after treatment, but a significantly greater improvement in glabellar lines was seen at maximum frown in the MT 10109L group at the 16-week follow-up (P=.0064).9
Administration Techniques
With regard to safe and effective BoNT product administration techniques, a variety of studies have provided insight into optimal practice methods. A 2015 expert consensus statement formed by an American Society for Dermatologic Surgery task force reviewed data from 42 papers and unanimously determined that for all current type A BoNT products available in the United States, a vial of BoNT reconstituted appropriately for the purpose of facial injections can be reconstituted at least 4 weeks prior to administration without contamination risk or decrease in efficacy and that multiple patients can be treated with the same vial.Although the statement was not explicit on whether or not preserved or unpreserved saline is to be used, it is considered routine practice to use preservative-containing saline to reconstitute BoNT, as it has been shown to reduce patient discomfort and is not associated with adverse effects.10
Pain Minimization
With respect to minimizing the pain associated with BoNT injections, several studies have assessed administration techniques to minimize patient discomfort. A split-face, double-blind study of 20 participants demonstrated that the use of a 32-gauge needle has a significantly greater chance of reducing clinically significant levels of pain as compared to a 30-gauge needle when performing facial injections (P=.04). Overall, however, injections of the face and arms were on average only nominally and not significantly more painful with 30-gauge needles compared to 32-gauge needles.11
Another technique that has been found to reduce patient discomfort is the application of cold packs prior to injection. A study of patients with chronic facial palsy observed a significant reduction in pain with the administration of a cold (3°C–5°C) gel pack for 1 minute compared to a room temperature (20°C) gel pack prior to the administration of onabotulinumtoxinA into the platysma (P<.001).12 In the matter of injection with rimabotulinumtoxinB, which has been shown to be considerably more painful to receive than its more popularly administered counterpart onabotulinumtoxinA, a split-face pilot study examined the effect of increasing the pH of rimabotulinumtoxinB to 7.5 with sodium bicarbonate from the usual pH of 5.6.13,14 Pain was reported to be considerably less in the higher pH group and no reduction of efficacy was seen over the 10-week follow-up period.14
Delivery Methods
Several preliminary studies also have examined novel delivery techniques to identify minimally painful yet effective methods for administering BoNT. It has been reported that standard BoNT formulations are not effective as topical agents in a comparison study in which onabotulinumtoxinA injection was compared to topically applied onabotulinumtoxinA.15 However, a follow-up prospective study by the same authors has demonstrated efficacy of topical onabotulinumtoxinA following pretreatment with a fractional ablative CO2 laser for treatment of crow’s-feet. In this randomized, split-face, controlled trial (N=10), participants were first pretreated with topical lidocaine 30% before receiving a single pass of fractional ablative CO2 laser with no overlap and a pulse energy of 100 mJ. Within 60 seconds of laser treatment, participants then received either 100 U of abobotulinumtoxinA diluted in 0.1 mL of saline or simple normal saline applied topically. A clinically significant improvement in periorbital wrinkles was seen both at 1-week and 1-month posttreatment in the laser and onabotulinumtoxinA–treated group compared to the laser and topical saline–treated group (P<.02).15
Another unique administration method studied, albeit with less successful results, involves the use of iontophoresis to deliver BoNT painlessly in a transdermal fashion with the assistance of an electrical current.16 This prospective, randomized, assessor-blinded, split-axilla, controlled trial of 11 participants compared the effectiveness of administering onabotulinumtoxinA via iontophoresis to traditional injection with onabotulinumtoxinA (250 U). Iontophoresis was accomplished with a single electrode pad soaked with 250 U of onabotulinumtoxinA applied directly to the axilla and a second electrode pad soaked in 0.9% saline applied to the hand to complete the circuit. An alternating electrical current was slowly increased for 30 minutes to a maximum current of 15 mA with a voltage of 12 V. Among the 11 participants recruited, the side treated with traditional injection showed a significantly greater percentage reduction in baseline sweating at the 1-week, 1-month, and 6-month posttreatment evaluations compared to iontophoresis (84%, 76%, and 50%, respectively vs 73%, 22%, and 32%, respectively)(P<.05). Despite being less efficacious than standard injection therapy, participants reported that iontophoresis delivery was significantly less painful (P<.05).16
A high-pressure oxygen delivery device, which utilizes a powerful jet of microdroplets containing water, the drug, air, and oxygen to deliver medication onto the skin surface, also has been studied as a means of delivery of BoNT in a minimally painful manner. In this study, the device was used to assess the efficacy of transdermal delivery of BoNT via jet nebulization in the treatment of primary palmar, plantar, and axillary hyperhidrosis.17 The 20 participants included in the study were randomized to receive either a combination of lidocaine and onabotulinumtoxinA (50 U) administered through the device or lidocaine delivered through the device followed by multiple transcutaneous injections of onabotulinumtoxinA (100 U). Both treatments significantly reduced sweating compared to baseline as measured by a visual analogue scale at 3-month follow-up (P<.001), but the combination delivery of lidocaine and onabotulinumtoxinA via the device resulted in significantly less procedure-related pain and sweating (P<.001) as well as significantly greater patient satisfaction (P<.001).17
Optimizing Aesthetic Outcomes
A frequent concern of patients receiving BoNT for cosmetic purposes is a desire to avoid a “frozen” or expressionless look. As such, many clinicians have attempted a variety of techniques to achieve more natural aesthetic results. One such method is known as the multipoint and multilevel injection technique, which consists of utilizing multiple injection sites at varying depths (intramuscular, subcutaneous, or intradermal) and doses (2–6 U) depending on the degree of contractility of the targeted muscle. In a preliminary study of 223 participants using this technique with a total dose of 125 U of abobotulinumtoxinA, good and natural results were reported with perseveration of facial emotion in all participants in addition to a mean overall satisfaction rate of 6.4 of 7 on the Facial Line Treatment Satisfaction Questionnaire with the maximum satisfaction rating possible reported in 66% of cases.18 It also has been postulated that injection depth of BoNT can affect brow elevation whereupon deeper injection depths can result in inactivation of the brow depressors and allow for increased elevation of the eyebrows. This technique has been applied in attempts to correct brow height asymmetry. However, a prospective, split-face study of 23 women suggested that this method is not effective.19 Participants received 64 U of onabotulinumtoxinA via 16 injection sites in the glabella, forehead, and lateral canthal area with either all deep or all shallow injections depending on the side treated and whether brow-lift was desired. Results at 4 weeks posttreatment showed no significant difference in brow height, and it was concluded that eyebrow depressor muscles cannot be accurately targeted with deep injection into the muscle belly for correction of eyebrow height discrepancies.19 Conversely, a 5-year retrospective, nonrandomized study of 227 patients with 563 treatments utilizing a “microdroplet” technique reported success at selectively targeting the eyebrow depressors while leaving the brow elevators unaffected.20 Here, a total dose of 33 U of onabotulinumtoxinA was administered via microdroplets of 10 to 20 μL, each with more than 60 to 100 injections into the brow, glabella, and crow’s-feet area. This method of injection resulted in a statistically significant improvement of forehead lines and brow ptosis and furrowing at follow-up between 10 and 45 days after treatment (P<.0001). Additionally, average brow height was significantly increased from 24.6 mm to 25 mm after treatment (P=.02).20
Conclusion
The use of BoNT products for both on- and off-label cosmetic and medical indications has rapidly grown over the past 2 decades. As demonstrated in this review, a variety of promising new products and delivery techniques are being developed. Given the rise in popularity of BoNT products among both physicians and consumers, clinicians should be aware of the current data and ongoing research.
- Carruthers JD, Carruthers JA. Treatment of glabellar frown lines with C. botulinum-A exotoxin. J Dermatol Surg Oncol. 1992;18:17-21.
- American Society for Aesthetic Plastic Surgery. Cosmetic Surgery National Data Bank statistics. American Society for Aesthetic Plastic Surgery website. http://www.surgery.org/sites/default/files/ASAPS-Stats2015.pdf. Accessed June 12, 2016.
- Walker TJ, Dayan SH. Comparison and overview of currently available neurotoxins. J Clin Aesthet Dermatol. 2014;7:31-39.
- Feng Z, Sun Q, He L, et al. Optimal dosage of botulinum toxin type A for treatment of glabellar frown lines: efficacy and safety in a clinical trial. Dermatol Surg. 2015;41(suppl 1):S56-S63.
- Jiang HY, Chen S, Zhou J, et al. Diffusion of two botulinum toxins type A on the forehead: double-blinded, randomized, controlled study. Dermatol Surg. 2014;40:184-192.
- Garcia-Murray E, Velasco Villasenor ML, Acevedo B, et al. Safety and efficacy of RT002, an injectable botulinum toxin type A, for treating glabellar lines: results of a phase 1/2, open-label, sequential dose-escalation study. Dermatol Surg. 2015;41(suppl 1):S47-S55.
- Won CH, Kim HK, Kim BJ, et al. Comparative trial of a novel botulinum neurotoxin type A versus onabotulinumtoxinA in the treatment of glabellar lines: a multicenter, randomized, double-blind, active-controlled study. Int J Dermatol. 2015;54:227-234.
- Kim BJ, Kwon HH, Park SY, et al. Double-blind, randomized non-inferiority trial of a novel botulinum toxin A processed from the strain CBFC26, compared with onabotulinumtoxin A in the treatment of glabellar lines. J Eur Acad Dermatol Venereol. 2014;28:1761-1767.
- Kim JE, Song EJ, Choi GS, et al. The efficacy and safety of liquid-type botulinum toxin type A for the management of moderate to severe glabellar frown lines. Plast Reconstr Surg. 2015;135:732-741.
- Alam M, Bolotin D, Carruthers J, et al. Consensus statement regarding storage and reuse of previously reconstituted neuromodulators. Dermatol Surg. 2015;41:321-326.
- Alam M, Geisler A, Sadhwani D, et al. Effect of needle size on pain perception in patients treated with botulinum toxin type A injections: a randomized clinical trial. JAMA Dermatol. 2015;151:1194-1199.
- Pucks N, Thomas A, Hallam MJ, et al. Cutaneous cooling to manage botulinum toxin injection-associated pain in patients with facial palsy: a randomised controlled trial. J Plast Reconstr Aesthet Surg. 2015;68:1701-1705.
- Kranz G, Sycha T, Voller B, et al. Pain sensation during intradermal injections of three different botulinum toxin preparations in different doses and dilutions. Dermatol Surg. 2006;32:886-890.
- Lowe PL, Lowe NJ. Botulinum toxin type B: pH change reduces injection pain, retains efficacy. Dermatol Surg. 2014;40:1328-1333.
- Mahmoud BH, Burnett C, Ozog D. Prospective randomized controlled study to determine the effect of topical application of botulinum toxin A for crow’s feet after treatment with ablative fractional CO2 laser. Dermatol Surg. 2015;41(suppl 1):S75-S81.
- Montaser-Kouhsari L, Zartab H, Fanian F, et al. Comparison of intradermal injection with iontophoresis of abo-botulinum toxin A for the treatment of primary axillary hyperhidrosis: a randomized, controlled trial. J Dermatolog Treat. 2014;25:337-341.
- Iannitti T, Palmieri B, Aspiro A, et al. A preliminary study of painless and effective transdermal botulinum toxin A delivery by jet nebulization for treatment of primary hyperhidrosis. Drug Des Devel Ther. 2014;8:931-935.
- Iozzo I, Tengattini V, Antonucci VA. Multipoint and multilevel injection technique of botulinum toxin A in facial aesthetics. J Cosmet Dermatol. 2014;13:135-142.
- Sneath J, Humphrey S, Carruthers A, et al. Injecting botulinum toxin at different depths is not effective for the correction of eyebrow asymmetry. Dermatol Surg. 2015;41(suppl 1):S82-S87.
- Steinsapir KD, Rootman D, Wulc A, et al. Cosmetic microdroplet botulinum toxin A forehead lift: a new treatment paradigm. Ophthal Plast Reconstr Surg. 2015;31:263-268.
- Carruthers JD, Carruthers JA. Treatment of glabellar frown lines with C. botulinum-A exotoxin. J Dermatol Surg Oncol. 1992;18:17-21.
- American Society for Aesthetic Plastic Surgery. Cosmetic Surgery National Data Bank statistics. American Society for Aesthetic Plastic Surgery website. http://www.surgery.org/sites/default/files/ASAPS-Stats2015.pdf. Accessed June 12, 2016.
- Walker TJ, Dayan SH. Comparison and overview of currently available neurotoxins. J Clin Aesthet Dermatol. 2014;7:31-39.
- Feng Z, Sun Q, He L, et al. Optimal dosage of botulinum toxin type A for treatment of glabellar frown lines: efficacy and safety in a clinical trial. Dermatol Surg. 2015;41(suppl 1):S56-S63.
- Jiang HY, Chen S, Zhou J, et al. Diffusion of two botulinum toxins type A on the forehead: double-blinded, randomized, controlled study. Dermatol Surg. 2014;40:184-192.
- Garcia-Murray E, Velasco Villasenor ML, Acevedo B, et al. Safety and efficacy of RT002, an injectable botulinum toxin type A, for treating glabellar lines: results of a phase 1/2, open-label, sequential dose-escalation study. Dermatol Surg. 2015;41(suppl 1):S47-S55.
- Won CH, Kim HK, Kim BJ, et al. Comparative trial of a novel botulinum neurotoxin type A versus onabotulinumtoxinA in the treatment of glabellar lines: a multicenter, randomized, double-blind, active-controlled study. Int J Dermatol. 2015;54:227-234.
- Kim BJ, Kwon HH, Park SY, et al. Double-blind, randomized non-inferiority trial of a novel botulinum toxin A processed from the strain CBFC26, compared with onabotulinumtoxin A in the treatment of glabellar lines. J Eur Acad Dermatol Venereol. 2014;28:1761-1767.
- Kim JE, Song EJ, Choi GS, et al. The efficacy and safety of liquid-type botulinum toxin type A for the management of moderate to severe glabellar frown lines. Plast Reconstr Surg. 2015;135:732-741.
- Alam M, Bolotin D, Carruthers J, et al. Consensus statement regarding storage and reuse of previously reconstituted neuromodulators. Dermatol Surg. 2015;41:321-326.
- Alam M, Geisler A, Sadhwani D, et al. Effect of needle size on pain perception in patients treated with botulinum toxin type A injections: a randomized clinical trial. JAMA Dermatol. 2015;151:1194-1199.
- Pucks N, Thomas A, Hallam MJ, et al. Cutaneous cooling to manage botulinum toxin injection-associated pain in patients with facial palsy: a randomised controlled trial. J Plast Reconstr Aesthet Surg. 2015;68:1701-1705.
- Kranz G, Sycha T, Voller B, et al. Pain sensation during intradermal injections of three different botulinum toxin preparations in different doses and dilutions. Dermatol Surg. 2006;32:886-890.
- Lowe PL, Lowe NJ. Botulinum toxin type B: pH change reduces injection pain, retains efficacy. Dermatol Surg. 2014;40:1328-1333.
- Mahmoud BH, Burnett C, Ozog D. Prospective randomized controlled study to determine the effect of topical application of botulinum toxin A for crow’s feet after treatment with ablative fractional CO2 laser. Dermatol Surg. 2015;41(suppl 1):S75-S81.
- Montaser-Kouhsari L, Zartab H, Fanian F, et al. Comparison of intradermal injection with iontophoresis of abo-botulinum toxin A for the treatment of primary axillary hyperhidrosis: a randomized, controlled trial. J Dermatolog Treat. 2014;25:337-341.
- Iannitti T, Palmieri B, Aspiro A, et al. A preliminary study of painless and effective transdermal botulinum toxin A delivery by jet nebulization for treatment of primary hyperhidrosis. Drug Des Devel Ther. 2014;8:931-935.
- Iozzo I, Tengattini V, Antonucci VA. Multipoint and multilevel injection technique of botulinum toxin A in facial aesthetics. J Cosmet Dermatol. 2014;13:135-142.
- Sneath J, Humphrey S, Carruthers A, et al. Injecting botulinum toxin at different depths is not effective for the correction of eyebrow asymmetry. Dermatol Surg. 2015;41(suppl 1):S82-S87.
- Steinsapir KD, Rootman D, Wulc A, et al. Cosmetic microdroplet botulinum toxin A forehead lift: a new treatment paradigm. Ophthal Plast Reconstr Surg. 2015;31:263-268.
Practice Points
- Botulinum neurotoxin (BoNT) injection is the most popular nonsurgical aesthetic procedure available of which there are currently 4 products approved by the US Food and Drug Administration.
- A variety of new BoNT products with unique properties and formulations are currently being studied, some of which are already available for clinical use in foreign markets.
- Administration technique and novel product delivery methods also can be utilized to minimize pain and maximize aesthetic outcomes.
Knowledge of the Platysma Muscle Anatomy in the Face Can Improve Cosmetic Outcomes
The platysma muscle has been widely studied in its course through the cervical region in an attempt to develop more effective surgical approaches for rejuvenation. As a person ages, the platysma muscle becomes thinner, less defined, and ptotic, which results in the clinical appearance of a sagging neck that prompts patients to undergo neck-lift procedures. However, little is known about the course of the platysma in the mid and lower face and the implications on facial aging.
Bae et al (Plast Reconstr Surg. 2016;138:365-371) performed a cadaveric dissection of the facial portion of the platysma muscle to delineate the morphology and extension of the muscle in the mid and lower face. Thirty-four adult hemifaces were dissected. The demographics were broken down by age (mean, 71.4 years; range, 41–93 years), gender (29 men; 5 women), and ethnicity (14 Korean; 20 Thai). The extension of the platysma was documented according to a grid the authors created. The grid was divided along 3 horizontal lines (H1–H3) and 3 vertical lines (V1–V3). The intersection of these lines created a grid that was comprised of 3×3 squares and was labeled superior (S1–S3), middle (M1–M3), and inferior (I1–I3). H1 was a line drawn horizontally from the tragus to the lateral orbit, H2 was a line from the earlobe to the nasal ala, and H3 was a line from the angle of the mandible to the corner of the mouth.
The extension pattern of the facial portion of the platysma muscle was classified into patterns: A (n=3; S1–S2, M1–M3, and I1-I3 were covered by the muscle), B-1 (n=20; M1–M3, I1–I3), B-2 (n=9; M1–M2, I1–I3), and C (n=2; I1-I3).
The platysma muscle is divided into anterior and posterior portions. The anterior platysma ascends superiorly and medially in the face to interdigitate with the depressor anguli oris and the depressor labii inferioris muscles. The morphology pattern of the posterior platysma varied and was classified into 1 of 3 patterns: straight (n=13), straight-curved (n=18), and curved (n=3). The straight pattern showed platysma fibers that travel parallel to the mandibular malar line and head toward the zygomaticus major or the orbicularis oculi muscles. The straight-curved type has fibers that travel straight from the neck to the face but then curve, heading toward the risorius, zygomaticus major, or orbicularis oculi muscle depending on the extent of the platysma muscle in the area. The curved type has fibers that run parallel to the zygomaticus arch and interdigitate with the orbicularis oculi muscle and either the risorius or zygomaticus major muscle.
What’s the issue?
For many years, the cervical platysma muscle was studied and surgical approaches were modified to enhance results. The authors of this study have taken the study of the platysma muscle further to delineate its course through the face. Knowledge of the various pathways of the muscle can help us determine the most natural direction to resuspend ptotic facial tissue during surgery and minimally invasive procedures such as thread-lifting. The most salient points include: (1) the lower one-third of the masseter muscle is covered by the platysma, and (2) straight-curved was the most common morphology type, suggesting that the vector for repositioning the platysma muscle is vertical.
Although Bae et al do not discuss the use of botulinum toxin (BTX) in the treatment of the platysma, the results of this study further support the importance of BTX to the lower face. We can look back at more than 14 years of cosmetic use of BTX for the glabella and see the reduced number of brow-lift surgeries nationwide. Although the preventative effects of BTX on brow depression still need to be scientifically proven, it may behoove us to think preventatively on the use of BTX in the platysma to minimize ptosis of the lower face and neck. The results of this anatomic study support the importance of tailoring the approach to the dynamic lines caused by the platysma muscle in each patient to address jowls and neck bands. Have you been offering lower face BTX treatments to patients in an effort to reduce lower face aging?
The platysma muscle has been widely studied in its course through the cervical region in an attempt to develop more effective surgical approaches for rejuvenation. As a person ages, the platysma muscle becomes thinner, less defined, and ptotic, which results in the clinical appearance of a sagging neck that prompts patients to undergo neck-lift procedures. However, little is known about the course of the platysma in the mid and lower face and the implications on facial aging.
Bae et al (Plast Reconstr Surg. 2016;138:365-371) performed a cadaveric dissection of the facial portion of the platysma muscle to delineate the morphology and extension of the muscle in the mid and lower face. Thirty-four adult hemifaces were dissected. The demographics were broken down by age (mean, 71.4 years; range, 41–93 years), gender (29 men; 5 women), and ethnicity (14 Korean; 20 Thai). The extension of the platysma was documented according to a grid the authors created. The grid was divided along 3 horizontal lines (H1–H3) and 3 vertical lines (V1–V3). The intersection of these lines created a grid that was comprised of 3×3 squares and was labeled superior (S1–S3), middle (M1–M3), and inferior (I1–I3). H1 was a line drawn horizontally from the tragus to the lateral orbit, H2 was a line from the earlobe to the nasal ala, and H3 was a line from the angle of the mandible to the corner of the mouth.
The extension pattern of the facial portion of the platysma muscle was classified into patterns: A (n=3; S1–S2, M1–M3, and I1-I3 were covered by the muscle), B-1 (n=20; M1–M3, I1–I3), B-2 (n=9; M1–M2, I1–I3), and C (n=2; I1-I3).
The platysma muscle is divided into anterior and posterior portions. The anterior platysma ascends superiorly and medially in the face to interdigitate with the depressor anguli oris and the depressor labii inferioris muscles. The morphology pattern of the posterior platysma varied and was classified into 1 of 3 patterns: straight (n=13), straight-curved (n=18), and curved (n=3). The straight pattern showed platysma fibers that travel parallel to the mandibular malar line and head toward the zygomaticus major or the orbicularis oculi muscles. The straight-curved type has fibers that travel straight from the neck to the face but then curve, heading toward the risorius, zygomaticus major, or orbicularis oculi muscle depending on the extent of the platysma muscle in the area. The curved type has fibers that run parallel to the zygomaticus arch and interdigitate with the orbicularis oculi muscle and either the risorius or zygomaticus major muscle.
What’s the issue?
For many years, the cervical platysma muscle was studied and surgical approaches were modified to enhance results. The authors of this study have taken the study of the platysma muscle further to delineate its course through the face. Knowledge of the various pathways of the muscle can help us determine the most natural direction to resuspend ptotic facial tissue during surgery and minimally invasive procedures such as thread-lifting. The most salient points include: (1) the lower one-third of the masseter muscle is covered by the platysma, and (2) straight-curved was the most common morphology type, suggesting that the vector for repositioning the platysma muscle is vertical.
Although Bae et al do not discuss the use of botulinum toxin (BTX) in the treatment of the platysma, the results of this study further support the importance of BTX to the lower face. We can look back at more than 14 years of cosmetic use of BTX for the glabella and see the reduced number of brow-lift surgeries nationwide. Although the preventative effects of BTX on brow depression still need to be scientifically proven, it may behoove us to think preventatively on the use of BTX in the platysma to minimize ptosis of the lower face and neck. The results of this anatomic study support the importance of tailoring the approach to the dynamic lines caused by the platysma muscle in each patient to address jowls and neck bands. Have you been offering lower face BTX treatments to patients in an effort to reduce lower face aging?
The platysma muscle has been widely studied in its course through the cervical region in an attempt to develop more effective surgical approaches for rejuvenation. As a person ages, the platysma muscle becomes thinner, less defined, and ptotic, which results in the clinical appearance of a sagging neck that prompts patients to undergo neck-lift procedures. However, little is known about the course of the platysma in the mid and lower face and the implications on facial aging.
Bae et al (Plast Reconstr Surg. 2016;138:365-371) performed a cadaveric dissection of the facial portion of the platysma muscle to delineate the morphology and extension of the muscle in the mid and lower face. Thirty-four adult hemifaces were dissected. The demographics were broken down by age (mean, 71.4 years; range, 41–93 years), gender (29 men; 5 women), and ethnicity (14 Korean; 20 Thai). The extension of the platysma was documented according to a grid the authors created. The grid was divided along 3 horizontal lines (H1–H3) and 3 vertical lines (V1–V3). The intersection of these lines created a grid that was comprised of 3×3 squares and was labeled superior (S1–S3), middle (M1–M3), and inferior (I1–I3). H1 was a line drawn horizontally from the tragus to the lateral orbit, H2 was a line from the earlobe to the nasal ala, and H3 was a line from the angle of the mandible to the corner of the mouth.
The extension pattern of the facial portion of the platysma muscle was classified into patterns: A (n=3; S1–S2, M1–M3, and I1-I3 were covered by the muscle), B-1 (n=20; M1–M3, I1–I3), B-2 (n=9; M1–M2, I1–I3), and C (n=2; I1-I3).
The platysma muscle is divided into anterior and posterior portions. The anterior platysma ascends superiorly and medially in the face to interdigitate with the depressor anguli oris and the depressor labii inferioris muscles. The morphology pattern of the posterior platysma varied and was classified into 1 of 3 patterns: straight (n=13), straight-curved (n=18), and curved (n=3). The straight pattern showed platysma fibers that travel parallel to the mandibular malar line and head toward the zygomaticus major or the orbicularis oculi muscles. The straight-curved type has fibers that travel straight from the neck to the face but then curve, heading toward the risorius, zygomaticus major, or orbicularis oculi muscle depending on the extent of the platysma muscle in the area. The curved type has fibers that run parallel to the zygomaticus arch and interdigitate with the orbicularis oculi muscle and either the risorius or zygomaticus major muscle.
What’s the issue?
For many years, the cervical platysma muscle was studied and surgical approaches were modified to enhance results. The authors of this study have taken the study of the platysma muscle further to delineate its course through the face. Knowledge of the various pathways of the muscle can help us determine the most natural direction to resuspend ptotic facial tissue during surgery and minimally invasive procedures such as thread-lifting. The most salient points include: (1) the lower one-third of the masseter muscle is covered by the platysma, and (2) straight-curved was the most common morphology type, suggesting that the vector for repositioning the platysma muscle is vertical.
Although Bae et al do not discuss the use of botulinum toxin (BTX) in the treatment of the platysma, the results of this study further support the importance of BTX to the lower face. We can look back at more than 14 years of cosmetic use of BTX for the glabella and see the reduced number of brow-lift surgeries nationwide. Although the preventative effects of BTX on brow depression still need to be scientifically proven, it may behoove us to think preventatively on the use of BTX in the platysma to minimize ptosis of the lower face and neck. The results of this anatomic study support the importance of tailoring the approach to the dynamic lines caused by the platysma muscle in each patient to address jowls and neck bands. Have you been offering lower face BTX treatments to patients in an effort to reduce lower face aging?
Preventing, Identifying, and Managing Cosmetic Procedure Complications, Part 2: Lasers and Chemical Peels
The primary cosmetic procedures that dermatology residents will perform or assist in performing during their training are soft-tissue augmentation, botulinum toxin injections, laser therapy, and chemical peels. Because complications can occur from these procedures, it is important for residents to learn how to prevent, identify, and manage them for optimal patient outcomes. In part 2 of this series, laser therapy and chemical peels are discussed.
Lasers
In dermatology, lasers are used to treat dyschromia, resurface scars, remove skin growths, and rejuvenate aging skin.1,2 Ablative resurfacing lasers such as the CO2 laser are the most likely to lead to unwanted side effects. There is a risk for herpes simplex virus reactivation, impetigo, persistent erythema, dyschromia, and scarring.1-3 Some patients who undergo facial ablative resurfacing may develop a visible hypopigmented line of demarcation between treated and untreated skin along the jawline.3 With the development of fractional resurfacing lasers, the risk for dyschromia, persistent erythema, and scarring was lessened.1-3
Regardless of the type of resurfacing laser used, patients should be given adequate prophylaxis with an antiviral and antibiotic. For skin of color, fractional resurfacing lasers should be set at lower density settings with a higher fluence.1-3 Sites with fewer adnexal structures (eg, neck, dorsal hands) also should be treated at lower densities.3 When using Q-switched lasers that target pigment, caution should be used to avoid vesicle formation and/or skin crusting, which may lead to scarring or dyschromia.1-3 Some tattoo inks may paradoxically darken when treated with lasers.3 A test spot is advised, especially prior to treatment of permanent makeup tattoos. A pigmented lesion should never be treated if the diagnosis is unclear (eg, a biopsy to establish the diagnosis may be the best appropriate step for some pigmented lesions). For laser hair removal, the Nd:YAG laser is the safest for skin of color.2,3
Lasers that target vascular structures may cause unwanted purpura, hypopigmentation, or thermal injury.1-3 A larger spot size may help decrease the risk for purpura. The skin should be cooled properly and caution should be used to avoid pulse stacking. For intense pulsed light devices, overlap pulses slightly to avoid a zebralike pattern of slivers of untreated skin.1-3 For all laser procedures, strict sun protection is advised before and after the procedure.
Chemical Peels
Chemical peels are versatile and varied in their composition. They are categorized based on the depth to which the skin is affected by the peel: superficial (stratum corneum), medium (full-thickness epidermis), or deep (mid reticular dermis).4 Peels are most commonly used to treat dyschromia, aging, rhytides, actinic damage, and superficial scars.4,5 The success of a chemical peel depends largely on patient selection and preprocedure preparation. Patients who tend to develop postinflammatory hyperpigmention, have an underlying inflammatory or scarring skin disorder, are on photosensitizing medications, or have continued work- or hobby-related sun exposure are generally poor peel candidates.4,5 Strict sun protection should be advised both before and after a chemical peel.
While in training, residents are unlikely to perform a medium or deep peel. Superficial peels can be accomplished with trichloroacetic acid 10%, glycolic acid (GA) 30% to 50%, salicylic acid (SA) 20% to 30%, Jessner solution (SA, lactic acid, and resorcinol with ethanol), and tretinoin 1% to 5%.4 Glycolic acid and SA are known to be safer for patients with skin of color.4,5
Care should always be taken to prepare the skin for an even peel. Mild peeling agents such as tretinoin or adapalene may be used to prepare the skin in the weeks before the procedure.4 Skin of color may benefit from hydroquinone used before and after a chemical peel.5 At the time of the peel, acetone can be used to degrease the skin for a more even, effective peel. If a peel needs to be neutralized (eg, GA), make sure to have the neutralization solution on hand, as leaving the peel solution on for too long can lead to severe epidermolysis, which can be visualized by a graying of the skin and will not be seen with a properly performed superficial peel.4 Care should be taken at all times to protect the patient’s eyes. Eye flushes should be readily available. The medial canthus and perinasal folds may be protected with petrolatum. For a superficial peel, some desquamation (less with GA) and erythema may be noted for a few days.
Final Thoughts
For any cosmetic procedure, the patient’s expectations should be discussed. The provider may adeptly guide the patient toward realistic expectations for the procedure. Pretreatment and posttreatment photographs should always be taken to help document treatment progress; it may be helpful to show the patient the photographs at each visit. The expected skin reactions, recovery time, and risks should be fully discussed. Full informed consent should be obtained. Complications from cosmetic procedures will inevitably arise. As residents, we can take the opportunity to learn how to prevent, identify, and manage them.
- Hirsch R, Stier M. Complications and their management in cosmetic dermatology. Dermatol Clin. 2009;27:507-520.
- Oliaei A, Nelson JS, Fitzpatrick R, et al. Laser treatment of scars. Facial Plast Surg. 2012;28:518-524.
- Al Nomair N, Nazarian R, Marmur E. Complications in lasers, lights, and radiofrequency devices. Facial Plast Surg. 2012;28:340-346.
- Khunger N, IADVL Task Force. Standard guidelines of care for chemical peels. Indian J Dermatol Venereol Leprol. 2008;74(suppl):S5-S12.
- Sarkar R, Bansal S, Garg VK. Chemical peels for melasma in dark-skinned patients. J Cutan Aesthet Surg. 2012;5:247-253.
The primary cosmetic procedures that dermatology residents will perform or assist in performing during their training are soft-tissue augmentation, botulinum toxin injections, laser therapy, and chemical peels. Because complications can occur from these procedures, it is important for residents to learn how to prevent, identify, and manage them for optimal patient outcomes. In part 2 of this series, laser therapy and chemical peels are discussed.
Lasers
In dermatology, lasers are used to treat dyschromia, resurface scars, remove skin growths, and rejuvenate aging skin.1,2 Ablative resurfacing lasers such as the CO2 laser are the most likely to lead to unwanted side effects. There is a risk for herpes simplex virus reactivation, impetigo, persistent erythema, dyschromia, and scarring.1-3 Some patients who undergo facial ablative resurfacing may develop a visible hypopigmented line of demarcation between treated and untreated skin along the jawline.3 With the development of fractional resurfacing lasers, the risk for dyschromia, persistent erythema, and scarring was lessened.1-3
Regardless of the type of resurfacing laser used, patients should be given adequate prophylaxis with an antiviral and antibiotic. For skin of color, fractional resurfacing lasers should be set at lower density settings with a higher fluence.1-3 Sites with fewer adnexal structures (eg, neck, dorsal hands) also should be treated at lower densities.3 When using Q-switched lasers that target pigment, caution should be used to avoid vesicle formation and/or skin crusting, which may lead to scarring or dyschromia.1-3 Some tattoo inks may paradoxically darken when treated with lasers.3 A test spot is advised, especially prior to treatment of permanent makeup tattoos. A pigmented lesion should never be treated if the diagnosis is unclear (eg, a biopsy to establish the diagnosis may be the best appropriate step for some pigmented lesions). For laser hair removal, the Nd:YAG laser is the safest for skin of color.2,3
Lasers that target vascular structures may cause unwanted purpura, hypopigmentation, or thermal injury.1-3 A larger spot size may help decrease the risk for purpura. The skin should be cooled properly and caution should be used to avoid pulse stacking. For intense pulsed light devices, overlap pulses slightly to avoid a zebralike pattern of slivers of untreated skin.1-3 For all laser procedures, strict sun protection is advised before and after the procedure.
Chemical Peels
Chemical peels are versatile and varied in their composition. They are categorized based on the depth to which the skin is affected by the peel: superficial (stratum corneum), medium (full-thickness epidermis), or deep (mid reticular dermis).4 Peels are most commonly used to treat dyschromia, aging, rhytides, actinic damage, and superficial scars.4,5 The success of a chemical peel depends largely on patient selection and preprocedure preparation. Patients who tend to develop postinflammatory hyperpigmention, have an underlying inflammatory or scarring skin disorder, are on photosensitizing medications, or have continued work- or hobby-related sun exposure are generally poor peel candidates.4,5 Strict sun protection should be advised both before and after a chemical peel.
While in training, residents are unlikely to perform a medium or deep peel. Superficial peels can be accomplished with trichloroacetic acid 10%, glycolic acid (GA) 30% to 50%, salicylic acid (SA) 20% to 30%, Jessner solution (SA, lactic acid, and resorcinol with ethanol), and tretinoin 1% to 5%.4 Glycolic acid and SA are known to be safer for patients with skin of color.4,5
Care should always be taken to prepare the skin for an even peel. Mild peeling agents such as tretinoin or adapalene may be used to prepare the skin in the weeks before the procedure.4 Skin of color may benefit from hydroquinone used before and after a chemical peel.5 At the time of the peel, acetone can be used to degrease the skin for a more even, effective peel. If a peel needs to be neutralized (eg, GA), make sure to have the neutralization solution on hand, as leaving the peel solution on for too long can lead to severe epidermolysis, which can be visualized by a graying of the skin and will not be seen with a properly performed superficial peel.4 Care should be taken at all times to protect the patient’s eyes. Eye flushes should be readily available. The medial canthus and perinasal folds may be protected with petrolatum. For a superficial peel, some desquamation (less with GA) and erythema may be noted for a few days.
Final Thoughts
For any cosmetic procedure, the patient’s expectations should be discussed. The provider may adeptly guide the patient toward realistic expectations for the procedure. Pretreatment and posttreatment photographs should always be taken to help document treatment progress; it may be helpful to show the patient the photographs at each visit. The expected skin reactions, recovery time, and risks should be fully discussed. Full informed consent should be obtained. Complications from cosmetic procedures will inevitably arise. As residents, we can take the opportunity to learn how to prevent, identify, and manage them.
The primary cosmetic procedures that dermatology residents will perform or assist in performing during their training are soft-tissue augmentation, botulinum toxin injections, laser therapy, and chemical peels. Because complications can occur from these procedures, it is important for residents to learn how to prevent, identify, and manage them for optimal patient outcomes. In part 2 of this series, laser therapy and chemical peels are discussed.
Lasers
In dermatology, lasers are used to treat dyschromia, resurface scars, remove skin growths, and rejuvenate aging skin.1,2 Ablative resurfacing lasers such as the CO2 laser are the most likely to lead to unwanted side effects. There is a risk for herpes simplex virus reactivation, impetigo, persistent erythema, dyschromia, and scarring.1-3 Some patients who undergo facial ablative resurfacing may develop a visible hypopigmented line of demarcation between treated and untreated skin along the jawline.3 With the development of fractional resurfacing lasers, the risk for dyschromia, persistent erythema, and scarring was lessened.1-3
Regardless of the type of resurfacing laser used, patients should be given adequate prophylaxis with an antiviral and antibiotic. For skin of color, fractional resurfacing lasers should be set at lower density settings with a higher fluence.1-3 Sites with fewer adnexal structures (eg, neck, dorsal hands) also should be treated at lower densities.3 When using Q-switched lasers that target pigment, caution should be used to avoid vesicle formation and/or skin crusting, which may lead to scarring or dyschromia.1-3 Some tattoo inks may paradoxically darken when treated with lasers.3 A test spot is advised, especially prior to treatment of permanent makeup tattoos. A pigmented lesion should never be treated if the diagnosis is unclear (eg, a biopsy to establish the diagnosis may be the best appropriate step for some pigmented lesions). For laser hair removal, the Nd:YAG laser is the safest for skin of color.2,3
Lasers that target vascular structures may cause unwanted purpura, hypopigmentation, or thermal injury.1-3 A larger spot size may help decrease the risk for purpura. The skin should be cooled properly and caution should be used to avoid pulse stacking. For intense pulsed light devices, overlap pulses slightly to avoid a zebralike pattern of slivers of untreated skin.1-3 For all laser procedures, strict sun protection is advised before and after the procedure.
Chemical Peels
Chemical peels are versatile and varied in their composition. They are categorized based on the depth to which the skin is affected by the peel: superficial (stratum corneum), medium (full-thickness epidermis), or deep (mid reticular dermis).4 Peels are most commonly used to treat dyschromia, aging, rhytides, actinic damage, and superficial scars.4,5 The success of a chemical peel depends largely on patient selection and preprocedure preparation. Patients who tend to develop postinflammatory hyperpigmention, have an underlying inflammatory or scarring skin disorder, are on photosensitizing medications, or have continued work- or hobby-related sun exposure are generally poor peel candidates.4,5 Strict sun protection should be advised both before and after a chemical peel.
While in training, residents are unlikely to perform a medium or deep peel. Superficial peels can be accomplished with trichloroacetic acid 10%, glycolic acid (GA) 30% to 50%, salicylic acid (SA) 20% to 30%, Jessner solution (SA, lactic acid, and resorcinol with ethanol), and tretinoin 1% to 5%.4 Glycolic acid and SA are known to be safer for patients with skin of color.4,5
Care should always be taken to prepare the skin for an even peel. Mild peeling agents such as tretinoin or adapalene may be used to prepare the skin in the weeks before the procedure.4 Skin of color may benefit from hydroquinone used before and after a chemical peel.5 At the time of the peel, acetone can be used to degrease the skin for a more even, effective peel. If a peel needs to be neutralized (eg, GA), make sure to have the neutralization solution on hand, as leaving the peel solution on for too long can lead to severe epidermolysis, which can be visualized by a graying of the skin and will not be seen with a properly performed superficial peel.4 Care should be taken at all times to protect the patient’s eyes. Eye flushes should be readily available. The medial canthus and perinasal folds may be protected with petrolatum. For a superficial peel, some desquamation (less with GA) and erythema may be noted for a few days.
Final Thoughts
For any cosmetic procedure, the patient’s expectations should be discussed. The provider may adeptly guide the patient toward realistic expectations for the procedure. Pretreatment and posttreatment photographs should always be taken to help document treatment progress; it may be helpful to show the patient the photographs at each visit. The expected skin reactions, recovery time, and risks should be fully discussed. Full informed consent should be obtained. Complications from cosmetic procedures will inevitably arise. As residents, we can take the opportunity to learn how to prevent, identify, and manage them.
- Hirsch R, Stier M. Complications and their management in cosmetic dermatology. Dermatol Clin. 2009;27:507-520.
- Oliaei A, Nelson JS, Fitzpatrick R, et al. Laser treatment of scars. Facial Plast Surg. 2012;28:518-524.
- Al Nomair N, Nazarian R, Marmur E. Complications in lasers, lights, and radiofrequency devices. Facial Plast Surg. 2012;28:340-346.
- Khunger N, IADVL Task Force. Standard guidelines of care for chemical peels. Indian J Dermatol Venereol Leprol. 2008;74(suppl):S5-S12.
- Sarkar R, Bansal S, Garg VK. Chemical peels for melasma in dark-skinned patients. J Cutan Aesthet Surg. 2012;5:247-253.
- Hirsch R, Stier M. Complications and their management in cosmetic dermatology. Dermatol Clin. 2009;27:507-520.
- Oliaei A, Nelson JS, Fitzpatrick R, et al. Laser treatment of scars. Facial Plast Surg. 2012;28:518-524.
- Al Nomair N, Nazarian R, Marmur E. Complications in lasers, lights, and radiofrequency devices. Facial Plast Surg. 2012;28:340-346.
- Khunger N, IADVL Task Force. Standard guidelines of care for chemical peels. Indian J Dermatol Venereol Leprol. 2008;74(suppl):S5-S12.
- Sarkar R, Bansal S, Garg VK. Chemical peels for melasma in dark-skinned patients. J Cutan Aesthet Surg. 2012;5:247-253.
Update on vitamin E
Available in the diet through fresh vegetables (particularly green leafy vegetables), vegetable oils, grains, nuts, seeds, corn, soy, whole wheat flour, margarine, and in some meat and dairy products, vitamin E, or tocopherol, is the primary lipid-soluble antioxidant found in human skin (via sebum), membranes, plasma, and tissues that protects cells from oxidative stress.1-4 Vitamin E is often used to treat minor burns, surgical scars, and other wounds, although the Food and Drug Administration has not approved its use for skin conditions.
In 1938, Karrer, Fritzsche, Ringier, and Salomon became the first to synthesize alpha-tocopherol,5,6 the main biologically active form of vitamin E.7 In the 1940s, vitamin E was labeled a “chain-breaking” antioxidant for its role in hindering the chain reaction induced by free radicals, and it is known to protect cutaneous cell membranes from peroxidation.8 Most topical formulations contain synthetic laboratory-made alpha-tocopherol or one of its many esters or ethers. As an ingredient in skin care agents, significant evidence has been amassed to suggest that topically applied vitamin E confers photoprotective activity against erythema, edema, sun burn cell formation, and other indicators of acute UV-induced damage as well as responses to chronic UVA and UVB exposure, including skin wrinkling and skin cancer.2,9-14 This column will focus on the topical applications of vitamin E.
Topical uses and findings
The lipophilic nature of vitamin E makes it suitable for topical application and percutaneous absorption through the skin.9,15 Vitamin E is generally used in 1%-5% concentrations alpha-tocopherol or tocopherol acetate in over-the-counter products.16 When topically applied, vitamin E has been shown to hydrate the stratum corneum (SC) and improve water-binding capacity.16 It is also considered an effective ingredient for imparting skin protection and treating atopic dermatitis (AD).2
In 2005, Ekanayake-Mudiyanselage et al. studied whether one application of an alpha-tocopherol–enriched rinse-off product could effectively lead to deposition of alpha-tocopherol on the SC in 13 volunteers. The researchers found that the alpha-tocopherol product raised alpha-tocopherol levels in surface lipids, which remained consistent for at least 24 hours, whereas such levels were reduced in the alpha-tocopherol–free vehicle control group. The alpha-tocopherol rinse-off product also significantly inhibited photo-oxidation of squalene.7
A 2009 6-month study in healthy human volunteers with actinic keratoses demonstrated that while topically applied dl-alpha-tocopherol, of which cutaneous levels were significantly increased at the end of the study, did not significantly change already present lesions, alterations in polyamine metabolism revealed that squamous cell carcinogenesis potential was significantly diminished.17
Patrizi et al., in a 2015 randomized, controlled, double-blind, single center study, assessed the safety and efficacy of MD2011001 cream (a nonsteroidal topical cream including vitamin E, epigallocatechin gallate and grape seed procyanidins) versus placebo, in 44 patients with mild to moderate AD in the perioral/periocular area and/or the neck. The researchers noted a significantly more rapid reduction in affected surface area with the test formulation, compared with placebo; the product was found to be well tolerated and safe as well as effective for mild to moderate AD.18
Also that year, Ruiz-Tovar et al. performed a prospective randomized clinical trial in 60 patients, showing that topical vitamin E ointment reduced postoperative pain.19
The vitamin C, vitamin E, ferulic acid combination
Vitamin E is perceived to be more effective when used in combination with other antioxidant ingredients. Some data suggest a cumulative benefit derived from using oral and topical antioxidant products in combination, including vitamins C and E in particular.20-22 Because vitamin C can restore oxidized vitamin E, combining the antioxidants is a stabilizing factor in topical formulations.23,24 Further, ferulic acid has been shown to stabilize both vitamins, with the topical combination yielding photoprotective effects against UVB exposure, including the significant reduction in thymine dimer formation.9,24,25
A small study of nine patients conducted by Murray et al. in 2008 found that a stable topical preparation of 15% l-ascorbic acid, 1% alpha-tocopherol, and 0.5% ferulic acid protected human skin in vivo from UV-induced damage, specifically erythema and apoptosis. The formulation also suppressed p53 activation and limited thymine dimer mutations, which are associated with skin cancer.26
Waibel et al. conducted a double-blind, prospective, single-center, randomized split-face trial in 2015 to study whether laser-assisted delivery of vitamins C and E and ferulic acid after fractional ablative laser procedures to treat photodamage could enhance wound healing. Fifteen healthy men and women (aged 30-55 years) were treated with the combination formulation on one side of the face and vehicle on the other side within 2 minutes of receiving fractional ablative CO2 laser surgery. They also received daily treatments and evaluations during days 1 through 7 of healing. Edema was found to be diminished on the sides treated with the antioxidant combination, compared with vehicle on day 7, and erythema, on days 3 and 5.27
Other vitamin E combinations
In 2014, Farris et al. found vitamin E to be a key ingredient, along with resveratrol and baicalin, in a nighttime antioxidant formulation that netted a statistically significant improvement in skin rejuvenation, specifically ameliorating fine lines and wrinkles, skin firmness, skin elasticity, skin laxity, hyperpigmentation, radiance, and skin roughness over 3 months, compared with baseline.28
Pereira et al. reported in 2014 that they found that the topical application of polymeric bioadhesive films containing aloe vera and vitamin E acetate appear to be an effective approach to burn treatment.29
A 2015 randomized, controlled, double-blind prospective study in 30 healthy volunteers also indicated that an SPF 30 sunscreen supplemented with an antioxidant combination containing grape seed extract, vitamin E, coenzyme Q10, and vitamin C effectively protected skin against infrared A radiation damage, unlike the use of the SPF 30 product without the antioxidant cocktail.30
Conclusion
Combining vitamin E with other antioxidants appears to enhance its bioactivity and, likely, that of the other interacting antioxidants. The potential therapeutic benefits of vitamin E in preventing and treating skin cancer and photoaging remain an important focus of research. As an ingredient in topical anti-aging skin care preparations, vitamin E displays emollient properties, and is stable, easy to formulate, and relatively inexpensive, making it a popular additive. More controlled trials are necessary to fully clarify the role of vitamin E in treating various dermatoses.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
References
1. J Mol Med (Berl). 1995 Jan;73(1):7-17.
2. Dermatol Surg. 2005 Jul;31(7 Pt 2):805-13.
3. J Cosmet Dermatol. 2004 Jul;3(3):149-55.
4. Skin Pharmacol Appl Skin Physiol. 2001;14 Suppl 1:87-91.
5. Am J Clin Nutr. 1987 Jul;46(1 Suppl):183-6.
6. Nutr Rev. 2012 Sep;70(9):483-90.
7. Skin Pharmacol Physiol. 2005 Jan-Feb;18(1):20-6.
8. Ann Nutr Metab. 2012;61(3):207-12.
9. J Am Acad Dermatol. 2003 Jun;48(6):866-74.
10. Plast Reconstr Surg. 1997 Sep;100(4):973-80.
11. Acta Derm Venereol. 1996 Jul;76(4):264-8.
12. J Invest Dermatol. 1995 Apr;104(4):484-8.
13. Photodermatol Photoimmunol Photomed. 1990 Apr;7(2):56-62.
14. Photodermatol. 1989 Oct;6(5):228-33.
15. Drug Metab Rev. 2000 Aug-Nov;32(3-4):413-20.
16. Clin Dermatol. 2009 Sep-Oct;27(5):469-74.
17. Cancer Prev Res (Phila). 2009 Apr;2(4):394-400.
18. J Dermatolog Treat. 2015 Dec 10:1-5. [Epub ahead of print].
19. Int J Colorectal Dis. 2016 Jul;31(7):1371-2.
20. Skin Pharmacol Appl Skin Physiol. 15:307;2002.
21. Biofactors. 2003;18(1-4):289-97.
22. J Drugs Dermatol. 2008 Jul;7(7 Suppl):s2-6.
23. Can J Physiol Pharmacol. 1993 Sep;71(9):725-31.
24. PLoS One. 2013 May 14;8(5):e63809.
25. J Invest Dermatol. 2005 Oct;125(4):826-32.
26. J Am Acad Dermatol. 2008 Sep;59(3):418-25.
27. Lasers Surg Med. 2016 Mar;48(3):238-44.
28. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
Available in the diet through fresh vegetables (particularly green leafy vegetables), vegetable oils, grains, nuts, seeds, corn, soy, whole wheat flour, margarine, and in some meat and dairy products, vitamin E, or tocopherol, is the primary lipid-soluble antioxidant found in human skin (via sebum), membranes, plasma, and tissues that protects cells from oxidative stress.1-4 Vitamin E is often used to treat minor burns, surgical scars, and other wounds, although the Food and Drug Administration has not approved its use for skin conditions.
In 1938, Karrer, Fritzsche, Ringier, and Salomon became the first to synthesize alpha-tocopherol,5,6 the main biologically active form of vitamin E.7 In the 1940s, vitamin E was labeled a “chain-breaking” antioxidant for its role in hindering the chain reaction induced by free radicals, and it is known to protect cutaneous cell membranes from peroxidation.8 Most topical formulations contain synthetic laboratory-made alpha-tocopherol or one of its many esters or ethers. As an ingredient in skin care agents, significant evidence has been amassed to suggest that topically applied vitamin E confers photoprotective activity against erythema, edema, sun burn cell formation, and other indicators of acute UV-induced damage as well as responses to chronic UVA and UVB exposure, including skin wrinkling and skin cancer.2,9-14 This column will focus on the topical applications of vitamin E.
Topical uses and findings
The lipophilic nature of vitamin E makes it suitable for topical application and percutaneous absorption through the skin.9,15 Vitamin E is generally used in 1%-5% concentrations alpha-tocopherol or tocopherol acetate in over-the-counter products.16 When topically applied, vitamin E has been shown to hydrate the stratum corneum (SC) and improve water-binding capacity.16 It is also considered an effective ingredient for imparting skin protection and treating atopic dermatitis (AD).2
In 2005, Ekanayake-Mudiyanselage et al. studied whether one application of an alpha-tocopherol–enriched rinse-off product could effectively lead to deposition of alpha-tocopherol on the SC in 13 volunteers. The researchers found that the alpha-tocopherol product raised alpha-tocopherol levels in surface lipids, which remained consistent for at least 24 hours, whereas such levels were reduced in the alpha-tocopherol–free vehicle control group. The alpha-tocopherol rinse-off product also significantly inhibited photo-oxidation of squalene.7
A 2009 6-month study in healthy human volunteers with actinic keratoses demonstrated that while topically applied dl-alpha-tocopherol, of which cutaneous levels were significantly increased at the end of the study, did not significantly change already present lesions, alterations in polyamine metabolism revealed that squamous cell carcinogenesis potential was significantly diminished.17
Patrizi et al., in a 2015 randomized, controlled, double-blind, single center study, assessed the safety and efficacy of MD2011001 cream (a nonsteroidal topical cream including vitamin E, epigallocatechin gallate and grape seed procyanidins) versus placebo, in 44 patients with mild to moderate AD in the perioral/periocular area and/or the neck. The researchers noted a significantly more rapid reduction in affected surface area with the test formulation, compared with placebo; the product was found to be well tolerated and safe as well as effective for mild to moderate AD.18
Also that year, Ruiz-Tovar et al. performed a prospective randomized clinical trial in 60 patients, showing that topical vitamin E ointment reduced postoperative pain.19
The vitamin C, vitamin E, ferulic acid combination
Vitamin E is perceived to be more effective when used in combination with other antioxidant ingredients. Some data suggest a cumulative benefit derived from using oral and topical antioxidant products in combination, including vitamins C and E in particular.20-22 Because vitamin C can restore oxidized vitamin E, combining the antioxidants is a stabilizing factor in topical formulations.23,24 Further, ferulic acid has been shown to stabilize both vitamins, with the topical combination yielding photoprotective effects against UVB exposure, including the significant reduction in thymine dimer formation.9,24,25
A small study of nine patients conducted by Murray et al. in 2008 found that a stable topical preparation of 15% l-ascorbic acid, 1% alpha-tocopherol, and 0.5% ferulic acid protected human skin in vivo from UV-induced damage, specifically erythema and apoptosis. The formulation also suppressed p53 activation and limited thymine dimer mutations, which are associated with skin cancer.26
Waibel et al. conducted a double-blind, prospective, single-center, randomized split-face trial in 2015 to study whether laser-assisted delivery of vitamins C and E and ferulic acid after fractional ablative laser procedures to treat photodamage could enhance wound healing. Fifteen healthy men and women (aged 30-55 years) were treated with the combination formulation on one side of the face and vehicle on the other side within 2 minutes of receiving fractional ablative CO2 laser surgery. They also received daily treatments and evaluations during days 1 through 7 of healing. Edema was found to be diminished on the sides treated with the antioxidant combination, compared with vehicle on day 7, and erythema, on days 3 and 5.27
Other vitamin E combinations
In 2014, Farris et al. found vitamin E to be a key ingredient, along with resveratrol and baicalin, in a nighttime antioxidant formulation that netted a statistically significant improvement in skin rejuvenation, specifically ameliorating fine lines and wrinkles, skin firmness, skin elasticity, skin laxity, hyperpigmentation, radiance, and skin roughness over 3 months, compared with baseline.28
Pereira et al. reported in 2014 that they found that the topical application of polymeric bioadhesive films containing aloe vera and vitamin E acetate appear to be an effective approach to burn treatment.29
A 2015 randomized, controlled, double-blind prospective study in 30 healthy volunteers also indicated that an SPF 30 sunscreen supplemented with an antioxidant combination containing grape seed extract, vitamin E, coenzyme Q10, and vitamin C effectively protected skin against infrared A radiation damage, unlike the use of the SPF 30 product without the antioxidant cocktail.30
Conclusion
Combining vitamin E with other antioxidants appears to enhance its bioactivity and, likely, that of the other interacting antioxidants. The potential therapeutic benefits of vitamin E in preventing and treating skin cancer and photoaging remain an important focus of research. As an ingredient in topical anti-aging skin care preparations, vitamin E displays emollient properties, and is stable, easy to formulate, and relatively inexpensive, making it a popular additive. More controlled trials are necessary to fully clarify the role of vitamin E in treating various dermatoses.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
References
1. J Mol Med (Berl). 1995 Jan;73(1):7-17.
2. Dermatol Surg. 2005 Jul;31(7 Pt 2):805-13.
3. J Cosmet Dermatol. 2004 Jul;3(3):149-55.
4. Skin Pharmacol Appl Skin Physiol. 2001;14 Suppl 1:87-91.
5. Am J Clin Nutr. 1987 Jul;46(1 Suppl):183-6.
6. Nutr Rev. 2012 Sep;70(9):483-90.
7. Skin Pharmacol Physiol. 2005 Jan-Feb;18(1):20-6.
8. Ann Nutr Metab. 2012;61(3):207-12.
9. J Am Acad Dermatol. 2003 Jun;48(6):866-74.
10. Plast Reconstr Surg. 1997 Sep;100(4):973-80.
11. Acta Derm Venereol. 1996 Jul;76(4):264-8.
12. J Invest Dermatol. 1995 Apr;104(4):484-8.
13. Photodermatol Photoimmunol Photomed. 1990 Apr;7(2):56-62.
14. Photodermatol. 1989 Oct;6(5):228-33.
15. Drug Metab Rev. 2000 Aug-Nov;32(3-4):413-20.
16. Clin Dermatol. 2009 Sep-Oct;27(5):469-74.
17. Cancer Prev Res (Phila). 2009 Apr;2(4):394-400.
18. J Dermatolog Treat. 2015 Dec 10:1-5. [Epub ahead of print].
19. Int J Colorectal Dis. 2016 Jul;31(7):1371-2.
20. Skin Pharmacol Appl Skin Physiol. 15:307;2002.
21. Biofactors. 2003;18(1-4):289-97.
22. J Drugs Dermatol. 2008 Jul;7(7 Suppl):s2-6.
23. Can J Physiol Pharmacol. 1993 Sep;71(9):725-31.
24. PLoS One. 2013 May 14;8(5):e63809.
25. J Invest Dermatol. 2005 Oct;125(4):826-32.
26. J Am Acad Dermatol. 2008 Sep;59(3):418-25.
27. Lasers Surg Med. 2016 Mar;48(3):238-44.
28. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
Available in the diet through fresh vegetables (particularly green leafy vegetables), vegetable oils, grains, nuts, seeds, corn, soy, whole wheat flour, margarine, and in some meat and dairy products, vitamin E, or tocopherol, is the primary lipid-soluble antioxidant found in human skin (via sebum), membranes, plasma, and tissues that protects cells from oxidative stress.1-4 Vitamin E is often used to treat minor burns, surgical scars, and other wounds, although the Food and Drug Administration has not approved its use for skin conditions.
In 1938, Karrer, Fritzsche, Ringier, and Salomon became the first to synthesize alpha-tocopherol,5,6 the main biologically active form of vitamin E.7 In the 1940s, vitamin E was labeled a “chain-breaking” antioxidant for its role in hindering the chain reaction induced by free radicals, and it is known to protect cutaneous cell membranes from peroxidation.8 Most topical formulations contain synthetic laboratory-made alpha-tocopherol or one of its many esters or ethers. As an ingredient in skin care agents, significant evidence has been amassed to suggest that topically applied vitamin E confers photoprotective activity against erythema, edema, sun burn cell formation, and other indicators of acute UV-induced damage as well as responses to chronic UVA and UVB exposure, including skin wrinkling and skin cancer.2,9-14 This column will focus on the topical applications of vitamin E.
Topical uses and findings
The lipophilic nature of vitamin E makes it suitable for topical application and percutaneous absorption through the skin.9,15 Vitamin E is generally used in 1%-5% concentrations alpha-tocopherol or tocopherol acetate in over-the-counter products.16 When topically applied, vitamin E has been shown to hydrate the stratum corneum (SC) and improve water-binding capacity.16 It is also considered an effective ingredient for imparting skin protection and treating atopic dermatitis (AD).2
In 2005, Ekanayake-Mudiyanselage et al. studied whether one application of an alpha-tocopherol–enriched rinse-off product could effectively lead to deposition of alpha-tocopherol on the SC in 13 volunteers. The researchers found that the alpha-tocopherol product raised alpha-tocopherol levels in surface lipids, which remained consistent for at least 24 hours, whereas such levels were reduced in the alpha-tocopherol–free vehicle control group. The alpha-tocopherol rinse-off product also significantly inhibited photo-oxidation of squalene.7
A 2009 6-month study in healthy human volunteers with actinic keratoses demonstrated that while topically applied dl-alpha-tocopherol, of which cutaneous levels were significantly increased at the end of the study, did not significantly change already present lesions, alterations in polyamine metabolism revealed that squamous cell carcinogenesis potential was significantly diminished.17
Patrizi et al., in a 2015 randomized, controlled, double-blind, single center study, assessed the safety and efficacy of MD2011001 cream (a nonsteroidal topical cream including vitamin E, epigallocatechin gallate and grape seed procyanidins) versus placebo, in 44 patients with mild to moderate AD in the perioral/periocular area and/or the neck. The researchers noted a significantly more rapid reduction in affected surface area with the test formulation, compared with placebo; the product was found to be well tolerated and safe as well as effective for mild to moderate AD.18
Also that year, Ruiz-Tovar et al. performed a prospective randomized clinical trial in 60 patients, showing that topical vitamin E ointment reduced postoperative pain.19
The vitamin C, vitamin E, ferulic acid combination
Vitamin E is perceived to be more effective when used in combination with other antioxidant ingredients. Some data suggest a cumulative benefit derived from using oral and topical antioxidant products in combination, including vitamins C and E in particular.20-22 Because vitamin C can restore oxidized vitamin E, combining the antioxidants is a stabilizing factor in topical formulations.23,24 Further, ferulic acid has been shown to stabilize both vitamins, with the topical combination yielding photoprotective effects against UVB exposure, including the significant reduction in thymine dimer formation.9,24,25
A small study of nine patients conducted by Murray et al. in 2008 found that a stable topical preparation of 15% l-ascorbic acid, 1% alpha-tocopherol, and 0.5% ferulic acid protected human skin in vivo from UV-induced damage, specifically erythema and apoptosis. The formulation also suppressed p53 activation and limited thymine dimer mutations, which are associated with skin cancer.26
Waibel et al. conducted a double-blind, prospective, single-center, randomized split-face trial in 2015 to study whether laser-assisted delivery of vitamins C and E and ferulic acid after fractional ablative laser procedures to treat photodamage could enhance wound healing. Fifteen healthy men and women (aged 30-55 years) were treated with the combination formulation on one side of the face and vehicle on the other side within 2 minutes of receiving fractional ablative CO2 laser surgery. They also received daily treatments and evaluations during days 1 through 7 of healing. Edema was found to be diminished on the sides treated with the antioxidant combination, compared with vehicle on day 7, and erythema, on days 3 and 5.27
Other vitamin E combinations
In 2014, Farris et al. found vitamin E to be a key ingredient, along with resveratrol and baicalin, in a nighttime antioxidant formulation that netted a statistically significant improvement in skin rejuvenation, specifically ameliorating fine lines and wrinkles, skin firmness, skin elasticity, skin laxity, hyperpigmentation, radiance, and skin roughness over 3 months, compared with baseline.28
Pereira et al. reported in 2014 that they found that the topical application of polymeric bioadhesive films containing aloe vera and vitamin E acetate appear to be an effective approach to burn treatment.29
A 2015 randomized, controlled, double-blind prospective study in 30 healthy volunteers also indicated that an SPF 30 sunscreen supplemented with an antioxidant combination containing grape seed extract, vitamin E, coenzyme Q10, and vitamin C effectively protected skin against infrared A radiation damage, unlike the use of the SPF 30 product without the antioxidant cocktail.30
Conclusion
Combining vitamin E with other antioxidants appears to enhance its bioactivity and, likely, that of the other interacting antioxidants. The potential therapeutic benefits of vitamin E in preventing and treating skin cancer and photoaging remain an important focus of research. As an ingredient in topical anti-aging skin care preparations, vitamin E displays emollient properties, and is stable, easy to formulate, and relatively inexpensive, making it a popular additive. More controlled trials are necessary to fully clarify the role of vitamin E in treating various dermatoses.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. She also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
References
1. J Mol Med (Berl). 1995 Jan;73(1):7-17.
2. Dermatol Surg. 2005 Jul;31(7 Pt 2):805-13.
3. J Cosmet Dermatol. 2004 Jul;3(3):149-55.
4. Skin Pharmacol Appl Skin Physiol. 2001;14 Suppl 1:87-91.
5. Am J Clin Nutr. 1987 Jul;46(1 Suppl):183-6.
6. Nutr Rev. 2012 Sep;70(9):483-90.
7. Skin Pharmacol Physiol. 2005 Jan-Feb;18(1):20-6.
8. Ann Nutr Metab. 2012;61(3):207-12.
9. J Am Acad Dermatol. 2003 Jun;48(6):866-74.
10. Plast Reconstr Surg. 1997 Sep;100(4):973-80.
11. Acta Derm Venereol. 1996 Jul;76(4):264-8.
12. J Invest Dermatol. 1995 Apr;104(4):484-8.
13. Photodermatol Photoimmunol Photomed. 1990 Apr;7(2):56-62.
14. Photodermatol. 1989 Oct;6(5):228-33.
15. Drug Metab Rev. 2000 Aug-Nov;32(3-4):413-20.
16. Clin Dermatol. 2009 Sep-Oct;27(5):469-74.
17. Cancer Prev Res (Phila). 2009 Apr;2(4):394-400.
18. J Dermatolog Treat. 2015 Dec 10:1-5. [Epub ahead of print].
19. Int J Colorectal Dis. 2016 Jul;31(7):1371-2.
20. Skin Pharmacol Appl Skin Physiol. 15:307;2002.
21. Biofactors. 2003;18(1-4):289-97.
22. J Drugs Dermatol. 2008 Jul;7(7 Suppl):s2-6.
23. Can J Physiol Pharmacol. 1993 Sep;71(9):725-31.
24. PLoS One. 2013 May 14;8(5):e63809.
25. J Invest Dermatol. 2005 Oct;125(4):826-32.
26. J Am Acad Dermatol. 2008 Sep;59(3):418-25.
27. Lasers Surg Med. 2016 Mar;48(3):238-44.
28. J Drugs Dermatol. 2014 Dec;13(12):1467-72.
• Copiously available through the diet, this main lipid-soluble antioxidant in human skin is used to treat some dermatoses but is not approved by the Food and Drug Administration for skin conditions.
• Its lipophilic nature renders it suitable for topical application and percutaneous absorption.
• Topical uses include skin hydration and treatment of atopic dermatitis.
• It is thought to be most effective when used with other antioxidant ingredients, particularly vitamin C and ferulic acid, with the combination yielding photoprotective activity.