User login
The male biological clock – How to tell the time
For decades, we have recognized the age-related natural decline in female fecundity (the ability to reproduce) after the age of 30 (Maturitas 1988;[Suppl]1:15-22). Advanced maternal age (AMA) has also been demonstrated to increase miscarriage and pregnancies with chromosomal abnormalities, presumably from the increased rate of oocyte aneuploidy. There has been a sixfold increase in the rate of first birth in women aged 35-39 years (NCHS Data Brief 2014;152:1-8). Consequently, over the last decade, women, often before they reach AMA, have turned to elective oocyte cryopreservation for fertility preservation.
Ovarian aging
Ovarian aging occurs through the decline in quality and quantity of oocytes. The former is a reflection of the woman’s chronologic age. Markers of female ovarian aging have been utilized, for the past 3 decades, most commonly by basal follicle stimulating hormone. Currently, to assess the quantity of ovarian follicles, antimüllerian hormone (AMH) and transvaginal ultrasound for ovarian antral follicle count (AFC) are the most accurate indicators (J Clin Endocrinol Metab 2004:89:2977-81). While ovarian age testing, particularly AMH, has been widely used to assess a woman’s “fertility potential,” it does not reflect her natural fecundity. In a prospective cohort study, AMH levels (ng/mL) divided into < 0.7, 0.7-8.4, and > 8.4, did not affect natural conception in women aged 30-44 who were divided into the categories of <35, 35-37, or 38-44 years (JAMA 2017;318:1367-76). Although AMH does reduce success with IVF, its main value is the inverse correlation when prescribing gonadotropin dosage for controlled ovarian stimulation.
Despite the familiarity with ovarian aging effects on fertility, the male biological clock remains less studied and understood. Over the last 4 decades, paternal age has increased an average of 3.5 years presumably due to delayed child rearing from professional or personal reasons, improved contraception as well as increased divorce, remarriage, and life expectancy (Hum Reprod. 2017;32:2110-6). Nevertheless, we have little data to definitively counsel men on the effects of advanced paternal age (APA) and no consensus on an actual defined age of designation. This month’s article will summarize the current literature on male age and its impact on fertility.
Testicular aging
Men older than 45 years require approximately five times longer to achieve a pregnancy as men less than 25 after adjustment for female age (Fertil Steril. 2003;79:1520-7). The most likely parameter to assess male fertility, other than pregnancy rates, would be the sperm. Sperm counts, beginning at age 41, may decline but concentrations have been shown to increase in older men apparently because of declining semen volume (Ageing Res Rev. 2015;19:22-33). Sperm motility, but not morphology, also declines while genetic alterations of sperm increase with age. The issue of chromosomal abnormalities in sperm from men of advanced age appears to be similar to that in the oocytes of women with AMA. Consequently, both sexes may contribute to embryo aneuploidy resulting in declining fertility and increasing miscarriage.
For all ages, studies have suggested that elevated male body mass index as well as alcohol consumption and cigarette smoking, including e-cigarettes, can lead to impaired sperm production (Hum Reprod Update 2013;19:221-31).
Fertility treatment outcomes
A mainstay of fertility treatment, particularly in men with mild to moderate impairments in semen parameters, is ovulation induction with intrauterine insemination. Male age has been shown to be a significant indicator for pregnancy rates, including those with normal semen parameters (J Obstet Gynaecol. 2011;31:420-3). Men above age 45 contributed to lower pregnancy rates and higher miscarriages during IUI treatment cycles (Reprod BioMed Online 2008;17:392-7).
During IVF cycles, the sperm of men with APA often undergo ICSI (intracytoplasmic sperm injection) due to higher fertilization rates compared with standard insemination. However, APA sperm appear to have lower fertilization rates and decreased embryo development to the blastocyst stage during cycles using donor oocytes, although pregnancy outcomes are inconsistent (Trans Androl Urol. 2019;8[Suppl 1]:S22-S30; Fertil Steril. 2008;90:97-103).
Perinatal and children’s health
The offspring from APA men appear to have higher rates of stillbirth, low birth weight, and preterm birth, as well as birth defects. Men older than 40-45 years have twice the risk of an autistic child and three times the risk of schizophrenia in their offspring (Transl Psychiatry 2017;7:e1019; Am J Psychiatry 2002;159:1528-33).
Conclusions
Most of the literature supports negative effects on sperm and reproduction from men with APA. The challenge in deciphering the true role of APA on fertility is that the partner is often of AMA. A consideration to avoid this effect would be sperm cryopreservation at a younger age, similar to the common trend among women. Preimplantation genetic testing of embryos from men with APA is also a potential option to reduce miscarriage and avoid a chromosomally abnormal pregnancy. Ethicists have pondered the impact of APA on parenthood and the detrimental effect of early paternal death on the child. Nevertheless, the effect of APA in reproduction is a vital area to study with the same fervor as AMA (Fertil Steril 2009;92:1772-5).
Dr. Trolice is director of Fertility CARE – The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando. He has no conflicts. Email him at [email protected].
For decades, we have recognized the age-related natural decline in female fecundity (the ability to reproduce) after the age of 30 (Maturitas 1988;[Suppl]1:15-22). Advanced maternal age (AMA) has also been demonstrated to increase miscarriage and pregnancies with chromosomal abnormalities, presumably from the increased rate of oocyte aneuploidy. There has been a sixfold increase in the rate of first birth in women aged 35-39 years (NCHS Data Brief 2014;152:1-8). Consequently, over the last decade, women, often before they reach AMA, have turned to elective oocyte cryopreservation for fertility preservation.
Ovarian aging
Ovarian aging occurs through the decline in quality and quantity of oocytes. The former is a reflection of the woman’s chronologic age. Markers of female ovarian aging have been utilized, for the past 3 decades, most commonly by basal follicle stimulating hormone. Currently, to assess the quantity of ovarian follicles, antimüllerian hormone (AMH) and transvaginal ultrasound for ovarian antral follicle count (AFC) are the most accurate indicators (J Clin Endocrinol Metab 2004:89:2977-81). While ovarian age testing, particularly AMH, has been widely used to assess a woman’s “fertility potential,” it does not reflect her natural fecundity. In a prospective cohort study, AMH levels (ng/mL) divided into < 0.7, 0.7-8.4, and > 8.4, did not affect natural conception in women aged 30-44 who were divided into the categories of <35, 35-37, or 38-44 years (JAMA 2017;318:1367-76). Although AMH does reduce success with IVF, its main value is the inverse correlation when prescribing gonadotropin dosage for controlled ovarian stimulation.
Despite the familiarity with ovarian aging effects on fertility, the male biological clock remains less studied and understood. Over the last 4 decades, paternal age has increased an average of 3.5 years presumably due to delayed child rearing from professional or personal reasons, improved contraception as well as increased divorce, remarriage, and life expectancy (Hum Reprod. 2017;32:2110-6). Nevertheless, we have little data to definitively counsel men on the effects of advanced paternal age (APA) and no consensus on an actual defined age of designation. This month’s article will summarize the current literature on male age and its impact on fertility.
Testicular aging
Men older than 45 years require approximately five times longer to achieve a pregnancy as men less than 25 after adjustment for female age (Fertil Steril. 2003;79:1520-7). The most likely parameter to assess male fertility, other than pregnancy rates, would be the sperm. Sperm counts, beginning at age 41, may decline but concentrations have been shown to increase in older men apparently because of declining semen volume (Ageing Res Rev. 2015;19:22-33). Sperm motility, but not morphology, also declines while genetic alterations of sperm increase with age. The issue of chromosomal abnormalities in sperm from men of advanced age appears to be similar to that in the oocytes of women with AMA. Consequently, both sexes may contribute to embryo aneuploidy resulting in declining fertility and increasing miscarriage.
For all ages, studies have suggested that elevated male body mass index as well as alcohol consumption and cigarette smoking, including e-cigarettes, can lead to impaired sperm production (Hum Reprod Update 2013;19:221-31).
Fertility treatment outcomes
A mainstay of fertility treatment, particularly in men with mild to moderate impairments in semen parameters, is ovulation induction with intrauterine insemination. Male age has been shown to be a significant indicator for pregnancy rates, including those with normal semen parameters (J Obstet Gynaecol. 2011;31:420-3). Men above age 45 contributed to lower pregnancy rates and higher miscarriages during IUI treatment cycles (Reprod BioMed Online 2008;17:392-7).
During IVF cycles, the sperm of men with APA often undergo ICSI (intracytoplasmic sperm injection) due to higher fertilization rates compared with standard insemination. However, APA sperm appear to have lower fertilization rates and decreased embryo development to the blastocyst stage during cycles using donor oocytes, although pregnancy outcomes are inconsistent (Trans Androl Urol. 2019;8[Suppl 1]:S22-S30; Fertil Steril. 2008;90:97-103).
Perinatal and children’s health
The offspring from APA men appear to have higher rates of stillbirth, low birth weight, and preterm birth, as well as birth defects. Men older than 40-45 years have twice the risk of an autistic child and three times the risk of schizophrenia in their offspring (Transl Psychiatry 2017;7:e1019; Am J Psychiatry 2002;159:1528-33).
Conclusions
Most of the literature supports negative effects on sperm and reproduction from men with APA. The challenge in deciphering the true role of APA on fertility is that the partner is often of AMA. A consideration to avoid this effect would be sperm cryopreservation at a younger age, similar to the common trend among women. Preimplantation genetic testing of embryos from men with APA is also a potential option to reduce miscarriage and avoid a chromosomally abnormal pregnancy. Ethicists have pondered the impact of APA on parenthood and the detrimental effect of early paternal death on the child. Nevertheless, the effect of APA in reproduction is a vital area to study with the same fervor as AMA (Fertil Steril 2009;92:1772-5).
Dr. Trolice is director of Fertility CARE – The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando. He has no conflicts. Email him at [email protected].
For decades, we have recognized the age-related natural decline in female fecundity (the ability to reproduce) after the age of 30 (Maturitas 1988;[Suppl]1:15-22). Advanced maternal age (AMA) has also been demonstrated to increase miscarriage and pregnancies with chromosomal abnormalities, presumably from the increased rate of oocyte aneuploidy. There has been a sixfold increase in the rate of first birth in women aged 35-39 years (NCHS Data Brief 2014;152:1-8). Consequently, over the last decade, women, often before they reach AMA, have turned to elective oocyte cryopreservation for fertility preservation.
Ovarian aging
Ovarian aging occurs through the decline in quality and quantity of oocytes. The former is a reflection of the woman’s chronologic age. Markers of female ovarian aging have been utilized, for the past 3 decades, most commonly by basal follicle stimulating hormone. Currently, to assess the quantity of ovarian follicles, antimüllerian hormone (AMH) and transvaginal ultrasound for ovarian antral follicle count (AFC) are the most accurate indicators (J Clin Endocrinol Metab 2004:89:2977-81). While ovarian age testing, particularly AMH, has been widely used to assess a woman’s “fertility potential,” it does not reflect her natural fecundity. In a prospective cohort study, AMH levels (ng/mL) divided into < 0.7, 0.7-8.4, and > 8.4, did not affect natural conception in women aged 30-44 who were divided into the categories of <35, 35-37, or 38-44 years (JAMA 2017;318:1367-76). Although AMH does reduce success with IVF, its main value is the inverse correlation when prescribing gonadotropin dosage for controlled ovarian stimulation.
Despite the familiarity with ovarian aging effects on fertility, the male biological clock remains less studied and understood. Over the last 4 decades, paternal age has increased an average of 3.5 years presumably due to delayed child rearing from professional or personal reasons, improved contraception as well as increased divorce, remarriage, and life expectancy (Hum Reprod. 2017;32:2110-6). Nevertheless, we have little data to definitively counsel men on the effects of advanced paternal age (APA) and no consensus on an actual defined age of designation. This month’s article will summarize the current literature on male age and its impact on fertility.
Testicular aging
Men older than 45 years require approximately five times longer to achieve a pregnancy as men less than 25 after adjustment for female age (Fertil Steril. 2003;79:1520-7). The most likely parameter to assess male fertility, other than pregnancy rates, would be the sperm. Sperm counts, beginning at age 41, may decline but concentrations have been shown to increase in older men apparently because of declining semen volume (Ageing Res Rev. 2015;19:22-33). Sperm motility, but not morphology, also declines while genetic alterations of sperm increase with age. The issue of chromosomal abnormalities in sperm from men of advanced age appears to be similar to that in the oocytes of women with AMA. Consequently, both sexes may contribute to embryo aneuploidy resulting in declining fertility and increasing miscarriage.
For all ages, studies have suggested that elevated male body mass index as well as alcohol consumption and cigarette smoking, including e-cigarettes, can lead to impaired sperm production (Hum Reprod Update 2013;19:221-31).
Fertility treatment outcomes
A mainstay of fertility treatment, particularly in men with mild to moderate impairments in semen parameters, is ovulation induction with intrauterine insemination. Male age has been shown to be a significant indicator for pregnancy rates, including those with normal semen parameters (J Obstet Gynaecol. 2011;31:420-3). Men above age 45 contributed to lower pregnancy rates and higher miscarriages during IUI treatment cycles (Reprod BioMed Online 2008;17:392-7).
During IVF cycles, the sperm of men with APA often undergo ICSI (intracytoplasmic sperm injection) due to higher fertilization rates compared with standard insemination. However, APA sperm appear to have lower fertilization rates and decreased embryo development to the blastocyst stage during cycles using donor oocytes, although pregnancy outcomes are inconsistent (Trans Androl Urol. 2019;8[Suppl 1]:S22-S30; Fertil Steril. 2008;90:97-103).
Perinatal and children’s health
The offspring from APA men appear to have higher rates of stillbirth, low birth weight, and preterm birth, as well as birth defects. Men older than 40-45 years have twice the risk of an autistic child and three times the risk of schizophrenia in their offspring (Transl Psychiatry 2017;7:e1019; Am J Psychiatry 2002;159:1528-33).
Conclusions
Most of the literature supports negative effects on sperm and reproduction from men with APA. The challenge in deciphering the true role of APA on fertility is that the partner is often of AMA. A consideration to avoid this effect would be sperm cryopreservation at a younger age, similar to the common trend among women. Preimplantation genetic testing of embryos from men with APA is also a potential option to reduce miscarriage and avoid a chromosomally abnormal pregnancy. Ethicists have pondered the impact of APA on parenthood and the detrimental effect of early paternal death on the child. Nevertheless, the effect of APA in reproduction is a vital area to study with the same fervor as AMA (Fertil Steril 2009;92:1772-5).
Dr. Trolice is director of Fertility CARE – The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando. He has no conflicts. Email him at [email protected].
USPSTF statement on aspirin: poor messaging at best
: “The USPSTF concludes with moderate certainty that initiating aspirin use for the primary prevention of CVD events in adults age 60 years or older has no net benefit.” I take no issue with the data and appreciate the efforts of the researchers, but at a minimum the public statement is incomplete. At most, it’s dangerously poor messaging.
As physicians, we understand how best to apply this information, but most laypeople, some at significant cardiovascular risk, closed their medicine cabinets this morning and left their aspirin bottle unopened on the shelf. Some of these patients have never spent an hour in the hospital for cardiac-related issues, but they have mitigated their risk for myocardial infarction by purposely poisoning their platelets daily with 81 mg of aspirin. And they should continue to do so.
Don’t forget the calcium score
Take, for instance, my patient Jack, who is typical of many patients I’ve seen throughout the years. Jack is 68 years old and has never had a cardiac event or a gastrointestinal bleed. His daily routine includes a walk, a statin, and a baby aspirin because his CT coronary artery calcium (CAC) score was 10,000 at age 58.
He first visited me 10 years ago because his father died of a myocardial infarction in his late 50s. Jack’s left ventricular ejection fraction is normal and his stress ECG shows 1-mm ST-segment depression at 8 minutes on a Bruce protocol stress test, without angina. Because Jack is well-educated and keeps up with the latest cardiology recommendations, he is precisely the type of patient who may be harmed by this new USPSTF statement by stopping his aspirin.
In October 2020, an analysis from the DALLAS Heart Study showed that persons with a CAC score greater than 100 had a higher cumulative incidence of bleeding and of atherosclerotic cardiovascular disease (ASCVD) events compared with those with no coronary calcium. After adjustment for clinical risk factors, the association between CAC and bleeding was attenuated and no longer statistically significant, whereas the relationship between CAC and ASCVD remained.
I asked one of the investigators, Amit Khera, MD, MSc, from UT Southwestern Medical Center, about the latest recommendations. He emphasized that both the American College of Cardiology/American Heart Association prevention guidelines and the USPSTF statement say that aspirin could still be considered among patients who are at higher risk for cardiovascular events. The USPSTF delineated this as a 10-year ASCVD risk greater than 10%.
Dr. Khera, who was an author of the 2019 guidelines, explained that the guideline committee purposely did not make specific recommendations as to what demarcated higher risk because the data were not clear at that time. Since then, a couple of papers, including the Dallas Heart Study analysis published in JAMA Cardiology, showed that patients at low bleeding risk with a calcium score above 100 may get a net benefit from aspirin. “Thus, in my patients who have a high calcium score and low bleeding risk, I do discuss the option to start or continue aspirin,” he said.
One size does not fit all
I watched ABC World News Tonight on Tuesday, October 12, and was immediately troubled about the coverage of the USPSTF statement. With viewership for the “Big Three” networks in the millions, the message to discontinue aspirin may have unintended consequences for many at-risk patients. The blood-thinning effects of a single dose of aspirin last about 10 days; it will be interesting to see if the rates of myocardial infarction increase over time. This could have been avoided with a better-worded statement – I’m concerned that the lack of nuance could spell big trouble for some.
In JAMA Cardiology, Dr. Khera and colleagues wrote that, “Aspirin use is not a one-size-fits-all therapy.” All physicians likely agree with that opinion. The USPSTF statement should have included the point that if you have a high CT coronary artery calcium score and a low bleeding risk, aspirin still fits very well even if you haven’t experienced a cardiac event. At a minimum, the USPSTF statement should have included the suggestion for patients to consult their physician for advice before discontinuing aspirin therapy.
I hope patients like Jack get the right message.
Melissa Walton-Shirley, MD, is a native Kentuckian who retired from full-time invasive cardiology.
A version of this article first appeared on Medscape.com.
: “The USPSTF concludes with moderate certainty that initiating aspirin use for the primary prevention of CVD events in adults age 60 years or older has no net benefit.” I take no issue with the data and appreciate the efforts of the researchers, but at a minimum the public statement is incomplete. At most, it’s dangerously poor messaging.
As physicians, we understand how best to apply this information, but most laypeople, some at significant cardiovascular risk, closed their medicine cabinets this morning and left their aspirin bottle unopened on the shelf. Some of these patients have never spent an hour in the hospital for cardiac-related issues, but they have mitigated their risk for myocardial infarction by purposely poisoning their platelets daily with 81 mg of aspirin. And they should continue to do so.
Don’t forget the calcium score
Take, for instance, my patient Jack, who is typical of many patients I’ve seen throughout the years. Jack is 68 years old and has never had a cardiac event or a gastrointestinal bleed. His daily routine includes a walk, a statin, and a baby aspirin because his CT coronary artery calcium (CAC) score was 10,000 at age 58.
He first visited me 10 years ago because his father died of a myocardial infarction in his late 50s. Jack’s left ventricular ejection fraction is normal and his stress ECG shows 1-mm ST-segment depression at 8 minutes on a Bruce protocol stress test, without angina. Because Jack is well-educated and keeps up with the latest cardiology recommendations, he is precisely the type of patient who may be harmed by this new USPSTF statement by stopping his aspirin.
In October 2020, an analysis from the DALLAS Heart Study showed that persons with a CAC score greater than 100 had a higher cumulative incidence of bleeding and of atherosclerotic cardiovascular disease (ASCVD) events compared with those with no coronary calcium. After adjustment for clinical risk factors, the association between CAC and bleeding was attenuated and no longer statistically significant, whereas the relationship between CAC and ASCVD remained.
I asked one of the investigators, Amit Khera, MD, MSc, from UT Southwestern Medical Center, about the latest recommendations. He emphasized that both the American College of Cardiology/American Heart Association prevention guidelines and the USPSTF statement say that aspirin could still be considered among patients who are at higher risk for cardiovascular events. The USPSTF delineated this as a 10-year ASCVD risk greater than 10%.
Dr. Khera, who was an author of the 2019 guidelines, explained that the guideline committee purposely did not make specific recommendations as to what demarcated higher risk because the data were not clear at that time. Since then, a couple of papers, including the Dallas Heart Study analysis published in JAMA Cardiology, showed that patients at low bleeding risk with a calcium score above 100 may get a net benefit from aspirin. “Thus, in my patients who have a high calcium score and low bleeding risk, I do discuss the option to start or continue aspirin,” he said.
One size does not fit all
I watched ABC World News Tonight on Tuesday, October 12, and was immediately troubled about the coverage of the USPSTF statement. With viewership for the “Big Three” networks in the millions, the message to discontinue aspirin may have unintended consequences for many at-risk patients. The blood-thinning effects of a single dose of aspirin last about 10 days; it will be interesting to see if the rates of myocardial infarction increase over time. This could have been avoided with a better-worded statement – I’m concerned that the lack of nuance could spell big trouble for some.
In JAMA Cardiology, Dr. Khera and colleagues wrote that, “Aspirin use is not a one-size-fits-all therapy.” All physicians likely agree with that opinion. The USPSTF statement should have included the point that if you have a high CT coronary artery calcium score and a low bleeding risk, aspirin still fits very well even if you haven’t experienced a cardiac event. At a minimum, the USPSTF statement should have included the suggestion for patients to consult their physician for advice before discontinuing aspirin therapy.
I hope patients like Jack get the right message.
Melissa Walton-Shirley, MD, is a native Kentuckian who retired from full-time invasive cardiology.
A version of this article first appeared on Medscape.com.
: “The USPSTF concludes with moderate certainty that initiating aspirin use for the primary prevention of CVD events in adults age 60 years or older has no net benefit.” I take no issue with the data and appreciate the efforts of the researchers, but at a minimum the public statement is incomplete. At most, it’s dangerously poor messaging.
As physicians, we understand how best to apply this information, but most laypeople, some at significant cardiovascular risk, closed their medicine cabinets this morning and left their aspirin bottle unopened on the shelf. Some of these patients have never spent an hour in the hospital for cardiac-related issues, but they have mitigated their risk for myocardial infarction by purposely poisoning their platelets daily with 81 mg of aspirin. And they should continue to do so.
Don’t forget the calcium score
Take, for instance, my patient Jack, who is typical of many patients I’ve seen throughout the years. Jack is 68 years old and has never had a cardiac event or a gastrointestinal bleed. His daily routine includes a walk, a statin, and a baby aspirin because his CT coronary artery calcium (CAC) score was 10,000 at age 58.
He first visited me 10 years ago because his father died of a myocardial infarction in his late 50s. Jack’s left ventricular ejection fraction is normal and his stress ECG shows 1-mm ST-segment depression at 8 minutes on a Bruce protocol stress test, without angina. Because Jack is well-educated and keeps up with the latest cardiology recommendations, he is precisely the type of patient who may be harmed by this new USPSTF statement by stopping his aspirin.
In October 2020, an analysis from the DALLAS Heart Study showed that persons with a CAC score greater than 100 had a higher cumulative incidence of bleeding and of atherosclerotic cardiovascular disease (ASCVD) events compared with those with no coronary calcium. After adjustment for clinical risk factors, the association between CAC and bleeding was attenuated and no longer statistically significant, whereas the relationship between CAC and ASCVD remained.
I asked one of the investigators, Amit Khera, MD, MSc, from UT Southwestern Medical Center, about the latest recommendations. He emphasized that both the American College of Cardiology/American Heart Association prevention guidelines and the USPSTF statement say that aspirin could still be considered among patients who are at higher risk for cardiovascular events. The USPSTF delineated this as a 10-year ASCVD risk greater than 10%.
Dr. Khera, who was an author of the 2019 guidelines, explained that the guideline committee purposely did not make specific recommendations as to what demarcated higher risk because the data were not clear at that time. Since then, a couple of papers, including the Dallas Heart Study analysis published in JAMA Cardiology, showed that patients at low bleeding risk with a calcium score above 100 may get a net benefit from aspirin. “Thus, in my patients who have a high calcium score and low bleeding risk, I do discuss the option to start or continue aspirin,” he said.
One size does not fit all
I watched ABC World News Tonight on Tuesday, October 12, and was immediately troubled about the coverage of the USPSTF statement. With viewership for the “Big Three” networks in the millions, the message to discontinue aspirin may have unintended consequences for many at-risk patients. The blood-thinning effects of a single dose of aspirin last about 10 days; it will be interesting to see if the rates of myocardial infarction increase over time. This could have been avoided with a better-worded statement – I’m concerned that the lack of nuance could spell big trouble for some.
In JAMA Cardiology, Dr. Khera and colleagues wrote that, “Aspirin use is not a one-size-fits-all therapy.” All physicians likely agree with that opinion. The USPSTF statement should have included the point that if you have a high CT coronary artery calcium score and a low bleeding risk, aspirin still fits very well even if you haven’t experienced a cardiac event. At a minimum, the USPSTF statement should have included the suggestion for patients to consult their physician for advice before discontinuing aspirin therapy.
I hope patients like Jack get the right message.
Melissa Walton-Shirley, MD, is a native Kentuckian who retired from full-time invasive cardiology.
A version of this article first appeared on Medscape.com.
Guidelines for dementia and age-related cognitive changes
It is estimated that by the year 2060, 13.9 million Americans over the age of 65 will be diagnosed with dementia. Few good treatments are currently available.
Earlier this year, the American Psychological Association (APA) Task Force issued clinical guidelines “for the Evaluation of Dementia and Age-Related Cognitive Change.” While these 16 guidelines are aimed at psychologists, primary care doctors are often the first ones to evaluate a patient who may have dementia. As a family physician, I find having these guidelines especially helpful.
Neuropsychiatric testing and defining severity and type
This new guidance places emphasis on neuropsychiatric testing and defining the severity and type of dementia present.
Over the past 2 decades, diagnoses of mild neurocognitive disorders have increased, and this, in part, is due to diagnosing these problems earlier and with greater precision. It is also important to know that biomarkers are being increasingly researched, and it is imperative that we stay current with this research.
Cognitive decline may also occur with the coexistence of other mental health disorders, such as depression, so it is important that we screen for these as well. This is often difficult given the behavioral changes that can arise in dementia, but, as primary care doctors, we must differentiate these to treat our patients appropriately.
Informed consent
Informed consent can become an issue with patients with dementia. It must be assessed whether the patient has the capacity to make an informed decision and can competently communicate that decision.
The diagnosis of dementia alone does not preclude a patient from giving informed consent. A patient’s mental capacity must be determined, and if they are not capable of making an informed decision, the person legally responsible for giving informed consent on behalf of the patient must be identified.
Patients with dementia often have other medical comorbidities and take several medications. It is imperative to keep accurate medical records and medication lists. Sometimes, patients with dementia cannot provide this information. If that is the case, every attempt should be made to obtain records from every possible source.
Cultural competence
The guidelines also stress that there may be cultural differences when applying neuropsychiatric tests. It is our duty to maintain cultural competence and understand these differences. We all need to work to ensure we control our biases, and it is suggested that we review relevant evidence-based literature.
While ageism is common in our society, it shouldn’t be in our practices. For these reasons, outreach in at-risk populations is very important.
Pertinent data
The guidelines also suggest obtaining all possible information in our evaluation, especially when the patient is unable to give it to us.
Often, as primary care physicians, we refer these patients to other providers, and we should be providing all pertinent data to those we are referring these patients to. If all information is not available at the time of evaluation, follow-up visits should be scheduled.
If possible, family members should be present at the time of visit. They often provide valuable information regarding the extent and progression of the decline. Also, they know how the patient is functioning in the home setting and how much assistance they need with activities of daily living.
Caretaker support
Another important factor to consider is caretaker burnout. Caretakers are often under a lot of stress and have high rates of depression. It is important to provide them with education and support, as well as resources that may be available to them. For some, accepting the diagnosis that their loved one has dementia may be a struggle.
As doctors treating dementia patients, we need to know the resources that are available to assist dementia patients and their families. There are many local organizations that can help.
Also, research into dementia is ongoing and we need to stay current. The diagnosis of dementia should be made as early as possible using appropriate screening tools. The sooner the diagnosis is made, the quicker interventions can be started and the family members, as well as the patient, can come to accept the diagnosis.
As the population ages, we can expect the demands of dementia to rise as well. Primary care doctors are in a unique position to diagnose dementia once it starts to appear.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, N.J. You can contact her at [email protected].
It is estimated that by the year 2060, 13.9 million Americans over the age of 65 will be diagnosed with dementia. Few good treatments are currently available.
Earlier this year, the American Psychological Association (APA) Task Force issued clinical guidelines “for the Evaluation of Dementia and Age-Related Cognitive Change.” While these 16 guidelines are aimed at psychologists, primary care doctors are often the first ones to evaluate a patient who may have dementia. As a family physician, I find having these guidelines especially helpful.
Neuropsychiatric testing and defining severity and type
This new guidance places emphasis on neuropsychiatric testing and defining the severity and type of dementia present.
Over the past 2 decades, diagnoses of mild neurocognitive disorders have increased, and this, in part, is due to diagnosing these problems earlier and with greater precision. It is also important to know that biomarkers are being increasingly researched, and it is imperative that we stay current with this research.
Cognitive decline may also occur with the coexistence of other mental health disorders, such as depression, so it is important that we screen for these as well. This is often difficult given the behavioral changes that can arise in dementia, but, as primary care doctors, we must differentiate these to treat our patients appropriately.
Informed consent
Informed consent can become an issue with patients with dementia. It must be assessed whether the patient has the capacity to make an informed decision and can competently communicate that decision.
The diagnosis of dementia alone does not preclude a patient from giving informed consent. A patient’s mental capacity must be determined, and if they are not capable of making an informed decision, the person legally responsible for giving informed consent on behalf of the patient must be identified.
Patients with dementia often have other medical comorbidities and take several medications. It is imperative to keep accurate medical records and medication lists. Sometimes, patients with dementia cannot provide this information. If that is the case, every attempt should be made to obtain records from every possible source.
Cultural competence
The guidelines also stress that there may be cultural differences when applying neuropsychiatric tests. It is our duty to maintain cultural competence and understand these differences. We all need to work to ensure we control our biases, and it is suggested that we review relevant evidence-based literature.
While ageism is common in our society, it shouldn’t be in our practices. For these reasons, outreach in at-risk populations is very important.
Pertinent data
The guidelines also suggest obtaining all possible information in our evaluation, especially when the patient is unable to give it to us.
Often, as primary care physicians, we refer these patients to other providers, and we should be providing all pertinent data to those we are referring these patients to. If all information is not available at the time of evaluation, follow-up visits should be scheduled.
If possible, family members should be present at the time of visit. They often provide valuable information regarding the extent and progression of the decline. Also, they know how the patient is functioning in the home setting and how much assistance they need with activities of daily living.
Caretaker support
Another important factor to consider is caretaker burnout. Caretakers are often under a lot of stress and have high rates of depression. It is important to provide them with education and support, as well as resources that may be available to them. For some, accepting the diagnosis that their loved one has dementia may be a struggle.
As doctors treating dementia patients, we need to know the resources that are available to assist dementia patients and their families. There are many local organizations that can help.
Also, research into dementia is ongoing and we need to stay current. The diagnosis of dementia should be made as early as possible using appropriate screening tools. The sooner the diagnosis is made, the quicker interventions can be started and the family members, as well as the patient, can come to accept the diagnosis.
As the population ages, we can expect the demands of dementia to rise as well. Primary care doctors are in a unique position to diagnose dementia once it starts to appear.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, N.J. You can contact her at [email protected].
It is estimated that by the year 2060, 13.9 million Americans over the age of 65 will be diagnosed with dementia. Few good treatments are currently available.
Earlier this year, the American Psychological Association (APA) Task Force issued clinical guidelines “for the Evaluation of Dementia and Age-Related Cognitive Change.” While these 16 guidelines are aimed at psychologists, primary care doctors are often the first ones to evaluate a patient who may have dementia. As a family physician, I find having these guidelines especially helpful.
Neuropsychiatric testing and defining severity and type
This new guidance places emphasis on neuropsychiatric testing and defining the severity and type of dementia present.
Over the past 2 decades, diagnoses of mild neurocognitive disorders have increased, and this, in part, is due to diagnosing these problems earlier and with greater precision. It is also important to know that biomarkers are being increasingly researched, and it is imperative that we stay current with this research.
Cognitive decline may also occur with the coexistence of other mental health disorders, such as depression, so it is important that we screen for these as well. This is often difficult given the behavioral changes that can arise in dementia, but, as primary care doctors, we must differentiate these to treat our patients appropriately.
Informed consent
Informed consent can become an issue with patients with dementia. It must be assessed whether the patient has the capacity to make an informed decision and can competently communicate that decision.
The diagnosis of dementia alone does not preclude a patient from giving informed consent. A patient’s mental capacity must be determined, and if they are not capable of making an informed decision, the person legally responsible for giving informed consent on behalf of the patient must be identified.
Patients with dementia often have other medical comorbidities and take several medications. It is imperative to keep accurate medical records and medication lists. Sometimes, patients with dementia cannot provide this information. If that is the case, every attempt should be made to obtain records from every possible source.
Cultural competence
The guidelines also stress that there may be cultural differences when applying neuropsychiatric tests. It is our duty to maintain cultural competence and understand these differences. We all need to work to ensure we control our biases, and it is suggested that we review relevant evidence-based literature.
While ageism is common in our society, it shouldn’t be in our practices. For these reasons, outreach in at-risk populations is very important.
Pertinent data
The guidelines also suggest obtaining all possible information in our evaluation, especially when the patient is unable to give it to us.
Often, as primary care physicians, we refer these patients to other providers, and we should be providing all pertinent data to those we are referring these patients to. If all information is not available at the time of evaluation, follow-up visits should be scheduled.
If possible, family members should be present at the time of visit. They often provide valuable information regarding the extent and progression of the decline. Also, they know how the patient is functioning in the home setting and how much assistance they need with activities of daily living.
Caretaker support
Another important factor to consider is caretaker burnout. Caretakers are often under a lot of stress and have high rates of depression. It is important to provide them with education and support, as well as resources that may be available to them. For some, accepting the diagnosis that their loved one has dementia may be a struggle.
As doctors treating dementia patients, we need to know the resources that are available to assist dementia patients and their families. There are many local organizations that can help.
Also, research into dementia is ongoing and we need to stay current. The diagnosis of dementia should be made as early as possible using appropriate screening tools. The sooner the diagnosis is made, the quicker interventions can be started and the family members, as well as the patient, can come to accept the diagnosis.
As the population ages, we can expect the demands of dementia to rise as well. Primary care doctors are in a unique position to diagnose dementia once it starts to appear.
Dr. Girgis practices family medicine in South River, N.J., and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, N.J. You can contact her at [email protected].
Teen boy’s knee lesion has changed
A biopsy of the lesion was performed which showed an increased number of eccrine glands and blood vessels within the dermis. Some areas showed an increase in adipocytes and smooth muscle bundles. The changes were consistent with eccrine angiomatous hamartoma (EAH).
The boy was referred to vascular laser therapy for treatment of the lesion.
EAH is a rare benign vascular growth characterized by an increased number of mature eccrine glands and blood vessels in the dermis and subcutis. The lesions are mostly present on the extremities, but cases of diffuse congenital lesions and lesions on the face and trunk have also been described. The lesions can be seen at birth or during the first years of life in about half of the cases, and the others tend to occur later in puberty and rarely in adulthood.1
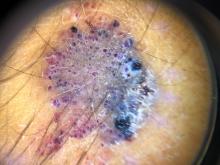
Clinically, EAH lesions present as red, yellow to brown papules and plaques. Different dermoscopic patterns have been described which include the popcorn pattern that presents as yellow, confluent nodules with popcornlike shapes over a background of erythema, and linear arborizing vessels. The spitzoid pattern are brown globules on a background of erythema and pseudoreticular pigmentation around the globules. The verrucous hemangiomalike pattern has a bluish-white hue, reddish-blue or bluish lacunae, as seen in our patient.2-4
Most of the lesions are asymptomatic, but in some patients, they can be associated with pain, hyperhidrosis, and sometimes bleeding. Hyperhidrosis has been reported early in the presentation or during puberty or pregnancy. Our patient had started on amphetamines when hyperhidrosis occurred. Hyperhidrosis is a knowns side effect of this type of medication and may have had a role in the increased sweating noted on the hamartoma.
EAH can clinically look like verrucous hemangiomas, angiokeratomas, and vascular malformations, and histopathology may be needed to differentiate between them. Eccrine nevi and EAH can be similar. Hyperhidrosis is an early and predominant component of eccrine nevi, compared with one-third of EAH.
The exact etiology of this lesion is not known. It is thought to be caused by an abnormal differentiation of the epithelium, adnexal structure, and the mesenchyme during organogenesis.3 No other associated conditions have been described with EAH.
EAH are benign lesions that rarely require treatment. If the lesions are symptomatic or because of cosmetic reasons, they can be removed surgically. There are some reports of successful treatment with pulse dual-wavelength sequential 595- and 1064-nm lasers.5 Botulinum toxin has also been used in cases of symptomatic hyperhidrosis.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She has no conflicts. Email her at [email protected].
References
1. Smith SD et al. Pediatr Dermatol. 2019 Nov;36(6):909-12.
2. Patterson AT et al. Am J Dermatopathol. 2016;38:413-7.
3. Garcıa-Garcıa SC et al. JAAD Case Rep. 2018;4(2):165-7.
4. Awatef Kelati et al. JAAD Case Rep. 2018;4(8)835-6.
5. Felgueiras J et al. Dermatol Surg. 2015 Mar;41(3):428-30.
A biopsy of the lesion was performed which showed an increased number of eccrine glands and blood vessels within the dermis. Some areas showed an increase in adipocytes and smooth muscle bundles. The changes were consistent with eccrine angiomatous hamartoma (EAH).
The boy was referred to vascular laser therapy for treatment of the lesion.
EAH is a rare benign vascular growth characterized by an increased number of mature eccrine glands and blood vessels in the dermis and subcutis. The lesions are mostly present on the extremities, but cases of diffuse congenital lesions and lesions on the face and trunk have also been described. The lesions can be seen at birth or during the first years of life in about half of the cases, and the others tend to occur later in puberty and rarely in adulthood.1
Clinically, EAH lesions present as red, yellow to brown papules and plaques. Different dermoscopic patterns have been described which include the popcorn pattern that presents as yellow, confluent nodules with popcornlike shapes over a background of erythema, and linear arborizing vessels. The spitzoid pattern are brown globules on a background of erythema and pseudoreticular pigmentation around the globules. The verrucous hemangiomalike pattern has a bluish-white hue, reddish-blue or bluish lacunae, as seen in our patient.2-4
Most of the lesions are asymptomatic, but in some patients, they can be associated with pain, hyperhidrosis, and sometimes bleeding. Hyperhidrosis has been reported early in the presentation or during puberty or pregnancy. Our patient had started on amphetamines when hyperhidrosis occurred. Hyperhidrosis is a knowns side effect of this type of medication and may have had a role in the increased sweating noted on the hamartoma.
EAH can clinically look like verrucous hemangiomas, angiokeratomas, and vascular malformations, and histopathology may be needed to differentiate between them. Eccrine nevi and EAH can be similar. Hyperhidrosis is an early and predominant component of eccrine nevi, compared with one-third of EAH.
The exact etiology of this lesion is not known. It is thought to be caused by an abnormal differentiation of the epithelium, adnexal structure, and the mesenchyme during organogenesis.3 No other associated conditions have been described with EAH.
EAH are benign lesions that rarely require treatment. If the lesions are symptomatic or because of cosmetic reasons, they can be removed surgically. There are some reports of successful treatment with pulse dual-wavelength sequential 595- and 1064-nm lasers.5 Botulinum toxin has also been used in cases of symptomatic hyperhidrosis.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She has no conflicts. Email her at [email protected].
References
1. Smith SD et al. Pediatr Dermatol. 2019 Nov;36(6):909-12.
2. Patterson AT et al. Am J Dermatopathol. 2016;38:413-7.
3. Garcıa-Garcıa SC et al. JAAD Case Rep. 2018;4(2):165-7.
4. Awatef Kelati et al. JAAD Case Rep. 2018;4(8)835-6.
5. Felgueiras J et al. Dermatol Surg. 2015 Mar;41(3):428-30.
A biopsy of the lesion was performed which showed an increased number of eccrine glands and blood vessels within the dermis. Some areas showed an increase in adipocytes and smooth muscle bundles. The changes were consistent with eccrine angiomatous hamartoma (EAH).
The boy was referred to vascular laser therapy for treatment of the lesion.
EAH is a rare benign vascular growth characterized by an increased number of mature eccrine glands and blood vessels in the dermis and subcutis. The lesions are mostly present on the extremities, but cases of diffuse congenital lesions and lesions on the face and trunk have also been described. The lesions can be seen at birth or during the first years of life in about half of the cases, and the others tend to occur later in puberty and rarely in adulthood.1
Clinically, EAH lesions present as red, yellow to brown papules and plaques. Different dermoscopic patterns have been described which include the popcorn pattern that presents as yellow, confluent nodules with popcornlike shapes over a background of erythema, and linear arborizing vessels. The spitzoid pattern are brown globules on a background of erythema and pseudoreticular pigmentation around the globules. The verrucous hemangiomalike pattern has a bluish-white hue, reddish-blue or bluish lacunae, as seen in our patient.2-4
Most of the lesions are asymptomatic, but in some patients, they can be associated with pain, hyperhidrosis, and sometimes bleeding. Hyperhidrosis has been reported early in the presentation or during puberty or pregnancy. Our patient had started on amphetamines when hyperhidrosis occurred. Hyperhidrosis is a knowns side effect of this type of medication and may have had a role in the increased sweating noted on the hamartoma.
EAH can clinically look like verrucous hemangiomas, angiokeratomas, and vascular malformations, and histopathology may be needed to differentiate between them. Eccrine nevi and EAH can be similar. Hyperhidrosis is an early and predominant component of eccrine nevi, compared with one-third of EAH.
The exact etiology of this lesion is not known. It is thought to be caused by an abnormal differentiation of the epithelium, adnexal structure, and the mesenchyme during organogenesis.3 No other associated conditions have been described with EAH.
EAH are benign lesions that rarely require treatment. If the lesions are symptomatic or because of cosmetic reasons, they can be removed surgically. There are some reports of successful treatment with pulse dual-wavelength sequential 595- and 1064-nm lasers.5 Botulinum toxin has also been used in cases of symptomatic hyperhidrosis.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She has no conflicts. Email her at [email protected].
References
1. Smith SD et al. Pediatr Dermatol. 2019 Nov;36(6):909-12.
2. Patterson AT et al. Am J Dermatopathol. 2016;38:413-7.
3. Garcıa-Garcıa SC et al. JAAD Case Rep. 2018;4(2):165-7.
4. Awatef Kelati et al. JAAD Case Rep. 2018;4(8)835-6.
5. Felgueiras J et al. Dermatol Surg. 2015 Mar;41(3):428-30.
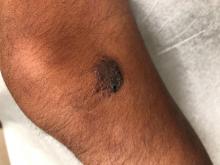
A 14-year-old male was referred to our pediatric dermatology clinic for evaluation of a lesion on the left knee that appeared at 1 year of age. The lesion has been growing with him and was not symptomatic until 6 months prior to the consultation, when it started bleeding and feeling wet.
He has a history of attention-deficit/hyperactivity disorder managed with dextroamphetamine-amphetamine. The changes noted on the knee lesion seem to occur at the same time that his ADHD medication was started.
On physical exam he had a violaceous circular plaque on the left knee.
On dermoscopy the lesion showed multiple dilated red and violaceous lacunae and whitish blue hue.
Sleep apnea has many faces
Fortunately her problem stemmed from sleep apnea, and resolved with continuous positive airway pressure (CPAP) therapy.
Wallace and Bucks performed a meta analysis of 42 studies of memory in patients with sleep apnea and found sleep apnea patients were impaired when compared to healthy controls on verbal episodic memory (immediate recall, delayed recall, learning, and recognition) and visuospatial episodic memory (immediate and delayed recall).1 A meta-analysis by Olaithe and associates found an improvement in executive function in patients with sleep apnea who were treated with CPAP.2 I think this is worth considering especially in your patients who have subjective memory disturbances and do not appear to have a mild cognitive impairment or dementia.
About 15 years ago I saw a 74-year-old man for nocturia. He had seen two urologists and had a transurethral resection of the prostate (TURP) without any real change in his nocturia. I trialed him on all sorts of medications, and he seemed to improve temporarily a little on trazodone (went from seven episodes a night to four).
Eventually, after several years, I sent him for a sleep study. He had severe sleep apnea (Apnea Hypopnea Index, 65; O2 saturations as low as 60%). With treatment, his nocturia resolved. He went from seven episodes to two each night.
Zhou and colleagues performed a meta-analysis of 13 studies looking at the association of sleep apnea with nocturia.3 They found that men with sleep apnea have a high incidence of nocturia.
Miyazato and colleagues looked at the effect of CPAP treatment on nighttime urine production in patients with obstructive sleep apnea.4 In this small study of 40 patients, mean nighttime voiding episodes decreased from 2.1 to 1.2 (P < .01).
I have seen several patients with night sweats who ended up having sleep apnea. These patients have had a resolution of their night sweats with sleep apnea treatment.
Arnardottir and colleagues found that obstructive sleep apnea was associated with frequent nocturnal sweating.5 They found that 31% of men and 33% of women with OSA had nocturnal sweating, compared with about 10% of the general population.
When the OSA patients were treated with positive airway pressure, the prevalence of nocturnal sweating decreased to 11.5%, which is similar to general population numbers. Given how common both sleep apnea and night sweats are, this is an important consideration as you evaluate night sweats.
I have seen many patients who have had atrial fibrillation and sleep apnea. Shapira-Daniels and colleagues did a prospective study of 188 patients with atrial fibrillation without a history of sleep apnea who were referred for ablation.6 All patients had home sleep studies, and testing was consistent with sleep apnea in 82% of patients.
Kanagala and associates found that patients with untreated sleep apnea had a greater chance of recurrent atrial fibrillation after cardioversion.7 Recurrence of atrial fibrillation at 12 months was 82% in untreated OSA patients, higher than the 42% recurrence in the treated OSA group (P = .013) and the 53% recurrence in control patients.
I think sleep apnea evaluation should be strongly considered in patients with atrial fibrillation and should be done before referral for ablations.
Pearl: Consider sleep apnea as a possible cause of or contributing factor to the common primary care problems of cognitive concerns, nocturia, night sweats, and atrial fibrillation.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Wallace A and Bucks RS. Memory and obstructive sleep apnea: a meta-analysis. Sleep. 2013;36(2):203. Epub 2013 Feb 1.
2. Olaithe M and Bucks RS. Executive dysfunction in OSA before and after treatment: a meta-analysis. Sleep. 2013;36(9):1297. Epub 2013 Sep 1.
3. Zhou J et al. Association between obstructive sleep apnea syndrome and nocturia: a meta-analysis. Sleep Breath. 2020 Dec;24(4):1293-8.
4. Miyauchi Y et al. Effect of the continuous positive airway pressure on the nocturnal urine volume or night-time frequency in patients with obstructive sleep apnea syndrome. Urology 2015;85:333.
5. Arnardottir ES et al. Nocturnal sweating–a common symptom of obstructive sleep apnoea: the Icelandic sleep apnoea cohort. BMJ Open. 2013 May 14;3(5):e002795. BMJ Open 2013;3:e002795
6. Shapira-Daniels A et al. Prevalence of undiagnosed sleep apnea in patients with atrial fibrillation and its impact on therapy. JACC Clin Electrophysiol. 2020;6(12):1499. Epub 2020 Aug 12.
7. Kanagala R et al. Obstructive sleep apnea and the recurrence of atrial fibrillation. Circulation. 2003;107(20):2589. Epub 2003 May 12.
Fortunately her problem stemmed from sleep apnea, and resolved with continuous positive airway pressure (CPAP) therapy.
Wallace and Bucks performed a meta analysis of 42 studies of memory in patients with sleep apnea and found sleep apnea patients were impaired when compared to healthy controls on verbal episodic memory (immediate recall, delayed recall, learning, and recognition) and visuospatial episodic memory (immediate and delayed recall).1 A meta-analysis by Olaithe and associates found an improvement in executive function in patients with sleep apnea who were treated with CPAP.2 I think this is worth considering especially in your patients who have subjective memory disturbances and do not appear to have a mild cognitive impairment or dementia.
About 15 years ago I saw a 74-year-old man for nocturia. He had seen two urologists and had a transurethral resection of the prostate (TURP) without any real change in his nocturia. I trialed him on all sorts of medications, and he seemed to improve temporarily a little on trazodone (went from seven episodes a night to four).
Eventually, after several years, I sent him for a sleep study. He had severe sleep apnea (Apnea Hypopnea Index, 65; O2 saturations as low as 60%). With treatment, his nocturia resolved. He went from seven episodes to two each night.
Zhou and colleagues performed a meta-analysis of 13 studies looking at the association of sleep apnea with nocturia.3 They found that men with sleep apnea have a high incidence of nocturia.
Miyazato and colleagues looked at the effect of CPAP treatment on nighttime urine production in patients with obstructive sleep apnea.4 In this small study of 40 patients, mean nighttime voiding episodes decreased from 2.1 to 1.2 (P < .01).
I have seen several patients with night sweats who ended up having sleep apnea. These patients have had a resolution of their night sweats with sleep apnea treatment.
Arnardottir and colleagues found that obstructive sleep apnea was associated with frequent nocturnal sweating.5 They found that 31% of men and 33% of women with OSA had nocturnal sweating, compared with about 10% of the general population.
When the OSA patients were treated with positive airway pressure, the prevalence of nocturnal sweating decreased to 11.5%, which is similar to general population numbers. Given how common both sleep apnea and night sweats are, this is an important consideration as you evaluate night sweats.
I have seen many patients who have had atrial fibrillation and sleep apnea. Shapira-Daniels and colleagues did a prospective study of 188 patients with atrial fibrillation without a history of sleep apnea who were referred for ablation.6 All patients had home sleep studies, and testing was consistent with sleep apnea in 82% of patients.
Kanagala and associates found that patients with untreated sleep apnea had a greater chance of recurrent atrial fibrillation after cardioversion.7 Recurrence of atrial fibrillation at 12 months was 82% in untreated OSA patients, higher than the 42% recurrence in the treated OSA group (P = .013) and the 53% recurrence in control patients.
I think sleep apnea evaluation should be strongly considered in patients with atrial fibrillation and should be done before referral for ablations.
Pearl: Consider sleep apnea as a possible cause of or contributing factor to the common primary care problems of cognitive concerns, nocturia, night sweats, and atrial fibrillation.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Wallace A and Bucks RS. Memory and obstructive sleep apnea: a meta-analysis. Sleep. 2013;36(2):203. Epub 2013 Feb 1.
2. Olaithe M and Bucks RS. Executive dysfunction in OSA before and after treatment: a meta-analysis. Sleep. 2013;36(9):1297. Epub 2013 Sep 1.
3. Zhou J et al. Association between obstructive sleep apnea syndrome and nocturia: a meta-analysis. Sleep Breath. 2020 Dec;24(4):1293-8.
4. Miyauchi Y et al. Effect of the continuous positive airway pressure on the nocturnal urine volume or night-time frequency in patients with obstructive sleep apnea syndrome. Urology 2015;85:333.
5. Arnardottir ES et al. Nocturnal sweating–a common symptom of obstructive sleep apnoea: the Icelandic sleep apnoea cohort. BMJ Open. 2013 May 14;3(5):e002795. BMJ Open 2013;3:e002795
6. Shapira-Daniels A et al. Prevalence of undiagnosed sleep apnea in patients with atrial fibrillation and its impact on therapy. JACC Clin Electrophysiol. 2020;6(12):1499. Epub 2020 Aug 12.
7. Kanagala R et al. Obstructive sleep apnea and the recurrence of atrial fibrillation. Circulation. 2003;107(20):2589. Epub 2003 May 12.
Fortunately her problem stemmed from sleep apnea, and resolved with continuous positive airway pressure (CPAP) therapy.
Wallace and Bucks performed a meta analysis of 42 studies of memory in patients with sleep apnea and found sleep apnea patients were impaired when compared to healthy controls on verbal episodic memory (immediate recall, delayed recall, learning, and recognition) and visuospatial episodic memory (immediate and delayed recall).1 A meta-analysis by Olaithe and associates found an improvement in executive function in patients with sleep apnea who were treated with CPAP.2 I think this is worth considering especially in your patients who have subjective memory disturbances and do not appear to have a mild cognitive impairment or dementia.
About 15 years ago I saw a 74-year-old man for nocturia. He had seen two urologists and had a transurethral resection of the prostate (TURP) without any real change in his nocturia. I trialed him on all sorts of medications, and he seemed to improve temporarily a little on trazodone (went from seven episodes a night to four).
Eventually, after several years, I sent him for a sleep study. He had severe sleep apnea (Apnea Hypopnea Index, 65; O2 saturations as low as 60%). With treatment, his nocturia resolved. He went from seven episodes to two each night.
Zhou and colleagues performed a meta-analysis of 13 studies looking at the association of sleep apnea with nocturia.3 They found that men with sleep apnea have a high incidence of nocturia.
Miyazato and colleagues looked at the effect of CPAP treatment on nighttime urine production in patients with obstructive sleep apnea.4 In this small study of 40 patients, mean nighttime voiding episodes decreased from 2.1 to 1.2 (P < .01).
I have seen several patients with night sweats who ended up having sleep apnea. These patients have had a resolution of their night sweats with sleep apnea treatment.
Arnardottir and colleagues found that obstructive sleep apnea was associated with frequent nocturnal sweating.5 They found that 31% of men and 33% of women with OSA had nocturnal sweating, compared with about 10% of the general population.
When the OSA patients were treated with positive airway pressure, the prevalence of nocturnal sweating decreased to 11.5%, which is similar to general population numbers. Given how common both sleep apnea and night sweats are, this is an important consideration as you evaluate night sweats.
I have seen many patients who have had atrial fibrillation and sleep apnea. Shapira-Daniels and colleagues did a prospective study of 188 patients with atrial fibrillation without a history of sleep apnea who were referred for ablation.6 All patients had home sleep studies, and testing was consistent with sleep apnea in 82% of patients.
Kanagala and associates found that patients with untreated sleep apnea had a greater chance of recurrent atrial fibrillation after cardioversion.7 Recurrence of atrial fibrillation at 12 months was 82% in untreated OSA patients, higher than the 42% recurrence in the treated OSA group (P = .013) and the 53% recurrence in control patients.
I think sleep apnea evaluation should be strongly considered in patients with atrial fibrillation and should be done before referral for ablations.
Pearl: Consider sleep apnea as a possible cause of or contributing factor to the common primary care problems of cognitive concerns, nocturia, night sweats, and atrial fibrillation.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as 3rd-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Wallace A and Bucks RS. Memory and obstructive sleep apnea: a meta-analysis. Sleep. 2013;36(2):203. Epub 2013 Feb 1.
2. Olaithe M and Bucks RS. Executive dysfunction in OSA before and after treatment: a meta-analysis. Sleep. 2013;36(9):1297. Epub 2013 Sep 1.
3. Zhou J et al. Association between obstructive sleep apnea syndrome and nocturia: a meta-analysis. Sleep Breath. 2020 Dec;24(4):1293-8.
4. Miyauchi Y et al. Effect of the continuous positive airway pressure on the nocturnal urine volume or night-time frequency in patients with obstructive sleep apnea syndrome. Urology 2015;85:333.
5. Arnardottir ES et al. Nocturnal sweating–a common symptom of obstructive sleep apnoea: the Icelandic sleep apnoea cohort. BMJ Open. 2013 May 14;3(5):e002795. BMJ Open 2013;3:e002795
6. Shapira-Daniels A et al. Prevalence of undiagnosed sleep apnea in patients with atrial fibrillation and its impact on therapy. JACC Clin Electrophysiol. 2020;6(12):1499. Epub 2020 Aug 12.
7. Kanagala R et al. Obstructive sleep apnea and the recurrence of atrial fibrillation. Circulation. 2003;107(20):2589. Epub 2003 May 12.
Sorting out the meaning of misbehavior: The invisible culprit
You have probably heard that determining the A(ntecedent)s, B(ehavior)s, and C(onsequence)s of a behavior is basic to counseling about oppositionality or aggression. But sorting out the As is especially important to going beyond disciplining a misbehavior to building insight for both parents and children.
Antecedents are of two types: triggers such as actions, words, or feelings that happen just before the behavior, and “setting events” that can occur intermittently hours or even days beforehand and lower the threshold for a trigger to cause a child to act out. Lack of sleep, hunger, or fatigue are common setting events that parents recognize and take into account as in “Oh, he missed his nap” to excuse a tantrum in younger children, but is less often considered or excused in older children in whom self-regulation is expected. Often, behavioral specialists in schools are asked to observe a child to identify the triggers and create a “functional behavioral assessment” based on what is observed.
While a functional behavioral assessment requires observations, invisible antecedents to consider include internal thoughts and feelings (meaning). A child feeling shame from a failed math test the day before may be on edge, then, when called on, may uncharacteristically talk back. The child may regard punishment for this “justified” response as unfair, accelerating anger. Feelings of shame or humiliation for failing one’s own standards (or perceived expectations of others the child cares about) are major setups for eliciting defiance.
Even more subtle are meanings the child creates for situations and people, whether real or imagined. A child’s behavior has meaning for the child and the family and can be initiated or maintained by that meaning. For example, a child may “live down” to what the family thinks of him/her; if you think I am bad, I will act badly.
Children may feel guilty about some real or imagined offense, such as divorce or death they think may be their fault, and act up with the family to elicit punishment as payment. When children feel conflicted in a relationship, such as a late adolescent feeling dependent on their mother when their age expectation is independence, they may act up expecting to be ejected from home when they are unable to gather the courage to voluntarily leave. This acting out may also occur with nonconflicted adults, who are actually safer targets. For example, school is often a safer place to express anger through aggression or bullying than home, the real source of the feelings, because family is the “lifeboat” of food and shelter they dare not upset.
Conflicted relationships may be present in blended families, especially if the ex speaks negatively about the other parent. The child of divorce, feeling himself composed of parts of each parent, has diminished self-esteem and anger on behalf of that side being put down. Marital conflict may set children up to feel they have to take sides to angrily defend the parent of like-gender by being oppositional to the other.
Just as we ponder whether the color blue looks the same to someone else, neurologically based differences in perception may make a child misinterpret or act inflexibly or explode in situations that seem normal to adults. While people joke about “being a little OCD,” for some children the distress caused by a change in routine, a messy room, a delayed bus, or loud music is enough to disrupt their functioning and coping enough to explode. Such hypersensitivity can be part of autism or obsessive compulsive disorder or a subthreshold variant. Children vary by age and individually in their ability to understand language, especially sarcastic humor, and often misinterpret it as insulting, threatening, or scary and act accordingly. While most common in children with autism, those with a language learning disability, intellectual disability, or who have English as a second language, or are anxious or vigilant may also take sarcasm the wrong way. Anxious children also may react aggressively from a “hostile bias attribution” of expecting the worst from others.
Another possible meaning of a behavior is that it is being used by the child to manage their feelings. I have found it useful to remind depressed children and parents that it “feels better to be mad than sad” as a reason for irritability. Anger can also push away a person whose otherwise sympathetic approach might release a collapse into tears the child can’t tolerate or would find embarrassing.
The meaning of a child’s misbehavior also resides in the minds of the adults. In addition to all the categories of meaning just described, a parent may be reminded by the child of someone else for whom the adult has strong or conflicted feelings (“projection”) such as a now-hated ex, a sibling of whom the adult is jealous, or a bully from childhood, thus eliciting a reaction falsely triggered by that connection rather than the actual child. Asking parents whom the child “takes after” may elicit such parental projections based on appearance, behavior, or temperament. Helping them pick a feature of the child to focus on to differentiate him/her can serve as an anchor to remind them to control these reactions. Other useful questions to detect meanings of behavior might include asking the child “What’s up with that?” or “What did that make you think/feel?” We can ask parents “How is that for you?” or “What do you think things will be like in 10 years?” to determine despair, mood disorders, or family discord contributing to maladaptive responses possibly maintaining unwanted behaviors.
Throughout life, putting feelings into words is the main way meanings that are contributing to misbehaviors or parenting dysfunction can be uncovered and shifted. For this, the child or adult must feel emotionally safe to talk with a person who conveys curiosity rather than judgment. Helping families explain that divorce is not the child’s fault; admit they also make mistakes; rebuild conflicted relationships through play or talking; identify hypersensitivities or triggers to avoid; and express confidence that the child is a good person, still young, and sure to do better over time, are all things we pediatricians can do to help sort out the meanings of behaviors.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
You have probably heard that determining the A(ntecedent)s, B(ehavior)s, and C(onsequence)s of a behavior is basic to counseling about oppositionality or aggression. But sorting out the As is especially important to going beyond disciplining a misbehavior to building insight for both parents and children.
Antecedents are of two types: triggers such as actions, words, or feelings that happen just before the behavior, and “setting events” that can occur intermittently hours or even days beforehand and lower the threshold for a trigger to cause a child to act out. Lack of sleep, hunger, or fatigue are common setting events that parents recognize and take into account as in “Oh, he missed his nap” to excuse a tantrum in younger children, but is less often considered or excused in older children in whom self-regulation is expected. Often, behavioral specialists in schools are asked to observe a child to identify the triggers and create a “functional behavioral assessment” based on what is observed.
While a functional behavioral assessment requires observations, invisible antecedents to consider include internal thoughts and feelings (meaning). A child feeling shame from a failed math test the day before may be on edge, then, when called on, may uncharacteristically talk back. The child may regard punishment for this “justified” response as unfair, accelerating anger. Feelings of shame or humiliation for failing one’s own standards (or perceived expectations of others the child cares about) are major setups for eliciting defiance.
Even more subtle are meanings the child creates for situations and people, whether real or imagined. A child’s behavior has meaning for the child and the family and can be initiated or maintained by that meaning. For example, a child may “live down” to what the family thinks of him/her; if you think I am bad, I will act badly.
Children may feel guilty about some real or imagined offense, such as divorce or death they think may be their fault, and act up with the family to elicit punishment as payment. When children feel conflicted in a relationship, such as a late adolescent feeling dependent on their mother when their age expectation is independence, they may act up expecting to be ejected from home when they are unable to gather the courage to voluntarily leave. This acting out may also occur with nonconflicted adults, who are actually safer targets. For example, school is often a safer place to express anger through aggression or bullying than home, the real source of the feelings, because family is the “lifeboat” of food and shelter they dare not upset.
Conflicted relationships may be present in blended families, especially if the ex speaks negatively about the other parent. The child of divorce, feeling himself composed of parts of each parent, has diminished self-esteem and anger on behalf of that side being put down. Marital conflict may set children up to feel they have to take sides to angrily defend the parent of like-gender by being oppositional to the other.
Just as we ponder whether the color blue looks the same to someone else, neurologically based differences in perception may make a child misinterpret or act inflexibly or explode in situations that seem normal to adults. While people joke about “being a little OCD,” for some children the distress caused by a change in routine, a messy room, a delayed bus, or loud music is enough to disrupt their functioning and coping enough to explode. Such hypersensitivity can be part of autism or obsessive compulsive disorder or a subthreshold variant. Children vary by age and individually in their ability to understand language, especially sarcastic humor, and often misinterpret it as insulting, threatening, or scary and act accordingly. While most common in children with autism, those with a language learning disability, intellectual disability, or who have English as a second language, or are anxious or vigilant may also take sarcasm the wrong way. Anxious children also may react aggressively from a “hostile bias attribution” of expecting the worst from others.
Another possible meaning of a behavior is that it is being used by the child to manage their feelings. I have found it useful to remind depressed children and parents that it “feels better to be mad than sad” as a reason for irritability. Anger can also push away a person whose otherwise sympathetic approach might release a collapse into tears the child can’t tolerate or would find embarrassing.
The meaning of a child’s misbehavior also resides in the minds of the adults. In addition to all the categories of meaning just described, a parent may be reminded by the child of someone else for whom the adult has strong or conflicted feelings (“projection”) such as a now-hated ex, a sibling of whom the adult is jealous, or a bully from childhood, thus eliciting a reaction falsely triggered by that connection rather than the actual child. Asking parents whom the child “takes after” may elicit such parental projections based on appearance, behavior, or temperament. Helping them pick a feature of the child to focus on to differentiate him/her can serve as an anchor to remind them to control these reactions. Other useful questions to detect meanings of behavior might include asking the child “What’s up with that?” or “What did that make you think/feel?” We can ask parents “How is that for you?” or “What do you think things will be like in 10 years?” to determine despair, mood disorders, or family discord contributing to maladaptive responses possibly maintaining unwanted behaviors.
Throughout life, putting feelings into words is the main way meanings that are contributing to misbehaviors or parenting dysfunction can be uncovered and shifted. For this, the child or adult must feel emotionally safe to talk with a person who conveys curiosity rather than judgment. Helping families explain that divorce is not the child’s fault; admit they also make mistakes; rebuild conflicted relationships through play or talking; identify hypersensitivities or triggers to avoid; and express confidence that the child is a good person, still young, and sure to do better over time, are all things we pediatricians can do to help sort out the meanings of behaviors.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
You have probably heard that determining the A(ntecedent)s, B(ehavior)s, and C(onsequence)s of a behavior is basic to counseling about oppositionality or aggression. But sorting out the As is especially important to going beyond disciplining a misbehavior to building insight for both parents and children.
Antecedents are of two types: triggers such as actions, words, or feelings that happen just before the behavior, and “setting events” that can occur intermittently hours or even days beforehand and lower the threshold for a trigger to cause a child to act out. Lack of sleep, hunger, or fatigue are common setting events that parents recognize and take into account as in “Oh, he missed his nap” to excuse a tantrum in younger children, but is less often considered or excused in older children in whom self-regulation is expected. Often, behavioral specialists in schools are asked to observe a child to identify the triggers and create a “functional behavioral assessment” based on what is observed.
While a functional behavioral assessment requires observations, invisible antecedents to consider include internal thoughts and feelings (meaning). A child feeling shame from a failed math test the day before may be on edge, then, when called on, may uncharacteristically talk back. The child may regard punishment for this “justified” response as unfair, accelerating anger. Feelings of shame or humiliation for failing one’s own standards (or perceived expectations of others the child cares about) are major setups for eliciting defiance.
Even more subtle are meanings the child creates for situations and people, whether real or imagined. A child’s behavior has meaning for the child and the family and can be initiated or maintained by that meaning. For example, a child may “live down” to what the family thinks of him/her; if you think I am bad, I will act badly.
Children may feel guilty about some real or imagined offense, such as divorce or death they think may be their fault, and act up with the family to elicit punishment as payment. When children feel conflicted in a relationship, such as a late adolescent feeling dependent on their mother when their age expectation is independence, they may act up expecting to be ejected from home when they are unable to gather the courage to voluntarily leave. This acting out may also occur with nonconflicted adults, who are actually safer targets. For example, school is often a safer place to express anger through aggression or bullying than home, the real source of the feelings, because family is the “lifeboat” of food and shelter they dare not upset.
Conflicted relationships may be present in blended families, especially if the ex speaks negatively about the other parent. The child of divorce, feeling himself composed of parts of each parent, has diminished self-esteem and anger on behalf of that side being put down. Marital conflict may set children up to feel they have to take sides to angrily defend the parent of like-gender by being oppositional to the other.
Just as we ponder whether the color blue looks the same to someone else, neurologically based differences in perception may make a child misinterpret or act inflexibly or explode in situations that seem normal to adults. While people joke about “being a little OCD,” for some children the distress caused by a change in routine, a messy room, a delayed bus, or loud music is enough to disrupt their functioning and coping enough to explode. Such hypersensitivity can be part of autism or obsessive compulsive disorder or a subthreshold variant. Children vary by age and individually in their ability to understand language, especially sarcastic humor, and often misinterpret it as insulting, threatening, or scary and act accordingly. While most common in children with autism, those with a language learning disability, intellectual disability, or who have English as a second language, or are anxious or vigilant may also take sarcasm the wrong way. Anxious children also may react aggressively from a “hostile bias attribution” of expecting the worst from others.
Another possible meaning of a behavior is that it is being used by the child to manage their feelings. I have found it useful to remind depressed children and parents that it “feels better to be mad than sad” as a reason for irritability. Anger can also push away a person whose otherwise sympathetic approach might release a collapse into tears the child can’t tolerate or would find embarrassing.
The meaning of a child’s misbehavior also resides in the minds of the adults. In addition to all the categories of meaning just described, a parent may be reminded by the child of someone else for whom the adult has strong or conflicted feelings (“projection”) such as a now-hated ex, a sibling of whom the adult is jealous, or a bully from childhood, thus eliciting a reaction falsely triggered by that connection rather than the actual child. Asking parents whom the child “takes after” may elicit such parental projections based on appearance, behavior, or temperament. Helping them pick a feature of the child to focus on to differentiate him/her can serve as an anchor to remind them to control these reactions. Other useful questions to detect meanings of behavior might include asking the child “What’s up with that?” or “What did that make you think/feel?” We can ask parents “How is that for you?” or “What do you think things will be like in 10 years?” to determine despair, mood disorders, or family discord contributing to maladaptive responses possibly maintaining unwanted behaviors.
Throughout life, putting feelings into words is the main way meanings that are contributing to misbehaviors or parenting dysfunction can be uncovered and shifted. For this, the child or adult must feel emotionally safe to talk with a person who conveys curiosity rather than judgment. Helping families explain that divorce is not the child’s fault; admit they also make mistakes; rebuild conflicted relationships through play or talking; identify hypersensitivities or triggers to avoid; and express confidence that the child is a good person, still young, and sure to do better over time, are all things we pediatricians can do to help sort out the meanings of behaviors.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS. She had no other relevant disclosures. Dr. Howard’s contribution to this publication was as a paid expert to MDedge News. E-mail her at [email protected].
Call them by their names in your office
Given that approximately 9.5% of youth aged 13-17 in the United States identify as lesbian, gay, bisexual, transgender, or queer (LGBTQ),1 it is likely that a general pediatrician or pediatric subspecialist is going to encounter at least one LGBTQ patient during the course of the average workweek. By having an easy way to identify these patients and store this data in a user-friendly manner, you can ensure that your practice is LGBTQ friendly and an affirming environment for all sexual- and gender-minority youth.
One way to do this is to look over any paper or electronic forms your practice uses and make sure that they provide patients and families a range of options to identify themselves. For example, you could provide more options for gender, other than male or female, including a nonbinary or “other” (with a free text line) option. This allows your patients to give you an accurate description of what their affirmed gender is. Instead of having a space for mother’s name and father’s name, you could list these fields as “parent/guardian #1” and “parent/guardian #2.” These labels allow for more inclusivity and to reflect the diverse makeup of modern families. Providing a space for a patient to put the name and pronouns that they use allows your staff to make sure that you are calling a patient by the correct name and using the correct pronouns.
Within your EMR, there may be editable fields that allow for you or your staff to list the patient’s affirmed name and pronouns. Making this small change allows any staff member who accesses the chart to have that information displayed correctly for them and reduces the chances of staff misgendering or dead-naming a patient. Underscoring the importance of this, Sequeira et al. found that in a sample of youth from a gender clinic, only 9% of those adolescents reported that they were asked their name/pronouns outside of the gender clinic.2 If those fields are not there, you may check with your IT staff or your EMR vendor to see if these fields may be added in. However, staff needs to make sure that they check with the child/adolescent first to discern with whom the patient has discussed their gender identity. If you were to put a patient’s affirmed name into the chart and then call the patient by that name in front of the parent/guardian, the parent/guardian may look at you quizzically about why you are calling their child by that name. This could then cause an uncomfortable conversation in the exam room or result in harm to the patient after the visit.
It is not just good clinical practice to ensure that you use a patient’s affirmed name and pronouns. Russell et al. looked at the relationship between depressive symptoms and suicidal ideation and whether an adolescent’s name/pronouns were used in the context of their home, school, work, and/or friend group. They found that use of an adolescent’s affirmed name in at least one of these contexts was associated with a decrease in depressive symptoms and a 29% decrease in suicidal ideation.3 Therefore, the use of an adolescent’s affirmed name and pronouns in your office contributes to the overall mental well-being of your patients.
Fortunately, there are many guides to help you and your practice be successful at implementing some of these changes. The Gay, Lesbian, Bisexual and Transgender Health Access Project put together its “Community Standards of Practice for the Provision of Quality Health Care Services to Lesbian, Gay, Bisexual, and Transgender Clients” to aid practices in developing environments that are LGBTQ affirming. The National LGBTQIA+ Health Education Center, a part of the Fenway Institute, has a series of learning modules that you and your staff can view for interactive training and tips for best practices. These resources offer pediatricians and their practices free resources to improve their policies and procedures. By instituting these small changes, you can ensure that your practice continues to be an affirming environment for your LGBTQ children and adolescents.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
1. Conran KJ. LGBT youth population in the United States, UCLA School of Law, Williams Institute, 2020 Sep.
2. Sequeira GM et al. Affirming transgender youths’ names and pronouns in the electronic medical record. JAMA Pediatr. 2020;174(5):501-3.
3. Russell ST et al. Chosen name use is linked to reduced depressive symptoms, suicidal ideation, and suicidal behavior among transgender youth. J Adolesc Health. 2018;63(4):503-5.
Given that approximately 9.5% of youth aged 13-17 in the United States identify as lesbian, gay, bisexual, transgender, or queer (LGBTQ),1 it is likely that a general pediatrician or pediatric subspecialist is going to encounter at least one LGBTQ patient during the course of the average workweek. By having an easy way to identify these patients and store this data in a user-friendly manner, you can ensure that your practice is LGBTQ friendly and an affirming environment for all sexual- and gender-minority youth.
One way to do this is to look over any paper or electronic forms your practice uses and make sure that they provide patients and families a range of options to identify themselves. For example, you could provide more options for gender, other than male or female, including a nonbinary or “other” (with a free text line) option. This allows your patients to give you an accurate description of what their affirmed gender is. Instead of having a space for mother’s name and father’s name, you could list these fields as “parent/guardian #1” and “parent/guardian #2.” These labels allow for more inclusivity and to reflect the diverse makeup of modern families. Providing a space for a patient to put the name and pronouns that they use allows your staff to make sure that you are calling a patient by the correct name and using the correct pronouns.
Within your EMR, there may be editable fields that allow for you or your staff to list the patient’s affirmed name and pronouns. Making this small change allows any staff member who accesses the chart to have that information displayed correctly for them and reduces the chances of staff misgendering or dead-naming a patient. Underscoring the importance of this, Sequeira et al. found that in a sample of youth from a gender clinic, only 9% of those adolescents reported that they were asked their name/pronouns outside of the gender clinic.2 If those fields are not there, you may check with your IT staff or your EMR vendor to see if these fields may be added in. However, staff needs to make sure that they check with the child/adolescent first to discern with whom the patient has discussed their gender identity. If you were to put a patient’s affirmed name into the chart and then call the patient by that name in front of the parent/guardian, the parent/guardian may look at you quizzically about why you are calling their child by that name. This could then cause an uncomfortable conversation in the exam room or result in harm to the patient after the visit.
It is not just good clinical practice to ensure that you use a patient’s affirmed name and pronouns. Russell et al. looked at the relationship between depressive symptoms and suicidal ideation and whether an adolescent’s name/pronouns were used in the context of their home, school, work, and/or friend group. They found that use of an adolescent’s affirmed name in at least one of these contexts was associated with a decrease in depressive symptoms and a 29% decrease in suicidal ideation.3 Therefore, the use of an adolescent’s affirmed name and pronouns in your office contributes to the overall mental well-being of your patients.
Fortunately, there are many guides to help you and your practice be successful at implementing some of these changes. The Gay, Lesbian, Bisexual and Transgender Health Access Project put together its “Community Standards of Practice for the Provision of Quality Health Care Services to Lesbian, Gay, Bisexual, and Transgender Clients” to aid practices in developing environments that are LGBTQ affirming. The National LGBTQIA+ Health Education Center, a part of the Fenway Institute, has a series of learning modules that you and your staff can view for interactive training and tips for best practices. These resources offer pediatricians and their practices free resources to improve their policies and procedures. By instituting these small changes, you can ensure that your practice continues to be an affirming environment for your LGBTQ children and adolescents.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
1. Conran KJ. LGBT youth population in the United States, UCLA School of Law, Williams Institute, 2020 Sep.
2. Sequeira GM et al. Affirming transgender youths’ names and pronouns in the electronic medical record. JAMA Pediatr. 2020;174(5):501-3.
3. Russell ST et al. Chosen name use is linked to reduced depressive symptoms, suicidal ideation, and suicidal behavior among transgender youth. J Adolesc Health. 2018;63(4):503-5.
Given that approximately 9.5% of youth aged 13-17 in the United States identify as lesbian, gay, bisexual, transgender, or queer (LGBTQ),1 it is likely that a general pediatrician or pediatric subspecialist is going to encounter at least one LGBTQ patient during the course of the average workweek. By having an easy way to identify these patients and store this data in a user-friendly manner, you can ensure that your practice is LGBTQ friendly and an affirming environment for all sexual- and gender-minority youth.
One way to do this is to look over any paper or electronic forms your practice uses and make sure that they provide patients and families a range of options to identify themselves. For example, you could provide more options for gender, other than male or female, including a nonbinary or “other” (with a free text line) option. This allows your patients to give you an accurate description of what their affirmed gender is. Instead of having a space for mother’s name and father’s name, you could list these fields as “parent/guardian #1” and “parent/guardian #2.” These labels allow for more inclusivity and to reflect the diverse makeup of modern families. Providing a space for a patient to put the name and pronouns that they use allows your staff to make sure that you are calling a patient by the correct name and using the correct pronouns.
Within your EMR, there may be editable fields that allow for you or your staff to list the patient’s affirmed name and pronouns. Making this small change allows any staff member who accesses the chart to have that information displayed correctly for them and reduces the chances of staff misgendering or dead-naming a patient. Underscoring the importance of this, Sequeira et al. found that in a sample of youth from a gender clinic, only 9% of those adolescents reported that they were asked their name/pronouns outside of the gender clinic.2 If those fields are not there, you may check with your IT staff or your EMR vendor to see if these fields may be added in. However, staff needs to make sure that they check with the child/adolescent first to discern with whom the patient has discussed their gender identity. If you were to put a patient’s affirmed name into the chart and then call the patient by that name in front of the parent/guardian, the parent/guardian may look at you quizzically about why you are calling their child by that name. This could then cause an uncomfortable conversation in the exam room or result in harm to the patient after the visit.
It is not just good clinical practice to ensure that you use a patient’s affirmed name and pronouns. Russell et al. looked at the relationship between depressive symptoms and suicidal ideation and whether an adolescent’s name/pronouns were used in the context of their home, school, work, and/or friend group. They found that use of an adolescent’s affirmed name in at least one of these contexts was associated with a decrease in depressive symptoms and a 29% decrease in suicidal ideation.3 Therefore, the use of an adolescent’s affirmed name and pronouns in your office contributes to the overall mental well-being of your patients.
Fortunately, there are many guides to help you and your practice be successful at implementing some of these changes. The Gay, Lesbian, Bisexual and Transgender Health Access Project put together its “Community Standards of Practice for the Provision of Quality Health Care Services to Lesbian, Gay, Bisexual, and Transgender Clients” to aid practices in developing environments that are LGBTQ affirming. The National LGBTQIA+ Health Education Center, a part of the Fenway Institute, has a series of learning modules that you and your staff can view for interactive training and tips for best practices. These resources offer pediatricians and their practices free resources to improve their policies and procedures. By instituting these small changes, you can ensure that your practice continues to be an affirming environment for your LGBTQ children and adolescents.
Dr. Cooper is assistant professor of pediatrics at University of Texas Southwestern, Dallas, and an adolescent medicine specialist at Children’s Medical Center Dallas.
References
1. Conran KJ. LGBT youth population in the United States, UCLA School of Law, Williams Institute, 2020 Sep.
2. Sequeira GM et al. Affirming transgender youths’ names and pronouns in the electronic medical record. JAMA Pediatr. 2020;174(5):501-3.
3. Russell ST et al. Chosen name use is linked to reduced depressive symptoms, suicidal ideation, and suicidal behavior among transgender youth. J Adolesc Health. 2018;63(4):503-5.
What’s behind the rise in youth anxiety and depression?
It’s well known that levels of anxiety and depression in youth are on the rise. While some of this increase may be because of other things, such as a lowering of the threshold for what counts as clinically relevant symptoms and decreased stigma when it comes to seeking out mental health services, there seems little debate that the number of children and adolescents who are actually struggling with their mental health is taking a sharp turn for the worse.
What is much less certain are the causes behind this surge. The answer to this important question will likely defy a clear answer from a definitive study. In its place then are a number of different theories that have been circulated and discussed. Each comes with some evidence to support the hypothesis, but none seems able to make a truly compelling argument as the single driving force behind this trend. This column briefly describes and examines some of the factors that may be contributing to the rise in anxiety and depression while providing some explanation for why each factor is unlikely to be the sole culprit.
Some of the biggest suggested causes for the rise in child and adolescent mental health problems include the following:
- COVID. Multiple studies have documented increases in mood and anxiety associated with the pandemic, which in turn, may be because of a number of factors such as social isolation, loss of family members, family financial stressors, and many other contributors.1 Yet, while it certainly makes sense that COVID is a powerful instigator of mood and anxiety problems, there is good evidence that the upward tic in emotional-behavioral problems began well before the COVID pandemic.2
- Smartphones. In 2017, psychologist Jean Twenge penned a provocative essay in the Atlantic with the title “Have Smartphones Destroyed a Generation?” and the basic answer was yes.3 The foundation for this conclusion was the tracking between the rise in mood and anxiety problems and the meteoric rise of smartphone use in youth. None of these associations, however, can be proven as casual, and more experimental data on the link between smartphone usage and mental health have been inconsistent.
- Bullying. The toxic effect of bullying and, in particular, online or “cyberbullying” has frequently been brought up as a potential cause. Yet while the negative effects of bullying have been well documented, there is evidence that overall bullying has actually decreased over recent years.4
These three factors have arguably been the most discussed, but a few others also probably deserve mention.
- Helicopter parenting. Critics of this common and increasingly popular approach to parenting are concerned that all the parental hovering and stepping in convey the message that the world is a very dangerous place while depriving children of opportunities to gain the exposure and competence they need to succeed. The critique is certainly logical and even has been supported in some studies but lacks the needed evidence for a more definitive conclusion.5
- Medications. Of course there will be stories blaming the mental health treatment itself, rather than the reasons people seek treatment, for this disturbing trend. And while it is always important to consider that medications can be part of the problem rather than the solution, the majority of evidence points overall to a lack of treatment rather than too much. A recent important study, for example, found that the peak of suicidal thoughts and behaviors occurred a month before medications were started, rather than after.6
- Cannabis. While there seems to be a lot of geographic variability with regard to whether or not the number of youth using cannabis is increasing or not, it’s clear that the product now being consumed is considerably stronger than what was used in decades past. This high-potency cannabis now being used has been shown to increase the risk for later mental health problems including psychosis and suicidal behavior.7 Unfortunately, these risks are not being heard as a powerful industry fights to increase their market share.
Putting all this together, it seems likely that a tidy and simple explanation for the alarming increase in youth mental health problems will be hard to pin down. It’s also worth pointing out that many of the above factors could work in a synergistic manner. For example, helicopter parenting may be keeping kids more confined to their rooms where they interact more and more on their phones and are exposed to higher amounts of online bullying, all of which has been magnified recently with the COVID pandemic. Obviously, understanding the causes behind this surge is much more than an academic exercise as the amount of stress and suffering rises and treatment resources get overwhelmed. In the meantime, addressing all of the above factors in both primary and specialty care is worthwhile in an effort to reverse this worrying and wide-ranging pattern.
Dr. Rettew is a child and adolescent psychiatrist and medical director of Lane County Behavioral Health in Eugene, Ore. He is the author of the 2021 book, “Parenting Made Complicated: What Science Really Knows about the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
References
1. Hawes MT et al. Psychol Med. 2021;13:1-9.
2. Twenge JM et al. J Abnorm Psych. 2019;128(3):185-99.
3. Twenge JM. Have Smartphones Destroyed a Generation? The Atlantic. 2017:September.
4. Rettew DC. Bullying: An update. Child Psych Clin North Am. 2021; in press.
5. Van Der Bruggen CO et al. J Child Psychol Psychiatry. 2008;49(12):1257-69.
6. Lagerberg T et al. Selective serotonin reuptake inhibitors and suicidal behaviour: A population-based cohort study. Neuropsychopharmacology 2021 Sep 24.
7. Gobbi G et al. JAMA Psychiatry. 2019;76(4):426-34.
It’s well known that levels of anxiety and depression in youth are on the rise. While some of this increase may be because of other things, such as a lowering of the threshold for what counts as clinically relevant symptoms and decreased stigma when it comes to seeking out mental health services, there seems little debate that the number of children and adolescents who are actually struggling with their mental health is taking a sharp turn for the worse.
What is much less certain are the causes behind this surge. The answer to this important question will likely defy a clear answer from a definitive study. In its place then are a number of different theories that have been circulated and discussed. Each comes with some evidence to support the hypothesis, but none seems able to make a truly compelling argument as the single driving force behind this trend. This column briefly describes and examines some of the factors that may be contributing to the rise in anxiety and depression while providing some explanation for why each factor is unlikely to be the sole culprit.
Some of the biggest suggested causes for the rise in child and adolescent mental health problems include the following:
- COVID. Multiple studies have documented increases in mood and anxiety associated with the pandemic, which in turn, may be because of a number of factors such as social isolation, loss of family members, family financial stressors, and many other contributors.1 Yet, while it certainly makes sense that COVID is a powerful instigator of mood and anxiety problems, there is good evidence that the upward tic in emotional-behavioral problems began well before the COVID pandemic.2
- Smartphones. In 2017, psychologist Jean Twenge penned a provocative essay in the Atlantic with the title “Have Smartphones Destroyed a Generation?” and the basic answer was yes.3 The foundation for this conclusion was the tracking between the rise in mood and anxiety problems and the meteoric rise of smartphone use in youth. None of these associations, however, can be proven as casual, and more experimental data on the link between smartphone usage and mental health have been inconsistent.
- Bullying. The toxic effect of bullying and, in particular, online or “cyberbullying” has frequently been brought up as a potential cause. Yet while the negative effects of bullying have been well documented, there is evidence that overall bullying has actually decreased over recent years.4
These three factors have arguably been the most discussed, but a few others also probably deserve mention.
- Helicopter parenting. Critics of this common and increasingly popular approach to parenting are concerned that all the parental hovering and stepping in convey the message that the world is a very dangerous place while depriving children of opportunities to gain the exposure and competence they need to succeed. The critique is certainly logical and even has been supported in some studies but lacks the needed evidence for a more definitive conclusion.5
- Medications. Of course there will be stories blaming the mental health treatment itself, rather than the reasons people seek treatment, for this disturbing trend. And while it is always important to consider that medications can be part of the problem rather than the solution, the majority of evidence points overall to a lack of treatment rather than too much. A recent important study, for example, found that the peak of suicidal thoughts and behaviors occurred a month before medications were started, rather than after.6
- Cannabis. While there seems to be a lot of geographic variability with regard to whether or not the number of youth using cannabis is increasing or not, it’s clear that the product now being consumed is considerably stronger than what was used in decades past. This high-potency cannabis now being used has been shown to increase the risk for later mental health problems including psychosis and suicidal behavior.7 Unfortunately, these risks are not being heard as a powerful industry fights to increase their market share.
Putting all this together, it seems likely that a tidy and simple explanation for the alarming increase in youth mental health problems will be hard to pin down. It’s also worth pointing out that many of the above factors could work in a synergistic manner. For example, helicopter parenting may be keeping kids more confined to their rooms where they interact more and more on their phones and are exposed to higher amounts of online bullying, all of which has been magnified recently with the COVID pandemic. Obviously, understanding the causes behind this surge is much more than an academic exercise as the amount of stress and suffering rises and treatment resources get overwhelmed. In the meantime, addressing all of the above factors in both primary and specialty care is worthwhile in an effort to reverse this worrying and wide-ranging pattern.
Dr. Rettew is a child and adolescent psychiatrist and medical director of Lane County Behavioral Health in Eugene, Ore. He is the author of the 2021 book, “Parenting Made Complicated: What Science Really Knows about the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
References
1. Hawes MT et al. Psychol Med. 2021;13:1-9.
2. Twenge JM et al. J Abnorm Psych. 2019;128(3):185-99.
3. Twenge JM. Have Smartphones Destroyed a Generation? The Atlantic. 2017:September.
4. Rettew DC. Bullying: An update. Child Psych Clin North Am. 2021; in press.
5. Van Der Bruggen CO et al. J Child Psychol Psychiatry. 2008;49(12):1257-69.
6. Lagerberg T et al. Selective serotonin reuptake inhibitors and suicidal behaviour: A population-based cohort study. Neuropsychopharmacology 2021 Sep 24.
7. Gobbi G et al. JAMA Psychiatry. 2019;76(4):426-34.
It’s well known that levels of anxiety and depression in youth are on the rise. While some of this increase may be because of other things, such as a lowering of the threshold for what counts as clinically relevant symptoms and decreased stigma when it comes to seeking out mental health services, there seems little debate that the number of children and adolescents who are actually struggling with their mental health is taking a sharp turn for the worse.
What is much less certain are the causes behind this surge. The answer to this important question will likely defy a clear answer from a definitive study. In its place then are a number of different theories that have been circulated and discussed. Each comes with some evidence to support the hypothesis, but none seems able to make a truly compelling argument as the single driving force behind this trend. This column briefly describes and examines some of the factors that may be contributing to the rise in anxiety and depression while providing some explanation for why each factor is unlikely to be the sole culprit.
Some of the biggest suggested causes for the rise in child and adolescent mental health problems include the following:
- COVID. Multiple studies have documented increases in mood and anxiety associated with the pandemic, which in turn, may be because of a number of factors such as social isolation, loss of family members, family financial stressors, and many other contributors.1 Yet, while it certainly makes sense that COVID is a powerful instigator of mood and anxiety problems, there is good evidence that the upward tic in emotional-behavioral problems began well before the COVID pandemic.2
- Smartphones. In 2017, psychologist Jean Twenge penned a provocative essay in the Atlantic with the title “Have Smartphones Destroyed a Generation?” and the basic answer was yes.3 The foundation for this conclusion was the tracking between the rise in mood and anxiety problems and the meteoric rise of smartphone use in youth. None of these associations, however, can be proven as casual, and more experimental data on the link between smartphone usage and mental health have been inconsistent.
- Bullying. The toxic effect of bullying and, in particular, online or “cyberbullying” has frequently been brought up as a potential cause. Yet while the negative effects of bullying have been well documented, there is evidence that overall bullying has actually decreased over recent years.4
These three factors have arguably been the most discussed, but a few others also probably deserve mention.
- Helicopter parenting. Critics of this common and increasingly popular approach to parenting are concerned that all the parental hovering and stepping in convey the message that the world is a very dangerous place while depriving children of opportunities to gain the exposure and competence they need to succeed. The critique is certainly logical and even has been supported in some studies but lacks the needed evidence for a more definitive conclusion.5
- Medications. Of course there will be stories blaming the mental health treatment itself, rather than the reasons people seek treatment, for this disturbing trend. And while it is always important to consider that medications can be part of the problem rather than the solution, the majority of evidence points overall to a lack of treatment rather than too much. A recent important study, for example, found that the peak of suicidal thoughts and behaviors occurred a month before medications were started, rather than after.6
- Cannabis. While there seems to be a lot of geographic variability with regard to whether or not the number of youth using cannabis is increasing or not, it’s clear that the product now being consumed is considerably stronger than what was used in decades past. This high-potency cannabis now being used has been shown to increase the risk for later mental health problems including psychosis and suicidal behavior.7 Unfortunately, these risks are not being heard as a powerful industry fights to increase their market share.
Putting all this together, it seems likely that a tidy and simple explanation for the alarming increase in youth mental health problems will be hard to pin down. It’s also worth pointing out that many of the above factors could work in a synergistic manner. For example, helicopter parenting may be keeping kids more confined to their rooms where they interact more and more on their phones and are exposed to higher amounts of online bullying, all of which has been magnified recently with the COVID pandemic. Obviously, understanding the causes behind this surge is much more than an academic exercise as the amount of stress and suffering rises and treatment resources get overwhelmed. In the meantime, addressing all of the above factors in both primary and specialty care is worthwhile in an effort to reverse this worrying and wide-ranging pattern.
Dr. Rettew is a child and adolescent psychiatrist and medical director of Lane County Behavioral Health in Eugene, Ore. He is the author of the 2021 book, “Parenting Made Complicated: What Science Really Knows about the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
References
1. Hawes MT et al. Psychol Med. 2021;13:1-9.
2. Twenge JM et al. J Abnorm Psych. 2019;128(3):185-99.
3. Twenge JM. Have Smartphones Destroyed a Generation? The Atlantic. 2017:September.
4. Rettew DC. Bullying: An update. Child Psych Clin North Am. 2021; in press.
5. Van Der Bruggen CO et al. J Child Psychol Psychiatry. 2008;49(12):1257-69.
6. Lagerberg T et al. Selective serotonin reuptake inhibitors and suicidal behaviour: A population-based cohort study. Neuropsychopharmacology 2021 Sep 24.
7. Gobbi G et al. JAMA Psychiatry. 2019;76(4):426-34.
You’ve been uneasy about the mother’s boyfriend: This may be why
The first patient of the afternoon is a 4-month-old in for his health maintenance visit. You’ve known his 20-year-old mother since she was a toddler. This infant has a 2-year-old sister. Also in the exam room is a young man you don’t recognize whom the mother introduces as Jason, her new boyfriend. He never makes eye contact and despite your best efforts you can’t get him to engage.
At the child’s next visit you are relieved to see the 6-month-old is alive and well and learn that your former patient and her two children have moved back in with her parents and Jason is no longer in the picture.
You don’t have to have been doing pediatrics very long to have learned that a “family” that includes an infant and a young adult male who is probably not the father is an environment in which the infant’s health and well-being is at significant risk. It is a situation in which child abuse even to the point of infanticide should be waving a red flag in your face.
Infanticide occurs in many animal species including our own. As abhorrent we may find the act, it occurs often enough to be, if not normal, at least not unexpected in certain circumstances. Theories abound as to what advantage the act of infanticide might convey to the success of a species. However, little if anything is known about any possible mechanisms that would allow it to occur.
Recently, a professor of molecular and cellular biology at Harvard University discovered a specific set of neurons in the mouse brain that controls aggressive behavior toward infants (Biological triggers for infant abuse, by Juan Siliezar, The Harvard Gazette, Sept 27, 2021). This same set of neurons also appears to trigger avoidance and neglect behaviors as well.
Research in other animal species has found that these antiparental behaviors occur in both virgins and sexually mature males who are strangers to the group. Interestingly, the behaviors switch off once individuals have their own offspring or have had the opportunity to familiarize themselves with infants. Not surprisingly, other studies have found that in some species environmental stress such as food shortage or threats of predation have triggered females to attack or ignore their offspring.
I think it is safe to assume a similar collection of neurons controlling aggressive behavior also exists in humans. One can imagine some well-read defense attorney dredging up this study and claiming that because his client had not yet fathered a child of his own that it was his nervous system’s normal response that made him toss his girlfriend’s baby against the wall.
The lead author of the study intends to study this collection of neurons in more depth to discover more about the process. It is conceivable that with more information her initial findings may help in the development of treatment and specific prevention strategies. Until that happens, we must rely on our intuition and keep our antennae tuned and alert for high-risk scenarios like the one I described at the opening of this letter.
We are left with leaning heavily on our community social work networks to keep close tabs on these high-risk families, offering both financial and emotional support. Parenting classes may be helpful, but some of this research leads me to suspect that immersing these young parents-to-be in hands-on child care situations might provide the best protection we can offer.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
The first patient of the afternoon is a 4-month-old in for his health maintenance visit. You’ve known his 20-year-old mother since she was a toddler. This infant has a 2-year-old sister. Also in the exam room is a young man you don’t recognize whom the mother introduces as Jason, her new boyfriend. He never makes eye contact and despite your best efforts you can’t get him to engage.
At the child’s next visit you are relieved to see the 6-month-old is alive and well and learn that your former patient and her two children have moved back in with her parents and Jason is no longer in the picture.
You don’t have to have been doing pediatrics very long to have learned that a “family” that includes an infant and a young adult male who is probably not the father is an environment in which the infant’s health and well-being is at significant risk. It is a situation in which child abuse even to the point of infanticide should be waving a red flag in your face.
Infanticide occurs in many animal species including our own. As abhorrent we may find the act, it occurs often enough to be, if not normal, at least not unexpected in certain circumstances. Theories abound as to what advantage the act of infanticide might convey to the success of a species. However, little if anything is known about any possible mechanisms that would allow it to occur.
Recently, a professor of molecular and cellular biology at Harvard University discovered a specific set of neurons in the mouse brain that controls aggressive behavior toward infants (Biological triggers for infant abuse, by Juan Siliezar, The Harvard Gazette, Sept 27, 2021). This same set of neurons also appears to trigger avoidance and neglect behaviors as well.
Research in other animal species has found that these antiparental behaviors occur in both virgins and sexually mature males who are strangers to the group. Interestingly, the behaviors switch off once individuals have their own offspring or have had the opportunity to familiarize themselves with infants. Not surprisingly, other studies have found that in some species environmental stress such as food shortage or threats of predation have triggered females to attack or ignore their offspring.
I think it is safe to assume a similar collection of neurons controlling aggressive behavior also exists in humans. One can imagine some well-read defense attorney dredging up this study and claiming that because his client had not yet fathered a child of his own that it was his nervous system’s normal response that made him toss his girlfriend’s baby against the wall.
The lead author of the study intends to study this collection of neurons in more depth to discover more about the process. It is conceivable that with more information her initial findings may help in the development of treatment and specific prevention strategies. Until that happens, we must rely on our intuition and keep our antennae tuned and alert for high-risk scenarios like the one I described at the opening of this letter.
We are left with leaning heavily on our community social work networks to keep close tabs on these high-risk families, offering both financial and emotional support. Parenting classes may be helpful, but some of this research leads me to suspect that immersing these young parents-to-be in hands-on child care situations might provide the best protection we can offer.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
The first patient of the afternoon is a 4-month-old in for his health maintenance visit. You’ve known his 20-year-old mother since she was a toddler. This infant has a 2-year-old sister. Also in the exam room is a young man you don’t recognize whom the mother introduces as Jason, her new boyfriend. He never makes eye contact and despite your best efforts you can’t get him to engage.
At the child’s next visit you are relieved to see the 6-month-old is alive and well and learn that your former patient and her two children have moved back in with her parents and Jason is no longer in the picture.
You don’t have to have been doing pediatrics very long to have learned that a “family” that includes an infant and a young adult male who is probably not the father is an environment in which the infant’s health and well-being is at significant risk. It is a situation in which child abuse even to the point of infanticide should be waving a red flag in your face.
Infanticide occurs in many animal species including our own. As abhorrent we may find the act, it occurs often enough to be, if not normal, at least not unexpected in certain circumstances. Theories abound as to what advantage the act of infanticide might convey to the success of a species. However, little if anything is known about any possible mechanisms that would allow it to occur.
Recently, a professor of molecular and cellular biology at Harvard University discovered a specific set of neurons in the mouse brain that controls aggressive behavior toward infants (Biological triggers for infant abuse, by Juan Siliezar, The Harvard Gazette, Sept 27, 2021). This same set of neurons also appears to trigger avoidance and neglect behaviors as well.
Research in other animal species has found that these antiparental behaviors occur in both virgins and sexually mature males who are strangers to the group. Interestingly, the behaviors switch off once individuals have their own offspring or have had the opportunity to familiarize themselves with infants. Not surprisingly, other studies have found that in some species environmental stress such as food shortage or threats of predation have triggered females to attack or ignore their offspring.
I think it is safe to assume a similar collection of neurons controlling aggressive behavior also exists in humans. One can imagine some well-read defense attorney dredging up this study and claiming that because his client had not yet fathered a child of his own that it was his nervous system’s normal response that made him toss his girlfriend’s baby against the wall.
The lead author of the study intends to study this collection of neurons in more depth to discover more about the process. It is conceivable that with more information her initial findings may help in the development of treatment and specific prevention strategies. Until that happens, we must rely on our intuition and keep our antennae tuned and alert for high-risk scenarios like the one I described at the opening of this letter.
We are left with leaning heavily on our community social work networks to keep close tabs on these high-risk families, offering both financial and emotional support. Parenting classes may be helpful, but some of this research leads me to suspect that immersing these young parents-to-be in hands-on child care situations might provide the best protection we can offer.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Effect of COVID-19 pandemic on respiratory infectious diseases in primary care practice
A secondary consequence of public health measures to prevent the spread of SARS-CoV-2 included a concurrent reduction in risk for children to acquire and spread other respiratory viral infectious diseases. In the Rochester, N.Y., area, we had an ongoing prospective study in primary care pediatric practices that afforded an opportunity to assess the effect of the pandemic control measures on all infectious disease illness visits in young children. Specifically, in children aged 6-36 months old, our study was in place when the pandemic began with a primary objective to evaluate the changing epidemiology of acute otitis media (AOM) and nasopharyngeal colonization by potential bacterial respiratory pathogens in community-based primary care pediatric practices. As the public health measures mandated by New York State Department of Health were implemented, we prospectively quantified their effect on physician-diagnosed infectious disease illness visits. The incidence of infectious disease visits by a cohort of young children during the COVID-19 pandemic period March 15, 2020, through Dec. 31, 2020, was compared with the same time frame in the preceding year, 2019.1
Recommendations of the New York State Department of Health for public health, changes in school and day care attendance, and clinical practice during the study time frame
On March 7, 2020, a state of emergency was declared in New York because of the COVID-19 pandemic. All schools were required to close. A mandated order for public use of masks in adults and children more than 2 years of age was enacted. In the Finger Lakes region of Upstate New York, where the two primary care pediatric practices reside, complete lockdown was partially lifted on May 15, 2020, and further lifted on June 26, 2020. Almost all regional school districts opened to at least hybrid learning models for all students starting Sept. 8, 2020. On March 6, 2020, video telehealth and telephone call visits were introduced as routine practice. Well-child visits were limited to those less than 2 years of age, then gradually expanded to all ages by late May 2020. During the “stay at home” phase of the New York State lockdown, day care services were considered an essential business. Day care child density was limited. All children less than 2 years old were required to wear a mask while in the facility. Upon arrival, children with any respiratory symptoms or fever were excluded. For the school year commencing September 2020, almost all regional school districts opened to virtual, hybrid, or in-person learning models. Exclusion occurred similar to that of the day care facilities.
Incidence of respiratory infectious disease illnesses
Clinical diagnoses and healthy visits of 144 children from March 15 to Dec. 31, 2020 (beginning of the pandemic) were compared to 215 children during the same months in 2019 (prepandemic). Pediatric SARS-CoV-2 positivity rates trended up alongside community spread. Pediatric practice positivity rates rose from 1.9% in October 2020 to 19% in December 2020.
The table shows the incidence of significantly different infectious disease illness visits in the two study cohorts.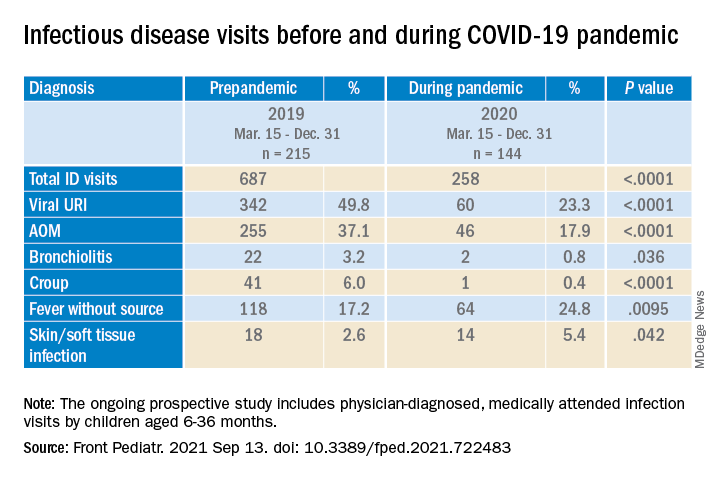
During the pandemic, 258 infection visits occurred among 144 pandemic cohort children, compared with 687 visits among 215 prepandemic cohort children, a 1.8-fold decrease (P < .0001). The proportion of children with visits for AOM (3.7-fold; P < .0001), bronchiolitis (7.4-fold; P = .036), croup (27.5-fold; P < .0001), and viral upper respiratory infection (3.8-fold; P < .0001) decreased significantly. Fever without a source (1.4-fold decrease; P = .009) and skin/soft tissue infection (2.1-fold decrease; P = .042) represented a higher proportion of visits during the pandemic.
Prescription of antibiotics significantly decreased (P < .001) during the pandemic.
Change in care practices
In the prepandemic period, virtual visits, leading to a diagnosis and treatment and referring children to an urgent care or hospital emergency department during regular office hours were rare. During the pandemic, this changed. Significantly increased use of telemedicine visits (P < .0001) and significantly decreased office and urgent care visits (P < .0001) occurred during the pandemic. Telehealth visits peaked the week of April 12, 2020, at 45% of all pediatric visits. In-person illness visits gradually returned to year-to-year volumes in August-September 2020 with school opening. Early in the pandemic, both pediatric offices limited patient encounters to well-child visits in the first 2 years of life to not miss opportunities for childhood vaccinations. However, some parents were reluctant to bring their children to those visits. There was no significant change in frequency of healthy child visits during the pandemic.
To our knowledge, this was the first study from primary care pediatric practices in the United States to analyze the effect on infectious diseases during the first 9 months of the pandemic, including the 6-month time period after the reopening from the first 3 months of lockdown. One prior study from a primary care network in Massachusetts reported significant decreases in respiratory infectious diseases for children aged 0-17 years during the first months of the pandemic during lockdown.2 A study in Tennessee that included hospital emergency department, urgent care, primary care, and retail health clinics also reported respiratory infection diagnoses as well as antibiotic prescription were reduced in the early months of the pandemic.3
Our study shows an overall reduction in frequency of respiratory illness visits in children 6-36 months old during the first 9 months of the COVID-19 pandemic. We learned the value of using technology in the form of virtual visits to render care. Perhaps as the pandemic subsides, many of the hand-washing and sanitizing practices will remain in place and lead to less frequent illness in children in the future. However, there may be temporary negative consequences from the “immune debt” that has occurred from a prolonged time span when children were not becoming infected with respiratory pathogens.4 We will see what unfolds in the future.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. Dr. Schulz is pediatric medical director at Rochester (N.Y.) Regional Health. Dr. Pichichero and Dr. Schulz have no conflicts of interest to disclose. This study was funded in part by the Centers for Disease Control and Prevention.
References
1. Kaur R et al. Front Pediatr. 2021;(9)722483:1-8.
2. Hatoun J et al. Pediatrics. 2020;146(4):e2020006460.
3. Katz SE et al. J Pediatric Infect Dis Soc. 2021;10(1):62-4.
4. Cohen R et al. Infect. Dis Now. 2021; 51(5)418-23.
A secondary consequence of public health measures to prevent the spread of SARS-CoV-2 included a concurrent reduction in risk for children to acquire and spread other respiratory viral infectious diseases. In the Rochester, N.Y., area, we had an ongoing prospective study in primary care pediatric practices that afforded an opportunity to assess the effect of the pandemic control measures on all infectious disease illness visits in young children. Specifically, in children aged 6-36 months old, our study was in place when the pandemic began with a primary objective to evaluate the changing epidemiology of acute otitis media (AOM) and nasopharyngeal colonization by potential bacterial respiratory pathogens in community-based primary care pediatric practices. As the public health measures mandated by New York State Department of Health were implemented, we prospectively quantified their effect on physician-diagnosed infectious disease illness visits. The incidence of infectious disease visits by a cohort of young children during the COVID-19 pandemic period March 15, 2020, through Dec. 31, 2020, was compared with the same time frame in the preceding year, 2019.1
Recommendations of the New York State Department of Health for public health, changes in school and day care attendance, and clinical practice during the study time frame
On March 7, 2020, a state of emergency was declared in New York because of the COVID-19 pandemic. All schools were required to close. A mandated order for public use of masks in adults and children more than 2 years of age was enacted. In the Finger Lakes region of Upstate New York, where the two primary care pediatric practices reside, complete lockdown was partially lifted on May 15, 2020, and further lifted on June 26, 2020. Almost all regional school districts opened to at least hybrid learning models for all students starting Sept. 8, 2020. On March 6, 2020, video telehealth and telephone call visits were introduced as routine practice. Well-child visits were limited to those less than 2 years of age, then gradually expanded to all ages by late May 2020. During the “stay at home” phase of the New York State lockdown, day care services were considered an essential business. Day care child density was limited. All children less than 2 years old were required to wear a mask while in the facility. Upon arrival, children with any respiratory symptoms or fever were excluded. For the school year commencing September 2020, almost all regional school districts opened to virtual, hybrid, or in-person learning models. Exclusion occurred similar to that of the day care facilities.
Incidence of respiratory infectious disease illnesses
Clinical diagnoses and healthy visits of 144 children from March 15 to Dec. 31, 2020 (beginning of the pandemic) were compared to 215 children during the same months in 2019 (prepandemic). Pediatric SARS-CoV-2 positivity rates trended up alongside community spread. Pediatric practice positivity rates rose from 1.9% in October 2020 to 19% in December 2020.
The table shows the incidence of significantly different infectious disease illness visits in the two study cohorts.
During the pandemic, 258 infection visits occurred among 144 pandemic cohort children, compared with 687 visits among 215 prepandemic cohort children, a 1.8-fold decrease (P < .0001). The proportion of children with visits for AOM (3.7-fold; P < .0001), bronchiolitis (7.4-fold; P = .036), croup (27.5-fold; P < .0001), and viral upper respiratory infection (3.8-fold; P < .0001) decreased significantly. Fever without a source (1.4-fold decrease; P = .009) and skin/soft tissue infection (2.1-fold decrease; P = .042) represented a higher proportion of visits during the pandemic.
Prescription of antibiotics significantly decreased (P < .001) during the pandemic.
Change in care practices
In the prepandemic period, virtual visits, leading to a diagnosis and treatment and referring children to an urgent care or hospital emergency department during regular office hours were rare. During the pandemic, this changed. Significantly increased use of telemedicine visits (P < .0001) and significantly decreased office and urgent care visits (P < .0001) occurred during the pandemic. Telehealth visits peaked the week of April 12, 2020, at 45% of all pediatric visits. In-person illness visits gradually returned to year-to-year volumes in August-September 2020 with school opening. Early in the pandemic, both pediatric offices limited patient encounters to well-child visits in the first 2 years of life to not miss opportunities for childhood vaccinations. However, some parents were reluctant to bring their children to those visits. There was no significant change in frequency of healthy child visits during the pandemic.
To our knowledge, this was the first study from primary care pediatric practices in the United States to analyze the effect on infectious diseases during the first 9 months of the pandemic, including the 6-month time period after the reopening from the first 3 months of lockdown. One prior study from a primary care network in Massachusetts reported significant decreases in respiratory infectious diseases for children aged 0-17 years during the first months of the pandemic during lockdown.2 A study in Tennessee that included hospital emergency department, urgent care, primary care, and retail health clinics also reported respiratory infection diagnoses as well as antibiotic prescription were reduced in the early months of the pandemic.3
Our study shows an overall reduction in frequency of respiratory illness visits in children 6-36 months old during the first 9 months of the COVID-19 pandemic. We learned the value of using technology in the form of virtual visits to render care. Perhaps as the pandemic subsides, many of the hand-washing and sanitizing practices will remain in place and lead to less frequent illness in children in the future. However, there may be temporary negative consequences from the “immune debt” that has occurred from a prolonged time span when children were not becoming infected with respiratory pathogens.4 We will see what unfolds in the future.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. Dr. Schulz is pediatric medical director at Rochester (N.Y.) Regional Health. Dr. Pichichero and Dr. Schulz have no conflicts of interest to disclose. This study was funded in part by the Centers for Disease Control and Prevention.
References
1. Kaur R et al. Front Pediatr. 2021;(9)722483:1-8.
2. Hatoun J et al. Pediatrics. 2020;146(4):e2020006460.
3. Katz SE et al. J Pediatric Infect Dis Soc. 2021;10(1):62-4.
4. Cohen R et al. Infect. Dis Now. 2021; 51(5)418-23.
A secondary consequence of public health measures to prevent the spread of SARS-CoV-2 included a concurrent reduction in risk for children to acquire and spread other respiratory viral infectious diseases. In the Rochester, N.Y., area, we had an ongoing prospective study in primary care pediatric practices that afforded an opportunity to assess the effect of the pandemic control measures on all infectious disease illness visits in young children. Specifically, in children aged 6-36 months old, our study was in place when the pandemic began with a primary objective to evaluate the changing epidemiology of acute otitis media (AOM) and nasopharyngeal colonization by potential bacterial respiratory pathogens in community-based primary care pediatric practices. As the public health measures mandated by New York State Department of Health were implemented, we prospectively quantified their effect on physician-diagnosed infectious disease illness visits. The incidence of infectious disease visits by a cohort of young children during the COVID-19 pandemic period March 15, 2020, through Dec. 31, 2020, was compared with the same time frame in the preceding year, 2019.1
Recommendations of the New York State Department of Health for public health, changes in school and day care attendance, and clinical practice during the study time frame
On March 7, 2020, a state of emergency was declared in New York because of the COVID-19 pandemic. All schools were required to close. A mandated order for public use of masks in adults and children more than 2 years of age was enacted. In the Finger Lakes region of Upstate New York, where the two primary care pediatric practices reside, complete lockdown was partially lifted on May 15, 2020, and further lifted on June 26, 2020. Almost all regional school districts opened to at least hybrid learning models for all students starting Sept. 8, 2020. On March 6, 2020, video telehealth and telephone call visits were introduced as routine practice. Well-child visits were limited to those less than 2 years of age, then gradually expanded to all ages by late May 2020. During the “stay at home” phase of the New York State lockdown, day care services were considered an essential business. Day care child density was limited. All children less than 2 years old were required to wear a mask while in the facility. Upon arrival, children with any respiratory symptoms or fever were excluded. For the school year commencing September 2020, almost all regional school districts opened to virtual, hybrid, or in-person learning models. Exclusion occurred similar to that of the day care facilities.
Incidence of respiratory infectious disease illnesses
Clinical diagnoses and healthy visits of 144 children from March 15 to Dec. 31, 2020 (beginning of the pandemic) were compared to 215 children during the same months in 2019 (prepandemic). Pediatric SARS-CoV-2 positivity rates trended up alongside community spread. Pediatric practice positivity rates rose from 1.9% in October 2020 to 19% in December 2020.
The table shows the incidence of significantly different infectious disease illness visits in the two study cohorts.
During the pandemic, 258 infection visits occurred among 144 pandemic cohort children, compared with 687 visits among 215 prepandemic cohort children, a 1.8-fold decrease (P < .0001). The proportion of children with visits for AOM (3.7-fold; P < .0001), bronchiolitis (7.4-fold; P = .036), croup (27.5-fold; P < .0001), and viral upper respiratory infection (3.8-fold; P < .0001) decreased significantly. Fever without a source (1.4-fold decrease; P = .009) and skin/soft tissue infection (2.1-fold decrease; P = .042) represented a higher proportion of visits during the pandemic.
Prescription of antibiotics significantly decreased (P < .001) during the pandemic.
Change in care practices
In the prepandemic period, virtual visits, leading to a diagnosis and treatment and referring children to an urgent care or hospital emergency department during regular office hours were rare. During the pandemic, this changed. Significantly increased use of telemedicine visits (P < .0001) and significantly decreased office and urgent care visits (P < .0001) occurred during the pandemic. Telehealth visits peaked the week of April 12, 2020, at 45% of all pediatric visits. In-person illness visits gradually returned to year-to-year volumes in August-September 2020 with school opening. Early in the pandemic, both pediatric offices limited patient encounters to well-child visits in the first 2 years of life to not miss opportunities for childhood vaccinations. However, some parents were reluctant to bring their children to those visits. There was no significant change in frequency of healthy child visits during the pandemic.
To our knowledge, this was the first study from primary care pediatric practices in the United States to analyze the effect on infectious diseases during the first 9 months of the pandemic, including the 6-month time period after the reopening from the first 3 months of lockdown. One prior study from a primary care network in Massachusetts reported significant decreases in respiratory infectious diseases for children aged 0-17 years during the first months of the pandemic during lockdown.2 A study in Tennessee that included hospital emergency department, urgent care, primary care, and retail health clinics also reported respiratory infection diagnoses as well as antibiotic prescription were reduced in the early months of the pandemic.3
Our study shows an overall reduction in frequency of respiratory illness visits in children 6-36 months old during the first 9 months of the COVID-19 pandemic. We learned the value of using technology in the form of virtual visits to render care. Perhaps as the pandemic subsides, many of the hand-washing and sanitizing practices will remain in place and lead to less frequent illness in children in the future. However, there may be temporary negative consequences from the “immune debt” that has occurred from a prolonged time span when children were not becoming infected with respiratory pathogens.4 We will see what unfolds in the future.
Dr. Pichichero is a specialist in pediatric infectious diseases and director of the Research Institute at Rochester (N.Y.) General Hospital. Dr. Schulz is pediatric medical director at Rochester (N.Y.) Regional Health. Dr. Pichichero and Dr. Schulz have no conflicts of interest to disclose. This study was funded in part by the Centers for Disease Control and Prevention.
References
1. Kaur R et al. Front Pediatr. 2021;(9)722483:1-8.
2. Hatoun J et al. Pediatrics. 2020;146(4):e2020006460.
3. Katz SE et al. J Pediatric Infect Dis Soc. 2021;10(1):62-4.
4. Cohen R et al. Infect. Dis Now. 2021; 51(5)418-23.