User login
The cell that might trigger Alzheimer’s disease
It all started with genetic data. A gene here, a gene there. Eventually the story became clearer: If scientists are to one day find a cure for Alzheimer’s disease, they should look to the immune system.
Over the past couple decades, researchers have identified numerous genes involved in various immune system functions that may also contribute to Alzheimer’s disease. Some of the prime suspects are genes that control microglia, now the focus of intense research in developing new Alzheimer’s drugs.
Microglia are amoeba-like cells that scour the brain for injuries and invaders. They help clear dead or impaired brain cells and literally gobble up invading microbes. Without them, we’d be in trouble.
In a normal brain, a protein called beta-amyloid is cleared away through our lymphatic system by microglia as molecular junk. But sometimes it builds up. Certain gene mutations are one culprit in this toxic accumulation. Traumatic brain injury is another, and, perhaps, impaired microglial function.
One thing everyone agrees on is that in people with Alzheimer’s disease, too much amyloid accumulates between their brain cells and in the vessels that supply the brain with blood. Once amyloid begins to clog networks of neurons, it triggers the accumulation of another protein, called tau, inside of these brain cells. The presence of tau sends microglia and other immune mechanisms into overdrive, resulting in the inflammatory immune response that many experts believe ultimately saps brain vitality in Alzheimer’s disease.
The gene scene
To date, nearly a dozen genes involved in immune and microglial function have been tied to Alzheimer’s disease. The first was CD33, identified in 2008.
“When we got the results, I literally ran to my colleague’s office next door and said, you gotta see this!” said Harvard neuroscientist Rudolph Tanzi. Dr. Tanzi, who goes by Rudy, led the CD33 research. The discovery was quickly named a top medical breakthrough of 2008 by Time magazine.
“We were laughing because what they didn’t know is we had no idea what this gene did,” he joked. But over time, research by Dr. Tanzi and his group revealed that CD33 is a kind of microglial on-off switch, activating the cells as part of an inflammatory pathway.
“We kind of got it all going when it came to the genetics,” he said.
Microglia normally recognize molecular patterns associated with microbes and cellular damage as unwanted. This is how they know to take action – to devour unfamiliar pathogens and dead tissue. Dr. Tanzi believes microglia sense any sign of brain damage as an infection, which causes them to become hyperactive.
Much of our modern human immune system, he explained, evolved many hundreds of thousands of years ago. Our lifespans at the time were far shorter than they are today, and the majority of people didn’t live long enough to develop dementia or the withered brain cells that come with it. So our immune system, he said, assumes any faulty brain tissue is due to a microbe, not dementia. Microglia react aggressively, clearing the area to prevent the spread of infection.
“They say, ‘We better wipe out this part of the brain that’s infected, even if it’s not.’ They don’t know,” quipped Dr. Tanzi. “That’s what causes neuroinflammation. And CD33 turns this response on. The microglia become killers, not just janitors.”
A brake on overactive microglia
If CD33 is the yin, a gene called TREM2 is the yang. Discovered a few years after CD33, TREM2 reins in microglial activation, returning them to their role as cellular housekeepers.
Neurologist David Holtzman, MD, of Washington University in St. Louis, who studies TREM2, agrees that wherever you find amyloid, tau, or dead brain cells, there are microglia raring to go and ready to scavenge.
“I think at first a lot of people thought these cells were reacting to Alzheimer’s pathology, and not necessarily a cause of the disease,” he said.
It was the discovery of TREM2 on the heels of CD33 that really shifted the thinking, in part because it produces a protein that in the brain is only found in microglia. “Many of us [in the field] immediately said, ‘Look, there’s now a risk factor that is only expressed in microglia. It must be that innate immune cells are important in some way in the pathogenesis of the disease,’ “ he added.
Dr. Holtzman sees microglial activation in impending dementia as a double-edged sword. In the beginning, microglia clear unwanted amyloid to maintain brain health. But once accumulated amyloid and tau have done enough damage, the neuroinflammation that comes with microglial activation does more harm than good. Neurons die en masse and dementia sets in.
But not all researchers are convinced.
Serge Revist, PhD, is a professor in the department of molecular medicine at the Laval University Medical School in Quebec. Based on his lab’s research, he believes that while impaired immune activity is involved in Alzheimer’s disease, it is not the root cause. “I don’t think it is the immune cells that do the damage, I still think it is the beta-amyloid itself,” he said, “In my lab, in mouse studies, we’ve never found that immune cells were directly responsible for killing neurons.”
He does believe that in some patients with Alzheimer’s disease, microglia may not be able to handle the excess amyloid that accumulates in the disease and that developing treatments that improve the ability of microglia and the immune system to clear the protein could be effective.
Microglial medicines
The biological cascade leading to Alzheimer’s disease is a tangled one. Gene variants influencing the accumulation and clearance of amyloid are likely a major contributor. But immune activity caused by early life infection might also be involved, at least in some cases. This infectious theory of Alzheimer’s disease was first proposed by Dr. Tanzi’s now-deceased colleague Robert Moir, PhD. Dr. Tanzi’s group even has evidence that amyloid itself is antimicrobial and evolved to protect us from pathogens, only to become a problem when overactive and aggregated.
And the same goes for microglia, cells whose over-ambition might cause much of the brain degeneration seen in Alzheimer’s disease.
In theory, if a treatment could decrease CD33 activity or increase that of TREM2, doctors might one day may be able to slow or even stop the progression of dementia. Instead of going after amyloid itself – the mechanism behind so many failed investigational Alzheimer’s drugs – a therapy that quells the immune response to amyloid might be the answer in treating dementia.
“There are a number of scientists and companies trying to figure out how to influence genes like TREM2 and CD33 and to both decrease amyloid and act on the downstream consequences of the protein,” said Dr. Holtzman. “All of this is to say that somewhere in the biology that causes Alzheimer’s disease, the immune system is involved.”
It seems that in many cases, the most common form of a dementia might be due to a well-intentioned immune cell going rogue. “I think you’d hear this from basically any researcher worth their salt,” said Dr. Tanzi. “I feel strongly that without microglial activation, you will not get Alzheimer’s disease.”
A version of this article first appeared on Medscape.com.
It all started with genetic data. A gene here, a gene there. Eventually the story became clearer: If scientists are to one day find a cure for Alzheimer’s disease, they should look to the immune system.
Over the past couple decades, researchers have identified numerous genes involved in various immune system functions that may also contribute to Alzheimer’s disease. Some of the prime suspects are genes that control microglia, now the focus of intense research in developing new Alzheimer’s drugs.
Microglia are amoeba-like cells that scour the brain for injuries and invaders. They help clear dead or impaired brain cells and literally gobble up invading microbes. Without them, we’d be in trouble.
In a normal brain, a protein called beta-amyloid is cleared away through our lymphatic system by microglia as molecular junk. But sometimes it builds up. Certain gene mutations are one culprit in this toxic accumulation. Traumatic brain injury is another, and, perhaps, impaired microglial function.
One thing everyone agrees on is that in people with Alzheimer’s disease, too much amyloid accumulates between their brain cells and in the vessels that supply the brain with blood. Once amyloid begins to clog networks of neurons, it triggers the accumulation of another protein, called tau, inside of these brain cells. The presence of tau sends microglia and other immune mechanisms into overdrive, resulting in the inflammatory immune response that many experts believe ultimately saps brain vitality in Alzheimer’s disease.
The gene scene
To date, nearly a dozen genes involved in immune and microglial function have been tied to Alzheimer’s disease. The first was CD33, identified in 2008.
“When we got the results, I literally ran to my colleague’s office next door and said, you gotta see this!” said Harvard neuroscientist Rudolph Tanzi. Dr. Tanzi, who goes by Rudy, led the CD33 research. The discovery was quickly named a top medical breakthrough of 2008 by Time magazine.
“We were laughing because what they didn’t know is we had no idea what this gene did,” he joked. But over time, research by Dr. Tanzi and his group revealed that CD33 is a kind of microglial on-off switch, activating the cells as part of an inflammatory pathway.
“We kind of got it all going when it came to the genetics,” he said.
Microglia normally recognize molecular patterns associated with microbes and cellular damage as unwanted. This is how they know to take action – to devour unfamiliar pathogens and dead tissue. Dr. Tanzi believes microglia sense any sign of brain damage as an infection, which causes them to become hyperactive.
Much of our modern human immune system, he explained, evolved many hundreds of thousands of years ago. Our lifespans at the time were far shorter than they are today, and the majority of people didn’t live long enough to develop dementia or the withered brain cells that come with it. So our immune system, he said, assumes any faulty brain tissue is due to a microbe, not dementia. Microglia react aggressively, clearing the area to prevent the spread of infection.
“They say, ‘We better wipe out this part of the brain that’s infected, even if it’s not.’ They don’t know,” quipped Dr. Tanzi. “That’s what causes neuroinflammation. And CD33 turns this response on. The microglia become killers, not just janitors.”
A brake on overactive microglia
If CD33 is the yin, a gene called TREM2 is the yang. Discovered a few years after CD33, TREM2 reins in microglial activation, returning them to their role as cellular housekeepers.
Neurologist David Holtzman, MD, of Washington University in St. Louis, who studies TREM2, agrees that wherever you find amyloid, tau, or dead brain cells, there are microglia raring to go and ready to scavenge.
“I think at first a lot of people thought these cells were reacting to Alzheimer’s pathology, and not necessarily a cause of the disease,” he said.
It was the discovery of TREM2 on the heels of CD33 that really shifted the thinking, in part because it produces a protein that in the brain is only found in microglia. “Many of us [in the field] immediately said, ‘Look, there’s now a risk factor that is only expressed in microglia. It must be that innate immune cells are important in some way in the pathogenesis of the disease,’ “ he added.
Dr. Holtzman sees microglial activation in impending dementia as a double-edged sword. In the beginning, microglia clear unwanted amyloid to maintain brain health. But once accumulated amyloid and tau have done enough damage, the neuroinflammation that comes with microglial activation does more harm than good. Neurons die en masse and dementia sets in.
But not all researchers are convinced.
Serge Revist, PhD, is a professor in the department of molecular medicine at the Laval University Medical School in Quebec. Based on his lab’s research, he believes that while impaired immune activity is involved in Alzheimer’s disease, it is not the root cause. “I don’t think it is the immune cells that do the damage, I still think it is the beta-amyloid itself,” he said, “In my lab, in mouse studies, we’ve never found that immune cells were directly responsible for killing neurons.”
He does believe that in some patients with Alzheimer’s disease, microglia may not be able to handle the excess amyloid that accumulates in the disease and that developing treatments that improve the ability of microglia and the immune system to clear the protein could be effective.
Microglial medicines
The biological cascade leading to Alzheimer’s disease is a tangled one. Gene variants influencing the accumulation and clearance of amyloid are likely a major contributor. But immune activity caused by early life infection might also be involved, at least in some cases. This infectious theory of Alzheimer’s disease was first proposed by Dr. Tanzi’s now-deceased colleague Robert Moir, PhD. Dr. Tanzi’s group even has evidence that amyloid itself is antimicrobial and evolved to protect us from pathogens, only to become a problem when overactive and aggregated.
And the same goes for microglia, cells whose over-ambition might cause much of the brain degeneration seen in Alzheimer’s disease.
In theory, if a treatment could decrease CD33 activity or increase that of TREM2, doctors might one day may be able to slow or even stop the progression of dementia. Instead of going after amyloid itself – the mechanism behind so many failed investigational Alzheimer’s drugs – a therapy that quells the immune response to amyloid might be the answer in treating dementia.
“There are a number of scientists and companies trying to figure out how to influence genes like TREM2 and CD33 and to both decrease amyloid and act on the downstream consequences of the protein,” said Dr. Holtzman. “All of this is to say that somewhere in the biology that causes Alzheimer’s disease, the immune system is involved.”
It seems that in many cases, the most common form of a dementia might be due to a well-intentioned immune cell going rogue. “I think you’d hear this from basically any researcher worth their salt,” said Dr. Tanzi. “I feel strongly that without microglial activation, you will not get Alzheimer’s disease.”
A version of this article first appeared on Medscape.com.
It all started with genetic data. A gene here, a gene there. Eventually the story became clearer: If scientists are to one day find a cure for Alzheimer’s disease, they should look to the immune system.
Over the past couple decades, researchers have identified numerous genes involved in various immune system functions that may also contribute to Alzheimer’s disease. Some of the prime suspects are genes that control microglia, now the focus of intense research in developing new Alzheimer’s drugs.
Microglia are amoeba-like cells that scour the brain for injuries and invaders. They help clear dead or impaired brain cells and literally gobble up invading microbes. Without them, we’d be in trouble.
In a normal brain, a protein called beta-amyloid is cleared away through our lymphatic system by microglia as molecular junk. But sometimes it builds up. Certain gene mutations are one culprit in this toxic accumulation. Traumatic brain injury is another, and, perhaps, impaired microglial function.
One thing everyone agrees on is that in people with Alzheimer’s disease, too much amyloid accumulates between their brain cells and in the vessels that supply the brain with blood. Once amyloid begins to clog networks of neurons, it triggers the accumulation of another protein, called tau, inside of these brain cells. The presence of tau sends microglia and other immune mechanisms into overdrive, resulting in the inflammatory immune response that many experts believe ultimately saps brain vitality in Alzheimer’s disease.
The gene scene
To date, nearly a dozen genes involved in immune and microglial function have been tied to Alzheimer’s disease. The first was CD33, identified in 2008.
“When we got the results, I literally ran to my colleague’s office next door and said, you gotta see this!” said Harvard neuroscientist Rudolph Tanzi. Dr. Tanzi, who goes by Rudy, led the CD33 research. The discovery was quickly named a top medical breakthrough of 2008 by Time magazine.
“We were laughing because what they didn’t know is we had no idea what this gene did,” he joked. But over time, research by Dr. Tanzi and his group revealed that CD33 is a kind of microglial on-off switch, activating the cells as part of an inflammatory pathway.
“We kind of got it all going when it came to the genetics,” he said.
Microglia normally recognize molecular patterns associated with microbes and cellular damage as unwanted. This is how they know to take action – to devour unfamiliar pathogens and dead tissue. Dr. Tanzi believes microglia sense any sign of brain damage as an infection, which causes them to become hyperactive.
Much of our modern human immune system, he explained, evolved many hundreds of thousands of years ago. Our lifespans at the time were far shorter than they are today, and the majority of people didn’t live long enough to develop dementia or the withered brain cells that come with it. So our immune system, he said, assumes any faulty brain tissue is due to a microbe, not dementia. Microglia react aggressively, clearing the area to prevent the spread of infection.
“They say, ‘We better wipe out this part of the brain that’s infected, even if it’s not.’ They don’t know,” quipped Dr. Tanzi. “That’s what causes neuroinflammation. And CD33 turns this response on. The microglia become killers, not just janitors.”
A brake on overactive microglia
If CD33 is the yin, a gene called TREM2 is the yang. Discovered a few years after CD33, TREM2 reins in microglial activation, returning them to their role as cellular housekeepers.
Neurologist David Holtzman, MD, of Washington University in St. Louis, who studies TREM2, agrees that wherever you find amyloid, tau, or dead brain cells, there are microglia raring to go and ready to scavenge.
“I think at first a lot of people thought these cells were reacting to Alzheimer’s pathology, and not necessarily a cause of the disease,” he said.
It was the discovery of TREM2 on the heels of CD33 that really shifted the thinking, in part because it produces a protein that in the brain is only found in microglia. “Many of us [in the field] immediately said, ‘Look, there’s now a risk factor that is only expressed in microglia. It must be that innate immune cells are important in some way in the pathogenesis of the disease,’ “ he added.
Dr. Holtzman sees microglial activation in impending dementia as a double-edged sword. In the beginning, microglia clear unwanted amyloid to maintain brain health. But once accumulated amyloid and tau have done enough damage, the neuroinflammation that comes with microglial activation does more harm than good. Neurons die en masse and dementia sets in.
But not all researchers are convinced.
Serge Revist, PhD, is a professor in the department of molecular medicine at the Laval University Medical School in Quebec. Based on his lab’s research, he believes that while impaired immune activity is involved in Alzheimer’s disease, it is not the root cause. “I don’t think it is the immune cells that do the damage, I still think it is the beta-amyloid itself,” he said, “In my lab, in mouse studies, we’ve never found that immune cells were directly responsible for killing neurons.”
He does believe that in some patients with Alzheimer’s disease, microglia may not be able to handle the excess amyloid that accumulates in the disease and that developing treatments that improve the ability of microglia and the immune system to clear the protein could be effective.
Microglial medicines
The biological cascade leading to Alzheimer’s disease is a tangled one. Gene variants influencing the accumulation and clearance of amyloid are likely a major contributor. But immune activity caused by early life infection might also be involved, at least in some cases. This infectious theory of Alzheimer’s disease was first proposed by Dr. Tanzi’s now-deceased colleague Robert Moir, PhD. Dr. Tanzi’s group even has evidence that amyloid itself is antimicrobial and evolved to protect us from pathogens, only to become a problem when overactive and aggregated.
And the same goes for microglia, cells whose over-ambition might cause much of the brain degeneration seen in Alzheimer’s disease.
In theory, if a treatment could decrease CD33 activity or increase that of TREM2, doctors might one day may be able to slow or even stop the progression of dementia. Instead of going after amyloid itself – the mechanism behind so many failed investigational Alzheimer’s drugs – a therapy that quells the immune response to amyloid might be the answer in treating dementia.
“There are a number of scientists and companies trying to figure out how to influence genes like TREM2 and CD33 and to both decrease amyloid and act on the downstream consequences of the protein,” said Dr. Holtzman. “All of this is to say that somewhere in the biology that causes Alzheimer’s disease, the immune system is involved.”
It seems that in many cases, the most common form of a dementia might be due to a well-intentioned immune cell going rogue. “I think you’d hear this from basically any researcher worth their salt,” said Dr. Tanzi. “I feel strongly that without microglial activation, you will not get Alzheimer’s disease.”
A version of this article first appeared on Medscape.com.
Psychedelic therapy and suicide: A myth busted?
A commonly held belief that classic psychedelic therapy can trigger suicidal thoughts, actions, or other types of self-harm is not supported by research, and, in fact, the opposite may be true.
Results from a meta-analysis of individual patient data showed that psychedelic therapy was associated with large, acute, and sustained decreases in suicidality across a range of clinical patient populations.
“ It gives us a better understanding of the effects of psychedelics on suicidality in the context of clinical trials,” study investigator Cory Weissman, MD, department of psychiatry, University of Toronto, told this news organization.
The evidence suggests psychedelic therapy “may reduce suicidal ideation when administered in the appropriate setting and offered to carefully screened patients,” Dr. Weissman said.
The findings were published online Jan. 18 in The Journal of Clinical Psychiatry.
More research needed
The analysis included seven psychedelic therapy clinical trials that had data on suicidality. Five of the trials used psilocybin plus psychotherapy and two used ayahuasca plus psychotherapy. All seven trials had a “low” risk of bias.
Patients included in the trials had treatment-resistant major depressive disorder (MDD), recurrent MDD, AIDS-related demoralization, and distress related to life-threatening cancer.
The meta-analytic results showed significant decreases in suicidality at all acute time points (80 to 240 minutes post administration) and at most post-acute time points (1 day to 4 months post administration).
Effect sizes for reductions in suicidality were “large” at all acute time points, with standardized mean differences (SMD) ranging from -1.48 to -1.72, and remained large from 1 day to 3-4 months after therapy (SMD range, -1.50 to -2.36).
At 6 months, the effect size for reductions in suicidality with psychedelic therapy was “medium” (SMD, -0.65).
Large effect sizes for reductions in suicidality occurred across the different patient populations represented in the trial, the investigators note.
No study reported any suicide-related adverse events because of administration of a psychedelic. There were also “very few” acute (6.5%) or postacute (3.0%) elevations in suicidality, “providing support for the safety of psychedelic therapy within controlled contexts,” the researchers write.
They caution, however, that large controlled trials that specifically evaluate the effect of psychedelic therapy on suicidality are needed.
Promising avenue
In an accompanying editorial, Daniel Grossman, BS, and Peter Hendricks, PhD, department of health behavior, University of Alabama at Birmingham, note that results of this review warrant “optimism” for use of psychedelics for treatment of suicidality.
Based on this study and others, classic psychedelic therapy for suicidality appears to be a “promising avenue” for further investigation, they write.
However, research and anecdotes about increased suicidality and other self-harm attributed to psychedelic therapy, “though evidently rare, remain a critical concern” for further research to address, Dr. Grossman and Dr. Hendricks add.
The hope is that future research “clarifies who is most subject to these risks, what factors best identify them, and how best to navigate their treatment safely,” they write.
The meta-analysis had no funding. Dr. Weissman receives funding from the Brain and Behavior Research Foundation and serves on the advisory board of GoodCap Pharmaceuticals. Dr. Hendricks is on the scientific advisory board of Bright Minds Biosciences, Eleusis Benefit Corporation, and Rest Pharmaceuticals.
A version of this article first appeared on Medscape.com.
A commonly held belief that classic psychedelic therapy can trigger suicidal thoughts, actions, or other types of self-harm is not supported by research, and, in fact, the opposite may be true.
Results from a meta-analysis of individual patient data showed that psychedelic therapy was associated with large, acute, and sustained decreases in suicidality across a range of clinical patient populations.
“ It gives us a better understanding of the effects of psychedelics on suicidality in the context of clinical trials,” study investigator Cory Weissman, MD, department of psychiatry, University of Toronto, told this news organization.
The evidence suggests psychedelic therapy “may reduce suicidal ideation when administered in the appropriate setting and offered to carefully screened patients,” Dr. Weissman said.
The findings were published online Jan. 18 in The Journal of Clinical Psychiatry.
More research needed
The analysis included seven psychedelic therapy clinical trials that had data on suicidality. Five of the trials used psilocybin plus psychotherapy and two used ayahuasca plus psychotherapy. All seven trials had a “low” risk of bias.
Patients included in the trials had treatment-resistant major depressive disorder (MDD), recurrent MDD, AIDS-related demoralization, and distress related to life-threatening cancer.
The meta-analytic results showed significant decreases in suicidality at all acute time points (80 to 240 minutes post administration) and at most post-acute time points (1 day to 4 months post administration).
Effect sizes for reductions in suicidality were “large” at all acute time points, with standardized mean differences (SMD) ranging from -1.48 to -1.72, and remained large from 1 day to 3-4 months after therapy (SMD range, -1.50 to -2.36).
At 6 months, the effect size for reductions in suicidality with psychedelic therapy was “medium” (SMD, -0.65).
Large effect sizes for reductions in suicidality occurred across the different patient populations represented in the trial, the investigators note.
No study reported any suicide-related adverse events because of administration of a psychedelic. There were also “very few” acute (6.5%) or postacute (3.0%) elevations in suicidality, “providing support for the safety of psychedelic therapy within controlled contexts,” the researchers write.
They caution, however, that large controlled trials that specifically evaluate the effect of psychedelic therapy on suicidality are needed.
Promising avenue
In an accompanying editorial, Daniel Grossman, BS, and Peter Hendricks, PhD, department of health behavior, University of Alabama at Birmingham, note that results of this review warrant “optimism” for use of psychedelics for treatment of suicidality.
Based on this study and others, classic psychedelic therapy for suicidality appears to be a “promising avenue” for further investigation, they write.
However, research and anecdotes about increased suicidality and other self-harm attributed to psychedelic therapy, “though evidently rare, remain a critical concern” for further research to address, Dr. Grossman and Dr. Hendricks add.
The hope is that future research “clarifies who is most subject to these risks, what factors best identify them, and how best to navigate their treatment safely,” they write.
The meta-analysis had no funding. Dr. Weissman receives funding from the Brain and Behavior Research Foundation and serves on the advisory board of GoodCap Pharmaceuticals. Dr. Hendricks is on the scientific advisory board of Bright Minds Biosciences, Eleusis Benefit Corporation, and Rest Pharmaceuticals.
A version of this article first appeared on Medscape.com.
A commonly held belief that classic psychedelic therapy can trigger suicidal thoughts, actions, or other types of self-harm is not supported by research, and, in fact, the opposite may be true.
Results from a meta-analysis of individual patient data showed that psychedelic therapy was associated with large, acute, and sustained decreases in suicidality across a range of clinical patient populations.
“ It gives us a better understanding of the effects of psychedelics on suicidality in the context of clinical trials,” study investigator Cory Weissman, MD, department of psychiatry, University of Toronto, told this news organization.
The evidence suggests psychedelic therapy “may reduce suicidal ideation when administered in the appropriate setting and offered to carefully screened patients,” Dr. Weissman said.
The findings were published online Jan. 18 in The Journal of Clinical Psychiatry.
More research needed
The analysis included seven psychedelic therapy clinical trials that had data on suicidality. Five of the trials used psilocybin plus psychotherapy and two used ayahuasca plus psychotherapy. All seven trials had a “low” risk of bias.
Patients included in the trials had treatment-resistant major depressive disorder (MDD), recurrent MDD, AIDS-related demoralization, and distress related to life-threatening cancer.
The meta-analytic results showed significant decreases in suicidality at all acute time points (80 to 240 minutes post administration) and at most post-acute time points (1 day to 4 months post administration).
Effect sizes for reductions in suicidality were “large” at all acute time points, with standardized mean differences (SMD) ranging from -1.48 to -1.72, and remained large from 1 day to 3-4 months after therapy (SMD range, -1.50 to -2.36).
At 6 months, the effect size for reductions in suicidality with psychedelic therapy was “medium” (SMD, -0.65).
Large effect sizes for reductions in suicidality occurred across the different patient populations represented in the trial, the investigators note.
No study reported any suicide-related adverse events because of administration of a psychedelic. There were also “very few” acute (6.5%) or postacute (3.0%) elevations in suicidality, “providing support for the safety of psychedelic therapy within controlled contexts,” the researchers write.
They caution, however, that large controlled trials that specifically evaluate the effect of psychedelic therapy on suicidality are needed.
Promising avenue
In an accompanying editorial, Daniel Grossman, BS, and Peter Hendricks, PhD, department of health behavior, University of Alabama at Birmingham, note that results of this review warrant “optimism” for use of psychedelics for treatment of suicidality.
Based on this study and others, classic psychedelic therapy for suicidality appears to be a “promising avenue” for further investigation, they write.
However, research and anecdotes about increased suicidality and other self-harm attributed to psychedelic therapy, “though evidently rare, remain a critical concern” for further research to address, Dr. Grossman and Dr. Hendricks add.
The hope is that future research “clarifies who is most subject to these risks, what factors best identify them, and how best to navigate their treatment safely,” they write.
The meta-analysis had no funding. Dr. Weissman receives funding from the Brain and Behavior Research Foundation and serves on the advisory board of GoodCap Pharmaceuticals. Dr. Hendricks is on the scientific advisory board of Bright Minds Biosciences, Eleusis Benefit Corporation, and Rest Pharmaceuticals.
A version of this article first appeared on Medscape.com.
What docs don’t know about the Disabilities Act can hurt them and patients
Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School and a disability researcher at Massachusetts General Hospital, both in Boston, has used a wheelchair for more than 30 years because of multiple sclerosis. When she visits her primary care doctor, she doesn’t get weighed because the scales are not wheelchair accessible.
This failure to weigh her and other patients in wheelchairs could lead to serious medical problems. Weight is used to monitor a person’s overall health and prenatal health and to determine accurate doses for medications such as some chemotherapies, said Dr. Iezzoni.
In another situation, a man who used a wheelchair said that his primary care doctor never got him out of it for a complete physical exam. The patient later developed lymphoma, which first appeared in his groin. The doctor should have accommodated his disability and used a height-adjustable exam table or a portable lift to transfer him onto the table.
When physicians don’t provide access to medical care that patients with disabilities need, they put themselves at greater risk of lawsuits, fines, and settlements.
Yet, a new study in Health Affairs suggests that a large percentage of doctors are not fully aware of what they are legally required to do.
Under federal nondiscrimination laws (Americans With Disabilities Act, American Rehabilitation Act, and ADA Amendments Act), medical practices must provide equal access to people with disabilities, accommodate their disability-related needs, and not refuse them medical services because of their disabilities, say disability experts.
Where doctors go wrong with disability laws
What doctors don’t know about providing reasonable accommodations makes them vulnerable to lawsuits, which worries more than two-thirds of the 714 outpatient doctors surveyed.
Not only are they required to provide reasonable accommodations, but they also have to pay for them, the researchers said. One-fifth of the surveyed doctors said they didn’t know that practice owners have to pay.
More than one practice has made patients pay for services needed for their disability, such as sign language interpreters – the patients later complained this violated the ADA to enforcement agencies.
Doctors also don’t know that they have to collaborate with patients to determine what reasonable accommodations they need – over two-thirds of those surveyed said they didn’t know it was a joint responsibility, the study found.
When doctors fail to accommodate patients’ disability needs, they engage in discrimination and violate the ADA, says Elizabeth Pendo, JD, a coauthor of the study and the Joseph J. Simeone Professor of Law at Saint Louis University.
The Department of Justice has investigated several patient complaints of alleged disability discrimination recently and resolved the disputes with agreements and small fines in some cases. “The goal is not to get large financial settlements but to work with practices to get the correct procedures in place to be compliant,” said Ms. Pendo.
Physicians would be wise to check out whether their practices are as accessible as they think. Even if there’s a ramp to the office building, the parking lot may not have a van-accessible space or enough handicapped parking signs, or the exam room may be too narrow for a wheelchair to navigate.
These practices violated the ADA and agreed to make changes:
- Hamden, Conn., has two buildings that patients with physical disabilities couldn’t easily enter. The physician owners agreed to change the buildings’ entrances and access routes and add features to make it easier to use examination rooms and restrooms and the check-in and check-out areas.
- Seven medical offices in Riverside, Calif., failed to communicate effectively with deaf and hard-of-hearing patients. They should have had a qualified sign language interpreter, an assistive listening device, or another appropriate aid or service available to a deaf patient and her family. Instead, the office relied on a video remote interpretation system that often failed to work. The agreement requires the clinic to provide those aids and services to patients and their companions who are deaf or hard of hearing, advertise their availability, assess each patient who is deaf or hard of hearing to determine the best aids and services for their needs, and pay $5,000 in compensation to the complainant and a $1,000 civil penalty to the United States.
- Springfield, Mass., refused to provide full joint replacements to two patients being treated with buprenorphine, a medication used to treat opioid use disorder. Rather than accommodate the patients, the surgeons referred them elsewhere because they were uncomfortable with the postoperative pain management protocol for patients prescribed buprenorphine. “The Americans With Disabilities Act protects health care access for people under medical treatment for opioid use disorder,” said Acting U.S. Attorney Nathaniel R. Mendell. “Health care providers must comply with the ADA, even when doing so is inconvenient or makes them uncomfortable.” The agreement requires the practice to adopt a nondiscrimination policy, provide training on the ADA and opioid use disorder, and pay two complainants $15,000 each for pain and suffering.
The DOJ has filed civil lawsuits against medical practices when they failed to resolve the allegations. Recent cases include an ophthalmology practice with 24 facilities in Arizona that refused to help transfer patients in wheelchairs to surgery tables for eye surgery and required them to pay for transfer support services and two obstetricians-gynecologists in Bakersfield, Calif., who refused to provide routine medical care to a patient because of her HIV status.
What doctors should know
Many people tend to think of a person with a disability as being in a wheelchair. But the ADA has a very broad definition of disability, which includes any physical or mental impairment that substantially limits any major life activity, said Ms. Pendo.
“It was amended in 2008 to clarify that the definition includes people with chronic diseases such as diabetes and cancer, cognitive and neurological disorders, substance abuse disorders, vision and hearing loss, and learning and other disabilities,” she said.
That means that doctors have to accommodate many types of disabilities, which can be challenging. The ADA only specifies that fixed structures need to be accessible, such as parking lots, driveways, and buildings, said Dr. Iezzoni.
When it comes to “reasonable accommodations,” doctors should decide that on a case-by-case basis, she said.
“We can say based on our study that 71% of doctors don’t know the right way to think about the accommodations – they don’t know they need to talk to patients so they can explain to them exactly what they need to accommodate their disability,” said Dr. Iezzoni.
Doctors are also required to provide effective communication for patients with sensory or cognitive disabilities, which can depend on the severity, said Ms. Pendo. Is the person deaf or hard of hearing, blind or partially sighted – is the dementia mild or severe?
“The requirement is there, but what that looks like will vary by patient. That’s what’s challenging,” said Ms. Pendo.
Dr. Iezzoni recommends that doctor’s offices ask patients whether they need special help or individual assistance when they make appointments and enter their responses in their records. She also suggests that patients be asked at follow-up appointments whether they still need the same help or not.
“Disabilities can change over time – a person with bad arthritis may need help getting onto an exam table, but later get a knee or hip replacement that is effective and no longer need that help,” said Dr. Iezzoni.
Benefits outweigh costs
Physicians have made progress in meeting the ADA’s physical accessibility requirements, said Dr. Iezzoni. “The literature suggests that doctors have done a good job at fixing the structural barriers people with mobility issues face, such as ramps and bathrooms.”
However, there are exceptions in rural older buildings which can be harder to retrofit for wheelchair accessibility, she said. “I recall interviewing a rural doctor several years ago who said that he knew his patients well and when a patient visits with mobility problems, he goes down and carries the patient up the steps to his office. My response was that is not respectful of the patient or safe for the patient or you. That doctor has since changed the location of his practice,” said Dr. Iezzoni.
Some doctors may resist paying for accessible medical equipment because of cost, but she said the benefits are worth it. These include preventing staff injuries when they transfer patients and being used by patients with temporary disabilities and aging people with bad knees, backs, hearing and sight. In addition, businesses may be eligible for federal and state tax credits.
Dr. Iezzoni recently visited her doctor where they finally got height-adjustable exam tables. “I asked the assistant, who really likes these tables? She said it’s the elderly ladies of short stature – the table is lowered and they sit down and get on it.”
But, Dr. Iezonni’s main message to doctors is that patients with disabilities deserve equal quality of care. “Just because we have a disability doesn’t mean we should get worse care than other people. It’s a matter of professionalism that doctors should want to give the same quality care to all their patients.”
A version of this article first appeared on Medscape.com.
Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School and a disability researcher at Massachusetts General Hospital, both in Boston, has used a wheelchair for more than 30 years because of multiple sclerosis. When she visits her primary care doctor, she doesn’t get weighed because the scales are not wheelchair accessible.
This failure to weigh her and other patients in wheelchairs could lead to serious medical problems. Weight is used to monitor a person’s overall health and prenatal health and to determine accurate doses for medications such as some chemotherapies, said Dr. Iezzoni.
In another situation, a man who used a wheelchair said that his primary care doctor never got him out of it for a complete physical exam. The patient later developed lymphoma, which first appeared in his groin. The doctor should have accommodated his disability and used a height-adjustable exam table or a portable lift to transfer him onto the table.
When physicians don’t provide access to medical care that patients with disabilities need, they put themselves at greater risk of lawsuits, fines, and settlements.
Yet, a new study in Health Affairs suggests that a large percentage of doctors are not fully aware of what they are legally required to do.
Under federal nondiscrimination laws (Americans With Disabilities Act, American Rehabilitation Act, and ADA Amendments Act), medical practices must provide equal access to people with disabilities, accommodate their disability-related needs, and not refuse them medical services because of their disabilities, say disability experts.
Where doctors go wrong with disability laws
What doctors don’t know about providing reasonable accommodations makes them vulnerable to lawsuits, which worries more than two-thirds of the 714 outpatient doctors surveyed.
Not only are they required to provide reasonable accommodations, but they also have to pay for them, the researchers said. One-fifth of the surveyed doctors said they didn’t know that practice owners have to pay.
More than one practice has made patients pay for services needed for their disability, such as sign language interpreters – the patients later complained this violated the ADA to enforcement agencies.
Doctors also don’t know that they have to collaborate with patients to determine what reasonable accommodations they need – over two-thirds of those surveyed said they didn’t know it was a joint responsibility, the study found.
When doctors fail to accommodate patients’ disability needs, they engage in discrimination and violate the ADA, says Elizabeth Pendo, JD, a coauthor of the study and the Joseph J. Simeone Professor of Law at Saint Louis University.
The Department of Justice has investigated several patient complaints of alleged disability discrimination recently and resolved the disputes with agreements and small fines in some cases. “The goal is not to get large financial settlements but to work with practices to get the correct procedures in place to be compliant,” said Ms. Pendo.
Physicians would be wise to check out whether their practices are as accessible as they think. Even if there’s a ramp to the office building, the parking lot may not have a van-accessible space or enough handicapped parking signs, or the exam room may be too narrow for a wheelchair to navigate.
These practices violated the ADA and agreed to make changes:
- Hamden, Conn., has two buildings that patients with physical disabilities couldn’t easily enter. The physician owners agreed to change the buildings’ entrances and access routes and add features to make it easier to use examination rooms and restrooms and the check-in and check-out areas.
- Seven medical offices in Riverside, Calif., failed to communicate effectively with deaf and hard-of-hearing patients. They should have had a qualified sign language interpreter, an assistive listening device, or another appropriate aid or service available to a deaf patient and her family. Instead, the office relied on a video remote interpretation system that often failed to work. The agreement requires the clinic to provide those aids and services to patients and their companions who are deaf or hard of hearing, advertise their availability, assess each patient who is deaf or hard of hearing to determine the best aids and services for their needs, and pay $5,000 in compensation to the complainant and a $1,000 civil penalty to the United States.
- Springfield, Mass., refused to provide full joint replacements to two patients being treated with buprenorphine, a medication used to treat opioid use disorder. Rather than accommodate the patients, the surgeons referred them elsewhere because they were uncomfortable with the postoperative pain management protocol for patients prescribed buprenorphine. “The Americans With Disabilities Act protects health care access for people under medical treatment for opioid use disorder,” said Acting U.S. Attorney Nathaniel R. Mendell. “Health care providers must comply with the ADA, even when doing so is inconvenient or makes them uncomfortable.” The agreement requires the practice to adopt a nondiscrimination policy, provide training on the ADA and opioid use disorder, and pay two complainants $15,000 each for pain and suffering.
The DOJ has filed civil lawsuits against medical practices when they failed to resolve the allegations. Recent cases include an ophthalmology practice with 24 facilities in Arizona that refused to help transfer patients in wheelchairs to surgery tables for eye surgery and required them to pay for transfer support services and two obstetricians-gynecologists in Bakersfield, Calif., who refused to provide routine medical care to a patient because of her HIV status.
What doctors should know
Many people tend to think of a person with a disability as being in a wheelchair. But the ADA has a very broad definition of disability, which includes any physical or mental impairment that substantially limits any major life activity, said Ms. Pendo.
“It was amended in 2008 to clarify that the definition includes people with chronic diseases such as diabetes and cancer, cognitive and neurological disorders, substance abuse disorders, vision and hearing loss, and learning and other disabilities,” she said.
That means that doctors have to accommodate many types of disabilities, which can be challenging. The ADA only specifies that fixed structures need to be accessible, such as parking lots, driveways, and buildings, said Dr. Iezzoni.
When it comes to “reasonable accommodations,” doctors should decide that on a case-by-case basis, she said.
“We can say based on our study that 71% of doctors don’t know the right way to think about the accommodations – they don’t know they need to talk to patients so they can explain to them exactly what they need to accommodate their disability,” said Dr. Iezzoni.
Doctors are also required to provide effective communication for patients with sensory or cognitive disabilities, which can depend on the severity, said Ms. Pendo. Is the person deaf or hard of hearing, blind or partially sighted – is the dementia mild or severe?
“The requirement is there, but what that looks like will vary by patient. That’s what’s challenging,” said Ms. Pendo.
Dr. Iezzoni recommends that doctor’s offices ask patients whether they need special help or individual assistance when they make appointments and enter their responses in their records. She also suggests that patients be asked at follow-up appointments whether they still need the same help or not.
“Disabilities can change over time – a person with bad arthritis may need help getting onto an exam table, but later get a knee or hip replacement that is effective and no longer need that help,” said Dr. Iezzoni.
Benefits outweigh costs
Physicians have made progress in meeting the ADA’s physical accessibility requirements, said Dr. Iezzoni. “The literature suggests that doctors have done a good job at fixing the structural barriers people with mobility issues face, such as ramps and bathrooms.”
However, there are exceptions in rural older buildings which can be harder to retrofit for wheelchair accessibility, she said. “I recall interviewing a rural doctor several years ago who said that he knew his patients well and when a patient visits with mobility problems, he goes down and carries the patient up the steps to his office. My response was that is not respectful of the patient or safe for the patient or you. That doctor has since changed the location of his practice,” said Dr. Iezzoni.
Some doctors may resist paying for accessible medical equipment because of cost, but she said the benefits are worth it. These include preventing staff injuries when they transfer patients and being used by patients with temporary disabilities and aging people with bad knees, backs, hearing and sight. In addition, businesses may be eligible for federal and state tax credits.
Dr. Iezzoni recently visited her doctor where they finally got height-adjustable exam tables. “I asked the assistant, who really likes these tables? She said it’s the elderly ladies of short stature – the table is lowered and they sit down and get on it.”
But, Dr. Iezonni’s main message to doctors is that patients with disabilities deserve equal quality of care. “Just because we have a disability doesn’t mean we should get worse care than other people. It’s a matter of professionalism that doctors should want to give the same quality care to all their patients.”
A version of this article first appeared on Medscape.com.
Lisa Iezzoni, MD, a professor of medicine at Harvard Medical School and a disability researcher at Massachusetts General Hospital, both in Boston, has used a wheelchair for more than 30 years because of multiple sclerosis. When she visits her primary care doctor, she doesn’t get weighed because the scales are not wheelchair accessible.
This failure to weigh her and other patients in wheelchairs could lead to serious medical problems. Weight is used to monitor a person’s overall health and prenatal health and to determine accurate doses for medications such as some chemotherapies, said Dr. Iezzoni.
In another situation, a man who used a wheelchair said that his primary care doctor never got him out of it for a complete physical exam. The patient later developed lymphoma, which first appeared in his groin. The doctor should have accommodated his disability and used a height-adjustable exam table or a portable lift to transfer him onto the table.
When physicians don’t provide access to medical care that patients with disabilities need, they put themselves at greater risk of lawsuits, fines, and settlements.
Yet, a new study in Health Affairs suggests that a large percentage of doctors are not fully aware of what they are legally required to do.
Under federal nondiscrimination laws (Americans With Disabilities Act, American Rehabilitation Act, and ADA Amendments Act), medical practices must provide equal access to people with disabilities, accommodate their disability-related needs, and not refuse them medical services because of their disabilities, say disability experts.
Where doctors go wrong with disability laws
What doctors don’t know about providing reasonable accommodations makes them vulnerable to lawsuits, which worries more than two-thirds of the 714 outpatient doctors surveyed.
Not only are they required to provide reasonable accommodations, but they also have to pay for them, the researchers said. One-fifth of the surveyed doctors said they didn’t know that practice owners have to pay.
More than one practice has made patients pay for services needed for their disability, such as sign language interpreters – the patients later complained this violated the ADA to enforcement agencies.
Doctors also don’t know that they have to collaborate with patients to determine what reasonable accommodations they need – over two-thirds of those surveyed said they didn’t know it was a joint responsibility, the study found.
When doctors fail to accommodate patients’ disability needs, they engage in discrimination and violate the ADA, says Elizabeth Pendo, JD, a coauthor of the study and the Joseph J. Simeone Professor of Law at Saint Louis University.
The Department of Justice has investigated several patient complaints of alleged disability discrimination recently and resolved the disputes with agreements and small fines in some cases. “The goal is not to get large financial settlements but to work with practices to get the correct procedures in place to be compliant,” said Ms. Pendo.
Physicians would be wise to check out whether their practices are as accessible as they think. Even if there’s a ramp to the office building, the parking lot may not have a van-accessible space or enough handicapped parking signs, or the exam room may be too narrow for a wheelchair to navigate.
These practices violated the ADA and agreed to make changes:
- Hamden, Conn., has two buildings that patients with physical disabilities couldn’t easily enter. The physician owners agreed to change the buildings’ entrances and access routes and add features to make it easier to use examination rooms and restrooms and the check-in and check-out areas.
- Seven medical offices in Riverside, Calif., failed to communicate effectively with deaf and hard-of-hearing patients. They should have had a qualified sign language interpreter, an assistive listening device, or another appropriate aid or service available to a deaf patient and her family. Instead, the office relied on a video remote interpretation system that often failed to work. The agreement requires the clinic to provide those aids and services to patients and their companions who are deaf or hard of hearing, advertise their availability, assess each patient who is deaf or hard of hearing to determine the best aids and services for their needs, and pay $5,000 in compensation to the complainant and a $1,000 civil penalty to the United States.
- Springfield, Mass., refused to provide full joint replacements to two patients being treated with buprenorphine, a medication used to treat opioid use disorder. Rather than accommodate the patients, the surgeons referred them elsewhere because they were uncomfortable with the postoperative pain management protocol for patients prescribed buprenorphine. “The Americans With Disabilities Act protects health care access for people under medical treatment for opioid use disorder,” said Acting U.S. Attorney Nathaniel R. Mendell. “Health care providers must comply with the ADA, even when doing so is inconvenient or makes them uncomfortable.” The agreement requires the practice to adopt a nondiscrimination policy, provide training on the ADA and opioid use disorder, and pay two complainants $15,000 each for pain and suffering.
The DOJ has filed civil lawsuits against medical practices when they failed to resolve the allegations. Recent cases include an ophthalmology practice with 24 facilities in Arizona that refused to help transfer patients in wheelchairs to surgery tables for eye surgery and required them to pay for transfer support services and two obstetricians-gynecologists in Bakersfield, Calif., who refused to provide routine medical care to a patient because of her HIV status.
What doctors should know
Many people tend to think of a person with a disability as being in a wheelchair. But the ADA has a very broad definition of disability, which includes any physical or mental impairment that substantially limits any major life activity, said Ms. Pendo.
“It was amended in 2008 to clarify that the definition includes people with chronic diseases such as diabetes and cancer, cognitive and neurological disorders, substance abuse disorders, vision and hearing loss, and learning and other disabilities,” she said.
That means that doctors have to accommodate many types of disabilities, which can be challenging. The ADA only specifies that fixed structures need to be accessible, such as parking lots, driveways, and buildings, said Dr. Iezzoni.
When it comes to “reasonable accommodations,” doctors should decide that on a case-by-case basis, she said.
“We can say based on our study that 71% of doctors don’t know the right way to think about the accommodations – they don’t know they need to talk to patients so they can explain to them exactly what they need to accommodate their disability,” said Dr. Iezzoni.
Doctors are also required to provide effective communication for patients with sensory or cognitive disabilities, which can depend on the severity, said Ms. Pendo. Is the person deaf or hard of hearing, blind or partially sighted – is the dementia mild or severe?
“The requirement is there, but what that looks like will vary by patient. That’s what’s challenging,” said Ms. Pendo.
Dr. Iezzoni recommends that doctor’s offices ask patients whether they need special help or individual assistance when they make appointments and enter their responses in their records. She also suggests that patients be asked at follow-up appointments whether they still need the same help or not.
“Disabilities can change over time – a person with bad arthritis may need help getting onto an exam table, but later get a knee or hip replacement that is effective and no longer need that help,” said Dr. Iezzoni.
Benefits outweigh costs
Physicians have made progress in meeting the ADA’s physical accessibility requirements, said Dr. Iezzoni. “The literature suggests that doctors have done a good job at fixing the structural barriers people with mobility issues face, such as ramps and bathrooms.”
However, there are exceptions in rural older buildings which can be harder to retrofit for wheelchair accessibility, she said. “I recall interviewing a rural doctor several years ago who said that he knew his patients well and when a patient visits with mobility problems, he goes down and carries the patient up the steps to his office. My response was that is not respectful of the patient or safe for the patient or you. That doctor has since changed the location of his practice,” said Dr. Iezzoni.
Some doctors may resist paying for accessible medical equipment because of cost, but she said the benefits are worth it. These include preventing staff injuries when they transfer patients and being used by patients with temporary disabilities and aging people with bad knees, backs, hearing and sight. In addition, businesses may be eligible for federal and state tax credits.
Dr. Iezzoni recently visited her doctor where they finally got height-adjustable exam tables. “I asked the assistant, who really likes these tables? She said it’s the elderly ladies of short stature – the table is lowered and they sit down and get on it.”
But, Dr. Iezonni’s main message to doctors is that patients with disabilities deserve equal quality of care. “Just because we have a disability doesn’t mean we should get worse care than other people. It’s a matter of professionalism that doctors should want to give the same quality care to all their patients.”
A version of this article first appeared on Medscape.com.
CDC issues new pneumococcal vaccine recommendations for adults
The recommendations, voted on by the CDC’s Advisory Committee on Immunization Practices (ACIP) in October and made final in January with publication in the agency’s Morbidity and Mortality Weekly Report (MMWR), call for use of the 15-valent pneumococcal conjugate vaccine (PCV15; Vaxneuvance, Merck Sharp & Dohme) or 20-valent PCV (PREVNAR20; Wyeth Pharmaceuticals).
The recommendations apply to PCV-naive adults in the United States who are either aged 65 years or older, or who are aged 19-64 years and have underlying conditions such as diabetes, chronic heart or liver disease, or HIV, and have not previously received a PCV or whose previous vaccination history is unknown.
If the PCV15 vaccine is used, a subsequent dose of the 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23, Merck Sharp & Dohme) should be provided, typically at least 1 year later, under the recommendations.
As reported by this news organization, PCV15 and PREVNAR20 received approval from the Food and Drug Administration last July.
Those approvals provided an impetus for the revised recommendations, “offer[ing] an opportunity to review the existing recommendations and available data,” Miwako Kobayashi, MD, first author of the MMWR report and a medical epidemiologist with the National Center for Immunization and Respiratory Diseases, CDC, in Atlanta, said in an interview.
“As part of that process, ACIP strived to simplify the recommendations,” she said.
The previous recommendations called for the PCV13 vaccine and the PPSV23 and had varying conditions (depending on certain age and risk groups) that added complexity to the process. Under the new approach, the same recommendation applies regardless of specific medical conditions or other risk factors.
“With the simplified recommendation for adults 19 through 64, we expect coverage may increase among this population,” Dr. Kobayashi said.
Compared with the PCV13 vaccine, PREVNAR20 protects against seven additional serotypes involved in cases of invasive pneumococcal disease (IPD) and pneumonia, which are responsible for up to 40% of all cases of pneumococcal disease and related deaths in the United States.
While the PREVNAR20 includes five more pneumococcal serotypes than PCV15, the
CDC does not recommend one over the other, Dr. Kobayashi noted.
More than 90% of cases of adult IPD involve older adults and adults with chronic medical conditions or immunocompromising conditions, cerebrospinal fluid leaks, or cochlear implants, the MMWR report notes.
Commenting on the recommendations, Amit A. Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, Ariz., underscored the need for clinicians to be proactive in recommending the vaccines to those patients.
“Despite only needing one vaccine dose after turning 65 to be considered vaccinated, only about 70% of people in this group have received any pneumococcal vaccination,” he said in an interview. “This percentage has not increased much over the past several years.”
The new approach should help change that, he said.
“These new recommendations are a significant simplification from the prior confusing and challenging-to-implement recommendations from 2019,” Dr. Shah explained.
Among the 2019 recommendations was a stipulation for “shared decision-making” with PCV13, and a conversation that often only complicated matters, he noted.
“Patients and providers alike had confusion about this since it was not a clear-cut ‘yes, give it’ or ‘no, do not give it any longer’ recommendation.”
“Now that this new recommendation will require no extra time for a discussion in the clinic, and just a simple ‘it’s time for your pneumonia shot’ offer, this may become more feasible,” Dr. Shah added. “In addition, removal of the shared decision-making stipulation allows for this immunization to be easily protocolized in the clinic, similar to automatic offers to the flu vaccine for patients each year.”
According to the CDC, pneumococcal pneumonia causes an estimated 150,000 hospitalizations each year in the United States, while pneumococcal meningitis and bacteremia killed approximately 3,250 people in the United States in 2019.
“Clinicians are patients’ most trusted resource when it comes to vaccine recommendations,” Dr. Kobayashi said. “We encourage all clinicians to recommend pneumococcal vaccines when indicated.”
Dr. Kobayashi and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The recommendations, voted on by the CDC’s Advisory Committee on Immunization Practices (ACIP) in October and made final in January with publication in the agency’s Morbidity and Mortality Weekly Report (MMWR), call for use of the 15-valent pneumococcal conjugate vaccine (PCV15; Vaxneuvance, Merck Sharp & Dohme) or 20-valent PCV (PREVNAR20; Wyeth Pharmaceuticals).
The recommendations apply to PCV-naive adults in the United States who are either aged 65 years or older, or who are aged 19-64 years and have underlying conditions such as diabetes, chronic heart or liver disease, or HIV, and have not previously received a PCV or whose previous vaccination history is unknown.
If the PCV15 vaccine is used, a subsequent dose of the 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23, Merck Sharp & Dohme) should be provided, typically at least 1 year later, under the recommendations.
As reported by this news organization, PCV15 and PREVNAR20 received approval from the Food and Drug Administration last July.
Those approvals provided an impetus for the revised recommendations, “offer[ing] an opportunity to review the existing recommendations and available data,” Miwako Kobayashi, MD, first author of the MMWR report and a medical epidemiologist with the National Center for Immunization and Respiratory Diseases, CDC, in Atlanta, said in an interview.
“As part of that process, ACIP strived to simplify the recommendations,” she said.
The previous recommendations called for the PCV13 vaccine and the PPSV23 and had varying conditions (depending on certain age and risk groups) that added complexity to the process. Under the new approach, the same recommendation applies regardless of specific medical conditions or other risk factors.
“With the simplified recommendation for adults 19 through 64, we expect coverage may increase among this population,” Dr. Kobayashi said.
Compared with the PCV13 vaccine, PREVNAR20 protects against seven additional serotypes involved in cases of invasive pneumococcal disease (IPD) and pneumonia, which are responsible for up to 40% of all cases of pneumococcal disease and related deaths in the United States.
While the PREVNAR20 includes five more pneumococcal serotypes than PCV15, the
CDC does not recommend one over the other, Dr. Kobayashi noted.
More than 90% of cases of adult IPD involve older adults and adults with chronic medical conditions or immunocompromising conditions, cerebrospinal fluid leaks, or cochlear implants, the MMWR report notes.
Commenting on the recommendations, Amit A. Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, Ariz., underscored the need for clinicians to be proactive in recommending the vaccines to those patients.
“Despite only needing one vaccine dose after turning 65 to be considered vaccinated, only about 70% of people in this group have received any pneumococcal vaccination,” he said in an interview. “This percentage has not increased much over the past several years.”
The new approach should help change that, he said.
“These new recommendations are a significant simplification from the prior confusing and challenging-to-implement recommendations from 2019,” Dr. Shah explained.
Among the 2019 recommendations was a stipulation for “shared decision-making” with PCV13, and a conversation that often only complicated matters, he noted.
“Patients and providers alike had confusion about this since it was not a clear-cut ‘yes, give it’ or ‘no, do not give it any longer’ recommendation.”
“Now that this new recommendation will require no extra time for a discussion in the clinic, and just a simple ‘it’s time for your pneumonia shot’ offer, this may become more feasible,” Dr. Shah added. “In addition, removal of the shared decision-making stipulation allows for this immunization to be easily protocolized in the clinic, similar to automatic offers to the flu vaccine for patients each year.”
According to the CDC, pneumococcal pneumonia causes an estimated 150,000 hospitalizations each year in the United States, while pneumococcal meningitis and bacteremia killed approximately 3,250 people in the United States in 2019.
“Clinicians are patients’ most trusted resource when it comes to vaccine recommendations,” Dr. Kobayashi said. “We encourage all clinicians to recommend pneumococcal vaccines when indicated.”
Dr. Kobayashi and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The recommendations, voted on by the CDC’s Advisory Committee on Immunization Practices (ACIP) in October and made final in January with publication in the agency’s Morbidity and Mortality Weekly Report (MMWR), call for use of the 15-valent pneumococcal conjugate vaccine (PCV15; Vaxneuvance, Merck Sharp & Dohme) or 20-valent PCV (PREVNAR20; Wyeth Pharmaceuticals).
The recommendations apply to PCV-naive adults in the United States who are either aged 65 years or older, or who are aged 19-64 years and have underlying conditions such as diabetes, chronic heart or liver disease, or HIV, and have not previously received a PCV or whose previous vaccination history is unknown.
If the PCV15 vaccine is used, a subsequent dose of the 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23, Merck Sharp & Dohme) should be provided, typically at least 1 year later, under the recommendations.
As reported by this news organization, PCV15 and PREVNAR20 received approval from the Food and Drug Administration last July.
Those approvals provided an impetus for the revised recommendations, “offer[ing] an opportunity to review the existing recommendations and available data,” Miwako Kobayashi, MD, first author of the MMWR report and a medical epidemiologist with the National Center for Immunization and Respiratory Diseases, CDC, in Atlanta, said in an interview.
“As part of that process, ACIP strived to simplify the recommendations,” she said.
The previous recommendations called for the PCV13 vaccine and the PPSV23 and had varying conditions (depending on certain age and risk groups) that added complexity to the process. Under the new approach, the same recommendation applies regardless of specific medical conditions or other risk factors.
“With the simplified recommendation for adults 19 through 64, we expect coverage may increase among this population,” Dr. Kobayashi said.
Compared with the PCV13 vaccine, PREVNAR20 protects against seven additional serotypes involved in cases of invasive pneumococcal disease (IPD) and pneumonia, which are responsible for up to 40% of all cases of pneumococcal disease and related deaths in the United States.
While the PREVNAR20 includes five more pneumococcal serotypes than PCV15, the
CDC does not recommend one over the other, Dr. Kobayashi noted.
More than 90% of cases of adult IPD involve older adults and adults with chronic medical conditions or immunocompromising conditions, cerebrospinal fluid leaks, or cochlear implants, the MMWR report notes.
Commenting on the recommendations, Amit A. Shah, MD, a geriatrician with the Mayo Clinic in Phoenix, Ariz., underscored the need for clinicians to be proactive in recommending the vaccines to those patients.
“Despite only needing one vaccine dose after turning 65 to be considered vaccinated, only about 70% of people in this group have received any pneumococcal vaccination,” he said in an interview. “This percentage has not increased much over the past several years.”
The new approach should help change that, he said.
“These new recommendations are a significant simplification from the prior confusing and challenging-to-implement recommendations from 2019,” Dr. Shah explained.
Among the 2019 recommendations was a stipulation for “shared decision-making” with PCV13, and a conversation that often only complicated matters, he noted.
“Patients and providers alike had confusion about this since it was not a clear-cut ‘yes, give it’ or ‘no, do not give it any longer’ recommendation.”
“Now that this new recommendation will require no extra time for a discussion in the clinic, and just a simple ‘it’s time for your pneumonia shot’ offer, this may become more feasible,” Dr. Shah added. “In addition, removal of the shared decision-making stipulation allows for this immunization to be easily protocolized in the clinic, similar to automatic offers to the flu vaccine for patients each year.”
According to the CDC, pneumococcal pneumonia causes an estimated 150,000 hospitalizations each year in the United States, while pneumococcal meningitis and bacteremia killed approximately 3,250 people in the United States in 2019.
“Clinicians are patients’ most trusted resource when it comes to vaccine recommendations,” Dr. Kobayashi said. “We encourage all clinicians to recommend pneumococcal vaccines when indicated.”
Dr. Kobayashi and Dr. Shah have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Newly approved drug improves sleep onset in insomnia
In the first of two studies, a 50-mg dose of daridorexant was associated with a reduction in latency to persistent sleep (LPS) of 11.7 minutes at month 3 versus placebo. The drug also was associated with improved daytime function.
Based on these results, the Food and Drug Administration approved daridorexant for the treatment of insomnia in adults earlier in January.
“The study shows that it is a really good drug that works differently from most other drugs,” said Emmanuel Mignot, MD, PhD, professor of sleep medicine at Stanford (Calif.) University. “It’s more specific to sleep,” Dr. Mignot added.
The findings were published in the February issue of The Lancet Neurology.
Two trials, three doses
Daridorexant is a dual orexin receptor antagonist intended to reduce excessive wakefulness. The investigators hypothesized that, because of its therapeutic target, the drug would not cause sleepiness on the morning after administration.
To examine daridorexant’s safety and efficacy, the researchers conducted two double-blind, parallel-group, phase 3 trials. Eligible participants were aged 18 years or older, had moderate to severe insomnia disorder, and had a self-reported history of disturbed sleep at least 3 nights per week for at least 3 months before screening.
In study 1, investigators randomly assigned participants in groups of equal size to daridorexant 25 mg, 50 mg, or placebo. In study 2, participants were randomly assigned to daridorexant 10 mg, 25 mg, or placebo.
During a placebo run-in period, participants underwent polysomnography on two consecutive nights to define baseline values. At the end of months 1 and 3 of the treatment period, participants again underwent 2 nights of polysomnography. A final night of polysomnography occurred during the placebo run-out period.
Self-assessments included the Insomnia Daytime Symptoms and Impacts Questionnaire (IDSIQ). This questionnaire, to which participants responded daily, is designed to measure the daytime impairments related to insomnia. The IDSIQ questions focus on sleepiness, mood, alertness, and cognition.
The study’s primary endpoints were change from baseline in wake after sleep onset (WASO) and LPS at months 1 and 3. Secondary endpoints were change from baseline in self-reported total sleep time and change in the IDSIQ sleepiness domain score at months 1 and 3.
The investigators enrolled 930 participants in study 1 and 924 in study 2. In each study, more than two-thirds of participants were women, 39% were aged 65 or older, and demographic and baseline characteristics were similar between treatment groups.
Dose-dependent effects
At month 1 in study 1, WASO was reduced by 22.8 minutes (P < .0001) in patients who received the 50-mg dose and by 12.2 minutes (P < .0001) in the 25-mg dose. At month 3, WASO was reduced by 18.3 minutes (P < .0001) in those assigned to 50 mg and by 11.9 minutes (P < .0001) in those assigned to 25 mg.
LPS was reduced by 11.4 minutes (P < .0001) at month 1 and by 11.7 minutes (P < .0001) at month 3 with the 50-mg dose versus placebo. LPS was reduced by 8.3 minutes (P = .0005) at month 1 and by 7.6 minutes (P = .0015) at month 3 with the 25-mg dose versus placebo.
At both time points, self-reported total sleep time was significantly increased and the IDSIQ sleepiness score significantly improved with the 50-mg dose. The 25-mg dose was associated with significant improvements in self-reported total sleep time at both time points, but not with significant improvements in IDSIQ sleepiness score.
In study 2, the 25-mg dose was associated with significant reductions in WASO at month 1 (11.6 minutes, P = .0001) and month 3 (10.3 minutes, P = .0028) compared with placebo. The 25-mg dose was not associated with significant differences in LPS at either time point, however.
Similarly, the 25-mg dose was associated with improvements in self-reported total sleep time, but not with the IDSIQ sleepiness score. The 10-mg dose was not associated with improvements on any endpoint compared with placebo.
Longer studies needed
In an accompanying editorial, Kai Spiegelhalder, PhD, University of Freiburg, Germany, and colleagues pointed out that although insomnia disorder is defined by self-reported difficulty initiating or maintaining sleep, none of the primary or secondary endpoints in these trials addressed these symptoms.
However, Dr. Mignot noted the use of the IDSIQ scale is the most interesting aspect of the study. Although difficulty with concentration and mood impairment are major symptoms of insomnia, they are often neglected. “This drug was reversing the daytime impairment that insomniacs have,” said Dr. Mignot. “We now need to systematically study people not only for the effect on sleep, but also that it makes them feel better the day after.”
He added that most of the current hypnotics were not developed to treat insomnia. Daridorexant, in contrast, targets the wake-promoting orexin system. “It works more selectively on sleep and not on other things. Most of the other sleeping pills have more complex effects on the brain,” Dr. Mignot said.
Commenting on the study, John Winkelman, MD, PhD, professor of psychiatry at Harvard Medical School, Boston, said the low prevalence of side effects associated with daridorexant was remarkable. “This is not what most of the benzodiazepine receptor agonists looked like,” said Dr. Winkelman, who was not involved with the research.
Many insomnia drugs affect transmitter systems that are widespread in the brain, thus provoking side effects. But orexin-receptor antagonists “don’t seem to produce a lot of side effects,” he noted.
Although the study duration was reasonable, longer studies will be necessary, he added. “And it would be nice to see comparative data. Prescribers want to see some context.” said Dr. Winkelman.
The study was funded by Idorsia Pharmaceuticals. Dr. Mignot reported receiving research or clinical trial funding from Axsome, Jazz Pharmaceuticals, Avadel, Apple, Huami, Sunovion, and Takeda. He has also received consulting fees or speakers’ conference reimbursement from Idorsia, Centessa Pharmaceuticals, Jazz Pharmaceuticals, Avadel, Dreem, and Takeda. Dr. Winkelman has consulted for Idorsia and has participated in investigator-initiated studies supported by Merck.
A version of this article first appeared on Medscape.com.
In the first of two studies, a 50-mg dose of daridorexant was associated with a reduction in latency to persistent sleep (LPS) of 11.7 minutes at month 3 versus placebo. The drug also was associated with improved daytime function.
Based on these results, the Food and Drug Administration approved daridorexant for the treatment of insomnia in adults earlier in January.
“The study shows that it is a really good drug that works differently from most other drugs,” said Emmanuel Mignot, MD, PhD, professor of sleep medicine at Stanford (Calif.) University. “It’s more specific to sleep,” Dr. Mignot added.
The findings were published in the February issue of The Lancet Neurology.
Two trials, three doses
Daridorexant is a dual orexin receptor antagonist intended to reduce excessive wakefulness. The investigators hypothesized that, because of its therapeutic target, the drug would not cause sleepiness on the morning after administration.
To examine daridorexant’s safety and efficacy, the researchers conducted two double-blind, parallel-group, phase 3 trials. Eligible participants were aged 18 years or older, had moderate to severe insomnia disorder, and had a self-reported history of disturbed sleep at least 3 nights per week for at least 3 months before screening.
In study 1, investigators randomly assigned participants in groups of equal size to daridorexant 25 mg, 50 mg, or placebo. In study 2, participants were randomly assigned to daridorexant 10 mg, 25 mg, or placebo.
During a placebo run-in period, participants underwent polysomnography on two consecutive nights to define baseline values. At the end of months 1 and 3 of the treatment period, participants again underwent 2 nights of polysomnography. A final night of polysomnography occurred during the placebo run-out period.
Self-assessments included the Insomnia Daytime Symptoms and Impacts Questionnaire (IDSIQ). This questionnaire, to which participants responded daily, is designed to measure the daytime impairments related to insomnia. The IDSIQ questions focus on sleepiness, mood, alertness, and cognition.
The study’s primary endpoints were change from baseline in wake after sleep onset (WASO) and LPS at months 1 and 3. Secondary endpoints were change from baseline in self-reported total sleep time and change in the IDSIQ sleepiness domain score at months 1 and 3.
The investigators enrolled 930 participants in study 1 and 924 in study 2. In each study, more than two-thirds of participants were women, 39% were aged 65 or older, and demographic and baseline characteristics were similar between treatment groups.
Dose-dependent effects
At month 1 in study 1, WASO was reduced by 22.8 minutes (P < .0001) in patients who received the 50-mg dose and by 12.2 minutes (P < .0001) in the 25-mg dose. At month 3, WASO was reduced by 18.3 minutes (P < .0001) in those assigned to 50 mg and by 11.9 minutes (P < .0001) in those assigned to 25 mg.
LPS was reduced by 11.4 minutes (P < .0001) at month 1 and by 11.7 minutes (P < .0001) at month 3 with the 50-mg dose versus placebo. LPS was reduced by 8.3 minutes (P = .0005) at month 1 and by 7.6 minutes (P = .0015) at month 3 with the 25-mg dose versus placebo.
At both time points, self-reported total sleep time was significantly increased and the IDSIQ sleepiness score significantly improved with the 50-mg dose. The 25-mg dose was associated with significant improvements in self-reported total sleep time at both time points, but not with significant improvements in IDSIQ sleepiness score.
In study 2, the 25-mg dose was associated with significant reductions in WASO at month 1 (11.6 minutes, P = .0001) and month 3 (10.3 minutes, P = .0028) compared with placebo. The 25-mg dose was not associated with significant differences in LPS at either time point, however.
Similarly, the 25-mg dose was associated with improvements in self-reported total sleep time, but not with the IDSIQ sleepiness score. The 10-mg dose was not associated with improvements on any endpoint compared with placebo.
Longer studies needed
In an accompanying editorial, Kai Spiegelhalder, PhD, University of Freiburg, Germany, and colleagues pointed out that although insomnia disorder is defined by self-reported difficulty initiating or maintaining sleep, none of the primary or secondary endpoints in these trials addressed these symptoms.
However, Dr. Mignot noted the use of the IDSIQ scale is the most interesting aspect of the study. Although difficulty with concentration and mood impairment are major symptoms of insomnia, they are often neglected. “This drug was reversing the daytime impairment that insomniacs have,” said Dr. Mignot. “We now need to systematically study people not only for the effect on sleep, but also that it makes them feel better the day after.”
He added that most of the current hypnotics were not developed to treat insomnia. Daridorexant, in contrast, targets the wake-promoting orexin system. “It works more selectively on sleep and not on other things. Most of the other sleeping pills have more complex effects on the brain,” Dr. Mignot said.
Commenting on the study, John Winkelman, MD, PhD, professor of psychiatry at Harvard Medical School, Boston, said the low prevalence of side effects associated with daridorexant was remarkable. “This is not what most of the benzodiazepine receptor agonists looked like,” said Dr. Winkelman, who was not involved with the research.
Many insomnia drugs affect transmitter systems that are widespread in the brain, thus provoking side effects. But orexin-receptor antagonists “don’t seem to produce a lot of side effects,” he noted.
Although the study duration was reasonable, longer studies will be necessary, he added. “And it would be nice to see comparative data. Prescribers want to see some context.” said Dr. Winkelman.
The study was funded by Idorsia Pharmaceuticals. Dr. Mignot reported receiving research or clinical trial funding from Axsome, Jazz Pharmaceuticals, Avadel, Apple, Huami, Sunovion, and Takeda. He has also received consulting fees or speakers’ conference reimbursement from Idorsia, Centessa Pharmaceuticals, Jazz Pharmaceuticals, Avadel, Dreem, and Takeda. Dr. Winkelman has consulted for Idorsia and has participated in investigator-initiated studies supported by Merck.
A version of this article first appeared on Medscape.com.
In the first of two studies, a 50-mg dose of daridorexant was associated with a reduction in latency to persistent sleep (LPS) of 11.7 minutes at month 3 versus placebo. The drug also was associated with improved daytime function.
Based on these results, the Food and Drug Administration approved daridorexant for the treatment of insomnia in adults earlier in January.
“The study shows that it is a really good drug that works differently from most other drugs,” said Emmanuel Mignot, MD, PhD, professor of sleep medicine at Stanford (Calif.) University. “It’s more specific to sleep,” Dr. Mignot added.
The findings were published in the February issue of The Lancet Neurology.
Two trials, three doses
Daridorexant is a dual orexin receptor antagonist intended to reduce excessive wakefulness. The investigators hypothesized that, because of its therapeutic target, the drug would not cause sleepiness on the morning after administration.
To examine daridorexant’s safety and efficacy, the researchers conducted two double-blind, parallel-group, phase 3 trials. Eligible participants were aged 18 years or older, had moderate to severe insomnia disorder, and had a self-reported history of disturbed sleep at least 3 nights per week for at least 3 months before screening.
In study 1, investigators randomly assigned participants in groups of equal size to daridorexant 25 mg, 50 mg, or placebo. In study 2, participants were randomly assigned to daridorexant 10 mg, 25 mg, or placebo.
During a placebo run-in period, participants underwent polysomnography on two consecutive nights to define baseline values. At the end of months 1 and 3 of the treatment period, participants again underwent 2 nights of polysomnography. A final night of polysomnography occurred during the placebo run-out period.
Self-assessments included the Insomnia Daytime Symptoms and Impacts Questionnaire (IDSIQ). This questionnaire, to which participants responded daily, is designed to measure the daytime impairments related to insomnia. The IDSIQ questions focus on sleepiness, mood, alertness, and cognition.
The study’s primary endpoints were change from baseline in wake after sleep onset (WASO) and LPS at months 1 and 3. Secondary endpoints were change from baseline in self-reported total sleep time and change in the IDSIQ sleepiness domain score at months 1 and 3.
The investigators enrolled 930 participants in study 1 and 924 in study 2. In each study, more than two-thirds of participants were women, 39% were aged 65 or older, and demographic and baseline characteristics were similar between treatment groups.
Dose-dependent effects
At month 1 in study 1, WASO was reduced by 22.8 minutes (P < .0001) in patients who received the 50-mg dose and by 12.2 minutes (P < .0001) in the 25-mg dose. At month 3, WASO was reduced by 18.3 minutes (P < .0001) in those assigned to 50 mg and by 11.9 minutes (P < .0001) in those assigned to 25 mg.
LPS was reduced by 11.4 minutes (P < .0001) at month 1 and by 11.7 minutes (P < .0001) at month 3 with the 50-mg dose versus placebo. LPS was reduced by 8.3 minutes (P = .0005) at month 1 and by 7.6 minutes (P = .0015) at month 3 with the 25-mg dose versus placebo.
At both time points, self-reported total sleep time was significantly increased and the IDSIQ sleepiness score significantly improved with the 50-mg dose. The 25-mg dose was associated with significant improvements in self-reported total sleep time at both time points, but not with significant improvements in IDSIQ sleepiness score.
In study 2, the 25-mg dose was associated with significant reductions in WASO at month 1 (11.6 minutes, P = .0001) and month 3 (10.3 minutes, P = .0028) compared with placebo. The 25-mg dose was not associated with significant differences in LPS at either time point, however.
Similarly, the 25-mg dose was associated with improvements in self-reported total sleep time, but not with the IDSIQ sleepiness score. The 10-mg dose was not associated with improvements on any endpoint compared with placebo.
Longer studies needed
In an accompanying editorial, Kai Spiegelhalder, PhD, University of Freiburg, Germany, and colleagues pointed out that although insomnia disorder is defined by self-reported difficulty initiating or maintaining sleep, none of the primary or secondary endpoints in these trials addressed these symptoms.
However, Dr. Mignot noted the use of the IDSIQ scale is the most interesting aspect of the study. Although difficulty with concentration and mood impairment are major symptoms of insomnia, they are often neglected. “This drug was reversing the daytime impairment that insomniacs have,” said Dr. Mignot. “We now need to systematically study people not only for the effect on sleep, but also that it makes them feel better the day after.”
He added that most of the current hypnotics were not developed to treat insomnia. Daridorexant, in contrast, targets the wake-promoting orexin system. “It works more selectively on sleep and not on other things. Most of the other sleeping pills have more complex effects on the brain,” Dr. Mignot said.
Commenting on the study, John Winkelman, MD, PhD, professor of psychiatry at Harvard Medical School, Boston, said the low prevalence of side effects associated with daridorexant was remarkable. “This is not what most of the benzodiazepine receptor agonists looked like,” said Dr. Winkelman, who was not involved with the research.
Many insomnia drugs affect transmitter systems that are widespread in the brain, thus provoking side effects. But orexin-receptor antagonists “don’t seem to produce a lot of side effects,” he noted.
Although the study duration was reasonable, longer studies will be necessary, he added. “And it would be nice to see comparative data. Prescribers want to see some context.” said Dr. Winkelman.
The study was funded by Idorsia Pharmaceuticals. Dr. Mignot reported receiving research or clinical trial funding from Axsome, Jazz Pharmaceuticals, Avadel, Apple, Huami, Sunovion, and Takeda. He has also received consulting fees or speakers’ conference reimbursement from Idorsia, Centessa Pharmaceuticals, Jazz Pharmaceuticals, Avadel, Dreem, and Takeda. Dr. Winkelman has consulted for Idorsia and has participated in investigator-initiated studies supported by Merck.
A version of this article first appeared on Medscape.com.
FROM THE LANCET NEUROLOGY
Pandemic pushed death rates to historic highs
Excess mortality is a way of quantifying the impact of a pandemic, based on overall mortality from nonpandemic periods. Mortality data over long periods of time are not available for many countries, but Switzerland, Sweden, and Spain have accumulated death count data for an uninterrupted period of more than 100 years.
In a study published in the Annals of Internal Medicine, Kaspar Staub, PhD, of the University of Zurich led a team of researchers in reviewing data on monthly excess deaths from all causes for Switzerland, Sweden, and Spain for 2020 to 2021. Dr. Staub and colleagues also compared these numbers to other pandemic and nonpandemic periods since the end of the 19th century. The starting years were 1877 for Switzerland, 1851 for Sweden, and 1908 for Spain.
The researchers collected data for monthly all-cause deaths from the statistical offices of each country and determined excess mortality by comparing these numbers to population size and age structure.
They found that 2020 showed the highest number of excess deaths since 1918, with relative excess of deaths of 12.5% in Switzerland, 8.5% in Sweden, and 17.3 % in Spain.
To put it another way, the number of excess deaths per 100,000 people was 100 for Switzerland, 75 for Sweden, and 155 for Spain.
“Our findings suggest that the pandemic led to the second-largest mortality disaster driven by a viral infection in more than 100 years in the three countries we studied, second only to the 1918 influenza pandemic,” the researchers wrote.
They explained that the excess mortality for the year 1918 was six to seven times higher than the 2020 numbers, but that the 2020 numbers might have been higher without the strong public health interventions taken worldwide to mitigate the impact of the COVID-19 pandemic.
“Early estimates suggest that vaccination prevented approximately 470,000 deaths in persons aged 60 years or older across 33 European countries between December 2019 and November 2021,” they wrote. However, because the COVID-19 pandemic is ongoing, “a more conclusive assessment will have to wait,” they added.
The 2020 numbers also were higher than most mortality rates since 1918, including peak years of previous influenza pandemics that occurred in 1957, 1968, 1977, and, most recently, the swine flu pandemic of 2009 which was caused by a novel strain of the H1N1 influenza virus.
The study findings had some limitations. For example, only three countries were included. Also, monthly death numbers according to sex, age, and cause of death were available only for the past 60 years, and data from years before the 20th century may not be reliable, the researchers said.
The new study does not account for the long-term effects of patients suffering from long COVID, they noted.
Study findings support strong public health response
“With the COVID-19 pandemic ongoing, this study reinforces the historic magnitude of the problem in terms of mortality and could add to the justification for ongoing public health measures such as vaccination drives and vaccine mandates to curb deaths,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
“The results are surprising because when we view the rapid advancement in medical science over the last few decades, which have led to a decline in mortality from many previously fatal diseases, the scale of excess mortality from COVID-19 seems to have offset many such gains in the past 2 years.”
Prior studies of United States mortality data have estimated that excess deaths in the United States in 2020 exceeded the deaths attributed to COVID-19, said Dr. Pal. “The findings of this study could help clinicians in their discussion of the need for COVID-19 prevention measures with their patients” and inform discussions between doctors and patients about prevention strategies, he explained.
“Emphasizing that this pandemic is the second-largest cause of death due to a viral infection in a century could help patients understand the need for public health measures that may be viewed as unprecedented, such as government-imposed lockdowns, contact tracing, mask requirements, restrictions on travel, and vaccine mandates,” Dr. Pal noted. Better understanding of the evidence behind such measures may decrease the public’s resistance to following them, he added.As for additional research, “region-specific analysis of excess deaths may help estimate the impact of COVID-19 better, especially in regions where data reporting may be unreliable.”
Dr. F. Perry Wilson's take on study
“All-cause mortality is a key metric to assess the impact of the pandemic, because each death is treated equally,” said F. Perry Wilson, MD, of Yale University, in an interview. “With this type of analysis, there is no vague definition of a death from COVID or with COVID,” he explained. “A death is a death, and more deaths than expected is, of course, a bad thing. These analyses give a high-level view of the true human cost of the pandemic,” he said.
Dr. Wilson said he was not surprised by the findings. “There have been multiple studies, across multiple countries including the United States, which show similar findings—that observed deaths during this pandemic are substantially higher than expected,” he said. The current study findings are unique in that they compare the current pandemic to death rates in a nearly unbroken chain into the last century using data that only a few countries can provide, he noted.
The mortality data are “quite similar to what we see in the United States, with the exception that Spain was particularly hard-hit in the first COVID-19 wave in April 2020, said Dr. Wilson. By contrast, “the U.S. had substantially more excess deaths in the recent Delta wave, presumably due to lower vaccination uptake,” he added.
The current study is important for clinicians and their patients, said Dr. Wilson. “Data like these can help cut through some of the misinformation, such as the idea that only people who would have died anyway die of COVID, or that COVID is not severe,” he emphasized. “Overall death data are quite clear that far more people, millions more people, died over the last 22 months than could possibly be explained except by a global-level mortality event,” he said.
“One thing this study reminds us of is the value of high-quality data,” said Dr. Wilson. “Few countries have near complete vital statistics records on their entire populations and these can be so crucial to understand the true impact of pandemics and other disasters,” he explained. Of course, mortality data also serve as a reminder “that COVID is a serious disease: a once-in-a-century (we hope) pandemic,” he added.
The current study showed that excess death rates were similar, but not the same, from country to country, Dr. Wilson noted. “Moving forward, we need to learn what factors, from vaccination to social distancing strategies,” saved lives around the world,” he said.
The study was supported by the Foundation for Research in Science and the Humanities at the University of Zurich, the Swiss National Science Foundation, and the U.S. National Institute of Allergy and Infectious Diseases. The researchers, Dr. Pal, and Dr. Wilson had no financial conflicts.
*This article was updated on 2/1/2022.
Excess mortality is a way of quantifying the impact of a pandemic, based on overall mortality from nonpandemic periods. Mortality data over long periods of time are not available for many countries, but Switzerland, Sweden, and Spain have accumulated death count data for an uninterrupted period of more than 100 years.
In a study published in the Annals of Internal Medicine, Kaspar Staub, PhD, of the University of Zurich led a team of researchers in reviewing data on monthly excess deaths from all causes for Switzerland, Sweden, and Spain for 2020 to 2021. Dr. Staub and colleagues also compared these numbers to other pandemic and nonpandemic periods since the end of the 19th century. The starting years were 1877 for Switzerland, 1851 for Sweden, and 1908 for Spain.
The researchers collected data for monthly all-cause deaths from the statistical offices of each country and determined excess mortality by comparing these numbers to population size and age structure.
They found that 2020 showed the highest number of excess deaths since 1918, with relative excess of deaths of 12.5% in Switzerland, 8.5% in Sweden, and 17.3 % in Spain.
To put it another way, the number of excess deaths per 100,000 people was 100 for Switzerland, 75 for Sweden, and 155 for Spain.
“Our findings suggest that the pandemic led to the second-largest mortality disaster driven by a viral infection in more than 100 years in the three countries we studied, second only to the 1918 influenza pandemic,” the researchers wrote.
They explained that the excess mortality for the year 1918 was six to seven times higher than the 2020 numbers, but that the 2020 numbers might have been higher without the strong public health interventions taken worldwide to mitigate the impact of the COVID-19 pandemic.
“Early estimates suggest that vaccination prevented approximately 470,000 deaths in persons aged 60 years or older across 33 European countries between December 2019 and November 2021,” they wrote. However, because the COVID-19 pandemic is ongoing, “a more conclusive assessment will have to wait,” they added.
The 2020 numbers also were higher than most mortality rates since 1918, including peak years of previous influenza pandemics that occurred in 1957, 1968, 1977, and, most recently, the swine flu pandemic of 2009 which was caused by a novel strain of the H1N1 influenza virus.
The study findings had some limitations. For example, only three countries were included. Also, monthly death numbers according to sex, age, and cause of death were available only for the past 60 years, and data from years before the 20th century may not be reliable, the researchers said.
The new study does not account for the long-term effects of patients suffering from long COVID, they noted.
Study findings support strong public health response
“With the COVID-19 pandemic ongoing, this study reinforces the historic magnitude of the problem in terms of mortality and could add to the justification for ongoing public health measures such as vaccination drives and vaccine mandates to curb deaths,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
“The results are surprising because when we view the rapid advancement in medical science over the last few decades, which have led to a decline in mortality from many previously fatal diseases, the scale of excess mortality from COVID-19 seems to have offset many such gains in the past 2 years.”
Prior studies of United States mortality data have estimated that excess deaths in the United States in 2020 exceeded the deaths attributed to COVID-19, said Dr. Pal. “The findings of this study could help clinicians in their discussion of the need for COVID-19 prevention measures with their patients” and inform discussions between doctors and patients about prevention strategies, he explained.
“Emphasizing that this pandemic is the second-largest cause of death due to a viral infection in a century could help patients understand the need for public health measures that may be viewed as unprecedented, such as government-imposed lockdowns, contact tracing, mask requirements, restrictions on travel, and vaccine mandates,” Dr. Pal noted. Better understanding of the evidence behind such measures may decrease the public’s resistance to following them, he added.As for additional research, “region-specific analysis of excess deaths may help estimate the impact of COVID-19 better, especially in regions where data reporting may be unreliable.”
Dr. F. Perry Wilson's take on study
“All-cause mortality is a key metric to assess the impact of the pandemic, because each death is treated equally,” said F. Perry Wilson, MD, of Yale University, in an interview. “With this type of analysis, there is no vague definition of a death from COVID or with COVID,” he explained. “A death is a death, and more deaths than expected is, of course, a bad thing. These analyses give a high-level view of the true human cost of the pandemic,” he said.
Dr. Wilson said he was not surprised by the findings. “There have been multiple studies, across multiple countries including the United States, which show similar findings—that observed deaths during this pandemic are substantially higher than expected,” he said. The current study findings are unique in that they compare the current pandemic to death rates in a nearly unbroken chain into the last century using data that only a few countries can provide, he noted.
The mortality data are “quite similar to what we see in the United States, with the exception that Spain was particularly hard-hit in the first COVID-19 wave in April 2020, said Dr. Wilson. By contrast, “the U.S. had substantially more excess deaths in the recent Delta wave, presumably due to lower vaccination uptake,” he added.
The current study is important for clinicians and their patients, said Dr. Wilson. “Data like these can help cut through some of the misinformation, such as the idea that only people who would have died anyway die of COVID, or that COVID is not severe,” he emphasized. “Overall death data are quite clear that far more people, millions more people, died over the last 22 months than could possibly be explained except by a global-level mortality event,” he said.
“One thing this study reminds us of is the value of high-quality data,” said Dr. Wilson. “Few countries have near complete vital statistics records on their entire populations and these can be so crucial to understand the true impact of pandemics and other disasters,” he explained. Of course, mortality data also serve as a reminder “that COVID is a serious disease: a once-in-a-century (we hope) pandemic,” he added.
The current study showed that excess death rates were similar, but not the same, from country to country, Dr. Wilson noted. “Moving forward, we need to learn what factors, from vaccination to social distancing strategies,” saved lives around the world,” he said.
The study was supported by the Foundation for Research in Science and the Humanities at the University of Zurich, the Swiss National Science Foundation, and the U.S. National Institute of Allergy and Infectious Diseases. The researchers, Dr. Pal, and Dr. Wilson had no financial conflicts.
*This article was updated on 2/1/2022.
Excess mortality is a way of quantifying the impact of a pandemic, based on overall mortality from nonpandemic periods. Mortality data over long periods of time are not available for many countries, but Switzerland, Sweden, and Spain have accumulated death count data for an uninterrupted period of more than 100 years.
In a study published in the Annals of Internal Medicine, Kaspar Staub, PhD, of the University of Zurich led a team of researchers in reviewing data on monthly excess deaths from all causes for Switzerland, Sweden, and Spain for 2020 to 2021. Dr. Staub and colleagues also compared these numbers to other pandemic and nonpandemic periods since the end of the 19th century. The starting years were 1877 for Switzerland, 1851 for Sweden, and 1908 for Spain.
The researchers collected data for monthly all-cause deaths from the statistical offices of each country and determined excess mortality by comparing these numbers to population size and age structure.
They found that 2020 showed the highest number of excess deaths since 1918, with relative excess of deaths of 12.5% in Switzerland, 8.5% in Sweden, and 17.3 % in Spain.
To put it another way, the number of excess deaths per 100,000 people was 100 for Switzerland, 75 for Sweden, and 155 for Spain.
“Our findings suggest that the pandemic led to the second-largest mortality disaster driven by a viral infection in more than 100 years in the three countries we studied, second only to the 1918 influenza pandemic,” the researchers wrote.
They explained that the excess mortality for the year 1918 was six to seven times higher than the 2020 numbers, but that the 2020 numbers might have been higher without the strong public health interventions taken worldwide to mitigate the impact of the COVID-19 pandemic.
“Early estimates suggest that vaccination prevented approximately 470,000 deaths in persons aged 60 years or older across 33 European countries between December 2019 and November 2021,” they wrote. However, because the COVID-19 pandemic is ongoing, “a more conclusive assessment will have to wait,” they added.
The 2020 numbers also were higher than most mortality rates since 1918, including peak years of previous influenza pandemics that occurred in 1957, 1968, 1977, and, most recently, the swine flu pandemic of 2009 which was caused by a novel strain of the H1N1 influenza virus.
The study findings had some limitations. For example, only three countries were included. Also, monthly death numbers according to sex, age, and cause of death were available only for the past 60 years, and data from years before the 20th century may not be reliable, the researchers said.
The new study does not account for the long-term effects of patients suffering from long COVID, they noted.
Study findings support strong public health response
“With the COVID-19 pandemic ongoing, this study reinforces the historic magnitude of the problem in terms of mortality and could add to the justification for ongoing public health measures such as vaccination drives and vaccine mandates to curb deaths,” said Suman Pal, MD, an internal medicine physician at the University of New Mexico, Albuquerque, in an interview.
“The results are surprising because when we view the rapid advancement in medical science over the last few decades, which have led to a decline in mortality from many previously fatal diseases, the scale of excess mortality from COVID-19 seems to have offset many such gains in the past 2 years.”
Prior studies of United States mortality data have estimated that excess deaths in the United States in 2020 exceeded the deaths attributed to COVID-19, said Dr. Pal. “The findings of this study could help clinicians in their discussion of the need for COVID-19 prevention measures with their patients” and inform discussions between doctors and patients about prevention strategies, he explained.
“Emphasizing that this pandemic is the second-largest cause of death due to a viral infection in a century could help patients understand the need for public health measures that may be viewed as unprecedented, such as government-imposed lockdowns, contact tracing, mask requirements, restrictions on travel, and vaccine mandates,” Dr. Pal noted. Better understanding of the evidence behind such measures may decrease the public’s resistance to following them, he added.As for additional research, “region-specific analysis of excess deaths may help estimate the impact of COVID-19 better, especially in regions where data reporting may be unreliable.”
Dr. F. Perry Wilson's take on study
“All-cause mortality is a key metric to assess the impact of the pandemic, because each death is treated equally,” said F. Perry Wilson, MD, of Yale University, in an interview. “With this type of analysis, there is no vague definition of a death from COVID or with COVID,” he explained. “A death is a death, and more deaths than expected is, of course, a bad thing. These analyses give a high-level view of the true human cost of the pandemic,” he said.
Dr. Wilson said he was not surprised by the findings. “There have been multiple studies, across multiple countries including the United States, which show similar findings—that observed deaths during this pandemic are substantially higher than expected,” he said. The current study findings are unique in that they compare the current pandemic to death rates in a nearly unbroken chain into the last century using data that only a few countries can provide, he noted.
The mortality data are “quite similar to what we see in the United States, with the exception that Spain was particularly hard-hit in the first COVID-19 wave in April 2020, said Dr. Wilson. By contrast, “the U.S. had substantially more excess deaths in the recent Delta wave, presumably due to lower vaccination uptake,” he added.
The current study is important for clinicians and their patients, said Dr. Wilson. “Data like these can help cut through some of the misinformation, such as the idea that only people who would have died anyway die of COVID, or that COVID is not severe,” he emphasized. “Overall death data are quite clear that far more people, millions more people, died over the last 22 months than could possibly be explained except by a global-level mortality event,” he said.
“One thing this study reminds us of is the value of high-quality data,” said Dr. Wilson. “Few countries have near complete vital statistics records on their entire populations and these can be so crucial to understand the true impact of pandemics and other disasters,” he explained. Of course, mortality data also serve as a reminder “that COVID is a serious disease: a once-in-a-century (we hope) pandemic,” he added.
The current study showed that excess death rates were similar, but not the same, from country to country, Dr. Wilson noted. “Moving forward, we need to learn what factors, from vaccination to social distancing strategies,” saved lives around the world,” he said.
The study was supported by the Foundation for Research in Science and the Humanities at the University of Zurich, the Swiss National Science Foundation, and the U.S. National Institute of Allergy and Infectious Diseases. The researchers, Dr. Pal, and Dr. Wilson had no financial conflicts.
*This article was updated on 2/1/2022.
FROM ANNALS OF INTERNAL MEDICINE
Buprenorphine may curb opioid-induced respiratory depression
High plasma concentrations of buprenorphine may reduce fentanyl-induced respiratory depression, new research suggests.
The primary endpoint measure in a small “proof of principal” pharmacology study was effect of escalating fentanyl dosing on respiratory depression by way of decreased isohypercapnic minute ventilation (VE) – or volume of gas inhaled or exhaled per minute from the lungs.
Results showed the maximum decrease in highest-dose fentanyl-induced VE was almost 50% less for opioid-tolerant patients receiving a 2.0 ng/mL concentration of steady-state plasma buprenorphine than when receiving matching placebo.
Risk for apnea requiring stimulation after fentanyl dosing was also significantly lower with buprenorphine.
“Even though the study is small, a lot of data were collected which will allow us to very accurately predict which plasma concentrations, and therefore drug doses, are needed to protect people adequately in practice,” study coinvestigator Geert Jan Groeneveld, MD, PhD, neurologist and clinical pharmacologist at the Centre for Human Drug Research, Leiden, the Netherlands, and professor of clinical neuropharmacology at Leiden University Medical Center, told this news organization.
He added the “beautiful results” were in line with what the researchers expected and although further research is needed, the study provides a lot of useful information for clinicians.
“I think this is an approach that works, and this study makes that clear,” Dr. Groeneveld added.
The findings were published online Jan. 27, 2022, in PLoS One.
High death rate from synthetic opioids
A recent report from the Centers for Disease Control and Prevention noted that, between June 2020 and June 2021, there were more than 100,000 drug overdose deaths in the United States. Of these, more than 73,000 were attributed to opioids and more than 60,000 to synthetic opioids such as fentanyl.
Most opioid-related overdose deaths in the United States are attributable to synthetic opioids “that can unexpectedly cause respiratory depression by being ingested as a substitute for heroin or with [other] drugs,” Indivior noted in a press release.
Buprenorphine is a partial agonist that “binds with high affinity to mu-opioid receptors but displays partial respiratory depression effects,” the investigators wrote.
As reported by this news organization, the Food and Drug Administration approved buprenorphine extended release (Sublocade, Indivior) in 2017 as the first once-monthly injection for the treatment of opioid use disorder.
In the current study, which was conducted in Leiden, the Netherlands, the investigators used continuous intravenous buprenorphine in order to “mimic” the sustained plasma concentrations of the drug that can be delivered with the long-acting injectable, noted Christian Heidbreder, PhD, chief scientific officer at Indivior.
“This was an experimental medicine study, whereby we used intravenous buprenorphine to really understand the interaction with escalating doses of fentanyl” on respiratory depression, he told this news organization.
Two-part, two-period study
In part A, period one of the two-period crossover study, 14 healthy volunteers were randomly assigned to receive for 360 minutes continuous infusion of 0.02 or 0.05 mg/70 kg per hour of buprenorphine to target plasma concentrations of 0.2 or 0.5 ng/mL, respectively, or matching placebo. In the second period, participants received the alternative infusion – either placebo or the active drug.
In part B, eight opioid-tolerant patients who had used high-dose opioids for at least 3 months prior received a higher infusion rate of 0.1, 0.2, or 0.5 mg/70 kg per hour to target plasma concentrations of 1, 2, or 5 ng/mL, respectively.
The 2 ng/mL “is a very important threshold for us” and the result from several previous experiments, Dr. Heidbreder noted. So the investigators targeted that concentration as well as one below and one “much higher” in the current study.
“Because tolerance to opioid effects is poorly characterized in patients receiving long-term opioids, opioid-tolerant participants in part B had a fixed treatment sequence, receiving placebo infusion plus fentanyl challenges in period 1 to optimize the fentanyl dose escalation before buprenorphine and fentanyl were coadministered in period 2,” the investigators reported.
All participants received up to four escalating doses of intravenous fentanyl after reaching target buprenorphine plasma concentrations.
For healthy volunteers, the planned fentanyl doses were 0.075, 0.15, 0.25, and 0.35 mg/70 kg. For the opioid-tolerant patients, the doses were 0.25, 0.35, 0.5, and 0.7 mg/70 kg.
The infusions began after baseline VE had stabilized at 20 plus or minus 2 L/min, which is about four times above normal resting VE.
First clinical evidence?
Results showed fentanyl-induced adverse changes in VE were less at higher concentrations of buprenorphine plasma.
Opioid-tolerant patients receiving the 2.0 ng/mL concentration of buprenorphine had a 33.7% decrease in highest dose fentanyl-induced VE versus an 82.3% decrease when receiving placebo.
In addition, fentanyl reduced VE up to 49% (95% confidence interval, 21%-76%) in opioid-tolerant patients in all buprenorphine concentration groups combined versus reducing VE up to 100% (95% CI, 68%-132%) during placebo infusion (P = .006).
In addition, buprenorphine was associated with a lower risk versus placebo for apnea requiring verbal stimulation after fentanyl dosing (odds ratio, 0.07; P = .001).
For the healthy volunteers, the first fentanyl bolus reduced VE by 26% for those at target buprenorphine concentration of 0.5 ng/mL versus 51% when receiving placebo (P = .001). The second bolus reduced VE by 47% versus 79%, respectively (P < .001).
“Discontinuations for apnea limited treatment comparisons beyond the second fentanyl injection,” the investigators reported.
Overall, the findings “provide the first clinical evidence that high sustained plasma concentrations of buprenorphine may protect against respiratory depression induced by potent opioids,” they added.
Additional research is now “warranted to assess the competitive interaction of buprenorphine and fentanyl (as well as other illicitly manufactured fentanyl analogs) as we continue to deepen our understanding of buprenorphine as an evidence-based treatment for patients struggling with opioid use disorder,” Dr. Heidbreder said in a press release.
It’s unclear whether the study’s findings are generalizable to other populations, said Dr. Heidbreder.
“So and for that we’ll be using [the injectable] Sublocade as the medication of choice,” said Dr. Heidbreder.
“Conceptually, we feel confident about these data, but now we need to demonstrate what is happening in the real world,” he added.
The study was funded by Indivior. Dr. Groeneveld has reported no relevant financial relationships. Dr. Heidbreder is an employee of Indivior.
A version of this article first appeared on Medscape.com.
High plasma concentrations of buprenorphine may reduce fentanyl-induced respiratory depression, new research suggests.
The primary endpoint measure in a small “proof of principal” pharmacology study was effect of escalating fentanyl dosing on respiratory depression by way of decreased isohypercapnic minute ventilation (VE) – or volume of gas inhaled or exhaled per minute from the lungs.
Results showed the maximum decrease in highest-dose fentanyl-induced VE was almost 50% less for opioid-tolerant patients receiving a 2.0 ng/mL concentration of steady-state plasma buprenorphine than when receiving matching placebo.
Risk for apnea requiring stimulation after fentanyl dosing was also significantly lower with buprenorphine.
“Even though the study is small, a lot of data were collected which will allow us to very accurately predict which plasma concentrations, and therefore drug doses, are needed to protect people adequately in practice,” study coinvestigator Geert Jan Groeneveld, MD, PhD, neurologist and clinical pharmacologist at the Centre for Human Drug Research, Leiden, the Netherlands, and professor of clinical neuropharmacology at Leiden University Medical Center, told this news organization.
He added the “beautiful results” were in line with what the researchers expected and although further research is needed, the study provides a lot of useful information for clinicians.
“I think this is an approach that works, and this study makes that clear,” Dr. Groeneveld added.
The findings were published online Jan. 27, 2022, in PLoS One.
High death rate from synthetic opioids
A recent report from the Centers for Disease Control and Prevention noted that, between June 2020 and June 2021, there were more than 100,000 drug overdose deaths in the United States. Of these, more than 73,000 were attributed to opioids and more than 60,000 to synthetic opioids such as fentanyl.
Most opioid-related overdose deaths in the United States are attributable to synthetic opioids “that can unexpectedly cause respiratory depression by being ingested as a substitute for heroin or with [other] drugs,” Indivior noted in a press release.
Buprenorphine is a partial agonist that “binds with high affinity to mu-opioid receptors but displays partial respiratory depression effects,” the investigators wrote.
As reported by this news organization, the Food and Drug Administration approved buprenorphine extended release (Sublocade, Indivior) in 2017 as the first once-monthly injection for the treatment of opioid use disorder.
In the current study, which was conducted in Leiden, the Netherlands, the investigators used continuous intravenous buprenorphine in order to “mimic” the sustained plasma concentrations of the drug that can be delivered with the long-acting injectable, noted Christian Heidbreder, PhD, chief scientific officer at Indivior.
“This was an experimental medicine study, whereby we used intravenous buprenorphine to really understand the interaction with escalating doses of fentanyl” on respiratory depression, he told this news organization.
Two-part, two-period study
In part A, period one of the two-period crossover study, 14 healthy volunteers were randomly assigned to receive for 360 minutes continuous infusion of 0.02 or 0.05 mg/70 kg per hour of buprenorphine to target plasma concentrations of 0.2 or 0.5 ng/mL, respectively, or matching placebo. In the second period, participants received the alternative infusion – either placebo or the active drug.
In part B, eight opioid-tolerant patients who had used high-dose opioids for at least 3 months prior received a higher infusion rate of 0.1, 0.2, or 0.5 mg/70 kg per hour to target plasma concentrations of 1, 2, or 5 ng/mL, respectively.
The 2 ng/mL “is a very important threshold for us” and the result from several previous experiments, Dr. Heidbreder noted. So the investigators targeted that concentration as well as one below and one “much higher” in the current study.
“Because tolerance to opioid effects is poorly characterized in patients receiving long-term opioids, opioid-tolerant participants in part B had a fixed treatment sequence, receiving placebo infusion plus fentanyl challenges in period 1 to optimize the fentanyl dose escalation before buprenorphine and fentanyl were coadministered in period 2,” the investigators reported.
All participants received up to four escalating doses of intravenous fentanyl after reaching target buprenorphine plasma concentrations.
For healthy volunteers, the planned fentanyl doses were 0.075, 0.15, 0.25, and 0.35 mg/70 kg. For the opioid-tolerant patients, the doses were 0.25, 0.35, 0.5, and 0.7 mg/70 kg.
The infusions began after baseline VE had stabilized at 20 plus or minus 2 L/min, which is about four times above normal resting VE.
First clinical evidence?
Results showed fentanyl-induced adverse changes in VE were less at higher concentrations of buprenorphine plasma.
Opioid-tolerant patients receiving the 2.0 ng/mL concentration of buprenorphine had a 33.7% decrease in highest dose fentanyl-induced VE versus an 82.3% decrease when receiving placebo.
In addition, fentanyl reduced VE up to 49% (95% confidence interval, 21%-76%) in opioid-tolerant patients in all buprenorphine concentration groups combined versus reducing VE up to 100% (95% CI, 68%-132%) during placebo infusion (P = .006).
In addition, buprenorphine was associated with a lower risk versus placebo for apnea requiring verbal stimulation after fentanyl dosing (odds ratio, 0.07; P = .001).
For the healthy volunteers, the first fentanyl bolus reduced VE by 26% for those at target buprenorphine concentration of 0.5 ng/mL versus 51% when receiving placebo (P = .001). The second bolus reduced VE by 47% versus 79%, respectively (P < .001).
“Discontinuations for apnea limited treatment comparisons beyond the second fentanyl injection,” the investigators reported.
Overall, the findings “provide the first clinical evidence that high sustained plasma concentrations of buprenorphine may protect against respiratory depression induced by potent opioids,” they added.
Additional research is now “warranted to assess the competitive interaction of buprenorphine and fentanyl (as well as other illicitly manufactured fentanyl analogs) as we continue to deepen our understanding of buprenorphine as an evidence-based treatment for patients struggling with opioid use disorder,” Dr. Heidbreder said in a press release.
It’s unclear whether the study’s findings are generalizable to other populations, said Dr. Heidbreder.
“So and for that we’ll be using [the injectable] Sublocade as the medication of choice,” said Dr. Heidbreder.
“Conceptually, we feel confident about these data, but now we need to demonstrate what is happening in the real world,” he added.
The study was funded by Indivior. Dr. Groeneveld has reported no relevant financial relationships. Dr. Heidbreder is an employee of Indivior.
A version of this article first appeared on Medscape.com.
High plasma concentrations of buprenorphine may reduce fentanyl-induced respiratory depression, new research suggests.
The primary endpoint measure in a small “proof of principal” pharmacology study was effect of escalating fentanyl dosing on respiratory depression by way of decreased isohypercapnic minute ventilation (VE) – or volume of gas inhaled or exhaled per minute from the lungs.
Results showed the maximum decrease in highest-dose fentanyl-induced VE was almost 50% less for opioid-tolerant patients receiving a 2.0 ng/mL concentration of steady-state plasma buprenorphine than when receiving matching placebo.
Risk for apnea requiring stimulation after fentanyl dosing was also significantly lower with buprenorphine.
“Even though the study is small, a lot of data were collected which will allow us to very accurately predict which plasma concentrations, and therefore drug doses, are needed to protect people adequately in practice,” study coinvestigator Geert Jan Groeneveld, MD, PhD, neurologist and clinical pharmacologist at the Centre for Human Drug Research, Leiden, the Netherlands, and professor of clinical neuropharmacology at Leiden University Medical Center, told this news organization.
He added the “beautiful results” were in line with what the researchers expected and although further research is needed, the study provides a lot of useful information for clinicians.
“I think this is an approach that works, and this study makes that clear,” Dr. Groeneveld added.
The findings were published online Jan. 27, 2022, in PLoS One.
High death rate from synthetic opioids
A recent report from the Centers for Disease Control and Prevention noted that, between June 2020 and June 2021, there were more than 100,000 drug overdose deaths in the United States. Of these, more than 73,000 were attributed to opioids and more than 60,000 to synthetic opioids such as fentanyl.
Most opioid-related overdose deaths in the United States are attributable to synthetic opioids “that can unexpectedly cause respiratory depression by being ingested as a substitute for heroin or with [other] drugs,” Indivior noted in a press release.
Buprenorphine is a partial agonist that “binds with high affinity to mu-opioid receptors but displays partial respiratory depression effects,” the investigators wrote.
As reported by this news organization, the Food and Drug Administration approved buprenorphine extended release (Sublocade, Indivior) in 2017 as the first once-monthly injection for the treatment of opioid use disorder.
In the current study, which was conducted in Leiden, the Netherlands, the investigators used continuous intravenous buprenorphine in order to “mimic” the sustained plasma concentrations of the drug that can be delivered with the long-acting injectable, noted Christian Heidbreder, PhD, chief scientific officer at Indivior.
“This was an experimental medicine study, whereby we used intravenous buprenorphine to really understand the interaction with escalating doses of fentanyl” on respiratory depression, he told this news organization.
Two-part, two-period study
In part A, period one of the two-period crossover study, 14 healthy volunteers were randomly assigned to receive for 360 minutes continuous infusion of 0.02 or 0.05 mg/70 kg per hour of buprenorphine to target plasma concentrations of 0.2 or 0.5 ng/mL, respectively, or matching placebo. In the second period, participants received the alternative infusion – either placebo or the active drug.
In part B, eight opioid-tolerant patients who had used high-dose opioids for at least 3 months prior received a higher infusion rate of 0.1, 0.2, or 0.5 mg/70 kg per hour to target plasma concentrations of 1, 2, or 5 ng/mL, respectively.
The 2 ng/mL “is a very important threshold for us” and the result from several previous experiments, Dr. Heidbreder noted. So the investigators targeted that concentration as well as one below and one “much higher” in the current study.
“Because tolerance to opioid effects is poorly characterized in patients receiving long-term opioids, opioid-tolerant participants in part B had a fixed treatment sequence, receiving placebo infusion plus fentanyl challenges in period 1 to optimize the fentanyl dose escalation before buprenorphine and fentanyl were coadministered in period 2,” the investigators reported.
All participants received up to four escalating doses of intravenous fentanyl after reaching target buprenorphine plasma concentrations.
For healthy volunteers, the planned fentanyl doses were 0.075, 0.15, 0.25, and 0.35 mg/70 kg. For the opioid-tolerant patients, the doses were 0.25, 0.35, 0.5, and 0.7 mg/70 kg.
The infusions began after baseline VE had stabilized at 20 plus or minus 2 L/min, which is about four times above normal resting VE.
First clinical evidence?
Results showed fentanyl-induced adverse changes in VE were less at higher concentrations of buprenorphine plasma.
Opioid-tolerant patients receiving the 2.0 ng/mL concentration of buprenorphine had a 33.7% decrease in highest dose fentanyl-induced VE versus an 82.3% decrease when receiving placebo.
In addition, fentanyl reduced VE up to 49% (95% confidence interval, 21%-76%) in opioid-tolerant patients in all buprenorphine concentration groups combined versus reducing VE up to 100% (95% CI, 68%-132%) during placebo infusion (P = .006).
In addition, buprenorphine was associated with a lower risk versus placebo for apnea requiring verbal stimulation after fentanyl dosing (odds ratio, 0.07; P = .001).
For the healthy volunteers, the first fentanyl bolus reduced VE by 26% for those at target buprenorphine concentration of 0.5 ng/mL versus 51% when receiving placebo (P = .001). The second bolus reduced VE by 47% versus 79%, respectively (P < .001).
“Discontinuations for apnea limited treatment comparisons beyond the second fentanyl injection,” the investigators reported.
Overall, the findings “provide the first clinical evidence that high sustained plasma concentrations of buprenorphine may protect against respiratory depression induced by potent opioids,” they added.
Additional research is now “warranted to assess the competitive interaction of buprenorphine and fentanyl (as well as other illicitly manufactured fentanyl analogs) as we continue to deepen our understanding of buprenorphine as an evidence-based treatment for patients struggling with opioid use disorder,” Dr. Heidbreder said in a press release.
It’s unclear whether the study’s findings are generalizable to other populations, said Dr. Heidbreder.
“So and for that we’ll be using [the injectable] Sublocade as the medication of choice,” said Dr. Heidbreder.
“Conceptually, we feel confident about these data, but now we need to demonstrate what is happening in the real world,” he added.
The study was funded by Indivior. Dr. Groeneveld has reported no relevant financial relationships. Dr. Heidbreder is an employee of Indivior.
A version of this article first appeared on Medscape.com.
FROM PLOS ONE
Yoga maneuver may prevent vasovagal syncope
The tadasana exercise – a movement-based contemplative practice that gradually corrects orthostatic imbalance by strengthening protective neuromuscular reflexes – practiced for just 15 minutes twice a day, was associated with the complete elimination of episodes of vasovagal syncope for many patients.
“These exercises are very easy to perform, inexpensive, and very effective. This is a very easy fix for a scary and potentially dangerous condition,” lead author Hygriv Rao, MD, said in an interview. “We are excited about these results. We thought it would work, but we did not expect it to be so effective. It seems to work for almost all patients.
“We found that, with the tadasana maneuver, episodes of full syncope, where the patient actually loses consciousness, ceased completely, and episodes of near-syncope, where the patient feels faint but does not completely pass out, were greatly reduced,” Dr. Rao added. “The actual loss of consciousness, which is the most dangerous part, is practically gone. This gives a lot of confidence to patients and their families.”
The researchers reported their initial results from a pilot study of the technique in a letter to JACC: Clinical Electrophysiology that was published online Jan. 26, 2022.
Dr. Rao, a cardiologist at the KIMS Hospitals, Hyderabad, India, explained that vasovagal syncope is a brief loss of consciousness caused by a neurologically induced drop in blood pressure caused by faulty neuromuscular reflexes.
It is typically triggered by emotional stress, prolonged standing, or getting up from a sitting position too quickly.
Very few treatments have been shown effective, with current management approaches involving avoiding triggers, increasing fluids, and if the individual feels an episode coming on, they can take steps to stop it by lying down, raising their legs, or lowering their head to increase blood flow to the brain.
“Recently, there has been a lot of interest in yoga as a preventative therapy for vasovagal syncope,” Dr. Rao noted. “We considered various yoga positions and we chose the tadasana maneuver to study in this context as it resembles exercises sometimes given to patients with vasovagal syncope but with some differences including the addition of synchronized breathing, which may help stabilize autonomic tone.”
For the tadasana maneuver, the individual stands straight with their feet together, arms by their side (against a wall if they need support), and alternatively lift the front and back part of their feet.
They first lift their toes with their weight resting on the ball of their feet, then after a few seconds they raise their heels with their weight on the front of the foot. Then after a few more seconds they lift their arms over their shoulders, stretching upward while standing on their toes.
These movements are synchronized with breathing exercises, with the individual taking a deep breath in as they lift their arms and breathing out again on lowering the arms.
“Each movement takes a few seconds, and each cycle of movements takes about 2 minutes. If this is performed 8 times, then this would take about 15 minutes. We recommend this 15-minute routine twice a day,” Dr. Rao said.
For the current study, 113 patients diagnosed with recurrent vasovagal disorder were counseled to practice standard physical maneuvers and maintain adequate hydration. Medications were prescribed at the discretion of the treating physician.
Of these, 61 patients were additionally trained to practice the tadasana maneuver and asked to practice the movement for 15 minutes twice a day. The mean durations of symptoms and follow-up in the two groups were similar. The average follow-up was about 20 months.
Results showed that episodes of both near-syncope and syncope decreased in both groups but there was a much larger reduction in the patients practicing the tadasana maneuver.
Before treatment, the 52 patients in the conventional group experienced 163 syncope or near-syncope events. At follow-up, 22 symptom recurrences occurred in 12 patients (23%). Total mean events per patient declined from 3 to 0.4.
Full syncope events in this group declined from 65 in 32 patients to 2 in 2 patients (mean per patient, 1.3 to 1), and near-syncope events fell from 98 in 34 patients to 20 in 10 patients (mean per patient, 2.0 to 0.4).
In the tadasana group, 61 patients had 378 syncope/near-syncope events before treatment; at follow-up, only 6 events occurred in 5 patients (8%). Per patient, total events declined from a mean of 6 to 0.1.
Full syncope events fell from 108 in 48 patients to 0 (mean per patient, 1.8 to 0), and near-syncope events declined from 269 in 33 patients to 6 in 5 patients (mean per patient, 4.4 to 0.1).
“This combination of exercise and breathing influences the neuromuscular reflex malfunction that occurs in vasovagal syncope,” Dr. Rao noted. “The movements focus on strengthening neuromuscular reflexes in the quadriceps and the calf muscles, which can increase the blood circulation and venous return, thus preventing blood pooling in the lower body.”
The researchers said this pilot study offers three main findings. First, both conventional therapy and conventional plus tadasana therapy appeared to benefit patients, compared with their respective baseline symptom burden. Second, application of tadasana as an adjunctive treatment was associated with fewer total event recurrences (that is, syncope and near-syncope combined), and third, tadasana was well tolerated, with no adverse events reported.
“The reduction in total events (i.e., syncope and near-syncope events), compared with pretreatment numbers, was substantial and most tadasana patients were managed without any pharmacotherapy,” the authors reported.
Dr. Rao noted that at baseline almost all patients in both groups were taking medications for the condition, but during the study these medications were reduced as fewer episodes occurred. At the end of the follow-up, 80% of the conventional group were still taking medication, compared with just 14% of those in the tadasana group.
Patients had an initial training session in person with a yoga instructor and then received follow-on training by video online. Dr. Rao said there was a very high rate of compliance, “almost 100%.”
He reports that a total of 200 patients have now been treated with this approach at his hospital with very similar results to those seen in the initial study.
This work was supported in part by a grant from the Dr Earl E. Bakken Family in support of heart-brain research. Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The tadasana exercise – a movement-based contemplative practice that gradually corrects orthostatic imbalance by strengthening protective neuromuscular reflexes – practiced for just 15 minutes twice a day, was associated with the complete elimination of episodes of vasovagal syncope for many patients.
“These exercises are very easy to perform, inexpensive, and very effective. This is a very easy fix for a scary and potentially dangerous condition,” lead author Hygriv Rao, MD, said in an interview. “We are excited about these results. We thought it would work, but we did not expect it to be so effective. It seems to work for almost all patients.
“We found that, with the tadasana maneuver, episodes of full syncope, where the patient actually loses consciousness, ceased completely, and episodes of near-syncope, where the patient feels faint but does not completely pass out, were greatly reduced,” Dr. Rao added. “The actual loss of consciousness, which is the most dangerous part, is practically gone. This gives a lot of confidence to patients and their families.”
The researchers reported their initial results from a pilot study of the technique in a letter to JACC: Clinical Electrophysiology that was published online Jan. 26, 2022.
Dr. Rao, a cardiologist at the KIMS Hospitals, Hyderabad, India, explained that vasovagal syncope is a brief loss of consciousness caused by a neurologically induced drop in blood pressure caused by faulty neuromuscular reflexes.
It is typically triggered by emotional stress, prolonged standing, or getting up from a sitting position too quickly.
Very few treatments have been shown effective, with current management approaches involving avoiding triggers, increasing fluids, and if the individual feels an episode coming on, they can take steps to stop it by lying down, raising their legs, or lowering their head to increase blood flow to the brain.
“Recently, there has been a lot of interest in yoga as a preventative therapy for vasovagal syncope,” Dr. Rao noted. “We considered various yoga positions and we chose the tadasana maneuver to study in this context as it resembles exercises sometimes given to patients with vasovagal syncope but with some differences including the addition of synchronized breathing, which may help stabilize autonomic tone.”
For the tadasana maneuver, the individual stands straight with their feet together, arms by their side (against a wall if they need support), and alternatively lift the front and back part of their feet.
They first lift their toes with their weight resting on the ball of their feet, then after a few seconds they raise their heels with their weight on the front of the foot. Then after a few more seconds they lift their arms over their shoulders, stretching upward while standing on their toes.
These movements are synchronized with breathing exercises, with the individual taking a deep breath in as they lift their arms and breathing out again on lowering the arms.
“Each movement takes a few seconds, and each cycle of movements takes about 2 minutes. If this is performed 8 times, then this would take about 15 minutes. We recommend this 15-minute routine twice a day,” Dr. Rao said.
For the current study, 113 patients diagnosed with recurrent vasovagal disorder were counseled to practice standard physical maneuvers and maintain adequate hydration. Medications were prescribed at the discretion of the treating physician.
Of these, 61 patients were additionally trained to practice the tadasana maneuver and asked to practice the movement for 15 minutes twice a day. The mean durations of symptoms and follow-up in the two groups were similar. The average follow-up was about 20 months.
Results showed that episodes of both near-syncope and syncope decreased in both groups but there was a much larger reduction in the patients practicing the tadasana maneuver.
Before treatment, the 52 patients in the conventional group experienced 163 syncope or near-syncope events. At follow-up, 22 symptom recurrences occurred in 12 patients (23%). Total mean events per patient declined from 3 to 0.4.
Full syncope events in this group declined from 65 in 32 patients to 2 in 2 patients (mean per patient, 1.3 to 1), and near-syncope events fell from 98 in 34 patients to 20 in 10 patients (mean per patient, 2.0 to 0.4).
In the tadasana group, 61 patients had 378 syncope/near-syncope events before treatment; at follow-up, only 6 events occurred in 5 patients (8%). Per patient, total events declined from a mean of 6 to 0.1.
Full syncope events fell from 108 in 48 patients to 0 (mean per patient, 1.8 to 0), and near-syncope events declined from 269 in 33 patients to 6 in 5 patients (mean per patient, 4.4 to 0.1).
“This combination of exercise and breathing influences the neuromuscular reflex malfunction that occurs in vasovagal syncope,” Dr. Rao noted. “The movements focus on strengthening neuromuscular reflexes in the quadriceps and the calf muscles, which can increase the blood circulation and venous return, thus preventing blood pooling in the lower body.”
The researchers said this pilot study offers three main findings. First, both conventional therapy and conventional plus tadasana therapy appeared to benefit patients, compared with their respective baseline symptom burden. Second, application of tadasana as an adjunctive treatment was associated with fewer total event recurrences (that is, syncope and near-syncope combined), and third, tadasana was well tolerated, with no adverse events reported.
“The reduction in total events (i.e., syncope and near-syncope events), compared with pretreatment numbers, was substantial and most tadasana patients were managed without any pharmacotherapy,” the authors reported.
Dr. Rao noted that at baseline almost all patients in both groups were taking medications for the condition, but during the study these medications were reduced as fewer episodes occurred. At the end of the follow-up, 80% of the conventional group were still taking medication, compared with just 14% of those in the tadasana group.
Patients had an initial training session in person with a yoga instructor and then received follow-on training by video online. Dr. Rao said there was a very high rate of compliance, “almost 100%.”
He reports that a total of 200 patients have now been treated with this approach at his hospital with very similar results to those seen in the initial study.
This work was supported in part by a grant from the Dr Earl E. Bakken Family in support of heart-brain research. Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The tadasana exercise – a movement-based contemplative practice that gradually corrects orthostatic imbalance by strengthening protective neuromuscular reflexes – practiced for just 15 minutes twice a day, was associated with the complete elimination of episodes of vasovagal syncope for many patients.
“These exercises are very easy to perform, inexpensive, and very effective. This is a very easy fix for a scary and potentially dangerous condition,” lead author Hygriv Rao, MD, said in an interview. “We are excited about these results. We thought it would work, but we did not expect it to be so effective. It seems to work for almost all patients.
“We found that, with the tadasana maneuver, episodes of full syncope, where the patient actually loses consciousness, ceased completely, and episodes of near-syncope, where the patient feels faint but does not completely pass out, were greatly reduced,” Dr. Rao added. “The actual loss of consciousness, which is the most dangerous part, is practically gone. This gives a lot of confidence to patients and their families.”
The researchers reported their initial results from a pilot study of the technique in a letter to JACC: Clinical Electrophysiology that was published online Jan. 26, 2022.
Dr. Rao, a cardiologist at the KIMS Hospitals, Hyderabad, India, explained that vasovagal syncope is a brief loss of consciousness caused by a neurologically induced drop in blood pressure caused by faulty neuromuscular reflexes.
It is typically triggered by emotional stress, prolonged standing, or getting up from a sitting position too quickly.
Very few treatments have been shown effective, with current management approaches involving avoiding triggers, increasing fluids, and if the individual feels an episode coming on, they can take steps to stop it by lying down, raising their legs, or lowering their head to increase blood flow to the brain.
“Recently, there has been a lot of interest in yoga as a preventative therapy for vasovagal syncope,” Dr. Rao noted. “We considered various yoga positions and we chose the tadasana maneuver to study in this context as it resembles exercises sometimes given to patients with vasovagal syncope but with some differences including the addition of synchronized breathing, which may help stabilize autonomic tone.”
For the tadasana maneuver, the individual stands straight with their feet together, arms by their side (against a wall if they need support), and alternatively lift the front and back part of their feet.
They first lift their toes with their weight resting on the ball of their feet, then after a few seconds they raise their heels with their weight on the front of the foot. Then after a few more seconds they lift their arms over their shoulders, stretching upward while standing on their toes.
These movements are synchronized with breathing exercises, with the individual taking a deep breath in as they lift their arms and breathing out again on lowering the arms.
“Each movement takes a few seconds, and each cycle of movements takes about 2 minutes. If this is performed 8 times, then this would take about 15 minutes. We recommend this 15-minute routine twice a day,” Dr. Rao said.
For the current study, 113 patients diagnosed with recurrent vasovagal disorder were counseled to practice standard physical maneuvers and maintain adequate hydration. Medications were prescribed at the discretion of the treating physician.
Of these, 61 patients were additionally trained to practice the tadasana maneuver and asked to practice the movement for 15 minutes twice a day. The mean durations of symptoms and follow-up in the two groups were similar. The average follow-up was about 20 months.
Results showed that episodes of both near-syncope and syncope decreased in both groups but there was a much larger reduction in the patients practicing the tadasana maneuver.
Before treatment, the 52 patients in the conventional group experienced 163 syncope or near-syncope events. At follow-up, 22 symptom recurrences occurred in 12 patients (23%). Total mean events per patient declined from 3 to 0.4.
Full syncope events in this group declined from 65 in 32 patients to 2 in 2 patients (mean per patient, 1.3 to 1), and near-syncope events fell from 98 in 34 patients to 20 in 10 patients (mean per patient, 2.0 to 0.4).
In the tadasana group, 61 patients had 378 syncope/near-syncope events before treatment; at follow-up, only 6 events occurred in 5 patients (8%). Per patient, total events declined from a mean of 6 to 0.1.
Full syncope events fell from 108 in 48 patients to 0 (mean per patient, 1.8 to 0), and near-syncope events declined from 269 in 33 patients to 6 in 5 patients (mean per patient, 4.4 to 0.1).
“This combination of exercise and breathing influences the neuromuscular reflex malfunction that occurs in vasovagal syncope,” Dr. Rao noted. “The movements focus on strengthening neuromuscular reflexes in the quadriceps and the calf muscles, which can increase the blood circulation and venous return, thus preventing blood pooling in the lower body.”
The researchers said this pilot study offers three main findings. First, both conventional therapy and conventional plus tadasana therapy appeared to benefit patients, compared with their respective baseline symptom burden. Second, application of tadasana as an adjunctive treatment was associated with fewer total event recurrences (that is, syncope and near-syncope combined), and third, tadasana was well tolerated, with no adverse events reported.
“The reduction in total events (i.e., syncope and near-syncope events), compared with pretreatment numbers, was substantial and most tadasana patients were managed without any pharmacotherapy,” the authors reported.
Dr. Rao noted that at baseline almost all patients in both groups were taking medications for the condition, but during the study these medications were reduced as fewer episodes occurred. At the end of the follow-up, 80% of the conventional group were still taking medication, compared with just 14% of those in the tadasana group.
Patients had an initial training session in person with a yoga instructor and then received follow-on training by video online. Dr. Rao said there was a very high rate of compliance, “almost 100%.”
He reports that a total of 200 patients have now been treated with this approach at his hospital with very similar results to those seen in the initial study.
This work was supported in part by a grant from the Dr Earl E. Bakken Family in support of heart-brain research. Dr. Rao disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JACC: CLINICAL ELECTROPHYSIOLOGY
Hong Kong, U.S., Israeli data illuminate COVID vaccine myocarditis
Why some COVID-19 vaccines seem occasionally to cause a distinctive form of myocarditis, and why adolescent boys and young men appear most vulnerable, remain a mystery. But the entity’s prevalence, nuances of presentation, and likely clinical course have come into sharper view after recent additions to the literature.
Two new publications all but confirm that the rare cases of myocarditis closely following vaccination against SARS-CoV-2, primarily with one of the mRNA-based vaccines from Pfizer-BioNTech and Moderna, is a clinically different creature from myocarditis physicians were likely to see before the pandemic.
A third report unveils rates of hospitalization for myocarditis linked to Pfizer-BioNTech vaccination in the 12- to 15-year age group, based on active surveillance across Israel. Of note, the rates were lower than corresponding numbers among the country’s 16- to 19-year-olds published in late 2021 by the same authors.
No link with CoronaVac
A case-control study covering almost the entire population of Hong Kong from February to August 2021 confirms a slight but significant excess risk for myocarditis and, to a lesser degree, pericarditis, after injections of the Pfizer-BioNTech vaccine. As consistently reported from other studies, the risks were highest in adolescent and young adult males and after a second dose.
The study estimated an overall carditis incidence of 5.7 cases per million doses of Pfizer-BioNTech, for a risk 3.5 times that in the unvaccinated Hong Kong population. Carditis rates after a first dose were about 2.5 per million and 10 per million after a second dose.
Hong Kong launched its public SARS-CoV-2 immunization program in late February 2021 with the Chinese-made CoronaVac (Sinovac) inactivated-virus vaccine, and introduced the mRNA-based alternative several weeks later. By August 2021, the vaccines had reached about 3.3 million people in the region – 49% of the Hong Kong population at least 12 years of age.
In a novel finding, there were no excesses in carditis cases after CoronaVac vaccination. The difference between vaccines likely isn’t caused by chance, because three-fourths of the carditis-associated Pfizer-BioNTech injections arose within a week, whereas “71% of cases following the use of CoronaVac occurred more than 30 days after vaccination,” senior author Ian Chi Kei Wong, PhD, University of Hong Kong, said in an interview.
“This onset distribution for cases having received CoronaVac demonstrates that it is highly unlikely the carditis cases are related to the vaccine,” he said. And that “plausibly implies a specific underlying mechanism between vaccination and carditis that may only be applicable to mRNA vaccines.”
That inference is in line with case reports and other research, including large population-based studies from Israel and Denmark, although a recent study from the United Kingdom hinted at a potential excess myocarditis risk associated with the adenovirus-based AstraZeneca-Oxford vaccine.
The Hong Kong study identified 160 patients age 12 or older with a first diagnosis of carditis during February to August 2021, in electronic health records covering nearly the entire region.
“We used laboratory test results of troponin levels to further eliminate unlikely cases of carditis,” Dr. Wong said. The health records were linked to a “population-based vaccination record” maintained by the government’s department of health.
About 10 control patients from among all hospitalized patients without carditis were matched by age, sex, and admission date to each of the 160 carditis cases. About 83% of cases and 92% of the controls were unvaccinated.
Among those who received the Pfizer-BioNTech vaccine, representing 12.5% of cases and 4.2% of controls, the estimated carditis incidence was 0.57 per 100,000 doses. For those who received CoronaVac, representing 4.4% of cases and 3.9% of controls, it was 0.31 per 100,000 doses.
In adjusted analysis, the odds ratios for carditis among Pfizer-BioNTech vaccine recipients, compared with unvaccinated controls, were 3.57 (95% confidence interval, 1.93-6.60) overall, 4.68 (95% CI, 2.25-9.71) for males, 2.22 (95% CI, 0.57-8.69) for females, 2.41 (95% CI, 1.18-4.90) for ages 18 and older, and 13.8 (95% CI, 2.86-110.4) for ages 12-17
Myocarditis accounted for most of the excess cases, with an overall OR of 9.29 (95% CI, 3.94-21.9). The OR reached only 1.06 (95% CI, 0.35-3.22) for pericarditis alone.
The case-control study is noteworthy for its design, which contrasts with the many recent case series and passive or active surveillance studies, and even the more robust population-based studies of vaccine-related myocarditis, observed Dongngan Truong, MD, University of Utah and Primary Children’s Hospital, both in Salt Lake City, who wasn’t part of the study.
Among its strengths, she said in an interview, are its linkage of comprehensive hospital and vaccination data sets for two different vaccines; and that it corroborates other research suggesting there is “something in particular about mRNA vaccination that seems to be associated with the development of myocarditis.”
Active surveillance in Israel
In an October 2021 report based on an Israeli Ministry of Health database covering up to May 2021, rates of myocarditis arising within 21 days of a second Pfizer-BioNTech dose in 16- to 19-year-olds reached about 1 per 6,637 males and 1 per 99,853 females. Those numbers compared with 1 per 26,000 males and 1 per 218,000 females across all age groups.
Now authors led by Dror Mevorach, MD, Hadassah Medical Center, Jerusalem, have published corresponding numbers from the same data base for myocarditis associated with the same vaccine in males and females aged 12-15.
Their research covers 404,407 people in that age group who received a first dose of the mRNA-based vaccine and 326,463 who received the second dose from June to October, 2021. Only 18 cases of myocarditis were observed within 21 days of either dose.
The estimated rates for males were 0.56 cases per 100,000 after a first dose and 8.09 cases per 100,000 after a second dose.
For females, the estimates were 0 cases per 100,000 after a first dose and 0.69 cases per 100,000 after a second dose.
“The pattern observed, mainly following the second vaccination in males, suggests causality,” the group wrote.
Leveraging passive surveillance reports
Another new report adds a twist to updated numbers from the U.S. Vaccine Adverse Event Reporting System (VAERS).
Prevalences derived from the passive-surveillance data base, known for including case records of inconsistent quality or completeness, are considered especially prone to reporting bias, the authors acknowledged.
The current analysis, however, plunges deep into VAERS-reported cases of presumed SARS-CoV-2 vaccine-associated myocarditis to help clarify “more of the characteristics of the patients and some of the treatments and short-term outcomes,” Matthew E. Oster, MD, MPH, said in an interview.
Dr. Oster, from the Centers for Disease Control and Prevention and Emory University, Atlanta, is lead author on the study’s Jan. 25, 2022, publication in JAMA.
The group reviewed charts and interviewed involved clinicians to adjudicate and document presentations, therapies, and the clinical course of cases reported as SARS-CoV-2 vaccine–associated myocarditis from December 2020 to August 2021. Out of the nearly 2000 reports, which were limited to patients younger than 30, the group identified 1,626 likely cases of such myocarditis arising within 7 days of a second mRNA vaccine dose.
The confirmed cases consistently represented higher prevalences than expected compared with prepandemic myocarditis claims data for both sexes and across age groups spanning 12-29 years.
For example, rates were highest for adolescent males – about 106 and 71 cases per million second doses of the Pfizer-BioNTech vaccine in those aged 16-17 and 12-16, respectively, for example. They were lowest for women aged 25-29, at 2.23 cases per million second Pfizer-BioNTech doses; the highest rate among females was about 11 per million for the 16-17 age group.
The observed rates, Dr. Oster said, represent an update to VAERS numbers published June 2021 in Morbidity and Mortality Weekly Report covering cases through June 2021.
“Overall, the general risk of having myocarditis from the vaccines is still extremely low. Even in the highest risk groups, it is still extremely low, and still lower than the risk of having cardiac complications from COVID,” he noted.
How do patients fare clinically?
From their chart reviews and interviews with case clinicians, Dr. Oster said, “we started to learn quickly that this is really a different type of myocarditis.”
For example, its onset, typically within a few days of the potential immunologic cause, was more rapid than in viral myocarditis, and its symptoms resolved faster, the report notes. Clinical presentations tended to be less severe, treatments not as intensive, and outcomes not as serious, compared with “the kind of typical viral myocarditis that most of the providers were used to taking care of in the past,” he said. “The pattern for these cases was very consistent.”
The study covered VAERS reports of suspected myocarditis arising within a week of first dose of a mRNA-based vaccine from the United States launch of public vaccination in December 2020 to August 2021, the CDC-based group reported. By then, more than 192 million people in the country had received either the Pfizer-BioNTech (age 12 or older) or Moderna (age 18 or older) vaccines.
Of the 1,991 reports of myocarditis, including 391 also involving pericarditis, 1,626 met the study’s definition for myocarditis on adjudication; about 82% of the latter cases were in males.
Based on the investigators’ review of charts and clinician interviews connected with 826 cases that met their definition of myocarditis in patients younger than 30, 89% reported “chest pain, pressure, or discomfort” and 30% reported dyspnea or shortness of breath. Troponin levels were elevated in 98%, 72% of patients who underwent electrocardiography showed abnormalities, and 12% of those with echocardiography had left ventricular ejection fractions less than 50%.
About 96% were hospitalized, and presenting symptoms resolved by discharge in 87% of those with available data, the group noted. Among patients with data on in-hospital therapy, they wrote, NSAIDs were the most common therapy, in 87%.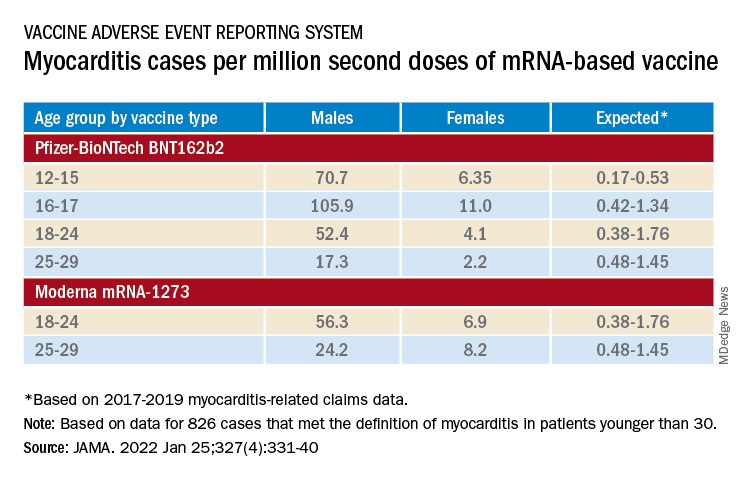
‘Mild and self-limiting’
The case-control study from Hong Kong didn’t specifically examine patients’ treatment and clinical course, but it does portray their vaccine-associated myocarditis as contrasting with more familiar viral myocarditis.
Patients with “typical” myocarditis tend to be “overall much sicker than what we’re seeing with myocarditis following vaccination,” Dr. Truong agreed. None of the 20 patients with myocarditis after Pfizer-BioNTech vaccination in Hong Kong were admitted to the intensive care unit. That, she added, suggests none required extracorporeal membrane oxygenation or vasoactive support, often necessary in viral myocarditis. “And they had shorter hospital stays.”
In contrast, Dr. Wong noted, 14 of the study’s unvaccinated patients required ICU admission; 12 of them died during the follow-up period. None with vaccine-related carditis died during the study’s follow-up. “We also showed that cases following [Pfizer-BioNTech] vaccination were all mild and self-limiting.”
Dr. Truong largely agreed that SARS-CoV-2 vaccine myocarditis and most myocarditis seen before the pandemic can be viewed as distinct clinical entities, “at least in the short term. I think we do need to follow these patients to look at more long-term outcomes, because at this point I don’t think we know the long-term implications. But at least in the short term, it seems like these patients are different, are much less sick, and recover pretty quickly overall.”
Dr. Oster emphasized that the many and varied acute and long-term hazards from contracting COVID-19 far outweigh any risk for myocarditis from vaccination. But for individuals who were hit with myocarditis soon after their first mRNA vaccine dose, who have already established their susceptibility, he and his colleagues would recommend that they “consider alternatives and not get the vaccine again.”
Dr. Oster reported no relevant financial relationships. Dr. Wong and colleagues did not report any relevant disclosures. Dr. Truong has previously disclosed serving as a consultant to Pfizer.
A version of this article first appeared on Medscape.com.
Why some COVID-19 vaccines seem occasionally to cause a distinctive form of myocarditis, and why adolescent boys and young men appear most vulnerable, remain a mystery. But the entity’s prevalence, nuances of presentation, and likely clinical course have come into sharper view after recent additions to the literature.
Two new publications all but confirm that the rare cases of myocarditis closely following vaccination against SARS-CoV-2, primarily with one of the mRNA-based vaccines from Pfizer-BioNTech and Moderna, is a clinically different creature from myocarditis physicians were likely to see before the pandemic.
A third report unveils rates of hospitalization for myocarditis linked to Pfizer-BioNTech vaccination in the 12- to 15-year age group, based on active surveillance across Israel. Of note, the rates were lower than corresponding numbers among the country’s 16- to 19-year-olds published in late 2021 by the same authors.
No link with CoronaVac
A case-control study covering almost the entire population of Hong Kong from February to August 2021 confirms a slight but significant excess risk for myocarditis and, to a lesser degree, pericarditis, after injections of the Pfizer-BioNTech vaccine. As consistently reported from other studies, the risks were highest in adolescent and young adult males and after a second dose.
The study estimated an overall carditis incidence of 5.7 cases per million doses of Pfizer-BioNTech, for a risk 3.5 times that in the unvaccinated Hong Kong population. Carditis rates after a first dose were about 2.5 per million and 10 per million after a second dose.
Hong Kong launched its public SARS-CoV-2 immunization program in late February 2021 with the Chinese-made CoronaVac (Sinovac) inactivated-virus vaccine, and introduced the mRNA-based alternative several weeks later. By August 2021, the vaccines had reached about 3.3 million people in the region – 49% of the Hong Kong population at least 12 years of age.
In a novel finding, there were no excesses in carditis cases after CoronaVac vaccination. The difference between vaccines likely isn’t caused by chance, because three-fourths of the carditis-associated Pfizer-BioNTech injections arose within a week, whereas “71% of cases following the use of CoronaVac occurred more than 30 days after vaccination,” senior author Ian Chi Kei Wong, PhD, University of Hong Kong, said in an interview.
“This onset distribution for cases having received CoronaVac demonstrates that it is highly unlikely the carditis cases are related to the vaccine,” he said. And that “plausibly implies a specific underlying mechanism between vaccination and carditis that may only be applicable to mRNA vaccines.”
That inference is in line with case reports and other research, including large population-based studies from Israel and Denmark, although a recent study from the United Kingdom hinted at a potential excess myocarditis risk associated with the adenovirus-based AstraZeneca-Oxford vaccine.
The Hong Kong study identified 160 patients age 12 or older with a first diagnosis of carditis during February to August 2021, in electronic health records covering nearly the entire region.
“We used laboratory test results of troponin levels to further eliminate unlikely cases of carditis,” Dr. Wong said. The health records were linked to a “population-based vaccination record” maintained by the government’s department of health.
About 10 control patients from among all hospitalized patients without carditis were matched by age, sex, and admission date to each of the 160 carditis cases. About 83% of cases and 92% of the controls were unvaccinated.
Among those who received the Pfizer-BioNTech vaccine, representing 12.5% of cases and 4.2% of controls, the estimated carditis incidence was 0.57 per 100,000 doses. For those who received CoronaVac, representing 4.4% of cases and 3.9% of controls, it was 0.31 per 100,000 doses.
In adjusted analysis, the odds ratios for carditis among Pfizer-BioNTech vaccine recipients, compared with unvaccinated controls, were 3.57 (95% confidence interval, 1.93-6.60) overall, 4.68 (95% CI, 2.25-9.71) for males, 2.22 (95% CI, 0.57-8.69) for females, 2.41 (95% CI, 1.18-4.90) for ages 18 and older, and 13.8 (95% CI, 2.86-110.4) for ages 12-17
Myocarditis accounted for most of the excess cases, with an overall OR of 9.29 (95% CI, 3.94-21.9). The OR reached only 1.06 (95% CI, 0.35-3.22) for pericarditis alone.
The case-control study is noteworthy for its design, which contrasts with the many recent case series and passive or active surveillance studies, and even the more robust population-based studies of vaccine-related myocarditis, observed Dongngan Truong, MD, University of Utah and Primary Children’s Hospital, both in Salt Lake City, who wasn’t part of the study.
Among its strengths, she said in an interview, are its linkage of comprehensive hospital and vaccination data sets for two different vaccines; and that it corroborates other research suggesting there is “something in particular about mRNA vaccination that seems to be associated with the development of myocarditis.”
Active surveillance in Israel
In an October 2021 report based on an Israeli Ministry of Health database covering up to May 2021, rates of myocarditis arising within 21 days of a second Pfizer-BioNTech dose in 16- to 19-year-olds reached about 1 per 6,637 males and 1 per 99,853 females. Those numbers compared with 1 per 26,000 males and 1 per 218,000 females across all age groups.
Now authors led by Dror Mevorach, MD, Hadassah Medical Center, Jerusalem, have published corresponding numbers from the same data base for myocarditis associated with the same vaccine in males and females aged 12-15.
Their research covers 404,407 people in that age group who received a first dose of the mRNA-based vaccine and 326,463 who received the second dose from June to October, 2021. Only 18 cases of myocarditis were observed within 21 days of either dose.
The estimated rates for males were 0.56 cases per 100,000 after a first dose and 8.09 cases per 100,000 after a second dose.
For females, the estimates were 0 cases per 100,000 after a first dose and 0.69 cases per 100,000 after a second dose.
“The pattern observed, mainly following the second vaccination in males, suggests causality,” the group wrote.
Leveraging passive surveillance reports
Another new report adds a twist to updated numbers from the U.S. Vaccine Adverse Event Reporting System (VAERS).
Prevalences derived from the passive-surveillance data base, known for including case records of inconsistent quality or completeness, are considered especially prone to reporting bias, the authors acknowledged.
The current analysis, however, plunges deep into VAERS-reported cases of presumed SARS-CoV-2 vaccine-associated myocarditis to help clarify “more of the characteristics of the patients and some of the treatments and short-term outcomes,” Matthew E. Oster, MD, MPH, said in an interview.
Dr. Oster, from the Centers for Disease Control and Prevention and Emory University, Atlanta, is lead author on the study’s Jan. 25, 2022, publication in JAMA.
The group reviewed charts and interviewed involved clinicians to adjudicate and document presentations, therapies, and the clinical course of cases reported as SARS-CoV-2 vaccine–associated myocarditis from December 2020 to August 2021. Out of the nearly 2000 reports, which were limited to patients younger than 30, the group identified 1,626 likely cases of such myocarditis arising within 7 days of a second mRNA vaccine dose.
The confirmed cases consistently represented higher prevalences than expected compared with prepandemic myocarditis claims data for both sexes and across age groups spanning 12-29 years.
For example, rates were highest for adolescent males – about 106 and 71 cases per million second doses of the Pfizer-BioNTech vaccine in those aged 16-17 and 12-16, respectively, for example. They were lowest for women aged 25-29, at 2.23 cases per million second Pfizer-BioNTech doses; the highest rate among females was about 11 per million for the 16-17 age group.
The observed rates, Dr. Oster said, represent an update to VAERS numbers published June 2021 in Morbidity and Mortality Weekly Report covering cases through June 2021.
“Overall, the general risk of having myocarditis from the vaccines is still extremely low. Even in the highest risk groups, it is still extremely low, and still lower than the risk of having cardiac complications from COVID,” he noted.
How do patients fare clinically?
From their chart reviews and interviews with case clinicians, Dr. Oster said, “we started to learn quickly that this is really a different type of myocarditis.”
For example, its onset, typically within a few days of the potential immunologic cause, was more rapid than in viral myocarditis, and its symptoms resolved faster, the report notes. Clinical presentations tended to be less severe, treatments not as intensive, and outcomes not as serious, compared with “the kind of typical viral myocarditis that most of the providers were used to taking care of in the past,” he said. “The pattern for these cases was very consistent.”
The study covered VAERS reports of suspected myocarditis arising within a week of first dose of a mRNA-based vaccine from the United States launch of public vaccination in December 2020 to August 2021, the CDC-based group reported. By then, more than 192 million people in the country had received either the Pfizer-BioNTech (age 12 or older) or Moderna (age 18 or older) vaccines.
Of the 1,991 reports of myocarditis, including 391 also involving pericarditis, 1,626 met the study’s definition for myocarditis on adjudication; about 82% of the latter cases were in males.
Based on the investigators’ review of charts and clinician interviews connected with 826 cases that met their definition of myocarditis in patients younger than 30, 89% reported “chest pain, pressure, or discomfort” and 30% reported dyspnea or shortness of breath. Troponin levels were elevated in 98%, 72% of patients who underwent electrocardiography showed abnormalities, and 12% of those with echocardiography had left ventricular ejection fractions less than 50%.
About 96% were hospitalized, and presenting symptoms resolved by discharge in 87% of those with available data, the group noted. Among patients with data on in-hospital therapy, they wrote, NSAIDs were the most common therapy, in 87%.
‘Mild and self-limiting’
The case-control study from Hong Kong didn’t specifically examine patients’ treatment and clinical course, but it does portray their vaccine-associated myocarditis as contrasting with more familiar viral myocarditis.
Patients with “typical” myocarditis tend to be “overall much sicker than what we’re seeing with myocarditis following vaccination,” Dr. Truong agreed. None of the 20 patients with myocarditis after Pfizer-BioNTech vaccination in Hong Kong were admitted to the intensive care unit. That, she added, suggests none required extracorporeal membrane oxygenation or vasoactive support, often necessary in viral myocarditis. “And they had shorter hospital stays.”
In contrast, Dr. Wong noted, 14 of the study’s unvaccinated patients required ICU admission; 12 of them died during the follow-up period. None with vaccine-related carditis died during the study’s follow-up. “We also showed that cases following [Pfizer-BioNTech] vaccination were all mild and self-limiting.”
Dr. Truong largely agreed that SARS-CoV-2 vaccine myocarditis and most myocarditis seen before the pandemic can be viewed as distinct clinical entities, “at least in the short term. I think we do need to follow these patients to look at more long-term outcomes, because at this point I don’t think we know the long-term implications. But at least in the short term, it seems like these patients are different, are much less sick, and recover pretty quickly overall.”
Dr. Oster emphasized that the many and varied acute and long-term hazards from contracting COVID-19 far outweigh any risk for myocarditis from vaccination. But for individuals who were hit with myocarditis soon after their first mRNA vaccine dose, who have already established their susceptibility, he and his colleagues would recommend that they “consider alternatives and not get the vaccine again.”
Dr. Oster reported no relevant financial relationships. Dr. Wong and colleagues did not report any relevant disclosures. Dr. Truong has previously disclosed serving as a consultant to Pfizer.
A version of this article first appeared on Medscape.com.
Why some COVID-19 vaccines seem occasionally to cause a distinctive form of myocarditis, and why adolescent boys and young men appear most vulnerable, remain a mystery. But the entity’s prevalence, nuances of presentation, and likely clinical course have come into sharper view after recent additions to the literature.
Two new publications all but confirm that the rare cases of myocarditis closely following vaccination against SARS-CoV-2, primarily with one of the mRNA-based vaccines from Pfizer-BioNTech and Moderna, is a clinically different creature from myocarditis physicians were likely to see before the pandemic.
A third report unveils rates of hospitalization for myocarditis linked to Pfizer-BioNTech vaccination in the 12- to 15-year age group, based on active surveillance across Israel. Of note, the rates were lower than corresponding numbers among the country’s 16- to 19-year-olds published in late 2021 by the same authors.
No link with CoronaVac
A case-control study covering almost the entire population of Hong Kong from February to August 2021 confirms a slight but significant excess risk for myocarditis and, to a lesser degree, pericarditis, after injections of the Pfizer-BioNTech vaccine. As consistently reported from other studies, the risks were highest in adolescent and young adult males and after a second dose.
The study estimated an overall carditis incidence of 5.7 cases per million doses of Pfizer-BioNTech, for a risk 3.5 times that in the unvaccinated Hong Kong population. Carditis rates after a first dose were about 2.5 per million and 10 per million after a second dose.
Hong Kong launched its public SARS-CoV-2 immunization program in late February 2021 with the Chinese-made CoronaVac (Sinovac) inactivated-virus vaccine, and introduced the mRNA-based alternative several weeks later. By August 2021, the vaccines had reached about 3.3 million people in the region – 49% of the Hong Kong population at least 12 years of age.
In a novel finding, there were no excesses in carditis cases after CoronaVac vaccination. The difference between vaccines likely isn’t caused by chance, because three-fourths of the carditis-associated Pfizer-BioNTech injections arose within a week, whereas “71% of cases following the use of CoronaVac occurred more than 30 days after vaccination,” senior author Ian Chi Kei Wong, PhD, University of Hong Kong, said in an interview.
“This onset distribution for cases having received CoronaVac demonstrates that it is highly unlikely the carditis cases are related to the vaccine,” he said. And that “plausibly implies a specific underlying mechanism between vaccination and carditis that may only be applicable to mRNA vaccines.”
That inference is in line with case reports and other research, including large population-based studies from Israel and Denmark, although a recent study from the United Kingdom hinted at a potential excess myocarditis risk associated with the adenovirus-based AstraZeneca-Oxford vaccine.
The Hong Kong study identified 160 patients age 12 or older with a first diagnosis of carditis during February to August 2021, in electronic health records covering nearly the entire region.
“We used laboratory test results of troponin levels to further eliminate unlikely cases of carditis,” Dr. Wong said. The health records were linked to a “population-based vaccination record” maintained by the government’s department of health.
About 10 control patients from among all hospitalized patients without carditis were matched by age, sex, and admission date to each of the 160 carditis cases. About 83% of cases and 92% of the controls were unvaccinated.
Among those who received the Pfizer-BioNTech vaccine, representing 12.5% of cases and 4.2% of controls, the estimated carditis incidence was 0.57 per 100,000 doses. For those who received CoronaVac, representing 4.4% of cases and 3.9% of controls, it was 0.31 per 100,000 doses.
In adjusted analysis, the odds ratios for carditis among Pfizer-BioNTech vaccine recipients, compared with unvaccinated controls, were 3.57 (95% confidence interval, 1.93-6.60) overall, 4.68 (95% CI, 2.25-9.71) for males, 2.22 (95% CI, 0.57-8.69) for females, 2.41 (95% CI, 1.18-4.90) for ages 18 and older, and 13.8 (95% CI, 2.86-110.4) for ages 12-17
Myocarditis accounted for most of the excess cases, with an overall OR of 9.29 (95% CI, 3.94-21.9). The OR reached only 1.06 (95% CI, 0.35-3.22) for pericarditis alone.
The case-control study is noteworthy for its design, which contrasts with the many recent case series and passive or active surveillance studies, and even the more robust population-based studies of vaccine-related myocarditis, observed Dongngan Truong, MD, University of Utah and Primary Children’s Hospital, both in Salt Lake City, who wasn’t part of the study.
Among its strengths, she said in an interview, are its linkage of comprehensive hospital and vaccination data sets for two different vaccines; and that it corroborates other research suggesting there is “something in particular about mRNA vaccination that seems to be associated with the development of myocarditis.”
Active surveillance in Israel
In an October 2021 report based on an Israeli Ministry of Health database covering up to May 2021, rates of myocarditis arising within 21 days of a second Pfizer-BioNTech dose in 16- to 19-year-olds reached about 1 per 6,637 males and 1 per 99,853 females. Those numbers compared with 1 per 26,000 males and 1 per 218,000 females across all age groups.
Now authors led by Dror Mevorach, MD, Hadassah Medical Center, Jerusalem, have published corresponding numbers from the same data base for myocarditis associated with the same vaccine in males and females aged 12-15.
Their research covers 404,407 people in that age group who received a first dose of the mRNA-based vaccine and 326,463 who received the second dose from June to October, 2021. Only 18 cases of myocarditis were observed within 21 days of either dose.
The estimated rates for males were 0.56 cases per 100,000 after a first dose and 8.09 cases per 100,000 after a second dose.
For females, the estimates were 0 cases per 100,000 after a first dose and 0.69 cases per 100,000 after a second dose.
“The pattern observed, mainly following the second vaccination in males, suggests causality,” the group wrote.
Leveraging passive surveillance reports
Another new report adds a twist to updated numbers from the U.S. Vaccine Adverse Event Reporting System (VAERS).
Prevalences derived from the passive-surveillance data base, known for including case records of inconsistent quality or completeness, are considered especially prone to reporting bias, the authors acknowledged.
The current analysis, however, plunges deep into VAERS-reported cases of presumed SARS-CoV-2 vaccine-associated myocarditis to help clarify “more of the characteristics of the patients and some of the treatments and short-term outcomes,” Matthew E. Oster, MD, MPH, said in an interview.
Dr. Oster, from the Centers for Disease Control and Prevention and Emory University, Atlanta, is lead author on the study’s Jan. 25, 2022, publication in JAMA.
The group reviewed charts and interviewed involved clinicians to adjudicate and document presentations, therapies, and the clinical course of cases reported as SARS-CoV-2 vaccine–associated myocarditis from December 2020 to August 2021. Out of the nearly 2000 reports, which were limited to patients younger than 30, the group identified 1,626 likely cases of such myocarditis arising within 7 days of a second mRNA vaccine dose.
The confirmed cases consistently represented higher prevalences than expected compared with prepandemic myocarditis claims data for both sexes and across age groups spanning 12-29 years.
For example, rates were highest for adolescent males – about 106 and 71 cases per million second doses of the Pfizer-BioNTech vaccine in those aged 16-17 and 12-16, respectively, for example. They were lowest for women aged 25-29, at 2.23 cases per million second Pfizer-BioNTech doses; the highest rate among females was about 11 per million for the 16-17 age group.
The observed rates, Dr. Oster said, represent an update to VAERS numbers published June 2021 in Morbidity and Mortality Weekly Report covering cases through June 2021.
“Overall, the general risk of having myocarditis from the vaccines is still extremely low. Even in the highest risk groups, it is still extremely low, and still lower than the risk of having cardiac complications from COVID,” he noted.
How do patients fare clinically?
From their chart reviews and interviews with case clinicians, Dr. Oster said, “we started to learn quickly that this is really a different type of myocarditis.”
For example, its onset, typically within a few days of the potential immunologic cause, was more rapid than in viral myocarditis, and its symptoms resolved faster, the report notes. Clinical presentations tended to be less severe, treatments not as intensive, and outcomes not as serious, compared with “the kind of typical viral myocarditis that most of the providers were used to taking care of in the past,” he said. “The pattern for these cases was very consistent.”
The study covered VAERS reports of suspected myocarditis arising within a week of first dose of a mRNA-based vaccine from the United States launch of public vaccination in December 2020 to August 2021, the CDC-based group reported. By then, more than 192 million people in the country had received either the Pfizer-BioNTech (age 12 or older) or Moderna (age 18 or older) vaccines.
Of the 1,991 reports of myocarditis, including 391 also involving pericarditis, 1,626 met the study’s definition for myocarditis on adjudication; about 82% of the latter cases were in males.
Based on the investigators’ review of charts and clinician interviews connected with 826 cases that met their definition of myocarditis in patients younger than 30, 89% reported “chest pain, pressure, or discomfort” and 30% reported dyspnea or shortness of breath. Troponin levels were elevated in 98%, 72% of patients who underwent electrocardiography showed abnormalities, and 12% of those with echocardiography had left ventricular ejection fractions less than 50%.
About 96% were hospitalized, and presenting symptoms resolved by discharge in 87% of those with available data, the group noted. Among patients with data on in-hospital therapy, they wrote, NSAIDs were the most common therapy, in 87%.
‘Mild and self-limiting’
The case-control study from Hong Kong didn’t specifically examine patients’ treatment and clinical course, but it does portray their vaccine-associated myocarditis as contrasting with more familiar viral myocarditis.
Patients with “typical” myocarditis tend to be “overall much sicker than what we’re seeing with myocarditis following vaccination,” Dr. Truong agreed. None of the 20 patients with myocarditis after Pfizer-BioNTech vaccination in Hong Kong were admitted to the intensive care unit. That, she added, suggests none required extracorporeal membrane oxygenation or vasoactive support, often necessary in viral myocarditis. “And they had shorter hospital stays.”
In contrast, Dr. Wong noted, 14 of the study’s unvaccinated patients required ICU admission; 12 of them died during the follow-up period. None with vaccine-related carditis died during the study’s follow-up. “We also showed that cases following [Pfizer-BioNTech] vaccination were all mild and self-limiting.”
Dr. Truong largely agreed that SARS-CoV-2 vaccine myocarditis and most myocarditis seen before the pandemic can be viewed as distinct clinical entities, “at least in the short term. I think we do need to follow these patients to look at more long-term outcomes, because at this point I don’t think we know the long-term implications. But at least in the short term, it seems like these patients are different, are much less sick, and recover pretty quickly overall.”
Dr. Oster emphasized that the many and varied acute and long-term hazards from contracting COVID-19 far outweigh any risk for myocarditis from vaccination. But for individuals who were hit with myocarditis soon after their first mRNA vaccine dose, who have already established their susceptibility, he and his colleagues would recommend that they “consider alternatives and not get the vaccine again.”
Dr. Oster reported no relevant financial relationships. Dr. Wong and colleagues did not report any relevant disclosures. Dr. Truong has previously disclosed serving as a consultant to Pfizer.
A version of this article first appeared on Medscape.com.
'The whitest specialty,' revisited
A recent STAT article by Usha Lee McFarling identified orthopedic surgery as “the whitest specialty.” That’s a problem many, perhaps most, orthopedic surgeons are aware of. But seeing it stated so bluntly is jolting. It’s disconcerting to think that the orthopedic community is making so little progress toward achieving the principal ideal articulated in our country’s fundamental declaration of moral values: that all people are created equal and that they have inalienable rights – in our case, that everyone, Black, brown, as well as White, has the right to the same high level of medical care.
Unfortunately, as study after study has shown, minorities do not enjoy the right to equitable care. Instead, they are subject to disparities in treatment and outcomes that speak to the prejudices that are built into the health care system and are present – sometimes consciously, but most often subconsciously – in the minds of physicians. One important contributing element to these disparities is the paucity of minority practitioners. Studies have also shown that Black patients, for example, respond better to Black physicians, who so often share a psychological and cultural sympathy unavailable to most White physicians. It’s for that reason that being identified as “the whitest specialty” is so immensely troubling.
In researching her STAT article, Ms. McFarling spoke with American Academy of Orthopaedic Surgeons leaders, practicing surgeons, residents, and med students about the dearth of minority and female orthopedic surgeons. What she heard was perplexity and frustration about why better progress hasn’t been made toward correcting the gross underrepresentation of everyone other than White men. The AAOS, she noted, was one of the first specialties to recognize the lack of diversity and over the years has put in great effort to address the problem, creating task forces, committees, and diversity awards and sponsoring conferences and discussions. Yet progress has been glacial, at best.
From her respondents, Ms. McFarling heard an array of reasons for this. Black, Hispanic, and Native American persons are underrepresented in medical schools, so the pool of potential applicants for orthopedic residencies is shallow to begin with. STEM studies are notoriously inadequate in poorer primary and secondary schools, in which so many minority students are educated. The MCAT and USMLE Step 1 test, which play a role in acceptance to residencies, have been shown to be biased. The specialty has few Black or brown role models and, consequently, few advocates and a lack of mentorship. Overt bias may be fairly rare (though microaggressions are still a common and ongoing problem), but most minority and female orthopedic surgeons feel strongly that implicit or subconscious bias is entrenched and works against acceptance to residencies, success in residencies, and advancement in the field.
One of this article’s authors (AW) saw all these factors at work as a resident, then as an admissions committee member at both Yale and Harvard. But the fact is that other medical specialties face exactly these problems and barriers, and yet have been substantially more successful in overcoming them.
What seems to be distinctive about orthopedics is that the mindset which perpetuated (and still perpetuates) In this regard, Kristy Weber, MD, the first female president of the AAOS, told Ms. McFarling that the critical first step to bringing in more women or people of color is changing the culture. There seems to be a consensus about that.
So, what does that mean, given that the AAOS has made serious efforts in that regard that have clearly been less than effective?
The answer, as we see it, is first – to not give in to frustration. The time frames involved in changing customary states of mind are typically elongated, and the deeper the habituation, the longer transformation takes. Deep changes always mean a long, hard slog. For transformations of this sort to take place, the requirements are a general agreement on the value of the transformation, exposure to the destructive consequences of the customary modus operandi, and persuasion for why change needs to happen.
In orthopedics, the first requirement has been met. The AAOS espouses diversity and inclusion as a high-level value. In terms of the second two requirements – exposure and persuasion – orthopedic surgeons have been witness to events, campaigns, conferences, et cetera. But these have not been enough, which means that efforts need to be focused, enlarged, sustained, determined, and innovative.
Does the orthopedic surgery community have the ability to do that?
The answer is: Yes, it does.
Currently the orthopedic surgeon community boasts a number of organizations, groups, and individuals pushing for change, in addition to the AAOS’s Diversity Advisory Board. The predominantly African American J. Robert Gladden Orthopaedic Society, the Ruth Jackson Orthopaedic Society of female orthopedic physicians, and the Association of Latino Orthopaedic Surgeons are all energetic advocates, as is Nth Dimensions, the Perry Initiative, and various ad hoc and individual endeavors.
These are all strong proponents for their own groups in their own way. But history has shown in so many cases that concerted rather than individual action empowers advocacy, and what orthopedic surgery needs in its current situation of gross underrepresentation of minorities and women is an enhanced campaign to raise awareness and redouble persuasion.
One of many examples of the power of collective action is the Association of Minority Health Professions Schools founded by Dr. Louis Sullivan in 1977.* Dr. Sullivan (later secretary of the Department of Health & Human Services) was at that time the founding dean of Morehouse School of Medicine. Morehouse had been launched on a shoestring and needed funding urgently. Other Black health schools, such as Meharry Medical College and Tuskegee College of Veterinary Medicine, were in even more pressing financial need. The coalition of schools that Dr. Sullivan organized became a powerful force in Congress and the National Institutes of Health, magnitudes more effective in raising funds from government and other sources than the best individual efforts of the separate institutions.
Dr. Sullivan’s association is only one of a multitude of historical examples of the effectiveness of unified action. AAOS currently has no single officer charged with bringing together the efforts of the change assets that already exist. It could, perhaps should, have someone in that position. AAOS could invest that same office with a mandate to survey the other medical specialties and bring to bear the most effective diversity, equity, and inclusion (DEI) practices in their arsenals.
Finally, despite the attention AAOS has brought to DEI needs, a look at the organization’s strategic goals, its core values, and its “key enablers” finds not a single mention of diversity or inclusion. Given the country’s current focus on the need for equality, given the poor performance of the orthopedic surgery specialty in terms of inclusion, the obvious question is: Should there not be an official declaration positing diversity as a primary AAOS desideratum?
There is recent precedent for this in the American College of Physicians/American Board of Internal Medicine’s Physician Charter on Professionalism, which includes “social justice” as a primary goal of medical practice. This highlights and reinforces the humanitarian strivings of the profession. In light of the paralysis illuminated by Ms. McFarling’s STAT article, a clear, concise declaration by the AAOS of the value and need for DEI as a central component of the organization’s values should be high on the AAOS order of business. A commitment in that form would serve as a powerful catalyst for bringing orthopedic surgery into step with its sister specialties, as well as affirming the core egalitarian principle that underlies all of medical care.
Dr. White is the Ellen and Melvin Gordon Distinguished Professor of Medical Education and Professor of Orthopedic Surgery at Harvard Medical School, Boston. Dr. Chanoff is a founding board member of the Augustus A. White III Institute for Healthcare Equity. Neither Dr. White nor Dr. Chanoff reported any conflicts of interest. A version of this article first appeared on Medscape.com.
Correction, 2/1/22: An earlier version of this article omitted the title of "Dr." before Dr. Louis Sullivan's name.
A recent STAT article by Usha Lee McFarling identified orthopedic surgery as “the whitest specialty.” That’s a problem many, perhaps most, orthopedic surgeons are aware of. But seeing it stated so bluntly is jolting. It’s disconcerting to think that the orthopedic community is making so little progress toward achieving the principal ideal articulated in our country’s fundamental declaration of moral values: that all people are created equal and that they have inalienable rights – in our case, that everyone, Black, brown, as well as White, has the right to the same high level of medical care.
Unfortunately, as study after study has shown, minorities do not enjoy the right to equitable care. Instead, they are subject to disparities in treatment and outcomes that speak to the prejudices that are built into the health care system and are present – sometimes consciously, but most often subconsciously – in the minds of physicians. One important contributing element to these disparities is the paucity of minority practitioners. Studies have also shown that Black patients, for example, respond better to Black physicians, who so often share a psychological and cultural sympathy unavailable to most White physicians. It’s for that reason that being identified as “the whitest specialty” is so immensely troubling.
In researching her STAT article, Ms. McFarling spoke with American Academy of Orthopaedic Surgeons leaders, practicing surgeons, residents, and med students about the dearth of minority and female orthopedic surgeons. What she heard was perplexity and frustration about why better progress hasn’t been made toward correcting the gross underrepresentation of everyone other than White men. The AAOS, she noted, was one of the first specialties to recognize the lack of diversity and over the years has put in great effort to address the problem, creating task forces, committees, and diversity awards and sponsoring conferences and discussions. Yet progress has been glacial, at best.
From her respondents, Ms. McFarling heard an array of reasons for this. Black, Hispanic, and Native American persons are underrepresented in medical schools, so the pool of potential applicants for orthopedic residencies is shallow to begin with. STEM studies are notoriously inadequate in poorer primary and secondary schools, in which so many minority students are educated. The MCAT and USMLE Step 1 test, which play a role in acceptance to residencies, have been shown to be biased. The specialty has few Black or brown role models and, consequently, few advocates and a lack of mentorship. Overt bias may be fairly rare (though microaggressions are still a common and ongoing problem), but most minority and female orthopedic surgeons feel strongly that implicit or subconscious bias is entrenched and works against acceptance to residencies, success in residencies, and advancement in the field.
One of this article’s authors (AW) saw all these factors at work as a resident, then as an admissions committee member at both Yale and Harvard. But the fact is that other medical specialties face exactly these problems and barriers, and yet have been substantially more successful in overcoming them.
What seems to be distinctive about orthopedics is that the mindset which perpetuated (and still perpetuates) In this regard, Kristy Weber, MD, the first female president of the AAOS, told Ms. McFarling that the critical first step to bringing in more women or people of color is changing the culture. There seems to be a consensus about that.
So, what does that mean, given that the AAOS has made serious efforts in that regard that have clearly been less than effective?
The answer, as we see it, is first – to not give in to frustration. The time frames involved in changing customary states of mind are typically elongated, and the deeper the habituation, the longer transformation takes. Deep changes always mean a long, hard slog. For transformations of this sort to take place, the requirements are a general agreement on the value of the transformation, exposure to the destructive consequences of the customary modus operandi, and persuasion for why change needs to happen.
In orthopedics, the first requirement has been met. The AAOS espouses diversity and inclusion as a high-level value. In terms of the second two requirements – exposure and persuasion – orthopedic surgeons have been witness to events, campaigns, conferences, et cetera. But these have not been enough, which means that efforts need to be focused, enlarged, sustained, determined, and innovative.
Does the orthopedic surgery community have the ability to do that?
The answer is: Yes, it does.
Currently the orthopedic surgeon community boasts a number of organizations, groups, and individuals pushing for change, in addition to the AAOS’s Diversity Advisory Board. The predominantly African American J. Robert Gladden Orthopaedic Society, the Ruth Jackson Orthopaedic Society of female orthopedic physicians, and the Association of Latino Orthopaedic Surgeons are all energetic advocates, as is Nth Dimensions, the Perry Initiative, and various ad hoc and individual endeavors.
These are all strong proponents for their own groups in their own way. But history has shown in so many cases that concerted rather than individual action empowers advocacy, and what orthopedic surgery needs in its current situation of gross underrepresentation of minorities and women is an enhanced campaign to raise awareness and redouble persuasion.
One of many examples of the power of collective action is the Association of Minority Health Professions Schools founded by Dr. Louis Sullivan in 1977.* Dr. Sullivan (later secretary of the Department of Health & Human Services) was at that time the founding dean of Morehouse School of Medicine. Morehouse had been launched on a shoestring and needed funding urgently. Other Black health schools, such as Meharry Medical College and Tuskegee College of Veterinary Medicine, were in even more pressing financial need. The coalition of schools that Dr. Sullivan organized became a powerful force in Congress and the National Institutes of Health, magnitudes more effective in raising funds from government and other sources than the best individual efforts of the separate institutions.
Dr. Sullivan’s association is only one of a multitude of historical examples of the effectiveness of unified action. AAOS currently has no single officer charged with bringing together the efforts of the change assets that already exist. It could, perhaps should, have someone in that position. AAOS could invest that same office with a mandate to survey the other medical specialties and bring to bear the most effective diversity, equity, and inclusion (DEI) practices in their arsenals.
Finally, despite the attention AAOS has brought to DEI needs, a look at the organization’s strategic goals, its core values, and its “key enablers” finds not a single mention of diversity or inclusion. Given the country’s current focus on the need for equality, given the poor performance of the orthopedic surgery specialty in terms of inclusion, the obvious question is: Should there not be an official declaration positing diversity as a primary AAOS desideratum?
There is recent precedent for this in the American College of Physicians/American Board of Internal Medicine’s Physician Charter on Professionalism, which includes “social justice” as a primary goal of medical practice. This highlights and reinforces the humanitarian strivings of the profession. In light of the paralysis illuminated by Ms. McFarling’s STAT article, a clear, concise declaration by the AAOS of the value and need for DEI as a central component of the organization’s values should be high on the AAOS order of business. A commitment in that form would serve as a powerful catalyst for bringing orthopedic surgery into step with its sister specialties, as well as affirming the core egalitarian principle that underlies all of medical care.
Dr. White is the Ellen and Melvin Gordon Distinguished Professor of Medical Education and Professor of Orthopedic Surgery at Harvard Medical School, Boston. Dr. Chanoff is a founding board member of the Augustus A. White III Institute for Healthcare Equity. Neither Dr. White nor Dr. Chanoff reported any conflicts of interest. A version of this article first appeared on Medscape.com.
Correction, 2/1/22: An earlier version of this article omitted the title of "Dr." before Dr. Louis Sullivan's name.
A recent STAT article by Usha Lee McFarling identified orthopedic surgery as “the whitest specialty.” That’s a problem many, perhaps most, orthopedic surgeons are aware of. But seeing it stated so bluntly is jolting. It’s disconcerting to think that the orthopedic community is making so little progress toward achieving the principal ideal articulated in our country’s fundamental declaration of moral values: that all people are created equal and that they have inalienable rights – in our case, that everyone, Black, brown, as well as White, has the right to the same high level of medical care.
Unfortunately, as study after study has shown, minorities do not enjoy the right to equitable care. Instead, they are subject to disparities in treatment and outcomes that speak to the prejudices that are built into the health care system and are present – sometimes consciously, but most often subconsciously – in the minds of physicians. One important contributing element to these disparities is the paucity of minority practitioners. Studies have also shown that Black patients, for example, respond better to Black physicians, who so often share a psychological and cultural sympathy unavailable to most White physicians. It’s for that reason that being identified as “the whitest specialty” is so immensely troubling.
In researching her STAT article, Ms. McFarling spoke with American Academy of Orthopaedic Surgeons leaders, practicing surgeons, residents, and med students about the dearth of minority and female orthopedic surgeons. What she heard was perplexity and frustration about why better progress hasn’t been made toward correcting the gross underrepresentation of everyone other than White men. The AAOS, she noted, was one of the first specialties to recognize the lack of diversity and over the years has put in great effort to address the problem, creating task forces, committees, and diversity awards and sponsoring conferences and discussions. Yet progress has been glacial, at best.
From her respondents, Ms. McFarling heard an array of reasons for this. Black, Hispanic, and Native American persons are underrepresented in medical schools, so the pool of potential applicants for orthopedic residencies is shallow to begin with. STEM studies are notoriously inadequate in poorer primary and secondary schools, in which so many minority students are educated. The MCAT and USMLE Step 1 test, which play a role in acceptance to residencies, have been shown to be biased. The specialty has few Black or brown role models and, consequently, few advocates and a lack of mentorship. Overt bias may be fairly rare (though microaggressions are still a common and ongoing problem), but most minority and female orthopedic surgeons feel strongly that implicit or subconscious bias is entrenched and works against acceptance to residencies, success in residencies, and advancement in the field.
One of this article’s authors (AW) saw all these factors at work as a resident, then as an admissions committee member at both Yale and Harvard. But the fact is that other medical specialties face exactly these problems and barriers, and yet have been substantially more successful in overcoming them.
What seems to be distinctive about orthopedics is that the mindset which perpetuated (and still perpetuates) In this regard, Kristy Weber, MD, the first female president of the AAOS, told Ms. McFarling that the critical first step to bringing in more women or people of color is changing the culture. There seems to be a consensus about that.
So, what does that mean, given that the AAOS has made serious efforts in that regard that have clearly been less than effective?
The answer, as we see it, is first – to not give in to frustration. The time frames involved in changing customary states of mind are typically elongated, and the deeper the habituation, the longer transformation takes. Deep changes always mean a long, hard slog. For transformations of this sort to take place, the requirements are a general agreement on the value of the transformation, exposure to the destructive consequences of the customary modus operandi, and persuasion for why change needs to happen.
In orthopedics, the first requirement has been met. The AAOS espouses diversity and inclusion as a high-level value. In terms of the second two requirements – exposure and persuasion – orthopedic surgeons have been witness to events, campaigns, conferences, et cetera. But these have not been enough, which means that efforts need to be focused, enlarged, sustained, determined, and innovative.
Does the orthopedic surgery community have the ability to do that?
The answer is: Yes, it does.
Currently the orthopedic surgeon community boasts a number of organizations, groups, and individuals pushing for change, in addition to the AAOS’s Diversity Advisory Board. The predominantly African American J. Robert Gladden Orthopaedic Society, the Ruth Jackson Orthopaedic Society of female orthopedic physicians, and the Association of Latino Orthopaedic Surgeons are all energetic advocates, as is Nth Dimensions, the Perry Initiative, and various ad hoc and individual endeavors.
These are all strong proponents for their own groups in their own way. But history has shown in so many cases that concerted rather than individual action empowers advocacy, and what orthopedic surgery needs in its current situation of gross underrepresentation of minorities and women is an enhanced campaign to raise awareness and redouble persuasion.
One of many examples of the power of collective action is the Association of Minority Health Professions Schools founded by Dr. Louis Sullivan in 1977.* Dr. Sullivan (later secretary of the Department of Health & Human Services) was at that time the founding dean of Morehouse School of Medicine. Morehouse had been launched on a shoestring and needed funding urgently. Other Black health schools, such as Meharry Medical College and Tuskegee College of Veterinary Medicine, were in even more pressing financial need. The coalition of schools that Dr. Sullivan organized became a powerful force in Congress and the National Institutes of Health, magnitudes more effective in raising funds from government and other sources than the best individual efforts of the separate institutions.
Dr. Sullivan’s association is only one of a multitude of historical examples of the effectiveness of unified action. AAOS currently has no single officer charged with bringing together the efforts of the change assets that already exist. It could, perhaps should, have someone in that position. AAOS could invest that same office with a mandate to survey the other medical specialties and bring to bear the most effective diversity, equity, and inclusion (DEI) practices in their arsenals.
Finally, despite the attention AAOS has brought to DEI needs, a look at the organization’s strategic goals, its core values, and its “key enablers” finds not a single mention of diversity or inclusion. Given the country’s current focus on the need for equality, given the poor performance of the orthopedic surgery specialty in terms of inclusion, the obvious question is: Should there not be an official declaration positing diversity as a primary AAOS desideratum?
There is recent precedent for this in the American College of Physicians/American Board of Internal Medicine’s Physician Charter on Professionalism, which includes “social justice” as a primary goal of medical practice. This highlights and reinforces the humanitarian strivings of the profession. In light of the paralysis illuminated by Ms. McFarling’s STAT article, a clear, concise declaration by the AAOS of the value and need for DEI as a central component of the organization’s values should be high on the AAOS order of business. A commitment in that form would serve as a powerful catalyst for bringing orthopedic surgery into step with its sister specialties, as well as affirming the core egalitarian principle that underlies all of medical care.
Dr. White is the Ellen and Melvin Gordon Distinguished Professor of Medical Education and Professor of Orthopedic Surgery at Harvard Medical School, Boston. Dr. Chanoff is a founding board member of the Augustus A. White III Institute for Healthcare Equity. Neither Dr. White nor Dr. Chanoff reported any conflicts of interest. A version of this article first appeared on Medscape.com.
Correction, 2/1/22: An earlier version of this article omitted the title of "Dr." before Dr. Louis Sullivan's name.