User login
Why is breast density a weighty matter?
Case: Patient seeks clarification and next steps on her breast density classification
Your patient, a 51-year-old postmenopausal woman (G0P0) in good health, had an annual screening mammogram that showed no evidence of malignancy. She is white and has a mother with a history of breast cancer. She has never had a breast biopsy. Following the mammogram, she received a letter from the imaging center, stating:
She calls your office and asks, “What should I do next?”
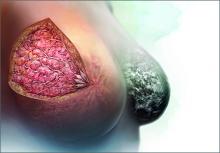
Breasts are composed of fibrous, glandular, and adipose tissue. If the breasts contain a lot of fibrous and glandular tissue, and little adipose tissue, they are considered to be “dense.” Using mammography, the current standard is to report the density of breast tissue using 4 categories:
- almost entirely fatty
- scattered fibroglandular densities
- heterogeneously dense
- extremely dense.
Dense breast tissue is defined to include the 2 categories heterogeneously dense and extremely dense.
Observational studies have reported that dense breast tissue is associated with an increased risk of breast cancer, and dense breast tissue makes it more difficult to detect breast cancer on mammography. According to data from the Breast Cancer Surveillance Consortium, among women aged 50 or older, the relative risk of breast cancer stratified by the 4 categories of breast density is 0.59, 1.00, 1.46, and 1.77, for almost entirely fatty, scattered fibroglandular densities, heterogeneously dense, and extremely dense, respectively.1 In one study, the sensitivity of mammography to detect breast cancer was 82% to 88% for women with nondense breasts and 62% to 69% in women with dense breasts.2 These data have catalyzed investigators to explore the use of supplemental imaging to enhance cancer detection in women with dense breasts.
The link between breast density and breast cancer risk and reduced sensitivity of mammography also has catalyzed activists and legislators to champion breast density notification laws, which have passed in more than 20 states. These laws require facilities that perform mammography to notify women with dense breasts that this finding is associated with an increased risk of breast cancer and that dense breasts reduce the ability of mammography to detect cancer. In some states, the law mandates that women with dense breasts be offered supplemental ultrasound imaging and that insurers must cover the cost of the ultrasound studies. Many of the laws recommend that the patient discuss the situation with the clinician who ordered the mammogram.
When I first saw the recommendation for patients to contact me about how to manage dense breasts, my initial response was, “Who? Me?” I felt ill equipped to provide any useful advice and suspected that many of my patients knew more than I about this issue.
Based on a review of the evidence, my current clinical recommendation is outlined in the 2 options below, including a low-resource utilization option and a high-resource utilization option. For patients, physicians, and health systems that are concerned that excessive breast cancer screening tests might cause more harm than benefit, the identification of dense breasts on mammogram is unlikely to be a trigger to perform any additional testing. In this situation, the pragmatic low-resource option is most relevant.
Alternatively, for patients and physicians who strongly believe in the value of screening mammography (see “Utilize tomosynthesis digital mammography technology for your patients” below), a reasonable strategy is to recommend that women with dense breasts and an increased risk for breast cancer be offered supplemental imaging.
In this editorial I elaborate these 2 approaches to breast cancer screening in women with dense breasts.
Utilize tomosynthesis digital mammography technology for your patients
Mammograms are the primary modality used for breast cancer screening because screening mammography has been shown to reduce breast cancer deaths by 15% to 30%.1,2 Annual or biennial mammograms are recommended for women aged 40 years or older by many professional organizations, including the American College of Obstetricians and Gynecologists and the American College of Radiology. However, mammography screening programs have been criticized because of false-positive tests resulting in unnecessary biopsies, limited sensitivity, and the theoretical risk of over-diagnosing clinically insignificant cancers.3,4
Mammography technology continues to evolve. Film-based mammography has been replaced by digital mammography. Tomosynthesis digital mammography, also known as 3-D mammography, is now replacing standard digital mammography.5
With tomosynthesis, digital mammography image acquisition is performed using an x-ray source that moves through an arc across the breast with the capture of a series of images from different angles and reconstruction of the data into thin slices approximately 1 mm in width. The presentation of breast images in thin slices permits superior detection of lesions. In addition, the collected images can be reconstructed to present a virtual 2-D image for analysis.
Tomosynthesis has been demonstrated to increase the sensitivity of mammography to detect cancer and reduce false-positive examinations. In a study of 454,850 mammography examinations, investigators found that the invasive cancer detection rate per 1,000 studies increased from 2.9 with standard digital mammography to 4.1 with tomosynthesis.6
Tomosynthesis also reduces the patient recall rate to perform additional views or subsequent ultrasound. In one large study, the recall rate was 12% for standard digital mammography and 8.4% for tomosynthesis.7
The limitations of tomosynthesis include higher costs and higher radiation doses.
If the technology is available, I recommend that women have their mammograms using the best technology, tomosynthesis digital mammography.8
References
1. Smith RA, Duffy SW, Gabe R, Tabar L, Yen AM, Chen TH. The randomized trials of breast cancer screening: what have we learned? Radiol Clin North Am. 2004;42(5):793–806.
2. Independent UK Panel on Breast Cancer Screening. The benefits and harms of breast cancer screening: an independent review. Lancet. 2012;380(9855):1778–1786.
3. US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendations statement. Ann Intern Med. 2009;151(10):716–726, W-236.
4. Welch HG, Passow HJ. Quantifying the benefits and harms of screening mammograms. JAMA Intern Med. 2014;174(3):448–454.
5. Destounis SV, Morgan R, Areino A. Screening for dense breasts: digital tomosynthesis. AJR Am J Roentgenol. 2015;204(2):261–264.
6. Friedewald SM, Rafferty EA, Rose SL, et al. Breast cancer screening using tomosynthesis in combination with digital mammography. JAMA. 2014;311(24):2499–2507.
7. Haas BM, Kalra V, Geisel J, Raghu M, Durand M, Philpotts LE. Comparison of tomosynthesis plus digital mammography and digital mammography alone for breast cancer screening. Radiology. 2013;269(3):694–700.
8. Pisano ED, Yaffe MJ. Breast cancer screening: should tomosynthesis replace digital mammography? JAMA. 2014;311(24):2488–2489.
A pragmatic, low-resource utilization screening approach for women with dense breasts
There are no published randomized clinical trials that provide high-quality evidence on what to do if dense breasts are identified on mammography.3 Authors of observational studies have evaluated the potential role of supplemental imaging, including ultrasound and magnetic resonance imaging (MRI), in the management of dense breast tissue (see “Supplemental breast cancer screening modalities” below). Supplemental imaging involves complex trade-offs, balancing the potential benefit of identifying occult early breast cancer lesions not identified by mammography with the risk of subjecting many women without cancer to additional testing and unnecessary biopsies.
A pragmatic, low-resource utilization plan for women with dense breasts involves emphasizing that mammography is the best available screening tool and that annual or biennial mammography is the foundation of all current approaches to breast cancer screening. Supplemental imaging is unnecessary with this approach because there is no evidence that it reduces breast cancer mortality. There is, however, substantial evidence that using supplemental imaging for all women with dense breasts will result in little benefit and great costs, including many unnecessary biopsies.1,4 Women with dense breasts also could consider annual clinical breast examination.
Supplemental breast cancer screening modalities
Ultrasound and magnetic resonance imaging (MRI) are available as supplemental imaging, although ultrasound is the only supplemental imaging test that is specifically approved for women with dense breasts. Among the clinically available imaging modalities, MRI can detect the greatest number of cancers.
Ultrasound
In women with dense breasts, ultrasound can detect another 3 to 4 cancers that were not detected by mammography. However, ultrasound imaging generates many false positive results that lead to additional biopsies. According to one analysis, compared with mammography alone, mammography plus ultrasound would prevent 0.36 breast cancer deaths and cause 354 additional biopsies per 1,000 women with dense breasts screened biennially for 25 years.1
Ultrasound commonly is used to follow up an abnormal mammogram to further evaluate masses and differentiate cysts from solid tumors. Ultrasound is also a useful breast-imaging tool for women who are pregnant. In 2012, the US Food and Drug Administration approved an automated breast ultrasound device to be used for supplemental imaging of asymptomatic women with dense breasts and a mammogram negative for cancer. This device may facilitate the use of ultrasound for supplemental imaging of women with dense breasts on mammography.
Magnetic resonance imaging
MRI can detect the greatest number of cancers of any clinically available modality.
It is almost never covered by insurance for women whose only breast cancer risk factor is the identification of dense breasts on mammography. The cost of MRI testing is, however, typically covered for women at very high risk for breast cancer.
Women who are known to be at very high risk for breast cancer should begin annual clinical breast examinations at age 25 years and alternate between screening mammography and screening MRI every 6 months or annually. These women include:
- carriers of clinically significant BRCA1 or BRCA2 mutations
- carriers of other high-risk genetic mutations such as Cowden syndrome (PTEN mutation), Lai-Fraumeni syndrome (TP53 mutation), and Peutz-Jeghers syndrome
- genetically untested women with a first-degree relative with a BRCA mutation.
Women who had thoracic radiation before age 30 also should be considered for this screening protocol beginning 8 to 10 years after the radiation exposure or at age 25 years.2
References
1. Sprague BL, Stout KN, Schechter MD, et al. Benefits, harms and cost-effectiveness of supplemental ultrasonography screening for women with dense breasts. Ann Intern Med. 2015;162(3):157–166.
2. CRICO Breast Care Management Algorithm. CRICO; Cambridge, Massachusetts; 2014. https://www.rmf.harvard.edu/~/media/Files/_Global/KC/PDFs/Guidelines/cricormfbca2014_locked.pdf. Accessed July 19, 2015.
A high-resource utilization screening approach
There are no randomized trials to help guide recommendations about how to respond to a finding of dense breasts on mammography. In addition to breast density, many factors influence breast cancer risk, including a patient’s:
- age
- family history
- history of previous breast biopsies
- many reproductive factors, including early age of menarche and late childbearing.
Women with both dense breasts and an increased risk of breast cancer may reap the greatest benefit from supplemental imaging, such as ultrasonography. Therefore, a two-step approach can help.
Step 1: Assess breast cancer risk. This can be accomplished using one of many calculators. Three that are commonly used are the:
- National Cancer Institute (NCI) Breast Cancer Surveillance Consortium (BCSC) calculator5
- NCI Breast Cancer Risk Assessment Tool, Gail model (BRCAT)6
- IBIS Breast Cancer Risk Evaluation Tool (Tyrer-Cuzick model).7
The BCSC calculator uses age, race/ethnicity, first-degree relatives with breast cancer, a history of a breast biopsy, and breast density to calculate a 5-year risk of developing breast cancer.
The BCRAT tool uses current age, race/ethnicity, age at menarche, age at first live-birth of a child, number of first-degree relatives with breast cancer, a history of breast biopsies, and the identification of atypical hyperplasia to calculate a 5-year risk of breast cancer.
The IBIS model uses many more variables, including a detailed family history to calculate a 10-year and lifetime risk of breast cancer. If a patient has ductal carcinoma in situ, lobular carcinoma in situ, chest irradiation before age 30 years, or known BRCA1 or BRCA2 mutations, she is instructed not to use the risk calculators because they are at very high risk for breast cancer, and they need an individualized intensive plan for monitoring and prevention (see MRI section in “Supplemental breast cancer screening modalities” above).
Step 2: Use breast density and breast cancer risk to develop a screening plan. The NIH Breast Cancer Surveillance Consortium has published data estimating the risk that a woman with a mammogram negative for cancer will develop breast cancer within the next 12 months (based on her age, breast density, and breast cancer risk—calculated with the BCSC tool).8
It reported an increased risk of breast cancer diagnosed within 12 months following a mammogram that was negative for cancer in women with extremely dense breasts and a BCSC 5-year risk of breast cancer of 1.67% or greater and in women with heterogeneously dense breasts and a BCSC 5-year risk of breast cancer of 2.5% or greater.8
Using these cutoffs it is estimated that 24% of all women with heterogeneously or extremely dense breasts would be offered supplemental screening with a modality such as ultrasound, and 76% would be guided not to have supplemental screening because their risk of developing breast cancer in the 12 months following their negative mammogram is low.
If this guidance is followed, it would require 694 supplemental ultrasound studies and many biopsies to detect 1 additional breast cancer, significantly increasing overall health care costs.8 In many states insurers do not cover supplemental ultrasound imaging of the breasts. In most states insurers require preauthorization for supplemental MRI of the breasts. You need to know the insurance practices in the state to help guide decision making about supplemental imaging. The approach described above is consistent with the American College of Obstetricians and Gynecologists recommendation that women with dense breasts, who are asymptomaticand have no additional risk factors for breast cancer, do not need to be offered supplemental imaging.9
Case: Next steps
The BCSC calculator reveals that the 51-year-old woman with a family history of breast cancer and a mammogram showing extremely dense breasts has a 5-year risk of breast cancer of 2.68%. Given that this risk is elevated, this patient could be offered supplemental ultrasound screening and annual breast clinical examination. In addition, she could be further counseled about breast cancer chemoprevention options.10
Women with a strong family history of breast and/or ovarian cancer also could be referred for genetic counseling and BRCA testing.11 The risk of having a BRCA mutation can be calculated using the BRCAPRO tool.12
Most women with dense breast tissue on mammography will never develop breast cancer. Yet the presence of dense breast tissue both increases the risk of breast cancer and decreases the sensitivity of mammography to detect cancer. There are no high-quality data from randomized trials to help guide our recommendations concerning the management of dense breasts identified on mammography. Yet many states have laws that suggest patients ask you to provide advice about breast density.
Patients, clinicians, and health systems vary in their confidence in the clinical value of breast cancer screening programs. Consequently, there is no “right answer” to this vexing problem. The standard of care is to support a range of options tailored to the specific clinical characteristics and needs of each patient.
Many states mandate that patients receive letters from their mammography center that report on breast density. In many states the law requires that the letter contain a statement that dense breasts increase the risk of breast cancer and reduce the ability of mammography to detect breast cancer. Do you believe these letters:
b) are beneficial because they provide the patient important information
c) both a and b
To weigh in and send your Letter to the Editor, visit obgmanagement.com and look for the “Quick Poll” on the right side of the home page.
1. Sprague BL, Stout KN, Schechter MD, et al. Benefits, harms and cost-effectiveness of supplemental ultrasonography screening for women with dense breasts. Ann Intern Med. 2015;162(3):157–166.
2. Carney PA, Miglioretti DL, Yankaskas BC, et al. Individual and combined effects of age, breast density and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med. 2003;138(3):168–175.
3. Gartlehner G, Thaler K, Chapman A, et al. Mammography in combination with breast ultrasonography versus mammography for breast cancer screening in women at average risk. Cochrane Database Syst Rev. 2013;4:CD009632.
4. Berg WA, Blume JD, Cormack JB, et al. Combined screening with ultrasound and mammography vs. mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299(18):2151–2163.
5. Breast Cancer Surveillance Consortium risk calculator. BCSC Web site. https://tools.bcsc-scc.org/BC5yearRisk/intro.htm. Updated February 13, 2015. Accessed July 17, 2015.
6. NCI Breast Cancer Risk Assessment Tool (Gail model). National Cancer Institute Web site. http://www.cancer.gov/BCRISKTOOL/. Accessed July 17, 2015.
7. IBIS Breast Cancer Risk Evaluation Tool. http://www.ems-trials.org/riskevaluator/. Updated January 9, 2015. Accessed July 17, 2015.
8. Kerlikowske K, Zhu W, Tosteson AN, et al; Breast Cancer Surveillance Consortium. Identifying women with dense breasts at high risk for interval cancer. Ann Intern Med. 2015;162(10):673–681.
9. Committee on Gynecologic Practice. Committee Opinion No. 625: Management of women with dense breasts diagnosed by mammography. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2015;125(3): 750–751.
10. Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013;31(34):2942–2962.
11. Profato JL, Arun BK. Genetic risk assessment for breast and gynecological malignancies. Curr Opin Obstet Gynecol. 2015;27(1):1–5.
12. BRCAPRO. BayesMendel Lab. Harvard University Web site. http://bcb.dfci.harvard.edu/bayesmendel/brcapro.php. Accessed July 19, 2015.
Case: Patient seeks clarification and next steps on her breast density classification
Your patient, a 51-year-old postmenopausal woman (G0P0) in good health, had an annual screening mammogram that showed no evidence of malignancy. She is white and has a mother with a history of breast cancer. She has never had a breast biopsy. Following the mammogram, she received a letter from the imaging center, stating:
She calls your office and asks, “What should I do next?”
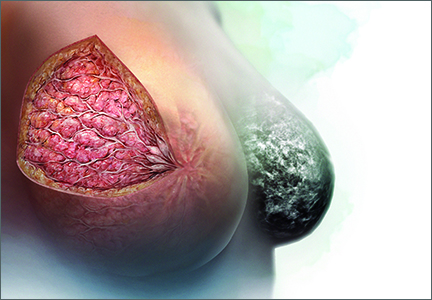
Breasts are composed of fibrous, glandular, and adipose tissue. If the breasts contain a lot of fibrous and glandular tissue, and little adipose tissue, they are considered to be “dense.” Using mammography, the current standard is to report the density of breast tissue using 4 categories:
- almost entirely fatty
- scattered fibroglandular densities
- heterogeneously dense
- extremely dense.
Dense breast tissue is defined to include the 2 categories heterogeneously dense and extremely dense.
Observational studies have reported that dense breast tissue is associated with an increased risk of breast cancer, and dense breast tissue makes it more difficult to detect breast cancer on mammography. According to data from the Breast Cancer Surveillance Consortium, among women aged 50 or older, the relative risk of breast cancer stratified by the 4 categories of breast density is 0.59, 1.00, 1.46, and 1.77, for almost entirely fatty, scattered fibroglandular densities, heterogeneously dense, and extremely dense, respectively.1 In one study, the sensitivity of mammography to detect breast cancer was 82% to 88% for women with nondense breasts and 62% to 69% in women with dense breasts.2 These data have catalyzed investigators to explore the use of supplemental imaging to enhance cancer detection in women with dense breasts.
The link between breast density and breast cancer risk and reduced sensitivity of mammography also has catalyzed activists and legislators to champion breast density notification laws, which have passed in more than 20 states. These laws require facilities that perform mammography to notify women with dense breasts that this finding is associated with an increased risk of breast cancer and that dense breasts reduce the ability of mammography to detect cancer. In some states, the law mandates that women with dense breasts be offered supplemental ultrasound imaging and that insurers must cover the cost of the ultrasound studies. Many of the laws recommend that the patient discuss the situation with the clinician who ordered the mammogram.
When I first saw the recommendation for patients to contact me about how to manage dense breasts, my initial response was, “Who? Me?” I felt ill equipped to provide any useful advice and suspected that many of my patients knew more than I about this issue.
Based on a review of the evidence, my current clinical recommendation is outlined in the 2 options below, including a low-resource utilization option and a high-resource utilization option. For patients, physicians, and health systems that are concerned that excessive breast cancer screening tests might cause more harm than benefit, the identification of dense breasts on mammogram is unlikely to be a trigger to perform any additional testing. In this situation, the pragmatic low-resource option is most relevant.
Alternatively, for patients and physicians who strongly believe in the value of screening mammography (see “Utilize tomosynthesis digital mammography technology for your patients” below), a reasonable strategy is to recommend that women with dense breasts and an increased risk for breast cancer be offered supplemental imaging.
In this editorial I elaborate these 2 approaches to breast cancer screening in women with dense breasts.
Utilize tomosynthesis digital mammography technology for your patients
Mammograms are the primary modality used for breast cancer screening because screening mammography has been shown to reduce breast cancer deaths by 15% to 30%.1,2 Annual or biennial mammograms are recommended for women aged 40 years or older by many professional organizations, including the American College of Obstetricians and Gynecologists and the American College of Radiology. However, mammography screening programs have been criticized because of false-positive tests resulting in unnecessary biopsies, limited sensitivity, and the theoretical risk of over-diagnosing clinically insignificant cancers.3,4
Mammography technology continues to evolve. Film-based mammography has been replaced by digital mammography. Tomosynthesis digital mammography, also known as 3-D mammography, is now replacing standard digital mammography.5
With tomosynthesis, digital mammography image acquisition is performed using an x-ray source that moves through an arc across the breast with the capture of a series of images from different angles and reconstruction of the data into thin slices approximately 1 mm in width. The presentation of breast images in thin slices permits superior detection of lesions. In addition, the collected images can be reconstructed to present a virtual 2-D image for analysis.
Tomosynthesis has been demonstrated to increase the sensitivity of mammography to detect cancer and reduce false-positive examinations. In a study of 454,850 mammography examinations, investigators found that the invasive cancer detection rate per 1,000 studies increased from 2.9 with standard digital mammography to 4.1 with tomosynthesis.6
Tomosynthesis also reduces the patient recall rate to perform additional views or subsequent ultrasound. In one large study, the recall rate was 12% for standard digital mammography and 8.4% for tomosynthesis.7
The limitations of tomosynthesis include higher costs and higher radiation doses.
If the technology is available, I recommend that women have their mammograms using the best technology, tomosynthesis digital mammography.8
References
1. Smith RA, Duffy SW, Gabe R, Tabar L, Yen AM, Chen TH. The randomized trials of breast cancer screening: what have we learned? Radiol Clin North Am. 2004;42(5):793–806.
2. Independent UK Panel on Breast Cancer Screening. The benefits and harms of breast cancer screening: an independent review. Lancet. 2012;380(9855):1778–1786.
3. US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendations statement. Ann Intern Med. 2009;151(10):716–726, W-236.
4. Welch HG, Passow HJ. Quantifying the benefits and harms of screening mammograms. JAMA Intern Med. 2014;174(3):448–454.
5. Destounis SV, Morgan R, Areino A. Screening for dense breasts: digital tomosynthesis. AJR Am J Roentgenol. 2015;204(2):261–264.
6. Friedewald SM, Rafferty EA, Rose SL, et al. Breast cancer screening using tomosynthesis in combination with digital mammography. JAMA. 2014;311(24):2499–2507.
7. Haas BM, Kalra V, Geisel J, Raghu M, Durand M, Philpotts LE. Comparison of tomosynthesis plus digital mammography and digital mammography alone for breast cancer screening. Radiology. 2013;269(3):694–700.
8. Pisano ED, Yaffe MJ. Breast cancer screening: should tomosynthesis replace digital mammography? JAMA. 2014;311(24):2488–2489.
A pragmatic, low-resource utilization screening approach for women with dense breasts
There are no published randomized clinical trials that provide high-quality evidence on what to do if dense breasts are identified on mammography.3 Authors of observational studies have evaluated the potential role of supplemental imaging, including ultrasound and magnetic resonance imaging (MRI), in the management of dense breast tissue (see “Supplemental breast cancer screening modalities” below). Supplemental imaging involves complex trade-offs, balancing the potential benefit of identifying occult early breast cancer lesions not identified by mammography with the risk of subjecting many women without cancer to additional testing and unnecessary biopsies.
A pragmatic, low-resource utilization plan for women with dense breasts involves emphasizing that mammography is the best available screening tool and that annual or biennial mammography is the foundation of all current approaches to breast cancer screening. Supplemental imaging is unnecessary with this approach because there is no evidence that it reduces breast cancer mortality. There is, however, substantial evidence that using supplemental imaging for all women with dense breasts will result in little benefit and great costs, including many unnecessary biopsies.1,4 Women with dense breasts also could consider annual clinical breast examination.
Supplemental breast cancer screening modalities
Ultrasound and magnetic resonance imaging (MRI) are available as supplemental imaging, although ultrasound is the only supplemental imaging test that is specifically approved for women with dense breasts. Among the clinically available imaging modalities, MRI can detect the greatest number of cancers.
Ultrasound
In women with dense breasts, ultrasound can detect another 3 to 4 cancers that were not detected by mammography. However, ultrasound imaging generates many false positive results that lead to additional biopsies. According to one analysis, compared with mammography alone, mammography plus ultrasound would prevent 0.36 breast cancer deaths and cause 354 additional biopsies per 1,000 women with dense breasts screened biennially for 25 years.1
Ultrasound commonly is used to follow up an abnormal mammogram to further evaluate masses and differentiate cysts from solid tumors. Ultrasound is also a useful breast-imaging tool for women who are pregnant. In 2012, the US Food and Drug Administration approved an automated breast ultrasound device to be used for supplemental imaging of asymptomatic women with dense breasts and a mammogram negative for cancer. This device may facilitate the use of ultrasound for supplemental imaging of women with dense breasts on mammography.
Magnetic resonance imaging
MRI can detect the greatest number of cancers of any clinically available modality.
It is almost never covered by insurance for women whose only breast cancer risk factor is the identification of dense breasts on mammography. The cost of MRI testing is, however, typically covered for women at very high risk for breast cancer.
Women who are known to be at very high risk for breast cancer should begin annual clinical breast examinations at age 25 years and alternate between screening mammography and screening MRI every 6 months or annually. These women include:
- carriers of clinically significant BRCA1 or BRCA2 mutations
- carriers of other high-risk genetic mutations such as Cowden syndrome (PTEN mutation), Lai-Fraumeni syndrome (TP53 mutation), and Peutz-Jeghers syndrome
- genetically untested women with a first-degree relative with a BRCA mutation.
Women who had thoracic radiation before age 30 also should be considered for this screening protocol beginning 8 to 10 years after the radiation exposure or at age 25 years.2
References
1. Sprague BL, Stout KN, Schechter MD, et al. Benefits, harms and cost-effectiveness of supplemental ultrasonography screening for women with dense breasts. Ann Intern Med. 2015;162(3):157–166.
2. CRICO Breast Care Management Algorithm. CRICO; Cambridge, Massachusetts; 2014. https://www.rmf.harvard.edu/~/media/Files/_Global/KC/PDFs/Guidelines/cricormfbca2014_locked.pdf. Accessed July 19, 2015.
A high-resource utilization screening approach
There are no randomized trials to help guide recommendations about how to respond to a finding of dense breasts on mammography. In addition to breast density, many factors influence breast cancer risk, including a patient’s:
- age
- family history
- history of previous breast biopsies
- many reproductive factors, including early age of menarche and late childbearing.
Women with both dense breasts and an increased risk of breast cancer may reap the greatest benefit from supplemental imaging, such as ultrasonography. Therefore, a two-step approach can help.
Step 1: Assess breast cancer risk. This can be accomplished using one of many calculators. Three that are commonly used are the:
- National Cancer Institute (NCI) Breast Cancer Surveillance Consortium (BCSC) calculator5
- NCI Breast Cancer Risk Assessment Tool, Gail model (BRCAT)6
- IBIS Breast Cancer Risk Evaluation Tool (Tyrer-Cuzick model).7
The BCSC calculator uses age, race/ethnicity, first-degree relatives with breast cancer, a history of a breast biopsy, and breast density to calculate a 5-year risk of developing breast cancer.
The BCRAT tool uses current age, race/ethnicity, age at menarche, age at first live-birth of a child, number of first-degree relatives with breast cancer, a history of breast biopsies, and the identification of atypical hyperplasia to calculate a 5-year risk of breast cancer.
The IBIS model uses many more variables, including a detailed family history to calculate a 10-year and lifetime risk of breast cancer. If a patient has ductal carcinoma in situ, lobular carcinoma in situ, chest irradiation before age 30 years, or known BRCA1 or BRCA2 mutations, she is instructed not to use the risk calculators because they are at very high risk for breast cancer, and they need an individualized intensive plan for monitoring and prevention (see MRI section in “Supplemental breast cancer screening modalities” above).
Step 2: Use breast density and breast cancer risk to develop a screening plan. The NIH Breast Cancer Surveillance Consortium has published data estimating the risk that a woman with a mammogram negative for cancer will develop breast cancer within the next 12 months (based on her age, breast density, and breast cancer risk—calculated with the BCSC tool).8
It reported an increased risk of breast cancer diagnosed within 12 months following a mammogram that was negative for cancer in women with extremely dense breasts and a BCSC 5-year risk of breast cancer of 1.67% or greater and in women with heterogeneously dense breasts and a BCSC 5-year risk of breast cancer of 2.5% or greater.8
Using these cutoffs it is estimated that 24% of all women with heterogeneously or extremely dense breasts would be offered supplemental screening with a modality such as ultrasound, and 76% would be guided not to have supplemental screening because their risk of developing breast cancer in the 12 months following their negative mammogram is low.
If this guidance is followed, it would require 694 supplemental ultrasound studies and many biopsies to detect 1 additional breast cancer, significantly increasing overall health care costs.8 In many states insurers do not cover supplemental ultrasound imaging of the breasts. In most states insurers require preauthorization for supplemental MRI of the breasts. You need to know the insurance practices in the state to help guide decision making about supplemental imaging. The approach described above is consistent with the American College of Obstetricians and Gynecologists recommendation that women with dense breasts, who are asymptomaticand have no additional risk factors for breast cancer, do not need to be offered supplemental imaging.9
Case: Next steps
The BCSC calculator reveals that the 51-year-old woman with a family history of breast cancer and a mammogram showing extremely dense breasts has a 5-year risk of breast cancer of 2.68%. Given that this risk is elevated, this patient could be offered supplemental ultrasound screening and annual breast clinical examination. In addition, she could be further counseled about breast cancer chemoprevention options.10
Women with a strong family history of breast and/or ovarian cancer also could be referred for genetic counseling and BRCA testing.11 The risk of having a BRCA mutation can be calculated using the BRCAPRO tool.12
Most women with dense breast tissue on mammography will never develop breast cancer. Yet the presence of dense breast tissue both increases the risk of breast cancer and decreases the sensitivity of mammography to detect cancer. There are no high-quality data from randomized trials to help guide our recommendations concerning the management of dense breasts identified on mammography. Yet many states have laws that suggest patients ask you to provide advice about breast density.
Patients, clinicians, and health systems vary in their confidence in the clinical value of breast cancer screening programs. Consequently, there is no “right answer” to this vexing problem. The standard of care is to support a range of options tailored to the specific clinical characteristics and needs of each patient.
Many states mandate that patients receive letters from their mammography center that report on breast density. In many states the law requires that the letter contain a statement that dense breasts increase the risk of breast cancer and reduce the ability of mammography to detect breast cancer. Do you believe these letters:
b) are beneficial because they provide the patient important information
c) both a and b
To weigh in and send your Letter to the Editor, visit obgmanagement.com and look for the “Quick Poll” on the right side of the home page.
Case: Patient seeks clarification and next steps on her breast density classification
Your patient, a 51-year-old postmenopausal woman (G0P0) in good health, had an annual screening mammogram that showed no evidence of malignancy. She is white and has a mother with a history of breast cancer. She has never had a breast biopsy. Following the mammogram, she received a letter from the imaging center, stating:
She calls your office and asks, “What should I do next?”
Breasts are composed of fibrous, glandular, and adipose tissue. If the breasts contain a lot of fibrous and glandular tissue, and little adipose tissue, they are considered to be “dense.” Using mammography, the current standard is to report the density of breast tissue using 4 categories:
- almost entirely fatty
- scattered fibroglandular densities
- heterogeneously dense
- extremely dense.
Dense breast tissue is defined to include the 2 categories heterogeneously dense and extremely dense.
Observational studies have reported that dense breast tissue is associated with an increased risk of breast cancer, and dense breast tissue makes it more difficult to detect breast cancer on mammography. According to data from the Breast Cancer Surveillance Consortium, among women aged 50 or older, the relative risk of breast cancer stratified by the 4 categories of breast density is 0.59, 1.00, 1.46, and 1.77, for almost entirely fatty, scattered fibroglandular densities, heterogeneously dense, and extremely dense, respectively.1 In one study, the sensitivity of mammography to detect breast cancer was 82% to 88% for women with nondense breasts and 62% to 69% in women with dense breasts.2 These data have catalyzed investigators to explore the use of supplemental imaging to enhance cancer detection in women with dense breasts.
The link between breast density and breast cancer risk and reduced sensitivity of mammography also has catalyzed activists and legislators to champion breast density notification laws, which have passed in more than 20 states. These laws require facilities that perform mammography to notify women with dense breasts that this finding is associated with an increased risk of breast cancer and that dense breasts reduce the ability of mammography to detect cancer. In some states, the law mandates that women with dense breasts be offered supplemental ultrasound imaging and that insurers must cover the cost of the ultrasound studies. Many of the laws recommend that the patient discuss the situation with the clinician who ordered the mammogram.
When I first saw the recommendation for patients to contact me about how to manage dense breasts, my initial response was, “Who? Me?” I felt ill equipped to provide any useful advice and suspected that many of my patients knew more than I about this issue.
Based on a review of the evidence, my current clinical recommendation is outlined in the 2 options below, including a low-resource utilization option and a high-resource utilization option. For patients, physicians, and health systems that are concerned that excessive breast cancer screening tests might cause more harm than benefit, the identification of dense breasts on mammogram is unlikely to be a trigger to perform any additional testing. In this situation, the pragmatic low-resource option is most relevant.
Alternatively, for patients and physicians who strongly believe in the value of screening mammography (see “Utilize tomosynthesis digital mammography technology for your patients” below), a reasonable strategy is to recommend that women with dense breasts and an increased risk for breast cancer be offered supplemental imaging.
In this editorial I elaborate these 2 approaches to breast cancer screening in women with dense breasts.
Utilize tomosynthesis digital mammography technology for your patients
Mammograms are the primary modality used for breast cancer screening because screening mammography has been shown to reduce breast cancer deaths by 15% to 30%.1,2 Annual or biennial mammograms are recommended for women aged 40 years or older by many professional organizations, including the American College of Obstetricians and Gynecologists and the American College of Radiology. However, mammography screening programs have been criticized because of false-positive tests resulting in unnecessary biopsies, limited sensitivity, and the theoretical risk of over-diagnosing clinically insignificant cancers.3,4
Mammography technology continues to evolve. Film-based mammography has been replaced by digital mammography. Tomosynthesis digital mammography, also known as 3-D mammography, is now replacing standard digital mammography.5
With tomosynthesis, digital mammography image acquisition is performed using an x-ray source that moves through an arc across the breast with the capture of a series of images from different angles and reconstruction of the data into thin slices approximately 1 mm in width. The presentation of breast images in thin slices permits superior detection of lesions. In addition, the collected images can be reconstructed to present a virtual 2-D image for analysis.
Tomosynthesis has been demonstrated to increase the sensitivity of mammography to detect cancer and reduce false-positive examinations. In a study of 454,850 mammography examinations, investigators found that the invasive cancer detection rate per 1,000 studies increased from 2.9 with standard digital mammography to 4.1 with tomosynthesis.6
Tomosynthesis also reduces the patient recall rate to perform additional views or subsequent ultrasound. In one large study, the recall rate was 12% for standard digital mammography and 8.4% for tomosynthesis.7
The limitations of tomosynthesis include higher costs and higher radiation doses.
If the technology is available, I recommend that women have their mammograms using the best technology, tomosynthesis digital mammography.8
References
1. Smith RA, Duffy SW, Gabe R, Tabar L, Yen AM, Chen TH. The randomized trials of breast cancer screening: what have we learned? Radiol Clin North Am. 2004;42(5):793–806.
2. Independent UK Panel on Breast Cancer Screening. The benefits and harms of breast cancer screening: an independent review. Lancet. 2012;380(9855):1778–1786.
3. US Preventive Services Task Force. Screening for breast cancer: US Preventive Services Task Force recommendations statement. Ann Intern Med. 2009;151(10):716–726, W-236.
4. Welch HG, Passow HJ. Quantifying the benefits and harms of screening mammograms. JAMA Intern Med. 2014;174(3):448–454.
5. Destounis SV, Morgan R, Areino A. Screening for dense breasts: digital tomosynthesis. AJR Am J Roentgenol. 2015;204(2):261–264.
6. Friedewald SM, Rafferty EA, Rose SL, et al. Breast cancer screening using tomosynthesis in combination with digital mammography. JAMA. 2014;311(24):2499–2507.
7. Haas BM, Kalra V, Geisel J, Raghu M, Durand M, Philpotts LE. Comparison of tomosynthesis plus digital mammography and digital mammography alone for breast cancer screening. Radiology. 2013;269(3):694–700.
8. Pisano ED, Yaffe MJ. Breast cancer screening: should tomosynthesis replace digital mammography? JAMA. 2014;311(24):2488–2489.
A pragmatic, low-resource utilization screening approach for women with dense breasts
There are no published randomized clinical trials that provide high-quality evidence on what to do if dense breasts are identified on mammography.3 Authors of observational studies have evaluated the potential role of supplemental imaging, including ultrasound and magnetic resonance imaging (MRI), in the management of dense breast tissue (see “Supplemental breast cancer screening modalities” below). Supplemental imaging involves complex trade-offs, balancing the potential benefit of identifying occult early breast cancer lesions not identified by mammography with the risk of subjecting many women without cancer to additional testing and unnecessary biopsies.
A pragmatic, low-resource utilization plan for women with dense breasts involves emphasizing that mammography is the best available screening tool and that annual or biennial mammography is the foundation of all current approaches to breast cancer screening. Supplemental imaging is unnecessary with this approach because there is no evidence that it reduces breast cancer mortality. There is, however, substantial evidence that using supplemental imaging for all women with dense breasts will result in little benefit and great costs, including many unnecessary biopsies.1,4 Women with dense breasts also could consider annual clinical breast examination.
Supplemental breast cancer screening modalities
Ultrasound and magnetic resonance imaging (MRI) are available as supplemental imaging, although ultrasound is the only supplemental imaging test that is specifically approved for women with dense breasts. Among the clinically available imaging modalities, MRI can detect the greatest number of cancers.
Ultrasound
In women with dense breasts, ultrasound can detect another 3 to 4 cancers that were not detected by mammography. However, ultrasound imaging generates many false positive results that lead to additional biopsies. According to one analysis, compared with mammography alone, mammography plus ultrasound would prevent 0.36 breast cancer deaths and cause 354 additional biopsies per 1,000 women with dense breasts screened biennially for 25 years.1
Ultrasound commonly is used to follow up an abnormal mammogram to further evaluate masses and differentiate cysts from solid tumors. Ultrasound is also a useful breast-imaging tool for women who are pregnant. In 2012, the US Food and Drug Administration approved an automated breast ultrasound device to be used for supplemental imaging of asymptomatic women with dense breasts and a mammogram negative for cancer. This device may facilitate the use of ultrasound for supplemental imaging of women with dense breasts on mammography.
Magnetic resonance imaging
MRI can detect the greatest number of cancers of any clinically available modality.
It is almost never covered by insurance for women whose only breast cancer risk factor is the identification of dense breasts on mammography. The cost of MRI testing is, however, typically covered for women at very high risk for breast cancer.
Women who are known to be at very high risk for breast cancer should begin annual clinical breast examinations at age 25 years and alternate between screening mammography and screening MRI every 6 months or annually. These women include:
- carriers of clinically significant BRCA1 or BRCA2 mutations
- carriers of other high-risk genetic mutations such as Cowden syndrome (PTEN mutation), Lai-Fraumeni syndrome (TP53 mutation), and Peutz-Jeghers syndrome
- genetically untested women with a first-degree relative with a BRCA mutation.
Women who had thoracic radiation before age 30 also should be considered for this screening protocol beginning 8 to 10 years after the radiation exposure or at age 25 years.2
References
1. Sprague BL, Stout KN, Schechter MD, et al. Benefits, harms and cost-effectiveness of supplemental ultrasonography screening for women with dense breasts. Ann Intern Med. 2015;162(3):157–166.
2. CRICO Breast Care Management Algorithm. CRICO; Cambridge, Massachusetts; 2014. https://www.rmf.harvard.edu/~/media/Files/_Global/KC/PDFs/Guidelines/cricormfbca2014_locked.pdf. Accessed July 19, 2015.
A high-resource utilization screening approach
There are no randomized trials to help guide recommendations about how to respond to a finding of dense breasts on mammography. In addition to breast density, many factors influence breast cancer risk, including a patient’s:
- age
- family history
- history of previous breast biopsies
- many reproductive factors, including early age of menarche and late childbearing.
Women with both dense breasts and an increased risk of breast cancer may reap the greatest benefit from supplemental imaging, such as ultrasonography. Therefore, a two-step approach can help.
Step 1: Assess breast cancer risk. This can be accomplished using one of many calculators. Three that are commonly used are the:
- National Cancer Institute (NCI) Breast Cancer Surveillance Consortium (BCSC) calculator5
- NCI Breast Cancer Risk Assessment Tool, Gail model (BRCAT)6
- IBIS Breast Cancer Risk Evaluation Tool (Tyrer-Cuzick model).7
The BCSC calculator uses age, race/ethnicity, first-degree relatives with breast cancer, a history of a breast biopsy, and breast density to calculate a 5-year risk of developing breast cancer.
The BCRAT tool uses current age, race/ethnicity, age at menarche, age at first live-birth of a child, number of first-degree relatives with breast cancer, a history of breast biopsies, and the identification of atypical hyperplasia to calculate a 5-year risk of breast cancer.
The IBIS model uses many more variables, including a detailed family history to calculate a 10-year and lifetime risk of breast cancer. If a patient has ductal carcinoma in situ, lobular carcinoma in situ, chest irradiation before age 30 years, or known BRCA1 or BRCA2 mutations, she is instructed not to use the risk calculators because they are at very high risk for breast cancer, and they need an individualized intensive plan for monitoring and prevention (see MRI section in “Supplemental breast cancer screening modalities” above).
Step 2: Use breast density and breast cancer risk to develop a screening plan. The NIH Breast Cancer Surveillance Consortium has published data estimating the risk that a woman with a mammogram negative for cancer will develop breast cancer within the next 12 months (based on her age, breast density, and breast cancer risk—calculated with the BCSC tool).8
It reported an increased risk of breast cancer diagnosed within 12 months following a mammogram that was negative for cancer in women with extremely dense breasts and a BCSC 5-year risk of breast cancer of 1.67% or greater and in women with heterogeneously dense breasts and a BCSC 5-year risk of breast cancer of 2.5% or greater.8
Using these cutoffs it is estimated that 24% of all women with heterogeneously or extremely dense breasts would be offered supplemental screening with a modality such as ultrasound, and 76% would be guided not to have supplemental screening because their risk of developing breast cancer in the 12 months following their negative mammogram is low.
If this guidance is followed, it would require 694 supplemental ultrasound studies and many biopsies to detect 1 additional breast cancer, significantly increasing overall health care costs.8 In many states insurers do not cover supplemental ultrasound imaging of the breasts. In most states insurers require preauthorization for supplemental MRI of the breasts. You need to know the insurance practices in the state to help guide decision making about supplemental imaging. The approach described above is consistent with the American College of Obstetricians and Gynecologists recommendation that women with dense breasts, who are asymptomaticand have no additional risk factors for breast cancer, do not need to be offered supplemental imaging.9
Case: Next steps
The BCSC calculator reveals that the 51-year-old woman with a family history of breast cancer and a mammogram showing extremely dense breasts has a 5-year risk of breast cancer of 2.68%. Given that this risk is elevated, this patient could be offered supplemental ultrasound screening and annual breast clinical examination. In addition, she could be further counseled about breast cancer chemoprevention options.10
Women with a strong family history of breast and/or ovarian cancer also could be referred for genetic counseling and BRCA testing.11 The risk of having a BRCA mutation can be calculated using the BRCAPRO tool.12
Most women with dense breast tissue on mammography will never develop breast cancer. Yet the presence of dense breast tissue both increases the risk of breast cancer and decreases the sensitivity of mammography to detect cancer. There are no high-quality data from randomized trials to help guide our recommendations concerning the management of dense breasts identified on mammography. Yet many states have laws that suggest patients ask you to provide advice about breast density.
Patients, clinicians, and health systems vary in their confidence in the clinical value of breast cancer screening programs. Consequently, there is no “right answer” to this vexing problem. The standard of care is to support a range of options tailored to the specific clinical characteristics and needs of each patient.
Many states mandate that patients receive letters from their mammography center that report on breast density. In many states the law requires that the letter contain a statement that dense breasts increase the risk of breast cancer and reduce the ability of mammography to detect breast cancer. Do you believe these letters:
b) are beneficial because they provide the patient important information
c) both a and b
To weigh in and send your Letter to the Editor, visit obgmanagement.com and look for the “Quick Poll” on the right side of the home page.
1. Sprague BL, Stout KN, Schechter MD, et al. Benefits, harms and cost-effectiveness of supplemental ultrasonography screening for women with dense breasts. Ann Intern Med. 2015;162(3):157–166.
2. Carney PA, Miglioretti DL, Yankaskas BC, et al. Individual and combined effects of age, breast density and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med. 2003;138(3):168–175.
3. Gartlehner G, Thaler K, Chapman A, et al. Mammography in combination with breast ultrasonography versus mammography for breast cancer screening in women at average risk. Cochrane Database Syst Rev. 2013;4:CD009632.
4. Berg WA, Blume JD, Cormack JB, et al. Combined screening with ultrasound and mammography vs. mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299(18):2151–2163.
5. Breast Cancer Surveillance Consortium risk calculator. BCSC Web site. https://tools.bcsc-scc.org/BC5yearRisk/intro.htm. Updated February 13, 2015. Accessed July 17, 2015.
6. NCI Breast Cancer Risk Assessment Tool (Gail model). National Cancer Institute Web site. http://www.cancer.gov/BCRISKTOOL/. Accessed July 17, 2015.
7. IBIS Breast Cancer Risk Evaluation Tool. http://www.ems-trials.org/riskevaluator/. Updated January 9, 2015. Accessed July 17, 2015.
8. Kerlikowske K, Zhu W, Tosteson AN, et al; Breast Cancer Surveillance Consortium. Identifying women with dense breasts at high risk for interval cancer. Ann Intern Med. 2015;162(10):673–681.
9. Committee on Gynecologic Practice. Committee Opinion No. 625: Management of women with dense breasts diagnosed by mammography. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2015;125(3): 750–751.
10. Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013;31(34):2942–2962.
11. Profato JL, Arun BK. Genetic risk assessment for breast and gynecological malignancies. Curr Opin Obstet Gynecol. 2015;27(1):1–5.
12. BRCAPRO. BayesMendel Lab. Harvard University Web site. http://bcb.dfci.harvard.edu/bayesmendel/brcapro.php. Accessed July 19, 2015.
1. Sprague BL, Stout KN, Schechter MD, et al. Benefits, harms and cost-effectiveness of supplemental ultrasonography screening for women with dense breasts. Ann Intern Med. 2015;162(3):157–166.
2. Carney PA, Miglioretti DL, Yankaskas BC, et al. Individual and combined effects of age, breast density and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med. 2003;138(3):168–175.
3. Gartlehner G, Thaler K, Chapman A, et al. Mammography in combination with breast ultrasonography versus mammography for breast cancer screening in women at average risk. Cochrane Database Syst Rev. 2013;4:CD009632.
4. Berg WA, Blume JD, Cormack JB, et al. Combined screening with ultrasound and mammography vs. mammography alone in women at elevated risk of breast cancer. JAMA. 2008;299(18):2151–2163.
5. Breast Cancer Surveillance Consortium risk calculator. BCSC Web site. https://tools.bcsc-scc.org/BC5yearRisk/intro.htm. Updated February 13, 2015. Accessed July 17, 2015.
6. NCI Breast Cancer Risk Assessment Tool (Gail model). National Cancer Institute Web site. http://www.cancer.gov/BCRISKTOOL/. Accessed July 17, 2015.
7. IBIS Breast Cancer Risk Evaluation Tool. http://www.ems-trials.org/riskevaluator/. Updated January 9, 2015. Accessed July 17, 2015.
8. Kerlikowske K, Zhu W, Tosteson AN, et al; Breast Cancer Surveillance Consortium. Identifying women with dense breasts at high risk for interval cancer. Ann Intern Med. 2015;162(10):673–681.
9. Committee on Gynecologic Practice. Committee Opinion No. 625: Management of women with dense breasts diagnosed by mammography. American College of Obstetricians and Gynecologists. Obstet Gynecol. 2015;125(3): 750–751.
10. Visvanathan K, Hurley P, Bantug E, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013;31(34):2942–2962.
11. Profato JL, Arun BK. Genetic risk assessment for breast and gynecological malignancies. Curr Opin Obstet Gynecol. 2015;27(1):1–5.
12. BRCAPRO. BayesMendel Lab. Harvard University Web site. http://bcb.dfci.harvard.edu/bayesmendel/brcapro.php. Accessed July 19, 2015.
Do you practice sophisticated psychiatry? 10 Proposed foundations of advanced care
Some psychiatrists are rapid adopters of the latest discoveries. Others wait before they adopt new modalities and change their practice accordingly. Then, there are some—admittedly, a minority—who stubbornly persist in practicing exactly as they did 30 or 40 years ago when they completed residency.
What are the foundations of exemplary, advanced, brain-based psychiatric care?
Here are my 10 proposed tenets of excellence in psychiatric practice. They reflect superior assessment and management of patients as well as personal growth and contributions to the specialty.
Provide a complete medical assessment for every patient at the first lifetime psychiatric contact, whether inpatient or outpatient. This includes routine physical and neurologic examinations and a panel of basic laboratory tests (complete blood count, liver and kidney functions, urine screen, thyroid-stimulating hormone, electrolytes, fasting glucose, and fasting lipids). All vital signs are measured and recorded. Referrals to other medical specialists are made as needed.
This medical assessment must, of course, include a comprehensive psychiatric evaluation: personal history, social history, medical history, family history, and a complete neuropsychiatric mental status examination.
Create a thorough 3-generation pedigree of all relatives, indicating not only psychopathology, addiction, and legal problems but also medical (especially neurologic) disorders and cause of death.
Perform basic assessment of brain structure and function (a MRI scan, a neurocognitive battery, and tests of neurologic soft signs).
Measure biomarkers that reflect potential harm to the brain according to emerging research—eg, pro-inflammatory markers (such as C-reactive protein [CRP], interleukin-6, and tumor necrosis factor alpha [TNF-α]) and oxidative stress biomarkers of increased free radical activity (superoxide dismutase [SOD], glutathione, thiobarbituric acid [GSH] reactive substances [TBARS], and catalase).
Maintain measurement-based practice, in which:
• severity of illness is measured by a specific, appropriate rating scale (eg, Positive and Negative Syndrome Scale for schizophrenia [PANSS], Young Mania Rating Scale [YMRS], Montgomery-Åsberg Depression Rating Scale [MADRS] for depression, Hamilton Anxiety Rating Scale [HAM-A] for anxiety, Yale-Brown Obsessive Compulsive Scale [Y-BOCS] for obsessions and compulsions)
• degree of response to treatment is measured as a reflection of the extent of drop in the total score of those rating scales, which are administered at every visit
• severity of common side effects is measured by the Simpson-Angus Scale (SAS) for parkinsonism, the Barnes Akathisia Rating Scale (BARS), the Abnormal Involuntary Movement Scale (AIMS) for tardive dyskinesia, the Glasgow Antipsychotic Side-effect Scale (GASS), etc.
Use tier-1 evidence-based psychiatry (that is, findings from large, placebo-controlled, double-blind studies) to select best treatments. This includes being familiar with:
• principles of meta-analysis
• the meaning of low, medium, and large effect sizes
• for every medication used, the calculation and clinical implications of number needed to treat (NNT) and number needed to harm (NNH).
Always combine the dual management approaches of pharmacotherapy plus psychotherapy/psychosocial therapy.
Share knowledge and experience gleaned from practice with the community of psychiatrists, including:
• writing letters to the editor about a clinical matter
• submitting case reports or case series for publication
• teaching students or residents at the local medical school (after obtaining adjunct faculty status).
In addition, psychiatrists should educate the public to eliminate misperceptions and erase stigma about mental illness.
Participate in creating new psychiatric knowledge by developing skills to become a clinical trialist, so that you can participate as an investigator in multicenter clinical trials of new medications, or, at least, refer patients for possible participation in ongoing clinical trials conducted at local academic centers.
Engage in effective and continuous life-learning, by:
• attending weekly Grand Rounds at the nearest academic department of psychiatry
• attending national continuing medical education conferences annually
• scanning PubMed regularly (at least 3 times a week, if not daily) for the latest research related to one’s patients or to read about advances in one’s clinical subspecialty; read the abstracts and download several PDFs a week for subsequent reading.
Some readers will agree with part, but not all, of these proposed components of advanced psychiatric practice. That’s to be expected; I welcome your letters rebutting some tenets, or proposing additional ones, of a sophisticated psychiatric practice. After all, sophistication is a journey, not a destination.
Some psychiatrists are rapid adopters of the latest discoveries. Others wait before they adopt new modalities and change their practice accordingly. Then, there are some—admittedly, a minority—who stubbornly persist in practicing exactly as they did 30 or 40 years ago when they completed residency.
What are the foundations of exemplary, advanced, brain-based psychiatric care?
Here are my 10 proposed tenets of excellence in psychiatric practice. They reflect superior assessment and management of patients as well as personal growth and contributions to the specialty.
Provide a complete medical assessment for every patient at the first lifetime psychiatric contact, whether inpatient or outpatient. This includes routine physical and neurologic examinations and a panel of basic laboratory tests (complete blood count, liver and kidney functions, urine screen, thyroid-stimulating hormone, electrolytes, fasting glucose, and fasting lipids). All vital signs are measured and recorded. Referrals to other medical specialists are made as needed.
This medical assessment must, of course, include a comprehensive psychiatric evaluation: personal history, social history, medical history, family history, and a complete neuropsychiatric mental status examination.
Create a thorough 3-generation pedigree of all relatives, indicating not only psychopathology, addiction, and legal problems but also medical (especially neurologic) disorders and cause of death.
Perform basic assessment of brain structure and function (a MRI scan, a neurocognitive battery, and tests of neurologic soft signs).
Measure biomarkers that reflect potential harm to the brain according to emerging research—eg, pro-inflammatory markers (such as C-reactive protein [CRP], interleukin-6, and tumor necrosis factor alpha [TNF-α]) and oxidative stress biomarkers of increased free radical activity (superoxide dismutase [SOD], glutathione, thiobarbituric acid [GSH] reactive substances [TBARS], and catalase).
Maintain measurement-based practice, in which:
• severity of illness is measured by a specific, appropriate rating scale (eg, Positive and Negative Syndrome Scale for schizophrenia [PANSS], Young Mania Rating Scale [YMRS], Montgomery-Åsberg Depression Rating Scale [MADRS] for depression, Hamilton Anxiety Rating Scale [HAM-A] for anxiety, Yale-Brown Obsessive Compulsive Scale [Y-BOCS] for obsessions and compulsions)
• degree of response to treatment is measured as a reflection of the extent of drop in the total score of those rating scales, which are administered at every visit
• severity of common side effects is measured by the Simpson-Angus Scale (SAS) for parkinsonism, the Barnes Akathisia Rating Scale (BARS), the Abnormal Involuntary Movement Scale (AIMS) for tardive dyskinesia, the Glasgow Antipsychotic Side-effect Scale (GASS), etc.
Use tier-1 evidence-based psychiatry (that is, findings from large, placebo-controlled, double-blind studies) to select best treatments. This includes being familiar with:
• principles of meta-analysis
• the meaning of low, medium, and large effect sizes
• for every medication used, the calculation and clinical implications of number needed to treat (NNT) and number needed to harm (NNH).
Always combine the dual management approaches of pharmacotherapy plus psychotherapy/psychosocial therapy.
Share knowledge and experience gleaned from practice with the community of psychiatrists, including:
• writing letters to the editor about a clinical matter
• submitting case reports or case series for publication
• teaching students or residents at the local medical school (after obtaining adjunct faculty status).
In addition, psychiatrists should educate the public to eliminate misperceptions and erase stigma about mental illness.
Participate in creating new psychiatric knowledge by developing skills to become a clinical trialist, so that you can participate as an investigator in multicenter clinical trials of new medications, or, at least, refer patients for possible participation in ongoing clinical trials conducted at local academic centers.
Engage in effective and continuous life-learning, by:
• attending weekly Grand Rounds at the nearest academic department of psychiatry
• attending national continuing medical education conferences annually
• scanning PubMed regularly (at least 3 times a week, if not daily) for the latest research related to one’s patients or to read about advances in one’s clinical subspecialty; read the abstracts and download several PDFs a week for subsequent reading.
Some readers will agree with part, but not all, of these proposed components of advanced psychiatric practice. That’s to be expected; I welcome your letters rebutting some tenets, or proposing additional ones, of a sophisticated psychiatric practice. After all, sophistication is a journey, not a destination.
Some psychiatrists are rapid adopters of the latest discoveries. Others wait before they adopt new modalities and change their practice accordingly. Then, there are some—admittedly, a minority—who stubbornly persist in practicing exactly as they did 30 or 40 years ago when they completed residency.
What are the foundations of exemplary, advanced, brain-based psychiatric care?
Here are my 10 proposed tenets of excellence in psychiatric practice. They reflect superior assessment and management of patients as well as personal growth and contributions to the specialty.
Provide a complete medical assessment for every patient at the first lifetime psychiatric contact, whether inpatient or outpatient. This includes routine physical and neurologic examinations and a panel of basic laboratory tests (complete blood count, liver and kidney functions, urine screen, thyroid-stimulating hormone, electrolytes, fasting glucose, and fasting lipids). All vital signs are measured and recorded. Referrals to other medical specialists are made as needed.
This medical assessment must, of course, include a comprehensive psychiatric evaluation: personal history, social history, medical history, family history, and a complete neuropsychiatric mental status examination.
Create a thorough 3-generation pedigree of all relatives, indicating not only psychopathology, addiction, and legal problems but also medical (especially neurologic) disorders and cause of death.
Perform basic assessment of brain structure and function (a MRI scan, a neurocognitive battery, and tests of neurologic soft signs).
Measure biomarkers that reflect potential harm to the brain according to emerging research—eg, pro-inflammatory markers (such as C-reactive protein [CRP], interleukin-6, and tumor necrosis factor alpha [TNF-α]) and oxidative stress biomarkers of increased free radical activity (superoxide dismutase [SOD], glutathione, thiobarbituric acid [GSH] reactive substances [TBARS], and catalase).
Maintain measurement-based practice, in which:
• severity of illness is measured by a specific, appropriate rating scale (eg, Positive and Negative Syndrome Scale for schizophrenia [PANSS], Young Mania Rating Scale [YMRS], Montgomery-Åsberg Depression Rating Scale [MADRS] for depression, Hamilton Anxiety Rating Scale [HAM-A] for anxiety, Yale-Brown Obsessive Compulsive Scale [Y-BOCS] for obsessions and compulsions)
• degree of response to treatment is measured as a reflection of the extent of drop in the total score of those rating scales, which are administered at every visit
• severity of common side effects is measured by the Simpson-Angus Scale (SAS) for parkinsonism, the Barnes Akathisia Rating Scale (BARS), the Abnormal Involuntary Movement Scale (AIMS) for tardive dyskinesia, the Glasgow Antipsychotic Side-effect Scale (GASS), etc.
Use tier-1 evidence-based psychiatry (that is, findings from large, placebo-controlled, double-blind studies) to select best treatments. This includes being familiar with:
• principles of meta-analysis
• the meaning of low, medium, and large effect sizes
• for every medication used, the calculation and clinical implications of number needed to treat (NNT) and number needed to harm (NNH).
Always combine the dual management approaches of pharmacotherapy plus psychotherapy/psychosocial therapy.
Share knowledge and experience gleaned from practice with the community of psychiatrists, including:
• writing letters to the editor about a clinical matter
• submitting case reports or case series for publication
• teaching students or residents at the local medical school (after obtaining adjunct faculty status).
In addition, psychiatrists should educate the public to eliminate misperceptions and erase stigma about mental illness.
Participate in creating new psychiatric knowledge by developing skills to become a clinical trialist, so that you can participate as an investigator in multicenter clinical trials of new medications, or, at least, refer patients for possible participation in ongoing clinical trials conducted at local academic centers.
Engage in effective and continuous life-learning, by:
• attending weekly Grand Rounds at the nearest academic department of psychiatry
• attending national continuing medical education conferences annually
• scanning PubMed regularly (at least 3 times a week, if not daily) for the latest research related to one’s patients or to read about advances in one’s clinical subspecialty; read the abstracts and download several PDFs a week for subsequent reading.
Some readers will agree with part, but not all, of these proposed components of advanced psychiatric practice. That’s to be expected; I welcome your letters rebutting some tenets, or proposing additional ones, of a sophisticated psychiatric practice. After all, sophistication is a journey, not a destination.
ASCO 2015: from data and learning, to daily practice
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
The cohabitation of art and genomic science
The art of medicine includes picking the right drug for the right patient, especially when we can choose between different classes of efficacious therapies. But, in view of our growing understanding of the human genome, can science replace art?
That question is part of the promise of pharmacogenetics, the study of how inter-individual genetic differences influence a patient’s response to a specific drug. A patient’s genome dictates the expression of specific enzymes that metabolize a drug with various efficiencies: variant alleles may result in slightly different proteins that express different enzymatic activity, ie, different substrate affinities for a drug resulting in more or less efficient metabolism. Genomic differences may also dictate whether a specific biochemical pathway is dominant in generating a specific pathophysiologic response, in which case drugs that affect that pathway may be strikingly effective. This may partly explain the various responses to different antihypertensive drugs.
Another less well-understood example of pharmacogenetics is the link between specific HLA haplotypes and a dramatic increase in allergic reactions to specific medications, such as the link between HLA-B*57:01 and abacavir hypersensitivity.
In this issue of the Journal, DiPiero et al discuss thiopurine methyltransferase (TPMT), an enzyme responsible for the degradation of azathioprine, and how knowing the genetically determined relative activity of this enzyme should influence our initial dosing of this and related drugs. Patients with certain variant alleles of TPMT degrade azathioprine more slowly, and these patients are at higher risk of myelosuppressive toxicity from the drug when it is given at the full weight-based dose. The TPMT test is expensive but not prohibitively so, and it would seem that genomic testing is a reasonable clinical and cost-effective option.
As in the abacavir scenario noted above, genomic-based dosing of azathioprine makes scientific sense and offers proof of principle for the validity of pharmacogenomics. But is it truly a clinical game-changer?
The answer depends in part on how the prescribing physician doses the drug, which depends in part on what disease is being treated, how fast the drug needs to be at full dose, and whether there are equally effective alternatives. Recommendations have been offered that state if TPMT activity is normal, we can start at the usual maintenance dose of 1.5 to 2 mg/kg/day (or occasionally more). But if the patient is heterozygous for the wild-type gene and thus is a slower drug metabolizer, then initial dosing “should” be reduced to 25 to 50 mg/day, with close observation of the white blood cell count as the dose is slowly increased to the target. The very rare patient who is homozygous for a non–wild-type allele should not be given the drug.
My usual practice has been to start patients on 50 mg or less daily and slowly titrate up, asking them how they are tolerating the drug and watching the white count—notably, the same approach to be taken if I had done genotyping before starting the drug and had found the patient to be heterozygous for the TPMT gene.
Interestingly, one pragmatic clinical trial tested whether genotyping patients before starting azathioprine—with subsequent suggested dosing of the drug based on the genotype as above—was safer and cheaper than letting physicians dose as they chose.1 It turned out that physicians participating in this study still dosed their patients conservatively. Even knowing that they might be able to give full doses from the start in patients with normal TPMT activity, many chose not to. I assume that many of those physicians felt as I do that there was no urgency in reaching the presumed-to-be-effective full weight-based therapeutic dose. (We don’t have a good clinical marker of azathioprine’s efficacy). At 4 months, the maintenance dose was about the same in all groups.
We have robust evidence to support the role of pharmacogenetics in informing the dosing of several medications, more than just the ones I have mentioned here. And in the right settings, we should use pharmacogenetic testing to limit toxicity and perhaps enhance efficacy in our drug selection. As the field moves rapidly forward, we will have many opportunities to improve clinical care by using our patients’ genomic information.
But like it or bemoan it, even when we have science in the house, the art of medicine still plays a role in our clinical decisions.
- Thompson AJ, Newman WG, Elliott RA, Roberts SA, Tricker K, Payne K. The cost-effectiveness of a pharmacogenetic test: a trial-based evaluation of TPMT genotyping for azathioprine. Value Health 2014; 17:22–33.
The art of medicine includes picking the right drug for the right patient, especially when we can choose between different classes of efficacious therapies. But, in view of our growing understanding of the human genome, can science replace art?
That question is part of the promise of pharmacogenetics, the study of how inter-individual genetic differences influence a patient’s response to a specific drug. A patient’s genome dictates the expression of specific enzymes that metabolize a drug with various efficiencies: variant alleles may result in slightly different proteins that express different enzymatic activity, ie, different substrate affinities for a drug resulting in more or less efficient metabolism. Genomic differences may also dictate whether a specific biochemical pathway is dominant in generating a specific pathophysiologic response, in which case drugs that affect that pathway may be strikingly effective. This may partly explain the various responses to different antihypertensive drugs.
Another less well-understood example of pharmacogenetics is the link between specific HLA haplotypes and a dramatic increase in allergic reactions to specific medications, such as the link between HLA-B*57:01 and abacavir hypersensitivity.
In this issue of the Journal, DiPiero et al discuss thiopurine methyltransferase (TPMT), an enzyme responsible for the degradation of azathioprine, and how knowing the genetically determined relative activity of this enzyme should influence our initial dosing of this and related drugs. Patients with certain variant alleles of TPMT degrade azathioprine more slowly, and these patients are at higher risk of myelosuppressive toxicity from the drug when it is given at the full weight-based dose. The TPMT test is expensive but not prohibitively so, and it would seem that genomic testing is a reasonable clinical and cost-effective option.
As in the abacavir scenario noted above, genomic-based dosing of azathioprine makes scientific sense and offers proof of principle for the validity of pharmacogenomics. But is it truly a clinical game-changer?
The answer depends in part on how the prescribing physician doses the drug, which depends in part on what disease is being treated, how fast the drug needs to be at full dose, and whether there are equally effective alternatives. Recommendations have been offered that state if TPMT activity is normal, we can start at the usual maintenance dose of 1.5 to 2 mg/kg/day (or occasionally more). But if the patient is heterozygous for the wild-type gene and thus is a slower drug metabolizer, then initial dosing “should” be reduced to 25 to 50 mg/day, with close observation of the white blood cell count as the dose is slowly increased to the target. The very rare patient who is homozygous for a non–wild-type allele should not be given the drug.
My usual practice has been to start patients on 50 mg or less daily and slowly titrate up, asking them how they are tolerating the drug and watching the white count—notably, the same approach to be taken if I had done genotyping before starting the drug and had found the patient to be heterozygous for the TPMT gene.
Interestingly, one pragmatic clinical trial tested whether genotyping patients before starting azathioprine—with subsequent suggested dosing of the drug based on the genotype as above—was safer and cheaper than letting physicians dose as they chose.1 It turned out that physicians participating in this study still dosed their patients conservatively. Even knowing that they might be able to give full doses from the start in patients with normal TPMT activity, many chose not to. I assume that many of those physicians felt as I do that there was no urgency in reaching the presumed-to-be-effective full weight-based therapeutic dose. (We don’t have a good clinical marker of azathioprine’s efficacy). At 4 months, the maintenance dose was about the same in all groups.
We have robust evidence to support the role of pharmacogenetics in informing the dosing of several medications, more than just the ones I have mentioned here. And in the right settings, we should use pharmacogenetic testing to limit toxicity and perhaps enhance efficacy in our drug selection. As the field moves rapidly forward, we will have many opportunities to improve clinical care by using our patients’ genomic information.
But like it or bemoan it, even when we have science in the house, the art of medicine still plays a role in our clinical decisions.
The art of medicine includes picking the right drug for the right patient, especially when we can choose between different classes of efficacious therapies. But, in view of our growing understanding of the human genome, can science replace art?
That question is part of the promise of pharmacogenetics, the study of how inter-individual genetic differences influence a patient’s response to a specific drug. A patient’s genome dictates the expression of specific enzymes that metabolize a drug with various efficiencies: variant alleles may result in slightly different proteins that express different enzymatic activity, ie, different substrate affinities for a drug resulting in more or less efficient metabolism. Genomic differences may also dictate whether a specific biochemical pathway is dominant in generating a specific pathophysiologic response, in which case drugs that affect that pathway may be strikingly effective. This may partly explain the various responses to different antihypertensive drugs.
Another less well-understood example of pharmacogenetics is the link between specific HLA haplotypes and a dramatic increase in allergic reactions to specific medications, such as the link between HLA-B*57:01 and abacavir hypersensitivity.
In this issue of the Journal, DiPiero et al discuss thiopurine methyltransferase (TPMT), an enzyme responsible for the degradation of azathioprine, and how knowing the genetically determined relative activity of this enzyme should influence our initial dosing of this and related drugs. Patients with certain variant alleles of TPMT degrade azathioprine more slowly, and these patients are at higher risk of myelosuppressive toxicity from the drug when it is given at the full weight-based dose. The TPMT test is expensive but not prohibitively so, and it would seem that genomic testing is a reasonable clinical and cost-effective option.
As in the abacavir scenario noted above, genomic-based dosing of azathioprine makes scientific sense and offers proof of principle for the validity of pharmacogenomics. But is it truly a clinical game-changer?
The answer depends in part on how the prescribing physician doses the drug, which depends in part on what disease is being treated, how fast the drug needs to be at full dose, and whether there are equally effective alternatives. Recommendations have been offered that state if TPMT activity is normal, we can start at the usual maintenance dose of 1.5 to 2 mg/kg/day (or occasionally more). But if the patient is heterozygous for the wild-type gene and thus is a slower drug metabolizer, then initial dosing “should” be reduced to 25 to 50 mg/day, with close observation of the white blood cell count as the dose is slowly increased to the target. The very rare patient who is homozygous for a non–wild-type allele should not be given the drug.
My usual practice has been to start patients on 50 mg or less daily and slowly titrate up, asking them how they are tolerating the drug and watching the white count—notably, the same approach to be taken if I had done genotyping before starting the drug and had found the patient to be heterozygous for the TPMT gene.
Interestingly, one pragmatic clinical trial tested whether genotyping patients before starting azathioprine—with subsequent suggested dosing of the drug based on the genotype as above—was safer and cheaper than letting physicians dose as they chose.1 It turned out that physicians participating in this study still dosed their patients conservatively. Even knowing that they might be able to give full doses from the start in patients with normal TPMT activity, many chose not to. I assume that many of those physicians felt as I do that there was no urgency in reaching the presumed-to-be-effective full weight-based therapeutic dose. (We don’t have a good clinical marker of azathioprine’s efficacy). At 4 months, the maintenance dose was about the same in all groups.
We have robust evidence to support the role of pharmacogenetics in informing the dosing of several medications, more than just the ones I have mentioned here. And in the right settings, we should use pharmacogenetic testing to limit toxicity and perhaps enhance efficacy in our drug selection. As the field moves rapidly forward, we will have many opportunities to improve clinical care by using our patients’ genomic information.
But like it or bemoan it, even when we have science in the house, the art of medicine still plays a role in our clinical decisions.
- Thompson AJ, Newman WG, Elliott RA, Roberts SA, Tricker K, Payne K. The cost-effectiveness of a pharmacogenetic test: a trial-based evaluation of TPMT genotyping for azathioprine. Value Health 2014; 17:22–33.
- Thompson AJ, Newman WG, Elliott RA, Roberts SA, Tricker K, Payne K. The cost-effectiveness of a pharmacogenetic test: a trial-based evaluation of TPMT genotyping for azathioprine. Value Health 2014; 17:22–33.
Is there only 1 neurobiologic psychiatric disorder, with different clinical expressions?
In a report of a study that was published recently in a top-tier psychiatry journal,1 researchers describe a stunning finding that challenges the notion that there is a plethora of psychiatric brain disorders. They conducted a large meta-analysis of 193 published brain imaging studies of people with schizophrenia, bipolar disorder, major depression, obsessive-compulsive disorder (OCD), anxiety, and addiction. They found that those 6 supposedly discrete illnesses are all associated with a varying degree of shrinkage (atrophy or hypoplasia) of the same 3 brain regions:
• Dorsal anterior cingulate cortex. This region around the frontal part of the corpus callosum controls rational cognitive processes, reward anticipation, decision making, empathy, impulse control, and emotional response. Francis Crick, the Nobel laureate who first described the structure of DNA, hypothesized that the anterior cingulate sulcus might even be the center of what we call “free will.”
• Left insula and right insula. The insulae are the cortical regions deep inside the lateral sulcus, which is the fissure that separates the temporal lobe from the parietal and frontal lobes. The functions of the insulae include consciousness, emotions, perceptions, motor control, self-awareness, cognitive functioning, and interpersonal experience. (In addicts, the insular cortex is activated when they are exposed to environmental cues that trigger craving because the insulae are a target for the dopamine system. Notably, it has been reported that, when cigarette addicts suffer a stroke that damages the insulae, they stop smoking completely.)
The 3 regions of the brain, in which pathology extends across 6 DSM-5 diagnoses, work together to manage high-level executive functions, such as working memory, reasoning, and flexible thinking. The degree of dysfunction varies among the 6 clinical disorders, with schizophrenia having the highest severity.
Neurobiological commonality
The idea of shared neurobiological underpinnings among 6 distinct psychiatric disorders flies in the face of the entrenched DSM model, in which those 6 disorders are distinct disease entities. Other studies (including the Bipolar Schizophrenia Network on Intermediate Phenotypes) also found prominent biological similarities in varying degrees across schizophrenia, schizoaffective disorder, and bipolar disorder.2,3 The Research Domain Criteria of the National Institute of Mental Health also embraces the dimensional approach to neurobiological biomarkers across various psychiatric disorders.
A common genetic substrate also is emerging. A recently published genome-wide association study, conducted on 33,332 psychiatric patients and 27,888 controls,4 revealed that a number of genes are shared by 5 different psychiatric disorders: schizophrenia, autism, bipolar disorder, major depression, and attention-deficit/hyperactivity disorder. The genetic phenomenon of the same genes manifesting in different clinical phenotypes is called pleiotropy, and is consistent with shared neurobiological findings.
Genetic and brain structural commonalities among multiple DSM diagnostic categories might explain some well-known clinical observations:
• frequent comorbidity of certain psychiatric disorders, such as depression and addiction in schizophrenia; anxiety and OCD in bipolar disorder; depression with OCD and addictions; and so on
• the presence of intermediate phenotypes in unaffected family members, such as cognitive dysfunction in the parents of patients with schizophrenia, compared with parents of matched healthy controls5
• the much higher rate of psychopathology among family members of patients with a major psychiatric disorder, compared with the general population.6
Core inflexible thinking. So what about clinical features across those disorders that share genetic and neurobiologic similarities? Psychiatrists may agree that symptoms of schizophrenia, bipolar disorder, major depression, OCD, anxiety, and addiction appear very different. However, given that reasoning and flexible thinking are functions of the insulae, which are shrunken in all 6 disorders, one can postulate that inflexible thinking (fixed false beliefs also are called psychotic delusions) might be a common feature across all those disorders. Namely:
• schizophrenia is known for paranoid or implausible delusions
• bipolar disorder is characterized by grandiose delusions
• major depressive disorder is associated with a fixed false belief of worthlessness as well as hopelessness
• anxiety patients harbor the fixed false belief of impending doom or death (the plane will crash if they are a passenger on it)
• OCD manifests as ego-dystonic false beliefs (obsessions) that can progress into ego-syntonic delusions
• people with an alcohol or tobacco addiction are in delusional denial that they are not really addicted or that they will not be harmed by their drug of abuse. Pathologic gamblers harbor the false belief that they will soon reverse their fortunes and “win big.”
It seems that poor reality testing and impaired reasoning is a common feature of not only all 6 psychiatric disorders with shared neurobiology, but others, too, including anorexia nervosa, body dysmorphic disorder, delirium, and dementia.
From a thick volume to… a booklet?
Can you envision a day when psychiatric disorders are conceptualized as having a common genetic, neurobiological, and clinical core, with some variability in phenotype and behavior? If further brain research steers psychiatric nosology in that direction, we might end up with a DSM of 10 pages instead of almost 1,000, with an “Appendix” of genetic, neuroimaging, and other emerging biomarkers.
Bold scientific prophecies often sound delusional—until they come true….
1. Goodkind M, Eickhoff SB, Oathes DJ, et al. Identification of a common neurobiological substrate for mental illness. JAMA Psychiatry. 2015;72(4):305-315.
2. Hill SK, Reilly JL, Keefe RS, et al. Neuropsychological impairments in schizophrenia and psychotic bipolar disorder: findings from the Bipolar- Schizophrenia Network on Intermediate Phenotypes (B-SNIP) study. Am J Psychiatry. 2013;170(11):1275-1284.
3. Skudlarski P, Scretlen DJ, Thaker GK, et al. Diffusion tensor imaging white matter endophenotypes in patients with schizophrenia or psychotic bipolar disorder and their relatives. Am J Psychiatry. 2013;170(8):886-898.
4. Cross-Disorder Group of Psychiatric Genomics Consortium. Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet. 2013;381(9875):1371-1379.
5. Appels MC, Sitskoorn MM, Westers P, et al. Cognitive dysfunction in parents of schizophrenia patients parallel the deficits found in patients. Schizophrenia Res. 2003;63(3):285-293.
6. Braff DL. The importance of endophenotypes in schizophrenia research. Schizophrenia Res. 2015;163(1-3):1-8.
In a report of a study that was published recently in a top-tier psychiatry journal,1 researchers describe a stunning finding that challenges the notion that there is a plethora of psychiatric brain disorders. They conducted a large meta-analysis of 193 published brain imaging studies of people with schizophrenia, bipolar disorder, major depression, obsessive-compulsive disorder (OCD), anxiety, and addiction. They found that those 6 supposedly discrete illnesses are all associated with a varying degree of shrinkage (atrophy or hypoplasia) of the same 3 brain regions:
• Dorsal anterior cingulate cortex. This region around the frontal part of the corpus callosum controls rational cognitive processes, reward anticipation, decision making, empathy, impulse control, and emotional response. Francis Crick, the Nobel laureate who first described the structure of DNA, hypothesized that the anterior cingulate sulcus might even be the center of what we call “free will.”
• Left insula and right insula. The insulae are the cortical regions deep inside the lateral sulcus, which is the fissure that separates the temporal lobe from the parietal and frontal lobes. The functions of the insulae include consciousness, emotions, perceptions, motor control, self-awareness, cognitive functioning, and interpersonal experience. (In addicts, the insular cortex is activated when they are exposed to environmental cues that trigger craving because the insulae are a target for the dopamine system. Notably, it has been reported that, when cigarette addicts suffer a stroke that damages the insulae, they stop smoking completely.)
The 3 regions of the brain, in which pathology extends across 6 DSM-5 diagnoses, work together to manage high-level executive functions, such as working memory, reasoning, and flexible thinking. The degree of dysfunction varies among the 6 clinical disorders, with schizophrenia having the highest severity.
Neurobiological commonality
The idea of shared neurobiological underpinnings among 6 distinct psychiatric disorders flies in the face of the entrenched DSM model, in which those 6 disorders are distinct disease entities. Other studies (including the Bipolar Schizophrenia Network on Intermediate Phenotypes) also found prominent biological similarities in varying degrees across schizophrenia, schizoaffective disorder, and bipolar disorder.2,3 The Research Domain Criteria of the National Institute of Mental Health also embraces the dimensional approach to neurobiological biomarkers across various psychiatric disorders.
A common genetic substrate also is emerging. A recently published genome-wide association study, conducted on 33,332 psychiatric patients and 27,888 controls,4 revealed that a number of genes are shared by 5 different psychiatric disorders: schizophrenia, autism, bipolar disorder, major depression, and attention-deficit/hyperactivity disorder. The genetic phenomenon of the same genes manifesting in different clinical phenotypes is called pleiotropy, and is consistent with shared neurobiological findings.
Genetic and brain structural commonalities among multiple DSM diagnostic categories might explain some well-known clinical observations:
• frequent comorbidity of certain psychiatric disorders, such as depression and addiction in schizophrenia; anxiety and OCD in bipolar disorder; depression with OCD and addictions; and so on
• the presence of intermediate phenotypes in unaffected family members, such as cognitive dysfunction in the parents of patients with schizophrenia, compared with parents of matched healthy controls5
• the much higher rate of psychopathology among family members of patients with a major psychiatric disorder, compared with the general population.6
Core inflexible thinking. So what about clinical features across those disorders that share genetic and neurobiologic similarities? Psychiatrists may agree that symptoms of schizophrenia, bipolar disorder, major depression, OCD, anxiety, and addiction appear very different. However, given that reasoning and flexible thinking are functions of the insulae, which are shrunken in all 6 disorders, one can postulate that inflexible thinking (fixed false beliefs also are called psychotic delusions) might be a common feature across all those disorders. Namely:
• schizophrenia is known for paranoid or implausible delusions
• bipolar disorder is characterized by grandiose delusions
• major depressive disorder is associated with a fixed false belief of worthlessness as well as hopelessness
• anxiety patients harbor the fixed false belief of impending doom or death (the plane will crash if they are a passenger on it)
• OCD manifests as ego-dystonic false beliefs (obsessions) that can progress into ego-syntonic delusions
• people with an alcohol or tobacco addiction are in delusional denial that they are not really addicted or that they will not be harmed by their drug of abuse. Pathologic gamblers harbor the false belief that they will soon reverse their fortunes and “win big.”
It seems that poor reality testing and impaired reasoning is a common feature of not only all 6 psychiatric disorders with shared neurobiology, but others, too, including anorexia nervosa, body dysmorphic disorder, delirium, and dementia.
From a thick volume to… a booklet?
Can you envision a day when psychiatric disorders are conceptualized as having a common genetic, neurobiological, and clinical core, with some variability in phenotype and behavior? If further brain research steers psychiatric nosology in that direction, we might end up with a DSM of 10 pages instead of almost 1,000, with an “Appendix” of genetic, neuroimaging, and other emerging biomarkers.
Bold scientific prophecies often sound delusional—until they come true….
In a report of a study that was published recently in a top-tier psychiatry journal,1 researchers describe a stunning finding that challenges the notion that there is a plethora of psychiatric brain disorders. They conducted a large meta-analysis of 193 published brain imaging studies of people with schizophrenia, bipolar disorder, major depression, obsessive-compulsive disorder (OCD), anxiety, and addiction. They found that those 6 supposedly discrete illnesses are all associated with a varying degree of shrinkage (atrophy or hypoplasia) of the same 3 brain regions:
• Dorsal anterior cingulate cortex. This region around the frontal part of the corpus callosum controls rational cognitive processes, reward anticipation, decision making, empathy, impulse control, and emotional response. Francis Crick, the Nobel laureate who first described the structure of DNA, hypothesized that the anterior cingulate sulcus might even be the center of what we call “free will.”
• Left insula and right insula. The insulae are the cortical regions deep inside the lateral sulcus, which is the fissure that separates the temporal lobe from the parietal and frontal lobes. The functions of the insulae include consciousness, emotions, perceptions, motor control, self-awareness, cognitive functioning, and interpersonal experience. (In addicts, the insular cortex is activated when they are exposed to environmental cues that trigger craving because the insulae are a target for the dopamine system. Notably, it has been reported that, when cigarette addicts suffer a stroke that damages the insulae, they stop smoking completely.)
The 3 regions of the brain, in which pathology extends across 6 DSM-5 diagnoses, work together to manage high-level executive functions, such as working memory, reasoning, and flexible thinking. The degree of dysfunction varies among the 6 clinical disorders, with schizophrenia having the highest severity.
Neurobiological commonality
The idea of shared neurobiological underpinnings among 6 distinct psychiatric disorders flies in the face of the entrenched DSM model, in which those 6 disorders are distinct disease entities. Other studies (including the Bipolar Schizophrenia Network on Intermediate Phenotypes) also found prominent biological similarities in varying degrees across schizophrenia, schizoaffective disorder, and bipolar disorder.2,3 The Research Domain Criteria of the National Institute of Mental Health also embraces the dimensional approach to neurobiological biomarkers across various psychiatric disorders.
A common genetic substrate also is emerging. A recently published genome-wide association study, conducted on 33,332 psychiatric patients and 27,888 controls,4 revealed that a number of genes are shared by 5 different psychiatric disorders: schizophrenia, autism, bipolar disorder, major depression, and attention-deficit/hyperactivity disorder. The genetic phenomenon of the same genes manifesting in different clinical phenotypes is called pleiotropy, and is consistent with shared neurobiological findings.
Genetic and brain structural commonalities among multiple DSM diagnostic categories might explain some well-known clinical observations:
• frequent comorbidity of certain psychiatric disorders, such as depression and addiction in schizophrenia; anxiety and OCD in bipolar disorder; depression with OCD and addictions; and so on
• the presence of intermediate phenotypes in unaffected family members, such as cognitive dysfunction in the parents of patients with schizophrenia, compared with parents of matched healthy controls5
• the much higher rate of psychopathology among family members of patients with a major psychiatric disorder, compared with the general population.6
Core inflexible thinking. So what about clinical features across those disorders that share genetic and neurobiologic similarities? Psychiatrists may agree that symptoms of schizophrenia, bipolar disorder, major depression, OCD, anxiety, and addiction appear very different. However, given that reasoning and flexible thinking are functions of the insulae, which are shrunken in all 6 disorders, one can postulate that inflexible thinking (fixed false beliefs also are called psychotic delusions) might be a common feature across all those disorders. Namely:
• schizophrenia is known for paranoid or implausible delusions
• bipolar disorder is characterized by grandiose delusions
• major depressive disorder is associated with a fixed false belief of worthlessness as well as hopelessness
• anxiety patients harbor the fixed false belief of impending doom or death (the plane will crash if they are a passenger on it)
• OCD manifests as ego-dystonic false beliefs (obsessions) that can progress into ego-syntonic delusions
• people with an alcohol or tobacco addiction are in delusional denial that they are not really addicted or that they will not be harmed by their drug of abuse. Pathologic gamblers harbor the false belief that they will soon reverse their fortunes and “win big.”
It seems that poor reality testing and impaired reasoning is a common feature of not only all 6 psychiatric disorders with shared neurobiology, but others, too, including anorexia nervosa, body dysmorphic disorder, delirium, and dementia.
From a thick volume to… a booklet?
Can you envision a day when psychiatric disorders are conceptualized as having a common genetic, neurobiological, and clinical core, with some variability in phenotype and behavior? If further brain research steers psychiatric nosology in that direction, we might end up with a DSM of 10 pages instead of almost 1,000, with an “Appendix” of genetic, neuroimaging, and other emerging biomarkers.
Bold scientific prophecies often sound delusional—until they come true….
1. Goodkind M, Eickhoff SB, Oathes DJ, et al. Identification of a common neurobiological substrate for mental illness. JAMA Psychiatry. 2015;72(4):305-315.
2. Hill SK, Reilly JL, Keefe RS, et al. Neuropsychological impairments in schizophrenia and psychotic bipolar disorder: findings from the Bipolar- Schizophrenia Network on Intermediate Phenotypes (B-SNIP) study. Am J Psychiatry. 2013;170(11):1275-1284.
3. Skudlarski P, Scretlen DJ, Thaker GK, et al. Diffusion tensor imaging white matter endophenotypes in patients with schizophrenia or psychotic bipolar disorder and their relatives. Am J Psychiatry. 2013;170(8):886-898.
4. Cross-Disorder Group of Psychiatric Genomics Consortium. Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet. 2013;381(9875):1371-1379.
5. Appels MC, Sitskoorn MM, Westers P, et al. Cognitive dysfunction in parents of schizophrenia patients parallel the deficits found in patients. Schizophrenia Res. 2003;63(3):285-293.
6. Braff DL. The importance of endophenotypes in schizophrenia research. Schizophrenia Res. 2015;163(1-3):1-8.
1. Goodkind M, Eickhoff SB, Oathes DJ, et al. Identification of a common neurobiological substrate for mental illness. JAMA Psychiatry. 2015;72(4):305-315.
2. Hill SK, Reilly JL, Keefe RS, et al. Neuropsychological impairments in schizophrenia and psychotic bipolar disorder: findings from the Bipolar- Schizophrenia Network on Intermediate Phenotypes (B-SNIP) study. Am J Psychiatry. 2013;170(11):1275-1284.
3. Skudlarski P, Scretlen DJ, Thaker GK, et al. Diffusion tensor imaging white matter endophenotypes in patients with schizophrenia or psychotic bipolar disorder and their relatives. Am J Psychiatry. 2013;170(8):886-898.
4. Cross-Disorder Group of Psychiatric Genomics Consortium. Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet. 2013;381(9875):1371-1379.
5. Appels MC, Sitskoorn MM, Westers P, et al. Cognitive dysfunction in parents of schizophrenia patients parallel the deficits found in patients. Schizophrenia Res. 2003;63(3):285-293.
6. Braff DL. The importance of endophenotypes in schizophrenia research. Schizophrenia Res. 2015;163(1-3):1-8.
It is not your mother/father’s ASCO anymore…
The vaccine safety belt
I’m not sure if I recall seeing kids in long lines outside of school waiting to receive the polio vaccine, or if these are just memories from movie film clips. I’ve never seen a patient with an active polio infection, and I’ve seen only a few with postpolio syndromes. I’ve never seen a patient with tetanus, smallpox, diphtheria, or typical measles. I’ve seen three cases of pertussis that I know of, and the long delay in diagnosing the first one (my wife) was clearly because at that time clinicians caring for adults were not attuned to a disease that had virtually disappeared from the American landscape. Once I was sensitized to its presence, it was far easier to make the diagnosis in the second case I encountered (myself). The list of infectious diseases that have almost vanished in the last 75 years with the development of specific vaccines is not long, but it is striking. We can easily lose sight of that when focusing on the less-than-perfect effectiveness of the pneumococcal and annual influenza vaccines.
My message in recounting these observations is that, growing up in the traditional Western medical establishment, I find it hard from a historical perspective to view vaccines as anything but a positive contribution to our public and personal health. And yet a vocal minority, generally outside the medical establishment, maintains that vaccination is a potentially dangerous practice to be avoided whenever possible. Their biological arguments are tenuous and rarely supported by controlled clinical outcomes or observational data. The elimination of trace amounts of mercury-containing preservatives from some vaccines has done little to dampen their concerns. The arguments against routine vaccination and mandated vaccination of schoolchildren to maintain herd immunity have acquired a libertarian tone. While I may share the philosophy behind their perspective—for example, I wear my seat belt while driving, but I don’t think I should be fined if I don’t—my not wearing a seat belt does not increase the chance that those who encounter me on a plane, in a movie theater, or at an amusement park will die when subsequently driving their car.
In all likelihood, I will retire from medicine before I ever see a case of typical diphtheria. I don’t think that is an accident of nature or the effect of better hygiene. I’m hoping that the generation of physicians to follow will see far less cervical cancer, and that physicians in Asia will see far less hepatitis B-associated hepatocellular carcinoma as a result of effective vaccination against the viruses associated with these cancers.
As Drs. Faria Farhat and Glenn Wortmann and Dr. Atul Khasnis discuss in their papers in this issue of the Journal, we have more to learn about how to most effectively use vaccines in special populations. It is clearly not a one-strategy-fits-all world. The decision to vaccinate these patients is usually less about public health than about the health of the individual patient.
The real-world effectiveness of many vaccines is less than it appeared to be in controlled clinical trials. Unfortunately, the patients who most need protection against infections, the immunosuppressed, have a blunted response to many vaccines and perhaps should not receive live vaccines. But we have too little evidence on how and when to optimally vaccinate these patients. It still feels a bit like a casino, not a clinic, when I discuss with a modestly immunosuppressed patient whether he or she should be vaccinated with a live vaccine to reduce the risk of shingles and postherpetic neuralgia.
If we have the opportunity, vaccinating before starting immunosuppressive drugs (or before splenectomy) makes sense. But often that is not an option. We are frequently faced with the need to extrapolate efficacy and safety experiences from clinical trials of vaccines that are conducted with healthier patients and with relatively short follow-up. The two vaccination papers in this issue of the Journal provide us with useful information about immunologic and other issues involved when making the decision to vaccinate special patient populations.
Buckle up wisely.
I’m not sure if I recall seeing kids in long lines outside of school waiting to receive the polio vaccine, or if these are just memories from movie film clips. I’ve never seen a patient with an active polio infection, and I’ve seen only a few with postpolio syndromes. I’ve never seen a patient with tetanus, smallpox, diphtheria, or typical measles. I’ve seen three cases of pertussis that I know of, and the long delay in diagnosing the first one (my wife) was clearly because at that time clinicians caring for adults were not attuned to a disease that had virtually disappeared from the American landscape. Once I was sensitized to its presence, it was far easier to make the diagnosis in the second case I encountered (myself). The list of infectious diseases that have almost vanished in the last 75 years with the development of specific vaccines is not long, but it is striking. We can easily lose sight of that when focusing on the less-than-perfect effectiveness of the pneumococcal and annual influenza vaccines.
My message in recounting these observations is that, growing up in the traditional Western medical establishment, I find it hard from a historical perspective to view vaccines as anything but a positive contribution to our public and personal health. And yet a vocal minority, generally outside the medical establishment, maintains that vaccination is a potentially dangerous practice to be avoided whenever possible. Their biological arguments are tenuous and rarely supported by controlled clinical outcomes or observational data. The elimination of trace amounts of mercury-containing preservatives from some vaccines has done little to dampen their concerns. The arguments against routine vaccination and mandated vaccination of schoolchildren to maintain herd immunity have acquired a libertarian tone. While I may share the philosophy behind their perspective—for example, I wear my seat belt while driving, but I don’t think I should be fined if I don’t—my not wearing a seat belt does not increase the chance that those who encounter me on a plane, in a movie theater, or at an amusement park will die when subsequently driving their car.
In all likelihood, I will retire from medicine before I ever see a case of typical diphtheria. I don’t think that is an accident of nature or the effect of better hygiene. I’m hoping that the generation of physicians to follow will see far less cervical cancer, and that physicians in Asia will see far less hepatitis B-associated hepatocellular carcinoma as a result of effective vaccination against the viruses associated with these cancers.
As Drs. Faria Farhat and Glenn Wortmann and Dr. Atul Khasnis discuss in their papers in this issue of the Journal, we have more to learn about how to most effectively use vaccines in special populations. It is clearly not a one-strategy-fits-all world. The decision to vaccinate these patients is usually less about public health than about the health of the individual patient.
The real-world effectiveness of many vaccines is less than it appeared to be in controlled clinical trials. Unfortunately, the patients who most need protection against infections, the immunosuppressed, have a blunted response to many vaccines and perhaps should not receive live vaccines. But we have too little evidence on how and when to optimally vaccinate these patients. It still feels a bit like a casino, not a clinic, when I discuss with a modestly immunosuppressed patient whether he or she should be vaccinated with a live vaccine to reduce the risk of shingles and postherpetic neuralgia.
If we have the opportunity, vaccinating before starting immunosuppressive drugs (or before splenectomy) makes sense. But often that is not an option. We are frequently faced with the need to extrapolate efficacy and safety experiences from clinical trials of vaccines that are conducted with healthier patients and with relatively short follow-up. The two vaccination papers in this issue of the Journal provide us with useful information about immunologic and other issues involved when making the decision to vaccinate special patient populations.
Buckle up wisely.
I’m not sure if I recall seeing kids in long lines outside of school waiting to receive the polio vaccine, or if these are just memories from movie film clips. I’ve never seen a patient with an active polio infection, and I’ve seen only a few with postpolio syndromes. I’ve never seen a patient with tetanus, smallpox, diphtheria, or typical measles. I’ve seen three cases of pertussis that I know of, and the long delay in diagnosing the first one (my wife) was clearly because at that time clinicians caring for adults were not attuned to a disease that had virtually disappeared from the American landscape. Once I was sensitized to its presence, it was far easier to make the diagnosis in the second case I encountered (myself). The list of infectious diseases that have almost vanished in the last 75 years with the development of specific vaccines is not long, but it is striking. We can easily lose sight of that when focusing on the less-than-perfect effectiveness of the pneumococcal and annual influenza vaccines.
My message in recounting these observations is that, growing up in the traditional Western medical establishment, I find it hard from a historical perspective to view vaccines as anything but a positive contribution to our public and personal health. And yet a vocal minority, generally outside the medical establishment, maintains that vaccination is a potentially dangerous practice to be avoided whenever possible. Their biological arguments are tenuous and rarely supported by controlled clinical outcomes or observational data. The elimination of trace amounts of mercury-containing preservatives from some vaccines has done little to dampen their concerns. The arguments against routine vaccination and mandated vaccination of schoolchildren to maintain herd immunity have acquired a libertarian tone. While I may share the philosophy behind their perspective—for example, I wear my seat belt while driving, but I don’t think I should be fined if I don’t—my not wearing a seat belt does not increase the chance that those who encounter me on a plane, in a movie theater, or at an amusement park will die when subsequently driving their car.
In all likelihood, I will retire from medicine before I ever see a case of typical diphtheria. I don’t think that is an accident of nature or the effect of better hygiene. I’m hoping that the generation of physicians to follow will see far less cervical cancer, and that physicians in Asia will see far less hepatitis B-associated hepatocellular carcinoma as a result of effective vaccination against the viruses associated with these cancers.
As Drs. Faria Farhat and Glenn Wortmann and Dr. Atul Khasnis discuss in their papers in this issue of the Journal, we have more to learn about how to most effectively use vaccines in special populations. It is clearly not a one-strategy-fits-all world. The decision to vaccinate these patients is usually less about public health than about the health of the individual patient.
The real-world effectiveness of many vaccines is less than it appeared to be in controlled clinical trials. Unfortunately, the patients who most need protection against infections, the immunosuppressed, have a blunted response to many vaccines and perhaps should not receive live vaccines. But we have too little evidence on how and when to optimally vaccinate these patients. It still feels a bit like a casino, not a clinic, when I discuss with a modestly immunosuppressed patient whether he or she should be vaccinated with a live vaccine to reduce the risk of shingles and postherpetic neuralgia.
If we have the opportunity, vaccinating before starting immunosuppressive drugs (or before splenectomy) makes sense. But often that is not an option. We are frequently faced with the need to extrapolate efficacy and safety experiences from clinical trials of vaccines that are conducted with healthier patients and with relatively short follow-up. The two vaccination papers in this issue of the Journal provide us with useful information about immunologic and other issues involved when making the decision to vaccinate special patient populations.
Buckle up wisely.
Advances in protection against oncogenic human papillomaviruses: The 9-valent vaccine
When Dr. Harald zur Hausen received the 2008 Nobel Prize in Physiology or Medicine for his discovery of the link between human papillomavirus (HPV) infections and genital cancers, he completed a 40-year odyssey to prove that viruses caused human cancer. Initially, zur Hausen, working in the University of Pennsylvania laboratory of the noted virologists Drs. Werner and Gertrude Henle, discovered that the Epstein-Barr virus was involved in the development of Burkitt lymphoma.1 On return to his native Germany, he sought a link between HPV and genital tumors.2
First he isolated HPV 6 and HPV 11 directly from genital warts.3 Then zur Hausen utilized the nucleic acid sequences from HPV6 and the technique of low stringency hybridization to discover HPV 16 and HPV 18 in cervical cancer specimens.4,5 Oncogenic HPV DNA contains 2 genes that produce the oncoproteins E6 and E7. E6 increases the degradation of p53 and E7 inactivates the retino-blastoma protein.6 The double-hit inactivation of 2 tumor suppressor genes, p53 and retinoblastoma protein, increases the mitotic activity of the infected cells, eventually leading to cancer.
zur Hausen tried to persuade companies to develop anti-HPV vaccines but was rebuffed for years. Today, building on his research, we have HPV vaccines that are 2-valent (against HPV types 16 and 18), 4-valent (against HPV types 6, 11, 16, and 18), and 9-valent (against HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58). zur Hausen richly deserved the Nobel Prize for his life-saving discoveries.
Cervical, vulvar, and vaginal cancers
HPV types 16 and 18 cause about 70% of cervical cancers. HPV types 31, 33, 45, 52, and 58 cause about 20% of cervical cancers.7 The 2-, 4-, and 9-valent HPV vaccines have been demonstrated to prevent premalignant cervical disease, including cervical intraepithelial neoplasia (CIN) 2 and CIN 3 and adenocarcinoma in situ.8–11 The development of a 9-valent HPV vaccine is an important advance because it provides more complete immunization against cancer causing viruses.
Approximately 70% of vaginal cancers are caused by HPV infections.12 Among squamous cell vulvar cancers, HPV is detected in approximately 70% of cancers with warty or basaloid histology and 12% of cancers with keratinizing histology.13 In vulvar cancer, HPV 16, 33, and 18 are the most common types detected, representing 73%, 7%, and 5% of cases, respectively. The HPV 4- and 9-valent vaccines have been reported to reduce precancerous lesions of the vagina and vulva.9,11 In most trials, vaccinations that occur before exposure to HPV through sexual encounters appear to provide greater protection than vaccinations that occur after HPV infection.
Anal cancer
Approximately 90% of anal cancers are caused by HPV infection, and HPV types 16 and 18 are detected in 81% and 4% of anal cancers, respectively.14 Among men who have sex with men, the HPV 4-valent vaccine reduced the rate of anal intraepithelial neoplasia, a precursor to anal cancer, by 50%.15 Women receiving the HPV 2-valent vaccine had an 84% reduction in the detection of anal cancer involving HPV types 16 and 18.16
Penile cancer
Approximately 48% of penile cancers harbor oncogenic HPV types.17 Among penile cancers the prevalence of HPV varies from 22% in verrucous cancer to 66% in basaloid and warty cancer. The most prevalent HPV types were 16, 6, and 18, which were observed in 31%, 7%, and 7% of the cancers, respectively.17 Penile cancer is not common and there are no studies directly demonstrating that HPV vaccination prevents penile cancer.
Oropharyngeal cancer
The rate of oropharyngeal cancer caused by HPV is rising rapidly and increasing more rapidly among men than among women.18 Remarkably, HPV-induced oropharyngeal cancer is projected to become more common than cervical cancer in 2020.18
In one report, 72% of oropharyngeal cancers harbored HPV 16, and antibodies against the HPV 16 oncoproteins E6 and E7 were detected in the blood of 64% of the cancer cases.19 In a case control study, having 6 or more lifetime oral-sex partners was associated with a 3.4-fold (95% confidence interval, 1.5 to 6.5) increased risk of developing oropharyngeal cancer.19
According to a population survey, 10% of men and 3.6% of women harbor HPV viruses in their oropharynx.20 In this study approximately 50% of the HPV viruses detected were high-risk types, with the following rank-order prevalence from highest to lowest: 16, 66, 51, 39, 56, 52, 59, 18, 53, 45, 35, 33, and 31.20 Theoretically, the 9-valent vaccine, with protection against HPV types 16, 18, 31, 33, 45, and 52, may be an optimal choice to prevent HPV-induced oropharyngeal cancer because of its broad coverage.
No study has yet proven that HPV vaccination reduces the risk of developing oropharyngeal cancer, but one study demonstrated that vaccination of girls against HPV types 16 and 18 reduced oral carriage of HPV 16 and HPV 18 by 93%.21 Vaccinating boys against HPV has been reported to be cost effective because it could reduce the high health care expenditures associated with treating oropharyngeal cancer.22
Will you be an immunization champion?
Although HPV vaccination reduces the disease burden of cervical, vulvar, vaginal, and anal neoplasia, the CDC reported that, as of 2013, only 38% of girls and 14% of boys in the United States had received 3 doses of HPV vaccine.23 The realization that oropharyngeal cancer caused by HPV is rapidly increasing may provide another catalyst to redouble our efforts to increase the vaccination rates for both boys and girls.
zur Hausen and many other experts have passionately advocated for vaccinating all boys and girls in order to maximize the beneficial effects of HPV vaccination.24 Every clinician can become an immunization champion by advocating that all boys and girls be vaccinated against HPV.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
1. zur Hausen H, Schulte-Holthausen H, Klein G, et al. EBV DNA in biopsies of Burkitt tumours and anaplastic carcinomas of the nasopharynx. Nature. 1970;228(5276):1056–1058.
2. zur Hausen H, Meinhof W, Scheiber W, Bornkamm GW. Attempts to detect virus-specific DNA sequences in human tumors: I. Nucleic acid hybridizations with complementary RNA of human wart virus. Int J Cancer. 1974;13(5):650–656.
3. Gissmann L, deVilliers EM, zur Hausen H. Analysis of human genital warts (condylomata acuminata) and other genital tumors for human papillomavirus type 6 DNA. Int J Cancer. 1982;29(2):143–146.
4. Dürst M, Gissman L, Ikenberg H, zur Hausen H. A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions. Proc Natl Acad Sci USA. 1983;80(12):3812–3815.
5. Boshart M, Gissmann L, Ikenberg H, Kleinheinz A, Scheulen W, zur Hausen H. A new type of papillomavirus DNA, its presence in genital cancer biopsies and in cell lines derived from cervical cancer. EMBO J. 1984;3(5):1151–1157.
6. Münger K, Phelps WC, Bubb V, Howley PM, Schlegel R. The E6 and E7 genes of human papillomavirus type 16 are necessary and sufficient for transformation of primary human keratinocytes. J Virol. 1989;63(10):4417–4423.
7. Serrano B, Alemany L. Tous S, et al. Potential impact of a 9-valent vaccine in human papillomavirus related cervical disease. Infect Agent Cancer. 2012;7(1):38.
8. Paavonen J, Naud P, Salmeron J, et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomized study in young women. Lancet. 2009;374(9686):301–314.
9. Garland SM, Hernandez-Avila M, Wheeler CM, et al; Females United to Unilaterally Reduce Endo/Ectocervical Disease (FUTURE) I Investigators. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928–1943.
10. FUTURE II Study Group. Quadrivalent vaccine against human papilloma virus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915–1927.
11. Joura EA, Giuliano AR, Iversen OE, et al; Broad Spectrum HPV Vaccine Study. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N Engl J Med. 2015;372(8): 711–723.
12. Forman D, de Martel C, Lacey CJ, et al. Global burden of human papillomavirus and related diseases. Vaccine. 2012; 30(suppl 5):F12–F23.
13. de Sanjose S, Alemany L, Ordi J, et al. Worldwide human papillomavirus genotype attribution in over 2000 cases of intraepithelial and invasive lesions of the vulva. Eur J Cancer. 2013;49(16):3450–3461.
14. Alemany L, Saunier M, Alvarado-Cabrero I, et al. Human papillomavirus DNA prevalence and type distribution in anal carcinomas worldwide. Int J Cancer. 2015;136(1):98–107.
15. Palefsky JM, Giuliano AR, Goldstone S, et al. HPV vaccine against anal HPV infection and anal intraepithelial neoplasia. N Engl J Med. 2011;365(17):1576–1585.
16. Kreimer AR, González P, Katki HA, et al. Efficacy of a bivalent HPV 16/18 vaccine against anal HPV 16/18 infection among young women: a nested analysis within the Costa Rica Vaccine Trial. Lancet Oncol. 2011;12(9):862–870.
17. Backes DM, Kurman RJ, Pimenta JM, Smith JS. Systematic review of human papillomavirus prevalence in invasive penile cancer. Cancer Causes Control. 2009;20(4):449–457.
18. Chaturvedi AK, Engels EA, Pfeiffer RM, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J Clin Oncol. 2011;29(32):4294–4301.
19. D’Souza G, Kreimer AR, Viscidi R, et al. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007;356(19):1944–1956.
20. Gillison ML, Broutian T, Pickard RK, et al. Prevalence of oral HPV infection in the United States, 2009-2010. JAMA. 2012; 307(7):693–703.
21. Herrero R, Quint W, Hildesheim A, et al. Reduced prevalence of oral human papillomavirus (HPV) 4 years after bivalent HPV vaccination in a randomised clinical trial in Costa Rica. PLoS One. 2013;8(7):e68329.
22. Graham DM, Isaranuwatchai W, Habbous S, et al. A cost-effectiveness analysis of human papillomavirus vaccination of boys for the prevention of oropharyngeal cancer [published online ahead of print April 13, 2015]. Cancer. doi: 10.1002/cncr.29111.
23. Stokley S, Jeyarajah J, Yankey D, et al. Human papillomavirus vaccination coverage among adolescents, 2007-2013, and postlicensure vaccine safety monitoring, 2006–2014—United States. MMWR Morb Mortal Wkly Rep. 2014;63(29):620–624.
24. Michels KB, zur Hausen H. HPV vaccine for all. Lancet. 2009;374(9686):268–270.
When Dr. Harald zur Hausen received the 2008 Nobel Prize in Physiology or Medicine for his discovery of the link between human papillomavirus (HPV) infections and genital cancers, he completed a 40-year odyssey to prove that viruses caused human cancer. Initially, zur Hausen, working in the University of Pennsylvania laboratory of the noted virologists Drs. Werner and Gertrude Henle, discovered that the Epstein-Barr virus was involved in the development of Burkitt lymphoma.1 On return to his native Germany, he sought a link between HPV and genital tumors.2
First he isolated HPV 6 and HPV 11 directly from genital warts.3 Then zur Hausen utilized the nucleic acid sequences from HPV6 and the technique of low stringency hybridization to discover HPV 16 and HPV 18 in cervical cancer specimens.4,5 Oncogenic HPV DNA contains 2 genes that produce the oncoproteins E6 and E7. E6 increases the degradation of p53 and E7 inactivates the retino-blastoma protein.6 The double-hit inactivation of 2 tumor suppressor genes, p53 and retinoblastoma protein, increases the mitotic activity of the infected cells, eventually leading to cancer.
zur Hausen tried to persuade companies to develop anti-HPV vaccines but was rebuffed for years. Today, building on his research, we have HPV vaccines that are 2-valent (against HPV types 16 and 18), 4-valent (against HPV types 6, 11, 16, and 18), and 9-valent (against HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58). zur Hausen richly deserved the Nobel Prize for his life-saving discoveries.
Cervical, vulvar, and vaginal cancers
HPV types 16 and 18 cause about 70% of cervical cancers. HPV types 31, 33, 45, 52, and 58 cause about 20% of cervical cancers.7 The 2-, 4-, and 9-valent HPV vaccines have been demonstrated to prevent premalignant cervical disease, including cervical intraepithelial neoplasia (CIN) 2 and CIN 3 and adenocarcinoma in situ.8–11 The development of a 9-valent HPV vaccine is an important advance because it provides more complete immunization against cancer causing viruses.
Approximately 70% of vaginal cancers are caused by HPV infections.12 Among squamous cell vulvar cancers, HPV is detected in approximately 70% of cancers with warty or basaloid histology and 12% of cancers with keratinizing histology.13 In vulvar cancer, HPV 16, 33, and 18 are the most common types detected, representing 73%, 7%, and 5% of cases, respectively. The HPV 4- and 9-valent vaccines have been reported to reduce precancerous lesions of the vagina and vulva.9,11 In most trials, vaccinations that occur before exposure to HPV through sexual encounters appear to provide greater protection than vaccinations that occur after HPV infection.
Anal cancer
Approximately 90% of anal cancers are caused by HPV infection, and HPV types 16 and 18 are detected in 81% and 4% of anal cancers, respectively.14 Among men who have sex with men, the HPV 4-valent vaccine reduced the rate of anal intraepithelial neoplasia, a precursor to anal cancer, by 50%.15 Women receiving the HPV 2-valent vaccine had an 84% reduction in the detection of anal cancer involving HPV types 16 and 18.16
Penile cancer
Approximately 48% of penile cancers harbor oncogenic HPV types.17 Among penile cancers the prevalence of HPV varies from 22% in verrucous cancer to 66% in basaloid and warty cancer. The most prevalent HPV types were 16, 6, and 18, which were observed in 31%, 7%, and 7% of the cancers, respectively.17 Penile cancer is not common and there are no studies directly demonstrating that HPV vaccination prevents penile cancer.
Oropharyngeal cancer
The rate of oropharyngeal cancer caused by HPV is rising rapidly and increasing more rapidly among men than among women.18 Remarkably, HPV-induced oropharyngeal cancer is projected to become more common than cervical cancer in 2020.18
In one report, 72% of oropharyngeal cancers harbored HPV 16, and antibodies against the HPV 16 oncoproteins E6 and E7 were detected in the blood of 64% of the cancer cases.19 In a case control study, having 6 or more lifetime oral-sex partners was associated with a 3.4-fold (95% confidence interval, 1.5 to 6.5) increased risk of developing oropharyngeal cancer.19
According to a population survey, 10% of men and 3.6% of women harbor HPV viruses in their oropharynx.20 In this study approximately 50% of the HPV viruses detected were high-risk types, with the following rank-order prevalence from highest to lowest: 16, 66, 51, 39, 56, 52, 59, 18, 53, 45, 35, 33, and 31.20 Theoretically, the 9-valent vaccine, with protection against HPV types 16, 18, 31, 33, 45, and 52, may be an optimal choice to prevent HPV-induced oropharyngeal cancer because of its broad coverage.
No study has yet proven that HPV vaccination reduces the risk of developing oropharyngeal cancer, but one study demonstrated that vaccination of girls against HPV types 16 and 18 reduced oral carriage of HPV 16 and HPV 18 by 93%.21 Vaccinating boys against HPV has been reported to be cost effective because it could reduce the high health care expenditures associated with treating oropharyngeal cancer.22
Will you be an immunization champion?
Although HPV vaccination reduces the disease burden of cervical, vulvar, vaginal, and anal neoplasia, the CDC reported that, as of 2013, only 38% of girls and 14% of boys in the United States had received 3 doses of HPV vaccine.23 The realization that oropharyngeal cancer caused by HPV is rapidly increasing may provide another catalyst to redouble our efforts to increase the vaccination rates for both boys and girls.
zur Hausen and many other experts have passionately advocated for vaccinating all boys and girls in order to maximize the beneficial effects of HPV vaccination.24 Every clinician can become an immunization champion by advocating that all boys and girls be vaccinated against HPV.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
When Dr. Harald zur Hausen received the 2008 Nobel Prize in Physiology or Medicine for his discovery of the link between human papillomavirus (HPV) infections and genital cancers, he completed a 40-year odyssey to prove that viruses caused human cancer. Initially, zur Hausen, working in the University of Pennsylvania laboratory of the noted virologists Drs. Werner and Gertrude Henle, discovered that the Epstein-Barr virus was involved in the development of Burkitt lymphoma.1 On return to his native Germany, he sought a link between HPV and genital tumors.2
First he isolated HPV 6 and HPV 11 directly from genital warts.3 Then zur Hausen utilized the nucleic acid sequences from HPV6 and the technique of low stringency hybridization to discover HPV 16 and HPV 18 in cervical cancer specimens.4,5 Oncogenic HPV DNA contains 2 genes that produce the oncoproteins E6 and E7. E6 increases the degradation of p53 and E7 inactivates the retino-blastoma protein.6 The double-hit inactivation of 2 tumor suppressor genes, p53 and retinoblastoma protein, increases the mitotic activity of the infected cells, eventually leading to cancer.
zur Hausen tried to persuade companies to develop anti-HPV vaccines but was rebuffed for years. Today, building on his research, we have HPV vaccines that are 2-valent (against HPV types 16 and 18), 4-valent (against HPV types 6, 11, 16, and 18), and 9-valent (against HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58). zur Hausen richly deserved the Nobel Prize for his life-saving discoveries.
Cervical, vulvar, and vaginal cancers
HPV types 16 and 18 cause about 70% of cervical cancers. HPV types 31, 33, 45, 52, and 58 cause about 20% of cervical cancers.7 The 2-, 4-, and 9-valent HPV vaccines have been demonstrated to prevent premalignant cervical disease, including cervical intraepithelial neoplasia (CIN) 2 and CIN 3 and adenocarcinoma in situ.8–11 The development of a 9-valent HPV vaccine is an important advance because it provides more complete immunization against cancer causing viruses.
Approximately 70% of vaginal cancers are caused by HPV infections.12 Among squamous cell vulvar cancers, HPV is detected in approximately 70% of cancers with warty or basaloid histology and 12% of cancers with keratinizing histology.13 In vulvar cancer, HPV 16, 33, and 18 are the most common types detected, representing 73%, 7%, and 5% of cases, respectively. The HPV 4- and 9-valent vaccines have been reported to reduce precancerous lesions of the vagina and vulva.9,11 In most trials, vaccinations that occur before exposure to HPV through sexual encounters appear to provide greater protection than vaccinations that occur after HPV infection.
Anal cancer
Approximately 90% of anal cancers are caused by HPV infection, and HPV types 16 and 18 are detected in 81% and 4% of anal cancers, respectively.14 Among men who have sex with men, the HPV 4-valent vaccine reduced the rate of anal intraepithelial neoplasia, a precursor to anal cancer, by 50%.15 Women receiving the HPV 2-valent vaccine had an 84% reduction in the detection of anal cancer involving HPV types 16 and 18.16
Penile cancer
Approximately 48% of penile cancers harbor oncogenic HPV types.17 Among penile cancers the prevalence of HPV varies from 22% in verrucous cancer to 66% in basaloid and warty cancer. The most prevalent HPV types were 16, 6, and 18, which were observed in 31%, 7%, and 7% of the cancers, respectively.17 Penile cancer is not common and there are no studies directly demonstrating that HPV vaccination prevents penile cancer.
Oropharyngeal cancer
The rate of oropharyngeal cancer caused by HPV is rising rapidly and increasing more rapidly among men than among women.18 Remarkably, HPV-induced oropharyngeal cancer is projected to become more common than cervical cancer in 2020.18
In one report, 72% of oropharyngeal cancers harbored HPV 16, and antibodies against the HPV 16 oncoproteins E6 and E7 were detected in the blood of 64% of the cancer cases.19 In a case control study, having 6 or more lifetime oral-sex partners was associated with a 3.4-fold (95% confidence interval, 1.5 to 6.5) increased risk of developing oropharyngeal cancer.19
According to a population survey, 10% of men and 3.6% of women harbor HPV viruses in their oropharynx.20 In this study approximately 50% of the HPV viruses detected were high-risk types, with the following rank-order prevalence from highest to lowest: 16, 66, 51, 39, 56, 52, 59, 18, 53, 45, 35, 33, and 31.20 Theoretically, the 9-valent vaccine, with protection against HPV types 16, 18, 31, 33, 45, and 52, may be an optimal choice to prevent HPV-induced oropharyngeal cancer because of its broad coverage.
No study has yet proven that HPV vaccination reduces the risk of developing oropharyngeal cancer, but one study demonstrated that vaccination of girls against HPV types 16 and 18 reduced oral carriage of HPV 16 and HPV 18 by 93%.21 Vaccinating boys against HPV has been reported to be cost effective because it could reduce the high health care expenditures associated with treating oropharyngeal cancer.22
Will you be an immunization champion?
Although HPV vaccination reduces the disease burden of cervical, vulvar, vaginal, and anal neoplasia, the CDC reported that, as of 2013, only 38% of girls and 14% of boys in the United States had received 3 doses of HPV vaccine.23 The realization that oropharyngeal cancer caused by HPV is rapidly increasing may provide another catalyst to redouble our efforts to increase the vaccination rates for both boys and girls.
zur Hausen and many other experts have passionately advocated for vaccinating all boys and girls in order to maximize the beneficial effects of HPV vaccination.24 Every clinician can become an immunization champion by advocating that all boys and girls be vaccinated against HPV.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
1. zur Hausen H, Schulte-Holthausen H, Klein G, et al. EBV DNA in biopsies of Burkitt tumours and anaplastic carcinomas of the nasopharynx. Nature. 1970;228(5276):1056–1058.
2. zur Hausen H, Meinhof W, Scheiber W, Bornkamm GW. Attempts to detect virus-specific DNA sequences in human tumors: I. Nucleic acid hybridizations with complementary RNA of human wart virus. Int J Cancer. 1974;13(5):650–656.
3. Gissmann L, deVilliers EM, zur Hausen H. Analysis of human genital warts (condylomata acuminata) and other genital tumors for human papillomavirus type 6 DNA. Int J Cancer. 1982;29(2):143–146.
4. Dürst M, Gissman L, Ikenberg H, zur Hausen H. A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions. Proc Natl Acad Sci USA. 1983;80(12):3812–3815.
5. Boshart M, Gissmann L, Ikenberg H, Kleinheinz A, Scheulen W, zur Hausen H. A new type of papillomavirus DNA, its presence in genital cancer biopsies and in cell lines derived from cervical cancer. EMBO J. 1984;3(5):1151–1157.
6. Münger K, Phelps WC, Bubb V, Howley PM, Schlegel R. The E6 and E7 genes of human papillomavirus type 16 are necessary and sufficient for transformation of primary human keratinocytes. J Virol. 1989;63(10):4417–4423.
7. Serrano B, Alemany L. Tous S, et al. Potential impact of a 9-valent vaccine in human papillomavirus related cervical disease. Infect Agent Cancer. 2012;7(1):38.
8. Paavonen J, Naud P, Salmeron J, et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomized study in young women. Lancet. 2009;374(9686):301–314.
9. Garland SM, Hernandez-Avila M, Wheeler CM, et al; Females United to Unilaterally Reduce Endo/Ectocervical Disease (FUTURE) I Investigators. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928–1943.
10. FUTURE II Study Group. Quadrivalent vaccine against human papilloma virus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915–1927.
11. Joura EA, Giuliano AR, Iversen OE, et al; Broad Spectrum HPV Vaccine Study. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N Engl J Med. 2015;372(8): 711–723.
12. Forman D, de Martel C, Lacey CJ, et al. Global burden of human papillomavirus and related diseases. Vaccine. 2012; 30(suppl 5):F12–F23.
13. de Sanjose S, Alemany L, Ordi J, et al. Worldwide human papillomavirus genotype attribution in over 2000 cases of intraepithelial and invasive lesions of the vulva. Eur J Cancer. 2013;49(16):3450–3461.
14. Alemany L, Saunier M, Alvarado-Cabrero I, et al. Human papillomavirus DNA prevalence and type distribution in anal carcinomas worldwide. Int J Cancer. 2015;136(1):98–107.
15. Palefsky JM, Giuliano AR, Goldstone S, et al. HPV vaccine against anal HPV infection and anal intraepithelial neoplasia. N Engl J Med. 2011;365(17):1576–1585.
16. Kreimer AR, González P, Katki HA, et al. Efficacy of a bivalent HPV 16/18 vaccine against anal HPV 16/18 infection among young women: a nested analysis within the Costa Rica Vaccine Trial. Lancet Oncol. 2011;12(9):862–870.
17. Backes DM, Kurman RJ, Pimenta JM, Smith JS. Systematic review of human papillomavirus prevalence in invasive penile cancer. Cancer Causes Control. 2009;20(4):449–457.
18. Chaturvedi AK, Engels EA, Pfeiffer RM, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J Clin Oncol. 2011;29(32):4294–4301.
19. D’Souza G, Kreimer AR, Viscidi R, et al. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007;356(19):1944–1956.
20. Gillison ML, Broutian T, Pickard RK, et al. Prevalence of oral HPV infection in the United States, 2009-2010. JAMA. 2012; 307(7):693–703.
21. Herrero R, Quint W, Hildesheim A, et al. Reduced prevalence of oral human papillomavirus (HPV) 4 years after bivalent HPV vaccination in a randomised clinical trial in Costa Rica. PLoS One. 2013;8(7):e68329.
22. Graham DM, Isaranuwatchai W, Habbous S, et al. A cost-effectiveness analysis of human papillomavirus vaccination of boys for the prevention of oropharyngeal cancer [published online ahead of print April 13, 2015]. Cancer. doi: 10.1002/cncr.29111.
23. Stokley S, Jeyarajah J, Yankey D, et al. Human papillomavirus vaccination coverage among adolescents, 2007-2013, and postlicensure vaccine safety monitoring, 2006–2014—United States. MMWR Morb Mortal Wkly Rep. 2014;63(29):620–624.
24. Michels KB, zur Hausen H. HPV vaccine for all. Lancet. 2009;374(9686):268–270.
1. zur Hausen H, Schulte-Holthausen H, Klein G, et al. EBV DNA in biopsies of Burkitt tumours and anaplastic carcinomas of the nasopharynx. Nature. 1970;228(5276):1056–1058.
2. zur Hausen H, Meinhof W, Scheiber W, Bornkamm GW. Attempts to detect virus-specific DNA sequences in human tumors: I. Nucleic acid hybridizations with complementary RNA of human wart virus. Int J Cancer. 1974;13(5):650–656.
3. Gissmann L, deVilliers EM, zur Hausen H. Analysis of human genital warts (condylomata acuminata) and other genital tumors for human papillomavirus type 6 DNA. Int J Cancer. 1982;29(2):143–146.
4. Dürst M, Gissman L, Ikenberg H, zur Hausen H. A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions. Proc Natl Acad Sci USA. 1983;80(12):3812–3815.
5. Boshart M, Gissmann L, Ikenberg H, Kleinheinz A, Scheulen W, zur Hausen H. A new type of papillomavirus DNA, its presence in genital cancer biopsies and in cell lines derived from cervical cancer. EMBO J. 1984;3(5):1151–1157.
6. Münger K, Phelps WC, Bubb V, Howley PM, Schlegel R. The E6 and E7 genes of human papillomavirus type 16 are necessary and sufficient for transformation of primary human keratinocytes. J Virol. 1989;63(10):4417–4423.
7. Serrano B, Alemany L. Tous S, et al. Potential impact of a 9-valent vaccine in human papillomavirus related cervical disease. Infect Agent Cancer. 2012;7(1):38.
8. Paavonen J, Naud P, Salmeron J, et al. Efficacy of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine against cervical infection and precancer caused by oncogenic HPV types (PATRICIA): final analysis of a double-blind, randomized study in young women. Lancet. 2009;374(9686):301–314.
9. Garland SM, Hernandez-Avila M, Wheeler CM, et al; Females United to Unilaterally Reduce Endo/Ectocervical Disease (FUTURE) I Investigators. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med. 2007;356(19):1928–1943.
10. FUTURE II Study Group. Quadrivalent vaccine against human papilloma virus to prevent high-grade cervical lesions. N Engl J Med. 2007;356(19):1915–1927.
11. Joura EA, Giuliano AR, Iversen OE, et al; Broad Spectrum HPV Vaccine Study. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N Engl J Med. 2015;372(8): 711–723.
12. Forman D, de Martel C, Lacey CJ, et al. Global burden of human papillomavirus and related diseases. Vaccine. 2012; 30(suppl 5):F12–F23.
13. de Sanjose S, Alemany L, Ordi J, et al. Worldwide human papillomavirus genotype attribution in over 2000 cases of intraepithelial and invasive lesions of the vulva. Eur J Cancer. 2013;49(16):3450–3461.
14. Alemany L, Saunier M, Alvarado-Cabrero I, et al. Human papillomavirus DNA prevalence and type distribution in anal carcinomas worldwide. Int J Cancer. 2015;136(1):98–107.
15. Palefsky JM, Giuliano AR, Goldstone S, et al. HPV vaccine against anal HPV infection and anal intraepithelial neoplasia. N Engl J Med. 2011;365(17):1576–1585.
16. Kreimer AR, González P, Katki HA, et al. Efficacy of a bivalent HPV 16/18 vaccine against anal HPV 16/18 infection among young women: a nested analysis within the Costa Rica Vaccine Trial. Lancet Oncol. 2011;12(9):862–870.
17. Backes DM, Kurman RJ, Pimenta JM, Smith JS. Systematic review of human papillomavirus prevalence in invasive penile cancer. Cancer Causes Control. 2009;20(4):449–457.
18. Chaturvedi AK, Engels EA, Pfeiffer RM, et al. Human papillomavirus and rising oropharyngeal cancer incidence in the United States. J Clin Oncol. 2011;29(32):4294–4301.
19. D’Souza G, Kreimer AR, Viscidi R, et al. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007;356(19):1944–1956.
20. Gillison ML, Broutian T, Pickard RK, et al. Prevalence of oral HPV infection in the United States, 2009-2010. JAMA. 2012; 307(7):693–703.
21. Herrero R, Quint W, Hildesheim A, et al. Reduced prevalence of oral human papillomavirus (HPV) 4 years after bivalent HPV vaccination in a randomised clinical trial in Costa Rica. PLoS One. 2013;8(7):e68329.
22. Graham DM, Isaranuwatchai W, Habbous S, et al. A cost-effectiveness analysis of human papillomavirus vaccination of boys for the prevention of oropharyngeal cancer [published online ahead of print April 13, 2015]. Cancer. doi: 10.1002/cncr.29111.
23. Stokley S, Jeyarajah J, Yankey D, et al. Human papillomavirus vaccination coverage among adolescents, 2007-2013, and postlicensure vaccine safety monitoring, 2006–2014—United States. MMWR Morb Mortal Wkly Rep. 2014;63(29):620–624.
24. Michels KB, zur Hausen H. HPV vaccine for all. Lancet. 2009;374(9686):268–270.
Beyond dopamine: Brain repair tactics in schizophrenia
For the past 60 years, the standard of care has remained one-dimensional in this brain syndrome, even though the clinical and neurobiological complexities of schizophrenia are multidimensional. Dopamine D2 receptor antagonists, discovered serendipitously in the 1950s, have remained the mainstay of treatment, despite momentous insights about the neurodevelopmental and neurodegenerative processes of schizophrenia.
Why do we ignore abundant evidence that the brain in schizophrenia needs extensive structural repair, not simply a reduction in the activity of a single neurotransmitter in the mesolimbic dopamine tract? Perhaps the age-old dogmatic pessimism that neurodegeneration cannot be reversed has inhibited the field from attempting to escape the dopamine box, so to speak, and from developing innovative, even radical, approaches to repair of the brain of persons with schizophrenia.
But radical thinking is justified when dealing with a cruel brain syndrome that disables young adults in the prime of life.
We should exploit neuroprotective tactics
Several neuroprotective approaches to preventing or reversing the degenerative changes across brain regions in schizophrenia are now recognized. Indirect evidence exists for such interventions in animal models, but the results of few controlled human studies have been published.
Here are my proposals for using neuroprotective tactics to address the unmet need to repair the brain of patients ravaged by neurotoxic psychotic relapses.
Promote 100% adherence to antipsychotic therapy. The simplest tactic to protect the brain from atrophy in patients with schizophrenia is to use long-acting injectable antipsychotic agents immediately after the first psychotic episode. The risk of a psychotic relapse is far lower (7-fold lower, according to a study performed at the University of California, Los Angeles, that soon will be published) with an injectable medication than with oral medication in first-episode patients. Preventing psychotic episodes is, logically, the most important neuroprotective tactic.
Enhance neurogenesis. The brain has 2 neurogenic regions that produce progenitor cells (stem cells) that gradually mature and differentiate into neurons and glia. That is how the brain naturally replenishes itself throughout life. This adult neurogenesis process, carried out in the dentate gyrus of the hippocampus and in the subventricular zone, stops during psychosis but resumes when psychosis remits.
Second-generation antipsychotics (but not first-generation agents) stimulate neurogenesis in animals.1 Haloperidol, in fact, does the opposite—suppressing neurogenesis and causing neuronal death via 15 different molecular mechanisms (see my editorial, “Haloperidol clearly is neurotoxic. Should it be banned?,” in the July 2013 issue).
Other psychotropics also induce neurogenesis, including selective serotonin reuptake inhibitors (SSRIs), which increase hippocampal neurogenesis (atypical antipsychotics appear to increase neurogenesis in the subventricular zone).2 SSRIs often have been used in schizophrenia patients for 2 common comorbid conditions: depression and anxiety. These agents can help regenerate brain tissue, in addition to providing their approved therapeutic indications.
Lithium and valproate have been shown to be neuroprotective3 and to stimulate neurogenesis. Both are often used in schizoaffective disorder, bipolar type; they can exert a neuroprotective effect in addition to their clinical usefulness. The combination of an SSRI or lithium with a second-generation antipsychotic could be synergistic in turbocharging neurogenesis. This sounds like polypharmacy—but it is a rational approach that deserves to be put to the test.
Increase neurotrophins, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF). When neurotrophin levels decline, the brain starts shrinking because of apoptosis. Psychosis lowers neurotrophins drastically—by approximately 60%. Atypical antipsychotics have been reported to increase the level of neurotrophins; haloperidol actually lowers those levels.4
Decrease inflammation. Psychosis has been shown to be associated with neuro-inflammation, as reflected in a surge of pro-inflammatory cytokines (released from activated microglia).5 A rise in interleukin-6, tumor necrosis factor-alpha, interferon-gamma, and other pro-inflammatory markers has been extensively documented in many studies.
With that observed rise in mind, several controlled studies have shown that adding an anti-inflammatory agent (aspirin, a nonsteroidal anti-inflammatory drug, a COX-2 inhibitor, or minocycline) to an antipsychotic can accentuate the therapeutic response, especially during a first episode of psychosis.6 Note also that second-generation antipsychotics have anti-inflammatory effects7 as well that might be part of their efficacy beyond blocking dopamine D2 receptors.
Decrease free radicals. Microglia are activated by psychosis to release free radicals, also known as reactive oxygen species; these include nitric oxide, superoxide, and peroxynitrate. All these species are destructive to brain tissue. Using an adjunctive strong antioxidant, such as N-acetyl cysteine,8 with an antipsychotic might help neutralize destructive effects of free radicals and protect the brain from tissue loss during a psychotic episode.
Avoid apoptosis inducers. Several substances can initiate programmed cell death (apoptosis), which is triggered during psychosis (believed to be caused by increased dopamine and, possibly, glutamate, activity) and which leads to brain atrophy. Patients with schizophrenia must be protected from these apoptosis inducers:
• amphetamine
• cocaine
• Cannabis
• lipid peroxidation products
• inflammatory cytokines.
Apoptosis can be inhibited by maintaining high levels of neurotrophic factors. Atypical, but not typical, antipsychotics increase levels of neurotrophins, such as NGF and BDNF.4 In addition, the Bcl-2 family of proteins inhibits apoptosis,9 and drugs such as lithium and valproate can induce Bcl-2 and protect against apoptosis and neuronal loss.3
Restore white-matter integrity. Numerous studies using diffusion tensor imaging have revealed that myelin is reduced or lacks integrity in schizophrenia. This results in loss of critical connectivity among brain regions, which might explain psychotic and cognitive symptoms. One possible way to repair white matter, which becomes more damaged after multiple psychotic episodes, is to use drugs indicated to treat the demyelinating disorder multiple sclerosis. Antagonists of LINGO-1, a negative regulator of axonal myelination, are a prominent possibility; a recent study reported altered signaling of LINGO-1 in schizophrenia.10
Decrease excessive glutamate. Because glutamate is neurotoxic and might contribute to brain-tissue loss during psychosis, it is important to reduce glutamate activity in schizophrenia. Lamotrigine and valproate are both known to do that.11 Several studies indicate that adjunctive lamotrigine might be helpful in schizophrenia.12
Inhibit caspase-3, also known as the “death cascade,” which is involved in brain-tissue loss. Eicosapentaenoic acid is an omega-3 fatty acid that inhibits caspase-3. Interestingly, omega-3 levels in patients with schizophrenia are significantly lower than in healthy subjects.13 Lithium also can inhibit caspase-3.
Do these proposals sound radical?
Most of the recommendations I’ve made here are not employed in the clinical practice of psychiatry. These ideas must be put to the test in controlled clinical trials.
The crux of my argument is that we need to think outside the “dopamine box” and focus on brain repair if we are to make progress in reversing, even preventing, neurodegeneration and clinical deterioration in this disabling brain syndrome. Just as cancer often is treated with rational polypharmacy, schizophrenia might need a similar approach. To vanquish schizophrenia—a goal that has eluded us—it is imperative to pursue radically novel and disruptive therapeutic strategies. The ideas I’ve listed here sound the call that the quest to repair the brain in schizophrenia must begin, and soon.
1. Agius N, Nandra, KS. Do atypical antipsychotics promote neurogenesis as a class effect? Psychiatr Danub. 2012;24(suppl 1):S191-S193.
2. Nasrallah HA, Hopkins T, Pixley SK. Differential effects of antipsychotic and antidepressant drugs on neurogenic regions in rats. Brain Res. 2010;1354:23-29.
3. Chiu CT, Wang Z, Hunsberger JG, et al. Therapeutic potential of mood stabilizers lithium and valproic acid: beyond bipolar disorder. Pharmacol Rev. 2013;65(1):105-142.
4. Parikh V, Khan MM, Terry A, et al. Differential effects of typical and atypical antipsychotics on nerve growth factor and choline acetyltransferase expression in the cortex and nucleus basalis of rats. J Psychiatr Res. 2004;38(5):521-529.
5. Monji A, Kato TA, Mizoguchi Y, et al. Neuro-inflammation in schizophrenia especially focused on the role of microglia. Prog Neuropsychopharmacol Biol Psychiatry. 2013;42:115-121.
6. Sommer IE, deWitte L, Begemann M, et al. Nonsteriodal anti-inflammatory drugs in schizophrenia: ready for practice or a good start? A meta-analysis. J Clin Psychiatry. 2012;73(4):414-419.
7. Bian Q, Kato T, Monji A, et al. The effect of atypical anti-psychotics perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
8. Berk M, Copolov D, Dean O, et al. N-acetyl cysteine as a glutathione precursor for schizophrenia—a double-blind, randomized, placebo-controlled trial. Biol Psychiatry. 2008;64(5):361-368.
9. Huang J, Fairbrother W, Reed JC, et al. Therapeutic targeting of Bcl-2 family for treatment of B-cell malignancies. Expert Rev Hematol. 2015;8(3):283-297.
10. Fernandez-Enright F, Andrews JL, Newell KA, et al. Novel implications of Lingo-1 and its signaling partners in schizophrenia. Transl Psychiatry. 2014; 4:e348.
11. Zink M, Correll CU. Glutamatergic agents for schizophrenia: current evidence and perspectives. Expert Rev Clin Pharmacol. 2015;8(3):335-352.
12. Kremer I, Vass A, Gorelik I, et al. Placebo-controlled trial of lamotrigine added to conventional and atypical antipsychotics in schizophrenia. Biol Psychiatry. 2004;56(6):444-446.
13. McEvoy J, Baillie RA, Zhu H, et al. Lipidomics reveals early metabolic changes in subjects with schizophrenia: effects of atypical antipsychotics. PLoS One. 2013;8(7):e68717.
For the past 60 years, the standard of care has remained one-dimensional in this brain syndrome, even though the clinical and neurobiological complexities of schizophrenia are multidimensional. Dopamine D2 receptor antagonists, discovered serendipitously in the 1950s, have remained the mainstay of treatment, despite momentous insights about the neurodevelopmental and neurodegenerative processes of schizophrenia.
Why do we ignore abundant evidence that the brain in schizophrenia needs extensive structural repair, not simply a reduction in the activity of a single neurotransmitter in the mesolimbic dopamine tract? Perhaps the age-old dogmatic pessimism that neurodegeneration cannot be reversed has inhibited the field from attempting to escape the dopamine box, so to speak, and from developing innovative, even radical, approaches to repair of the brain of persons with schizophrenia.
But radical thinking is justified when dealing with a cruel brain syndrome that disables young adults in the prime of life.
We should exploit neuroprotective tactics
Several neuroprotective approaches to preventing or reversing the degenerative changes across brain regions in schizophrenia are now recognized. Indirect evidence exists for such interventions in animal models, but the results of few controlled human studies have been published.
Here are my proposals for using neuroprotective tactics to address the unmet need to repair the brain of patients ravaged by neurotoxic psychotic relapses.
Promote 100% adherence to antipsychotic therapy. The simplest tactic to protect the brain from atrophy in patients with schizophrenia is to use long-acting injectable antipsychotic agents immediately after the first psychotic episode. The risk of a psychotic relapse is far lower (7-fold lower, according to a study performed at the University of California, Los Angeles, that soon will be published) with an injectable medication than with oral medication in first-episode patients. Preventing psychotic episodes is, logically, the most important neuroprotective tactic.
Enhance neurogenesis. The brain has 2 neurogenic regions that produce progenitor cells (stem cells) that gradually mature and differentiate into neurons and glia. That is how the brain naturally replenishes itself throughout life. This adult neurogenesis process, carried out in the dentate gyrus of the hippocampus and in the subventricular zone, stops during psychosis but resumes when psychosis remits.
Second-generation antipsychotics (but not first-generation agents) stimulate neurogenesis in animals.1 Haloperidol, in fact, does the opposite—suppressing neurogenesis and causing neuronal death via 15 different molecular mechanisms (see my editorial, “Haloperidol clearly is neurotoxic. Should it be banned?,” in the July 2013 issue).
Other psychotropics also induce neurogenesis, including selective serotonin reuptake inhibitors (SSRIs), which increase hippocampal neurogenesis (atypical antipsychotics appear to increase neurogenesis in the subventricular zone).2 SSRIs often have been used in schizophrenia patients for 2 common comorbid conditions: depression and anxiety. These agents can help regenerate brain tissue, in addition to providing their approved therapeutic indications.
Lithium and valproate have been shown to be neuroprotective3 and to stimulate neurogenesis. Both are often used in schizoaffective disorder, bipolar type; they can exert a neuroprotective effect in addition to their clinical usefulness. The combination of an SSRI or lithium with a second-generation antipsychotic could be synergistic in turbocharging neurogenesis. This sounds like polypharmacy—but it is a rational approach that deserves to be put to the test.
Increase neurotrophins, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF). When neurotrophin levels decline, the brain starts shrinking because of apoptosis. Psychosis lowers neurotrophins drastically—by approximately 60%. Atypical antipsychotics have been reported to increase the level of neurotrophins; haloperidol actually lowers those levels.4
Decrease inflammation. Psychosis has been shown to be associated with neuro-inflammation, as reflected in a surge of pro-inflammatory cytokines (released from activated microglia).5 A rise in interleukin-6, tumor necrosis factor-alpha, interferon-gamma, and other pro-inflammatory markers has been extensively documented in many studies.
With that observed rise in mind, several controlled studies have shown that adding an anti-inflammatory agent (aspirin, a nonsteroidal anti-inflammatory drug, a COX-2 inhibitor, or minocycline) to an antipsychotic can accentuate the therapeutic response, especially during a first episode of psychosis.6 Note also that second-generation antipsychotics have anti-inflammatory effects7 as well that might be part of their efficacy beyond blocking dopamine D2 receptors.
Decrease free radicals. Microglia are activated by psychosis to release free radicals, also known as reactive oxygen species; these include nitric oxide, superoxide, and peroxynitrate. All these species are destructive to brain tissue. Using an adjunctive strong antioxidant, such as N-acetyl cysteine,8 with an antipsychotic might help neutralize destructive effects of free radicals and protect the brain from tissue loss during a psychotic episode.
Avoid apoptosis inducers. Several substances can initiate programmed cell death (apoptosis), which is triggered during psychosis (believed to be caused by increased dopamine and, possibly, glutamate, activity) and which leads to brain atrophy. Patients with schizophrenia must be protected from these apoptosis inducers:
• amphetamine
• cocaine
• Cannabis
• lipid peroxidation products
• inflammatory cytokines.
Apoptosis can be inhibited by maintaining high levels of neurotrophic factors. Atypical, but not typical, antipsychotics increase levels of neurotrophins, such as NGF and BDNF.4 In addition, the Bcl-2 family of proteins inhibits apoptosis,9 and drugs such as lithium and valproate can induce Bcl-2 and protect against apoptosis and neuronal loss.3
Restore white-matter integrity. Numerous studies using diffusion tensor imaging have revealed that myelin is reduced or lacks integrity in schizophrenia. This results in loss of critical connectivity among brain regions, which might explain psychotic and cognitive symptoms. One possible way to repair white matter, which becomes more damaged after multiple psychotic episodes, is to use drugs indicated to treat the demyelinating disorder multiple sclerosis. Antagonists of LINGO-1, a negative regulator of axonal myelination, are a prominent possibility; a recent study reported altered signaling of LINGO-1 in schizophrenia.10
Decrease excessive glutamate. Because glutamate is neurotoxic and might contribute to brain-tissue loss during psychosis, it is important to reduce glutamate activity in schizophrenia. Lamotrigine and valproate are both known to do that.11 Several studies indicate that adjunctive lamotrigine might be helpful in schizophrenia.12
Inhibit caspase-3, also known as the “death cascade,” which is involved in brain-tissue loss. Eicosapentaenoic acid is an omega-3 fatty acid that inhibits caspase-3. Interestingly, omega-3 levels in patients with schizophrenia are significantly lower than in healthy subjects.13 Lithium also can inhibit caspase-3.
Do these proposals sound radical?
Most of the recommendations I’ve made here are not employed in the clinical practice of psychiatry. These ideas must be put to the test in controlled clinical trials.
The crux of my argument is that we need to think outside the “dopamine box” and focus on brain repair if we are to make progress in reversing, even preventing, neurodegeneration and clinical deterioration in this disabling brain syndrome. Just as cancer often is treated with rational polypharmacy, schizophrenia might need a similar approach. To vanquish schizophrenia—a goal that has eluded us—it is imperative to pursue radically novel and disruptive therapeutic strategies. The ideas I’ve listed here sound the call that the quest to repair the brain in schizophrenia must begin, and soon.
For the past 60 years, the standard of care has remained one-dimensional in this brain syndrome, even though the clinical and neurobiological complexities of schizophrenia are multidimensional. Dopamine D2 receptor antagonists, discovered serendipitously in the 1950s, have remained the mainstay of treatment, despite momentous insights about the neurodevelopmental and neurodegenerative processes of schizophrenia.
Why do we ignore abundant evidence that the brain in schizophrenia needs extensive structural repair, not simply a reduction in the activity of a single neurotransmitter in the mesolimbic dopamine tract? Perhaps the age-old dogmatic pessimism that neurodegeneration cannot be reversed has inhibited the field from attempting to escape the dopamine box, so to speak, and from developing innovative, even radical, approaches to repair of the brain of persons with schizophrenia.
But radical thinking is justified when dealing with a cruel brain syndrome that disables young adults in the prime of life.
We should exploit neuroprotective tactics
Several neuroprotective approaches to preventing or reversing the degenerative changes across brain regions in schizophrenia are now recognized. Indirect evidence exists for such interventions in animal models, but the results of few controlled human studies have been published.
Here are my proposals for using neuroprotective tactics to address the unmet need to repair the brain of patients ravaged by neurotoxic psychotic relapses.
Promote 100% adherence to antipsychotic therapy. The simplest tactic to protect the brain from atrophy in patients with schizophrenia is to use long-acting injectable antipsychotic agents immediately after the first psychotic episode. The risk of a psychotic relapse is far lower (7-fold lower, according to a study performed at the University of California, Los Angeles, that soon will be published) with an injectable medication than with oral medication in first-episode patients. Preventing psychotic episodes is, logically, the most important neuroprotective tactic.
Enhance neurogenesis. The brain has 2 neurogenic regions that produce progenitor cells (stem cells) that gradually mature and differentiate into neurons and glia. That is how the brain naturally replenishes itself throughout life. This adult neurogenesis process, carried out in the dentate gyrus of the hippocampus and in the subventricular zone, stops during psychosis but resumes when psychosis remits.
Second-generation antipsychotics (but not first-generation agents) stimulate neurogenesis in animals.1 Haloperidol, in fact, does the opposite—suppressing neurogenesis and causing neuronal death via 15 different molecular mechanisms (see my editorial, “Haloperidol clearly is neurotoxic. Should it be banned?,” in the July 2013 issue).
Other psychotropics also induce neurogenesis, including selective serotonin reuptake inhibitors (SSRIs), which increase hippocampal neurogenesis (atypical antipsychotics appear to increase neurogenesis in the subventricular zone).2 SSRIs often have been used in schizophrenia patients for 2 common comorbid conditions: depression and anxiety. These agents can help regenerate brain tissue, in addition to providing their approved therapeutic indications.
Lithium and valproate have been shown to be neuroprotective3 and to stimulate neurogenesis. Both are often used in schizoaffective disorder, bipolar type; they can exert a neuroprotective effect in addition to their clinical usefulness. The combination of an SSRI or lithium with a second-generation antipsychotic could be synergistic in turbocharging neurogenesis. This sounds like polypharmacy—but it is a rational approach that deserves to be put to the test.
Increase neurotrophins, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF). When neurotrophin levels decline, the brain starts shrinking because of apoptosis. Psychosis lowers neurotrophins drastically—by approximately 60%. Atypical antipsychotics have been reported to increase the level of neurotrophins; haloperidol actually lowers those levels.4
Decrease inflammation. Psychosis has been shown to be associated with neuro-inflammation, as reflected in a surge of pro-inflammatory cytokines (released from activated microglia).5 A rise in interleukin-6, tumor necrosis factor-alpha, interferon-gamma, and other pro-inflammatory markers has been extensively documented in many studies.
With that observed rise in mind, several controlled studies have shown that adding an anti-inflammatory agent (aspirin, a nonsteroidal anti-inflammatory drug, a COX-2 inhibitor, or minocycline) to an antipsychotic can accentuate the therapeutic response, especially during a first episode of psychosis.6 Note also that second-generation antipsychotics have anti-inflammatory effects7 as well that might be part of their efficacy beyond blocking dopamine D2 receptors.
Decrease free radicals. Microglia are activated by psychosis to release free radicals, also known as reactive oxygen species; these include nitric oxide, superoxide, and peroxynitrate. All these species are destructive to brain tissue. Using an adjunctive strong antioxidant, such as N-acetyl cysteine,8 with an antipsychotic might help neutralize destructive effects of free radicals and protect the brain from tissue loss during a psychotic episode.
Avoid apoptosis inducers. Several substances can initiate programmed cell death (apoptosis), which is triggered during psychosis (believed to be caused by increased dopamine and, possibly, glutamate, activity) and which leads to brain atrophy. Patients with schizophrenia must be protected from these apoptosis inducers:
• amphetamine
• cocaine
• Cannabis
• lipid peroxidation products
• inflammatory cytokines.
Apoptosis can be inhibited by maintaining high levels of neurotrophic factors. Atypical, but not typical, antipsychotics increase levels of neurotrophins, such as NGF and BDNF.4 In addition, the Bcl-2 family of proteins inhibits apoptosis,9 and drugs such as lithium and valproate can induce Bcl-2 and protect against apoptosis and neuronal loss.3
Restore white-matter integrity. Numerous studies using diffusion tensor imaging have revealed that myelin is reduced or lacks integrity in schizophrenia. This results in loss of critical connectivity among brain regions, which might explain psychotic and cognitive symptoms. One possible way to repair white matter, which becomes more damaged after multiple psychotic episodes, is to use drugs indicated to treat the demyelinating disorder multiple sclerosis. Antagonists of LINGO-1, a negative regulator of axonal myelination, are a prominent possibility; a recent study reported altered signaling of LINGO-1 in schizophrenia.10
Decrease excessive glutamate. Because glutamate is neurotoxic and might contribute to brain-tissue loss during psychosis, it is important to reduce glutamate activity in schizophrenia. Lamotrigine and valproate are both known to do that.11 Several studies indicate that adjunctive lamotrigine might be helpful in schizophrenia.12
Inhibit caspase-3, also known as the “death cascade,” which is involved in brain-tissue loss. Eicosapentaenoic acid is an omega-3 fatty acid that inhibits caspase-3. Interestingly, omega-3 levels in patients with schizophrenia are significantly lower than in healthy subjects.13 Lithium also can inhibit caspase-3.
Do these proposals sound radical?
Most of the recommendations I’ve made here are not employed in the clinical practice of psychiatry. These ideas must be put to the test in controlled clinical trials.
The crux of my argument is that we need to think outside the “dopamine box” and focus on brain repair if we are to make progress in reversing, even preventing, neurodegeneration and clinical deterioration in this disabling brain syndrome. Just as cancer often is treated with rational polypharmacy, schizophrenia might need a similar approach. To vanquish schizophrenia—a goal that has eluded us—it is imperative to pursue radically novel and disruptive therapeutic strategies. The ideas I’ve listed here sound the call that the quest to repair the brain in schizophrenia must begin, and soon.
1. Agius N, Nandra, KS. Do atypical antipsychotics promote neurogenesis as a class effect? Psychiatr Danub. 2012;24(suppl 1):S191-S193.
2. Nasrallah HA, Hopkins T, Pixley SK. Differential effects of antipsychotic and antidepressant drugs on neurogenic regions in rats. Brain Res. 2010;1354:23-29.
3. Chiu CT, Wang Z, Hunsberger JG, et al. Therapeutic potential of mood stabilizers lithium and valproic acid: beyond bipolar disorder. Pharmacol Rev. 2013;65(1):105-142.
4. Parikh V, Khan MM, Terry A, et al. Differential effects of typical and atypical antipsychotics on nerve growth factor and choline acetyltransferase expression in the cortex and nucleus basalis of rats. J Psychiatr Res. 2004;38(5):521-529.
5. Monji A, Kato TA, Mizoguchi Y, et al. Neuro-inflammation in schizophrenia especially focused on the role of microglia. Prog Neuropsychopharmacol Biol Psychiatry. 2013;42:115-121.
6. Sommer IE, deWitte L, Begemann M, et al. Nonsteriodal anti-inflammatory drugs in schizophrenia: ready for practice or a good start? A meta-analysis. J Clin Psychiatry. 2012;73(4):414-419.
7. Bian Q, Kato T, Monji A, et al. The effect of atypical anti-psychotics perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
8. Berk M, Copolov D, Dean O, et al. N-acetyl cysteine as a glutathione precursor for schizophrenia—a double-blind, randomized, placebo-controlled trial. Biol Psychiatry. 2008;64(5):361-368.
9. Huang J, Fairbrother W, Reed JC, et al. Therapeutic targeting of Bcl-2 family for treatment of B-cell malignancies. Expert Rev Hematol. 2015;8(3):283-297.
10. Fernandez-Enright F, Andrews JL, Newell KA, et al. Novel implications of Lingo-1 and its signaling partners in schizophrenia. Transl Psychiatry. 2014; 4:e348.
11. Zink M, Correll CU. Glutamatergic agents for schizophrenia: current evidence and perspectives. Expert Rev Clin Pharmacol. 2015;8(3):335-352.
12. Kremer I, Vass A, Gorelik I, et al. Placebo-controlled trial of lamotrigine added to conventional and atypical antipsychotics in schizophrenia. Biol Psychiatry. 2004;56(6):444-446.
13. McEvoy J, Baillie RA, Zhu H, et al. Lipidomics reveals early metabolic changes in subjects with schizophrenia: effects of atypical antipsychotics. PLoS One. 2013;8(7):e68717.
1. Agius N, Nandra, KS. Do atypical antipsychotics promote neurogenesis as a class effect? Psychiatr Danub. 2012;24(suppl 1):S191-S193.
2. Nasrallah HA, Hopkins T, Pixley SK. Differential effects of antipsychotic and antidepressant drugs on neurogenic regions in rats. Brain Res. 2010;1354:23-29.
3. Chiu CT, Wang Z, Hunsberger JG, et al. Therapeutic potential of mood stabilizers lithium and valproic acid: beyond bipolar disorder. Pharmacol Rev. 2013;65(1):105-142.
4. Parikh V, Khan MM, Terry A, et al. Differential effects of typical and atypical antipsychotics on nerve growth factor and choline acetyltransferase expression in the cortex and nucleus basalis of rats. J Psychiatr Res. 2004;38(5):521-529.
5. Monji A, Kato TA, Mizoguchi Y, et al. Neuro-inflammation in schizophrenia especially focused on the role of microglia. Prog Neuropsychopharmacol Biol Psychiatry. 2013;42:115-121.
6. Sommer IE, deWitte L, Begemann M, et al. Nonsteriodal anti-inflammatory drugs in schizophrenia: ready for practice or a good start? A meta-analysis. J Clin Psychiatry. 2012;73(4):414-419.
7. Bian Q, Kato T, Monji A, et al. The effect of atypical anti-psychotics perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
8. Berk M, Copolov D, Dean O, et al. N-acetyl cysteine as a glutathione precursor for schizophrenia—a double-blind, randomized, placebo-controlled trial. Biol Psychiatry. 2008;64(5):361-368.
9. Huang J, Fairbrother W, Reed JC, et al. Therapeutic targeting of Bcl-2 family for treatment of B-cell malignancies. Expert Rev Hematol. 2015;8(3):283-297.
10. Fernandez-Enright F, Andrews JL, Newell KA, et al. Novel implications of Lingo-1 and its signaling partners in schizophrenia. Transl Psychiatry. 2014; 4:e348.
11. Zink M, Correll CU. Glutamatergic agents for schizophrenia: current evidence and perspectives. Expert Rev Clin Pharmacol. 2015;8(3):335-352.
12. Kremer I, Vass A, Gorelik I, et al. Placebo-controlled trial of lamotrigine added to conventional and atypical antipsychotics in schizophrenia. Biol Psychiatry. 2004;56(6):444-446.
13. McEvoy J, Baillie RA, Zhu H, et al. Lipidomics reveals early metabolic changes in subjects with schizophrenia: effects of atypical antipsychotics. PLoS One. 2013;8(7):e68717.
Do unto others...
In his most recent bestseller, Being Mortal, Dr. Atul Gawande has again raised social awareness of the inadequacies of our health care system in assisting patients with end-of-life decisions and care. In his treatise, he laments his own lack of training in medical school and residency regarding what he has emphasized should be a key component of any physician’s education.
To some degree, there has been a greater emphasis on palliative care training in our medical schools since Dr. Gawande graduated two decades or so ago. The subject of physician-patient communication, including those difficult discussions that should occur near the end of life, is now incorporated into most medical school curricula. Additionally, palliative care has become a respected and growing subspecialty within both medicine and surgery.* Despite these improvements, still far too many patients die while receiving futile end-of-life care in our nation’s intensive care units and hospital wards rather than in the comfort of their homes surrounded by loved ones. Although referrals have increased, too few patients are afforded the opportunity to utilize hospice care and, those that do, are often referred too late in the course of their terminal disease to obtain full benefit
Why are we not doing better? Two likely contributors include a physician mindset that only cure is success and death represents failure, and unrealistic expectations of patients as to what modern medicine can accomplish. A more fixable and probably more important factor is the failure of doctors to effectively communicate during these highly stressful circumstances. As emphasized by Gawande and from my own experience, the key to negotiating a sensible path in hopeless, end-of-life situations is frequent, reasonable, and realistic consultation with our patients.
Not only are the conversations usually difficult and demanding, but the choices of whether to pursue treatment or remove life-sustaining efforts are frequently not well-defined. While these challenging clinical scenarios are often painted as black and white in the lay press, any physician or surgeon who has cared for such patients realizes that there is a delicate and precarious balance between providing hope, administering appropriate aggressive treatment, and ensuring patient comfort. In a well-intentioned attempt to leave some remnant of hope, we physicians too frequently paint an unrealistic picture for our patients.
Advance patient directives have been promoted as one means for patients to avoid futile, uncomfortable, and unnecessary care during the last stage of their lives. Though I by no means wish to discourage these often useful legal documents, they should not be entered into naively. For example, aggressive life-sustaining care for a patient with extensive metastatic lung cancer is likely inappropriate. On the other hand, short-term ventilator assistance for an elderly unconscious person recovering from an automobile accident may result in many additional years of enjoyable and productive life. Patients need to understand that all grave clinical situations are not equal and that their advance directives should be flexible enough to cover a variety of circumstances.
It has been well established that most patients and families have selective hearing and understanding. Even when the details of a major operation with a greater likelihood of a negative rather than a positive outcome are carefully and clearly presented using lay language, the potential positive outcomes frequently push the more probable adverse consequences into hidden recesses of the brain. In my experience, the more desperate the situation, the more often it is that the possibility of an unsuccessful outcome will be masked or denied by patients or their family members. Even though in my practice I carefully explained the high probability of eventual recurrence when operating on patients with pancreatic cancer, many of them were surprised and some were even quite indignant when this disappointing consequence developed. In my opinion, the most effective means to avoid such misunderstandings is to always have the patient and/or family relate their comprehension of the just-completed conversation. It is then essential to re-emphasize the important details that they suppressed and pushed to the background from your initial explanation.
In these challenging end-of-life moments, what advice should we offer? One question that should almost never be asked of the patient or his/her representative is: “Would you like everything possible done?” Especially for a family member who may take on considerable guilt by answering in the negative, the response will nearly always be “yes” no matter how unlikely a successful outcome. Rather, I believe that recommending only reasonable options, including and possibly emphasizing the choice of comfort therapy alone despite the certainty of death, is our obligation. We should be cognizant of the fact that the decision made by the patient is often highly dependent on how the alternatives are presented by his/her doctor. After clearly presenting the therapeutic options and their likely consequences, it may be helpful to relate what you would do yourself for a loved one in the same circumstances.
As in many other aspects of our lives, a useful guidepost in these situations is the Golden Rule: “Do unto others as you would have done unto yourself.” Interestingly, most probably based on our intimate exposure to numerous unnecessarily complicated and uncomfortable deaths, there is evidence that we physicians choose to die differently than our patients. In a recent essay, Dr. Ken Murray presented data from the John Hopkins Precursors Study that suggested doctors are less likely than their patients to submit themselves to futile end-of-life care. (Murray K: Doctors really do die differently. Zocalopublicsquare.org; accessed March 29, 2015). The proof he presents is from a survey of graduates of Johns Hopkins School of Medicine between 1948 and 1964. It revealed that 65% of them had written their own advance directives in comparison to 20% for the public at large. In addition, only 10% of the graduates would opt for cardiopulmonary resuscitation if they were comatose, compared with 75% of the general population.
I suspect that most surgeons, desiring a dignified death for themselves, are not surprised by these statistics. Therefore, we owe it to our patients to be as compassionate and thoughtful in managing the last stage of their lives as we have traditionally been trained to do in earlier phases when cure was a realistic expectation.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
*Recognizing the importance of end-of-life issues in a surgeon’s education, in 2012 ACS Surgery News initiated a series of articles on various aspects of palliative care.
In his most recent bestseller, Being Mortal, Dr. Atul Gawande has again raised social awareness of the inadequacies of our health care system in assisting patients with end-of-life decisions and care. In his treatise, he laments his own lack of training in medical school and residency regarding what he has emphasized should be a key component of any physician’s education.
To some degree, there has been a greater emphasis on palliative care training in our medical schools since Dr. Gawande graduated two decades or so ago. The subject of physician-patient communication, including those difficult discussions that should occur near the end of life, is now incorporated into most medical school curricula. Additionally, palliative care has become a respected and growing subspecialty within both medicine and surgery.* Despite these improvements, still far too many patients die while receiving futile end-of-life care in our nation’s intensive care units and hospital wards rather than in the comfort of their homes surrounded by loved ones. Although referrals have increased, too few patients are afforded the opportunity to utilize hospice care and, those that do, are often referred too late in the course of their terminal disease to obtain full benefit
Why are we not doing better? Two likely contributors include a physician mindset that only cure is success and death represents failure, and unrealistic expectations of patients as to what modern medicine can accomplish. A more fixable and probably more important factor is the failure of doctors to effectively communicate during these highly stressful circumstances. As emphasized by Gawande and from my own experience, the key to negotiating a sensible path in hopeless, end-of-life situations is frequent, reasonable, and realistic consultation with our patients.
Not only are the conversations usually difficult and demanding, but the choices of whether to pursue treatment or remove life-sustaining efforts are frequently not well-defined. While these challenging clinical scenarios are often painted as black and white in the lay press, any physician or surgeon who has cared for such patients realizes that there is a delicate and precarious balance between providing hope, administering appropriate aggressive treatment, and ensuring patient comfort. In a well-intentioned attempt to leave some remnant of hope, we physicians too frequently paint an unrealistic picture for our patients.
Advance patient directives have been promoted as one means for patients to avoid futile, uncomfortable, and unnecessary care during the last stage of their lives. Though I by no means wish to discourage these often useful legal documents, they should not be entered into naively. For example, aggressive life-sustaining care for a patient with extensive metastatic lung cancer is likely inappropriate. On the other hand, short-term ventilator assistance for an elderly unconscious person recovering from an automobile accident may result in many additional years of enjoyable and productive life. Patients need to understand that all grave clinical situations are not equal and that their advance directives should be flexible enough to cover a variety of circumstances.
It has been well established that most patients and families have selective hearing and understanding. Even when the details of a major operation with a greater likelihood of a negative rather than a positive outcome are carefully and clearly presented using lay language, the potential positive outcomes frequently push the more probable adverse consequences into hidden recesses of the brain. In my experience, the more desperate the situation, the more often it is that the possibility of an unsuccessful outcome will be masked or denied by patients or their family members. Even though in my practice I carefully explained the high probability of eventual recurrence when operating on patients with pancreatic cancer, many of them were surprised and some were even quite indignant when this disappointing consequence developed. In my opinion, the most effective means to avoid such misunderstandings is to always have the patient and/or family relate their comprehension of the just-completed conversation. It is then essential to re-emphasize the important details that they suppressed and pushed to the background from your initial explanation.
In these challenging end-of-life moments, what advice should we offer? One question that should almost never be asked of the patient or his/her representative is: “Would you like everything possible done?” Especially for a family member who may take on considerable guilt by answering in the negative, the response will nearly always be “yes” no matter how unlikely a successful outcome. Rather, I believe that recommending only reasonable options, including and possibly emphasizing the choice of comfort therapy alone despite the certainty of death, is our obligation. We should be cognizant of the fact that the decision made by the patient is often highly dependent on how the alternatives are presented by his/her doctor. After clearly presenting the therapeutic options and their likely consequences, it may be helpful to relate what you would do yourself for a loved one in the same circumstances.
As in many other aspects of our lives, a useful guidepost in these situations is the Golden Rule: “Do unto others as you would have done unto yourself.” Interestingly, most probably based on our intimate exposure to numerous unnecessarily complicated and uncomfortable deaths, there is evidence that we physicians choose to die differently than our patients. In a recent essay, Dr. Ken Murray presented data from the John Hopkins Precursors Study that suggested doctors are less likely than their patients to submit themselves to futile end-of-life care. (Murray K: Doctors really do die differently. Zocalopublicsquare.org; accessed March 29, 2015). The proof he presents is from a survey of graduates of Johns Hopkins School of Medicine between 1948 and 1964. It revealed that 65% of them had written their own advance directives in comparison to 20% for the public at large. In addition, only 10% of the graduates would opt for cardiopulmonary resuscitation if they were comatose, compared with 75% of the general population.
I suspect that most surgeons, desiring a dignified death for themselves, are not surprised by these statistics. Therefore, we owe it to our patients to be as compassionate and thoughtful in managing the last stage of their lives as we have traditionally been trained to do in earlier phases when cure was a realistic expectation.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
*Recognizing the importance of end-of-life issues in a surgeon’s education, in 2012 ACS Surgery News initiated a series of articles on various aspects of palliative care.
In his most recent bestseller, Being Mortal, Dr. Atul Gawande has again raised social awareness of the inadequacies of our health care system in assisting patients with end-of-life decisions and care. In his treatise, he laments his own lack of training in medical school and residency regarding what he has emphasized should be a key component of any physician’s education.
To some degree, there has been a greater emphasis on palliative care training in our medical schools since Dr. Gawande graduated two decades or so ago. The subject of physician-patient communication, including those difficult discussions that should occur near the end of life, is now incorporated into most medical school curricula. Additionally, palliative care has become a respected and growing subspecialty within both medicine and surgery.* Despite these improvements, still far too many patients die while receiving futile end-of-life care in our nation’s intensive care units and hospital wards rather than in the comfort of their homes surrounded by loved ones. Although referrals have increased, too few patients are afforded the opportunity to utilize hospice care and, those that do, are often referred too late in the course of their terminal disease to obtain full benefit
Why are we not doing better? Two likely contributors include a physician mindset that only cure is success and death represents failure, and unrealistic expectations of patients as to what modern medicine can accomplish. A more fixable and probably more important factor is the failure of doctors to effectively communicate during these highly stressful circumstances. As emphasized by Gawande and from my own experience, the key to negotiating a sensible path in hopeless, end-of-life situations is frequent, reasonable, and realistic consultation with our patients.
Not only are the conversations usually difficult and demanding, but the choices of whether to pursue treatment or remove life-sustaining efforts are frequently not well-defined. While these challenging clinical scenarios are often painted as black and white in the lay press, any physician or surgeon who has cared for such patients realizes that there is a delicate and precarious balance between providing hope, administering appropriate aggressive treatment, and ensuring patient comfort. In a well-intentioned attempt to leave some remnant of hope, we physicians too frequently paint an unrealistic picture for our patients.
Advance patient directives have been promoted as one means for patients to avoid futile, uncomfortable, and unnecessary care during the last stage of their lives. Though I by no means wish to discourage these often useful legal documents, they should not be entered into naively. For example, aggressive life-sustaining care for a patient with extensive metastatic lung cancer is likely inappropriate. On the other hand, short-term ventilator assistance for an elderly unconscious person recovering from an automobile accident may result in many additional years of enjoyable and productive life. Patients need to understand that all grave clinical situations are not equal and that their advance directives should be flexible enough to cover a variety of circumstances.
It has been well established that most patients and families have selective hearing and understanding. Even when the details of a major operation with a greater likelihood of a negative rather than a positive outcome are carefully and clearly presented using lay language, the potential positive outcomes frequently push the more probable adverse consequences into hidden recesses of the brain. In my experience, the more desperate the situation, the more often it is that the possibility of an unsuccessful outcome will be masked or denied by patients or their family members. Even though in my practice I carefully explained the high probability of eventual recurrence when operating on patients with pancreatic cancer, many of them were surprised and some were even quite indignant when this disappointing consequence developed. In my opinion, the most effective means to avoid such misunderstandings is to always have the patient and/or family relate their comprehension of the just-completed conversation. It is then essential to re-emphasize the important details that they suppressed and pushed to the background from your initial explanation.
In these challenging end-of-life moments, what advice should we offer? One question that should almost never be asked of the patient or his/her representative is: “Would you like everything possible done?” Especially for a family member who may take on considerable guilt by answering in the negative, the response will nearly always be “yes” no matter how unlikely a successful outcome. Rather, I believe that recommending only reasonable options, including and possibly emphasizing the choice of comfort therapy alone despite the certainty of death, is our obligation. We should be cognizant of the fact that the decision made by the patient is often highly dependent on how the alternatives are presented by his/her doctor. After clearly presenting the therapeutic options and their likely consequences, it may be helpful to relate what you would do yourself for a loved one in the same circumstances.
As in many other aspects of our lives, a useful guidepost in these situations is the Golden Rule: “Do unto others as you would have done unto yourself.” Interestingly, most probably based on our intimate exposure to numerous unnecessarily complicated and uncomfortable deaths, there is evidence that we physicians choose to die differently than our patients. In a recent essay, Dr. Ken Murray presented data from the John Hopkins Precursors Study that suggested doctors are less likely than their patients to submit themselves to futile end-of-life care. (Murray K: Doctors really do die differently. Zocalopublicsquare.org; accessed March 29, 2015). The proof he presents is from a survey of graduates of Johns Hopkins School of Medicine between 1948 and 1964. It revealed that 65% of them had written their own advance directives in comparison to 20% for the public at large. In addition, only 10% of the graduates would opt for cardiopulmonary resuscitation if they were comatose, compared with 75% of the general population.
I suspect that most surgeons, desiring a dignified death for themselves, are not surprised by these statistics. Therefore, we owe it to our patients to be as compassionate and thoughtful in managing the last stage of their lives as we have traditionally been trained to do in earlier phases when cure was a realistic expectation.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
*Recognizing the importance of end-of-life issues in a surgeon’s education, in 2012 ACS Surgery News initiated a series of articles on various aspects of palliative care.