User login
How to fix clinical trial accrual
Editor Jame Abraham argues that once there is appropriate clinical trial infrastructure, there is only one person who can increase the clinical trial accrual: the treating oncologist.
Click on the PDF icon at the top of this introduction to read the full article.
Editor Jame Abraham argues that once there is appropriate clinical trial infrastructure, there is only one person who can increase the clinical trial accrual: the treating oncologist.
Click on the PDF icon at the top of this introduction to read the full article.
Editor Jame Abraham argues that once there is appropriate clinical trial infrastructure, there is only one person who can increase the clinical trial accrual: the treating oncologist.
Click on the PDF icon at the top of this introduction to read the full article.
Your College: A Remarkable Organization
Peering out from 28 floors above the busy, early morning streets of downtown Chicago, I was entranced by the view. The rising sun in the east created a shimmering, iridescent play of light on the waters of Lake Michigan that extended as far as the eye could see. The room I was in also commanded my attention. Several rows of desks, each with a computer screen, faced a single elevated line of chairs for the leaders of the deliberations that were about to take place. Above this row on the front wall of this imposing room is emblazoned the seal of our College with its mission statement, “The ACS is dedicated to improving the care of the surgical patient and to safeguarding standards of care in an optimal and ethical practice environment,” to remind those in the room of the ultimate purpose in serving this professional organization. So the Regents room and view appeared to me, a newcomer to these meetings of the leadership of the American College of Surgeons.
I hope I can convince you in the paragraphs that follow that this mission and its execution by Regents, Governors, and Fellows of the ACS, are every bit as noble as the architecture of the room and the view it affords.
I have been a Fellow of the American College of Surgeons since 1980. My membership in this extraordinary association has provided me many benefits and numerous opportunities. In the early years, I significantly expanded the knowledge base I had gained in residency by attending every fall Clinical Congress and consuming as many educational offerings as time would permit during this nearly week-long learning marathon. After a few years, I was granted the privilege of being on the instructor end of several of these educational exchanges. At that time in my career, it appeared to me that the ACS’s main and almost sole purpose was to provide continuing education for surgeons who attended the annual Clinical Congress. I have subsequently found that it is so much more.
In 2005, I was invited to represent one of my specialist surgical societies as a Governor. During my 6-year term, I came to realize that the key purpose of the Board of Governors (BOG) is to provide an effective communication conduit between the Fellows and the sole policy-making body of the ACS, the Board of Regents (BOR). In recent years, most of the Regents have attended the annual BOG meeting in order to facilitate this interaction.
In 2012, I was elected First Vice-President of the ACS. Now as an officer of the College, I was invited for 2 years to attend all BOR meetings and to be in the mainstream of all communications relating to strategy and policy. These opportunities provided me with an intimate, inside look at how this large organization of nearly 80,000 members functions to serve the entire surgical profession including each of its many specialties. What I learned about the internal workings of the leadership and those who dedicate their time to this work has reinforced my own commitment to the ACS. It is a remarkable organization.
This brings us back to the well-designed and impressive Regents’ room high above Chicago. The hum of numerous disjointed conversations ceased as the Chair of the ACS Board of Regents called the June 2012 meeting to order. My attention is now focused on the proceedings rather than on the beauty of Lake Michigan below. Although every surgical specialty is represented among the 22 Regents, all discussion was invariably directed toward the betterment of the surgical profession as a whole rather than about any specific specialty’s interests.
The Regents are dedicated servants of the ACS. In addition to three one-and-a-half day meetings annually, each of which requires hours of reading in preparation, most of the Regents serve on at least two committees of the Board. Regents are nominated by Fellows, advisory councils, and committees, and are elected by the much larger BOG which represents every state and Canadian province, several countries, and many surgery specialist societies. In addition to assuring that all surgical specialties are represented, bylaws of the ACS state that the President of the ACS and two Canadian Fellows must be among the BOR membership. Based on my 2-year experience, the BOG has exhibited considerable wisdom in their choice of Regents.
The officers of the ACS (President-Elect, First and Second Vice-Presidents and Vice-Presidents-Elect, Secretary, Treasurer), and the officers of the BOG (Chair, Vice-Chair, and Secretary/Treasurer) attend all BOR meetings and serve in an advisory capacity. Also in attendance and providing essential input are executive members of the ACS staff and, representing the younger ACS membership, the chairs of the Resident Associates Society (RAS) and the Young Fellows Association (YAF). Although only Regents can vote and are therefore responsible for establishing ACS policy, I discovered they welcome participation from all in attendance. I always felt that my contributions and those of other non-voting attendees were thoughtfully and carefully considered.
Much of the preparatory work for BOR meetings is done in the committees that meet just prior to the full Board meeting. I had the pleasure of being on the Honors Committee that selects Honorary Fellows of the ACS from regions throughout the world and selects Fellows for special awards such as the Distinguished Service Award, and on the Members Services Liaison Committee that concentrates on expanding ACS membership and on more fully informing the Fellows of BOR activities. Among several other important committees are the Central Judiciary Committee that is responsible for disciplining Fellows who breach the ethical standards of our College and the Finance Committee that assures responsible fiscal stewardship of the ACS. Deliberations of all of the committees are brought before the full Board for final approval
Although the BOR has been the policy-making body since the founding of the ACS 102 years ago, the structure of our society has evolved considerably, especially during the past 2 decades. The ACS is organized around five major Divisions: Advocacy and Health Policy, Education, Integrated Communications, Member Services, and Research and Optimal Patient Care. The Directors of these Divisions report on a regular basis to the BOR to keep the Regents’ knowledge up-to-date and to assist them in determining the strategic direction of the ACS. Much of the discussion, modifications, and innovations center around these Divisions, also represented as pillars in the recent BOG re-organization. I trust you are aware of the many achievements that have resulted: NSQIP, legislative elimination of the flawed Sustainable Growth Rate (SGR) formula, reorganization of the Clinical Congress, and a re-emphasis on global surgery and the Operation Giving Back Program to name but a few.
Finally, a key role of the BOR is to select the Executive Director of the ACS who manages the day-to-day operations of the College with the Board’s strategic guidance. The ACS has been blessed with a number of excellent Directors, none more visionary and competent than the present Director, David Hoyt, MD, FACS, who is 1 year into his second 5-year term.
I hope that this discussion provides you with a better understanding of the role and functioning of the BOR and the College of which you are a member. The grandeur of the BOR room appropriately parallels the excellence of what takes place within it.
Take time to visit the next time you are in Chicago. I am certain the ACS staff would be pleased and proud to meet you, show you around, and have you experience what I have tried to describe in this brief discourse.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
Peering out from 28 floors above the busy, early morning streets of downtown Chicago, I was entranced by the view. The rising sun in the east created a shimmering, iridescent play of light on the waters of Lake Michigan that extended as far as the eye could see. The room I was in also commanded my attention. Several rows of desks, each with a computer screen, faced a single elevated line of chairs for the leaders of the deliberations that were about to take place. Above this row on the front wall of this imposing room is emblazoned the seal of our College with its mission statement, “The ACS is dedicated to improving the care of the surgical patient and to safeguarding standards of care in an optimal and ethical practice environment,” to remind those in the room of the ultimate purpose in serving this professional organization. So the Regents room and view appeared to me, a newcomer to these meetings of the leadership of the American College of Surgeons.
I hope I can convince you in the paragraphs that follow that this mission and its execution by Regents, Governors, and Fellows of the ACS, are every bit as noble as the architecture of the room and the view it affords.
I have been a Fellow of the American College of Surgeons since 1980. My membership in this extraordinary association has provided me many benefits and numerous opportunities. In the early years, I significantly expanded the knowledge base I had gained in residency by attending every fall Clinical Congress and consuming as many educational offerings as time would permit during this nearly week-long learning marathon. After a few years, I was granted the privilege of being on the instructor end of several of these educational exchanges. At that time in my career, it appeared to me that the ACS’s main and almost sole purpose was to provide continuing education for surgeons who attended the annual Clinical Congress. I have subsequently found that it is so much more.
In 2005, I was invited to represent one of my specialist surgical societies as a Governor. During my 6-year term, I came to realize that the key purpose of the Board of Governors (BOG) is to provide an effective communication conduit between the Fellows and the sole policy-making body of the ACS, the Board of Regents (BOR). In recent years, most of the Regents have attended the annual BOG meeting in order to facilitate this interaction.
In 2012, I was elected First Vice-President of the ACS. Now as an officer of the College, I was invited for 2 years to attend all BOR meetings and to be in the mainstream of all communications relating to strategy and policy. These opportunities provided me with an intimate, inside look at how this large organization of nearly 80,000 members functions to serve the entire surgical profession including each of its many specialties. What I learned about the internal workings of the leadership and those who dedicate their time to this work has reinforced my own commitment to the ACS. It is a remarkable organization.
This brings us back to the well-designed and impressive Regents’ room high above Chicago. The hum of numerous disjointed conversations ceased as the Chair of the ACS Board of Regents called the June 2012 meeting to order. My attention is now focused on the proceedings rather than on the beauty of Lake Michigan below. Although every surgical specialty is represented among the 22 Regents, all discussion was invariably directed toward the betterment of the surgical profession as a whole rather than about any specific specialty’s interests.
The Regents are dedicated servants of the ACS. In addition to three one-and-a-half day meetings annually, each of which requires hours of reading in preparation, most of the Regents serve on at least two committees of the Board. Regents are nominated by Fellows, advisory councils, and committees, and are elected by the much larger BOG which represents every state and Canadian province, several countries, and many surgery specialist societies. In addition to assuring that all surgical specialties are represented, bylaws of the ACS state that the President of the ACS and two Canadian Fellows must be among the BOR membership. Based on my 2-year experience, the BOG has exhibited considerable wisdom in their choice of Regents.
The officers of the ACS (President-Elect, First and Second Vice-Presidents and Vice-Presidents-Elect, Secretary, Treasurer), and the officers of the BOG (Chair, Vice-Chair, and Secretary/Treasurer) attend all BOR meetings and serve in an advisory capacity. Also in attendance and providing essential input are executive members of the ACS staff and, representing the younger ACS membership, the chairs of the Resident Associates Society (RAS) and the Young Fellows Association (YAF). Although only Regents can vote and are therefore responsible for establishing ACS policy, I discovered they welcome participation from all in attendance. I always felt that my contributions and those of other non-voting attendees were thoughtfully and carefully considered.
Much of the preparatory work for BOR meetings is done in the committees that meet just prior to the full Board meeting. I had the pleasure of being on the Honors Committee that selects Honorary Fellows of the ACS from regions throughout the world and selects Fellows for special awards such as the Distinguished Service Award, and on the Members Services Liaison Committee that concentrates on expanding ACS membership and on more fully informing the Fellows of BOR activities. Among several other important committees are the Central Judiciary Committee that is responsible for disciplining Fellows who breach the ethical standards of our College and the Finance Committee that assures responsible fiscal stewardship of the ACS. Deliberations of all of the committees are brought before the full Board for final approval
Although the BOR has been the policy-making body since the founding of the ACS 102 years ago, the structure of our society has evolved considerably, especially during the past 2 decades. The ACS is organized around five major Divisions: Advocacy and Health Policy, Education, Integrated Communications, Member Services, and Research and Optimal Patient Care. The Directors of these Divisions report on a regular basis to the BOR to keep the Regents’ knowledge up-to-date and to assist them in determining the strategic direction of the ACS. Much of the discussion, modifications, and innovations center around these Divisions, also represented as pillars in the recent BOG re-organization. I trust you are aware of the many achievements that have resulted: NSQIP, legislative elimination of the flawed Sustainable Growth Rate (SGR) formula, reorganization of the Clinical Congress, and a re-emphasis on global surgery and the Operation Giving Back Program to name but a few.
Finally, a key role of the BOR is to select the Executive Director of the ACS who manages the day-to-day operations of the College with the Board’s strategic guidance. The ACS has been blessed with a number of excellent Directors, none more visionary and competent than the present Director, David Hoyt, MD, FACS, who is 1 year into his second 5-year term.
I hope that this discussion provides you with a better understanding of the role and functioning of the BOR and the College of which you are a member. The grandeur of the BOR room appropriately parallels the excellence of what takes place within it.
Take time to visit the next time you are in Chicago. I am certain the ACS staff would be pleased and proud to meet you, show you around, and have you experience what I have tried to describe in this brief discourse.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
Peering out from 28 floors above the busy, early morning streets of downtown Chicago, I was entranced by the view. The rising sun in the east created a shimmering, iridescent play of light on the waters of Lake Michigan that extended as far as the eye could see. The room I was in also commanded my attention. Several rows of desks, each with a computer screen, faced a single elevated line of chairs for the leaders of the deliberations that were about to take place. Above this row on the front wall of this imposing room is emblazoned the seal of our College with its mission statement, “The ACS is dedicated to improving the care of the surgical patient and to safeguarding standards of care in an optimal and ethical practice environment,” to remind those in the room of the ultimate purpose in serving this professional organization. So the Regents room and view appeared to me, a newcomer to these meetings of the leadership of the American College of Surgeons.
I hope I can convince you in the paragraphs that follow that this mission and its execution by Regents, Governors, and Fellows of the ACS, are every bit as noble as the architecture of the room and the view it affords.
I have been a Fellow of the American College of Surgeons since 1980. My membership in this extraordinary association has provided me many benefits and numerous opportunities. In the early years, I significantly expanded the knowledge base I had gained in residency by attending every fall Clinical Congress and consuming as many educational offerings as time would permit during this nearly week-long learning marathon. After a few years, I was granted the privilege of being on the instructor end of several of these educational exchanges. At that time in my career, it appeared to me that the ACS’s main and almost sole purpose was to provide continuing education for surgeons who attended the annual Clinical Congress. I have subsequently found that it is so much more.
In 2005, I was invited to represent one of my specialist surgical societies as a Governor. During my 6-year term, I came to realize that the key purpose of the Board of Governors (BOG) is to provide an effective communication conduit between the Fellows and the sole policy-making body of the ACS, the Board of Regents (BOR). In recent years, most of the Regents have attended the annual BOG meeting in order to facilitate this interaction.
In 2012, I was elected First Vice-President of the ACS. Now as an officer of the College, I was invited for 2 years to attend all BOR meetings and to be in the mainstream of all communications relating to strategy and policy. These opportunities provided me with an intimate, inside look at how this large organization of nearly 80,000 members functions to serve the entire surgical profession including each of its many specialties. What I learned about the internal workings of the leadership and those who dedicate their time to this work has reinforced my own commitment to the ACS. It is a remarkable organization.
This brings us back to the well-designed and impressive Regents’ room high above Chicago. The hum of numerous disjointed conversations ceased as the Chair of the ACS Board of Regents called the June 2012 meeting to order. My attention is now focused on the proceedings rather than on the beauty of Lake Michigan below. Although every surgical specialty is represented among the 22 Regents, all discussion was invariably directed toward the betterment of the surgical profession as a whole rather than about any specific specialty’s interests.
The Regents are dedicated servants of the ACS. In addition to three one-and-a-half day meetings annually, each of which requires hours of reading in preparation, most of the Regents serve on at least two committees of the Board. Regents are nominated by Fellows, advisory councils, and committees, and are elected by the much larger BOG which represents every state and Canadian province, several countries, and many surgery specialist societies. In addition to assuring that all surgical specialties are represented, bylaws of the ACS state that the President of the ACS and two Canadian Fellows must be among the BOR membership. Based on my 2-year experience, the BOG has exhibited considerable wisdom in their choice of Regents.
The officers of the ACS (President-Elect, First and Second Vice-Presidents and Vice-Presidents-Elect, Secretary, Treasurer), and the officers of the BOG (Chair, Vice-Chair, and Secretary/Treasurer) attend all BOR meetings and serve in an advisory capacity. Also in attendance and providing essential input are executive members of the ACS staff and, representing the younger ACS membership, the chairs of the Resident Associates Society (RAS) and the Young Fellows Association (YAF). Although only Regents can vote and are therefore responsible for establishing ACS policy, I discovered they welcome participation from all in attendance. I always felt that my contributions and those of other non-voting attendees were thoughtfully and carefully considered.
Much of the preparatory work for BOR meetings is done in the committees that meet just prior to the full Board meeting. I had the pleasure of being on the Honors Committee that selects Honorary Fellows of the ACS from regions throughout the world and selects Fellows for special awards such as the Distinguished Service Award, and on the Members Services Liaison Committee that concentrates on expanding ACS membership and on more fully informing the Fellows of BOR activities. Among several other important committees are the Central Judiciary Committee that is responsible for disciplining Fellows who breach the ethical standards of our College and the Finance Committee that assures responsible fiscal stewardship of the ACS. Deliberations of all of the committees are brought before the full Board for final approval
Although the BOR has been the policy-making body since the founding of the ACS 102 years ago, the structure of our society has evolved considerably, especially during the past 2 decades. The ACS is organized around five major Divisions: Advocacy and Health Policy, Education, Integrated Communications, Member Services, and Research and Optimal Patient Care. The Directors of these Divisions report on a regular basis to the BOR to keep the Regents’ knowledge up-to-date and to assist them in determining the strategic direction of the ACS. Much of the discussion, modifications, and innovations center around these Divisions, also represented as pillars in the recent BOG re-organization. I trust you are aware of the many achievements that have resulted: NSQIP, legislative elimination of the flawed Sustainable Growth Rate (SGR) formula, reorganization of the Clinical Congress, and a re-emphasis on global surgery and the Operation Giving Back Program to name but a few.
Finally, a key role of the BOR is to select the Executive Director of the ACS who manages the day-to-day operations of the College with the Board’s strategic guidance. The ACS has been blessed with a number of excellent Directors, none more visionary and competent than the present Director, David Hoyt, MD, FACS, who is 1 year into his second 5-year term.
I hope that this discussion provides you with a better understanding of the role and functioning of the BOR and the College of which you are a member. The grandeur of the BOR room appropriately parallels the excellence of what takes place within it.
Take time to visit the next time you are in Chicago. I am certain the ACS staff would be pleased and proud to meet you, show you around, and have you experience what I have tried to describe in this brief discourse.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
Reflux redux
Symptoms compatible with gastroesophageal reflux disease (GERD) are incredibly prevalent. The typical ones are common, and the atypical ones are so often attributed to GERD that they too are extremely common. It seems that few patients in my clinic are not taking a proton pump inhibitor (PPI).
Drs. Alzubaidi and Gabbard, in their review of GERD in this issue, note that up to 40% of people experience symptoms of GERD at least once monthly. Since these symptoms can be intermittent, diagnosis poses a problem when the diagnostic algorithm includes a trial of a PPI. It is sometimes unclear whether PPI therapy relieved the symptoms or whether the symptoms abated for other reasons. I suspect that many patients remain on PPI therapy longer than needed (and often longer than initially intended) because of a false sense of improvement and continued need. When patients are diagnosed on clinical grounds, we need to intermittently reassess the continued need for PPI therapy. The authors discuss and place in reasonable perspective a few of the potential complications of chronic PPI use, but not the effects on absorption of iron, calcium, and micronutrients, or PPI-associated gastric polyposis. These can be clinically significant in some patients.
I believe that some atypical symptoms such as cough and hoarseness are overly attributed to GERD, so that PPI therapy is started, continued, and escalated due to premature closure of the diagnosis. I believe that the diagnosis should be reassessed at least once with observed withdrawal of PPI therapy in patients who did not have a firm physiologic diagnosis. Asking the patient to keep a symptom diary may help.
Lack of a significant response to PPI therapy should cast doubt on the diagnosis of GERD and warrant exploration for an alternative cause of the symptoms (eg, eosinophilic esophagitis, bile reflux, sinus disease, dysmotility). The possibility that the patient was not given an optimal trial of a PPI must also be considered: eg, the dose may have been inadequate, the timing of administration may have been suboptimal (not preprandial), or the patient may have been taking over-the-counter NSAIDs.
GERD is so prevalent in the general population that we must train ourselves to consider the possibility that, even if totally relieved by PPI therapy, the symptoms might be associated with aggravating comorbid conditions such as obstructive sleep apnea, Raynaud phenomenon, drugs that can decrease the tone of the lower esophageal sphincter, or even scleroderma.
Finally, in patients who have had a less-than-total response to full-dose PPI therapy and have had other diagnoses excluded, we shouldn’t forget the value of adding appropriately timed histamine 2 receptor antagonist therapy (and asking the patient about use of medications that can exacerbate symptoms).
Even the diseases we deal with every day sometimes warrant a second look.
Symptoms compatible with gastroesophageal reflux disease (GERD) are incredibly prevalent. The typical ones are common, and the atypical ones are so often attributed to GERD that they too are extremely common. It seems that few patients in my clinic are not taking a proton pump inhibitor (PPI).
Drs. Alzubaidi and Gabbard, in their review of GERD in this issue, note that up to 40% of people experience symptoms of GERD at least once monthly. Since these symptoms can be intermittent, diagnosis poses a problem when the diagnostic algorithm includes a trial of a PPI. It is sometimes unclear whether PPI therapy relieved the symptoms or whether the symptoms abated for other reasons. I suspect that many patients remain on PPI therapy longer than needed (and often longer than initially intended) because of a false sense of improvement and continued need. When patients are diagnosed on clinical grounds, we need to intermittently reassess the continued need for PPI therapy. The authors discuss and place in reasonable perspective a few of the potential complications of chronic PPI use, but not the effects on absorption of iron, calcium, and micronutrients, or PPI-associated gastric polyposis. These can be clinically significant in some patients.
I believe that some atypical symptoms such as cough and hoarseness are overly attributed to GERD, so that PPI therapy is started, continued, and escalated due to premature closure of the diagnosis. I believe that the diagnosis should be reassessed at least once with observed withdrawal of PPI therapy in patients who did not have a firm physiologic diagnosis. Asking the patient to keep a symptom diary may help.
Lack of a significant response to PPI therapy should cast doubt on the diagnosis of GERD and warrant exploration for an alternative cause of the symptoms (eg, eosinophilic esophagitis, bile reflux, sinus disease, dysmotility). The possibility that the patient was not given an optimal trial of a PPI must also be considered: eg, the dose may have been inadequate, the timing of administration may have been suboptimal (not preprandial), or the patient may have been taking over-the-counter NSAIDs.
GERD is so prevalent in the general population that we must train ourselves to consider the possibility that, even if totally relieved by PPI therapy, the symptoms might be associated with aggravating comorbid conditions such as obstructive sleep apnea, Raynaud phenomenon, drugs that can decrease the tone of the lower esophageal sphincter, or even scleroderma.
Finally, in patients who have had a less-than-total response to full-dose PPI therapy and have had other diagnoses excluded, we shouldn’t forget the value of adding appropriately timed histamine 2 receptor antagonist therapy (and asking the patient about use of medications that can exacerbate symptoms).
Even the diseases we deal with every day sometimes warrant a second look.
Symptoms compatible with gastroesophageal reflux disease (GERD) are incredibly prevalent. The typical ones are common, and the atypical ones are so often attributed to GERD that they too are extremely common. It seems that few patients in my clinic are not taking a proton pump inhibitor (PPI).
Drs. Alzubaidi and Gabbard, in their review of GERD in this issue, note that up to 40% of people experience symptoms of GERD at least once monthly. Since these symptoms can be intermittent, diagnosis poses a problem when the diagnostic algorithm includes a trial of a PPI. It is sometimes unclear whether PPI therapy relieved the symptoms or whether the symptoms abated for other reasons. I suspect that many patients remain on PPI therapy longer than needed (and often longer than initially intended) because of a false sense of improvement and continued need. When patients are diagnosed on clinical grounds, we need to intermittently reassess the continued need for PPI therapy. The authors discuss and place in reasonable perspective a few of the potential complications of chronic PPI use, but not the effects on absorption of iron, calcium, and micronutrients, or PPI-associated gastric polyposis. These can be clinically significant in some patients.
I believe that some atypical symptoms such as cough and hoarseness are overly attributed to GERD, so that PPI therapy is started, continued, and escalated due to premature closure of the diagnosis. I believe that the diagnosis should be reassessed at least once with observed withdrawal of PPI therapy in patients who did not have a firm physiologic diagnosis. Asking the patient to keep a symptom diary may help.
Lack of a significant response to PPI therapy should cast doubt on the diagnosis of GERD and warrant exploration for an alternative cause of the symptoms (eg, eosinophilic esophagitis, bile reflux, sinus disease, dysmotility). The possibility that the patient was not given an optimal trial of a PPI must also be considered: eg, the dose may have been inadequate, the timing of administration may have been suboptimal (not preprandial), or the patient may have been taking over-the-counter NSAIDs.
GERD is so prevalent in the general population that we must train ourselves to consider the possibility that, even if totally relieved by PPI therapy, the symptoms might be associated with aggravating comorbid conditions such as obstructive sleep apnea, Raynaud phenomenon, drugs that can decrease the tone of the lower esophageal sphincter, or even scleroderma.
Finally, in patients who have had a less-than-total response to full-dose PPI therapy and have had other diagnoses excluded, we shouldn’t forget the value of adding appropriately timed histamine 2 receptor antagonist therapy (and asking the patient about use of medications that can exacerbate symptoms).
Even the diseases we deal with every day sometimes warrant a second look.
Does hormone therapy reduce mortality in recently menopausal women?
Clinicians work to maximize the quality of life and longevity of every patient. For women with moderate to severe menopausal symptoms, oral estrogen therapy can improve quality of life, but at the cost of significant adverse effects. The Women’s Health Initiative (WHI) reported that for postmenopausal women with a uterus, conjugated estrogen plus medroxyprogesterone acetate (CEE+MPA) hormone therapy (HT) versus placebo significantly increased the risk of cardiovascular events (relative risk [RR], 1.13), breast cancer (RR, 1.24), stroke (RR, 1.37), deep vein thrombosis (RR, 1.87), and pulmonary embolism (RR, 1.98).1 In postmeno pausal women without a uterus, CEE HT did not increase the risk of breast cancer (RR, 0.79), compared with placebo, but it did significantly in crease the risk of cardiovascular events (RR, 1.11), stroke (RR, 1.35), deep vein thrombosis (RR, 1.48), and pulmonary embolism (RR, 1.35).1
Clinicians prescribing estrogen must individualize therapy according to its benefits and risks. An important issue that has received insufficient at tention is, “What is the effect of HT on mortality in recently menopausal women?” Here, I examine this issue.
HT reduces mortality in recently menopausal women
Pooling the results of the WHI CEE+MPA and CEE-only trials reveals that there were 70 deaths in the HT-treated groups and 98 deaths in the placebo groups among women aged 50 to 59 years.1 With 4,706 and 4,259 women alive at the conclusion of the study in the HT and placebo groups, respectively, the women in the placebo group had significantly more deaths than the women in the HT-treated groups (Fisher exact test, P = .0194, χ2 test with Yates correction, P = .0226).
Using pooled data from the WHI, the RR of death in the HT versus placebo group was estimated at 0.70 (95% confidence interval [CI], 0.51−0.96), representing approximately 5 fewer deaths per 1,000 women per 5 years of therapy.2 In women aged 60 to 69 years and 70 to 79 years there were no significant differences in death rates between the HT- and placebo-treated women.
My interpretation of these results is that HT likely is associated with a reduced risk of death in recently menopausal women, but not in women distant from menopause onset.
Cochrane review of HT and mortality
Consistent with the WHI findings, authors of a recent Cochrane meta-analysis of 19 randomized trials including 40,410 menopausal women reported that HT significantly increased the risk of stroke (RR, 1.24; 95% CI, 1.10−1.41), venous thromboembolism (RR, 1.92; 95% CI, 1.36−2.69), and pulmonary emboli (RR, 1.81; 95% CI, 1.32−2.48).3 However, among women treated with oral HT within 10 years after the start of menopause, there was a reduced risk of coronary heart disease (RR, 0.52; 95% CI, 0.29−0.96). Using data from 5 clinical trials, the Cochrane meta-analysis researchers reported that, compared with placebo, HT reduced mortality (RR, 0.70; 95% CI, 0.52−0.95).3
Results of the Cochrane meta-analysis are consistent with those of a previous meta-analysis of 19 randomized trials involving 16,000 women. In this analysis, investigators found a reduced risk of death in recently menopausal women treated with hormone therapy (RR, 0.73; 95% CI, 0.52−0.96).4
Early menopause, HT, and mortality
Authors of multiple large epidemiologic studies have reported that early menopause is associated with an increased risk of death if HT is not initiated.5−7 For example, results of a study of women in Olmsted County, Minnesota, conducted from 1950 to 1987, indicated that, for women younger than age 45 years who underwent bilateral oophorectomy, the risk of death was increased among those who did not initiate HT, compared with women who did not undergo oophorectomy (hazard ratio [HR], 1.84; 95% CI, 1.27−2.68; P = .001).7
By contrast, women younger than 45 years who underwent bilateral oophorectomy and initiated estrogen therapy did not have an increased risk of death compared with women who did not undergo oophorectomy (HR, 0.65; 95% CI, 0.30−1.41; P = .28).7 An excess number of cardiovascular events appeared to account for the increased mortality among women with early surgical menopause who did not initiate HT.
The “timing hypothesis” proposes that the initiation of HT soon after the onset of menopause is associated with beneficial cardiovascular effects, but initiation more than 10 years after the onset of menopause is not associated with beneficial cardiovascular effects. The timing hypothesis is supported by the finding that, in recently menopausal women, HT is associated with reduced carotid intima-media thickness (CIMT), compared with placebo.8 Greater CIMT thickness is associated with an increased risk of cardiovascular events.
In my experience, few primary care clinicians are aware of these data. Often, these clinicians over-emphasize the risks and withhold HT in this vulnerable group of women.
HT: Minimizing the risks of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer
Results of multiple studies have shown that certain HT regimens increase the risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer. Is it possible to prescribe HT in a way that reduces these risks?
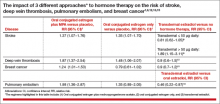
Results of observational studies indicate that, compared with oral estrogen therapy, transdermal HT is associated with a lower risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer (TABLE).9−15
Reducing the risk of stroke caused by HT is an important goal. In a study of 15,710 women who had stroke and 59,958 control women aged 50 to 79 years, transdermal estradiol at a dose of 50 µg or less daily was not associated with an increased risk of stroke, compared with HT nonuse (rate ratio, 0.81; 95% CI, 0.62−1.05).9 Compared with HT nonuse, the use of oral estrogen (rate ratio, 1.28; 95% CI, 1.15−1.42) or transdermal estradiol 50 µg or greater daily (rate ratio, 1.89; 95% CI, 1.15−3.11) was associated with an increased risk of stroke.9
Reducing the risks of deep venous thromboembolism (VTE) and pulmonary embolism caused by HT is an important goal. In a meta-analysis of the risk of VTE with HT, compared with nonusers, oral estrogen therapy was associated with a significantly increased risk of VTE (odds ratio [OR], 2.5; 95% CI, 1.9−3.4). Compared with nonuse, transdermal estrogen therapy was not associated with an increased risk of VTE (OR, 1.2; 95% CI, 0.9−1.7).11 In a study comparing oral versus transdermal estradiol, transdermal estradiol was associated with a reduced risk of pulmonary embolism (0.46 [95% CI, 0.22−0.97]).13
Reducing the risk of breast cancer caused by HT is an important goal. Results of one study showed that the combination of oral estrogen plus synthetic progestin was associated with an increased risk of breast cancer, compared with nonuse (RR, 1.5; 95% CI, 1.1−1.9). By contrast, the combination of transdermal estradiol plus micronized progesterone was not associated with an increased risk of breast cancer, compared with nonuse (RR, 0.9; 95% CI, 0.7−1.2).15
The bottom line
In recently menopausal women with moderate to severe hot flashes, HT improves quality of life and appears to decrease mortality. However, HT with oral estrogen plus synthetic progestin is associated with an increased risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer. Compared with oral estrogen, transdermal estradiol treatment is associated with a lower risk of stroke, deep vein thrombosis, and pulmonary embolism. Compared with oral estrogen plus a synthetic progestin, transdermal estradiol plus micronized progesterone is associated with a lower risk of breast cancer. The benefits of HT are likely maximized by initiating therapy in the perimenopause transition or early in the postmenopause, and the risks are minimized by using transdermal estradiol.16−18
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended post-stopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310(13):1353−1368.
- Santen RJ, Allred DC, Ardoin SP, et al. J Clin Endocrinol Metab. 2010;95(suppl 1):S1−S66.
- Boardman HM, Hartley L, Eisinga A, et al. Hormone therapy for preventing cardiovascular disease in postmenopausal women. Cochrane Database Syst Rev. 2015;3:CD002229.
- Salpeter SR, Cheng J, Thabane L, Buckley NS, Salpeter EE. Bayesian meta-analysis of hormone therapy and mortality in younger post-menopausal women. Am J Med. 2009;122(11):1016−1022.
- Gordon T, Kannel WB, Hjortland MC, McNamara PM. Menopause and coronary heart disease: The Framingham Study. Ann Intern Med. 1978;89(2):157−161.
- Stampfer MJ, Colditz GA, Willet WC, et al. Postmenopausal estrogen therapy and cardiovascular disease. Ten-year follow-up from the Nurses Health Study. N Engl J Med. 1991;325(11):756−762.
- Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16(1):15−23.
- Hodis HN, Mack WJ, Shoupe D, et al. Testing the menopausal hormone therapy timing hypothesis: the early versus late intervention trial with estradiol [abstract 13283]. American Heart Association Meeting 2014. Circulation. 2014;130:A13283.
- Renoux C, Dell’Aniello S, Garbe E, Suissa S. Transdermal and oral hormone replacement therapy and the risk of stroke: a nested case-control study. BMJ. 2010;340:c2519
- Renoux C, Dell’Aniello S, Suissa S. Hormone replacement therapy and the risk of venous thromboembolism: a population-based study. J Thromb Haemost. 2010;8(5):979−986.
- Canonico M, Plu-Bureau G, Lowe GD, Scarabin PY. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ. 2008;336(7655):1227−1231.
- Canonico M, Fournier A, Carcaillon L, et al. Postmenopausal hormone therapy and risk of idiopathic venous thromboembolism: results from the E3N cohort study. Arterioscler Thromb Vasc Biol. 2010;30(2):340−345.
- Laliberte F, Dea K, Duh MS, Kahler KH, Rolli M, Lefebvre P. Does the route of administration for estrogen hormone therapy impact the risk of venous thromboembolism? Estradiol transdermal system versus oral estrogen-only hormone therapy. Menopause. 2011;18(10):1052−1059.
- Sweetland S, Beral V, Balkwill A, et al. Venous thromboembolism risk in relation to different types of postmenopausal hormone therapy in a large prospective study. J Thromb Haemost. 2012;10(11):2277−2286.
- Fournier A, Berrino F, Riboli E, Avenel V, Clavel-Chapelon F. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114(3):448−454.
- L’Hermite M. HRT optimization, using transdermal estradiol plus micronized progesterone, a safer HRT. Climacteric. 2013;16(suppl 1):44−53.
- Simon JA. What’s new in hormone replacement therapy: focus on transdermal estradiol and micronized progesterone. Climacteric. 2012;15(suppl 1):3−10.
- Mueck AO. Postmenopausal hormone replacement therapy and cardiovascular disease: the value of transdermal estradiol and micronized progesterone. Climacteric. 2012;15(suppl 1): 11−17.
Clinicians work to maximize the quality of life and longevity of every patient. For women with moderate to severe menopausal symptoms, oral estrogen therapy can improve quality of life, but at the cost of significant adverse effects. The Women’s Health Initiative (WHI) reported that for postmenopausal women with a uterus, conjugated estrogen plus medroxyprogesterone acetate (CEE+MPA) hormone therapy (HT) versus placebo significantly increased the risk of cardiovascular events (relative risk [RR], 1.13), breast cancer (RR, 1.24), stroke (RR, 1.37), deep vein thrombosis (RR, 1.87), and pulmonary embolism (RR, 1.98).1 In postmeno pausal women without a uterus, CEE HT did not increase the risk of breast cancer (RR, 0.79), compared with placebo, but it did significantly in crease the risk of cardiovascular events (RR, 1.11), stroke (RR, 1.35), deep vein thrombosis (RR, 1.48), and pulmonary embolism (RR, 1.35).1
Clinicians prescribing estrogen must individualize therapy according to its benefits and risks. An important issue that has received insufficient at tention is, “What is the effect of HT on mortality in recently menopausal women?” Here, I examine this issue.
HT reduces mortality in recently menopausal women
Pooling the results of the WHI CEE+MPA and CEE-only trials reveals that there were 70 deaths in the HT-treated groups and 98 deaths in the placebo groups among women aged 50 to 59 years.1 With 4,706 and 4,259 women alive at the conclusion of the study in the HT and placebo groups, respectively, the women in the placebo group had significantly more deaths than the women in the HT-treated groups (Fisher exact test, P = .0194, χ2 test with Yates correction, P = .0226).
Using pooled data from the WHI, the RR of death in the HT versus placebo group was estimated at 0.70 (95% confidence interval [CI], 0.51−0.96), representing approximately 5 fewer deaths per 1,000 women per 5 years of therapy.2 In women aged 60 to 69 years and 70 to 79 years there were no significant differences in death rates between the HT- and placebo-treated women.
My interpretation of these results is that HT likely is associated with a reduced risk of death in recently menopausal women, but not in women distant from menopause onset.
Cochrane review of HT and mortality
Consistent with the WHI findings, authors of a recent Cochrane meta-analysis of 19 randomized trials including 40,410 menopausal women reported that HT significantly increased the risk of stroke (RR, 1.24; 95% CI, 1.10−1.41), venous thromboembolism (RR, 1.92; 95% CI, 1.36−2.69), and pulmonary emboli (RR, 1.81; 95% CI, 1.32−2.48).3 However, among women treated with oral HT within 10 years after the start of menopause, there was a reduced risk of coronary heart disease (RR, 0.52; 95% CI, 0.29−0.96). Using data from 5 clinical trials, the Cochrane meta-analysis researchers reported that, compared with placebo, HT reduced mortality (RR, 0.70; 95% CI, 0.52−0.95).3
Results of the Cochrane meta-analysis are consistent with those of a previous meta-analysis of 19 randomized trials involving 16,000 women. In this analysis, investigators found a reduced risk of death in recently menopausal women treated with hormone therapy (RR, 0.73; 95% CI, 0.52−0.96).4
Early menopause, HT, and mortality
Authors of multiple large epidemiologic studies have reported that early menopause is associated with an increased risk of death if HT is not initiated.5−7 For example, results of a study of women in Olmsted County, Minnesota, conducted from 1950 to 1987, indicated that, for women younger than age 45 years who underwent bilateral oophorectomy, the risk of death was increased among those who did not initiate HT, compared with women who did not undergo oophorectomy (hazard ratio [HR], 1.84; 95% CI, 1.27−2.68; P = .001).7
By contrast, women younger than 45 years who underwent bilateral oophorectomy and initiated estrogen therapy did not have an increased risk of death compared with women who did not undergo oophorectomy (HR, 0.65; 95% CI, 0.30−1.41; P = .28).7 An excess number of cardiovascular events appeared to account for the increased mortality among women with early surgical menopause who did not initiate HT.
The “timing hypothesis” proposes that the initiation of HT soon after the onset of menopause is associated with beneficial cardiovascular effects, but initiation more than 10 years after the onset of menopause is not associated with beneficial cardiovascular effects. The timing hypothesis is supported by the finding that, in recently menopausal women, HT is associated with reduced carotid intima-media thickness (CIMT), compared with placebo.8 Greater CIMT thickness is associated with an increased risk of cardiovascular events.
In my experience, few primary care clinicians are aware of these data. Often, these clinicians over-emphasize the risks and withhold HT in this vulnerable group of women.
HT: Minimizing the risks of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer
Results of multiple studies have shown that certain HT regimens increase the risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer. Is it possible to prescribe HT in a way that reduces these risks?
Results of observational studies indicate that, compared with oral estrogen therapy, transdermal HT is associated with a lower risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer (TABLE).9−15
Reducing the risk of stroke caused by HT is an important goal. In a study of 15,710 women who had stroke and 59,958 control women aged 50 to 79 years, transdermal estradiol at a dose of 50 µg or less daily was not associated with an increased risk of stroke, compared with HT nonuse (rate ratio, 0.81; 95% CI, 0.62−1.05).9 Compared with HT nonuse, the use of oral estrogen (rate ratio, 1.28; 95% CI, 1.15−1.42) or transdermal estradiol 50 µg or greater daily (rate ratio, 1.89; 95% CI, 1.15−3.11) was associated with an increased risk of stroke.9
Reducing the risks of deep venous thromboembolism (VTE) and pulmonary embolism caused by HT is an important goal. In a meta-analysis of the risk of VTE with HT, compared with nonusers, oral estrogen therapy was associated with a significantly increased risk of VTE (odds ratio [OR], 2.5; 95% CI, 1.9−3.4). Compared with nonuse, transdermal estrogen therapy was not associated with an increased risk of VTE (OR, 1.2; 95% CI, 0.9−1.7).11 In a study comparing oral versus transdermal estradiol, transdermal estradiol was associated with a reduced risk of pulmonary embolism (0.46 [95% CI, 0.22−0.97]).13
Reducing the risk of breast cancer caused by HT is an important goal. Results of one study showed that the combination of oral estrogen plus synthetic progestin was associated with an increased risk of breast cancer, compared with nonuse (RR, 1.5; 95% CI, 1.1−1.9). By contrast, the combination of transdermal estradiol plus micronized progesterone was not associated with an increased risk of breast cancer, compared with nonuse (RR, 0.9; 95% CI, 0.7−1.2).15
The bottom line
In recently menopausal women with moderate to severe hot flashes, HT improves quality of life and appears to decrease mortality. However, HT with oral estrogen plus synthetic progestin is associated with an increased risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer. Compared with oral estrogen, transdermal estradiol treatment is associated with a lower risk of stroke, deep vein thrombosis, and pulmonary embolism. Compared with oral estrogen plus a synthetic progestin, transdermal estradiol plus micronized progesterone is associated with a lower risk of breast cancer. The benefits of HT are likely maximized by initiating therapy in the perimenopause transition or early in the postmenopause, and the risks are minimized by using transdermal estradiol.16−18
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Clinicians work to maximize the quality of life and longevity of every patient. For women with moderate to severe menopausal symptoms, oral estrogen therapy can improve quality of life, but at the cost of significant adverse effects. The Women’s Health Initiative (WHI) reported that for postmenopausal women with a uterus, conjugated estrogen plus medroxyprogesterone acetate (CEE+MPA) hormone therapy (HT) versus placebo significantly increased the risk of cardiovascular events (relative risk [RR], 1.13), breast cancer (RR, 1.24), stroke (RR, 1.37), deep vein thrombosis (RR, 1.87), and pulmonary embolism (RR, 1.98).1 In postmeno pausal women without a uterus, CEE HT did not increase the risk of breast cancer (RR, 0.79), compared with placebo, but it did significantly in crease the risk of cardiovascular events (RR, 1.11), stroke (RR, 1.35), deep vein thrombosis (RR, 1.48), and pulmonary embolism (RR, 1.35).1
Clinicians prescribing estrogen must individualize therapy according to its benefits and risks. An important issue that has received insufficient at tention is, “What is the effect of HT on mortality in recently menopausal women?” Here, I examine this issue.
HT reduces mortality in recently menopausal women
Pooling the results of the WHI CEE+MPA and CEE-only trials reveals that there were 70 deaths in the HT-treated groups and 98 deaths in the placebo groups among women aged 50 to 59 years.1 With 4,706 and 4,259 women alive at the conclusion of the study in the HT and placebo groups, respectively, the women in the placebo group had significantly more deaths than the women in the HT-treated groups (Fisher exact test, P = .0194, χ2 test with Yates correction, P = .0226).
Using pooled data from the WHI, the RR of death in the HT versus placebo group was estimated at 0.70 (95% confidence interval [CI], 0.51−0.96), representing approximately 5 fewer deaths per 1,000 women per 5 years of therapy.2 In women aged 60 to 69 years and 70 to 79 years there were no significant differences in death rates between the HT- and placebo-treated women.
My interpretation of these results is that HT likely is associated with a reduced risk of death in recently menopausal women, but not in women distant from menopause onset.
Cochrane review of HT and mortality
Consistent with the WHI findings, authors of a recent Cochrane meta-analysis of 19 randomized trials including 40,410 menopausal women reported that HT significantly increased the risk of stroke (RR, 1.24; 95% CI, 1.10−1.41), venous thromboembolism (RR, 1.92; 95% CI, 1.36−2.69), and pulmonary emboli (RR, 1.81; 95% CI, 1.32−2.48).3 However, among women treated with oral HT within 10 years after the start of menopause, there was a reduced risk of coronary heart disease (RR, 0.52; 95% CI, 0.29−0.96). Using data from 5 clinical trials, the Cochrane meta-analysis researchers reported that, compared with placebo, HT reduced mortality (RR, 0.70; 95% CI, 0.52−0.95).3
Results of the Cochrane meta-analysis are consistent with those of a previous meta-analysis of 19 randomized trials involving 16,000 women. In this analysis, investigators found a reduced risk of death in recently menopausal women treated with hormone therapy (RR, 0.73; 95% CI, 0.52−0.96).4
Early menopause, HT, and mortality
Authors of multiple large epidemiologic studies have reported that early menopause is associated with an increased risk of death if HT is not initiated.5−7 For example, results of a study of women in Olmsted County, Minnesota, conducted from 1950 to 1987, indicated that, for women younger than age 45 years who underwent bilateral oophorectomy, the risk of death was increased among those who did not initiate HT, compared with women who did not undergo oophorectomy (hazard ratio [HR], 1.84; 95% CI, 1.27−2.68; P = .001).7
By contrast, women younger than 45 years who underwent bilateral oophorectomy and initiated estrogen therapy did not have an increased risk of death compared with women who did not undergo oophorectomy (HR, 0.65; 95% CI, 0.30−1.41; P = .28).7 An excess number of cardiovascular events appeared to account for the increased mortality among women with early surgical menopause who did not initiate HT.
The “timing hypothesis” proposes that the initiation of HT soon after the onset of menopause is associated with beneficial cardiovascular effects, but initiation more than 10 years after the onset of menopause is not associated with beneficial cardiovascular effects. The timing hypothesis is supported by the finding that, in recently menopausal women, HT is associated with reduced carotid intima-media thickness (CIMT), compared with placebo.8 Greater CIMT thickness is associated with an increased risk of cardiovascular events.
In my experience, few primary care clinicians are aware of these data. Often, these clinicians over-emphasize the risks and withhold HT in this vulnerable group of women.
HT: Minimizing the risks of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer
Results of multiple studies have shown that certain HT regimens increase the risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer. Is it possible to prescribe HT in a way that reduces these risks?
Results of observational studies indicate that, compared with oral estrogen therapy, transdermal HT is associated with a lower risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer (TABLE).9−15
Reducing the risk of stroke caused by HT is an important goal. In a study of 15,710 women who had stroke and 59,958 control women aged 50 to 79 years, transdermal estradiol at a dose of 50 µg or less daily was not associated with an increased risk of stroke, compared with HT nonuse (rate ratio, 0.81; 95% CI, 0.62−1.05).9 Compared with HT nonuse, the use of oral estrogen (rate ratio, 1.28; 95% CI, 1.15−1.42) or transdermal estradiol 50 µg or greater daily (rate ratio, 1.89; 95% CI, 1.15−3.11) was associated with an increased risk of stroke.9
Reducing the risks of deep venous thromboembolism (VTE) and pulmonary embolism caused by HT is an important goal. In a meta-analysis of the risk of VTE with HT, compared with nonusers, oral estrogen therapy was associated with a significantly increased risk of VTE (odds ratio [OR], 2.5; 95% CI, 1.9−3.4). Compared with nonuse, transdermal estrogen therapy was not associated with an increased risk of VTE (OR, 1.2; 95% CI, 0.9−1.7).11 In a study comparing oral versus transdermal estradiol, transdermal estradiol was associated with a reduced risk of pulmonary embolism (0.46 [95% CI, 0.22−0.97]).13
Reducing the risk of breast cancer caused by HT is an important goal. Results of one study showed that the combination of oral estrogen plus synthetic progestin was associated with an increased risk of breast cancer, compared with nonuse (RR, 1.5; 95% CI, 1.1−1.9). By contrast, the combination of transdermal estradiol plus micronized progesterone was not associated with an increased risk of breast cancer, compared with nonuse (RR, 0.9; 95% CI, 0.7−1.2).15
The bottom line
In recently menopausal women with moderate to severe hot flashes, HT improves quality of life and appears to decrease mortality. However, HT with oral estrogen plus synthetic progestin is associated with an increased risk of stroke, deep vein thrombosis, pulmonary embolism, and breast cancer. Compared with oral estrogen, transdermal estradiol treatment is associated with a lower risk of stroke, deep vein thrombosis, and pulmonary embolism. Compared with oral estrogen plus a synthetic progestin, transdermal estradiol plus micronized progesterone is associated with a lower risk of breast cancer. The benefits of HT are likely maximized by initiating therapy in the perimenopause transition or early in the postmenopause, and the risks are minimized by using transdermal estradiol.16−18
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended post-stopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310(13):1353−1368.
- Santen RJ, Allred DC, Ardoin SP, et al. J Clin Endocrinol Metab. 2010;95(suppl 1):S1−S66.
- Boardman HM, Hartley L, Eisinga A, et al. Hormone therapy for preventing cardiovascular disease in postmenopausal women. Cochrane Database Syst Rev. 2015;3:CD002229.
- Salpeter SR, Cheng J, Thabane L, Buckley NS, Salpeter EE. Bayesian meta-analysis of hormone therapy and mortality in younger post-menopausal women. Am J Med. 2009;122(11):1016−1022.
- Gordon T, Kannel WB, Hjortland MC, McNamara PM. Menopause and coronary heart disease: The Framingham Study. Ann Intern Med. 1978;89(2):157−161.
- Stampfer MJ, Colditz GA, Willet WC, et al. Postmenopausal estrogen therapy and cardiovascular disease. Ten-year follow-up from the Nurses Health Study. N Engl J Med. 1991;325(11):756−762.
- Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16(1):15−23.
- Hodis HN, Mack WJ, Shoupe D, et al. Testing the menopausal hormone therapy timing hypothesis: the early versus late intervention trial with estradiol [abstract 13283]. American Heart Association Meeting 2014. Circulation. 2014;130:A13283.
- Renoux C, Dell’Aniello S, Garbe E, Suissa S. Transdermal and oral hormone replacement therapy and the risk of stroke: a nested case-control study. BMJ. 2010;340:c2519
- Renoux C, Dell’Aniello S, Suissa S. Hormone replacement therapy and the risk of venous thromboembolism: a population-based study. J Thromb Haemost. 2010;8(5):979−986.
- Canonico M, Plu-Bureau G, Lowe GD, Scarabin PY. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ. 2008;336(7655):1227−1231.
- Canonico M, Fournier A, Carcaillon L, et al. Postmenopausal hormone therapy and risk of idiopathic venous thromboembolism: results from the E3N cohort study. Arterioscler Thromb Vasc Biol. 2010;30(2):340−345.
- Laliberte F, Dea K, Duh MS, Kahler KH, Rolli M, Lefebvre P. Does the route of administration for estrogen hormone therapy impact the risk of venous thromboembolism? Estradiol transdermal system versus oral estrogen-only hormone therapy. Menopause. 2011;18(10):1052−1059.
- Sweetland S, Beral V, Balkwill A, et al. Venous thromboembolism risk in relation to different types of postmenopausal hormone therapy in a large prospective study. J Thromb Haemost. 2012;10(11):2277−2286.
- Fournier A, Berrino F, Riboli E, Avenel V, Clavel-Chapelon F. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114(3):448−454.
- L’Hermite M. HRT optimization, using transdermal estradiol plus micronized progesterone, a safer HRT. Climacteric. 2013;16(suppl 1):44−53.
- Simon JA. What’s new in hormone replacement therapy: focus on transdermal estradiol and micronized progesterone. Climacteric. 2012;15(suppl 1):3−10.
- Mueck AO. Postmenopausal hormone replacement therapy and cardiovascular disease: the value of transdermal estradiol and micronized progesterone. Climacteric. 2012;15(suppl 1): 11−17.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended post-stopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310(13):1353−1368.
- Santen RJ, Allred DC, Ardoin SP, et al. J Clin Endocrinol Metab. 2010;95(suppl 1):S1−S66.
- Boardman HM, Hartley L, Eisinga A, et al. Hormone therapy for preventing cardiovascular disease in postmenopausal women. Cochrane Database Syst Rev. 2015;3:CD002229.
- Salpeter SR, Cheng J, Thabane L, Buckley NS, Salpeter EE. Bayesian meta-analysis of hormone therapy and mortality in younger post-menopausal women. Am J Med. 2009;122(11):1016−1022.
- Gordon T, Kannel WB, Hjortland MC, McNamara PM. Menopause and coronary heart disease: The Framingham Study. Ann Intern Med. 1978;89(2):157−161.
- Stampfer MJ, Colditz GA, Willet WC, et al. Postmenopausal estrogen therapy and cardiovascular disease. Ten-year follow-up from the Nurses Health Study. N Engl J Med. 1991;325(11):756−762.
- Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16(1):15−23.
- Hodis HN, Mack WJ, Shoupe D, et al. Testing the menopausal hormone therapy timing hypothesis: the early versus late intervention trial with estradiol [abstract 13283]. American Heart Association Meeting 2014. Circulation. 2014;130:A13283.
- Renoux C, Dell’Aniello S, Garbe E, Suissa S. Transdermal and oral hormone replacement therapy and the risk of stroke: a nested case-control study. BMJ. 2010;340:c2519
- Renoux C, Dell’Aniello S, Suissa S. Hormone replacement therapy and the risk of venous thromboembolism: a population-based study. J Thromb Haemost. 2010;8(5):979−986.
- Canonico M, Plu-Bureau G, Lowe GD, Scarabin PY. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. BMJ. 2008;336(7655):1227−1231.
- Canonico M, Fournier A, Carcaillon L, et al. Postmenopausal hormone therapy and risk of idiopathic venous thromboembolism: results from the E3N cohort study. Arterioscler Thromb Vasc Biol. 2010;30(2):340−345.
- Laliberte F, Dea K, Duh MS, Kahler KH, Rolli M, Lefebvre P. Does the route of administration for estrogen hormone therapy impact the risk of venous thromboembolism? Estradiol transdermal system versus oral estrogen-only hormone therapy. Menopause. 2011;18(10):1052−1059.
- Sweetland S, Beral V, Balkwill A, et al. Venous thromboembolism risk in relation to different types of postmenopausal hormone therapy in a large prospective study. J Thromb Haemost. 2012;10(11):2277−2286.
- Fournier A, Berrino F, Riboli E, Avenel V, Clavel-Chapelon F. Breast cancer risk in relation to different types of hormone replacement therapy in the E3N-EPIC cohort. Int J Cancer. 2005;114(3):448−454.
- L’Hermite M. HRT optimization, using transdermal estradiol plus micronized progesterone, a safer HRT. Climacteric. 2013;16(suppl 1):44−53.
- Simon JA. What’s new in hormone replacement therapy: focus on transdermal estradiol and micronized progesterone. Climacteric. 2012;15(suppl 1):3−10.
- Mueck AO. Postmenopausal hormone replacement therapy and cardiovascular disease: the value of transdermal estradiol and micronized progesterone. Climacteric. 2012;15(suppl 1): 11−17.
Needed: A biopsychosocial ‘therapeutic placenta’ for people with schizophrenia
Consider stroke. Guidelines for acute treatment, access, intervention, prevention of post-hospitalization relapse, and rehabilitation are extensively spelled out and implemented.1 (The Box outlines Mayo Clinic guidelines for stroke management, as a demonstration of the comprehensiveness of the approach.)
Schizophrenia and related severe mental illnesses (SMI) need a similar all-inclusive system that seamlessly provides the myriad components of care needed for this vulnerable population. I propose the term “therapeutic placenta” to describe what people with a disabling SMI brain disorder deserve, just as stroke patients do.
Closing asylums: Psychosocial abruptio placentae
In a past Editorial,2 I described the appalling consequences of eliminating the asylum, an entity that I believe must be a key component of the SMI therapeutic placenta. The asylum is to schizophrenia as the skilled nursing home is to stroke. SMI patients suffered extensively when asylums were shut down; they lost a medical refuge with psychiatric and primary care, nursing and social work support, occupational and recreational therapies, and work therapy (farming, carpentry shop, cafeteria, laundry, etc.). For SMI, these services are the psychosocial counterpart of various physical rehabilitation therapies for stroke patients that no one would ever dare to eliminate.
Persons with schizophrenia and other SMI have suffered tragically with rupture of the main components of the therapeutic placenta that existed for decades before the advent of medications. The massive homelessness, widespread incarceration, persistent poverty, rampant access to alcohol and drugs of abuse, early death due to lack of primary care, and absence of meaningful opportunities for vocational rehabilitation are all consequences of a neglectful society that refuses to fund a therapeutic placenta for the SMI population.
The public mental health system in charge of SMI patients is broken, disconnected, and failing to provide the necessary components of a therapeutic placenta. It should not be surprising to witness the terribly stressful life and premature mortality of SMI patients, who are modern-day les misérables.
The Table lists what I consider to be the necessary spectrum of health care services through the life of an SMI patient that an optimal therapeutic placenta must provide until an effective prevention or a cure for SMI is discovered.
Reasons to be hopeful
Admittedly, encouraging steps are being made toward establishing a therapeutic placenta for SMI:
The RAISE Study3and Navigate Program4 demonstrate that implementing a comprehensive program of acute treatment and psychosocial interventions and rehabilitation yields better outcomes in SMI.
The Institute of Medicine released a landmark report on psychosocial interventions for mental illness and substance abuse disorders. It outlines a new model for establishing the effectiveness of intervention and the implementation of psychosocial strategies in clinical practice.5
The 21st Century Cures Act, if passed by Congress and signed by the President, will increase funding for the National Institutes of Health, which in turn will bolster the budgets of the National Institute of Mental Health, National Institute on Drug Abuse, and the National Institute on Alcohol Abuse and Alcoholism and enhance the chances of discovering better treatments and prevention of SMI.
The Helping Families in Mental Health Crisis Act, more directly relevant to mental health and psychiatry, proposes, if passed, to:
• enhance evidence-based and scientifically validated interventions in the public sector
• raise the profile of mental health within the federal government by creating a position of Assistant Secretary for Mental Health in the U.S. Department of Health and Human Services, who will have oversight of both research and mental health care within the federal government.
Unacceptable disparity must be remedied
Planning an effective therapeutic placenta is imperative if health care for SMI patients is to approach the comprehensive spectrum of treatment, rehabilitation, and prevention available to stroke patients. Although stroke is regarded as a sensory-motor brain disorder, it is also associated with mental symptoms, just as schizophrenia is associated with sensory-motor symptoms. Both are disabling brain disorders: one, physically and cognitively; the other, mentally and socially. Both require a therapeutic placenta: Stroke is supported by one; schizophrenia is not. This is an unacceptable disparity that must be addressed—soon.
1. Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870-947.
2. Nasrallah HA. Bring back the asylums? Current Psychiatry. 2008;7(3):19-20.
3. Kane JM, Schooler NR, Marcy P, et al. The RAISE early treatment program for first-episode psychosis: background, rationale, and study design. J Clin Psychiatry. 2015;76(3):240-246.
4. Mueser KT, Penn DL, Addington J, et al. The NAVIGATE program for first-episode psychosis: rationale, overview, and description of psychosocial components. Psychiatr Serv. 2015;66(7):680-690.
5. The National Academy of Sciences. Psychosocial interventions for mental and substance use disorders: a framework for establishing evidence-based standards. Washington, DC. http:// iom.nationalacademies.org/Reports/2015/ Psychosocial-Interventions-Mental-Substance- Abuse-Disorders.aspx. Published July 14, 2015. Accessed September 3, 2015.
Consider stroke. Guidelines for acute treatment, access, intervention, prevention of post-hospitalization relapse, and rehabilitation are extensively spelled out and implemented.1 (The Box outlines Mayo Clinic guidelines for stroke management, as a demonstration of the comprehensiveness of the approach.)
Schizophrenia and related severe mental illnesses (SMI) need a similar all-inclusive system that seamlessly provides the myriad components of care needed for this vulnerable population. I propose the term “therapeutic placenta” to describe what people with a disabling SMI brain disorder deserve, just as stroke patients do.
Closing asylums: Psychosocial abruptio placentae
In a past Editorial,2 I described the appalling consequences of eliminating the asylum, an entity that I believe must be a key component of the SMI therapeutic placenta. The asylum is to schizophrenia as the skilled nursing home is to stroke. SMI patients suffered extensively when asylums were shut down; they lost a medical refuge with psychiatric and primary care, nursing and social work support, occupational and recreational therapies, and work therapy (farming, carpentry shop, cafeteria, laundry, etc.). For SMI, these services are the psychosocial counterpart of various physical rehabilitation therapies for stroke patients that no one would ever dare to eliminate.
Persons with schizophrenia and other SMI have suffered tragically with rupture of the main components of the therapeutic placenta that existed for decades before the advent of medications. The massive homelessness, widespread incarceration, persistent poverty, rampant access to alcohol and drugs of abuse, early death due to lack of primary care, and absence of meaningful opportunities for vocational rehabilitation are all consequences of a neglectful society that refuses to fund a therapeutic placenta for the SMI population.
The public mental health system in charge of SMI patients is broken, disconnected, and failing to provide the necessary components of a therapeutic placenta. It should not be surprising to witness the terribly stressful life and premature mortality of SMI patients, who are modern-day les misérables.
The Table lists what I consider to be the necessary spectrum of health care services through the life of an SMI patient that an optimal therapeutic placenta must provide until an effective prevention or a cure for SMI is discovered.
Reasons to be hopeful
Admittedly, encouraging steps are being made toward establishing a therapeutic placenta for SMI:
The RAISE Study3and Navigate Program4 demonstrate that implementing a comprehensive program of acute treatment and psychosocial interventions and rehabilitation yields better outcomes in SMI.
The Institute of Medicine released a landmark report on psychosocial interventions for mental illness and substance abuse disorders. It outlines a new model for establishing the effectiveness of intervention and the implementation of psychosocial strategies in clinical practice.5
The 21st Century Cures Act, if passed by Congress and signed by the President, will increase funding for the National Institutes of Health, which in turn will bolster the budgets of the National Institute of Mental Health, National Institute on Drug Abuse, and the National Institute on Alcohol Abuse and Alcoholism and enhance the chances of discovering better treatments and prevention of SMI.
The Helping Families in Mental Health Crisis Act, more directly relevant to mental health and psychiatry, proposes, if passed, to:
• enhance evidence-based and scientifically validated interventions in the public sector
• raise the profile of mental health within the federal government by creating a position of Assistant Secretary for Mental Health in the U.S. Department of Health and Human Services, who will have oversight of both research and mental health care within the federal government.
Unacceptable disparity must be remedied
Planning an effective therapeutic placenta is imperative if health care for SMI patients is to approach the comprehensive spectrum of treatment, rehabilitation, and prevention available to stroke patients. Although stroke is regarded as a sensory-motor brain disorder, it is also associated with mental symptoms, just as schizophrenia is associated with sensory-motor symptoms. Both are disabling brain disorders: one, physically and cognitively; the other, mentally and socially. Both require a therapeutic placenta: Stroke is supported by one; schizophrenia is not. This is an unacceptable disparity that must be addressed—soon.
Consider stroke. Guidelines for acute treatment, access, intervention, prevention of post-hospitalization relapse, and rehabilitation are extensively spelled out and implemented.1 (The Box outlines Mayo Clinic guidelines for stroke management, as a demonstration of the comprehensiveness of the approach.)
Schizophrenia and related severe mental illnesses (SMI) need a similar all-inclusive system that seamlessly provides the myriad components of care needed for this vulnerable population. I propose the term “therapeutic placenta” to describe what people with a disabling SMI brain disorder deserve, just as stroke patients do.
Closing asylums: Psychosocial abruptio placentae
In a past Editorial,2 I described the appalling consequences of eliminating the asylum, an entity that I believe must be a key component of the SMI therapeutic placenta. The asylum is to schizophrenia as the skilled nursing home is to stroke. SMI patients suffered extensively when asylums were shut down; they lost a medical refuge with psychiatric and primary care, nursing and social work support, occupational and recreational therapies, and work therapy (farming, carpentry shop, cafeteria, laundry, etc.). For SMI, these services are the psychosocial counterpart of various physical rehabilitation therapies for stroke patients that no one would ever dare to eliminate.
Persons with schizophrenia and other SMI have suffered tragically with rupture of the main components of the therapeutic placenta that existed for decades before the advent of medications. The massive homelessness, widespread incarceration, persistent poverty, rampant access to alcohol and drugs of abuse, early death due to lack of primary care, and absence of meaningful opportunities for vocational rehabilitation are all consequences of a neglectful society that refuses to fund a therapeutic placenta for the SMI population.
The public mental health system in charge of SMI patients is broken, disconnected, and failing to provide the necessary components of a therapeutic placenta. It should not be surprising to witness the terribly stressful life and premature mortality of SMI patients, who are modern-day les misérables.
The Table lists what I consider to be the necessary spectrum of health care services through the life of an SMI patient that an optimal therapeutic placenta must provide until an effective prevention or a cure for SMI is discovered.
Reasons to be hopeful
Admittedly, encouraging steps are being made toward establishing a therapeutic placenta for SMI:
The RAISE Study3and Navigate Program4 demonstrate that implementing a comprehensive program of acute treatment and psychosocial interventions and rehabilitation yields better outcomes in SMI.
The Institute of Medicine released a landmark report on psychosocial interventions for mental illness and substance abuse disorders. It outlines a new model for establishing the effectiveness of intervention and the implementation of psychosocial strategies in clinical practice.5
The 21st Century Cures Act, if passed by Congress and signed by the President, will increase funding for the National Institutes of Health, which in turn will bolster the budgets of the National Institute of Mental Health, National Institute on Drug Abuse, and the National Institute on Alcohol Abuse and Alcoholism and enhance the chances of discovering better treatments and prevention of SMI.
The Helping Families in Mental Health Crisis Act, more directly relevant to mental health and psychiatry, proposes, if passed, to:
• enhance evidence-based and scientifically validated interventions in the public sector
• raise the profile of mental health within the federal government by creating a position of Assistant Secretary for Mental Health in the U.S. Department of Health and Human Services, who will have oversight of both research and mental health care within the federal government.
Unacceptable disparity must be remedied
Planning an effective therapeutic placenta is imperative if health care for SMI patients is to approach the comprehensive spectrum of treatment, rehabilitation, and prevention available to stroke patients. Although stroke is regarded as a sensory-motor brain disorder, it is also associated with mental symptoms, just as schizophrenia is associated with sensory-motor symptoms. Both are disabling brain disorders: one, physically and cognitively; the other, mentally and socially. Both require a therapeutic placenta: Stroke is supported by one; schizophrenia is not. This is an unacceptable disparity that must be addressed—soon.
1. Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870-947.
2. Nasrallah HA. Bring back the asylums? Current Psychiatry. 2008;7(3):19-20.
3. Kane JM, Schooler NR, Marcy P, et al. The RAISE early treatment program for first-episode psychosis: background, rationale, and study design. J Clin Psychiatry. 2015;76(3):240-246.
4. Mueser KT, Penn DL, Addington J, et al. The NAVIGATE program for first-episode psychosis: rationale, overview, and description of psychosocial components. Psychiatr Serv. 2015;66(7):680-690.
5. The National Academy of Sciences. Psychosocial interventions for mental and substance use disorders: a framework for establishing evidence-based standards. Washington, DC. http:// iom.nationalacademies.org/Reports/2015/ Psychosocial-Interventions-Mental-Substance- Abuse-Disorders.aspx. Published July 14, 2015. Accessed September 3, 2015.
1. Jauch EC, Saver JL, Adams HP Jr, et al; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870-947.
2. Nasrallah HA. Bring back the asylums? Current Psychiatry. 2008;7(3):19-20.
3. Kane JM, Schooler NR, Marcy P, et al. The RAISE early treatment program for first-episode psychosis: background, rationale, and study design. J Clin Psychiatry. 2015;76(3):240-246.
4. Mueser KT, Penn DL, Addington J, et al. The NAVIGATE program for first-episode psychosis: rationale, overview, and description of psychosocial components. Psychiatr Serv. 2015;66(7):680-690.
5. The National Academy of Sciences. Psychosocial interventions for mental and substance use disorders: a framework for establishing evidence-based standards. Washington, DC. http:// iom.nationalacademies.org/Reports/2015/ Psychosocial-Interventions-Mental-Substance- Abuse-Disorders.aspx. Published July 14, 2015. Accessed September 3, 2015.
Changing oncology compliance standards: step 1 in re-valuing clinician workload for value-based cancer care
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
Reducing maternal mortality in the United States—Let’s get organized!
A mother’s untimely death in childbirth is a grave loss that sends shock waves of grief across generations of her family and community. As obstetricians practicing in the United States, we face a terrible problem. We have a continually rising rate of maternal death in a country with exceptional medical resources (FIGURE).1 Our national decentralized approach to dealing with maternal mortality is a factor contributing to the decades-long increase in the maternal mortality ratio. Let’s get organized to better respond to this public health crisis.
Medical education— Let’s get focused on maternal mortality
The 140-page Council on Resident Education in Obstetrics and Gynecology CREOG Educational Objectives: Core Curriculum in Obstetrics and Gynecology provides a detailed enumeration of the key learning objectives for residents in obstetrics and gynecology.2 Surprisingly, the CREOG objectives do not mention reducing maternal mortality as an important curricular goal. Learning clinical processes and practices that decrease the risk of maternal mortality should be an important educational goal for all residents training in obstetrics and gynecology.
Nationwide action is needed to address the problem
Many countries have organized widespread efforts to reduce maternal mortality. In the United Kingdom and France there are nationwide reviews of maternal deaths with detailed analyses of clinical events and identification of areas for future improvement. These reviews result in the dissemination of countrywide clinical recommendations that change practice and hopefully reduce the risk of future maternal deaths. For example, following the identification of pulmonary embolism as a leading cause of maternal death in the United Kingdom there was a nationwide effort to increase the use of mechanical and pharmacologic prophylaxis to prevent deep venous thrombosis.
In the United States, experts have proposed that a national program of clinical review of severe maternal morbidity cases should be mandatory. (There are many more cases of “near misses” with severe maternal morbidity than there are maternal deaths.) The greater number of cases available for review should help institutions to quickly recognize potential areas for clinical improvement. One group of experts has recommended that all deliveries in which a pregnant woman received 4 or more units of blood or was admitted to an intensive care unit should be thoroughly reviewed to identify opportunities for clinical improvement.3
In the United Kingdom a contemporary clinical problem that is being addressed in an organized and systematic manner is how to respond to the rising rate of severe maternal morbidity caused by placenta accreta. Experts have concluded that women with a suspected placenta accreta should deliver in regional centers with advanced clinical resources—including an emergency surgical response team, interventional radiology, a high capacity blood bank, and an intensive care unit.
A similar approach has been proposed for managing placenta accreta in the United States.4 The American College of Obstetricians and Gynecologists (ACOG) and the Society of Maternal Fetal Medicine (SMFM) have proposed a tiered system of obstetric care with more complex cases being referred to regional perinatal centers.5 Regionalization of trauma services has been an important part of the US health care system for decades. Cases of severe trauma are brought to regional centers equipped to emergently treat complex injuries. A similar system of regulation and regionalization could be adapted for optimizing maternity care.
High-risk clinical events: Is your unit prepared?
In the United States the leading causes of maternal mortality, in descending order, are6−8:
- cardiovascular diseases
- infection
- hemorrhage
- cardiomyopathy
- pulmonary embolism
- hypertension
- amniotic fluid embolism
- stroke
- anesthesia complications.
Over the last decade, the Joint Commission has recommended that birthing centers develop standardized protocols and use simulation to improve the institution’s ability to respond in a timely manner to clinical events that may result in maternal morbidity or death.
The quality of published protocols dealing with hemorrhage, hypertension, and thromboembolism is continuously improving, and every birthing center should have written protocols that are updated on a regular timetable for these common high-risk events.9,10 Does your birthing unit have written protocols to deal with cardiac diseases, infection, obstetric hemorrhage, thromboembolism, and severe hypertension? Are simulation exercises used to strengthen familiarity with the protocols?
High-risk patients
An amazing fact of today’s medical care is that sexually active women of reproductive age who have high-risk medical problems often have not been counseled to use a highly effective contraceptive, resulting in an increased risk of unintended pregnancy and maternal death. For example, adult women with a history of congenital heart disease are known to be at increased risk of death if they become pregnant. In a recent study, women with a history of congenital heart disease had 178 maternal deaths per 100,000 deliveries—a rate approximately 10-fold higher than the US maternal mortality ratio.11 Yet, many of these women are not using a highly effective contraceptive, and this results in a high rate of unplanned pregnancy.12
In order to reduce the risk of unintended pregnancy in women with high-risk medical problems, health systems could make contraception an important “vital sign” for women with high-risk medical conditions.
Race and age matter greatly when it comes to maternal mortality risk
There are major racial differences in pregnancy-related mortality, with black women having much higher rates than white women. In the United States in 2011, the pregnancy-related mortality ratio for white, black, and women of other races was 12.5, 42.8, and 17.3 deaths per 100,000 live births, respectively. This represents a major racial disparity in pregnancy outcomes.1
The age of the mother is an important determinant of the risk of maternal death. Women younger than age 35 years have the lowest risk of maternal death. From 2006 to 2010, pregnant women older than age 40 had a risk of death approximately 3 times greater than women aged 34 or younger.2
References
- Pregnancy Mortality Surveillance System. Centers for Disease Control and Prevention Web site. http://www .cdc.gov/reproductivehealth/maternalinfanthealth/pmss.html. Accessed August 20, 2015.
- Creanga AA, Berg CJ, Syverson C, Seed K, Bruce FC, Callaghan WM. Pregnancy-related mortality in the United States, 2006-2010. Obstet Gynecol. 2015;125(1):5−12.
Let’s get organized
In a country with a history of embracing the “live free or die” ethic, it is often difficult for physicians to enthusiastically embrace the need for a higher level of organization and a potential reduction in individual freedom in order to improve health outcomes. And with a US maternal mortality ratio of 1 maternal death for every 5,400 births, many obstetricians will never have one of their patients die in childbirth. In fact, most obstetricians will have only 1 maternal death during their entire career. In this reality, when clinical events occur rarely, it is not possible for any single clinician, working alone, to impact the overall outcomes of those rare events. Therefore, teamwork and national efforts, such as the National Partnership for Maternal Safety,13 will be necessary to reverse our alarming trend of increasing maternal mortality. Let’s get organized to stop the rise of maternal deaths in the United States.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Chescheir NC. Enough already! Obstet Gynecol. 2015;125(1):2−4.
- Council on Resident Education in Obstetrics and Gynecology (CREOG) Educational Objectives: Core Curriculum in Obstetrics and Gynecology. 10th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2013:140.
- Callaghan WM, Grobman WA, Kilpatrick SJ, Main EK, D’Alton M. Facility-based identification of women with severe maternal morbidity: it is time to start. Obstet Gynecol. 2014;123(5):978−981.
- Silver RM, Fox KA, Barton JR, et al. Center of excellence for placenta accreta. Am J Obstet Gynecol. 2015;212(5):561−568.
- American College of Obstetricians and Gynecologists and the Society of Maternal Fetal Medicine. Obstetric care consensus No 2: levels of maternal care. Obstet Gynecol. 2015;125(2):502−515.
- Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy-related mortality in the United States, 1998−2005. Obstet Gynecol. 2010;116(6):1302−1309.
- Creanga AA, Berg CJ, Syverson C, Seed K, Bruce FC, Callaghan WM. Pregnancy-related mortality in the United States, 2006−2010. Obstet Gynecol. 2015;125(1):5−12.
- Pregnancy Mortality Surveillance System. Centers for Disease Control and Prevention Web site. http://www.cdc.gov/reproductivehealth/maternalinfanthealth/pmss.html. Accessed August 20, 2015.
- Shields LE, Wiesner S, Fulton J, Pelletreau B. Comprehensive maternal hemorrhage protocols reduce the use of blood products and improve patient safety. Am J Obstet Gynecol. 2015;212(3):272−280.
- James A. Committee on Practice Bulletins—Obstetrics. Practice bulletin No. 123: thromboembolism in pregnancy. ACOG. Obstet Gynecol. 2011;118(3):718−729.
- Thompson JL, Kuklina EV, Bateman BT, Callaghan WM, James AH, Grotegut CA. Medical and obstetrical outcomes among pregnant women with congenital heart disease. Obstet Gynecol. 2015;126(2):346−354.
- Lindley KJ, Madden T, Cahill AG, Ludbrook PA, Billadello JJ. Contraceptive use and unintended pregnancy in women with congenital heart disease. Obstet Gynecol. 2015;126(2):363−369.
- D’Alton ME, Main EK, Menard MK, Levy BS. The National Partnership for Maternal Safety. Obstet Gynecol. 2014;123(5):973−977.
A mother’s untimely death in childbirth is a grave loss that sends shock waves of grief across generations of her family and community. As obstetricians practicing in the United States, we face a terrible problem. We have a continually rising rate of maternal death in a country with exceptional medical resources (FIGURE).1 Our national decentralized approach to dealing with maternal mortality is a factor contributing to the decades-long increase in the maternal mortality ratio. Let’s get organized to better respond to this public health crisis.
Medical education— Let’s get focused on maternal mortality
The 140-page Council on Resident Education in Obstetrics and Gynecology CREOG Educational Objectives: Core Curriculum in Obstetrics and Gynecology provides a detailed enumeration of the key learning objectives for residents in obstetrics and gynecology.2 Surprisingly, the CREOG objectives do not mention reducing maternal mortality as an important curricular goal. Learning clinical processes and practices that decrease the risk of maternal mortality should be an important educational goal for all residents training in obstetrics and gynecology.
Nationwide action is needed to address the problem
Many countries have organized widespread efforts to reduce maternal mortality. In the United Kingdom and France there are nationwide reviews of maternal deaths with detailed analyses of clinical events and identification of areas for future improvement. These reviews result in the dissemination of countrywide clinical recommendations that change practice and hopefully reduce the risk of future maternal deaths. For example, following the identification of pulmonary embolism as a leading cause of maternal death in the United Kingdom there was a nationwide effort to increase the use of mechanical and pharmacologic prophylaxis to prevent deep venous thrombosis.
In the United States, experts have proposed that a national program of clinical review of severe maternal morbidity cases should be mandatory. (There are many more cases of “near misses” with severe maternal morbidity than there are maternal deaths.) The greater number of cases available for review should help institutions to quickly recognize potential areas for clinical improvement. One group of experts has recommended that all deliveries in which a pregnant woman received 4 or more units of blood or was admitted to an intensive care unit should be thoroughly reviewed to identify opportunities for clinical improvement.3
In the United Kingdom a contemporary clinical problem that is being addressed in an organized and systematic manner is how to respond to the rising rate of severe maternal morbidity caused by placenta accreta. Experts have concluded that women with a suspected placenta accreta should deliver in regional centers with advanced clinical resources—including an emergency surgical response team, interventional radiology, a high capacity blood bank, and an intensive care unit.
A similar approach has been proposed for managing placenta accreta in the United States.4 The American College of Obstetricians and Gynecologists (ACOG) and the Society of Maternal Fetal Medicine (SMFM) have proposed a tiered system of obstetric care with more complex cases being referred to regional perinatal centers.5 Regionalization of trauma services has been an important part of the US health care system for decades. Cases of severe trauma are brought to regional centers equipped to emergently treat complex injuries. A similar system of regulation and regionalization could be adapted for optimizing maternity care.
High-risk clinical events: Is your unit prepared?
In the United States the leading causes of maternal mortality, in descending order, are6−8:
- cardiovascular diseases
- infection
- hemorrhage
- cardiomyopathy
- pulmonary embolism
- hypertension
- amniotic fluid embolism
- stroke
- anesthesia complications.
Over the last decade, the Joint Commission has recommended that birthing centers develop standardized protocols and use simulation to improve the institution’s ability to respond in a timely manner to clinical events that may result in maternal morbidity or death.
The quality of published protocols dealing with hemorrhage, hypertension, and thromboembolism is continuously improving, and every birthing center should have written protocols that are updated on a regular timetable for these common high-risk events.9,10 Does your birthing unit have written protocols to deal with cardiac diseases, infection, obstetric hemorrhage, thromboembolism, and severe hypertension? Are simulation exercises used to strengthen familiarity with the protocols?
High-risk patients
An amazing fact of today’s medical care is that sexually active women of reproductive age who have high-risk medical problems often have not been counseled to use a highly effective contraceptive, resulting in an increased risk of unintended pregnancy and maternal death. For example, adult women with a history of congenital heart disease are known to be at increased risk of death if they become pregnant. In a recent study, women with a history of congenital heart disease had 178 maternal deaths per 100,000 deliveries—a rate approximately 10-fold higher than the US maternal mortality ratio.11 Yet, many of these women are not using a highly effective contraceptive, and this results in a high rate of unplanned pregnancy.12
In order to reduce the risk of unintended pregnancy in women with high-risk medical problems, health systems could make contraception an important “vital sign” for women with high-risk medical conditions.
Race and age matter greatly when it comes to maternal mortality risk
There are major racial differences in pregnancy-related mortality, with black women having much higher rates than white women. In the United States in 2011, the pregnancy-related mortality ratio for white, black, and women of other races was 12.5, 42.8, and 17.3 deaths per 100,000 live births, respectively. This represents a major racial disparity in pregnancy outcomes.1
The age of the mother is an important determinant of the risk of maternal death. Women younger than age 35 years have the lowest risk of maternal death. From 2006 to 2010, pregnant women older than age 40 had a risk of death approximately 3 times greater than women aged 34 or younger.2
References
- Pregnancy Mortality Surveillance System. Centers for Disease Control and Prevention Web site. http://www .cdc.gov/reproductivehealth/maternalinfanthealth/pmss.html. Accessed August 20, 2015.
- Creanga AA, Berg CJ, Syverson C, Seed K, Bruce FC, Callaghan WM. Pregnancy-related mortality in the United States, 2006-2010. Obstet Gynecol. 2015;125(1):5−12.
Let’s get organized
In a country with a history of embracing the “live free or die” ethic, it is often difficult for physicians to enthusiastically embrace the need for a higher level of organization and a potential reduction in individual freedom in order to improve health outcomes. And with a US maternal mortality ratio of 1 maternal death for every 5,400 births, many obstetricians will never have one of their patients die in childbirth. In fact, most obstetricians will have only 1 maternal death during their entire career. In this reality, when clinical events occur rarely, it is not possible for any single clinician, working alone, to impact the overall outcomes of those rare events. Therefore, teamwork and national efforts, such as the National Partnership for Maternal Safety,13 will be necessary to reverse our alarming trend of increasing maternal mortality. Let’s get organized to stop the rise of maternal deaths in the United States.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
A mother’s untimely death in childbirth is a grave loss that sends shock waves of grief across generations of her family and community. As obstetricians practicing in the United States, we face a terrible problem. We have a continually rising rate of maternal death in a country with exceptional medical resources (FIGURE).1 Our national decentralized approach to dealing with maternal mortality is a factor contributing to the decades-long increase in the maternal mortality ratio. Let’s get organized to better respond to this public health crisis.
Medical education— Let’s get focused on maternal mortality
The 140-page Council on Resident Education in Obstetrics and Gynecology CREOG Educational Objectives: Core Curriculum in Obstetrics and Gynecology provides a detailed enumeration of the key learning objectives for residents in obstetrics and gynecology.2 Surprisingly, the CREOG objectives do not mention reducing maternal mortality as an important curricular goal. Learning clinical processes and practices that decrease the risk of maternal mortality should be an important educational goal for all residents training in obstetrics and gynecology.
Nationwide action is needed to address the problem
Many countries have organized widespread efforts to reduce maternal mortality. In the United Kingdom and France there are nationwide reviews of maternal deaths with detailed analyses of clinical events and identification of areas for future improvement. These reviews result in the dissemination of countrywide clinical recommendations that change practice and hopefully reduce the risk of future maternal deaths. For example, following the identification of pulmonary embolism as a leading cause of maternal death in the United Kingdom there was a nationwide effort to increase the use of mechanical and pharmacologic prophylaxis to prevent deep venous thrombosis.
In the United States, experts have proposed that a national program of clinical review of severe maternal morbidity cases should be mandatory. (There are many more cases of “near misses” with severe maternal morbidity than there are maternal deaths.) The greater number of cases available for review should help institutions to quickly recognize potential areas for clinical improvement. One group of experts has recommended that all deliveries in which a pregnant woman received 4 or more units of blood or was admitted to an intensive care unit should be thoroughly reviewed to identify opportunities for clinical improvement.3
In the United Kingdom a contemporary clinical problem that is being addressed in an organized and systematic manner is how to respond to the rising rate of severe maternal morbidity caused by placenta accreta. Experts have concluded that women with a suspected placenta accreta should deliver in regional centers with advanced clinical resources—including an emergency surgical response team, interventional radiology, a high capacity blood bank, and an intensive care unit.
A similar approach has been proposed for managing placenta accreta in the United States.4 The American College of Obstetricians and Gynecologists (ACOG) and the Society of Maternal Fetal Medicine (SMFM) have proposed a tiered system of obstetric care with more complex cases being referred to regional perinatal centers.5 Regionalization of trauma services has been an important part of the US health care system for decades. Cases of severe trauma are brought to regional centers equipped to emergently treat complex injuries. A similar system of regulation and regionalization could be adapted for optimizing maternity care.
High-risk clinical events: Is your unit prepared?
In the United States the leading causes of maternal mortality, in descending order, are6−8:
- cardiovascular diseases
- infection
- hemorrhage
- cardiomyopathy
- pulmonary embolism
- hypertension
- amniotic fluid embolism
- stroke
- anesthesia complications.
Over the last decade, the Joint Commission has recommended that birthing centers develop standardized protocols and use simulation to improve the institution’s ability to respond in a timely manner to clinical events that may result in maternal morbidity or death.
The quality of published protocols dealing with hemorrhage, hypertension, and thromboembolism is continuously improving, and every birthing center should have written protocols that are updated on a regular timetable for these common high-risk events.9,10 Does your birthing unit have written protocols to deal with cardiac diseases, infection, obstetric hemorrhage, thromboembolism, and severe hypertension? Are simulation exercises used to strengthen familiarity with the protocols?
High-risk patients
An amazing fact of today’s medical care is that sexually active women of reproductive age who have high-risk medical problems often have not been counseled to use a highly effective contraceptive, resulting in an increased risk of unintended pregnancy and maternal death. For example, adult women with a history of congenital heart disease are known to be at increased risk of death if they become pregnant. In a recent study, women with a history of congenital heart disease had 178 maternal deaths per 100,000 deliveries—a rate approximately 10-fold higher than the US maternal mortality ratio.11 Yet, many of these women are not using a highly effective contraceptive, and this results in a high rate of unplanned pregnancy.12
In order to reduce the risk of unintended pregnancy in women with high-risk medical problems, health systems could make contraception an important “vital sign” for women with high-risk medical conditions.
Race and age matter greatly when it comes to maternal mortality risk
There are major racial differences in pregnancy-related mortality, with black women having much higher rates than white women. In the United States in 2011, the pregnancy-related mortality ratio for white, black, and women of other races was 12.5, 42.8, and 17.3 deaths per 100,000 live births, respectively. This represents a major racial disparity in pregnancy outcomes.1
The age of the mother is an important determinant of the risk of maternal death. Women younger than age 35 years have the lowest risk of maternal death. From 2006 to 2010, pregnant women older than age 40 had a risk of death approximately 3 times greater than women aged 34 or younger.2
References
- Pregnancy Mortality Surveillance System. Centers for Disease Control and Prevention Web site. http://www .cdc.gov/reproductivehealth/maternalinfanthealth/pmss.html. Accessed August 20, 2015.
- Creanga AA, Berg CJ, Syverson C, Seed K, Bruce FC, Callaghan WM. Pregnancy-related mortality in the United States, 2006-2010. Obstet Gynecol. 2015;125(1):5−12.
Let’s get organized
In a country with a history of embracing the “live free or die” ethic, it is often difficult for physicians to enthusiastically embrace the need for a higher level of organization and a potential reduction in individual freedom in order to improve health outcomes. And with a US maternal mortality ratio of 1 maternal death for every 5,400 births, many obstetricians will never have one of their patients die in childbirth. In fact, most obstetricians will have only 1 maternal death during their entire career. In this reality, when clinical events occur rarely, it is not possible for any single clinician, working alone, to impact the overall outcomes of those rare events. Therefore, teamwork and national efforts, such as the National Partnership for Maternal Safety,13 will be necessary to reverse our alarming trend of increasing maternal mortality. Let’s get organized to stop the rise of maternal deaths in the United States.
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Chescheir NC. Enough already! Obstet Gynecol. 2015;125(1):2−4.
- Council on Resident Education in Obstetrics and Gynecology (CREOG) Educational Objectives: Core Curriculum in Obstetrics and Gynecology. 10th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2013:140.
- Callaghan WM, Grobman WA, Kilpatrick SJ, Main EK, D’Alton M. Facility-based identification of women with severe maternal morbidity: it is time to start. Obstet Gynecol. 2014;123(5):978−981.
- Silver RM, Fox KA, Barton JR, et al. Center of excellence for placenta accreta. Am J Obstet Gynecol. 2015;212(5):561−568.
- American College of Obstetricians and Gynecologists and the Society of Maternal Fetal Medicine. Obstetric care consensus No 2: levels of maternal care. Obstet Gynecol. 2015;125(2):502−515.
- Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy-related mortality in the United States, 1998−2005. Obstet Gynecol. 2010;116(6):1302−1309.
- Creanga AA, Berg CJ, Syverson C, Seed K, Bruce FC, Callaghan WM. Pregnancy-related mortality in the United States, 2006−2010. Obstet Gynecol. 2015;125(1):5−12.
- Pregnancy Mortality Surveillance System. Centers for Disease Control and Prevention Web site. http://www.cdc.gov/reproductivehealth/maternalinfanthealth/pmss.html. Accessed August 20, 2015.
- Shields LE, Wiesner S, Fulton J, Pelletreau B. Comprehensive maternal hemorrhage protocols reduce the use of blood products and improve patient safety. Am J Obstet Gynecol. 2015;212(3):272−280.
- James A. Committee on Practice Bulletins—Obstetrics. Practice bulletin No. 123: thromboembolism in pregnancy. ACOG. Obstet Gynecol. 2011;118(3):718−729.
- Thompson JL, Kuklina EV, Bateman BT, Callaghan WM, James AH, Grotegut CA. Medical and obstetrical outcomes among pregnant women with congenital heart disease. Obstet Gynecol. 2015;126(2):346−354.
- Lindley KJ, Madden T, Cahill AG, Ludbrook PA, Billadello JJ. Contraceptive use and unintended pregnancy in women with congenital heart disease. Obstet Gynecol. 2015;126(2):363−369.
- D’Alton ME, Main EK, Menard MK, Levy BS. The National Partnership for Maternal Safety. Obstet Gynecol. 2014;123(5):973−977.
- Chescheir NC. Enough already! Obstet Gynecol. 2015;125(1):2−4.
- Council on Resident Education in Obstetrics and Gynecology (CREOG) Educational Objectives: Core Curriculum in Obstetrics and Gynecology. 10th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2013:140.
- Callaghan WM, Grobman WA, Kilpatrick SJ, Main EK, D’Alton M. Facility-based identification of women with severe maternal morbidity: it is time to start. Obstet Gynecol. 2014;123(5):978−981.
- Silver RM, Fox KA, Barton JR, et al. Center of excellence for placenta accreta. Am J Obstet Gynecol. 2015;212(5):561−568.
- American College of Obstetricians and Gynecologists and the Society of Maternal Fetal Medicine. Obstetric care consensus No 2: levels of maternal care. Obstet Gynecol. 2015;125(2):502−515.
- Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy-related mortality in the United States, 1998−2005. Obstet Gynecol. 2010;116(6):1302−1309.
- Creanga AA, Berg CJ, Syverson C, Seed K, Bruce FC, Callaghan WM. Pregnancy-related mortality in the United States, 2006−2010. Obstet Gynecol. 2015;125(1):5−12.
- Pregnancy Mortality Surveillance System. Centers for Disease Control and Prevention Web site. http://www.cdc.gov/reproductivehealth/maternalinfanthealth/pmss.html. Accessed August 20, 2015.
- Shields LE, Wiesner S, Fulton J, Pelletreau B. Comprehensive maternal hemorrhage protocols reduce the use of blood products and improve patient safety. Am J Obstet Gynecol. 2015;212(3):272−280.
- James A. Committee on Practice Bulletins—Obstetrics. Practice bulletin No. 123: thromboembolism in pregnancy. ACOG. Obstet Gynecol. 2011;118(3):718−729.
- Thompson JL, Kuklina EV, Bateman BT, Callaghan WM, James AH, Grotegut CA. Medical and obstetrical outcomes among pregnant women with congenital heart disease. Obstet Gynecol. 2015;126(2):346−354.
- Lindley KJ, Madden T, Cahill AG, Ludbrook PA, Billadello JJ. Contraceptive use and unintended pregnancy in women with congenital heart disease. Obstet Gynecol. 2015;126(2):363−369.
- D’Alton ME, Main EK, Menard MK, Levy BS. The National Partnership for Maternal Safety. Obstet Gynecol. 2014;123(5):973−977.
Perioperative MI: Data, practice, and questions
Except in emergency or specific high-risk surgery, or for extremely fragile high-risk patients, we anticipate a successful outcome from noncardiac surgery. The skills and tools of our anesthesiology colleagues have advanced to the point that severe intraoperative and immediate postoperative complications are rare.
Preoperative risk assessment and perioperative medical management in large medical centers are now largely done by hospital-based physicians with interest and expertise in this subspecialty, and are integrated into the care of the surgical patient. This has likely contributed to improved patient outcomes. Yet postoperative cardiovascular events still cause significant morbidity (although they generally occur in less than 10% of patients).
The entity of perioperative myocardial infarction (MI) has an interesting history. We have recognized for several decades that its presentation is often different than the usually diagnosed MI: perioperative MI is often painless and may manifest as unexplained sinus tachycardia, subtle changes in mental status, or mild dyspnea. These symptoms, if they occurred while the patient was at home, would often be mild enough that the patient would not seek immediate medical attention. Autopsy studies suggested that many of these MIs result from a different pathophysiology than the garden variety MI; plaque rupture with or without secondary thrombosis may be less common than myocardial injury resulting from an imbalance between cardiac demand and blood flow. Studies initially suggested that postoperative MI occurred many days after the surgery. But as tests to diagnose myocyte injury became more sensitive (electrocardiography, creatine kinase, creatine kinase MB, and now troponin), it was recognized that cardiac injury actually occurred very soon after or even during surgery.
With the advent of highly sensitive and fairly specific troponin assays, it seems that perioperative cardiac injury is extremely common, perhaps occurring in up to 20% of patients (if we include patients at high risk based on traditional criteria). This has led to the newly described entity of “myocardial injury after noncardiac surgery” (MINS). MINS patients, diagnosed by troponin elevations, usually are asymptomatic, and many do not meet criteria for any type of MI. But strikingly, as discussed in this issue of the Journal by Horr et al, simply having a postoperative troponin elevation predicts an increased risk of clinical cardiovascular events and a decreased 30-day survival rate.
Adding postoperative troponin measurement to the usual preoperative screening protocol significantly increases our ability to predict delayed cardiovascular events and mortality. As pointed out by Cohn in his accompanying editorial, the benefit, if any, of screening low-risk patients remains to be defined. But an even more important issue, as commented upon in both papers, is what to do when an elevated troponin is detected in a postoperative patient who is otherwise doing perfectly well. Given our current knowledge of the pathophysiology of postoperative MI and the still overall low mortality, it seems unreasonable to immediately take all of these patients to the catheterization suite. Yet with current knowledge of the prognostic significance of troponin elevation, this can’t be ignored. Should all patients receive immediate high-intensity statin therapy, antiplatelet therapy if safe in the specific perioperative setting, and postdischarge physiologic stress studies, or should we “just” take it as a potential high-impact teaching moment and advise patients of their increased cardiovascular risk and offer our usual heart-healthy admonitions?
The confirmed observation that postoperative troponin elevation predicts morbidity and mortality over the subsequent 30 days, and perhaps even longer, has triggered the start of several interventional trials. The results of these will, hopefully, help us to further improve perioperative outcomes.
Except in emergency or specific high-risk surgery, or for extremely fragile high-risk patients, we anticipate a successful outcome from noncardiac surgery. The skills and tools of our anesthesiology colleagues have advanced to the point that severe intraoperative and immediate postoperative complications are rare.
Preoperative risk assessment and perioperative medical management in large medical centers are now largely done by hospital-based physicians with interest and expertise in this subspecialty, and are integrated into the care of the surgical patient. This has likely contributed to improved patient outcomes. Yet postoperative cardiovascular events still cause significant morbidity (although they generally occur in less than 10% of patients).
The entity of perioperative myocardial infarction (MI) has an interesting history. We have recognized for several decades that its presentation is often different than the usually diagnosed MI: perioperative MI is often painless and may manifest as unexplained sinus tachycardia, subtle changes in mental status, or mild dyspnea. These symptoms, if they occurred while the patient was at home, would often be mild enough that the patient would not seek immediate medical attention. Autopsy studies suggested that many of these MIs result from a different pathophysiology than the garden variety MI; plaque rupture with or without secondary thrombosis may be less common than myocardial injury resulting from an imbalance between cardiac demand and blood flow. Studies initially suggested that postoperative MI occurred many days after the surgery. But as tests to diagnose myocyte injury became more sensitive (electrocardiography, creatine kinase, creatine kinase MB, and now troponin), it was recognized that cardiac injury actually occurred very soon after or even during surgery.
With the advent of highly sensitive and fairly specific troponin assays, it seems that perioperative cardiac injury is extremely common, perhaps occurring in up to 20% of patients (if we include patients at high risk based on traditional criteria). This has led to the newly described entity of “myocardial injury after noncardiac surgery” (MINS). MINS patients, diagnosed by troponin elevations, usually are asymptomatic, and many do not meet criteria for any type of MI. But strikingly, as discussed in this issue of the Journal by Horr et al, simply having a postoperative troponin elevation predicts an increased risk of clinical cardiovascular events and a decreased 30-day survival rate.
Adding postoperative troponin measurement to the usual preoperative screening protocol significantly increases our ability to predict delayed cardiovascular events and mortality. As pointed out by Cohn in his accompanying editorial, the benefit, if any, of screening low-risk patients remains to be defined. But an even more important issue, as commented upon in both papers, is what to do when an elevated troponin is detected in a postoperative patient who is otherwise doing perfectly well. Given our current knowledge of the pathophysiology of postoperative MI and the still overall low mortality, it seems unreasonable to immediately take all of these patients to the catheterization suite. Yet with current knowledge of the prognostic significance of troponin elevation, this can’t be ignored. Should all patients receive immediate high-intensity statin therapy, antiplatelet therapy if safe in the specific perioperative setting, and postdischarge physiologic stress studies, or should we “just” take it as a potential high-impact teaching moment and advise patients of their increased cardiovascular risk and offer our usual heart-healthy admonitions?
The confirmed observation that postoperative troponin elevation predicts morbidity and mortality over the subsequent 30 days, and perhaps even longer, has triggered the start of several interventional trials. The results of these will, hopefully, help us to further improve perioperative outcomes.
Except in emergency or specific high-risk surgery, or for extremely fragile high-risk patients, we anticipate a successful outcome from noncardiac surgery. The skills and tools of our anesthesiology colleagues have advanced to the point that severe intraoperative and immediate postoperative complications are rare.
Preoperative risk assessment and perioperative medical management in large medical centers are now largely done by hospital-based physicians with interest and expertise in this subspecialty, and are integrated into the care of the surgical patient. This has likely contributed to improved patient outcomes. Yet postoperative cardiovascular events still cause significant morbidity (although they generally occur in less than 10% of patients).
The entity of perioperative myocardial infarction (MI) has an interesting history. We have recognized for several decades that its presentation is often different than the usually diagnosed MI: perioperative MI is often painless and may manifest as unexplained sinus tachycardia, subtle changes in mental status, or mild dyspnea. These symptoms, if they occurred while the patient was at home, would often be mild enough that the patient would not seek immediate medical attention. Autopsy studies suggested that many of these MIs result from a different pathophysiology than the garden variety MI; plaque rupture with or without secondary thrombosis may be less common than myocardial injury resulting from an imbalance between cardiac demand and blood flow. Studies initially suggested that postoperative MI occurred many days after the surgery. But as tests to diagnose myocyte injury became more sensitive (electrocardiography, creatine kinase, creatine kinase MB, and now troponin), it was recognized that cardiac injury actually occurred very soon after or even during surgery.
With the advent of highly sensitive and fairly specific troponin assays, it seems that perioperative cardiac injury is extremely common, perhaps occurring in up to 20% of patients (if we include patients at high risk based on traditional criteria). This has led to the newly described entity of “myocardial injury after noncardiac surgery” (MINS). MINS patients, diagnosed by troponin elevations, usually are asymptomatic, and many do not meet criteria for any type of MI. But strikingly, as discussed in this issue of the Journal by Horr et al, simply having a postoperative troponin elevation predicts an increased risk of clinical cardiovascular events and a decreased 30-day survival rate.
Adding postoperative troponin measurement to the usual preoperative screening protocol significantly increases our ability to predict delayed cardiovascular events and mortality. As pointed out by Cohn in his accompanying editorial, the benefit, if any, of screening low-risk patients remains to be defined. But an even more important issue, as commented upon in both papers, is what to do when an elevated troponin is detected in a postoperative patient who is otherwise doing perfectly well. Given our current knowledge of the pathophysiology of postoperative MI and the still overall low mortality, it seems unreasonable to immediately take all of these patients to the catheterization suite. Yet with current knowledge of the prognostic significance of troponin elevation, this can’t be ignored. Should all patients receive immediate high-intensity statin therapy, antiplatelet therapy if safe in the specific perioperative setting, and postdischarge physiologic stress studies, or should we “just” take it as a potential high-impact teaching moment and advise patients of their increased cardiovascular risk and offer our usual heart-healthy admonitions?
The confirmed observation that postoperative troponin elevation predicts morbidity and mortality over the subsequent 30 days, and perhaps even longer, has triggered the start of several interventional trials. The results of these will, hopefully, help us to further improve perioperative outcomes.
Re-personalizing precision medicine: is there a role for patient-reported outcomes?
In the opinion of most, precision medicine is the future of cancer therapeutics. By producing response rates well into double digits, and substantially extending progression-free and overall survival, the molecular testing of tumors to select optimal treatment may be a way to justify the high cost of new and emerging therapeutics. The road to this future will likely be long and winding, however, with a string of incremental successes amid inevitable disappointments. Our patients will walk this road with us, agreeing to testing and treatment when those tests come back positive for an eligible mutation.
Click on the PDF icon at the top of this introduction to read the full article.
In the opinion of most, precision medicine is the future of cancer therapeutics. By producing response rates well into double digits, and substantially extending progression-free and overall survival, the molecular testing of tumors to select optimal treatment may be a way to justify the high cost of new and emerging therapeutics. The road to this future will likely be long and winding, however, with a string of incremental successes amid inevitable disappointments. Our patients will walk this road with us, agreeing to testing and treatment when those tests come back positive for an eligible mutation.
Click on the PDF icon at the top of this introduction to read the full article.
In the opinion of most, precision medicine is the future of cancer therapeutics. By producing response rates well into double digits, and substantially extending progression-free and overall survival, the molecular testing of tumors to select optimal treatment may be a way to justify the high cost of new and emerging therapeutics. The road to this future will likely be long and winding, however, with a string of incremental successes amid inevitable disappointments. Our patients will walk this road with us, agreeing to testing and treatment when those tests come back positive for an eligible mutation.
Click on the PDF icon at the top of this introduction to read the full article.
Remembering that old dogs can still do tricks
More and more we are realizing that we need trials that use hard clinical end points to inform our clinical practice. Several things we used to do based on observational studies have fallen from grace after being evaluated in interventional trials. And faced with the US Food and Drug Administration’s mandate to demonstrate clinical impact, pharmaceutical companies can rarely count on using even well-accepted biomarkers instead of clinical outcomes when trying to bring new drugs to market.
This atmosphere often makes us a bit uncomfortable when prescribing older drugs that have passed the test of time and collective anecdotal experience but not rigorous clinical testing. In some cases this is good, and robust evaluation provides greater confidence in our choice of therapy: witness the demise of digoxin for heart failure.
Many older drugs have never been compared with newer drugs in well-designed trials using hard clinical outcomes and likely never will, owing to cost, marketing, and logistic reasons. But sometimes these trials are done, and the results are surprising. For instance, methotrexate in appropriate doses may actually be comparable to newer and far more expensive tumor necrosis factor inhibitors when used to treat rheumatoid arthritis.
Should we be willing to sometimes accept data on surrogate markers (eg, low-density lipoprotein cholesterol levels, blood pressure, hemoglobin A1c ) or even extensive clinical experience in the absence of hard outcome data when using older, tried-and-true drugs? Markers can mislead: consider the higher number of deaths recorded in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial in the group receiving more aggressive control of their glucose levels.
So we should not be totally sanguine when using older drugs instead of newer ones. But some drugs may have slipped out of our mental formularies yet still have real value in niche or even common settings. Methyldopa remains an effective antihypertensive drug and may be especially useful in peripartum patients. Yet relatively few young physicians know the drug.
And so it may be with chlorthalidone. In this issue of the Journal, Cooney et al remind us not only that this drug is still around, but that it has proven efficacy and, compared with its more popular cousin hydrochlorothiazide, favorable pharmacokinetic properties such as longer action. Not to mention that it was a comparator drug in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT) trial.
In our current cost-saving environment, we should remember that some old dogs can still do good tricks.
More and more we are realizing that we need trials that use hard clinical end points to inform our clinical practice. Several things we used to do based on observational studies have fallen from grace after being evaluated in interventional trials. And faced with the US Food and Drug Administration’s mandate to demonstrate clinical impact, pharmaceutical companies can rarely count on using even well-accepted biomarkers instead of clinical outcomes when trying to bring new drugs to market.
This atmosphere often makes us a bit uncomfortable when prescribing older drugs that have passed the test of time and collective anecdotal experience but not rigorous clinical testing. In some cases this is good, and robust evaluation provides greater confidence in our choice of therapy: witness the demise of digoxin for heart failure.
Many older drugs have never been compared with newer drugs in well-designed trials using hard clinical outcomes and likely never will, owing to cost, marketing, and logistic reasons. But sometimes these trials are done, and the results are surprising. For instance, methotrexate in appropriate doses may actually be comparable to newer and far more expensive tumor necrosis factor inhibitors when used to treat rheumatoid arthritis.
Should we be willing to sometimes accept data on surrogate markers (eg, low-density lipoprotein cholesterol levels, blood pressure, hemoglobin A1c ) or even extensive clinical experience in the absence of hard outcome data when using older, tried-and-true drugs? Markers can mislead: consider the higher number of deaths recorded in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial in the group receiving more aggressive control of their glucose levels.
So we should not be totally sanguine when using older drugs instead of newer ones. But some drugs may have slipped out of our mental formularies yet still have real value in niche or even common settings. Methyldopa remains an effective antihypertensive drug and may be especially useful in peripartum patients. Yet relatively few young physicians know the drug.
And so it may be with chlorthalidone. In this issue of the Journal, Cooney et al remind us not only that this drug is still around, but that it has proven efficacy and, compared with its more popular cousin hydrochlorothiazide, favorable pharmacokinetic properties such as longer action. Not to mention that it was a comparator drug in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT) trial.
In our current cost-saving environment, we should remember that some old dogs can still do good tricks.
More and more we are realizing that we need trials that use hard clinical end points to inform our clinical practice. Several things we used to do based on observational studies have fallen from grace after being evaluated in interventional trials. And faced with the US Food and Drug Administration’s mandate to demonstrate clinical impact, pharmaceutical companies can rarely count on using even well-accepted biomarkers instead of clinical outcomes when trying to bring new drugs to market.
This atmosphere often makes us a bit uncomfortable when prescribing older drugs that have passed the test of time and collective anecdotal experience but not rigorous clinical testing. In some cases this is good, and robust evaluation provides greater confidence in our choice of therapy: witness the demise of digoxin for heart failure.
Many older drugs have never been compared with newer drugs in well-designed trials using hard clinical outcomes and likely never will, owing to cost, marketing, and logistic reasons. But sometimes these trials are done, and the results are surprising. For instance, methotrexate in appropriate doses may actually be comparable to newer and far more expensive tumor necrosis factor inhibitors when used to treat rheumatoid arthritis.
Should we be willing to sometimes accept data on surrogate markers (eg, low-density lipoprotein cholesterol levels, blood pressure, hemoglobin A1c ) or even extensive clinical experience in the absence of hard outcome data when using older, tried-and-true drugs? Markers can mislead: consider the higher number of deaths recorded in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial in the group receiving more aggressive control of their glucose levels.
So we should not be totally sanguine when using older drugs instead of newer ones. But some drugs may have slipped out of our mental formularies yet still have real value in niche or even common settings. Methyldopa remains an effective antihypertensive drug and may be especially useful in peripartum patients. Yet relatively few young physicians know the drug.
And so it may be with chlorthalidone. In this issue of the Journal, Cooney et al remind us not only that this drug is still around, but that it has proven efficacy and, compared with its more popular cousin hydrochlorothiazide, favorable pharmacokinetic properties such as longer action. Not to mention that it was a comparator drug in the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack (ALLHAT) trial.
In our current cost-saving environment, we should remember that some old dogs can still do good tricks.