User login
The simple lab test is sometimes more complex than we think, if we think about it at all
We are all exposed to initiatives from multiple stakeholders telling us to order fewer tests. Many of these efforts to control costs and improve efficiency and quality of care are directed at populations of patients and are broad concepts: provide screening only to those most likely to benefit (eg, don’t screen for prostate cancer in men with a lifespan < 10 years); avoid procedures that provided limited benefit in controlled trials (eg, limit routine arthroscopic treatment of knee osteoarthritis); and avoid reflexive practices unlikely to improve patient outcomes (eg, eliminate routine preoperative testing before elective procedures in otherwise healthy patients).
Whether system-based changes will be implemented and have an impact remains to be determined. But I sense that with all the attention being focused on population management and healthcare practices, including an emphasis on documenting and coding our encounters with patients, whether substantive or simply digital housekeeping, we are increasingly distracted from the patient in front of us and are spending less time reviewing the principles underlying the diagnoses we make and the tests we order—just as we are taking less time to perform relevant physical examinations.1 The latter may mostly relate to time pressures. The former, I believe, is a product of both time pressures and a false sense of confidence in our knowledge of seemingly commonplace laboratory tests.
As I lecture, work with trainees, and reflect on my own patients, I realize that we are slowly but progressively minimizing the importance of a working knowledge of the basic foundations of clinical practice—perhaps because facts can always be looked up. I am not referring to knowledge of arcane biochemical pathways, eponymous references, or the latest recommended treatment of inclusion body myositis. I am thinking instead of the value of regularly refreshing our knowledge of laboratory tests and diagnoses we frequently encounter.
Having access to multiple clinical databases literally in our pockets is likely bolstering a false sense of confidence in our knowledge. The National Library of Medicine may be only a tap on a smart phone away, but accessing it regularly is a different thing. Attending conferences and reading educational journals help to keep our broad-based knowledge of internal medicine refreshed, but time pressures may significantly limit our ability to regularly pursue these activities.
In this issue of the Journal, Drs. Moghadam-Kia et al discuss their approach to asymptomatic elevations in creatine kinase (CK). Although no longer included in the most commonly used lab panels, CK measurement is often ordered in patients taking statins, even if they have no relevant muscle-related symptoms. Thus, evaluating a patient with an asymptomatic elevated CK level is not rare. The authors delve into the clinically relevant test characteristics, and their important caveats about interpreting elevated CK levels are germane not only to the asymptomatic patient, but also to the patient being evaluated for myalgia or weakness. This latter situation is one I frequently face in both the hospital and the outpatient clinic. I am often asked to consult on patients who have incompletely defined symptoms and elevated CK.
As discussed in the article, the laboratory definition of “normal” must first be considered. Laboratory test results must always be interpreted in the clinical context. An isolated elevation in parathyroid hormone cannot be interpreted without knowing the patient’s vitamin D level. Nor can “normal” low-density lipoprotein or serum urate levels be interpreted properly without knowing if the patient is accumulating excess cholesterol or urate deposits. As we order and interpret test results, we must consider the biology of the substance being measured as well as the test characteristics; too often, we react to abnormal laboratory results with an incomplete understanding of these aspects.
Moghadam-Kia et al do not dwell on the organ involvement causing CK elevations, but specificity is another very important aspect when clinically interpreting the results of a CK test. Many patients with muscle damage or inflammation have elevations in serum aspartate aminotransferase and alanine aminotransferase levels (the ratio of elevation depends on the time course of the muscle damage and on the relative clearance rate of the two enzymes). Without knowing that the CK is elevated, one might assume that an aminotransferase elevation reflects hepatitis. I have seen several patients with elevated aminotransferases and complaints of weakness and fatigue who were subjected to liver biopsy before it was recognized that the source of the enzyme elevation (“liver function test changes”) was muscle (or hemolysis). Frequently unrecognized is that aldolase, which has a cell distribution similar to that of lactate dehydrogenase, does not have the relative specificity of localization to muscle that CK has. CK is quite useful in distinguishing myocyte from hepatocyte damage.
This paper presents a wonderful reminder of the value of updating and reviewing what we know about tests that we order, even if we feel comfortable when ordering them. Before initiating a cascade of additional tests and consultations to explore the cause of an abnormal test result, a little time spent reviewing its basic characteristics and biology may pay dividends.
As 2015 comes to a close, we at the Journal share with you our sincere wishes for personal satisfaction and a globally more peaceful 2016.
- Verghese A, Charlton B, Kassirer JP, Ramsey M, Ioannidis JP. Inadequacies of physical examination as a cause of medical errors and adverse events: a collection of vignettes. Am J Med 2015; 128:1322–1324.
We are all exposed to initiatives from multiple stakeholders telling us to order fewer tests. Many of these efforts to control costs and improve efficiency and quality of care are directed at populations of patients and are broad concepts: provide screening only to those most likely to benefit (eg, don’t screen for prostate cancer in men with a lifespan < 10 years); avoid procedures that provided limited benefit in controlled trials (eg, limit routine arthroscopic treatment of knee osteoarthritis); and avoid reflexive practices unlikely to improve patient outcomes (eg, eliminate routine preoperative testing before elective procedures in otherwise healthy patients).
Whether system-based changes will be implemented and have an impact remains to be determined. But I sense that with all the attention being focused on population management and healthcare practices, including an emphasis on documenting and coding our encounters with patients, whether substantive or simply digital housekeeping, we are increasingly distracted from the patient in front of us and are spending less time reviewing the principles underlying the diagnoses we make and the tests we order—just as we are taking less time to perform relevant physical examinations.1 The latter may mostly relate to time pressures. The former, I believe, is a product of both time pressures and a false sense of confidence in our knowledge of seemingly commonplace laboratory tests.
As I lecture, work with trainees, and reflect on my own patients, I realize that we are slowly but progressively minimizing the importance of a working knowledge of the basic foundations of clinical practice—perhaps because facts can always be looked up. I am not referring to knowledge of arcane biochemical pathways, eponymous references, or the latest recommended treatment of inclusion body myositis. I am thinking instead of the value of regularly refreshing our knowledge of laboratory tests and diagnoses we frequently encounter.
Having access to multiple clinical databases literally in our pockets is likely bolstering a false sense of confidence in our knowledge. The National Library of Medicine may be only a tap on a smart phone away, but accessing it regularly is a different thing. Attending conferences and reading educational journals help to keep our broad-based knowledge of internal medicine refreshed, but time pressures may significantly limit our ability to regularly pursue these activities.
In this issue of the Journal, Drs. Moghadam-Kia et al discuss their approach to asymptomatic elevations in creatine kinase (CK). Although no longer included in the most commonly used lab panels, CK measurement is often ordered in patients taking statins, even if they have no relevant muscle-related symptoms. Thus, evaluating a patient with an asymptomatic elevated CK level is not rare. The authors delve into the clinically relevant test characteristics, and their important caveats about interpreting elevated CK levels are germane not only to the asymptomatic patient, but also to the patient being evaluated for myalgia or weakness. This latter situation is one I frequently face in both the hospital and the outpatient clinic. I am often asked to consult on patients who have incompletely defined symptoms and elevated CK.
As discussed in the article, the laboratory definition of “normal” must first be considered. Laboratory test results must always be interpreted in the clinical context. An isolated elevation in parathyroid hormone cannot be interpreted without knowing the patient’s vitamin D level. Nor can “normal” low-density lipoprotein or serum urate levels be interpreted properly without knowing if the patient is accumulating excess cholesterol or urate deposits. As we order and interpret test results, we must consider the biology of the substance being measured as well as the test characteristics; too often, we react to abnormal laboratory results with an incomplete understanding of these aspects.
Moghadam-Kia et al do not dwell on the organ involvement causing CK elevations, but specificity is another very important aspect when clinically interpreting the results of a CK test. Many patients with muscle damage or inflammation have elevations in serum aspartate aminotransferase and alanine aminotransferase levels (the ratio of elevation depends on the time course of the muscle damage and on the relative clearance rate of the two enzymes). Without knowing that the CK is elevated, one might assume that an aminotransferase elevation reflects hepatitis. I have seen several patients with elevated aminotransferases and complaints of weakness and fatigue who were subjected to liver biopsy before it was recognized that the source of the enzyme elevation (“liver function test changes”) was muscle (or hemolysis). Frequently unrecognized is that aldolase, which has a cell distribution similar to that of lactate dehydrogenase, does not have the relative specificity of localization to muscle that CK has. CK is quite useful in distinguishing myocyte from hepatocyte damage.
This paper presents a wonderful reminder of the value of updating and reviewing what we know about tests that we order, even if we feel comfortable when ordering them. Before initiating a cascade of additional tests and consultations to explore the cause of an abnormal test result, a little time spent reviewing its basic characteristics and biology may pay dividends.
As 2015 comes to a close, we at the Journal share with you our sincere wishes for personal satisfaction and a globally more peaceful 2016.
We are all exposed to initiatives from multiple stakeholders telling us to order fewer tests. Many of these efforts to control costs and improve efficiency and quality of care are directed at populations of patients and are broad concepts: provide screening only to those most likely to benefit (eg, don’t screen for prostate cancer in men with a lifespan < 10 years); avoid procedures that provided limited benefit in controlled trials (eg, limit routine arthroscopic treatment of knee osteoarthritis); and avoid reflexive practices unlikely to improve patient outcomes (eg, eliminate routine preoperative testing before elective procedures in otherwise healthy patients).
Whether system-based changes will be implemented and have an impact remains to be determined. But I sense that with all the attention being focused on population management and healthcare practices, including an emphasis on documenting and coding our encounters with patients, whether substantive or simply digital housekeeping, we are increasingly distracted from the patient in front of us and are spending less time reviewing the principles underlying the diagnoses we make and the tests we order—just as we are taking less time to perform relevant physical examinations.1 The latter may mostly relate to time pressures. The former, I believe, is a product of both time pressures and a false sense of confidence in our knowledge of seemingly commonplace laboratory tests.
As I lecture, work with trainees, and reflect on my own patients, I realize that we are slowly but progressively minimizing the importance of a working knowledge of the basic foundations of clinical practice—perhaps because facts can always be looked up. I am not referring to knowledge of arcane biochemical pathways, eponymous references, or the latest recommended treatment of inclusion body myositis. I am thinking instead of the value of regularly refreshing our knowledge of laboratory tests and diagnoses we frequently encounter.
Having access to multiple clinical databases literally in our pockets is likely bolstering a false sense of confidence in our knowledge. The National Library of Medicine may be only a tap on a smart phone away, but accessing it regularly is a different thing. Attending conferences and reading educational journals help to keep our broad-based knowledge of internal medicine refreshed, but time pressures may significantly limit our ability to regularly pursue these activities.
In this issue of the Journal, Drs. Moghadam-Kia et al discuss their approach to asymptomatic elevations in creatine kinase (CK). Although no longer included in the most commonly used lab panels, CK measurement is often ordered in patients taking statins, even if they have no relevant muscle-related symptoms. Thus, evaluating a patient with an asymptomatic elevated CK level is not rare. The authors delve into the clinically relevant test characteristics, and their important caveats about interpreting elevated CK levels are germane not only to the asymptomatic patient, but also to the patient being evaluated for myalgia or weakness. This latter situation is one I frequently face in both the hospital and the outpatient clinic. I am often asked to consult on patients who have incompletely defined symptoms and elevated CK.
As discussed in the article, the laboratory definition of “normal” must first be considered. Laboratory test results must always be interpreted in the clinical context. An isolated elevation in parathyroid hormone cannot be interpreted without knowing the patient’s vitamin D level. Nor can “normal” low-density lipoprotein or serum urate levels be interpreted properly without knowing if the patient is accumulating excess cholesterol or urate deposits. As we order and interpret test results, we must consider the biology of the substance being measured as well as the test characteristics; too often, we react to abnormal laboratory results with an incomplete understanding of these aspects.
Moghadam-Kia et al do not dwell on the organ involvement causing CK elevations, but specificity is another very important aspect when clinically interpreting the results of a CK test. Many patients with muscle damage or inflammation have elevations in serum aspartate aminotransferase and alanine aminotransferase levels (the ratio of elevation depends on the time course of the muscle damage and on the relative clearance rate of the two enzymes). Without knowing that the CK is elevated, one might assume that an aminotransferase elevation reflects hepatitis. I have seen several patients with elevated aminotransferases and complaints of weakness and fatigue who were subjected to liver biopsy before it was recognized that the source of the enzyme elevation (“liver function test changes”) was muscle (or hemolysis). Frequently unrecognized is that aldolase, which has a cell distribution similar to that of lactate dehydrogenase, does not have the relative specificity of localization to muscle that CK has. CK is quite useful in distinguishing myocyte from hepatocyte damage.
This paper presents a wonderful reminder of the value of updating and reviewing what we know about tests that we order, even if we feel comfortable when ordering them. Before initiating a cascade of additional tests and consultations to explore the cause of an abnormal test result, a little time spent reviewing its basic characteristics and biology may pay dividends.
As 2015 comes to a close, we at the Journal share with you our sincere wishes for personal satisfaction and a globally more peaceful 2016.
- Verghese A, Charlton B, Kassirer JP, Ramsey M, Ioannidis JP. Inadequacies of physical examination as a cause of medical errors and adverse events: a collection of vignettes. Am J Med 2015; 128:1322–1324.
- Verghese A, Charlton B, Kassirer JP, Ramsey M, Ioannidis JP. Inadequacies of physical examination as a cause of medical errors and adverse events: a collection of vignettes. Am J Med 2015; 128:1322–1324.
Should you adopt the practice of vaginal cleansing with povidone-iodine prior to cesarean delivery?
There are approximately 4,000,000 births annually in the United States, and about 32% of them occur by cesarean delivery. Compared with vaginal birth, cesarean delivery is associated with an increased risk of endometritis (defined as fever plus uterine or abdominal tenderness). Although surgical complications cannot be eliminated entirely, surgeons are deeply dedicated to the continuous improvement of surgical practice in order to reduce the risk of complications.
With cesarean delivery, many surgical practices have been adopted universally to reduce postoperative complications, including administration of intravenous (IV) antibiotics before skin incision to minimize postoperative infection and the use of postoperative mechanical or pharmacologic interventions to help prevent venous thromboembolism and pulmonary embolism. Preoperative vaginal cleansing with povidone-iodine may reduce the risk of postoperative endometritis, but the practice is not currently common in the United States.
Should you adopt a policy of preoperative vaginal cleansing prior to cesarean delivery? The data suggest perhaps you should.
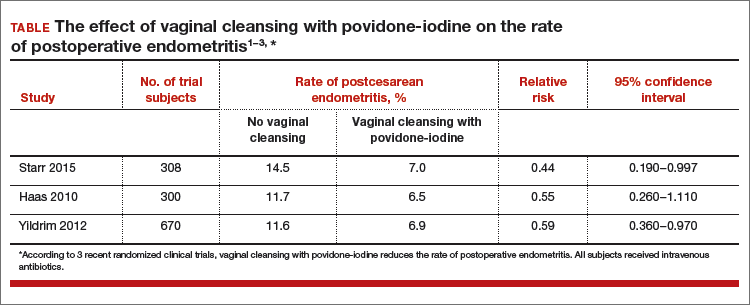
Data-driven support for povidone-iodine precesareanThree large randomized trials published within the past 10 years concluded that preoperative vaginal cleansing with povidone-iodine reduced the risk of postcesarean endometritis in women who also received prophylactic IV antibiotics (TABLE).1−3 Vaginal cleansing did not reduce the rate of postpartum fever or wound infection in these studies.
Clinical factors that increased the risk of postpartum endometritis independent of vaginal cleansing included:
- extended duration of cesarean surgery
- being in labor prior to cesarean delivery
- ruptured membranes
- advanced cervical examination
- maternal anemia
- use of intrapartum internal monitors
- prior history of genitourinary infection.
Authors of two recent, large nonrandomized studies also have reported that vaginal cleansing reduced the risk of postcesarean endometritis.4,5 By contrast, investigators from one large trial from 2001 did not observe a decrease in endometritis with vaginal cleansing.6
To test the impact of metronidazole vaginal gel on post‑cesarean endometritis, 224 women undergoing cesarean delivery for various indications were randomly assigned to placebo vaginal gel or metronidazole vaginal gel 5 g prior to surgery initiation.1 Most women also received intravenous antibiotics. The rates of endometritis were 17% and 7% in the placebo and metronidazole groups, respectively (relative risk, 0.42; 95% confidence interval, 0.19−0.92).
Vaginal antibiotic administration shows promise as an alternative to povidone-iodine cleansing in the prevention of postcesarean endometritis. Additional randomized clinical trials are necessary to fully evaluate the benefits and risks of this practice.
Reference
1. Pitt C, Sanchez-Ramos L, Kaunitz AM. Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol. 2001;98(5 pt 1):745−750.
Cochrane review of precesarean vaginal cleansingAuthors of a Cochrane review, in which they synthesized 7 studies involving 2,635 women, reported that vaginal cleansing with povidone-iodine immediately before cesarean delivery was associated with a reduced risk of postcesarean endometritis: 8.3% vs 4.3% in the control and vaginal cleansing groups, respectively, (risk ratio [RR], 0.45; 95% confidence interval [CI], 0.25−0.81).7
The positive effect of vaginal cleansing was particularly noteworthy in the subgroup of women with ruptured membranes (3 trials involving 272 women). The rates of endometritis in the control versus vaginal cleansing groups were 17.9% and 4.3%, respectively (RR, 0.24; 95% CI, 0.10−0.55).
Women who went into labor prior to cesarean delivery (523 women from 3 trials) also benefitted from vaginal cleansing, with endometritis rates of 13.0% and 7.4% in the control and vaginal cleansing groups, respectively (RR, 0.56; 95% CI, 0.34−0.95).
In this review, again, vaginal cleansing did not significantly reduce the rate of postoperative fever or wound infection.
The American College of Obstetricians and Gynecologists has noted that chlorohexidine gluconate solutions with high concentrations of alcohol are contraindicated for vaginal cleansing.1 However, although not approved for vaginal cleansing, solutions of chlorohexidine gluconate with low alcohol content (4% alcohol concentration) are safe and may be effective for off-label use as vaginal cleansings.
Reference
1. American College of Obstetricians and Gynecologists Women’s Health Care Physicians; Committee on Gynecologic Practice. Committee Opinion No. 571: solutions for surgical cleansing of the vagina. Obstet Gynecol. 2013;122(3):718−720.
Is vaginal cleansing prior to cesarean delivery best practice?In the United States, precesarean vaginal cleansing is not a common practice. To close the gap between current practice and what is potentially a best practice, two approaches to using vaginal cleansing could be instituted in delivery units.
Approach #1: A liberal clinical protocol. In this scenario, all women (who are not allergic to iodine or povidone-iodine) undergoing cesarean delivery should undergo vaginal cleansing. The World Health Organization conditionally recommends vaginal cleansing for all women undergoing a cesarean delivery.8
Approach #2: A focused clinical protocol. For this protocol, only women (again, who are not allergic to iodine or povidone-iodine) who have ruptured membranes or are in labor upon advanced cervical examination should receive vaginal cleansing.
The advantage of a liberal protocol is that vaginal preparation becomes embedded within the standard practice of cesarean delivery and, hence, is seldom overlooked. The upside of the focused protocol is that only those women most likely to benefit receive the intervention.
Tell me what you thinkWill you consider using vaginal cleansing in your practice? Please let me know your views on vaginal cleansing for cesarean delivery, as well as your clinical pearls on cesarean delivery surgery, at obgmanagement.com. In addition, weigh in on the Quick Poll posted to OBG Management’s homepage. Send your letter to the editor to [email protected].
- Starr RV, Zurawski J, Ismail M. Preoperative vaginal preparation with povidone-iodine and the risk of postcesarean endometritis. Obstet Gynecol. 2015;105(5 pt 1):1024–1029.
- Haas DM, Pazouki F, Smith RR, et al. Vaginal cleansing before cesarean delivery to reduce postoperative infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2010;202(3):310.e1–e6.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to cesarean delivery reduce the risk of endometritis?A randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(11):2316–2321.
- Asghania M, Mirblouk F, Shakiba M, Faraji R. Preoperative vaginal preparation with povidone-iodine on post-cesarean infectious morbidity. J Obstet Gynaecol. 2011;31(5):400–403.
- Memon S, Qazi RA, Bibi S, Parveen N. Effect of preoperative vaginal cleansing with an antiseptic solution to reduce post caesarean infectious morbidity. J Pak Med Assoc. 2011;61(12):1179–1183.
- Reid VC, Hartmann KE, McMahon M, Fry EP. Vaginal preparation with povidone-iodine and postcesarean infectious morbidity: a randomized controlled trial. Obstet Gynecol. 2001;97(1):147–152.
- Haas DM, Morgan S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2014;12:CD007892.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to
There are approximately 4,000,000 births annually in the United States, and about 32% of them occur by cesarean delivery. Compared with vaginal birth, cesarean delivery is associated with an increased risk of endometritis (defined as fever plus uterine or abdominal tenderness). Although surgical complications cannot be eliminated entirely, surgeons are deeply dedicated to the continuous improvement of surgical practice in order to reduce the risk of complications.
With cesarean delivery, many surgical practices have been adopted universally to reduce postoperative complications, including administration of intravenous (IV) antibiotics before skin incision to minimize postoperative infection and the use of postoperative mechanical or pharmacologic interventions to help prevent venous thromboembolism and pulmonary embolism. Preoperative vaginal cleansing with povidone-iodine may reduce the risk of postoperative endometritis, but the practice is not currently common in the United States.
Should you adopt a policy of preoperative vaginal cleansing prior to cesarean delivery? The data suggest perhaps you should.
Data-driven support for povidone-iodine precesareanThree large randomized trials published within the past 10 years concluded that preoperative vaginal cleansing with povidone-iodine reduced the risk of postcesarean endometritis in women who also received prophylactic IV antibiotics (TABLE).1−3 Vaginal cleansing did not reduce the rate of postpartum fever or wound infection in these studies.

Clinical factors that increased the risk of postpartum endometritis independent of vaginal cleansing included:
- extended duration of cesarean surgery
- being in labor prior to cesarean delivery
- ruptured membranes
- advanced cervical examination
- maternal anemia
- use of intrapartum internal monitors
- prior history of genitourinary infection.
Authors of two recent, large nonrandomized studies also have reported that vaginal cleansing reduced the risk of postcesarean endometritis.4,5 By contrast, investigators from one large trial from 2001 did not observe a decrease in endometritis with vaginal cleansing.6
To test the impact of metronidazole vaginal gel on post‑cesarean endometritis, 224 women undergoing cesarean delivery for various indications were randomly assigned to placebo vaginal gel or metronidazole vaginal gel 5 g prior to surgery initiation.1 Most women also received intravenous antibiotics. The rates of endometritis were 17% and 7% in the placebo and metronidazole groups, respectively (relative risk, 0.42; 95% confidence interval, 0.19−0.92).
Vaginal antibiotic administration shows promise as an alternative to povidone-iodine cleansing in the prevention of postcesarean endometritis. Additional randomized clinical trials are necessary to fully evaluate the benefits and risks of this practice.
Reference
1. Pitt C, Sanchez-Ramos L, Kaunitz AM. Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol. 2001;98(5 pt 1):745−750.
Cochrane review of precesarean vaginal cleansingAuthors of a Cochrane review, in which they synthesized 7 studies involving 2,635 women, reported that vaginal cleansing with povidone-iodine immediately before cesarean delivery was associated with a reduced risk of postcesarean endometritis: 8.3% vs 4.3% in the control and vaginal cleansing groups, respectively, (risk ratio [RR], 0.45; 95% confidence interval [CI], 0.25−0.81).7
The positive effect of vaginal cleansing was particularly noteworthy in the subgroup of women with ruptured membranes (3 trials involving 272 women). The rates of endometritis in the control versus vaginal cleansing groups were 17.9% and 4.3%, respectively (RR, 0.24; 95% CI, 0.10−0.55).
Women who went into labor prior to cesarean delivery (523 women from 3 trials) also benefitted from vaginal cleansing, with endometritis rates of 13.0% and 7.4% in the control and vaginal cleansing groups, respectively (RR, 0.56; 95% CI, 0.34−0.95).
In this review, again, vaginal cleansing did not significantly reduce the rate of postoperative fever or wound infection.
The American College of Obstetricians and Gynecologists has noted that chlorohexidine gluconate solutions with high concentrations of alcohol are contraindicated for vaginal cleansing.1 However, although not approved for vaginal cleansing, solutions of chlorohexidine gluconate with low alcohol content (4% alcohol concentration) are safe and may be effective for off-label use as vaginal cleansings.
Reference
1. American College of Obstetricians and Gynecologists Women’s Health Care Physicians; Committee on Gynecologic Practice. Committee Opinion No. 571: solutions for surgical cleansing of the vagina. Obstet Gynecol. 2013;122(3):718−720.
Is vaginal cleansing prior to cesarean delivery best practice?In the United States, precesarean vaginal cleansing is not a common practice. To close the gap between current practice and what is potentially a best practice, two approaches to using vaginal cleansing could be instituted in delivery units.
Approach #1: A liberal clinical protocol. In this scenario, all women (who are not allergic to iodine or povidone-iodine) undergoing cesarean delivery should undergo vaginal cleansing. The World Health Organization conditionally recommends vaginal cleansing for all women undergoing a cesarean delivery.8
Approach #2: A focused clinical protocol. For this protocol, only women (again, who are not allergic to iodine or povidone-iodine) who have ruptured membranes or are in labor upon advanced cervical examination should receive vaginal cleansing.
The advantage of a liberal protocol is that vaginal preparation becomes embedded within the standard practice of cesarean delivery and, hence, is seldom overlooked. The upside of the focused protocol is that only those women most likely to benefit receive the intervention.
Tell me what you thinkWill you consider using vaginal cleansing in your practice? Please let me know your views on vaginal cleansing for cesarean delivery, as well as your clinical pearls on cesarean delivery surgery, at obgmanagement.com. In addition, weigh in on the Quick Poll posted to OBG Management’s homepage. Send your letter to the editor to [email protected].
There are approximately 4,000,000 births annually in the United States, and about 32% of them occur by cesarean delivery. Compared with vaginal birth, cesarean delivery is associated with an increased risk of endometritis (defined as fever plus uterine or abdominal tenderness). Although surgical complications cannot be eliminated entirely, surgeons are deeply dedicated to the continuous improvement of surgical practice in order to reduce the risk of complications.
With cesarean delivery, many surgical practices have been adopted universally to reduce postoperative complications, including administration of intravenous (IV) antibiotics before skin incision to minimize postoperative infection and the use of postoperative mechanical or pharmacologic interventions to help prevent venous thromboembolism and pulmonary embolism. Preoperative vaginal cleansing with povidone-iodine may reduce the risk of postoperative endometritis, but the practice is not currently common in the United States.
Should you adopt a policy of preoperative vaginal cleansing prior to cesarean delivery? The data suggest perhaps you should.
Data-driven support for povidone-iodine precesareanThree large randomized trials published within the past 10 years concluded that preoperative vaginal cleansing with povidone-iodine reduced the risk of postcesarean endometritis in women who also received prophylactic IV antibiotics (TABLE).1−3 Vaginal cleansing did not reduce the rate of postpartum fever or wound infection in these studies.

Clinical factors that increased the risk of postpartum endometritis independent of vaginal cleansing included:
- extended duration of cesarean surgery
- being in labor prior to cesarean delivery
- ruptured membranes
- advanced cervical examination
- maternal anemia
- use of intrapartum internal monitors
- prior history of genitourinary infection.
Authors of two recent, large nonrandomized studies also have reported that vaginal cleansing reduced the risk of postcesarean endometritis.4,5 By contrast, investigators from one large trial from 2001 did not observe a decrease in endometritis with vaginal cleansing.6
To test the impact of metronidazole vaginal gel on post‑cesarean endometritis, 224 women undergoing cesarean delivery for various indications were randomly assigned to placebo vaginal gel or metronidazole vaginal gel 5 g prior to surgery initiation.1 Most women also received intravenous antibiotics. The rates of endometritis were 17% and 7% in the placebo and metronidazole groups, respectively (relative risk, 0.42; 95% confidence interval, 0.19−0.92).
Vaginal antibiotic administration shows promise as an alternative to povidone-iodine cleansing in the prevention of postcesarean endometritis. Additional randomized clinical trials are necessary to fully evaluate the benefits and risks of this practice.
Reference
1. Pitt C, Sanchez-Ramos L, Kaunitz AM. Adjunctive intravaginal metronidazole for the prevention of postcesarean endometritis: a randomized controlled trial. Obstet Gynecol. 2001;98(5 pt 1):745−750.
Cochrane review of precesarean vaginal cleansingAuthors of a Cochrane review, in which they synthesized 7 studies involving 2,635 women, reported that vaginal cleansing with povidone-iodine immediately before cesarean delivery was associated with a reduced risk of postcesarean endometritis: 8.3% vs 4.3% in the control and vaginal cleansing groups, respectively, (risk ratio [RR], 0.45; 95% confidence interval [CI], 0.25−0.81).7
The positive effect of vaginal cleansing was particularly noteworthy in the subgroup of women with ruptured membranes (3 trials involving 272 women). The rates of endometritis in the control versus vaginal cleansing groups were 17.9% and 4.3%, respectively (RR, 0.24; 95% CI, 0.10−0.55).
Women who went into labor prior to cesarean delivery (523 women from 3 trials) also benefitted from vaginal cleansing, with endometritis rates of 13.0% and 7.4% in the control and vaginal cleansing groups, respectively (RR, 0.56; 95% CI, 0.34−0.95).
In this review, again, vaginal cleansing did not significantly reduce the rate of postoperative fever or wound infection.
The American College of Obstetricians and Gynecologists has noted that chlorohexidine gluconate solutions with high concentrations of alcohol are contraindicated for vaginal cleansing.1 However, although not approved for vaginal cleansing, solutions of chlorohexidine gluconate with low alcohol content (4% alcohol concentration) are safe and may be effective for off-label use as vaginal cleansings.
Reference
1. American College of Obstetricians and Gynecologists Women’s Health Care Physicians; Committee on Gynecologic Practice. Committee Opinion No. 571: solutions for surgical cleansing of the vagina. Obstet Gynecol. 2013;122(3):718−720.
Is vaginal cleansing prior to cesarean delivery best practice?In the United States, precesarean vaginal cleansing is not a common practice. To close the gap between current practice and what is potentially a best practice, two approaches to using vaginal cleansing could be instituted in delivery units.
Approach #1: A liberal clinical protocol. In this scenario, all women (who are not allergic to iodine or povidone-iodine) undergoing cesarean delivery should undergo vaginal cleansing. The World Health Organization conditionally recommends vaginal cleansing for all women undergoing a cesarean delivery.8
Approach #2: A focused clinical protocol. For this protocol, only women (again, who are not allergic to iodine or povidone-iodine) who have ruptured membranes or are in labor upon advanced cervical examination should receive vaginal cleansing.
The advantage of a liberal protocol is that vaginal preparation becomes embedded within the standard practice of cesarean delivery and, hence, is seldom overlooked. The upside of the focused protocol is that only those women most likely to benefit receive the intervention.
Tell me what you thinkWill you consider using vaginal cleansing in your practice? Please let me know your views on vaginal cleansing for cesarean delivery, as well as your clinical pearls on cesarean delivery surgery, at obgmanagement.com. In addition, weigh in on the Quick Poll posted to OBG Management’s homepage. Send your letter to the editor to [email protected].
- Starr RV, Zurawski J, Ismail M. Preoperative vaginal preparation with povidone-iodine and the risk of postcesarean endometritis. Obstet Gynecol. 2015;105(5 pt 1):1024–1029.
- Haas DM, Pazouki F, Smith RR, et al. Vaginal cleansing before cesarean delivery to reduce postoperative infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2010;202(3):310.e1–e6.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to cesarean delivery reduce the risk of endometritis?A randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(11):2316–2321.
- Asghania M, Mirblouk F, Shakiba M, Faraji R. Preoperative vaginal preparation with povidone-iodine on post-cesarean infectious morbidity. J Obstet Gynaecol. 2011;31(5):400–403.
- Memon S, Qazi RA, Bibi S, Parveen N. Effect of preoperative vaginal cleansing with an antiseptic solution to reduce post caesarean infectious morbidity. J Pak Med Assoc. 2011;61(12):1179–1183.
- Reid VC, Hartmann KE, McMahon M, Fry EP. Vaginal preparation with povidone-iodine and postcesarean infectious morbidity: a randomized controlled trial. Obstet Gynecol. 2001;97(1):147–152.
- Haas DM, Morgan S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2014;12:CD007892.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to
- Starr RV, Zurawski J, Ismail M. Preoperative vaginal preparation with povidone-iodine and the risk of postcesarean endometritis. Obstet Gynecol. 2015;105(5 pt 1):1024–1029.
- Haas DM, Pazouki F, Smith RR, et al. Vaginal cleansing before cesarean delivery to reduce postoperative infectious morbidity: a randomized, controlled trial. Am J Obstet Gynecol. 2010;202(3):310.e1–e6.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to cesarean delivery reduce the risk of endometritis?A randomized controlled trial. J Matern Fetal Neonatal Med. 2012;25(11):2316–2321.
- Asghania M, Mirblouk F, Shakiba M, Faraji R. Preoperative vaginal preparation with povidone-iodine on post-cesarean infectious morbidity. J Obstet Gynaecol. 2011;31(5):400–403.
- Memon S, Qazi RA, Bibi S, Parveen N. Effect of preoperative vaginal cleansing with an antiseptic solution to reduce post caesarean infectious morbidity. J Pak Med Assoc. 2011;61(12):1179–1183.
- Reid VC, Hartmann KE, McMahon M, Fry EP. Vaginal preparation with povidone-iodine and postcesarean infectious morbidity: a randomized controlled trial. Obstet Gynecol. 2001;97(1):147–152.
- Haas DM, Morgan S, Contreras K. Vaginal preparation with antiseptic solution before cesarean section for preventing postoperative infections. Cochrane Database Syst Rev. 2014;12:CD007892.
- Yildrim G, Gungorduk K, Asicioglu O, et al. Does vaginal preparation with povidone-iodine prior to
16 New Year’s resolutions for psychiatrists in 2016
Such decisions can be made at any time, but the dawn of a year is a powerful signal of a new beginning—another lease on life, a potential turning point. Imbedded in those resolutions is a subliminal sense of urgency to correct one’s long-neglected shortcomings as the calendar ruthlessly points to inevitable aging and the relentless march of time.
A psychiatric perspective
For psychiatrists, New Year’s resolutions transcend the (often ephemeral) impulse to go on a diet or buy a membership at the local gym. We have a unique perspective on the challenges that our patients face every day as they cope with the complex demands of life despite their anxiety, depression, or psychosis.
We are aware of the many unmet needs in managing complex neuropsychiatric brain disorders and the major challenges of erasing the burdensome stigma that engulfs our patients and the practice of psychiatry itself—despite its noble mission of repairing fractured brains, mending tortured souls, and restoring peace of mind and wellness. We are proud of our clinical and scientific accomplishments but are painfully cognizant of our limitations and the huge chasm between what we know and what we will eventually know once the brain reveals its glorious mysteries through neuroscientific research.
What can you resolve?
Here is my proposed list of pragmatic resolutions that most psychiatrists would regard as part of a perpetual to-do list—a must-do bucket of cherished goals and brave new horizons to bring complete mental health for our patients and immeasurable gratification for us, who dream of cures for brain disorders that trigger various ailments of the mind.
- Practice like a physician to emphasize the medical foundation of psychiatry: Always check on a patient’s physical health, and monitor his (her) cardiometabolic status. Wear the symbolic white coat that often enhances the physician−patient relationship.
- Dedicate a significant percentage of your practice to the sickest patients. There are enough non-physician mental health professionals to handle the walking wounded and worried well.
- Advocate relentlessly throughout your sphere of influence, and publicly, for true parity between psychiatric and non-mental medical disorders—not only for insurance coverage but for overall societal acceptance and compassion as well.
- Lobby vigorously for hospitalization instead of imprisonment of the seriously mentally ill because psychosis is a brain disease, not a criminal offense.
- Adopt evidence-based psychiatric practice whenever possible to achieve the best outcomes. Judiciously implement off-label practices, however, if no evidence-based treatments exist for a suffering patient.
- Avoid senseless and irrational polypharmacy but do not hesitate to use rational, beneficial combination therapy.
- Provide 1 hour a week of pro bono psychiatric work for the indigent and underserved. The rewards of giving what amounts to 1 week a year are immeasurably more gratifying than a few more dollars in your bank account.
- Resist calling an ill person a ‘client’ or ‘consumer’—at least until oncologists and cardiologists start doing so. Refuse to give up your medical identify in the many de-medicalized mental health clinics.
- Never let a patient leave your office without some psychotherapy, even as part of a 15-minute med-check.
- Stay current and on the cutting edge of evolving psychiatric practice by logging into PubMed every day (even briefly) to read a few abstracts of the latest studies related to patients you saw that day.
- Think like a neurologist by identifying the neural circuits of psychiatric symptoms. Act like a cardiologist by doing everything medically possible to prevent recurrence of psychotic, manic, or depressive episodes because they damage brain tissue just as a myocardial infarction damages the heart.
- Support research with words, money, and passion. Psychiatric neuroscientific breakthroughs generate superior treatments, erase stigma, and advance the quality of life for patients. Donate annually to the researchers of your choice, at the medical school where you were trained, or at a nonprofit research institute.
- Make time to write for publication, annually, at least 1 case report or a letter to the editor about observations from your practice. You can contribute immensely to the discovery process by sharing novel clinical insights.
- Never give up on any patient or set expectations too low, regardless of the diagnosis or severity of illness. Giving up destroys hope and ushers in despondency. Get a second opinion if you run out of options for a patient.
- Always set remission followed by recovery as the therapeutic goal for every patient. Let the patient know this and ask him (her) commit to that goal with you.
- Be genuinely proud to be a psychiatrist. You assess and rectify disorders of the mind, the most complex and magical product of the human brain that determines who we are and how we think, emote, communicate, verbalize, empathize, love, hate, remember, plan, problem-solve, and, of course, make resolutions.
Back to diet and exercise—for our patients and for us!
It’s OK to include, among your New Year’s resolutions, a pledge to strongly encourage patients to diet and exercise. Given the tendency of many of them to gain weight and die prematurely as a consequence of obesity-related cardiometabolic risk factors, you should urge them to eat healthy and exercise every time you see them, not only on New Year’s Day.
Such decisions can be made at any time, but the dawn of a year is a powerful signal of a new beginning—another lease on life, a potential turning point. Imbedded in those resolutions is a subliminal sense of urgency to correct one’s long-neglected shortcomings as the calendar ruthlessly points to inevitable aging and the relentless march of time.
A psychiatric perspective
For psychiatrists, New Year’s resolutions transcend the (often ephemeral) impulse to go on a diet or buy a membership at the local gym. We have a unique perspective on the challenges that our patients face every day as they cope with the complex demands of life despite their anxiety, depression, or psychosis.
We are aware of the many unmet needs in managing complex neuropsychiatric brain disorders and the major challenges of erasing the burdensome stigma that engulfs our patients and the practice of psychiatry itself—despite its noble mission of repairing fractured brains, mending tortured souls, and restoring peace of mind and wellness. We are proud of our clinical and scientific accomplishments but are painfully cognizant of our limitations and the huge chasm between what we know and what we will eventually know once the brain reveals its glorious mysteries through neuroscientific research.
What can you resolve?
Here is my proposed list of pragmatic resolutions that most psychiatrists would regard as part of a perpetual to-do list—a must-do bucket of cherished goals and brave new horizons to bring complete mental health for our patients and immeasurable gratification for us, who dream of cures for brain disorders that trigger various ailments of the mind.
- Practice like a physician to emphasize the medical foundation of psychiatry: Always check on a patient’s physical health, and monitor his (her) cardiometabolic status. Wear the symbolic white coat that often enhances the physician−patient relationship.
- Dedicate a significant percentage of your practice to the sickest patients. There are enough non-physician mental health professionals to handle the walking wounded and worried well.
- Advocate relentlessly throughout your sphere of influence, and publicly, for true parity between psychiatric and non-mental medical disorders—not only for insurance coverage but for overall societal acceptance and compassion as well.
- Lobby vigorously for hospitalization instead of imprisonment of the seriously mentally ill because psychosis is a brain disease, not a criminal offense.
- Adopt evidence-based psychiatric practice whenever possible to achieve the best outcomes. Judiciously implement off-label practices, however, if no evidence-based treatments exist for a suffering patient.
- Avoid senseless and irrational polypharmacy but do not hesitate to use rational, beneficial combination therapy.
- Provide 1 hour a week of pro bono psychiatric work for the indigent and underserved. The rewards of giving what amounts to 1 week a year are immeasurably more gratifying than a few more dollars in your bank account.
- Resist calling an ill person a ‘client’ or ‘consumer’—at least until oncologists and cardiologists start doing so. Refuse to give up your medical identify in the many de-medicalized mental health clinics.
- Never let a patient leave your office without some psychotherapy, even as part of a 15-minute med-check.
- Stay current and on the cutting edge of evolving psychiatric practice by logging into PubMed every day (even briefly) to read a few abstracts of the latest studies related to patients you saw that day.
- Think like a neurologist by identifying the neural circuits of psychiatric symptoms. Act like a cardiologist by doing everything medically possible to prevent recurrence of psychotic, manic, or depressive episodes because they damage brain tissue just as a myocardial infarction damages the heart.
- Support research with words, money, and passion. Psychiatric neuroscientific breakthroughs generate superior treatments, erase stigma, and advance the quality of life for patients. Donate annually to the researchers of your choice, at the medical school where you were trained, or at a nonprofit research institute.
- Make time to write for publication, annually, at least 1 case report or a letter to the editor about observations from your practice. You can contribute immensely to the discovery process by sharing novel clinical insights.
- Never give up on any patient or set expectations too low, regardless of the diagnosis or severity of illness. Giving up destroys hope and ushers in despondency. Get a second opinion if you run out of options for a patient.
- Always set remission followed by recovery as the therapeutic goal for every patient. Let the patient know this and ask him (her) commit to that goal with you.
- Be genuinely proud to be a psychiatrist. You assess and rectify disorders of the mind, the most complex and magical product of the human brain that determines who we are and how we think, emote, communicate, verbalize, empathize, love, hate, remember, plan, problem-solve, and, of course, make resolutions.
Back to diet and exercise—for our patients and for us!
It’s OK to include, among your New Year’s resolutions, a pledge to strongly encourage patients to diet and exercise. Given the tendency of many of them to gain weight and die prematurely as a consequence of obesity-related cardiometabolic risk factors, you should urge them to eat healthy and exercise every time you see them, not only on New Year’s Day.
Such decisions can be made at any time, but the dawn of a year is a powerful signal of a new beginning—another lease on life, a potential turning point. Imbedded in those resolutions is a subliminal sense of urgency to correct one’s long-neglected shortcomings as the calendar ruthlessly points to inevitable aging and the relentless march of time.
A psychiatric perspective
For psychiatrists, New Year’s resolutions transcend the (often ephemeral) impulse to go on a diet or buy a membership at the local gym. We have a unique perspective on the challenges that our patients face every day as they cope with the complex demands of life despite their anxiety, depression, or psychosis.
We are aware of the many unmet needs in managing complex neuropsychiatric brain disorders and the major challenges of erasing the burdensome stigma that engulfs our patients and the practice of psychiatry itself—despite its noble mission of repairing fractured brains, mending tortured souls, and restoring peace of mind and wellness. We are proud of our clinical and scientific accomplishments but are painfully cognizant of our limitations and the huge chasm between what we know and what we will eventually know once the brain reveals its glorious mysteries through neuroscientific research.
What can you resolve?
Here is my proposed list of pragmatic resolutions that most psychiatrists would regard as part of a perpetual to-do list—a must-do bucket of cherished goals and brave new horizons to bring complete mental health for our patients and immeasurable gratification for us, who dream of cures for brain disorders that trigger various ailments of the mind.
- Practice like a physician to emphasize the medical foundation of psychiatry: Always check on a patient’s physical health, and monitor his (her) cardiometabolic status. Wear the symbolic white coat that often enhances the physician−patient relationship.
- Dedicate a significant percentage of your practice to the sickest patients. There are enough non-physician mental health professionals to handle the walking wounded and worried well.
- Advocate relentlessly throughout your sphere of influence, and publicly, for true parity between psychiatric and non-mental medical disorders—not only for insurance coverage but for overall societal acceptance and compassion as well.
- Lobby vigorously for hospitalization instead of imprisonment of the seriously mentally ill because psychosis is a brain disease, not a criminal offense.
- Adopt evidence-based psychiatric practice whenever possible to achieve the best outcomes. Judiciously implement off-label practices, however, if no evidence-based treatments exist for a suffering patient.
- Avoid senseless and irrational polypharmacy but do not hesitate to use rational, beneficial combination therapy.
- Provide 1 hour a week of pro bono psychiatric work for the indigent and underserved. The rewards of giving what amounts to 1 week a year are immeasurably more gratifying than a few more dollars in your bank account.
- Resist calling an ill person a ‘client’ or ‘consumer’—at least until oncologists and cardiologists start doing so. Refuse to give up your medical identify in the many de-medicalized mental health clinics.
- Never let a patient leave your office without some psychotherapy, even as part of a 15-minute med-check.
- Stay current and on the cutting edge of evolving psychiatric practice by logging into PubMed every day (even briefly) to read a few abstracts of the latest studies related to patients you saw that day.
- Think like a neurologist by identifying the neural circuits of psychiatric symptoms. Act like a cardiologist by doing everything medically possible to prevent recurrence of psychotic, manic, or depressive episodes because they damage brain tissue just as a myocardial infarction damages the heart.
- Support research with words, money, and passion. Psychiatric neuroscientific breakthroughs generate superior treatments, erase stigma, and advance the quality of life for patients. Donate annually to the researchers of your choice, at the medical school where you were trained, or at a nonprofit research institute.
- Make time to write for publication, annually, at least 1 case report or a letter to the editor about observations from your practice. You can contribute immensely to the discovery process by sharing novel clinical insights.
- Never give up on any patient or set expectations too low, regardless of the diagnosis or severity of illness. Giving up destroys hope and ushers in despondency. Get a second opinion if you run out of options for a patient.
- Always set remission followed by recovery as the therapeutic goal for every patient. Let the patient know this and ask him (her) commit to that goal with you.
- Be genuinely proud to be a psychiatrist. You assess and rectify disorders of the mind, the most complex and magical product of the human brain that determines who we are and how we think, emote, communicate, verbalize, empathize, love, hate, remember, plan, problem-solve, and, of course, make resolutions.
Back to diet and exercise—for our patients and for us!
It’s OK to include, among your New Year’s resolutions, a pledge to strongly encourage patients to diet and exercise. Given the tendency of many of them to gain weight and die prematurely as a consequence of obesity-related cardiometabolic risk factors, you should urge them to eat healthy and exercise every time you see them, not only on New Year’s Day.
Oncology 2015: new therapies and new transitions toward value-based cancer care
The past year has been an exciting one for new oncology and hematology drug approvals and the continued evolution of our oncology delivery system toward high quality and value. In all, at press time in mid-November, the US Food and Drug Administration (FDA) had approved or granted expanded indications for 24 drugs, compared with 19 in the 2 preceding years. Of those 24 approvals, 7 were accelerated and 6 were expanded approvals, and 3 alone were for the immunotherapeutic drug, nivolumab – 2 for non-small-cell lung cancer (NSCLC) and 1 for metastatic melanoma.
Click on the PDF icon at the top of this introduction to read the full article.
The past year has been an exciting one for new oncology and hematology drug approvals and the continued evolution of our oncology delivery system toward high quality and value. In all, at press time in mid-November, the US Food and Drug Administration (FDA) had approved or granted expanded indications for 24 drugs, compared with 19 in the 2 preceding years. Of those 24 approvals, 7 were accelerated and 6 were expanded approvals, and 3 alone were for the immunotherapeutic drug, nivolumab – 2 for non-small-cell lung cancer (NSCLC) and 1 for metastatic melanoma.
Click on the PDF icon at the top of this introduction to read the full article.
The past year has been an exciting one for new oncology and hematology drug approvals and the continued evolution of our oncology delivery system toward high quality and value. In all, at press time in mid-November, the US Food and Drug Administration (FDA) had approved or granted expanded indications for 24 drugs, compared with 19 in the 2 preceding years. Of those 24 approvals, 7 were accelerated and 6 were expanded approvals, and 3 alone were for the immunotherapeutic drug, nivolumab – 2 for non-small-cell lung cancer (NSCLC) and 1 for metastatic melanoma.
Click on the PDF icon at the top of this introduction to read the full article.
Should newborns at 22 or 23 weeks’ gestational age be aggressively resuscitated?
For many decades the limit of viability was believed to be approximately 24 weeks of gestation. In many medical centers, newborns delivered at less than 25 weeks are evaluated in the delivery room and the decision to resuscitate is based on the infant’s clinical response. In the past, aggressive and extended resuscitation of newborns at 22 and 23 weeks was not common because the prognosis was bleak and clinicians did not want to inflict unnecessary pain when the chances for survival were limited. Recent advances in obstetric and pediatric care, however, have resulted in the survival of some infants born at 22 weeks’ gestation, calling into question long-held beliefs about the limits of viability.
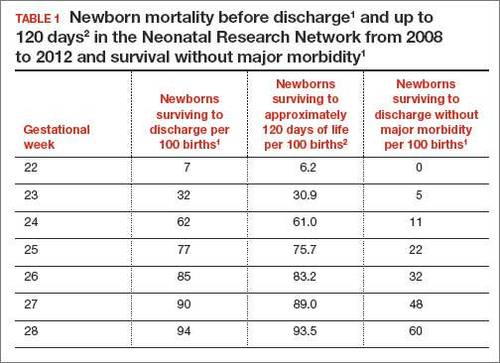
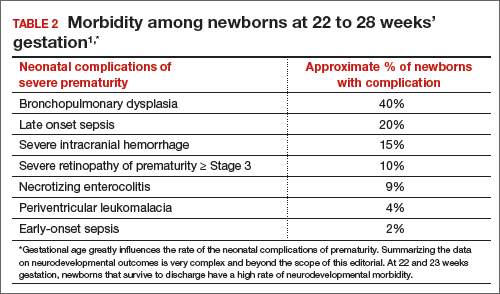
In 2 recent reports, investigators used data from the National Institute of Child Health and Human Development (NICHD) Neonatal Research Network to acquire detailed information about newborn survival and morbidity at 22 through 28 weeks’ gestation (TABLES 1 and 2).1,2 These data show that the survival of newborns at 23 through 27 weeks’ gestation is increasing, albeit slowly. Survival, without major morbidity, is gradually improving for newborns at 25 through 28 weeks.1,2 But what is the prognosis for a fetus born at 22 or 23 weeks?
There are several aspects of this issue to consider, including accurate dating of the gestational age and current viability outcomes data.
Determining the limit of viability: Accurate dating is essentialThe limit of viability is the milestone in gestation when there is a high probability of extrauterine survival. A major challenge in studies of the limit of viability for newborns is that accurate gestational dating is not always available. For example, in recent reports from the NICHD Neonatal Research Network the gestational age was determined by the best obstetric estimate, or the Ballard or Dubowitz examination, of the newborn.1,2
It is well known that ultrasound dating early in gestation is a better estimate of gestational age than last menstrual period, uterine sizing, or pediatric examination of the newborn. Hence, the available data are limited by the absence of precise gestational dating with early ultrasound. Data on the limit of viability with large numbers of births between 22 and 24 weeks with early ultrasound dating would help to refine our understanding of the limit of viability.
At 23 weeks, each day of in utero development is criticalThe importance of each additional day spent in utero during the 23rd week of gestation was demonstrated in a small cohort in 2001.4 Overall, during the 23rd week of gestation the survival of newborns to discharge was 33%.4 This finding is similar to the survival rate reported by the NICHD Neonatal Research Network in 2012.1 However, survival was vastly different early, compared with later, in the 23rd week4:
- from 23 weeks 0 days to 23 weeks 2 days: no newborn survived
- at 23 weeks 3 days and 23 weeks 4 days: 40% of newborns survived
- at 23 weeks 5 days and 23 weeks 6 days: 63% of newborns survived (a similar survival rate of 24-week gestations was reported by the NICHD Neonatal Research Network1).
The development of the fetus across the 23rd week of gestation appears to be critical to newborn survival. Hence, every day of in utero development during the 23rd week is critically important. A great challenge for obstetricians is how to approach the woman with threatened preterm birth at 22 weeks 0 days’ gestation. If the woman delivers within a few days, the likelihood of survival is minimal. However, if the pregnancy can be extended to 23 weeks and 5 days, survival rates increase significantly.
Aligning the actions of birth team, mother, and familyFactors that influence the limit of viability include:
- gestational age
- gender of the fetus (Females are more likely than males to survive.)
- treatment of the mother with glucocorticoids prior to birth
- newborn weight.
To increase the likelihood of newborn survival, obstetricians need to treat women at risk for preterm birth with antenatal glucocorticoids and antibiotics for rupture of membranes and to limit fetal stress during the birth process. Guidelines have evolved to encourage clinicians to treat women at preterm birth risk with glucocorticoids either at:
- 23 weeks’ gestation or
- 22 weeks’ gestation, if birth is anticipated to occur at 23 weeks or later.5
At birth, pediatricians are then faced with the very difficult decision of whether or not to aggressively resuscitate the severely preterm infant. Complex medical, social, and ethical issues ultimately guide pediatricians’ actions in this challenging situation. It is important for their actions to be in consensus with the obstetrician, the mother, and the mother’s family and for a consensus to be reached. Dissonant plans may increase adverse outcomes for the newborn. In one study when pediatricians and obstetricians were not aligned in their actions, the risk of death of an extremely preterm newborn significantly increased.6
Prior to birth, team meetings that include the obstetricians, pediatricians, mother, and family will help to set expectations about the course of care and, in turn, improve perceived outcomes.5 If feasible, obstetricians and pediatricians should develop joint institutional guidelines about the general approach to pregnant women when birth may occur at 22 or 23 weeks’ gestation.5
A neonatal outcomes predictor
The National Institute of Child Health and Human Development provides a Web-based tool for estimating newborn outcomes based on gestational age (22 to 25 weeks), birth weight, gender, singleton or multiple gestation, and exposure to antenatal glucocorticoid treatment. The outcomes tool provides estimates for survival and survival with severe morbidity. It uses data collected by the Neonatal Research Network to predict outcomes. To access the outcomes data assessment, visit https://www.nichd.nih.gov/about/org/der/branches/ppb/programs/epbo/Pages/epbo_case.aspx.
Is aggressive management of preterm birth and neonatal resuscitation a self-fulfilling prophecy?The beliefs and training of clinicians may influence the outcome of extremely preterm newborns. For example, if obstetricians and pediatricians focus on the fact that birth at 23 weeks is not likely to result in survival without severe morbidity, they may withhold key interventions such as antenatal glucocorticoids, antibiotics for rupture of the membranes, and aggressive newborn resuscitation.7 Consequently the likelihood of survival may be reduced.
If clinicians believe in maximal interventions for all newborns at 22 and 23 weeks’ gestation, their actions may result in a small increase in newborn survival—but at the cost of painful and unnecessary interventions in many newborns who are destined to die. Finding the right balance along the broad spectrum from expectant management to aggressive and extended resuscitation is challenging. Clearly there is no “right answer” with these extremely difficult decisions.
Future trends in the limit of viabilityIn 1963, Jacqueline Bouvier Kennedy, at 34 weeks’ gestation, went into preterm labor and delivered her son Patrick at a community hospital. Patrick developed respiratory distress syndrome and was transferred to the Boston Children’s Hospital. He died shortly thereafter.8 Would Patrick have survived if he had been delivered at an institution capable of providing high-risk obstetric and newborn services? Would such modern interventions as antenatal glucocorticoids, antibiotics for ruptured membranes, liberal use of cesarean delivery, and aggressive neonatal resuscitation have improved his chances for survival?
From our current perspective, it is surprising that a 34-week newborn died shortly after birth. With modern obstetric and pediatric care that scenario is unusual. It is possible that future advances in medical care will push the limit of viability to 22 weeks’ gestation. Future generations of clinicians may be surprised that the medicine we practice today is so limited.
However, given our current resources, it is unlikely that newborns at 22 weeks’ gestation will survive, or survive without severe morbidity. Consequently, routine aggressive resuscitation of newborns at 22 weeks should be approached with caution. At 23 weeks and later, many newborns will survive and a few will survive without severe morbidity. Given the complexity of the issues, the approach to resuscitation of infants at 22 and 23 weeks must account for the perspectives of the birth mother and her family, obstetricians, and pediatricians. Managing threatened preterm birth at 22 and 23 weeks is one of our greatest challenges as obstetricians, and we need to meet this challenge with grace and skill.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Stoll BJ, Hansen NI, Bell EF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Trends in care practices, morbidity and mortality of extremely preterm neonates, 1993-2012. JAMA. 2015;314(10):1039–1051.
- Patel RM, Kandefer S, Walsh MC, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Causes and timing of death in extremely premature infants from 2000 through 2011. N Engl J Med. 2015;372(4):331–340.
- Donovan EF, Tyson JE, Ehrenkranz RA, et al. Inaccuracy of Ballard scores before 28 weeks’ gestation. National Institute of Child Health and Human Development Neonatal Research Network. J Pediatr. 1999;135(2 pt 1):147–152.
- McElrath TF, Robinson JN, Ecker JL, Ringer SA, Norwitz ER. Neonatal outcome of infants born at 23 weeks’ gestation. Obstet Gynecol. 2001;97(1):49–52.
- Raju TN, Mercer BM, Burchfield DJ, Joseph GF Jr. Periviable birth: executive summary of a joint workshop by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for Maternal-Fetal Medicine, American Academy of Pediatrics, and American College of Obstetricians and Gynecologists. Obstet Gynecol. 2014;123(5):1083–1096.
- Guinsburg R, Branco de Almeida MF, dos Santos Rodrigues Sadeck L, et al; Brazilian Network on Neonatal Research. Proactive management of extreme prematurity: disagreement between obstetricians and neonatologists. J Perinatol. 2012;32(12):913-919.
- Tucker Emonds B, McKenzie F, Farrow V, Raglan G, Schulkin J. A national survey of obstetricians’ attitudes toward and practice of periviable interventions. J Perinatol. 2015;35(5):338–343.
- Altman LK. A Kennedy baby’s life and death. New York Times. http://www.nytimes.com/2013/07/30/health/a-kennedy-babys-life-and-death.html?_r=0. Published July 29, 2013. Accessed November 19, 2015.
For many decades the limit of viability was believed to be approximately 24 weeks of gestation. In many medical centers, newborns delivered at less than 25 weeks are evaluated in the delivery room and the decision to resuscitate is based on the infant’s clinical response. In the past, aggressive and extended resuscitation of newborns at 22 and 23 weeks was not common because the prognosis was bleak and clinicians did not want to inflict unnecessary pain when the chances for survival were limited. Recent advances in obstetric and pediatric care, however, have resulted in the survival of some infants born at 22 weeks’ gestation, calling into question long-held beliefs about the limits of viability.
In 2 recent reports, investigators used data from the National Institute of Child Health and Human Development (NICHD) Neonatal Research Network to acquire detailed information about newborn survival and morbidity at 22 through 28 weeks’ gestation (TABLES 1 and 2).1,2 These data show that the survival of newborns at 23 through 27 weeks’ gestation is increasing, albeit slowly. Survival, without major morbidity, is gradually improving for newborns at 25 through 28 weeks.1,2 But what is the prognosis for a fetus born at 22 or 23 weeks?


There are several aspects of this issue to consider, including accurate dating of the gestational age and current viability outcomes data.
Determining the limit of viability: Accurate dating is essentialThe limit of viability is the milestone in gestation when there is a high probability of extrauterine survival. A major challenge in studies of the limit of viability for newborns is that accurate gestational dating is not always available. For example, in recent reports from the NICHD Neonatal Research Network the gestational age was determined by the best obstetric estimate, or the Ballard or Dubowitz examination, of the newborn.1,2
It is well known that ultrasound dating early in gestation is a better estimate of gestational age than last menstrual period, uterine sizing, or pediatric examination of the newborn. Hence, the available data are limited by the absence of precise gestational dating with early ultrasound. Data on the limit of viability with large numbers of births between 22 and 24 weeks with early ultrasound dating would help to refine our understanding of the limit of viability.
At 23 weeks, each day of in utero development is criticalThe importance of each additional day spent in utero during the 23rd week of gestation was demonstrated in a small cohort in 2001.4 Overall, during the 23rd week of gestation the survival of newborns to discharge was 33%.4 This finding is similar to the survival rate reported by the NICHD Neonatal Research Network in 2012.1 However, survival was vastly different early, compared with later, in the 23rd week4:
- from 23 weeks 0 days to 23 weeks 2 days: no newborn survived
- at 23 weeks 3 days and 23 weeks 4 days: 40% of newborns survived
- at 23 weeks 5 days and 23 weeks 6 days: 63% of newborns survived (a similar survival rate of 24-week gestations was reported by the NICHD Neonatal Research Network1).
The development of the fetus across the 23rd week of gestation appears to be critical to newborn survival. Hence, every day of in utero development during the 23rd week is critically important. A great challenge for obstetricians is how to approach the woman with threatened preterm birth at 22 weeks 0 days’ gestation. If the woman delivers within a few days, the likelihood of survival is minimal. However, if the pregnancy can be extended to 23 weeks and 5 days, survival rates increase significantly.
Aligning the actions of birth team, mother, and familyFactors that influence the limit of viability include:
- gestational age
- gender of the fetus (Females are more likely than males to survive.)
- treatment of the mother with glucocorticoids prior to birth
- newborn weight.
To increase the likelihood of newborn survival, obstetricians need to treat women at risk for preterm birth with antenatal glucocorticoids and antibiotics for rupture of membranes and to limit fetal stress during the birth process. Guidelines have evolved to encourage clinicians to treat women at preterm birth risk with glucocorticoids either at:
- 23 weeks’ gestation or
- 22 weeks’ gestation, if birth is anticipated to occur at 23 weeks or later.5
At birth, pediatricians are then faced with the very difficult decision of whether or not to aggressively resuscitate the severely preterm infant. Complex medical, social, and ethical issues ultimately guide pediatricians’ actions in this challenging situation. It is important for their actions to be in consensus with the obstetrician, the mother, and the mother’s family and for a consensus to be reached. Dissonant plans may increase adverse outcomes for the newborn. In one study when pediatricians and obstetricians were not aligned in their actions, the risk of death of an extremely preterm newborn significantly increased.6
Prior to birth, team meetings that include the obstetricians, pediatricians, mother, and family will help to set expectations about the course of care and, in turn, improve perceived outcomes.5 If feasible, obstetricians and pediatricians should develop joint institutional guidelines about the general approach to pregnant women when birth may occur at 22 or 23 weeks’ gestation.5
A neonatal outcomes predictor
The National Institute of Child Health and Human Development provides a Web-based tool for estimating newborn outcomes based on gestational age (22 to 25 weeks), birth weight, gender, singleton or multiple gestation, and exposure to antenatal glucocorticoid treatment. The outcomes tool provides estimates for survival and survival with severe morbidity. It uses data collected by the Neonatal Research Network to predict outcomes. To access the outcomes data assessment, visit https://www.nichd.nih.gov/about/org/der/branches/ppb/programs/epbo/Pages/epbo_case.aspx.
Is aggressive management of preterm birth and neonatal resuscitation a self-fulfilling prophecy?The beliefs and training of clinicians may influence the outcome of extremely preterm newborns. For example, if obstetricians and pediatricians focus on the fact that birth at 23 weeks is not likely to result in survival without severe morbidity, they may withhold key interventions such as antenatal glucocorticoids, antibiotics for rupture of the membranes, and aggressive newborn resuscitation.7 Consequently the likelihood of survival may be reduced.
If clinicians believe in maximal interventions for all newborns at 22 and 23 weeks’ gestation, their actions may result in a small increase in newborn survival—but at the cost of painful and unnecessary interventions in many newborns who are destined to die. Finding the right balance along the broad spectrum from expectant management to aggressive and extended resuscitation is challenging. Clearly there is no “right answer” with these extremely difficult decisions.
Future trends in the limit of viabilityIn 1963, Jacqueline Bouvier Kennedy, at 34 weeks’ gestation, went into preterm labor and delivered her son Patrick at a community hospital. Patrick developed respiratory distress syndrome and was transferred to the Boston Children’s Hospital. He died shortly thereafter.8 Would Patrick have survived if he had been delivered at an institution capable of providing high-risk obstetric and newborn services? Would such modern interventions as antenatal glucocorticoids, antibiotics for ruptured membranes, liberal use of cesarean delivery, and aggressive neonatal resuscitation have improved his chances for survival?
From our current perspective, it is surprising that a 34-week newborn died shortly after birth. With modern obstetric and pediatric care that scenario is unusual. It is possible that future advances in medical care will push the limit of viability to 22 weeks’ gestation. Future generations of clinicians may be surprised that the medicine we practice today is so limited.
However, given our current resources, it is unlikely that newborns at 22 weeks’ gestation will survive, or survive without severe morbidity. Consequently, routine aggressive resuscitation of newborns at 22 weeks should be approached with caution. At 23 weeks and later, many newborns will survive and a few will survive without severe morbidity. Given the complexity of the issues, the approach to resuscitation of infants at 22 and 23 weeks must account for the perspectives of the birth mother and her family, obstetricians, and pediatricians. Managing threatened preterm birth at 22 and 23 weeks is one of our greatest challenges as obstetricians, and we need to meet this challenge with grace and skill.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
For many decades the limit of viability was believed to be approximately 24 weeks of gestation. In many medical centers, newborns delivered at less than 25 weeks are evaluated in the delivery room and the decision to resuscitate is based on the infant’s clinical response. In the past, aggressive and extended resuscitation of newborns at 22 and 23 weeks was not common because the prognosis was bleak and clinicians did not want to inflict unnecessary pain when the chances for survival were limited. Recent advances in obstetric and pediatric care, however, have resulted in the survival of some infants born at 22 weeks’ gestation, calling into question long-held beliefs about the limits of viability.
In 2 recent reports, investigators used data from the National Institute of Child Health and Human Development (NICHD) Neonatal Research Network to acquire detailed information about newborn survival and morbidity at 22 through 28 weeks’ gestation (TABLES 1 and 2).1,2 These data show that the survival of newborns at 23 through 27 weeks’ gestation is increasing, albeit slowly. Survival, without major morbidity, is gradually improving for newborns at 25 through 28 weeks.1,2 But what is the prognosis for a fetus born at 22 or 23 weeks?


There are several aspects of this issue to consider, including accurate dating of the gestational age and current viability outcomes data.
Determining the limit of viability: Accurate dating is essentialThe limit of viability is the milestone in gestation when there is a high probability of extrauterine survival. A major challenge in studies of the limit of viability for newborns is that accurate gestational dating is not always available. For example, in recent reports from the NICHD Neonatal Research Network the gestational age was determined by the best obstetric estimate, or the Ballard or Dubowitz examination, of the newborn.1,2
It is well known that ultrasound dating early in gestation is a better estimate of gestational age than last menstrual period, uterine sizing, or pediatric examination of the newborn. Hence, the available data are limited by the absence of precise gestational dating with early ultrasound. Data on the limit of viability with large numbers of births between 22 and 24 weeks with early ultrasound dating would help to refine our understanding of the limit of viability.
At 23 weeks, each day of in utero development is criticalThe importance of each additional day spent in utero during the 23rd week of gestation was demonstrated in a small cohort in 2001.4 Overall, during the 23rd week of gestation the survival of newborns to discharge was 33%.4 This finding is similar to the survival rate reported by the NICHD Neonatal Research Network in 2012.1 However, survival was vastly different early, compared with later, in the 23rd week4:
- from 23 weeks 0 days to 23 weeks 2 days: no newborn survived
- at 23 weeks 3 days and 23 weeks 4 days: 40% of newborns survived
- at 23 weeks 5 days and 23 weeks 6 days: 63% of newborns survived (a similar survival rate of 24-week gestations was reported by the NICHD Neonatal Research Network1).
The development of the fetus across the 23rd week of gestation appears to be critical to newborn survival. Hence, every day of in utero development during the 23rd week is critically important. A great challenge for obstetricians is how to approach the woman with threatened preterm birth at 22 weeks 0 days’ gestation. If the woman delivers within a few days, the likelihood of survival is minimal. However, if the pregnancy can be extended to 23 weeks and 5 days, survival rates increase significantly.
Aligning the actions of birth team, mother, and familyFactors that influence the limit of viability include:
- gestational age
- gender of the fetus (Females are more likely than males to survive.)
- treatment of the mother with glucocorticoids prior to birth
- newborn weight.
To increase the likelihood of newborn survival, obstetricians need to treat women at risk for preterm birth with antenatal glucocorticoids and antibiotics for rupture of membranes and to limit fetal stress during the birth process. Guidelines have evolved to encourage clinicians to treat women at preterm birth risk with glucocorticoids either at:
- 23 weeks’ gestation or
- 22 weeks’ gestation, if birth is anticipated to occur at 23 weeks or later.5
At birth, pediatricians are then faced with the very difficult decision of whether or not to aggressively resuscitate the severely preterm infant. Complex medical, social, and ethical issues ultimately guide pediatricians’ actions in this challenging situation. It is important for their actions to be in consensus with the obstetrician, the mother, and the mother’s family and for a consensus to be reached. Dissonant plans may increase adverse outcomes for the newborn. In one study when pediatricians and obstetricians were not aligned in their actions, the risk of death of an extremely preterm newborn significantly increased.6
Prior to birth, team meetings that include the obstetricians, pediatricians, mother, and family will help to set expectations about the course of care and, in turn, improve perceived outcomes.5 If feasible, obstetricians and pediatricians should develop joint institutional guidelines about the general approach to pregnant women when birth may occur at 22 or 23 weeks’ gestation.5
A neonatal outcomes predictor
The National Institute of Child Health and Human Development provides a Web-based tool for estimating newborn outcomes based on gestational age (22 to 25 weeks), birth weight, gender, singleton or multiple gestation, and exposure to antenatal glucocorticoid treatment. The outcomes tool provides estimates for survival and survival with severe morbidity. It uses data collected by the Neonatal Research Network to predict outcomes. To access the outcomes data assessment, visit https://www.nichd.nih.gov/about/org/der/branches/ppb/programs/epbo/Pages/epbo_case.aspx.
Is aggressive management of preterm birth and neonatal resuscitation a self-fulfilling prophecy?The beliefs and training of clinicians may influence the outcome of extremely preterm newborns. For example, if obstetricians and pediatricians focus on the fact that birth at 23 weeks is not likely to result in survival without severe morbidity, they may withhold key interventions such as antenatal glucocorticoids, antibiotics for rupture of the membranes, and aggressive newborn resuscitation.7 Consequently the likelihood of survival may be reduced.
If clinicians believe in maximal interventions for all newborns at 22 and 23 weeks’ gestation, their actions may result in a small increase in newborn survival—but at the cost of painful and unnecessary interventions in many newborns who are destined to die. Finding the right balance along the broad spectrum from expectant management to aggressive and extended resuscitation is challenging. Clearly there is no “right answer” with these extremely difficult decisions.
Future trends in the limit of viabilityIn 1963, Jacqueline Bouvier Kennedy, at 34 weeks’ gestation, went into preterm labor and delivered her son Patrick at a community hospital. Patrick developed respiratory distress syndrome and was transferred to the Boston Children’s Hospital. He died shortly thereafter.8 Would Patrick have survived if he had been delivered at an institution capable of providing high-risk obstetric and newborn services? Would such modern interventions as antenatal glucocorticoids, antibiotics for ruptured membranes, liberal use of cesarean delivery, and aggressive neonatal resuscitation have improved his chances for survival?
From our current perspective, it is surprising that a 34-week newborn died shortly after birth. With modern obstetric and pediatric care that scenario is unusual. It is possible that future advances in medical care will push the limit of viability to 22 weeks’ gestation. Future generations of clinicians may be surprised that the medicine we practice today is so limited.
However, given our current resources, it is unlikely that newborns at 22 weeks’ gestation will survive, or survive without severe morbidity. Consequently, routine aggressive resuscitation of newborns at 22 weeks should be approached with caution. At 23 weeks and later, many newborns will survive and a few will survive without severe morbidity. Given the complexity of the issues, the approach to resuscitation of infants at 22 and 23 weeks must account for the perspectives of the birth mother and her family, obstetricians, and pediatricians. Managing threatened preterm birth at 22 and 23 weeks is one of our greatest challenges as obstetricians, and we need to meet this challenge with grace and skill.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Stoll BJ, Hansen NI, Bell EF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Trends in care practices, morbidity and mortality of extremely preterm neonates, 1993-2012. JAMA. 2015;314(10):1039–1051.
- Patel RM, Kandefer S, Walsh MC, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Causes and timing of death in extremely premature infants from 2000 through 2011. N Engl J Med. 2015;372(4):331–340.
- Donovan EF, Tyson JE, Ehrenkranz RA, et al. Inaccuracy of Ballard scores before 28 weeks’ gestation. National Institute of Child Health and Human Development Neonatal Research Network. J Pediatr. 1999;135(2 pt 1):147–152.
- McElrath TF, Robinson JN, Ecker JL, Ringer SA, Norwitz ER. Neonatal outcome of infants born at 23 weeks’ gestation. Obstet Gynecol. 2001;97(1):49–52.
- Raju TN, Mercer BM, Burchfield DJ, Joseph GF Jr. Periviable birth: executive summary of a joint workshop by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for Maternal-Fetal Medicine, American Academy of Pediatrics, and American College of Obstetricians and Gynecologists. Obstet Gynecol. 2014;123(5):1083–1096.
- Guinsburg R, Branco de Almeida MF, dos Santos Rodrigues Sadeck L, et al; Brazilian Network on Neonatal Research. Proactive management of extreme prematurity: disagreement between obstetricians and neonatologists. J Perinatol. 2012;32(12):913-919.
- Tucker Emonds B, McKenzie F, Farrow V, Raglan G, Schulkin J. A national survey of obstetricians’ attitudes toward and practice of periviable interventions. J Perinatol. 2015;35(5):338–343.
- Altman LK. A Kennedy baby’s life and death. New York Times. http://www.nytimes.com/2013/07/30/health/a-kennedy-babys-life-and-death.html?_r=0. Published July 29, 2013. Accessed November 19, 2015.
- Stoll BJ, Hansen NI, Bell EF, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Trends in care practices, morbidity and mortality of extremely preterm neonates, 1993-2012. JAMA. 2015;314(10):1039–1051.
- Patel RM, Kandefer S, Walsh MC, et al; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Causes and timing of death in extremely premature infants from 2000 through 2011. N Engl J Med. 2015;372(4):331–340.
- Donovan EF, Tyson JE, Ehrenkranz RA, et al. Inaccuracy of Ballard scores before 28 weeks’ gestation. National Institute of Child Health and Human Development Neonatal Research Network. J Pediatr. 1999;135(2 pt 1):147–152.
- McElrath TF, Robinson JN, Ecker JL, Ringer SA, Norwitz ER. Neonatal outcome of infants born at 23 weeks’ gestation. Obstet Gynecol. 2001;97(1):49–52.
- Raju TN, Mercer BM, Burchfield DJ, Joseph GF Jr. Periviable birth: executive summary of a joint workshop by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for Maternal-Fetal Medicine, American Academy of Pediatrics, and American College of Obstetricians and Gynecologists. Obstet Gynecol. 2014;123(5):1083–1096.
- Guinsburg R, Branco de Almeida MF, dos Santos Rodrigues Sadeck L, et al; Brazilian Network on Neonatal Research. Proactive management of extreme prematurity: disagreement between obstetricians and neonatologists. J Perinatol. 2012;32(12):913-919.
- Tucker Emonds B, McKenzie F, Farrow V, Raglan G, Schulkin J. A national survey of obstetricians’ attitudes toward and practice of periviable interventions. J Perinatol. 2015;35(5):338–343.
- Altman LK. A Kennedy baby’s life and death. New York Times. http://www.nytimes.com/2013/07/30/health/a-kennedy-babys-life-and-death.html?_r=0. Published July 29, 2013. Accessed November 19, 2015.
It’s not the noise, it’s what you do with it
In his physician-coming-of-age novel House of God, published in 1978, Dr. Steven Bergman (aka Sam Shem) presented rules for an intern’s survival devised by the senior resident, the Fat Man. Rule X was that there is no fever if you don’t check the patient’s temperature, implying that if the physician is unaware of an elevated temperature, no “fever workup” is warranted. A fever workup back then was not just a few keystrokes to order a chest x-ray, complete blood cell count, and blood cultures. The intern had to go to the bedside, awaken and examine the patient, draw the blood, perhaps transport the blood samples to the lab, do a urinalysis, and take the patient to the radiology department to get the chest x-ray. There was often little thought to the intern’s action; a fever in the hospital automatically meant there needed to be a fever workup.
A covering senior resident might have gotten the same notification of a fever, quickly reviewed the chart, gone to the bedside, and assessed whether a bacterial infection was likely enough to warrant the time and annoyance of a full fever workup. As supervising faculty, I will accept that assessment from a senior resident in June more willingly than from an intern in July. Tests and physical findings must be evaluated in context, taking into consideration the patient as well as the skill and experience of the physician.
So how should we react to guidelines that seem to be based on the premise that a positive finding will result in reflexive ordering of additional tests or initiating a therapeutic intervention, and thus should be avoided by all of us—young intern and senior cardiologist alike?
In this issue of the Journal, Dr. Aldo Schenone et al discuss the management of the asymptomatic patient who has carotid artery stenosis. They put into perspective the risks and benefits of medical or surgical intervention as initially defined by several landmark trials, noting how those conclusions should now be modified by knowledge of the efficacy of current medical therapy.
The US Preventive Services Task Force (USPSTF)1 has recommended against screening for asymptomatic carotid artery stenosis in the general population, noting the limited sensitivity (71%) and specificity (98%) of auscultation to diagnose significant stenosis and lumping it with other ineffective screening tests. In other words, we should not examine asymptomatic patients for carotid bruits, just as we should not look for the fever because finding it could lead to additional testing and potentially unnecessary therapy.
But there are broader implications when a bruit is discovered, beyond simply trodding the algorithmic path toward stenting or endarterectomy. A bruit can suggest occult atherosclerotic disease that warrants medical attention, even if traditional risk factors for atherosclerosis are not prominent. Its discovery can be a wake-up call to the patient (and physician) that the hackneyed admonitions to eat healthy, lose weight, and exercise are actually relevant. Its discovery may lead to medical intervention with a potent statin or with a more aggressive target for blood pressure control. It may color the interpretation of the patient’s described vague arm tingling when bowling.
I may well be misleading myself, but I am more comfortable in dealing with whatever oddities I discover on a physical examination than not doing the examination at all. I’d rather know about the bruit (or the fever) and then think about our options. The stethoscope indeed has limited test reliability, but the real action takes place between its earpieces; the bruit is merely the catalyst for thought. There must be a guideline somewhere that says that a thoughtful, informed, commonsense evaluation is a useful contributor to patient care.
- LeFevre ML; US Preventive Services Task Force. Screening for asymptomatic carotid artery stenosis: US Preventive Services Task Force recommendation statement. Ann Intern Med 2014; 161:356–362.
In his physician-coming-of-age novel House of God, published in 1978, Dr. Steven Bergman (aka Sam Shem) presented rules for an intern’s survival devised by the senior resident, the Fat Man. Rule X was that there is no fever if you don’t check the patient’s temperature, implying that if the physician is unaware of an elevated temperature, no “fever workup” is warranted. A fever workup back then was not just a few keystrokes to order a chest x-ray, complete blood cell count, and blood cultures. The intern had to go to the bedside, awaken and examine the patient, draw the blood, perhaps transport the blood samples to the lab, do a urinalysis, and take the patient to the radiology department to get the chest x-ray. There was often little thought to the intern’s action; a fever in the hospital automatically meant there needed to be a fever workup.
A covering senior resident might have gotten the same notification of a fever, quickly reviewed the chart, gone to the bedside, and assessed whether a bacterial infection was likely enough to warrant the time and annoyance of a full fever workup. As supervising faculty, I will accept that assessment from a senior resident in June more willingly than from an intern in July. Tests and physical findings must be evaluated in context, taking into consideration the patient as well as the skill and experience of the physician.
So how should we react to guidelines that seem to be based on the premise that a positive finding will result in reflexive ordering of additional tests or initiating a therapeutic intervention, and thus should be avoided by all of us—young intern and senior cardiologist alike?
In this issue of the Journal, Dr. Aldo Schenone et al discuss the management of the asymptomatic patient who has carotid artery stenosis. They put into perspective the risks and benefits of medical or surgical intervention as initially defined by several landmark trials, noting how those conclusions should now be modified by knowledge of the efficacy of current medical therapy.
The US Preventive Services Task Force (USPSTF)1 has recommended against screening for asymptomatic carotid artery stenosis in the general population, noting the limited sensitivity (71%) and specificity (98%) of auscultation to diagnose significant stenosis and lumping it with other ineffective screening tests. In other words, we should not examine asymptomatic patients for carotid bruits, just as we should not look for the fever because finding it could lead to additional testing and potentially unnecessary therapy.
But there are broader implications when a bruit is discovered, beyond simply trodding the algorithmic path toward stenting or endarterectomy. A bruit can suggest occult atherosclerotic disease that warrants medical attention, even if traditional risk factors for atherosclerosis are not prominent. Its discovery can be a wake-up call to the patient (and physician) that the hackneyed admonitions to eat healthy, lose weight, and exercise are actually relevant. Its discovery may lead to medical intervention with a potent statin or with a more aggressive target for blood pressure control. It may color the interpretation of the patient’s described vague arm tingling when bowling.
I may well be misleading myself, but I am more comfortable in dealing with whatever oddities I discover on a physical examination than not doing the examination at all. I’d rather know about the bruit (or the fever) and then think about our options. The stethoscope indeed has limited test reliability, but the real action takes place between its earpieces; the bruit is merely the catalyst for thought. There must be a guideline somewhere that says that a thoughtful, informed, commonsense evaluation is a useful contributor to patient care.
In his physician-coming-of-age novel House of God, published in 1978, Dr. Steven Bergman (aka Sam Shem) presented rules for an intern’s survival devised by the senior resident, the Fat Man. Rule X was that there is no fever if you don’t check the patient’s temperature, implying that if the physician is unaware of an elevated temperature, no “fever workup” is warranted. A fever workup back then was not just a few keystrokes to order a chest x-ray, complete blood cell count, and blood cultures. The intern had to go to the bedside, awaken and examine the patient, draw the blood, perhaps transport the blood samples to the lab, do a urinalysis, and take the patient to the radiology department to get the chest x-ray. There was often little thought to the intern’s action; a fever in the hospital automatically meant there needed to be a fever workup.
A covering senior resident might have gotten the same notification of a fever, quickly reviewed the chart, gone to the bedside, and assessed whether a bacterial infection was likely enough to warrant the time and annoyance of a full fever workup. As supervising faculty, I will accept that assessment from a senior resident in June more willingly than from an intern in July. Tests and physical findings must be evaluated in context, taking into consideration the patient as well as the skill and experience of the physician.
So how should we react to guidelines that seem to be based on the premise that a positive finding will result in reflexive ordering of additional tests or initiating a therapeutic intervention, and thus should be avoided by all of us—young intern and senior cardiologist alike?
In this issue of the Journal, Dr. Aldo Schenone et al discuss the management of the asymptomatic patient who has carotid artery stenosis. They put into perspective the risks and benefits of medical or surgical intervention as initially defined by several landmark trials, noting how those conclusions should now be modified by knowledge of the efficacy of current medical therapy.
The US Preventive Services Task Force (USPSTF)1 has recommended against screening for asymptomatic carotid artery stenosis in the general population, noting the limited sensitivity (71%) and specificity (98%) of auscultation to diagnose significant stenosis and lumping it with other ineffective screening tests. In other words, we should not examine asymptomatic patients for carotid bruits, just as we should not look for the fever because finding it could lead to additional testing and potentially unnecessary therapy.
But there are broader implications when a bruit is discovered, beyond simply trodding the algorithmic path toward stenting or endarterectomy. A bruit can suggest occult atherosclerotic disease that warrants medical attention, even if traditional risk factors for atherosclerosis are not prominent. Its discovery can be a wake-up call to the patient (and physician) that the hackneyed admonitions to eat healthy, lose weight, and exercise are actually relevant. Its discovery may lead to medical intervention with a potent statin or with a more aggressive target for blood pressure control. It may color the interpretation of the patient’s described vague arm tingling when bowling.
I may well be misleading myself, but I am more comfortable in dealing with whatever oddities I discover on a physical examination than not doing the examination at all. I’d rather know about the bruit (or the fever) and then think about our options. The stethoscope indeed has limited test reliability, but the real action takes place between its earpieces; the bruit is merely the catalyst for thought. There must be a guideline somewhere that says that a thoughtful, informed, commonsense evaluation is a useful contributor to patient care.
- LeFevre ML; US Preventive Services Task Force. Screening for asymptomatic carotid artery stenosis: US Preventive Services Task Force recommendation statement. Ann Intern Med 2014; 161:356–362.
- LeFevre ML; US Preventive Services Task Force. Screening for asymptomatic carotid artery stenosis: US Preventive Services Task Force recommendation statement. Ann Intern Med 2014; 161:356–362.
A decade after the CATIE study, the focus has shifted from effectiveness to neuroprotection
This past September, exactly 10 years after publication of the primary findings of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study1—namely, that effectiveness (defined as all-cause discontinuation) was not different across first-generation antipsychotics (FGAs) and second generation antipsychotics (SGAs)— a new meta-analysis by Vita et al2 of differences in cortical gray-matter change between those 2 classes of antipsychotics offers a reminder: The clinical focus of the CATIE study overlooked important neurobiological and neuroprotective differences between FGAs and SGAs.
How drastically 1 decade can change the scientific perspective! Vita et al’s meta-analysis and meta-regression encompassed all 18 MRI studies of cortical gray matter in patients with schizophrenia.2 Earlier studies (published between 1983 and 2014) had lumped together patients who were receiving an FGA and those receiving an SGA, and authors reported overall reduction in cortical gray matter with prolonged antipsychotic treatment.
Remarkable findings emerge
When Vita et al2 analyzed FGA- and SGA-treated patients separately, however, they found a significant reduction in cortical gray matter in the FGA group but not in the SGA group. In fact, while higher daily dosages of FGAs were associated with greater reduction in cortical gray matter, higher dosages of SGAs were associated with lower cortical gray matter reduction and, in some samples, with an increase in volume of cortical gray matter.
The researchers hypothesized that the differential effects of FGAs and SGAs might be attributable to the neurotoxicity of typical FGAs and the neuroprotective effect of atypical SGAs.
Hindsight
The key neurobiological difference between FGAs and SGAs reported by Vita et al2 was not addressed in the CATIE study, leading, at that time, to a rush to judgment that all antipsychotics are the same. This conclusion emboldened managed-care organizations to mandate use of older (and cheaper) generic FGAs instead of newer (and more expensive) SGAs— most of which have become available as generic equivalents since the CATIE study was completed.
Investigators in the CATIE study— of which I was one—cannot be blamed for not focusing on neurotoxicity and neuroprotection; those data were not on the psychiatry’s radar when the CATIE study was designed in 1998. The major focus was on whether SGAs (new on the scene in the late 1990s) were more efficacious, safe, and tolerable (that is, more effective) than FGAs.
In fact, the first study reporting that SGAs stimulated neurogenesis (in animals) was published in 2002,3 when the CATIE study was more than half complete. Research into the neuroprotective properties of SGAs then grew rapidly. In fact, the principal investigator of the CATIE study conducted a head-to-head comparison of FGA haloperidol and SGA olanzapine in a sample of first-episode schizophrenia patients4; over 1 year of follow-up, it was determined that patients in the haloperidol-treated group exhibited significant brain volume loss on MRI but those in the olanzapine-treated group did not. This study was published in 2005—the same year the CATIE study was published!
SGAs offer neuroprotection
Over the past decade, the neuroprotective effects of SGAs5 and the neurotoxic effects of FGAs6 have been studied intensively, revealing that SGAs have multiple neuroprotective effects. These effects include:
• stimulation of the production of new brain cells (neurons and glia), known as neurogenesis5,7,8
• an increase in neurotrophic factors, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF),9 which are found at a significantly low level in patients with psychosis10
• reversal of phencyclidine (PCP)-induced changes in gene expression11
• neuroprotection against ischemic stroke12-14
• reversal of PCP-induced loss of dendritic spines in the frontal cortex15
• prevention of oligodendrocyte damage caused by interferon gamma-stimulated microglia16,17
• reversal of loss of dendritic spines in the prefrontal cortex induced by dopamine depletion18
• an anti-inflammatory effect19,20
• protection against β-amyloid and hydrogen peroxide-induced cell death21
• protection against prefrontal cortical neuronal damage caused by dizocilpine (MK-801)22
• reversal of a PCP-induced decrease in the glutathione level and alteration of antioxidant defenses23
• protection of cortical neurons from glutamate neurotoxicity.24
One reason why SGAs are neuroprotective, but FGAs are not, can be attributed to their receptor profiles. FGAs block dopamine D2 receptors far more than serotonin 2A receptors, whereas SGAs do the opposite: They block 5-HT2A receptors 500% to 1,000% more than they block D2 receptors. This difference is associated in turn with a different neurobiological and neuroprotective profiles, such as a decrease or an increase in BDNF.25,26
Neither similar nor interchangeable
Since publication of the findings of the CATIE study, the primary investigator has proposed that neuroprotection can be a therapeutic strategy to prevent neurodegeneration and neurodeterioration associated with schizophrenia.27 Given the preponderance of data showing that SGAs have numerous neuroprotective properties but FGAs have many neurotoxic effects,6 the message to psychiatric practitioners, a decade after the CATIE study, is that the 2 generations of antipsychotic agents are not really similar or interchangeable. They might have similar clinical effectiveness but they exert very different neurobiological effects.
The proof of the pudding is in the eating: Despite the findings of the CATIE study, the vast majority of psychiatrists would prefer to treat their own family members with an SGA, not an FGA, if the need for antipsychotic medication arises.
1. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223.
2. Vita A, De Peri L, Deste G, et al. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol Psychiatry. 2015;78(6):403-412.
3. Wakade CG, Mahadik SP, Waller JL, et al. Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res. 2002;69(1):72-79.
4. Lieberman JA, Tollefson GD, Charles C, et al; HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370.
5. Nasrallah HA. Impaired neuroplasticity in schizophrenia and the neuro-regenerative effects of atypical antipsychotics. Medscape Psychiatry. http://www.medscape.org/viewarticle/569521. Published January 31, 2008. Accessed November 10, 2015.
6. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2012;12(7):7-8.
7. Nandra KS, Agius M. The differences between typical and atypical antipsychotics: the effects on neurogenesis. Psychiatr Danub. 2012;24(suppl 1):S95-S99.
8. Nasrallah HA, Hopkins T, Pixley SK, et al. Differential effects of antipsychotic and antidepressant drugs on neurogenic region in rats. Brain Res. 2010;354:23-29.
9. Pillai A, Tery AV, Mahadik SP. Differential effects of long-term treatment with typical and atypical antipsychotics on NGF and BNDF levels in rat striatum and hippocampus. Schizophr Res. 2006;82(1):95-106.
10. Buckley PF, Pillai A, Evans D, et al. Brain derived neurotropic factor in first-episode psychosis. Schizophr Res. 2007;91(1-3):1-5.
11. Martin MV, Mimics K, Nisenbaum LK, et al. Olanzapine reversed brain gene expression changes induced by phencyclidines treatment in non-human primates. Mol Neuropsychiatry. 2015;1(2):82-93.
12. Yan BC, Park JH, Ahn JH, et al. Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res. 2014;92(6):795-807.
13. Yulug B, Yildiz A, Güzel O, et al. Risperidone attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;69(6):656-659.
14. Yulug B, Yildiz A, Hüdaoglu O, et al. Olanzapine attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;71(1-3):296-300.
15. Elsworth JD, Morrow BA. Hajszan T, et al. Phencyclidine-induced loss of asymmetric spine synapses in rodent prefrontal cortex is reversed by acute and chronic treatment with olanzapine. Neuropsychopharmacology. 2001;36(10):2054-2061.
16. Seki Y, Kato TA, Monji A, et al. Pretreatment of aripiprazole and minocycline, but not haloperidol, suppresses oligodendrocyte damage from interferon-y-stimulated microglia in co-culture model. Schizophr Res. 2013;151(1-3):20-28.
17. Bian Q, Kato T, Monji A, et al. The effect of atypical antipsychotics, perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
18. Wang HD, Deutch AY. Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment. Neuropsychopharmacology. 2008;33(6):1276-1286.
19. Miller BJ, Buckley P, Seabolt W, et al. Meta-analysis of cytokine alternations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663-671.
20. Nasrallah HA. Beyond dopamine: The ‘other’ effects of antipsychotics. Current Psychiatry. 2013;12(6):8-9.
21. Yang MC, Lung FW. Neuroprotection of paliperidone on SH-SY5Y cells against β-amyloid peptide(25-35), N-methyl-4-phenylpyridinium ion, and hydrogen peroxide-induced cell death. Psychopharmacology (Berl). 2011;217(3):397-410.
22. Peng L, Zhu D, Feng X, et al. Paliperidone protects prefrontal cortical neurons from damages caused by MK-801 via Akt1/GSK3β _signaling pathway. Schizophr Res. 2013;147(1):14-23.23.
Stojkovic´ T, Radonjic´ NV, Velimirovic´ M, et al. Risperidone reverses phencyclidine induced decrease in glutathione levels and alternations of antioxidant defense in rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39(1):192-199.
24. Koprivica V, Regardie K, Wolff C, et al. Aripiprazole protects cortical neurons from glutamate toxicity. Eur J Pharmacol. 2011;651(1-3):73-76.
25. Vaidya VA, Marek GJ, Aghajanian GK, et al. 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J Neurosci. 1997;17(8):2785-2795.
26. Meridith GE, Switzer RC 3rd, Napier TC. Short-term, D2 receptor blockade induces synaptic degeneration, reduces levels of tyrosine hydroxylase and brain-derived neurotrophic factor, and enhances D2-mediated firing in the ventral pallidum. Brain Res. 2004;995(1):14-22.
27. Lieberman JA, Perkins DO, Jarskog LF. Neuroprotection: a therapeutic strategy to prevent deterioration associated with schizophrenia. CNS Spectr. 2007;12(suppl 4):1-13; quiz 14.
This past September, exactly 10 years after publication of the primary findings of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study1—namely, that effectiveness (defined as all-cause discontinuation) was not different across first-generation antipsychotics (FGAs) and second generation antipsychotics (SGAs)— a new meta-analysis by Vita et al2 of differences in cortical gray-matter change between those 2 classes of antipsychotics offers a reminder: The clinical focus of the CATIE study overlooked important neurobiological and neuroprotective differences between FGAs and SGAs.
How drastically 1 decade can change the scientific perspective! Vita et al’s meta-analysis and meta-regression encompassed all 18 MRI studies of cortical gray matter in patients with schizophrenia.2 Earlier studies (published between 1983 and 2014) had lumped together patients who were receiving an FGA and those receiving an SGA, and authors reported overall reduction in cortical gray matter with prolonged antipsychotic treatment.
Remarkable findings emerge
When Vita et al2 analyzed FGA- and SGA-treated patients separately, however, they found a significant reduction in cortical gray matter in the FGA group but not in the SGA group. In fact, while higher daily dosages of FGAs were associated with greater reduction in cortical gray matter, higher dosages of SGAs were associated with lower cortical gray matter reduction and, in some samples, with an increase in volume of cortical gray matter.
The researchers hypothesized that the differential effects of FGAs and SGAs might be attributable to the neurotoxicity of typical FGAs and the neuroprotective effect of atypical SGAs.
Hindsight
The key neurobiological difference between FGAs and SGAs reported by Vita et al2 was not addressed in the CATIE study, leading, at that time, to a rush to judgment that all antipsychotics are the same. This conclusion emboldened managed-care organizations to mandate use of older (and cheaper) generic FGAs instead of newer (and more expensive) SGAs— most of which have become available as generic equivalents since the CATIE study was completed.
Investigators in the CATIE study— of which I was one—cannot be blamed for not focusing on neurotoxicity and neuroprotection; those data were not on the psychiatry’s radar when the CATIE study was designed in 1998. The major focus was on whether SGAs (new on the scene in the late 1990s) were more efficacious, safe, and tolerable (that is, more effective) than FGAs.
In fact, the first study reporting that SGAs stimulated neurogenesis (in animals) was published in 2002,3 when the CATIE study was more than half complete. Research into the neuroprotective properties of SGAs then grew rapidly. In fact, the principal investigator of the CATIE study conducted a head-to-head comparison of FGA haloperidol and SGA olanzapine in a sample of first-episode schizophrenia patients4; over 1 year of follow-up, it was determined that patients in the haloperidol-treated group exhibited significant brain volume loss on MRI but those in the olanzapine-treated group did not. This study was published in 2005—the same year the CATIE study was published!
SGAs offer neuroprotection
Over the past decade, the neuroprotective effects of SGAs5 and the neurotoxic effects of FGAs6 have been studied intensively, revealing that SGAs have multiple neuroprotective effects. These effects include:
• stimulation of the production of new brain cells (neurons and glia), known as neurogenesis5,7,8
• an increase in neurotrophic factors, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF),9 which are found at a significantly low level in patients with psychosis10
• reversal of phencyclidine (PCP)-induced changes in gene expression11
• neuroprotection against ischemic stroke12-14
• reversal of PCP-induced loss of dendritic spines in the frontal cortex15
• prevention of oligodendrocyte damage caused by interferon gamma-stimulated microglia16,17
• reversal of loss of dendritic spines in the prefrontal cortex induced by dopamine depletion18
• an anti-inflammatory effect19,20
• protection against β-amyloid and hydrogen peroxide-induced cell death21
• protection against prefrontal cortical neuronal damage caused by dizocilpine (MK-801)22
• reversal of a PCP-induced decrease in the glutathione level and alteration of antioxidant defenses23
• protection of cortical neurons from glutamate neurotoxicity.24
One reason why SGAs are neuroprotective, but FGAs are not, can be attributed to their receptor profiles. FGAs block dopamine D2 receptors far more than serotonin 2A receptors, whereas SGAs do the opposite: They block 5-HT2A receptors 500% to 1,000% more than they block D2 receptors. This difference is associated in turn with a different neurobiological and neuroprotective profiles, such as a decrease or an increase in BDNF.25,26
Neither similar nor interchangeable
Since publication of the findings of the CATIE study, the primary investigator has proposed that neuroprotection can be a therapeutic strategy to prevent neurodegeneration and neurodeterioration associated with schizophrenia.27 Given the preponderance of data showing that SGAs have numerous neuroprotective properties but FGAs have many neurotoxic effects,6 the message to psychiatric practitioners, a decade after the CATIE study, is that the 2 generations of antipsychotic agents are not really similar or interchangeable. They might have similar clinical effectiveness but they exert very different neurobiological effects.
The proof of the pudding is in the eating: Despite the findings of the CATIE study, the vast majority of psychiatrists would prefer to treat their own family members with an SGA, not an FGA, if the need for antipsychotic medication arises.
This past September, exactly 10 years after publication of the primary findings of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study1—namely, that effectiveness (defined as all-cause discontinuation) was not different across first-generation antipsychotics (FGAs) and second generation antipsychotics (SGAs)— a new meta-analysis by Vita et al2 of differences in cortical gray-matter change between those 2 classes of antipsychotics offers a reminder: The clinical focus of the CATIE study overlooked important neurobiological and neuroprotective differences between FGAs and SGAs.
How drastically 1 decade can change the scientific perspective! Vita et al’s meta-analysis and meta-regression encompassed all 18 MRI studies of cortical gray matter in patients with schizophrenia.2 Earlier studies (published between 1983 and 2014) had lumped together patients who were receiving an FGA and those receiving an SGA, and authors reported overall reduction in cortical gray matter with prolonged antipsychotic treatment.
Remarkable findings emerge
When Vita et al2 analyzed FGA- and SGA-treated patients separately, however, they found a significant reduction in cortical gray matter in the FGA group but not in the SGA group. In fact, while higher daily dosages of FGAs were associated with greater reduction in cortical gray matter, higher dosages of SGAs were associated with lower cortical gray matter reduction and, in some samples, with an increase in volume of cortical gray matter.
The researchers hypothesized that the differential effects of FGAs and SGAs might be attributable to the neurotoxicity of typical FGAs and the neuroprotective effect of atypical SGAs.
Hindsight
The key neurobiological difference between FGAs and SGAs reported by Vita et al2 was not addressed in the CATIE study, leading, at that time, to a rush to judgment that all antipsychotics are the same. This conclusion emboldened managed-care organizations to mandate use of older (and cheaper) generic FGAs instead of newer (and more expensive) SGAs— most of which have become available as generic equivalents since the CATIE study was completed.
Investigators in the CATIE study— of which I was one—cannot be blamed for not focusing on neurotoxicity and neuroprotection; those data were not on the psychiatry’s radar when the CATIE study was designed in 1998. The major focus was on whether SGAs (new on the scene in the late 1990s) were more efficacious, safe, and tolerable (that is, more effective) than FGAs.
In fact, the first study reporting that SGAs stimulated neurogenesis (in animals) was published in 2002,3 when the CATIE study was more than half complete. Research into the neuroprotective properties of SGAs then grew rapidly. In fact, the principal investigator of the CATIE study conducted a head-to-head comparison of FGA haloperidol and SGA olanzapine in a sample of first-episode schizophrenia patients4; over 1 year of follow-up, it was determined that patients in the haloperidol-treated group exhibited significant brain volume loss on MRI but those in the olanzapine-treated group did not. This study was published in 2005—the same year the CATIE study was published!
SGAs offer neuroprotection
Over the past decade, the neuroprotective effects of SGAs5 and the neurotoxic effects of FGAs6 have been studied intensively, revealing that SGAs have multiple neuroprotective effects. These effects include:
• stimulation of the production of new brain cells (neurons and glia), known as neurogenesis5,7,8
• an increase in neurotrophic factors, such as nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF),9 which are found at a significantly low level in patients with psychosis10
• reversal of phencyclidine (PCP)-induced changes in gene expression11
• neuroprotection against ischemic stroke12-14
• reversal of PCP-induced loss of dendritic spines in the frontal cortex15
• prevention of oligodendrocyte damage caused by interferon gamma-stimulated microglia16,17
• reversal of loss of dendritic spines in the prefrontal cortex induced by dopamine depletion18
• an anti-inflammatory effect19,20
• protection against β-amyloid and hydrogen peroxide-induced cell death21
• protection against prefrontal cortical neuronal damage caused by dizocilpine (MK-801)22
• reversal of a PCP-induced decrease in the glutathione level and alteration of antioxidant defenses23
• protection of cortical neurons from glutamate neurotoxicity.24
One reason why SGAs are neuroprotective, but FGAs are not, can be attributed to their receptor profiles. FGAs block dopamine D2 receptors far more than serotonin 2A receptors, whereas SGAs do the opposite: They block 5-HT2A receptors 500% to 1,000% more than they block D2 receptors. This difference is associated in turn with a different neurobiological and neuroprotective profiles, such as a decrease or an increase in BDNF.25,26
Neither similar nor interchangeable
Since publication of the findings of the CATIE study, the primary investigator has proposed that neuroprotection can be a therapeutic strategy to prevent neurodegeneration and neurodeterioration associated with schizophrenia.27 Given the preponderance of data showing that SGAs have numerous neuroprotective properties but FGAs have many neurotoxic effects,6 the message to psychiatric practitioners, a decade after the CATIE study, is that the 2 generations of antipsychotic agents are not really similar or interchangeable. They might have similar clinical effectiveness but they exert very different neurobiological effects.
The proof of the pudding is in the eating: Despite the findings of the CATIE study, the vast majority of psychiatrists would prefer to treat their own family members with an SGA, not an FGA, if the need for antipsychotic medication arises.
1. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223.
2. Vita A, De Peri L, Deste G, et al. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol Psychiatry. 2015;78(6):403-412.
3. Wakade CG, Mahadik SP, Waller JL, et al. Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res. 2002;69(1):72-79.
4. Lieberman JA, Tollefson GD, Charles C, et al; HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370.
5. Nasrallah HA. Impaired neuroplasticity in schizophrenia and the neuro-regenerative effects of atypical antipsychotics. Medscape Psychiatry. http://www.medscape.org/viewarticle/569521. Published January 31, 2008. Accessed November 10, 2015.
6. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2012;12(7):7-8.
7. Nandra KS, Agius M. The differences between typical and atypical antipsychotics: the effects on neurogenesis. Psychiatr Danub. 2012;24(suppl 1):S95-S99.
8. Nasrallah HA, Hopkins T, Pixley SK, et al. Differential effects of antipsychotic and antidepressant drugs on neurogenic region in rats. Brain Res. 2010;354:23-29.
9. Pillai A, Tery AV, Mahadik SP. Differential effects of long-term treatment with typical and atypical antipsychotics on NGF and BNDF levels in rat striatum and hippocampus. Schizophr Res. 2006;82(1):95-106.
10. Buckley PF, Pillai A, Evans D, et al. Brain derived neurotropic factor in first-episode psychosis. Schizophr Res. 2007;91(1-3):1-5.
11. Martin MV, Mimics K, Nisenbaum LK, et al. Olanzapine reversed brain gene expression changes induced by phencyclidines treatment in non-human primates. Mol Neuropsychiatry. 2015;1(2):82-93.
12. Yan BC, Park JH, Ahn JH, et al. Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res. 2014;92(6):795-807.
13. Yulug B, Yildiz A, Güzel O, et al. Risperidone attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;69(6):656-659.
14. Yulug B, Yildiz A, Hüdaoglu O, et al. Olanzapine attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;71(1-3):296-300.
15. Elsworth JD, Morrow BA. Hajszan T, et al. Phencyclidine-induced loss of asymmetric spine synapses in rodent prefrontal cortex is reversed by acute and chronic treatment with olanzapine. Neuropsychopharmacology. 2001;36(10):2054-2061.
16. Seki Y, Kato TA, Monji A, et al. Pretreatment of aripiprazole and minocycline, but not haloperidol, suppresses oligodendrocyte damage from interferon-y-stimulated microglia in co-culture model. Schizophr Res. 2013;151(1-3):20-28.
17. Bian Q, Kato T, Monji A, et al. The effect of atypical antipsychotics, perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
18. Wang HD, Deutch AY. Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment. Neuropsychopharmacology. 2008;33(6):1276-1286.
19. Miller BJ, Buckley P, Seabolt W, et al. Meta-analysis of cytokine alternations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663-671.
20. Nasrallah HA. Beyond dopamine: The ‘other’ effects of antipsychotics. Current Psychiatry. 2013;12(6):8-9.
21. Yang MC, Lung FW. Neuroprotection of paliperidone on SH-SY5Y cells against β-amyloid peptide(25-35), N-methyl-4-phenylpyridinium ion, and hydrogen peroxide-induced cell death. Psychopharmacology (Berl). 2011;217(3):397-410.
22. Peng L, Zhu D, Feng X, et al. Paliperidone protects prefrontal cortical neurons from damages caused by MK-801 via Akt1/GSK3β _signaling pathway. Schizophr Res. 2013;147(1):14-23.23.
Stojkovic´ T, Radonjic´ NV, Velimirovic´ M, et al. Risperidone reverses phencyclidine induced decrease in glutathione levels and alternations of antioxidant defense in rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39(1):192-199.
24. Koprivica V, Regardie K, Wolff C, et al. Aripiprazole protects cortical neurons from glutamate toxicity. Eur J Pharmacol. 2011;651(1-3):73-76.
25. Vaidya VA, Marek GJ, Aghajanian GK, et al. 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J Neurosci. 1997;17(8):2785-2795.
26. Meridith GE, Switzer RC 3rd, Napier TC. Short-term, D2 receptor blockade induces synaptic degeneration, reduces levels of tyrosine hydroxylase and brain-derived neurotrophic factor, and enhances D2-mediated firing in the ventral pallidum. Brain Res. 2004;995(1):14-22.
27. Lieberman JA, Perkins DO, Jarskog LF. Neuroprotection: a therapeutic strategy to prevent deterioration associated with schizophrenia. CNS Spectr. 2007;12(suppl 4):1-13; quiz 14.
1. Lieberman JA, Stroup TS, McEvoy JP, et al; Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Investigators. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353(12):1209-1223.
2. Vita A, De Peri L, Deste G, et al. The effect of antipsychotic treatment on cortical gray matter changes in schizophrenia: does the class matter? A meta-analysis and meta-regression of longitudinal magnetic resonance imaging studies. Biol Psychiatry. 2015;78(6):403-412.
3. Wakade CG, Mahadik SP, Waller JL, et al. Atypical neuroleptics stimulate neurogenesis in adult rat brain. J Neurosci Res. 2002;69(1):72-79.
4. Lieberman JA, Tollefson GD, Charles C, et al; HGDH Study Group. Antipsychotic drug effects on brain morphology in first-episode psychosis. Arch Gen Psychiatry. 2005;62(4):361-370.
5. Nasrallah HA. Impaired neuroplasticity in schizophrenia and the neuro-regenerative effects of atypical antipsychotics. Medscape Psychiatry. http://www.medscape.org/viewarticle/569521. Published January 31, 2008. Accessed November 10, 2015.
6. Nasrallah HA. Haloperidol clearly is neurotoxic. Should it be banned? Current Psychiatry. 2012;12(7):7-8.
7. Nandra KS, Agius M. The differences between typical and atypical antipsychotics: the effects on neurogenesis. Psychiatr Danub. 2012;24(suppl 1):S95-S99.
8. Nasrallah HA, Hopkins T, Pixley SK, et al. Differential effects of antipsychotic and antidepressant drugs on neurogenic region in rats. Brain Res. 2010;354:23-29.
9. Pillai A, Tery AV, Mahadik SP. Differential effects of long-term treatment with typical and atypical antipsychotics on NGF and BNDF levels in rat striatum and hippocampus. Schizophr Res. 2006;82(1):95-106.
10. Buckley PF, Pillai A, Evans D, et al. Brain derived neurotropic factor in first-episode psychosis. Schizophr Res. 2007;91(1-3):1-5.
11. Martin MV, Mimics K, Nisenbaum LK, et al. Olanzapine reversed brain gene expression changes induced by phencyclidines treatment in non-human primates. Mol Neuropsychiatry. 2015;1(2):82-93.
12. Yan BC, Park JH, Ahn JH, et al. Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res. 2014;92(6):795-807.
13. Yulug B, Yildiz A, Güzel O, et al. Risperidone attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;69(6):656-659.
14. Yulug B, Yildiz A, Hüdaoglu O, et al. Olanzapine attenuates brain damage after focal cerebral ischemia in vivo. Brain Res Bull. 2006;71(1-3):296-300.
15. Elsworth JD, Morrow BA. Hajszan T, et al. Phencyclidine-induced loss of asymmetric spine synapses in rodent prefrontal cortex is reversed by acute and chronic treatment with olanzapine. Neuropsychopharmacology. 2001;36(10):2054-2061.
16. Seki Y, Kato TA, Monji A, et al. Pretreatment of aripiprazole and minocycline, but not haloperidol, suppresses oligodendrocyte damage from interferon-y-stimulated microglia in co-culture model. Schizophr Res. 2013;151(1-3):20-28.
17. Bian Q, Kato T, Monji A, et al. The effect of atypical antipsychotics, perospirone, ziprasidone and quetiapine on microglial activation induced by interferon-gamma. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(1):42-48.
18. Wang HD, Deutch AY. Dopamine depletion of the prefrontal cortex induces dendritic spine loss: reversal by atypical antipsychotic drug treatment. Neuropsychopharmacology. 2008;33(6):1276-1286.
19. Miller BJ, Buckley P, Seabolt W, et al. Meta-analysis of cytokine alternations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663-671.
20. Nasrallah HA. Beyond dopamine: The ‘other’ effects of antipsychotics. Current Psychiatry. 2013;12(6):8-9.
21. Yang MC, Lung FW. Neuroprotection of paliperidone on SH-SY5Y cells against β-amyloid peptide(25-35), N-methyl-4-phenylpyridinium ion, and hydrogen peroxide-induced cell death. Psychopharmacology (Berl). 2011;217(3):397-410.
22. Peng L, Zhu D, Feng X, et al. Paliperidone protects prefrontal cortical neurons from damages caused by MK-801 via Akt1/GSK3β _signaling pathway. Schizophr Res. 2013;147(1):14-23.23.
Stojkovic´ T, Radonjic´ NV, Velimirovic´ M, et al. Risperidone reverses phencyclidine induced decrease in glutathione levels and alternations of antioxidant defense in rat brain. Prog Neuropsychopharmacol Biol Psychiatry. 2012;39(1):192-199.
24. Koprivica V, Regardie K, Wolff C, et al. Aripiprazole protects cortical neurons from glutamate toxicity. Eur J Pharmacol. 2011;651(1-3):73-76.
25. Vaidya VA, Marek GJ, Aghajanian GK, et al. 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J Neurosci. 1997;17(8):2785-2795.
26. Meridith GE, Switzer RC 3rd, Napier TC. Short-term, D2 receptor blockade induces synaptic degeneration, reduces levels of tyrosine hydroxylase and brain-derived neurotrophic factor, and enhances D2-mediated firing in the ventral pallidum. Brain Res. 2004;995(1):14-22.
27. Lieberman JA, Perkins DO, Jarskog LF. Neuroprotection: a therapeutic strategy to prevent deterioration associated with schizophrenia. CNS Spectr. 2007;12(suppl 4):1-13; quiz 14.
Balancing clinical and supportive care at every step of the disease continuum
It seems it was just yesterday that we did our first “mutation analysis” to help guide us in our treatment of patients with a drug that was more likely to work than not. Of course, I am referring to estrogen-receptor/progesterone receptor (ER/PR) blocking therapy, and “yesterday” actually goes all the way back to the 1970s! When tamoxifen was first given to unselected metastatic breast cancer patients, the response rate was low, but when the study population was “enriched” with breast cancer patients who were ER/ PR-positive, the response rates improved and the outcomes were more favorable. So began the era of tumor markers and enriching patient populations, and the process now referred to as mutation analysis, which is becoming more broadly applicable to other tumors as well.
Click on the PDF icon at the top of this introduction to read the full article.
It seems it was just yesterday that we did our first “mutation analysis” to help guide us in our treatment of patients with a drug that was more likely to work than not. Of course, I am referring to estrogen-receptor/progesterone receptor (ER/PR) blocking therapy, and “yesterday” actually goes all the way back to the 1970s! When tamoxifen was first given to unselected metastatic breast cancer patients, the response rate was low, but when the study population was “enriched” with breast cancer patients who were ER/ PR-positive, the response rates improved and the outcomes were more favorable. So began the era of tumor markers and enriching patient populations, and the process now referred to as mutation analysis, which is becoming more broadly applicable to other tumors as well.
Click on the PDF icon at the top of this introduction to read the full article.
It seems it was just yesterday that we did our first “mutation analysis” to help guide us in our treatment of patients with a drug that was more likely to work than not. Of course, I am referring to estrogen-receptor/progesterone receptor (ER/PR) blocking therapy, and “yesterday” actually goes all the way back to the 1970s! When tamoxifen was first given to unselected metastatic breast cancer patients, the response rate was low, but when the study population was “enriched” with breast cancer patients who were ER/ PR-positive, the response rates improved and the outcomes were more favorable. So began the era of tumor markers and enriching patient populations, and the process now referred to as mutation analysis, which is becoming more broadly applicable to other tumors as well.
Click on the PDF icon at the top of this introduction to read the full article.
To err is human, but…
In Being Wrong,1 her treatise on the psychology of human error, Kathryn Schulz quotes William James: “Our errors are surely not such awfully solemn things.”2 Being wrong, she argues, is part of the human genome. Despite aphorisms such as “we learn from our mistakes,” we are far from accepting of mistakes in medical practice. Perhaps naively, I do not believe that our need to understand how clinical errors occur and how to avoid them is based on the fear of legal repercussion. And of course we do not want to harm our patients. But our relationship with medical errors is far more complex than that. We really don’t want to be wrong.
Dr. Atul Gawande3 has promoted using checklists and a structured system to limit errors of misapplication of knowledge. Diagnostic and therapeutic algorithms, once the province of trauma surgeons, are increasingly becoming part of internal medicine.
When I was a house officer we all had our “pocket brains” in our white coats—lists of disease complications, drug doses and interactions, causes of IgA deposition in the kidney, and treatment algorithms. But we believed (probably correctly) that our teachers expected us to commit all these facts to memory in our fleshy brains. The elitist and hubristic belief that this was uniformly possible has lingered in academic medicine, still permeating even the fabric of certification examinations. We learn that it is OK to be honest and say that we don’t know the answer, but we don’t like to have to say it. Physicians finish the academic game of Chutes and Ladders with a strong aversion to being wrong.
Younger doctors today seem more comfortable with not knowing so many facts and bits of medical trivia, being able to find answers instantly using their smart phones. But a challenge is knowing at a glance the context and veracity of the answers you find. And whether the knowledge comes from our anatomic, pocket, or cyber brain, the overarching challenge is to avoid Gawande’s error of misapplication.
In this issue of the Journal, Dr. Nikhil Mull and colleagues dissect a clinical case that did not proceed as expected. They discuss, in reference to the described patient, some of the published analyses of the clinical decision-making process, highlighting various ways that our reasoning can be led astray. Having just finished a stint on the inpatient consultation service, I wish I could have read the article a few weeks ago. A bit of reflection on how we reach decisions can be as powerful as knowing the source of the facts in our pocket brain.
Being wrong, as Schulz has written, is part of the human experience, but I don’t like it. Ways to limit the chances of it’s happening in the clinic are worth keeping on a personal checklist, or perhaps as an app on my smart phone.
- Schulz K. Being Wrong: Adventures in the Margin of Error. New York: Harper Collins, 2010.
- James W. The will to believe. An address to the philosophical clubs of Yale and Brown Universities, 1896. http://educ.jmu.edu//~omearawm/ph101willtobelieve.html. Accessed October 12, 2015.
- Gawande A. The Checklist Manifesto. How to Get Things Right. New York: Metropolitan Books, 2009.
In Being Wrong,1 her treatise on the psychology of human error, Kathryn Schulz quotes William James: “Our errors are surely not such awfully solemn things.”2 Being wrong, she argues, is part of the human genome. Despite aphorisms such as “we learn from our mistakes,” we are far from accepting of mistakes in medical practice. Perhaps naively, I do not believe that our need to understand how clinical errors occur and how to avoid them is based on the fear of legal repercussion. And of course we do not want to harm our patients. But our relationship with medical errors is far more complex than that. We really don’t want to be wrong.
Dr. Atul Gawande3 has promoted using checklists and a structured system to limit errors of misapplication of knowledge. Diagnostic and therapeutic algorithms, once the province of trauma surgeons, are increasingly becoming part of internal medicine.
When I was a house officer we all had our “pocket brains” in our white coats—lists of disease complications, drug doses and interactions, causes of IgA deposition in the kidney, and treatment algorithms. But we believed (probably correctly) that our teachers expected us to commit all these facts to memory in our fleshy brains. The elitist and hubristic belief that this was uniformly possible has lingered in academic medicine, still permeating even the fabric of certification examinations. We learn that it is OK to be honest and say that we don’t know the answer, but we don’t like to have to say it. Physicians finish the academic game of Chutes and Ladders with a strong aversion to being wrong.
Younger doctors today seem more comfortable with not knowing so many facts and bits of medical trivia, being able to find answers instantly using their smart phones. But a challenge is knowing at a glance the context and veracity of the answers you find. And whether the knowledge comes from our anatomic, pocket, or cyber brain, the overarching challenge is to avoid Gawande’s error of misapplication.
In this issue of the Journal, Dr. Nikhil Mull and colleagues dissect a clinical case that did not proceed as expected. They discuss, in reference to the described patient, some of the published analyses of the clinical decision-making process, highlighting various ways that our reasoning can be led astray. Having just finished a stint on the inpatient consultation service, I wish I could have read the article a few weeks ago. A bit of reflection on how we reach decisions can be as powerful as knowing the source of the facts in our pocket brain.
Being wrong, as Schulz has written, is part of the human experience, but I don’t like it. Ways to limit the chances of it’s happening in the clinic are worth keeping on a personal checklist, or perhaps as an app on my smart phone.
In Being Wrong,1 her treatise on the psychology of human error, Kathryn Schulz quotes William James: “Our errors are surely not such awfully solemn things.”2 Being wrong, she argues, is part of the human genome. Despite aphorisms such as “we learn from our mistakes,” we are far from accepting of mistakes in medical practice. Perhaps naively, I do not believe that our need to understand how clinical errors occur and how to avoid them is based on the fear of legal repercussion. And of course we do not want to harm our patients. But our relationship with medical errors is far more complex than that. We really don’t want to be wrong.
Dr. Atul Gawande3 has promoted using checklists and a structured system to limit errors of misapplication of knowledge. Diagnostic and therapeutic algorithms, once the province of trauma surgeons, are increasingly becoming part of internal medicine.
When I was a house officer we all had our “pocket brains” in our white coats—lists of disease complications, drug doses and interactions, causes of IgA deposition in the kidney, and treatment algorithms. But we believed (probably correctly) that our teachers expected us to commit all these facts to memory in our fleshy brains. The elitist and hubristic belief that this was uniformly possible has lingered in academic medicine, still permeating even the fabric of certification examinations. We learn that it is OK to be honest and say that we don’t know the answer, but we don’t like to have to say it. Physicians finish the academic game of Chutes and Ladders with a strong aversion to being wrong.
Younger doctors today seem more comfortable with not knowing so many facts and bits of medical trivia, being able to find answers instantly using their smart phones. But a challenge is knowing at a glance the context and veracity of the answers you find. And whether the knowledge comes from our anatomic, pocket, or cyber brain, the overarching challenge is to avoid Gawande’s error of misapplication.
In this issue of the Journal, Dr. Nikhil Mull and colleagues dissect a clinical case that did not proceed as expected. They discuss, in reference to the described patient, some of the published analyses of the clinical decision-making process, highlighting various ways that our reasoning can be led astray. Having just finished a stint on the inpatient consultation service, I wish I could have read the article a few weeks ago. A bit of reflection on how we reach decisions can be as powerful as knowing the source of the facts in our pocket brain.
Being wrong, as Schulz has written, is part of the human experience, but I don’t like it. Ways to limit the chances of it’s happening in the clinic are worth keeping on a personal checklist, or perhaps as an app on my smart phone.
- Schulz K. Being Wrong: Adventures in the Margin of Error. New York: Harper Collins, 2010.
- James W. The will to believe. An address to the philosophical clubs of Yale and Brown Universities, 1896. http://educ.jmu.edu//~omearawm/ph101willtobelieve.html. Accessed October 12, 2015.
- Gawande A. The Checklist Manifesto. How to Get Things Right. New York: Metropolitan Books, 2009.
- Schulz K. Being Wrong: Adventures in the Margin of Error. New York: Harper Collins, 2010.
- James W. The will to believe. An address to the philosophical clubs of Yale and Brown Universities, 1896. http://educ.jmu.edu//~omearawm/ph101willtobelieve.html. Accessed October 12, 2015.
- Gawande A. The Checklist Manifesto. How to Get Things Right. New York: Metropolitan Books, 2009.
Can we reduce the use of abdominal hysterectomy and increase the use of vaginal and laparoscopic approaches?
Hysterectomy for benign disease is a very effective operation to treat moderate to severe uterine bleeding or pain caused by uterine problems. There are 3 main surgical approaches to performing a hysterectomy: vaginal, laparoscopic, and abdominal.
“Abdominal hysterectomy” is a term that indicates the procedure was performed using a relatively large incision in the abdominal wall. It also is possible for 2 surgical routes to be combined into one operation, such as a laparoscopically assisted vaginal hysterectomy or a laparoscopic hysterectomy with a mini-laparotomy incision to remove the uterus.
Substantial evidence indicates that vaginal and laparoscopic approaches to hysterectomy result in superior outcomes when compared with abdominal hysterectomy. In this editorial I highlight that data, as well as offer concrete ways in which we can increase the use of vaginal and laparoscopic hysterectomy while reducing the current reliance on an abdominal approach.
Vaginal and laparoscopic hysterectomy are associated with more rapid recoveryAuthors of a meta-analysis of 47 randomized trials involving 5,102 women concluded that women who underwent vaginal and laparoscopic hysterectomy had faster return to full activity, compared with women who had an abdominal hysterectomy. Compared with vaginal hysterectomy, the abdominal approach required an additional 12 days of postoperative recovery before return to normal activities and 1 additional day of postoperative hospitalization.1
In the same meta-analysis, when compared with the laparoscopic approach, abdominal hysterectomy required 15 additional days to return to normal activity and 2.6 more days of postoperative hospitalization.1 The evidence indicates that to maximize rapid return of the patient to full activity, we should reduce the use of abdominal hysterectomy for benign disease.
Abdominal hysterectomy is the most frequent US surgical approach In the United States in 2010, the rates of hysterectomy by route were 56% abdominal, 25% laparoscopic, and 19% vaginal.2 In contrast to US practice, French, German, and Australian gynecologists prioritize the vaginal route. In France in 2004, the rates of hysterectomy by route were 48% vaginal, 27% laparoscopic, and 25% abdominal.3 In Australia and Germany, vaginal hysterectomy is performed in 39% and 55% of all hysterectomy cases, respectively—a greater rate of vaginal hysterectomy than observed in the United States (TABLE 1).4,5
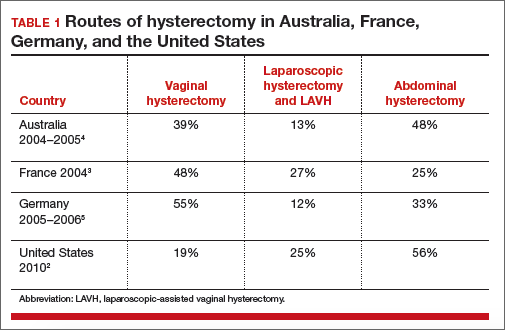
Our goal should be 40% or less for abdominal hysterectomy. Based on the experience of French,3 Australian,4 and German5 surgeons, a realistic goal is to reduce the use of abdominal hysterectomy in the United States to a rate of 40% or less and to increase the use of vaginal and laparoscopic hysterectomy to a combined rate of 60% or more.
Perceived contraindications for vaginal hysterectomy may not be valid
Surgeons may avoid selecting a vaginal route for hysterectomy for benign uterine disease when the patient has a markedly enlarged uterus (for example, >16 weeks’ size) or a markedly enlarged cervix or lower uterine segment. The large uterus may be difficult to remove through the vagina and an enlarged cervix or lower uterine segment may make it difficult to enter the peritoneal cavity.
However, large uteri can be removed through the vagina using uterine reduction techniques, including uterine bisection and intramyometrial coring. In one randomized clinical trial,1 women with enlarged uteri were randomly assigned to vaginal or abdominal hysterectomy. Both approaches were successful in removing large uteri. When compared with abdominal hysterectomy, the vaginal approach was associated with shorter operative time, less postoperative fever, less postoperative pain, and fewer hospital days following surgery.
Reference
How will we increase vaginal and laparoscopic hysterectomy for benign disease?In order to reduce the use of abdominal hysterectomy, a multipronged effort is needed:
- Leaders in gynecology need to champion the use of vaginal and laparoscopic hysterectomy.
- Educators in gynecology need to refocus and intensify surgical training to ensure that trainees are confident in their ability to perform both vaginal and laparoscopic hysterectomy.
- Hospital departments need to provide the continuing education and senior surgical mentoring that will facilitate reducing the use of abdominal hysterectomy.
- Quality review committees need to review the indication for abdominal hysterectomy procedures and question whether they could be better performed by a vaginal or laparoscopic route.
AAGL launches comprehensive video cooperative. A major new educational video offering on vaginal hysterectomy recently was released by the AAGL (TABLE 2). Produced by the AAGL and cosponsored by the American College of Obstetricians and Gynecologists and the Society of Gynecologic Surgeons, this resource includes detailed videos focused on basic instrumentation and technique, techniques for adnexal surgery at vaginal hysterectomy, and managing complications. I believe this resource will be of great value as momentum builds to increase the use of vaginal hysterectomy. In addition, OBG Management will continue to publish major articles by leading surgeons focused on vaginal hysterectomy.

Are you a champion of vaginal and laparoscopic hysterectomy? Every hospital should identify champions of these approaches. These master surgeons could help advance the capability of the hospital staff to confidently and safely prioritize the use of vaginal and laparoscopic hysterectomy for benign disease by mentoring other surgeons. If we reduce the use of abdominal hysterectomy we will improve outcomes and significantly advance women’s health.
Select OBG Management publications on vaginal surgery and minimally invasive gynecology
Transforming vaginal hysterectomy: 7 solutions to the most
daunting challenges
Rosanne M. Kho, MD (July 2014)
The Extracorporeal C-Incision Tissue Extraction ExCITE technique
Mireille D. Truong, MD, and Arnold P. Advincula, MD (November 2014)
Update on vaginal hysterectomy
Barbara S. Levy, MD (September 2015)
The ExCITE technique, Part 2: Simulation made simple
Mireille D. Truong, MD, and Arnold P. Advincula, MD (Coming soon)
The following articles are based on the master class in vaginal hysterectomy produced by AAGL and cosponsored by ACOG and SGS.
Vaginal hysterectomy with basic instrumentation
Barbara S. Levy, MD (October 2015)
Technique for salpingectomy and salpingo-oophorectomy
John B. Gebhart, MD, MS (In this issue, page 26)
Managing complications in vaginal hysterectomy
John B. Gebhart, MD, MS (Coming soon)
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Aarts JW, Nieboer TE, Johnson N, et al. Surgical approach to hysterectomy for benign gynaecological disease. Cochrane Database Syst Rev. 2015;(8):CD003677. doi:10.1002/14651858.CD003677.pub5.
- Cohen SL, Vitonis AF, Einarsson JI. Updated hysterectomy surveillance and factors associated with minimally invasive hysterectomy. JSLS. 2014;18(3):e2014.00096.
- David-Montefiore E, Rouzier R, Chapron C, Darai E; Collegiale d’Obstétrique et Gynécologie de Paris-Ile de France. Surgical routes and complications of hysterectomy for benign disorders: a prospective observational study in French university hospitals. Hum Reprod. 2007;22(1):260–265.
- Hill E, Graham M, Shelley J. Hysterectomy trends in Australia—between 2000/01 and 2004/05. Aust N Z J Obstet Gynaecol. 2010;50(2):153–158.
- Stang A, Merrill RM, Kuss O. Nationwide rates of conversion from laparoscopic or vaginal hysterectomy to open abdominal hysterectomy in Germany. Eur J Epidemiol. 2011;26(2):125–133.
Hysterectomy for benign disease is a very effective operation to treat moderate to severe uterine bleeding or pain caused by uterine problems. There are 3 main surgical approaches to performing a hysterectomy: vaginal, laparoscopic, and abdominal.
“Abdominal hysterectomy” is a term that indicates the procedure was performed using a relatively large incision in the abdominal wall. It also is possible for 2 surgical routes to be combined into one operation, such as a laparoscopically assisted vaginal hysterectomy or a laparoscopic hysterectomy with a mini-laparotomy incision to remove the uterus.
Substantial evidence indicates that vaginal and laparoscopic approaches to hysterectomy result in superior outcomes when compared with abdominal hysterectomy. In this editorial I highlight that data, as well as offer concrete ways in which we can increase the use of vaginal and laparoscopic hysterectomy while reducing the current reliance on an abdominal approach.
Vaginal and laparoscopic hysterectomy are associated with more rapid recoveryAuthors of a meta-analysis of 47 randomized trials involving 5,102 women concluded that women who underwent vaginal and laparoscopic hysterectomy had faster return to full activity, compared with women who had an abdominal hysterectomy. Compared with vaginal hysterectomy, the abdominal approach required an additional 12 days of postoperative recovery before return to normal activities and 1 additional day of postoperative hospitalization.1
In the same meta-analysis, when compared with the laparoscopic approach, abdominal hysterectomy required 15 additional days to return to normal activity and 2.6 more days of postoperative hospitalization.1 The evidence indicates that to maximize rapid return of the patient to full activity, we should reduce the use of abdominal hysterectomy for benign disease.
Abdominal hysterectomy is the most frequent US surgical approach In the United States in 2010, the rates of hysterectomy by route were 56% abdominal, 25% laparoscopic, and 19% vaginal.2 In contrast to US practice, French, German, and Australian gynecologists prioritize the vaginal route. In France in 2004, the rates of hysterectomy by route were 48% vaginal, 27% laparoscopic, and 25% abdominal.3 In Australia and Germany, vaginal hysterectomy is performed in 39% and 55% of all hysterectomy cases, respectively—a greater rate of vaginal hysterectomy than observed in the United States (TABLE 1).4,5

Our goal should be 40% or less for abdominal hysterectomy. Based on the experience of French,3 Australian,4 and German5 surgeons, a realistic goal is to reduce the use of abdominal hysterectomy in the United States to a rate of 40% or less and to increase the use of vaginal and laparoscopic hysterectomy to a combined rate of 60% or more.
Perceived contraindications for vaginal hysterectomy may not be valid
Surgeons may avoid selecting a vaginal route for hysterectomy for benign uterine disease when the patient has a markedly enlarged uterus (for example, >16 weeks’ size) or a markedly enlarged cervix or lower uterine segment. The large uterus may be difficult to remove through the vagina and an enlarged cervix or lower uterine segment may make it difficult to enter the peritoneal cavity.
However, large uteri can be removed through the vagina using uterine reduction techniques, including uterine bisection and intramyometrial coring. In one randomized clinical trial,1 women with enlarged uteri were randomly assigned to vaginal or abdominal hysterectomy. Both approaches were successful in removing large uteri. When compared with abdominal hysterectomy, the vaginal approach was associated with shorter operative time, less postoperative fever, less postoperative pain, and fewer hospital days following surgery.
Reference
How will we increase vaginal and laparoscopic hysterectomy for benign disease?In order to reduce the use of abdominal hysterectomy, a multipronged effort is needed:
- Leaders in gynecology need to champion the use of vaginal and laparoscopic hysterectomy.
- Educators in gynecology need to refocus and intensify surgical training to ensure that trainees are confident in their ability to perform both vaginal and laparoscopic hysterectomy.
- Hospital departments need to provide the continuing education and senior surgical mentoring that will facilitate reducing the use of abdominal hysterectomy.
- Quality review committees need to review the indication for abdominal hysterectomy procedures and question whether they could be better performed by a vaginal or laparoscopic route.
AAGL launches comprehensive video cooperative. A major new educational video offering on vaginal hysterectomy recently was released by the AAGL (TABLE 2). Produced by the AAGL and cosponsored by the American College of Obstetricians and Gynecologists and the Society of Gynecologic Surgeons, this resource includes detailed videos focused on basic instrumentation and technique, techniques for adnexal surgery at vaginal hysterectomy, and managing complications. I believe this resource will be of great value as momentum builds to increase the use of vaginal hysterectomy. In addition, OBG Management will continue to publish major articles by leading surgeons focused on vaginal hysterectomy.

Are you a champion of vaginal and laparoscopic hysterectomy? Every hospital should identify champions of these approaches. These master surgeons could help advance the capability of the hospital staff to confidently and safely prioritize the use of vaginal and laparoscopic hysterectomy for benign disease by mentoring other surgeons. If we reduce the use of abdominal hysterectomy we will improve outcomes and significantly advance women’s health.
Select OBG Management publications on vaginal surgery and minimally invasive gynecology
Transforming vaginal hysterectomy: 7 solutions to the most
daunting challenges
Rosanne M. Kho, MD (July 2014)
The Extracorporeal C-Incision Tissue Extraction ExCITE technique
Mireille D. Truong, MD, and Arnold P. Advincula, MD (November 2014)
Update on vaginal hysterectomy
Barbara S. Levy, MD (September 2015)
The ExCITE technique, Part 2: Simulation made simple
Mireille D. Truong, MD, and Arnold P. Advincula, MD (Coming soon)
The following articles are based on the master class in vaginal hysterectomy produced by AAGL and cosponsored by ACOG and SGS.
Vaginal hysterectomy with basic instrumentation
Barbara S. Levy, MD (October 2015)
Technique for salpingectomy and salpingo-oophorectomy
John B. Gebhart, MD, MS (In this issue, page 26)
Managing complications in vaginal hysterectomy
John B. Gebhart, MD, MS (Coming soon)
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Hysterectomy for benign disease is a very effective operation to treat moderate to severe uterine bleeding or pain caused by uterine problems. There are 3 main surgical approaches to performing a hysterectomy: vaginal, laparoscopic, and abdominal.
“Abdominal hysterectomy” is a term that indicates the procedure was performed using a relatively large incision in the abdominal wall. It also is possible for 2 surgical routes to be combined into one operation, such as a laparoscopically assisted vaginal hysterectomy or a laparoscopic hysterectomy with a mini-laparotomy incision to remove the uterus.
Substantial evidence indicates that vaginal and laparoscopic approaches to hysterectomy result in superior outcomes when compared with abdominal hysterectomy. In this editorial I highlight that data, as well as offer concrete ways in which we can increase the use of vaginal and laparoscopic hysterectomy while reducing the current reliance on an abdominal approach.
Vaginal and laparoscopic hysterectomy are associated with more rapid recoveryAuthors of a meta-analysis of 47 randomized trials involving 5,102 women concluded that women who underwent vaginal and laparoscopic hysterectomy had faster return to full activity, compared with women who had an abdominal hysterectomy. Compared with vaginal hysterectomy, the abdominal approach required an additional 12 days of postoperative recovery before return to normal activities and 1 additional day of postoperative hospitalization.1
In the same meta-analysis, when compared with the laparoscopic approach, abdominal hysterectomy required 15 additional days to return to normal activity and 2.6 more days of postoperative hospitalization.1 The evidence indicates that to maximize rapid return of the patient to full activity, we should reduce the use of abdominal hysterectomy for benign disease.
Abdominal hysterectomy is the most frequent US surgical approach In the United States in 2010, the rates of hysterectomy by route were 56% abdominal, 25% laparoscopic, and 19% vaginal.2 In contrast to US practice, French, German, and Australian gynecologists prioritize the vaginal route. In France in 2004, the rates of hysterectomy by route were 48% vaginal, 27% laparoscopic, and 25% abdominal.3 In Australia and Germany, vaginal hysterectomy is performed in 39% and 55% of all hysterectomy cases, respectively—a greater rate of vaginal hysterectomy than observed in the United States (TABLE 1).4,5

Our goal should be 40% or less for abdominal hysterectomy. Based on the experience of French,3 Australian,4 and German5 surgeons, a realistic goal is to reduce the use of abdominal hysterectomy in the United States to a rate of 40% or less and to increase the use of vaginal and laparoscopic hysterectomy to a combined rate of 60% or more.
Perceived contraindications for vaginal hysterectomy may not be valid
Surgeons may avoid selecting a vaginal route for hysterectomy for benign uterine disease when the patient has a markedly enlarged uterus (for example, >16 weeks’ size) or a markedly enlarged cervix or lower uterine segment. The large uterus may be difficult to remove through the vagina and an enlarged cervix or lower uterine segment may make it difficult to enter the peritoneal cavity.
However, large uteri can be removed through the vagina using uterine reduction techniques, including uterine bisection and intramyometrial coring. In one randomized clinical trial,1 women with enlarged uteri were randomly assigned to vaginal or abdominal hysterectomy. Both approaches were successful in removing large uteri. When compared with abdominal hysterectomy, the vaginal approach was associated with shorter operative time, less postoperative fever, less postoperative pain, and fewer hospital days following surgery.
Reference
How will we increase vaginal and laparoscopic hysterectomy for benign disease?In order to reduce the use of abdominal hysterectomy, a multipronged effort is needed:
- Leaders in gynecology need to champion the use of vaginal and laparoscopic hysterectomy.
- Educators in gynecology need to refocus and intensify surgical training to ensure that trainees are confident in their ability to perform both vaginal and laparoscopic hysterectomy.
- Hospital departments need to provide the continuing education and senior surgical mentoring that will facilitate reducing the use of abdominal hysterectomy.
- Quality review committees need to review the indication for abdominal hysterectomy procedures and question whether they could be better performed by a vaginal or laparoscopic route.
AAGL launches comprehensive video cooperative. A major new educational video offering on vaginal hysterectomy recently was released by the AAGL (TABLE 2). Produced by the AAGL and cosponsored by the American College of Obstetricians and Gynecologists and the Society of Gynecologic Surgeons, this resource includes detailed videos focused on basic instrumentation and technique, techniques for adnexal surgery at vaginal hysterectomy, and managing complications. I believe this resource will be of great value as momentum builds to increase the use of vaginal hysterectomy. In addition, OBG Management will continue to publish major articles by leading surgeons focused on vaginal hysterectomy.

Are you a champion of vaginal and laparoscopic hysterectomy? Every hospital should identify champions of these approaches. These master surgeons could help advance the capability of the hospital staff to confidently and safely prioritize the use of vaginal and laparoscopic hysterectomy for benign disease by mentoring other surgeons. If we reduce the use of abdominal hysterectomy we will improve outcomes and significantly advance women’s health.
Select OBG Management publications on vaginal surgery and minimally invasive gynecology
Transforming vaginal hysterectomy: 7 solutions to the most
daunting challenges
Rosanne M. Kho, MD (July 2014)
The Extracorporeal C-Incision Tissue Extraction ExCITE technique
Mireille D. Truong, MD, and Arnold P. Advincula, MD (November 2014)
Update on vaginal hysterectomy
Barbara S. Levy, MD (September 2015)
The ExCITE technique, Part 2: Simulation made simple
Mireille D. Truong, MD, and Arnold P. Advincula, MD (Coming soon)
The following articles are based on the master class in vaginal hysterectomy produced by AAGL and cosponsored by ACOG and SGS.
Vaginal hysterectomy with basic instrumentation
Barbara S. Levy, MD (October 2015)
Technique for salpingectomy and salpingo-oophorectomy
John B. Gebhart, MD, MS (In this issue, page 26)
Managing complications in vaginal hysterectomy
John B. Gebhart, MD, MS (Coming soon)
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Aarts JW, Nieboer TE, Johnson N, et al. Surgical approach to hysterectomy for benign gynaecological disease. Cochrane Database Syst Rev. 2015;(8):CD003677. doi:10.1002/14651858.CD003677.pub5.
- Cohen SL, Vitonis AF, Einarsson JI. Updated hysterectomy surveillance and factors associated with minimally invasive hysterectomy. JSLS. 2014;18(3):e2014.00096.
- David-Montefiore E, Rouzier R, Chapron C, Darai E; Collegiale d’Obstétrique et Gynécologie de Paris-Ile de France. Surgical routes and complications of hysterectomy for benign disorders: a prospective observational study in French university hospitals. Hum Reprod. 2007;22(1):260–265.
- Hill E, Graham M, Shelley J. Hysterectomy trends in Australia—between 2000/01 and 2004/05. Aust N Z J Obstet Gynaecol. 2010;50(2):153–158.
- Stang A, Merrill RM, Kuss O. Nationwide rates of conversion from laparoscopic or vaginal hysterectomy to open abdominal hysterectomy in Germany. Eur J Epidemiol. 2011;26(2):125–133.
- Aarts JW, Nieboer TE, Johnson N, et al. Surgical approach to hysterectomy for benign gynaecological disease. Cochrane Database Syst Rev. 2015;(8):CD003677. doi:10.1002/14651858.CD003677.pub5.
- Cohen SL, Vitonis AF, Einarsson JI. Updated hysterectomy surveillance and factors associated with minimally invasive hysterectomy. JSLS. 2014;18(3):e2014.00096.
- David-Montefiore E, Rouzier R, Chapron C, Darai E; Collegiale d’Obstétrique et Gynécologie de Paris-Ile de France. Surgical routes and complications of hysterectomy for benign disorders: a prospective observational study in French university hospitals. Hum Reprod. 2007;22(1):260–265.
- Hill E, Graham M, Shelley J. Hysterectomy trends in Australia—between 2000/01 and 2004/05. Aust N Z J Obstet Gynaecol. 2010;50(2):153–158.
- Stang A, Merrill RM, Kuss O. Nationwide rates of conversion from laparoscopic or vaginal hysterectomy to open abdominal hysterectomy in Germany. Eur J Epidemiol. 2011;26(2):125–133.