User login
The Power of Quiet
In his insightful book “The Wisdom of Crowds” (New York: Anchor Books, 2004), James Surowiecke makes the convincing argument that many heads are wiser than one, even if that one is the sole expert regarding the subject under discussion. As long as the decision-making group is diverse, with each individual being allowed to come to an independent conclusion, this tenet appears to hold, whether the group is estimating the number of jelly beans in a jar or resolving a difficult issue. The message is clear: As a leader your leadership will be more effective if you solicit input from all members of your group, including those who may be reluctant to offer it.
In another excellent book, “Quiet” (New York: Crown Publishers, 2012), Susan Cain posits that, from early in the 20th century on, despite the considerable value it has to offer, introversion has become a “second-class personality trait.” Although highly valued earlier in our history, the thoughtful, introspective temperament was replaced by the aggressive, decisive character as the ideal.
Cain delves deeply into the substantial differences between extroverts and introverts, acknowledging that there are many gradations between the extremes. Extroverts tend to be loquacious and are seldom hesitant to offer their opinions on complex, difficult issues, even when their understanding of them is limited. They don’t always think before speaking and are less skilled listeners than introverts. They prefer to come to decisions rapidly, sometimes with incomplete data, and are much more decisive than introverts.
Introverts, on the other hand, prefer to listen rather than talk and to thoroughly vet an issue before reaching a decision. When they do, they are uncomfortable expressing it in a group setting. They prefer to work alone rather than in groups and, because of their thoughtful approach, their solutions to problems may be more innovative and sound than the shoot-from-the-hip, rapid answers that extroverts frequently propose. They abhor conflict and are likely to remain silent during controversy. In sum, although more difficult to elicit, obtaining input from the quiet members of the group is very worthwhile.
Often the most timely and ideal resolution is reached by balanced contributions from both personality types, the decision-making extroverts and the more thoughtful but reticent introverts. In fact, some of the best team members are those who are not on either extreme of the extrovert-introvert scale. But considering the fact that one-third to one-half of Americans are introverts (I suspect the fraction is a bit less among surgeons) and hesitant to offer their opinions in a group setting, how is this to be accomplished?
First, as a leader, you need to be sensitive to the fact that the introverts in your group are likely out of their comfort zone during communal meetings. It may even be embarrassing for them if they are called upon to offer their advice or opinion. To some degree this reluctance can be overcome by a leader who always attempts to reach consensus by valuing everyone’s opinion. Even the arrangement of the meeting room is important. The ideal is for all participants to be situated around a table rather than facing an imposing leader at the front of the room. This “leveling of the play field” emphasizes equality, de-emphasizes hierarchy, and encourages all to participate. The least likely to contribute can often be nudged from their quiet solitude by gentle urging from the leader with a statement such as: “Joe, I know you have a thoughtful perspective on this. Can you share it with the group?”
However, even the best-run meeting may not result in satisfactory resolution of difficult issues. In my experience, even those toward the extrovert end of the spectrum may be hesitant to offer their honest opinion in a meeting if it is in conflict with that of the leader. It is not uncommon to come to a consensus resolution of a controversial issue in a group meeting only to find out from hallway chatter that many disagree with the agreement reached. It is essential that the leader have access to this hallway chatter. This can be accomplished by way of confidantes who have the trust of both the troops and the leader.
During my years of leadership, a useful and productive technique I fostered to prompt input from introverts and honest assessments from all was to visit individual offices after the busy work day had quieted down, usually after 5 p.m. Meeting with individual faculty in their offices rather than in mine lent an informality to the conversation that could not be duplicated in the office of the chairman. In these one-on-one encounters, I found that even my relatively quiet faculty members felt comfortable in expressing their views regarding controversial issues facing our department. These informal chats also allowed me to become aware of problems they were facing in their professional and personal lives. They were great opportunities for mentoring and bonding as well. When these individual discussions precede what is anticipated to be a contentious group meeting, the likelihood of a successful conclusion is significantly enhanced.
Although my leadership experience was confined within the walls of academe, I believe these principles apply to anyone invited to lead a group in virtually any setting. Individual meetings are not an efficient way to lead, but they may provide a more effective and, in some cases, more rapid means of reaching consensus than innumerable group meetings with follow-up emails. When the group is too large to conference with everyone individually, one-on-one meetings with several key players may achieve the same result. During the process, don’t forget the quiet ones. They sometimes contribute the best and most innovative solutions to complex problems. There is power in quiet.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
In his insightful book “The Wisdom of Crowds” (New York: Anchor Books, 2004), James Surowiecke makes the convincing argument that many heads are wiser than one, even if that one is the sole expert regarding the subject under discussion. As long as the decision-making group is diverse, with each individual being allowed to come to an independent conclusion, this tenet appears to hold, whether the group is estimating the number of jelly beans in a jar or resolving a difficult issue. The message is clear: As a leader your leadership will be more effective if you solicit input from all members of your group, including those who may be reluctant to offer it.
In another excellent book, “Quiet” (New York: Crown Publishers, 2012), Susan Cain posits that, from early in the 20th century on, despite the considerable value it has to offer, introversion has become a “second-class personality trait.” Although highly valued earlier in our history, the thoughtful, introspective temperament was replaced by the aggressive, decisive character as the ideal.
Cain delves deeply into the substantial differences between extroverts and introverts, acknowledging that there are many gradations between the extremes. Extroverts tend to be loquacious and are seldom hesitant to offer their opinions on complex, difficult issues, even when their understanding of them is limited. They don’t always think before speaking and are less skilled listeners than introverts. They prefer to come to decisions rapidly, sometimes with incomplete data, and are much more decisive than introverts.
Introverts, on the other hand, prefer to listen rather than talk and to thoroughly vet an issue before reaching a decision. When they do, they are uncomfortable expressing it in a group setting. They prefer to work alone rather than in groups and, because of their thoughtful approach, their solutions to problems may be more innovative and sound than the shoot-from-the-hip, rapid answers that extroverts frequently propose. They abhor conflict and are likely to remain silent during controversy. In sum, although more difficult to elicit, obtaining input from the quiet members of the group is very worthwhile.
Often the most timely and ideal resolution is reached by balanced contributions from both personality types, the decision-making extroverts and the more thoughtful but reticent introverts. In fact, some of the best team members are those who are not on either extreme of the extrovert-introvert scale. But considering the fact that one-third to one-half of Americans are introverts (I suspect the fraction is a bit less among surgeons) and hesitant to offer their opinions in a group setting, how is this to be accomplished?
First, as a leader, you need to be sensitive to the fact that the introverts in your group are likely out of their comfort zone during communal meetings. It may even be embarrassing for them if they are called upon to offer their advice or opinion. To some degree this reluctance can be overcome by a leader who always attempts to reach consensus by valuing everyone’s opinion. Even the arrangement of the meeting room is important. The ideal is for all participants to be situated around a table rather than facing an imposing leader at the front of the room. This “leveling of the play field” emphasizes equality, de-emphasizes hierarchy, and encourages all to participate. The least likely to contribute can often be nudged from their quiet solitude by gentle urging from the leader with a statement such as: “Joe, I know you have a thoughtful perspective on this. Can you share it with the group?”
However, even the best-run meeting may not result in satisfactory resolution of difficult issues. In my experience, even those toward the extrovert end of the spectrum may be hesitant to offer their honest opinion in a meeting if it is in conflict with that of the leader. It is not uncommon to come to a consensus resolution of a controversial issue in a group meeting only to find out from hallway chatter that many disagree with the agreement reached. It is essential that the leader have access to this hallway chatter. This can be accomplished by way of confidantes who have the trust of both the troops and the leader.
During my years of leadership, a useful and productive technique I fostered to prompt input from introverts and honest assessments from all was to visit individual offices after the busy work day had quieted down, usually after 5 p.m. Meeting with individual faculty in their offices rather than in mine lent an informality to the conversation that could not be duplicated in the office of the chairman. In these one-on-one encounters, I found that even my relatively quiet faculty members felt comfortable in expressing their views regarding controversial issues facing our department. These informal chats also allowed me to become aware of problems they were facing in their professional and personal lives. They were great opportunities for mentoring and bonding as well. When these individual discussions precede what is anticipated to be a contentious group meeting, the likelihood of a successful conclusion is significantly enhanced.
Although my leadership experience was confined within the walls of academe, I believe these principles apply to anyone invited to lead a group in virtually any setting. Individual meetings are not an efficient way to lead, but they may provide a more effective and, in some cases, more rapid means of reaching consensus than innumerable group meetings with follow-up emails. When the group is too large to conference with everyone individually, one-on-one meetings with several key players may achieve the same result. During the process, don’t forget the quiet ones. They sometimes contribute the best and most innovative solutions to complex problems. There is power in quiet.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
In his insightful book “The Wisdom of Crowds” (New York: Anchor Books, 2004), James Surowiecke makes the convincing argument that many heads are wiser than one, even if that one is the sole expert regarding the subject under discussion. As long as the decision-making group is diverse, with each individual being allowed to come to an independent conclusion, this tenet appears to hold, whether the group is estimating the number of jelly beans in a jar or resolving a difficult issue. The message is clear: As a leader your leadership will be more effective if you solicit input from all members of your group, including those who may be reluctant to offer it.
In another excellent book, “Quiet” (New York: Crown Publishers, 2012), Susan Cain posits that, from early in the 20th century on, despite the considerable value it has to offer, introversion has become a “second-class personality trait.” Although highly valued earlier in our history, the thoughtful, introspective temperament was replaced by the aggressive, decisive character as the ideal.
Cain delves deeply into the substantial differences between extroverts and introverts, acknowledging that there are many gradations between the extremes. Extroverts tend to be loquacious and are seldom hesitant to offer their opinions on complex, difficult issues, even when their understanding of them is limited. They don’t always think before speaking and are less skilled listeners than introverts. They prefer to come to decisions rapidly, sometimes with incomplete data, and are much more decisive than introverts.
Introverts, on the other hand, prefer to listen rather than talk and to thoroughly vet an issue before reaching a decision. When they do, they are uncomfortable expressing it in a group setting. They prefer to work alone rather than in groups and, because of their thoughtful approach, their solutions to problems may be more innovative and sound than the shoot-from-the-hip, rapid answers that extroverts frequently propose. They abhor conflict and are likely to remain silent during controversy. In sum, although more difficult to elicit, obtaining input from the quiet members of the group is very worthwhile.
Often the most timely and ideal resolution is reached by balanced contributions from both personality types, the decision-making extroverts and the more thoughtful but reticent introverts. In fact, some of the best team members are those who are not on either extreme of the extrovert-introvert scale. But considering the fact that one-third to one-half of Americans are introverts (I suspect the fraction is a bit less among surgeons) and hesitant to offer their opinions in a group setting, how is this to be accomplished?
First, as a leader, you need to be sensitive to the fact that the introverts in your group are likely out of their comfort zone during communal meetings. It may even be embarrassing for them if they are called upon to offer their advice or opinion. To some degree this reluctance can be overcome by a leader who always attempts to reach consensus by valuing everyone’s opinion. Even the arrangement of the meeting room is important. The ideal is for all participants to be situated around a table rather than facing an imposing leader at the front of the room. This “leveling of the play field” emphasizes equality, de-emphasizes hierarchy, and encourages all to participate. The least likely to contribute can often be nudged from their quiet solitude by gentle urging from the leader with a statement such as: “Joe, I know you have a thoughtful perspective on this. Can you share it with the group?”
However, even the best-run meeting may not result in satisfactory resolution of difficult issues. In my experience, even those toward the extrovert end of the spectrum may be hesitant to offer their honest opinion in a meeting if it is in conflict with that of the leader. It is not uncommon to come to a consensus resolution of a controversial issue in a group meeting only to find out from hallway chatter that many disagree with the agreement reached. It is essential that the leader have access to this hallway chatter. This can be accomplished by way of confidantes who have the trust of both the troops and the leader.
During my years of leadership, a useful and productive technique I fostered to prompt input from introverts and honest assessments from all was to visit individual offices after the busy work day had quieted down, usually after 5 p.m. Meeting with individual faculty in their offices rather than in mine lent an informality to the conversation that could not be duplicated in the office of the chairman. In these one-on-one encounters, I found that even my relatively quiet faculty members felt comfortable in expressing their views regarding controversial issues facing our department. These informal chats also allowed me to become aware of problems they were facing in their professional and personal lives. They were great opportunities for mentoring and bonding as well. When these individual discussions precede what is anticipated to be a contentious group meeting, the likelihood of a successful conclusion is significantly enhanced.
Although my leadership experience was confined within the walls of academe, I believe these principles apply to anyone invited to lead a group in virtually any setting. Individual meetings are not an efficient way to lead, but they may provide a more effective and, in some cases, more rapid means of reaching consensus than innumerable group meetings with follow-up emails. When the group is too large to conference with everyone individually, one-on-one meetings with several key players may achieve the same result. During the process, don’t forget the quiet ones. They sometimes contribute the best and most innovative solutions to complex problems. There is power in quiet.
Dr. Rikkers is Editor in Chief of ACS Surgery News.
Intractable shoulder dystocia: A posterior axilla maneuver may save the day
Shoulder dystocia is an unpredictable obstetric emergency that challenges all obstetricians and midwives. In response to a shoulder dystocia emergency, most clinicians implement a sequence of well-practiced steps that begin with early recognition of the problem, clear communication of the emergency with delivery room staff, and a call for help to available clinicians. Management steps may include:
- instructing the mother to stop pushing and moving the mother's buttocks to the edge of the bed
- ensuring there is not a tight nuchal cord
- committing to avoiding the use of excessive force on the fetal head and neck
- considering performing an episiotomy
- performing the McRoberts maneuver combined with suprapubic pressure
- using a rotational maneuver, such as the Woods maneuver or the Rubin maneuver
- delivering the posterior arm
- considering the Gaskin all-four maneuver.
When initial management steps are not enoughIf this sequence of steps does not result in successful vaginal delivery, additional options include: clavicle fracture, cephalic replacement followed by cesarean delivery (Zavanelli maneuver), symphysiotomy, or fundal pressure combined with a rotational maneuver. Another simple intervention that is not discussed widely in medical textbooks or taught during training is the posterior axilla maneuver.
Posterior axilla maneuversVarying posterior axilla maneuvers have been described by many expert obstetricians, including Willughby (17th Century),1 Holman (1963),2 Schramm (1983),3 Menticoglou (2006),4 and Hofmeyr and Cluver (2009, 2015).5−7
Willughby maneuverPercival Willughby’s (1596−1685) description of a posterior axilla maneuver was brief1:
After the head is born, if the child through the greatness of the shoulders, should stick at the neck, let the midwife put her fingers under the child's armpit and give it a nudge, thrusting it to the other side with her finger, drawing the child or she may quickly bring forth the shoulders, without offering to put it forth by her hands clasped about the neck, which might endanger the breaking of the neck.
Holman maneuverHolman described a maneuver with the following steps2:
- perform an episiotomy
- place a finger in the posterior axilla and draw the posterior shoulder down along the pelvic axis
- simultaneously have an assistant perform suprapubic pressure and
- if necessary, insert two supinated fingers under the pubic arch and press and rock the anterior shoulder, tilting the anterior shoulder toward the hollow of the sacrum while simultaneously gently pulling the posterior axilla along the pelvic axis.
Schramm maneuverSchramm, working with a population enriched with women with diabetes, frequently encountered shoulder dystocia and recommended3:
If the posterior axilla can be reached—in other words, if the posterior shoulder is engaged—in my experience it can always be delivered by rotating it to the anterior position while at the same time applying traction....I normally place 1 or 2 fingers of my right hand in the posterior axilla and “scruff” the neck with my left hand, applying both rotation and traction. Because this grip is somewhat insecure, the resultant tractive force is limited and I consider this manoeuvre to be the most effective and least traumatic method of relieving moderate to severe obstruction.
Practice your shoulder dystocia maneuvers using simulation
Obstetric emergencies trigger a rush of adrenaline and great stress for the obstetrician and delivery room team. This may adversely impact motor performance, decision making, and communication skills.1 Low- and high-fidelity simulation exercises create an environment in which the obstetrics team can practice the sequence of maneuvers and seamless teamwork needed to successfully resolve a shoulder dystocia.2,3 Implementing a shoulder dystocia protocol and practicing the protocol using team-based simulation may help to reduce the adverse outcomes of shoulder dystocia.3,4
Reference
1. Wetzel CM, Kneebone RL, Woloshynowych M, et al. The effects of stress on surgical performance. Am J Surg. 2006;191(1):5−10.
2. Crofts JF, Fox R, Ellis D, Winter C, Hinshaw K, Draycott TJ. Observations from 450 shoulder dystocia simulations. Obstet Gynecol. 2008;112(4):906−912.
3. Draycott TJ, Crofts JF, Ash JP, et al. Improving neonatal outcome through practical shoulder dystocia training. Obstet Gynecol. 2008;112(1):14−20.
4. Grobman WA, Miller D, Burke C, Hornbogen A, Tam K, Costello R. Outcomes associated with introduction of a shoulder dystocia protocol. Am J Obstet Gynecol. 2011;205(6):513−517.
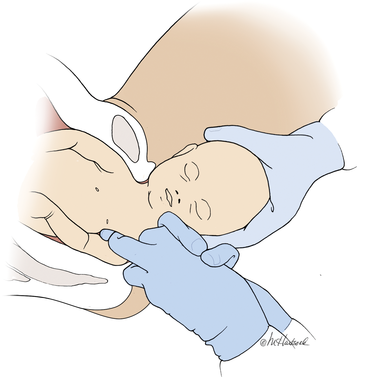
Manipulation of the posterior axilla |
|
The right and left third fingers are locked into the posterior axilla, one finger from the front and one from the back of the fetus. Gentle downward guidance is provided by the fingers to draw the posterior shoulder down and out along the curve of the sacrum, thus releasing the anterior shoulder.4 In this drawing, an assistant gently holds the head up. |
Menticoglou maneuverMenticoglou noted that delivery of the posterior arm generally resolves almost all cases of shoulder dystocia. However, if the posterior arm is extended and trapped between the fetus and maternal pelvic side-wall, it may be difficult to deliver the posterior arm. In these cases he recommended having an assistant gently hold, not pull, the fetal head upward and, at the same time, having the obstetrician get on one knee, placing the middle fingers of both hands into the posterior axilla of the fetus.4
The right middle finger is placed into the axilla from the left side of the maternal pelvis, and the left middle finger is placed into the axilla from the right side of the maternal pelvis, resulting in the two middle fingers overlapping in the fetal axilla (FIGURE).4 Gentle force is then used to pull the posterior shoulder and arm downward and outward along the curve of the sacrum. Once the shoulder has emerged from the pelvis, the posterior arm is delivered. Alternatively, if the posterior shoulder is brought well down into the pelvis, another attempt can be made at delivering the posterior arm.4
My preferred approach. The Menticoglou maneuver is my preferred posterior axilla maneuver because it can be accomplished rapidly; requires no equipment, such as a sling catheter; and the obstetrician has good tactile feedback throughout the application of gentle force.
Hofmeyr-Cluver maneuverIn cases of difficult shoulder dystocia, Dr. William Smellie (1762)8 recommended placing one or two fingers in the anterior or posterior fetal axilla and gentling pulling on the axilla to deliver the body. If the axillae were too high to reach, he recommended using a blunt hook in the axilla to draw forth the impacted child. He advised caution when using a blunt hook because the fetus might be injured or lacerated.
Instead of using a hook, Hofmeyr and Cluver5−7 have recommended using a catheter sling to deliver the posterior shoulder. In this maneuver, a loop of a suction catheter or firm urinary catheter is placed over the obstetrician’s index finger and the loop is pushed through the posterior axilla, back to front, with guidance from the index finger. The index finger of the opposite hand is used to catch the loop and pull the catheter through, creating a single-stranded sling that is positioned in the axilla. Gentle force is then applied to the sling in the axis of the pelvis to deliver the posterior shoulder.
“If the posterior arm does not follow it is then swept out easily because room has been created by delivering the posterior shoulder. If the aforementioned procedure fails, the sling can be used to rotate the shoulder. To perform a rotational maneuver, sling traction is directed laterally towards the side of the baby’s back then anteriorly while digital pressure is applied behind the anterior shoulder to assist rotation.”7
Use ACOG’s checklist for documenting a shoulder dystocia
Following the resolution of a shoulder dystocia, it is important to gather all the necessary facts to complete a detailed medical record entry describing the situation and interventions used. The checklist from the American College of Obstetricians and Gynecologists (ACOG) helps you to prepare a standardized medical record entry that is comprehensive.
My experience is that “free form” medical record entries describing the events at a shoulder dystocia event are generally not optimally organized, creating future problems when the case is reviewed.
ACOG obstetric checklists are available for download at http://www.acog.org/Resources-And-Publications, or use your web browser to search for “ACOG Shoulder Dystocia checklist.”
With scant literature, know the benefits and risksThe world’s literature on posterior axilla maneuvers to resolve shoulder dystocia consists of case series and individual case reports.2−7 Hence, the quality of the data supporting this intervention is not optimal, and risks associated with the maneuver are not well characterized. Application of a controlled and gentle force to the posterior axilla may cause fracture of the fetal humerus5 or dislocation of the fetal shoulder. The posterior axilla maneuver also may increase the risk of a maternal third- or fourth-degree perineal laceration.
As a general rule, as the number of maneuvers used to resolve a difficult shoulder dystocia increase, the risk of neonatal injury increases.9 Since the posterior axilla maneuver typically is only attempted after multiple previous maneuvers have failed, the risk of fetal injury is increased. However, as time passes and a shoulder dystocia remains unresolved for 4 or 5 minutes, the risk of neurologic injury and fetal death increases.10
In resolving a shoulder dystocia, speed and skill are essential. A posterior axilla maneuver can be performed more rapidly than a Zavanelli maneuver or a symphysiotomy. Although manipulation of the posterior axilla and arm may cause a fracture of the humerus, this complication is a modest price to pay for preventing permanent fetal brain injury or fetal death.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Willughby P. Observations in midwifery. New York, NY: MW Books; 1972:312−313.
- Holman MS. A new manoeuvre for delivery of an impacted shoulder based on a mechanical analysis. S Afr Med J. 1963;37:247−249.
- Schramm M. Impacted shoulders—a personal experience. Aust N Z J Obstet Gynaecol. 1983;23(1):28−31.
- Menticoglou SM. A modified technique to deliver the posterior arm in severe shoulder dystocia. Obstet Gynecol. 2006;108(3 pt 2):755−757.
- Cluver CA, Hofmeyr GJ. Posterior axilla sling traction: a technique for intractable shoulder dystocia. Obstet Gynecol. 2009;113(2 pt 2):486–488.
- Hofmeyr GJ, Cluver CA. Posterior axilla sling traction for intractable shoulder dystocia. BJOG. 2009;116(13):1818−1820.
- Cluver CA, Hofmeyr GJ. Posterior axilla sling traction for shoulder dystocia: case review and a new method for shoulder rotation with the sling. Am J Obstet Gynecol. 2015;212(6):784.e1−e7.
- Smellie W. A treatise on the theory and practice of midwifery. 4th ed. London, England; 1762:226−227.
- Hoffman MK, Bailit JL, Branch DW, et al; Consortium on Safe Labor. A comparison of obstetric maneuvers for the acute management of shoulder dystocia. Obstet Gynecol. 2011;117(6):1272−1278.
- Lerner H, Durlacher K, Smith S, Hamilton E. Relationship between head-to-body delivery interval in shoulder dystocia and neonatal depression. Obstet Gynecol. 2011;118(2 pt 1):318−322.
Shoulder dystocia is an unpredictable obstetric emergency that challenges all obstetricians and midwives. In response to a shoulder dystocia emergency, most clinicians implement a sequence of well-practiced steps that begin with early recognition of the problem, clear communication of the emergency with delivery room staff, and a call for help to available clinicians. Management steps may include:
- instructing the mother to stop pushing and moving the mother's buttocks to the edge of the bed
- ensuring there is not a tight nuchal cord
- committing to avoiding the use of excessive force on the fetal head and neck
- considering performing an episiotomy
- performing the McRoberts maneuver combined with suprapubic pressure
- using a rotational maneuver, such as the Woods maneuver or the Rubin maneuver
- delivering the posterior arm
- considering the Gaskin all-four maneuver.
When initial management steps are not enoughIf this sequence of steps does not result in successful vaginal delivery, additional options include: clavicle fracture, cephalic replacement followed by cesarean delivery (Zavanelli maneuver), symphysiotomy, or fundal pressure combined with a rotational maneuver. Another simple intervention that is not discussed widely in medical textbooks or taught during training is the posterior axilla maneuver.
Posterior axilla maneuversVarying posterior axilla maneuvers have been described by many expert obstetricians, including Willughby (17th Century),1 Holman (1963),2 Schramm (1983),3 Menticoglou (2006),4 and Hofmeyr and Cluver (2009, 2015).5−7
Willughby maneuverPercival Willughby’s (1596−1685) description of a posterior axilla maneuver was brief1:
After the head is born, if the child through the greatness of the shoulders, should stick at the neck, let the midwife put her fingers under the child's armpit and give it a nudge, thrusting it to the other side with her finger, drawing the child or she may quickly bring forth the shoulders, without offering to put it forth by her hands clasped about the neck, which might endanger the breaking of the neck.
Holman maneuverHolman described a maneuver with the following steps2:
- perform an episiotomy
- place a finger in the posterior axilla and draw the posterior shoulder down along the pelvic axis
- simultaneously have an assistant perform suprapubic pressure and
- if necessary, insert two supinated fingers under the pubic arch and press and rock the anterior shoulder, tilting the anterior shoulder toward the hollow of the sacrum while simultaneously gently pulling the posterior axilla along the pelvic axis.
Schramm maneuverSchramm, working with a population enriched with women with diabetes, frequently encountered shoulder dystocia and recommended3:
If the posterior axilla can be reached—in other words, if the posterior shoulder is engaged—in my experience it can always be delivered by rotating it to the anterior position while at the same time applying traction....I normally place 1 or 2 fingers of my right hand in the posterior axilla and “scruff” the neck with my left hand, applying both rotation and traction. Because this grip is somewhat insecure, the resultant tractive force is limited and I consider this manoeuvre to be the most effective and least traumatic method of relieving moderate to severe obstruction.
Practice your shoulder dystocia maneuvers using simulation
Obstetric emergencies trigger a rush of adrenaline and great stress for the obstetrician and delivery room team. This may adversely impact motor performance, decision making, and communication skills.1 Low- and high-fidelity simulation exercises create an environment in which the obstetrics team can practice the sequence of maneuvers and seamless teamwork needed to successfully resolve a shoulder dystocia.2,3 Implementing a shoulder dystocia protocol and practicing the protocol using team-based simulation may help to reduce the adverse outcomes of shoulder dystocia.3,4
Reference
1. Wetzel CM, Kneebone RL, Woloshynowych M, et al. The effects of stress on surgical performance. Am J Surg. 2006;191(1):5−10.
2. Crofts JF, Fox R, Ellis D, Winter C, Hinshaw K, Draycott TJ. Observations from 450 shoulder dystocia simulations. Obstet Gynecol. 2008;112(4):906−912.
3. Draycott TJ, Crofts JF, Ash JP, et al. Improving neonatal outcome through practical shoulder dystocia training. Obstet Gynecol. 2008;112(1):14−20.
4. Grobman WA, Miller D, Burke C, Hornbogen A, Tam K, Costello R. Outcomes associated with introduction of a shoulder dystocia protocol. Am J Obstet Gynecol. 2011;205(6):513−517.
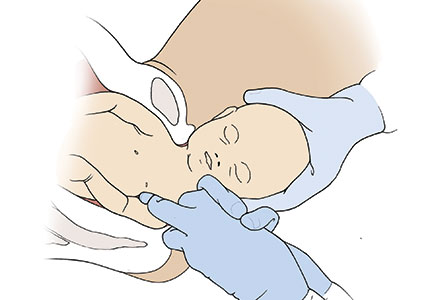
Manipulation of the posterior axilla |
|
The right and left third fingers are locked into the posterior axilla, one finger from the front and one from the back of the fetus. Gentle downward guidance is provided by the fingers to draw the posterior shoulder down and out along the curve of the sacrum, thus releasing the anterior shoulder.4 In this drawing, an assistant gently holds the head up. |
Menticoglou maneuverMenticoglou noted that delivery of the posterior arm generally resolves almost all cases of shoulder dystocia. However, if the posterior arm is extended and trapped between the fetus and maternal pelvic side-wall, it may be difficult to deliver the posterior arm. In these cases he recommended having an assistant gently hold, not pull, the fetal head upward and, at the same time, having the obstetrician get on one knee, placing the middle fingers of both hands into the posterior axilla of the fetus.4
The right middle finger is placed into the axilla from the left side of the maternal pelvis, and the left middle finger is placed into the axilla from the right side of the maternal pelvis, resulting in the two middle fingers overlapping in the fetal axilla (FIGURE).4 Gentle force is then used to pull the posterior shoulder and arm downward and outward along the curve of the sacrum. Once the shoulder has emerged from the pelvis, the posterior arm is delivered. Alternatively, if the posterior shoulder is brought well down into the pelvis, another attempt can be made at delivering the posterior arm.4
My preferred approach. The Menticoglou maneuver is my preferred posterior axilla maneuver because it can be accomplished rapidly; requires no equipment, such as a sling catheter; and the obstetrician has good tactile feedback throughout the application of gentle force.
Hofmeyr-Cluver maneuverIn cases of difficult shoulder dystocia, Dr. William Smellie (1762)8 recommended placing one or two fingers in the anterior or posterior fetal axilla and gentling pulling on the axilla to deliver the body. If the axillae were too high to reach, he recommended using a blunt hook in the axilla to draw forth the impacted child. He advised caution when using a blunt hook because the fetus might be injured or lacerated.
Instead of using a hook, Hofmeyr and Cluver5−7 have recommended using a catheter sling to deliver the posterior shoulder. In this maneuver, a loop of a suction catheter or firm urinary catheter is placed over the obstetrician’s index finger and the loop is pushed through the posterior axilla, back to front, with guidance from the index finger. The index finger of the opposite hand is used to catch the loop and pull the catheter through, creating a single-stranded sling that is positioned in the axilla. Gentle force is then applied to the sling in the axis of the pelvis to deliver the posterior shoulder.
“If the posterior arm does not follow it is then swept out easily because room has been created by delivering the posterior shoulder. If the aforementioned procedure fails, the sling can be used to rotate the shoulder. To perform a rotational maneuver, sling traction is directed laterally towards the side of the baby’s back then anteriorly while digital pressure is applied behind the anterior shoulder to assist rotation.”7
Use ACOG’s checklist for documenting a shoulder dystocia
Following the resolution of a shoulder dystocia, it is important to gather all the necessary facts to complete a detailed medical record entry describing the situation and interventions used. The checklist from the American College of Obstetricians and Gynecologists (ACOG) helps you to prepare a standardized medical record entry that is comprehensive.
My experience is that “free form” medical record entries describing the events at a shoulder dystocia event are generally not optimally organized, creating future problems when the case is reviewed.
ACOG obstetric checklists are available for download at http://www.acog.org/Resources-And-Publications, or use your web browser to search for “ACOG Shoulder Dystocia checklist.”
With scant literature, know the benefits and risksThe world’s literature on posterior axilla maneuvers to resolve shoulder dystocia consists of case series and individual case reports.2−7 Hence, the quality of the data supporting this intervention is not optimal, and risks associated with the maneuver are not well characterized. Application of a controlled and gentle force to the posterior axilla may cause fracture of the fetal humerus5 or dislocation of the fetal shoulder. The posterior axilla maneuver also may increase the risk of a maternal third- or fourth-degree perineal laceration.
As a general rule, as the number of maneuvers used to resolve a difficult shoulder dystocia increase, the risk of neonatal injury increases.9 Since the posterior axilla maneuver typically is only attempted after multiple previous maneuvers have failed, the risk of fetal injury is increased. However, as time passes and a shoulder dystocia remains unresolved for 4 or 5 minutes, the risk of neurologic injury and fetal death increases.10
In resolving a shoulder dystocia, speed and skill are essential. A posterior axilla maneuver can be performed more rapidly than a Zavanelli maneuver or a symphysiotomy. Although manipulation of the posterior axilla and arm may cause a fracture of the humerus, this complication is a modest price to pay for preventing permanent fetal brain injury or fetal death.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Shoulder dystocia is an unpredictable obstetric emergency that challenges all obstetricians and midwives. In response to a shoulder dystocia emergency, most clinicians implement a sequence of well-practiced steps that begin with early recognition of the problem, clear communication of the emergency with delivery room staff, and a call for help to available clinicians. Management steps may include:
- instructing the mother to stop pushing and moving the mother's buttocks to the edge of the bed
- ensuring there is not a tight nuchal cord
- committing to avoiding the use of excessive force on the fetal head and neck
- considering performing an episiotomy
- performing the McRoberts maneuver combined with suprapubic pressure
- using a rotational maneuver, such as the Woods maneuver or the Rubin maneuver
- delivering the posterior arm
- considering the Gaskin all-four maneuver.
When initial management steps are not enoughIf this sequence of steps does not result in successful vaginal delivery, additional options include: clavicle fracture, cephalic replacement followed by cesarean delivery (Zavanelli maneuver), symphysiotomy, or fundal pressure combined with a rotational maneuver. Another simple intervention that is not discussed widely in medical textbooks or taught during training is the posterior axilla maneuver.
Posterior axilla maneuversVarying posterior axilla maneuvers have been described by many expert obstetricians, including Willughby (17th Century),1 Holman (1963),2 Schramm (1983),3 Menticoglou (2006),4 and Hofmeyr and Cluver (2009, 2015).5−7
Willughby maneuverPercival Willughby’s (1596−1685) description of a posterior axilla maneuver was brief1:
After the head is born, if the child through the greatness of the shoulders, should stick at the neck, let the midwife put her fingers under the child's armpit and give it a nudge, thrusting it to the other side with her finger, drawing the child or she may quickly bring forth the shoulders, without offering to put it forth by her hands clasped about the neck, which might endanger the breaking of the neck.
Holman maneuverHolman described a maneuver with the following steps2:
- perform an episiotomy
- place a finger in the posterior axilla and draw the posterior shoulder down along the pelvic axis
- simultaneously have an assistant perform suprapubic pressure and
- if necessary, insert two supinated fingers under the pubic arch and press and rock the anterior shoulder, tilting the anterior shoulder toward the hollow of the sacrum while simultaneously gently pulling the posterior axilla along the pelvic axis.
Schramm maneuverSchramm, working with a population enriched with women with diabetes, frequently encountered shoulder dystocia and recommended3:
If the posterior axilla can be reached—in other words, if the posterior shoulder is engaged—in my experience it can always be delivered by rotating it to the anterior position while at the same time applying traction....I normally place 1 or 2 fingers of my right hand in the posterior axilla and “scruff” the neck with my left hand, applying both rotation and traction. Because this grip is somewhat insecure, the resultant tractive force is limited and I consider this manoeuvre to be the most effective and least traumatic method of relieving moderate to severe obstruction.
Practice your shoulder dystocia maneuvers using simulation
Obstetric emergencies trigger a rush of adrenaline and great stress for the obstetrician and delivery room team. This may adversely impact motor performance, decision making, and communication skills.1 Low- and high-fidelity simulation exercises create an environment in which the obstetrics team can practice the sequence of maneuvers and seamless teamwork needed to successfully resolve a shoulder dystocia.2,3 Implementing a shoulder dystocia protocol and practicing the protocol using team-based simulation may help to reduce the adverse outcomes of shoulder dystocia.3,4
Reference
1. Wetzel CM, Kneebone RL, Woloshynowych M, et al. The effects of stress on surgical performance. Am J Surg. 2006;191(1):5−10.
2. Crofts JF, Fox R, Ellis D, Winter C, Hinshaw K, Draycott TJ. Observations from 450 shoulder dystocia simulations. Obstet Gynecol. 2008;112(4):906−912.
3. Draycott TJ, Crofts JF, Ash JP, et al. Improving neonatal outcome through practical shoulder dystocia training. Obstet Gynecol. 2008;112(1):14−20.
4. Grobman WA, Miller D, Burke C, Hornbogen A, Tam K, Costello R. Outcomes associated with introduction of a shoulder dystocia protocol. Am J Obstet Gynecol. 2011;205(6):513−517.
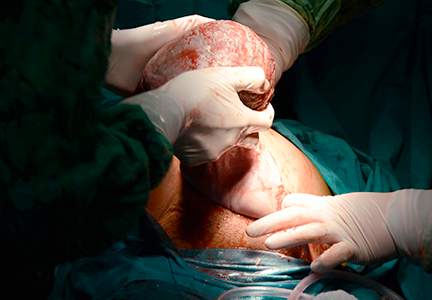
Manipulation of the posterior axilla |
|
The right and left third fingers are locked into the posterior axilla, one finger from the front and one from the back of the fetus. Gentle downward guidance is provided by the fingers to draw the posterior shoulder down and out along the curve of the sacrum, thus releasing the anterior shoulder.4 In this drawing, an assistant gently holds the head up. |
Menticoglou maneuverMenticoglou noted that delivery of the posterior arm generally resolves almost all cases of shoulder dystocia. However, if the posterior arm is extended and trapped between the fetus and maternal pelvic side-wall, it may be difficult to deliver the posterior arm. In these cases he recommended having an assistant gently hold, not pull, the fetal head upward and, at the same time, having the obstetrician get on one knee, placing the middle fingers of both hands into the posterior axilla of the fetus.4
The right middle finger is placed into the axilla from the left side of the maternal pelvis, and the left middle finger is placed into the axilla from the right side of the maternal pelvis, resulting in the two middle fingers overlapping in the fetal axilla (FIGURE).4 Gentle force is then used to pull the posterior shoulder and arm downward and outward along the curve of the sacrum. Once the shoulder has emerged from the pelvis, the posterior arm is delivered. Alternatively, if the posterior shoulder is brought well down into the pelvis, another attempt can be made at delivering the posterior arm.4
My preferred approach. The Menticoglou maneuver is my preferred posterior axilla maneuver because it can be accomplished rapidly; requires no equipment, such as a sling catheter; and the obstetrician has good tactile feedback throughout the application of gentle force.
Hofmeyr-Cluver maneuverIn cases of difficult shoulder dystocia, Dr. William Smellie (1762)8 recommended placing one or two fingers in the anterior or posterior fetal axilla and gentling pulling on the axilla to deliver the body. If the axillae were too high to reach, he recommended using a blunt hook in the axilla to draw forth the impacted child. He advised caution when using a blunt hook because the fetus might be injured or lacerated.
Instead of using a hook, Hofmeyr and Cluver5−7 have recommended using a catheter sling to deliver the posterior shoulder. In this maneuver, a loop of a suction catheter or firm urinary catheter is placed over the obstetrician’s index finger and the loop is pushed through the posterior axilla, back to front, with guidance from the index finger. The index finger of the opposite hand is used to catch the loop and pull the catheter through, creating a single-stranded sling that is positioned in the axilla. Gentle force is then applied to the sling in the axis of the pelvis to deliver the posterior shoulder.
“If the posterior arm does not follow it is then swept out easily because room has been created by delivering the posterior shoulder. If the aforementioned procedure fails, the sling can be used to rotate the shoulder. To perform a rotational maneuver, sling traction is directed laterally towards the side of the baby’s back then anteriorly while digital pressure is applied behind the anterior shoulder to assist rotation.”7
Use ACOG’s checklist for documenting a shoulder dystocia
Following the resolution of a shoulder dystocia, it is important to gather all the necessary facts to complete a detailed medical record entry describing the situation and interventions used. The checklist from the American College of Obstetricians and Gynecologists (ACOG) helps you to prepare a standardized medical record entry that is comprehensive.
My experience is that “free form” medical record entries describing the events at a shoulder dystocia event are generally not optimally organized, creating future problems when the case is reviewed.
ACOG obstetric checklists are available for download at http://www.acog.org/Resources-And-Publications, or use your web browser to search for “ACOG Shoulder Dystocia checklist.”
With scant literature, know the benefits and risksThe world’s literature on posterior axilla maneuvers to resolve shoulder dystocia consists of case series and individual case reports.2−7 Hence, the quality of the data supporting this intervention is not optimal, and risks associated with the maneuver are not well characterized. Application of a controlled and gentle force to the posterior axilla may cause fracture of the fetal humerus5 or dislocation of the fetal shoulder. The posterior axilla maneuver also may increase the risk of a maternal third- or fourth-degree perineal laceration.
As a general rule, as the number of maneuvers used to resolve a difficult shoulder dystocia increase, the risk of neonatal injury increases.9 Since the posterior axilla maneuver typically is only attempted after multiple previous maneuvers have failed, the risk of fetal injury is increased. However, as time passes and a shoulder dystocia remains unresolved for 4 or 5 minutes, the risk of neurologic injury and fetal death increases.10
In resolving a shoulder dystocia, speed and skill are essential. A posterior axilla maneuver can be performed more rapidly than a Zavanelli maneuver or a symphysiotomy. Although manipulation of the posterior axilla and arm may cause a fracture of the humerus, this complication is a modest price to pay for preventing permanent fetal brain injury or fetal death.
Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Willughby P. Observations in midwifery. New York, NY: MW Books; 1972:312−313.
- Holman MS. A new manoeuvre for delivery of an impacted shoulder based on a mechanical analysis. S Afr Med J. 1963;37:247−249.
- Schramm M. Impacted shoulders—a personal experience. Aust N Z J Obstet Gynaecol. 1983;23(1):28−31.
- Menticoglou SM. A modified technique to deliver the posterior arm in severe shoulder dystocia. Obstet Gynecol. 2006;108(3 pt 2):755−757.
- Cluver CA, Hofmeyr GJ. Posterior axilla sling traction: a technique for intractable shoulder dystocia. Obstet Gynecol. 2009;113(2 pt 2):486–488.
- Hofmeyr GJ, Cluver CA. Posterior axilla sling traction for intractable shoulder dystocia. BJOG. 2009;116(13):1818−1820.
- Cluver CA, Hofmeyr GJ. Posterior axilla sling traction for shoulder dystocia: case review and a new method for shoulder rotation with the sling. Am J Obstet Gynecol. 2015;212(6):784.e1−e7.
- Smellie W. A treatise on the theory and practice of midwifery. 4th ed. London, England; 1762:226−227.
- Hoffman MK, Bailit JL, Branch DW, et al; Consortium on Safe Labor. A comparison of obstetric maneuvers for the acute management of shoulder dystocia. Obstet Gynecol. 2011;117(6):1272−1278.
- Lerner H, Durlacher K, Smith S, Hamilton E. Relationship between head-to-body delivery interval in shoulder dystocia and neonatal depression. Obstet Gynecol. 2011;118(2 pt 1):318−322.
- Willughby P. Observations in midwifery. New York, NY: MW Books; 1972:312−313.
- Holman MS. A new manoeuvre for delivery of an impacted shoulder based on a mechanical analysis. S Afr Med J. 1963;37:247−249.
- Schramm M. Impacted shoulders—a personal experience. Aust N Z J Obstet Gynaecol. 1983;23(1):28−31.
- Menticoglou SM. A modified technique to deliver the posterior arm in severe shoulder dystocia. Obstet Gynecol. 2006;108(3 pt 2):755−757.
- Cluver CA, Hofmeyr GJ. Posterior axilla sling traction: a technique for intractable shoulder dystocia. Obstet Gynecol. 2009;113(2 pt 2):486–488.
- Hofmeyr GJ, Cluver CA. Posterior axilla sling traction for intractable shoulder dystocia. BJOG. 2009;116(13):1818−1820.
- Cluver CA, Hofmeyr GJ. Posterior axilla sling traction for shoulder dystocia: case review and a new method for shoulder rotation with the sling. Am J Obstet Gynecol. 2015;212(6):784.e1−e7.
- Smellie W. A treatise on the theory and practice of midwifery. 4th ed. London, England; 1762:226−227.
- Hoffman MK, Bailit JL, Branch DW, et al; Consortium on Safe Labor. A comparison of obstetric maneuvers for the acute management of shoulder dystocia. Obstet Gynecol. 2011;117(6):1272−1278.
- Lerner H, Durlacher K, Smith S, Hamilton E. Relationship between head-to-body delivery interval in shoulder dystocia and neonatal depression. Obstet Gynecol. 2011;118(2 pt 1):318−322.
In this article
- Menticoglou maneuver
- Importance of simulation
Zika—a new continent and new complications?
The latest reminders that we live in a medically connected global community are the appearance of the Africa-born Zika virus infection in Brazil and other areas within the Western hemisphere and the subsequent apparent transmission of the disease to female sexual contacts of infected males in the United States. Zika virus’ geographic travels are most certainly of interest; they can be traced from sub-Saharan Africa, where serologically identified outbreaks have continued since 1947, through Asia, Micronesia, Polynesia, and now South and Central America. But what may turn out to be even more interesting than the virus’s travel itinerary is what we may learn about the Zika virus-human host interaction and the subsequent spectrum of clinical disease.
The primary clinical illness following serologically defined infection seems to be relatively uncommon and generally mild: a fairly nondistinctive febrile episode with mild rash, small- and large-joint arthralgias or arthritis, and nonpurulent conjunctivitis. But what has fostered the greatest concern is the epidemiologic association of Zika infection with the neurologic complications of microcephaly and Guillain-Barré syndrome (GBS).
During the 2013–2014 outbreak of Zika infection in French Polynesia, 42 patients with GBS were identified, 100% of whom had serologic evidence suggestive of recent Zika infection, compared with 56% of control patients without GBS.1 Serologic determination of recent infection can be difficult due to cross-reactivity with other flaviviruses, but it seems that in the Polynesian outbreak the risk of GBS might be much less than 1 in 1,000 patients. This is not unlike the incidence of GBS following influenza, Campylobacter, and cytomegalovirus. One explanation for why GBS may follow certain infections is that the infection can trigger antibodies that cross-react with neuronal membrane components. However, those antiganglioside antibodies were not uniformly present in the Polynesian patients who developed GBS following Zika infection. Thus, this may provide an opportunity to further understand the mechanism by which GBS is associated with some infections, in selected patients.
Patients with post-Zika GBS seem to fare well, with a very good prognosis for complete recovery. That is not the case, however, for infants born with microcephaly, another epidemiologically linked complication of Zika infection. In Brazil, the exact incidence rate remains to be determined, and it is not yet certain whether the rate is higher than in the previous Polynesian epidemic (the number of infections is far greater in Brazil, and thus the accuracy of estimated frequency may also be greater), but there may have been a significantly increased frequency of microcephaly in the Polynesian outbreak as well. Like the related West Nile, Saint Louis encephalitis, and Japanese encephalitis viruses, Zika virus has the ability to directly attack certain neurons, and the Zika genome has been detected in brains of infected babies at autopsy. So this particularly devastating aspect of Zika infection may turn out to be relatively easy to understand—perhaps the portal for viral infection of specific neurons is expressed only at certain times during brain development. I’m sure these investigations are under way at a feverish pitch.
Recognizing that new information is being released virtually daily, Flores et al provide a current overview of our understanding of the virus and some practical advice regarding diagnosis and prevention.
As laboratories gear up to devise rapid and more specific diagnostic tests and develop effective anti-Zika vaccines, we hope to learn more about how a seemingly minimally relevant virus, when introduced into a new environment, can wreak clinical havoc. Possible explanations abound—genetic differences in the population, altered immunologic background of infected patients due to prior infection with related viruses such as dengue, or the direct impact of other coinfections. Or, with careful study, it may be discovered that these neurologic issues have been present elsewhere all along, but not previously linked to the Zika virus.
- Cao-Lormeau VM, Blake A, Mons S, et al. Guillain-Barré syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet 2016 Feb 29. pii: S0140-6736(16)00562-6. doi: 10.1016/S0140-6736(16)00562-6. [Epub ahead of print].
The latest reminders that we live in a medically connected global community are the appearance of the Africa-born Zika virus infection in Brazil and other areas within the Western hemisphere and the subsequent apparent transmission of the disease to female sexual contacts of infected males in the United States. Zika virus’ geographic travels are most certainly of interest; they can be traced from sub-Saharan Africa, where serologically identified outbreaks have continued since 1947, through Asia, Micronesia, Polynesia, and now South and Central America. But what may turn out to be even more interesting than the virus’s travel itinerary is what we may learn about the Zika virus-human host interaction and the subsequent spectrum of clinical disease.
The primary clinical illness following serologically defined infection seems to be relatively uncommon and generally mild: a fairly nondistinctive febrile episode with mild rash, small- and large-joint arthralgias or arthritis, and nonpurulent conjunctivitis. But what has fostered the greatest concern is the epidemiologic association of Zika infection with the neurologic complications of microcephaly and Guillain-Barré syndrome (GBS).
During the 2013–2014 outbreak of Zika infection in French Polynesia, 42 patients with GBS were identified, 100% of whom had serologic evidence suggestive of recent Zika infection, compared with 56% of control patients without GBS.1 Serologic determination of recent infection can be difficult due to cross-reactivity with other flaviviruses, but it seems that in the Polynesian outbreak the risk of GBS might be much less than 1 in 1,000 patients. This is not unlike the incidence of GBS following influenza, Campylobacter, and cytomegalovirus. One explanation for why GBS may follow certain infections is that the infection can trigger antibodies that cross-react with neuronal membrane components. However, those antiganglioside antibodies were not uniformly present in the Polynesian patients who developed GBS following Zika infection. Thus, this may provide an opportunity to further understand the mechanism by which GBS is associated with some infections, in selected patients.
Patients with post-Zika GBS seem to fare well, with a very good prognosis for complete recovery. That is not the case, however, for infants born with microcephaly, another epidemiologically linked complication of Zika infection. In Brazil, the exact incidence rate remains to be determined, and it is not yet certain whether the rate is higher than in the previous Polynesian epidemic (the number of infections is far greater in Brazil, and thus the accuracy of estimated frequency may also be greater), but there may have been a significantly increased frequency of microcephaly in the Polynesian outbreak as well. Like the related West Nile, Saint Louis encephalitis, and Japanese encephalitis viruses, Zika virus has the ability to directly attack certain neurons, and the Zika genome has been detected in brains of infected babies at autopsy. So this particularly devastating aspect of Zika infection may turn out to be relatively easy to understand—perhaps the portal for viral infection of specific neurons is expressed only at certain times during brain development. I’m sure these investigations are under way at a feverish pitch.
Recognizing that new information is being released virtually daily, Flores et al provide a current overview of our understanding of the virus and some practical advice regarding diagnosis and prevention.
As laboratories gear up to devise rapid and more specific diagnostic tests and develop effective anti-Zika vaccines, we hope to learn more about how a seemingly minimally relevant virus, when introduced into a new environment, can wreak clinical havoc. Possible explanations abound—genetic differences in the population, altered immunologic background of infected patients due to prior infection with related viruses such as dengue, or the direct impact of other coinfections. Or, with careful study, it may be discovered that these neurologic issues have been present elsewhere all along, but not previously linked to the Zika virus.
The latest reminders that we live in a medically connected global community are the appearance of the Africa-born Zika virus infection in Brazil and other areas within the Western hemisphere and the subsequent apparent transmission of the disease to female sexual contacts of infected males in the United States. Zika virus’ geographic travels are most certainly of interest; they can be traced from sub-Saharan Africa, where serologically identified outbreaks have continued since 1947, through Asia, Micronesia, Polynesia, and now South and Central America. But what may turn out to be even more interesting than the virus’s travel itinerary is what we may learn about the Zika virus-human host interaction and the subsequent spectrum of clinical disease.
The primary clinical illness following serologically defined infection seems to be relatively uncommon and generally mild: a fairly nondistinctive febrile episode with mild rash, small- and large-joint arthralgias or arthritis, and nonpurulent conjunctivitis. But what has fostered the greatest concern is the epidemiologic association of Zika infection with the neurologic complications of microcephaly and Guillain-Barré syndrome (GBS).
During the 2013–2014 outbreak of Zika infection in French Polynesia, 42 patients with GBS were identified, 100% of whom had serologic evidence suggestive of recent Zika infection, compared with 56% of control patients without GBS.1 Serologic determination of recent infection can be difficult due to cross-reactivity with other flaviviruses, but it seems that in the Polynesian outbreak the risk of GBS might be much less than 1 in 1,000 patients. This is not unlike the incidence of GBS following influenza, Campylobacter, and cytomegalovirus. One explanation for why GBS may follow certain infections is that the infection can trigger antibodies that cross-react with neuronal membrane components. However, those antiganglioside antibodies were not uniformly present in the Polynesian patients who developed GBS following Zika infection. Thus, this may provide an opportunity to further understand the mechanism by which GBS is associated with some infections, in selected patients.
Patients with post-Zika GBS seem to fare well, with a very good prognosis for complete recovery. That is not the case, however, for infants born with microcephaly, another epidemiologically linked complication of Zika infection. In Brazil, the exact incidence rate remains to be determined, and it is not yet certain whether the rate is higher than in the previous Polynesian epidemic (the number of infections is far greater in Brazil, and thus the accuracy of estimated frequency may also be greater), but there may have been a significantly increased frequency of microcephaly in the Polynesian outbreak as well. Like the related West Nile, Saint Louis encephalitis, and Japanese encephalitis viruses, Zika virus has the ability to directly attack certain neurons, and the Zika genome has been detected in brains of infected babies at autopsy. So this particularly devastating aspect of Zika infection may turn out to be relatively easy to understand—perhaps the portal for viral infection of specific neurons is expressed only at certain times during brain development. I’m sure these investigations are under way at a feverish pitch.
Recognizing that new information is being released virtually daily, Flores et al provide a current overview of our understanding of the virus and some practical advice regarding diagnosis and prevention.
As laboratories gear up to devise rapid and more specific diagnostic tests and develop effective anti-Zika vaccines, we hope to learn more about how a seemingly minimally relevant virus, when introduced into a new environment, can wreak clinical havoc. Possible explanations abound—genetic differences in the population, altered immunologic background of infected patients due to prior infection with related viruses such as dengue, or the direct impact of other coinfections. Or, with careful study, it may be discovered that these neurologic issues have been present elsewhere all along, but not previously linked to the Zika virus.
- Cao-Lormeau VM, Blake A, Mons S, et al. Guillain-Barré syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet 2016 Feb 29. pii: S0140-6736(16)00562-6. doi: 10.1016/S0140-6736(16)00562-6. [Epub ahead of print].
- Cao-Lormeau VM, Blake A, Mons S, et al. Guillain-Barré syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet 2016 Feb 29. pii: S0140-6736(16)00562-6. doi: 10.1016/S0140-6736(16)00562-6. [Epub ahead of print].
Approvals and presentations flag notable advances in the hem-onc space
Scientific advances and their translation from bench to bedside were front and foremost in the hematology-oncology sphere during 2015 and were bolstered by a record number of therapy approvals by the US Food and Drug Administration (FDA).1 The most recent of those approvals included elotuzumab and ixazomib (both with lenalidomide plus dexamethasone) for previously treated patients with multiple myeloma; and daratumumab as a single agent, also for previously treated multiple myeloma.
Click on the PDF icon at the top of this introduction to read the full article.
Scientific advances and their translation from bench to bedside were front and foremost in the hematology-oncology sphere during 2015 and were bolstered by a record number of therapy approvals by the US Food and Drug Administration (FDA).1 The most recent of those approvals included elotuzumab and ixazomib (both with lenalidomide plus dexamethasone) for previously treated patients with multiple myeloma; and daratumumab as a single agent, also for previously treated multiple myeloma.
Click on the PDF icon at the top of this introduction to read the full article.
Scientific advances and their translation from bench to bedside were front and foremost in the hematology-oncology sphere during 2015 and were bolstered by a record number of therapy approvals by the US Food and Drug Administration (FDA).1 The most recent of those approvals included elotuzumab and ixazomib (both with lenalidomide plus dexamethasone) for previously treated patients with multiple myeloma; and daratumumab as a single agent, also for previously treated multiple myeloma.
Click on the PDF icon at the top of this introduction to read the full article.
When we need to remember that it is more than a job
“I am forever humbled.” So said a heart failure specialist on rounds when I was a resident in the intensive care unit several decades ago. He was talking about the perpetual mismatch between a physician’s level of knowledge and the unpredictability inherent in the management and outcome of critically ill patients. His words ring true for me nearly every day. We should never think we are so smart that we are truly in control of our patients’ outcomes or that we don’t make mistakes—but we also cannot become so paralyzed by the awareness of our limitations that we don’t make decisions.
I have spoken those same powerful words many times on teaching rounds. I also frequently push them to the back of my mind. As a consultant at a major medical center, I am supposed to know. It is a fine line we walk.
I know I am not alone in harboring these self-doubts. Ready access to online information does little to assuage the concern that we can never know enough. Have I ordered enough diagnostic tests to be sure? Have I ordered too many tests and thus will be penalized for providing cost-ineffective care? Should we follow generic guidelines, or deviate from the guidelines based on our clinical instincts, our own interpretation of the literature, and the patient’s unique circumstances and desires?
And then what happens when we make wrong decisions, or even the right decisions that result in a poor patient outcome, which of course is at some point inevitable? We are told to be open about errors, to be honest and transparent about our limitations, to throw down our elaborate emotional and intellectual defensive shields and expose our vulnerability.
But what do we experience emotionally when we are named in a malpractice suit? We may have done all that we thought we could do: we responsibly explored the diagnostic and therapeutic options, provided empathetic care, and listened to the voice of the patient. Yet an adverse outcome still occurred. The practice of medicine is indeed humbling. We feel crushed. We revisit the patient’s care in a vivid perpetual replay loop in our head. Maybe we didn’t evaluate all the options as we should have. If we had been a bit smarter, a bit more efficient, maybe the outcome would have been different.
Then during a deposition, the plaintiff’s counsel points out the temporal and documentary inconsistencies in the electronic medical record: “Doctor, you say you saw the patient at 2:00 pm, but there was no note finalized until 10:00 pm…and why was your documented physical exam exactly the same as the one from the day before and exactly the same as that of the resident who saw the patient that afternoon?” We now feel crushed, totally vulnerable, emotionally trampled, and often isolated and disconnected from our patients and peers. The intellectualized humility becomes transformed into a sense of inadequacy. Why should I keep doing this?
In this issue of the Journal, experienced malpractice attorney Kevin Giordano explores aspects of the malpractice process as they relate to the physicians he defends. He notes how the electronic medical record, a tool ostensibly in place to improve communication and the sharing of medical information between caregivers and patients, can be our worst enemy in a courtroom. He discusses the pressures of our complicated healthcare system that promote documentation errors that he must try to explain away to the jury in our defense, demonstrating that these documentation errors do not necessarily mirror the care and caring of the named physicians. This is critically important information for us to understand and to act on for our personal protection, but it is not his most important message to us.
Mr. Giordano is a sincere, empathetic, and proficient professional. He has spoken for and to many physicians. He has listened to us and observed our behaviors. And as he has defended many of us in a court of law, he has learned to diagnose in his clients the damage that can persist following involvement in a malpractice case and the emotional scars the malpractice experience leaves on physicians. He emphasizes that we must not let the event of a malpractice suit force us to withdraw and strip us of our connection and engagement to patients. If anything, he and Drs. Susan Rehm and Bradford Borden, in an accompanying editorial, urge us to keep in mind that our personal engagement with patients and the mindful practice of medicine is our raison d’être as physicians.
I am continuously humbled by the breadth of the pathology, clinical medicine, and social challenges that I encounter on a daily basis, armed with limited knowledge and experience. It is intellectually rewarding to make an arcane diagnosis or to see an individualized therapy work as I had hoped. But I agree with Mr. Giordano that it is the genuine engagement with patients that provides us with the real joy in the practice of medicine and pushes us to deliver care at a consistently proficient level. We must not forget that, even in the face of significant and emotionally challenging events such as being named in a malpractice suit. It is the nature of our engagement with our patients and our colleagues that make what we do more than a job.
As more physicians in the United States become employed by health systems, I hope that the administrative leaders within these systems truly recognize these issues. As they struggle to balance the provision of safe high-quality care to patients with their increasingly complex financial spreadsheets, I hope that the emotional health of their physician employees is not forgotten. And not just after a malpractice suit.
“I am forever humbled.” So said a heart failure specialist on rounds when I was a resident in the intensive care unit several decades ago. He was talking about the perpetual mismatch between a physician’s level of knowledge and the unpredictability inherent in the management and outcome of critically ill patients. His words ring true for me nearly every day. We should never think we are so smart that we are truly in control of our patients’ outcomes or that we don’t make mistakes—but we also cannot become so paralyzed by the awareness of our limitations that we don’t make decisions.
I have spoken those same powerful words many times on teaching rounds. I also frequently push them to the back of my mind. As a consultant at a major medical center, I am supposed to know. It is a fine line we walk.
I know I am not alone in harboring these self-doubts. Ready access to online information does little to assuage the concern that we can never know enough. Have I ordered enough diagnostic tests to be sure? Have I ordered too many tests and thus will be penalized for providing cost-ineffective care? Should we follow generic guidelines, or deviate from the guidelines based on our clinical instincts, our own interpretation of the literature, and the patient’s unique circumstances and desires?
And then what happens when we make wrong decisions, or even the right decisions that result in a poor patient outcome, which of course is at some point inevitable? We are told to be open about errors, to be honest and transparent about our limitations, to throw down our elaborate emotional and intellectual defensive shields and expose our vulnerability.
But what do we experience emotionally when we are named in a malpractice suit? We may have done all that we thought we could do: we responsibly explored the diagnostic and therapeutic options, provided empathetic care, and listened to the voice of the patient. Yet an adverse outcome still occurred. The practice of medicine is indeed humbling. We feel crushed. We revisit the patient’s care in a vivid perpetual replay loop in our head. Maybe we didn’t evaluate all the options as we should have. If we had been a bit smarter, a bit more efficient, maybe the outcome would have been different.
Then during a deposition, the plaintiff’s counsel points out the temporal and documentary inconsistencies in the electronic medical record: “Doctor, you say you saw the patient at 2:00 pm, but there was no note finalized until 10:00 pm…and why was your documented physical exam exactly the same as the one from the day before and exactly the same as that of the resident who saw the patient that afternoon?” We now feel crushed, totally vulnerable, emotionally trampled, and often isolated and disconnected from our patients and peers. The intellectualized humility becomes transformed into a sense of inadequacy. Why should I keep doing this?
In this issue of the Journal, experienced malpractice attorney Kevin Giordano explores aspects of the malpractice process as they relate to the physicians he defends. He notes how the electronic medical record, a tool ostensibly in place to improve communication and the sharing of medical information between caregivers and patients, can be our worst enemy in a courtroom. He discusses the pressures of our complicated healthcare system that promote documentation errors that he must try to explain away to the jury in our defense, demonstrating that these documentation errors do not necessarily mirror the care and caring of the named physicians. This is critically important information for us to understand and to act on for our personal protection, but it is not his most important message to us.
Mr. Giordano is a sincere, empathetic, and proficient professional. He has spoken for and to many physicians. He has listened to us and observed our behaviors. And as he has defended many of us in a court of law, he has learned to diagnose in his clients the damage that can persist following involvement in a malpractice case and the emotional scars the malpractice experience leaves on physicians. He emphasizes that we must not let the event of a malpractice suit force us to withdraw and strip us of our connection and engagement to patients. If anything, he and Drs. Susan Rehm and Bradford Borden, in an accompanying editorial, urge us to keep in mind that our personal engagement with patients and the mindful practice of medicine is our raison d’être as physicians.
I am continuously humbled by the breadth of the pathology, clinical medicine, and social challenges that I encounter on a daily basis, armed with limited knowledge and experience. It is intellectually rewarding to make an arcane diagnosis or to see an individualized therapy work as I had hoped. But I agree with Mr. Giordano that it is the genuine engagement with patients that provides us with the real joy in the practice of medicine and pushes us to deliver care at a consistently proficient level. We must not forget that, even in the face of significant and emotionally challenging events such as being named in a malpractice suit. It is the nature of our engagement with our patients and our colleagues that make what we do more than a job.
As more physicians in the United States become employed by health systems, I hope that the administrative leaders within these systems truly recognize these issues. As they struggle to balance the provision of safe high-quality care to patients with their increasingly complex financial spreadsheets, I hope that the emotional health of their physician employees is not forgotten. And not just after a malpractice suit.
“I am forever humbled.” So said a heart failure specialist on rounds when I was a resident in the intensive care unit several decades ago. He was talking about the perpetual mismatch between a physician’s level of knowledge and the unpredictability inherent in the management and outcome of critically ill patients. His words ring true for me nearly every day. We should never think we are so smart that we are truly in control of our patients’ outcomes or that we don’t make mistakes—but we also cannot become so paralyzed by the awareness of our limitations that we don’t make decisions.
I have spoken those same powerful words many times on teaching rounds. I also frequently push them to the back of my mind. As a consultant at a major medical center, I am supposed to know. It is a fine line we walk.
I know I am not alone in harboring these self-doubts. Ready access to online information does little to assuage the concern that we can never know enough. Have I ordered enough diagnostic tests to be sure? Have I ordered too many tests and thus will be penalized for providing cost-ineffective care? Should we follow generic guidelines, or deviate from the guidelines based on our clinical instincts, our own interpretation of the literature, and the patient’s unique circumstances and desires?
And then what happens when we make wrong decisions, or even the right decisions that result in a poor patient outcome, which of course is at some point inevitable? We are told to be open about errors, to be honest and transparent about our limitations, to throw down our elaborate emotional and intellectual defensive shields and expose our vulnerability.
But what do we experience emotionally when we are named in a malpractice suit? We may have done all that we thought we could do: we responsibly explored the diagnostic and therapeutic options, provided empathetic care, and listened to the voice of the patient. Yet an adverse outcome still occurred. The practice of medicine is indeed humbling. We feel crushed. We revisit the patient’s care in a vivid perpetual replay loop in our head. Maybe we didn’t evaluate all the options as we should have. If we had been a bit smarter, a bit more efficient, maybe the outcome would have been different.
Then during a deposition, the plaintiff’s counsel points out the temporal and documentary inconsistencies in the electronic medical record: “Doctor, you say you saw the patient at 2:00 pm, but there was no note finalized until 10:00 pm…and why was your documented physical exam exactly the same as the one from the day before and exactly the same as that of the resident who saw the patient that afternoon?” We now feel crushed, totally vulnerable, emotionally trampled, and often isolated and disconnected from our patients and peers. The intellectualized humility becomes transformed into a sense of inadequacy. Why should I keep doing this?
In this issue of the Journal, experienced malpractice attorney Kevin Giordano explores aspects of the malpractice process as they relate to the physicians he defends. He notes how the electronic medical record, a tool ostensibly in place to improve communication and the sharing of medical information between caregivers and patients, can be our worst enemy in a courtroom. He discusses the pressures of our complicated healthcare system that promote documentation errors that he must try to explain away to the jury in our defense, demonstrating that these documentation errors do not necessarily mirror the care and caring of the named physicians. This is critically important information for us to understand and to act on for our personal protection, but it is not his most important message to us.
Mr. Giordano is a sincere, empathetic, and proficient professional. He has spoken for and to many physicians. He has listened to us and observed our behaviors. And as he has defended many of us in a court of law, he has learned to diagnose in his clients the damage that can persist following involvement in a malpractice case and the emotional scars the malpractice experience leaves on physicians. He emphasizes that we must not let the event of a malpractice suit force us to withdraw and strip us of our connection and engagement to patients. If anything, he and Drs. Susan Rehm and Bradford Borden, in an accompanying editorial, urge us to keep in mind that our personal engagement with patients and the mindful practice of medicine is our raison d’être as physicians.
I am continuously humbled by the breadth of the pathology, clinical medicine, and social challenges that I encounter on a daily basis, armed with limited knowledge and experience. It is intellectually rewarding to make an arcane diagnosis or to see an individualized therapy work as I had hoped. But I agree with Mr. Giordano that it is the genuine engagement with patients that provides us with the real joy in the practice of medicine and pushes us to deliver care at a consistently proficient level. We must not forget that, even in the face of significant and emotionally challenging events such as being named in a malpractice suit. It is the nature of our engagement with our patients and our colleagues that make what we do more than a job.
As more physicians in the United States become employed by health systems, I hope that the administrative leaders within these systems truly recognize these issues. As they struggle to balance the provision of safe high-quality care to patients with their increasingly complex financial spreadsheets, I hope that the emotional health of their physician employees is not forgotten. And not just after a malpractice suit.
Making immunotherapy part of routine breast cancer treatment
Cancer treatment is evolving rapidly, and 2015 was no exception. It was the year of immunotherapy. Following the approval by the US Food and Drug Administration in 2014 of pembrolizumab for melanoma, 2015 saw approvals of nivolumab for melanoma, lung cancer, and kidney cancer; pembrolizumab for lung cancer; and the combination of ipilimumab and nivolimab for melanoma. That’s an impressive list of immunotherapy approvals for a single year.
Click on the PDF icon at the top of this introduction to read the full article.
Cancer treatment is evolving rapidly, and 2015 was no exception. It was the year of immunotherapy. Following the approval by the US Food and Drug Administration in 2014 of pembrolizumab for melanoma, 2015 saw approvals of nivolumab for melanoma, lung cancer, and kidney cancer; pembrolizumab for lung cancer; and the combination of ipilimumab and nivolimab for melanoma. That’s an impressive list of immunotherapy approvals for a single year.
Click on the PDF icon at the top of this introduction to read the full article.
Cancer treatment is evolving rapidly, and 2015 was no exception. It was the year of immunotherapy. Following the approval by the US Food and Drug Administration in 2014 of pembrolizumab for melanoma, 2015 saw approvals of nivolumab for melanoma, lung cancer, and kidney cancer; pembrolizumab for lung cancer; and the combination of ipilimumab and nivolimab for melanoma. That’s an impressive list of immunotherapy approvals for a single year.
Click on the PDF icon at the top of this introduction to read the full article.
Can we solve the problem of inadequate contraception for women at high risk for adverse pregnancy outcomes?
In the United States contraception practices are slowly improving, with robust evidence for the increased use of long-acting reversible contraceptives and preliminary data that the unintended pregnancy rate may be decreasing for the first time in many years.1 There remains a major gap in contraception practice, however: US women with chronic disease who are at high risk for adverse pregnancy outcomes are not receiving adequate contraceptive counseling or adequate contraception.2,3 In one study, the majority of women with hypertension, diabetes, epilepsy, stroke, heart disease, lupus, or thrombophilia were not using a prescription contraceptive.3
Recently, I have seen women with major medical problems, who have had many visits with specialists and primary care clinicians, but who have not had their contraceptive needs prioritized. Here are but a few examples:
- We recently cared for a patient with heart disease and severe pulmonary hypertension, who had many procedures performed by cardiologists and cardiac surgeons, but contraception had not been prioritized as one of her foremost medical needs.
- A young woman who had a pulmonary embolism 1-month postpartum reported that her primary care clinician said that she could never use any hormonal contraceptive, including the progestin-only pill, progestin-implant, and the progestin-releasing intrauterine device (IUD). She was not taking a contraceptive and had an unplanned pregnancy.
- A middle-aged woman with diabetes and a glycosylated hemoglo‑ bin A1c (HbA1c) value greater than 10% was regularly seeing her primary care clinician, but was not using an effective contraceptive. She became pregnant with a fetus that had a major congenital anomaly.
Clearly, there is a major gap between current and optimal contraceptive services for women with chronic medical problems. Women with diabetes and heart disease are affected substantially, as the evidence I present in this editorial indicates. Studies also show that women taking teratogenic medications do not receive the vital counseling that they should regarding contraception. Given the potential detrimental adverse events to both mother and fetus, obstetrician-gynecologists are poised to offer solutions to this concerning inadequacy of care.
DiabetesWomen with diabetes and an abnormally elevated HbA1c level are at high risk for many adverse pregnancy outcomes, including major congenital malformations and intrauterine fetal demise.4 Unfortunately, results of many studies indicate that women with diabetes are not receiving adequate contraceptive services.5–7 In one review of records at Kaiser Permanente Northern California, investigators reported that 62% of 122,921 healthy women, but only 48% of 8,182 women with diabetes, received contraceptive counseling, a contraceptive prescription, or contraceptive services.5
Why is it that so many women with diabetes do not receive contraceptive services? One possibility is that clinicians are reluctant to prescribe oral hormonal contraceptives that contain estrogen to their patients with diabetes because of a perceived increased risk of cardiovascular events.8 In the Kaiser study, 31% of the healthy women, and only 13% of the women with diabetes, were using a pill, patch, or ring (most of which contain estrogen).5 In this same study, the rate of utilization of an IUD was similar in the healthy (6.5%) and diabetic (5.6%) women. The IUD is known to be safe for use in women with diabetes.9
The low rate of utilization of intrauterine contraception by women with diabetes is a gap that gynecologists are well positioned to help solve.
Heart diseaseIn developed countries, a major cause of maternal mortality is pregnancy among women with congenital or acquired heart disease.10 Misinformation is a common problem in contraceptive counseling. In a recent study of 83 sexually active women with congenital heart disease, 6 women were told that they could not use an IUD or progestin-implant because they were unsafe for those with repaired congenital heart disease.11 In this cohort of women, who were at high risk for adverse pregnancy outcomes, 45% of pregnancies were unplanned, similar to the rate among healthy women.
On a positive note, authors of a small study from Maryland found that, among women with heart disease, the self-reported use of a contraceptive increased from 60% prepregnancy to 93% following delivery.12
Clearly, patient interaction with qualified women’s health clinicians can increase contraceptive use in those with high-risk medical issues.
Teratogenic medicationsShould reproductive-age women taking long-term methotrexate for treatment of rheumatoid conditions receive contraceptive counseling? The answer is clearly, “yes.” Methotrexate can cause fetal death or major congenital malformations, such as absence of digits and oxycephaly (premature closure of the skull sutures). All women of reproductive age prescribed known teratogens should receive effective contraception. Unfortunately, data do not indicate this is occurring.
In one study of 1,694 adolescents and young women aged 14 to 25 years who were prescribed a teratogen, only 29% received documented contraception counseling, and only 11% received a contraceptive prescription or were documented to be actively using a contraceptive.13 The most commonly prescribed teratogens in this study were topiramate, methotrexate, and isoretinoin. Among the specialists who prescribed the medications, dermatologists documented contraceptive counseling in 47% of visits—likely because of the federally mandated risk mitigation system for prescribing isoretinoin. Neurologists and hematologists were least likely to document contraceptive counseling, at 16% and 28%, respectively.13
In a study of 488,175 women aged 15 to 44 years receiving care from clinicians at Kaiser Permanente Northern California, contraceptive counseling documentation was compared among women prescribed US Food and Drug Administration (FDA) category A or B medications (nonteratogenic) versus FDA category D or X medications (teratogenic).14 The rate at which women had no contraceptive counseling recorded was similar whether a teratogenic (47.6%) or nonteratogenic (46%) medication was prescribed. Clearly, there is a gap between current and optimal practice when teratogens are prescribed to women of reproductive age.
What could improve contraceptive services for women with serious medical problems?One promising approach is to include contraception status as a vital sign for all women and men of reproductive age. Most electronic medical records prioritize assessment of such health vital signs as allergies, smoking status, depression screening, falls prevention, blood pressure, temperature, heart rate, weight, and height. Contraception status is of equal importance to these vital signs in women and men of reproductive age and should be routinely documented.
Another intervention is to create a standard of care in which reproductive-age women with major medical problems are routinely referred to a clinician who has the time and skill to provide a comprehensive contraception visit. Health systems could take greater responsibility for managing the contraception practices of their members. For example, within a given accountable care organization the electronic health record could be used to identify adult women of reproductive age with diabetes and an HbA1c level greater than 7%. These women could be contacted to ascertain their contraception status and their need for a contraception health visit. Electronic health records could be utilized to identify all reproductive-age women taking a teratogenic medication. A computer-generated alert could be sent to the responsible clinician recommending referral to an obstetrician-gynecologist for a contraceptive services visit.15
Pharmacists could be more proactive in highlighting the importance of contraception for women prescribed teratogens and in recommending a contraceptive visit. In some states pharmacists can offer an oral hormonal contraceptive to women who are prescribed a teratogen and at risk for becoming pregnant.
How do you propose to address lack of counseling?As an experienced clinician, you likely have ideas about how to improve contraceptive counseling for women with significant medical problems. Please let me know what interventions you think would best improve the use of contraception in this group of high-risk women by emailing me at OBG Management: [email protected].
Tell us…What are your ideas to improve contraception counseling for women with significant medical problems that put them at high risk for adverse pregnancy outcomes?
Send your letter to the editor to [email protected]. Please include the city and state in which you practice
- Kavanaugh ML, Jerman J, Finer LB. Changes in use of long-acting reversible contraceptive methods among US women, 2009-2012. Obstet Gynecol. 2015;126(5):917–927.
- DeNoble AE, Hall KS, Xu X, Zochowski MK, Piehl K, Dalton VK. Receipt of prescription contraception by commercially insured women with chronic medical conditions. Obstet Gynecol. 2014;123(6):1213–1220.
- Champaloux SW, Tepper NK, Curtis KM, et al. Contraceptive use among women with medical conditions in a nationwide privately insured population. Obstet Gynecol. 2015;126(6):1151–1159.
- Klingensmith GJ, Pyle L, Nadeau KJ, et al; TODAY Study Group. Pregnancy outcomes in youth with type 2 diabetes: the TODAY study experience. Diabetes Care. 2016;39(1):122–129.
- Schwarz EB, Postlethwaite D, Hung YY, Lantzman E, Armstrong MA, Horberg MA. Provision of contraceptive services to women with diabetes mellitus. J Gen Int Med. 2011;27(2):196–201.
- Schwarz EB, Maselli J, Gonzales R. Contraceptive counseling of diabetic women of reproductive age. Obstet Gynecol. 2006;107(5):1070–1074.
- Chuang CH, Chase GA, Bensyl DM, Weisman CS. Contraceptive use by diabetic and obese women. Womens Health Issues. 2005;15(4):167–173.
- Lidegaard O. Hormonal contraception, thrombosis and age. Expert Opin Drug Safe. 2014;13(10):1353–1360.
- Goldstuck ND, Steyn PS. The intrauterine device in women with diabetes mellitus type I and II: a systematic review. ISRN Obstet Gynecol. 2013:814062. doi.10.1155/2013/814062.
- Thompson JL, Kuklina EV, Bateman BT, Callaghan WM, James AH, Grotegut CA. Medical and obstetric outcomes among pregnant women with congenital heart disease. Obstet Gynecol. 2015;126(2):346–354.
- Lindley KJ, Madden T, Cahill AG, Ludbrook PA, Billadello JJ. Contraceptive use and unintended pregnancy in women with congenital heart disease. Obstet Gynecol. 2015;126(2):363–369.
- Perritt JB, Burke A, Jasmshidli R, Wang J, Fox M. Contraception counseling, pregnancy intention and contraception use in women with medical problems: an analysis of data from the Maryland Pregnancy Risk Assessment Monitoring System (PRAMS). Contraception. 2013;88(2):263–268.
- Stancil SL, Miller M, Briggs H, Lynch D, Goggin K, Kearns G. Contraceptive provision to adolescent females prescribed teratogenic medications. Pediatrics. 2016;137(1):1–8.
- Schwarz EB, Postlewaite DA, Hung YY, Armstrong MA. Documentation of contraception and pregnancy when prescribing potentially teratogenic medications for reproductive-age women. Ann Int Med. 2007;147(6):370–376.
- Mody SK, Wu J, Ornelas M, et al. Using the electronic medical record to refer women taking category D or X medications for teratogen and contraceptive counseling. Birth Defects Res A Clin Mol Teratol. 2015;103(7):644–647.
In the United States contraception practices are slowly improving, with robust evidence for the increased use of long-acting reversible contraceptives and preliminary data that the unintended pregnancy rate may be decreasing for the first time in many years.1 There remains a major gap in contraception practice, however: US women with chronic disease who are at high risk for adverse pregnancy outcomes are not receiving adequate contraceptive counseling or adequate contraception.2,3 In one study, the majority of women with hypertension, diabetes, epilepsy, stroke, heart disease, lupus, or thrombophilia were not using a prescription contraceptive.3
Recently, I have seen women with major medical problems, who have had many visits with specialists and primary care clinicians, but who have not had their contraceptive needs prioritized. Here are but a few examples:
- We recently cared for a patient with heart disease and severe pulmonary hypertension, who had many procedures performed by cardiologists and cardiac surgeons, but contraception had not been prioritized as one of her foremost medical needs.
- A young woman who had a pulmonary embolism 1-month postpartum reported that her primary care clinician said that she could never use any hormonal contraceptive, including the progestin-only pill, progestin-implant, and the progestin-releasing intrauterine device (IUD). She was not taking a contraceptive and had an unplanned pregnancy.
- A middle-aged woman with diabetes and a glycosylated hemoglo‑ bin A1c (HbA1c) value greater than 10% was regularly seeing her primary care clinician, but was not using an effective contraceptive. She became pregnant with a fetus that had a major congenital anomaly.
Clearly, there is a major gap between current and optimal contraceptive services for women with chronic medical problems. Women with diabetes and heart disease are affected substantially, as the evidence I present in this editorial indicates. Studies also show that women taking teratogenic medications do not receive the vital counseling that they should regarding contraception. Given the potential detrimental adverse events to both mother and fetus, obstetrician-gynecologists are poised to offer solutions to this concerning inadequacy of care.
DiabetesWomen with diabetes and an abnormally elevated HbA1c level are at high risk for many adverse pregnancy outcomes, including major congenital malformations and intrauterine fetal demise.4 Unfortunately, results of many studies indicate that women with diabetes are not receiving adequate contraceptive services.5–7 In one review of records at Kaiser Permanente Northern California, investigators reported that 62% of 122,921 healthy women, but only 48% of 8,182 women with diabetes, received contraceptive counseling, a contraceptive prescription, or contraceptive services.5
Why is it that so many women with diabetes do not receive contraceptive services? One possibility is that clinicians are reluctant to prescribe oral hormonal contraceptives that contain estrogen to their patients with diabetes because of a perceived increased risk of cardiovascular events.8 In the Kaiser study, 31% of the healthy women, and only 13% of the women with diabetes, were using a pill, patch, or ring (most of which contain estrogen).5 In this same study, the rate of utilization of an IUD was similar in the healthy (6.5%) and diabetic (5.6%) women. The IUD is known to be safe for use in women with diabetes.9
The low rate of utilization of intrauterine contraception by women with diabetes is a gap that gynecologists are well positioned to help solve.
Heart diseaseIn developed countries, a major cause of maternal mortality is pregnancy among women with congenital or acquired heart disease.10 Misinformation is a common problem in contraceptive counseling. In a recent study of 83 sexually active women with congenital heart disease, 6 women were told that they could not use an IUD or progestin-implant because they were unsafe for those with repaired congenital heart disease.11 In this cohort of women, who were at high risk for adverse pregnancy outcomes, 45% of pregnancies were unplanned, similar to the rate among healthy women.
On a positive note, authors of a small study from Maryland found that, among women with heart disease, the self-reported use of a contraceptive increased from 60% prepregnancy to 93% following delivery.12
Clearly, patient interaction with qualified women’s health clinicians can increase contraceptive use in those with high-risk medical issues.
Teratogenic medicationsShould reproductive-age women taking long-term methotrexate for treatment of rheumatoid conditions receive contraceptive counseling? The answer is clearly, “yes.” Methotrexate can cause fetal death or major congenital malformations, such as absence of digits and oxycephaly (premature closure of the skull sutures). All women of reproductive age prescribed known teratogens should receive effective contraception. Unfortunately, data do not indicate this is occurring.
In one study of 1,694 adolescents and young women aged 14 to 25 years who were prescribed a teratogen, only 29% received documented contraception counseling, and only 11% received a contraceptive prescription or were documented to be actively using a contraceptive.13 The most commonly prescribed teratogens in this study were topiramate, methotrexate, and isoretinoin. Among the specialists who prescribed the medications, dermatologists documented contraceptive counseling in 47% of visits—likely because of the federally mandated risk mitigation system for prescribing isoretinoin. Neurologists and hematologists were least likely to document contraceptive counseling, at 16% and 28%, respectively.13
In a study of 488,175 women aged 15 to 44 years receiving care from clinicians at Kaiser Permanente Northern California, contraceptive counseling documentation was compared among women prescribed US Food and Drug Administration (FDA) category A or B medications (nonteratogenic) versus FDA category D or X medications (teratogenic).14 The rate at which women had no contraceptive counseling recorded was similar whether a teratogenic (47.6%) or nonteratogenic (46%) medication was prescribed. Clearly, there is a gap between current and optimal practice when teratogens are prescribed to women of reproductive age.
What could improve contraceptive services for women with serious medical problems?One promising approach is to include contraception status as a vital sign for all women and men of reproductive age. Most electronic medical records prioritize assessment of such health vital signs as allergies, smoking status, depression screening, falls prevention, blood pressure, temperature, heart rate, weight, and height. Contraception status is of equal importance to these vital signs in women and men of reproductive age and should be routinely documented.
Another intervention is to create a standard of care in which reproductive-age women with major medical problems are routinely referred to a clinician who has the time and skill to provide a comprehensive contraception visit. Health systems could take greater responsibility for managing the contraception practices of their members. For example, within a given accountable care organization the electronic health record could be used to identify adult women of reproductive age with diabetes and an HbA1c level greater than 7%. These women could be contacted to ascertain their contraception status and their need for a contraception health visit. Electronic health records could be utilized to identify all reproductive-age women taking a teratogenic medication. A computer-generated alert could be sent to the responsible clinician recommending referral to an obstetrician-gynecologist for a contraceptive services visit.15
Pharmacists could be more proactive in highlighting the importance of contraception for women prescribed teratogens and in recommending a contraceptive visit. In some states pharmacists can offer an oral hormonal contraceptive to women who are prescribed a teratogen and at risk for becoming pregnant.
How do you propose to address lack of counseling?As an experienced clinician, you likely have ideas about how to improve contraceptive counseling for women with significant medical problems. Please let me know what interventions you think would best improve the use of contraception in this group of high-risk women by emailing me at OBG Management: [email protected].
Tell us…What are your ideas to improve contraception counseling for women with significant medical problems that put them at high risk for adverse pregnancy outcomes?
Send your letter to the editor to [email protected]. Please include the city and state in which you practice
In the United States contraception practices are slowly improving, with robust evidence for the increased use of long-acting reversible contraceptives and preliminary data that the unintended pregnancy rate may be decreasing for the first time in many years.1 There remains a major gap in contraception practice, however: US women with chronic disease who are at high risk for adverse pregnancy outcomes are not receiving adequate contraceptive counseling or adequate contraception.2,3 In one study, the majority of women with hypertension, diabetes, epilepsy, stroke, heart disease, lupus, or thrombophilia were not using a prescription contraceptive.3
Recently, I have seen women with major medical problems, who have had many visits with specialists and primary care clinicians, but who have not had their contraceptive needs prioritized. Here are but a few examples:
- We recently cared for a patient with heart disease and severe pulmonary hypertension, who had many procedures performed by cardiologists and cardiac surgeons, but contraception had not been prioritized as one of her foremost medical needs.
- A young woman who had a pulmonary embolism 1-month postpartum reported that her primary care clinician said that she could never use any hormonal contraceptive, including the progestin-only pill, progestin-implant, and the progestin-releasing intrauterine device (IUD). She was not taking a contraceptive and had an unplanned pregnancy.
- A middle-aged woman with diabetes and a glycosylated hemoglo‑ bin A1c (HbA1c) value greater than 10% was regularly seeing her primary care clinician, but was not using an effective contraceptive. She became pregnant with a fetus that had a major congenital anomaly.
Clearly, there is a major gap between current and optimal contraceptive services for women with chronic medical problems. Women with diabetes and heart disease are affected substantially, as the evidence I present in this editorial indicates. Studies also show that women taking teratogenic medications do not receive the vital counseling that they should regarding contraception. Given the potential detrimental adverse events to both mother and fetus, obstetrician-gynecologists are poised to offer solutions to this concerning inadequacy of care.
DiabetesWomen with diabetes and an abnormally elevated HbA1c level are at high risk for many adverse pregnancy outcomes, including major congenital malformations and intrauterine fetal demise.4 Unfortunately, results of many studies indicate that women with diabetes are not receiving adequate contraceptive services.5–7 In one review of records at Kaiser Permanente Northern California, investigators reported that 62% of 122,921 healthy women, but only 48% of 8,182 women with diabetes, received contraceptive counseling, a contraceptive prescription, or contraceptive services.5
Why is it that so many women with diabetes do not receive contraceptive services? One possibility is that clinicians are reluctant to prescribe oral hormonal contraceptives that contain estrogen to their patients with diabetes because of a perceived increased risk of cardiovascular events.8 In the Kaiser study, 31% of the healthy women, and only 13% of the women with diabetes, were using a pill, patch, or ring (most of which contain estrogen).5 In this same study, the rate of utilization of an IUD was similar in the healthy (6.5%) and diabetic (5.6%) women. The IUD is known to be safe for use in women with diabetes.9
The low rate of utilization of intrauterine contraception by women with diabetes is a gap that gynecologists are well positioned to help solve.
Heart diseaseIn developed countries, a major cause of maternal mortality is pregnancy among women with congenital or acquired heart disease.10 Misinformation is a common problem in contraceptive counseling. In a recent study of 83 sexually active women with congenital heart disease, 6 women were told that they could not use an IUD or progestin-implant because they were unsafe for those with repaired congenital heart disease.11 In this cohort of women, who were at high risk for adverse pregnancy outcomes, 45% of pregnancies were unplanned, similar to the rate among healthy women.
On a positive note, authors of a small study from Maryland found that, among women with heart disease, the self-reported use of a contraceptive increased from 60% prepregnancy to 93% following delivery.12
Clearly, patient interaction with qualified women’s health clinicians can increase contraceptive use in those with high-risk medical issues.
Teratogenic medicationsShould reproductive-age women taking long-term methotrexate for treatment of rheumatoid conditions receive contraceptive counseling? The answer is clearly, “yes.” Methotrexate can cause fetal death or major congenital malformations, such as absence of digits and oxycephaly (premature closure of the skull sutures). All women of reproductive age prescribed known teratogens should receive effective contraception. Unfortunately, data do not indicate this is occurring.
In one study of 1,694 adolescents and young women aged 14 to 25 years who were prescribed a teratogen, only 29% received documented contraception counseling, and only 11% received a contraceptive prescription or were documented to be actively using a contraceptive.13 The most commonly prescribed teratogens in this study were topiramate, methotrexate, and isoretinoin. Among the specialists who prescribed the medications, dermatologists documented contraceptive counseling in 47% of visits—likely because of the federally mandated risk mitigation system for prescribing isoretinoin. Neurologists and hematologists were least likely to document contraceptive counseling, at 16% and 28%, respectively.13
In a study of 488,175 women aged 15 to 44 years receiving care from clinicians at Kaiser Permanente Northern California, contraceptive counseling documentation was compared among women prescribed US Food and Drug Administration (FDA) category A or B medications (nonteratogenic) versus FDA category D or X medications (teratogenic).14 The rate at which women had no contraceptive counseling recorded was similar whether a teratogenic (47.6%) or nonteratogenic (46%) medication was prescribed. Clearly, there is a gap between current and optimal practice when teratogens are prescribed to women of reproductive age.
What could improve contraceptive services for women with serious medical problems?One promising approach is to include contraception status as a vital sign for all women and men of reproductive age. Most electronic medical records prioritize assessment of such health vital signs as allergies, smoking status, depression screening, falls prevention, blood pressure, temperature, heart rate, weight, and height. Contraception status is of equal importance to these vital signs in women and men of reproductive age and should be routinely documented.
Another intervention is to create a standard of care in which reproductive-age women with major medical problems are routinely referred to a clinician who has the time and skill to provide a comprehensive contraception visit. Health systems could take greater responsibility for managing the contraception practices of their members. For example, within a given accountable care organization the electronic health record could be used to identify adult women of reproductive age with diabetes and an HbA1c level greater than 7%. These women could be contacted to ascertain their contraception status and their need for a contraception health visit. Electronic health records could be utilized to identify all reproductive-age women taking a teratogenic medication. A computer-generated alert could be sent to the responsible clinician recommending referral to an obstetrician-gynecologist for a contraceptive services visit.15
Pharmacists could be more proactive in highlighting the importance of contraception for women prescribed teratogens and in recommending a contraceptive visit. In some states pharmacists can offer an oral hormonal contraceptive to women who are prescribed a teratogen and at risk for becoming pregnant.
How do you propose to address lack of counseling?As an experienced clinician, you likely have ideas about how to improve contraceptive counseling for women with significant medical problems. Please let me know what interventions you think would best improve the use of contraception in this group of high-risk women by emailing me at OBG Management: [email protected].
Tell us…What are your ideas to improve contraception counseling for women with significant medical problems that put them at high risk for adverse pregnancy outcomes?
Send your letter to the editor to [email protected]. Please include the city and state in which you practice
- Kavanaugh ML, Jerman J, Finer LB. Changes in use of long-acting reversible contraceptive methods among US women, 2009-2012. Obstet Gynecol. 2015;126(5):917–927.
- DeNoble AE, Hall KS, Xu X, Zochowski MK, Piehl K, Dalton VK. Receipt of prescription contraception by commercially insured women with chronic medical conditions. Obstet Gynecol. 2014;123(6):1213–1220.
- Champaloux SW, Tepper NK, Curtis KM, et al. Contraceptive use among women with medical conditions in a nationwide privately insured population. Obstet Gynecol. 2015;126(6):1151–1159.
- Klingensmith GJ, Pyle L, Nadeau KJ, et al; TODAY Study Group. Pregnancy outcomes in youth with type 2 diabetes: the TODAY study experience. Diabetes Care. 2016;39(1):122–129.
- Schwarz EB, Postlethwaite D, Hung YY, Lantzman E, Armstrong MA, Horberg MA. Provision of contraceptive services to women with diabetes mellitus. J Gen Int Med. 2011;27(2):196–201.
- Schwarz EB, Maselli J, Gonzales R. Contraceptive counseling of diabetic women of reproductive age. Obstet Gynecol. 2006;107(5):1070–1074.
- Chuang CH, Chase GA, Bensyl DM, Weisman CS. Contraceptive use by diabetic and obese women. Womens Health Issues. 2005;15(4):167–173.
- Lidegaard O. Hormonal contraception, thrombosis and age. Expert Opin Drug Safe. 2014;13(10):1353–1360.
- Goldstuck ND, Steyn PS. The intrauterine device in women with diabetes mellitus type I and II: a systematic review. ISRN Obstet Gynecol. 2013:814062. doi.10.1155/2013/814062.
- Thompson JL, Kuklina EV, Bateman BT, Callaghan WM, James AH, Grotegut CA. Medical and obstetric outcomes among pregnant women with congenital heart disease. Obstet Gynecol. 2015;126(2):346–354.
- Lindley KJ, Madden T, Cahill AG, Ludbrook PA, Billadello JJ. Contraceptive use and unintended pregnancy in women with congenital heart disease. Obstet Gynecol. 2015;126(2):363–369.
- Perritt JB, Burke A, Jasmshidli R, Wang J, Fox M. Contraception counseling, pregnancy intention and contraception use in women with medical problems: an analysis of data from the Maryland Pregnancy Risk Assessment Monitoring System (PRAMS). Contraception. 2013;88(2):263–268.
- Stancil SL, Miller M, Briggs H, Lynch D, Goggin K, Kearns G. Contraceptive provision to adolescent females prescribed teratogenic medications. Pediatrics. 2016;137(1):1–8.
- Schwarz EB, Postlewaite DA, Hung YY, Armstrong MA. Documentation of contraception and pregnancy when prescribing potentially teratogenic medications for reproductive-age women. Ann Int Med. 2007;147(6):370–376.
- Mody SK, Wu J, Ornelas M, et al. Using the electronic medical record to refer women taking category D or X medications for teratogen and contraceptive counseling. Birth Defects Res A Clin Mol Teratol. 2015;103(7):644–647.
- Kavanaugh ML, Jerman J, Finer LB. Changes in use of long-acting reversible contraceptive methods among US women, 2009-2012. Obstet Gynecol. 2015;126(5):917–927.
- DeNoble AE, Hall KS, Xu X, Zochowski MK, Piehl K, Dalton VK. Receipt of prescription contraception by commercially insured women with chronic medical conditions. Obstet Gynecol. 2014;123(6):1213–1220.
- Champaloux SW, Tepper NK, Curtis KM, et al. Contraceptive use among women with medical conditions in a nationwide privately insured population. Obstet Gynecol. 2015;126(6):1151–1159.
- Klingensmith GJ, Pyle L, Nadeau KJ, et al; TODAY Study Group. Pregnancy outcomes in youth with type 2 diabetes: the TODAY study experience. Diabetes Care. 2016;39(1):122–129.
- Schwarz EB, Postlethwaite D, Hung YY, Lantzman E, Armstrong MA, Horberg MA. Provision of contraceptive services to women with diabetes mellitus. J Gen Int Med. 2011;27(2):196–201.
- Schwarz EB, Maselli J, Gonzales R. Contraceptive counseling of diabetic women of reproductive age. Obstet Gynecol. 2006;107(5):1070–1074.
- Chuang CH, Chase GA, Bensyl DM, Weisman CS. Contraceptive use by diabetic and obese women. Womens Health Issues. 2005;15(4):167–173.
- Lidegaard O. Hormonal contraception, thrombosis and age. Expert Opin Drug Safe. 2014;13(10):1353–1360.
- Goldstuck ND, Steyn PS. The intrauterine device in women with diabetes mellitus type I and II: a systematic review. ISRN Obstet Gynecol. 2013:814062. doi.10.1155/2013/814062.
- Thompson JL, Kuklina EV, Bateman BT, Callaghan WM, James AH, Grotegut CA. Medical and obstetric outcomes among pregnant women with congenital heart disease. Obstet Gynecol. 2015;126(2):346–354.
- Lindley KJ, Madden T, Cahill AG, Ludbrook PA, Billadello JJ. Contraceptive use and unintended pregnancy in women with congenital heart disease. Obstet Gynecol. 2015;126(2):363–369.
- Perritt JB, Burke A, Jasmshidli R, Wang J, Fox M. Contraception counseling, pregnancy intention and contraception use in women with medical problems: an analysis of data from the Maryland Pregnancy Risk Assessment Monitoring System (PRAMS). Contraception. 2013;88(2):263–268.
- Stancil SL, Miller M, Briggs H, Lynch D, Goggin K, Kearns G. Contraceptive provision to adolescent females prescribed teratogenic medications. Pediatrics. 2016;137(1):1–8.
- Schwarz EB, Postlewaite DA, Hung YY, Armstrong MA. Documentation of contraception and pregnancy when prescribing potentially teratogenic medications for reproductive-age women. Ann Int Med. 2007;147(6):370–376.
- Mody SK, Wu J, Ornelas M, et al. Using the electronic medical record to refer women taking category D or X medications for teratogen and contraceptive counseling. Birth Defects Res A Clin Mol Teratol. 2015;103(7):644–647.
Hope may not be the best component of an exercise regimen
Judging by the crowd and the difficulty in finding a locker at my gym on January 1, a lot of people are serious about their 2016 New Year’s resolution to exercise and lose weight. But as most of us have experienced personally and professionally, embarking on a well-intended effort to exercise in the hope of losing weight more often results in frustration than a trip to the store to buy smaller-sized clothes.
The frequent answer to the question “What did the doctor say at your visit?” provides a partial explanation of this phenomenon: “Nothing, just that I should exercise and lose weight” is the usual hackneyed response. Nothing—as in nothing unexpected, nothing significant, and nothing specific was said. It is with this lack of specific advice that I feel many of us let our patients down.
We admonish patients to eat fewer calories, avoid the evil carbs, walk 10,000 steps, ride a bike, use the elliptical, or swim three times a week. There is a concrete but broad nature to these suggestions, but there is also a familiarity and a lack of specificity that leaves patients feeling that there is no science behind them. And the truth is that many of us are not comfortable enough with current data from our exercise physiology colleagues to have a detailed discussion with our patients that pairs their specific goals with an exercise regimen and diet most likely to be beneficial. We may fear sounding like the morning talk show doctors, offering sound bites instead of engaging in an evidence-based dialogue with our patients.
Many of our patients cannot afford a personal trainer to guide and cajole them through a successful regimen—assuming that they, or we, can separate myth and fact and choose an appropriate trainer. We should try to be their guide and sounding board as well as coach and cheerleader.
In this issue of the Journal, John and Christopher Higgins present a primer on the background information to use when talking with our patients about starting an exercise program focused on weight loss. They provide useful references that support specific approaches to achieve realistic expectations, and they review and compare various strategies.
I’m sure by March it will again be easier to find a locker at my gym. And I hope by then that my new workout plan will be more scientifically based, as well as a bit more effective. Even data-based hope springs eternal.
Judging by the crowd and the difficulty in finding a locker at my gym on January 1, a lot of people are serious about their 2016 New Year’s resolution to exercise and lose weight. But as most of us have experienced personally and professionally, embarking on a well-intended effort to exercise in the hope of losing weight more often results in frustration than a trip to the store to buy smaller-sized clothes.
The frequent answer to the question “What did the doctor say at your visit?” provides a partial explanation of this phenomenon: “Nothing, just that I should exercise and lose weight” is the usual hackneyed response. Nothing—as in nothing unexpected, nothing significant, and nothing specific was said. It is with this lack of specific advice that I feel many of us let our patients down.
We admonish patients to eat fewer calories, avoid the evil carbs, walk 10,000 steps, ride a bike, use the elliptical, or swim three times a week. There is a concrete but broad nature to these suggestions, but there is also a familiarity and a lack of specificity that leaves patients feeling that there is no science behind them. And the truth is that many of us are not comfortable enough with current data from our exercise physiology colleagues to have a detailed discussion with our patients that pairs their specific goals with an exercise regimen and diet most likely to be beneficial. We may fear sounding like the morning talk show doctors, offering sound bites instead of engaging in an evidence-based dialogue with our patients.
Many of our patients cannot afford a personal trainer to guide and cajole them through a successful regimen—assuming that they, or we, can separate myth and fact and choose an appropriate trainer. We should try to be their guide and sounding board as well as coach and cheerleader.
In this issue of the Journal, John and Christopher Higgins present a primer on the background information to use when talking with our patients about starting an exercise program focused on weight loss. They provide useful references that support specific approaches to achieve realistic expectations, and they review and compare various strategies.
I’m sure by March it will again be easier to find a locker at my gym. And I hope by then that my new workout plan will be more scientifically based, as well as a bit more effective. Even data-based hope springs eternal.
Judging by the crowd and the difficulty in finding a locker at my gym on January 1, a lot of people are serious about their 2016 New Year’s resolution to exercise and lose weight. But as most of us have experienced personally and professionally, embarking on a well-intended effort to exercise in the hope of losing weight more often results in frustration than a trip to the store to buy smaller-sized clothes.
The frequent answer to the question “What did the doctor say at your visit?” provides a partial explanation of this phenomenon: “Nothing, just that I should exercise and lose weight” is the usual hackneyed response. Nothing—as in nothing unexpected, nothing significant, and nothing specific was said. It is with this lack of specific advice that I feel many of us let our patients down.
We admonish patients to eat fewer calories, avoid the evil carbs, walk 10,000 steps, ride a bike, use the elliptical, or swim three times a week. There is a concrete but broad nature to these suggestions, but there is also a familiarity and a lack of specificity that leaves patients feeling that there is no science behind them. And the truth is that many of us are not comfortable enough with current data from our exercise physiology colleagues to have a detailed discussion with our patients that pairs their specific goals with an exercise regimen and diet most likely to be beneficial. We may fear sounding like the morning talk show doctors, offering sound bites instead of engaging in an evidence-based dialogue with our patients.
Many of our patients cannot afford a personal trainer to guide and cajole them through a successful regimen—assuming that they, or we, can separate myth and fact and choose an appropriate trainer. We should try to be their guide and sounding board as well as coach and cheerleader.
In this issue of the Journal, John and Christopher Higgins present a primer on the background information to use when talking with our patients about starting an exercise program focused on weight loss. They provide useful references that support specific approaches to achieve realistic expectations, and they review and compare various strategies.
I’m sure by March it will again be easier to find a locker at my gym. And I hope by then that my new workout plan will be more scientifically based, as well as a bit more effective. Even data-based hope springs eternal.
Stop blaming ‘demons’ for bizarre delusions or behavior!
That expression is a residue of the absurd belief during the Middle Ages that mental illness is caused by evil spirits—that justified burning the afflicted person at the stake. (Remember Joan of Arc?)
This ignorant, even maliciously unscientific, portrayal of psychiatric symptoms is an appalling disservice to all our patients who struggle with a potentially disabling neuropsychiatric disorder. Regrettably, some religious entities still propagate the fallacy of possession by an evil spirit and call for exorcism of bizarre behaviors sometimes associated with psychosis.1 What is really needed is an exorcism of unscientific and harmful misconceptions that mental illness is the nefarious work of Beelzebub or Lucifer.
Strange manifestations beget weird explanations
I can understand how ignorance about the neurologic basis of unusual delusions and behavior can trigger absurd religious explanations for their cause. Sometimes, brain pathology can have strange clinical manifestations that are beyond the ken of the average layperson, which invites metaphysical, religious, or philosophical explanation. Here are examples of neuropsychiatric symptoms in the category of “very unusual” that might summon a demonic malfeasance.
Delusion of possession or alien control. Some people complain of being possessed2; delusional people who have a strong religious background might believe they are possessed by Satan himself. Some of my patients with psychotic depression believe this, expressing great guilt and anguish about being doomed to go to Hell.
Alternately, patients with schizophrenia often think they are under the control of an “alien force” that shapes their behavior, feelings, and thoughts (a Schneiderian first-rank symptom). In a 2012 editorial, I proposed that this “alien intruder” is the unintegrated right hemispheric consciousness,3 and that disintegration of the 200 million inter-hemispheric white matter fibers of the corpus callosum might be the cause of the loss of integration of the right hemisphere into the dominant left hemisphere.
Some people attribute external control on their lives to a government agency, a foreign country, or a spiteful neighbor; others believe it is the work of evil spirits. Whereas the foundation of the delusion is brain pathology, the content of the delusion is colored by the affected person’s cultural and religious background.
Apotemnophilia. A neurologic disorder that manifests in a bizarre clinical symptom that invites faulty explanation: A person demands amputation of a leg because “it doesn’t belong to my body.”4 The cause of this strange and confusing disorder has been misinterpreted as a paraphilia, a desire by the affected person to achieve greater sexual satisfaction by having a stump. It was first reported in the September/October 1972 issue of the magazine Penthouse, where it was described as the motivation to heighten one’s sexual appeal because stumps can be sexually exciting to their partners.
It took many years of neurologic research to demonstrate that apotemnophilia is caused by pathology in the parietal lobe, where the physical representation of the body is located. Incomplete neurodevelopment of the parietal lobe can cause a person to fail to recognize a leg as a “legitimate” part of his body, and he (she) then desperately seeks amputation of the so-called alien limb (see the description of xenomelia below) that is attached to his body.
When an affected person is asked to delineate the borders of an alien limb, he draws a line on the skin at the precise border between the alien limb and the rest of his body—where the amputation should take place. Requests for surgical amputation were adamantly denied when the disorder was thought to be a weird sexual practice, but elective amputation in the context of neuropsychopathology is seriously debated now—and has, in fact, been reported.5 The term “body impaired integrity disorder” has been proposed, but neurologists consider the disorder an example of xenomelia.
Xenomelia (‘alien limb syndrome’). An odd neurologic disorder produced by brain pathology, in which a person has a sense of estrangement about 1 or more limbs.5 The disorder can be caused by a neurologic lesion such as tumor, Creutzfeldt-Jakob disease, hereditary diffuse leukoencephalopathy, demyelinating disease, progressive dementia, corpus callosotomy, intracerebral hemorrhage, or thalamic degeneration.6
So-called “alien hand syndrome,” or asomatognosia, is a widely recognized example of xenomelia, and is associated with medial frontal lobe damage.
Another variant of xenomelia is somatoparaphrenia, unawareness of a part of one’s body.7
Cotard syndrome. A nihilistic delusion of the nonexistence or dissolution of a body part; in extreme form, the delusion of being dead or nonexistent.8 The syndrome sometimes occurs in the setting of severe depression. Research has shown an association with atrophy of the insula,9 which is responsible for internal proprioception (interoception).
Delusional misidentification syndrome. A set of neuropsychiatric conditions in which a person misidentifies people, places, objects, or events10:
- Capgras syndrome (one perceives a familiar person as an imposter)
- Fregoli syndrome (one perceives that a familiar person is repeatedly disguised to change appearance)
- intermetamorphosis (one perceives that a person changes his external appearance and personality or identity)
- lycanthropy (one delusionally misidentifies one’s self as an animal—eg, a wolf, rabbit, or snake, and behaves accordingly)
- Ekbom syndrome (delusional belief of being infested with parasites )
- delusion of hermaphroditism (one has merged in the same body with another person of the opposite sex)
- delusion of sexual transformation (one has changed to the opposite sex)
- delusion of being the Antichrist.
Delusional misidentification syndrome can develop after the onset of focal or diffuse brain pathology, such as right hemispheric stroke, multiple sclerosis, hyperparathyroidism, traumatic brain injury, dementia, and schizophrenia. In several studies, researchers have reported an increased risk of violence in delusional misidentification syndromes.11
Neurological, not diabolical!
A disruption in brain anatomy, neurodevelopment, or circuitry/interconnectivity can produce odd beliefs and bizarre behavior that might prompt a lay observer to believe that a demon or an evil spirt is responsible for the incomprehensible symptoms. I have one response to the “blame-the-devil” proponents: It’s the brain pathology, stupid!
1. Irmak MK. Schizophrenia or possession? J Relig Health. 2014;53(3):773-777.
2. Goff DC, Brotman AW, Kindlon D, et al. The delusion of possession in chronically psychotic patients. J Nerv Ment Dis. 1991;179(9):567-571.
3. Nasrallah HA. Impaired mental proprioception in schizophrenia. Current Psychiatry. 2012;11(8):4-5.
4. Brang D, McGeoch PD, Ramachandran VS. Apotemnophilia: a neurological disorder. Neuroreport. 2008;19(13):1305-1306.
5. McGeoch PD, Brang D, Song T, et al. Xenomelia: a new right parietal lobe syndrome. J Neurol Neurosurg Psychiatry. 2011;82(12):1314-1319.
6. Graff-Radford J, Rubin MN, Jones DT, et al. The alien limb phenomenon. J Neurol. 2013;260(7):1880-1888.
7. Feinberg TE, Venneri A, Simone AM, et al. The neuroanatomy of asomatognosia and somatoparaphrenia. J Neurol Neurosurg Psychiatry. 2010;81(3):276-281.
8. Ramirez-Bermudez J, Aguilar-Venegas LC, Crail- Melendez D, et al. Cotard syndrome in neurological and psychiatric patients. J Neuropsychiatry Clin Neurosci. 2010;22(4):409-416.
9. Chatterjee SS, Mitra S. “I do not exist”-Cotard syndrome in insular cortex atrophy. Biol Psychiatry. 2015;77(11):e52-e53.
10. Cipriani G, Vedovello M, Ulivi M, et al. Delusional misidentification syndromes and dementia: a border zone between neurology and psychiatry. Am J Alzheimers Dis Other Demen. 2013;28(7):671-678.
11. Klein CA, Hirachan S. The masks of identities: who’s who? Delusional misidentification syndromes. J Am Acad Psychiatry Law. 2014;42(3):369-378.
That expression is a residue of the absurd belief during the Middle Ages that mental illness is caused by evil spirits—that justified burning the afflicted person at the stake. (Remember Joan of Arc?)
This ignorant, even maliciously unscientific, portrayal of psychiatric symptoms is an appalling disservice to all our patients who struggle with a potentially disabling neuropsychiatric disorder. Regrettably, some religious entities still propagate the fallacy of possession by an evil spirit and call for exorcism of bizarre behaviors sometimes associated with psychosis.1 What is really needed is an exorcism of unscientific and harmful misconceptions that mental illness is the nefarious work of Beelzebub or Lucifer.
Strange manifestations beget weird explanations
I can understand how ignorance about the neurologic basis of unusual delusions and behavior can trigger absurd religious explanations for their cause. Sometimes, brain pathology can have strange clinical manifestations that are beyond the ken of the average layperson, which invites metaphysical, religious, or philosophical explanation. Here are examples of neuropsychiatric symptoms in the category of “very unusual” that might summon a demonic malfeasance.
Delusion of possession or alien control. Some people complain of being possessed2; delusional people who have a strong religious background might believe they are possessed by Satan himself. Some of my patients with psychotic depression believe this, expressing great guilt and anguish about being doomed to go to Hell.
Alternately, patients with schizophrenia often think they are under the control of an “alien force” that shapes their behavior, feelings, and thoughts (a Schneiderian first-rank symptom). In a 2012 editorial, I proposed that this “alien intruder” is the unintegrated right hemispheric consciousness,3 and that disintegration of the 200 million inter-hemispheric white matter fibers of the corpus callosum might be the cause of the loss of integration of the right hemisphere into the dominant left hemisphere.
Some people attribute external control on their lives to a government agency, a foreign country, or a spiteful neighbor; others believe it is the work of evil spirits. Whereas the foundation of the delusion is brain pathology, the content of the delusion is colored by the affected person’s cultural and religious background.
Apotemnophilia. A neurologic disorder that manifests in a bizarre clinical symptom that invites faulty explanation: A person demands amputation of a leg because “it doesn’t belong to my body.”4 The cause of this strange and confusing disorder has been misinterpreted as a paraphilia, a desire by the affected person to achieve greater sexual satisfaction by having a stump. It was first reported in the September/October 1972 issue of the magazine Penthouse, where it was described as the motivation to heighten one’s sexual appeal because stumps can be sexually exciting to their partners.
It took many years of neurologic research to demonstrate that apotemnophilia is caused by pathology in the parietal lobe, where the physical representation of the body is located. Incomplete neurodevelopment of the parietal lobe can cause a person to fail to recognize a leg as a “legitimate” part of his body, and he (she) then desperately seeks amputation of the so-called alien limb (see the description of xenomelia below) that is attached to his body.
When an affected person is asked to delineate the borders of an alien limb, he draws a line on the skin at the precise border between the alien limb and the rest of his body—where the amputation should take place. Requests for surgical amputation were adamantly denied when the disorder was thought to be a weird sexual practice, but elective amputation in the context of neuropsychopathology is seriously debated now—and has, in fact, been reported.5 The term “body impaired integrity disorder” has been proposed, but neurologists consider the disorder an example of xenomelia.
Xenomelia (‘alien limb syndrome’). An odd neurologic disorder produced by brain pathology, in which a person has a sense of estrangement about 1 or more limbs.5 The disorder can be caused by a neurologic lesion such as tumor, Creutzfeldt-Jakob disease, hereditary diffuse leukoencephalopathy, demyelinating disease, progressive dementia, corpus callosotomy, intracerebral hemorrhage, or thalamic degeneration.6
So-called “alien hand syndrome,” or asomatognosia, is a widely recognized example of xenomelia, and is associated with medial frontal lobe damage.
Another variant of xenomelia is somatoparaphrenia, unawareness of a part of one’s body.7
Cotard syndrome. A nihilistic delusion of the nonexistence or dissolution of a body part; in extreme form, the delusion of being dead or nonexistent.8 The syndrome sometimes occurs in the setting of severe depression. Research has shown an association with atrophy of the insula,9 which is responsible for internal proprioception (interoception).
Delusional misidentification syndrome. A set of neuropsychiatric conditions in which a person misidentifies people, places, objects, or events10:
- Capgras syndrome (one perceives a familiar person as an imposter)
- Fregoli syndrome (one perceives that a familiar person is repeatedly disguised to change appearance)
- intermetamorphosis (one perceives that a person changes his external appearance and personality or identity)
- lycanthropy (one delusionally misidentifies one’s self as an animal—eg, a wolf, rabbit, or snake, and behaves accordingly)
- Ekbom syndrome (delusional belief of being infested with parasites )
- delusion of hermaphroditism (one has merged in the same body with another person of the opposite sex)
- delusion of sexual transformation (one has changed to the opposite sex)
- delusion of being the Antichrist.
Delusional misidentification syndrome can develop after the onset of focal or diffuse brain pathology, such as right hemispheric stroke, multiple sclerosis, hyperparathyroidism, traumatic brain injury, dementia, and schizophrenia. In several studies, researchers have reported an increased risk of violence in delusional misidentification syndromes.11
Neurological, not diabolical!
A disruption in brain anatomy, neurodevelopment, or circuitry/interconnectivity can produce odd beliefs and bizarre behavior that might prompt a lay observer to believe that a demon or an evil spirt is responsible for the incomprehensible symptoms. I have one response to the “blame-the-devil” proponents: It’s the brain pathology, stupid!
That expression is a residue of the absurd belief during the Middle Ages that mental illness is caused by evil spirits—that justified burning the afflicted person at the stake. (Remember Joan of Arc?)
This ignorant, even maliciously unscientific, portrayal of psychiatric symptoms is an appalling disservice to all our patients who struggle with a potentially disabling neuropsychiatric disorder. Regrettably, some religious entities still propagate the fallacy of possession by an evil spirit and call for exorcism of bizarre behaviors sometimes associated with psychosis.1 What is really needed is an exorcism of unscientific and harmful misconceptions that mental illness is the nefarious work of Beelzebub or Lucifer.
Strange manifestations beget weird explanations
I can understand how ignorance about the neurologic basis of unusual delusions and behavior can trigger absurd religious explanations for their cause. Sometimes, brain pathology can have strange clinical manifestations that are beyond the ken of the average layperson, which invites metaphysical, religious, or philosophical explanation. Here are examples of neuropsychiatric symptoms in the category of “very unusual” that might summon a demonic malfeasance.
Delusion of possession or alien control. Some people complain of being possessed2; delusional people who have a strong religious background might believe they are possessed by Satan himself. Some of my patients with psychotic depression believe this, expressing great guilt and anguish about being doomed to go to Hell.
Alternately, patients with schizophrenia often think they are under the control of an “alien force” that shapes their behavior, feelings, and thoughts (a Schneiderian first-rank symptom). In a 2012 editorial, I proposed that this “alien intruder” is the unintegrated right hemispheric consciousness,3 and that disintegration of the 200 million inter-hemispheric white matter fibers of the corpus callosum might be the cause of the loss of integration of the right hemisphere into the dominant left hemisphere.
Some people attribute external control on their lives to a government agency, a foreign country, or a spiteful neighbor; others believe it is the work of evil spirits. Whereas the foundation of the delusion is brain pathology, the content of the delusion is colored by the affected person’s cultural and religious background.
Apotemnophilia. A neurologic disorder that manifests in a bizarre clinical symptom that invites faulty explanation: A person demands amputation of a leg because “it doesn’t belong to my body.”4 The cause of this strange and confusing disorder has been misinterpreted as a paraphilia, a desire by the affected person to achieve greater sexual satisfaction by having a stump. It was first reported in the September/October 1972 issue of the magazine Penthouse, where it was described as the motivation to heighten one’s sexual appeal because stumps can be sexually exciting to their partners.
It took many years of neurologic research to demonstrate that apotemnophilia is caused by pathology in the parietal lobe, where the physical representation of the body is located. Incomplete neurodevelopment of the parietal lobe can cause a person to fail to recognize a leg as a “legitimate” part of his body, and he (she) then desperately seeks amputation of the so-called alien limb (see the description of xenomelia below) that is attached to his body.
When an affected person is asked to delineate the borders of an alien limb, he draws a line on the skin at the precise border between the alien limb and the rest of his body—where the amputation should take place. Requests for surgical amputation were adamantly denied when the disorder was thought to be a weird sexual practice, but elective amputation in the context of neuropsychopathology is seriously debated now—and has, in fact, been reported.5 The term “body impaired integrity disorder” has been proposed, but neurologists consider the disorder an example of xenomelia.
Xenomelia (‘alien limb syndrome’). An odd neurologic disorder produced by brain pathology, in which a person has a sense of estrangement about 1 or more limbs.5 The disorder can be caused by a neurologic lesion such as tumor, Creutzfeldt-Jakob disease, hereditary diffuse leukoencephalopathy, demyelinating disease, progressive dementia, corpus callosotomy, intracerebral hemorrhage, or thalamic degeneration.6
So-called “alien hand syndrome,” or asomatognosia, is a widely recognized example of xenomelia, and is associated with medial frontal lobe damage.
Another variant of xenomelia is somatoparaphrenia, unawareness of a part of one’s body.7
Cotard syndrome. A nihilistic delusion of the nonexistence or dissolution of a body part; in extreme form, the delusion of being dead or nonexistent.8 The syndrome sometimes occurs in the setting of severe depression. Research has shown an association with atrophy of the insula,9 which is responsible for internal proprioception (interoception).
Delusional misidentification syndrome. A set of neuropsychiatric conditions in which a person misidentifies people, places, objects, or events10:
- Capgras syndrome (one perceives a familiar person as an imposter)
- Fregoli syndrome (one perceives that a familiar person is repeatedly disguised to change appearance)
- intermetamorphosis (one perceives that a person changes his external appearance and personality or identity)
- lycanthropy (one delusionally misidentifies one’s self as an animal—eg, a wolf, rabbit, or snake, and behaves accordingly)
- Ekbom syndrome (delusional belief of being infested with parasites )
- delusion of hermaphroditism (one has merged in the same body with another person of the opposite sex)
- delusion of sexual transformation (one has changed to the opposite sex)
- delusion of being the Antichrist.
Delusional misidentification syndrome can develop after the onset of focal or diffuse brain pathology, such as right hemispheric stroke, multiple sclerosis, hyperparathyroidism, traumatic brain injury, dementia, and schizophrenia. In several studies, researchers have reported an increased risk of violence in delusional misidentification syndromes.11
Neurological, not diabolical!
A disruption in brain anatomy, neurodevelopment, or circuitry/interconnectivity can produce odd beliefs and bizarre behavior that might prompt a lay observer to believe that a demon or an evil spirt is responsible for the incomprehensible symptoms. I have one response to the “blame-the-devil” proponents: It’s the brain pathology, stupid!
1. Irmak MK. Schizophrenia or possession? J Relig Health. 2014;53(3):773-777.
2. Goff DC, Brotman AW, Kindlon D, et al. The delusion of possession in chronically psychotic patients. J Nerv Ment Dis. 1991;179(9):567-571.
3. Nasrallah HA. Impaired mental proprioception in schizophrenia. Current Psychiatry. 2012;11(8):4-5.
4. Brang D, McGeoch PD, Ramachandran VS. Apotemnophilia: a neurological disorder. Neuroreport. 2008;19(13):1305-1306.
5. McGeoch PD, Brang D, Song T, et al. Xenomelia: a new right parietal lobe syndrome. J Neurol Neurosurg Psychiatry. 2011;82(12):1314-1319.
6. Graff-Radford J, Rubin MN, Jones DT, et al. The alien limb phenomenon. J Neurol. 2013;260(7):1880-1888.
7. Feinberg TE, Venneri A, Simone AM, et al. The neuroanatomy of asomatognosia and somatoparaphrenia. J Neurol Neurosurg Psychiatry. 2010;81(3):276-281.
8. Ramirez-Bermudez J, Aguilar-Venegas LC, Crail- Melendez D, et al. Cotard syndrome in neurological and psychiatric patients. J Neuropsychiatry Clin Neurosci. 2010;22(4):409-416.
9. Chatterjee SS, Mitra S. “I do not exist”-Cotard syndrome in insular cortex atrophy. Biol Psychiatry. 2015;77(11):e52-e53.
10. Cipriani G, Vedovello M, Ulivi M, et al. Delusional misidentification syndromes and dementia: a border zone between neurology and psychiatry. Am J Alzheimers Dis Other Demen. 2013;28(7):671-678.
11. Klein CA, Hirachan S. The masks of identities: who’s who? Delusional misidentification syndromes. J Am Acad Psychiatry Law. 2014;42(3):369-378.
1. Irmak MK. Schizophrenia or possession? J Relig Health. 2014;53(3):773-777.
2. Goff DC, Brotman AW, Kindlon D, et al. The delusion of possession in chronically psychotic patients. J Nerv Ment Dis. 1991;179(9):567-571.
3. Nasrallah HA. Impaired mental proprioception in schizophrenia. Current Psychiatry. 2012;11(8):4-5.
4. Brang D, McGeoch PD, Ramachandran VS. Apotemnophilia: a neurological disorder. Neuroreport. 2008;19(13):1305-1306.
5. McGeoch PD, Brang D, Song T, et al. Xenomelia: a new right parietal lobe syndrome. J Neurol Neurosurg Psychiatry. 2011;82(12):1314-1319.
6. Graff-Radford J, Rubin MN, Jones DT, et al. The alien limb phenomenon. J Neurol. 2013;260(7):1880-1888.
7. Feinberg TE, Venneri A, Simone AM, et al. The neuroanatomy of asomatognosia and somatoparaphrenia. J Neurol Neurosurg Psychiatry. 2010;81(3):276-281.
8. Ramirez-Bermudez J, Aguilar-Venegas LC, Crail- Melendez D, et al. Cotard syndrome in neurological and psychiatric patients. J Neuropsychiatry Clin Neurosci. 2010;22(4):409-416.
9. Chatterjee SS, Mitra S. “I do not exist”-Cotard syndrome in insular cortex atrophy. Biol Psychiatry. 2015;77(11):e52-e53.
10. Cipriani G, Vedovello M, Ulivi M, et al. Delusional misidentification syndromes and dementia: a border zone between neurology and psychiatry. Am J Alzheimers Dis Other Demen. 2013;28(7):671-678.
11. Klein CA, Hirachan S. The masks of identities: who’s who? Delusional misidentification syndromes. J Am Acad Psychiatry Law. 2014;42(3):369-378.
Cannabinoids in cancer treatment settings
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.
Click on the PDF icon at the top of this introduction to read the full article.