User login
A welcome addition
On behalf of the SVS Publications Committee I am delighted to announce the appointment of Malachi Sheahan, III, M.D., as Associate Medical Editor of Vascular Specialist. I am sure readers will realize this official newspaper of the Society for Vascular Surgery has grown substantially over the last few years. With the help of generous advertisers we now “publish” Vascular Specialist not only in print but in multiple formats. These include electronic versions on the Web, Facebook, and Twitter, as well as mobile versions for smartphones and tablets. The print version is now published monthly and most often as 20 pages.
Further, www.vascularspecialistonline.com includes many articles not found in the print edition. Articles can be searched based on subject and author and the print version can be seen in PDF version. The web version also allows readers to answer polls posed in the print edition. These are archived for review.
A recent survey of 250 members of the SVS confirmed that Vascular Specialist is now the most widely read vascular news publication. More importantly, it was considered by far the most authoritative.
This expansion requires a significant expenditure of time by the Associate Editors and the Medical Editor in particular. Accordingly, it is essential that the medical editorial staff be supplemented by an Associate Medical Editor. This will also allow the smooth transition when the Medical Editor’s appointment terminates.
Mal has already proven to be an excellent writer, and his commitment to the SVS, resident/fellow training, and his sense of humor make him an outstanding choice to assume this position. Mal completed his vascular surgery training at the Beth Israel Deaconess program in 2003. Shortly thereafter he joined the faculty at the Louisiana State University Health Sciences Center in New Orleans. He currently serves as the program director for both their integrated and independent vascular training programs. In 2014, he was named the Claude C. Craighead Jr. Professor and Chair of the Division of Vascular and Endovascular Surgery.
I have previously requested that all members consider themselves not only readers but also contributors. Once again I encourage you to send Mal and me opinion pieces or letters to the editor. We also welcome Tips and Tricks. These can be quite short and do not require any references. They are meant to highlight a technique that you have found to be helpful in performing an open or endovascular procedure. One or two photos often help the piece but they must be in .jpg format and at least 124kb. If you are so inclined we will also print your photo and affiliation alongside. However, we regret you cannot submit your high school photo or substitute one of Tom Cruise or Beyoncé! Don’t worry that you may not be a Pulitzer Prize journalist since we have professional writers who will tidy up your writing as necessary. We also welcome suggestions about news items that we may have missed or overlooked. These may be news items from the lay press or a manuscript you read in a journal other than the JVS. Again, our professional reporters will turn the item into a news article. You can also send us a comment and, if appropriate, we will print it as a sidebar to the article.
Further, we encourage news about your comings and goings which we can publish in the “From the community” section. Executive members of the various regional Societies can also utilize this section to bring us news about their Societies activities.
In order to keep the commentaries fresh, we do need to rotate the members of the editorial board. So, if you would like to be considered for such a position, please send us a short biography and your area of interest. This could be related to a disease process, treatment, or socioeconomic issues. Associate Editors will be required to read short news items about 12 times a year and to provide commentary when requested. Even if you are not selected we may occasionally turn to you to write a comment on a news item related to your stated interest.
The SVS wants Vascular Specialist to be YOUR newspaper. Please help us achieve that goal as we continue to make Vascular Specialist the most read and respected supplier of news and opinions about vascular surgery and vascular surgeons.
You can contact me at [email protected] and Dr. Sheahan at [email protected].
On behalf of the SVS Publications Committee I am delighted to announce the appointment of Malachi Sheahan, III, M.D., as Associate Medical Editor of Vascular Specialist. I am sure readers will realize this official newspaper of the Society for Vascular Surgery has grown substantially over the last few years. With the help of generous advertisers we now “publish” Vascular Specialist not only in print but in multiple formats. These include electronic versions on the Web, Facebook, and Twitter, as well as mobile versions for smartphones and tablets. The print version is now published monthly and most often as 20 pages.
Further, www.vascularspecialistonline.com includes many articles not found in the print edition. Articles can be searched based on subject and author and the print version can be seen in PDF version. The web version also allows readers to answer polls posed in the print edition. These are archived for review.
A recent survey of 250 members of the SVS confirmed that Vascular Specialist is now the most widely read vascular news publication. More importantly, it was considered by far the most authoritative.
This expansion requires a significant expenditure of time by the Associate Editors and the Medical Editor in particular. Accordingly, it is essential that the medical editorial staff be supplemented by an Associate Medical Editor. This will also allow the smooth transition when the Medical Editor’s appointment terminates.
Mal has already proven to be an excellent writer, and his commitment to the SVS, resident/fellow training, and his sense of humor make him an outstanding choice to assume this position. Mal completed his vascular surgery training at the Beth Israel Deaconess program in 2003. Shortly thereafter he joined the faculty at the Louisiana State University Health Sciences Center in New Orleans. He currently serves as the program director for both their integrated and independent vascular training programs. In 2014, he was named the Claude C. Craighead Jr. Professor and Chair of the Division of Vascular and Endovascular Surgery.
I have previously requested that all members consider themselves not only readers but also contributors. Once again I encourage you to send Mal and me opinion pieces or letters to the editor. We also welcome Tips and Tricks. These can be quite short and do not require any references. They are meant to highlight a technique that you have found to be helpful in performing an open or endovascular procedure. One or two photos often help the piece but they must be in .jpg format and at least 124kb. If you are so inclined we will also print your photo and affiliation alongside. However, we regret you cannot submit your high school photo or substitute one of Tom Cruise or Beyoncé! Don’t worry that you may not be a Pulitzer Prize journalist since we have professional writers who will tidy up your writing as necessary. We also welcome suggestions about news items that we may have missed or overlooked. These may be news items from the lay press or a manuscript you read in a journal other than the JVS. Again, our professional reporters will turn the item into a news article. You can also send us a comment and, if appropriate, we will print it as a sidebar to the article.
Further, we encourage news about your comings and goings which we can publish in the “From the community” section. Executive members of the various regional Societies can also utilize this section to bring us news about their Societies activities.
In order to keep the commentaries fresh, we do need to rotate the members of the editorial board. So, if you would like to be considered for such a position, please send us a short biography and your area of interest. This could be related to a disease process, treatment, or socioeconomic issues. Associate Editors will be required to read short news items about 12 times a year and to provide commentary when requested. Even if you are not selected we may occasionally turn to you to write a comment on a news item related to your stated interest.
The SVS wants Vascular Specialist to be YOUR newspaper. Please help us achieve that goal as we continue to make Vascular Specialist the most read and respected supplier of news and opinions about vascular surgery and vascular surgeons.
You can contact me at [email protected] and Dr. Sheahan at [email protected].
On behalf of the SVS Publications Committee I am delighted to announce the appointment of Malachi Sheahan, III, M.D., as Associate Medical Editor of Vascular Specialist. I am sure readers will realize this official newspaper of the Society for Vascular Surgery has grown substantially over the last few years. With the help of generous advertisers we now “publish” Vascular Specialist not only in print but in multiple formats. These include electronic versions on the Web, Facebook, and Twitter, as well as mobile versions for smartphones and tablets. The print version is now published monthly and most often as 20 pages.
Further, www.vascularspecialistonline.com includes many articles not found in the print edition. Articles can be searched based on subject and author and the print version can be seen in PDF version. The web version also allows readers to answer polls posed in the print edition. These are archived for review.
A recent survey of 250 members of the SVS confirmed that Vascular Specialist is now the most widely read vascular news publication. More importantly, it was considered by far the most authoritative.
This expansion requires a significant expenditure of time by the Associate Editors and the Medical Editor in particular. Accordingly, it is essential that the medical editorial staff be supplemented by an Associate Medical Editor. This will also allow the smooth transition when the Medical Editor’s appointment terminates.
Mal has already proven to be an excellent writer, and his commitment to the SVS, resident/fellow training, and his sense of humor make him an outstanding choice to assume this position. Mal completed his vascular surgery training at the Beth Israel Deaconess program in 2003. Shortly thereafter he joined the faculty at the Louisiana State University Health Sciences Center in New Orleans. He currently serves as the program director for both their integrated and independent vascular training programs. In 2014, he was named the Claude C. Craighead Jr. Professor and Chair of the Division of Vascular and Endovascular Surgery.
I have previously requested that all members consider themselves not only readers but also contributors. Once again I encourage you to send Mal and me opinion pieces or letters to the editor. We also welcome Tips and Tricks. These can be quite short and do not require any references. They are meant to highlight a technique that you have found to be helpful in performing an open or endovascular procedure. One or two photos often help the piece but they must be in .jpg format and at least 124kb. If you are so inclined we will also print your photo and affiliation alongside. However, we regret you cannot submit your high school photo or substitute one of Tom Cruise or Beyoncé! Don’t worry that you may not be a Pulitzer Prize journalist since we have professional writers who will tidy up your writing as necessary. We also welcome suggestions about news items that we may have missed or overlooked. These may be news items from the lay press or a manuscript you read in a journal other than the JVS. Again, our professional reporters will turn the item into a news article. You can also send us a comment and, if appropriate, we will print it as a sidebar to the article.
Further, we encourage news about your comings and goings which we can publish in the “From the community” section. Executive members of the various regional Societies can also utilize this section to bring us news about their Societies activities.
In order to keep the commentaries fresh, we do need to rotate the members of the editorial board. So, if you would like to be considered for such a position, please send us a short biography and your area of interest. This could be related to a disease process, treatment, or socioeconomic issues. Associate Editors will be required to read short news items about 12 times a year and to provide commentary when requested. Even if you are not selected we may occasionally turn to you to write a comment on a news item related to your stated interest.
The SVS wants Vascular Specialist to be YOUR newspaper. Please help us achieve that goal as we continue to make Vascular Specialist the most read and respected supplier of news and opinions about vascular surgery and vascular surgeons.
You can contact me at [email protected] and Dr. Sheahan at [email protected].
Stop using rectal misoprostol for the treatment of postpartum hemorrhage caused by uterine atony
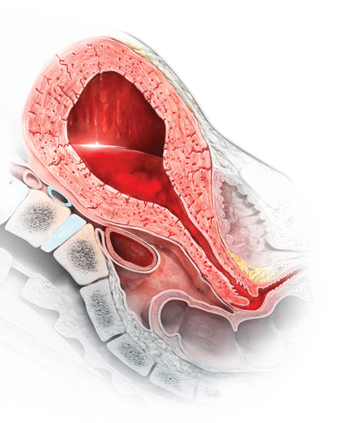
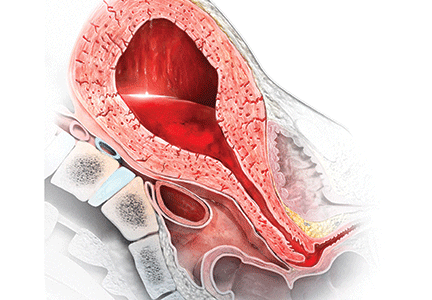
Most authorities recommend that, following delivery, all women should receive a uterotonic medication to reduce the risk of postpartum hemorrhage (PPH).1 In the United States, the preferred uterotonic for this preventive effort is oxytocin—a low-cost, highly effective agent that typically is administered as an intravenous (IV) infusion or intramuscular (IM) injection. Unfortunately, even with the universal administration of oxytocin in the third stage of labor, PPH occurs in about 3% of vaginal deliveries.
A key decision in treating a PPH due to uterine atony is treatment with an optimal uterotonic. The options include:
- additional oxytocin
- carboprost tromethamine(Hemabate)
- methylergonovine (Methergine)
- misoprostol.
Many obstetricians choose rectal misoprostol alone or in combination with oxytocin as the preferred treatment of PPH. However, evidence from clinical trials and pharmacokinetic studies suggest that rectal misoprostol is not an optimal choice if parenteral uterotonics are available. Here I pre-sent this evidence and urge you to stop the practice of using rectal misoprostol in efforts to manage PPH.
RCTs do not support the use of rectal misoprostolRandomized clinical trials (RCTs) have not demonstrated that misoprostol is superior to oxytocin for the treatment of PPH caused by uterine atony.2 For example, Blum and colleagues studied 31,055 women who received oxytocin (by IV or IM route) at vaginal delivery and observed that 809 (3%) developed a PPH.3 The women who developed PPH were randomly assigned to treatment with misoprostol 800 µg sublingual or oxytocin 40 U in 1,000 mL as an IV infusion over 15 minutes.
Both oxytocin and misoprostol had similar efficacy for controlling bleeding within 20 minutes (90% and 89%, respectively). Fewer women had blood loss of 1,000 mL or greater when treated with oxytocin compared with misoprostol (1% vs 3%, respectively; P = .062). In addition, oxytocin was associated with fewer temperature elevations of 38°C (100.4°F) or above (15% vs 22% for misoprostol, P = .007) and fewer temperature elevations of 40°C (104°F) or above (0.2% vs 1.2% for misoprostol, P = .11).
In another trial, women with a vaginal delivery who were not treated with a uterotonic in the third stage were monitored for the development of a PPH.4 PPH did develop in 1,422 women, who were then randomly assigned to receive oxytocin (10 U IV or IM) plus a placebo tablet or oxytocin plus misoprostol (600 µg sublingual).
Comparing oxytocin alone versus oxytocin plus misoprostol, there was no difference in blood loss of 500 mL or greater after treatment initiation (14% vs 14%). However, 90 minutes following treatment, temperature elevations occurred much more often in the women who received oxytocin plus misoprostol compared with the women who received oxytocin alone (temperature ≥38°C: 58% vs 19%; temperature ≥40°C: 6.8% vs 0.4%).
Bottom line: If you have access to oxytocin, there is no advantage to using misoprostol to treat a PPH due to uterine atony.5
Rectal misoprostol does not achieve optimal circulating concentrations of the drugMisoprostol tablets are formulated for oral administration, not rectal administration. The studies in the TABLE show that rectal administration of misoprostol results in lower circulating concentration of the medication compared to oral, buccal, or vaginal administration.6−8 After rectal administration it takes about 60 minutes to reach the peak circulating concentration of misoprostol.6,7 By contrast, parenteral oxytocin, methylergonovine, and carboprost tromethamine reach peak serum concentration much more quickly after administration.

In a study of misoprostol stimulation of uterine contractility as measured by an intrauterine pressure catheter, buccal administration resulted in higher peak uterine tone than rectal administration (49 vs 31 mm Hg).8 In addition, time to onset of uterine contractility was 41 minutes and 103 minutes, respectively, for buccal and rectal administration.
These studies show that rectal misoprostol is associated with lower serum concentrations, longer time to onset of uterine contraction, and less contractility than buccal administration. The one advantage of rectal administration is that it has a longer duration of action than the oral, buccal, or sublingual routes. In pharmacokinetic comparisons of buccal versus sublingual administration of misoprostol, the sublingual route results in greater peak concentration, which may cause more adverse effects.9,10
Misoprostol is a useful uterotonic if parenteral agents are not available
Worldwide, approximately one maternal death occurs every 7 minutes. Postpartum hemorrhage (PPH) is a common cause of maternal death. Oxytocin, methylergonovine, and carboprost tromethamine should be stored in a refrigerated environment to ensure the stability and bioavailability of the drug. In settings in which reliable refrigeration is not available, misoprostol, a medication that is heat-stable, is often used to prevent and treat PPH.
One approach to preventing PPH is to provide 600 µg of misoprostol to women delivering at home without a skilled birth attendant that they can self- administer after the delivery.1,2 Another approach is to recommend that skilled birth attendants administer misoprostol following the delivery.3
Although I am recommending that we not use rectal misoprostol to treat PPH in the United States, it is clear that misoprostol plays an important role in preventing PPH in countries where parenteral uterotonics are not available. If a clinician in the United States was involved in a home birth complicated by PPH due to uterine atony, and if misoprostol was the only available uterotonic, it would be wise to administer it promptly.
References
- Rajbhandari S, Hodgins S, Sanghvi H, McPherson R, Pradhan YV, Baqui AH; Misoprostol Study Group. Expanding uterotonic protection following childbirth through community-based distribution of misoprostol: operations research study in Nepal. Int J Gynaecol Obstet. 2010;108(3):282–288.
- Sanghvi H, Ansari N, Prata NJ, Gibson H, Ehsan AT, Smith JM. Prevention of postpartum hemorrhage at home birth in Afghanistan. Int J Gynaecol Obstet. 2010;108(3):276–281.
- Prata N, Mbaruku G, Campbell M, Potts M, Vahidnia F. Controlling postpartum hemorrhage after home births in Tanzania. Int J Gynaecol Obstet. 2005;90(1):51–55.
Prioritize oxytocin, methergine, and carboprost tromethamineWhen treating PPH, administration of oxytocin, methylergonovine, or carboprost tromethamine rapidly provides therapeutic concentration of medication. For oxytocin, 40 U in 1 L, administered at a rate sufficient to control atony, or 10 U IM injection are often effective in controlling bleeding due to atony. Carboprost tromethamine 0.25 mg administered intramuscularly every 15 minutes up to 8 doses provides an excellent second-line agent. Carboprost tromethamine is contraindicated for women with asthma.
Methylergonovine 0.2 mg administered intramuscularly only can be given every 2 to 4 hours. Consequently, because time is of the essence in managing a severe PPH, it is unusual to be able to administer more than one dose of the agent during the course of treatment. Methylergonovine is contraindicated for women with hypertension.
There is scant evidence that misoprostol is more effective than oxytocin, and misoprostol clearly causes a higher rate of elevated temperature than any of the parenteral uterotonic agents. In your practice stop using rectal misoprostol for the treatment of PPH caused by uterine atony, and prioritize the use of parenteral uterotonics.

Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Westhoff G, Cotter AM, Tolosa JE. Prophylactic oxytocin for the third stage of labour to prevent postpartum hemorrhage. Cochrane Database Syst Rev. 2013;(10):CD001808.
- Gibbons KJ, Albright CM, Rouse DJ. Postpartum hemorrhage in the developed world: whither misoprostol? Am J Obstet Gynecol. 2013;208(3):181−183.
- Blum J, Winikoff B, Raghavan S, et al. Treatment of postpartum hemorrhage with sublingual misoprostol versus oxytocin in women receiving prophylactic oxytocin: a double-blind, randomised, non-inferiority trial. Lancet. 2010;375(9710):217−223.
- Widmer M, Blum J, Hofmeyr GJ, et al. Misoprostolas an adjunct to standard uterotonics for treatment of post-partum haemorrhage: a multicentre, double-blind randomised trial. Lancet. 2010;375(9728):1808−1813.
- Weeks A. The prevention and treatment of postpartum hemorrhage: what do we know and where do we go to next? BJOG. 2015;122(2):202−210.
- Khan RU, El-Refaey H. Pharmacokinetics and adverse-effect profile of rectally administered misoprostol in the third stage of labor. Obstet Gynecol. 2003;101(5 pt 1):968−974.
- Khan RU, El-Refaey H, Sharma S, Sooranna D, Stafford M. Oral, rectal and vaginal pharmacokinetics of misoprostol. Obstet Gynecol. 2004;103(5 pt 1):866−870.
- Meckstroth KR, Whitaker AK, Bertisch S, Goldberg AB, Darney PD. Misoprostol administered by epithelial routes: drug absorption and uterine response. Obstet Gynecol. 2006;108(3 pt 1):582−590.
- Schaff EA, DiCenzo R, Fielding SL. Comparison of misoprostol plasma concentrations following buccal and sublingual administration. Contraception. 2005;71(1):22−25.
- Frye LJ, Byrne ME, Winikoff B. A crossover pharmacokinetic study of misoprostol by the oral, sublingual and buccal routes [published online ahead of print April 22, 2016]. Eur J Contracept Reprod Health Care. doi:10.3109/13625187.2016.1168799.

Most authorities recommend that, following delivery, all women should receive a uterotonic medication to reduce the risk of postpartum hemorrhage (PPH).1 In the United States, the preferred uterotonic for this preventive effort is oxytocin—a low-cost, highly effective agent that typically is administered as an intravenous (IV) infusion or intramuscular (IM) injection. Unfortunately, even with the universal administration of oxytocin in the third stage of labor, PPH occurs in about 3% of vaginal deliveries.
A key decision in treating a PPH due to uterine atony is treatment with an optimal uterotonic. The options include:
- additional oxytocin
- carboprost tromethamine(Hemabate)
- methylergonovine (Methergine)
- misoprostol.
Many obstetricians choose rectal misoprostol alone or in combination with oxytocin as the preferred treatment of PPH. However, evidence from clinical trials and pharmacokinetic studies suggest that rectal misoprostol is not an optimal choice if parenteral uterotonics are available. Here I pre-sent this evidence and urge you to stop the practice of using rectal misoprostol in efforts to manage PPH.
RCTs do not support the use of rectal misoprostolRandomized clinical trials (RCTs) have not demonstrated that misoprostol is superior to oxytocin for the treatment of PPH caused by uterine atony.2 For example, Blum and colleagues studied 31,055 women who received oxytocin (by IV or IM route) at vaginal delivery and observed that 809 (3%) developed a PPH.3 The women who developed PPH were randomly assigned to treatment with misoprostol 800 µg sublingual or oxytocin 40 U in 1,000 mL as an IV infusion over 15 minutes.
Both oxytocin and misoprostol had similar efficacy for controlling bleeding within 20 minutes (90% and 89%, respectively). Fewer women had blood loss of 1,000 mL or greater when treated with oxytocin compared with misoprostol (1% vs 3%, respectively; P = .062). In addition, oxytocin was associated with fewer temperature elevations of 38°C (100.4°F) or above (15% vs 22% for misoprostol, P = .007) and fewer temperature elevations of 40°C (104°F) or above (0.2% vs 1.2% for misoprostol, P = .11).
In another trial, women with a vaginal delivery who were not treated with a uterotonic in the third stage were monitored for the development of a PPH.4 PPH did develop in 1,422 women, who were then randomly assigned to receive oxytocin (10 U IV or IM) plus a placebo tablet or oxytocin plus misoprostol (600 µg sublingual).
Comparing oxytocin alone versus oxytocin plus misoprostol, there was no difference in blood loss of 500 mL or greater after treatment initiation (14% vs 14%). However, 90 minutes following treatment, temperature elevations occurred much more often in the women who received oxytocin plus misoprostol compared with the women who received oxytocin alone (temperature ≥38°C: 58% vs 19%; temperature ≥40°C: 6.8% vs 0.4%).
Bottom line: If you have access to oxytocin, there is no advantage to using misoprostol to treat a PPH due to uterine atony.5
Rectal misoprostol does not achieve optimal circulating concentrations of the drugMisoprostol tablets are formulated for oral administration, not rectal administration. The studies in the TABLE show that rectal administration of misoprostol results in lower circulating concentration of the medication compared to oral, buccal, or vaginal administration.6−8 After rectal administration it takes about 60 minutes to reach the peak circulating concentration of misoprostol.6,7 By contrast, parenteral oxytocin, methylergonovine, and carboprost tromethamine reach peak serum concentration much more quickly after administration.

In a study of misoprostol stimulation of uterine contractility as measured by an intrauterine pressure catheter, buccal administration resulted in higher peak uterine tone than rectal administration (49 vs 31 mm Hg).8 In addition, time to onset of uterine contractility was 41 minutes and 103 minutes, respectively, for buccal and rectal administration.
These studies show that rectal misoprostol is associated with lower serum concentrations, longer time to onset of uterine contraction, and less contractility than buccal administration. The one advantage of rectal administration is that it has a longer duration of action than the oral, buccal, or sublingual routes. In pharmacokinetic comparisons of buccal versus sublingual administration of misoprostol, the sublingual route results in greater peak concentration, which may cause more adverse effects.9,10
Misoprostol is a useful uterotonic if parenteral agents are not available
Worldwide, approximately one maternal death occurs every 7 minutes. Postpartum hemorrhage (PPH) is a common cause of maternal death. Oxytocin, methylergonovine, and carboprost tromethamine should be stored in a refrigerated environment to ensure the stability and bioavailability of the drug. In settings in which reliable refrigeration is not available, misoprostol, a medication that is heat-stable, is often used to prevent and treat PPH.
One approach to preventing PPH is to provide 600 µg of misoprostol to women delivering at home without a skilled birth attendant that they can self- administer after the delivery.1,2 Another approach is to recommend that skilled birth attendants administer misoprostol following the delivery.3
Although I am recommending that we not use rectal misoprostol to treat PPH in the United States, it is clear that misoprostol plays an important role in preventing PPH in countries where parenteral uterotonics are not available. If a clinician in the United States was involved in a home birth complicated by PPH due to uterine atony, and if misoprostol was the only available uterotonic, it would be wise to administer it promptly.
References
- Rajbhandari S, Hodgins S, Sanghvi H, McPherson R, Pradhan YV, Baqui AH; Misoprostol Study Group. Expanding uterotonic protection following childbirth through community-based distribution of misoprostol: operations research study in Nepal. Int J Gynaecol Obstet. 2010;108(3):282–288.
- Sanghvi H, Ansari N, Prata NJ, Gibson H, Ehsan AT, Smith JM. Prevention of postpartum hemorrhage at home birth in Afghanistan. Int J Gynaecol Obstet. 2010;108(3):276–281.
- Prata N, Mbaruku G, Campbell M, Potts M, Vahidnia F. Controlling postpartum hemorrhage after home births in Tanzania. Int J Gynaecol Obstet. 2005;90(1):51–55.
Prioritize oxytocin, methergine, and carboprost tromethamineWhen treating PPH, administration of oxytocin, methylergonovine, or carboprost tromethamine rapidly provides therapeutic concentration of medication. For oxytocin, 40 U in 1 L, administered at a rate sufficient to control atony, or 10 U IM injection are often effective in controlling bleeding due to atony. Carboprost tromethamine 0.25 mg administered intramuscularly every 15 minutes up to 8 doses provides an excellent second-line agent. Carboprost tromethamine is contraindicated for women with asthma.
Methylergonovine 0.2 mg administered intramuscularly only can be given every 2 to 4 hours. Consequently, because time is of the essence in managing a severe PPH, it is unusual to be able to administer more than one dose of the agent during the course of treatment. Methylergonovine is contraindicated for women with hypertension.
There is scant evidence that misoprostol is more effective than oxytocin, and misoprostol clearly causes a higher rate of elevated temperature than any of the parenteral uterotonic agents. In your practice stop using rectal misoprostol for the treatment of PPH caused by uterine atony, and prioritize the use of parenteral uterotonics.

Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.

Most authorities recommend that, following delivery, all women should receive a uterotonic medication to reduce the risk of postpartum hemorrhage (PPH).1 In the United States, the preferred uterotonic for this preventive effort is oxytocin—a low-cost, highly effective agent that typically is administered as an intravenous (IV) infusion or intramuscular (IM) injection. Unfortunately, even with the universal administration of oxytocin in the third stage of labor, PPH occurs in about 3% of vaginal deliveries.
A key decision in treating a PPH due to uterine atony is treatment with an optimal uterotonic. The options include:
- additional oxytocin
- carboprost tromethamine(Hemabate)
- methylergonovine (Methergine)
- misoprostol.
Many obstetricians choose rectal misoprostol alone or in combination with oxytocin as the preferred treatment of PPH. However, evidence from clinical trials and pharmacokinetic studies suggest that rectal misoprostol is not an optimal choice if parenteral uterotonics are available. Here I pre-sent this evidence and urge you to stop the practice of using rectal misoprostol in efforts to manage PPH.
RCTs do not support the use of rectal misoprostolRandomized clinical trials (RCTs) have not demonstrated that misoprostol is superior to oxytocin for the treatment of PPH caused by uterine atony.2 For example, Blum and colleagues studied 31,055 women who received oxytocin (by IV or IM route) at vaginal delivery and observed that 809 (3%) developed a PPH.3 The women who developed PPH were randomly assigned to treatment with misoprostol 800 µg sublingual or oxytocin 40 U in 1,000 mL as an IV infusion over 15 minutes.
Both oxytocin and misoprostol had similar efficacy for controlling bleeding within 20 minutes (90% and 89%, respectively). Fewer women had blood loss of 1,000 mL or greater when treated with oxytocin compared with misoprostol (1% vs 3%, respectively; P = .062). In addition, oxytocin was associated with fewer temperature elevations of 38°C (100.4°F) or above (15% vs 22% for misoprostol, P = .007) and fewer temperature elevations of 40°C (104°F) or above (0.2% vs 1.2% for misoprostol, P = .11).
In another trial, women with a vaginal delivery who were not treated with a uterotonic in the third stage were monitored for the development of a PPH.4 PPH did develop in 1,422 women, who were then randomly assigned to receive oxytocin (10 U IV or IM) plus a placebo tablet or oxytocin plus misoprostol (600 µg sublingual).
Comparing oxytocin alone versus oxytocin plus misoprostol, there was no difference in blood loss of 500 mL or greater after treatment initiation (14% vs 14%). However, 90 minutes following treatment, temperature elevations occurred much more often in the women who received oxytocin plus misoprostol compared with the women who received oxytocin alone (temperature ≥38°C: 58% vs 19%; temperature ≥40°C: 6.8% vs 0.4%).
Bottom line: If you have access to oxytocin, there is no advantage to using misoprostol to treat a PPH due to uterine atony.5
Rectal misoprostol does not achieve optimal circulating concentrations of the drugMisoprostol tablets are formulated for oral administration, not rectal administration. The studies in the TABLE show that rectal administration of misoprostol results in lower circulating concentration of the medication compared to oral, buccal, or vaginal administration.6−8 After rectal administration it takes about 60 minutes to reach the peak circulating concentration of misoprostol.6,7 By contrast, parenteral oxytocin, methylergonovine, and carboprost tromethamine reach peak serum concentration much more quickly after administration.

In a study of misoprostol stimulation of uterine contractility as measured by an intrauterine pressure catheter, buccal administration resulted in higher peak uterine tone than rectal administration (49 vs 31 mm Hg).8 In addition, time to onset of uterine contractility was 41 minutes and 103 minutes, respectively, for buccal and rectal administration.
These studies show that rectal misoprostol is associated with lower serum concentrations, longer time to onset of uterine contraction, and less contractility than buccal administration. The one advantage of rectal administration is that it has a longer duration of action than the oral, buccal, or sublingual routes. In pharmacokinetic comparisons of buccal versus sublingual administration of misoprostol, the sublingual route results in greater peak concentration, which may cause more adverse effects.9,10
Misoprostol is a useful uterotonic if parenteral agents are not available
Worldwide, approximately one maternal death occurs every 7 minutes. Postpartum hemorrhage (PPH) is a common cause of maternal death. Oxytocin, methylergonovine, and carboprost tromethamine should be stored in a refrigerated environment to ensure the stability and bioavailability of the drug. In settings in which reliable refrigeration is not available, misoprostol, a medication that is heat-stable, is often used to prevent and treat PPH.
One approach to preventing PPH is to provide 600 µg of misoprostol to women delivering at home without a skilled birth attendant that they can self- administer after the delivery.1,2 Another approach is to recommend that skilled birth attendants administer misoprostol following the delivery.3
Although I am recommending that we not use rectal misoprostol to treat PPH in the United States, it is clear that misoprostol plays an important role in preventing PPH in countries where parenteral uterotonics are not available. If a clinician in the United States was involved in a home birth complicated by PPH due to uterine atony, and if misoprostol was the only available uterotonic, it would be wise to administer it promptly.
References
- Rajbhandari S, Hodgins S, Sanghvi H, McPherson R, Pradhan YV, Baqui AH; Misoprostol Study Group. Expanding uterotonic protection following childbirth through community-based distribution of misoprostol: operations research study in Nepal. Int J Gynaecol Obstet. 2010;108(3):282–288.
- Sanghvi H, Ansari N, Prata NJ, Gibson H, Ehsan AT, Smith JM. Prevention of postpartum hemorrhage at home birth in Afghanistan. Int J Gynaecol Obstet. 2010;108(3):276–281.
- Prata N, Mbaruku G, Campbell M, Potts M, Vahidnia F. Controlling postpartum hemorrhage after home births in Tanzania. Int J Gynaecol Obstet. 2005;90(1):51–55.
Prioritize oxytocin, methergine, and carboprost tromethamineWhen treating PPH, administration of oxytocin, methylergonovine, or carboprost tromethamine rapidly provides therapeutic concentration of medication. For oxytocin, 40 U in 1 L, administered at a rate sufficient to control atony, or 10 U IM injection are often effective in controlling bleeding due to atony. Carboprost tromethamine 0.25 mg administered intramuscularly every 15 minutes up to 8 doses provides an excellent second-line agent. Carboprost tromethamine is contraindicated for women with asthma.
Methylergonovine 0.2 mg administered intramuscularly only can be given every 2 to 4 hours. Consequently, because time is of the essence in managing a severe PPH, it is unusual to be able to administer more than one dose of the agent during the course of treatment. Methylergonovine is contraindicated for women with hypertension.
There is scant evidence that misoprostol is more effective than oxytocin, and misoprostol clearly causes a higher rate of elevated temperature than any of the parenteral uterotonic agents. In your practice stop using rectal misoprostol for the treatment of PPH caused by uterine atony, and prioritize the use of parenteral uterotonics.

Share your thoughts! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
- Westhoff G, Cotter AM, Tolosa JE. Prophylactic oxytocin for the third stage of labour to prevent postpartum hemorrhage. Cochrane Database Syst Rev. 2013;(10):CD001808.
- Gibbons KJ, Albright CM, Rouse DJ. Postpartum hemorrhage in the developed world: whither misoprostol? Am J Obstet Gynecol. 2013;208(3):181−183.
- Blum J, Winikoff B, Raghavan S, et al. Treatment of postpartum hemorrhage with sublingual misoprostol versus oxytocin in women receiving prophylactic oxytocin: a double-blind, randomised, non-inferiority trial. Lancet. 2010;375(9710):217−223.
- Widmer M, Blum J, Hofmeyr GJ, et al. Misoprostolas an adjunct to standard uterotonics for treatment of post-partum haemorrhage: a multicentre, double-blind randomised trial. Lancet. 2010;375(9728):1808−1813.
- Weeks A. The prevention and treatment of postpartum hemorrhage: what do we know and where do we go to next? BJOG. 2015;122(2):202−210.
- Khan RU, El-Refaey H. Pharmacokinetics and adverse-effect profile of rectally administered misoprostol in the third stage of labor. Obstet Gynecol. 2003;101(5 pt 1):968−974.
- Khan RU, El-Refaey H, Sharma S, Sooranna D, Stafford M. Oral, rectal and vaginal pharmacokinetics of misoprostol. Obstet Gynecol. 2004;103(5 pt 1):866−870.
- Meckstroth KR, Whitaker AK, Bertisch S, Goldberg AB, Darney PD. Misoprostol administered by epithelial routes: drug absorption and uterine response. Obstet Gynecol. 2006;108(3 pt 1):582−590.
- Schaff EA, DiCenzo R, Fielding SL. Comparison of misoprostol plasma concentrations following buccal and sublingual administration. Contraception. 2005;71(1):22−25.
- Frye LJ, Byrne ME, Winikoff B. A crossover pharmacokinetic study of misoprostol by the oral, sublingual and buccal routes [published online ahead of print April 22, 2016]. Eur J Contracept Reprod Health Care. doi:10.3109/13625187.2016.1168799.
- Westhoff G, Cotter AM, Tolosa JE. Prophylactic oxytocin for the third stage of labour to prevent postpartum hemorrhage. Cochrane Database Syst Rev. 2013;(10):CD001808.
- Gibbons KJ, Albright CM, Rouse DJ. Postpartum hemorrhage in the developed world: whither misoprostol? Am J Obstet Gynecol. 2013;208(3):181−183.
- Blum J, Winikoff B, Raghavan S, et al. Treatment of postpartum hemorrhage with sublingual misoprostol versus oxytocin in women receiving prophylactic oxytocin: a double-blind, randomised, non-inferiority trial. Lancet. 2010;375(9710):217−223.
- Widmer M, Blum J, Hofmeyr GJ, et al. Misoprostolas an adjunct to standard uterotonics for treatment of post-partum haemorrhage: a multicentre, double-blind randomised trial. Lancet. 2010;375(9728):1808−1813.
- Weeks A. The prevention and treatment of postpartum hemorrhage: what do we know and where do we go to next? BJOG. 2015;122(2):202−210.
- Khan RU, El-Refaey H. Pharmacokinetics and adverse-effect profile of rectally administered misoprostol in the third stage of labor. Obstet Gynecol. 2003;101(5 pt 1):968−974.
- Khan RU, El-Refaey H, Sharma S, Sooranna D, Stafford M. Oral, rectal and vaginal pharmacokinetics of misoprostol. Obstet Gynecol. 2004;103(5 pt 1):866−870.
- Meckstroth KR, Whitaker AK, Bertisch S, Goldberg AB, Darney PD. Misoprostol administered by epithelial routes: drug absorption and uterine response. Obstet Gynecol. 2006;108(3 pt 1):582−590.
- Schaff EA, DiCenzo R, Fielding SL. Comparison of misoprostol plasma concentrations following buccal and sublingual administration. Contraception. 2005;71(1):22−25.
- Frye LJ, Byrne ME, Winikoff B. A crossover pharmacokinetic study of misoprostol by the oral, sublingual and buccal routes [published online ahead of print April 22, 2016]. Eur J Contracept Reprod Health Care. doi:10.3109/13625187.2016.1168799.
Fear and loathing abound in the ‘off-label’ presidential election of 2016
A day in the psychiatry clinic? No—just scenes from that high-stakes festival of intense human competitiveness gone awry: the current presidential election. Alas, we have no FDA-approved treatments for any of these unusual political behaviors.
More stunning is how blind some loyal voters are to the flaws of their candidate of choice. They seem to be joyfully intoxicated by sharing the unusual beliefs of the candidate, in a cultish folie en masse of epidemic proportion.
Other (rational) voters are stunned and jarred by what they see and hear; they appear to be in need of Rx: an intellectual antiemetic.
The rise of uber-narcissism
A certain amount of narcissism is, understandably, necessary to run for the nation’s highest office and to believe, against all odds, that winning is certain despite microscopic favorability in the polls. In this election cycle, the cup of narcissism has run over; yet, to adoring fans, narcissism only adds a wondrous halo to their candidate.
The history of the United States is rife with similar behavior by elected officials, including our revered Founding Fathers.1 But today’s psychiatrists, witnessing this national charade, are perplexed and question the rationality of the national psyche. Established rules for seeking the Presidency have been demolished and the show goes on as if heightened narcissism is the new normal in human behavior.
Giving voice to my colleagues’ consternation
Here are a few thoughts that might cross the mind of psychiatrists as they watch, with a frown and pursed lips, this unconventional election cycle:
From a psychoanalytic perspective, the id has left the ego in its dust, and the super-ego went home to hide.
When boorishness trumps civility, hillaryous consequences ensue.
The gullibility of voters deserves serious scientific study. Jeste and Harris2 reviewed the evidence for a neurobiology of wisdom; The National Institutes of Health should fund research into how some voters believe the candidate of their choice will provide them with everything they wish. The chicken in every pot expands to 100 in every pot, and money grows on trees (at least on 1% of the forest!).
From an evolutionary standpoint, survival of the fittest has become survival of the most bombastic.
The zeitgeist reflects an electorate that oscillates agonizingly from surprise to anger to cynicism to disgust.
The traditional internal conflict (studied by political scientists) of choosing between 2 reasonably meritorious candidates has been transformed into a conflict over whether to vote at all.
This is the least nuanced presidential campaign—ever.
All decision-making in politics is unconscious, political scientist Jon A. Krosnick proposed. In this election, however, candidates’ enunciations are so overt that it’s hard to believe there’s anything left in the unconscious. Freud spoke of the “primary process” arising from the unconscious; he definitely was not referring to the primary process we experienced during this election cycle.
From a neuropsychiatric perspective, the limbic system has kicked the cortex in the metaphorical derrière in this election campaign.
Unabashed display of character flaws, personal shortcomings, and biases prove that anyone can run for president in a democracy, and that some voters will display a flight of reason and vote for a flawed candidate.
Even an inept demagogue can be misperceived as a savior by followers. Some voters could use a few sessions of insight-oriented therapy or cognitive-behavioral therapy for their unrealistic expectations.
It is dizzying, mentally, to watch candidates’ verbal acrobatics as they try to pass several litmus tests to satisfy disparate demands of sundry constituencies and mendaciously flip-flop on many issues—ignoring the fact that everything they have said was recorded or videotaped. Intellectual transvestism is a political sin, and sinners abound.
Oh, for a Jenner, Pasteur, or Sabin to discover vaccines for the intellect
Writing this editorial has been therapeutic. It feels good to ventilate about this bizarre election process that has the nation in its grip. I would feel much better if neuroscientists would develop and license vaccines that would broadly inoculate candidates against demagoguery, dishonesty, and pandering and voters against mind-boggling gullibility.
That would make elections so boring. But also so on-label….
1. Gartner JD. The hypomanic edge: the link between (a little) craziness and (a lot of) success in America. New York, NY: Simon & Schuster; 2005.
2. Jeste DV, Harris JC. Wisdom—a neuroscience perspective. JAMA. 2013;304(14):1602-1603.
A day in the psychiatry clinic? No—just scenes from that high-stakes festival of intense human competitiveness gone awry: the current presidential election. Alas, we have no FDA-approved treatments for any of these unusual political behaviors.
More stunning is how blind some loyal voters are to the flaws of their candidate of choice. They seem to be joyfully intoxicated by sharing the unusual beliefs of the candidate, in a cultish folie en masse of epidemic proportion.
Other (rational) voters are stunned and jarred by what they see and hear; they appear to be in need of Rx: an intellectual antiemetic.
The rise of uber-narcissism
A certain amount of narcissism is, understandably, necessary to run for the nation’s highest office and to believe, against all odds, that winning is certain despite microscopic favorability in the polls. In this election cycle, the cup of narcissism has run over; yet, to adoring fans, narcissism only adds a wondrous halo to their candidate.
The history of the United States is rife with similar behavior by elected officials, including our revered Founding Fathers.1 But today’s psychiatrists, witnessing this national charade, are perplexed and question the rationality of the national psyche. Established rules for seeking the Presidency have been demolished and the show goes on as if heightened narcissism is the new normal in human behavior.
Giving voice to my colleagues’ consternation
Here are a few thoughts that might cross the mind of psychiatrists as they watch, with a frown and pursed lips, this unconventional election cycle:
From a psychoanalytic perspective, the id has left the ego in its dust, and the super-ego went home to hide.
When boorishness trumps civility, hillaryous consequences ensue.
The gullibility of voters deserves serious scientific study. Jeste and Harris2 reviewed the evidence for a neurobiology of wisdom; The National Institutes of Health should fund research into how some voters believe the candidate of their choice will provide them with everything they wish. The chicken in every pot expands to 100 in every pot, and money grows on trees (at least on 1% of the forest!).
From an evolutionary standpoint, survival of the fittest has become survival of the most bombastic.
The zeitgeist reflects an electorate that oscillates agonizingly from surprise to anger to cynicism to disgust.
The traditional internal conflict (studied by political scientists) of choosing between 2 reasonably meritorious candidates has been transformed into a conflict over whether to vote at all.
This is the least nuanced presidential campaign—ever.
All decision-making in politics is unconscious, political scientist Jon A. Krosnick proposed. In this election, however, candidates’ enunciations are so overt that it’s hard to believe there’s anything left in the unconscious. Freud spoke of the “primary process” arising from the unconscious; he definitely was not referring to the primary process we experienced during this election cycle.
From a neuropsychiatric perspective, the limbic system has kicked the cortex in the metaphorical derrière in this election campaign.
Unabashed display of character flaws, personal shortcomings, and biases prove that anyone can run for president in a democracy, and that some voters will display a flight of reason and vote for a flawed candidate.
Even an inept demagogue can be misperceived as a savior by followers. Some voters could use a few sessions of insight-oriented therapy or cognitive-behavioral therapy for their unrealistic expectations.
It is dizzying, mentally, to watch candidates’ verbal acrobatics as they try to pass several litmus tests to satisfy disparate demands of sundry constituencies and mendaciously flip-flop on many issues—ignoring the fact that everything they have said was recorded or videotaped. Intellectual transvestism is a political sin, and sinners abound.
Oh, for a Jenner, Pasteur, or Sabin to discover vaccines for the intellect
Writing this editorial has been therapeutic. It feels good to ventilate about this bizarre election process that has the nation in its grip. I would feel much better if neuroscientists would develop and license vaccines that would broadly inoculate candidates against demagoguery, dishonesty, and pandering and voters against mind-boggling gullibility.
That would make elections so boring. But also so on-label….
A day in the psychiatry clinic? No—just scenes from that high-stakes festival of intense human competitiveness gone awry: the current presidential election. Alas, we have no FDA-approved treatments for any of these unusual political behaviors.
More stunning is how blind some loyal voters are to the flaws of their candidate of choice. They seem to be joyfully intoxicated by sharing the unusual beliefs of the candidate, in a cultish folie en masse of epidemic proportion.
Other (rational) voters are stunned and jarred by what they see and hear; they appear to be in need of Rx: an intellectual antiemetic.
The rise of uber-narcissism
A certain amount of narcissism is, understandably, necessary to run for the nation’s highest office and to believe, against all odds, that winning is certain despite microscopic favorability in the polls. In this election cycle, the cup of narcissism has run over; yet, to adoring fans, narcissism only adds a wondrous halo to their candidate.
The history of the United States is rife with similar behavior by elected officials, including our revered Founding Fathers.1 But today’s psychiatrists, witnessing this national charade, are perplexed and question the rationality of the national psyche. Established rules for seeking the Presidency have been demolished and the show goes on as if heightened narcissism is the new normal in human behavior.
Giving voice to my colleagues’ consternation
Here are a few thoughts that might cross the mind of psychiatrists as they watch, with a frown and pursed lips, this unconventional election cycle:
From a psychoanalytic perspective, the id has left the ego in its dust, and the super-ego went home to hide.
When boorishness trumps civility, hillaryous consequences ensue.
The gullibility of voters deserves serious scientific study. Jeste and Harris2 reviewed the evidence for a neurobiology of wisdom; The National Institutes of Health should fund research into how some voters believe the candidate of their choice will provide them with everything they wish. The chicken in every pot expands to 100 in every pot, and money grows on trees (at least on 1% of the forest!).
From an evolutionary standpoint, survival of the fittest has become survival of the most bombastic.
The zeitgeist reflects an electorate that oscillates agonizingly from surprise to anger to cynicism to disgust.
The traditional internal conflict (studied by political scientists) of choosing between 2 reasonably meritorious candidates has been transformed into a conflict over whether to vote at all.
This is the least nuanced presidential campaign—ever.
All decision-making in politics is unconscious, political scientist Jon A. Krosnick proposed. In this election, however, candidates’ enunciations are so overt that it’s hard to believe there’s anything left in the unconscious. Freud spoke of the “primary process” arising from the unconscious; he definitely was not referring to the primary process we experienced during this election cycle.
From a neuropsychiatric perspective, the limbic system has kicked the cortex in the metaphorical derrière in this election campaign.
Unabashed display of character flaws, personal shortcomings, and biases prove that anyone can run for president in a democracy, and that some voters will display a flight of reason and vote for a flawed candidate.
Even an inept demagogue can be misperceived as a savior by followers. Some voters could use a few sessions of insight-oriented therapy or cognitive-behavioral therapy for their unrealistic expectations.
It is dizzying, mentally, to watch candidates’ verbal acrobatics as they try to pass several litmus tests to satisfy disparate demands of sundry constituencies and mendaciously flip-flop on many issues—ignoring the fact that everything they have said was recorded or videotaped. Intellectual transvestism is a political sin, and sinners abound.
Oh, for a Jenner, Pasteur, or Sabin to discover vaccines for the intellect
Writing this editorial has been therapeutic. It feels good to ventilate about this bizarre election process that has the nation in its grip. I would feel much better if neuroscientists would develop and license vaccines that would broadly inoculate candidates against demagoguery, dishonesty, and pandering and voters against mind-boggling gullibility.
That would make elections so boring. But also so on-label….
1. Gartner JD. The hypomanic edge: the link between (a little) craziness and (a lot of) success in America. New York, NY: Simon & Schuster; 2005.
2. Jeste DV, Harris JC. Wisdom—a neuroscience perspective. JAMA. 2013;304(14):1602-1603.
1. Gartner JD. The hypomanic edge: the link between (a little) craziness and (a lot of) success in America. New York, NY: Simon & Schuster; 2005.
2. Jeste DV, Harris JC. Wisdom—a neuroscience perspective. JAMA. 2013;304(14):1602-1603.
LGBT care: There has been progress
McNamara and Ng, in this issue of the Journal, discuss the psychosocial and clinical aspects of caring for people who are lesbian, gay, bisexual, or transgender (LGBT), a community that numbers more than 9 million in the United States. Choices for health maintenance and screening are influenced by the patient’s age, sexual practices, comorbidities, and in some transgender patients by current and previous therapeutic hormone manipulations. Although these medical decisions must be personalized, many are informed by existing guidelines for the general population and thus do not represent a dramatic departure from decision-making in other patients.
More difficult is acquiring the requisite understanding and appreciation of the special challenges each LGBT patient brings to the office visit. We need to understand these challenges to provide medically appropriate, compassionate, patient-centered care.
McNamara and Ng suggest simple acts of kindness and consideration to increase the comfort of this group of patients who historically, for a myriad of complex reasons, have not been uniformly made to feel comfortable receiving routine care in our medical system. A tectonic shift has taken place in the way society at large perceives and interacts with members of the LGBT community. Large pockets of intolerance and lack of understanding certainly still exist. But I want to believe that as a medical community, we have changed dramatically over the past several decades.
Early in my medical career, as the AIDS epidemic expanded from unexplained Pneumocystis carinii (now jirovecii) and fungal infections and virulent Kaposi sarcoma to include Guillain-Barré syndrome, central nervous system tumors and infections, and so much more, I watched the gay community rally around specific doctors and medical centers—and avoid others. It was more than just the perception that some hospitals were “gay-friendly.” Care was more compassionate, supportive, and thorough at some hospitals than others. I well recall having difficulty finding a neurosurgeon willing to biopsy a brain mass in one of my patients with AIDS, and finding an orthopedic surgeon willing to manage an infected hip prosthesis in another one. Fast forward 3 decades, and now in a different hospital I am managing gout in a patient who is infected with HIV, is in apparent remission without detectable virus on highly active antiretroviral therapy, and recently received a heart transplant.
As controversies continue to swirl and external acts of discrimination continue to impact the LGBT community on many fronts—bathroom laws, spousal rights, child adoption, gay political leaders, xenophobia, and even hate crimes—it is encouraging to read a matter-of-fact, practical approach to “best practices” in LGBT care. No hype. No judgment. Just compassionate, appropriate medical care.
The journey isn’t over, but there has been progress.
McNamara and Ng, in this issue of the Journal, discuss the psychosocial and clinical aspects of caring for people who are lesbian, gay, bisexual, or transgender (LGBT), a community that numbers more than 9 million in the United States. Choices for health maintenance and screening are influenced by the patient’s age, sexual practices, comorbidities, and in some transgender patients by current and previous therapeutic hormone manipulations. Although these medical decisions must be personalized, many are informed by existing guidelines for the general population and thus do not represent a dramatic departure from decision-making in other patients.
More difficult is acquiring the requisite understanding and appreciation of the special challenges each LGBT patient brings to the office visit. We need to understand these challenges to provide medically appropriate, compassionate, patient-centered care.
McNamara and Ng suggest simple acts of kindness and consideration to increase the comfort of this group of patients who historically, for a myriad of complex reasons, have not been uniformly made to feel comfortable receiving routine care in our medical system. A tectonic shift has taken place in the way society at large perceives and interacts with members of the LGBT community. Large pockets of intolerance and lack of understanding certainly still exist. But I want to believe that as a medical community, we have changed dramatically over the past several decades.
Early in my medical career, as the AIDS epidemic expanded from unexplained Pneumocystis carinii (now jirovecii) and fungal infections and virulent Kaposi sarcoma to include Guillain-Barré syndrome, central nervous system tumors and infections, and so much more, I watched the gay community rally around specific doctors and medical centers—and avoid others. It was more than just the perception that some hospitals were “gay-friendly.” Care was more compassionate, supportive, and thorough at some hospitals than others. I well recall having difficulty finding a neurosurgeon willing to biopsy a brain mass in one of my patients with AIDS, and finding an orthopedic surgeon willing to manage an infected hip prosthesis in another one. Fast forward 3 decades, and now in a different hospital I am managing gout in a patient who is infected with HIV, is in apparent remission without detectable virus on highly active antiretroviral therapy, and recently received a heart transplant.
As controversies continue to swirl and external acts of discrimination continue to impact the LGBT community on many fronts—bathroom laws, spousal rights, child adoption, gay political leaders, xenophobia, and even hate crimes—it is encouraging to read a matter-of-fact, practical approach to “best practices” in LGBT care. No hype. No judgment. Just compassionate, appropriate medical care.
The journey isn’t over, but there has been progress.
McNamara and Ng, in this issue of the Journal, discuss the psychosocial and clinical aspects of caring for people who are lesbian, gay, bisexual, or transgender (LGBT), a community that numbers more than 9 million in the United States. Choices for health maintenance and screening are influenced by the patient’s age, sexual practices, comorbidities, and in some transgender patients by current and previous therapeutic hormone manipulations. Although these medical decisions must be personalized, many are informed by existing guidelines for the general population and thus do not represent a dramatic departure from decision-making in other patients.
More difficult is acquiring the requisite understanding and appreciation of the special challenges each LGBT patient brings to the office visit. We need to understand these challenges to provide medically appropriate, compassionate, patient-centered care.
McNamara and Ng suggest simple acts of kindness and consideration to increase the comfort of this group of patients who historically, for a myriad of complex reasons, have not been uniformly made to feel comfortable receiving routine care in our medical system. A tectonic shift has taken place in the way society at large perceives and interacts with members of the LGBT community. Large pockets of intolerance and lack of understanding certainly still exist. But I want to believe that as a medical community, we have changed dramatically over the past several decades.
Early in my medical career, as the AIDS epidemic expanded from unexplained Pneumocystis carinii (now jirovecii) and fungal infections and virulent Kaposi sarcoma to include Guillain-Barré syndrome, central nervous system tumors and infections, and so much more, I watched the gay community rally around specific doctors and medical centers—and avoid others. It was more than just the perception that some hospitals were “gay-friendly.” Care was more compassionate, supportive, and thorough at some hospitals than others. I well recall having difficulty finding a neurosurgeon willing to biopsy a brain mass in one of my patients with AIDS, and finding an orthopedic surgeon willing to manage an infected hip prosthesis in another one. Fast forward 3 decades, and now in a different hospital I am managing gout in a patient who is infected with HIV, is in apparent remission without detectable virus on highly active antiretroviral therapy, and recently received a heart transplant.
As controversies continue to swirl and external acts of discrimination continue to impact the LGBT community on many fronts—bathroom laws, spousal rights, child adoption, gay political leaders, xenophobia, and even hate crimes—it is encouraging to read a matter-of-fact, practical approach to “best practices” in LGBT care. No hype. No judgment. Just compassionate, appropriate medical care.
The journey isn’t over, but there has been progress.
The fifth vital sign: A complex story of politics and patient care
In this issue of the Journal, Dr. Marissa Galicia-Castillo discusses the use of opioids in older patients with persistent (formerly known as chronic) pain. Even though she devotes one and a half pages to the side effects of chronic opioid therapy, I am sure that in the current environment many readers will perceive her as expressing a surprisingly supportive tone regarding the use of these medications. The times have changed, and the difficulties and complexities of trying to help patients with ongoing pain have increased.
In the mid-1990s, the American Pain Society aggressively pushed the concept of pain as the fifth vital sign.1 Their stated goals included raising awareness that patients with pain were undertreated, in large part because in the Society’s opinion pain was not regularly assessed at physician office visits or even in the hospital after surgery. Half a decade later the Joint Commission and others hopped on this train, emphasizing that pain needs to be regularly assessed in all patients, that pain is a subjective measure, unlike the heart rate or blood pressure, and that physicians must accept and respect patient self-reporting of pain. Concurrent with these efforts was the enhanced promotion of pain medications—new highly touted and frequently prescribed narcotics as well as nonnarcotic medications re-marketed as analgesics. Opportunistically, or perhaps wielding inappropriate and sketchy influence, some drug manufacturers in the early 2000s funded publications and physician presentations to encourage the expanded use of opioids and other medications for pain control. In a recent CNN report on the opioid epidemic, it was noted that the Joint Commission published a book in 2000 for purchase by doctors as part of required continuing education seminars, and that the book cited studies claiming “there is no evidence that addiction is a significant issue when persons are given opioids for pain control.”2 According to the CNN report, the book was sponsored by a manufacturer of narcotic analgesics.2 Lack of evidence is not evidence supporting a lack of known concern.
Step forward in time, and pain control has become a measure of patient satisfaction, and thus potentially another physician and institutional rating score that can be linked to reimbursement. This despite reports suggesting that incorporation of this required pain scale did not actually improve the quality of pain management.3 I suspect that most of my peers function in the outpatient clinic as I do, without much interest in what was recorded on the intake pain scale, since I will be taking a more focused and detailed history from the patient if pain is any part of the reason for visiting with me. The goal of alleviating a patient’s pain, whenever reasonable, must always be on our agenda. Yet, while we need to respond to scores on a somewhat silly screening pain scale, the hurdles to prescribing analgesics are getting higher.
The latest data on opioid-related deaths are sobering and scary. Organized medicine must absolutely push to close the pain-pill mills, but is the link really so strong between thoughtful prescribing of short- or even long-term opioids and the escalating “epidemic” of opioid complications that we should not prescribe these drugs? Does the fact that we don’t have good data demonstrating long-term efficacy mean that these drugs are not effective in appropriately selected patients? Is it warranted to require regular database reviews of all patients who are prescribed these medications? Is it warranted, as one patient said to me, that she be treated like a potential criminal begging for drugs when her prescriptions are up, and that she be “looked at funny” by the pharmacist when she fills them?
An increasingly discussed concept is that of central generalization of pain, and patients who have this may be opioid-resistant and, perhaps, prone to developing opioid hyperalgesia. It has been studied in patients with fibromyalgia and is now felt by some to include patients with osteoarthritis and other initially localized painful conditions. Whether or not this concept ultimately turns out to be correct, it adds another dimension to our assessment of patients with pain.
The time has come to move past using a one-size-fits-all fifth vital sign (“How would you rate your pain on a scale of 1 to 10?”) and reflexively prescribing an opioid when pain is characterized as severe. But, if the patient truly needs the drug, we also need to move past not writing that prescription because of headlines and administrative hurdles. This is a much more complex story.
- American Pain Society Quality of Care Committee. Quality improvement guidelines for the treatment of acute pain and cancer pain. JAMA 1995; 274:1874–1880.
- Moghe S. Opioid history: from ‘wonder drug’ to abuse epidemic. www.cnn.com/2016/05/12/health/opioid-addiction-history/. Accessed May 16, 2016.
- Mularski RA, White-Chu F, Overbay D, et al. Measuring pain as the 5th vital sign does not improve quality of pain management. J Gen Intern Med 2006; 21:607–612.
In this issue of the Journal, Dr. Marissa Galicia-Castillo discusses the use of opioids in older patients with persistent (formerly known as chronic) pain. Even though she devotes one and a half pages to the side effects of chronic opioid therapy, I am sure that in the current environment many readers will perceive her as expressing a surprisingly supportive tone regarding the use of these medications. The times have changed, and the difficulties and complexities of trying to help patients with ongoing pain have increased.
In the mid-1990s, the American Pain Society aggressively pushed the concept of pain as the fifth vital sign.1 Their stated goals included raising awareness that patients with pain were undertreated, in large part because in the Society’s opinion pain was not regularly assessed at physician office visits or even in the hospital after surgery. Half a decade later the Joint Commission and others hopped on this train, emphasizing that pain needs to be regularly assessed in all patients, that pain is a subjective measure, unlike the heart rate or blood pressure, and that physicians must accept and respect patient self-reporting of pain. Concurrent with these efforts was the enhanced promotion of pain medications—new highly touted and frequently prescribed narcotics as well as nonnarcotic medications re-marketed as analgesics. Opportunistically, or perhaps wielding inappropriate and sketchy influence, some drug manufacturers in the early 2000s funded publications and physician presentations to encourage the expanded use of opioids and other medications for pain control. In a recent CNN report on the opioid epidemic, it was noted that the Joint Commission published a book in 2000 for purchase by doctors as part of required continuing education seminars, and that the book cited studies claiming “there is no evidence that addiction is a significant issue when persons are given opioids for pain control.”2 According to the CNN report, the book was sponsored by a manufacturer of narcotic analgesics.2 Lack of evidence is not evidence supporting a lack of known concern.
Step forward in time, and pain control has become a measure of patient satisfaction, and thus potentially another physician and institutional rating score that can be linked to reimbursement. This despite reports suggesting that incorporation of this required pain scale did not actually improve the quality of pain management.3 I suspect that most of my peers function in the outpatient clinic as I do, without much interest in what was recorded on the intake pain scale, since I will be taking a more focused and detailed history from the patient if pain is any part of the reason for visiting with me. The goal of alleviating a patient’s pain, whenever reasonable, must always be on our agenda. Yet, while we need to respond to scores on a somewhat silly screening pain scale, the hurdles to prescribing analgesics are getting higher.
The latest data on opioid-related deaths are sobering and scary. Organized medicine must absolutely push to close the pain-pill mills, but is the link really so strong between thoughtful prescribing of short- or even long-term opioids and the escalating “epidemic” of opioid complications that we should not prescribe these drugs? Does the fact that we don’t have good data demonstrating long-term efficacy mean that these drugs are not effective in appropriately selected patients? Is it warranted to require regular database reviews of all patients who are prescribed these medications? Is it warranted, as one patient said to me, that she be treated like a potential criminal begging for drugs when her prescriptions are up, and that she be “looked at funny” by the pharmacist when she fills them?
An increasingly discussed concept is that of central generalization of pain, and patients who have this may be opioid-resistant and, perhaps, prone to developing opioid hyperalgesia. It has been studied in patients with fibromyalgia and is now felt by some to include patients with osteoarthritis and other initially localized painful conditions. Whether or not this concept ultimately turns out to be correct, it adds another dimension to our assessment of patients with pain.
The time has come to move past using a one-size-fits-all fifth vital sign (“How would you rate your pain on a scale of 1 to 10?”) and reflexively prescribing an opioid when pain is characterized as severe. But, if the patient truly needs the drug, we also need to move past not writing that prescription because of headlines and administrative hurdles. This is a much more complex story.
In this issue of the Journal, Dr. Marissa Galicia-Castillo discusses the use of opioids in older patients with persistent (formerly known as chronic) pain. Even though she devotes one and a half pages to the side effects of chronic opioid therapy, I am sure that in the current environment many readers will perceive her as expressing a surprisingly supportive tone regarding the use of these medications. The times have changed, and the difficulties and complexities of trying to help patients with ongoing pain have increased.
In the mid-1990s, the American Pain Society aggressively pushed the concept of pain as the fifth vital sign.1 Their stated goals included raising awareness that patients with pain were undertreated, in large part because in the Society’s opinion pain was not regularly assessed at physician office visits or even in the hospital after surgery. Half a decade later the Joint Commission and others hopped on this train, emphasizing that pain needs to be regularly assessed in all patients, that pain is a subjective measure, unlike the heart rate or blood pressure, and that physicians must accept and respect patient self-reporting of pain. Concurrent with these efforts was the enhanced promotion of pain medications—new highly touted and frequently prescribed narcotics as well as nonnarcotic medications re-marketed as analgesics. Opportunistically, or perhaps wielding inappropriate and sketchy influence, some drug manufacturers in the early 2000s funded publications and physician presentations to encourage the expanded use of opioids and other medications for pain control. In a recent CNN report on the opioid epidemic, it was noted that the Joint Commission published a book in 2000 for purchase by doctors as part of required continuing education seminars, and that the book cited studies claiming “there is no evidence that addiction is a significant issue when persons are given opioids for pain control.”2 According to the CNN report, the book was sponsored by a manufacturer of narcotic analgesics.2 Lack of evidence is not evidence supporting a lack of known concern.
Step forward in time, and pain control has become a measure of patient satisfaction, and thus potentially another physician and institutional rating score that can be linked to reimbursement. This despite reports suggesting that incorporation of this required pain scale did not actually improve the quality of pain management.3 I suspect that most of my peers function in the outpatient clinic as I do, without much interest in what was recorded on the intake pain scale, since I will be taking a more focused and detailed history from the patient if pain is any part of the reason for visiting with me. The goal of alleviating a patient’s pain, whenever reasonable, must always be on our agenda. Yet, while we need to respond to scores on a somewhat silly screening pain scale, the hurdles to prescribing analgesics are getting higher.
The latest data on opioid-related deaths are sobering and scary. Organized medicine must absolutely push to close the pain-pill mills, but is the link really so strong between thoughtful prescribing of short- or even long-term opioids and the escalating “epidemic” of opioid complications that we should not prescribe these drugs? Does the fact that we don’t have good data demonstrating long-term efficacy mean that these drugs are not effective in appropriately selected patients? Is it warranted to require regular database reviews of all patients who are prescribed these medications? Is it warranted, as one patient said to me, that she be treated like a potential criminal begging for drugs when her prescriptions are up, and that she be “looked at funny” by the pharmacist when she fills them?
An increasingly discussed concept is that of central generalization of pain, and patients who have this may be opioid-resistant and, perhaps, prone to developing opioid hyperalgesia. It has been studied in patients with fibromyalgia and is now felt by some to include patients with osteoarthritis and other initially localized painful conditions. Whether or not this concept ultimately turns out to be correct, it adds another dimension to our assessment of patients with pain.
The time has come to move past using a one-size-fits-all fifth vital sign (“How would you rate your pain on a scale of 1 to 10?”) and reflexively prescribing an opioid when pain is characterized as severe. But, if the patient truly needs the drug, we also need to move past not writing that prescription because of headlines and administrative hurdles. This is a much more complex story.
- American Pain Society Quality of Care Committee. Quality improvement guidelines for the treatment of acute pain and cancer pain. JAMA 1995; 274:1874–1880.
- Moghe S. Opioid history: from ‘wonder drug’ to abuse epidemic. www.cnn.com/2016/05/12/health/opioid-addiction-history/. Accessed May 16, 2016.
- Mularski RA, White-Chu F, Overbay D, et al. Measuring pain as the 5th vital sign does not improve quality of pain management. J Gen Intern Med 2006; 21:607–612.
- American Pain Society Quality of Care Committee. Quality improvement guidelines for the treatment of acute pain and cancer pain. JAMA 1995; 274:1874–1880.
- Moghe S. Opioid history: from ‘wonder drug’ to abuse epidemic. www.cnn.com/2016/05/12/health/opioid-addiction-history/. Accessed May 16, 2016.
- Mularski RA, White-Chu F, Overbay D, et al. Measuring pain as the 5th vital sign does not improve quality of pain management. J Gen Intern Med 2006; 21:607–612.
The scourge of societal anosognosia about the mentally ill
What if this increase had occurred in cardiovascular disease or cancer (both on the decline, in fact, thanks to the intense attention they receive)? I think there would have been a public outcry, followed by demands by Congress that the National Institutes of Health and the CDC address this catastrophic rise immediately. And billions of dollars would then be earmarked to prevent these 2 diseases.
How sad that society has “forgotten” that mental illness has deadly consequences, often leading to suicide (42,773 deaths in 2014 alone2—the second most common cause among people age 15 to 253)! Hundreds of thousands of people attempt suicide every year, and those who do not lose their life often end up injured or maimed. Millions who suffer depression, bipolar disorder, schizophrenia, anxiety, posttraumatic stress disorder, or a substance use disorder are at high risk of suicide, and many never receive the timely intervention that might save their life.
Our national blind spot
It is poignant that the CDC report was released in spring: The rate of suicide is highest in April and May, when the light-dark cycle is reversed. This springtime peak runs contrary to the common belief that the rate of suicide is highest during winter months. The Annual Meeting of the American Psychiatric Association convenes in May, such that, ironically, thousands of psychiatrists are away from their office exactly when their patients might need them most
Lack of attention to the high risk of suicide among all ages and both sexes is emblematic of society’s inexplicable neglect of the needs of the mentally ill. That neglect is fueled, and exacerbated, by the destructive stigma attached to brain disorders that display psychiatric symptoms. As a neuropsychiatrist, I label this neglect societal anosognosia—the same as the lack of insight seen in patients with acute schizophrenia, who are unaware of how impaired they are and insist that they are not sick. (Anosognosia also occurs in stroke patients who deny that their limb is paralyzed and insist that all is well.)
Loss of insight can have serious consequences for patients who lose the ability to monitor and evaluate their physical and mental health. Just as patients with anosognosia think they do not need help, a society that fails to attend to the mental illness of its citizens endangers their overall health and welfare.
From neglect of mental illness many hazards arise
Tens of millions of Americans suffer from mental illness, according to the National Institute of Mental Health-sponsored Epidemiologic Catchment Area Study.4 The last thing these people can afford is societal anosognosia, which deprives them of necessary and timely access to psychiatric care.
Societal anosognosia is associated with numerous hazards for persons with mental illness, including:
- Lack of compassion, which is readily available for people with a medical ailment (broken bones, cardiovascular disease, cancer).
- Lack of adequate, affordable health insurance and financial support, compared with what is available for non-psychiatric disorders.
- Shortage of publicly funded programs and mental health practitioners to provide prevention and intervention for those who consider ending their life during an episode of depression, psychosis, stress, or a panic attack.
- Allowing the stigma to continue unabated. Why are there strict laws about hate crimes, but not about stigma? Why does society continue to portray depression and anxiety as a personal weakness or failure, while patients with Parkinson’s disease or multiple sclerosis who have motor weakness are not stigmatized for their physical deficits?
- Transforming the seriously mentally ill into felons by arresting and jailing them because of erratic behavior—instead of hospitalizing them for the medical care they need. The transinstitutionalization of the mentally ill—from state hospitals to prisons—is one of the most shameful consequences of societal anosognosia, burdening our patients with the dual stigma of being a criminal and mentally ill.
- Turning a blind eye to abuses by insurance companies. More appalling is the perpetuation of restricted health coverage despite the passage of parity laws! Why are sensory and motor disorders of brain lesions covered fully, while the thought, emotional, and behavior disorders of the brain covered only partially?
- Consent laws that restrict psychiatrists from medicating acutely psychotic or depressed patients unless they consent—but no laws that restrict a cardiologist from immediately treating an unconscious heart attack patient who cannot consent, or an obtunded stroke patient who cannot communicate? The duration of untreated psychosis or depression has been shown repeatedly to have deleterious effects on brain tissue and functional outcomes, yet treatment of an acutely ill psychiatric patient is often delayed until a court order is obtained. When was the last time a court order was needed to treat an acute myocardial infarction?
- Failure to recognize that premature mortality (by approximately 25 years) is a devastating consequence of mental illness, whether from suicide or cardiometabolic risk factors due to smoking, substance use (often used to self-medicate because proper treatment is lacking), poor diet, and sedentary living.
- Failure to provide basic primary care to people with severe mental illness, and the much lower use of life-saving diagnostic and treatment procedures offered to these patients, compared with non-psychiatric patients.
- Inadequate funding for research on psychiatric disorders, compared with other medical disorders—even though direct and indirect costs of mental illness to society (hundreds of billions of dollars a year) far exceed costs of most medical disorders.
- Severe shortage of rehabilitation programs for the mentally ill, compared with many other medical disorders. Why does paralysis of the mind receive far less support than paralysis of the legs or arms?
The rising suicide rate reflects poorly on us
Societal anosognosia is a global scourge, affecting many underdeveloped countries. Why do developed nations, like ours, have the same blind spot for mental illness? Might ignorance and discrimination be universal?
The tragic rise in the rate of death by suicide in men and women, among all age groups, year after year, is stunningly incongruent when juxtaposed against the elimination of smallpox and other communicable diseases through a concerted societal effort to support scientific advances in vaccine development. Societal anosognosia appears to be selective: We have comprehensive insight about diseases of the body but not diseases of the mind.
The essence and soul of a society are the collective minds of its citizens, not their bodies. Societal anosognosia is a serious dysfunction of its mind, and a rising suicide rate is a symptom of that pathological dysfunction.
1. Curtin SC, Warner M, Hedegaard H, et al. Increase in suicide in the United States, 1999-2014. National Center for Health Statistics Data Brief No. 241. Atlanta, GA: National Center for Health Statistics, U.S. Department of Health and Human Services; 2016.
2. Ten leading causes of death by age group, United States – 2014. Centers for Disease Control and Prevention. http://www.cdc.gov/injury/images/lc-charts/leading_causes_of_death_age_group_2014_1050w760h.gif. Accessed May 20, 2016.
3. Morris M. Stemming the rising tide of suicide. Clinical Psychiatry News. http://www.clinicalpsychiatrynews.com/specialty-focus/depression/single-article-page/stemming-the-rising-tide-of-suicide/01cd45cabfc693bedb0e30bb6cb0b89e.html. Published April 26, 2016. Accessed May 13, 2016.
4. Robins LN, Regier DA, eds. Psychiatric disorders in America: the Epidemiologic Catchment Area Study. New York, NY: Free Press; 1990.
What if this increase had occurred in cardiovascular disease or cancer (both on the decline, in fact, thanks to the intense attention they receive)? I think there would have been a public outcry, followed by demands by Congress that the National Institutes of Health and the CDC address this catastrophic rise immediately. And billions of dollars would then be earmarked to prevent these 2 diseases.
How sad that society has “forgotten” that mental illness has deadly consequences, often leading to suicide (42,773 deaths in 2014 alone2—the second most common cause among people age 15 to 253)! Hundreds of thousands of people attempt suicide every year, and those who do not lose their life often end up injured or maimed. Millions who suffer depression, bipolar disorder, schizophrenia, anxiety, posttraumatic stress disorder, or a substance use disorder are at high risk of suicide, and many never receive the timely intervention that might save their life.
Our national blind spot
It is poignant that the CDC report was released in spring: The rate of suicide is highest in April and May, when the light-dark cycle is reversed. This springtime peak runs contrary to the common belief that the rate of suicide is highest during winter months. The Annual Meeting of the American Psychiatric Association convenes in May, such that, ironically, thousands of psychiatrists are away from their office exactly when their patients might need them most
Lack of attention to the high risk of suicide among all ages and both sexes is emblematic of society’s inexplicable neglect of the needs of the mentally ill. That neglect is fueled, and exacerbated, by the destructive stigma attached to brain disorders that display psychiatric symptoms. As a neuropsychiatrist, I label this neglect societal anosognosia—the same as the lack of insight seen in patients with acute schizophrenia, who are unaware of how impaired they are and insist that they are not sick. (Anosognosia also occurs in stroke patients who deny that their limb is paralyzed and insist that all is well.)
Loss of insight can have serious consequences for patients who lose the ability to monitor and evaluate their physical and mental health. Just as patients with anosognosia think they do not need help, a society that fails to attend to the mental illness of its citizens endangers their overall health and welfare.
From neglect of mental illness many hazards arise
Tens of millions of Americans suffer from mental illness, according to the National Institute of Mental Health-sponsored Epidemiologic Catchment Area Study.4 The last thing these people can afford is societal anosognosia, which deprives them of necessary and timely access to psychiatric care.
Societal anosognosia is associated with numerous hazards for persons with mental illness, including:
- Lack of compassion, which is readily available for people with a medical ailment (broken bones, cardiovascular disease, cancer).
- Lack of adequate, affordable health insurance and financial support, compared with what is available for non-psychiatric disorders.
- Shortage of publicly funded programs and mental health practitioners to provide prevention and intervention for those who consider ending their life during an episode of depression, psychosis, stress, or a panic attack.
- Allowing the stigma to continue unabated. Why are there strict laws about hate crimes, but not about stigma? Why does society continue to portray depression and anxiety as a personal weakness or failure, while patients with Parkinson’s disease or multiple sclerosis who have motor weakness are not stigmatized for their physical deficits?
- Transforming the seriously mentally ill into felons by arresting and jailing them because of erratic behavior—instead of hospitalizing them for the medical care they need. The transinstitutionalization of the mentally ill—from state hospitals to prisons—is one of the most shameful consequences of societal anosognosia, burdening our patients with the dual stigma of being a criminal and mentally ill.
- Turning a blind eye to abuses by insurance companies. More appalling is the perpetuation of restricted health coverage despite the passage of parity laws! Why are sensory and motor disorders of brain lesions covered fully, while the thought, emotional, and behavior disorders of the brain covered only partially?
- Consent laws that restrict psychiatrists from medicating acutely psychotic or depressed patients unless they consent—but no laws that restrict a cardiologist from immediately treating an unconscious heart attack patient who cannot consent, or an obtunded stroke patient who cannot communicate? The duration of untreated psychosis or depression has been shown repeatedly to have deleterious effects on brain tissue and functional outcomes, yet treatment of an acutely ill psychiatric patient is often delayed until a court order is obtained. When was the last time a court order was needed to treat an acute myocardial infarction?
- Failure to recognize that premature mortality (by approximately 25 years) is a devastating consequence of mental illness, whether from suicide or cardiometabolic risk factors due to smoking, substance use (often used to self-medicate because proper treatment is lacking), poor diet, and sedentary living.
- Failure to provide basic primary care to people with severe mental illness, and the much lower use of life-saving diagnostic and treatment procedures offered to these patients, compared with non-psychiatric patients.
- Inadequate funding for research on psychiatric disorders, compared with other medical disorders—even though direct and indirect costs of mental illness to society (hundreds of billions of dollars a year) far exceed costs of most medical disorders.
- Severe shortage of rehabilitation programs for the mentally ill, compared with many other medical disorders. Why does paralysis of the mind receive far less support than paralysis of the legs or arms?
The rising suicide rate reflects poorly on us
Societal anosognosia is a global scourge, affecting many underdeveloped countries. Why do developed nations, like ours, have the same blind spot for mental illness? Might ignorance and discrimination be universal?
The tragic rise in the rate of death by suicide in men and women, among all age groups, year after year, is stunningly incongruent when juxtaposed against the elimination of smallpox and other communicable diseases through a concerted societal effort to support scientific advances in vaccine development. Societal anosognosia appears to be selective: We have comprehensive insight about diseases of the body but not diseases of the mind.
The essence and soul of a society are the collective minds of its citizens, not their bodies. Societal anosognosia is a serious dysfunction of its mind, and a rising suicide rate is a symptom of that pathological dysfunction.
What if this increase had occurred in cardiovascular disease or cancer (both on the decline, in fact, thanks to the intense attention they receive)? I think there would have been a public outcry, followed by demands by Congress that the National Institutes of Health and the CDC address this catastrophic rise immediately. And billions of dollars would then be earmarked to prevent these 2 diseases.
How sad that society has “forgotten” that mental illness has deadly consequences, often leading to suicide (42,773 deaths in 2014 alone2—the second most common cause among people age 15 to 253)! Hundreds of thousands of people attempt suicide every year, and those who do not lose their life often end up injured or maimed. Millions who suffer depression, bipolar disorder, schizophrenia, anxiety, posttraumatic stress disorder, or a substance use disorder are at high risk of suicide, and many never receive the timely intervention that might save their life.
Our national blind spot
It is poignant that the CDC report was released in spring: The rate of suicide is highest in April and May, when the light-dark cycle is reversed. This springtime peak runs contrary to the common belief that the rate of suicide is highest during winter months. The Annual Meeting of the American Psychiatric Association convenes in May, such that, ironically, thousands of psychiatrists are away from their office exactly when their patients might need them most
Lack of attention to the high risk of suicide among all ages and both sexes is emblematic of society’s inexplicable neglect of the needs of the mentally ill. That neglect is fueled, and exacerbated, by the destructive stigma attached to brain disorders that display psychiatric symptoms. As a neuropsychiatrist, I label this neglect societal anosognosia—the same as the lack of insight seen in patients with acute schizophrenia, who are unaware of how impaired they are and insist that they are not sick. (Anosognosia also occurs in stroke patients who deny that their limb is paralyzed and insist that all is well.)
Loss of insight can have serious consequences for patients who lose the ability to monitor and evaluate their physical and mental health. Just as patients with anosognosia think they do not need help, a society that fails to attend to the mental illness of its citizens endangers their overall health and welfare.
From neglect of mental illness many hazards arise
Tens of millions of Americans suffer from mental illness, according to the National Institute of Mental Health-sponsored Epidemiologic Catchment Area Study.4 The last thing these people can afford is societal anosognosia, which deprives them of necessary and timely access to psychiatric care.
Societal anosognosia is associated with numerous hazards for persons with mental illness, including:
- Lack of compassion, which is readily available for people with a medical ailment (broken bones, cardiovascular disease, cancer).
- Lack of adequate, affordable health insurance and financial support, compared with what is available for non-psychiatric disorders.
- Shortage of publicly funded programs and mental health practitioners to provide prevention and intervention for those who consider ending their life during an episode of depression, psychosis, stress, or a panic attack.
- Allowing the stigma to continue unabated. Why are there strict laws about hate crimes, but not about stigma? Why does society continue to portray depression and anxiety as a personal weakness or failure, while patients with Parkinson’s disease or multiple sclerosis who have motor weakness are not stigmatized for their physical deficits?
- Transforming the seriously mentally ill into felons by arresting and jailing them because of erratic behavior—instead of hospitalizing them for the medical care they need. The transinstitutionalization of the mentally ill—from state hospitals to prisons—is one of the most shameful consequences of societal anosognosia, burdening our patients with the dual stigma of being a criminal and mentally ill.
- Turning a blind eye to abuses by insurance companies. More appalling is the perpetuation of restricted health coverage despite the passage of parity laws! Why are sensory and motor disorders of brain lesions covered fully, while the thought, emotional, and behavior disorders of the brain covered only partially?
- Consent laws that restrict psychiatrists from medicating acutely psychotic or depressed patients unless they consent—but no laws that restrict a cardiologist from immediately treating an unconscious heart attack patient who cannot consent, or an obtunded stroke patient who cannot communicate? The duration of untreated psychosis or depression has been shown repeatedly to have deleterious effects on brain tissue and functional outcomes, yet treatment of an acutely ill psychiatric patient is often delayed until a court order is obtained. When was the last time a court order was needed to treat an acute myocardial infarction?
- Failure to recognize that premature mortality (by approximately 25 years) is a devastating consequence of mental illness, whether from suicide or cardiometabolic risk factors due to smoking, substance use (often used to self-medicate because proper treatment is lacking), poor diet, and sedentary living.
- Failure to provide basic primary care to people with severe mental illness, and the much lower use of life-saving diagnostic and treatment procedures offered to these patients, compared with non-psychiatric patients.
- Inadequate funding for research on psychiatric disorders, compared with other medical disorders—even though direct and indirect costs of mental illness to society (hundreds of billions of dollars a year) far exceed costs of most medical disorders.
- Severe shortage of rehabilitation programs for the mentally ill, compared with many other medical disorders. Why does paralysis of the mind receive far less support than paralysis of the legs or arms?
The rising suicide rate reflects poorly on us
Societal anosognosia is a global scourge, affecting many underdeveloped countries. Why do developed nations, like ours, have the same blind spot for mental illness? Might ignorance and discrimination be universal?
The tragic rise in the rate of death by suicide in men and women, among all age groups, year after year, is stunningly incongruent when juxtaposed against the elimination of smallpox and other communicable diseases through a concerted societal effort to support scientific advances in vaccine development. Societal anosognosia appears to be selective: We have comprehensive insight about diseases of the body but not diseases of the mind.
The essence and soul of a society are the collective minds of its citizens, not their bodies. Societal anosognosia is a serious dysfunction of its mind, and a rising suicide rate is a symptom of that pathological dysfunction.
1. Curtin SC, Warner M, Hedegaard H, et al. Increase in suicide in the United States, 1999-2014. National Center for Health Statistics Data Brief No. 241. Atlanta, GA: National Center for Health Statistics, U.S. Department of Health and Human Services; 2016.
2. Ten leading causes of death by age group, United States – 2014. Centers for Disease Control and Prevention. http://www.cdc.gov/injury/images/lc-charts/leading_causes_of_death_age_group_2014_1050w760h.gif. Accessed May 20, 2016.
3. Morris M. Stemming the rising tide of suicide. Clinical Psychiatry News. http://www.clinicalpsychiatrynews.com/specialty-focus/depression/single-article-page/stemming-the-rising-tide-of-suicide/01cd45cabfc693bedb0e30bb6cb0b89e.html. Published April 26, 2016. Accessed May 13, 2016.
4. Robins LN, Regier DA, eds. Psychiatric disorders in America: the Epidemiologic Catchment Area Study. New York, NY: Free Press; 1990.
1. Curtin SC, Warner M, Hedegaard H, et al. Increase in suicide in the United States, 1999-2014. National Center for Health Statistics Data Brief No. 241. Atlanta, GA: National Center for Health Statistics, U.S. Department of Health and Human Services; 2016.
2. Ten leading causes of death by age group, United States – 2014. Centers for Disease Control and Prevention. http://www.cdc.gov/injury/images/lc-charts/leading_causes_of_death_age_group_2014_1050w760h.gif. Accessed May 20, 2016.
3. Morris M. Stemming the rising tide of suicide. Clinical Psychiatry News. http://www.clinicalpsychiatrynews.com/specialty-focus/depression/single-article-page/stemming-the-rising-tide-of-suicide/01cd45cabfc693bedb0e30bb6cb0b89e.html. Published April 26, 2016. Accessed May 13, 2016.
4. Robins LN, Regier DA, eds. Psychiatric disorders in America: the Epidemiologic Catchment Area Study. New York, NY: Free Press; 1990.
Start offering aspirin to pregnant women at high risk for preeclampsia
Obstetricians work diligently to anticipate, diagnose, and treat preeclampsia because the maternal and perinatal health burden of the disease is enormous. Many meta-analyses have reported that aspirin treatment of women at high risk for preeclampsia reduces the risk of developing the disease by about 10% to 23%.1–5 In addition, for women at high risk for preeclampsia, aspirin treatment reduces the risk of preterm birth and intrauterine growth restriction (IUGR). In your practice you should start offering aspirin to pregnant women at high risk for preeclampsia.
Aspirin reduces the risk of preeclampsia, preterm birth, and IUGRBased on the results of multiple meta-analyses of clinical trials involving more than 35,000 women, investigators consistently have concluded that aspirin treatment reduces the risk of preeclampsia in women at high risk for the disease.1–5 The magnitude of the effect is difficult to define with precision, but the risk reduction is likely in the range of 10% to 23%.1
In addition to reducing the risk of preeclampsia, aspirin also reduces the risk of 2 associated problems: preterm birth and IUGR. For preterm birth, the risk reduction is estimated to be in the range of 11% to 31%. For IUGR, the estimation for risk reduction is in the range of 7% to 24%.1 Although these benefits are modest, the burden of maternal and perinatal morbidity associated with preeclampsia is great, making even a modest benefit clinically significant.
Potential harms of aspirin treatmentIn the most recent meta-analysis from the US Preventive Services Task Force (USPSTF),1 low-dose aspirin treatment was associated with no significant perinatal or maternal harms, but rare harms could not be ruled out. A small increase in the risk of placental abruption was noted, but this increase did not reach significance (relative risk [RR], 1.17; 95% confidence interval [CI], 0.93–1.48).1 There was no increased risk of maternal postpartum hemorrhage or blood loss at delivery.1 In one meta-analysis, aspirin treatment did not increase the risk of newborn intracranial hemorrhage.1
Other potential adverse effects of aspirin treatment include maternal gastrointestinal bleeding and exacerbation of respiratory disorders such as asthma, but these effects have not been reported as significant associations in clinical trials of preeclampsia prevention.
Dueling recommendations: Restrictive or liberal use of aspirin?The American College of Obstetricians and Gynecologists (ACOG) recommends use of aspirin to prevent preeclampsia in women who have a personal history of early-onset preeclampsia with delivery before 34 weeks of gestation and in women with preeclampsia in 2 or more prior pregnancies.6 The restrictive ACOG guideline recommends aspirin treatment for a very small group of women. In one analysis, using the ACOG guideline, only 0.35% of all pregnant women would be eligible for treatment with aspirin to prevent preeclampsia.7
The USPSTF recommends that all pregnant women with one major risk factor for preeclampsia—including multifetal gestation, chronic hypertension, type 1 or 2 pregestational diabetes, renal disease, autoimmune disease, or prior personal history of preeclampsia—receive treatment with aspirin to prevent preeclampsia.8 The Task Force also recommends that women with multiple moderate risk factors for preeclampsia, such as nulliparity, body mass index greater than 30 kg/m2, family history of preeclampsia in a mother or sister, age 35 years or older, and certain sociodemographic risk factors (African American race, low socioeconomic status) also be offered aspirin treatment.
The USPSTF guideline advises aspirin treatment for many women. According to one analysis, the USPSTFguideline would result in approximately 24% of all pregnant women being offered aspirin treatment.7
The USPSTF guideline would result in 67 times more pregnant women being treated with aspirin than the ACOG guideline. The narrowly focused ACOG recommendation is problematic because it recommends against aspirin treatment in women who are at very high risk for developing preeclampsia, for example, a 41-year-old woman in her first pregnancy with twins and pregestational diabetes. In addition, the ACOG recommendation is not consistent with the recommendations of most other major health organizations.
The World Health Organization,9 the United Kingdom’s National Institute for Health and Care Excellence (NICE),10 and the Society of Obstetricians and Gynaecologists of Canada11 all recommend aspirin treatment to prevent preeclampsia in pregnant women at high risk for the disease and utilize an expanded definition of “high risk” (TABLE). Some experts have observed that, in actual clinical practice, it is often difficult to consistently implement a prevention plan based on a complex assessment of clinical risk factors.7
An alternative to guidelines that use clinical risk factors to identify women at high risk is universal treatment. With universal treatment all pregnant women are prescribed aspirin, thereby maximizing the clinical benefit but unnecessarily treating many women with aspirin.7 Universal treatment of pregnant women with aspirin appears to be cost-effective and would be associated with annual health care savings of $365 million.7
Timing of aspirin initiationIn one meta-analysis, initiating aspirin before 16 weeks’ gestation resulted in a greater reduction in preeclampsia than starting aspirin after 16 weeks.12 The USPSTF cautions that meta-analysis of the available data is not well suited for identifying the optimal time to initiate aspirin therapy.13 ACOG, USPSTF, and NICE recommend initiating aspirin therapy at approximately 12 weeks’ gestation—the end of the first trimester.
Ideal aspirin doseThe optimal dose of aspirin to prevent preeclampsia is not precisely defined. Aspirin doses ranging from 50 mg to 162 mg have been proposed for the prevention of preeclampsia. Most authorities recommend a daily dose between 80 mg and less than 300 mg to prevent preeclampsia.14 ACOG and USPSTF recommend aspirin at a dose of 81 mg daily,6,8 because this dose is widely available in the United States.
Let’s close the gap between current and optimal practiceAccording to the USPSTF guidelines, approximately 24% of the pregnant women in our practices have risk factors that would justify the initiation of aspirin treatment for the prevention of preeclampsia.8 This approach would modestly reduce the rate of preeclampsia and the associated problems of preterm birth and IUGR with little cost and few adverse effects. Yet relatively few pregnant women in the United States are currently receiving aspirin therapy. We could close this clinical gap between current and optimal practice by reflecting on the USPSTF recommendations and implementing them in our practices, as appropriate.
Tell us…What are your thoughts about the use of aspirin in pregnant women who are at high risk for preeclampsia?
Send your letter to the editor to [email protected]. Please include the city and state in which you practice.
- Henderson JT, Whitlock EP, O'Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2014;160(10):695-703.
- Roberge S, Nicolaides KH, Demers S, Villa P, Bujold E. Prevention of perinatal death and adverse perinatal outcome using low-dose aspirin: a meta-analysis. Ultrasound Obstet Gynecol. 2013;41(5):491-499.
- Bujold E, Roberge S, Lacasse Y, et al. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: a meta-analysis. Obstet Gynecol. 2010;116(2 pt 1):402-414.
- Duley L, Henderson-Smart DJ, Meher S, King JF. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2007;(2):CD004659.
- Askie LM, Duley L, Henderson-Smart DJ, Stewart LA; PARIS Collaborative Group. Antiplatelet agents for prevention of pre-eclampsia: a meta-analysis of individual patient data. Lancet. 2007;369(9575):1791-1798.
- American College of Obstetricians and Gynecologists, Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122(5):1122-1131.
- Werner EF, Hauspurg AK, Rouse DJ. A cost-benefit analysis of low-dose aspirin prophylaxis for the prevention of preeclampsia in the United States. Obstet Gynecol. 2015;126(6):1242-1250.
- LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819-826.
- World Health Organization. WHO recommendations for prevention and treatment of pre-eclampsia and eclampsia. Geneva, Switzerland: WHO; 2011:13-15. https://www.preeclampsia.org/images/pdf/2011c-who_pe_final.pdf. Accessed January 4, 2016.
- National Institute for Health and Care Excellence. Hypertension in pregnancy: diagnosis and management. Clinical guideline 107. Manchester, United Kingdom: NICE; 2010:7. https://www.nice.org.uk/guidance/cg107/resources/hypertension-in-pregnancy-diagnosis-and-management-35109334009285. Accessed April 4, 2016.
- Magee LA, Pels A, Helewa M, Rey E, von Dadelszen P; Canadian Hypertensive Disorders of Pregnancy Working Group. Diagnosis, evaluation, and management of hypertensive disorders of pregnancy: executive summary. J Obstet Gynaecol Can. 2014;36(5):416-441.
- Roberge S, Demers S, Bujold E. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia [letter to the editor]. Ann Intern Med. 2014;161(8):613.
- Henderson JT, O'Connor E, Whitlock EP. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia [letter to the editor]. Ann Intern Med. 2014;161(8):613-614.
- Bujold E, Roberge S, Nicolaides KH. Low-dose aspirin for prevention of adverse outcomes related to abnormal placentation. Prenat Diagn. 2014;34(7):642-648.
Obstetricians work diligently to anticipate, diagnose, and treat preeclampsia because the maternal and perinatal health burden of the disease is enormous. Many meta-analyses have reported that aspirin treatment of women at high risk for preeclampsia reduces the risk of developing the disease by about 10% to 23%.1–5 In addition, for women at high risk for preeclampsia, aspirin treatment reduces the risk of preterm birth and intrauterine growth restriction (IUGR). In your practice you should start offering aspirin to pregnant women at high risk for preeclampsia.
Aspirin reduces the risk of preeclampsia, preterm birth, and IUGRBased on the results of multiple meta-analyses of clinical trials involving more than 35,000 women, investigators consistently have concluded that aspirin treatment reduces the risk of preeclampsia in women at high risk for the disease.1–5 The magnitude of the effect is difficult to define with precision, but the risk reduction is likely in the range of 10% to 23%.1
In addition to reducing the risk of preeclampsia, aspirin also reduces the risk of 2 associated problems: preterm birth and IUGR. For preterm birth, the risk reduction is estimated to be in the range of 11% to 31%. For IUGR, the estimation for risk reduction is in the range of 7% to 24%.1 Although these benefits are modest, the burden of maternal and perinatal morbidity associated with preeclampsia is great, making even a modest benefit clinically significant.
Potential harms of aspirin treatmentIn the most recent meta-analysis from the US Preventive Services Task Force (USPSTF),1 low-dose aspirin treatment was associated with no significant perinatal or maternal harms, but rare harms could not be ruled out. A small increase in the risk of placental abruption was noted, but this increase did not reach significance (relative risk [RR], 1.17; 95% confidence interval [CI], 0.93–1.48).1 There was no increased risk of maternal postpartum hemorrhage or blood loss at delivery.1 In one meta-analysis, aspirin treatment did not increase the risk of newborn intracranial hemorrhage.1
Other potential adverse effects of aspirin treatment include maternal gastrointestinal bleeding and exacerbation of respiratory disorders such as asthma, but these effects have not been reported as significant associations in clinical trials of preeclampsia prevention.
Dueling recommendations: Restrictive or liberal use of aspirin?The American College of Obstetricians and Gynecologists (ACOG) recommends use of aspirin to prevent preeclampsia in women who have a personal history of early-onset preeclampsia with delivery before 34 weeks of gestation and in women with preeclampsia in 2 or more prior pregnancies.6 The restrictive ACOG guideline recommends aspirin treatment for a very small group of women. In one analysis, using the ACOG guideline, only 0.35% of all pregnant women would be eligible for treatment with aspirin to prevent preeclampsia.7
The USPSTF recommends that all pregnant women with one major risk factor for preeclampsia—including multifetal gestation, chronic hypertension, type 1 or 2 pregestational diabetes, renal disease, autoimmune disease, or prior personal history of preeclampsia—receive treatment with aspirin to prevent preeclampsia.8 The Task Force also recommends that women with multiple moderate risk factors for preeclampsia, such as nulliparity, body mass index greater than 30 kg/m2, family history of preeclampsia in a mother or sister, age 35 years or older, and certain sociodemographic risk factors (African American race, low socioeconomic status) also be offered aspirin treatment.
The USPSTF guideline advises aspirin treatment for many women. According to one analysis, the USPSTFguideline would result in approximately 24% of all pregnant women being offered aspirin treatment.7
The USPSTF guideline would result in 67 times more pregnant women being treated with aspirin than the ACOG guideline. The narrowly focused ACOG recommendation is problematic because it recommends against aspirin treatment in women who are at very high risk for developing preeclampsia, for example, a 41-year-old woman in her first pregnancy with twins and pregestational diabetes. In addition, the ACOG recommendation is not consistent with the recommendations of most other major health organizations.
The World Health Organization,9 the United Kingdom’s National Institute for Health and Care Excellence (NICE),10 and the Society of Obstetricians and Gynaecologists of Canada11 all recommend aspirin treatment to prevent preeclampsia in pregnant women at high risk for the disease and utilize an expanded definition of “high risk” (TABLE). Some experts have observed that, in actual clinical practice, it is often difficult to consistently implement a prevention plan based on a complex assessment of clinical risk factors.7

An alternative to guidelines that use clinical risk factors to identify women at high risk is universal treatment. With universal treatment all pregnant women are prescribed aspirin, thereby maximizing the clinical benefit but unnecessarily treating many women with aspirin.7 Universal treatment of pregnant women with aspirin appears to be cost-effective and would be associated with annual health care savings of $365 million.7
Timing of aspirin initiationIn one meta-analysis, initiating aspirin before 16 weeks’ gestation resulted in a greater reduction in preeclampsia than starting aspirin after 16 weeks.12 The USPSTF cautions that meta-analysis of the available data is not well suited for identifying the optimal time to initiate aspirin therapy.13 ACOG, USPSTF, and NICE recommend initiating aspirin therapy at approximately 12 weeks’ gestation—the end of the first trimester.
Ideal aspirin doseThe optimal dose of aspirin to prevent preeclampsia is not precisely defined. Aspirin doses ranging from 50 mg to 162 mg have been proposed for the prevention of preeclampsia. Most authorities recommend a daily dose between 80 mg and less than 300 mg to prevent preeclampsia.14 ACOG and USPSTF recommend aspirin at a dose of 81 mg daily,6,8 because this dose is widely available in the United States.
Let’s close the gap between current and optimal practiceAccording to the USPSTF guidelines, approximately 24% of the pregnant women in our practices have risk factors that would justify the initiation of aspirin treatment for the prevention of preeclampsia.8 This approach would modestly reduce the rate of preeclampsia and the associated problems of preterm birth and IUGR with little cost and few adverse effects. Yet relatively few pregnant women in the United States are currently receiving aspirin therapy. We could close this clinical gap between current and optimal practice by reflecting on the USPSTF recommendations and implementing them in our practices, as appropriate.
Tell us…What are your thoughts about the use of aspirin in pregnant women who are at high risk for preeclampsia?
Send your letter to the editor to [email protected]. Please include the city and state in which you practice.
Obstetricians work diligently to anticipate, diagnose, and treat preeclampsia because the maternal and perinatal health burden of the disease is enormous. Many meta-analyses have reported that aspirin treatment of women at high risk for preeclampsia reduces the risk of developing the disease by about 10% to 23%.1–5 In addition, for women at high risk for preeclampsia, aspirin treatment reduces the risk of preterm birth and intrauterine growth restriction (IUGR). In your practice you should start offering aspirin to pregnant women at high risk for preeclampsia.
Aspirin reduces the risk of preeclampsia, preterm birth, and IUGRBased on the results of multiple meta-analyses of clinical trials involving more than 35,000 women, investigators consistently have concluded that aspirin treatment reduces the risk of preeclampsia in women at high risk for the disease.1–5 The magnitude of the effect is difficult to define with precision, but the risk reduction is likely in the range of 10% to 23%.1
In addition to reducing the risk of preeclampsia, aspirin also reduces the risk of 2 associated problems: preterm birth and IUGR. For preterm birth, the risk reduction is estimated to be in the range of 11% to 31%. For IUGR, the estimation for risk reduction is in the range of 7% to 24%.1 Although these benefits are modest, the burden of maternal and perinatal morbidity associated with preeclampsia is great, making even a modest benefit clinically significant.
Potential harms of aspirin treatmentIn the most recent meta-analysis from the US Preventive Services Task Force (USPSTF),1 low-dose aspirin treatment was associated with no significant perinatal or maternal harms, but rare harms could not be ruled out. A small increase in the risk of placental abruption was noted, but this increase did not reach significance (relative risk [RR], 1.17; 95% confidence interval [CI], 0.93–1.48).1 There was no increased risk of maternal postpartum hemorrhage or blood loss at delivery.1 In one meta-analysis, aspirin treatment did not increase the risk of newborn intracranial hemorrhage.1
Other potential adverse effects of aspirin treatment include maternal gastrointestinal bleeding and exacerbation of respiratory disorders such as asthma, but these effects have not been reported as significant associations in clinical trials of preeclampsia prevention.
Dueling recommendations: Restrictive or liberal use of aspirin?The American College of Obstetricians and Gynecologists (ACOG) recommends use of aspirin to prevent preeclampsia in women who have a personal history of early-onset preeclampsia with delivery before 34 weeks of gestation and in women with preeclampsia in 2 or more prior pregnancies.6 The restrictive ACOG guideline recommends aspirin treatment for a very small group of women. In one analysis, using the ACOG guideline, only 0.35% of all pregnant women would be eligible for treatment with aspirin to prevent preeclampsia.7
The USPSTF recommends that all pregnant women with one major risk factor for preeclampsia—including multifetal gestation, chronic hypertension, type 1 or 2 pregestational diabetes, renal disease, autoimmune disease, or prior personal history of preeclampsia—receive treatment with aspirin to prevent preeclampsia.8 The Task Force also recommends that women with multiple moderate risk factors for preeclampsia, such as nulliparity, body mass index greater than 30 kg/m2, family history of preeclampsia in a mother or sister, age 35 years or older, and certain sociodemographic risk factors (African American race, low socioeconomic status) also be offered aspirin treatment.
The USPSTF guideline advises aspirin treatment for many women. According to one analysis, the USPSTFguideline would result in approximately 24% of all pregnant women being offered aspirin treatment.7
The USPSTF guideline would result in 67 times more pregnant women being treated with aspirin than the ACOG guideline. The narrowly focused ACOG recommendation is problematic because it recommends against aspirin treatment in women who are at very high risk for developing preeclampsia, for example, a 41-year-old woman in her first pregnancy with twins and pregestational diabetes. In addition, the ACOG recommendation is not consistent with the recommendations of most other major health organizations.
The World Health Organization,9 the United Kingdom’s National Institute for Health and Care Excellence (NICE),10 and the Society of Obstetricians and Gynaecologists of Canada11 all recommend aspirin treatment to prevent preeclampsia in pregnant women at high risk for the disease and utilize an expanded definition of “high risk” (TABLE). Some experts have observed that, in actual clinical practice, it is often difficult to consistently implement a prevention plan based on a complex assessment of clinical risk factors.7

An alternative to guidelines that use clinical risk factors to identify women at high risk is universal treatment. With universal treatment all pregnant women are prescribed aspirin, thereby maximizing the clinical benefit but unnecessarily treating many women with aspirin.7 Universal treatment of pregnant women with aspirin appears to be cost-effective and would be associated with annual health care savings of $365 million.7
Timing of aspirin initiationIn one meta-analysis, initiating aspirin before 16 weeks’ gestation resulted in a greater reduction in preeclampsia than starting aspirin after 16 weeks.12 The USPSTF cautions that meta-analysis of the available data is not well suited for identifying the optimal time to initiate aspirin therapy.13 ACOG, USPSTF, and NICE recommend initiating aspirin therapy at approximately 12 weeks’ gestation—the end of the first trimester.
Ideal aspirin doseThe optimal dose of aspirin to prevent preeclampsia is not precisely defined. Aspirin doses ranging from 50 mg to 162 mg have been proposed for the prevention of preeclampsia. Most authorities recommend a daily dose between 80 mg and less than 300 mg to prevent preeclampsia.14 ACOG and USPSTF recommend aspirin at a dose of 81 mg daily,6,8 because this dose is widely available in the United States.
Let’s close the gap between current and optimal practiceAccording to the USPSTF guidelines, approximately 24% of the pregnant women in our practices have risk factors that would justify the initiation of aspirin treatment for the prevention of preeclampsia.8 This approach would modestly reduce the rate of preeclampsia and the associated problems of preterm birth and IUGR with little cost and few adverse effects. Yet relatively few pregnant women in the United States are currently receiving aspirin therapy. We could close this clinical gap between current and optimal practice by reflecting on the USPSTF recommendations and implementing them in our practices, as appropriate.
Tell us…What are your thoughts about the use of aspirin in pregnant women who are at high risk for preeclampsia?
Send your letter to the editor to [email protected]. Please include the city and state in which you practice.
- Henderson JT, Whitlock EP, O'Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2014;160(10):695-703.
- Roberge S, Nicolaides KH, Demers S, Villa P, Bujold E. Prevention of perinatal death and adverse perinatal outcome using low-dose aspirin: a meta-analysis. Ultrasound Obstet Gynecol. 2013;41(5):491-499.
- Bujold E, Roberge S, Lacasse Y, et al. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: a meta-analysis. Obstet Gynecol. 2010;116(2 pt 1):402-414.
- Duley L, Henderson-Smart DJ, Meher S, King JF. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2007;(2):CD004659.
- Askie LM, Duley L, Henderson-Smart DJ, Stewart LA; PARIS Collaborative Group. Antiplatelet agents for prevention of pre-eclampsia: a meta-analysis of individual patient data. Lancet. 2007;369(9575):1791-1798.
- American College of Obstetricians and Gynecologists, Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122(5):1122-1131.
- Werner EF, Hauspurg AK, Rouse DJ. A cost-benefit analysis of low-dose aspirin prophylaxis for the prevention of preeclampsia in the United States. Obstet Gynecol. 2015;126(6):1242-1250.
- LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819-826.
- World Health Organization. WHO recommendations for prevention and treatment of pre-eclampsia and eclampsia. Geneva, Switzerland: WHO; 2011:13-15. https://www.preeclampsia.org/images/pdf/2011c-who_pe_final.pdf. Accessed January 4, 2016.
- National Institute for Health and Care Excellence. Hypertension in pregnancy: diagnosis and management. Clinical guideline 107. Manchester, United Kingdom: NICE; 2010:7. https://www.nice.org.uk/guidance/cg107/resources/hypertension-in-pregnancy-diagnosis-and-management-35109334009285. Accessed April 4, 2016.
- Magee LA, Pels A, Helewa M, Rey E, von Dadelszen P; Canadian Hypertensive Disorders of Pregnancy Working Group. Diagnosis, evaluation, and management of hypertensive disorders of pregnancy: executive summary. J Obstet Gynaecol Can. 2014;36(5):416-441.
- Roberge S, Demers S, Bujold E. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia [letter to the editor]. Ann Intern Med. 2014;161(8):613.
- Henderson JT, O'Connor E, Whitlock EP. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia [letter to the editor]. Ann Intern Med. 2014;161(8):613-614.
- Bujold E, Roberge S, Nicolaides KH. Low-dose aspirin for prevention of adverse outcomes related to abnormal placentation. Prenat Diagn. 2014;34(7):642-648.
- Henderson JT, Whitlock EP, O'Connor E, Senger CA, Thompson JH, Rowland MG. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2014;160(10):695-703.
- Roberge S, Nicolaides KH, Demers S, Villa P, Bujold E. Prevention of perinatal death and adverse perinatal outcome using low-dose aspirin: a meta-analysis. Ultrasound Obstet Gynecol. 2013;41(5):491-499.
- Bujold E, Roberge S, Lacasse Y, et al. Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: a meta-analysis. Obstet Gynecol. 2010;116(2 pt 1):402-414.
- Duley L, Henderson-Smart DJ, Meher S, King JF. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2007;(2):CD004659.
- Askie LM, Duley L, Henderson-Smart DJ, Stewart LA; PARIS Collaborative Group. Antiplatelet agents for prevention of pre-eclampsia: a meta-analysis of individual patient data. Lancet. 2007;369(9575):1791-1798.
- American College of Obstetricians and Gynecologists, Task Force on Hypertension in Pregnancy. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists' Task Force on Hypertension in Pregnancy. Obstet Gynecol. 2013;122(5):1122-1131.
- Werner EF, Hauspurg AK, Rouse DJ. A cost-benefit analysis of low-dose aspirin prophylaxis for the prevention of preeclampsia in the United States. Obstet Gynecol. 2015;126(6):1242-1250.
- LeFevre ML; US Preventive Services Task Force. Low-dose aspirin use for the prevention of morbidity and mortality from preeclampsia: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;161(11):819-826.
- World Health Organization. WHO recommendations for prevention and treatment of pre-eclampsia and eclampsia. Geneva, Switzerland: WHO; 2011:13-15. https://www.preeclampsia.org/images/pdf/2011c-who_pe_final.pdf. Accessed January 4, 2016.
- National Institute for Health and Care Excellence. Hypertension in pregnancy: diagnosis and management. Clinical guideline 107. Manchester, United Kingdom: NICE; 2010:7. https://www.nice.org.uk/guidance/cg107/resources/hypertension-in-pregnancy-diagnosis-and-management-35109334009285. Accessed April 4, 2016.
- Magee LA, Pels A, Helewa M, Rey E, von Dadelszen P; Canadian Hypertensive Disorders of Pregnancy Working Group. Diagnosis, evaluation, and management of hypertensive disorders of pregnancy: executive summary. J Obstet Gynaecol Can. 2014;36(5):416-441.
- Roberge S, Demers S, Bujold E. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia [letter to the editor]. Ann Intern Med. 2014;161(8):613.
- Henderson JT, O'Connor E, Whitlock EP. Low-dose aspirin for prevention of morbidity and mortality from preeclampsia [letter to the editor]. Ann Intern Med. 2014;161(8):613-614.
- Bujold E, Roberge S, Nicolaides KH. Low-dose aspirin for prevention of adverse outcomes related to abnormal placentation. Prenat Diagn. 2014;34(7):642-648.
Predicting is tough, especially about the future
Quinn and Fang, in this issue of the Journal discuss efforts to predict bleeding complications associated with anticoagulant therapy in elderly patients. They note, as others have suggested, that we may fear the risk of severe anticoagulant-associated bleeding more than is warranted based on the data. The level of that fear and the risk of bleeding depend on the specific need for anticoagulant therapy in a given patient and on the risk of serious adverse outcomes from thrombosis that the anticoagulation is supposed to prevent. All prediction models are based on an “average” patient with certain characteristics. But of course none of our patients are average.
The studies Quinn and Fang discuss focus on vitamin K antagonist therapy. There is probably not enough practice-based or trial-based evidence yet to evaluate the risks associated with the new generation of anticoagulants.
All prediction models have limitations. The recent discussion on establishing a risk-based strategy to guide institution of lipid-lowering therapy highlights the challenges inherent in trying to base therapeutic decisions on predictive models. But however imperfect, models are still widely used to predict fracture risk in patients being considered for bone antiresorptive therapy and to predict the need for anticoagulation therapy or further diagnostic testing in patients with potential deep vein thrombosis or atrial fibrillation.
The decision to start anticoagulation in an elderly patient is often informed by the possibility of an easily recognized and feared risk factor for bleeding complications—falling. Falls are certainly important and are a major contributor to subdural hematoma and complicated hip fracture. But there are more common causes of severe bleeding complications that are less easily predicted by functional assessment of the patient. Nonetheless, fall risk can be lessened by prescribing exercise programs such as tai chi to improve balance, limiting the use of drugs associated with falls in the elderly, perhaps correcting hyponatremia, and testing for orthostatic hypotension as part of the physical examination. (Mild compression stockings and medication adjustment may reduce orthostasis.) Some of these interventions are easily accomplished, and probably should be done with all of our elderly and frail patients.
As we build more risk calculators into our electronic medical records, we must continue to consider their limitations as well as their specific utility. To paraphrase Yogi Berra, making predictions is tough, especially about the future.
Quinn and Fang, in this issue of the Journal discuss efforts to predict bleeding complications associated with anticoagulant therapy in elderly patients. They note, as others have suggested, that we may fear the risk of severe anticoagulant-associated bleeding more than is warranted based on the data. The level of that fear and the risk of bleeding depend on the specific need for anticoagulant therapy in a given patient and on the risk of serious adverse outcomes from thrombosis that the anticoagulation is supposed to prevent. All prediction models are based on an “average” patient with certain characteristics. But of course none of our patients are average.
The studies Quinn and Fang discuss focus on vitamin K antagonist therapy. There is probably not enough practice-based or trial-based evidence yet to evaluate the risks associated with the new generation of anticoagulants.
All prediction models have limitations. The recent discussion on establishing a risk-based strategy to guide institution of lipid-lowering therapy highlights the challenges inherent in trying to base therapeutic decisions on predictive models. But however imperfect, models are still widely used to predict fracture risk in patients being considered for bone antiresorptive therapy and to predict the need for anticoagulation therapy or further diagnostic testing in patients with potential deep vein thrombosis or atrial fibrillation.
The decision to start anticoagulation in an elderly patient is often informed by the possibility of an easily recognized and feared risk factor for bleeding complications—falling. Falls are certainly important and are a major contributor to subdural hematoma and complicated hip fracture. But there are more common causes of severe bleeding complications that are less easily predicted by functional assessment of the patient. Nonetheless, fall risk can be lessened by prescribing exercise programs such as tai chi to improve balance, limiting the use of drugs associated with falls in the elderly, perhaps correcting hyponatremia, and testing for orthostatic hypotension as part of the physical examination. (Mild compression stockings and medication adjustment may reduce orthostasis.) Some of these interventions are easily accomplished, and probably should be done with all of our elderly and frail patients.
As we build more risk calculators into our electronic medical records, we must continue to consider their limitations as well as their specific utility. To paraphrase Yogi Berra, making predictions is tough, especially about the future.
Quinn and Fang, in this issue of the Journal discuss efforts to predict bleeding complications associated with anticoagulant therapy in elderly patients. They note, as others have suggested, that we may fear the risk of severe anticoagulant-associated bleeding more than is warranted based on the data. The level of that fear and the risk of bleeding depend on the specific need for anticoagulant therapy in a given patient and on the risk of serious adverse outcomes from thrombosis that the anticoagulation is supposed to prevent. All prediction models are based on an “average” patient with certain characteristics. But of course none of our patients are average.
The studies Quinn and Fang discuss focus on vitamin K antagonist therapy. There is probably not enough practice-based or trial-based evidence yet to evaluate the risks associated with the new generation of anticoagulants.
All prediction models have limitations. The recent discussion on establishing a risk-based strategy to guide institution of lipid-lowering therapy highlights the challenges inherent in trying to base therapeutic decisions on predictive models. But however imperfect, models are still widely used to predict fracture risk in patients being considered for bone antiresorptive therapy and to predict the need for anticoagulation therapy or further diagnostic testing in patients with potential deep vein thrombosis or atrial fibrillation.
The decision to start anticoagulation in an elderly patient is often informed by the possibility of an easily recognized and feared risk factor for bleeding complications—falling. Falls are certainly important and are a major contributor to subdural hematoma and complicated hip fracture. But there are more common causes of severe bleeding complications that are less easily predicted by functional assessment of the patient. Nonetheless, fall risk can be lessened by prescribing exercise programs such as tai chi to improve balance, limiting the use of drugs associated with falls in the elderly, perhaps correcting hyponatremia, and testing for orthostatic hypotension as part of the physical examination. (Mild compression stockings and medication adjustment may reduce orthostasis.) Some of these interventions are easily accomplished, and probably should be done with all of our elderly and frail patients.
As we build more risk calculators into our electronic medical records, we must continue to consider their limitations as well as their specific utility. To paraphrase Yogi Berra, making predictions is tough, especially about the future.
Change, challenge, and a farewell
‘Druggable’ genes, promiscuous drugs, repurposed medications
Unprecedented collaboration among 900 genetics investigators across 40 countries led to creation of the highly productive Psychiatric Genomics Consortium (PGC), which is analyzing 400,000 individual DNA samples.1 The Consortium has an open-source approach, with data freely available to all who are interested.a
The PGC recently published the results of a large Genome Wide Association Study (GWAS) of 36,989 people with schizophrenia and 113,075 controls. Investigators discovered 108 genetic loci (each containing as many as 26 genes), adding up to 341 protein-coding risk genes for schizophrenia, distributed across all 23 chromosomes.2 One of these risk genes, on chromosome 6, is in the major histocompatibility complex and has the strongest association with schizophrenia (P = 10–31). This finding provides insight that schizophrenia might be related to immune dysfunction, supported by evidence for neuro-inflammation and elevated pro-inflammatory biomarkers in this syndrome.3
In addition to heritable risk genes, the PGC has found many copy number variants (CNVs) and rare de novo mutations that are found significantly more often (10-fold or greater) in schizophrenia. But, as reflected by the 50% concordance rate for schizophrenia in monozygotic twins, non-genetic pathways to schizophrenia obviously exist; this is especially so through adverse events during pregnancy, which can disrupt brain development in a manner similar to disruption caused by risk genes, CNVs, and mutations.
The most exciting consequence of these breakthroughs?These genetic discoveries have great implications for novel drug development for the hundreds of biological subtypes of schizophrenia. At latest count, 23,345 genes that code for proteins, the building blocks of the body, are found in the human set of 23 chromosomes.2 Approximately 7,000 of those genes are druggable and can open the way to developing new agents. In fact, identifying potential targets for pharmacotherapeutic intervention is the major goal of conducting a GWAS.4
What it means to be ‘druggable.’ Two conditions must be met for a gene to be druggable: First, it must code for a protein with folds that can interact with chemical compounds; second, that protein must be associated with a human disease.5 A drug that interacts with several target proteins (eg, kinases, proteases, transporters, enzymes) is considered promiscuous. After such a drug is found to have efficacy in 1 disease, it can be repurposed for treating other diseases. Such repositioning of an already approved drug for other conditions could save the pharmaceutical industry an enormous amount of time and billions of research and development dollars in developing new drugs for psychiatric illnesses that might have been used to treat various other medical conditions.
To exploit the principle of re-purposing, Lencz and Malhotra2 examined the 341 coding genes associated with schizophrenia, to determine whether available drugs interact with the proteins produced by some of those genes. They identified 40 druggable genes (11.7% of the 341) and reported that:
- 27 coding genes (7.92% of the 341) are drug targets6
- 20 of the 40 druggable genes are already approved by the FDA to treat a range of medical disorders, including glaucoma, epilepsy, hypertension, angina, irritable bowel syndrome, incontinence, smoking cessation, nausea, hypertension, prostate cancer, type 2 diabetes mellitus, pulmonary fibrosis, and acute promyelocytic leukemia; in addition, some genes act as a diuretic or an nonsteroidal anti-inflammatory drug
- another 20 druggable genes are not approved for use but are in clinical trials for disorders such as Alzheimer’s disease, heart failure, neuropathic pain, depression, cancer, immune-supported acne psoriasis, and myeloma.
The opportunity to repurpose some of those promiscuous drugs for various medical indications for the treatment of schizophrenia is exciting, and presents Pandora’s box of new mechanisms of action.7 It is intriguing how therapeutic mechanisms for a wide range of unrelated medical conditions may have commonality with the neurobiological underpinnings of a serious brain disorder such as schizophrenia.
Journey from genome to clinicPsychiatrists should be heartened by this translational research into the pharmacotherapeutic promise of emerging genetic advances. The parched terrain of psychopharmacology—the result of a drought of truly innovative medications for serious psychiatric brain disorders—soon may be drenched by a shower of translational discoveries from druggable genes.8 An auspicious scientific journey, from the genome to the clinic, has begun in earnest.
That is great news for our patients, and uplifting to us as well. Breakthroughs to cure intractable and persistent psychiatric brain disorders will not only vanquish disability and restore functioning, but also will be a powerful, long-awaited antidote to the virulent stigma of mental illness.
aAvailable at http://pgc.unc.edu/downloads.
1. Corvin A, Sullivan PF. What next in schizophrenia genetics for the Psychiatric Genomics Consortium [published online March 18, 2016]. Schizophr Bull. pii: sbw014.
2. Lencz T, Malhotra AK. Targeting the schizophrenia genome: a fast-track strategy from GWAS to clinic. Mol Psychiatry. 2015;20(7):820-826.
3. Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421-427.
4. Russ AP, Lampels S. The druggable genome: an update. Drug Discov Today. 2005;10(23-24):1607-1610.
5. Sakharkar MK, Sakharkar KR. Targetability of human disease genes. Curr Drug Discov Technol. 2007;4(1):48-58.
6. Rask-Anderson M, Masuram S, Schiöth HB. The druggable genome: evaluation of drug targets in clinical trials supports major shifts in molecular class and indication. Annu Rev Pharmacol Toxicol. 2014;54:9-26.
7. Hopkins AL, Groom CR. The druggable genome. Nat Rev Drug Discov. 2002;1(9):727-730.
8. Lipinski CA, Lombardo F, Dominy BW, et al. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev. 2001;46(1-3):3-26.
Unprecedented collaboration among 900 genetics investigators across 40 countries led to creation of the highly productive Psychiatric Genomics Consortium (PGC), which is analyzing 400,000 individual DNA samples.1 The Consortium has an open-source approach, with data freely available to all who are interested.a
The PGC recently published the results of a large Genome Wide Association Study (GWAS) of 36,989 people with schizophrenia and 113,075 controls. Investigators discovered 108 genetic loci (each containing as many as 26 genes), adding up to 341 protein-coding risk genes for schizophrenia, distributed across all 23 chromosomes.2 One of these risk genes, on chromosome 6, is in the major histocompatibility complex and has the strongest association with schizophrenia (P = 10–31). This finding provides insight that schizophrenia might be related to immune dysfunction, supported by evidence for neuro-inflammation and elevated pro-inflammatory biomarkers in this syndrome.3
In addition to heritable risk genes, the PGC has found many copy number variants (CNVs) and rare de novo mutations that are found significantly more often (10-fold or greater) in schizophrenia. But, as reflected by the 50% concordance rate for schizophrenia in monozygotic twins, non-genetic pathways to schizophrenia obviously exist; this is especially so through adverse events during pregnancy, which can disrupt brain development in a manner similar to disruption caused by risk genes, CNVs, and mutations.
The most exciting consequence of these breakthroughs?These genetic discoveries have great implications for novel drug development for the hundreds of biological subtypes of schizophrenia. At latest count, 23,345 genes that code for proteins, the building blocks of the body, are found in the human set of 23 chromosomes.2 Approximately 7,000 of those genes are druggable and can open the way to developing new agents. In fact, identifying potential targets for pharmacotherapeutic intervention is the major goal of conducting a GWAS.4
What it means to be ‘druggable.’ Two conditions must be met for a gene to be druggable: First, it must code for a protein with folds that can interact with chemical compounds; second, that protein must be associated with a human disease.5 A drug that interacts with several target proteins (eg, kinases, proteases, transporters, enzymes) is considered promiscuous. After such a drug is found to have efficacy in 1 disease, it can be repurposed for treating other diseases. Such repositioning of an already approved drug for other conditions could save the pharmaceutical industry an enormous amount of time and billions of research and development dollars in developing new drugs for psychiatric illnesses that might have been used to treat various other medical conditions.
To exploit the principle of re-purposing, Lencz and Malhotra2 examined the 341 coding genes associated with schizophrenia, to determine whether available drugs interact with the proteins produced by some of those genes. They identified 40 druggable genes (11.7% of the 341) and reported that:
- 27 coding genes (7.92% of the 341) are drug targets6
- 20 of the 40 druggable genes are already approved by the FDA to treat a range of medical disorders, including glaucoma, epilepsy, hypertension, angina, irritable bowel syndrome, incontinence, smoking cessation, nausea, hypertension, prostate cancer, type 2 diabetes mellitus, pulmonary fibrosis, and acute promyelocytic leukemia; in addition, some genes act as a diuretic or an nonsteroidal anti-inflammatory drug
- another 20 druggable genes are not approved for use but are in clinical trials for disorders such as Alzheimer’s disease, heart failure, neuropathic pain, depression, cancer, immune-supported acne psoriasis, and myeloma.
The opportunity to repurpose some of those promiscuous drugs for various medical indications for the treatment of schizophrenia is exciting, and presents Pandora’s box of new mechanisms of action.7 It is intriguing how therapeutic mechanisms for a wide range of unrelated medical conditions may have commonality with the neurobiological underpinnings of a serious brain disorder such as schizophrenia.
Journey from genome to clinicPsychiatrists should be heartened by this translational research into the pharmacotherapeutic promise of emerging genetic advances. The parched terrain of psychopharmacology—the result of a drought of truly innovative medications for serious psychiatric brain disorders—soon may be drenched by a shower of translational discoveries from druggable genes.8 An auspicious scientific journey, from the genome to the clinic, has begun in earnest.
That is great news for our patients, and uplifting to us as well. Breakthroughs to cure intractable and persistent psychiatric brain disorders will not only vanquish disability and restore functioning, but also will be a powerful, long-awaited antidote to the virulent stigma of mental illness.
aAvailable at http://pgc.unc.edu/downloads.
Unprecedented collaboration among 900 genetics investigators across 40 countries led to creation of the highly productive Psychiatric Genomics Consortium (PGC), which is analyzing 400,000 individual DNA samples.1 The Consortium has an open-source approach, with data freely available to all who are interested.a
The PGC recently published the results of a large Genome Wide Association Study (GWAS) of 36,989 people with schizophrenia and 113,075 controls. Investigators discovered 108 genetic loci (each containing as many as 26 genes), adding up to 341 protein-coding risk genes for schizophrenia, distributed across all 23 chromosomes.2 One of these risk genes, on chromosome 6, is in the major histocompatibility complex and has the strongest association with schizophrenia (P = 10–31). This finding provides insight that schizophrenia might be related to immune dysfunction, supported by evidence for neuro-inflammation and elevated pro-inflammatory biomarkers in this syndrome.3
In addition to heritable risk genes, the PGC has found many copy number variants (CNVs) and rare de novo mutations that are found significantly more often (10-fold or greater) in schizophrenia. But, as reflected by the 50% concordance rate for schizophrenia in monozygotic twins, non-genetic pathways to schizophrenia obviously exist; this is especially so through adverse events during pregnancy, which can disrupt brain development in a manner similar to disruption caused by risk genes, CNVs, and mutations.
The most exciting consequence of these breakthroughs?These genetic discoveries have great implications for novel drug development for the hundreds of biological subtypes of schizophrenia. At latest count, 23,345 genes that code for proteins, the building blocks of the body, are found in the human set of 23 chromosomes.2 Approximately 7,000 of those genes are druggable and can open the way to developing new agents. In fact, identifying potential targets for pharmacotherapeutic intervention is the major goal of conducting a GWAS.4
What it means to be ‘druggable.’ Two conditions must be met for a gene to be druggable: First, it must code for a protein with folds that can interact with chemical compounds; second, that protein must be associated with a human disease.5 A drug that interacts with several target proteins (eg, kinases, proteases, transporters, enzymes) is considered promiscuous. After such a drug is found to have efficacy in 1 disease, it can be repurposed for treating other diseases. Such repositioning of an already approved drug for other conditions could save the pharmaceutical industry an enormous amount of time and billions of research and development dollars in developing new drugs for psychiatric illnesses that might have been used to treat various other medical conditions.
To exploit the principle of re-purposing, Lencz and Malhotra2 examined the 341 coding genes associated with schizophrenia, to determine whether available drugs interact with the proteins produced by some of those genes. They identified 40 druggable genes (11.7% of the 341) and reported that:
- 27 coding genes (7.92% of the 341) are drug targets6
- 20 of the 40 druggable genes are already approved by the FDA to treat a range of medical disorders, including glaucoma, epilepsy, hypertension, angina, irritable bowel syndrome, incontinence, smoking cessation, nausea, hypertension, prostate cancer, type 2 diabetes mellitus, pulmonary fibrosis, and acute promyelocytic leukemia; in addition, some genes act as a diuretic or an nonsteroidal anti-inflammatory drug
- another 20 druggable genes are not approved for use but are in clinical trials for disorders such as Alzheimer’s disease, heart failure, neuropathic pain, depression, cancer, immune-supported acne psoriasis, and myeloma.
The opportunity to repurpose some of those promiscuous drugs for various medical indications for the treatment of schizophrenia is exciting, and presents Pandora’s box of new mechanisms of action.7 It is intriguing how therapeutic mechanisms for a wide range of unrelated medical conditions may have commonality with the neurobiological underpinnings of a serious brain disorder such as schizophrenia.
Journey from genome to clinicPsychiatrists should be heartened by this translational research into the pharmacotherapeutic promise of emerging genetic advances. The parched terrain of psychopharmacology—the result of a drought of truly innovative medications for serious psychiatric brain disorders—soon may be drenched by a shower of translational discoveries from druggable genes.8 An auspicious scientific journey, from the genome to the clinic, has begun in earnest.
That is great news for our patients, and uplifting to us as well. Breakthroughs to cure intractable and persistent psychiatric brain disorders will not only vanquish disability and restore functioning, but also will be a powerful, long-awaited antidote to the virulent stigma of mental illness.
aAvailable at http://pgc.unc.edu/downloads.
1. Corvin A, Sullivan PF. What next in schizophrenia genetics for the Psychiatric Genomics Consortium [published online March 18, 2016]. Schizophr Bull. pii: sbw014.
2. Lencz T, Malhotra AK. Targeting the schizophrenia genome: a fast-track strategy from GWAS to clinic. Mol Psychiatry. 2015;20(7):820-826.
3. Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421-427.
4. Russ AP, Lampels S. The druggable genome: an update. Drug Discov Today. 2005;10(23-24):1607-1610.
5. Sakharkar MK, Sakharkar KR. Targetability of human disease genes. Curr Drug Discov Technol. 2007;4(1):48-58.
6. Rask-Anderson M, Masuram S, Schiöth HB. The druggable genome: evaluation of drug targets in clinical trials supports major shifts in molecular class and indication. Annu Rev Pharmacol Toxicol. 2014;54:9-26.
7. Hopkins AL, Groom CR. The druggable genome. Nat Rev Drug Discov. 2002;1(9):727-730.
8. Lipinski CA, Lombardo F, Dominy BW, et al. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev. 2001;46(1-3):3-26.
1. Corvin A, Sullivan PF. What next in schizophrenia genetics for the Psychiatric Genomics Consortium [published online March 18, 2016]. Schizophr Bull. pii: sbw014.
2. Lencz T, Malhotra AK. Targeting the schizophrenia genome: a fast-track strategy from GWAS to clinic. Mol Psychiatry. 2015;20(7):820-826.
3. Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511(7510):421-427.
4. Russ AP, Lampels S. The druggable genome: an update. Drug Discov Today. 2005;10(23-24):1607-1610.
5. Sakharkar MK, Sakharkar KR. Targetability of human disease genes. Curr Drug Discov Technol. 2007;4(1):48-58.
6. Rask-Anderson M, Masuram S, Schiöth HB. The druggable genome: evaluation of drug targets in clinical trials supports major shifts in molecular class and indication. Annu Rev Pharmacol Toxicol. 2014;54:9-26.
7. Hopkins AL, Groom CR. The druggable genome. Nat Rev Drug Discov. 2002;1(9):727-730.
8. Lipinski CA, Lombardo F, Dominy BW, et al. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev. 2001;46(1-3):3-26.