User login
Real-world safety, efficacy found for fecal transplants
Fecal microbiota transplantation (FMT) appears safe and effective as a treatment for most Clostridioides difficile infections as it is currently being administered, researchers say.
“We actually didn’t see any infections that were definitely transmissible via fecal transplant,” Colleen Kelly, MD, an associate professor of medicine at Brown University, Providence, R.I., said in an interview.
The findings, published online Oct. 1 in the journal Gastroenterology, come from the American Gastroenterological Association (AGA) NIH-funded FMT National Registry and could allay concerns about a treatment that has yet to gain full approval by the Food and Drug Administration, despite successful clinical trials.
C. diff infections are common and increasing in the United States, often can’t be cured with conventional treatments such as antibiotics, and can be deadly.
Transplanting fecal matter from a donor to the patient appears to work by restoring beneficial microorganisms to the patient’s gut. The procedure is also under investigation for a wide range of other ailments, from irritable bowel syndrome to mood disorders.
But much remains unknown. Researchers have counted a thousand bacterial species along with viruses, bacteriophages, archaea, and fungi in the human gut that interact in complex ways, not all of them beneficial.
The FDA has not enforced regulations that would prohibit the procedure, but in March, it warned about infections with enteropathogenic Escherichia coli and Shiga toxin–producing E. coli following fecal transplants.
As a result of these reports, and the theoretical risk of spreading SARS-CoV-2, OpenBiome, the largest stool bank in the United States, has suspended shipments except for emergency orders, and asked clinicians to quarantine any of its products they already have on hand.
In the meantime, long-term effects of the treatment have not been well documented. And clinical trials have excluded patients who might benefit, such as those who have been immunocompromised or have inflammatory bowel disease.
National registry follows patients outside clinical trials
To better understand how patients fare outside these trials, AGA and other organizations developed a national registry, funded by a grant from the National Institute of Allergy and Infectious Diseases.
The current report summarizes results on 259 patients enrolled between Dec. 5, 2017, and Sept. 2, 2019 at 20 sites.
At baseline, 44% of these patients suffered moderate and 36% mild C. diff infections. The duration of the diagnosis ranged from less than 1 week to 9 years, with a median duration of 20 weeks. They ranged from 1 to 15 episodes with a mean of 3.5.
Almost all had received vancomycin, and 62% had at least two courses. About 40% had received metronidazole and 28% had received fidaxomicin.
Almost all participants received stool from an unknown donor, mostly from stool banks, with OpenBiome accounting for 67%. About 85% of the transplants were administered through colonoscopy and 6% by upper endoscopy.
Out of 222 patients who returned for a 1-month follow-up, 90% met the investigators’ definition of cure: resolution of diarrhea without need for further anti–C. diff therapy. About 98% received only one transplant. An intent to treat analysis produced a cure rate of 86%.
Results were good in patients with comorbidities, including 12% who had irritable bowel syndrome, 9% who had ulcerative colitis, and 7% who had Crohn’s disease, Dr. Kelly said. “I hope everybody sees the importance of it. In these patients that are more complicated, who may have underlying comorbidities, who may not have been in the clinical trials, it looks effective in that group, and also incredibly safe.”
She added that the risk of transmitting SARS-CoV-2 is minor. “I think it would be a very, very unlikely way for someone to get a respiratory pathogen.”
Of the 112 participants who were cured at 1 month and returned for follow-up after 6 months, 4 developed recurrent C. diff infection. Eleven patients who were not cured in the first month returned after 6 months. Of these, seven were reported cured at this later follow-up.
Three complications occurred as result of the procedure: one colonoscopic perforation and two episodes of gastrointestinal bleeding.
About 45% of participants reported at least one symptom, with diarrhea not related to C. difficile the most common, followed by abdominal pain, bloating, and constipation.
Eleven patients suffered infections, including two which the investigators thought might be related to the procedure: Bacteroides fragilis in one participant with severe diarrhea, and enteropathogenic E. coli in another with loose stools. Other infections included four urinary tract infections, three cases of pneumonia, one E. coli bacteremia and one tooth infection.
Within a month of the procedure, 27 patients were hospitalized, with 3 of these cases considered possibly related to the procedure.
Findings may not apply to all clinical settings
Vincent B. Young, MD, PhD, a professor of medicine and infectious diseases at the University of Michigan, Ann Arbor, pointed out that the findings might not apply to all clinical settings. The participating clinicians were almost all gastroenterologists working in academic centers.
“Most of them are not Joe Doctor at the doctor’s office,” said Dr. Young, who was not involved with the study. Clinicians in other specialties, such as infectious diseases, might be more inclined to administer fecal transplants through capsules rather than colonoscopies.
And he added that the study does not address effects of the transplant that might develop over years. “Some people talk about how changes in the microbiota lead to increased risk for long-term complications, things like cancer or heart disease. You’re not going to see those in 6 months.”
Also, the study didn’t yield any findings on indications other than C. diff. “In no way, shape, or form does it mean you can use it for autism, depression, heart disease, or [irritable bowel syndrome],” he said.
Still, he said, the study “confirms the fact that fecal cell transplantation is an effective treatment for recurrent C. diff infection when administered as they administered it.”
The National Institute of Allergy and Infectious Diseases funded the registry. Dr. Kelly reported a relationship with Finch Therapeutics. Dr. Young reports financial relationships with Vedanta Biosciences and Bio-K+.
This story was updated on Oct. 4, 2020.
A version of this article originally appeared on Medscape.com.
Fecal microbiota transplantation (FMT) appears safe and effective as a treatment for most Clostridioides difficile infections as it is currently being administered, researchers say.
“We actually didn’t see any infections that were definitely transmissible via fecal transplant,” Colleen Kelly, MD, an associate professor of medicine at Brown University, Providence, R.I., said in an interview.
The findings, published online Oct. 1 in the journal Gastroenterology, come from the American Gastroenterological Association (AGA) NIH-funded FMT National Registry and could allay concerns about a treatment that has yet to gain full approval by the Food and Drug Administration, despite successful clinical trials.
C. diff infections are common and increasing in the United States, often can’t be cured with conventional treatments such as antibiotics, and can be deadly.
Transplanting fecal matter from a donor to the patient appears to work by restoring beneficial microorganisms to the patient’s gut. The procedure is also under investigation for a wide range of other ailments, from irritable bowel syndrome to mood disorders.
But much remains unknown. Researchers have counted a thousand bacterial species along with viruses, bacteriophages, archaea, and fungi in the human gut that interact in complex ways, not all of them beneficial.
The FDA has not enforced regulations that would prohibit the procedure, but in March, it warned about infections with enteropathogenic Escherichia coli and Shiga toxin–producing E. coli following fecal transplants.
As a result of these reports, and the theoretical risk of spreading SARS-CoV-2, OpenBiome, the largest stool bank in the United States, has suspended shipments except for emergency orders, and asked clinicians to quarantine any of its products they already have on hand.
In the meantime, long-term effects of the treatment have not been well documented. And clinical trials have excluded patients who might benefit, such as those who have been immunocompromised or have inflammatory bowel disease.
National registry follows patients outside clinical trials
To better understand how patients fare outside these trials, AGA and other organizations developed a national registry, funded by a grant from the National Institute of Allergy and Infectious Diseases.
The current report summarizes results on 259 patients enrolled between Dec. 5, 2017, and Sept. 2, 2019 at 20 sites.
At baseline, 44% of these patients suffered moderate and 36% mild C. diff infections. The duration of the diagnosis ranged from less than 1 week to 9 years, with a median duration of 20 weeks. They ranged from 1 to 15 episodes with a mean of 3.5.
Almost all had received vancomycin, and 62% had at least two courses. About 40% had received metronidazole and 28% had received fidaxomicin.
Almost all participants received stool from an unknown donor, mostly from stool banks, with OpenBiome accounting for 67%. About 85% of the transplants were administered through colonoscopy and 6% by upper endoscopy.
Out of 222 patients who returned for a 1-month follow-up, 90% met the investigators’ definition of cure: resolution of diarrhea without need for further anti–C. diff therapy. About 98% received only one transplant. An intent to treat analysis produced a cure rate of 86%.
Results were good in patients with comorbidities, including 12% who had irritable bowel syndrome, 9% who had ulcerative colitis, and 7% who had Crohn’s disease, Dr. Kelly said. “I hope everybody sees the importance of it. In these patients that are more complicated, who may have underlying comorbidities, who may not have been in the clinical trials, it looks effective in that group, and also incredibly safe.”
She added that the risk of transmitting SARS-CoV-2 is minor. “I think it would be a very, very unlikely way for someone to get a respiratory pathogen.”
Of the 112 participants who were cured at 1 month and returned for follow-up after 6 months, 4 developed recurrent C. diff infection. Eleven patients who were not cured in the first month returned after 6 months. Of these, seven were reported cured at this later follow-up.
Three complications occurred as result of the procedure: one colonoscopic perforation and two episodes of gastrointestinal bleeding.
About 45% of participants reported at least one symptom, with diarrhea not related to C. difficile the most common, followed by abdominal pain, bloating, and constipation.
Eleven patients suffered infections, including two which the investigators thought might be related to the procedure: Bacteroides fragilis in one participant with severe diarrhea, and enteropathogenic E. coli in another with loose stools. Other infections included four urinary tract infections, three cases of pneumonia, one E. coli bacteremia and one tooth infection.
Within a month of the procedure, 27 patients were hospitalized, with 3 of these cases considered possibly related to the procedure.
Findings may not apply to all clinical settings
Vincent B. Young, MD, PhD, a professor of medicine and infectious diseases at the University of Michigan, Ann Arbor, pointed out that the findings might not apply to all clinical settings. The participating clinicians were almost all gastroenterologists working in academic centers.
“Most of them are not Joe Doctor at the doctor’s office,” said Dr. Young, who was not involved with the study. Clinicians in other specialties, such as infectious diseases, might be more inclined to administer fecal transplants through capsules rather than colonoscopies.
And he added that the study does not address effects of the transplant that might develop over years. “Some people talk about how changes in the microbiota lead to increased risk for long-term complications, things like cancer or heart disease. You’re not going to see those in 6 months.”
Also, the study didn’t yield any findings on indications other than C. diff. “In no way, shape, or form does it mean you can use it for autism, depression, heart disease, or [irritable bowel syndrome],” he said.
Still, he said, the study “confirms the fact that fecal cell transplantation is an effective treatment for recurrent C. diff infection when administered as they administered it.”
The National Institute of Allergy and Infectious Diseases funded the registry. Dr. Kelly reported a relationship with Finch Therapeutics. Dr. Young reports financial relationships with Vedanta Biosciences and Bio-K+.
This story was updated on Oct. 4, 2020.
A version of this article originally appeared on Medscape.com.
Fecal microbiota transplantation (FMT) appears safe and effective as a treatment for most Clostridioides difficile infections as it is currently being administered, researchers say.
“We actually didn’t see any infections that were definitely transmissible via fecal transplant,” Colleen Kelly, MD, an associate professor of medicine at Brown University, Providence, R.I., said in an interview.
The findings, published online Oct. 1 in the journal Gastroenterology, come from the American Gastroenterological Association (AGA) NIH-funded FMT National Registry and could allay concerns about a treatment that has yet to gain full approval by the Food and Drug Administration, despite successful clinical trials.
C. diff infections are common and increasing in the United States, often can’t be cured with conventional treatments such as antibiotics, and can be deadly.
Transplanting fecal matter from a donor to the patient appears to work by restoring beneficial microorganisms to the patient’s gut. The procedure is also under investigation for a wide range of other ailments, from irritable bowel syndrome to mood disorders.
But much remains unknown. Researchers have counted a thousand bacterial species along with viruses, bacteriophages, archaea, and fungi in the human gut that interact in complex ways, not all of them beneficial.
The FDA has not enforced regulations that would prohibit the procedure, but in March, it warned about infections with enteropathogenic Escherichia coli and Shiga toxin–producing E. coli following fecal transplants.
As a result of these reports, and the theoretical risk of spreading SARS-CoV-2, OpenBiome, the largest stool bank in the United States, has suspended shipments except for emergency orders, and asked clinicians to quarantine any of its products they already have on hand.
In the meantime, long-term effects of the treatment have not been well documented. And clinical trials have excluded patients who might benefit, such as those who have been immunocompromised or have inflammatory bowel disease.
National registry follows patients outside clinical trials
To better understand how patients fare outside these trials, AGA and other organizations developed a national registry, funded by a grant from the National Institute of Allergy and Infectious Diseases.
The current report summarizes results on 259 patients enrolled between Dec. 5, 2017, and Sept. 2, 2019 at 20 sites.
At baseline, 44% of these patients suffered moderate and 36% mild C. diff infections. The duration of the diagnosis ranged from less than 1 week to 9 years, with a median duration of 20 weeks. They ranged from 1 to 15 episodes with a mean of 3.5.
Almost all had received vancomycin, and 62% had at least two courses. About 40% had received metronidazole and 28% had received fidaxomicin.
Almost all participants received stool from an unknown donor, mostly from stool banks, with OpenBiome accounting for 67%. About 85% of the transplants were administered through colonoscopy and 6% by upper endoscopy.
Out of 222 patients who returned for a 1-month follow-up, 90% met the investigators’ definition of cure: resolution of diarrhea without need for further anti–C. diff therapy. About 98% received only one transplant. An intent to treat analysis produced a cure rate of 86%.
Results were good in patients with comorbidities, including 12% who had irritable bowel syndrome, 9% who had ulcerative colitis, and 7% who had Crohn’s disease, Dr. Kelly said. “I hope everybody sees the importance of it. In these patients that are more complicated, who may have underlying comorbidities, who may not have been in the clinical trials, it looks effective in that group, and also incredibly safe.”
She added that the risk of transmitting SARS-CoV-2 is minor. “I think it would be a very, very unlikely way for someone to get a respiratory pathogen.”
Of the 112 participants who were cured at 1 month and returned for follow-up after 6 months, 4 developed recurrent C. diff infection. Eleven patients who were not cured in the first month returned after 6 months. Of these, seven were reported cured at this later follow-up.
Three complications occurred as result of the procedure: one colonoscopic perforation and two episodes of gastrointestinal bleeding.
About 45% of participants reported at least one symptom, with diarrhea not related to C. difficile the most common, followed by abdominal pain, bloating, and constipation.
Eleven patients suffered infections, including two which the investigators thought might be related to the procedure: Bacteroides fragilis in one participant with severe diarrhea, and enteropathogenic E. coli in another with loose stools. Other infections included four urinary tract infections, three cases of pneumonia, one E. coli bacteremia and one tooth infection.
Within a month of the procedure, 27 patients were hospitalized, with 3 of these cases considered possibly related to the procedure.
Findings may not apply to all clinical settings
Vincent B. Young, MD, PhD, a professor of medicine and infectious diseases at the University of Michigan, Ann Arbor, pointed out that the findings might not apply to all clinical settings. The participating clinicians were almost all gastroenterologists working in academic centers.
“Most of them are not Joe Doctor at the doctor’s office,” said Dr. Young, who was not involved with the study. Clinicians in other specialties, such as infectious diseases, might be more inclined to administer fecal transplants through capsules rather than colonoscopies.
And he added that the study does not address effects of the transplant that might develop over years. “Some people talk about how changes in the microbiota lead to increased risk for long-term complications, things like cancer or heart disease. You’re not going to see those in 6 months.”
Also, the study didn’t yield any findings on indications other than C. diff. “In no way, shape, or form does it mean you can use it for autism, depression, heart disease, or [irritable bowel syndrome],” he said.
Still, he said, the study “confirms the fact that fecal cell transplantation is an effective treatment for recurrent C. diff infection when administered as they administered it.”
The National Institute of Allergy and Infectious Diseases funded the registry. Dr. Kelly reported a relationship with Finch Therapeutics. Dr. Young reports financial relationships with Vedanta Biosciences and Bio-K+.
This story was updated on Oct. 4, 2020.
A version of this article originally appeared on Medscape.com.
‘Celebration’ will be ‘short-lived’ if COVID vaccine rushed: Experts
on Wednesday.
The career staff of the Food and Drug Administration can be counted on to appropriately weigh whether a vaccine should be cleared for use in preventing COVID-19, witnesses, including Paul A. Offit, MD, of Children’s Hospital of Philadelphia, told the House Energy and Commerce Committee’s oversight and investigations panel.
FDA staffers would object to attempts by the Trump administration to rush a vaccine to the public without proper vetting, as would veteran federal researchers, including National Institutes of Health Director Francis S. Collins, MD, PhD, and Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, Offit said.
“If COVID-19 vaccines are released before they’re ready to be released, you will hear from these people, and you will also hear from people like Dr. Francis Collins and Tony Fauci, both of whom are trusted by the American public, as well as many other academicians and researchers who wouldn’t stand for this,” he said.
“The public is already nervous about these vaccines,” said Offit, who serves on key FDA and Centers for Disease Control and Prevention committees overseeing vaccine policy. “If trusted health officials stand up and decry a premature release, the celebration by the administration will be short-lived.”
Overly optimistic estimates about a potential approval can only serve to erode the public’s trust in these crucial vaccines, said another witness, Ashish K. Jha, MD, MPH, the dean of Brown University’s School of Public Health, in Providence, Rhode Island.
“All political leaders need to stop talking about things like time lines,” Jha told the lawmakers.
President Donald Trump has several times suggested that a COVID vaccine might be approved ahead of the November 3 election, where he faces a significant challenge from his Democratic rival, former Vice President Joe Biden.
In a Tuesday night debate with Biden, Trump again raised the idea of a quick approval. “Now we’re weeks away from a vaccine,” Trump said during the debate.
Trump’s estimates, though, are not in line with those offered by most firms involved with making vaccines. The most optimistic projections have come from Pfizer Inc. The drugmaker’s chief executive, Albert Bourla, has spoken about his company possibly having data to present to the FDA as early as late October about the safety and effectiveness of a vaccine.
In a September 8 interview with the Today show, Bourla said there was a 60% chance his company would meet that goal. In response to a question, he made it clear his comments applied to a potential Pfizer application, not an approval or release of a vaccine by that time.
In response to concerns about political pressures, the FDA in June issued guidance outlining what its staff would require for approval of a COVID-19 vaccine.
Pushback on politics
Another witness at the Wednesday hearing, Mark McClellan, MD, PhD, a former FDA commissioner (2002 – 2004), pushed back on objections to a potential release of further guidance from the agency.
“Some recent statements from the White House have implied that FDA’s plan to release additional written guidance on its expectations for emergency use authorization of a vaccine is unnecessarily raising the bar on regulatory standards for authorization,” said McClellan in his testimony for the House panel. “That is not the case.”
Instead, further FDA guidance would be a welcome form of feedback for the firms trying to develop COVID-19 vaccines, according to McClellan, who also serves on the board of directors for Johnson & Johnson. Johnson & Johnson is among the firms that have advanced a COVID-19 vaccine candidate to phase 3 testing. In his role as a director, he serves on the board’s regulatory compliance committee.
Along with politics, recent stumbles at FDA with emergency use authorizations (EUAs) of treatments for COVID-19 have eroded the public’s confidence in the agency, Jha told the House panel. The FDA approved hydroxychloroquine, a medicine promoted by Trump for use in COVID, under an EUA in March and then revoked this clearance in June.
Jha said the FDA’s most serious misstep was its handling of convalescent plasma, which was approved through an EUA on August 23 “in a highly advertised and widely televised announcement including the president.
“The announcement solidified in the public conversation the impression that, increasingly with this administration, politics are taking over trusted, nonpartisan scientific institutions,” he said in his testimony.
Approving a COVID-19 vaccine on the limited evidence through an EUA would mark a serious departure from FDA policy, according to Jha.
“While we sometimes accept a certain level of potential harm in experimental treatments for those who are severely ill, vaccines are given to healthy people and therefore need to have a substantially higher measure of safety and effectiveness,” he explained.
Jha said the FDA has only once before used this EUA approach for a vaccine. That was for a vaccine against inhaled anthrax and was mostly distributed to high-risk soldiers and civilians in war zones.
COVID-19, in contrast, is an infection that has changed lives around the world. The virus has contributed to more than 1 million deaths, including more than 200,000 in the United States, according to the World Health Organization.
Scientists are hoping vaccines will help curb this infection, although much of the future success of vaccines depends on how widely they are used, witnesses told the House panel.
Debate on approaches for vaccine effectiveness
In his testimony, Jha also noted concerns about COVID-19 vaccine trials. He included a reference to a Sept. 22 opinion article titled, “These Coronavirus Trials Don›t Answer the One Question We Need to Know,” which was written by Peter Doshi, PhD, of the University of Maryland School of Pharmacy, in Baltimore, and Eric Topol, MD, a professor of molecular medicine at Scripps Research in La Jolla, Calif. Topol is also editor in chief of Medscape.
Topol and Doshi questioned why the firms Moderna, Pfizer, and AstraZeneca structured their competing trials such that “a vaccine could meet the companies’ benchmark for success if it lowered the risk of mild Covid-19, but was never shown to reduce moderate or severe forms of the disease, or the risk of hospitalization, admissions to the intensive care unit or death.”
“To say a vaccine works should mean that most people no longer run the risk of getting seriously sick,” Topol and Doshi wrote. “That’s not what these trials will determine.”
There was disagreement about this point at the hearing. U.S. Representative Morgan Griffith (R-Va.) read the section of the Doshi-Topol article quoted above and asked one witness, Offit, to weigh in.
“Do you agree with those concerns? And either way, tell me why,” Griffith asked.
“I don’t agree,” Offit responded.
“I think it’s actually much harder to prevent asymptomatic infection or mildly symptomatic infection,” he said. “If you can prevent that, you are much more likely to prevent moderate to severe disease. So I think they have it backwards.”
But other researchers also question the approaches used with the current crop of COVID-19 vaccines.
“With the current protocols, it is conceivable that a vaccine might be considered effective – and eventually approved – based primarily on its ability to prevent mild cases alone,” wrote William Haseltine, PhD, president of the nonprofit ACCESS Health International, in a September 22 opinion article in the Washington Post titled: “Beware of COVID-19 Vaccine Trials Designed to Succeed From the Start.”
In an interview with Medscape Medical News on Wednesday, Haseltine said he maintains these concerns about the tests. Earlier in his career, he was a leader in HIV research through his lab at Harvard University in Cambridge, Massachusetts, and he subsequently led a biotech company, Human Genome Sciences.
He fears consumers will not get what they might expect from the vaccines being tested.
“What people care about is if this is going to keep them out of the hospital and will it keep them alive. And that’s not even part of this protocol,” Haseltine said.
This article first appeared on Medscape.com.
on Wednesday.
The career staff of the Food and Drug Administration can be counted on to appropriately weigh whether a vaccine should be cleared for use in preventing COVID-19, witnesses, including Paul A. Offit, MD, of Children’s Hospital of Philadelphia, told the House Energy and Commerce Committee’s oversight and investigations panel.
FDA staffers would object to attempts by the Trump administration to rush a vaccine to the public without proper vetting, as would veteran federal researchers, including National Institutes of Health Director Francis S. Collins, MD, PhD, and Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, Offit said.
“If COVID-19 vaccines are released before they’re ready to be released, you will hear from these people, and you will also hear from people like Dr. Francis Collins and Tony Fauci, both of whom are trusted by the American public, as well as many other academicians and researchers who wouldn’t stand for this,” he said.
“The public is already nervous about these vaccines,” said Offit, who serves on key FDA and Centers for Disease Control and Prevention committees overseeing vaccine policy. “If trusted health officials stand up and decry a premature release, the celebration by the administration will be short-lived.”
Overly optimistic estimates about a potential approval can only serve to erode the public’s trust in these crucial vaccines, said another witness, Ashish K. Jha, MD, MPH, the dean of Brown University’s School of Public Health, in Providence, Rhode Island.
“All political leaders need to stop talking about things like time lines,” Jha told the lawmakers.
President Donald Trump has several times suggested that a COVID vaccine might be approved ahead of the November 3 election, where he faces a significant challenge from his Democratic rival, former Vice President Joe Biden.
In a Tuesday night debate with Biden, Trump again raised the idea of a quick approval. “Now we’re weeks away from a vaccine,” Trump said during the debate.
Trump’s estimates, though, are not in line with those offered by most firms involved with making vaccines. The most optimistic projections have come from Pfizer Inc. The drugmaker’s chief executive, Albert Bourla, has spoken about his company possibly having data to present to the FDA as early as late October about the safety and effectiveness of a vaccine.
In a September 8 interview with the Today show, Bourla said there was a 60% chance his company would meet that goal. In response to a question, he made it clear his comments applied to a potential Pfizer application, not an approval or release of a vaccine by that time.
In response to concerns about political pressures, the FDA in June issued guidance outlining what its staff would require for approval of a COVID-19 vaccine.
Pushback on politics
Another witness at the Wednesday hearing, Mark McClellan, MD, PhD, a former FDA commissioner (2002 – 2004), pushed back on objections to a potential release of further guidance from the agency.
“Some recent statements from the White House have implied that FDA’s plan to release additional written guidance on its expectations for emergency use authorization of a vaccine is unnecessarily raising the bar on regulatory standards for authorization,” said McClellan in his testimony for the House panel. “That is not the case.”
Instead, further FDA guidance would be a welcome form of feedback for the firms trying to develop COVID-19 vaccines, according to McClellan, who also serves on the board of directors for Johnson & Johnson. Johnson & Johnson is among the firms that have advanced a COVID-19 vaccine candidate to phase 3 testing. In his role as a director, he serves on the board’s regulatory compliance committee.
Along with politics, recent stumbles at FDA with emergency use authorizations (EUAs) of treatments for COVID-19 have eroded the public’s confidence in the agency, Jha told the House panel. The FDA approved hydroxychloroquine, a medicine promoted by Trump for use in COVID, under an EUA in March and then revoked this clearance in June.
Jha said the FDA’s most serious misstep was its handling of convalescent plasma, which was approved through an EUA on August 23 “in a highly advertised and widely televised announcement including the president.
“The announcement solidified in the public conversation the impression that, increasingly with this administration, politics are taking over trusted, nonpartisan scientific institutions,” he said in his testimony.
Approving a COVID-19 vaccine on the limited evidence through an EUA would mark a serious departure from FDA policy, according to Jha.
“While we sometimes accept a certain level of potential harm in experimental treatments for those who are severely ill, vaccines are given to healthy people and therefore need to have a substantially higher measure of safety and effectiveness,” he explained.
Jha said the FDA has only once before used this EUA approach for a vaccine. That was for a vaccine against inhaled anthrax and was mostly distributed to high-risk soldiers and civilians in war zones.
COVID-19, in contrast, is an infection that has changed lives around the world. The virus has contributed to more than 1 million deaths, including more than 200,000 in the United States, according to the World Health Organization.
Scientists are hoping vaccines will help curb this infection, although much of the future success of vaccines depends on how widely they are used, witnesses told the House panel.
Debate on approaches for vaccine effectiveness
In his testimony, Jha also noted concerns about COVID-19 vaccine trials. He included a reference to a Sept. 22 opinion article titled, “These Coronavirus Trials Don›t Answer the One Question We Need to Know,” which was written by Peter Doshi, PhD, of the University of Maryland School of Pharmacy, in Baltimore, and Eric Topol, MD, a professor of molecular medicine at Scripps Research in La Jolla, Calif. Topol is also editor in chief of Medscape.
Topol and Doshi questioned why the firms Moderna, Pfizer, and AstraZeneca structured their competing trials such that “a vaccine could meet the companies’ benchmark for success if it lowered the risk of mild Covid-19, but was never shown to reduce moderate or severe forms of the disease, or the risk of hospitalization, admissions to the intensive care unit or death.”
“To say a vaccine works should mean that most people no longer run the risk of getting seriously sick,” Topol and Doshi wrote. “That’s not what these trials will determine.”
There was disagreement about this point at the hearing. U.S. Representative Morgan Griffith (R-Va.) read the section of the Doshi-Topol article quoted above and asked one witness, Offit, to weigh in.
“Do you agree with those concerns? And either way, tell me why,” Griffith asked.
“I don’t agree,” Offit responded.
“I think it’s actually much harder to prevent asymptomatic infection or mildly symptomatic infection,” he said. “If you can prevent that, you are much more likely to prevent moderate to severe disease. So I think they have it backwards.”
But other researchers also question the approaches used with the current crop of COVID-19 vaccines.
“With the current protocols, it is conceivable that a vaccine might be considered effective – and eventually approved – based primarily on its ability to prevent mild cases alone,” wrote William Haseltine, PhD, president of the nonprofit ACCESS Health International, in a September 22 opinion article in the Washington Post titled: “Beware of COVID-19 Vaccine Trials Designed to Succeed From the Start.”
In an interview with Medscape Medical News on Wednesday, Haseltine said he maintains these concerns about the tests. Earlier in his career, he was a leader in HIV research through his lab at Harvard University in Cambridge, Massachusetts, and he subsequently led a biotech company, Human Genome Sciences.
He fears consumers will not get what they might expect from the vaccines being tested.
“What people care about is if this is going to keep them out of the hospital and will it keep them alive. And that’s not even part of this protocol,” Haseltine said.
This article first appeared on Medscape.com.
on Wednesday.
The career staff of the Food and Drug Administration can be counted on to appropriately weigh whether a vaccine should be cleared for use in preventing COVID-19, witnesses, including Paul A. Offit, MD, of Children’s Hospital of Philadelphia, told the House Energy and Commerce Committee’s oversight and investigations panel.
FDA staffers would object to attempts by the Trump administration to rush a vaccine to the public without proper vetting, as would veteran federal researchers, including National Institutes of Health Director Francis S. Collins, MD, PhD, and Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, Offit said.
“If COVID-19 vaccines are released before they’re ready to be released, you will hear from these people, and you will also hear from people like Dr. Francis Collins and Tony Fauci, both of whom are trusted by the American public, as well as many other academicians and researchers who wouldn’t stand for this,” he said.
“The public is already nervous about these vaccines,” said Offit, who serves on key FDA and Centers for Disease Control and Prevention committees overseeing vaccine policy. “If trusted health officials stand up and decry a premature release, the celebration by the administration will be short-lived.”
Overly optimistic estimates about a potential approval can only serve to erode the public’s trust in these crucial vaccines, said another witness, Ashish K. Jha, MD, MPH, the dean of Brown University’s School of Public Health, in Providence, Rhode Island.
“All political leaders need to stop talking about things like time lines,” Jha told the lawmakers.
President Donald Trump has several times suggested that a COVID vaccine might be approved ahead of the November 3 election, where he faces a significant challenge from his Democratic rival, former Vice President Joe Biden.
In a Tuesday night debate with Biden, Trump again raised the idea of a quick approval. “Now we’re weeks away from a vaccine,” Trump said during the debate.
Trump’s estimates, though, are not in line with those offered by most firms involved with making vaccines. The most optimistic projections have come from Pfizer Inc. The drugmaker’s chief executive, Albert Bourla, has spoken about his company possibly having data to present to the FDA as early as late October about the safety and effectiveness of a vaccine.
In a September 8 interview with the Today show, Bourla said there was a 60% chance his company would meet that goal. In response to a question, he made it clear his comments applied to a potential Pfizer application, not an approval or release of a vaccine by that time.
In response to concerns about political pressures, the FDA in June issued guidance outlining what its staff would require for approval of a COVID-19 vaccine.
Pushback on politics
Another witness at the Wednesday hearing, Mark McClellan, MD, PhD, a former FDA commissioner (2002 – 2004), pushed back on objections to a potential release of further guidance from the agency.
“Some recent statements from the White House have implied that FDA’s plan to release additional written guidance on its expectations for emergency use authorization of a vaccine is unnecessarily raising the bar on regulatory standards for authorization,” said McClellan in his testimony for the House panel. “That is not the case.”
Instead, further FDA guidance would be a welcome form of feedback for the firms trying to develop COVID-19 vaccines, according to McClellan, who also serves on the board of directors for Johnson & Johnson. Johnson & Johnson is among the firms that have advanced a COVID-19 vaccine candidate to phase 3 testing. In his role as a director, he serves on the board’s regulatory compliance committee.
Along with politics, recent stumbles at FDA with emergency use authorizations (EUAs) of treatments for COVID-19 have eroded the public’s confidence in the agency, Jha told the House panel. The FDA approved hydroxychloroquine, a medicine promoted by Trump for use in COVID, under an EUA in March and then revoked this clearance in June.
Jha said the FDA’s most serious misstep was its handling of convalescent plasma, which was approved through an EUA on August 23 “in a highly advertised and widely televised announcement including the president.
“The announcement solidified in the public conversation the impression that, increasingly with this administration, politics are taking over trusted, nonpartisan scientific institutions,” he said in his testimony.
Approving a COVID-19 vaccine on the limited evidence through an EUA would mark a serious departure from FDA policy, according to Jha.
“While we sometimes accept a certain level of potential harm in experimental treatments for those who are severely ill, vaccines are given to healthy people and therefore need to have a substantially higher measure of safety and effectiveness,” he explained.
Jha said the FDA has only once before used this EUA approach for a vaccine. That was for a vaccine against inhaled anthrax and was mostly distributed to high-risk soldiers and civilians in war zones.
COVID-19, in contrast, is an infection that has changed lives around the world. The virus has contributed to more than 1 million deaths, including more than 200,000 in the United States, according to the World Health Organization.
Scientists are hoping vaccines will help curb this infection, although much of the future success of vaccines depends on how widely they are used, witnesses told the House panel.
Debate on approaches for vaccine effectiveness
In his testimony, Jha also noted concerns about COVID-19 vaccine trials. He included a reference to a Sept. 22 opinion article titled, “These Coronavirus Trials Don›t Answer the One Question We Need to Know,” which was written by Peter Doshi, PhD, of the University of Maryland School of Pharmacy, in Baltimore, and Eric Topol, MD, a professor of molecular medicine at Scripps Research in La Jolla, Calif. Topol is also editor in chief of Medscape.
Topol and Doshi questioned why the firms Moderna, Pfizer, and AstraZeneca structured their competing trials such that “a vaccine could meet the companies’ benchmark for success if it lowered the risk of mild Covid-19, but was never shown to reduce moderate or severe forms of the disease, or the risk of hospitalization, admissions to the intensive care unit or death.”
“To say a vaccine works should mean that most people no longer run the risk of getting seriously sick,” Topol and Doshi wrote. “That’s not what these trials will determine.”
There was disagreement about this point at the hearing. U.S. Representative Morgan Griffith (R-Va.) read the section of the Doshi-Topol article quoted above and asked one witness, Offit, to weigh in.
“Do you agree with those concerns? And either way, tell me why,” Griffith asked.
“I don’t agree,” Offit responded.
“I think it’s actually much harder to prevent asymptomatic infection or mildly symptomatic infection,” he said. “If you can prevent that, you are much more likely to prevent moderate to severe disease. So I think they have it backwards.”
But other researchers also question the approaches used with the current crop of COVID-19 vaccines.
“With the current protocols, it is conceivable that a vaccine might be considered effective – and eventually approved – based primarily on its ability to prevent mild cases alone,” wrote William Haseltine, PhD, president of the nonprofit ACCESS Health International, in a September 22 opinion article in the Washington Post titled: “Beware of COVID-19 Vaccine Trials Designed to Succeed From the Start.”
In an interview with Medscape Medical News on Wednesday, Haseltine said he maintains these concerns about the tests. Earlier in his career, he was a leader in HIV research through his lab at Harvard University in Cambridge, Massachusetts, and he subsequently led a biotech company, Human Genome Sciences.
He fears consumers will not get what they might expect from the vaccines being tested.
“What people care about is if this is going to keep them out of the hospital and will it keep them alive. And that’s not even part of this protocol,” Haseltine said.
This article first appeared on Medscape.com.
Post-COVID clinics get jump-start from patients with lingering illness
Clarence Troutman survived a 2-month hospital stay with COVID-19, then went home in early June. But he’s far from over the disease, still suffering from limited endurance, shortness of breath and hands that can be stiff and swollen.
“Before COVID, I was a 59-year-old, relatively healthy man,” said the broadband technician from Denver. “If I had to say where I’m at now, I’d say about 50% of where I was, but when I first went home, I was at 20%.”
He credits much of his progress to the “motivation and education” gleaned from a new program for post-COVID patients at the University of Colorado at Denver, Aurora, one of a small but growing number of clinics aimed at treating and studying those who have had the unpredictable coronavirus.
As the election nears, much attention is focused on daily infection numbers or the climbing death toll, but another measure matters: Patients who survive but continue to wrestle with a range of physical or mental effects, including lung damage, heart or neurologic concerns, anxiety, and depression.
“We need to think about how we’re going to provide care for patients who may be recovering for years after the virus,” said Sarah Jolley, MD, a pulmonologist with UCHealth University of Colorado Hospital and director of UCHealth’s Post-Covid Clinic, where Mr. Troutman is seen.
That need has jump-started post-COVID clinics, which bring together a range of specialists into a one-stop shop.
One of the first and largest such clinics is at Mount Sinai in New York City, but programs have also launched at the University of California,San Francisco; Stanford (Calif.) University Medical Center; and the University of Pennsylvania, Philadelphia. The Cleveland Clinic plans to open one early next year. And it’s not just academic medical centers: St. John’s Well Child and Family Center, part of a network of community clinics in south central Los Angeles, said this month it aims to test thousands of its patients who were diagnosed with COVID-19 since March for long-term effects.
Mental health specialists are also involved, along with social workers and pharmacists. Many of the centers also do research studies, aiming to better understand why the virus hits certain patients so hard.
“Some of our patients, even those on a ventilator on death’s door, will come out remarkably unscathed,” said Lekshmi Santhosh, MD, an assistant professor of pulmonary critical care and a leader of the post-COVID program at UCSF, called the OPTIMAL clinic. “Others, even those who were never hospitalized, have disabling fatigue, ongoing chest pain, and shortness of breath, and there’s a whole spectrum in between.”
‘Staggering’ medical need
It’s too early to know how long the persistent medical effects and symptoms will linger, or to make accurate estimates on the percentage of patients affected.
Some early studies are sobering. An Austrian report released this month found that 76 of the first 86 patients studied had evidence of lung damage 6 weeks after hospital discharge, but that dropped to 48 patients at 12 weeks.
Some researchers and clinics say about 10% of U.S. COVID patients they see may have longer-running effects, said Zijian Chen, MD, medical director of the Center for Post-COVID Care at Mount Sinai, which has enrolled 400 patients so far.
If that estimate is correct – and Dr. Chen emphasized that more research is needed to make sure – it translates to patients entering the medical system in droves, often with multiple issues.
How health systems and insurers respond will be key, he said. More than 6.5 million U.S. residents have tested positive for the disease. If fewer than 10% – say 500,000 – already have long-lasting symptoms, “that number is staggering,” Dr. Chen said. “How much medical care will be needed for that?”
Though start-up costs could be a hurdle, the clinics themselves may eventually draw much-needed revenue to medical centers by attracting patients, many of whom have insurance to cover some or all of the cost of repeated visits.
Dr. Chen said the specialized centers can help lower health spending by providing more cost effective, coordinated care that avoids duplicative testing a patient might otherwise undergo.
“We’ve seen patients that when they come in, they’ve already had four MRI or CT scans and a stack of bloodwork,” he said.
The program consolidates those earlier results and determines if any additional testing is needed. Sometimes the answer to what’s causing patients’ long-lasting symptoms remains elusive. One problem for patients seeking help outside of dedicated clinics is that when there is no clear cause for their condition, they may be told the symptoms are imagined.
“I believe in the patients,” said Dr. Chen.
About half the clinic’s patients have received test results showing damage, said Dr. Chen, an endocrinologist and internal medicine physician. For those patients, the clinic can develop a treatment plan. But, frustratingly, the other half have inconclusive test results yet exhibit a range of symptoms.
“That makes it more difficult to treat,” said Dr. Chen.
Experts see parallels to a push in the past decade to establish special clinics to treat patients released from ICU wards, who may have problems related to long-term bed rest or the delirium many experience while hospitalized. Some of the current post-COVID clinics are modeled after the post-ICU clinics or are expanded versions of them.
The ICU Recovery Center at Vanderbilt University Medical Center, Nashville, Tenn., for instance, which opened in 2012, is accepting post-COVID patients.
There are about a dozen post-ICU clinics nationally, some of which are also now working with COVID patients, said James Jackson, director of long-term outcomes at the Vanderbilt center. In addition, he’s heard of at least another dozen post-COVID centers in development.
The centers generally do an initial assessment a few weeks after a patient is diagnosed or discharged from the hospital, often by video call. Check-in and repeat visits are scheduled every month or so after that.
“In an ideal world, with these post-COVID clinics, you can identify the patients and get them into rehab,” he said. “Even if the primary thing these clinics did was to say to patients: ‘This is real, it is not all in your head,’ that impact would be important.”
A question of feasibility
Financing is the largest obstacle, program proponents said. Many hospitals lost substantial revenue to canceled elective procedures during stay-at-home periods.
“So, it’s not a great time to be pitching a new activity that requires a start-up subsidy,” said Glenn Melnick, PhD, a professor of health economics at the University of Southern California.
At UCSF, a select group of faculty members staff the post-COVID clinics and some mental health professionals volunteer their time, said Dr. Santhosh.
Dr. Chen said he was able to recruit team members and support staff from the ranks of those whose elective patient caseload had dropped.
Dr. Jackson said unfortunately there’s not been enough research into the cost-and-clinical effectiveness of post-ICU centers.
“In the early days, there may have been questions about how much value does this add,” he noted. “Now, the question is not so much is it a good idea, but is it feasible?”
Right now, the post-COVID centers are foremost a research effort, said Len Nichols, an economist and nonresident fellow at the Urban Institute. “If these guys get good at treating long-term symptoms, that’s good for all of us. There’s not enough patients to make it a business model yet, but if they become the place to go when you get it, it could become a business model for some of the elite institutions.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Clarence Troutman survived a 2-month hospital stay with COVID-19, then went home in early June. But he’s far from over the disease, still suffering from limited endurance, shortness of breath and hands that can be stiff and swollen.
“Before COVID, I was a 59-year-old, relatively healthy man,” said the broadband technician from Denver. “If I had to say where I’m at now, I’d say about 50% of where I was, but when I first went home, I was at 20%.”
He credits much of his progress to the “motivation and education” gleaned from a new program for post-COVID patients at the University of Colorado at Denver, Aurora, one of a small but growing number of clinics aimed at treating and studying those who have had the unpredictable coronavirus.
As the election nears, much attention is focused on daily infection numbers or the climbing death toll, but another measure matters: Patients who survive but continue to wrestle with a range of physical or mental effects, including lung damage, heart or neurologic concerns, anxiety, and depression.
“We need to think about how we’re going to provide care for patients who may be recovering for years after the virus,” said Sarah Jolley, MD, a pulmonologist with UCHealth University of Colorado Hospital and director of UCHealth’s Post-Covid Clinic, where Mr. Troutman is seen.
That need has jump-started post-COVID clinics, which bring together a range of specialists into a one-stop shop.
One of the first and largest such clinics is at Mount Sinai in New York City, but programs have also launched at the University of California,San Francisco; Stanford (Calif.) University Medical Center; and the University of Pennsylvania, Philadelphia. The Cleveland Clinic plans to open one early next year. And it’s not just academic medical centers: St. John’s Well Child and Family Center, part of a network of community clinics in south central Los Angeles, said this month it aims to test thousands of its patients who were diagnosed with COVID-19 since March for long-term effects.
Mental health specialists are also involved, along with social workers and pharmacists. Many of the centers also do research studies, aiming to better understand why the virus hits certain patients so hard.
“Some of our patients, even those on a ventilator on death’s door, will come out remarkably unscathed,” said Lekshmi Santhosh, MD, an assistant professor of pulmonary critical care and a leader of the post-COVID program at UCSF, called the OPTIMAL clinic. “Others, even those who were never hospitalized, have disabling fatigue, ongoing chest pain, and shortness of breath, and there’s a whole spectrum in between.”
‘Staggering’ medical need
It’s too early to know how long the persistent medical effects and symptoms will linger, or to make accurate estimates on the percentage of patients affected.
Some early studies are sobering. An Austrian report released this month found that 76 of the first 86 patients studied had evidence of lung damage 6 weeks after hospital discharge, but that dropped to 48 patients at 12 weeks.
Some researchers and clinics say about 10% of U.S. COVID patients they see may have longer-running effects, said Zijian Chen, MD, medical director of the Center for Post-COVID Care at Mount Sinai, which has enrolled 400 patients so far.
If that estimate is correct – and Dr. Chen emphasized that more research is needed to make sure – it translates to patients entering the medical system in droves, often with multiple issues.
How health systems and insurers respond will be key, he said. More than 6.5 million U.S. residents have tested positive for the disease. If fewer than 10% – say 500,000 – already have long-lasting symptoms, “that number is staggering,” Dr. Chen said. “How much medical care will be needed for that?”
Though start-up costs could be a hurdle, the clinics themselves may eventually draw much-needed revenue to medical centers by attracting patients, many of whom have insurance to cover some or all of the cost of repeated visits.
Dr. Chen said the specialized centers can help lower health spending by providing more cost effective, coordinated care that avoids duplicative testing a patient might otherwise undergo.
“We’ve seen patients that when they come in, they’ve already had four MRI or CT scans and a stack of bloodwork,” he said.
The program consolidates those earlier results and determines if any additional testing is needed. Sometimes the answer to what’s causing patients’ long-lasting symptoms remains elusive. One problem for patients seeking help outside of dedicated clinics is that when there is no clear cause for their condition, they may be told the symptoms are imagined.
“I believe in the patients,” said Dr. Chen.
About half the clinic’s patients have received test results showing damage, said Dr. Chen, an endocrinologist and internal medicine physician. For those patients, the clinic can develop a treatment plan. But, frustratingly, the other half have inconclusive test results yet exhibit a range of symptoms.
“That makes it more difficult to treat,” said Dr. Chen.
Experts see parallels to a push in the past decade to establish special clinics to treat patients released from ICU wards, who may have problems related to long-term bed rest or the delirium many experience while hospitalized. Some of the current post-COVID clinics are modeled after the post-ICU clinics or are expanded versions of them.
The ICU Recovery Center at Vanderbilt University Medical Center, Nashville, Tenn., for instance, which opened in 2012, is accepting post-COVID patients.
There are about a dozen post-ICU clinics nationally, some of which are also now working with COVID patients, said James Jackson, director of long-term outcomes at the Vanderbilt center. In addition, he’s heard of at least another dozen post-COVID centers in development.
The centers generally do an initial assessment a few weeks after a patient is diagnosed or discharged from the hospital, often by video call. Check-in and repeat visits are scheduled every month or so after that.
“In an ideal world, with these post-COVID clinics, you can identify the patients and get them into rehab,” he said. “Even if the primary thing these clinics did was to say to patients: ‘This is real, it is not all in your head,’ that impact would be important.”
A question of feasibility
Financing is the largest obstacle, program proponents said. Many hospitals lost substantial revenue to canceled elective procedures during stay-at-home periods.
“So, it’s not a great time to be pitching a new activity that requires a start-up subsidy,” said Glenn Melnick, PhD, a professor of health economics at the University of Southern California.
At UCSF, a select group of faculty members staff the post-COVID clinics and some mental health professionals volunteer their time, said Dr. Santhosh.
Dr. Chen said he was able to recruit team members and support staff from the ranks of those whose elective patient caseload had dropped.
Dr. Jackson said unfortunately there’s not been enough research into the cost-and-clinical effectiveness of post-ICU centers.
“In the early days, there may have been questions about how much value does this add,” he noted. “Now, the question is not so much is it a good idea, but is it feasible?”
Right now, the post-COVID centers are foremost a research effort, said Len Nichols, an economist and nonresident fellow at the Urban Institute. “If these guys get good at treating long-term symptoms, that’s good for all of us. There’s not enough patients to make it a business model yet, but if they become the place to go when you get it, it could become a business model for some of the elite institutions.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Clarence Troutman survived a 2-month hospital stay with COVID-19, then went home in early June. But he’s far from over the disease, still suffering from limited endurance, shortness of breath and hands that can be stiff and swollen.
“Before COVID, I was a 59-year-old, relatively healthy man,” said the broadband technician from Denver. “If I had to say where I’m at now, I’d say about 50% of where I was, but when I first went home, I was at 20%.”
He credits much of his progress to the “motivation and education” gleaned from a new program for post-COVID patients at the University of Colorado at Denver, Aurora, one of a small but growing number of clinics aimed at treating and studying those who have had the unpredictable coronavirus.
As the election nears, much attention is focused on daily infection numbers or the climbing death toll, but another measure matters: Patients who survive but continue to wrestle with a range of physical or mental effects, including lung damage, heart or neurologic concerns, anxiety, and depression.
“We need to think about how we’re going to provide care for patients who may be recovering for years after the virus,” said Sarah Jolley, MD, a pulmonologist with UCHealth University of Colorado Hospital and director of UCHealth’s Post-Covid Clinic, where Mr. Troutman is seen.
That need has jump-started post-COVID clinics, which bring together a range of specialists into a one-stop shop.
One of the first and largest such clinics is at Mount Sinai in New York City, but programs have also launched at the University of California,San Francisco; Stanford (Calif.) University Medical Center; and the University of Pennsylvania, Philadelphia. The Cleveland Clinic plans to open one early next year. And it’s not just academic medical centers: St. John’s Well Child and Family Center, part of a network of community clinics in south central Los Angeles, said this month it aims to test thousands of its patients who were diagnosed with COVID-19 since March for long-term effects.
Mental health specialists are also involved, along with social workers and pharmacists. Many of the centers also do research studies, aiming to better understand why the virus hits certain patients so hard.
“Some of our patients, even those on a ventilator on death’s door, will come out remarkably unscathed,” said Lekshmi Santhosh, MD, an assistant professor of pulmonary critical care and a leader of the post-COVID program at UCSF, called the OPTIMAL clinic. “Others, even those who were never hospitalized, have disabling fatigue, ongoing chest pain, and shortness of breath, and there’s a whole spectrum in between.”
‘Staggering’ medical need
It’s too early to know how long the persistent medical effects and symptoms will linger, or to make accurate estimates on the percentage of patients affected.
Some early studies are sobering. An Austrian report released this month found that 76 of the first 86 patients studied had evidence of lung damage 6 weeks after hospital discharge, but that dropped to 48 patients at 12 weeks.
Some researchers and clinics say about 10% of U.S. COVID patients they see may have longer-running effects, said Zijian Chen, MD, medical director of the Center for Post-COVID Care at Mount Sinai, which has enrolled 400 patients so far.
If that estimate is correct – and Dr. Chen emphasized that more research is needed to make sure – it translates to patients entering the medical system in droves, often with multiple issues.
How health systems and insurers respond will be key, he said. More than 6.5 million U.S. residents have tested positive for the disease. If fewer than 10% – say 500,000 – already have long-lasting symptoms, “that number is staggering,” Dr. Chen said. “How much medical care will be needed for that?”
Though start-up costs could be a hurdle, the clinics themselves may eventually draw much-needed revenue to medical centers by attracting patients, many of whom have insurance to cover some or all of the cost of repeated visits.
Dr. Chen said the specialized centers can help lower health spending by providing more cost effective, coordinated care that avoids duplicative testing a patient might otherwise undergo.
“We’ve seen patients that when they come in, they’ve already had four MRI or CT scans and a stack of bloodwork,” he said.
The program consolidates those earlier results and determines if any additional testing is needed. Sometimes the answer to what’s causing patients’ long-lasting symptoms remains elusive. One problem for patients seeking help outside of dedicated clinics is that when there is no clear cause for their condition, they may be told the symptoms are imagined.
“I believe in the patients,” said Dr. Chen.
About half the clinic’s patients have received test results showing damage, said Dr. Chen, an endocrinologist and internal medicine physician. For those patients, the clinic can develop a treatment plan. But, frustratingly, the other half have inconclusive test results yet exhibit a range of symptoms.
“That makes it more difficult to treat,” said Dr. Chen.
Experts see parallels to a push in the past decade to establish special clinics to treat patients released from ICU wards, who may have problems related to long-term bed rest or the delirium many experience while hospitalized. Some of the current post-COVID clinics are modeled after the post-ICU clinics or are expanded versions of them.
The ICU Recovery Center at Vanderbilt University Medical Center, Nashville, Tenn., for instance, which opened in 2012, is accepting post-COVID patients.
There are about a dozen post-ICU clinics nationally, some of which are also now working with COVID patients, said James Jackson, director of long-term outcomes at the Vanderbilt center. In addition, he’s heard of at least another dozen post-COVID centers in development.
The centers generally do an initial assessment a few weeks after a patient is diagnosed or discharged from the hospital, often by video call. Check-in and repeat visits are scheduled every month or so after that.
“In an ideal world, with these post-COVID clinics, you can identify the patients and get them into rehab,” he said. “Even if the primary thing these clinics did was to say to patients: ‘This is real, it is not all in your head,’ that impact would be important.”
A question of feasibility
Financing is the largest obstacle, program proponents said. Many hospitals lost substantial revenue to canceled elective procedures during stay-at-home periods.
“So, it’s not a great time to be pitching a new activity that requires a start-up subsidy,” said Glenn Melnick, PhD, a professor of health economics at the University of Southern California.
At UCSF, a select group of faculty members staff the post-COVID clinics and some mental health professionals volunteer their time, said Dr. Santhosh.
Dr. Chen said he was able to recruit team members and support staff from the ranks of those whose elective patient caseload had dropped.
Dr. Jackson said unfortunately there’s not been enough research into the cost-and-clinical effectiveness of post-ICU centers.
“In the early days, there may have been questions about how much value does this add,” he noted. “Now, the question is not so much is it a good idea, but is it feasible?”
Right now, the post-COVID centers are foremost a research effort, said Len Nichols, an economist and nonresident fellow at the Urban Institute. “If these guys get good at treating long-term symptoms, that’s good for all of us. There’s not enough patients to make it a business model yet, but if they become the place to go when you get it, it could become a business model for some of the elite institutions.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
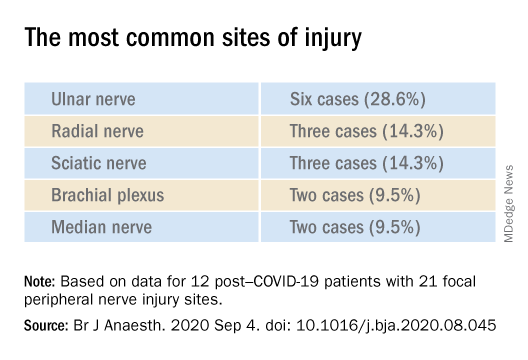
Nerve damage linked to prone positioning in COVID-19
A new case series describes peripheral nerve injuries associated with this type of positioning and suggests ways to minimize the potential damage.
“Physicians should remain aware of increased susceptibility to peripheral nerve damage in patients with severe COVID-19 after prone positioning, since it is surprisingly common among these patients, and should refine standard protocols accordingly to reduce that risk,” said senior author Colin Franz, MD, PhD, director of the Electrodiagnostic Laboratory, Shirley Ryan AbilityLab, Chicago.
The article was published online Sept. 4 in the British Journal of Anaesthesiology.
Unique type of nerve injury
Many patients who are admitted to the intensive care unit with COVID-19 undergo invasive mechanical ventilation because of acute respiratory distress syndrome (ARDS). Clinical guidelines recommend that such patients lie in the prone position 12-16 hours per day.
“Prone positioning for up to 16 hours is a therapy we use for patients with more severe forms of ARDS, and high-level evidence points to mortality benefit in patients with moderate to severe ARDS if [mechanical] ventilation occurs,” said study coauthor James McCauley Walter, MD, of the pulmonary division at Northwestern University, Chicago.
With a “significant number of COVID-19 patients flooding the ICU, we quickly started to prone a lot of them, but if you are in a specific position for multiple hours a day, coupled with the neurotoxic effects of the SARS-CoV-2 virus itself, you may be exposed to a unique type of nerve injury,” he said.
Dr. Walter said that the “incidence of asymmetric neuropathies seems out of proportion to what has been reported in non–COVID-19 settings, which is what caught our attention.”
Many of these patients are discharged to rehabilitation hospitals, and “what we noticed, which was unique about COVID-19 patients coming to our rehab hospital, was that, compared with other patients who had been critically ill with a long hospital stay, there was a significantly higher percentage of COVID-19 patients who had peripheral nerve damage,” Dr. Franz said.
The authors described 12 of these patients who were admitted between April 24 and June 30, 2020 (mean age, 60.3 years; range, 23-80 years). The sample included White, Black, and Hispanic individuals. Eleven of the 12 post–COVID-19 patients with peripheral nerve damage had experienced prone positioning during acute management.
The average number of days patients received mechanical ventilation was 33.6 (range, 12-62 days). The average number of proning sessions was 4.5 (range, 1-16) with an average of 81.2 hours (range, 16-252 hours) spent prone.
A major contributor
Dr. Franz suggested that prone positioning is likely not the only cause of peripheral nerve damage but “may play a big role in these patients who are vulnerable because of viral infection and the critical illness that causes damage and nerve injuries.”
“The first component of lifesaving care for the critically ill in the ICU is intravenous fluids, mechanical ventilation, steroids, and antibiotics for infection,” said Dr. Walter.
“We are trying to come up with ways to place patients in prone position in safer ways, to pay attention to pressure points and areas of injury that we have seen and try to offload them, to see if we can decrease the rate of these injuries,” he added.
The researchers’ article includes a heat map diagram as a “template for where to focus the most efforts, in terms of decreasing pressure,” Dr. Walter said.
“The nerves are accepting too much force for gravely ill COVID-19 patients to handle, so we suggest using the template to determine where extra padding might be needed, or a protocol that might include changes in positioning,” he added.
Dr. Franz described the interventions used for COVID-19 patients with prone positioning–related peripheral nerve damage. “The first step is trying to address the problems one by one, either trying to solve them through exercise or teaching new skills, new ways to compensate, beginning with basic activities, such as getting out of bed and self-care,” he said.
Long-term recovery of nerve injuries depends on how severe the injuries are. Some nerves can slowly regenerate – possibly at the rate of 1 inch per month – which can be a long process, taking between a year and 18 months.
Dr. Franz said that therapies for this condition are “extrapolated from clinical trial work” on promoting nerve regeneration after surgery using electrical stimulation to enable nerves to regrow at a faster rate.
“Regeneration is not only slow, but it may not happen completely, leaving the patient with permanent nerve damage – in fact, based on our experience and what has been reported, the percentage of patients with full recovery is only 10%,” he said.
The most common symptomatic complaint other than lack of movement or feeling is neuropathic pain, “which may require medication to take the edge off the pain,” Dr. Franz added.
Irreversible damage?
Commenting on the study, Tae Chung, MD, of the departments of physical medicine, rehabilitation, and neurology, Johns Hopkins University, Baltimore, said the study “provides one of the first and the largest description of peripheral nerve injury associated with prone positioning for management of ARDS from COVID-19.”
Dr. Chung, who was not involved in the research, noted that “various neurological complications from COVID-19 have been reported, and some of them may result in irreversible neurological damage or delay the recovery from COVID-19 infection,” so “accurate and timely diagnosis of such neurological complications is critical for rehabilitation of the COVID-19 survivors.”
The study received no funding. Dr. Franz, Dr. Walter, study coauthors, and Dr. Chung report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A new case series describes peripheral nerve injuries associated with this type of positioning and suggests ways to minimize the potential damage.

“Physicians should remain aware of increased susceptibility to peripheral nerve damage in patients with severe COVID-19 after prone positioning, since it is surprisingly common among these patients, and should refine standard protocols accordingly to reduce that risk,” said senior author Colin Franz, MD, PhD, director of the Electrodiagnostic Laboratory, Shirley Ryan AbilityLab, Chicago.
The article was published online Sept. 4 in the British Journal of Anaesthesiology.
Unique type of nerve injury
Many patients who are admitted to the intensive care unit with COVID-19 undergo invasive mechanical ventilation because of acute respiratory distress syndrome (ARDS). Clinical guidelines recommend that such patients lie in the prone position 12-16 hours per day.
“Prone positioning for up to 16 hours is a therapy we use for patients with more severe forms of ARDS, and high-level evidence points to mortality benefit in patients with moderate to severe ARDS if [mechanical] ventilation occurs,” said study coauthor James McCauley Walter, MD, of the pulmonary division at Northwestern University, Chicago.
With a “significant number of COVID-19 patients flooding the ICU, we quickly started to prone a lot of them, but if you are in a specific position for multiple hours a day, coupled with the neurotoxic effects of the SARS-CoV-2 virus itself, you may be exposed to a unique type of nerve injury,” he said.
Dr. Walter said that the “incidence of asymmetric neuropathies seems out of proportion to what has been reported in non–COVID-19 settings, which is what caught our attention.”
Many of these patients are discharged to rehabilitation hospitals, and “what we noticed, which was unique about COVID-19 patients coming to our rehab hospital, was that, compared with other patients who had been critically ill with a long hospital stay, there was a significantly higher percentage of COVID-19 patients who had peripheral nerve damage,” Dr. Franz said.
The authors described 12 of these patients who were admitted between April 24 and June 30, 2020 (mean age, 60.3 years; range, 23-80 years). The sample included White, Black, and Hispanic individuals. Eleven of the 12 post–COVID-19 patients with peripheral nerve damage had experienced prone positioning during acute management.
The average number of days patients received mechanical ventilation was 33.6 (range, 12-62 days). The average number of proning sessions was 4.5 (range, 1-16) with an average of 81.2 hours (range, 16-252 hours) spent prone.
A major contributor
Dr. Franz suggested that prone positioning is likely not the only cause of peripheral nerve damage but “may play a big role in these patients who are vulnerable because of viral infection and the critical illness that causes damage and nerve injuries.”
“The first component of lifesaving care for the critically ill in the ICU is intravenous fluids, mechanical ventilation, steroids, and antibiotics for infection,” said Dr. Walter.
“We are trying to come up with ways to place patients in prone position in safer ways, to pay attention to pressure points and areas of injury that we have seen and try to offload them, to see if we can decrease the rate of these injuries,” he added.
The researchers’ article includes a heat map diagram as a “template for where to focus the most efforts, in terms of decreasing pressure,” Dr. Walter said.
“The nerves are accepting too much force for gravely ill COVID-19 patients to handle, so we suggest using the template to determine where extra padding might be needed, or a protocol that might include changes in positioning,” he added.
Dr. Franz described the interventions used for COVID-19 patients with prone positioning–related peripheral nerve damage. “The first step is trying to address the problems one by one, either trying to solve them through exercise or teaching new skills, new ways to compensate, beginning with basic activities, such as getting out of bed and self-care,” he said.
Long-term recovery of nerve injuries depends on how severe the injuries are. Some nerves can slowly regenerate – possibly at the rate of 1 inch per month – which can be a long process, taking between a year and 18 months.
Dr. Franz said that therapies for this condition are “extrapolated from clinical trial work” on promoting nerve regeneration after surgery using electrical stimulation to enable nerves to regrow at a faster rate.
“Regeneration is not only slow, but it may not happen completely, leaving the patient with permanent nerve damage – in fact, based on our experience and what has been reported, the percentage of patients with full recovery is only 10%,” he said.
The most common symptomatic complaint other than lack of movement or feeling is neuropathic pain, “which may require medication to take the edge off the pain,” Dr. Franz added.
Irreversible damage?
Commenting on the study, Tae Chung, MD, of the departments of physical medicine, rehabilitation, and neurology, Johns Hopkins University, Baltimore, said the study “provides one of the first and the largest description of peripheral nerve injury associated with prone positioning for management of ARDS from COVID-19.”
Dr. Chung, who was not involved in the research, noted that “various neurological complications from COVID-19 have been reported, and some of them may result in irreversible neurological damage or delay the recovery from COVID-19 infection,” so “accurate and timely diagnosis of such neurological complications is critical for rehabilitation of the COVID-19 survivors.”
The study received no funding. Dr. Franz, Dr. Walter, study coauthors, and Dr. Chung report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A new case series describes peripheral nerve injuries associated with this type of positioning and suggests ways to minimize the potential damage.

“Physicians should remain aware of increased susceptibility to peripheral nerve damage in patients with severe COVID-19 after prone positioning, since it is surprisingly common among these patients, and should refine standard protocols accordingly to reduce that risk,” said senior author Colin Franz, MD, PhD, director of the Electrodiagnostic Laboratory, Shirley Ryan AbilityLab, Chicago.
The article was published online Sept. 4 in the British Journal of Anaesthesiology.
Unique type of nerve injury
Many patients who are admitted to the intensive care unit with COVID-19 undergo invasive mechanical ventilation because of acute respiratory distress syndrome (ARDS). Clinical guidelines recommend that such patients lie in the prone position 12-16 hours per day.
“Prone positioning for up to 16 hours is a therapy we use for patients with more severe forms of ARDS, and high-level evidence points to mortality benefit in patients with moderate to severe ARDS if [mechanical] ventilation occurs,” said study coauthor James McCauley Walter, MD, of the pulmonary division at Northwestern University, Chicago.
With a “significant number of COVID-19 patients flooding the ICU, we quickly started to prone a lot of them, but if you are in a specific position for multiple hours a day, coupled with the neurotoxic effects of the SARS-CoV-2 virus itself, you may be exposed to a unique type of nerve injury,” he said.
Dr. Walter said that the “incidence of asymmetric neuropathies seems out of proportion to what has been reported in non–COVID-19 settings, which is what caught our attention.”
Many of these patients are discharged to rehabilitation hospitals, and “what we noticed, which was unique about COVID-19 patients coming to our rehab hospital, was that, compared with other patients who had been critically ill with a long hospital stay, there was a significantly higher percentage of COVID-19 patients who had peripheral nerve damage,” Dr. Franz said.
The authors described 12 of these patients who were admitted between April 24 and June 30, 2020 (mean age, 60.3 years; range, 23-80 years). The sample included White, Black, and Hispanic individuals. Eleven of the 12 post–COVID-19 patients with peripheral nerve damage had experienced prone positioning during acute management.
The average number of days patients received mechanical ventilation was 33.6 (range, 12-62 days). The average number of proning sessions was 4.5 (range, 1-16) with an average of 81.2 hours (range, 16-252 hours) spent prone.
A major contributor
Dr. Franz suggested that prone positioning is likely not the only cause of peripheral nerve damage but “may play a big role in these patients who are vulnerable because of viral infection and the critical illness that causes damage and nerve injuries.”
“The first component of lifesaving care for the critically ill in the ICU is intravenous fluids, mechanical ventilation, steroids, and antibiotics for infection,” said Dr. Walter.
“We are trying to come up with ways to place patients in prone position in safer ways, to pay attention to pressure points and areas of injury that we have seen and try to offload them, to see if we can decrease the rate of these injuries,” he added.
The researchers’ article includes a heat map diagram as a “template for where to focus the most efforts, in terms of decreasing pressure,” Dr. Walter said.
“The nerves are accepting too much force for gravely ill COVID-19 patients to handle, so we suggest using the template to determine where extra padding might be needed, or a protocol that might include changes in positioning,” he added.
Dr. Franz described the interventions used for COVID-19 patients with prone positioning–related peripheral nerve damage. “The first step is trying to address the problems one by one, either trying to solve them through exercise or teaching new skills, new ways to compensate, beginning with basic activities, such as getting out of bed and self-care,” he said.
Long-term recovery of nerve injuries depends on how severe the injuries are. Some nerves can slowly regenerate – possibly at the rate of 1 inch per month – which can be a long process, taking between a year and 18 months.
Dr. Franz said that therapies for this condition are “extrapolated from clinical trial work” on promoting nerve regeneration after surgery using electrical stimulation to enable nerves to regrow at a faster rate.
“Regeneration is not only slow, but it may not happen completely, leaving the patient with permanent nerve damage – in fact, based on our experience and what has been reported, the percentage of patients with full recovery is only 10%,” he said.
The most common symptomatic complaint other than lack of movement or feeling is neuropathic pain, “which may require medication to take the edge off the pain,” Dr. Franz added.
Irreversible damage?
Commenting on the study, Tae Chung, MD, of the departments of physical medicine, rehabilitation, and neurology, Johns Hopkins University, Baltimore, said the study “provides one of the first and the largest description of peripheral nerve injury associated with prone positioning for management of ARDS from COVID-19.”
Dr. Chung, who was not involved in the research, noted that “various neurological complications from COVID-19 have been reported, and some of them may result in irreversible neurological damage or delay the recovery from COVID-19 infection,” so “accurate and timely diagnosis of such neurological complications is critical for rehabilitation of the COVID-19 survivors.”
The study received no funding. Dr. Franz, Dr. Walter, study coauthors, and Dr. Chung report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM THE BRITISH JOURNAL OF ANAESTHESIOLOGY
AHA scientific statement highlights cardiorenal benefit of new diabetes drugs
To protect the heart and kidneys, sodium-glucose transporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists should be considered for people with type 2 diabetes and chronic kidney disease (CKD), the American Heart Association advised in a new scientific statement.
Taken together, the results of relevant clinical trials indicate that SGLT2 inhibitors and GLP-1 receptor agonists safely and significantly reduce the risk for cardiovascular (CV) events, death, and the slow progression of CKD to end-stage kidney disease, including the risks for dialysis, transplantation, and death, the writing group says.
The scientific statement was published online Sept. 28 in Circulation.
“There has been rapid reporting of high-quality data in the cardio-renal-metabolic space with significant heart and kidney benefits, particularly with these two newer classes of antihyperglycemic agents,” Janani Rangaswami, MD, who chaired the writing group, said in an interview.
“More recent data show benefits in chronic kidney disease and heart failure even in patients without diabetes,” said Dr. Rangaswami, Einstein Medical Center and Sidney Kimmel Medical College, both in Philadelphia.
“These data are practice-changing in both cardiology and nephrology, and usher in a new era of disease-modifying therapies in heart and kidney disease,” Dr. Rangaswami added.
Recommendations at a glance
- Provide early and ongoing assessment of risks for CVD and CKD to patients who may benefit from SGLT2 inhibitors of GLP-1 receptor agonists.
- Tailor medication choices that meet the needs of individual patients. Realize that, given “consistent class-wide effects,” the choice of a specific SGLT2 inhibitor or GLP-1 receptor agonist may be dictated by affordability, coverage, and formulary considerations.
- Adjust all medications in tandem with these medicines and consider the burden of polypharmacy, which is common among people with type 2 diabetes. Adjust concomitant therapies and deprescribe where possible.
- Identify risks for hypoglycemia and educate patients on the signs so they can seek treatment quickly.
- Monitor and control high blood pressure.
- Counsel patients about the risks for and symptoms of euglycemic diabetic ketoacidosis when taking SGLT2 inhibitors, as well as classic DKA, which can be fatal.
- Regularly screen and counsel patients about foot care to prevent foot ulcers or blisters that can quickly become infected and lead to amputation.
The writing group identified two additional patient subgroups that may benefit from SGLT2 inhibitors and GLP-1 receptor agonists: those with heart failure with reduced ejection fraction with or without diabetes; and those with CKD who do not have diabetes. They say more data are anticipated to validate the use of SGLT2 inhibitors and GLP-1 receptor agonists in these “at-risk” patients.
Collaborative care model
The writing group proposed a collaborative care model, bridging cardiologists, nephrologists, endocrinologists, and primary care physicians, to help facilitate the “prompt and appropriate” integration of these new classes of medications in the management of patients with type 2 diabetes and CKD.
There is “an unmet need for a cardio-renal-metabolic care model that incorporates best practices in the real world to help align these therapies, especially with vulnerable high-risk patients with cardiorenal disease, and to overcome barriers toward uptake of these agents. Hopefully this statement provides some guidance to the cardiology and nephrology communities in that area,” Dr. Rangaswami said in an interview.
But old habits die hard, as research continues to show the slow adoption of these newer medications in the real world.
For example, a large observational study published last year showed a “striking” discordance between evidence-based, guideline-recommended use of SGLT2 inhibitors for the treatment of type 2 diabetes and their actual uptake in clinical practice.
Paradoxically, patients with CVD, heart failure, hypertension, CKD, and those at risk for hypoglycemia were less apt to receive an SGLT2 inhibitor than other patients.
“The relatively slow uptake of these agents is multifactorial,” Dr. Rangaswami said. “Cardiologists and nephrologists may suffer from some level of ‘therapeutic inertia’ when using new agents they are unfamiliar with and originally branded as ‘antidiabetic’ agents, with the perception of these agents being outside the scope of their practice.”
Two other factors are also at play. “The current health care system is based on ‘specialty silos,’ where specialists tend to stick to the traditional scope of their specialty and are reluctant to view these agents as part of their therapeutic armamentarium. Finally, insurance coverage barriers and affordability also limit the use on a widespread basis,” Dr. Rangaswami said.
A version of this article originally appeared on Medscape.com .
To protect the heart and kidneys, sodium-glucose transporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists should be considered for people with type 2 diabetes and chronic kidney disease (CKD), the American Heart Association advised in a new scientific statement.
Taken together, the results of relevant clinical trials indicate that SGLT2 inhibitors and GLP-1 receptor agonists safely and significantly reduce the risk for cardiovascular (CV) events, death, and the slow progression of CKD to end-stage kidney disease, including the risks for dialysis, transplantation, and death, the writing group says.
The scientific statement was published online Sept. 28 in Circulation.
“There has been rapid reporting of high-quality data in the cardio-renal-metabolic space with significant heart and kidney benefits, particularly with these two newer classes of antihyperglycemic agents,” Janani Rangaswami, MD, who chaired the writing group, said in an interview.
“More recent data show benefits in chronic kidney disease and heart failure even in patients without diabetes,” said Dr. Rangaswami, Einstein Medical Center and Sidney Kimmel Medical College, both in Philadelphia.
“These data are practice-changing in both cardiology and nephrology, and usher in a new era of disease-modifying therapies in heart and kidney disease,” Dr. Rangaswami added.
Recommendations at a glance
- Provide early and ongoing assessment of risks for CVD and CKD to patients who may benefit from SGLT2 inhibitors of GLP-1 receptor agonists.
- Tailor medication choices that meet the needs of individual patients. Realize that, given “consistent class-wide effects,” the choice of a specific SGLT2 inhibitor or GLP-1 receptor agonist may be dictated by affordability, coverage, and formulary considerations.
- Adjust all medications in tandem with these medicines and consider the burden of polypharmacy, which is common among people with type 2 diabetes. Adjust concomitant therapies and deprescribe where possible.
- Identify risks for hypoglycemia and educate patients on the signs so they can seek treatment quickly.
- Monitor and control high blood pressure.
- Counsel patients about the risks for and symptoms of euglycemic diabetic ketoacidosis when taking SGLT2 inhibitors, as well as classic DKA, which can be fatal.
- Regularly screen and counsel patients about foot care to prevent foot ulcers or blisters that can quickly become infected and lead to amputation.
The writing group identified two additional patient subgroups that may benefit from SGLT2 inhibitors and GLP-1 receptor agonists: those with heart failure with reduced ejection fraction with or without diabetes; and those with CKD who do not have diabetes. They say more data are anticipated to validate the use of SGLT2 inhibitors and GLP-1 receptor agonists in these “at-risk” patients.
Collaborative care model
The writing group proposed a collaborative care model, bridging cardiologists, nephrologists, endocrinologists, and primary care physicians, to help facilitate the “prompt and appropriate” integration of these new classes of medications in the management of patients with type 2 diabetes and CKD.
There is “an unmet need for a cardio-renal-metabolic care model that incorporates best practices in the real world to help align these therapies, especially with vulnerable high-risk patients with cardiorenal disease, and to overcome barriers toward uptake of these agents. Hopefully this statement provides some guidance to the cardiology and nephrology communities in that area,” Dr. Rangaswami said in an interview.
But old habits die hard, as research continues to show the slow adoption of these newer medications in the real world.
For example, a large observational study published last year showed a “striking” discordance between evidence-based, guideline-recommended use of SGLT2 inhibitors for the treatment of type 2 diabetes and their actual uptake in clinical practice.
Paradoxically, patients with CVD, heart failure, hypertension, CKD, and those at risk for hypoglycemia were less apt to receive an SGLT2 inhibitor than other patients.
“The relatively slow uptake of these agents is multifactorial,” Dr. Rangaswami said. “Cardiologists and nephrologists may suffer from some level of ‘therapeutic inertia’ when using new agents they are unfamiliar with and originally branded as ‘antidiabetic’ agents, with the perception of these agents being outside the scope of their practice.”
Two other factors are also at play. “The current health care system is based on ‘specialty silos,’ where specialists tend to stick to the traditional scope of their specialty and are reluctant to view these agents as part of their therapeutic armamentarium. Finally, insurance coverage barriers and affordability also limit the use on a widespread basis,” Dr. Rangaswami said.
A version of this article originally appeared on Medscape.com .
To protect the heart and kidneys, sodium-glucose transporter 2 (SGLT2) inhibitors and glucagonlike peptide–1 (GLP-1) receptor agonists should be considered for people with type 2 diabetes and chronic kidney disease (CKD), the American Heart Association advised in a new scientific statement.
Taken together, the results of relevant clinical trials indicate that SGLT2 inhibitors and GLP-1 receptor agonists safely and significantly reduce the risk for cardiovascular (CV) events, death, and the slow progression of CKD to end-stage kidney disease, including the risks for dialysis, transplantation, and death, the writing group says.
The scientific statement was published online Sept. 28 in Circulation.
“There has been rapid reporting of high-quality data in the cardio-renal-metabolic space with significant heart and kidney benefits, particularly with these two newer classes of antihyperglycemic agents,” Janani Rangaswami, MD, who chaired the writing group, said in an interview.
“More recent data show benefits in chronic kidney disease and heart failure even in patients without diabetes,” said Dr. Rangaswami, Einstein Medical Center and Sidney Kimmel Medical College, both in Philadelphia.
“These data are practice-changing in both cardiology and nephrology, and usher in a new era of disease-modifying therapies in heart and kidney disease,” Dr. Rangaswami added.
Recommendations at a glance
- Provide early and ongoing assessment of risks for CVD and CKD to patients who may benefit from SGLT2 inhibitors of GLP-1 receptor agonists.
- Tailor medication choices that meet the needs of individual patients. Realize that, given “consistent class-wide effects,” the choice of a specific SGLT2 inhibitor or GLP-1 receptor agonist may be dictated by affordability, coverage, and formulary considerations.
- Adjust all medications in tandem with these medicines and consider the burden of polypharmacy, which is common among people with type 2 diabetes. Adjust concomitant therapies and deprescribe where possible.
- Identify risks for hypoglycemia and educate patients on the signs so they can seek treatment quickly.
- Monitor and control high blood pressure.
- Counsel patients about the risks for and symptoms of euglycemic diabetic ketoacidosis when taking SGLT2 inhibitors, as well as classic DKA, which can be fatal.
- Regularly screen and counsel patients about foot care to prevent foot ulcers or blisters that can quickly become infected and lead to amputation.
The writing group identified two additional patient subgroups that may benefit from SGLT2 inhibitors and GLP-1 receptor agonists: those with heart failure with reduced ejection fraction with or without diabetes; and those with CKD who do not have diabetes. They say more data are anticipated to validate the use of SGLT2 inhibitors and GLP-1 receptor agonists in these “at-risk” patients.
Collaborative care model
The writing group proposed a collaborative care model, bridging cardiologists, nephrologists, endocrinologists, and primary care physicians, to help facilitate the “prompt and appropriate” integration of these new classes of medications in the management of patients with type 2 diabetes and CKD.
There is “an unmet need for a cardio-renal-metabolic care model that incorporates best practices in the real world to help align these therapies, especially with vulnerable high-risk patients with cardiorenal disease, and to overcome barriers toward uptake of these agents. Hopefully this statement provides some guidance to the cardiology and nephrology communities in that area,” Dr. Rangaswami said in an interview.
But old habits die hard, as research continues to show the slow adoption of these newer medications in the real world.
For example, a large observational study published last year showed a “striking” discordance between evidence-based, guideline-recommended use of SGLT2 inhibitors for the treatment of type 2 diabetes and their actual uptake in clinical practice.
Paradoxically, patients with CVD, heart failure, hypertension, CKD, and those at risk for hypoglycemia were less apt to receive an SGLT2 inhibitor than other patients.
“The relatively slow uptake of these agents is multifactorial,” Dr. Rangaswami said. “Cardiologists and nephrologists may suffer from some level of ‘therapeutic inertia’ when using new agents they are unfamiliar with and originally branded as ‘antidiabetic’ agents, with the perception of these agents being outside the scope of their practice.”
Two other factors are also at play. “The current health care system is based on ‘specialty silos,’ where specialists tend to stick to the traditional scope of their specialty and are reluctant to view these agents as part of their therapeutic armamentarium. Finally, insurance coverage barriers and affordability also limit the use on a widespread basis,” Dr. Rangaswami said.
A version of this article originally appeared on Medscape.com .
COVID-19 shutdown fuels sharp rise in alcohol use
Americans sharply increased their alcohol intake last spring as many areas of the country shutdown because of the coronavirus pandemic, results of a national survey show.
The overall frequency of alcohol consumption increased by 14% among adults over age 30 in the spring of 2020 versus the same period a year earlier.
The increase was most evident in adults aged 30-59, women, and non-Hispanic Whites.
“Alcohol consumption can have significant negative health consequences, so this information suggests another way that the pandemic may be affecting the physical and mental health of Americans,” Michael Pollard, PhD, lead investigator and sociologist at Rand, said in a news release.
The results were published online as a research letter Sept. 29 in JAMA Network Open.
Booming business
After some U.S. states issued stay-at-home orders to fight the spread of SARS-CoV-2, one study noted a 54% increase in national sales of alcohol for the week ending March 21, 2020, relative to 1 year earlier and a 262% increase in online alcohol sales.
“We’ve had anecdotal information about people buying and consuming more alcohol,” Dr. Pollard said, but the Rand study provides the first survey-based information that shows how much alcohol consumption has increased during the pandemic.
The findings are based on 1,540 adults (mean age, 56.6 years; 57% women) from the Rand American Life Panel, a nationally representative sample of Americans who were surveyed about their alcohol consumption before the pandemic in the spring of 2019, and again in the spring of 2020 during the early months of the shutdown.
Overall, in spring 2020, respondents reported drinking alcohol 6.22 days in the prior month on average, a 14% increase from the monthly average of 5.48 days reported in spring 2019.
Among adults aged 30 to 59 years, the frequency of alcohol consumption increased from 4.98 days prepandemic to 5.91 days during the pandemic, a 19% increase.
Women reported drinking an average of 5.36 days in the prior month in the early pandemic period, a 17% increase from 4.58 monthly drinking days before the pandemic.
In addition, compared with spring 2019, in spring 2020 women reported a 41% increase in heavy drinking days – four or more drinks in a couple of hours.
Independent of consumption level, nearly 1 in 10 women had an increase in alcohol-related problems in the pandemic period, based on responses to the Short Inventory of Problems scale.
For non-Hispanic White individuals, the overall frequency of alcohol intake rose 10% during the early pandemic period.
“The population level changes for women, younger, and non-Hispanic White individuals highlight that health systems may need to educate consumers through print or online media about increased alcohol use during the pandemic and identify factors associated with susceptibility and resilience to the impacts of COVID-19,” write Dr. Pollard and colleagues.
The authors note , and whether psychological and physical well-being are subsequently affected.
The study was supported by the National Institute of Alcohol Abuse and Alcoholism. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Americans sharply increased their alcohol intake last spring as many areas of the country shutdown because of the coronavirus pandemic, results of a national survey show.
The overall frequency of alcohol consumption increased by 14% among adults over age 30 in the spring of 2020 versus the same period a year earlier.
The increase was most evident in adults aged 30-59, women, and non-Hispanic Whites.
“Alcohol consumption can have significant negative health consequences, so this information suggests another way that the pandemic may be affecting the physical and mental health of Americans,” Michael Pollard, PhD, lead investigator and sociologist at Rand, said in a news release.
The results were published online as a research letter Sept. 29 in JAMA Network Open.
Booming business
After some U.S. states issued stay-at-home orders to fight the spread of SARS-CoV-2, one study noted a 54% increase in national sales of alcohol for the week ending March 21, 2020, relative to 1 year earlier and a 262% increase in online alcohol sales.
“We’ve had anecdotal information about people buying and consuming more alcohol,” Dr. Pollard said, but the Rand study provides the first survey-based information that shows how much alcohol consumption has increased during the pandemic.
The findings are based on 1,540 adults (mean age, 56.6 years; 57% women) from the Rand American Life Panel, a nationally representative sample of Americans who were surveyed about their alcohol consumption before the pandemic in the spring of 2019, and again in the spring of 2020 during the early months of the shutdown.
Overall, in spring 2020, respondents reported drinking alcohol 6.22 days in the prior month on average, a 14% increase from the monthly average of 5.48 days reported in spring 2019.
Among adults aged 30 to 59 years, the frequency of alcohol consumption increased from 4.98 days prepandemic to 5.91 days during the pandemic, a 19% increase.
Women reported drinking an average of 5.36 days in the prior month in the early pandemic period, a 17% increase from 4.58 monthly drinking days before the pandemic.
In addition, compared with spring 2019, in spring 2020 women reported a 41% increase in heavy drinking days – four or more drinks in a couple of hours.
Independent of consumption level, nearly 1 in 10 women had an increase in alcohol-related problems in the pandemic period, based on responses to the Short Inventory of Problems scale.
For non-Hispanic White individuals, the overall frequency of alcohol intake rose 10% during the early pandemic period.
“The population level changes for women, younger, and non-Hispanic White individuals highlight that health systems may need to educate consumers through print or online media about increased alcohol use during the pandemic and identify factors associated with susceptibility and resilience to the impacts of COVID-19,” write Dr. Pollard and colleagues.
The authors note , and whether psychological and physical well-being are subsequently affected.
The study was supported by the National Institute of Alcohol Abuse and Alcoholism. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Americans sharply increased their alcohol intake last spring as many areas of the country shutdown because of the coronavirus pandemic, results of a national survey show.
The overall frequency of alcohol consumption increased by 14% among adults over age 30 in the spring of 2020 versus the same period a year earlier.
The increase was most evident in adults aged 30-59, women, and non-Hispanic Whites.
“Alcohol consumption can have significant negative health consequences, so this information suggests another way that the pandemic may be affecting the physical and mental health of Americans,” Michael Pollard, PhD, lead investigator and sociologist at Rand, said in a news release.
The results were published online as a research letter Sept. 29 in JAMA Network Open.
Booming business
After some U.S. states issued stay-at-home orders to fight the spread of SARS-CoV-2, one study noted a 54% increase in national sales of alcohol for the week ending March 21, 2020, relative to 1 year earlier and a 262% increase in online alcohol sales.
“We’ve had anecdotal information about people buying and consuming more alcohol,” Dr. Pollard said, but the Rand study provides the first survey-based information that shows how much alcohol consumption has increased during the pandemic.
The findings are based on 1,540 adults (mean age, 56.6 years; 57% women) from the Rand American Life Panel, a nationally representative sample of Americans who were surveyed about their alcohol consumption before the pandemic in the spring of 2019, and again in the spring of 2020 during the early months of the shutdown.
Overall, in spring 2020, respondents reported drinking alcohol 6.22 days in the prior month on average, a 14% increase from the monthly average of 5.48 days reported in spring 2019.
Among adults aged 30 to 59 years, the frequency of alcohol consumption increased from 4.98 days prepandemic to 5.91 days during the pandemic, a 19% increase.
Women reported drinking an average of 5.36 days in the prior month in the early pandemic period, a 17% increase from 4.58 monthly drinking days before the pandemic.
In addition, compared with spring 2019, in spring 2020 women reported a 41% increase in heavy drinking days – four or more drinks in a couple of hours.
Independent of consumption level, nearly 1 in 10 women had an increase in alcohol-related problems in the pandemic period, based on responses to the Short Inventory of Problems scale.
For non-Hispanic White individuals, the overall frequency of alcohol intake rose 10% during the early pandemic period.
“The population level changes for women, younger, and non-Hispanic White individuals highlight that health systems may need to educate consumers through print or online media about increased alcohol use during the pandemic and identify factors associated with susceptibility and resilience to the impacts of COVID-19,” write Dr. Pollard and colleagues.
The authors note , and whether psychological and physical well-being are subsequently affected.
The study was supported by the National Institute of Alcohol Abuse and Alcoholism. The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Children’s share of new COVID-19 cases is on the rise
The cumulative percentage of COVID-19 cases reported in children continues to climb, but “the history behind that cumulative number shows substantial change,” according to a new analysis of state health department data.
As of Sept. 10, the 549,432 cases in children represented 10.0% of all reported COVID-19 cases in the United States following a substantial rise over the course of the pandemic – the figure was 7.7% on July 16 and 3.2% on May 7, Blake Sisk, PhD, of the American Academy of Pediatrics and associates reported Sept. 29 in Pediatrics.
Unlike the cumulative number, the weekly proportion of cases in children fell early in the summer but then started climbing again in late July. Dr. Sisk and associates wrote.
Despite the increase, however, the proportion of pediatric COVID-19 cases is still well below children’s share of the overall population (22.6%). Also, “it is unclear how much of the increase in child cases is due to increased testing capacity, although CDC data from public and commercial laboratories show the share of all tests administered to children ages 0-17 has remained stable at 5%-7% since late April,” they said.
Data for the current report were drawn from 49 state health department websites (New York state does not report ages for COVID-19 cases), along with New York City, the District of Columbia, Puerto Rico, and Guam. Alabama changed its definition of a child case in August and was not included in the trend analysis (see graph), the investigators explained.
Those data show “substantial variation in case growth by region: in April, a preponderance of cases was in the Northeast. In June, cases surged in the South and West, followed by mid-July increases in the Midwest,” Dr. Sisk and associates said.
The increase among children in Midwest states is ongoing with the number of new cases reaching its highest level yet during the week ending Sept. 10, they reported.
SOURCE: Sisk B et al. Pediatrics. 2020 Sep 29. doi: 10.1542/peds.2020-027425.
The cumulative percentage of COVID-19 cases reported in children continues to climb, but “the history behind that cumulative number shows substantial change,” according to a new analysis of state health department data.

As of Sept. 10, the 549,432 cases in children represented 10.0% of all reported COVID-19 cases in the United States following a substantial rise over the course of the pandemic – the figure was 7.7% on July 16 and 3.2% on May 7, Blake Sisk, PhD, of the American Academy of Pediatrics and associates reported Sept. 29 in Pediatrics.
Unlike the cumulative number, the weekly proportion of cases in children fell early in the summer but then started climbing again in late July. Dr. Sisk and associates wrote.
Despite the increase, however, the proportion of pediatric COVID-19 cases is still well below children’s share of the overall population (22.6%). Also, “it is unclear how much of the increase in child cases is due to increased testing capacity, although CDC data from public and commercial laboratories show the share of all tests administered to children ages 0-17 has remained stable at 5%-7% since late April,” they said.
Data for the current report were drawn from 49 state health department websites (New York state does not report ages for COVID-19 cases), along with New York City, the District of Columbia, Puerto Rico, and Guam. Alabama changed its definition of a child case in August and was not included in the trend analysis (see graph), the investigators explained.
Those data show “substantial variation in case growth by region: in April, a preponderance of cases was in the Northeast. In June, cases surged in the South and West, followed by mid-July increases in the Midwest,” Dr. Sisk and associates said.
The increase among children in Midwest states is ongoing with the number of new cases reaching its highest level yet during the week ending Sept. 10, they reported.
SOURCE: Sisk B et al. Pediatrics. 2020 Sep 29. doi: 10.1542/peds.2020-027425.
The cumulative percentage of COVID-19 cases reported in children continues to climb, but “the history behind that cumulative number shows substantial change,” according to a new analysis of state health department data.

As of Sept. 10, the 549,432 cases in children represented 10.0% of all reported COVID-19 cases in the United States following a substantial rise over the course of the pandemic – the figure was 7.7% on July 16 and 3.2% on May 7, Blake Sisk, PhD, of the American Academy of Pediatrics and associates reported Sept. 29 in Pediatrics.
Unlike the cumulative number, the weekly proportion of cases in children fell early in the summer but then started climbing again in late July. Dr. Sisk and associates wrote.
Despite the increase, however, the proportion of pediatric COVID-19 cases is still well below children’s share of the overall population (22.6%). Also, “it is unclear how much of the increase in child cases is due to increased testing capacity, although CDC data from public and commercial laboratories show the share of all tests administered to children ages 0-17 has remained stable at 5%-7% since late April,” they said.
Data for the current report were drawn from 49 state health department websites (New York state does not report ages for COVID-19 cases), along with New York City, the District of Columbia, Puerto Rico, and Guam. Alabama changed its definition of a child case in August and was not included in the trend analysis (see graph), the investigators explained.
Those data show “substantial variation in case growth by region: in April, a preponderance of cases was in the Northeast. In June, cases surged in the South and West, followed by mid-July increases in the Midwest,” Dr. Sisk and associates said.
The increase among children in Midwest states is ongoing with the number of new cases reaching its highest level yet during the week ending Sept. 10, they reported.
SOURCE: Sisk B et al. Pediatrics. 2020 Sep 29. doi: 10.1542/peds.2020-027425.
FROM PEDIATRICS
Pandemic poses new challenges for rural doctors
These include struggling with seeing patients virtually and treating patients who have politicized the virus. Additionally, the pandemic has exposed rural practices to greater financial difficulties.
Before the pandemic some rurally based primary care physicians were already working through big challenges, such as having few local medical colleagues to consult and working in small practices with lean budgets. In fact, data gathered by the National Rural Health Association showed that there are only 40 primary care physicians per 100,000 patients in rural regions, compared with 53 in urban areas – and the number of physicians overall is 13 per 10,000 in rural areas, compared with 31 in cities.
In the prepandemic world, for some doctors, the challenges were balanced by the benefits of practicing in these sparsely populated communities with scenic, low-traffic roads. Some perks of practicing in rural areas touted by doctors included having a fast commute, being able to swim in a lake near the office before work, having a low cost of living, and feeling like they are making a difference in their communities as they treat generations of the families they see around town.
But today, new hurdles to practicing medicine in rural America created by the COVID-19 pandemic have caused the hardships to feel heavier than the joys at times for some physicians interviewed by MDedge.
Many independent rural practices in need of assistance were not able to get much from the federal Provider Relief Funds, said John M. Westfall, MD, who is director of the Robert Graham Center for Policy Studies in Family Medicine and Primary Care, in an interview.
“Rural primary care doctors function independently or in smaller critical access hospitals and community health centers,” said Dr. Westfall, who previously practiced family medicine in a small town in Colorado. “Many of these have much less financial reserves so are at risk of cutbacks and closure.”
Jacqueline W. Fincher, MD, an internist based in a tiny Georgia community along the highway between Atlanta and Augusta, said her small practice works on really thin margins and doesn’t have much cushion. At the beginning of the pandemic, all visits were down, and her practice operated at a loss. To help, Dr. Fincher and her colleagues applied for funding from the Small Business Administration’s Paycheck Protection Program (PPP) through the CARES Act.
“COVID-19 has had a tremendous impact especially on primary care practices. We live and die by volume. … Our volume in mid-March to mid-May really dropped dramatically,” explained Dr. Fincher, who is also president of the American College of Physicians. “The PPP sustained us for 2 months, enabling us to pay our staff and to remain open and get us up and running on telehealth.”
Starting up telemedicine
Experiencing spotty or no access to broadband Internet is nothing new to rural physicians, but having this problem interfere with their ability to provide care to patients is.
As much of the American health system rapidly embraced telehealth during the pandemic, obtaining access to high-speed Internet has been a major challenge for rural patients, noted Dr. Westfall.
“Some practices were able to quickly adopt some telehealth capacity with phone and video. Changes in payment for telehealth helped. But in some rural communities there was not adequate Internet bandwidth for quality video connections. And some patients did not have the means for high-speed video connections,” Dr. Westfall said.
Indeed, according to a 2019 Pew Research Center survey, 63% of rural Americans say they can access the Internet through a broadband connection at home, compared with 75% and 79% in suburban and urban areas, respectively.
In the Appalachian town of Zanesville, Ohio, for example, family physician Shelly L. Dunmyer, MD, and her colleagues discovered that many patients don’t have Internet access at home. Dr. Fincher has to go to the office to conduct telehealth visits because her own Internet access at home is unpredictable. As for patients, it may take 15 minutes for them to work out technical glitches and find good Internet reception, said Dr. Fincher. For internist Y. Ki Shin, MD, who practices in the coastal town of Montesano in Washington state, about 25% of his practice’s telehealth visits must be conducted by phone because of limitations on video, such as lack of high-speed access.
But telephone visits are often insufficient replacements for appointments via video, according to several rural physicians interviewed for this piece.
“Telehealth can be frustrating at times due to connectivity issues which can be difficult at times in the rural areas,” said Dr. Fincher. “In order for telehealth to be reasonably helpful to patients and physicians to care for people with chronic problems, the patients must have things like blood pressure monitors, glucometers, and scales to address problems like hypertension, diabetes myelitis, and congestive heart failure.”
“If you have the audio and video and the data from these devices, you’re good. If you don’t have these data, and/or don’t have the video you just can’t provide good care,” she explained.
Dr. Dunmyer and her colleagues at Medical Home Primary Care Center in Zanesville, Ohio, found a way to get around the problem of patients not being able to access Internet to participate in video visits from their homes. This involved having her patients drive into her practice’s parking lot to participate in modified telehealth visits. Staffers gave iPads to patients in their cars, and Dr. Dunmyer conducted visits from her office, about 50 yards away.
“We were even doing Medicare wellness visits: Instead of asking them to get up and move around the room, we would sit at the window and wave at them, ask them to get out, walk around the car. We were able to check mobility and all kinds of things that we’d normally do in the office,” Dr. Dunmyer explained in an interview.
The family physician noted that her practice is now conducting fewer parking lot visits since her office is allowing in-person appointments, but that they’re still an option for her patients.
Treating political adversaries
Some rural physicians have experienced strained relationships with patients for reasons other than technology – stark differences in opinion over the pandemic itself. Certain patients are following President Trump’s lead and questioning everything from the pandemic death toll to preventive measures recommended by scientists and medical experts, physicians interviewed by MDedge said.
Patients everywhere share these viewpoints, of course, but research and election results confirm that rural areas are more receptive to conservative viewpoints. In 2018, a Pew Research Center survey reported that rural and urban areas are “becoming more polarized politically,” and “rural areas tend to have a higher concentration of Republicans and Republican-leaning independents.” For example, 40% of rural respondents reported “very warm” or “somewhat warm” feelings toward Donald Trump, compared with just 19% in urban areas.
Dr. Shin has struggled to cope with patients who want to argue about pandemic safety precautions like wearing masks and seem to question whether systemic racism exists.
“We are seeing a lot more people who feel that this pandemic is not real, that it’s a political and not-true infection,” he said in an interview. “We’ve had patients who were angry at us because we made them wear masks, and some were demanding hydroxychloroquine and wanted to have an argument because we’re not going to prescribe it for them.”
In one situation, which he found especially disturbing, Dr. Shin had to leave the exam room because a patient wouldn’t stop challenging him regarding the pandemic. Things have gotten so bad that Dr. Shin has even questioned whether he wants to continue his long career in his small town because of local political attitudes such as opposition to mask-wearing and social distancing.
“Mr. Trump’s misinformation on this pandemic made my job much more difficult. As a minority, I feel less safe in my community than ever,” said Dr. Shin, who described himself as Asian American.
Despite these new stressors, Dr. Shin has experienced some joyful moments while practicing medicine in the pandemic.
He said a recent home visit to a patient who had been hospitalized for over 3 months and nearly died helped him put political disputes with his patients into perspective.
“He was discharged home but is bedbound. He had gangrene on his toes, and I could not fully examine him using video,” Dr. Shin recalled. “It was tricky to find the house, but a very large Trump sign was very helpful in locating it. It was a good visit: He was happy to see me, and I was happy to see that he was doing okay at home.”
“I need to remind myself that supporting Mr. Trump does not always mean that my patient supports Mr. Trump’s view on the pandemic and the race issues in our country,” Dr. Shin added.
The Washington-based internist said he also tells himself that, even if his patients refuse to follow his strong advice regarding pandemic precautions, it does not mean he has failed as a doctor.
“I need to continue to educate patients about the dangers of COVID infection but cannot be angry if they don’t choose to follow my recommendations,” he noted.
Dr. Fincher says her close connection with patients has allowed her to smooth over politically charged claims about the pandemic in the town of Thomson, Georgia, with a population 6,800.
“I have a sense that, even though we may differ in our understanding of some basic facts, they appreciate what I say since we have a long-term relationship built on trust,” she said. This kind of trust, Dr. Fincher suggested, may be more common than in urban areas where there’s a larger supply of physicians, and patients don’t see the same doctors for long periods of time.
“It’s more meaningful when it comes from me, rather than doctors who are [new to patients] every year when their employer changes their insurance,” she noted.
These include struggling with seeing patients virtually and treating patients who have politicized the virus. Additionally, the pandemic has exposed rural practices to greater financial difficulties.
Before the pandemic some rurally based primary care physicians were already working through big challenges, such as having few local medical colleagues to consult and working in small practices with lean budgets. In fact, data gathered by the National Rural Health Association showed that there are only 40 primary care physicians per 100,000 patients in rural regions, compared with 53 in urban areas – and the number of physicians overall is 13 per 10,000 in rural areas, compared with 31 in cities.
In the prepandemic world, for some doctors, the challenges were balanced by the benefits of practicing in these sparsely populated communities with scenic, low-traffic roads. Some perks of practicing in rural areas touted by doctors included having a fast commute, being able to swim in a lake near the office before work, having a low cost of living, and feeling like they are making a difference in their communities as they treat generations of the families they see around town.
But today, new hurdles to practicing medicine in rural America created by the COVID-19 pandemic have caused the hardships to feel heavier than the joys at times for some physicians interviewed by MDedge.
Many independent rural practices in need of assistance were not able to get much from the federal Provider Relief Funds, said John M. Westfall, MD, who is director of the Robert Graham Center for Policy Studies in Family Medicine and Primary Care, in an interview.
“Rural primary care doctors function independently or in smaller critical access hospitals and community health centers,” said Dr. Westfall, who previously practiced family medicine in a small town in Colorado. “Many of these have much less financial reserves so are at risk of cutbacks and closure.”
Jacqueline W. Fincher, MD, an internist based in a tiny Georgia community along the highway between Atlanta and Augusta, said her small practice works on really thin margins and doesn’t have much cushion. At the beginning of the pandemic, all visits were down, and her practice operated at a loss. To help, Dr. Fincher and her colleagues applied for funding from the Small Business Administration’s Paycheck Protection Program (PPP) through the CARES Act.
“COVID-19 has had a tremendous impact especially on primary care practices. We live and die by volume. … Our volume in mid-March to mid-May really dropped dramatically,” explained Dr. Fincher, who is also president of the American College of Physicians. “The PPP sustained us for 2 months, enabling us to pay our staff and to remain open and get us up and running on telehealth.”
Starting up telemedicine
Experiencing spotty or no access to broadband Internet is nothing new to rural physicians, but having this problem interfere with their ability to provide care to patients is.
As much of the American health system rapidly embraced telehealth during the pandemic, obtaining access to high-speed Internet has been a major challenge for rural patients, noted Dr. Westfall.
“Some practices were able to quickly adopt some telehealth capacity with phone and video. Changes in payment for telehealth helped. But in some rural communities there was not adequate Internet bandwidth for quality video connections. And some patients did not have the means for high-speed video connections,” Dr. Westfall said.
Indeed, according to a 2019 Pew Research Center survey, 63% of rural Americans say they can access the Internet through a broadband connection at home, compared with 75% and 79% in suburban and urban areas, respectively.
In the Appalachian town of Zanesville, Ohio, for example, family physician Shelly L. Dunmyer, MD, and her colleagues discovered that many patients don’t have Internet access at home. Dr. Fincher has to go to the office to conduct telehealth visits because her own Internet access at home is unpredictable. As for patients, it may take 15 minutes for them to work out technical glitches and find good Internet reception, said Dr. Fincher. For internist Y. Ki Shin, MD, who practices in the coastal town of Montesano in Washington state, about 25% of his practice’s telehealth visits must be conducted by phone because of limitations on video, such as lack of high-speed access.
But telephone visits are often insufficient replacements for appointments via video, according to several rural physicians interviewed for this piece.
“Telehealth can be frustrating at times due to connectivity issues which can be difficult at times in the rural areas,” said Dr. Fincher. “In order for telehealth to be reasonably helpful to patients and physicians to care for people with chronic problems, the patients must have things like blood pressure monitors, glucometers, and scales to address problems like hypertension, diabetes myelitis, and congestive heart failure.”
“If you have the audio and video and the data from these devices, you’re good. If you don’t have these data, and/or don’t have the video you just can’t provide good care,” she explained.
Dr. Dunmyer and her colleagues at Medical Home Primary Care Center in Zanesville, Ohio, found a way to get around the problem of patients not being able to access Internet to participate in video visits from their homes. This involved having her patients drive into her practice’s parking lot to participate in modified telehealth visits. Staffers gave iPads to patients in their cars, and Dr. Dunmyer conducted visits from her office, about 50 yards away.
“We were even doing Medicare wellness visits: Instead of asking them to get up and move around the room, we would sit at the window and wave at them, ask them to get out, walk around the car. We were able to check mobility and all kinds of things that we’d normally do in the office,” Dr. Dunmyer explained in an interview.
The family physician noted that her practice is now conducting fewer parking lot visits since her office is allowing in-person appointments, but that they’re still an option for her patients.
Treating political adversaries
Some rural physicians have experienced strained relationships with patients for reasons other than technology – stark differences in opinion over the pandemic itself. Certain patients are following President Trump’s lead and questioning everything from the pandemic death toll to preventive measures recommended by scientists and medical experts, physicians interviewed by MDedge said.
Patients everywhere share these viewpoints, of course, but research and election results confirm that rural areas are more receptive to conservative viewpoints. In 2018, a Pew Research Center survey reported that rural and urban areas are “becoming more polarized politically,” and “rural areas tend to have a higher concentration of Republicans and Republican-leaning independents.” For example, 40% of rural respondents reported “very warm” or “somewhat warm” feelings toward Donald Trump, compared with just 19% in urban areas.
Dr. Shin has struggled to cope with patients who want to argue about pandemic safety precautions like wearing masks and seem to question whether systemic racism exists.
“We are seeing a lot more people who feel that this pandemic is not real, that it’s a political and not-true infection,” he said in an interview. “We’ve had patients who were angry at us because we made them wear masks, and some were demanding hydroxychloroquine and wanted to have an argument because we’re not going to prescribe it for them.”
In one situation, which he found especially disturbing, Dr. Shin had to leave the exam room because a patient wouldn’t stop challenging him regarding the pandemic. Things have gotten so bad that Dr. Shin has even questioned whether he wants to continue his long career in his small town because of local political attitudes such as opposition to mask-wearing and social distancing.
“Mr. Trump’s misinformation on this pandemic made my job much more difficult. As a minority, I feel less safe in my community than ever,” said Dr. Shin, who described himself as Asian American.
Despite these new stressors, Dr. Shin has experienced some joyful moments while practicing medicine in the pandemic.
He said a recent home visit to a patient who had been hospitalized for over 3 months and nearly died helped him put political disputes with his patients into perspective.
“He was discharged home but is bedbound. He had gangrene on his toes, and I could not fully examine him using video,” Dr. Shin recalled. “It was tricky to find the house, but a very large Trump sign was very helpful in locating it. It was a good visit: He was happy to see me, and I was happy to see that he was doing okay at home.”
“I need to remind myself that supporting Mr. Trump does not always mean that my patient supports Mr. Trump’s view on the pandemic and the race issues in our country,” Dr. Shin added.
The Washington-based internist said he also tells himself that, even if his patients refuse to follow his strong advice regarding pandemic precautions, it does not mean he has failed as a doctor.
“I need to continue to educate patients about the dangers of COVID infection but cannot be angry if they don’t choose to follow my recommendations,” he noted.
Dr. Fincher says her close connection with patients has allowed her to smooth over politically charged claims about the pandemic in the town of Thomson, Georgia, with a population 6,800.
“I have a sense that, even though we may differ in our understanding of some basic facts, they appreciate what I say since we have a long-term relationship built on trust,” she said. This kind of trust, Dr. Fincher suggested, may be more common than in urban areas where there’s a larger supply of physicians, and patients don’t see the same doctors for long periods of time.
“It’s more meaningful when it comes from me, rather than doctors who are [new to patients] every year when their employer changes their insurance,” she noted.
These include struggling with seeing patients virtually and treating patients who have politicized the virus. Additionally, the pandemic has exposed rural practices to greater financial difficulties.
Before the pandemic some rurally based primary care physicians were already working through big challenges, such as having few local medical colleagues to consult and working in small practices with lean budgets. In fact, data gathered by the National Rural Health Association showed that there are only 40 primary care physicians per 100,000 patients in rural regions, compared with 53 in urban areas – and the number of physicians overall is 13 per 10,000 in rural areas, compared with 31 in cities.
In the prepandemic world, for some doctors, the challenges were balanced by the benefits of practicing in these sparsely populated communities with scenic, low-traffic roads. Some perks of practicing in rural areas touted by doctors included having a fast commute, being able to swim in a lake near the office before work, having a low cost of living, and feeling like they are making a difference in their communities as they treat generations of the families they see around town.
But today, new hurdles to practicing medicine in rural America created by the COVID-19 pandemic have caused the hardships to feel heavier than the joys at times for some physicians interviewed by MDedge.
Many independent rural practices in need of assistance were not able to get much from the federal Provider Relief Funds, said John M. Westfall, MD, who is director of the Robert Graham Center for Policy Studies in Family Medicine and Primary Care, in an interview.
“Rural primary care doctors function independently or in smaller critical access hospitals and community health centers,” said Dr. Westfall, who previously practiced family medicine in a small town in Colorado. “Many of these have much less financial reserves so are at risk of cutbacks and closure.”
Jacqueline W. Fincher, MD, an internist based in a tiny Georgia community along the highway between Atlanta and Augusta, said her small practice works on really thin margins and doesn’t have much cushion. At the beginning of the pandemic, all visits were down, and her practice operated at a loss. To help, Dr. Fincher and her colleagues applied for funding from the Small Business Administration’s Paycheck Protection Program (PPP) through the CARES Act.
“COVID-19 has had a tremendous impact especially on primary care practices. We live and die by volume. … Our volume in mid-March to mid-May really dropped dramatically,” explained Dr. Fincher, who is also president of the American College of Physicians. “The PPP sustained us for 2 months, enabling us to pay our staff and to remain open and get us up and running on telehealth.”
Starting up telemedicine
Experiencing spotty or no access to broadband Internet is nothing new to rural physicians, but having this problem interfere with their ability to provide care to patients is.
As much of the American health system rapidly embraced telehealth during the pandemic, obtaining access to high-speed Internet has been a major challenge for rural patients, noted Dr. Westfall.
“Some practices were able to quickly adopt some telehealth capacity with phone and video. Changes in payment for telehealth helped. But in some rural communities there was not adequate Internet bandwidth for quality video connections. And some patients did not have the means for high-speed video connections,” Dr. Westfall said.
Indeed, according to a 2019 Pew Research Center survey, 63% of rural Americans say they can access the Internet through a broadband connection at home, compared with 75% and 79% in suburban and urban areas, respectively.
In the Appalachian town of Zanesville, Ohio, for example, family physician Shelly L. Dunmyer, MD, and her colleagues discovered that many patients don’t have Internet access at home. Dr. Fincher has to go to the office to conduct telehealth visits because her own Internet access at home is unpredictable. As for patients, it may take 15 minutes for them to work out technical glitches and find good Internet reception, said Dr. Fincher. For internist Y. Ki Shin, MD, who practices in the coastal town of Montesano in Washington state, about 25% of his practice’s telehealth visits must be conducted by phone because of limitations on video, such as lack of high-speed access.
But telephone visits are often insufficient replacements for appointments via video, according to several rural physicians interviewed for this piece.
“Telehealth can be frustrating at times due to connectivity issues which can be difficult at times in the rural areas,” said Dr. Fincher. “In order for telehealth to be reasonably helpful to patients and physicians to care for people with chronic problems, the patients must have things like blood pressure monitors, glucometers, and scales to address problems like hypertension, diabetes myelitis, and congestive heart failure.”
“If you have the audio and video and the data from these devices, you’re good. If you don’t have these data, and/or don’t have the video you just can’t provide good care,” she explained.
Dr. Dunmyer and her colleagues at Medical Home Primary Care Center in Zanesville, Ohio, found a way to get around the problem of patients not being able to access Internet to participate in video visits from their homes. This involved having her patients drive into her practice’s parking lot to participate in modified telehealth visits. Staffers gave iPads to patients in their cars, and Dr. Dunmyer conducted visits from her office, about 50 yards away.
“We were even doing Medicare wellness visits: Instead of asking them to get up and move around the room, we would sit at the window and wave at them, ask them to get out, walk around the car. We were able to check mobility and all kinds of things that we’d normally do in the office,” Dr. Dunmyer explained in an interview.
The family physician noted that her practice is now conducting fewer parking lot visits since her office is allowing in-person appointments, but that they’re still an option for her patients.
Treating political adversaries
Some rural physicians have experienced strained relationships with patients for reasons other than technology – stark differences in opinion over the pandemic itself. Certain patients are following President Trump’s lead and questioning everything from the pandemic death toll to preventive measures recommended by scientists and medical experts, physicians interviewed by MDedge said.
Patients everywhere share these viewpoints, of course, but research and election results confirm that rural areas are more receptive to conservative viewpoints. In 2018, a Pew Research Center survey reported that rural and urban areas are “becoming more polarized politically,” and “rural areas tend to have a higher concentration of Republicans and Republican-leaning independents.” For example, 40% of rural respondents reported “very warm” or “somewhat warm” feelings toward Donald Trump, compared with just 19% in urban areas.
Dr. Shin has struggled to cope with patients who want to argue about pandemic safety precautions like wearing masks and seem to question whether systemic racism exists.
“We are seeing a lot more people who feel that this pandemic is not real, that it’s a political and not-true infection,” he said in an interview. “We’ve had patients who were angry at us because we made them wear masks, and some were demanding hydroxychloroquine and wanted to have an argument because we’re not going to prescribe it for them.”
In one situation, which he found especially disturbing, Dr. Shin had to leave the exam room because a patient wouldn’t stop challenging him regarding the pandemic. Things have gotten so bad that Dr. Shin has even questioned whether he wants to continue his long career in his small town because of local political attitudes such as opposition to mask-wearing and social distancing.
“Mr. Trump’s misinformation on this pandemic made my job much more difficult. As a minority, I feel less safe in my community than ever,” said Dr. Shin, who described himself as Asian American.
Despite these new stressors, Dr. Shin has experienced some joyful moments while practicing medicine in the pandemic.
He said a recent home visit to a patient who had been hospitalized for over 3 months and nearly died helped him put political disputes with his patients into perspective.
“He was discharged home but is bedbound. He had gangrene on his toes, and I could not fully examine him using video,” Dr. Shin recalled. “It was tricky to find the house, but a very large Trump sign was very helpful in locating it. It was a good visit: He was happy to see me, and I was happy to see that he was doing okay at home.”
“I need to remind myself that supporting Mr. Trump does not always mean that my patient supports Mr. Trump’s view on the pandemic and the race issues in our country,” Dr. Shin added.
The Washington-based internist said he also tells himself that, even if his patients refuse to follow his strong advice regarding pandemic precautions, it does not mean he has failed as a doctor.
“I need to continue to educate patients about the dangers of COVID infection but cannot be angry if they don’t choose to follow my recommendations,” he noted.
Dr. Fincher says her close connection with patients has allowed her to smooth over politically charged claims about the pandemic in the town of Thomson, Georgia, with a population 6,800.
“I have a sense that, even though we may differ in our understanding of some basic facts, they appreciate what I say since we have a long-term relationship built on trust,” she said. This kind of trust, Dr. Fincher suggested, may be more common than in urban areas where there’s a larger supply of physicians, and patients don’t see the same doctors for long periods of time.
“It’s more meaningful when it comes from me, rather than doctors who are [new to patients] every year when their employer changes their insurance,” she noted.
HM20 Virtual: Combating racism in medicine
HM20 Virtual session title
When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics
Presenter
Kimberly Manning, MD, FACP, FAAP
Session summary
Dr. Kimberly Manning, associate vice chair of diversity, equity, and inclusion at Emory University, Atlanta, masterfully discussed the dual pandemics of COVID-19 and racism that we are currently experiencing and tried to describe the unique perspective of Black Americans.
Though it is easy to see that COVID-19 is a pandemic, racism is not always seen in this way. Dr. Manning demonstrated that when a pandemic is defined as “that which occurs over a wide geographic area and affects a high proportion of the population,” racism is absolutely a pandemic. She gave a great analogy: when sticking your hand into a bowl of Lucky Charms cereal, you do not expect to always end up with marshmallows alone, yet repeatedly, we see that Black Americans have been disproportionately affected by COVID-19. We often hear that we are in unprecedented times but as far as racism is concerned, there is nothing new about this.
Dr. Manning discussed the life stories of her grandfather, her father, and even her own life’s milestones such as starting college, getting into medical school, finishing residency – all the way to becoming a full professor. She described how each of these instances, though marked by something beautiful, was also marked by something truly awful. Each time she had a reason to smile and laugh, there was something awful happening in the country simultaneously that showed us how racism was still present. Though this was one person’s story, all Black Americans, not just those working in health care, can recount similar stories, emotions, and feelings of grief.
Dr. Manning concluded by telling us how we can “Do the Work” to combat the pandemic of racism:
- Broaden your fund of knowledge: Read books, listen to podcasts, watch documentaries.
- Remember that people are grieving.
- Explore your implicit biases.
- Be a brave bystander.
- Avoid performative allyship.
Key takeaways
- Though the COVID-19 pandemic is unprecedented, the pandemic of racism is not.
- The story of COVID-19 is the story of social determinants of health.
- We all must “Do the Work” to combat everyday racism and be cognizant of what our Black colleagues are going through every day.
Dr. Doraiswamy is an assistant professor of medicine and pediatrics and a med-peds hospitalist at The Ohio State University and Nationwide Children’s Hospital, Columbus.
HM20 Virtual session title
When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics
Presenter
Kimberly Manning, MD, FACP, FAAP
Session summary
Dr. Kimberly Manning, associate vice chair of diversity, equity, and inclusion at Emory University, Atlanta, masterfully discussed the dual pandemics of COVID-19 and racism that we are currently experiencing and tried to describe the unique perspective of Black Americans.
Though it is easy to see that COVID-19 is a pandemic, racism is not always seen in this way. Dr. Manning demonstrated that when a pandemic is defined as “that which occurs over a wide geographic area and affects a high proportion of the population,” racism is absolutely a pandemic. She gave a great analogy: when sticking your hand into a bowl of Lucky Charms cereal, you do not expect to always end up with marshmallows alone, yet repeatedly, we see that Black Americans have been disproportionately affected by COVID-19. We often hear that we are in unprecedented times but as far as racism is concerned, there is nothing new about this.
Dr. Manning discussed the life stories of her grandfather, her father, and even her own life’s milestones such as starting college, getting into medical school, finishing residency – all the way to becoming a full professor. She described how each of these instances, though marked by something beautiful, was also marked by something truly awful. Each time she had a reason to smile and laugh, there was something awful happening in the country simultaneously that showed us how racism was still present. Though this was one person’s story, all Black Americans, not just those working in health care, can recount similar stories, emotions, and feelings of grief.
Dr. Manning concluded by telling us how we can “Do the Work” to combat the pandemic of racism:
- Broaden your fund of knowledge: Read books, listen to podcasts, watch documentaries.
- Remember that people are grieving.
- Explore your implicit biases.
- Be a brave bystander.
- Avoid performative allyship.
Key takeaways
- Though the COVID-19 pandemic is unprecedented, the pandemic of racism is not.
- The story of COVID-19 is the story of social determinants of health.
- We all must “Do the Work” to combat everyday racism and be cognizant of what our Black colleagues are going through every day.
Dr. Doraiswamy is an assistant professor of medicine and pediatrics and a med-peds hospitalist at The Ohio State University and Nationwide Children’s Hospital, Columbus.
HM20 Virtual session title
When Grief and Crises Intersect: Perspectives of a Black Physician in the Time of Two Pandemics
Presenter
Kimberly Manning, MD, FACP, FAAP
Session summary
Dr. Kimberly Manning, associate vice chair of diversity, equity, and inclusion at Emory University, Atlanta, masterfully discussed the dual pandemics of COVID-19 and racism that we are currently experiencing and tried to describe the unique perspective of Black Americans.
Though it is easy to see that COVID-19 is a pandemic, racism is not always seen in this way. Dr. Manning demonstrated that when a pandemic is defined as “that which occurs over a wide geographic area and affects a high proportion of the population,” racism is absolutely a pandemic. She gave a great analogy: when sticking your hand into a bowl of Lucky Charms cereal, you do not expect to always end up with marshmallows alone, yet repeatedly, we see that Black Americans have been disproportionately affected by COVID-19. We often hear that we are in unprecedented times but as far as racism is concerned, there is nothing new about this.
Dr. Manning discussed the life stories of her grandfather, her father, and even her own life’s milestones such as starting college, getting into medical school, finishing residency – all the way to becoming a full professor. She described how each of these instances, though marked by something beautiful, was also marked by something truly awful. Each time she had a reason to smile and laugh, there was something awful happening in the country simultaneously that showed us how racism was still present. Though this was one person’s story, all Black Americans, not just those working in health care, can recount similar stories, emotions, and feelings of grief.
Dr. Manning concluded by telling us how we can “Do the Work” to combat the pandemic of racism:
- Broaden your fund of knowledge: Read books, listen to podcasts, watch documentaries.
- Remember that people are grieving.
- Explore your implicit biases.
- Be a brave bystander.
- Avoid performative allyship.
Key takeaways
- Though the COVID-19 pandemic is unprecedented, the pandemic of racism is not.
- The story of COVID-19 is the story of social determinants of health.
- We all must “Do the Work” to combat everyday racism and be cognizant of what our Black colleagues are going through every day.
Dr. Doraiswamy is an assistant professor of medicine and pediatrics and a med-peds hospitalist at The Ohio State University and Nationwide Children’s Hospital, Columbus.
DAPA-CKD resets eGFR floor for safe SGLT2 inhibitor use
The dramatically positive safety and efficacy results from the DAPA-CKD trial, which showed that treatment with the sodium-glucose transporter 2 (SGLT2) inhibitor dapagliflozin significantly cut both chronic kidney disease progression and all-cause death in patients with or without type 2 diabetes, were also notable for broadening the population of patients eligible for this treatment to those in the upper range of stage 4 CKD.
Of the 4,304 CKD patients enrolled in DAPA-CKD, 624 (14%) had an estimated glomerular filtration rate (eGFR) of 25-29 mL/min per 1.73m2, an unprecedented population to receive a drug from the SGLT2 inhibitor class in a reported study. The results provided definitive evidence for efficacy and safety in this range of renal function, said Hiddo J.L. Heerspink, Ph.D., at the virtual annual meeting of the European Association for the Study of Diabetes.
Until now, the widely accepted lowest level for starting an SGLT2 inhibitor in routine practice has been an eGFR as low as 30 mL/min per 1.73 m2.
Using SGLT2 inhibitors when eGFR is as low as 25
“It’s time to reduce the eGFR level for initiating an SGLT2 inhibitor to as low as 25,” said Dr. Heerspink, a professor of clinical pharmacology at the University of Groningen (the Netherlands).
While conceding that this is primarily a decision to be made by guideline writers and regulatory bodies, he declared what he believed was established by the DAPA-CKD findings: “We’ve shown that dapagliflozin can be safely used in these patients. It is effective across the spectrum of kidney function.”
Other experts not associated with the study agreed.
The trial researchers were “brave” to enroll patients with eGFRs as low as 25 mL/min per 1.73 m2, and “we urgently need these agents in patients with an eGFR this low,” commented Chantal Mathieu, MD, an endocrinologist and professor of medicine at Catholic University in Leuven, Belgium, and designated discussant for the report. Overall, she called the findings “spectacular,” a “landmark trial,” and a “winner.”
The study also set an new, lower floor for the level of albuminuria that can be usefully treated with dapagliflozin (Farxiga) by enrolling patients with a urinary albumin-to-creatinine ratio as low as 200 mg/g; the previous lower limit had been 300 mg/g, noted Dr. Mathieu. The new findings pose challenges to guideline writers, regulators who approve drug labels, and payers to a quickly make changes that will bring dapagliflozin to a wider number of patients with CKD.
Once the full DAPA-CKD results are reported, “it will change practice, and push the eGFR needle down” to as low as 25. It will also lower the albuminuria threshold for using dapagliflozin or other drugs in the class, commented David Z.I. Cherney, MD, a nephrologist at the University of Toronto. “It’s just one study,” he admitted, but the consistent renal benefits seen across several studies involving all four drugs in the SGLT2 inhibitor class will help hasten this change in identifying treatable patients, as well as expand the drug class to patients with CKD but no type 2 diabetes (T2D).
“I don’t think we’ve ever had stronger evidence” for drugs that can benefit both heart and renal function, plus the drug class is “very safe, and really easy to start” and maintain in patients, Dr. Cherney said in an interview. “It’s wonderful for these patients that we now have something new for treatment,” a drug with a “very favorable benefit-to-risk ratio.”
Results show many dapagliflozin benefits
While this broadening of the range of patients proven to tolerate and benefit from an SGLT2 inhibitor was an important consequence of DAPA-CKD, the study’s primary finding – that dapagliflozin was as safe and effective for slowing CKD progression in patients regardless of whether they also had T2D – will have an even bigger impact on expanding the target patient population. Showing efficacy in patients with CKD but without a T2D etiology, the status of about a third of the enrolled 4,304 patients, makes this treatment an option for “millions” of additional patients worldwide, said Dr. Heerspink. “These are the most common patients nephrologists see.” A major challenge now will be to do a better job finding patients with CKD who could benefit from dapagliflozin.
DAPA-CKD enrolled CKD patients based primarily on prespecified albuminuria and eGFR levels at more than 300 centers in 34 countries, including the United States. Virtually all patients, 97%, were on the only treatment now available with proven efficacy for slowing CKD, either an ACE inhibitor or an angiotensin receptor blocker. The small number of patients not on one of these drugs was because of poor tolerance.
The study’s primary endpoint was the combined rate of cardiovascular death, renal death, end-stage renal disease, or a drop in eGFR of at least 50% from baseline. This occurred in 14.5% of patients who received placebo and in 9.2% of those who received dapagliflozin during a median follow-up of 2.4 years, a highly significant 39% relative risk reduction. Concurrently with the report at the virtual meeting the results also appeared online in the New England Journal of Medicine. This 5.3% cut in the absolute rate of the combined, primary adverse outcome converted into a number needed to treat of 19 to prevent 1 event during 2.4 years, a “much lower” number needed to treat than reported for renin-angiotensin system inhibitors in these types of patients, Dr. Heerspink said.
Notable positive secondary outcomes included a significant 31% relative cut (a 2% absolute decline) in all-cause mortality, “a major highlight” of the findings, Dr. Heerspink said. Dapagliflozin treatment also linked with a significant 29% relative cut in the incidence of cardiovascular death or hospitalization for heart failure.
“Cardiovascular disease is the most common cause of death in patients with CKD,” explained David C. Wheeler, MD, a coinvestigator on the study and professor of kidney medicine at University College London. “The heart and kidney are intertwined. This is about cardiorenal disease.”
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin. Dr. Heerspink has been a consultant to and received research funding from AstraZeneca. He has also received personal fees from Mundipharma and Novo Nordisk, and he has also served as consultant to several other companies with the honoraria being paid to his institution. Dr. Mathieu has had relationships with AstraZeneca and several other companies. Dr. Cherney has been a consultant to and has received research funding from AstraZeneca and several other companies. Dr. Wheeler has received personal fees from AstraZeneca and from several other companies.
SOURCE: Heerspink HJL et al. EASD 2020 and N Engl J Med. 2020 Sep 24. doi: 10.1056/NEJMoa2024816.
The dramatically positive safety and efficacy results from the DAPA-CKD trial, which showed that treatment with the sodium-glucose transporter 2 (SGLT2) inhibitor dapagliflozin significantly cut both chronic kidney disease progression and all-cause death in patients with or without type 2 diabetes, were also notable for broadening the population of patients eligible for this treatment to those in the upper range of stage 4 CKD.
Of the 4,304 CKD patients enrolled in DAPA-CKD, 624 (14%) had an estimated glomerular filtration rate (eGFR) of 25-29 mL/min per 1.73m2, an unprecedented population to receive a drug from the SGLT2 inhibitor class in a reported study. The results provided definitive evidence for efficacy and safety in this range of renal function, said Hiddo J.L. Heerspink, Ph.D., at the virtual annual meeting of the European Association for the Study of Diabetes.
Until now, the widely accepted lowest level for starting an SGLT2 inhibitor in routine practice has been an eGFR as low as 30 mL/min per 1.73 m2.
Using SGLT2 inhibitors when eGFR is as low as 25
“It’s time to reduce the eGFR level for initiating an SGLT2 inhibitor to as low as 25,” said Dr. Heerspink, a professor of clinical pharmacology at the University of Groningen (the Netherlands).
While conceding that this is primarily a decision to be made by guideline writers and regulatory bodies, he declared what he believed was established by the DAPA-CKD findings: “We’ve shown that dapagliflozin can be safely used in these patients. It is effective across the spectrum of kidney function.”
Other experts not associated with the study agreed.
The trial researchers were “brave” to enroll patients with eGFRs as low as 25 mL/min per 1.73 m2, and “we urgently need these agents in patients with an eGFR this low,” commented Chantal Mathieu, MD, an endocrinologist and professor of medicine at Catholic University in Leuven, Belgium, and designated discussant for the report. Overall, she called the findings “spectacular,” a “landmark trial,” and a “winner.”
The study also set an new, lower floor for the level of albuminuria that can be usefully treated with dapagliflozin (Farxiga) by enrolling patients with a urinary albumin-to-creatinine ratio as low as 200 mg/g; the previous lower limit had been 300 mg/g, noted Dr. Mathieu. The new findings pose challenges to guideline writers, regulators who approve drug labels, and payers to a quickly make changes that will bring dapagliflozin to a wider number of patients with CKD.
Once the full DAPA-CKD results are reported, “it will change practice, and push the eGFR needle down” to as low as 25. It will also lower the albuminuria threshold for using dapagliflozin or other drugs in the class, commented David Z.I. Cherney, MD, a nephrologist at the University of Toronto. “It’s just one study,” he admitted, but the consistent renal benefits seen across several studies involving all four drugs in the SGLT2 inhibitor class will help hasten this change in identifying treatable patients, as well as expand the drug class to patients with CKD but no type 2 diabetes (T2D).
“I don’t think we’ve ever had stronger evidence” for drugs that can benefit both heart and renal function, plus the drug class is “very safe, and really easy to start” and maintain in patients, Dr. Cherney said in an interview. “It’s wonderful for these patients that we now have something new for treatment,” a drug with a “very favorable benefit-to-risk ratio.”
Results show many dapagliflozin benefits
While this broadening of the range of patients proven to tolerate and benefit from an SGLT2 inhibitor was an important consequence of DAPA-CKD, the study’s primary finding – that dapagliflozin was as safe and effective for slowing CKD progression in patients regardless of whether they also had T2D – will have an even bigger impact on expanding the target patient population. Showing efficacy in patients with CKD but without a T2D etiology, the status of about a third of the enrolled 4,304 patients, makes this treatment an option for “millions” of additional patients worldwide, said Dr. Heerspink. “These are the most common patients nephrologists see.” A major challenge now will be to do a better job finding patients with CKD who could benefit from dapagliflozin.
DAPA-CKD enrolled CKD patients based primarily on prespecified albuminuria and eGFR levels at more than 300 centers in 34 countries, including the United States. Virtually all patients, 97%, were on the only treatment now available with proven efficacy for slowing CKD, either an ACE inhibitor or an angiotensin receptor blocker. The small number of patients not on one of these drugs was because of poor tolerance.
The study’s primary endpoint was the combined rate of cardiovascular death, renal death, end-stage renal disease, or a drop in eGFR of at least 50% from baseline. This occurred in 14.5% of patients who received placebo and in 9.2% of those who received dapagliflozin during a median follow-up of 2.4 years, a highly significant 39% relative risk reduction. Concurrently with the report at the virtual meeting the results also appeared online in the New England Journal of Medicine. This 5.3% cut in the absolute rate of the combined, primary adverse outcome converted into a number needed to treat of 19 to prevent 1 event during 2.4 years, a “much lower” number needed to treat than reported for renin-angiotensin system inhibitors in these types of patients, Dr. Heerspink said.
Notable positive secondary outcomes included a significant 31% relative cut (a 2% absolute decline) in all-cause mortality, “a major highlight” of the findings, Dr. Heerspink said. Dapagliflozin treatment also linked with a significant 29% relative cut in the incidence of cardiovascular death or hospitalization for heart failure.
“Cardiovascular disease is the most common cause of death in patients with CKD,” explained David C. Wheeler, MD, a coinvestigator on the study and professor of kidney medicine at University College London. “The heart and kidney are intertwined. This is about cardiorenal disease.”
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin. Dr. Heerspink has been a consultant to and received research funding from AstraZeneca. He has also received personal fees from Mundipharma and Novo Nordisk, and he has also served as consultant to several other companies with the honoraria being paid to his institution. Dr. Mathieu has had relationships with AstraZeneca and several other companies. Dr. Cherney has been a consultant to and has received research funding from AstraZeneca and several other companies. Dr. Wheeler has received personal fees from AstraZeneca and from several other companies.
SOURCE: Heerspink HJL et al. EASD 2020 and N Engl J Med. 2020 Sep 24. doi: 10.1056/NEJMoa2024816.
The dramatically positive safety and efficacy results from the DAPA-CKD trial, which showed that treatment with the sodium-glucose transporter 2 (SGLT2) inhibitor dapagliflozin significantly cut both chronic kidney disease progression and all-cause death in patients with or without type 2 diabetes, were also notable for broadening the population of patients eligible for this treatment to those in the upper range of stage 4 CKD.
Of the 4,304 CKD patients enrolled in DAPA-CKD, 624 (14%) had an estimated glomerular filtration rate (eGFR) of 25-29 mL/min per 1.73m2, an unprecedented population to receive a drug from the SGLT2 inhibitor class in a reported study. The results provided definitive evidence for efficacy and safety in this range of renal function, said Hiddo J.L. Heerspink, Ph.D., at the virtual annual meeting of the European Association for the Study of Diabetes.
Until now, the widely accepted lowest level for starting an SGLT2 inhibitor in routine practice has been an eGFR as low as 30 mL/min per 1.73 m2.
Using SGLT2 inhibitors when eGFR is as low as 25
“It’s time to reduce the eGFR level for initiating an SGLT2 inhibitor to as low as 25,” said Dr. Heerspink, a professor of clinical pharmacology at the University of Groningen (the Netherlands).
While conceding that this is primarily a decision to be made by guideline writers and regulatory bodies, he declared what he believed was established by the DAPA-CKD findings: “We’ve shown that dapagliflozin can be safely used in these patients. It is effective across the spectrum of kidney function.”
Other experts not associated with the study agreed.
The trial researchers were “brave” to enroll patients with eGFRs as low as 25 mL/min per 1.73 m2, and “we urgently need these agents in patients with an eGFR this low,” commented Chantal Mathieu, MD, an endocrinologist and professor of medicine at Catholic University in Leuven, Belgium, and designated discussant for the report. Overall, she called the findings “spectacular,” a “landmark trial,” and a “winner.”
The study also set an new, lower floor for the level of albuminuria that can be usefully treated with dapagliflozin (Farxiga) by enrolling patients with a urinary albumin-to-creatinine ratio as low as 200 mg/g; the previous lower limit had been 300 mg/g, noted Dr. Mathieu. The new findings pose challenges to guideline writers, regulators who approve drug labels, and payers to a quickly make changes that will bring dapagliflozin to a wider number of patients with CKD.
Once the full DAPA-CKD results are reported, “it will change practice, and push the eGFR needle down” to as low as 25. It will also lower the albuminuria threshold for using dapagliflozin or other drugs in the class, commented David Z.I. Cherney, MD, a nephrologist at the University of Toronto. “It’s just one study,” he admitted, but the consistent renal benefits seen across several studies involving all four drugs in the SGLT2 inhibitor class will help hasten this change in identifying treatable patients, as well as expand the drug class to patients with CKD but no type 2 diabetes (T2D).
“I don’t think we’ve ever had stronger evidence” for drugs that can benefit both heart and renal function, plus the drug class is “very safe, and really easy to start” and maintain in patients, Dr. Cherney said in an interview. “It’s wonderful for these patients that we now have something new for treatment,” a drug with a “very favorable benefit-to-risk ratio.”
Results show many dapagliflozin benefits
While this broadening of the range of patients proven to tolerate and benefit from an SGLT2 inhibitor was an important consequence of DAPA-CKD, the study’s primary finding – that dapagliflozin was as safe and effective for slowing CKD progression in patients regardless of whether they also had T2D – will have an even bigger impact on expanding the target patient population. Showing efficacy in patients with CKD but without a T2D etiology, the status of about a third of the enrolled 4,304 patients, makes this treatment an option for “millions” of additional patients worldwide, said Dr. Heerspink. “These are the most common patients nephrologists see.” A major challenge now will be to do a better job finding patients with CKD who could benefit from dapagliflozin.
DAPA-CKD enrolled CKD patients based primarily on prespecified albuminuria and eGFR levels at more than 300 centers in 34 countries, including the United States. Virtually all patients, 97%, were on the only treatment now available with proven efficacy for slowing CKD, either an ACE inhibitor or an angiotensin receptor blocker. The small number of patients not on one of these drugs was because of poor tolerance.
The study’s primary endpoint was the combined rate of cardiovascular death, renal death, end-stage renal disease, or a drop in eGFR of at least 50% from baseline. This occurred in 14.5% of patients who received placebo and in 9.2% of those who received dapagliflozin during a median follow-up of 2.4 years, a highly significant 39% relative risk reduction. Concurrently with the report at the virtual meeting the results also appeared online in the New England Journal of Medicine. This 5.3% cut in the absolute rate of the combined, primary adverse outcome converted into a number needed to treat of 19 to prevent 1 event during 2.4 years, a “much lower” number needed to treat than reported for renin-angiotensin system inhibitors in these types of patients, Dr. Heerspink said.
Notable positive secondary outcomes included a significant 31% relative cut (a 2% absolute decline) in all-cause mortality, “a major highlight” of the findings, Dr. Heerspink said. Dapagliflozin treatment also linked with a significant 29% relative cut in the incidence of cardiovascular death or hospitalization for heart failure.
“Cardiovascular disease is the most common cause of death in patients with CKD,” explained David C. Wheeler, MD, a coinvestigator on the study and professor of kidney medicine at University College London. “The heart and kidney are intertwined. This is about cardiorenal disease.”
DAPA-CKD was funded by AstraZeneca, the company that markets dapagliflozin. Dr. Heerspink has been a consultant to and received research funding from AstraZeneca. He has also received personal fees from Mundipharma and Novo Nordisk, and he has also served as consultant to several other companies with the honoraria being paid to his institution. Dr. Mathieu has had relationships with AstraZeneca and several other companies. Dr. Cherney has been a consultant to and has received research funding from AstraZeneca and several other companies. Dr. Wheeler has received personal fees from AstraZeneca and from several other companies.
SOURCE: Heerspink HJL et al. EASD 2020 and N Engl J Med. 2020 Sep 24. doi: 10.1056/NEJMoa2024816.
FROM EASD 2020












