User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Pertussis vaccination of infants reduces death, hospitalization risk
Receiving the first dose of the pertussis vaccine significantly reduces the risk of death, hospitalization, and pneumonia among infants who contract the illness, a recent study found.
“Health professionals should ensure on-time first-dose pertussis vaccination as early as 6 weeks of age during pertussis outbreaks and provide early recommended antibiotic treatment,” said Dr. Tejpratap S.P. Tiwari and colleagues at the Centers for Disease Control and Prevention, Atlanta. “Infants who are age-ineligible for vaccination will benefit from strengthening strategies that provide immunity to newborns and prevent exposure to [Bordetella] pertussis,” they wrote (Pediatrics 2015 (doi:10.1542/peds.2014-2291).
Dr. Tiwari’s team reviewed all cases of pertussis in infants under age 1 year that had been reported to the National Notifiable Diseases Surveillance System (NNDSS) and the Centers for Disease Control and Prevention between 1991 and 2008. Among 45,404 reported cases, 258 deaths (0.57%) occurred.
All of the children who died had contracted pertussis before age 34 weeks, and 64% of the deaths occurred in children younger than 6 weeks. Overall, 90% of nonfatal cases occurred in infants younger than 34 weeks. The older children were, the less likely they were to die from the disease: the case fatality rate dropped from 2.36% at age 1 week to 0.28% at age 12 weeks. No significant differences in sex were observed.
Infants at least 42 days old who had received at least one dose of DTaP were 72% less likely to die from the illness after adjusting for onset age, race, and antibiotic treatment (adjusted odds ratio, 0.28). They were also 31% less likely to be hospitalized and 20% less likely to develop pneumonia (aOR, 0.69 and 0.80, respectively). Babies who received the recommended antibiotics were 72% less likely to die than those who didn’t receive the drugs (aOR, 0.28).
Hispanic infants with pertussis were more than twice as likely to die than were non-Hispanic infants with the illness, with a case fatality rate of 1.26% for Hispanic infants and 0.75% for non-Hispanic infants (aOR, 2.28, compared with white infants). American Indian/Alaska Native infants had a more than five times greater odds of death and a case fatality rate of 1.85% (aOR, 5.15).
The researchers calculated that on-time vaccination could have prevented 72% of the deaths occurring after 8 weeks, resulting in a 16% reduced fatality rate.
The research was funded by the CDC. The authors reported no relevant financial disclosures.
Receiving the first dose of the pertussis vaccine significantly reduces the risk of death, hospitalization, and pneumonia among infants who contract the illness, a recent study found.
“Health professionals should ensure on-time first-dose pertussis vaccination as early as 6 weeks of age during pertussis outbreaks and provide early recommended antibiotic treatment,” said Dr. Tejpratap S.P. Tiwari and colleagues at the Centers for Disease Control and Prevention, Atlanta. “Infants who are age-ineligible for vaccination will benefit from strengthening strategies that provide immunity to newborns and prevent exposure to [Bordetella] pertussis,” they wrote (Pediatrics 2015 (doi:10.1542/peds.2014-2291).
Dr. Tiwari’s team reviewed all cases of pertussis in infants under age 1 year that had been reported to the National Notifiable Diseases Surveillance System (NNDSS) and the Centers for Disease Control and Prevention between 1991 and 2008. Among 45,404 reported cases, 258 deaths (0.57%) occurred.
All of the children who died had contracted pertussis before age 34 weeks, and 64% of the deaths occurred in children younger than 6 weeks. Overall, 90% of nonfatal cases occurred in infants younger than 34 weeks. The older children were, the less likely they were to die from the disease: the case fatality rate dropped from 2.36% at age 1 week to 0.28% at age 12 weeks. No significant differences in sex were observed.
Infants at least 42 days old who had received at least one dose of DTaP were 72% less likely to die from the illness after adjusting for onset age, race, and antibiotic treatment (adjusted odds ratio, 0.28). They were also 31% less likely to be hospitalized and 20% less likely to develop pneumonia (aOR, 0.69 and 0.80, respectively). Babies who received the recommended antibiotics were 72% less likely to die than those who didn’t receive the drugs (aOR, 0.28).
Hispanic infants with pertussis were more than twice as likely to die than were non-Hispanic infants with the illness, with a case fatality rate of 1.26% for Hispanic infants and 0.75% for non-Hispanic infants (aOR, 2.28, compared with white infants). American Indian/Alaska Native infants had a more than five times greater odds of death and a case fatality rate of 1.85% (aOR, 5.15).
The researchers calculated that on-time vaccination could have prevented 72% of the deaths occurring after 8 weeks, resulting in a 16% reduced fatality rate.
The research was funded by the CDC. The authors reported no relevant financial disclosures.
Receiving the first dose of the pertussis vaccine significantly reduces the risk of death, hospitalization, and pneumonia among infants who contract the illness, a recent study found.
“Health professionals should ensure on-time first-dose pertussis vaccination as early as 6 weeks of age during pertussis outbreaks and provide early recommended antibiotic treatment,” said Dr. Tejpratap S.P. Tiwari and colleagues at the Centers for Disease Control and Prevention, Atlanta. “Infants who are age-ineligible for vaccination will benefit from strengthening strategies that provide immunity to newborns and prevent exposure to [Bordetella] pertussis,” they wrote (Pediatrics 2015 (doi:10.1542/peds.2014-2291).
Dr. Tiwari’s team reviewed all cases of pertussis in infants under age 1 year that had been reported to the National Notifiable Diseases Surveillance System (NNDSS) and the Centers for Disease Control and Prevention between 1991 and 2008. Among 45,404 reported cases, 258 deaths (0.57%) occurred.
All of the children who died had contracted pertussis before age 34 weeks, and 64% of the deaths occurred in children younger than 6 weeks. Overall, 90% of nonfatal cases occurred in infants younger than 34 weeks. The older children were, the less likely they were to die from the disease: the case fatality rate dropped from 2.36% at age 1 week to 0.28% at age 12 weeks. No significant differences in sex were observed.
Infants at least 42 days old who had received at least one dose of DTaP were 72% less likely to die from the illness after adjusting for onset age, race, and antibiotic treatment (adjusted odds ratio, 0.28). They were also 31% less likely to be hospitalized and 20% less likely to develop pneumonia (aOR, 0.69 and 0.80, respectively). Babies who received the recommended antibiotics were 72% less likely to die than those who didn’t receive the drugs (aOR, 0.28).
Hispanic infants with pertussis were more than twice as likely to die than were non-Hispanic infants with the illness, with a case fatality rate of 1.26% for Hispanic infants and 0.75% for non-Hispanic infants (aOR, 2.28, compared with white infants). American Indian/Alaska Native infants had a more than five times greater odds of death and a case fatality rate of 1.85% (aOR, 5.15).
The researchers calculated that on-time vaccination could have prevented 72% of the deaths occurring after 8 weeks, resulting in a 16% reduced fatality rate.
The research was funded by the CDC. The authors reported no relevant financial disclosures.
FROM PEDIATRICS
Key clinical point: Pertussis vaccination reduces death, hospitalization, and pneumonia in infected infants.
Major finding: Vaccinated infants with pertussis had 72% lower odds of dying, 31% lower odds of hospitalization, and 20% lower odds of pneumonia than unvaccinated infants with pertussis.
Data source: Retrospective analysis of all 45,404 confirmed or probable cases of pertussis in infants under age 1 year reported to the National Notifiable Diseases Surveillance System and the Centers for Disease Control and Prevention between 1991 and 2008.
Disclosures: The research was funded by the CDC. The authors reported no relevant financial disclosures.
Teens’ Tdap booster dose begins to wane after 2 years
A sixth dose of Tdap is initially effective in preventing pertussis in teens, but its effectiveness declines by half within 4 years after the booster, a recent study found.
“This waning is likely contributing to the increase in pertussis among adolescents,” reported Dr. Anna M. Acosta of the Centers for Disease Control and Prevention, Atlanta, and her associates (Pediatrics 2015 [doi: 10.1542/peds.2014-3358]).
“Advances in our understanding of the immunology and bacteriology of Bordetella pertussis are essential to optimize future prevention and control measures,” they wrote. “However, novel pertussis vaccines that effectively limit infection and transmission are also likely needed to reduce the burden of pertussis disease in the United States.”
The researchers matched three controls by birth year and primary provider practice (total 2,322 controls) to each of 836 cases of pertussis in seven counties of Washington during the 2012 pertussis epidemic. Cases were more likely than were controls to be non-Hispanic and white, but there was a lack of race/ethnicity data.
Receipt of the five childhood series doses was similar among cases (74%) and controls (75%), but a smaller proportion of both (60% cases, 58% controls) were on schedule. Among more than 84% of participants who received Tdap between ages 11 and 12, 81% of the cases and 90% of the controls received the sixth dose.
Among the 450 cases and 1,246 controls who received all acellular vaccines for the primary series, Tdap effectiveness was 63.9% overall. Stratified by time since Tdap vaccination, however, it was 73.1% within 12 months, 54.9% within 12-23 months, and 34.2% within 24-47 months.
Those born from 1999 to 2000 were presumed to have received the DTaP in infancy, and those born from 1993 to 1997 were presumed to have received a mixture of the acellular and whole-cell (DTwP) vaccines. Among those with vaccine lot numbers recorded, all vaccines administered after 1998 were acellular. A direct comparison of Tdap effectiveness between those vaccinated with the acellular primary series and those vaccinated with a mixed primary series was not possible because of time differences in vaccination between the two groups.
The research did not receive external funding, and the authors reported no relevant financial disclosures.
In the study by Acosta et al., the duration of Tdap effectiveness is disappointing, particularly because case-control studies tend to inflate efficacy. The resurgence of pertussis is often attributed to the switch from whole-cell pertussis vaccines to acellular products. However, the increase in reported pertussis began 14 years before the universal use of diphtheria-tetanus-acellular pertussis (DTaP) vaccines in childhood commenced. The two greatest contributors to the resurgence of pertussis are greater awareness and more sensitive diagnosis (routine use of polymerase chain reaction).
In the pre-DTaP and pre-Tdap eras, the pertussis attack rate in nonepidemic periods in largely whole-cell pertussis vaccine–primed adolescents and adults was 370 to 500 per 100,000 per year. These rates are underestimates because of clear evidence of “observer bias” in both studies. In this present Washington State study, the attack rate during the epidemic was only 182.3 per 100,000 for the one-half-year study period.
Factors that I think are most important relating to DTaP vaccine failure are as follows: decay in antibody over time; a T helper (Th) 1/Th2 versus a Th1, Th17 cellular response; incomplete antigen package; incorrect balance of antigens in the vaccine; linked-epitope suppression; and the occurrence of pertactin-deficient B. pertussis strains. Some, but not all, of these factors also may relate to Tdap failure over time.
Although adequate data are not available, it can be assumed that adolescents and adults who were primed in infancy by infection or DTP will have a Th1, Th17 response to Tdap. In contrast, those who were primed by DTaP will have a Th1/Th2 response. In line with the results of these two recent Tdap effectiveness studies, we should examine our present Tdap immunization recommendations. It is my opinion that we should continue with our present Tdap schedules. Of most importance is to see that all pregnant women receive Tdap with each pregnancy. This alone can prevent virtually all pertussis deaths in young infants.
James D. Cherry, M.D., is professor of pediatrics at the University of California, Los Angeles. These comments are excerpted from an editorial (Pediatrics 2015 [doi:10.1542/peds.2014-4118]) accompanying Dr. Acosta’s study. Dr. Cherry is a member of the Sanofi Pasteur speakers bureau and of the Global Pertussis Initiative, supported by Sanofi Pastuer. He receives royalties from Elsevier for “Feigin and Cherry’s Textbook of Pediatric Infectious Diseases.”
In the study by Acosta et al., the duration of Tdap effectiveness is disappointing, particularly because case-control studies tend to inflate efficacy. The resurgence of pertussis is often attributed to the switch from whole-cell pertussis vaccines to acellular products. However, the increase in reported pertussis began 14 years before the universal use of diphtheria-tetanus-acellular pertussis (DTaP) vaccines in childhood commenced. The two greatest contributors to the resurgence of pertussis are greater awareness and more sensitive diagnosis (routine use of polymerase chain reaction).
In the pre-DTaP and pre-Tdap eras, the pertussis attack rate in nonepidemic periods in largely whole-cell pertussis vaccine–primed adolescents and adults was 370 to 500 per 100,000 per year. These rates are underestimates because of clear evidence of “observer bias” in both studies. In this present Washington State study, the attack rate during the epidemic was only 182.3 per 100,000 for the one-half-year study period.
Factors that I think are most important relating to DTaP vaccine failure are as follows: decay in antibody over time; a T helper (Th) 1/Th2 versus a Th1, Th17 cellular response; incomplete antigen package; incorrect balance of antigens in the vaccine; linked-epitope suppression; and the occurrence of pertactin-deficient B. pertussis strains. Some, but not all, of these factors also may relate to Tdap failure over time.
Although adequate data are not available, it can be assumed that adolescents and adults who were primed in infancy by infection or DTP will have a Th1, Th17 response to Tdap. In contrast, those who were primed by DTaP will have a Th1/Th2 response. In line with the results of these two recent Tdap effectiveness studies, we should examine our present Tdap immunization recommendations. It is my opinion that we should continue with our present Tdap schedules. Of most importance is to see that all pregnant women receive Tdap with each pregnancy. This alone can prevent virtually all pertussis deaths in young infants.
James D. Cherry, M.D., is professor of pediatrics at the University of California, Los Angeles. These comments are excerpted from an editorial (Pediatrics 2015 [doi:10.1542/peds.2014-4118]) accompanying Dr. Acosta’s study. Dr. Cherry is a member of the Sanofi Pasteur speakers bureau and of the Global Pertussis Initiative, supported by Sanofi Pastuer. He receives royalties from Elsevier for “Feigin and Cherry’s Textbook of Pediatric Infectious Diseases.”
In the study by Acosta et al., the duration of Tdap effectiveness is disappointing, particularly because case-control studies tend to inflate efficacy. The resurgence of pertussis is often attributed to the switch from whole-cell pertussis vaccines to acellular products. However, the increase in reported pertussis began 14 years before the universal use of diphtheria-tetanus-acellular pertussis (DTaP) vaccines in childhood commenced. The two greatest contributors to the resurgence of pertussis are greater awareness and more sensitive diagnosis (routine use of polymerase chain reaction).
In the pre-DTaP and pre-Tdap eras, the pertussis attack rate in nonepidemic periods in largely whole-cell pertussis vaccine–primed adolescents and adults was 370 to 500 per 100,000 per year. These rates are underestimates because of clear evidence of “observer bias” in both studies. In this present Washington State study, the attack rate during the epidemic was only 182.3 per 100,000 for the one-half-year study period.
Factors that I think are most important relating to DTaP vaccine failure are as follows: decay in antibody over time; a T helper (Th) 1/Th2 versus a Th1, Th17 cellular response; incomplete antigen package; incorrect balance of antigens in the vaccine; linked-epitope suppression; and the occurrence of pertactin-deficient B. pertussis strains. Some, but not all, of these factors also may relate to Tdap failure over time.
Although adequate data are not available, it can be assumed that adolescents and adults who were primed in infancy by infection or DTP will have a Th1, Th17 response to Tdap. In contrast, those who were primed by DTaP will have a Th1/Th2 response. In line with the results of these two recent Tdap effectiveness studies, we should examine our present Tdap immunization recommendations. It is my opinion that we should continue with our present Tdap schedules. Of most importance is to see that all pregnant women receive Tdap with each pregnancy. This alone can prevent virtually all pertussis deaths in young infants.
James D. Cherry, M.D., is professor of pediatrics at the University of California, Los Angeles. These comments are excerpted from an editorial (Pediatrics 2015 [doi:10.1542/peds.2014-4118]) accompanying Dr. Acosta’s study. Dr. Cherry is a member of the Sanofi Pasteur speakers bureau and of the Global Pertussis Initiative, supported by Sanofi Pastuer. He receives royalties from Elsevier for “Feigin and Cherry’s Textbook of Pediatric Infectious Diseases.”
A sixth dose of Tdap is initially effective in preventing pertussis in teens, but its effectiveness declines by half within 4 years after the booster, a recent study found.
“This waning is likely contributing to the increase in pertussis among adolescents,” reported Dr. Anna M. Acosta of the Centers for Disease Control and Prevention, Atlanta, and her associates (Pediatrics 2015 [doi: 10.1542/peds.2014-3358]).
“Advances in our understanding of the immunology and bacteriology of Bordetella pertussis are essential to optimize future prevention and control measures,” they wrote. “However, novel pertussis vaccines that effectively limit infection and transmission are also likely needed to reduce the burden of pertussis disease in the United States.”
The researchers matched three controls by birth year and primary provider practice (total 2,322 controls) to each of 836 cases of pertussis in seven counties of Washington during the 2012 pertussis epidemic. Cases were more likely than were controls to be non-Hispanic and white, but there was a lack of race/ethnicity data.
Receipt of the five childhood series doses was similar among cases (74%) and controls (75%), but a smaller proportion of both (60% cases, 58% controls) were on schedule. Among more than 84% of participants who received Tdap between ages 11 and 12, 81% of the cases and 90% of the controls received the sixth dose.
Among the 450 cases and 1,246 controls who received all acellular vaccines for the primary series, Tdap effectiveness was 63.9% overall. Stratified by time since Tdap vaccination, however, it was 73.1% within 12 months, 54.9% within 12-23 months, and 34.2% within 24-47 months.
Those born from 1999 to 2000 were presumed to have received the DTaP in infancy, and those born from 1993 to 1997 were presumed to have received a mixture of the acellular and whole-cell (DTwP) vaccines. Among those with vaccine lot numbers recorded, all vaccines administered after 1998 were acellular. A direct comparison of Tdap effectiveness between those vaccinated with the acellular primary series and those vaccinated with a mixed primary series was not possible because of time differences in vaccination between the two groups.
The research did not receive external funding, and the authors reported no relevant financial disclosures.
A sixth dose of Tdap is initially effective in preventing pertussis in teens, but its effectiveness declines by half within 4 years after the booster, a recent study found.
“This waning is likely contributing to the increase in pertussis among adolescents,” reported Dr. Anna M. Acosta of the Centers for Disease Control and Prevention, Atlanta, and her associates (Pediatrics 2015 [doi: 10.1542/peds.2014-3358]).
“Advances in our understanding of the immunology and bacteriology of Bordetella pertussis are essential to optimize future prevention and control measures,” they wrote. “However, novel pertussis vaccines that effectively limit infection and transmission are also likely needed to reduce the burden of pertussis disease in the United States.”
The researchers matched three controls by birth year and primary provider practice (total 2,322 controls) to each of 836 cases of pertussis in seven counties of Washington during the 2012 pertussis epidemic. Cases were more likely than were controls to be non-Hispanic and white, but there was a lack of race/ethnicity data.
Receipt of the five childhood series doses was similar among cases (74%) and controls (75%), but a smaller proportion of both (60% cases, 58% controls) were on schedule. Among more than 84% of participants who received Tdap between ages 11 and 12, 81% of the cases and 90% of the controls received the sixth dose.
Among the 450 cases and 1,246 controls who received all acellular vaccines for the primary series, Tdap effectiveness was 63.9% overall. Stratified by time since Tdap vaccination, however, it was 73.1% within 12 months, 54.9% within 12-23 months, and 34.2% within 24-47 months.
Those born from 1999 to 2000 were presumed to have received the DTaP in infancy, and those born from 1993 to 1997 were presumed to have received a mixture of the acellular and whole-cell (DTwP) vaccines. Among those with vaccine lot numbers recorded, all vaccines administered after 1998 were acellular. A direct comparison of Tdap effectiveness between those vaccinated with the acellular primary series and those vaccinated with a mixed primary series was not possible because of time differences in vaccination between the two groups.
The research did not receive external funding, and the authors reported no relevant financial disclosures.
FROM PEDIATRICS
Key clinical point: Tdap effectiveness for pertussis prevention wanes 2 years after the sixth dose in teens.
Major finding: Tdap booster effectiveness drops from 73% to 34% within 4 years of the sixth dose in teens.
Data source: A case-control study of 3,158 teens in seven counties during the 2012 pertussis epidemic in Washington State.
Disclosures: The research did not receive external funding, and the authors reported no relevant financial disclosures.
Baseline CT scans predict lung fibrosis in SSc
Patients with newly diagnosed systemic sclerosis should have a high-resolution lung CT and pulmonary function tests, because when assessed together, their findings identify patients at high risk of interstitial lung disease, researchers say.
In a study of 305 patients with systemic sclerosis (SSc), a baseline high-resolution lung CT showing no fibrosis was highly predictive of a fibrosis free follow-up scan at 3 years, reported the researchers from Oslo (Norway) University Hospital. SSc associated interstitial lung–disease (SSc-ILD) typically has an insidious onset with subtle clinical symptoms, accoording to the authors in background information to the paper published in Arthritis & Rheumatology (Arthritis & Rheumatology 2015; [doi:10.1002/art.39166]).
This may explain why SSc-ILD is often diagnosed at an advanced stage, when extensive lung fibrosis is already present. “Better, and more targeted, strategies for SSc-ILD identification and risk stratification early in the disease course are therefore warranted,” they wrote.
In order to assess serial lung fibrosis measurements and paired pulmonary function tests (PFTs) as outcome prediction tools, the researchers prospectively analyzed both modalities at baseline and at an average of 3 years follow-up in 305 patients with SSc. The extent of fibrosis was scored on 10 sections from every high-resolution CT (HRCT) and expressed as a percentage of total lung volumes.
The researchers identified three groups of patients: More than 20% lung fibrosis (n=40), between 1%- 20% fibrosis (n=157), and no fibrosis (n=108). Results showed that all 108 patients who had no lung fibrosis at baseline remained free of fibrosis at a 3-year follow-up scan. These patients were predominantly female (88%) had limited (lc) SSc (84%), and were positive for anticentromere antibodies (ACA) (70%). They also had a high baseline decline in diffusing lung capacity for carbon monoxide (DLCO) that declined by 8.2%, the same degree as patients with lung fibrosis.
“This finding emphasizes that the mechanics behind SSc related DCLO changes probably are multifactorial and may involve pathology in the vasculature,” the study authors wrote. This was underscored by the observation that pulmonary hypertension (PH) was present in all groups of patients and supported the notion that PH screening should be conducted independently of fibrosis screening, the researchers noted.
For patients in the 1%-20% group, 146 were the same at follow-up, whereas the remaining patients progressed to more than 20% fibrosis. These 11 patients were characterized by significantly shorter average disease duration at baseline (1.3 years) compared with the other groups.
The 40 patients with more than 20% fibrosis at baseline had a higher annual fibrosis progression rate (aFPR), declining PFT values, and development of pulmonary hypertension (PH). Most of this group had diffuse SSc (55%), were positive for anti-topoisomerase antiboidies (ATA) (48%), and had a higher frequency of PH (28%), the researchers said. The rate of annual fibrosis progression differed across all groups and correlated with total FVC decline.
Surprisingly, neither baseline fibrosis nor annual fibrosis progression significantly predicted mortality. This finding may be partly due to survival bias in the cohort and the statistical power of the study, the researchers said. “The results indicate that a baseline examination in newly diagnosed SSc patients should include lung HRCT and PFTs,” the researchers concluded. Patients with low ILD risk should not undergo serial HCRT examination but probably need serial PFTs as an adjunct PH detection tool, they said.
Patients with newly diagnosed systemic sclerosis should have a high-resolution lung CT and pulmonary function tests, because when assessed together, their findings identify patients at high risk of interstitial lung disease, researchers say.
In a study of 305 patients with systemic sclerosis (SSc), a baseline high-resolution lung CT showing no fibrosis was highly predictive of a fibrosis free follow-up scan at 3 years, reported the researchers from Oslo (Norway) University Hospital. SSc associated interstitial lung–disease (SSc-ILD) typically has an insidious onset with subtle clinical symptoms, accoording to the authors in background information to the paper published in Arthritis & Rheumatology (Arthritis & Rheumatology 2015; [doi:10.1002/art.39166]).
This may explain why SSc-ILD is often diagnosed at an advanced stage, when extensive lung fibrosis is already present. “Better, and more targeted, strategies for SSc-ILD identification and risk stratification early in the disease course are therefore warranted,” they wrote.
In order to assess serial lung fibrosis measurements and paired pulmonary function tests (PFTs) as outcome prediction tools, the researchers prospectively analyzed both modalities at baseline and at an average of 3 years follow-up in 305 patients with SSc. The extent of fibrosis was scored on 10 sections from every high-resolution CT (HRCT) and expressed as a percentage of total lung volumes.
The researchers identified three groups of patients: More than 20% lung fibrosis (n=40), between 1%- 20% fibrosis (n=157), and no fibrosis (n=108). Results showed that all 108 patients who had no lung fibrosis at baseline remained free of fibrosis at a 3-year follow-up scan. These patients were predominantly female (88%) had limited (lc) SSc (84%), and were positive for anticentromere antibodies (ACA) (70%). They also had a high baseline decline in diffusing lung capacity for carbon monoxide (DLCO) that declined by 8.2%, the same degree as patients with lung fibrosis.
“This finding emphasizes that the mechanics behind SSc related DCLO changes probably are multifactorial and may involve pathology in the vasculature,” the study authors wrote. This was underscored by the observation that pulmonary hypertension (PH) was present in all groups of patients and supported the notion that PH screening should be conducted independently of fibrosis screening, the researchers noted.
For patients in the 1%-20% group, 146 were the same at follow-up, whereas the remaining patients progressed to more than 20% fibrosis. These 11 patients were characterized by significantly shorter average disease duration at baseline (1.3 years) compared with the other groups.
The 40 patients with more than 20% fibrosis at baseline had a higher annual fibrosis progression rate (aFPR), declining PFT values, and development of pulmonary hypertension (PH). Most of this group had diffuse SSc (55%), were positive for anti-topoisomerase antiboidies (ATA) (48%), and had a higher frequency of PH (28%), the researchers said. The rate of annual fibrosis progression differed across all groups and correlated with total FVC decline.
Surprisingly, neither baseline fibrosis nor annual fibrosis progression significantly predicted mortality. This finding may be partly due to survival bias in the cohort and the statistical power of the study, the researchers said. “The results indicate that a baseline examination in newly diagnosed SSc patients should include lung HRCT and PFTs,” the researchers concluded. Patients with low ILD risk should not undergo serial HCRT examination but probably need serial PFTs as an adjunct PH detection tool, they said.
Patients with newly diagnosed systemic sclerosis should have a high-resolution lung CT and pulmonary function tests, because when assessed together, their findings identify patients at high risk of interstitial lung disease, researchers say.
In a study of 305 patients with systemic sclerosis (SSc), a baseline high-resolution lung CT showing no fibrosis was highly predictive of a fibrosis free follow-up scan at 3 years, reported the researchers from Oslo (Norway) University Hospital. SSc associated interstitial lung–disease (SSc-ILD) typically has an insidious onset with subtle clinical symptoms, accoording to the authors in background information to the paper published in Arthritis & Rheumatology (Arthritis & Rheumatology 2015; [doi:10.1002/art.39166]).
This may explain why SSc-ILD is often diagnosed at an advanced stage, when extensive lung fibrosis is already present. “Better, and more targeted, strategies for SSc-ILD identification and risk stratification early in the disease course are therefore warranted,” they wrote.
In order to assess serial lung fibrosis measurements and paired pulmonary function tests (PFTs) as outcome prediction tools, the researchers prospectively analyzed both modalities at baseline and at an average of 3 years follow-up in 305 patients with SSc. The extent of fibrosis was scored on 10 sections from every high-resolution CT (HRCT) and expressed as a percentage of total lung volumes.
The researchers identified three groups of patients: More than 20% lung fibrosis (n=40), between 1%- 20% fibrosis (n=157), and no fibrosis (n=108). Results showed that all 108 patients who had no lung fibrosis at baseline remained free of fibrosis at a 3-year follow-up scan. These patients were predominantly female (88%) had limited (lc) SSc (84%), and were positive for anticentromere antibodies (ACA) (70%). They also had a high baseline decline in diffusing lung capacity for carbon monoxide (DLCO) that declined by 8.2%, the same degree as patients with lung fibrosis.
“This finding emphasizes that the mechanics behind SSc related DCLO changes probably are multifactorial and may involve pathology in the vasculature,” the study authors wrote. This was underscored by the observation that pulmonary hypertension (PH) was present in all groups of patients and supported the notion that PH screening should be conducted independently of fibrosis screening, the researchers noted.
For patients in the 1%-20% group, 146 were the same at follow-up, whereas the remaining patients progressed to more than 20% fibrosis. These 11 patients were characterized by significantly shorter average disease duration at baseline (1.3 years) compared with the other groups.
The 40 patients with more than 20% fibrosis at baseline had a higher annual fibrosis progression rate (aFPR), declining PFT values, and development of pulmonary hypertension (PH). Most of this group had diffuse SSc (55%), were positive for anti-topoisomerase antiboidies (ATA) (48%), and had a higher frequency of PH (28%), the researchers said. The rate of annual fibrosis progression differed across all groups and correlated with total FVC decline.
Surprisingly, neither baseline fibrosis nor annual fibrosis progression significantly predicted mortality. This finding may be partly due to survival bias in the cohort and the statistical power of the study, the researchers said. “The results indicate that a baseline examination in newly diagnosed SSc patients should include lung HRCT and PFTs,” the researchers concluded. Patients with low ILD risk should not undergo serial HCRT examination but probably need serial PFTs as an adjunct PH detection tool, they said.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Patients with newly diagnosed SSc should have baseline lung CT and pulmonary function tests
Major finding: Baseline high-resolution CT scans were highly predictive of fibrosis development and progression.
Data source: A prospective study of 305 consecutive SSc patients attending Oslo University Hospital.
Disclosures: No conflicts of interest were reported.
Pleuritic chest pain and globus pharyngeus
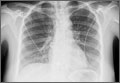
A 22-year-old woman with a history of attention-deficit/hyperactivity disorder and childhood asthma came to the emergency department (ED) for treatment of a cramping, substernal, pleuritic chest pain she’d had for a week and the feeling of a “lump in her throat” that made it difficult and painful for her to swallow. The patient’s vital signs were normal and her substernal chest pain was reproducible with palpation. An anteroposterior (AP) chest x-ray (CXR) was unremarkable.
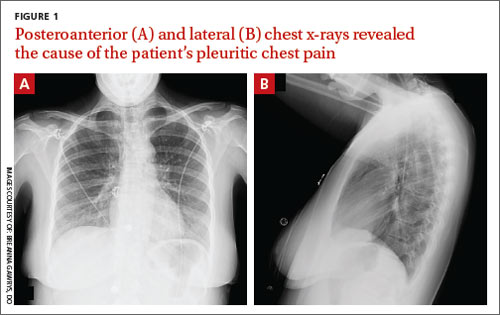
A “GI cocktail” (lidocaine, Mylanta and Donnatal), ketorolac, morphine, and lorazepam were administered in the ED, but did not provide the patient with any relief. She was admitted to the hospital to rule out acute coronary syndrome and was kept NPO overnight. A repeat CXR with posteroanterior (PA) and lateral views was also obtained (FIGURE 1A AND 1B).
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pneumomediastinum
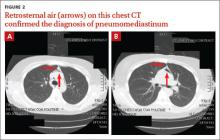
The PA and lateral view CXRs revealed the presence of retrosternal air, suggesting the patient had pneumomediastinum. A computed tomography (CT) scan of the chest also showed retrosternal air (FIGURE 2A AND 2B, arrows) and confirmed this diagnosis. To rule out esophageal perforation, the team ordered Gastrografin and barium swallow studies. The patient was kept NPO until both studies were confirmed to be negative.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by Hamman’s sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Differential diagnosis includes inflammatory conditions
The differential diagnosis for pneumomediastinum includes pericarditis, mediastinitis, Boerhaave syndrome, and acute coronary syndrome.
Pericarditis. In a patient with inflammation of the pericardium, you would hear reduced heart sounds and observe electrocardiogram (EKG) changes (eg, diffuse ST elevation in acute pericarditis). These signs typically would not be present in a patient with pneumomediastinum.1
Mediastinitis. Patients with mediastinitis—inflammation of the mediastinum—are more likely to have hypotension and shock.1
Boerhaave syndrome, or spontaneous esophageal perforation, has a similar presentation to pneumomediastinum but is more likely to be accompanied by hypotension and shock. Additionally, there would be extravasation of the contrast agent during swallow studies.2
Acute coronary syndrome is also part of the differential. However, in ACS, you would see ST changes on the patient’s EKG and elevated cardiac enzymes.1
Lateral x-rays are especially useful in making the diagnosis
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
For most patients, rest and pain control are key
Because pneumomediastinum is generally a self-limiting condition, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home.
If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation. If the result of this test is abnormal, a follow-up study with barium is recommended.3 Gastrografin swallow studies are the preferred initial study.3 A barium swallow study is more sensitive, but has a higher risk of causing pneumomediastinitis if an esophageal perforation is present.2
If the swallow study reveals a perforation, surgical decompression and antibiotics may be necessary.1,4,5
Our patient received subsequent serial CXRs that showed improvement in pneumomediastinum. Once our patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged home after a 3-day hospitalization with close follow-up. One week later, she had no further complaints and her pain had almost entirely resolved.
CORRESPONDENCE
Breanna Gawrys, DO, Fort Belvoir Community Hospital Family Medicine Residency, 9300 DeWitt Loop, Fort Belvoir, VA 22060; [email protected]
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Philadelphia, PA: Elsevier Health Sciences; 2005:2039–2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057.
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334.
4. Macia I, Moya J, Ramos R, et al. Spontaneous pneumomediastinum: 41 cases. Eur J Cardiothorac Surg. 2007;31:1110-1114.
5. Chalumeau M, Le Clainche L, Sayeg N, et al. Spontaneous pneumomediastinum in children. Pediatr Pulmonol. 2001;31:67-75.
A 22-year-old woman with a history of attention-deficit/hyperactivity disorder and childhood asthma came to the emergency department (ED) for treatment of a cramping, substernal, pleuritic chest pain she’d had for a week and the feeling of a “lump in her throat” that made it difficult and painful for her to swallow. The patient’s vital signs were normal and her substernal chest pain was reproducible with palpation. An anteroposterior (AP) chest x-ray (CXR) was unremarkable.
A “GI cocktail” (lidocaine, Mylanta and Donnatal), ketorolac, morphine, and lorazepam were administered in the ED, but did not provide the patient with any relief. She was admitted to the hospital to rule out acute coronary syndrome and was kept NPO overnight. A repeat CXR with posteroanterior (PA) and lateral views was also obtained (FIGURE 1A AND 1B).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pneumomediastinum
The PA and lateral view CXRs revealed the presence of retrosternal air, suggesting the patient had pneumomediastinum. A computed tomography (CT) scan of the chest also showed retrosternal air (FIGURE 2A AND 2B, arrows) and confirmed this diagnosis. To rule out esophageal perforation, the team ordered Gastrografin and barium swallow studies. The patient was kept NPO until both studies were confirmed to be negative.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by Hamman’s sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Differential diagnosis includes inflammatory conditions
The differential diagnosis for pneumomediastinum includes pericarditis, mediastinitis, Boerhaave syndrome, and acute coronary syndrome.
Pericarditis. In a patient with inflammation of the pericardium, you would hear reduced heart sounds and observe electrocardiogram (EKG) changes (eg, diffuse ST elevation in acute pericarditis). These signs typically would not be present in a patient with pneumomediastinum.1
Mediastinitis. Patients with mediastinitis—inflammation of the mediastinum—are more likely to have hypotension and shock.1
Boerhaave syndrome, or spontaneous esophageal perforation, has a similar presentation to pneumomediastinum but is more likely to be accompanied by hypotension and shock. Additionally, there would be extravasation of the contrast agent during swallow studies.2
Acute coronary syndrome is also part of the differential. However, in ACS, you would see ST changes on the patient’s EKG and elevated cardiac enzymes.1
Lateral x-rays are especially useful in making the diagnosis
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
For most patients, rest and pain control are key
Because pneumomediastinum is generally a self-limiting condition, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home.
If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation. If the result of this test is abnormal, a follow-up study with barium is recommended.3 Gastrografin swallow studies are the preferred initial study.3 A barium swallow study is more sensitive, but has a higher risk of causing pneumomediastinitis if an esophageal perforation is present.2
If the swallow study reveals a perforation, surgical decompression and antibiotics may be necessary.1,4,5
Our patient received subsequent serial CXRs that showed improvement in pneumomediastinum. Once our patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged home after a 3-day hospitalization with close follow-up. One week later, she had no further complaints and her pain had almost entirely resolved.
CORRESPONDENCE
Breanna Gawrys, DO, Fort Belvoir Community Hospital Family Medicine Residency, 9300 DeWitt Loop, Fort Belvoir, VA 22060; [email protected]
A 22-year-old woman with a history of attention-deficit/hyperactivity disorder and childhood asthma came to the emergency department (ED) for treatment of a cramping, substernal, pleuritic chest pain she’d had for a week and the feeling of a “lump in her throat” that made it difficult and painful for her to swallow. The patient’s vital signs were normal and her substernal chest pain was reproducible with palpation. An anteroposterior (AP) chest x-ray (CXR) was unremarkable.
A “GI cocktail” (lidocaine, Mylanta and Donnatal), ketorolac, morphine, and lorazepam were administered in the ED, but did not provide the patient with any relief. She was admitted to the hospital to rule out acute coronary syndrome and was kept NPO overnight. A repeat CXR with posteroanterior (PA) and lateral views was also obtained (FIGURE 1A AND 1B).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pneumomediastinum
The PA and lateral view CXRs revealed the presence of retrosternal air, suggesting the patient had pneumomediastinum. A computed tomography (CT) scan of the chest also showed retrosternal air (FIGURE 2A AND 2B, arrows) and confirmed this diagnosis. To rule out esophageal perforation, the team ordered Gastrografin and barium swallow studies. The patient was kept NPO until both studies were confirmed to be negative.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by Hamman’s sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Differential diagnosis includes inflammatory conditions
The differential diagnosis for pneumomediastinum includes pericarditis, mediastinitis, Boerhaave syndrome, and acute coronary syndrome.
Pericarditis. In a patient with inflammation of the pericardium, you would hear reduced heart sounds and observe electrocardiogram (EKG) changes (eg, diffuse ST elevation in acute pericarditis). These signs typically would not be present in a patient with pneumomediastinum.1
Mediastinitis. Patients with mediastinitis—inflammation of the mediastinum—are more likely to have hypotension and shock.1
Boerhaave syndrome, or spontaneous esophageal perforation, has a similar presentation to pneumomediastinum but is more likely to be accompanied by hypotension and shock. Additionally, there would be extravasation of the contrast agent during swallow studies.2
Acute coronary syndrome is also part of the differential. However, in ACS, you would see ST changes on the patient’s EKG and elevated cardiac enzymes.1
Lateral x-rays are especially useful in making the diagnosis
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
For most patients, rest and pain control are key
Because pneumomediastinum is generally a self-limiting condition, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home.
If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation. If the result of this test is abnormal, a follow-up study with barium is recommended.3 Gastrografin swallow studies are the preferred initial study.3 A barium swallow study is more sensitive, but has a higher risk of causing pneumomediastinitis if an esophageal perforation is present.2
If the swallow study reveals a perforation, surgical decompression and antibiotics may be necessary.1,4,5
Our patient received subsequent serial CXRs that showed improvement in pneumomediastinum. Once our patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged home after a 3-day hospitalization with close follow-up. One week later, she had no further complaints and her pain had almost entirely resolved.
CORRESPONDENCE
Breanna Gawrys, DO, Fort Belvoir Community Hospital Family Medicine Residency, 9300 DeWitt Loop, Fort Belvoir, VA 22060; [email protected]
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Philadelphia, PA: Elsevier Health Sciences; 2005:2039–2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057.
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334.
4. Macia I, Moya J, Ramos R, et al. Spontaneous pneumomediastinum: 41 cases. Eur J Cardiothorac Surg. 2007;31:1110-1114.
5. Chalumeau M, Le Clainche L, Sayeg N, et al. Spontaneous pneumomediastinum in children. Pediatr Pulmonol. 2001;31:67-75.
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Philadelphia, PA: Elsevier Health Sciences; 2005:2039–2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057.
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334.
4. Macia I, Moya J, Ramos R, et al. Spontaneous pneumomediastinum: 41 cases. Eur J Cardiothorac Surg. 2007;31:1110-1114.
5. Chalumeau M, Le Clainche L, Sayeg N, et al. Spontaneous pneumomediastinum in children. Pediatr Pulmonol. 2001;31:67-75.
Once-daily inhaled fluticasone-vilanterol approved for adult asthma
Fixed-dose combinations of the inhaled corticosteroid (ICS) fluticasone and the long-acting beta-agonist (LABA) vilanterol have been approved by the Food and Drug Administration for treating asthma in adults, the manufacturer, GlaxoSmithKline, announced on April 30.
Vilanterol, as part of the combination product, is the first new LABA approved for asthma in 15 years and fluticasone-vilanterol is the first once-daily inhaled ICS-LABA combination treatment approved for asthma.
Two strengths have been approved, 100 mcg or 200 mcg of fluticasone with 25 mcg of vilanterol, administered once a day with a dry powder inhaler, for people aged 18 years and older, according to the company’s statement. The products are marketed as Breo Ellipta. In 2013, the 100/25 mcg dose was approved to treat chronic obstructive pulmonary disease.
The company had filed for approval for treating asthma in adolescents aged 12-17 years who were studied in the same trials as adults, but the FDA did not approve the product in this age group. In the statement, GSK said that the FDA issued a “complete response” letter for the younger age group, “stating that the data submitted do not show adequate risk-benefit to support the approval in these patients,” and that more data “would be required to further demonstrate the safety and efficacy in this population.”
(The FDA issues complete response letters to companies when approval is not supported by the company’s application; the agency does not make this information public, but manufacturers often make that information available.)
The FDA decision reflects the recommendations of the FDA’s Pulmonary-Allergy Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committee, which, at a meeting on March 19, voted 16-4 that the efficacy and safety data supported the approval of both proposed doses in adults. But the panels voted 19-1 that the data did not support approval for adolescents, aged 12-17 years, citing uncertainty about safety and inconsistent efficacy results in this age group; panelists said that a separate safety and efficacy study in adolescents was needed.
As with other LABA products, the prescribing information for Breo Ellipta includes a boxed warning about the increased risk of asthma-related deaths associated with LABAs.
Unlike the LABAs salmeterol and formoterol, vilanterol is not approved for use as a single agent. Fluticasone (100 mcg and 200 mcg) is approved for treating asthma.
Serious adverse events associated with Breo Ellipta should be reported to the FDA’s MedWatch program at 800-332-1088. Prescribing information can be found at www.gsksource.com.
Fixed-dose combinations of the inhaled corticosteroid (ICS) fluticasone and the long-acting beta-agonist (LABA) vilanterol have been approved by the Food and Drug Administration for treating asthma in adults, the manufacturer, GlaxoSmithKline, announced on April 30.
Vilanterol, as part of the combination product, is the first new LABA approved for asthma in 15 years and fluticasone-vilanterol is the first once-daily inhaled ICS-LABA combination treatment approved for asthma.
Two strengths have been approved, 100 mcg or 200 mcg of fluticasone with 25 mcg of vilanterol, administered once a day with a dry powder inhaler, for people aged 18 years and older, according to the company’s statement. The products are marketed as Breo Ellipta. In 2013, the 100/25 mcg dose was approved to treat chronic obstructive pulmonary disease.
The company had filed for approval for treating asthma in adolescents aged 12-17 years who were studied in the same trials as adults, but the FDA did not approve the product in this age group. In the statement, GSK said that the FDA issued a “complete response” letter for the younger age group, “stating that the data submitted do not show adequate risk-benefit to support the approval in these patients,” and that more data “would be required to further demonstrate the safety and efficacy in this population.”
(The FDA issues complete response letters to companies when approval is not supported by the company’s application; the agency does not make this information public, but manufacturers often make that information available.)
The FDA decision reflects the recommendations of the FDA’s Pulmonary-Allergy Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committee, which, at a meeting on March 19, voted 16-4 that the efficacy and safety data supported the approval of both proposed doses in adults. But the panels voted 19-1 that the data did not support approval for adolescents, aged 12-17 years, citing uncertainty about safety and inconsistent efficacy results in this age group; panelists said that a separate safety and efficacy study in adolescents was needed.
As with other LABA products, the prescribing information for Breo Ellipta includes a boxed warning about the increased risk of asthma-related deaths associated with LABAs.
Unlike the LABAs salmeterol and formoterol, vilanterol is not approved for use as a single agent. Fluticasone (100 mcg and 200 mcg) is approved for treating asthma.
Serious adverse events associated with Breo Ellipta should be reported to the FDA’s MedWatch program at 800-332-1088. Prescribing information can be found at www.gsksource.com.
Fixed-dose combinations of the inhaled corticosteroid (ICS) fluticasone and the long-acting beta-agonist (LABA) vilanterol have been approved by the Food and Drug Administration for treating asthma in adults, the manufacturer, GlaxoSmithKline, announced on April 30.
Vilanterol, as part of the combination product, is the first new LABA approved for asthma in 15 years and fluticasone-vilanterol is the first once-daily inhaled ICS-LABA combination treatment approved for asthma.
Two strengths have been approved, 100 mcg or 200 mcg of fluticasone with 25 mcg of vilanterol, administered once a day with a dry powder inhaler, for people aged 18 years and older, according to the company’s statement. The products are marketed as Breo Ellipta. In 2013, the 100/25 mcg dose was approved to treat chronic obstructive pulmonary disease.
The company had filed for approval for treating asthma in adolescents aged 12-17 years who were studied in the same trials as adults, but the FDA did not approve the product in this age group. In the statement, GSK said that the FDA issued a “complete response” letter for the younger age group, “stating that the data submitted do not show adequate risk-benefit to support the approval in these patients,” and that more data “would be required to further demonstrate the safety and efficacy in this population.”
(The FDA issues complete response letters to companies when approval is not supported by the company’s application; the agency does not make this information public, but manufacturers often make that information available.)
The FDA decision reflects the recommendations of the FDA’s Pulmonary-Allergy Drugs Advisory Committee and Drug Safety and Risk Management Advisory Committee, which, at a meeting on March 19, voted 16-4 that the efficacy and safety data supported the approval of both proposed doses in adults. But the panels voted 19-1 that the data did not support approval for adolescents, aged 12-17 years, citing uncertainty about safety and inconsistent efficacy results in this age group; panelists said that a separate safety and efficacy study in adolescents was needed.
As with other LABA products, the prescribing information for Breo Ellipta includes a boxed warning about the increased risk of asthma-related deaths associated with LABAs.
Unlike the LABAs salmeterol and formoterol, vilanterol is not approved for use as a single agent. Fluticasone (100 mcg and 200 mcg) is approved for treating asthma.
Serious adverse events associated with Breo Ellipta should be reported to the FDA’s MedWatch program at 800-332-1088. Prescribing information can be found at www.gsksource.com.
ELCC: Urine tumor DNA shows high testing promise
GENEVA – Blood sampling provides a less invasive alternative to tumor biopsy for collecting cancer DNA for mutation testing, but retrieving tumor DNA from a patient’s urine is least invasive of all.
In an early phase of clinical investigation, researchers assessed the feasibility of using urine to collect cell-free tumor DNA to detect mutations in 34 patients with advanced non–small cell lung cancer. The results suggested that testing circulating tumor (ct) DNA isolated from patients’ urine was sensitive compared with testing DNA from biopsied specimens of the primary tumor, and it was able to flag tumor changes early, before clinically-identifiable effects appeared, Dr. Hatim Husain said at the European Lung Cancer Conference.
He used ctDNA isolated from patients’ urine to test for the presence of three different resistance mutations within the epidermal growth factor receptor (EGFR) gene. Genetic testing of biopsy specimens showed 10 patients carried the T790M mutation, 18 carried an exon 19 deletion, and eight carried an exon 21 L858R mutation. Of these 36 mutations in 34 patients, testing ctDNA isolated from urine identified 35 as positive, a 97% overall concordance rate.
In addition, testing with ctDNA from urine also picked up three additional T790M mutations not seen in the three corresponding tumor-biopsy specimens, but in patients with high clinical suspicion for carrying an EGFR mutation, Dr. Husain reported.
Further evidence for the utility of urinary ctDNA came from following 22 patients on treatment with erlotinib (Tarceva) and monitored for their acquisition of an EGFR-gene mutation making the tumor erlotinib resistant. Dr. Husain and his associates ran a DNA test every 3-6 weeks and tracked the time until patients developed radiographic progression. Using urinary ctDNA, they found four patients who developed EGFR mutations 29-111 days before clinical progression of the tumor became radiographically apparent.
The ctDNA that ends up in a patient’s urine starts out circulating in the blood; urine works as the main elimination route. Urine ctDNA is more concentrated than in blood, and ctDNA remains stable in urine at ambient temperature for 2 weeks, said Dr. Husain, an oncology researcher at the University of California San Diego, La Jolla.
“These interim results suggest that use of urinary ctDNA has potential to detect EGFR T790M status in a higher number of study subjects and may make some patients eligible for therapy who would by tissue biopsy be falsely classified as negative,” Dr. Husain said in a written statement. “Detecting the emergence of EGFR T790M mutations before progression has the potential to enable physicians to better align therapeutic selection and inform early therapeutic decision making,” he said.
Testing ctDNA in patients’ urine is a “novel way to do noninvasive testing,” said Dr. Egbert F. Smit, professor of pulmonary medicine at VU University Medical Center in Amsterdam and the meeting’s designated discussant for Dr. Husain’s report. “It’s attractive for collecting ctDNA because you get a high concentration, and it has potential for a high level of sensitivity. It may have potential for showing how a tumor reacts to treatment. The method seems robust, but we still need data on reproducibility and cost effectiveness,” Dr. Smit cautioned.
On Twitter @mitchelzoler
GENEVA – Blood sampling provides a less invasive alternative to tumor biopsy for collecting cancer DNA for mutation testing, but retrieving tumor DNA from a patient’s urine is least invasive of all.
In an early phase of clinical investigation, researchers assessed the feasibility of using urine to collect cell-free tumor DNA to detect mutations in 34 patients with advanced non–small cell lung cancer. The results suggested that testing circulating tumor (ct) DNA isolated from patients’ urine was sensitive compared with testing DNA from biopsied specimens of the primary tumor, and it was able to flag tumor changes early, before clinically-identifiable effects appeared, Dr. Hatim Husain said at the European Lung Cancer Conference.
He used ctDNA isolated from patients’ urine to test for the presence of three different resistance mutations within the epidermal growth factor receptor (EGFR) gene. Genetic testing of biopsy specimens showed 10 patients carried the T790M mutation, 18 carried an exon 19 deletion, and eight carried an exon 21 L858R mutation. Of these 36 mutations in 34 patients, testing ctDNA isolated from urine identified 35 as positive, a 97% overall concordance rate.
In addition, testing with ctDNA from urine also picked up three additional T790M mutations not seen in the three corresponding tumor-biopsy specimens, but in patients with high clinical suspicion for carrying an EGFR mutation, Dr. Husain reported.
Further evidence for the utility of urinary ctDNA came from following 22 patients on treatment with erlotinib (Tarceva) and monitored for their acquisition of an EGFR-gene mutation making the tumor erlotinib resistant. Dr. Husain and his associates ran a DNA test every 3-6 weeks and tracked the time until patients developed radiographic progression. Using urinary ctDNA, they found four patients who developed EGFR mutations 29-111 days before clinical progression of the tumor became radiographically apparent.
The ctDNA that ends up in a patient’s urine starts out circulating in the blood; urine works as the main elimination route. Urine ctDNA is more concentrated than in blood, and ctDNA remains stable in urine at ambient temperature for 2 weeks, said Dr. Husain, an oncology researcher at the University of California San Diego, La Jolla.
“These interim results suggest that use of urinary ctDNA has potential to detect EGFR T790M status in a higher number of study subjects and may make some patients eligible for therapy who would by tissue biopsy be falsely classified as negative,” Dr. Husain said in a written statement. “Detecting the emergence of EGFR T790M mutations before progression has the potential to enable physicians to better align therapeutic selection and inform early therapeutic decision making,” he said.
Testing ctDNA in patients’ urine is a “novel way to do noninvasive testing,” said Dr. Egbert F. Smit, professor of pulmonary medicine at VU University Medical Center in Amsterdam and the meeting’s designated discussant for Dr. Husain’s report. “It’s attractive for collecting ctDNA because you get a high concentration, and it has potential for a high level of sensitivity. It may have potential for showing how a tumor reacts to treatment. The method seems robust, but we still need data on reproducibility and cost effectiveness,” Dr. Smit cautioned.
On Twitter @mitchelzoler
GENEVA – Blood sampling provides a less invasive alternative to tumor biopsy for collecting cancer DNA for mutation testing, but retrieving tumor DNA from a patient’s urine is least invasive of all.
In an early phase of clinical investigation, researchers assessed the feasibility of using urine to collect cell-free tumor DNA to detect mutations in 34 patients with advanced non–small cell lung cancer. The results suggested that testing circulating tumor (ct) DNA isolated from patients’ urine was sensitive compared with testing DNA from biopsied specimens of the primary tumor, and it was able to flag tumor changes early, before clinically-identifiable effects appeared, Dr. Hatim Husain said at the European Lung Cancer Conference.
He used ctDNA isolated from patients’ urine to test for the presence of three different resistance mutations within the epidermal growth factor receptor (EGFR) gene. Genetic testing of biopsy specimens showed 10 patients carried the T790M mutation, 18 carried an exon 19 deletion, and eight carried an exon 21 L858R mutation. Of these 36 mutations in 34 patients, testing ctDNA isolated from urine identified 35 as positive, a 97% overall concordance rate.
In addition, testing with ctDNA from urine also picked up three additional T790M mutations not seen in the three corresponding tumor-biopsy specimens, but in patients with high clinical suspicion for carrying an EGFR mutation, Dr. Husain reported.
Further evidence for the utility of urinary ctDNA came from following 22 patients on treatment with erlotinib (Tarceva) and monitored for their acquisition of an EGFR-gene mutation making the tumor erlotinib resistant. Dr. Husain and his associates ran a DNA test every 3-6 weeks and tracked the time until patients developed radiographic progression. Using urinary ctDNA, they found four patients who developed EGFR mutations 29-111 days before clinical progression of the tumor became radiographically apparent.
The ctDNA that ends up in a patient’s urine starts out circulating in the blood; urine works as the main elimination route. Urine ctDNA is more concentrated than in blood, and ctDNA remains stable in urine at ambient temperature for 2 weeks, said Dr. Husain, an oncology researcher at the University of California San Diego, La Jolla.
“These interim results suggest that use of urinary ctDNA has potential to detect EGFR T790M status in a higher number of study subjects and may make some patients eligible for therapy who would by tissue biopsy be falsely classified as negative,” Dr. Husain said in a written statement. “Detecting the emergence of EGFR T790M mutations before progression has the potential to enable physicians to better align therapeutic selection and inform early therapeutic decision making,” he said.
Testing ctDNA in patients’ urine is a “novel way to do noninvasive testing,” said Dr. Egbert F. Smit, professor of pulmonary medicine at VU University Medical Center in Amsterdam and the meeting’s designated discussant for Dr. Husain’s report. “It’s attractive for collecting ctDNA because you get a high concentration, and it has potential for a high level of sensitivity. It may have potential for showing how a tumor reacts to treatment. The method seems robust, but we still need data on reproducibility and cost effectiveness,” Dr. Smit cautioned.
On Twitter @mitchelzoler
AT ELCC 2015
Key clinical point: Urine showed potential as a fully noninvasive source for circulating tumor DNA with high testing concordance compared with tumor-biopsy DNA in initial clinical experience with 34 lung cancer patients.
Major finding: Urine circulating tumor DNA had overall concordance of 97% compared with tumor-biopsy DNA for detecting treatment-altering mutations.
Data source: Single-center study of 34 patients with advanced non-small cell lung cancer.
Disclosures: The study was sponsored by Trovagene, the company developing the tests used in the study. Several coauthors on the study are Trovagene employees. Dr. Husain had no personal disclosures.
PAS: Low-dose hydrocortisone improves outcomes in extreme preemies
SAN DIEGO – Early prophylactic very-low-dose hydrocortisone improved survival free of bronchopulmonary dysplasia in extremely preterm neonates in a large French randomized trial.
The intervention also brought positive results on multiple secondary endpoints, including rates of extubation by day 10 and patent ductus arteriosus closure without resort to ligation, Dr. Olivier Baud reported at the annual meeting of the Pediatric Academic Societies.
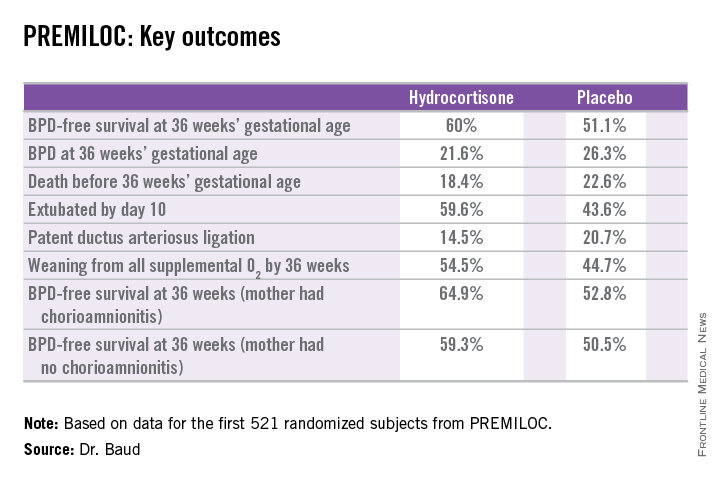
He presented the findings of PREMILOC, a double-blind, randomized, placebo-controlled, 21-center French study. The study was halted by the data safety monitoring board for ethical reasons, based upon compelling evidence of superiority after scrutinizing results in the first 521 randomized subjects.
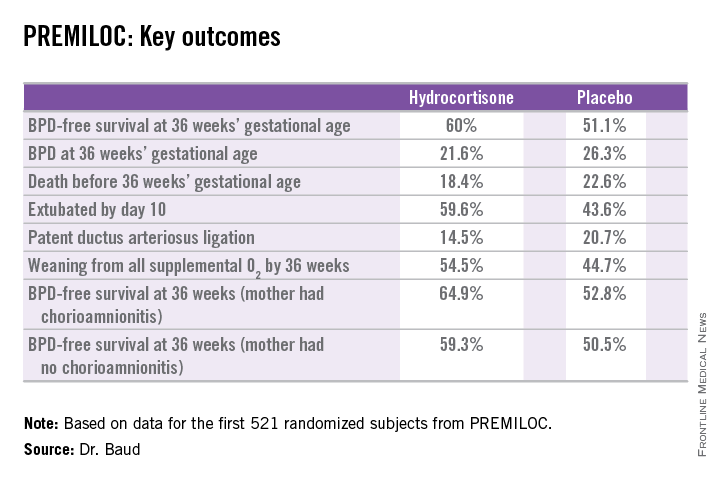
The primary outcome – survival free of bronchopulmonary dysplasia (BPD) at 36 weeks of gestational age – occurred in 60% of the hydrocortisone group, compared with 51% of placebo-treated controls, for an adjusted 48% increased likelihood of favorable outcome. The number-needed-to-treat was 11 patients, said Dr. Baud, professor of pediatrics and chief of the neonatal medicine unit at Robert Debré Hospital in Paris and Paris Diderot University.
The intervention involved administration of intravenous hydrocortisone sodium succinate for the first 10 postnatal days. The dose was 0.5 mg/kg/12 hours for 7 days followed by 0.5 mg/kg/24 hours for 3 days. Treatment began as soon as possible after birth and always within 24 hours. The total cumulative dose of hydrocortisone was 8.5 mg/kg. That’s 15-30 times less corticosteroid than employed in earlier studies of higher-dose dexamethasone, where gastrointestinal bleeding, intestinal perforation, and other serious adverse events were a major problem.
“This is the lowest hydrocortisone dose ever studied in a randomized controlled trial,” according to the neonatologist.
All study participants were born at 24-27 weeks’ gestational age. In order to minimize the risk of treatment-related serious adverse events, especially GI perforation – patients with intrauterine growth retardation or who were on nonsteroidal anti-inflammatory agents were ineligible for the trial.
The rationale for the study, Dr. Baud explained, comes from the landmark work of Dr. Kristi L. Watterberg, professor of pediatrics at the University of New Mexico, Albuquerque, who has argued that early adrenal insufficiency in extremely small-for-gestational-age infants is linked to BPD, patent ductus arteriosus, persistent lung inflammation, and poor enteral nutrition. She introduced the concept that administration of very-low-dose corticosteroids at a dose comparable to normal endogenous steroid production could serve as a global therapy addressing these multiple health problems.
Two prior studies of low-dose hydrocortisone in extremely preterm babies were halted due to an increase in GI perforation, Dr. Baud noted, which is why the French investigators excluded neonates at increased risk for this complication.
In a prespecified PREMILOC subgroup analysis stratified by gestational age, the rate of survival free of BPD at 36 weeks among neonates born at 24-25 weeks was 33.7% with hydrocortisone, compared with 23.3% with placebo, for an adjusted 67% increased likelihood of a positive outcome with active treatment. In babies born at 26-27 weeks, the rates were 72.7% and 65.3%, respectively. On the other hand, hydrocortisone was associated with a highly significant 55% relative risk reduction for neonatal mortality in infants born at 26-27 weeks.
Post hoc analysis spotlighted several factors which were unexpectedly associated with differential impacts on outcome. For example, hydrocortisone achieved a significant benefit in terms of survival free of BPD at 36 weeks only in females, where the rates were 69.4% versus 53%, for an adjusted 2.25-fold increased rate. In males, the rate was 51.1% with hydrocortisone, compared with 49.7% in controls.
There were no differences between the hydrocortisone and placebo groups in rates of any serious adverse events, including GI perforation, necrotizing enterocolitis, air leaks, severe sepsis, or persistent pulmonary hypertension.
Dr. Baud said the next stage of the PREMILOC study will be to report 2-year outcomes; a greater than 90% follow-up rate is anticipated. Also, he and his coinvestigators are planning ancillary studies examining placental findings, the impact of pretreatment serum cortisol levels, thyroid function, and other issues.
“Our goal is to identify a targeted population that could strongly benefit from prophylactic hydrocortisone,” the neonatologist said.
He reported having no financial conflicts regarding the study, supported by INSERM and other French national research organizations.
SAN DIEGO – Early prophylactic very-low-dose hydrocortisone improved survival free of bronchopulmonary dysplasia in extremely preterm neonates in a large French randomized trial.
The intervention also brought positive results on multiple secondary endpoints, including rates of extubation by day 10 and patent ductus arteriosus closure without resort to ligation, Dr. Olivier Baud reported at the annual meeting of the Pediatric Academic Societies.

He presented the findings of PREMILOC, a double-blind, randomized, placebo-controlled, 21-center French study. The study was halted by the data safety monitoring board for ethical reasons, based upon compelling evidence of superiority after scrutinizing results in the first 521 randomized subjects.
The primary outcome – survival free of bronchopulmonary dysplasia (BPD) at 36 weeks of gestational age – occurred in 60% of the hydrocortisone group, compared with 51% of placebo-treated controls, for an adjusted 48% increased likelihood of favorable outcome. The number-needed-to-treat was 11 patients, said Dr. Baud, professor of pediatrics and chief of the neonatal medicine unit at Robert Debré Hospital in Paris and Paris Diderot University.
The intervention involved administration of intravenous hydrocortisone sodium succinate for the first 10 postnatal days. The dose was 0.5 mg/kg/12 hours for 7 days followed by 0.5 mg/kg/24 hours for 3 days. Treatment began as soon as possible after birth and always within 24 hours. The total cumulative dose of hydrocortisone was 8.5 mg/kg. That’s 15-30 times less corticosteroid than employed in earlier studies of higher-dose dexamethasone, where gastrointestinal bleeding, intestinal perforation, and other serious adverse events were a major problem.
“This is the lowest hydrocortisone dose ever studied in a randomized controlled trial,” according to the neonatologist.
All study participants were born at 24-27 weeks’ gestational age. In order to minimize the risk of treatment-related serious adverse events, especially GI perforation – patients with intrauterine growth retardation or who were on nonsteroidal anti-inflammatory agents were ineligible for the trial.
The rationale for the study, Dr. Baud explained, comes from the landmark work of Dr. Kristi L. Watterberg, professor of pediatrics at the University of New Mexico, Albuquerque, who has argued that early adrenal insufficiency in extremely small-for-gestational-age infants is linked to BPD, patent ductus arteriosus, persistent lung inflammation, and poor enteral nutrition. She introduced the concept that administration of very-low-dose corticosteroids at a dose comparable to normal endogenous steroid production could serve as a global therapy addressing these multiple health problems.
Two prior studies of low-dose hydrocortisone in extremely preterm babies were halted due to an increase in GI perforation, Dr. Baud noted, which is why the French investigators excluded neonates at increased risk for this complication.
In a prespecified PREMILOC subgroup analysis stratified by gestational age, the rate of survival free of BPD at 36 weeks among neonates born at 24-25 weeks was 33.7% with hydrocortisone, compared with 23.3% with placebo, for an adjusted 67% increased likelihood of a positive outcome with active treatment. In babies born at 26-27 weeks, the rates were 72.7% and 65.3%, respectively. On the other hand, hydrocortisone was associated with a highly significant 55% relative risk reduction for neonatal mortality in infants born at 26-27 weeks.
Post hoc analysis spotlighted several factors which were unexpectedly associated with differential impacts on outcome. For example, hydrocortisone achieved a significant benefit in terms of survival free of BPD at 36 weeks only in females, where the rates were 69.4% versus 53%, for an adjusted 2.25-fold increased rate. In males, the rate was 51.1% with hydrocortisone, compared with 49.7% in controls.
There were no differences between the hydrocortisone and placebo groups in rates of any serious adverse events, including GI perforation, necrotizing enterocolitis, air leaks, severe sepsis, or persistent pulmonary hypertension.
Dr. Baud said the next stage of the PREMILOC study will be to report 2-year outcomes; a greater than 90% follow-up rate is anticipated. Also, he and his coinvestigators are planning ancillary studies examining placental findings, the impact of pretreatment serum cortisol levels, thyroid function, and other issues.
“Our goal is to identify a targeted population that could strongly benefit from prophylactic hydrocortisone,” the neonatologist said.
He reported having no financial conflicts regarding the study, supported by INSERM and other French national research organizations.
SAN DIEGO – Early prophylactic very-low-dose hydrocortisone improved survival free of bronchopulmonary dysplasia in extremely preterm neonates in a large French randomized trial.
The intervention also brought positive results on multiple secondary endpoints, including rates of extubation by day 10 and patent ductus arteriosus closure without resort to ligation, Dr. Olivier Baud reported at the annual meeting of the Pediatric Academic Societies.

He presented the findings of PREMILOC, a double-blind, randomized, placebo-controlled, 21-center French study. The study was halted by the data safety monitoring board for ethical reasons, based upon compelling evidence of superiority after scrutinizing results in the first 521 randomized subjects.
The primary outcome – survival free of bronchopulmonary dysplasia (BPD) at 36 weeks of gestational age – occurred in 60% of the hydrocortisone group, compared with 51% of placebo-treated controls, for an adjusted 48% increased likelihood of favorable outcome. The number-needed-to-treat was 11 patients, said Dr. Baud, professor of pediatrics and chief of the neonatal medicine unit at Robert Debré Hospital in Paris and Paris Diderot University.
The intervention involved administration of intravenous hydrocortisone sodium succinate for the first 10 postnatal days. The dose was 0.5 mg/kg/12 hours for 7 days followed by 0.5 mg/kg/24 hours for 3 days. Treatment began as soon as possible after birth and always within 24 hours. The total cumulative dose of hydrocortisone was 8.5 mg/kg. That’s 15-30 times less corticosteroid than employed in earlier studies of higher-dose dexamethasone, where gastrointestinal bleeding, intestinal perforation, and other serious adverse events were a major problem.
“This is the lowest hydrocortisone dose ever studied in a randomized controlled trial,” according to the neonatologist.
All study participants were born at 24-27 weeks’ gestational age. In order to minimize the risk of treatment-related serious adverse events, especially GI perforation – patients with intrauterine growth retardation or who were on nonsteroidal anti-inflammatory agents were ineligible for the trial.
The rationale for the study, Dr. Baud explained, comes from the landmark work of Dr. Kristi L. Watterberg, professor of pediatrics at the University of New Mexico, Albuquerque, who has argued that early adrenal insufficiency in extremely small-for-gestational-age infants is linked to BPD, patent ductus arteriosus, persistent lung inflammation, and poor enteral nutrition. She introduced the concept that administration of very-low-dose corticosteroids at a dose comparable to normal endogenous steroid production could serve as a global therapy addressing these multiple health problems.
Two prior studies of low-dose hydrocortisone in extremely preterm babies were halted due to an increase in GI perforation, Dr. Baud noted, which is why the French investigators excluded neonates at increased risk for this complication.
In a prespecified PREMILOC subgroup analysis stratified by gestational age, the rate of survival free of BPD at 36 weeks among neonates born at 24-25 weeks was 33.7% with hydrocortisone, compared with 23.3% with placebo, for an adjusted 67% increased likelihood of a positive outcome with active treatment. In babies born at 26-27 weeks, the rates were 72.7% and 65.3%, respectively. On the other hand, hydrocortisone was associated with a highly significant 55% relative risk reduction for neonatal mortality in infants born at 26-27 weeks.
Post hoc analysis spotlighted several factors which were unexpectedly associated with differential impacts on outcome. For example, hydrocortisone achieved a significant benefit in terms of survival free of BPD at 36 weeks only in females, where the rates were 69.4% versus 53%, for an adjusted 2.25-fold increased rate. In males, the rate was 51.1% with hydrocortisone, compared with 49.7% in controls.
There were no differences between the hydrocortisone and placebo groups in rates of any serious adverse events, including GI perforation, necrotizing enterocolitis, air leaks, severe sepsis, or persistent pulmonary hypertension.
Dr. Baud said the next stage of the PREMILOC study will be to report 2-year outcomes; a greater than 90% follow-up rate is anticipated. Also, he and his coinvestigators are planning ancillary studies examining placental findings, the impact of pretreatment serum cortisol levels, thyroid function, and other issues.
“Our goal is to identify a targeted population that could strongly benefit from prophylactic hydrocortisone,” the neonatologist said.
He reported having no financial conflicts regarding the study, supported by INSERM and other French national research organizations.
AT THE PAS ANNUAL MEETING
Key clinical point: In extremely premature neonates, prophylactic very-low-dose hydrocortisone improved the likelihood of survival free of bronchopulmonary dysplasia at 36 weeks’ gestational age by roughly 50%.
Major finding: The number needed to treat to achieve one additional survival without bronchopulmonary dysplasia at 36 weeks’ gestational age was 11.
Data source: This was a randomized, double-blind, placebo-controlled, multicenter French study involving 521 patients born at 24-27 weeks’ gestational age.
Disclosures: The PREMILOC study was supported by INSERM and other French scientific research organizations. The presenter reported having no financial conflicts.
Rociletinib active against resistant NSCLC
Rociletinib, a third-generation EGFR tyrosine kinase inhibitor that targets mutations causing treatment resistance, showed sustained activity against resistant non–small cell lung cancer (NSCLC) in a phase I clinical trial, according to a report published online April 30 in the New England Journal of Medicine.
The study, sponsored by Clovis Oncology, the maker of rociletinib, involved 130 patients enrolled over the course of 2 years at 10 medical centers in the United States, France, and Australia. All the study participants had received at least one first- or second-generation EGFR inhibitor (usually erlotinib), and their NSCLC tumors had mutated and developed resistance to the therapy. Half of these patients had three or more sites of metastasis, and 44% had brain involvement, said Dr. Lecia V. Sequist of the deparment of medicine, Massachusetts General Hospital, Boston, and her associates.
A total of 92 patients received therapeutic doses of rociletinib. After a median follow-up of 10 weeks, the response rate among the 46 whose cancer had known EGFR T790M mutations was 59%; 43 of these 46 patients achieved a partial response, a complete response, or disease stabilization. As expected, the response rate was lower, at 29%, among the 17 patients whose tumors were T790M negative, the investigators reported (New Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMoa1413654]).
“Rociletinib did not cause the syndrome of rash, stomatitis, and paronychia that is associated with inhibition of nonmutant EGFR,” they wrote, and the most common grade 3 toxic effect was hyperglycemia. No hyperglycemic events led to discontinuation of treatment, and most were managed with a dose reduction plus use of an oral hypoglycemic agent, usually metformin. Both the hyperglycemia and the metformin may have contributed to gastrointestinal adverse events, which were generally mild.
The main limitation of this study is the small number of patients who have received rociletinib. Larger studies of the agent are now underway, Dr. Sequist and her associates said.
This study was funded by Clovis Oncology, maker of rociletinib, which also participated in study design and data collection and analysis. Dr. Sequist reported receiving personal fees from, and consulting for, Clovis Oncology, AstraZeneca, Boehringer Ingelheim, Novartis, Genentech, Merrimack, and Taiho, and her associates reported ties to numerous industry sources.
Rociletinib, a third-generation EGFR tyrosine kinase inhibitor that targets mutations causing treatment resistance, showed sustained activity against resistant non–small cell lung cancer (NSCLC) in a phase I clinical trial, according to a report published online April 30 in the New England Journal of Medicine.
The study, sponsored by Clovis Oncology, the maker of rociletinib, involved 130 patients enrolled over the course of 2 years at 10 medical centers in the United States, France, and Australia. All the study participants had received at least one first- or second-generation EGFR inhibitor (usually erlotinib), and their NSCLC tumors had mutated and developed resistance to the therapy. Half of these patients had three or more sites of metastasis, and 44% had brain involvement, said Dr. Lecia V. Sequist of the deparment of medicine, Massachusetts General Hospital, Boston, and her associates.
A total of 92 patients received therapeutic doses of rociletinib. After a median follow-up of 10 weeks, the response rate among the 46 whose cancer had known EGFR T790M mutations was 59%; 43 of these 46 patients achieved a partial response, a complete response, or disease stabilization. As expected, the response rate was lower, at 29%, among the 17 patients whose tumors were T790M negative, the investigators reported (New Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMoa1413654]).
“Rociletinib did not cause the syndrome of rash, stomatitis, and paronychia that is associated with inhibition of nonmutant EGFR,” they wrote, and the most common grade 3 toxic effect was hyperglycemia. No hyperglycemic events led to discontinuation of treatment, and most were managed with a dose reduction plus use of an oral hypoglycemic agent, usually metformin. Both the hyperglycemia and the metformin may have contributed to gastrointestinal adverse events, which were generally mild.
The main limitation of this study is the small number of patients who have received rociletinib. Larger studies of the agent are now underway, Dr. Sequist and her associates said.
This study was funded by Clovis Oncology, maker of rociletinib, which also participated in study design and data collection and analysis. Dr. Sequist reported receiving personal fees from, and consulting for, Clovis Oncology, AstraZeneca, Boehringer Ingelheim, Novartis, Genentech, Merrimack, and Taiho, and her associates reported ties to numerous industry sources.
Rociletinib, a third-generation EGFR tyrosine kinase inhibitor that targets mutations causing treatment resistance, showed sustained activity against resistant non–small cell lung cancer (NSCLC) in a phase I clinical trial, according to a report published online April 30 in the New England Journal of Medicine.
The study, sponsored by Clovis Oncology, the maker of rociletinib, involved 130 patients enrolled over the course of 2 years at 10 medical centers in the United States, France, and Australia. All the study participants had received at least one first- or second-generation EGFR inhibitor (usually erlotinib), and their NSCLC tumors had mutated and developed resistance to the therapy. Half of these patients had three or more sites of metastasis, and 44% had brain involvement, said Dr. Lecia V. Sequist of the deparment of medicine, Massachusetts General Hospital, Boston, and her associates.
A total of 92 patients received therapeutic doses of rociletinib. After a median follow-up of 10 weeks, the response rate among the 46 whose cancer had known EGFR T790M mutations was 59%; 43 of these 46 patients achieved a partial response, a complete response, or disease stabilization. As expected, the response rate was lower, at 29%, among the 17 patients whose tumors were T790M negative, the investigators reported (New Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMoa1413654]).
“Rociletinib did not cause the syndrome of rash, stomatitis, and paronychia that is associated with inhibition of nonmutant EGFR,” they wrote, and the most common grade 3 toxic effect was hyperglycemia. No hyperglycemic events led to discontinuation of treatment, and most were managed with a dose reduction plus use of an oral hypoglycemic agent, usually metformin. Both the hyperglycemia and the metformin may have contributed to gastrointestinal adverse events, which were generally mild.
The main limitation of this study is the small number of patients who have received rociletinib. Larger studies of the agent are now underway, Dr. Sequist and her associates said.
This study was funded by Clovis Oncology, maker of rociletinib, which also participated in study design and data collection and analysis. Dr. Sequist reported receiving personal fees from, and consulting for, Clovis Oncology, AstraZeneca, Boehringer Ingelheim, Novartis, Genentech, Merrimack, and Taiho, and her associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Rociletinib exerted prolonged disease control in a phase I study of resistant non–small cell lung cancer.
Major finding: After a median follow-up of 10 weeks, the response rate among the 46 patients whose NSCLC had known EGFR T790M mutations was 59%.
Data source: An international phase I trial of rociletinib in 130 patients whose NSCLC developed resistance to EGFR tyrosine kinase inhibitors.
Disclosures: This study was funded by Clovis Oncology, maker of rociletinib, which also participated in study design and data collection and analysis. Dr. Sequist reported receiving personal fees from, and consulting for, Clovis Oncology, AstraZeneca, Boehringer Ingelheim, Novartis, Genentech, Merrimack, and Taiho, and her associates reported ties to numerous industry sources.
New agent targets EGFR resistance in NSCLC
A new agent targeting tumors that develop resistance to EGFR tyrosine kinase inhibitors proved to be “highly active” in a phase I clinical trial involving 253 patients with advanced, resistant NSCLC, according to a report published online April 30 in the New England Journal of Medicine.
Most patients who initially respond to EGFR tyrosine kinase inhibitors such as gefitinib, erlotinib, and afatinib develop resistance and show disease progression within 2 years, because the tumors develop additional EGFR mutations, particularly T790M resistance mutations. Preliminary studies suggested that a new oral agent, AZD9291, would target T790M-mediated resistance. In a phase I trial to assess its safety and efficacy, 127 study participants had known EGFR T790M mutations, and the remainder had other or unknown mutations, said Dr. Pasi A. Janne of the Lowe Center for Thoracic Oncology and the Belfer Institute for Applied Cancer Science, Dana-Farber Institute, Boston, and his associates.
The study – designed and funded by AstraZeneca, which also collected and analyzed the data in conjunction with the scientific authors – was conducted at 33 sites in Japan, South Korea, Taiwan, France, Spain, Germany, Australia, the United Kingdom, and the United States. Of the 239 patients who could be evaluated for a response, 123 (51%) showed a partial or complete response. In the subset of patients with known EGFR T790M mutations, the objective response rate was even higher, at 61%.
In contrast, the objective response rate was 21% in the subset of 61 patients who did not have known EGFR T790M mutations, the investigators said (N. Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMoa1411817]). Median progression-free survival was estimated to be 9.6 months in patients with EGFR T790M-positive tumors, although many are still alive and the final survival data have yet to be calculated. In contrast, progression-free survival was 2.8 months in patients with EGFR T790M-negative tumors.
No dose-limiting toxic effects occurred and therefore the maximal efficacy dose with an acceptable level of adverse events cannot be established yet. The most common adverse events were diarrhea (47% of patients), rash (40%), nausea (22%), and decreased appetite (21%). Serious events that were considered to be possibly treatment-related occurred in 6% of patients, and included one fatal case of pneumonia and six cases of pneumonitis-like events that resolved when the drug was discontinued.
These findings demonstrate that “a structurally distinct EGFR inhibitor, one that is selective for the mutated form of EGFR, can be clinically effective and has a side-effect profile that is not dose limiting in the majority of patients in whom T790M-mediated drug resistance has developed,” Dr. Janne and his associates said.
Astra Zeneca is the maker of AZD9291. Dr. Janne reported ties to AstraZeneca, Boehringer Ingelheim, Chugai, Pfizer, Merrimack, Clovis Oncology, Roche, and Gatekeeper, as well as several patents related to genetically targeted cancer treatments; his associates reported ties to numerous industry sources.
Finding effective and relatively safe agents to address drug resistance in NSCLC is encouraging, and there is every reason to be cautiously optimistic about lung cancer treatment. But it is almost certain that cancer cells will continue to evolve and eventually develop resistance to these agents also.
The keys to keeping up with evolving cancers are to continue performing genomic analysis of resistant lesions and to translate laboratory findings to the clinic as rapidly as possible.
Dr. Ramaswamy Govindan is with the Alvin J. Siteman Cancer Center at Washington University, St. Louis. He reported receiving consulting fees and honoraria from Pfizer, Merck, Boehringer Ingelheim, Clovis Oncology, Helsinn Healthcare, Genentech, AbbVie, and GlaxoSmithKline. Dr. Govindan made these remarks in an editorial accompanying Dr. Janne’s report (N. Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMe1500181]).
Finding effective and relatively safe agents to address drug resistance in NSCLC is encouraging, and there is every reason to be cautiously optimistic about lung cancer treatment. But it is almost certain that cancer cells will continue to evolve and eventually develop resistance to these agents also.
The keys to keeping up with evolving cancers are to continue performing genomic analysis of resistant lesions and to translate laboratory findings to the clinic as rapidly as possible.
Dr. Ramaswamy Govindan is with the Alvin J. Siteman Cancer Center at Washington University, St. Louis. He reported receiving consulting fees and honoraria from Pfizer, Merck, Boehringer Ingelheim, Clovis Oncology, Helsinn Healthcare, Genentech, AbbVie, and GlaxoSmithKline. Dr. Govindan made these remarks in an editorial accompanying Dr. Janne’s report (N. Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMe1500181]).
Finding effective and relatively safe agents to address drug resistance in NSCLC is encouraging, and there is every reason to be cautiously optimistic about lung cancer treatment. But it is almost certain that cancer cells will continue to evolve and eventually develop resistance to these agents also.
The keys to keeping up with evolving cancers are to continue performing genomic analysis of resistant lesions and to translate laboratory findings to the clinic as rapidly as possible.
Dr. Ramaswamy Govindan is with the Alvin J. Siteman Cancer Center at Washington University, St. Louis. He reported receiving consulting fees and honoraria from Pfizer, Merck, Boehringer Ingelheim, Clovis Oncology, Helsinn Healthcare, Genentech, AbbVie, and GlaxoSmithKline. Dr. Govindan made these remarks in an editorial accompanying Dr. Janne’s report (N. Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMe1500181]).
A new agent targeting tumors that develop resistance to EGFR tyrosine kinase inhibitors proved to be “highly active” in a phase I clinical trial involving 253 patients with advanced, resistant NSCLC, according to a report published online April 30 in the New England Journal of Medicine.
Most patients who initially respond to EGFR tyrosine kinase inhibitors such as gefitinib, erlotinib, and afatinib develop resistance and show disease progression within 2 years, because the tumors develop additional EGFR mutations, particularly T790M resistance mutations. Preliminary studies suggested that a new oral agent, AZD9291, would target T790M-mediated resistance. In a phase I trial to assess its safety and efficacy, 127 study participants had known EGFR T790M mutations, and the remainder had other or unknown mutations, said Dr. Pasi A. Janne of the Lowe Center for Thoracic Oncology and the Belfer Institute for Applied Cancer Science, Dana-Farber Institute, Boston, and his associates.
The study – designed and funded by AstraZeneca, which also collected and analyzed the data in conjunction with the scientific authors – was conducted at 33 sites in Japan, South Korea, Taiwan, France, Spain, Germany, Australia, the United Kingdom, and the United States. Of the 239 patients who could be evaluated for a response, 123 (51%) showed a partial or complete response. In the subset of patients with known EGFR T790M mutations, the objective response rate was even higher, at 61%.
In contrast, the objective response rate was 21% in the subset of 61 patients who did not have known EGFR T790M mutations, the investigators said (N. Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMoa1411817]). Median progression-free survival was estimated to be 9.6 months in patients with EGFR T790M-positive tumors, although many are still alive and the final survival data have yet to be calculated. In contrast, progression-free survival was 2.8 months in patients with EGFR T790M-negative tumors.
No dose-limiting toxic effects occurred and therefore the maximal efficacy dose with an acceptable level of adverse events cannot be established yet. The most common adverse events were diarrhea (47% of patients), rash (40%), nausea (22%), and decreased appetite (21%). Serious events that were considered to be possibly treatment-related occurred in 6% of patients, and included one fatal case of pneumonia and six cases of pneumonitis-like events that resolved when the drug was discontinued.
These findings demonstrate that “a structurally distinct EGFR inhibitor, one that is selective for the mutated form of EGFR, can be clinically effective and has a side-effect profile that is not dose limiting in the majority of patients in whom T790M-mediated drug resistance has developed,” Dr. Janne and his associates said.
Astra Zeneca is the maker of AZD9291. Dr. Janne reported ties to AstraZeneca, Boehringer Ingelheim, Chugai, Pfizer, Merrimack, Clovis Oncology, Roche, and Gatekeeper, as well as several patents related to genetically targeted cancer treatments; his associates reported ties to numerous industry sources.
A new agent targeting tumors that develop resistance to EGFR tyrosine kinase inhibitors proved to be “highly active” in a phase I clinical trial involving 253 patients with advanced, resistant NSCLC, according to a report published online April 30 in the New England Journal of Medicine.
Most patients who initially respond to EGFR tyrosine kinase inhibitors such as gefitinib, erlotinib, and afatinib develop resistance and show disease progression within 2 years, because the tumors develop additional EGFR mutations, particularly T790M resistance mutations. Preliminary studies suggested that a new oral agent, AZD9291, would target T790M-mediated resistance. In a phase I trial to assess its safety and efficacy, 127 study participants had known EGFR T790M mutations, and the remainder had other or unknown mutations, said Dr. Pasi A. Janne of the Lowe Center for Thoracic Oncology and the Belfer Institute for Applied Cancer Science, Dana-Farber Institute, Boston, and his associates.
The study – designed and funded by AstraZeneca, which also collected and analyzed the data in conjunction with the scientific authors – was conducted at 33 sites in Japan, South Korea, Taiwan, France, Spain, Germany, Australia, the United Kingdom, and the United States. Of the 239 patients who could be evaluated for a response, 123 (51%) showed a partial or complete response. In the subset of patients with known EGFR T790M mutations, the objective response rate was even higher, at 61%.
In contrast, the objective response rate was 21% in the subset of 61 patients who did not have known EGFR T790M mutations, the investigators said (N. Engl. J. Med. 2015 April 30 [doi:10.1056/NEJMoa1411817]). Median progression-free survival was estimated to be 9.6 months in patients with EGFR T790M-positive tumors, although many are still alive and the final survival data have yet to be calculated. In contrast, progression-free survival was 2.8 months in patients with EGFR T790M-negative tumors.
No dose-limiting toxic effects occurred and therefore the maximal efficacy dose with an acceptable level of adverse events cannot be established yet. The most common adverse events were diarrhea (47% of patients), rash (40%), nausea (22%), and decreased appetite (21%). Serious events that were considered to be possibly treatment-related occurred in 6% of patients, and included one fatal case of pneumonia and six cases of pneumonitis-like events that resolved when the drug was discontinued.
These findings demonstrate that “a structurally distinct EGFR inhibitor, one that is selective for the mutated form of EGFR, can be clinically effective and has a side-effect profile that is not dose limiting in the majority of patients in whom T790M-mediated drug resistance has developed,” Dr. Janne and his associates said.
Astra Zeneca is the maker of AZD9291. Dr. Janne reported ties to AstraZeneca, Boehringer Ingelheim, Chugai, Pfizer, Merrimack, Clovis Oncology, Roche, and Gatekeeper, as well as several patents related to genetically targeted cancer treatments; his associates reported ties to numerous industry sources.
Key clinical point: AZD9291 was highly active against advanced NSCLC that had mutated to become resistant to treatment with EGFR tyrosine kinase inhibitors.
Major finding: The objective response rate was 51% in the entire study population and 61% in the subset of patients with known EGFR T790M mutations.
Data source: An industry-funded phase I clinical trial of 253 patients in nine countries whose advanced NSCLC had developed treatment resistance.
Disclosures: This study was funded by Astra Zeneca, maker of AZD9291. Dr. Janne reported ties to AstraZeneca, Boehringer Ingelheim, Chugai, Pfizer, Merrimack, Clovis Oncology, Roche, and Gatekeeper, as well as several patents related to genetically targeted cancer treatments; his associates reported ties to numerous industry sources.
Implantable filter doesn’t cut rate of recurrent PE
Implanting a retrievable filter in the inferior vena cava did not reduce the rate of recurrent pulmonary embolism or mortality in high-risk patients, according to a report published online April 28 in JAMA.
In recent years, there has been a sharp increase in the use of these devices as an add-on to anticoagulant therapy among patients hospitalized for acute PE associated with lower-limb deep or superficial vein thrombosis. Several clinical guidelines advocate this strategy, though others do not, citing the paucity of reliable data concerning both risks and benefits.
The findings in this study “do not support the use of this type of filter in patients who can be treated with anticoagulation alone,” and clinical guidelines recommending this approach should be reexamined, Dr. Patrick Mismetti of the University Hospital of Saint-Etienne, France, and his associates said.
They performed a randomized, open-label clinical study at 17 French medical centers to compare anticoagulation alone against anticoagulation plus implanting a filter to be retrieved 3 months later.
The study participants were 399 adults enrolled during a 6-year period who were deemed at high risk for recurrent PE because of advanced age, active cancer, chronic cardiac or respiratory insufficiency, ischemic stroke with leg paralysis, DVT that was bilateral or affected the iliocaval segment, or signs of right ventricular dysfunction or myocardial injury.
The primary efficacy outcome, recurrent PE within 3 months of hospitalization, developed in 6 of 200 patients assigned to receive an implantable filter (3%) and 3 of the 199 assigned to the control group (1.5%). All but one of these episodes of recurrent PE were fatal. One additional PE developed in each study group between 3 and 6 months. There were no differences between patients who received an inferior vena cava filter and those who did not in the incidence of DVT, major bleeding, or death from any cause at 3 or 6 months, the investigators said (JAMA 2015 April 28 [doi:10.1001/jama.2015.3780]).
Besides failing to prevent recurrent PE, the filter implantation caused access site hematomas in five patients, and the filter itself caused thrombosis formation in three. One patient developed cardiac arrest during the procedure. In addition, retrieval of the device failed in 11 patients because of mechanical problems.
Implanting a retrievable filter in the inferior vena cava did not reduce the rate of recurrent pulmonary embolism or mortality in high-risk patients, according to a report published online April 28 in JAMA.
In recent years, there has been a sharp increase in the use of these devices as an add-on to anticoagulant therapy among patients hospitalized for acute PE associated with lower-limb deep or superficial vein thrombosis. Several clinical guidelines advocate this strategy, though others do not, citing the paucity of reliable data concerning both risks and benefits.
The findings in this study “do not support the use of this type of filter in patients who can be treated with anticoagulation alone,” and clinical guidelines recommending this approach should be reexamined, Dr. Patrick Mismetti of the University Hospital of Saint-Etienne, France, and his associates said.
They performed a randomized, open-label clinical study at 17 French medical centers to compare anticoagulation alone against anticoagulation plus implanting a filter to be retrieved 3 months later.
The study participants were 399 adults enrolled during a 6-year period who were deemed at high risk for recurrent PE because of advanced age, active cancer, chronic cardiac or respiratory insufficiency, ischemic stroke with leg paralysis, DVT that was bilateral or affected the iliocaval segment, or signs of right ventricular dysfunction or myocardial injury.
The primary efficacy outcome, recurrent PE within 3 months of hospitalization, developed in 6 of 200 patients assigned to receive an implantable filter (3%) and 3 of the 199 assigned to the control group (1.5%). All but one of these episodes of recurrent PE were fatal. One additional PE developed in each study group between 3 and 6 months. There were no differences between patients who received an inferior vena cava filter and those who did not in the incidence of DVT, major bleeding, or death from any cause at 3 or 6 months, the investigators said (JAMA 2015 April 28 [doi:10.1001/jama.2015.3780]).
Besides failing to prevent recurrent PE, the filter implantation caused access site hematomas in five patients, and the filter itself caused thrombosis formation in three. One patient developed cardiac arrest during the procedure. In addition, retrieval of the device failed in 11 patients because of mechanical problems.
Implanting a retrievable filter in the inferior vena cava did not reduce the rate of recurrent pulmonary embolism or mortality in high-risk patients, according to a report published online April 28 in JAMA.
In recent years, there has been a sharp increase in the use of these devices as an add-on to anticoagulant therapy among patients hospitalized for acute PE associated with lower-limb deep or superficial vein thrombosis. Several clinical guidelines advocate this strategy, though others do not, citing the paucity of reliable data concerning both risks and benefits.
The findings in this study “do not support the use of this type of filter in patients who can be treated with anticoagulation alone,” and clinical guidelines recommending this approach should be reexamined, Dr. Patrick Mismetti of the University Hospital of Saint-Etienne, France, and his associates said.
They performed a randomized, open-label clinical study at 17 French medical centers to compare anticoagulation alone against anticoagulation plus implanting a filter to be retrieved 3 months later.
The study participants were 399 adults enrolled during a 6-year period who were deemed at high risk for recurrent PE because of advanced age, active cancer, chronic cardiac or respiratory insufficiency, ischemic stroke with leg paralysis, DVT that was bilateral or affected the iliocaval segment, or signs of right ventricular dysfunction or myocardial injury.
The primary efficacy outcome, recurrent PE within 3 months of hospitalization, developed in 6 of 200 patients assigned to receive an implantable filter (3%) and 3 of the 199 assigned to the control group (1.5%). All but one of these episodes of recurrent PE were fatal. One additional PE developed in each study group between 3 and 6 months. There were no differences between patients who received an inferior vena cava filter and those who did not in the incidence of DVT, major bleeding, or death from any cause at 3 or 6 months, the investigators said (JAMA 2015 April 28 [doi:10.1001/jama.2015.3780]).
Besides failing to prevent recurrent PE, the filter implantation caused access site hematomas in five patients, and the filter itself caused thrombosis formation in three. One patient developed cardiac arrest during the procedure. In addition, retrieval of the device failed in 11 patients because of mechanical problems.
FROM JAMA
Key clinical point: Use of a retrievable filter implanted in the inferior vena cava did not reduce the rate of recurrent pulmonary embolism.
Major finding: The primary efficacy outcome, recurrent PE within 3 months of hospitalization, developed in 6 of 200 patients assigned to receive an implantable filter (3%) and 3 of 199 assigned to the control group (1.5%).
Data source: An open-label randomized trial involving 399 adults hospitalized in France for acute PE.
Disclosures: This study was sponsored by the University Hospital of Saint-Etienne and supported by the French Department of Health, Fondation de l’Avenir, and Fondation de France. Vena cava filters were provided free of charge by ALN Implants Chirurgicaux. Dr. Mismetti and his associates reported ties to numerous industry sources.