User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Adding salmeterol to steroids didn’t boost kids’ serious asthma events
Adding the long-acting beta-agonist salmeterol to fluticasone in a fixed-dose combination didn’t increase serious asthma-related events among children aged 4-11 years, according to a report published online Sept. 1 in the New England Journal of Medicine.
After long-acting beta-agonists were introduced as add-on therapy for uncontrolled asthma, two large studies involving adults linked the treatment to an increase in asthma-related death. Other studies found no such association.
The FDA mandated that all four manufacturers of those agents in the United States perform large postmarketing safety trials to establish the noninferiority of the approach. In response, GlaxoSmithKline, the only maker of a long-acting beta-agonist with a pediatric indication (salmeterol), performed this international randomized, double-blind, controlled trial at 567 medical centers in 32 countries, said David A. Stempel, MD, of Respiratory Clinical Development, GSK, Research Triangle Park, N.C., and his associates.
The trial involved 6,208 children aged 4-11 years who had controlled or uncontrolled asthma with a history of exacerbations during the preceding year. The participants were randomly assigned to receive 26 weeks of a lower fixed-dose combination of salmeterol plus fluticasone, a higher fixed-dose combination, a lower dose of fluticasone alone, or a higher dose of fluticasone alone, delivered twice daily via a disk device.
The primary safety endpoint was a composite of death, endotracheal intubation, and hospitalization. No deaths or intubations occurred.
A total of 27 patients taking combined therapy and 21 taking fluticasone alone required hospitalization for asthma (hazard ratio, 1.28). The number of severe asthma exacerbations was 14% lower when salmeterol was added to fluticasone, a nonsignificant difference.
The results demonstrate the noninferiority of the combined therapy, Dr. Stempel and his associates said (N Engl J Med. 2016 Sep 1;375[9]:840-9).
The percentage of children who withdrew from the study because of asthma exacerbations was identical in the two groups (1.1% of each), and the percentage who had a serious adverse event was nearly identical (1.8% vs 1.7%, respectively). The mean percentage of rescue therapy–free days also was similar (83.0% vs 81.9%), as was the mean percentage of days in which asthma was controlled (74.8% vs. 73.4%).
At the conclusion of the study, 88.1% of the fluticasone-plus-salmeterol group had controlled asthma, as did 88.5% of the fluticasone-only group. Meaningful differences between the two treatments could not be identified among various subgroups of patients – defined by age, sex, and race – because the overall number of adverse events was so low, the investigators added.
They cautioned that the trial excluded children who had a history of multiple asthma-related hospitalizations and intubations. Therefore, the findings may not be applicable to patients with very severe asthma, the researchers cautioned.
GlaxoSmithKline sponsored the trial in response to a Food and Drug Administration mandate for large postmarketing safety studies from the marketers of long-acting beta agonist–containing products sold in the United States. Dr. Stempel is an employee of GSK; his associates reported ties to numerous industry sources.
These study findings provide reassuring evidence that combination inhalers are safe for the unusual child with asthma who needs more than inhaled glucocorticoids to control the disease or who has persistent, objectively documented variable airflow obstruction.

|
Dr. Andrew Bush |
But it’s important to emphasize that a combined inhaler is never indicated as first-line preventive therapy in children, because such use is increasingly creeping into practice. And monotherapy with a long-acting beta-agonist in a child should be considered medical negligence.
Andrew Bush, MD, is in the department of respiratory medicine at Royal Brompton Hospital, London. Urs Frey, MD, PhD, is in the department of pediatrics at the University of Basel (Switzerland) Children’s Hospital. They reported having no relevant financial disclosures. Dr. Bush and Dr. Frey made these remarks in an editorial accompanying Dr. Stempel’s report (N Engl J Med. 2016 Sep 1;375[9]:889-91).
These study findings provide reassuring evidence that combination inhalers are safe for the unusual child with asthma who needs more than inhaled glucocorticoids to control the disease or who has persistent, objectively documented variable airflow obstruction.

|
Dr. Andrew Bush |
But it’s important to emphasize that a combined inhaler is never indicated as first-line preventive therapy in children, because such use is increasingly creeping into practice. And monotherapy with a long-acting beta-agonist in a child should be considered medical negligence.
Andrew Bush, MD, is in the department of respiratory medicine at Royal Brompton Hospital, London. Urs Frey, MD, PhD, is in the department of pediatrics at the University of Basel (Switzerland) Children’s Hospital. They reported having no relevant financial disclosures. Dr. Bush and Dr. Frey made these remarks in an editorial accompanying Dr. Stempel’s report (N Engl J Med. 2016 Sep 1;375[9]:889-91).
These study findings provide reassuring evidence that combination inhalers are safe for the unusual child with asthma who needs more than inhaled glucocorticoids to control the disease or who has persistent, objectively documented variable airflow obstruction.

|
Dr. Andrew Bush |
But it’s important to emphasize that a combined inhaler is never indicated as first-line preventive therapy in children, because such use is increasingly creeping into practice. And monotherapy with a long-acting beta-agonist in a child should be considered medical negligence.
Andrew Bush, MD, is in the department of respiratory medicine at Royal Brompton Hospital, London. Urs Frey, MD, PhD, is in the department of pediatrics at the University of Basel (Switzerland) Children’s Hospital. They reported having no relevant financial disclosures. Dr. Bush and Dr. Frey made these remarks in an editorial accompanying Dr. Stempel’s report (N Engl J Med. 2016 Sep 1;375[9]:889-91).
Adding the long-acting beta-agonist salmeterol to fluticasone in a fixed-dose combination didn’t increase serious asthma-related events among children aged 4-11 years, according to a report published online Sept. 1 in the New England Journal of Medicine.
After long-acting beta-agonists were introduced as add-on therapy for uncontrolled asthma, two large studies involving adults linked the treatment to an increase in asthma-related death. Other studies found no such association.
The FDA mandated that all four manufacturers of those agents in the United States perform large postmarketing safety trials to establish the noninferiority of the approach. In response, GlaxoSmithKline, the only maker of a long-acting beta-agonist with a pediatric indication (salmeterol), performed this international randomized, double-blind, controlled trial at 567 medical centers in 32 countries, said David A. Stempel, MD, of Respiratory Clinical Development, GSK, Research Triangle Park, N.C., and his associates.
The trial involved 6,208 children aged 4-11 years who had controlled or uncontrolled asthma with a history of exacerbations during the preceding year. The participants were randomly assigned to receive 26 weeks of a lower fixed-dose combination of salmeterol plus fluticasone, a higher fixed-dose combination, a lower dose of fluticasone alone, or a higher dose of fluticasone alone, delivered twice daily via a disk device.
The primary safety endpoint was a composite of death, endotracheal intubation, and hospitalization. No deaths or intubations occurred.
A total of 27 patients taking combined therapy and 21 taking fluticasone alone required hospitalization for asthma (hazard ratio, 1.28). The number of severe asthma exacerbations was 14% lower when salmeterol was added to fluticasone, a nonsignificant difference.
The results demonstrate the noninferiority of the combined therapy, Dr. Stempel and his associates said (N Engl J Med. 2016 Sep 1;375[9]:840-9).
The percentage of children who withdrew from the study because of asthma exacerbations was identical in the two groups (1.1% of each), and the percentage who had a serious adverse event was nearly identical (1.8% vs 1.7%, respectively). The mean percentage of rescue therapy–free days also was similar (83.0% vs 81.9%), as was the mean percentage of days in which asthma was controlled (74.8% vs. 73.4%).
At the conclusion of the study, 88.1% of the fluticasone-plus-salmeterol group had controlled asthma, as did 88.5% of the fluticasone-only group. Meaningful differences between the two treatments could not be identified among various subgroups of patients – defined by age, sex, and race – because the overall number of adverse events was so low, the investigators added.
They cautioned that the trial excluded children who had a history of multiple asthma-related hospitalizations and intubations. Therefore, the findings may not be applicable to patients with very severe asthma, the researchers cautioned.
GlaxoSmithKline sponsored the trial in response to a Food and Drug Administration mandate for large postmarketing safety studies from the marketers of long-acting beta agonist–containing products sold in the United States. Dr. Stempel is an employee of GSK; his associates reported ties to numerous industry sources.
Adding the long-acting beta-agonist salmeterol to fluticasone in a fixed-dose combination didn’t increase serious asthma-related events among children aged 4-11 years, according to a report published online Sept. 1 in the New England Journal of Medicine.
After long-acting beta-agonists were introduced as add-on therapy for uncontrolled asthma, two large studies involving adults linked the treatment to an increase in asthma-related death. Other studies found no such association.
The FDA mandated that all four manufacturers of those agents in the United States perform large postmarketing safety trials to establish the noninferiority of the approach. In response, GlaxoSmithKline, the only maker of a long-acting beta-agonist with a pediatric indication (salmeterol), performed this international randomized, double-blind, controlled trial at 567 medical centers in 32 countries, said David A. Stempel, MD, of Respiratory Clinical Development, GSK, Research Triangle Park, N.C., and his associates.
The trial involved 6,208 children aged 4-11 years who had controlled or uncontrolled asthma with a history of exacerbations during the preceding year. The participants were randomly assigned to receive 26 weeks of a lower fixed-dose combination of salmeterol plus fluticasone, a higher fixed-dose combination, a lower dose of fluticasone alone, or a higher dose of fluticasone alone, delivered twice daily via a disk device.
The primary safety endpoint was a composite of death, endotracheal intubation, and hospitalization. No deaths or intubations occurred.
A total of 27 patients taking combined therapy and 21 taking fluticasone alone required hospitalization for asthma (hazard ratio, 1.28). The number of severe asthma exacerbations was 14% lower when salmeterol was added to fluticasone, a nonsignificant difference.
The results demonstrate the noninferiority of the combined therapy, Dr. Stempel and his associates said (N Engl J Med. 2016 Sep 1;375[9]:840-9).
The percentage of children who withdrew from the study because of asthma exacerbations was identical in the two groups (1.1% of each), and the percentage who had a serious adverse event was nearly identical (1.8% vs 1.7%, respectively). The mean percentage of rescue therapy–free days also was similar (83.0% vs 81.9%), as was the mean percentage of days in which asthma was controlled (74.8% vs. 73.4%).
At the conclusion of the study, 88.1% of the fluticasone-plus-salmeterol group had controlled asthma, as did 88.5% of the fluticasone-only group. Meaningful differences between the two treatments could not be identified among various subgroups of patients – defined by age, sex, and race – because the overall number of adverse events was so low, the investigators added.
They cautioned that the trial excluded children who had a history of multiple asthma-related hospitalizations and intubations. Therefore, the findings may not be applicable to patients with very severe asthma, the researchers cautioned.
GlaxoSmithKline sponsored the trial in response to a Food and Drug Administration mandate for large postmarketing safety studies from the marketers of long-acting beta agonist–containing products sold in the United States. Dr. Stempel is an employee of GSK; his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Adding salmeterol to fluticasone therapy didn’t increase serious asthma-related events among children.
Major finding: 27 patients taking combined therapy and 21 taking fluticasone alone required hospitalization for asthma (HR, 1.28).
Data source: A 26-week international randomized, double-blind trial involving 6,208 patients aged 4-11 years.
Disclosures: GlaxoSmithKline sponsored the trial in response to a Food and Drug Administration mandate for large postmarketing safety studies from the marketers of long-acting beta agonist–containing products sold in the United States. Dr. Stempel is an employee of GSK; his associates reported ties to numerous industry sources.
Adding formoterol to budesonide does not increase serious asthma events
Adding formoterol to budesonide in a fixed-dose combination does not increase serous asthma-related events in adolescents and adults, according to a report published online Sept. 1 in the New England Journal of Medicine.
This finding from a multicenter randomized double-blind clinical trial involving 11,693 patients should allay safety concerns about adding long-acting beta-agonists to inhaled glucocorticoids in moderate to severe asthma. Previously, two large studies linked such additive therapy to increased asthma-related deaths and other serious outcomes, but other clinical trials and numerous meta-analyses found no such increase.
In 2009, the Food and Drug Administration mandated that the four manufacturers of long-acting beta-agonists available in the United States conduct postmarketing safety analyses of these agents. The current trial is AstraZeneca’s response to the mandate, said Stephen P. Peters, MD, PhD, of Wake Forest University, Winston-Salem N.C., and his associates.
They assessed patients aged 12 years and older who had taken daily asthma medication for at least 1 year before enrollment and had a history of at least one exacerbation during that year. These participants were enrolled at 534 medical centers in 25 countries during 2011-2015 and randomly assigned to receive either budesonide plus formoterol (5,846 patients) or budesonide alone (5,847 patients) through an inhaler twice daily for 26 weeks. The primary endpoint was a composite of asthma-related death, intubation, and hospitalization.
A total of 43 patients in the combined-therapy group had 49 serious asthma-related events, while 40 patients in the budesonide-only group had 45 such events. This is a nonsignificant difference and establishes the noninferiority of the combined treatment regarding this outcome, the investigators said (N Engl J Med. 2016 Sept 1. doi: 10.1056/NEJMoa1511190).
In addition, 539 (9.2%) of the patients in the combined-therapy group reported 637 asthma exacerbations, while 633 in the budesonide-only group had 762 exacerbations. Thus, the risk of having an asthma exacerbation was 16.5% lower with combined therapy (HR, 0.84).
Both study groups had a clinically relevant improvement in asthma control as measured by the ACQ-6, and the combined therapy yielded a significantly greater benefit. The percentage of patients who had a clinically relevant improvement in asthma control at the conclusion of treatment also favored budesonide plus formoterol (58.7% vs. 54.4%). And the combined-therapy group also had a greater mean number of symptom-free days, had fewer night-time awakenings, and used fewer doses of rescue medications, Dr. Peters and his associates said.
Given that asthma-related deaths are rare, none of the four individual manufacturer-sponsored postmarketing studies required by the FDA can be powered for a separate analysis of that endpoint. “Any between-group differences in asthma-related death will need to be evaluated in the context of pooled data from the four studies, once they are all completed,” the investigators added.
Dr. Peters and his associates reported ties to numerous industry sources.
Adding formoterol to budesonide in a fixed-dose combination does not increase serous asthma-related events in adolescents and adults, according to a report published online Sept. 1 in the New England Journal of Medicine.
This finding from a multicenter randomized double-blind clinical trial involving 11,693 patients should allay safety concerns about adding long-acting beta-agonists to inhaled glucocorticoids in moderate to severe asthma. Previously, two large studies linked such additive therapy to increased asthma-related deaths and other serious outcomes, but other clinical trials and numerous meta-analyses found no such increase.
In 2009, the Food and Drug Administration mandated that the four manufacturers of long-acting beta-agonists available in the United States conduct postmarketing safety analyses of these agents. The current trial is AstraZeneca’s response to the mandate, said Stephen P. Peters, MD, PhD, of Wake Forest University, Winston-Salem N.C., and his associates.
They assessed patients aged 12 years and older who had taken daily asthma medication for at least 1 year before enrollment and had a history of at least one exacerbation during that year. These participants were enrolled at 534 medical centers in 25 countries during 2011-2015 and randomly assigned to receive either budesonide plus formoterol (5,846 patients) or budesonide alone (5,847 patients) through an inhaler twice daily for 26 weeks. The primary endpoint was a composite of asthma-related death, intubation, and hospitalization.
A total of 43 patients in the combined-therapy group had 49 serious asthma-related events, while 40 patients in the budesonide-only group had 45 such events. This is a nonsignificant difference and establishes the noninferiority of the combined treatment regarding this outcome, the investigators said (N Engl J Med. 2016 Sept 1. doi: 10.1056/NEJMoa1511190).
In addition, 539 (9.2%) of the patients in the combined-therapy group reported 637 asthma exacerbations, while 633 in the budesonide-only group had 762 exacerbations. Thus, the risk of having an asthma exacerbation was 16.5% lower with combined therapy (HR, 0.84).
Both study groups had a clinically relevant improvement in asthma control as measured by the ACQ-6, and the combined therapy yielded a significantly greater benefit. The percentage of patients who had a clinically relevant improvement in asthma control at the conclusion of treatment also favored budesonide plus formoterol (58.7% vs. 54.4%). And the combined-therapy group also had a greater mean number of symptom-free days, had fewer night-time awakenings, and used fewer doses of rescue medications, Dr. Peters and his associates said.
Given that asthma-related deaths are rare, none of the four individual manufacturer-sponsored postmarketing studies required by the FDA can be powered for a separate analysis of that endpoint. “Any between-group differences in asthma-related death will need to be evaluated in the context of pooled data from the four studies, once they are all completed,” the investigators added.
Dr. Peters and his associates reported ties to numerous industry sources.
Adding formoterol to budesonide in a fixed-dose combination does not increase serous asthma-related events in adolescents and adults, according to a report published online Sept. 1 in the New England Journal of Medicine.
This finding from a multicenter randomized double-blind clinical trial involving 11,693 patients should allay safety concerns about adding long-acting beta-agonists to inhaled glucocorticoids in moderate to severe asthma. Previously, two large studies linked such additive therapy to increased asthma-related deaths and other serious outcomes, but other clinical trials and numerous meta-analyses found no such increase.
In 2009, the Food and Drug Administration mandated that the four manufacturers of long-acting beta-agonists available in the United States conduct postmarketing safety analyses of these agents. The current trial is AstraZeneca’s response to the mandate, said Stephen P. Peters, MD, PhD, of Wake Forest University, Winston-Salem N.C., and his associates.
They assessed patients aged 12 years and older who had taken daily asthma medication for at least 1 year before enrollment and had a history of at least one exacerbation during that year. These participants were enrolled at 534 medical centers in 25 countries during 2011-2015 and randomly assigned to receive either budesonide plus formoterol (5,846 patients) or budesonide alone (5,847 patients) through an inhaler twice daily for 26 weeks. The primary endpoint was a composite of asthma-related death, intubation, and hospitalization.
A total of 43 patients in the combined-therapy group had 49 serious asthma-related events, while 40 patients in the budesonide-only group had 45 such events. This is a nonsignificant difference and establishes the noninferiority of the combined treatment regarding this outcome, the investigators said (N Engl J Med. 2016 Sept 1. doi: 10.1056/NEJMoa1511190).
In addition, 539 (9.2%) of the patients in the combined-therapy group reported 637 asthma exacerbations, while 633 in the budesonide-only group had 762 exacerbations. Thus, the risk of having an asthma exacerbation was 16.5% lower with combined therapy (HR, 0.84).
Both study groups had a clinically relevant improvement in asthma control as measured by the ACQ-6, and the combined therapy yielded a significantly greater benefit. The percentage of patients who had a clinically relevant improvement in asthma control at the conclusion of treatment also favored budesonide plus formoterol (58.7% vs. 54.4%). And the combined-therapy group also had a greater mean number of symptom-free days, had fewer night-time awakenings, and used fewer doses of rescue medications, Dr. Peters and his associates said.
Given that asthma-related deaths are rare, none of the four individual manufacturer-sponsored postmarketing studies required by the FDA can be powered for a separate analysis of that endpoint. “Any between-group differences in asthma-related death will need to be evaluated in the context of pooled data from the four studies, once they are all completed,” the investigators added.
Dr. Peters and his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Adding formoterol to budesonide in a fixed-dose combination does not increase serious asthma-related events in adolescents and adults.
Major finding: The risk of having an asthma exacerbation was 16.5% lower with combined therapy than with budesonide alone (HR, 0.84).
Data source: A 26-week multicenter randomized double-blind trial involving 11,693 asthma patients aged 12 and older.
Disclosures: This trial was sponsored by AstraZeneca in response to an FDA mandate for large postmarketing safety studies from the four marketers of long-acting beta-agonist-containing products sold in the United States. Dr. Peters and his associates reported ties to numerous industry sources.
PAI-1 modifications, early-life LRIs increase asthma risk
A genetic modification of the plasminogen activator inhibitor-1 gene in conjunction with lower respiratory infections during early life was associated with increased risk of asthma, morbidities, and reduced lung function, according to Seong H. Cho, MD, and his associates.
A history of respiratory syncytial virus (RSV) and a history of other lower respiratory infections (LRIs) before the age of 2 were independently associated with asthma in Latino people aged 8-21, with odd ratios of 9.9 and 9.1, respectively, while PAI-1 was not independently associated. In combination, the OR for PAI-1/RSV increased to 17.7, and the OR for PAI-1/other LRIs increased to 11.7.
Lung function was also adversely affected by the joint effect of PAI-1 and early life infection. In patients with PAI-1/LRI, forced expiratory volume in 1 second (FEV1) percent predicted and FEV1/forced vital capacity (FVC) percent predicted were significantly less than in the control group. Similar but less significant results were seen in the PAI-1/RSV group. Recurring hospitalizations were also significantly more likely in the PAI-1/RSV group, with an OR of 3.1.
“Further prospective studies are needed to replicate our RSV-genotype findings in other non-Latino populations, and determine if PAI-1 variants may serve as a biomarker of risk, which may provide impetus for clinical trials of primary prevention of asthma. In the interim, PAI-1 genotype in combination with significant LRI identifies individuals at increased risk of developing asthma,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0157848).
A genetic modification of the plasminogen activator inhibitor-1 gene in conjunction with lower respiratory infections during early life was associated with increased risk of asthma, morbidities, and reduced lung function, according to Seong H. Cho, MD, and his associates.
A history of respiratory syncytial virus (RSV) and a history of other lower respiratory infections (LRIs) before the age of 2 were independently associated with asthma in Latino people aged 8-21, with odd ratios of 9.9 and 9.1, respectively, while PAI-1 was not independently associated. In combination, the OR for PAI-1/RSV increased to 17.7, and the OR for PAI-1/other LRIs increased to 11.7.
Lung function was also adversely affected by the joint effect of PAI-1 and early life infection. In patients with PAI-1/LRI, forced expiratory volume in 1 second (FEV1) percent predicted and FEV1/forced vital capacity (FVC) percent predicted were significantly less than in the control group. Similar but less significant results were seen in the PAI-1/RSV group. Recurring hospitalizations were also significantly more likely in the PAI-1/RSV group, with an OR of 3.1.
“Further prospective studies are needed to replicate our RSV-genotype findings in other non-Latino populations, and determine if PAI-1 variants may serve as a biomarker of risk, which may provide impetus for clinical trials of primary prevention of asthma. In the interim, PAI-1 genotype in combination with significant LRI identifies individuals at increased risk of developing asthma,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0157848).
A genetic modification of the plasminogen activator inhibitor-1 gene in conjunction with lower respiratory infections during early life was associated with increased risk of asthma, morbidities, and reduced lung function, according to Seong H. Cho, MD, and his associates.
A history of respiratory syncytial virus (RSV) and a history of other lower respiratory infections (LRIs) before the age of 2 were independently associated with asthma in Latino people aged 8-21, with odd ratios of 9.9 and 9.1, respectively, while PAI-1 was not independently associated. In combination, the OR for PAI-1/RSV increased to 17.7, and the OR for PAI-1/other LRIs increased to 11.7.
Lung function was also adversely affected by the joint effect of PAI-1 and early life infection. In patients with PAI-1/LRI, forced expiratory volume in 1 second (FEV1) percent predicted and FEV1/forced vital capacity (FVC) percent predicted were significantly less than in the control group. Similar but less significant results were seen in the PAI-1/RSV group. Recurring hospitalizations were also significantly more likely in the PAI-1/RSV group, with an OR of 3.1.
“Further prospective studies are needed to replicate our RSV-genotype findings in other non-Latino populations, and determine if PAI-1 variants may serve as a biomarker of risk, which may provide impetus for clinical trials of primary prevention of asthma. In the interim, PAI-1 genotype in combination with significant LRI identifies individuals at increased risk of developing asthma,” the investigators wrote.
Find the full study in PLoS One (doi: 10.1371/journal.pone.0157848).
FROM PLOS ONE
Peanut-allergic preschoolers benefit from oral immunotherapy
Early intervention oral immunotherapy (OIT) improved a majority of peanut-allergic preschoolers’ ability to eat peanut protein with no reaction, based on data from a randomized trial of 40 children aged 9-36 months.
“We postulated that targeting newly diagnosed young peanut-allergic children would provide the best opportunity to enhance the clinical effectiveness of OIT as an immunomodulatory and disease-modifying treatment by interrupting allergic priming before its full maturation,” wrote Brian P. Vickery, MD, of the University of North Carolina, Chapel Hill, and his colleagues.
The children received doses of either 300 mg/day or 3,000 mg/day of peanut protein for an average of 29 months. Overall, 78% of the 37 children in the intent-to-treat analysis met the primary endpoint of unresponsiveness to peanut protein 4 weeks after discontinuing oral immunotherapy (85% of the 300-mg group and 71% of the 3,000-mg group). Peanut-specific levels of IgE dropped significantly in the treatment group, and the treated children were 19 times more likely to eat 5 g of peanut protein without reaction than were 154 untreated matched controls.
Three children discontinued the study because of treatment-related adverse events, but no treatment-related severe adverse events, hospitalizations, or deaths were reported.
The findings suggest “that allergic responses may be more easily and durably corrected in young children, and that in this context, relatively low OIT doses are sufficiently potent in suppressing IgE responses and stimulating IgG4 production,” the researchers said.
Find the full study here in the Journal of Allergy and Clinical Immunology (2016 Aug. doi: 10.1016/j.jaci.2016.05.027).
Early intervention oral immunotherapy (OIT) improved a majority of peanut-allergic preschoolers’ ability to eat peanut protein with no reaction, based on data from a randomized trial of 40 children aged 9-36 months.
“We postulated that targeting newly diagnosed young peanut-allergic children would provide the best opportunity to enhance the clinical effectiveness of OIT as an immunomodulatory and disease-modifying treatment by interrupting allergic priming before its full maturation,” wrote Brian P. Vickery, MD, of the University of North Carolina, Chapel Hill, and his colleagues.
The children received doses of either 300 mg/day or 3,000 mg/day of peanut protein for an average of 29 months. Overall, 78% of the 37 children in the intent-to-treat analysis met the primary endpoint of unresponsiveness to peanut protein 4 weeks after discontinuing oral immunotherapy (85% of the 300-mg group and 71% of the 3,000-mg group). Peanut-specific levels of IgE dropped significantly in the treatment group, and the treated children were 19 times more likely to eat 5 g of peanut protein without reaction than were 154 untreated matched controls.
Three children discontinued the study because of treatment-related adverse events, but no treatment-related severe adverse events, hospitalizations, or deaths were reported.
The findings suggest “that allergic responses may be more easily and durably corrected in young children, and that in this context, relatively low OIT doses are sufficiently potent in suppressing IgE responses and stimulating IgG4 production,” the researchers said.
Find the full study here in the Journal of Allergy and Clinical Immunology (2016 Aug. doi: 10.1016/j.jaci.2016.05.027).
Early intervention oral immunotherapy (OIT) improved a majority of peanut-allergic preschoolers’ ability to eat peanut protein with no reaction, based on data from a randomized trial of 40 children aged 9-36 months.
“We postulated that targeting newly diagnosed young peanut-allergic children would provide the best opportunity to enhance the clinical effectiveness of OIT as an immunomodulatory and disease-modifying treatment by interrupting allergic priming before its full maturation,” wrote Brian P. Vickery, MD, of the University of North Carolina, Chapel Hill, and his colleagues.
The children received doses of either 300 mg/day or 3,000 mg/day of peanut protein for an average of 29 months. Overall, 78% of the 37 children in the intent-to-treat analysis met the primary endpoint of unresponsiveness to peanut protein 4 weeks after discontinuing oral immunotherapy (85% of the 300-mg group and 71% of the 3,000-mg group). Peanut-specific levels of IgE dropped significantly in the treatment group, and the treated children were 19 times more likely to eat 5 g of peanut protein without reaction than were 154 untreated matched controls.
Three children discontinued the study because of treatment-related adverse events, but no treatment-related severe adverse events, hospitalizations, or deaths were reported.
The findings suggest “that allergic responses may be more easily and durably corrected in young children, and that in this context, relatively low OIT doses are sufficiently potent in suppressing IgE responses and stimulating IgG4 production,” the researchers said.
Find the full study here in the Journal of Allergy and Clinical Immunology (2016 Aug. doi: 10.1016/j.jaci.2016.05.027).
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
Antibiotics overprescribed during asthma-related hospitalizations
Antibiotics are overprescribed in asthma-related hospitalizations, even though guidelines recommend against prescribing antibiotics during exacerbations of asthma in the absence of concurrent infection, reported Peter K. Lindenauer, MD, MSc, of Baystate Medical Center in Springfield, Mass., and his colleagues.
They examined the hospitalization records of 51,951 individuals admitted to 577 hospitals in the United States between 2013 and 2014 with a principal diagnosis of either asthma or acute respiratory failure combined with asthma as a secondary diagnosis. Each patient type and the timing of antibiotic therapy was noted.
A total of 30,226 of the 51,951 patients (58.2%) were prescribed antibiotics at some point during their hospitalization, while 21,248 (40.9%) were prescribed antibiotics on the first day of hospitalization, without “documentation of an indication for antibiotic therapy.”
Macrolides were most commonly prescribed, given to 9,633 (18.5%) of patients, followed by quinolones (8,632, 16.1%), third-generation cephalosporins (4,420, 8.5%), and tetracyclines (1,858, 3.6%). After adjustment for risk variables, chronic obstructive asthma hospitalizations were found to be those most highly associated with receiving antibiotics (odds ratio 1.6, 95% confidence interval 1.5-1.7).
“Possible explanations for this high rate of potentially inappropriate treatment include the challenge of differentiating bacterial from nonbacterial infections, distinguishing asthma from chronic obstructive pulmonary disease in the acute care setting, and gaps in knowledge about the benefits of antibiotic therapy,” the authors posited, adding that these findings “suggest a significant opportunity to improve patient safety, reduce the spread of resistance, and lower spending through greater adherence to guideline recommendations.”
The National Heart, Lung, and Blood Institute and Veterans Affairs Health Services Research and Development funded the study. Dr. Lindenauer and his coauthors did not report any relevant financial disclosures.
Antibiotics are overprescribed in asthma-related hospitalizations, even though guidelines recommend against prescribing antibiotics during exacerbations of asthma in the absence of concurrent infection, reported Peter K. Lindenauer, MD, MSc, of Baystate Medical Center in Springfield, Mass., and his colleagues.
They examined the hospitalization records of 51,951 individuals admitted to 577 hospitals in the United States between 2013 and 2014 with a principal diagnosis of either asthma or acute respiratory failure combined with asthma as a secondary diagnosis. Each patient type and the timing of antibiotic therapy was noted.
A total of 30,226 of the 51,951 patients (58.2%) were prescribed antibiotics at some point during their hospitalization, while 21,248 (40.9%) were prescribed antibiotics on the first day of hospitalization, without “documentation of an indication for antibiotic therapy.”
Macrolides were most commonly prescribed, given to 9,633 (18.5%) of patients, followed by quinolones (8,632, 16.1%), third-generation cephalosporins (4,420, 8.5%), and tetracyclines (1,858, 3.6%). After adjustment for risk variables, chronic obstructive asthma hospitalizations were found to be those most highly associated with receiving antibiotics (odds ratio 1.6, 95% confidence interval 1.5-1.7).
“Possible explanations for this high rate of potentially inappropriate treatment include the challenge of differentiating bacterial from nonbacterial infections, distinguishing asthma from chronic obstructive pulmonary disease in the acute care setting, and gaps in knowledge about the benefits of antibiotic therapy,” the authors posited, adding that these findings “suggest a significant opportunity to improve patient safety, reduce the spread of resistance, and lower spending through greater adherence to guideline recommendations.”
The National Heart, Lung, and Blood Institute and Veterans Affairs Health Services Research and Development funded the study. Dr. Lindenauer and his coauthors did not report any relevant financial disclosures.
Antibiotics are overprescribed in asthma-related hospitalizations, even though guidelines recommend against prescribing antibiotics during exacerbations of asthma in the absence of concurrent infection, reported Peter K. Lindenauer, MD, MSc, of Baystate Medical Center in Springfield, Mass., and his colleagues.
They examined the hospitalization records of 51,951 individuals admitted to 577 hospitals in the United States between 2013 and 2014 with a principal diagnosis of either asthma or acute respiratory failure combined with asthma as a secondary diagnosis. Each patient type and the timing of antibiotic therapy was noted.
A total of 30,226 of the 51,951 patients (58.2%) were prescribed antibiotics at some point during their hospitalization, while 21,248 (40.9%) were prescribed antibiotics on the first day of hospitalization, without “documentation of an indication for antibiotic therapy.”
Macrolides were most commonly prescribed, given to 9,633 (18.5%) of patients, followed by quinolones (8,632, 16.1%), third-generation cephalosporins (4,420, 8.5%), and tetracyclines (1,858, 3.6%). After adjustment for risk variables, chronic obstructive asthma hospitalizations were found to be those most highly associated with receiving antibiotics (odds ratio 1.6, 95% confidence interval 1.5-1.7).
“Possible explanations for this high rate of potentially inappropriate treatment include the challenge of differentiating bacterial from nonbacterial infections, distinguishing asthma from chronic obstructive pulmonary disease in the acute care setting, and gaps in knowledge about the benefits of antibiotic therapy,” the authors posited, adding that these findings “suggest a significant opportunity to improve patient safety, reduce the spread of resistance, and lower spending through greater adherence to guideline recommendations.”
The National Heart, Lung, and Blood Institute and Veterans Affairs Health Services Research and Development funded the study. Dr. Lindenauer and his coauthors did not report any relevant financial disclosures.
FROM JAMA INTERNAL MEDICINE
Key clinical point: Antibiotics are overprescribed in asthma-related hospitalizations.
Major finding: Among patients hospitalized for asthma, 58.2% had received antibiotics without any documentation or indication for such therapy.
Data source: Retrospective study of 51,951 patients in 577 U.S. hospitals from 2013 to 2014.
Disclosures: The National Heart, Lung, and Blood Institute and Veterans Affairs Health Services Research and Development funded the study. The researchers reported no relevant financial disclosures.
CPAP fell short for preventing cardiovascular events
Adults with moderate to severe sleep apnea and coronary or cerebrovascular disease had about the same frequency of cardiovascular events whether they received continuous positive airway pressure (CPAP) therapy or usual care alone, according to a large randomized trial.
But CPAP was used for only 3.3 hours per night by these patients and might have been “insufficient to provide the level of effect on cardiovascular outcomes that had been hypothesized,” Dr. Doug McEvoy of the Adelaide Institute for Sleep Health, Flinders University, Adelaide, Australia and his associates reported at the annual congress of the European Society of Cardiology. Their study was simultaneously published in the New England Journal of Medicine (N Engl J Med. 2016 Aug 28. doi: 10.1056/NEJMoa1606599).
Notably, CPAP did show a trend toward significance in a prespecified subgroup analysis that matched 561 patients who used CPAP for a longer period – more than 4 hours a night – with the same number of controls (hazard ratio, 0.8; 95% CI, 0.6 to 1.1; P = .1). Dr. McEvoy discussed the implications of prolonged CPAP use in a video interview with Bruce Jancin, our reporter at the ESC Congress in Rome.
Obstructive sleep apnea causes episodic hypoxemia, sympathetic nervous system activation; intrathoracic pressure swings strain the heart and great vessels, and increases markers of oxidative stress, hypercoagulation, and inflammation. Randomized trials have linked CPAP therapy to lower systolic blood pressure measures and improved endothelial function and insulin sensitivity. Observational studies suggest that CPAP might help prevent cardiovascular events and death if used consistently, the investigators noted.
Because cardiovascular disease and obstructive sleep apnea often co-occur, the researchers carried out a secondary prevention trial, Sleep Apnea Cardiovascular Endpoints (SAVE), to quantify rates of major cardiovascular events among 2,717 adults aged 45-75 years with obstructive sleep apnea and established coronary or cerebrovascular disease. Patients were randomly assigned to receive CPAP therapy plus usual care, or usual care alone. The primary endpoint was a composite of cardiovascular death, myocardial infarction, stroke, or hospitalization from unstable angina, transient ischemic attack, or heart failure. The researchers also looked at other cardiovascular outcomes, snoring symptoms, mood, daytime sleepiness, and health-related quality of life. They used a 1-week run-in period of sham CPAP (administered at subtherapeutic pressure) to ensure what they considered an adequate level of adherence.
The average apnea-hypopnea index (that is, the average number of apnea or hypopnea events recorded per hour) was 29 at baseline and 3.7 after initiating CPAP, the investigators said. At a mean of 3.7 years of follow-up, 17% of CPAP users (220 patients) and 15.4% of controls had a cardiovascular event, for a hazard ratio of 1.1 (95% confidence interval, 0.9 to 1.3; P = 0.3).
Not only did CPAP fail to meet the composite primary endpoint, but it did not significantly affect any cause-specific cardiovascular outcome, the researchers said. However, CPAP users did improve significantly more than controls on measures of daytime sleepiness (the Epworth Sleepiness Scale), anxiety and depression (Hospital Anxiety and Depression Scale), self-reported physical and mental health (Short-Form Health Survey), and quality of life (European Quality of Life-5 Dimensions questionnaire). They also missed fewer days of work than did controls.
Study funders included the National Health and Medical Research Council of Australia, Respironics Sleep and Respiratory Research Foundation, and Phillips Respironics. Dr. McEvoy reported receiving research equipment for the study from AirLiquide. Several coinvestigators reported other ties to industry.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
This trial raises several issues. One major issue is whether the results were negative because obstructive sleep apnea does not have clinically significant adverse cardiovascular effects or because the patients did not use CPAP for a long enough duration each night to derive cardiovascular benefits. Given the substantial human and animal data that have consistently documented links between obstructive sleep apnea and cardiovascular health, we suspect that mean CPAP duration may have been inadequate at 3.3 hours per night, which is probably less than half the time the patient was asleep.
What do these results mean for clinical practice? We believe that symptomatic patients with obstructive sleep apnea should be offered a trial of CPAP therapy. However, on the basis of results from the SAVE trial, prescribing CPAP with the sole purpose of reducing future cardiovascular events in asymptomatic patients with obstructive sleep apnea and established cardiovascular disease cannot be recommended. Ongoing clinical trials will shed further light on the effects of CPAP therapy in nonsleepy patients with obstructive sleep apnea and acute coronary syndromes.
Babak Mokhlesi, MD, is with the Sleep Disorders Center at the University of Chicago. Najib Ayas, MD, is with the Sleep Disorders Program at the University of British Columbia, Vancouver. The remarks are excerpted from their editorial (N Engl J Med. 2016 Aug 28. doi: 10.1056/NEJMe1609704).
This trial raises several issues. One major issue is whether the results were negative because obstructive sleep apnea does not have clinically significant adverse cardiovascular effects or because the patients did not use CPAP for a long enough duration each night to derive cardiovascular benefits. Given the substantial human and animal data that have consistently documented links between obstructive sleep apnea and cardiovascular health, we suspect that mean CPAP duration may have been inadequate at 3.3 hours per night, which is probably less than half the time the patient was asleep.
What do these results mean for clinical practice? We believe that symptomatic patients with obstructive sleep apnea should be offered a trial of CPAP therapy. However, on the basis of results from the SAVE trial, prescribing CPAP with the sole purpose of reducing future cardiovascular events in asymptomatic patients with obstructive sleep apnea and established cardiovascular disease cannot be recommended. Ongoing clinical trials will shed further light on the effects of CPAP therapy in nonsleepy patients with obstructive sleep apnea and acute coronary syndromes.
Babak Mokhlesi, MD, is with the Sleep Disorders Center at the University of Chicago. Najib Ayas, MD, is with the Sleep Disorders Program at the University of British Columbia, Vancouver. The remarks are excerpted from their editorial (N Engl J Med. 2016 Aug 28. doi: 10.1056/NEJMe1609704).
This trial raises several issues. One major issue is whether the results were negative because obstructive sleep apnea does not have clinically significant adverse cardiovascular effects or because the patients did not use CPAP for a long enough duration each night to derive cardiovascular benefits. Given the substantial human and animal data that have consistently documented links between obstructive sleep apnea and cardiovascular health, we suspect that mean CPAP duration may have been inadequate at 3.3 hours per night, which is probably less than half the time the patient was asleep.
What do these results mean for clinical practice? We believe that symptomatic patients with obstructive sleep apnea should be offered a trial of CPAP therapy. However, on the basis of results from the SAVE trial, prescribing CPAP with the sole purpose of reducing future cardiovascular events in asymptomatic patients with obstructive sleep apnea and established cardiovascular disease cannot be recommended. Ongoing clinical trials will shed further light on the effects of CPAP therapy in nonsleepy patients with obstructive sleep apnea and acute coronary syndromes.
Babak Mokhlesi, MD, is with the Sleep Disorders Center at the University of Chicago. Najib Ayas, MD, is with the Sleep Disorders Program at the University of British Columbia, Vancouver. The remarks are excerpted from their editorial (N Engl J Med. 2016 Aug 28. doi: 10.1056/NEJMe1609704).
Adults with moderate to severe sleep apnea and coronary or cerebrovascular disease had about the same frequency of cardiovascular events whether they received continuous positive airway pressure (CPAP) therapy or usual care alone, according to a large randomized trial.
But CPAP was used for only 3.3 hours per night by these patients and might have been “insufficient to provide the level of effect on cardiovascular outcomes that had been hypothesized,” Dr. Doug McEvoy of the Adelaide Institute for Sleep Health, Flinders University, Adelaide, Australia and his associates reported at the annual congress of the European Society of Cardiology. Their study was simultaneously published in the New England Journal of Medicine (N Engl J Med. 2016 Aug 28. doi: 10.1056/NEJMoa1606599).
Notably, CPAP did show a trend toward significance in a prespecified subgroup analysis that matched 561 patients who used CPAP for a longer period – more than 4 hours a night – with the same number of controls (hazard ratio, 0.8; 95% CI, 0.6 to 1.1; P = .1). Dr. McEvoy discussed the implications of prolonged CPAP use in a video interview with Bruce Jancin, our reporter at the ESC Congress in Rome.
Obstructive sleep apnea causes episodic hypoxemia, sympathetic nervous system activation; intrathoracic pressure swings strain the heart and great vessels, and increases markers of oxidative stress, hypercoagulation, and inflammation. Randomized trials have linked CPAP therapy to lower systolic blood pressure measures and improved endothelial function and insulin sensitivity. Observational studies suggest that CPAP might help prevent cardiovascular events and death if used consistently, the investigators noted.
Because cardiovascular disease and obstructive sleep apnea often co-occur, the researchers carried out a secondary prevention trial, Sleep Apnea Cardiovascular Endpoints (SAVE), to quantify rates of major cardiovascular events among 2,717 adults aged 45-75 years with obstructive sleep apnea and established coronary or cerebrovascular disease. Patients were randomly assigned to receive CPAP therapy plus usual care, or usual care alone. The primary endpoint was a composite of cardiovascular death, myocardial infarction, stroke, or hospitalization from unstable angina, transient ischemic attack, or heart failure. The researchers also looked at other cardiovascular outcomes, snoring symptoms, mood, daytime sleepiness, and health-related quality of life. They used a 1-week run-in period of sham CPAP (administered at subtherapeutic pressure) to ensure what they considered an adequate level of adherence.
The average apnea-hypopnea index (that is, the average number of apnea or hypopnea events recorded per hour) was 29 at baseline and 3.7 after initiating CPAP, the investigators said. At a mean of 3.7 years of follow-up, 17% of CPAP users (220 patients) and 15.4% of controls had a cardiovascular event, for a hazard ratio of 1.1 (95% confidence interval, 0.9 to 1.3; P = 0.3).
Not only did CPAP fail to meet the composite primary endpoint, but it did not significantly affect any cause-specific cardiovascular outcome, the researchers said. However, CPAP users did improve significantly more than controls on measures of daytime sleepiness (the Epworth Sleepiness Scale), anxiety and depression (Hospital Anxiety and Depression Scale), self-reported physical and mental health (Short-Form Health Survey), and quality of life (European Quality of Life-5 Dimensions questionnaire). They also missed fewer days of work than did controls.
Study funders included the National Health and Medical Research Council of Australia, Respironics Sleep and Respiratory Research Foundation, and Phillips Respironics. Dr. McEvoy reported receiving research equipment for the study from AirLiquide. Several coinvestigators reported other ties to industry.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Adults with moderate to severe sleep apnea and coronary or cerebrovascular disease had about the same frequency of cardiovascular events whether they received continuous positive airway pressure (CPAP) therapy or usual care alone, according to a large randomized trial.
But CPAP was used for only 3.3 hours per night by these patients and might have been “insufficient to provide the level of effect on cardiovascular outcomes that had been hypothesized,” Dr. Doug McEvoy of the Adelaide Institute for Sleep Health, Flinders University, Adelaide, Australia and his associates reported at the annual congress of the European Society of Cardiology. Their study was simultaneously published in the New England Journal of Medicine (N Engl J Med. 2016 Aug 28. doi: 10.1056/NEJMoa1606599).
Notably, CPAP did show a trend toward significance in a prespecified subgroup analysis that matched 561 patients who used CPAP for a longer period – more than 4 hours a night – with the same number of controls (hazard ratio, 0.8; 95% CI, 0.6 to 1.1; P = .1). Dr. McEvoy discussed the implications of prolonged CPAP use in a video interview with Bruce Jancin, our reporter at the ESC Congress in Rome.
Obstructive sleep apnea causes episodic hypoxemia, sympathetic nervous system activation; intrathoracic pressure swings strain the heart and great vessels, and increases markers of oxidative stress, hypercoagulation, and inflammation. Randomized trials have linked CPAP therapy to lower systolic blood pressure measures and improved endothelial function and insulin sensitivity. Observational studies suggest that CPAP might help prevent cardiovascular events and death if used consistently, the investigators noted.
Because cardiovascular disease and obstructive sleep apnea often co-occur, the researchers carried out a secondary prevention trial, Sleep Apnea Cardiovascular Endpoints (SAVE), to quantify rates of major cardiovascular events among 2,717 adults aged 45-75 years with obstructive sleep apnea and established coronary or cerebrovascular disease. Patients were randomly assigned to receive CPAP therapy plus usual care, or usual care alone. The primary endpoint was a composite of cardiovascular death, myocardial infarction, stroke, or hospitalization from unstable angina, transient ischemic attack, or heart failure. The researchers also looked at other cardiovascular outcomes, snoring symptoms, mood, daytime sleepiness, and health-related quality of life. They used a 1-week run-in period of sham CPAP (administered at subtherapeutic pressure) to ensure what they considered an adequate level of adherence.
The average apnea-hypopnea index (that is, the average number of apnea or hypopnea events recorded per hour) was 29 at baseline and 3.7 after initiating CPAP, the investigators said. At a mean of 3.7 years of follow-up, 17% of CPAP users (220 patients) and 15.4% of controls had a cardiovascular event, for a hazard ratio of 1.1 (95% confidence interval, 0.9 to 1.3; P = 0.3).
Not only did CPAP fail to meet the composite primary endpoint, but it did not significantly affect any cause-specific cardiovascular outcome, the researchers said. However, CPAP users did improve significantly more than controls on measures of daytime sleepiness (the Epworth Sleepiness Scale), anxiety and depression (Hospital Anxiety and Depression Scale), self-reported physical and mental health (Short-Form Health Survey), and quality of life (European Quality of Life-5 Dimensions questionnaire). They also missed fewer days of work than did controls.
Study funders included the National Health and Medical Research Council of Australia, Respironics Sleep and Respiratory Research Foundation, and Phillips Respironics. Dr. McEvoy reported receiving research equipment for the study from AirLiquide. Several coinvestigators reported other ties to industry.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
FROM THE ESC CONGRESS 2016
Key clinical point: About 3.3. hours a night of continuous positive airway pressure (CPAP) therapy did not prevent more serious cardiovascular events than usual care alone for adults with moderate to severe obstructive sleep apnea and established cardiovascular or cerebrovascular disease.
Major finding: At 3.7 years of follow-up, 17% of CPAP patients and 15.4% of controls had experienced a major cardiovascular event (hazard ratio, 1.1; P = .3).
Data source: An international, multicenter, randomized, parallel-group, open-label trial of 2,717 adults with blinded endpoint assessment.
Disclosures: Study funders included the National Health and Medical Research Council of Australia, Respironics Sleep and Respiratory Research Foundation, and Phillips Respironics. Dr. McEvoy reported receiving research equipment for the study from AirLiquide. Several coinvestigators reported a number of other ties to industry.
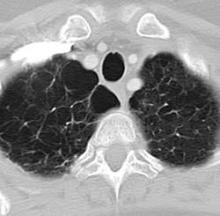
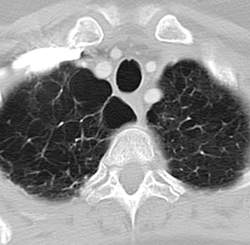
NAF1 gene mutations predispose to pulmonary fibrosis, emphysema
Rare frameshift mutations in the NAF1 gene were discovered to cause a telomere-shortening syndrome which, among other adverse effects, predisposes carriers to develop pulmonary fibrosis (PF) and emphysema, according to a report published in Science Translational Medicine.
“Our findings here ... highlight how telomere shortening is a relevant mechanism for PF-emphysema susceptibility in a subset of patients beyond those with mutations in the telomerase core components. It is thus possible that efforts to reverse the telomere defect, or other regenerative approaches, will influence the natural history of these progressive pathologies in patients with telomere-mediated lung disease,” said Susan E. Stanley, an MD-PhD candidate in the department of oncology, Johns Hopkins University, Baltimore, and her associates.
Pulmonary fibrosis and emphysema cluster in some families, but the genetic basis of such cases is poorly understood. Both PF and emphysema have been linked to premature aging of lung tissue and to abnormalities in the maintenance of telomere length. In addition, at least half of patients with familial and sporadic PF, and many with emphysema, have the clinical features of a short-telomere syndrome, including bone marrow failure/myelodysplastic syndrome, liver disease, and infertility.
The diagnosis of a short-telomere syndrome, as opposed to isolated PF-emphysema, is essential for appropriate treatment because if the defect is systemic, patients will “show exquisite sensitivity to otherwise tolerated medications and procedures, especially in the setting of lung transplantation,” the investigators said (Sci Transl Med. 2016;8:351ra107).
To explore the genetic basis of familial PF-emphysema, the researchers performed a series of studies, beginning with whole-genome sequencing on peripheral blood samples from five unrelated probands in familial PF-emphysema pedigrees. These participants had abnormally short telomeres and extrapulmonary features of short-telomere syndrome. Three of them who had low levels of the telomerase RNA component TR were selected for a candidate gene search, which revealed the NAF1 mutations.
The mutations were then found to be present in 2 of 30 (7%) affected members of a prevalence cohort but in none of 134 unaffected control subjects (0%), and in none of 9,006 samples from a public database of unaffected people (0%). Further genetic laboratory and mouse studies were performed to link the mutations with specific pathologies and to trace their functional effects. Their results led the researchers to conclude that these rare NAF1 variants interfere with RNA biogenesis, causing short telomeres resulting in lung disease and other abnormalities.
This work was supported by the National Institutes of Health, the Commonwealth Foundation, and the American Cancer Society. Ms. Stanley and her associates reported having no relevant financial disclosures.
Rare frameshift mutations in the NAF1 gene were discovered to cause a telomere-shortening syndrome which, among other adverse effects, predisposes carriers to develop pulmonary fibrosis (PF) and emphysema, according to a report published in Science Translational Medicine.
“Our findings here ... highlight how telomere shortening is a relevant mechanism for PF-emphysema susceptibility in a subset of patients beyond those with mutations in the telomerase core components. It is thus possible that efforts to reverse the telomere defect, or other regenerative approaches, will influence the natural history of these progressive pathologies in patients with telomere-mediated lung disease,” said Susan E. Stanley, an MD-PhD candidate in the department of oncology, Johns Hopkins University, Baltimore, and her associates.
Pulmonary fibrosis and emphysema cluster in some families, but the genetic basis of such cases is poorly understood. Both PF and emphysema have been linked to premature aging of lung tissue and to abnormalities in the maintenance of telomere length. In addition, at least half of patients with familial and sporadic PF, and many with emphysema, have the clinical features of a short-telomere syndrome, including bone marrow failure/myelodysplastic syndrome, liver disease, and infertility.
The diagnosis of a short-telomere syndrome, as opposed to isolated PF-emphysema, is essential for appropriate treatment because if the defect is systemic, patients will “show exquisite sensitivity to otherwise tolerated medications and procedures, especially in the setting of lung transplantation,” the investigators said (Sci Transl Med. 2016;8:351ra107).
To explore the genetic basis of familial PF-emphysema, the researchers performed a series of studies, beginning with whole-genome sequencing on peripheral blood samples from five unrelated probands in familial PF-emphysema pedigrees. These participants had abnormally short telomeres and extrapulmonary features of short-telomere syndrome. Three of them who had low levels of the telomerase RNA component TR were selected for a candidate gene search, which revealed the NAF1 mutations.
The mutations were then found to be present in 2 of 30 (7%) affected members of a prevalence cohort but in none of 134 unaffected control subjects (0%), and in none of 9,006 samples from a public database of unaffected people (0%). Further genetic laboratory and mouse studies were performed to link the mutations with specific pathologies and to trace their functional effects. Their results led the researchers to conclude that these rare NAF1 variants interfere with RNA biogenesis, causing short telomeres resulting in lung disease and other abnormalities.
This work was supported by the National Institutes of Health, the Commonwealth Foundation, and the American Cancer Society. Ms. Stanley and her associates reported having no relevant financial disclosures.
Rare frameshift mutations in the NAF1 gene were discovered to cause a telomere-shortening syndrome which, among other adverse effects, predisposes carriers to develop pulmonary fibrosis (PF) and emphysema, according to a report published in Science Translational Medicine.
“Our findings here ... highlight how telomere shortening is a relevant mechanism for PF-emphysema susceptibility in a subset of patients beyond those with mutations in the telomerase core components. It is thus possible that efforts to reverse the telomere defect, or other regenerative approaches, will influence the natural history of these progressive pathologies in patients with telomere-mediated lung disease,” said Susan E. Stanley, an MD-PhD candidate in the department of oncology, Johns Hopkins University, Baltimore, and her associates.
Pulmonary fibrosis and emphysema cluster in some families, but the genetic basis of such cases is poorly understood. Both PF and emphysema have been linked to premature aging of lung tissue and to abnormalities in the maintenance of telomere length. In addition, at least half of patients with familial and sporadic PF, and many with emphysema, have the clinical features of a short-telomere syndrome, including bone marrow failure/myelodysplastic syndrome, liver disease, and infertility.
The diagnosis of a short-telomere syndrome, as opposed to isolated PF-emphysema, is essential for appropriate treatment because if the defect is systemic, patients will “show exquisite sensitivity to otherwise tolerated medications and procedures, especially in the setting of lung transplantation,” the investigators said (Sci Transl Med. 2016;8:351ra107).
To explore the genetic basis of familial PF-emphysema, the researchers performed a series of studies, beginning with whole-genome sequencing on peripheral blood samples from five unrelated probands in familial PF-emphysema pedigrees. These participants had abnormally short telomeres and extrapulmonary features of short-telomere syndrome. Three of them who had low levels of the telomerase RNA component TR were selected for a candidate gene search, which revealed the NAF1 mutations.
The mutations were then found to be present in 2 of 30 (7%) affected members of a prevalence cohort but in none of 134 unaffected control subjects (0%), and in none of 9,006 samples from a public database of unaffected people (0%). Further genetic laboratory and mouse studies were performed to link the mutations with specific pathologies and to trace their functional effects. Their results led the researchers to conclude that these rare NAF1 variants interfere with RNA biogenesis, causing short telomeres resulting in lung disease and other abnormalities.
This work was supported by the National Institutes of Health, the Commonwealth Foundation, and the American Cancer Society. Ms. Stanley and her associates reported having no relevant financial disclosures.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: Certain rare mutations in the NAF1 gene were discovered to predispose carriers to develop pulmonary fibrosis and emphysema.
Major finding: The rare NAF1 mutations were detected in 2 of 30 (7%) family members in an affected pedigree but in 0 of 134 controls.
Data source: A series of genetic sequencing and other studies involving five affected probands, 30 unrelated but affected patients, and 134 control subjects.
Disclosures: This work was supported by the National Institutes of Health, the Commonwealth Foundation, and the American Cancer Society. Ms. Stanley and her associates reported having no relevant financial disclosures.
Flu vaccine prevented hospitalizations in patients 50 and older
The seasonal influenza vaccination reduced flu-related hospitalizations by 56.8% among people aged 50 and older during a recent flu season, according to a report published in Clinical Infectious Diseases.
Even in the oldest age group – the population with the highest risk of developing flu complications and perhaps the weakest immune response – influenza vaccination prevented serious complications, said Fiona P. Havers, MD, of the influenza division, Centers for Disease Control and Prevention, Atlanta, and her associates.
Data on vaccine efficacy in older adults are sparse, and randomized, placebo-controlled trials to gather evidence would be unethical. Dr. Havers and her colleagues studied the issue using a case-control design, focusing on community-dwelling adults aged 50 years and older during the 2010-2011 flu season. They identified 368 patients across 10 states who were hospitalized for polymerase chain reaction–confirmed influenza and matched them for age and county of residence with 773 control subjects.
Hospitalized case-patients were less likely to have been vaccinated (55%) than were control subjects (63%). Thus, the flu vaccine reduced the risk of hospitalization for influenza by 56.8% overall.
Vaccination reduced hospitalization for influenza by 63.9% in the youngest age group (50-64 years), by 61.0% in the intermediate age group (65-74 years), and by 57.3% in the oldest age group (75 years and older).
These results are similar to those reported in other studies assessing the same time period, including one that evaluated vaccine efficacy in ambulatory adults in the United States and Europe. They also are consistent with the results of observational studies performed during different flu seasons, the investigators said (Clin Infect Dis. 2016 Aug 2. doi: 10.1093/cid/ciw512).
Compared with control subjects, case-patients were more likely to be of nonwhite race, to be of Hispanic ethnicity, to have a lower income, to have had fewer years of education, to have two or more chronic health conditions, to have required recent hospitalization for respiratory problems, to have impaired mobility, and to have lower functional status.
“These findings support current U.S. recommendations for annual influenza vaccination in older adults, especially in adults aged 65 and older who are at higher risk of influenza-associated complications,” Dr. Havers and her associates said.
The Centers for Disease Control and Prevention supported the study. Dr. Havers reported having no relevant financial disclosures; one of her associates reported ties to Genentech, Merck, Novavax, and Pfizer.
The seasonal influenza vaccination reduced flu-related hospitalizations by 56.8% among people aged 50 and older during a recent flu season, according to a report published in Clinical Infectious Diseases.
Even in the oldest age group – the population with the highest risk of developing flu complications and perhaps the weakest immune response – influenza vaccination prevented serious complications, said Fiona P. Havers, MD, of the influenza division, Centers for Disease Control and Prevention, Atlanta, and her associates.
Data on vaccine efficacy in older adults are sparse, and randomized, placebo-controlled trials to gather evidence would be unethical. Dr. Havers and her colleagues studied the issue using a case-control design, focusing on community-dwelling adults aged 50 years and older during the 2010-2011 flu season. They identified 368 patients across 10 states who were hospitalized for polymerase chain reaction–confirmed influenza and matched them for age and county of residence with 773 control subjects.
Hospitalized case-patients were less likely to have been vaccinated (55%) than were control subjects (63%). Thus, the flu vaccine reduced the risk of hospitalization for influenza by 56.8% overall.
Vaccination reduced hospitalization for influenza by 63.9% in the youngest age group (50-64 years), by 61.0% in the intermediate age group (65-74 years), and by 57.3% in the oldest age group (75 years and older).
These results are similar to those reported in other studies assessing the same time period, including one that evaluated vaccine efficacy in ambulatory adults in the United States and Europe. They also are consistent with the results of observational studies performed during different flu seasons, the investigators said (Clin Infect Dis. 2016 Aug 2. doi: 10.1093/cid/ciw512).
Compared with control subjects, case-patients were more likely to be of nonwhite race, to be of Hispanic ethnicity, to have a lower income, to have had fewer years of education, to have two or more chronic health conditions, to have required recent hospitalization for respiratory problems, to have impaired mobility, and to have lower functional status.
“These findings support current U.S. recommendations for annual influenza vaccination in older adults, especially in adults aged 65 and older who are at higher risk of influenza-associated complications,” Dr. Havers and her associates said.
The Centers for Disease Control and Prevention supported the study. Dr. Havers reported having no relevant financial disclosures; one of her associates reported ties to Genentech, Merck, Novavax, and Pfizer.
The seasonal influenza vaccination reduced flu-related hospitalizations by 56.8% among people aged 50 and older during a recent flu season, according to a report published in Clinical Infectious Diseases.
Even in the oldest age group – the population with the highest risk of developing flu complications and perhaps the weakest immune response – influenza vaccination prevented serious complications, said Fiona P. Havers, MD, of the influenza division, Centers for Disease Control and Prevention, Atlanta, and her associates.
Data on vaccine efficacy in older adults are sparse, and randomized, placebo-controlled trials to gather evidence would be unethical. Dr. Havers and her colleagues studied the issue using a case-control design, focusing on community-dwelling adults aged 50 years and older during the 2010-2011 flu season. They identified 368 patients across 10 states who were hospitalized for polymerase chain reaction–confirmed influenza and matched them for age and county of residence with 773 control subjects.
Hospitalized case-patients were less likely to have been vaccinated (55%) than were control subjects (63%). Thus, the flu vaccine reduced the risk of hospitalization for influenza by 56.8% overall.
Vaccination reduced hospitalization for influenza by 63.9% in the youngest age group (50-64 years), by 61.0% in the intermediate age group (65-74 years), and by 57.3% in the oldest age group (75 years and older).
These results are similar to those reported in other studies assessing the same time period, including one that evaluated vaccine efficacy in ambulatory adults in the United States and Europe. They also are consistent with the results of observational studies performed during different flu seasons, the investigators said (Clin Infect Dis. 2016 Aug 2. doi: 10.1093/cid/ciw512).
Compared with control subjects, case-patients were more likely to be of nonwhite race, to be of Hispanic ethnicity, to have a lower income, to have had fewer years of education, to have two or more chronic health conditions, to have required recent hospitalization for respiratory problems, to have impaired mobility, and to have lower functional status.
“These findings support current U.S. recommendations for annual influenza vaccination in older adults, especially in adults aged 65 and older who are at higher risk of influenza-associated complications,” Dr. Havers and her associates said.
The Centers for Disease Control and Prevention supported the study. Dr. Havers reported having no relevant financial disclosures; one of her associates reported ties to Genentech, Merck, Novavax, and Pfizer.
FROM CLINICAL INFECTIOUS DISEASES
Key clinical point: Seasonal influenza vaccination reduced flu-related hospitalizations by 56.8% in people aged 50 years and older.
Major finding: Vaccination reduced hospitalization for influenza by 63.9% in people aged 50-64 years, by 61.0% in those aged 65-74 years, and by 57.3% in those aged 75 years and older.
Data source: A retrospective case-control study involving 368 cases and 773 matched controls assessed during a single recent flu season.
Disclosures: The Centers for Disease Control and Prevention supported the study. Dr. Havers reported having no relevant financial disclosures; one of her associates reported ties to Genentech, Merck, Novavax, and Pfizer.
Nonpharmacologic AD therapy: Strongest evidence supports moisturizers
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
BOSTON – Moisturizers are “a cornerstone” of therapy for children with atopic dermatitis, according to Julie V. Schaffer, MD.
Moisturizers improve skin hydration, increase the time between flares, and reduce xerosis and pruritus, Dr. Schaffer of Hackensack (N.J.) University Medical Group said at the American Academy of Dermatology summer meeting.
In 2014, the AAD released guidelines that “very strongly” recommended moisturizers as an important nonpharmacologic intervention for patients with AD, stating that moisturizer use decreases disease severity and can reduce the need for pharmacologic intervention, she said.
In fact, the recommendation for moisturizer was based on “strength A, level 1 evidence,” she noted.
The role of bathing is a bit less clear; bathing is suggested as part of treatment and maintenance, but no standard exists with respect to frequency or duration for those with AD (evidence level: III, strength of recommendation: C). In general, the AAD recommends daily or less frequent bathing in warm water for 5-10 minutes, but surveys suggest that bathing recommendations vary widely among specialists and primary care providers, Dr. Schaffer said.
She noted that she sometimes sees children who have been told to bathe only once a week.
“They will come in just covered with disgusting gunk and it can’t be good for them,” she said. Bathing, especially if they have crusting and scaling, removes irritants and potential allergens, and provides hydration. It can also improve penetration of topical medications, as well as tolerance of those medications so that they burn less.
“So I give a thumbs up to daily bathing,” she said.
It is generally agreed that moisturizers should be applied soon after bathing (after applying medication) to improve skin hydration in patients with AD, Dr. Schaffer said.
The AAD says that moisturizers should be applied liberally and frequently, but the ideal frequency and type of moisturizer remains “a bit of an art form rather than a precise science,” she added.
The ideal moisturizer is one that is safe, effective, and free of fragrance, irritants, and potential sensitizers, she said, noting that “an individualized approach to moisturizer and vehicle selection can be very helpful.”
For young children, it is important that the product doesn’t sting; an ointment may be preferable in this population. Preteens and teenagers may dislike greasiness, so that is an important consideration, she said.
Dr. Schaffer pointed out that lotion formulations typically have water content that is too high to be helpful for patients with substantial xerosis. Creams or ointments may be a better bet, but take care to avoid contamination in large jars of such products, she advised.
“I’ve had a couple times when patients were getting recurrent infections, and we traced it down to a nasty jar that had a little too much bacteria in it,” she said, noting that using a clean scoop or pump can help prevent contamination.
As for cleansers, the “pretty clear winner” is a nonsoap cleanser, Dr. Schaffer said.
The AAD recommends limited use of hypoallergenic, fragrance-free, nonsoap cleansers with neutral to low pH, but the evidence is insufficient for recommending the addition of bath oils, emollients, oatmeal, and most other additives to bath water, as well as for the use of acidic spring water, she said (evidence level: III, strength of recommendation: C). An exception is bleach baths, as adding a small amount of bleach to bath water has been shown to improve symptoms, but the other products have not been shown to be beneficial.
The AAD notes that wet wrap therapy, either with or without a topical corticosteroid, can be recommended for patients with moderate to severe AD, as this can decrease disease severity and water loss during flares (evidence level: II, strength of recommendation: B).
Use moisturizer in newborns at risk for AD
Moisturizers don’t just help improve atopic dermatitis in children, they may also prevent the condition in at risk newborns.
Parents of a child with eczema who are concerned about the condition developing in their next child may find hope in the findings from two studies published in 2014, Dr. Schaffer said.
In a study of 124 newborns at high risk for AD who were randomized to daily emollient therapy or usual infant skin care started by age 3 weeks, the incidence of AD over 6 months was 43% in the control group, vs. 22% in the emollient group, a relative risk reduction of 50% (J Allergy Clin Immunol. 2014 Oct;134[4]:818-23). Parents in the emollient therapy group were allowed to choose between sunflower oil, Cetaphil cream, or Aquaphor Healing Ointment.
In a similar Japanese study of 118 high risk infants who were randomized to daily treatment with an emulsion-type emollient or usual skin care starting the first week of life, the AD/eczema rates at 32 weeks were 47% and 32% in the control and emollient groups, respectively (J Allergy Clin Immunol. 2014 Oct;134[4], 824-30). Both groups were allowed to use petroleum jelly.
“So that is something you can potentially make a recommendation for,” she said.
Dr. Schaffer reported having no conflicts of interest.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY 2016
Clinical decision tree pinpointed risk of extended-spectrum beta-lactamase bacteremia
A new classification tool helped guide the treatment of bacteremic patients while clinicians awaited antibiotic resistance results, investigators reported.
The clinical decision tree had a positive predictive value of 91% and a negative predictive value of 92% for determining whether certain gram-negative infections produced extended-spectrum beta-lactamase (ESBL), Catherine Goodman, PhD, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, and her associates wrote online in Clinical Infectious Diseases. “These predictions may assist empiric treatment decisions in order to optimize clinical outcomes while reducing administration of overly broad antibiotic agents that can select for further resistance emergence,” they added.
Bacteria that produce ESBL can hydrolyze all broad-spectrum beta-lactam antibiotics except carbapenems. Rapid tests for beta-lactamase genes can shorten the lag time between gram-stain identification and antimicrobial resistance results, but are cost prohibitive for most clinical laboratories and often do not assess ESBL gene groups, the researchers said. To find a way to predict which infections are characterized by ESBL production, they studied adults hospitalized at Johns Hopkins from October 2008 to March 2015 with bloodstream isolates of Klebsiella pneumoniae (40% of patients), Klebsiella oxytoca (4% of patients), and Escherichia coli (56% of patients). Most bacteremias began as urinary tract infections (34% of cases), followed by intra-abdominal infections (24%), catheter-related infections (16%), and biliary infections (14%) (Clin Infect Dis. 2016 Jul 26. doi:10.1093/cid/ciw425).
A total of 194 patients (15%) had bacteremias that produced ESBL, according to the investigators. Using a technique called binary recursive partitioning, they compared these patients with ESBL-negative patients to create a clinical decision tree based on five yes-or-no questions. The tree first asked if the patient had been colonized or infected with ESBL-producing bacteria within 6 months, and if so, whether the patient currently had an indwelling catheter. Patients meeting both criteria had a 92% chance of being ESBL positive. Patients with a recent history of ESBL but no catheter had an 81% chance of being ESBL positive if they were at least 43 years old, but a 75% chance of being ESBL negative if they were under age 43 years.
Among patients with no recent history of ESBL, the decision tree asked about hospitalization in a country with a high ESBL burden and antibiotic therapy during the past 6 months. Patients responding “yes” to both questions had a 100% chance of being ESBL positive. Patients with only the geographic risk factor had a 63% chance of being ESBL negative, and patients with neither risk factor had a 93% chance of being ESBL negative.
The decision tree detected only half of ESBL cases because there was a subgroup with no recent ESBL history or geographic exposure, the investigators noted. “The poor predictive nature of health care–associated variables within this patient subset may suggest a high proportion of community-acquired ESBL infections. Indeed, although risk factors for ESBLs have traditionally focused on the health care setting, increasing reports describe the community as an important ESBL reservoir,” they added. Nonetheless, of 194 patients with ESBL bacteremia, 35% received empiric carbapenem treatment within 6 hours after identification of the bacterial genus and species, the investigators emphasized. “Utilization of the decision tree would have increased ESBL case detection during the empiric treatment window by approximately 50%.”
The National Institutes of Health funded the study. The researchers reported having no conflicts of interest.
A new classification tool helped guide the treatment of bacteremic patients while clinicians awaited antibiotic resistance results, investigators reported.
The clinical decision tree had a positive predictive value of 91% and a negative predictive value of 92% for determining whether certain gram-negative infections produced extended-spectrum beta-lactamase (ESBL), Catherine Goodman, PhD, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, and her associates wrote online in Clinical Infectious Diseases. “These predictions may assist empiric treatment decisions in order to optimize clinical outcomes while reducing administration of overly broad antibiotic agents that can select for further resistance emergence,” they added.
Bacteria that produce ESBL can hydrolyze all broad-spectrum beta-lactam antibiotics except carbapenems. Rapid tests for beta-lactamase genes can shorten the lag time between gram-stain identification and antimicrobial resistance results, but are cost prohibitive for most clinical laboratories and often do not assess ESBL gene groups, the researchers said. To find a way to predict which infections are characterized by ESBL production, they studied adults hospitalized at Johns Hopkins from October 2008 to March 2015 with bloodstream isolates of Klebsiella pneumoniae (40% of patients), Klebsiella oxytoca (4% of patients), and Escherichia coli (56% of patients). Most bacteremias began as urinary tract infections (34% of cases), followed by intra-abdominal infections (24%), catheter-related infections (16%), and biliary infections (14%) (Clin Infect Dis. 2016 Jul 26. doi:10.1093/cid/ciw425).
A total of 194 patients (15%) had bacteremias that produced ESBL, according to the investigators. Using a technique called binary recursive partitioning, they compared these patients with ESBL-negative patients to create a clinical decision tree based on five yes-or-no questions. The tree first asked if the patient had been colonized or infected with ESBL-producing bacteria within 6 months, and if so, whether the patient currently had an indwelling catheter. Patients meeting both criteria had a 92% chance of being ESBL positive. Patients with a recent history of ESBL but no catheter had an 81% chance of being ESBL positive if they were at least 43 years old, but a 75% chance of being ESBL negative if they were under age 43 years.
Among patients with no recent history of ESBL, the decision tree asked about hospitalization in a country with a high ESBL burden and antibiotic therapy during the past 6 months. Patients responding “yes” to both questions had a 100% chance of being ESBL positive. Patients with only the geographic risk factor had a 63% chance of being ESBL negative, and patients with neither risk factor had a 93% chance of being ESBL negative.
The decision tree detected only half of ESBL cases because there was a subgroup with no recent ESBL history or geographic exposure, the investigators noted. “The poor predictive nature of health care–associated variables within this patient subset may suggest a high proportion of community-acquired ESBL infections. Indeed, although risk factors for ESBLs have traditionally focused on the health care setting, increasing reports describe the community as an important ESBL reservoir,” they added. Nonetheless, of 194 patients with ESBL bacteremia, 35% received empiric carbapenem treatment within 6 hours after identification of the bacterial genus and species, the investigators emphasized. “Utilization of the decision tree would have increased ESBL case detection during the empiric treatment window by approximately 50%.”
The National Institutes of Health funded the study. The researchers reported having no conflicts of interest.
A new classification tool helped guide the treatment of bacteremic patients while clinicians awaited antibiotic resistance results, investigators reported.
The clinical decision tree had a positive predictive value of 91% and a negative predictive value of 92% for determining whether certain gram-negative infections produced extended-spectrum beta-lactamase (ESBL), Catherine Goodman, PhD, of the Johns Hopkins Bloomberg School of Public Health, Baltimore, and her associates wrote online in Clinical Infectious Diseases. “These predictions may assist empiric treatment decisions in order to optimize clinical outcomes while reducing administration of overly broad antibiotic agents that can select for further resistance emergence,” they added.
Bacteria that produce ESBL can hydrolyze all broad-spectrum beta-lactam antibiotics except carbapenems. Rapid tests for beta-lactamase genes can shorten the lag time between gram-stain identification and antimicrobial resistance results, but are cost prohibitive for most clinical laboratories and often do not assess ESBL gene groups, the researchers said. To find a way to predict which infections are characterized by ESBL production, they studied adults hospitalized at Johns Hopkins from October 2008 to March 2015 with bloodstream isolates of Klebsiella pneumoniae (40% of patients), Klebsiella oxytoca (4% of patients), and Escherichia coli (56% of patients). Most bacteremias began as urinary tract infections (34% of cases), followed by intra-abdominal infections (24%), catheter-related infections (16%), and biliary infections (14%) (Clin Infect Dis. 2016 Jul 26. doi:10.1093/cid/ciw425).
A total of 194 patients (15%) had bacteremias that produced ESBL, according to the investigators. Using a technique called binary recursive partitioning, they compared these patients with ESBL-negative patients to create a clinical decision tree based on five yes-or-no questions. The tree first asked if the patient had been colonized or infected with ESBL-producing bacteria within 6 months, and if so, whether the patient currently had an indwelling catheter. Patients meeting both criteria had a 92% chance of being ESBL positive. Patients with a recent history of ESBL but no catheter had an 81% chance of being ESBL positive if they were at least 43 years old, but a 75% chance of being ESBL negative if they were under age 43 years.
Among patients with no recent history of ESBL, the decision tree asked about hospitalization in a country with a high ESBL burden and antibiotic therapy during the past 6 months. Patients responding “yes” to both questions had a 100% chance of being ESBL positive. Patients with only the geographic risk factor had a 63% chance of being ESBL negative, and patients with neither risk factor had a 93% chance of being ESBL negative.
The decision tree detected only half of ESBL cases because there was a subgroup with no recent ESBL history or geographic exposure, the investigators noted. “The poor predictive nature of health care–associated variables within this patient subset may suggest a high proportion of community-acquired ESBL infections. Indeed, although risk factors for ESBLs have traditionally focused on the health care setting, increasing reports describe the community as an important ESBL reservoir,” they added. Nonetheless, of 194 patients with ESBL bacteremia, 35% received empiric carbapenem treatment within 6 hours after identification of the bacterial genus and species, the investigators emphasized. “Utilization of the decision tree would have increased ESBL case detection during the empiric treatment window by approximately 50%.”
The National Institutes of Health funded the study. The researchers reported having no conflicts of interest.
FROM CLINICAL INFECTIOUS DISEASES
Key clinical point: A clinical decision tree helped identify bacteria producing extended-spectrum beta-lactamases.
Major finding: The positive predictive value was 91%, and the negative predictive value was 92%.
Data source: A single-center retrospective study of 1,288 adults with blood isolates of Klebsiella pneumoniae, Klebsiella oxytoca, or Escherichia coli.
Disclosures: The National Institutes of Health funded the study. The researchers reported having no conflicts of interest.