User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
E-cigarettes most popular among youngest adults
Over 15% of adults have used electronic cigarettes at some time, and about 3% reported current use when they were surveyed in 2016, according to the Centers for Disease Control and Prevention.
When those numbers are broken down by age group, the youngest adults are the most likely e-cigarette users: 23.5% of those aged 18-24 years had ever vaped and 4.5% were currently vaping either every day or on some days, the CDC reported (MMWR. 2017;66[33]:892).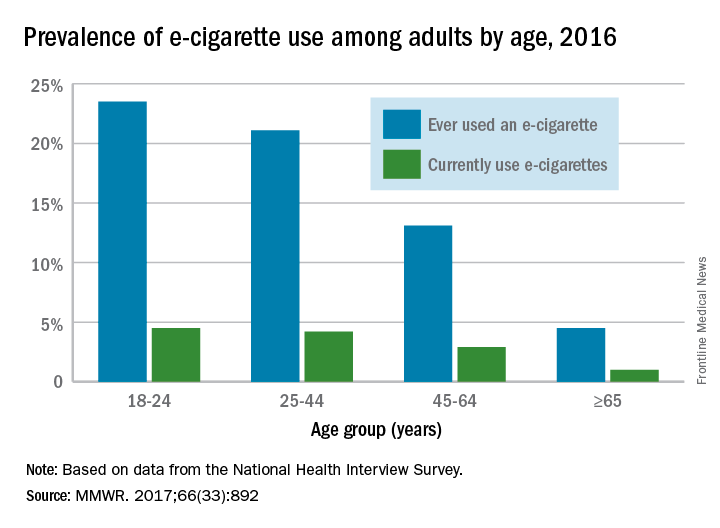
Over 15% of adults have used electronic cigarettes at some time, and about 3% reported current use when they were surveyed in 2016, according to the Centers for Disease Control and Prevention.
When those numbers are broken down by age group, the youngest adults are the most likely e-cigarette users: 23.5% of those aged 18-24 years had ever vaped and 4.5% were currently vaping either every day or on some days, the CDC reported (MMWR. 2017;66[33]:892).
Over 15% of adults have used electronic cigarettes at some time, and about 3% reported current use when they were surveyed in 2016, according to the Centers for Disease Control and Prevention.
When those numbers are broken down by age group, the youngest adults are the most likely e-cigarette users: 23.5% of those aged 18-24 years had ever vaped and 4.5% were currently vaping either every day or on some days, the CDC reported (MMWR. 2017;66[33]:892).
FROM MMWR
Statins linked to lower death rates in COPD
Receiving a statin prescription within a year after diagnosis of chronic obstructive pulmonary disease was associated with a 21% decrease in the subsequent risk of all-cause mortality and a 45% drop in risk of pulmonary mortality, according to the results of a large retrospective administrative database study.
The findings belie those of the recent Simvastatin in the Prevention of COPD Exacerbation (STATCOPE) trial, in which daily simvastatin (40 mg) did not affect exacerbation rates or time to first exacerbation in high-risk COPD patients, wrote Adam Raymakers, MSc, a doctoral candidate at the University of British Columbia, Vancouver, and his associates. Their study was observational, but the association between statin use and decreased mortality “persisted across several measures of statin exposure,” they wrote. “Our findings, in conjunction with previously reported evidence, suggests that there may be a specific subtype of COPD patients that may benefit from statin use.” The study appears in the September issue of Chest (2017;152;486-93).
To further explore the question, the researchers analyzed linked health databases from nearly 40,000 patients aged 50 years and older who had received at least three prescriptions for an anticholinergic or a short-acting beta agonist in 12 months some time between 1998 and 2007. The first prescription was considered the date of COPD “diagnosis.” The average age of the patients was 71 years; 55% were female.
A total of 7,775 patients (19.6%) who met this definition of incident COPD were prescribed a statin at least once during the subsequent year. These patients had a significantly reduced risk of subsequent all-cause mortality in univariate and multivariate analyses, with hazard ratios of 0.79 (95% confidence intervals, 0.68 to 0.91; P less than .002). Statins also showed a protective effect against pulmonary mortality, with univariate and multivariate hazard ratios of 0.52 (P = .01) and 0.55 (P = .03), respectively.
The protective effect of statins held up when the investigators narrowed the exposure period to 6 months after COPD diagnosis and when they expanded it to 18 months. Exposure to statins for 80% of the 1-year window after COPD diagnosis – a proxy for statin adherence – also led to a reduced risk of all-cause mortality, but the 95% confidence interval for the hazard ratio did not reach statistical significance (0.71 to 1.01; P = .06).
The most common prescription was for atorvastatin (49%), usually for 90 days (23%), 100 days (20%), or 30 days (15%), the researchers said. While the “possibility of the ‘healthy user’ or the ‘healthy adherer’ cannot be ignored,” they adjusted for other prescriptions, comorbidities, and income level, which should have helped eliminate this effect, they added. However, they lacked data on smoking and lung function assessments, both of which are “important confounders and contributors to mortality,” they acknowledged.
Canadian Institutes of Health Research supported the study. One coinvestigator disclosed consulting relationships with Teva, Pfizer, and Novartis. The others had no conflicts of interest.
Despite [its] limitations, the study results are intriguing and in line with findings from other retrospective cohorts. How then can we reconcile the apparent benefits observed in retrospective studies with the lack of clinical effect seen in prospective trials, particularly the Simvastatin in the Prevention of COPD Exacerbation (STATCOPE) study? Could it be that both negative and positive studies are “correct”? Prospective studies have thus far not been adequately powered for mortality as an endpoint. Perhaps the choice of the particular statin matters? While STATCOPE involved simvastatin, the majority of the cohort reported by Raymakers et al. received atorvastatin. [Or perhaps] the negative results of STATCOPE could be related to careful selection of study participants with a low burden of systemic inflammation.
This most recent study reinforces the idea that statins may play a beneficial role in COPD, but it isn’t clear which patients to target for therapy. It is unlikely that the findings by Raymakers et al. will reverse recent recommendations by the American College of Chest Physicians and Canadian Thoracic Society against the use of statins for the purpose of prevention of COPD exacerbations, but the suggestion of survival advantage related to statins certainly may breathe new life into an enthusiasm greatly tempered by STATCOPE.
Or Kalchiem-Dekel, MD, and Robert M. Reed, MD, are at the pulmonary and critical care medicine division, University of Maryland, Baltimore. Neither editorialist had conflicts of interest (Chest. 2017;152:456-7. doi: 10.1016/j.chest.2017.04.156).
Despite [its] limitations, the study results are intriguing and in line with findings from other retrospective cohorts. How then can we reconcile the apparent benefits observed in retrospective studies with the lack of clinical effect seen in prospective trials, particularly the Simvastatin in the Prevention of COPD Exacerbation (STATCOPE) study? Could it be that both negative and positive studies are “correct”? Prospective studies have thus far not been adequately powered for mortality as an endpoint. Perhaps the choice of the particular statin matters? While STATCOPE involved simvastatin, the majority of the cohort reported by Raymakers et al. received atorvastatin. [Or perhaps] the negative results of STATCOPE could be related to careful selection of study participants with a low burden of systemic inflammation.
This most recent study reinforces the idea that statins may play a beneficial role in COPD, but it isn’t clear which patients to target for therapy. It is unlikely that the findings by Raymakers et al. will reverse recent recommendations by the American College of Chest Physicians and Canadian Thoracic Society against the use of statins for the purpose of prevention of COPD exacerbations, but the suggestion of survival advantage related to statins certainly may breathe new life into an enthusiasm greatly tempered by STATCOPE.
Or Kalchiem-Dekel, MD, and Robert M. Reed, MD, are at the pulmonary and critical care medicine division, University of Maryland, Baltimore. Neither editorialist had conflicts of interest (Chest. 2017;152:456-7. doi: 10.1016/j.chest.2017.04.156).
Despite [its] limitations, the study results are intriguing and in line with findings from other retrospective cohorts. How then can we reconcile the apparent benefits observed in retrospective studies with the lack of clinical effect seen in prospective trials, particularly the Simvastatin in the Prevention of COPD Exacerbation (STATCOPE) study? Could it be that both negative and positive studies are “correct”? Prospective studies have thus far not been adequately powered for mortality as an endpoint. Perhaps the choice of the particular statin matters? While STATCOPE involved simvastatin, the majority of the cohort reported by Raymakers et al. received atorvastatin. [Or perhaps] the negative results of STATCOPE could be related to careful selection of study participants with a low burden of systemic inflammation.
This most recent study reinforces the idea that statins may play a beneficial role in COPD, but it isn’t clear which patients to target for therapy. It is unlikely that the findings by Raymakers et al. will reverse recent recommendations by the American College of Chest Physicians and Canadian Thoracic Society against the use of statins for the purpose of prevention of COPD exacerbations, but the suggestion of survival advantage related to statins certainly may breathe new life into an enthusiasm greatly tempered by STATCOPE.
Or Kalchiem-Dekel, MD, and Robert M. Reed, MD, are at the pulmonary and critical care medicine division, University of Maryland, Baltimore. Neither editorialist had conflicts of interest (Chest. 2017;152:456-7. doi: 10.1016/j.chest.2017.04.156).
Receiving a statin prescription within a year after diagnosis of chronic obstructive pulmonary disease was associated with a 21% decrease in the subsequent risk of all-cause mortality and a 45% drop in risk of pulmonary mortality, according to the results of a large retrospective administrative database study.
The findings belie those of the recent Simvastatin in the Prevention of COPD Exacerbation (STATCOPE) trial, in which daily simvastatin (40 mg) did not affect exacerbation rates or time to first exacerbation in high-risk COPD patients, wrote Adam Raymakers, MSc, a doctoral candidate at the University of British Columbia, Vancouver, and his associates. Their study was observational, but the association between statin use and decreased mortality “persisted across several measures of statin exposure,” they wrote. “Our findings, in conjunction with previously reported evidence, suggests that there may be a specific subtype of COPD patients that may benefit from statin use.” The study appears in the September issue of Chest (2017;152;486-93).
To further explore the question, the researchers analyzed linked health databases from nearly 40,000 patients aged 50 years and older who had received at least three prescriptions for an anticholinergic or a short-acting beta agonist in 12 months some time between 1998 and 2007. The first prescription was considered the date of COPD “diagnosis.” The average age of the patients was 71 years; 55% were female.
A total of 7,775 patients (19.6%) who met this definition of incident COPD were prescribed a statin at least once during the subsequent year. These patients had a significantly reduced risk of subsequent all-cause mortality in univariate and multivariate analyses, with hazard ratios of 0.79 (95% confidence intervals, 0.68 to 0.91; P less than .002). Statins also showed a protective effect against pulmonary mortality, with univariate and multivariate hazard ratios of 0.52 (P = .01) and 0.55 (P = .03), respectively.
The protective effect of statins held up when the investigators narrowed the exposure period to 6 months after COPD diagnosis and when they expanded it to 18 months. Exposure to statins for 80% of the 1-year window after COPD diagnosis – a proxy for statin adherence – also led to a reduced risk of all-cause mortality, but the 95% confidence interval for the hazard ratio did not reach statistical significance (0.71 to 1.01; P = .06).
The most common prescription was for atorvastatin (49%), usually for 90 days (23%), 100 days (20%), or 30 days (15%), the researchers said. While the “possibility of the ‘healthy user’ or the ‘healthy adherer’ cannot be ignored,” they adjusted for other prescriptions, comorbidities, and income level, which should have helped eliminate this effect, they added. However, they lacked data on smoking and lung function assessments, both of which are “important confounders and contributors to mortality,” they acknowledged.
Canadian Institutes of Health Research supported the study. One coinvestigator disclosed consulting relationships with Teva, Pfizer, and Novartis. The others had no conflicts of interest.
Receiving a statin prescription within a year after diagnosis of chronic obstructive pulmonary disease was associated with a 21% decrease in the subsequent risk of all-cause mortality and a 45% drop in risk of pulmonary mortality, according to the results of a large retrospective administrative database study.
The findings belie those of the recent Simvastatin in the Prevention of COPD Exacerbation (STATCOPE) trial, in which daily simvastatin (40 mg) did not affect exacerbation rates or time to first exacerbation in high-risk COPD patients, wrote Adam Raymakers, MSc, a doctoral candidate at the University of British Columbia, Vancouver, and his associates. Their study was observational, but the association between statin use and decreased mortality “persisted across several measures of statin exposure,” they wrote. “Our findings, in conjunction with previously reported evidence, suggests that there may be a specific subtype of COPD patients that may benefit from statin use.” The study appears in the September issue of Chest (2017;152;486-93).
To further explore the question, the researchers analyzed linked health databases from nearly 40,000 patients aged 50 years and older who had received at least three prescriptions for an anticholinergic or a short-acting beta agonist in 12 months some time between 1998 and 2007. The first prescription was considered the date of COPD “diagnosis.” The average age of the patients was 71 years; 55% were female.
A total of 7,775 patients (19.6%) who met this definition of incident COPD were prescribed a statin at least once during the subsequent year. These patients had a significantly reduced risk of subsequent all-cause mortality in univariate and multivariate analyses, with hazard ratios of 0.79 (95% confidence intervals, 0.68 to 0.91; P less than .002). Statins also showed a protective effect against pulmonary mortality, with univariate and multivariate hazard ratios of 0.52 (P = .01) and 0.55 (P = .03), respectively.
The protective effect of statins held up when the investigators narrowed the exposure period to 6 months after COPD diagnosis and when they expanded it to 18 months. Exposure to statins for 80% of the 1-year window after COPD diagnosis – a proxy for statin adherence – also led to a reduced risk of all-cause mortality, but the 95% confidence interval for the hazard ratio did not reach statistical significance (0.71 to 1.01; P = .06).
The most common prescription was for atorvastatin (49%), usually for 90 days (23%), 100 days (20%), or 30 days (15%), the researchers said. While the “possibility of the ‘healthy user’ or the ‘healthy adherer’ cannot be ignored,” they adjusted for other prescriptions, comorbidities, and income level, which should have helped eliminate this effect, they added. However, they lacked data on smoking and lung function assessments, both of which are “important confounders and contributors to mortality,” they acknowledged.
Canadian Institutes of Health Research supported the study. One coinvestigator disclosed consulting relationships with Teva, Pfizer, and Novartis. The others had no conflicts of interest.
FROM CHEST
Key clinical point: Statins might reduce the risk of death among patients with chronic obstructive pulmonary disease.
Major finding: Statin use was associated with a 21% decrease in risk of all-cause mortality and a 45% decrease in risk of pulmonary mortality.
Data source: A retrospective cohort study of 39,678 patients with COPD, including 7,775 prescribed statins.
Disclosures: Canadian Institutes of Health Research supported the study. One coinvestigator disclosed consulting relationships with Teva, Pfizer, and Novartis. The others had no conflicts of interest.
Riociguat may benefit subset of PAH patients
Switching to riociguat may be an effective strategy for pulmonary arterial hypertension (PAH) patients who respond inadequately to phosphodiesterase-5 inhibitors, results from a small open-label study demonstrated.
“This study represents an important step towards determining if this new treatment strategy is an effective approach to the management of patients with PAH, although additional data from larger, randomised, controlled studies are needed to further establish the safety and efficacy of this approach,” researchers led by Marius M. Hoeper, MD, wrote in a study published online Sept. 9, 2017, in the European Respiratory Journal.
Current clinical data indicate that many patients with PAH who receive phosphodiesterase-5 inhibitors do not reach treatment goals. “For example, in the AMBITION study, 73% of patients with PAH receiving tadalafil monotherapy and 61% of those receiving tadalafil in combination with ambrisentan did not achieve a satisfactory clinical response at week 24 of the study (N Engl J Med. 2015;373:834-44),” Dr. Hoeper of the Clinic for Respiratory Medicine at Hannover Medical School Germany and his associates wrote. “Furthermore, in the SERAPHIN study, event-free survival of patients receiving [phosphodiesterase-5 inhibitors] monotherapy was approximately 50% at 3 years (N Engl J Med. 2013;369:809-18).”
For the current trial, known as RESPITE, investigators from nine countries in Europe and North America enrolled 61 PAH patients in a 24-week, open-label uncontrolled analysis to investigate the safety, feasibility, and benefit of switching them from phosphodiesterase-5 inhibitors to riociguat. The patients underwent 1-3 days free of phosphodiesterase-5 inhibitors before receiving riociguat in a maximum dose of up to 2.5 mg t.i.d. Most patients (74%) were female, and 92% were Caucasian. In all, 51 patients (84%) completed all 24 weeks of treatment, while the remaining 10 discontinued treatment, 4 of whom due to adverse events.
Additionally, 54% of the patients studied experienced an improvement in their the World Health Organization Functional Class. However, 32 patients (52%) experienced study drug–related adverse events and 10 (16%) experienced serious adverse events, two of which were related to the drug being studied. Six patients (10%) experienced clinical worsening, including death in two, though the deaths were deemed to be unrelated to the drug being studied.
“Although not mechanistically studied, the findings of RESPITE support the hypothesis that a defective [nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate] pathway might explain why some patients have no sufficient or sustained response to [phosphodiesterase-5 inhibitors] therapy,” the researchers noted. “In such patients, direct stimulation of [soluble guanylate cyclase] may be more effective than inhibition of [phosphodiesterase-5], but this hypothesis is still unproven.”
They acknowledged certain limitations of the study, including its prospective design and the relatively homogenous patient population. “Other limitations include the lack of a long-term continuation phase, and the absence of mechanistic data allowing identification of patients likely to respond or not respond to switching,” they wrote. “Two deaths were observed in this study, which might raise concerns, although neither of the deaths (one due to pneumonia and one due to subdural haematoma) was considered by the investigators to be study drug-related or due to worsening PAH. Given the lack of a control group and the rate of study withdrawals and clinical worsening events, further evaluation to clarify the safety of switching is required.”
The study was funded by Bayer AG, Berlin. Dr. Hoeper and his coauthors disclosed having financial ties to numerous pharmaceutical companies, including Bayer, which makes riociguat.
PRIMARY SOURCE: Eur Respir J. 2017 Sep 9. doi: 10.1183/13993003.02425-2016.
Switching to riociguat may be an effective strategy for pulmonary arterial hypertension (PAH) patients who respond inadequately to phosphodiesterase-5 inhibitors, results from a small open-label study demonstrated.
“This study represents an important step towards determining if this new treatment strategy is an effective approach to the management of patients with PAH, although additional data from larger, randomised, controlled studies are needed to further establish the safety and efficacy of this approach,” researchers led by Marius M. Hoeper, MD, wrote in a study published online Sept. 9, 2017, in the European Respiratory Journal.
Current clinical data indicate that many patients with PAH who receive phosphodiesterase-5 inhibitors do not reach treatment goals. “For example, in the AMBITION study, 73% of patients with PAH receiving tadalafil monotherapy and 61% of those receiving tadalafil in combination with ambrisentan did not achieve a satisfactory clinical response at week 24 of the study (N Engl J Med. 2015;373:834-44),” Dr. Hoeper of the Clinic for Respiratory Medicine at Hannover Medical School Germany and his associates wrote. “Furthermore, in the SERAPHIN study, event-free survival of patients receiving [phosphodiesterase-5 inhibitors] monotherapy was approximately 50% at 3 years (N Engl J Med. 2013;369:809-18).”
For the current trial, known as RESPITE, investigators from nine countries in Europe and North America enrolled 61 PAH patients in a 24-week, open-label uncontrolled analysis to investigate the safety, feasibility, and benefit of switching them from phosphodiesterase-5 inhibitors to riociguat. The patients underwent 1-3 days free of phosphodiesterase-5 inhibitors before receiving riociguat in a maximum dose of up to 2.5 mg t.i.d. Most patients (74%) were female, and 92% were Caucasian. In all, 51 patients (84%) completed all 24 weeks of treatment, while the remaining 10 discontinued treatment, 4 of whom due to adverse events.
Additionally, 54% of the patients studied experienced an improvement in their the World Health Organization Functional Class. However, 32 patients (52%) experienced study drug–related adverse events and 10 (16%) experienced serious adverse events, two of which were related to the drug being studied. Six patients (10%) experienced clinical worsening, including death in two, though the deaths were deemed to be unrelated to the drug being studied.
“Although not mechanistically studied, the findings of RESPITE support the hypothesis that a defective [nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate] pathway might explain why some patients have no sufficient or sustained response to [phosphodiesterase-5 inhibitors] therapy,” the researchers noted. “In such patients, direct stimulation of [soluble guanylate cyclase] may be more effective than inhibition of [phosphodiesterase-5], but this hypothesis is still unproven.”
They acknowledged certain limitations of the study, including its prospective design and the relatively homogenous patient population. “Other limitations include the lack of a long-term continuation phase, and the absence of mechanistic data allowing identification of patients likely to respond or not respond to switching,” they wrote. “Two deaths were observed in this study, which might raise concerns, although neither of the deaths (one due to pneumonia and one due to subdural haematoma) was considered by the investigators to be study drug-related or due to worsening PAH. Given the lack of a control group and the rate of study withdrawals and clinical worsening events, further evaluation to clarify the safety of switching is required.”
The study was funded by Bayer AG, Berlin. Dr. Hoeper and his coauthors disclosed having financial ties to numerous pharmaceutical companies, including Bayer, which makes riociguat.
PRIMARY SOURCE: Eur Respir J. 2017 Sep 9. doi: 10.1183/13993003.02425-2016.
Switching to riociguat may be an effective strategy for pulmonary arterial hypertension (PAH) patients who respond inadequately to phosphodiesterase-5 inhibitors, results from a small open-label study demonstrated.
“This study represents an important step towards determining if this new treatment strategy is an effective approach to the management of patients with PAH, although additional data from larger, randomised, controlled studies are needed to further establish the safety and efficacy of this approach,” researchers led by Marius M. Hoeper, MD, wrote in a study published online Sept. 9, 2017, in the European Respiratory Journal.
Current clinical data indicate that many patients with PAH who receive phosphodiesterase-5 inhibitors do not reach treatment goals. “For example, in the AMBITION study, 73% of patients with PAH receiving tadalafil monotherapy and 61% of those receiving tadalafil in combination with ambrisentan did not achieve a satisfactory clinical response at week 24 of the study (N Engl J Med. 2015;373:834-44),” Dr. Hoeper of the Clinic for Respiratory Medicine at Hannover Medical School Germany and his associates wrote. “Furthermore, in the SERAPHIN study, event-free survival of patients receiving [phosphodiesterase-5 inhibitors] monotherapy was approximately 50% at 3 years (N Engl J Med. 2013;369:809-18).”
For the current trial, known as RESPITE, investigators from nine countries in Europe and North America enrolled 61 PAH patients in a 24-week, open-label uncontrolled analysis to investigate the safety, feasibility, and benefit of switching them from phosphodiesterase-5 inhibitors to riociguat. The patients underwent 1-3 days free of phosphodiesterase-5 inhibitors before receiving riociguat in a maximum dose of up to 2.5 mg t.i.d. Most patients (74%) were female, and 92% were Caucasian. In all, 51 patients (84%) completed all 24 weeks of treatment, while the remaining 10 discontinued treatment, 4 of whom due to adverse events.
Additionally, 54% of the patients studied experienced an improvement in their the World Health Organization Functional Class. However, 32 patients (52%) experienced study drug–related adverse events and 10 (16%) experienced serious adverse events, two of which were related to the drug being studied. Six patients (10%) experienced clinical worsening, including death in two, though the deaths were deemed to be unrelated to the drug being studied.
“Although not mechanistically studied, the findings of RESPITE support the hypothesis that a defective [nitric oxide–soluble guanylate cyclase–cyclic guanosine monophosphate] pathway might explain why some patients have no sufficient or sustained response to [phosphodiesterase-5 inhibitors] therapy,” the researchers noted. “In such patients, direct stimulation of [soluble guanylate cyclase] may be more effective than inhibition of [phosphodiesterase-5], but this hypothesis is still unproven.”
They acknowledged certain limitations of the study, including its prospective design and the relatively homogenous patient population. “Other limitations include the lack of a long-term continuation phase, and the absence of mechanistic data allowing identification of patients likely to respond or not respond to switching,” they wrote. “Two deaths were observed in this study, which might raise concerns, although neither of the deaths (one due to pneumonia and one due to subdural haematoma) was considered by the investigators to be study drug-related or due to worsening PAH. Given the lack of a control group and the rate of study withdrawals and clinical worsening events, further evaluation to clarify the safety of switching is required.”
The study was funded by Bayer AG, Berlin. Dr. Hoeper and his coauthors disclosed having financial ties to numerous pharmaceutical companies, including Bayer, which makes riociguat.
PRIMARY SOURCE: Eur Respir J. 2017 Sep 9. doi: 10.1183/13993003.02425-2016.
FROM THE EUROPEAN RESPIRATORY JOURNAL
Key clinical point: Selected patients with pulmonary artery hypertension (PAH) may benefit from switching from phosphodiesterase-5 inhibitors to riociguat.
Major finding: Among patients who completed all 24 weeks of treatment with riociguat, their mean 6-minute walking distance had increased by a mean of 31 meters, and their N-terminal pro b-type natriuretic peptide level decreased by a mean of 347 pg/mL.
Study details: A multicenter, open-label trial of 61 patients with PAH.
Disclosures: The study was funded by Bayer AG, Berlin. Dr. Hoeper and his coauthors disclosed having financial ties to numerous pharmaceutical companies, including Bayer, which makes riociguat.
Source: Marius M. Hoeper, et al. RESPITE: switching to riociguat in pulmonary arterial hypertension patients with inadequate response to phosphdiesterase-5 inhibitors. Eur Resp J. 2017 Sep 09. doi: 10.1183/13993003.02425-2016.
Hospital-led interventions cut pediatric asthma hospitalizations
Hospital-driven interventions designed to improve management of asthma in children achieved significant reductions in monthly asthma-related hospitalizations and emergency department visits, according to a paper published online Sept. 18 in JAMA Pediatrics.
Long-term management of pediatric asthma is challenging, and around 40% of children and adolescents hospitalized with the disease tend to be rehospitalized or revisit the emergency department (ED) within 12 months, according to Carolyn M. Kercsmar, MD, of Children’s Hospital Medical Center in Cincinnati, and her coauthors.
“Traditional care models do not adequately address underlying risk factors, propagating disparities and costly health care use,” they wrote (JAMA Pediatrics 2017, Sep 18. doi: 10.1001/jamapediatrics.2017.2600).
This study, initiated by Cincinnati Children’s Hospital Medical Center, involved a range of interventions implemented with inpatients and outpatients and through the community setting, targeting the region’s more than 36,000 children and adolescents with asthma, approximately 13,000 of whom were Medicaid insured.
Over the 5-year study, researchers saw a 41.8% relative reduction in asthma-related hospitalizations – from 8.1 to 4.7 per 10,000 Medicaid patients per month. Asthma-related visits to the ED decreased by 42.4%, from 21.5 to 12.4 per 10,000 Medicaid patients per month, and the percentage of patients rehospitalized or who returned to the ED for asthma within 30 days declined from 12% to 7%, “within 3 years of implementation of the inpatient care interventions,” the researchers noted.
There was also a significant increase in the percentage of patients discharged with a 30-day supply of inhaled controller medications, from 50% in May 2008 to 90% in May 2010, and the percentage of patients discharged with a short course of oral corticosteroids increased from 0% to 70% by March 2011.
Outpatient processes ensured that Asthma Control Test scores were collected and that patients were provided with asthma action plans. This was associated with an increase in the percentage of patients with well-controlled asthma from 48% to 54%.
“Implementation of an integrated, multilevel approach focused on enhancing availability and accessibility of treatments, removing barriers to adherence, mitigating risks related to adverse exposures, and augmenting self-management and collaborative relationships between the family and the health care system was associated with improved asthma outcomes,” the authors wrote.
Noting that previous research has found 38%-70% of patients do not get their prescribed medications at hospital discharge, the authors said they believed giving a 30-day supply of all daily asthma medications at discharge was a key part of their success.
The study was supported by the Cincinnati Children’s Hospital Medical Center and one author received a grant from the National Institutes of Health. One author declared compensation for a committee role on a study of asthma treatments in children. No other conflicts of interest were declared.
Of importance, any future efforts to replicate this work in a patient-centered way should include consideration of how information on asthma management is communicated to and understood by patients. Standard tools such as asthma action plans often contain language and other information that is inaccessible to populations with low health literacy levels.
After years of elevated morbidity, the work of Kercsmar et al. is a demonstration of how interdisciplinary care focused within a biopsychosocial model can improve outcomes for vulnerable children. Future efforts to replicate these results in other communities should continue to emphasize this patient-centered, biopsychosocial philosophy, with heightened attention to the challenges that remain for children and families.
Dr. Sean M. Frey and Dr. Jill S. Halterman are in the department of pediatrics at the University of Rochester (N.Y.) School of Medicine and Dentistry. These comments are taken from an accompanying editorial (JAMA Pediatrics 2017, Sep 18. doi: 10.1001/jamapediatrics.2017.2609). No conflicts of interest were declared.
Of importance, any future efforts to replicate this work in a patient-centered way should include consideration of how information on asthma management is communicated to and understood by patients. Standard tools such as asthma action plans often contain language and other information that is inaccessible to populations with low health literacy levels.
After years of elevated morbidity, the work of Kercsmar et al. is a demonstration of how interdisciplinary care focused within a biopsychosocial model can improve outcomes for vulnerable children. Future efforts to replicate these results in other communities should continue to emphasize this patient-centered, biopsychosocial philosophy, with heightened attention to the challenges that remain for children and families.
Dr. Sean M. Frey and Dr. Jill S. Halterman are in the department of pediatrics at the University of Rochester (N.Y.) School of Medicine and Dentistry. These comments are taken from an accompanying editorial (JAMA Pediatrics 2017, Sep 18. doi: 10.1001/jamapediatrics.2017.2609). No conflicts of interest were declared.
Of importance, any future efforts to replicate this work in a patient-centered way should include consideration of how information on asthma management is communicated to and understood by patients. Standard tools such as asthma action plans often contain language and other information that is inaccessible to populations with low health literacy levels.
After years of elevated morbidity, the work of Kercsmar et al. is a demonstration of how interdisciplinary care focused within a biopsychosocial model can improve outcomes for vulnerable children. Future efforts to replicate these results in other communities should continue to emphasize this patient-centered, biopsychosocial philosophy, with heightened attention to the challenges that remain for children and families.
Dr. Sean M. Frey and Dr. Jill S. Halterman are in the department of pediatrics at the University of Rochester (N.Y.) School of Medicine and Dentistry. These comments are taken from an accompanying editorial (JAMA Pediatrics 2017, Sep 18. doi: 10.1001/jamapediatrics.2017.2609). No conflicts of interest were declared.
Hospital-driven interventions designed to improve management of asthma in children achieved significant reductions in monthly asthma-related hospitalizations and emergency department visits, according to a paper published online Sept. 18 in JAMA Pediatrics.
Long-term management of pediatric asthma is challenging, and around 40% of children and adolescents hospitalized with the disease tend to be rehospitalized or revisit the emergency department (ED) within 12 months, according to Carolyn M. Kercsmar, MD, of Children’s Hospital Medical Center in Cincinnati, and her coauthors.
“Traditional care models do not adequately address underlying risk factors, propagating disparities and costly health care use,” they wrote (JAMA Pediatrics 2017, Sep 18. doi: 10.1001/jamapediatrics.2017.2600).
This study, initiated by Cincinnati Children’s Hospital Medical Center, involved a range of interventions implemented with inpatients and outpatients and through the community setting, targeting the region’s more than 36,000 children and adolescents with asthma, approximately 13,000 of whom were Medicaid insured.
Over the 5-year study, researchers saw a 41.8% relative reduction in asthma-related hospitalizations – from 8.1 to 4.7 per 10,000 Medicaid patients per month. Asthma-related visits to the ED decreased by 42.4%, from 21.5 to 12.4 per 10,000 Medicaid patients per month, and the percentage of patients rehospitalized or who returned to the ED for asthma within 30 days declined from 12% to 7%, “within 3 years of implementation of the inpatient care interventions,” the researchers noted.
There was also a significant increase in the percentage of patients discharged with a 30-day supply of inhaled controller medications, from 50% in May 2008 to 90% in May 2010, and the percentage of patients discharged with a short course of oral corticosteroids increased from 0% to 70% by March 2011.
Outpatient processes ensured that Asthma Control Test scores were collected and that patients were provided with asthma action plans. This was associated with an increase in the percentage of patients with well-controlled asthma from 48% to 54%.
“Implementation of an integrated, multilevel approach focused on enhancing availability and accessibility of treatments, removing barriers to adherence, mitigating risks related to adverse exposures, and augmenting self-management and collaborative relationships between the family and the health care system was associated with improved asthma outcomes,” the authors wrote.
Noting that previous research has found 38%-70% of patients do not get their prescribed medications at hospital discharge, the authors said they believed giving a 30-day supply of all daily asthma medications at discharge was a key part of their success.
The study was supported by the Cincinnati Children’s Hospital Medical Center and one author received a grant from the National Institutes of Health. One author declared compensation for a committee role on a study of asthma treatments in children. No other conflicts of interest were declared.
Hospital-driven interventions designed to improve management of asthma in children achieved significant reductions in monthly asthma-related hospitalizations and emergency department visits, according to a paper published online Sept. 18 in JAMA Pediatrics.
Long-term management of pediatric asthma is challenging, and around 40% of children and adolescents hospitalized with the disease tend to be rehospitalized or revisit the emergency department (ED) within 12 months, according to Carolyn M. Kercsmar, MD, of Children’s Hospital Medical Center in Cincinnati, and her coauthors.
“Traditional care models do not adequately address underlying risk factors, propagating disparities and costly health care use,” they wrote (JAMA Pediatrics 2017, Sep 18. doi: 10.1001/jamapediatrics.2017.2600).
This study, initiated by Cincinnati Children’s Hospital Medical Center, involved a range of interventions implemented with inpatients and outpatients and through the community setting, targeting the region’s more than 36,000 children and adolescents with asthma, approximately 13,000 of whom were Medicaid insured.
Over the 5-year study, researchers saw a 41.8% relative reduction in asthma-related hospitalizations – from 8.1 to 4.7 per 10,000 Medicaid patients per month. Asthma-related visits to the ED decreased by 42.4%, from 21.5 to 12.4 per 10,000 Medicaid patients per month, and the percentage of patients rehospitalized or who returned to the ED for asthma within 30 days declined from 12% to 7%, “within 3 years of implementation of the inpatient care interventions,” the researchers noted.
There was also a significant increase in the percentage of patients discharged with a 30-day supply of inhaled controller medications, from 50% in May 2008 to 90% in May 2010, and the percentage of patients discharged with a short course of oral corticosteroids increased from 0% to 70% by March 2011.
Outpatient processes ensured that Asthma Control Test scores were collected and that patients were provided with asthma action plans. This was associated with an increase in the percentage of patients with well-controlled asthma from 48% to 54%.
“Implementation of an integrated, multilevel approach focused on enhancing availability and accessibility of treatments, removing barriers to adherence, mitigating risks related to adverse exposures, and augmenting self-management and collaborative relationships between the family and the health care system was associated with improved asthma outcomes,” the authors wrote.
Noting that previous research has found 38%-70% of patients do not get their prescribed medications at hospital discharge, the authors said they believed giving a 30-day supply of all daily asthma medications at discharge was a key part of their success.
The study was supported by the Cincinnati Children’s Hospital Medical Center and one author received a grant from the National Institutes of Health. One author declared compensation for a committee role on a study of asthma treatments in children. No other conflicts of interest were declared.
FROM JAMA PEDIATRICS
Key clinical point: A hospital-driven intervention to improve management of asthma in children has achieved significant reductions in asthma-related hospitalizations and emergency department visits and increased medication uptake.
Major finding: A multifactorial intervention to improve asthma management in children was associated with a 41.8% relative reduction in asthma-related hospitalizations and a 42.4% reduction in emergency department visits.
Data source: A hospital-based intervention.
Disclosures: The study was supported by the Cincinnati Children’s Hospital Medical Center and one author received a grant from the National Institutes of Health. One author declared compensation for a committee role on a study of asthma treatments in children. No other conflicts of interest were declared.
COPD in Primary Care
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Tezepelumab reduces exacerbations in persistent, treatment-resistant asthma
Patients whose asthma remains uncontrolled despite treatment may benefit from a new monoclonal antibody that targets an inflammatory cytokine known to be promoted in asthmatic airways, according to data presented at the annual congress of the European Respiratory Society.
Writing in the Sept. 7 issue of the New England Journal of Medicine, researchers reported on a phase 2, randomized placebo-controlled trial of three dosing regimens of subcutaneous tezepelumab, which targets the epithelial cell–derived cytokine thymic stromal lymphopoietin (TSLP). The trial involved 584 patients with uncontrolled asthma, despite treatment with long-acting beta-agonists and medium to high doses of inhaled glucocorticoids.
At 70 mg every 4 weeks, exacerbation rates were 61% lower than in the placebo group; at 210 mg every 4 weeks, they were 71% lower; and at 280 mg every 2 weeks, they were 66% lower (P was less than 001 in comparisons between each group and the placebo).
The overall annualized exacerbation rates by week 52 were 0.26 for the 70-mg group, 0.19 for the 210-mg group, and 0.22 for the 280-mg group, compared with 0.67 in the placebo group, regardless of a patient’s baseline eosinophil count. Patients treated with tezepelumab had a longer time to first asthma exacerbation. They also experienced a significantly higher change from baseline in their prebronchodilator forced expiratory volume in 1 second at week 52, when compared with patients on the placebo.
“The observed improvements in disease control in patients who received tezepelumab highlight the potential pathogenic role of TSLP across different asthma phenotypes,” reported Jonathan Corren, MD, of the University of California, Los Angeles, and his coauthors. “... Although TSLP is central to the regulation of type 2 immunity, many cell types that are activated by or respond to TSLP, such as mast cells, basophils, natural killer T cells, innate lymphoid cells, and neutrophils, may play a role in inflammation in asthma beyond type 2 inflammation.”
The incidences of adverse events and serious adverse events were similar across all groups in the study. Three serious adverse events – pneumonia and stroke in the same patient and one case of Guillain-Barré syndrome – in patients taking tezepelumab, were deemed to be related to the treatment.
The study was supported by tezepelumab manufacturers MedImmune (a member of the AstraZeneca group) and Amgen. Six of the seven authors are employees of MedImmune or Amgen. One author declared support and honoraria from several pharmaceutical companies, one declared a related patent, and five also had stock options in either MedImmune or Amgen.
Tezepelumab is the first biologic that has a substantial positive effect on two important markers of the inflammation of asthma – namely, blood eosinophil counts and the fraction of exhaled nitric oxide. It appears to be the broadest and most promising biologic for the treatment of persistent uncontrolled asthma to date.
The observation that tezepelumab reduces the level of both inflammatory markers shows that it hits a more upstream target and that it blocks at least two relevant inflammatory pathways in asthma. This is likely to be clinically relevant, since simultaneously increased exhaled nitric oxide levels and blood eosinophil counts are related to increased morbidity due to asthma.
Elisabeth H. Bel, MD, PhD, is with the department of respiratory medicine, Academic Medical Center, the University of Amsterdam. These comments were taken from an accompanying editorial (N Engl J Med. 2017;377:989-91). Dr. Bel declared consultancies and grants from pharmaceutical companies including AstraZeneca.
Tezepelumab is the first biologic that has a substantial positive effect on two important markers of the inflammation of asthma – namely, blood eosinophil counts and the fraction of exhaled nitric oxide. It appears to be the broadest and most promising biologic for the treatment of persistent uncontrolled asthma to date.
The observation that tezepelumab reduces the level of both inflammatory markers shows that it hits a more upstream target and that it blocks at least two relevant inflammatory pathways in asthma. This is likely to be clinically relevant, since simultaneously increased exhaled nitric oxide levels and blood eosinophil counts are related to increased morbidity due to asthma.
Elisabeth H. Bel, MD, PhD, is with the department of respiratory medicine, Academic Medical Center, the University of Amsterdam. These comments were taken from an accompanying editorial (N Engl J Med. 2017;377:989-91). Dr. Bel declared consultancies and grants from pharmaceutical companies including AstraZeneca.
Tezepelumab is the first biologic that has a substantial positive effect on two important markers of the inflammation of asthma – namely, blood eosinophil counts and the fraction of exhaled nitric oxide. It appears to be the broadest and most promising biologic for the treatment of persistent uncontrolled asthma to date.
The observation that tezepelumab reduces the level of both inflammatory markers shows that it hits a more upstream target and that it blocks at least two relevant inflammatory pathways in asthma. This is likely to be clinically relevant, since simultaneously increased exhaled nitric oxide levels and blood eosinophil counts are related to increased morbidity due to asthma.
Elisabeth H. Bel, MD, PhD, is with the department of respiratory medicine, Academic Medical Center, the University of Amsterdam. These comments were taken from an accompanying editorial (N Engl J Med. 2017;377:989-91). Dr. Bel declared consultancies and grants from pharmaceutical companies including AstraZeneca.
Patients whose asthma remains uncontrolled despite treatment may benefit from a new monoclonal antibody that targets an inflammatory cytokine known to be promoted in asthmatic airways, according to data presented at the annual congress of the European Respiratory Society.
Writing in the Sept. 7 issue of the New England Journal of Medicine, researchers reported on a phase 2, randomized placebo-controlled trial of three dosing regimens of subcutaneous tezepelumab, which targets the epithelial cell–derived cytokine thymic stromal lymphopoietin (TSLP). The trial involved 584 patients with uncontrolled asthma, despite treatment with long-acting beta-agonists and medium to high doses of inhaled glucocorticoids.
At 70 mg every 4 weeks, exacerbation rates were 61% lower than in the placebo group; at 210 mg every 4 weeks, they were 71% lower; and at 280 mg every 2 weeks, they were 66% lower (P was less than 001 in comparisons between each group and the placebo).
The overall annualized exacerbation rates by week 52 were 0.26 for the 70-mg group, 0.19 for the 210-mg group, and 0.22 for the 280-mg group, compared with 0.67 in the placebo group, regardless of a patient’s baseline eosinophil count. Patients treated with tezepelumab had a longer time to first asthma exacerbation. They also experienced a significantly higher change from baseline in their prebronchodilator forced expiratory volume in 1 second at week 52, when compared with patients on the placebo.
“The observed improvements in disease control in patients who received tezepelumab highlight the potential pathogenic role of TSLP across different asthma phenotypes,” reported Jonathan Corren, MD, of the University of California, Los Angeles, and his coauthors. “... Although TSLP is central to the regulation of type 2 immunity, many cell types that are activated by or respond to TSLP, such as mast cells, basophils, natural killer T cells, innate lymphoid cells, and neutrophils, may play a role in inflammation in asthma beyond type 2 inflammation.”
The incidences of adverse events and serious adverse events were similar across all groups in the study. Three serious adverse events – pneumonia and stroke in the same patient and one case of Guillain-Barré syndrome – in patients taking tezepelumab, were deemed to be related to the treatment.
The study was supported by tezepelumab manufacturers MedImmune (a member of the AstraZeneca group) and Amgen. Six of the seven authors are employees of MedImmune or Amgen. One author declared support and honoraria from several pharmaceutical companies, one declared a related patent, and five also had stock options in either MedImmune or Amgen.
Patients whose asthma remains uncontrolled despite treatment may benefit from a new monoclonal antibody that targets an inflammatory cytokine known to be promoted in asthmatic airways, according to data presented at the annual congress of the European Respiratory Society.
Writing in the Sept. 7 issue of the New England Journal of Medicine, researchers reported on a phase 2, randomized placebo-controlled trial of three dosing regimens of subcutaneous tezepelumab, which targets the epithelial cell–derived cytokine thymic stromal lymphopoietin (TSLP). The trial involved 584 patients with uncontrolled asthma, despite treatment with long-acting beta-agonists and medium to high doses of inhaled glucocorticoids.
At 70 mg every 4 weeks, exacerbation rates were 61% lower than in the placebo group; at 210 mg every 4 weeks, they were 71% lower; and at 280 mg every 2 weeks, they were 66% lower (P was less than 001 in comparisons between each group and the placebo).
The overall annualized exacerbation rates by week 52 were 0.26 for the 70-mg group, 0.19 for the 210-mg group, and 0.22 for the 280-mg group, compared with 0.67 in the placebo group, regardless of a patient’s baseline eosinophil count. Patients treated with tezepelumab had a longer time to first asthma exacerbation. They also experienced a significantly higher change from baseline in their prebronchodilator forced expiratory volume in 1 second at week 52, when compared with patients on the placebo.
“The observed improvements in disease control in patients who received tezepelumab highlight the potential pathogenic role of TSLP across different asthma phenotypes,” reported Jonathan Corren, MD, of the University of California, Los Angeles, and his coauthors. “... Although TSLP is central to the regulation of type 2 immunity, many cell types that are activated by or respond to TSLP, such as mast cells, basophils, natural killer T cells, innate lymphoid cells, and neutrophils, may play a role in inflammation in asthma beyond type 2 inflammation.”
The incidences of adverse events and serious adverse events were similar across all groups in the study. Three serious adverse events – pneumonia and stroke in the same patient and one case of Guillain-Barré syndrome – in patients taking tezepelumab, were deemed to be related to the treatment.
The study was supported by tezepelumab manufacturers MedImmune (a member of the AstraZeneca group) and Amgen. Six of the seven authors are employees of MedImmune or Amgen. One author declared support and honoraria from several pharmaceutical companies, one declared a related patent, and five also had stock options in either MedImmune or Amgen.
FROM THE ERS CONGRESS 2017
Key clinical point: The monoclonal antibody tezepelumab is associated with a significant reduction in asthma exacerbations in patients with treatment-resistant and persistent disease.
Major finding: Patients treated with tezepelumab had a 34% reduction in the risk of asthma exacerbations, compared with those on placebo.
Data source: A phase 2, randomized placebo-controlled trial in 584 patients with persistent asthma.
Disclosures: The study was supported by tezepelumab manufacturers MedImmune (a member of the AstraZeneca group) and Amgen. Six of the seven authors are employees of MedImmune or Amgen. One author declared support and honoraria from several pharmaceutical companies, one declared a related patent, and five also had stock options in either MedImmune or Amgen.
FDA approves biosimilar to bevacizumab
The Food and Drug Administration has approved a biosimilar to bevacizumab (Avastin) for the treatment of certain colorectal, lung, brain, kidney, and cervical cancers.
Bevacizumab-awwb is the first biosimilar approved in the United States for the treatment of cancer, the FDA said in a press release.
Approval is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrate bevacizumab-awwb is biosimilar to bevacizumab, the FDA said.
• Metastatic colorectal cancer, in combination with intravenous 5-fluorouracil-based chemotherapy for first- or second-line treatment.
• Metastatic colorectal cancer, in combination with fluoropyrimidine-irinotecan–based or fluoropyrimidine-oxaliplatin–based chemotherapy for the second-line treatment of patients who have progressed on a first-line bevacizumab product–containing regimen.
• Non-squamous non–small cell lung cancer, in combination with carboplatin and paclitaxel for first line treatment of unresectable, locally advanced, recurrent, or metastatic disease.
• Glioblastoma with progressive disease following prior therapy, based on improvement in objective response rate.
• Metastatic renal cell carcinoma, in combination with interferon alfa.
• Cervical cancer that is persistent, recurrent, or metastatic, in combination with paclitaxel and cisplatin or paclitaxel and topotecan.
Common expected side effects of the biosimilar include epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Serious expected side effects include perforation or fistula, arterial and venous thromboembolic events, hypertension, posterior reversible encephalopathy syndrome, proteinuria, infusion-related reactions, and ovarian failure. Women who are pregnant should not take bevacizumab-awwb.
The biosimilar to bevacizumab carries a similar boxed warning regarding the increased risk of gastrointestinal perforations; surgery and wound healing complications; and severe or fatal pulmonary, gastrointestinal, central nervous system, and vaginal hemorrhage.
The biosimilar approval was granted to Amgen, which will market the drug under the trade name Mvasi.
The Food and Drug Administration has approved a biosimilar to bevacizumab (Avastin) for the treatment of certain colorectal, lung, brain, kidney, and cervical cancers.
Bevacizumab-awwb is the first biosimilar approved in the United States for the treatment of cancer, the FDA said in a press release.
Approval is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrate bevacizumab-awwb is biosimilar to bevacizumab, the FDA said.
• Metastatic colorectal cancer, in combination with intravenous 5-fluorouracil-based chemotherapy for first- or second-line treatment.
• Metastatic colorectal cancer, in combination with fluoropyrimidine-irinotecan–based or fluoropyrimidine-oxaliplatin–based chemotherapy for the second-line treatment of patients who have progressed on a first-line bevacizumab product–containing regimen.
• Non-squamous non–small cell lung cancer, in combination with carboplatin and paclitaxel for first line treatment of unresectable, locally advanced, recurrent, or metastatic disease.
• Glioblastoma with progressive disease following prior therapy, based on improvement in objective response rate.
• Metastatic renal cell carcinoma, in combination with interferon alfa.
• Cervical cancer that is persistent, recurrent, or metastatic, in combination with paclitaxel and cisplatin or paclitaxel and topotecan.
Common expected side effects of the biosimilar include epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Serious expected side effects include perforation or fistula, arterial and venous thromboembolic events, hypertension, posterior reversible encephalopathy syndrome, proteinuria, infusion-related reactions, and ovarian failure. Women who are pregnant should not take bevacizumab-awwb.
The biosimilar to bevacizumab carries a similar boxed warning regarding the increased risk of gastrointestinal perforations; surgery and wound healing complications; and severe or fatal pulmonary, gastrointestinal, central nervous system, and vaginal hemorrhage.
The biosimilar approval was granted to Amgen, which will market the drug under the trade name Mvasi.
The Food and Drug Administration has approved a biosimilar to bevacizumab (Avastin) for the treatment of certain colorectal, lung, brain, kidney, and cervical cancers.
Bevacizumab-awwb is the first biosimilar approved in the United States for the treatment of cancer, the FDA said in a press release.
Approval is based on structural and functional characterization, animal study data, human pharmacokinetic and pharmacodynamics data, clinical immunogenicity data, and other clinical safety and effectiveness data that demonstrate bevacizumab-awwb is biosimilar to bevacizumab, the FDA said.
• Metastatic colorectal cancer, in combination with intravenous 5-fluorouracil-based chemotherapy for first- or second-line treatment.
• Metastatic colorectal cancer, in combination with fluoropyrimidine-irinotecan–based or fluoropyrimidine-oxaliplatin–based chemotherapy for the second-line treatment of patients who have progressed on a first-line bevacizumab product–containing regimen.
• Non-squamous non–small cell lung cancer, in combination with carboplatin and paclitaxel for first line treatment of unresectable, locally advanced, recurrent, or metastatic disease.
• Glioblastoma with progressive disease following prior therapy, based on improvement in objective response rate.
• Metastatic renal cell carcinoma, in combination with interferon alfa.
• Cervical cancer that is persistent, recurrent, or metastatic, in combination with paclitaxel and cisplatin or paclitaxel and topotecan.
Common expected side effects of the biosimilar include epistaxis, headache, hypertension, rhinitis, proteinuria, taste alteration, dry skin, hemorrhage, lacrimation disorder, back pain, and exfoliative dermatitis.
Serious expected side effects include perforation or fistula, arterial and venous thromboembolic events, hypertension, posterior reversible encephalopathy syndrome, proteinuria, infusion-related reactions, and ovarian failure. Women who are pregnant should not take bevacizumab-awwb.
The biosimilar to bevacizumab carries a similar boxed warning regarding the increased risk of gastrointestinal perforations; surgery and wound healing complications; and severe or fatal pulmonary, gastrointestinal, central nervous system, and vaginal hemorrhage.
The biosimilar approval was granted to Amgen, which will market the drug under the trade name Mvasi.
Bedside imaging allowed for individualized PEEP adjustments
A noninvasive bedside imaging technique can individually calibrate positive end-expiratory pressure settings in patients on extracorporeal membrane oxygenation (ECMO) for severe acute respiratory distress syndrome (ARDS), a study showed.
The step-down PEEP (positive end-expiratory pressure) trial could not identify a single PEEP setting that optimally balanced lung overdistension and lung collapse for all 15 patients. But, electrical impedance tomography (EIT) allowed investigators to individually titrate PEEP settings for each patient, Guillaume Franchineau, MD, wrote (Am J Respir Crit Care Med. 2017;196[4]:447-57 doi: 10.1164/rccm.201605-1055OC).
The 4-month study involved 15 patients (aged, 18-79 years) who were in acute respiratory distress syndrome for a variety of reasons, including influenza (7 patients), pneumonia (3), leukemia (2), and 1 case each of Pneumocystis, antisynthetase syndrome, and trauma. All patients were receiving ECMO with a constant driving pressure of 14 cm H2O. After verifying that the inspiratory flow was 0 at the end of inspiration, PEEP was increased to 20 cm H2O (PEEP 20) with a peak inspiratory pressure of 34 cm H2O. PEEP 20 was held for 20 minutes and then lowered by 5 cm H2O decrements with the potential of reaching PEEP 0.
The EIT device, consisting of a silicone belt with 16 surface electrodes, was placed around the thorax aligning with the sixth intercostal parasternal space and connected to a monitor. By measuring conductivity and impeditivity in the underlying tissues, the device generates a low-resolution, two-dimensional image. The image was sufficient to show lung distension and collapse as the PEEP settings changed. Investigators looked for the best compromise between overdistension and collapsed zones, which they defined as the lowest pressure able to limit EIT-assessed collapse to no more than 15% with the least overdistension.
There was no one-size-fits-all PEEP setting, the authors found. The setting that minimized both overdistension and collapse was PEEP 15 in seven patients, PEEP 10 in six patients, and PEEP 5 in two patients.
At each patient’s optimal PEEP setting, the median tidal volume was similar: 3.8 mL/kg ideal body weight for PEEP 15, 3.9 mL/kg ideal body weight for PEEP 10, and 4.3 mL/kg ideal body weight for PEEP 5.
Respiratory system compliance was also similar among the groups, at 20 mL/cm H2O, 18 mL/cm H2O, and 21 mL/cm H2O, respectively. However, arterial partial pressure of oxygen decreased as the PEEP setting decreased, dropping from 148 mm Hg to 128 mm Hg to 100 mm Hg, respectively. Conversely, arterial partial pressure of CO2 increased (32-41 mm Hg).
EIT also allowed clinicians to pinpoint areas of distension or collapse. As PEEP decreased, there was steady ventilation loss in the medial-dorsal and dorsal regions, which shifted to the medial-ventral and ventral regions.
“Most end-expiratory lung impedances were located in medial-dorsal and medial-ventral regions, whereas the dorsal region constantly contributed less than 10% of total end-expiratory lung impedance,” the authors noted.
“The broad variability of EIT-based best compromise PEEPs in these patients with severe ARDS reinforces the need to provide ventilation settings individually tailored to the regional ARDS-lesion distribution,” they concluded. “To achieve that goal, EIT seems to be an interesting bedside noninvasive tool to provide real-time monitoring of the PEEP effect and ventilation distribution on ECMO.”
Dr. Franchineau reported receiving speakers fees from Mapquet.
[email protected]
On Twitter @Alz_Gal
This first study to examine electrical impedance tomography (EIT) in patients under extracorporeal membrane oxygenation shows important clinical potential, but also raises important questions, Claude Guerin, MD, wrote in an accompanying editorial. (Am J Respir Crit Care Med. doi: 10.1164/rccm.201701-0167ed).
The ability to titrate PEEP settings to a patient’s individual needs could substantially reduce the risk of lung derecruitment or damage by overdistension.
The current study, however, has limitations that must be addressed in the next phase of research, before this technique can be adopted into clinical practice, Dr. Guerin said: The 5-cm H20 PEEP steps may be too large to detect relevant changes.
In several other studies, PEEP was reduced more gradually in 2- to 3-cm H2O increments. “Surprisingly, PEEP was reduced to 0 cm H2O in this study, with this step maintained for 20 minutes, raising the risk of derecruitment and further stretching once higher PEEP levels were resumed.”
The investigators did not perform any recruitment maneuvers before proceeding with PEEP adjustment. This is contrary to what has been done in prior animal and human studies.
The computation of driving pressure was done without taking total PEEP into account. “As total PEEP is frequently greater than PEEP in patients with [acute respiratory distress syndrome], driving pressure can be overestimated with the common computation.”
The optimal PEEP that the investigators aimed for was determined retrospectively from an offline analysis of the data; this technique would not be suitable for bedside management. “When ‘optimal’ PEEP was defined from [EIT criteria], from a higher PaO2 [arterial partial pressure of oxygen] or from a higher compliance of the respiratory system during the decremental PEEP trial, these three criteria were observed together in only four patients with [acute respiratory distress syndrome].”
The study was done only once and cannot comply with the need for regular PEEP-level assessments over time, as could be done with some other strategies.
“Further studies should also consider taking into account the role of chest wall mechanics,” Dr. Guerin said.
Nevertheless, he concluded, EIT-based PEEP titration for each individual patient represents a prospective tool for assisting with the treatment of acute respiratory distress syndrome, and should be fully investigated in a large, prospective trial.
Dr. Guerin is a pulmonologist at the Hospital de la Croix Rousse, Lyon, France. He had no relevant financial disclosures.
This first study to examine electrical impedance tomography (EIT) in patients under extracorporeal membrane oxygenation shows important clinical potential, but also raises important questions, Claude Guerin, MD, wrote in an accompanying editorial. (Am J Respir Crit Care Med. doi: 10.1164/rccm.201701-0167ed).
The ability to titrate PEEP settings to a patient’s individual needs could substantially reduce the risk of lung derecruitment or damage by overdistension.
The current study, however, has limitations that must be addressed in the next phase of research, before this technique can be adopted into clinical practice, Dr. Guerin said: The 5-cm H20 PEEP steps may be too large to detect relevant changes.
In several other studies, PEEP was reduced more gradually in 2- to 3-cm H2O increments. “Surprisingly, PEEP was reduced to 0 cm H2O in this study, with this step maintained for 20 minutes, raising the risk of derecruitment and further stretching once higher PEEP levels were resumed.”
The investigators did not perform any recruitment maneuvers before proceeding with PEEP adjustment. This is contrary to what has been done in prior animal and human studies.
The computation of driving pressure was done without taking total PEEP into account. “As total PEEP is frequently greater than PEEP in patients with [acute respiratory distress syndrome], driving pressure can be overestimated with the common computation.”
The optimal PEEP that the investigators aimed for was determined retrospectively from an offline analysis of the data; this technique would not be suitable for bedside management. “When ‘optimal’ PEEP was defined from [EIT criteria], from a higher PaO2 [arterial partial pressure of oxygen] or from a higher compliance of the respiratory system during the decremental PEEP trial, these three criteria were observed together in only four patients with [acute respiratory distress syndrome].”
The study was done only once and cannot comply with the need for regular PEEP-level assessments over time, as could be done with some other strategies.
“Further studies should also consider taking into account the role of chest wall mechanics,” Dr. Guerin said.
Nevertheless, he concluded, EIT-based PEEP titration for each individual patient represents a prospective tool for assisting with the treatment of acute respiratory distress syndrome, and should be fully investigated in a large, prospective trial.
Dr. Guerin is a pulmonologist at the Hospital de la Croix Rousse, Lyon, France. He had no relevant financial disclosures.
This first study to examine electrical impedance tomography (EIT) in patients under extracorporeal membrane oxygenation shows important clinical potential, but also raises important questions, Claude Guerin, MD, wrote in an accompanying editorial. (Am J Respir Crit Care Med. doi: 10.1164/rccm.201701-0167ed).
The ability to titrate PEEP settings to a patient’s individual needs could substantially reduce the risk of lung derecruitment or damage by overdistension.
The current study, however, has limitations that must be addressed in the next phase of research, before this technique can be adopted into clinical practice, Dr. Guerin said: The 5-cm H20 PEEP steps may be too large to detect relevant changes.
In several other studies, PEEP was reduced more gradually in 2- to 3-cm H2O increments. “Surprisingly, PEEP was reduced to 0 cm H2O in this study, with this step maintained for 20 minutes, raising the risk of derecruitment and further stretching once higher PEEP levels were resumed.”
The investigators did not perform any recruitment maneuvers before proceeding with PEEP adjustment. This is contrary to what has been done in prior animal and human studies.
The computation of driving pressure was done without taking total PEEP into account. “As total PEEP is frequently greater than PEEP in patients with [acute respiratory distress syndrome], driving pressure can be overestimated with the common computation.”
The optimal PEEP that the investigators aimed for was determined retrospectively from an offline analysis of the data; this technique would not be suitable for bedside management. “When ‘optimal’ PEEP was defined from [EIT criteria], from a higher PaO2 [arterial partial pressure of oxygen] or from a higher compliance of the respiratory system during the decremental PEEP trial, these three criteria were observed together in only four patients with [acute respiratory distress syndrome].”
The study was done only once and cannot comply with the need for regular PEEP-level assessments over time, as could be done with some other strategies.
“Further studies should also consider taking into account the role of chest wall mechanics,” Dr. Guerin said.
Nevertheless, he concluded, EIT-based PEEP titration for each individual patient represents a prospective tool for assisting with the treatment of acute respiratory distress syndrome, and should be fully investigated in a large, prospective trial.
Dr. Guerin is a pulmonologist at the Hospital de la Croix Rousse, Lyon, France. He had no relevant financial disclosures.
A noninvasive bedside imaging technique can individually calibrate positive end-expiratory pressure settings in patients on extracorporeal membrane oxygenation (ECMO) for severe acute respiratory distress syndrome (ARDS), a study showed.
The step-down PEEP (positive end-expiratory pressure) trial could not identify a single PEEP setting that optimally balanced lung overdistension and lung collapse for all 15 patients. But, electrical impedance tomography (EIT) allowed investigators to individually titrate PEEP settings for each patient, Guillaume Franchineau, MD, wrote (Am J Respir Crit Care Med. 2017;196[4]:447-57 doi: 10.1164/rccm.201605-1055OC).
The 4-month study involved 15 patients (aged, 18-79 years) who were in acute respiratory distress syndrome for a variety of reasons, including influenza (7 patients), pneumonia (3), leukemia (2), and 1 case each of Pneumocystis, antisynthetase syndrome, and trauma. All patients were receiving ECMO with a constant driving pressure of 14 cm H2O. After verifying that the inspiratory flow was 0 at the end of inspiration, PEEP was increased to 20 cm H2O (PEEP 20) with a peak inspiratory pressure of 34 cm H2O. PEEP 20 was held for 20 minutes and then lowered by 5 cm H2O decrements with the potential of reaching PEEP 0.
The EIT device, consisting of a silicone belt with 16 surface electrodes, was placed around the thorax aligning with the sixth intercostal parasternal space and connected to a monitor. By measuring conductivity and impeditivity in the underlying tissues, the device generates a low-resolution, two-dimensional image. The image was sufficient to show lung distension and collapse as the PEEP settings changed. Investigators looked for the best compromise between overdistension and collapsed zones, which they defined as the lowest pressure able to limit EIT-assessed collapse to no more than 15% with the least overdistension.
There was no one-size-fits-all PEEP setting, the authors found. The setting that minimized both overdistension and collapse was PEEP 15 in seven patients, PEEP 10 in six patients, and PEEP 5 in two patients.
At each patient’s optimal PEEP setting, the median tidal volume was similar: 3.8 mL/kg ideal body weight for PEEP 15, 3.9 mL/kg ideal body weight for PEEP 10, and 4.3 mL/kg ideal body weight for PEEP 5.
Respiratory system compliance was also similar among the groups, at 20 mL/cm H2O, 18 mL/cm H2O, and 21 mL/cm H2O, respectively. However, arterial partial pressure of oxygen decreased as the PEEP setting decreased, dropping from 148 mm Hg to 128 mm Hg to 100 mm Hg, respectively. Conversely, arterial partial pressure of CO2 increased (32-41 mm Hg).
EIT also allowed clinicians to pinpoint areas of distension or collapse. As PEEP decreased, there was steady ventilation loss in the medial-dorsal and dorsal regions, which shifted to the medial-ventral and ventral regions.
“Most end-expiratory lung impedances were located in medial-dorsal and medial-ventral regions, whereas the dorsal region constantly contributed less than 10% of total end-expiratory lung impedance,” the authors noted.
“The broad variability of EIT-based best compromise PEEPs in these patients with severe ARDS reinforces the need to provide ventilation settings individually tailored to the regional ARDS-lesion distribution,” they concluded. “To achieve that goal, EIT seems to be an interesting bedside noninvasive tool to provide real-time monitoring of the PEEP effect and ventilation distribution on ECMO.”
Dr. Franchineau reported receiving speakers fees from Mapquet.
[email protected]
On Twitter @Alz_Gal
A noninvasive bedside imaging technique can individually calibrate positive end-expiratory pressure settings in patients on extracorporeal membrane oxygenation (ECMO) for severe acute respiratory distress syndrome (ARDS), a study showed.
The step-down PEEP (positive end-expiratory pressure) trial could not identify a single PEEP setting that optimally balanced lung overdistension and lung collapse for all 15 patients. But, electrical impedance tomography (EIT) allowed investigators to individually titrate PEEP settings for each patient, Guillaume Franchineau, MD, wrote (Am J Respir Crit Care Med. 2017;196[4]:447-57 doi: 10.1164/rccm.201605-1055OC).
The 4-month study involved 15 patients (aged, 18-79 years) who were in acute respiratory distress syndrome for a variety of reasons, including influenza (7 patients), pneumonia (3), leukemia (2), and 1 case each of Pneumocystis, antisynthetase syndrome, and trauma. All patients were receiving ECMO with a constant driving pressure of 14 cm H2O. After verifying that the inspiratory flow was 0 at the end of inspiration, PEEP was increased to 20 cm H2O (PEEP 20) with a peak inspiratory pressure of 34 cm H2O. PEEP 20 was held for 20 minutes and then lowered by 5 cm H2O decrements with the potential of reaching PEEP 0.
The EIT device, consisting of a silicone belt with 16 surface electrodes, was placed around the thorax aligning with the sixth intercostal parasternal space and connected to a monitor. By measuring conductivity and impeditivity in the underlying tissues, the device generates a low-resolution, two-dimensional image. The image was sufficient to show lung distension and collapse as the PEEP settings changed. Investigators looked for the best compromise between overdistension and collapsed zones, which they defined as the lowest pressure able to limit EIT-assessed collapse to no more than 15% with the least overdistension.
There was no one-size-fits-all PEEP setting, the authors found. The setting that minimized both overdistension and collapse was PEEP 15 in seven patients, PEEP 10 in six patients, and PEEP 5 in two patients.
At each patient’s optimal PEEP setting, the median tidal volume was similar: 3.8 mL/kg ideal body weight for PEEP 15, 3.9 mL/kg ideal body weight for PEEP 10, and 4.3 mL/kg ideal body weight for PEEP 5.
Respiratory system compliance was also similar among the groups, at 20 mL/cm H2O, 18 mL/cm H2O, and 21 mL/cm H2O, respectively. However, arterial partial pressure of oxygen decreased as the PEEP setting decreased, dropping from 148 mm Hg to 128 mm Hg to 100 mm Hg, respectively. Conversely, arterial partial pressure of CO2 increased (32-41 mm Hg).
EIT also allowed clinicians to pinpoint areas of distension or collapse. As PEEP decreased, there was steady ventilation loss in the medial-dorsal and dorsal regions, which shifted to the medial-ventral and ventral regions.
“Most end-expiratory lung impedances were located in medial-dorsal and medial-ventral regions, whereas the dorsal region constantly contributed less than 10% of total end-expiratory lung impedance,” the authors noted.
“The broad variability of EIT-based best compromise PEEPs in these patients with severe ARDS reinforces the need to provide ventilation settings individually tailored to the regional ARDS-lesion distribution,” they concluded. “To achieve that goal, EIT seems to be an interesting bedside noninvasive tool to provide real-time monitoring of the PEEP effect and ventilation distribution on ECMO.”
Dr. Franchineau reported receiving speakers fees from Mapquet.
[email protected]
On Twitter @Alz_Gal
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Key clinical point:
Major finding: The PEEP settings that minimized both overdistension and collapse were PEEP 15 in seven patients, PEEP 10 in six patients, and PEEP 5 in two patients.
Data source: A prospective study of 15 patients.
Disclosures: Dr. Franchineau reported receiving speakers fees from Mapquet. Dr. Guerin had no relevant financial disclosures.
Making Practice Perfect Download
Please click either of the links below to download your free eBook