User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Cases of Legionnaires’ continue to rise in the United States
SAN DIEGO –
“Improved testing and surveillance are needed to improve understanding of disease and outbreak burden,” Laura A. Cooley, MD, said at an annual scientific meeting on infectious diseases. “There is more to learn about environmental sources of Legionella for cases not associated with known outbreaks and about the distribution of Legionella in the environment.”
A Gram-negative bacillus, Legionella is an intracellular parasite of free-living protozoa primarily found in freshwater. “It can live and grow in biofilm, and there are more than 60 species of the bacterium,” she said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Cases are higher in the warmer months, and the rates are highest among the elderly, men, and those of black race. Currently, L. pneumophila accounts for about 90% of cases in the United States. “Once it’s transmitted, it has to hit a susceptible population to cause disease, generally older individuals and people with underlying conditions,” Dr. Cooley said.
A separate analysis evaluated Legionella cases reported among U.S. residents between 2005 and 2009 (MMWR. 2011;60[32]:1083-6). It found that only 4% were associated with outbreaks, and 96% were sporadic. “That doesn’t mean that [the cases] weren’t associated with the same kind of source, they just weren’t identified as an outbreak,” Dr. Cooley said. “It shows that there is a lot to learn about transmission of Legionella.”
Data from the National Notifiable Diseases Surveillance System indicate that rates of Legionella continue to rise nationwide, especially in the Midwest and Northeast. “Why? It’s possible that there are differences in testing preferences and reporting preferences in this region of the country,” Dr. Cooley noted. “Maybe people are more tuned in to the potential for outbreaks, but there are reasons why there could be differences in disease, like differences in infrastructure, climate, population density, and cooling tower density.” CDC data from 2015 indicate that most cases are not associated with a known exposure, and that the case fatality rate differs by exposure type: 12% for cases reporting health care exposure during the 10 days before symptom onset (25% for definite cases), 9% for cases reporting assisted or senior living exposure, 7% when no specific exposure is reported, and 4% for cases reporting travel exposure (MMWR. 2017;66[22]:584-9).
The U.S. case definition of Legionnaires’ disease consists of clinical or radiologic pneumonia plus confirmatory laboratory testing, either by urinary antigen test (UAT), lower respiratory culture, or appropriate serological testing. Polymerase chain reaction can be used as a presumptive test for a suspect case. “UAT is easy and it detects L. pneumophila serogroup 1 (Lp1), but it has some gaps,” Dr. Cooley said. “It isn’t completely sensitive for Lp1, and it doesn’t detect any other species or serogroups. That’s why we also recommend that a culture of respiratory secretions on selective media be performed at the same time. That being said, in the U.S., nearly all reported cases of Legionella are diagnosed by UAT only.”
A 2016 CDC MMWR and Vital Signs report found that almost all Legionella outbreaks could be prevented with effective water management, and the CDC has published a step-by-step guide to creating a water management program to reduce Legionella growth and spread in buildings. The 2017 MMWR Report found that definite health care–associated Legionnaires’ disease is deadly for one in four people who get it. The report also found that this issue is widespread; 76% of complete reporting jurisdictions reported at least one definite case of health care–associated Legionella disease in 2015. More recently, the Centers for Medicare & Medicaid Services issued a requirement to reduce risk in health care facility water systems to prevent cases and outbreaks. It applies to hospitals, skilled nursing facilities, and critical access hospitals.
Dr. Cooley reported having no financial disclosures.
SAN DIEGO –
“Improved testing and surveillance are needed to improve understanding of disease and outbreak burden,” Laura A. Cooley, MD, said at an annual scientific meeting on infectious diseases. “There is more to learn about environmental sources of Legionella for cases not associated with known outbreaks and about the distribution of Legionella in the environment.”
A Gram-negative bacillus, Legionella is an intracellular parasite of free-living protozoa primarily found in freshwater. “It can live and grow in biofilm, and there are more than 60 species of the bacterium,” she said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Cases are higher in the warmer months, and the rates are highest among the elderly, men, and those of black race. Currently, L. pneumophila accounts for about 90% of cases in the United States. “Once it’s transmitted, it has to hit a susceptible population to cause disease, generally older individuals and people with underlying conditions,” Dr. Cooley said.
A separate analysis evaluated Legionella cases reported among U.S. residents between 2005 and 2009 (MMWR. 2011;60[32]:1083-6). It found that only 4% were associated with outbreaks, and 96% were sporadic. “That doesn’t mean that [the cases] weren’t associated with the same kind of source, they just weren’t identified as an outbreak,” Dr. Cooley said. “It shows that there is a lot to learn about transmission of Legionella.”
Data from the National Notifiable Diseases Surveillance System indicate that rates of Legionella continue to rise nationwide, especially in the Midwest and Northeast. “Why? It’s possible that there are differences in testing preferences and reporting preferences in this region of the country,” Dr. Cooley noted. “Maybe people are more tuned in to the potential for outbreaks, but there are reasons why there could be differences in disease, like differences in infrastructure, climate, population density, and cooling tower density.” CDC data from 2015 indicate that most cases are not associated with a known exposure, and that the case fatality rate differs by exposure type: 12% for cases reporting health care exposure during the 10 days before symptom onset (25% for definite cases), 9% for cases reporting assisted or senior living exposure, 7% when no specific exposure is reported, and 4% for cases reporting travel exposure (MMWR. 2017;66[22]:584-9).
The U.S. case definition of Legionnaires’ disease consists of clinical or radiologic pneumonia plus confirmatory laboratory testing, either by urinary antigen test (UAT), lower respiratory culture, or appropriate serological testing. Polymerase chain reaction can be used as a presumptive test for a suspect case. “UAT is easy and it detects L. pneumophila serogroup 1 (Lp1), but it has some gaps,” Dr. Cooley said. “It isn’t completely sensitive for Lp1, and it doesn’t detect any other species or serogroups. That’s why we also recommend that a culture of respiratory secretions on selective media be performed at the same time. That being said, in the U.S., nearly all reported cases of Legionella are diagnosed by UAT only.”
A 2016 CDC MMWR and Vital Signs report found that almost all Legionella outbreaks could be prevented with effective water management, and the CDC has published a step-by-step guide to creating a water management program to reduce Legionella growth and spread in buildings. The 2017 MMWR Report found that definite health care–associated Legionnaires’ disease is deadly for one in four people who get it. The report also found that this issue is widespread; 76% of complete reporting jurisdictions reported at least one definite case of health care–associated Legionella disease in 2015. More recently, the Centers for Medicare & Medicaid Services issued a requirement to reduce risk in health care facility water systems to prevent cases and outbreaks. It applies to hospitals, skilled nursing facilities, and critical access hospitals.
Dr. Cooley reported having no financial disclosures.
SAN DIEGO –
“Improved testing and surveillance are needed to improve understanding of disease and outbreak burden,” Laura A. Cooley, MD, said at an annual scientific meeting on infectious diseases. “There is more to learn about environmental sources of Legionella for cases not associated with known outbreaks and about the distribution of Legionella in the environment.”
A Gram-negative bacillus, Legionella is an intracellular parasite of free-living protozoa primarily found in freshwater. “It can live and grow in biofilm, and there are more than 60 species of the bacterium,” she said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Cases are higher in the warmer months, and the rates are highest among the elderly, men, and those of black race. Currently, L. pneumophila accounts for about 90% of cases in the United States. “Once it’s transmitted, it has to hit a susceptible population to cause disease, generally older individuals and people with underlying conditions,” Dr. Cooley said.
A separate analysis evaluated Legionella cases reported among U.S. residents between 2005 and 2009 (MMWR. 2011;60[32]:1083-6). It found that only 4% were associated with outbreaks, and 96% were sporadic. “That doesn’t mean that [the cases] weren’t associated with the same kind of source, they just weren’t identified as an outbreak,” Dr. Cooley said. “It shows that there is a lot to learn about transmission of Legionella.”
Data from the National Notifiable Diseases Surveillance System indicate that rates of Legionella continue to rise nationwide, especially in the Midwest and Northeast. “Why? It’s possible that there are differences in testing preferences and reporting preferences in this region of the country,” Dr. Cooley noted. “Maybe people are more tuned in to the potential for outbreaks, but there are reasons why there could be differences in disease, like differences in infrastructure, climate, population density, and cooling tower density.” CDC data from 2015 indicate that most cases are not associated with a known exposure, and that the case fatality rate differs by exposure type: 12% for cases reporting health care exposure during the 10 days before symptom onset (25% for definite cases), 9% for cases reporting assisted or senior living exposure, 7% when no specific exposure is reported, and 4% for cases reporting travel exposure (MMWR. 2017;66[22]:584-9).
The U.S. case definition of Legionnaires’ disease consists of clinical or radiologic pneumonia plus confirmatory laboratory testing, either by urinary antigen test (UAT), lower respiratory culture, or appropriate serological testing. Polymerase chain reaction can be used as a presumptive test for a suspect case. “UAT is easy and it detects L. pneumophila serogroup 1 (Lp1), but it has some gaps,” Dr. Cooley said. “It isn’t completely sensitive for Lp1, and it doesn’t detect any other species or serogroups. That’s why we also recommend that a culture of respiratory secretions on selective media be performed at the same time. That being said, in the U.S., nearly all reported cases of Legionella are diagnosed by UAT only.”
A 2016 CDC MMWR and Vital Signs report found that almost all Legionella outbreaks could be prevented with effective water management, and the CDC has published a step-by-step guide to creating a water management program to reduce Legionella growth and spread in buildings. The 2017 MMWR Report found that definite health care–associated Legionnaires’ disease is deadly for one in four people who get it. The report also found that this issue is widespread; 76% of complete reporting jurisdictions reported at least one definite case of health care–associated Legionella disease in 2015. More recently, the Centers for Medicare & Medicaid Services issued a requirement to reduce risk in health care facility water systems to prevent cases and outbreaks. It applies to hospitals, skilled nursing facilities, and critical access hospitals.
Dr. Cooley reported having no financial disclosures.
REPORTING FROM ID WEEK 2017
Nearly 80% of health care personnel stepped up for flu shots
Nearly four out of five health care personnel in the United States received a flu vaccination during the 2016-2017 flu season, but a majority of those working in long-term care settings were not vaccinated, based on data from an Internet survey of more than 2,000 individuals that was conducted by the Centers for Disease Control and Prevention.
A total of 78.6% of the survey’s respondents said they’d been vaccinated during the 2016-2017 season. Vaccination coverage for health care personnel overall has remained in the 77%-79% range in recent years, but that represents an increase from 64% in 2010-2011.
“As in previous seasons, the highest coverage was among HCP whose workplace had vaccination requirements,” noted Carla L. Black, PhD, of the CDC, and colleagues (MMWR Morb Mortal Wkly Rep. 2017 Sep 29;66[38]:1009-15). The researchers reviewed data collected from an Internet panel survey of 2,438 health care personnel between March 28, 2017, and April 19, 2017.
Physicians boasted the highest vaccination coverage in 2016-2017 (96%), followed by pharmacists (94%), nurses (93%), nurse practitioners and physician assistants (92%), other clinical providers (80%), nonclinical health care providers (74%), and aides and assistants (69%).
Flu vaccination rates were highest among HCPs working in a hospital setting (92%); 94% of survey respondents in hospitals reported either having a vaccination requirement at work or being provided at least 1 day of on-site vaccination.
Vaccination rates were lowest among health care personnel in long-term care settings (68%), where only 26% reported a workplace vaccination requirement. However, vaccination rates in long-term care rose to 90% when employers required vaccination.
The report’s findings were limited by several factors, including the use of a volunteer sample, the reliance on self-reports, and the potential differences between Internet survey results and population-based estimates of flu vaccination.
However, “in the absence of vaccination requirements, the findings in this study support the recommendations found in the Guide to Community Preventive Services, which include active promotion of on-site vaccination at no cost or low cost to increase influenza vaccination coverage among HCPs,” the researchers said.
The researchers had no financial conflicts to disclose.
Nearly four out of five health care personnel in the United States received a flu vaccination during the 2016-2017 flu season, but a majority of those working in long-term care settings were not vaccinated, based on data from an Internet survey of more than 2,000 individuals that was conducted by the Centers for Disease Control and Prevention.
A total of 78.6% of the survey’s respondents said they’d been vaccinated during the 2016-2017 season. Vaccination coverage for health care personnel overall has remained in the 77%-79% range in recent years, but that represents an increase from 64% in 2010-2011.
“As in previous seasons, the highest coverage was among HCP whose workplace had vaccination requirements,” noted Carla L. Black, PhD, of the CDC, and colleagues (MMWR Morb Mortal Wkly Rep. 2017 Sep 29;66[38]:1009-15). The researchers reviewed data collected from an Internet panel survey of 2,438 health care personnel between March 28, 2017, and April 19, 2017.
Physicians boasted the highest vaccination coverage in 2016-2017 (96%), followed by pharmacists (94%), nurses (93%), nurse practitioners and physician assistants (92%), other clinical providers (80%), nonclinical health care providers (74%), and aides and assistants (69%).
Flu vaccination rates were highest among HCPs working in a hospital setting (92%); 94% of survey respondents in hospitals reported either having a vaccination requirement at work or being provided at least 1 day of on-site vaccination.
Vaccination rates were lowest among health care personnel in long-term care settings (68%), where only 26% reported a workplace vaccination requirement. However, vaccination rates in long-term care rose to 90% when employers required vaccination.
The report’s findings were limited by several factors, including the use of a volunteer sample, the reliance on self-reports, and the potential differences between Internet survey results and population-based estimates of flu vaccination.
However, “in the absence of vaccination requirements, the findings in this study support the recommendations found in the Guide to Community Preventive Services, which include active promotion of on-site vaccination at no cost or low cost to increase influenza vaccination coverage among HCPs,” the researchers said.
The researchers had no financial conflicts to disclose.
Nearly four out of five health care personnel in the United States received a flu vaccination during the 2016-2017 flu season, but a majority of those working in long-term care settings were not vaccinated, based on data from an Internet survey of more than 2,000 individuals that was conducted by the Centers for Disease Control and Prevention.
A total of 78.6% of the survey’s respondents said they’d been vaccinated during the 2016-2017 season. Vaccination coverage for health care personnel overall has remained in the 77%-79% range in recent years, but that represents an increase from 64% in 2010-2011.
“As in previous seasons, the highest coverage was among HCP whose workplace had vaccination requirements,” noted Carla L. Black, PhD, of the CDC, and colleagues (MMWR Morb Mortal Wkly Rep. 2017 Sep 29;66[38]:1009-15). The researchers reviewed data collected from an Internet panel survey of 2,438 health care personnel between March 28, 2017, and April 19, 2017.
Physicians boasted the highest vaccination coverage in 2016-2017 (96%), followed by pharmacists (94%), nurses (93%), nurse practitioners and physician assistants (92%), other clinical providers (80%), nonclinical health care providers (74%), and aides and assistants (69%).
Flu vaccination rates were highest among HCPs working in a hospital setting (92%); 94% of survey respondents in hospitals reported either having a vaccination requirement at work or being provided at least 1 day of on-site vaccination.
Vaccination rates were lowest among health care personnel in long-term care settings (68%), where only 26% reported a workplace vaccination requirement. However, vaccination rates in long-term care rose to 90% when employers required vaccination.
The report’s findings were limited by several factors, including the use of a volunteer sample, the reliance on self-reports, and the potential differences between Internet survey results and population-based estimates of flu vaccination.
However, “in the absence of vaccination requirements, the findings in this study support the recommendations found in the Guide to Community Preventive Services, which include active promotion of on-site vaccination at no cost or low cost to increase influenza vaccination coverage among HCPs,” the researchers said.
The researchers had no financial conflicts to disclose.
FROM MMWR
Key clinical point:
Major finding: Overall flu vaccination coverage among U.S. health care personnel was 78.6% in the 2016-2017 season
Data source: The data come from an Internet survey of 2,438 health care personnel.
Disclosures: The researchers had no financial conflicts to disclose.
Avoid sildenafil for pulmonary hypertension after corrected valvular disease
BARCELONA – Off-label use of the phosphodiesterase-5 inhibitor sildenafil to treat residual pulmonary hypertension after successful correction of valvular heart disease is not merely ineffective, it’s counterproductive, according to the results of the randomized, placebo-controlled SIOVAC study.
“We believe based upon our results that off-label use of sildenafil in patients with left heart disease-pulmonary hypertension due to valvular disease should be discouraged,” Javier Bermejo, MD, declared at the annual congress of the European Society of Cardiology.

Sildenafil is approved with a solid, evidence-based indication for treating some other types of pulmonary hypertension. Many cardiologists also prescribe the drug off label for residual pulmonary hypertension in patients with corrected valve disease, hoping that it will be of benefit, since there is currently no approved treatment for this common and serious condition associated with increased mortality. But because the anecdotal literature on sildenafil for this specific type of pulmonary hypertension is mixed, Dr. Bermejo and his coinvestigators in the Spanish Network Center for Cardiovascular Research decided to conduct a multicenter randomized trial.
SIOVAC (Sildenafil for Improving Clinical Outcomes After Valvular Correction) comprised 200 patients with residual pulmonary hypertension after corrected valvular heart disease at 17 Spanish general hospitals. The patients were randomized to receive sildenafil at 40 mg t.i.d. or placebo for 6 months in this double-blind trial.
The primary endpoint was a standardized composite clinical score widely used in heart failure trials. It consists of all-cause mortality, hospital admission for heart failure, worsening exercise tolerance, and deterioration in a global self-assessment rating.
The shocker for the investigators – who had expected a positive study – was that 33% of patients in the sildenafil group worsened significantly on the composite clinical score at 6 months, compared with 14% of placebo-treated controls, said Dr. Bermejo, a cardiologist at Gregorio Marañón University Hospital in Madrid.
Moreover, only 27% of the sildenafil group improved, compared with 44% of controls. About one-third of patients in both groups remained unchanged over the course of the 6-month trial.
Dr. Bermejo noted that valvular disease is considered the next cardiac epidemic because of its strong association with advancing age and the rapid aging of the population worldwide. Pulmonary hypertension occurs is virtually all patients with severe mitral disease and in up to two-thirds of those with asymptomatic aortic stenosis. Regression of the pulmonary hypertension is often incomplete after successful surgical or transcatheter correction of the valvular lesion.
Discussant Irene M. Lang, MD, called SIOVAC “a very clear study.” It convincingly establishes that sildenafil – a vasodilator – is ineffective for the treatment of what the current ESC/European Respiratory Society guidelines on pulmonary hypertension call isolated post-capillary pulmonary hypertension, a condition defined hemodynamically by a diastolic pulmonary vascular pressure gradient of less than 7 mm Hg and/or a pulmonary vascular resistance below 3 Wood units (Eur Heart J. 2016 Jan 1;37[1]:67-119.)
The SIOVAC findings underscore the strong IIIC recommendation in the European guidelines that the use of approved therapies for pulmonary arterial hypertension is not recommended in patients with left heart disease-pulmonary hypertension, added Dr. Lang, a coauthor of the guidelines and professor of vascular biology at the Medical University of Vienna.
The Spanish government funded SIOVAC. Dr. Bermejo reported having no financial conflicts of interest.
BARCELONA – Off-label use of the phosphodiesterase-5 inhibitor sildenafil to treat residual pulmonary hypertension after successful correction of valvular heart disease is not merely ineffective, it’s counterproductive, according to the results of the randomized, placebo-controlled SIOVAC study.
“We believe based upon our results that off-label use of sildenafil in patients with left heart disease-pulmonary hypertension due to valvular disease should be discouraged,” Javier Bermejo, MD, declared at the annual congress of the European Society of Cardiology.

Sildenafil is approved with a solid, evidence-based indication for treating some other types of pulmonary hypertension. Many cardiologists also prescribe the drug off label for residual pulmonary hypertension in patients with corrected valve disease, hoping that it will be of benefit, since there is currently no approved treatment for this common and serious condition associated with increased mortality. But because the anecdotal literature on sildenafil for this specific type of pulmonary hypertension is mixed, Dr. Bermejo and his coinvestigators in the Spanish Network Center for Cardiovascular Research decided to conduct a multicenter randomized trial.
SIOVAC (Sildenafil for Improving Clinical Outcomes After Valvular Correction) comprised 200 patients with residual pulmonary hypertension after corrected valvular heart disease at 17 Spanish general hospitals. The patients were randomized to receive sildenafil at 40 mg t.i.d. or placebo for 6 months in this double-blind trial.
The primary endpoint was a standardized composite clinical score widely used in heart failure trials. It consists of all-cause mortality, hospital admission for heart failure, worsening exercise tolerance, and deterioration in a global self-assessment rating.
The shocker for the investigators – who had expected a positive study – was that 33% of patients in the sildenafil group worsened significantly on the composite clinical score at 6 months, compared with 14% of placebo-treated controls, said Dr. Bermejo, a cardiologist at Gregorio Marañón University Hospital in Madrid.
Moreover, only 27% of the sildenafil group improved, compared with 44% of controls. About one-third of patients in both groups remained unchanged over the course of the 6-month trial.
Dr. Bermejo noted that valvular disease is considered the next cardiac epidemic because of its strong association with advancing age and the rapid aging of the population worldwide. Pulmonary hypertension occurs is virtually all patients with severe mitral disease and in up to two-thirds of those with asymptomatic aortic stenosis. Regression of the pulmonary hypertension is often incomplete after successful surgical or transcatheter correction of the valvular lesion.
Discussant Irene M. Lang, MD, called SIOVAC “a very clear study.” It convincingly establishes that sildenafil – a vasodilator – is ineffective for the treatment of what the current ESC/European Respiratory Society guidelines on pulmonary hypertension call isolated post-capillary pulmonary hypertension, a condition defined hemodynamically by a diastolic pulmonary vascular pressure gradient of less than 7 mm Hg and/or a pulmonary vascular resistance below 3 Wood units (Eur Heart J. 2016 Jan 1;37[1]:67-119.)
The SIOVAC findings underscore the strong IIIC recommendation in the European guidelines that the use of approved therapies for pulmonary arterial hypertension is not recommended in patients with left heart disease-pulmonary hypertension, added Dr. Lang, a coauthor of the guidelines and professor of vascular biology at the Medical University of Vienna.
The Spanish government funded SIOVAC. Dr. Bermejo reported having no financial conflicts of interest.
BARCELONA – Off-label use of the phosphodiesterase-5 inhibitor sildenafil to treat residual pulmonary hypertension after successful correction of valvular heart disease is not merely ineffective, it’s counterproductive, according to the results of the randomized, placebo-controlled SIOVAC study.
“We believe based upon our results that off-label use of sildenafil in patients with left heart disease-pulmonary hypertension due to valvular disease should be discouraged,” Javier Bermejo, MD, declared at the annual congress of the European Society of Cardiology.

Sildenafil is approved with a solid, evidence-based indication for treating some other types of pulmonary hypertension. Many cardiologists also prescribe the drug off label for residual pulmonary hypertension in patients with corrected valve disease, hoping that it will be of benefit, since there is currently no approved treatment for this common and serious condition associated with increased mortality. But because the anecdotal literature on sildenafil for this specific type of pulmonary hypertension is mixed, Dr. Bermejo and his coinvestigators in the Spanish Network Center for Cardiovascular Research decided to conduct a multicenter randomized trial.
SIOVAC (Sildenafil for Improving Clinical Outcomes After Valvular Correction) comprised 200 patients with residual pulmonary hypertension after corrected valvular heart disease at 17 Spanish general hospitals. The patients were randomized to receive sildenafil at 40 mg t.i.d. or placebo for 6 months in this double-blind trial.
The primary endpoint was a standardized composite clinical score widely used in heart failure trials. It consists of all-cause mortality, hospital admission for heart failure, worsening exercise tolerance, and deterioration in a global self-assessment rating.
The shocker for the investigators – who had expected a positive study – was that 33% of patients in the sildenafil group worsened significantly on the composite clinical score at 6 months, compared with 14% of placebo-treated controls, said Dr. Bermejo, a cardiologist at Gregorio Marañón University Hospital in Madrid.
Moreover, only 27% of the sildenafil group improved, compared with 44% of controls. About one-third of patients in both groups remained unchanged over the course of the 6-month trial.
Dr. Bermejo noted that valvular disease is considered the next cardiac epidemic because of its strong association with advancing age and the rapid aging of the population worldwide. Pulmonary hypertension occurs is virtually all patients with severe mitral disease and in up to two-thirds of those with asymptomatic aortic stenosis. Regression of the pulmonary hypertension is often incomplete after successful surgical or transcatheter correction of the valvular lesion.
Discussant Irene M. Lang, MD, called SIOVAC “a very clear study.” It convincingly establishes that sildenafil – a vasodilator – is ineffective for the treatment of what the current ESC/European Respiratory Society guidelines on pulmonary hypertension call isolated post-capillary pulmonary hypertension, a condition defined hemodynamically by a diastolic pulmonary vascular pressure gradient of less than 7 mm Hg and/or a pulmonary vascular resistance below 3 Wood units (Eur Heart J. 2016 Jan 1;37[1]:67-119.)
The SIOVAC findings underscore the strong IIIC recommendation in the European guidelines that the use of approved therapies for pulmonary arterial hypertension is not recommended in patients with left heart disease-pulmonary hypertension, added Dr. Lang, a coauthor of the guidelines and professor of vascular biology at the Medical University of Vienna.
The Spanish government funded SIOVAC. Dr. Bermejo reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2017
Key clinical point:
Major finding: One-third of patients with residual pulmonary hypertension after successful correction of valvular heart disease experienced significant clinical worsening during 6 months of sildenafil therapy, compared with 14% on placebo.
Data source: SIOVAC, a 6-month, double-blind, placebo-controlled study of 200 patients with residual pulmonary hypertension after correction for valvular heart disease.
Disclosures: The Spanish government funded SIOVAC. The presenter reported having no financial conflicts of interest.
FDA approves implantable device for central sleep apnea
The U.S. Food and Drug Administration on Oct. 6 approved an implantable device for the treatment of moderate to severe central sleep apnea.
The remedē System consists of a battery pack and small, thin wires placed under the skin in the upper chest area. The wires are inserted into the blood vessels in the chest to stimulate the phrenic nerve. The system monitors respiratory signals and, when it stimulates the nerve, the diaphragm moves to restore normal breathing.
The agency’s approval comes on the basis of study results showing that the system reduced the apnea–hypopnea index scores by 50% or more in 51% of patients studied. Control patients in the study saw an 11% reduction in their score.
Adverse events reported in the study included concomitant device interaction, implant site infection, and swelling and local tissue damage or pocket erosion. The remedē System is contraindicated for patients with active infection or who are known to require an MRI.
The U.S. Food and Drug Administration on Oct. 6 approved an implantable device for the treatment of moderate to severe central sleep apnea.
The remedē System consists of a battery pack and small, thin wires placed under the skin in the upper chest area. The wires are inserted into the blood vessels in the chest to stimulate the phrenic nerve. The system monitors respiratory signals and, when it stimulates the nerve, the diaphragm moves to restore normal breathing.
The agency’s approval comes on the basis of study results showing that the system reduced the apnea–hypopnea index scores by 50% or more in 51% of patients studied. Control patients in the study saw an 11% reduction in their score.
Adverse events reported in the study included concomitant device interaction, implant site infection, and swelling and local tissue damage or pocket erosion. The remedē System is contraindicated for patients with active infection or who are known to require an MRI.
The U.S. Food and Drug Administration on Oct. 6 approved an implantable device for the treatment of moderate to severe central sleep apnea.
The remedē System consists of a battery pack and small, thin wires placed under the skin in the upper chest area. The wires are inserted into the blood vessels in the chest to stimulate the phrenic nerve. The system monitors respiratory signals and, when it stimulates the nerve, the diaphragm moves to restore normal breathing.
The agency’s approval comes on the basis of study results showing that the system reduced the apnea–hypopnea index scores by 50% or more in 51% of patients studied. Control patients in the study saw an 11% reduction in their score.
Adverse events reported in the study included concomitant device interaction, implant site infection, and swelling and local tissue damage or pocket erosion. The remedē System is contraindicated for patients with active infection or who are known to require an MRI.
Lifesaving future seen for electronic cigarettes
A switch from cigarettes to e-cigarettes has the potential to prevent almost 90,000 premature deaths in the United States in the year 2026, according to a study examining e-cigarette substitution scenarios.
The investigators’ “optimistic scenario” – in which new smokers use e-cigarettes instead of cigarettes, smoking prevalence falls to 5% over a 10-year period, and e-cigarettes have a 5% excess risk over regular cigarettes – projects 380,832 premature deaths from smoking in the year 2026. Under a “status quo scenario,” which projected current cigarette initiation and cessation rates and did not include e-cigarettes or other tobacco products, there would be 470,743 deaths, reported David T. Levy, PhD, and his associates (Tob Control. 2017 Oct 2. doi: 10.1136/tobaccocontrol-2017-053759).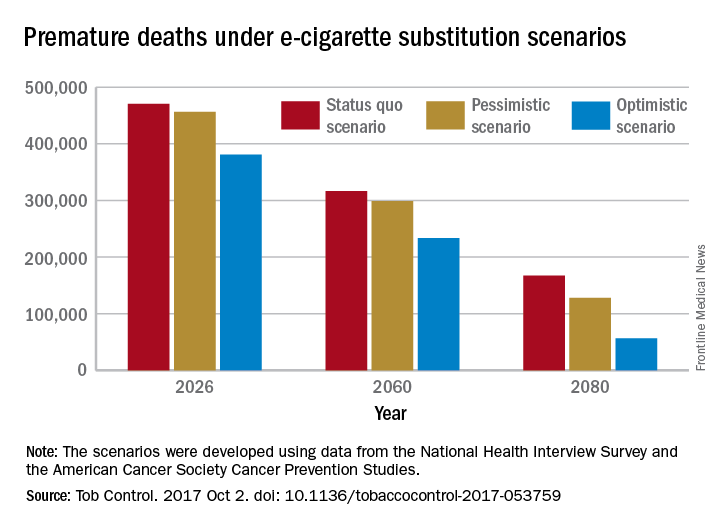
Further projections suggest that the optimistic scenario could result in almost 6.6 million fewer premature deaths and 86.7 million years of life gained by the year 2100, compared with the status quo scenario, while the pessimistic scenario would prevent 1.6 million deaths and add an extra 20.8 million years of life, they noted.
Since “a strategy of replacing cigarette by e-cigarette use can yield substantial gains, even with conservative assumptions about related risks … an endgame scenario for cigarettes might well be within reach, if new technologies for delivering nicotine with substantially less harm, but sufficient satisfaction, are harnessed with sufficient passion and political will to aggressively phase out tobacco cigarettes,” Dr. Levy and his associates wrote.
The study was funded by grants from the National Institute on Drug Abuse and the National Cancer Institute. One investigator received a research grant from Pfizer and served as an advisory board member to Johnson & Johnson, which manufactures smoking cessation medications. No other conflicts of interest were declared.
A switch from cigarettes to e-cigarettes has the potential to prevent almost 90,000 premature deaths in the United States in the year 2026, according to a study examining e-cigarette substitution scenarios.
The investigators’ “optimistic scenario” – in which new smokers use e-cigarettes instead of cigarettes, smoking prevalence falls to 5% over a 10-year period, and e-cigarettes have a 5% excess risk over regular cigarettes – projects 380,832 premature deaths from smoking in the year 2026. Under a “status quo scenario,” which projected current cigarette initiation and cessation rates and did not include e-cigarettes or other tobacco products, there would be 470,743 deaths, reported David T. Levy, PhD, and his associates (Tob Control. 2017 Oct 2. doi: 10.1136/tobaccocontrol-2017-053759).
Further projections suggest that the optimistic scenario could result in almost 6.6 million fewer premature deaths and 86.7 million years of life gained by the year 2100, compared with the status quo scenario, while the pessimistic scenario would prevent 1.6 million deaths and add an extra 20.8 million years of life, they noted.
Since “a strategy of replacing cigarette by e-cigarette use can yield substantial gains, even with conservative assumptions about related risks … an endgame scenario for cigarettes might well be within reach, if new technologies for delivering nicotine with substantially less harm, but sufficient satisfaction, are harnessed with sufficient passion and political will to aggressively phase out tobacco cigarettes,” Dr. Levy and his associates wrote.
The study was funded by grants from the National Institute on Drug Abuse and the National Cancer Institute. One investigator received a research grant from Pfizer and served as an advisory board member to Johnson & Johnson, which manufactures smoking cessation medications. No other conflicts of interest were declared.
A switch from cigarettes to e-cigarettes has the potential to prevent almost 90,000 premature deaths in the United States in the year 2026, according to a study examining e-cigarette substitution scenarios.
The investigators’ “optimistic scenario” – in which new smokers use e-cigarettes instead of cigarettes, smoking prevalence falls to 5% over a 10-year period, and e-cigarettes have a 5% excess risk over regular cigarettes – projects 380,832 premature deaths from smoking in the year 2026. Under a “status quo scenario,” which projected current cigarette initiation and cessation rates and did not include e-cigarettes or other tobacco products, there would be 470,743 deaths, reported David T. Levy, PhD, and his associates (Tob Control. 2017 Oct 2. doi: 10.1136/tobaccocontrol-2017-053759).
Further projections suggest that the optimistic scenario could result in almost 6.6 million fewer premature deaths and 86.7 million years of life gained by the year 2100, compared with the status quo scenario, while the pessimistic scenario would prevent 1.6 million deaths and add an extra 20.8 million years of life, they noted.
Since “a strategy of replacing cigarette by e-cigarette use can yield substantial gains, even with conservative assumptions about related risks … an endgame scenario for cigarettes might well be within reach, if new technologies for delivering nicotine with substantially less harm, but sufficient satisfaction, are harnessed with sufficient passion and political will to aggressively phase out tobacco cigarettes,” Dr. Levy and his associates wrote.
The study was funded by grants from the National Institute on Drug Abuse and the National Cancer Institute. One investigator received a research grant from Pfizer and served as an advisory board member to Johnson & Johnson, which manufactures smoking cessation medications. No other conflicts of interest were declared.
FROM TOBACCO CONTROL
Just over half of pregnant women got flu vaccine in 2016-2017
Influenza vaccination among pregnant women during the 2016-2017 flu season was slightly higher than during the 2015-2016 season, according to the Centers for Disease Control and Prevention.
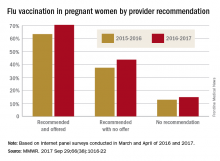
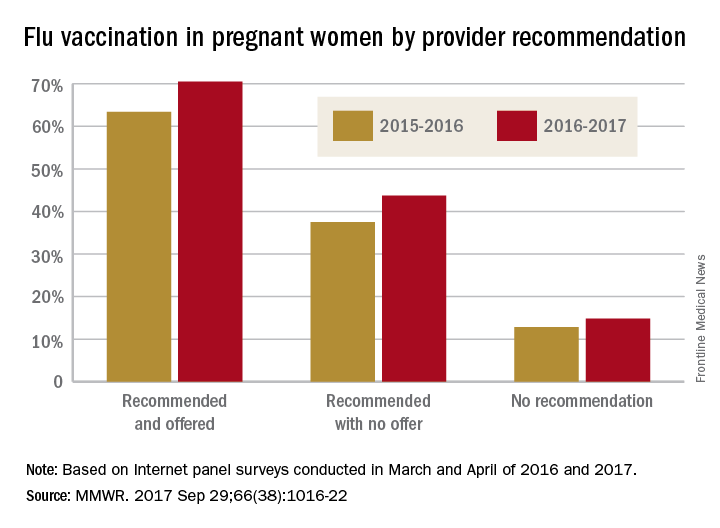
Overall coverage for 2016-2017 was 53.6% among pregnant women, compared with 49.9% in 2015-2016, continuing the overall rise seen over the last several flu seasons. Among pregnant women who received a recommendation from a health care provider and were offered vaccination, coverage was 70.5% in 2016-2017, while coverage was 43.7% among women who received a recommendation but no offer and 14.8% among those who did not receive a recommendation, the CDC reported (MMWR. 2017 Sep 29;66[38]:1016-22).
Among other subgroups, coverage by age for the 2016-2017 flu season was 41.7% for those aged 18-24 years, 58.4% for those aged 25-34 years, and 58.5% for those 35-49 years old. There also was considerable variation by race/ethnicity, with coverage at 61.2% for Hispanics, 55.4% for whites, 42.3% for blacks, and 51.7% for others. Coverage for the subgroups corresponded with the rates at which vaccination was recommended: Younger women were less likely than older women to receive a recommendation, and Hispanic and white women more likely to receive recommendations than did blacks and other races/ethnicities, the CDC said.
The 2017 data include 1,893 responses to an Internet panel survey conducted from March 28 to April 7, 2017. The analysis of the 2016 panel survey, which was conducted from March 29 to April 7, 2016, included responses from 1,692 women.
Influenza vaccination among pregnant women during the 2016-2017 flu season was slightly higher than during the 2015-2016 season, according to the Centers for Disease Control and Prevention.
Overall coverage for 2016-2017 was 53.6% among pregnant women, compared with 49.9% in 2015-2016, continuing the overall rise seen over the last several flu seasons. Among pregnant women who received a recommendation from a health care provider and were offered vaccination, coverage was 70.5% in 2016-2017, while coverage was 43.7% among women who received a recommendation but no offer and 14.8% among those who did not receive a recommendation, the CDC reported (MMWR. 2017 Sep 29;66[38]:1016-22).
Among other subgroups, coverage by age for the 2016-2017 flu season was 41.7% for those aged 18-24 years, 58.4% for those aged 25-34 years, and 58.5% for those 35-49 years old. There also was considerable variation by race/ethnicity, with coverage at 61.2% for Hispanics, 55.4% for whites, 42.3% for blacks, and 51.7% for others. Coverage for the subgroups corresponded with the rates at which vaccination was recommended: Younger women were less likely than older women to receive a recommendation, and Hispanic and white women more likely to receive recommendations than did blacks and other races/ethnicities, the CDC said.
The 2017 data include 1,893 responses to an Internet panel survey conducted from March 28 to April 7, 2017. The analysis of the 2016 panel survey, which was conducted from March 29 to April 7, 2016, included responses from 1,692 women.
Influenza vaccination among pregnant women during the 2016-2017 flu season was slightly higher than during the 2015-2016 season, according to the Centers for Disease Control and Prevention.
Overall coverage for 2016-2017 was 53.6% among pregnant women, compared with 49.9% in 2015-2016, continuing the overall rise seen over the last several flu seasons. Among pregnant women who received a recommendation from a health care provider and were offered vaccination, coverage was 70.5% in 2016-2017, while coverage was 43.7% among women who received a recommendation but no offer and 14.8% among those who did not receive a recommendation, the CDC reported (MMWR. 2017 Sep 29;66[38]:1016-22).
Among other subgroups, coverage by age for the 2016-2017 flu season was 41.7% for those aged 18-24 years, 58.4% for those aged 25-34 years, and 58.5% for those 35-49 years old. There also was considerable variation by race/ethnicity, with coverage at 61.2% for Hispanics, 55.4% for whites, 42.3% for blacks, and 51.7% for others. Coverage for the subgroups corresponded with the rates at which vaccination was recommended: Younger women were less likely than older women to receive a recommendation, and Hispanic and white women more likely to receive recommendations than did blacks and other races/ethnicities, the CDC said.
The 2017 data include 1,893 responses to an Internet panel survey conducted from March 28 to April 7, 2017. The analysis of the 2016 panel survey, which was conducted from March 29 to April 7, 2016, included responses from 1,692 women.
FROM MMWR
Clinicians: Be clear about flu vaccine’s value
WASHINGTON – Flu vaccination rates remain below the 70% Healthy People 2020 goal for most of the U.S. population, but data show that a recommendation from a clinician can encourage individuals to get vaccinated and to vaccinate their children, according to a panel of experts who spoke at a press briefing sponsored by the National Foundation for Infectious Diseases.
“Annual vaccination is our first line of defense against the flu,” William Schaffner, MD, of Vanderbilt University, Nashville, Tenn., said at the briefing. The unpredictable nature of the flu makes annual vaccination even more important – and the earlier, the better, said Dr. Schaffner. “If you have seen one flu season, you have seen ... one flu season.”
In a video interview at the briefing, experts emphasized the safety and effectiveness of the flu vaccine for a range of populations, including children, pregnant women, and older adults. And they offered tips to convince patients of the importance of vaccination, as well as the need to make sure health care staff are protected.
Briefing participants included former Department of Health and Human Services Secretary Thomas A. Price, MD; Patricia A. Stinchfield, RN, MS, CPNP, CIC of Children’s Hospitals and Clinics of Minnesota, St. Paul; Kathleen M. Neuzil, MD, of the University of Maryland; and Daniel B. Jernigan, MD, of the Centers for Disease Control and Prevention.
The clinicians interviewed had no financial conflicts to disclose.
WASHINGTON – Flu vaccination rates remain below the 70% Healthy People 2020 goal for most of the U.S. population, but data show that a recommendation from a clinician can encourage individuals to get vaccinated and to vaccinate their children, according to a panel of experts who spoke at a press briefing sponsored by the National Foundation for Infectious Diseases.
“Annual vaccination is our first line of defense against the flu,” William Schaffner, MD, of Vanderbilt University, Nashville, Tenn., said at the briefing. The unpredictable nature of the flu makes annual vaccination even more important – and the earlier, the better, said Dr. Schaffner. “If you have seen one flu season, you have seen ... one flu season.”
In a video interview at the briefing, experts emphasized the safety and effectiveness of the flu vaccine for a range of populations, including children, pregnant women, and older adults. And they offered tips to convince patients of the importance of vaccination, as well as the need to make sure health care staff are protected.
Briefing participants included former Department of Health and Human Services Secretary Thomas A. Price, MD; Patricia A. Stinchfield, RN, MS, CPNP, CIC of Children’s Hospitals and Clinics of Minnesota, St. Paul; Kathleen M. Neuzil, MD, of the University of Maryland; and Daniel B. Jernigan, MD, of the Centers for Disease Control and Prevention.
The clinicians interviewed had no financial conflicts to disclose.
WASHINGTON – Flu vaccination rates remain below the 70% Healthy People 2020 goal for most of the U.S. population, but data show that a recommendation from a clinician can encourage individuals to get vaccinated and to vaccinate their children, according to a panel of experts who spoke at a press briefing sponsored by the National Foundation for Infectious Diseases.
“Annual vaccination is our first line of defense against the flu,” William Schaffner, MD, of Vanderbilt University, Nashville, Tenn., said at the briefing. The unpredictable nature of the flu makes annual vaccination even more important – and the earlier, the better, said Dr. Schaffner. “If you have seen one flu season, you have seen ... one flu season.”
In a video interview at the briefing, experts emphasized the safety and effectiveness of the flu vaccine for a range of populations, including children, pregnant women, and older adults. And they offered tips to convince patients of the importance of vaccination, as well as the need to make sure health care staff are protected.
Briefing participants included former Department of Health and Human Services Secretary Thomas A. Price, MD; Patricia A. Stinchfield, RN, MS, CPNP, CIC of Children’s Hospitals and Clinics of Minnesota, St. Paul; Kathleen M. Neuzil, MD, of the University of Maryland; and Daniel B. Jernigan, MD, of the Centers for Disease Control and Prevention.
The clinicians interviewed had no financial conflicts to disclose.
AT A PRESS BRIEFING BY THE NATIONAL FOUNDATION FOR INFECTIOUS DISEASES
New AAP policy recommends flu vaccination regardless of egg allergy
CHICAGO – The American Academy of Pediatrics has released a new policy statement that in part suggests that physicians can administer influenza vaccine to children and teenagers with egg allergies without any special precautions beyond those that apply to other vaccines.
This is some “egg-citing news,” said Mary Ann Jackson, MD. “In 28 studies with 4,315 egg allergic subjects, 656 of whom had severe allergies, there were no serious allergic reactions.” In other words, there was no respiratory distress or hypotension observed after participants received the influenza vaccine in these studies, she added.
Officially, the policy statement from the academy’s Committee on Infectious Diseases reads: “All children with egg allergy of any severity can receive influenza vaccine without any additional precautions beyond those recommended for any vaccine. Special precautions for egg-allergic recipients of IIV [inactivated influenza vaccine] are not warranted, as the rate of anaphylaxis after IIV administration is no greater in egg-allergic than non–egg-allergic recipients or from other universally recommended vaccines. Standard vaccination practice for all vaccines in children should include the ability to respond to rare acute hypersensitivity reactions.” The full policy statement was published online in the journal Pediatrics (2017 Sep. 6; doi: 10.1542/peds.2017-2550.)
This new policy “makes your life so much easier … and allows you to continue to recommend the vaccine strongly,” said Dr. Jackson, division director of infectious diseases at Children’s Mercy Hospital and professor of pediatrics at the University of Missouri at Kansas City.
Other reasons to recommend flu vaccination
Dr. Jackson also gave an overview of influenza epidemiology and why ongoing education of patients and families remains essential. “It’s almost flu season now. It’s inevitable – like RSV [respiratory syncytial virus] is inevitable – but when the seasons starts is unknown,” she said. “How severe and long the season will be is also unpredictable.” Which viruses are spread and whether there is a good match between circulating virus and the vaccine are additional unknowns each year.
Patient and family education are important for these reasons, and because “it’s the most likely vaccine to be opted out of by the otherwise fully immunized,” she noted.
The lowest covered population are individuals between 18 years and 49 years, Dr. Jackson said. “My young adult son did not think he needed to worry about flu for himself; he said he was healthy and well. But it’s a bummer when your mom is an ID doctor who likes vaccines, because I made it happen for him.”
Dr. Jackson had no relevant financial disclosures.
CHICAGO – The American Academy of Pediatrics has released a new policy statement that in part suggests that physicians can administer influenza vaccine to children and teenagers with egg allergies without any special precautions beyond those that apply to other vaccines.
This is some “egg-citing news,” said Mary Ann Jackson, MD. “In 28 studies with 4,315 egg allergic subjects, 656 of whom had severe allergies, there were no serious allergic reactions.” In other words, there was no respiratory distress or hypotension observed after participants received the influenza vaccine in these studies, she added.
Officially, the policy statement from the academy’s Committee on Infectious Diseases reads: “All children with egg allergy of any severity can receive influenza vaccine without any additional precautions beyond those recommended for any vaccine. Special precautions for egg-allergic recipients of IIV [inactivated influenza vaccine] are not warranted, as the rate of anaphylaxis after IIV administration is no greater in egg-allergic than non–egg-allergic recipients or from other universally recommended vaccines. Standard vaccination practice for all vaccines in children should include the ability to respond to rare acute hypersensitivity reactions.” The full policy statement was published online in the journal Pediatrics (2017 Sep. 6; doi: 10.1542/peds.2017-2550.)
This new policy “makes your life so much easier … and allows you to continue to recommend the vaccine strongly,” said Dr. Jackson, division director of infectious diseases at Children’s Mercy Hospital and professor of pediatrics at the University of Missouri at Kansas City.
Other reasons to recommend flu vaccination
Dr. Jackson also gave an overview of influenza epidemiology and why ongoing education of patients and families remains essential. “It’s almost flu season now. It’s inevitable – like RSV [respiratory syncytial virus] is inevitable – but when the seasons starts is unknown,” she said. “How severe and long the season will be is also unpredictable.” Which viruses are spread and whether there is a good match between circulating virus and the vaccine are additional unknowns each year.
Patient and family education are important for these reasons, and because “it’s the most likely vaccine to be opted out of by the otherwise fully immunized,” she noted.
The lowest covered population are individuals between 18 years and 49 years, Dr. Jackson said. “My young adult son did not think he needed to worry about flu for himself; he said he was healthy and well. But it’s a bummer when your mom is an ID doctor who likes vaccines, because I made it happen for him.”
Dr. Jackson had no relevant financial disclosures.
CHICAGO – The American Academy of Pediatrics has released a new policy statement that in part suggests that physicians can administer influenza vaccine to children and teenagers with egg allergies without any special precautions beyond those that apply to other vaccines.
This is some “egg-citing news,” said Mary Ann Jackson, MD. “In 28 studies with 4,315 egg allergic subjects, 656 of whom had severe allergies, there were no serious allergic reactions.” In other words, there was no respiratory distress or hypotension observed after participants received the influenza vaccine in these studies, she added.
Officially, the policy statement from the academy’s Committee on Infectious Diseases reads: “All children with egg allergy of any severity can receive influenza vaccine without any additional precautions beyond those recommended for any vaccine. Special precautions for egg-allergic recipients of IIV [inactivated influenza vaccine] are not warranted, as the rate of anaphylaxis after IIV administration is no greater in egg-allergic than non–egg-allergic recipients or from other universally recommended vaccines. Standard vaccination practice for all vaccines in children should include the ability to respond to rare acute hypersensitivity reactions.” The full policy statement was published online in the journal Pediatrics (2017 Sep. 6; doi: 10.1542/peds.2017-2550.)
This new policy “makes your life so much easier … and allows you to continue to recommend the vaccine strongly,” said Dr. Jackson, division director of infectious diseases at Children’s Mercy Hospital and professor of pediatrics at the University of Missouri at Kansas City.
Other reasons to recommend flu vaccination
Dr. Jackson also gave an overview of influenza epidemiology and why ongoing education of patients and families remains essential. “It’s almost flu season now. It’s inevitable – like RSV [respiratory syncytial virus] is inevitable – but when the seasons starts is unknown,” she said. “How severe and long the season will be is also unpredictable.” Which viruses are spread and whether there is a good match between circulating virus and the vaccine are additional unknowns each year.
Patient and family education are important for these reasons, and because “it’s the most likely vaccine to be opted out of by the otherwise fully immunized,” she noted.
The lowest covered population are individuals between 18 years and 49 years, Dr. Jackson said. “My young adult son did not think he needed to worry about flu for himself; he said he was healthy and well. But it’s a bummer when your mom is an ID doctor who likes vaccines, because I made it happen for him.”
Dr. Jackson had no relevant financial disclosures.
AT AAP 2017
With inpatient flu shots, providers’ attitude problem may outweigh parents’
reported Suchitra Rao, MD, of the University of Colorado, Aurora, and her colleagues.
Surveys assessing attitudes toward inpatient influenza vaccination were given to parents/caregivers of general pediatric inpatients and to inpatient physicians, residents, nurses, physician assistants, and nurse practitioners at Children’s Hospital Colorado in Aurora between October 2014 and March 2015. Response rates were 95% of the 1,053 parents/caregivers and 58% of the 339 providers.
The parents agreed that the flu is a serious disease (92%), that flu vaccines work (58%), that flu vaccines are safe (76%), and that the vaccines are needed annually (76%), the Dr. Rao and her colleagues found.
The providers thought the most common barriers to vaccination were parental refusal because of child illness (80%) and family misconceptions about the vaccine (74%). Also, 54% of providers forgot to ask about flu vaccination status and 46% forgot to order flu vaccines.
When asked what interventions might increase flu vaccination rates in the inpatient setting, 73% of providers agreed that personal reminders might help increase vaccination rates, but only 48% thought that provider education might help do so.
Read more in the journal Influenza and Other Respiratory Viruses (2017 Sep 5. doi: 10.1111/irv.12482.)
reported Suchitra Rao, MD, of the University of Colorado, Aurora, and her colleagues.
Surveys assessing attitudes toward inpatient influenza vaccination were given to parents/caregivers of general pediatric inpatients and to inpatient physicians, residents, nurses, physician assistants, and nurse practitioners at Children’s Hospital Colorado in Aurora between October 2014 and March 2015. Response rates were 95% of the 1,053 parents/caregivers and 58% of the 339 providers.
The parents agreed that the flu is a serious disease (92%), that flu vaccines work (58%), that flu vaccines are safe (76%), and that the vaccines are needed annually (76%), the Dr. Rao and her colleagues found.
The providers thought the most common barriers to vaccination were parental refusal because of child illness (80%) and family misconceptions about the vaccine (74%). Also, 54% of providers forgot to ask about flu vaccination status and 46% forgot to order flu vaccines.
When asked what interventions might increase flu vaccination rates in the inpatient setting, 73% of providers agreed that personal reminders might help increase vaccination rates, but only 48% thought that provider education might help do so.
Read more in the journal Influenza and Other Respiratory Viruses (2017 Sep 5. doi: 10.1111/irv.12482.)
reported Suchitra Rao, MD, of the University of Colorado, Aurora, and her colleagues.
Surveys assessing attitudes toward inpatient influenza vaccination were given to parents/caregivers of general pediatric inpatients and to inpatient physicians, residents, nurses, physician assistants, and nurse practitioners at Children’s Hospital Colorado in Aurora between October 2014 and March 2015. Response rates were 95% of the 1,053 parents/caregivers and 58% of the 339 providers.
The parents agreed that the flu is a serious disease (92%), that flu vaccines work (58%), that flu vaccines are safe (76%), and that the vaccines are needed annually (76%), the Dr. Rao and her colleagues found.
The providers thought the most common barriers to vaccination were parental refusal because of child illness (80%) and family misconceptions about the vaccine (74%). Also, 54% of providers forgot to ask about flu vaccination status and 46% forgot to order flu vaccines.
When asked what interventions might increase flu vaccination rates in the inpatient setting, 73% of providers agreed that personal reminders might help increase vaccination rates, but only 48% thought that provider education might help do so.
Read more in the journal Influenza and Other Respiratory Viruses (2017 Sep 5. doi: 10.1111/irv.12482.)
FDA approves triple-therapy inhaler for COPD
The Food and Drug Administration has approved Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol), a triple-therapy inhaler for the treatment of chronic obstructive pulmonary disease (COPD) in adult patients, according to a press release from GlaxoSmithKline and Innoviva.
Trelegy Ellipta combines an inhaled corticosteroid, a long-acting muscarinic antagonist, and a long-acting beta2-adrenergic agonist into an inhaler meant for once-daily use in people with COPD. Chronic bronchitis and/or emphysema patients are also indicated for treatment. The FDA-approved dosage is 100 mcg of fluticasone furoate, 62.5 mcg of umeclidinium, and 25 mcg of vilanterol.
“This approval represents a significant therapeutic convenience for those appropriate patients already on Breo Ellipta, that require additional bronchodilation or for those patients already on a combination of Breo Ellipta and Incruse Ellipta,” Mike Aguiar, CEO of Innoviva said in the press release.
In results supporting the FDA approval, the IMPACT study, a 52-week phase 3 clinical trial including 10,355 COPD patients sponsored by GSK, found that patients receiving Trelegy Ellipta experienced a 25% reduction in moderate to severe exacerbations compared to patients receiving Anoro Ellipta, and a 15% reduction in moderate to severe exacerbations, compared with patients receiving Relvar/Breo Ellipta. Change from baseline FEV1, change from baseline scores on the St George’s Respiratory Questionnaire, and time to first moderate/severe COPD exacerbation also were improved in the Trelegy Ellipta study group compared to the others.
“This is the first study to report a comparison of a single inhaler triple therapy with two dual therapies, providing much needed clinical evidence about the ability of a single inhaler triple therapy to reduce exacerbations,” Patrick Vallance, President of R&D at GSK, noted in a press release announcing the results of the IMPACT study.
The Food and Drug Administration has approved Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol), a triple-therapy inhaler for the treatment of chronic obstructive pulmonary disease (COPD) in adult patients, according to a press release from GlaxoSmithKline and Innoviva.
Trelegy Ellipta combines an inhaled corticosteroid, a long-acting muscarinic antagonist, and a long-acting beta2-adrenergic agonist into an inhaler meant for once-daily use in people with COPD. Chronic bronchitis and/or emphysema patients are also indicated for treatment. The FDA-approved dosage is 100 mcg of fluticasone furoate, 62.5 mcg of umeclidinium, and 25 mcg of vilanterol.
“This approval represents a significant therapeutic convenience for those appropriate patients already on Breo Ellipta, that require additional bronchodilation or for those patients already on a combination of Breo Ellipta and Incruse Ellipta,” Mike Aguiar, CEO of Innoviva said in the press release.
In results supporting the FDA approval, the IMPACT study, a 52-week phase 3 clinical trial including 10,355 COPD patients sponsored by GSK, found that patients receiving Trelegy Ellipta experienced a 25% reduction in moderate to severe exacerbations compared to patients receiving Anoro Ellipta, and a 15% reduction in moderate to severe exacerbations, compared with patients receiving Relvar/Breo Ellipta. Change from baseline FEV1, change from baseline scores on the St George’s Respiratory Questionnaire, and time to first moderate/severe COPD exacerbation also were improved in the Trelegy Ellipta study group compared to the others.
“This is the first study to report a comparison of a single inhaler triple therapy with two dual therapies, providing much needed clinical evidence about the ability of a single inhaler triple therapy to reduce exacerbations,” Patrick Vallance, President of R&D at GSK, noted in a press release announcing the results of the IMPACT study.
The Food and Drug Administration has approved Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol), a triple-therapy inhaler for the treatment of chronic obstructive pulmonary disease (COPD) in adult patients, according to a press release from GlaxoSmithKline and Innoviva.
Trelegy Ellipta combines an inhaled corticosteroid, a long-acting muscarinic antagonist, and a long-acting beta2-adrenergic agonist into an inhaler meant for once-daily use in people with COPD. Chronic bronchitis and/or emphysema patients are also indicated for treatment. The FDA-approved dosage is 100 mcg of fluticasone furoate, 62.5 mcg of umeclidinium, and 25 mcg of vilanterol.
“This approval represents a significant therapeutic convenience for those appropriate patients already on Breo Ellipta, that require additional bronchodilation or for those patients already on a combination of Breo Ellipta and Incruse Ellipta,” Mike Aguiar, CEO of Innoviva said in the press release.
In results supporting the FDA approval, the IMPACT study, a 52-week phase 3 clinical trial including 10,355 COPD patients sponsored by GSK, found that patients receiving Trelegy Ellipta experienced a 25% reduction in moderate to severe exacerbations compared to patients receiving Anoro Ellipta, and a 15% reduction in moderate to severe exacerbations, compared with patients receiving Relvar/Breo Ellipta. Change from baseline FEV1, change from baseline scores on the St George’s Respiratory Questionnaire, and time to first moderate/severe COPD exacerbation also were improved in the Trelegy Ellipta study group compared to the others.
“This is the first study to report a comparison of a single inhaler triple therapy with two dual therapies, providing much needed clinical evidence about the ability of a single inhaler triple therapy to reduce exacerbations,” Patrick Vallance, President of R&D at GSK, noted in a press release announcing the results of the IMPACT study.