User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Benralizumab approved for eosinophilic asthma
This is the only respiratory biologic to provide fast and “near-complete depletion of eosinophils within 24 hours,” according to the statement from AstraZeneca.
Similar adverse events were seen in patients who took benralizumab and the placebo. AstraZeneca will market benralizumab under the trade name Fasenra.
“This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long,” said Eugene Bleecker, MD, professor and codirector of genetics, genomics, and precision medicine at the University of Arizona in Tucson, in the statement.
This is the only respiratory biologic to provide fast and “near-complete depletion of eosinophils within 24 hours,” according to the statement from AstraZeneca.
Similar adverse events were seen in patients who took benralizumab and the placebo. AstraZeneca will market benralizumab under the trade name Fasenra.
“This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long,” said Eugene Bleecker, MD, professor and codirector of genetics, genomics, and precision medicine at the University of Arizona in Tucson, in the statement.
This is the only respiratory biologic to provide fast and “near-complete depletion of eosinophils within 24 hours,” according to the statement from AstraZeneca.
Similar adverse events were seen in patients who took benralizumab and the placebo. AstraZeneca will market benralizumab under the trade name Fasenra.
“This is an important day for severe, eosinophilic asthma patients who have had limited treatment options for far too long,” said Eugene Bleecker, MD, professor and codirector of genetics, genomics, and precision medicine at the University of Arizona in Tucson, in the statement.
DTaP vaccination rate highest in Maryland
, according to the Centers for Disease Control and Prevention.
Maryland’s coverage of required DTaP vaccine doses came in at a national high of 99.6% for children entering kindergarten in 2016-2017, while the District of Columbia had the nation’s lowest rate at 82.2%, Ranee Seither, MPH, of the National Center for Immunization and Respiratory Disease, and associates at the CDC, Atlanta, reported (MMWR. 2017 Oct 13;66[40]:1073-80).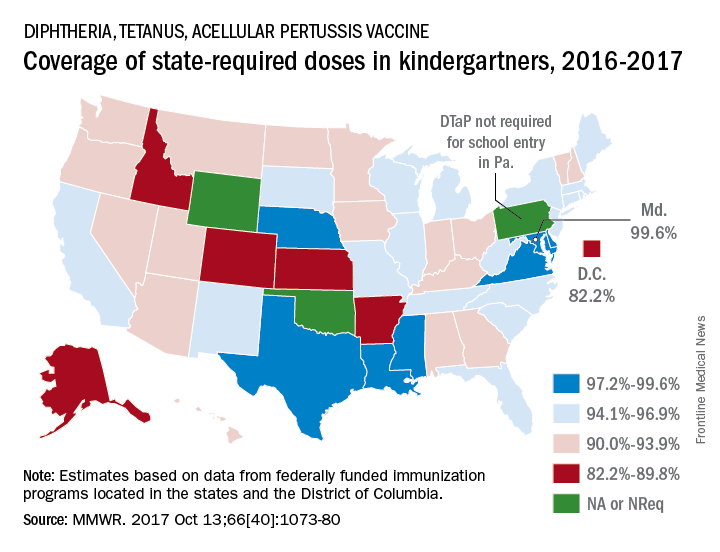
There is also variation among the states in the number of doses required for kindergarten entry: Most require five, but Illinois, Maryland, Virginia, and Wisconsin require four; Nebraska requires three; and Pennsylvania does not require pertussis vaccine. Oklahoma and Wyoming did not report vaccination coverage “because of widespread problems with the quality of data reported by schools,” they noted.
Nationally, median coverage for state-required doses of the DTaP vaccine was 94.5%, according to data from federally funded immunization programs in the 50 states and D.C., which included 3,973,172 kindergartners for the 2016-2017 school year.
, according to the Centers for Disease Control and Prevention.
Maryland’s coverage of required DTaP vaccine doses came in at a national high of 99.6% for children entering kindergarten in 2016-2017, while the District of Columbia had the nation’s lowest rate at 82.2%, Ranee Seither, MPH, of the National Center for Immunization and Respiratory Disease, and associates at the CDC, Atlanta, reported (MMWR. 2017 Oct 13;66[40]:1073-80).
There is also variation among the states in the number of doses required for kindergarten entry: Most require five, but Illinois, Maryland, Virginia, and Wisconsin require four; Nebraska requires three; and Pennsylvania does not require pertussis vaccine. Oklahoma and Wyoming did not report vaccination coverage “because of widespread problems with the quality of data reported by schools,” they noted.
Nationally, median coverage for state-required doses of the DTaP vaccine was 94.5%, according to data from federally funded immunization programs in the 50 states and D.C., which included 3,973,172 kindergartners for the 2016-2017 school year.
, according to the Centers for Disease Control and Prevention.
Maryland’s coverage of required DTaP vaccine doses came in at a national high of 99.6% for children entering kindergarten in 2016-2017, while the District of Columbia had the nation’s lowest rate at 82.2%, Ranee Seither, MPH, of the National Center for Immunization and Respiratory Disease, and associates at the CDC, Atlanta, reported (MMWR. 2017 Oct 13;66[40]:1073-80).
There is also variation among the states in the number of doses required for kindergarten entry: Most require five, but Illinois, Maryland, Virginia, and Wisconsin require four; Nebraska requires three; and Pennsylvania does not require pertussis vaccine. Oklahoma and Wyoming did not report vaccination coverage “because of widespread problems with the quality of data reported by schools,” they noted.
Nationally, median coverage for state-required doses of the DTaP vaccine was 94.5%, according to data from federally funded immunization programs in the 50 states and D.C., which included 3,973,172 kindergartners for the 2016-2017 school year.
FROM MMWR
ARDS incidence is declining. Is it a preventable syndrome?
TORONTO – The incidence of acute respiratory distress syndrome (ARDS) is on the decline, according to a retrospective, population-based cohort study conducted at the Mayo Clinic in Rochester, Minn.
“This is very promising data in combating this syndrome,” reported Augustin Joseph of the Mayo Clinic, and “it suggests that ARDS may in part be a completely preventable disease.”
To see if ARDS incidence has continued to decline, Mr. Joseph’s group studied all patients admitted during 2009-2014 to the Mayo Clinic’s ICU, the only facility in the county that cares for ARDS patients. From 82,388 ICU admissions, they identified 505 patients with ARDS according to the Berlin definition of ARDS developed in 2012.
The number of annual cases dropped from 108 in 2009 to 59 in 2014, and the incidence steadily declined from 74.5 cases per 100,000 in 2009 to 39.3 per 100,000 in 2014.
Median age was 67 years in 2009 and 62 years in 2014. Hospital mortality ranged from 15% to 26% during the study period, while hospital length of stay ranged from 8 to 15 days, with no clear decline in either.
“For hospital and ICU mortality and hospital and ICU length of stay, we did not see much difference [from 2009 to 2014], so the overall picture between the Guangxi Li study and mine was that we did not see much of a difference in the patients who had ARDS, but [in terms of] preventing ARDS, the incidence has continued to decline,” Mr. Joseph reported.
While the earlier study used the American-European Consensus Conference (AECC) definition of ARDS, Mr. Joseph and his colleagues diagnosed ARDS according to the Berlin definition. One of the major changes seen in the new Berlin rules is that acute lung injury no longer exists and patients with a P/F ratio (PaO2/FiO2 ratio, or the ratio of arterial oxygen partial pressure to fractional inspired oxygen) between 200 and 300 are now considered to have “mild ARDS,” Mr. Joseph explained. With the AECC definition, a P/F ratio in this range was classified as acute lung injury and only one less than 200 was considered ARDS.
The researchers are now trying to parse out how changing ARDS diagnosis and management at their institution might be contributing to declining incidence, said Mr. Joseph.
TORONTO – The incidence of acute respiratory distress syndrome (ARDS) is on the decline, according to a retrospective, population-based cohort study conducted at the Mayo Clinic in Rochester, Minn.
“This is very promising data in combating this syndrome,” reported Augustin Joseph of the Mayo Clinic, and “it suggests that ARDS may in part be a completely preventable disease.”
To see if ARDS incidence has continued to decline, Mr. Joseph’s group studied all patients admitted during 2009-2014 to the Mayo Clinic’s ICU, the only facility in the county that cares for ARDS patients. From 82,388 ICU admissions, they identified 505 patients with ARDS according to the Berlin definition of ARDS developed in 2012.
The number of annual cases dropped from 108 in 2009 to 59 in 2014, and the incidence steadily declined from 74.5 cases per 100,000 in 2009 to 39.3 per 100,000 in 2014.
Median age was 67 years in 2009 and 62 years in 2014. Hospital mortality ranged from 15% to 26% during the study period, while hospital length of stay ranged from 8 to 15 days, with no clear decline in either.
“For hospital and ICU mortality and hospital and ICU length of stay, we did not see much difference [from 2009 to 2014], so the overall picture between the Guangxi Li study and mine was that we did not see much of a difference in the patients who had ARDS, but [in terms of] preventing ARDS, the incidence has continued to decline,” Mr. Joseph reported.
While the earlier study used the American-European Consensus Conference (AECC) definition of ARDS, Mr. Joseph and his colleagues diagnosed ARDS according to the Berlin definition. One of the major changes seen in the new Berlin rules is that acute lung injury no longer exists and patients with a P/F ratio (PaO2/FiO2 ratio, or the ratio of arterial oxygen partial pressure to fractional inspired oxygen) between 200 and 300 are now considered to have “mild ARDS,” Mr. Joseph explained. With the AECC definition, a P/F ratio in this range was classified as acute lung injury and only one less than 200 was considered ARDS.
The researchers are now trying to parse out how changing ARDS diagnosis and management at their institution might be contributing to declining incidence, said Mr. Joseph.
TORONTO – The incidence of acute respiratory distress syndrome (ARDS) is on the decline, according to a retrospective, population-based cohort study conducted at the Mayo Clinic in Rochester, Minn.
“This is very promising data in combating this syndrome,” reported Augustin Joseph of the Mayo Clinic, and “it suggests that ARDS may in part be a completely preventable disease.”
To see if ARDS incidence has continued to decline, Mr. Joseph’s group studied all patients admitted during 2009-2014 to the Mayo Clinic’s ICU, the only facility in the county that cares for ARDS patients. From 82,388 ICU admissions, they identified 505 patients with ARDS according to the Berlin definition of ARDS developed in 2012.
The number of annual cases dropped from 108 in 2009 to 59 in 2014, and the incidence steadily declined from 74.5 cases per 100,000 in 2009 to 39.3 per 100,000 in 2014.
Median age was 67 years in 2009 and 62 years in 2014. Hospital mortality ranged from 15% to 26% during the study period, while hospital length of stay ranged from 8 to 15 days, with no clear decline in either.
“For hospital and ICU mortality and hospital and ICU length of stay, we did not see much difference [from 2009 to 2014], so the overall picture between the Guangxi Li study and mine was that we did not see much of a difference in the patients who had ARDS, but [in terms of] preventing ARDS, the incidence has continued to decline,” Mr. Joseph reported.
While the earlier study used the American-European Consensus Conference (AECC) definition of ARDS, Mr. Joseph and his colleagues diagnosed ARDS according to the Berlin definition. One of the major changes seen in the new Berlin rules is that acute lung injury no longer exists and patients with a P/F ratio (PaO2/FiO2 ratio, or the ratio of arterial oxygen partial pressure to fractional inspired oxygen) between 200 and 300 are now considered to have “mild ARDS,” Mr. Joseph explained. With the AECC definition, a P/F ratio in this range was classified as acute lung injury and only one less than 200 was considered ARDS.
The researchers are now trying to parse out how changing ARDS diagnosis and management at their institution might be contributing to declining incidence, said Mr. Joseph.
AT CHEST 2017
Key clinical point: The incidence of acute respiratory distress syndrome is declining, an indication that it may be preventable, according to researchers.
Major finding: The number of annual cases dropped from 108 in 2009 to 59 in 2014, and the incidence steadily declined from 74.5 cases per 100,000 in 2009 to 39.3 per 100,000 in 2014.
Data source: Retrospective, population-based cohort study of all (505) patients admitted to the ICU for ARDS at a single center.
Disclosures: The authors reported having no relevant disclosures.
HPV vaccination cuts incidence of juvenile respiratory papillomatosis
Introduction of a national human papillomavirus vaccination program in Australia has been associated with declines in the incidence of juvenile-onset recurrent respiratory papillomatosis, according to a nationwide study.
Juvenile-onset recurrent respiratory papillomatosis (JORRP) is a rare condition characterized by recurring growths in the larynx that often require multiple operations to remove. The disease typically emerges around age 3-4 years and most cases are thought to be caused by human papillomavirus (HPV) subtypes 6 and 11, which are acquired from the mother during birth.
Overall, just 15 cases were reported during the course of the study; 7 in the 1st year, 3 in the 2nd year, 2 each in the 3rd and 4th years, and 1 case in the last year. The annual rates declined from 0.16 per 100,000 children aged 0-14 years in 2012 to 0.02 per 100,000 in 2016.
Of the cases identified, none of the mothers had been vaccinated against HPV before pregnancy and 20% had a history of genital warts. Seven cases were genotyped; 4 were HPV-6 and 3 were HPV-11, and 13 of the 15 cases were born vaginally.
“Our data strongly suggest that the previously documented impact of quadrivalent HPV vaccination in dramatically reducing the prevalence of HPV-6 and HPV-11 genital infection in the Australian population is translating to a reduction in the risk of transmission to infants intrapartum and subsequent development in some of these children of JORRP,” wrote Daniel Novakovic, MD, of the University of Sydney Medical School, and his coinvestigators.
The authors noted that their initial estimate of infection rates was lower than that seen in other studies, such as the 0.5 per 100,000 rate seen in private health insurance data, and the 1.0 per 100,000 seen with Medicaid data in the United States.
Given that the study period started nearly 5 years after the vaccination program began, they suggested that this lower prevalence may reflect the early impact of the vaccine, particularly given that the prevalence of genital warts had already dramatically declined by that point.
However they also stressed that their study relied on clinicians actively reporting cases, and that given surveillance only began after the introduction of the vaccination program, no data were available on the incidence before that point.
The study was supported by a research grant from Merck and by the Australian Paediatric Surveillance Unit, which is supported by the Australian Government Department of Health. Three authors declared research funding from Merck/Seqirus for HPV studies. Two authors declared funding, speaking fees, and other support from a range of pharmaceutical companies. No other conflicts of interest were declared.
Introduction of a national human papillomavirus vaccination program in Australia has been associated with declines in the incidence of juvenile-onset recurrent respiratory papillomatosis, according to a nationwide study.
Juvenile-onset recurrent respiratory papillomatosis (JORRP) is a rare condition characterized by recurring growths in the larynx that often require multiple operations to remove. The disease typically emerges around age 3-4 years and most cases are thought to be caused by human papillomavirus (HPV) subtypes 6 and 11, which are acquired from the mother during birth.
Overall, just 15 cases were reported during the course of the study; 7 in the 1st year, 3 in the 2nd year, 2 each in the 3rd and 4th years, and 1 case in the last year. The annual rates declined from 0.16 per 100,000 children aged 0-14 years in 2012 to 0.02 per 100,000 in 2016.
Of the cases identified, none of the mothers had been vaccinated against HPV before pregnancy and 20% had a history of genital warts. Seven cases were genotyped; 4 were HPV-6 and 3 were HPV-11, and 13 of the 15 cases were born vaginally.
“Our data strongly suggest that the previously documented impact of quadrivalent HPV vaccination in dramatically reducing the prevalence of HPV-6 and HPV-11 genital infection in the Australian population is translating to a reduction in the risk of transmission to infants intrapartum and subsequent development in some of these children of JORRP,” wrote Daniel Novakovic, MD, of the University of Sydney Medical School, and his coinvestigators.
The authors noted that their initial estimate of infection rates was lower than that seen in other studies, such as the 0.5 per 100,000 rate seen in private health insurance data, and the 1.0 per 100,000 seen with Medicaid data in the United States.
Given that the study period started nearly 5 years after the vaccination program began, they suggested that this lower prevalence may reflect the early impact of the vaccine, particularly given that the prevalence of genital warts had already dramatically declined by that point.
However they also stressed that their study relied on clinicians actively reporting cases, and that given surveillance only began after the introduction of the vaccination program, no data were available on the incidence before that point.
The study was supported by a research grant from Merck and by the Australian Paediatric Surveillance Unit, which is supported by the Australian Government Department of Health. Three authors declared research funding from Merck/Seqirus for HPV studies. Two authors declared funding, speaking fees, and other support from a range of pharmaceutical companies. No other conflicts of interest were declared.
Introduction of a national human papillomavirus vaccination program in Australia has been associated with declines in the incidence of juvenile-onset recurrent respiratory papillomatosis, according to a nationwide study.
Juvenile-onset recurrent respiratory papillomatosis (JORRP) is a rare condition characterized by recurring growths in the larynx that often require multiple operations to remove. The disease typically emerges around age 3-4 years and most cases are thought to be caused by human papillomavirus (HPV) subtypes 6 and 11, which are acquired from the mother during birth.
Overall, just 15 cases were reported during the course of the study; 7 in the 1st year, 3 in the 2nd year, 2 each in the 3rd and 4th years, and 1 case in the last year. The annual rates declined from 0.16 per 100,000 children aged 0-14 years in 2012 to 0.02 per 100,000 in 2016.
Of the cases identified, none of the mothers had been vaccinated against HPV before pregnancy and 20% had a history of genital warts. Seven cases were genotyped; 4 were HPV-6 and 3 were HPV-11, and 13 of the 15 cases were born vaginally.
“Our data strongly suggest that the previously documented impact of quadrivalent HPV vaccination in dramatically reducing the prevalence of HPV-6 and HPV-11 genital infection in the Australian population is translating to a reduction in the risk of transmission to infants intrapartum and subsequent development in some of these children of JORRP,” wrote Daniel Novakovic, MD, of the University of Sydney Medical School, and his coinvestigators.
The authors noted that their initial estimate of infection rates was lower than that seen in other studies, such as the 0.5 per 100,000 rate seen in private health insurance data, and the 1.0 per 100,000 seen with Medicaid data in the United States.
Given that the study period started nearly 5 years after the vaccination program began, they suggested that this lower prevalence may reflect the early impact of the vaccine, particularly given that the prevalence of genital warts had already dramatically declined by that point.
However they also stressed that their study relied on clinicians actively reporting cases, and that given surveillance only began after the introduction of the vaccination program, no data were available on the incidence before that point.
The study was supported by a research grant from Merck and by the Australian Paediatric Surveillance Unit, which is supported by the Australian Government Department of Health. Three authors declared research funding from Merck/Seqirus for HPV studies. Two authors declared funding, speaking fees, and other support from a range of pharmaceutical companies. No other conflicts of interest were declared.
FROM THE JOURNAL OF INFECTIOUS DISEASES
Key clinical point: The introduction of a human papillomavirus vaccination program in Australia has seen a decline in the incidence of juvenile-onset recurrent respiratory papillomatosis.
Major finding: The incidence of juvenile recurrent respiratory papillomatosis decreased from 0.16 per 100,000 children aged 0-14 years to 0.02 per 100,000 over 5 years shortly after the national HPV vaccination program was introduced.
Data source: Prospective study using data from the Australian Paediatric Surveillance Unit.
Disclosures: The study was supported by a research grant from Merck and by the Australian Paediatric Surveillance Unit, which is supported by the Australian Government Department of Health. Three authors declared research funding from Merck/Seqirus for HPV studies. Two authors declared funding, speaking fees, and other support from a range of pharmaceutical companies. No other conflicts of interest were declared.
Tezacaftor-ivacaftor safe, effective in Phe508del CF
, according to results of a 24-week randomized, placebo-controlled clinical trial.
Patients receiving the tezacaftor-ivacaftor combination experienced a mean increase in their percentage of predicted forced expiratory volume in 1 second of 3.4 percentage points, compared with a mean decrease of 0.6 percentage points in the control group, at the end of the trial (P less than .001). The pulmonary exacerbation rate was 35% lower in the tezacaftor-ivacaftor treatment arm than in the placebo arm (P = .005), data show. These results were recently published in the New England Journal of Medicine (2017 Nov 3. doi: 10.1056/NEJMoa1709846).
Ivacaftor was the first approved modulator of the cystic fibrosis transmembrane conductance regulator (CFTR) protein, and tezacaftor is an investigational CFTR corrector. Tezacaftor demonstrated efficacy in a previous phase 2 trial that included patients either homozygous for the Phe508del mutation or heterozygous for the Phe508del and G551D mutations, Dr. Taylor-Cousar and her coauthors said in their report.
The combination of ivacaftor and another CFTR corrector, lumacaftor, is already available to treat cystic fibrosis patients who are homozygous for the Phe508del CFTR mutation. However, not all patients can receive lumacaftor-ivacaftor because of its respiratory side effects, and lumacaftor is associated with “prohibitive drug-drug interactions” due to considerable cytochrome P-450-3A induction, according to the study authors.
“The improved safety profile of combination therapy with tezacaftor-ivacaftor, as compared with currently available therapy, in addition to its effect on multiple efficacy end points, supports its use in a broad range of patients with cystic fibrosis,” wrote Dr. Taylor-Cousar and her colleagues.
The phase 3 trial included 509 cystic fibrosis patients at least 12 years of age who were homozygous for the CFTR Phe508del mutation. The mean percentage of predicted forced expiratory volume in 1 second of the patients was 60.0, at baseline.
All patients were randomized to combination therapy with tezacaftor 100 mg once daily and ivacaftor 150 mg twice daily, or matched placebo. A total of 475 patients completed the 24-week trial. The incidence of serious adverse events was just 12.4% of tezacaftor-ivacaftor–treated patients, compared with 18.2% in the placebo arm, and no serious adverse events led to treatment discontinuation.
“The rate of respiratory adverse events was not higher in the tezacaftor-ivacaftor group than in the placebo group, which shows that the safety profile for tezacaftor-ivacaftor is better than that reported for lumacaftor-ivacaftor,” Dr. Taylor-Cousar and her colleagues wrote.
Treatments that modulate CFTR are promising, according to the authors, because they treat the underlying cause of cystic fibrosis.
Vertex Pharmaceuticals supported the study. Dr. Taylor-Cousar reported personal fees from Vertex Pharmaceuticals outside of the submitted work. Full disclosures for all authors were published on the New England Journal of Medicine website.
, according to results of a 24-week randomized, placebo-controlled clinical trial.
Patients receiving the tezacaftor-ivacaftor combination experienced a mean increase in their percentage of predicted forced expiratory volume in 1 second of 3.4 percentage points, compared with a mean decrease of 0.6 percentage points in the control group, at the end of the trial (P less than .001). The pulmonary exacerbation rate was 35% lower in the tezacaftor-ivacaftor treatment arm than in the placebo arm (P = .005), data show. These results were recently published in the New England Journal of Medicine (2017 Nov 3. doi: 10.1056/NEJMoa1709846).
Ivacaftor was the first approved modulator of the cystic fibrosis transmembrane conductance regulator (CFTR) protein, and tezacaftor is an investigational CFTR corrector. Tezacaftor demonstrated efficacy in a previous phase 2 trial that included patients either homozygous for the Phe508del mutation or heterozygous for the Phe508del and G551D mutations, Dr. Taylor-Cousar and her coauthors said in their report.
The combination of ivacaftor and another CFTR corrector, lumacaftor, is already available to treat cystic fibrosis patients who are homozygous for the Phe508del CFTR mutation. However, not all patients can receive lumacaftor-ivacaftor because of its respiratory side effects, and lumacaftor is associated with “prohibitive drug-drug interactions” due to considerable cytochrome P-450-3A induction, according to the study authors.
“The improved safety profile of combination therapy with tezacaftor-ivacaftor, as compared with currently available therapy, in addition to its effect on multiple efficacy end points, supports its use in a broad range of patients with cystic fibrosis,” wrote Dr. Taylor-Cousar and her colleagues.
The phase 3 trial included 509 cystic fibrosis patients at least 12 years of age who were homozygous for the CFTR Phe508del mutation. The mean percentage of predicted forced expiratory volume in 1 second of the patients was 60.0, at baseline.
All patients were randomized to combination therapy with tezacaftor 100 mg once daily and ivacaftor 150 mg twice daily, or matched placebo. A total of 475 patients completed the 24-week trial. The incidence of serious adverse events was just 12.4% of tezacaftor-ivacaftor–treated patients, compared with 18.2% in the placebo arm, and no serious adverse events led to treatment discontinuation.
“The rate of respiratory adverse events was not higher in the tezacaftor-ivacaftor group than in the placebo group, which shows that the safety profile for tezacaftor-ivacaftor is better than that reported for lumacaftor-ivacaftor,” Dr. Taylor-Cousar and her colleagues wrote.
Treatments that modulate CFTR are promising, according to the authors, because they treat the underlying cause of cystic fibrosis.
Vertex Pharmaceuticals supported the study. Dr. Taylor-Cousar reported personal fees from Vertex Pharmaceuticals outside of the submitted work. Full disclosures for all authors were published on the New England Journal of Medicine website.
, according to results of a 24-week randomized, placebo-controlled clinical trial.
Patients receiving the tezacaftor-ivacaftor combination experienced a mean increase in their percentage of predicted forced expiratory volume in 1 second of 3.4 percentage points, compared with a mean decrease of 0.6 percentage points in the control group, at the end of the trial (P less than .001). The pulmonary exacerbation rate was 35% lower in the tezacaftor-ivacaftor treatment arm than in the placebo arm (P = .005), data show. These results were recently published in the New England Journal of Medicine (2017 Nov 3. doi: 10.1056/NEJMoa1709846).
Ivacaftor was the first approved modulator of the cystic fibrosis transmembrane conductance regulator (CFTR) protein, and tezacaftor is an investigational CFTR corrector. Tezacaftor demonstrated efficacy in a previous phase 2 trial that included patients either homozygous for the Phe508del mutation or heterozygous for the Phe508del and G551D mutations, Dr. Taylor-Cousar and her coauthors said in their report.
The combination of ivacaftor and another CFTR corrector, lumacaftor, is already available to treat cystic fibrosis patients who are homozygous for the Phe508del CFTR mutation. However, not all patients can receive lumacaftor-ivacaftor because of its respiratory side effects, and lumacaftor is associated with “prohibitive drug-drug interactions” due to considerable cytochrome P-450-3A induction, according to the study authors.
“The improved safety profile of combination therapy with tezacaftor-ivacaftor, as compared with currently available therapy, in addition to its effect on multiple efficacy end points, supports its use in a broad range of patients with cystic fibrosis,” wrote Dr. Taylor-Cousar and her colleagues.
The phase 3 trial included 509 cystic fibrosis patients at least 12 years of age who were homozygous for the CFTR Phe508del mutation. The mean percentage of predicted forced expiratory volume in 1 second of the patients was 60.0, at baseline.
All patients were randomized to combination therapy with tezacaftor 100 mg once daily and ivacaftor 150 mg twice daily, or matched placebo. A total of 475 patients completed the 24-week trial. The incidence of serious adverse events was just 12.4% of tezacaftor-ivacaftor–treated patients, compared with 18.2% in the placebo arm, and no serious adverse events led to treatment discontinuation.
“The rate of respiratory adverse events was not higher in the tezacaftor-ivacaftor group than in the placebo group, which shows that the safety profile for tezacaftor-ivacaftor is better than that reported for lumacaftor-ivacaftor,” Dr. Taylor-Cousar and her colleagues wrote.
Treatments that modulate CFTR are promising, according to the authors, because they treat the underlying cause of cystic fibrosis.
Vertex Pharmaceuticals supported the study. Dr. Taylor-Cousar reported personal fees from Vertex Pharmaceuticals outside of the submitted work. Full disclosures for all authors were published on the New England Journal of Medicine website.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: In patients with cystic fibrosis homozygous for the Phe508del mutation, combined tezacaftor-ivacaftor treatment was effective, with a safety profile comparing favorably to currently available therapies.
Major finding: Patients receiving the tezacaftor-ivacaftor combination experienced a mean increase in their percentage of predicted forced expiratory volume in 1 second of 3.4 percentage points, compared with a decrease of 0.6 percentage points in the control group, at the end of the trial (P less than .001).
Data source: A phase 3, randomized, double-blind, multicenter, placebo-controlled, trial (EVOLVE) including 509 patients at least 12 years of age with cystic fibrosis homozygous for the Phe508del CFTR mutation.
Disclosures: Vertex Pharmaceuticals supported the study. First author Jennifer L. Taylor-Cousar, MD, reported personal fees from Vertex Pharmaceuticals outside of the submitted work. Full disclosures for all authors were published on the New England Journal of Medicine website.
VIDEO: Revised guidelines raise lung cancer screening age ceiling
TORONTO – A proposed change to CHEST’s lung cancer screening guideline calls for raising the upper age for screening recent cigarette smokers to 77 years of age from 74 years of age.
This proposal is part of draft guideline that was unveiled during the CHEST annual meeting but is still subject to tweaking by peer review until formal release in early 2018. The draft also offers expanded guidance on how to implement screening, containing three times as many recommendations as the current lung cancer screening guidelines (Chest. 2013 May; 143[5 Suppl]:e78S-e92S).
“We want screening to expand in a safe and effective way,” said Peter J. Mazzone, MD, chair of the expert panel that is preparing the revision for CHEST and a pulmonologist at the Cleveland Clinic. “We are less restrictive with these guidelines” than in the 2013 version.
Dr. Mazzone cited two major changes that will produce modest broadening of the criteria that determine which patients can appropriately get screening. The clearest change was the age range, which expanded from 55-74 years of age set in 2013 to reflect the age criterion for enrollment in the National Lung Screening Trial (New Engl J Med. 2011 Aug 4; 365[5]:395-409). Dr. Mazzone explained, though it remains short of the 80-year old ceiling recommended by the U.S. Preventive Services Task Force.
The second, subtler change eased back on the outright ban that the 2013 guidelines placed on screening anyone who falls outside the target age range and smoking history (at least 30 pack years and either being a current smoker or having recently quit within the past 15 years) and who is without severe comorbidities.
The guidelines from 2013 said that screening people who fell outside these limits “should not be performed.” In contrast, the new draft guideline simply said that people who fall outside of the age and smoking-history criteria but who are still considered high risk for lung cancer based on a risk-prediction calculator should not “routinely” undergo screening. Additionally, exceptions could be made for certain patients whose high risk appears to warrant screening, Dr. Mazzone and others from the expert panel noted.
The revision specified that a high-risk person outside of the core criteria might still be a reasonable candidate for screening if this person tallies at least a 1.51% risk of developing lung cancer during the next 6 years according to the PLCOM2012 risk calculator (New Engl J Med. 2013 Feb 21; 368[8]:728-36).
“Some of the evidence allowed us to be a little more flexible,” though not to the point of “opening screening widely” to people who fall outside the core target population; rather, clinicians get to have a little more discretion, said Dr. Mazzone, who directs the Cleveland Clinic’s Lung Cancer Program. “We hope this will lead to more patients being screened in a high quality way,” he said in an interview. The panel strove to “look beyond the National Lung Screening Trial and find other groups of patients who could benefit” from screening. “We say that other high-risk people should not, on the whole, be screened” but that clinicians could consider individuals as appropriate for screening on a case-by-case basis.
The revision “fills in the outline” for screening that was established in the 2013 guidelines, said Gerard A. Silvestri, MD, a member of the revision panel, in a video interview. The updated guideline better detailed who benefits the most from screening and who benefits less, as well as the potential complications screening may cause, said Dr. Silvestri, a professor of medicine and lung cancer pulmonologist at the Medical University of South Carolina in Charleston.
“The sweet spot for screening is patients with a medium lung cancer risk without many comorbidities. We are trying to come up with individualized risk profiling,” explained Dr. Silvestri during the CHEST session. He noted that, in the screening program he runs in Charleston, every person who contacts the program and is interested in screening undergoes risk profiling. Are there people with a risk profile that justifies screening but fall outside the proposed criteria? “Absolutely,” Dr. Silvestri said.
People considering screening also need to recognize its potential harms, noted Renda Soylemez Wiener, MD, another member of the expert panel who spoke at the meeting. She cited five potential harms: death or complications from a biopsy of a screen-detected nodule, surgery for a screen-detected lesion that turns out to be benign, the psychosocial impact of finding a lung nodule, over diagnosis, and the cumulative radiation exposure from serial low-dose chest CT scans. “All of these dangers are real and may be magnified or mitigated as low-dose CT screening is implemented in real world practice,” said Dr. Wiener, a pulmonologist at Boston University.
In addition to four evidence-based recommendations that help define who is and isn’t an appropriate screening candidate, the revised guideline also included 11 mostly consensus-based “suggestions” about how screening programs should ideally operate. These covered issues such as identifying symptomatic patients who require diagnosis rather than screening, having strategies to encourage compliance with annual screening, including smoking cessation treatments in screening programs, and having strategies that minimize overtreatment of potentially indolent cancers.
The goal of these suggestions is to help in the design of high-quality screening programs, said Dr. Mazzone. “It’s not just who you screen but also how you screen.”
On Twitter @mitchelzoler
M. Patricia Rivera, MD, FCCP, comments: The revised ACCP screening guidelines recommend expanding the screening age from 55-74 years (age criterion used in the National Lung Screening Trial) to 55-77 years. While this may be interpreted as raising the screening age ceiling, the new recommendation is in line with the age range approved by Medicare. We should also keep in mind that after modeling studies to predict the benefits and harms of screening programs using different screening intervals, age ranges, and smoking histories (duration and time since quitting), the USPSTF concluded in their final recommendation that screening adults aged 55-80 years with same smoking history and time since quitting used in the National Lung Screening Trial (NLST) had a reasonable balance of benefits and harms. Implementing lung cancer screening has been challenging, and studies have reported many patient-, provider-, and system-based barriers including conflicting upper age range recommendations.
While age and smoking history are important in identifying individuals at risk for lung cancer, development of lung cancer is likely multifactorial and several other risk factors need to be considered. The ACCP's revised guidelines provide flexibility when evaluating patients who do not meet the age and smoking history criteria for screening but who have a high risk for developing lung cancer based on risk prediction models. Following publication of the NLST results, secondary analysis of the data using a risk prediction model that takes into account additional risk factors for the development of lung cancer (race, COPD, and family history of lung cancer, among others) suggests risk prediction modeling may be helpful at identifying the individuals who are at highest risk for developing lung cancer. As pointed out by Dr. Mazzone and Dr. Silvestri, the ACCP lung cancer screening guidelines promote the expansion of lung cancer screening in a safe and effective way and encourage individualized risk profiling to aid in the selection of all individuals who will benefit from lung cancer screening.
M. Patricia Rivera, MD, FCCP, comments: The revised ACCP screening guidelines recommend expanding the screening age from 55-74 years (age criterion used in the National Lung Screening Trial) to 55-77 years. While this may be interpreted as raising the screening age ceiling, the new recommendation is in line with the age range approved by Medicare. We should also keep in mind that after modeling studies to predict the benefits and harms of screening programs using different screening intervals, age ranges, and smoking histories (duration and time since quitting), the USPSTF concluded in their final recommendation that screening adults aged 55-80 years with same smoking history and time since quitting used in the National Lung Screening Trial (NLST) had a reasonable balance of benefits and harms. Implementing lung cancer screening has been challenging, and studies have reported many patient-, provider-, and system-based barriers including conflicting upper age range recommendations.
While age and smoking history are important in identifying individuals at risk for lung cancer, development of lung cancer is likely multifactorial and several other risk factors need to be considered. The ACCP's revised guidelines provide flexibility when evaluating patients who do not meet the age and smoking history criteria for screening but who have a high risk for developing lung cancer based on risk prediction models. Following publication of the NLST results, secondary analysis of the data using a risk prediction model that takes into account additional risk factors for the development of lung cancer (race, COPD, and family history of lung cancer, among others) suggests risk prediction modeling may be helpful at identifying the individuals who are at highest risk for developing lung cancer. As pointed out by Dr. Mazzone and Dr. Silvestri, the ACCP lung cancer screening guidelines promote the expansion of lung cancer screening in a safe and effective way and encourage individualized risk profiling to aid in the selection of all individuals who will benefit from lung cancer screening.
M. Patricia Rivera, MD, FCCP, comments: The revised ACCP screening guidelines recommend expanding the screening age from 55-74 years (age criterion used in the National Lung Screening Trial) to 55-77 years. While this may be interpreted as raising the screening age ceiling, the new recommendation is in line with the age range approved by Medicare. We should also keep in mind that after modeling studies to predict the benefits and harms of screening programs using different screening intervals, age ranges, and smoking histories (duration and time since quitting), the USPSTF concluded in their final recommendation that screening adults aged 55-80 years with same smoking history and time since quitting used in the National Lung Screening Trial (NLST) had a reasonable balance of benefits and harms. Implementing lung cancer screening has been challenging, and studies have reported many patient-, provider-, and system-based barriers including conflicting upper age range recommendations.
While age and smoking history are important in identifying individuals at risk for lung cancer, development of lung cancer is likely multifactorial and several other risk factors need to be considered. The ACCP's revised guidelines provide flexibility when evaluating patients who do not meet the age and smoking history criteria for screening but who have a high risk for developing lung cancer based on risk prediction models. Following publication of the NLST results, secondary analysis of the data using a risk prediction model that takes into account additional risk factors for the development of lung cancer (race, COPD, and family history of lung cancer, among others) suggests risk prediction modeling may be helpful at identifying the individuals who are at highest risk for developing lung cancer. As pointed out by Dr. Mazzone and Dr. Silvestri, the ACCP lung cancer screening guidelines promote the expansion of lung cancer screening in a safe and effective way and encourage individualized risk profiling to aid in the selection of all individuals who will benefit from lung cancer screening.
TORONTO – A proposed change to CHEST’s lung cancer screening guideline calls for raising the upper age for screening recent cigarette smokers to 77 years of age from 74 years of age.
This proposal is part of draft guideline that was unveiled during the CHEST annual meeting but is still subject to tweaking by peer review until formal release in early 2018. The draft also offers expanded guidance on how to implement screening, containing three times as many recommendations as the current lung cancer screening guidelines (Chest. 2013 May; 143[5 Suppl]:e78S-e92S).
“We want screening to expand in a safe and effective way,” said Peter J. Mazzone, MD, chair of the expert panel that is preparing the revision for CHEST and a pulmonologist at the Cleveland Clinic. “We are less restrictive with these guidelines” than in the 2013 version.
Dr. Mazzone cited two major changes that will produce modest broadening of the criteria that determine which patients can appropriately get screening. The clearest change was the age range, which expanded from 55-74 years of age set in 2013 to reflect the age criterion for enrollment in the National Lung Screening Trial (New Engl J Med. 2011 Aug 4; 365[5]:395-409). Dr. Mazzone explained, though it remains short of the 80-year old ceiling recommended by the U.S. Preventive Services Task Force.
The second, subtler change eased back on the outright ban that the 2013 guidelines placed on screening anyone who falls outside the target age range and smoking history (at least 30 pack years and either being a current smoker or having recently quit within the past 15 years) and who is without severe comorbidities.
The guidelines from 2013 said that screening people who fell outside these limits “should not be performed.” In contrast, the new draft guideline simply said that people who fall outside of the age and smoking-history criteria but who are still considered high risk for lung cancer based on a risk-prediction calculator should not “routinely” undergo screening. Additionally, exceptions could be made for certain patients whose high risk appears to warrant screening, Dr. Mazzone and others from the expert panel noted.
The revision specified that a high-risk person outside of the core criteria might still be a reasonable candidate for screening if this person tallies at least a 1.51% risk of developing lung cancer during the next 6 years according to the PLCOM2012 risk calculator (New Engl J Med. 2013 Feb 21; 368[8]:728-36).
“Some of the evidence allowed us to be a little more flexible,” though not to the point of “opening screening widely” to people who fall outside the core target population; rather, clinicians get to have a little more discretion, said Dr. Mazzone, who directs the Cleveland Clinic’s Lung Cancer Program. “We hope this will lead to more patients being screened in a high quality way,” he said in an interview. The panel strove to “look beyond the National Lung Screening Trial and find other groups of patients who could benefit” from screening. “We say that other high-risk people should not, on the whole, be screened” but that clinicians could consider individuals as appropriate for screening on a case-by-case basis.
The revision “fills in the outline” for screening that was established in the 2013 guidelines, said Gerard A. Silvestri, MD, a member of the revision panel, in a video interview. The updated guideline better detailed who benefits the most from screening and who benefits less, as well as the potential complications screening may cause, said Dr. Silvestri, a professor of medicine and lung cancer pulmonologist at the Medical University of South Carolina in Charleston.
“The sweet spot for screening is patients with a medium lung cancer risk without many comorbidities. We are trying to come up with individualized risk profiling,” explained Dr. Silvestri during the CHEST session. He noted that, in the screening program he runs in Charleston, every person who contacts the program and is interested in screening undergoes risk profiling. Are there people with a risk profile that justifies screening but fall outside the proposed criteria? “Absolutely,” Dr. Silvestri said.
People considering screening also need to recognize its potential harms, noted Renda Soylemez Wiener, MD, another member of the expert panel who spoke at the meeting. She cited five potential harms: death or complications from a biopsy of a screen-detected nodule, surgery for a screen-detected lesion that turns out to be benign, the psychosocial impact of finding a lung nodule, over diagnosis, and the cumulative radiation exposure from serial low-dose chest CT scans. “All of these dangers are real and may be magnified or mitigated as low-dose CT screening is implemented in real world practice,” said Dr. Wiener, a pulmonologist at Boston University.
In addition to four evidence-based recommendations that help define who is and isn’t an appropriate screening candidate, the revised guideline also included 11 mostly consensus-based “suggestions” about how screening programs should ideally operate. These covered issues such as identifying symptomatic patients who require diagnosis rather than screening, having strategies to encourage compliance with annual screening, including smoking cessation treatments in screening programs, and having strategies that minimize overtreatment of potentially indolent cancers.
The goal of these suggestions is to help in the design of high-quality screening programs, said Dr. Mazzone. “It’s not just who you screen but also how you screen.”
On Twitter @mitchelzoler
TORONTO – A proposed change to CHEST’s lung cancer screening guideline calls for raising the upper age for screening recent cigarette smokers to 77 years of age from 74 years of age.
This proposal is part of draft guideline that was unveiled during the CHEST annual meeting but is still subject to tweaking by peer review until formal release in early 2018. The draft also offers expanded guidance on how to implement screening, containing three times as many recommendations as the current lung cancer screening guidelines (Chest. 2013 May; 143[5 Suppl]:e78S-e92S).
“We want screening to expand in a safe and effective way,” said Peter J. Mazzone, MD, chair of the expert panel that is preparing the revision for CHEST and a pulmonologist at the Cleveland Clinic. “We are less restrictive with these guidelines” than in the 2013 version.
Dr. Mazzone cited two major changes that will produce modest broadening of the criteria that determine which patients can appropriately get screening. The clearest change was the age range, which expanded from 55-74 years of age set in 2013 to reflect the age criterion for enrollment in the National Lung Screening Trial (New Engl J Med. 2011 Aug 4; 365[5]:395-409). Dr. Mazzone explained, though it remains short of the 80-year old ceiling recommended by the U.S. Preventive Services Task Force.
The second, subtler change eased back on the outright ban that the 2013 guidelines placed on screening anyone who falls outside the target age range and smoking history (at least 30 pack years and either being a current smoker or having recently quit within the past 15 years) and who is without severe comorbidities.
The guidelines from 2013 said that screening people who fell outside these limits “should not be performed.” In contrast, the new draft guideline simply said that people who fall outside of the age and smoking-history criteria but who are still considered high risk for lung cancer based on a risk-prediction calculator should not “routinely” undergo screening. Additionally, exceptions could be made for certain patients whose high risk appears to warrant screening, Dr. Mazzone and others from the expert panel noted.
The revision specified that a high-risk person outside of the core criteria might still be a reasonable candidate for screening if this person tallies at least a 1.51% risk of developing lung cancer during the next 6 years according to the PLCOM2012 risk calculator (New Engl J Med. 2013 Feb 21; 368[8]:728-36).
“Some of the evidence allowed us to be a little more flexible,” though not to the point of “opening screening widely” to people who fall outside the core target population; rather, clinicians get to have a little more discretion, said Dr. Mazzone, who directs the Cleveland Clinic’s Lung Cancer Program. “We hope this will lead to more patients being screened in a high quality way,” he said in an interview. The panel strove to “look beyond the National Lung Screening Trial and find other groups of patients who could benefit” from screening. “We say that other high-risk people should not, on the whole, be screened” but that clinicians could consider individuals as appropriate for screening on a case-by-case basis.
The revision “fills in the outline” for screening that was established in the 2013 guidelines, said Gerard A. Silvestri, MD, a member of the revision panel, in a video interview. The updated guideline better detailed who benefits the most from screening and who benefits less, as well as the potential complications screening may cause, said Dr. Silvestri, a professor of medicine and lung cancer pulmonologist at the Medical University of South Carolina in Charleston.
“The sweet spot for screening is patients with a medium lung cancer risk without many comorbidities. We are trying to come up with individualized risk profiling,” explained Dr. Silvestri during the CHEST session. He noted that, in the screening program he runs in Charleston, every person who contacts the program and is interested in screening undergoes risk profiling. Are there people with a risk profile that justifies screening but fall outside the proposed criteria? “Absolutely,” Dr. Silvestri said.
People considering screening also need to recognize its potential harms, noted Renda Soylemez Wiener, MD, another member of the expert panel who spoke at the meeting. She cited five potential harms: death or complications from a biopsy of a screen-detected nodule, surgery for a screen-detected lesion that turns out to be benign, the psychosocial impact of finding a lung nodule, over diagnosis, and the cumulative radiation exposure from serial low-dose chest CT scans. “All of these dangers are real and may be magnified or mitigated as low-dose CT screening is implemented in real world practice,” said Dr. Wiener, a pulmonologist at Boston University.
In addition to four evidence-based recommendations that help define who is and isn’t an appropriate screening candidate, the revised guideline also included 11 mostly consensus-based “suggestions” about how screening programs should ideally operate. These covered issues such as identifying symptomatic patients who require diagnosis rather than screening, having strategies to encourage compliance with annual screening, including smoking cessation treatments in screening programs, and having strategies that minimize overtreatment of potentially indolent cancers.
The goal of these suggestions is to help in the design of high-quality screening programs, said Dr. Mazzone. “It’s not just who you screen but also how you screen.”
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM CHEST 2017
Omalizumab helps asthma COPD overlap patients
TORONTO – Omalizumab (Xolair, Genentech) decreased asthma exacerbations and improved symptom control to a similar extent in patients with asthma chronic obstructive pulmonary disease (ACO) overlap as seen in patients with asthma but no COPD, in a study presented at the CHEST annual meeting.
While patients with COPD typically experience annual declines in lung function, at least some of the ACO patients in this study, which included one of the largest observational cohorts to date of patients with ACO, showed preserved lung function after 48 weeks of omalizumab treatment.
Dr. Hanania presented data from the “real-world” PROSPERO (Prospective Study to Evaluate Predictors of Clinical Effectiveness in Response to Omalizumab), which unlike many asthma studies, did not exclude patients with comorbid COPD. PROSPERO was a prospective, multicenter, observational, 48-week study of patients (n = 806) who were 12 years of age and older who were initiating omalizumab treatment for moderate to severe allergic asthma. Asthma control was assessed monthly using the Asthma Control Test (ACT).
Participants were identified as having ACO based on two approaches: 1. A positive medical history of asthma and COPD, or 2. A medical history of asthma (but not COPD), at least a 10-pack per year smoking history, and an forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) of less than 0.7. From the 728 study participants included in this secondary analysis, 56 were classified as ACO according to the first definition (ACO cohort A) and 59 according to the second (ACO cohort B). Thirty-seven patients fell into both groups.
“All groups had a reduction in their exacerbation rates through 12 months, and it didn’t differ whether they had ACO in cohort A or cohort B, or no ACO,” Dr. Hanania reported.
Additionally, all three groups showed clinically meaningful improvements in their ACT scores, with mean improvements of 4.1, 4.7, and 4.4 units for ACO cohort A, ACO cohort B, and non-ACO patients, respectively.
Postbronchodilator FEV1 at study end was improved by 36 mL in ACO cohort A and by 23 mL in the non-ACO cohort. But a 14 mL reduction in postbronchodilator FEV1 was noted in ACO cohort B, “a reminder that the cohort B population was those patients with fixed airway obstruction and smoking history,” said Dr. Hanania.
Mean age in the non-ACO population was 50 years, rising to 57.6 years in ACO cohort A and 55 years in ACO cohort B. All three groups had three or more asthma exacerbations in the 12 months before starting omalizumab, and all groups had mean ACT scores of less than 15 at baseline, indicating that they were all symptomatic.
Adverse events were consistent with the known safety profile of omalizumab.
“The significance of this study [is that] it’s one of the largest ACO cohorts that we know of and I think it encourages all of us to look at or re-visit both COPD therapies and asthma therapies in populations [not included] in clinical trials because in real life, these are the patients we see … and we don’t have evidence,” Dr. Hanania said.
Dr. Hanania reported receiving research support from Roche/Genentech, among other companies. Three of the investigators are employees of Genentech, the study’s sponsor.
TORONTO – Omalizumab (Xolair, Genentech) decreased asthma exacerbations and improved symptom control to a similar extent in patients with asthma chronic obstructive pulmonary disease (ACO) overlap as seen in patients with asthma but no COPD, in a study presented at the CHEST annual meeting.
While patients with COPD typically experience annual declines in lung function, at least some of the ACO patients in this study, which included one of the largest observational cohorts to date of patients with ACO, showed preserved lung function after 48 weeks of omalizumab treatment.
Dr. Hanania presented data from the “real-world” PROSPERO (Prospective Study to Evaluate Predictors of Clinical Effectiveness in Response to Omalizumab), which unlike many asthma studies, did not exclude patients with comorbid COPD. PROSPERO was a prospective, multicenter, observational, 48-week study of patients (n = 806) who were 12 years of age and older who were initiating omalizumab treatment for moderate to severe allergic asthma. Asthma control was assessed monthly using the Asthma Control Test (ACT).
Participants were identified as having ACO based on two approaches: 1. A positive medical history of asthma and COPD, or 2. A medical history of asthma (but not COPD), at least a 10-pack per year smoking history, and an forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) of less than 0.7. From the 728 study participants included in this secondary analysis, 56 were classified as ACO according to the first definition (ACO cohort A) and 59 according to the second (ACO cohort B). Thirty-seven patients fell into both groups.
“All groups had a reduction in their exacerbation rates through 12 months, and it didn’t differ whether they had ACO in cohort A or cohort B, or no ACO,” Dr. Hanania reported.
Additionally, all three groups showed clinically meaningful improvements in their ACT scores, with mean improvements of 4.1, 4.7, and 4.4 units for ACO cohort A, ACO cohort B, and non-ACO patients, respectively.
Postbronchodilator FEV1 at study end was improved by 36 mL in ACO cohort A and by 23 mL in the non-ACO cohort. But a 14 mL reduction in postbronchodilator FEV1 was noted in ACO cohort B, “a reminder that the cohort B population was those patients with fixed airway obstruction and smoking history,” said Dr. Hanania.
Mean age in the non-ACO population was 50 years, rising to 57.6 years in ACO cohort A and 55 years in ACO cohort B. All three groups had three or more asthma exacerbations in the 12 months before starting omalizumab, and all groups had mean ACT scores of less than 15 at baseline, indicating that they were all symptomatic.
Adverse events were consistent with the known safety profile of omalizumab.
“The significance of this study [is that] it’s one of the largest ACO cohorts that we know of and I think it encourages all of us to look at or re-visit both COPD therapies and asthma therapies in populations [not included] in clinical trials because in real life, these are the patients we see … and we don’t have evidence,” Dr. Hanania said.
Dr. Hanania reported receiving research support from Roche/Genentech, among other companies. Three of the investigators are employees of Genentech, the study’s sponsor.
TORONTO – Omalizumab (Xolair, Genentech) decreased asthma exacerbations and improved symptom control to a similar extent in patients with asthma chronic obstructive pulmonary disease (ACO) overlap as seen in patients with asthma but no COPD, in a study presented at the CHEST annual meeting.
While patients with COPD typically experience annual declines in lung function, at least some of the ACO patients in this study, which included one of the largest observational cohorts to date of patients with ACO, showed preserved lung function after 48 weeks of omalizumab treatment.
Dr. Hanania presented data from the “real-world” PROSPERO (Prospective Study to Evaluate Predictors of Clinical Effectiveness in Response to Omalizumab), which unlike many asthma studies, did not exclude patients with comorbid COPD. PROSPERO was a prospective, multicenter, observational, 48-week study of patients (n = 806) who were 12 years of age and older who were initiating omalizumab treatment for moderate to severe allergic asthma. Asthma control was assessed monthly using the Asthma Control Test (ACT).
Participants were identified as having ACO based on two approaches: 1. A positive medical history of asthma and COPD, or 2. A medical history of asthma (but not COPD), at least a 10-pack per year smoking history, and an forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) of less than 0.7. From the 728 study participants included in this secondary analysis, 56 were classified as ACO according to the first definition (ACO cohort A) and 59 according to the second (ACO cohort B). Thirty-seven patients fell into both groups.
“All groups had a reduction in their exacerbation rates through 12 months, and it didn’t differ whether they had ACO in cohort A or cohort B, or no ACO,” Dr. Hanania reported.
Additionally, all three groups showed clinically meaningful improvements in their ACT scores, with mean improvements of 4.1, 4.7, and 4.4 units for ACO cohort A, ACO cohort B, and non-ACO patients, respectively.
Postbronchodilator FEV1 at study end was improved by 36 mL in ACO cohort A and by 23 mL in the non-ACO cohort. But a 14 mL reduction in postbronchodilator FEV1 was noted in ACO cohort B, “a reminder that the cohort B population was those patients with fixed airway obstruction and smoking history,” said Dr. Hanania.
Mean age in the non-ACO population was 50 years, rising to 57.6 years in ACO cohort A and 55 years in ACO cohort B. All three groups had three or more asthma exacerbations in the 12 months before starting omalizumab, and all groups had mean ACT scores of less than 15 at baseline, indicating that they were all symptomatic.
Adverse events were consistent with the known safety profile of omalizumab.
“The significance of this study [is that] it’s one of the largest ACO cohorts that we know of and I think it encourages all of us to look at or re-visit both COPD therapies and asthma therapies in populations [not included] in clinical trials because in real life, these are the patients we see … and we don’t have evidence,” Dr. Hanania said.
Dr. Hanania reported receiving research support from Roche/Genentech, among other companies. Three of the investigators are employees of Genentech, the study’s sponsor.
AT CHEST 2017
Key clinical point: In patients with asthma COPD overlap (ACO), treatment with omalizumab was associated with decreased asthma exacerbations and improved symptom control, similar to that seen in non-ACO asthma patients.
Major finding: Asthma exacerbation numbers were reduced from baseline levels though month 12, from 3 or more exacerbations in both ACO and non ACO groups to 1.1 or less.
Data source: Subgroup analysis from a prospective observational study of omalizumab that focused on 78 patients (from 737 total) that had asthma and comorbid COPD according to one of two definitions.
Disclosures: Dr. Hanania reported receiving research support from Roche/Genentech, among other companies. Genentech sponsored the study and employs three of the investigators.
Lung injury risk higher with apheresis blood products
SAN DIEGO – , according to research presented at the annual meeting of the American Association of Blood Banks.
Compared with other RBC products, those derived from apheresis significantly increased pulmonary cell interleukin (IL)–6 and IL-8 production, and this was further exacerbated by cell stretching. Conversely, red cell–filtered products appeared to be the least likely to cause cell injury.
“Several studies have shown that red blood cell transfusion is associated with acute lung injury, and transfusion induces leakage in ICU patients,” said lead study author Mathijs Wirtz, MD, of the Academic Medical Center, Amsterdam.
ICU patients who did not receive any transfusions had significantly lower leakage than those who were transfused. “There also seems to be a synergy between transfusion and mechanical ventilation,” Dr. Wirtz said.
Studies have also shown that there are differences in the prevalence of transfusion-related acute lung injury, when comparing Europe to the United States. Storage and manufacturing methods do differ between Europe and the United States, Dr. Wirtz noted. “This led to our hypothesis that lung injury inflicted by red blood cell transfusion is influenced by manufacturing methods.”
In this study, Dr. Wirtz and his colleagues investigated the response of pulmonary cells to the different methods of manufacturing RBC products. Using type A or B blood obtained from eight donors, a variety of RBC products were manufactured for the study, including whole-blood filtered, red-cell filtered, apheresis derived, and whole-blood derived.
For measuring thrombin generation and analyzing extracellular vesicles (EV), supernatants were prepared after 4-5 days of storage for fresh and 41-42 days for stored. The researchers selected A549 type II alveolar cells to seed onto flexible membranes, which were then incubated with RBC supernatant also stretched 25% using a cell stretcher.
After 24 hours, the production of IL-8 and IL-6 was measured.
Both fresh and stored supernatants that were derived from apheresis significantly increased the production of IL-6 and IL-8 in pulmonary cells, compared with nonincubated controls and most of the other RBC products. The production of IL-6 and IL-8 was exacerbated by cell stretching.
Average IL-6 production in nonstretched cells was 91 pg/mL for fresh and 87 pg/mL for expired (P less than .05 vs. control and other RBC products). For stretched cells, it was 130 pg/mL and 150 pg/mL (P less than .05 vs. control). For controls, mean nonstretched and stretched production was 21 pg/mL and 85 pg/mL.
Mean IL-8 production in nonstretched cells was 2,100 pg/mL for fresh and 1,900 pg/mL for stored (P less than .05 vs. control and other RBC products). For stretched cells, the means were 4,100 pg/mL for fresh and 5,200 pg/mL for stored (P less than .05 vs. control).
The average nonstretched and stretched control IL-8 production was 1,200 pg/mL for fresh and 4,300 pg/mL for stored.
Products derived from apheresis also demonstrated a significantly higher ability to generate thrombin, compared with other RBC products, and a significantly increased number of RBC-derived EVs, compared with filtered red cell and whole blood–derived products (P less than .05).
However, incubated stretched cells from stored whole blood–filtered products had higher IL-8 production (16,000 pg/mL), compared with other products and stretched controls. The lowest mean levels of IL-6 were observed in supernatants derived from red cell–filtered products (nonstretched fresh and expired, 12 pg/mL and 8 pg/mL; stretched, 40 pg/mL and 36 pg/mL) and they did not appear to activate pulmonary cells. Levels of EVs were also low, compared with other blood products.
“We can conclude that manufacturing methods contribute to the differences in inducing lung injury, and especially the apheresis-derived products, which induced the most consistent injury in our model,” Dr. Wirtz said. “The red cell–filtered products appeared to be the safest.”
Dr. Wirtz had no disclosures.
SAN DIEGO – , according to research presented at the annual meeting of the American Association of Blood Banks.
Compared with other RBC products, those derived from apheresis significantly increased pulmonary cell interleukin (IL)–6 and IL-8 production, and this was further exacerbated by cell stretching. Conversely, red cell–filtered products appeared to be the least likely to cause cell injury.
“Several studies have shown that red blood cell transfusion is associated with acute lung injury, and transfusion induces leakage in ICU patients,” said lead study author Mathijs Wirtz, MD, of the Academic Medical Center, Amsterdam.
ICU patients who did not receive any transfusions had significantly lower leakage than those who were transfused. “There also seems to be a synergy between transfusion and mechanical ventilation,” Dr. Wirtz said.
Studies have also shown that there are differences in the prevalence of transfusion-related acute lung injury, when comparing Europe to the United States. Storage and manufacturing methods do differ between Europe and the United States, Dr. Wirtz noted. “This led to our hypothesis that lung injury inflicted by red blood cell transfusion is influenced by manufacturing methods.”
In this study, Dr. Wirtz and his colleagues investigated the response of pulmonary cells to the different methods of manufacturing RBC products. Using type A or B blood obtained from eight donors, a variety of RBC products were manufactured for the study, including whole-blood filtered, red-cell filtered, apheresis derived, and whole-blood derived.
For measuring thrombin generation and analyzing extracellular vesicles (EV), supernatants were prepared after 4-5 days of storage for fresh and 41-42 days for stored. The researchers selected A549 type II alveolar cells to seed onto flexible membranes, which were then incubated with RBC supernatant also stretched 25% using a cell stretcher.
After 24 hours, the production of IL-8 and IL-6 was measured.
Both fresh and stored supernatants that were derived from apheresis significantly increased the production of IL-6 and IL-8 in pulmonary cells, compared with nonincubated controls and most of the other RBC products. The production of IL-6 and IL-8 was exacerbated by cell stretching.
Average IL-6 production in nonstretched cells was 91 pg/mL for fresh and 87 pg/mL for expired (P less than .05 vs. control and other RBC products). For stretched cells, it was 130 pg/mL and 150 pg/mL (P less than .05 vs. control). For controls, mean nonstretched and stretched production was 21 pg/mL and 85 pg/mL.
Mean IL-8 production in nonstretched cells was 2,100 pg/mL for fresh and 1,900 pg/mL for stored (P less than .05 vs. control and other RBC products). For stretched cells, the means were 4,100 pg/mL for fresh and 5,200 pg/mL for stored (P less than .05 vs. control).
The average nonstretched and stretched control IL-8 production was 1,200 pg/mL for fresh and 4,300 pg/mL for stored.
Products derived from apheresis also demonstrated a significantly higher ability to generate thrombin, compared with other RBC products, and a significantly increased number of RBC-derived EVs, compared with filtered red cell and whole blood–derived products (P less than .05).
However, incubated stretched cells from stored whole blood–filtered products had higher IL-8 production (16,000 pg/mL), compared with other products and stretched controls. The lowest mean levels of IL-6 were observed in supernatants derived from red cell–filtered products (nonstretched fresh and expired, 12 pg/mL and 8 pg/mL; stretched, 40 pg/mL and 36 pg/mL) and they did not appear to activate pulmonary cells. Levels of EVs were also low, compared with other blood products.
“We can conclude that manufacturing methods contribute to the differences in inducing lung injury, and especially the apheresis-derived products, which induced the most consistent injury in our model,” Dr. Wirtz said. “The red cell–filtered products appeared to be the safest.”
Dr. Wirtz had no disclosures.
SAN DIEGO – , according to research presented at the annual meeting of the American Association of Blood Banks.
Compared with other RBC products, those derived from apheresis significantly increased pulmonary cell interleukin (IL)–6 and IL-8 production, and this was further exacerbated by cell stretching. Conversely, red cell–filtered products appeared to be the least likely to cause cell injury.
“Several studies have shown that red blood cell transfusion is associated with acute lung injury, and transfusion induces leakage in ICU patients,” said lead study author Mathijs Wirtz, MD, of the Academic Medical Center, Amsterdam.
ICU patients who did not receive any transfusions had significantly lower leakage than those who were transfused. “There also seems to be a synergy between transfusion and mechanical ventilation,” Dr. Wirtz said.
Studies have also shown that there are differences in the prevalence of transfusion-related acute lung injury, when comparing Europe to the United States. Storage and manufacturing methods do differ between Europe and the United States, Dr. Wirtz noted. “This led to our hypothesis that lung injury inflicted by red blood cell transfusion is influenced by manufacturing methods.”
In this study, Dr. Wirtz and his colleagues investigated the response of pulmonary cells to the different methods of manufacturing RBC products. Using type A or B blood obtained from eight donors, a variety of RBC products were manufactured for the study, including whole-blood filtered, red-cell filtered, apheresis derived, and whole-blood derived.
For measuring thrombin generation and analyzing extracellular vesicles (EV), supernatants were prepared after 4-5 days of storage for fresh and 41-42 days for stored. The researchers selected A549 type II alveolar cells to seed onto flexible membranes, which were then incubated with RBC supernatant also stretched 25% using a cell stretcher.
After 24 hours, the production of IL-8 and IL-6 was measured.
Both fresh and stored supernatants that were derived from apheresis significantly increased the production of IL-6 and IL-8 in pulmonary cells, compared with nonincubated controls and most of the other RBC products. The production of IL-6 and IL-8 was exacerbated by cell stretching.
Average IL-6 production in nonstretched cells was 91 pg/mL for fresh and 87 pg/mL for expired (P less than .05 vs. control and other RBC products). For stretched cells, it was 130 pg/mL and 150 pg/mL (P less than .05 vs. control). For controls, mean nonstretched and stretched production was 21 pg/mL and 85 pg/mL.
Mean IL-8 production in nonstretched cells was 2,100 pg/mL for fresh and 1,900 pg/mL for stored (P less than .05 vs. control and other RBC products). For stretched cells, the means were 4,100 pg/mL for fresh and 5,200 pg/mL for stored (P less than .05 vs. control).
The average nonstretched and stretched control IL-8 production was 1,200 pg/mL for fresh and 4,300 pg/mL for stored.
Products derived from apheresis also demonstrated a significantly higher ability to generate thrombin, compared with other RBC products, and a significantly increased number of RBC-derived EVs, compared with filtered red cell and whole blood–derived products (P less than .05).
However, incubated stretched cells from stored whole blood–filtered products had higher IL-8 production (16,000 pg/mL), compared with other products and stretched controls. The lowest mean levels of IL-6 were observed in supernatants derived from red cell–filtered products (nonstretched fresh and expired, 12 pg/mL and 8 pg/mL; stretched, 40 pg/mL and 36 pg/mL) and they did not appear to activate pulmonary cells. Levels of EVs were also low, compared with other blood products.
“We can conclude that manufacturing methods contribute to the differences in inducing lung injury, and especially the apheresis-derived products, which induced the most consistent injury in our model,” Dr. Wirtz said. “The red cell–filtered products appeared to be the safest.”
Dr. Wirtz had no disclosures.
From AABB17
Key clinical point: The method of manufacturing blood products can markedly influence the interaction of RBC products with lung cells, especially in patients on mechanical ventilation.
Major finding: Apheresis-derived products are the most consistent in causing injuries, while red cell–filtered products appear to be the safest in avoiding lung injury.
Data source: An experimental study that investigated different manufacturing methods of RBC products and the response of pulmonary cells in an in vitro model of mechanical ventilation.
Disclosures: Dr. Wirtz had no disclosures.
OSA home testing less expensive than polysomnography
Home respiratory polygraphy had similar efficacy with substantially lower per-patient cost, compared with traditional polysomnography for diagnosing obstructive sleep apnea, a study showed.
Obstructive sleep apnea (OSA) is a common chronic disease associated with higher risk of cardiovascular disease and traffic accidents and a lower quality of life. Although expensive and time intensive, the polysomnography (PSG) has been the preferred test for diagnosing OSA. Home respiratory polygraphy (HRP) uses portable devices that are less complex than polysomnography and has been shown to have similar effectiveness in diagnosing OSA, compared with PSG, in patients with a high clinical suspicion of OSA. However, there is limited evidence for the cost effectiveness of HRP, compared with PSG (Am J Respir Crit Care Med. 2017 Nov 1;196[9]:1181-90).
The investigators conducted a multicenter, randomized controlled, noninferiority trial and cost-effectiveness analysis comparing PSG with HRP. Inclusion criteria included snoring or observed sleep apnea, Epworth Sleepiness Scale (ESS)of 10 or higher, and no suspicion of alternative causes for daytime sleepiness. Patients with a suspicion for OSA were randomized to polysomnography or respiratory polygraphy protocols. Both arms received counseling on proper sleep hygiene; counseling on weight loss, if overweight; and auto-CPAP titration if continuous positive airway pressure (CPAP) was clinically indicated.
Assessment of CPAP compliance or dietary and sleep hygiene compliance was assessed at months 1 and 3. ESS, quality of life measures, well-being measures, 24-hour blood pressure monitoring, auto accidents, and cardiovascular events were assessed at baseline and at month 6.
CPAP treatment was indicated in 68% of the PSG arm, compared with 53% of the HRP arm. After intention-to-treat analysis, there was no statistically significant difference between the two groups for ESS improvement (HRP mean, –4.2, vs. PSG mean, –4.9; P = .14). The groups demonstrated similar results for quality of life, blood pressure, polysomnographic assessment at 6 months, CPAP compliance, and rates of cardiovascular events and accidents at follow-up.
The cost-effective analysis demonstrated respiratory polygraphy was less expensive, saving more than 400 euros/patient. “Because the effectiveness (ESS and QALYs [quality-adjusted life-years]) was similar between arms, the HRP protocol is preferable due to its lower cost,” the authors wrote.
In all, 430 patients were randomized to HRP or PSG and consisted mostly of men (70.5%) with a mean body mass index of 30.7 kg/m2. The groups had similar rates of alcohol consumption and hypertension.
Limitations of the study included unblinded randomization to the participants and researchers and the possibility of variability in therapeutic decisions. However, the authors noted that intraobserver variability was minimized by using the Spanish Sleep Network guidelines and centralized assessment.
“[The] HRP management protocol is not inferior to PSG and presents substantially lower costs. Therefore, PSG is not necessary for most patients with suspicion of OSA. This finding could change established clinical practice, with a clear economic benefit,” the authors concluded.
Home respiratory polygraphy continues to impress
This study adds strong evidence to support the use of home respiratory polygraphy for the diagnosis of obstructive sleep apnea in patients without major comorbidities such as severe chronic restrictive or obstructive lung disease, heart failure or unstable cardiovascular disease, major psychiatric diagnoses, and neuromuscular conditions, noted Ching Li Chai-Coetzer, MBBS, PhD, and R.
Doug McEvoy, MBBS, MD, in an accompanying editorial (Am J Respir Crit Care Med. 2017 Nov 1;196[9]:1096-8). However, lower-cost methods to diagnose OSA would still not address unmet needs such as the cost of continuous positive airway pressure and scarcity of sleep physicians to assess patients with OSA, and still may be too expensive for underresourced populations, they said.
Dr. Chai-Coetzer and Dr. McEvoy are affiliated with the Adelaide Institute for Sleep Health at Flinders University and the Sleep Health Service, Southern Adelaide Local Health Network, both in South Australia.
The study was supported by Sociedad Española de Neumología, Air Liquide (Spain), Asociacion de Neumologos del Sur, and Sociedad Extremeña de Neumología. The investigators report no disclosures.
Dr. Chai-Coetzer reported grants from National Health and Medical Research Council of Australia and nonfinancial support from Biotech Pharmaceuticals. Dr. McEvoy reported grants and nonfinancial support from Philips Respironics, nonfinancial support from ResMed, and grants from Fisher & Paykel.
Home respiratory polygraphy had similar efficacy with substantially lower per-patient cost, compared with traditional polysomnography for diagnosing obstructive sleep apnea, a study showed.
Obstructive sleep apnea (OSA) is a common chronic disease associated with higher risk of cardiovascular disease and traffic accidents and a lower quality of life. Although expensive and time intensive, the polysomnography (PSG) has been the preferred test for diagnosing OSA. Home respiratory polygraphy (HRP) uses portable devices that are less complex than polysomnography and has been shown to have similar effectiveness in diagnosing OSA, compared with PSG, in patients with a high clinical suspicion of OSA. However, there is limited evidence for the cost effectiveness of HRP, compared with PSG (Am J Respir Crit Care Med. 2017 Nov 1;196[9]:1181-90).
The investigators conducted a multicenter, randomized controlled, noninferiority trial and cost-effectiveness analysis comparing PSG with HRP. Inclusion criteria included snoring or observed sleep apnea, Epworth Sleepiness Scale (ESS)of 10 or higher, and no suspicion of alternative causes for daytime sleepiness. Patients with a suspicion for OSA were randomized to polysomnography or respiratory polygraphy protocols. Both arms received counseling on proper sleep hygiene; counseling on weight loss, if overweight; and auto-CPAP titration if continuous positive airway pressure (CPAP) was clinically indicated.
Assessment of CPAP compliance or dietary and sleep hygiene compliance was assessed at months 1 and 3. ESS, quality of life measures, well-being measures, 24-hour blood pressure monitoring, auto accidents, and cardiovascular events were assessed at baseline and at month 6.
CPAP treatment was indicated in 68% of the PSG arm, compared with 53% of the HRP arm. After intention-to-treat analysis, there was no statistically significant difference between the two groups for ESS improvement (HRP mean, –4.2, vs. PSG mean, –4.9; P = .14). The groups demonstrated similar results for quality of life, blood pressure, polysomnographic assessment at 6 months, CPAP compliance, and rates of cardiovascular events and accidents at follow-up.
The cost-effective analysis demonstrated respiratory polygraphy was less expensive, saving more than 400 euros/patient. “Because the effectiveness (ESS and QALYs [quality-adjusted life-years]) was similar between arms, the HRP protocol is preferable due to its lower cost,” the authors wrote.
In all, 430 patients were randomized to HRP or PSG and consisted mostly of men (70.5%) with a mean body mass index of 30.7 kg/m2. The groups had similar rates of alcohol consumption and hypertension.
Limitations of the study included unblinded randomization to the participants and researchers and the possibility of variability in therapeutic decisions. However, the authors noted that intraobserver variability was minimized by using the Spanish Sleep Network guidelines and centralized assessment.
“[The] HRP management protocol is not inferior to PSG and presents substantially lower costs. Therefore, PSG is not necessary for most patients with suspicion of OSA. This finding could change established clinical practice, with a clear economic benefit,” the authors concluded.
Home respiratory polygraphy continues to impress
This study adds strong evidence to support the use of home respiratory polygraphy for the diagnosis of obstructive sleep apnea in patients without major comorbidities such as severe chronic restrictive or obstructive lung disease, heart failure or unstable cardiovascular disease, major psychiatric diagnoses, and neuromuscular conditions, noted Ching Li Chai-Coetzer, MBBS, PhD, and R.
Doug McEvoy, MBBS, MD, in an accompanying editorial (Am J Respir Crit Care Med. 2017 Nov 1;196[9]:1096-8). However, lower-cost methods to diagnose OSA would still not address unmet needs such as the cost of continuous positive airway pressure and scarcity of sleep physicians to assess patients with OSA, and still may be too expensive for underresourced populations, they said.
Dr. Chai-Coetzer and Dr. McEvoy are affiliated with the Adelaide Institute for Sleep Health at Flinders University and the Sleep Health Service, Southern Adelaide Local Health Network, both in South Australia.
The study was supported by Sociedad Española de Neumología, Air Liquide (Spain), Asociacion de Neumologos del Sur, and Sociedad Extremeña de Neumología. The investigators report no disclosures.
Dr. Chai-Coetzer reported grants from National Health and Medical Research Council of Australia and nonfinancial support from Biotech Pharmaceuticals. Dr. McEvoy reported grants and nonfinancial support from Philips Respironics, nonfinancial support from ResMed, and grants from Fisher & Paykel.
Home respiratory polygraphy had similar efficacy with substantially lower per-patient cost, compared with traditional polysomnography for diagnosing obstructive sleep apnea, a study showed.
Obstructive sleep apnea (OSA) is a common chronic disease associated with higher risk of cardiovascular disease and traffic accidents and a lower quality of life. Although expensive and time intensive, the polysomnography (PSG) has been the preferred test for diagnosing OSA. Home respiratory polygraphy (HRP) uses portable devices that are less complex than polysomnography and has been shown to have similar effectiveness in diagnosing OSA, compared with PSG, in patients with a high clinical suspicion of OSA. However, there is limited evidence for the cost effectiveness of HRP, compared with PSG (Am J Respir Crit Care Med. 2017 Nov 1;196[9]:1181-90).
The investigators conducted a multicenter, randomized controlled, noninferiority trial and cost-effectiveness analysis comparing PSG with HRP. Inclusion criteria included snoring or observed sleep apnea, Epworth Sleepiness Scale (ESS)of 10 or higher, and no suspicion of alternative causes for daytime sleepiness. Patients with a suspicion for OSA were randomized to polysomnography or respiratory polygraphy protocols. Both arms received counseling on proper sleep hygiene; counseling on weight loss, if overweight; and auto-CPAP titration if continuous positive airway pressure (CPAP) was clinically indicated.
Assessment of CPAP compliance or dietary and sleep hygiene compliance was assessed at months 1 and 3. ESS, quality of life measures, well-being measures, 24-hour blood pressure monitoring, auto accidents, and cardiovascular events were assessed at baseline and at month 6.
CPAP treatment was indicated in 68% of the PSG arm, compared with 53% of the HRP arm. After intention-to-treat analysis, there was no statistically significant difference between the two groups for ESS improvement (HRP mean, –4.2, vs. PSG mean, –4.9; P = .14). The groups demonstrated similar results for quality of life, blood pressure, polysomnographic assessment at 6 months, CPAP compliance, and rates of cardiovascular events and accidents at follow-up.
The cost-effective analysis demonstrated respiratory polygraphy was less expensive, saving more than 400 euros/patient. “Because the effectiveness (ESS and QALYs [quality-adjusted life-years]) was similar between arms, the HRP protocol is preferable due to its lower cost,” the authors wrote.
In all, 430 patients were randomized to HRP or PSG and consisted mostly of men (70.5%) with a mean body mass index of 30.7 kg/m2. The groups had similar rates of alcohol consumption and hypertension.
Limitations of the study included unblinded randomization to the participants and researchers and the possibility of variability in therapeutic decisions. However, the authors noted that intraobserver variability was minimized by using the Spanish Sleep Network guidelines and centralized assessment.
“[The] HRP management protocol is not inferior to PSG and presents substantially lower costs. Therefore, PSG is not necessary for most patients with suspicion of OSA. This finding could change established clinical practice, with a clear economic benefit,” the authors concluded.
Home respiratory polygraphy continues to impress
This study adds strong evidence to support the use of home respiratory polygraphy for the diagnosis of obstructive sleep apnea in patients without major comorbidities such as severe chronic restrictive or obstructive lung disease, heart failure or unstable cardiovascular disease, major psychiatric diagnoses, and neuromuscular conditions, noted Ching Li Chai-Coetzer, MBBS, PhD, and R.
Doug McEvoy, MBBS, MD, in an accompanying editorial (Am J Respir Crit Care Med. 2017 Nov 1;196[9]:1096-8). However, lower-cost methods to diagnose OSA would still not address unmet needs such as the cost of continuous positive airway pressure and scarcity of sleep physicians to assess patients with OSA, and still may be too expensive for underresourced populations, they said.
Dr. Chai-Coetzer and Dr. McEvoy are affiliated with the Adelaide Institute for Sleep Health at Flinders University and the Sleep Health Service, Southern Adelaide Local Health Network, both in South Australia.
The study was supported by Sociedad Española de Neumología, Air Liquide (Spain), Asociacion de Neumologos del Sur, and Sociedad Extremeña de Neumología. The investigators report no disclosures.
Dr. Chai-Coetzer reported grants from National Health and Medical Research Council of Australia and nonfinancial support from Biotech Pharmaceuticals. Dr. McEvoy reported grants and nonfinancial support from Philips Respironics, nonfinancial support from ResMed, and grants from Fisher & Paykel.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Key clinical point: Home obstructive sleep apnea testing was less costly and noninferior to polysomnography.
Major finding: Using respiratory polygraphy instead of polysomnography results in savings of more than 400 euros/patient.
Data source: A multicenter, randomized controlled, noninferiority trial and cost-effectiveness analysis of 430 patients suspected of having OSA.
Disclosures: The study was supported by Sociedad Española de Neumología, Air Liquide (Spain), Asociacion de Neumologos del Sur, and Sociedad Extremeña de Neumología. The authors report no disclosures.
FDA asked to approve add-on drug for eosinophilic COPD
GlaxoSmithKline asked the Food and Drug Administration to approve an interleuklin-5 antagonist as an add-on maintenance therapy for patients with eosinophilic chronic obstructive pulmonary disease (COPD).
The pharmaceutical and health care company is seeking approval of mepolizumab to be used specifically to treat COPD patients with an eosinophilic phenotype. The drug currently is indicated to treat patients aged 12 years or older with severe asthma and asthma with an eosinophilic phenotype and is sold under the name Nucala, according to a GlaxoSmithKline statement issued November 7.
Headache, injection site reaction, back pain, and fatigue are the most common adverse reactions seen in patients who took mepolizumab during clinical trials.
Mepolizumab is not approved for the treatment of COPD anywhere in the world, and GlaxoSmithKline intends to also ask other countries’ regulatory authorities to allow this drug to be sold as a therapy for COPD.
GlaxoSmithKline asked the Food and Drug Administration to approve an interleuklin-5 antagonist as an add-on maintenance therapy for patients with eosinophilic chronic obstructive pulmonary disease (COPD).
The pharmaceutical and health care company is seeking approval of mepolizumab to be used specifically to treat COPD patients with an eosinophilic phenotype. The drug currently is indicated to treat patients aged 12 years or older with severe asthma and asthma with an eosinophilic phenotype and is sold under the name Nucala, according to a GlaxoSmithKline statement issued November 7.
Headache, injection site reaction, back pain, and fatigue are the most common adverse reactions seen in patients who took mepolizumab during clinical trials.
Mepolizumab is not approved for the treatment of COPD anywhere in the world, and GlaxoSmithKline intends to also ask other countries’ regulatory authorities to allow this drug to be sold as a therapy for COPD.
GlaxoSmithKline asked the Food and Drug Administration to approve an interleuklin-5 antagonist as an add-on maintenance therapy for patients with eosinophilic chronic obstructive pulmonary disease (COPD).
The pharmaceutical and health care company is seeking approval of mepolizumab to be used specifically to treat COPD patients with an eosinophilic phenotype. The drug currently is indicated to treat patients aged 12 years or older with severe asthma and asthma with an eosinophilic phenotype and is sold under the name Nucala, according to a GlaxoSmithKline statement issued November 7.
Headache, injection site reaction, back pain, and fatigue are the most common adverse reactions seen in patients who took mepolizumab during clinical trials.
Mepolizumab is not approved for the treatment of COPD anywhere in the world, and GlaxoSmithKline intends to also ask other countries’ regulatory authorities to allow this drug to be sold as a therapy for COPD.