User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
Multidisciplinary teams improve diagnoses in ILD
New research provides strong statistical support for the use of dynamic multidisciplinary discussion in the diagnosis of patients who may have interstitial lung diseases (ILD).
and it changed the diagnosis in 41% of the other cases.
The American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association adopted joint guidelines for the treatment of idiopathic pulmonary fibrosis in 2015, and the ATS and ERS updated guidelines for the classification and terminology for idiopathic interstitial pneumonias in 2013. The Lancet Respiratory Medicine published what some consider to be a landmark evaluation of multidisciplinary team agreement on diagnosis of interstitial lung disease following the adoption of these guidelines (Walsh SLF et al. 2016;4[7]:557-65). This study showed that in idiopathic pulmonary fibrosis, multidisciplinary team meetings “have a higher level of agreement on diagnoses, assign diagnoses with higher confidence more frequently, and provide diagnoses that have nonsignificant greater prognostic separation than do clinicians or radiologists in most cases,” the researchers wrote.
In the new study, MDD failed to produce a diagnosis or suggestions about a way forward in only 3.5% of patients, according to the study, which appeared March 30 in CHEST®.
According to Dr. Antin-Ozerkis, accurate diagnosis of ILD is crucial to treatment, but it can be challenging to achieve. The MDD approach has been recommended since 2002 by the ATS and ERS, she said.
The study authors, led by Laurens J. De Sadeleer, MD, of Belgium’s University Hospitals Leuven, define the MDD approach as one “in which expert ILD clinicians, radiologists, and pathologists integrate all available clinical data, laboratory results, high-resolution computed tomography [HRCT] findings, and lung biopsy [when performed].”
For the study, the researchers tracked pre-MDD and MDD diagnoses of 938 consecutive patients with possible ILD who were discussed during 2005-2015. Of these patients, referring physicians made preliminary diagnoses in 49% of cases; in the rest, physicians either failed to develop a diagnosis or offered multiple possible diagnoses.
MDD teams produced a change in diagnosis in 191 – 42% – of patients with a pre-MDD diagnosis. Another condition was diagnosed in 118 of these patients, and the MDD teams declined to classify the other 73 patients pending further investigation.
The MDD teams also were able to produce diagnoses in 80% of cases when referring physicians could not come up with diagnoses.
“Discrepancy between pre-MDD diagnosis before work-up and discussion was remarkable,” the study authors wrote, estimating that MDD added value for 70% of referred patients.
“We believe MDD should be a common practice in the diagnosis of every patient with suspected ILD,” the researchers said.
The study doesn’t examine the challenges of putting MDD into practice, but Dr. Antin-Ozerkis provided some perspective. “It may be difficult for physicians to take the time from a busy practice to meet with a multidisciplinary team. It can require resources to gather the data necessary to comprehensively assess each patient case. Additionally, maintaining staff with experienced pulmonologists, radiologists and pathologists may be costly.”
She added that “there are various ways in which MDD may occur,” and that the pros and cons of different methods have not been well studied. “This practice will likely evolve with the development of new biomarkers and other diagnostic strategies in IPF [idiopathic pulmonary fibrosis].”
Still, she said, “this joint undertaking is clearly vital in helping to guide clinical practice, including therapeutic decisions and discussion of prognosis. For now, any discussion between clinician, radiologist, and pathologist is of benefit.”
Research Foundation-Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for IPF and other ILDs by Boehringer, Promedior, Fibrogen and Roche. She noted that payments go directly to the university with no direct payments to the investigator.
SOURCE: De Sadeleer LJ et al. Chest. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
MDD strategy is crucial for accurate ILD diagnoses
The field of interstitial lung diseases (ILD) is challenging, with more than 200 disorders as possible diagnoses for patients who present to clinicians with similar symptoms and chest x-ray findings. The multidisciplinary discussion (MDD) strategy is very important for attaining an accurate ILD diagnosis.
We have had routine, formal, multidisciplinary discussions at our center since 2008. My guesstimate is that at least a third of patients referred as having idiopathic pulmonary fibrosis or another form of ILD by pulmonologists had been given the wrong diagnosis. Frequently, this was because of incorrect impressions provided by local radiologists and/or pathologists along with the clinician’s own limited knowledge of ILD.
In my experience, some patients described their pulmonologists as becoming irate with them when they asked for a second opinion, and I have had to try to avoid confrontations with referring physicians when trying to explain why the referral diagnosis was inaccurate.
Challenges to instituting the multidisciplinary discussion approach include coverage by health plans for a second-opinion evaluation, the willingness of physicians (for example, pulmonologists) outside of academic referral centers to refer patients to a center capable of adequately conducting an MDD, and patients’ desire to undergo an evaluation at centers of excellence where an MDD can be performed.
One must have also adequate resources to perform a proper MDD. But even in centers that refer patients, pulmonologists should confer with their colleague radiologists – and pathologists when appropriate – to try to make the most accurate diagnosis. And they should continue to question their diagnosis at follow-up appointments, as new symptoms and findings may arise or additional crucial information can become available over time that can point to an alternative diagnosis.
Kenneth C. Meyer, MD, MS, served as medical director of the lung transplant program and head of ILD at the University of Wisconsin–Madison. He reported no relevant disclosures.
Second MDD may be helpful for CTD-related ILD
Accumulating evidence suggests that multidisciplinary committees play a central role in improving the diagnostic accuracy of complex medical conditions. Interstitial lung disease (ILD) encompasses a number of clinical entities and no single diagnostic test alone can discriminate among the various causes of ILD. Instead, these diagnoses are based on a constellation of signs and symptoms, and radiographic, pathologic, and laboratory studies.
However, unanswered questions remain. First, it is unclear whether a single MDD is sufficient. The present study found that 20% of cases were unclassifiable after the MDD. A second MDD may be helpful, especially in patients with ILDs related to connective tissue disease (CTD). The rheumatic diseases most commonly associated with ILD (for example, systemic sclerosis, rheumatoid arthritis, myositis) often evolve at different rates, and not all of the signs and symptoms of these conditions may be present or apparent at the time of the ILD presentation. A second MDD discussion may be particularly helpful in patients presenting with a specific CTD-related autoantibody in the absence of clinical signs and symptoms of a CTD. Another unanswered question is whether MDDs actually improve clinically meaningful outcomes for patients, such as survival and quality of life. At our CTD-ILD Program at the University of California, Los Angeles, we have found that our MDD has augmented patient satisfaction with their care, and it has also improved our ability to identify patients who are eligible for specific clinical studies. Future research is needed to determine to assess the impact of MDD on a variety of patient-centered and practice/research-focused outcomes.
Elizabeth Volkmann, MD, is founder and codirector of the CTD-ILD Program at the University of California, Los Angeles. She disclosed serving as a consultant or as a member of an advisory board for Boehringer Ingelheim and Astellas Pharma. She has received grants from Boehringer Ingelheim, Merck Serono, and the Rheumatology Research Foundation.
MDD strategy is crucial for accurate ILD diagnoses
The field of interstitial lung diseases (ILD) is challenging, with more than 200 disorders as possible diagnoses for patients who present to clinicians with similar symptoms and chest x-ray findings. The multidisciplinary discussion (MDD) strategy is very important for attaining an accurate ILD diagnosis.
We have had routine, formal, multidisciplinary discussions at our center since 2008. My guesstimate is that at least a third of patients referred as having idiopathic pulmonary fibrosis or another form of ILD by pulmonologists had been given the wrong diagnosis. Frequently, this was because of incorrect impressions provided by local radiologists and/or pathologists along with the clinician’s own limited knowledge of ILD.
In my experience, some patients described their pulmonologists as becoming irate with them when they asked for a second opinion, and I have had to try to avoid confrontations with referring physicians when trying to explain why the referral diagnosis was inaccurate.
Challenges to instituting the multidisciplinary discussion approach include coverage by health plans for a second-opinion evaluation, the willingness of physicians (for example, pulmonologists) outside of academic referral centers to refer patients to a center capable of adequately conducting an MDD, and patients’ desire to undergo an evaluation at centers of excellence where an MDD can be performed.
One must have also adequate resources to perform a proper MDD. But even in centers that refer patients, pulmonologists should confer with their colleague radiologists – and pathologists when appropriate – to try to make the most accurate diagnosis. And they should continue to question their diagnosis at follow-up appointments, as new symptoms and findings may arise or additional crucial information can become available over time that can point to an alternative diagnosis.
Kenneth C. Meyer, MD, MS, served as medical director of the lung transplant program and head of ILD at the University of Wisconsin–Madison. He reported no relevant disclosures.
Second MDD may be helpful for CTD-related ILD
Accumulating evidence suggests that multidisciplinary committees play a central role in improving the diagnostic accuracy of complex medical conditions. Interstitial lung disease (ILD) encompasses a number of clinical entities and no single diagnostic test alone can discriminate among the various causes of ILD. Instead, these diagnoses are based on a constellation of signs and symptoms, and radiographic, pathologic, and laboratory studies.
However, unanswered questions remain. First, it is unclear whether a single MDD is sufficient. The present study found that 20% of cases were unclassifiable after the MDD. A second MDD may be helpful, especially in patients with ILDs related to connective tissue disease (CTD). The rheumatic diseases most commonly associated with ILD (for example, systemic sclerosis, rheumatoid arthritis, myositis) often evolve at different rates, and not all of the signs and symptoms of these conditions may be present or apparent at the time of the ILD presentation. A second MDD discussion may be particularly helpful in patients presenting with a specific CTD-related autoantibody in the absence of clinical signs and symptoms of a CTD. Another unanswered question is whether MDDs actually improve clinically meaningful outcomes for patients, such as survival and quality of life. At our CTD-ILD Program at the University of California, Los Angeles, we have found that our MDD has augmented patient satisfaction with their care, and it has also improved our ability to identify patients who are eligible for specific clinical studies. Future research is needed to determine to assess the impact of MDD on a variety of patient-centered and practice/research-focused outcomes.
Elizabeth Volkmann, MD, is founder and codirector of the CTD-ILD Program at the University of California, Los Angeles. She disclosed serving as a consultant or as a member of an advisory board for Boehringer Ingelheim and Astellas Pharma. She has received grants from Boehringer Ingelheim, Merck Serono, and the Rheumatology Research Foundation.
MDD strategy is crucial for accurate ILD diagnoses
The field of interstitial lung diseases (ILD) is challenging, with more than 200 disorders as possible diagnoses for patients who present to clinicians with similar symptoms and chest x-ray findings. The multidisciplinary discussion (MDD) strategy is very important for attaining an accurate ILD diagnosis.
We have had routine, formal, multidisciplinary discussions at our center since 2008. My guesstimate is that at least a third of patients referred as having idiopathic pulmonary fibrosis or another form of ILD by pulmonologists had been given the wrong diagnosis. Frequently, this was because of incorrect impressions provided by local radiologists and/or pathologists along with the clinician’s own limited knowledge of ILD.
In my experience, some patients described their pulmonologists as becoming irate with them when they asked for a second opinion, and I have had to try to avoid confrontations with referring physicians when trying to explain why the referral diagnosis was inaccurate.
Challenges to instituting the multidisciplinary discussion approach include coverage by health plans for a second-opinion evaluation, the willingness of physicians (for example, pulmonologists) outside of academic referral centers to refer patients to a center capable of adequately conducting an MDD, and patients’ desire to undergo an evaluation at centers of excellence where an MDD can be performed.
One must have also adequate resources to perform a proper MDD. But even in centers that refer patients, pulmonologists should confer with their colleague radiologists – and pathologists when appropriate – to try to make the most accurate diagnosis. And they should continue to question their diagnosis at follow-up appointments, as new symptoms and findings may arise or additional crucial information can become available over time that can point to an alternative diagnosis.
Kenneth C. Meyer, MD, MS, served as medical director of the lung transplant program and head of ILD at the University of Wisconsin–Madison. He reported no relevant disclosures.
Second MDD may be helpful for CTD-related ILD
Accumulating evidence suggests that multidisciplinary committees play a central role in improving the diagnostic accuracy of complex medical conditions. Interstitial lung disease (ILD) encompasses a number of clinical entities and no single diagnostic test alone can discriminate among the various causes of ILD. Instead, these diagnoses are based on a constellation of signs and symptoms, and radiographic, pathologic, and laboratory studies.
However, unanswered questions remain. First, it is unclear whether a single MDD is sufficient. The present study found that 20% of cases were unclassifiable after the MDD. A second MDD may be helpful, especially in patients with ILDs related to connective tissue disease (CTD). The rheumatic diseases most commonly associated with ILD (for example, systemic sclerosis, rheumatoid arthritis, myositis) often evolve at different rates, and not all of the signs and symptoms of these conditions may be present or apparent at the time of the ILD presentation. A second MDD discussion may be particularly helpful in patients presenting with a specific CTD-related autoantibody in the absence of clinical signs and symptoms of a CTD. Another unanswered question is whether MDDs actually improve clinically meaningful outcomes for patients, such as survival and quality of life. At our CTD-ILD Program at the University of California, Los Angeles, we have found that our MDD has augmented patient satisfaction with their care, and it has also improved our ability to identify patients who are eligible for specific clinical studies. Future research is needed to determine to assess the impact of MDD on a variety of patient-centered and practice/research-focused outcomes.
Elizabeth Volkmann, MD, is founder and codirector of the CTD-ILD Program at the University of California, Los Angeles. She disclosed serving as a consultant or as a member of an advisory board for Boehringer Ingelheim and Astellas Pharma. She has received grants from Boehringer Ingelheim, Merck Serono, and the Rheumatology Research Foundation.
New research provides strong statistical support for the use of dynamic multidisciplinary discussion in the diagnosis of patients who may have interstitial lung diseases (ILD).
and it changed the diagnosis in 41% of the other cases.
The American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association adopted joint guidelines for the treatment of idiopathic pulmonary fibrosis in 2015, and the ATS and ERS updated guidelines for the classification and terminology for idiopathic interstitial pneumonias in 2013. The Lancet Respiratory Medicine published what some consider to be a landmark evaluation of multidisciplinary team agreement on diagnosis of interstitial lung disease following the adoption of these guidelines (Walsh SLF et al. 2016;4[7]:557-65). This study showed that in idiopathic pulmonary fibrosis, multidisciplinary team meetings “have a higher level of agreement on diagnoses, assign diagnoses with higher confidence more frequently, and provide diagnoses that have nonsignificant greater prognostic separation than do clinicians or radiologists in most cases,” the researchers wrote.
In the new study, MDD failed to produce a diagnosis or suggestions about a way forward in only 3.5% of patients, according to the study, which appeared March 30 in CHEST®.
According to Dr. Antin-Ozerkis, accurate diagnosis of ILD is crucial to treatment, but it can be challenging to achieve. The MDD approach has been recommended since 2002 by the ATS and ERS, she said.
The study authors, led by Laurens J. De Sadeleer, MD, of Belgium’s University Hospitals Leuven, define the MDD approach as one “in which expert ILD clinicians, radiologists, and pathologists integrate all available clinical data, laboratory results, high-resolution computed tomography [HRCT] findings, and lung biopsy [when performed].”
For the study, the researchers tracked pre-MDD and MDD diagnoses of 938 consecutive patients with possible ILD who were discussed during 2005-2015. Of these patients, referring physicians made preliminary diagnoses in 49% of cases; in the rest, physicians either failed to develop a diagnosis or offered multiple possible diagnoses.
MDD teams produced a change in diagnosis in 191 – 42% – of patients with a pre-MDD diagnosis. Another condition was diagnosed in 118 of these patients, and the MDD teams declined to classify the other 73 patients pending further investigation.
The MDD teams also were able to produce diagnoses in 80% of cases when referring physicians could not come up with diagnoses.
“Discrepancy between pre-MDD diagnosis before work-up and discussion was remarkable,” the study authors wrote, estimating that MDD added value for 70% of referred patients.
“We believe MDD should be a common practice in the diagnosis of every patient with suspected ILD,” the researchers said.
The study doesn’t examine the challenges of putting MDD into practice, but Dr. Antin-Ozerkis provided some perspective. “It may be difficult for physicians to take the time from a busy practice to meet with a multidisciplinary team. It can require resources to gather the data necessary to comprehensively assess each patient case. Additionally, maintaining staff with experienced pulmonologists, radiologists and pathologists may be costly.”
She added that “there are various ways in which MDD may occur,” and that the pros and cons of different methods have not been well studied. “This practice will likely evolve with the development of new biomarkers and other diagnostic strategies in IPF [idiopathic pulmonary fibrosis].”
Still, she said, “this joint undertaking is clearly vital in helping to guide clinical practice, including therapeutic decisions and discussion of prognosis. For now, any discussion between clinician, radiologist, and pathologist is of benefit.”
Research Foundation-Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for IPF and other ILDs by Boehringer, Promedior, Fibrogen and Roche. She noted that payments go directly to the university with no direct payments to the investigator.
SOURCE: De Sadeleer LJ et al. Chest. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
New research provides strong statistical support for the use of dynamic multidisciplinary discussion in the diagnosis of patients who may have interstitial lung diseases (ILD).
and it changed the diagnosis in 41% of the other cases.
The American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association adopted joint guidelines for the treatment of idiopathic pulmonary fibrosis in 2015, and the ATS and ERS updated guidelines for the classification and terminology for idiopathic interstitial pneumonias in 2013. The Lancet Respiratory Medicine published what some consider to be a landmark evaluation of multidisciplinary team agreement on diagnosis of interstitial lung disease following the adoption of these guidelines (Walsh SLF et al. 2016;4[7]:557-65). This study showed that in idiopathic pulmonary fibrosis, multidisciplinary team meetings “have a higher level of agreement on diagnoses, assign diagnoses with higher confidence more frequently, and provide diagnoses that have nonsignificant greater prognostic separation than do clinicians or radiologists in most cases,” the researchers wrote.
In the new study, MDD failed to produce a diagnosis or suggestions about a way forward in only 3.5% of patients, according to the study, which appeared March 30 in CHEST®.
According to Dr. Antin-Ozerkis, accurate diagnosis of ILD is crucial to treatment, but it can be challenging to achieve. The MDD approach has been recommended since 2002 by the ATS and ERS, she said.
The study authors, led by Laurens J. De Sadeleer, MD, of Belgium’s University Hospitals Leuven, define the MDD approach as one “in which expert ILD clinicians, radiologists, and pathologists integrate all available clinical data, laboratory results, high-resolution computed tomography [HRCT] findings, and lung biopsy [when performed].”
For the study, the researchers tracked pre-MDD and MDD diagnoses of 938 consecutive patients with possible ILD who were discussed during 2005-2015. Of these patients, referring physicians made preliminary diagnoses in 49% of cases; in the rest, physicians either failed to develop a diagnosis or offered multiple possible diagnoses.
MDD teams produced a change in diagnosis in 191 – 42% – of patients with a pre-MDD diagnosis. Another condition was diagnosed in 118 of these patients, and the MDD teams declined to classify the other 73 patients pending further investigation.
The MDD teams also were able to produce diagnoses in 80% of cases when referring physicians could not come up with diagnoses.
“Discrepancy between pre-MDD diagnosis before work-up and discussion was remarkable,” the study authors wrote, estimating that MDD added value for 70% of referred patients.
“We believe MDD should be a common practice in the diagnosis of every patient with suspected ILD,” the researchers said.
The study doesn’t examine the challenges of putting MDD into practice, but Dr. Antin-Ozerkis provided some perspective. “It may be difficult for physicians to take the time from a busy practice to meet with a multidisciplinary team. It can require resources to gather the data necessary to comprehensively assess each patient case. Additionally, maintaining staff with experienced pulmonologists, radiologists and pathologists may be costly.”
She added that “there are various ways in which MDD may occur,” and that the pros and cons of different methods have not been well studied. “This practice will likely evolve with the development of new biomarkers and other diagnostic strategies in IPF [idiopathic pulmonary fibrosis].”
Still, she said, “this joint undertaking is clearly vital in helping to guide clinical practice, including therapeutic decisions and discussion of prognosis. For now, any discussion between clinician, radiologist, and pathologist is of benefit.”
Research Foundation-Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for IPF and other ILDs by Boehringer, Promedior, Fibrogen and Roche. She noted that payments go directly to the university with no direct payments to the investigator.
SOURCE: De Sadeleer LJ et al. Chest. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
FROM CHEST
Key clinical point: Multidisciplinary discussion (MDD) in cases of suspected interstitial lung disease frequently produces adjustments of previous diagnoses and new diagnoses when none existed previously.
Major finding: MDD teams produced a change in diagnosis in 42% of patients with a pre-MDD diagnosis and in 80% of those without one.
Study details: 938 consecutive patients at University Hospitals Leuven, Belgium, with possible ILD who underwent MDD diagnostics during 2005-2015.
Disclosures: Research Foundation–Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for idiopathic pulmonary fibrosis and other ILDs by Boehringer, Promedior, FibroGen, and Roche. She noted that payments go directly to the university, with no direct payments to the investigator.
Source: De Sadeleer LJ et al. Chest 2018. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
Asthma flourishing in its medical home
according to the Centers for Disease Control and Prevention.
Current asthma prevalence was 8.8% for adults aged 18 years and older who worked in health care and social assistance in 2011-2016, which put them above those in education services (8.2%); arts, entertainment, and recreation (8.1%); accommodation and food services (7.7%); and finance and insurance (7.5%). The overall rate for all working adults was 6.8%, Jacek M. Mazurek, MD, PhD, and Girija Syamlal, MBBS, reported in the Morbidity and Mortality Weekly Report.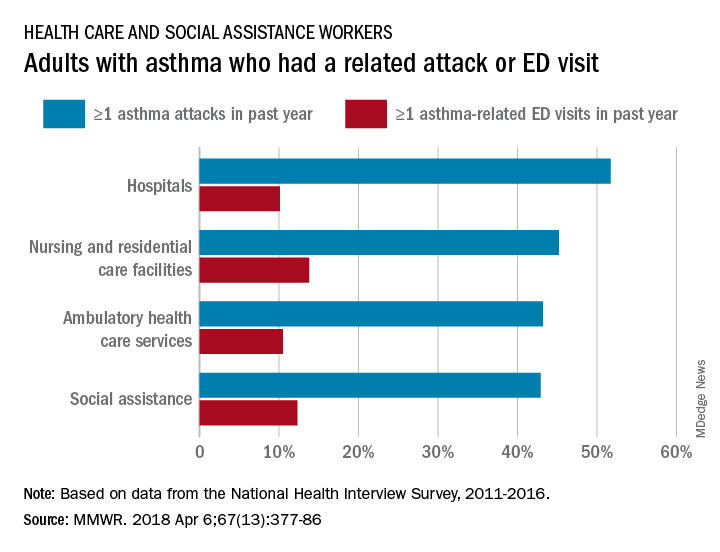
Among persons with asthma who were employed in health care and social assistance, 45.8% reported having at least one asthma attack in the previous year. Among the subgroups of the industry, those working in hospitals were highest with a 51.7% rate of past-year asthma attacks, followed by those working in nursing and residential care facilities at 45.2%, those working in ambulatory health care services at 43.2%, and those working in social assistance at 42.9%. The highest asthma attack rates among all industries were 57.3% for wood product manufacturing and 56.7% for plastics and rubber products manufacturing, the investigators said, based on data from the National Health Interview Survey.
Asthma-related visits to the emergency department in the past year were much less common for those in health care – 11.3% overall – and followed a pattern different from asthma attacks. Those working in nursing and residential care facilities were highest at 13.8%, with those in social assistance at 12.3%, those in ambulatory care at 10.5%, and those in hospitals the lowest at 10.1%. The highest ED-visit rate for any industry, 22.9%, was for workers in private households, said Dr. Mazurek and Dr. Syamlal, both of the respiratory health division at the CDC’s National Institute for Occupational Safety and Health in Morgantown, W.Va.
SOURCE: Mazurek JM, Syamlal G. MMWR. 2018 Apr 6;67(13):377-86.
according to the Centers for Disease Control and Prevention.
Current asthma prevalence was 8.8% for adults aged 18 years and older who worked in health care and social assistance in 2011-2016, which put them above those in education services (8.2%); arts, entertainment, and recreation (8.1%); accommodation and food services (7.7%); and finance and insurance (7.5%). The overall rate for all working adults was 6.8%, Jacek M. Mazurek, MD, PhD, and Girija Syamlal, MBBS, reported in the Morbidity and Mortality Weekly Report.
Among persons with asthma who were employed in health care and social assistance, 45.8% reported having at least one asthma attack in the previous year. Among the subgroups of the industry, those working in hospitals were highest with a 51.7% rate of past-year asthma attacks, followed by those working in nursing and residential care facilities at 45.2%, those working in ambulatory health care services at 43.2%, and those working in social assistance at 42.9%. The highest asthma attack rates among all industries were 57.3% for wood product manufacturing and 56.7% for plastics and rubber products manufacturing, the investigators said, based on data from the National Health Interview Survey.
Asthma-related visits to the emergency department in the past year were much less common for those in health care – 11.3% overall – and followed a pattern different from asthma attacks. Those working in nursing and residential care facilities were highest at 13.8%, with those in social assistance at 12.3%, those in ambulatory care at 10.5%, and those in hospitals the lowest at 10.1%. The highest ED-visit rate for any industry, 22.9%, was for workers in private households, said Dr. Mazurek and Dr. Syamlal, both of the respiratory health division at the CDC’s National Institute for Occupational Safety and Health in Morgantown, W.Va.
SOURCE: Mazurek JM, Syamlal G. MMWR. 2018 Apr 6;67(13):377-86.
according to the Centers for Disease Control and Prevention.
Current asthma prevalence was 8.8% for adults aged 18 years and older who worked in health care and social assistance in 2011-2016, which put them above those in education services (8.2%); arts, entertainment, and recreation (8.1%); accommodation and food services (7.7%); and finance and insurance (7.5%). The overall rate for all working adults was 6.8%, Jacek M. Mazurek, MD, PhD, and Girija Syamlal, MBBS, reported in the Morbidity and Mortality Weekly Report.
Among persons with asthma who were employed in health care and social assistance, 45.8% reported having at least one asthma attack in the previous year. Among the subgroups of the industry, those working in hospitals were highest with a 51.7% rate of past-year asthma attacks, followed by those working in nursing and residential care facilities at 45.2%, those working in ambulatory health care services at 43.2%, and those working in social assistance at 42.9%. The highest asthma attack rates among all industries were 57.3% for wood product manufacturing and 56.7% for plastics and rubber products manufacturing, the investigators said, based on data from the National Health Interview Survey.
Asthma-related visits to the emergency department in the past year were much less common for those in health care – 11.3% overall – and followed a pattern different from asthma attacks. Those working in nursing and residential care facilities were highest at 13.8%, with those in social assistance at 12.3%, those in ambulatory care at 10.5%, and those in hospitals the lowest at 10.1%. The highest ED-visit rate for any industry, 22.9%, was for workers in private households, said Dr. Mazurek and Dr. Syamlal, both of the respiratory health division at the CDC’s National Institute for Occupational Safety and Health in Morgantown, W.Va.
SOURCE: Mazurek JM, Syamlal G. MMWR. 2018 Apr 6;67(13):377-86.
FROM MMWR
EAGLES: Smoking cessation therapy did not up cardiovascular risk
among stable adult smokers with up to one year of follow-up.
“In what we believe to be the largest smoking cessation clinical trial and the only trial comparing NRT, bupropion, and varenicline [with] placebo, we found no signal that smoking cessation pharmacotherapy increases the risk of serious cardiovascular disease or cardiovascular adverse events in a general population of smokers,” concluded Neal L. Benowitz, MD, of the University of California, San Francisco, and his associates. “While the number of events was small, the incidence of serious cardiovascular events was low, suggesting that any absolute increase in risk that we might have missed would be low and not clinically meaningful.” The findings were reported online April 9 in JAMA Internal Medicine.
In this double-blind, multicenter, triple-dummy trial (EAGLES), Dr. Benowitz and his associates randomly assigned 8,058 adult smokers, who did not have acute or unstable cardiovascular disease, to receive bupropion (150 mg twice daily), varenicline (1 mg twice daily), NRT (21-mg/day patch with tapering), or placebo for 12 weeks, followed by 12 weeks of follow-up. A total of 4,595 patients agreed to be followed for another 28 weeks during an extension phase of the trial. More than half of the patients were women and the average age of a participant was 47 years. The primary endpoint was time to major adverse cardiovascular event (MACE), including cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. The researchers selected time to MACE as their primary endpoint to better detect differences among groups. One of the secondary end points was the occurrence of MACEs over the same 3 time intervals. Additionally, cardiovascular deaths, nonfatal MI, and nonfatal stroke (the components of MACE) were evaluated individually, as were hospitalizations for congestive heart failure and serious arrhythmias.
Differences in time to onset of MACE between all four patient groups, were not significant. The overall incidence of MACEs was less than 0.5% during all observation periods. There were also no significant differences in rates of the individual types of MACE, coronary revascularization, hospitalization for unstable angina, or new or worsening peripheral vascular disease requiring treatment among groups. Changes in body weight, blood pressure, and heart rate also were similar across patients.
There were five cardiovascular deaths, including one in the varenicline group, two in the bupropion group and two in the placebo group, according to the researchers. Overall the trial results “are consistent with and support previously published findings from meta-analyses and small clinical trials in smokers with known [cardiovascular disease],” they wrote.
GlaxoSmithKline and Pfizer, who make and market smoking cessation therapies, sponsored the study. Dr. Benowitz disclosed a consulting relationship with Pfizer and other pharmaceutical companies. He also has been a paid expert witness in litigation against tobacco companies. Eight coinvestigators disclosed ties to Pfizer, GlaxoSmithKline, and other companies.
SOURCE: Benowitz NL et al. JAMA Intern Med. 2018 Apr 9. doi: 10.1001/jamainternmed.2018.0397)
among stable adult smokers with up to one year of follow-up.
“In what we believe to be the largest smoking cessation clinical trial and the only trial comparing NRT, bupropion, and varenicline [with] placebo, we found no signal that smoking cessation pharmacotherapy increases the risk of serious cardiovascular disease or cardiovascular adverse events in a general population of smokers,” concluded Neal L. Benowitz, MD, of the University of California, San Francisco, and his associates. “While the number of events was small, the incidence of serious cardiovascular events was low, suggesting that any absolute increase in risk that we might have missed would be low and not clinically meaningful.” The findings were reported online April 9 in JAMA Internal Medicine.
In this double-blind, multicenter, triple-dummy trial (EAGLES), Dr. Benowitz and his associates randomly assigned 8,058 adult smokers, who did not have acute or unstable cardiovascular disease, to receive bupropion (150 mg twice daily), varenicline (1 mg twice daily), NRT (21-mg/day patch with tapering), or placebo for 12 weeks, followed by 12 weeks of follow-up. A total of 4,595 patients agreed to be followed for another 28 weeks during an extension phase of the trial. More than half of the patients were women and the average age of a participant was 47 years. The primary endpoint was time to major adverse cardiovascular event (MACE), including cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. The researchers selected time to MACE as their primary endpoint to better detect differences among groups. One of the secondary end points was the occurrence of MACEs over the same 3 time intervals. Additionally, cardiovascular deaths, nonfatal MI, and nonfatal stroke (the components of MACE) were evaluated individually, as were hospitalizations for congestive heart failure and serious arrhythmias.
Differences in time to onset of MACE between all four patient groups, were not significant. The overall incidence of MACEs was less than 0.5% during all observation periods. There were also no significant differences in rates of the individual types of MACE, coronary revascularization, hospitalization for unstable angina, or new or worsening peripheral vascular disease requiring treatment among groups. Changes in body weight, blood pressure, and heart rate also were similar across patients.
There were five cardiovascular deaths, including one in the varenicline group, two in the bupropion group and two in the placebo group, according to the researchers. Overall the trial results “are consistent with and support previously published findings from meta-analyses and small clinical trials in smokers with known [cardiovascular disease],” they wrote.
GlaxoSmithKline and Pfizer, who make and market smoking cessation therapies, sponsored the study. Dr. Benowitz disclosed a consulting relationship with Pfizer and other pharmaceutical companies. He also has been a paid expert witness in litigation against tobacco companies. Eight coinvestigators disclosed ties to Pfizer, GlaxoSmithKline, and other companies.
SOURCE: Benowitz NL et al. JAMA Intern Med. 2018 Apr 9. doi: 10.1001/jamainternmed.2018.0397)
among stable adult smokers with up to one year of follow-up.
“In what we believe to be the largest smoking cessation clinical trial and the only trial comparing NRT, bupropion, and varenicline [with] placebo, we found no signal that smoking cessation pharmacotherapy increases the risk of serious cardiovascular disease or cardiovascular adverse events in a general population of smokers,” concluded Neal L. Benowitz, MD, of the University of California, San Francisco, and his associates. “While the number of events was small, the incidence of serious cardiovascular events was low, suggesting that any absolute increase in risk that we might have missed would be low and not clinically meaningful.” The findings were reported online April 9 in JAMA Internal Medicine.
In this double-blind, multicenter, triple-dummy trial (EAGLES), Dr. Benowitz and his associates randomly assigned 8,058 adult smokers, who did not have acute or unstable cardiovascular disease, to receive bupropion (150 mg twice daily), varenicline (1 mg twice daily), NRT (21-mg/day patch with tapering), or placebo for 12 weeks, followed by 12 weeks of follow-up. A total of 4,595 patients agreed to be followed for another 28 weeks during an extension phase of the trial. More than half of the patients were women and the average age of a participant was 47 years. The primary endpoint was time to major adverse cardiovascular event (MACE), including cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. The researchers selected time to MACE as their primary endpoint to better detect differences among groups. One of the secondary end points was the occurrence of MACEs over the same 3 time intervals. Additionally, cardiovascular deaths, nonfatal MI, and nonfatal stroke (the components of MACE) were evaluated individually, as were hospitalizations for congestive heart failure and serious arrhythmias.
Differences in time to onset of MACE between all four patient groups, were not significant. The overall incidence of MACEs was less than 0.5% during all observation periods. There were also no significant differences in rates of the individual types of MACE, coronary revascularization, hospitalization for unstable angina, or new or worsening peripheral vascular disease requiring treatment among groups. Changes in body weight, blood pressure, and heart rate also were similar across patients.
There were five cardiovascular deaths, including one in the varenicline group, two in the bupropion group and two in the placebo group, according to the researchers. Overall the trial results “are consistent with and support previously published findings from meta-analyses and small clinical trials in smokers with known [cardiovascular disease],” they wrote.
GlaxoSmithKline and Pfizer, who make and market smoking cessation therapies, sponsored the study. Dr. Benowitz disclosed a consulting relationship with Pfizer and other pharmaceutical companies. He also has been a paid expert witness in litigation against tobacco companies. Eight coinvestigators disclosed ties to Pfizer, GlaxoSmithKline, and other companies.
SOURCE: Benowitz NL et al. JAMA Intern Med. 2018 Apr 9. doi: 10.1001/jamainternmed.2018.0397)
FROM JAMA INTERNAL MEDICINE
Key clinical point: The use of smoking cessation therapy did not increase the risk of cardiovascular events in adult smokers.
Major finding: There were no significant differences among groups in rates of major adverse cardiovascular events, rates of other pertinent cardiovascular events, time to cardiovascular events, blood pressure, or heart rate.
Study details: Double-blind, randomized, multicenter, triple-dummy trial of 8,058 adult smokers receiving nicotine replacement therapy, bupropion, varenicline, or placebo (EAGLES).
Disclosures: GlaxoSmithKline and Pfizer sponsored the study and make the drugs. Dr. Benowitz disclosed a consulting relationship with Pfizer and other pharmaceutical companies. He also has been a paid expert witness in litigation against tobacco companies. Eight coinvestigators disclosed ties to Pfizer, GlaxoSmithKline, and other companies.
Source: Benowitz NL et al. JAMA Intern Med. 2018 Apr 9. doi: 10.1001/jamainternmed.2018.0397.
The outcomes of “GOLD 2017”
After the Global Initiative for Chronic Obstructive Lung Disease released updated recommendations for grading COPD patients’ level of disease in November of 2016, Imran Iftikhar, MD, tried to incorporate them into his practice, but he encountered problems.
For one thing, the new classification system, which became known as GOLD 2017, uncoupled spirometry results from the ABCD treatment algorithm. “I found it wasn’t really helping me in terms of prognostication or COPD management,” said Dr. Iftikhar, section chief of pulmonary and critical care at Emory Saint Joseph’s Hospital, Atlanta. “Although the purpose of the GOLD classification was not really meant for prognostication, most practicing physicians are frequently asked about prognosis by patients, and I am not sure if the 2017 reclassification really helps with that.”
The GOLD 2017 classification simplified the chronic obstructive pulmonary disease staging that was available from 2011 to 2015 from three variables (spirometry thresholds, exacerbation risk, and dyspnea scale) to two variables (exacerbation risk and dyspnea scale). In the 2017 report, authors of the new guidelines characterized forced expiratory volume in 1 second (FEV1) as “a poor predictor of disease status” and proposed that clinicians derive ABCD groups exclusively from patient symptoms and their exacerbations. FEV1 is an “important parameter at the population level” in predicting hospitalization and mortality, the authors wrote, but keeping results separate “acknowledges the limitations of FEV1 in making treatment decisions for individualized patient care and highlights the importance of patient symptoms and exacerbation risks in guiding therapies in COPD.”
According to Meilan Han, MD, MS, a member of the GOLD Science Committee, since release of the 2017 guidelines, “clinicians have indicated that they like the flexibility the system provides in separating spirometry, symptoms, and exacerbation risk as this more accurately reflects the heterogeneity we see in the COPD patient population.” Nevertheless, how this approach influences long-term outcomes remains unclear.
Daniel Ouellette, MD, FCCP, a pulmonologist with the Henry Ford Health System in Detroit, described the GOLD 2017 criteria as “a good step forward” but said he wasn’t sure if the optimal or perfect tool exists for categorizing COPD patients’ level of disease.
“I think what we see is an effort to use all of these criteria to help us better treat our patients. I think ,” he said in an interview.
“All guidelines need to be modified as further research becomes available. I think that the frontiers of this area are going to be to incorporate new elements such as tobacco history, more emphasis on clinical signs and symptoms, and use of markers other than spirometry, such as eosinophil count, to categorize patients with COPD,” Dr. Ouellette added.
In an analysis of the GOLD 2017 criteria applied to 819 COPD patients in Spain and the United States, published online Nov. 3, 2017, in the American Journal of Respiratory and Critical Care Medicine, Carlos Cabrera López, MD, and his colleagues concluded that the mortality risk was better predicted by the 2015 GOLD classification system than by the 2017 iteration (Am J Respir Crit Care Med. 2018 Feb. doi: 10.101164/rccm.201707-1363OC).
The distribution of Charlson index scores also changed. Whereas group D was higher than B in 2015, they became similar in the 2017 system. For her part, Dr. Han emphasized that the primary goal of the GOLD ABCD classification system is to categorize patients with respect to treatment groups. “Current therapy targets symptoms and exacerbations, which are the key current elements of the classification schema,” she said in an interview. “The results of the Cabrera Lopez analysis are not necessarily unexpected, as FEV1 is associated with mortality.”
In a prospective, multicenter analysis, Portuguese researchers compared the performance of GOLD 2011 and 2017 in terms of how 200 COPD patients were reclassified, the level of agreement between the two iterations, and the performance of each to predict future exacerbations (COPD. 2018 Feb;15[1]; 21-6). They found that about half of patients classified as GOLD D under the 2011 guidelines became classified as GOLD B when the 2017 version was used, and the extent of agreement between the two iterations was moderate (P less than .001). They also found that the two versions of the guidelines were equivalently effective at predicting exacerbations (69.7% vs. 67.6% in the 2011 and 2017 iterations, respectively). In addition, patients who met the criteria for a GOLD B grouping in the 2017 iteration exacerbated 17% more often and had a lower percent predicted post bronchodilator FEV1 than did those who met the criteria for a GOLD B classification under the 2011 guidelines.
Dr. Han, who is also an associate professor of medicine at the University of Michigan Hospital, acknowledged that GOLD 2017 has resulted in the reclassification of some previously group D patients as group B patients. “Our primary goal is to aid clinicians with the diagnosis and management of patients with COPD,” she said. “We look forward to additional data coming in from ongoing clinical trials that will provide longer term data to further refine treatment algorithms.”
In a recent study of more than 33,000 Danish patients older than age 30 with COPD, researchers led by Anne Gedebjerg, MD, found that the GOLD 2017 ABCD classification did not predict all-cause and respiratory mortality more accurately than previous GOLD iterations from 2007 and 2011. Area under the curve for all-cause mortality was 0.61 for GOLD 2007, 0.61 for GOLD 2011, and 0.63 for GOLD 2017, while the area under the curve for respiratory mortality was 0.64 for GOLD 2007, 0.63 for GOLD 2011, and 0.65 for GOLD 2017 (Lancet Respir Med. 2018 Jan;6[3]:204-12).
However, when the spirometric stages 1-4 were combined with the A to D groupings based on symptoms and exacerbations, the 2017 classification predicted mortality with greater accuracy, compared with previous iterations (P less than .0001). “My practice is very much like this paper,” Dr. Iftikhar said. “I use both the spirometric grade and the ABCD grouping to specify which ‘group’ and ‘grade’ my patient belongs to. I think future investigators need to combine ABCD with spirometry classification to see how we can improve the classification system.”
In a commentary published in the same issue of the Lancet Respiratory Medicine as the large Danish study, Joan B. Soriano, MD, PhD, wrote that the 2011 GOLD guideline’s collapse of four spirometric thresholds (greater than 80%, 50%-80%, 30%-50%, and less than 30%) into just two (greater than 50% or 50% or less) “reduced the system’s ability to inform and predict mortality from the short term up to 10 years” (Lancet Respir Med. 2018 Jan;6[3]:165-6).
Lung function remains the best available biomarker for life expectancy in both patients with COPD and the general population,” wrote Dr. Soriano, a respiratory medicine researcher based in Madrid, Spain.
Additional important outcomes
Dr. Ouellette noted that while mortality is an important outcome for COPD patients, it’s not the only outcome of interest. “In addition to [trying to] help people live longer, which is certainly a desirable goal, we also want to make people be able to be more functional during their life, have fewer hospitalizations, and have less of a need of other types of supportive medical care for worsening of their disease,” he said. “The fact that the current guidelines don’t improve mortality more than the previous ones may not be a negative thing. It may tell us that the previous guidelines already did a pretty good job of helping us to improve mortality.”
Dr. Ouellette was quick to add that none of inhaled drugs currently available to treat COPD have been conclusively shown to improve mortality. “The only things we know that improve mortality for COPD patients are quitting smoking and using oxygen if a patient meets predefined goals for oxygen,” he said. “So the fact that GOLD criteria doesn’t improve mortality shouldn’t make us think that it’s not a useful tool. We already know that the medicines may not help people live longer.”
Dr. Han pointed out that spirometry “is still used to further clarify the choice of therapy recommended based on the nature and degree of airflow obstruction in light of severity of patient symptoms. The data are still designed to be used in conjunction to personalize therapy for patients.”
She added that the GOLD Science Committee “welcomes additional data analyses so that future recommendations can be further refined.”
Dr. Han disclosed that she has consulted for Boehringer Ingelheim, AstraZeneca, and GlaxoSmithKline. She has also received in-kind research support from Novartis and Sunovion.
Dr. Iftikhar reported having no financial disclosures. Dr. Ouellette is a member of CHEST® Physician’s editorial advisory board. He disclosed being part of a federally funded study being carried out by the Patient-Centered Outcomes Research Institute.
There was no industry involvement in the GOLD 2017 report, but many of its authors and board members had pharmaceutical company ties, and GOLD’s treatment advice relies on data from industry-sponsored studies.
After the Global Initiative for Chronic Obstructive Lung Disease released updated recommendations for grading COPD patients’ level of disease in November of 2016, Imran Iftikhar, MD, tried to incorporate them into his practice, but he encountered problems.
For one thing, the new classification system, which became known as GOLD 2017, uncoupled spirometry results from the ABCD treatment algorithm. “I found it wasn’t really helping me in terms of prognostication or COPD management,” said Dr. Iftikhar, section chief of pulmonary and critical care at Emory Saint Joseph’s Hospital, Atlanta. “Although the purpose of the GOLD classification was not really meant for prognostication, most practicing physicians are frequently asked about prognosis by patients, and I am not sure if the 2017 reclassification really helps with that.”
The GOLD 2017 classification simplified the chronic obstructive pulmonary disease staging that was available from 2011 to 2015 from three variables (spirometry thresholds, exacerbation risk, and dyspnea scale) to two variables (exacerbation risk and dyspnea scale). In the 2017 report, authors of the new guidelines characterized forced expiratory volume in 1 second (FEV1) as “a poor predictor of disease status” and proposed that clinicians derive ABCD groups exclusively from patient symptoms and their exacerbations. FEV1 is an “important parameter at the population level” in predicting hospitalization and mortality, the authors wrote, but keeping results separate “acknowledges the limitations of FEV1 in making treatment decisions for individualized patient care and highlights the importance of patient symptoms and exacerbation risks in guiding therapies in COPD.”
According to Meilan Han, MD, MS, a member of the GOLD Science Committee, since release of the 2017 guidelines, “clinicians have indicated that they like the flexibility the system provides in separating spirometry, symptoms, and exacerbation risk as this more accurately reflects the heterogeneity we see in the COPD patient population.” Nevertheless, how this approach influences long-term outcomes remains unclear.
Daniel Ouellette, MD, FCCP, a pulmonologist with the Henry Ford Health System in Detroit, described the GOLD 2017 criteria as “a good step forward” but said he wasn’t sure if the optimal or perfect tool exists for categorizing COPD patients’ level of disease.
“I think what we see is an effort to use all of these criteria to help us better treat our patients. I think ,” he said in an interview.
“All guidelines need to be modified as further research becomes available. I think that the frontiers of this area are going to be to incorporate new elements such as tobacco history, more emphasis on clinical signs and symptoms, and use of markers other than spirometry, such as eosinophil count, to categorize patients with COPD,” Dr. Ouellette added.
In an analysis of the GOLD 2017 criteria applied to 819 COPD patients in Spain and the United States, published online Nov. 3, 2017, in the American Journal of Respiratory and Critical Care Medicine, Carlos Cabrera López, MD, and his colleagues concluded that the mortality risk was better predicted by the 2015 GOLD classification system than by the 2017 iteration (Am J Respir Crit Care Med. 2018 Feb. doi: 10.101164/rccm.201707-1363OC).
The distribution of Charlson index scores also changed. Whereas group D was higher than B in 2015, they became similar in the 2017 system. For her part, Dr. Han emphasized that the primary goal of the GOLD ABCD classification system is to categorize patients with respect to treatment groups. “Current therapy targets symptoms and exacerbations, which are the key current elements of the classification schema,” she said in an interview. “The results of the Cabrera Lopez analysis are not necessarily unexpected, as FEV1 is associated with mortality.”
In a prospective, multicenter analysis, Portuguese researchers compared the performance of GOLD 2011 and 2017 in terms of how 200 COPD patients were reclassified, the level of agreement between the two iterations, and the performance of each to predict future exacerbations (COPD. 2018 Feb;15[1]; 21-6). They found that about half of patients classified as GOLD D under the 2011 guidelines became classified as GOLD B when the 2017 version was used, and the extent of agreement between the two iterations was moderate (P less than .001). They also found that the two versions of the guidelines were equivalently effective at predicting exacerbations (69.7% vs. 67.6% in the 2011 and 2017 iterations, respectively). In addition, patients who met the criteria for a GOLD B grouping in the 2017 iteration exacerbated 17% more often and had a lower percent predicted post bronchodilator FEV1 than did those who met the criteria for a GOLD B classification under the 2011 guidelines.
Dr. Han, who is also an associate professor of medicine at the University of Michigan Hospital, acknowledged that GOLD 2017 has resulted in the reclassification of some previously group D patients as group B patients. “Our primary goal is to aid clinicians with the diagnosis and management of patients with COPD,” she said. “We look forward to additional data coming in from ongoing clinical trials that will provide longer term data to further refine treatment algorithms.”
In a recent study of more than 33,000 Danish patients older than age 30 with COPD, researchers led by Anne Gedebjerg, MD, found that the GOLD 2017 ABCD classification did not predict all-cause and respiratory mortality more accurately than previous GOLD iterations from 2007 and 2011. Area under the curve for all-cause mortality was 0.61 for GOLD 2007, 0.61 for GOLD 2011, and 0.63 for GOLD 2017, while the area under the curve for respiratory mortality was 0.64 for GOLD 2007, 0.63 for GOLD 2011, and 0.65 for GOLD 2017 (Lancet Respir Med. 2018 Jan;6[3]:204-12).
However, when the spirometric stages 1-4 were combined with the A to D groupings based on symptoms and exacerbations, the 2017 classification predicted mortality with greater accuracy, compared with previous iterations (P less than .0001). “My practice is very much like this paper,” Dr. Iftikhar said. “I use both the spirometric grade and the ABCD grouping to specify which ‘group’ and ‘grade’ my patient belongs to. I think future investigators need to combine ABCD with spirometry classification to see how we can improve the classification system.”
In a commentary published in the same issue of the Lancet Respiratory Medicine as the large Danish study, Joan B. Soriano, MD, PhD, wrote that the 2011 GOLD guideline’s collapse of four spirometric thresholds (greater than 80%, 50%-80%, 30%-50%, and less than 30%) into just two (greater than 50% or 50% or less) “reduced the system’s ability to inform and predict mortality from the short term up to 10 years” (Lancet Respir Med. 2018 Jan;6[3]:165-6).
Lung function remains the best available biomarker for life expectancy in both patients with COPD and the general population,” wrote Dr. Soriano, a respiratory medicine researcher based in Madrid, Spain.
Additional important outcomes
Dr. Ouellette noted that while mortality is an important outcome for COPD patients, it’s not the only outcome of interest. “In addition to [trying to] help people live longer, which is certainly a desirable goal, we also want to make people be able to be more functional during their life, have fewer hospitalizations, and have less of a need of other types of supportive medical care for worsening of their disease,” he said. “The fact that the current guidelines don’t improve mortality more than the previous ones may not be a negative thing. It may tell us that the previous guidelines already did a pretty good job of helping us to improve mortality.”
Dr. Ouellette was quick to add that none of inhaled drugs currently available to treat COPD have been conclusively shown to improve mortality. “The only things we know that improve mortality for COPD patients are quitting smoking and using oxygen if a patient meets predefined goals for oxygen,” he said. “So the fact that GOLD criteria doesn’t improve mortality shouldn’t make us think that it’s not a useful tool. We already know that the medicines may not help people live longer.”
Dr. Han pointed out that spirometry “is still used to further clarify the choice of therapy recommended based on the nature and degree of airflow obstruction in light of severity of patient symptoms. The data are still designed to be used in conjunction to personalize therapy for patients.”
She added that the GOLD Science Committee “welcomes additional data analyses so that future recommendations can be further refined.”
Dr. Han disclosed that she has consulted for Boehringer Ingelheim, AstraZeneca, and GlaxoSmithKline. She has also received in-kind research support from Novartis and Sunovion.
Dr. Iftikhar reported having no financial disclosures. Dr. Ouellette is a member of CHEST® Physician’s editorial advisory board. He disclosed being part of a federally funded study being carried out by the Patient-Centered Outcomes Research Institute.
There was no industry involvement in the GOLD 2017 report, but many of its authors and board members had pharmaceutical company ties, and GOLD’s treatment advice relies on data from industry-sponsored studies.
After the Global Initiative for Chronic Obstructive Lung Disease released updated recommendations for grading COPD patients’ level of disease in November of 2016, Imran Iftikhar, MD, tried to incorporate them into his practice, but he encountered problems.
For one thing, the new classification system, which became known as GOLD 2017, uncoupled spirometry results from the ABCD treatment algorithm. “I found it wasn’t really helping me in terms of prognostication or COPD management,” said Dr. Iftikhar, section chief of pulmonary and critical care at Emory Saint Joseph’s Hospital, Atlanta. “Although the purpose of the GOLD classification was not really meant for prognostication, most practicing physicians are frequently asked about prognosis by patients, and I am not sure if the 2017 reclassification really helps with that.”
The GOLD 2017 classification simplified the chronic obstructive pulmonary disease staging that was available from 2011 to 2015 from three variables (spirometry thresholds, exacerbation risk, and dyspnea scale) to two variables (exacerbation risk and dyspnea scale). In the 2017 report, authors of the new guidelines characterized forced expiratory volume in 1 second (FEV1) as “a poor predictor of disease status” and proposed that clinicians derive ABCD groups exclusively from patient symptoms and their exacerbations. FEV1 is an “important parameter at the population level” in predicting hospitalization and mortality, the authors wrote, but keeping results separate “acknowledges the limitations of FEV1 in making treatment decisions for individualized patient care and highlights the importance of patient symptoms and exacerbation risks in guiding therapies in COPD.”
According to Meilan Han, MD, MS, a member of the GOLD Science Committee, since release of the 2017 guidelines, “clinicians have indicated that they like the flexibility the system provides in separating spirometry, symptoms, and exacerbation risk as this more accurately reflects the heterogeneity we see in the COPD patient population.” Nevertheless, how this approach influences long-term outcomes remains unclear.
Daniel Ouellette, MD, FCCP, a pulmonologist with the Henry Ford Health System in Detroit, described the GOLD 2017 criteria as “a good step forward” but said he wasn’t sure if the optimal or perfect tool exists for categorizing COPD patients’ level of disease.
“I think what we see is an effort to use all of these criteria to help us better treat our patients. I think ,” he said in an interview.
“All guidelines need to be modified as further research becomes available. I think that the frontiers of this area are going to be to incorporate new elements such as tobacco history, more emphasis on clinical signs and symptoms, and use of markers other than spirometry, such as eosinophil count, to categorize patients with COPD,” Dr. Ouellette added.
In an analysis of the GOLD 2017 criteria applied to 819 COPD patients in Spain and the United States, published online Nov. 3, 2017, in the American Journal of Respiratory and Critical Care Medicine, Carlos Cabrera López, MD, and his colleagues concluded that the mortality risk was better predicted by the 2015 GOLD classification system than by the 2017 iteration (Am J Respir Crit Care Med. 2018 Feb. doi: 10.101164/rccm.201707-1363OC).
The distribution of Charlson index scores also changed. Whereas group D was higher than B in 2015, they became similar in the 2017 system. For her part, Dr. Han emphasized that the primary goal of the GOLD ABCD classification system is to categorize patients with respect to treatment groups. “Current therapy targets symptoms and exacerbations, which are the key current elements of the classification schema,” she said in an interview. “The results of the Cabrera Lopez analysis are not necessarily unexpected, as FEV1 is associated with mortality.”
In a prospective, multicenter analysis, Portuguese researchers compared the performance of GOLD 2011 and 2017 in terms of how 200 COPD patients were reclassified, the level of agreement between the two iterations, and the performance of each to predict future exacerbations (COPD. 2018 Feb;15[1]; 21-6). They found that about half of patients classified as GOLD D under the 2011 guidelines became classified as GOLD B when the 2017 version was used, and the extent of agreement between the two iterations was moderate (P less than .001). They also found that the two versions of the guidelines were equivalently effective at predicting exacerbations (69.7% vs. 67.6% in the 2011 and 2017 iterations, respectively). In addition, patients who met the criteria for a GOLD B grouping in the 2017 iteration exacerbated 17% more often and had a lower percent predicted post bronchodilator FEV1 than did those who met the criteria for a GOLD B classification under the 2011 guidelines.
Dr. Han, who is also an associate professor of medicine at the University of Michigan Hospital, acknowledged that GOLD 2017 has resulted in the reclassification of some previously group D patients as group B patients. “Our primary goal is to aid clinicians with the diagnosis and management of patients with COPD,” she said. “We look forward to additional data coming in from ongoing clinical trials that will provide longer term data to further refine treatment algorithms.”
In a recent study of more than 33,000 Danish patients older than age 30 with COPD, researchers led by Anne Gedebjerg, MD, found that the GOLD 2017 ABCD classification did not predict all-cause and respiratory mortality more accurately than previous GOLD iterations from 2007 and 2011. Area under the curve for all-cause mortality was 0.61 for GOLD 2007, 0.61 for GOLD 2011, and 0.63 for GOLD 2017, while the area under the curve for respiratory mortality was 0.64 for GOLD 2007, 0.63 for GOLD 2011, and 0.65 for GOLD 2017 (Lancet Respir Med. 2018 Jan;6[3]:204-12).
However, when the spirometric stages 1-4 were combined with the A to D groupings based on symptoms and exacerbations, the 2017 classification predicted mortality with greater accuracy, compared with previous iterations (P less than .0001). “My practice is very much like this paper,” Dr. Iftikhar said. “I use both the spirometric grade and the ABCD grouping to specify which ‘group’ and ‘grade’ my patient belongs to. I think future investigators need to combine ABCD with spirometry classification to see how we can improve the classification system.”
In a commentary published in the same issue of the Lancet Respiratory Medicine as the large Danish study, Joan B. Soriano, MD, PhD, wrote that the 2011 GOLD guideline’s collapse of four spirometric thresholds (greater than 80%, 50%-80%, 30%-50%, and less than 30%) into just two (greater than 50% or 50% or less) “reduced the system’s ability to inform and predict mortality from the short term up to 10 years” (Lancet Respir Med. 2018 Jan;6[3]:165-6).
Lung function remains the best available biomarker for life expectancy in both patients with COPD and the general population,” wrote Dr. Soriano, a respiratory medicine researcher based in Madrid, Spain.
Additional important outcomes
Dr. Ouellette noted that while mortality is an important outcome for COPD patients, it’s not the only outcome of interest. “In addition to [trying to] help people live longer, which is certainly a desirable goal, we also want to make people be able to be more functional during their life, have fewer hospitalizations, and have less of a need of other types of supportive medical care for worsening of their disease,” he said. “The fact that the current guidelines don’t improve mortality more than the previous ones may not be a negative thing. It may tell us that the previous guidelines already did a pretty good job of helping us to improve mortality.”
Dr. Ouellette was quick to add that none of inhaled drugs currently available to treat COPD have been conclusively shown to improve mortality. “The only things we know that improve mortality for COPD patients are quitting smoking and using oxygen if a patient meets predefined goals for oxygen,” he said. “So the fact that GOLD criteria doesn’t improve mortality shouldn’t make us think that it’s not a useful tool. We already know that the medicines may not help people live longer.”
Dr. Han pointed out that spirometry “is still used to further clarify the choice of therapy recommended based on the nature and degree of airflow obstruction in light of severity of patient symptoms. The data are still designed to be used in conjunction to personalize therapy for patients.”
She added that the GOLD Science Committee “welcomes additional data analyses so that future recommendations can be further refined.”
Dr. Han disclosed that she has consulted for Boehringer Ingelheim, AstraZeneca, and GlaxoSmithKline. She has also received in-kind research support from Novartis and Sunovion.
Dr. Iftikhar reported having no financial disclosures. Dr. Ouellette is a member of CHEST® Physician’s editorial advisory board. He disclosed being part of a federally funded study being carried out by the Patient-Centered Outcomes Research Institute.
There was no industry involvement in the GOLD 2017 report, but many of its authors and board members had pharmaceutical company ties, and GOLD’s treatment advice relies on data from industry-sponsored studies.
Outpatient talc administration improves malignant effusion outcomes
Patients with malignant pleural effusion treated with an indwelling pleural catheter have an improved chance of a positive outcome when talc administration is part of their procedure, suggest the results of a randomized, placebo-controlled study.
Malignant pleural effusion, which is usually caused by the spread of metastatic cancer, is typically treated by inducement of pleurodesis. Talc is probably the most effective agent for achieving this result, but there are drawbacks to using talc to induce pleurodesis. Patients who receive this treatment often need to stay in the hospital for 4-7 days, according to Rahul Bhatnagar, PhD, and the coauthors of a study published in the New England Journal of Medicine). Indwelling pleural catheters provide an “ambulatory alternative” for fluid management, they noted. In a noncomparative series of 22 patients, administering talc through such a catheter produced high rates of pleurodesis, they added.
In the new study, Dr. Bhatnagar of the Academic Respiratory Unit, University of Bristol, England, and his coauthors evaluated the use of an indwelling catheter, with or without talc, in patients with malignant pleural effusion recruited at 18 centers in the United Kingdom over 4 years.
“Our primary-outcome results, which were backed up by robust sensitivity analyses, strongly suggest that the administration of talc through an indwelling pleural catheter was significantly more efficacious than the use of an indwelling pleural catheter alone among patients without substantial lung entrapment,” the authors wrote.
A total of 154 patients underwent randomization to the talc or placebo group, and 139 had sufficient data to evaluate the primary outcome of successful pleurodesis at 35 days after randomization. The researchers excluded patients with evidence of lung entrapment, or nonexpandable lung, according to the study report.
In the talc group, pleurodesis was successful at day 35 in 30 of 69 patients (43%) versus 16 of 70 patients (23%) in the placebo group (P = .008).
At day 70, the success rate was 51% for the talc group vs. 27% for the placebo group, respectively.
The rate of pleurodesis was significantly higher when talc was administered through an indwelling pleural catheter, Dr. Bhatnagar and his colleagues noted.
“Success rates at day 70 suggested that pleurodesis was maintained to a point that is clinically relevant for patients with short median survival,” they added.
No excess of side effects or catheter blockages were associated with talc vs. placebo administration through a catheter. Additionally, no differences were seen between the talc and placebo groups in the number of adverse events, number of inpatient days, mortality, or other outcomes tracked by the researchers.
Dr. Bhatnagar reported he had no disclosures related to the study. Study coauthors reported disclosures related to Becton Dickinson – CareFusion, Rosetrees Trust, GE Medical, and Rocket Medical. Becton Dickinson supported the trial with an unrestricted research grant and supplied catheters and drainage bottles for the study’s participants.
SOURCE: Bhatnagar R et al. N Engl J Med. 2018;378:1313-22.
Patients with malignant pleural effusion treated with an indwelling pleural catheter have an improved chance of a positive outcome when talc administration is part of their procedure, suggest the results of a randomized, placebo-controlled study.
Malignant pleural effusion, which is usually caused by the spread of metastatic cancer, is typically treated by inducement of pleurodesis. Talc is probably the most effective agent for achieving this result, but there are drawbacks to using talc to induce pleurodesis. Patients who receive this treatment often need to stay in the hospital for 4-7 days, according to Rahul Bhatnagar, PhD, and the coauthors of a study published in the New England Journal of Medicine). Indwelling pleural catheters provide an “ambulatory alternative” for fluid management, they noted. In a noncomparative series of 22 patients, administering talc through such a catheter produced high rates of pleurodesis, they added.
In the new study, Dr. Bhatnagar of the Academic Respiratory Unit, University of Bristol, England, and his coauthors evaluated the use of an indwelling catheter, with or without talc, in patients with malignant pleural effusion recruited at 18 centers in the United Kingdom over 4 years.
“Our primary-outcome results, which were backed up by robust sensitivity analyses, strongly suggest that the administration of talc through an indwelling pleural catheter was significantly more efficacious than the use of an indwelling pleural catheter alone among patients without substantial lung entrapment,” the authors wrote.
A total of 154 patients underwent randomization to the talc or placebo group, and 139 had sufficient data to evaluate the primary outcome of successful pleurodesis at 35 days after randomization. The researchers excluded patients with evidence of lung entrapment, or nonexpandable lung, according to the study report.
In the talc group, pleurodesis was successful at day 35 in 30 of 69 patients (43%) versus 16 of 70 patients (23%) in the placebo group (P = .008).
At day 70, the success rate was 51% for the talc group vs. 27% for the placebo group, respectively.
The rate of pleurodesis was significantly higher when talc was administered through an indwelling pleural catheter, Dr. Bhatnagar and his colleagues noted.
“Success rates at day 70 suggested that pleurodesis was maintained to a point that is clinically relevant for patients with short median survival,” they added.
No excess of side effects or catheter blockages were associated with talc vs. placebo administration through a catheter. Additionally, no differences were seen between the talc and placebo groups in the number of adverse events, number of inpatient days, mortality, or other outcomes tracked by the researchers.
Dr. Bhatnagar reported he had no disclosures related to the study. Study coauthors reported disclosures related to Becton Dickinson – CareFusion, Rosetrees Trust, GE Medical, and Rocket Medical. Becton Dickinson supported the trial with an unrestricted research grant and supplied catheters and drainage bottles for the study’s participants.
SOURCE: Bhatnagar R et al. N Engl J Med. 2018;378:1313-22.
Patients with malignant pleural effusion treated with an indwelling pleural catheter have an improved chance of a positive outcome when talc administration is part of their procedure, suggest the results of a randomized, placebo-controlled study.
Malignant pleural effusion, which is usually caused by the spread of metastatic cancer, is typically treated by inducement of pleurodesis. Talc is probably the most effective agent for achieving this result, but there are drawbacks to using talc to induce pleurodesis. Patients who receive this treatment often need to stay in the hospital for 4-7 days, according to Rahul Bhatnagar, PhD, and the coauthors of a study published in the New England Journal of Medicine). Indwelling pleural catheters provide an “ambulatory alternative” for fluid management, they noted. In a noncomparative series of 22 patients, administering talc through such a catheter produced high rates of pleurodesis, they added.
In the new study, Dr. Bhatnagar of the Academic Respiratory Unit, University of Bristol, England, and his coauthors evaluated the use of an indwelling catheter, with or without talc, in patients with malignant pleural effusion recruited at 18 centers in the United Kingdom over 4 years.
“Our primary-outcome results, which were backed up by robust sensitivity analyses, strongly suggest that the administration of talc through an indwelling pleural catheter was significantly more efficacious than the use of an indwelling pleural catheter alone among patients without substantial lung entrapment,” the authors wrote.
A total of 154 patients underwent randomization to the talc or placebo group, and 139 had sufficient data to evaluate the primary outcome of successful pleurodesis at 35 days after randomization. The researchers excluded patients with evidence of lung entrapment, or nonexpandable lung, according to the study report.
In the talc group, pleurodesis was successful at day 35 in 30 of 69 patients (43%) versus 16 of 70 patients (23%) in the placebo group (P = .008).
At day 70, the success rate was 51% for the talc group vs. 27% for the placebo group, respectively.
The rate of pleurodesis was significantly higher when talc was administered through an indwelling pleural catheter, Dr. Bhatnagar and his colleagues noted.
“Success rates at day 70 suggested that pleurodesis was maintained to a point that is clinically relevant for patients with short median survival,” they added.
No excess of side effects or catheter blockages were associated with talc vs. placebo administration through a catheter. Additionally, no differences were seen between the talc and placebo groups in the number of adverse events, number of inpatient days, mortality, or other outcomes tracked by the researchers.
Dr. Bhatnagar reported he had no disclosures related to the study. Study coauthors reported disclosures related to Becton Dickinson – CareFusion, Rosetrees Trust, GE Medical, and Rocket Medical. Becton Dickinson supported the trial with an unrestricted research grant and supplied catheters and drainage bottles for the study’s participants.
SOURCE: Bhatnagar R et al. N Engl J Med. 2018;378:1313-22.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: with no deleterious effects.
Major finding: At 35 days post randomization, pleurodesis was successful in 30 of 69 patients (43%) in the talc group versus 16 of 70 patients (23%) in the placebo group (P = .008).
Study details: A randomized, placebo-controlled, single-blind, parallel-group trial including 154 patients with malignant pleural effusion recruited at 18 U.K. centers over a period of 4 years.
Disclosures: Becton Dickinson supported the trial with an unrestricted research grant and supplied catheters and drainage bottles for participants. Study authors reported disclosures related to Becton Dickinson – CareFusion, Rosetrees Trust, GE Medical, and Rocket Medical.
Source: Bhatnagar R et al. N Engl J Med. 2018;378:1313-22.
Evaluating fever in the first 90 days of life
Fever in the youngest of infants creates a challenge for the pediatric clinician. Fever is a common presentation for serious bacterial infection (SBI) although most fevers are due to viral infection. However, the clinical presentation does not necessarily differ, and the risk for a poor outcome in this age group is substantial.
In the early stages of my pediatric career, most febrile infants less than 90 days of age were evaluated for sepsis, admitted, and treated with antibiotics pending culture results. Group B streptococcal sepsis or Escherichia coli sepsis were common in the first month of life, and Haemophilus influenza type B or Streptococcus pneumoniae in the second and third months of life. The approach to fever in the first 90 days has changed following both the introduction of haemophilus and pneumococcal conjugate vaccines, the experience with risk stratification criteria for identifying infants at low risk for SBI, and the recognition of urinary tract infection (UTI) as a common source of infection in this age group as well as development of criteria for diagnosis.
A further nuance was subsequently added with the introduction of rapid diagnostics for viral infection. Byington et al. found that the majority of febrile infants less than 90 days of age had viral infection with enterovirus, respiratory syncytial virus (RSV), influenza or rotavirus.1 Using the Rochester risk stratification and the presence or absence of viral infection, she demonstrated that the risk of SBI was reduced in both high- and low-risk infants in the presence of viral infection; in low risk infants with viral infection, SBI was identified in 1.8%, compared with 3.1% in those without viral infection, and in high-risk infants. 5.5% has SBI when viral infection was found, compared to 16.7% in the absence of viral infection. She also proposed risk features to identify those infected with herpes simplex virus; age less than 42 days, vesicular rash, elevated alanine transaminase (ALT) and aspartate aminotransferase (AST), CSF pleocytosis, and seizure or twitching.
Greenhow et al. reported on the experience with “serious” bacterial infection in infants less than 90 days of age receiving care at Northern California Kaiser Permanente during the period 2005-2011.2 As pictured, the majority of children have UTI, and smaller numbers have bacteremia or meningitis. A small group of children with UTI have urosepsis as well; those with urosepsis can be differentiated from those with only UTI by age (less than 21 days), clinical exam (ill appearing), and elevated C reactive protein (greater than 20 mg/L) or elevated procalcitonin (greater than 0.5 ng/mL).3 Further evaluation of procalcitonin by other groups appears to validate its role in identifying children at low risk of SBI (procalcitonin less than 0.3 ng/mL).4
Currently, studies of febrile infants less than 90 days of age demonstrate that E. coli dominates in bacteremia, UTI, and meningitis, with Group B streptococcus as the next most frequent pathogen identified.2 Increasingly ampicillin resistance has been reported among E. coli isolates from both early- and late-onset disease as well as rare isolates that are resistant to third generation cephalosporins or gentamicin. Surveillance to identify changes in antimicrobial susceptibility will need to be ongoing to ensure that current approaches for initial therapy in high-risk infants aligns with current susceptibility patterns.
Dr. Pelton is chief of the section of pediatric infectious diseases and coordinator of the maternal-child HIV program at Boston Medical Center. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Pediatrics. 2004 Jun;113(6):1662-6.
2. Pediatr Infect Dis J. 2014 Jun;33(6):595-9.
3. Pediatr Infect Dis J. 2015 Jan;34(1):17-21.
4. JAMA Pediatr. 2016;170(1):17-18.
5. “AAP Proposes Update to Evaluating, Managing Febrile Infants Guideline,” The Hospitalist, 2016.
Fever in the youngest of infants creates a challenge for the pediatric clinician. Fever is a common presentation for serious bacterial infection (SBI) although most fevers are due to viral infection. However, the clinical presentation does not necessarily differ, and the risk for a poor outcome in this age group is substantial.
In the early stages of my pediatric career, most febrile infants less than 90 days of age were evaluated for sepsis, admitted, and treated with antibiotics pending culture results. Group B streptococcal sepsis or Escherichia coli sepsis were common in the first month of life, and Haemophilus influenza type B or Streptococcus pneumoniae in the second and third months of life. The approach to fever in the first 90 days has changed following both the introduction of haemophilus and pneumococcal conjugate vaccines, the experience with risk stratification criteria for identifying infants at low risk for SBI, and the recognition of urinary tract infection (UTI) as a common source of infection in this age group as well as development of criteria for diagnosis.
A further nuance was subsequently added with the introduction of rapid diagnostics for viral infection. Byington et al. found that the majority of febrile infants less than 90 days of age had viral infection with enterovirus, respiratory syncytial virus (RSV), influenza or rotavirus.1 Using the Rochester risk stratification and the presence or absence of viral infection, she demonstrated that the risk of SBI was reduced in both high- and low-risk infants in the presence of viral infection; in low risk infants with viral infection, SBI was identified in 1.8%, compared with 3.1% in those without viral infection, and in high-risk infants. 5.5% has SBI when viral infection was found, compared to 16.7% in the absence of viral infection. She also proposed risk features to identify those infected with herpes simplex virus; age less than 42 days, vesicular rash, elevated alanine transaminase (ALT) and aspartate aminotransferase (AST), CSF pleocytosis, and seizure or twitching.
Greenhow et al. reported on the experience with “serious” bacterial infection in infants less than 90 days of age receiving care at Northern California Kaiser Permanente during the period 2005-2011.2 As pictured, the majority of children have UTI, and smaller numbers have bacteremia or meningitis. A small group of children with UTI have urosepsis as well; those with urosepsis can be differentiated from those with only UTI by age (less than 21 days), clinical exam (ill appearing), and elevated C reactive protein (greater than 20 mg/L) or elevated procalcitonin (greater than 0.5 ng/mL).3 Further evaluation of procalcitonin by other groups appears to validate its role in identifying children at low risk of SBI (procalcitonin less than 0.3 ng/mL).4
Currently, studies of febrile infants less than 90 days of age demonstrate that E. coli dominates in bacteremia, UTI, and meningitis, with Group B streptococcus as the next most frequent pathogen identified.2 Increasingly ampicillin resistance has been reported among E. coli isolates from both early- and late-onset disease as well as rare isolates that are resistant to third generation cephalosporins or gentamicin. Surveillance to identify changes in antimicrobial susceptibility will need to be ongoing to ensure that current approaches for initial therapy in high-risk infants aligns with current susceptibility patterns.
Dr. Pelton is chief of the section of pediatric infectious diseases and coordinator of the maternal-child HIV program at Boston Medical Center. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Pediatrics. 2004 Jun;113(6):1662-6.
2. Pediatr Infect Dis J. 2014 Jun;33(6):595-9.
3. Pediatr Infect Dis J. 2015 Jan;34(1):17-21.
4. JAMA Pediatr. 2016;170(1):17-18.
5. “AAP Proposes Update to Evaluating, Managing Febrile Infants Guideline,” The Hospitalist, 2016.
Fever in the youngest of infants creates a challenge for the pediatric clinician. Fever is a common presentation for serious bacterial infection (SBI) although most fevers are due to viral infection. However, the clinical presentation does not necessarily differ, and the risk for a poor outcome in this age group is substantial.
In the early stages of my pediatric career, most febrile infants less than 90 days of age were evaluated for sepsis, admitted, and treated with antibiotics pending culture results. Group B streptococcal sepsis or Escherichia coli sepsis were common in the first month of life, and Haemophilus influenza type B or Streptococcus pneumoniae in the second and third months of life. The approach to fever in the first 90 days has changed following both the introduction of haemophilus and pneumococcal conjugate vaccines, the experience with risk stratification criteria for identifying infants at low risk for SBI, and the recognition of urinary tract infection (UTI) as a common source of infection in this age group as well as development of criteria for diagnosis.
A further nuance was subsequently added with the introduction of rapid diagnostics for viral infection. Byington et al. found that the majority of febrile infants less than 90 days of age had viral infection with enterovirus, respiratory syncytial virus (RSV), influenza or rotavirus.1 Using the Rochester risk stratification and the presence or absence of viral infection, she demonstrated that the risk of SBI was reduced in both high- and low-risk infants in the presence of viral infection; in low risk infants with viral infection, SBI was identified in 1.8%, compared with 3.1% in those without viral infection, and in high-risk infants. 5.5% has SBI when viral infection was found, compared to 16.7% in the absence of viral infection. She also proposed risk features to identify those infected with herpes simplex virus; age less than 42 days, vesicular rash, elevated alanine transaminase (ALT) and aspartate aminotransferase (AST), CSF pleocytosis, and seizure or twitching.
Greenhow et al. reported on the experience with “serious” bacterial infection in infants less than 90 days of age receiving care at Northern California Kaiser Permanente during the period 2005-2011.2 As pictured, the majority of children have UTI, and smaller numbers have bacteremia or meningitis. A small group of children with UTI have urosepsis as well; those with urosepsis can be differentiated from those with only UTI by age (less than 21 days), clinical exam (ill appearing), and elevated C reactive protein (greater than 20 mg/L) or elevated procalcitonin (greater than 0.5 ng/mL).3 Further evaluation of procalcitonin by other groups appears to validate its role in identifying children at low risk of SBI (procalcitonin less than 0.3 ng/mL).4
Currently, studies of febrile infants less than 90 days of age demonstrate that E. coli dominates in bacteremia, UTI, and meningitis, with Group B streptococcus as the next most frequent pathogen identified.2 Increasingly ampicillin resistance has been reported among E. coli isolates from both early- and late-onset disease as well as rare isolates that are resistant to third generation cephalosporins or gentamicin. Surveillance to identify changes in antimicrobial susceptibility will need to be ongoing to ensure that current approaches for initial therapy in high-risk infants aligns with current susceptibility patterns.
Dr. Pelton is chief of the section of pediatric infectious diseases and coordinator of the maternal-child HIV program at Boston Medical Center. He said he had no relevant financial disclosures. Email him at [email protected].
References
1. Pediatrics. 2004 Jun;113(6):1662-6.
2. Pediatr Infect Dis J. 2014 Jun;33(6):595-9.
3. Pediatr Infect Dis J. 2015 Jan;34(1):17-21.
4. JAMA Pediatr. 2016;170(1):17-18.
5. “AAP Proposes Update to Evaluating, Managing Febrile Infants Guideline,” The Hospitalist, 2016.
Few acutely ill hospitalized patients receive VTE prophylaxis
SAN DIEGO – Among patients hospitalized for acute medical illnesses, the risk of venous thromboembolism (VTE) remained elevated 30-40 days after discharge, results from a large analysis of national data showed.
Moreover, only 7% of at-risk patients received VTE prophylaxis in both the inpatient and outpatient setting.
“The results of this real-world study imply that there is a significantly unmet medical need for effective VTE prophylaxis in both the inpatient and outpatient continuum of care among patients hospitalized for acute medical illnesses,” researchers led by Alpesh Amin, MD, wrote in a poster presented at the biennial summit of the Thrombosis & Hemostasis Societies of North America.
According to Dr. Amin, who chairs the department of medicine at the University of California, Irvine, hospitalized patients with acute medical illnesses face an increased risk for VTE during hospital discharge, mainly within 40 days following hospital admission. However, the treatment patterns of VTE prophylaxis in this patient population have not been well studied in the “real-world” setting. In an effort to improve this area of clinical practice, the researchers used the Marketscan database between Jan. 1, 2012, and June 30, 2015, to identify acutely ill hospitalized patients, such as those with heart failure, respiratory diseases, ischemic stroke, cancer, infectious diseases, and rheumatic diseases. The key outcomes of interest were the proportion of patients receiving inpatient and outpatient VTE prophylaxis and the proportion of patients with VTE events during and after the index hospitalization. They used Kaplan-Meier analysis to examine the risk for VTE events after the index inpatient admission.
The mean age of the 17,895 patients was 58 years, 55% were female, and most (77%) were from the Southern area of the United States. Their mean Charlson Comborbidity Index score prior to hospitalization was 2.2. Nearly all hospitals (87%) were urban based, nonteaching (95%), and large, with 68% having at least 300 beds. Nearly three-quarters of patients (72%) were hospitalized for infectious and respiratory diseases, and the mean length of stay was 5 days.
Dr. Amin and his associates found that 59% of hospitalized patients did not receive any VTE prophylaxis, while only 7% received prophylaxis in both the inpatient and outpatient continuum of care. At the same time, cumulative VTE rates within 40 days of index admission were highest among patients hospitalized for infectious diseases and cancer (3.4% each), followed by those with heart failure (3.1%), respiratory diseases (2%), ischemic stroke (1.5%), and rheumatic diseases (1.3%). The cumulative VTE event rate for the overall study population within 40 days from index hospitalization was nearly 3%, with 60% of VTE events having occurred within 40 days.
The study was funded by Portola Pharmaceuticals. Dr. Amin reported having no financial disclosures.
SOURCE: Amin A et al. THSNA 2018, Poster 51.
SAN DIEGO – Among patients hospitalized for acute medical illnesses, the risk of venous thromboembolism (VTE) remained elevated 30-40 days after discharge, results from a large analysis of national data showed.
Moreover, only 7% of at-risk patients received VTE prophylaxis in both the inpatient and outpatient setting.
“The results of this real-world study imply that there is a significantly unmet medical need for effective VTE prophylaxis in both the inpatient and outpatient continuum of care among patients hospitalized for acute medical illnesses,” researchers led by Alpesh Amin, MD, wrote in a poster presented at the biennial summit of the Thrombosis & Hemostasis Societies of North America.
According to Dr. Amin, who chairs the department of medicine at the University of California, Irvine, hospitalized patients with acute medical illnesses face an increased risk for VTE during hospital discharge, mainly within 40 days following hospital admission. However, the treatment patterns of VTE prophylaxis in this patient population have not been well studied in the “real-world” setting. In an effort to improve this area of clinical practice, the researchers used the Marketscan database between Jan. 1, 2012, and June 30, 2015, to identify acutely ill hospitalized patients, such as those with heart failure, respiratory diseases, ischemic stroke, cancer, infectious diseases, and rheumatic diseases. The key outcomes of interest were the proportion of patients receiving inpatient and outpatient VTE prophylaxis and the proportion of patients with VTE events during and after the index hospitalization. They used Kaplan-Meier analysis to examine the risk for VTE events after the index inpatient admission.
The mean age of the 17,895 patients was 58 years, 55% were female, and most (77%) were from the Southern area of the United States. Their mean Charlson Comborbidity Index score prior to hospitalization was 2.2. Nearly all hospitals (87%) were urban based, nonteaching (95%), and large, with 68% having at least 300 beds. Nearly three-quarters of patients (72%) were hospitalized for infectious and respiratory diseases, and the mean length of stay was 5 days.
Dr. Amin and his associates found that 59% of hospitalized patients did not receive any VTE prophylaxis, while only 7% received prophylaxis in both the inpatient and outpatient continuum of care. At the same time, cumulative VTE rates within 40 days of index admission were highest among patients hospitalized for infectious diseases and cancer (3.4% each), followed by those with heart failure (3.1%), respiratory diseases (2%), ischemic stroke (1.5%), and rheumatic diseases (1.3%). The cumulative VTE event rate for the overall study population within 40 days from index hospitalization was nearly 3%, with 60% of VTE events having occurred within 40 days.
The study was funded by Portola Pharmaceuticals. Dr. Amin reported having no financial disclosures.
SOURCE: Amin A et al. THSNA 2018, Poster 51.
SAN DIEGO – Among patients hospitalized for acute medical illnesses, the risk of venous thromboembolism (VTE) remained elevated 30-40 days after discharge, results from a large analysis of national data showed.
Moreover, only 7% of at-risk patients received VTE prophylaxis in both the inpatient and outpatient setting.
“The results of this real-world study imply that there is a significantly unmet medical need for effective VTE prophylaxis in both the inpatient and outpatient continuum of care among patients hospitalized for acute medical illnesses,” researchers led by Alpesh Amin, MD, wrote in a poster presented at the biennial summit of the Thrombosis & Hemostasis Societies of North America.
According to Dr. Amin, who chairs the department of medicine at the University of California, Irvine, hospitalized patients with acute medical illnesses face an increased risk for VTE during hospital discharge, mainly within 40 days following hospital admission. However, the treatment patterns of VTE prophylaxis in this patient population have not been well studied in the “real-world” setting. In an effort to improve this area of clinical practice, the researchers used the Marketscan database between Jan. 1, 2012, and June 30, 2015, to identify acutely ill hospitalized patients, such as those with heart failure, respiratory diseases, ischemic stroke, cancer, infectious diseases, and rheumatic diseases. The key outcomes of interest were the proportion of patients receiving inpatient and outpatient VTE prophylaxis and the proportion of patients with VTE events during and after the index hospitalization. They used Kaplan-Meier analysis to examine the risk for VTE events after the index inpatient admission.
The mean age of the 17,895 patients was 58 years, 55% were female, and most (77%) were from the Southern area of the United States. Their mean Charlson Comborbidity Index score prior to hospitalization was 2.2. Nearly all hospitals (87%) were urban based, nonteaching (95%), and large, with 68% having at least 300 beds. Nearly three-quarters of patients (72%) were hospitalized for infectious and respiratory diseases, and the mean length of stay was 5 days.
Dr. Amin and his associates found that 59% of hospitalized patients did not receive any VTE prophylaxis, while only 7% received prophylaxis in both the inpatient and outpatient continuum of care. At the same time, cumulative VTE rates within 40 days of index admission were highest among patients hospitalized for infectious diseases and cancer (3.4% each), followed by those with heart failure (3.1%), respiratory diseases (2%), ischemic stroke (1.5%), and rheumatic diseases (1.3%). The cumulative VTE event rate for the overall study population within 40 days from index hospitalization was nearly 3%, with 60% of VTE events having occurred within 40 days.
The study was funded by Portola Pharmaceuticals. Dr. Amin reported having no financial disclosures.
SOURCE: Amin A et al. THSNA 2018, Poster 51.
REPORTING FROM THSNA 2018
Key clinical point: There is a significant unmet medical need for VTE prophylaxis in the continuum of care of patients hospitalized for acute medical illnesses.
Major finding: Of the overall study population, only 7% received both inpatient and outpatient VTE prophylaxis.
Study details: An analysis of national data from 17,895 acutely ill hospitalized patients.
Disclosures: The study was funded by Portola Pharmaceuticals. The presenter reported having no financial conflicts.
Source: Amin A et al. THSNA 2018, Poster 51.
Waning vaccine immunity linked to pertussis resurgence
researchers said.
In the March 28, 2018, edition of Science Translational Medicine, researchers reported on a study that used different models of transmission to explore what might be the cause of the steady increase in pertussis infections since the mid-1970s.
Using 16 years’ worth of detailed, age-stratified incidence data from Massachusetts, researchers found that the model which assumed a gradual waning in protection was the best fit for the observed patterns of pertussis incidence across the population.
This model suggested significant variability in how the level of protection changes over time, with a 10% risk of vaccine protection waning to zero within 10 years of completing routine vaccination and a 55% chance that the vaccine would confer lifelong protection.
“Crucially, we find that the vaccine is effective at reducing pathogen circulation but not so effective that eradication of this highly contagious bacterium should be possible without targeted booster campaigns,” wrote Dr. Matthieu Domenech de Cellès, PhD, of the Institut Pasteur at the University of Versailles (France) and his coauthors.
The model also considered the possibility that the whole-cell and acellular pertussis vaccines might show differences in immunity, which had been suggested as one explanation for the resurgence of the disease. However, the authors found little evidence of a marked epidemiological switch from the whole-cell to acellular vaccines, although their results did suggest the acellular vaccine has a moderately reduced efficacy.
“Our results suggest that the train of events leading to the resurgence of pertussis was set in motion well before the shift to the DTaP vaccine,” Dr. Domenech de Cellès and his associates said.
The model also pointed to big shifts in the age-specific immunological profile caused by introduction of vaccination, which led to a reduction in transmission and also a reduction in natural infections both in vaccinated and unvaccinated individuals.
This meant individuals who either did not get vaccinated as children or who did not gain immunity from vaccination were growing to adulthood without ever being exposed to natural infection.
“Concurrently, older cohorts, with their long-lived immunity derived from natural infections experienced during the prevaccine period, were gradually dying out,” the authors said. “The resulting rise in the number of susceptible adults sets the stage for the pertussis resurgence, especially among adults.”
Two authors were supported by the National Institutes of Health and by Models of Infectious Disease Agent Study–National Institute of General Medical Sciences. No conflicts of interest were declared.
SOURCE: Domenech de Cellès M et al. Sci Transl Med. 2018 Mar 28;10:eaaj1748.
researchers said.
In the March 28, 2018, edition of Science Translational Medicine, researchers reported on a study that used different models of transmission to explore what might be the cause of the steady increase in pertussis infections since the mid-1970s.
Using 16 years’ worth of detailed, age-stratified incidence data from Massachusetts, researchers found that the model which assumed a gradual waning in protection was the best fit for the observed patterns of pertussis incidence across the population.
This model suggested significant variability in how the level of protection changes over time, with a 10% risk of vaccine protection waning to zero within 10 years of completing routine vaccination and a 55% chance that the vaccine would confer lifelong protection.
“Crucially, we find that the vaccine is effective at reducing pathogen circulation but not so effective that eradication of this highly contagious bacterium should be possible without targeted booster campaigns,” wrote Dr. Matthieu Domenech de Cellès, PhD, of the Institut Pasteur at the University of Versailles (France) and his coauthors.
The model also considered the possibility that the whole-cell and acellular pertussis vaccines might show differences in immunity, which had been suggested as one explanation for the resurgence of the disease. However, the authors found little evidence of a marked epidemiological switch from the whole-cell to acellular vaccines, although their results did suggest the acellular vaccine has a moderately reduced efficacy.
“Our results suggest that the train of events leading to the resurgence of pertussis was set in motion well before the shift to the DTaP vaccine,” Dr. Domenech de Cellès and his associates said.
The model also pointed to big shifts in the age-specific immunological profile caused by introduction of vaccination, which led to a reduction in transmission and also a reduction in natural infections both in vaccinated and unvaccinated individuals.
This meant individuals who either did not get vaccinated as children or who did not gain immunity from vaccination were growing to adulthood without ever being exposed to natural infection.
“Concurrently, older cohorts, with their long-lived immunity derived from natural infections experienced during the prevaccine period, were gradually dying out,” the authors said. “The resulting rise in the number of susceptible adults sets the stage for the pertussis resurgence, especially among adults.”
Two authors were supported by the National Institutes of Health and by Models of Infectious Disease Agent Study–National Institute of General Medical Sciences. No conflicts of interest were declared.
SOURCE: Domenech de Cellès M et al. Sci Transl Med. 2018 Mar 28;10:eaaj1748.
researchers said.
In the March 28, 2018, edition of Science Translational Medicine, researchers reported on a study that used different models of transmission to explore what might be the cause of the steady increase in pertussis infections since the mid-1970s.
Using 16 years’ worth of detailed, age-stratified incidence data from Massachusetts, researchers found that the model which assumed a gradual waning in protection was the best fit for the observed patterns of pertussis incidence across the population.
This model suggested significant variability in how the level of protection changes over time, with a 10% risk of vaccine protection waning to zero within 10 years of completing routine vaccination and a 55% chance that the vaccine would confer lifelong protection.
“Crucially, we find that the vaccine is effective at reducing pathogen circulation but not so effective that eradication of this highly contagious bacterium should be possible without targeted booster campaigns,” wrote Dr. Matthieu Domenech de Cellès, PhD, of the Institut Pasteur at the University of Versailles (France) and his coauthors.
The model also considered the possibility that the whole-cell and acellular pertussis vaccines might show differences in immunity, which had been suggested as one explanation for the resurgence of the disease. However, the authors found little evidence of a marked epidemiological switch from the whole-cell to acellular vaccines, although their results did suggest the acellular vaccine has a moderately reduced efficacy.
“Our results suggest that the train of events leading to the resurgence of pertussis was set in motion well before the shift to the DTaP vaccine,” Dr. Domenech de Cellès and his associates said.
The model also pointed to big shifts in the age-specific immunological profile caused by introduction of vaccination, which led to a reduction in transmission and also a reduction in natural infections both in vaccinated and unvaccinated individuals.
This meant individuals who either did not get vaccinated as children or who did not gain immunity from vaccination were growing to adulthood without ever being exposed to natural infection.
“Concurrently, older cohorts, with their long-lived immunity derived from natural infections experienced during the prevaccine period, were gradually dying out,” the authors said. “The resulting rise in the number of susceptible adults sets the stage for the pertussis resurgence, especially among adults.”
Two authors were supported by the National Institutes of Health and by Models of Infectious Disease Agent Study–National Institute of General Medical Sciences. No conflicts of interest were declared.
SOURCE: Domenech de Cellès M et al. Sci Transl Med. 2018 Mar 28;10:eaaj1748.
FROM SCIENCE TRANSLATIONAL MEDICINE
TB in 2017: Good news and bad news
according to the Centers for Disease Control and Prevention.
Those new lows – TB incidence of 2.8 per 100,000 persons and 9,093 new cases – continue a downward trend that started in 1993, but the current rate of decline is much lower than the threshold needed to eliminate TB by the year 2100, Rebekah J. Stewart and her associates at the CDC’s Division of Tuberculosis Elimination, Atlanta, wrote in the Morbidity and Mortality Weekly Report.
Geographically, at least, the states with populations at the highest risk are Hawaii, which had a TB incidence of 8.1 per 100,000 persons in 2017, and Alaska, with an incidence of 7.0 per 100,000. California and the District of Columbia were next, each with an incidence of 5.2. The states with the lowest rates were Montana and Wyoming at 0.3 per 100,000, the investigators reported, based on data from the National Tuberculosis Surveillance System as of Feb. 12, 2018.
Groups most affected by TB include persons housed in congregate settings – homeless shelters, long-term care facilities, and correctional facilities – and those from countries that have high TB prevalence. Overall incidence for non–U.S. born residents was 14.6 per 100,000 in 2017, compared with 1.0 for the native born, with large discrepancies seen between U.S. and non–U.S. born blacks (2.8 vs. 22.0), native Hawaiian/Pacific Islanders (6.5 vs. 21.0), and Asians (2.0 vs. 27.0), Ms. Stewart and her associates said.
“Increased support of global TB elimination efforts would help to reduce global … prevalence, thereby indirectly reducing the incidence of reactivation TB in the United States among non–U.S. born persons from higher-prevalence countries,” they wrote.
The issue of global action on TB was addressed by the Forum of International Respiratory Societies in a statement recognizing World TB Day (March 24). “TB is the world’s most common infectious disease killer, yet is identifiable, treatable and preventable; what is missing is the political will to dedicate the resources necessary to eradicate it, once and for all,” said Dean E. Schraufnagel, MD, the organization’s executive director.
SOURCE: Stewart RJ et al. MMWR 2018 Mar 23;67(11):317-23.
according to the Centers for Disease Control and Prevention.
Those new lows – TB incidence of 2.8 per 100,000 persons and 9,093 new cases – continue a downward trend that started in 1993, but the current rate of decline is much lower than the threshold needed to eliminate TB by the year 2100, Rebekah J. Stewart and her associates at the CDC’s Division of Tuberculosis Elimination, Atlanta, wrote in the Morbidity and Mortality Weekly Report.
Geographically, at least, the states with populations at the highest risk are Hawaii, which had a TB incidence of 8.1 per 100,000 persons in 2017, and Alaska, with an incidence of 7.0 per 100,000. California and the District of Columbia were next, each with an incidence of 5.2. The states with the lowest rates were Montana and Wyoming at 0.3 per 100,000, the investigators reported, based on data from the National Tuberculosis Surveillance System as of Feb. 12, 2018.
Groups most affected by TB include persons housed in congregate settings – homeless shelters, long-term care facilities, and correctional facilities – and those from countries that have high TB prevalence. Overall incidence for non–U.S. born residents was 14.6 per 100,000 in 2017, compared with 1.0 for the native born, with large discrepancies seen between U.S. and non–U.S. born blacks (2.8 vs. 22.0), native Hawaiian/Pacific Islanders (6.5 vs. 21.0), and Asians (2.0 vs. 27.0), Ms. Stewart and her associates said.
“Increased support of global TB elimination efforts would help to reduce global … prevalence, thereby indirectly reducing the incidence of reactivation TB in the United States among non–U.S. born persons from higher-prevalence countries,” they wrote.
The issue of global action on TB was addressed by the Forum of International Respiratory Societies in a statement recognizing World TB Day (March 24). “TB is the world’s most common infectious disease killer, yet is identifiable, treatable and preventable; what is missing is the political will to dedicate the resources necessary to eradicate it, once and for all,” said Dean E. Schraufnagel, MD, the organization’s executive director.
SOURCE: Stewart RJ et al. MMWR 2018 Mar 23;67(11):317-23.
according to the Centers for Disease Control and Prevention.
Those new lows – TB incidence of 2.8 per 100,000 persons and 9,093 new cases – continue a downward trend that started in 1993, but the current rate of decline is much lower than the threshold needed to eliminate TB by the year 2100, Rebekah J. Stewart and her associates at the CDC’s Division of Tuberculosis Elimination, Atlanta, wrote in the Morbidity and Mortality Weekly Report.
Geographically, at least, the states with populations at the highest risk are Hawaii, which had a TB incidence of 8.1 per 100,000 persons in 2017, and Alaska, with an incidence of 7.0 per 100,000. California and the District of Columbia were next, each with an incidence of 5.2. The states with the lowest rates were Montana and Wyoming at 0.3 per 100,000, the investigators reported, based on data from the National Tuberculosis Surveillance System as of Feb. 12, 2018.
Groups most affected by TB include persons housed in congregate settings – homeless shelters, long-term care facilities, and correctional facilities – and those from countries that have high TB prevalence. Overall incidence for non–U.S. born residents was 14.6 per 100,000 in 2017, compared with 1.0 for the native born, with large discrepancies seen between U.S. and non–U.S. born blacks (2.8 vs. 22.0), native Hawaiian/Pacific Islanders (6.5 vs. 21.0), and Asians (2.0 vs. 27.0), Ms. Stewart and her associates said.
“Increased support of global TB elimination efforts would help to reduce global … prevalence, thereby indirectly reducing the incidence of reactivation TB in the United States among non–U.S. born persons from higher-prevalence countries,” they wrote.
The issue of global action on TB was addressed by the Forum of International Respiratory Societies in a statement recognizing World TB Day (March 24). “TB is the world’s most common infectious disease killer, yet is identifiable, treatable and preventable; what is missing is the political will to dedicate the resources necessary to eradicate it, once and for all,” said Dean E. Schraufnagel, MD, the organization’s executive director.
SOURCE: Stewart RJ et al. MMWR 2018 Mar 23;67(11):317-23.
FROM MMWR
RSV immunoprophylaxis in premature infants doesn’t prevent later asthma
reported Nienke M. Scheltema, MD, of Wilhelmina Children’s Hospital, Utrecht, the Netherlands, and associates.
In a study of 395 otherwise healthy premature infants who were randomized to receive palivizumab for respiratory syncytial virus (RSV) immunoprophylaxis or placebo and followed for 6 years, 14% of the 199 infants in the RSV prevention group had parent-reported asthma, compared with 24% of the 196 in the placebo group (absolute risk reduction, 9.9%). This was explained mostly by differences in infrequent wheeze, the researchers said. However, physician-diagnosed asthma in the past 12 months was not significantly different between the two groups at 6 years: 10.3% in the RSV prevention group and 9.9% in the placebo group.
SOURCE: Scheltema NM et al. Lancet. 2018 Feb 27. doi: 10.1016/S2213-2600(18)30055-9.
reported Nienke M. Scheltema, MD, of Wilhelmina Children’s Hospital, Utrecht, the Netherlands, and associates.
In a study of 395 otherwise healthy premature infants who were randomized to receive palivizumab for respiratory syncytial virus (RSV) immunoprophylaxis or placebo and followed for 6 years, 14% of the 199 infants in the RSV prevention group had parent-reported asthma, compared with 24% of the 196 in the placebo group (absolute risk reduction, 9.9%). This was explained mostly by differences in infrequent wheeze, the researchers said. However, physician-diagnosed asthma in the past 12 months was not significantly different between the two groups at 6 years: 10.3% in the RSV prevention group and 9.9% in the placebo group.
SOURCE: Scheltema NM et al. Lancet. 2018 Feb 27. doi: 10.1016/S2213-2600(18)30055-9.
reported Nienke M. Scheltema, MD, of Wilhelmina Children’s Hospital, Utrecht, the Netherlands, and associates.
In a study of 395 otherwise healthy premature infants who were randomized to receive palivizumab for respiratory syncytial virus (RSV) immunoprophylaxis or placebo and followed for 6 years, 14% of the 199 infants in the RSV prevention group had parent-reported asthma, compared with 24% of the 196 in the placebo group (absolute risk reduction, 9.9%). This was explained mostly by differences in infrequent wheeze, the researchers said. However, physician-diagnosed asthma in the past 12 months was not significantly different between the two groups at 6 years: 10.3% in the RSV prevention group and 9.9% in the placebo group.
SOURCE: Scheltema NM et al. Lancet. 2018 Feb 27. doi: 10.1016/S2213-2600(18)30055-9.
FROM THE LANCET