User login
News and Views that Matter to Pediatricians
The leading independent newspaper covering news and commentary in pediatrics.
Lucid abductions and Candy Crush addiction
I dream of alien abductions
There he goes! It’s lunchtime and your colleague Tom is going on and on again about that time he was abducted by aliens. It sounds ridiculous, but he does make some convincing arguments. Tom thinks it was real, but could it have all just been in his head?
Lucid dreaming may help explain alleged alien abductions. During a lucid dream, people know that they’re dreaming, and can also have some control over how the dreams play out. During some dream states, a person can feel intense sensations, such as terror and paralysis, so it’s no wonder these dreams feel so real.
In a recent study, scientists encouraged 152 participants who had self-identified as lucid dreamers to dream about aliens. Many (75%) of the participants were able to dream about alien encounters, and 15% “achieved relatively realistic experiences,” the investigators reported.
So cut Tom some slack. He’s not crazy, he might just have lucid dreaming privileges. Tell him he should dream about something more fun, like a vacation in the Bahamas.
Follow your heart: Drink more coffee
It seems like the world is divided into coffee drinkers and non–coffee drinkers. Then there’s decaf and regular drinkers. Whichever camp you fall into, know this: The widespread belief that caffeine consumption has an effect on your heart is all beans.
In what is the largest investigation of its kind, researchers from the University of California, San Francisco, looked into whether drinking caffeinated coffee was linked to a risk for heart arrhythmia. They also researched whether patients with genetic variants that affect their metabolism could change that association. Almost 400,000 people with a mean age of 56 years participated in the study. More than half of the participants were women.
The investigators analyzed the participants’ self-reported coffee consumption using a technique called Mendelian randomization to leverage genetic data with the participants’ relationship with caffeine, making it an even field and not relying on the participant consumption self-reporting for outcomes as in previous studies.
What they found, after the 4-year follow up, was nothing short of myth busting.
“We found no evidence that caffeine consumption leads to a greater risk of arrhythmias,” said senior and corresponding author Gregory Marcus, MD. “Our population-based study provides reassurance that common prohibitions against caffeine to reduce arrhythmia risk are likely unwarranted.”
There was no evidence of a heightened risk of arrhythmias in participants who were genetically predisposed to metabolize caffeine differently from those who were not. And, there was a 3% reduction of arrhythmias in patients who consumed higher amounts of coffee.
We are not lobbying for Big Caffeine, but this study adds to the reported health benefits linked to coffee, which already include reduced risk for cancer, diabetes, and Parkinson’s disease, with an added bonus of anti-inflammatory benefits. So, the next time you’re hesitant to pour that second cup of Joe, just go for it. Your heart can take it.
Bored? Feeling down? Don’t play Candy Crush
Now hang on, aren’t those the perfect times to play video games? If there’s nothing else to do, why not open Candy Crush and mindlessly power through the levels?
Because, according to a study by a group of Canadian researchers, it’s actually the worst thing you can do. Well, maybe not literally, but it’s not helpful. Researchers recruited 60 Candy Crush players who were at various levels in the game. They had the participants play early levels that were far too easy or levels balanced with their gameplay abilities.
Players in the easy-level group got bored and quit far earlier than did those in the advanced-level group. The group playing to their abilities were able to access a “flow” state and focus all their attention on the game. While this is all well and good for their gaming performance, according to the researchers, it confirms the theory that playing to escape boredom or negative emotions is more likely to lead to addiction. As with all addictions, the temporary high can give way to a self-repeating loop, causing patients to ignore real life and deepen depression.
The researchers hope their findings will encourage game developers to “consider implementing responsible video gaming tools directly within their games.” Comedy gold. Perhaps Canadians’ idea of capitalism is a little different from that of those south of the border.
Hiccups and vaccine refusal
Tonight, LOTME News dives into the fetid cesspool that is international politics and comes out with … hiccups?
But first, a word from our sponsor, Fearless Boxing Club of South Etobicoke, Ontario.
Are you looking to flout public health restrictions? Do you want to spend time in an enclosed space with other people who haven’t gotten the COVID-19 vaccine? Do you “feel safer waiting until more research is done on the side effects being discovered right now”? (We are not making this up.)
Then join the Fearless Boxing Club, because we “will not be accepting any vaccinated members.” Our founders, Mohammed Abedeen and Krystal Glazier-Roscoe, are working hard to exclude “those who received the experimental COVID vaccine.” (Still not making it up.)
And now, back to the news.
Brazilian president Jair Bolsonaro was hospitalized recently for a severe case of hiccups that may have been related to a stab wound he received in 2018. [Nope, didn’t make that up, either.]
Mr. Bolsonaro had been hiccuping for 10 days, and was experiencing abdominal pain and difficulty speaking, when he entered the hospital on July 14. Since being stabbed while on the campaign trail, he has undergone several operations, which may have led to the partial intestinal obstruction that caused his latest symptoms.
His medical team advised Mr. Bolsonaro to go on a diet to aid his recovery, but when he was released on July 18 he said, “I hope in 10 days I’ll be eating barbecued ribs.” (Maybe this is all just a lucid dream. Probably shouldn’t have had ribs right before bed.)
I dream of alien abductions
There he goes! It’s lunchtime and your colleague Tom is going on and on again about that time he was abducted by aliens. It sounds ridiculous, but he does make some convincing arguments. Tom thinks it was real, but could it have all just been in his head?
Lucid dreaming may help explain alleged alien abductions. During a lucid dream, people know that they’re dreaming, and can also have some control over how the dreams play out. During some dream states, a person can feel intense sensations, such as terror and paralysis, so it’s no wonder these dreams feel so real.
In a recent study, scientists encouraged 152 participants who had self-identified as lucid dreamers to dream about aliens. Many (75%) of the participants were able to dream about alien encounters, and 15% “achieved relatively realistic experiences,” the investigators reported.
So cut Tom some slack. He’s not crazy, he might just have lucid dreaming privileges. Tell him he should dream about something more fun, like a vacation in the Bahamas.
Follow your heart: Drink more coffee
It seems like the world is divided into coffee drinkers and non–coffee drinkers. Then there’s decaf and regular drinkers. Whichever camp you fall into, know this: The widespread belief that caffeine consumption has an effect on your heart is all beans.
In what is the largest investigation of its kind, researchers from the University of California, San Francisco, looked into whether drinking caffeinated coffee was linked to a risk for heart arrhythmia. They also researched whether patients with genetic variants that affect their metabolism could change that association. Almost 400,000 people with a mean age of 56 years participated in the study. More than half of the participants were women.
The investigators analyzed the participants’ self-reported coffee consumption using a technique called Mendelian randomization to leverage genetic data with the participants’ relationship with caffeine, making it an even field and not relying on the participant consumption self-reporting for outcomes as in previous studies.
What they found, after the 4-year follow up, was nothing short of myth busting.
“We found no evidence that caffeine consumption leads to a greater risk of arrhythmias,” said senior and corresponding author Gregory Marcus, MD. “Our population-based study provides reassurance that common prohibitions against caffeine to reduce arrhythmia risk are likely unwarranted.”
There was no evidence of a heightened risk of arrhythmias in participants who were genetically predisposed to metabolize caffeine differently from those who were not. And, there was a 3% reduction of arrhythmias in patients who consumed higher amounts of coffee.
We are not lobbying for Big Caffeine, but this study adds to the reported health benefits linked to coffee, which already include reduced risk for cancer, diabetes, and Parkinson’s disease, with an added bonus of anti-inflammatory benefits. So, the next time you’re hesitant to pour that second cup of Joe, just go for it. Your heart can take it.
Bored? Feeling down? Don’t play Candy Crush
Now hang on, aren’t those the perfect times to play video games? If there’s nothing else to do, why not open Candy Crush and mindlessly power through the levels?
Because, according to a study by a group of Canadian researchers, it’s actually the worst thing you can do. Well, maybe not literally, but it’s not helpful. Researchers recruited 60 Candy Crush players who were at various levels in the game. They had the participants play early levels that were far too easy or levels balanced with their gameplay abilities.
Players in the easy-level group got bored and quit far earlier than did those in the advanced-level group. The group playing to their abilities were able to access a “flow” state and focus all their attention on the game. While this is all well and good for their gaming performance, according to the researchers, it confirms the theory that playing to escape boredom or negative emotions is more likely to lead to addiction. As with all addictions, the temporary high can give way to a self-repeating loop, causing patients to ignore real life and deepen depression.
The researchers hope their findings will encourage game developers to “consider implementing responsible video gaming tools directly within their games.” Comedy gold. Perhaps Canadians’ idea of capitalism is a little different from that of those south of the border.
Hiccups and vaccine refusal
Tonight, LOTME News dives into the fetid cesspool that is international politics and comes out with … hiccups?
But first, a word from our sponsor, Fearless Boxing Club of South Etobicoke, Ontario.
Are you looking to flout public health restrictions? Do you want to spend time in an enclosed space with other people who haven’t gotten the COVID-19 vaccine? Do you “feel safer waiting until more research is done on the side effects being discovered right now”? (We are not making this up.)
Then join the Fearless Boxing Club, because we “will not be accepting any vaccinated members.” Our founders, Mohammed Abedeen and Krystal Glazier-Roscoe, are working hard to exclude “those who received the experimental COVID vaccine.” (Still not making it up.)
And now, back to the news.
Brazilian president Jair Bolsonaro was hospitalized recently for a severe case of hiccups that may have been related to a stab wound he received in 2018. [Nope, didn’t make that up, either.]
Mr. Bolsonaro had been hiccuping for 10 days, and was experiencing abdominal pain and difficulty speaking, when he entered the hospital on July 14. Since being stabbed while on the campaign trail, he has undergone several operations, which may have led to the partial intestinal obstruction that caused his latest symptoms.
His medical team advised Mr. Bolsonaro to go on a diet to aid his recovery, but when he was released on July 18 he said, “I hope in 10 days I’ll be eating barbecued ribs.” (Maybe this is all just a lucid dream. Probably shouldn’t have had ribs right before bed.)
I dream of alien abductions
There he goes! It’s lunchtime and your colleague Tom is going on and on again about that time he was abducted by aliens. It sounds ridiculous, but he does make some convincing arguments. Tom thinks it was real, but could it have all just been in his head?
Lucid dreaming may help explain alleged alien abductions. During a lucid dream, people know that they’re dreaming, and can also have some control over how the dreams play out. During some dream states, a person can feel intense sensations, such as terror and paralysis, so it’s no wonder these dreams feel so real.
In a recent study, scientists encouraged 152 participants who had self-identified as lucid dreamers to dream about aliens. Many (75%) of the participants were able to dream about alien encounters, and 15% “achieved relatively realistic experiences,” the investigators reported.
So cut Tom some slack. He’s not crazy, he might just have lucid dreaming privileges. Tell him he should dream about something more fun, like a vacation in the Bahamas.
Follow your heart: Drink more coffee
It seems like the world is divided into coffee drinkers and non–coffee drinkers. Then there’s decaf and regular drinkers. Whichever camp you fall into, know this: The widespread belief that caffeine consumption has an effect on your heart is all beans.
In what is the largest investigation of its kind, researchers from the University of California, San Francisco, looked into whether drinking caffeinated coffee was linked to a risk for heart arrhythmia. They also researched whether patients with genetic variants that affect their metabolism could change that association. Almost 400,000 people with a mean age of 56 years participated in the study. More than half of the participants were women.
The investigators analyzed the participants’ self-reported coffee consumption using a technique called Mendelian randomization to leverage genetic data with the participants’ relationship with caffeine, making it an even field and not relying on the participant consumption self-reporting for outcomes as in previous studies.
What they found, after the 4-year follow up, was nothing short of myth busting.
“We found no evidence that caffeine consumption leads to a greater risk of arrhythmias,” said senior and corresponding author Gregory Marcus, MD. “Our population-based study provides reassurance that common prohibitions against caffeine to reduce arrhythmia risk are likely unwarranted.”
There was no evidence of a heightened risk of arrhythmias in participants who were genetically predisposed to metabolize caffeine differently from those who were not. And, there was a 3% reduction of arrhythmias in patients who consumed higher amounts of coffee.
We are not lobbying for Big Caffeine, but this study adds to the reported health benefits linked to coffee, which already include reduced risk for cancer, diabetes, and Parkinson’s disease, with an added bonus of anti-inflammatory benefits. So, the next time you’re hesitant to pour that second cup of Joe, just go for it. Your heart can take it.
Bored? Feeling down? Don’t play Candy Crush
Now hang on, aren’t those the perfect times to play video games? If there’s nothing else to do, why not open Candy Crush and mindlessly power through the levels?
Because, according to a study by a group of Canadian researchers, it’s actually the worst thing you can do. Well, maybe not literally, but it’s not helpful. Researchers recruited 60 Candy Crush players who were at various levels in the game. They had the participants play early levels that were far too easy or levels balanced with their gameplay abilities.
Players in the easy-level group got bored and quit far earlier than did those in the advanced-level group. The group playing to their abilities were able to access a “flow” state and focus all their attention on the game. While this is all well and good for their gaming performance, according to the researchers, it confirms the theory that playing to escape boredom or negative emotions is more likely to lead to addiction. As with all addictions, the temporary high can give way to a self-repeating loop, causing patients to ignore real life and deepen depression.
The researchers hope their findings will encourage game developers to “consider implementing responsible video gaming tools directly within their games.” Comedy gold. Perhaps Canadians’ idea of capitalism is a little different from that of those south of the border.
Hiccups and vaccine refusal
Tonight, LOTME News dives into the fetid cesspool that is international politics and comes out with … hiccups?
But first, a word from our sponsor, Fearless Boxing Club of South Etobicoke, Ontario.
Are you looking to flout public health restrictions? Do you want to spend time in an enclosed space with other people who haven’t gotten the COVID-19 vaccine? Do you “feel safer waiting until more research is done on the side effects being discovered right now”? (We are not making this up.)
Then join the Fearless Boxing Club, because we “will not be accepting any vaccinated members.” Our founders, Mohammed Abedeen and Krystal Glazier-Roscoe, are working hard to exclude “those who received the experimental COVID vaccine.” (Still not making it up.)
And now, back to the news.
Brazilian president Jair Bolsonaro was hospitalized recently for a severe case of hiccups that may have been related to a stab wound he received in 2018. [Nope, didn’t make that up, either.]
Mr. Bolsonaro had been hiccuping for 10 days, and was experiencing abdominal pain and difficulty speaking, when he entered the hospital on July 14. Since being stabbed while on the campaign trail, he has undergone several operations, which may have led to the partial intestinal obstruction that caused his latest symptoms.
His medical team advised Mr. Bolsonaro to go on a diet to aid his recovery, but when he was released on July 18 he said, “I hope in 10 days I’ll be eating barbecued ribs.” (Maybe this is all just a lucid dream. Probably shouldn’t have had ribs right before bed.)
Sickle cell disease, trait may up risk for poor COVID outcomes
Sickle cell disease (SCD) was associated with a greater than fourfold excess risk for COVID-19–related hospitalization and a greater than twofold risk for COVID-19–related death, according to a big-data analysis from the United Kingdom.
SCD was associated with an adjusted hazard ratio (HR) of 4.11 (95% confidence interval, 2.98-5.66) for admission to hospital and an HR of 2.55 (95% CI, 1.36-4.75) for death, report Ashley K. Clift, MBBS, a clinical research fellow at the University of Oxford, and colleagues. The results were published online July 20 in Annals of Internal Medicine.
Even those who carry just one copy of the sickle cell gene – the carrier status for sickle cell disease – appeared to be at heightened risk for these outcomes (HR for hospitalization, 1.38; 95% CI, 1.12-1.70; HR for death, 1.51; 95% CI, 1.13-2.00).
“Given the well-known ethnic patterning of sickle cell disorders, the predisposition they pose to other infections, and early evidence from smaller registries, we thought this would be an important analysis to run at the population level,” Dr. Clift said in an interview.
in terms of vaccination strategies and advice on nonpharmacological interventions,” he said.
“The best course of action for managing risk in this group is vaccination,” said Enrico M. Novelli, MD, director of the adult sickle cell program at the University of Pittsburgh Medical Center. Dr. Novelli, who is also section chief of benign hematology in the university’s School of Medicine, was not involved in the study. “To date, there are no specific studies of the effect of COVID-19 vaccination in patients with SCD, but there is no reason to believe it would be less effective or more risky in this patient population,” he said.
In addition, common-sense measures, such as masking and physical distancing, particularly at large, indoor gatherings, should be encouraged, Dr. Novelli added. Keeping SCD under good control with available treatments is also important. “Any patient with SCD who contracts COVID-19 should undergo close, outpatient monitoring with pulse oxygen measurements. If sick, they should be hospitalized in a center familiar with the care of SCD patients.”
The U.K. results are in line with and expand on earlier evidence from specialist centers and registries, but the association with sickle cell trait has been unclear and is notable in these findings, Dr. Clift said.
“The finding of the association with sickle cell trait is somewhat unexpected,” pediatric hematologist/oncologist Rabi Hanna, MD, director of pediatric bone marrow transplantation at Cleveland Clinic Children’s, told this news organization. “But I would question the accuracy of the numbers, since not all people with the trait realize they have it. In other respects, the study confirms earlier hypotheses and data from single-center studies.” Dr. Hanna did not participate in the U.K. study.
Study details
The SCD cohort consisted of 5,059 persons with SCD and 25,682 carriers, those with just one copy of the trait. Data were drawn from the United Kingdom’s large primary-care QResearch database. Follow-up for hospitalizations was conducted from Jan. 24, 2020 to Sept. 30, 2020; follow-up for deaths was conducted from Jan. 24, 2020 to Jan. 18, 2021. Among adults with SCD, there were 40 hospitalizations and 10 deaths. Among those with sickle cell trait, there were 98 hospitalizations and 50 deaths. No children died, and only a few (<5) required hospitalization.
Previous registry research showed similarly elevated risks for severe disease and fatality among patients with SCD who were infected with SARS-CoV-2.
Because SCD affects 8 to 12 million people globally – 100,000 in the United States – the authors say their results are important for policymakers and for prioritizing vaccination. They also note that trait carriers may be underdiagnosed.
“While SCD is part of newborn screening, there may be undiagnosed older people with the trait in the general population, but it’s difficult to quantify how much this is undiagnosed,” Dr. Clift said. “But now we have these results, it’s not that surprising that sickle cell trait is also associated with increased risk, albeit to a lower extent. This could suggest an almost dose-like effect of the sickle mutations on COVID hospitalization risk.”
Neonatal screening for the most common form of SCD is currently mandatory in the United States, but the Centers for Disease Control and Prevention has no clear data on how many people are aware they are carriers, Dr. Hanna said. “The states didn’t all begin screening at the same time – some started in the 1990s, others started in the 2000s – so many young adults may be unaware they have the trait,” he said.
Dr. Clift said the multiorgan complications of SCD, such as cardiac and immune problems, may be contributing to the heightened risk in individuals infected with SARS-CoV-2. “For example, we know that people with sickle cell disease are more susceptible to other viral infections. There is also some pathophysiological overlap between SCD disease and severe COVID, such as clotting dysfunction, so that may be worth further exploration,” he said.
The overlapping clotting problems associated with both COVID-19 and SCD could increase the risk for severe venous thromboembolism. In addition, experts noted that patients with SCD often have pre-COVID endothelial damage and baseline inflammation and are very sensitive to hypoxia; as well, a sizable proportion have lung disease.
The message to patients and physicians counseling patients is twofold, said Dr. Hanna: “SCD patients are at higher risk of COVID complications, and these are preventable with vaccination.”
The study was supported by the UK Medical Research Council. Dr. Clift is supported by Cancer Research UK. Coauthor Dr. Hippisley-Cox has received fees from ClinRisk and nonfinancial support from QResearch outside of the submitted work. Dr. Hanna has disclosed no relevant financial relationships. Dr. Novelli is a consultant for Novartis.
A version of this article first appeared on Medscape.com.
Sickle cell disease (SCD) was associated with a greater than fourfold excess risk for COVID-19–related hospitalization and a greater than twofold risk for COVID-19–related death, according to a big-data analysis from the United Kingdom.
SCD was associated with an adjusted hazard ratio (HR) of 4.11 (95% confidence interval, 2.98-5.66) for admission to hospital and an HR of 2.55 (95% CI, 1.36-4.75) for death, report Ashley K. Clift, MBBS, a clinical research fellow at the University of Oxford, and colleagues. The results were published online July 20 in Annals of Internal Medicine.
Even those who carry just one copy of the sickle cell gene – the carrier status for sickle cell disease – appeared to be at heightened risk for these outcomes (HR for hospitalization, 1.38; 95% CI, 1.12-1.70; HR for death, 1.51; 95% CI, 1.13-2.00).
“Given the well-known ethnic patterning of sickle cell disorders, the predisposition they pose to other infections, and early evidence from smaller registries, we thought this would be an important analysis to run at the population level,” Dr. Clift said in an interview.
in terms of vaccination strategies and advice on nonpharmacological interventions,” he said.
“The best course of action for managing risk in this group is vaccination,” said Enrico M. Novelli, MD, director of the adult sickle cell program at the University of Pittsburgh Medical Center. Dr. Novelli, who is also section chief of benign hematology in the university’s School of Medicine, was not involved in the study. “To date, there are no specific studies of the effect of COVID-19 vaccination in patients with SCD, but there is no reason to believe it would be less effective or more risky in this patient population,” he said.
In addition, common-sense measures, such as masking and physical distancing, particularly at large, indoor gatherings, should be encouraged, Dr. Novelli added. Keeping SCD under good control with available treatments is also important. “Any patient with SCD who contracts COVID-19 should undergo close, outpatient monitoring with pulse oxygen measurements. If sick, they should be hospitalized in a center familiar with the care of SCD patients.”
The U.K. results are in line with and expand on earlier evidence from specialist centers and registries, but the association with sickle cell trait has been unclear and is notable in these findings, Dr. Clift said.
“The finding of the association with sickle cell trait is somewhat unexpected,” pediatric hematologist/oncologist Rabi Hanna, MD, director of pediatric bone marrow transplantation at Cleveland Clinic Children’s, told this news organization. “But I would question the accuracy of the numbers, since not all people with the trait realize they have it. In other respects, the study confirms earlier hypotheses and data from single-center studies.” Dr. Hanna did not participate in the U.K. study.
Study details
The SCD cohort consisted of 5,059 persons with SCD and 25,682 carriers, those with just one copy of the trait. Data were drawn from the United Kingdom’s large primary-care QResearch database. Follow-up for hospitalizations was conducted from Jan. 24, 2020 to Sept. 30, 2020; follow-up for deaths was conducted from Jan. 24, 2020 to Jan. 18, 2021. Among adults with SCD, there were 40 hospitalizations and 10 deaths. Among those with sickle cell trait, there were 98 hospitalizations and 50 deaths. No children died, and only a few (<5) required hospitalization.
Previous registry research showed similarly elevated risks for severe disease and fatality among patients with SCD who were infected with SARS-CoV-2.
Because SCD affects 8 to 12 million people globally – 100,000 in the United States – the authors say their results are important for policymakers and for prioritizing vaccination. They also note that trait carriers may be underdiagnosed.
“While SCD is part of newborn screening, there may be undiagnosed older people with the trait in the general population, but it’s difficult to quantify how much this is undiagnosed,” Dr. Clift said. “But now we have these results, it’s not that surprising that sickle cell trait is also associated with increased risk, albeit to a lower extent. This could suggest an almost dose-like effect of the sickle mutations on COVID hospitalization risk.”
Neonatal screening for the most common form of SCD is currently mandatory in the United States, but the Centers for Disease Control and Prevention has no clear data on how many people are aware they are carriers, Dr. Hanna said. “The states didn’t all begin screening at the same time – some started in the 1990s, others started in the 2000s – so many young adults may be unaware they have the trait,” he said.
Dr. Clift said the multiorgan complications of SCD, such as cardiac and immune problems, may be contributing to the heightened risk in individuals infected with SARS-CoV-2. “For example, we know that people with sickle cell disease are more susceptible to other viral infections. There is also some pathophysiological overlap between SCD disease and severe COVID, such as clotting dysfunction, so that may be worth further exploration,” he said.
The overlapping clotting problems associated with both COVID-19 and SCD could increase the risk for severe venous thromboembolism. In addition, experts noted that patients with SCD often have pre-COVID endothelial damage and baseline inflammation and are very sensitive to hypoxia; as well, a sizable proportion have lung disease.
The message to patients and physicians counseling patients is twofold, said Dr. Hanna: “SCD patients are at higher risk of COVID complications, and these are preventable with vaccination.”
The study was supported by the UK Medical Research Council. Dr. Clift is supported by Cancer Research UK. Coauthor Dr. Hippisley-Cox has received fees from ClinRisk and nonfinancial support from QResearch outside of the submitted work. Dr. Hanna has disclosed no relevant financial relationships. Dr. Novelli is a consultant for Novartis.
A version of this article first appeared on Medscape.com.
Sickle cell disease (SCD) was associated with a greater than fourfold excess risk for COVID-19–related hospitalization and a greater than twofold risk for COVID-19–related death, according to a big-data analysis from the United Kingdom.
SCD was associated with an adjusted hazard ratio (HR) of 4.11 (95% confidence interval, 2.98-5.66) for admission to hospital and an HR of 2.55 (95% CI, 1.36-4.75) for death, report Ashley K. Clift, MBBS, a clinical research fellow at the University of Oxford, and colleagues. The results were published online July 20 in Annals of Internal Medicine.
Even those who carry just one copy of the sickle cell gene – the carrier status for sickle cell disease – appeared to be at heightened risk for these outcomes (HR for hospitalization, 1.38; 95% CI, 1.12-1.70; HR for death, 1.51; 95% CI, 1.13-2.00).
“Given the well-known ethnic patterning of sickle cell disorders, the predisposition they pose to other infections, and early evidence from smaller registries, we thought this would be an important analysis to run at the population level,” Dr. Clift said in an interview.
in terms of vaccination strategies and advice on nonpharmacological interventions,” he said.
“The best course of action for managing risk in this group is vaccination,” said Enrico M. Novelli, MD, director of the adult sickle cell program at the University of Pittsburgh Medical Center. Dr. Novelli, who is also section chief of benign hematology in the university’s School of Medicine, was not involved in the study. “To date, there are no specific studies of the effect of COVID-19 vaccination in patients with SCD, but there is no reason to believe it would be less effective or more risky in this patient population,” he said.
In addition, common-sense measures, such as masking and physical distancing, particularly at large, indoor gatherings, should be encouraged, Dr. Novelli added. Keeping SCD under good control with available treatments is also important. “Any patient with SCD who contracts COVID-19 should undergo close, outpatient monitoring with pulse oxygen measurements. If sick, they should be hospitalized in a center familiar with the care of SCD patients.”
The U.K. results are in line with and expand on earlier evidence from specialist centers and registries, but the association with sickle cell trait has been unclear and is notable in these findings, Dr. Clift said.
“The finding of the association with sickle cell trait is somewhat unexpected,” pediatric hematologist/oncologist Rabi Hanna, MD, director of pediatric bone marrow transplantation at Cleveland Clinic Children’s, told this news organization. “But I would question the accuracy of the numbers, since not all people with the trait realize they have it. In other respects, the study confirms earlier hypotheses and data from single-center studies.” Dr. Hanna did not participate in the U.K. study.
Study details
The SCD cohort consisted of 5,059 persons with SCD and 25,682 carriers, those with just one copy of the trait. Data were drawn from the United Kingdom’s large primary-care QResearch database. Follow-up for hospitalizations was conducted from Jan. 24, 2020 to Sept. 30, 2020; follow-up for deaths was conducted from Jan. 24, 2020 to Jan. 18, 2021. Among adults with SCD, there were 40 hospitalizations and 10 deaths. Among those with sickle cell trait, there were 98 hospitalizations and 50 deaths. No children died, and only a few (<5) required hospitalization.
Previous registry research showed similarly elevated risks for severe disease and fatality among patients with SCD who were infected with SARS-CoV-2.
Because SCD affects 8 to 12 million people globally – 100,000 in the United States – the authors say their results are important for policymakers and for prioritizing vaccination. They also note that trait carriers may be underdiagnosed.
“While SCD is part of newborn screening, there may be undiagnosed older people with the trait in the general population, but it’s difficult to quantify how much this is undiagnosed,” Dr. Clift said. “But now we have these results, it’s not that surprising that sickle cell trait is also associated with increased risk, albeit to a lower extent. This could suggest an almost dose-like effect of the sickle mutations on COVID hospitalization risk.”
Neonatal screening for the most common form of SCD is currently mandatory in the United States, but the Centers for Disease Control and Prevention has no clear data on how many people are aware they are carriers, Dr. Hanna said. “The states didn’t all begin screening at the same time – some started in the 1990s, others started in the 2000s – so many young adults may be unaware they have the trait,” he said.
Dr. Clift said the multiorgan complications of SCD, such as cardiac and immune problems, may be contributing to the heightened risk in individuals infected with SARS-CoV-2. “For example, we know that people with sickle cell disease are more susceptible to other viral infections. There is also some pathophysiological overlap between SCD disease and severe COVID, such as clotting dysfunction, so that may be worth further exploration,” he said.
The overlapping clotting problems associated with both COVID-19 and SCD could increase the risk for severe venous thromboembolism. In addition, experts noted that patients with SCD often have pre-COVID endothelial damage and baseline inflammation and are very sensitive to hypoxia; as well, a sizable proportion have lung disease.
The message to patients and physicians counseling patients is twofold, said Dr. Hanna: “SCD patients are at higher risk of COVID complications, and these are preventable with vaccination.”
The study was supported by the UK Medical Research Council. Dr. Clift is supported by Cancer Research UK. Coauthor Dr. Hippisley-Cox has received fees from ClinRisk and nonfinancial support from QResearch outside of the submitted work. Dr. Hanna has disclosed no relevant financial relationships. Dr. Novelli is a consultant for Novartis.
A version of this article first appeared on Medscape.com.
‘Dealing with a different beast’: Why Delta has doctors worried
Catherine O’Neal, MD, an infectious disease physician, took to the podium of the Louisiana governor’s press conference recently and did not mince words.
“The Delta variant is not last year’s virus, and it’s become incredibly apparent to healthcare workers that we are dealing with a different beast,” she said.
Louisiana is one of the least vaccinated states in the country. In the United States as a whole, 48.6% of the population is fully vaccinated. In Louisiana, it’s just 36%, and Delta is bearing down.
Dr. O’Neal spoke about the pressure that rising COVID cases were already putting on her hospital, Our Lady of the Lake Regional Medical Center in Baton Rouge. She talked about watching her peers, 30- and 40-year-olds, become severely ill with the latest iteration of the new coronavirus — the Delta variant — which is sweeping through the United States with astonishing speed, causing new cases, hospitalizations, and deaths to rise again.
Dr. O’Neal talked about parents who might not be alive to see their children go off to college in a few weeks. She talked about increasing hospital admissions for infected kids and pregnant women on ventilators.
“I want to be clear after seeing what we’ve seen the last two weeks. We only have two choices: We are either going to get vaccinated and end the pandemic, or we’re going to accept death and a lot of it,” Dr. O’Neal said, her voice choked by emotion.
Where Delta goes, death follows
Delta was first identified in India, where it caused a devastating surge in the spring. In a population that was largely unvaccinated, researchers think it may have caused as many as three million deaths. In just a few months’ time, it has sped across the globe.
, which was first identified in the United Kingdom).
Where a single infected person might have spread older versions of the virus to two or three others, mathematician and epidemiologist Adam Kucharski, PhD, an associate professor at the London School of Hygiene and Tropical Medicine, thinks that number — called the basic reproduction number — might be around six for Delta, meaning that, on average, each infected person spreads the virus to six others.
“The Delta variant is the most able and fastest and fittest of those viruses,” said Mike Ryan, executive director of the World Health Organization’s Health Emergencies Programme, in a recent press briefing.
Early evidence suggests it may also cause more severe disease in people who are not vaccinated.
“There’s clearly increased risk of ICU admission, hospitalization, and death,” said Ashleigh Tuite, PhD, MPH, an infectious disease epidemiologist at the University of Toronto in Ontario.
In a study published ahead of peer review, Dr. Tuite and her coauthor, David Fisman, MD, MPH, reviewed the health outcomes for more than 200,000 people who tested positive for SARS-CoV-2 in Ontario between February and June of 2021. Starting in February, Ontario began screening all positive COVID tests for mutations in the N501Y region for signs of mutation.
Compared with versions of the coronavirus that circulated in 2020, having an Alpha, Beta, or Gamma variant modestly increased the odds that an infected person would become sicker. The Delta variant raised the risk even higher, more than doubling the odds that an infected person would need to be hospitalized or could die from their infection.
Emerging evidence from England and Scotland, analyzed by Public Health England, also shows an increased risk for hospitalization with Delta. The increases are in line with the Canadian data. Experts caution that the picture may change over time as more evidence is gathered.
“What is causing that? We don’t know,” Dr. Tuite said.
Enhanced virus
The Delta variants (there’s actually more than one in the same viral family) have about 15 different mutations compared with the original virus. Two of these, L452R and E484Q, are mutations to the spike protein that were first flagged as problematic in other variants because they appear to help the virus escape the antibodies we make to fight it.
It has another mutation away from its binding site that’s also getting researchers’ attention — P681R.
This mutation appears to enhance the “springiness” of the parts of the virus that dock onto our cells, said Alexander Greninger, MD, PhD, assistant director of the UW Medicine Clinical Virology Laboratory at the University of Washington in Seattle. So it’s more likely to be in the right position to infect our cells if we come into contact with it.
Another theory is that P681R may also enhance the virus’s ability to fuse cells together into clumps that have several different nuclei. These balls of fused cells are called syncytia.
“So it turns into a big factory for making viruses,” said Kamran Kadkhoda, PhD, medical director of immunopathology at the Cleveland Clinic in Ohio.
This capability is not unique to Delta or even to the new coronavirus. Earlier versions and other viruses can do the same thing, but according to a recent paper in Nature, the syncytia that Delta creates are larger than the ones created by previous variants.
Scientists aren’t sure what these supersized syncytia mean, exactly, but they have some theories. They may help the virus copy itself more quickly, so a person’s viral load builds up quickly. That may enhance the ability of the virus to transmit from person to person.
And at least one recent study from China supports this idea. That study, which was posted ahead of peer review on the website Virological.org, tracked 167 people infected with Delta back to a single index case.
China has used extensive contact tracing to identify people that may have been exposed to the virus and sequester them quickly to tamp down its spread. Once a person is isolated or quarantined, they are tested daily with gold-standard PCR testing to determine whether or not they were infected.
Researchers compared the characteristics of Delta cases with those of people infected in 2020 with previous versions of the virus.
This study found that people infected by Delta tested positive more quickly than their predecessors did. In 2020, it took an average of 6 days for someone to test positive after an exposure. With Delta, it took an average of about 4 days.
When people tested positive, they had more than 1,000 times more virus in their bodies, suggesting that the Delta variant has a higher growth rate in the body.
This gives Delta a big advantage. According to Angie Rasmussen, PhD, a virologist at the Vaccine and Infectious Disease Organization at the University of Saskatchewan in Canada, who posted a thread about the study on Twitter, if people are shedding 1,000 times more virus, it is much more likely that close contacts will be exposed to enough of it to become infected themselves.
And if they’re shedding earlier in the course of their infections, the virus has more opportunity to spread.
This may help explain why Delta is so much more contagious.
Beyond transmission, Delta’s ability to form syncytia may have two other important consequences. It may help the virus hide from our immune system, and it may make the virus more damaging to the body.
Commonly, when a virus infects a cell, it will corrupt the cell’s protein-making machinery to crank out more copies of itself. When the cell dies, these new copies are released into the plasma outside the cell where they can float over and infect new cells. It’s in this extracellular space where a virus can also be attacked by the neutralizing antibodies our immune system makes to fight it off.
“Antibodies don’t penetrate inside the cell. If these viruses are going from one cell to another by just fusing to each other, antibodies become less useful,” Dr. Kadkhoda said.
Escape artist
Recent studies show that Delta is also able to escape antibodies made in response to vaccination more effectively than the Alpha, or B.1.1.7 strain. The effect was more pronounced in older adults, who tend to have weaker responses to vaccines in general.
This evasion of the immune system is particularly problematic for people who are only partially vaccinated. Data from the United Kingdom show that a single dose of vaccine is only about 31% effective at preventing illness with Delta, and 75% effective at preventing hospitalization.
After two doses, the vaccines are still highly effective — even against Delta — reaching 80% protection for illness, and 94% for hospitalization, which is why U.S. officials are begging people to get both doses of their shots, and do it as quickly as possible.
Finally, the virus’s ability to form syncytia may leave greater damage behind in the body’s tissues and organs.
“Especially in the lungs,” Dr. Kadkhoda said. The lungs are very fragile tissues. Their tiny air sacs — the alveoli — are only a single-cell thick. They have to be very thin to exchange oxygen in the blood.
“Any damage like that can severely affect any oxygen exchange and the normal housekeeping activities of that tissue,” he said. “In those vital organs, it may be very problematic.”
The research is still early, but studies in animals and cell lines are backing up what doctors say they are seeing in hospitalized patients.
A recent preprint study from researchers in Japan found that hamsters infected with Delta lost more weight — a proxy for how sick they were — compared with hamsters infected with an older version of the virus. The researchers attribute this to the viruses› ability to fuse cells together to form syncytia.
Another investigation, from researchers in India, infected two groups of hamsters — one with the original “wild type” strain of the virus, the other with the Delta variant of the new coronavirus.
As in the Japanese study, the hamsters infected with Delta lost more weight. When the researchers performed necropsies on the animals, they found more lung damage and bleeding in hamsters infected with Delta. This study was also posted as a preprint ahead of peer review.
German researchers working with pseudotyped versions of the new coronavirus — viruses that have been genetically changed to make them safer to work with — watched what happened after they used these pseudoviruses to infect lung, colon, and kidney cells in the lab.
They, too, found that cells infected with the Delta variant formed more and larger syncytia compared with cells infected with the wild type strain of the virus. The authors write that their findings suggest Delta could “cause more tissue damage, and thus be more pathogenic, than previous variants.”Researchers say it’s important to remember that, while interesting, this research isn’t conclusive. Hamsters and cells aren’t humans. More studies are needed to prove these theories.
Scientists say that what we already know about Delta makes vaccination more important than ever.
“The net effect is really that, you know, this is worrisome in people who are unvaccinated and then people who have breakthrough infections, but it’s not…a reason to panic or to throw up our hands and say you know, this pandemic is never going to end,” Dr. Tuite said, “[b]ecause what we do see is that the vaccines continue to be highly protective.”
A version of this article first appeared on Medscape.com.
Catherine O’Neal, MD, an infectious disease physician, took to the podium of the Louisiana governor’s press conference recently and did not mince words.
“The Delta variant is not last year’s virus, and it’s become incredibly apparent to healthcare workers that we are dealing with a different beast,” she said.
Louisiana is one of the least vaccinated states in the country. In the United States as a whole, 48.6% of the population is fully vaccinated. In Louisiana, it’s just 36%, and Delta is bearing down.
Dr. O’Neal spoke about the pressure that rising COVID cases were already putting on her hospital, Our Lady of the Lake Regional Medical Center in Baton Rouge. She talked about watching her peers, 30- and 40-year-olds, become severely ill with the latest iteration of the new coronavirus — the Delta variant — which is sweeping through the United States with astonishing speed, causing new cases, hospitalizations, and deaths to rise again.
Dr. O’Neal talked about parents who might not be alive to see their children go off to college in a few weeks. She talked about increasing hospital admissions for infected kids and pregnant women on ventilators.
“I want to be clear after seeing what we’ve seen the last two weeks. We only have two choices: We are either going to get vaccinated and end the pandemic, or we’re going to accept death and a lot of it,” Dr. O’Neal said, her voice choked by emotion.
Where Delta goes, death follows
Delta was first identified in India, where it caused a devastating surge in the spring. In a population that was largely unvaccinated, researchers think it may have caused as many as three million deaths. In just a few months’ time, it has sped across the globe.
, which was first identified in the United Kingdom).
Where a single infected person might have spread older versions of the virus to two or three others, mathematician and epidemiologist Adam Kucharski, PhD, an associate professor at the London School of Hygiene and Tropical Medicine, thinks that number — called the basic reproduction number — might be around six for Delta, meaning that, on average, each infected person spreads the virus to six others.
“The Delta variant is the most able and fastest and fittest of those viruses,” said Mike Ryan, executive director of the World Health Organization’s Health Emergencies Programme, in a recent press briefing.
Early evidence suggests it may also cause more severe disease in people who are not vaccinated.
“There’s clearly increased risk of ICU admission, hospitalization, and death,” said Ashleigh Tuite, PhD, MPH, an infectious disease epidemiologist at the University of Toronto in Ontario.
In a study published ahead of peer review, Dr. Tuite and her coauthor, David Fisman, MD, MPH, reviewed the health outcomes for more than 200,000 people who tested positive for SARS-CoV-2 in Ontario between February and June of 2021. Starting in February, Ontario began screening all positive COVID tests for mutations in the N501Y region for signs of mutation.
Compared with versions of the coronavirus that circulated in 2020, having an Alpha, Beta, or Gamma variant modestly increased the odds that an infected person would become sicker. The Delta variant raised the risk even higher, more than doubling the odds that an infected person would need to be hospitalized or could die from their infection.
Emerging evidence from England and Scotland, analyzed by Public Health England, also shows an increased risk for hospitalization with Delta. The increases are in line with the Canadian data. Experts caution that the picture may change over time as more evidence is gathered.
“What is causing that? We don’t know,” Dr. Tuite said.
Enhanced virus
The Delta variants (there’s actually more than one in the same viral family) have about 15 different mutations compared with the original virus. Two of these, L452R and E484Q, are mutations to the spike protein that were first flagged as problematic in other variants because they appear to help the virus escape the antibodies we make to fight it.
It has another mutation away from its binding site that’s also getting researchers’ attention — P681R.
This mutation appears to enhance the “springiness” of the parts of the virus that dock onto our cells, said Alexander Greninger, MD, PhD, assistant director of the UW Medicine Clinical Virology Laboratory at the University of Washington in Seattle. So it’s more likely to be in the right position to infect our cells if we come into contact with it.
Another theory is that P681R may also enhance the virus’s ability to fuse cells together into clumps that have several different nuclei. These balls of fused cells are called syncytia.
“So it turns into a big factory for making viruses,” said Kamran Kadkhoda, PhD, medical director of immunopathology at the Cleveland Clinic in Ohio.
This capability is not unique to Delta or even to the new coronavirus. Earlier versions and other viruses can do the same thing, but according to a recent paper in Nature, the syncytia that Delta creates are larger than the ones created by previous variants.
Scientists aren’t sure what these supersized syncytia mean, exactly, but they have some theories. They may help the virus copy itself more quickly, so a person’s viral load builds up quickly. That may enhance the ability of the virus to transmit from person to person.
And at least one recent study from China supports this idea. That study, which was posted ahead of peer review on the website Virological.org, tracked 167 people infected with Delta back to a single index case.
China has used extensive contact tracing to identify people that may have been exposed to the virus and sequester them quickly to tamp down its spread. Once a person is isolated or quarantined, they are tested daily with gold-standard PCR testing to determine whether or not they were infected.
Researchers compared the characteristics of Delta cases with those of people infected in 2020 with previous versions of the virus.
This study found that people infected by Delta tested positive more quickly than their predecessors did. In 2020, it took an average of 6 days for someone to test positive after an exposure. With Delta, it took an average of about 4 days.
When people tested positive, they had more than 1,000 times more virus in their bodies, suggesting that the Delta variant has a higher growth rate in the body.
This gives Delta a big advantage. According to Angie Rasmussen, PhD, a virologist at the Vaccine and Infectious Disease Organization at the University of Saskatchewan in Canada, who posted a thread about the study on Twitter, if people are shedding 1,000 times more virus, it is much more likely that close contacts will be exposed to enough of it to become infected themselves.
And if they’re shedding earlier in the course of their infections, the virus has more opportunity to spread.
This may help explain why Delta is so much more contagious.
Beyond transmission, Delta’s ability to form syncytia may have two other important consequences. It may help the virus hide from our immune system, and it may make the virus more damaging to the body.
Commonly, when a virus infects a cell, it will corrupt the cell’s protein-making machinery to crank out more copies of itself. When the cell dies, these new copies are released into the plasma outside the cell where they can float over and infect new cells. It’s in this extracellular space where a virus can also be attacked by the neutralizing antibodies our immune system makes to fight it off.
“Antibodies don’t penetrate inside the cell. If these viruses are going from one cell to another by just fusing to each other, antibodies become less useful,” Dr. Kadkhoda said.
Escape artist
Recent studies show that Delta is also able to escape antibodies made in response to vaccination more effectively than the Alpha, or B.1.1.7 strain. The effect was more pronounced in older adults, who tend to have weaker responses to vaccines in general.
This evasion of the immune system is particularly problematic for people who are only partially vaccinated. Data from the United Kingdom show that a single dose of vaccine is only about 31% effective at preventing illness with Delta, and 75% effective at preventing hospitalization.
After two doses, the vaccines are still highly effective — even against Delta — reaching 80% protection for illness, and 94% for hospitalization, which is why U.S. officials are begging people to get both doses of their shots, and do it as quickly as possible.
Finally, the virus’s ability to form syncytia may leave greater damage behind in the body’s tissues and organs.
“Especially in the lungs,” Dr. Kadkhoda said. The lungs are very fragile tissues. Their tiny air sacs — the alveoli — are only a single-cell thick. They have to be very thin to exchange oxygen in the blood.
“Any damage like that can severely affect any oxygen exchange and the normal housekeeping activities of that tissue,” he said. “In those vital organs, it may be very problematic.”
The research is still early, but studies in animals and cell lines are backing up what doctors say they are seeing in hospitalized patients.
A recent preprint study from researchers in Japan found that hamsters infected with Delta lost more weight — a proxy for how sick they were — compared with hamsters infected with an older version of the virus. The researchers attribute this to the viruses› ability to fuse cells together to form syncytia.
Another investigation, from researchers in India, infected two groups of hamsters — one with the original “wild type” strain of the virus, the other with the Delta variant of the new coronavirus.
As in the Japanese study, the hamsters infected with Delta lost more weight. When the researchers performed necropsies on the animals, they found more lung damage and bleeding in hamsters infected with Delta. This study was also posted as a preprint ahead of peer review.
German researchers working with pseudotyped versions of the new coronavirus — viruses that have been genetically changed to make them safer to work with — watched what happened after they used these pseudoviruses to infect lung, colon, and kidney cells in the lab.
They, too, found that cells infected with the Delta variant formed more and larger syncytia compared with cells infected with the wild type strain of the virus. The authors write that their findings suggest Delta could “cause more tissue damage, and thus be more pathogenic, than previous variants.”Researchers say it’s important to remember that, while interesting, this research isn’t conclusive. Hamsters and cells aren’t humans. More studies are needed to prove these theories.
Scientists say that what we already know about Delta makes vaccination more important than ever.
“The net effect is really that, you know, this is worrisome in people who are unvaccinated and then people who have breakthrough infections, but it’s not…a reason to panic or to throw up our hands and say you know, this pandemic is never going to end,” Dr. Tuite said, “[b]ecause what we do see is that the vaccines continue to be highly protective.”
A version of this article first appeared on Medscape.com.
Catherine O’Neal, MD, an infectious disease physician, took to the podium of the Louisiana governor’s press conference recently and did not mince words.
“The Delta variant is not last year’s virus, and it’s become incredibly apparent to healthcare workers that we are dealing with a different beast,” she said.
Louisiana is one of the least vaccinated states in the country. In the United States as a whole, 48.6% of the population is fully vaccinated. In Louisiana, it’s just 36%, and Delta is bearing down.
Dr. O’Neal spoke about the pressure that rising COVID cases were already putting on her hospital, Our Lady of the Lake Regional Medical Center in Baton Rouge. She talked about watching her peers, 30- and 40-year-olds, become severely ill with the latest iteration of the new coronavirus — the Delta variant — which is sweeping through the United States with astonishing speed, causing new cases, hospitalizations, and deaths to rise again.
Dr. O’Neal talked about parents who might not be alive to see their children go off to college in a few weeks. She talked about increasing hospital admissions for infected kids and pregnant women on ventilators.
“I want to be clear after seeing what we’ve seen the last two weeks. We only have two choices: We are either going to get vaccinated and end the pandemic, or we’re going to accept death and a lot of it,” Dr. O’Neal said, her voice choked by emotion.
Where Delta goes, death follows
Delta was first identified in India, where it caused a devastating surge in the spring. In a population that was largely unvaccinated, researchers think it may have caused as many as three million deaths. In just a few months’ time, it has sped across the globe.
, which was first identified in the United Kingdom).
Where a single infected person might have spread older versions of the virus to two or three others, mathematician and epidemiologist Adam Kucharski, PhD, an associate professor at the London School of Hygiene and Tropical Medicine, thinks that number — called the basic reproduction number — might be around six for Delta, meaning that, on average, each infected person spreads the virus to six others.
“The Delta variant is the most able and fastest and fittest of those viruses,” said Mike Ryan, executive director of the World Health Organization’s Health Emergencies Programme, in a recent press briefing.
Early evidence suggests it may also cause more severe disease in people who are not vaccinated.
“There’s clearly increased risk of ICU admission, hospitalization, and death,” said Ashleigh Tuite, PhD, MPH, an infectious disease epidemiologist at the University of Toronto in Ontario.
In a study published ahead of peer review, Dr. Tuite and her coauthor, David Fisman, MD, MPH, reviewed the health outcomes for more than 200,000 people who tested positive for SARS-CoV-2 in Ontario between February and June of 2021. Starting in February, Ontario began screening all positive COVID tests for mutations in the N501Y region for signs of mutation.
Compared with versions of the coronavirus that circulated in 2020, having an Alpha, Beta, or Gamma variant modestly increased the odds that an infected person would become sicker. The Delta variant raised the risk even higher, more than doubling the odds that an infected person would need to be hospitalized or could die from their infection.
Emerging evidence from England and Scotland, analyzed by Public Health England, also shows an increased risk for hospitalization with Delta. The increases are in line with the Canadian data. Experts caution that the picture may change over time as more evidence is gathered.
“What is causing that? We don’t know,” Dr. Tuite said.
Enhanced virus
The Delta variants (there’s actually more than one in the same viral family) have about 15 different mutations compared with the original virus. Two of these, L452R and E484Q, are mutations to the spike protein that were first flagged as problematic in other variants because they appear to help the virus escape the antibodies we make to fight it.
It has another mutation away from its binding site that’s also getting researchers’ attention — P681R.
This mutation appears to enhance the “springiness” of the parts of the virus that dock onto our cells, said Alexander Greninger, MD, PhD, assistant director of the UW Medicine Clinical Virology Laboratory at the University of Washington in Seattle. So it’s more likely to be in the right position to infect our cells if we come into contact with it.
Another theory is that P681R may also enhance the virus’s ability to fuse cells together into clumps that have several different nuclei. These balls of fused cells are called syncytia.
“So it turns into a big factory for making viruses,” said Kamran Kadkhoda, PhD, medical director of immunopathology at the Cleveland Clinic in Ohio.
This capability is not unique to Delta or even to the new coronavirus. Earlier versions and other viruses can do the same thing, but according to a recent paper in Nature, the syncytia that Delta creates are larger than the ones created by previous variants.
Scientists aren’t sure what these supersized syncytia mean, exactly, but they have some theories. They may help the virus copy itself more quickly, so a person’s viral load builds up quickly. That may enhance the ability of the virus to transmit from person to person.
And at least one recent study from China supports this idea. That study, which was posted ahead of peer review on the website Virological.org, tracked 167 people infected with Delta back to a single index case.
China has used extensive contact tracing to identify people that may have been exposed to the virus and sequester them quickly to tamp down its spread. Once a person is isolated or quarantined, they are tested daily with gold-standard PCR testing to determine whether or not they were infected.
Researchers compared the characteristics of Delta cases with those of people infected in 2020 with previous versions of the virus.
This study found that people infected by Delta tested positive more quickly than their predecessors did. In 2020, it took an average of 6 days for someone to test positive after an exposure. With Delta, it took an average of about 4 days.
When people tested positive, they had more than 1,000 times more virus in their bodies, suggesting that the Delta variant has a higher growth rate in the body.
This gives Delta a big advantage. According to Angie Rasmussen, PhD, a virologist at the Vaccine and Infectious Disease Organization at the University of Saskatchewan in Canada, who posted a thread about the study on Twitter, if people are shedding 1,000 times more virus, it is much more likely that close contacts will be exposed to enough of it to become infected themselves.
And if they’re shedding earlier in the course of their infections, the virus has more opportunity to spread.
This may help explain why Delta is so much more contagious.
Beyond transmission, Delta’s ability to form syncytia may have two other important consequences. It may help the virus hide from our immune system, and it may make the virus more damaging to the body.
Commonly, when a virus infects a cell, it will corrupt the cell’s protein-making machinery to crank out more copies of itself. When the cell dies, these new copies are released into the plasma outside the cell where they can float over and infect new cells. It’s in this extracellular space where a virus can also be attacked by the neutralizing antibodies our immune system makes to fight it off.
“Antibodies don’t penetrate inside the cell. If these viruses are going from one cell to another by just fusing to each other, antibodies become less useful,” Dr. Kadkhoda said.
Escape artist
Recent studies show that Delta is also able to escape antibodies made in response to vaccination more effectively than the Alpha, or B.1.1.7 strain. The effect was more pronounced in older adults, who tend to have weaker responses to vaccines in general.
This evasion of the immune system is particularly problematic for people who are only partially vaccinated. Data from the United Kingdom show that a single dose of vaccine is only about 31% effective at preventing illness with Delta, and 75% effective at preventing hospitalization.
After two doses, the vaccines are still highly effective — even against Delta — reaching 80% protection for illness, and 94% for hospitalization, which is why U.S. officials are begging people to get both doses of their shots, and do it as quickly as possible.
Finally, the virus’s ability to form syncytia may leave greater damage behind in the body’s tissues and organs.
“Especially in the lungs,” Dr. Kadkhoda said. The lungs are very fragile tissues. Their tiny air sacs — the alveoli — are only a single-cell thick. They have to be very thin to exchange oxygen in the blood.
“Any damage like that can severely affect any oxygen exchange and the normal housekeeping activities of that tissue,” he said. “In those vital organs, it may be very problematic.”
The research is still early, but studies in animals and cell lines are backing up what doctors say they are seeing in hospitalized patients.
A recent preprint study from researchers in Japan found that hamsters infected with Delta lost more weight — a proxy for how sick they were — compared with hamsters infected with an older version of the virus. The researchers attribute this to the viruses› ability to fuse cells together to form syncytia.
Another investigation, from researchers in India, infected two groups of hamsters — one with the original “wild type” strain of the virus, the other with the Delta variant of the new coronavirus.
As in the Japanese study, the hamsters infected with Delta lost more weight. When the researchers performed necropsies on the animals, they found more lung damage and bleeding in hamsters infected with Delta. This study was also posted as a preprint ahead of peer review.
German researchers working with pseudotyped versions of the new coronavirus — viruses that have been genetically changed to make them safer to work with — watched what happened after they used these pseudoviruses to infect lung, colon, and kidney cells in the lab.
They, too, found that cells infected with the Delta variant formed more and larger syncytia compared with cells infected with the wild type strain of the virus. The authors write that their findings suggest Delta could “cause more tissue damage, and thus be more pathogenic, than previous variants.”Researchers say it’s important to remember that, while interesting, this research isn’t conclusive. Hamsters and cells aren’t humans. More studies are needed to prove these theories.
Scientists say that what we already know about Delta makes vaccination more important than ever.
“The net effect is really that, you know, this is worrisome in people who are unvaccinated and then people who have breakthrough infections, but it’s not…a reason to panic or to throw up our hands and say you know, this pandemic is never going to end,” Dr. Tuite said, “[b]ecause what we do see is that the vaccines continue to be highly protective.”
A version of this article first appeared on Medscape.com.
Children and COVID: New vaccinations increase as cases continue to climb
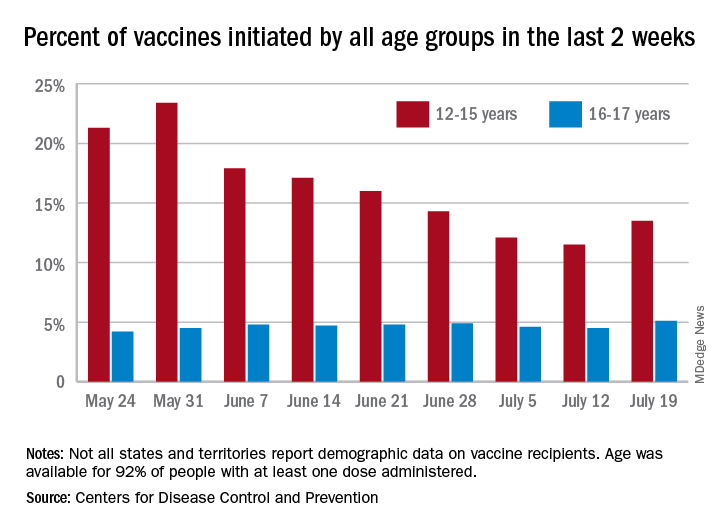
Children aged 12-15 years represented 13.5% of all first vaccinations received during the 2 weeks ending July 19, compared with 11.5% for the 2 weeks ending July 12, marking the first increase since the end of May. First vaccinations in 16- and 17-year-olds, who make up a much smaller share of the U.S. population, also went up, topping 5%, the Centers for Disease Control and Prevention said in its COVID Data Tracker.
The total number of vaccine initiations was almost 250,000 for the week ending July 19, after dropping to a low of 201,000 the previous week. Before that, first vaccinations had fallen in 5 of the previous 6 weeks, going from 1.4 million on May 24 to 307,000 on July 5, the CDC said.
New cases of COVID-19, unfortunately, continued to follow the trend among the larger population: As of July 15, weekly cases in children were up by 179% since dropping to 8,400 on June 24, the American Academy of Pediatrics and the Children’s Hospital Association said in a joint report. The 23,551 new cases in children for the week ending July 15 were 15.9% of all cases reported.
With those new cases, the total number of children infected with COVID-19 comes to almost 4.1 million since the start of the pandemic, the AAP and CHA said. The CDC data indicate that just over 5.35 million children aged 12-15 years and 3.53 million 16- and 17-year-olds have received at least one dose of the COVID-19 vaccine and that 6.8 million children aged 12-17 are fully vaccinated.
Fully vaccinated children represent 26.4% of all 12- to 15-year-olds and 38.3% of the 16- 17-year-olds as of July 19. The corresponding numbers for those who have received at least one dose are 35.2% (ages 12-15) and 46.8% (16-17), the CDC said.
The AAP recently recommended in-person learning with universal masking in schools this fall “because a significant portion of the student population is not yet eligible for vaccines. ... Many schools will not have a system to monitor vaccine status of students, teachers and staff, and some communities overall have low vaccination uptake where the virus may be circulating more prominently.”

Children aged 12-15 years represented 13.5% of all first vaccinations received during the 2 weeks ending July 19, compared with 11.5% for the 2 weeks ending July 12, marking the first increase since the end of May. First vaccinations in 16- and 17-year-olds, who make up a much smaller share of the U.S. population, also went up, topping 5%, the Centers for Disease Control and Prevention said in its COVID Data Tracker.
The total number of vaccine initiations was almost 250,000 for the week ending July 19, after dropping to a low of 201,000 the previous week. Before that, first vaccinations had fallen in 5 of the previous 6 weeks, going from 1.4 million on May 24 to 307,000 on July 5, the CDC said.
New cases of COVID-19, unfortunately, continued to follow the trend among the larger population: As of July 15, weekly cases in children were up by 179% since dropping to 8,400 on June 24, the American Academy of Pediatrics and the Children’s Hospital Association said in a joint report. The 23,551 new cases in children for the week ending July 15 were 15.9% of all cases reported.
With those new cases, the total number of children infected with COVID-19 comes to almost 4.1 million since the start of the pandemic, the AAP and CHA said. The CDC data indicate that just over 5.35 million children aged 12-15 years and 3.53 million 16- and 17-year-olds have received at least one dose of the COVID-19 vaccine and that 6.8 million children aged 12-17 are fully vaccinated.
Fully vaccinated children represent 26.4% of all 12- to 15-year-olds and 38.3% of the 16- 17-year-olds as of July 19. The corresponding numbers for those who have received at least one dose are 35.2% (ages 12-15) and 46.8% (16-17), the CDC said.
The AAP recently recommended in-person learning with universal masking in schools this fall “because a significant portion of the student population is not yet eligible for vaccines. ... Many schools will not have a system to monitor vaccine status of students, teachers and staff, and some communities overall have low vaccination uptake where the virus may be circulating more prominently.”

Children aged 12-15 years represented 13.5% of all first vaccinations received during the 2 weeks ending July 19, compared with 11.5% for the 2 weeks ending July 12, marking the first increase since the end of May. First vaccinations in 16- and 17-year-olds, who make up a much smaller share of the U.S. population, also went up, topping 5%, the Centers for Disease Control and Prevention said in its COVID Data Tracker.
The total number of vaccine initiations was almost 250,000 for the week ending July 19, after dropping to a low of 201,000 the previous week. Before that, first vaccinations had fallen in 5 of the previous 6 weeks, going from 1.4 million on May 24 to 307,000 on July 5, the CDC said.
New cases of COVID-19, unfortunately, continued to follow the trend among the larger population: As of July 15, weekly cases in children were up by 179% since dropping to 8,400 on June 24, the American Academy of Pediatrics and the Children’s Hospital Association said in a joint report. The 23,551 new cases in children for the week ending July 15 were 15.9% of all cases reported.
With those new cases, the total number of children infected with COVID-19 comes to almost 4.1 million since the start of the pandemic, the AAP and CHA said. The CDC data indicate that just over 5.35 million children aged 12-15 years and 3.53 million 16- and 17-year-olds have received at least one dose of the COVID-19 vaccine and that 6.8 million children aged 12-17 are fully vaccinated.
Fully vaccinated children represent 26.4% of all 12- to 15-year-olds and 38.3% of the 16- 17-year-olds as of July 19. The corresponding numbers for those who have received at least one dose are 35.2% (ages 12-15) and 46.8% (16-17), the CDC said.
The AAP recently recommended in-person learning with universal masking in schools this fall “because a significant portion of the student population is not yet eligible for vaccines. ... Many schools will not have a system to monitor vaccine status of students, teachers and staff, and some communities overall have low vaccination uptake where the virus may be circulating more prominently.”
Pandemic drives drop in prescription drugs for children
The amount of prescription drugs given to children in the United States decreased by 27.1% between April and December 2020, compared with the same period in 2019, based on data from a national database.
Overall, dispensing of prescription drugs to all patients in the United States decreased in the wake of COVID-19 but has since rebounded, wrote Kao-Ping Chua, MD, of the University of Michigan, Ann Arbor, and colleagues. “However, whether these same trends occurred for children is unknown.”
In a study published in Pediatrics, the researchers used the IQVIA National Prescription Audit, a database that contains monthly dispensing details from 92% of retail pharmacies in the United States. They compared changes in the dispensing of prescriptions with children aged 0-19 years during 2018-2020.
In the April 2020–December 2020 time period, prescriptions for children aged 1-2 years, 3-9 years, and 10-19 years decreased by 48.7%, 40.6%, and 16.8%, respectively, compared with the same time period in 2019.
The overall dispensing total for children from April 2020 to December 2020 was 160,630,406, representing a 27.1% reduction, compared with the 220,284,613 total from April 2019 to December 2019.
By drug class, prescriptions for antibiotics, ADHD medications, and antidepressants decreased by 55.6%, 11.8%, and 0.1%, respectively, in comparing the two time periods. Prescriptions for drug classes used typically for acute infections decreased by 51.3%, and those used for chronic diseases decreased by 17.4%.
From January 2018 to February 2020, a median of 25,744,758 prescriptions were dispensed to children aged 0-19 years each month. The total prescriptions decreased from 25,684,219 in March 2020 to 16,742,568 in April 2020, increased to 19,657,289 in October 2020, and decreased again to 15,821,914 during December 2020.
In a subgroup analysis, the decline in prescriptions was greater in children aged 0-9 years, compared with those aged 10-19 years. “Because young children have a higher rate of antibiotic use than older children, declines in antibiotic dispensing might affect overall dispensing totals to a greater degree in young children,” the researchers said.
The study findings were limited by several factors including the lack of information on clinical outcomes, disease severity, and details of new versus ongoing prescriptions, as well as the possible heterogeneity in indications within drug classes, and lack of data from small pharmacies, the researchers noted. However, the results were strengthened by the use of a national all-payer database that including most prescriptions dispensed in the United States, and the use of objective measurements of prescribing practices rather than self-reports.
Despite concerns for the decreased dispensing of chronic disease drugs to children during the pandemic, “declines in dispensing of infection-related drugs, such as antitussives and antibiotics, may be welcome developments,” the researchers said. “These declines reveal that substantial reductions in prescribing of these drugs are possible,” and ongoing monitoring is needed to follow whether the reductions continue long term.
COVID precautions contributed to prescription declines
The mask-wearing and social distancing imposed by the COVID-19 pandemic has contributed to reduced rates of other illnesses, Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn., said in an interview.
“On the surface, with masks and social isolation, we have seen a drastic reduction in infectious disease,” she said. Fewer infections mean a reduced need for prescriptions to treat them. However, Dr. Kinsella expects the situation to change as more venues and activities open. “I expect that, as things continue to open, we will continue to see more infectious disease,” which will likely lead to more prescription drug use.
Part of the study data were provided through the IQVIA Institute’s Human Data Science Research Collaborative. Lead author Dr. Chua was supported by a career development award from the National Institute on Drug Abuse, but had no financial conflicts to disclose. Dr. Kinsella had no financial conflicts to disclose, but serves as a member of the Pediatric News editorial advisory board.
The amount of prescription drugs given to children in the United States decreased by 27.1% between April and December 2020, compared with the same period in 2019, based on data from a national database.
Overall, dispensing of prescription drugs to all patients in the United States decreased in the wake of COVID-19 but has since rebounded, wrote Kao-Ping Chua, MD, of the University of Michigan, Ann Arbor, and colleagues. “However, whether these same trends occurred for children is unknown.”
In a study published in Pediatrics, the researchers used the IQVIA National Prescription Audit, a database that contains monthly dispensing details from 92% of retail pharmacies in the United States. They compared changes in the dispensing of prescriptions with children aged 0-19 years during 2018-2020.
In the April 2020–December 2020 time period, prescriptions for children aged 1-2 years, 3-9 years, and 10-19 years decreased by 48.7%, 40.6%, and 16.8%, respectively, compared with the same time period in 2019.
The overall dispensing total for children from April 2020 to December 2020 was 160,630,406, representing a 27.1% reduction, compared with the 220,284,613 total from April 2019 to December 2019.
By drug class, prescriptions for antibiotics, ADHD medications, and antidepressants decreased by 55.6%, 11.8%, and 0.1%, respectively, in comparing the two time periods. Prescriptions for drug classes used typically for acute infections decreased by 51.3%, and those used for chronic diseases decreased by 17.4%.
From January 2018 to February 2020, a median of 25,744,758 prescriptions were dispensed to children aged 0-19 years each month. The total prescriptions decreased from 25,684,219 in March 2020 to 16,742,568 in April 2020, increased to 19,657,289 in October 2020, and decreased again to 15,821,914 during December 2020.
In a subgroup analysis, the decline in prescriptions was greater in children aged 0-9 years, compared with those aged 10-19 years. “Because young children have a higher rate of antibiotic use than older children, declines in antibiotic dispensing might affect overall dispensing totals to a greater degree in young children,” the researchers said.
The study findings were limited by several factors including the lack of information on clinical outcomes, disease severity, and details of new versus ongoing prescriptions, as well as the possible heterogeneity in indications within drug classes, and lack of data from small pharmacies, the researchers noted. However, the results were strengthened by the use of a national all-payer database that including most prescriptions dispensed in the United States, and the use of objective measurements of prescribing practices rather than self-reports.
Despite concerns for the decreased dispensing of chronic disease drugs to children during the pandemic, “declines in dispensing of infection-related drugs, such as antitussives and antibiotics, may be welcome developments,” the researchers said. “These declines reveal that substantial reductions in prescribing of these drugs are possible,” and ongoing monitoring is needed to follow whether the reductions continue long term.
COVID precautions contributed to prescription declines
The mask-wearing and social distancing imposed by the COVID-19 pandemic has contributed to reduced rates of other illnesses, Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn., said in an interview.
“On the surface, with masks and social isolation, we have seen a drastic reduction in infectious disease,” she said. Fewer infections mean a reduced need for prescriptions to treat them. However, Dr. Kinsella expects the situation to change as more venues and activities open. “I expect that, as things continue to open, we will continue to see more infectious disease,” which will likely lead to more prescription drug use.
Part of the study data were provided through the IQVIA Institute’s Human Data Science Research Collaborative. Lead author Dr. Chua was supported by a career development award from the National Institute on Drug Abuse, but had no financial conflicts to disclose. Dr. Kinsella had no financial conflicts to disclose, but serves as a member of the Pediatric News editorial advisory board.
The amount of prescription drugs given to children in the United States decreased by 27.1% between April and December 2020, compared with the same period in 2019, based on data from a national database.
Overall, dispensing of prescription drugs to all patients in the United States decreased in the wake of COVID-19 but has since rebounded, wrote Kao-Ping Chua, MD, of the University of Michigan, Ann Arbor, and colleagues. “However, whether these same trends occurred for children is unknown.”
In a study published in Pediatrics, the researchers used the IQVIA National Prescription Audit, a database that contains monthly dispensing details from 92% of retail pharmacies in the United States. They compared changes in the dispensing of prescriptions with children aged 0-19 years during 2018-2020.
In the April 2020–December 2020 time period, prescriptions for children aged 1-2 years, 3-9 years, and 10-19 years decreased by 48.7%, 40.6%, and 16.8%, respectively, compared with the same time period in 2019.
The overall dispensing total for children from April 2020 to December 2020 was 160,630,406, representing a 27.1% reduction, compared with the 220,284,613 total from April 2019 to December 2019.
By drug class, prescriptions for antibiotics, ADHD medications, and antidepressants decreased by 55.6%, 11.8%, and 0.1%, respectively, in comparing the two time periods. Prescriptions for drug classes used typically for acute infections decreased by 51.3%, and those used for chronic diseases decreased by 17.4%.
From January 2018 to February 2020, a median of 25,744,758 prescriptions were dispensed to children aged 0-19 years each month. The total prescriptions decreased from 25,684,219 in March 2020 to 16,742,568 in April 2020, increased to 19,657,289 in October 2020, and decreased again to 15,821,914 during December 2020.
In a subgroup analysis, the decline in prescriptions was greater in children aged 0-9 years, compared with those aged 10-19 years. “Because young children have a higher rate of antibiotic use than older children, declines in antibiotic dispensing might affect overall dispensing totals to a greater degree in young children,” the researchers said.
The study findings were limited by several factors including the lack of information on clinical outcomes, disease severity, and details of new versus ongoing prescriptions, as well as the possible heterogeneity in indications within drug classes, and lack of data from small pharmacies, the researchers noted. However, the results were strengthened by the use of a national all-payer database that including most prescriptions dispensed in the United States, and the use of objective measurements of prescribing practices rather than self-reports.
Despite concerns for the decreased dispensing of chronic disease drugs to children during the pandemic, “declines in dispensing of infection-related drugs, such as antitussives and antibiotics, may be welcome developments,” the researchers said. “These declines reveal that substantial reductions in prescribing of these drugs are possible,” and ongoing monitoring is needed to follow whether the reductions continue long term.
COVID precautions contributed to prescription declines
The mask-wearing and social distancing imposed by the COVID-19 pandemic has contributed to reduced rates of other illnesses, Karalyn Kinsella, MD, a pediatrician in private practice in Cheshire, Conn., said in an interview.
“On the surface, with masks and social isolation, we have seen a drastic reduction in infectious disease,” she said. Fewer infections mean a reduced need for prescriptions to treat them. However, Dr. Kinsella expects the situation to change as more venues and activities open. “I expect that, as things continue to open, we will continue to see more infectious disease,” which will likely lead to more prescription drug use.
Part of the study data were provided through the IQVIA Institute’s Human Data Science Research Collaborative. Lead author Dr. Chua was supported by a career development award from the National Institute on Drug Abuse, but had no financial conflicts to disclose. Dr. Kinsella had no financial conflicts to disclose, but serves as a member of the Pediatric News editorial advisory board.
FROM PEDIATRICS
Long COVID seen in patients with severe and mild disease
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM EUROPEAN CONGRESS OF CLINICAL MICROBIOLOGY & INFECTIOUS DISEASES
The febrile infant: New AAP guidance for the first 2 months of life
Sixteen years in the making, the American Academy of Pediatrics just released a new clinical practice guideline (CPG), “Evaluation and Management of Well-Appearing Febrile Infants 8-60 Days Old”. The recommendations were derived from interpretations of sequential studies in young, febrile, but well-appearing infants that covered invasive bacterial infection (IBI) incidence, diagnostic modalities, and treatment during the first 2 months of life, further refining approaches to evaluation and empirical treatment.
Pediatricians have long had solid information to help assess the risk for IBI among febrile infants aged 0-3 months, but there has been an ongoing desire to further refine the suggested evaluation of these very young infants. A study of febrile infants from the Pediatric Research in Office Settings network along with subsequent evidence has identified the first 3 weeks of life as the period of highest risk for IBI, with risk declining in a graded fashion aged between 22 and 56 days.
Critical caveats
First, some caveats. Infants 0-7 days are not addressed in the CPG, and all should be treated as high risk and receive full IBI evaluation according to newborn protocols. Second, the recommendations apply only to “well-appearing” infants. Any ill-appearing infant should be treated as high risk and receive full IBI evaluation and begun on empirical antimicrobials. Third, even though the CPG deals with infants as young as 8-21 days old, the recommendations are to treat all infants in this age group as high risk, even if well-appearing, and complete full IBI evaluation and empirical therapy while awaiting results. Fourth, these guidelines apply only to infants born at 37 weeks’ gestation or more. Finally, the new CPG action statements are meant to be recommendations rather than a standard of medical care, leaving some leeway for clinician interpretation of individual patient scenarios. Where appropriate, parents’ values and preferences should be incorporated as part of shared decision-making.
The CPG divides young, febrile infants into three cohorts based on age:
- 8-21 days old
- 22-28 days old
- 29-60 days old
Age 8-21 days
For well-appearing febrile infants 8-21 days old, the CPG recommends a complete IBI evaluation that includes urine, blood, and cerebrospinal fluid (CSF) for culture, approaching all infants in this cohort as high risk. Inflammatory markers may be obtained, but the evidence is not comprehensive enough to evaluate their role in decision-making for this age group. A two-step urine evaluation method (urine analysis followed by culture if the urine analysis looks concerning) is not recommended for infants aged 8-21 days. Urine samples for culture from these young infants should be obtained by catheterization or suprapubic aspiration.
The CPG recommends drawing blood cultures and CSF by lumbar puncture from this cohort. These infants should be admitted to the hospital, treated empirically with antimicrobials, and actively monitored. However, if the cultures are negative at 24-36 hours, the clinician should discontinue antimicrobials and discharge the infant if there is no other reason for continued hospitalization.
Age 22-28 days
Well-appearing, febrile infants 22-28 days old are in an intermediate-risk zone. The recommendation for infants in this cohort is to obtain a urine specimen by catheterization or suprapubic aspiration for both urine analysis and culture. Clinicians may consider obtaining urine samples for analysis noninvasively (e.g., urine bag) in this cohort, but this is not the preferred method.
Blood culture should be obtained from all infants in this group. Inflammatory markers can help clinicians identify infants at greater risk for IBI, including meningitis. Previous data suggested that inflammatory markers such as serum white blood cell counts greater than 11,000/mcL, a serum absolute neutrophil count of greater than 4,000/mcL, and elevated C-reactive protein and procalcitonin levels could help providers identify febrile infants with true IBI. A 2008 study demonstrated that procalcitonin had the best receiver operating characteristic curve in regard to predicting IBI in young febrile infants. Other research backed up that finding and identified cutoff values for procalcitonin levels greater than 1.0 ng/mL. The CPG recommends considering a procalcitonin value of 0.5 ng/mL or higher as positive, indicating that the infant is at greater risk for IBI and potentially should undergo an expanded IBI workup. Therefore, in infants aged 22-28 days, inflammatory markers can play a role in deciding whether to perform a lumbar puncture.
Many more nuanced recommendations for whether to and how to empirically treat with antimicrobials in this cohort can be found in the CPG, including whether to manage in the hospital or at home. Treatment recommendations vary greatly for this cohort on the basis of the tests obtained and whether tests were positive or negative at the initial evaluation.
Age 29-60 days
The CPG will be most helpful when clinicians are faced with well-appearing, febrile infants in the 29- to 60-day age group. As with the other groups, a urine evaluation is recommended; however, the CPG suggests that the two-step approach – obtaining a urine analysis by a noninvasive method and only obtaining culture if the urine analysis is positive – is reasonable. This means that a bag or free-flowing urine specimen would be appropriate for urinalysis, followed by catheterization/suprapubic aspiration if a culture is necessary. This would save approximately 90% of infants from invasive urine collection. Regardless, only catheter or suprapubic specimens are appropriate for urine culture.
The CPG also recommends that clinicians obtain blood culture on all of these infants. Inflammatory markers should be assessed in this cohort because avoiding lumbar puncture for CSF culture would be appropriate in this cohort if the inflammatory markers are negative. If CSF is obtained in this age cohort, enterovirus testing should be added to the testing regimen. Again, for any infant considered at higher risk for IBI on the basis of screening tests, the CPG recommends a 24- to 36-hour rule-out period with empirical antimicrobial treatment and active monitoring in the hospital.
Summary
The recommended approach for febrile infants 8-21 days old is relatively aggressive, with urine, blood, and CSF evaluation for IBI. Clinicians gain some leeway for infants age 22-28 days, but the guidelines recommend a more flexible approach to evaluating well-appearing, febrile infants age 29-60 days, when a two-step urine evaluation and inflammatory marker assessment can help clinicians and parents have a better discussion about the risk-benefit trade-offs of more aggressive testing and empirical treatment.
The author would like to thank Ken Roberts, MD, for his review and helpful comments on this summary of the CPG highlights. Summary points of the CPG were presented by the writing group at the 2021 Pediatric Academic Societies meeting.
William T. Basco, Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Sixteen years in the making, the American Academy of Pediatrics just released a new clinical practice guideline (CPG), “Evaluation and Management of Well-Appearing Febrile Infants 8-60 Days Old”. The recommendations were derived from interpretations of sequential studies in young, febrile, but well-appearing infants that covered invasive bacterial infection (IBI) incidence, diagnostic modalities, and treatment during the first 2 months of life, further refining approaches to evaluation and empirical treatment.
Pediatricians have long had solid information to help assess the risk for IBI among febrile infants aged 0-3 months, but there has been an ongoing desire to further refine the suggested evaluation of these very young infants. A study of febrile infants from the Pediatric Research in Office Settings network along with subsequent evidence has identified the first 3 weeks of life as the period of highest risk for IBI, with risk declining in a graded fashion aged between 22 and 56 days.
Critical caveats
First, some caveats. Infants 0-7 days are not addressed in the CPG, and all should be treated as high risk and receive full IBI evaluation according to newborn protocols. Second, the recommendations apply only to “well-appearing” infants. Any ill-appearing infant should be treated as high risk and receive full IBI evaluation and begun on empirical antimicrobials. Third, even though the CPG deals with infants as young as 8-21 days old, the recommendations are to treat all infants in this age group as high risk, even if well-appearing, and complete full IBI evaluation and empirical therapy while awaiting results. Fourth, these guidelines apply only to infants born at 37 weeks’ gestation or more. Finally, the new CPG action statements are meant to be recommendations rather than a standard of medical care, leaving some leeway for clinician interpretation of individual patient scenarios. Where appropriate, parents’ values and preferences should be incorporated as part of shared decision-making.
The CPG divides young, febrile infants into three cohorts based on age:
- 8-21 days old
- 22-28 days old
- 29-60 days old
Age 8-21 days
For well-appearing febrile infants 8-21 days old, the CPG recommends a complete IBI evaluation that includes urine, blood, and cerebrospinal fluid (CSF) for culture, approaching all infants in this cohort as high risk. Inflammatory markers may be obtained, but the evidence is not comprehensive enough to evaluate their role in decision-making for this age group. A two-step urine evaluation method (urine analysis followed by culture if the urine analysis looks concerning) is not recommended for infants aged 8-21 days. Urine samples for culture from these young infants should be obtained by catheterization or suprapubic aspiration.
The CPG recommends drawing blood cultures and CSF by lumbar puncture from this cohort. These infants should be admitted to the hospital, treated empirically with antimicrobials, and actively monitored. However, if the cultures are negative at 24-36 hours, the clinician should discontinue antimicrobials and discharge the infant if there is no other reason for continued hospitalization.
Age 22-28 days
Well-appearing, febrile infants 22-28 days old are in an intermediate-risk zone. The recommendation for infants in this cohort is to obtain a urine specimen by catheterization or suprapubic aspiration for both urine analysis and culture. Clinicians may consider obtaining urine samples for analysis noninvasively (e.g., urine bag) in this cohort, but this is not the preferred method.
Blood culture should be obtained from all infants in this group. Inflammatory markers can help clinicians identify infants at greater risk for IBI, including meningitis. Previous data suggested that inflammatory markers such as serum white blood cell counts greater than 11,000/mcL, a serum absolute neutrophil count of greater than 4,000/mcL, and elevated C-reactive protein and procalcitonin levels could help providers identify febrile infants with true IBI. A 2008 study demonstrated that procalcitonin had the best receiver operating characteristic curve in regard to predicting IBI in young febrile infants. Other research backed up that finding and identified cutoff values for procalcitonin levels greater than 1.0 ng/mL. The CPG recommends considering a procalcitonin value of 0.5 ng/mL or higher as positive, indicating that the infant is at greater risk for IBI and potentially should undergo an expanded IBI workup. Therefore, in infants aged 22-28 days, inflammatory markers can play a role in deciding whether to perform a lumbar puncture.
Many more nuanced recommendations for whether to and how to empirically treat with antimicrobials in this cohort can be found in the CPG, including whether to manage in the hospital or at home. Treatment recommendations vary greatly for this cohort on the basis of the tests obtained and whether tests were positive or negative at the initial evaluation.
Age 29-60 days
The CPG will be most helpful when clinicians are faced with well-appearing, febrile infants in the 29- to 60-day age group. As with the other groups, a urine evaluation is recommended; however, the CPG suggests that the two-step approach – obtaining a urine analysis by a noninvasive method and only obtaining culture if the urine analysis is positive – is reasonable. This means that a bag or free-flowing urine specimen would be appropriate for urinalysis, followed by catheterization/suprapubic aspiration if a culture is necessary. This would save approximately 90% of infants from invasive urine collection. Regardless, only catheter or suprapubic specimens are appropriate for urine culture.
The CPG also recommends that clinicians obtain blood culture on all of these infants. Inflammatory markers should be assessed in this cohort because avoiding lumbar puncture for CSF culture would be appropriate in this cohort if the inflammatory markers are negative. If CSF is obtained in this age cohort, enterovirus testing should be added to the testing regimen. Again, for any infant considered at higher risk for IBI on the basis of screening tests, the CPG recommends a 24- to 36-hour rule-out period with empirical antimicrobial treatment and active monitoring in the hospital.
Summary
The recommended approach for febrile infants 8-21 days old is relatively aggressive, with urine, blood, and CSF evaluation for IBI. Clinicians gain some leeway for infants age 22-28 days, but the guidelines recommend a more flexible approach to evaluating well-appearing, febrile infants age 29-60 days, when a two-step urine evaluation and inflammatory marker assessment can help clinicians and parents have a better discussion about the risk-benefit trade-offs of more aggressive testing and empirical treatment.
The author would like to thank Ken Roberts, MD, for his review and helpful comments on this summary of the CPG highlights. Summary points of the CPG were presented by the writing group at the 2021 Pediatric Academic Societies meeting.
William T. Basco, Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
Sixteen years in the making, the American Academy of Pediatrics just released a new clinical practice guideline (CPG), “Evaluation and Management of Well-Appearing Febrile Infants 8-60 Days Old”. The recommendations were derived from interpretations of sequential studies in young, febrile, but well-appearing infants that covered invasive bacterial infection (IBI) incidence, diagnostic modalities, and treatment during the first 2 months of life, further refining approaches to evaluation and empirical treatment.
Pediatricians have long had solid information to help assess the risk for IBI among febrile infants aged 0-3 months, but there has been an ongoing desire to further refine the suggested evaluation of these very young infants. A study of febrile infants from the Pediatric Research in Office Settings network along with subsequent evidence has identified the first 3 weeks of life as the period of highest risk for IBI, with risk declining in a graded fashion aged between 22 and 56 days.
Critical caveats
First, some caveats. Infants 0-7 days are not addressed in the CPG, and all should be treated as high risk and receive full IBI evaluation according to newborn protocols. Second, the recommendations apply only to “well-appearing” infants. Any ill-appearing infant should be treated as high risk and receive full IBI evaluation and begun on empirical antimicrobials. Third, even though the CPG deals with infants as young as 8-21 days old, the recommendations are to treat all infants in this age group as high risk, even if well-appearing, and complete full IBI evaluation and empirical therapy while awaiting results. Fourth, these guidelines apply only to infants born at 37 weeks’ gestation or more. Finally, the new CPG action statements are meant to be recommendations rather than a standard of medical care, leaving some leeway for clinician interpretation of individual patient scenarios. Where appropriate, parents’ values and preferences should be incorporated as part of shared decision-making.
The CPG divides young, febrile infants into three cohorts based on age:
- 8-21 days old
- 22-28 days old
- 29-60 days old
Age 8-21 days
For well-appearing febrile infants 8-21 days old, the CPG recommends a complete IBI evaluation that includes urine, blood, and cerebrospinal fluid (CSF) for culture, approaching all infants in this cohort as high risk. Inflammatory markers may be obtained, but the evidence is not comprehensive enough to evaluate their role in decision-making for this age group. A two-step urine evaluation method (urine analysis followed by culture if the urine analysis looks concerning) is not recommended for infants aged 8-21 days. Urine samples for culture from these young infants should be obtained by catheterization or suprapubic aspiration.
The CPG recommends drawing blood cultures and CSF by lumbar puncture from this cohort. These infants should be admitted to the hospital, treated empirically with antimicrobials, and actively monitored. However, if the cultures are negative at 24-36 hours, the clinician should discontinue antimicrobials and discharge the infant if there is no other reason for continued hospitalization.
Age 22-28 days
Well-appearing, febrile infants 22-28 days old are in an intermediate-risk zone. The recommendation for infants in this cohort is to obtain a urine specimen by catheterization or suprapubic aspiration for both urine analysis and culture. Clinicians may consider obtaining urine samples for analysis noninvasively (e.g., urine bag) in this cohort, but this is not the preferred method.
Blood culture should be obtained from all infants in this group. Inflammatory markers can help clinicians identify infants at greater risk for IBI, including meningitis. Previous data suggested that inflammatory markers such as serum white blood cell counts greater than 11,000/mcL, a serum absolute neutrophil count of greater than 4,000/mcL, and elevated C-reactive protein and procalcitonin levels could help providers identify febrile infants with true IBI. A 2008 study demonstrated that procalcitonin had the best receiver operating characteristic curve in regard to predicting IBI in young febrile infants. Other research backed up that finding and identified cutoff values for procalcitonin levels greater than 1.0 ng/mL. The CPG recommends considering a procalcitonin value of 0.5 ng/mL or higher as positive, indicating that the infant is at greater risk for IBI and potentially should undergo an expanded IBI workup. Therefore, in infants aged 22-28 days, inflammatory markers can play a role in deciding whether to perform a lumbar puncture.
Many more nuanced recommendations for whether to and how to empirically treat with antimicrobials in this cohort can be found in the CPG, including whether to manage in the hospital or at home. Treatment recommendations vary greatly for this cohort on the basis of the tests obtained and whether tests were positive or negative at the initial evaluation.
Age 29-60 days
The CPG will be most helpful when clinicians are faced with well-appearing, febrile infants in the 29- to 60-day age group. As with the other groups, a urine evaluation is recommended; however, the CPG suggests that the two-step approach – obtaining a urine analysis by a noninvasive method and only obtaining culture if the urine analysis is positive – is reasonable. This means that a bag or free-flowing urine specimen would be appropriate for urinalysis, followed by catheterization/suprapubic aspiration if a culture is necessary. This would save approximately 90% of infants from invasive urine collection. Regardless, only catheter or suprapubic specimens are appropriate for urine culture.
The CPG also recommends that clinicians obtain blood culture on all of these infants. Inflammatory markers should be assessed in this cohort because avoiding lumbar puncture for CSF culture would be appropriate in this cohort if the inflammatory markers are negative. If CSF is obtained in this age cohort, enterovirus testing should be added to the testing regimen. Again, for any infant considered at higher risk for IBI on the basis of screening tests, the CPG recommends a 24- to 36-hour rule-out period with empirical antimicrobial treatment and active monitoring in the hospital.
Summary
The recommended approach for febrile infants 8-21 days old is relatively aggressive, with urine, blood, and CSF evaluation for IBI. Clinicians gain some leeway for infants age 22-28 days, but the guidelines recommend a more flexible approach to evaluating well-appearing, febrile infants age 29-60 days, when a two-step urine evaluation and inflammatory marker assessment can help clinicians and parents have a better discussion about the risk-benefit trade-offs of more aggressive testing and empirical treatment.
The author would like to thank Ken Roberts, MD, for his review and helpful comments on this summary of the CPG highlights. Summary points of the CPG were presented by the writing group at the 2021 Pediatric Academic Societies meeting.
William T. Basco, Jr, MD, MS, is a professor of pediatrics at the Medical University of South Carolina, Charleston, and director of the division of general pediatrics. He is an active health services researcher and has published more than 60 manuscripts in the peer-reviewed literature.
A version of this article first appeared on Medscape.com.
FDA warns of potential mechanical concerns with MAGEC devices
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
MAGEC is a surgical magnetic rod system used to treat early-onset scoliosis (EOS) in children under 10 years of age. The magnetic system can help avoid invasive surgeries, as growth rods can be adjusted with an external remote control. MAGEC is the only FDA-approved pure distraction-based system for EOS and is the most-used technology for EOS treatment in the United States, Aakash Agarwal, PhD, director of research and clinical affairs at Spinal Balance in Swanton, Ohio, said in an interview.
According to the notice, there are reports of endcap separation and O-ring seal failure in the following six MAGEC devices:
- MAGEC Spinal Bracing and Distraction System
- MAGEC 2 Spinal Bracing and Distraction System
- MAGEC System
- MAGEC System Model X Device
- MAGEC System Model X Rod
- MAGEC System Rods
Endcap separation can potentially expose the patient’s tissue to internal components of the device that have not been completely tested for biocompatibility.
In February 2020, NuVasive recalled its MAGEC System Model X rods to address reports of endcap separation issues. The FDA cleared a modified version of the device designed to mitigate these events in July 2020. In April 2021, NuVasive informed providers of potential biocompatibility concerns and placed a voluntary shipping hold on the MAGEC device system. The shipping hold was lifted July 15, the company announced.
The FDA is currently not recommending removal of functioning MAGEC devices, noting that it is “in the best interest of patients” to continue to make the system available. The overall benefits of the device outweigh the known risks, and the restricted use for a 2-year implantation time for children under 10 years of age will further mitigate these risks, the FDA said in the statement.
To report adverse events related to MAGEC devices, patients, caregivers, and providers can submit a report through MedWatch, the FDA safety information and adverse event reporting program.
A version of this article first appeared on Medscape.com.
Does early delivery for FGR affect school outcomes?
Iatrogenic delivery for suspected fetal growth restriction (FGR) may be associated with an increased likelihood of poorer school outcomes among infants born severely small for gestational age, a study of children in Australia suggests.
researchers reported in JAMA.
“It raises the question: in our efforts to improve outcomes in babies that are small, are we potentially doing more harm than good?” said Robert M. Silver, MD, of the department of obstetrics and gynecology at the University of Utah, Salt Lake City, who was not involved in the study. “I think that is a very important question to ask.”
However, “we can’t make that conclusion based on this one study,” he said in an interview. It could be that, in cases where severely small infants were delivered early, there may have been testing that indicated acute risks, and these infants may have tended to be sicker overall. “It may have been that if those babies weren’t delivered, they would have suffered a stillbirth or major brain injury,” Dr. Silver said. “It is really important that we acknowledge that we shouldn’t change our clinical practice” based on this one study.”
At the same time, the study underscores questions and challenges that surround the definition, identification, and management of suspected FGR, Dr. Silver said.
The study authors described their research as exploratory. In a related editorial Dr. Silver and Nathan R. Blue, MD said the findings should be considered hypothesis generating.
For the study, Roshan John Selvaratnam, BMedSc(Hons), a researcher affiliated with Monash University, Melbourne, and colleagues analyzed data from 181,902 children with developmental outcomes and 425,717 children with educational outcomes in Australia. They included children born at 32 weeks’ or more gestation between 2003 and 2013.
Severely small infants delivered early for suspected FGR had an average gestation of 37.9 weeks, whereas those not suspected of having FGR had an average gestation of 39.4 weeks.
Among infants who were severely small for gestational age, those delivered early for suspected FGR were more likely to be in the bottom 10th percentile on at least two developmental domains when they started school, compared with those not suspected of having FGR (16.2% vs. 12.7%; adjusted odds ratio, 1.36). They also were more likely to have low test scores in subsequent years. In grade 7, for example, the adjusted odds ratio for scoring below the national minimum standard on at least two educational domains was 1.33 (13.4% vs. 10.5%).
The researchers defined severely small for gestational age as birth weight below the third percentile. Among infants with normal growth, defined as birth weight at the 10th percentile or greater, school outcomes did not significantly differ between those with early delivery for suspected FGR and those not suspected of having FGR. Approximately 8% of the infants with normal growth had poor developmental outcomes.
The study authors described the dilemma that clinicians face with suspected FGR: “Either intervene early to prevent a small risk of stillbirth but potentially cause immediate and lifelong harm to the child or accept the increasing risk of stillbirth associated with prolonging the pregnancy to avoid more common neonatal and longer-term morbidities.”
It could be that severely small infants with suspected FGR in the study were “more compromised than those not suspected of having FGR,” which might explain the outcomes, Mr. Selvaratnam and coauthors wrote.
Another more plausible explanation is that “iatrogenic prematurity was harmful,” they said.
The researchers were unable to adjust for many factors that may influence academic success, including smoking and alcohol use during pregnancy, maternal body mass index, and breastfeeding, they noted. They also lacked information about the etiology for FGR and whether children had genetic abnormalities.
The study also does not take into account neonatal, infant, and childhood complications, Dr. Silver and Dr. Blue wrote in their editorial. “Nonetheless, these data are a welcome contribution given the knowledge gaps with regard to the optimal obstetric management of FGR.”
The establishment of a diagnostic standard for FGR is needed to properly investigate ways to improve risk stratification, diagnosis, and management, Dr. Silver and Dr. Blue added.
“What we have to do is get better at predicting which babies are at very high risk for continuing the pregnancy and which babies are at low risk for continuing the pregnancy so that we can better decide which babies would benefit from slightly early delivery,” Dr. Silver said.
Improved detection and management of FGR may be on the horizon. “Our ability to image the placental function has gotten a lot better, and I think that is really going to help us,” Dr. Silver said. Studies that aim to further improve the ability to assess whether babies are getting adequate blood flow during pregnancy are ongoing, which could further help doctors evaluate risks.
The study investigators and Dr. Silver had no conflict of interest disclosures. Dr. Blue disclosed grants from Samsung Medison and personal fees from Elsevier. The study was supported by a grant from the Australian government’s National Health and Medical Research Council Program, and Mr. Selvaratnam is supported by scholarships from an Australian government research training program and the National Centre of Research Excellence in Stillbirth.
Iatrogenic delivery for suspected fetal growth restriction (FGR) may be associated with an increased likelihood of poorer school outcomes among infants born severely small for gestational age, a study of children in Australia suggests.
researchers reported in JAMA.
“It raises the question: in our efforts to improve outcomes in babies that are small, are we potentially doing more harm than good?” said Robert M. Silver, MD, of the department of obstetrics and gynecology at the University of Utah, Salt Lake City, who was not involved in the study. “I think that is a very important question to ask.”
However, “we can’t make that conclusion based on this one study,” he said in an interview. It could be that, in cases where severely small infants were delivered early, there may have been testing that indicated acute risks, and these infants may have tended to be sicker overall. “It may have been that if those babies weren’t delivered, they would have suffered a stillbirth or major brain injury,” Dr. Silver said. “It is really important that we acknowledge that we shouldn’t change our clinical practice” based on this one study.”
At the same time, the study underscores questions and challenges that surround the definition, identification, and management of suspected FGR, Dr. Silver said.
The study authors described their research as exploratory. In a related editorial Dr. Silver and Nathan R. Blue, MD said the findings should be considered hypothesis generating.
For the study, Roshan John Selvaratnam, BMedSc(Hons), a researcher affiliated with Monash University, Melbourne, and colleagues analyzed data from 181,902 children with developmental outcomes and 425,717 children with educational outcomes in Australia. They included children born at 32 weeks’ or more gestation between 2003 and 2013.
Severely small infants delivered early for suspected FGR had an average gestation of 37.9 weeks, whereas those not suspected of having FGR had an average gestation of 39.4 weeks.
Among infants who were severely small for gestational age, those delivered early for suspected FGR were more likely to be in the bottom 10th percentile on at least two developmental domains when they started school, compared with those not suspected of having FGR (16.2% vs. 12.7%; adjusted odds ratio, 1.36). They also were more likely to have low test scores in subsequent years. In grade 7, for example, the adjusted odds ratio for scoring below the national minimum standard on at least two educational domains was 1.33 (13.4% vs. 10.5%).
The researchers defined severely small for gestational age as birth weight below the third percentile. Among infants with normal growth, defined as birth weight at the 10th percentile or greater, school outcomes did not significantly differ between those with early delivery for suspected FGR and those not suspected of having FGR. Approximately 8% of the infants with normal growth had poor developmental outcomes.
The study authors described the dilemma that clinicians face with suspected FGR: “Either intervene early to prevent a small risk of stillbirth but potentially cause immediate and lifelong harm to the child or accept the increasing risk of stillbirth associated with prolonging the pregnancy to avoid more common neonatal and longer-term morbidities.”
It could be that severely small infants with suspected FGR in the study were “more compromised than those not suspected of having FGR,” which might explain the outcomes, Mr. Selvaratnam and coauthors wrote.
Another more plausible explanation is that “iatrogenic prematurity was harmful,” they said.
The researchers were unable to adjust for many factors that may influence academic success, including smoking and alcohol use during pregnancy, maternal body mass index, and breastfeeding, they noted. They also lacked information about the etiology for FGR and whether children had genetic abnormalities.
The study also does not take into account neonatal, infant, and childhood complications, Dr. Silver and Dr. Blue wrote in their editorial. “Nonetheless, these data are a welcome contribution given the knowledge gaps with regard to the optimal obstetric management of FGR.”
The establishment of a diagnostic standard for FGR is needed to properly investigate ways to improve risk stratification, diagnosis, and management, Dr. Silver and Dr. Blue added.
“What we have to do is get better at predicting which babies are at very high risk for continuing the pregnancy and which babies are at low risk for continuing the pregnancy so that we can better decide which babies would benefit from slightly early delivery,” Dr. Silver said.
Improved detection and management of FGR may be on the horizon. “Our ability to image the placental function has gotten a lot better, and I think that is really going to help us,” Dr. Silver said. Studies that aim to further improve the ability to assess whether babies are getting adequate blood flow during pregnancy are ongoing, which could further help doctors evaluate risks.
The study investigators and Dr. Silver had no conflict of interest disclosures. Dr. Blue disclosed grants from Samsung Medison and personal fees from Elsevier. The study was supported by a grant from the Australian government’s National Health and Medical Research Council Program, and Mr. Selvaratnam is supported by scholarships from an Australian government research training program and the National Centre of Research Excellence in Stillbirth.
Iatrogenic delivery for suspected fetal growth restriction (FGR) may be associated with an increased likelihood of poorer school outcomes among infants born severely small for gestational age, a study of children in Australia suggests.
researchers reported in JAMA.
“It raises the question: in our efforts to improve outcomes in babies that are small, are we potentially doing more harm than good?” said Robert M. Silver, MD, of the department of obstetrics and gynecology at the University of Utah, Salt Lake City, who was not involved in the study. “I think that is a very important question to ask.”
However, “we can’t make that conclusion based on this one study,” he said in an interview. It could be that, in cases where severely small infants were delivered early, there may have been testing that indicated acute risks, and these infants may have tended to be sicker overall. “It may have been that if those babies weren’t delivered, they would have suffered a stillbirth or major brain injury,” Dr. Silver said. “It is really important that we acknowledge that we shouldn’t change our clinical practice” based on this one study.”
At the same time, the study underscores questions and challenges that surround the definition, identification, and management of suspected FGR, Dr. Silver said.
The study authors described their research as exploratory. In a related editorial Dr. Silver and Nathan R. Blue, MD said the findings should be considered hypothesis generating.
For the study, Roshan John Selvaratnam, BMedSc(Hons), a researcher affiliated with Monash University, Melbourne, and colleagues analyzed data from 181,902 children with developmental outcomes and 425,717 children with educational outcomes in Australia. They included children born at 32 weeks’ or more gestation between 2003 and 2013.
Severely small infants delivered early for suspected FGR had an average gestation of 37.9 weeks, whereas those not suspected of having FGR had an average gestation of 39.4 weeks.
Among infants who were severely small for gestational age, those delivered early for suspected FGR were more likely to be in the bottom 10th percentile on at least two developmental domains when they started school, compared with those not suspected of having FGR (16.2% vs. 12.7%; adjusted odds ratio, 1.36). They also were more likely to have low test scores in subsequent years. In grade 7, for example, the adjusted odds ratio for scoring below the national minimum standard on at least two educational domains was 1.33 (13.4% vs. 10.5%).
The researchers defined severely small for gestational age as birth weight below the third percentile. Among infants with normal growth, defined as birth weight at the 10th percentile or greater, school outcomes did not significantly differ between those with early delivery for suspected FGR and those not suspected of having FGR. Approximately 8% of the infants with normal growth had poor developmental outcomes.
The study authors described the dilemma that clinicians face with suspected FGR: “Either intervene early to prevent a small risk of stillbirth but potentially cause immediate and lifelong harm to the child or accept the increasing risk of stillbirth associated with prolonging the pregnancy to avoid more common neonatal and longer-term morbidities.”
It could be that severely small infants with suspected FGR in the study were “more compromised than those not suspected of having FGR,” which might explain the outcomes, Mr. Selvaratnam and coauthors wrote.
Another more plausible explanation is that “iatrogenic prematurity was harmful,” they said.
The researchers were unable to adjust for many factors that may influence academic success, including smoking and alcohol use during pregnancy, maternal body mass index, and breastfeeding, they noted. They also lacked information about the etiology for FGR and whether children had genetic abnormalities.
The study also does not take into account neonatal, infant, and childhood complications, Dr. Silver and Dr. Blue wrote in their editorial. “Nonetheless, these data are a welcome contribution given the knowledge gaps with regard to the optimal obstetric management of FGR.”
The establishment of a diagnostic standard for FGR is needed to properly investigate ways to improve risk stratification, diagnosis, and management, Dr. Silver and Dr. Blue added.
“What we have to do is get better at predicting which babies are at very high risk for continuing the pregnancy and which babies are at low risk for continuing the pregnancy so that we can better decide which babies would benefit from slightly early delivery,” Dr. Silver said.
Improved detection and management of FGR may be on the horizon. “Our ability to image the placental function has gotten a lot better, and I think that is really going to help us,” Dr. Silver said. Studies that aim to further improve the ability to assess whether babies are getting adequate blood flow during pregnancy are ongoing, which could further help doctors evaluate risks.
The study investigators and Dr. Silver had no conflict of interest disclosures. Dr. Blue disclosed grants from Samsung Medison and personal fees from Elsevier. The study was supported by a grant from the Australian government’s National Health and Medical Research Council Program, and Mr. Selvaratnam is supported by scholarships from an Australian government research training program and the National Centre of Research Excellence in Stillbirth.
FROM JAMA
When patients demand vaccinated health care providers
Should a hospital or medical practice fulfill a patient’s request to be treated or cared for only by vaccinated health care providers?The answer is yes, in a perfect world. Patients should feel assured that their health care providers – clinicians and caregivers – are not exposing them to infectious diseases.But issues are being raised – subquestions that need to be answered to understand the current situation and assist health care employers in their decision-making:
- Must health care employers ensure that their employees are vaccinated?
- Can health care employers require that their employees be vaccinated?
- Do employees have any rights to refuse vaccination or to refuse to supply their employer with their vaccination status?
- Can a health care employer terminate an employee who refuses vaccination?
- Does a patient have a legal right to a vaccinated health care provider?
At present, federal policy says that employers may, but are not required to, insist that employees be vaccinated. The currently prevailing state case law says that hospitals and other employers can require staff to be vaccinated and can terminate employees who refuse vaccination. In June, a Texas court dismissed a case in which 117 employees sued a hospital for requiring that employees be vaccinated. More cases are pending in other states, and there may be differing decisions in other states and on appeal.
State laws enacted years ago also weigh in on employer obligations. In at least one state, Oregon, employers of health care providers may not require vaccination, even though other employers may. Other states have laws about what an employer may or may not require of an employee regarding vaccination, and some have introduced laws which are pending.
So, in most states, health care employers may, not must, require that employees be vaccinated. In most states, hospitals and medical practices may terminate employees who refuse vaccination. However, employers should research the laws of their own states before requiring vaccinations and before terminating employees who are not vaccinated.
The issue of employer mandates is complicated further by the practicality that, in some areas of the country, health care providers are in scarce supply. Employers don’t want to lose the providers they have.
And there are additional questions about how certain federal laws affect the situation. Federal law that may apply includes:
- U.S. Food and Drug Administration regulation on approval of vaccines
- The Americans With Disabilities Act (ADA)
- The Health Insurance Portability and Accountability Act of 1996, which protects sensitive patient health information from being disclosed without the patient’s consent
- Civil rights laws
- Patients’ rights
FDA. Some health care providers who refuse vaccination argue that employers have no legal right to require a vaccine that is not fully approved by the FDA. COVID-19 vaccinations have emergency use authorization – something less than full approval. Courts have not yet ruled on this issue.
ADA. Some attorneys believe that honoring a patient’s request to be attended only by a vaccinated health care provider can implicate the ADA. However, the ADA doesn’t protect healthy individuals who don’t want to be vaccinated. The ADA protects the person who, because of their disability, shouldn’t get the vaccination. If an employer mandates vaccination, the employer must, under the ADA, consider requests for exemptions from disabled individuals. However, even when an employee has a disability that may qualify the employee for an exemption to the vaccination requirement, an employer may argue that giving an exemption would be a direct threat to the safety of others; in that case, the ADA may require that the disabled employee and hospital work something out. A compromise might be that the unvaccinated disabled individual would not provide direct patient care or would wear a mask and maintain physical distance.
HIPAA. Some argue that federal privacy law enters into the discussion, maintaining that health care employers can’t disclose employees’ vaccination status under HIPAA. That is not true. Employers are not “covered entities” under HIPAA. It is health care providers who are precluded under HIPAA from disclosing a patient’s personal information. So, if an employer were to ask an employee’s health care provider about the employee’s vaccination status, the health care provider could disclose that status only if the employee consented to the disclosure. An employer may ask an employee for the employee’s proof of vaccination card. However, employers must not ask for unnecessary details that might reveal disability information protected by the ADA.
Civil rights law. Civil rights laws may protect certain individuals from employment consequences of refusing vaccination. Specifically, individuals with sincerely held religious convictions against vaccinations are protected from retaliation by employers for refusing vaccination, under the Constitutional right of freedom of religion. The individual without sincerely held religious convictions against vaccinations and without a relevant disability doesn’t have legal remedies under civil rights laws.
Civil rights laws may apply if employers don’t apply their vaccination requirements to all employees equally. That is, employers can’t require vaccinations of some employees but not others.
Patients’ rights. Legal protections for patients who want a vaccinated health care provider are nowhere to be seen, at this time. It is unlikely that a single patient will be able to convince a hospital or medical practice to require that its staff be vaccinated. However, if a patient becomes infected with COVID-19 and can prove that the illness is causally related to interacting with an unvaccinated health care worker, the patient may have a case against the employer. The legal theory would be malpractice or negligence under informed consent law: That is, the patient did not consent to be treated by an unvaccinated person.
Employer options
So, what can health care employers do? They have three options:
- Require vaccination of all employees, independent contractors, and other providers who have privileges to see patients. Then, as long as the employer enforces the vaccination mandate, the employer can tell patients that all providers are vaccinated.
- Not require that employees and others with access to patients be vaccinated, and if a patient requests to be seen only by vaccinated providers, provide that patient with a vaccinated provider. It is especially important that health care employers take care with patients who are unvaccinated and who have been advised not to be vaccinated because of a medical condition. Both the patient and the health care employer would be protected best by avoiding having two unvaccinated individuals interact. Masks and physical distancing may decrease the risk.
- Not require that employees be vaccinated and refuse to guarantee that providers are vaccinated. To avoid risk for future lawsuits, employers should inform patients that there is no assurance that providers are vaccinated. That leaves it to each patient to ask individual providers about the provider’s vaccination status. If a patient doesn’t like a provider’s answer, then the patient has the right to leave. It’s not clear that the patient has a legal right to stay and demand a vaccinated provider.
Option three is problematic for a number of reasons. Patients aren’t always in a position to query each provider who enters the room about vaccination status. Patients may be sedated or too ill to exert that effort. And it puts supervisors in the position of having to mediate situations where a patient wants to leave against medical advice but the option of staying may also be dangerous.
Health care employers should discuss the options with their legal counsel before deciding which option to adopt.
A version of this article first appeared on Medscape.com.
Should a hospital or medical practice fulfill a patient’s request to be treated or cared for only by vaccinated health care providers?The answer is yes, in a perfect world. Patients should feel assured that their health care providers – clinicians and caregivers – are not exposing them to infectious diseases.But issues are being raised – subquestions that need to be answered to understand the current situation and assist health care employers in their decision-making:
- Must health care employers ensure that their employees are vaccinated?
- Can health care employers require that their employees be vaccinated?
- Do employees have any rights to refuse vaccination or to refuse to supply their employer with their vaccination status?
- Can a health care employer terminate an employee who refuses vaccination?
- Does a patient have a legal right to a vaccinated health care provider?
At present, federal policy says that employers may, but are not required to, insist that employees be vaccinated. The currently prevailing state case law says that hospitals and other employers can require staff to be vaccinated and can terminate employees who refuse vaccination. In June, a Texas court dismissed a case in which 117 employees sued a hospital for requiring that employees be vaccinated. More cases are pending in other states, and there may be differing decisions in other states and on appeal.
State laws enacted years ago also weigh in on employer obligations. In at least one state, Oregon, employers of health care providers may not require vaccination, even though other employers may. Other states have laws about what an employer may or may not require of an employee regarding vaccination, and some have introduced laws which are pending.
So, in most states, health care employers may, not must, require that employees be vaccinated. In most states, hospitals and medical practices may terminate employees who refuse vaccination. However, employers should research the laws of their own states before requiring vaccinations and before terminating employees who are not vaccinated.
The issue of employer mandates is complicated further by the practicality that, in some areas of the country, health care providers are in scarce supply. Employers don’t want to lose the providers they have.
And there are additional questions about how certain federal laws affect the situation. Federal law that may apply includes:
- U.S. Food and Drug Administration regulation on approval of vaccines
- The Americans With Disabilities Act (ADA)
- The Health Insurance Portability and Accountability Act of 1996, which protects sensitive patient health information from being disclosed without the patient’s consent
- Civil rights laws
- Patients’ rights
FDA. Some health care providers who refuse vaccination argue that employers have no legal right to require a vaccine that is not fully approved by the FDA. COVID-19 vaccinations have emergency use authorization – something less than full approval. Courts have not yet ruled on this issue.
ADA. Some attorneys believe that honoring a patient’s request to be attended only by a vaccinated health care provider can implicate the ADA. However, the ADA doesn’t protect healthy individuals who don’t want to be vaccinated. The ADA protects the person who, because of their disability, shouldn’t get the vaccination. If an employer mandates vaccination, the employer must, under the ADA, consider requests for exemptions from disabled individuals. However, even when an employee has a disability that may qualify the employee for an exemption to the vaccination requirement, an employer may argue that giving an exemption would be a direct threat to the safety of others; in that case, the ADA may require that the disabled employee and hospital work something out. A compromise might be that the unvaccinated disabled individual would not provide direct patient care or would wear a mask and maintain physical distance.
HIPAA. Some argue that federal privacy law enters into the discussion, maintaining that health care employers can’t disclose employees’ vaccination status under HIPAA. That is not true. Employers are not “covered entities” under HIPAA. It is health care providers who are precluded under HIPAA from disclosing a patient’s personal information. So, if an employer were to ask an employee’s health care provider about the employee’s vaccination status, the health care provider could disclose that status only if the employee consented to the disclosure. An employer may ask an employee for the employee’s proof of vaccination card. However, employers must not ask for unnecessary details that might reveal disability information protected by the ADA.
Civil rights law. Civil rights laws may protect certain individuals from employment consequences of refusing vaccination. Specifically, individuals with sincerely held religious convictions against vaccinations are protected from retaliation by employers for refusing vaccination, under the Constitutional right of freedom of religion. The individual without sincerely held religious convictions against vaccinations and without a relevant disability doesn’t have legal remedies under civil rights laws.
Civil rights laws may apply if employers don’t apply their vaccination requirements to all employees equally. That is, employers can’t require vaccinations of some employees but not others.
Patients’ rights. Legal protections for patients who want a vaccinated health care provider are nowhere to be seen, at this time. It is unlikely that a single patient will be able to convince a hospital or medical practice to require that its staff be vaccinated. However, if a patient becomes infected with COVID-19 and can prove that the illness is causally related to interacting with an unvaccinated health care worker, the patient may have a case against the employer. The legal theory would be malpractice or negligence under informed consent law: That is, the patient did not consent to be treated by an unvaccinated person.
Employer options
So, what can health care employers do? They have three options:
- Require vaccination of all employees, independent contractors, and other providers who have privileges to see patients. Then, as long as the employer enforces the vaccination mandate, the employer can tell patients that all providers are vaccinated.
- Not require that employees and others with access to patients be vaccinated, and if a patient requests to be seen only by vaccinated providers, provide that patient with a vaccinated provider. It is especially important that health care employers take care with patients who are unvaccinated and who have been advised not to be vaccinated because of a medical condition. Both the patient and the health care employer would be protected best by avoiding having two unvaccinated individuals interact. Masks and physical distancing may decrease the risk.
- Not require that employees be vaccinated and refuse to guarantee that providers are vaccinated. To avoid risk for future lawsuits, employers should inform patients that there is no assurance that providers are vaccinated. That leaves it to each patient to ask individual providers about the provider’s vaccination status. If a patient doesn’t like a provider’s answer, then the patient has the right to leave. It’s not clear that the patient has a legal right to stay and demand a vaccinated provider.
Option three is problematic for a number of reasons. Patients aren’t always in a position to query each provider who enters the room about vaccination status. Patients may be sedated or too ill to exert that effort. And it puts supervisors in the position of having to mediate situations where a patient wants to leave against medical advice but the option of staying may also be dangerous.
Health care employers should discuss the options with their legal counsel before deciding which option to adopt.
A version of this article first appeared on Medscape.com.
Should a hospital or medical practice fulfill a patient’s request to be treated or cared for only by vaccinated health care providers?The answer is yes, in a perfect world. Patients should feel assured that their health care providers – clinicians and caregivers – are not exposing them to infectious diseases.But issues are being raised – subquestions that need to be answered to understand the current situation and assist health care employers in their decision-making:
- Must health care employers ensure that their employees are vaccinated?
- Can health care employers require that their employees be vaccinated?
- Do employees have any rights to refuse vaccination or to refuse to supply their employer with their vaccination status?
- Can a health care employer terminate an employee who refuses vaccination?
- Does a patient have a legal right to a vaccinated health care provider?
At present, federal policy says that employers may, but are not required to, insist that employees be vaccinated. The currently prevailing state case law says that hospitals and other employers can require staff to be vaccinated and can terminate employees who refuse vaccination. In June, a Texas court dismissed a case in which 117 employees sued a hospital for requiring that employees be vaccinated. More cases are pending in other states, and there may be differing decisions in other states and on appeal.
State laws enacted years ago also weigh in on employer obligations. In at least one state, Oregon, employers of health care providers may not require vaccination, even though other employers may. Other states have laws about what an employer may or may not require of an employee regarding vaccination, and some have introduced laws which are pending.
So, in most states, health care employers may, not must, require that employees be vaccinated. In most states, hospitals and medical practices may terminate employees who refuse vaccination. However, employers should research the laws of their own states before requiring vaccinations and before terminating employees who are not vaccinated.
The issue of employer mandates is complicated further by the practicality that, in some areas of the country, health care providers are in scarce supply. Employers don’t want to lose the providers they have.
And there are additional questions about how certain federal laws affect the situation. Federal law that may apply includes:
- U.S. Food and Drug Administration regulation on approval of vaccines
- The Americans With Disabilities Act (ADA)
- The Health Insurance Portability and Accountability Act of 1996, which protects sensitive patient health information from being disclosed without the patient’s consent
- Civil rights laws
- Patients’ rights
FDA. Some health care providers who refuse vaccination argue that employers have no legal right to require a vaccine that is not fully approved by the FDA. COVID-19 vaccinations have emergency use authorization – something less than full approval. Courts have not yet ruled on this issue.
ADA. Some attorneys believe that honoring a patient’s request to be attended only by a vaccinated health care provider can implicate the ADA. However, the ADA doesn’t protect healthy individuals who don’t want to be vaccinated. The ADA protects the person who, because of their disability, shouldn’t get the vaccination. If an employer mandates vaccination, the employer must, under the ADA, consider requests for exemptions from disabled individuals. However, even when an employee has a disability that may qualify the employee for an exemption to the vaccination requirement, an employer may argue that giving an exemption would be a direct threat to the safety of others; in that case, the ADA may require that the disabled employee and hospital work something out. A compromise might be that the unvaccinated disabled individual would not provide direct patient care or would wear a mask and maintain physical distance.
HIPAA. Some argue that federal privacy law enters into the discussion, maintaining that health care employers can’t disclose employees’ vaccination status under HIPAA. That is not true. Employers are not “covered entities” under HIPAA. It is health care providers who are precluded under HIPAA from disclosing a patient’s personal information. So, if an employer were to ask an employee’s health care provider about the employee’s vaccination status, the health care provider could disclose that status only if the employee consented to the disclosure. An employer may ask an employee for the employee’s proof of vaccination card. However, employers must not ask for unnecessary details that might reveal disability information protected by the ADA.
Civil rights law. Civil rights laws may protect certain individuals from employment consequences of refusing vaccination. Specifically, individuals with sincerely held religious convictions against vaccinations are protected from retaliation by employers for refusing vaccination, under the Constitutional right of freedom of religion. The individual without sincerely held religious convictions against vaccinations and without a relevant disability doesn’t have legal remedies under civil rights laws.
Civil rights laws may apply if employers don’t apply their vaccination requirements to all employees equally. That is, employers can’t require vaccinations of some employees but not others.
Patients’ rights. Legal protections for patients who want a vaccinated health care provider are nowhere to be seen, at this time. It is unlikely that a single patient will be able to convince a hospital or medical practice to require that its staff be vaccinated. However, if a patient becomes infected with COVID-19 and can prove that the illness is causally related to interacting with an unvaccinated health care worker, the patient may have a case against the employer. The legal theory would be malpractice or negligence under informed consent law: That is, the patient did not consent to be treated by an unvaccinated person.
Employer options
So, what can health care employers do? They have three options:
- Require vaccination of all employees, independent contractors, and other providers who have privileges to see patients. Then, as long as the employer enforces the vaccination mandate, the employer can tell patients that all providers are vaccinated.
- Not require that employees and others with access to patients be vaccinated, and if a patient requests to be seen only by vaccinated providers, provide that patient with a vaccinated provider. It is especially important that health care employers take care with patients who are unvaccinated and who have been advised not to be vaccinated because of a medical condition. Both the patient and the health care employer would be protected best by avoiding having two unvaccinated individuals interact. Masks and physical distancing may decrease the risk.
- Not require that employees be vaccinated and refuse to guarantee that providers are vaccinated. To avoid risk for future lawsuits, employers should inform patients that there is no assurance that providers are vaccinated. That leaves it to each patient to ask individual providers about the provider’s vaccination status. If a patient doesn’t like a provider’s answer, then the patient has the right to leave. It’s not clear that the patient has a legal right to stay and demand a vaccinated provider.
Option three is problematic for a number of reasons. Patients aren’t always in a position to query each provider who enters the room about vaccination status. Patients may be sedated or too ill to exert that effort. And it puts supervisors in the position of having to mediate situations where a patient wants to leave against medical advice but the option of staying may also be dangerous.
Health care employers should discuss the options with their legal counsel before deciding which option to adopt.
A version of this article first appeared on Medscape.com.






