User login
Eli Lilly Offers Obesity Drug Directly to Consumers
Eli Lilly, maker of the anti-obesity drug Zepbound, announced this week the launch of LillyDirect, a direct-to-patient portal, allowing some patients to obtain its drug for as little as $25 a month.
The move is seen as a major shift in the way these popular medications can reach patients.
For many of the 42 million Americans with obesity, weight loss medications such as Wegovy, Saxenda, and the brand-new Zepbound can be a godsend, helping them lose the excess pounds they’ve struggled with for decades or a lifetime.
But getting these medications has been a struggle for many who are eligible. Shortages of the drugs have been one barrier, and costs of up to $1,300 monthly — the price tag without insurance coverage — are another hurdle.
But 2024 may be a much brighter year, thanks to Lilly’s new portal as well as other developments:
Insurance coverage on private health plans, while still spotty, may be improving. Federal legislators are fighting a 2003 law that forbids Medicare from paying for the medications when prescribed for obesity.
New research found that semaglutide (Wegovy) can reduce the risk of recurrent strokes and heart attacks as well as deaths from cardiovascular events in those with obesity and preexisting cardiovascular disease (or diseases of the heart and blood vessels), a finding experts said should get the attention of health insurers.
The medications, also referred to as GLP-1 agonists, work by activating the receptors of hormones (called glucagon-like peptide 1 and others) that are naturally released after eating. That, in turn, makes you feel more full, leading to weight loss of up to 22% for some. The medications are approved for those with a body mass index (BMI) of 30 or a BMI of 27 with at least one other weight-related health condition such as high blood pressure or high cholesterol. The medicines, injected weekly or more often, are prescribed along with advice about a reduced-calorie diet and increased physical activity.
LillyDirect
Patients can access the obesity medicines through the telehealth platform FORM. Patients reach independent telehealth providers, according to Lilly, who can complement a patient’s current doctor or be an alternative to in-patient care in some cases.
Eli Lilly officials did not respond to requests for comment.
Some obesity experts welcomed the new service. “Any program that improves availability and affordability of these ground-breaking medications is welcome news for our long-suffering patients,” said Louis Aronne, MD, director of the Comprehensive Weight Control Center at Weill Cornell Medicine in New York City, a long-time obesity researcher.
“It’s a great move for Lilly to do,” agreed Caroline Apovian, MD, a professor of medicine at Harvard Medical School and co-director of the Center for Weight Management and Wellness at Brigham & Women’s Hospital in Boston, who is also a veteran obesity specialist. “It is trying to help the accessibility issue and do it responsibly.”
“The bottom line is, there is an overwhelming amount of consumer need and desire for these medications and not enough channels [to provide them],” said Zeev Neuwirth, MD, a former executive at Atrium Health who writes about health care trends. “Eli Lilly is responding to a market need that is out there and quite honestly continuing to grow.”
There are still concerns and questions, Dr. Neuwirth said, “especially since this is to my knowledge the first of its kind in terms of a pharmaceutical manufacturer directly dispensing medication in this nontraditional way.”
He called for transparency between telehealth providers and the pharmaceutical company to rule out any conflicts of interest.
The American College of Physicians, an organization of internal medicine doctors and others, issued a statement expressing concern. Omar T. Atiq, MD, group’s president, said his organization is “concerned by the development of websites that enable patients to order prescription medications directly from the drugmakers. While information on in-person care is available, this direct-to-consumer approach is primarily oriented around the use of telehealth services to prescribe a drug maker’s products.”
The group urged that an established patient-doctor relationship be present, or that care should happen in consultation with a doctor who does have an established relationship (the latter an option offered by Lilly). “These direct-to-consumer services have the potential to leave patients confused and misinformed about medications.”
Heart Attack, Stroke Reduction Benefits
Previous research has found that the GLP-1 medicines such as Ozempic (semaglutide), which the FDA approved to treat diabetes, also reduce the risk of cardiovascular issues such as strokes and heart attacks. Now, new research finds that semaglutide at the Wegovy dose (usually slightly higher than the Ozempic dose for diabetes) also has those benefits in those who don›t have a diabetes diagnosis but do have obesity and cardiovascular disease.
In a clinical trial sponsored by Novo Nordisk, the maker of Wegovy, half of more than 17,000 people with obesity were given semaglutide (Wegovy); the other half got a placebo. Compared to those on the placebo, those who took the Wegovy had a 20% reduction in strokes, heart attacks, and deaths from cardiovascular causes over a 33-month period.
The study results are a “big deal,” Dr. Aronne said. The results make it clear that those with obesity but not diabetes will get the cardiovascular benefits from the treatment as well. While more analysis is necessary, he said the important point is that the study showed that reducing body weight is linked to improvement in critical health outcomes.
As the research evolves, he said, it’s going to be difficult for insurers to deny medications in the face of those findings, which promise reductions in long-term health care costs.
Insurance Coverage
In November, the American Medical Association voted to adopt a policy to urge insurance coverage for evidence-based treatment for obesity, including the new obesity medications.
“No single organization is going to be able to convince insurers and employers to cover this,” Dr. Aronne said. “But I think a prominent organization like the AMA adding their voice to the rising chorus is going to help.”
Coverage of GLP-1 medications could nearly double in 2024, according to a survey of 500 human resources decision-makers released in October by Accolade, a personalized health care advocacy and delivery company. While 25% of respondents said they currently offered coverage when the survey was done in August and September, 43% said they intend to offer coverage in 2024.
In an email, David Allen, a spokesperson for America’s Health Insurance Plans, a health care industry association, said: “Every American deserves affordable coverage and high-quality care, and that includes coverage and care for evidence-based obesity treatments and therapies.”
He said “clinical leaders and other experts at health insurance providers routinely review the evidence for all types of treatments, including treatments for obesity, and offer multiple options to patients — ranging from lifestyle changes and nutrition counseling, to surgical interventions, to prescription drugs.”
Mr. Allen said the evidence that obesity drugs help with weight loss “is still evolving.”
“And some patients are experiencing bad effects related to these drugs such as vomiting and nausea, for example, and the likelihood of gaining the weight back when discontinuing the drugs,” he said.
Others are fighting for Medicare coverage, while some experts contend the costs of that coverage would be overwhelming. A bipartisan bill, the Treat and Reduce Obesity Act of 2023, would allow coverage under Medicare›s prescription drug benefit for drugs used for the treatment of obesity or for weigh loss management for people who are overweight. Some say it›s an uphill climb, citing a Vanderbilt University analysis that found giving just 10% of Medicare-eligible patients the drugs would cost $13.6 billion to more than $26 billion.
However, a white paper from the University of Southern California concluded that the value to society of covering the drugs for Medicare recipients would equal nearly $1 trillion over 10 years, citing savings in hospitalizations and other health care costs.
Comprehensive insurance coverage is needed, Dr. Apovian said. Private insurance plans, Medicare, and Medicaid must all realize the importance of covering what has been now shown to be life-saving drugs, she said.
Broader coverage might also reduce the number of patients getting obesity drugs from unreliable sources, in an effort to save money, and having adverse effects. The FDA warned against counterfeit semaglutide in December.
Long-Term Picture
Research suggests the obesity medications must be taken continuously, at least for most people, to maintain the weight loss. In a study of patients on Zepbound, Dr. Aronne and colleagues found that withdrawing the medication led people to regain weight, while continuing it led to maintaining and even increasing the initial weight loss. While some may be able to use the medications only from time to time, “the majority will have to take these on a chronic basis,” Dr. Aronne said.
Obesity, like high blood pressure and other chronic conditions, needs continuous treatment, Dr. Apovian said. No one would suggest withdrawing blood pressure medications that stabilize blood pressure; the same should be true for the obesity drugs, she said.
Dr. Apovian consults for FORM, the telehealth platform Lilly uses for LillyDirect, and consults for Novo Nordisk, which makes Saxenda and Wegovy. Dr. Aronne is a consultant and investigator for Novo Nordisk, Eli Lilly, and other companies.
A version of this article appeared on WebMD.com.
Eli Lilly, maker of the anti-obesity drug Zepbound, announced this week the launch of LillyDirect, a direct-to-patient portal, allowing some patients to obtain its drug for as little as $25 a month.
The move is seen as a major shift in the way these popular medications can reach patients.
For many of the 42 million Americans with obesity, weight loss medications such as Wegovy, Saxenda, and the brand-new Zepbound can be a godsend, helping them lose the excess pounds they’ve struggled with for decades or a lifetime.
But getting these medications has been a struggle for many who are eligible. Shortages of the drugs have been one barrier, and costs of up to $1,300 monthly — the price tag without insurance coverage — are another hurdle.
But 2024 may be a much brighter year, thanks to Lilly’s new portal as well as other developments:
Insurance coverage on private health plans, while still spotty, may be improving. Federal legislators are fighting a 2003 law that forbids Medicare from paying for the medications when prescribed for obesity.
New research found that semaglutide (Wegovy) can reduce the risk of recurrent strokes and heart attacks as well as deaths from cardiovascular events in those with obesity and preexisting cardiovascular disease (or diseases of the heart and blood vessels), a finding experts said should get the attention of health insurers.
The medications, also referred to as GLP-1 agonists, work by activating the receptors of hormones (called glucagon-like peptide 1 and others) that are naturally released after eating. That, in turn, makes you feel more full, leading to weight loss of up to 22% for some. The medications are approved for those with a body mass index (BMI) of 30 or a BMI of 27 with at least one other weight-related health condition such as high blood pressure or high cholesterol. The medicines, injected weekly or more often, are prescribed along with advice about a reduced-calorie diet and increased physical activity.
LillyDirect
Patients can access the obesity medicines through the telehealth platform FORM. Patients reach independent telehealth providers, according to Lilly, who can complement a patient’s current doctor or be an alternative to in-patient care in some cases.
Eli Lilly officials did not respond to requests for comment.
Some obesity experts welcomed the new service. “Any program that improves availability and affordability of these ground-breaking medications is welcome news for our long-suffering patients,” said Louis Aronne, MD, director of the Comprehensive Weight Control Center at Weill Cornell Medicine in New York City, a long-time obesity researcher.
“It’s a great move for Lilly to do,” agreed Caroline Apovian, MD, a professor of medicine at Harvard Medical School and co-director of the Center for Weight Management and Wellness at Brigham & Women’s Hospital in Boston, who is also a veteran obesity specialist. “It is trying to help the accessibility issue and do it responsibly.”
“The bottom line is, there is an overwhelming amount of consumer need and desire for these medications and not enough channels [to provide them],” said Zeev Neuwirth, MD, a former executive at Atrium Health who writes about health care trends. “Eli Lilly is responding to a market need that is out there and quite honestly continuing to grow.”
There are still concerns and questions, Dr. Neuwirth said, “especially since this is to my knowledge the first of its kind in terms of a pharmaceutical manufacturer directly dispensing medication in this nontraditional way.”
He called for transparency between telehealth providers and the pharmaceutical company to rule out any conflicts of interest.
The American College of Physicians, an organization of internal medicine doctors and others, issued a statement expressing concern. Omar T. Atiq, MD, group’s president, said his organization is “concerned by the development of websites that enable patients to order prescription medications directly from the drugmakers. While information on in-person care is available, this direct-to-consumer approach is primarily oriented around the use of telehealth services to prescribe a drug maker’s products.”
The group urged that an established patient-doctor relationship be present, or that care should happen in consultation with a doctor who does have an established relationship (the latter an option offered by Lilly). “These direct-to-consumer services have the potential to leave patients confused and misinformed about medications.”
Heart Attack, Stroke Reduction Benefits
Previous research has found that the GLP-1 medicines such as Ozempic (semaglutide), which the FDA approved to treat diabetes, also reduce the risk of cardiovascular issues such as strokes and heart attacks. Now, new research finds that semaglutide at the Wegovy dose (usually slightly higher than the Ozempic dose for diabetes) also has those benefits in those who don›t have a diabetes diagnosis but do have obesity and cardiovascular disease.
In a clinical trial sponsored by Novo Nordisk, the maker of Wegovy, half of more than 17,000 people with obesity were given semaglutide (Wegovy); the other half got a placebo. Compared to those on the placebo, those who took the Wegovy had a 20% reduction in strokes, heart attacks, and deaths from cardiovascular causes over a 33-month period.
The study results are a “big deal,” Dr. Aronne said. The results make it clear that those with obesity but not diabetes will get the cardiovascular benefits from the treatment as well. While more analysis is necessary, he said the important point is that the study showed that reducing body weight is linked to improvement in critical health outcomes.
As the research evolves, he said, it’s going to be difficult for insurers to deny medications in the face of those findings, which promise reductions in long-term health care costs.
Insurance Coverage
In November, the American Medical Association voted to adopt a policy to urge insurance coverage for evidence-based treatment for obesity, including the new obesity medications.
“No single organization is going to be able to convince insurers and employers to cover this,” Dr. Aronne said. “But I think a prominent organization like the AMA adding their voice to the rising chorus is going to help.”
Coverage of GLP-1 medications could nearly double in 2024, according to a survey of 500 human resources decision-makers released in October by Accolade, a personalized health care advocacy and delivery company. While 25% of respondents said they currently offered coverage when the survey was done in August and September, 43% said they intend to offer coverage in 2024.
In an email, David Allen, a spokesperson for America’s Health Insurance Plans, a health care industry association, said: “Every American deserves affordable coverage and high-quality care, and that includes coverage and care for evidence-based obesity treatments and therapies.”
He said “clinical leaders and other experts at health insurance providers routinely review the evidence for all types of treatments, including treatments for obesity, and offer multiple options to patients — ranging from lifestyle changes and nutrition counseling, to surgical interventions, to prescription drugs.”
Mr. Allen said the evidence that obesity drugs help with weight loss “is still evolving.”
“And some patients are experiencing bad effects related to these drugs such as vomiting and nausea, for example, and the likelihood of gaining the weight back when discontinuing the drugs,” he said.
Others are fighting for Medicare coverage, while some experts contend the costs of that coverage would be overwhelming. A bipartisan bill, the Treat and Reduce Obesity Act of 2023, would allow coverage under Medicare›s prescription drug benefit for drugs used for the treatment of obesity or for weigh loss management for people who are overweight. Some say it›s an uphill climb, citing a Vanderbilt University analysis that found giving just 10% of Medicare-eligible patients the drugs would cost $13.6 billion to more than $26 billion.
However, a white paper from the University of Southern California concluded that the value to society of covering the drugs for Medicare recipients would equal nearly $1 trillion over 10 years, citing savings in hospitalizations and other health care costs.
Comprehensive insurance coverage is needed, Dr. Apovian said. Private insurance plans, Medicare, and Medicaid must all realize the importance of covering what has been now shown to be life-saving drugs, she said.
Broader coverage might also reduce the number of patients getting obesity drugs from unreliable sources, in an effort to save money, and having adverse effects. The FDA warned against counterfeit semaglutide in December.
Long-Term Picture
Research suggests the obesity medications must be taken continuously, at least for most people, to maintain the weight loss. In a study of patients on Zepbound, Dr. Aronne and colleagues found that withdrawing the medication led people to regain weight, while continuing it led to maintaining and even increasing the initial weight loss. While some may be able to use the medications only from time to time, “the majority will have to take these on a chronic basis,” Dr. Aronne said.
Obesity, like high blood pressure and other chronic conditions, needs continuous treatment, Dr. Apovian said. No one would suggest withdrawing blood pressure medications that stabilize blood pressure; the same should be true for the obesity drugs, she said.
Dr. Apovian consults for FORM, the telehealth platform Lilly uses for LillyDirect, and consults for Novo Nordisk, which makes Saxenda and Wegovy. Dr. Aronne is a consultant and investigator for Novo Nordisk, Eli Lilly, and other companies.
A version of this article appeared on WebMD.com.
Eli Lilly, maker of the anti-obesity drug Zepbound, announced this week the launch of LillyDirect, a direct-to-patient portal, allowing some patients to obtain its drug for as little as $25 a month.
The move is seen as a major shift in the way these popular medications can reach patients.
For many of the 42 million Americans with obesity, weight loss medications such as Wegovy, Saxenda, and the brand-new Zepbound can be a godsend, helping them lose the excess pounds they’ve struggled with for decades or a lifetime.
But getting these medications has been a struggle for many who are eligible. Shortages of the drugs have been one barrier, and costs of up to $1,300 monthly — the price tag without insurance coverage — are another hurdle.
But 2024 may be a much brighter year, thanks to Lilly’s new portal as well as other developments:
Insurance coverage on private health plans, while still spotty, may be improving. Federal legislators are fighting a 2003 law that forbids Medicare from paying for the medications when prescribed for obesity.
New research found that semaglutide (Wegovy) can reduce the risk of recurrent strokes and heart attacks as well as deaths from cardiovascular events in those with obesity and preexisting cardiovascular disease (or diseases of the heart and blood vessels), a finding experts said should get the attention of health insurers.
The medications, also referred to as GLP-1 agonists, work by activating the receptors of hormones (called glucagon-like peptide 1 and others) that are naturally released after eating. That, in turn, makes you feel more full, leading to weight loss of up to 22% for some. The medications are approved for those with a body mass index (BMI) of 30 or a BMI of 27 with at least one other weight-related health condition such as high blood pressure or high cholesterol. The medicines, injected weekly or more often, are prescribed along with advice about a reduced-calorie diet and increased physical activity.
LillyDirect
Patients can access the obesity medicines through the telehealth platform FORM. Patients reach independent telehealth providers, according to Lilly, who can complement a patient’s current doctor or be an alternative to in-patient care in some cases.
Eli Lilly officials did not respond to requests for comment.
Some obesity experts welcomed the new service. “Any program that improves availability and affordability of these ground-breaking medications is welcome news for our long-suffering patients,” said Louis Aronne, MD, director of the Comprehensive Weight Control Center at Weill Cornell Medicine in New York City, a long-time obesity researcher.
“It’s a great move for Lilly to do,” agreed Caroline Apovian, MD, a professor of medicine at Harvard Medical School and co-director of the Center for Weight Management and Wellness at Brigham & Women’s Hospital in Boston, who is also a veteran obesity specialist. “It is trying to help the accessibility issue and do it responsibly.”
“The bottom line is, there is an overwhelming amount of consumer need and desire for these medications and not enough channels [to provide them],” said Zeev Neuwirth, MD, a former executive at Atrium Health who writes about health care trends. “Eli Lilly is responding to a market need that is out there and quite honestly continuing to grow.”
There are still concerns and questions, Dr. Neuwirth said, “especially since this is to my knowledge the first of its kind in terms of a pharmaceutical manufacturer directly dispensing medication in this nontraditional way.”
He called for transparency between telehealth providers and the pharmaceutical company to rule out any conflicts of interest.
The American College of Physicians, an organization of internal medicine doctors and others, issued a statement expressing concern. Omar T. Atiq, MD, group’s president, said his organization is “concerned by the development of websites that enable patients to order prescription medications directly from the drugmakers. While information on in-person care is available, this direct-to-consumer approach is primarily oriented around the use of telehealth services to prescribe a drug maker’s products.”
The group urged that an established patient-doctor relationship be present, or that care should happen in consultation with a doctor who does have an established relationship (the latter an option offered by Lilly). “These direct-to-consumer services have the potential to leave patients confused and misinformed about medications.”
Heart Attack, Stroke Reduction Benefits
Previous research has found that the GLP-1 medicines such as Ozempic (semaglutide), which the FDA approved to treat diabetes, also reduce the risk of cardiovascular issues such as strokes and heart attacks. Now, new research finds that semaglutide at the Wegovy dose (usually slightly higher than the Ozempic dose for diabetes) also has those benefits in those who don›t have a diabetes diagnosis but do have obesity and cardiovascular disease.
In a clinical trial sponsored by Novo Nordisk, the maker of Wegovy, half of more than 17,000 people with obesity were given semaglutide (Wegovy); the other half got a placebo. Compared to those on the placebo, those who took the Wegovy had a 20% reduction in strokes, heart attacks, and deaths from cardiovascular causes over a 33-month period.
The study results are a “big deal,” Dr. Aronne said. The results make it clear that those with obesity but not diabetes will get the cardiovascular benefits from the treatment as well. While more analysis is necessary, he said the important point is that the study showed that reducing body weight is linked to improvement in critical health outcomes.
As the research evolves, he said, it’s going to be difficult for insurers to deny medications in the face of those findings, which promise reductions in long-term health care costs.
Insurance Coverage
In November, the American Medical Association voted to adopt a policy to urge insurance coverage for evidence-based treatment for obesity, including the new obesity medications.
“No single organization is going to be able to convince insurers and employers to cover this,” Dr. Aronne said. “But I think a prominent organization like the AMA adding their voice to the rising chorus is going to help.”
Coverage of GLP-1 medications could nearly double in 2024, according to a survey of 500 human resources decision-makers released in October by Accolade, a personalized health care advocacy and delivery company. While 25% of respondents said they currently offered coverage when the survey was done in August and September, 43% said they intend to offer coverage in 2024.
In an email, David Allen, a spokesperson for America’s Health Insurance Plans, a health care industry association, said: “Every American deserves affordable coverage and high-quality care, and that includes coverage and care for evidence-based obesity treatments and therapies.”
He said “clinical leaders and other experts at health insurance providers routinely review the evidence for all types of treatments, including treatments for obesity, and offer multiple options to patients — ranging from lifestyle changes and nutrition counseling, to surgical interventions, to prescription drugs.”
Mr. Allen said the evidence that obesity drugs help with weight loss “is still evolving.”
“And some patients are experiencing bad effects related to these drugs such as vomiting and nausea, for example, and the likelihood of gaining the weight back when discontinuing the drugs,” he said.
Others are fighting for Medicare coverage, while some experts contend the costs of that coverage would be overwhelming. A bipartisan bill, the Treat and Reduce Obesity Act of 2023, would allow coverage under Medicare›s prescription drug benefit for drugs used for the treatment of obesity or for weigh loss management for people who are overweight. Some say it›s an uphill climb, citing a Vanderbilt University analysis that found giving just 10% of Medicare-eligible patients the drugs would cost $13.6 billion to more than $26 billion.
However, a white paper from the University of Southern California concluded that the value to society of covering the drugs for Medicare recipients would equal nearly $1 trillion over 10 years, citing savings in hospitalizations and other health care costs.
Comprehensive insurance coverage is needed, Dr. Apovian said. Private insurance plans, Medicare, and Medicaid must all realize the importance of covering what has been now shown to be life-saving drugs, she said.
Broader coverage might also reduce the number of patients getting obesity drugs from unreliable sources, in an effort to save money, and having adverse effects. The FDA warned against counterfeit semaglutide in December.
Long-Term Picture
Research suggests the obesity medications must be taken continuously, at least for most people, to maintain the weight loss. In a study of patients on Zepbound, Dr. Aronne and colleagues found that withdrawing the medication led people to regain weight, while continuing it led to maintaining and even increasing the initial weight loss. While some may be able to use the medications only from time to time, “the majority will have to take these on a chronic basis,” Dr. Aronne said.
Obesity, like high blood pressure and other chronic conditions, needs continuous treatment, Dr. Apovian said. No one would suggest withdrawing blood pressure medications that stabilize blood pressure; the same should be true for the obesity drugs, she said.
Dr. Apovian consults for FORM, the telehealth platform Lilly uses for LillyDirect, and consults for Novo Nordisk, which makes Saxenda and Wegovy. Dr. Aronne is a consultant and investigator for Novo Nordisk, Eli Lilly, and other companies.
A version of this article appeared on WebMD.com.
Do Statins Offset Venous Thrombosis Risk With Hormone Therapy?
This transcript has been edited for clarity.
This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. I’d like to talk with you about a recent report in JAMA Network Open on the subject of whether statin therapy may be able to offset some of the excess risk for venous thromboembolism (VTE) among women taking menopausal hormone therapy.
It’s an important issue because we know that menopausal hormone therapy, especially oral therapy, is linked to an excess risk for VTE, approximately doubling of risk in the randomized clinical trials. There is also emerging evidence from some randomized trials, such as the Jupiter trial, that step therapy may be linked to a reduction in risk. This may be related to anti-inflammatory or antithrombotic effects of statin therapy.
The authors made use of a very large administrative claims database, Optum Health, to look at more than 15 million annual members. They were able to identify 2000 women with a diagnostic code for VTE treatment. The women were between ages 50 and 64 years, and they were compared with 200,000 controls without VTE, matched in 10-to-1 fashion.
About 50% of the women were taking oral hormone therapy, and about 50% took non-oral transdermal or other non-oral formulations of hormone therapy. The odds ratio for VTE was 1.53 among the women who did not also have prescription records for statin therapy. They were able to look at prescribed prescriptions for both the hormone therapy and the statins. Among the women prescribed hormone therapy and also low- to intermediate-dose statins, the odds ratio was 1.29. So that was quite a mitigation of the elevated risk. Among the women taking high-intensity statins, the odds ratio was 1.06, and there was no significant elevation.
We do need more data and more research on this question. One approach would be a meta-analysis of all of the existing randomized trials of hormone therapy in recent years wherein there was increased uptake of statin therapy to look at this question not only for VTE but also for coronary heart disease, stroke, and other CVD outcomes to see whether statin therapy is associated with some attenuation of the excess risk. We also need a targeted randomized trial of statins vs placebo among women who have clear indications for hormone therapy but may be at some increased risk for VTE. That type of trial would be extremely helpful.
These include choosing a transdermal rather than an oral formulation of hormone therapy and using lower doses of hormone therapy. Also, women who are clear candidates for hormone therapy and also for statins, it’s obvious that statins could be co-prescribed. Even among women who are clear candidates for hormone therapy but only intermediate borderline candidates for statin therapy, the prescription of statins might be considered in that clinical scenario to try to mitigate that excess risk for VTE.
JoAnn E. Manson, MD, DrPH, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from: Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. I’d like to talk with you about a recent report in JAMA Network Open on the subject of whether statin therapy may be able to offset some of the excess risk for venous thromboembolism (VTE) among women taking menopausal hormone therapy.
It’s an important issue because we know that menopausal hormone therapy, especially oral therapy, is linked to an excess risk for VTE, approximately doubling of risk in the randomized clinical trials. There is also emerging evidence from some randomized trials, such as the Jupiter trial, that step therapy may be linked to a reduction in risk. This may be related to anti-inflammatory or antithrombotic effects of statin therapy.
The authors made use of a very large administrative claims database, Optum Health, to look at more than 15 million annual members. They were able to identify 2000 women with a diagnostic code for VTE treatment. The women were between ages 50 and 64 years, and they were compared with 200,000 controls without VTE, matched in 10-to-1 fashion.
About 50% of the women were taking oral hormone therapy, and about 50% took non-oral transdermal or other non-oral formulations of hormone therapy. The odds ratio for VTE was 1.53 among the women who did not also have prescription records for statin therapy. They were able to look at prescribed prescriptions for both the hormone therapy and the statins. Among the women prescribed hormone therapy and also low- to intermediate-dose statins, the odds ratio was 1.29. So that was quite a mitigation of the elevated risk. Among the women taking high-intensity statins, the odds ratio was 1.06, and there was no significant elevation.
We do need more data and more research on this question. One approach would be a meta-analysis of all of the existing randomized trials of hormone therapy in recent years wherein there was increased uptake of statin therapy to look at this question not only for VTE but also for coronary heart disease, stroke, and other CVD outcomes to see whether statin therapy is associated with some attenuation of the excess risk. We also need a targeted randomized trial of statins vs placebo among women who have clear indications for hormone therapy but may be at some increased risk for VTE. That type of trial would be extremely helpful.
These include choosing a transdermal rather than an oral formulation of hormone therapy and using lower doses of hormone therapy. Also, women who are clear candidates for hormone therapy and also for statins, it’s obvious that statins could be co-prescribed. Even among women who are clear candidates for hormone therapy but only intermediate borderline candidates for statin therapy, the prescription of statins might be considered in that clinical scenario to try to mitigate that excess risk for VTE.
JoAnn E. Manson, MD, DrPH, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from: Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This is Dr JoAnn Manson, professor of medicine at Harvard Medical School and Brigham and Women’s Hospital. I’d like to talk with you about a recent report in JAMA Network Open on the subject of whether statin therapy may be able to offset some of the excess risk for venous thromboembolism (VTE) among women taking menopausal hormone therapy.
It’s an important issue because we know that menopausal hormone therapy, especially oral therapy, is linked to an excess risk for VTE, approximately doubling of risk in the randomized clinical trials. There is also emerging evidence from some randomized trials, such as the Jupiter trial, that step therapy may be linked to a reduction in risk. This may be related to anti-inflammatory or antithrombotic effects of statin therapy.
The authors made use of a very large administrative claims database, Optum Health, to look at more than 15 million annual members. They were able to identify 2000 women with a diagnostic code for VTE treatment. The women were between ages 50 and 64 years, and they were compared with 200,000 controls without VTE, matched in 10-to-1 fashion.
About 50% of the women were taking oral hormone therapy, and about 50% took non-oral transdermal or other non-oral formulations of hormone therapy. The odds ratio for VTE was 1.53 among the women who did not also have prescription records for statin therapy. They were able to look at prescribed prescriptions for both the hormone therapy and the statins. Among the women prescribed hormone therapy and also low- to intermediate-dose statins, the odds ratio was 1.29. So that was quite a mitigation of the elevated risk. Among the women taking high-intensity statins, the odds ratio was 1.06, and there was no significant elevation.
We do need more data and more research on this question. One approach would be a meta-analysis of all of the existing randomized trials of hormone therapy in recent years wherein there was increased uptake of statin therapy to look at this question not only for VTE but also for coronary heart disease, stroke, and other CVD outcomes to see whether statin therapy is associated with some attenuation of the excess risk. We also need a targeted randomized trial of statins vs placebo among women who have clear indications for hormone therapy but may be at some increased risk for VTE. That type of trial would be extremely helpful.
These include choosing a transdermal rather than an oral formulation of hormone therapy and using lower doses of hormone therapy. Also, women who are clear candidates for hormone therapy and also for statins, it’s obvious that statins could be co-prescribed. Even among women who are clear candidates for hormone therapy but only intermediate borderline candidates for statin therapy, the prescription of statins might be considered in that clinical scenario to try to mitigate that excess risk for VTE.
JoAnn E. Manson, MD, DrPH, has disclosed the following relevant financial relationships: Received study pill donation and infrastructure support from: Mars Symbioscience (for the COSMOS trial).
A version of this article appeared on Medscape.com.
Noninvasive Brain Stimulation a Breakthrough for Hypnotherapy?
Less than 2 minutes of transcranial magnetic stimulation (TMS) targeting specific areas of the brain can boost an individual’s ability to be hypnotized, in new findings that could increase the efficacy of therapeutic hypnosis and expand the pool of patients who can benefit from it.
“We were able to increase hypnotizability, a neuropsychological trait previously shown to be as stable as IQ in adulthood,” said co-senior author David Spiegel, MD, professor of psychiatry and behavioral sciences, Stanford University, Palo Alto, California.
“Our findings would allow us to combine neurostimulation with hypnosis to expand the number of people able to benefit from hypnosis and enhance their responsiveness to treatment,” Dr. Spiegel added.
The study was published online on January 4, 2024, in Nature Mental Health.
A Breakthrough for Hypnotherapy?
About two thirds of the general adult population are estimated to be at least somewhat hypnotizable, and 15% are highly hypnotizable.
Through brain imaging, the Stanford team found that high hypnotizability is associated with greater functional connectivity between the left dorsolateral prefrontal cortex (DLPFC) and the dorsal anterior cingulate cortex.
In the double-blind study, they randomly assigned 80 patients (mean age, 48 years; 94% women) with fibromyalgia syndrome to active, or sham, continuous theta-burst stimulation over a personalized neuroimaging-derived left DLPFC target — a technique known as Stanford Hypnosis Integrated with Functional Connectivity-targeted Transcranial Stimulation (SHIFT). Individuals who were naturally highly hypnotizable were excluded.
“A novel aspect of this trial is that we used the person’s own brain networks, based on brain imaging, to target the right spot,” Co-senior author Nolan Williams, MD, with Stanford University, California, said in a news release.
The team chose patients with chronic pain because hypnosis has been shown to be a “highly effective analgesic that has a far better risk/benefit ratio than widely overutilized opioids that have serious fatal overdose potential,” Spiegel told this news organization.
The pre-to-post SHIFT change in hypnotic induction profile scores, a standardized measure of hypnotizability, was significantly greater in the active vs sham group after just 92 seconds of stimulation (P = .046).
Only the active SHIFT group showed a significant increase in hypnotizability following stimulation, an effect that lasted for about 1 hour.
“Increasing hypnotizability in people who are low-to-medium hypnotizable individuals could improve both the efficacy and effectiveness of therapeutic hypnosis as a clinical intervention,” the researchers wrote.
They note that because this was a “mechanistic study,” it did not explore the impact of increased hypnotizability on disease symptoms. They also note that further studies are needed to assess the dose-response relationships of SHIFT.
Transformative Research
“This line of research is fascinating,” Shaheen Lakhan, MD, PhD, neurologist, and researcher in Boston, told this news organization.
“We are nearing an era of personalized, noninvasive brain modulation. The ability to individually modulate the DLPFC opens new possibilities for brain health beyond hypnotizability for fibromyalgia,” said Dr. Lakhan, who wasn’t involved in the study.
“The DLPFC is involved in executive functions (and disorders) like attention (ADHD), emotional regulation (depression), motivation (schizophrenia), and impulse control (addiction),” he noted.
“Soon we may no longer need large expensive devices like transcranial magnetic stimulators as in this research study. Smartphones could deliver tailored digital therapeutics by engaging specific brain circuits,” Dr. Lakhan predicted.
“Imagine using an app to receive treatment customized to your unique brain and needs — all without anything implanted and delivered anywhere. The potential to precisely modulate the brain’s wiring to enhance cognition and mental health, without surgery or physical constraints, is incredibly promising. The possibilities are intriguing and could truly transform how we address brain diseases,” he added.
The study was supported by a grant from the National Center for Complementary and Integrative Health (NCCIH), part of the National Institutes of Health (NIH). Dr. Williams is a named inventor on Stanford-owned intellectual property relating to accelerated TMS pulse pattern sequences and neuroimaging-based TMS targeting; has served on scientific advisory boards for Otsuka, NeuraWell, Magnus Medical, and Nooma as a paid advisor; and holds equity/stock options in Magnus Medical, NeuraWell, and Nooma. Dr. Spiegel is a cofounder of Reveri Health, Inc., an interactive hypnosis app (not utilized in the current study).
A version of this article appeared on Medscape.com.
Less than 2 minutes of transcranial magnetic stimulation (TMS) targeting specific areas of the brain can boost an individual’s ability to be hypnotized, in new findings that could increase the efficacy of therapeutic hypnosis and expand the pool of patients who can benefit from it.
“We were able to increase hypnotizability, a neuropsychological trait previously shown to be as stable as IQ in adulthood,” said co-senior author David Spiegel, MD, professor of psychiatry and behavioral sciences, Stanford University, Palo Alto, California.
“Our findings would allow us to combine neurostimulation with hypnosis to expand the number of people able to benefit from hypnosis and enhance their responsiveness to treatment,” Dr. Spiegel added.
The study was published online on January 4, 2024, in Nature Mental Health.
A Breakthrough for Hypnotherapy?
About two thirds of the general adult population are estimated to be at least somewhat hypnotizable, and 15% are highly hypnotizable.
Through brain imaging, the Stanford team found that high hypnotizability is associated with greater functional connectivity between the left dorsolateral prefrontal cortex (DLPFC) and the dorsal anterior cingulate cortex.
In the double-blind study, they randomly assigned 80 patients (mean age, 48 years; 94% women) with fibromyalgia syndrome to active, or sham, continuous theta-burst stimulation over a personalized neuroimaging-derived left DLPFC target — a technique known as Stanford Hypnosis Integrated with Functional Connectivity-targeted Transcranial Stimulation (SHIFT). Individuals who were naturally highly hypnotizable were excluded.
“A novel aspect of this trial is that we used the person’s own brain networks, based on brain imaging, to target the right spot,” Co-senior author Nolan Williams, MD, with Stanford University, California, said in a news release.
The team chose patients with chronic pain because hypnosis has been shown to be a “highly effective analgesic that has a far better risk/benefit ratio than widely overutilized opioids that have serious fatal overdose potential,” Spiegel told this news organization.
The pre-to-post SHIFT change in hypnotic induction profile scores, a standardized measure of hypnotizability, was significantly greater in the active vs sham group after just 92 seconds of stimulation (P = .046).
Only the active SHIFT group showed a significant increase in hypnotizability following stimulation, an effect that lasted for about 1 hour.
“Increasing hypnotizability in people who are low-to-medium hypnotizable individuals could improve both the efficacy and effectiveness of therapeutic hypnosis as a clinical intervention,” the researchers wrote.
They note that because this was a “mechanistic study,” it did not explore the impact of increased hypnotizability on disease symptoms. They also note that further studies are needed to assess the dose-response relationships of SHIFT.
Transformative Research
“This line of research is fascinating,” Shaheen Lakhan, MD, PhD, neurologist, and researcher in Boston, told this news organization.
“We are nearing an era of personalized, noninvasive brain modulation. The ability to individually modulate the DLPFC opens new possibilities for brain health beyond hypnotizability for fibromyalgia,” said Dr. Lakhan, who wasn’t involved in the study.
“The DLPFC is involved in executive functions (and disorders) like attention (ADHD), emotional regulation (depression), motivation (schizophrenia), and impulse control (addiction),” he noted.
“Soon we may no longer need large expensive devices like transcranial magnetic stimulators as in this research study. Smartphones could deliver tailored digital therapeutics by engaging specific brain circuits,” Dr. Lakhan predicted.
“Imagine using an app to receive treatment customized to your unique brain and needs — all without anything implanted and delivered anywhere. The potential to precisely modulate the brain’s wiring to enhance cognition and mental health, without surgery or physical constraints, is incredibly promising. The possibilities are intriguing and could truly transform how we address brain diseases,” he added.
The study was supported by a grant from the National Center for Complementary and Integrative Health (NCCIH), part of the National Institutes of Health (NIH). Dr. Williams is a named inventor on Stanford-owned intellectual property relating to accelerated TMS pulse pattern sequences and neuroimaging-based TMS targeting; has served on scientific advisory boards for Otsuka, NeuraWell, Magnus Medical, and Nooma as a paid advisor; and holds equity/stock options in Magnus Medical, NeuraWell, and Nooma. Dr. Spiegel is a cofounder of Reveri Health, Inc., an interactive hypnosis app (not utilized in the current study).
A version of this article appeared on Medscape.com.
Less than 2 minutes of transcranial magnetic stimulation (TMS) targeting specific areas of the brain can boost an individual’s ability to be hypnotized, in new findings that could increase the efficacy of therapeutic hypnosis and expand the pool of patients who can benefit from it.
“We were able to increase hypnotizability, a neuropsychological trait previously shown to be as stable as IQ in adulthood,” said co-senior author David Spiegel, MD, professor of psychiatry and behavioral sciences, Stanford University, Palo Alto, California.
“Our findings would allow us to combine neurostimulation with hypnosis to expand the number of people able to benefit from hypnosis and enhance their responsiveness to treatment,” Dr. Spiegel added.
The study was published online on January 4, 2024, in Nature Mental Health.
A Breakthrough for Hypnotherapy?
About two thirds of the general adult population are estimated to be at least somewhat hypnotizable, and 15% are highly hypnotizable.
Through brain imaging, the Stanford team found that high hypnotizability is associated with greater functional connectivity between the left dorsolateral prefrontal cortex (DLPFC) and the dorsal anterior cingulate cortex.
In the double-blind study, they randomly assigned 80 patients (mean age, 48 years; 94% women) with fibromyalgia syndrome to active, or sham, continuous theta-burst stimulation over a personalized neuroimaging-derived left DLPFC target — a technique known as Stanford Hypnosis Integrated with Functional Connectivity-targeted Transcranial Stimulation (SHIFT). Individuals who were naturally highly hypnotizable were excluded.
“A novel aspect of this trial is that we used the person’s own brain networks, based on brain imaging, to target the right spot,” Co-senior author Nolan Williams, MD, with Stanford University, California, said in a news release.
The team chose patients with chronic pain because hypnosis has been shown to be a “highly effective analgesic that has a far better risk/benefit ratio than widely overutilized opioids that have serious fatal overdose potential,” Spiegel told this news organization.
The pre-to-post SHIFT change in hypnotic induction profile scores, a standardized measure of hypnotizability, was significantly greater in the active vs sham group after just 92 seconds of stimulation (P = .046).
Only the active SHIFT group showed a significant increase in hypnotizability following stimulation, an effect that lasted for about 1 hour.
“Increasing hypnotizability in people who are low-to-medium hypnotizable individuals could improve both the efficacy and effectiveness of therapeutic hypnosis as a clinical intervention,” the researchers wrote.
They note that because this was a “mechanistic study,” it did not explore the impact of increased hypnotizability on disease symptoms. They also note that further studies are needed to assess the dose-response relationships of SHIFT.
Transformative Research
“This line of research is fascinating,” Shaheen Lakhan, MD, PhD, neurologist, and researcher in Boston, told this news organization.
“We are nearing an era of personalized, noninvasive brain modulation. The ability to individually modulate the DLPFC opens new possibilities for brain health beyond hypnotizability for fibromyalgia,” said Dr. Lakhan, who wasn’t involved in the study.
“The DLPFC is involved in executive functions (and disorders) like attention (ADHD), emotional regulation (depression), motivation (schizophrenia), and impulse control (addiction),” he noted.
“Soon we may no longer need large expensive devices like transcranial magnetic stimulators as in this research study. Smartphones could deliver tailored digital therapeutics by engaging specific brain circuits,” Dr. Lakhan predicted.
“Imagine using an app to receive treatment customized to your unique brain and needs — all without anything implanted and delivered anywhere. The potential to precisely modulate the brain’s wiring to enhance cognition and mental health, without surgery or physical constraints, is incredibly promising. The possibilities are intriguing and could truly transform how we address brain diseases,” he added.
The study was supported by a grant from the National Center for Complementary and Integrative Health (NCCIH), part of the National Institutes of Health (NIH). Dr. Williams is a named inventor on Stanford-owned intellectual property relating to accelerated TMS pulse pattern sequences and neuroimaging-based TMS targeting; has served on scientific advisory boards for Otsuka, NeuraWell, Magnus Medical, and Nooma as a paid advisor; and holds equity/stock options in Magnus Medical, NeuraWell, and Nooma. Dr. Spiegel is a cofounder of Reveri Health, Inc., an interactive hypnosis app (not utilized in the current study).
A version of this article appeared on Medscape.com.
A Counterintuitive Approach to Lowering Cholesterol in Children
With the flip of the calendar a few short weeks ago, gyms and fitness centers began ramping up their advertising campaigns in hopes of attracting the horde of resolution makers searching for a place where they can inject some exercise into their sedentary lives. A recent survey by C.S. Mott’s Children’s Hospital found that even young people are setting health-related goals with more than half of the parents of 11- to 18-year-olds reporting their children were setting personal goals for themselves. More than 40% of the young people listed more exercise as a target.
However, our personal and professional experiences have taught us that achieving goals, particularly when it comes to exercise, is far more difficult than setting the target. Finding an exercise buddy can be an important motivator on the days when just lacing up one’s sneakers is a stumbling block. Investing in a gym membership and sweating with a peer group can help. However, it is an investment that rarely pays a dividend. Exercise isn’t fun for everyone. For adults, showing up at a gym may be just one more reminder of how they have already lost their competitive edge over their leaner and fitter peers. If they aren’t lucky enough to find a sport or activity that they enjoy, the loneliness of the long-distance runner has little appeal.
A recent study on children in the United Kingdom suggests that at least when it comes to teens and young adults we as physicians may actually have been making things worse for our obese patients by urging them to accept unrealistic activity goals. While it is already known that sedentary time is responsible for 70% of the total increase in cholesterol as children advance to young adulthood an unqualified recommendation for more exercise may not be the best advice.
In an interview with the study author, Andre O. Agbaje MD, MPH, said that in his large study population “light physical activity outperforms moderate to vigorous physical activity by five to eight times in lowering lipids”. While we may be surprised by this counterintuitive finding, Dr. Agbaje points out that an increase in sedentariness from 6 to 9 hours per day translates into a loss of 3 hours of light physical activity. In other words if you’re not sedentary you must be standing at attention or engaged in some light activity.
In my experience, and I suspect yours, it is difficult to get adults to do something, particularly if that something involves exerting energy, even a small amount of energy. The general admonishment of “be more active” is often met with a blank stare and the sometimes unspoken question “Like what?”
You could fall into a bottomless trap with them by suggesting a long list of activities, many of which are probably ones you do or would enjoy but don’t happen to fit with any of their interests or capabilities. Your chances of hitting on a perfect activity that the patient will attempt, let alone adopt, is very slim. Those of you with more patience than I have may choose to persist with this strategy. You could argue that even if the patient only dabbles briefly in one of your recommended activities, this is a minor victory worth celebrating. Who knows? The brief jolt of energy they received from this activity may prompt them to seek and find something else that works.
My interpretation of Dr. Agbaje’s findings is this: If we are going to suggest more activity, aim low. Don’t even mention the heavily weighted words “sport” or “exercise,” which are likely to dredge up bad memories. For adults, “Go shopping” or “Visit a friend” may be sufficient to at least get the person off the couch and on their feet and moving, even if very briefly.
The second message from this study applies more to children and adolescents and is one of those unusual instances in which a negative intervention may be more effective than a positive approach. Acknowledging that we are likely to have difficulty finding even a light activity that the child enjoys, why not pivot to the other side of the equation? Make a list of the child’s primary sedentary “activities.” Then suggest the parents put the child on a couch potato diet by immediately cutting in half the time he or she spends being sedentary. By definition, this will automatically increase his or her light physical activity by 50%. According to Dr. Agbaje’s data, this should be more effective in lowering lipids than in the unlikely event of finding a moderate activity the child accepts.
You can argue that the child will hound his or her parents unmercifully asking to be entertained. This may be true and this persistent complaining will be more likely to come from the older the child and the longer that the child has been allowed to be sedentary. Although the child may appear to have lost the ability to self amuse, I contend this isn’t a permanent loss and, This is another example of how saying “No!” in the right circumstances is often the most effective remedy for an unhealthy situation. I would never claim saying “No” is easy and helping parents to learn how to say “No” is one of our most difficult challenges. But, nothing else seems to be working.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
With the flip of the calendar a few short weeks ago, gyms and fitness centers began ramping up their advertising campaigns in hopes of attracting the horde of resolution makers searching for a place where they can inject some exercise into their sedentary lives. A recent survey by C.S. Mott’s Children’s Hospital found that even young people are setting health-related goals with more than half of the parents of 11- to 18-year-olds reporting their children were setting personal goals for themselves. More than 40% of the young people listed more exercise as a target.
However, our personal and professional experiences have taught us that achieving goals, particularly when it comes to exercise, is far more difficult than setting the target. Finding an exercise buddy can be an important motivator on the days when just lacing up one’s sneakers is a stumbling block. Investing in a gym membership and sweating with a peer group can help. However, it is an investment that rarely pays a dividend. Exercise isn’t fun for everyone. For adults, showing up at a gym may be just one more reminder of how they have already lost their competitive edge over their leaner and fitter peers. If they aren’t lucky enough to find a sport or activity that they enjoy, the loneliness of the long-distance runner has little appeal.
A recent study on children in the United Kingdom suggests that at least when it comes to teens and young adults we as physicians may actually have been making things worse for our obese patients by urging them to accept unrealistic activity goals. While it is already known that sedentary time is responsible for 70% of the total increase in cholesterol as children advance to young adulthood an unqualified recommendation for more exercise may not be the best advice.
In an interview with the study author, Andre O. Agbaje MD, MPH, said that in his large study population “light physical activity outperforms moderate to vigorous physical activity by five to eight times in lowering lipids”. While we may be surprised by this counterintuitive finding, Dr. Agbaje points out that an increase in sedentariness from 6 to 9 hours per day translates into a loss of 3 hours of light physical activity. In other words if you’re not sedentary you must be standing at attention or engaged in some light activity.
In my experience, and I suspect yours, it is difficult to get adults to do something, particularly if that something involves exerting energy, even a small amount of energy. The general admonishment of “be more active” is often met with a blank stare and the sometimes unspoken question “Like what?”
You could fall into a bottomless trap with them by suggesting a long list of activities, many of which are probably ones you do or would enjoy but don’t happen to fit with any of their interests or capabilities. Your chances of hitting on a perfect activity that the patient will attempt, let alone adopt, is very slim. Those of you with more patience than I have may choose to persist with this strategy. You could argue that even if the patient only dabbles briefly in one of your recommended activities, this is a minor victory worth celebrating. Who knows? The brief jolt of energy they received from this activity may prompt them to seek and find something else that works.
My interpretation of Dr. Agbaje’s findings is this: If we are going to suggest more activity, aim low. Don’t even mention the heavily weighted words “sport” or “exercise,” which are likely to dredge up bad memories. For adults, “Go shopping” or “Visit a friend” may be sufficient to at least get the person off the couch and on their feet and moving, even if very briefly.
The second message from this study applies more to children and adolescents and is one of those unusual instances in which a negative intervention may be more effective than a positive approach. Acknowledging that we are likely to have difficulty finding even a light activity that the child enjoys, why not pivot to the other side of the equation? Make a list of the child’s primary sedentary “activities.” Then suggest the parents put the child on a couch potato diet by immediately cutting in half the time he or she spends being sedentary. By definition, this will automatically increase his or her light physical activity by 50%. According to Dr. Agbaje’s data, this should be more effective in lowering lipids than in the unlikely event of finding a moderate activity the child accepts.
You can argue that the child will hound his or her parents unmercifully asking to be entertained. This may be true and this persistent complaining will be more likely to come from the older the child and the longer that the child has been allowed to be sedentary. Although the child may appear to have lost the ability to self amuse, I contend this isn’t a permanent loss and, This is another example of how saying “No!” in the right circumstances is often the most effective remedy for an unhealthy situation. I would never claim saying “No” is easy and helping parents to learn how to say “No” is one of our most difficult challenges. But, nothing else seems to be working.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
With the flip of the calendar a few short weeks ago, gyms and fitness centers began ramping up their advertising campaigns in hopes of attracting the horde of resolution makers searching for a place where they can inject some exercise into their sedentary lives. A recent survey by C.S. Mott’s Children’s Hospital found that even young people are setting health-related goals with more than half of the parents of 11- to 18-year-olds reporting their children were setting personal goals for themselves. More than 40% of the young people listed more exercise as a target.
However, our personal and professional experiences have taught us that achieving goals, particularly when it comes to exercise, is far more difficult than setting the target. Finding an exercise buddy can be an important motivator on the days when just lacing up one’s sneakers is a stumbling block. Investing in a gym membership and sweating with a peer group can help. However, it is an investment that rarely pays a dividend. Exercise isn’t fun for everyone. For adults, showing up at a gym may be just one more reminder of how they have already lost their competitive edge over their leaner and fitter peers. If they aren’t lucky enough to find a sport or activity that they enjoy, the loneliness of the long-distance runner has little appeal.
A recent study on children in the United Kingdom suggests that at least when it comes to teens and young adults we as physicians may actually have been making things worse for our obese patients by urging them to accept unrealistic activity goals. While it is already known that sedentary time is responsible for 70% of the total increase in cholesterol as children advance to young adulthood an unqualified recommendation for more exercise may not be the best advice.
In an interview with the study author, Andre O. Agbaje MD, MPH, said that in his large study population “light physical activity outperforms moderate to vigorous physical activity by five to eight times in lowering lipids”. While we may be surprised by this counterintuitive finding, Dr. Agbaje points out that an increase in sedentariness from 6 to 9 hours per day translates into a loss of 3 hours of light physical activity. In other words if you’re not sedentary you must be standing at attention or engaged in some light activity.
In my experience, and I suspect yours, it is difficult to get adults to do something, particularly if that something involves exerting energy, even a small amount of energy. The general admonishment of “be more active” is often met with a blank stare and the sometimes unspoken question “Like what?”
You could fall into a bottomless trap with them by suggesting a long list of activities, many of which are probably ones you do or would enjoy but don’t happen to fit with any of their interests or capabilities. Your chances of hitting on a perfect activity that the patient will attempt, let alone adopt, is very slim. Those of you with more patience than I have may choose to persist with this strategy. You could argue that even if the patient only dabbles briefly in one of your recommended activities, this is a minor victory worth celebrating. Who knows? The brief jolt of energy they received from this activity may prompt them to seek and find something else that works.
My interpretation of Dr. Agbaje’s findings is this: If we are going to suggest more activity, aim low. Don’t even mention the heavily weighted words “sport” or “exercise,” which are likely to dredge up bad memories. For adults, “Go shopping” or “Visit a friend” may be sufficient to at least get the person off the couch and on their feet and moving, even if very briefly.
The second message from this study applies more to children and adolescents and is one of those unusual instances in which a negative intervention may be more effective than a positive approach. Acknowledging that we are likely to have difficulty finding even a light activity that the child enjoys, why not pivot to the other side of the equation? Make a list of the child’s primary sedentary “activities.” Then suggest the parents put the child on a couch potato diet by immediately cutting in half the time he or she spends being sedentary. By definition, this will automatically increase his or her light physical activity by 50%. According to Dr. Agbaje’s data, this should be more effective in lowering lipids than in the unlikely event of finding a moderate activity the child accepts.
You can argue that the child will hound his or her parents unmercifully asking to be entertained. This may be true and this persistent complaining will be more likely to come from the older the child and the longer that the child has been allowed to be sedentary. Although the child may appear to have lost the ability to self amuse, I contend this isn’t a permanent loss and, This is another example of how saying “No!” in the right circumstances is often the most effective remedy for an unhealthy situation. I would never claim saying “No” is easy and helping parents to learn how to say “No” is one of our most difficult challenges. But, nothing else seems to be working.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Healthcare Violence: Doctors and Nurses Are Bearing the Brunt of Business Pressures
This transcript has been edited for clarity.
This month, I want to tackle the difficult subject of violence toward healthcare workers. There’s a reason this is top of mind for me in my practice, but I want to start by acknowledging that this has been a much larger issue for our profession and one that has been growing for a number of years now.
They also estimate that that rate doubled between 2011 and 2018. I think that range is important because it proves this was a problem, and a crescendoing problem, even before COVID.
Another thing I think is relevant is to look at where in the healthcare system are these attacks most likely. In the emergency room, ER staff have seen hostility toward them rise by at least 25% over the past several years. Some of the seeds of mistrust that were sown between the general public and the scientific and medical communities around the pandemic. I think there’s some explanation there for why that might be a particular crucible.
Perhaps most disturbingly of all, 60% of the victims of healthcare workplace violence are bedside nurses. There is something about the intensity of the inpatient setting that makes nerves particularly frayed and unfortunately makes patients and family members more likely to lash out. I think it’s actually the heightened sense of mortality.
I’m not excusing any of these behaviors, but maybe it’s akin to road rage. On the road, behind the wheel, tiny gestures can actually be, on some level, perceived as threats to our survival. Another driver swerving into your lane activates a fight-or-flight response, you feel threatened, and you might respond in the moment very rashly. I wonder if we’re not seeing that, quite unfairly, play out against bedside staff in our hospitals.
Here’s the thing. Those of us who practice in the outpatient setting — 95% of my work, for instance, happens in clinic — are not immune to this either. There are some very harrowing recent examples of physicians being killed, typically at gunpoint, often by patients, sometimes by aggrieved family members, in their offices. An orthopedist in Tennessee, a back surgeon in Tulsa, along with three of their colleagues. In the latter case, the assailant specifically blamed the surgeon for their pain.
This is where I think things get even more scary. We have to be the bearers of bad news in our profession. This has long been the task of the oncologist, in particular, to convey things that people don’t want to hear.
I think what brought this to my mind in terms of my reading was an incredible article in The ASCO Post and also in the Journal of Clinical Oncology by Dr. Noelle LoConte, who’s a medical oncologist in Wisconsin. The article is called, “I Want to Kill You,” and it recounts her telling a previously stage III colon cancer patient, with whom she thought she had good rapport, that the disease had recurred. The patient’s immediate reaction in the heat of that moment was to say, Dr LoConte, I want to kill you. I want to blow your face off.
Already, there’s clearly tension when we are telling people what they don’t want to hear. I think the final piece of the puzzle goes back to the intrusion of the business of healthcare on the practice of medicine. This is what I witnessed very recently. One of the things that’s interesting to think about is how what we do is now framed as customer service. I know there’s deriding of this model, but if perception is reality, we have a system where patients are set up to view themselves as consumers.
Let’s say, for instance, you’re in the unfortunate circumstance of being diagnosed with cancer and your insurer gives you the option to go to multiple oncologists. If you’re online browsing for oncologists, how do you differentiate me from some of my colleagues? The answer on these rating websites often has to do with domains that are about the overall experience — not just the patient-doctor interaction but also things like wait time, friendliness of staff, and promptness of care delivery.
That, I think, is the final piece of the puzzle, because what I really risk when I sit down with a patient and lay out a treatment plan is overpromising and underdelivering. I am long used to citing median overall survival for expectation of outcome. Of course, every patient wants to be an exceptional responder. Most patients want to be on the latter half of median survival. No one wants to be on the disappointingly shorter half.
My point is that I’ve long been able to mitigate that uncertainty for patients. What is getting harder and harder to explain away is the delay incurred between someone’s diagnosis, my meeting them and laying out a treatment plan, and their actual initiation of that therapy.
This finally brings me to my recent personal encounter. I have long taken care of a patient, much like Dr LoConte’s, with an extremely calm demeanor. I thought we had a great therapeutic alliance. I had to tell the patient that the disease had recurred, and then I laid out a treatment plan. It took weeks and then months for the insurer to approve this plan despite my providing my note in a timely fashion with a mountain of evidence behind the regimen that I’d selected.
This is where I think insurers — when they deny, deflect, and delay — are not taking adequate responsibility for the impact that has on the therapeutic alliance between a patient and their doctor. These people are trusting us with their lives. As an oncologist, I’ve already told them something they didn’t want to hear, and now I’m compounding that with the uncertainty of when we can actually begin treatment.
This gentleman — who, again, is normally extremely kind and affable — showed up at my office and was incredibly hostile toward me and my staff because of the delay that he was encountering. We literally couldn’t tell him when his insurer was going to approve his treatment, which would have been financially disastrous if he had tried to pay for it himself out of pocket. He needed his insurer’s approval before we could start, but we didn’t know when he could start. That uncertainty and not knowing was gnawing away at him until he was at the end of his rope.
What I’m here to say is that this has been a difficult couple of years in healthcare. I’m well aware that our ER staff are on the front lines, as are our bedside and inpatient teams. Even in the outpatient setting, I think we’re seeing this crucible and we’re seeing the pressure just grow, and grow, and grow. It’s like fracking. The more you increase the pressure, the more eventually you’re going to find out where the cracks are.
These patients are the ultimate stakeholders. It’s their lives on the line, and we should be concerned, but perhaps ultimately not surprised, that they’re lashing out to be heard. Given no other resort, they are taking out their frustration and their aggression on us. It›s not fair, but I am newly aware of it because, in a patient with whom I thought we had a superb rapport, I saw that vanish. As soon as he thought that his life was at risk, his fight-or-flight response kicked in. I was not dealing with the same man I knew. I was dealing with someone who was desperate and who just wanted to know when he could get the treatment.
I think this has taken the likelihood of workplace hostility to a whole other level for those of us in healthcare.
For any patients listening, I beg of you, please don’t shoot the messenger. We are here to serve you the best we can, but there are many external factors at play. We are doing our best to mitigate those for you so we can deliver the care that we promised in as timely a fashion as we can.
I hope everyone out there can stay safe. Thank you.
Dr. Lewis is director of gastrointestinal oncology at Intermountain Healthcare in Salt Lake City, Utah. He has an interest in neuroendocrine tumors, hereditary cancer syndromes, and patient-physician communication. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This month, I want to tackle the difficult subject of violence toward healthcare workers. There’s a reason this is top of mind for me in my practice, but I want to start by acknowledging that this has been a much larger issue for our profession and one that has been growing for a number of years now.
They also estimate that that rate doubled between 2011 and 2018. I think that range is important because it proves this was a problem, and a crescendoing problem, even before COVID.
Another thing I think is relevant is to look at where in the healthcare system are these attacks most likely. In the emergency room, ER staff have seen hostility toward them rise by at least 25% over the past several years. Some of the seeds of mistrust that were sown between the general public and the scientific and medical communities around the pandemic. I think there’s some explanation there for why that might be a particular crucible.
Perhaps most disturbingly of all, 60% of the victims of healthcare workplace violence are bedside nurses. There is something about the intensity of the inpatient setting that makes nerves particularly frayed and unfortunately makes patients and family members more likely to lash out. I think it’s actually the heightened sense of mortality.
I’m not excusing any of these behaviors, but maybe it’s akin to road rage. On the road, behind the wheel, tiny gestures can actually be, on some level, perceived as threats to our survival. Another driver swerving into your lane activates a fight-or-flight response, you feel threatened, and you might respond in the moment very rashly. I wonder if we’re not seeing that, quite unfairly, play out against bedside staff in our hospitals.
Here’s the thing. Those of us who practice in the outpatient setting — 95% of my work, for instance, happens in clinic — are not immune to this either. There are some very harrowing recent examples of physicians being killed, typically at gunpoint, often by patients, sometimes by aggrieved family members, in their offices. An orthopedist in Tennessee, a back surgeon in Tulsa, along with three of their colleagues. In the latter case, the assailant specifically blamed the surgeon for their pain.
This is where I think things get even more scary. We have to be the bearers of bad news in our profession. This has long been the task of the oncologist, in particular, to convey things that people don’t want to hear.
I think what brought this to my mind in terms of my reading was an incredible article in The ASCO Post and also in the Journal of Clinical Oncology by Dr. Noelle LoConte, who’s a medical oncologist in Wisconsin. The article is called, “I Want to Kill You,” and it recounts her telling a previously stage III colon cancer patient, with whom she thought she had good rapport, that the disease had recurred. The patient’s immediate reaction in the heat of that moment was to say, Dr LoConte, I want to kill you. I want to blow your face off.
Already, there’s clearly tension when we are telling people what they don’t want to hear. I think the final piece of the puzzle goes back to the intrusion of the business of healthcare on the practice of medicine. This is what I witnessed very recently. One of the things that’s interesting to think about is how what we do is now framed as customer service. I know there’s deriding of this model, but if perception is reality, we have a system where patients are set up to view themselves as consumers.
Let’s say, for instance, you’re in the unfortunate circumstance of being diagnosed with cancer and your insurer gives you the option to go to multiple oncologists. If you’re online browsing for oncologists, how do you differentiate me from some of my colleagues? The answer on these rating websites often has to do with domains that are about the overall experience — not just the patient-doctor interaction but also things like wait time, friendliness of staff, and promptness of care delivery.
That, I think, is the final piece of the puzzle, because what I really risk when I sit down with a patient and lay out a treatment plan is overpromising and underdelivering. I am long used to citing median overall survival for expectation of outcome. Of course, every patient wants to be an exceptional responder. Most patients want to be on the latter half of median survival. No one wants to be on the disappointingly shorter half.
My point is that I’ve long been able to mitigate that uncertainty for patients. What is getting harder and harder to explain away is the delay incurred between someone’s diagnosis, my meeting them and laying out a treatment plan, and their actual initiation of that therapy.
This finally brings me to my recent personal encounter. I have long taken care of a patient, much like Dr LoConte’s, with an extremely calm demeanor. I thought we had a great therapeutic alliance. I had to tell the patient that the disease had recurred, and then I laid out a treatment plan. It took weeks and then months for the insurer to approve this plan despite my providing my note in a timely fashion with a mountain of evidence behind the regimen that I’d selected.
This is where I think insurers — when they deny, deflect, and delay — are not taking adequate responsibility for the impact that has on the therapeutic alliance between a patient and their doctor. These people are trusting us with their lives. As an oncologist, I’ve already told them something they didn’t want to hear, and now I’m compounding that with the uncertainty of when we can actually begin treatment.
This gentleman — who, again, is normally extremely kind and affable — showed up at my office and was incredibly hostile toward me and my staff because of the delay that he was encountering. We literally couldn’t tell him when his insurer was going to approve his treatment, which would have been financially disastrous if he had tried to pay for it himself out of pocket. He needed his insurer’s approval before we could start, but we didn’t know when he could start. That uncertainty and not knowing was gnawing away at him until he was at the end of his rope.
What I’m here to say is that this has been a difficult couple of years in healthcare. I’m well aware that our ER staff are on the front lines, as are our bedside and inpatient teams. Even in the outpatient setting, I think we’re seeing this crucible and we’re seeing the pressure just grow, and grow, and grow. It’s like fracking. The more you increase the pressure, the more eventually you’re going to find out where the cracks are.
These patients are the ultimate stakeholders. It’s their lives on the line, and we should be concerned, but perhaps ultimately not surprised, that they’re lashing out to be heard. Given no other resort, they are taking out their frustration and their aggression on us. It›s not fair, but I am newly aware of it because, in a patient with whom I thought we had a superb rapport, I saw that vanish. As soon as he thought that his life was at risk, his fight-or-flight response kicked in. I was not dealing with the same man I knew. I was dealing with someone who was desperate and who just wanted to know when he could get the treatment.
I think this has taken the likelihood of workplace hostility to a whole other level for those of us in healthcare.
For any patients listening, I beg of you, please don’t shoot the messenger. We are here to serve you the best we can, but there are many external factors at play. We are doing our best to mitigate those for you so we can deliver the care that we promised in as timely a fashion as we can.
I hope everyone out there can stay safe. Thank you.
Dr. Lewis is director of gastrointestinal oncology at Intermountain Healthcare in Salt Lake City, Utah. He has an interest in neuroendocrine tumors, hereditary cancer syndromes, and patient-physician communication. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
This month, I want to tackle the difficult subject of violence toward healthcare workers. There’s a reason this is top of mind for me in my practice, but I want to start by acknowledging that this has been a much larger issue for our profession and one that has been growing for a number of years now.
They also estimate that that rate doubled between 2011 and 2018. I think that range is important because it proves this was a problem, and a crescendoing problem, even before COVID.
Another thing I think is relevant is to look at where in the healthcare system are these attacks most likely. In the emergency room, ER staff have seen hostility toward them rise by at least 25% over the past several years. Some of the seeds of mistrust that were sown between the general public and the scientific and medical communities around the pandemic. I think there’s some explanation there for why that might be a particular crucible.
Perhaps most disturbingly of all, 60% of the victims of healthcare workplace violence are bedside nurses. There is something about the intensity of the inpatient setting that makes nerves particularly frayed and unfortunately makes patients and family members more likely to lash out. I think it’s actually the heightened sense of mortality.
I’m not excusing any of these behaviors, but maybe it’s akin to road rage. On the road, behind the wheel, tiny gestures can actually be, on some level, perceived as threats to our survival. Another driver swerving into your lane activates a fight-or-flight response, you feel threatened, and you might respond in the moment very rashly. I wonder if we’re not seeing that, quite unfairly, play out against bedside staff in our hospitals.
Here’s the thing. Those of us who practice in the outpatient setting — 95% of my work, for instance, happens in clinic — are not immune to this either. There are some very harrowing recent examples of physicians being killed, typically at gunpoint, often by patients, sometimes by aggrieved family members, in their offices. An orthopedist in Tennessee, a back surgeon in Tulsa, along with three of their colleagues. In the latter case, the assailant specifically blamed the surgeon for their pain.
This is where I think things get even more scary. We have to be the bearers of bad news in our profession. This has long been the task of the oncologist, in particular, to convey things that people don’t want to hear.
I think what brought this to my mind in terms of my reading was an incredible article in The ASCO Post and also in the Journal of Clinical Oncology by Dr. Noelle LoConte, who’s a medical oncologist in Wisconsin. The article is called, “I Want to Kill You,” and it recounts her telling a previously stage III colon cancer patient, with whom she thought she had good rapport, that the disease had recurred. The patient’s immediate reaction in the heat of that moment was to say, Dr LoConte, I want to kill you. I want to blow your face off.
Already, there’s clearly tension when we are telling people what they don’t want to hear. I think the final piece of the puzzle goes back to the intrusion of the business of healthcare on the practice of medicine. This is what I witnessed very recently. One of the things that’s interesting to think about is how what we do is now framed as customer service. I know there’s deriding of this model, but if perception is reality, we have a system where patients are set up to view themselves as consumers.
Let’s say, for instance, you’re in the unfortunate circumstance of being diagnosed with cancer and your insurer gives you the option to go to multiple oncologists. If you’re online browsing for oncologists, how do you differentiate me from some of my colleagues? The answer on these rating websites often has to do with domains that are about the overall experience — not just the patient-doctor interaction but also things like wait time, friendliness of staff, and promptness of care delivery.
That, I think, is the final piece of the puzzle, because what I really risk when I sit down with a patient and lay out a treatment plan is overpromising and underdelivering. I am long used to citing median overall survival for expectation of outcome. Of course, every patient wants to be an exceptional responder. Most patients want to be on the latter half of median survival. No one wants to be on the disappointingly shorter half.
My point is that I’ve long been able to mitigate that uncertainty for patients. What is getting harder and harder to explain away is the delay incurred between someone’s diagnosis, my meeting them and laying out a treatment plan, and their actual initiation of that therapy.
This finally brings me to my recent personal encounter. I have long taken care of a patient, much like Dr LoConte’s, with an extremely calm demeanor. I thought we had a great therapeutic alliance. I had to tell the patient that the disease had recurred, and then I laid out a treatment plan. It took weeks and then months for the insurer to approve this plan despite my providing my note in a timely fashion with a mountain of evidence behind the regimen that I’d selected.
This is where I think insurers — when they deny, deflect, and delay — are not taking adequate responsibility for the impact that has on the therapeutic alliance between a patient and their doctor. These people are trusting us with their lives. As an oncologist, I’ve already told them something they didn’t want to hear, and now I’m compounding that with the uncertainty of when we can actually begin treatment.
This gentleman — who, again, is normally extremely kind and affable — showed up at my office and was incredibly hostile toward me and my staff because of the delay that he was encountering. We literally couldn’t tell him when his insurer was going to approve his treatment, which would have been financially disastrous if he had tried to pay for it himself out of pocket. He needed his insurer’s approval before we could start, but we didn’t know when he could start. That uncertainty and not knowing was gnawing away at him until he was at the end of his rope.
What I’m here to say is that this has been a difficult couple of years in healthcare. I’m well aware that our ER staff are on the front lines, as are our bedside and inpatient teams. Even in the outpatient setting, I think we’re seeing this crucible and we’re seeing the pressure just grow, and grow, and grow. It’s like fracking. The more you increase the pressure, the more eventually you’re going to find out where the cracks are.
These patients are the ultimate stakeholders. It’s their lives on the line, and we should be concerned, but perhaps ultimately not surprised, that they’re lashing out to be heard. Given no other resort, they are taking out their frustration and their aggression on us. It›s not fair, but I am newly aware of it because, in a patient with whom I thought we had a superb rapport, I saw that vanish. As soon as he thought that his life was at risk, his fight-or-flight response kicked in. I was not dealing with the same man I knew. I was dealing with someone who was desperate and who just wanted to know when he could get the treatment.
I think this has taken the likelihood of workplace hostility to a whole other level for those of us in healthcare.
For any patients listening, I beg of you, please don’t shoot the messenger. We are here to serve you the best we can, but there are many external factors at play. We are doing our best to mitigate those for you so we can deliver the care that we promised in as timely a fashion as we can.
I hope everyone out there can stay safe. Thank you.
Dr. Lewis is director of gastrointestinal oncology at Intermountain Healthcare in Salt Lake City, Utah. He has an interest in neuroendocrine tumors, hereditary cancer syndromes, and patient-physician communication. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Botanical Briefs: Neem Oil (Azadirachta indica)
Commonly known as neem or nimba, Azadirachta indica traditionally has been used as an oil or poultice to lighten skin pigment and reduce joint inflammation. Neem is a drought-resistant evergreen tree with thin serrated leaves, white fragrant flowers, and olivelike fruit (Figure 1). This plant is indigenous to India but also is readily found within tropical and semitropical environments throughout the Middle East, Southeast Asia, North Africa, and Australia.

Traditional Uses
For more than 4000 years, neem leaves, bark, fruit, and seeds have been used in food, insecticide, and herbal medicine cross-culturally in Indian Ayurvedic medicine and across Southeast Asia, particularly in Cambodia, Laos, Thailand, Myanmar, and Vietnam.1-3 Because of its many essential nutrients—oleic acid, palmitic acid, stearic acid, linoleic acid, behenic acid, arachidic acid, and palmitoleic acid—and readily available nature, some ethnic groups include neem in their diet.4 Neem commonly is used as a seasoning in soups and rice, eaten as a cooked vegetable, infused into teas and tonics, and pickled with other spices.5
All parts of the neem tree—both externally and internally—have been utilized in traditional medicine for the treatment of various diseases and ailments. The flowers have been used to treat eye diseases and dyspepsia, the fruit has been employed as an anthelmintic, the seeds and leaves have been used for malaria treatment and insecticide, the stem bark has been used for the treatment of diarrhea, and the root bark has been used for skin diseases and inflammation.6 Neem oil is a yellow-brown bitter substance that often is utilized to treat skin diseases such as psoriasis, eczema, fungal infections, and abscesses.
Case Report—A 77-year-old man presented with a diffuse rash across the lower back. He reported that he had been using topical neem oil to alleviate lower back pain and arthritis for the last 6 months with noted relief and improvement of back pain. After roughly 3 to 4 months of using neem oil, he noted a rash on the lower back, bilateral flanks, and buttocks (Figure 2). The rash was asymptomatic, and he denied any pruritus, scaling, pain, or burning. The patient was referred to dermatology and received a diagnosis of chemical leukoderma secondary to contact with A indica. The patient was advised to stop using the topical neem oil, and the rash was simply monitored, as it was asymptomatic.
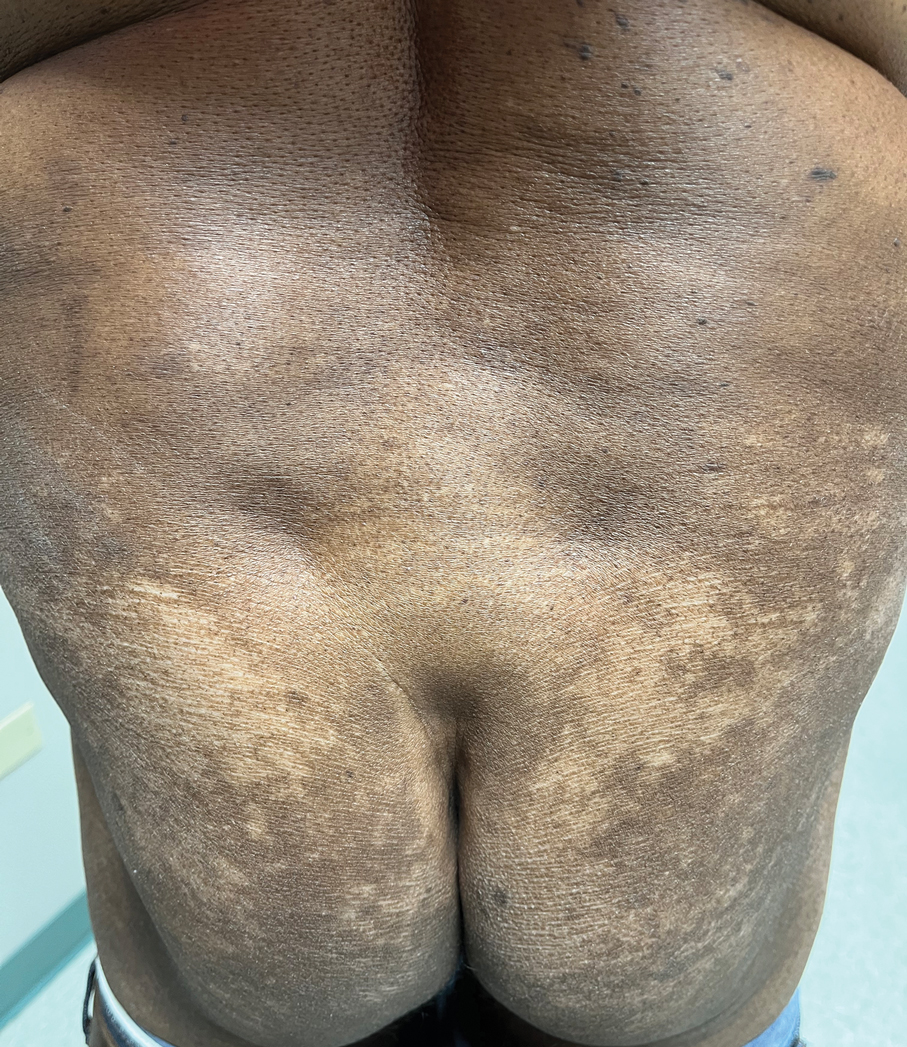
Bioactivity
Research has elucidated multiple bioactivity mechanisms of neem, including melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity.1,7-9 Literature on the diverse phytochemical components of A indica indicate high levels of limonoids, flavonoids, and triterpenoids that are responsible for much of its antioxidant, anti-inflammatory, and insecticide properties.1,10
Melanogenesis-Inhibitory Activity—To date, neem has been added to a number of cosmetic products used in Ayurvedic medicine. One study of isolated compounds in A indica showed superior inhibitory activities against melanogenesis with minimal toxicity to cells (86.5%–105.1% cell viability). Western blot analysis of samples extracted and isolated from neem root and bark showed melanogenesis-inhibitory activities in B16 melanoma cells through the inhibition of microphthalmia-associated transcription factor expression and decreased expression of tyrosinase, as well as tyrosinase-related proteins 1 and 2, which are largely responsible for melanin synthesis.11 In another study, A indica flowers and their extracted constituents—6-deacetylnimbin and kaempferide—suggest melanogenesis-inhibitory activities in B16 melanoma cells with little to no toxicity to the cells (81.0%–111.7% cell viability).1 In an evaluationof A indica seed extracts, some of the isolated limonoids and diterpenoids exhibited a marked melanogenesis-inhibitory effect (74%–91% reduction of melanin content) with no toxicity to the cell.5 All of these studies indicate that active compounds in neem root, bark, flowers, and seeds may be potential skin-lightening agents.
Toxicity Against Pests—Neem seeds have phytochemicals that convey some insecticidal properties. The seeds often are ground into a powder, combined with water, and sprayed onto crops to act as an insecticide. As a natural method of nonpesticidal management, A indica acts as an antifeedant, insect repellent, and egg-laying deterrent that protects crops from damage. Studies of A indica have noted effective nonpesticidal management against arthropod pests such as armyworm, termites, and the oriental fruit fly.7,12,13
Antimalarial Activity—One study indicated that nimbolide, a limonoid from the neem plant, demonstrated antimalarial activity against Plasmodium falciparum. In separate cultures of asexual parasites and mature gametocytes, parasite numbers were less than 50% of the number in control cultures (8.0% vs 8.5% parasitemia, respectively).14 Thus, the lower parasite numbers indicated by this study highlight the antimalarial utility of nimbolide and neem oil.
Antioxidant and Anti-inflammatory Activity—Neem bark has been reported to have considerable antioxidant activity due to its high phenolic content.1,15 One study showed that azadirachtin and nimbolide in neem exhibited concentration-dependent antiradical scavenging activity and antioxidant properties.16
The anti-inflammatory potential for neem may occur via the inhibition of the nuclear factor-κB signaling pathway, which is linked to cancer, inflammation, and apoptosis.17 It also has been observed that nimbidin within neem extracts—such as leaves, bark, and seed extract—suppresses the function of macrophages and neutrophils relevant to inflammation.16 Another study indicated neem’s anti-inflammatory activity due to the regulation of proinflammatory enzymes such as cyclooxygenase and lipoxygenase.18
Safety, Toxicity, and Risks
Ingestion—Although neem is safe to use in the general population, neem oil poisoning has been reported, particularly in young children. Ingesting large quantities of neem has resulted in vomiting, hepatic toxicity, metabolic acidosis, late neurologic sequelae, and encephalopathy in young children.19 The diagnosis of neem oil poisoning is based on patient history, clinical examination, and imaging findings. Poisoning can manifest as drowsiness, tachypnea, and generalized seizures.20
Topical Application—Topical use of neem appears to be safe if the substance is diluted with other ingredients. However, direct application to the skin is not advised, as it may cause leukoderma and could induce allergic contact dermatitis and other allergic reactions.4
Final Thoughts
The use of neem extract for disease prevention and treatment has been prevalent around the world since ancient times. Neem has been documented to possess melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity by means of tyrosinase inhibition, phytochemical production, limonoid expression, and nuclear factor-κB regulation, respectively. However, topical use of neem may trigger a cutaneous response, highlighting the importance of considering a diagnosis of neem oil–induced chemical leukoderma when patients present with a hypopigmented rash and relevant history.
- Kitdamrongtham W, Ishii K, Ebina K, et al. Limonoids and flavonoids from the flowers of Azadirachta indica var. siamensis, and their melanogenesis-inhibitory and cytotoxic activities. Chem Biodivers. 2014;11:73-84. doi:10.1002/cbdv.201300266
- Singh A, Srivastava PS, Lakshmikumaran M. Comparison of AFLP and SAMPL markers for assessment of intra-population genetic variation in Azadirachta indica A. Juss. Plant Sci. 2002;162:17-25. doi:10.1016/S0168-9452(01)00503-9
- Pandey G, Verma K, Singh M. Evaluation of phytochemical, antibacterial and free radical scavenging properties of Azadirachta Indica (neem) leaves. Int J Pharm Pharmaceut Sci. 2014;6:444-447.
- Romita P, Calogiuri G, Bellino M, et al. Allergic contact dermatitis caused by neem oil: an underrated allergen. Contact Dermatitis. 2019;81:133-134. doi:10.1111/cod. 13256
- Akihisa T, Noto T, Takahashi A, et al. Melanogenesis inhibitory, anti-inflammatory, and chemopreventive effects of limonoids from the seeds of Azadirachta indica A. Juss. (neem). J Oleo Sci. 2009;58:581-594.
- Subapriya R, Nagini S. Medicinal properties of neem leaves: a review. Curr Med Chem Anticancer Agents. 2005;5:149-156. doi:10.2174/1568011053174828
- Areekul S, Sinchaisri P, Tigvatananon S. Effect of Thai plant extracts on the Oriental fruit fly. I: toxicity test. Agriculture and Natural Resources. 1987;21:395-407.
- Rochanakij S, Thebtaranonth Y, Yenjai C, et al. Nimbolide, a constituent of Azadirachta indica, inhibits Plasmodium falciparum in culture. Southeast Asian J Trop Med Public Health. 1985;16:66-72.
- Sithisarn P, Supabphol R, Gritsanapan W. Antioxidant activity of Siamese neem tree (VP1209). J Ethnopharmacol. 2005;99:109-112. doi:10.1016/j.jep.2005.02.008
- Yin F, Lei XX, Cheng L, et al. Isolation and structure identification of the compounds from the seeds and leaves of Azadirachta indica A. Juss. J China Pharmaceut University. 2005;36:10-12.
- Su S, Cheng J, Zhang C, et al. Melanogenesis-inhibitory activities of limonoids and tricyclic diterpenoids from Azadirachta indica. Bioorganic Chemistry. 2020;100:103941. doi:j.bioorg.2020.103941
- Tulashie SK, Adjei F, Abraham J, et al. Potential of neem extracts as natural insecticide against fall armyworm (Spodoptera frugiperda (JE Smith)(Lepidoptera: Noctuidae). Case Stud Chem Environ Eng. 2021;4:100130. doi:10.1016/j.cscee.2021.100130
- Yashroy RC, Gupta PK. Neem-seed oil inhibits growth of termite surface-tunnels. Indian J Toxicol. 2000;7:49-50.
- Udeinya JI, Shu EN, Quakyi I, et al. An antimalarial neem leaf extract has both schizonticidal and gametocytocidal activities. Am J Therapeutics. 2008;15:108-110. doi:10.1097/MJT.0b013e31804c6d1d
- Bindurani R, Kumar K. Evaluation of antioxidant activity of hydro distilled extracts of leaf, heart wood and flower of Azadirachta indica. Int J Pharm Sci Rev Res. 2013;20:222.
- Alzohairy MA. Therapeutics role of Azadirachta indica (Neem) and their active constituents in diseases prevention and treatment [published online March 1, 2016]. Evid Based Complement Alternat Med. doi:10.1155/2016/7382506
- Schumacher M, Cerella C, Reuter S, et al. Anti-inflammatory, pro-apoptotic, and anti-proliferative effects of a methanolic neem (Azadirachta indica) leaf extract are mediated via modulation of the nuclear factor-κB pathway. Genes Nutr. 2011;6:149-160. doi:10.1007/s12263-010-0194-6
- Kaur G, Sarwar Alam M, Athar M. Nimbidin suppresses functions of macrophages and neutrophils: relevance to its anti-inflammatory mechanisms. Phytotherapy Res. 2004;18:419-424. doi:10.1002/ptr.1474
- Dhongade RK, Kavade SG, Damle RS. Neem oil poisoning. Indian Pediatr. 2008;45:56-57.
- Bhaskar MV, Pramod SJ, Jeevika MU, et al. MR imaging findings of neem oil poisoning. Am J Neuroradiol. 2010;31:E60-E61. doi:10.3174/ajnr.A2146
Commonly known as neem or nimba, Azadirachta indica traditionally has been used as an oil or poultice to lighten skin pigment and reduce joint inflammation. Neem is a drought-resistant evergreen tree with thin serrated leaves, white fragrant flowers, and olivelike fruit (Figure 1). This plant is indigenous to India but also is readily found within tropical and semitropical environments throughout the Middle East, Southeast Asia, North Africa, and Australia.

Traditional Uses
For more than 4000 years, neem leaves, bark, fruit, and seeds have been used in food, insecticide, and herbal medicine cross-culturally in Indian Ayurvedic medicine and across Southeast Asia, particularly in Cambodia, Laos, Thailand, Myanmar, and Vietnam.1-3 Because of its many essential nutrients—oleic acid, palmitic acid, stearic acid, linoleic acid, behenic acid, arachidic acid, and palmitoleic acid—and readily available nature, some ethnic groups include neem in their diet.4 Neem commonly is used as a seasoning in soups and rice, eaten as a cooked vegetable, infused into teas and tonics, and pickled with other spices.5
All parts of the neem tree—both externally and internally—have been utilized in traditional medicine for the treatment of various diseases and ailments. The flowers have been used to treat eye diseases and dyspepsia, the fruit has been employed as an anthelmintic, the seeds and leaves have been used for malaria treatment and insecticide, the stem bark has been used for the treatment of diarrhea, and the root bark has been used for skin diseases and inflammation.6 Neem oil is a yellow-brown bitter substance that often is utilized to treat skin diseases such as psoriasis, eczema, fungal infections, and abscesses.
Case Report—A 77-year-old man presented with a diffuse rash across the lower back. He reported that he had been using topical neem oil to alleviate lower back pain and arthritis for the last 6 months with noted relief and improvement of back pain. After roughly 3 to 4 months of using neem oil, he noted a rash on the lower back, bilateral flanks, and buttocks (Figure 2). The rash was asymptomatic, and he denied any pruritus, scaling, pain, or burning. The patient was referred to dermatology and received a diagnosis of chemical leukoderma secondary to contact with A indica. The patient was advised to stop using the topical neem oil, and the rash was simply monitored, as it was asymptomatic.

Bioactivity
Research has elucidated multiple bioactivity mechanisms of neem, including melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity.1,7-9 Literature on the diverse phytochemical components of A indica indicate high levels of limonoids, flavonoids, and triterpenoids that are responsible for much of its antioxidant, anti-inflammatory, and insecticide properties.1,10
Melanogenesis-Inhibitory Activity—To date, neem has been added to a number of cosmetic products used in Ayurvedic medicine. One study of isolated compounds in A indica showed superior inhibitory activities against melanogenesis with minimal toxicity to cells (86.5%–105.1% cell viability). Western blot analysis of samples extracted and isolated from neem root and bark showed melanogenesis-inhibitory activities in B16 melanoma cells through the inhibition of microphthalmia-associated transcription factor expression and decreased expression of tyrosinase, as well as tyrosinase-related proteins 1 and 2, which are largely responsible for melanin synthesis.11 In another study, A indica flowers and their extracted constituents—6-deacetylnimbin and kaempferide—suggest melanogenesis-inhibitory activities in B16 melanoma cells with little to no toxicity to the cells (81.0%–111.7% cell viability).1 In an evaluationof A indica seed extracts, some of the isolated limonoids and diterpenoids exhibited a marked melanogenesis-inhibitory effect (74%–91% reduction of melanin content) with no toxicity to the cell.5 All of these studies indicate that active compounds in neem root, bark, flowers, and seeds may be potential skin-lightening agents.
Toxicity Against Pests—Neem seeds have phytochemicals that convey some insecticidal properties. The seeds often are ground into a powder, combined with water, and sprayed onto crops to act as an insecticide. As a natural method of nonpesticidal management, A indica acts as an antifeedant, insect repellent, and egg-laying deterrent that protects crops from damage. Studies of A indica have noted effective nonpesticidal management against arthropod pests such as armyworm, termites, and the oriental fruit fly.7,12,13
Antimalarial Activity—One study indicated that nimbolide, a limonoid from the neem plant, demonstrated antimalarial activity against Plasmodium falciparum. In separate cultures of asexual parasites and mature gametocytes, parasite numbers were less than 50% of the number in control cultures (8.0% vs 8.5% parasitemia, respectively).14 Thus, the lower parasite numbers indicated by this study highlight the antimalarial utility of nimbolide and neem oil.
Antioxidant and Anti-inflammatory Activity—Neem bark has been reported to have considerable antioxidant activity due to its high phenolic content.1,15 One study showed that azadirachtin and nimbolide in neem exhibited concentration-dependent antiradical scavenging activity and antioxidant properties.16
The anti-inflammatory potential for neem may occur via the inhibition of the nuclear factor-κB signaling pathway, which is linked to cancer, inflammation, and apoptosis.17 It also has been observed that nimbidin within neem extracts—such as leaves, bark, and seed extract—suppresses the function of macrophages and neutrophils relevant to inflammation.16 Another study indicated neem’s anti-inflammatory activity due to the regulation of proinflammatory enzymes such as cyclooxygenase and lipoxygenase.18
Safety, Toxicity, and Risks
Ingestion—Although neem is safe to use in the general population, neem oil poisoning has been reported, particularly in young children. Ingesting large quantities of neem has resulted in vomiting, hepatic toxicity, metabolic acidosis, late neurologic sequelae, and encephalopathy in young children.19 The diagnosis of neem oil poisoning is based on patient history, clinical examination, and imaging findings. Poisoning can manifest as drowsiness, tachypnea, and generalized seizures.20
Topical Application—Topical use of neem appears to be safe if the substance is diluted with other ingredients. However, direct application to the skin is not advised, as it may cause leukoderma and could induce allergic contact dermatitis and other allergic reactions.4
Final Thoughts
The use of neem extract for disease prevention and treatment has been prevalent around the world since ancient times. Neem has been documented to possess melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity by means of tyrosinase inhibition, phytochemical production, limonoid expression, and nuclear factor-κB regulation, respectively. However, topical use of neem may trigger a cutaneous response, highlighting the importance of considering a diagnosis of neem oil–induced chemical leukoderma when patients present with a hypopigmented rash and relevant history.
Commonly known as neem or nimba, Azadirachta indica traditionally has been used as an oil or poultice to lighten skin pigment and reduce joint inflammation. Neem is a drought-resistant evergreen tree with thin serrated leaves, white fragrant flowers, and olivelike fruit (Figure 1). This plant is indigenous to India but also is readily found within tropical and semitropical environments throughout the Middle East, Southeast Asia, North Africa, and Australia.

Traditional Uses
For more than 4000 years, neem leaves, bark, fruit, and seeds have been used in food, insecticide, and herbal medicine cross-culturally in Indian Ayurvedic medicine and across Southeast Asia, particularly in Cambodia, Laos, Thailand, Myanmar, and Vietnam.1-3 Because of its many essential nutrients—oleic acid, palmitic acid, stearic acid, linoleic acid, behenic acid, arachidic acid, and palmitoleic acid—and readily available nature, some ethnic groups include neem in their diet.4 Neem commonly is used as a seasoning in soups and rice, eaten as a cooked vegetable, infused into teas and tonics, and pickled with other spices.5
All parts of the neem tree—both externally and internally—have been utilized in traditional medicine for the treatment of various diseases and ailments. The flowers have been used to treat eye diseases and dyspepsia, the fruit has been employed as an anthelmintic, the seeds and leaves have been used for malaria treatment and insecticide, the stem bark has been used for the treatment of diarrhea, and the root bark has been used for skin diseases and inflammation.6 Neem oil is a yellow-brown bitter substance that often is utilized to treat skin diseases such as psoriasis, eczema, fungal infections, and abscesses.
Case Report—A 77-year-old man presented with a diffuse rash across the lower back. He reported that he had been using topical neem oil to alleviate lower back pain and arthritis for the last 6 months with noted relief and improvement of back pain. After roughly 3 to 4 months of using neem oil, he noted a rash on the lower back, bilateral flanks, and buttocks (Figure 2). The rash was asymptomatic, and he denied any pruritus, scaling, pain, or burning. The patient was referred to dermatology and received a diagnosis of chemical leukoderma secondary to contact with A indica. The patient was advised to stop using the topical neem oil, and the rash was simply monitored, as it was asymptomatic.

Bioactivity
Research has elucidated multiple bioactivity mechanisms of neem, including melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity.1,7-9 Literature on the diverse phytochemical components of A indica indicate high levels of limonoids, flavonoids, and triterpenoids that are responsible for much of its antioxidant, anti-inflammatory, and insecticide properties.1,10
Melanogenesis-Inhibitory Activity—To date, neem has been added to a number of cosmetic products used in Ayurvedic medicine. One study of isolated compounds in A indica showed superior inhibitory activities against melanogenesis with minimal toxicity to cells (86.5%–105.1% cell viability). Western blot analysis of samples extracted and isolated from neem root and bark showed melanogenesis-inhibitory activities in B16 melanoma cells through the inhibition of microphthalmia-associated transcription factor expression and decreased expression of tyrosinase, as well as tyrosinase-related proteins 1 and 2, which are largely responsible for melanin synthesis.11 In another study, A indica flowers and their extracted constituents—6-deacetylnimbin and kaempferide—suggest melanogenesis-inhibitory activities in B16 melanoma cells with little to no toxicity to the cells (81.0%–111.7% cell viability).1 In an evaluationof A indica seed extracts, some of the isolated limonoids and diterpenoids exhibited a marked melanogenesis-inhibitory effect (74%–91% reduction of melanin content) with no toxicity to the cell.5 All of these studies indicate that active compounds in neem root, bark, flowers, and seeds may be potential skin-lightening agents.
Toxicity Against Pests—Neem seeds have phytochemicals that convey some insecticidal properties. The seeds often are ground into a powder, combined with water, and sprayed onto crops to act as an insecticide. As a natural method of nonpesticidal management, A indica acts as an antifeedant, insect repellent, and egg-laying deterrent that protects crops from damage. Studies of A indica have noted effective nonpesticidal management against arthropod pests such as armyworm, termites, and the oriental fruit fly.7,12,13
Antimalarial Activity—One study indicated that nimbolide, a limonoid from the neem plant, demonstrated antimalarial activity against Plasmodium falciparum. In separate cultures of asexual parasites and mature gametocytes, parasite numbers were less than 50% of the number in control cultures (8.0% vs 8.5% parasitemia, respectively).14 Thus, the lower parasite numbers indicated by this study highlight the antimalarial utility of nimbolide and neem oil.
Antioxidant and Anti-inflammatory Activity—Neem bark has been reported to have considerable antioxidant activity due to its high phenolic content.1,15 One study showed that azadirachtin and nimbolide in neem exhibited concentration-dependent antiradical scavenging activity and antioxidant properties.16
The anti-inflammatory potential for neem may occur via the inhibition of the nuclear factor-κB signaling pathway, which is linked to cancer, inflammation, and apoptosis.17 It also has been observed that nimbidin within neem extracts—such as leaves, bark, and seed extract—suppresses the function of macrophages and neutrophils relevant to inflammation.16 Another study indicated neem’s anti-inflammatory activity due to the regulation of proinflammatory enzymes such as cyclooxygenase and lipoxygenase.18
Safety, Toxicity, and Risks
Ingestion—Although neem is safe to use in the general population, neem oil poisoning has been reported, particularly in young children. Ingesting large quantities of neem has resulted in vomiting, hepatic toxicity, metabolic acidosis, late neurologic sequelae, and encephalopathy in young children.19 The diagnosis of neem oil poisoning is based on patient history, clinical examination, and imaging findings. Poisoning can manifest as drowsiness, tachypnea, and generalized seizures.20
Topical Application—Topical use of neem appears to be safe if the substance is diluted with other ingredients. However, direct application to the skin is not advised, as it may cause leukoderma and could induce allergic contact dermatitis and other allergic reactions.4
Final Thoughts
The use of neem extract for disease prevention and treatment has been prevalent around the world since ancient times. Neem has been documented to possess melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity by means of tyrosinase inhibition, phytochemical production, limonoid expression, and nuclear factor-κB regulation, respectively. However, topical use of neem may trigger a cutaneous response, highlighting the importance of considering a diagnosis of neem oil–induced chemical leukoderma when patients present with a hypopigmented rash and relevant history.
- Kitdamrongtham W, Ishii K, Ebina K, et al. Limonoids and flavonoids from the flowers of Azadirachta indica var. siamensis, and their melanogenesis-inhibitory and cytotoxic activities. Chem Biodivers. 2014;11:73-84. doi:10.1002/cbdv.201300266
- Singh A, Srivastava PS, Lakshmikumaran M. Comparison of AFLP and SAMPL markers for assessment of intra-population genetic variation in Azadirachta indica A. Juss. Plant Sci. 2002;162:17-25. doi:10.1016/S0168-9452(01)00503-9
- Pandey G, Verma K, Singh M. Evaluation of phytochemical, antibacterial and free radical scavenging properties of Azadirachta Indica (neem) leaves. Int J Pharm Pharmaceut Sci. 2014;6:444-447.
- Romita P, Calogiuri G, Bellino M, et al. Allergic contact dermatitis caused by neem oil: an underrated allergen. Contact Dermatitis. 2019;81:133-134. doi:10.1111/cod. 13256
- Akihisa T, Noto T, Takahashi A, et al. Melanogenesis inhibitory, anti-inflammatory, and chemopreventive effects of limonoids from the seeds of Azadirachta indica A. Juss. (neem). J Oleo Sci. 2009;58:581-594.
- Subapriya R, Nagini S. Medicinal properties of neem leaves: a review. Curr Med Chem Anticancer Agents. 2005;5:149-156. doi:10.2174/1568011053174828
- Areekul S, Sinchaisri P, Tigvatananon S. Effect of Thai plant extracts on the Oriental fruit fly. I: toxicity test. Agriculture and Natural Resources. 1987;21:395-407.
- Rochanakij S, Thebtaranonth Y, Yenjai C, et al. Nimbolide, a constituent of Azadirachta indica, inhibits Plasmodium falciparum in culture. Southeast Asian J Trop Med Public Health. 1985;16:66-72.
- Sithisarn P, Supabphol R, Gritsanapan W. Antioxidant activity of Siamese neem tree (VP1209). J Ethnopharmacol. 2005;99:109-112. doi:10.1016/j.jep.2005.02.008
- Yin F, Lei XX, Cheng L, et al. Isolation and structure identification of the compounds from the seeds and leaves of Azadirachta indica A. Juss. J China Pharmaceut University. 2005;36:10-12.
- Su S, Cheng J, Zhang C, et al. Melanogenesis-inhibitory activities of limonoids and tricyclic diterpenoids from Azadirachta indica. Bioorganic Chemistry. 2020;100:103941. doi:j.bioorg.2020.103941
- Tulashie SK, Adjei F, Abraham J, et al. Potential of neem extracts as natural insecticide against fall armyworm (Spodoptera frugiperda (JE Smith)(Lepidoptera: Noctuidae). Case Stud Chem Environ Eng. 2021;4:100130. doi:10.1016/j.cscee.2021.100130
- Yashroy RC, Gupta PK. Neem-seed oil inhibits growth of termite surface-tunnels. Indian J Toxicol. 2000;7:49-50.
- Udeinya JI, Shu EN, Quakyi I, et al. An antimalarial neem leaf extract has both schizonticidal and gametocytocidal activities. Am J Therapeutics. 2008;15:108-110. doi:10.1097/MJT.0b013e31804c6d1d
- Bindurani R, Kumar K. Evaluation of antioxidant activity of hydro distilled extracts of leaf, heart wood and flower of Azadirachta indica. Int J Pharm Sci Rev Res. 2013;20:222.
- Alzohairy MA. Therapeutics role of Azadirachta indica (Neem) and their active constituents in diseases prevention and treatment [published online March 1, 2016]. Evid Based Complement Alternat Med. doi:10.1155/2016/7382506
- Schumacher M, Cerella C, Reuter S, et al. Anti-inflammatory, pro-apoptotic, and anti-proliferative effects of a methanolic neem (Azadirachta indica) leaf extract are mediated via modulation of the nuclear factor-κB pathway. Genes Nutr. 2011;6:149-160. doi:10.1007/s12263-010-0194-6
- Kaur G, Sarwar Alam M, Athar M. Nimbidin suppresses functions of macrophages and neutrophils: relevance to its anti-inflammatory mechanisms. Phytotherapy Res. 2004;18:419-424. doi:10.1002/ptr.1474
- Dhongade RK, Kavade SG, Damle RS. Neem oil poisoning. Indian Pediatr. 2008;45:56-57.
- Bhaskar MV, Pramod SJ, Jeevika MU, et al. MR imaging findings of neem oil poisoning. Am J Neuroradiol. 2010;31:E60-E61. doi:10.3174/ajnr.A2146
- Kitdamrongtham W, Ishii K, Ebina K, et al. Limonoids and flavonoids from the flowers of Azadirachta indica var. siamensis, and their melanogenesis-inhibitory and cytotoxic activities. Chem Biodivers. 2014;11:73-84. doi:10.1002/cbdv.201300266
- Singh A, Srivastava PS, Lakshmikumaran M. Comparison of AFLP and SAMPL markers for assessment of intra-population genetic variation in Azadirachta indica A. Juss. Plant Sci. 2002;162:17-25. doi:10.1016/S0168-9452(01)00503-9
- Pandey G, Verma K, Singh M. Evaluation of phytochemical, antibacterial and free radical scavenging properties of Azadirachta Indica (neem) leaves. Int J Pharm Pharmaceut Sci. 2014;6:444-447.
- Romita P, Calogiuri G, Bellino M, et al. Allergic contact dermatitis caused by neem oil: an underrated allergen. Contact Dermatitis. 2019;81:133-134. doi:10.1111/cod. 13256
- Akihisa T, Noto T, Takahashi A, et al. Melanogenesis inhibitory, anti-inflammatory, and chemopreventive effects of limonoids from the seeds of Azadirachta indica A. Juss. (neem). J Oleo Sci. 2009;58:581-594.
- Subapriya R, Nagini S. Medicinal properties of neem leaves: a review. Curr Med Chem Anticancer Agents. 2005;5:149-156. doi:10.2174/1568011053174828
- Areekul S, Sinchaisri P, Tigvatananon S. Effect of Thai plant extracts on the Oriental fruit fly. I: toxicity test. Agriculture and Natural Resources. 1987;21:395-407.
- Rochanakij S, Thebtaranonth Y, Yenjai C, et al. Nimbolide, a constituent of Azadirachta indica, inhibits Plasmodium falciparum in culture. Southeast Asian J Trop Med Public Health. 1985;16:66-72.
- Sithisarn P, Supabphol R, Gritsanapan W. Antioxidant activity of Siamese neem tree (VP1209). J Ethnopharmacol. 2005;99:109-112. doi:10.1016/j.jep.2005.02.008
- Yin F, Lei XX, Cheng L, et al. Isolation and structure identification of the compounds from the seeds and leaves of Azadirachta indica A. Juss. J China Pharmaceut University. 2005;36:10-12.
- Su S, Cheng J, Zhang C, et al. Melanogenesis-inhibitory activities of limonoids and tricyclic diterpenoids from Azadirachta indica. Bioorganic Chemistry. 2020;100:103941. doi:j.bioorg.2020.103941
- Tulashie SK, Adjei F, Abraham J, et al. Potential of neem extracts as natural insecticide against fall armyworm (Spodoptera frugiperda (JE Smith)(Lepidoptera: Noctuidae). Case Stud Chem Environ Eng. 2021;4:100130. doi:10.1016/j.cscee.2021.100130
- Yashroy RC, Gupta PK. Neem-seed oil inhibits growth of termite surface-tunnels. Indian J Toxicol. 2000;7:49-50.
- Udeinya JI, Shu EN, Quakyi I, et al. An antimalarial neem leaf extract has both schizonticidal and gametocytocidal activities. Am J Therapeutics. 2008;15:108-110. doi:10.1097/MJT.0b013e31804c6d1d
- Bindurani R, Kumar K. Evaluation of antioxidant activity of hydro distilled extracts of leaf, heart wood and flower of Azadirachta indica. Int J Pharm Sci Rev Res. 2013;20:222.
- Alzohairy MA. Therapeutics role of Azadirachta indica (Neem) and their active constituents in diseases prevention and treatment [published online March 1, 2016]. Evid Based Complement Alternat Med. doi:10.1155/2016/7382506
- Schumacher M, Cerella C, Reuter S, et al. Anti-inflammatory, pro-apoptotic, and anti-proliferative effects of a methanolic neem (Azadirachta indica) leaf extract are mediated via modulation of the nuclear factor-κB pathway. Genes Nutr. 2011;6:149-160. doi:10.1007/s12263-010-0194-6
- Kaur G, Sarwar Alam M, Athar M. Nimbidin suppresses functions of macrophages and neutrophils: relevance to its anti-inflammatory mechanisms. Phytotherapy Res. 2004;18:419-424. doi:10.1002/ptr.1474
- Dhongade RK, Kavade SG, Damle RS. Neem oil poisoning. Indian Pediatr. 2008;45:56-57.
- Bhaskar MV, Pramod SJ, Jeevika MU, et al. MR imaging findings of neem oil poisoning. Am J Neuroradiol. 2010;31:E60-E61. doi:10.3174/ajnr.A2146
Practice Points
- Neem is a traditional herb with various bioactivities, such as melanogenesis-inhibitory activity, toxicity against pests, antimalarial activity, and antioxidant activity.
- Neem should be used with caution as a remedy because of its skin-lightening properties, which are attributed to melanogenesis-inhibitory activity via tyrosinase inhibition.
- Chemical leukoderma should be included in the differential diagnosis when a patient presents with a hypopigmented rash after topical use of neem products.
Analysis of Online Diet Recommendations for Vitiligo
Internet platforms have become a common source of medical information for individuals with a broad range of skin conditions including vitiligo. The prevalence of vitiligo among US adults ranges from 0.76% to 1.11%, with approximately 40% of adult cases of vitiligo in the United States remaining undiagnosed.1 The vitiligo community has become more inquisitive of the relationship between diet and vitiligo, turning to online sources for suggestions on diet modifications that may be beneficial for their condition. Although there is an abundance of online information, few diets or foods have been medically recognized to definitively improve or worsen vitiligo symptoms. We reviewed the top online web pages accessible to the public regarding diet suggestions that affect vitiligo symptoms. We then compared these online results to published peer-reviewed scientific literature.
Methods
Two independent online searches were performed by Researcher 1 (Y.A.) and Researcher 2 (I.M.) using Google Advanced Search. The independent searches were performed by the reviewers in neighboring areas of Chicago, Illinois, using the same Internet browser (Google Chrome). The primary search terms were diet and vitiligo along with the optional additional terms dietary supplement(s), food(s), nutrition, herb(s), or vitamin(s). Our search included any web pages published or updated from January 1, 2010, to December 31, 2021, and originally scribed in the English language. The domains “.com,” “.org,” “.edu,” and “.cc” were included.
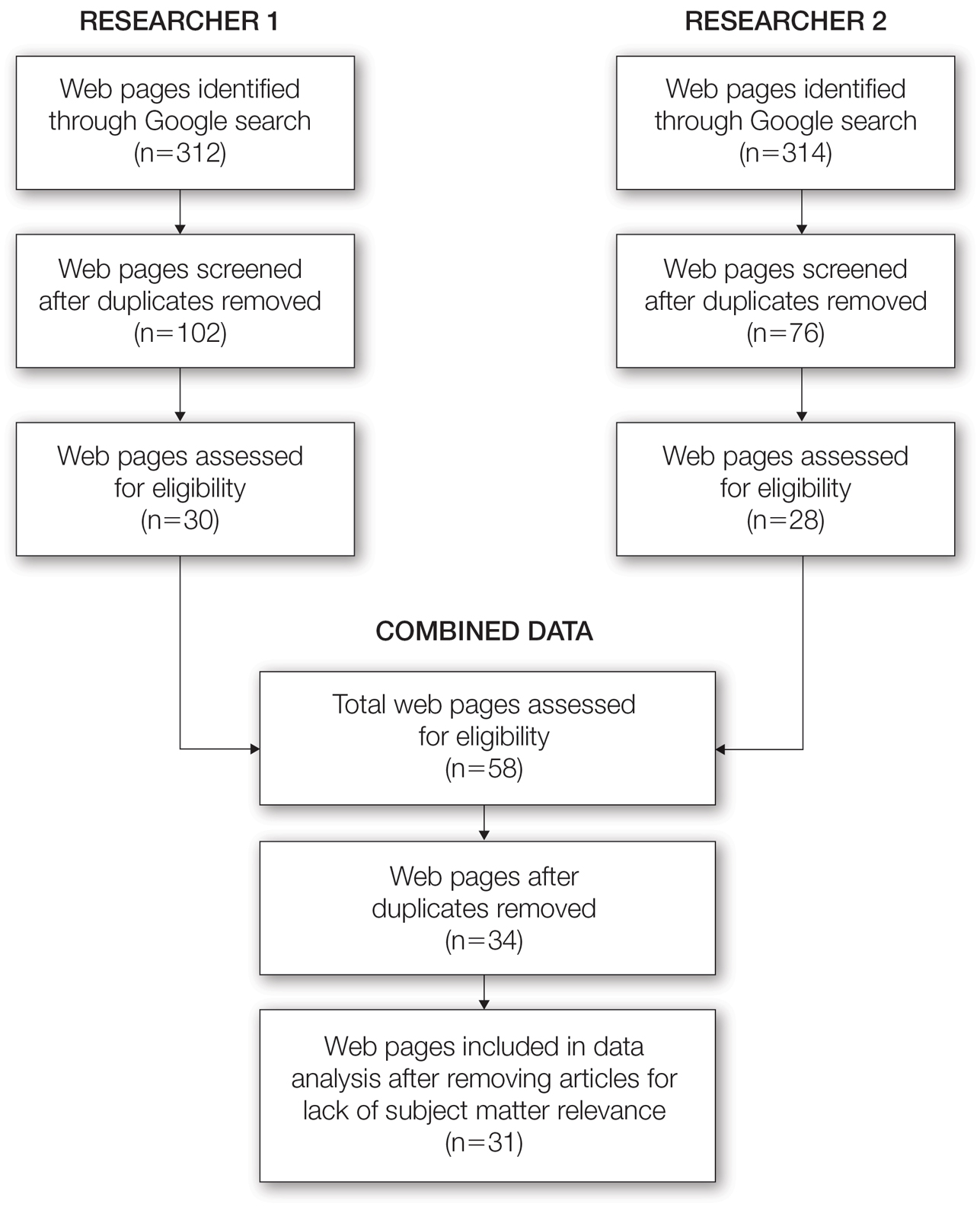
From this initial search, Researcher 1 identified 312 web pages and Researcher 2 identified 314 web pages. Each reviewer sorted their respective search results to identify the number of eligible records to be screened. Records were defined as unique web pages that met the search criteria. After removing duplicates, Researcher 1 screened 102 web pages and Researcher 2 screened 76 web pages. Of these records, web pages were excluded if they did not include any diet recommendations for vitiligo patients. Each reviewer independently created a list of eligible records, and the independent lists were then merged for a total of 58 web pages. Among these 58 web pages, there were 24 duplicate records and 3 records that were deemed ineligible for the study due to lack of subject matter relevance. A final total of 31 web pages were included in the data analysis (Figure). Of the 31 records selected, the reviewers jointly evaluated each web page and recorded the diet components that were recommended for individuals with vitiligo to either include or avoid (eTable).
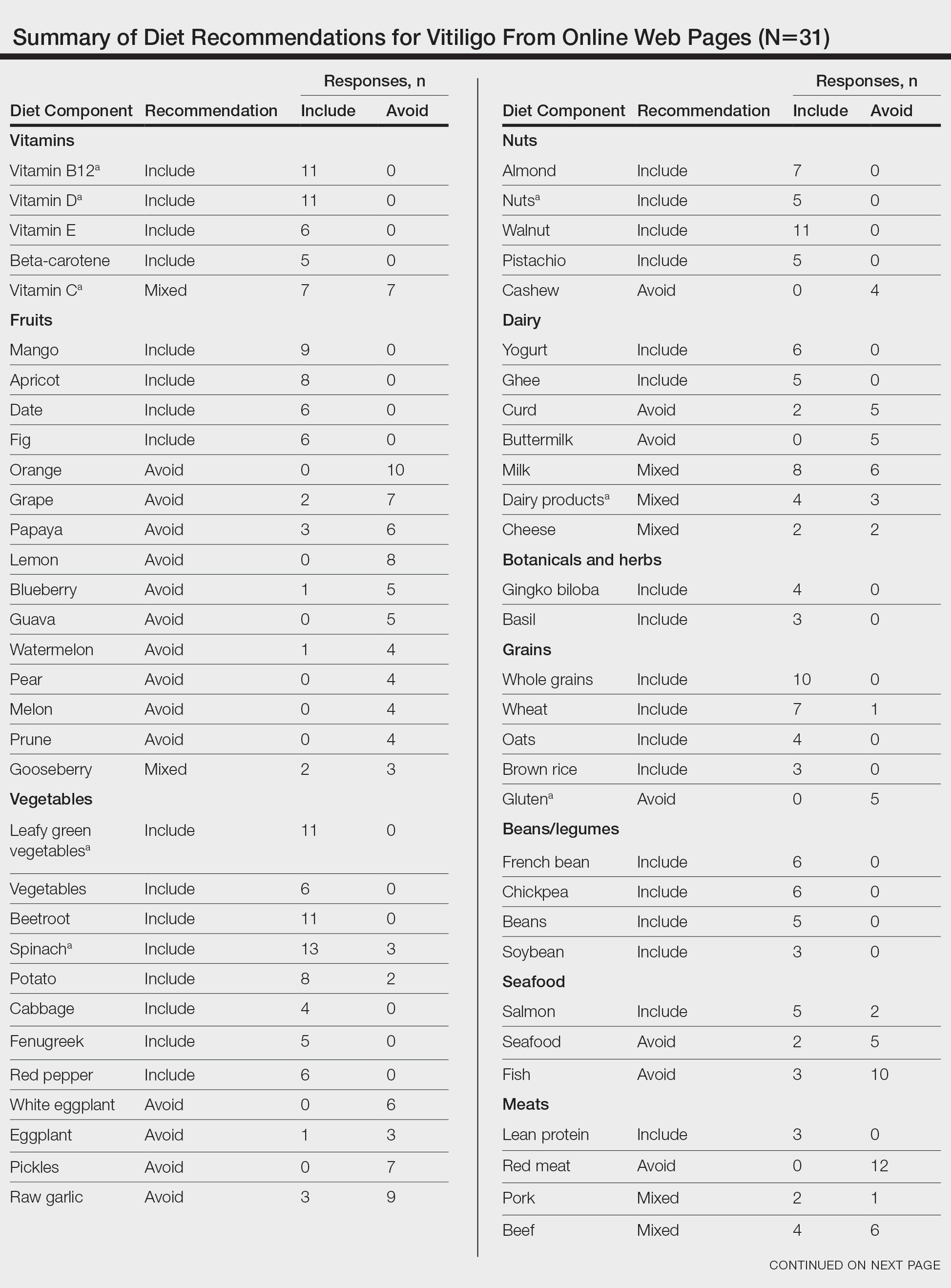
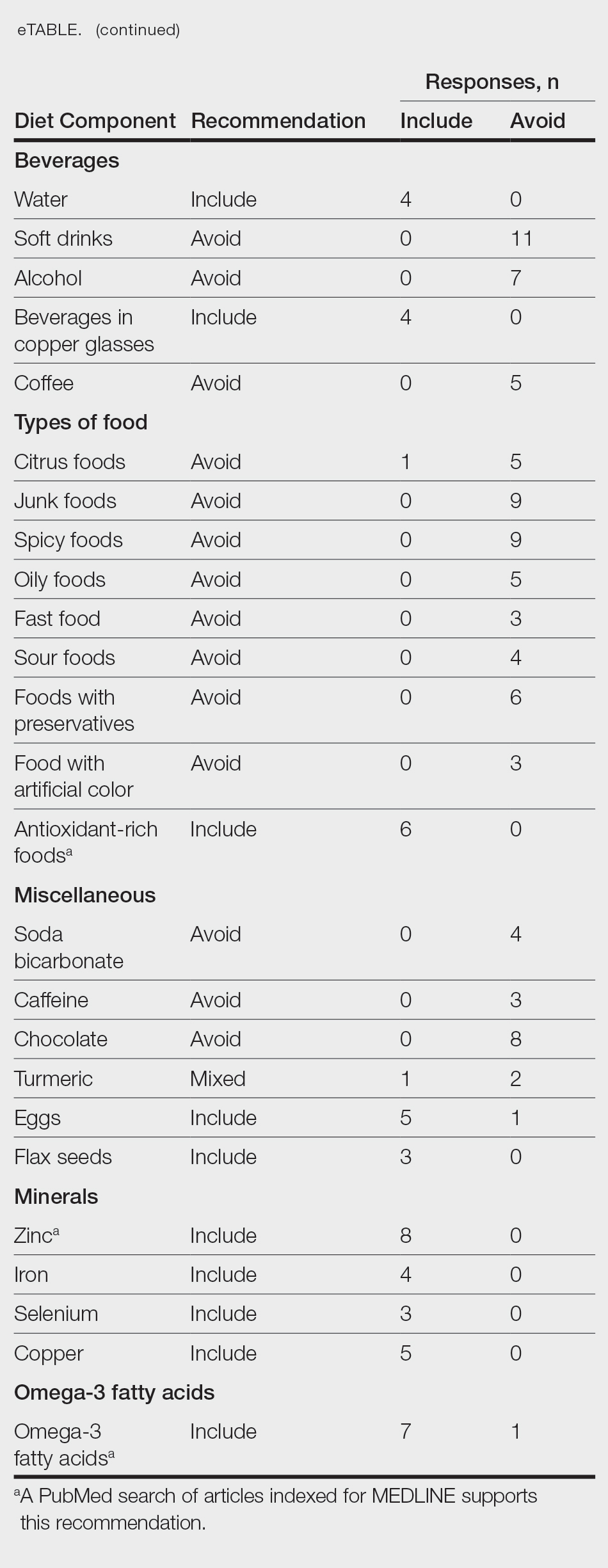
For comparison and support from published scientific literature, a search of PubMed articles indexed for MEDLINE was conducted using the terms diet and vitiligo. Relevant human clinical studies published in the English-language literature were reviewed for content regarding the relationship between diet and vitiligo.
Results
Our online search revealed an abundance of information regarding various dietary modifications suggested to aid in the management of vitiligo symptoms. Most web pages (27/31 [87%]) were not authored by medical professionals or dermatologists. There were 27 diet components mentioned 8 or more times within the 31 total web pages. These diet components were selected for further review via PubMed. Each item was searched on PubMed using the term “[respective diet component] and vitiligo” among all published literature in the English language. Our study focused on summarizing the data on dietary components for which we were able to gather scientific support. These data have been organized into the following categories: vitamins, fruits, omega-3 fatty acids, grains, minerals, vegetables, and nuts.
Vitamins—The online literature recommended inclusion of vitamin supplements, in particular vitamins D and B12, which aligned with published scientific literature.2,3 Eleven of 31 (35%) web pages recommended vitamin D in vitiligo. A 2010 study analyzing patients with vitiligo vulgaris (N=45) found that 68.9% of the cohort had insufficient (<30 ng/mL) 25-hydroxyvitamin D levels.2 A prospective study of 30 individuals found that the use of tacrolimus ointment plus oral vitamin D supplementation was found to be more successful in repigmentation than topical tacrolimus alone.3 Vitamin D dosage ranged from 1500 IU/d if the patient’s serum 25-hydroxyvitamin D levels were less than 20 ng/mL to 3000 IU/d if the serum levels were less than 10 ng/mL for 6 months.
Dairy products are a source of vitamin D.2,3 Of the web pages that mentioned dairy, a subtle majority (4/7 [57%]) recommended the inclusion of dairy products. Although many web pages did not specify whether oral vitamin D supplementation vs dietary food consumption is preferred, a 2013 controlled study of 16 vitiligo patients who received high doses of vitamin D supplementation with a low-calcium diet found that 4 patients showed 1% to 25% repigmentation, 5 patients showed 26% to 50% repigmentation, and 5 patients showed 51% to 75% repigmentation of the affected areas.4
Eleven of 31 (35%) web pages recommended inclusion of vitamin B12 supplementation in vitiligo. A 2-year study with 100 participants showed that supplementation with folic acid and vitamin B12 along with sun exposure yielded more effective repigmentation than either vitamins or sun exposure alone.5 An additional hypothesis suggested vitamin B12 may aid in repigmentation through its role in the homocysteine pathway. Although the theory is unproven, it is proposed that inhibition of homocysteine via vitamin B12 or folic acid supplementation may play a role in reducing melanocyte destruction and restoring melanin synthesis.6
There were mixed recommendations regarding vitamin C via supplementation and/or eating citrus fruits such as oranges. Although there are limited clinical studies on the use of vitamin C and the treatment of vitiligo, a 6-year prospective study from Madagascar consisting of approximately 300 participants with vitiligo who were treated with a combination of topical corticosteroids, oral vitamin C, and oral vitamin B12 supplementation showed excellent repigmentation (defined by repigmentation of more than 76% of the originally affected area) in 50 participants.7
Fruits—Most web pages had mixed recommendations on whether to include or avoid certain fruits. Interestingly, inclusion of mangoes and apricots in the diet were highly recommended (9/31 [29%] and 8/31 [26%], respectively) while fruits such as oranges, lemons, papayas, and grapes were discouraged (10/31 [32%], 8/31 [26%], 6/31 [19%], and 7/31 [23%], respectively). Although some web pages suggested that vitamin C–rich produce including citrus and berries may help to increase melanin formation, others strongly suggested avoiding these fruits. There is limited information on the effects of citrus on vitiligo, but a 2022 study indicated that 5-demethylnobiletin, a flavonoid found in sweet citrus fruits, may stimulate melanin synthesis, which can possibly be beneficial for vitiligo.8
Omega-3 Fatty Acids—Seven of 31 (23%) web pages recommended the inclusion of omega-3 fatty acids for their role as antioxidants to improve vitiligo symptoms. Research has indicated a strong association between vitiligo and oxidative stress.9 A 2007 controlled clinical trial that included 28 vitiligo patients demonstrated that oral antioxidant supplementation in combination with narrowband UVB phototherapy can significantly decrease vitiligo-associated oxidative stress (P<.05); 8 of 17 (47%) patients in the treatment group saw greater than 75% repigmentation after antioxidant treatment.10
Grains—Five of 31 (16%) web pages suggested avoiding gluten—a protein naturally found in some grains including wheat, barley, and rye—to improve vitiligo symptoms. A 2021 review suggested that a gluten-free diet may be effective in managing celiac disease, and it is hypothesized that vitiligo may be managed with similar dietary adjustments.11 Studies have shown that celiac disease and vitiligo—both autoimmune conditions—involve IL-2, IL-6, IL-7, and IL-21 in their disease pathways.12,13 Their shared immunogenic mechanism may account for similar management options.
Upon review, 2 case reports were identified that discussed a relationship between a gluten-free diet and vitiligo symptom improvement. In one report, a 9-year-old child diagnosed with both celiac disease and vitiligo saw intense repigmentation of the skin after adhering to a gluten-free diet for 1 year.14 Another case study reported a 22-year-old woman with vitiligo whose symptoms improved after 1 month of a gluten-free diet following 2 years of failed treatment with a topical steroid and phototherapy.15
Seven of 31 (23%) web pages suggested that individuals with vitiligo should include wheat in their diet. There is no published literature discussing the relationship between vitiligo and wheat. Of the 31 web pages reviewed, 10 (32%) suggested including whole grain. There is no relevant scientific evidence or hypotheses describing how whole grains may be beneficial in vitiligo.
Minerals—Eight of 31 (26%) web pages suggested including zinc in the diet to improve vitiligo symptoms. A 2020 study evaluated how different serum levels of zinc in vitiligo patients might be affiliated with interleukin activity. Fifty patients diagnosed with active vitiligo were tested for serum levels of zinc, IL-4, IL-6, and IL-17.16 The results showed that mean serum levels of zinc were lower in vitiligo patients compared with patients without vitiligo. The study concluded that zinc could possibly be used as a supplement to improve vitiligo, though the dosage needs to be further studied and confirmed.16
Vegetables—Eleven of 31 (35%) web pages recommended leafy green vegetables and 13 of 31 (42%) recommended spinach for patients with vitiligo. Spinach and other leafy green vegetables are known to be rich in antioxidants, which may have protective effects against reactive oxygen species that are thought to contribute to vitiligo progression.17,18
Nuts—Walnuts were recommended in 11 of 31 (35%) web pages. Nuts may be beneficial in reducing inflammation and providing protection against oxidative stress.9 However, there is no specific scientific literature that supports the inclusion of nuts in the diet to manage vitiligo symptoms.
Comment
With a growing amount of research suggesting that diet modifications may contribute to management of certain skin conditions, vitiligo patients often inquire about foods or supplements that may help improve their condition.19 Our review highlighted what information was available to the public regarding diet and vitiligo, with preliminary support of the following primary diet components: vitamin D, vitamin B12, zinc, and omega-3 fatty acids. Our review showed no support in the literature for the items that were recommended to avoid. It is important to note that 27 of 31 (87%) web pages from our online search were not authored by medical professionals or dermatologists. Additionally, many web pages suggested conflicting information, making it difficult to draw concrete conclusions about what diet modifications will be beneficial to the vitiligo community. Further controlled clinical trials are warranted due to the lack of formal studies that assess the relationship between diet and vitiligo.
- Gandhi K, Ezzedine K, Anastassopoulos KP, et al. Prevalence of vitiligo among adults in the United States. JAMA Dermatol. 2022;158:43-50. doi:10.1001/jamadermatol.2021.4724
- Silverberg JI, Silverberg AI, Malka E, et al. A pilot study assessing the role of 25 hydroxy vitamin D levels in patients with vitiligo vulgaris. J Am Acad Dermatol. 2010;62:937-941. doi:10.1016/j.jaad.2009.11.024
- Karagüzel G, Sakarya NP, Bahadır S, et al. Vitamin D status and the effects of oral vitamin D treatment in children with vitiligo: a prospective study. Clin Nutr ESPEN. 2016;15:28-31. doi:10.1016/j.clnesp.2016.05.006.
- Finamor DC, Sinigaglia-Coimbra R, Neves LC, et al. A pilot study assessing the effect of prolonged administration of high daily doses of vitamin D on the clinical course of vitiligo and psoriasis. Dermatoendocrinol. 2013;5:222-234. doi:10.4161/derm.24808
- Juhlin L, Olsson MJ. Improvement of vitiligo after oral treatment with vitamin B12 and folic acid and the importance of sun exposure. Acta Derm Venereol. 1997;77:460-462. doi:10.2340/000155555577460462
- Chen J, Zhuang T, Chen J, et al. Homocysteine induces melanocytes apoptosis via PERK-eIF2α-CHOP pathway in vitiligo. Clin Sci (Lond). 2020;134:1127-1141. doi:10.1042/CS20200218
- Sendrasoa FA, Ranaivo IM, Sata M, et al. Treatment responses in patients with vitiligo to very potent topical corticosteroids combined with vitamin therapy in Madagascar. Int J Dermatol. 2019;58:908-911. doi:10.1111/ijd.14510
- Wang HM, Qu LQ, Ng JPL, et al. Natural citrus flavanone 5-demethylnobiletin stimulates melanogenesis through the activation of cAMP/CREB pathway in B16F10 cells. Phytomedicine. 2022;98:153941. doi:10.1016/j.phymed.2022.153941
- Ros E. Health benefits of nut consumption. Nutrients. 2010;2:652-682.
- Dell’Anna ML, Mastrofrancesco A, Sala R, et al. Antioxidants and narrow band-UVB in the treatment of vitiligo: a double-blind placebo controlled trial. Clin Exp Dermatol. 2007;32:631-636.
- Gastrointestinal microbiome and gluten in celiac disease. Ann Med. 2021;53:1797-1805. doi:10.1080/07853890.2021.1990392
- Forabosco P, Neuhausen SL, Greco L, et al. Meta-analysis of genome-wide linkage studies in celiac disease. Hum Hered. 2009;68:223-230. doi:10.1159/000228920
- Akbulut UE, Çebi AH, Sag˘ E, et al. Interleukin-6 and interleukin-17 gene polymorphism association with celiac disease in children. Turk J Gastroenterol. 2017;28:471-475. doi:10.5152/tjg.2017.17092
- Rodríguez-García C, González-Hernández S, Pérez-Robayna N, et al. Repigmentation of vitiligo lesions in a child with celiac disease after a gluten-free diet. Pediatr Dermatol. 2011;28:209-210. doi:10.1111/j.1525-1470.2011.01388.x
- Khandalavala BN, Nirmalraj MC. Rapid partial repigmentation ofvitiligo in a young female adult with a gluten-free diet. Case Rep Dermatol. 2014;6:283-287.
- Sanad EM, El-Fallah AA, Al-Doori AR, et al. Serum zinc and inflammatory cytokines in vitiligo. J Clin Aesthet Dermatol. 2020;13:(12 suppl 1):S29-S33.
- Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci U S A. 1993;90:7915-7922. doi:10.1073/pnas.90.17.7915
- Xian D, Guo M, Xu J, et al. Current evidence to support the therapeutic potential of flavonoids in oxidative stress-related dermatoses. Redox Rep. 2021;26:134-146. doi:10.1080 /13510002.2021.1962094
- Katta R, Kramer MJ. Skin and diet: an update on the role of dietary change as a treatment strategy for skin disease. Skin Therapy Lett. 2018;23:1-5.
Internet platforms have become a common source of medical information for individuals with a broad range of skin conditions including vitiligo. The prevalence of vitiligo among US adults ranges from 0.76% to 1.11%, with approximately 40% of adult cases of vitiligo in the United States remaining undiagnosed.1 The vitiligo community has become more inquisitive of the relationship between diet and vitiligo, turning to online sources for suggestions on diet modifications that may be beneficial for their condition. Although there is an abundance of online information, few diets or foods have been medically recognized to definitively improve or worsen vitiligo symptoms. We reviewed the top online web pages accessible to the public regarding diet suggestions that affect vitiligo symptoms. We then compared these online results to published peer-reviewed scientific literature.
Methods
Two independent online searches were performed by Researcher 1 (Y.A.) and Researcher 2 (I.M.) using Google Advanced Search. The independent searches were performed by the reviewers in neighboring areas of Chicago, Illinois, using the same Internet browser (Google Chrome). The primary search terms were diet and vitiligo along with the optional additional terms dietary supplement(s), food(s), nutrition, herb(s), or vitamin(s). Our search included any web pages published or updated from January 1, 2010, to December 31, 2021, and originally scribed in the English language. The domains “.com,” “.org,” “.edu,” and “.cc” were included.

From this initial search, Researcher 1 identified 312 web pages and Researcher 2 identified 314 web pages. Each reviewer sorted their respective search results to identify the number of eligible records to be screened. Records were defined as unique web pages that met the search criteria. After removing duplicates, Researcher 1 screened 102 web pages and Researcher 2 screened 76 web pages. Of these records, web pages were excluded if they did not include any diet recommendations for vitiligo patients. Each reviewer independently created a list of eligible records, and the independent lists were then merged for a total of 58 web pages. Among these 58 web pages, there were 24 duplicate records and 3 records that were deemed ineligible for the study due to lack of subject matter relevance. A final total of 31 web pages were included in the data analysis (Figure). Of the 31 records selected, the reviewers jointly evaluated each web page and recorded the diet components that were recommended for individuals with vitiligo to either include or avoid (eTable).


For comparison and support from published scientific literature, a search of PubMed articles indexed for MEDLINE was conducted using the terms diet and vitiligo. Relevant human clinical studies published in the English-language literature were reviewed for content regarding the relationship between diet and vitiligo.
Results
Our online search revealed an abundance of information regarding various dietary modifications suggested to aid in the management of vitiligo symptoms. Most web pages (27/31 [87%]) were not authored by medical professionals or dermatologists. There were 27 diet components mentioned 8 or more times within the 31 total web pages. These diet components were selected for further review via PubMed. Each item was searched on PubMed using the term “[respective diet component] and vitiligo” among all published literature in the English language. Our study focused on summarizing the data on dietary components for which we were able to gather scientific support. These data have been organized into the following categories: vitamins, fruits, omega-3 fatty acids, grains, minerals, vegetables, and nuts.
Vitamins—The online literature recommended inclusion of vitamin supplements, in particular vitamins D and B12, which aligned with published scientific literature.2,3 Eleven of 31 (35%) web pages recommended vitamin D in vitiligo. A 2010 study analyzing patients with vitiligo vulgaris (N=45) found that 68.9% of the cohort had insufficient (<30 ng/mL) 25-hydroxyvitamin D levels.2 A prospective study of 30 individuals found that the use of tacrolimus ointment plus oral vitamin D supplementation was found to be more successful in repigmentation than topical tacrolimus alone.3 Vitamin D dosage ranged from 1500 IU/d if the patient’s serum 25-hydroxyvitamin D levels were less than 20 ng/mL to 3000 IU/d if the serum levels were less than 10 ng/mL for 6 months.
Dairy products are a source of vitamin D.2,3 Of the web pages that mentioned dairy, a subtle majority (4/7 [57%]) recommended the inclusion of dairy products. Although many web pages did not specify whether oral vitamin D supplementation vs dietary food consumption is preferred, a 2013 controlled study of 16 vitiligo patients who received high doses of vitamin D supplementation with a low-calcium diet found that 4 patients showed 1% to 25% repigmentation, 5 patients showed 26% to 50% repigmentation, and 5 patients showed 51% to 75% repigmentation of the affected areas.4
Eleven of 31 (35%) web pages recommended inclusion of vitamin B12 supplementation in vitiligo. A 2-year study with 100 participants showed that supplementation with folic acid and vitamin B12 along with sun exposure yielded more effective repigmentation than either vitamins or sun exposure alone.5 An additional hypothesis suggested vitamin B12 may aid in repigmentation through its role in the homocysteine pathway. Although the theory is unproven, it is proposed that inhibition of homocysteine via vitamin B12 or folic acid supplementation may play a role in reducing melanocyte destruction and restoring melanin synthesis.6
There were mixed recommendations regarding vitamin C via supplementation and/or eating citrus fruits such as oranges. Although there are limited clinical studies on the use of vitamin C and the treatment of vitiligo, a 6-year prospective study from Madagascar consisting of approximately 300 participants with vitiligo who were treated with a combination of topical corticosteroids, oral vitamin C, and oral vitamin B12 supplementation showed excellent repigmentation (defined by repigmentation of more than 76% of the originally affected area) in 50 participants.7
Fruits—Most web pages had mixed recommendations on whether to include or avoid certain fruits. Interestingly, inclusion of mangoes and apricots in the diet were highly recommended (9/31 [29%] and 8/31 [26%], respectively) while fruits such as oranges, lemons, papayas, and grapes were discouraged (10/31 [32%], 8/31 [26%], 6/31 [19%], and 7/31 [23%], respectively). Although some web pages suggested that vitamin C–rich produce including citrus and berries may help to increase melanin formation, others strongly suggested avoiding these fruits. There is limited information on the effects of citrus on vitiligo, but a 2022 study indicated that 5-demethylnobiletin, a flavonoid found in sweet citrus fruits, may stimulate melanin synthesis, which can possibly be beneficial for vitiligo.8
Omega-3 Fatty Acids—Seven of 31 (23%) web pages recommended the inclusion of omega-3 fatty acids for their role as antioxidants to improve vitiligo symptoms. Research has indicated a strong association between vitiligo and oxidative stress.9 A 2007 controlled clinical trial that included 28 vitiligo patients demonstrated that oral antioxidant supplementation in combination with narrowband UVB phototherapy can significantly decrease vitiligo-associated oxidative stress (P<.05); 8 of 17 (47%) patients in the treatment group saw greater than 75% repigmentation after antioxidant treatment.10
Grains—Five of 31 (16%) web pages suggested avoiding gluten—a protein naturally found in some grains including wheat, barley, and rye—to improve vitiligo symptoms. A 2021 review suggested that a gluten-free diet may be effective in managing celiac disease, and it is hypothesized that vitiligo may be managed with similar dietary adjustments.11 Studies have shown that celiac disease and vitiligo—both autoimmune conditions—involve IL-2, IL-6, IL-7, and IL-21 in their disease pathways.12,13 Their shared immunogenic mechanism may account for similar management options.
Upon review, 2 case reports were identified that discussed a relationship between a gluten-free diet and vitiligo symptom improvement. In one report, a 9-year-old child diagnosed with both celiac disease and vitiligo saw intense repigmentation of the skin after adhering to a gluten-free diet for 1 year.14 Another case study reported a 22-year-old woman with vitiligo whose symptoms improved after 1 month of a gluten-free diet following 2 years of failed treatment with a topical steroid and phototherapy.15
Seven of 31 (23%) web pages suggested that individuals with vitiligo should include wheat in their diet. There is no published literature discussing the relationship between vitiligo and wheat. Of the 31 web pages reviewed, 10 (32%) suggested including whole grain. There is no relevant scientific evidence or hypotheses describing how whole grains may be beneficial in vitiligo.
Minerals—Eight of 31 (26%) web pages suggested including zinc in the diet to improve vitiligo symptoms. A 2020 study evaluated how different serum levels of zinc in vitiligo patients might be affiliated with interleukin activity. Fifty patients diagnosed with active vitiligo were tested for serum levels of zinc, IL-4, IL-6, and IL-17.16 The results showed that mean serum levels of zinc were lower in vitiligo patients compared with patients without vitiligo. The study concluded that zinc could possibly be used as a supplement to improve vitiligo, though the dosage needs to be further studied and confirmed.16
Vegetables—Eleven of 31 (35%) web pages recommended leafy green vegetables and 13 of 31 (42%) recommended spinach for patients with vitiligo. Spinach and other leafy green vegetables are known to be rich in antioxidants, which may have protective effects against reactive oxygen species that are thought to contribute to vitiligo progression.17,18
Nuts—Walnuts were recommended in 11 of 31 (35%) web pages. Nuts may be beneficial in reducing inflammation and providing protection against oxidative stress.9 However, there is no specific scientific literature that supports the inclusion of nuts in the diet to manage vitiligo symptoms.
Comment
With a growing amount of research suggesting that diet modifications may contribute to management of certain skin conditions, vitiligo patients often inquire about foods or supplements that may help improve their condition.19 Our review highlighted what information was available to the public regarding diet and vitiligo, with preliminary support of the following primary diet components: vitamin D, vitamin B12, zinc, and omega-3 fatty acids. Our review showed no support in the literature for the items that were recommended to avoid. It is important to note that 27 of 31 (87%) web pages from our online search were not authored by medical professionals or dermatologists. Additionally, many web pages suggested conflicting information, making it difficult to draw concrete conclusions about what diet modifications will be beneficial to the vitiligo community. Further controlled clinical trials are warranted due to the lack of formal studies that assess the relationship between diet and vitiligo.
Internet platforms have become a common source of medical information for individuals with a broad range of skin conditions including vitiligo. The prevalence of vitiligo among US adults ranges from 0.76% to 1.11%, with approximately 40% of adult cases of vitiligo in the United States remaining undiagnosed.1 The vitiligo community has become more inquisitive of the relationship between diet and vitiligo, turning to online sources for suggestions on diet modifications that may be beneficial for their condition. Although there is an abundance of online information, few diets or foods have been medically recognized to definitively improve or worsen vitiligo symptoms. We reviewed the top online web pages accessible to the public regarding diet suggestions that affect vitiligo symptoms. We then compared these online results to published peer-reviewed scientific literature.
Methods
Two independent online searches were performed by Researcher 1 (Y.A.) and Researcher 2 (I.M.) using Google Advanced Search. The independent searches were performed by the reviewers in neighboring areas of Chicago, Illinois, using the same Internet browser (Google Chrome). The primary search terms were diet and vitiligo along with the optional additional terms dietary supplement(s), food(s), nutrition, herb(s), or vitamin(s). Our search included any web pages published or updated from January 1, 2010, to December 31, 2021, and originally scribed in the English language. The domains “.com,” “.org,” “.edu,” and “.cc” were included.

From this initial search, Researcher 1 identified 312 web pages and Researcher 2 identified 314 web pages. Each reviewer sorted their respective search results to identify the number of eligible records to be screened. Records were defined as unique web pages that met the search criteria. After removing duplicates, Researcher 1 screened 102 web pages and Researcher 2 screened 76 web pages. Of these records, web pages were excluded if they did not include any diet recommendations for vitiligo patients. Each reviewer independently created a list of eligible records, and the independent lists were then merged for a total of 58 web pages. Among these 58 web pages, there were 24 duplicate records and 3 records that were deemed ineligible for the study due to lack of subject matter relevance. A final total of 31 web pages were included in the data analysis (Figure). Of the 31 records selected, the reviewers jointly evaluated each web page and recorded the diet components that were recommended for individuals with vitiligo to either include or avoid (eTable).


For comparison and support from published scientific literature, a search of PubMed articles indexed for MEDLINE was conducted using the terms diet and vitiligo. Relevant human clinical studies published in the English-language literature were reviewed for content regarding the relationship between diet and vitiligo.
Results
Our online search revealed an abundance of information regarding various dietary modifications suggested to aid in the management of vitiligo symptoms. Most web pages (27/31 [87%]) were not authored by medical professionals or dermatologists. There were 27 diet components mentioned 8 or more times within the 31 total web pages. These diet components were selected for further review via PubMed. Each item was searched on PubMed using the term “[respective diet component] and vitiligo” among all published literature in the English language. Our study focused on summarizing the data on dietary components for which we were able to gather scientific support. These data have been organized into the following categories: vitamins, fruits, omega-3 fatty acids, grains, minerals, vegetables, and nuts.
Vitamins—The online literature recommended inclusion of vitamin supplements, in particular vitamins D and B12, which aligned with published scientific literature.2,3 Eleven of 31 (35%) web pages recommended vitamin D in vitiligo. A 2010 study analyzing patients with vitiligo vulgaris (N=45) found that 68.9% of the cohort had insufficient (<30 ng/mL) 25-hydroxyvitamin D levels.2 A prospective study of 30 individuals found that the use of tacrolimus ointment plus oral vitamin D supplementation was found to be more successful in repigmentation than topical tacrolimus alone.3 Vitamin D dosage ranged from 1500 IU/d if the patient’s serum 25-hydroxyvitamin D levels were less than 20 ng/mL to 3000 IU/d if the serum levels were less than 10 ng/mL for 6 months.
Dairy products are a source of vitamin D.2,3 Of the web pages that mentioned dairy, a subtle majority (4/7 [57%]) recommended the inclusion of dairy products. Although many web pages did not specify whether oral vitamin D supplementation vs dietary food consumption is preferred, a 2013 controlled study of 16 vitiligo patients who received high doses of vitamin D supplementation with a low-calcium diet found that 4 patients showed 1% to 25% repigmentation, 5 patients showed 26% to 50% repigmentation, and 5 patients showed 51% to 75% repigmentation of the affected areas.4
Eleven of 31 (35%) web pages recommended inclusion of vitamin B12 supplementation in vitiligo. A 2-year study with 100 participants showed that supplementation with folic acid and vitamin B12 along with sun exposure yielded more effective repigmentation than either vitamins or sun exposure alone.5 An additional hypothesis suggested vitamin B12 may aid in repigmentation through its role in the homocysteine pathway. Although the theory is unproven, it is proposed that inhibition of homocysteine via vitamin B12 or folic acid supplementation may play a role in reducing melanocyte destruction and restoring melanin synthesis.6
There were mixed recommendations regarding vitamin C via supplementation and/or eating citrus fruits such as oranges. Although there are limited clinical studies on the use of vitamin C and the treatment of vitiligo, a 6-year prospective study from Madagascar consisting of approximately 300 participants with vitiligo who were treated with a combination of topical corticosteroids, oral vitamin C, and oral vitamin B12 supplementation showed excellent repigmentation (defined by repigmentation of more than 76% of the originally affected area) in 50 participants.7
Fruits—Most web pages had mixed recommendations on whether to include or avoid certain fruits. Interestingly, inclusion of mangoes and apricots in the diet were highly recommended (9/31 [29%] and 8/31 [26%], respectively) while fruits such as oranges, lemons, papayas, and grapes were discouraged (10/31 [32%], 8/31 [26%], 6/31 [19%], and 7/31 [23%], respectively). Although some web pages suggested that vitamin C–rich produce including citrus and berries may help to increase melanin formation, others strongly suggested avoiding these fruits. There is limited information on the effects of citrus on vitiligo, but a 2022 study indicated that 5-demethylnobiletin, a flavonoid found in sweet citrus fruits, may stimulate melanin synthesis, which can possibly be beneficial for vitiligo.8
Omega-3 Fatty Acids—Seven of 31 (23%) web pages recommended the inclusion of omega-3 fatty acids for their role as antioxidants to improve vitiligo symptoms. Research has indicated a strong association between vitiligo and oxidative stress.9 A 2007 controlled clinical trial that included 28 vitiligo patients demonstrated that oral antioxidant supplementation in combination with narrowband UVB phototherapy can significantly decrease vitiligo-associated oxidative stress (P<.05); 8 of 17 (47%) patients in the treatment group saw greater than 75% repigmentation after antioxidant treatment.10
Grains—Five of 31 (16%) web pages suggested avoiding gluten—a protein naturally found in some grains including wheat, barley, and rye—to improve vitiligo symptoms. A 2021 review suggested that a gluten-free diet may be effective in managing celiac disease, and it is hypothesized that vitiligo may be managed with similar dietary adjustments.11 Studies have shown that celiac disease and vitiligo—both autoimmune conditions—involve IL-2, IL-6, IL-7, and IL-21 in their disease pathways.12,13 Their shared immunogenic mechanism may account for similar management options.
Upon review, 2 case reports were identified that discussed a relationship between a gluten-free diet and vitiligo symptom improvement. In one report, a 9-year-old child diagnosed with both celiac disease and vitiligo saw intense repigmentation of the skin after adhering to a gluten-free diet for 1 year.14 Another case study reported a 22-year-old woman with vitiligo whose symptoms improved after 1 month of a gluten-free diet following 2 years of failed treatment with a topical steroid and phototherapy.15
Seven of 31 (23%) web pages suggested that individuals with vitiligo should include wheat in their diet. There is no published literature discussing the relationship between vitiligo and wheat. Of the 31 web pages reviewed, 10 (32%) suggested including whole grain. There is no relevant scientific evidence or hypotheses describing how whole grains may be beneficial in vitiligo.
Minerals—Eight of 31 (26%) web pages suggested including zinc in the diet to improve vitiligo symptoms. A 2020 study evaluated how different serum levels of zinc in vitiligo patients might be affiliated with interleukin activity. Fifty patients diagnosed with active vitiligo were tested for serum levels of zinc, IL-4, IL-6, and IL-17.16 The results showed that mean serum levels of zinc were lower in vitiligo patients compared with patients without vitiligo. The study concluded that zinc could possibly be used as a supplement to improve vitiligo, though the dosage needs to be further studied and confirmed.16
Vegetables—Eleven of 31 (35%) web pages recommended leafy green vegetables and 13 of 31 (42%) recommended spinach for patients with vitiligo. Spinach and other leafy green vegetables are known to be rich in antioxidants, which may have protective effects against reactive oxygen species that are thought to contribute to vitiligo progression.17,18
Nuts—Walnuts were recommended in 11 of 31 (35%) web pages. Nuts may be beneficial in reducing inflammation and providing protection against oxidative stress.9 However, there is no specific scientific literature that supports the inclusion of nuts in the diet to manage vitiligo symptoms.
Comment
With a growing amount of research suggesting that diet modifications may contribute to management of certain skin conditions, vitiligo patients often inquire about foods or supplements that may help improve their condition.19 Our review highlighted what information was available to the public regarding diet and vitiligo, with preliminary support of the following primary diet components: vitamin D, vitamin B12, zinc, and omega-3 fatty acids. Our review showed no support in the literature for the items that were recommended to avoid. It is important to note that 27 of 31 (87%) web pages from our online search were not authored by medical professionals or dermatologists. Additionally, many web pages suggested conflicting information, making it difficult to draw concrete conclusions about what diet modifications will be beneficial to the vitiligo community. Further controlled clinical trials are warranted due to the lack of formal studies that assess the relationship between diet and vitiligo.
- Gandhi K, Ezzedine K, Anastassopoulos KP, et al. Prevalence of vitiligo among adults in the United States. JAMA Dermatol. 2022;158:43-50. doi:10.1001/jamadermatol.2021.4724
- Silverberg JI, Silverberg AI, Malka E, et al. A pilot study assessing the role of 25 hydroxy vitamin D levels in patients with vitiligo vulgaris. J Am Acad Dermatol. 2010;62:937-941. doi:10.1016/j.jaad.2009.11.024
- Karagüzel G, Sakarya NP, Bahadır S, et al. Vitamin D status and the effects of oral vitamin D treatment in children with vitiligo: a prospective study. Clin Nutr ESPEN. 2016;15:28-31. doi:10.1016/j.clnesp.2016.05.006.
- Finamor DC, Sinigaglia-Coimbra R, Neves LC, et al. A pilot study assessing the effect of prolonged administration of high daily doses of vitamin D on the clinical course of vitiligo and psoriasis. Dermatoendocrinol. 2013;5:222-234. doi:10.4161/derm.24808
- Juhlin L, Olsson MJ. Improvement of vitiligo after oral treatment with vitamin B12 and folic acid and the importance of sun exposure. Acta Derm Venereol. 1997;77:460-462. doi:10.2340/000155555577460462
- Chen J, Zhuang T, Chen J, et al. Homocysteine induces melanocytes apoptosis via PERK-eIF2α-CHOP pathway in vitiligo. Clin Sci (Lond). 2020;134:1127-1141. doi:10.1042/CS20200218
- Sendrasoa FA, Ranaivo IM, Sata M, et al. Treatment responses in patients with vitiligo to very potent topical corticosteroids combined with vitamin therapy in Madagascar. Int J Dermatol. 2019;58:908-911. doi:10.1111/ijd.14510
- Wang HM, Qu LQ, Ng JPL, et al. Natural citrus flavanone 5-demethylnobiletin stimulates melanogenesis through the activation of cAMP/CREB pathway in B16F10 cells. Phytomedicine. 2022;98:153941. doi:10.1016/j.phymed.2022.153941
- Ros E. Health benefits of nut consumption. Nutrients. 2010;2:652-682.
- Dell’Anna ML, Mastrofrancesco A, Sala R, et al. Antioxidants and narrow band-UVB in the treatment of vitiligo: a double-blind placebo controlled trial. Clin Exp Dermatol. 2007;32:631-636.
- Gastrointestinal microbiome and gluten in celiac disease. Ann Med. 2021;53:1797-1805. doi:10.1080/07853890.2021.1990392
- Forabosco P, Neuhausen SL, Greco L, et al. Meta-analysis of genome-wide linkage studies in celiac disease. Hum Hered. 2009;68:223-230. doi:10.1159/000228920
- Akbulut UE, Çebi AH, Sag˘ E, et al. Interleukin-6 and interleukin-17 gene polymorphism association with celiac disease in children. Turk J Gastroenterol. 2017;28:471-475. doi:10.5152/tjg.2017.17092
- Rodríguez-García C, González-Hernández S, Pérez-Robayna N, et al. Repigmentation of vitiligo lesions in a child with celiac disease after a gluten-free diet. Pediatr Dermatol. 2011;28:209-210. doi:10.1111/j.1525-1470.2011.01388.x
- Khandalavala BN, Nirmalraj MC. Rapid partial repigmentation ofvitiligo in a young female adult with a gluten-free diet. Case Rep Dermatol. 2014;6:283-287.
- Sanad EM, El-Fallah AA, Al-Doori AR, et al. Serum zinc and inflammatory cytokines in vitiligo. J Clin Aesthet Dermatol. 2020;13:(12 suppl 1):S29-S33.
- Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci U S A. 1993;90:7915-7922. doi:10.1073/pnas.90.17.7915
- Xian D, Guo M, Xu J, et al. Current evidence to support the therapeutic potential of flavonoids in oxidative stress-related dermatoses. Redox Rep. 2021;26:134-146. doi:10.1080 /13510002.2021.1962094
- Katta R, Kramer MJ. Skin and diet: an update on the role of dietary change as a treatment strategy for skin disease. Skin Therapy Lett. 2018;23:1-5.
- Gandhi K, Ezzedine K, Anastassopoulos KP, et al. Prevalence of vitiligo among adults in the United States. JAMA Dermatol. 2022;158:43-50. doi:10.1001/jamadermatol.2021.4724
- Silverberg JI, Silverberg AI, Malka E, et al. A pilot study assessing the role of 25 hydroxy vitamin D levels in patients with vitiligo vulgaris. J Am Acad Dermatol. 2010;62:937-941. doi:10.1016/j.jaad.2009.11.024
- Karagüzel G, Sakarya NP, Bahadır S, et al. Vitamin D status and the effects of oral vitamin D treatment in children with vitiligo: a prospective study. Clin Nutr ESPEN. 2016;15:28-31. doi:10.1016/j.clnesp.2016.05.006.
- Finamor DC, Sinigaglia-Coimbra R, Neves LC, et al. A pilot study assessing the effect of prolonged administration of high daily doses of vitamin D on the clinical course of vitiligo and psoriasis. Dermatoendocrinol. 2013;5:222-234. doi:10.4161/derm.24808
- Juhlin L, Olsson MJ. Improvement of vitiligo after oral treatment with vitamin B12 and folic acid and the importance of sun exposure. Acta Derm Venereol. 1997;77:460-462. doi:10.2340/000155555577460462
- Chen J, Zhuang T, Chen J, et al. Homocysteine induces melanocytes apoptosis via PERK-eIF2α-CHOP pathway in vitiligo. Clin Sci (Lond). 2020;134:1127-1141. doi:10.1042/CS20200218
- Sendrasoa FA, Ranaivo IM, Sata M, et al. Treatment responses in patients with vitiligo to very potent topical corticosteroids combined with vitamin therapy in Madagascar. Int J Dermatol. 2019;58:908-911. doi:10.1111/ijd.14510
- Wang HM, Qu LQ, Ng JPL, et al. Natural citrus flavanone 5-demethylnobiletin stimulates melanogenesis through the activation of cAMP/CREB pathway in B16F10 cells. Phytomedicine. 2022;98:153941. doi:10.1016/j.phymed.2022.153941
- Ros E. Health benefits of nut consumption. Nutrients. 2010;2:652-682.
- Dell’Anna ML, Mastrofrancesco A, Sala R, et al. Antioxidants and narrow band-UVB in the treatment of vitiligo: a double-blind placebo controlled trial. Clin Exp Dermatol. 2007;32:631-636.
- Gastrointestinal microbiome and gluten in celiac disease. Ann Med. 2021;53:1797-1805. doi:10.1080/07853890.2021.1990392
- Forabosco P, Neuhausen SL, Greco L, et al. Meta-analysis of genome-wide linkage studies in celiac disease. Hum Hered. 2009;68:223-230. doi:10.1159/000228920
- Akbulut UE, Çebi AH, Sag˘ E, et al. Interleukin-6 and interleukin-17 gene polymorphism association with celiac disease in children. Turk J Gastroenterol. 2017;28:471-475. doi:10.5152/tjg.2017.17092
- Rodríguez-García C, González-Hernández S, Pérez-Robayna N, et al. Repigmentation of vitiligo lesions in a child with celiac disease after a gluten-free diet. Pediatr Dermatol. 2011;28:209-210. doi:10.1111/j.1525-1470.2011.01388.x
- Khandalavala BN, Nirmalraj MC. Rapid partial repigmentation ofvitiligo in a young female adult with a gluten-free diet. Case Rep Dermatol. 2014;6:283-287.
- Sanad EM, El-Fallah AA, Al-Doori AR, et al. Serum zinc and inflammatory cytokines in vitiligo. J Clin Aesthet Dermatol. 2020;13:(12 suppl 1):S29-S33.
- Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci U S A. 1993;90:7915-7922. doi:10.1073/pnas.90.17.7915
- Xian D, Guo M, Xu J, et al. Current evidence to support the therapeutic potential of flavonoids in oxidative stress-related dermatoses. Redox Rep. 2021;26:134-146. doi:10.1080 /13510002.2021.1962094
- Katta R, Kramer MJ. Skin and diet: an update on the role of dietary change as a treatment strategy for skin disease. Skin Therapy Lett. 2018;23:1-5.
Practice Points
- There are numerous online dietary and supplement recommendations that claim to impact vitiligo but most are not authored by medical professionals or dermatologists.
- Scientific evidence supporting specific dietary and supplement recommendations for vitiligo is limited.
- Current preliminary data support the potential recommendation for dietary supplementation with vitamin D, vitamin B12, zinc, and omega-3 fatty acids.
Study: Early Tecovirimat Stops Mpox Progression in HIV Patients
A new analysis supports using the smallpox antiviral tecovirimat (TPOXX/ST-246) in HIV patients showing the first symptoms of the human smallpox disease mpox (monkeypox), caused by the variola virus.
In a small prospective matched cohort analysis, people with HIV (PWH) and mpox disease who received tecovirimat within 7 days of symptom onset were 13 times less likely to experience progression, compared with PWH not prescribed tecovirimat within that window. In a matched cohort of 112 PWH, mpox disease progression occurred in 5.4% in an early tecovirimat group and in 26.8% in a late- or no-tecovirimat group, for a paired odds ratio of 13.00 (95% CI, 1.71-99.40; P = .002).
“Results of the present study suggest that tecovirimat treatment should be started early at the time of suspected mpox diagnosis in all PWH, especially in those with nonsuppressed HIV viremia or mucosal site involvement,” wrote a team led by Bruce Aldred, MD, of the Division of Infectious Diseases in the Department of Medicine at Emory University School of Medicine in Atlanta, in JAMA Internal Medicine. Early symptoms of mpox include skin rash and mucosal lesions, along with viral symptoms such as fever, headache, muscle aches, back pain, low energy, and swollen lymph nodes.
As of March 1 of last year, the United States reported more than 30,000 cases, while cases numbered more than 86,000 worldwide.
Despite a lack of effectiveness data in humans, tecovirimat was widely prescribed to PWH with mpox during the 2022 epidemic, which disproportionately affected PWH, particularly those with low CD4+ T-cell counts or severe mpox clinical manifestations who needed urgent therapy. Developed to treat smallpox, tecovirimat has antiviral activity against other orthopoxviruses, and has reduced mpox-related morbidity and mortality in animals.
Based on the animal data, approval was granted by the US Food and Drug Administration (FDA) for human mpox treatment. Dr. Aldred and colleagues undertook this cohort analysis in the absence of human data and with the postoutbreak decline in cases impeding recruitment to a full-scale clinical trial.
Study design
The preponderantly Black cohort included 112 PWH diagnosed with mpox at four Atlanta hospitals from June 1 to October 7, 2022. Patients were grouped in an early cohort receiving tecovirimat within 7 days of symptom onset or a no or late cohort (no tecovirimat or treatment more than 7 days after symptom onset. Multivariate logistic regression models identified factors associated with progression, defined as development of at least one severe CDC mpox criterion after symptom day 7.
The cohorts were then matched 1:1 using propensity scores based on the identified factors, and mpox disease progression was compared.
Of 112 PWH, 56 receive early tecovirimat and 56 received no or late treatment. In the early group, the median (interquartile range [IQR]) age was 35 (30-42) years; 54 individuals (96.4%) were cisgender men, 46 (82.1%) were Black, and 10 (17.9%) were, variously, White, American Indian, Alaska Native, Asian, Native Hawaiian or Other Pacific Islander, or of unknown race.
In the late- or no-tecovirimat group, the median (IQR) age was 36 (32-43) years; 54 (96.4%) were cisgender men, 49 (87.5%) were Black, and 7 (12.5%) were individuals of other or unknown race. Mpox disease progression occurred in 3 PWH in the early-tecovirimat group and 15 PWH (26.8%) in the late- or no-tecovirimat group.
Dr. Aldred and colleagues acknowledged that more research is needed to confirm the findings and cited several study limitations. These included the small sample size, the preponderance of Black participants, and the possibility that unmatched confounding variables could have led to the observation of fewer cases of severe disease in the early-tecovirimat cohort.
This study was supported by a grant from the Emory Center for AIDS Research. Coauthors reported grants from various institutes at the National Institutes of Health as well as from multiple pharmaceutical companies.
A new analysis supports using the smallpox antiviral tecovirimat (TPOXX/ST-246) in HIV patients showing the first symptoms of the human smallpox disease mpox (monkeypox), caused by the variola virus.
In a small prospective matched cohort analysis, people with HIV (PWH) and mpox disease who received tecovirimat within 7 days of symptom onset were 13 times less likely to experience progression, compared with PWH not prescribed tecovirimat within that window. In a matched cohort of 112 PWH, mpox disease progression occurred in 5.4% in an early tecovirimat group and in 26.8% in a late- or no-tecovirimat group, for a paired odds ratio of 13.00 (95% CI, 1.71-99.40; P = .002).
“Results of the present study suggest that tecovirimat treatment should be started early at the time of suspected mpox diagnosis in all PWH, especially in those with nonsuppressed HIV viremia or mucosal site involvement,” wrote a team led by Bruce Aldred, MD, of the Division of Infectious Diseases in the Department of Medicine at Emory University School of Medicine in Atlanta, in JAMA Internal Medicine. Early symptoms of mpox include skin rash and mucosal lesions, along with viral symptoms such as fever, headache, muscle aches, back pain, low energy, and swollen lymph nodes.
As of March 1 of last year, the United States reported more than 30,000 cases, while cases numbered more than 86,000 worldwide.
Despite a lack of effectiveness data in humans, tecovirimat was widely prescribed to PWH with mpox during the 2022 epidemic, which disproportionately affected PWH, particularly those with low CD4+ T-cell counts or severe mpox clinical manifestations who needed urgent therapy. Developed to treat smallpox, tecovirimat has antiviral activity against other orthopoxviruses, and has reduced mpox-related morbidity and mortality in animals.
Based on the animal data, approval was granted by the US Food and Drug Administration (FDA) for human mpox treatment. Dr. Aldred and colleagues undertook this cohort analysis in the absence of human data and with the postoutbreak decline in cases impeding recruitment to a full-scale clinical trial.
Study design
The preponderantly Black cohort included 112 PWH diagnosed with mpox at four Atlanta hospitals from June 1 to October 7, 2022. Patients were grouped in an early cohort receiving tecovirimat within 7 days of symptom onset or a no or late cohort (no tecovirimat or treatment more than 7 days after symptom onset. Multivariate logistic regression models identified factors associated with progression, defined as development of at least one severe CDC mpox criterion after symptom day 7.
The cohorts were then matched 1:1 using propensity scores based on the identified factors, and mpox disease progression was compared.
Of 112 PWH, 56 receive early tecovirimat and 56 received no or late treatment. In the early group, the median (interquartile range [IQR]) age was 35 (30-42) years; 54 individuals (96.4%) were cisgender men, 46 (82.1%) were Black, and 10 (17.9%) were, variously, White, American Indian, Alaska Native, Asian, Native Hawaiian or Other Pacific Islander, or of unknown race.
In the late- or no-tecovirimat group, the median (IQR) age was 36 (32-43) years; 54 (96.4%) were cisgender men, 49 (87.5%) were Black, and 7 (12.5%) were individuals of other or unknown race. Mpox disease progression occurred in 3 PWH in the early-tecovirimat group and 15 PWH (26.8%) in the late- or no-tecovirimat group.
Dr. Aldred and colleagues acknowledged that more research is needed to confirm the findings and cited several study limitations. These included the small sample size, the preponderance of Black participants, and the possibility that unmatched confounding variables could have led to the observation of fewer cases of severe disease in the early-tecovirimat cohort.
This study was supported by a grant from the Emory Center for AIDS Research. Coauthors reported grants from various institutes at the National Institutes of Health as well as from multiple pharmaceutical companies.
A new analysis supports using the smallpox antiviral tecovirimat (TPOXX/ST-246) in HIV patients showing the first symptoms of the human smallpox disease mpox (monkeypox), caused by the variola virus.
In a small prospective matched cohort analysis, people with HIV (PWH) and mpox disease who received tecovirimat within 7 days of symptom onset were 13 times less likely to experience progression, compared with PWH not prescribed tecovirimat within that window. In a matched cohort of 112 PWH, mpox disease progression occurred in 5.4% in an early tecovirimat group and in 26.8% in a late- or no-tecovirimat group, for a paired odds ratio of 13.00 (95% CI, 1.71-99.40; P = .002).
“Results of the present study suggest that tecovirimat treatment should be started early at the time of suspected mpox diagnosis in all PWH, especially in those with nonsuppressed HIV viremia or mucosal site involvement,” wrote a team led by Bruce Aldred, MD, of the Division of Infectious Diseases in the Department of Medicine at Emory University School of Medicine in Atlanta, in JAMA Internal Medicine. Early symptoms of mpox include skin rash and mucosal lesions, along with viral symptoms such as fever, headache, muscle aches, back pain, low energy, and swollen lymph nodes.
As of March 1 of last year, the United States reported more than 30,000 cases, while cases numbered more than 86,000 worldwide.
Despite a lack of effectiveness data in humans, tecovirimat was widely prescribed to PWH with mpox during the 2022 epidemic, which disproportionately affected PWH, particularly those with low CD4+ T-cell counts or severe mpox clinical manifestations who needed urgent therapy. Developed to treat smallpox, tecovirimat has antiviral activity against other orthopoxviruses, and has reduced mpox-related morbidity and mortality in animals.
Based on the animal data, approval was granted by the US Food and Drug Administration (FDA) for human mpox treatment. Dr. Aldred and colleagues undertook this cohort analysis in the absence of human data and with the postoutbreak decline in cases impeding recruitment to a full-scale clinical trial.
Study design
The preponderantly Black cohort included 112 PWH diagnosed with mpox at four Atlanta hospitals from June 1 to October 7, 2022. Patients were grouped in an early cohort receiving tecovirimat within 7 days of symptom onset or a no or late cohort (no tecovirimat or treatment more than 7 days after symptom onset. Multivariate logistic regression models identified factors associated with progression, defined as development of at least one severe CDC mpox criterion after symptom day 7.
The cohorts were then matched 1:1 using propensity scores based on the identified factors, and mpox disease progression was compared.
Of 112 PWH, 56 receive early tecovirimat and 56 received no or late treatment. In the early group, the median (interquartile range [IQR]) age was 35 (30-42) years; 54 individuals (96.4%) were cisgender men, 46 (82.1%) were Black, and 10 (17.9%) were, variously, White, American Indian, Alaska Native, Asian, Native Hawaiian or Other Pacific Islander, or of unknown race.
In the late- or no-tecovirimat group, the median (IQR) age was 36 (32-43) years; 54 (96.4%) were cisgender men, 49 (87.5%) were Black, and 7 (12.5%) were individuals of other or unknown race. Mpox disease progression occurred in 3 PWH in the early-tecovirimat group and 15 PWH (26.8%) in the late- or no-tecovirimat group.
Dr. Aldred and colleagues acknowledged that more research is needed to confirm the findings and cited several study limitations. These included the small sample size, the preponderance of Black participants, and the possibility that unmatched confounding variables could have led to the observation of fewer cases of severe disease in the early-tecovirimat cohort.
This study was supported by a grant from the Emory Center for AIDS Research. Coauthors reported grants from various institutes at the National Institutes of Health as well as from multiple pharmaceutical companies.
FROM JAMA INTERNAL MEDICINE
Culprits of Medication-Induced Telogen Effluvium, Part 2
Medication-induced telogen effluvium (TE) is a nonscarring alopecia that typically is reversible. Appropriate management requires identification of the underlying trigger and cessation of potential culprit medications. In part 2 of this series, we review anticoagulant and antihypertensive medications as potential contributors to TE.
Anticoagulants
Anticoagulants target various parts of the coagulation cascade to prevent clot formation in patients with conditions that increase their risk for thromboembolic events. Common indications for initiating anticoagulant therapy include atrial fibrillation,1 venous thromboembolism,2 acute myocardial infarction,3 malignancy,4 and hypercoagulable states.5 Traditional anticoagulants include heparin and warfarin. Heparin is a glycosaminoglycan that exerts its anticoagulant effects through binding with antithrombin, greatly increasing its inactivation of thrombin and factor Xa of the coagulation cascade.6 Warfarin is a coumarin derivative that inhibits activation of vitamin K, subsequently limiting the function of vitamin K–dependent factors II, VII, IX, and X.7,8 Watras et al9 noted that heparin and warfarin were implicated in alopecia as their clinical use became widespread throughout the mid-20th century. Onset of alopecia following the use of heparin or warfarin was reported at 3 weeks to 3 months following medication initiation, with most cases clinically consistent with TE.9 Heparin and warfarin both have alopecia reported as a potential adverse effect in their structured product labeling documents.10,11
Heparin is further classified into unfractionated heparin (UFH) and low-molecular-weight heparin (LMWH); the latter is a heterogeneous group of medications derived from chemical or enzymatic depolymerization of UFH.12 In contrast to UFH, LMWH exerts its anticoagulant effects through inactivation of factor Xa without the ability to bind thrombin.12 An animal study using anagen-induced mice demonstrated that intraperitoneal administration of heparin inhibited the development of anagen follicles, while in vitro studies showed that the addition of heparin inhibited mouse dermal papilla cell proliferation.13 Other animal and in vitro studies have examined the inhibitory effects of heparin on signaling pathways in tumor lymphangiogenesis, including the vascular endothelial growth factor C/vascular endothelial growth factor receptor 3 axis.14,15 Clinically, it has been demonstrated that heparin, especially LMWHs, may be associated with a survival benefit among certain cancer patients,16,17 with the impact of LMWHs attributed to antimitotic and antimetastatic effects of heparin on tumor growth.14 It is hypothesized that such antiangiogenic and antimitotic effects also are involved in the pathomechanisms of heparin-induced alopecia.18
More recently, the use of direct oral anticoagulants (DOACs) such as dabigatran, rivaroxaban, and apixaban has increased due to their more favorable adverse-effect profile and minimal monitoring requirements. Bonaldo et al19 conducted an analysis of reports submitted to the World Health Organization’s VigiBase database of alopecia associated with DOACs until May 2, 2018. They found 1316 nonduplicate DOAC-induced cases of alopecia, with rivaroxaban as the most reported drug associated with alopecia development (58.8% [774/1316]). Only 4 cases demonstrated alopecia with DOAC rechallenge, suggesting onset of alopecia may have been unrelated to DOAC use or caused by a different trigger. Among 243 cases with a documented time to onset of alopecia, the median was 28 days, with an interquartile range of 63 days. Because TE most commonly occurs 3 to 4 months after the inciting event or medication trigger, there is little evidence to suggest DOACs as the cause of TE, and the observed cases of alopecia may be attributable to another preceding medical event and/or medication exposure.19 More studies are needed to examine the impact of anticoagulant medications on the hair cycle.
Antihypertensives
Hypertension is a modifiable risk factor for several cardiovascular diseases.20 According to the 2019 American College of Cardiology/American Heart Association Guideline on the Primary Prevention of Cardiovascular Disease,21 first-line medications include thiazide diuretics, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, and angiotensin receptor blockers (ARBs).
Angiotensin-converting enzyme inhibitors exert their antihypertensive effects by reducing conversion of angiotensin I to angiotensin II, thereby limiting the downstream effects of vasoconstriction as well as sodium and water retention. Given the proven mortality benefit of ACE inhibition in patients with congestive heart failure, ACE inhibitors are used as first-line therapy in these patients.22,23 Alopecia associated with ACE inhibitors is rare and limited to case reports following their introduction and approval in 1981.24-28 In one case, a woman in her 60s with congestive heart failure initiated captopril with development of an erythematous pruritic rash on the extremities and diffuse scalp hair loss 2 months later; spontaneous hair growth resumed 1 month following captopril discontinuation.25 In this case, the hair loss may be secondary to the drug eruption rather than true medication-induced TE. Initiation of enalapril in a woman in her 30s with hypertension was associated with diffuse scalp alopecia 4 weeks later that resolved with cessation of the suspected culprit, enalapril; rechallenge with enalapril several months later reproduced the hair loss.27 Given limited reports of ACE inhibitor–associated hair loss relative to their pervasive use, a direct causal role between ACE inhibition and TE is unlikely, or it has not been rigorously identified. The structured product labeling for captopril includes alopecia in its list of adverse effects reported in approximately 0.5% to 2% of patients but did not appear at increased frequency compared to placebo or other treatments used in controlled trials.29 Alternative inciting causes of alopecia in patients prescribed ACE inhibitors may include use of other medications, hospitalization, or metabolic derangements related to their underlying cardiac disease.
Although not indicated as a primary treatment for hypertension, β-blockers have US Food and Drug Administration approval for the treatment of certain arrhythmias, hypertension, heart failure, myocardial infarction, hyperthyroidism, and other conditions.30 β-Blockers are competitive antagonists of β-adrenergic receptors that limit the production of intracellular cyclic adenosine monophosphate, but the mechanism of β-blockers as antihypertensives is unclear.31 Evidence supporting the role of β-adrenergic antagonists in TE is limited to case reports. Widespread alopecia across the scalp and arms was noted in a man in his 30s several months after starting propranolol.32 Biopsy of an affected area of the scalp demonstrated an increased number of telogen follicles with no other abnormalities. Near-complete resolution of alopecia was seen 4 months following cessation of propranolol, which recurred within 4 weeks of rechallenge.
Minoxidil—Oral minoxidil originally was approved for use in patients with resistant hypertension, defined as blood pressure elevated above goal despite concurrent use of the maximum dose of 3 classes of antihypertensives.36 Unlike other antihypertensive medications, minoxidil appears to cause reversible hypertrichosis that affects nearly all patients using oral minoxidil for longer than 1 month.37 This common adverse effect was a desired outcome in patients affected by hair loss, and a topical formulation of minoxidil was approved for androgenetic alopecia in men and women in 1988 and 1991, respectively.38 Since its approval, topical minoxidil has been commonly prescribed in the treatment of several types of alopecia, though evidence of its efficacy in the treatment of TE is limited.39,40 Low-dose oral minoxidil also has been reported to aid hair growth in androgenetic alopecia and TE.41 Taken orally, minoxidil is converted by sulfotransferases in the liver to minoxidil sulfate, which causes opening of plasma membrane adenosine triphosphate–sensitive potassium channels.42-44 The subsequent membrane hyperpolarization reduces calcium ion influx, which also reduces cell excitability, and inhibits contraction in vascular smooth muscle cells, which results in the arteriolar vasodilatory and antihypertensive effects of minoxidil.43,45 The potassium channel–opening effects of minoxidil may underly its hair growth stimulatory action. Unrelated potassium channel openers such as diazoxide and pinacidil also cause hypertrichosis.46-48 An animal study showed that topical minoxidil, cromakalim (potassium channel opener), and P1075 (pinacidil analog) applied daily to the scalps of balding stump-tailed macaques led to significant increases in hair weight over a 20-week treatment period compared with the vehicle control group (P<.05 for minoxidil 100 mM and 250 mM, cromakalim 100 mM, and P1075 100 mM and 250 mM).50 For minoxidil, this effect on hair growth appears to be dose dependent, as cumulative hair weights for the study period were significantly greater in the 250-mM concentration compared with 100-mM minoxidil (P<.05).49 The potassium channel–opening activity of minoxidil may induce stimulation of microcirculation around hair follicles conducive to hair growth.50 Other proposed mechanisms for hair growth with minoxidil include effects on keratinocyte and fibroblast cell proliferation,51-53 collagen synthesis,52,54 and prostaglandin activity.44,55
Final Thoughts
Medication-induced TE is an undesired adverse effect of many commonly used medications, including retinoids, azole antifungals, mood stabilizers, anticoagulants, and antihypertensives. In part 156 of this 2-part series, we reviewed the existing literature on hair loss from retinoids, antifungals, and psychotropic medications. Herein, we focused on anticoagulant and antihypertensive medications as potential culprits of TE. Heparin and its derivatives have been associated with development of diffuse alopecia weeks to months after the start of treatment. Alopecia associated with ACE inhibitors and β-blockers has been described only in case reports, suggesting that they may be unlikely causes of TE. In contrast, minoxidil is an antihypertensive that can result in hypertrichosis and is used in the treatment of androgenetic alopecia. It should not be assumed that medications that share an indication or are part of the same medication class would similarly induce TE. The development of diffuse nonscarring alopecia should prompt suspicion for TE and thorough investigation of medications initiated 1 to 6 months prior to onset of clinically apparent alopecia. Suspected culprit medications should be carefully assessed for their likelihood of inducing TE.
- Angiolillo DJ, Bhatt DL, Cannon CP, et al. Antithrombotic therapy in patients with atrial fibrillation treated with oral anticoagulation undergoing percutaneous coronary intervention: a North American perspective: 2021 update. Circulation. 2021;143:583-596. doi:10.1161 /circulationaha.120.050438
- Kearon C, Kahn SR. Long-term treatment of venous thromboembolism. Blood. 2020;135:317-325. doi:10.1182/blood.2019002364
- Frishman WH, Ribner HS. Anticoagulation in myocardial infarction: modern approach to an old problem. Am J Cardiol. 1979;43:1207-1213. doi:10.1016/0002-9149(79)90155-3
- Khorana AA, Mackman N, Falanga A, et al. Cancer-associated venous thromboembolism. Nat Rev Dis Primers. 2022;8:11. doi:10.1038 /s41572-022-00336-y
- Umerah CO, Momodu, II. Anticoagulation. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK560651/
- Beurskens DMH, Huckriede JP, Schrijver R, et al. The anticoagulant and nonanticoagulant properties of heparin. Thromb Haemost. 2020;120:1371-1383. doi:10.1055/s-0040-1715460
- Hirsh J, Dalen J, Anderson DR, et al. Oral anticoagulants: mechanism of action, clinical effectiveness, and optimal therapeutic range. Chest. 2001;119(1 suppl):8S-21S. doi:10.1378/chest.119.1_suppl.8s
- Holbrook AM, Pereira JA, Labiris R, et al. Systematic overview of warfarin and its drug and food interactions. Arch Intern Med. 2005;165:1095-1106. doi:10.1001/archinte.165.10.1095
- Watras MM, Patel JP, Arya R. Traditional anticoagulants and hair loss: a role for direct oral anticoagulants? a review of the literature. Drugs Real World Outcomes. 2016;3:1-6. doi:10.1007/s40801-015-0056-z
- Heparin sodium. Product information. Hepalink USA Inc; January 2022. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/c4c6bc1f-e0c7-fd0d-e053-2995a90abdef/spl-doc?hl=heparin
- Warfarin sodium. Product information. Bryant Ranch Prepack; April 2023. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/c41b7c23-8053-428a-ac1d-8395e714c2f1/spl-doc?hl=alopecia%7Cwarfarin#section-6
- Hirsh J. Low-molecular-weight heparin. Circulation. 1998;98:1575-1582. doi:10.1161/01.CIR.98.15.1575
- Paus R. Hair growth inhibition by heparin in mice: a model system for studying the modulation of epithelial cell growth by glycosaminoglycans? Br J Dermatol. 1991;124:415-422. doi:10.1111/j.1365-2133.1991.tb00618.x
- Ma SN, Mao ZX, Wu Y, et al. The anti-cancer properties of heparin and its derivatives: a review and prospect. Cell Adh Migr. 2020;14:118-128. doi:10.1080/19336918.2020.1767489
- Choi JU, Chung SW, Al-Hilal TA, et al. A heparin conjugate, LHbisD4, inhibits lymphangiogenesis and attenuates lymph node metastasis by blocking VEGF-C signaling pathway. Biomaterials. 2017;139:56-66. doi:0.1016/j.biomaterials.2017.05.026
- Klerk CP, Smorenburg SM, Otten HM, et al. The effect of low molecular weight heparin on survival in patients with advanced malignancy. J Clin Oncol. 2005;23:2130-2135. doi:10.1200/jco.2005.03.134
- Altinbas M, Coskun HS, Er O, et al. A randomized clinical trial of combination chemotherapy with and without low-molecular-weight heparin in small cell lung cancer. J Thromb Haemost. 2004;2:1266-1271. doi:10.1111/j.1538-7836.2004.00871.x
- Weyand AC, Shavit JA. Agent specific effects of anticoagulant induced alopecia. Res Pract Thromb Haemost. 2017;1:90-92. doi:10.1002 /rth2.12001
- Bonaldo G, Vaccheri A, Motola D. Direct-acting oral anticoagulants and alopecia: the valuable support of postmarketing data. Br J Clin Pharmacol. 2020;86:1654-1660. doi:10.1111/bcp.14221
- Fuchs FD, Whelton PK. High blood pressure and cardiovascular disease. Hypertension. 2020;75:285-292. doi:10.1161 /HYPERTENSIONAHA.119.14240
- Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140:E596-E646. doi:10.1161/CIR.0000000000000678
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2013;128:E240-E327. doi:10.1161 /CIR.0b013e31829e8776
- Effects of enalapril on mortality in severe congestive heart failure. results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). N Engl J Med. 1987;316:1429-1435. doi:10.1056 /nejm198706043162301
- Kataria V, Wang H, Wald JW, et al. Lisinopril-induced alopecia: a case report. J Pharm Pract. 2017;30:562-566. doi:10.1177/0897190016652554
- Motel PJ. Captopril and alopecia: a case report and review of known cutaneous reactions in captopril use. J Am Acad Dermatol. 1990;23:124-125. doi:10.1016/s0190-9622(08)81205-4
- Leaker B, Whitworth JA. Alopecia associated with captopril treatment. Aust N Z J Med. 1984;14:866. doi:10.1111/j.1445-5994.1984.tb03797.x
- Ahmad S. Enalapril and reversible alopecia. Arch Intern Med. 1991;151:404.
- Bicket DP. Using ACE inhibitors appropriately. Am Fam Physician. 2002;66:461-468.
- Captopril. Product information. Bryant Ranch Prepack; May 2023. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/563737c5-4d63-4957-8022-e3bc3112dfac/spl-doc?hl=captopril
- Farzam K, Jan A. Beta blockers. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK532906/
- Mason RP, Giles TD, Sowers JR. Evolving mechanisms of action of beta blockers: focus on nebivolol. J Cardiovasc Pharmacol. 2009; 54:123-128.
- Martin CM, Southwick EG, Maibach HI. Propranolol induced alopecia. Am Heart J. 1973;86:236-237. doi:10.1016/0002-8703(73)90250-0
- Graeber CW, Lapkin RA. Metoprolol and alopecia. Cutis. 1981; 28:633-634.
- Hilder RJ. Propranolol and alopecia. Cutis. 1979;24:63-64.
- Coreg. Prescribing information. Woodward Pharma Services LLC; 2023. Accessed December 11, 2023. https://www.accessdata.fda.gov/spl/data/34aa881a-3df4-460b-acad-fb9975ca3a06/34aa881a-3df4-460b-acad-fb9975ca3a06.xml
- Carey RM, Calhoun DA, Bakris GL, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72:E53-E90. doi:10.1161/hyp.0000000000000084
- Campese VM. Minoxidil: a review of its pharmacological properties and therapeutic use. Drugs. 1981;22:257-278. doi:10.2165/00003495-198122040-00001
- Heymann WR. Coming full circle (almost): low dose oral minoxidil for alopecia. J Am Acad Dermatol. 2021;84:613-614. doi:10.1016/j .jaad.2020.12.053
- Yin S, Zhang B, Lin J, et al. Development of purification process for dual-function recombinant human heavy-chain ferritin by the investigation of genetic modification impact on conformation. Eng Life Sci. 2021;21:630-642. doi:10.1002/elsc.202000105
- Mysore V, Parthasaradhi A, Kharkar RD, et al. Expert consensus on the management of telogen effluvium in India. Int J Trichology. 2019;11:107-112.
- Gupta AK, Talukder M, Shemar A, et al. Low-dose oral minoxidil for alopecia: a comprehensive review [published online September 27, 2023]. Skin Appendage Disord. doi:10.1159/000531890
- Meisheri KD, Cipkus LA, Taylor CJ. Mechanism of action of minoxidil sulfate-induced vasodilation: a role for increased K+ permeability. J Pharmacol Exp Ther. 1988;245:751-760.
- Winquist RJ, Heaney LA, Wallace AA, et al. Glyburide blocks the relaxation response to BRL 34915 (cromakalim), minoxidil sulfate and diazoxide in vascular smooth muscle. J Pharmacol Exp Ther. 1989;248:149-56.
- Messenger AG, Rundegren J. Minoxidil: mechanisms of action on hair growth. Br J Dermatol. 2004;150:186-194. doi:10.1111/j .1365-2133.2004.05785.x
- Alijotas-Reig J, García GV, Velthuis PJ, et al. Inflammatory immunemediated adverse reactions induced by COVID-19 vaccines in previously injected patients with soft tissue fillers: a case series of 20 patients. J Cosmet Dermatol. 2022;21:3181-3187. doi: 10.1111/jocd.15117
- Boskabadi SJ, Ramezaninejad S, Sohrab M, et al. Diazoxideinduced hypertrichosis in a neonate with transient hyperinsulinism. Clin Med Insights Case Rep. 2023;16:11795476231151330. doi:10.1177/11795476231151330
- Burton JL, Schutt WH, Caldwell IW. Hypertrichosis due to diazoxide. Br J Dermatol. 1975;93:707-711. doi:10.1111/j.1365-2133.1975.tb05123.x
- Goldberg MR. Clinical pharmacology of pinacidil, a prototype for drugs that affect potassium channels. J Cardiovasc Pharmacol. 1988;12 suppl 2:S41-S47. doi: 10.1097/00005344-198812002-00008
- Buhl AE, Waldon DJ, Conrad SJ, et al. Potassium channel conductance: a mechanism affecting hair growth both in vitro and in vivo. J Invest Dermatol. 1992;98:315-319. doi:10.1111/1523-1747.ep12499788
- Patel P, Nessel TA, Kumar DD. Minoxidil. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK482378/
- O’Keefe E, Payne RE Jr. Minoxidil: inhibition of proliferation of keratinocytes in vitro. J Invest Dermatol. 1991;97:534-536. doi:10.1111/1523-1747.ep12481560
- Murad S, Pinnell SR. Suppression of fibroblast proliferation and lysyl hydroxylase activity by minoxidil. J Biol Chem. 1987;262:11973-11978.
- Baden HP, Kubilus J. Effect of minoxidil on cultured keratinocytes. J Invest Dermatol. 1983;81:558-560. doi:10.1111/1523-1747.ep12523220
- Murad S, Walker LC, Tajima S, et al. Minimum structural requirements for minoxidil inhibition of lysyl hydroxylase in cultured fibroblasts. Arch Biochem Biophys. 1994;308:42-47. doi:10.1006/abbi.1994.1006
- Kvedar JC, Baden HP, Levine L. Selective inhibition by minoxidil of prostacyclin production by cells in culture. Biochem Pharmacol. 1988;37:867-874. doi:0.1016/0006-2952(88)90174-8
- Zhang D, LaSenna C, Shields BE. Culprits of medication-induced telogen effluvium, part 1. Cutis. 2023;112:267-271.
Medication-induced telogen effluvium (TE) is a nonscarring alopecia that typically is reversible. Appropriate management requires identification of the underlying trigger and cessation of potential culprit medications. In part 2 of this series, we review anticoagulant and antihypertensive medications as potential contributors to TE.
Anticoagulants
Anticoagulants target various parts of the coagulation cascade to prevent clot formation in patients with conditions that increase their risk for thromboembolic events. Common indications for initiating anticoagulant therapy include atrial fibrillation,1 venous thromboembolism,2 acute myocardial infarction,3 malignancy,4 and hypercoagulable states.5 Traditional anticoagulants include heparin and warfarin. Heparin is a glycosaminoglycan that exerts its anticoagulant effects through binding with antithrombin, greatly increasing its inactivation of thrombin and factor Xa of the coagulation cascade.6 Warfarin is a coumarin derivative that inhibits activation of vitamin K, subsequently limiting the function of vitamin K–dependent factors II, VII, IX, and X.7,8 Watras et al9 noted that heparin and warfarin were implicated in alopecia as their clinical use became widespread throughout the mid-20th century. Onset of alopecia following the use of heparin or warfarin was reported at 3 weeks to 3 months following medication initiation, with most cases clinically consistent with TE.9 Heparin and warfarin both have alopecia reported as a potential adverse effect in their structured product labeling documents.10,11
Heparin is further classified into unfractionated heparin (UFH) and low-molecular-weight heparin (LMWH); the latter is a heterogeneous group of medications derived from chemical or enzymatic depolymerization of UFH.12 In contrast to UFH, LMWH exerts its anticoagulant effects through inactivation of factor Xa without the ability to bind thrombin.12 An animal study using anagen-induced mice demonstrated that intraperitoneal administration of heparin inhibited the development of anagen follicles, while in vitro studies showed that the addition of heparin inhibited mouse dermal papilla cell proliferation.13 Other animal and in vitro studies have examined the inhibitory effects of heparin on signaling pathways in tumor lymphangiogenesis, including the vascular endothelial growth factor C/vascular endothelial growth factor receptor 3 axis.14,15 Clinically, it has been demonstrated that heparin, especially LMWHs, may be associated with a survival benefit among certain cancer patients,16,17 with the impact of LMWHs attributed to antimitotic and antimetastatic effects of heparin on tumor growth.14 It is hypothesized that such antiangiogenic and antimitotic effects also are involved in the pathomechanisms of heparin-induced alopecia.18
More recently, the use of direct oral anticoagulants (DOACs) such as dabigatran, rivaroxaban, and apixaban has increased due to their more favorable adverse-effect profile and minimal monitoring requirements. Bonaldo et al19 conducted an analysis of reports submitted to the World Health Organization’s VigiBase database of alopecia associated with DOACs until May 2, 2018. They found 1316 nonduplicate DOAC-induced cases of alopecia, with rivaroxaban as the most reported drug associated with alopecia development (58.8% [774/1316]). Only 4 cases demonstrated alopecia with DOAC rechallenge, suggesting onset of alopecia may have been unrelated to DOAC use or caused by a different trigger. Among 243 cases with a documented time to onset of alopecia, the median was 28 days, with an interquartile range of 63 days. Because TE most commonly occurs 3 to 4 months after the inciting event or medication trigger, there is little evidence to suggest DOACs as the cause of TE, and the observed cases of alopecia may be attributable to another preceding medical event and/or medication exposure.19 More studies are needed to examine the impact of anticoagulant medications on the hair cycle.
Antihypertensives
Hypertension is a modifiable risk factor for several cardiovascular diseases.20 According to the 2019 American College of Cardiology/American Heart Association Guideline on the Primary Prevention of Cardiovascular Disease,21 first-line medications include thiazide diuretics, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, and angiotensin receptor blockers (ARBs).
Angiotensin-converting enzyme inhibitors exert their antihypertensive effects by reducing conversion of angiotensin I to angiotensin II, thereby limiting the downstream effects of vasoconstriction as well as sodium and water retention. Given the proven mortality benefit of ACE inhibition in patients with congestive heart failure, ACE inhibitors are used as first-line therapy in these patients.22,23 Alopecia associated with ACE inhibitors is rare and limited to case reports following their introduction and approval in 1981.24-28 In one case, a woman in her 60s with congestive heart failure initiated captopril with development of an erythematous pruritic rash on the extremities and diffuse scalp hair loss 2 months later; spontaneous hair growth resumed 1 month following captopril discontinuation.25 In this case, the hair loss may be secondary to the drug eruption rather than true medication-induced TE. Initiation of enalapril in a woman in her 30s with hypertension was associated with diffuse scalp alopecia 4 weeks later that resolved with cessation of the suspected culprit, enalapril; rechallenge with enalapril several months later reproduced the hair loss.27 Given limited reports of ACE inhibitor–associated hair loss relative to their pervasive use, a direct causal role between ACE inhibition and TE is unlikely, or it has not been rigorously identified. The structured product labeling for captopril includes alopecia in its list of adverse effects reported in approximately 0.5% to 2% of patients but did not appear at increased frequency compared to placebo or other treatments used in controlled trials.29 Alternative inciting causes of alopecia in patients prescribed ACE inhibitors may include use of other medications, hospitalization, or metabolic derangements related to their underlying cardiac disease.
Although not indicated as a primary treatment for hypertension, β-blockers have US Food and Drug Administration approval for the treatment of certain arrhythmias, hypertension, heart failure, myocardial infarction, hyperthyroidism, and other conditions.30 β-Blockers are competitive antagonists of β-adrenergic receptors that limit the production of intracellular cyclic adenosine monophosphate, but the mechanism of β-blockers as antihypertensives is unclear.31 Evidence supporting the role of β-adrenergic antagonists in TE is limited to case reports. Widespread alopecia across the scalp and arms was noted in a man in his 30s several months after starting propranolol.32 Biopsy of an affected area of the scalp demonstrated an increased number of telogen follicles with no other abnormalities. Near-complete resolution of alopecia was seen 4 months following cessation of propranolol, which recurred within 4 weeks of rechallenge.
Minoxidil—Oral minoxidil originally was approved for use in patients with resistant hypertension, defined as blood pressure elevated above goal despite concurrent use of the maximum dose of 3 classes of antihypertensives.36 Unlike other antihypertensive medications, minoxidil appears to cause reversible hypertrichosis that affects nearly all patients using oral minoxidil for longer than 1 month.37 This common adverse effect was a desired outcome in patients affected by hair loss, and a topical formulation of minoxidil was approved for androgenetic alopecia in men and women in 1988 and 1991, respectively.38 Since its approval, topical minoxidil has been commonly prescribed in the treatment of several types of alopecia, though evidence of its efficacy in the treatment of TE is limited.39,40 Low-dose oral minoxidil also has been reported to aid hair growth in androgenetic alopecia and TE.41 Taken orally, minoxidil is converted by sulfotransferases in the liver to minoxidil sulfate, which causes opening of plasma membrane adenosine triphosphate–sensitive potassium channels.42-44 The subsequent membrane hyperpolarization reduces calcium ion influx, which also reduces cell excitability, and inhibits contraction in vascular smooth muscle cells, which results in the arteriolar vasodilatory and antihypertensive effects of minoxidil.43,45 The potassium channel–opening effects of minoxidil may underly its hair growth stimulatory action. Unrelated potassium channel openers such as diazoxide and pinacidil also cause hypertrichosis.46-48 An animal study showed that topical minoxidil, cromakalim (potassium channel opener), and P1075 (pinacidil analog) applied daily to the scalps of balding stump-tailed macaques led to significant increases in hair weight over a 20-week treatment period compared with the vehicle control group (P<.05 for minoxidil 100 mM and 250 mM, cromakalim 100 mM, and P1075 100 mM and 250 mM).50 For minoxidil, this effect on hair growth appears to be dose dependent, as cumulative hair weights for the study period were significantly greater in the 250-mM concentration compared with 100-mM minoxidil (P<.05).49 The potassium channel–opening activity of minoxidil may induce stimulation of microcirculation around hair follicles conducive to hair growth.50 Other proposed mechanisms for hair growth with minoxidil include effects on keratinocyte and fibroblast cell proliferation,51-53 collagen synthesis,52,54 and prostaglandin activity.44,55
Final Thoughts
Medication-induced TE is an undesired adverse effect of many commonly used medications, including retinoids, azole antifungals, mood stabilizers, anticoagulants, and antihypertensives. In part 156 of this 2-part series, we reviewed the existing literature on hair loss from retinoids, antifungals, and psychotropic medications. Herein, we focused on anticoagulant and antihypertensive medications as potential culprits of TE. Heparin and its derivatives have been associated with development of diffuse alopecia weeks to months after the start of treatment. Alopecia associated with ACE inhibitors and β-blockers has been described only in case reports, suggesting that they may be unlikely causes of TE. In contrast, minoxidil is an antihypertensive that can result in hypertrichosis and is used in the treatment of androgenetic alopecia. It should not be assumed that medications that share an indication or are part of the same medication class would similarly induce TE. The development of diffuse nonscarring alopecia should prompt suspicion for TE and thorough investigation of medications initiated 1 to 6 months prior to onset of clinically apparent alopecia. Suspected culprit medications should be carefully assessed for their likelihood of inducing TE.
Medication-induced telogen effluvium (TE) is a nonscarring alopecia that typically is reversible. Appropriate management requires identification of the underlying trigger and cessation of potential culprit medications. In part 2 of this series, we review anticoagulant and antihypertensive medications as potential contributors to TE.
Anticoagulants
Anticoagulants target various parts of the coagulation cascade to prevent clot formation in patients with conditions that increase their risk for thromboembolic events. Common indications for initiating anticoagulant therapy include atrial fibrillation,1 venous thromboembolism,2 acute myocardial infarction,3 malignancy,4 and hypercoagulable states.5 Traditional anticoagulants include heparin and warfarin. Heparin is a glycosaminoglycan that exerts its anticoagulant effects through binding with antithrombin, greatly increasing its inactivation of thrombin and factor Xa of the coagulation cascade.6 Warfarin is a coumarin derivative that inhibits activation of vitamin K, subsequently limiting the function of vitamin K–dependent factors II, VII, IX, and X.7,8 Watras et al9 noted that heparin and warfarin were implicated in alopecia as their clinical use became widespread throughout the mid-20th century. Onset of alopecia following the use of heparin or warfarin was reported at 3 weeks to 3 months following medication initiation, with most cases clinically consistent with TE.9 Heparin and warfarin both have alopecia reported as a potential adverse effect in their structured product labeling documents.10,11
Heparin is further classified into unfractionated heparin (UFH) and low-molecular-weight heparin (LMWH); the latter is a heterogeneous group of medications derived from chemical or enzymatic depolymerization of UFH.12 In contrast to UFH, LMWH exerts its anticoagulant effects through inactivation of factor Xa without the ability to bind thrombin.12 An animal study using anagen-induced mice demonstrated that intraperitoneal administration of heparin inhibited the development of anagen follicles, while in vitro studies showed that the addition of heparin inhibited mouse dermal papilla cell proliferation.13 Other animal and in vitro studies have examined the inhibitory effects of heparin on signaling pathways in tumor lymphangiogenesis, including the vascular endothelial growth factor C/vascular endothelial growth factor receptor 3 axis.14,15 Clinically, it has been demonstrated that heparin, especially LMWHs, may be associated with a survival benefit among certain cancer patients,16,17 with the impact of LMWHs attributed to antimitotic and antimetastatic effects of heparin on tumor growth.14 It is hypothesized that such antiangiogenic and antimitotic effects also are involved in the pathomechanisms of heparin-induced alopecia.18
More recently, the use of direct oral anticoagulants (DOACs) such as dabigatran, rivaroxaban, and apixaban has increased due to their more favorable adverse-effect profile and minimal monitoring requirements. Bonaldo et al19 conducted an analysis of reports submitted to the World Health Organization’s VigiBase database of alopecia associated with DOACs until May 2, 2018. They found 1316 nonduplicate DOAC-induced cases of alopecia, with rivaroxaban as the most reported drug associated with alopecia development (58.8% [774/1316]). Only 4 cases demonstrated alopecia with DOAC rechallenge, suggesting onset of alopecia may have been unrelated to DOAC use or caused by a different trigger. Among 243 cases with a documented time to onset of alopecia, the median was 28 days, with an interquartile range of 63 days. Because TE most commonly occurs 3 to 4 months after the inciting event or medication trigger, there is little evidence to suggest DOACs as the cause of TE, and the observed cases of alopecia may be attributable to another preceding medical event and/or medication exposure.19 More studies are needed to examine the impact of anticoagulant medications on the hair cycle.
Antihypertensives
Hypertension is a modifiable risk factor for several cardiovascular diseases.20 According to the 2019 American College of Cardiology/American Heart Association Guideline on the Primary Prevention of Cardiovascular Disease,21 first-line medications include thiazide diuretics, calcium channel blockers, angiotensin-converting enzyme (ACE) inhibitors, and angiotensin receptor blockers (ARBs).
Angiotensin-converting enzyme inhibitors exert their antihypertensive effects by reducing conversion of angiotensin I to angiotensin II, thereby limiting the downstream effects of vasoconstriction as well as sodium and water retention. Given the proven mortality benefit of ACE inhibition in patients with congestive heart failure, ACE inhibitors are used as first-line therapy in these patients.22,23 Alopecia associated with ACE inhibitors is rare and limited to case reports following their introduction and approval in 1981.24-28 In one case, a woman in her 60s with congestive heart failure initiated captopril with development of an erythematous pruritic rash on the extremities and diffuse scalp hair loss 2 months later; spontaneous hair growth resumed 1 month following captopril discontinuation.25 In this case, the hair loss may be secondary to the drug eruption rather than true medication-induced TE. Initiation of enalapril in a woman in her 30s with hypertension was associated with diffuse scalp alopecia 4 weeks later that resolved with cessation of the suspected culprit, enalapril; rechallenge with enalapril several months later reproduced the hair loss.27 Given limited reports of ACE inhibitor–associated hair loss relative to their pervasive use, a direct causal role between ACE inhibition and TE is unlikely, or it has not been rigorously identified. The structured product labeling for captopril includes alopecia in its list of adverse effects reported in approximately 0.5% to 2% of patients but did not appear at increased frequency compared to placebo or other treatments used in controlled trials.29 Alternative inciting causes of alopecia in patients prescribed ACE inhibitors may include use of other medications, hospitalization, or metabolic derangements related to their underlying cardiac disease.
Although not indicated as a primary treatment for hypertension, β-blockers have US Food and Drug Administration approval for the treatment of certain arrhythmias, hypertension, heart failure, myocardial infarction, hyperthyroidism, and other conditions.30 β-Blockers are competitive antagonists of β-adrenergic receptors that limit the production of intracellular cyclic adenosine monophosphate, but the mechanism of β-blockers as antihypertensives is unclear.31 Evidence supporting the role of β-adrenergic antagonists in TE is limited to case reports. Widespread alopecia across the scalp and arms was noted in a man in his 30s several months after starting propranolol.32 Biopsy of an affected area of the scalp demonstrated an increased number of telogen follicles with no other abnormalities. Near-complete resolution of alopecia was seen 4 months following cessation of propranolol, which recurred within 4 weeks of rechallenge.
Minoxidil—Oral minoxidil originally was approved for use in patients with resistant hypertension, defined as blood pressure elevated above goal despite concurrent use of the maximum dose of 3 classes of antihypertensives.36 Unlike other antihypertensive medications, minoxidil appears to cause reversible hypertrichosis that affects nearly all patients using oral minoxidil for longer than 1 month.37 This common adverse effect was a desired outcome in patients affected by hair loss, and a topical formulation of minoxidil was approved for androgenetic alopecia in men and women in 1988 and 1991, respectively.38 Since its approval, topical minoxidil has been commonly prescribed in the treatment of several types of alopecia, though evidence of its efficacy in the treatment of TE is limited.39,40 Low-dose oral minoxidil also has been reported to aid hair growth in androgenetic alopecia and TE.41 Taken orally, minoxidil is converted by sulfotransferases in the liver to minoxidil sulfate, which causes opening of plasma membrane adenosine triphosphate–sensitive potassium channels.42-44 The subsequent membrane hyperpolarization reduces calcium ion influx, which also reduces cell excitability, and inhibits contraction in vascular smooth muscle cells, which results in the arteriolar vasodilatory and antihypertensive effects of minoxidil.43,45 The potassium channel–opening effects of minoxidil may underly its hair growth stimulatory action. Unrelated potassium channel openers such as diazoxide and pinacidil also cause hypertrichosis.46-48 An animal study showed that topical minoxidil, cromakalim (potassium channel opener), and P1075 (pinacidil analog) applied daily to the scalps of balding stump-tailed macaques led to significant increases in hair weight over a 20-week treatment period compared with the vehicle control group (P<.05 for minoxidil 100 mM and 250 mM, cromakalim 100 mM, and P1075 100 mM and 250 mM).50 For minoxidil, this effect on hair growth appears to be dose dependent, as cumulative hair weights for the study period were significantly greater in the 250-mM concentration compared with 100-mM minoxidil (P<.05).49 The potassium channel–opening activity of minoxidil may induce stimulation of microcirculation around hair follicles conducive to hair growth.50 Other proposed mechanisms for hair growth with minoxidil include effects on keratinocyte and fibroblast cell proliferation,51-53 collagen synthesis,52,54 and prostaglandin activity.44,55
Final Thoughts
Medication-induced TE is an undesired adverse effect of many commonly used medications, including retinoids, azole antifungals, mood stabilizers, anticoagulants, and antihypertensives. In part 156 of this 2-part series, we reviewed the existing literature on hair loss from retinoids, antifungals, and psychotropic medications. Herein, we focused on anticoagulant and antihypertensive medications as potential culprits of TE. Heparin and its derivatives have been associated with development of diffuse alopecia weeks to months after the start of treatment. Alopecia associated with ACE inhibitors and β-blockers has been described only in case reports, suggesting that they may be unlikely causes of TE. In contrast, minoxidil is an antihypertensive that can result in hypertrichosis and is used in the treatment of androgenetic alopecia. It should not be assumed that medications that share an indication or are part of the same medication class would similarly induce TE. The development of diffuse nonscarring alopecia should prompt suspicion for TE and thorough investigation of medications initiated 1 to 6 months prior to onset of clinically apparent alopecia. Suspected culprit medications should be carefully assessed for their likelihood of inducing TE.
- Angiolillo DJ, Bhatt DL, Cannon CP, et al. Antithrombotic therapy in patients with atrial fibrillation treated with oral anticoagulation undergoing percutaneous coronary intervention: a North American perspective: 2021 update. Circulation. 2021;143:583-596. doi:10.1161 /circulationaha.120.050438
- Kearon C, Kahn SR. Long-term treatment of venous thromboembolism. Blood. 2020;135:317-325. doi:10.1182/blood.2019002364
- Frishman WH, Ribner HS. Anticoagulation in myocardial infarction: modern approach to an old problem. Am J Cardiol. 1979;43:1207-1213. doi:10.1016/0002-9149(79)90155-3
- Khorana AA, Mackman N, Falanga A, et al. Cancer-associated venous thromboembolism. Nat Rev Dis Primers. 2022;8:11. doi:10.1038 /s41572-022-00336-y
- Umerah CO, Momodu, II. Anticoagulation. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK560651/
- Beurskens DMH, Huckriede JP, Schrijver R, et al. The anticoagulant and nonanticoagulant properties of heparin. Thromb Haemost. 2020;120:1371-1383. doi:10.1055/s-0040-1715460
- Hirsh J, Dalen J, Anderson DR, et al. Oral anticoagulants: mechanism of action, clinical effectiveness, and optimal therapeutic range. Chest. 2001;119(1 suppl):8S-21S. doi:10.1378/chest.119.1_suppl.8s
- Holbrook AM, Pereira JA, Labiris R, et al. Systematic overview of warfarin and its drug and food interactions. Arch Intern Med. 2005;165:1095-1106. doi:10.1001/archinte.165.10.1095
- Watras MM, Patel JP, Arya R. Traditional anticoagulants and hair loss: a role for direct oral anticoagulants? a review of the literature. Drugs Real World Outcomes. 2016;3:1-6. doi:10.1007/s40801-015-0056-z
- Heparin sodium. Product information. Hepalink USA Inc; January 2022. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/c4c6bc1f-e0c7-fd0d-e053-2995a90abdef/spl-doc?hl=heparin
- Warfarin sodium. Product information. Bryant Ranch Prepack; April 2023. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/c41b7c23-8053-428a-ac1d-8395e714c2f1/spl-doc?hl=alopecia%7Cwarfarin#section-6
- Hirsh J. Low-molecular-weight heparin. Circulation. 1998;98:1575-1582. doi:10.1161/01.CIR.98.15.1575
- Paus R. Hair growth inhibition by heparin in mice: a model system for studying the modulation of epithelial cell growth by glycosaminoglycans? Br J Dermatol. 1991;124:415-422. doi:10.1111/j.1365-2133.1991.tb00618.x
- Ma SN, Mao ZX, Wu Y, et al. The anti-cancer properties of heparin and its derivatives: a review and prospect. Cell Adh Migr. 2020;14:118-128. doi:10.1080/19336918.2020.1767489
- Choi JU, Chung SW, Al-Hilal TA, et al. A heparin conjugate, LHbisD4, inhibits lymphangiogenesis and attenuates lymph node metastasis by blocking VEGF-C signaling pathway. Biomaterials. 2017;139:56-66. doi:0.1016/j.biomaterials.2017.05.026
- Klerk CP, Smorenburg SM, Otten HM, et al. The effect of low molecular weight heparin on survival in patients with advanced malignancy. J Clin Oncol. 2005;23:2130-2135. doi:10.1200/jco.2005.03.134
- Altinbas M, Coskun HS, Er O, et al. A randomized clinical trial of combination chemotherapy with and without low-molecular-weight heparin in small cell lung cancer. J Thromb Haemost. 2004;2:1266-1271. doi:10.1111/j.1538-7836.2004.00871.x
- Weyand AC, Shavit JA. Agent specific effects of anticoagulant induced alopecia. Res Pract Thromb Haemost. 2017;1:90-92. doi:10.1002 /rth2.12001
- Bonaldo G, Vaccheri A, Motola D. Direct-acting oral anticoagulants and alopecia: the valuable support of postmarketing data. Br J Clin Pharmacol. 2020;86:1654-1660. doi:10.1111/bcp.14221
- Fuchs FD, Whelton PK. High blood pressure and cardiovascular disease. Hypertension. 2020;75:285-292. doi:10.1161 /HYPERTENSIONAHA.119.14240
- Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140:E596-E646. doi:10.1161/CIR.0000000000000678
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2013;128:E240-E327. doi:10.1161 /CIR.0b013e31829e8776
- Effects of enalapril on mortality in severe congestive heart failure. results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). N Engl J Med. 1987;316:1429-1435. doi:10.1056 /nejm198706043162301
- Kataria V, Wang H, Wald JW, et al. Lisinopril-induced alopecia: a case report. J Pharm Pract. 2017;30:562-566. doi:10.1177/0897190016652554
- Motel PJ. Captopril and alopecia: a case report and review of known cutaneous reactions in captopril use. J Am Acad Dermatol. 1990;23:124-125. doi:10.1016/s0190-9622(08)81205-4
- Leaker B, Whitworth JA. Alopecia associated with captopril treatment. Aust N Z J Med. 1984;14:866. doi:10.1111/j.1445-5994.1984.tb03797.x
- Ahmad S. Enalapril and reversible alopecia. Arch Intern Med. 1991;151:404.
- Bicket DP. Using ACE inhibitors appropriately. Am Fam Physician. 2002;66:461-468.
- Captopril. Product information. Bryant Ranch Prepack; May 2023. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/563737c5-4d63-4957-8022-e3bc3112dfac/spl-doc?hl=captopril
- Farzam K, Jan A. Beta blockers. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK532906/
- Mason RP, Giles TD, Sowers JR. Evolving mechanisms of action of beta blockers: focus on nebivolol. J Cardiovasc Pharmacol. 2009; 54:123-128.
- Martin CM, Southwick EG, Maibach HI. Propranolol induced alopecia. Am Heart J. 1973;86:236-237. doi:10.1016/0002-8703(73)90250-0
- Graeber CW, Lapkin RA. Metoprolol and alopecia. Cutis. 1981; 28:633-634.
- Hilder RJ. Propranolol and alopecia. Cutis. 1979;24:63-64.
- Coreg. Prescribing information. Woodward Pharma Services LLC; 2023. Accessed December 11, 2023. https://www.accessdata.fda.gov/spl/data/34aa881a-3df4-460b-acad-fb9975ca3a06/34aa881a-3df4-460b-acad-fb9975ca3a06.xml
- Carey RM, Calhoun DA, Bakris GL, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72:E53-E90. doi:10.1161/hyp.0000000000000084
- Campese VM. Minoxidil: a review of its pharmacological properties and therapeutic use. Drugs. 1981;22:257-278. doi:10.2165/00003495-198122040-00001
- Heymann WR. Coming full circle (almost): low dose oral minoxidil for alopecia. J Am Acad Dermatol. 2021;84:613-614. doi:10.1016/j .jaad.2020.12.053
- Yin S, Zhang B, Lin J, et al. Development of purification process for dual-function recombinant human heavy-chain ferritin by the investigation of genetic modification impact on conformation. Eng Life Sci. 2021;21:630-642. doi:10.1002/elsc.202000105
- Mysore V, Parthasaradhi A, Kharkar RD, et al. Expert consensus on the management of telogen effluvium in India. Int J Trichology. 2019;11:107-112.
- Gupta AK, Talukder M, Shemar A, et al. Low-dose oral minoxidil for alopecia: a comprehensive review [published online September 27, 2023]. Skin Appendage Disord. doi:10.1159/000531890
- Meisheri KD, Cipkus LA, Taylor CJ. Mechanism of action of minoxidil sulfate-induced vasodilation: a role for increased K+ permeability. J Pharmacol Exp Ther. 1988;245:751-760.
- Winquist RJ, Heaney LA, Wallace AA, et al. Glyburide blocks the relaxation response to BRL 34915 (cromakalim), minoxidil sulfate and diazoxide in vascular smooth muscle. J Pharmacol Exp Ther. 1989;248:149-56.
- Messenger AG, Rundegren J. Minoxidil: mechanisms of action on hair growth. Br J Dermatol. 2004;150:186-194. doi:10.1111/j .1365-2133.2004.05785.x
- Alijotas-Reig J, García GV, Velthuis PJ, et al. Inflammatory immunemediated adverse reactions induced by COVID-19 vaccines in previously injected patients with soft tissue fillers: a case series of 20 patients. J Cosmet Dermatol. 2022;21:3181-3187. doi: 10.1111/jocd.15117
- Boskabadi SJ, Ramezaninejad S, Sohrab M, et al. Diazoxideinduced hypertrichosis in a neonate with transient hyperinsulinism. Clin Med Insights Case Rep. 2023;16:11795476231151330. doi:10.1177/11795476231151330
- Burton JL, Schutt WH, Caldwell IW. Hypertrichosis due to diazoxide. Br J Dermatol. 1975;93:707-711. doi:10.1111/j.1365-2133.1975.tb05123.x
- Goldberg MR. Clinical pharmacology of pinacidil, a prototype for drugs that affect potassium channels. J Cardiovasc Pharmacol. 1988;12 suppl 2:S41-S47. doi: 10.1097/00005344-198812002-00008
- Buhl AE, Waldon DJ, Conrad SJ, et al. Potassium channel conductance: a mechanism affecting hair growth both in vitro and in vivo. J Invest Dermatol. 1992;98:315-319. doi:10.1111/1523-1747.ep12499788
- Patel P, Nessel TA, Kumar DD. Minoxidil. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK482378/
- O’Keefe E, Payne RE Jr. Minoxidil: inhibition of proliferation of keratinocytes in vitro. J Invest Dermatol. 1991;97:534-536. doi:10.1111/1523-1747.ep12481560
- Murad S, Pinnell SR. Suppression of fibroblast proliferation and lysyl hydroxylase activity by minoxidil. J Biol Chem. 1987;262:11973-11978.
- Baden HP, Kubilus J. Effect of minoxidil on cultured keratinocytes. J Invest Dermatol. 1983;81:558-560. doi:10.1111/1523-1747.ep12523220
- Murad S, Walker LC, Tajima S, et al. Minimum structural requirements for minoxidil inhibition of lysyl hydroxylase in cultured fibroblasts. Arch Biochem Biophys. 1994;308:42-47. doi:10.1006/abbi.1994.1006
- Kvedar JC, Baden HP, Levine L. Selective inhibition by minoxidil of prostacyclin production by cells in culture. Biochem Pharmacol. 1988;37:867-874. doi:0.1016/0006-2952(88)90174-8
- Zhang D, LaSenna C, Shields BE. Culprits of medication-induced telogen effluvium, part 1. Cutis. 2023;112:267-271.
- Angiolillo DJ, Bhatt DL, Cannon CP, et al. Antithrombotic therapy in patients with atrial fibrillation treated with oral anticoagulation undergoing percutaneous coronary intervention: a North American perspective: 2021 update. Circulation. 2021;143:583-596. doi:10.1161 /circulationaha.120.050438
- Kearon C, Kahn SR. Long-term treatment of venous thromboembolism. Blood. 2020;135:317-325. doi:10.1182/blood.2019002364
- Frishman WH, Ribner HS. Anticoagulation in myocardial infarction: modern approach to an old problem. Am J Cardiol. 1979;43:1207-1213. doi:10.1016/0002-9149(79)90155-3
- Khorana AA, Mackman N, Falanga A, et al. Cancer-associated venous thromboembolism. Nat Rev Dis Primers. 2022;8:11. doi:10.1038 /s41572-022-00336-y
- Umerah CO, Momodu, II. Anticoagulation. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK560651/
- Beurskens DMH, Huckriede JP, Schrijver R, et al. The anticoagulant and nonanticoagulant properties of heparin. Thromb Haemost. 2020;120:1371-1383. doi:10.1055/s-0040-1715460
- Hirsh J, Dalen J, Anderson DR, et al. Oral anticoagulants: mechanism of action, clinical effectiveness, and optimal therapeutic range. Chest. 2001;119(1 suppl):8S-21S. doi:10.1378/chest.119.1_suppl.8s
- Holbrook AM, Pereira JA, Labiris R, et al. Systematic overview of warfarin and its drug and food interactions. Arch Intern Med. 2005;165:1095-1106. doi:10.1001/archinte.165.10.1095
- Watras MM, Patel JP, Arya R. Traditional anticoagulants and hair loss: a role for direct oral anticoagulants? a review of the literature. Drugs Real World Outcomes. 2016;3:1-6. doi:10.1007/s40801-015-0056-z
- Heparin sodium. Product information. Hepalink USA Inc; January 2022. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/c4c6bc1f-e0c7-fd0d-e053-2995a90abdef/spl-doc?hl=heparin
- Warfarin sodium. Product information. Bryant Ranch Prepack; April 2023. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/c41b7c23-8053-428a-ac1d-8395e714c2f1/spl-doc?hl=alopecia%7Cwarfarin#section-6
- Hirsh J. Low-molecular-weight heparin. Circulation. 1998;98:1575-1582. doi:10.1161/01.CIR.98.15.1575
- Paus R. Hair growth inhibition by heparin in mice: a model system for studying the modulation of epithelial cell growth by glycosaminoglycans? Br J Dermatol. 1991;124:415-422. doi:10.1111/j.1365-2133.1991.tb00618.x
- Ma SN, Mao ZX, Wu Y, et al. The anti-cancer properties of heparin and its derivatives: a review and prospect. Cell Adh Migr. 2020;14:118-128. doi:10.1080/19336918.2020.1767489
- Choi JU, Chung SW, Al-Hilal TA, et al. A heparin conjugate, LHbisD4, inhibits lymphangiogenesis and attenuates lymph node metastasis by blocking VEGF-C signaling pathway. Biomaterials. 2017;139:56-66. doi:0.1016/j.biomaterials.2017.05.026
- Klerk CP, Smorenburg SM, Otten HM, et al. The effect of low molecular weight heparin on survival in patients with advanced malignancy. J Clin Oncol. 2005;23:2130-2135. doi:10.1200/jco.2005.03.134
- Altinbas M, Coskun HS, Er O, et al. A randomized clinical trial of combination chemotherapy with and without low-molecular-weight heparin in small cell lung cancer. J Thromb Haemost. 2004;2:1266-1271. doi:10.1111/j.1538-7836.2004.00871.x
- Weyand AC, Shavit JA. Agent specific effects of anticoagulant induced alopecia. Res Pract Thromb Haemost. 2017;1:90-92. doi:10.1002 /rth2.12001
- Bonaldo G, Vaccheri A, Motola D. Direct-acting oral anticoagulants and alopecia: the valuable support of postmarketing data. Br J Clin Pharmacol. 2020;86:1654-1660. doi:10.1111/bcp.14221
- Fuchs FD, Whelton PK. High blood pressure and cardiovascular disease. Hypertension. 2020;75:285-292. doi:10.1161 /HYPERTENSIONAHA.119.14240
- Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2019;140:E596-E646. doi:10.1161/CIR.0000000000000678
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2013;128:E240-E327. doi:10.1161 /CIR.0b013e31829e8776
- Effects of enalapril on mortality in severe congestive heart failure. results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). N Engl J Med. 1987;316:1429-1435. doi:10.1056 /nejm198706043162301
- Kataria V, Wang H, Wald JW, et al. Lisinopril-induced alopecia: a case report. J Pharm Pract. 2017;30:562-566. doi:10.1177/0897190016652554
- Motel PJ. Captopril and alopecia: a case report and review of known cutaneous reactions in captopril use. J Am Acad Dermatol. 1990;23:124-125. doi:10.1016/s0190-9622(08)81205-4
- Leaker B, Whitworth JA. Alopecia associated with captopril treatment. Aust N Z J Med. 1984;14:866. doi:10.1111/j.1445-5994.1984.tb03797.x
- Ahmad S. Enalapril and reversible alopecia. Arch Intern Med. 1991;151:404.
- Bicket DP. Using ACE inhibitors appropriately. Am Fam Physician. 2002;66:461-468.
- Captopril. Product information. Bryant Ranch Prepack; May 2023. Accessed December 11, 2023. https://nctr-crs.fda.gov/fdalabel/services/spl/set-ids/563737c5-4d63-4957-8022-e3bc3112dfac/spl-doc?hl=captopril
- Farzam K, Jan A. Beta blockers. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK532906/
- Mason RP, Giles TD, Sowers JR. Evolving mechanisms of action of beta blockers: focus on nebivolol. J Cardiovasc Pharmacol. 2009; 54:123-128.
- Martin CM, Southwick EG, Maibach HI. Propranolol induced alopecia. Am Heart J. 1973;86:236-237. doi:10.1016/0002-8703(73)90250-0
- Graeber CW, Lapkin RA. Metoprolol and alopecia. Cutis. 1981; 28:633-634.
- Hilder RJ. Propranolol and alopecia. Cutis. 1979;24:63-64.
- Coreg. Prescribing information. Woodward Pharma Services LLC; 2023. Accessed December 11, 2023. https://www.accessdata.fda.gov/spl/data/34aa881a-3df4-460b-acad-fb9975ca3a06/34aa881a-3df4-460b-acad-fb9975ca3a06.xml
- Carey RM, Calhoun DA, Bakris GL, et al. Resistant hypertension: detection, evaluation, and management: a scientific statement from the American Heart Association. Hypertension. 2018;72:E53-E90. doi:10.1161/hyp.0000000000000084
- Campese VM. Minoxidil: a review of its pharmacological properties and therapeutic use. Drugs. 1981;22:257-278. doi:10.2165/00003495-198122040-00001
- Heymann WR. Coming full circle (almost): low dose oral minoxidil for alopecia. J Am Acad Dermatol. 2021;84:613-614. doi:10.1016/j .jaad.2020.12.053
- Yin S, Zhang B, Lin J, et al. Development of purification process for dual-function recombinant human heavy-chain ferritin by the investigation of genetic modification impact on conformation. Eng Life Sci. 2021;21:630-642. doi:10.1002/elsc.202000105
- Mysore V, Parthasaradhi A, Kharkar RD, et al. Expert consensus on the management of telogen effluvium in India. Int J Trichology. 2019;11:107-112.
- Gupta AK, Talukder M, Shemar A, et al. Low-dose oral minoxidil for alopecia: a comprehensive review [published online September 27, 2023]. Skin Appendage Disord. doi:10.1159/000531890
- Meisheri KD, Cipkus LA, Taylor CJ. Mechanism of action of minoxidil sulfate-induced vasodilation: a role for increased K+ permeability. J Pharmacol Exp Ther. 1988;245:751-760.
- Winquist RJ, Heaney LA, Wallace AA, et al. Glyburide blocks the relaxation response to BRL 34915 (cromakalim), minoxidil sulfate and diazoxide in vascular smooth muscle. J Pharmacol Exp Ther. 1989;248:149-56.
- Messenger AG, Rundegren J. Minoxidil: mechanisms of action on hair growth. Br J Dermatol. 2004;150:186-194. doi:10.1111/j .1365-2133.2004.05785.x
- Alijotas-Reig J, García GV, Velthuis PJ, et al. Inflammatory immunemediated adverse reactions induced by COVID-19 vaccines in previously injected patients with soft tissue fillers: a case series of 20 patients. J Cosmet Dermatol. 2022;21:3181-3187. doi: 10.1111/jocd.15117
- Boskabadi SJ, Ramezaninejad S, Sohrab M, et al. Diazoxideinduced hypertrichosis in a neonate with transient hyperinsulinism. Clin Med Insights Case Rep. 2023;16:11795476231151330. doi:10.1177/11795476231151330
- Burton JL, Schutt WH, Caldwell IW. Hypertrichosis due to diazoxide. Br J Dermatol. 1975;93:707-711. doi:10.1111/j.1365-2133.1975.tb05123.x
- Goldberg MR. Clinical pharmacology of pinacidil, a prototype for drugs that affect potassium channels. J Cardiovasc Pharmacol. 1988;12 suppl 2:S41-S47. doi: 10.1097/00005344-198812002-00008
- Buhl AE, Waldon DJ, Conrad SJ, et al. Potassium channel conductance: a mechanism affecting hair growth both in vitro and in vivo. J Invest Dermatol. 1992;98:315-319. doi:10.1111/1523-1747.ep12499788
- Patel P, Nessel TA, Kumar DD. Minoxidil. StatPearls [Internet]. StatPearls Publishing; 2023. Accessed December 11, 2023. https://www.ncbi.nlm.nih.gov/books/NBK482378/
- O’Keefe E, Payne RE Jr. Minoxidil: inhibition of proliferation of keratinocytes in vitro. J Invest Dermatol. 1991;97:534-536. doi:10.1111/1523-1747.ep12481560
- Murad S, Pinnell SR. Suppression of fibroblast proliferation and lysyl hydroxylase activity by minoxidil. J Biol Chem. 1987;262:11973-11978.
- Baden HP, Kubilus J. Effect of minoxidil on cultured keratinocytes. J Invest Dermatol. 1983;81:558-560. doi:10.1111/1523-1747.ep12523220
- Murad S, Walker LC, Tajima S, et al. Minimum structural requirements for minoxidil inhibition of lysyl hydroxylase in cultured fibroblasts. Arch Biochem Biophys. 1994;308:42-47. doi:10.1006/abbi.1994.1006
- Kvedar JC, Baden HP, Levine L. Selective inhibition by minoxidil of prostacyclin production by cells in culture. Biochem Pharmacol. 1988;37:867-874. doi:0.1016/0006-2952(88)90174-8
- Zhang D, LaSenna C, Shields BE. Culprits of medication-induced telogen effluvium, part 1. Cutis. 2023;112:267-271.
Practice Points
- Medications are a common culprit of telogen effluvium (TE), and medication-induced TE should be suspected in patients presenting with diffuse nonscarring alopecia who are taking systemic medication(s) such as heparin and its derivatives.
- Infection, illness, or hospitalization around the time of initiation of the suspected culprit medication may complicate identification of the inciting cause and may contribute to TE.
- Angiotensin-converting enzyme inhibitors and β-blockers are unlikely culprits of medication-induced TE, and the benefits of discontinuing a suspected culprit medication should be weighed carefully against the risks of medication cessation.
Pink Papules on the Cheek
The Diagnosis: Cutaneous Rosai-Dorfman Disease
Rosai-Dorfman disease is a rare benign non- Langerhans cell histiocytopathy that can manifest initially with lymph node involvement—classically, massive painless cervical lymphadenopathy.1 Cutaneous Rosai-Dorfman disease (CRDD) is a variant that can be associated with lymph node and internal involvement, but more than 80% of cases lack extracutaneous involvement.2,3 In cases with extracutaneous involvement, lymph node disease is most frequent.3 Cutaneous Rosai-Dorfman disease unassociated with extracutaneous disease is a benign self-limiting histiocytopathy that manifests as painless red-brown, yellow, or fleshcolored nodules, plaques, or papules that may become tender or ulcerated.4
Cutaneous Rosai-Dorfman disease represents a benign histiocytopathy of resident dendritic cell derivation.3 A characteristic immunohistochemical finding is S-100 positivity, which might suggest a Langerhans cell transdifferentiation phenotype, but other markers corroborative of a Langerhans cell phenotype—namely CD1a and langerin—will be negative. Biopsies typically show a mid to deep dermal histiocytic infiltration in a variably dense polymorphous inflammatory cell background comprised of a mixture of lymphocytes, plasma cells, and neutrophils.3 At times the extent of lymphocytic infiltration can be to a magnitude that resembles a lymphoma on histopathology. In our patient, lymphoma was excluded based on clinical presentation, as this patient lacked the typical symptoms of lymphadenopathy or B symptoms that come with B-cell lymphoma.5
The histiocytes in CRDD are characteristically large mononuclear cells exhibiting a low nuclear to cytoplasmic ratio reflective of the voluminous, nonvacuolated, watery cytoplasm. They have ill-defined cytoplasmic membranes resulting in a seemingly syncytial growth pattern. A hallmark of the histiocytes is emperipolesis characterized by intracytoplasmic localization of intact inflammatory cells including neutrophils, lymphocytes, and plasma cells.3
The differential diagnosis of CRDD includes Langerhans cell histiocytosis (LCH), indeterminate cell histiocytosis, xanthogranuloma, and reticulohistiocytoma. All of these conditions can be differentiated by their unique histopathologic and phenotypic characteristics.
Langerhans cell histiocytosis is a distinct clonal histiocytopathy that has a varied presentation ranging from cutaneous confined cases manifesting as a solitary lesion to one of disseminated cutaneous disease with the potential for multiorgan involvement. Regardless of the variant of LCH, the hallmark cell is one showing an eccentrically disposed, reniform nucleus with an open chromatin and abundant eosinophilic cytoplasm (Figure 1).6 Both LCH and CRDD stain positive for S-100. However, unlike the histiocytes in CRDD, those seen in LCH stain positive for CD1a and langerin and would not express factor XIIIA. Additionally, the neoplastic cells would not exhibit the same extent of CD68 positivity seen in CRDD.6 Treatment of LCH depends on the extent of disease, especially for the presence or absence of extracutaneous disease.7
A variant of LCH is indeterminate cell histiocytosis, which can be seen in neonates or adults. It represents a neoplastic proliferation of Langerhans cells that are devoid of Birbeck granules, reflective of an immature early phase of differentiation in the skin prior to the cells acquiring the Birbeck granule (as would be seen in neonates) or a later phase of differentiation after the mature Langerhans cell has encountered antigen and is en route to the lymph node (typically seen in adults).8 The phenotypic profile is identical to conventional LCH except the cells do not express langerin. Microscopically, the infiltrates are composed of Langerhans cells that are morphologically indistinguishable from classic LCH but without epidermotropism and exhibit a dominant localization in the dermis typically unassociated with other inflammatory cell elements (Figure 2).9
Xanthogranuloma is seen in young children (juvenile xanthogranuloma) as a solitary lesion, though a multifocal cutaneous variant and extracutaneous presentations have been described. Similar lesions can be seen in adults.10 These lesions are evolutionary in their morphology. In the inception of a juvenile xanthogranuloma, the lesions are highly cellular, and the histiocytes typically are poorly lipidized; there may be a dearth of other inflammatory cell elements. As the lesions mature, the histiocytes become lipidized, and characteristic Touton giant cells that exhibit a wreath of nuclei with peripheral lipidization may develop (Figure 3). In the later stages, there is considerable hyalinizing fibrosis, and the cells can acquire a spindled appearance. The absence of emperipolesis and the presence of notable lipidization are useful light microscopy features differentiating xanthogranuloma from CRDD.11 Treatment of xanthogranuloma can range from a conservative monitoring approach to an aggressive approach combining various antineoplastic therapies with immunosuppressive agents.12
Solitary and multicentric reticulohistiocytoma is another form of resident dendritic cell histiocytopathy that can resemble Rosai-Dorfman disease. It is a dermal histiocytic infiltrate accompanied by a polymorphous inflammatory cell infiltrate (Figure 4) and can show variable fibrosis.13 One of the hallmarks is the distinct amphophilic cytoplasms, possibly attributable to nuclear DNA released into the cytoplasm from effete nuclei.13 The solitary form is unassociated with systemic disease, whereas the multicentric variant can be a paraneoplastic syndrome in the setting of solid and hematologic malignancies.14 In addition, in the multicentric variant, similar lesions can affect any organ but there can be a proclivity to involve the hand and knee joints, leading to a crippling arthritis.15 We presented a case of CRDD, a benign resident dendritic cell histiocytopathy that can manifest as a cutaneous confined process in the skin where the clinical course is characteristically benign. It potentially can be confused with LCH, indeterminate cell histiocytosis, xanthogranuloma, and reticulohistiocytoma. For a solitary lesion, intralesional triamcinolone injection and excision are options. Multifocal cutaneous disease or CRDD with notable extracutaneous disease may require systemic treatment.16 In our patient, one intralesional triamcinolone injection was performed with notable resolution.
- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy: a newly recognized benign clinicopathological entity. Arch Pathol. 1969;87:63-70.
- Brenn T, Calonje E, Granter SR, et al. Cutaneous Rosai-Dorfman disease is a distinct clinical entity. Am J Dermatopathol. 2002;24:385.
- Ahmed A, Crowson N, Magro CM. A comprehensive assessment of cutaneous Rosai-Dorfman disease. Ann Diagn Pathol. 2019;40:166-173.
- Frater JL, Maddox JS, Obadiah JM, et al. Cutaneous Rosai-Dorfman disease: comprehensive review of cases reported in the medical literature since 1990 and presentation of an illustrative case. J Cutan Med Surg. 2006;10:281-290.
- Friedberg JW, Fisher RI. Diffuse large B-cell lymphoma. Hematol Oncol Clin North Am. 2008;22:941-952. Doi:10.1016/j.hoc.2008.07.002
- Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379:856-868.
- Board PPTE. Langerhans cell histiocytosis treatment (PDQ®). In: PDQ Cancer Information Summaries [Internet]. National Cancer Institute (US); 2009.
- Chu A, Eisinger M, Lee JS, et al. Immunoelectron microscopic identification of Langerhans cells using a new antigenic marker. J Invest Dermatol. 1982;78:177-180. doi:10.1111/1523-1747.ep12506352
- Berti E, Gianotti R, Alessi E. Unusual cutaneous histiocytosis expressing an intermediate immunophenotype between Langerhans’ cells and dermal macrophages. Arch Dermatol. 1988;124:1250-1253. doi:10.1001/archderm.1988.01670080062020
- Cypel TKS, Zuker RM. Juvenile xanthogranuloma: case report and review of the literature. Can J Plast Surg. 2008;16:175-177.
- Rodriguez J, Ackerman AB. Xanthogranuloma in adults. Arch Dermatol. 1976;112:43-44.
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2022.
- Tajirian AL, Malik MK, Robinson-Bostom L, et al. Multicentric reticulohistiocytosis. Clin Dermatol. 2006;24:486-492. doi:10.1016/j. clindermatol.2006.07.010
- Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol. 2006;30:521.
- Gold RH, Metzger AL, Mirra JM, et al. Multicentric reticulohistiocytosis (lipoid dermato-arthritis). An erosive polyarthritis with distinctive clinical, roentgenographic and pathologic features. Am J Roentgenol Radium Ther Nucl Med. 1975;124:610-624. doi:10.2214/ajr.124.4.610
- Dalia S, Sagatys E, Sokol L, et al. Rosai-Dorfman disease: tumor biology, clinical features, pathology, and treatment. Cancer Control. 2014;21:322-327. doi:10.1177/107327481402100408
The Diagnosis: Cutaneous Rosai-Dorfman Disease
Rosai-Dorfman disease is a rare benign non- Langerhans cell histiocytopathy that can manifest initially with lymph node involvement—classically, massive painless cervical lymphadenopathy.1 Cutaneous Rosai-Dorfman disease (CRDD) is a variant that can be associated with lymph node and internal involvement, but more than 80% of cases lack extracutaneous involvement.2,3 In cases with extracutaneous involvement, lymph node disease is most frequent.3 Cutaneous Rosai-Dorfman disease unassociated with extracutaneous disease is a benign self-limiting histiocytopathy that manifests as painless red-brown, yellow, or fleshcolored nodules, plaques, or papules that may become tender or ulcerated.4
Cutaneous Rosai-Dorfman disease represents a benign histiocytopathy of resident dendritic cell derivation.3 A characteristic immunohistochemical finding is S-100 positivity, which might suggest a Langerhans cell transdifferentiation phenotype, but other markers corroborative of a Langerhans cell phenotype—namely CD1a and langerin—will be negative. Biopsies typically show a mid to deep dermal histiocytic infiltration in a variably dense polymorphous inflammatory cell background comprised of a mixture of lymphocytes, plasma cells, and neutrophils.3 At times the extent of lymphocytic infiltration can be to a magnitude that resembles a lymphoma on histopathology. In our patient, lymphoma was excluded based on clinical presentation, as this patient lacked the typical symptoms of lymphadenopathy or B symptoms that come with B-cell lymphoma.5
The histiocytes in CRDD are characteristically large mononuclear cells exhibiting a low nuclear to cytoplasmic ratio reflective of the voluminous, nonvacuolated, watery cytoplasm. They have ill-defined cytoplasmic membranes resulting in a seemingly syncytial growth pattern. A hallmark of the histiocytes is emperipolesis characterized by intracytoplasmic localization of intact inflammatory cells including neutrophils, lymphocytes, and plasma cells.3
The differential diagnosis of CRDD includes Langerhans cell histiocytosis (LCH), indeterminate cell histiocytosis, xanthogranuloma, and reticulohistiocytoma. All of these conditions can be differentiated by their unique histopathologic and phenotypic characteristics.
Langerhans cell histiocytosis is a distinct clonal histiocytopathy that has a varied presentation ranging from cutaneous confined cases manifesting as a solitary lesion to one of disseminated cutaneous disease with the potential for multiorgan involvement. Regardless of the variant of LCH, the hallmark cell is one showing an eccentrically disposed, reniform nucleus with an open chromatin and abundant eosinophilic cytoplasm (Figure 1).6 Both LCH and CRDD stain positive for S-100. However, unlike the histiocytes in CRDD, those seen in LCH stain positive for CD1a and langerin and would not express factor XIIIA. Additionally, the neoplastic cells would not exhibit the same extent of CD68 positivity seen in CRDD.6 Treatment of LCH depends on the extent of disease, especially for the presence or absence of extracutaneous disease.7

A variant of LCH is indeterminate cell histiocytosis, which can be seen in neonates or adults. It represents a neoplastic proliferation of Langerhans cells that are devoid of Birbeck granules, reflective of an immature early phase of differentiation in the skin prior to the cells acquiring the Birbeck granule (as would be seen in neonates) or a later phase of differentiation after the mature Langerhans cell has encountered antigen and is en route to the lymph node (typically seen in adults).8 The phenotypic profile is identical to conventional LCH except the cells do not express langerin. Microscopically, the infiltrates are composed of Langerhans cells that are morphologically indistinguishable from classic LCH but without epidermotropism and exhibit a dominant localization in the dermis typically unassociated with other inflammatory cell elements (Figure 2).9

Xanthogranuloma is seen in young children (juvenile xanthogranuloma) as a solitary lesion, though a multifocal cutaneous variant and extracutaneous presentations have been described. Similar lesions can be seen in adults.10 These lesions are evolutionary in their morphology. In the inception of a juvenile xanthogranuloma, the lesions are highly cellular, and the histiocytes typically are poorly lipidized; there may be a dearth of other inflammatory cell elements. As the lesions mature, the histiocytes become lipidized, and characteristic Touton giant cells that exhibit a wreath of nuclei with peripheral lipidization may develop (Figure 3). In the later stages, there is considerable hyalinizing fibrosis, and the cells can acquire a spindled appearance. The absence of emperipolesis and the presence of notable lipidization are useful light microscopy features differentiating xanthogranuloma from CRDD.11 Treatment of xanthogranuloma can range from a conservative monitoring approach to an aggressive approach combining various antineoplastic therapies with immunosuppressive agents.12

Solitary and multicentric reticulohistiocytoma is another form of resident dendritic cell histiocytopathy that can resemble Rosai-Dorfman disease. It is a dermal histiocytic infiltrate accompanied by a polymorphous inflammatory cell infiltrate (Figure 4) and can show variable fibrosis.13 One of the hallmarks is the distinct amphophilic cytoplasms, possibly attributable to nuclear DNA released into the cytoplasm from effete nuclei.13 The solitary form is unassociated with systemic disease, whereas the multicentric variant can be a paraneoplastic syndrome in the setting of solid and hematologic malignancies.14 In addition, in the multicentric variant, similar lesions can affect any organ but there can be a proclivity to involve the hand and knee joints, leading to a crippling arthritis.15 We presented a case of CRDD, a benign resident dendritic cell histiocytopathy that can manifest as a cutaneous confined process in the skin where the clinical course is characteristically benign. It potentially can be confused with LCH, indeterminate cell histiocytosis, xanthogranuloma, and reticulohistiocytoma. For a solitary lesion, intralesional triamcinolone injection and excision are options. Multifocal cutaneous disease or CRDD with notable extracutaneous disease may require systemic treatment.16 In our patient, one intralesional triamcinolone injection was performed with notable resolution.

The Diagnosis: Cutaneous Rosai-Dorfman Disease
Rosai-Dorfman disease is a rare benign non- Langerhans cell histiocytopathy that can manifest initially with lymph node involvement—classically, massive painless cervical lymphadenopathy.1 Cutaneous Rosai-Dorfman disease (CRDD) is a variant that can be associated with lymph node and internal involvement, but more than 80% of cases lack extracutaneous involvement.2,3 In cases with extracutaneous involvement, lymph node disease is most frequent.3 Cutaneous Rosai-Dorfman disease unassociated with extracutaneous disease is a benign self-limiting histiocytopathy that manifests as painless red-brown, yellow, or fleshcolored nodules, plaques, or papules that may become tender or ulcerated.4
Cutaneous Rosai-Dorfman disease represents a benign histiocytopathy of resident dendritic cell derivation.3 A characteristic immunohistochemical finding is S-100 positivity, which might suggest a Langerhans cell transdifferentiation phenotype, but other markers corroborative of a Langerhans cell phenotype—namely CD1a and langerin—will be negative. Biopsies typically show a mid to deep dermal histiocytic infiltration in a variably dense polymorphous inflammatory cell background comprised of a mixture of lymphocytes, plasma cells, and neutrophils.3 At times the extent of lymphocytic infiltration can be to a magnitude that resembles a lymphoma on histopathology. In our patient, lymphoma was excluded based on clinical presentation, as this patient lacked the typical symptoms of lymphadenopathy or B symptoms that come with B-cell lymphoma.5
The histiocytes in CRDD are characteristically large mononuclear cells exhibiting a low nuclear to cytoplasmic ratio reflective of the voluminous, nonvacuolated, watery cytoplasm. They have ill-defined cytoplasmic membranes resulting in a seemingly syncytial growth pattern. A hallmark of the histiocytes is emperipolesis characterized by intracytoplasmic localization of intact inflammatory cells including neutrophils, lymphocytes, and plasma cells.3
The differential diagnosis of CRDD includes Langerhans cell histiocytosis (LCH), indeterminate cell histiocytosis, xanthogranuloma, and reticulohistiocytoma. All of these conditions can be differentiated by their unique histopathologic and phenotypic characteristics.
Langerhans cell histiocytosis is a distinct clonal histiocytopathy that has a varied presentation ranging from cutaneous confined cases manifesting as a solitary lesion to one of disseminated cutaneous disease with the potential for multiorgan involvement. Regardless of the variant of LCH, the hallmark cell is one showing an eccentrically disposed, reniform nucleus with an open chromatin and abundant eosinophilic cytoplasm (Figure 1).6 Both LCH and CRDD stain positive for S-100. However, unlike the histiocytes in CRDD, those seen in LCH stain positive for CD1a and langerin and would not express factor XIIIA. Additionally, the neoplastic cells would not exhibit the same extent of CD68 positivity seen in CRDD.6 Treatment of LCH depends on the extent of disease, especially for the presence or absence of extracutaneous disease.7

A variant of LCH is indeterminate cell histiocytosis, which can be seen in neonates or adults. It represents a neoplastic proliferation of Langerhans cells that are devoid of Birbeck granules, reflective of an immature early phase of differentiation in the skin prior to the cells acquiring the Birbeck granule (as would be seen in neonates) or a later phase of differentiation after the mature Langerhans cell has encountered antigen and is en route to the lymph node (typically seen in adults).8 The phenotypic profile is identical to conventional LCH except the cells do not express langerin. Microscopically, the infiltrates are composed of Langerhans cells that are morphologically indistinguishable from classic LCH but without epidermotropism and exhibit a dominant localization in the dermis typically unassociated with other inflammatory cell elements (Figure 2).9

Xanthogranuloma is seen in young children (juvenile xanthogranuloma) as a solitary lesion, though a multifocal cutaneous variant and extracutaneous presentations have been described. Similar lesions can be seen in adults.10 These lesions are evolutionary in their morphology. In the inception of a juvenile xanthogranuloma, the lesions are highly cellular, and the histiocytes typically are poorly lipidized; there may be a dearth of other inflammatory cell elements. As the lesions mature, the histiocytes become lipidized, and characteristic Touton giant cells that exhibit a wreath of nuclei with peripheral lipidization may develop (Figure 3). In the later stages, there is considerable hyalinizing fibrosis, and the cells can acquire a spindled appearance. The absence of emperipolesis and the presence of notable lipidization are useful light microscopy features differentiating xanthogranuloma from CRDD.11 Treatment of xanthogranuloma can range from a conservative monitoring approach to an aggressive approach combining various antineoplastic therapies with immunosuppressive agents.12

Solitary and multicentric reticulohistiocytoma is another form of resident dendritic cell histiocytopathy that can resemble Rosai-Dorfman disease. It is a dermal histiocytic infiltrate accompanied by a polymorphous inflammatory cell infiltrate (Figure 4) and can show variable fibrosis.13 One of the hallmarks is the distinct amphophilic cytoplasms, possibly attributable to nuclear DNA released into the cytoplasm from effete nuclei.13 The solitary form is unassociated with systemic disease, whereas the multicentric variant can be a paraneoplastic syndrome in the setting of solid and hematologic malignancies.14 In addition, in the multicentric variant, similar lesions can affect any organ but there can be a proclivity to involve the hand and knee joints, leading to a crippling arthritis.15 We presented a case of CRDD, a benign resident dendritic cell histiocytopathy that can manifest as a cutaneous confined process in the skin where the clinical course is characteristically benign. It potentially can be confused with LCH, indeterminate cell histiocytosis, xanthogranuloma, and reticulohistiocytoma. For a solitary lesion, intralesional triamcinolone injection and excision are options. Multifocal cutaneous disease or CRDD with notable extracutaneous disease may require systemic treatment.16 In our patient, one intralesional triamcinolone injection was performed with notable resolution.

- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy: a newly recognized benign clinicopathological entity. Arch Pathol. 1969;87:63-70.
- Brenn T, Calonje E, Granter SR, et al. Cutaneous Rosai-Dorfman disease is a distinct clinical entity. Am J Dermatopathol. 2002;24:385.
- Ahmed A, Crowson N, Magro CM. A comprehensive assessment of cutaneous Rosai-Dorfman disease. Ann Diagn Pathol. 2019;40:166-173.
- Frater JL, Maddox JS, Obadiah JM, et al. Cutaneous Rosai-Dorfman disease: comprehensive review of cases reported in the medical literature since 1990 and presentation of an illustrative case. J Cutan Med Surg. 2006;10:281-290.
- Friedberg JW, Fisher RI. Diffuse large B-cell lymphoma. Hematol Oncol Clin North Am. 2008;22:941-952. Doi:10.1016/j.hoc.2008.07.002
- Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379:856-868.
- Board PPTE. Langerhans cell histiocytosis treatment (PDQ®). In: PDQ Cancer Information Summaries [Internet]. National Cancer Institute (US); 2009.
- Chu A, Eisinger M, Lee JS, et al. Immunoelectron microscopic identification of Langerhans cells using a new antigenic marker. J Invest Dermatol. 1982;78:177-180. doi:10.1111/1523-1747.ep12506352
- Berti E, Gianotti R, Alessi E. Unusual cutaneous histiocytosis expressing an intermediate immunophenotype between Langerhans’ cells and dermal macrophages. Arch Dermatol. 1988;124:1250-1253. doi:10.1001/archderm.1988.01670080062020
- Cypel TKS, Zuker RM. Juvenile xanthogranuloma: case report and review of the literature. Can J Plast Surg. 2008;16:175-177.
- Rodriguez J, Ackerman AB. Xanthogranuloma in adults. Arch Dermatol. 1976;112:43-44.
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2022.
- Tajirian AL, Malik MK, Robinson-Bostom L, et al. Multicentric reticulohistiocytosis. Clin Dermatol. 2006;24:486-492. doi:10.1016/j. clindermatol.2006.07.010
- Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol. 2006;30:521.
- Gold RH, Metzger AL, Mirra JM, et al. Multicentric reticulohistiocytosis (lipoid dermato-arthritis). An erosive polyarthritis with distinctive clinical, roentgenographic and pathologic features. Am J Roentgenol Radium Ther Nucl Med. 1975;124:610-624. doi:10.2214/ajr.124.4.610
- Dalia S, Sagatys E, Sokol L, et al. Rosai-Dorfman disease: tumor biology, clinical features, pathology, and treatment. Cancer Control. 2014;21:322-327. doi:10.1177/107327481402100408
- Rosai J, Dorfman RF. Sinus histiocytosis with massive lymphadenopathy: a newly recognized benign clinicopathological entity. Arch Pathol. 1969;87:63-70.
- Brenn T, Calonje E, Granter SR, et al. Cutaneous Rosai-Dorfman disease is a distinct clinical entity. Am J Dermatopathol. 2002;24:385.
- Ahmed A, Crowson N, Magro CM. A comprehensive assessment of cutaneous Rosai-Dorfman disease. Ann Diagn Pathol. 2019;40:166-173.
- Frater JL, Maddox JS, Obadiah JM, et al. Cutaneous Rosai-Dorfman disease: comprehensive review of cases reported in the medical literature since 1990 and presentation of an illustrative case. J Cutan Med Surg. 2006;10:281-290.
- Friedberg JW, Fisher RI. Diffuse large B-cell lymphoma. Hematol Oncol Clin North Am. 2008;22:941-952. Doi:10.1016/j.hoc.2008.07.002
- Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379:856-868.
- Board PPTE. Langerhans cell histiocytosis treatment (PDQ®). In: PDQ Cancer Information Summaries [Internet]. National Cancer Institute (US); 2009.
- Chu A, Eisinger M, Lee JS, et al. Immunoelectron microscopic identification of Langerhans cells using a new antigenic marker. J Invest Dermatol. 1982;78:177-180. doi:10.1111/1523-1747.ep12506352
- Berti E, Gianotti R, Alessi E. Unusual cutaneous histiocytosis expressing an intermediate immunophenotype between Langerhans’ cells and dermal macrophages. Arch Dermatol. 1988;124:1250-1253. doi:10.1001/archderm.1988.01670080062020
- Cypel TKS, Zuker RM. Juvenile xanthogranuloma: case report and review of the literature. Can J Plast Surg. 2008;16:175-177.
- Rodriguez J, Ackerman AB. Xanthogranuloma in adults. Arch Dermatol. 1976;112:43-44.
- Collie JS, Harper CD, Fillman EP. Juvenile xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2022.
- Tajirian AL, Malik MK, Robinson-Bostom L, et al. Multicentric reticulohistiocytosis. Clin Dermatol. 2006;24:486-492. doi:10.1016/j. clindermatol.2006.07.010
- Miettinen M, Fetsch JF. Reticulohistiocytoma (solitary epithelioid histiocytoma): a clinicopathologic and immunohistochemical study of 44 cases. Am J Surg Pathol. 2006;30:521.
- Gold RH, Metzger AL, Mirra JM, et al. Multicentric reticulohistiocytosis (lipoid dermato-arthritis). An erosive polyarthritis with distinctive clinical, roentgenographic and pathologic features. Am J Roentgenol Radium Ther Nucl Med. 1975;124:610-624. doi:10.2214/ajr.124.4.610
- Dalia S, Sagatys E, Sokol L, et al. Rosai-Dorfman disease: tumor biology, clinical features, pathology, and treatment. Cancer Control. 2014;21:322-327. doi:10.1177/107327481402100408
A 31-year-old woman presented with a slow-growing, tender, pruritic lesion on the right cheek of 4 to 5 months’ duration. She had been applying petroleum jelly and hydrocortisone cream 2.5% without any improvement. Physical examination revealed a 1×5-mm, pearly pink, erythematous, crusted papule with arborizing vessels surrounded by scattered pink papules with white dots within. No cervical lymphadenopathy was appreciated on physical examination, and the patient denied any other systemic symptoms. Shave and punch biopsies of the lesion were performed; stains for microorganisms were negative. The biopsy showed a dense reticular mixed inflammatory cell infiltrate comprised of a mixture of histiocytes (top), lymphocytes, neutrophils, and plasma cells that assumed a diffuse growth pattern within the dermis. The histiocytes exhibited abundant watery cytoplasms with ill-defined cytoplasmic membranes; intact leukocytes were found within the cytoplasms. The histiocytes demonstrated a unique phenotype characterized by S-100 (bottom) and CD68 positivity.