User login
Partnering with dietitians can bridge gaps in IBD care
Working with a registered dietitian (RD) can help ensure that changing the way patients with inflammatory bowel disease (IBD) eat won’t deprive them of the nutrients they need.
Depending on the location and resources of a medical practice, calling in a dietitian may seem like a luxury. But making those connections can be more accessible during the COVID-19 pandemic as more dietitians are working virtually.
Kelly Issokson, MS, RD, clinical nutrition coordinator for IBD at Cedars Sinai Medical Center in Los Angeles, suggested two websites that allow users to search for RDs by ZIP code or by those working virtually: the International Foundation for Gastrointestinal Disorders and eatright.org, the website for the professional body for the dietetics community, which also has a searchable database.
Ashwin N. Ananthakrishnan, MD, MPH, director of the Crohn’s and colitis center at Massachusetts General Hospital in Boston, said it’s key for gastroenterologists to communicate what exactly they want the dietitian to address and not merely refer the patient.
The provider should know what therapies exist and then have the dietitian walk the patient through the plan, he said.
Mark Mattar, MD, with MedStar Georgetown University Hospital in Washington, said that, in addition to connecting patients with dietitians, “I always refer my patient to the Crohn’s and Colitis Foundation for the most recently updated patient education materials on nutrition.”
Panelists at the Advances in Inflammatory Bowel Diseases 2020 annual meeting on Wednesday weighed in on dietary considerations for two patient scenarios posed by Maria Abreu, MD, director of the Crohn’s & Colitis Center at the University of Miami.
The first scenario involved a 54-year-old man with long-standing fibrostenotic Crohn’s disease, recently hospitalized for obstruction and discharged with a prescription for prednisone 40 mg daily. The patient had been on infliximab (Remicade), and now is taking now adalimumab (Humira) weekly. He will undergo surgery to remove an ileal stricture. Dr. Abreu asked what dietary changes the panelists would make to ensure adequate nutrition prior to surgery and prevent another obstruction.
Dr. Ananthakrishnan said he would check vitamin B₁₂, vitamin D, iron, and albumin levels to see if any micronutrients need to be replaced.
He said that, although he thinks low-fiber diets are used too often as the default for Crohn’s and ulcerative colitis, in this case he would recommend low fiber and urge the patient to avoid raw fruits, vegetables, nuts, and seeds.
The patient can remove the skins and still have shakes and smoothies to get the benefits of fiber-containing foods without the fiber component, he said.
Discussing a pediatric version of that scenario, Andrew Grossman, MD, a pediatric gastroenterologist at the Children’s Hospital of Philadelphia, said he would turn to enteral nutrition therapy.
“We would strongly encourage using a formula to try to improve nutritional status, which we know can improve surgical outcomes,” he said.
The second case was a 15-year-old girl with growth stunting. She was diagnosed at age 10 with Crohn’s disease, currently has moderate disease, and continues to have five to seven liquid bowel movements daily, along with abdominal pain after meals. She is starting adalimumab induction.
Dr. Grossman said, “first, I would not be managing this alone. I would be managing this with a dietitian and working together to improve outcomes. We need to consider aggressive therapy, and to me that would include consideration of biological therapy but also possible dietary therapy – the Crohn’s Disease Exclusion Diet or enteral nutrition therapy as possibilities.”
He pointed out that in pediatrics there must be consideration both for what the parent wants the child to do and what the child is willing to do.
“My primary focus would be on improving caloric intake, working with the dietitian to avoid foods that bother the most,” he said.
Dr. Issokson said she would recommend either exclusive enteral nutrition or a specific carbohydrate diet (SCD) for the teen.
“We see [SCD] doesn’t impair growth in our patients as long as they are being followed by a dietitian, and we’re making sure they are getting adequate nutrient intake,” she said.
Dr. Abreu said in an interview that “diet is important in patients with IBD; it is a complement to the therapies that we use and a potential opportunity to solidify a long-lived remission.”
“Although studies of diet are only now being done,” she said, “we already have some good foundational ideas about diet and its role in reducing inflammation and reducing symptoms.” And she added that treating gastroenterologists should certainly avoid telling patients that “diet does not matter.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Ananthakrishnan, Dr. Grossman, and Dr. Issokson have disclosed no relevant financial relationships.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
A version of this article originally appeared on Medscape.com.
Working with a registered dietitian (RD) can help ensure that changing the way patients with inflammatory bowel disease (IBD) eat won’t deprive them of the nutrients they need.
Depending on the location and resources of a medical practice, calling in a dietitian may seem like a luxury. But making those connections can be more accessible during the COVID-19 pandemic as more dietitians are working virtually.
Kelly Issokson, MS, RD, clinical nutrition coordinator for IBD at Cedars Sinai Medical Center in Los Angeles, suggested two websites that allow users to search for RDs by ZIP code or by those working virtually: the International Foundation for Gastrointestinal Disorders and eatright.org, the website for the professional body for the dietetics community, which also has a searchable database.
Ashwin N. Ananthakrishnan, MD, MPH, director of the Crohn’s and colitis center at Massachusetts General Hospital in Boston, said it’s key for gastroenterologists to communicate what exactly they want the dietitian to address and not merely refer the patient.
The provider should know what therapies exist and then have the dietitian walk the patient through the plan, he said.
Mark Mattar, MD, with MedStar Georgetown University Hospital in Washington, said that, in addition to connecting patients with dietitians, “I always refer my patient to the Crohn’s and Colitis Foundation for the most recently updated patient education materials on nutrition.”
Panelists at the Advances in Inflammatory Bowel Diseases 2020 annual meeting on Wednesday weighed in on dietary considerations for two patient scenarios posed by Maria Abreu, MD, director of the Crohn’s & Colitis Center at the University of Miami.
The first scenario involved a 54-year-old man with long-standing fibrostenotic Crohn’s disease, recently hospitalized for obstruction and discharged with a prescription for prednisone 40 mg daily. The patient had been on infliximab (Remicade), and now is taking now adalimumab (Humira) weekly. He will undergo surgery to remove an ileal stricture. Dr. Abreu asked what dietary changes the panelists would make to ensure adequate nutrition prior to surgery and prevent another obstruction.
Dr. Ananthakrishnan said he would check vitamin B₁₂, vitamin D, iron, and albumin levels to see if any micronutrients need to be replaced.
He said that, although he thinks low-fiber diets are used too often as the default for Crohn’s and ulcerative colitis, in this case he would recommend low fiber and urge the patient to avoid raw fruits, vegetables, nuts, and seeds.
The patient can remove the skins and still have shakes and smoothies to get the benefits of fiber-containing foods without the fiber component, he said.
Discussing a pediatric version of that scenario, Andrew Grossman, MD, a pediatric gastroenterologist at the Children’s Hospital of Philadelphia, said he would turn to enteral nutrition therapy.
“We would strongly encourage using a formula to try to improve nutritional status, which we know can improve surgical outcomes,” he said.
The second case was a 15-year-old girl with growth stunting. She was diagnosed at age 10 with Crohn’s disease, currently has moderate disease, and continues to have five to seven liquid bowel movements daily, along with abdominal pain after meals. She is starting adalimumab induction.
Dr. Grossman said, “first, I would not be managing this alone. I would be managing this with a dietitian and working together to improve outcomes. We need to consider aggressive therapy, and to me that would include consideration of biological therapy but also possible dietary therapy – the Crohn’s Disease Exclusion Diet or enteral nutrition therapy as possibilities.”
He pointed out that in pediatrics there must be consideration both for what the parent wants the child to do and what the child is willing to do.
“My primary focus would be on improving caloric intake, working with the dietitian to avoid foods that bother the most,” he said.
Dr. Issokson said she would recommend either exclusive enteral nutrition or a specific carbohydrate diet (SCD) for the teen.
“We see [SCD] doesn’t impair growth in our patients as long as they are being followed by a dietitian, and we’re making sure they are getting adequate nutrient intake,” she said.
Dr. Abreu said in an interview that “diet is important in patients with IBD; it is a complement to the therapies that we use and a potential opportunity to solidify a long-lived remission.”
“Although studies of diet are only now being done,” she said, “we already have some good foundational ideas about diet and its role in reducing inflammation and reducing symptoms.” And she added that treating gastroenterologists should certainly avoid telling patients that “diet does not matter.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Ananthakrishnan, Dr. Grossman, and Dr. Issokson have disclosed no relevant financial relationships.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
A version of this article originally appeared on Medscape.com.
Working with a registered dietitian (RD) can help ensure that changing the way patients with inflammatory bowel disease (IBD) eat won’t deprive them of the nutrients they need.
Depending on the location and resources of a medical practice, calling in a dietitian may seem like a luxury. But making those connections can be more accessible during the COVID-19 pandemic as more dietitians are working virtually.
Kelly Issokson, MS, RD, clinical nutrition coordinator for IBD at Cedars Sinai Medical Center in Los Angeles, suggested two websites that allow users to search for RDs by ZIP code or by those working virtually: the International Foundation for Gastrointestinal Disorders and eatright.org, the website for the professional body for the dietetics community, which also has a searchable database.
Ashwin N. Ananthakrishnan, MD, MPH, director of the Crohn’s and colitis center at Massachusetts General Hospital in Boston, said it’s key for gastroenterologists to communicate what exactly they want the dietitian to address and not merely refer the patient.
The provider should know what therapies exist and then have the dietitian walk the patient through the plan, he said.
Mark Mattar, MD, with MedStar Georgetown University Hospital in Washington, said that, in addition to connecting patients with dietitians, “I always refer my patient to the Crohn’s and Colitis Foundation for the most recently updated patient education materials on nutrition.”
Panelists at the Advances in Inflammatory Bowel Diseases 2020 annual meeting on Wednesday weighed in on dietary considerations for two patient scenarios posed by Maria Abreu, MD, director of the Crohn’s & Colitis Center at the University of Miami.
The first scenario involved a 54-year-old man with long-standing fibrostenotic Crohn’s disease, recently hospitalized for obstruction and discharged with a prescription for prednisone 40 mg daily. The patient had been on infliximab (Remicade), and now is taking now adalimumab (Humira) weekly. He will undergo surgery to remove an ileal stricture. Dr. Abreu asked what dietary changes the panelists would make to ensure adequate nutrition prior to surgery and prevent another obstruction.
Dr. Ananthakrishnan said he would check vitamin B₁₂, vitamin D, iron, and albumin levels to see if any micronutrients need to be replaced.
He said that, although he thinks low-fiber diets are used too often as the default for Crohn’s and ulcerative colitis, in this case he would recommend low fiber and urge the patient to avoid raw fruits, vegetables, nuts, and seeds.
The patient can remove the skins and still have shakes and smoothies to get the benefits of fiber-containing foods without the fiber component, he said.
Discussing a pediatric version of that scenario, Andrew Grossman, MD, a pediatric gastroenterologist at the Children’s Hospital of Philadelphia, said he would turn to enteral nutrition therapy.
“We would strongly encourage using a formula to try to improve nutritional status, which we know can improve surgical outcomes,” he said.
The second case was a 15-year-old girl with growth stunting. She was diagnosed at age 10 with Crohn’s disease, currently has moderate disease, and continues to have five to seven liquid bowel movements daily, along with abdominal pain after meals. She is starting adalimumab induction.
Dr. Grossman said, “first, I would not be managing this alone. I would be managing this with a dietitian and working together to improve outcomes. We need to consider aggressive therapy, and to me that would include consideration of biological therapy but also possible dietary therapy – the Crohn’s Disease Exclusion Diet or enteral nutrition therapy as possibilities.”
He pointed out that in pediatrics there must be consideration both for what the parent wants the child to do and what the child is willing to do.
“My primary focus would be on improving caloric intake, working with the dietitian to avoid foods that bother the most,” he said.
Dr. Issokson said she would recommend either exclusive enteral nutrition or a specific carbohydrate diet (SCD) for the teen.
“We see [SCD] doesn’t impair growth in our patients as long as they are being followed by a dietitian, and we’re making sure they are getting adequate nutrient intake,” she said.
Dr. Abreu said in an interview that “diet is important in patients with IBD; it is a complement to the therapies that we use and a potential opportunity to solidify a long-lived remission.”
“Although studies of diet are only now being done,” she said, “we already have some good foundational ideas about diet and its role in reducing inflammation and reducing symptoms.” And she added that treating gastroenterologists should certainly avoid telling patients that “diet does not matter.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Ananthakrishnan, Dr. Grossman, and Dr. Issokson have disclosed no relevant financial relationships.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
A version of this article originally appeared on Medscape.com.
Partnering with dietitians can bridge gaps in IBD care
Working with a registered dietitian (RD) can help ensure that changing the way patients with inflammatory bowel disease (IBD) eat won’t deprive them of the nutrients they need.
Depending on the location and resources of a medical practice, calling in a dietitian may seem like a luxury. But making those connections can be more accessible during the COVID-19 pandemic as more dietitians are working virtually.
Kelly Issokson, MS, RD, clinical nutrition coordinator for IBD at Cedars Sinai Medical Center in Los Angeles, suggested two websites that allow users to search for RDs by ZIP code or by those working virtually: the International Foundation for Gastrointestinal Disorders and eatright.org, the website for the professional body for the dietetics community, which also has a searchable database.
Ashwin N. Ananthakrishnan, MD, MPH, director of the Crohn’s and colitis center at Massachusetts General Hospital in Boston, said it’s key for gastroenterologists to communicate what exactly they want the dietitian to address and not merely refer the patient.
The provider should know what therapies exist and then have the dietitian walk the patient through the plan, he said.
Mark Mattar, MD, with MedStar Georgetown University Hospital in Washington, said that, in addition to connecting patients with dietitians, “I always refer my patient to the Crohn’s and Colitis Foundation for the most recently updated patient education materials on nutrition.”
Panelists at the Advances in Inflammatory Bowel Diseases 2020 annual meeting on Wednesday weighed in on dietary considerations for two patient scenarios posed by Maria Abreu, MD, director of the Crohn’s & Colitis Center at the University of Miami.
The first scenario involved a 54-year-old man with long-standing fibrostenotic Crohn’s disease, recently hospitalized for obstruction and discharged with a prescription for prednisone 40 mg daily. The patient had been on infliximab (Remicade), and now is taking now adalimumab (Humira) weekly. He will undergo surgery to remove an ileal stricture. Dr. Abreu asked what dietary changes the panelists would make to ensure adequate nutrition prior to surgery and prevent another obstruction.
Dr. Ananthakrishnan said he would check vitamin B₁₂, vitamin D, iron, and albumin levels to see if any micronutrients need to be replaced.
He said that, although he thinks low-fiber diets are used too often as the default for Crohn’s and ulcerative colitis, in this case he would recommend low fiber and urge the patient to avoid raw fruits, vegetables, nuts, and seeds.
The patient can remove the skins and still have shakes and smoothies to get the benefits of fiber-containing foods without the fiber component, he said.
Discussing a pediatric version of that scenario, Andrew Grossman, MD, a pediatric gastroenterologist at the Children’s Hospital of Philadelphia, said he would turn to enteral nutrition therapy.
“We would strongly encourage using a formula to try to improve nutritional status, which we know can improve surgical outcomes,” he said.
The second case was a 15-year-old girl with growth stunting. She was diagnosed at age 10 with Crohn’s disease, currently has moderate disease, and continues to have five to seven liquid bowel movements daily, along with abdominal pain after meals. She is starting adalimumab induction.
Dr. Grossman said, “first, I would not be managing this alone. I would be managing this with a dietitian and working together to improve outcomes. We need to consider aggressive therapy, and to me that would include consideration of biological therapy but also possible dietary therapy – the Crohn’s Disease Exclusion Diet or enteral nutrition therapy as possibilities.”
He pointed out that in pediatrics there must be consideration both for what the parent wants the child to do and what the child is willing to do.
“My primary focus would be on improving caloric intake, working with the dietitian to avoid foods that bother the most,” he said.
Dr. Issokson said she would recommend either exclusive enteral nutrition or a specific carbohydrate diet (SCD) for the teen.
“We see [SCD] doesn’t impair growth in our patients as long as they are being followed by a dietitian, and we’re making sure they are getting adequate nutrient intake,” she said.
Dr. Abreu said in an interview that “diet is important in patients with IBD; it is a complement to the therapies that we use and a potential opportunity to solidify a long-lived remission.”
“Although studies of diet are only now being done,” she said, “we already have some good foundational ideas about diet and its role in reducing inflammation and reducing symptoms.” And she added that treating gastroenterologists should certainly avoid telling patients that “diet does not matter.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Ananthakrishnan, Dr. Grossman, and Dr. Issokson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Working with a registered dietitian (RD) can help ensure that changing the way patients with inflammatory bowel disease (IBD) eat won’t deprive them of the nutrients they need.
Depending on the location and resources of a medical practice, calling in a dietitian may seem like a luxury. But making those connections can be more accessible during the COVID-19 pandemic as more dietitians are working virtually.
Kelly Issokson, MS, RD, clinical nutrition coordinator for IBD at Cedars Sinai Medical Center in Los Angeles, suggested two websites that allow users to search for RDs by ZIP code or by those working virtually: the International Foundation for Gastrointestinal Disorders and eatright.org, the website for the professional body for the dietetics community, which also has a searchable database.
Ashwin N. Ananthakrishnan, MD, MPH, director of the Crohn’s and colitis center at Massachusetts General Hospital in Boston, said it’s key for gastroenterologists to communicate what exactly they want the dietitian to address and not merely refer the patient.
The provider should know what therapies exist and then have the dietitian walk the patient through the plan, he said.
Mark Mattar, MD, with MedStar Georgetown University Hospital in Washington, said that, in addition to connecting patients with dietitians, “I always refer my patient to the Crohn’s and Colitis Foundation for the most recently updated patient education materials on nutrition.”
Panelists at the Advances in Inflammatory Bowel Diseases 2020 annual meeting on Wednesday weighed in on dietary considerations for two patient scenarios posed by Maria Abreu, MD, director of the Crohn’s & Colitis Center at the University of Miami.
The first scenario involved a 54-year-old man with long-standing fibrostenotic Crohn’s disease, recently hospitalized for obstruction and discharged with a prescription for prednisone 40 mg daily. The patient had been on infliximab (Remicade), and now is taking now adalimumab (Humira) weekly. He will undergo surgery to remove an ileal stricture. Dr. Abreu asked what dietary changes the panelists would make to ensure adequate nutrition prior to surgery and prevent another obstruction.
Dr. Ananthakrishnan said he would check vitamin B₁₂, vitamin D, iron, and albumin levels to see if any micronutrients need to be replaced.
He said that, although he thinks low-fiber diets are used too often as the default for Crohn’s and ulcerative colitis, in this case he would recommend low fiber and urge the patient to avoid raw fruits, vegetables, nuts, and seeds.
The patient can remove the skins and still have shakes and smoothies to get the benefits of fiber-containing foods without the fiber component, he said.
Discussing a pediatric version of that scenario, Andrew Grossman, MD, a pediatric gastroenterologist at the Children’s Hospital of Philadelphia, said he would turn to enteral nutrition therapy.
“We would strongly encourage using a formula to try to improve nutritional status, which we know can improve surgical outcomes,” he said.
The second case was a 15-year-old girl with growth stunting. She was diagnosed at age 10 with Crohn’s disease, currently has moderate disease, and continues to have five to seven liquid bowel movements daily, along with abdominal pain after meals. She is starting adalimumab induction.
Dr. Grossman said, “first, I would not be managing this alone. I would be managing this with a dietitian and working together to improve outcomes. We need to consider aggressive therapy, and to me that would include consideration of biological therapy but also possible dietary therapy – the Crohn’s Disease Exclusion Diet or enteral nutrition therapy as possibilities.”
He pointed out that in pediatrics there must be consideration both for what the parent wants the child to do and what the child is willing to do.
“My primary focus would be on improving caloric intake, working with the dietitian to avoid foods that bother the most,” he said.
Dr. Issokson said she would recommend either exclusive enteral nutrition or a specific carbohydrate diet (SCD) for the teen.
“We see [SCD] doesn’t impair growth in our patients as long as they are being followed by a dietitian, and we’re making sure they are getting adequate nutrient intake,” she said.
Dr. Abreu said in an interview that “diet is important in patients with IBD; it is a complement to the therapies that we use and a potential opportunity to solidify a long-lived remission.”
“Although studies of diet are only now being done,” she said, “we already have some good foundational ideas about diet and its role in reducing inflammation and reducing symptoms.” And she added that treating gastroenterologists should certainly avoid telling patients that “diet does not matter.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Ananthakrishnan, Dr. Grossman, and Dr. Issokson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Working with a registered dietitian (RD) can help ensure that changing the way patients with inflammatory bowel disease (IBD) eat won’t deprive them of the nutrients they need.
Depending on the location and resources of a medical practice, calling in a dietitian may seem like a luxury. But making those connections can be more accessible during the COVID-19 pandemic as more dietitians are working virtually.
Kelly Issokson, MS, RD, clinical nutrition coordinator for IBD at Cedars Sinai Medical Center in Los Angeles, suggested two websites that allow users to search for RDs by ZIP code or by those working virtually: the International Foundation for Gastrointestinal Disorders and eatright.org, the website for the professional body for the dietetics community, which also has a searchable database.
Ashwin N. Ananthakrishnan, MD, MPH, director of the Crohn’s and colitis center at Massachusetts General Hospital in Boston, said it’s key for gastroenterologists to communicate what exactly they want the dietitian to address and not merely refer the patient.
The provider should know what therapies exist and then have the dietitian walk the patient through the plan, he said.
Mark Mattar, MD, with MedStar Georgetown University Hospital in Washington, said that, in addition to connecting patients with dietitians, “I always refer my patient to the Crohn’s and Colitis Foundation for the most recently updated patient education materials on nutrition.”
Panelists at the Advances in Inflammatory Bowel Diseases 2020 annual meeting on Wednesday weighed in on dietary considerations for two patient scenarios posed by Maria Abreu, MD, director of the Crohn’s & Colitis Center at the University of Miami.
The first scenario involved a 54-year-old man with long-standing fibrostenotic Crohn’s disease, recently hospitalized for obstruction and discharged with a prescription for prednisone 40 mg daily. The patient had been on infliximab (Remicade), and now is taking now adalimumab (Humira) weekly. He will undergo surgery to remove an ileal stricture. Dr. Abreu asked what dietary changes the panelists would make to ensure adequate nutrition prior to surgery and prevent another obstruction.
Dr. Ananthakrishnan said he would check vitamin B₁₂, vitamin D, iron, and albumin levels to see if any micronutrients need to be replaced.
He said that, although he thinks low-fiber diets are used too often as the default for Crohn’s and ulcerative colitis, in this case he would recommend low fiber and urge the patient to avoid raw fruits, vegetables, nuts, and seeds.
The patient can remove the skins and still have shakes and smoothies to get the benefits of fiber-containing foods without the fiber component, he said.
Discussing a pediatric version of that scenario, Andrew Grossman, MD, a pediatric gastroenterologist at the Children’s Hospital of Philadelphia, said he would turn to enteral nutrition therapy.
“We would strongly encourage using a formula to try to improve nutritional status, which we know can improve surgical outcomes,” he said.
The second case was a 15-year-old girl with growth stunting. She was diagnosed at age 10 with Crohn’s disease, currently has moderate disease, and continues to have five to seven liquid bowel movements daily, along with abdominal pain after meals. She is starting adalimumab induction.
Dr. Grossman said, “first, I would not be managing this alone. I would be managing this with a dietitian and working together to improve outcomes. We need to consider aggressive therapy, and to me that would include consideration of biological therapy but also possible dietary therapy – the Crohn’s Disease Exclusion Diet or enteral nutrition therapy as possibilities.”
He pointed out that in pediatrics there must be consideration both for what the parent wants the child to do and what the child is willing to do.
“My primary focus would be on improving caloric intake, working with the dietitian to avoid foods that bother the most,” he said.
Dr. Issokson said she would recommend either exclusive enteral nutrition or a specific carbohydrate diet (SCD) for the teen.
“We see [SCD] doesn’t impair growth in our patients as long as they are being followed by a dietitian, and we’re making sure they are getting adequate nutrient intake,” she said.
Dr. Abreu said in an interview that “diet is important in patients with IBD; it is a complement to the therapies that we use and a potential opportunity to solidify a long-lived remission.”
“Although studies of diet are only now being done,” she said, “we already have some good foundational ideas about diet and its role in reducing inflammation and reducing symptoms.” And she added that treating gastroenterologists should certainly avoid telling patients that “diet does not matter.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Ananthakrishnan, Dr. Grossman, and Dr. Issokson have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
COVID-19 and patient safety in the medical office
Editor’s note: This article has been provided by The Doctors Company, the exclusively endorsed medical malpractice carrier for the Society of Hospital Medicine.
As the pandemic hits its third nationwide surge, families are gathering for the holidays, and medical practices are preparing for a potential increase in cases. Medical offices in states that were not strongly affected by the first and second waves of the virus may now be facing an influx of COVID-19 patients. Therefore, medical offices must remain very attentive to the widespread outbreak of COVID-19, continuing to proactively take steps to safely manage patients while protecting clinical staff.
Here are tips and resources for this season of the pandemic:
- Documentation: Maintain administrative records of how you have adapted to the evolving crisis, including the challenges you faced. For details, see Keep a COVID-19 Diary: Document Now in Case of Future Lawsuits.
- Legislation and Guidance: Reference the CDC; your state medical board; professional societies; and federal, state, and local authorities daily for public health guidance and new legislation, as this continues to be a fluid situation.
- Screening Criteria: Follow the CDC’s patient assessment protocol for early disease detection for patients presenting to your practice. Patients should be screened using these guidelines: Overview of Testing for SARS-CoV-2 (COVID-19). Essential visitors to your facility should also be assessed for symptoms of coronavirus and contact exposure and redirected to remain outside if suspect.
- Accepting Patients: Do not turn patients away simply because a patient calls with acute respiratory symptoms. Refusing assessment/care may lead to concerns of patient abandonment.
- Designated Triage Location: Check with your local public health authorities for locations designated to triage suspected patients, so exposure is limited in general medical offices.
- Telehealth Triage: Licensed staff should be trained in triage protocol to determine which patients can be managed safely at home. See Healthcare Facilities: Managing Operations During the COVID-19 Pandemic. The Doctors Company offers resources on telemedicine in our COVID-19 Telehealth Resource Center.
- Patient Testing: When there is a reasonable presumption that a patient may have been exposed to COVID-19, contact the local or state health department to coordinate testing using available community resources. See the CDC’s Testing for COVID-19 , the COVID-19 Testing Overview, and the Clinician Call Center.
- Elective Services: Check with regional governmental and health authorities on the provision of nonessential and elective health care visits and group-related activities. Many states continue or have reinstated restrictions on the provision of nonurgent, elective surgeries and procedures.
- Patient Precautions: Educational resources, including posters for use in the medical office, are available from the WHO and for health care workers from the CDC (Contact Precautions, Droplet Precautions, and Airborne Precautions). Reference the CDC’s and Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19) for patient management guidance.
- Provider/Staff Precautions: Follow Standard Precautions and Transmission-Based Precautions, including gloves, gowns, protective eyewear, and NIOSH-certified N95 respirators that have been properly fit-tested. If there is a shortage of N95 respirators in your facility, access current CDC respirator recommendations and review Optimizing Personal Protective Equipment (PPE) Supplies.
- Limit Exposure: Limit staff exposure to suspected patients, with the exam room door kept closed. Ideally, the designated exam room should be at the back of the office, far away from other staff and patients.
- Surface Disinfection: Once the patient exits the room, conduct surface disinfection while staff continues to wear PPE.
- Patient Education: Provide up-to-date, factual information on the virus to suspected COVID-19–positive patients and their close contacts.
- Provider/Staff Exposure: Screen health care personnel daily for symptoms/contacts relevant to COVID-19. Any unprotected occupational exposure by staff members should be assessed and monitored. See Interim U.S. Guidance for Risk Assessment and Work Restrictions for Healthcare Personnel with Potential Exposure to COVID-19. Should providers and/or staff test positive within your facility, conduct and document a risk assessment identifying contacts, type of interaction, and PPE in use, then contact local health authorities for additional instruction. The CDC provides guidance here under the section “Infection Control.” The health department may assist with patient notification if determined to be necessary. For return-to-work guidance, review the Criteria for Return to Work for Healthcare Personnel with SARS-CoV-2 Infection (Interim Guidance).
- Staff Training: Provide and document additional staff training as protocols change. Maintain training records in administrative files.
Ms. Hill is senior patient safety risk manager at The Doctors Company. The guidelines suggested here are not rules, do not constitute legal advice, and do not ensure a successful outcome. The ultimate decision regarding the appropriateness of any treatment must be made by each health care provider considering the circumstances of the individual situation and in accordance with the laws of the jurisdiction in which the care is rendered.
Editor’s note: This article has been provided by The Doctors Company, the exclusively endorsed medical malpractice carrier for the Society of Hospital Medicine.
As the pandemic hits its third nationwide surge, families are gathering for the holidays, and medical practices are preparing for a potential increase in cases. Medical offices in states that were not strongly affected by the first and second waves of the virus may now be facing an influx of COVID-19 patients. Therefore, medical offices must remain very attentive to the widespread outbreak of COVID-19, continuing to proactively take steps to safely manage patients while protecting clinical staff.
Here are tips and resources for this season of the pandemic:
- Documentation: Maintain administrative records of how you have adapted to the evolving crisis, including the challenges you faced. For details, see Keep a COVID-19 Diary: Document Now in Case of Future Lawsuits.
- Legislation and Guidance: Reference the CDC; your state medical board; professional societies; and federal, state, and local authorities daily for public health guidance and new legislation, as this continues to be a fluid situation.
- Screening Criteria: Follow the CDC’s patient assessment protocol for early disease detection for patients presenting to your practice. Patients should be screened using these guidelines: Overview of Testing for SARS-CoV-2 (COVID-19). Essential visitors to your facility should also be assessed for symptoms of coronavirus and contact exposure and redirected to remain outside if suspect.
- Accepting Patients: Do not turn patients away simply because a patient calls with acute respiratory symptoms. Refusing assessment/care may lead to concerns of patient abandonment.
- Designated Triage Location: Check with your local public health authorities for locations designated to triage suspected patients, so exposure is limited in general medical offices.
- Telehealth Triage: Licensed staff should be trained in triage protocol to determine which patients can be managed safely at home. See Healthcare Facilities: Managing Operations During the COVID-19 Pandemic. The Doctors Company offers resources on telemedicine in our COVID-19 Telehealth Resource Center.
- Patient Testing: When there is a reasonable presumption that a patient may have been exposed to COVID-19, contact the local or state health department to coordinate testing using available community resources. See the CDC’s Testing for COVID-19 , the COVID-19 Testing Overview, and the Clinician Call Center.
- Elective Services: Check with regional governmental and health authorities on the provision of nonessential and elective health care visits and group-related activities. Many states continue or have reinstated restrictions on the provision of nonurgent, elective surgeries and procedures.
- Patient Precautions: Educational resources, including posters for use in the medical office, are available from the WHO and for health care workers from the CDC (Contact Precautions, Droplet Precautions, and Airborne Precautions). Reference the CDC’s and Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19) for patient management guidance.
- Provider/Staff Precautions: Follow Standard Precautions and Transmission-Based Precautions, including gloves, gowns, protective eyewear, and NIOSH-certified N95 respirators that have been properly fit-tested. If there is a shortage of N95 respirators in your facility, access current CDC respirator recommendations and review Optimizing Personal Protective Equipment (PPE) Supplies.
- Limit Exposure: Limit staff exposure to suspected patients, with the exam room door kept closed. Ideally, the designated exam room should be at the back of the office, far away from other staff and patients.
- Surface Disinfection: Once the patient exits the room, conduct surface disinfection while staff continues to wear PPE.
- Patient Education: Provide up-to-date, factual information on the virus to suspected COVID-19–positive patients and their close contacts.
- Provider/Staff Exposure: Screen health care personnel daily for symptoms/contacts relevant to COVID-19. Any unprotected occupational exposure by staff members should be assessed and monitored. See Interim U.S. Guidance for Risk Assessment and Work Restrictions for Healthcare Personnel with Potential Exposure to COVID-19. Should providers and/or staff test positive within your facility, conduct and document a risk assessment identifying contacts, type of interaction, and PPE in use, then contact local health authorities for additional instruction. The CDC provides guidance here under the section “Infection Control.” The health department may assist with patient notification if determined to be necessary. For return-to-work guidance, review the Criteria for Return to Work for Healthcare Personnel with SARS-CoV-2 Infection (Interim Guidance).
- Staff Training: Provide and document additional staff training as protocols change. Maintain training records in administrative files.
Ms. Hill is senior patient safety risk manager at The Doctors Company. The guidelines suggested here are not rules, do not constitute legal advice, and do not ensure a successful outcome. The ultimate decision regarding the appropriateness of any treatment must be made by each health care provider considering the circumstances of the individual situation and in accordance with the laws of the jurisdiction in which the care is rendered.
Editor’s note: This article has been provided by The Doctors Company, the exclusively endorsed medical malpractice carrier for the Society of Hospital Medicine.
As the pandemic hits its third nationwide surge, families are gathering for the holidays, and medical practices are preparing for a potential increase in cases. Medical offices in states that were not strongly affected by the first and second waves of the virus may now be facing an influx of COVID-19 patients. Therefore, medical offices must remain very attentive to the widespread outbreak of COVID-19, continuing to proactively take steps to safely manage patients while protecting clinical staff.
Here are tips and resources for this season of the pandemic:
- Documentation: Maintain administrative records of how you have adapted to the evolving crisis, including the challenges you faced. For details, see Keep a COVID-19 Diary: Document Now in Case of Future Lawsuits.
- Legislation and Guidance: Reference the CDC; your state medical board; professional societies; and federal, state, and local authorities daily for public health guidance and new legislation, as this continues to be a fluid situation.
- Screening Criteria: Follow the CDC’s patient assessment protocol for early disease detection for patients presenting to your practice. Patients should be screened using these guidelines: Overview of Testing for SARS-CoV-2 (COVID-19). Essential visitors to your facility should also be assessed for symptoms of coronavirus and contact exposure and redirected to remain outside if suspect.
- Accepting Patients: Do not turn patients away simply because a patient calls with acute respiratory symptoms. Refusing assessment/care may lead to concerns of patient abandonment.
- Designated Triage Location: Check with your local public health authorities for locations designated to triage suspected patients, so exposure is limited in general medical offices.
- Telehealth Triage: Licensed staff should be trained in triage protocol to determine which patients can be managed safely at home. See Healthcare Facilities: Managing Operations During the COVID-19 Pandemic. The Doctors Company offers resources on telemedicine in our COVID-19 Telehealth Resource Center.
- Patient Testing: When there is a reasonable presumption that a patient may have been exposed to COVID-19, contact the local or state health department to coordinate testing using available community resources. See the CDC’s Testing for COVID-19 , the COVID-19 Testing Overview, and the Clinician Call Center.
- Elective Services: Check with regional governmental and health authorities on the provision of nonessential and elective health care visits and group-related activities. Many states continue or have reinstated restrictions on the provision of nonurgent, elective surgeries and procedures.
- Patient Precautions: Educational resources, including posters for use in the medical office, are available from the WHO and for health care workers from the CDC (Contact Precautions, Droplet Precautions, and Airborne Precautions). Reference the CDC’s and Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19) for patient management guidance.
- Provider/Staff Precautions: Follow Standard Precautions and Transmission-Based Precautions, including gloves, gowns, protective eyewear, and NIOSH-certified N95 respirators that have been properly fit-tested. If there is a shortage of N95 respirators in your facility, access current CDC respirator recommendations and review Optimizing Personal Protective Equipment (PPE) Supplies.
- Limit Exposure: Limit staff exposure to suspected patients, with the exam room door kept closed. Ideally, the designated exam room should be at the back of the office, far away from other staff and patients.
- Surface Disinfection: Once the patient exits the room, conduct surface disinfection while staff continues to wear PPE.
- Patient Education: Provide up-to-date, factual information on the virus to suspected COVID-19–positive patients and their close contacts.
- Provider/Staff Exposure: Screen health care personnel daily for symptoms/contacts relevant to COVID-19. Any unprotected occupational exposure by staff members should be assessed and monitored. See Interim U.S. Guidance for Risk Assessment and Work Restrictions for Healthcare Personnel with Potential Exposure to COVID-19. Should providers and/or staff test positive within your facility, conduct and document a risk assessment identifying contacts, type of interaction, and PPE in use, then contact local health authorities for additional instruction. The CDC provides guidance here under the section “Infection Control.” The health department may assist with patient notification if determined to be necessary. For return-to-work guidance, review the Criteria for Return to Work for Healthcare Personnel with SARS-CoV-2 Infection (Interim Guidance).
- Staff Training: Provide and document additional staff training as protocols change. Maintain training records in administrative files.
Ms. Hill is senior patient safety risk manager at The Doctors Company. The guidelines suggested here are not rules, do not constitute legal advice, and do not ensure a successful outcome. The ultimate decision regarding the appropriateness of any treatment must be made by each health care provider considering the circumstances of the individual situation and in accordance with the laws of the jurisdiction in which the care is rendered.
Etonogestrel implants may be bent, fractured by trauma or during sports
In 2017, Global Pediatric Health published a case report series associated with the use of long-acting reversible contraceptives, specifically the etonogestrel implant.
In November 2020, the makers of the etonogestrel implant (Merck) recommended a change in practice with the release of a notice to health care providers certified in the training of this product. This mass marketing blast included an updated warning and cautions for prescribers as well as patient information on the potential risks of migration, fracture, and bent devices attributable to trauma or sports. “Broken or Bent Implant (Section 5.16). The addition of the following underlined language: “There have been reports of broken or bent implants, which may be related to external forces (e.g., manipulation of the implant or contact sports) while in the patient’s arm. There have also been reports of migration of a broken implant fragment within the arm.”
Clearly the etonogestrel subdermal hormonal implant is an effective form of contraception and particularly beneficial in nonadherent sexually active teens who struggle to remember oral contraceptives. But it is important to be aware of this alert. Little is known about the type of trauma or rate of external force required to cause migration, fracture, or bend implants. This update requires adequate counseling of potential risks and complications of the etonogestrel implant, including the risk of migration, fracture, or bent devices specifically in the event of contact sports and trauma.
Ms. Thew is medical director of the department of adolescent medicine at Children’s Wisconsin in Milwaukee. She is a member of the Pediatric News editorial advisory board. She had no relevant financial disclosures. Email Ms. Thew at [email protected].
In 2017, Global Pediatric Health published a case report series associated with the use of long-acting reversible contraceptives, specifically the etonogestrel implant.
In November 2020, the makers of the etonogestrel implant (Merck) recommended a change in practice with the release of a notice to health care providers certified in the training of this product. This mass marketing blast included an updated warning and cautions for prescribers as well as patient information on the potential risks of migration, fracture, and bent devices attributable to trauma or sports. “Broken or Bent Implant (Section 5.16). The addition of the following underlined language: “There have been reports of broken or bent implants, which may be related to external forces (e.g., manipulation of the implant or contact sports) while in the patient’s arm. There have also been reports of migration of a broken implant fragment within the arm.”
Clearly the etonogestrel subdermal hormonal implant is an effective form of contraception and particularly beneficial in nonadherent sexually active teens who struggle to remember oral contraceptives. But it is important to be aware of this alert. Little is known about the type of trauma or rate of external force required to cause migration, fracture, or bend implants. This update requires adequate counseling of potential risks and complications of the etonogestrel implant, including the risk of migration, fracture, or bent devices specifically in the event of contact sports and trauma.
Ms. Thew is medical director of the department of adolescent medicine at Children’s Wisconsin in Milwaukee. She is a member of the Pediatric News editorial advisory board. She had no relevant financial disclosures. Email Ms. Thew at [email protected].
In 2017, Global Pediatric Health published a case report series associated with the use of long-acting reversible contraceptives, specifically the etonogestrel implant.
In November 2020, the makers of the etonogestrel implant (Merck) recommended a change in practice with the release of a notice to health care providers certified in the training of this product. This mass marketing blast included an updated warning and cautions for prescribers as well as patient information on the potential risks of migration, fracture, and bent devices attributable to trauma or sports. “Broken or Bent Implant (Section 5.16). The addition of the following underlined language: “There have been reports of broken or bent implants, which may be related to external forces (e.g., manipulation of the implant or contact sports) while in the patient’s arm. There have also been reports of migration of a broken implant fragment within the arm.”
Clearly the etonogestrel subdermal hormonal implant is an effective form of contraception and particularly beneficial in nonadherent sexually active teens who struggle to remember oral contraceptives. But it is important to be aware of this alert. Little is known about the type of trauma or rate of external force required to cause migration, fracture, or bend implants. This update requires adequate counseling of potential risks and complications of the etonogestrel implant, including the risk of migration, fracture, or bent devices specifically in the event of contact sports and trauma.
Ms. Thew is medical director of the department of adolescent medicine at Children’s Wisconsin in Milwaukee. She is a member of the Pediatric News editorial advisory board. She had no relevant financial disclosures. Email Ms. Thew at [email protected].
To vape or not to vape: Is that really a question?
All pediatricians are relieved that the rates of children smoking cigarettes has dropped steadily since 2011. This decline seems to be associated with education on the dangers of cigarettes and fewer parents smoking. Perhaps less modeling of cigarette use in movies (although it increased again from 2010 to 2019) and lawsuits against advertisements targeting children also has helped.
“Whew,” we may have said, “we can relax our efforts to convince children to avoid smoking.” But, as is commonly true in medicine, the next threat was right around the corner – in this case vaping or e-cigarettes, also called vapes, e-hookahs, vape pens, tank systems, mods, and electronic nicotine delivery systems. And the size of the problem is huge – over 20% of high school students report using e-cigarettes – and immediate, as vaping can kill in the short term as well as causing long-term harm.
“E-cigarette, or vaping, product use–associated Lung Injury” – EVALI for short – has killed 68 vapers and hospitalized thousands. EVALI is thought to be caused by a vitamin E acetate additive used when vaping marijuana, particularly from informal sources like friends, family, or in-person or online dealers.
Vaping increases the risk of severe COVID-19 disease
While EVALI deaths dropped in months after being explained, the COVID-19 epidemic is now a much greater threat to vapers. Vaping increases risk of severe COVID-19 disease because of its immediate paralysis of lung cilia. Sharing vape devices and touching one’s lips while using also increase the risk of virus transmission. Vaping and smoking increase the number of ACE2 receptors to which the SARS-CoV-2 virus attaches causing the characteristic cell damage, and suppresses macrophages and neutrophils, resulting in more smokers testing positive, being twice as likely to develop a severe illness and get hospitalized because of pneumonia from COVID-19, and being less likely to recover. Unfortunately, addressing this new threat to the immediate and long-term health of our patients appears to be more complicated than for addressing smoking tobacco. First of all, vaping is much more difficult to detect than smelly cigarettes sending smoke signals from behind the garage or in the school bathrooms. Many, if not most, adults do not recognize the vaping devices when they see them, as many are tiny and some look like computer thumb drives. The aerosol emitted when in use, while containing dangerous toxins, has less odor than tobacco smoke. Vaping equipment and ads have been designed to attract youth, including linking them to sports and music events. Vaping has been advertised as a way to wean off nicotine addiction, a claim that has some scientific evidence in adults, but at a lower dose of nicotine. Warning children about the dangers of marijuana vaping has been made less credible by the rapid expansion of legalization of marijuana around the United States, eliciting “I told you it was fine” reactions from youth. And the person vaping does not know what or how much of the psychoactive components are being delivered into their bodies. One Juul pod, for example, has the equivalent in nicotine of an entire pack of 20 cigarettes. They are highly addictive, especially to the developing brain, such that youth who vape are more likely to become addicted and to smoke cigarettes in the future.
Help from federal regulation has been weak
While all 50 states ban sales to youth, adults can still buy. Food and Drug Administration limitations on kid-friendly ads, and use of sweet, fruity, and mint flavorings that are most preferred by children, apply only to new producers. The FDA does not yet regulate content of vaping solutions.
So we pediatricians are on the front line for this new threat to prevent vaping or convince youth to cut down or quit. The first step in addressing vaping is being knowledgeable about its many known and emerging health risks. It may seem obvious that the dangers of vaping microscopic particles depends on the contents. Water vapor alone is not dangerous; in fact, we prescribe it in nebulizers. Unfortunately, the contents of different vaping products vary and are not well defined in different vape products. The process of using an electric current to vaporize a substance can make it more toxic than the precursor, and teens have little idea about the substances they are inhaling. The psychoactive components vary from nicotine to tetrahydrocannabinol in varying amounts. These have the well known effects of stimulation or a high, but also the potential adverse effects of poor concentration, agitation, and even psychosis. Most e-cigarettes contain nicotine, which is highly addictive and can harm adolescent brain development, which continues into the early- to mid-20s. About two-thirds of Juul users aged 15-24 years did not know that it always contains nicotine, as do 99% of all vape solutions (Centers for Disease Control and Prevention, 2020). Earlier use of nicotine is more highly associated with later addiction to tobacco products that cause lung damage, acid reflux, insulin resistance, harm to the testes, harm to fetuses, cancer, and heart disease.
E-cigarette aerosols also contain dozens of other harmful substances besides nicotine ranging from acetone, propylene glycol, and metals to formaldehyde and ethyl benzene. These same chemicals are part of familiar toxic substances such as antifreeze, paint thinner, and pesticides. These cause ear, eye and throat irritation, and impairments in the cardiovascular system reducing athletic ability – at the least. Some flavorings in vape fluids also are toxic. Even the residual left on furniture and floors is harmful to those coming in contact, including pets.
How to encourage teens not to vaping
Trying to scare youth about health hazards is not generally effective in stopping risk behaviors since adolescence is a time of perceived singularity (it does not apply to me) and even a sense of immortality. Teens also see peers who vape as being unaffected and decide on using based on this small personal sample instead of valid statistics.
But teens do pay some attention to peer models or influencers saying why they do not use. One source of such testimony you can refer to is videos of inspiring athletes, musicians, and other “cool” young adults found on the naturalhigh.org website. You may know other examples of community teens desisting you can reference.
Parent rules, and less so advice, against smoking have been shown to be effective in deterring youth cigarette smoking. Because parents are less aware of vaping and its dangers, another step we can take is educating parents in our practices about vaping, its variable forms, its effects, and dangers, supplying authoritative materials, and advising them to talk with their children. Other steps the American Academy of Pediatrics recommends regarding smoking is for parents to be a role model of not using or try to quit, designate the house and car as smoking free, avoid children viewing smoking in media, tell their children about the side effects, and encourage their children who use to quit. Parents also can encourage schools to teach and have rules about smoking and vaping (e.g., med.stanford.edu/tobaccopreventiontoolkit.html).
Another approach we have been using is to not only screen for all substance use, but also to gather information about the teen’s strengths, activities, and life goals both to enhance rapport and to reference during motivational interviewing as reasons to avoid, reduce, or quit vaping. Motivational interviewing has been shown to help patients make healthier lifestyle choices by nonjudgmentally exploring their pros and cons in a conversation that takes into account readiness to change. This fits well with the stage of developing autonomy when teens want above all to make their own decisions. The cons of using can be discussed as including the effects and side effects of vaping interfering with their favored activities and moving towards their identified goals. Praising abstinence and asking them to show you how they could decline offers to vape are valuable reinforcement you can provide.
Finally, we all know that teens hate being manipulated. Vaping education we provide can make it clear that youth are being tricked by companies – most being large cigarette producers who know the dangers of vaping – into getting addicted so these companies can get rich on their money.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She has no other relevant disclosures. Dr. Howard’s contribution to this publication is as a paid expert to MDedge News. Email her at [email protected].
All pediatricians are relieved that the rates of children smoking cigarettes has dropped steadily since 2011. This decline seems to be associated with education on the dangers of cigarettes and fewer parents smoking. Perhaps less modeling of cigarette use in movies (although it increased again from 2010 to 2019) and lawsuits against advertisements targeting children also has helped.
“Whew,” we may have said, “we can relax our efforts to convince children to avoid smoking.” But, as is commonly true in medicine, the next threat was right around the corner – in this case vaping or e-cigarettes, also called vapes, e-hookahs, vape pens, tank systems, mods, and electronic nicotine delivery systems. And the size of the problem is huge – over 20% of high school students report using e-cigarettes – and immediate, as vaping can kill in the short term as well as causing long-term harm.
“E-cigarette, or vaping, product use–associated Lung Injury” – EVALI for short – has killed 68 vapers and hospitalized thousands. EVALI is thought to be caused by a vitamin E acetate additive used when vaping marijuana, particularly from informal sources like friends, family, or in-person or online dealers.
Vaping increases the risk of severe COVID-19 disease
While EVALI deaths dropped in months after being explained, the COVID-19 epidemic is now a much greater threat to vapers. Vaping increases risk of severe COVID-19 disease because of its immediate paralysis of lung cilia. Sharing vape devices and touching one’s lips while using also increase the risk of virus transmission. Vaping and smoking increase the number of ACE2 receptors to which the SARS-CoV-2 virus attaches causing the characteristic cell damage, and suppresses macrophages and neutrophils, resulting in more smokers testing positive, being twice as likely to develop a severe illness and get hospitalized because of pneumonia from COVID-19, and being less likely to recover. Unfortunately, addressing this new threat to the immediate and long-term health of our patients appears to be more complicated than for addressing smoking tobacco. First of all, vaping is much more difficult to detect than smelly cigarettes sending smoke signals from behind the garage or in the school bathrooms. Many, if not most, adults do not recognize the vaping devices when they see them, as many are tiny and some look like computer thumb drives. The aerosol emitted when in use, while containing dangerous toxins, has less odor than tobacco smoke. Vaping equipment and ads have been designed to attract youth, including linking them to sports and music events. Vaping has been advertised as a way to wean off nicotine addiction, a claim that has some scientific evidence in adults, but at a lower dose of nicotine. Warning children about the dangers of marijuana vaping has been made less credible by the rapid expansion of legalization of marijuana around the United States, eliciting “I told you it was fine” reactions from youth. And the person vaping does not know what or how much of the psychoactive components are being delivered into their bodies. One Juul pod, for example, has the equivalent in nicotine of an entire pack of 20 cigarettes. They are highly addictive, especially to the developing brain, such that youth who vape are more likely to become addicted and to smoke cigarettes in the future.
Help from federal regulation has been weak
While all 50 states ban sales to youth, adults can still buy. Food and Drug Administration limitations on kid-friendly ads, and use of sweet, fruity, and mint flavorings that are most preferred by children, apply only to new producers. The FDA does not yet regulate content of vaping solutions.
So we pediatricians are on the front line for this new threat to prevent vaping or convince youth to cut down or quit. The first step in addressing vaping is being knowledgeable about its many known and emerging health risks. It may seem obvious that the dangers of vaping microscopic particles depends on the contents. Water vapor alone is not dangerous; in fact, we prescribe it in nebulizers. Unfortunately, the contents of different vaping products vary and are not well defined in different vape products. The process of using an electric current to vaporize a substance can make it more toxic than the precursor, and teens have little idea about the substances they are inhaling. The psychoactive components vary from nicotine to tetrahydrocannabinol in varying amounts. These have the well known effects of stimulation or a high, but also the potential adverse effects of poor concentration, agitation, and even psychosis. Most e-cigarettes contain nicotine, which is highly addictive and can harm adolescent brain development, which continues into the early- to mid-20s. About two-thirds of Juul users aged 15-24 years did not know that it always contains nicotine, as do 99% of all vape solutions (Centers for Disease Control and Prevention, 2020). Earlier use of nicotine is more highly associated with later addiction to tobacco products that cause lung damage, acid reflux, insulin resistance, harm to the testes, harm to fetuses, cancer, and heart disease.
E-cigarette aerosols also contain dozens of other harmful substances besides nicotine ranging from acetone, propylene glycol, and metals to formaldehyde and ethyl benzene. These same chemicals are part of familiar toxic substances such as antifreeze, paint thinner, and pesticides. These cause ear, eye and throat irritation, and impairments in the cardiovascular system reducing athletic ability – at the least. Some flavorings in vape fluids also are toxic. Even the residual left on furniture and floors is harmful to those coming in contact, including pets.
How to encourage teens not to vaping
Trying to scare youth about health hazards is not generally effective in stopping risk behaviors since adolescence is a time of perceived singularity (it does not apply to me) and even a sense of immortality. Teens also see peers who vape as being unaffected and decide on using based on this small personal sample instead of valid statistics.
But teens do pay some attention to peer models or influencers saying why they do not use. One source of such testimony you can refer to is videos of inspiring athletes, musicians, and other “cool” young adults found on the naturalhigh.org website. You may know other examples of community teens desisting you can reference.
Parent rules, and less so advice, against smoking have been shown to be effective in deterring youth cigarette smoking. Because parents are less aware of vaping and its dangers, another step we can take is educating parents in our practices about vaping, its variable forms, its effects, and dangers, supplying authoritative materials, and advising them to talk with their children. Other steps the American Academy of Pediatrics recommends regarding smoking is for parents to be a role model of not using or try to quit, designate the house and car as smoking free, avoid children viewing smoking in media, tell their children about the side effects, and encourage their children who use to quit. Parents also can encourage schools to teach and have rules about smoking and vaping (e.g., med.stanford.edu/tobaccopreventiontoolkit.html).
Another approach we have been using is to not only screen for all substance use, but also to gather information about the teen’s strengths, activities, and life goals both to enhance rapport and to reference during motivational interviewing as reasons to avoid, reduce, or quit vaping. Motivational interviewing has been shown to help patients make healthier lifestyle choices by nonjudgmentally exploring their pros and cons in a conversation that takes into account readiness to change. This fits well with the stage of developing autonomy when teens want above all to make their own decisions. The cons of using can be discussed as including the effects and side effects of vaping interfering with their favored activities and moving towards their identified goals. Praising abstinence and asking them to show you how they could decline offers to vape are valuable reinforcement you can provide.
Finally, we all know that teens hate being manipulated. Vaping education we provide can make it clear that youth are being tricked by companies – most being large cigarette producers who know the dangers of vaping – into getting addicted so these companies can get rich on their money.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She has no other relevant disclosures. Dr. Howard’s contribution to this publication is as a paid expert to MDedge News. Email her at [email protected].
All pediatricians are relieved that the rates of children smoking cigarettes has dropped steadily since 2011. This decline seems to be associated with education on the dangers of cigarettes and fewer parents smoking. Perhaps less modeling of cigarette use in movies (although it increased again from 2010 to 2019) and lawsuits against advertisements targeting children also has helped.
“Whew,” we may have said, “we can relax our efforts to convince children to avoid smoking.” But, as is commonly true in medicine, the next threat was right around the corner – in this case vaping or e-cigarettes, also called vapes, e-hookahs, vape pens, tank systems, mods, and electronic nicotine delivery systems. And the size of the problem is huge – over 20% of high school students report using e-cigarettes – and immediate, as vaping can kill in the short term as well as causing long-term harm.
“E-cigarette, or vaping, product use–associated Lung Injury” – EVALI for short – has killed 68 vapers and hospitalized thousands. EVALI is thought to be caused by a vitamin E acetate additive used when vaping marijuana, particularly from informal sources like friends, family, or in-person or online dealers.
Vaping increases the risk of severe COVID-19 disease
While EVALI deaths dropped in months after being explained, the COVID-19 epidemic is now a much greater threat to vapers. Vaping increases risk of severe COVID-19 disease because of its immediate paralysis of lung cilia. Sharing vape devices and touching one’s lips while using also increase the risk of virus transmission. Vaping and smoking increase the number of ACE2 receptors to which the SARS-CoV-2 virus attaches causing the characteristic cell damage, and suppresses macrophages and neutrophils, resulting in more smokers testing positive, being twice as likely to develop a severe illness and get hospitalized because of pneumonia from COVID-19, and being less likely to recover. Unfortunately, addressing this new threat to the immediate and long-term health of our patients appears to be more complicated than for addressing smoking tobacco. First of all, vaping is much more difficult to detect than smelly cigarettes sending smoke signals from behind the garage or in the school bathrooms. Many, if not most, adults do not recognize the vaping devices when they see them, as many are tiny and some look like computer thumb drives. The aerosol emitted when in use, while containing dangerous toxins, has less odor than tobacco smoke. Vaping equipment and ads have been designed to attract youth, including linking them to sports and music events. Vaping has been advertised as a way to wean off nicotine addiction, a claim that has some scientific evidence in adults, but at a lower dose of nicotine. Warning children about the dangers of marijuana vaping has been made less credible by the rapid expansion of legalization of marijuana around the United States, eliciting “I told you it was fine” reactions from youth. And the person vaping does not know what or how much of the psychoactive components are being delivered into their bodies. One Juul pod, for example, has the equivalent in nicotine of an entire pack of 20 cigarettes. They are highly addictive, especially to the developing brain, such that youth who vape are more likely to become addicted and to smoke cigarettes in the future.
Help from federal regulation has been weak
While all 50 states ban sales to youth, adults can still buy. Food and Drug Administration limitations on kid-friendly ads, and use of sweet, fruity, and mint flavorings that are most preferred by children, apply only to new producers. The FDA does not yet regulate content of vaping solutions.
So we pediatricians are on the front line for this new threat to prevent vaping or convince youth to cut down or quit. The first step in addressing vaping is being knowledgeable about its many known and emerging health risks. It may seem obvious that the dangers of vaping microscopic particles depends on the contents. Water vapor alone is not dangerous; in fact, we prescribe it in nebulizers. Unfortunately, the contents of different vaping products vary and are not well defined in different vape products. The process of using an electric current to vaporize a substance can make it more toxic than the precursor, and teens have little idea about the substances they are inhaling. The psychoactive components vary from nicotine to tetrahydrocannabinol in varying amounts. These have the well known effects of stimulation or a high, but also the potential adverse effects of poor concentration, agitation, and even psychosis. Most e-cigarettes contain nicotine, which is highly addictive and can harm adolescent brain development, which continues into the early- to mid-20s. About two-thirds of Juul users aged 15-24 years did not know that it always contains nicotine, as do 99% of all vape solutions (Centers for Disease Control and Prevention, 2020). Earlier use of nicotine is more highly associated with later addiction to tobacco products that cause lung damage, acid reflux, insulin resistance, harm to the testes, harm to fetuses, cancer, and heart disease.
E-cigarette aerosols also contain dozens of other harmful substances besides nicotine ranging from acetone, propylene glycol, and metals to formaldehyde and ethyl benzene. These same chemicals are part of familiar toxic substances such as antifreeze, paint thinner, and pesticides. These cause ear, eye and throat irritation, and impairments in the cardiovascular system reducing athletic ability – at the least. Some flavorings in vape fluids also are toxic. Even the residual left on furniture and floors is harmful to those coming in contact, including pets.
How to encourage teens not to vaping
Trying to scare youth about health hazards is not generally effective in stopping risk behaviors since adolescence is a time of perceived singularity (it does not apply to me) and even a sense of immortality. Teens also see peers who vape as being unaffected and decide on using based on this small personal sample instead of valid statistics.
But teens do pay some attention to peer models or influencers saying why they do not use. One source of such testimony you can refer to is videos of inspiring athletes, musicians, and other “cool” young adults found on the naturalhigh.org website. You may know other examples of community teens desisting you can reference.
Parent rules, and less so advice, against smoking have been shown to be effective in deterring youth cigarette smoking. Because parents are less aware of vaping and its dangers, another step we can take is educating parents in our practices about vaping, its variable forms, its effects, and dangers, supplying authoritative materials, and advising them to talk with their children. Other steps the American Academy of Pediatrics recommends regarding smoking is for parents to be a role model of not using or try to quit, designate the house and car as smoking free, avoid children viewing smoking in media, tell their children about the side effects, and encourage their children who use to quit. Parents also can encourage schools to teach and have rules about smoking and vaping (e.g., med.stanford.edu/tobaccopreventiontoolkit.html).
Another approach we have been using is to not only screen for all substance use, but also to gather information about the teen’s strengths, activities, and life goals both to enhance rapport and to reference during motivational interviewing as reasons to avoid, reduce, or quit vaping. Motivational interviewing has been shown to help patients make healthier lifestyle choices by nonjudgmentally exploring their pros and cons in a conversation that takes into account readiness to change. This fits well with the stage of developing autonomy when teens want above all to make their own decisions. The cons of using can be discussed as including the effects and side effects of vaping interfering with their favored activities and moving towards their identified goals. Praising abstinence and asking them to show you how they could decline offers to vape are valuable reinforcement you can provide.
Finally, we all know that teens hate being manipulated. Vaping education we provide can make it clear that youth are being tricked by companies – most being large cigarette producers who know the dangers of vaping – into getting addicted so these companies can get rich on their money.
Dr. Howard is assistant professor of pediatrics at Johns Hopkins University, Baltimore, and creator of CHADIS (www.CHADIS.com). She has no other relevant disclosures. Dr. Howard’s contribution to this publication is as a paid expert to MDedge News. Email her at [email protected].
GI physicians urge COVID-19 vaccines for all IBD patients
Gastroenterologists at the Advances in Inflammatory Bowel Disease 2020 annual meeting said they will strongly advise their patients to take the COVID-19 vaccines as they become available.
Announcement that the first vaccine, Pfizer’s, was recommended for emergency use authorization came in the middle of AIBD’s Thursday evening COVID-19 session.
Miguel Regueiro, MD, chair of the department of gastroenterology, hepatology, and nutrition at Cleveland Clinic in Ohio, said, “We’re uniformly recommending this to all our patients.”
“The [vaccines] leading the pack do not have any replicating virus and thus can be used in immunocompromised people,” Maria Abreu, MD, director of the Crohn’s & colitis center at the University of Miami, told this news organization. “Although it is true that we don’t know – and won’t know for a while – whether the high levels of efficacy seen with the mRNA vaccines so far will be achieved in patients who are immunocompromised, there is every reason to believe that [the vaccine] will still be enough to protect them from complications of COVID-19.”
The bottom line, she said, is that “it’s much safer to get a vaccine than it is to take your chances of getting COVID-19.”
David T. Rubin, MD, chief of gastroenterology, hepatology, and nutrition at UChicago Medicine, said in a session earlier in the day, “Emerging information about the messenger RNA looks like it’s going to be safe for our population, but of course we want to see more. Messenger RNA degrades within days of giving it, so it’s not expected to linger or generate any other problems we can think of.”
Dr. Abreu said there’s no evidence that inflammatory bowel disease (IBD) patients are more susceptible to COVID-19 infection even though the entry molecules are expressed in the GI tract. “They are really not differentially expressed in IBD and, if anything, some of our more potent therapies reduce the expression of these molecules in the GI tract,” she said.
Regarding how IBD medications affect outcomes if patients are infected with COVID-19, Dr. Abreu pointed out that corticosteroids seem to be associated with worse outcomes. “I would posit that it has to do with initially allowing there to be a lot of very rapid viral replication,” she said.
And she also noted that any of the mainstay drugs for IBD – the anti–tumor necrosis factor (TNF) therapies – are showing promise as treatments for COVID-19.
Updates from the IBD-COVID-19 registry
Michael Kappelman, MD, MPH, from the University of North Carolina at Chapel Hill said information from the Secure-IBD registry, which collects real-time global information on how COVID-19 affects IBD patients, suggests that these patients “may have a more severe course than the general population, but not by much.”
He reported the registry had logged more than 3,300 reported COVID-19 cases among IBD patients from 62 countries.
Registry outcomes through the end of November have found a mean age of reported cases of 40 years, and that 21% of patients were hospitalized with an average length of stay of 10.2 days, 4% required intensive care unit admission, and 2% died.
The majority of the deaths reported to Secure-IBD occurred in patients older than 60 years, Dr. Kappelman said, adding that the hospitalizations and death rates in IBD patients with no comorbidities were relatively low.
“My belief is that available data are actually more reassuring than alarming,” he said.
Dr. Kappelman and other investigators found that combination therapy that includes thiopurines and thiopurine monotherapy are “associated with about a fourfold risk of the requirement for intensive care or mortality from COVID,” compared with anti-TNF monotherapy.
In cases reported to Secure-IBD, about 25% of IBD patients with COVID-19 developed new GI symptoms, primarily diarrhea and abdominal pain, he said.
In his practice, Dr. Kappelman said, he minimizes use of steroids and has found that COVID-19 adds a reason to favor anti-TNF over 6-mercaptopurine (6-MP) plus azathioprine.
He also advises “a high alert for COVID-19 in patients with new GI symptoms.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Rubin has served as a director, officer, partner, employee, adviser, consultant, or trustee for AbbVie, Abgenomics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dizal Pharmaceuticals, GalenPharma/Atlantica, Genentech/Roche, Gilead Sciences, Ichnos Sciences, GlaxoSmithKline, Janssen, Eli Lilly, Pfizer, Prometheus Laboratories, Reistone, Shire, Takeda Pharmaceutical, and Techlab. In addition, he has received research grants from AbbVie, Genentech/Roche, Janssen Pharmaceuticals, Prometheus Laboratories, Shire, and Takeda Pharmaceutical Company; and holds stock options in Abgenomics and Biomica. Regueiro and Kappelman have disclosed no relevant financial relationships.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
A version of this article originally appeared on Medscape.com.
Gastroenterologists at the Advances in Inflammatory Bowel Disease 2020 annual meeting said they will strongly advise their patients to take the COVID-19 vaccines as they become available.
Announcement that the first vaccine, Pfizer’s, was recommended for emergency use authorization came in the middle of AIBD’s Thursday evening COVID-19 session.
Miguel Regueiro, MD, chair of the department of gastroenterology, hepatology, and nutrition at Cleveland Clinic in Ohio, said, “We’re uniformly recommending this to all our patients.”
“The [vaccines] leading the pack do not have any replicating virus and thus can be used in immunocompromised people,” Maria Abreu, MD, director of the Crohn’s & colitis center at the University of Miami, told this news organization. “Although it is true that we don’t know – and won’t know for a while – whether the high levels of efficacy seen with the mRNA vaccines so far will be achieved in patients who are immunocompromised, there is every reason to believe that [the vaccine] will still be enough to protect them from complications of COVID-19.”
The bottom line, she said, is that “it’s much safer to get a vaccine than it is to take your chances of getting COVID-19.”
David T. Rubin, MD, chief of gastroenterology, hepatology, and nutrition at UChicago Medicine, said in a session earlier in the day, “Emerging information about the messenger RNA looks like it’s going to be safe for our population, but of course we want to see more. Messenger RNA degrades within days of giving it, so it’s not expected to linger or generate any other problems we can think of.”
Dr. Abreu said there’s no evidence that inflammatory bowel disease (IBD) patients are more susceptible to COVID-19 infection even though the entry molecules are expressed in the GI tract. “They are really not differentially expressed in IBD and, if anything, some of our more potent therapies reduce the expression of these molecules in the GI tract,” she said.
Regarding how IBD medications affect outcomes if patients are infected with COVID-19, Dr. Abreu pointed out that corticosteroids seem to be associated with worse outcomes. “I would posit that it has to do with initially allowing there to be a lot of very rapid viral replication,” she said.
And she also noted that any of the mainstay drugs for IBD – the anti–tumor necrosis factor (TNF) therapies – are showing promise as treatments for COVID-19.
Updates from the IBD-COVID-19 registry
Michael Kappelman, MD, MPH, from the University of North Carolina at Chapel Hill said information from the Secure-IBD registry, which collects real-time global information on how COVID-19 affects IBD patients, suggests that these patients “may have a more severe course than the general population, but not by much.”
He reported the registry had logged more than 3,300 reported COVID-19 cases among IBD patients from 62 countries.
Registry outcomes through the end of November have found a mean age of reported cases of 40 years, and that 21% of patients were hospitalized with an average length of stay of 10.2 days, 4% required intensive care unit admission, and 2% died.
The majority of the deaths reported to Secure-IBD occurred in patients older than 60 years, Dr. Kappelman said, adding that the hospitalizations and death rates in IBD patients with no comorbidities were relatively low.
“My belief is that available data are actually more reassuring than alarming,” he said.
Dr. Kappelman and other investigators found that combination therapy that includes thiopurines and thiopurine monotherapy are “associated with about a fourfold risk of the requirement for intensive care or mortality from COVID,” compared with anti-TNF monotherapy.
In cases reported to Secure-IBD, about 25% of IBD patients with COVID-19 developed new GI symptoms, primarily diarrhea and abdominal pain, he said.
In his practice, Dr. Kappelman said, he minimizes use of steroids and has found that COVID-19 adds a reason to favor anti-TNF over 6-mercaptopurine (6-MP) plus azathioprine.
He also advises “a high alert for COVID-19 in patients with new GI symptoms.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Rubin has served as a director, officer, partner, employee, adviser, consultant, or trustee for AbbVie, Abgenomics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dizal Pharmaceuticals, GalenPharma/Atlantica, Genentech/Roche, Gilead Sciences, Ichnos Sciences, GlaxoSmithKline, Janssen, Eli Lilly, Pfizer, Prometheus Laboratories, Reistone, Shire, Takeda Pharmaceutical, and Techlab. In addition, he has received research grants from AbbVie, Genentech/Roche, Janssen Pharmaceuticals, Prometheus Laboratories, Shire, and Takeda Pharmaceutical Company; and holds stock options in Abgenomics and Biomica. Regueiro and Kappelman have disclosed no relevant financial relationships.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
A version of this article originally appeared on Medscape.com.
Gastroenterologists at the Advances in Inflammatory Bowel Disease 2020 annual meeting said they will strongly advise their patients to take the COVID-19 vaccines as they become available.
Announcement that the first vaccine, Pfizer’s, was recommended for emergency use authorization came in the middle of AIBD’s Thursday evening COVID-19 session.
Miguel Regueiro, MD, chair of the department of gastroenterology, hepatology, and nutrition at Cleveland Clinic in Ohio, said, “We’re uniformly recommending this to all our patients.”
“The [vaccines] leading the pack do not have any replicating virus and thus can be used in immunocompromised people,” Maria Abreu, MD, director of the Crohn’s & colitis center at the University of Miami, told this news organization. “Although it is true that we don’t know – and won’t know for a while – whether the high levels of efficacy seen with the mRNA vaccines so far will be achieved in patients who are immunocompromised, there is every reason to believe that [the vaccine] will still be enough to protect them from complications of COVID-19.”
The bottom line, she said, is that “it’s much safer to get a vaccine than it is to take your chances of getting COVID-19.”
David T. Rubin, MD, chief of gastroenterology, hepatology, and nutrition at UChicago Medicine, said in a session earlier in the day, “Emerging information about the messenger RNA looks like it’s going to be safe for our population, but of course we want to see more. Messenger RNA degrades within days of giving it, so it’s not expected to linger or generate any other problems we can think of.”
Dr. Abreu said there’s no evidence that inflammatory bowel disease (IBD) patients are more susceptible to COVID-19 infection even though the entry molecules are expressed in the GI tract. “They are really not differentially expressed in IBD and, if anything, some of our more potent therapies reduce the expression of these molecules in the GI tract,” she said.
Regarding how IBD medications affect outcomes if patients are infected with COVID-19, Dr. Abreu pointed out that corticosteroids seem to be associated with worse outcomes. “I would posit that it has to do with initially allowing there to be a lot of very rapid viral replication,” she said.
And she also noted that any of the mainstay drugs for IBD – the anti–tumor necrosis factor (TNF) therapies – are showing promise as treatments for COVID-19.
Updates from the IBD-COVID-19 registry
Michael Kappelman, MD, MPH, from the University of North Carolina at Chapel Hill said information from the Secure-IBD registry, which collects real-time global information on how COVID-19 affects IBD patients, suggests that these patients “may have a more severe course than the general population, but not by much.”
He reported the registry had logged more than 3,300 reported COVID-19 cases among IBD patients from 62 countries.
Registry outcomes through the end of November have found a mean age of reported cases of 40 years, and that 21% of patients were hospitalized with an average length of stay of 10.2 days, 4% required intensive care unit admission, and 2% died.
The majority of the deaths reported to Secure-IBD occurred in patients older than 60 years, Dr. Kappelman said, adding that the hospitalizations and death rates in IBD patients with no comorbidities were relatively low.
“My belief is that available data are actually more reassuring than alarming,” he said.
Dr. Kappelman and other investigators found that combination therapy that includes thiopurines and thiopurine monotherapy are “associated with about a fourfold risk of the requirement for intensive care or mortality from COVID,” compared with anti-TNF monotherapy.
In cases reported to Secure-IBD, about 25% of IBD patients with COVID-19 developed new GI symptoms, primarily diarrhea and abdominal pain, he said.
In his practice, Dr. Kappelman said, he minimizes use of steroids and has found that COVID-19 adds a reason to favor anti-TNF over 6-mercaptopurine (6-MP) plus azathioprine.
He also advises “a high alert for COVID-19 in patients with new GI symptoms.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Rubin has served as a director, officer, partner, employee, adviser, consultant, or trustee for AbbVie, Abgenomics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dizal Pharmaceuticals, GalenPharma/Atlantica, Genentech/Roche, Gilead Sciences, Ichnos Sciences, GlaxoSmithKline, Janssen, Eli Lilly, Pfizer, Prometheus Laboratories, Reistone, Shire, Takeda Pharmaceutical, and Techlab. In addition, he has received research grants from AbbVie, Genentech/Roche, Janssen Pharmaceuticals, Prometheus Laboratories, Shire, and Takeda Pharmaceutical Company; and holds stock options in Abgenomics and Biomica. Regueiro and Kappelman have disclosed no relevant financial relationships.
For the latest clinical guidance, education, research and physician resources about coronavirus, visit the AGA COVID-19 Resource Center at www.gastro.org/COVID.
A version of this article originally appeared on Medscape.com.
Asthma guidelines update FeNO, intermittent ICS use
The updated guidelines address six priority topics, including refined recommendations for the use of fractional exhaled nitric oxide (FeNO) testing, intermittent inhaled corticosteroids (ICS), long-acting muscarinic antagonists (LAMA), and bronchial thermoplasty, but notably exclude any recommendations for the use of fast-emerging biological therapy.
“Biological therapy is the major step forward,” said William W. Busse, MD, professor of allergy and immunology at the University of Wisconsin–Madison, and lead author of the previous guidelines (Bethesda, Md.: NHLBI, 2007). “It wasn’t within the scope of work, so it’s not a criticism, but it is an important shortcoming,” he said. The omission identifies the need for the next update. “This is an area that has to be dealt with,” Dr. Busse stated in an interview.
Including biologic agents would have delayed the release of the recommendations for another year or 2, wrote the expert panel working group of the NHLBI, “and this was felt to be unacceptable.” The working group, overseen by the National Asthma Education and Prevention Program Coordinating Committee, also acknowledged the update is “not a complete revision” of the 2007 guidelines.
The update provides an evidenced-based review of six key topics in asthma care, as Mary Cataletto, MD, FCCP, professor of pediatrics at New York University Long Island, Mineola, pointed out: use of FeNO, indoor allergen mitigation, use of intermittent ICS and LAMA for asthma, role of subcutaneous and sublingual immunotherapy in the treatment of allergic asthma, and the use of bronchial thermoplasty.
“It has been 13 years since the last update and substantial progress has been made since then in understanding how to best treat children and adults with asthma,” said working group member Michael Schatz, MD, MS, FCCP, an allergy specialist at Kaiser Permanente Medical Center in San Diego.
According to Dr. Schatz, the most important updated recommendations are:
- Conditional recommendation for the use of ICS in children aged infant to 4 years with recurrent wheezing with respiratory infections.
- Use of combination ICS-formoterol for maintenance and to relieve flares in patients with moderate to severe asthma.
- Addition of the LAMA inhaled bronchodilator as add-on therapy for severe asthma not controlled by long-acting beta-agonist (LABA)/ICS combination medications.
Another important update, Dr. Cataletto said, is “shared decision-making among members of asthma teams in order to improve asthma care across all age groups.”
In all, the update includes 19 recommendations in the six subject areas. Each recommendation is notated with two values: its strength, either strong or conditional, and the certainty of evidence behind it, either low, moderate, or high. For example, the recommendation for ICS in young children that Dr. Schatz referred to has a conditional strength of recommendation with moderate certainty of evidence.
Using the GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) methodology to determine strength of recommendation is a notable innovation of the latest guidelines, Dr. Busse noted.
Recommendations (strength of recommendation/certainty of evidence) include:
- Use of FeNO in children and adults when the asthma diagnosis is uncertain (conditional/moderate) or in those with allergic asthma and an uncertain course of management (conditional/low).
- Avoid standalone FeNO to evaluate asthma control or the likelihood or severity of future exacerbations, or for in infants to 4-year-olds with recurrent wheezing (strong/low for both).
- Avoid allergen mitigation in routine asthma management for patients who don’t have sensitivity to specific indoor allergens (conditional/low).
- Multicomponent allergen-specific mitigation when specific allergen sensitivity has been identified and pest management alone for symptoms related to specific pest exposure (conditional/low for both).
- Impermeable bedding covers should be part of a multicomponent mitigation strategy, not as a standalone tool, for patients with asthma and dust mite sensitivity (conditional/moderate).
- Daily ICS at onset of a respiratory tract infection along with as-needed short-acting beta-agonists in children aged 4 years and younger with recurrent wheezing but no wheezing between infections rather than as-needed standalone SABA (conditional/high).
- For adults and children aged 12 years and older with mild persistent asthma, either daily low-dose ICS with as-needed SABA or as-needed ICS and SABA concomitantly (conditional/moderate).
- Avoid short-course increased ICS dosing for patients aged 4 years and older with good adherence to daily ICS therapy (conditional/low).
- For patients aged 4 years and older with moderate to severe persistent asthma, a preference for combined ICS-formoterol inhaler over higher dose ICS daily and intermittent SABA or daily ICS-LABA with intermittent SABA (strong/high [aged 12 years and older]; moderate [aged 4-11 years]).
- A preference for combined ICS-formoterol for both daily and relief therapy for patients 12 years and older with severe persistent asthma over higher-dose ICS-LABA daily and intermittent SABA (conditional/high).
- A preference for adding LABA rather than LAMA to ICS in patients aged 12 years and older with uncontrolled persistent asthma (conditional/moderate).
- If LABA isn’t used, add LAMA to ICS in patients aged 12 years and older with uncontrolled persistent asthma rather than continuing the same dose of ICS alone (conditional/moderate).
- In those same patients already on combined ICS-LABA therapy, add LAMA rather than continuing the same dose of ICS-LABA (conditional/moderate).
- Use subcutaneous immunotherapy as a potential adjunct to standard drug therapy in patients aged 5 years and older with mild to moderate allergic asthma when their asthma is controlled on immunotherapy (conditional/moderate).
- Avoid sublingual immunotherapy in patients with persistent allergic asthma (conditional/moderate).
- Avoid bronchial thermoplasty in those 18 years and older with persistent asthma, but consider it in patients who can accept the short-term worsening symptoms or unknown long-term side effects in exchange for the potential benefits (conditional/moderate).
One of the key elements of the guidelines is the use of the SMART (single maintenance and reliever therapy) approach to evaluate the comparative effectiveness of intermittent ICS with formoterol, Dr. Busse noted. “I think that’s a very significant advance. The literature is replete with evidence to support this. Secondly, it really makes life convenient for patients; you have one inhaler.”
The recommendation on SABA use is also significant, Dr. Busse said. “Data have emerged to suggest that if you’re having a need for one of these rescue medications, it’s due to an increase in inflammation in the lower airway, and you want to give an ICS which will act on the inflammation along with the bronchodilator. That’s a new concept, and it’s a very significant step forward.”
Dr. Schatz disclosed financial relationships with Merck, Teva, and ALK-Abello, but was recused from the writing, discussion, and voting related to the immunotherapy recommendation. Dr. Cataletto and Dr. Busse have no relevant relationships to disclose.
SOURCE: Schatz M et al. J Allergy Clin Immunol. 2020;146:1217-70.
The updated guidelines address six priority topics, including refined recommendations for the use of fractional exhaled nitric oxide (FeNO) testing, intermittent inhaled corticosteroids (ICS), long-acting muscarinic antagonists (LAMA), and bronchial thermoplasty, but notably exclude any recommendations for the use of fast-emerging biological therapy.
“Biological therapy is the major step forward,” said William W. Busse, MD, professor of allergy and immunology at the University of Wisconsin–Madison, and lead author of the previous guidelines (Bethesda, Md.: NHLBI, 2007). “It wasn’t within the scope of work, so it’s not a criticism, but it is an important shortcoming,” he said. The omission identifies the need for the next update. “This is an area that has to be dealt with,” Dr. Busse stated in an interview.
Including biologic agents would have delayed the release of the recommendations for another year or 2, wrote the expert panel working group of the NHLBI, “and this was felt to be unacceptable.” The working group, overseen by the National Asthma Education and Prevention Program Coordinating Committee, also acknowledged the update is “not a complete revision” of the 2007 guidelines.
The update provides an evidenced-based review of six key topics in asthma care, as Mary Cataletto, MD, FCCP, professor of pediatrics at New York University Long Island, Mineola, pointed out: use of FeNO, indoor allergen mitigation, use of intermittent ICS and LAMA for asthma, role of subcutaneous and sublingual immunotherapy in the treatment of allergic asthma, and the use of bronchial thermoplasty.
“It has been 13 years since the last update and substantial progress has been made since then in understanding how to best treat children and adults with asthma,” said working group member Michael Schatz, MD, MS, FCCP, an allergy specialist at Kaiser Permanente Medical Center in San Diego.
According to Dr. Schatz, the most important updated recommendations are:
- Conditional recommendation for the use of ICS in children aged infant to 4 years with recurrent wheezing with respiratory infections.
- Use of combination ICS-formoterol for maintenance and to relieve flares in patients with moderate to severe asthma.
- Addition of the LAMA inhaled bronchodilator as add-on therapy for severe asthma not controlled by long-acting beta-agonist (LABA)/ICS combination medications.
Another important update, Dr. Cataletto said, is “shared decision-making among members of asthma teams in order to improve asthma care across all age groups.”
In all, the update includes 19 recommendations in the six subject areas. Each recommendation is notated with two values: its strength, either strong or conditional, and the certainty of evidence behind it, either low, moderate, or high. For example, the recommendation for ICS in young children that Dr. Schatz referred to has a conditional strength of recommendation with moderate certainty of evidence.
Using the GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) methodology to determine strength of recommendation is a notable innovation of the latest guidelines, Dr. Busse noted.
Recommendations (strength of recommendation/certainty of evidence) include:
- Use of FeNO in children and adults when the asthma diagnosis is uncertain (conditional/moderate) or in those with allergic asthma and an uncertain course of management (conditional/low).
- Avoid standalone FeNO to evaluate asthma control or the likelihood or severity of future exacerbations, or for in infants to 4-year-olds with recurrent wheezing (strong/low for both).
- Avoid allergen mitigation in routine asthma management for patients who don’t have sensitivity to specific indoor allergens (conditional/low).
- Multicomponent allergen-specific mitigation when specific allergen sensitivity has been identified and pest management alone for symptoms related to specific pest exposure (conditional/low for both).
- Impermeable bedding covers should be part of a multicomponent mitigation strategy, not as a standalone tool, for patients with asthma and dust mite sensitivity (conditional/moderate).
- Daily ICS at onset of a respiratory tract infection along with as-needed short-acting beta-agonists in children aged 4 years and younger with recurrent wheezing but no wheezing between infections rather than as-needed standalone SABA (conditional/high).
- For adults and children aged 12 years and older with mild persistent asthma, either daily low-dose ICS with as-needed SABA or as-needed ICS and SABA concomitantly (conditional/moderate).
- Avoid short-course increased ICS dosing for patients aged 4 years and older with good adherence to daily ICS therapy (conditional/low).
- For patients aged 4 years and older with moderate to severe persistent asthma, a preference for combined ICS-formoterol inhaler over higher dose ICS daily and intermittent SABA or daily ICS-LABA with intermittent SABA (strong/high [aged 12 years and older]; moderate [aged 4-11 years]).
- A preference for combined ICS-formoterol for both daily and relief therapy for patients 12 years and older with severe persistent asthma over higher-dose ICS-LABA daily and intermittent SABA (conditional/high).
- A preference for adding LABA rather than LAMA to ICS in patients aged 12 years and older with uncontrolled persistent asthma (conditional/moderate).
- If LABA isn’t used, add LAMA to ICS in patients aged 12 years and older with uncontrolled persistent asthma rather than continuing the same dose of ICS alone (conditional/moderate).
- In those same patients already on combined ICS-LABA therapy, add LAMA rather than continuing the same dose of ICS-LABA (conditional/moderate).
- Use subcutaneous immunotherapy as a potential adjunct to standard drug therapy in patients aged 5 years and older with mild to moderate allergic asthma when their asthma is controlled on immunotherapy (conditional/moderate).
- Avoid sublingual immunotherapy in patients with persistent allergic asthma (conditional/moderate).
- Avoid bronchial thermoplasty in those 18 years and older with persistent asthma, but consider it in patients who can accept the short-term worsening symptoms or unknown long-term side effects in exchange for the potential benefits (conditional/moderate).
One of the key elements of the guidelines is the use of the SMART (single maintenance and reliever therapy) approach to evaluate the comparative effectiveness of intermittent ICS with formoterol, Dr. Busse noted. “I think that’s a very significant advance. The literature is replete with evidence to support this. Secondly, it really makes life convenient for patients; you have one inhaler.”
The recommendation on SABA use is also significant, Dr. Busse said. “Data have emerged to suggest that if you’re having a need for one of these rescue medications, it’s due to an increase in inflammation in the lower airway, and you want to give an ICS which will act on the inflammation along with the bronchodilator. That’s a new concept, and it’s a very significant step forward.”
Dr. Schatz disclosed financial relationships with Merck, Teva, and ALK-Abello, but was recused from the writing, discussion, and voting related to the immunotherapy recommendation. Dr. Cataletto and Dr. Busse have no relevant relationships to disclose.
SOURCE: Schatz M et al. J Allergy Clin Immunol. 2020;146:1217-70.
The updated guidelines address six priority topics, including refined recommendations for the use of fractional exhaled nitric oxide (FeNO) testing, intermittent inhaled corticosteroids (ICS), long-acting muscarinic antagonists (LAMA), and bronchial thermoplasty, but notably exclude any recommendations for the use of fast-emerging biological therapy.
“Biological therapy is the major step forward,” said William W. Busse, MD, professor of allergy and immunology at the University of Wisconsin–Madison, and lead author of the previous guidelines (Bethesda, Md.: NHLBI, 2007). “It wasn’t within the scope of work, so it’s not a criticism, but it is an important shortcoming,” he said. The omission identifies the need for the next update. “This is an area that has to be dealt with,” Dr. Busse stated in an interview.
Including biologic agents would have delayed the release of the recommendations for another year or 2, wrote the expert panel working group of the NHLBI, “and this was felt to be unacceptable.” The working group, overseen by the National Asthma Education and Prevention Program Coordinating Committee, also acknowledged the update is “not a complete revision” of the 2007 guidelines.
The update provides an evidenced-based review of six key topics in asthma care, as Mary Cataletto, MD, FCCP, professor of pediatrics at New York University Long Island, Mineola, pointed out: use of FeNO, indoor allergen mitigation, use of intermittent ICS and LAMA for asthma, role of subcutaneous and sublingual immunotherapy in the treatment of allergic asthma, and the use of bronchial thermoplasty.
“It has been 13 years since the last update and substantial progress has been made since then in understanding how to best treat children and adults with asthma,” said working group member Michael Schatz, MD, MS, FCCP, an allergy specialist at Kaiser Permanente Medical Center in San Diego.
According to Dr. Schatz, the most important updated recommendations are:
- Conditional recommendation for the use of ICS in children aged infant to 4 years with recurrent wheezing with respiratory infections.
- Use of combination ICS-formoterol for maintenance and to relieve flares in patients with moderate to severe asthma.
- Addition of the LAMA inhaled bronchodilator as add-on therapy for severe asthma not controlled by long-acting beta-agonist (LABA)/ICS combination medications.
Another important update, Dr. Cataletto said, is “shared decision-making among members of asthma teams in order to improve asthma care across all age groups.”
In all, the update includes 19 recommendations in the six subject areas. Each recommendation is notated with two values: its strength, either strong or conditional, and the certainty of evidence behind it, either low, moderate, or high. For example, the recommendation for ICS in young children that Dr. Schatz referred to has a conditional strength of recommendation with moderate certainty of evidence.
Using the GRADE (Grading of Recommendations, Assessment, Development, and Evaluation) methodology to determine strength of recommendation is a notable innovation of the latest guidelines, Dr. Busse noted.
Recommendations (strength of recommendation/certainty of evidence) include:
- Use of FeNO in children and adults when the asthma diagnosis is uncertain (conditional/moderate) or in those with allergic asthma and an uncertain course of management (conditional/low).
- Avoid standalone FeNO to evaluate asthma control or the likelihood or severity of future exacerbations, or for in infants to 4-year-olds with recurrent wheezing (strong/low for both).
- Avoid allergen mitigation in routine asthma management for patients who don’t have sensitivity to specific indoor allergens (conditional/low).
- Multicomponent allergen-specific mitigation when specific allergen sensitivity has been identified and pest management alone for symptoms related to specific pest exposure (conditional/low for both).
- Impermeable bedding covers should be part of a multicomponent mitigation strategy, not as a standalone tool, for patients with asthma and dust mite sensitivity (conditional/moderate).
- Daily ICS at onset of a respiratory tract infection along with as-needed short-acting beta-agonists in children aged 4 years and younger with recurrent wheezing but no wheezing between infections rather than as-needed standalone SABA (conditional/high).
- For adults and children aged 12 years and older with mild persistent asthma, either daily low-dose ICS with as-needed SABA or as-needed ICS and SABA concomitantly (conditional/moderate).
- Avoid short-course increased ICS dosing for patients aged 4 years and older with good adherence to daily ICS therapy (conditional/low).
- For patients aged 4 years and older with moderate to severe persistent asthma, a preference for combined ICS-formoterol inhaler over higher dose ICS daily and intermittent SABA or daily ICS-LABA with intermittent SABA (strong/high [aged 12 years and older]; moderate [aged 4-11 years]).
- A preference for combined ICS-formoterol for both daily and relief therapy for patients 12 years and older with severe persistent asthma over higher-dose ICS-LABA daily and intermittent SABA (conditional/high).
- A preference for adding LABA rather than LAMA to ICS in patients aged 12 years and older with uncontrolled persistent asthma (conditional/moderate).
- If LABA isn’t used, add LAMA to ICS in patients aged 12 years and older with uncontrolled persistent asthma rather than continuing the same dose of ICS alone (conditional/moderate).
- In those same patients already on combined ICS-LABA therapy, add LAMA rather than continuing the same dose of ICS-LABA (conditional/moderate).
- Use subcutaneous immunotherapy as a potential adjunct to standard drug therapy in patients aged 5 years and older with mild to moderate allergic asthma when their asthma is controlled on immunotherapy (conditional/moderate).
- Avoid sublingual immunotherapy in patients with persistent allergic asthma (conditional/moderate).
- Avoid bronchial thermoplasty in those 18 years and older with persistent asthma, but consider it in patients who can accept the short-term worsening symptoms or unknown long-term side effects in exchange for the potential benefits (conditional/moderate).
One of the key elements of the guidelines is the use of the SMART (single maintenance and reliever therapy) approach to evaluate the comparative effectiveness of intermittent ICS with formoterol, Dr. Busse noted. “I think that’s a very significant advance. The literature is replete with evidence to support this. Secondly, it really makes life convenient for patients; you have one inhaler.”
The recommendation on SABA use is also significant, Dr. Busse said. “Data have emerged to suggest that if you’re having a need for one of these rescue medications, it’s due to an increase in inflammation in the lower airway, and you want to give an ICS which will act on the inflammation along with the bronchodilator. That’s a new concept, and it’s a very significant step forward.”
Dr. Schatz disclosed financial relationships with Merck, Teva, and ALK-Abello, but was recused from the writing, discussion, and voting related to the immunotherapy recommendation. Dr. Cataletto and Dr. Busse have no relevant relationships to disclose.
SOURCE: Schatz M et al. J Allergy Clin Immunol. 2020;146:1217-70.
FROM THE JOURNAL OF ALLERGY AND CLINICAL IMMUNOLOGY
GI physicians urge COVID-19 vaccines for all IBD patients
Gastroenterologists at the Advances in Inflammatory Bowel Disease 2020 annual meeting said they will strongly advise their patients to take the COVID-19 vaccines as they become available.
Announcement that the first vaccine, Pfizer’s, was recommended for emergency use authorization came in the middle of AIBD’s Thursday evening COVID-19 session.
Miguel Regueiro, MD, chair of the department of gastroenterology, hepatology, and nutrition at Cleveland Clinic in Ohio, said, “We’re uniformly recommending this to all our patients.”
“The [vaccines] leading the pack do not have any replicating virus and thus can be used in immunocompromised people,” Maria Abreu, MD, director of the Crohn’s & colitis center at the University of Miami, told this news organization. “Although it is true that we don’t know – and won’t know for a while – whether the high levels of efficacy seen with the mRNA vaccines so far will be achieved in patients who are immunocompromised, there is every reason to believe that [the vaccine] will still be enough to protect them from complications of COVID-19.”
The bottom line, she said, is that “it’s much safer to get a vaccine than it is to take your chances of getting COVID-19.”
David T. Rubin, MD, chief of gastroenterology, hepatology, and nutrition at UChicago Medicine, said in a session earlier in the day, “Emerging information about the messenger RNA looks like it’s going to be safe for our population, but of course we want to see more. Messenger RNA degrades within days of giving it, so it’s not expected to linger or generate any other problems we can think of.”
Dr. Abreu said there’s no evidence that inflammatory bowel disease (IBD) patients are more susceptible to COVID-19 infection even though the entry molecules are expressed in the GI tract. “They are really not differentially expressed in IBD and, if anything, some of our more potent therapies reduce the expression of these molecules in the GI tract,” she said.
Regarding how IBD medications affect outcomes if patients are infected with COVID-19, Dr. Abreu pointed out that corticosteroids seem to be associated with worse outcomes. “I would posit that it has to do with initially allowing there to be a lot of very rapid viral replication,” she said.
And she also noted that any of the mainstay drugs for IBD – the anti–tumor necrosis factor (TNF) therapies – are showing promise as treatments for COVID-19.
Updates from the IBD-COVID-19 registry
Michael Kappelman, MD, MPH, from the University of North Carolina at Chapel Hill said information from the Secure-IBD registry, which collects real-time global information on how COVID-19 affects IBD patients, suggests that these patients “may have a more severe course than the general population, but not by much.”
He reported the registry had logged more than 3,300 reported COVID-19 cases among IBD patients from 62 countries.
Registry outcomes through the end of November have found a mean age of reported cases of 40 years, and that 21% of patients were hospitalized with an average length of stay of 10.2 days, 4% required intensive care unit admission, and 2% died.
The majority of the deaths reported to Secure-IBD occurred in patients older than 60 years, Dr. Kappelman said, adding that the hospitalizations and death rates in IBD patients with no comorbidities were relatively low.
“My belief is that available data are actually more reassuring than alarming,” he said.
Dr. Kappelman and other investigators found that combination therapy that includes thiopurines and thiopurine monotherapy are “associated with about a fourfold risk of the requirement for intensive care or mortality from COVID,” compared with anti-TNF monotherapy.
In cases reported to Secure-IBD, about 25% of IBD patients with COVID-19 developed new GI symptoms, primarily diarrhea and abdominal pain, he said.
In his practice, Dr. Kappelman said, he minimizes use of steroids and has found that COVID-19 adds a reason to favor anti-TNF over 6-mercaptopurine (6-MP) plus azathioprine.
He also advises “a high alert for COVID-19 in patients with new GI symptoms.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Rubin has served as a director, officer, partner, employee, adviser, consultant, or trustee for AbbVie, Abgenomics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dizal Pharmaceuticals, GalenPharma/Atlantica, Genentech/Roche, Gilead Sciences, Ichnos Sciences, GlaxoSmithKline, Janssen, Eli Lilly, Pfizer, Prometheus Laboratories, Reistone, Shire, Takeda Pharmaceutical, and Techlab. In addition, he has received research grants from AbbVie, Genentech/Roche, Janssen Pharmaceuticals, Prometheus Laboratories, Shire, and Takeda Pharmaceutical Company; and holds stock options in Abgenomics and Biomica. Regueiro and Kappelman have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Gastroenterologists at the Advances in Inflammatory Bowel Disease 2020 annual meeting said they will strongly advise their patients to take the COVID-19 vaccines as they become available.
Announcement that the first vaccine, Pfizer’s, was recommended for emergency use authorization came in the middle of AIBD’s Thursday evening COVID-19 session.
Miguel Regueiro, MD, chair of the department of gastroenterology, hepatology, and nutrition at Cleveland Clinic in Ohio, said, “We’re uniformly recommending this to all our patients.”
“The [vaccines] leading the pack do not have any replicating virus and thus can be used in immunocompromised people,” Maria Abreu, MD, director of the Crohn’s & colitis center at the University of Miami, told this news organization. “Although it is true that we don’t know – and won’t know for a while – whether the high levels of efficacy seen with the mRNA vaccines so far will be achieved in patients who are immunocompromised, there is every reason to believe that [the vaccine] will still be enough to protect them from complications of COVID-19.”
The bottom line, she said, is that “it’s much safer to get a vaccine than it is to take your chances of getting COVID-19.”
David T. Rubin, MD, chief of gastroenterology, hepatology, and nutrition at UChicago Medicine, said in a session earlier in the day, “Emerging information about the messenger RNA looks like it’s going to be safe for our population, but of course we want to see more. Messenger RNA degrades within days of giving it, so it’s not expected to linger or generate any other problems we can think of.”
Dr. Abreu said there’s no evidence that inflammatory bowel disease (IBD) patients are more susceptible to COVID-19 infection even though the entry molecules are expressed in the GI tract. “They are really not differentially expressed in IBD and, if anything, some of our more potent therapies reduce the expression of these molecules in the GI tract,” she said.
Regarding how IBD medications affect outcomes if patients are infected with COVID-19, Dr. Abreu pointed out that corticosteroids seem to be associated with worse outcomes. “I would posit that it has to do with initially allowing there to be a lot of very rapid viral replication,” she said.
And she also noted that any of the mainstay drugs for IBD – the anti–tumor necrosis factor (TNF) therapies – are showing promise as treatments for COVID-19.
Updates from the IBD-COVID-19 registry
Michael Kappelman, MD, MPH, from the University of North Carolina at Chapel Hill said information from the Secure-IBD registry, which collects real-time global information on how COVID-19 affects IBD patients, suggests that these patients “may have a more severe course than the general population, but not by much.”
He reported the registry had logged more than 3,300 reported COVID-19 cases among IBD patients from 62 countries.
Registry outcomes through the end of November have found a mean age of reported cases of 40 years, and that 21% of patients were hospitalized with an average length of stay of 10.2 days, 4% required intensive care unit admission, and 2% died.
The majority of the deaths reported to Secure-IBD occurred in patients older than 60 years, Dr. Kappelman said, adding that the hospitalizations and death rates in IBD patients with no comorbidities were relatively low.
“My belief is that available data are actually more reassuring than alarming,” he said.
Dr. Kappelman and other investigators found that combination therapy that includes thiopurines and thiopurine monotherapy are “associated with about a fourfold risk of the requirement for intensive care or mortality from COVID,” compared with anti-TNF monotherapy.
In cases reported to Secure-IBD, about 25% of IBD patients with COVID-19 developed new GI symptoms, primarily diarrhea and abdominal pain, he said.
In his practice, Dr. Kappelman said, he minimizes use of steroids and has found that COVID-19 adds a reason to favor anti-TNF over 6-mercaptopurine (6-MP) plus azathioprine.
He also advises “a high alert for COVID-19 in patients with new GI symptoms.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Rubin has served as a director, officer, partner, employee, adviser, consultant, or trustee for AbbVie, Abgenomics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dizal Pharmaceuticals, GalenPharma/Atlantica, Genentech/Roche, Gilead Sciences, Ichnos Sciences, GlaxoSmithKline, Janssen, Eli Lilly, Pfizer, Prometheus Laboratories, Reistone, Shire, Takeda Pharmaceutical, and Techlab. In addition, he has received research grants from AbbVie, Genentech/Roche, Janssen Pharmaceuticals, Prometheus Laboratories, Shire, and Takeda Pharmaceutical Company; and holds stock options in Abgenomics and Biomica. Regueiro and Kappelman have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Gastroenterologists at the Advances in Inflammatory Bowel Disease 2020 annual meeting said they will strongly advise their patients to take the COVID-19 vaccines as they become available.
Announcement that the first vaccine, Pfizer’s, was recommended for emergency use authorization came in the middle of AIBD’s Thursday evening COVID-19 session.
Miguel Regueiro, MD, chair of the department of gastroenterology, hepatology, and nutrition at Cleveland Clinic in Ohio, said, “We’re uniformly recommending this to all our patients.”
“The [vaccines] leading the pack do not have any replicating virus and thus can be used in immunocompromised people,” Maria Abreu, MD, director of the Crohn’s & colitis center at the University of Miami, told this news organization. “Although it is true that we don’t know – and won’t know for a while – whether the high levels of efficacy seen with the mRNA vaccines so far will be achieved in patients who are immunocompromised, there is every reason to believe that [the vaccine] will still be enough to protect them from complications of COVID-19.”
The bottom line, she said, is that “it’s much safer to get a vaccine than it is to take your chances of getting COVID-19.”
David T. Rubin, MD, chief of gastroenterology, hepatology, and nutrition at UChicago Medicine, said in a session earlier in the day, “Emerging information about the messenger RNA looks like it’s going to be safe for our population, but of course we want to see more. Messenger RNA degrades within days of giving it, so it’s not expected to linger or generate any other problems we can think of.”
Dr. Abreu said there’s no evidence that inflammatory bowel disease (IBD) patients are more susceptible to COVID-19 infection even though the entry molecules are expressed in the GI tract. “They are really not differentially expressed in IBD and, if anything, some of our more potent therapies reduce the expression of these molecules in the GI tract,” she said.
Regarding how IBD medications affect outcomes if patients are infected with COVID-19, Dr. Abreu pointed out that corticosteroids seem to be associated with worse outcomes. “I would posit that it has to do with initially allowing there to be a lot of very rapid viral replication,” she said.
And she also noted that any of the mainstay drugs for IBD – the anti–tumor necrosis factor (TNF) therapies – are showing promise as treatments for COVID-19.
Updates from the IBD-COVID-19 registry
Michael Kappelman, MD, MPH, from the University of North Carolina at Chapel Hill said information from the Secure-IBD registry, which collects real-time global information on how COVID-19 affects IBD patients, suggests that these patients “may have a more severe course than the general population, but not by much.”
He reported the registry had logged more than 3,300 reported COVID-19 cases among IBD patients from 62 countries.
Registry outcomes through the end of November have found a mean age of reported cases of 40 years, and that 21% of patients were hospitalized with an average length of stay of 10.2 days, 4% required intensive care unit admission, and 2% died.
The majority of the deaths reported to Secure-IBD occurred in patients older than 60 years, Dr. Kappelman said, adding that the hospitalizations and death rates in IBD patients with no comorbidities were relatively low.
“My belief is that available data are actually more reassuring than alarming,” he said.
Dr. Kappelman and other investigators found that combination therapy that includes thiopurines and thiopurine monotherapy are “associated with about a fourfold risk of the requirement for intensive care or mortality from COVID,” compared with anti-TNF monotherapy.
In cases reported to Secure-IBD, about 25% of IBD patients with COVID-19 developed new GI symptoms, primarily diarrhea and abdominal pain, he said.
In his practice, Dr. Kappelman said, he minimizes use of steroids and has found that COVID-19 adds a reason to favor anti-TNF over 6-mercaptopurine (6-MP) plus azathioprine.
He also advises “a high alert for COVID-19 in patients with new GI symptoms.”
Dr. Abreu has relationships with Boehringer Ingelheim, Cosmo Biopharma, Eli Lilly, Gilead, Janssen, Landos Biopharma, Prometheus Bioscience, Takeda, UCB Biopharma, Pfizer, and Prometheus Laboratories. Dr. Rubin has served as a director, officer, partner, employee, adviser, consultant, or trustee for AbbVie, Abgenomics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dizal Pharmaceuticals, GalenPharma/Atlantica, Genentech/Roche, Gilead Sciences, Ichnos Sciences, GlaxoSmithKline, Janssen, Eli Lilly, Pfizer, Prometheus Laboratories, Reistone, Shire, Takeda Pharmaceutical, and Techlab. In addition, he has received research grants from AbbVie, Genentech/Roche, Janssen Pharmaceuticals, Prometheus Laboratories, Shire, and Takeda Pharmaceutical Company; and holds stock options in Abgenomics and Biomica. Regueiro and Kappelman have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
COVID-related harm to HCWs must be tracked more rigorously: NAS panel
A panel of scientific experts is urging the nation to do more to track morbidity and mortality among health care workers (HCWs), given the large and disproportionate number who have been infected with or died from SARS-CoV-2.
The National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats issued a 10-page “rapid expert consultation” on what is known about deaths and mental health problems among HCWs associated with the COVID-19 pandemic and how to protect workers.
“The absence of a uniform national framework and inconsistent requirements across states for collecting, recording, and reporting HCW mortality and morbidity data associated with COVID-19 impairs anyone’s ability to make comparisons, do combined analyses, or draw conclusions about the scale of the problem,” says the panel in the report.
Mental health, in particular, needs to be examined, it says. Although the data are still limited, the prevalence of burnout and suicide “points to a serious concern,” according to the report.
“As with mortality due to COVID-19, there are currently no national systems nor reporting standards for morbidity measures related to the pandemic, such as mental health status, provider well-being, and other psychological effects on HCWs,” the report says.
A more robust national system that collected data on circumstances and interventions that may raise or lower risk, as well as on where the infection occurred, “would support the adoption of effective mitigation strategies,” says the report. It would also facilitate epidemiologic studies on risk factors, such as face-to-face contact with COVID-19 patients and the availability and use of personal protective equipment (PPE). Studies could also examine the impact of institutional requirements for masking.
Studies have consistently shown that universal mask wearing and access to appropriate PPE support the physical safety and mental health of HCWs, says the report.
Track scale of crisis
The committee cited many gaps in the current system. The Occupational Safety and Health Administration, for instance, doesn’t count deaths from occupationally acquired infection. Many states don’t report COVID-19 deaths by profession. The Centers for Disease Control and Prevention (CDC) relies on case report forms from local health departments for all COVID-19 cases, which typically are lacking in specifics, such as occupation or job setting, says the committee’s report.
As of Nov. 3, the CDC had reported 786 deaths among HCWs that were attributable to COVID-19 – a far lower number than other sources have reported.
The committee notes that much could be done immediately. A National Academy of Medicine panel on clinician well-being and resilience in August recommended that the CDC establish a national epidemiologic tracking program to measure HCWs’ well-being, assess the acute and long-term effects of COVID-19 on those workers, and report on the outcomes of interventions.
Such a program “is needed to comprehensively acknowledge the scale of the COVID-19 crisis and protect the health care workforce that is already stretched to the breaking point in many locations,” the committee says in its report.
A version of this article originally appeared on Medscape.com.
A panel of scientific experts is urging the nation to do more to track morbidity and mortality among health care workers (HCWs), given the large and disproportionate number who have been infected with or died from SARS-CoV-2.
The National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats issued a 10-page “rapid expert consultation” on what is known about deaths and mental health problems among HCWs associated with the COVID-19 pandemic and how to protect workers.
“The absence of a uniform national framework and inconsistent requirements across states for collecting, recording, and reporting HCW mortality and morbidity data associated with COVID-19 impairs anyone’s ability to make comparisons, do combined analyses, or draw conclusions about the scale of the problem,” says the panel in the report.
Mental health, in particular, needs to be examined, it says. Although the data are still limited, the prevalence of burnout and suicide “points to a serious concern,” according to the report.
“As with mortality due to COVID-19, there are currently no national systems nor reporting standards for morbidity measures related to the pandemic, such as mental health status, provider well-being, and other psychological effects on HCWs,” the report says.
A more robust national system that collected data on circumstances and interventions that may raise or lower risk, as well as on where the infection occurred, “would support the adoption of effective mitigation strategies,” says the report. It would also facilitate epidemiologic studies on risk factors, such as face-to-face contact with COVID-19 patients and the availability and use of personal protective equipment (PPE). Studies could also examine the impact of institutional requirements for masking.
Studies have consistently shown that universal mask wearing and access to appropriate PPE support the physical safety and mental health of HCWs, says the report.
Track scale of crisis
The committee cited many gaps in the current system. The Occupational Safety and Health Administration, for instance, doesn’t count deaths from occupationally acquired infection. Many states don’t report COVID-19 deaths by profession. The Centers for Disease Control and Prevention (CDC) relies on case report forms from local health departments for all COVID-19 cases, which typically are lacking in specifics, such as occupation or job setting, says the committee’s report.
As of Nov. 3, the CDC had reported 786 deaths among HCWs that were attributable to COVID-19 – a far lower number than other sources have reported.
The committee notes that much could be done immediately. A National Academy of Medicine panel on clinician well-being and resilience in August recommended that the CDC establish a national epidemiologic tracking program to measure HCWs’ well-being, assess the acute and long-term effects of COVID-19 on those workers, and report on the outcomes of interventions.
Such a program “is needed to comprehensively acknowledge the scale of the COVID-19 crisis and protect the health care workforce that is already stretched to the breaking point in many locations,” the committee says in its report.
A version of this article originally appeared on Medscape.com.
A panel of scientific experts is urging the nation to do more to track morbidity and mortality among health care workers (HCWs), given the large and disproportionate number who have been infected with or died from SARS-CoV-2.
The National Academies of Sciences, Engineering, and Medicine’s Standing Committee on Emerging Infectious Diseases and 21st Century Health Threats issued a 10-page “rapid expert consultation” on what is known about deaths and mental health problems among HCWs associated with the COVID-19 pandemic and how to protect workers.
“The absence of a uniform national framework and inconsistent requirements across states for collecting, recording, and reporting HCW mortality and morbidity data associated with COVID-19 impairs anyone’s ability to make comparisons, do combined analyses, or draw conclusions about the scale of the problem,” says the panel in the report.
Mental health, in particular, needs to be examined, it says. Although the data are still limited, the prevalence of burnout and suicide “points to a serious concern,” according to the report.
“As with mortality due to COVID-19, there are currently no national systems nor reporting standards for morbidity measures related to the pandemic, such as mental health status, provider well-being, and other psychological effects on HCWs,” the report says.
A more robust national system that collected data on circumstances and interventions that may raise or lower risk, as well as on where the infection occurred, “would support the adoption of effective mitigation strategies,” says the report. It would also facilitate epidemiologic studies on risk factors, such as face-to-face contact with COVID-19 patients and the availability and use of personal protective equipment (PPE). Studies could also examine the impact of institutional requirements for masking.
Studies have consistently shown that universal mask wearing and access to appropriate PPE support the physical safety and mental health of HCWs, says the report.
Track scale of crisis
The committee cited many gaps in the current system. The Occupational Safety and Health Administration, for instance, doesn’t count deaths from occupationally acquired infection. Many states don’t report COVID-19 deaths by profession. The Centers for Disease Control and Prevention (CDC) relies on case report forms from local health departments for all COVID-19 cases, which typically are lacking in specifics, such as occupation or job setting, says the committee’s report.
As of Nov. 3, the CDC had reported 786 deaths among HCWs that were attributable to COVID-19 – a far lower number than other sources have reported.
The committee notes that much could be done immediately. A National Academy of Medicine panel on clinician well-being and resilience in August recommended that the CDC establish a national epidemiologic tracking program to measure HCWs’ well-being, assess the acute and long-term effects of COVID-19 on those workers, and report on the outcomes of interventions.
Such a program “is needed to comprehensively acknowledge the scale of the COVID-19 crisis and protect the health care workforce that is already stretched to the breaking point in many locations,” the committee says in its report.
A version of this article originally appeared on Medscape.com.
Coronavirus has infected over 2% of U.S. children
After last week’s ever-so-slightly positive news, the COVID-19 numbers in children have gone back to their old ways.
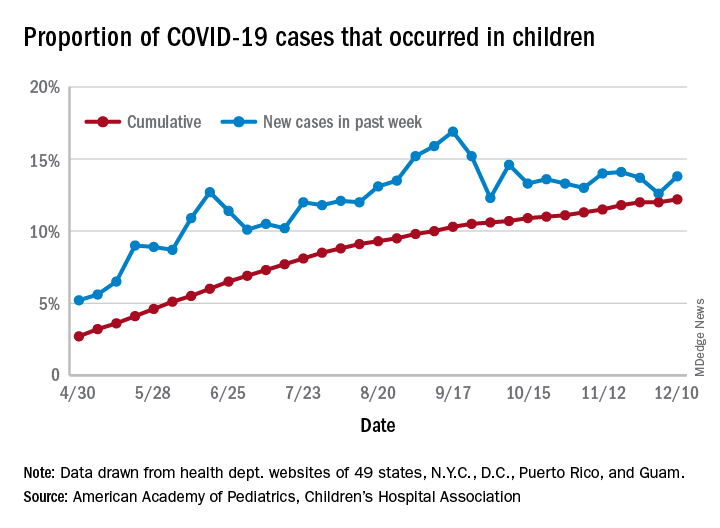
For the week ending Dec. 10, there were 178,823 new COVID-19 cases reported in U.S. children, the highest weekly total yet during the pandemic. The number of new cases had dropped the week before after setting a new high of almost 154,000 during the last full week of November, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
A new weekly high has been seen in 9 of the last 10 weeks, during which time the weekly total of child cases has gone from just over 40,000 (week ending Oct. 8) to almost 179,000, the two organizations said.
and that 2.1% of all children (2,179 per 100,000) in the United States have been infected with the coronavirus, the AAP and CHA said in their weekly report, which includes health department data from 49 states (New York does not report age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
The cumulative proportion of 12.2% has been exceeded in 27 states, as well as Puerto Rico and Guam, with the highest coming in Wyoming (21.3%), South Carolina (18.1%), and Tennessee (18.1%) and the lowest in Florida (6.7%, but the state uses an age range of 0-14 years) and New Jersey (7.6%), the AAP/CHA data show.
In a separate statement, AAP president Sally Goza, MD, welcomed the approval of the Pfizer-BioNTech COVID-19 vaccine but noted that the “virus is at unprecedented levels in nearly every community in the U.S., and in many areas, our health care system is terribly overburdened. The vaccine will not solve this overnight. I urge everyone to continue to practice social distancing, and wear masks or cloth face coverings, and get a flu shot, so we can protect the people we care about.”
Dr. Goza continued: “We applaud Pfizer-BioNTech for including children ages 12 through 17 in their clinical trials and we look forward to learning more about the data from children aged 12-15. We also want to acknowledge the discussion during the committee meeting on including 16- to 17-year-olds in the EUA [emergency-use authorization]. We believe that discussion underscores the need to keep expanding these trials to the pediatric population so we can collect robust data on this age group.”
[email protected]
After last week’s ever-so-slightly positive news, the COVID-19 numbers in children have gone back to their old ways.

For the week ending Dec. 10, there were 178,823 new COVID-19 cases reported in U.S. children, the highest weekly total yet during the pandemic. The number of new cases had dropped the week before after setting a new high of almost 154,000 during the last full week of November, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
A new weekly high has been seen in 9 of the last 10 weeks, during which time the weekly total of child cases has gone from just over 40,000 (week ending Oct. 8) to almost 179,000, the two organizations said.
and that 2.1% of all children (2,179 per 100,000) in the United States have been infected with the coronavirus, the AAP and CHA said in their weekly report, which includes health department data from 49 states (New York does not report age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
The cumulative proportion of 12.2% has been exceeded in 27 states, as well as Puerto Rico and Guam, with the highest coming in Wyoming (21.3%), South Carolina (18.1%), and Tennessee (18.1%) and the lowest in Florida (6.7%, but the state uses an age range of 0-14 years) and New Jersey (7.6%), the AAP/CHA data show.
In a separate statement, AAP president Sally Goza, MD, welcomed the approval of the Pfizer-BioNTech COVID-19 vaccine but noted that the “virus is at unprecedented levels in nearly every community in the U.S., and in many areas, our health care system is terribly overburdened. The vaccine will not solve this overnight. I urge everyone to continue to practice social distancing, and wear masks or cloth face coverings, and get a flu shot, so we can protect the people we care about.”
Dr. Goza continued: “We applaud Pfizer-BioNTech for including children ages 12 through 17 in their clinical trials and we look forward to learning more about the data from children aged 12-15. We also want to acknowledge the discussion during the committee meeting on including 16- to 17-year-olds in the EUA [emergency-use authorization]. We believe that discussion underscores the need to keep expanding these trials to the pediatric population so we can collect robust data on this age group.”
[email protected]
After last week’s ever-so-slightly positive news, the COVID-19 numbers in children have gone back to their old ways.

For the week ending Dec. 10, there were 178,823 new COVID-19 cases reported in U.S. children, the highest weekly total yet during the pandemic. The number of new cases had dropped the week before after setting a new high of almost 154,000 during the last full week of November, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
A new weekly high has been seen in 9 of the last 10 weeks, during which time the weekly total of child cases has gone from just over 40,000 (week ending Oct. 8) to almost 179,000, the two organizations said.
and that 2.1% of all children (2,179 per 100,000) in the United States have been infected with the coronavirus, the AAP and CHA said in their weekly report, which includes health department data from 49 states (New York does not report age distribution), the District of Columbia, New York City, Puerto Rico, and Guam.
The cumulative proportion of 12.2% has been exceeded in 27 states, as well as Puerto Rico and Guam, with the highest coming in Wyoming (21.3%), South Carolina (18.1%), and Tennessee (18.1%) and the lowest in Florida (6.7%, but the state uses an age range of 0-14 years) and New Jersey (7.6%), the AAP/CHA data show.
In a separate statement, AAP president Sally Goza, MD, welcomed the approval of the Pfizer-BioNTech COVID-19 vaccine but noted that the “virus is at unprecedented levels in nearly every community in the U.S., and in many areas, our health care system is terribly overburdened. The vaccine will not solve this overnight. I urge everyone to continue to practice social distancing, and wear masks or cloth face coverings, and get a flu shot, so we can protect the people we care about.”
Dr. Goza continued: “We applaud Pfizer-BioNTech for including children ages 12 through 17 in their clinical trials and we look forward to learning more about the data from children aged 12-15. We also want to acknowledge the discussion during the committee meeting on including 16- to 17-year-olds in the EUA [emergency-use authorization]. We believe that discussion underscores the need to keep expanding these trials to the pediatric population so we can collect robust data on this age group.”
[email protected]






