User login
Strategies to identify and prevent penicillin allergy mislabeling and appropriately de-label patients
In North America and Europe, penicillin allergy is the most common drug-allergy label.1 Carrying a penicillin-allergy label, which has recently gained more attention in health care systems, leads to suboptimal outcomes, increased use of broad-spectrum antibiotics, increased risk of adverse reactions, and increased cost of care.2,3 Despite the high rate of reported reactions, clinically significant immunoglobulin E (IgE)-mediated and T cell–mediated hypersensitivity reactions to penicillins are uncommon.2
Through the Choosing Wisely initiative of the American Board of Internal Medicine Foundation, the American Academy of Allergy, Asthma, and Immunology has issued a recommendation: “Don’t overuse non-beta lactam antibiotics in patients with a history of penicillin allergy without an appropriate evaluation.”4 The primary care physician (PCP) plays a critical role in the appropriate evaluation and accurate initial labeling of penicillin allergy. Furthermore, the PCP plays an integral part, in conjunction with the allergist, in removing the “penicillin allergy” label from a patient’s chart when feasible.
The history of penicillin and prevalence of allergy
History. Penicillin, the first antibiotic, was discovered in 1928 by physician and microbiologist Alexander Fleming when he observed that a mold of the Penicillium genus inhibited growth of gram-positive pathogens.5 Along with pharmacologist Howard Florey and chemist Ernst Chain, both of whom assisted in the large-scale isolation and production of the antibiotic, Fleming won the Nobel Prize in Physiology or Medicine in 1945 for this discovery.5
Antibiotics transformed the practice of medicine across a spectrum, including safer childbirth, surgical procedures, and transplantation.6 Penicillin remains first-line therapy for many infections, such as streptococcal pharyngitis,7 and is the only recommended medication for treating syphilis during pregnancy.8 Continued effectiveness of penicillin in these cases allows broad-spectrum antibiotics to be reserved for more severe infections. Regrettably, incorrect antibiotic allergy labeling poses a significant risk to the patient and health care system.
Epidemiology. As with all medications, the potential for anaphylaxis exists after administration of penicillin. Because its use is widespread, penicillin is the most common cause of drug-induced anaphylaxis. However, the incidence of penicillin-induced anaphylaxis is low9: A 1968 World Health Organization report stated that the rate of penicillin anaphylaxis was between 0.015% and 0.04%.10 A more recent study reported an incidence of 1 in 207,191 patients after an oral dose and 1 in 95,298 after a parenteral dose.11 The most common reactions to penicillins are urticaria and delayed maculopapular rash.8
In the United States, the prevalence of reported penicillin allergy is approximately 10% (estimated range, 8% to 12%)3,12-15; among hospitalized patients, that prevalence is estimated to be as high as 15%.13,15 However, the prevalence of confirmed penicillin allergy is low and has decreased over time—demonstrated in a longitudinal study in which the rate of a positive skin test fell from 15% in 1995 to 0.8% in 2013.16,17
Studies have confirmed that as many as 90% of patients who report penicillin allergy are, in fact, able to tolerate penicillins.14,18-20 This finding might be a consequence of initial mislabeling of penicillin allergy; often, adverse reactions are documented as “allergy” when no risk of anaphylaxis exists. Furthermore, patients can outgrow IgE-mediated penicillin allergy because the presence of penicillin IgE antibodies wanes over time.14,15
Continue to: Consequences of mislabeling
Consequences of mislabeling
Clinical consequences. A multitude of clinical consequences result from carrying a “penicillin allergy” label.
Use of broad-spectrum antibiotics leads to increased risk of Clostridium difficile infection and to development of resistant bacteria, such as methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococcus.2,15
Alternative antibiotics used in the setting of a “penicillin allergy” label might be less efficacious and result in suboptimal outcomes. For example, vancomycin is less effective against methicillin-sensitive S aureus bacteremia than nafcillin or cefazolin.2,21 Beta-lactam antibiotics—in particular, cefazolin—are often first-line for perioperative prophylaxis; patients with reported penicillin allergy often receive a less-optimal alternative, such as clindamycin, vancomycin, or gentamicin.22 These patients are at increased risk of surgical site infection.2,22
In addition, using penicillin alternatives can result in greater risk of drug reactions and adverse effects.2
Increased health care costs. Primarily through observational studies, penicillin allergy has been associated with higher health care costs.23 Patients with reported penicillin allergy had, on average, a longer inpatient stay than patients without penicillin allergy, at a 3-year total estimated additional cost of $64.6 million.24 Inpatients with a listed penicillin allergy had direct drug costs ranging from “no difference” to $609 per patient more than patients without a listed penicillin allergy. Outpatient prescription costs were $14 to $193 higher per patient for patients with a listed penicillin allergy.23
Continue to: Considerations in special populations
Considerations in special populations. Evaluating penicillin allergy during routine care is key to decreasing the necessity for urgent penicillin evaluation and possible desensitization at the time of serious infection. Certain patient populations pose specific challenges:
- Pregnant patients. Unverified penicillin allergy during pregnancy is associated with an increased rate of cesarean section and longer postpartum hospitalization.25 Additionally, group B streptococcus-positive women have increased exposure to alternative antibiotics and an increased incidence of adverse drug reactions.25
- Elderly patients. Drug allergy increases with aging.1 Elderly patients in a long-term care facility are more likely to experience adverse drug effects or drug–drug interactions from the use of penicillin alternatives, such as clindamycin, vancomycin, and fluoroquinolones.2
- Oncology patients often require antibiotic prophylaxis as well as treatment for illnesses, such as neutropenic fever, for which beta-lactam antibiotics are often used as initial treatment.2,26
- Other important populations that present specific challenges include hospitalized patients, pediatric patients, and patients with a sexually transmitted infection.2
Active management of a penicillin-allergy label
Greater recognition of the consequences of penicillin allergy in recent years has led to efforts by hospitals and other health care organizations to develop processes by which patients can be successfully de-labeled as part of antibiotic stewardship programs9 and other initiatives. Ideally, every patient who has a “penicillin allergy” label would be referred to an allergist for evaluation; however, the number of allergy specialists is limited, and access to such specialists might be restricted in some areas, making this approach impracticable. Active management of penicillin allergy requires strategies to both test and de-label patients, as well as proactive approaches to prevent incorrect labeling. These proactive approaches require involvement of all members of the health care team—especially PCPs.
Preventing incorrect labeling. PCPs are the most likely to initially label a patient as allergic to penicillin.27 Most physicians rely on a reported history of allergy alone when selecting medication12; once a patient has been labeled “penicillin allergic,” they often retain that mislabel through adulthood.27,28 A qualitative study of PCPs’ views on prescribing penicillin found that many were aware that documented allergies were incorrect but were uncomfortable using their clinical judgment to prescribe a penicillin or change the record, for fear of a future anaphylactic reaction.29 The first step in the case of any reported reaction should be for you to elicit an accurate drug allergy history (TABLE 1).

As with other drug reactions, you should consider the context surrounding the reaction to a penicillin. Take care to review signs and symptoms of the reaction to look for clues that make a true allergic reaction more, or less, likely.
Symptoms can generally be divided into low-risk and high-risk categories27 (TABLE 2). An example of a commonly reported low-risk symptom is diarrhea that develops after several doses of a penicillin. In the absence of other symptoms, this finding is most likely due to elimination of normal gut flora,30 not to an allergic reaction to the medication. Symptoms of intolerance to the medication, such as headache and nausea, are also low risk.27,31 In contrast, immediate onset of abdominal pain after a dose of penicillin and lip or throat swelling are considered high risk.
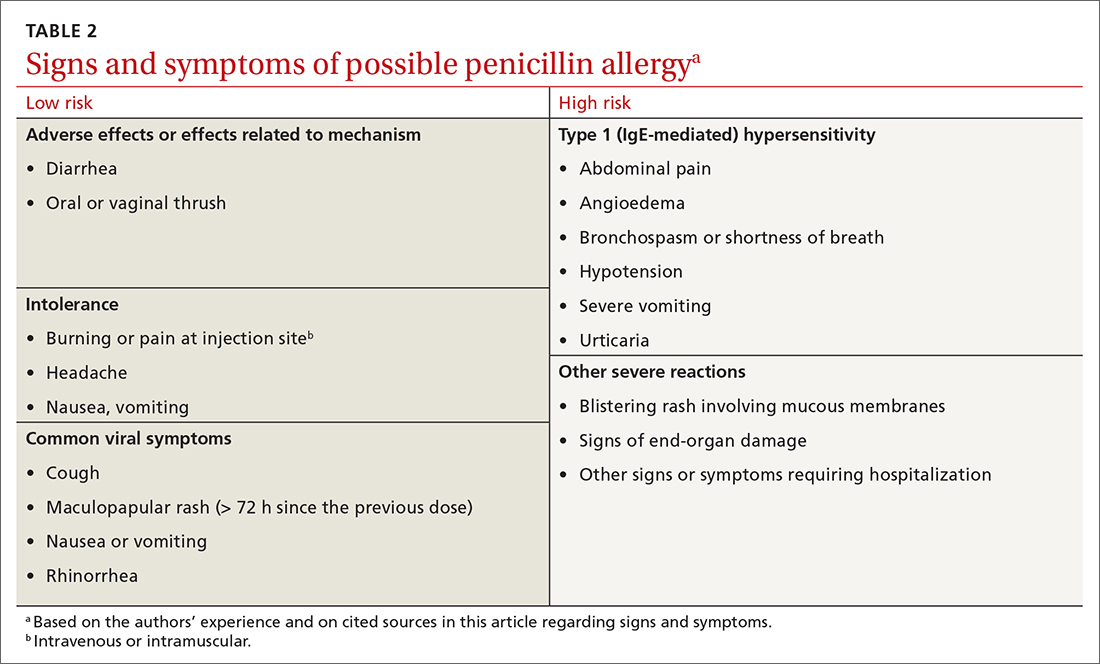
Continue to: Patients presenting with urticaria...
Patients presenting with urticaria or maculopapular rash after taking penicillin are particularly challenging.30 A study of patients in a primary care pediatrics practice found that 7.4% of children receiving a prescription for a penicillin reported a rash.32 Here, timing of onset of symptoms provides some clarity about the likelihood of true allergy. Rashes that manifest during the first hours after exposure are more likely to be IgE mediated, particularly when accompanied by other systemic symptoms; they should be considered high risk. Delayed-onset rashes (> 72 hours after exposure) are usually non-IgE mediated and therefore are generally lower risk,8,30,33 except when associated with certain features, such as mucosal involvement and skin peeling.
Despite acknowledging viral exanthems in the differential, many physicians still label patients presenting with any rash as “allergic.”28 Take care to look for other potential causes of a rash; for example, patients taking amoxicillin who have concurrent Epstein-Barr virus infection frequently develop a maculopapular rash.34 Caubet and colleagues found that 56% of pediatric patients with a history of nonimmediate rash and a negative oral challenge to amoxicillin tested positive for viral infection.28
A family history of penicillin allergy alone should not preclude the use of penicillin.8,27,31 Similarly, if a patient has already received and tolerated a subsequent course of the same penicillin derivative after the initial reaction, the “penicillin allergy” label can be removed. If the reaction history is unknown, refer the patient to an allergist for further evaluation.
Accurate charting is key. With most hospital systems and physician practices now documenting in an electronic health record, there exists the ability to document, in great detail, patients’ reactions to medications. Previous studies have found, however, that such documentation is often done poorly, or not done at all. One such study found that (1) > 20% of patients with a “penicillin allergy” label did not have reaction details listed and (2) when reactions were listed, many were incorrectly labeled as “allergy,” not “intolerance.”35
Many electronic health record systems lump drug allergies, adverse effects, and food and environmental allergies into a single section, leading to a lack of distinction between adverse reactions and true allergy.31 Although many PCPs report that it is easy to change a patient’s allergy label in the record,29 more often, a nurse, resident, or consultant actually documents the reaction.35
Continue to: Documentation at the time of the reaction...
Documentation at the time of the reaction, within the encounter note and the allergy tab, is essential, so that other physicians caring for the patient, in the future, will be knowledgeable about the details of the reaction. Make it your responsibility to accurately document penicillin allergy in patients’ charts, including removing the “penicillin allergy” label from the chart of patients whose history is inconsistent with allergy, who have tolerated subsequent courses of the same penicillin derivative, or who have passed testing in an allergist’s office. In a study of 639 patients who tested negative for penicillin allergy, 51% still had a “penicillin allergy” label in their chart more than 4 years later.36
Penicillin allergy evaluation. When a patient cannot be cleared of a “penicillin allergy” label by history alone, and in the absence of severe features such as mucous membrane involvement, they should be further evaluated through objective testing for potential IgE-mediated allergy. This assessment includes penicillin skin testing or an oral challenge, or both.
Skin testing involves skin-prick testing of major and minor determinants of penicillin; when skin-prick testing is negative, intradermal testing of major and minor determinants should follow. The negative predictive value of penicillin skin testing is high: In a prospective, multicenter investigation, researchers demonstrated that, when both the major penicillin determinant and a minor determinant mixture were used, negative predictive value was 97.9%.37
However, a minor determinant mixture is not commercially available in the United States; therefore, penicillin G is often used alone as the minor determinant. Typically, if a patient passes skin testing, a challenge dose of penicillin or amoxicillin is administered, followed by an observation period. The risk of re-sensitization after oral penicillin is thought to be low and does not preclude future use.38
Although drug testing is most often performed in an allergist’s office, several groups have developed protocols that allow for limited testing of low-risk patients in a primary care setting.8,31 For example, several studies have demonstrated that patients presenting with low-risk skin rash can be safely tested with a supervised oral challenge alone.18,28 The FIGURE8,27,30,31,33,34 outlines our proposed workflow for risk stratification and subsequent management of patients with a “penicillin allergy” label.
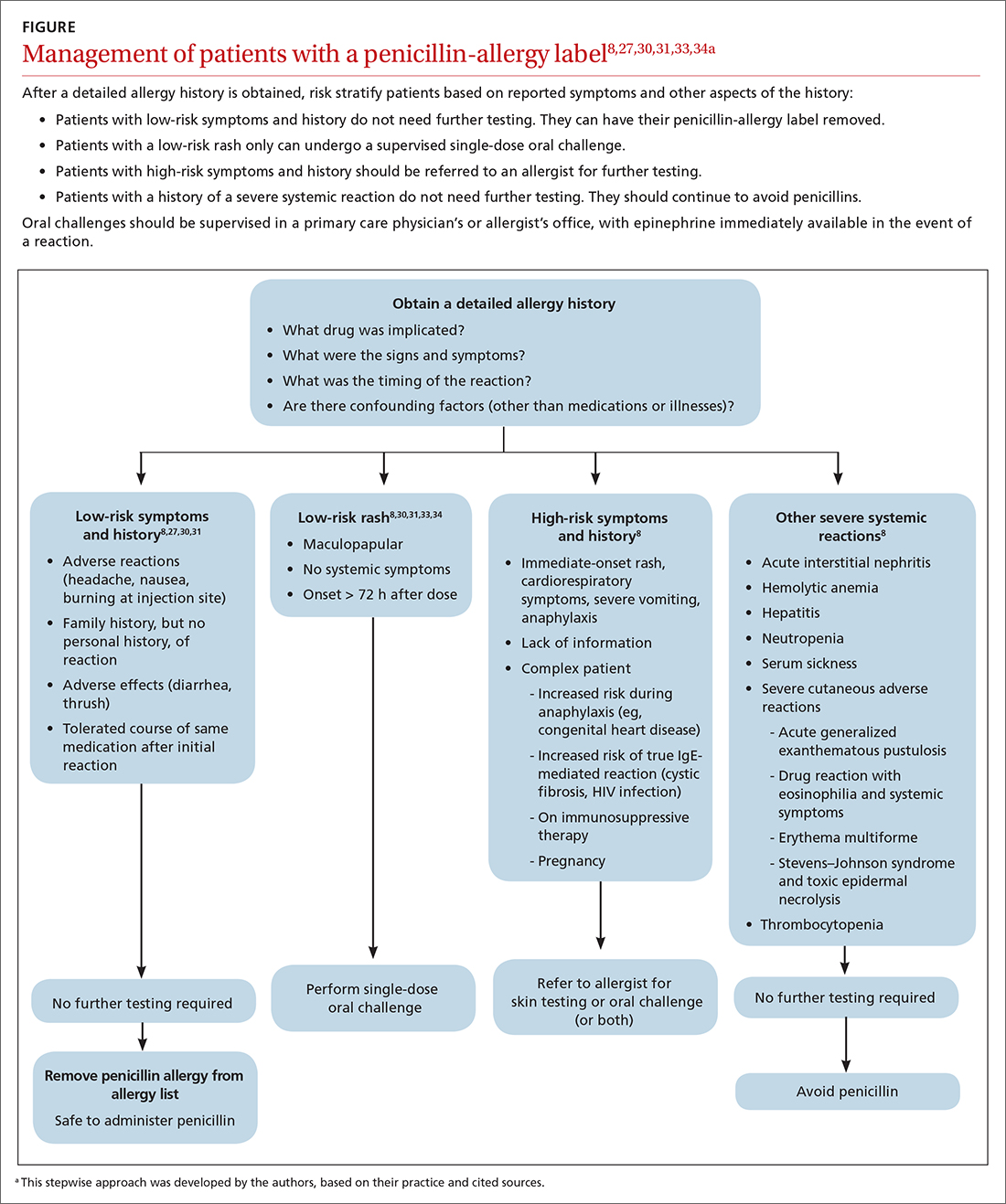
Continue to: De-labeling requires a systems approach
De-labeling requires a systems approach. Given the mismatch between the large number of patients labeled “penicillin allergic” and the few allergy specialists, referral alone is not enough to solve the problem of mislabeling. Targeting specific populations for testing, such as patients presenting to an inner-city sexually transmitted infection clinic19 or preoperative patients, as is done at the Mayo Clinic,9 has been successful. Skin testing in an inpatient setting has also been shown to be safe and effective,13 allowing for protocol-driven testing under the supervision of trained pharmacists (and others), to relieve the burden on allergy specialists.9
CORRESPONDENCE
Andrew Lutzkanin, MD, 500 University Drive, PO Box 850, Hershey, PA 17033; [email protected]
1. Macy E. The clinical evaluation of penicillin allergy: what is necessary, sufficient and safe given the materials currently available? Clin Exp Allergy. 2011;41:1498-1501. doi: 10.1111/j.1365-2222.2011.03837.x
2. Shenoy ES, Macy E, Rowe T, et al. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321:188-199. doi: 10.1001/jama.2018.19283
3. Blumenthal KG, Li Y, Banerji A, et al. The cost of penicillin allergy evaluation. J Allergy Clin Immunol Pract. 2018;6:1019-1027.e2. doi: 10.1016/j.jaip.2017.08.006
4. American Academy of Allergy, Asthma & Immunology: Ten things physicians and patients should question. American Board of Medicine Foundation Choosing Wisely website. 2018. Accessed July 7, 2021. www.choosingwisely.org/doctor-patient-lists/american-academy-of-allergy-asthma-immunology
5. Tan SY, Tatsumura Y. Alexander Fleming (1881-1955): discoverer of penicillin. Singapore Med J. 2015;56:366-367. doi: 10.11622/smedj.2015105
6. Marston HD, Dixon DM, Knisely JM, et al. Antimicrobial resistance. JAMA. 2016;316:1193-1204. doi: 10.1001/jama.2016.11764
7. Spinks A, Glasziou PP, Del Mar CB. Antibiotics for sore throat. Cochrane Database Syst Rev. 2013;2013:CD000023. doi: 10.1002/14651858.CD000023.pub4
8. Castells M, Khan DA, Phillips EJ. Penicillin allergy. N Engl J Med. 2019;381:2338-2351. doi: 10.1056/NEJMra1807761
9. Khan DA. Proactive management of penicillin and other antibiotic allergies. Allergy Asthma Proc. 2020;41:82-89. doi: 10.2500/aap.2020.41.190024
10. Idsoe O, Guthe T, Willcox RR, et al. Nature and extent of penicillin side-reactions, with particular reference to fatalities from anaphylactic shock. Bull World Health Organ. 1968;38:159-188.
11. Chiriac AM, Macy E. Large health system databases and drug hypersensitivity. J Allergy Clin Immunol Pract. 2019;7:2125-2131. doi: 10.1016/j.jaip.2019.04.014
12. Albin S, Agarwal S. Prevalence and characteristics of reported penicillin allergy in an urban outpatient adult population. Allergy Asthma Proc. 2014;35:489-494. doi: 10.2500/aap.2014.35.3791
13. Sacco KA, Bates A, Brigham TJ, et al. Clinical outcomes following inpatient penicillin allergy testing: a systematic review and meta-analysis. Allergy. 2017;72:1288-1296. doi: 10.1111/all.13168
14. Khan DA, Solensky R. Drug allergy. J Allergy Clin Immunol. 2010;125(2 suppl 2):S126-S137. doi: 10.1016/j.jaci.2009.10.028
15. Blumenthal KG, Shenoy ES, Varughese CA, et al. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann Allergy Asthma Immunol. 2015;115:294-300.e2. doi: 10.1016/j.anai.2015.05.011
16. Macy E, Schatz M, Lin C, et al. The falling rate of positive penicillin skin tests from 1995 to 2007. Perm J. 2009;13:12-18. doi: 10.7812/tpp/08-073
17. Macy E, Ngor EW. Safely diagnosing clinically significant penicillin allergy using only penicilloyl-poly-lysine, penicillin, and oral amoxicillin. J Allergy Clin Immunol Pract. 2013;1:258-263. doi: 10.1016/j.jaip.2013.02.002
18. Bourke J, Pavlos R, James I, et al. Improving the effectiveness of penicillin allergy de-labeling. J Allergy Clin Immunol Pract. 2015;3:365-374.e1. doi: 10.1016/j.jaip.2014.11.002
19. Gadde J, Spence M, Wheeler B, et al. Clinical experience with penicillin skin testing in a large inner-city STD clinic. JAMA. 1993;270:2456-2463.
20. Klaustermeyer WB, Gowda VC. Penicillin skin testing: a 20-year study at the West Los Angeles Veterans Affairs Medical Center. Mil Med. 2005;170:701-704. doi: 10.7205/milmed.170.8.701.
21. McDanel JS, Perencevich EN, Diekema DJ, et al. Comparative effectiveness of beta-lactams versus vancomycin for treatment of methicillin-susceptible Staphylococcus aureus bloodstream infections among 122 hospitals. Clin Infect Dis. 2015;61:361-367. doi: 10.1093/cid/civ308
22. Blumenthal KG, Ryan EE, Li Y, et al. The impact of a reported penicillin allergy on surgical site infection risk. Clin Infect Dis. 2018;66:329-336. doi: 10.1093/cid/cix794
23. Mattingly TJ 2nd, Fulton A, Lumish RA, et al. The cost of self-reported penicillin allergy: a systematic review. J Allergy Clin Immunol Pract. 2018;6:1649-1654.e4. doi: 10.1016/j.jaip.2017.12.033
24. Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133:790-796. doi: 10.1016/j.jaci.2013.09.021
25. Desai SH, Kaplan MS, Chen Q, et al. Morbidity in pregnant women associated with unverified penicillin allergies, antibiotic use, and group B streptococcus infections. Perm J. 2017;21:16-80. doi: 10.7812/TPP/16-080
26. Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2011;52:e56-e93. doi: 10.1093/cid/cir073
27. Vyles D, Mistry RD, Heffner V, et al. Reported knowledge and management of potential penicillin allergy in children. Acad Pediatr. 2019;19:684-690. doi: 10.1016/j.acap.2019.01.002
28. Caubet J-C, Kaiser L, Lemaître B, et al. The role of penicillin in benign skin rashes in childhood: a prospective study based on drug rechallenge. J Allergy Clin Immunol. 2011;127:218-222. doi: 10.1016/j.jaci.2010.08.025
29. Wanat M, Anthierens S, Butler CC, et al. Patient and primary care physician perceptions of penicillin allergy testing and subsequent use of penicillin-containing antibiotics: a qualitative study. J Allergy Clin Immunol Pract. 2019;7:1888-1893.e1. doi: 10.1016/j.jaip.2019.02.036
30. Norton AE, Konvinse K, Phillips EJ, et al. Antibiotic allergy in pediatrics. Pediatrics. 2018;141: e20172497. doi: 10.1542/peds.2017-2497
31. Collins C. The low risks and high rewards of penicillin allergy delabeling: an algorithm to expedite the evaluation. J Pediatr. 2019;212:216-223. doi: 10.1016/j.jpeds.2019.05.060
32. Ibia EO, Schwartz RH, Wiedermann BL. Antibiotic rashes in children: a survey in a private practice setting. Arch Dermatol. 2000;136:849-854. doi: 10.1001/archderm.136.7.849
33. Salkind AR, Cuddy PG, Foxworth JW. The rational clinical examination. Is this patient allergic to penicillin? An evidence-based analysis of the likelihood of penicillin allergy. JAMA. 2001;285:2498-2505. doi: 10.1001/jama.285.19.2498
34. Patel BM. Skin rash with infectious mononucleosis and ampicillin. Pediatrics. 1967;40:910-911.
35. Inglis JM, Caughey GE, Smith W, et al. Documentation of penicillin adverse drug reactions in electronic health records: inconsistent use of allergy and intolerance labels. Intern Med J. 2017;47:1292-1297. doi: 10.1111/imj.13558
36. Lachover-Roth I, Sharon S, Rosman Y, et al. Long-term follow-up after penicillin allergy delabeling in ambulatory patients. J Allergy Clin Immunol Pract. 2019;7:231-235.e1. doi: 10.1016/j.jaip.2018.04.042
37. Solensky R, Jacobs J, Lester M, et al. Penicillin allergy evaluation: a prospective, multicenter, open-label evaluation of a comprehensive penicillin skin test kit. J Allergy Clin Immunol Pract. 2019;7:1876-1885.e3. doi: 10.1016/j.jaip.2019.02.040
38. A; ; . Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273. doi: 10.1016/j.anai.2010.08.002
In North America and Europe, penicillin allergy is the most common drug-allergy label.1 Carrying a penicillin-allergy label, which has recently gained more attention in health care systems, leads to suboptimal outcomes, increased use of broad-spectrum antibiotics, increased risk of adverse reactions, and increased cost of care.2,3 Despite the high rate of reported reactions, clinically significant immunoglobulin E (IgE)-mediated and T cell–mediated hypersensitivity reactions to penicillins are uncommon.2
Through the Choosing Wisely initiative of the American Board of Internal Medicine Foundation, the American Academy of Allergy, Asthma, and Immunology has issued a recommendation: “Don’t overuse non-beta lactam antibiotics in patients with a history of penicillin allergy without an appropriate evaluation.”4 The primary care physician (PCP) plays a critical role in the appropriate evaluation and accurate initial labeling of penicillin allergy. Furthermore, the PCP plays an integral part, in conjunction with the allergist, in removing the “penicillin allergy” label from a patient’s chart when feasible.

The history of penicillin and prevalence of allergy
History. Penicillin, the first antibiotic, was discovered in 1928 by physician and microbiologist Alexander Fleming when he observed that a mold of the Penicillium genus inhibited growth of gram-positive pathogens.5 Along with pharmacologist Howard Florey and chemist Ernst Chain, both of whom assisted in the large-scale isolation and production of the antibiotic, Fleming won the Nobel Prize in Physiology or Medicine in 1945 for this discovery.5
Antibiotics transformed the practice of medicine across a spectrum, including safer childbirth, surgical procedures, and transplantation.6 Penicillin remains first-line therapy for many infections, such as streptococcal pharyngitis,7 and is the only recommended medication for treating syphilis during pregnancy.8 Continued effectiveness of penicillin in these cases allows broad-spectrum antibiotics to be reserved for more severe infections. Regrettably, incorrect antibiotic allergy labeling poses a significant risk to the patient and health care system.
Epidemiology. As with all medications, the potential for anaphylaxis exists after administration of penicillin. Because its use is widespread, penicillin is the most common cause of drug-induced anaphylaxis. However, the incidence of penicillin-induced anaphylaxis is low9: A 1968 World Health Organization report stated that the rate of penicillin anaphylaxis was between 0.015% and 0.04%.10 A more recent study reported an incidence of 1 in 207,191 patients after an oral dose and 1 in 95,298 after a parenteral dose.11 The most common reactions to penicillins are urticaria and delayed maculopapular rash.8
In the United States, the prevalence of reported penicillin allergy is approximately 10% (estimated range, 8% to 12%)3,12-15; among hospitalized patients, that prevalence is estimated to be as high as 15%.13,15 However, the prevalence of confirmed penicillin allergy is low and has decreased over time—demonstrated in a longitudinal study in which the rate of a positive skin test fell from 15% in 1995 to 0.8% in 2013.16,17
Studies have confirmed that as many as 90% of patients who report penicillin allergy are, in fact, able to tolerate penicillins.14,18-20 This finding might be a consequence of initial mislabeling of penicillin allergy; often, adverse reactions are documented as “allergy” when no risk of anaphylaxis exists. Furthermore, patients can outgrow IgE-mediated penicillin allergy because the presence of penicillin IgE antibodies wanes over time.14,15
Continue to: Consequences of mislabeling
Consequences of mislabeling
Clinical consequences. A multitude of clinical consequences result from carrying a “penicillin allergy” label.
Use of broad-spectrum antibiotics leads to increased risk of Clostridium difficile infection and to development of resistant bacteria, such as methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococcus.2,15
Alternative antibiotics used in the setting of a “penicillin allergy” label might be less efficacious and result in suboptimal outcomes. For example, vancomycin is less effective against methicillin-sensitive S aureus bacteremia than nafcillin or cefazolin.2,21 Beta-lactam antibiotics—in particular, cefazolin—are often first-line for perioperative prophylaxis; patients with reported penicillin allergy often receive a less-optimal alternative, such as clindamycin, vancomycin, or gentamicin.22 These patients are at increased risk of surgical site infection.2,22
In addition, using penicillin alternatives can result in greater risk of drug reactions and adverse effects.2
Increased health care costs. Primarily through observational studies, penicillin allergy has been associated with higher health care costs.23 Patients with reported penicillin allergy had, on average, a longer inpatient stay than patients without penicillin allergy, at a 3-year total estimated additional cost of $64.6 million.24 Inpatients with a listed penicillin allergy had direct drug costs ranging from “no difference” to $609 per patient more than patients without a listed penicillin allergy. Outpatient prescription costs were $14 to $193 higher per patient for patients with a listed penicillin allergy.23
Continue to: Considerations in special populations
Considerations in special populations. Evaluating penicillin allergy during routine care is key to decreasing the necessity for urgent penicillin evaluation and possible desensitization at the time of serious infection. Certain patient populations pose specific challenges:
- Pregnant patients. Unverified penicillin allergy during pregnancy is associated with an increased rate of cesarean section and longer postpartum hospitalization.25 Additionally, group B streptococcus-positive women have increased exposure to alternative antibiotics and an increased incidence of adverse drug reactions.25
- Elderly patients. Drug allergy increases with aging.1 Elderly patients in a long-term care facility are more likely to experience adverse drug effects or drug–drug interactions from the use of penicillin alternatives, such as clindamycin, vancomycin, and fluoroquinolones.2
- Oncology patients often require antibiotic prophylaxis as well as treatment for illnesses, such as neutropenic fever, for which beta-lactam antibiotics are often used as initial treatment.2,26
- Other important populations that present specific challenges include hospitalized patients, pediatric patients, and patients with a sexually transmitted infection.2
Active management of a penicillin-allergy label
Greater recognition of the consequences of penicillin allergy in recent years has led to efforts by hospitals and other health care organizations to develop processes by which patients can be successfully de-labeled as part of antibiotic stewardship programs9 and other initiatives. Ideally, every patient who has a “penicillin allergy” label would be referred to an allergist for evaluation; however, the number of allergy specialists is limited, and access to such specialists might be restricted in some areas, making this approach impracticable. Active management of penicillin allergy requires strategies to both test and de-label patients, as well as proactive approaches to prevent incorrect labeling. These proactive approaches require involvement of all members of the health care team—especially PCPs.
Preventing incorrect labeling. PCPs are the most likely to initially label a patient as allergic to penicillin.27 Most physicians rely on a reported history of allergy alone when selecting medication12; once a patient has been labeled “penicillin allergic,” they often retain that mislabel through adulthood.27,28 A qualitative study of PCPs’ views on prescribing penicillin found that many were aware that documented allergies were incorrect but were uncomfortable using their clinical judgment to prescribe a penicillin or change the record, for fear of a future anaphylactic reaction.29 The first step in the case of any reported reaction should be for you to elicit an accurate drug allergy history (TABLE 1).

As with other drug reactions, you should consider the context surrounding the reaction to a penicillin. Take care to review signs and symptoms of the reaction to look for clues that make a true allergic reaction more, or less, likely.
Symptoms can generally be divided into low-risk and high-risk categories27 (TABLE 2). An example of a commonly reported low-risk symptom is diarrhea that develops after several doses of a penicillin. In the absence of other symptoms, this finding is most likely due to elimination of normal gut flora,30 not to an allergic reaction to the medication. Symptoms of intolerance to the medication, such as headache and nausea, are also low risk.27,31 In contrast, immediate onset of abdominal pain after a dose of penicillin and lip or throat swelling are considered high risk.

Continue to: Patients presenting with urticaria...
Patients presenting with urticaria or maculopapular rash after taking penicillin are particularly challenging.30 A study of patients in a primary care pediatrics practice found that 7.4% of children receiving a prescription for a penicillin reported a rash.32 Here, timing of onset of symptoms provides some clarity about the likelihood of true allergy. Rashes that manifest during the first hours after exposure are more likely to be IgE mediated, particularly when accompanied by other systemic symptoms; they should be considered high risk. Delayed-onset rashes (> 72 hours after exposure) are usually non-IgE mediated and therefore are generally lower risk,8,30,33 except when associated with certain features, such as mucosal involvement and skin peeling.
Despite acknowledging viral exanthems in the differential, many physicians still label patients presenting with any rash as “allergic.”28 Take care to look for other potential causes of a rash; for example, patients taking amoxicillin who have concurrent Epstein-Barr virus infection frequently develop a maculopapular rash.34 Caubet and colleagues found that 56% of pediatric patients with a history of nonimmediate rash and a negative oral challenge to amoxicillin tested positive for viral infection.28
A family history of penicillin allergy alone should not preclude the use of penicillin.8,27,31 Similarly, if a patient has already received and tolerated a subsequent course of the same penicillin derivative after the initial reaction, the “penicillin allergy” label can be removed. If the reaction history is unknown, refer the patient to an allergist for further evaluation.
Accurate charting is key. With most hospital systems and physician practices now documenting in an electronic health record, there exists the ability to document, in great detail, patients’ reactions to medications. Previous studies have found, however, that such documentation is often done poorly, or not done at all. One such study found that (1) > 20% of patients with a “penicillin allergy” label did not have reaction details listed and (2) when reactions were listed, many were incorrectly labeled as “allergy,” not “intolerance.”35
Many electronic health record systems lump drug allergies, adverse effects, and food and environmental allergies into a single section, leading to a lack of distinction between adverse reactions and true allergy.31 Although many PCPs report that it is easy to change a patient’s allergy label in the record,29 more often, a nurse, resident, or consultant actually documents the reaction.35
Continue to: Documentation at the time of the reaction...
Documentation at the time of the reaction, within the encounter note and the allergy tab, is essential, so that other physicians caring for the patient, in the future, will be knowledgeable about the details of the reaction. Make it your responsibility to accurately document penicillin allergy in patients’ charts, including removing the “penicillin allergy” label from the chart of patients whose history is inconsistent with allergy, who have tolerated subsequent courses of the same penicillin derivative, or who have passed testing in an allergist’s office. In a study of 639 patients who tested negative for penicillin allergy, 51% still had a “penicillin allergy” label in their chart more than 4 years later.36
Penicillin allergy evaluation. When a patient cannot be cleared of a “penicillin allergy” label by history alone, and in the absence of severe features such as mucous membrane involvement, they should be further evaluated through objective testing for potential IgE-mediated allergy. This assessment includes penicillin skin testing or an oral challenge, or both.
Skin testing involves skin-prick testing of major and minor determinants of penicillin; when skin-prick testing is negative, intradermal testing of major and minor determinants should follow. The negative predictive value of penicillin skin testing is high: In a prospective, multicenter investigation, researchers demonstrated that, when both the major penicillin determinant and a minor determinant mixture were used, negative predictive value was 97.9%.37
However, a minor determinant mixture is not commercially available in the United States; therefore, penicillin G is often used alone as the minor determinant. Typically, if a patient passes skin testing, a challenge dose of penicillin or amoxicillin is administered, followed by an observation period. The risk of re-sensitization after oral penicillin is thought to be low and does not preclude future use.38
Although drug testing is most often performed in an allergist’s office, several groups have developed protocols that allow for limited testing of low-risk patients in a primary care setting.8,31 For example, several studies have demonstrated that patients presenting with low-risk skin rash can be safely tested with a supervised oral challenge alone.18,28 The FIGURE8,27,30,31,33,34 outlines our proposed workflow for risk stratification and subsequent management of patients with a “penicillin allergy” label.

Continue to: De-labeling requires a systems approach
De-labeling requires a systems approach. Given the mismatch between the large number of patients labeled “penicillin allergic” and the few allergy specialists, referral alone is not enough to solve the problem of mislabeling. Targeting specific populations for testing, such as patients presenting to an inner-city sexually transmitted infection clinic19 or preoperative patients, as is done at the Mayo Clinic,9 has been successful. Skin testing in an inpatient setting has also been shown to be safe and effective,13 allowing for protocol-driven testing under the supervision of trained pharmacists (and others), to relieve the burden on allergy specialists.9
CORRESPONDENCE
Andrew Lutzkanin, MD, 500 University Drive, PO Box 850, Hershey, PA 17033; [email protected]
In North America and Europe, penicillin allergy is the most common drug-allergy label.1 Carrying a penicillin-allergy label, which has recently gained more attention in health care systems, leads to suboptimal outcomes, increased use of broad-spectrum antibiotics, increased risk of adverse reactions, and increased cost of care.2,3 Despite the high rate of reported reactions, clinically significant immunoglobulin E (IgE)-mediated and T cell–mediated hypersensitivity reactions to penicillins are uncommon.2
Through the Choosing Wisely initiative of the American Board of Internal Medicine Foundation, the American Academy of Allergy, Asthma, and Immunology has issued a recommendation: “Don’t overuse non-beta lactam antibiotics in patients with a history of penicillin allergy without an appropriate evaluation.”4 The primary care physician (PCP) plays a critical role in the appropriate evaluation and accurate initial labeling of penicillin allergy. Furthermore, the PCP plays an integral part, in conjunction with the allergist, in removing the “penicillin allergy” label from a patient’s chart when feasible.

The history of penicillin and prevalence of allergy
History. Penicillin, the first antibiotic, was discovered in 1928 by physician and microbiologist Alexander Fleming when he observed that a mold of the Penicillium genus inhibited growth of gram-positive pathogens.5 Along with pharmacologist Howard Florey and chemist Ernst Chain, both of whom assisted in the large-scale isolation and production of the antibiotic, Fleming won the Nobel Prize in Physiology or Medicine in 1945 for this discovery.5
Antibiotics transformed the practice of medicine across a spectrum, including safer childbirth, surgical procedures, and transplantation.6 Penicillin remains first-line therapy for many infections, such as streptococcal pharyngitis,7 and is the only recommended medication for treating syphilis during pregnancy.8 Continued effectiveness of penicillin in these cases allows broad-spectrum antibiotics to be reserved for more severe infections. Regrettably, incorrect antibiotic allergy labeling poses a significant risk to the patient and health care system.
Epidemiology. As with all medications, the potential for anaphylaxis exists after administration of penicillin. Because its use is widespread, penicillin is the most common cause of drug-induced anaphylaxis. However, the incidence of penicillin-induced anaphylaxis is low9: A 1968 World Health Organization report stated that the rate of penicillin anaphylaxis was between 0.015% and 0.04%.10 A more recent study reported an incidence of 1 in 207,191 patients after an oral dose and 1 in 95,298 after a parenteral dose.11 The most common reactions to penicillins are urticaria and delayed maculopapular rash.8
In the United States, the prevalence of reported penicillin allergy is approximately 10% (estimated range, 8% to 12%)3,12-15; among hospitalized patients, that prevalence is estimated to be as high as 15%.13,15 However, the prevalence of confirmed penicillin allergy is low and has decreased over time—demonstrated in a longitudinal study in which the rate of a positive skin test fell from 15% in 1995 to 0.8% in 2013.16,17
Studies have confirmed that as many as 90% of patients who report penicillin allergy are, in fact, able to tolerate penicillins.14,18-20 This finding might be a consequence of initial mislabeling of penicillin allergy; often, adverse reactions are documented as “allergy” when no risk of anaphylaxis exists. Furthermore, patients can outgrow IgE-mediated penicillin allergy because the presence of penicillin IgE antibodies wanes over time.14,15
Continue to: Consequences of mislabeling
Consequences of mislabeling
Clinical consequences. A multitude of clinical consequences result from carrying a “penicillin allergy” label.
Use of broad-spectrum antibiotics leads to increased risk of Clostridium difficile infection and to development of resistant bacteria, such as methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococcus.2,15
Alternative antibiotics used in the setting of a “penicillin allergy” label might be less efficacious and result in suboptimal outcomes. For example, vancomycin is less effective against methicillin-sensitive S aureus bacteremia than nafcillin or cefazolin.2,21 Beta-lactam antibiotics—in particular, cefazolin—are often first-line for perioperative prophylaxis; patients with reported penicillin allergy often receive a less-optimal alternative, such as clindamycin, vancomycin, or gentamicin.22 These patients are at increased risk of surgical site infection.2,22
In addition, using penicillin alternatives can result in greater risk of drug reactions and adverse effects.2
Increased health care costs. Primarily through observational studies, penicillin allergy has been associated with higher health care costs.23 Patients with reported penicillin allergy had, on average, a longer inpatient stay than patients without penicillin allergy, at a 3-year total estimated additional cost of $64.6 million.24 Inpatients with a listed penicillin allergy had direct drug costs ranging from “no difference” to $609 per patient more than patients without a listed penicillin allergy. Outpatient prescription costs were $14 to $193 higher per patient for patients with a listed penicillin allergy.23
Continue to: Considerations in special populations
Considerations in special populations. Evaluating penicillin allergy during routine care is key to decreasing the necessity for urgent penicillin evaluation and possible desensitization at the time of serious infection. Certain patient populations pose specific challenges:
- Pregnant patients. Unverified penicillin allergy during pregnancy is associated with an increased rate of cesarean section and longer postpartum hospitalization.25 Additionally, group B streptococcus-positive women have increased exposure to alternative antibiotics and an increased incidence of adverse drug reactions.25
- Elderly patients. Drug allergy increases with aging.1 Elderly patients in a long-term care facility are more likely to experience adverse drug effects or drug–drug interactions from the use of penicillin alternatives, such as clindamycin, vancomycin, and fluoroquinolones.2
- Oncology patients often require antibiotic prophylaxis as well as treatment for illnesses, such as neutropenic fever, for which beta-lactam antibiotics are often used as initial treatment.2,26
- Other important populations that present specific challenges include hospitalized patients, pediatric patients, and patients with a sexually transmitted infection.2
Active management of a penicillin-allergy label
Greater recognition of the consequences of penicillin allergy in recent years has led to efforts by hospitals and other health care organizations to develop processes by which patients can be successfully de-labeled as part of antibiotic stewardship programs9 and other initiatives. Ideally, every patient who has a “penicillin allergy” label would be referred to an allergist for evaluation; however, the number of allergy specialists is limited, and access to such specialists might be restricted in some areas, making this approach impracticable. Active management of penicillin allergy requires strategies to both test and de-label patients, as well as proactive approaches to prevent incorrect labeling. These proactive approaches require involvement of all members of the health care team—especially PCPs.
Preventing incorrect labeling. PCPs are the most likely to initially label a patient as allergic to penicillin.27 Most physicians rely on a reported history of allergy alone when selecting medication12; once a patient has been labeled “penicillin allergic,” they often retain that mislabel through adulthood.27,28 A qualitative study of PCPs’ views on prescribing penicillin found that many were aware that documented allergies were incorrect but were uncomfortable using their clinical judgment to prescribe a penicillin or change the record, for fear of a future anaphylactic reaction.29 The first step in the case of any reported reaction should be for you to elicit an accurate drug allergy history (TABLE 1).

As with other drug reactions, you should consider the context surrounding the reaction to a penicillin. Take care to review signs and symptoms of the reaction to look for clues that make a true allergic reaction more, or less, likely.
Symptoms can generally be divided into low-risk and high-risk categories27 (TABLE 2). An example of a commonly reported low-risk symptom is diarrhea that develops after several doses of a penicillin. In the absence of other symptoms, this finding is most likely due to elimination of normal gut flora,30 not to an allergic reaction to the medication. Symptoms of intolerance to the medication, such as headache and nausea, are also low risk.27,31 In contrast, immediate onset of abdominal pain after a dose of penicillin and lip or throat swelling are considered high risk.

Continue to: Patients presenting with urticaria...
Patients presenting with urticaria or maculopapular rash after taking penicillin are particularly challenging.30 A study of patients in a primary care pediatrics practice found that 7.4% of children receiving a prescription for a penicillin reported a rash.32 Here, timing of onset of symptoms provides some clarity about the likelihood of true allergy. Rashes that manifest during the first hours after exposure are more likely to be IgE mediated, particularly when accompanied by other systemic symptoms; they should be considered high risk. Delayed-onset rashes (> 72 hours after exposure) are usually non-IgE mediated and therefore are generally lower risk,8,30,33 except when associated with certain features, such as mucosal involvement and skin peeling.
Despite acknowledging viral exanthems in the differential, many physicians still label patients presenting with any rash as “allergic.”28 Take care to look for other potential causes of a rash; for example, patients taking amoxicillin who have concurrent Epstein-Barr virus infection frequently develop a maculopapular rash.34 Caubet and colleagues found that 56% of pediatric patients with a history of nonimmediate rash and a negative oral challenge to amoxicillin tested positive for viral infection.28
A family history of penicillin allergy alone should not preclude the use of penicillin.8,27,31 Similarly, if a patient has already received and tolerated a subsequent course of the same penicillin derivative after the initial reaction, the “penicillin allergy” label can be removed. If the reaction history is unknown, refer the patient to an allergist for further evaluation.
Accurate charting is key. With most hospital systems and physician practices now documenting in an electronic health record, there exists the ability to document, in great detail, patients’ reactions to medications. Previous studies have found, however, that such documentation is often done poorly, or not done at all. One such study found that (1) > 20% of patients with a “penicillin allergy” label did not have reaction details listed and (2) when reactions were listed, many were incorrectly labeled as “allergy,” not “intolerance.”35
Many electronic health record systems lump drug allergies, adverse effects, and food and environmental allergies into a single section, leading to a lack of distinction between adverse reactions and true allergy.31 Although many PCPs report that it is easy to change a patient’s allergy label in the record,29 more often, a nurse, resident, or consultant actually documents the reaction.35
Continue to: Documentation at the time of the reaction...
Documentation at the time of the reaction, within the encounter note and the allergy tab, is essential, so that other physicians caring for the patient, in the future, will be knowledgeable about the details of the reaction. Make it your responsibility to accurately document penicillin allergy in patients’ charts, including removing the “penicillin allergy” label from the chart of patients whose history is inconsistent with allergy, who have tolerated subsequent courses of the same penicillin derivative, or who have passed testing in an allergist’s office. In a study of 639 patients who tested negative for penicillin allergy, 51% still had a “penicillin allergy” label in their chart more than 4 years later.36
Penicillin allergy evaluation. When a patient cannot be cleared of a “penicillin allergy” label by history alone, and in the absence of severe features such as mucous membrane involvement, they should be further evaluated through objective testing for potential IgE-mediated allergy. This assessment includes penicillin skin testing or an oral challenge, or both.
Skin testing involves skin-prick testing of major and minor determinants of penicillin; when skin-prick testing is negative, intradermal testing of major and minor determinants should follow. The negative predictive value of penicillin skin testing is high: In a prospective, multicenter investigation, researchers demonstrated that, when both the major penicillin determinant and a minor determinant mixture were used, negative predictive value was 97.9%.37
However, a minor determinant mixture is not commercially available in the United States; therefore, penicillin G is often used alone as the minor determinant. Typically, if a patient passes skin testing, a challenge dose of penicillin or amoxicillin is administered, followed by an observation period. The risk of re-sensitization after oral penicillin is thought to be low and does not preclude future use.38
Although drug testing is most often performed in an allergist’s office, several groups have developed protocols that allow for limited testing of low-risk patients in a primary care setting.8,31 For example, several studies have demonstrated that patients presenting with low-risk skin rash can be safely tested with a supervised oral challenge alone.18,28 The FIGURE8,27,30,31,33,34 outlines our proposed workflow for risk stratification and subsequent management of patients with a “penicillin allergy” label.

Continue to: De-labeling requires a systems approach
De-labeling requires a systems approach. Given the mismatch between the large number of patients labeled “penicillin allergic” and the few allergy specialists, referral alone is not enough to solve the problem of mislabeling. Targeting specific populations for testing, such as patients presenting to an inner-city sexually transmitted infection clinic19 or preoperative patients, as is done at the Mayo Clinic,9 has been successful. Skin testing in an inpatient setting has also been shown to be safe and effective,13 allowing for protocol-driven testing under the supervision of trained pharmacists (and others), to relieve the burden on allergy specialists.9
CORRESPONDENCE
Andrew Lutzkanin, MD, 500 University Drive, PO Box 850, Hershey, PA 17033; [email protected]
1. Macy E. The clinical evaluation of penicillin allergy: what is necessary, sufficient and safe given the materials currently available? Clin Exp Allergy. 2011;41:1498-1501. doi: 10.1111/j.1365-2222.2011.03837.x
2. Shenoy ES, Macy E, Rowe T, et al. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321:188-199. doi: 10.1001/jama.2018.19283
3. Blumenthal KG, Li Y, Banerji A, et al. The cost of penicillin allergy evaluation. J Allergy Clin Immunol Pract. 2018;6:1019-1027.e2. doi: 10.1016/j.jaip.2017.08.006
4. American Academy of Allergy, Asthma & Immunology: Ten things physicians and patients should question. American Board of Medicine Foundation Choosing Wisely website. 2018. Accessed July 7, 2021. www.choosingwisely.org/doctor-patient-lists/american-academy-of-allergy-asthma-immunology
5. Tan SY, Tatsumura Y. Alexander Fleming (1881-1955): discoverer of penicillin. Singapore Med J. 2015;56:366-367. doi: 10.11622/smedj.2015105
6. Marston HD, Dixon DM, Knisely JM, et al. Antimicrobial resistance. JAMA. 2016;316:1193-1204. doi: 10.1001/jama.2016.11764
7. Spinks A, Glasziou PP, Del Mar CB. Antibiotics for sore throat. Cochrane Database Syst Rev. 2013;2013:CD000023. doi: 10.1002/14651858.CD000023.pub4
8. Castells M, Khan DA, Phillips EJ. Penicillin allergy. N Engl J Med. 2019;381:2338-2351. doi: 10.1056/NEJMra1807761
9. Khan DA. Proactive management of penicillin and other antibiotic allergies. Allergy Asthma Proc. 2020;41:82-89. doi: 10.2500/aap.2020.41.190024
10. Idsoe O, Guthe T, Willcox RR, et al. Nature and extent of penicillin side-reactions, with particular reference to fatalities from anaphylactic shock. Bull World Health Organ. 1968;38:159-188.
11. Chiriac AM, Macy E. Large health system databases and drug hypersensitivity. J Allergy Clin Immunol Pract. 2019;7:2125-2131. doi: 10.1016/j.jaip.2019.04.014
12. Albin S, Agarwal S. Prevalence and characteristics of reported penicillin allergy in an urban outpatient adult population. Allergy Asthma Proc. 2014;35:489-494. doi: 10.2500/aap.2014.35.3791
13. Sacco KA, Bates A, Brigham TJ, et al. Clinical outcomes following inpatient penicillin allergy testing: a systematic review and meta-analysis. Allergy. 2017;72:1288-1296. doi: 10.1111/all.13168
14. Khan DA, Solensky R. Drug allergy. J Allergy Clin Immunol. 2010;125(2 suppl 2):S126-S137. doi: 10.1016/j.jaci.2009.10.028
15. Blumenthal KG, Shenoy ES, Varughese CA, et al. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann Allergy Asthma Immunol. 2015;115:294-300.e2. doi: 10.1016/j.anai.2015.05.011
16. Macy E, Schatz M, Lin C, et al. The falling rate of positive penicillin skin tests from 1995 to 2007. Perm J. 2009;13:12-18. doi: 10.7812/tpp/08-073
17. Macy E, Ngor EW. Safely diagnosing clinically significant penicillin allergy using only penicilloyl-poly-lysine, penicillin, and oral amoxicillin. J Allergy Clin Immunol Pract. 2013;1:258-263. doi: 10.1016/j.jaip.2013.02.002
18. Bourke J, Pavlos R, James I, et al. Improving the effectiveness of penicillin allergy de-labeling. J Allergy Clin Immunol Pract. 2015;3:365-374.e1. doi: 10.1016/j.jaip.2014.11.002
19. Gadde J, Spence M, Wheeler B, et al. Clinical experience with penicillin skin testing in a large inner-city STD clinic. JAMA. 1993;270:2456-2463.
20. Klaustermeyer WB, Gowda VC. Penicillin skin testing: a 20-year study at the West Los Angeles Veterans Affairs Medical Center. Mil Med. 2005;170:701-704. doi: 10.7205/milmed.170.8.701.
21. McDanel JS, Perencevich EN, Diekema DJ, et al. Comparative effectiveness of beta-lactams versus vancomycin for treatment of methicillin-susceptible Staphylococcus aureus bloodstream infections among 122 hospitals. Clin Infect Dis. 2015;61:361-367. doi: 10.1093/cid/civ308
22. Blumenthal KG, Ryan EE, Li Y, et al. The impact of a reported penicillin allergy on surgical site infection risk. Clin Infect Dis. 2018;66:329-336. doi: 10.1093/cid/cix794
23. Mattingly TJ 2nd, Fulton A, Lumish RA, et al. The cost of self-reported penicillin allergy: a systematic review. J Allergy Clin Immunol Pract. 2018;6:1649-1654.e4. doi: 10.1016/j.jaip.2017.12.033
24. Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133:790-796. doi: 10.1016/j.jaci.2013.09.021
25. Desai SH, Kaplan MS, Chen Q, et al. Morbidity in pregnant women associated with unverified penicillin allergies, antibiotic use, and group B streptococcus infections. Perm J. 2017;21:16-80. doi: 10.7812/TPP/16-080
26. Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2011;52:e56-e93. doi: 10.1093/cid/cir073
27. Vyles D, Mistry RD, Heffner V, et al. Reported knowledge and management of potential penicillin allergy in children. Acad Pediatr. 2019;19:684-690. doi: 10.1016/j.acap.2019.01.002
28. Caubet J-C, Kaiser L, Lemaître B, et al. The role of penicillin in benign skin rashes in childhood: a prospective study based on drug rechallenge. J Allergy Clin Immunol. 2011;127:218-222. doi: 10.1016/j.jaci.2010.08.025
29. Wanat M, Anthierens S, Butler CC, et al. Patient and primary care physician perceptions of penicillin allergy testing and subsequent use of penicillin-containing antibiotics: a qualitative study. J Allergy Clin Immunol Pract. 2019;7:1888-1893.e1. doi: 10.1016/j.jaip.2019.02.036
30. Norton AE, Konvinse K, Phillips EJ, et al. Antibiotic allergy in pediatrics. Pediatrics. 2018;141: e20172497. doi: 10.1542/peds.2017-2497
31. Collins C. The low risks and high rewards of penicillin allergy delabeling: an algorithm to expedite the evaluation. J Pediatr. 2019;212:216-223. doi: 10.1016/j.jpeds.2019.05.060
32. Ibia EO, Schwartz RH, Wiedermann BL. Antibiotic rashes in children: a survey in a private practice setting. Arch Dermatol. 2000;136:849-854. doi: 10.1001/archderm.136.7.849
33. Salkind AR, Cuddy PG, Foxworth JW. The rational clinical examination. Is this patient allergic to penicillin? An evidence-based analysis of the likelihood of penicillin allergy. JAMA. 2001;285:2498-2505. doi: 10.1001/jama.285.19.2498
34. Patel BM. Skin rash with infectious mononucleosis and ampicillin. Pediatrics. 1967;40:910-911.
35. Inglis JM, Caughey GE, Smith W, et al. Documentation of penicillin adverse drug reactions in electronic health records: inconsistent use of allergy and intolerance labels. Intern Med J. 2017;47:1292-1297. doi: 10.1111/imj.13558
36. Lachover-Roth I, Sharon S, Rosman Y, et al. Long-term follow-up after penicillin allergy delabeling in ambulatory patients. J Allergy Clin Immunol Pract. 2019;7:231-235.e1. doi: 10.1016/j.jaip.2018.04.042
37. Solensky R, Jacobs J, Lester M, et al. Penicillin allergy evaluation: a prospective, multicenter, open-label evaluation of a comprehensive penicillin skin test kit. J Allergy Clin Immunol Pract. 2019;7:1876-1885.e3. doi: 10.1016/j.jaip.2019.02.040
38. A; ; . Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273. doi: 10.1016/j.anai.2010.08.002
1. Macy E. The clinical evaluation of penicillin allergy: what is necessary, sufficient and safe given the materials currently available? Clin Exp Allergy. 2011;41:1498-1501. doi: 10.1111/j.1365-2222.2011.03837.x
2. Shenoy ES, Macy E, Rowe T, et al. Evaluation and management of penicillin allergy: a review. JAMA. 2019;321:188-199. doi: 10.1001/jama.2018.19283
3. Blumenthal KG, Li Y, Banerji A, et al. The cost of penicillin allergy evaluation. J Allergy Clin Immunol Pract. 2018;6:1019-1027.e2. doi: 10.1016/j.jaip.2017.08.006
4. American Academy of Allergy, Asthma & Immunology: Ten things physicians and patients should question. American Board of Medicine Foundation Choosing Wisely website. 2018. Accessed July 7, 2021. www.choosingwisely.org/doctor-patient-lists/american-academy-of-allergy-asthma-immunology
5. Tan SY, Tatsumura Y. Alexander Fleming (1881-1955): discoverer of penicillin. Singapore Med J. 2015;56:366-367. doi: 10.11622/smedj.2015105
6. Marston HD, Dixon DM, Knisely JM, et al. Antimicrobial resistance. JAMA. 2016;316:1193-1204. doi: 10.1001/jama.2016.11764
7. Spinks A, Glasziou PP, Del Mar CB. Antibiotics for sore throat. Cochrane Database Syst Rev. 2013;2013:CD000023. doi: 10.1002/14651858.CD000023.pub4
8. Castells M, Khan DA, Phillips EJ. Penicillin allergy. N Engl J Med. 2019;381:2338-2351. doi: 10.1056/NEJMra1807761
9. Khan DA. Proactive management of penicillin and other antibiotic allergies. Allergy Asthma Proc. 2020;41:82-89. doi: 10.2500/aap.2020.41.190024
10. Idsoe O, Guthe T, Willcox RR, et al. Nature and extent of penicillin side-reactions, with particular reference to fatalities from anaphylactic shock. Bull World Health Organ. 1968;38:159-188.
11. Chiriac AM, Macy E. Large health system databases and drug hypersensitivity. J Allergy Clin Immunol Pract. 2019;7:2125-2131. doi: 10.1016/j.jaip.2019.04.014
12. Albin S, Agarwal S. Prevalence and characteristics of reported penicillin allergy in an urban outpatient adult population. Allergy Asthma Proc. 2014;35:489-494. doi: 10.2500/aap.2014.35.3791
13. Sacco KA, Bates A, Brigham TJ, et al. Clinical outcomes following inpatient penicillin allergy testing: a systematic review and meta-analysis. Allergy. 2017;72:1288-1296. doi: 10.1111/all.13168
14. Khan DA, Solensky R. Drug allergy. J Allergy Clin Immunol. 2010;125(2 suppl 2):S126-S137. doi: 10.1016/j.jaci.2009.10.028
15. Blumenthal KG, Shenoy ES, Varughese CA, et al. Impact of a clinical guideline for prescribing antibiotics to inpatients reporting penicillin or cephalosporin allergy. Ann Allergy Asthma Immunol. 2015;115:294-300.e2. doi: 10.1016/j.anai.2015.05.011
16. Macy E, Schatz M, Lin C, et al. The falling rate of positive penicillin skin tests from 1995 to 2007. Perm J. 2009;13:12-18. doi: 10.7812/tpp/08-073
17. Macy E, Ngor EW. Safely diagnosing clinically significant penicillin allergy using only penicilloyl-poly-lysine, penicillin, and oral amoxicillin. J Allergy Clin Immunol Pract. 2013;1:258-263. doi: 10.1016/j.jaip.2013.02.002
18. Bourke J, Pavlos R, James I, et al. Improving the effectiveness of penicillin allergy de-labeling. J Allergy Clin Immunol Pract. 2015;3:365-374.e1. doi: 10.1016/j.jaip.2014.11.002
19. Gadde J, Spence M, Wheeler B, et al. Clinical experience with penicillin skin testing in a large inner-city STD clinic. JAMA. 1993;270:2456-2463.
20. Klaustermeyer WB, Gowda VC. Penicillin skin testing: a 20-year study at the West Los Angeles Veterans Affairs Medical Center. Mil Med. 2005;170:701-704. doi: 10.7205/milmed.170.8.701.
21. McDanel JS, Perencevich EN, Diekema DJ, et al. Comparative effectiveness of beta-lactams versus vancomycin for treatment of methicillin-susceptible Staphylococcus aureus bloodstream infections among 122 hospitals. Clin Infect Dis. 2015;61:361-367. doi: 10.1093/cid/civ308
22. Blumenthal KG, Ryan EE, Li Y, et al. The impact of a reported penicillin allergy on surgical site infection risk. Clin Infect Dis. 2018;66:329-336. doi: 10.1093/cid/cix794
23. Mattingly TJ 2nd, Fulton A, Lumish RA, et al. The cost of self-reported penicillin allergy: a systematic review. J Allergy Clin Immunol Pract. 2018;6:1649-1654.e4. doi: 10.1016/j.jaip.2017.12.033
24. Macy E, Contreras R. Health care use and serious infection prevalence associated with penicillin “allergy” in hospitalized patients: a cohort study. J Allergy Clin Immunol. 2014;133:790-796. doi: 10.1016/j.jaci.2013.09.021
25. Desai SH, Kaplan MS, Chen Q, et al. Morbidity in pregnant women associated with unverified penicillin allergies, antibiotic use, and group B streptococcus infections. Perm J. 2017;21:16-80. doi: 10.7812/TPP/16-080
26. Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2011;52:e56-e93. doi: 10.1093/cid/cir073
27. Vyles D, Mistry RD, Heffner V, et al. Reported knowledge and management of potential penicillin allergy in children. Acad Pediatr. 2019;19:684-690. doi: 10.1016/j.acap.2019.01.002
28. Caubet J-C, Kaiser L, Lemaître B, et al. The role of penicillin in benign skin rashes in childhood: a prospective study based on drug rechallenge. J Allergy Clin Immunol. 2011;127:218-222. doi: 10.1016/j.jaci.2010.08.025
29. Wanat M, Anthierens S, Butler CC, et al. Patient and primary care physician perceptions of penicillin allergy testing and subsequent use of penicillin-containing antibiotics: a qualitative study. J Allergy Clin Immunol Pract. 2019;7:1888-1893.e1. doi: 10.1016/j.jaip.2019.02.036
30. Norton AE, Konvinse K, Phillips EJ, et al. Antibiotic allergy in pediatrics. Pediatrics. 2018;141: e20172497. doi: 10.1542/peds.2017-2497
31. Collins C. The low risks and high rewards of penicillin allergy delabeling: an algorithm to expedite the evaluation. J Pediatr. 2019;212:216-223. doi: 10.1016/j.jpeds.2019.05.060
32. Ibia EO, Schwartz RH, Wiedermann BL. Antibiotic rashes in children: a survey in a private practice setting. Arch Dermatol. 2000;136:849-854. doi: 10.1001/archderm.136.7.849
33. Salkind AR, Cuddy PG, Foxworth JW. The rational clinical examination. Is this patient allergic to penicillin? An evidence-based analysis of the likelihood of penicillin allergy. JAMA. 2001;285:2498-2505. doi: 10.1001/jama.285.19.2498
34. Patel BM. Skin rash with infectious mononucleosis and ampicillin. Pediatrics. 1967;40:910-911.
35. Inglis JM, Caughey GE, Smith W, et al. Documentation of penicillin adverse drug reactions in electronic health records: inconsistent use of allergy and intolerance labels. Intern Med J. 2017;47:1292-1297. doi: 10.1111/imj.13558
36. Lachover-Roth I, Sharon S, Rosman Y, et al. Long-term follow-up after penicillin allergy delabeling in ambulatory patients. J Allergy Clin Immunol Pract. 2019;7:231-235.e1. doi: 10.1016/j.jaip.2018.04.042
37. Solensky R, Jacobs J, Lester M, et al. Penicillin allergy evaluation: a prospective, multicenter, open-label evaluation of a comprehensive penicillin skin test kit. J Allergy Clin Immunol Pract. 2019;7:1876-1885.e3. doi: 10.1016/j.jaip.2019.02.040
38. A; ; . Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273. doi: 10.1016/j.anai.2010.08.002
PRACTICE RECOMMENDATIONS
› Obtain an accurate drug allergy history from all patients who have a listed penicillin allergy. B
› De-label penicillin allergy in patients who report symptoms of an adverse reaction (diarrhea, headache, or nausea) but who (1) do not have other systemic symptoms; (2) do have a family history, but no personal history, of a reaction; or (3) have tolerated the same penicillin derivative since the initial reaction. B
› Refer patients whose reaction history includes hives, shortness of breath, or other allergic-type signs and symptoms for potential skin testing or oral challenge, or both. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Children and COVID: New cases down slightly from record high
Weekly cases of COVID-19 in children dropped for the first time since June, and daily hospitalizations appear to be falling, even as the pace of vaccinations continues to slow among the youngest eligible recipients, according to new data.
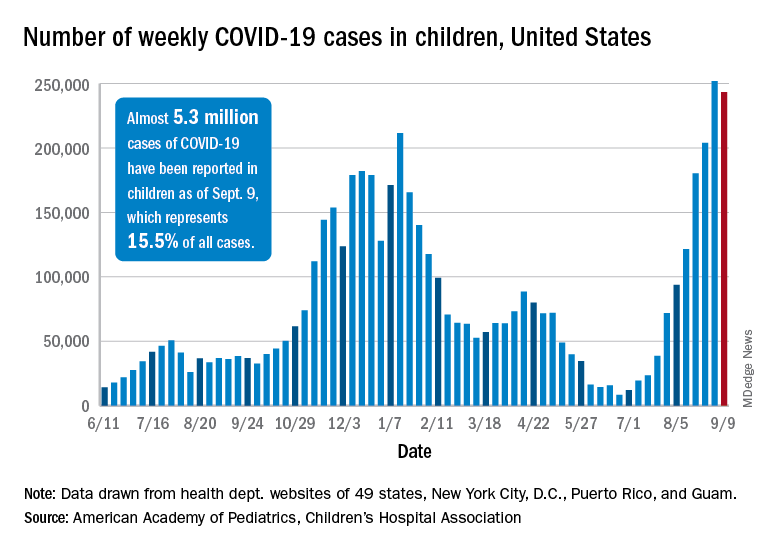
Despite the 3.3% decline from the previous week’s record high, the new-case count still topped 243,000 for the week of Sept. 3-9, putting the total number of cases in children at almost 5.3 million since the pandemic began.
Hospitalizations seem to have peaked on Sept. 4, when the rate for children aged 0-17 years reached 0.51 per 100,000 population. The admission rate for confirmed COVID-19 has dropped steadily since then and was down to 0.45 per 100,000 on Sept. 11, the last day for which preliminary data from the Centers for Disease Control and Prevention were available.
On the prevention side, fully vaccinated children aged 12-17 years represented 5.5% of all Americans who had completed the vaccine regimen as of Sept. 13. Vaccine initiation, however, has dropped for 5 consecutive weeks in 12- to 15-year-olds and in 4 of the last 5 weeks among 16- and 17-year-olds, the CDC said on its COVID Data Tracker.
Just under 199,000 children aged 12-15 received their first dose of the COVID-19 vaccine during the week of Sept. 7-13. That’s down by 18.5% from the week before and by 51.6% since Aug. 9, the last week that vaccine initiation increased for the age group. Among 16- and 17-year-olds, the 83,000 new recipients that week was a decrease of 25.7% from the previous week and a decline of 47% since the summer peak of Aug. 9, the CDC data show.
Those newest recipients bring at-least-one-dose status to 52.0% of those aged 12-15 and 59.9% of the 16- and 17-year-olds, while 40.3% and 48.9% were fully vaccinated as of Sept. 13. Corresponding figures for some of the older groups are 61.6%/49.7% (age 18-24 years), 73.8%/63.1% (40-49 years), and 95.1%/84.5% (65-74 years), the CDC said.
Vaccine coverage for children at the state level deviates considerably from the national averages. The highest rates for children aged 12-17 are to be found in Vermont, where 76% have received at least one dose, the AAP reported in a separate analysis. Massachusetts is just below that but also comes in at 76% by virtue of a rounding error. The other states in the top five are Connecticut (74%), Hawaii (73%), and Rhode Island (71%).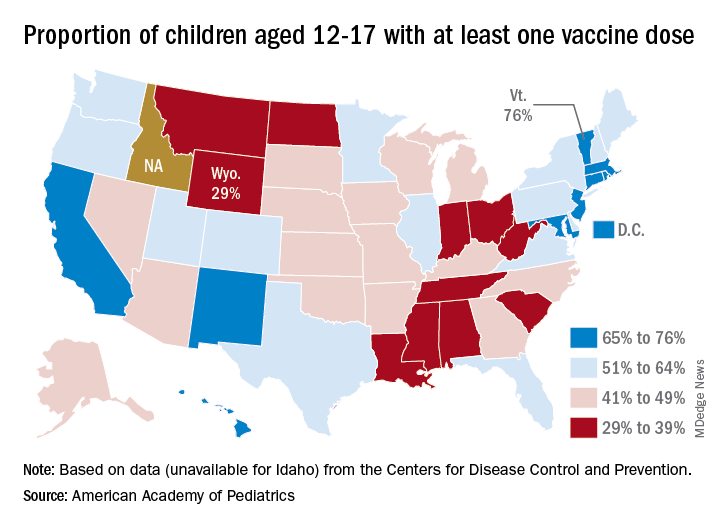
The lowest vaccination rate for children comes from Wyoming (29%), which is preceded by North Dakota (33%), West Virginia (33%), Alabama (33%), and Mississippi (34%). the AAP said based on data from the CDC, which does not include Idaho.
In a bit of a side note, West Virginia’s Republican governor, Jim Justice, recently said this about vaccine reluctance in his state: “For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the same very people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Over the last 3 weeks, the District of Columbia has had the largest increase in children having received at least one dose: 10 percentage points, as it went from 58% to 68%. The next-largest improvement – 7 percentage points – occurred in Georgia (34% to 41%), New Mexico (61% to 68%), New York (55% to 62%), and Washington (57% to 64%), the AAP said in its weekly vaccination trends report.
Weekly cases of COVID-19 in children dropped for the first time since June, and daily hospitalizations appear to be falling, even as the pace of vaccinations continues to slow among the youngest eligible recipients, according to new data.

Despite the 3.3% decline from the previous week’s record high, the new-case count still topped 243,000 for the week of Sept. 3-9, putting the total number of cases in children at almost 5.3 million since the pandemic began.
Hospitalizations seem to have peaked on Sept. 4, when the rate for children aged 0-17 years reached 0.51 per 100,000 population. The admission rate for confirmed COVID-19 has dropped steadily since then and was down to 0.45 per 100,000 on Sept. 11, the last day for which preliminary data from the Centers for Disease Control and Prevention were available.
On the prevention side, fully vaccinated children aged 12-17 years represented 5.5% of all Americans who had completed the vaccine regimen as of Sept. 13. Vaccine initiation, however, has dropped for 5 consecutive weeks in 12- to 15-year-olds and in 4 of the last 5 weeks among 16- and 17-year-olds, the CDC said on its COVID Data Tracker.
Just under 199,000 children aged 12-15 received their first dose of the COVID-19 vaccine during the week of Sept. 7-13. That’s down by 18.5% from the week before and by 51.6% since Aug. 9, the last week that vaccine initiation increased for the age group. Among 16- and 17-year-olds, the 83,000 new recipients that week was a decrease of 25.7% from the previous week and a decline of 47% since the summer peak of Aug. 9, the CDC data show.
Those newest recipients bring at-least-one-dose status to 52.0% of those aged 12-15 and 59.9% of the 16- and 17-year-olds, while 40.3% and 48.9% were fully vaccinated as of Sept. 13. Corresponding figures for some of the older groups are 61.6%/49.7% (age 18-24 years), 73.8%/63.1% (40-49 years), and 95.1%/84.5% (65-74 years), the CDC said.
Vaccine coverage for children at the state level deviates considerably from the national averages. The highest rates for children aged 12-17 are to be found in Vermont, where 76% have received at least one dose, the AAP reported in a separate analysis. Massachusetts is just below that but also comes in at 76% by virtue of a rounding error. The other states in the top five are Connecticut (74%), Hawaii (73%), and Rhode Island (71%).
The lowest vaccination rate for children comes from Wyoming (29%), which is preceded by North Dakota (33%), West Virginia (33%), Alabama (33%), and Mississippi (34%). the AAP said based on data from the CDC, which does not include Idaho.
In a bit of a side note, West Virginia’s Republican governor, Jim Justice, recently said this about vaccine reluctance in his state: “For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the same very people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Over the last 3 weeks, the District of Columbia has had the largest increase in children having received at least one dose: 10 percentage points, as it went from 58% to 68%. The next-largest improvement – 7 percentage points – occurred in Georgia (34% to 41%), New Mexico (61% to 68%), New York (55% to 62%), and Washington (57% to 64%), the AAP said in its weekly vaccination trends report.
Weekly cases of COVID-19 in children dropped for the first time since June, and daily hospitalizations appear to be falling, even as the pace of vaccinations continues to slow among the youngest eligible recipients, according to new data.

Despite the 3.3% decline from the previous week’s record high, the new-case count still topped 243,000 for the week of Sept. 3-9, putting the total number of cases in children at almost 5.3 million since the pandemic began.
Hospitalizations seem to have peaked on Sept. 4, when the rate for children aged 0-17 years reached 0.51 per 100,000 population. The admission rate for confirmed COVID-19 has dropped steadily since then and was down to 0.45 per 100,000 on Sept. 11, the last day for which preliminary data from the Centers for Disease Control and Prevention were available.
On the prevention side, fully vaccinated children aged 12-17 years represented 5.5% of all Americans who had completed the vaccine regimen as of Sept. 13. Vaccine initiation, however, has dropped for 5 consecutive weeks in 12- to 15-year-olds and in 4 of the last 5 weeks among 16- and 17-year-olds, the CDC said on its COVID Data Tracker.
Just under 199,000 children aged 12-15 received their first dose of the COVID-19 vaccine during the week of Sept. 7-13. That’s down by 18.5% from the week before and by 51.6% since Aug. 9, the last week that vaccine initiation increased for the age group. Among 16- and 17-year-olds, the 83,000 new recipients that week was a decrease of 25.7% from the previous week and a decline of 47% since the summer peak of Aug. 9, the CDC data show.
Those newest recipients bring at-least-one-dose status to 52.0% of those aged 12-15 and 59.9% of the 16- and 17-year-olds, while 40.3% and 48.9% were fully vaccinated as of Sept. 13. Corresponding figures for some of the older groups are 61.6%/49.7% (age 18-24 years), 73.8%/63.1% (40-49 years), and 95.1%/84.5% (65-74 years), the CDC said.
Vaccine coverage for children at the state level deviates considerably from the national averages. The highest rates for children aged 12-17 are to be found in Vermont, where 76% have received at least one dose, the AAP reported in a separate analysis. Massachusetts is just below that but also comes in at 76% by virtue of a rounding error. The other states in the top five are Connecticut (74%), Hawaii (73%), and Rhode Island (71%).
The lowest vaccination rate for children comes from Wyoming (29%), which is preceded by North Dakota (33%), West Virginia (33%), Alabama (33%), and Mississippi (34%). the AAP said based on data from the CDC, which does not include Idaho.
In a bit of a side note, West Virginia’s Republican governor, Jim Justice, recently said this about vaccine reluctance in his state: “For God’s sakes a livin’, how difficult is this to understand? Why in the world do we have to come up with these crazy ideas – and they’re crazy ideas – that the vaccine’s got something in it and it’s tracing people wherever they go? And the same very people that are saying that are carrying their cellphones around. I mean, come on. Come on.”
Over the last 3 weeks, the District of Columbia has had the largest increase in children having received at least one dose: 10 percentage points, as it went from 58% to 68%. The next-largest improvement – 7 percentage points – occurred in Georgia (34% to 41%), New Mexico (61% to 68%), New York (55% to 62%), and Washington (57% to 64%), the AAP said in its weekly vaccination trends report.
PHM 2021: Achieving gender equity in medicine
PHM 2021 session
Accelerating Patient Care and Healthcare Workforce Diversity and Inclusion
Presenter
Julie Silver, MD
Session summary
Gender inequity in medicine has been well documented and further highlighted by the tremendous impact of the COVID-19 pandemic on women in medicine. While more women than men are entering medical schools across the U.S., women still struggle to reach the highest levels of academic rank, achieve leadership positions of power and influence, receive fair equitable pay, attain leadership roles in national societies, and receive funding from national agencies. They also continue to face discrimination and implicit and explicit biases. Women of color or from other minority backgrounds face even greater barriers and biases. Despite being a specialty in which women represent almost 70% of the workforce, pediatrics is not immune to these disparities.
In her PHM21 plenary on Aug. 3, 2021, Dr. Julie Silver, a national expert in gender equity disparities, detailed the landscape for women in medicine and proposed some solutions to accelerate systemic change for gender equity. In order to understand and mitigate gender inequity, Dr. Silver encouraged the PHM community to identify influential “gatekeepers” of promotion, advancement, and salary compensation. In academic medicine medical schools, funding agencies, professional societies, and journals are the gatekeepers to advancement and compensation for women. Women are traditionally underrepresented as members and influential leaders of these gatekeeping organizations and in their recognition structures, therefore their advancement, compensation, and wellbeing are hindered.
Key takeaways
- Critical mass theory will not help alleviate gender inequity in medicine, as women make up a critical mass in pediatrics and are still experiencing stark inequities. Critical actor leaders are needed to highlight disparities and drive change even once a critical mass is reached.
- Our current diversity, equity, and inclusion (DEI) efforts are ineffective and are creating an “illusion of fairness that causes majority group members to become less sensitive to recognizing discrimination against minorities.” Many of the activities that are considered citizenship, including committees focused on DEI efforts, should be counted as scholarship, and appropriately compensated to ensure promotion of our women and minority colleagues.
- Male allies are critical to documenting, disseminating, and addressing gender inequality. Without the support of men in the field, we will see little progress.
- While there are numerous advocacy angles we can take when advocating for gender equity, the most effective will be the financial angle. Gender pay gaps at the start of a career can lead to roughly 2 million dollars of salary loss for a woman over the course of her career. In order to alleviate those salary pay gaps our institutions must not expect women to negotiate for fair pay, make salary benchmarks transparent, continue to monitor and conduct research on compensation disparities, and attempt to alleviate the weight of educational debt.
- COVID-19 is causing immense stress on women in medicine, and the impact could be disastrous. We must recognize and reward the “4th shift” women are working for COVID-19–related activities at home and at work, and put measures in place to #GiveHerAReasonToStay in health care.
- Men and other women leaders have a responsibility to sponsor the many and well-qualified women in medicine for awards, committees, and speaking engagements. These opportunities are key markers of success in academic medicine and are critical to advancement and salary compensation.
Dr. Casillas is the internal medicine-pediatric chief resident for the University of Cincinnati/Cincinnati Children’s Internal Medicine-Pediatric program. His career goal is to serve as a hospitalist for children and adults, and he is interested in health equity and Latinx health. Dr. O’Toole is a pediatric and adult hospitalist at Cincinnati Children’s Hospital Medical Center and University of Cincinnati Medical Center, and a professor of pediatrics and internal medicine at the University of Cincinnati College of Medicine. She serves as program director of Cincinnati’s Combined Internal Medicine and Pediatrics Residency Program.
PHM 2021 session
Accelerating Patient Care and Healthcare Workforce Diversity and Inclusion
Presenter
Julie Silver, MD
Session summary
Gender inequity in medicine has been well documented and further highlighted by the tremendous impact of the COVID-19 pandemic on women in medicine. While more women than men are entering medical schools across the U.S., women still struggle to reach the highest levels of academic rank, achieve leadership positions of power and influence, receive fair equitable pay, attain leadership roles in national societies, and receive funding from national agencies. They also continue to face discrimination and implicit and explicit biases. Women of color or from other minority backgrounds face even greater barriers and biases. Despite being a specialty in which women represent almost 70% of the workforce, pediatrics is not immune to these disparities.
In her PHM21 plenary on Aug. 3, 2021, Dr. Julie Silver, a national expert in gender equity disparities, detailed the landscape for women in medicine and proposed some solutions to accelerate systemic change for gender equity. In order to understand and mitigate gender inequity, Dr. Silver encouraged the PHM community to identify influential “gatekeepers” of promotion, advancement, and salary compensation. In academic medicine medical schools, funding agencies, professional societies, and journals are the gatekeepers to advancement and compensation for women. Women are traditionally underrepresented as members and influential leaders of these gatekeeping organizations and in their recognition structures, therefore their advancement, compensation, and wellbeing are hindered.
Key takeaways
- Critical mass theory will not help alleviate gender inequity in medicine, as women make up a critical mass in pediatrics and are still experiencing stark inequities. Critical actor leaders are needed to highlight disparities and drive change even once a critical mass is reached.
- Our current diversity, equity, and inclusion (DEI) efforts are ineffective and are creating an “illusion of fairness that causes majority group members to become less sensitive to recognizing discrimination against minorities.” Many of the activities that are considered citizenship, including committees focused on DEI efforts, should be counted as scholarship, and appropriately compensated to ensure promotion of our women and minority colleagues.
- Male allies are critical to documenting, disseminating, and addressing gender inequality. Without the support of men in the field, we will see little progress.
- While there are numerous advocacy angles we can take when advocating for gender equity, the most effective will be the financial angle. Gender pay gaps at the start of a career can lead to roughly 2 million dollars of salary loss for a woman over the course of her career. In order to alleviate those salary pay gaps our institutions must not expect women to negotiate for fair pay, make salary benchmarks transparent, continue to monitor and conduct research on compensation disparities, and attempt to alleviate the weight of educational debt.
- COVID-19 is causing immense stress on women in medicine, and the impact could be disastrous. We must recognize and reward the “4th shift” women are working for COVID-19–related activities at home and at work, and put measures in place to #GiveHerAReasonToStay in health care.
- Men and other women leaders have a responsibility to sponsor the many and well-qualified women in medicine for awards, committees, and speaking engagements. These opportunities are key markers of success in academic medicine and are critical to advancement and salary compensation.
Dr. Casillas is the internal medicine-pediatric chief resident for the University of Cincinnati/Cincinnati Children’s Internal Medicine-Pediatric program. His career goal is to serve as a hospitalist for children and adults, and he is interested in health equity and Latinx health. Dr. O’Toole is a pediatric and adult hospitalist at Cincinnati Children’s Hospital Medical Center and University of Cincinnati Medical Center, and a professor of pediatrics and internal medicine at the University of Cincinnati College of Medicine. She serves as program director of Cincinnati’s Combined Internal Medicine and Pediatrics Residency Program.
PHM 2021 session
Accelerating Patient Care and Healthcare Workforce Diversity and Inclusion
Presenter
Julie Silver, MD
Session summary
Gender inequity in medicine has been well documented and further highlighted by the tremendous impact of the COVID-19 pandemic on women in medicine. While more women than men are entering medical schools across the U.S., women still struggle to reach the highest levels of academic rank, achieve leadership positions of power and influence, receive fair equitable pay, attain leadership roles in national societies, and receive funding from national agencies. They also continue to face discrimination and implicit and explicit biases. Women of color or from other minority backgrounds face even greater barriers and biases. Despite being a specialty in which women represent almost 70% of the workforce, pediatrics is not immune to these disparities.
In her PHM21 plenary on Aug. 3, 2021, Dr. Julie Silver, a national expert in gender equity disparities, detailed the landscape for women in medicine and proposed some solutions to accelerate systemic change for gender equity. In order to understand and mitigate gender inequity, Dr. Silver encouraged the PHM community to identify influential “gatekeepers” of promotion, advancement, and salary compensation. In academic medicine medical schools, funding agencies, professional societies, and journals are the gatekeepers to advancement and compensation for women. Women are traditionally underrepresented as members and influential leaders of these gatekeeping organizations and in their recognition structures, therefore their advancement, compensation, and wellbeing are hindered.
Key takeaways
- Critical mass theory will not help alleviate gender inequity in medicine, as women make up a critical mass in pediatrics and are still experiencing stark inequities. Critical actor leaders are needed to highlight disparities and drive change even once a critical mass is reached.
- Our current diversity, equity, and inclusion (DEI) efforts are ineffective and are creating an “illusion of fairness that causes majority group members to become less sensitive to recognizing discrimination against minorities.” Many of the activities that are considered citizenship, including committees focused on DEI efforts, should be counted as scholarship, and appropriately compensated to ensure promotion of our women and minority colleagues.
- Male allies are critical to documenting, disseminating, and addressing gender inequality. Without the support of men in the field, we will see little progress.
- While there are numerous advocacy angles we can take when advocating for gender equity, the most effective will be the financial angle. Gender pay gaps at the start of a career can lead to roughly 2 million dollars of salary loss for a woman over the course of her career. In order to alleviate those salary pay gaps our institutions must not expect women to negotiate for fair pay, make salary benchmarks transparent, continue to monitor and conduct research on compensation disparities, and attempt to alleviate the weight of educational debt.
- COVID-19 is causing immense stress on women in medicine, and the impact could be disastrous. We must recognize and reward the “4th shift” women are working for COVID-19–related activities at home and at work, and put measures in place to #GiveHerAReasonToStay in health care.
- Men and other women leaders have a responsibility to sponsor the many and well-qualified women in medicine for awards, committees, and speaking engagements. These opportunities are key markers of success in academic medicine and are critical to advancement and salary compensation.
Dr. Casillas is the internal medicine-pediatric chief resident for the University of Cincinnati/Cincinnati Children’s Internal Medicine-Pediatric program. His career goal is to serve as a hospitalist for children and adults, and he is interested in health equity and Latinx health. Dr. O’Toole is a pediatric and adult hospitalist at Cincinnati Children’s Hospital Medical Center and University of Cincinnati Medical Center, and a professor of pediatrics and internal medicine at the University of Cincinnati College of Medicine. She serves as program director of Cincinnati’s Combined Internal Medicine and Pediatrics Residency Program.
New CMS rule challenges hospitals, but not vendors, to make EHRs safer
In a little-noticed action last month, so as to meet an objective of the Medicare Promoting Interoperability Program, starting next year.
Experts praised the move but said that EHR developers should share the responsibility for ensuring that the use of their products doesn’t harm patients.
A number of safety problems are associated with hospital EHR systems, ranging from insufficient protection against medication errors and inadvertent turnoffs of drug interaction checkers to allowing physicians to use free text instead of coded data for key patient indicators. Although hospitals aren’t required to do anything about safety problems that turn up in their self-audits, practitioners who perform the self-assessment will likely encounter challenges that they were previously unaware of and will fix them, experts say.
Studies over the past decade have shown that improper configuration and use of EHRs, as well as design flaws in the systems, can cause avoidable patient injuries or can fail to prevent them. For example, one large study found that clinical decision support (CDS) features in EHRs prevented adverse drug events (ADEs) in only 61.6% of cases in 2016. That was an improvement over the ADE prevention rate of 54% in 2009. Nevertheless, nearly 40% of ADEs were not averted.
Another study, sponsored by the Leapfrog Group, found that EHRs used in U.S. hospitals failed to detect up to 1 in 3 potentially harmful drug interactions and other medication errors. In this study, about 10% of the detection failures were attributed to design problems in EHRs.
The new CMS measure requires hospitals to evaluate their EHRs using safety guides that were developed in 2014 and were revised in 2016 by the Office of the National Coordinator for Health IT (ONC). Known as the Safety Assurance Factors for Resilience (SAFER) guides, they include a set of recommendations to help health care organizations optimize the safety of EHRs.
Surprises in store for hospitals
Dean Sittig, PhD, a professor at the University of Texas Health Science Center, Houston, told this news organization that a 2018 study he conducted with his colleague Hardeep Singh, MD, MPH, found that eight surveyed health care organizations were following about 75% of the SAFER recommendations.
He said that when hospitals and health care systems start to assess their systems, many will be surprised at what they are not doing or not doing right. Although the new CMS rule doesn’t require them to correct deficiencies, he expects that many will.
For this reason, Dr. Sittig believes the requirement will have a positive effect on patient safety. But the regulation may not go far enough because it doesn’t impose any requirements on EHR vendors, he said.
In a commentary published in JAMA, Dr. Sittig and Dr. Hardeep, a professor at the Michael E. DeBakey VA Medical Center and Baylor College of Medicine, cite a study showing that 40% of “EHR-related products” had “nonconformities” with EHR certification regulations that could potentially harm patients. “Many nonconformities could have been identified by the developer prior to product release,” they say.
Shared responsibility
According to the JAMA commentary, the SAFER guides were developed “to help health care organizations and EHR developers conduct voluntary self-assessments to help eliminate or minimize EHR-related safety risks and hazards.”
In response to a query from this news organization, ONC confirmed that the SAFER guides were intended for use by developers as well as practitioners. ONC said it supports CMS’s approach to incentivize collaborations between EHR vendors and health care organizations. It noted that some entities have already teamed up to the meet the SAFER guides’ recommendations.
Hospitals and EHR developers must share responsibility for safety, Dr. Sittig and Dr. Singh argue, because many SAFER recommendations are based on EHR features that have to be programmed by developers.
For example, one recommendation is that patient identification information be displayed on all portions of the EHR user interface, wristbands, and printouts. Hospitals can’t implement this feature if the developer hasn’t built it into its product.
Dr. Sittig and Dr. Singh suggest three strategies to complement CMS’s new regulation:
- Because in their view, ONC’s EHR certification criteria are insufficient to address many patient safety concerns, CMS should require EHR developers to assess their products annually.
- ONC should conduct annual reviews of the SAFER recommendations with input from EHR developers and safety experts.
- EHR vendors should disseminate guidance to their customers on how to address safety practices, perhaps including EHR configuration guides related to safety.
Safety in EHR certification
At a recent press conference that ONC held to update reporters on its current plans, officials were asked to comment on Dr. Sittig’s and Dr. Singh’s proposition that EHR developers, as well as hospitals, do more to ensure system safety.
Steve Posnack, deputy national coordinator of health IT, noted that the ONC-supervised certification process requires developers to pay attention to how they “implement and integrate safety practices in their software design. We have certification criteria ... around what’s called safety-enhanced design – specific capabilities in the EHR that are sensitive to safety in areas like e-prescribing, medication, and high-risk events, where you want to make sure there’s more attention paid to the safety-related dynamics.”
After the conference, ONC told this news organization that among the safety-related certification criteria is one on user-centered design, which must be used in programming certain EHR features. Another is on the use of a quality management system to guide the creation of each EHR capability.
Nevertheless, there is evidence that not all EHR developers have paid sufficient attention to safety in their products. This is shown in the corporate integrity agreements with the Office of the Inspector General (OIG) of the Department of Health and Human Services (HHS) that developers eClinicalWorks and Greenway agreed to sign because, according to the government, they had not met all of the certification criteria they’d claimed to satisfy.
Under these agreements, the vendors agreed to follow “relevant standards, checklists, self-assessment tools, and other practices identified in the ONC SAFER guides and the ICE Report(s) to optimize the safety and safe use of EHRs” in a number of specific areas.
Even if all EHRs conformed to the certification requirements for safety, they would fall short of the SAFER recommendations, Dr. Sittig says. “Those certification criteria are pretty general and not as comprehensive as the SAFER guides. Some SAFER guide recommendations are in existing certification requirements, like you’re supposed to have drug-drug interaction checking capabilities, and they’re supposed to be on. But it doesn’t say you need to have the patient’s identification on every screen. It’s easy to assume good software design, development, and testing principles are a given, but our experience suggests otherwise.”
Configuration problems
A handful of vendors are working on what the JAMA article suggests, but there are about 1,000 EHR developers, Dr. Sittig notes. Moreover, there are configuration problems in the design of many EHRs, even if the products have the recommended features.
“For example, it’s often possible to meet the SAFER recommendations, but not all the vendors make that the default setting. That’s one of the things our paper says they should do,” Dr. Sittig says.
Conversely, some hospitals turn off certain features because they annoy doctors, he notes. For instance, the SAFER guides recommend that allergies, problem list entries, and diagnostic test results be entered and stored using standard, coded data elements in the EHR, but often the EHR makes it easier to enter free text data.
Default settings can be wiped out during system upgrades, he added. That has happened with drug interaction checkers. “If you don’t test the system after upgrades and reassess it annually, you might go several months without your drug-drug interaction checker on. And your doctors aren’t complaining about not getting alerts. Those kinds of mistakes are hard to catch.”
Some errors in an EHR may be caught fairly quickly, but in a health system that treats thousands of patients at any given time, those mistakes can still cause a lot of potential patient harm, Dr. Sittig points out. Some vendors, he says, are building tools to help health care organizations catch those errors through what is called “anomaly detection.” This is similar to what credit card companies do when they notice you’ve bought a carpet in Saudi Arabia, although you’ve never traveled abroad, he notes.
“You can look at alert firing data and notice that all of a sudden an alert fired 500 times today when it usually fires 10 times, or it stopped firing,” Dr. Sittig observes. “Those kinds of things should be built into all EHRs. That would be an excellent step forward.”
A version of this article first appeared on Medscape.com.
In a little-noticed action last month, so as to meet an objective of the Medicare Promoting Interoperability Program, starting next year.
Experts praised the move but said that EHR developers should share the responsibility for ensuring that the use of their products doesn’t harm patients.
A number of safety problems are associated with hospital EHR systems, ranging from insufficient protection against medication errors and inadvertent turnoffs of drug interaction checkers to allowing physicians to use free text instead of coded data for key patient indicators. Although hospitals aren’t required to do anything about safety problems that turn up in their self-audits, practitioners who perform the self-assessment will likely encounter challenges that they were previously unaware of and will fix them, experts say.
Studies over the past decade have shown that improper configuration and use of EHRs, as well as design flaws in the systems, can cause avoidable patient injuries or can fail to prevent them. For example, one large study found that clinical decision support (CDS) features in EHRs prevented adverse drug events (ADEs) in only 61.6% of cases in 2016. That was an improvement over the ADE prevention rate of 54% in 2009. Nevertheless, nearly 40% of ADEs were not averted.
Another study, sponsored by the Leapfrog Group, found that EHRs used in U.S. hospitals failed to detect up to 1 in 3 potentially harmful drug interactions and other medication errors. In this study, about 10% of the detection failures were attributed to design problems in EHRs.
The new CMS measure requires hospitals to evaluate their EHRs using safety guides that were developed in 2014 and were revised in 2016 by the Office of the National Coordinator for Health IT (ONC). Known as the Safety Assurance Factors for Resilience (SAFER) guides, they include a set of recommendations to help health care organizations optimize the safety of EHRs.
Surprises in store for hospitals
Dean Sittig, PhD, a professor at the University of Texas Health Science Center, Houston, told this news organization that a 2018 study he conducted with his colleague Hardeep Singh, MD, MPH, found that eight surveyed health care organizations were following about 75% of the SAFER recommendations.
He said that when hospitals and health care systems start to assess their systems, many will be surprised at what they are not doing or not doing right. Although the new CMS rule doesn’t require them to correct deficiencies, he expects that many will.
For this reason, Dr. Sittig believes the requirement will have a positive effect on patient safety. But the regulation may not go far enough because it doesn’t impose any requirements on EHR vendors, he said.
In a commentary published in JAMA, Dr. Sittig and Dr. Hardeep, a professor at the Michael E. DeBakey VA Medical Center and Baylor College of Medicine, cite a study showing that 40% of “EHR-related products” had “nonconformities” with EHR certification regulations that could potentially harm patients. “Many nonconformities could have been identified by the developer prior to product release,” they say.
Shared responsibility
According to the JAMA commentary, the SAFER guides were developed “to help health care organizations and EHR developers conduct voluntary self-assessments to help eliminate or minimize EHR-related safety risks and hazards.”
In response to a query from this news organization, ONC confirmed that the SAFER guides were intended for use by developers as well as practitioners. ONC said it supports CMS’s approach to incentivize collaborations between EHR vendors and health care organizations. It noted that some entities have already teamed up to the meet the SAFER guides’ recommendations.
Hospitals and EHR developers must share responsibility for safety, Dr. Sittig and Dr. Singh argue, because many SAFER recommendations are based on EHR features that have to be programmed by developers.
For example, one recommendation is that patient identification information be displayed on all portions of the EHR user interface, wristbands, and printouts. Hospitals can’t implement this feature if the developer hasn’t built it into its product.
Dr. Sittig and Dr. Singh suggest three strategies to complement CMS’s new regulation:
- Because in their view, ONC’s EHR certification criteria are insufficient to address many patient safety concerns, CMS should require EHR developers to assess their products annually.
- ONC should conduct annual reviews of the SAFER recommendations with input from EHR developers and safety experts.
- EHR vendors should disseminate guidance to their customers on how to address safety practices, perhaps including EHR configuration guides related to safety.
Safety in EHR certification
At a recent press conference that ONC held to update reporters on its current plans, officials were asked to comment on Dr. Sittig’s and Dr. Singh’s proposition that EHR developers, as well as hospitals, do more to ensure system safety.
Steve Posnack, deputy national coordinator of health IT, noted that the ONC-supervised certification process requires developers to pay attention to how they “implement and integrate safety practices in their software design. We have certification criteria ... around what’s called safety-enhanced design – specific capabilities in the EHR that are sensitive to safety in areas like e-prescribing, medication, and high-risk events, where you want to make sure there’s more attention paid to the safety-related dynamics.”
After the conference, ONC told this news organization that among the safety-related certification criteria is one on user-centered design, which must be used in programming certain EHR features. Another is on the use of a quality management system to guide the creation of each EHR capability.
Nevertheless, there is evidence that not all EHR developers have paid sufficient attention to safety in their products. This is shown in the corporate integrity agreements with the Office of the Inspector General (OIG) of the Department of Health and Human Services (HHS) that developers eClinicalWorks and Greenway agreed to sign because, according to the government, they had not met all of the certification criteria they’d claimed to satisfy.
Under these agreements, the vendors agreed to follow “relevant standards, checklists, self-assessment tools, and other practices identified in the ONC SAFER guides and the ICE Report(s) to optimize the safety and safe use of EHRs” in a number of specific areas.
Even if all EHRs conformed to the certification requirements for safety, they would fall short of the SAFER recommendations, Dr. Sittig says. “Those certification criteria are pretty general and not as comprehensive as the SAFER guides. Some SAFER guide recommendations are in existing certification requirements, like you’re supposed to have drug-drug interaction checking capabilities, and they’re supposed to be on. But it doesn’t say you need to have the patient’s identification on every screen. It’s easy to assume good software design, development, and testing principles are a given, but our experience suggests otherwise.”
Configuration problems
A handful of vendors are working on what the JAMA article suggests, but there are about 1,000 EHR developers, Dr. Sittig notes. Moreover, there are configuration problems in the design of many EHRs, even if the products have the recommended features.
“For example, it’s often possible to meet the SAFER recommendations, but not all the vendors make that the default setting. That’s one of the things our paper says they should do,” Dr. Sittig says.
Conversely, some hospitals turn off certain features because they annoy doctors, he notes. For instance, the SAFER guides recommend that allergies, problem list entries, and diagnostic test results be entered and stored using standard, coded data elements in the EHR, but often the EHR makes it easier to enter free text data.
Default settings can be wiped out during system upgrades, he added. That has happened with drug interaction checkers. “If you don’t test the system after upgrades and reassess it annually, you might go several months without your drug-drug interaction checker on. And your doctors aren’t complaining about not getting alerts. Those kinds of mistakes are hard to catch.”
Some errors in an EHR may be caught fairly quickly, but in a health system that treats thousands of patients at any given time, those mistakes can still cause a lot of potential patient harm, Dr. Sittig points out. Some vendors, he says, are building tools to help health care organizations catch those errors through what is called “anomaly detection.” This is similar to what credit card companies do when they notice you’ve bought a carpet in Saudi Arabia, although you’ve never traveled abroad, he notes.
“You can look at alert firing data and notice that all of a sudden an alert fired 500 times today when it usually fires 10 times, or it stopped firing,” Dr. Sittig observes. “Those kinds of things should be built into all EHRs. That would be an excellent step forward.”
A version of this article first appeared on Medscape.com.
In a little-noticed action last month, so as to meet an objective of the Medicare Promoting Interoperability Program, starting next year.
Experts praised the move but said that EHR developers should share the responsibility for ensuring that the use of their products doesn’t harm patients.
A number of safety problems are associated with hospital EHR systems, ranging from insufficient protection against medication errors and inadvertent turnoffs of drug interaction checkers to allowing physicians to use free text instead of coded data for key patient indicators. Although hospitals aren’t required to do anything about safety problems that turn up in their self-audits, practitioners who perform the self-assessment will likely encounter challenges that they were previously unaware of and will fix them, experts say.
Studies over the past decade have shown that improper configuration and use of EHRs, as well as design flaws in the systems, can cause avoidable patient injuries or can fail to prevent them. For example, one large study found that clinical decision support (CDS) features in EHRs prevented adverse drug events (ADEs) in only 61.6% of cases in 2016. That was an improvement over the ADE prevention rate of 54% in 2009. Nevertheless, nearly 40% of ADEs were not averted.
Another study, sponsored by the Leapfrog Group, found that EHRs used in U.S. hospitals failed to detect up to 1 in 3 potentially harmful drug interactions and other medication errors. In this study, about 10% of the detection failures were attributed to design problems in EHRs.
The new CMS measure requires hospitals to evaluate their EHRs using safety guides that were developed in 2014 and were revised in 2016 by the Office of the National Coordinator for Health IT (ONC). Known as the Safety Assurance Factors for Resilience (SAFER) guides, they include a set of recommendations to help health care organizations optimize the safety of EHRs.
Surprises in store for hospitals
Dean Sittig, PhD, a professor at the University of Texas Health Science Center, Houston, told this news organization that a 2018 study he conducted with his colleague Hardeep Singh, MD, MPH, found that eight surveyed health care organizations were following about 75% of the SAFER recommendations.
He said that when hospitals and health care systems start to assess their systems, many will be surprised at what they are not doing or not doing right. Although the new CMS rule doesn’t require them to correct deficiencies, he expects that many will.
For this reason, Dr. Sittig believes the requirement will have a positive effect on patient safety. But the regulation may not go far enough because it doesn’t impose any requirements on EHR vendors, he said.
In a commentary published in JAMA, Dr. Sittig and Dr. Hardeep, a professor at the Michael E. DeBakey VA Medical Center and Baylor College of Medicine, cite a study showing that 40% of “EHR-related products” had “nonconformities” with EHR certification regulations that could potentially harm patients. “Many nonconformities could have been identified by the developer prior to product release,” they say.
Shared responsibility
According to the JAMA commentary, the SAFER guides were developed “to help health care organizations and EHR developers conduct voluntary self-assessments to help eliminate or minimize EHR-related safety risks and hazards.”
In response to a query from this news organization, ONC confirmed that the SAFER guides were intended for use by developers as well as practitioners. ONC said it supports CMS’s approach to incentivize collaborations between EHR vendors and health care organizations. It noted that some entities have already teamed up to the meet the SAFER guides’ recommendations.
Hospitals and EHR developers must share responsibility for safety, Dr. Sittig and Dr. Singh argue, because many SAFER recommendations are based on EHR features that have to be programmed by developers.
For example, one recommendation is that patient identification information be displayed on all portions of the EHR user interface, wristbands, and printouts. Hospitals can’t implement this feature if the developer hasn’t built it into its product.
Dr. Sittig and Dr. Singh suggest three strategies to complement CMS’s new regulation:
- Because in their view, ONC’s EHR certification criteria are insufficient to address many patient safety concerns, CMS should require EHR developers to assess their products annually.
- ONC should conduct annual reviews of the SAFER recommendations with input from EHR developers and safety experts.
- EHR vendors should disseminate guidance to their customers on how to address safety practices, perhaps including EHR configuration guides related to safety.
Safety in EHR certification
At a recent press conference that ONC held to update reporters on its current plans, officials were asked to comment on Dr. Sittig’s and Dr. Singh’s proposition that EHR developers, as well as hospitals, do more to ensure system safety.
Steve Posnack, deputy national coordinator of health IT, noted that the ONC-supervised certification process requires developers to pay attention to how they “implement and integrate safety practices in their software design. We have certification criteria ... around what’s called safety-enhanced design – specific capabilities in the EHR that are sensitive to safety in areas like e-prescribing, medication, and high-risk events, where you want to make sure there’s more attention paid to the safety-related dynamics.”
After the conference, ONC told this news organization that among the safety-related certification criteria is one on user-centered design, which must be used in programming certain EHR features. Another is on the use of a quality management system to guide the creation of each EHR capability.
Nevertheless, there is evidence that not all EHR developers have paid sufficient attention to safety in their products. This is shown in the corporate integrity agreements with the Office of the Inspector General (OIG) of the Department of Health and Human Services (HHS) that developers eClinicalWorks and Greenway agreed to sign because, according to the government, they had not met all of the certification criteria they’d claimed to satisfy.
Under these agreements, the vendors agreed to follow “relevant standards, checklists, self-assessment tools, and other practices identified in the ONC SAFER guides and the ICE Report(s) to optimize the safety and safe use of EHRs” in a number of specific areas.
Even if all EHRs conformed to the certification requirements for safety, they would fall short of the SAFER recommendations, Dr. Sittig says. “Those certification criteria are pretty general and not as comprehensive as the SAFER guides. Some SAFER guide recommendations are in existing certification requirements, like you’re supposed to have drug-drug interaction checking capabilities, and they’re supposed to be on. But it doesn’t say you need to have the patient’s identification on every screen. It’s easy to assume good software design, development, and testing principles are a given, but our experience suggests otherwise.”
Configuration problems
A handful of vendors are working on what the JAMA article suggests, but there are about 1,000 EHR developers, Dr. Sittig notes. Moreover, there are configuration problems in the design of many EHRs, even if the products have the recommended features.
“For example, it’s often possible to meet the SAFER recommendations, but not all the vendors make that the default setting. That’s one of the things our paper says they should do,” Dr. Sittig says.
Conversely, some hospitals turn off certain features because they annoy doctors, he notes. For instance, the SAFER guides recommend that allergies, problem list entries, and diagnostic test results be entered and stored using standard, coded data elements in the EHR, but often the EHR makes it easier to enter free text data.
Default settings can be wiped out during system upgrades, he added. That has happened with drug interaction checkers. “If you don’t test the system after upgrades and reassess it annually, you might go several months without your drug-drug interaction checker on. And your doctors aren’t complaining about not getting alerts. Those kinds of mistakes are hard to catch.”
Some errors in an EHR may be caught fairly quickly, but in a health system that treats thousands of patients at any given time, those mistakes can still cause a lot of potential patient harm, Dr. Sittig points out. Some vendors, he says, are building tools to help health care organizations catch those errors through what is called “anomaly detection.” This is similar to what credit card companies do when they notice you’ve bought a carpet in Saudi Arabia, although you’ve never traveled abroad, he notes.
“You can look at alert firing data and notice that all of a sudden an alert fired 500 times today when it usually fires 10 times, or it stopped firing,” Dr. Sittig observes. “Those kinds of things should be built into all EHRs. That would be an excellent step forward.”
A version of this article first appeared on Medscape.com.
Antibiotic use and colon cancer: More evidence of link
The latest data come from a Swedish population study. Investigators analyzed data from more than 40,000 colorectal cancer patients and 200,000 cancer-free control persons.
They found that moderate use of antibiotics increased the risk for proximal colon cancer by 9% and that very high antibiotic use increased the risk by 17%.
In contrast, the risk for rectal cancer was reduced by 4% with moderate use and 9% with very high use, but this association was confined to women.
Antibiotic use was categorized as no use (no reported use of antibiotics during the study period), low (use during a period of 1-10 days), moderate (11-60 days), high (61-180 days), and very high (>180 days).
The study, led by Sophia Harlid, PhD, department of radiation sciences, oncology, Umeå University, Sweden, was published online on Sept. 1 in the Journal of the National Cancer Institute.
The results complement findings from a recent study from Scotland, which found that a history of antibiotic use among individuals younger than 50 appeared to increase the risk of developing colon cancer but not rectal cancer by 49%.
The new data from Sweden “strengthen prior evidence and provide new insights into site-specific carcinogenesis as well as indirect support for the role of gut microbiota,” lead author Dr. Dr. Harlid commented in an interview.
“The positive associations between antibiotics use and proximal colon cancer began at the lowest level of antibiotics use, providing a potential justification for reducing antibiotics prescriptions in clinical practice,” she added.
In their article, the team suggests that the increased risk could be a result of antibiotics having a “disruptive effect” on the gut microbiome.
The finding of an increased risk for cancer in the proximal colon but not further along the alimentary tract “is consistent with a high microbial impact in the proximal colon and a decreasing concentration of short-chain fatty acids along the colon,” the authors comment.
This results “in higher bacterial activity, biofilm formation, and fermentation in the proximal compared with the distal colon and rectum.”
A further analysis showed that the use of quinolones and sulfonamides and/or trimethoprims was associated with an increased risk for proximal colon cancer, whereas use of nitrofurantoins, macrolides and/or lincosamides, and metronidazoles and/or tinidazoles was inversely associated with rectal cancer.
Details of the study findings
For their study, the team analyzed complete-population data from Swedish national registers for the period July 1, 2005 to Dec. 31, 2016.
They matched case patients who were diagnosed with colorectal cancer from Jan. 1, 2010 to Dec. 31, 2016 with cancer-free control persons in a 1:5 ratio. Data on antibiotic use were extracted from the Swedish Prescribed Drug Register.
Other variables, such as socioeconomic factors and health care utilization, were obtained from the Swedish Inpatient Register and the Longitudinal Integration Database for Health Insurance and Labor Market Studies.
The team identified 40,545 patients with colorectal cancer cases; there were 202,720 control persons. Just over half (52.9%) of the participants were men; the mean age at cancer diagnosis was 72 years. Among the cases, 36.4% were proximal colon cancers, 29.3% were distal colon cancers, and 33.0% rectal cancers.
Control patients were more likely to have been prescribed no antibiotics, at 22.4% versus 18.7% for case patients. Case patients were more likely than control persons to have used antibiotics for more than 2 months, at 20.8% versus 19.3% (P < .001).
Overall, antibiotic use was positively associated with colorectal cancer. In comparison with no use, the odds ratio for moderate use was 1.15; for very high use, it was 1.17 (P < .001 for trend).
Excluding all antibiotic use during the 2 years prior to a colorectal cancer diagnosis attenuated the association, such that it was no longer significant for very high use versus no antibiotic use.
Applying this cutoff to the remaining analyses, the team found that the dose-response relationship between antibiotic use and colorectal cancer was largely confined to proximal colon cancer, at an odds ratio of 1.09 for moderate use and 1.17 for very high use in comparison with no use (P < .001 for trend).
For distal colon cancer, the relationship was “close to null.”
There was a slight inverse relationship between rectal cancer and antibiotic use, at an odds rate of 0.96 for moderate use and 0.91 for very high use versus no use (P < .001 for trend). This association was found in women only, whereas the other associations were seen in both men and women.
The study was supported by the Lion’s Cancer Research Foundation, Umeå University, and Region Västerbotten. Dr. Harlid has disclosed no relevant financial relationships. Three coauthors report various relationships with industry, as noted in the original article.
A version of this article first appeared on Medscape.com.
The latest data come from a Swedish population study. Investigators analyzed data from more than 40,000 colorectal cancer patients and 200,000 cancer-free control persons.
They found that moderate use of antibiotics increased the risk for proximal colon cancer by 9% and that very high antibiotic use increased the risk by 17%.
In contrast, the risk for rectal cancer was reduced by 4% with moderate use and 9% with very high use, but this association was confined to women.
Antibiotic use was categorized as no use (no reported use of antibiotics during the study period), low (use during a period of 1-10 days), moderate (11-60 days), high (61-180 days), and very high (>180 days).
The study, led by Sophia Harlid, PhD, department of radiation sciences, oncology, Umeå University, Sweden, was published online on Sept. 1 in the Journal of the National Cancer Institute.
The results complement findings from a recent study from Scotland, which found that a history of antibiotic use among individuals younger than 50 appeared to increase the risk of developing colon cancer but not rectal cancer by 49%.
The new data from Sweden “strengthen prior evidence and provide new insights into site-specific carcinogenesis as well as indirect support for the role of gut microbiota,” lead author Dr. Dr. Harlid commented in an interview.
“The positive associations between antibiotics use and proximal colon cancer began at the lowest level of antibiotics use, providing a potential justification for reducing antibiotics prescriptions in clinical practice,” she added.
In their article, the team suggests that the increased risk could be a result of antibiotics having a “disruptive effect” on the gut microbiome.
The finding of an increased risk for cancer in the proximal colon but not further along the alimentary tract “is consistent with a high microbial impact in the proximal colon and a decreasing concentration of short-chain fatty acids along the colon,” the authors comment.
This results “in higher bacterial activity, biofilm formation, and fermentation in the proximal compared with the distal colon and rectum.”
A further analysis showed that the use of quinolones and sulfonamides and/or trimethoprims was associated with an increased risk for proximal colon cancer, whereas use of nitrofurantoins, macrolides and/or lincosamides, and metronidazoles and/or tinidazoles was inversely associated with rectal cancer.
Details of the study findings
For their study, the team analyzed complete-population data from Swedish national registers for the period July 1, 2005 to Dec. 31, 2016.
They matched case patients who were diagnosed with colorectal cancer from Jan. 1, 2010 to Dec. 31, 2016 with cancer-free control persons in a 1:5 ratio. Data on antibiotic use were extracted from the Swedish Prescribed Drug Register.
Other variables, such as socioeconomic factors and health care utilization, were obtained from the Swedish Inpatient Register and the Longitudinal Integration Database for Health Insurance and Labor Market Studies.
The team identified 40,545 patients with colorectal cancer cases; there were 202,720 control persons. Just over half (52.9%) of the participants were men; the mean age at cancer diagnosis was 72 years. Among the cases, 36.4% were proximal colon cancers, 29.3% were distal colon cancers, and 33.0% rectal cancers.
Control patients were more likely to have been prescribed no antibiotics, at 22.4% versus 18.7% for case patients. Case patients were more likely than control persons to have used antibiotics for more than 2 months, at 20.8% versus 19.3% (P < .001).
Overall, antibiotic use was positively associated with colorectal cancer. In comparison with no use, the odds ratio for moderate use was 1.15; for very high use, it was 1.17 (P < .001 for trend).
Excluding all antibiotic use during the 2 years prior to a colorectal cancer diagnosis attenuated the association, such that it was no longer significant for very high use versus no antibiotic use.
Applying this cutoff to the remaining analyses, the team found that the dose-response relationship between antibiotic use and colorectal cancer was largely confined to proximal colon cancer, at an odds ratio of 1.09 for moderate use and 1.17 for very high use in comparison with no use (P < .001 for trend).
For distal colon cancer, the relationship was “close to null.”
There was a slight inverse relationship between rectal cancer and antibiotic use, at an odds rate of 0.96 for moderate use and 0.91 for very high use versus no use (P < .001 for trend). This association was found in women only, whereas the other associations were seen in both men and women.
The study was supported by the Lion’s Cancer Research Foundation, Umeå University, and Region Västerbotten. Dr. Harlid has disclosed no relevant financial relationships. Three coauthors report various relationships with industry, as noted in the original article.
A version of this article first appeared on Medscape.com.
The latest data come from a Swedish population study. Investigators analyzed data from more than 40,000 colorectal cancer patients and 200,000 cancer-free control persons.
They found that moderate use of antibiotics increased the risk for proximal colon cancer by 9% and that very high antibiotic use increased the risk by 17%.
In contrast, the risk for rectal cancer was reduced by 4% with moderate use and 9% with very high use, but this association was confined to women.
Antibiotic use was categorized as no use (no reported use of antibiotics during the study period), low (use during a period of 1-10 days), moderate (11-60 days), high (61-180 days), and very high (>180 days).
The study, led by Sophia Harlid, PhD, department of radiation sciences, oncology, Umeå University, Sweden, was published online on Sept. 1 in the Journal of the National Cancer Institute.
The results complement findings from a recent study from Scotland, which found that a history of antibiotic use among individuals younger than 50 appeared to increase the risk of developing colon cancer but not rectal cancer by 49%.
The new data from Sweden “strengthen prior evidence and provide new insights into site-specific carcinogenesis as well as indirect support for the role of gut microbiota,” lead author Dr. Dr. Harlid commented in an interview.
“The positive associations between antibiotics use and proximal colon cancer began at the lowest level of antibiotics use, providing a potential justification for reducing antibiotics prescriptions in clinical practice,” she added.
In their article, the team suggests that the increased risk could be a result of antibiotics having a “disruptive effect” on the gut microbiome.
The finding of an increased risk for cancer in the proximal colon but not further along the alimentary tract “is consistent with a high microbial impact in the proximal colon and a decreasing concentration of short-chain fatty acids along the colon,” the authors comment.
This results “in higher bacterial activity, biofilm formation, and fermentation in the proximal compared with the distal colon and rectum.”
A further analysis showed that the use of quinolones and sulfonamides and/or trimethoprims was associated with an increased risk for proximal colon cancer, whereas use of nitrofurantoins, macrolides and/or lincosamides, and metronidazoles and/or tinidazoles was inversely associated with rectal cancer.
Details of the study findings
For their study, the team analyzed complete-population data from Swedish national registers for the period July 1, 2005 to Dec. 31, 2016.
They matched case patients who were diagnosed with colorectal cancer from Jan. 1, 2010 to Dec. 31, 2016 with cancer-free control persons in a 1:5 ratio. Data on antibiotic use were extracted from the Swedish Prescribed Drug Register.
Other variables, such as socioeconomic factors and health care utilization, were obtained from the Swedish Inpatient Register and the Longitudinal Integration Database for Health Insurance and Labor Market Studies.
The team identified 40,545 patients with colorectal cancer cases; there were 202,720 control persons. Just over half (52.9%) of the participants were men; the mean age at cancer diagnosis was 72 years. Among the cases, 36.4% were proximal colon cancers, 29.3% were distal colon cancers, and 33.0% rectal cancers.
Control patients were more likely to have been prescribed no antibiotics, at 22.4% versus 18.7% for case patients. Case patients were more likely than control persons to have used antibiotics for more than 2 months, at 20.8% versus 19.3% (P < .001).
Overall, antibiotic use was positively associated with colorectal cancer. In comparison with no use, the odds ratio for moderate use was 1.15; for very high use, it was 1.17 (P < .001 for trend).
Excluding all antibiotic use during the 2 years prior to a colorectal cancer diagnosis attenuated the association, such that it was no longer significant for very high use versus no antibiotic use.
Applying this cutoff to the remaining analyses, the team found that the dose-response relationship between antibiotic use and colorectal cancer was largely confined to proximal colon cancer, at an odds ratio of 1.09 for moderate use and 1.17 for very high use in comparison with no use (P < .001 for trend).
For distal colon cancer, the relationship was “close to null.”
There was a slight inverse relationship between rectal cancer and antibiotic use, at an odds rate of 0.96 for moderate use and 0.91 for very high use versus no use (P < .001 for trend). This association was found in women only, whereas the other associations were seen in both men and women.
The study was supported by the Lion’s Cancer Research Foundation, Umeå University, and Region Västerbotten. Dr. Harlid has disclosed no relevant financial relationships. Three coauthors report various relationships with industry, as noted in the original article.
A version of this article first appeared on Medscape.com.
Man dies after 43 full ICUs turn him away
Ray Martin DeMonia, 73, of Cullman, Alabama, ran an antiques business for 40 years and served as an auctioneer at charity events, the obituary said.
He had a stroke in 2020 during the first months of the COVID pandemic and made sure to get vaccinated, his daughter, Raven DeMonia, told The Washington Post.
“He knew what the vaccine meant for his health and what it meant to staying alive,” she said. “He said, ‘I just want to get back to shaking hands with people, selling stuff, and talking antiques.’”
His daughter told the Post that her father went to Cullman Regional Medical Center on Aug. 23 with heart problems.
About 12 hours after he was admitted, her mother got a call from the hospital saying they’d called 43 hospitals and were unable to find a “specialized cardiac ICU bed” for him, Ms. DeMonia told the Post.
He was finally airlifted to Rush Foundation Hospital in Meridian, Mississippi, almost 200 miles from his home, but died there Sept. 1. His family decided to make a plea for increased vaccinations in his obituary.
“In honor of Ray, please get vaccinated if you have not, in an effort to free up resources for non COVID related emergencies,” the obit said. “Due to COVID 19, CRMC emergency staff contacted 43 hospitals in 3 states in search of a Cardiac ICU bed and finally located one in Meridian, MS. He would not want any other family to go through what his did.”
Mr. DeMonia is survived by his wife, daughter, grandson, and other family members.
The Alabama Hospital Association says state hospitals are still short of ICU beds. On Sept. 12, the AHA website said the state had 1,530 staffed ICU beds to accommodate 1,541 ICU patients.
The AHA said 83% of COVID patients in ICU had not been vaccinated against COVID, 4% were partially vaccinated, and 13% were fully vaccinated. Alabama trails other states in vaccination rates. Newsweek, citing CDC data, said 53.7% of people in Alabama were fully vaccinated. In comparison, 53.8% of all Americans nationally are fully vaccinated.
A version of this article first appeared on WebMD.com.
Ray Martin DeMonia, 73, of Cullman, Alabama, ran an antiques business for 40 years and served as an auctioneer at charity events, the obituary said.
He had a stroke in 2020 during the first months of the COVID pandemic and made sure to get vaccinated, his daughter, Raven DeMonia, told The Washington Post.
“He knew what the vaccine meant for his health and what it meant to staying alive,” she said. “He said, ‘I just want to get back to shaking hands with people, selling stuff, and talking antiques.’”
His daughter told the Post that her father went to Cullman Regional Medical Center on Aug. 23 with heart problems.
About 12 hours after he was admitted, her mother got a call from the hospital saying they’d called 43 hospitals and were unable to find a “specialized cardiac ICU bed” for him, Ms. DeMonia told the Post.
He was finally airlifted to Rush Foundation Hospital in Meridian, Mississippi, almost 200 miles from his home, but died there Sept. 1. His family decided to make a plea for increased vaccinations in his obituary.
“In honor of Ray, please get vaccinated if you have not, in an effort to free up resources for non COVID related emergencies,” the obit said. “Due to COVID 19, CRMC emergency staff contacted 43 hospitals in 3 states in search of a Cardiac ICU bed and finally located one in Meridian, MS. He would not want any other family to go through what his did.”
Mr. DeMonia is survived by his wife, daughter, grandson, and other family members.
The Alabama Hospital Association says state hospitals are still short of ICU beds. On Sept. 12, the AHA website said the state had 1,530 staffed ICU beds to accommodate 1,541 ICU patients.
The AHA said 83% of COVID patients in ICU had not been vaccinated against COVID, 4% were partially vaccinated, and 13% were fully vaccinated. Alabama trails other states in vaccination rates. Newsweek, citing CDC data, said 53.7% of people in Alabama were fully vaccinated. In comparison, 53.8% of all Americans nationally are fully vaccinated.
A version of this article first appeared on WebMD.com.
Ray Martin DeMonia, 73, of Cullman, Alabama, ran an antiques business for 40 years and served as an auctioneer at charity events, the obituary said.
He had a stroke in 2020 during the first months of the COVID pandemic and made sure to get vaccinated, his daughter, Raven DeMonia, told The Washington Post.
“He knew what the vaccine meant for his health and what it meant to staying alive,” she said. “He said, ‘I just want to get back to shaking hands with people, selling stuff, and talking antiques.’”
His daughter told the Post that her father went to Cullman Regional Medical Center on Aug. 23 with heart problems.
About 12 hours after he was admitted, her mother got a call from the hospital saying they’d called 43 hospitals and were unable to find a “specialized cardiac ICU bed” for him, Ms. DeMonia told the Post.
He was finally airlifted to Rush Foundation Hospital in Meridian, Mississippi, almost 200 miles from his home, but died there Sept. 1. His family decided to make a plea for increased vaccinations in his obituary.
“In honor of Ray, please get vaccinated if you have not, in an effort to free up resources for non COVID related emergencies,” the obit said. “Due to COVID 19, CRMC emergency staff contacted 43 hospitals in 3 states in search of a Cardiac ICU bed and finally located one in Meridian, MS. He would not want any other family to go through what his did.”
Mr. DeMonia is survived by his wife, daughter, grandson, and other family members.
The Alabama Hospital Association says state hospitals are still short of ICU beds. On Sept. 12, the AHA website said the state had 1,530 staffed ICU beds to accommodate 1,541 ICU patients.
The AHA said 83% of COVID patients in ICU had not been vaccinated against COVID, 4% were partially vaccinated, and 13% were fully vaccinated. Alabama trails other states in vaccination rates. Newsweek, citing CDC data, said 53.7% of people in Alabama were fully vaccinated. In comparison, 53.8% of all Americans nationally are fully vaccinated.
A version of this article first appeared on WebMD.com.
PRESERVED-HF: Dapagliflozin improves physical limitations in patients with HFpEF
The SGLT2 inhibitor dapagliflozin scored a clear win in a randomized, controlled trial with more than 300 U.S. patients with heart failure with preserved ejection fraction (HFpEF), showing a significant and clinically meaningful benefit for the primary endpoint, a KCCQ measure of symptoms and physical limitations, after 12 weeks of treatment.
These results in the PRESERVED-HF study follow closely on the heals of the initial report from the EMPEROR-Preserved trial that showed a benefit from a different sodium-glucose cotransporter 2 (SGLT2) inhibitor, empagliflozin (Jardiance) in nearly 6,000 randomized patients for the primary endpoint of preventing cardiovascular death or hospitalizations for heart failure.
In PRESERVED-HF, patients with HFpEF who received a standard, once-daily dose of dapagliflozin (Farxiga) had an average 5.8-point improvement in their condition as measured by the Kansas City Cardiomyopathy Questionnaire clinical summary score (KCCQ-CS), the study’s primary endpoint.
This is “the first study to demonstrate that an SGLT2 inhibitor dapagliflozin significantly improves symptoms, physical limitations, and 6-minute walking distance in patients with HFpEF,” Mikhail N. Kosiborod, MD, reported at the annual scientific meeting of the Heart Failure Society of America. The secondary endpoint of 6-minute walking distance “has been very difficult to improve in many previous studies of other treatments” tested in patients with HFpEF, noted Dr. Kosiborod, a cardiologist and codirector of the Cardiometabolic Center of Excellence at Saint Luke’s Mid-America Heart Institute.
The results are “highly complementary” to the findings from large outcome trials, such as the findings from EMPEROR-Preserved, he said, and collectively the recent findings from these studies of SGLT2 inhibitors in patients with HFpEF identify drugs in this class as a “new treatment option” for patients with a disorder that until now had no treatment with unequivocally proven efficacy and safety.
‘Impressive and unprecedented’ findings
The findings are “really impressive and unprecedented,” said Milton Packer, MD, a cardiologist at Baylor University Medical Center in Dallas who was not involved in the study. “This is the largest KCCQ benefit ever seen in either patients with HFpEF or in patients with heart failure with reduced ejection fraction,” said Dr. Packer, one of the investigators who led the EMPEROR-Preserved trial.
PRESERVED-HF randomized 324 patients diagnosed with heart failure and with a left ventricular ejection fraction of 45% or higher at any of 26 U.S. centers, with 304 patients completing the planned final analysis after 12 weeks on treatment. Patients could be in New York Heart Association (NYHA) functional class II-IV, they had to have a baseline N-terminal pro-brain natriuretic peptide (NT-proBNP) level of at least 225 pg/mL (or higher if they also had atrial fibrillation), and they required at least one of three markers of established heart failure: recent hospitalization for heart failure or an urgent outpatient visit that required treatment with an IV diuretic, elevated filling pressure measured by left or right catheterization, or structural heart disease detected by echocardiography.
The average age of the enrolled patients was 70 years, and they had been diagnosed with heart failure for about 3 years; 57% were women, 30% were African American, and their median body mass index was 35 kg/m2. Roughly 42% had NYHA class III or IV disease, 56% had type 2 diabetes, their median estimated glomerular filtration rate was about 55 mL/min per 1.73m2, their median KCCQ-CS score at baseline was about 62, and their average 6-minute walk distance was 244 m.
These and other features of the enrolled population define a distinctly U.S. patient population, stressed Dr. Kosiborod, professor of medicine at the University of Missouri–Kansas City.
“The patients we enrolled are the patients we see in U.S. clinical practice,” he said in an interview. Importantly, the patient profile of a median BMI of 35 kg/m2, a median KCCQ-CS score of 62 – “quite low,” noted Dr. Kosiborod – and having more than 40% of patients in NYHA functional class III defines a study population with a substantially greater burden of obesity, symptoms, and functional impairment compared with those enrolled in prior trials involving patients with HFpEF such as EMPEROR-Preserved.
Results complement findings from larger trials
PRESERVED-HF was an investigator-initiated study designed to inform clinical practice, not as a pivotal trial like EMPEROR-Preserved, which aims to gather evidence to support a new indication for regulatory approval. (On Sept. 9, 2021, the Food and Drug Administration granted empagliflozin “breakthrough therapy” status for treating HFpEF based on the EMPEROR-Preserved results, which will fast-track the agency’s decision on this indication.)
Dr. Kosiborod noted that he and his associates designed PRESERVED-HF with adequate patient numbers to power a statistically valid assessment of effect on KCCQ-CS score. While the new findings will not by themselves lead to a new indication for dapagliflozin to treat patients with HFpEF, they will potentially complement the pending results of another trial, DELIVER, by showing efficacy and safety in a uniquely U.S. patient population. DELIVER is a pivotal, global trial of dapagliflozin in more than 6,000 patients with HFpEF that’s on track to report findings in 2022.
Dr. Kosiborod also stressed that dapagliflozin has U.S.-approved indications for treating patients with type 2 diabetes, and for patients with chronic kidney disease, and that a majority of patients enrolled in PRESERVED-HF had one or both of these conditions. That makes the new findings especially compelling for patients with either type 2 diabetes or chronic kidney disease and HFpEF who are not already receiving an SGLT2 inhibitor.
Other findings that he reported showed a range of benefits consistent with the primary endpoint, including the KCCQ overall summary score, which also showed a significant 4.5-point average increase over placebo after 12 weeks. Analysis by the percentage of patients achieving at least a 5-point improvement in the KCCQ clinical summary score (the threshold for a clinically meaningful improvement) showed that about 45% of patients treated with dapagliflozin reached this mark compared with roughly 35% of patients in the placebo arm, indicating a number needed to treat of nine to have one additional patient achieve this threshold after 12 weeks. Average improvement in 6-minute walk distance was about 20 m with dapagliflozin compared with placebo.
No heterogeneity of effect by baseline ejection fraction.
Subgroup analyses showed no heterogeneity of response across 12 different ways of subdividing the study population, including age, sex, race, diabetes status, and BMI. The median left ventricular ejection fraction among enrolled patients was 60%, and the findings showed identical KCCQ improvements among patients with ejection fractions less than the median and those with an ejection fraction above the median.
This last finding was especially relevant because the EMPEROR-Preserved results showed a possible signal of heterogeneity by ejection fraction and an attenuated effect among patients with HFpEF and an ejection fraction above the 60%-65% range, although the certainty of this finding is currently controversial.
The impact of empagliflozin on KCCQ clinical summary score in EMPEROR-Preserved showed an average incremental improvement of 1.32 points compared with placebo, a significant difference, but more modest than the increment from dapagliflozin treatment seen in PRESERVED-HF. Dr. Kosiborod hypothesized that this difference might be mostly because of the different patient populations enrolled in the two studies.
Dr. Kosiborod noted that a report on the PRESERVED-HF results will soon appear in Nature Medicine.
PRESERVED-HF was funded by AstraZeneca, which markets dapagliflozin (Farxiga), but the trials’ design and conduct were independent of this funding source. Dr. Kosiborod has been a consultant to AstraZeneca and numerous other companies, and he has received research funding from AstraZeneca and Boehringer Ingelheim. Dr. Packer has had financial relationships with AstraZeneca and numerous other companies.
The SGLT2 inhibitor dapagliflozin scored a clear win in a randomized, controlled trial with more than 300 U.S. patients with heart failure with preserved ejection fraction (HFpEF), showing a significant and clinically meaningful benefit for the primary endpoint, a KCCQ measure of symptoms and physical limitations, after 12 weeks of treatment.
These results in the PRESERVED-HF study follow closely on the heals of the initial report from the EMPEROR-Preserved trial that showed a benefit from a different sodium-glucose cotransporter 2 (SGLT2) inhibitor, empagliflozin (Jardiance) in nearly 6,000 randomized patients for the primary endpoint of preventing cardiovascular death or hospitalizations for heart failure.
In PRESERVED-HF, patients with HFpEF who received a standard, once-daily dose of dapagliflozin (Farxiga) had an average 5.8-point improvement in their condition as measured by the Kansas City Cardiomyopathy Questionnaire clinical summary score (KCCQ-CS), the study’s primary endpoint.
This is “the first study to demonstrate that an SGLT2 inhibitor dapagliflozin significantly improves symptoms, physical limitations, and 6-minute walking distance in patients with HFpEF,” Mikhail N. Kosiborod, MD, reported at the annual scientific meeting of the Heart Failure Society of America. The secondary endpoint of 6-minute walking distance “has been very difficult to improve in many previous studies of other treatments” tested in patients with HFpEF, noted Dr. Kosiborod, a cardiologist and codirector of the Cardiometabolic Center of Excellence at Saint Luke’s Mid-America Heart Institute.
The results are “highly complementary” to the findings from large outcome trials, such as the findings from EMPEROR-Preserved, he said, and collectively the recent findings from these studies of SGLT2 inhibitors in patients with HFpEF identify drugs in this class as a “new treatment option” for patients with a disorder that until now had no treatment with unequivocally proven efficacy and safety.
‘Impressive and unprecedented’ findings
The findings are “really impressive and unprecedented,” said Milton Packer, MD, a cardiologist at Baylor University Medical Center in Dallas who was not involved in the study. “This is the largest KCCQ benefit ever seen in either patients with HFpEF or in patients with heart failure with reduced ejection fraction,” said Dr. Packer, one of the investigators who led the EMPEROR-Preserved trial.
PRESERVED-HF randomized 324 patients diagnosed with heart failure and with a left ventricular ejection fraction of 45% or higher at any of 26 U.S. centers, with 304 patients completing the planned final analysis after 12 weeks on treatment. Patients could be in New York Heart Association (NYHA) functional class II-IV, they had to have a baseline N-terminal pro-brain natriuretic peptide (NT-proBNP) level of at least 225 pg/mL (or higher if they also had atrial fibrillation), and they required at least one of three markers of established heart failure: recent hospitalization for heart failure or an urgent outpatient visit that required treatment with an IV diuretic, elevated filling pressure measured by left or right catheterization, or structural heart disease detected by echocardiography.
The average age of the enrolled patients was 70 years, and they had been diagnosed with heart failure for about 3 years; 57% were women, 30% were African American, and their median body mass index was 35 kg/m2. Roughly 42% had NYHA class III or IV disease, 56% had type 2 diabetes, their median estimated glomerular filtration rate was about 55 mL/min per 1.73m2, their median KCCQ-CS score at baseline was about 62, and their average 6-minute walk distance was 244 m.
These and other features of the enrolled population define a distinctly U.S. patient population, stressed Dr. Kosiborod, professor of medicine at the University of Missouri–Kansas City.
“The patients we enrolled are the patients we see in U.S. clinical practice,” he said in an interview. Importantly, the patient profile of a median BMI of 35 kg/m2, a median KCCQ-CS score of 62 – “quite low,” noted Dr. Kosiborod – and having more than 40% of patients in NYHA functional class III defines a study population with a substantially greater burden of obesity, symptoms, and functional impairment compared with those enrolled in prior trials involving patients with HFpEF such as EMPEROR-Preserved.
Results complement findings from larger trials
PRESERVED-HF was an investigator-initiated study designed to inform clinical practice, not as a pivotal trial like EMPEROR-Preserved, which aims to gather evidence to support a new indication for regulatory approval. (On Sept. 9, 2021, the Food and Drug Administration granted empagliflozin “breakthrough therapy” status for treating HFpEF based on the EMPEROR-Preserved results, which will fast-track the agency’s decision on this indication.)
Dr. Kosiborod noted that he and his associates designed PRESERVED-HF with adequate patient numbers to power a statistically valid assessment of effect on KCCQ-CS score. While the new findings will not by themselves lead to a new indication for dapagliflozin to treat patients with HFpEF, they will potentially complement the pending results of another trial, DELIVER, by showing efficacy and safety in a uniquely U.S. patient population. DELIVER is a pivotal, global trial of dapagliflozin in more than 6,000 patients with HFpEF that’s on track to report findings in 2022.
Dr. Kosiborod also stressed that dapagliflozin has U.S.-approved indications for treating patients with type 2 diabetes, and for patients with chronic kidney disease, and that a majority of patients enrolled in PRESERVED-HF had one or both of these conditions. That makes the new findings especially compelling for patients with either type 2 diabetes or chronic kidney disease and HFpEF who are not already receiving an SGLT2 inhibitor.
Other findings that he reported showed a range of benefits consistent with the primary endpoint, including the KCCQ overall summary score, which also showed a significant 4.5-point average increase over placebo after 12 weeks. Analysis by the percentage of patients achieving at least a 5-point improvement in the KCCQ clinical summary score (the threshold for a clinically meaningful improvement) showed that about 45% of patients treated with dapagliflozin reached this mark compared with roughly 35% of patients in the placebo arm, indicating a number needed to treat of nine to have one additional patient achieve this threshold after 12 weeks. Average improvement in 6-minute walk distance was about 20 m with dapagliflozin compared with placebo.
No heterogeneity of effect by baseline ejection fraction.
Subgroup analyses showed no heterogeneity of response across 12 different ways of subdividing the study population, including age, sex, race, diabetes status, and BMI. The median left ventricular ejection fraction among enrolled patients was 60%, and the findings showed identical KCCQ improvements among patients with ejection fractions less than the median and those with an ejection fraction above the median.
This last finding was especially relevant because the EMPEROR-Preserved results showed a possible signal of heterogeneity by ejection fraction and an attenuated effect among patients with HFpEF and an ejection fraction above the 60%-65% range, although the certainty of this finding is currently controversial.
The impact of empagliflozin on KCCQ clinical summary score in EMPEROR-Preserved showed an average incremental improvement of 1.32 points compared with placebo, a significant difference, but more modest than the increment from dapagliflozin treatment seen in PRESERVED-HF. Dr. Kosiborod hypothesized that this difference might be mostly because of the different patient populations enrolled in the two studies.
Dr. Kosiborod noted that a report on the PRESERVED-HF results will soon appear in Nature Medicine.
PRESERVED-HF was funded by AstraZeneca, which markets dapagliflozin (Farxiga), but the trials’ design and conduct were independent of this funding source. Dr. Kosiborod has been a consultant to AstraZeneca and numerous other companies, and he has received research funding from AstraZeneca and Boehringer Ingelheim. Dr. Packer has had financial relationships with AstraZeneca and numerous other companies.
The SGLT2 inhibitor dapagliflozin scored a clear win in a randomized, controlled trial with more than 300 U.S. patients with heart failure with preserved ejection fraction (HFpEF), showing a significant and clinically meaningful benefit for the primary endpoint, a KCCQ measure of symptoms and physical limitations, after 12 weeks of treatment.
These results in the PRESERVED-HF study follow closely on the heals of the initial report from the EMPEROR-Preserved trial that showed a benefit from a different sodium-glucose cotransporter 2 (SGLT2) inhibitor, empagliflozin (Jardiance) in nearly 6,000 randomized patients for the primary endpoint of preventing cardiovascular death or hospitalizations for heart failure.
In PRESERVED-HF, patients with HFpEF who received a standard, once-daily dose of dapagliflozin (Farxiga) had an average 5.8-point improvement in their condition as measured by the Kansas City Cardiomyopathy Questionnaire clinical summary score (KCCQ-CS), the study’s primary endpoint.
This is “the first study to demonstrate that an SGLT2 inhibitor dapagliflozin significantly improves symptoms, physical limitations, and 6-minute walking distance in patients with HFpEF,” Mikhail N. Kosiborod, MD, reported at the annual scientific meeting of the Heart Failure Society of America. The secondary endpoint of 6-minute walking distance “has been very difficult to improve in many previous studies of other treatments” tested in patients with HFpEF, noted Dr. Kosiborod, a cardiologist and codirector of the Cardiometabolic Center of Excellence at Saint Luke’s Mid-America Heart Institute.
The results are “highly complementary” to the findings from large outcome trials, such as the findings from EMPEROR-Preserved, he said, and collectively the recent findings from these studies of SGLT2 inhibitors in patients with HFpEF identify drugs in this class as a “new treatment option” for patients with a disorder that until now had no treatment with unequivocally proven efficacy and safety.
‘Impressive and unprecedented’ findings
The findings are “really impressive and unprecedented,” said Milton Packer, MD, a cardiologist at Baylor University Medical Center in Dallas who was not involved in the study. “This is the largest KCCQ benefit ever seen in either patients with HFpEF or in patients with heart failure with reduced ejection fraction,” said Dr. Packer, one of the investigators who led the EMPEROR-Preserved trial.
PRESERVED-HF randomized 324 patients diagnosed with heart failure and with a left ventricular ejection fraction of 45% or higher at any of 26 U.S. centers, with 304 patients completing the planned final analysis after 12 weeks on treatment. Patients could be in New York Heart Association (NYHA) functional class II-IV, they had to have a baseline N-terminal pro-brain natriuretic peptide (NT-proBNP) level of at least 225 pg/mL (or higher if they also had atrial fibrillation), and they required at least one of three markers of established heart failure: recent hospitalization for heart failure or an urgent outpatient visit that required treatment with an IV diuretic, elevated filling pressure measured by left or right catheterization, or structural heart disease detected by echocardiography.
The average age of the enrolled patients was 70 years, and they had been diagnosed with heart failure for about 3 years; 57% were women, 30% were African American, and their median body mass index was 35 kg/m2. Roughly 42% had NYHA class III or IV disease, 56% had type 2 diabetes, their median estimated glomerular filtration rate was about 55 mL/min per 1.73m2, their median KCCQ-CS score at baseline was about 62, and their average 6-minute walk distance was 244 m.
These and other features of the enrolled population define a distinctly U.S. patient population, stressed Dr. Kosiborod, professor of medicine at the University of Missouri–Kansas City.
“The patients we enrolled are the patients we see in U.S. clinical practice,” he said in an interview. Importantly, the patient profile of a median BMI of 35 kg/m2, a median KCCQ-CS score of 62 – “quite low,” noted Dr. Kosiborod – and having more than 40% of patients in NYHA functional class III defines a study population with a substantially greater burden of obesity, symptoms, and functional impairment compared with those enrolled in prior trials involving patients with HFpEF such as EMPEROR-Preserved.
Results complement findings from larger trials
PRESERVED-HF was an investigator-initiated study designed to inform clinical practice, not as a pivotal trial like EMPEROR-Preserved, which aims to gather evidence to support a new indication for regulatory approval. (On Sept. 9, 2021, the Food and Drug Administration granted empagliflozin “breakthrough therapy” status for treating HFpEF based on the EMPEROR-Preserved results, which will fast-track the agency’s decision on this indication.)
Dr. Kosiborod noted that he and his associates designed PRESERVED-HF with adequate patient numbers to power a statistically valid assessment of effect on KCCQ-CS score. While the new findings will not by themselves lead to a new indication for dapagliflozin to treat patients with HFpEF, they will potentially complement the pending results of another trial, DELIVER, by showing efficacy and safety in a uniquely U.S. patient population. DELIVER is a pivotal, global trial of dapagliflozin in more than 6,000 patients with HFpEF that’s on track to report findings in 2022.
Dr. Kosiborod also stressed that dapagliflozin has U.S.-approved indications for treating patients with type 2 diabetes, and for patients with chronic kidney disease, and that a majority of patients enrolled in PRESERVED-HF had one or both of these conditions. That makes the new findings especially compelling for patients with either type 2 diabetes or chronic kidney disease and HFpEF who are not already receiving an SGLT2 inhibitor.
Other findings that he reported showed a range of benefits consistent with the primary endpoint, including the KCCQ overall summary score, which also showed a significant 4.5-point average increase over placebo after 12 weeks. Analysis by the percentage of patients achieving at least a 5-point improvement in the KCCQ clinical summary score (the threshold for a clinically meaningful improvement) showed that about 45% of patients treated with dapagliflozin reached this mark compared with roughly 35% of patients in the placebo arm, indicating a number needed to treat of nine to have one additional patient achieve this threshold after 12 weeks. Average improvement in 6-minute walk distance was about 20 m with dapagliflozin compared with placebo.
No heterogeneity of effect by baseline ejection fraction.
Subgroup analyses showed no heterogeneity of response across 12 different ways of subdividing the study population, including age, sex, race, diabetes status, and BMI. The median left ventricular ejection fraction among enrolled patients was 60%, and the findings showed identical KCCQ improvements among patients with ejection fractions less than the median and those with an ejection fraction above the median.
This last finding was especially relevant because the EMPEROR-Preserved results showed a possible signal of heterogeneity by ejection fraction and an attenuated effect among patients with HFpEF and an ejection fraction above the 60%-65% range, although the certainty of this finding is currently controversial.
The impact of empagliflozin on KCCQ clinical summary score in EMPEROR-Preserved showed an average incremental improvement of 1.32 points compared with placebo, a significant difference, but more modest than the increment from dapagliflozin treatment seen in PRESERVED-HF. Dr. Kosiborod hypothesized that this difference might be mostly because of the different patient populations enrolled in the two studies.
Dr. Kosiborod noted that a report on the PRESERVED-HF results will soon appear in Nature Medicine.
PRESERVED-HF was funded by AstraZeneca, which markets dapagliflozin (Farxiga), but the trials’ design and conduct were independent of this funding source. Dr. Kosiborod has been a consultant to AstraZeneca and numerous other companies, and he has received research funding from AstraZeneca and Boehringer Ingelheim. Dr. Packer has had financial relationships with AstraZeneca and numerous other companies.
FROM HFSA 2021
Fewer inpatient work hours linked with worse patient outcomes
The number of physicians working part time in the United States has increased by nearly 11% since 1993, and as more physicians opt for part-time work, quality of care deserves further study, the investigators wrote in JAMA Internal Medicine. Most studies comparing outcomes for patients treated by full-timers and part-timers have focused on outpatient care settings, where mortality is low and the potential for confounding is high, according to the study authors Hirotaka Kato, PhD, of Keio University in Tokyo, and colleagues. The new study, in contrast, is based on data from nearly 400,000 hospitalizations.
The researchers conducted a cross-sectional analysis on a 20% random sample of Medicare patients aged 65 years and older who were treated by a hospitalist for an emergency medical condition between 2011 and 2016. They examined associations between the number of days per year worked by hospitalists and they 30-day mortality rates among the patients they treated. The researchers analyzed a total of 392,797 hospitalizations in which patients were treated by 19,170 hospitalists. The mean age of the hospitalists was 41 years; 39% were female. Clinician work days were divided into quartiles.
Overall, the 30-day mortality was significantly higher among patients treated by clinicians in the bottom quartile with the fewest number of days worked, compared with those treated by clinicians in the top quartile with the most days worked (10.5% vs. 9.6%). The rates were similar in the second and third quartiles (10.0% and 9.5%).
The average number of days worked clinically per year was 57.6 in the lowest quartile versus 163.3 in the highest quartile, a 65% difference. No significant associations were noted between days worked and patient outcomes with regard to physician age, gender, or hospital teaching status.
Hospital 30-day readmission rates were examined as a secondary outcome, but there was no association between patient readmission and the number of days worked by the clinician. The adjusted 30-day readmission rate for clinicians in the bottom quartile of days worked, compared with those in the top quartile, was 15.3% versus 15.2% (P = .61).
The researchers found no difference in patients’ severity of illness (defined by expected mortality) or reason for admission between physicians in the different quartiles of days worked. They eliminated confounding from hospital-level differences by comparing outcomes of patients between physicians in the same hospital.
Possible explanations for worse patient outcomes
“As the number of physicians who engage in part-time clinical work continues to increase, these findings should lead to careful consideration by health systems to reevaluate preventive measures to address potential unintended patient harm,” the researchers wrote.
The researchers proposed several reasons for the association between fewer clinical work days and worse patient outcomes. First, physicians putting in less clinical time may be less updated on the latest guidelines, their skills may decline with less frequent patient care, and they may be less familiar with the nurses, medical assistants, and support staff, which may contribute to poor teamwork. The researchers also stated that some part-time physicians may need to balance nonclinical responsibilities, such as research or administrative tasks, concurrently with inpatient care. “It is also possible that physicians with less clinical knowledge or skills select to become part-time physicians, whereas physicians with higher clinical performance decide to work full time,” they noted.
The study findings were limited by several factors including the observational design and potential for unmeasured confounding variables, and the results may not generalize to younger patients or surgical patients, the researchers noted. Also, the study did not include care by hospitalists that was not billed, days in which clinicians treated non-Medicare patients or patients not part of the Medicare sample, or information about the reasons for clinicians’ part-time work.
However, the results were strengthened by the large sample size, and suggest the need for better institutional support to maintain the clinical performance of physicians who may be balancing a range of obligations, they concluded.
Clinician work issues have renewed relevance
“The data in this paper are from 2016 and earlier, but it is possibly event more relevant today than then,” Eileen Barrett, MD, of the University of New Mexico, Albuquerque, said in an interview. “The pandemic has exacerbated stressors being experienced by physicians and other health care workers, including higher clinical workloads and burnout, and spotlighted gendered effects on women in the workforce, which is likely to drive more physicians to part-time work.
“Reporting these findings now is so important so they can contribute to a shared mental model of the challenges physicians and hospitals face as we seek solutions to deliver high-quality and high-value care with an engaged, professionally fulfilled workforce,” she emphasized.
Dr. Barrett said she was surprised that the study did not show differences in readmission rates depending on the number of shifts worked, and also that the results were not different when considering expected mortality.
“However unpopular it may be to say so, physicians and administrators should assume these results apply to their practice unless they have examined their own data and know it does not,” Dr. Barrett said. “With that in mind, hospitals, administrators, and regulatory bodies have an urgent need to examine and reduce the forces driving physicians to part-time clinical work. Some of these factors include the absence of childcare, excessive paperwork, burnout, administrative duties, and valued experiences such as teaching, leadership, and research that keep clinicians from the bedside.
“Additionally, steps should be taken to reduce the administrative complexity that makes providing the best care to patients difficult and requires hospitalists to create ‘workarounds,’ because those who work fewer clinical hours may not know how to do these, nor how to advocate for their patients,” Dr. Barrett emphasized.
“Additional research is needed to determine how mortality varies by number of clinical shifts for pediatric and obstetric patients who are infrequently covered by Medicare, also how the pandemic and increasing administrative complexity since the time the data was obtained affect patient care,” Dr. Barrett noted.
The study was supported by a grant from the Japan Society for the Promotion of Science to lead author Dr. Kato, who had no financial conflicts to disclose. Dr. Barrett, who serves on the editorial advisory board of Internal Medicine News, had no financial conflicts.
The number of physicians working part time in the United States has increased by nearly 11% since 1993, and as more physicians opt for part-time work, quality of care deserves further study, the investigators wrote in JAMA Internal Medicine. Most studies comparing outcomes for patients treated by full-timers and part-timers have focused on outpatient care settings, where mortality is low and the potential for confounding is high, according to the study authors Hirotaka Kato, PhD, of Keio University in Tokyo, and colleagues. The new study, in contrast, is based on data from nearly 400,000 hospitalizations.
The researchers conducted a cross-sectional analysis on a 20% random sample of Medicare patients aged 65 years and older who were treated by a hospitalist for an emergency medical condition between 2011 and 2016. They examined associations between the number of days per year worked by hospitalists and they 30-day mortality rates among the patients they treated. The researchers analyzed a total of 392,797 hospitalizations in which patients were treated by 19,170 hospitalists. The mean age of the hospitalists was 41 years; 39% were female. Clinician work days were divided into quartiles.
Overall, the 30-day mortality was significantly higher among patients treated by clinicians in the bottom quartile with the fewest number of days worked, compared with those treated by clinicians in the top quartile with the most days worked (10.5% vs. 9.6%). The rates were similar in the second and third quartiles (10.0% and 9.5%).
The average number of days worked clinically per year was 57.6 in the lowest quartile versus 163.3 in the highest quartile, a 65% difference. No significant associations were noted between days worked and patient outcomes with regard to physician age, gender, or hospital teaching status.
Hospital 30-day readmission rates were examined as a secondary outcome, but there was no association between patient readmission and the number of days worked by the clinician. The adjusted 30-day readmission rate for clinicians in the bottom quartile of days worked, compared with those in the top quartile, was 15.3% versus 15.2% (P = .61).
The researchers found no difference in patients’ severity of illness (defined by expected mortality) or reason for admission between physicians in the different quartiles of days worked. They eliminated confounding from hospital-level differences by comparing outcomes of patients between physicians in the same hospital.
Possible explanations for worse patient outcomes
“As the number of physicians who engage in part-time clinical work continues to increase, these findings should lead to careful consideration by health systems to reevaluate preventive measures to address potential unintended patient harm,” the researchers wrote.
The researchers proposed several reasons for the association between fewer clinical work days and worse patient outcomes. First, physicians putting in less clinical time may be less updated on the latest guidelines, their skills may decline with less frequent patient care, and they may be less familiar with the nurses, medical assistants, and support staff, which may contribute to poor teamwork. The researchers also stated that some part-time physicians may need to balance nonclinical responsibilities, such as research or administrative tasks, concurrently with inpatient care. “It is also possible that physicians with less clinical knowledge or skills select to become part-time physicians, whereas physicians with higher clinical performance decide to work full time,” they noted.
The study findings were limited by several factors including the observational design and potential for unmeasured confounding variables, and the results may not generalize to younger patients or surgical patients, the researchers noted. Also, the study did not include care by hospitalists that was not billed, days in which clinicians treated non-Medicare patients or patients not part of the Medicare sample, or information about the reasons for clinicians’ part-time work.
However, the results were strengthened by the large sample size, and suggest the need for better institutional support to maintain the clinical performance of physicians who may be balancing a range of obligations, they concluded.
Clinician work issues have renewed relevance
“The data in this paper are from 2016 and earlier, but it is possibly event more relevant today than then,” Eileen Barrett, MD, of the University of New Mexico, Albuquerque, said in an interview. “The pandemic has exacerbated stressors being experienced by physicians and other health care workers, including higher clinical workloads and burnout, and spotlighted gendered effects on women in the workforce, which is likely to drive more physicians to part-time work.
“Reporting these findings now is so important so they can contribute to a shared mental model of the challenges physicians and hospitals face as we seek solutions to deliver high-quality and high-value care with an engaged, professionally fulfilled workforce,” she emphasized.
Dr. Barrett said she was surprised that the study did not show differences in readmission rates depending on the number of shifts worked, and also that the results were not different when considering expected mortality.
“However unpopular it may be to say so, physicians and administrators should assume these results apply to their practice unless they have examined their own data and know it does not,” Dr. Barrett said. “With that in mind, hospitals, administrators, and regulatory bodies have an urgent need to examine and reduce the forces driving physicians to part-time clinical work. Some of these factors include the absence of childcare, excessive paperwork, burnout, administrative duties, and valued experiences such as teaching, leadership, and research that keep clinicians from the bedside.
“Additionally, steps should be taken to reduce the administrative complexity that makes providing the best care to patients difficult and requires hospitalists to create ‘workarounds,’ because those who work fewer clinical hours may not know how to do these, nor how to advocate for their patients,” Dr. Barrett emphasized.
“Additional research is needed to determine how mortality varies by number of clinical shifts for pediatric and obstetric patients who are infrequently covered by Medicare, also how the pandemic and increasing administrative complexity since the time the data was obtained affect patient care,” Dr. Barrett noted.
The study was supported by a grant from the Japan Society for the Promotion of Science to lead author Dr. Kato, who had no financial conflicts to disclose. Dr. Barrett, who serves on the editorial advisory board of Internal Medicine News, had no financial conflicts.
The number of physicians working part time in the United States has increased by nearly 11% since 1993, and as more physicians opt for part-time work, quality of care deserves further study, the investigators wrote in JAMA Internal Medicine. Most studies comparing outcomes for patients treated by full-timers and part-timers have focused on outpatient care settings, where mortality is low and the potential for confounding is high, according to the study authors Hirotaka Kato, PhD, of Keio University in Tokyo, and colleagues. The new study, in contrast, is based on data from nearly 400,000 hospitalizations.
The researchers conducted a cross-sectional analysis on a 20% random sample of Medicare patients aged 65 years and older who were treated by a hospitalist for an emergency medical condition between 2011 and 2016. They examined associations between the number of days per year worked by hospitalists and they 30-day mortality rates among the patients they treated. The researchers analyzed a total of 392,797 hospitalizations in which patients were treated by 19,170 hospitalists. The mean age of the hospitalists was 41 years; 39% were female. Clinician work days were divided into quartiles.
Overall, the 30-day mortality was significantly higher among patients treated by clinicians in the bottom quartile with the fewest number of days worked, compared with those treated by clinicians in the top quartile with the most days worked (10.5% vs. 9.6%). The rates were similar in the second and third quartiles (10.0% and 9.5%).
The average number of days worked clinically per year was 57.6 in the lowest quartile versus 163.3 in the highest quartile, a 65% difference. No significant associations were noted between days worked and patient outcomes with regard to physician age, gender, or hospital teaching status.
Hospital 30-day readmission rates were examined as a secondary outcome, but there was no association between patient readmission and the number of days worked by the clinician. The adjusted 30-day readmission rate for clinicians in the bottom quartile of days worked, compared with those in the top quartile, was 15.3% versus 15.2% (P = .61).
The researchers found no difference in patients’ severity of illness (defined by expected mortality) or reason for admission between physicians in the different quartiles of days worked. They eliminated confounding from hospital-level differences by comparing outcomes of patients between physicians in the same hospital.
Possible explanations for worse patient outcomes
“As the number of physicians who engage in part-time clinical work continues to increase, these findings should lead to careful consideration by health systems to reevaluate preventive measures to address potential unintended patient harm,” the researchers wrote.
The researchers proposed several reasons for the association between fewer clinical work days and worse patient outcomes. First, physicians putting in less clinical time may be less updated on the latest guidelines, their skills may decline with less frequent patient care, and they may be less familiar with the nurses, medical assistants, and support staff, which may contribute to poor teamwork. The researchers also stated that some part-time physicians may need to balance nonclinical responsibilities, such as research or administrative tasks, concurrently with inpatient care. “It is also possible that physicians with less clinical knowledge or skills select to become part-time physicians, whereas physicians with higher clinical performance decide to work full time,” they noted.
The study findings were limited by several factors including the observational design and potential for unmeasured confounding variables, and the results may not generalize to younger patients or surgical patients, the researchers noted. Also, the study did not include care by hospitalists that was not billed, days in which clinicians treated non-Medicare patients or patients not part of the Medicare sample, or information about the reasons for clinicians’ part-time work.
However, the results were strengthened by the large sample size, and suggest the need for better institutional support to maintain the clinical performance of physicians who may be balancing a range of obligations, they concluded.
Clinician work issues have renewed relevance
“The data in this paper are from 2016 and earlier, but it is possibly event more relevant today than then,” Eileen Barrett, MD, of the University of New Mexico, Albuquerque, said in an interview. “The pandemic has exacerbated stressors being experienced by physicians and other health care workers, including higher clinical workloads and burnout, and spotlighted gendered effects on women in the workforce, which is likely to drive more physicians to part-time work.
“Reporting these findings now is so important so they can contribute to a shared mental model of the challenges physicians and hospitals face as we seek solutions to deliver high-quality and high-value care with an engaged, professionally fulfilled workforce,” she emphasized.
Dr. Barrett said she was surprised that the study did not show differences in readmission rates depending on the number of shifts worked, and also that the results were not different when considering expected mortality.
“However unpopular it may be to say so, physicians and administrators should assume these results apply to their practice unless they have examined their own data and know it does not,” Dr. Barrett said. “With that in mind, hospitals, administrators, and regulatory bodies have an urgent need to examine and reduce the forces driving physicians to part-time clinical work. Some of these factors include the absence of childcare, excessive paperwork, burnout, administrative duties, and valued experiences such as teaching, leadership, and research that keep clinicians from the bedside.
“Additionally, steps should be taken to reduce the administrative complexity that makes providing the best care to patients difficult and requires hospitalists to create ‘workarounds,’ because those who work fewer clinical hours may not know how to do these, nor how to advocate for their patients,” Dr. Barrett emphasized.
“Additional research is needed to determine how mortality varies by number of clinical shifts for pediatric and obstetric patients who are infrequently covered by Medicare, also how the pandemic and increasing administrative complexity since the time the data was obtained affect patient care,” Dr. Barrett noted.
The study was supported by a grant from the Japan Society for the Promotion of Science to lead author Dr. Kato, who had no financial conflicts to disclose. Dr. Barrett, who serves on the editorial advisory board of Internal Medicine News, had no financial conflicts.
FROM JAMA INTERNAL MEDICINE
Obese children with asthma are resistant to ICS
Obese or overweight children with asthma could be using inhaled corticosteroids (ICS) to no avail, combined results from observational studies suggest.
Using Mendelian randomization, a method for reducing bias in observational studies, investigators from the University of Amsterdam Medical Center performed an analysis of data from four cross-sectional studies and one cohort study on a total of 1,511 children with asthma.
They showed that every 1-unit increase in the body mass index (BMI) z score was associated with a more than twofold higher odds ratio for exacerbation, reported Cristina Longo, PhD, a former postdoctoral fellow at AMC, and assistant professor of medicine at the University of Montreal.
“In this large, multicenter Mendelian randomization study, our findings support current evidence that children with higher BMI status respond inadequately to inhaled corticosteroids, and that this association is likely not explained by measured confounding or reverse causation,” she said in an oral abstract presentation during the European Respiratory Society International Congress.
Unmeasured confounding
The obese-asthma phenotype in children is characterized by reduced lung function, high symptom expression, poor response to ICS, and high health care utilization.
“While most observational studies suggest that weight status is associated with asthma exacerbations, despite using inhaled corticosteroids, it’s unclear whether these associations may be due to unmeasured confounding or reverse causation, which captures the idea that perhaps obesity is a consequence rather than a cause of uncontrolled severe asthma,” she said.
Traditional observational studies of the obesity-asthma link rely on comparing data on asthma in a target population and comparing nonobese patients with obese patients. The problem with this method, Dr. Longo contended, is that the exposure assignment – weight status – is not random, and could lead to bias from potential imbalance of confounders, leading to unintentionally biased results.
In contrast, Mendelian randomization uses genetic data to approximate random assignment of exposures, using a risk score for BMI based on genetic susceptibility. The score is based on the accumulation of genetic variants (single-nucleotide polymorphisms, or SNPs) that predispose individuals to obesity, with higher numbers of variants results in a higher risk score.
The scores are then used to determine the comparison groups for evaluating the obesity-asthma association.
Alphabet soup
Dr. Longo and colleagues analyzed data on a total 1,511 children enrolled in four observational studies (PACMAN, PAGES, HPR, CLARA) and one cohort study (ALSPAC).
They included children with an asthma diagnosis who used ICS and had available information on both BMI and genetics.
The Mendelian randomization analysis was based on a weighted allele score based on 97 SNPs predictive of BMI based on large-scale genomewide association studies. The exposure for the analysis was age- and sex-adjusted BMI z scores based on World Health Organization growth charts for children.
They found that using the Mendelian randomization approach, for each standard deviation increase in BMI, the OR for any parent-reported asthma exacerbations, including urgent care visits or use of oral corticosteroids, was 2.31 (95% confidence interval, 1.26-4.25).
In contrast, if the traditional observational model had been used, the OR would be a nonsignificant 1.10 (95% CI, 0.99-1.22).
“Treatment guidelines recommend steroids for children with asthma who have a higher-than-normal BMI,” Dr. Longo said in a statement. “Our research group felt that the one-size fits-all approach to treating children with asthma with inhaled steroids as their first-line treatment, particularly those with excess weight, warrants revision. At the very least, research identifying potential alternative treatments should be encouraged and prioritized, especially since 30% of children with asthma are also obese. With the childhood obesity epidemic rising, we expect this percentage to increase meaning this problem of poor control will be seen more frequently in routine clinical practice.”
Christopher E. Brightling, PhD, professor of respiratory medicine at the University of Leicester (England), commented that “this is very good and fascinating research with findings that are important and novel.
“It sheds light on the complex interplay between genes, weight, and response to inhaled corticosteroids, underscoring the need to combine drug treatments with lifestyle and diet modifications. Policy makers, health care providers and families need to do much more to tackle the growing obesity epidemic in young people,” he said.
Dr. Brightling was not involved in the study.
The study was supported by the ERS and the European Union’s H2020 research and innovation program. Dr. Longo was a Horizon 2020 Marie-Sklodowska Cure Respire-3 fellow. Dr. Brightling reported no relevant disclosures.
Obese or overweight children with asthma could be using inhaled corticosteroids (ICS) to no avail, combined results from observational studies suggest.
Using Mendelian randomization, a method for reducing bias in observational studies, investigators from the University of Amsterdam Medical Center performed an analysis of data from four cross-sectional studies and one cohort study on a total of 1,511 children with asthma.
They showed that every 1-unit increase in the body mass index (BMI) z score was associated with a more than twofold higher odds ratio for exacerbation, reported Cristina Longo, PhD, a former postdoctoral fellow at AMC, and assistant professor of medicine at the University of Montreal.
“In this large, multicenter Mendelian randomization study, our findings support current evidence that children with higher BMI status respond inadequately to inhaled corticosteroids, and that this association is likely not explained by measured confounding or reverse causation,” she said in an oral abstract presentation during the European Respiratory Society International Congress.
Unmeasured confounding
The obese-asthma phenotype in children is characterized by reduced lung function, high symptom expression, poor response to ICS, and high health care utilization.
“While most observational studies suggest that weight status is associated with asthma exacerbations, despite using inhaled corticosteroids, it’s unclear whether these associations may be due to unmeasured confounding or reverse causation, which captures the idea that perhaps obesity is a consequence rather than a cause of uncontrolled severe asthma,” she said.
Traditional observational studies of the obesity-asthma link rely on comparing data on asthma in a target population and comparing nonobese patients with obese patients. The problem with this method, Dr. Longo contended, is that the exposure assignment – weight status – is not random, and could lead to bias from potential imbalance of confounders, leading to unintentionally biased results.
In contrast, Mendelian randomization uses genetic data to approximate random assignment of exposures, using a risk score for BMI based on genetic susceptibility. The score is based on the accumulation of genetic variants (single-nucleotide polymorphisms, or SNPs) that predispose individuals to obesity, with higher numbers of variants results in a higher risk score.
The scores are then used to determine the comparison groups for evaluating the obesity-asthma association.
Alphabet soup
Dr. Longo and colleagues analyzed data on a total 1,511 children enrolled in four observational studies (PACMAN, PAGES, HPR, CLARA) and one cohort study (ALSPAC).
They included children with an asthma diagnosis who used ICS and had available information on both BMI and genetics.
The Mendelian randomization analysis was based on a weighted allele score based on 97 SNPs predictive of BMI based on large-scale genomewide association studies. The exposure for the analysis was age- and sex-adjusted BMI z scores based on World Health Organization growth charts for children.
They found that using the Mendelian randomization approach, for each standard deviation increase in BMI, the OR for any parent-reported asthma exacerbations, including urgent care visits or use of oral corticosteroids, was 2.31 (95% confidence interval, 1.26-4.25).
In contrast, if the traditional observational model had been used, the OR would be a nonsignificant 1.10 (95% CI, 0.99-1.22).
“Treatment guidelines recommend steroids for children with asthma who have a higher-than-normal BMI,” Dr. Longo said in a statement. “Our research group felt that the one-size fits-all approach to treating children with asthma with inhaled steroids as their first-line treatment, particularly those with excess weight, warrants revision. At the very least, research identifying potential alternative treatments should be encouraged and prioritized, especially since 30% of children with asthma are also obese. With the childhood obesity epidemic rising, we expect this percentage to increase meaning this problem of poor control will be seen more frequently in routine clinical practice.”
Christopher E. Brightling, PhD, professor of respiratory medicine at the University of Leicester (England), commented that “this is very good and fascinating research with findings that are important and novel.
“It sheds light on the complex interplay between genes, weight, and response to inhaled corticosteroids, underscoring the need to combine drug treatments with lifestyle and diet modifications. Policy makers, health care providers and families need to do much more to tackle the growing obesity epidemic in young people,” he said.
Dr. Brightling was not involved in the study.
The study was supported by the ERS and the European Union’s H2020 research and innovation program. Dr. Longo was a Horizon 2020 Marie-Sklodowska Cure Respire-3 fellow. Dr. Brightling reported no relevant disclosures.
Obese or overweight children with asthma could be using inhaled corticosteroids (ICS) to no avail, combined results from observational studies suggest.
Using Mendelian randomization, a method for reducing bias in observational studies, investigators from the University of Amsterdam Medical Center performed an analysis of data from four cross-sectional studies and one cohort study on a total of 1,511 children with asthma.
They showed that every 1-unit increase in the body mass index (BMI) z score was associated with a more than twofold higher odds ratio for exacerbation, reported Cristina Longo, PhD, a former postdoctoral fellow at AMC, and assistant professor of medicine at the University of Montreal.
“In this large, multicenter Mendelian randomization study, our findings support current evidence that children with higher BMI status respond inadequately to inhaled corticosteroids, and that this association is likely not explained by measured confounding or reverse causation,” she said in an oral abstract presentation during the European Respiratory Society International Congress.
Unmeasured confounding
The obese-asthma phenotype in children is characterized by reduced lung function, high symptom expression, poor response to ICS, and high health care utilization.
“While most observational studies suggest that weight status is associated with asthma exacerbations, despite using inhaled corticosteroids, it’s unclear whether these associations may be due to unmeasured confounding or reverse causation, which captures the idea that perhaps obesity is a consequence rather than a cause of uncontrolled severe asthma,” she said.
Traditional observational studies of the obesity-asthma link rely on comparing data on asthma in a target population and comparing nonobese patients with obese patients. The problem with this method, Dr. Longo contended, is that the exposure assignment – weight status – is not random, and could lead to bias from potential imbalance of confounders, leading to unintentionally biased results.
In contrast, Mendelian randomization uses genetic data to approximate random assignment of exposures, using a risk score for BMI based on genetic susceptibility. The score is based on the accumulation of genetic variants (single-nucleotide polymorphisms, or SNPs) that predispose individuals to obesity, with higher numbers of variants results in a higher risk score.
The scores are then used to determine the comparison groups for evaluating the obesity-asthma association.
Alphabet soup
Dr. Longo and colleagues analyzed data on a total 1,511 children enrolled in four observational studies (PACMAN, PAGES, HPR, CLARA) and one cohort study (ALSPAC).
They included children with an asthma diagnosis who used ICS and had available information on both BMI and genetics.
The Mendelian randomization analysis was based on a weighted allele score based on 97 SNPs predictive of BMI based on large-scale genomewide association studies. The exposure for the analysis was age- and sex-adjusted BMI z scores based on World Health Organization growth charts for children.
They found that using the Mendelian randomization approach, for each standard deviation increase in BMI, the OR for any parent-reported asthma exacerbations, including urgent care visits or use of oral corticosteroids, was 2.31 (95% confidence interval, 1.26-4.25).
In contrast, if the traditional observational model had been used, the OR would be a nonsignificant 1.10 (95% CI, 0.99-1.22).
“Treatment guidelines recommend steroids for children with asthma who have a higher-than-normal BMI,” Dr. Longo said in a statement. “Our research group felt that the one-size fits-all approach to treating children with asthma with inhaled steroids as their first-line treatment, particularly those with excess weight, warrants revision. At the very least, research identifying potential alternative treatments should be encouraged and prioritized, especially since 30% of children with asthma are also obese. With the childhood obesity epidemic rising, we expect this percentage to increase meaning this problem of poor control will be seen more frequently in routine clinical practice.”
Christopher E. Brightling, PhD, professor of respiratory medicine at the University of Leicester (England), commented that “this is very good and fascinating research with findings that are important and novel.
“It sheds light on the complex interplay between genes, weight, and response to inhaled corticosteroids, underscoring the need to combine drug treatments with lifestyle and diet modifications. Policy makers, health care providers and families need to do much more to tackle the growing obesity epidemic in young people,” he said.
Dr. Brightling was not involved in the study.
The study was supported by the ERS and the European Union’s H2020 research and innovation program. Dr. Longo was a Horizon 2020 Marie-Sklodowska Cure Respire-3 fellow. Dr. Brightling reported no relevant disclosures.
FROM ERS 2021
USPSTF update: Screen young asymptomatic women for chlamydia and gonorrhea
But evidence for screening men remains insufficient, task force says
The U.S. Preventive Services Task Force has updated its 2014 statement on screening asymptomatic individuals for chlamydia and gonorrhea infection.
Published online in JAMA, the 2021 version recommends that all sexually active women aged 24 years or younger and at-risk women 25 years or older should be screened for chlamydia and gonorrhea.
As in 2014, the task force made no screening recommendation for men owing to inconclusive evidence of benefit.
With cases of sexually transmitted infections reaching all-time highs, Amy G. Cantor, MD, MPH, of the Pacific Northwest Evidence-based Practice Center at Oregon Health & Science University, Portland, and colleagues noted that chlamydia and gonorrhea are among the most common STIs in this country. According to the Centers for Disease Control and Prevention, 2019 saw approximately 1.8 million reported cases of chlamydia and more than 600,000 of gonorrhea.
In the current analysis of 27 observational and randomized studies comprising 179,515 patients, the USPSTF panel found that, compared with no screening, chlamydia screening was significantly associated with a reduced risk of pelvic inflammatory disease (PID) in young women in 2 out of 4 trials.
The authors cautioned, however, that the magnitude of benefit was relatively small. No studies reported on screening effectiveness in men, except for one reporting rates of epididymitis, and no studies were done on pregnant women for any outcome.
The largest and newest study, the Australian Chlamydia Control Effectiveness Pilot trial of 2018, assessed chlamydia screening against usual care in 180,355 men and women aged 16-29 years in 130 rural Australian primary care clinics. Screening was associated with a reduced risk of hospital-diagnosed PID: the absolute risk was 0.24% for screening versus 0.38% for usual care (unadjusted risk ratio, 0.6; 95% confidence interval, 0.4-1.0). It was not, however, significantly associated with a reduced risk of clinic-diagnosed PID, with an absolute risk of 0.45% versus 0.39% (RR, 1.1; 95% CI, 0.7-18). Nor did it correlate with a risk reduction for clinic-diagnosed epididymitis: 0.26% vs. 0.27% (RR, 0.9; 95% CI, 0.6-1.4).
While risk prediction criteria apart from age were only minimally accurate, testing for asymptomatic chlamydial and gonococcal infections was highly accurate at most anatomical sites, including urine and self-collected specimens, the investigators observed. Age 22 years or younger alone versus multi-item risk criteria demonstrated similar discrimination in a study that included symptomatic and asymptomatic women.
Sensitivity of chlamydial testing was similar at endocervical (89%-100%) and self- and clinician-collected vaginal (90%-100%) sites for women and at meatal (100%), urethral (99%), and rectal (92%) sites for men. It was lower, however, at pharyngeal sites (69.2%) for men who have sex with men (MSM).
Sensitivity of gonococcal testing was 89% or greater for all anatomical samples. False-positive and false-negative testing rates were low across anatomical sites and collection methods.
“Effectiveness of screening in men and during pregnancy, optimal screening intervals, and adverse effects of screening require further evaluation, Dr. Cantor and associates concluded.
In an accompanying editorial, Jeanne Marrazzo, MD, MPH, and Jodie Dionne-Odom, MD, MSPH, of the division of infectious diseases at the University of Alabama at Birmingham, called the guidelines “timely” and “powerful agents of change” that “influence a wide spectrum of health-based metrics, from quality assurance measures to criteria for financial reimbursement.”
They pointed out that men who have sex with men are experiencing historically high rates of gonorrhea, with most infections occurring extragenitally at the pharynx or rectum. In 2019 CDC data, MSM had substantially higher rates of gonorrhea than men who had sex only with women. They recommended that guidelines for men consider STI risk because of sexual relations with men, women, or both.
“Comprehensive screening guidelines for common STIs like chlamydia and gonorrhea could incorporate the limited evidence base for MSM, whether it is regular practice or not,” they wrote, with the same approach for women who have sex with women but may be at risk for chlamydia, particularly if they also have sex with men.
In their view, these latest guidelines appropriately prioritize high-level clinically based data. They pointed, however, to recent progress in understanding the pathogenesis of upper reproductive tract infection in women and the sexual networks behind the current resurgence of STIs in the United States in the failure to manage exposed sex partners.
“Considering these critical advances in the evolution of clinic-based screening guidelines is a work in progress,” they wrote, “the dialogue among basic scientists, clinical trial investigators, and public health professionals to inform the next version of updated USPSTF chlamydia and gonorrhea screening guidelines should start now.”
In the opinion of Jennifer L. Reed, MD, MS, a professor of pediatrics and an emergency medicine physician at Cincinnati Children’s Hospital Medical Center and not involved in the updated statement, the recommendations are very reasonable. “The highest rates of infection occur in females 15-24 years of age, and therefore asymptomatic screening for chlamydia and gonorrhea is imperative at least annually or more often if they are high risk,” she said in an interview.
“I would hope that providers increase their asymptomatic screening as a result of these recommendations and highly consider it in the younger men,” Dr. Reed added. “I see a very high rate of gonorrhea and chlamydia infections.” Her center is studying the implementation of gonorrhea and chlamydia asymptomatic screening for adolescents in the pediatric emergency department, a high-risk patient population that will benefit from STI screening opportunities in nontraditional settings.
This research was funded by the Agency for Healthcare Research and Quality and the Department of Health & Human Services under a contract to support the USPSTF. One statement coauthor reported personal fees from Insmed, Paratek, RedHill, and Spero, as well as grants from Insmed. No other disclosures were reported. Dr. Dionne-Odom reported grants from the National Institutes of Health/National Institute of Child Health and Development. Dr. Reed reported a grant from NIH/NICHD for a pragmatic trial of improving STI detection in the pediatric ED.
But evidence for screening men remains insufficient, task force says
But evidence for screening men remains insufficient, task force says
The U.S. Preventive Services Task Force has updated its 2014 statement on screening asymptomatic individuals for chlamydia and gonorrhea infection.
Published online in JAMA, the 2021 version recommends that all sexually active women aged 24 years or younger and at-risk women 25 years or older should be screened for chlamydia and gonorrhea.
As in 2014, the task force made no screening recommendation for men owing to inconclusive evidence of benefit.
With cases of sexually transmitted infections reaching all-time highs, Amy G. Cantor, MD, MPH, of the Pacific Northwest Evidence-based Practice Center at Oregon Health & Science University, Portland, and colleagues noted that chlamydia and gonorrhea are among the most common STIs in this country. According to the Centers for Disease Control and Prevention, 2019 saw approximately 1.8 million reported cases of chlamydia and more than 600,000 of gonorrhea.
In the current analysis of 27 observational and randomized studies comprising 179,515 patients, the USPSTF panel found that, compared with no screening, chlamydia screening was significantly associated with a reduced risk of pelvic inflammatory disease (PID) in young women in 2 out of 4 trials.
The authors cautioned, however, that the magnitude of benefit was relatively small. No studies reported on screening effectiveness in men, except for one reporting rates of epididymitis, and no studies were done on pregnant women for any outcome.
The largest and newest study, the Australian Chlamydia Control Effectiveness Pilot trial of 2018, assessed chlamydia screening against usual care in 180,355 men and women aged 16-29 years in 130 rural Australian primary care clinics. Screening was associated with a reduced risk of hospital-diagnosed PID: the absolute risk was 0.24% for screening versus 0.38% for usual care (unadjusted risk ratio, 0.6; 95% confidence interval, 0.4-1.0). It was not, however, significantly associated with a reduced risk of clinic-diagnosed PID, with an absolute risk of 0.45% versus 0.39% (RR, 1.1; 95% CI, 0.7-18). Nor did it correlate with a risk reduction for clinic-diagnosed epididymitis: 0.26% vs. 0.27% (RR, 0.9; 95% CI, 0.6-1.4).
While risk prediction criteria apart from age were only minimally accurate, testing for asymptomatic chlamydial and gonococcal infections was highly accurate at most anatomical sites, including urine and self-collected specimens, the investigators observed. Age 22 years or younger alone versus multi-item risk criteria demonstrated similar discrimination in a study that included symptomatic and asymptomatic women.
Sensitivity of chlamydial testing was similar at endocervical (89%-100%) and self- and clinician-collected vaginal (90%-100%) sites for women and at meatal (100%), urethral (99%), and rectal (92%) sites for men. It was lower, however, at pharyngeal sites (69.2%) for men who have sex with men (MSM).
Sensitivity of gonococcal testing was 89% or greater for all anatomical samples. False-positive and false-negative testing rates were low across anatomical sites and collection methods.
“Effectiveness of screening in men and during pregnancy, optimal screening intervals, and adverse effects of screening require further evaluation, Dr. Cantor and associates concluded.
In an accompanying editorial, Jeanne Marrazzo, MD, MPH, and Jodie Dionne-Odom, MD, MSPH, of the division of infectious diseases at the University of Alabama at Birmingham, called the guidelines “timely” and “powerful agents of change” that “influence a wide spectrum of health-based metrics, from quality assurance measures to criteria for financial reimbursement.”
They pointed out that men who have sex with men are experiencing historically high rates of gonorrhea, with most infections occurring extragenitally at the pharynx or rectum. In 2019 CDC data, MSM had substantially higher rates of gonorrhea than men who had sex only with women. They recommended that guidelines for men consider STI risk because of sexual relations with men, women, or both.
“Comprehensive screening guidelines for common STIs like chlamydia and gonorrhea could incorporate the limited evidence base for MSM, whether it is regular practice or not,” they wrote, with the same approach for women who have sex with women but may be at risk for chlamydia, particularly if they also have sex with men.
In their view, these latest guidelines appropriately prioritize high-level clinically based data. They pointed, however, to recent progress in understanding the pathogenesis of upper reproductive tract infection in women and the sexual networks behind the current resurgence of STIs in the United States in the failure to manage exposed sex partners.
“Considering these critical advances in the evolution of clinic-based screening guidelines is a work in progress,” they wrote, “the dialogue among basic scientists, clinical trial investigators, and public health professionals to inform the next version of updated USPSTF chlamydia and gonorrhea screening guidelines should start now.”
In the opinion of Jennifer L. Reed, MD, MS, a professor of pediatrics and an emergency medicine physician at Cincinnati Children’s Hospital Medical Center and not involved in the updated statement, the recommendations are very reasonable. “The highest rates of infection occur in females 15-24 years of age, and therefore asymptomatic screening for chlamydia and gonorrhea is imperative at least annually or more often if they are high risk,” she said in an interview.
“I would hope that providers increase their asymptomatic screening as a result of these recommendations and highly consider it in the younger men,” Dr. Reed added. “I see a very high rate of gonorrhea and chlamydia infections.” Her center is studying the implementation of gonorrhea and chlamydia asymptomatic screening for adolescents in the pediatric emergency department, a high-risk patient population that will benefit from STI screening opportunities in nontraditional settings.
This research was funded by the Agency for Healthcare Research and Quality and the Department of Health & Human Services under a contract to support the USPSTF. One statement coauthor reported personal fees from Insmed, Paratek, RedHill, and Spero, as well as grants from Insmed. No other disclosures were reported. Dr. Dionne-Odom reported grants from the National Institutes of Health/National Institute of Child Health and Development. Dr. Reed reported a grant from NIH/NICHD for a pragmatic trial of improving STI detection in the pediatric ED.
The U.S. Preventive Services Task Force has updated its 2014 statement on screening asymptomatic individuals for chlamydia and gonorrhea infection.
Published online in JAMA, the 2021 version recommends that all sexually active women aged 24 years or younger and at-risk women 25 years or older should be screened for chlamydia and gonorrhea.
As in 2014, the task force made no screening recommendation for men owing to inconclusive evidence of benefit.
With cases of sexually transmitted infections reaching all-time highs, Amy G. Cantor, MD, MPH, of the Pacific Northwest Evidence-based Practice Center at Oregon Health & Science University, Portland, and colleagues noted that chlamydia and gonorrhea are among the most common STIs in this country. According to the Centers for Disease Control and Prevention, 2019 saw approximately 1.8 million reported cases of chlamydia and more than 600,000 of gonorrhea.
In the current analysis of 27 observational and randomized studies comprising 179,515 patients, the USPSTF panel found that, compared with no screening, chlamydia screening was significantly associated with a reduced risk of pelvic inflammatory disease (PID) in young women in 2 out of 4 trials.
The authors cautioned, however, that the magnitude of benefit was relatively small. No studies reported on screening effectiveness in men, except for one reporting rates of epididymitis, and no studies were done on pregnant women for any outcome.
The largest and newest study, the Australian Chlamydia Control Effectiveness Pilot trial of 2018, assessed chlamydia screening against usual care in 180,355 men and women aged 16-29 years in 130 rural Australian primary care clinics. Screening was associated with a reduced risk of hospital-diagnosed PID: the absolute risk was 0.24% for screening versus 0.38% for usual care (unadjusted risk ratio, 0.6; 95% confidence interval, 0.4-1.0). It was not, however, significantly associated with a reduced risk of clinic-diagnosed PID, with an absolute risk of 0.45% versus 0.39% (RR, 1.1; 95% CI, 0.7-18). Nor did it correlate with a risk reduction for clinic-diagnosed epididymitis: 0.26% vs. 0.27% (RR, 0.9; 95% CI, 0.6-1.4).
While risk prediction criteria apart from age were only minimally accurate, testing for asymptomatic chlamydial and gonococcal infections was highly accurate at most anatomical sites, including urine and self-collected specimens, the investigators observed. Age 22 years or younger alone versus multi-item risk criteria demonstrated similar discrimination in a study that included symptomatic and asymptomatic women.
Sensitivity of chlamydial testing was similar at endocervical (89%-100%) and self- and clinician-collected vaginal (90%-100%) sites for women and at meatal (100%), urethral (99%), and rectal (92%) sites for men. It was lower, however, at pharyngeal sites (69.2%) for men who have sex with men (MSM).
Sensitivity of gonococcal testing was 89% or greater for all anatomical samples. False-positive and false-negative testing rates were low across anatomical sites and collection methods.
“Effectiveness of screening in men and during pregnancy, optimal screening intervals, and adverse effects of screening require further evaluation, Dr. Cantor and associates concluded.
In an accompanying editorial, Jeanne Marrazzo, MD, MPH, and Jodie Dionne-Odom, MD, MSPH, of the division of infectious diseases at the University of Alabama at Birmingham, called the guidelines “timely” and “powerful agents of change” that “influence a wide spectrum of health-based metrics, from quality assurance measures to criteria for financial reimbursement.”
They pointed out that men who have sex with men are experiencing historically high rates of gonorrhea, with most infections occurring extragenitally at the pharynx or rectum. In 2019 CDC data, MSM had substantially higher rates of gonorrhea than men who had sex only with women. They recommended that guidelines for men consider STI risk because of sexual relations with men, women, or both.
“Comprehensive screening guidelines for common STIs like chlamydia and gonorrhea could incorporate the limited evidence base for MSM, whether it is regular practice or not,” they wrote, with the same approach for women who have sex with women but may be at risk for chlamydia, particularly if they also have sex with men.
In their view, these latest guidelines appropriately prioritize high-level clinically based data. They pointed, however, to recent progress in understanding the pathogenesis of upper reproductive tract infection in women and the sexual networks behind the current resurgence of STIs in the United States in the failure to manage exposed sex partners.
“Considering these critical advances in the evolution of clinic-based screening guidelines is a work in progress,” they wrote, “the dialogue among basic scientists, clinical trial investigators, and public health professionals to inform the next version of updated USPSTF chlamydia and gonorrhea screening guidelines should start now.”
In the opinion of Jennifer L. Reed, MD, MS, a professor of pediatrics and an emergency medicine physician at Cincinnati Children’s Hospital Medical Center and not involved in the updated statement, the recommendations are very reasonable. “The highest rates of infection occur in females 15-24 years of age, and therefore asymptomatic screening for chlamydia and gonorrhea is imperative at least annually or more often if they are high risk,” she said in an interview.
“I would hope that providers increase their asymptomatic screening as a result of these recommendations and highly consider it in the younger men,” Dr. Reed added. “I see a very high rate of gonorrhea and chlamydia infections.” Her center is studying the implementation of gonorrhea and chlamydia asymptomatic screening for adolescents in the pediatric emergency department, a high-risk patient population that will benefit from STI screening opportunities in nontraditional settings.
This research was funded by the Agency for Healthcare Research and Quality and the Department of Health & Human Services under a contract to support the USPSTF. One statement coauthor reported personal fees from Insmed, Paratek, RedHill, and Spero, as well as grants from Insmed. No other disclosures were reported. Dr. Dionne-Odom reported grants from the National Institutes of Health/National Institute of Child Health and Development. Dr. Reed reported a grant from NIH/NICHD for a pragmatic trial of improving STI detection in the pediatric ED.
FROM JAMA