User login
Eating disorder may be common in celiac disease
A new study examining avoidant/restrictive food intake disorder (ARFID) among patients with celiac disease found that the condition is common but is not associated with any difference in disease control. The findings suggest that some with celiac disease may pursue dietary control too far, but experts warn that ARFID is only recently being recognized in patients with GI diseases, the definition is in flux, and it’s important to not overpathologize patient behavior.
The new study, published in Gastro Hep Advances, comes in the wake of a 2021 cross-sectional study, which found that 53.7% of celiac disease patients met the criteria for ARFID based on the Nine-Item ARFID Screen, and were more likely to have anxiety, depression, and reduced food-related quality of life.
“I think both studies are hypothesizing that there might be greater fear around eating in these patients with celiac, but that the possible outcomes related to their disease may not actually be different,” said Helen Burton Murray, PhD, director of the GI behavioral health program and staff psychologist at Massachusetts General Hospital, Boston, who was asked to comment on the study.
She also noted that ARFID may represent a subgroup of celiac patients with more severe disease or worse quality of life, though the two studies can’t definitively prove that. The surveys used are intended for screening rather than diagnosis and have not yet been validated in patients with a gastrointestinal disease like celiac.
Although the symptoms of ARFID have been recognized for many years, it only became an official diagnosis with its inclusion in DSM-5 in 2013. Physicians are becoming increasingly aware of this potential comorbidity, but it can be difficult to diagnose or understand the impact of an eating disorder in a condition like celiac disease, where intense dietary management is the key to controlling it. “There’s concern about overpathologizing patients where dietary management can be a normative strategy, and overpathologizing by diagnosing ARFID. Is diagnosing ARFID going to change the patient’s treatment course and improve outcomes for them?” asked Dr. Burton Murray.
In some cases, the answer may be yes. Patients may be so restrictive in their eating that it impacts physical health or lifestyle. “Hypervigilance or worry around eating could extend to even non–gluten based foods. That may be a marker of where a patient’s eating behaviors are crossing the line into ARFID, if their diet is so limited when it doesn’t need to be, and those limitations might be harming them nutritionally, leading to weight loss or making it difficult to live their life in the way that they would like to,” said Dr. Burton Murray.
Still, the results of these studies shouldn’t be overinterpreted, according to Anne R. Lee, EdD, RDN, LD, associate professor of nutritional medicine at the celiac disease center at Columbia University, New York. “In the world of eating disorders, ARFID is the newest kid on the block, and one that’s in transition,” she said. What differentiates ARFID from other eating disorders is that food behavior is related to things like appetite or picky eating, but not body shape and size. Therefore, it helps to combine the ARFID screen with other eating disorder screening tools, Dr. Lee said.
“We need to differentiate between diagnosing someone with a disordered eating pattern versus helping them navigate their life within a gluten-free diet. We need to help them with developing strategies to maneuver through work lunches and social outings and all of those things so that we don’t overdiagnose,” said Dr. Lee.
In the new study, researchers retrospectively analyzed data from 137 patients with celiac disease at the Center for Human Nutrition at Vanderbilt University Medical Center; 107 were women, and the median age was 37 years. The researchers used questionnaires to evaluate diet, including the ARFID Symptom Checklist.
Seventy-eight participants (57%) had suspected ARFID; 30 had symptoms consistent with clinical ARFID and 48 consistent with subclinical ARFID. There were no differences between patients with and without ARFID with respect to anxiety and depression, length of illness, age, gender, body mass index, bone disease, or micronutrient or vitamin deficiency. Serology studies revealed only one difference: a higher frequency of tissue transglutaminase IgG antibody in the ARFID group (15% vs. 2%; P = .007).
There was a strong correlation between ARFID and the Impact of the Gluten Free Diet questionnaire (IGFDQ), with patients scoring higher on the social and food components more likely to also have ARFID. It was also the only predictor of ARFID in a multivariable analysis, with associations in the food (odds ratio, 1.64; P = .01), emotional (OR, 1.66; P = .05), and social (OR, 1.59; P = .01) sections.
The authors concluded that, although there were some study limitations, including possible patient misunderstanding of the survey questions and lack of knowledge of whether the patients had access to gluten-free foods, AFID is not only common, but it also has a significant impact on patients with celiac disease. The authors also noted that this assessment occurred over a 2-year period, with patients attending clinic only once a year. Follow-up surveys, duodenal biopsies, and bone density assessments could identify more differences over time.
Dr. Burton Murray and Dr. Lee have no relevant financial disclosures.
A new study examining avoidant/restrictive food intake disorder (ARFID) among patients with celiac disease found that the condition is common but is not associated with any difference in disease control. The findings suggest that some with celiac disease may pursue dietary control too far, but experts warn that ARFID is only recently being recognized in patients with GI diseases, the definition is in flux, and it’s important to not overpathologize patient behavior.
The new study, published in Gastro Hep Advances, comes in the wake of a 2021 cross-sectional study, which found that 53.7% of celiac disease patients met the criteria for ARFID based on the Nine-Item ARFID Screen, and were more likely to have anxiety, depression, and reduced food-related quality of life.
“I think both studies are hypothesizing that there might be greater fear around eating in these patients with celiac, but that the possible outcomes related to their disease may not actually be different,” said Helen Burton Murray, PhD, director of the GI behavioral health program and staff psychologist at Massachusetts General Hospital, Boston, who was asked to comment on the study.
She also noted that ARFID may represent a subgroup of celiac patients with more severe disease or worse quality of life, though the two studies can’t definitively prove that. The surveys used are intended for screening rather than diagnosis and have not yet been validated in patients with a gastrointestinal disease like celiac.
Although the symptoms of ARFID have been recognized for many years, it only became an official diagnosis with its inclusion in DSM-5 in 2013. Physicians are becoming increasingly aware of this potential comorbidity, but it can be difficult to diagnose or understand the impact of an eating disorder in a condition like celiac disease, where intense dietary management is the key to controlling it. “There’s concern about overpathologizing patients where dietary management can be a normative strategy, and overpathologizing by diagnosing ARFID. Is diagnosing ARFID going to change the patient’s treatment course and improve outcomes for them?” asked Dr. Burton Murray.
In some cases, the answer may be yes. Patients may be so restrictive in their eating that it impacts physical health or lifestyle. “Hypervigilance or worry around eating could extend to even non–gluten based foods. That may be a marker of where a patient’s eating behaviors are crossing the line into ARFID, if their diet is so limited when it doesn’t need to be, and those limitations might be harming them nutritionally, leading to weight loss or making it difficult to live their life in the way that they would like to,” said Dr. Burton Murray.
Still, the results of these studies shouldn’t be overinterpreted, according to Anne R. Lee, EdD, RDN, LD, associate professor of nutritional medicine at the celiac disease center at Columbia University, New York. “In the world of eating disorders, ARFID is the newest kid on the block, and one that’s in transition,” she said. What differentiates ARFID from other eating disorders is that food behavior is related to things like appetite or picky eating, but not body shape and size. Therefore, it helps to combine the ARFID screen with other eating disorder screening tools, Dr. Lee said.
“We need to differentiate between diagnosing someone with a disordered eating pattern versus helping them navigate their life within a gluten-free diet. We need to help them with developing strategies to maneuver through work lunches and social outings and all of those things so that we don’t overdiagnose,” said Dr. Lee.
In the new study, researchers retrospectively analyzed data from 137 patients with celiac disease at the Center for Human Nutrition at Vanderbilt University Medical Center; 107 were women, and the median age was 37 years. The researchers used questionnaires to evaluate diet, including the ARFID Symptom Checklist.
Seventy-eight participants (57%) had suspected ARFID; 30 had symptoms consistent with clinical ARFID and 48 consistent with subclinical ARFID. There were no differences between patients with and without ARFID with respect to anxiety and depression, length of illness, age, gender, body mass index, bone disease, or micronutrient or vitamin deficiency. Serology studies revealed only one difference: a higher frequency of tissue transglutaminase IgG antibody in the ARFID group (15% vs. 2%; P = .007).
There was a strong correlation between ARFID and the Impact of the Gluten Free Diet questionnaire (IGFDQ), with patients scoring higher on the social and food components more likely to also have ARFID. It was also the only predictor of ARFID in a multivariable analysis, with associations in the food (odds ratio, 1.64; P = .01), emotional (OR, 1.66; P = .05), and social (OR, 1.59; P = .01) sections.
The authors concluded that, although there were some study limitations, including possible patient misunderstanding of the survey questions and lack of knowledge of whether the patients had access to gluten-free foods, AFID is not only common, but it also has a significant impact on patients with celiac disease. The authors also noted that this assessment occurred over a 2-year period, with patients attending clinic only once a year. Follow-up surveys, duodenal biopsies, and bone density assessments could identify more differences over time.
Dr. Burton Murray and Dr. Lee have no relevant financial disclosures.
A new study examining avoidant/restrictive food intake disorder (ARFID) among patients with celiac disease found that the condition is common but is not associated with any difference in disease control. The findings suggest that some with celiac disease may pursue dietary control too far, but experts warn that ARFID is only recently being recognized in patients with GI diseases, the definition is in flux, and it’s important to not overpathologize patient behavior.
The new study, published in Gastro Hep Advances, comes in the wake of a 2021 cross-sectional study, which found that 53.7% of celiac disease patients met the criteria for ARFID based on the Nine-Item ARFID Screen, and were more likely to have anxiety, depression, and reduced food-related quality of life.
“I think both studies are hypothesizing that there might be greater fear around eating in these patients with celiac, but that the possible outcomes related to their disease may not actually be different,” said Helen Burton Murray, PhD, director of the GI behavioral health program and staff psychologist at Massachusetts General Hospital, Boston, who was asked to comment on the study.
She also noted that ARFID may represent a subgroup of celiac patients with more severe disease or worse quality of life, though the two studies can’t definitively prove that. The surveys used are intended for screening rather than diagnosis and have not yet been validated in patients with a gastrointestinal disease like celiac.
Although the symptoms of ARFID have been recognized for many years, it only became an official diagnosis with its inclusion in DSM-5 in 2013. Physicians are becoming increasingly aware of this potential comorbidity, but it can be difficult to diagnose or understand the impact of an eating disorder in a condition like celiac disease, where intense dietary management is the key to controlling it. “There’s concern about overpathologizing patients where dietary management can be a normative strategy, and overpathologizing by diagnosing ARFID. Is diagnosing ARFID going to change the patient’s treatment course and improve outcomes for them?” asked Dr. Burton Murray.
In some cases, the answer may be yes. Patients may be so restrictive in their eating that it impacts physical health or lifestyle. “Hypervigilance or worry around eating could extend to even non–gluten based foods. That may be a marker of where a patient’s eating behaviors are crossing the line into ARFID, if their diet is so limited when it doesn’t need to be, and those limitations might be harming them nutritionally, leading to weight loss or making it difficult to live their life in the way that they would like to,” said Dr. Burton Murray.
Still, the results of these studies shouldn’t be overinterpreted, according to Anne R. Lee, EdD, RDN, LD, associate professor of nutritional medicine at the celiac disease center at Columbia University, New York. “In the world of eating disorders, ARFID is the newest kid on the block, and one that’s in transition,” she said. What differentiates ARFID from other eating disorders is that food behavior is related to things like appetite or picky eating, but not body shape and size. Therefore, it helps to combine the ARFID screen with other eating disorder screening tools, Dr. Lee said.
“We need to differentiate between diagnosing someone with a disordered eating pattern versus helping them navigate their life within a gluten-free diet. We need to help them with developing strategies to maneuver through work lunches and social outings and all of those things so that we don’t overdiagnose,” said Dr. Lee.
In the new study, researchers retrospectively analyzed data from 137 patients with celiac disease at the Center for Human Nutrition at Vanderbilt University Medical Center; 107 were women, and the median age was 37 years. The researchers used questionnaires to evaluate diet, including the ARFID Symptom Checklist.
Seventy-eight participants (57%) had suspected ARFID; 30 had symptoms consistent with clinical ARFID and 48 consistent with subclinical ARFID. There were no differences between patients with and without ARFID with respect to anxiety and depression, length of illness, age, gender, body mass index, bone disease, or micronutrient or vitamin deficiency. Serology studies revealed only one difference: a higher frequency of tissue transglutaminase IgG antibody in the ARFID group (15% vs. 2%; P = .007).
There was a strong correlation between ARFID and the Impact of the Gluten Free Diet questionnaire (IGFDQ), with patients scoring higher on the social and food components more likely to also have ARFID. It was also the only predictor of ARFID in a multivariable analysis, with associations in the food (odds ratio, 1.64; P = .01), emotional (OR, 1.66; P = .05), and social (OR, 1.59; P = .01) sections.
The authors concluded that, although there were some study limitations, including possible patient misunderstanding of the survey questions and lack of knowledge of whether the patients had access to gluten-free foods, AFID is not only common, but it also has a significant impact on patients with celiac disease. The authors also noted that this assessment occurred over a 2-year period, with patients attending clinic only once a year. Follow-up surveys, duodenal biopsies, and bone density assessments could identify more differences over time.
Dr. Burton Murray and Dr. Lee have no relevant financial disclosures.
FROM GASTRO HEP ADVANCES
Pandemic-stressed youths call runaway hotline
The calls kept coming into the National Runaway Safeline during the pandemic: the desperate kids who wanted to bike away from home in the middle of the night, the isolated youths who felt suicidal, the teens whose parents had forced them out of the house.
To the surprise of experts who help runaway youths, the pandemic didn’t appear to produce a big rise or fall in the numbers of children and teens who had left home. Still, the crisis hit hard. As schools closed and households sheltered in place, youths reached out to the National Runaway Safeline to report heightened family conflicts and worsening mental health.
The Safeline, based in Chicago, is the country’s 24/7, federally designated communications system for runaway and homeless youths. Each year, it makes about 125,000 connections with young people and their family members through its hotline and other services.
In a typical year, teens aged 15-17 years are the main group that gets in touch by phone, live chat, email, or an online crisis forum, according to Jeff Stern, chief engagement officer at the Safeline.
But in the past 2 years, “contacts have skewed younger,” including many more children under age 12.
“I think this is showing what a hit this is taking on young children,” he said.
Without school, sports, and other activities, younger children might be reaching out because they’ve lost trusted sources of support. Callers have been as young as 9.
“Those ones stand out,” said a crisis center supervisor who asked to go by Michael, which is not his real name, to protect the privacy of his clients.
In November 2020, a child posted in the crisis forum: “I’m 11 and my parents treat me poorly. They have told me many times to ‘kill myself’ and I didn’t let that settle well with me. ... I have tried to run away one time from my house, but they found out, so they took my phone away and put screws on my windows so I couldn’t leave.”
Increasing numbers of children told Safeline counselors that their parents were emotionally or verbally abusive, while others reported physical abuse. Some said they experienced neglect, while others had been thrown out.
“We absolutely have had youths who have either been physically kicked out of the house or just verbally told to leave,” Michael said, “and then the kid does.”
Heightened family conflicts
The Safeline partners with the National Center for Missing and Exploited Children, which, despite widespread public perception, doesn’t work mainly with child abduction cases. Each year, the center assists with 29,000-31,000 cases, and 92% involve “endangered runaways,” said John Bischoff, vice president of the Missing Children Division. These children could be running away from home or foster care.
During the pandemic, the center didn’t spot major changes in its missing child numbers, “which honestly was shocking,” Mr. Bischoff said. “We figured we were either going to see an extreme rise or a decrease.
“But the reasons for the run were changing,” he said.
Many youths were fleeing out of frustration with quarantine restrictions, Mr. Bischoff said, as well as frustration with the unknown and their own lack of control over many situations.
At the runaway hotline, calls have been longer and more intense, with family problems topping the list of concerns. In 2019, about 57% of all contacts mentioned family dynamics. In 2020, that number jumped to 88%, according to Mr. Stern.
Some kids sought support for family problems that involved school. In October 2020, one 13-year-old wrote in the Safeline forum: “My mom constantly yells at me for no reason. I want to leave, but I don’t know how. I have also been really stressed about school because they haven’t been giving me the grades I would normally receive during actual school. She thinks I’m lying and that I don’t care. I just need somebody to help me.”
Many adults are under tremendous strain, too, Michael said.
“Parents might have gotten COVID last month and haven’t been able to work for 2 weeks, and they’re missing a paycheck now. Money is tight, there might not be food, everyone’s angry at everything.”
During the pandemic, the National Runaway Safeline found a 16% increase in contacts citing financial challenges.
Some children have felt confined in unsafe homes or have endured violence, as one 15-year-old reported in the forum: “I am the scapegoat out of four kids. Unfortunately, my mom has always been a toxic person. ... I’m the only kid she still hits really hard. She’s left bruises and scratches recently. ... I just have no solution to this.”
Worsening mental health
Besides family dynamics, mental health emerged as a top concern that youths reported in 2020. “This is something notable. It increased by 30% just in 1 year,” Mr. Stern said.
In November 2020, a 16-year-old wrote: “I can’t ever go outside. I’ve been stuck in the house for a very long time now since quarantine started. I’m scared. ... My mother has been taking her anger out on me emotionally. ... I have severe depression and I need help. Please, if there’s any way I can get out of here, let me know.”
The Safeline also has seen a rise in suicide-related contacts. Among children and teens who had cited a mental health concern, 18% said they were suicidal, Stern said. Most were between ages 12 and 16, but some were younger than 12.
When children couldn’t hang out with peers, they felt even more isolated if parents confiscated their phones, a common punishment, Michael said.
During the winter of 2020-21, “It felt like almost every digital contact was a youth reaching out on their Chromebook because they had gotten their phone taken away and they were either suicidal or considering running away,” he said. “That’s kind of their entire social sphere getting taken away.”
Reality check
Roughly 7 in 10 youths report still being at home when they reach out to the Safeline. Among those who do leave, Michael said, “They’re going sometimes to friends’ houses, oftentimes to a significant other’s house, sometimes to extended family members’ houses. Often, they don’t have a place that they’re planning to go. They just left, and that’s why they’re calling us.”
While some youths have been afraid of catching COVID-19 in general, the coronavirus threat hasn’t deterred those who have decided to run away, Michael said. “Usually, they’re more worried about being returned home.”
Many can’t comprehend the risks of setting off on their own.
In October 2021, a 15-year-old boy posted on the forum that his verbally abusive parents had called him a mistake and said they couldn’t wait for him to move out.
“So I’m going to make their dreams come true,” he wrote. “I’m going to go live in California with my friend who is a young YouTuber. I need help getting money to either fly or get a bus ticket, even though I’m all right with trying to ride a bike or fixing my dirt bike and getting the wagon to pull my stuff. But I’m looking for apartments in Los Angeles so I’m not living on the streets and I’m looking for a job. Please help me. My friend can’t send me money because I don’t have a bank account.”
“Often,” Michael said, “we’re reality-checking kids who want to hitchhike 5 hours away to either a friend’s or the closest shelter that we could find them. Or walk for 5 hours at 3 a.m. or bike, so we try to safety-check that.”
Another concern: online enticement by predators. During the pandemic, the National Center for Missing and Exploited Children saw cases in which children ran away from home “to go meet with someone who may not be who they thought they were talking to online,” Mr. Bischoff said. “It’s certainly something we’re keeping a close eye on.”
Fewer resources in the pandemic
The National Runaway Safeline provides information and referrals to other hotlines and services, including suicide prevention and mental health organizations. When youths have already run away and have no place to go, Michael said, the Safeline tries to find shelter options or seek out a relative who can provide a safe place to stay.
But finding shelters became tougher during the pandemic, when many had no room or shelter supply was limited. Some had to shut down for COVID-19–related deep cleanings, Michael said. Helping youths find transportation, especially with public transportation shutdowns, also was tough.
The Huckleberry House, a six-bed youth shelter in San Francisco, has stayed open throughout the pandemic with limited staffing, said Douglas Styles, PsyD. He’s the executive director of the Huckleberry Youth Programs, which runs the house.
The shelter, which serves Bay Area runaway and homeless youths ages 12-17, hasn’t seen an overall spike in demand, Dr. Styles said. But “what’s expanded is undocumented [youths] and young people who don’t have any family connections in the area, so they’re unaccompanied as well. We’ve seen that here and there throughout the years, but during the pandemic, that population has actually increased quite a bit.”
The Huckleberry House has sheltered children and teens who have run away from all kinds of homes, including affluent ones, Dr. Styles said.
Once children leave home, the lack of adult supervision leaves them vulnerable. They face multiple dangers, including child sex trafficking and exploitation, substance abuse, gang involvement, and violence. “As an organization, that scares us,” Mr. Bischoff said. “What’s happening at home, we’ll sort that out. The biggest thing we as an organization are trying to do is locate them and ensure their safety.”
To help runaways and their families get in touch, the National Runaway Safeline provides a message service and conference calling. “We can play the middleman, really acting on behalf of the young person – not because they’re right or wrong, but to ensure that their voice is really heard,” Mr. Stern said.
Through its national Home Free program, the Safeline partners with Greyhound to bring children back home or into an alternative, safe living environment by providing a free bus ticket.
These days, technology can expose children to harm online, but it can also speed their return home.
“When I was growing up, if you weren’t home by 5 o’clock, Mom would start to worry, but she really didn’t have any way of reaching you,” Mr. Bischoff said. “More children today have cellphones. More children are easily reachable. That’s a benefit.”
A version of this article first appeared on WebMD.com.
The calls kept coming into the National Runaway Safeline during the pandemic: the desperate kids who wanted to bike away from home in the middle of the night, the isolated youths who felt suicidal, the teens whose parents had forced them out of the house.
To the surprise of experts who help runaway youths, the pandemic didn’t appear to produce a big rise or fall in the numbers of children and teens who had left home. Still, the crisis hit hard. As schools closed and households sheltered in place, youths reached out to the National Runaway Safeline to report heightened family conflicts and worsening mental health.
The Safeline, based in Chicago, is the country’s 24/7, federally designated communications system for runaway and homeless youths. Each year, it makes about 125,000 connections with young people and their family members through its hotline and other services.
In a typical year, teens aged 15-17 years are the main group that gets in touch by phone, live chat, email, or an online crisis forum, according to Jeff Stern, chief engagement officer at the Safeline.
But in the past 2 years, “contacts have skewed younger,” including many more children under age 12.
“I think this is showing what a hit this is taking on young children,” he said.
Without school, sports, and other activities, younger children might be reaching out because they’ve lost trusted sources of support. Callers have been as young as 9.
“Those ones stand out,” said a crisis center supervisor who asked to go by Michael, which is not his real name, to protect the privacy of his clients.
In November 2020, a child posted in the crisis forum: “I’m 11 and my parents treat me poorly. They have told me many times to ‘kill myself’ and I didn’t let that settle well with me. ... I have tried to run away one time from my house, but they found out, so they took my phone away and put screws on my windows so I couldn’t leave.”
Increasing numbers of children told Safeline counselors that their parents were emotionally or verbally abusive, while others reported physical abuse. Some said they experienced neglect, while others had been thrown out.
“We absolutely have had youths who have either been physically kicked out of the house or just verbally told to leave,” Michael said, “and then the kid does.”
Heightened family conflicts
The Safeline partners with the National Center for Missing and Exploited Children, which, despite widespread public perception, doesn’t work mainly with child abduction cases. Each year, the center assists with 29,000-31,000 cases, and 92% involve “endangered runaways,” said John Bischoff, vice president of the Missing Children Division. These children could be running away from home or foster care.
During the pandemic, the center didn’t spot major changes in its missing child numbers, “which honestly was shocking,” Mr. Bischoff said. “We figured we were either going to see an extreme rise or a decrease.
“But the reasons for the run were changing,” he said.
Many youths were fleeing out of frustration with quarantine restrictions, Mr. Bischoff said, as well as frustration with the unknown and their own lack of control over many situations.
At the runaway hotline, calls have been longer and more intense, with family problems topping the list of concerns. In 2019, about 57% of all contacts mentioned family dynamics. In 2020, that number jumped to 88%, according to Mr. Stern.
Some kids sought support for family problems that involved school. In October 2020, one 13-year-old wrote in the Safeline forum: “My mom constantly yells at me for no reason. I want to leave, but I don’t know how. I have also been really stressed about school because they haven’t been giving me the grades I would normally receive during actual school. She thinks I’m lying and that I don’t care. I just need somebody to help me.”
Many adults are under tremendous strain, too, Michael said.
“Parents might have gotten COVID last month and haven’t been able to work for 2 weeks, and they’re missing a paycheck now. Money is tight, there might not be food, everyone’s angry at everything.”
During the pandemic, the National Runaway Safeline found a 16% increase in contacts citing financial challenges.
Some children have felt confined in unsafe homes or have endured violence, as one 15-year-old reported in the forum: “I am the scapegoat out of four kids. Unfortunately, my mom has always been a toxic person. ... I’m the only kid she still hits really hard. She’s left bruises and scratches recently. ... I just have no solution to this.”
Worsening mental health
Besides family dynamics, mental health emerged as a top concern that youths reported in 2020. “This is something notable. It increased by 30% just in 1 year,” Mr. Stern said.
In November 2020, a 16-year-old wrote: “I can’t ever go outside. I’ve been stuck in the house for a very long time now since quarantine started. I’m scared. ... My mother has been taking her anger out on me emotionally. ... I have severe depression and I need help. Please, if there’s any way I can get out of here, let me know.”
The Safeline also has seen a rise in suicide-related contacts. Among children and teens who had cited a mental health concern, 18% said they were suicidal, Stern said. Most were between ages 12 and 16, but some were younger than 12.
When children couldn’t hang out with peers, they felt even more isolated if parents confiscated their phones, a common punishment, Michael said.
During the winter of 2020-21, “It felt like almost every digital contact was a youth reaching out on their Chromebook because they had gotten their phone taken away and they were either suicidal or considering running away,” he said. “That’s kind of their entire social sphere getting taken away.”
Reality check
Roughly 7 in 10 youths report still being at home when they reach out to the Safeline. Among those who do leave, Michael said, “They’re going sometimes to friends’ houses, oftentimes to a significant other’s house, sometimes to extended family members’ houses. Often, they don’t have a place that they’re planning to go. They just left, and that’s why they’re calling us.”
While some youths have been afraid of catching COVID-19 in general, the coronavirus threat hasn’t deterred those who have decided to run away, Michael said. “Usually, they’re more worried about being returned home.”
Many can’t comprehend the risks of setting off on their own.
In October 2021, a 15-year-old boy posted on the forum that his verbally abusive parents had called him a mistake and said they couldn’t wait for him to move out.
“So I’m going to make their dreams come true,” he wrote. “I’m going to go live in California with my friend who is a young YouTuber. I need help getting money to either fly or get a bus ticket, even though I’m all right with trying to ride a bike or fixing my dirt bike and getting the wagon to pull my stuff. But I’m looking for apartments in Los Angeles so I’m not living on the streets and I’m looking for a job. Please help me. My friend can’t send me money because I don’t have a bank account.”
“Often,” Michael said, “we’re reality-checking kids who want to hitchhike 5 hours away to either a friend’s or the closest shelter that we could find them. Or walk for 5 hours at 3 a.m. or bike, so we try to safety-check that.”
Another concern: online enticement by predators. During the pandemic, the National Center for Missing and Exploited Children saw cases in which children ran away from home “to go meet with someone who may not be who they thought they were talking to online,” Mr. Bischoff said. “It’s certainly something we’re keeping a close eye on.”
Fewer resources in the pandemic
The National Runaway Safeline provides information and referrals to other hotlines and services, including suicide prevention and mental health organizations. When youths have already run away and have no place to go, Michael said, the Safeline tries to find shelter options or seek out a relative who can provide a safe place to stay.
But finding shelters became tougher during the pandemic, when many had no room or shelter supply was limited. Some had to shut down for COVID-19–related deep cleanings, Michael said. Helping youths find transportation, especially with public transportation shutdowns, also was tough.
The Huckleberry House, a six-bed youth shelter in San Francisco, has stayed open throughout the pandemic with limited staffing, said Douglas Styles, PsyD. He’s the executive director of the Huckleberry Youth Programs, which runs the house.
The shelter, which serves Bay Area runaway and homeless youths ages 12-17, hasn’t seen an overall spike in demand, Dr. Styles said. But “what’s expanded is undocumented [youths] and young people who don’t have any family connections in the area, so they’re unaccompanied as well. We’ve seen that here and there throughout the years, but during the pandemic, that population has actually increased quite a bit.”
The Huckleberry House has sheltered children and teens who have run away from all kinds of homes, including affluent ones, Dr. Styles said.
Once children leave home, the lack of adult supervision leaves them vulnerable. They face multiple dangers, including child sex trafficking and exploitation, substance abuse, gang involvement, and violence. “As an organization, that scares us,” Mr. Bischoff said. “What’s happening at home, we’ll sort that out. The biggest thing we as an organization are trying to do is locate them and ensure their safety.”
To help runaways and their families get in touch, the National Runaway Safeline provides a message service and conference calling. “We can play the middleman, really acting on behalf of the young person – not because they’re right or wrong, but to ensure that their voice is really heard,” Mr. Stern said.
Through its national Home Free program, the Safeline partners with Greyhound to bring children back home or into an alternative, safe living environment by providing a free bus ticket.
These days, technology can expose children to harm online, but it can also speed their return home.
“When I was growing up, if you weren’t home by 5 o’clock, Mom would start to worry, but she really didn’t have any way of reaching you,” Mr. Bischoff said. “More children today have cellphones. More children are easily reachable. That’s a benefit.”
A version of this article first appeared on WebMD.com.
The calls kept coming into the National Runaway Safeline during the pandemic: the desperate kids who wanted to bike away from home in the middle of the night, the isolated youths who felt suicidal, the teens whose parents had forced them out of the house.
To the surprise of experts who help runaway youths, the pandemic didn’t appear to produce a big rise or fall in the numbers of children and teens who had left home. Still, the crisis hit hard. As schools closed and households sheltered in place, youths reached out to the National Runaway Safeline to report heightened family conflicts and worsening mental health.
The Safeline, based in Chicago, is the country’s 24/7, federally designated communications system for runaway and homeless youths. Each year, it makes about 125,000 connections with young people and their family members through its hotline and other services.
In a typical year, teens aged 15-17 years are the main group that gets in touch by phone, live chat, email, or an online crisis forum, according to Jeff Stern, chief engagement officer at the Safeline.
But in the past 2 years, “contacts have skewed younger,” including many more children under age 12.
“I think this is showing what a hit this is taking on young children,” he said.
Without school, sports, and other activities, younger children might be reaching out because they’ve lost trusted sources of support. Callers have been as young as 9.
“Those ones stand out,” said a crisis center supervisor who asked to go by Michael, which is not his real name, to protect the privacy of his clients.
In November 2020, a child posted in the crisis forum: “I’m 11 and my parents treat me poorly. They have told me many times to ‘kill myself’ and I didn’t let that settle well with me. ... I have tried to run away one time from my house, but they found out, so they took my phone away and put screws on my windows so I couldn’t leave.”
Increasing numbers of children told Safeline counselors that their parents were emotionally or verbally abusive, while others reported physical abuse. Some said they experienced neglect, while others had been thrown out.
“We absolutely have had youths who have either been physically kicked out of the house or just verbally told to leave,” Michael said, “and then the kid does.”
Heightened family conflicts
The Safeline partners with the National Center for Missing and Exploited Children, which, despite widespread public perception, doesn’t work mainly with child abduction cases. Each year, the center assists with 29,000-31,000 cases, and 92% involve “endangered runaways,” said John Bischoff, vice president of the Missing Children Division. These children could be running away from home or foster care.
During the pandemic, the center didn’t spot major changes in its missing child numbers, “which honestly was shocking,” Mr. Bischoff said. “We figured we were either going to see an extreme rise or a decrease.
“But the reasons for the run were changing,” he said.
Many youths were fleeing out of frustration with quarantine restrictions, Mr. Bischoff said, as well as frustration with the unknown and their own lack of control over many situations.
At the runaway hotline, calls have been longer and more intense, with family problems topping the list of concerns. In 2019, about 57% of all contacts mentioned family dynamics. In 2020, that number jumped to 88%, according to Mr. Stern.
Some kids sought support for family problems that involved school. In October 2020, one 13-year-old wrote in the Safeline forum: “My mom constantly yells at me for no reason. I want to leave, but I don’t know how. I have also been really stressed about school because they haven’t been giving me the grades I would normally receive during actual school. She thinks I’m lying and that I don’t care. I just need somebody to help me.”
Many adults are under tremendous strain, too, Michael said.
“Parents might have gotten COVID last month and haven’t been able to work for 2 weeks, and they’re missing a paycheck now. Money is tight, there might not be food, everyone’s angry at everything.”
During the pandemic, the National Runaway Safeline found a 16% increase in contacts citing financial challenges.
Some children have felt confined in unsafe homes or have endured violence, as one 15-year-old reported in the forum: “I am the scapegoat out of four kids. Unfortunately, my mom has always been a toxic person. ... I’m the only kid she still hits really hard. She’s left bruises and scratches recently. ... I just have no solution to this.”
Worsening mental health
Besides family dynamics, mental health emerged as a top concern that youths reported in 2020. “This is something notable. It increased by 30% just in 1 year,” Mr. Stern said.
In November 2020, a 16-year-old wrote: “I can’t ever go outside. I’ve been stuck in the house for a very long time now since quarantine started. I’m scared. ... My mother has been taking her anger out on me emotionally. ... I have severe depression and I need help. Please, if there’s any way I can get out of here, let me know.”
The Safeline also has seen a rise in suicide-related contacts. Among children and teens who had cited a mental health concern, 18% said they were suicidal, Stern said. Most were between ages 12 and 16, but some were younger than 12.
When children couldn’t hang out with peers, they felt even more isolated if parents confiscated their phones, a common punishment, Michael said.
During the winter of 2020-21, “It felt like almost every digital contact was a youth reaching out on their Chromebook because they had gotten their phone taken away and they were either suicidal or considering running away,” he said. “That’s kind of their entire social sphere getting taken away.”
Reality check
Roughly 7 in 10 youths report still being at home when they reach out to the Safeline. Among those who do leave, Michael said, “They’re going sometimes to friends’ houses, oftentimes to a significant other’s house, sometimes to extended family members’ houses. Often, they don’t have a place that they’re planning to go. They just left, and that’s why they’re calling us.”
While some youths have been afraid of catching COVID-19 in general, the coronavirus threat hasn’t deterred those who have decided to run away, Michael said. “Usually, they’re more worried about being returned home.”
Many can’t comprehend the risks of setting off on their own.
In October 2021, a 15-year-old boy posted on the forum that his verbally abusive parents had called him a mistake and said they couldn’t wait for him to move out.
“So I’m going to make their dreams come true,” he wrote. “I’m going to go live in California with my friend who is a young YouTuber. I need help getting money to either fly or get a bus ticket, even though I’m all right with trying to ride a bike or fixing my dirt bike and getting the wagon to pull my stuff. But I’m looking for apartments in Los Angeles so I’m not living on the streets and I’m looking for a job. Please help me. My friend can’t send me money because I don’t have a bank account.”
“Often,” Michael said, “we’re reality-checking kids who want to hitchhike 5 hours away to either a friend’s or the closest shelter that we could find them. Or walk for 5 hours at 3 a.m. or bike, so we try to safety-check that.”
Another concern: online enticement by predators. During the pandemic, the National Center for Missing and Exploited Children saw cases in which children ran away from home “to go meet with someone who may not be who they thought they were talking to online,” Mr. Bischoff said. “It’s certainly something we’re keeping a close eye on.”
Fewer resources in the pandemic
The National Runaway Safeline provides information and referrals to other hotlines and services, including suicide prevention and mental health organizations. When youths have already run away and have no place to go, Michael said, the Safeline tries to find shelter options or seek out a relative who can provide a safe place to stay.
But finding shelters became tougher during the pandemic, when many had no room or shelter supply was limited. Some had to shut down for COVID-19–related deep cleanings, Michael said. Helping youths find transportation, especially with public transportation shutdowns, also was tough.
The Huckleberry House, a six-bed youth shelter in San Francisco, has stayed open throughout the pandemic with limited staffing, said Douglas Styles, PsyD. He’s the executive director of the Huckleberry Youth Programs, which runs the house.
The shelter, which serves Bay Area runaway and homeless youths ages 12-17, hasn’t seen an overall spike in demand, Dr. Styles said. But “what’s expanded is undocumented [youths] and young people who don’t have any family connections in the area, so they’re unaccompanied as well. We’ve seen that here and there throughout the years, but during the pandemic, that population has actually increased quite a bit.”
The Huckleberry House has sheltered children and teens who have run away from all kinds of homes, including affluent ones, Dr. Styles said.
Once children leave home, the lack of adult supervision leaves them vulnerable. They face multiple dangers, including child sex trafficking and exploitation, substance abuse, gang involvement, and violence. “As an organization, that scares us,” Mr. Bischoff said. “What’s happening at home, we’ll sort that out. The biggest thing we as an organization are trying to do is locate them and ensure their safety.”
To help runaways and their families get in touch, the National Runaway Safeline provides a message service and conference calling. “We can play the middleman, really acting on behalf of the young person – not because they’re right or wrong, but to ensure that their voice is really heard,” Mr. Stern said.
Through its national Home Free program, the Safeline partners with Greyhound to bring children back home or into an alternative, safe living environment by providing a free bus ticket.
These days, technology can expose children to harm online, but it can also speed their return home.
“When I was growing up, if you weren’t home by 5 o’clock, Mom would start to worry, but she really didn’t have any way of reaching you,” Mr. Bischoff said. “More children today have cellphones. More children are easily reachable. That’s a benefit.”
A version of this article first appeared on WebMD.com.
Psychiatrist found guilty in $110 million fraud scheme
Mark Agresti, MD, 59, was convicted of unlawfully billing approximately $110 million of medically unnecessary urinalysis services for patients living at Good Decisions Sober Living (GDSL), a drug rehabilitation facility in West Palm Beach, Fla. Dr. Agresti was once the director of psychiatry at the former Columbia Hospital in West Palm Beach.
According to evidence presented at the trial, Dr. Agresti, as medical director of GDSL, agreed to provide standing orders for urinalysis for GDSL patients in exchange for a monthly fee. Three to four times a week, patients were required to submit to excessive and medically unnecessary urine tests, costing $6,000 to $9,000 per test, according to federal officials. Dr. Agresti did not review the results of these tests and did not use the tests to treat patients. The evidence also showed that he had also done this at other addiction facilities in the West Palm Beach area, resulting in thousands of additional fraudulent tests. Dr. Agresti had GDSL patients visit his private practice so he could fraudulently bill for services there as well.
“This defendant used his medical license to facilitate an egregious, multimillion-dollar fraud scheme that exploited vulnerable substance abusers,” said Juan Antonio Gonzalez, U.S. Attorney for the Southern District of Florida, in a statement announcing the conviction.
Three other defendants, including GDSL’s owner, Kenneth Bailynson, were previously indicted and pleaded guilty to related charges connected with the scheme. Dr. Agresti, however, insisted on his innocence throughout the trial, arguing that he was himself the victim of Mr. Bailynson. Mr. Bailynson, as a part of his plea deal, testified against Dr. Agresti at the trial.
According to a report in the Palm Beach Post, Dr. Agresti’s lawyers, in closing statements, argued, “The government did a fantastic job of proving that Dr. Agresti was negligent, maybe even grossly negligent, but that’s not the issue in this case.” The jury disagreed.
Dr. Agresti faces a maximum penalty of 20 years in prison for the healthcare fraud and wire fraud conspiracy charges and 10 years for each count of healthcare fraud. Sentencing is scheduled for April 21.
Ten people, including two physicians, charged in Texas kickback scheme
Jose Maldonado, MD, 48, a family medicine physician, and Eduardo Carlos Canova, MD, 44, an internal medicine specialist, both based in Laredo, Tex., were among ten people indicted in connection with a $300 million healthcare fraud scheme
According to the federal indictment, the founders of several lab companies, including Unified Laboratory Services, Spectrum Diagnostic Laboratory, and Reliable Labs, LLC, allegedly paid kickbacks to medical practitioners in exchange for orders for medically unnecessary lab tests, which were then billed to Medicare and other federal healthcare programs. The medical professionals, including Dr. Maldonado and Dr. Canova, are alleged to have accepted bribes for ordering millions of dollars’ worth of tests.
The scheme was facilitated by marketing firms, which paid doctors hundreds of thousands of dollars for “advisory services,” according to officials. Rather than providing advice, however, the physicians provided lab test referrals. The labs are accused of paying portions of the salaries of the doctors’ staffs as well as portions of their leases. In some instances, direct payments were made to the practitioner’s spouse. In one case, the labs informed one practitioner that the payments would cease if he didn’t refer more tests. He responded to the threat by immediately increasing his lab referrals, averaging 20 to 30 referrals a day.
Jeffrey Madison, the founder of Spectrum and Unified, is accused of convincing Reliable’s founders to convert their business to a physician-owned lab to disguise the kickbacks. Reliable offered physicians ownership opportunities only if they referred enough lab tests. In some cases, says the indictment, the labs made advance payments to physicians to ensure that those physicians would not send samples to other labs.
The scheme resulted in more than $300 million in billing to federal government healthcare programs. Between 2015 and 2018, Dr. Maldonado alone received more than $400,000 in kickbacks for ordering more than $4 million worth of lab tests, and Dr. Canova received more than $300,000 for ordering more than $12 million in tests, according to the indictment.
The 26-count indictment included charges of conspiracy to commit healthcare fraud, conspiracy to pay and receive healthcare kickbacks, offering or paying illegal kickbacks, and soliciting or receiving illegal kickbacks.
N.J. doc charged on 34 counts of illegally prescribing controlled substances
A N.J. orthopedic surgeon has been charged with 34 counts of illegally prescribing controlled substances.
According to documents filed in the case, from November 2016 to February 2018 Evangelos Megariotis, MD, 68, of Passaic County, N.J., prescribed opiates — including oxycodone, alprazolam (Xanax), and cough syrup with codeine — as well as stimulants without a legitimate medical purpose and outside the usual course of his practice.
Before the State of New Jersey permanently banned him from practicing medicine, Dr. Megariotis owned and operated Clifton Orthopedic Associates, in Clifton, N.J.
According to The Bergin Record, the state alleges that Dr. Megariotis prescribed thousands of doses of painkillers even though he knew his patients were abusing them and in some cases were getting similar drugs from other doctors. In addition, Dr. Megariotis failed to diagnose or treat these patients’ underlying conditions, treated illnesses outside his scope of practice, and performed surgery in the absence of medical need.
If convicted, Dr. Megariotis could face a lengthy prison sentence as well as substantial fines. The counts of illegally prescribing Schedule II opioids and stimulants are punishable by a maximum of 20 years in prison and a $1 million fine. Illegally prescribing Schedule IV controlled substances brings a maximum of 5 years in prison and a $250,000 fine. The counts of illegally prescribing cough syrup with codeine, a Schedule V controlled substance, are punishable by a maximum of 1 year in prison and a $100,000 fine.
Michigan surgeon pleads guilty to fraud scheme
A Bay City, Mich., vascular surgeon has pleaded guilty to engaging in a scheme to defraud Medicare, Medicaid, and Blue Cross/Blue Shield of Michigan out of approximately $19.5 million
According to the plea agreement, in 2009, Vasso Godiali, MD, 59, began false and fraudulent billing to the three insurers. False claims included billing for the placement of stents in dialysis patients and for the treatment of arterial blood clots. The surgeon admitted to billing for the placement of multiple stents in the same vessel and preparing medical records to support the necessity of placing those stents. However, he never placed the stents.
In the plea document, Dr. Godiali also admitted that his medical records would describe occluded arteries that would justify arterial thrombectomies. However, in many cases, there were no occluded arteries, and no thrombectomies were performed, even though Dr. Godiali billed insurers for them.
“When Dr Godiali submitted claims for medical services that were never provided, he violated the trust of his patients and defrauded taxpayer-funded health care programs,” Mario M. Pinto, special agent in charge, said in a statement announcing the plea.
In 2014, The Bay City Times interviewed Dr. Godiali after he was featured by USA Today as one of seven physicians in the United States to receive more than $10 million for Medicare reimbursements in 2012. He told The Times he was a workaholic. “I work a lot — I work probably more than surgeons that you meet,” he told the paper.
Dr. Godiali faces up to 10 years in prison. In addition, he will be required to pay $19.5 million in restitution to the three insurers. A civil forfeiture case against approximately $39.9 million seized from accounts controlled by Dr. Godiali is pending. Sentencing is scheduled for September 15.
A version of this article first appeared on Medscape.com.
Mark Agresti, MD, 59, was convicted of unlawfully billing approximately $110 million of medically unnecessary urinalysis services for patients living at Good Decisions Sober Living (GDSL), a drug rehabilitation facility in West Palm Beach, Fla. Dr. Agresti was once the director of psychiatry at the former Columbia Hospital in West Palm Beach.
According to evidence presented at the trial, Dr. Agresti, as medical director of GDSL, agreed to provide standing orders for urinalysis for GDSL patients in exchange for a monthly fee. Three to four times a week, patients were required to submit to excessive and medically unnecessary urine tests, costing $6,000 to $9,000 per test, according to federal officials. Dr. Agresti did not review the results of these tests and did not use the tests to treat patients. The evidence also showed that he had also done this at other addiction facilities in the West Palm Beach area, resulting in thousands of additional fraudulent tests. Dr. Agresti had GDSL patients visit his private practice so he could fraudulently bill for services there as well.
“This defendant used his medical license to facilitate an egregious, multimillion-dollar fraud scheme that exploited vulnerable substance abusers,” said Juan Antonio Gonzalez, U.S. Attorney for the Southern District of Florida, in a statement announcing the conviction.
Three other defendants, including GDSL’s owner, Kenneth Bailynson, were previously indicted and pleaded guilty to related charges connected with the scheme. Dr. Agresti, however, insisted on his innocence throughout the trial, arguing that he was himself the victim of Mr. Bailynson. Mr. Bailynson, as a part of his plea deal, testified against Dr. Agresti at the trial.
According to a report in the Palm Beach Post, Dr. Agresti’s lawyers, in closing statements, argued, “The government did a fantastic job of proving that Dr. Agresti was negligent, maybe even grossly negligent, but that’s not the issue in this case.” The jury disagreed.
Dr. Agresti faces a maximum penalty of 20 years in prison for the healthcare fraud and wire fraud conspiracy charges and 10 years for each count of healthcare fraud. Sentencing is scheduled for April 21.
Ten people, including two physicians, charged in Texas kickback scheme
Jose Maldonado, MD, 48, a family medicine physician, and Eduardo Carlos Canova, MD, 44, an internal medicine specialist, both based in Laredo, Tex., were among ten people indicted in connection with a $300 million healthcare fraud scheme
According to the federal indictment, the founders of several lab companies, including Unified Laboratory Services, Spectrum Diagnostic Laboratory, and Reliable Labs, LLC, allegedly paid kickbacks to medical practitioners in exchange for orders for medically unnecessary lab tests, which were then billed to Medicare and other federal healthcare programs. The medical professionals, including Dr. Maldonado and Dr. Canova, are alleged to have accepted bribes for ordering millions of dollars’ worth of tests.
The scheme was facilitated by marketing firms, which paid doctors hundreds of thousands of dollars for “advisory services,” according to officials. Rather than providing advice, however, the physicians provided lab test referrals. The labs are accused of paying portions of the salaries of the doctors’ staffs as well as portions of their leases. In some instances, direct payments were made to the practitioner’s spouse. In one case, the labs informed one practitioner that the payments would cease if he didn’t refer more tests. He responded to the threat by immediately increasing his lab referrals, averaging 20 to 30 referrals a day.
Jeffrey Madison, the founder of Spectrum and Unified, is accused of convincing Reliable’s founders to convert their business to a physician-owned lab to disguise the kickbacks. Reliable offered physicians ownership opportunities only if they referred enough lab tests. In some cases, says the indictment, the labs made advance payments to physicians to ensure that those physicians would not send samples to other labs.
The scheme resulted in more than $300 million in billing to federal government healthcare programs. Between 2015 and 2018, Dr. Maldonado alone received more than $400,000 in kickbacks for ordering more than $4 million worth of lab tests, and Dr. Canova received more than $300,000 for ordering more than $12 million in tests, according to the indictment.
The 26-count indictment included charges of conspiracy to commit healthcare fraud, conspiracy to pay and receive healthcare kickbacks, offering or paying illegal kickbacks, and soliciting or receiving illegal kickbacks.
N.J. doc charged on 34 counts of illegally prescribing controlled substances
A N.J. orthopedic surgeon has been charged with 34 counts of illegally prescribing controlled substances.
According to documents filed in the case, from November 2016 to February 2018 Evangelos Megariotis, MD, 68, of Passaic County, N.J., prescribed opiates — including oxycodone, alprazolam (Xanax), and cough syrup with codeine — as well as stimulants without a legitimate medical purpose and outside the usual course of his practice.
Before the State of New Jersey permanently banned him from practicing medicine, Dr. Megariotis owned and operated Clifton Orthopedic Associates, in Clifton, N.J.
According to The Bergin Record, the state alleges that Dr. Megariotis prescribed thousands of doses of painkillers even though he knew his patients were abusing them and in some cases were getting similar drugs from other doctors. In addition, Dr. Megariotis failed to diagnose or treat these patients’ underlying conditions, treated illnesses outside his scope of practice, and performed surgery in the absence of medical need.
If convicted, Dr. Megariotis could face a lengthy prison sentence as well as substantial fines. The counts of illegally prescribing Schedule II opioids and stimulants are punishable by a maximum of 20 years in prison and a $1 million fine. Illegally prescribing Schedule IV controlled substances brings a maximum of 5 years in prison and a $250,000 fine. The counts of illegally prescribing cough syrup with codeine, a Schedule V controlled substance, are punishable by a maximum of 1 year in prison and a $100,000 fine.
Michigan surgeon pleads guilty to fraud scheme
A Bay City, Mich., vascular surgeon has pleaded guilty to engaging in a scheme to defraud Medicare, Medicaid, and Blue Cross/Blue Shield of Michigan out of approximately $19.5 million
According to the plea agreement, in 2009, Vasso Godiali, MD, 59, began false and fraudulent billing to the three insurers. False claims included billing for the placement of stents in dialysis patients and for the treatment of arterial blood clots. The surgeon admitted to billing for the placement of multiple stents in the same vessel and preparing medical records to support the necessity of placing those stents. However, he never placed the stents.
In the plea document, Dr. Godiali also admitted that his medical records would describe occluded arteries that would justify arterial thrombectomies. However, in many cases, there were no occluded arteries, and no thrombectomies were performed, even though Dr. Godiali billed insurers for them.
“When Dr Godiali submitted claims for medical services that were never provided, he violated the trust of his patients and defrauded taxpayer-funded health care programs,” Mario M. Pinto, special agent in charge, said in a statement announcing the plea.
In 2014, The Bay City Times interviewed Dr. Godiali after he was featured by USA Today as one of seven physicians in the United States to receive more than $10 million for Medicare reimbursements in 2012. He told The Times he was a workaholic. “I work a lot — I work probably more than surgeons that you meet,” he told the paper.
Dr. Godiali faces up to 10 years in prison. In addition, he will be required to pay $19.5 million in restitution to the three insurers. A civil forfeiture case against approximately $39.9 million seized from accounts controlled by Dr. Godiali is pending. Sentencing is scheduled for September 15.
A version of this article first appeared on Medscape.com.
Mark Agresti, MD, 59, was convicted of unlawfully billing approximately $110 million of medically unnecessary urinalysis services for patients living at Good Decisions Sober Living (GDSL), a drug rehabilitation facility in West Palm Beach, Fla. Dr. Agresti was once the director of psychiatry at the former Columbia Hospital in West Palm Beach.
According to evidence presented at the trial, Dr. Agresti, as medical director of GDSL, agreed to provide standing orders for urinalysis for GDSL patients in exchange for a monthly fee. Three to four times a week, patients were required to submit to excessive and medically unnecessary urine tests, costing $6,000 to $9,000 per test, according to federal officials. Dr. Agresti did not review the results of these tests and did not use the tests to treat patients. The evidence also showed that he had also done this at other addiction facilities in the West Palm Beach area, resulting in thousands of additional fraudulent tests. Dr. Agresti had GDSL patients visit his private practice so he could fraudulently bill for services there as well.
“This defendant used his medical license to facilitate an egregious, multimillion-dollar fraud scheme that exploited vulnerable substance abusers,” said Juan Antonio Gonzalez, U.S. Attorney for the Southern District of Florida, in a statement announcing the conviction.
Three other defendants, including GDSL’s owner, Kenneth Bailynson, were previously indicted and pleaded guilty to related charges connected with the scheme. Dr. Agresti, however, insisted on his innocence throughout the trial, arguing that he was himself the victim of Mr. Bailynson. Mr. Bailynson, as a part of his plea deal, testified against Dr. Agresti at the trial.
According to a report in the Palm Beach Post, Dr. Agresti’s lawyers, in closing statements, argued, “The government did a fantastic job of proving that Dr. Agresti was negligent, maybe even grossly negligent, but that’s not the issue in this case.” The jury disagreed.
Dr. Agresti faces a maximum penalty of 20 years in prison for the healthcare fraud and wire fraud conspiracy charges and 10 years for each count of healthcare fraud. Sentencing is scheduled for April 21.
Ten people, including two physicians, charged in Texas kickback scheme
Jose Maldonado, MD, 48, a family medicine physician, and Eduardo Carlos Canova, MD, 44, an internal medicine specialist, both based in Laredo, Tex., were among ten people indicted in connection with a $300 million healthcare fraud scheme
According to the federal indictment, the founders of several lab companies, including Unified Laboratory Services, Spectrum Diagnostic Laboratory, and Reliable Labs, LLC, allegedly paid kickbacks to medical practitioners in exchange for orders for medically unnecessary lab tests, which were then billed to Medicare and other federal healthcare programs. The medical professionals, including Dr. Maldonado and Dr. Canova, are alleged to have accepted bribes for ordering millions of dollars’ worth of tests.
The scheme was facilitated by marketing firms, which paid doctors hundreds of thousands of dollars for “advisory services,” according to officials. Rather than providing advice, however, the physicians provided lab test referrals. The labs are accused of paying portions of the salaries of the doctors’ staffs as well as portions of their leases. In some instances, direct payments were made to the practitioner’s spouse. In one case, the labs informed one practitioner that the payments would cease if he didn’t refer more tests. He responded to the threat by immediately increasing his lab referrals, averaging 20 to 30 referrals a day.
Jeffrey Madison, the founder of Spectrum and Unified, is accused of convincing Reliable’s founders to convert their business to a physician-owned lab to disguise the kickbacks. Reliable offered physicians ownership opportunities only if they referred enough lab tests. In some cases, says the indictment, the labs made advance payments to physicians to ensure that those physicians would not send samples to other labs.
The scheme resulted in more than $300 million in billing to federal government healthcare programs. Between 2015 and 2018, Dr. Maldonado alone received more than $400,000 in kickbacks for ordering more than $4 million worth of lab tests, and Dr. Canova received more than $300,000 for ordering more than $12 million in tests, according to the indictment.
The 26-count indictment included charges of conspiracy to commit healthcare fraud, conspiracy to pay and receive healthcare kickbacks, offering or paying illegal kickbacks, and soliciting or receiving illegal kickbacks.
N.J. doc charged on 34 counts of illegally prescribing controlled substances
A N.J. orthopedic surgeon has been charged with 34 counts of illegally prescribing controlled substances.
According to documents filed in the case, from November 2016 to February 2018 Evangelos Megariotis, MD, 68, of Passaic County, N.J., prescribed opiates — including oxycodone, alprazolam (Xanax), and cough syrup with codeine — as well as stimulants without a legitimate medical purpose and outside the usual course of his practice.
Before the State of New Jersey permanently banned him from practicing medicine, Dr. Megariotis owned and operated Clifton Orthopedic Associates, in Clifton, N.J.
According to The Bergin Record, the state alleges that Dr. Megariotis prescribed thousands of doses of painkillers even though he knew his patients were abusing them and in some cases were getting similar drugs from other doctors. In addition, Dr. Megariotis failed to diagnose or treat these patients’ underlying conditions, treated illnesses outside his scope of practice, and performed surgery in the absence of medical need.
If convicted, Dr. Megariotis could face a lengthy prison sentence as well as substantial fines. The counts of illegally prescribing Schedule II opioids and stimulants are punishable by a maximum of 20 years in prison and a $1 million fine. Illegally prescribing Schedule IV controlled substances brings a maximum of 5 years in prison and a $250,000 fine. The counts of illegally prescribing cough syrup with codeine, a Schedule V controlled substance, are punishable by a maximum of 1 year in prison and a $100,000 fine.
Michigan surgeon pleads guilty to fraud scheme
A Bay City, Mich., vascular surgeon has pleaded guilty to engaging in a scheme to defraud Medicare, Medicaid, and Blue Cross/Blue Shield of Michigan out of approximately $19.5 million
According to the plea agreement, in 2009, Vasso Godiali, MD, 59, began false and fraudulent billing to the three insurers. False claims included billing for the placement of stents in dialysis patients and for the treatment of arterial blood clots. The surgeon admitted to billing for the placement of multiple stents in the same vessel and preparing medical records to support the necessity of placing those stents. However, he never placed the stents.
In the plea document, Dr. Godiali also admitted that his medical records would describe occluded arteries that would justify arterial thrombectomies. However, in many cases, there were no occluded arteries, and no thrombectomies were performed, even though Dr. Godiali billed insurers for them.
“When Dr Godiali submitted claims for medical services that were never provided, he violated the trust of his patients and defrauded taxpayer-funded health care programs,” Mario M. Pinto, special agent in charge, said in a statement announcing the plea.
In 2014, The Bay City Times interviewed Dr. Godiali after he was featured by USA Today as one of seven physicians in the United States to receive more than $10 million for Medicare reimbursements in 2012. He told The Times he was a workaholic. “I work a lot — I work probably more than surgeons that you meet,” he told the paper.
Dr. Godiali faces up to 10 years in prison. In addition, he will be required to pay $19.5 million in restitution to the three insurers. A civil forfeiture case against approximately $39.9 million seized from accounts controlled by Dr. Godiali is pending. Sentencing is scheduled for September 15.
A version of this article first appeared on Medscape.com.
Robotic transcranial Doppler improves PFO detection after stroke
in a new study.
Being far easier to perform than regular transcranial Doppler ultrasound, it’s hoped that use of the robotic device will enable many more patients to undergo the more sensitive transcranial screening modality and increase the number of shunts identified.
“I believe robot-assisted transcranial Doppler ultrasound can fill the gap between the gold standard transcranial Doppler and transthoracic echocardiography, which is the current standard of care,” said lead author Mark Rubin, MD.
Dr. Rubin, who is assistant professor of neurology at University of Tennessee Health Science Center, Memphis, presented results of the BUBL study at the International Stroke Conference (ISC) 2022, where they were greeted with applause from the floor.
An improvement in the current standard of care
Dr. Rubin explained that patients with suspected embolic stroke are routinely screened for shunts in the heart, such as patent foramen ovale (PFO), that allow blood to flow from the right chamber to the left chamber and can lead to clots from the venous system, accessing the arterial system, then traveling to the brain and causing a stroke.
The current standard of care in screening for such shunts is the use of transthoracic echocardiography (TTE), a widely available and easy to perform, non-invasive procedure. “But we have known for decades that TTE does not pick up these shunts very well. With a sensitivity of only around 45%, it identifies less than half of the patients affected,” Dr. Rubin noted.
The more sensitive transesophageal echocardiography (TEE) gives much better results, but it is an invasive and unpleasant procedure with the ultrasound probe being passed down the throat, and the patient needing to be sedated, so it’s not appropriate for everyone, he noted.
“Transcranial Doppler ultrasound (TCD) also gives excellent results, with a sensitivity of about 96% for detecting PFO, but this procedure is difficult to perform and requires a great deal of skill in placing the probes in the right position and interpreting the signal,” Dr. Rubin said. “TCD has been around for decades, but it hasn’t caught on, as it is too difficult to do. It takes a lot of time to learn the technique.”
“With the robotic-assisted transcranial Doppler device, we can achieve the sensitivity of TCD without needing expert operators. This should vastly improve accessibility to this technology,” he said. “With such technology we can make significant strides into more accurate diagnoses on the cause of stroke, which should lead to better preventive treatments in those found to have right-to-left shunts.”
Robotic detection of shunts
For the BUBL study, the robotic TCD technique was compared with the standard TTE in 129 patients who had a diagnosis of presumed embolic stroke or transient ischemic attack (TIA), with all patients undergoing both procedures.
The robotic TCD device resembles a giant pair of headphones containing the ultrasound probes, which are attached to a frame. In the study, it was operated by a health care professional without TCD skills. Each ultrasound probe independently scans the temporal area autonomously – with angling and positive pressure against the scalp akin to a sonographer – to find and optimize bilateral middle cerebral artery signals, Dr. Rubin explained.
The primary endpoint was the detection of a right-to-left shunt. This occurred in 82 of the 129 patients (63.6%) with the robotic TCD device but in only 27 patients (20.9%) when TTE was used. This gives an absolute difference of 42.6% (95% confidence interval, 28.6%-56.7%; P < .001), which Dr. Rubin described as “astounding.”
However, he said he was not surprised by these results.
“In my experience with transcranial Doppler, I find shunts in patients every day that have not been seen with transthoracic echo,” he commented.
He noted that a previous meta-analysis has suggested a similar difference between TCD and transthoracic echo, but the current study provides prospectively collected data produced in a clinical trial setting and is therefore more reliable.
“What I hope comes from this is that more patients will be able to undergo transcranial Doppler, which is a far superior screening technique for identifying right-to-left shunts. There is so much evidence to support the use of transcranial Doppler, but with this new artificial-intelligence robotic device, we don’t need an expert to use it,” Dr. Rubin said.
He explained that finding a right-to-left shunt in stroke patients is particularly important, as it can direct treatment strategies to reduce future risk of recurrent strokes.
“If a patient has a large shunt, then they have a high risk of having another stroke, and the PFO should be closed.”
In this study, the robotic-assisted TCD detected three times as many large shunts that were considered “intervenable,” compared with transthoracic echo, identifying these shunts in 35 patients (27%) compared to just 13 (10%) with TTE.
“Of the 35 patients with intervenable shunts detected with robotic transcranial Doppler, TTE was completely negative in 18 of them and only suggested a small shunt in the others. So, the standard of care (TTE) missed half the patients with intervenable PFOs,” Dr. Rubin reported.
Study should ‘dramatically change’ practice
Commenting on the study, Patrick Lyden, MD, professor of physiology and neuroscience and of neurology, University of Southern California, Los Angeles, said: “Most clinicians hesitate to use transcranial Doppler given the need for specialized technical expertise to obtain a reliable result. This study showed that a robotic transcranial Doppler device – which can be applied by any cardiac non-invasive lab technician – provides reliable and rigorous data.”
He added: “This result will dramatically change the typical evaluation of patients with suspected PFO: In place of an invasive transesophageal echo that requires anesthesia and a cardiologist, most patients can have a non-invasive, robotic-guided transcranial Doppler and get the same diagnostic benefit.”
Dr. Lyden also pointed out that the cost of TCD is typically one-tenth that of TEE, although he said the cost of the robotic guided TCD “is not clear.”
A representative of the company that makes the robotic assisted device, NovaSignal, says the cost of the equipment is approximately $250,000, but “understanding the importance of the technology, we work with each hospital to meet their unique needs.”
The company adds that it currently has “over 45 commercial solutions deployed across 25 centers with 3-4 times growth expected year over year.”
The study was supported by NovaSignal, the company which makes the robotic device. Dr. Rubin reports acting as a consultant for the NovaSignal.
A version of this article first appeared on Medscape.com.
in a new study.
Being far easier to perform than regular transcranial Doppler ultrasound, it’s hoped that use of the robotic device will enable many more patients to undergo the more sensitive transcranial screening modality and increase the number of shunts identified.
“I believe robot-assisted transcranial Doppler ultrasound can fill the gap between the gold standard transcranial Doppler and transthoracic echocardiography, which is the current standard of care,” said lead author Mark Rubin, MD.
Dr. Rubin, who is assistant professor of neurology at University of Tennessee Health Science Center, Memphis, presented results of the BUBL study at the International Stroke Conference (ISC) 2022, where they were greeted with applause from the floor.
An improvement in the current standard of care
Dr. Rubin explained that patients with suspected embolic stroke are routinely screened for shunts in the heart, such as patent foramen ovale (PFO), that allow blood to flow from the right chamber to the left chamber and can lead to clots from the venous system, accessing the arterial system, then traveling to the brain and causing a stroke.
The current standard of care in screening for such shunts is the use of transthoracic echocardiography (TTE), a widely available and easy to perform, non-invasive procedure. “But we have known for decades that TTE does not pick up these shunts very well. With a sensitivity of only around 45%, it identifies less than half of the patients affected,” Dr. Rubin noted.
The more sensitive transesophageal echocardiography (TEE) gives much better results, but it is an invasive and unpleasant procedure with the ultrasound probe being passed down the throat, and the patient needing to be sedated, so it’s not appropriate for everyone, he noted.
“Transcranial Doppler ultrasound (TCD) also gives excellent results, with a sensitivity of about 96% for detecting PFO, but this procedure is difficult to perform and requires a great deal of skill in placing the probes in the right position and interpreting the signal,” Dr. Rubin said. “TCD has been around for decades, but it hasn’t caught on, as it is too difficult to do. It takes a lot of time to learn the technique.”
“With the robotic-assisted transcranial Doppler device, we can achieve the sensitivity of TCD without needing expert operators. This should vastly improve accessibility to this technology,” he said. “With such technology we can make significant strides into more accurate diagnoses on the cause of stroke, which should lead to better preventive treatments in those found to have right-to-left shunts.”
Robotic detection of shunts
For the BUBL study, the robotic TCD technique was compared with the standard TTE in 129 patients who had a diagnosis of presumed embolic stroke or transient ischemic attack (TIA), with all patients undergoing both procedures.
The robotic TCD device resembles a giant pair of headphones containing the ultrasound probes, which are attached to a frame. In the study, it was operated by a health care professional without TCD skills. Each ultrasound probe independently scans the temporal area autonomously – with angling and positive pressure against the scalp akin to a sonographer – to find and optimize bilateral middle cerebral artery signals, Dr. Rubin explained.
The primary endpoint was the detection of a right-to-left shunt. This occurred in 82 of the 129 patients (63.6%) with the robotic TCD device but in only 27 patients (20.9%) when TTE was used. This gives an absolute difference of 42.6% (95% confidence interval, 28.6%-56.7%; P < .001), which Dr. Rubin described as “astounding.”
However, he said he was not surprised by these results.
“In my experience with transcranial Doppler, I find shunts in patients every day that have not been seen with transthoracic echo,” he commented.
He noted that a previous meta-analysis has suggested a similar difference between TCD and transthoracic echo, but the current study provides prospectively collected data produced in a clinical trial setting and is therefore more reliable.
“What I hope comes from this is that more patients will be able to undergo transcranial Doppler, which is a far superior screening technique for identifying right-to-left shunts. There is so much evidence to support the use of transcranial Doppler, but with this new artificial-intelligence robotic device, we don’t need an expert to use it,” Dr. Rubin said.
He explained that finding a right-to-left shunt in stroke patients is particularly important, as it can direct treatment strategies to reduce future risk of recurrent strokes.
“If a patient has a large shunt, then they have a high risk of having another stroke, and the PFO should be closed.”
In this study, the robotic-assisted TCD detected three times as many large shunts that were considered “intervenable,” compared with transthoracic echo, identifying these shunts in 35 patients (27%) compared to just 13 (10%) with TTE.
“Of the 35 patients with intervenable shunts detected with robotic transcranial Doppler, TTE was completely negative in 18 of them and only suggested a small shunt in the others. So, the standard of care (TTE) missed half the patients with intervenable PFOs,” Dr. Rubin reported.
Study should ‘dramatically change’ practice
Commenting on the study, Patrick Lyden, MD, professor of physiology and neuroscience and of neurology, University of Southern California, Los Angeles, said: “Most clinicians hesitate to use transcranial Doppler given the need for specialized technical expertise to obtain a reliable result. This study showed that a robotic transcranial Doppler device – which can be applied by any cardiac non-invasive lab technician – provides reliable and rigorous data.”
He added: “This result will dramatically change the typical evaluation of patients with suspected PFO: In place of an invasive transesophageal echo that requires anesthesia and a cardiologist, most patients can have a non-invasive, robotic-guided transcranial Doppler and get the same diagnostic benefit.”
Dr. Lyden also pointed out that the cost of TCD is typically one-tenth that of TEE, although he said the cost of the robotic guided TCD “is not clear.”
A representative of the company that makes the robotic assisted device, NovaSignal, says the cost of the equipment is approximately $250,000, but “understanding the importance of the technology, we work with each hospital to meet their unique needs.”
The company adds that it currently has “over 45 commercial solutions deployed across 25 centers with 3-4 times growth expected year over year.”
The study was supported by NovaSignal, the company which makes the robotic device. Dr. Rubin reports acting as a consultant for the NovaSignal.
A version of this article first appeared on Medscape.com.
in a new study.
Being far easier to perform than regular transcranial Doppler ultrasound, it’s hoped that use of the robotic device will enable many more patients to undergo the more sensitive transcranial screening modality and increase the number of shunts identified.
“I believe robot-assisted transcranial Doppler ultrasound can fill the gap between the gold standard transcranial Doppler and transthoracic echocardiography, which is the current standard of care,” said lead author Mark Rubin, MD.
Dr. Rubin, who is assistant professor of neurology at University of Tennessee Health Science Center, Memphis, presented results of the BUBL study at the International Stroke Conference (ISC) 2022, where they were greeted with applause from the floor.
An improvement in the current standard of care
Dr. Rubin explained that patients with suspected embolic stroke are routinely screened for shunts in the heart, such as patent foramen ovale (PFO), that allow blood to flow from the right chamber to the left chamber and can lead to clots from the venous system, accessing the arterial system, then traveling to the brain and causing a stroke.
The current standard of care in screening for such shunts is the use of transthoracic echocardiography (TTE), a widely available and easy to perform, non-invasive procedure. “But we have known for decades that TTE does not pick up these shunts very well. With a sensitivity of only around 45%, it identifies less than half of the patients affected,” Dr. Rubin noted.
The more sensitive transesophageal echocardiography (TEE) gives much better results, but it is an invasive and unpleasant procedure with the ultrasound probe being passed down the throat, and the patient needing to be sedated, so it’s not appropriate for everyone, he noted.
“Transcranial Doppler ultrasound (TCD) also gives excellent results, with a sensitivity of about 96% for detecting PFO, but this procedure is difficult to perform and requires a great deal of skill in placing the probes in the right position and interpreting the signal,” Dr. Rubin said. “TCD has been around for decades, but it hasn’t caught on, as it is too difficult to do. It takes a lot of time to learn the technique.”
“With the robotic-assisted transcranial Doppler device, we can achieve the sensitivity of TCD without needing expert operators. This should vastly improve accessibility to this technology,” he said. “With such technology we can make significant strides into more accurate diagnoses on the cause of stroke, which should lead to better preventive treatments in those found to have right-to-left shunts.”
Robotic detection of shunts
For the BUBL study, the robotic TCD technique was compared with the standard TTE in 129 patients who had a diagnosis of presumed embolic stroke or transient ischemic attack (TIA), with all patients undergoing both procedures.
The robotic TCD device resembles a giant pair of headphones containing the ultrasound probes, which are attached to a frame. In the study, it was operated by a health care professional without TCD skills. Each ultrasound probe independently scans the temporal area autonomously – with angling and positive pressure against the scalp akin to a sonographer – to find and optimize bilateral middle cerebral artery signals, Dr. Rubin explained.
The primary endpoint was the detection of a right-to-left shunt. This occurred in 82 of the 129 patients (63.6%) with the robotic TCD device but in only 27 patients (20.9%) when TTE was used. This gives an absolute difference of 42.6% (95% confidence interval, 28.6%-56.7%; P < .001), which Dr. Rubin described as “astounding.”
However, he said he was not surprised by these results.
“In my experience with transcranial Doppler, I find shunts in patients every day that have not been seen with transthoracic echo,” he commented.
He noted that a previous meta-analysis has suggested a similar difference between TCD and transthoracic echo, but the current study provides prospectively collected data produced in a clinical trial setting and is therefore more reliable.
“What I hope comes from this is that more patients will be able to undergo transcranial Doppler, which is a far superior screening technique for identifying right-to-left shunts. There is so much evidence to support the use of transcranial Doppler, but with this new artificial-intelligence robotic device, we don’t need an expert to use it,” Dr. Rubin said.
He explained that finding a right-to-left shunt in stroke patients is particularly important, as it can direct treatment strategies to reduce future risk of recurrent strokes.
“If a patient has a large shunt, then they have a high risk of having another stroke, and the PFO should be closed.”
In this study, the robotic-assisted TCD detected three times as many large shunts that were considered “intervenable,” compared with transthoracic echo, identifying these shunts in 35 patients (27%) compared to just 13 (10%) with TTE.
“Of the 35 patients with intervenable shunts detected with robotic transcranial Doppler, TTE was completely negative in 18 of them and only suggested a small shunt in the others. So, the standard of care (TTE) missed half the patients with intervenable PFOs,” Dr. Rubin reported.
Study should ‘dramatically change’ practice
Commenting on the study, Patrick Lyden, MD, professor of physiology and neuroscience and of neurology, University of Southern California, Los Angeles, said: “Most clinicians hesitate to use transcranial Doppler given the need for specialized technical expertise to obtain a reliable result. This study showed that a robotic transcranial Doppler device – which can be applied by any cardiac non-invasive lab technician – provides reliable and rigorous data.”
He added: “This result will dramatically change the typical evaluation of patients with suspected PFO: In place of an invasive transesophageal echo that requires anesthesia and a cardiologist, most patients can have a non-invasive, robotic-guided transcranial Doppler and get the same diagnostic benefit.”
Dr. Lyden also pointed out that the cost of TCD is typically one-tenth that of TEE, although he said the cost of the robotic guided TCD “is not clear.”
A representative of the company that makes the robotic assisted device, NovaSignal, says the cost of the equipment is approximately $250,000, but “understanding the importance of the technology, we work with each hospital to meet their unique needs.”
The company adds that it currently has “over 45 commercial solutions deployed across 25 centers with 3-4 times growth expected year over year.”
The study was supported by NovaSignal, the company which makes the robotic device. Dr. Rubin reports acting as a consultant for the NovaSignal.
A version of this article first appeared on Medscape.com.
FROM ISC 2022
USPSTF tweaks primary prevention statin recommendations in new draft guidance
Given the expansive contemporary role of statins for primary cardiovascular disease (CVD) prevention, the language in the new U.S. Preventive Services Task Force draft guidance on their use in that setting may seem conservative. Even so, the proposed recommendations, open to public comment until March 21, take more recent data into account but don’t substantially vary from the 2016 USPSTF document they are intended to replace.
The task force concluded “with moderate certainty” that a statin prescription will clinically benefit adults aged 40-75 years without CVD but with at least one of several risk factors, such as dyslipidemia or diabetes, who have a 10-year CVD risk of at least 7.5%.
the new report states. That, says an accompanying USPSTF press release, means such people “may benefit from statin use and should decide with their clinician if taking a statin is right for them.”
Also, notes the report, the net benefit of statin therapy is “at least moderate” for individuals with a 10% or greater CVD risk over the next decade who, the press release states, “should take a statin to prevent a first heart attack or stroke.”
The evidence review on which the task force based the guidance, the report says, lacked sufficient basis for determining statin benefit versus risk in adults older than 75 years without a history of CVD. “In the absence of this evidence, clinicians should use their judgment as to whether to offer a statin to a patient in this age group,” according to the press release.
The review focused on 22 clinical trials for data on the statin benefits and saw significantly decreased associated risks for death from any cause, fatal or nonfatal stroke, and fatal or nonfatal myocardial infarction with treatment. The combined trial populations exceeded 85,000 for assessing all-cause mortality and 76,000 for each of the other two endpoints.
To assess any potential statin therapy harms, the evidence review covered 19 clinical trials with a combined enrollment of about 75,000 – two more trials than considered in the 2016 document – plus three observational studies with more than 400,000 participants. Statins were found not to be associated with an increased risk for study withdrawal because of adverse events, nor were there signs of greater risk for myalgia or new-onset diabetes, compared with placebo.
“A majority of the trials reviewed by the USPSTF used moderate-intensity statin therapy,” the report states. “Based on available evidence, use of moderate-intensity statin therapy seems reasonable for the primary prevention of CVD in most persons.”
A version of this article first appeared on Medscape.com.
Given the expansive contemporary role of statins for primary cardiovascular disease (CVD) prevention, the language in the new U.S. Preventive Services Task Force draft guidance on their use in that setting may seem conservative. Even so, the proposed recommendations, open to public comment until March 21, take more recent data into account but don’t substantially vary from the 2016 USPSTF document they are intended to replace.
The task force concluded “with moderate certainty” that a statin prescription will clinically benefit adults aged 40-75 years without CVD but with at least one of several risk factors, such as dyslipidemia or diabetes, who have a 10-year CVD risk of at least 7.5%.
the new report states. That, says an accompanying USPSTF press release, means such people “may benefit from statin use and should decide with their clinician if taking a statin is right for them.”
Also, notes the report, the net benefit of statin therapy is “at least moderate” for individuals with a 10% or greater CVD risk over the next decade who, the press release states, “should take a statin to prevent a first heart attack or stroke.”
The evidence review on which the task force based the guidance, the report says, lacked sufficient basis for determining statin benefit versus risk in adults older than 75 years without a history of CVD. “In the absence of this evidence, clinicians should use their judgment as to whether to offer a statin to a patient in this age group,” according to the press release.
The review focused on 22 clinical trials for data on the statin benefits and saw significantly decreased associated risks for death from any cause, fatal or nonfatal stroke, and fatal or nonfatal myocardial infarction with treatment. The combined trial populations exceeded 85,000 for assessing all-cause mortality and 76,000 for each of the other two endpoints.
To assess any potential statin therapy harms, the evidence review covered 19 clinical trials with a combined enrollment of about 75,000 – two more trials than considered in the 2016 document – plus three observational studies with more than 400,000 participants. Statins were found not to be associated with an increased risk for study withdrawal because of adverse events, nor were there signs of greater risk for myalgia or new-onset diabetes, compared with placebo.
“A majority of the trials reviewed by the USPSTF used moderate-intensity statin therapy,” the report states. “Based on available evidence, use of moderate-intensity statin therapy seems reasonable for the primary prevention of CVD in most persons.”
A version of this article first appeared on Medscape.com.
Given the expansive contemporary role of statins for primary cardiovascular disease (CVD) prevention, the language in the new U.S. Preventive Services Task Force draft guidance on their use in that setting may seem conservative. Even so, the proposed recommendations, open to public comment until March 21, take more recent data into account but don’t substantially vary from the 2016 USPSTF document they are intended to replace.
The task force concluded “with moderate certainty” that a statin prescription will clinically benefit adults aged 40-75 years without CVD but with at least one of several risk factors, such as dyslipidemia or diabetes, who have a 10-year CVD risk of at least 7.5%.
the new report states. That, says an accompanying USPSTF press release, means such people “may benefit from statin use and should decide with their clinician if taking a statin is right for them.”
Also, notes the report, the net benefit of statin therapy is “at least moderate” for individuals with a 10% or greater CVD risk over the next decade who, the press release states, “should take a statin to prevent a first heart attack or stroke.”
The evidence review on which the task force based the guidance, the report says, lacked sufficient basis for determining statin benefit versus risk in adults older than 75 years without a history of CVD. “In the absence of this evidence, clinicians should use their judgment as to whether to offer a statin to a patient in this age group,” according to the press release.
The review focused on 22 clinical trials for data on the statin benefits and saw significantly decreased associated risks for death from any cause, fatal or nonfatal stroke, and fatal or nonfatal myocardial infarction with treatment. The combined trial populations exceeded 85,000 for assessing all-cause mortality and 76,000 for each of the other two endpoints.
To assess any potential statin therapy harms, the evidence review covered 19 clinical trials with a combined enrollment of about 75,000 – two more trials than considered in the 2016 document – plus three observational studies with more than 400,000 participants. Statins were found not to be associated with an increased risk for study withdrawal because of adverse events, nor were there signs of greater risk for myalgia or new-onset diabetes, compared with placebo.
“A majority of the trials reviewed by the USPSTF used moderate-intensity statin therapy,” the report states. “Based on available evidence, use of moderate-intensity statin therapy seems reasonable for the primary prevention of CVD in most persons.”
A version of this article first appeared on Medscape.com.
‘In the presence of kindness’: humanitarian Paul Farmer dies
Renowned infectious disease specialist, humanitarian, and healthcare champion for many of the world’s most vulnerable patient populations, Paul Edward Farmer, MD, died suddenly in his sleep from an acute cardiac event on Feb. 21 in Rwanda, where he had been teaching. He was 62.
Dr. Farmer cofounded the Boston-based global nonprofit Partners In Health and spent decades providing healthcare to impoverished communities worldwide, fighting on the frontline to protect underserved communities against deadly pandemics.
Dr. Farmer was the Kolokotrones University Professor and chair of the department of global health and social medicine in the Blavatnik Institute at Harvard Medical School, Boston. He served as chief of the division of global health equity at Brigham and Women’s Hospital, also in Boston.
“Paul dedicated his life to improving human health and advocating for health equity and social justice on a global scale,” said HMS dean George Q. Daley in a letter to the HMS community. “I am particularly shaken by his passing because he was not only a consummate colleague and a beloved mentor, but a close friend. To me, Paul represented the heart and soul of Harvard Medical School.”
He was also chancellor and cofounder of the University of Global Health Equity in Kigali, Rwanda. Before his death, he spent several weeks teaching at the university.
“Paul Farmer’s loss is devastating, but his vision for the world will live on through Partners In Health,” said Partners In Health CEO Sheila Davis in a statement. “Paul taught all those around him the power of accompaniment, love for one another, and solidarity. Our deepest sympathies are with his family.”
Dr. Farmer was born in North Adams, Mass., and grew up in Florida with his parents and five siblings. He attended Duke University on a Benjamin N. Duke Scholarship and received his medical degree in 1988, followed by his PhD in 1990 from Harvard University.
His humanitarian work began when he was a college student volunteering in Haiti in 1983 working with dispossessed farmers. In 1987, he cofounded Partners In Health with the goal of helping patients in poverty-stricken corners of the world.
Under Dr. Farmer’s leadership, the nonprofit tackled major public health crises: Haiti’s devastating 2010 earthquake, drug-resistant tuberculosis in Peru and other countries, and an Ebola outbreak that tore through West Africa.
Dr. Farmer documented his 2014-2015 experience treating Africa’s Ebola patients in a book called “Fevers, Feuds, and Diamonds: Ebola and the Ravages of History.”
He wrote that by the time he arrived, “western Sierra Leone was ground zero of the epidemic, and Upper West Africa was just about the worst place in the world to be critically ill or injured.”
One of his greatest qualities was his ability to connect with patients – to treat them “not like ones who suffered, but like a pal you’d joke with,” said Pardis Sabeti, MD, PhD, a Harvard University geneticist who also spent time in Africa and famously sequenced samples of the Ebola virus’ genome.
Dr. Sabeti and Dr. Farmer bonded over their love for Sierra Leone, and their grief over losing a close colleague to Ebola, Sheik Humarr Khan, who was one of the area’s leading infectious disease experts.
Dr. Sabeti first met Dr. Farmer years earlier as a first-year Harvard medical student when she enrolled in one of his courses. She said students introduced themselves, one by one, each veering into heartfelt testimonies about what Dr. Farmer’s work had meant to them.
Dr. Farmer and Dr. Sabeti were just texting on Feb. 19, and the two were “goofing around in our usual way, and scheming about how to make the world better, as we always did.”
Dr. Farmer was funny, mischievous, and above all, exactly what you would expect upon meeting him, Dr. Sabeti said.
“It’s cliché, but the energetic kick you get from just being in his presence, it’s almost otherworldly,” she said. “It’s not even otherworldly in the sense of: ‘I just came across – greatness.’ It’s more: ‘I just came across kindness.’ ”
Dr. Farmer’s work has been widely distributed in publications including Bulletin of the World Health Organization, The Lancet, the New England Journal of Medicine, Clinical Infectious Diseases, and Social Science & Medicine.
He was awarded the 2020 Berggruen Prize for Philosophy & Culture, the Margaret Mead Award from the American Anthropological Association, the American Medical Association’s Outstanding International Physician (Nathan Davis) Award, and, with his Partners In Health colleagues, the Hilton Humanitarian Prize.
He is survived by his wife, Didi Bertrand Farmer, and their three children.
A verison of this article first appeared on Medscape.com.
Renowned infectious disease specialist, humanitarian, and healthcare champion for many of the world’s most vulnerable patient populations, Paul Edward Farmer, MD, died suddenly in his sleep from an acute cardiac event on Feb. 21 in Rwanda, where he had been teaching. He was 62.
Dr. Farmer cofounded the Boston-based global nonprofit Partners In Health and spent decades providing healthcare to impoverished communities worldwide, fighting on the frontline to protect underserved communities against deadly pandemics.
Dr. Farmer was the Kolokotrones University Professor and chair of the department of global health and social medicine in the Blavatnik Institute at Harvard Medical School, Boston. He served as chief of the division of global health equity at Brigham and Women’s Hospital, also in Boston.
“Paul dedicated his life to improving human health and advocating for health equity and social justice on a global scale,” said HMS dean George Q. Daley in a letter to the HMS community. “I am particularly shaken by his passing because he was not only a consummate colleague and a beloved mentor, but a close friend. To me, Paul represented the heart and soul of Harvard Medical School.”
He was also chancellor and cofounder of the University of Global Health Equity in Kigali, Rwanda. Before his death, he spent several weeks teaching at the university.
“Paul Farmer’s loss is devastating, but his vision for the world will live on through Partners In Health,” said Partners In Health CEO Sheila Davis in a statement. “Paul taught all those around him the power of accompaniment, love for one another, and solidarity. Our deepest sympathies are with his family.”
Dr. Farmer was born in North Adams, Mass., and grew up in Florida with his parents and five siblings. He attended Duke University on a Benjamin N. Duke Scholarship and received his medical degree in 1988, followed by his PhD in 1990 from Harvard University.
His humanitarian work began when he was a college student volunteering in Haiti in 1983 working with dispossessed farmers. In 1987, he cofounded Partners In Health with the goal of helping patients in poverty-stricken corners of the world.
Under Dr. Farmer’s leadership, the nonprofit tackled major public health crises: Haiti’s devastating 2010 earthquake, drug-resistant tuberculosis in Peru and other countries, and an Ebola outbreak that tore through West Africa.
Dr. Farmer documented his 2014-2015 experience treating Africa’s Ebola patients in a book called “Fevers, Feuds, and Diamonds: Ebola and the Ravages of History.”
He wrote that by the time he arrived, “western Sierra Leone was ground zero of the epidemic, and Upper West Africa was just about the worst place in the world to be critically ill or injured.”
One of his greatest qualities was his ability to connect with patients – to treat them “not like ones who suffered, but like a pal you’d joke with,” said Pardis Sabeti, MD, PhD, a Harvard University geneticist who also spent time in Africa and famously sequenced samples of the Ebola virus’ genome.
Dr. Sabeti and Dr. Farmer bonded over their love for Sierra Leone, and their grief over losing a close colleague to Ebola, Sheik Humarr Khan, who was one of the area’s leading infectious disease experts.
Dr. Sabeti first met Dr. Farmer years earlier as a first-year Harvard medical student when she enrolled in one of his courses. She said students introduced themselves, one by one, each veering into heartfelt testimonies about what Dr. Farmer’s work had meant to them.
Dr. Farmer and Dr. Sabeti were just texting on Feb. 19, and the two were “goofing around in our usual way, and scheming about how to make the world better, as we always did.”
Dr. Farmer was funny, mischievous, and above all, exactly what you would expect upon meeting him, Dr. Sabeti said.
“It’s cliché, but the energetic kick you get from just being in his presence, it’s almost otherworldly,” she said. “It’s not even otherworldly in the sense of: ‘I just came across – greatness.’ It’s more: ‘I just came across kindness.’ ”
Dr. Farmer’s work has been widely distributed in publications including Bulletin of the World Health Organization, The Lancet, the New England Journal of Medicine, Clinical Infectious Diseases, and Social Science & Medicine.
He was awarded the 2020 Berggruen Prize for Philosophy & Culture, the Margaret Mead Award from the American Anthropological Association, the American Medical Association’s Outstanding International Physician (Nathan Davis) Award, and, with his Partners In Health colleagues, the Hilton Humanitarian Prize.
He is survived by his wife, Didi Bertrand Farmer, and their three children.
A verison of this article first appeared on Medscape.com.
Renowned infectious disease specialist, humanitarian, and healthcare champion for many of the world’s most vulnerable patient populations, Paul Edward Farmer, MD, died suddenly in his sleep from an acute cardiac event on Feb. 21 in Rwanda, where he had been teaching. He was 62.
Dr. Farmer cofounded the Boston-based global nonprofit Partners In Health and spent decades providing healthcare to impoverished communities worldwide, fighting on the frontline to protect underserved communities against deadly pandemics.
Dr. Farmer was the Kolokotrones University Professor and chair of the department of global health and social medicine in the Blavatnik Institute at Harvard Medical School, Boston. He served as chief of the division of global health equity at Brigham and Women’s Hospital, also in Boston.
“Paul dedicated his life to improving human health and advocating for health equity and social justice on a global scale,” said HMS dean George Q. Daley in a letter to the HMS community. “I am particularly shaken by his passing because he was not only a consummate colleague and a beloved mentor, but a close friend. To me, Paul represented the heart and soul of Harvard Medical School.”
He was also chancellor and cofounder of the University of Global Health Equity in Kigali, Rwanda. Before his death, he spent several weeks teaching at the university.
“Paul Farmer’s loss is devastating, but his vision for the world will live on through Partners In Health,” said Partners In Health CEO Sheila Davis in a statement. “Paul taught all those around him the power of accompaniment, love for one another, and solidarity. Our deepest sympathies are with his family.”
Dr. Farmer was born in North Adams, Mass., and grew up in Florida with his parents and five siblings. He attended Duke University on a Benjamin N. Duke Scholarship and received his medical degree in 1988, followed by his PhD in 1990 from Harvard University.
His humanitarian work began when he was a college student volunteering in Haiti in 1983 working with dispossessed farmers. In 1987, he cofounded Partners In Health with the goal of helping patients in poverty-stricken corners of the world.
Under Dr. Farmer’s leadership, the nonprofit tackled major public health crises: Haiti’s devastating 2010 earthquake, drug-resistant tuberculosis in Peru and other countries, and an Ebola outbreak that tore through West Africa.
Dr. Farmer documented his 2014-2015 experience treating Africa’s Ebola patients in a book called “Fevers, Feuds, and Diamonds: Ebola and the Ravages of History.”
He wrote that by the time he arrived, “western Sierra Leone was ground zero of the epidemic, and Upper West Africa was just about the worst place in the world to be critically ill or injured.”
One of his greatest qualities was his ability to connect with patients – to treat them “not like ones who suffered, but like a pal you’d joke with,” said Pardis Sabeti, MD, PhD, a Harvard University geneticist who also spent time in Africa and famously sequenced samples of the Ebola virus’ genome.
Dr. Sabeti and Dr. Farmer bonded over their love for Sierra Leone, and their grief over losing a close colleague to Ebola, Sheik Humarr Khan, who was one of the area’s leading infectious disease experts.
Dr. Sabeti first met Dr. Farmer years earlier as a first-year Harvard medical student when she enrolled in one of his courses. She said students introduced themselves, one by one, each veering into heartfelt testimonies about what Dr. Farmer’s work had meant to them.
Dr. Farmer and Dr. Sabeti were just texting on Feb. 19, and the two were “goofing around in our usual way, and scheming about how to make the world better, as we always did.”
Dr. Farmer was funny, mischievous, and above all, exactly what you would expect upon meeting him, Dr. Sabeti said.
“It’s cliché, but the energetic kick you get from just being in his presence, it’s almost otherworldly,” she said. “It’s not even otherworldly in the sense of: ‘I just came across – greatness.’ It’s more: ‘I just came across kindness.’ ”
Dr. Farmer’s work has been widely distributed in publications including Bulletin of the World Health Organization, The Lancet, the New England Journal of Medicine, Clinical Infectious Diseases, and Social Science & Medicine.
He was awarded the 2020 Berggruen Prize for Philosophy & Culture, the Margaret Mead Award from the American Anthropological Association, the American Medical Association’s Outstanding International Physician (Nathan Davis) Award, and, with his Partners In Health colleagues, the Hilton Humanitarian Prize.
He is survived by his wife, Didi Bertrand Farmer, and their three children.
A verison of this article first appeared on Medscape.com.
Pediatrics group stresses benefits of vitamin K shots for infants
After the American Academy of Pediatrics (AAP) began recommending vitamin K shots for newborns in 1961, infant bleeding as a result of vitamin K deficiency plummeted. The life-threatening disorder is so rare that some parents now question the need for injections to safeguard against it.
The situation amounts to “a failure of our success,” Ivan Hand, MD, a coauthor of a new AAP statement on vitamin K, told this news organization. Much like diseases that can be prevented with vaccines, vitamin K deficiency bleeding isn’t top of mind for parents. “It’s not something they’re aware of or afraid of,” he said.
In 2019, however, the AAP listed public education about the importance of the shots in its 10 most important priorities.
The policy update urges clinicians to bone up on the benefits and perceived risks of vitamin K deficiency, which is essential for clotting, and to “strongly advocate” for the shot in discussions with parents who may get competing messages from their social circles, the internet, and other health care professionals.
Dr. Hand, director of neonatology at NYC Health + Hospitals Kings County, Brooklyn, said clinicians walk a line between educating and alienating parents who favor natural birth processes. “We’re hoping that by talking to the families and answering their questions and explaining the risks, parents will accept vitamin K as a necessary treatment for their babies,” he said.
Vitamin K does not easily pass through the placenta and is not plentiful in breast milk, the preferred nutrition source for newborns. It takes months for babies to build their stores through food and gut bacteria.
Infants who do not receive vitamin K at birth are 81 times more likely to develop late-onset vitamin K deficiency bleeding, which occurs a week to 6 months after birth, according to the Centers for Disease Control and Prevention. One in five babies with the disorder dies, and about half have bleeding in the skull that can lead to brain damage.
New dosing for premature infants
The AAP’s new statement, published in the journal Pediatrics, reaffirms the administration of a 1-mg intramuscular dose for infants weighing more than 1,500 grams, or about 3 lb 5 oz, within 6 hours of birth. For premature infants who weigh less, the guidance recommends an intramuscular dose of 0.3 to 0.5 mg/kg.
The group notes that oral preparations of vitamin K have proven less effective because of malabsorption and challenges with adhering to dosing regimens.
The document also warns that breastfed babies can experience vitamin K deficiency bleeding even if they have received the shot, because concentration of vitamin K often wanes before a baby starts eating solid food. The disorder “should be considered when evaluating bleeding in the first 6 months of life, even in infants who received prophylaxis, and especially in exclusively breastfed infants,” it states.
Accounts of parental refusals date back to 2013, when the CDC reported four cases of deficiency bleeding in Tennessee. The infants’ parents said they declined vitamin K because they worried about increased risk of leukemia, thought the injection was unnecessary, or wanted to minimize the baby’s exposure to “toxins.” Leukemia concern stemmed from a 1992 report linking vitamin K to childhood cancer, an association that did not hold up in subsequent studies.
More recent research has documented parental concerns about preservatives and injection pain as well as distrust of medical and public health authorities. Some parents have been accused of neglect for refusing to allow their babies to receive the shots.
Phoebe Danziger, MD, a pediatrician and writer in rural Michigan who has studied parental refusal of standard-of-care interventions, called the document a “welcome update” to the AAP’s last statement on the topic, in 2003. She told this news organization that lower dosing for premature infants may reassure some vitamin K–hesitant parents who worry about one-size-fits-all dosing.
But Dr. Danziger added that “evidence is lacking to support the claim that pediatricians can really move the needle on parental hesitancy and refusal simply through better listening and more persuasive counseling.” She said the AAP should do more to address “the broader social climate of mistrust and misinformation” that fuels refusal.
Dr. Hand and Dr. Danziger have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
After the American Academy of Pediatrics (AAP) began recommending vitamin K shots for newborns in 1961, infant bleeding as a result of vitamin K deficiency plummeted. The life-threatening disorder is so rare that some parents now question the need for injections to safeguard against it.
The situation amounts to “a failure of our success,” Ivan Hand, MD, a coauthor of a new AAP statement on vitamin K, told this news organization. Much like diseases that can be prevented with vaccines, vitamin K deficiency bleeding isn’t top of mind for parents. “It’s not something they’re aware of or afraid of,” he said.
In 2019, however, the AAP listed public education about the importance of the shots in its 10 most important priorities.
The policy update urges clinicians to bone up on the benefits and perceived risks of vitamin K deficiency, which is essential for clotting, and to “strongly advocate” for the shot in discussions with parents who may get competing messages from their social circles, the internet, and other health care professionals.
Dr. Hand, director of neonatology at NYC Health + Hospitals Kings County, Brooklyn, said clinicians walk a line between educating and alienating parents who favor natural birth processes. “We’re hoping that by talking to the families and answering their questions and explaining the risks, parents will accept vitamin K as a necessary treatment for their babies,” he said.
Vitamin K does not easily pass through the placenta and is not plentiful in breast milk, the preferred nutrition source for newborns. It takes months for babies to build their stores through food and gut bacteria.
Infants who do not receive vitamin K at birth are 81 times more likely to develop late-onset vitamin K deficiency bleeding, which occurs a week to 6 months after birth, according to the Centers for Disease Control and Prevention. One in five babies with the disorder dies, and about half have bleeding in the skull that can lead to brain damage.
New dosing for premature infants
The AAP’s new statement, published in the journal Pediatrics, reaffirms the administration of a 1-mg intramuscular dose for infants weighing more than 1,500 grams, or about 3 lb 5 oz, within 6 hours of birth. For premature infants who weigh less, the guidance recommends an intramuscular dose of 0.3 to 0.5 mg/kg.
The group notes that oral preparations of vitamin K have proven less effective because of malabsorption and challenges with adhering to dosing regimens.
The document also warns that breastfed babies can experience vitamin K deficiency bleeding even if they have received the shot, because concentration of vitamin K often wanes before a baby starts eating solid food. The disorder “should be considered when evaluating bleeding in the first 6 months of life, even in infants who received prophylaxis, and especially in exclusively breastfed infants,” it states.
Accounts of parental refusals date back to 2013, when the CDC reported four cases of deficiency bleeding in Tennessee. The infants’ parents said they declined vitamin K because they worried about increased risk of leukemia, thought the injection was unnecessary, or wanted to minimize the baby’s exposure to “toxins.” Leukemia concern stemmed from a 1992 report linking vitamin K to childhood cancer, an association that did not hold up in subsequent studies.
More recent research has documented parental concerns about preservatives and injection pain as well as distrust of medical and public health authorities. Some parents have been accused of neglect for refusing to allow their babies to receive the shots.
Phoebe Danziger, MD, a pediatrician and writer in rural Michigan who has studied parental refusal of standard-of-care interventions, called the document a “welcome update” to the AAP’s last statement on the topic, in 2003. She told this news organization that lower dosing for premature infants may reassure some vitamin K–hesitant parents who worry about one-size-fits-all dosing.
But Dr. Danziger added that “evidence is lacking to support the claim that pediatricians can really move the needle on parental hesitancy and refusal simply through better listening and more persuasive counseling.” She said the AAP should do more to address “the broader social climate of mistrust and misinformation” that fuels refusal.
Dr. Hand and Dr. Danziger have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
After the American Academy of Pediatrics (AAP) began recommending vitamin K shots for newborns in 1961, infant bleeding as a result of vitamin K deficiency plummeted. The life-threatening disorder is so rare that some parents now question the need for injections to safeguard against it.
The situation amounts to “a failure of our success,” Ivan Hand, MD, a coauthor of a new AAP statement on vitamin K, told this news organization. Much like diseases that can be prevented with vaccines, vitamin K deficiency bleeding isn’t top of mind for parents. “It’s not something they’re aware of or afraid of,” he said.
In 2019, however, the AAP listed public education about the importance of the shots in its 10 most important priorities.
The policy update urges clinicians to bone up on the benefits and perceived risks of vitamin K deficiency, which is essential for clotting, and to “strongly advocate” for the shot in discussions with parents who may get competing messages from their social circles, the internet, and other health care professionals.
Dr. Hand, director of neonatology at NYC Health + Hospitals Kings County, Brooklyn, said clinicians walk a line between educating and alienating parents who favor natural birth processes. “We’re hoping that by talking to the families and answering their questions and explaining the risks, parents will accept vitamin K as a necessary treatment for their babies,” he said.
Vitamin K does not easily pass through the placenta and is not plentiful in breast milk, the preferred nutrition source for newborns. It takes months for babies to build their stores through food and gut bacteria.
Infants who do not receive vitamin K at birth are 81 times more likely to develop late-onset vitamin K deficiency bleeding, which occurs a week to 6 months after birth, according to the Centers for Disease Control and Prevention. One in five babies with the disorder dies, and about half have bleeding in the skull that can lead to brain damage.
New dosing for premature infants
The AAP’s new statement, published in the journal Pediatrics, reaffirms the administration of a 1-mg intramuscular dose for infants weighing more than 1,500 grams, or about 3 lb 5 oz, within 6 hours of birth. For premature infants who weigh less, the guidance recommends an intramuscular dose of 0.3 to 0.5 mg/kg.
The group notes that oral preparations of vitamin K have proven less effective because of malabsorption and challenges with adhering to dosing regimens.
The document also warns that breastfed babies can experience vitamin K deficiency bleeding even if they have received the shot, because concentration of vitamin K often wanes before a baby starts eating solid food. The disorder “should be considered when evaluating bleeding in the first 6 months of life, even in infants who received prophylaxis, and especially in exclusively breastfed infants,” it states.
Accounts of parental refusals date back to 2013, when the CDC reported four cases of deficiency bleeding in Tennessee. The infants’ parents said they declined vitamin K because they worried about increased risk of leukemia, thought the injection was unnecessary, or wanted to minimize the baby’s exposure to “toxins.” Leukemia concern stemmed from a 1992 report linking vitamin K to childhood cancer, an association that did not hold up in subsequent studies.
More recent research has documented parental concerns about preservatives and injection pain as well as distrust of medical and public health authorities. Some parents have been accused of neglect for refusing to allow their babies to receive the shots.
Phoebe Danziger, MD, a pediatrician and writer in rural Michigan who has studied parental refusal of standard-of-care interventions, called the document a “welcome update” to the AAP’s last statement on the topic, in 2003. She told this news organization that lower dosing for premature infants may reassure some vitamin K–hesitant parents who worry about one-size-fits-all dosing.
But Dr. Danziger added that “evidence is lacking to support the claim that pediatricians can really move the needle on parental hesitancy and refusal simply through better listening and more persuasive counseling.” She said the AAP should do more to address “the broader social climate of mistrust and misinformation” that fuels refusal.
Dr. Hand and Dr. Danziger have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Researchers tout new CLL prognostic tool
Researchers report that they’ve confirmed the usefulness of a new tool to help physicians pinpoint prognoses for patients with chronic lymphocytic leukemia (CLL).
“Physicians may use this tool to support decisions regarding supportive care, manage the patient’s and physician’s expectations, and potentially tailor therapy,” study lead author and epidemiologist Emelie Rotbain, MD, PhD, of Rigshospitalet in Copenhagen, said in an interview.
The study appeared Jan. 10 in the journal Blood Advances.
According to Dr. Rotbain,
Researchers developed the questions based on an analysis of categories in the Cumulative Illness Rating Scale that are most linked to event-free survival (EFS) from time of treatment.
The tool looks at three organ systems – vascular, upper GI, and endocrine – and asks about conditions such as diabetes and chronic use of a proton pump inhibitor, study coauthor and hematologist/oncologist Alexey V. Danilov, MD, PhD, codirector of the Toni Stephenson Lymphoma Center at the City of Hope National Medical Center, Duarte, Calif., said in an interview. The tool then generates a score based on the variables.
For the new study, the researchers retrospectively applied the tool to 4,975 patients who appeared in the Danish National CLL Register from 2008 to 2018 (61% male, median age 70.7.). Of those, 1,513 received first-line treatment during follow-up (median = 4.39 years).
At diagnosis, nearly two-thirds (63%) of patients were considered to be low risk. None of these had endocrinological, upper gastrointestinal, or vascular disease. Another 30% were considered to be at intermediate risk. The remaining 7% were at high risk. They had high levels of endocrinological (55.6%), upper gastrointestinal (64.6%), and vascular disease (91.0%).
The high-risk patients had a median survival of 6.0 years. The intermediate-risk patients lived for a median of 8.5 years, while the low-risk patients didn’t reach a median survival level.
Fifty-six percent of high-risk patients were treated within 4 years, compared to 20%-30% of intermediate- and low-risk patients. Median event-free survival from time of treatment was 8.4, 4.4, and 2.2 years for the low-, intermediate-, and high-risk groups, respectively.
The authors cautioned that “differences in survival by type of treatment, particularly in patients treated with targeted therapies who were underrepresented in this study, could influence survival and limit the generalizability of these results.”
They added that “while prognostic factors should remain key for treatment decisions, clinical trial data from pivotal phase 3 trials with novel targeted agents versus chemoimmunotherapy should be reanalyzed with addition of CLL-CI to assess the optimal treatment for patients according to CLL-CI.”
The tool is not yet available online, Dr. Danilov said, “but that is something that we as a group could potentially work on.”
Joanna Rhodes, MD, assistant professor with Northwell Health Cancer Institute/Donald and Barbara Zucker School of Medicine at Hofstra/Northwell, Hempstead, N.Y., said in an interview that the tool is easy to use and appropriate to apply at first consultation. It should be used in conjunction with the International Prognostic Index for Chronic Lymphocytic Leukemia (CLL-IPI), she said.
“We often discuss frailty as a factor in types of and timing of treatment for patients with CLL, but often this is not directly measured in clinical practice,” she said. “The CLL-CI is associated with important outcomes, particularly overall survival, which is our most important metric in oncology. Additionally, it provides important information on time to first treatment and overall survival, which are useful when we are counseling patients.”
Like the study authors, Dr. Rhodes cautioned that the CLL-CI has not been validated specifically in patients treated with targeted therapies. “It may not be applicable in this setting, particularly in the front-line setting, as these treatments were underrepresented in this cohort. Further studies in this population are needed to answer this question.”
The study is funded in part by Novo Nordisk Foundation. Several study authors report various disclosures outside the scope of this study. Dr. Rhodes has no disclosures.
Researchers report that they’ve confirmed the usefulness of a new tool to help physicians pinpoint prognoses for patients with chronic lymphocytic leukemia (CLL).
“Physicians may use this tool to support decisions regarding supportive care, manage the patient’s and physician’s expectations, and potentially tailor therapy,” study lead author and epidemiologist Emelie Rotbain, MD, PhD, of Rigshospitalet in Copenhagen, said in an interview.
The study appeared Jan. 10 in the journal Blood Advances.
According to Dr. Rotbain,
Researchers developed the questions based on an analysis of categories in the Cumulative Illness Rating Scale that are most linked to event-free survival (EFS) from time of treatment.
The tool looks at three organ systems – vascular, upper GI, and endocrine – and asks about conditions such as diabetes and chronic use of a proton pump inhibitor, study coauthor and hematologist/oncologist Alexey V. Danilov, MD, PhD, codirector of the Toni Stephenson Lymphoma Center at the City of Hope National Medical Center, Duarte, Calif., said in an interview. The tool then generates a score based on the variables.
For the new study, the researchers retrospectively applied the tool to 4,975 patients who appeared in the Danish National CLL Register from 2008 to 2018 (61% male, median age 70.7.). Of those, 1,513 received first-line treatment during follow-up (median = 4.39 years).
At diagnosis, nearly two-thirds (63%) of patients were considered to be low risk. None of these had endocrinological, upper gastrointestinal, or vascular disease. Another 30% were considered to be at intermediate risk. The remaining 7% were at high risk. They had high levels of endocrinological (55.6%), upper gastrointestinal (64.6%), and vascular disease (91.0%).
The high-risk patients had a median survival of 6.0 years. The intermediate-risk patients lived for a median of 8.5 years, while the low-risk patients didn’t reach a median survival level.
Fifty-six percent of high-risk patients were treated within 4 years, compared to 20%-30% of intermediate- and low-risk patients. Median event-free survival from time of treatment was 8.4, 4.4, and 2.2 years for the low-, intermediate-, and high-risk groups, respectively.
The authors cautioned that “differences in survival by type of treatment, particularly in patients treated with targeted therapies who were underrepresented in this study, could influence survival and limit the generalizability of these results.”
They added that “while prognostic factors should remain key for treatment decisions, clinical trial data from pivotal phase 3 trials with novel targeted agents versus chemoimmunotherapy should be reanalyzed with addition of CLL-CI to assess the optimal treatment for patients according to CLL-CI.”
The tool is not yet available online, Dr. Danilov said, “but that is something that we as a group could potentially work on.”
Joanna Rhodes, MD, assistant professor with Northwell Health Cancer Institute/Donald and Barbara Zucker School of Medicine at Hofstra/Northwell, Hempstead, N.Y., said in an interview that the tool is easy to use and appropriate to apply at first consultation. It should be used in conjunction with the International Prognostic Index for Chronic Lymphocytic Leukemia (CLL-IPI), she said.
“We often discuss frailty as a factor in types of and timing of treatment for patients with CLL, but often this is not directly measured in clinical practice,” she said. “The CLL-CI is associated with important outcomes, particularly overall survival, which is our most important metric in oncology. Additionally, it provides important information on time to first treatment and overall survival, which are useful when we are counseling patients.”
Like the study authors, Dr. Rhodes cautioned that the CLL-CI has not been validated specifically in patients treated with targeted therapies. “It may not be applicable in this setting, particularly in the front-line setting, as these treatments were underrepresented in this cohort. Further studies in this population are needed to answer this question.”
The study is funded in part by Novo Nordisk Foundation. Several study authors report various disclosures outside the scope of this study. Dr. Rhodes has no disclosures.
Researchers report that they’ve confirmed the usefulness of a new tool to help physicians pinpoint prognoses for patients with chronic lymphocytic leukemia (CLL).
“Physicians may use this tool to support decisions regarding supportive care, manage the patient’s and physician’s expectations, and potentially tailor therapy,” study lead author and epidemiologist Emelie Rotbain, MD, PhD, of Rigshospitalet in Copenhagen, said in an interview.
The study appeared Jan. 10 in the journal Blood Advances.
According to Dr. Rotbain,
Researchers developed the questions based on an analysis of categories in the Cumulative Illness Rating Scale that are most linked to event-free survival (EFS) from time of treatment.
The tool looks at three organ systems – vascular, upper GI, and endocrine – and asks about conditions such as diabetes and chronic use of a proton pump inhibitor, study coauthor and hematologist/oncologist Alexey V. Danilov, MD, PhD, codirector of the Toni Stephenson Lymphoma Center at the City of Hope National Medical Center, Duarte, Calif., said in an interview. The tool then generates a score based on the variables.
For the new study, the researchers retrospectively applied the tool to 4,975 patients who appeared in the Danish National CLL Register from 2008 to 2018 (61% male, median age 70.7.). Of those, 1,513 received first-line treatment during follow-up (median = 4.39 years).
At diagnosis, nearly two-thirds (63%) of patients were considered to be low risk. None of these had endocrinological, upper gastrointestinal, or vascular disease. Another 30% were considered to be at intermediate risk. The remaining 7% were at high risk. They had high levels of endocrinological (55.6%), upper gastrointestinal (64.6%), and vascular disease (91.0%).
The high-risk patients had a median survival of 6.0 years. The intermediate-risk patients lived for a median of 8.5 years, while the low-risk patients didn’t reach a median survival level.
Fifty-six percent of high-risk patients were treated within 4 years, compared to 20%-30% of intermediate- and low-risk patients. Median event-free survival from time of treatment was 8.4, 4.4, and 2.2 years for the low-, intermediate-, and high-risk groups, respectively.
The authors cautioned that “differences in survival by type of treatment, particularly in patients treated with targeted therapies who were underrepresented in this study, could influence survival and limit the generalizability of these results.”
They added that “while prognostic factors should remain key for treatment decisions, clinical trial data from pivotal phase 3 trials with novel targeted agents versus chemoimmunotherapy should be reanalyzed with addition of CLL-CI to assess the optimal treatment for patients according to CLL-CI.”
The tool is not yet available online, Dr. Danilov said, “but that is something that we as a group could potentially work on.”
Joanna Rhodes, MD, assistant professor with Northwell Health Cancer Institute/Donald and Barbara Zucker School of Medicine at Hofstra/Northwell, Hempstead, N.Y., said in an interview that the tool is easy to use and appropriate to apply at first consultation. It should be used in conjunction with the International Prognostic Index for Chronic Lymphocytic Leukemia (CLL-IPI), she said.
“We often discuss frailty as a factor in types of and timing of treatment for patients with CLL, but often this is not directly measured in clinical practice,” she said. “The CLL-CI is associated with important outcomes, particularly overall survival, which is our most important metric in oncology. Additionally, it provides important information on time to first treatment and overall survival, which are useful when we are counseling patients.”
Like the study authors, Dr. Rhodes cautioned that the CLL-CI has not been validated specifically in patients treated with targeted therapies. “It may not be applicable in this setting, particularly in the front-line setting, as these treatments were underrepresented in this cohort. Further studies in this population are needed to answer this question.”
The study is funded in part by Novo Nordisk Foundation. Several study authors report various disclosures outside the scope of this study. Dr. Rhodes has no disclosures.
FROM BLOOD ADVANCES
Children and COVID: The Omicron surge has become a retreat
The Omicron decline continued for a fourth consecutive week as new cases of COVID-19 in children fell by 42% from the week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
That 42% represents a drop from the 299,000 new cases reported for Feb. 4-10 down to 174,000 for the most recent week, Feb. 11-17. 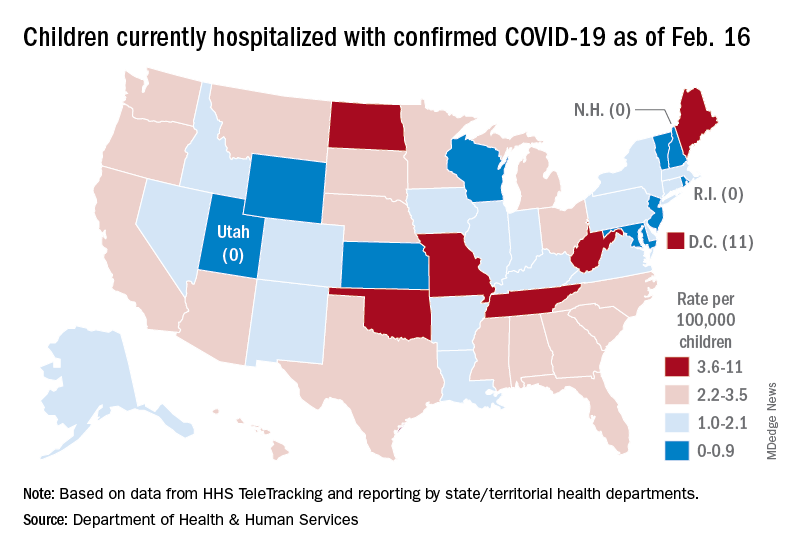
The overall count of COVID-19 cases in children is 12.5 million over the course of the pandemic, and that represents 19% of cases reported among all ages, the AAP and CHA said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Hospital admissions also continued to fall, with the rate for children aged 0-17 at 0.43 per 100,000 population as of Feb. 20, down by almost 66% from the peak of 1.25 per 100,000 reached on Jan. 16, the Centers for Disease Control and Prevention reported.
A snapshot of the hospitalization situation shows that 1,687 children were occupying inpatient beds on Feb. 16, compared with 4,070 on Jan. 19, which appears to be the peak of the Omicron surge, according to data from the Department of Health & Human Services.
The state with the highest rate – 5.6 per 100,000 children – on Feb. 16 was North Dakota, although the District of Columbia came in at 11.0 per 100,000. They were followed by Oklahoma (5.3), Missouri (5.2), and West Virginia (4.1). There were three states – New Hampshire, Rhode Island, and Utah – with no children in the hospital on that date, the HHS said.
New vaccinations in children aged 5-11 years, which declined in mid- and late January, even as Omicron surged, continued to decline, as did vaccine completions. Vaccinations also fell among children aged 12-17 for the latest reporting week, Feb. 10-16, the AAP said in a separate report.
As more states and school districts drop mask mandates, data from the CDC indicate that 32.5% of 5- to 11-year olds and 67.4% of 12- to 17-year-olds have gotten at least one dose of the COVID-19 vaccine and that 25.1% and 57.3%, respectively, are fully vaccinated. Meanwhile, 20.5% of those fully vaccinated 12- to 17-year-olds have gotten a booster dose, the CDC said.
The Omicron decline continued for a fourth consecutive week as new cases of COVID-19 in children fell by 42% from the week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
That 42% represents a drop from the 299,000 new cases reported for Feb. 4-10 down to 174,000 for the most recent week, Feb. 11-17. 
The overall count of COVID-19 cases in children is 12.5 million over the course of the pandemic, and that represents 19% of cases reported among all ages, the AAP and CHA said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Hospital admissions also continued to fall, with the rate for children aged 0-17 at 0.43 per 100,000 population as of Feb. 20, down by almost 66% from the peak of 1.25 per 100,000 reached on Jan. 16, the Centers for Disease Control and Prevention reported.
A snapshot of the hospitalization situation shows that 1,687 children were occupying inpatient beds on Feb. 16, compared with 4,070 on Jan. 19, which appears to be the peak of the Omicron surge, according to data from the Department of Health & Human Services.
The state with the highest rate – 5.6 per 100,000 children – on Feb. 16 was North Dakota, although the District of Columbia came in at 11.0 per 100,000. They were followed by Oklahoma (5.3), Missouri (5.2), and West Virginia (4.1). There were three states – New Hampshire, Rhode Island, and Utah – with no children in the hospital on that date, the HHS said.
New vaccinations in children aged 5-11 years, which declined in mid- and late January, even as Omicron surged, continued to decline, as did vaccine completions. Vaccinations also fell among children aged 12-17 for the latest reporting week, Feb. 10-16, the AAP said in a separate report.
As more states and school districts drop mask mandates, data from the CDC indicate that 32.5% of 5- to 11-year olds and 67.4% of 12- to 17-year-olds have gotten at least one dose of the COVID-19 vaccine and that 25.1% and 57.3%, respectively, are fully vaccinated. Meanwhile, 20.5% of those fully vaccinated 12- to 17-year-olds have gotten a booster dose, the CDC said.
The Omicron decline continued for a fourth consecutive week as new cases of COVID-19 in children fell by 42% from the week before, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
That 42% represents a drop from the 299,000 new cases reported for Feb. 4-10 down to 174,000 for the most recent week, Feb. 11-17. 
The overall count of COVID-19 cases in children is 12.5 million over the course of the pandemic, and that represents 19% of cases reported among all ages, the AAP and CHA said based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Hospital admissions also continued to fall, with the rate for children aged 0-17 at 0.43 per 100,000 population as of Feb. 20, down by almost 66% from the peak of 1.25 per 100,000 reached on Jan. 16, the Centers for Disease Control and Prevention reported.
A snapshot of the hospitalization situation shows that 1,687 children were occupying inpatient beds on Feb. 16, compared with 4,070 on Jan. 19, which appears to be the peak of the Omicron surge, according to data from the Department of Health & Human Services.
The state with the highest rate – 5.6 per 100,000 children – on Feb. 16 was North Dakota, although the District of Columbia came in at 11.0 per 100,000. They were followed by Oklahoma (5.3), Missouri (5.2), and West Virginia (4.1). There were three states – New Hampshire, Rhode Island, and Utah – with no children in the hospital on that date, the HHS said.
New vaccinations in children aged 5-11 years, which declined in mid- and late January, even as Omicron surged, continued to decline, as did vaccine completions. Vaccinations also fell among children aged 12-17 for the latest reporting week, Feb. 10-16, the AAP said in a separate report.
As more states and school districts drop mask mandates, data from the CDC indicate that 32.5% of 5- to 11-year olds and 67.4% of 12- to 17-year-olds have gotten at least one dose of the COVID-19 vaccine and that 25.1% and 57.3%, respectively, are fully vaccinated. Meanwhile, 20.5% of those fully vaccinated 12- to 17-year-olds have gotten a booster dose, the CDC said.
Women with von Willebrand disease: Managing menstrual and postpartum bleeding
Women with von Willebrand disease (VWD) experience many obstetric and gynecologic challenges, including higher levels of von Willebrand factor (VWF) in pregnancy, Romina Brignardello-Petersen, PhD, of McMaster University, Hamilton, Ont., and colleagues wrote.
The American Society of Hematology, the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation, and the World Federation of Hemophilia convened a working group in 2017 to address updated guidelines on VWD with a focus on women, the researchers said.
In an article published in Blood Advances, the researchers described the evidence from three systematic reviews conducted to inform three recommendations for the guidelines: first-line management of heavy menstrual bleeding (HMB), treatment of women requiring or desiring neuraxial analgesia, and management of postpartum hemorrhage. The authors identified studies published through October 2019.
The first systematic review of first-line therapies for HMB included five case series, one retrospective cohort study, and one randomized controlled trial. In the randomized controlled trial of 232 patients, low-certainty evidence suggested less reduction of blood loss with desmopressin, compared with tranexamic acid (TxA), with no significant differences in side effects. Very-low-certainty evidence from an observational study also supported lower effectiveness of desmopressin versus hormonal therapy. Finally, the case series showed very-low-certainty evidence for the comparative effectiveness of hormonal therapy delivered via a levonorgestrel-releasing intrauterine system (LNG-IUS) and other therapies for HMB control.
The second systematic review compared VWF levels in women who received neuraxial anesthesia during labor.
The review included five case series that described outcomes of women with VWF levels greater than 0.50 IU/mL; however, the studies did not describe outcomes according to VWF levels and did not cite the proportion of women with VWF levels greater than 1.50 IU/mL. Consequently, the evidence for the effects of increasing VWF levels was very low certainty, the authors said. In a meta-analysis, the proportion of anesthesia complications in these women was 6% (very low certainty). The complications included hypotension, accidental dural puncture, inadequate analgesia, bloody tap with no further complications, and failed block requiring general anesthesia.
The third systemic review included two retrospective cohort studies on the use of TxA during the postpartum period. In these studies, the authors found very-low-certainty evidence that TxA reduced the risk of severe primary postpartum hemorrhage, primary postpartum hemorrhage, and secondary postpartum hemorrhage (risk ratios, 0.36, 0.25, and 0.42, respectively). The effects of TxA on blood transfusions, vaginal hematoma, blood loss, and thrombotic complications also showed very-low-certainty evidence.
The currently available evidence for treatment options in women with VWD remains very low certainty, the researchers wrote in their discussion. “Because hormonal therapy is effective in controlling HMB (based on data from women without bleeding disorders), we believe the most effective strategy to be hormonal therapy with a LNG-IUS or combined oral contraceptives, followed by TxA and desmopressin.”
The study findings were limited by several factors including scarce evidence, the risk of bias in the observational studies, and lack of comparisons/controls in the case series, the researchers noted. Notable literature gaps included data on outcomes including major bleeding and the need for surgery or additional treatments in the first review; mortality, major bleeding, spinal hematoma, transfusion, and thrombotic events in the second review; and mortality, major bleeding, and the need for other procedures in the third review.
However, the findings were strengthened by the use of broad eligibility criteria to include any studies with potential useful advice, including case series, if these were the only available options. In developing recommendations, “the guideline panel interpreted the evidence adding their experience and knowledge of indirect evidence,” the authors noted.
The current evidence, though mainly very low certainty, “is the best available to inform decisions about management. Clinicians seeking advice on how to manage their patients with VWD should refer to the practice guidelines and assess to what extent they are applicable to their patients,” the researchers concluded.
Meeting the need for evidence-based guidelines
The review is important at this time because current evidence-based guidelines are limited, said coauthor Veronica Flood, MD, a pediatric hematologist at the Medical College of Wisconsin, Milwaukee, and a VWD researcher.
“While we have some guidelines that address von Willebrand disease, these were primarily based on expert opinion and not necessarily based on the best available evidence,” said Dr. Flood.
“Given how many people have von Willebrand disease, it is important that we actually base our recommendations on the data,” she emphasized. The new guidelines also incorporate patient feedback, with the inclusion of multiple panelists who are individuals living with VWD. “The final recommendations looked at not only the evidence, but the cost effectiveness, feasibility, and patient values and preferences,” she added.
“I was surprised we did not have better evidence for some of these common issues for patients with VWD,” said Dr. Flood. “I think that speaks to the need to do more high-quality research in this area.”
From a clinical standpoint, “we now have evidence-based guidelines that support the use of prophylaxis in patients with VWD and significant bleeding, as well as recommendations for surgery and bleeding issues around menstruation,” said Dr. Flood. “I do think it is also important to recognize that many of these are conditional recommendations, meaning there is room for patient preferences in implementation, which is helpful since we know that some people will have different priorities.”
Dr. Flood noted that more research is needed in many aspects of VWD. “We definitely need to better understand best options for surgical treatment, and I consider that a high priority. We are also hoping, along with the National Hemophilia Foundation, to develop some patient decision aids to help with some of these issues.”
Coauthor Nathan T. Connell, MD, an adult hematologist at the Brigham and Women’s Hospital and Harvard Medical School, both in Boston, served as the vice chair for the guideline panel. Dr. Connell agreed with the importance of the reviews and the need for additional research. “I, too, was surprised to see the lack of robust data to answer many of the basic questions about how to manage people living with VWD. Regarding the systematic reviews, I was surprised to see the power of combining the limited data in this way to come up with an evidence base for the panels to review,” he added.
The study was supported by the ASH, ISTH, NHF, and the WFH 2020 Guidelines for Management of VWD. The researchers had no financial conflicts to disclose.
Women with von Willebrand disease (VWD) experience many obstetric and gynecologic challenges, including higher levels of von Willebrand factor (VWF) in pregnancy, Romina Brignardello-Petersen, PhD, of McMaster University, Hamilton, Ont., and colleagues wrote.
The American Society of Hematology, the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation, and the World Federation of Hemophilia convened a working group in 2017 to address updated guidelines on VWD with a focus on women, the researchers said.
In an article published in Blood Advances, the researchers described the evidence from three systematic reviews conducted to inform three recommendations for the guidelines: first-line management of heavy menstrual bleeding (HMB), treatment of women requiring or desiring neuraxial analgesia, and management of postpartum hemorrhage. The authors identified studies published through October 2019.
The first systematic review of first-line therapies for HMB included five case series, one retrospective cohort study, and one randomized controlled trial. In the randomized controlled trial of 232 patients, low-certainty evidence suggested less reduction of blood loss with desmopressin, compared with tranexamic acid (TxA), with no significant differences in side effects. Very-low-certainty evidence from an observational study also supported lower effectiveness of desmopressin versus hormonal therapy. Finally, the case series showed very-low-certainty evidence for the comparative effectiveness of hormonal therapy delivered via a levonorgestrel-releasing intrauterine system (LNG-IUS) and other therapies for HMB control.
The second systematic review compared VWF levels in women who received neuraxial anesthesia during labor.
The review included five case series that described outcomes of women with VWF levels greater than 0.50 IU/mL; however, the studies did not describe outcomes according to VWF levels and did not cite the proportion of women with VWF levels greater than 1.50 IU/mL. Consequently, the evidence for the effects of increasing VWF levels was very low certainty, the authors said. In a meta-analysis, the proportion of anesthesia complications in these women was 6% (very low certainty). The complications included hypotension, accidental dural puncture, inadequate analgesia, bloody tap with no further complications, and failed block requiring general anesthesia.
The third systemic review included two retrospective cohort studies on the use of TxA during the postpartum period. In these studies, the authors found very-low-certainty evidence that TxA reduced the risk of severe primary postpartum hemorrhage, primary postpartum hemorrhage, and secondary postpartum hemorrhage (risk ratios, 0.36, 0.25, and 0.42, respectively). The effects of TxA on blood transfusions, vaginal hematoma, blood loss, and thrombotic complications also showed very-low-certainty evidence.
The currently available evidence for treatment options in women with VWD remains very low certainty, the researchers wrote in their discussion. “Because hormonal therapy is effective in controlling HMB (based on data from women without bleeding disorders), we believe the most effective strategy to be hormonal therapy with a LNG-IUS or combined oral contraceptives, followed by TxA and desmopressin.”
The study findings were limited by several factors including scarce evidence, the risk of bias in the observational studies, and lack of comparisons/controls in the case series, the researchers noted. Notable literature gaps included data on outcomes including major bleeding and the need for surgery or additional treatments in the first review; mortality, major bleeding, spinal hematoma, transfusion, and thrombotic events in the second review; and mortality, major bleeding, and the need for other procedures in the third review.
However, the findings were strengthened by the use of broad eligibility criteria to include any studies with potential useful advice, including case series, if these were the only available options. In developing recommendations, “the guideline panel interpreted the evidence adding their experience and knowledge of indirect evidence,” the authors noted.
The current evidence, though mainly very low certainty, “is the best available to inform decisions about management. Clinicians seeking advice on how to manage their patients with VWD should refer to the practice guidelines and assess to what extent they are applicable to their patients,” the researchers concluded.
Meeting the need for evidence-based guidelines
The review is important at this time because current evidence-based guidelines are limited, said coauthor Veronica Flood, MD, a pediatric hematologist at the Medical College of Wisconsin, Milwaukee, and a VWD researcher.
“While we have some guidelines that address von Willebrand disease, these were primarily based on expert opinion and not necessarily based on the best available evidence,” said Dr. Flood.
“Given how many people have von Willebrand disease, it is important that we actually base our recommendations on the data,” she emphasized. The new guidelines also incorporate patient feedback, with the inclusion of multiple panelists who are individuals living with VWD. “The final recommendations looked at not only the evidence, but the cost effectiveness, feasibility, and patient values and preferences,” she added.
“I was surprised we did not have better evidence for some of these common issues for patients with VWD,” said Dr. Flood. “I think that speaks to the need to do more high-quality research in this area.”
From a clinical standpoint, “we now have evidence-based guidelines that support the use of prophylaxis in patients with VWD and significant bleeding, as well as recommendations for surgery and bleeding issues around menstruation,” said Dr. Flood. “I do think it is also important to recognize that many of these are conditional recommendations, meaning there is room for patient preferences in implementation, which is helpful since we know that some people will have different priorities.”
Dr. Flood noted that more research is needed in many aspects of VWD. “We definitely need to better understand best options for surgical treatment, and I consider that a high priority. We are also hoping, along with the National Hemophilia Foundation, to develop some patient decision aids to help with some of these issues.”
Coauthor Nathan T. Connell, MD, an adult hematologist at the Brigham and Women’s Hospital and Harvard Medical School, both in Boston, served as the vice chair for the guideline panel. Dr. Connell agreed with the importance of the reviews and the need for additional research. “I, too, was surprised to see the lack of robust data to answer many of the basic questions about how to manage people living with VWD. Regarding the systematic reviews, I was surprised to see the power of combining the limited data in this way to come up with an evidence base for the panels to review,” he added.
The study was supported by the ASH, ISTH, NHF, and the WFH 2020 Guidelines for Management of VWD. The researchers had no financial conflicts to disclose.
Women with von Willebrand disease (VWD) experience many obstetric and gynecologic challenges, including higher levels of von Willebrand factor (VWF) in pregnancy, Romina Brignardello-Petersen, PhD, of McMaster University, Hamilton, Ont., and colleagues wrote.
The American Society of Hematology, the International Society on Thrombosis and Haemostasis, the National Hemophilia Foundation, and the World Federation of Hemophilia convened a working group in 2017 to address updated guidelines on VWD with a focus on women, the researchers said.
In an article published in Blood Advances, the researchers described the evidence from three systematic reviews conducted to inform three recommendations for the guidelines: first-line management of heavy menstrual bleeding (HMB), treatment of women requiring or desiring neuraxial analgesia, and management of postpartum hemorrhage. The authors identified studies published through October 2019.
The first systematic review of first-line therapies for HMB included five case series, one retrospective cohort study, and one randomized controlled trial. In the randomized controlled trial of 232 patients, low-certainty evidence suggested less reduction of blood loss with desmopressin, compared with tranexamic acid (TxA), with no significant differences in side effects. Very-low-certainty evidence from an observational study also supported lower effectiveness of desmopressin versus hormonal therapy. Finally, the case series showed very-low-certainty evidence for the comparative effectiveness of hormonal therapy delivered via a levonorgestrel-releasing intrauterine system (LNG-IUS) and other therapies for HMB control.
The second systematic review compared VWF levels in women who received neuraxial anesthesia during labor.
The review included five case series that described outcomes of women with VWF levels greater than 0.50 IU/mL; however, the studies did not describe outcomes according to VWF levels and did not cite the proportion of women with VWF levels greater than 1.50 IU/mL. Consequently, the evidence for the effects of increasing VWF levels was very low certainty, the authors said. In a meta-analysis, the proportion of anesthesia complications in these women was 6% (very low certainty). The complications included hypotension, accidental dural puncture, inadequate analgesia, bloody tap with no further complications, and failed block requiring general anesthesia.
The third systemic review included two retrospective cohort studies on the use of TxA during the postpartum period. In these studies, the authors found very-low-certainty evidence that TxA reduced the risk of severe primary postpartum hemorrhage, primary postpartum hemorrhage, and secondary postpartum hemorrhage (risk ratios, 0.36, 0.25, and 0.42, respectively). The effects of TxA on blood transfusions, vaginal hematoma, blood loss, and thrombotic complications also showed very-low-certainty evidence.
The currently available evidence for treatment options in women with VWD remains very low certainty, the researchers wrote in their discussion. “Because hormonal therapy is effective in controlling HMB (based on data from women without bleeding disorders), we believe the most effective strategy to be hormonal therapy with a LNG-IUS or combined oral contraceptives, followed by TxA and desmopressin.”
The study findings were limited by several factors including scarce evidence, the risk of bias in the observational studies, and lack of comparisons/controls in the case series, the researchers noted. Notable literature gaps included data on outcomes including major bleeding and the need for surgery or additional treatments in the first review; mortality, major bleeding, spinal hematoma, transfusion, and thrombotic events in the second review; and mortality, major bleeding, and the need for other procedures in the third review.
However, the findings were strengthened by the use of broad eligibility criteria to include any studies with potential useful advice, including case series, if these were the only available options. In developing recommendations, “the guideline panel interpreted the evidence adding their experience and knowledge of indirect evidence,” the authors noted.
The current evidence, though mainly very low certainty, “is the best available to inform decisions about management. Clinicians seeking advice on how to manage their patients with VWD should refer to the practice guidelines and assess to what extent they are applicable to their patients,” the researchers concluded.
Meeting the need for evidence-based guidelines
The review is important at this time because current evidence-based guidelines are limited, said coauthor Veronica Flood, MD, a pediatric hematologist at the Medical College of Wisconsin, Milwaukee, and a VWD researcher.
“While we have some guidelines that address von Willebrand disease, these were primarily based on expert opinion and not necessarily based on the best available evidence,” said Dr. Flood.
“Given how many people have von Willebrand disease, it is important that we actually base our recommendations on the data,” she emphasized. The new guidelines also incorporate patient feedback, with the inclusion of multiple panelists who are individuals living with VWD. “The final recommendations looked at not only the evidence, but the cost effectiveness, feasibility, and patient values and preferences,” she added.
“I was surprised we did not have better evidence for some of these common issues for patients with VWD,” said Dr. Flood. “I think that speaks to the need to do more high-quality research in this area.”
From a clinical standpoint, “we now have evidence-based guidelines that support the use of prophylaxis in patients with VWD and significant bleeding, as well as recommendations for surgery and bleeding issues around menstruation,” said Dr. Flood. “I do think it is also important to recognize that many of these are conditional recommendations, meaning there is room for patient preferences in implementation, which is helpful since we know that some people will have different priorities.”
Dr. Flood noted that more research is needed in many aspects of VWD. “We definitely need to better understand best options for surgical treatment, and I consider that a high priority. We are also hoping, along with the National Hemophilia Foundation, to develop some patient decision aids to help with some of these issues.”
Coauthor Nathan T. Connell, MD, an adult hematologist at the Brigham and Women’s Hospital and Harvard Medical School, both in Boston, served as the vice chair for the guideline panel. Dr. Connell agreed with the importance of the reviews and the need for additional research. “I, too, was surprised to see the lack of robust data to answer many of the basic questions about how to manage people living with VWD. Regarding the systematic reviews, I was surprised to see the power of combining the limited data in this way to come up with an evidence base for the panels to review,” he added.
The study was supported by the ASH, ISTH, NHF, and the WFH 2020 Guidelines for Management of VWD. The researchers had no financial conflicts to disclose.
FROM BLOOD ADVANCES