User login
It’s Never Too Late to Convince Patients to Quit Smoking
An estimated 450,000 US deaths are expected this year from conditions attributed to cigarette smoking. Although the percentage of adults who smoke declined from 21% in 2005 to 11% in 2022, the annual death toll has been stable since 2005 and isn’t expected to decline until 2030, owing to an aging population of current and former smokers.
In 2022, based on a national survey, two thirds of the 28.8 million US adult smokers wanted to quit, and more than half tried quitting on their own or with the help of clinicians, but less than 9% succeeded in kicking the habit. The health benefits of quitting, summarized in a patient education handout from the American Cancer Society, include a lower risk for cancer, diabetes, and cardiovascular disease. Furthermore, the handout states, “quitting smoking can add as much as 10 years to your life, compared to if you continued to smoke.”
For my patients older than age 50 who are lifelong smokers, the qualifier “as much as” can be a sticking point. Although most recognize that continuing to smoke exposes them to greater health risks and are willing to undergo lung cancer screening and receive pneumococcal vaccines, a kind of fatalism frequently sets in. I’ve heard more times than I can recall some version of the declaration, “It’s too late for quitting to make much difference for me.” Many smokers think that once they reach middle age, gains in life expectancy will be too small to be worth the intense effort and multiple failed attempts that are typically required to quit permanently. Until recently, there were few data I could call on to persuade them they were wrong.
In February 2024, Dr. Eo Rin Cho and colleagues pooled data from four national cohort studies (United States, United Kingdom, Norway, and Canada) to calculate mortality differences among current, former, and never smokers aged 20-79 years. Compared with never smokers, lifelong smokers died an average of 12-13 years earlier. However, quitting before age 50 nearly eliminated the excess mortality associated with smoking, and in the 50- to 59-year-old age group, cessation eventually reduced excess mortality by 92%-95%. Better yet, more than half of the benefits occurred within the first 3 years after cessation.
At first glance, these estimates may seem too good to be true. A few months later, though, a different research group, using data from a large cancer prevention study and 2018 US population census and mortality rates, largely confirmed their findings. Dr. Thuy Le and colleagues found that quitting at age 35, 45, 55, 65, or 75 years resulted in average life gains of 8, 5.6, 3.5, 1.7, and 0.7 years, respectively, relative to continuing to smoke. Because no patient is average, the analysis also presented some helpful probabilities. For example, a smoker who quits at age 65 has about a 1 in 4 chance of gaining at least 1 full year of life and a 1 in 6 chance of gaining at least 4 years. In other words, from a life expectancy perspective alone, it’s almost never too late to quit smoking.
Dr. Lin is a family physician and Associate Director, Family Medicine Residency Program, Lancaster General Hospital, Lancaster, Pennsylvania. He blogs at Common Sense Family Doctor. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An estimated 450,000 US deaths are expected this year from conditions attributed to cigarette smoking. Although the percentage of adults who smoke declined from 21% in 2005 to 11% in 2022, the annual death toll has been stable since 2005 and isn’t expected to decline until 2030, owing to an aging population of current and former smokers.
In 2022, based on a national survey, two thirds of the 28.8 million US adult smokers wanted to quit, and more than half tried quitting on their own or with the help of clinicians, but less than 9% succeeded in kicking the habit. The health benefits of quitting, summarized in a patient education handout from the American Cancer Society, include a lower risk for cancer, diabetes, and cardiovascular disease. Furthermore, the handout states, “quitting smoking can add as much as 10 years to your life, compared to if you continued to smoke.”
For my patients older than age 50 who are lifelong smokers, the qualifier “as much as” can be a sticking point. Although most recognize that continuing to smoke exposes them to greater health risks and are willing to undergo lung cancer screening and receive pneumococcal vaccines, a kind of fatalism frequently sets in. I’ve heard more times than I can recall some version of the declaration, “It’s too late for quitting to make much difference for me.” Many smokers think that once they reach middle age, gains in life expectancy will be too small to be worth the intense effort and multiple failed attempts that are typically required to quit permanently. Until recently, there were few data I could call on to persuade them they were wrong.
In February 2024, Dr. Eo Rin Cho and colleagues pooled data from four national cohort studies (United States, United Kingdom, Norway, and Canada) to calculate mortality differences among current, former, and never smokers aged 20-79 years. Compared with never smokers, lifelong smokers died an average of 12-13 years earlier. However, quitting before age 50 nearly eliminated the excess mortality associated with smoking, and in the 50- to 59-year-old age group, cessation eventually reduced excess mortality by 92%-95%. Better yet, more than half of the benefits occurred within the first 3 years after cessation.
At first glance, these estimates may seem too good to be true. A few months later, though, a different research group, using data from a large cancer prevention study and 2018 US population census and mortality rates, largely confirmed their findings. Dr. Thuy Le and colleagues found that quitting at age 35, 45, 55, 65, or 75 years resulted in average life gains of 8, 5.6, 3.5, 1.7, and 0.7 years, respectively, relative to continuing to smoke. Because no patient is average, the analysis also presented some helpful probabilities. For example, a smoker who quits at age 65 has about a 1 in 4 chance of gaining at least 1 full year of life and a 1 in 6 chance of gaining at least 4 years. In other words, from a life expectancy perspective alone, it’s almost never too late to quit smoking.
Dr. Lin is a family physician and Associate Director, Family Medicine Residency Program, Lancaster General Hospital, Lancaster, Pennsylvania. He blogs at Common Sense Family Doctor. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An estimated 450,000 US deaths are expected this year from conditions attributed to cigarette smoking. Although the percentage of adults who smoke declined from 21% in 2005 to 11% in 2022, the annual death toll has been stable since 2005 and isn’t expected to decline until 2030, owing to an aging population of current and former smokers.
In 2022, based on a national survey, two thirds of the 28.8 million US adult smokers wanted to quit, and more than half tried quitting on their own or with the help of clinicians, but less than 9% succeeded in kicking the habit. The health benefits of quitting, summarized in a patient education handout from the American Cancer Society, include a lower risk for cancer, diabetes, and cardiovascular disease. Furthermore, the handout states, “quitting smoking can add as much as 10 years to your life, compared to if you continued to smoke.”
For my patients older than age 50 who are lifelong smokers, the qualifier “as much as” can be a sticking point. Although most recognize that continuing to smoke exposes them to greater health risks and are willing to undergo lung cancer screening and receive pneumococcal vaccines, a kind of fatalism frequently sets in. I’ve heard more times than I can recall some version of the declaration, “It’s too late for quitting to make much difference for me.” Many smokers think that once they reach middle age, gains in life expectancy will be too small to be worth the intense effort and multiple failed attempts that are typically required to quit permanently. Until recently, there were few data I could call on to persuade them they were wrong.
In February 2024, Dr. Eo Rin Cho and colleagues pooled data from four national cohort studies (United States, United Kingdom, Norway, and Canada) to calculate mortality differences among current, former, and never smokers aged 20-79 years. Compared with never smokers, lifelong smokers died an average of 12-13 years earlier. However, quitting before age 50 nearly eliminated the excess mortality associated with smoking, and in the 50- to 59-year-old age group, cessation eventually reduced excess mortality by 92%-95%. Better yet, more than half of the benefits occurred within the first 3 years after cessation.
At first glance, these estimates may seem too good to be true. A few months later, though, a different research group, using data from a large cancer prevention study and 2018 US population census and mortality rates, largely confirmed their findings. Dr. Thuy Le and colleagues found that quitting at age 35, 45, 55, 65, or 75 years resulted in average life gains of 8, 5.6, 3.5, 1.7, and 0.7 years, respectively, relative to continuing to smoke. Because no patient is average, the analysis also presented some helpful probabilities. For example, a smoker who quits at age 65 has about a 1 in 4 chance of gaining at least 1 full year of life and a 1 in 6 chance of gaining at least 4 years. In other words, from a life expectancy perspective alone, it’s almost never too late to quit smoking.
Dr. Lin is a family physician and Associate Director, Family Medicine Residency Program, Lancaster General Hospital, Lancaster, Pennsylvania. He blogs at Common Sense Family Doctor. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Primary Care: Re Carpe Your Diem
William Fox, MD, a self-described “dinosaur,” works in an independent internal medicine practice with two other physicians in Charlottesville, Virginia. He is rarely able to accept new patients, and when he does see one, they often have to wait months for the appointment. He accepts the burden of many pent-up needs, along with the huge administrative chore of coordinating their care with subspecialists.
“I will probably have to make multiple visits in a quick succession in order to make sure that I stabilize all the various issues,” Dr. Fox said. Doing so for a complicated new patient is nearly impossible because of time pressures, especially as it has become increasingly difficult for his patients to access subspecialists.
Dr. Fox traced the roots of the problem to a shortage of primary care physicians.
“Primary care is a vital part of healthcare and infrastructure in the United States, and it is being eroded unfortunately, as fewer and fewer medical students and residents choose to go into the primary care field,” Fox said. “And the reason it’s being eroded partly is because it is undervalued” and under-reimbursed, he added.
A study published 2 years ago in the Journal of General Internal Medicine proved what every primary care clinician already knows: The 24-hour day simply isn’t long enough.
Assuming an average panel of 2500 patients, the authors estimated the average time needed to provide currently recommended preventive care services and vaccines, chronic disease care, and management of acute illnesses. The answer: 26.7 hours a day.
However, using a team-based approach in which the necessary care was divided between physicians, advanced practice providers, and medical assistants, the physician component could be whittled down to 9 hours.
As chair of the Board of Regents of the American College of Physicians (ACP), Dr. Fox champions the ACP’s endorsement of physician-led team-based care, which improves patient outcomes and increases well-being among health professionals. But practices like his rarely have the necessary resources to support advanced practice clinicians or social workers. “Team-based care can be achieved in larger healthcare systems that have the resources to do it,” said Dr. Fox. “We need to find a way for smaller independent practices to also participate in team-based care.”
The solution? Major reform in the current fee-for-service payment structure, which incentivizes patient volume over patient outcomes. Dr. Fox co-authored a 2022 position statement from ACP outlining strategies such as prospective payment models that could achieve high-quality care and address social inequities. “We need to evolve our payment system from a fee-for-service system into a blended system where you have some population-based payments along with fee-for-service, or a fully capitated system,” he said.
Advantages of Team-Based Care
When a patient wrote on a satisfaction survey “the doctor spent more time examining the computer than examining me,” Kevin Hopkins, MD, decided he needed to change some things. Now the vice chief of Cleveland Clinic’s Primary Care Institute in Cleveland, in 2010, he developed a rudimentary team-based care model consisting of himself and two medical assistants at Cleveland Clinic.
The assistants did much of the patient intake, served as scribes while Dr. Hopkins saw his patients, and completed most of the required documentation. “I was able to see 30% more patients in a day and still take great care of them,” Dr. Hopkins said.
The concept of team-based care has evolved since then, often including some combination of advanced practice providers such as physician assistants (PAs) or nurse practitioners (NPs), nurses or medical assistants, and social workers or case managers working under the leadership of a physician. According to Dr. Hopkins, the basic strategy should be that “the physician does what only the physician is uniquely trained and qualified to do.” All other tasks, such as data entry, handling refill requests and messages from the patient portal, scheduling, or patient education, can — and should — be done by someone else.
Dr. Hopkins also serves as a senior physician advisor to the American Medical Association (AMA) and an instructor for workshops like “Saving Time: Practice Innovation Boot Camp.” His advice for clinicians who would like to streamline their workflow is to start with small steps.
“You’re not going to be able to hire all the people that you’d like to have,” he said. “I encourage physicians to look around at the people that they have and what they are currently doing as a part of their roles and responsibilities.”
The AMA Team-Based Care and Workflow website provides brief continuing medical education activities on topics such as implementing lab testing prior to office visits or advanced protocols for rooming and discharge; adopting any of these strategies can help save steps during office visits.
Dr. Hopkins said the AMA is committed to reducing the regulatory burden on clinicians. Clinical compliance officers may misinterpret regulatory requirements, putting into place overly conservative internal policies and procedures. The AMA’s “Reducing Regulatory Burden Playbook” offers advice on practices that could be stopped, such as two-factor authentication for approving or signing orders unless they are for controlled substances, or started, such as writing prescriptions for chronic daily medications for the maximum allowed length. Reducing a few clicks with each log-on to the electronic health record or reducing the number of tasks physicians must complete to log on can dramatically reduce hours spent on the computer.
Telehealth Changes Everything
But it might be even more efficient to keep patients out of the office.
Initially piloted in Palo Alto, California, from 2018 to 2022, the Tera Practice adopted team-based care strategies in a practice that conducts most of its work virtually. Sutter Health has since expanded the concept to three “Connected Care Clinics” in the San Francisco area, which provide 80% of their care via secure messaging, telephone, or video visit. Staff work primarily from home, and patients do not need to take off work or spend time driving to in-person visits to access care.
Matthew Sakumoto, MD, is a virtualist primary care physician at Sutter Health’s San Francisco clinic and an adjunct assistant clinical professor at the University of California, San Francisco. He leads a three-person team, which typically has included either an NP or a PA, along with a licensed vocational nurse or medical assistant.
“I go into clinic once a week, but a lot of the visits are video visits, so I don’t feel the pressure to address everything all in one visit,” Dr. Sakumoto said. “It is really freeing for both me and the patient.”
For more complex patients, for example, he prioritizes one problem at a visit because his team will follow up virtually for administrative issues like refills or specialty referrals. He can easily find time to schedule a patient for a follow-up virtual visit in the next week or 2 weeks to address additional needs. And on days he and his staff work from home, patients who message with an urgent concern can often be seen by video that same day.
Dr. Sakumoto and his team have a traditional huddle in the office the morning of their clinic day to plan for the scheduled visits, but most of their days are less structured. On non-clinic days, their morning Zoom huddle has a much different focus.
“We’re saying, ‘Okay, who are our high-risk patients? Who haven’t we seen in a while?’ ” Dr. Sakumoto said. They group patients into tiers based on factors such as age, number of complex medical needs, and frequency of emergency visits or hospitalizations. They also check in with higher-risk patients who haven’t contacted the office or been seen recently.
Dr. Sakumoto noticed in medical school doctors take excellent care of the patients who show up to the clinic. “We don’t do quite as well for those that don’t show up,” he said. The hybrid-virtual model gives him time to think in a more population-based way about engaging his entire panel of patients. The majority have capitated or value-based insurance plans, providing a dedicated monthly revenue stream that funds his virtual practice.
More clinicians may be able to hire staff and adopt some of these innovative approaches if Congress approves proposed changes to Medicare and Medicaid increasing the use of value-based payment systems. Although these changes may not provide all the funding needed to reinvigorate the field of primary care medicine, Dr. Fox said, “at least some people in positions of power and responsibility are beginning to think more seriously about these issues.”
Dr. Sakumoto reported personal fees from the following companies within the past 24 months: Clearstep Health, Carbon Health, Matter Health, CareAlign, PlushCare, Teladoc, and Nabla Health. Dr. Fox and Dr. Hopkins reported no financial conflicts of interest.
A former pediatrician and disease detective, Ann Thomas, MD, MPH, is a freelance science writer living in Portland, Oregon.
A version of this article first appeared on Medscape.com.
William Fox, MD, a self-described “dinosaur,” works in an independent internal medicine practice with two other physicians in Charlottesville, Virginia. He is rarely able to accept new patients, and when he does see one, they often have to wait months for the appointment. He accepts the burden of many pent-up needs, along with the huge administrative chore of coordinating their care with subspecialists.
“I will probably have to make multiple visits in a quick succession in order to make sure that I stabilize all the various issues,” Dr. Fox said. Doing so for a complicated new patient is nearly impossible because of time pressures, especially as it has become increasingly difficult for his patients to access subspecialists.
Dr. Fox traced the roots of the problem to a shortage of primary care physicians.
“Primary care is a vital part of healthcare and infrastructure in the United States, and it is being eroded unfortunately, as fewer and fewer medical students and residents choose to go into the primary care field,” Fox said. “And the reason it’s being eroded partly is because it is undervalued” and under-reimbursed, he added.
A study published 2 years ago in the Journal of General Internal Medicine proved what every primary care clinician already knows: The 24-hour day simply isn’t long enough.
Assuming an average panel of 2500 patients, the authors estimated the average time needed to provide currently recommended preventive care services and vaccines, chronic disease care, and management of acute illnesses. The answer: 26.7 hours a day.
However, using a team-based approach in which the necessary care was divided between physicians, advanced practice providers, and medical assistants, the physician component could be whittled down to 9 hours.
As chair of the Board of Regents of the American College of Physicians (ACP), Dr. Fox champions the ACP’s endorsement of physician-led team-based care, which improves patient outcomes and increases well-being among health professionals. But practices like his rarely have the necessary resources to support advanced practice clinicians or social workers. “Team-based care can be achieved in larger healthcare systems that have the resources to do it,” said Dr. Fox. “We need to find a way for smaller independent practices to also participate in team-based care.”
The solution? Major reform in the current fee-for-service payment structure, which incentivizes patient volume over patient outcomes. Dr. Fox co-authored a 2022 position statement from ACP outlining strategies such as prospective payment models that could achieve high-quality care and address social inequities. “We need to evolve our payment system from a fee-for-service system into a blended system where you have some population-based payments along with fee-for-service, or a fully capitated system,” he said.
Advantages of Team-Based Care
When a patient wrote on a satisfaction survey “the doctor spent more time examining the computer than examining me,” Kevin Hopkins, MD, decided he needed to change some things. Now the vice chief of Cleveland Clinic’s Primary Care Institute in Cleveland, in 2010, he developed a rudimentary team-based care model consisting of himself and two medical assistants at Cleveland Clinic.
The assistants did much of the patient intake, served as scribes while Dr. Hopkins saw his patients, and completed most of the required documentation. “I was able to see 30% more patients in a day and still take great care of them,” Dr. Hopkins said.
The concept of team-based care has evolved since then, often including some combination of advanced practice providers such as physician assistants (PAs) or nurse practitioners (NPs), nurses or medical assistants, and social workers or case managers working under the leadership of a physician. According to Dr. Hopkins, the basic strategy should be that “the physician does what only the physician is uniquely trained and qualified to do.” All other tasks, such as data entry, handling refill requests and messages from the patient portal, scheduling, or patient education, can — and should — be done by someone else.
Dr. Hopkins also serves as a senior physician advisor to the American Medical Association (AMA) and an instructor for workshops like “Saving Time: Practice Innovation Boot Camp.” His advice for clinicians who would like to streamline their workflow is to start with small steps.
“You’re not going to be able to hire all the people that you’d like to have,” he said. “I encourage physicians to look around at the people that they have and what they are currently doing as a part of their roles and responsibilities.”
The AMA Team-Based Care and Workflow website provides brief continuing medical education activities on topics such as implementing lab testing prior to office visits or advanced protocols for rooming and discharge; adopting any of these strategies can help save steps during office visits.
Dr. Hopkins said the AMA is committed to reducing the regulatory burden on clinicians. Clinical compliance officers may misinterpret regulatory requirements, putting into place overly conservative internal policies and procedures. The AMA’s “Reducing Regulatory Burden Playbook” offers advice on practices that could be stopped, such as two-factor authentication for approving or signing orders unless they are for controlled substances, or started, such as writing prescriptions for chronic daily medications for the maximum allowed length. Reducing a few clicks with each log-on to the electronic health record or reducing the number of tasks physicians must complete to log on can dramatically reduce hours spent on the computer.
Telehealth Changes Everything
But it might be even more efficient to keep patients out of the office.
Initially piloted in Palo Alto, California, from 2018 to 2022, the Tera Practice adopted team-based care strategies in a practice that conducts most of its work virtually. Sutter Health has since expanded the concept to three “Connected Care Clinics” in the San Francisco area, which provide 80% of their care via secure messaging, telephone, or video visit. Staff work primarily from home, and patients do not need to take off work or spend time driving to in-person visits to access care.
Matthew Sakumoto, MD, is a virtualist primary care physician at Sutter Health’s San Francisco clinic and an adjunct assistant clinical professor at the University of California, San Francisco. He leads a three-person team, which typically has included either an NP or a PA, along with a licensed vocational nurse or medical assistant.
“I go into clinic once a week, but a lot of the visits are video visits, so I don’t feel the pressure to address everything all in one visit,” Dr. Sakumoto said. “It is really freeing for both me and the patient.”
For more complex patients, for example, he prioritizes one problem at a visit because his team will follow up virtually for administrative issues like refills or specialty referrals. He can easily find time to schedule a patient for a follow-up virtual visit in the next week or 2 weeks to address additional needs. And on days he and his staff work from home, patients who message with an urgent concern can often be seen by video that same day.
Dr. Sakumoto and his team have a traditional huddle in the office the morning of their clinic day to plan for the scheduled visits, but most of their days are less structured. On non-clinic days, their morning Zoom huddle has a much different focus.
“We’re saying, ‘Okay, who are our high-risk patients? Who haven’t we seen in a while?’ ” Dr. Sakumoto said. They group patients into tiers based on factors such as age, number of complex medical needs, and frequency of emergency visits or hospitalizations. They also check in with higher-risk patients who haven’t contacted the office or been seen recently.
Dr. Sakumoto noticed in medical school doctors take excellent care of the patients who show up to the clinic. “We don’t do quite as well for those that don’t show up,” he said. The hybrid-virtual model gives him time to think in a more population-based way about engaging his entire panel of patients. The majority have capitated or value-based insurance plans, providing a dedicated monthly revenue stream that funds his virtual practice.
More clinicians may be able to hire staff and adopt some of these innovative approaches if Congress approves proposed changes to Medicare and Medicaid increasing the use of value-based payment systems. Although these changes may not provide all the funding needed to reinvigorate the field of primary care medicine, Dr. Fox said, “at least some people in positions of power and responsibility are beginning to think more seriously about these issues.”
Dr. Sakumoto reported personal fees from the following companies within the past 24 months: Clearstep Health, Carbon Health, Matter Health, CareAlign, PlushCare, Teladoc, and Nabla Health. Dr. Fox and Dr. Hopkins reported no financial conflicts of interest.
A former pediatrician and disease detective, Ann Thomas, MD, MPH, is a freelance science writer living in Portland, Oregon.
A version of this article first appeared on Medscape.com.
William Fox, MD, a self-described “dinosaur,” works in an independent internal medicine practice with two other physicians in Charlottesville, Virginia. He is rarely able to accept new patients, and when he does see one, they often have to wait months for the appointment. He accepts the burden of many pent-up needs, along with the huge administrative chore of coordinating their care with subspecialists.
“I will probably have to make multiple visits in a quick succession in order to make sure that I stabilize all the various issues,” Dr. Fox said. Doing so for a complicated new patient is nearly impossible because of time pressures, especially as it has become increasingly difficult for his patients to access subspecialists.
Dr. Fox traced the roots of the problem to a shortage of primary care physicians.
“Primary care is a vital part of healthcare and infrastructure in the United States, and it is being eroded unfortunately, as fewer and fewer medical students and residents choose to go into the primary care field,” Fox said. “And the reason it’s being eroded partly is because it is undervalued” and under-reimbursed, he added.
A study published 2 years ago in the Journal of General Internal Medicine proved what every primary care clinician already knows: The 24-hour day simply isn’t long enough.
Assuming an average panel of 2500 patients, the authors estimated the average time needed to provide currently recommended preventive care services and vaccines, chronic disease care, and management of acute illnesses. The answer: 26.7 hours a day.
However, using a team-based approach in which the necessary care was divided between physicians, advanced practice providers, and medical assistants, the physician component could be whittled down to 9 hours.
As chair of the Board of Regents of the American College of Physicians (ACP), Dr. Fox champions the ACP’s endorsement of physician-led team-based care, which improves patient outcomes and increases well-being among health professionals. But practices like his rarely have the necessary resources to support advanced practice clinicians or social workers. “Team-based care can be achieved in larger healthcare systems that have the resources to do it,” said Dr. Fox. “We need to find a way for smaller independent practices to also participate in team-based care.”
The solution? Major reform in the current fee-for-service payment structure, which incentivizes patient volume over patient outcomes. Dr. Fox co-authored a 2022 position statement from ACP outlining strategies such as prospective payment models that could achieve high-quality care and address social inequities. “We need to evolve our payment system from a fee-for-service system into a blended system where you have some population-based payments along with fee-for-service, or a fully capitated system,” he said.
Advantages of Team-Based Care
When a patient wrote on a satisfaction survey “the doctor spent more time examining the computer than examining me,” Kevin Hopkins, MD, decided he needed to change some things. Now the vice chief of Cleveland Clinic’s Primary Care Institute in Cleveland, in 2010, he developed a rudimentary team-based care model consisting of himself and two medical assistants at Cleveland Clinic.
The assistants did much of the patient intake, served as scribes while Dr. Hopkins saw his patients, and completed most of the required documentation. “I was able to see 30% more patients in a day and still take great care of them,” Dr. Hopkins said.
The concept of team-based care has evolved since then, often including some combination of advanced practice providers such as physician assistants (PAs) or nurse practitioners (NPs), nurses or medical assistants, and social workers or case managers working under the leadership of a physician. According to Dr. Hopkins, the basic strategy should be that “the physician does what only the physician is uniquely trained and qualified to do.” All other tasks, such as data entry, handling refill requests and messages from the patient portal, scheduling, or patient education, can — and should — be done by someone else.
Dr. Hopkins also serves as a senior physician advisor to the American Medical Association (AMA) and an instructor for workshops like “Saving Time: Practice Innovation Boot Camp.” His advice for clinicians who would like to streamline their workflow is to start with small steps.
“You’re not going to be able to hire all the people that you’d like to have,” he said. “I encourage physicians to look around at the people that they have and what they are currently doing as a part of their roles and responsibilities.”
The AMA Team-Based Care and Workflow website provides brief continuing medical education activities on topics such as implementing lab testing prior to office visits or advanced protocols for rooming and discharge; adopting any of these strategies can help save steps during office visits.
Dr. Hopkins said the AMA is committed to reducing the regulatory burden on clinicians. Clinical compliance officers may misinterpret regulatory requirements, putting into place overly conservative internal policies and procedures. The AMA’s “Reducing Regulatory Burden Playbook” offers advice on practices that could be stopped, such as two-factor authentication for approving or signing orders unless they are for controlled substances, or started, such as writing prescriptions for chronic daily medications for the maximum allowed length. Reducing a few clicks with each log-on to the electronic health record or reducing the number of tasks physicians must complete to log on can dramatically reduce hours spent on the computer.
Telehealth Changes Everything
But it might be even more efficient to keep patients out of the office.
Initially piloted in Palo Alto, California, from 2018 to 2022, the Tera Practice adopted team-based care strategies in a practice that conducts most of its work virtually. Sutter Health has since expanded the concept to three “Connected Care Clinics” in the San Francisco area, which provide 80% of their care via secure messaging, telephone, or video visit. Staff work primarily from home, and patients do not need to take off work or spend time driving to in-person visits to access care.
Matthew Sakumoto, MD, is a virtualist primary care physician at Sutter Health’s San Francisco clinic and an adjunct assistant clinical professor at the University of California, San Francisco. He leads a three-person team, which typically has included either an NP or a PA, along with a licensed vocational nurse or medical assistant.
“I go into clinic once a week, but a lot of the visits are video visits, so I don’t feel the pressure to address everything all in one visit,” Dr. Sakumoto said. “It is really freeing for both me and the patient.”
For more complex patients, for example, he prioritizes one problem at a visit because his team will follow up virtually for administrative issues like refills or specialty referrals. He can easily find time to schedule a patient for a follow-up virtual visit in the next week or 2 weeks to address additional needs. And on days he and his staff work from home, patients who message with an urgent concern can often be seen by video that same day.
Dr. Sakumoto and his team have a traditional huddle in the office the morning of their clinic day to plan for the scheduled visits, but most of their days are less structured. On non-clinic days, their morning Zoom huddle has a much different focus.
“We’re saying, ‘Okay, who are our high-risk patients? Who haven’t we seen in a while?’ ” Dr. Sakumoto said. They group patients into tiers based on factors such as age, number of complex medical needs, and frequency of emergency visits or hospitalizations. They also check in with higher-risk patients who haven’t contacted the office or been seen recently.
Dr. Sakumoto noticed in medical school doctors take excellent care of the patients who show up to the clinic. “We don’t do quite as well for those that don’t show up,” he said. The hybrid-virtual model gives him time to think in a more population-based way about engaging his entire panel of patients. The majority have capitated or value-based insurance plans, providing a dedicated monthly revenue stream that funds his virtual practice.
More clinicians may be able to hire staff and adopt some of these innovative approaches if Congress approves proposed changes to Medicare and Medicaid increasing the use of value-based payment systems. Although these changes may not provide all the funding needed to reinvigorate the field of primary care medicine, Dr. Fox said, “at least some people in positions of power and responsibility are beginning to think more seriously about these issues.”
Dr. Sakumoto reported personal fees from the following companies within the past 24 months: Clearstep Health, Carbon Health, Matter Health, CareAlign, PlushCare, Teladoc, and Nabla Health. Dr. Fox and Dr. Hopkins reported no financial conflicts of interest.
A former pediatrician and disease detective, Ann Thomas, MD, MPH, is a freelance science writer living in Portland, Oregon.
A version of this article first appeared on Medscape.com.
Rural Women Face Greater Challenges in Perimenopause
Women in the perimenopausal period who live in rural areas have a higher prevalence of symptoms typical of this period and a poorer health-related quality of life than women living in urban areas, according to a cross-sectional study that was conducted in Spain.
Cristina Llaneza Suárez, a specialist in family and community medicine and the lead author of the study, told this news organization that women living in rural areas face greater difficulties with access to healthcare services, employment, and transportation and a heavier burden of caregiving. She mentioned that these barriers “can represent an added challenge during the perimenopausal stage, when significant life changes generally occur for all women.” The challenges may lead to “poorer health-related quality of life during perimenopause, compared with women living in urban areas.”
The research group led by Dr. Llaneza aimed to test the hypothesis that sociodemographic characteristics influence symptoms and quality of life in women in perimenopause. They enrolled 270 women aged 45-55 years from eight autonomous communities in Spain who had variability in their menstrual cycles (lasting more than 7 days or amenorrhea greater than 60 days but less than a year).
This cross-sectional study was conducted from December 2019 to April 2023, using the short version of the Cervantes scale to assess health-related quality of life and the Beck Depression Inventory to evaluate associated depressive symptoms.
Among the main findings of the study was that sociocultural factors can influence the perception of perimenopausal symptoms. Living in rural areas has a negative effect on health-related quality of life scales, and this finding is consistent with those of previous studies conducted on women in India, Turkey, Poland, and Peru.
In addition, the selected sample of women experiencing changes in their menstrual cycles and residing in rural areas showed a high prevalence of hot flashes (70% overall and 80% in rural areas) and a poorer quality of life in women with obesity.
“It is striking that, although there is a worse perception of quality of life during perimenopause in women living in rural areas, the proportion of women experiencing some degree of depressive symptoms, according to the Beck inventory, was similar to that of women residing in urban areas,” said Dr. Llaneza. She noted that “no worse scores were observed in sexuality or in the couple relationship.”
Rural Physicians’ Role
Women in the perimenopausal period face significant challenges resulting from inadequate access to healthcare services and limited awareness about menopause. In many countries, this topic is still taboo, both in the family environment and in workplaces and health centers.
Dr. Llaneza mentioned that when she began her training as a primary care physician in a rural population, she witnessed firsthand some of the barriers that women in this age group face, such as limited access to healthcare due to a lack of public transportation. She added that, coupled with this challenge, “there are no regular public transport services that allow independent access for patients, and many [women] lack a driver’s license, making them dependent on others to receive healthcare.” Another important point that she identified was the lack of health education in rural populations, which leads to a minimization of perimenopausal symptoms and causes delays in prevention and early detection.
According to the World Health Organization, healthcare professionals often lack the necessary training to recognize and treat the symptoms of perimenopause and postmenopause. This situation, coupled with the limited attention given to the sexual well-being of menopausal women, contributes to gynecological problems and risks for sexually transmitted infections in this population. The absence of specific health policies and funding for menopause exacerbates the situation, particularly in regions where other health needs compete for limited resources.
Dr. Llaneza noted that primary care physicians in rural areas are responsible for leading primary prevention actions through community interventions that contribute to improving health. Community physicians in rural areas have a lower patient load than urban physicians do. Therefore, “this allows for a more thorough management and closer monitoring of these conditions, which highlights the importance of prevention of perimenopausal symptoms and community education,” she said.
An important goal in improving the quality of life of women in the perimenopausal period is reducing symptoms. Hormone replacement therapy is the cornerstone of treatment, along with nonhormonal therapies such as the use of isoflavones. However, the aforementioned barriers lead to a delay in initiating effective treatment.
Dr. Llaneza added that the main limitation that she encountered during her clinical practice in rural areas regarding the initiation of hormonal therapy was “the reluctance of certain professionals to start it, as they consider that these drugs should be prescribed by menopause specialists because of potential side effects and the increased risk for developing breast cancer.”
Call for Training
Dr. Llaneza and her research team emphasized the need for further research on new drugs for controlling vasomotor symptoms, expressing their interest in conducting additional studies. “We would like to conduct a study on the use of these therapies in perimenopausal and postmenopausal women residing in rural areas.
“We believe that our data may be of interest to healthcare authorities seeking to combat population exodus in rural areas,” they wrote. In addition, they recommended additional training for rural primary care physicians on perimenopause and menopause topics regarding prevention, management, and access, as well as further awareness about preventing depressive symptoms in this population.
Dr. Llaneza declared that she has no relevant financial relationships.
This story was translated from the Medscape Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Women in the perimenopausal period who live in rural areas have a higher prevalence of symptoms typical of this period and a poorer health-related quality of life than women living in urban areas, according to a cross-sectional study that was conducted in Spain.
Cristina Llaneza Suárez, a specialist in family and community medicine and the lead author of the study, told this news organization that women living in rural areas face greater difficulties with access to healthcare services, employment, and transportation and a heavier burden of caregiving. She mentioned that these barriers “can represent an added challenge during the perimenopausal stage, when significant life changes generally occur for all women.” The challenges may lead to “poorer health-related quality of life during perimenopause, compared with women living in urban areas.”
The research group led by Dr. Llaneza aimed to test the hypothesis that sociodemographic characteristics influence symptoms and quality of life in women in perimenopause. They enrolled 270 women aged 45-55 years from eight autonomous communities in Spain who had variability in their menstrual cycles (lasting more than 7 days or amenorrhea greater than 60 days but less than a year).
This cross-sectional study was conducted from December 2019 to April 2023, using the short version of the Cervantes scale to assess health-related quality of life and the Beck Depression Inventory to evaluate associated depressive symptoms.
Among the main findings of the study was that sociocultural factors can influence the perception of perimenopausal symptoms. Living in rural areas has a negative effect on health-related quality of life scales, and this finding is consistent with those of previous studies conducted on women in India, Turkey, Poland, and Peru.
In addition, the selected sample of women experiencing changes in their menstrual cycles and residing in rural areas showed a high prevalence of hot flashes (70% overall and 80% in rural areas) and a poorer quality of life in women with obesity.
“It is striking that, although there is a worse perception of quality of life during perimenopause in women living in rural areas, the proportion of women experiencing some degree of depressive symptoms, according to the Beck inventory, was similar to that of women residing in urban areas,” said Dr. Llaneza. She noted that “no worse scores were observed in sexuality or in the couple relationship.”
Rural Physicians’ Role
Women in the perimenopausal period face significant challenges resulting from inadequate access to healthcare services and limited awareness about menopause. In many countries, this topic is still taboo, both in the family environment and in workplaces and health centers.
Dr. Llaneza mentioned that when she began her training as a primary care physician in a rural population, she witnessed firsthand some of the barriers that women in this age group face, such as limited access to healthcare due to a lack of public transportation. She added that, coupled with this challenge, “there are no regular public transport services that allow independent access for patients, and many [women] lack a driver’s license, making them dependent on others to receive healthcare.” Another important point that she identified was the lack of health education in rural populations, which leads to a minimization of perimenopausal symptoms and causes delays in prevention and early detection.
According to the World Health Organization, healthcare professionals often lack the necessary training to recognize and treat the symptoms of perimenopause and postmenopause. This situation, coupled with the limited attention given to the sexual well-being of menopausal women, contributes to gynecological problems and risks for sexually transmitted infections in this population. The absence of specific health policies and funding for menopause exacerbates the situation, particularly in regions where other health needs compete for limited resources.
Dr. Llaneza noted that primary care physicians in rural areas are responsible for leading primary prevention actions through community interventions that contribute to improving health. Community physicians in rural areas have a lower patient load than urban physicians do. Therefore, “this allows for a more thorough management and closer monitoring of these conditions, which highlights the importance of prevention of perimenopausal symptoms and community education,” she said.
An important goal in improving the quality of life of women in the perimenopausal period is reducing symptoms. Hormone replacement therapy is the cornerstone of treatment, along with nonhormonal therapies such as the use of isoflavones. However, the aforementioned barriers lead to a delay in initiating effective treatment.
Dr. Llaneza added that the main limitation that she encountered during her clinical practice in rural areas regarding the initiation of hormonal therapy was “the reluctance of certain professionals to start it, as they consider that these drugs should be prescribed by menopause specialists because of potential side effects and the increased risk for developing breast cancer.”
Call for Training
Dr. Llaneza and her research team emphasized the need for further research on new drugs for controlling vasomotor symptoms, expressing their interest in conducting additional studies. “We would like to conduct a study on the use of these therapies in perimenopausal and postmenopausal women residing in rural areas.
“We believe that our data may be of interest to healthcare authorities seeking to combat population exodus in rural areas,” they wrote. In addition, they recommended additional training for rural primary care physicians on perimenopause and menopause topics regarding prevention, management, and access, as well as further awareness about preventing depressive symptoms in this population.
Dr. Llaneza declared that she has no relevant financial relationships.
This story was translated from the Medscape Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Women in the perimenopausal period who live in rural areas have a higher prevalence of symptoms typical of this period and a poorer health-related quality of life than women living in urban areas, according to a cross-sectional study that was conducted in Spain.
Cristina Llaneza Suárez, a specialist in family and community medicine and the lead author of the study, told this news organization that women living in rural areas face greater difficulties with access to healthcare services, employment, and transportation and a heavier burden of caregiving. She mentioned that these barriers “can represent an added challenge during the perimenopausal stage, when significant life changes generally occur for all women.” The challenges may lead to “poorer health-related quality of life during perimenopause, compared with women living in urban areas.”
The research group led by Dr. Llaneza aimed to test the hypothesis that sociodemographic characteristics influence symptoms and quality of life in women in perimenopause. They enrolled 270 women aged 45-55 years from eight autonomous communities in Spain who had variability in their menstrual cycles (lasting more than 7 days or amenorrhea greater than 60 days but less than a year).
This cross-sectional study was conducted from December 2019 to April 2023, using the short version of the Cervantes scale to assess health-related quality of life and the Beck Depression Inventory to evaluate associated depressive symptoms.
Among the main findings of the study was that sociocultural factors can influence the perception of perimenopausal symptoms. Living in rural areas has a negative effect on health-related quality of life scales, and this finding is consistent with those of previous studies conducted on women in India, Turkey, Poland, and Peru.
In addition, the selected sample of women experiencing changes in their menstrual cycles and residing in rural areas showed a high prevalence of hot flashes (70% overall and 80% in rural areas) and a poorer quality of life in women with obesity.
“It is striking that, although there is a worse perception of quality of life during perimenopause in women living in rural areas, the proportion of women experiencing some degree of depressive symptoms, according to the Beck inventory, was similar to that of women residing in urban areas,” said Dr. Llaneza. She noted that “no worse scores were observed in sexuality or in the couple relationship.”
Rural Physicians’ Role
Women in the perimenopausal period face significant challenges resulting from inadequate access to healthcare services and limited awareness about menopause. In many countries, this topic is still taboo, both in the family environment and in workplaces and health centers.
Dr. Llaneza mentioned that when she began her training as a primary care physician in a rural population, she witnessed firsthand some of the barriers that women in this age group face, such as limited access to healthcare due to a lack of public transportation. She added that, coupled with this challenge, “there are no regular public transport services that allow independent access for patients, and many [women] lack a driver’s license, making them dependent on others to receive healthcare.” Another important point that she identified was the lack of health education in rural populations, which leads to a minimization of perimenopausal symptoms and causes delays in prevention and early detection.
According to the World Health Organization, healthcare professionals often lack the necessary training to recognize and treat the symptoms of perimenopause and postmenopause. This situation, coupled with the limited attention given to the sexual well-being of menopausal women, contributes to gynecological problems and risks for sexually transmitted infections in this population. The absence of specific health policies and funding for menopause exacerbates the situation, particularly in regions where other health needs compete for limited resources.
Dr. Llaneza noted that primary care physicians in rural areas are responsible for leading primary prevention actions through community interventions that contribute to improving health. Community physicians in rural areas have a lower patient load than urban physicians do. Therefore, “this allows for a more thorough management and closer monitoring of these conditions, which highlights the importance of prevention of perimenopausal symptoms and community education,” she said.
An important goal in improving the quality of life of women in the perimenopausal period is reducing symptoms. Hormone replacement therapy is the cornerstone of treatment, along with nonhormonal therapies such as the use of isoflavones. However, the aforementioned barriers lead to a delay in initiating effective treatment.
Dr. Llaneza added that the main limitation that she encountered during her clinical practice in rural areas regarding the initiation of hormonal therapy was “the reluctance of certain professionals to start it, as they consider that these drugs should be prescribed by menopause specialists because of potential side effects and the increased risk for developing breast cancer.”
Call for Training
Dr. Llaneza and her research team emphasized the need for further research on new drugs for controlling vasomotor symptoms, expressing their interest in conducting additional studies. “We would like to conduct a study on the use of these therapies in perimenopausal and postmenopausal women residing in rural areas.
“We believe that our data may be of interest to healthcare authorities seeking to combat population exodus in rural areas,” they wrote. In addition, they recommended additional training for rural primary care physicians on perimenopause and menopause topics regarding prevention, management, and access, as well as further awareness about preventing depressive symptoms in this population.
Dr. Llaneza declared that she has no relevant financial relationships.
This story was translated from the Medscape Spanish edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Misdiagnosis of Crusted Scabies: Skin Excoriations Resembling Brown Sugar Are Characteristic
To the Editor:
Crusted scabies (formerly known as Norwegian scabies) is a rare and highly contagious variant of scabies, in which the skin is infested with thousands to millions of Sarcoptes scabiei var hominis mites. We present a case of skin changes that were misdiagnosed as atopic dermatitis, seborrhea, xerosis, and drug eruption on initial presentation, which prompted treatment with a corticosteroid that inadvertently caused progression to crusted scabies.
A 79-year-old woman who uses a wheelchair presented to the clinic with skin changes that consisted of diffuse, severely pruritic, erythematous plaques on the head, neck, trunk, face, and extremities of 2 years’ duration. She had a medical history of hyperlipidemia, hypertension, and hyperglycemia, as well as a stroke that required hospitalization 2 years prior to the onset of the skin changes. She had no history of allergies.
Prior clinical diagnoses by primary care and dermatology included xerosis, atopic dermatitis, seborrhea, and drug eruption. She was treated with a mid-potency topical corticosteroid (triamcinolone acetonide cream 0.1%) twice daily and prednisone 40 mg once daily for 2- to 4-week courses over an 8-month period without reduction in symptoms.
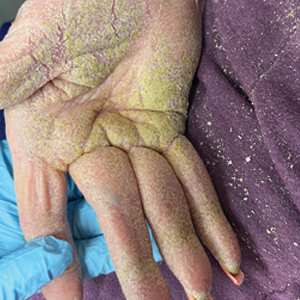
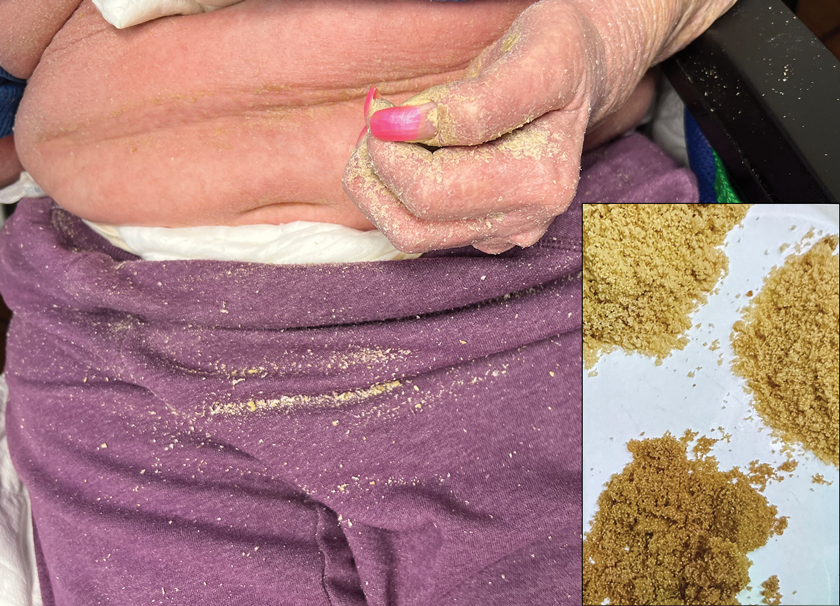
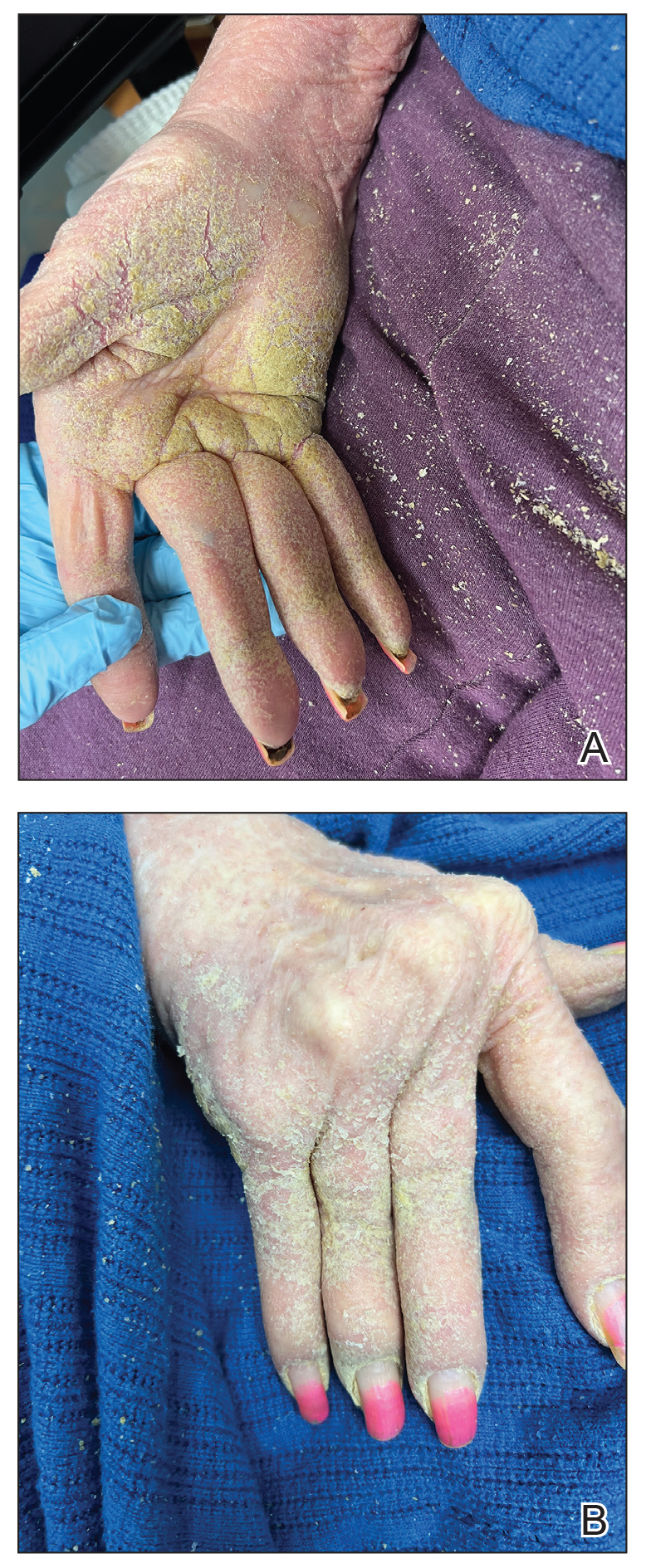
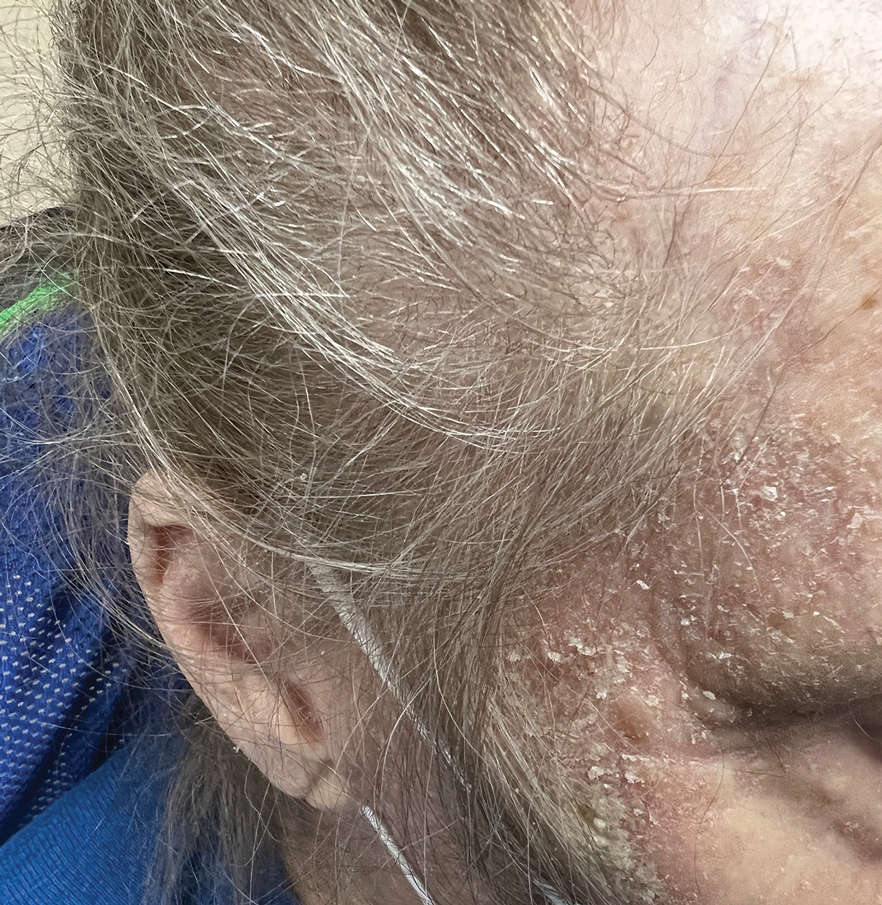
Physical examination at the current presentation revealed golden, crusted, fine, powdery but slightly sticky flakes that spread diffusely across the entire body and came off in crumbles with a simple touch. These widespread crusts were easily visible on clothing. There was underlying diffuse erythema beneath the flaking skin on the trunk and proximal extremities. The scale and shedding skin laid in piles on the patient’s lap and resembled brown sugar (Figure 1). The patient also reported decreased hand function and dexterity due to the yellowbrown, thick, crusty plaques that had developed on both the palmar and dorsal sides of the hands (Figure 2). Erythematous plaques on the scalp, forehead, and inner ears resembled seborrhea (Figure 3). Pruritus severity was rated by the patient as 10 of 10, and she scratched her skin the entire time she was in the clinic. The patient was emotional and stated that she had not been able to sleep due to the discomfort. We suspected scabies, and the patient was reassured to learn that it could be confirmed with a simple skin scrape test.
The crusted lesions on the patient's hands were scraped with a #15-blade scalpel, and a routine potassium hydroxide mount was performed. The skin scrapings were placed on a slide with a drop of 10% potassium hydroxide and observed under low-power (×10) and high-power (×40) microscopy, which revealed thousands of mites and eggs (along with previously hatched eggs) (Figure 4) and quickly confirmed a diagnosis of crusted scabies.an extremely contagious form of scabies seen in older patients with compromised immune systems, malnutrition, or disabilities. The patient was prescribed oral ivermectin (3 mg dosed at 200 μg/kg of body weight) and topical permethrin 5%, neither of which she took, as she died of a COVID-19 infection complication 3 days after this diagnostic clinic visit.
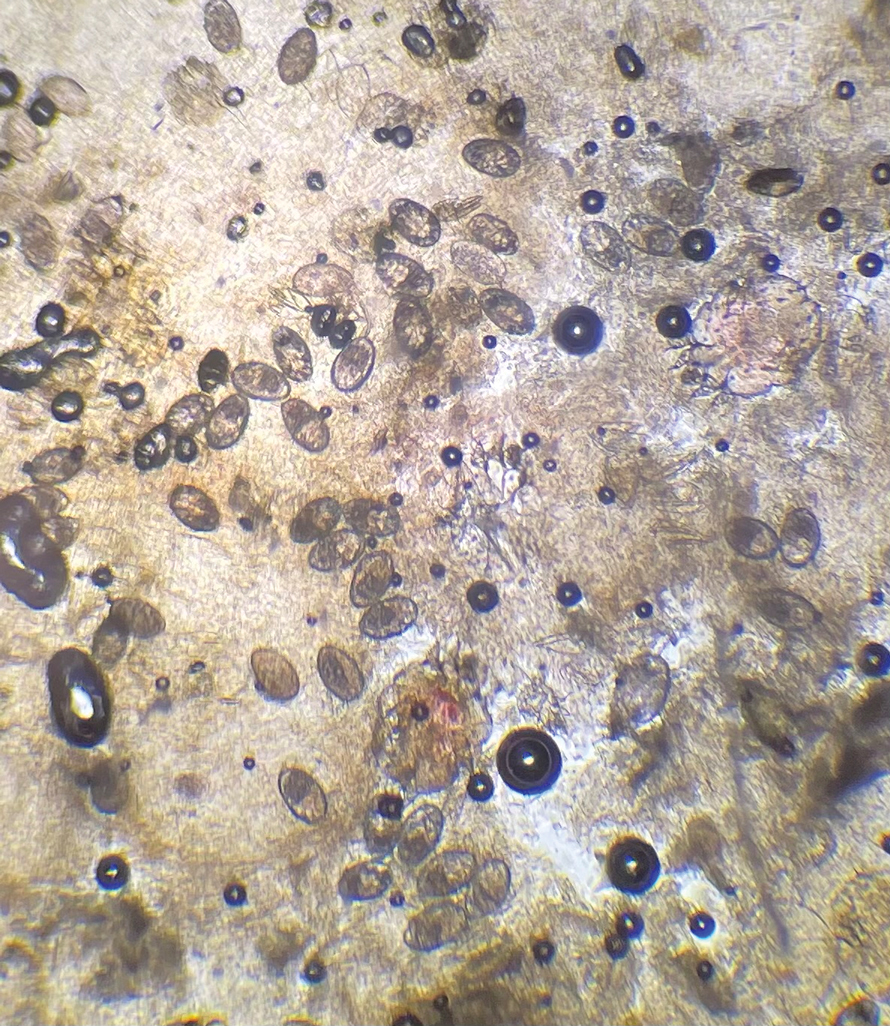
Classic and crusted scabies are both caused by infestation of the Sarcoptes scabiei var hominis mite. Classic scabies is a result of an infestation of a small number of mites (commonly 5–15 mites), while crusted scabies is due to hyperinfestation by as many as millions of mites, the latter often requiring more aggressive treatment. The mites are first transmitted to humans by either skin-toskin contact or fomites on bedding and clothing. The scabies mite undergoes 4 life cycle stages: egg, larvae, nymph, and adult. Once female mites are transmitted, they burrow under the skin and lay 2 to 3 eggs per day. The eggs hatch within 3 to 4 days, after which the larvae migrate to the skin surface. The larval stage lasts for 3 to 4 days, during which the larvae burrow into the stratum corneum to create molting pouches, until they molt into slightly larger nymphs. Nymphs can be found in hair follicles or molting pouches until they further molt within 3 to 4 days into adults, which are round, saclike mites. The adult male and female mites then mate, leaving the female fertile for the rest of her 1- to 2-month lifespan. Impregnated female mites traverse the skin surface in search of a burrow site, using the pulvilli on the anterior aspect of 2 legs to hold onto the skin. Once burrowed, the female mite continues to lay eggs for the rest of her life, with approximately 10% of her eggs resulting in adult mites. Male mites feed in shallow pits of the skin until they find a female burrow site for mating.1 This continuous life cycle of the scabies mite gives rise to highly transmissible, pruritic skin excoriations, as demonstrated in our patient.
The skin has a relatively late inflammatory and adaptive immune response to scabies, typically occurring 4 to 6 weeks after the initial infestation.2 This delayed inflammatory response and onset of symptoms may be due to the scabies mite’s ability to alter aspects of the host’s immune response, which differs in classic vs crusted scabies. In classic scabies, there is a predominance of CD4+ T cells in the dermis and minimal CD8+ T cells. The opposite is true in crusted scabies— there is an overwhelming infiltration of CD8+ T cells and minimal CD4+ T cells.3 The CD8+ T-cell predominance in crusted scabies is hypothesized to be the cause of keratinocyte apoptosis, resulting in epidermal hyperproliferation. Keratinocyte apoptosis also secretes cytokines, which may lead to the immunologic targeting of healthy skin cells. The damage of healthy dermal cells contributes to the inability of the skin’s immune system to mount an effective response, allowing the parasite to grow uncontrollably in patients with crusted scabies.4
This ineffective immune response is further exacerbated by corticosteroids, which are commonly prescribed for pruritus experienced by patients with scabies infestations. The mechanism of action of corticosteroids is the production of anti-inflammatory, antimitotic, and immunosuppressive effects.5 Because the integumentary immune system is imbalanced during crusted scabies infestation, the immunosuppressive mechanism of oral and topical corticosteroids further reduces the cellular immune response to scabies. The flourishing of the scabies mites along with keratinocyte apoptosis4 results in the development of hyperkeratotic skin crusting, most frequently on the palms, soles, arms, and legs. Risk factors for crusted scabies include immunosuppression, hospitalization, crowded living conditions, and poor hygiene, though no known risk factors were documented in up to 42% (33/78) of patients with crusted scabies in one study.6
Patients with crusted scabies typically present with generalized, poorly defined, erythematous, fissured plaques covered by scaling and crusts. Plaques on bony prominences such as finger articulations and elbows may have a thick verrucous aspect.1 Skin flaking that resembles brown sugar—a mixture of white sugar and molasses—is a clue to the diagnosis of crusted scabies. Brown sugar has a slightly sandy and sticky texture that ranges in color from very light brown to very dark brown. When present, flakes always appears slightly lighter than the patient’s skin tone. Although skin burrows are pathognomonic and clinically recognizable features of scabies, these burrows can be disguised by lesions, such as the hyperkeratotic plaques seen in our patient. The lesions may or may not be associated with pruritus, which may occur only at night, and bacterial superinfection has been reported in severe cases of crusted scabies,7 as scratching can cause sores, which may lead to infection. In severe cases, the constant scratching could lead to sepsis if the infection enters the bloodstream.8 Another symptom of scabies is a rash that causes small bumps that tend to form in a line, resembling small bites, hives, or pimples, and scaly plaques can lead to misdiagnosis as atopic dermatitis.
Treatment often is delayed due to misdiagnosis, as seen in our patient. Common misdiagnoses include atopic dermatitis, pityriasis rosea, systemic lupus erythematosus, bullous pemphigoid, lichen planus, pediculosis corporis, seborrheic scalp dermatitis, and adverse drug reactions.9 Patients with extensive infestations of crusted scabies should be treated with a 4-week course of permethrin cream 5% daily for 1 week, then twice per week until resolved, and oral ivermectin 200 μg/kg dosed 1 week apart for up to 4 weeks, if needed.1 Topical permethrin works by producing a selective neurotoxic effect on invertebrates such as scabies mites, which disrupts the function of voltage-gated sodium channels, thereby paralyzing the adult mites to halt the spread of infestation. However, treatment with topical medications can be difficult due to the thick crusts that have formed, which make it more challenging for the skin to properly absorb the treatment. Additionally, surgical debridement as an adjunct procedure has been done to improve the effectiveness of topical medications by removing all the mites in skin.10 On the other hand, the mechanism in which ivermectin treats scabies infestations is poorly understood. Current research suggests that ivermectin works by causing persistent opening of pH-gated chloride channels in scabies mites.11 There is emerging concern for drug resistance to these scabicides,12 revealing a need for further research of treatment options.
Patients with crusted scabies can have an extremely large number of mites (up to 2 million), making them more infectious than patients with classic scabies.13 As a result, it is imperative to reduce environmental transmission and risk for reinfection with mites during treatment. Because crusted scabies is transmitted by prolonged skinto- skin contact or by contact with personal items of an infected person (eg, bedding, clothing), treatment guidelines require all clothing, bedding, and towels of a patient with scabies to be machine-washed and dried with hot water and hot dryer cycles. If an item cannot be washed, it should be stored in a sealed plastic bag for 1 week, as scabies mites cannot survive more than 2 to 3 days away from their host of human skin.13 Treatment of close contacts of patients with scabies is recommended, as well as for those in endemic areas or closed communities, such as nursing homes or jails.
- Salavastru CM, Chosidow O, Boffa MJ, et al. European guideline for the management of scabies. J Eur Acad Dermatol Venereol. 2017;31:1248-1253. doi:10.1111/jdv.14351
- Morgan MS, Arlian LG, Markey MP. Sarcoptes scabiei mites modulate gene expression in human skin equivalents. PLoS One. 2013;8:e71143. doi:10.1371/journal.pone.0071143
- Walton SF, Beroukas D, Roberts-Thomson P, et al. New insights into disease pathogenesis in crusted (Norwegian) scabies: the skin immune response in crusted scabies. Br J Dermatol. 2008;158:1247-1255. doi:10.1111/j.1365-2133.2008.08541.x
- Bhat SA, Mounsey KE, Liu X, et al. Host immune responses to the itch mite, Sarcoptes scabiei, in humans. Parasit Vectors. 2017;10:385. doi:10.1186/s13071-017-2320-4
- Binic´ I, Jankovic´ A, Jovanovic´ D, et al. Crusted (Norwegian) scabies following systemic and topical corticosteroid therapy. J Korean Med Sci. 2009;25:188-191. doi:10.3346/jkms.2010.25.1.188
- Roberts LJ, Huffam SE, Walton SF, et al. Crusted scabies: clinical and immunological findings in seventy-eight patients and a review of the literature. J Infect. 2005;50:375-381. doi:10.1016/j.jinf.2004.08.033
- Yari N, Malone CH, Rivas A. Misdiagnosed crusted scabies in an AIDS patient leads to hyperinfestation. Cutis. 2017;99:202-204.
- American Academy of Dermatology Association. Scabies: signs and symptoms. Accessed July 12, 2024. https://www.aad.org/public/diseases/a-z/scabies-symptoms
- Siegfried EC, Hebert AA. Diagnosis of atopic dermatitis: mimics, overlaps, and complications. J Clin Med. 2015;4:884-917. doi:10.3390/jcm4050884
- Maghrabi MM, Lum S, Joba AT, et al. Norwegian crusted scabies: an unusual case presentation. J Foot Ankle Surg. 2014;53:62-66. doi:10.1053/j.jfas.2013.09.002
- Currie BJ, McCarthy JS. Permethrin and ivermectin for scabies. N Engl J Med. 2010;362:717-725. doi:10.1056/NEJMct0910329
- Andriantsoanirina V, Izri A, Botterel F, et al. Molecular survey of knockdown resistance to pyrethroids in human scabies mites. Clin Microbiol Infect. 2014;20:O139-O141. doi:10.1111/1469-0691.12334
- Centers for Disease Control and Prevention. Preventing scabies. Published December 18, 2023. Accessed August 9, 2024. https://www.cdc.gov/scabies/prevention/index.html
To the Editor:
Crusted scabies (formerly known as Norwegian scabies) is a rare and highly contagious variant of scabies, in which the skin is infested with thousands to millions of Sarcoptes scabiei var hominis mites. We present a case of skin changes that were misdiagnosed as atopic dermatitis, seborrhea, xerosis, and drug eruption on initial presentation, which prompted treatment with a corticosteroid that inadvertently caused progression to crusted scabies.
A 79-year-old woman who uses a wheelchair presented to the clinic with skin changes that consisted of diffuse, severely pruritic, erythematous plaques on the head, neck, trunk, face, and extremities of 2 years’ duration. She had a medical history of hyperlipidemia, hypertension, and hyperglycemia, as well as a stroke that required hospitalization 2 years prior to the onset of the skin changes. She had no history of allergies.
Prior clinical diagnoses by primary care and dermatology included xerosis, atopic dermatitis, seborrhea, and drug eruption. She was treated with a mid-potency topical corticosteroid (triamcinolone acetonide cream 0.1%) twice daily and prednisone 40 mg once daily for 2- to 4-week courses over an 8-month period without reduction in symptoms.
Physical examination at the current presentation revealed golden, crusted, fine, powdery but slightly sticky flakes that spread diffusely across the entire body and came off in crumbles with a simple touch. These widespread crusts were easily visible on clothing. There was underlying diffuse erythema beneath the flaking skin on the trunk and proximal extremities. The scale and shedding skin laid in piles on the patient’s lap and resembled brown sugar (Figure 1). The patient also reported decreased hand function and dexterity due to the yellowbrown, thick, crusty plaques that had developed on both the palmar and dorsal sides of the hands (Figure 2). Erythematous plaques on the scalp, forehead, and inner ears resembled seborrhea (Figure 3). Pruritus severity was rated by the patient as 10 of 10, and she scratched her skin the entire time she was in the clinic. The patient was emotional and stated that she had not been able to sleep due to the discomfort. We suspected scabies, and the patient was reassured to learn that it could be confirmed with a simple skin scrape test.



The crusted lesions on the patient's hands were scraped with a #15-blade scalpel, and a routine potassium hydroxide mount was performed. The skin scrapings were placed on a slide with a drop of 10% potassium hydroxide and observed under low-power (×10) and high-power (×40) microscopy, which revealed thousands of mites and eggs (along with previously hatched eggs) (Figure 4) and quickly confirmed a diagnosis of crusted scabies.an extremely contagious form of scabies seen in older patients with compromised immune systems, malnutrition, or disabilities. The patient was prescribed oral ivermectin (3 mg dosed at 200 μg/kg of body weight) and topical permethrin 5%, neither of which she took, as she died of a COVID-19 infection complication 3 days after this diagnostic clinic visit.

Classic and crusted scabies are both caused by infestation of the Sarcoptes scabiei var hominis mite. Classic scabies is a result of an infestation of a small number of mites (commonly 5–15 mites), while crusted scabies is due to hyperinfestation by as many as millions of mites, the latter often requiring more aggressive treatment. The mites are first transmitted to humans by either skin-toskin contact or fomites on bedding and clothing. The scabies mite undergoes 4 life cycle stages: egg, larvae, nymph, and adult. Once female mites are transmitted, they burrow under the skin and lay 2 to 3 eggs per day. The eggs hatch within 3 to 4 days, after which the larvae migrate to the skin surface. The larval stage lasts for 3 to 4 days, during which the larvae burrow into the stratum corneum to create molting pouches, until they molt into slightly larger nymphs. Nymphs can be found in hair follicles or molting pouches until they further molt within 3 to 4 days into adults, which are round, saclike mites. The adult male and female mites then mate, leaving the female fertile for the rest of her 1- to 2-month lifespan. Impregnated female mites traverse the skin surface in search of a burrow site, using the pulvilli on the anterior aspect of 2 legs to hold onto the skin. Once burrowed, the female mite continues to lay eggs for the rest of her life, with approximately 10% of her eggs resulting in adult mites. Male mites feed in shallow pits of the skin until they find a female burrow site for mating.1 This continuous life cycle of the scabies mite gives rise to highly transmissible, pruritic skin excoriations, as demonstrated in our patient.
The skin has a relatively late inflammatory and adaptive immune response to scabies, typically occurring 4 to 6 weeks after the initial infestation.2 This delayed inflammatory response and onset of symptoms may be due to the scabies mite’s ability to alter aspects of the host’s immune response, which differs in classic vs crusted scabies. In classic scabies, there is a predominance of CD4+ T cells in the dermis and minimal CD8+ T cells. The opposite is true in crusted scabies— there is an overwhelming infiltration of CD8+ T cells and minimal CD4+ T cells.3 The CD8+ T-cell predominance in crusted scabies is hypothesized to be the cause of keratinocyte apoptosis, resulting in epidermal hyperproliferation. Keratinocyte apoptosis also secretes cytokines, which may lead to the immunologic targeting of healthy skin cells. The damage of healthy dermal cells contributes to the inability of the skin’s immune system to mount an effective response, allowing the parasite to grow uncontrollably in patients with crusted scabies.4
This ineffective immune response is further exacerbated by corticosteroids, which are commonly prescribed for pruritus experienced by patients with scabies infestations. The mechanism of action of corticosteroids is the production of anti-inflammatory, antimitotic, and immunosuppressive effects.5 Because the integumentary immune system is imbalanced during crusted scabies infestation, the immunosuppressive mechanism of oral and topical corticosteroids further reduces the cellular immune response to scabies. The flourishing of the scabies mites along with keratinocyte apoptosis4 results in the development of hyperkeratotic skin crusting, most frequently on the palms, soles, arms, and legs. Risk factors for crusted scabies include immunosuppression, hospitalization, crowded living conditions, and poor hygiene, though no known risk factors were documented in up to 42% (33/78) of patients with crusted scabies in one study.6
Patients with crusted scabies typically present with generalized, poorly defined, erythematous, fissured plaques covered by scaling and crusts. Plaques on bony prominences such as finger articulations and elbows may have a thick verrucous aspect.1 Skin flaking that resembles brown sugar—a mixture of white sugar and molasses—is a clue to the diagnosis of crusted scabies. Brown sugar has a slightly sandy and sticky texture that ranges in color from very light brown to very dark brown. When present, flakes always appears slightly lighter than the patient’s skin tone. Although skin burrows are pathognomonic and clinically recognizable features of scabies, these burrows can be disguised by lesions, such as the hyperkeratotic plaques seen in our patient. The lesions may or may not be associated with pruritus, which may occur only at night, and bacterial superinfection has been reported in severe cases of crusted scabies,7 as scratching can cause sores, which may lead to infection. In severe cases, the constant scratching could lead to sepsis if the infection enters the bloodstream.8 Another symptom of scabies is a rash that causes small bumps that tend to form in a line, resembling small bites, hives, or pimples, and scaly plaques can lead to misdiagnosis as atopic dermatitis.
Treatment often is delayed due to misdiagnosis, as seen in our patient. Common misdiagnoses include atopic dermatitis, pityriasis rosea, systemic lupus erythematosus, bullous pemphigoid, lichen planus, pediculosis corporis, seborrheic scalp dermatitis, and adverse drug reactions.9 Patients with extensive infestations of crusted scabies should be treated with a 4-week course of permethrin cream 5% daily for 1 week, then twice per week until resolved, and oral ivermectin 200 μg/kg dosed 1 week apart for up to 4 weeks, if needed.1 Topical permethrin works by producing a selective neurotoxic effect on invertebrates such as scabies mites, which disrupts the function of voltage-gated sodium channels, thereby paralyzing the adult mites to halt the spread of infestation. However, treatment with topical medications can be difficult due to the thick crusts that have formed, which make it more challenging for the skin to properly absorb the treatment. Additionally, surgical debridement as an adjunct procedure has been done to improve the effectiveness of topical medications by removing all the mites in skin.10 On the other hand, the mechanism in which ivermectin treats scabies infestations is poorly understood. Current research suggests that ivermectin works by causing persistent opening of pH-gated chloride channels in scabies mites.11 There is emerging concern for drug resistance to these scabicides,12 revealing a need for further research of treatment options.
Patients with crusted scabies can have an extremely large number of mites (up to 2 million), making them more infectious than patients with classic scabies.13 As a result, it is imperative to reduce environmental transmission and risk for reinfection with mites during treatment. Because crusted scabies is transmitted by prolonged skinto- skin contact or by contact with personal items of an infected person (eg, bedding, clothing), treatment guidelines require all clothing, bedding, and towels of a patient with scabies to be machine-washed and dried with hot water and hot dryer cycles. If an item cannot be washed, it should be stored in a sealed plastic bag for 1 week, as scabies mites cannot survive more than 2 to 3 days away from their host of human skin.13 Treatment of close contacts of patients with scabies is recommended, as well as for those in endemic areas or closed communities, such as nursing homes or jails.
To the Editor:
Crusted scabies (formerly known as Norwegian scabies) is a rare and highly contagious variant of scabies, in which the skin is infested with thousands to millions of Sarcoptes scabiei var hominis mites. We present a case of skin changes that were misdiagnosed as atopic dermatitis, seborrhea, xerosis, and drug eruption on initial presentation, which prompted treatment with a corticosteroid that inadvertently caused progression to crusted scabies.
A 79-year-old woman who uses a wheelchair presented to the clinic with skin changes that consisted of diffuse, severely pruritic, erythematous plaques on the head, neck, trunk, face, and extremities of 2 years’ duration. She had a medical history of hyperlipidemia, hypertension, and hyperglycemia, as well as a stroke that required hospitalization 2 years prior to the onset of the skin changes. She had no history of allergies.
Prior clinical diagnoses by primary care and dermatology included xerosis, atopic dermatitis, seborrhea, and drug eruption. She was treated with a mid-potency topical corticosteroid (triamcinolone acetonide cream 0.1%) twice daily and prednisone 40 mg once daily for 2- to 4-week courses over an 8-month period without reduction in symptoms.
Physical examination at the current presentation revealed golden, crusted, fine, powdery but slightly sticky flakes that spread diffusely across the entire body and came off in crumbles with a simple touch. These widespread crusts were easily visible on clothing. There was underlying diffuse erythema beneath the flaking skin on the trunk and proximal extremities. The scale and shedding skin laid in piles on the patient’s lap and resembled brown sugar (Figure 1). The patient also reported decreased hand function and dexterity due to the yellowbrown, thick, crusty plaques that had developed on both the palmar and dorsal sides of the hands (Figure 2). Erythematous plaques on the scalp, forehead, and inner ears resembled seborrhea (Figure 3). Pruritus severity was rated by the patient as 10 of 10, and she scratched her skin the entire time she was in the clinic. The patient was emotional and stated that she had not been able to sleep due to the discomfort. We suspected scabies, and the patient was reassured to learn that it could be confirmed with a simple skin scrape test.



The crusted lesions on the patient's hands were scraped with a #15-blade scalpel, and a routine potassium hydroxide mount was performed. The skin scrapings were placed on a slide with a drop of 10% potassium hydroxide and observed under low-power (×10) and high-power (×40) microscopy, which revealed thousands of mites and eggs (along with previously hatched eggs) (Figure 4) and quickly confirmed a diagnosis of crusted scabies.an extremely contagious form of scabies seen in older patients with compromised immune systems, malnutrition, or disabilities. The patient was prescribed oral ivermectin (3 mg dosed at 200 μg/kg of body weight) and topical permethrin 5%, neither of which she took, as she died of a COVID-19 infection complication 3 days after this diagnostic clinic visit.

Classic and crusted scabies are both caused by infestation of the Sarcoptes scabiei var hominis mite. Classic scabies is a result of an infestation of a small number of mites (commonly 5–15 mites), while crusted scabies is due to hyperinfestation by as many as millions of mites, the latter often requiring more aggressive treatment. The mites are first transmitted to humans by either skin-toskin contact or fomites on bedding and clothing. The scabies mite undergoes 4 life cycle stages: egg, larvae, nymph, and adult. Once female mites are transmitted, they burrow under the skin and lay 2 to 3 eggs per day. The eggs hatch within 3 to 4 days, after which the larvae migrate to the skin surface. The larval stage lasts for 3 to 4 days, during which the larvae burrow into the stratum corneum to create molting pouches, until they molt into slightly larger nymphs. Nymphs can be found in hair follicles or molting pouches until they further molt within 3 to 4 days into adults, which are round, saclike mites. The adult male and female mites then mate, leaving the female fertile for the rest of her 1- to 2-month lifespan. Impregnated female mites traverse the skin surface in search of a burrow site, using the pulvilli on the anterior aspect of 2 legs to hold onto the skin. Once burrowed, the female mite continues to lay eggs for the rest of her life, with approximately 10% of her eggs resulting in adult mites. Male mites feed in shallow pits of the skin until they find a female burrow site for mating.1 This continuous life cycle of the scabies mite gives rise to highly transmissible, pruritic skin excoriations, as demonstrated in our patient.
The skin has a relatively late inflammatory and adaptive immune response to scabies, typically occurring 4 to 6 weeks after the initial infestation.2 This delayed inflammatory response and onset of symptoms may be due to the scabies mite’s ability to alter aspects of the host’s immune response, which differs in classic vs crusted scabies. In classic scabies, there is a predominance of CD4+ T cells in the dermis and minimal CD8+ T cells. The opposite is true in crusted scabies— there is an overwhelming infiltration of CD8+ T cells and minimal CD4+ T cells.3 The CD8+ T-cell predominance in crusted scabies is hypothesized to be the cause of keratinocyte apoptosis, resulting in epidermal hyperproliferation. Keratinocyte apoptosis also secretes cytokines, which may lead to the immunologic targeting of healthy skin cells. The damage of healthy dermal cells contributes to the inability of the skin’s immune system to mount an effective response, allowing the parasite to grow uncontrollably in patients with crusted scabies.4
This ineffective immune response is further exacerbated by corticosteroids, which are commonly prescribed for pruritus experienced by patients with scabies infestations. The mechanism of action of corticosteroids is the production of anti-inflammatory, antimitotic, and immunosuppressive effects.5 Because the integumentary immune system is imbalanced during crusted scabies infestation, the immunosuppressive mechanism of oral and topical corticosteroids further reduces the cellular immune response to scabies. The flourishing of the scabies mites along with keratinocyte apoptosis4 results in the development of hyperkeratotic skin crusting, most frequently on the palms, soles, arms, and legs. Risk factors for crusted scabies include immunosuppression, hospitalization, crowded living conditions, and poor hygiene, though no known risk factors were documented in up to 42% (33/78) of patients with crusted scabies in one study.6
Patients with crusted scabies typically present with generalized, poorly defined, erythematous, fissured plaques covered by scaling and crusts. Plaques on bony prominences such as finger articulations and elbows may have a thick verrucous aspect.1 Skin flaking that resembles brown sugar—a mixture of white sugar and molasses—is a clue to the diagnosis of crusted scabies. Brown sugar has a slightly sandy and sticky texture that ranges in color from very light brown to very dark brown. When present, flakes always appears slightly lighter than the patient’s skin tone. Although skin burrows are pathognomonic and clinically recognizable features of scabies, these burrows can be disguised by lesions, such as the hyperkeratotic plaques seen in our patient. The lesions may or may not be associated with pruritus, which may occur only at night, and bacterial superinfection has been reported in severe cases of crusted scabies,7 as scratching can cause sores, which may lead to infection. In severe cases, the constant scratching could lead to sepsis if the infection enters the bloodstream.8 Another symptom of scabies is a rash that causes small bumps that tend to form in a line, resembling small bites, hives, or pimples, and scaly plaques can lead to misdiagnosis as atopic dermatitis.
Treatment often is delayed due to misdiagnosis, as seen in our patient. Common misdiagnoses include atopic dermatitis, pityriasis rosea, systemic lupus erythematosus, bullous pemphigoid, lichen planus, pediculosis corporis, seborrheic scalp dermatitis, and adverse drug reactions.9 Patients with extensive infestations of crusted scabies should be treated with a 4-week course of permethrin cream 5% daily for 1 week, then twice per week until resolved, and oral ivermectin 200 μg/kg dosed 1 week apart for up to 4 weeks, if needed.1 Topical permethrin works by producing a selective neurotoxic effect on invertebrates such as scabies mites, which disrupts the function of voltage-gated sodium channels, thereby paralyzing the adult mites to halt the spread of infestation. However, treatment with topical medications can be difficult due to the thick crusts that have formed, which make it more challenging for the skin to properly absorb the treatment. Additionally, surgical debridement as an adjunct procedure has been done to improve the effectiveness of topical medications by removing all the mites in skin.10 On the other hand, the mechanism in which ivermectin treats scabies infestations is poorly understood. Current research suggests that ivermectin works by causing persistent opening of pH-gated chloride channels in scabies mites.11 There is emerging concern for drug resistance to these scabicides,12 revealing a need for further research of treatment options.
Patients with crusted scabies can have an extremely large number of mites (up to 2 million), making them more infectious than patients with classic scabies.13 As a result, it is imperative to reduce environmental transmission and risk for reinfection with mites during treatment. Because crusted scabies is transmitted by prolonged skinto- skin contact or by contact with personal items of an infected person (eg, bedding, clothing), treatment guidelines require all clothing, bedding, and towels of a patient with scabies to be machine-washed and dried with hot water and hot dryer cycles. If an item cannot be washed, it should be stored in a sealed plastic bag for 1 week, as scabies mites cannot survive more than 2 to 3 days away from their host of human skin.13 Treatment of close contacts of patients with scabies is recommended, as well as for those in endemic areas or closed communities, such as nursing homes or jails.
- Salavastru CM, Chosidow O, Boffa MJ, et al. European guideline for the management of scabies. J Eur Acad Dermatol Venereol. 2017;31:1248-1253. doi:10.1111/jdv.14351
- Morgan MS, Arlian LG, Markey MP. Sarcoptes scabiei mites modulate gene expression in human skin equivalents. PLoS One. 2013;8:e71143. doi:10.1371/journal.pone.0071143
- Walton SF, Beroukas D, Roberts-Thomson P, et al. New insights into disease pathogenesis in crusted (Norwegian) scabies: the skin immune response in crusted scabies. Br J Dermatol. 2008;158:1247-1255. doi:10.1111/j.1365-2133.2008.08541.x
- Bhat SA, Mounsey KE, Liu X, et al. Host immune responses to the itch mite, Sarcoptes scabiei, in humans. Parasit Vectors. 2017;10:385. doi:10.1186/s13071-017-2320-4
- Binic´ I, Jankovic´ A, Jovanovic´ D, et al. Crusted (Norwegian) scabies following systemic and topical corticosteroid therapy. J Korean Med Sci. 2009;25:188-191. doi:10.3346/jkms.2010.25.1.188
- Roberts LJ, Huffam SE, Walton SF, et al. Crusted scabies: clinical and immunological findings in seventy-eight patients and a review of the literature. J Infect. 2005;50:375-381. doi:10.1016/j.jinf.2004.08.033
- Yari N, Malone CH, Rivas A. Misdiagnosed crusted scabies in an AIDS patient leads to hyperinfestation. Cutis. 2017;99:202-204.
- American Academy of Dermatology Association. Scabies: signs and symptoms. Accessed July 12, 2024. https://www.aad.org/public/diseases/a-z/scabies-symptoms
- Siegfried EC, Hebert AA. Diagnosis of atopic dermatitis: mimics, overlaps, and complications. J Clin Med. 2015;4:884-917. doi:10.3390/jcm4050884
- Maghrabi MM, Lum S, Joba AT, et al. Norwegian crusted scabies: an unusual case presentation. J Foot Ankle Surg. 2014;53:62-66. doi:10.1053/j.jfas.2013.09.002
- Currie BJ, McCarthy JS. Permethrin and ivermectin for scabies. N Engl J Med. 2010;362:717-725. doi:10.1056/NEJMct0910329
- Andriantsoanirina V, Izri A, Botterel F, et al. Molecular survey of knockdown resistance to pyrethroids in human scabies mites. Clin Microbiol Infect. 2014;20:O139-O141. doi:10.1111/1469-0691.12334
- Centers for Disease Control and Prevention. Preventing scabies. Published December 18, 2023. Accessed August 9, 2024. https://www.cdc.gov/scabies/prevention/index.html
- Salavastru CM, Chosidow O, Boffa MJ, et al. European guideline for the management of scabies. J Eur Acad Dermatol Venereol. 2017;31:1248-1253. doi:10.1111/jdv.14351
- Morgan MS, Arlian LG, Markey MP. Sarcoptes scabiei mites modulate gene expression in human skin equivalents. PLoS One. 2013;8:e71143. doi:10.1371/journal.pone.0071143
- Walton SF, Beroukas D, Roberts-Thomson P, et al. New insights into disease pathogenesis in crusted (Norwegian) scabies: the skin immune response in crusted scabies. Br J Dermatol. 2008;158:1247-1255. doi:10.1111/j.1365-2133.2008.08541.x
- Bhat SA, Mounsey KE, Liu X, et al. Host immune responses to the itch mite, Sarcoptes scabiei, in humans. Parasit Vectors. 2017;10:385. doi:10.1186/s13071-017-2320-4
- Binic´ I, Jankovic´ A, Jovanovic´ D, et al. Crusted (Norwegian) scabies following systemic and topical corticosteroid therapy. J Korean Med Sci. 2009;25:188-191. doi:10.3346/jkms.2010.25.1.188
- Roberts LJ, Huffam SE, Walton SF, et al. Crusted scabies: clinical and immunological findings in seventy-eight patients and a review of the literature. J Infect. 2005;50:375-381. doi:10.1016/j.jinf.2004.08.033
- Yari N, Malone CH, Rivas A. Misdiagnosed crusted scabies in an AIDS patient leads to hyperinfestation. Cutis. 2017;99:202-204.
- American Academy of Dermatology Association. Scabies: signs and symptoms. Accessed July 12, 2024. https://www.aad.org/public/diseases/a-z/scabies-symptoms
- Siegfried EC, Hebert AA. Diagnosis of atopic dermatitis: mimics, overlaps, and complications. J Clin Med. 2015;4:884-917. doi:10.3390/jcm4050884
- Maghrabi MM, Lum S, Joba AT, et al. Norwegian crusted scabies: an unusual case presentation. J Foot Ankle Surg. 2014;53:62-66. doi:10.1053/j.jfas.2013.09.002
- Currie BJ, McCarthy JS. Permethrin and ivermectin for scabies. N Engl J Med. 2010;362:717-725. doi:10.1056/NEJMct0910329
- Andriantsoanirina V, Izri A, Botterel F, et al. Molecular survey of knockdown resistance to pyrethroids in human scabies mites. Clin Microbiol Infect. 2014;20:O139-O141. doi:10.1111/1469-0691.12334
- Centers for Disease Control and Prevention. Preventing scabies. Published December 18, 2023. Accessed August 9, 2024. https://www.cdc.gov/scabies/prevention/index.html
PRACTICE POINTS
- Crusted scabies often is misdiagnosed because it mimics common dermatologic conditions, such as atopic dermatitis, psoriasis, drug eruption, and seborrhea. A unique feature of crusted scabies is fine or coarse scaling that resembles brown sugar.
- Immunosuppressants, such as topical corticosteroids, worsen the skin’s immune response to classic scabies infestations, which leads to parasitic overgrowth and the development of crusted scabies.
- Treatment of crusted scabies requires topical and oral scabicide; in addition, all clothing, bedding, and towels should be machine-washed and dried with hot water and hot dryer cycles to prevent environmental transmission and reinfection.
Recurrent Pancreatitis Triples Risk for Chronic Disease
TOPLINE:
The overall progression to chronic pancreatitis among adults was three times higher following recurrent episodes of acute pancreatitis than occurring after just the first acute pancreatitis episode.
METHODOLOGY:
- The progression of acute pancreatitis is time-dependent, with the recurrence and progression rates to recurrent acute pancreatitis and chronic pancreatitis varying based on the follow-up duration and may be affected by the cause and severity of the first acute episode.
- To better understand the progression of acute pancreatitis to recurrent acute pancreatitis and chronic pancreatitis, researchers conducted a systematic review and meta-analysis of 119 studies, all of which were used for qualitative and quantitative synthesis and 29 of which also were used for calculating incidence rates.
- The primary outcomes were the incidence rates of recurrent acute and chronic pancreatitis following the initial episode of acute pancreatitis and the incidence rate of chronic pancreatitis after recurrent episodes of acute pancreatitis.
- The secondary outcomes were the cumulative incidences and proportions of recurrent acute and chronic pancreatitis following the initial acute pancreatitis episode and the proportion of chronic pancreatitis occurring after recurrent acute pancreatitis episodes.
TAKEAWAY:
- The incidence rate of recurrent acute pancreatitis after the first acute episode was 5.26 per 100 person-years in adults and 4.64 per 100 person-years in children, a difference that did not reach statistical significance.
- The progression rate to chronic pancreatitis in adults was threefold higher after recurrent acute pancreatitis episodes than after the first acute pancreatitis episode (4.31 vs 1.38 per 100 person-years).
- Hypertriglyceridemia-induced acute pancreatitis had the highest recurrence rates, followed by alcohol-induced, idiopathic, and biliary pancreatitis.
- The overall progression rate into chronic pancreatitis was 8% after the first acute pancreatitis episode and 24% after recurrent episodes of acute pancreatitis. Progression to chronic pancreatitis among adults was highest among those with alcohol-induced disease, followed by idiopathic and biliary pancreatitis.
- A moderately severe first episode of acute pancreatitis was associated with the highest recurrence rate, followed by mild and severe first episodes.
IN PRACTICE:
The authors emphasized the need to develop new interventions to address the factors associated with acute pancreatitis and its progression and to better utilize existing approaches, such as brief and repeated psychological interventions and alcohol and smoking cessation programs. Deeper investigation into the underlying causes of the disease’s etiology is warranted to reduce recurrence and progression rates, they noted.
SOURCE:
The study, led by Endre-Botond Gagyi, MD, of the Center for Translational Medicine, Semmelweis University, Budapest, Hungary, was published online in Therapeutic Advances in Gastroenterology.
LIMITATIONS:
Most of the studies included in the analysis were retrospective, and there was high heterogeneity between them. The researchers could only analyze the presence of recurrent acute pancreatitis but could not explore the number of episodes or their impact on progression due to the lack of reported data.
DISCLOSURES:
The study was funded by the New National Excellence Program of the Ministry for Innovation and Technology from the National Research, Development and Innovation Fund. The authors declared no conflict of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
The overall progression to chronic pancreatitis among adults was three times higher following recurrent episodes of acute pancreatitis than occurring after just the first acute pancreatitis episode.
METHODOLOGY:
- The progression of acute pancreatitis is time-dependent, with the recurrence and progression rates to recurrent acute pancreatitis and chronic pancreatitis varying based on the follow-up duration and may be affected by the cause and severity of the first acute episode.
- To better understand the progression of acute pancreatitis to recurrent acute pancreatitis and chronic pancreatitis, researchers conducted a systematic review and meta-analysis of 119 studies, all of which were used for qualitative and quantitative synthesis and 29 of which also were used for calculating incidence rates.
- The primary outcomes were the incidence rates of recurrent acute and chronic pancreatitis following the initial episode of acute pancreatitis and the incidence rate of chronic pancreatitis after recurrent episodes of acute pancreatitis.
- The secondary outcomes were the cumulative incidences and proportions of recurrent acute and chronic pancreatitis following the initial acute pancreatitis episode and the proportion of chronic pancreatitis occurring after recurrent acute pancreatitis episodes.
TAKEAWAY:
- The incidence rate of recurrent acute pancreatitis after the first acute episode was 5.26 per 100 person-years in adults and 4.64 per 100 person-years in children, a difference that did not reach statistical significance.
- The progression rate to chronic pancreatitis in adults was threefold higher after recurrent acute pancreatitis episodes than after the first acute pancreatitis episode (4.31 vs 1.38 per 100 person-years).
- Hypertriglyceridemia-induced acute pancreatitis had the highest recurrence rates, followed by alcohol-induced, idiopathic, and biliary pancreatitis.
- The overall progression rate into chronic pancreatitis was 8% after the first acute pancreatitis episode and 24% after recurrent episodes of acute pancreatitis. Progression to chronic pancreatitis among adults was highest among those with alcohol-induced disease, followed by idiopathic and biliary pancreatitis.
- A moderately severe first episode of acute pancreatitis was associated with the highest recurrence rate, followed by mild and severe first episodes.
IN PRACTICE:
The authors emphasized the need to develop new interventions to address the factors associated with acute pancreatitis and its progression and to better utilize existing approaches, such as brief and repeated psychological interventions and alcohol and smoking cessation programs. Deeper investigation into the underlying causes of the disease’s etiology is warranted to reduce recurrence and progression rates, they noted.
SOURCE:
The study, led by Endre-Botond Gagyi, MD, of the Center for Translational Medicine, Semmelweis University, Budapest, Hungary, was published online in Therapeutic Advances in Gastroenterology.
LIMITATIONS:
Most of the studies included in the analysis were retrospective, and there was high heterogeneity between them. The researchers could only analyze the presence of recurrent acute pancreatitis but could not explore the number of episodes or their impact on progression due to the lack of reported data.
DISCLOSURES:
The study was funded by the New National Excellence Program of the Ministry for Innovation and Technology from the National Research, Development and Innovation Fund. The authors declared no conflict of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
The overall progression to chronic pancreatitis among adults was three times higher following recurrent episodes of acute pancreatitis than occurring after just the first acute pancreatitis episode.
METHODOLOGY:
- The progression of acute pancreatitis is time-dependent, with the recurrence and progression rates to recurrent acute pancreatitis and chronic pancreatitis varying based on the follow-up duration and may be affected by the cause and severity of the first acute episode.
- To better understand the progression of acute pancreatitis to recurrent acute pancreatitis and chronic pancreatitis, researchers conducted a systematic review and meta-analysis of 119 studies, all of which were used for qualitative and quantitative synthesis and 29 of which also were used for calculating incidence rates.
- The primary outcomes were the incidence rates of recurrent acute and chronic pancreatitis following the initial episode of acute pancreatitis and the incidence rate of chronic pancreatitis after recurrent episodes of acute pancreatitis.
- The secondary outcomes were the cumulative incidences and proportions of recurrent acute and chronic pancreatitis following the initial acute pancreatitis episode and the proportion of chronic pancreatitis occurring after recurrent acute pancreatitis episodes.
TAKEAWAY:
- The incidence rate of recurrent acute pancreatitis after the first acute episode was 5.26 per 100 person-years in adults and 4.64 per 100 person-years in children, a difference that did not reach statistical significance.
- The progression rate to chronic pancreatitis in adults was threefold higher after recurrent acute pancreatitis episodes than after the first acute pancreatitis episode (4.31 vs 1.38 per 100 person-years).
- Hypertriglyceridemia-induced acute pancreatitis had the highest recurrence rates, followed by alcohol-induced, idiopathic, and biliary pancreatitis.
- The overall progression rate into chronic pancreatitis was 8% after the first acute pancreatitis episode and 24% after recurrent episodes of acute pancreatitis. Progression to chronic pancreatitis among adults was highest among those with alcohol-induced disease, followed by idiopathic and biliary pancreatitis.
- A moderately severe first episode of acute pancreatitis was associated with the highest recurrence rate, followed by mild and severe first episodes.
IN PRACTICE:
The authors emphasized the need to develop new interventions to address the factors associated with acute pancreatitis and its progression and to better utilize existing approaches, such as brief and repeated psychological interventions and alcohol and smoking cessation programs. Deeper investigation into the underlying causes of the disease’s etiology is warranted to reduce recurrence and progression rates, they noted.
SOURCE:
The study, led by Endre-Botond Gagyi, MD, of the Center for Translational Medicine, Semmelweis University, Budapest, Hungary, was published online in Therapeutic Advances in Gastroenterology.
LIMITATIONS:
Most of the studies included in the analysis were retrospective, and there was high heterogeneity between them. The researchers could only analyze the presence of recurrent acute pancreatitis but could not explore the number of episodes or their impact on progression due to the lack of reported data.
DISCLOSURES:
The study was funded by the New National Excellence Program of the Ministry for Innovation and Technology from the National Research, Development and Innovation Fund. The authors declared no conflict of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Beers Criteria Update for Inappropriate Medication Use in Older Adults
Primary care physicians know the complexities of treating older patients, from increased complications from medications and procedures to comorbidities stemming from having multiple medical conditions. The Beers Criteria were established by the American Geriatrics Society as a guide for physicians about medications that may possess more risks than benefits in older patients, specifically those aged 65 years and older.
There are approximately 100 medications on the list. Criteria used to establish the list include medications to avoid over the age of 65 in an outpatient setting, medications to avoid in certain medical conditions, medications to avoid that may interact with other medications, medications to avoid with renal impairment, and medications to avoid where harmful side effects outweigh the possible benefits. The American Geriatrics Society updates the list as new published evidence becomes available.
The latest updates to the Beers Criteria include several medications commonly used in primary care. Regarding anticoagulation, warfarin should be avoided as initial therapy and apixaban should be used in patients with reduced renal function. These guidelines looked particularly at antithrombotic medications because of new evidence arising in nonvalvular atrial fibrillation and venous thromboembolism. In addition to the previous recommendations, the use of aspirin is no longer recommended in older adults.
The latest guidelines also make recommendations regarding certain diabetic medications as well as combinations to avoid. The Beers Criteria now place all sulfonylureas in the class to avoid, and not just the long-acting formulations as was recommended in the previous guidelines. If a sulfonylurea is necessary, use of a short-acting one is advised. Several other classes of medications were addressed and doctors practicing primary care medicine should be aware of these guidelines, especially as the population continues to age.
Overall, these guidelines are a great resource for treating patients aged 65 and older. It is important to keep in mind that they look at a whole population of patients and it is not patient specific. As primary care doctors, we know many of our patients don’t fit into the textbook box. While these guidelines consider the dangers of a certain medication, sometimes the benefits do outweigh the risks at the patient-specific level.
As doctors, we are trained to weigh the risks and benefits when prescribing any medication to our patients. These guidelines shouldn’t be approached as a do or don’t list but should be considered in the overall plan when prescribing for our patients. Sometimes, these medications can be used with careful observation by the prescribing physician. When they are utilized, we need to make the patient aware of specific side effects and what to watch out for. We need to make these decisions together with our patients and their caregivers.
For example, we all know how agonizing taking care of an older dementia patient can be, and sometimes there is nothing left to try except one of the medications on the list.
An additional practical point not considered in the guidelines is real-world use. Often, certain medications are not covered by a patient’s insurance company. The cost can be prohibitive to use the recommended agent. We are left in the middle to go off script with a medication that the patient may be able to access easily or keep pushing for the most appropriate medication for the patient. Unfortunately, in our current healthcare climate, prior authorizations can sometimes take weeks to obtain (or to be denied). For most of the conditions we treat in our older patients, it is not safe to leave them without any medication while we fight this prior authorizations war.
Our older patients often have multiple specialists as well. Each of these specialists may be prescribing different medications. It is imperative that we know all the medications a patient is taking so that we may look for potentially dangerous drug interactions. Many patients don’t remember the names of all their medications, nor do they realize that many classes of medications are “little white pills.” Asking them to bring their pill bottles to every visit can be a great help in searching out interactions.
That being said, the Beers Criteria do an excellent job reviewing the latest evidence and developing guidelines. As primary care physicians, we have never been busier and having someone do the research and set it forth so clearly is a great tool. We should be aware of the Beers Criteria and the medications and interactions listed there.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no conflicts of interest.
Primary care physicians know the complexities of treating older patients, from increased complications from medications and procedures to comorbidities stemming from having multiple medical conditions. The Beers Criteria were established by the American Geriatrics Society as a guide for physicians about medications that may possess more risks than benefits in older patients, specifically those aged 65 years and older.
There are approximately 100 medications on the list. Criteria used to establish the list include medications to avoid over the age of 65 in an outpatient setting, medications to avoid in certain medical conditions, medications to avoid that may interact with other medications, medications to avoid with renal impairment, and medications to avoid where harmful side effects outweigh the possible benefits. The American Geriatrics Society updates the list as new published evidence becomes available.
The latest updates to the Beers Criteria include several medications commonly used in primary care. Regarding anticoagulation, warfarin should be avoided as initial therapy and apixaban should be used in patients with reduced renal function. These guidelines looked particularly at antithrombotic medications because of new evidence arising in nonvalvular atrial fibrillation and venous thromboembolism. In addition to the previous recommendations, the use of aspirin is no longer recommended in older adults.
The latest guidelines also make recommendations regarding certain diabetic medications as well as combinations to avoid. The Beers Criteria now place all sulfonylureas in the class to avoid, and not just the long-acting formulations as was recommended in the previous guidelines. If a sulfonylurea is necessary, use of a short-acting one is advised. Several other classes of medications were addressed and doctors practicing primary care medicine should be aware of these guidelines, especially as the population continues to age.
Overall, these guidelines are a great resource for treating patients aged 65 and older. It is important to keep in mind that they look at a whole population of patients and it is not patient specific. As primary care doctors, we know many of our patients don’t fit into the textbook box. While these guidelines consider the dangers of a certain medication, sometimes the benefits do outweigh the risks at the patient-specific level.
As doctors, we are trained to weigh the risks and benefits when prescribing any medication to our patients. These guidelines shouldn’t be approached as a do or don’t list but should be considered in the overall plan when prescribing for our patients. Sometimes, these medications can be used with careful observation by the prescribing physician. When they are utilized, we need to make the patient aware of specific side effects and what to watch out for. We need to make these decisions together with our patients and their caregivers.
For example, we all know how agonizing taking care of an older dementia patient can be, and sometimes there is nothing left to try except one of the medications on the list.
An additional practical point not considered in the guidelines is real-world use. Often, certain medications are not covered by a patient’s insurance company. The cost can be prohibitive to use the recommended agent. We are left in the middle to go off script with a medication that the patient may be able to access easily or keep pushing for the most appropriate medication for the patient. Unfortunately, in our current healthcare climate, prior authorizations can sometimes take weeks to obtain (or to be denied). For most of the conditions we treat in our older patients, it is not safe to leave them without any medication while we fight this prior authorizations war.
Our older patients often have multiple specialists as well. Each of these specialists may be prescribing different medications. It is imperative that we know all the medications a patient is taking so that we may look for potentially dangerous drug interactions. Many patients don’t remember the names of all their medications, nor do they realize that many classes of medications are “little white pills.” Asking them to bring their pill bottles to every visit can be a great help in searching out interactions.
That being said, the Beers Criteria do an excellent job reviewing the latest evidence and developing guidelines. As primary care physicians, we have never been busier and having someone do the research and set it forth so clearly is a great tool. We should be aware of the Beers Criteria and the medications and interactions listed there.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no conflicts of interest.
Primary care physicians know the complexities of treating older patients, from increased complications from medications and procedures to comorbidities stemming from having multiple medical conditions. The Beers Criteria were established by the American Geriatrics Society as a guide for physicians about medications that may possess more risks than benefits in older patients, specifically those aged 65 years and older.
There are approximately 100 medications on the list. Criteria used to establish the list include medications to avoid over the age of 65 in an outpatient setting, medications to avoid in certain medical conditions, medications to avoid that may interact with other medications, medications to avoid with renal impairment, and medications to avoid where harmful side effects outweigh the possible benefits. The American Geriatrics Society updates the list as new published evidence becomes available.
The latest updates to the Beers Criteria include several medications commonly used in primary care. Regarding anticoagulation, warfarin should be avoided as initial therapy and apixaban should be used in patients with reduced renal function. These guidelines looked particularly at antithrombotic medications because of new evidence arising in nonvalvular atrial fibrillation and venous thromboembolism. In addition to the previous recommendations, the use of aspirin is no longer recommended in older adults.
The latest guidelines also make recommendations regarding certain diabetic medications as well as combinations to avoid. The Beers Criteria now place all sulfonylureas in the class to avoid, and not just the long-acting formulations as was recommended in the previous guidelines. If a sulfonylurea is necessary, use of a short-acting one is advised. Several other classes of medications were addressed and doctors practicing primary care medicine should be aware of these guidelines, especially as the population continues to age.
Overall, these guidelines are a great resource for treating patients aged 65 and older. It is important to keep in mind that they look at a whole population of patients and it is not patient specific. As primary care doctors, we know many of our patients don’t fit into the textbook box. While these guidelines consider the dangers of a certain medication, sometimes the benefits do outweigh the risks at the patient-specific level.
As doctors, we are trained to weigh the risks and benefits when prescribing any medication to our patients. These guidelines shouldn’t be approached as a do or don’t list but should be considered in the overall plan when prescribing for our patients. Sometimes, these medications can be used with careful observation by the prescribing physician. When they are utilized, we need to make the patient aware of specific side effects and what to watch out for. We need to make these decisions together with our patients and their caregivers.
For example, we all know how agonizing taking care of an older dementia patient can be, and sometimes there is nothing left to try except one of the medications on the list.
An additional practical point not considered in the guidelines is real-world use. Often, certain medications are not covered by a patient’s insurance company. The cost can be prohibitive to use the recommended agent. We are left in the middle to go off script with a medication that the patient may be able to access easily or keep pushing for the most appropriate medication for the patient. Unfortunately, in our current healthcare climate, prior authorizations can sometimes take weeks to obtain (or to be denied). For most of the conditions we treat in our older patients, it is not safe to leave them without any medication while we fight this prior authorizations war.
Our older patients often have multiple specialists as well. Each of these specialists may be prescribing different medications. It is imperative that we know all the medications a patient is taking so that we may look for potentially dangerous drug interactions. Many patients don’t remember the names of all their medications, nor do they realize that many classes of medications are “little white pills.” Asking them to bring their pill bottles to every visit can be a great help in searching out interactions.
That being said, the Beers Criteria do an excellent job reviewing the latest evidence and developing guidelines. As primary care physicians, we have never been busier and having someone do the research and set it forth so clearly is a great tool. We should be aware of the Beers Criteria and the medications and interactions listed there.
Dr. Girgis practices family medicine in South River, New Jersey, and is a clinical assistant professor of family medicine at Robert Wood Johnson Medical School, New Brunswick, New Jersey. She has no conflicts of interest.
Is Vision Loss a New Dementia Risk Factor? What Do the Data Say?
In 2019, 57 million people worldwide were living with dementia, a figure expected to soar to 153 million by 2050. A recent Lancet Commission report suggests that nearly half of dementia cases could be prevented or delayed by addressing 14 modifiable risk factors, including impaired vision.
The report’s authors recommend that vision-loss screening and treatment be universally available. But are these recommendations warranted? What is the evidence? What is the potential mechanism? And what are the potential implications for clinical practice?
Worldwide, the prevalence of avoidable vision loss and blindness in adults aged 50 years or older is estimated to hover around 13%.
“There is now overwhelming evidence that vision impairment in later life is associated with more rapid cognitive decline and an increased risk of dementia,” said Joshua Ehrlich, MD, MPH, associate professor in ophthalmology and visual sciences, the Institute for Social Research at the University of Michigan, Ann Arbor.
The evidence includes a meta-analysis of 14 prospective cohort studies with roughly 6.2 million older adults who were cognitively intact at baseline. Over the course of up to 14 years, 171,888 developed dementia. Vision loss was associated with a pooled relative risk (RR) for dementia of 1.47.
A separate meta-analysis also identified an increased risk for dementia (RR, 1.38) with visual loss. When broken down into different eye conditions, an increased dementia risk was associated with cataracts and diabetic retinopathy but not with glaucoma or age-related macular degeneration.
A US study that followed roughly 3000 older adults with cataracts and normal cognition at baseline for more than 20 years found that those who had cataract extraction had significantly reduced risk for dementia compared with those who did not have cataract extraction (hazard ratio, 0.71), after controlling for age, race, APOE genotype, education, smoking, and an extensive list of comorbidities.
Causation or Coincidence?
The mechanisms behind these associations might be related to underlying illness, such as diabetes, which is a risk factor for dementia; vision loss itself, as might be suggested by a possible effect of cataract surgery; or shared neuropathologic processes in the retina and the brain.
A longitudinal study from Korea that included roughly 6 million adults showed that dementia risk increased with severity of visual loss, which supports the hypothesis that vision loss in itself might be causal or that there is a dose-response effect to a shared causal factor.
“Work is still needed to sort out” exactly how visual deficits may raise dementia risk, although several hypotheses exist, Dr. Ehrlich said.
For example, “decreased input to the brain via the visual pathways may directly induce brain changes. Also, consequences of vision loss, like social isolation, physical inactivity, and depression, are themselves risk factors for dementia and may explain the pathways through which vision impairment increases risk,” he said.
Is the link causal? “We’ll never know definitively because we can’t randomize people to not get cataract surgery versus getting cataract surgery, because we know that improving vision improves quality of life, so we’d never want to do that. But the new evidence that’s come in over the last 5 years or so is pretty promising,” said Esme Fuller-Thomson, PhD, director of the Institute for Life Course and Aging and professor, Department of Family and Community Medicine and Faculty of Nursing, at the University of Toronto, Ontario, Canada.
She noted that results of two studies that have looked at this “seem to indicate that those who have cataract surgery are not nearly at as high risk of dementia as those who have cataracts but don’t have the surgery. That’s leaning towards causality.”
A study published in July suggests that cataracts increase dementia risk through vascular and non–Alzheimer’s disease mechanisms.
Clear Clinical Implications
Dr. Ehrlich said that evidence for an association between untreated vision loss and dementia risk and potential modification by treatment has clear implications for care.
“Loss of vision impacts so many aspects of people’s lives beyond just how they see the world and losing vision in later life is not a normal part of aging. Thus, when older adults experience vision loss, this should be a cause for concern and prompt an immediate referral to an eye care professional,” he noted.
Dr. Fuller-Thomson agrees. “Addressing vision loss will certainly help people see better and function at a higher level and improve quality of life, and it seems probable that it might decrease dementia risk so it’s a win-win,” she said.
In her own research, Dr. Fuller-Thomson has found that the combination of hearing loss and vision loss is linked to an eightfold increased risk for cognitive impairment.
“The idea is that vision and/or hearing loss makes it harder for you to be physically active, to be socially engaged, to be mentally stimulated. They are equally important in terms of social isolation, which could lead to loneliness, and we know that loneliness is not good for dementia,” she said.
“With dual sensory impairment, you don’t have as much information coming in — your brain is not engaged as much — and having an engaged brain, doing hobbies, having intellectually stimulating conversation, all of those are factors are associated with lowering risk of dementia,” Dr. Fuller-Thomson said.
The latest Lancet Commission report noted that treatment for visual loss is “effective and cost-effective” for an estimated 90% of people. However, across the world, particularly in low- and middle-income countries, visual loss often goes untreated.
the report concluded.
Dr. Ehrlich and Dr. Fuller-Thomson have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
In 2019, 57 million people worldwide were living with dementia, a figure expected to soar to 153 million by 2050. A recent Lancet Commission report suggests that nearly half of dementia cases could be prevented or delayed by addressing 14 modifiable risk factors, including impaired vision.
The report’s authors recommend that vision-loss screening and treatment be universally available. But are these recommendations warranted? What is the evidence? What is the potential mechanism? And what are the potential implications for clinical practice?
Worldwide, the prevalence of avoidable vision loss and blindness in adults aged 50 years or older is estimated to hover around 13%.
“There is now overwhelming evidence that vision impairment in later life is associated with more rapid cognitive decline and an increased risk of dementia,” said Joshua Ehrlich, MD, MPH, associate professor in ophthalmology and visual sciences, the Institute for Social Research at the University of Michigan, Ann Arbor.
The evidence includes a meta-analysis of 14 prospective cohort studies with roughly 6.2 million older adults who were cognitively intact at baseline. Over the course of up to 14 years, 171,888 developed dementia. Vision loss was associated with a pooled relative risk (RR) for dementia of 1.47.
A separate meta-analysis also identified an increased risk for dementia (RR, 1.38) with visual loss. When broken down into different eye conditions, an increased dementia risk was associated with cataracts and diabetic retinopathy but not with glaucoma or age-related macular degeneration.
A US study that followed roughly 3000 older adults with cataracts and normal cognition at baseline for more than 20 years found that those who had cataract extraction had significantly reduced risk for dementia compared with those who did not have cataract extraction (hazard ratio, 0.71), after controlling for age, race, APOE genotype, education, smoking, and an extensive list of comorbidities.
Causation or Coincidence?
The mechanisms behind these associations might be related to underlying illness, such as diabetes, which is a risk factor for dementia; vision loss itself, as might be suggested by a possible effect of cataract surgery; or shared neuropathologic processes in the retina and the brain.
A longitudinal study from Korea that included roughly 6 million adults showed that dementia risk increased with severity of visual loss, which supports the hypothesis that vision loss in itself might be causal or that there is a dose-response effect to a shared causal factor.
“Work is still needed to sort out” exactly how visual deficits may raise dementia risk, although several hypotheses exist, Dr. Ehrlich said.
For example, “decreased input to the brain via the visual pathways may directly induce brain changes. Also, consequences of vision loss, like social isolation, physical inactivity, and depression, are themselves risk factors for dementia and may explain the pathways through which vision impairment increases risk,” he said.
Is the link causal? “We’ll never know definitively because we can’t randomize people to not get cataract surgery versus getting cataract surgery, because we know that improving vision improves quality of life, so we’d never want to do that. But the new evidence that’s come in over the last 5 years or so is pretty promising,” said Esme Fuller-Thomson, PhD, director of the Institute for Life Course and Aging and professor, Department of Family and Community Medicine and Faculty of Nursing, at the University of Toronto, Ontario, Canada.
She noted that results of two studies that have looked at this “seem to indicate that those who have cataract surgery are not nearly at as high risk of dementia as those who have cataracts but don’t have the surgery. That’s leaning towards causality.”
A study published in July suggests that cataracts increase dementia risk through vascular and non–Alzheimer’s disease mechanisms.
Clear Clinical Implications
Dr. Ehrlich said that evidence for an association between untreated vision loss and dementia risk and potential modification by treatment has clear implications for care.
“Loss of vision impacts so many aspects of people’s lives beyond just how they see the world and losing vision in later life is not a normal part of aging. Thus, when older adults experience vision loss, this should be a cause for concern and prompt an immediate referral to an eye care professional,” he noted.
Dr. Fuller-Thomson agrees. “Addressing vision loss will certainly help people see better and function at a higher level and improve quality of life, and it seems probable that it might decrease dementia risk so it’s a win-win,” she said.
In her own research, Dr. Fuller-Thomson has found that the combination of hearing loss and vision loss is linked to an eightfold increased risk for cognitive impairment.
“The idea is that vision and/or hearing loss makes it harder for you to be physically active, to be socially engaged, to be mentally stimulated. They are equally important in terms of social isolation, which could lead to loneliness, and we know that loneliness is not good for dementia,” she said.
“With dual sensory impairment, you don’t have as much information coming in — your brain is not engaged as much — and having an engaged brain, doing hobbies, having intellectually stimulating conversation, all of those are factors are associated with lowering risk of dementia,” Dr. Fuller-Thomson said.
The latest Lancet Commission report noted that treatment for visual loss is “effective and cost-effective” for an estimated 90% of people. However, across the world, particularly in low- and middle-income countries, visual loss often goes untreated.
the report concluded.
Dr. Ehrlich and Dr. Fuller-Thomson have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
In 2019, 57 million people worldwide were living with dementia, a figure expected to soar to 153 million by 2050. A recent Lancet Commission report suggests that nearly half of dementia cases could be prevented or delayed by addressing 14 modifiable risk factors, including impaired vision.
The report’s authors recommend that vision-loss screening and treatment be universally available. But are these recommendations warranted? What is the evidence? What is the potential mechanism? And what are the potential implications for clinical practice?
Worldwide, the prevalence of avoidable vision loss and blindness in adults aged 50 years or older is estimated to hover around 13%.
“There is now overwhelming evidence that vision impairment in later life is associated with more rapid cognitive decline and an increased risk of dementia,” said Joshua Ehrlich, MD, MPH, associate professor in ophthalmology and visual sciences, the Institute for Social Research at the University of Michigan, Ann Arbor.
The evidence includes a meta-analysis of 14 prospective cohort studies with roughly 6.2 million older adults who were cognitively intact at baseline. Over the course of up to 14 years, 171,888 developed dementia. Vision loss was associated with a pooled relative risk (RR) for dementia of 1.47.
A separate meta-analysis also identified an increased risk for dementia (RR, 1.38) with visual loss. When broken down into different eye conditions, an increased dementia risk was associated with cataracts and diabetic retinopathy but not with glaucoma or age-related macular degeneration.
A US study that followed roughly 3000 older adults with cataracts and normal cognition at baseline for more than 20 years found that those who had cataract extraction had significantly reduced risk for dementia compared with those who did not have cataract extraction (hazard ratio, 0.71), after controlling for age, race, APOE genotype, education, smoking, and an extensive list of comorbidities.
Causation or Coincidence?
The mechanisms behind these associations might be related to underlying illness, such as diabetes, which is a risk factor for dementia; vision loss itself, as might be suggested by a possible effect of cataract surgery; or shared neuropathologic processes in the retina and the brain.
A longitudinal study from Korea that included roughly 6 million adults showed that dementia risk increased with severity of visual loss, which supports the hypothesis that vision loss in itself might be causal or that there is a dose-response effect to a shared causal factor.
“Work is still needed to sort out” exactly how visual deficits may raise dementia risk, although several hypotheses exist, Dr. Ehrlich said.
For example, “decreased input to the brain via the visual pathways may directly induce brain changes. Also, consequences of vision loss, like social isolation, physical inactivity, and depression, are themselves risk factors for dementia and may explain the pathways through which vision impairment increases risk,” he said.
Is the link causal? “We’ll never know definitively because we can’t randomize people to not get cataract surgery versus getting cataract surgery, because we know that improving vision improves quality of life, so we’d never want to do that. But the new evidence that’s come in over the last 5 years or so is pretty promising,” said Esme Fuller-Thomson, PhD, director of the Institute for Life Course and Aging and professor, Department of Family and Community Medicine and Faculty of Nursing, at the University of Toronto, Ontario, Canada.
She noted that results of two studies that have looked at this “seem to indicate that those who have cataract surgery are not nearly at as high risk of dementia as those who have cataracts but don’t have the surgery. That’s leaning towards causality.”
A study published in July suggests that cataracts increase dementia risk through vascular and non–Alzheimer’s disease mechanisms.
Clear Clinical Implications
Dr. Ehrlich said that evidence for an association between untreated vision loss and dementia risk and potential modification by treatment has clear implications for care.
“Loss of vision impacts so many aspects of people’s lives beyond just how they see the world and losing vision in later life is not a normal part of aging. Thus, when older adults experience vision loss, this should be a cause for concern and prompt an immediate referral to an eye care professional,” he noted.
Dr. Fuller-Thomson agrees. “Addressing vision loss will certainly help people see better and function at a higher level and improve quality of life, and it seems probable that it might decrease dementia risk so it’s a win-win,” she said.
In her own research, Dr. Fuller-Thomson has found that the combination of hearing loss and vision loss is linked to an eightfold increased risk for cognitive impairment.
“The idea is that vision and/or hearing loss makes it harder for you to be physically active, to be socially engaged, to be mentally stimulated. They are equally important in terms of social isolation, which could lead to loneliness, and we know that loneliness is not good for dementia,” she said.
“With dual sensory impairment, you don’t have as much information coming in — your brain is not engaged as much — and having an engaged brain, doing hobbies, having intellectually stimulating conversation, all of those are factors are associated with lowering risk of dementia,” Dr. Fuller-Thomson said.
The latest Lancet Commission report noted that treatment for visual loss is “effective and cost-effective” for an estimated 90% of people. However, across the world, particularly in low- and middle-income countries, visual loss often goes untreated.
the report concluded.
Dr. Ehrlich and Dr. Fuller-Thomson have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Doctors Are Seeking Professional Coaches More Often. Here’s Why
When Andrea Austin, MD, an emergency medicine specialist, left the military in 2020, she knew the adjustment to civilian life and practice might be difficult. To help smooth the transition, she reached out to a physician mentor who also had a professional coaching certificate. After a conversation, Dr. Austin signed up for 6 months of career coaching.
It was time well spent, according to Dr. Austin, who today is a coach herself. “It was really the first time I had the ability to choose what I wanted to do, and that required a mindset shift,” she explains. “A big part of coaching is helping physicians discover their agency so that they can make the best career choices.”
Physicians have long lacked the coaching resources typically made available to corporate executives. But that’s changing. In today’s high-pressure environment, where doctors are burning out at a rapid pace, coaching can sometimes be an avenue to staying in the field, especially if that coach is a fellow physician who understands what you’re facing.
With a physician shortage that the Association of American Medical Colleges expects to hit 86,000 in the next decade or so, coaching could be a stone worth turning over. A 2024 report in JAMA Network Open found that coaching provided by physician peers led to a significant reduction in interpersonal disengagement and burnout.
“What I think is exciting about coaching is that it allows you to better understand yourself and know your strengths and weaknesses,” said Dr. Austin. “It might seem simple, but many ‘soft skills’ aren’t considered mainstream in medicine. Coaching allows us to understand them and ourselves better.”
Why Are Doctors Using Coaches?
Although it’s hard to put a number on how many physicians are turning to coaches, the number of coaches available for doctors is growing rapidly. The American Medical Women’s Association maintains a database of physician coaches. According to deputy director Jodi Godfrey, MS, RDN, the number of members who have added coaching to their skill set has tripled in the past 4 years. “Many cite burnout as the reason they sought coaching support, and then they decided to go on to get certified in coaching.”
The pandemic is one reason physician coaching has grown, said Elizabeth Esparaz, MD, an ophthalmologist and physician coach. “Since the pandemic, the word ‘burnout’ is thrown around a good deal.” And the causes are clear. “Doctors are facing longer hours, they must make split-second decisions, they’re multitasking, and they have less support staff.”
Among her coaching clients, Dr. Austin has noticed other common struggles: fears of litigation, time scarcity with patients, declining reimbursement that hasn’t kept up with inflation, and loss of autonomy because of the corporatization of healthcare.
Coaching, Dr. Esparaz believes, can be an antidote to many of these issues. “Coaches help doctors see their strengths and find better ways of applying them,” she said. “We help them move forward, and also see their blind spots.”
Clarity, Goals, and Making the Right Choices
Physician coaching comes in a variety of flavors — some one on one, and others in the form of group sessions. All, however, serve the purpose of helping physicians gain career clarity. “Sometimes clients realize their job may not be working for them, but that there are things they can do to change that without having to leave the field,” said Jattu Senesie, MD, a former ob.gyn. who is now a physician coach.
Dr. Esparaz works with doctors to establish SMART goals: specific, measurable, attainable, realistic, and time based. She gave the example of learning how to set boundaries. “If a physician is asked to create a presentation for work, I encourage them to ask for compensation or administrative time before committing to unpaid tasks.”
Another big issue: charting. It’s increasingly burdensome, and many doctors find it encroaching on their home lives. “If we can identify a problem like that, we can come up with a strategy for mitigating it,” Dr. Esparaz said. This might include setting a goal of getting 80% of charting completed immediately after the patient encounter on the busiest clinic day of the week. The client tests the experiment and then revisits it with the coach to discuss what worked and what didn’t, refining the process until it has freed up the physician’s home life.
The younger generation of doctors often struggles with career choices, too, because it’s the first time they are without structure, said Dr. Senesie. There’s med school and residency, which puts a framework around every move a doctor makes. But once they become attending physicians, the choices are endless. “Coaching can help them find a new structure and systems that will allow them to thrive.”
Although mentoring has been a well-embraced concept for decades, it “hits a wall,” at some point in terms of what it can offer, Dr. Austin said. That’s where coaching can take over. “There’s a point where a mentor cannot help someone self-actualize. As a coach, you don’t need to know everything about a doctor’s life, but you can help them learn to ask themselves the right questions to solve problems.”
Should You Stay or Should You Go?
Dr. Austin’s approach begins with the premise that healthcare today is challenging and dysfunctional — but doctors still have agency. She has worked with clients on the verge of leaving the field and helped them find their way back.
“They have a light bulb moment and open up to the idea that they have much to give still,” she said. “We take an inventory to help them better communicate their needs and make changes, and I help them connect to their values. Sometimes that exercise allows them to reframe their current work environment.”
Not every doctor who goes through coaching remains in the field. But “that’s the exception, not the rule,” Dr. Austin said. And that’s okay. “If that’s the outcome, coaching probably helped them get to that point faster, and with an informed decision.”
Dr. Senesie has been coaching for about a decade, and in that time, she’s seen a shift that goes beyond figuring out career goals. “Doctors are more aware of the need for well-being today. The pandemic made it impossible to ignore what doesn’t work for us. When I work with clients, we look for ways to make the job more tenable.”
According to Dr. Senesie, younger doctors are looking for that balance at the outset. “They want to be physicians, but they also want a life,” she said. “It’s a challenge for them because in addition to that mindset, they’re also coming out with more debt than older generations. They want out from underneath that.”
When It’s Time to Find a Physician Coach
Wondering whether coaching is right for you? Consider these symptoms:
- You need help setting boundaries at work.
- You feel like you’re sacrificing your own well-being for your job.
- You’re using maladaptive strategies to cope with the stress at work.
- You’ve reached a point where you are considering leaving the field.
If you’re interested in finding a physician coach, there are several places to begin your search, word of mouth being one of them. “Conferences and social media can also expose you to coaches,” suggested Dr. Esparaz. There are different methods and approaches to coaching. So, as you research, “make sure the coach you choose has techniques and a framework that fit what you’re after.”
Dr. Austin warned that it is an unregulated industry, so buyer beware. To ensure you’re getting an accredited physician coach, look for people who have obtained an International Coach Federation (ICF) accreditation. These coaches will hold an associate certified coach credential, which requires at least 60 hours of coaching-specific training approved by the ICF, in addition to other assessments and education.
Ensure that the coach you choose is within your budget. “There are some people charging astronomical rates out there,” Dr. Austin said. “If you’re burned out or struggling, it can be easy to reach for your credit card.”
Dr. Austin also cautioned doctors seeking a coach to avoid promises that sound too good to be true. Some coaching can have a gaslighting quality to it, she warned, “suggesting it can allow you to endure any environment.” But positive self-talk alone won’t cure an abusive or discriminatory situation. “If a client describes a toxic work environment,” the coach has an “ethical imperative” to help that person protect themselves.
A Side Gig or a New Career Path
After Dr. Austin’s experience with her coach, she made the choice to continue as an emergency physician part-time while starting her own coaching business. “It’s important for me personally to keep in touch with what’s happening on the ground, but I have no judgment for anyone who chooses to leave clinical practice to become a coach.”
When Dr. Senesie looks back on her own struggles as a clinician, she recognizes the state of burnout she was in 10 years ago. “I knew there was an issue, but I didn’t have the mindset to find a way to make it work,” she said. “I left the field when I was at my depths of burnout, which is generally not the best way to go about it.”
Guidance might have allowed her to take into account other avenues and helped her remain in the field, said Dr. Senesie. She has since learned that “there are many ways to practice medicine, and the way we’ve gone about it traditionally has worked for some, but not necessarily for everyone.”
There may be more possibilities than you think. By helping you assess your path and make meaningful changes, a physician coach might be the key to remaining in the field you love.
A version of this article first appeared on Medscape.com.
When Andrea Austin, MD, an emergency medicine specialist, left the military in 2020, she knew the adjustment to civilian life and practice might be difficult. To help smooth the transition, she reached out to a physician mentor who also had a professional coaching certificate. After a conversation, Dr. Austin signed up for 6 months of career coaching.
It was time well spent, according to Dr. Austin, who today is a coach herself. “It was really the first time I had the ability to choose what I wanted to do, and that required a mindset shift,” she explains. “A big part of coaching is helping physicians discover their agency so that they can make the best career choices.”
Physicians have long lacked the coaching resources typically made available to corporate executives. But that’s changing. In today’s high-pressure environment, where doctors are burning out at a rapid pace, coaching can sometimes be an avenue to staying in the field, especially if that coach is a fellow physician who understands what you’re facing.
With a physician shortage that the Association of American Medical Colleges expects to hit 86,000 in the next decade or so, coaching could be a stone worth turning over. A 2024 report in JAMA Network Open found that coaching provided by physician peers led to a significant reduction in interpersonal disengagement and burnout.
“What I think is exciting about coaching is that it allows you to better understand yourself and know your strengths and weaknesses,” said Dr. Austin. “It might seem simple, but many ‘soft skills’ aren’t considered mainstream in medicine. Coaching allows us to understand them and ourselves better.”
Why Are Doctors Using Coaches?
Although it’s hard to put a number on how many physicians are turning to coaches, the number of coaches available for doctors is growing rapidly. The American Medical Women’s Association maintains a database of physician coaches. According to deputy director Jodi Godfrey, MS, RDN, the number of members who have added coaching to their skill set has tripled in the past 4 years. “Many cite burnout as the reason they sought coaching support, and then they decided to go on to get certified in coaching.”
The pandemic is one reason physician coaching has grown, said Elizabeth Esparaz, MD, an ophthalmologist and physician coach. “Since the pandemic, the word ‘burnout’ is thrown around a good deal.” And the causes are clear. “Doctors are facing longer hours, they must make split-second decisions, they’re multitasking, and they have less support staff.”
Among her coaching clients, Dr. Austin has noticed other common struggles: fears of litigation, time scarcity with patients, declining reimbursement that hasn’t kept up with inflation, and loss of autonomy because of the corporatization of healthcare.
Coaching, Dr. Esparaz believes, can be an antidote to many of these issues. “Coaches help doctors see their strengths and find better ways of applying them,” she said. “We help them move forward, and also see their blind spots.”
Clarity, Goals, and Making the Right Choices
Physician coaching comes in a variety of flavors — some one on one, and others in the form of group sessions. All, however, serve the purpose of helping physicians gain career clarity. “Sometimes clients realize their job may not be working for them, but that there are things they can do to change that without having to leave the field,” said Jattu Senesie, MD, a former ob.gyn. who is now a physician coach.
Dr. Esparaz works with doctors to establish SMART goals: specific, measurable, attainable, realistic, and time based. She gave the example of learning how to set boundaries. “If a physician is asked to create a presentation for work, I encourage them to ask for compensation or administrative time before committing to unpaid tasks.”
Another big issue: charting. It’s increasingly burdensome, and many doctors find it encroaching on their home lives. “If we can identify a problem like that, we can come up with a strategy for mitigating it,” Dr. Esparaz said. This might include setting a goal of getting 80% of charting completed immediately after the patient encounter on the busiest clinic day of the week. The client tests the experiment and then revisits it with the coach to discuss what worked and what didn’t, refining the process until it has freed up the physician’s home life.
The younger generation of doctors often struggles with career choices, too, because it’s the first time they are without structure, said Dr. Senesie. There’s med school and residency, which puts a framework around every move a doctor makes. But once they become attending physicians, the choices are endless. “Coaching can help them find a new structure and systems that will allow them to thrive.”
Although mentoring has been a well-embraced concept for decades, it “hits a wall,” at some point in terms of what it can offer, Dr. Austin said. That’s where coaching can take over. “There’s a point where a mentor cannot help someone self-actualize. As a coach, you don’t need to know everything about a doctor’s life, but you can help them learn to ask themselves the right questions to solve problems.”
Should You Stay or Should You Go?
Dr. Austin’s approach begins with the premise that healthcare today is challenging and dysfunctional — but doctors still have agency. She has worked with clients on the verge of leaving the field and helped them find their way back.
“They have a light bulb moment and open up to the idea that they have much to give still,” she said. “We take an inventory to help them better communicate their needs and make changes, and I help them connect to their values. Sometimes that exercise allows them to reframe their current work environment.”
Not every doctor who goes through coaching remains in the field. But “that’s the exception, not the rule,” Dr. Austin said. And that’s okay. “If that’s the outcome, coaching probably helped them get to that point faster, and with an informed decision.”
Dr. Senesie has been coaching for about a decade, and in that time, she’s seen a shift that goes beyond figuring out career goals. “Doctors are more aware of the need for well-being today. The pandemic made it impossible to ignore what doesn’t work for us. When I work with clients, we look for ways to make the job more tenable.”
According to Dr. Senesie, younger doctors are looking for that balance at the outset. “They want to be physicians, but they also want a life,” she said. “It’s a challenge for them because in addition to that mindset, they’re also coming out with more debt than older generations. They want out from underneath that.”
When It’s Time to Find a Physician Coach
Wondering whether coaching is right for you? Consider these symptoms:
- You need help setting boundaries at work.
- You feel like you’re sacrificing your own well-being for your job.
- You’re using maladaptive strategies to cope with the stress at work.
- You’ve reached a point where you are considering leaving the field.
If you’re interested in finding a physician coach, there are several places to begin your search, word of mouth being one of them. “Conferences and social media can also expose you to coaches,” suggested Dr. Esparaz. There are different methods and approaches to coaching. So, as you research, “make sure the coach you choose has techniques and a framework that fit what you’re after.”
Dr. Austin warned that it is an unregulated industry, so buyer beware. To ensure you’re getting an accredited physician coach, look for people who have obtained an International Coach Federation (ICF) accreditation. These coaches will hold an associate certified coach credential, which requires at least 60 hours of coaching-specific training approved by the ICF, in addition to other assessments and education.
Ensure that the coach you choose is within your budget. “There are some people charging astronomical rates out there,” Dr. Austin said. “If you’re burned out or struggling, it can be easy to reach for your credit card.”
Dr. Austin also cautioned doctors seeking a coach to avoid promises that sound too good to be true. Some coaching can have a gaslighting quality to it, she warned, “suggesting it can allow you to endure any environment.” But positive self-talk alone won’t cure an abusive or discriminatory situation. “If a client describes a toxic work environment,” the coach has an “ethical imperative” to help that person protect themselves.
A Side Gig or a New Career Path
After Dr. Austin’s experience with her coach, she made the choice to continue as an emergency physician part-time while starting her own coaching business. “It’s important for me personally to keep in touch with what’s happening on the ground, but I have no judgment for anyone who chooses to leave clinical practice to become a coach.”
When Dr. Senesie looks back on her own struggles as a clinician, she recognizes the state of burnout she was in 10 years ago. “I knew there was an issue, but I didn’t have the mindset to find a way to make it work,” she said. “I left the field when I was at my depths of burnout, which is generally not the best way to go about it.”
Guidance might have allowed her to take into account other avenues and helped her remain in the field, said Dr. Senesie. She has since learned that “there are many ways to practice medicine, and the way we’ve gone about it traditionally has worked for some, but not necessarily for everyone.”
There may be more possibilities than you think. By helping you assess your path and make meaningful changes, a physician coach might be the key to remaining in the field you love.
A version of this article first appeared on Medscape.com.
When Andrea Austin, MD, an emergency medicine specialist, left the military in 2020, she knew the adjustment to civilian life and practice might be difficult. To help smooth the transition, she reached out to a physician mentor who also had a professional coaching certificate. After a conversation, Dr. Austin signed up for 6 months of career coaching.
It was time well spent, according to Dr. Austin, who today is a coach herself. “It was really the first time I had the ability to choose what I wanted to do, and that required a mindset shift,” she explains. “A big part of coaching is helping physicians discover their agency so that they can make the best career choices.”
Physicians have long lacked the coaching resources typically made available to corporate executives. But that’s changing. In today’s high-pressure environment, where doctors are burning out at a rapid pace, coaching can sometimes be an avenue to staying in the field, especially if that coach is a fellow physician who understands what you’re facing.
With a physician shortage that the Association of American Medical Colleges expects to hit 86,000 in the next decade or so, coaching could be a stone worth turning over. A 2024 report in JAMA Network Open found that coaching provided by physician peers led to a significant reduction in interpersonal disengagement and burnout.
“What I think is exciting about coaching is that it allows you to better understand yourself and know your strengths and weaknesses,” said Dr. Austin. “It might seem simple, but many ‘soft skills’ aren’t considered mainstream in medicine. Coaching allows us to understand them and ourselves better.”
Why Are Doctors Using Coaches?
Although it’s hard to put a number on how many physicians are turning to coaches, the number of coaches available for doctors is growing rapidly. The American Medical Women’s Association maintains a database of physician coaches. According to deputy director Jodi Godfrey, MS, RDN, the number of members who have added coaching to their skill set has tripled in the past 4 years. “Many cite burnout as the reason they sought coaching support, and then they decided to go on to get certified in coaching.”
The pandemic is one reason physician coaching has grown, said Elizabeth Esparaz, MD, an ophthalmologist and physician coach. “Since the pandemic, the word ‘burnout’ is thrown around a good deal.” And the causes are clear. “Doctors are facing longer hours, they must make split-second decisions, they’re multitasking, and they have less support staff.”
Among her coaching clients, Dr. Austin has noticed other common struggles: fears of litigation, time scarcity with patients, declining reimbursement that hasn’t kept up with inflation, and loss of autonomy because of the corporatization of healthcare.
Coaching, Dr. Esparaz believes, can be an antidote to many of these issues. “Coaches help doctors see their strengths and find better ways of applying them,” she said. “We help them move forward, and also see their blind spots.”
Clarity, Goals, and Making the Right Choices
Physician coaching comes in a variety of flavors — some one on one, and others in the form of group sessions. All, however, serve the purpose of helping physicians gain career clarity. “Sometimes clients realize their job may not be working for them, but that there are things they can do to change that without having to leave the field,” said Jattu Senesie, MD, a former ob.gyn. who is now a physician coach.
Dr. Esparaz works with doctors to establish SMART goals: specific, measurable, attainable, realistic, and time based. She gave the example of learning how to set boundaries. “If a physician is asked to create a presentation for work, I encourage them to ask for compensation or administrative time before committing to unpaid tasks.”
Another big issue: charting. It’s increasingly burdensome, and many doctors find it encroaching on their home lives. “If we can identify a problem like that, we can come up with a strategy for mitigating it,” Dr. Esparaz said. This might include setting a goal of getting 80% of charting completed immediately after the patient encounter on the busiest clinic day of the week. The client tests the experiment and then revisits it with the coach to discuss what worked and what didn’t, refining the process until it has freed up the physician’s home life.
The younger generation of doctors often struggles with career choices, too, because it’s the first time they are without structure, said Dr. Senesie. There’s med school and residency, which puts a framework around every move a doctor makes. But once they become attending physicians, the choices are endless. “Coaching can help them find a new structure and systems that will allow them to thrive.”
Although mentoring has been a well-embraced concept for decades, it “hits a wall,” at some point in terms of what it can offer, Dr. Austin said. That’s where coaching can take over. “There’s a point where a mentor cannot help someone self-actualize. As a coach, you don’t need to know everything about a doctor’s life, but you can help them learn to ask themselves the right questions to solve problems.”
Should You Stay or Should You Go?
Dr. Austin’s approach begins with the premise that healthcare today is challenging and dysfunctional — but doctors still have agency. She has worked with clients on the verge of leaving the field and helped them find their way back.
“They have a light bulb moment and open up to the idea that they have much to give still,” she said. “We take an inventory to help them better communicate their needs and make changes, and I help them connect to their values. Sometimes that exercise allows them to reframe their current work environment.”
Not every doctor who goes through coaching remains in the field. But “that’s the exception, not the rule,” Dr. Austin said. And that’s okay. “If that’s the outcome, coaching probably helped them get to that point faster, and with an informed decision.”
Dr. Senesie has been coaching for about a decade, and in that time, she’s seen a shift that goes beyond figuring out career goals. “Doctors are more aware of the need for well-being today. The pandemic made it impossible to ignore what doesn’t work for us. When I work with clients, we look for ways to make the job more tenable.”
According to Dr. Senesie, younger doctors are looking for that balance at the outset. “They want to be physicians, but they also want a life,” she said. “It’s a challenge for them because in addition to that mindset, they’re also coming out with more debt than older generations. They want out from underneath that.”
When It’s Time to Find a Physician Coach
Wondering whether coaching is right for you? Consider these symptoms:
- You need help setting boundaries at work.
- You feel like you’re sacrificing your own well-being for your job.
- You’re using maladaptive strategies to cope with the stress at work.
- You’ve reached a point where you are considering leaving the field.
If you’re interested in finding a physician coach, there are several places to begin your search, word of mouth being one of them. “Conferences and social media can also expose you to coaches,” suggested Dr. Esparaz. There are different methods and approaches to coaching. So, as you research, “make sure the coach you choose has techniques and a framework that fit what you’re after.”
Dr. Austin warned that it is an unregulated industry, so buyer beware. To ensure you’re getting an accredited physician coach, look for people who have obtained an International Coach Federation (ICF) accreditation. These coaches will hold an associate certified coach credential, which requires at least 60 hours of coaching-specific training approved by the ICF, in addition to other assessments and education.
Ensure that the coach you choose is within your budget. “There are some people charging astronomical rates out there,” Dr. Austin said. “If you’re burned out or struggling, it can be easy to reach for your credit card.”
Dr. Austin also cautioned doctors seeking a coach to avoid promises that sound too good to be true. Some coaching can have a gaslighting quality to it, she warned, “suggesting it can allow you to endure any environment.” But positive self-talk alone won’t cure an abusive or discriminatory situation. “If a client describes a toxic work environment,” the coach has an “ethical imperative” to help that person protect themselves.
A Side Gig or a New Career Path
After Dr. Austin’s experience with her coach, she made the choice to continue as an emergency physician part-time while starting her own coaching business. “It’s important for me personally to keep in touch with what’s happening on the ground, but I have no judgment for anyone who chooses to leave clinical practice to become a coach.”
When Dr. Senesie looks back on her own struggles as a clinician, she recognizes the state of burnout she was in 10 years ago. “I knew there was an issue, but I didn’t have the mindset to find a way to make it work,” she said. “I left the field when I was at my depths of burnout, which is generally not the best way to go about it.”
Guidance might have allowed her to take into account other avenues and helped her remain in the field, said Dr. Senesie. She has since learned that “there are many ways to practice medicine, and the way we’ve gone about it traditionally has worked for some, but not necessarily for everyone.”
There may be more possibilities than you think. By helping you assess your path and make meaningful changes, a physician coach might be the key to remaining in the field you love.
A version of this article first appeared on Medscape.com.
Fillers, Hyaluronidase Relieve Orofacial Changes in Patients with Scleroderma
CARLSBAD, CALIFORNIA — In 2003, researchers asked 303 patients with systemic sclerosis (scleroderma) what bothered them most about their disease from an aesthetic standpoint: Orofacial features, such as thin lips and mouth furrows, or non-facial features, such as fingertip ulceration and waxy changes to the skin.
Respondents expressed significant concern about specific orofacial features, including thin lips (73%), mouth furrows (80%), loss of facial lines (68%), and a smaller, tighter mouth (77%).
“Patients with systemic sclerosis may have loss of vermilion lip, microstomia, and perioral rhytids,” Kathleen Cook Suozzi, MD, who directs the Aesthetic Dermatology Program at Yale University School of Medicine, New Haven, Connecticut, said at the Controversies and Conversations in Laser and Cosmetic Surgery annual symposium. “How can we address these changes for our patients?”
Recent research has shown that hyaluronidase injections can help improve orofacial changes commonly experienced by patients with scleroderma. In 2019, researchers in Alabama reported the case of a 53-year-old woman treated with hyaluronidase for scleroderma-induced microstomia. After four visits over 7 months and a total hyaluronidase dose of 470 IU, the patient reported an improved Mouth Handicap in Systemic Sclerosis (MHISS) score (38 of 48); subjective improvement of symptoms, including greater ease in eating and undergoing dental treatment; and improved mouth closure.
In 2023, researchers published a cohort study of four women between the ages of 43 and 61 with autoimmune sclerosing conditions that resulted in oral microstomia. Following hyaluronidase injections, all improved in mouth opening capacity and MHISS, with change stabilizing between three and five treatments. More recently, in a study pending publication in JAAD Case Reports, Dr. Suozzi and colleagues retrospectively evaluated 12 women with scleroderma who received between 150 and 300 units of hyaluronic acid (HA) filler for microstomia between 2020 and 2023. Of the 12 women, 58% had diffuse disease, and 42% had limited disease. Overall, oral aperture width increased by 0.65 cm (P = .0027) and oral aperture height increased by 0.88 cm (P < .0001). “In general, patients needed three to four treatments to reach peak effect, and then they reached a plateau,” Dr. Suozzi said. “It wasn’t that the treatment wasn’t working anymore, but it was because their oral aperture had gotten to a size of around 5 cm, which is clinically normal. Interestingly, we found that if the patient’s disease flared and their microstomia started to return, when you rechallenged them, they continued to respond. So, patients can continue to use this treatment over time.”
In a separate case series of seven patients, Dr. Suozzi and colleagues prospectively evaluated the effect of HA soft tissue filler with Restylane Silk for lip augmentation. Study participants experienced statistically significant increases in the difference between pre- and postinjection fullness in both upper and lower lips. Also, the mean posttreatment score fell between “much improved” (2) and “improved” (3) on both the Investigator Global Aesthetic Improvement Scale and the Subject Global Aesthetic Improvement Scale.
Dr. Suozzi recommends using nerve blocks for injecting HA filler or hyaluronidase in patients with scleroderma and minimizing the injection points. “Initially, we were using 30% lidocaine preparations around the mouth for an hour before the procedure, and patients were still having pain, so now we use nerve blocks,” she said. “For hyaluronidase, we do perform a test dose of 75-100 units, usually in the commissure. It’s amazing how well it works; people will usually come back after their test dose and have improvements in their measurements. This is a really easy treatment to perform, and I think it can be done in the office of a general dermatologist. There is concern about cross-reactivity with bee venom, so you want to ask patients about that.”
Dr. Suozzi reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CARLSBAD, CALIFORNIA — In 2003, researchers asked 303 patients with systemic sclerosis (scleroderma) what bothered them most about their disease from an aesthetic standpoint: Orofacial features, such as thin lips and mouth furrows, or non-facial features, such as fingertip ulceration and waxy changes to the skin.
Respondents expressed significant concern about specific orofacial features, including thin lips (73%), mouth furrows (80%), loss of facial lines (68%), and a smaller, tighter mouth (77%).
“Patients with systemic sclerosis may have loss of vermilion lip, microstomia, and perioral rhytids,” Kathleen Cook Suozzi, MD, who directs the Aesthetic Dermatology Program at Yale University School of Medicine, New Haven, Connecticut, said at the Controversies and Conversations in Laser and Cosmetic Surgery annual symposium. “How can we address these changes for our patients?”
Recent research has shown that hyaluronidase injections can help improve orofacial changes commonly experienced by patients with scleroderma. In 2019, researchers in Alabama reported the case of a 53-year-old woman treated with hyaluronidase for scleroderma-induced microstomia. After four visits over 7 months and a total hyaluronidase dose of 470 IU, the patient reported an improved Mouth Handicap in Systemic Sclerosis (MHISS) score (38 of 48); subjective improvement of symptoms, including greater ease in eating and undergoing dental treatment; and improved mouth closure.
In 2023, researchers published a cohort study of four women between the ages of 43 and 61 with autoimmune sclerosing conditions that resulted in oral microstomia. Following hyaluronidase injections, all improved in mouth opening capacity and MHISS, with change stabilizing between three and five treatments. More recently, in a study pending publication in JAAD Case Reports, Dr. Suozzi and colleagues retrospectively evaluated 12 women with scleroderma who received between 150 and 300 units of hyaluronic acid (HA) filler for microstomia between 2020 and 2023. Of the 12 women, 58% had diffuse disease, and 42% had limited disease. Overall, oral aperture width increased by 0.65 cm (P = .0027) and oral aperture height increased by 0.88 cm (P < .0001). “In general, patients needed three to four treatments to reach peak effect, and then they reached a plateau,” Dr. Suozzi said. “It wasn’t that the treatment wasn’t working anymore, but it was because their oral aperture had gotten to a size of around 5 cm, which is clinically normal. Interestingly, we found that if the patient’s disease flared and their microstomia started to return, when you rechallenged them, they continued to respond. So, patients can continue to use this treatment over time.”
In a separate case series of seven patients, Dr. Suozzi and colleagues prospectively evaluated the effect of HA soft tissue filler with Restylane Silk for lip augmentation. Study participants experienced statistically significant increases in the difference between pre- and postinjection fullness in both upper and lower lips. Also, the mean posttreatment score fell between “much improved” (2) and “improved” (3) on both the Investigator Global Aesthetic Improvement Scale and the Subject Global Aesthetic Improvement Scale.
Dr. Suozzi recommends using nerve blocks for injecting HA filler or hyaluronidase in patients with scleroderma and minimizing the injection points. “Initially, we were using 30% lidocaine preparations around the mouth for an hour before the procedure, and patients were still having pain, so now we use nerve blocks,” she said. “For hyaluronidase, we do perform a test dose of 75-100 units, usually in the commissure. It’s amazing how well it works; people will usually come back after their test dose and have improvements in their measurements. This is a really easy treatment to perform, and I think it can be done in the office of a general dermatologist. There is concern about cross-reactivity with bee venom, so you want to ask patients about that.”
Dr. Suozzi reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CARLSBAD, CALIFORNIA — In 2003, researchers asked 303 patients with systemic sclerosis (scleroderma) what bothered them most about their disease from an aesthetic standpoint: Orofacial features, such as thin lips and mouth furrows, or non-facial features, such as fingertip ulceration and waxy changes to the skin.
Respondents expressed significant concern about specific orofacial features, including thin lips (73%), mouth furrows (80%), loss of facial lines (68%), and a smaller, tighter mouth (77%).
“Patients with systemic sclerosis may have loss of vermilion lip, microstomia, and perioral rhytids,” Kathleen Cook Suozzi, MD, who directs the Aesthetic Dermatology Program at Yale University School of Medicine, New Haven, Connecticut, said at the Controversies and Conversations in Laser and Cosmetic Surgery annual symposium. “How can we address these changes for our patients?”
Recent research has shown that hyaluronidase injections can help improve orofacial changes commonly experienced by patients with scleroderma. In 2019, researchers in Alabama reported the case of a 53-year-old woman treated with hyaluronidase for scleroderma-induced microstomia. After four visits over 7 months and a total hyaluronidase dose of 470 IU, the patient reported an improved Mouth Handicap in Systemic Sclerosis (MHISS) score (38 of 48); subjective improvement of symptoms, including greater ease in eating and undergoing dental treatment; and improved mouth closure.
In 2023, researchers published a cohort study of four women between the ages of 43 and 61 with autoimmune sclerosing conditions that resulted in oral microstomia. Following hyaluronidase injections, all improved in mouth opening capacity and MHISS, with change stabilizing between three and five treatments. More recently, in a study pending publication in JAAD Case Reports, Dr. Suozzi and colleagues retrospectively evaluated 12 women with scleroderma who received between 150 and 300 units of hyaluronic acid (HA) filler for microstomia between 2020 and 2023. Of the 12 women, 58% had diffuse disease, and 42% had limited disease. Overall, oral aperture width increased by 0.65 cm (P = .0027) and oral aperture height increased by 0.88 cm (P < .0001). “In general, patients needed three to four treatments to reach peak effect, and then they reached a plateau,” Dr. Suozzi said. “It wasn’t that the treatment wasn’t working anymore, but it was because their oral aperture had gotten to a size of around 5 cm, which is clinically normal. Interestingly, we found that if the patient’s disease flared and their microstomia started to return, when you rechallenged them, they continued to respond. So, patients can continue to use this treatment over time.”
In a separate case series of seven patients, Dr. Suozzi and colleagues prospectively evaluated the effect of HA soft tissue filler with Restylane Silk for lip augmentation. Study participants experienced statistically significant increases in the difference between pre- and postinjection fullness in both upper and lower lips. Also, the mean posttreatment score fell between “much improved” (2) and “improved” (3) on both the Investigator Global Aesthetic Improvement Scale and the Subject Global Aesthetic Improvement Scale.
Dr. Suozzi recommends using nerve blocks for injecting HA filler or hyaluronidase in patients with scleroderma and minimizing the injection points. “Initially, we were using 30% lidocaine preparations around the mouth for an hour before the procedure, and patients were still having pain, so now we use nerve blocks,” she said. “For hyaluronidase, we do perform a test dose of 75-100 units, usually in the commissure. It’s amazing how well it works; people will usually come back after their test dose and have improvements in their measurements. This is a really easy treatment to perform, and I think it can be done in the office of a general dermatologist. There is concern about cross-reactivity with bee venom, so you want to ask patients about that.”
Dr. Suozzi reported having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Is There a Role for GLP-1s in Neurology and Psychiatry?
This transcript has been edited for clarity.
I usually report five or six studies in the field of neurology that were published in the last months, but July was a vacation month.
I decided to cover another topic, which is the role of glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) receptor agonists beyond diabetes and obesity, and in particular, for the field of neurology and psychiatry. Until a few years ago, the treatment of diabetes with traditional antidiabetic drugs was frustrating for vascular neurologists.
These drugs would lower glucose and had an impact on small-vessel disease, but they had no impact on large-vessel disease, stroke, and vascular mortality. This changed with the sodium-glucose cotransporter 2 antagonists because these drugs were not only effective for diabetes, but they also lowered cardiac mortality, in particular, in patients with cardiac failure.
The next generation of antidiabetic drugs were the GLP-1 receptor agonists and the combined GIP/GLP-1 receptor agonists. These two polypeptides and their receptors play a very important role in diabetes and in obesity. The receptors are found not only in the pancreas but also in the intestinal system, the liver, and the central nervous system.
We have a number of preclinical models, mostly in transgenic mice, which show that these drugs are not effective only in diabetes and obesity, but also in liver disease, kidney failure, and neurodegenerative diseases. GLP-1 receptor agonists also have powerful anti-inflammatory properties. These drugs reduce body weight, and they have positive effects on blood pressure and lipid metabolism.
In the studies on the use of GLP-1 receptor agonists in diabetes, a meta-analysis with more than 58,000 patients showed a significant risk reduction for stroke compared with placebo, and this risk reduction was in the range of 80%.
Stroke, Smoking, and Alcohol
A meta-analysis on the use of GLP-1 receptor agonists in over 30,000 nondiabetic patients with obesity found a significant reduction in blood pressure, mortality, and the risk of myocardial infarction. There was no significant decrease in the risk of stroke, but most probably this is due to the fact that strokes are much less frequent in obesity than in diabetes.
You all know that obesity is also a major risk factor for sleep apnea syndrome. Recently, two large studies with the GIP/GLP-1 receptor agonist tirzepatide found a significant improvement in sleep apnea syndrome compared to placebo, regardless of whether patients needed continuous positive airway pressure therapy or not.
In the therapy studies on diabetes and obesity, there were indications that some smokers in the studies stopped their nicotine consumption. A small pilot study with exenatide in 84 overweight patients who were smokers showed that 46% of patients on exenatide stopped smoking compared with 27% in the placebo group. This could be an indication that GLP-1 receptor agonists have activity on the reward system in the brain. Currently, there are a number of larger placebo-controlled trials ongoing.
Another aspect is alcohol consumption. An epidemiologic study in Denmark using data from the National Health Registry showed that the incidence of alcohol-related events decreased significantly in almost 40,000 patients with diabetes when they were treated with GLP-1 receptor agonists compared with other antidiabetic drugs.
A retrospective cohort study from the United States with over 80,000 patients with obesity showed that treatment with GLP-1 receptor agonists was associated with a 50%-60% lower risk for occurrence or recurrence of high alcohol consumption. There is only one small study with exenatide, which was not really informative.
There are a number of studies underway for GLP-1 receptor agonists compared with placebo in patients with alcohol dependence or alcohol consumption. Preclinical models also indicate that these drugs might be effective in cocaine abuse, and there is one placebo-controlled study ongoing.
Parkinson’s Disease
Let’s come to neurology. Preclinical models of Parkinson’s disease have shown neuroprotective activities of GLP-1. Until now, we have three randomized placebo-controlled trials with exenatide, NLY01, and lixisenatide. Two of these studies were positive, showing that the symptoms of Parkinson’s disease were stable over time and deteriorated with placebo. One study was neutral. This means we need more large-scale placebo-controlled studies in the early phases of Parkinson’s disease.
Another potential use of GIP/GLP-1 receptor agonists is in dementia. These substances, as you know, have positive effects on high blood pressure and vascular risk factors.
A working group in China analyzed 27 studies on the treatment of diabetes. A small number of randomized studies and a large number of cohort studies showed that modern antidiabetic drugs reduce the risk for dementia. The risk reduction for dementia for the GLP-1 receptor agonists was 75%. At the moment, there are only small prospective studies and they are not conclusive. Again, we need large-scale placebo-controlled studies.
The most important limitation at the moment beyond the cost is the other adverse drug reactions with the GLP-1 receptor agonists; these include nausea, vomiting, diarrhea, and constipation. There might be a slightly increased risk for pancreatitis. The US Food and Drug Administration recently reported there is no increased risk for suicide. Another potential adverse drug reaction is nonatherosclerotic anterior optic neuropathy.
These drugs, GLP-1 receptor agonists and GIP agonists, are also investigated in a variety of other non-neurologic diseases. The focus here is on metabolic liver disease, such as fatty liver and kidney diseases. Smaller, positive studies have been conducted in this area, and large placebo-controlled trials for both indications are currently underway.
If these diverse therapeutic properties would turn out to be really the case with GLP-1 receptor agonists, this would lead to a significant expansion of the range of indications. If we consider cost, this would be the end of our healthcare systems because we cannot afford this. In addition, the new antidiabetic drugs and the treatment of obesity are available only to a limited extent.
Finally, at least for neurology, it’s unclear whether the impact of these diseases is in the brain or whether it’s indirect, due to the effectiveness on vascular risk factors and concomitant diseases.
Dr. Diener is Professor in the Department of Neurology, Stroke Center-Headache Center, University Duisburg-Essen, Essen, Germany; he has disclosed conflicts of interest with numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I usually report five or six studies in the field of neurology that were published in the last months, but July was a vacation month.
I decided to cover another topic, which is the role of glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) receptor agonists beyond diabetes and obesity, and in particular, for the field of neurology and psychiatry. Until a few years ago, the treatment of diabetes with traditional antidiabetic drugs was frustrating for vascular neurologists.
These drugs would lower glucose and had an impact on small-vessel disease, but they had no impact on large-vessel disease, stroke, and vascular mortality. This changed with the sodium-glucose cotransporter 2 antagonists because these drugs were not only effective for diabetes, but they also lowered cardiac mortality, in particular, in patients with cardiac failure.
The next generation of antidiabetic drugs were the GLP-1 receptor agonists and the combined GIP/GLP-1 receptor agonists. These two polypeptides and their receptors play a very important role in diabetes and in obesity. The receptors are found not only in the pancreas but also in the intestinal system, the liver, and the central nervous system.
We have a number of preclinical models, mostly in transgenic mice, which show that these drugs are not effective only in diabetes and obesity, but also in liver disease, kidney failure, and neurodegenerative diseases. GLP-1 receptor agonists also have powerful anti-inflammatory properties. These drugs reduce body weight, and they have positive effects on blood pressure and lipid metabolism.
In the studies on the use of GLP-1 receptor agonists in diabetes, a meta-analysis with more than 58,000 patients showed a significant risk reduction for stroke compared with placebo, and this risk reduction was in the range of 80%.
Stroke, Smoking, and Alcohol
A meta-analysis on the use of GLP-1 receptor agonists in over 30,000 nondiabetic patients with obesity found a significant reduction in blood pressure, mortality, and the risk of myocardial infarction. There was no significant decrease in the risk of stroke, but most probably this is due to the fact that strokes are much less frequent in obesity than in diabetes.
You all know that obesity is also a major risk factor for sleep apnea syndrome. Recently, two large studies with the GIP/GLP-1 receptor agonist tirzepatide found a significant improvement in sleep apnea syndrome compared to placebo, regardless of whether patients needed continuous positive airway pressure therapy or not.
In the therapy studies on diabetes and obesity, there were indications that some smokers in the studies stopped their nicotine consumption. A small pilot study with exenatide in 84 overweight patients who were smokers showed that 46% of patients on exenatide stopped smoking compared with 27% in the placebo group. This could be an indication that GLP-1 receptor agonists have activity on the reward system in the brain. Currently, there are a number of larger placebo-controlled trials ongoing.
Another aspect is alcohol consumption. An epidemiologic study in Denmark using data from the National Health Registry showed that the incidence of alcohol-related events decreased significantly in almost 40,000 patients with diabetes when they were treated with GLP-1 receptor agonists compared with other antidiabetic drugs.
A retrospective cohort study from the United States with over 80,000 patients with obesity showed that treatment with GLP-1 receptor agonists was associated with a 50%-60% lower risk for occurrence or recurrence of high alcohol consumption. There is only one small study with exenatide, which was not really informative.
There are a number of studies underway for GLP-1 receptor agonists compared with placebo in patients with alcohol dependence or alcohol consumption. Preclinical models also indicate that these drugs might be effective in cocaine abuse, and there is one placebo-controlled study ongoing.
Parkinson’s Disease
Let’s come to neurology. Preclinical models of Parkinson’s disease have shown neuroprotective activities of GLP-1. Until now, we have three randomized placebo-controlled trials with exenatide, NLY01, and lixisenatide. Two of these studies were positive, showing that the symptoms of Parkinson’s disease were stable over time and deteriorated with placebo. One study was neutral. This means we need more large-scale placebo-controlled studies in the early phases of Parkinson’s disease.
Another potential use of GIP/GLP-1 receptor agonists is in dementia. These substances, as you know, have positive effects on high blood pressure and vascular risk factors.
A working group in China analyzed 27 studies on the treatment of diabetes. A small number of randomized studies and a large number of cohort studies showed that modern antidiabetic drugs reduce the risk for dementia. The risk reduction for dementia for the GLP-1 receptor agonists was 75%. At the moment, there are only small prospective studies and they are not conclusive. Again, we need large-scale placebo-controlled studies.
The most important limitation at the moment beyond the cost is the other adverse drug reactions with the GLP-1 receptor agonists; these include nausea, vomiting, diarrhea, and constipation. There might be a slightly increased risk for pancreatitis. The US Food and Drug Administration recently reported there is no increased risk for suicide. Another potential adverse drug reaction is nonatherosclerotic anterior optic neuropathy.
These drugs, GLP-1 receptor agonists and GIP agonists, are also investigated in a variety of other non-neurologic diseases. The focus here is on metabolic liver disease, such as fatty liver and kidney diseases. Smaller, positive studies have been conducted in this area, and large placebo-controlled trials for both indications are currently underway.
If these diverse therapeutic properties would turn out to be really the case with GLP-1 receptor agonists, this would lead to a significant expansion of the range of indications. If we consider cost, this would be the end of our healthcare systems because we cannot afford this. In addition, the new antidiabetic drugs and the treatment of obesity are available only to a limited extent.
Finally, at least for neurology, it’s unclear whether the impact of these diseases is in the brain or whether it’s indirect, due to the effectiveness on vascular risk factors and concomitant diseases.
Dr. Diener is Professor in the Department of Neurology, Stroke Center-Headache Center, University Duisburg-Essen, Essen, Germany; he has disclosed conflicts of interest with numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I usually report five or six studies in the field of neurology that were published in the last months, but July was a vacation month.
I decided to cover another topic, which is the role of glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) receptor agonists beyond diabetes and obesity, and in particular, for the field of neurology and psychiatry. Until a few years ago, the treatment of diabetes with traditional antidiabetic drugs was frustrating for vascular neurologists.
These drugs would lower glucose and had an impact on small-vessel disease, but they had no impact on large-vessel disease, stroke, and vascular mortality. This changed with the sodium-glucose cotransporter 2 antagonists because these drugs were not only effective for diabetes, but they also lowered cardiac mortality, in particular, in patients with cardiac failure.
The next generation of antidiabetic drugs were the GLP-1 receptor agonists and the combined GIP/GLP-1 receptor agonists. These two polypeptides and their receptors play a very important role in diabetes and in obesity. The receptors are found not only in the pancreas but also in the intestinal system, the liver, and the central nervous system.
We have a number of preclinical models, mostly in transgenic mice, which show that these drugs are not effective only in diabetes and obesity, but also in liver disease, kidney failure, and neurodegenerative diseases. GLP-1 receptor agonists also have powerful anti-inflammatory properties. These drugs reduce body weight, and they have positive effects on blood pressure and lipid metabolism.
In the studies on the use of GLP-1 receptor agonists in diabetes, a meta-analysis with more than 58,000 patients showed a significant risk reduction for stroke compared with placebo, and this risk reduction was in the range of 80%.
Stroke, Smoking, and Alcohol
A meta-analysis on the use of GLP-1 receptor agonists in over 30,000 nondiabetic patients with obesity found a significant reduction in blood pressure, mortality, and the risk of myocardial infarction. There was no significant decrease in the risk of stroke, but most probably this is due to the fact that strokes are much less frequent in obesity than in diabetes.
You all know that obesity is also a major risk factor for sleep apnea syndrome. Recently, two large studies with the GIP/GLP-1 receptor agonist tirzepatide found a significant improvement in sleep apnea syndrome compared to placebo, regardless of whether patients needed continuous positive airway pressure therapy or not.
In the therapy studies on diabetes and obesity, there were indications that some smokers in the studies stopped their nicotine consumption. A small pilot study with exenatide in 84 overweight patients who were smokers showed that 46% of patients on exenatide stopped smoking compared with 27% in the placebo group. This could be an indication that GLP-1 receptor agonists have activity on the reward system in the brain. Currently, there are a number of larger placebo-controlled trials ongoing.
Another aspect is alcohol consumption. An epidemiologic study in Denmark using data from the National Health Registry showed that the incidence of alcohol-related events decreased significantly in almost 40,000 patients with diabetes when they were treated with GLP-1 receptor agonists compared with other antidiabetic drugs.
A retrospective cohort study from the United States with over 80,000 patients with obesity showed that treatment with GLP-1 receptor agonists was associated with a 50%-60% lower risk for occurrence or recurrence of high alcohol consumption. There is only one small study with exenatide, which was not really informative.
There are a number of studies underway for GLP-1 receptor agonists compared with placebo in patients with alcohol dependence or alcohol consumption. Preclinical models also indicate that these drugs might be effective in cocaine abuse, and there is one placebo-controlled study ongoing.
Parkinson’s Disease
Let’s come to neurology. Preclinical models of Parkinson’s disease have shown neuroprotective activities of GLP-1. Until now, we have three randomized placebo-controlled trials with exenatide, NLY01, and lixisenatide. Two of these studies were positive, showing that the symptoms of Parkinson’s disease were stable over time and deteriorated with placebo. One study was neutral. This means we need more large-scale placebo-controlled studies in the early phases of Parkinson’s disease.
Another potential use of GIP/GLP-1 receptor agonists is in dementia. These substances, as you know, have positive effects on high blood pressure and vascular risk factors.
A working group in China analyzed 27 studies on the treatment of diabetes. A small number of randomized studies and a large number of cohort studies showed that modern antidiabetic drugs reduce the risk for dementia. The risk reduction for dementia for the GLP-1 receptor agonists was 75%. At the moment, there are only small prospective studies and they are not conclusive. Again, we need large-scale placebo-controlled studies.
The most important limitation at the moment beyond the cost is the other adverse drug reactions with the GLP-1 receptor agonists; these include nausea, vomiting, diarrhea, and constipation. There might be a slightly increased risk for pancreatitis. The US Food and Drug Administration recently reported there is no increased risk for suicide. Another potential adverse drug reaction is nonatherosclerotic anterior optic neuropathy.
These drugs, GLP-1 receptor agonists and GIP agonists, are also investigated in a variety of other non-neurologic diseases. The focus here is on metabolic liver disease, such as fatty liver and kidney diseases. Smaller, positive studies have been conducted in this area, and large placebo-controlled trials for both indications are currently underway.
If these diverse therapeutic properties would turn out to be really the case with GLP-1 receptor agonists, this would lead to a significant expansion of the range of indications. If we consider cost, this would be the end of our healthcare systems because we cannot afford this. In addition, the new antidiabetic drugs and the treatment of obesity are available only to a limited extent.
Finally, at least for neurology, it’s unclear whether the impact of these diseases is in the brain or whether it’s indirect, due to the effectiveness on vascular risk factors and concomitant diseases.
Dr. Diener is Professor in the Department of Neurology, Stroke Center-Headache Center, University Duisburg-Essen, Essen, Germany; he has disclosed conflicts of interest with numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.