User login
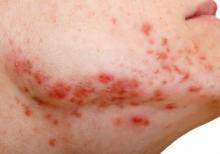
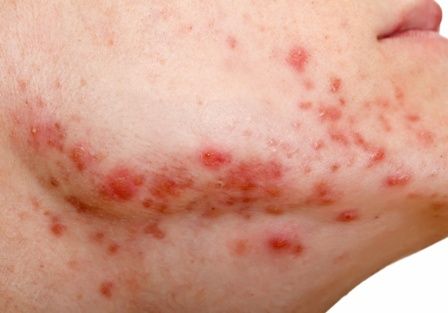
Keto for life? Reasons to think twice
Is the ketogenic diet the only way to lose weight? Of course not! Keep track of calories in vs. calories out and almost anyone can lose weight. The problem is keeping it off. To understand that, we need to look at metabolic adaptation and the biology of obesity.
Our bodies have a “set point” that is epigenetically latched onto the environment the brain senses, just as the fetal environment responds to the maternal environment.
If food is plentiful, our hormones force us to eat until our bodies feel that there are enough fat stores to survive. Because of environmental influences such as highly processed food, preservatives, climate change, and regulation of temperature, our brains have decided that we need more adipose tissue than we did 50-100 years ago. It could be that an element in food has caused a dysfunction of the pathways that regulate our body weight, and most of us “defend” a higher body weight in this environment.
How to counteract that? Not easily. The ketogenic diet works temporarily just like any other diet where calorie intake is lower than usual. It seems to be agreeable to many people because they say they feel full after eating protein, fat, and perhaps some vegetables. Protein and fat are certainly more satiating than simple carbohydrates.
If strictly followed, a ketogenic diet will force the body to burn fat and go into ketosis. Without a source for glucose, the brain will burn ketones from fat stores. Owen and colleagues discovered this in 1969 when they did their now-famous studies of fasting in inpatients at Brigham and Women’s hospital, using IV amino acids to protect muscle mass.
Keto for life?
Is the ketogenic diet a healthy diet for the long term? That is a different question.
Of course not – we need high-fiber carbohydrate sources such as whole grains, fruits, and vegetables to keep the colon healthy and obtain the vitamins and minerals needed to make the Krebs cycle, or citric acid cycle, work at its best.
Why, then, are we promoting ketogenic diets for those with obesity and type 2 diabetes? Ketogenic or low-carbohydrate diets are easy to teach and can rapidly help patients lose weight and return their blood glucose, blood pressure, and other metabolic parameters to normal.
The patient will be instructed to avoid all highly processed foods. Studies have shown that highly processed foods, created to maximize flavor, “coerce” people to eat more calories than when presented with the same number of calories in unprocessed foods, a way to fool the brain.
Why are we fooling the brain?
We circumvent the natural satiety mechanisms that start with the gut. When we eat, our gastric fundus and intestinal stretch receptors start the process that informs the hypothalamus about food intake. Highly processed foods are usually devoid of fiber and volume, and pack in the calories in small volumes so that the stretch receptors are not activated until more calories are ingested. The study mentioned above developed two ad lib diets with the same number of calories, sugar, fat, and carbohydrate content – one ultraprocessed and the other unprocessed.
That explanation is just the tip of the iceberg, because a lot more than primitive stretch receptors is informing the brain. There are gut hormones that are secreted before and after meals, such as ghrelin, glucagon-like peptide 1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and cholecystokinin (CCK), among a slew of others. These peptide hormones are all secreted from gut cells into the blood or vagus nerve, or both, and alert the brain that there is or is not enough food to maintain body weight at its set point.
It’s a highly regulated and precise system that regulates body weight for survival of the species in this environment. However, the environment has changed over the past 100 years but our genetic makeup for survival of the fittest has not. The mechanism of action for defense of a higher body weight set point in this new environment has not been elucidated as yet. Most likely, there are many players or instigators involved, such as food-supply changes, sedentary lifestyle, ambient temperature, fetal programming, air quality, and global warming and climate change, to name a few.
The goal of obesity researchers is to investigate the underlying mechanisms of the increased prevalence of obesity over the past 100 years. The goal of obesity medicine specialists is to treat obesity in adults and children, and to prevent obesity as much as possible with lifestyle change and medications that have been shown to help “reverse” the metabolic adaptation to this environment. Our newest GLP-1/GIP receptor agonists have been shown in animal models to hit several pathways that lead to obesity. They are not just appetite suppressants. Yes, they do modulate appetite and satiety, but they also affect energy expenditure. The body’s normal reaction to a lack of calorie intake is to reduce resting energy expenditure until body weight increases back to “set point levels.” These agonists prevent that metabolic adaptation. That is why they are true agents that can treat obesity – the disease.
Back to the ketogenic diet. The ketogenic diet can fool the brain temporarily by using protein and fat to elicit satiety with less food intake in calories. After a while, however, gut hormones and other factors begin to counteract the weight loss with a reduction in resting energy and total energy expenditure, and other metabolic measures, to get the body back to a certain body weight set point.
The ketogenic diet also can help dieters avoid ultra- and highly processed foods. In the end, any type of diet that lowers caloric intake will work for weight loss, but it’s the maintenance of that weight loss that makes a long-term difference, and that involves closing the metabolic gap that the body generates to defend fat mass. Understanding this pathophysiology will allow obesity medicine specialists to assist patients with obesity to lose weight and keep it off.
Dr. Apovian is in the department of medicine, division of endocrinology, diabetes, and hypertension, and codirector, Center for Weight Management and Wellness, Harvard Medical School, Boston. She disclosed ties with Altimmune, Cowen and Company, Currax Pharmaceuticals, EPG Communication Holdings, Gelesis Srl, L-Nutra, NeuroBo Pharmaceuticals, National Institutes of Health, Patient-Centered Outcomes Research Institute, GI Dynamics, and Novo Nordisk. A version of this article first appeared on Medscape.com.
Is the ketogenic diet the only way to lose weight? Of course not! Keep track of calories in vs. calories out and almost anyone can lose weight. The problem is keeping it off. To understand that, we need to look at metabolic adaptation and the biology of obesity.
Our bodies have a “set point” that is epigenetically latched onto the environment the brain senses, just as the fetal environment responds to the maternal environment.
If food is plentiful, our hormones force us to eat until our bodies feel that there are enough fat stores to survive. Because of environmental influences such as highly processed food, preservatives, climate change, and regulation of temperature, our brains have decided that we need more adipose tissue than we did 50-100 years ago. It could be that an element in food has caused a dysfunction of the pathways that regulate our body weight, and most of us “defend” a higher body weight in this environment.
How to counteract that? Not easily. The ketogenic diet works temporarily just like any other diet where calorie intake is lower than usual. It seems to be agreeable to many people because they say they feel full after eating protein, fat, and perhaps some vegetables. Protein and fat are certainly more satiating than simple carbohydrates.
If strictly followed, a ketogenic diet will force the body to burn fat and go into ketosis. Without a source for glucose, the brain will burn ketones from fat stores. Owen and colleagues discovered this in 1969 when they did their now-famous studies of fasting in inpatients at Brigham and Women’s hospital, using IV amino acids to protect muscle mass.
Keto for life?
Is the ketogenic diet a healthy diet for the long term? That is a different question.
Of course not – we need high-fiber carbohydrate sources such as whole grains, fruits, and vegetables to keep the colon healthy and obtain the vitamins and minerals needed to make the Krebs cycle, or citric acid cycle, work at its best.
Why, then, are we promoting ketogenic diets for those with obesity and type 2 diabetes? Ketogenic or low-carbohydrate diets are easy to teach and can rapidly help patients lose weight and return their blood glucose, blood pressure, and other metabolic parameters to normal.
The patient will be instructed to avoid all highly processed foods. Studies have shown that highly processed foods, created to maximize flavor, “coerce” people to eat more calories than when presented with the same number of calories in unprocessed foods, a way to fool the brain.
Why are we fooling the brain?
We circumvent the natural satiety mechanisms that start with the gut. When we eat, our gastric fundus and intestinal stretch receptors start the process that informs the hypothalamus about food intake. Highly processed foods are usually devoid of fiber and volume, and pack in the calories in small volumes so that the stretch receptors are not activated until more calories are ingested. The study mentioned above developed two ad lib diets with the same number of calories, sugar, fat, and carbohydrate content – one ultraprocessed and the other unprocessed.
That explanation is just the tip of the iceberg, because a lot more than primitive stretch receptors is informing the brain. There are gut hormones that are secreted before and after meals, such as ghrelin, glucagon-like peptide 1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and cholecystokinin (CCK), among a slew of others. These peptide hormones are all secreted from gut cells into the blood or vagus nerve, or both, and alert the brain that there is or is not enough food to maintain body weight at its set point.
It’s a highly regulated and precise system that regulates body weight for survival of the species in this environment. However, the environment has changed over the past 100 years but our genetic makeup for survival of the fittest has not. The mechanism of action for defense of a higher body weight set point in this new environment has not been elucidated as yet. Most likely, there are many players or instigators involved, such as food-supply changes, sedentary lifestyle, ambient temperature, fetal programming, air quality, and global warming and climate change, to name a few.
The goal of obesity researchers is to investigate the underlying mechanisms of the increased prevalence of obesity over the past 100 years. The goal of obesity medicine specialists is to treat obesity in adults and children, and to prevent obesity as much as possible with lifestyle change and medications that have been shown to help “reverse” the metabolic adaptation to this environment. Our newest GLP-1/GIP receptor agonists have been shown in animal models to hit several pathways that lead to obesity. They are not just appetite suppressants. Yes, they do modulate appetite and satiety, but they also affect energy expenditure. The body’s normal reaction to a lack of calorie intake is to reduce resting energy expenditure until body weight increases back to “set point levels.” These agonists prevent that metabolic adaptation. That is why they are true agents that can treat obesity – the disease.
Back to the ketogenic diet. The ketogenic diet can fool the brain temporarily by using protein and fat to elicit satiety with less food intake in calories. After a while, however, gut hormones and other factors begin to counteract the weight loss with a reduction in resting energy and total energy expenditure, and other metabolic measures, to get the body back to a certain body weight set point.
The ketogenic diet also can help dieters avoid ultra- and highly processed foods. In the end, any type of diet that lowers caloric intake will work for weight loss, but it’s the maintenance of that weight loss that makes a long-term difference, and that involves closing the metabolic gap that the body generates to defend fat mass. Understanding this pathophysiology will allow obesity medicine specialists to assist patients with obesity to lose weight and keep it off.
Dr. Apovian is in the department of medicine, division of endocrinology, diabetes, and hypertension, and codirector, Center for Weight Management and Wellness, Harvard Medical School, Boston. She disclosed ties with Altimmune, Cowen and Company, Currax Pharmaceuticals, EPG Communication Holdings, Gelesis Srl, L-Nutra, NeuroBo Pharmaceuticals, National Institutes of Health, Patient-Centered Outcomes Research Institute, GI Dynamics, and Novo Nordisk. A version of this article first appeared on Medscape.com.
Is the ketogenic diet the only way to lose weight? Of course not! Keep track of calories in vs. calories out and almost anyone can lose weight. The problem is keeping it off. To understand that, we need to look at metabolic adaptation and the biology of obesity.
Our bodies have a “set point” that is epigenetically latched onto the environment the brain senses, just as the fetal environment responds to the maternal environment.
If food is plentiful, our hormones force us to eat until our bodies feel that there are enough fat stores to survive. Because of environmental influences such as highly processed food, preservatives, climate change, and regulation of temperature, our brains have decided that we need more adipose tissue than we did 50-100 years ago. It could be that an element in food has caused a dysfunction of the pathways that regulate our body weight, and most of us “defend” a higher body weight in this environment.
How to counteract that? Not easily. The ketogenic diet works temporarily just like any other diet where calorie intake is lower than usual. It seems to be agreeable to many people because they say they feel full after eating protein, fat, and perhaps some vegetables. Protein and fat are certainly more satiating than simple carbohydrates.
If strictly followed, a ketogenic diet will force the body to burn fat and go into ketosis. Without a source for glucose, the brain will burn ketones from fat stores. Owen and colleagues discovered this in 1969 when they did their now-famous studies of fasting in inpatients at Brigham and Women’s hospital, using IV amino acids to protect muscle mass.
Keto for life?
Is the ketogenic diet a healthy diet for the long term? That is a different question.
Of course not – we need high-fiber carbohydrate sources such as whole grains, fruits, and vegetables to keep the colon healthy and obtain the vitamins and minerals needed to make the Krebs cycle, or citric acid cycle, work at its best.
Why, then, are we promoting ketogenic diets for those with obesity and type 2 diabetes? Ketogenic or low-carbohydrate diets are easy to teach and can rapidly help patients lose weight and return their blood glucose, blood pressure, and other metabolic parameters to normal.
The patient will be instructed to avoid all highly processed foods. Studies have shown that highly processed foods, created to maximize flavor, “coerce” people to eat more calories than when presented with the same number of calories in unprocessed foods, a way to fool the brain.
Why are we fooling the brain?
We circumvent the natural satiety mechanisms that start with the gut. When we eat, our gastric fundus and intestinal stretch receptors start the process that informs the hypothalamus about food intake. Highly processed foods are usually devoid of fiber and volume, and pack in the calories in small volumes so that the stretch receptors are not activated until more calories are ingested. The study mentioned above developed two ad lib diets with the same number of calories, sugar, fat, and carbohydrate content – one ultraprocessed and the other unprocessed.
That explanation is just the tip of the iceberg, because a lot more than primitive stretch receptors is informing the brain. There are gut hormones that are secreted before and after meals, such as ghrelin, glucagon-like peptide 1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and cholecystokinin (CCK), among a slew of others. These peptide hormones are all secreted from gut cells into the blood or vagus nerve, or both, and alert the brain that there is or is not enough food to maintain body weight at its set point.
It’s a highly regulated and precise system that regulates body weight for survival of the species in this environment. However, the environment has changed over the past 100 years but our genetic makeup for survival of the fittest has not. The mechanism of action for defense of a higher body weight set point in this new environment has not been elucidated as yet. Most likely, there are many players or instigators involved, such as food-supply changes, sedentary lifestyle, ambient temperature, fetal programming, air quality, and global warming and climate change, to name a few.
The goal of obesity researchers is to investigate the underlying mechanisms of the increased prevalence of obesity over the past 100 years. The goal of obesity medicine specialists is to treat obesity in adults and children, and to prevent obesity as much as possible with lifestyle change and medications that have been shown to help “reverse” the metabolic adaptation to this environment. Our newest GLP-1/GIP receptor agonists have been shown in animal models to hit several pathways that lead to obesity. They are not just appetite suppressants. Yes, they do modulate appetite and satiety, but they also affect energy expenditure. The body’s normal reaction to a lack of calorie intake is to reduce resting energy expenditure until body weight increases back to “set point levels.” These agonists prevent that metabolic adaptation. That is why they are true agents that can treat obesity – the disease.
Back to the ketogenic diet. The ketogenic diet can fool the brain temporarily by using protein and fat to elicit satiety with less food intake in calories. After a while, however, gut hormones and other factors begin to counteract the weight loss with a reduction in resting energy and total energy expenditure, and other metabolic measures, to get the body back to a certain body weight set point.
The ketogenic diet also can help dieters avoid ultra- and highly processed foods. In the end, any type of diet that lowers caloric intake will work for weight loss, but it’s the maintenance of that weight loss that makes a long-term difference, and that involves closing the metabolic gap that the body generates to defend fat mass. Understanding this pathophysiology will allow obesity medicine specialists to assist patients with obesity to lose weight and keep it off.
Dr. Apovian is in the department of medicine, division of endocrinology, diabetes, and hypertension, and codirector, Center for Weight Management and Wellness, Harvard Medical School, Boston. She disclosed ties with Altimmune, Cowen and Company, Currax Pharmaceuticals, EPG Communication Holdings, Gelesis Srl, L-Nutra, NeuroBo Pharmaceuticals, National Institutes of Health, Patient-Centered Outcomes Research Institute, GI Dynamics, and Novo Nordisk. A version of this article first appeared on Medscape.com.
Persistent gaps in drug use by patients with type 2 diabetes
Adults with mainly type 2 diabetes had gaps in the use of medications for managing blood glucose, hypertension, and lipids, in an analysis of nationally representative U.S. survey data.
A mean of 19.5%, 17.1%, and 43.3% of survey participants had inconsistent use of glucose-, BP-, or lipid-lowering medications, respectively, over 2 years in a series of successive 2-year surveys in 2005-2019.
A new group of participants was enrolled for each successive 2-year survey.
“We found persistent and sometimes increasing gaps in continuity of use of these [glycemia, hypertension, and lipid] treatments at the national level,” the researchers wrote.
Moreover, “this outcome was found despite long-lasting guidelines that generally recommend medications as an ongoing part of therapy for adults with type 2 diabetes to reduce macrovascular and microvascular disease risk,” they stressed.
The data did not distinguish between type 1 and type 2 diabetes, but more than 90% of diabetes diagnoses in the United States are type 2 diabetes, the researchers noted.
Therefore, it is “correct, our findings primarily reflect type 2 diabetes,” lead author Puneet Kaur Chehal, PhD, assistant professor, Emory University, Atlanta, clarified in an email.
“The clinical guidelines for treatment of type 1 diabetes are distinct,” she added, so “it is difficult to draw any conclusions from our study for this population.”
“To observe national trends in continuous use decrease at the same time that diabetes complications are increasing and physicians are guided to shift away from treat-to-target and towards individual patient needs certainly caught our attention,” she said.
“Our findings highlight the need for additional research to understand what is going on here,” according to Dr. Chehal.
“We did not observe levels of glucose (or blood pressure and lipids) to explore if the decrease in glucose-lowering drugs was warranted,” she added. “Our evidence of differences in continuity in use across subgroups (by race/ethnicity, payer, and age) does warrant further analysis of whether the decreasing trends we observe are lapses in access or deliberate changes in treatment.”
The study was published online in JAMA Network Open.
Investigating trends in medication adherence
Type 2 diabetes is a chronic condition and medications to control blood glucose, BP, and lipids lower the risk of diabetes-associated complications, Dr. Chehal and colleagues wrote.
After years of improvement, these cardiometabolic parameters plateaued and even decreased in 2013-2021, in parallel with increasing rates of diabetes complications, especially in younger adults, certain ethnic minority groups, and people with increased risks.
Suboptimal medication adherence among people with type 2 diabetes is associated with preventable complications and onset of heart disease, kidney disease, or diabetic neuropathy, which can lead to amputation.
However, previous studies of medication adherence were typically limited to patients covered by Medicare or commercial insurance, or studies only had 1-year follow-up.
Therefore, the researchers performed a cross-sectional analysis of a series of 2-year data from the Medical Expenditure Panel Survey (MEPS), in which participants reply to five interviews in 2 years and new participants are selected each year.
The researchers analyzed data from 15,237 adults aged 18 and older with type 2 diabetes who participated in 1 of 14 2-year MEPS survey panels in 2005-2019.
About half of participants (47.4%) were age 45-64 and about half (54.2%) were women. They were also racially diverse (43% non-Latino White, 25% Latino, and 24% non-Latino Black).
Participants were classified as having “inconsistent use” of glucose-lowering medication, for example, if they did not fill at least one prescription for a glucose-lowering drug in each of the 2 years.
“As long as [the medication] was some type of glucose-, blood pressure–, or lipid-lowering medication and was filled, it counted as continued use for that category,” Dr. Chehal explained.
They are preparing another paper that explores changes in medication regimens.
The current study showed continued use of glucose-lowering medication in both years decreased from 84.5% in 2005-2006 to 77.4% in 2018-2019, no use of glucose-lowering medication in either of the 2 years increased from 8.1% in 2005-2006 to 12.9% in 2018-2019, inconsistent use of glucose-lowering medication increased from 3.3% in 2005-2006 to 7.1% in 2018-2019, and new use of glucose-lowering medications in year 2 fluctuated between 2% and 4% across panels.
It also showed inconsistent use of BP-lowering medication increased from 3.9% in 2005-2006 to 9.0% in 2016-2017 and inconsistent use of lipid-lowering medication increased to a high of 9.9% in 2017-2018.
Younger and Black participants were less likely to consistently use glucose-lowering medication, Latino patients were less likely to consistently use BP-lowering medications, and Black and Latino patients were less likely to continuously use lipid-lowering medications. Uninsured adults were more likely to use no medications or use medications inconsistently.
“Changes and inconsistencies in payer formularies and out-of-pocket cost burden, especially among adults with no or insufficient insurance (i.e., Medicare Part D), remain prominent issues,” according to Dr. Chehal and colleagues.
“Decreases in continuity in use of glucose-lowering medications in recent panels may explain worsening diabetes complications,” they wrote.
This may be partly caused by recommended decreases in sulfonylurea and thiazolidinedione use and increased prescribing of new and more cost-prohibitive medications, they suggested.
Or this may be caused by the shift away from treating aggressively until a target is achieved toward individualizing treatment based on a patient’s age, phenotype, or comorbidities (for example, kidney disease).
The study was supported by a grant from MSD, a subsidiary of Merck, to Emory University. Some of the researchers received grants from Merck for the submitted work or were partially supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health to the Georgia Center for Diabetes Translation Research. Dr. Chehal reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Adults with mainly type 2 diabetes had gaps in the use of medications for managing blood glucose, hypertension, and lipids, in an analysis of nationally representative U.S. survey data.
A mean of 19.5%, 17.1%, and 43.3% of survey participants had inconsistent use of glucose-, BP-, or lipid-lowering medications, respectively, over 2 years in a series of successive 2-year surveys in 2005-2019.
A new group of participants was enrolled for each successive 2-year survey.
“We found persistent and sometimes increasing gaps in continuity of use of these [glycemia, hypertension, and lipid] treatments at the national level,” the researchers wrote.
Moreover, “this outcome was found despite long-lasting guidelines that generally recommend medications as an ongoing part of therapy for adults with type 2 diabetes to reduce macrovascular and microvascular disease risk,” they stressed.
The data did not distinguish between type 1 and type 2 diabetes, but more than 90% of diabetes diagnoses in the United States are type 2 diabetes, the researchers noted.
Therefore, it is “correct, our findings primarily reflect type 2 diabetes,” lead author Puneet Kaur Chehal, PhD, assistant professor, Emory University, Atlanta, clarified in an email.
“The clinical guidelines for treatment of type 1 diabetes are distinct,” she added, so “it is difficult to draw any conclusions from our study for this population.”
“To observe national trends in continuous use decrease at the same time that diabetes complications are increasing and physicians are guided to shift away from treat-to-target and towards individual patient needs certainly caught our attention,” she said.
“Our findings highlight the need for additional research to understand what is going on here,” according to Dr. Chehal.
“We did not observe levels of glucose (or blood pressure and lipids) to explore if the decrease in glucose-lowering drugs was warranted,” she added. “Our evidence of differences in continuity in use across subgroups (by race/ethnicity, payer, and age) does warrant further analysis of whether the decreasing trends we observe are lapses in access or deliberate changes in treatment.”
The study was published online in JAMA Network Open.
Investigating trends in medication adherence
Type 2 diabetes is a chronic condition and medications to control blood glucose, BP, and lipids lower the risk of diabetes-associated complications, Dr. Chehal and colleagues wrote.
After years of improvement, these cardiometabolic parameters plateaued and even decreased in 2013-2021, in parallel with increasing rates of diabetes complications, especially in younger adults, certain ethnic minority groups, and people with increased risks.
Suboptimal medication adherence among people with type 2 diabetes is associated with preventable complications and onset of heart disease, kidney disease, or diabetic neuropathy, which can lead to amputation.
However, previous studies of medication adherence were typically limited to patients covered by Medicare or commercial insurance, or studies only had 1-year follow-up.
Therefore, the researchers performed a cross-sectional analysis of a series of 2-year data from the Medical Expenditure Panel Survey (MEPS), in which participants reply to five interviews in 2 years and new participants are selected each year.
The researchers analyzed data from 15,237 adults aged 18 and older with type 2 diabetes who participated in 1 of 14 2-year MEPS survey panels in 2005-2019.
About half of participants (47.4%) were age 45-64 and about half (54.2%) were women. They were also racially diverse (43% non-Latino White, 25% Latino, and 24% non-Latino Black).
Participants were classified as having “inconsistent use” of glucose-lowering medication, for example, if they did not fill at least one prescription for a glucose-lowering drug in each of the 2 years.
“As long as [the medication] was some type of glucose-, blood pressure–, or lipid-lowering medication and was filled, it counted as continued use for that category,” Dr. Chehal explained.
They are preparing another paper that explores changes in medication regimens.
The current study showed continued use of glucose-lowering medication in both years decreased from 84.5% in 2005-2006 to 77.4% in 2018-2019, no use of glucose-lowering medication in either of the 2 years increased from 8.1% in 2005-2006 to 12.9% in 2018-2019, inconsistent use of glucose-lowering medication increased from 3.3% in 2005-2006 to 7.1% in 2018-2019, and new use of glucose-lowering medications in year 2 fluctuated between 2% and 4% across panels.
It also showed inconsistent use of BP-lowering medication increased from 3.9% in 2005-2006 to 9.0% in 2016-2017 and inconsistent use of lipid-lowering medication increased to a high of 9.9% in 2017-2018.
Younger and Black participants were less likely to consistently use glucose-lowering medication, Latino patients were less likely to consistently use BP-lowering medications, and Black and Latino patients were less likely to continuously use lipid-lowering medications. Uninsured adults were more likely to use no medications or use medications inconsistently.
“Changes and inconsistencies in payer formularies and out-of-pocket cost burden, especially among adults with no or insufficient insurance (i.e., Medicare Part D), remain prominent issues,” according to Dr. Chehal and colleagues.
“Decreases in continuity in use of glucose-lowering medications in recent panels may explain worsening diabetes complications,” they wrote.
This may be partly caused by recommended decreases in sulfonylurea and thiazolidinedione use and increased prescribing of new and more cost-prohibitive medications, they suggested.
Or this may be caused by the shift away from treating aggressively until a target is achieved toward individualizing treatment based on a patient’s age, phenotype, or comorbidities (for example, kidney disease).
The study was supported by a grant from MSD, a subsidiary of Merck, to Emory University. Some of the researchers received grants from Merck for the submitted work or were partially supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health to the Georgia Center for Diabetes Translation Research. Dr. Chehal reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Adults with mainly type 2 diabetes had gaps in the use of medications for managing blood glucose, hypertension, and lipids, in an analysis of nationally representative U.S. survey data.
A mean of 19.5%, 17.1%, and 43.3% of survey participants had inconsistent use of glucose-, BP-, or lipid-lowering medications, respectively, over 2 years in a series of successive 2-year surveys in 2005-2019.
A new group of participants was enrolled for each successive 2-year survey.
“We found persistent and sometimes increasing gaps in continuity of use of these [glycemia, hypertension, and lipid] treatments at the national level,” the researchers wrote.
Moreover, “this outcome was found despite long-lasting guidelines that generally recommend medications as an ongoing part of therapy for adults with type 2 diabetes to reduce macrovascular and microvascular disease risk,” they stressed.
The data did not distinguish between type 1 and type 2 diabetes, but more than 90% of diabetes diagnoses in the United States are type 2 diabetes, the researchers noted.
Therefore, it is “correct, our findings primarily reflect type 2 diabetes,” lead author Puneet Kaur Chehal, PhD, assistant professor, Emory University, Atlanta, clarified in an email.
“The clinical guidelines for treatment of type 1 diabetes are distinct,” she added, so “it is difficult to draw any conclusions from our study for this population.”
“To observe national trends in continuous use decrease at the same time that diabetes complications are increasing and physicians are guided to shift away from treat-to-target and towards individual patient needs certainly caught our attention,” she said.
“Our findings highlight the need for additional research to understand what is going on here,” according to Dr. Chehal.
“We did not observe levels of glucose (or blood pressure and lipids) to explore if the decrease in glucose-lowering drugs was warranted,” she added. “Our evidence of differences in continuity in use across subgroups (by race/ethnicity, payer, and age) does warrant further analysis of whether the decreasing trends we observe are lapses in access or deliberate changes in treatment.”
The study was published online in JAMA Network Open.
Investigating trends in medication adherence
Type 2 diabetes is a chronic condition and medications to control blood glucose, BP, and lipids lower the risk of diabetes-associated complications, Dr. Chehal and colleagues wrote.
After years of improvement, these cardiometabolic parameters plateaued and even decreased in 2013-2021, in parallel with increasing rates of diabetes complications, especially in younger adults, certain ethnic minority groups, and people with increased risks.
Suboptimal medication adherence among people with type 2 diabetes is associated with preventable complications and onset of heart disease, kidney disease, or diabetic neuropathy, which can lead to amputation.
However, previous studies of medication adherence were typically limited to patients covered by Medicare or commercial insurance, or studies only had 1-year follow-up.
Therefore, the researchers performed a cross-sectional analysis of a series of 2-year data from the Medical Expenditure Panel Survey (MEPS), in which participants reply to five interviews in 2 years and new participants are selected each year.
The researchers analyzed data from 15,237 adults aged 18 and older with type 2 diabetes who participated in 1 of 14 2-year MEPS survey panels in 2005-2019.
About half of participants (47.4%) were age 45-64 and about half (54.2%) were women. They were also racially diverse (43% non-Latino White, 25% Latino, and 24% non-Latino Black).
Participants were classified as having “inconsistent use” of glucose-lowering medication, for example, if they did not fill at least one prescription for a glucose-lowering drug in each of the 2 years.
“As long as [the medication] was some type of glucose-, blood pressure–, or lipid-lowering medication and was filled, it counted as continued use for that category,” Dr. Chehal explained.
They are preparing another paper that explores changes in medication regimens.
The current study showed continued use of glucose-lowering medication in both years decreased from 84.5% in 2005-2006 to 77.4% in 2018-2019, no use of glucose-lowering medication in either of the 2 years increased from 8.1% in 2005-2006 to 12.9% in 2018-2019, inconsistent use of glucose-lowering medication increased from 3.3% in 2005-2006 to 7.1% in 2018-2019, and new use of glucose-lowering medications in year 2 fluctuated between 2% and 4% across panels.
It also showed inconsistent use of BP-lowering medication increased from 3.9% in 2005-2006 to 9.0% in 2016-2017 and inconsistent use of lipid-lowering medication increased to a high of 9.9% in 2017-2018.
Younger and Black participants were less likely to consistently use glucose-lowering medication, Latino patients were less likely to consistently use BP-lowering medications, and Black and Latino patients were less likely to continuously use lipid-lowering medications. Uninsured adults were more likely to use no medications or use medications inconsistently.
“Changes and inconsistencies in payer formularies and out-of-pocket cost burden, especially among adults with no or insufficient insurance (i.e., Medicare Part D), remain prominent issues,” according to Dr. Chehal and colleagues.
“Decreases in continuity in use of glucose-lowering medications in recent panels may explain worsening diabetes complications,” they wrote.
This may be partly caused by recommended decreases in sulfonylurea and thiazolidinedione use and increased prescribing of new and more cost-prohibitive medications, they suggested.
Or this may be caused by the shift away from treating aggressively until a target is achieved toward individualizing treatment based on a patient’s age, phenotype, or comorbidities (for example, kidney disease).
The study was supported by a grant from MSD, a subsidiary of Merck, to Emory University. Some of the researchers received grants from Merck for the submitted work or were partially supported by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health to the Georgia Center for Diabetes Translation Research. Dr. Chehal reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM JAMA NETWORK OPEN
After decade of effort, AGA welcomes change to colonoscopy coverage
When the Affordable Care Act was signed in 2010, patients became eligible for coverage of screening examinations, which was good news for gastroenterologists and their patients in the fight to prevent colorectal cancer.
But there was a problem: While noninvasive stool tests – measuring microscopic amounts of blood or key DNA mutations, both tip-offs to increased colorectal cancer risk – were considered a screen, the necessary follow-up colonoscopy was not, leaving patients with expensive copays or other payment responsibility.
That interpretation prompted a years-long effort to have the federal agencies who oversee the health insurance sector reinterpret their policy, which finally changed in May 2022. Medicare, which is not covered by the ACA and is administered by a different agency, announced a change to its policy in July 2022, and the change went into effect in January 2023.
“It’s a major victory for patients, and I couldn’t be more delighted,” said David Lieberman, MD, AGAF, professor of medicine in gastroenterology at Oregon Health & Science University, Portland, and former American Gastroenterological Association president, who participated in the effort.
AGA Regulatory Affairs Director Leslie Narramore said AGA Vice President of Public Policy and Advocacy Kathleen Teixeira worked for 10 years, alongside other GI societies and patient advocacy groups, to close the “colonoscopy loophole.” In 2022, the AGA, Fight Colorectal Cancer, and the American Cancer Society–Cancer Action Network met with White House officials and senior officials with the Department of Health & Human Services about expanding the coverage. They also lobbied to eliminate cost-sharing, another way in which preventive colonoscopy is discouraged.
“The COVID-19 public health emergency highlighted health disparities and barriers to access to care,” said Ms. Narramore, who gave input about the effort along with Ms. Teixeira and AGA Director of Government Affairs Sarah Ankney. “The temporary suspension of elective procedures, including screening colonoscopies, exacerbated the existing low colorectal screening rates and created momentum and willingness in agency officials to create positive change.”
Without coverage of the colonoscopy, patients have needed to cover at least part of the cost of the procedure, which could be $1,000 or more with private insurance, or $100 or more with Medicare, Dr. Lieberman said. So, he noted, unsuspecting patients might receive a positive result on a noninvasive test and have a colonoscopy, only to get a “surprise bill.” Or they would know about the lack of coverage and not get the colonoscopy.
“Prior to the policy change, gastroenterologists and their staff had to explain to patients that their insurer would not fully pay for a colonoscopy following a positive noninvasive stool test, and field questions from upset patients who weren’t aware of their insurance plan’s cost-sharing requirements for a cancer screening procedure they thought was free,” Ms. Narramore said.
There’s little doubt the change will help save lives and improve quality of life, Dr. Lieberman said.
“We don’t know the full impact of this new ruling, but we know that financial barriers are important for some patients,” he said. “And so by removing these barriers we hope that we’re going to see improved adherence to follow-up with colonoscopy after a positive stool test, and that would result in reductions in incidence, mortality, and increased life-years gained.”
Ms. Narramore said the changes show the importance of pushing for policy change.
“Our physician advocates were effective in educating policy makers on the need for coverage of the full colorectal cancer screening continuum, and how colorectal cancer needs to be viewed as a program given the various steps necessary for a complete screening,” she said. “These successful efforts demonstrate to our members that advocacy works and that they can be a voice for their patients in improving their access to care.”
When the Affordable Care Act was signed in 2010, patients became eligible for coverage of screening examinations, which was good news for gastroenterologists and their patients in the fight to prevent colorectal cancer.
But there was a problem: While noninvasive stool tests – measuring microscopic amounts of blood or key DNA mutations, both tip-offs to increased colorectal cancer risk – were considered a screen, the necessary follow-up colonoscopy was not, leaving patients with expensive copays or other payment responsibility.
That interpretation prompted a years-long effort to have the federal agencies who oversee the health insurance sector reinterpret their policy, which finally changed in May 2022. Medicare, which is not covered by the ACA and is administered by a different agency, announced a change to its policy in July 2022, and the change went into effect in January 2023.
“It’s a major victory for patients, and I couldn’t be more delighted,” said David Lieberman, MD, AGAF, professor of medicine in gastroenterology at Oregon Health & Science University, Portland, and former American Gastroenterological Association president, who participated in the effort.
AGA Regulatory Affairs Director Leslie Narramore said AGA Vice President of Public Policy and Advocacy Kathleen Teixeira worked for 10 years, alongside other GI societies and patient advocacy groups, to close the “colonoscopy loophole.” In 2022, the AGA, Fight Colorectal Cancer, and the American Cancer Society–Cancer Action Network met with White House officials and senior officials with the Department of Health & Human Services about expanding the coverage. They also lobbied to eliminate cost-sharing, another way in which preventive colonoscopy is discouraged.
“The COVID-19 public health emergency highlighted health disparities and barriers to access to care,” said Ms. Narramore, who gave input about the effort along with Ms. Teixeira and AGA Director of Government Affairs Sarah Ankney. “The temporary suspension of elective procedures, including screening colonoscopies, exacerbated the existing low colorectal screening rates and created momentum and willingness in agency officials to create positive change.”
Without coverage of the colonoscopy, patients have needed to cover at least part of the cost of the procedure, which could be $1,000 or more with private insurance, or $100 or more with Medicare, Dr. Lieberman said. So, he noted, unsuspecting patients might receive a positive result on a noninvasive test and have a colonoscopy, only to get a “surprise bill.” Or they would know about the lack of coverage and not get the colonoscopy.
“Prior to the policy change, gastroenterologists and their staff had to explain to patients that their insurer would not fully pay for a colonoscopy following a positive noninvasive stool test, and field questions from upset patients who weren’t aware of their insurance plan’s cost-sharing requirements for a cancer screening procedure they thought was free,” Ms. Narramore said.
There’s little doubt the change will help save lives and improve quality of life, Dr. Lieberman said.
“We don’t know the full impact of this new ruling, but we know that financial barriers are important for some patients,” he said. “And so by removing these barriers we hope that we’re going to see improved adherence to follow-up with colonoscopy after a positive stool test, and that would result in reductions in incidence, mortality, and increased life-years gained.”
Ms. Narramore said the changes show the importance of pushing for policy change.
“Our physician advocates were effective in educating policy makers on the need for coverage of the full colorectal cancer screening continuum, and how colorectal cancer needs to be viewed as a program given the various steps necessary for a complete screening,” she said. “These successful efforts demonstrate to our members that advocacy works and that they can be a voice for their patients in improving their access to care.”
When the Affordable Care Act was signed in 2010, patients became eligible for coverage of screening examinations, which was good news for gastroenterologists and their patients in the fight to prevent colorectal cancer.
But there was a problem: While noninvasive stool tests – measuring microscopic amounts of blood or key DNA mutations, both tip-offs to increased colorectal cancer risk – were considered a screen, the necessary follow-up colonoscopy was not, leaving patients with expensive copays or other payment responsibility.
That interpretation prompted a years-long effort to have the federal agencies who oversee the health insurance sector reinterpret their policy, which finally changed in May 2022. Medicare, which is not covered by the ACA and is administered by a different agency, announced a change to its policy in July 2022, and the change went into effect in January 2023.
“It’s a major victory for patients, and I couldn’t be more delighted,” said David Lieberman, MD, AGAF, professor of medicine in gastroenterology at Oregon Health & Science University, Portland, and former American Gastroenterological Association president, who participated in the effort.
AGA Regulatory Affairs Director Leslie Narramore said AGA Vice President of Public Policy and Advocacy Kathleen Teixeira worked for 10 years, alongside other GI societies and patient advocacy groups, to close the “colonoscopy loophole.” In 2022, the AGA, Fight Colorectal Cancer, and the American Cancer Society–Cancer Action Network met with White House officials and senior officials with the Department of Health & Human Services about expanding the coverage. They also lobbied to eliminate cost-sharing, another way in which preventive colonoscopy is discouraged.
“The COVID-19 public health emergency highlighted health disparities and barriers to access to care,” said Ms. Narramore, who gave input about the effort along with Ms. Teixeira and AGA Director of Government Affairs Sarah Ankney. “The temporary suspension of elective procedures, including screening colonoscopies, exacerbated the existing low colorectal screening rates and created momentum and willingness in agency officials to create positive change.”
Without coverage of the colonoscopy, patients have needed to cover at least part of the cost of the procedure, which could be $1,000 or more with private insurance, or $100 or more with Medicare, Dr. Lieberman said. So, he noted, unsuspecting patients might receive a positive result on a noninvasive test and have a colonoscopy, only to get a “surprise bill.” Or they would know about the lack of coverage and not get the colonoscopy.
“Prior to the policy change, gastroenterologists and their staff had to explain to patients that their insurer would not fully pay for a colonoscopy following a positive noninvasive stool test, and field questions from upset patients who weren’t aware of their insurance plan’s cost-sharing requirements for a cancer screening procedure they thought was free,” Ms. Narramore said.
There’s little doubt the change will help save lives and improve quality of life, Dr. Lieberman said.
“We don’t know the full impact of this new ruling, but we know that financial barriers are important for some patients,” he said. “And so by removing these barriers we hope that we’re going to see improved adherence to follow-up with colonoscopy after a positive stool test, and that would result in reductions in incidence, mortality, and increased life-years gained.”
Ms. Narramore said the changes show the importance of pushing for policy change.
“Our physician advocates were effective in educating policy makers on the need for coverage of the full colorectal cancer screening continuum, and how colorectal cancer needs to be viewed as a program given the various steps necessary for a complete screening,” she said. “These successful efforts demonstrate to our members that advocacy works and that they can be a voice for their patients in improving their access to care.”
In adults with prediabetes, vitamin D cuts diabetes risk
Results of the analysis, led by Anastassios G. Pittas, MD, MS, with the division of endocrinology, diabetes, and metabolism at Tufts Medical Center, in Boston, were published online in Annals of Internal Medicine (2023 Feb 7. doi: 10.7326/M22-3018).
All three eligible trials included in the analysis were randomized, double blinded, and placebo controlled. The three eligible trials tested three oral formulations of Vitamin D: cholecalciferol, 20,000 IU (500 mcg) weekly; cholecalciferol, 4,000 IU (100 mcg) daily; or eldecalcitol, 0.75 mcg daily, against placebos.
The authors of the new paper found that vitamin D reduced the risk for diabetes in people with prediabetes by a statistically significant 15% in adjusted analyses. The 3-year absolute risk reduction was 3.3%.
They found no difference in the rate ratios for adverse events (kidney stones, 1.17, 95% confidence interval, 0.69-1.99; hypercalcemia, 2.34; 95% CI, 0.83-6.66]; hypercalciuria, 1.65; 95% CI, 0.83-3.28]; death, 0.85; 95% CI, 0.31-2.36]) when study participants got vitamin D instead of placebo.
Differences from previous analyses
The relationship between vitamin D levels and risk for type 2 diabetes has been studied in previous trials and results have been mixed.
The authors note that two previous meta-analyses included trials “that had relatively short durations for assessment of diabetes risk (for example, ≤ 1 year), had high risk of bias (for example, open-label trials), or were not specifically designed and conducted for primary prevention of type 2 diabetes, potentially undermining the validity of the results.”
Each of the trials in this meta-analysis had a low risk of bias as determined by the Cochrane risk-of-bias tool, Dr. Pittas and colleagues said.
“The present study does not reach an opposite conclusion from the D2d study,” said Dr. Pittas, who coauthored that paper as well. “Rather, it confirms the results of the D2d study. In D2d and two other similar vitamin D and diabetes prevention trials (one in Norway and one in Japan), vitamin D reduced the rate of progression to diabetes in adults with prediabetes, but the observed differences were not statistically significant because the reported relative risk reductions (10%-13%) were smaller than each trial was powered to detect (25%-36%).”
“Individual participant data meta-analyses increase the statistical power to detect an effect. After combining data, we found that vitamin D reduced the risk of progression from prediabetes to diabetes by 15% and this result was statistically significant. So, the conclusion of the meta-analysis is essentially the same conclusion as in D2d and the other two trials. The difference is that the result is now statistically significant,” Dr. Pittas added.
Small reduction but large population
The authors acknowledged that the absolute risk reduction number is small, especially when compared with the risk reduction seen with intensive lifestyle changes (58%) and metformin (31%), as reported in an article published in the New England of Journal of Medicine (2002 Feb 7;346:393-403). But “extrapolating to the more than 374 million adults worldwide who have prediabetes suggests that inexpensive vitamin D supplementation could delay the development of diabetes in more than 10 million people,” they said.
As for how high vitamin D levels need to be, the authors write that their research indicates that the optimal level of vitamin D in the blood needed to reduce diabetes risk may be higher than an Institute of Medicine committee recommendation in 2011.
“The blood 25-hydroxy vitamin D level needed to optimally reduce diabetes risk may be near and possibly above the range of 125-150 nmol/L (50-60 ng/mL) that the 2011 Institute of Medicine Committee to Review Dietary Reference Intakes for Calcium and Vitamin D provided as the range corresponding to the tolerable upper intake level (UL) of 4,000 IU/d for vitamin D,” the authors of the new paper said.
Editorialists urge caution
In an accompanying editorial also published in the Annals of Internal Medicine, Malachi J. McKenna, MD, with the department of clinical chemistry, at St. Vincent’s University Hospital, and Mary A.T. Flynn, PhD, RD, with the Food Safety Authority of Ireland in Dublin, urge caution regarding vitamin D dosing.
They write that there are important distinctions between vitamin D supplements and vitamin D therapy, and the potential harms of high-dose vitamin D are still unclear.
“Vitamin D supplementation of 10 to 20 mcg (400 to 800 IU) daily can be applied safely at the population level to prevent skeletal and possibly nonskeletal disease. Very-high-dose vitamin D therapy might prevent type 2 diabetes in some patients but may also cause harm,” they note.
Dr. Pittas said in an interview that there have been some studies with high-dose vitamin D (up to 500,000 IU a year in one study) that reported an increased fall risk in older adults who had high fall risk. “However, these findings are not generalizable to other populations that are younger and at low or average fall risk, such as the prediabetes population to which the results of this meta-analysis apply,” he noted.
“The benefit-to-risk ratio for vitamin D depends on the target population and medical condition,” Dr. Pittas said. “The editorial refers to the NAM (National Academy of Medicine) vitamin D guidelines for the general, healthy population to promote bone health. The guidelines should not be extrapolated to specific populations, for example [patients with] prediabetes,” where the vitamin D benefit-to-risk ratio would be different from that in the general population.
Dr. Pittas and colleagues caution that the people studied in this meta-analysis were at high risk for type 2 diabetes, so these results do not apply to the general healthy population. The results also should not be extrapolated to people at average risk for any type of diabetes, they add.
Several physicians either declined to comment or did not respond to requests for comment on this research.
Dr. Pittas reports the National Institutes of Health and the American Diabetes Association made payments to his institution to conduct Vitamin D-related research. He is an unpaid cochair of the Endocrine Society’s Evaluation, Treatment and Prevention of Vitamin D Deficiency Clinical Practice Guideline team.
Coauthor Dr. Jorde reports grants from Novo Nordisk Foundation, North Norwegian Regional Health Authorities, and the Research Council of Norway.
Dr. Dawson-Hughes reports she is on the DSMB for AgNovos Healthcare. AgNovos is developing a bone implant to reduce hip fracture risk and she gets a stipend from the company. She reports Helsinn Therapeutics provided anamorelin and matching placebo for an NIH-funded clinical trial.
Dr. Trikalinos was supported by the D2d study. He is a technical methodological consultant to Latham and Watkins, who is retained by Pacira Pharmaceuticals.
Dr. Angellotti has been employed by Takeda and owns stock in the company.
The editorialists report no relevant financial relationships.
Results of the analysis, led by Anastassios G. Pittas, MD, MS, with the division of endocrinology, diabetes, and metabolism at Tufts Medical Center, in Boston, were published online in Annals of Internal Medicine (2023 Feb 7. doi: 10.7326/M22-3018).
All three eligible trials included in the analysis were randomized, double blinded, and placebo controlled. The three eligible trials tested three oral formulations of Vitamin D: cholecalciferol, 20,000 IU (500 mcg) weekly; cholecalciferol, 4,000 IU (100 mcg) daily; or eldecalcitol, 0.75 mcg daily, against placebos.
The authors of the new paper found that vitamin D reduced the risk for diabetes in people with prediabetes by a statistically significant 15% in adjusted analyses. The 3-year absolute risk reduction was 3.3%.
They found no difference in the rate ratios for adverse events (kidney stones, 1.17, 95% confidence interval, 0.69-1.99; hypercalcemia, 2.34; 95% CI, 0.83-6.66]; hypercalciuria, 1.65; 95% CI, 0.83-3.28]; death, 0.85; 95% CI, 0.31-2.36]) when study participants got vitamin D instead of placebo.
Differences from previous analyses
The relationship between vitamin D levels and risk for type 2 diabetes has been studied in previous trials and results have been mixed.
The authors note that two previous meta-analyses included trials “that had relatively short durations for assessment of diabetes risk (for example, ≤ 1 year), had high risk of bias (for example, open-label trials), or were not specifically designed and conducted for primary prevention of type 2 diabetes, potentially undermining the validity of the results.”
Each of the trials in this meta-analysis had a low risk of bias as determined by the Cochrane risk-of-bias tool, Dr. Pittas and colleagues said.
“The present study does not reach an opposite conclusion from the D2d study,” said Dr. Pittas, who coauthored that paper as well. “Rather, it confirms the results of the D2d study. In D2d and two other similar vitamin D and diabetes prevention trials (one in Norway and one in Japan), vitamin D reduced the rate of progression to diabetes in adults with prediabetes, but the observed differences were not statistically significant because the reported relative risk reductions (10%-13%) were smaller than each trial was powered to detect (25%-36%).”
“Individual participant data meta-analyses increase the statistical power to detect an effect. After combining data, we found that vitamin D reduced the risk of progression from prediabetes to diabetes by 15% and this result was statistically significant. So, the conclusion of the meta-analysis is essentially the same conclusion as in D2d and the other two trials. The difference is that the result is now statistically significant,” Dr. Pittas added.
Small reduction but large population
The authors acknowledged that the absolute risk reduction number is small, especially when compared with the risk reduction seen with intensive lifestyle changes (58%) and metformin (31%), as reported in an article published in the New England of Journal of Medicine (2002 Feb 7;346:393-403). But “extrapolating to the more than 374 million adults worldwide who have prediabetes suggests that inexpensive vitamin D supplementation could delay the development of diabetes in more than 10 million people,” they said.
As for how high vitamin D levels need to be, the authors write that their research indicates that the optimal level of vitamin D in the blood needed to reduce diabetes risk may be higher than an Institute of Medicine committee recommendation in 2011.
“The blood 25-hydroxy vitamin D level needed to optimally reduce diabetes risk may be near and possibly above the range of 125-150 nmol/L (50-60 ng/mL) that the 2011 Institute of Medicine Committee to Review Dietary Reference Intakes for Calcium and Vitamin D provided as the range corresponding to the tolerable upper intake level (UL) of 4,000 IU/d for vitamin D,” the authors of the new paper said.
Editorialists urge caution
In an accompanying editorial also published in the Annals of Internal Medicine, Malachi J. McKenna, MD, with the department of clinical chemistry, at St. Vincent’s University Hospital, and Mary A.T. Flynn, PhD, RD, with the Food Safety Authority of Ireland in Dublin, urge caution regarding vitamin D dosing.
They write that there are important distinctions between vitamin D supplements and vitamin D therapy, and the potential harms of high-dose vitamin D are still unclear.
“Vitamin D supplementation of 10 to 20 mcg (400 to 800 IU) daily can be applied safely at the population level to prevent skeletal and possibly nonskeletal disease. Very-high-dose vitamin D therapy might prevent type 2 diabetes in some patients but may also cause harm,” they note.
Dr. Pittas said in an interview that there have been some studies with high-dose vitamin D (up to 500,000 IU a year in one study) that reported an increased fall risk in older adults who had high fall risk. “However, these findings are not generalizable to other populations that are younger and at low or average fall risk, such as the prediabetes population to which the results of this meta-analysis apply,” he noted.
“The benefit-to-risk ratio for vitamin D depends on the target population and medical condition,” Dr. Pittas said. “The editorial refers to the NAM (National Academy of Medicine) vitamin D guidelines for the general, healthy population to promote bone health. The guidelines should not be extrapolated to specific populations, for example [patients with] prediabetes,” where the vitamin D benefit-to-risk ratio would be different from that in the general population.
Dr. Pittas and colleagues caution that the people studied in this meta-analysis were at high risk for type 2 diabetes, so these results do not apply to the general healthy population. The results also should not be extrapolated to people at average risk for any type of diabetes, they add.
Several physicians either declined to comment or did not respond to requests for comment on this research.
Dr. Pittas reports the National Institutes of Health and the American Diabetes Association made payments to his institution to conduct Vitamin D-related research. He is an unpaid cochair of the Endocrine Society’s Evaluation, Treatment and Prevention of Vitamin D Deficiency Clinical Practice Guideline team.
Coauthor Dr. Jorde reports grants from Novo Nordisk Foundation, North Norwegian Regional Health Authorities, and the Research Council of Norway.
Dr. Dawson-Hughes reports she is on the DSMB for AgNovos Healthcare. AgNovos is developing a bone implant to reduce hip fracture risk and she gets a stipend from the company. She reports Helsinn Therapeutics provided anamorelin and matching placebo for an NIH-funded clinical trial.
Dr. Trikalinos was supported by the D2d study. He is a technical methodological consultant to Latham and Watkins, who is retained by Pacira Pharmaceuticals.
Dr. Angellotti has been employed by Takeda and owns stock in the company.
The editorialists report no relevant financial relationships.
Results of the analysis, led by Anastassios G. Pittas, MD, MS, with the division of endocrinology, diabetes, and metabolism at Tufts Medical Center, in Boston, were published online in Annals of Internal Medicine (2023 Feb 7. doi: 10.7326/M22-3018).
All three eligible trials included in the analysis were randomized, double blinded, and placebo controlled. The three eligible trials tested three oral formulations of Vitamin D: cholecalciferol, 20,000 IU (500 mcg) weekly; cholecalciferol, 4,000 IU (100 mcg) daily; or eldecalcitol, 0.75 mcg daily, against placebos.
The authors of the new paper found that vitamin D reduced the risk for diabetes in people with prediabetes by a statistically significant 15% in adjusted analyses. The 3-year absolute risk reduction was 3.3%.
They found no difference in the rate ratios for adverse events (kidney stones, 1.17, 95% confidence interval, 0.69-1.99; hypercalcemia, 2.34; 95% CI, 0.83-6.66]; hypercalciuria, 1.65; 95% CI, 0.83-3.28]; death, 0.85; 95% CI, 0.31-2.36]) when study participants got vitamin D instead of placebo.
Differences from previous analyses
The relationship between vitamin D levels and risk for type 2 diabetes has been studied in previous trials and results have been mixed.
The authors note that two previous meta-analyses included trials “that had relatively short durations for assessment of diabetes risk (for example, ≤ 1 year), had high risk of bias (for example, open-label trials), or were not specifically designed and conducted for primary prevention of type 2 diabetes, potentially undermining the validity of the results.”
Each of the trials in this meta-analysis had a low risk of bias as determined by the Cochrane risk-of-bias tool, Dr. Pittas and colleagues said.
“The present study does not reach an opposite conclusion from the D2d study,” said Dr. Pittas, who coauthored that paper as well. “Rather, it confirms the results of the D2d study. In D2d and two other similar vitamin D and diabetes prevention trials (one in Norway and one in Japan), vitamin D reduced the rate of progression to diabetes in adults with prediabetes, but the observed differences were not statistically significant because the reported relative risk reductions (10%-13%) were smaller than each trial was powered to detect (25%-36%).”
“Individual participant data meta-analyses increase the statistical power to detect an effect. After combining data, we found that vitamin D reduced the risk of progression from prediabetes to diabetes by 15% and this result was statistically significant. So, the conclusion of the meta-analysis is essentially the same conclusion as in D2d and the other two trials. The difference is that the result is now statistically significant,” Dr. Pittas added.
Small reduction but large population
The authors acknowledged that the absolute risk reduction number is small, especially when compared with the risk reduction seen with intensive lifestyle changes (58%) and metformin (31%), as reported in an article published in the New England of Journal of Medicine (2002 Feb 7;346:393-403). But “extrapolating to the more than 374 million adults worldwide who have prediabetes suggests that inexpensive vitamin D supplementation could delay the development of diabetes in more than 10 million people,” they said.
As for how high vitamin D levels need to be, the authors write that their research indicates that the optimal level of vitamin D in the blood needed to reduce diabetes risk may be higher than an Institute of Medicine committee recommendation in 2011.
“The blood 25-hydroxy vitamin D level needed to optimally reduce diabetes risk may be near and possibly above the range of 125-150 nmol/L (50-60 ng/mL) that the 2011 Institute of Medicine Committee to Review Dietary Reference Intakes for Calcium and Vitamin D provided as the range corresponding to the tolerable upper intake level (UL) of 4,000 IU/d for vitamin D,” the authors of the new paper said.
Editorialists urge caution
In an accompanying editorial also published in the Annals of Internal Medicine, Malachi J. McKenna, MD, with the department of clinical chemistry, at St. Vincent’s University Hospital, and Mary A.T. Flynn, PhD, RD, with the Food Safety Authority of Ireland in Dublin, urge caution regarding vitamin D dosing.
They write that there are important distinctions between vitamin D supplements and vitamin D therapy, and the potential harms of high-dose vitamin D are still unclear.
“Vitamin D supplementation of 10 to 20 mcg (400 to 800 IU) daily can be applied safely at the population level to prevent skeletal and possibly nonskeletal disease. Very-high-dose vitamin D therapy might prevent type 2 diabetes in some patients but may also cause harm,” they note.
Dr. Pittas said in an interview that there have been some studies with high-dose vitamin D (up to 500,000 IU a year in one study) that reported an increased fall risk in older adults who had high fall risk. “However, these findings are not generalizable to other populations that are younger and at low or average fall risk, such as the prediabetes population to which the results of this meta-analysis apply,” he noted.
“The benefit-to-risk ratio for vitamin D depends on the target population and medical condition,” Dr. Pittas said. “The editorial refers to the NAM (National Academy of Medicine) vitamin D guidelines for the general, healthy population to promote bone health. The guidelines should not be extrapolated to specific populations, for example [patients with] prediabetes,” where the vitamin D benefit-to-risk ratio would be different from that in the general population.
Dr. Pittas and colleagues caution that the people studied in this meta-analysis were at high risk for type 2 diabetes, so these results do not apply to the general healthy population. The results also should not be extrapolated to people at average risk for any type of diabetes, they add.
Several physicians either declined to comment or did not respond to requests for comment on this research.
Dr. Pittas reports the National Institutes of Health and the American Diabetes Association made payments to his institution to conduct Vitamin D-related research. He is an unpaid cochair of the Endocrine Society’s Evaluation, Treatment and Prevention of Vitamin D Deficiency Clinical Practice Guideline team.
Coauthor Dr. Jorde reports grants from Novo Nordisk Foundation, North Norwegian Regional Health Authorities, and the Research Council of Norway.
Dr. Dawson-Hughes reports she is on the DSMB for AgNovos Healthcare. AgNovos is developing a bone implant to reduce hip fracture risk and she gets a stipend from the company. She reports Helsinn Therapeutics provided anamorelin and matching placebo for an NIH-funded clinical trial.
Dr. Trikalinos was supported by the D2d study. He is a technical methodological consultant to Latham and Watkins, who is retained by Pacira Pharmaceuticals.
Dr. Angellotti has been employed by Takeda and owns stock in the company.
The editorialists report no relevant financial relationships.
FROM ANNALS OF INTERNAL MEDICINE
Primary care providers are increasingly addressing mental health concerns
particularly anxiety and stress-related diagnoses, based on a recent study.
These findings point to a sizable gap in psychiatric care that has likely been exacerbated by the pandemic, reported lead author Lisa S. Rotenstein, MD, MBA, assistant professor of medicine at Harvard Medical School and Medical Director of Population Health at Brigham and Women’s Hospital, both in Boston, and colleagues.
To ensure that PCPs can effectively manage this burden, innovative approaches are needed, such as value-based care models, billing codes for integrated behavioral health, and e-consultations with psychiatric colleagues, they added.
“Previous studies demonstrated that the rate of adult mental health outpatient visits increased between 1995 and 2010,” Dr. Rotenstein and colleagues wrote in Health Affairs. “However, more than a decade later, the extent to which the rate of primary care visits addressing mental health concerns has changed is unclear, with multiple health care delivery trends potentially influencing a further increase in prevalence.”
To address this knowledge gap, the investigators turned to the 2006-2018 National Ambulatory Medical Care Surveys, a nationally representative, serial, cross-sectional dataset. The present analysis included 109,898 visits representing 3,891,233,060 weighted visits.
Over the study period, the proportion of PCP visits that addressed mental health concerns rose from 10.7% to 15.9%.
This latter figure has probably increased since the onset of the pandemic, the investigators wrote, while availability of psychiatric care hasn’t kept pace, meaning PCPs are increasingly on the hook for managing mental illness.
“Even before the pandemic, one in five Americans lived with a mental health condition,” Dr. Rotenstein said in a written comment. “The COVID pandemic has only accelerated demand for mental health treatment. ... We know that there aren’t enough psychiatrists to meet this demand.”
Over the course of the study period, the rate of depression and affective disorders diagnoses slowed while anxiety and stress-related disorders were increasingly diagnosed.
“Particularly given the common co-occurrence of anxiety and depression, the trends we identified may represent physicians’ greater comfort over time with accurately diagnosing anxiety in the primary care setting, potentially for diagnoses that previously would have been classified as depression,” the investigators wrote, noting these findings align with a 2014 study by Olfson and colleagues.
Multiple factors associated with primary care mental health visits
Several variables were associated with significantly greater likelihood that a mental health concern would be addressed at a given visit, including female sex, younger age, payment via Medicare or Medicaid, and the physician being the patient’s regular physician.
“Our study demonstrated that mental health concerns were significantly more likely to be addressed in a visit with one’s usual primary care physician,” Dr. Rotenstein said. “This finding emphasizes the value of the longitudinal, supportive relationship developed in primary care for raising and addressing the full continuum of a patient’s needs, including mental health concerns.”
The investigators also observed significant associations between race/ethnicity and likelihood of addressing a mental health concern.
Compared with White patients, Black patients were 40% less likely to have a primary care visit with a mental health concern (odds ratio, 0.6; P less than .001). Similarly, Hispanic patients were 40% less likely than non-Hispanic patients to have a visit with a mental health concern (OR, 0.6; P less than .001).
“Unfortunately, our data don’t give us insight into why Black and Hispanic patients were less likely to have a mental health concern addressed in the context of a primary care visit,” Dr. Rotenstein said. “However, the data do suggest an urgent need to better understand and subsequently address the underlying causes of these disparities.”
She suggested several possible explanations, including differences in rates of screening, issues with access to care, insurance coverage disparities, and communication or cultural barriers.
Stuck in the reimbursement trap
Michael Klinkman, MD , professor of family medicine and learning health sciences at the University of Michigan Medical School, Ann Arbor, said the data align with his own clinical experience.
“The proportion of visits where depression was addresed went down, but the baseline is going up, so I don’t think we’re dealing with any less depression,” Dr. Klinkman said in an interview. “It’s just that there’s a lot more anxiety and stress that we’re finding and dealing with in primary care.”
While most family doctors are comfortable with best practices in managing these conditions, they may feel increasingly overburdened by the sheer number of patients with mental illness under their care alone, according to Dr. Klinkman.
“Primary care docs are increasingly feeling like they’re on their own in dealing with mental health problems,” he said.
While he agreed in theory with the interventions proposed by Dr. Rotenstein and colleagues, some solutions, like billing code changes, may ultimately worsen the burden on primary care providers.
“My fear in all of this, frankly, is that we’re going to create a better sense of the need for primary care practice in general to address mental health and social care issues, and we’re just going to create a lot more work and more widget-counting around doing that,” said Dr. Klinkman.
Value-based care appears to be a better solution, he said, since “we’re trying to take care of a human being, not the 1,050 pieces of that human being’s care that we’re trying to bundle up with different codes.”
A flat-fee, per-patient model, however, is unlikely to gain traction in the United States.
Dr. Klinkman has been involved in health care system reform up to the federal level, where he has encountered politicians who understood the issues but were incapable of helping because of partisan gridlock, he said. “It’s just politically near impossible to make changes in this basic health care business model.”
Policymakers advised Dr. Klinkman and his colleagues to strive for incremental changes, leaving them to grapple with increasingly complex reimbursement rules.
“We’re kind of stuck in this trap of trying to create new codes for services that we think ought to be better reimbursed,” Dr. Klinkman said. “We’re missing the person in all of this – the human being we’re trying to serve.”
The investigators, Dr. Cain, and Dr. Klinkman disclosed no conflicts of interest.
*This article was updated on 2/27/2023.
particularly anxiety and stress-related diagnoses, based on a recent study.
These findings point to a sizable gap in psychiatric care that has likely been exacerbated by the pandemic, reported lead author Lisa S. Rotenstein, MD, MBA, assistant professor of medicine at Harvard Medical School and Medical Director of Population Health at Brigham and Women’s Hospital, both in Boston, and colleagues.
To ensure that PCPs can effectively manage this burden, innovative approaches are needed, such as value-based care models, billing codes for integrated behavioral health, and e-consultations with psychiatric colleagues, they added.
“Previous studies demonstrated that the rate of adult mental health outpatient visits increased between 1995 and 2010,” Dr. Rotenstein and colleagues wrote in Health Affairs. “However, more than a decade later, the extent to which the rate of primary care visits addressing mental health concerns has changed is unclear, with multiple health care delivery trends potentially influencing a further increase in prevalence.”
To address this knowledge gap, the investigators turned to the 2006-2018 National Ambulatory Medical Care Surveys, a nationally representative, serial, cross-sectional dataset. The present analysis included 109,898 visits representing 3,891,233,060 weighted visits.
Over the study period, the proportion of PCP visits that addressed mental health concerns rose from 10.7% to 15.9%.
This latter figure has probably increased since the onset of the pandemic, the investigators wrote, while availability of psychiatric care hasn’t kept pace, meaning PCPs are increasingly on the hook for managing mental illness.
“Even before the pandemic, one in five Americans lived with a mental health condition,” Dr. Rotenstein said in a written comment. “The COVID pandemic has only accelerated demand for mental health treatment. ... We know that there aren’t enough psychiatrists to meet this demand.”
Over the course of the study period, the rate of depression and affective disorders diagnoses slowed while anxiety and stress-related disorders were increasingly diagnosed.
“Particularly given the common co-occurrence of anxiety and depression, the trends we identified may represent physicians’ greater comfort over time with accurately diagnosing anxiety in the primary care setting, potentially for diagnoses that previously would have been classified as depression,” the investigators wrote, noting these findings align with a 2014 study by Olfson and colleagues.
Multiple factors associated with primary care mental health visits
Several variables were associated with significantly greater likelihood that a mental health concern would be addressed at a given visit, including female sex, younger age, payment via Medicare or Medicaid, and the physician being the patient’s regular physician.
“Our study demonstrated that mental health concerns were significantly more likely to be addressed in a visit with one’s usual primary care physician,” Dr. Rotenstein said. “This finding emphasizes the value of the longitudinal, supportive relationship developed in primary care for raising and addressing the full continuum of a patient’s needs, including mental health concerns.”
The investigators also observed significant associations between race/ethnicity and likelihood of addressing a mental health concern.
Compared with White patients, Black patients were 40% less likely to have a primary care visit with a mental health concern (odds ratio, 0.6; P less than .001). Similarly, Hispanic patients were 40% less likely than non-Hispanic patients to have a visit with a mental health concern (OR, 0.6; P less than .001).
“Unfortunately, our data don’t give us insight into why Black and Hispanic patients were less likely to have a mental health concern addressed in the context of a primary care visit,” Dr. Rotenstein said. “However, the data do suggest an urgent need to better understand and subsequently address the underlying causes of these disparities.”
She suggested several possible explanations, including differences in rates of screening, issues with access to care, insurance coverage disparities, and communication or cultural barriers.
Stuck in the reimbursement trap
Michael Klinkman, MD , professor of family medicine and learning health sciences at the University of Michigan Medical School, Ann Arbor, said the data align with his own clinical experience.
“The proportion of visits where depression was addresed went down, but the baseline is going up, so I don’t think we’re dealing with any less depression,” Dr. Klinkman said in an interview. “It’s just that there’s a lot more anxiety and stress that we’re finding and dealing with in primary care.”
While most family doctors are comfortable with best practices in managing these conditions, they may feel increasingly overburdened by the sheer number of patients with mental illness under their care alone, according to Dr. Klinkman.
“Primary care docs are increasingly feeling like they’re on their own in dealing with mental health problems,” he said.
While he agreed in theory with the interventions proposed by Dr. Rotenstein and colleagues, some solutions, like billing code changes, may ultimately worsen the burden on primary care providers.
“My fear in all of this, frankly, is that we’re going to create a better sense of the need for primary care practice in general to address mental health and social care issues, and we’re just going to create a lot more work and more widget-counting around doing that,” said Dr. Klinkman.
Value-based care appears to be a better solution, he said, since “we’re trying to take care of a human being, not the 1,050 pieces of that human being’s care that we’re trying to bundle up with different codes.”
A flat-fee, per-patient model, however, is unlikely to gain traction in the United States.
Dr. Klinkman has been involved in health care system reform up to the federal level, where he has encountered politicians who understood the issues but were incapable of helping because of partisan gridlock, he said. “It’s just politically near impossible to make changes in this basic health care business model.”
Policymakers advised Dr. Klinkman and his colleagues to strive for incremental changes, leaving them to grapple with increasingly complex reimbursement rules.
“We’re kind of stuck in this trap of trying to create new codes for services that we think ought to be better reimbursed,” Dr. Klinkman said. “We’re missing the person in all of this – the human being we’re trying to serve.”
The investigators, Dr. Cain, and Dr. Klinkman disclosed no conflicts of interest.
*This article was updated on 2/27/2023.
particularly anxiety and stress-related diagnoses, based on a recent study.
These findings point to a sizable gap in psychiatric care that has likely been exacerbated by the pandemic, reported lead author Lisa S. Rotenstein, MD, MBA, assistant professor of medicine at Harvard Medical School and Medical Director of Population Health at Brigham and Women’s Hospital, both in Boston, and colleagues.
To ensure that PCPs can effectively manage this burden, innovative approaches are needed, such as value-based care models, billing codes for integrated behavioral health, and e-consultations with psychiatric colleagues, they added.
“Previous studies demonstrated that the rate of adult mental health outpatient visits increased between 1995 and 2010,” Dr. Rotenstein and colleagues wrote in Health Affairs. “However, more than a decade later, the extent to which the rate of primary care visits addressing mental health concerns has changed is unclear, with multiple health care delivery trends potentially influencing a further increase in prevalence.”
To address this knowledge gap, the investigators turned to the 2006-2018 National Ambulatory Medical Care Surveys, a nationally representative, serial, cross-sectional dataset. The present analysis included 109,898 visits representing 3,891,233,060 weighted visits.
Over the study period, the proportion of PCP visits that addressed mental health concerns rose from 10.7% to 15.9%.
This latter figure has probably increased since the onset of the pandemic, the investigators wrote, while availability of psychiatric care hasn’t kept pace, meaning PCPs are increasingly on the hook for managing mental illness.
“Even before the pandemic, one in five Americans lived with a mental health condition,” Dr. Rotenstein said in a written comment. “The COVID pandemic has only accelerated demand for mental health treatment. ... We know that there aren’t enough psychiatrists to meet this demand.”
Over the course of the study period, the rate of depression and affective disorders diagnoses slowed while anxiety and stress-related disorders were increasingly diagnosed.
“Particularly given the common co-occurrence of anxiety and depression, the trends we identified may represent physicians’ greater comfort over time with accurately diagnosing anxiety in the primary care setting, potentially for diagnoses that previously would have been classified as depression,” the investigators wrote, noting these findings align with a 2014 study by Olfson and colleagues.
Multiple factors associated with primary care mental health visits
Several variables were associated with significantly greater likelihood that a mental health concern would be addressed at a given visit, including female sex, younger age, payment via Medicare or Medicaid, and the physician being the patient’s regular physician.
“Our study demonstrated that mental health concerns were significantly more likely to be addressed in a visit with one’s usual primary care physician,” Dr. Rotenstein said. “This finding emphasizes the value of the longitudinal, supportive relationship developed in primary care for raising and addressing the full continuum of a patient’s needs, including mental health concerns.”
The investigators also observed significant associations between race/ethnicity and likelihood of addressing a mental health concern.
Compared with White patients, Black patients were 40% less likely to have a primary care visit with a mental health concern (odds ratio, 0.6; P less than .001). Similarly, Hispanic patients were 40% less likely than non-Hispanic patients to have a visit with a mental health concern (OR, 0.6; P less than .001).
“Unfortunately, our data don’t give us insight into why Black and Hispanic patients were less likely to have a mental health concern addressed in the context of a primary care visit,” Dr. Rotenstein said. “However, the data do suggest an urgent need to better understand and subsequently address the underlying causes of these disparities.”
She suggested several possible explanations, including differences in rates of screening, issues with access to care, insurance coverage disparities, and communication or cultural barriers.
Stuck in the reimbursement trap
Michael Klinkman, MD , professor of family medicine and learning health sciences at the University of Michigan Medical School, Ann Arbor, said the data align with his own clinical experience.
“The proportion of visits where depression was addresed went down, but the baseline is going up, so I don’t think we’re dealing with any less depression,” Dr. Klinkman said in an interview. “It’s just that there’s a lot more anxiety and stress that we’re finding and dealing with in primary care.”
While most family doctors are comfortable with best practices in managing these conditions, they may feel increasingly overburdened by the sheer number of patients with mental illness under their care alone, according to Dr. Klinkman.
“Primary care docs are increasingly feeling like they’re on their own in dealing with mental health problems,” he said.
While he agreed in theory with the interventions proposed by Dr. Rotenstein and colleagues, some solutions, like billing code changes, may ultimately worsen the burden on primary care providers.
“My fear in all of this, frankly, is that we’re going to create a better sense of the need for primary care practice in general to address mental health and social care issues, and we’re just going to create a lot more work and more widget-counting around doing that,” said Dr. Klinkman.
Value-based care appears to be a better solution, he said, since “we’re trying to take care of a human being, not the 1,050 pieces of that human being’s care that we’re trying to bundle up with different codes.”
A flat-fee, per-patient model, however, is unlikely to gain traction in the United States.
Dr. Klinkman has been involved in health care system reform up to the federal level, where he has encountered politicians who understood the issues but were incapable of helping because of partisan gridlock, he said. “It’s just politically near impossible to make changes in this basic health care business model.”
Policymakers advised Dr. Klinkman and his colleagues to strive for incremental changes, leaving them to grapple with increasingly complex reimbursement rules.
“We’re kind of stuck in this trap of trying to create new codes for services that we think ought to be better reimbursed,” Dr. Klinkman said. “We’re missing the person in all of this – the human being we’re trying to serve.”
The investigators, Dr. Cain, and Dr. Klinkman disclosed no conflicts of interest.
*This article was updated on 2/27/2023.
FROM HEALTH AFFAIRS
Consider cultural differences in IBD diet planning
AURORA, COLO. – Inflammatory bowel disease doesn’t respect international borders, and
“Many patients are in an environment that they’re not used to, an environment where most people speak English and their customs and their language may differ from the individual providing care to them. They’re often told, in addition, to eat foods that they may not even have heard of. It can really be a scary situation for many of these patients,” said Neha D. Shah, MPH, RD, CNSC, a dietitian at University of California San Francisco Health.
“Put yourself in their shoes. [Consider] what would make you feel more comfortable in that environment, and then apply that perspective to the care of your patient,” she advised colleagues at the annual Crohn’s & Colitis Congress®, a partnership of the Crohn’s & Colitis Foundation and the American Gastroenterological Association.
Ms. Shah explained that by incorporating understanding of cultural differences and food culture into the care of persons with IBD, clinicians can help patients from different ethnic backgrounds accept diets that both contain familiar foods and also help to ameliorate their gastrointestinal symptoms.
Food culture and acculturation
As of 2016, the estimated prevalence of IBD among pediatric patients in the United States was 77 per 100,000, and the prevalence in adults was estimated at 478.4 per 100,000. In a 2021 study of the effects of race and ethnicity on the diagnosis and management of IBD, the authors estimated that the prevalence of IBD in the United States was about 3.1 million persons, or 1.3% of the population, with an increase in prevalence in non-White persons and ethnicities, she noted.
Some of the increasing prevalence among minority populations may be attributable to diet acculturation, when members of a particular group partially or completely adopt the eating patterns and/or food choices of the host country.
Culturally appropriate foods
The term “food culture” refers to “the sociocultural aspect of eating, and include[s] the beliefs, values, and attitudes a community may accept around food,” she said.
Ms. Shah provided examples of culturally appropriate foods that may be tolerated by patients with IBD, such as beans, tortillas, chicken with rice, guacamole, mangos, and tomatoes in persons from South America, or lentils, breads, rice, oats, spinach, and tea among patients from the Indian subcontinent.
By understanding and respecting cultural differences, learning how to best communicate with persons of other cultures, and by being aware of one’s own biases, clinicians can better help patients create diet plans that fit within their expectations and lifestyles, she said.
For example, patients can be encouraged to incorporate more culturally familiar plant-based foods such as legumes to manage active disease and maintain remissions.
Patients with active disease should have at least one-half cup of one form of culturally appropriate fiber at each meal. The dietitian should consider recommending blending fiber into other foods or serving it cooked, mashed, or minced, depending upon the patient’s level of tolerance.
During the transition phase, patients can reintroduce an additional half cup of fiber at one meal, then at two meals, and finally at three daily meals. Patients can see whether they can tolerate more raw or whole high-fiber foods at this stage.
During remissions, patients should be advised to add two to three foods containing culturally appropriate fiber at each meal, she said.
‘Eye-opening’ realization
“I think it’s really eye-opening for us to think about how we have to have culturally sensitive discussions with our patients,” commented Sandra Kim, MD, from the University of Pittsburgh Medical Center, who moderated the session.
Dr. Kim asked Ms. Shah what advice she’d give to pediatric gastroenterologists about engaging patients and their families.
The clinician should ask both patients and parents about what the child eats and what the challenges of eating under certain circumstances are, and have culturally appropriate resources on hand.
Ms. Shah did not report a funding source for her work. She disclosed compensation as editor of the Journal of Practical Gastroeneterology and as GI on Demand–consultant for a joint virtual platform from the American College of Gastroenterology and Gastro Girl. She also serves as treasurer and director of operations for the South Asian IBD Alliance.
AURORA, COLO. – Inflammatory bowel disease doesn’t respect international borders, and
“Many patients are in an environment that they’re not used to, an environment where most people speak English and their customs and their language may differ from the individual providing care to them. They’re often told, in addition, to eat foods that they may not even have heard of. It can really be a scary situation for many of these patients,” said Neha D. Shah, MPH, RD, CNSC, a dietitian at University of California San Francisco Health.
“Put yourself in their shoes. [Consider] what would make you feel more comfortable in that environment, and then apply that perspective to the care of your patient,” she advised colleagues at the annual Crohn’s & Colitis Congress®, a partnership of the Crohn’s & Colitis Foundation and the American Gastroenterological Association.
Ms. Shah explained that by incorporating understanding of cultural differences and food culture into the care of persons with IBD, clinicians can help patients from different ethnic backgrounds accept diets that both contain familiar foods and also help to ameliorate their gastrointestinal symptoms.
Food culture and acculturation
As of 2016, the estimated prevalence of IBD among pediatric patients in the United States was 77 per 100,000, and the prevalence in adults was estimated at 478.4 per 100,000. In a 2021 study of the effects of race and ethnicity on the diagnosis and management of IBD, the authors estimated that the prevalence of IBD in the United States was about 3.1 million persons, or 1.3% of the population, with an increase in prevalence in non-White persons and ethnicities, she noted.
Some of the increasing prevalence among minority populations may be attributable to diet acculturation, when members of a particular group partially or completely adopt the eating patterns and/or food choices of the host country.
Culturally appropriate foods
The term “food culture” refers to “the sociocultural aspect of eating, and include[s] the beliefs, values, and attitudes a community may accept around food,” she said.
Ms. Shah provided examples of culturally appropriate foods that may be tolerated by patients with IBD, such as beans, tortillas, chicken with rice, guacamole, mangos, and tomatoes in persons from South America, or lentils, breads, rice, oats, spinach, and tea among patients from the Indian subcontinent.
By understanding and respecting cultural differences, learning how to best communicate with persons of other cultures, and by being aware of one’s own biases, clinicians can better help patients create diet plans that fit within their expectations and lifestyles, she said.
For example, patients can be encouraged to incorporate more culturally familiar plant-based foods such as legumes to manage active disease and maintain remissions.
Patients with active disease should have at least one-half cup of one form of culturally appropriate fiber at each meal. The dietitian should consider recommending blending fiber into other foods or serving it cooked, mashed, or minced, depending upon the patient’s level of tolerance.
During the transition phase, patients can reintroduce an additional half cup of fiber at one meal, then at two meals, and finally at three daily meals. Patients can see whether they can tolerate more raw or whole high-fiber foods at this stage.
During remissions, patients should be advised to add two to three foods containing culturally appropriate fiber at each meal, she said.
‘Eye-opening’ realization
“I think it’s really eye-opening for us to think about how we have to have culturally sensitive discussions with our patients,” commented Sandra Kim, MD, from the University of Pittsburgh Medical Center, who moderated the session.
Dr. Kim asked Ms. Shah what advice she’d give to pediatric gastroenterologists about engaging patients and their families.
The clinician should ask both patients and parents about what the child eats and what the challenges of eating under certain circumstances are, and have culturally appropriate resources on hand.
Ms. Shah did not report a funding source for her work. She disclosed compensation as editor of the Journal of Practical Gastroeneterology and as GI on Demand–consultant for a joint virtual platform from the American College of Gastroenterology and Gastro Girl. She also serves as treasurer and director of operations for the South Asian IBD Alliance.
AURORA, COLO. – Inflammatory bowel disease doesn’t respect international borders, and
“Many patients are in an environment that they’re not used to, an environment where most people speak English and their customs and their language may differ from the individual providing care to them. They’re often told, in addition, to eat foods that they may not even have heard of. It can really be a scary situation for many of these patients,” said Neha D. Shah, MPH, RD, CNSC, a dietitian at University of California San Francisco Health.
“Put yourself in their shoes. [Consider] what would make you feel more comfortable in that environment, and then apply that perspective to the care of your patient,” she advised colleagues at the annual Crohn’s & Colitis Congress®, a partnership of the Crohn’s & Colitis Foundation and the American Gastroenterological Association.
Ms. Shah explained that by incorporating understanding of cultural differences and food culture into the care of persons with IBD, clinicians can help patients from different ethnic backgrounds accept diets that both contain familiar foods and also help to ameliorate their gastrointestinal symptoms.
Food culture and acculturation
As of 2016, the estimated prevalence of IBD among pediatric patients in the United States was 77 per 100,000, and the prevalence in adults was estimated at 478.4 per 100,000. In a 2021 study of the effects of race and ethnicity on the diagnosis and management of IBD, the authors estimated that the prevalence of IBD in the United States was about 3.1 million persons, or 1.3% of the population, with an increase in prevalence in non-White persons and ethnicities, she noted.
Some of the increasing prevalence among minority populations may be attributable to diet acculturation, when members of a particular group partially or completely adopt the eating patterns and/or food choices of the host country.
Culturally appropriate foods
The term “food culture” refers to “the sociocultural aspect of eating, and include[s] the beliefs, values, and attitudes a community may accept around food,” she said.
Ms. Shah provided examples of culturally appropriate foods that may be tolerated by patients with IBD, such as beans, tortillas, chicken with rice, guacamole, mangos, and tomatoes in persons from South America, or lentils, breads, rice, oats, spinach, and tea among patients from the Indian subcontinent.
By understanding and respecting cultural differences, learning how to best communicate with persons of other cultures, and by being aware of one’s own biases, clinicians can better help patients create diet plans that fit within their expectations and lifestyles, she said.
For example, patients can be encouraged to incorporate more culturally familiar plant-based foods such as legumes to manage active disease and maintain remissions.
Patients with active disease should have at least one-half cup of one form of culturally appropriate fiber at each meal. The dietitian should consider recommending blending fiber into other foods or serving it cooked, mashed, or minced, depending upon the patient’s level of tolerance.
During the transition phase, patients can reintroduce an additional half cup of fiber at one meal, then at two meals, and finally at three daily meals. Patients can see whether they can tolerate more raw or whole high-fiber foods at this stage.
During remissions, patients should be advised to add two to three foods containing culturally appropriate fiber at each meal, she said.
‘Eye-opening’ realization
“I think it’s really eye-opening for us to think about how we have to have culturally sensitive discussions with our patients,” commented Sandra Kim, MD, from the University of Pittsburgh Medical Center, who moderated the session.
Dr. Kim asked Ms. Shah what advice she’d give to pediatric gastroenterologists about engaging patients and their families.
The clinician should ask both patients and parents about what the child eats and what the challenges of eating under certain circumstances are, and have culturally appropriate resources on hand.
Ms. Shah did not report a funding source for her work. She disclosed compensation as editor of the Journal of Practical Gastroeneterology and as GI on Demand–consultant for a joint virtual platform from the American College of Gastroenterology and Gastro Girl. She also serves as treasurer and director of operations for the South Asian IBD Alliance.
AT THE CROHN’S & COLITIS CONGRESS
Go outside: It’s good for you
“Go outside and play!”
How many times did your mother tell you that?
Turns out that, like with chicken soup, she was right.
A recent article in Occupational and Environmental Medicine found that urban dwellers who spent time outdoors in green areas, such as parks and forests, had lower use of antihypertensive, antidepressant, and antianxiety medications than those who didn’t. People who just looked at such areas from a window didn’t have lower medication use than those who weren’t exposed to them at all.
In other words:
1. Being outside in nature is good for you.
2. Staring at a forest on your screensaver isn’t a substitute.
But this shouldn’t really surprise anyone.
While I’m not going to say our technologically driven lifestyles are a bad thing - they’ve certainly made many parts of our lives easier – they do tend to take us away from other activities. As I’ve mentioned in this column before, there’s no shortage of recent studies suggesting social media use and regularly following the news are bad for you. These are things that become emotionally taxing and upsetting, increasing anxiety, raising blood pressure, and increasing stress eating and other not-so-good-for-you consequences.
Of course, this is easier said than done. Getting outside takes time, something it seems we have precious little of these days. Our lives are constantly in a rush from the house to the office to the hospital to the store to the house. Wash, rinse, repeat. Breaking the cycle is harder than just going along with it.
Yet, as these things show, . While I still enjoy my job, I’m obviously not the only one who needs to step back and go to the park, or hiking trail, or whatever.
Studies agree that it’s a good idea. And my mom told me to.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“Go outside and play!”
How many times did your mother tell you that?
Turns out that, like with chicken soup, she was right.
A recent article in Occupational and Environmental Medicine found that urban dwellers who spent time outdoors in green areas, such as parks and forests, had lower use of antihypertensive, antidepressant, and antianxiety medications than those who didn’t. People who just looked at such areas from a window didn’t have lower medication use than those who weren’t exposed to them at all.
In other words:
1. Being outside in nature is good for you.
2. Staring at a forest on your screensaver isn’t a substitute.
But this shouldn’t really surprise anyone.
While I’m not going to say our technologically driven lifestyles are a bad thing - they’ve certainly made many parts of our lives easier – they do tend to take us away from other activities. As I’ve mentioned in this column before, there’s no shortage of recent studies suggesting social media use and regularly following the news are bad for you. These are things that become emotionally taxing and upsetting, increasing anxiety, raising blood pressure, and increasing stress eating and other not-so-good-for-you consequences.
Of course, this is easier said than done. Getting outside takes time, something it seems we have precious little of these days. Our lives are constantly in a rush from the house to the office to the hospital to the store to the house. Wash, rinse, repeat. Breaking the cycle is harder than just going along with it.
Yet, as these things show, . While I still enjoy my job, I’m obviously not the only one who needs to step back and go to the park, or hiking trail, or whatever.
Studies agree that it’s a good idea. And my mom told me to.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
“Go outside and play!”
How many times did your mother tell you that?
Turns out that, like with chicken soup, she was right.
A recent article in Occupational and Environmental Medicine found that urban dwellers who spent time outdoors in green areas, such as parks and forests, had lower use of antihypertensive, antidepressant, and antianxiety medications than those who didn’t. People who just looked at such areas from a window didn’t have lower medication use than those who weren’t exposed to them at all.
In other words:
1. Being outside in nature is good for you.
2. Staring at a forest on your screensaver isn’t a substitute.
But this shouldn’t really surprise anyone.
While I’m not going to say our technologically driven lifestyles are a bad thing - they’ve certainly made many parts of our lives easier – they do tend to take us away from other activities. As I’ve mentioned in this column before, there’s no shortage of recent studies suggesting social media use and regularly following the news are bad for you. These are things that become emotionally taxing and upsetting, increasing anxiety, raising blood pressure, and increasing stress eating and other not-so-good-for-you consequences.
Of course, this is easier said than done. Getting outside takes time, something it seems we have precious little of these days. Our lives are constantly in a rush from the house to the office to the hospital to the store to the house. Wash, rinse, repeat. Breaking the cycle is harder than just going along with it.
Yet, as these things show, . While I still enjoy my job, I’m obviously not the only one who needs to step back and go to the park, or hiking trail, or whatever.
Studies agree that it’s a good idea. And my mom told me to.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Systemic sclerosis antibodies show link to interstitial lung disease in RA
Adults with rheumatoid arthritis or primary Sjogren’s syndrome plus interstitial lung disease had higher levels of systemic sclerosis–specific antibodies than those without lung disease, based on data from 101 individuals.
Systemic sclerosis (SSc) has been associated with the development of interstitial lung disease (ILD), but the prevalence of SSc autoantibodies in patients with rheumatoid arthritis (RA) and primary Sjogren’s syndrome (SS) has not been explored, wrote Vasilike Koulouri, MD, of Kapodistrian University of Athens, and colleagues.
In a study published in the Journal of Translational Autoimmunity, the researchers reviewed serum data from patients with RA and SS using immunoblot assays to determine the prevalence of SSc-specific and anti-Ro52 autoantibodies, both of which have been associated with ILD in SSc patients.
The study population included 28 RA patients with ILD, 32 RA patients without ILD, 9 primary SS patients with ILD, and 32 primary SS patients with no ILD. The mean age of the RA participants was 63.4 years, 70% were women, and the mean age at RA diagnosis was 50.2 years. The mean age of the primary SS group was 60.3 years, 87.8% were female, and the mean age at diagnosis was 52.7 years.
Overall, SSc-specific antibodies across all titers were detected more frequently in RA patients with ILD compared with those with no ILD, though not statistically significant (42.9% vs. 21.9%, P = .08). However, “This trend was mainly attributed to the statistically significant difference between the two groups at strong titers (25% vs. 3.1%, P = .01),” the researchers wrote. Notably, they added.
No significant differences appeared in the prevalence of SSc-specific or Ro52 autoantibodies between primary SS patients with and without ILD, which might be attributable in part to the increased prevalence of anticentromere antibodies in primary SS, the researchers said.
RA patients who were positive for SSc-specific antibodies at strong titers were significantly more likely to have respiratory abnormalities than those who were negative (87.5% vs. 47.2%, P = .04), but no such differences appeared in primary SS patients.
“Early detection of SSc antibodies could be important in clinical practice as it may mandate further diagnostic (for example, screening for pulmonary hypertension) and therapeutic approaches of these patients,” the researchers wrote in their discussion.
The study findings were limited by several factors, mainly the small sample size, but also the potential for false-positive results on antibody titers, lack of data on the clinical significance of medium autoantibody titers, and the lack of long-term follow-up data, the researchers noted.
However, the results suggest that many seropositive RA patients with evidence of ILD “may evolve to a clinically evident overlap of RA and SSc” that would benefit from targeted treatment, they concluded.
The study was supported by a grant from Novartis AG and by the Molecular Immunology and Clinical Applications Unit, Department of Physiology, School of Medicine, National and Kapodistrian University of Athens. The researchers had no financial conflicts to disclose.
Adults with rheumatoid arthritis or primary Sjogren’s syndrome plus interstitial lung disease had higher levels of systemic sclerosis–specific antibodies than those without lung disease, based on data from 101 individuals.
Systemic sclerosis (SSc) has been associated with the development of interstitial lung disease (ILD), but the prevalence of SSc autoantibodies in patients with rheumatoid arthritis (RA) and primary Sjogren’s syndrome (SS) has not been explored, wrote Vasilike Koulouri, MD, of Kapodistrian University of Athens, and colleagues.
In a study published in the Journal of Translational Autoimmunity, the researchers reviewed serum data from patients with RA and SS using immunoblot assays to determine the prevalence of SSc-specific and anti-Ro52 autoantibodies, both of which have been associated with ILD in SSc patients.
The study population included 28 RA patients with ILD, 32 RA patients without ILD, 9 primary SS patients with ILD, and 32 primary SS patients with no ILD. The mean age of the RA participants was 63.4 years, 70% were women, and the mean age at RA diagnosis was 50.2 years. The mean age of the primary SS group was 60.3 years, 87.8% were female, and the mean age at diagnosis was 52.7 years.
Overall, SSc-specific antibodies across all titers were detected more frequently in RA patients with ILD compared with those with no ILD, though not statistically significant (42.9% vs. 21.9%, P = .08). However, “This trend was mainly attributed to the statistically significant difference between the two groups at strong titers (25% vs. 3.1%, P = .01),” the researchers wrote. Notably, they added.
No significant differences appeared in the prevalence of SSc-specific or Ro52 autoantibodies between primary SS patients with and without ILD, which might be attributable in part to the increased prevalence of anticentromere antibodies in primary SS, the researchers said.
RA patients who were positive for SSc-specific antibodies at strong titers were significantly more likely to have respiratory abnormalities than those who were negative (87.5% vs. 47.2%, P = .04), but no such differences appeared in primary SS patients.
“Early detection of SSc antibodies could be important in clinical practice as it may mandate further diagnostic (for example, screening for pulmonary hypertension) and therapeutic approaches of these patients,” the researchers wrote in their discussion.
The study findings were limited by several factors, mainly the small sample size, but also the potential for false-positive results on antibody titers, lack of data on the clinical significance of medium autoantibody titers, and the lack of long-term follow-up data, the researchers noted.
However, the results suggest that many seropositive RA patients with evidence of ILD “may evolve to a clinically evident overlap of RA and SSc” that would benefit from targeted treatment, they concluded.
The study was supported by a grant from Novartis AG and by the Molecular Immunology and Clinical Applications Unit, Department of Physiology, School of Medicine, National and Kapodistrian University of Athens. The researchers had no financial conflicts to disclose.
Adults with rheumatoid arthritis or primary Sjogren’s syndrome plus interstitial lung disease had higher levels of systemic sclerosis–specific antibodies than those without lung disease, based on data from 101 individuals.
Systemic sclerosis (SSc) has been associated with the development of interstitial lung disease (ILD), but the prevalence of SSc autoantibodies in patients with rheumatoid arthritis (RA) and primary Sjogren’s syndrome (SS) has not been explored, wrote Vasilike Koulouri, MD, of Kapodistrian University of Athens, and colleagues.
In a study published in the Journal of Translational Autoimmunity, the researchers reviewed serum data from patients with RA and SS using immunoblot assays to determine the prevalence of SSc-specific and anti-Ro52 autoantibodies, both of which have been associated with ILD in SSc patients.
The study population included 28 RA patients with ILD, 32 RA patients without ILD, 9 primary SS patients with ILD, and 32 primary SS patients with no ILD. The mean age of the RA participants was 63.4 years, 70% were women, and the mean age at RA diagnosis was 50.2 years. The mean age of the primary SS group was 60.3 years, 87.8% were female, and the mean age at diagnosis was 52.7 years.
Overall, SSc-specific antibodies across all titers were detected more frequently in RA patients with ILD compared with those with no ILD, though not statistically significant (42.9% vs. 21.9%, P = .08). However, “This trend was mainly attributed to the statistically significant difference between the two groups at strong titers (25% vs. 3.1%, P = .01),” the researchers wrote. Notably, they added.
No significant differences appeared in the prevalence of SSc-specific or Ro52 autoantibodies between primary SS patients with and without ILD, which might be attributable in part to the increased prevalence of anticentromere antibodies in primary SS, the researchers said.
RA patients who were positive for SSc-specific antibodies at strong titers were significantly more likely to have respiratory abnormalities than those who were negative (87.5% vs. 47.2%, P = .04), but no such differences appeared in primary SS patients.
“Early detection of SSc antibodies could be important in clinical practice as it may mandate further diagnostic (for example, screening for pulmonary hypertension) and therapeutic approaches of these patients,” the researchers wrote in their discussion.
The study findings were limited by several factors, mainly the small sample size, but also the potential for false-positive results on antibody titers, lack of data on the clinical significance of medium autoantibody titers, and the lack of long-term follow-up data, the researchers noted.
However, the results suggest that many seropositive RA patients with evidence of ILD “may evolve to a clinically evident overlap of RA and SSc” that would benefit from targeted treatment, they concluded.
The study was supported by a grant from Novartis AG and by the Molecular Immunology and Clinical Applications Unit, Department of Physiology, School of Medicine, National and Kapodistrian University of Athens. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF TRANSLATIONAL AUTOIMMUNITY
Remote electrical neuromodulation device helps reduce migraine days
, according to recent research published in the journal Headache.
The prospective, randomized, double-blind, placebo-controlled, multicenter trial showed that remote electrical neuromodulation (REN) with Nerivio (Theranica Bio-Electronics Ltd.; Bridgewater, N.J.) found a mean reduction/decrease in the number of migraine days by an average of 4.0 days per month, according to Stewart J. Tepper MD, of the Geisel School of Medicine at Dartmouth in Hanover, N.H., and colleagues.*
“The statistically significant results were maintained in separate subanalyses of the chronic and episodic subsamples, as well as in the separate subanalyses of participants who used and did not use migraine prophylaxis,” Dr. Tepper and colleagues wrote.
A nonpharmacological alternative
Researchers randomized 248 participants into active and placebo groups, with 95 participants in the active group and 84 participants in the placebo group meeting the criteria for a modified intention-to-treat (mITT) analysis. Most of the participants in the ITT dataset were women (85.9%) with an average age of 41.7 years, and a baseline average of 12.2 migraine days and 15.6 headache days. Overall, 52.4% of participants in the ITT dataset had chronic migraine, 25.0% had migraine with aura, and 41.1% were taking preventative medication.
Dr. Tepper and colleagues followed participants for 4 weeks at baseline for observation followed by 8 weeks of participants using the REN device every other day for 45 minutes, or a placebo device that “produces electrical pulses of the same maximum intensity (34 mA) and overall energy, but with different pulse durations and much lower frequencies compared with the active device.” Participants completed a daily diary where they recorded their symptoms.
Researchers assessed the mean change in number of migraine days per month as a primary outcome, and evaluated participants who experienced episodic and chronic migraines separately in subgroup analyses. Secondary outcome measures included mean change in number of moderate or severe headache days, 50% reduction in mean number of headache days compared with baseline, Headache Impact Test short form (HIT-6) and Migraine Specific Quality of Life Questionnaire (MSQ) Role Function Domain total score mean change at 12 weeks compared with week 1, and reduction in mean number of days taking acute headache or migraine medication.
Participants receiving REN treatment had a significant reduction in mean migraine days per month compared with the placebo group (4.0 days vs. 1.3 days; 95% confidence interval, –3.9 days to –1.5 days; P < .001). In subgroup analyses, a significant reduction in migraine days was seen in participants receiving REN treatment with episodic migraine (3.2 days vs. 1.0 days; P = .003) and chronic migraine (4.7 days vs. 1.6 days; P = .001) compared with placebo.
Dr. Tepper and colleagues found a significant reduction in moderate and/or severe headache days among participants receiving REN treatment compared with placebo (3.8 days vs. 2.2 days; P = .005), a significant reduction in headache days overall compared with placebo (4.5 days vs. 1.8 days; P < .001), a significant percentage of patients who experienced 50% reduction in moderate and/or severe headache days compared with placebo (51.6% vs. 35.7%; P = .033), and a significant reduction in acute medication days compared with placebo (3.5 days vs. 1.4 days; P = .001). Dr. Tepper and colleagues found no serious device-related adverse events in either group.
The researchers noted that REN therapy is a “much-needed nonpharmacological alternative” to other preventive and acute treatments for migraine. “Given the previously well-established clinical efficacy and high safety profile in acute treatment of migraine, REN can cover the entire treatment spectrum of migraine, including both acute and preventive treatments,” they said.
‘A good place to start’
Commenting on the study, Alan M. Rapoport, MD, clinical professor of neurology at University of California, Los Angeles; past president of the International Headache Society; and editor-in-chief of Neurology Reviews, said the study was well designed, but acknowledged the 8-week follow-up time for participants as one potential area where he would have wanted to see more data.
As a medical device cleared for use by the Food and Drug Administration for acute treatment of migraine, the REM device also appears to be effective as a migraine preventative based on the results of the study with “virtually no adverse events,” he noted.
“I think this is a great treatment. I think it’s a good place to start,” Dr. Rapoport said. Given the low adverse event rate, he said he would be willing to offer the device to patients as a first option for preventing migraine and either switch to another preventative option or add an additional medication in combination based on how the patient responds. However, at the moment, he noted that this device is not covered by insurance.
Now that a REN device has been shown to work in the acute setting and as a preventative, Dr. Rapoport said he is interested in seeing other devices that have been cleared by the FDA as migraine treatments evaluated in migraine prevention. “I think we need more patients tried on the devices so we get an idea of which ones work acutely, which ones work preventively,” he said.
The authors reported personal and institutional relationships in the form of advisory board positions, consultancies, grants, research principal investigator roles, royalties, speakers bureau positions, and stockholders for a variety of pharmaceutical companies, agencies, and other organizations. Several authors disclosed ties with Theranica, the manufacturer of the REN device used in the study. Dr. Rapoport is editor-in-chief of Neurology Reviews and a consultant for Theranica, but was not involved in studies associated with the REN device.
Correction, 2/10/23: An earlier version of this article misstated the reduction in number of migraine days.
, according to recent research published in the journal Headache.
The prospective, randomized, double-blind, placebo-controlled, multicenter trial showed that remote electrical neuromodulation (REN) with Nerivio (Theranica Bio-Electronics Ltd.; Bridgewater, N.J.) found a mean reduction/decrease in the number of migraine days by an average of 4.0 days per month, according to Stewart J. Tepper MD, of the Geisel School of Medicine at Dartmouth in Hanover, N.H., and colleagues.*
“The statistically significant results were maintained in separate subanalyses of the chronic and episodic subsamples, as well as in the separate subanalyses of participants who used and did not use migraine prophylaxis,” Dr. Tepper and colleagues wrote.
A nonpharmacological alternative
Researchers randomized 248 participants into active and placebo groups, with 95 participants in the active group and 84 participants in the placebo group meeting the criteria for a modified intention-to-treat (mITT) analysis. Most of the participants in the ITT dataset were women (85.9%) with an average age of 41.7 years, and a baseline average of 12.2 migraine days and 15.6 headache days. Overall, 52.4% of participants in the ITT dataset had chronic migraine, 25.0% had migraine with aura, and 41.1% were taking preventative medication.
Dr. Tepper and colleagues followed participants for 4 weeks at baseline for observation followed by 8 weeks of participants using the REN device every other day for 45 minutes, or a placebo device that “produces electrical pulses of the same maximum intensity (34 mA) and overall energy, but with different pulse durations and much lower frequencies compared with the active device.” Participants completed a daily diary where they recorded their symptoms.
Researchers assessed the mean change in number of migraine days per month as a primary outcome, and evaluated participants who experienced episodic and chronic migraines separately in subgroup analyses. Secondary outcome measures included mean change in number of moderate or severe headache days, 50% reduction in mean number of headache days compared with baseline, Headache Impact Test short form (HIT-6) and Migraine Specific Quality of Life Questionnaire (MSQ) Role Function Domain total score mean change at 12 weeks compared with week 1, and reduction in mean number of days taking acute headache or migraine medication.
Participants receiving REN treatment had a significant reduction in mean migraine days per month compared with the placebo group (4.0 days vs. 1.3 days; 95% confidence interval, –3.9 days to –1.5 days; P < .001). In subgroup analyses, a significant reduction in migraine days was seen in participants receiving REN treatment with episodic migraine (3.2 days vs. 1.0 days; P = .003) and chronic migraine (4.7 days vs. 1.6 days; P = .001) compared with placebo.
Dr. Tepper and colleagues found a significant reduction in moderate and/or severe headache days among participants receiving REN treatment compared with placebo (3.8 days vs. 2.2 days; P = .005), a significant reduction in headache days overall compared with placebo (4.5 days vs. 1.8 days; P < .001), a significant percentage of patients who experienced 50% reduction in moderate and/or severe headache days compared with placebo (51.6% vs. 35.7%; P = .033), and a significant reduction in acute medication days compared with placebo (3.5 days vs. 1.4 days; P = .001). Dr. Tepper and colleagues found no serious device-related adverse events in either group.
The researchers noted that REN therapy is a “much-needed nonpharmacological alternative” to other preventive and acute treatments for migraine. “Given the previously well-established clinical efficacy and high safety profile in acute treatment of migraine, REN can cover the entire treatment spectrum of migraine, including both acute and preventive treatments,” they said.
‘A good place to start’
Commenting on the study, Alan M. Rapoport, MD, clinical professor of neurology at University of California, Los Angeles; past president of the International Headache Society; and editor-in-chief of Neurology Reviews, said the study was well designed, but acknowledged the 8-week follow-up time for participants as one potential area where he would have wanted to see more data.
As a medical device cleared for use by the Food and Drug Administration for acute treatment of migraine, the REM device also appears to be effective as a migraine preventative based on the results of the study with “virtually no adverse events,” he noted.
“I think this is a great treatment. I think it’s a good place to start,” Dr. Rapoport said. Given the low adverse event rate, he said he would be willing to offer the device to patients as a first option for preventing migraine and either switch to another preventative option or add an additional medication in combination based on how the patient responds. However, at the moment, he noted that this device is not covered by insurance.
Now that a REN device has been shown to work in the acute setting and as a preventative, Dr. Rapoport said he is interested in seeing other devices that have been cleared by the FDA as migraine treatments evaluated in migraine prevention. “I think we need more patients tried on the devices so we get an idea of which ones work acutely, which ones work preventively,” he said.
The authors reported personal and institutional relationships in the form of advisory board positions, consultancies, grants, research principal investigator roles, royalties, speakers bureau positions, and stockholders for a variety of pharmaceutical companies, agencies, and other organizations. Several authors disclosed ties with Theranica, the manufacturer of the REN device used in the study. Dr. Rapoport is editor-in-chief of Neurology Reviews and a consultant for Theranica, but was not involved in studies associated with the REN device.
Correction, 2/10/23: An earlier version of this article misstated the reduction in number of migraine days.
, according to recent research published in the journal Headache.
The prospective, randomized, double-blind, placebo-controlled, multicenter trial showed that remote electrical neuromodulation (REN) with Nerivio (Theranica Bio-Electronics Ltd.; Bridgewater, N.J.) found a mean reduction/decrease in the number of migraine days by an average of 4.0 days per month, according to Stewart J. Tepper MD, of the Geisel School of Medicine at Dartmouth in Hanover, N.H., and colleagues.*
“The statistically significant results were maintained in separate subanalyses of the chronic and episodic subsamples, as well as in the separate subanalyses of participants who used and did not use migraine prophylaxis,” Dr. Tepper and colleagues wrote.
A nonpharmacological alternative
Researchers randomized 248 participants into active and placebo groups, with 95 participants in the active group and 84 participants in the placebo group meeting the criteria for a modified intention-to-treat (mITT) analysis. Most of the participants in the ITT dataset were women (85.9%) with an average age of 41.7 years, and a baseline average of 12.2 migraine days and 15.6 headache days. Overall, 52.4% of participants in the ITT dataset had chronic migraine, 25.0% had migraine with aura, and 41.1% were taking preventative medication.
Dr. Tepper and colleagues followed participants for 4 weeks at baseline for observation followed by 8 weeks of participants using the REN device every other day for 45 minutes, or a placebo device that “produces electrical pulses of the same maximum intensity (34 mA) and overall energy, but with different pulse durations and much lower frequencies compared with the active device.” Participants completed a daily diary where they recorded their symptoms.
Researchers assessed the mean change in number of migraine days per month as a primary outcome, and evaluated participants who experienced episodic and chronic migraines separately in subgroup analyses. Secondary outcome measures included mean change in number of moderate or severe headache days, 50% reduction in mean number of headache days compared with baseline, Headache Impact Test short form (HIT-6) and Migraine Specific Quality of Life Questionnaire (MSQ) Role Function Domain total score mean change at 12 weeks compared with week 1, and reduction in mean number of days taking acute headache or migraine medication.
Participants receiving REN treatment had a significant reduction in mean migraine days per month compared with the placebo group (4.0 days vs. 1.3 days; 95% confidence interval, –3.9 days to –1.5 days; P < .001). In subgroup analyses, a significant reduction in migraine days was seen in participants receiving REN treatment with episodic migraine (3.2 days vs. 1.0 days; P = .003) and chronic migraine (4.7 days vs. 1.6 days; P = .001) compared with placebo.
Dr. Tepper and colleagues found a significant reduction in moderate and/or severe headache days among participants receiving REN treatment compared with placebo (3.8 days vs. 2.2 days; P = .005), a significant reduction in headache days overall compared with placebo (4.5 days vs. 1.8 days; P < .001), a significant percentage of patients who experienced 50% reduction in moderate and/or severe headache days compared with placebo (51.6% vs. 35.7%; P = .033), and a significant reduction in acute medication days compared with placebo (3.5 days vs. 1.4 days; P = .001). Dr. Tepper and colleagues found no serious device-related adverse events in either group.
The researchers noted that REN therapy is a “much-needed nonpharmacological alternative” to other preventive and acute treatments for migraine. “Given the previously well-established clinical efficacy and high safety profile in acute treatment of migraine, REN can cover the entire treatment spectrum of migraine, including both acute and preventive treatments,” they said.
‘A good place to start’
Commenting on the study, Alan M. Rapoport, MD, clinical professor of neurology at University of California, Los Angeles; past president of the International Headache Society; and editor-in-chief of Neurology Reviews, said the study was well designed, but acknowledged the 8-week follow-up time for participants as one potential area where he would have wanted to see more data.
As a medical device cleared for use by the Food and Drug Administration for acute treatment of migraine, the REM device also appears to be effective as a migraine preventative based on the results of the study with “virtually no adverse events,” he noted.
“I think this is a great treatment. I think it’s a good place to start,” Dr. Rapoport said. Given the low adverse event rate, he said he would be willing to offer the device to patients as a first option for preventing migraine and either switch to another preventative option or add an additional medication in combination based on how the patient responds. However, at the moment, he noted that this device is not covered by insurance.
Now that a REN device has been shown to work in the acute setting and as a preventative, Dr. Rapoport said he is interested in seeing other devices that have been cleared by the FDA as migraine treatments evaluated in migraine prevention. “I think we need more patients tried on the devices so we get an idea of which ones work acutely, which ones work preventively,” he said.
The authors reported personal and institutional relationships in the form of advisory board positions, consultancies, grants, research principal investigator roles, royalties, speakers bureau positions, and stockholders for a variety of pharmaceutical companies, agencies, and other organizations. Several authors disclosed ties with Theranica, the manufacturer of the REN device used in the study. Dr. Rapoport is editor-in-chief of Neurology Reviews and a consultant for Theranica, but was not involved in studies associated with the REN device.
Correction, 2/10/23: An earlier version of this article misstated the reduction in number of migraine days.
FROM HEADACHE
Large cohort study finds isotretinoin not associated with IBD
that also found no significant association of oral tetracycline-class antibiotics with IBD – and a small but statistically significant association of acne itself with the inflammatory disorders that make up IBD.
For the study, senior author John S. Barbieri, MD, MBA, of the department of dermatology, at Brigham and Women’s Hospital, Boston, and his colleagues used data from the TriNetX global research platform, which mines patient-level electronic medical record data from dozens of health care organizations, mainly in the United States. The network includes over 106 million patients. They looked at four cohorts: Patients without acne; those with acne but no current or prior use of systemic medications; those with acne managed with isotretinoin (and no prior use of oral tetracycline-class antibiotics); and those with acne managed with oral tetracycline-class antibiotics (and no exposure to isotretinoin).
For the acne cohorts, the investigators captured first encounters with a diagnosis of acne and first prescriptions of interest. And studywide, they used propensity score matching to balance cohorts for age, sex, race, ethnicity, and combined oral contraceptive use.
“These data should provide more reassurance to patients and prescribers that isotretinoin does not appear to result in a meaningfully increased risk of inflammatory bowel disease,” they wrote in the study, published online in the Journal of the American Academy of Dermatology.
“These are important findings as isotretinoin is a valuable treatment for acne that can result in a durable remission of disease activity, prevent acne scarring, and reduce our overreliance on oral antibiotics for acne,” they added.
Indeed, dermatologist Jonathan S. Weiss, MD, who was not involved in the research and was asked to comment on the study, said that the findings “are reassuring given the large numbers of patients evaluated and treated.” The smallest cohort – the isotretinoin group – had over 11,000 patients, and the other cohorts had over 100,000 patients each, he said in an interview.
“At this point, I’m not sure we need any other immediate information to feel comfortable using isotretinoin with respect to a potential to cause IBD, but it would be nice to see some longitudinal follow-up data for longer-term reassurance,” added Dr. Weiss, who practices in Snellville, Georgia, and is on the board of the directors of the American Acne and Rosacea Society.
The findings: Risk with acne
To assess the potential association between acne and IBD, the researchers identified more than 350,000 patients with acne managed without systemic medications, and propensity score matched them with patients who did not have acne. Altogether, their mean age was 22; 32.1% were male, and 59.6% were White.
Compared with the controls who did not have acne, they found a statistically significant association between acne and risk of incident IBD (odds ratio, 1.42; 95% confidence interval, 1.23-1.65) and an absolute risk difference of .04%. Separated into Crohn’s disease (CD) and ulcerative colitis (UC), ORs were 1.56 and 1.62, respectively.
Tetracyclines
To assess the association of oral tetracycline use and IBD, they compared more than 144,000 patients whose acne was managed with antibiotics with patients whose acne was managed without systemic medications. The patients had a mean age of 24.4; 34.7% were male, and 68.2% were White.
Compared with the patients who were not on systemic medications, there were no significant associations among those on oral tetracyclines, with an OR for incident IBD of 1 (95% CI, 0.82-1.22), an OR for incident CD of 1.09 (95% CI, 0.86-1.38), and an OR for UC of 0.78 (95% CI, 0.61-1.00).
Isotretinoin
To evaluate the association of isotretinoin and IBD, the researchers compared more than 11,000 patients treated with isotretinoin with two matched groups: patients with acne managed without systemic medications, and patients with acne managed with oral tetracyclines. The latter comparison was made to minimize potential confounding by acne severity. These patients had a mean age of 21.1; 49.5% were male, and 75.3% were White.
In the first comparison, compared with patients not treated with systemic medications, the OR for 1-year incidence of IBD among patients treated with isotretinoin was 1.29 (95% CI, 0.64-2.59), with an absolute risk difference of .036%. The ORs for CD and UC were 1.00 (95% CI, 0.45-2.23) and 1.27 (95% CI, .58-2.80), respectively.
And compared with the antibiotic-managed group, the OR for incident IBD among those on isotretinoin was 1.13 (95% CI, 0.57-2.21), with an absolute risk difference of .018%. The OR for CD was 1.00 (95% CI, 0.45-2.23). The OR for UC could not be accurately estimated because of an insufficient number of events in the tetracycline-treated group.
‘Challenging’ area of research
Researching acne treatments and the potential risk of IBD has been a methodologically “challenging topic to study” because of possible confounding and surveillance bias depending on study designs, Dr. Barbieri, director of the Brigham and Women’s Advanced Acne Therapeutics Clinic, said in an interview.
Studies that have identified a potential association between isotretinoin and IBD often have not adequately controlled for prior antibiotic exposure, for instance. And other studies, including a retrospective cohort study also published recently in JAAD using the same TriNetX database, have found 6-month isotretinoin-related risks of IBD but no increased risk at 1 year or more of follow-up – a finding that suggests a role of surveillance bias, Dr. Barbieri said.
The follow-up period of 1 year in their new study was chosen to minimize the risk of such bias. “Since patients on isotretinoin are seen more often, and since there are historical concerns about isotretinoin and IBD, patients on isotretinoin may be more likely to be screened earlier and thus could be diagnosed sooner than those not on [the medication],” he said.
He and his coauthors considered similar potential bias in designing the no-acne cohort, choosing patients who had routine primary care visits without abnormal findings in order to “reduce potential for bias due to frequency of interaction with the health care system,” they noted in their paper. (Patients had no prior encounters for acne and no history of acne treatments.)
Antibiotics, acne itself
Research on antibiotic use for acne and risk of IBD is scant, and the few studies that have been published show conflicting findings, Dr. Barbieri noted. In the meantime, studies and meta-analyses in the general medical literature – not involving acne – have identified an association between lifetime oral antibiotic exposure and IBD, he said.
While the results of the new study “are reassuring that oral tetracycline-class exposure for acne may not be associated with a significant absolute risk of inflammatory bowel disease, given the potential for antibiotic resistance and other antibiotic-associated complications, it remains important to be judicious” with their use in acne management, he and his coauthors wrote in the study.
The potential association between antibiotics for acne and IBD needs further study, preferably with longer follow-up duration, Dr. Barbieri said in the interview, but researchers are challenged by the lack of datasets with high-quality longitudinal data “beyond a few years of follow-up.”
The extent to which acne itself is associated with IBD is another area ripe for more research. Thus far, it seems that IBD and acne – and other chronic inflammatory skin diseases such as psoriasis – involve similar pathogenic pathways. “We know that in IBD Th17 and TNF immunologic pathways are important, so it’s not surprising that there may be associations,” he said.
In their paper, Dr. Barbieri and his coauthors emphasize, however, that the absolute risk difference between acne and IBD is small. It’s “unlikely that population level screening is warranted among patients with acne,” they wrote.
A second new study
The other study, also published recently in JAAD, used the same TriNetX research platform to identify approximately 77,000 patients with acne starting isotretinoin and matched them with patients starting oral antibiotics.
The investigators, Khalaf Kridin MD, PhD, and Ralf J. Ludwig, MD, of the Lübeck Institute of Experimental Dermatology, University of Lübeck (Germany), found that the lifetime risks (greater than 6 months) for patients on isotretinoin were not significantly elevated, compared with those on oral antibiotics for either CD (hazard ratio 1.05; 95% CI, 0.89-1.24, P = .583) or UC (HR, 1.13; 95% CI, 0.95-1.34; P = .162) They also looked at the risk of irritable bowel syndrome (IBS) and found a lower lifetime risk in the isotretinoin group.
In the short term, during the first 6 months after drug initiation, there was a significant, but slight increase in UC in the isotretinoin group. But this risk decreased to the level of the antibiotic group with longer follow up. “The absolute incidence rates [of IBD] and the risk difference of UC within the first 6 months are of limited clinical significance,” they wrote.
It may be, Dr. Weiss said in commenting on this study, “that isotretinoin unmasks an already-existing genetic tendency to UC early on in the course of treatment, but that it does not truly cause an increased incidence of any type of IBD.”
Both studies, said Dr. Barbieri, “add to an extensive body of literature that supports that isotretinoin is not associated with IBD.”
Dr. Barbieri had no disclosures for the study, for which Matthew T. Taylor served as first author. Coauthor Shawn Kwatra, MD, disclosed that he is an advisory board member/consultant for numerous pharmaceutical companies and has served as an investigator for several. Both are supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. The other authors had no disclosures. Dr. Kridin and Dr. Ludwig had no disclosures for their study. Dr. Weiss had no disclosures.
that also found no significant association of oral tetracycline-class antibiotics with IBD – and a small but statistically significant association of acne itself with the inflammatory disorders that make up IBD.
For the study, senior author John S. Barbieri, MD, MBA, of the department of dermatology, at Brigham and Women’s Hospital, Boston, and his colleagues used data from the TriNetX global research platform, which mines patient-level electronic medical record data from dozens of health care organizations, mainly in the United States. The network includes over 106 million patients. They looked at four cohorts: Patients without acne; those with acne but no current or prior use of systemic medications; those with acne managed with isotretinoin (and no prior use of oral tetracycline-class antibiotics); and those with acne managed with oral tetracycline-class antibiotics (and no exposure to isotretinoin).
For the acne cohorts, the investigators captured first encounters with a diagnosis of acne and first prescriptions of interest. And studywide, they used propensity score matching to balance cohorts for age, sex, race, ethnicity, and combined oral contraceptive use.
“These data should provide more reassurance to patients and prescribers that isotretinoin does not appear to result in a meaningfully increased risk of inflammatory bowel disease,” they wrote in the study, published online in the Journal of the American Academy of Dermatology.
“These are important findings as isotretinoin is a valuable treatment for acne that can result in a durable remission of disease activity, prevent acne scarring, and reduce our overreliance on oral antibiotics for acne,” they added.
Indeed, dermatologist Jonathan S. Weiss, MD, who was not involved in the research and was asked to comment on the study, said that the findings “are reassuring given the large numbers of patients evaluated and treated.” The smallest cohort – the isotretinoin group – had over 11,000 patients, and the other cohorts had over 100,000 patients each, he said in an interview.
“At this point, I’m not sure we need any other immediate information to feel comfortable using isotretinoin with respect to a potential to cause IBD, but it would be nice to see some longitudinal follow-up data for longer-term reassurance,” added Dr. Weiss, who practices in Snellville, Georgia, and is on the board of the directors of the American Acne and Rosacea Society.
The findings: Risk with acne
To assess the potential association between acne and IBD, the researchers identified more than 350,000 patients with acne managed without systemic medications, and propensity score matched them with patients who did not have acne. Altogether, their mean age was 22; 32.1% were male, and 59.6% were White.
Compared with the controls who did not have acne, they found a statistically significant association between acne and risk of incident IBD (odds ratio, 1.42; 95% confidence interval, 1.23-1.65) and an absolute risk difference of .04%. Separated into Crohn’s disease (CD) and ulcerative colitis (UC), ORs were 1.56 and 1.62, respectively.
Tetracyclines
To assess the association of oral tetracycline use and IBD, they compared more than 144,000 patients whose acne was managed with antibiotics with patients whose acne was managed without systemic medications. The patients had a mean age of 24.4; 34.7% were male, and 68.2% were White.
Compared with the patients who were not on systemic medications, there were no significant associations among those on oral tetracyclines, with an OR for incident IBD of 1 (95% CI, 0.82-1.22), an OR for incident CD of 1.09 (95% CI, 0.86-1.38), and an OR for UC of 0.78 (95% CI, 0.61-1.00).
Isotretinoin
To evaluate the association of isotretinoin and IBD, the researchers compared more than 11,000 patients treated with isotretinoin with two matched groups: patients with acne managed without systemic medications, and patients with acne managed with oral tetracyclines. The latter comparison was made to minimize potential confounding by acne severity. These patients had a mean age of 21.1; 49.5% were male, and 75.3% were White.
In the first comparison, compared with patients not treated with systemic medications, the OR for 1-year incidence of IBD among patients treated with isotretinoin was 1.29 (95% CI, 0.64-2.59), with an absolute risk difference of .036%. The ORs for CD and UC were 1.00 (95% CI, 0.45-2.23) and 1.27 (95% CI, .58-2.80), respectively.
And compared with the antibiotic-managed group, the OR for incident IBD among those on isotretinoin was 1.13 (95% CI, 0.57-2.21), with an absolute risk difference of .018%. The OR for CD was 1.00 (95% CI, 0.45-2.23). The OR for UC could not be accurately estimated because of an insufficient number of events in the tetracycline-treated group.
‘Challenging’ area of research
Researching acne treatments and the potential risk of IBD has been a methodologically “challenging topic to study” because of possible confounding and surveillance bias depending on study designs, Dr. Barbieri, director of the Brigham and Women’s Advanced Acne Therapeutics Clinic, said in an interview.
Studies that have identified a potential association between isotretinoin and IBD often have not adequately controlled for prior antibiotic exposure, for instance. And other studies, including a retrospective cohort study also published recently in JAAD using the same TriNetX database, have found 6-month isotretinoin-related risks of IBD but no increased risk at 1 year or more of follow-up – a finding that suggests a role of surveillance bias, Dr. Barbieri said.
The follow-up period of 1 year in their new study was chosen to minimize the risk of such bias. “Since patients on isotretinoin are seen more often, and since there are historical concerns about isotretinoin and IBD, patients on isotretinoin may be more likely to be screened earlier and thus could be diagnosed sooner than those not on [the medication],” he said.
He and his coauthors considered similar potential bias in designing the no-acne cohort, choosing patients who had routine primary care visits without abnormal findings in order to “reduce potential for bias due to frequency of interaction with the health care system,” they noted in their paper. (Patients had no prior encounters for acne and no history of acne treatments.)
Antibiotics, acne itself
Research on antibiotic use for acne and risk of IBD is scant, and the few studies that have been published show conflicting findings, Dr. Barbieri noted. In the meantime, studies and meta-analyses in the general medical literature – not involving acne – have identified an association between lifetime oral antibiotic exposure and IBD, he said.
While the results of the new study “are reassuring that oral tetracycline-class exposure for acne may not be associated with a significant absolute risk of inflammatory bowel disease, given the potential for antibiotic resistance and other antibiotic-associated complications, it remains important to be judicious” with their use in acne management, he and his coauthors wrote in the study.
The potential association between antibiotics for acne and IBD needs further study, preferably with longer follow-up duration, Dr. Barbieri said in the interview, but researchers are challenged by the lack of datasets with high-quality longitudinal data “beyond a few years of follow-up.”
The extent to which acne itself is associated with IBD is another area ripe for more research. Thus far, it seems that IBD and acne – and other chronic inflammatory skin diseases such as psoriasis – involve similar pathogenic pathways. “We know that in IBD Th17 and TNF immunologic pathways are important, so it’s not surprising that there may be associations,” he said.
In their paper, Dr. Barbieri and his coauthors emphasize, however, that the absolute risk difference between acne and IBD is small. It’s “unlikely that population level screening is warranted among patients with acne,” they wrote.
A second new study
The other study, also published recently in JAAD, used the same TriNetX research platform to identify approximately 77,000 patients with acne starting isotretinoin and matched them with patients starting oral antibiotics.
The investigators, Khalaf Kridin MD, PhD, and Ralf J. Ludwig, MD, of the Lübeck Institute of Experimental Dermatology, University of Lübeck (Germany), found that the lifetime risks (greater than 6 months) for patients on isotretinoin were not significantly elevated, compared with those on oral antibiotics for either CD (hazard ratio 1.05; 95% CI, 0.89-1.24, P = .583) or UC (HR, 1.13; 95% CI, 0.95-1.34; P = .162) They also looked at the risk of irritable bowel syndrome (IBS) and found a lower lifetime risk in the isotretinoin group.
In the short term, during the first 6 months after drug initiation, there was a significant, but slight increase in UC in the isotretinoin group. But this risk decreased to the level of the antibiotic group with longer follow up. “The absolute incidence rates [of IBD] and the risk difference of UC within the first 6 months are of limited clinical significance,” they wrote.
It may be, Dr. Weiss said in commenting on this study, “that isotretinoin unmasks an already-existing genetic tendency to UC early on in the course of treatment, but that it does not truly cause an increased incidence of any type of IBD.”
Both studies, said Dr. Barbieri, “add to an extensive body of literature that supports that isotretinoin is not associated with IBD.”
Dr. Barbieri had no disclosures for the study, for which Matthew T. Taylor served as first author. Coauthor Shawn Kwatra, MD, disclosed that he is an advisory board member/consultant for numerous pharmaceutical companies and has served as an investigator for several. Both are supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. The other authors had no disclosures. Dr. Kridin and Dr. Ludwig had no disclosures for their study. Dr. Weiss had no disclosures.
that also found no significant association of oral tetracycline-class antibiotics with IBD – and a small but statistically significant association of acne itself with the inflammatory disorders that make up IBD.
For the study, senior author John S. Barbieri, MD, MBA, of the department of dermatology, at Brigham and Women’s Hospital, Boston, and his colleagues used data from the TriNetX global research platform, which mines patient-level electronic medical record data from dozens of health care organizations, mainly in the United States. The network includes over 106 million patients. They looked at four cohorts: Patients without acne; those with acne but no current or prior use of systemic medications; those with acne managed with isotretinoin (and no prior use of oral tetracycline-class antibiotics); and those with acne managed with oral tetracycline-class antibiotics (and no exposure to isotretinoin).
For the acne cohorts, the investigators captured first encounters with a diagnosis of acne and first prescriptions of interest. And studywide, they used propensity score matching to balance cohorts for age, sex, race, ethnicity, and combined oral contraceptive use.
“These data should provide more reassurance to patients and prescribers that isotretinoin does not appear to result in a meaningfully increased risk of inflammatory bowel disease,” they wrote in the study, published online in the Journal of the American Academy of Dermatology.
“These are important findings as isotretinoin is a valuable treatment for acne that can result in a durable remission of disease activity, prevent acne scarring, and reduce our overreliance on oral antibiotics for acne,” they added.
Indeed, dermatologist Jonathan S. Weiss, MD, who was not involved in the research and was asked to comment on the study, said that the findings “are reassuring given the large numbers of patients evaluated and treated.” The smallest cohort – the isotretinoin group – had over 11,000 patients, and the other cohorts had over 100,000 patients each, he said in an interview.
“At this point, I’m not sure we need any other immediate information to feel comfortable using isotretinoin with respect to a potential to cause IBD, but it would be nice to see some longitudinal follow-up data for longer-term reassurance,” added Dr. Weiss, who practices in Snellville, Georgia, and is on the board of the directors of the American Acne and Rosacea Society.
The findings: Risk with acne
To assess the potential association between acne and IBD, the researchers identified more than 350,000 patients with acne managed without systemic medications, and propensity score matched them with patients who did not have acne. Altogether, their mean age was 22; 32.1% were male, and 59.6% were White.
Compared with the controls who did not have acne, they found a statistically significant association between acne and risk of incident IBD (odds ratio, 1.42; 95% confidence interval, 1.23-1.65) and an absolute risk difference of .04%. Separated into Crohn’s disease (CD) and ulcerative colitis (UC), ORs were 1.56 and 1.62, respectively.
Tetracyclines
To assess the association of oral tetracycline use and IBD, they compared more than 144,000 patients whose acne was managed with antibiotics with patients whose acne was managed without systemic medications. The patients had a mean age of 24.4; 34.7% were male, and 68.2% were White.
Compared with the patients who were not on systemic medications, there were no significant associations among those on oral tetracyclines, with an OR for incident IBD of 1 (95% CI, 0.82-1.22), an OR for incident CD of 1.09 (95% CI, 0.86-1.38), and an OR for UC of 0.78 (95% CI, 0.61-1.00).
Isotretinoin
To evaluate the association of isotretinoin and IBD, the researchers compared more than 11,000 patients treated with isotretinoin with two matched groups: patients with acne managed without systemic medications, and patients with acne managed with oral tetracyclines. The latter comparison was made to minimize potential confounding by acne severity. These patients had a mean age of 21.1; 49.5% were male, and 75.3% were White.
In the first comparison, compared with patients not treated with systemic medications, the OR for 1-year incidence of IBD among patients treated with isotretinoin was 1.29 (95% CI, 0.64-2.59), with an absolute risk difference of .036%. The ORs for CD and UC were 1.00 (95% CI, 0.45-2.23) and 1.27 (95% CI, .58-2.80), respectively.
And compared with the antibiotic-managed group, the OR for incident IBD among those on isotretinoin was 1.13 (95% CI, 0.57-2.21), with an absolute risk difference of .018%. The OR for CD was 1.00 (95% CI, 0.45-2.23). The OR for UC could not be accurately estimated because of an insufficient number of events in the tetracycline-treated group.
‘Challenging’ area of research
Researching acne treatments and the potential risk of IBD has been a methodologically “challenging topic to study” because of possible confounding and surveillance bias depending on study designs, Dr. Barbieri, director of the Brigham and Women’s Advanced Acne Therapeutics Clinic, said in an interview.
Studies that have identified a potential association between isotretinoin and IBD often have not adequately controlled for prior antibiotic exposure, for instance. And other studies, including a retrospective cohort study also published recently in JAAD using the same TriNetX database, have found 6-month isotretinoin-related risks of IBD but no increased risk at 1 year or more of follow-up – a finding that suggests a role of surveillance bias, Dr. Barbieri said.
The follow-up period of 1 year in their new study was chosen to minimize the risk of such bias. “Since patients on isotretinoin are seen more often, and since there are historical concerns about isotretinoin and IBD, patients on isotretinoin may be more likely to be screened earlier and thus could be diagnosed sooner than those not on [the medication],” he said.
He and his coauthors considered similar potential bias in designing the no-acne cohort, choosing patients who had routine primary care visits without abnormal findings in order to “reduce potential for bias due to frequency of interaction with the health care system,” they noted in their paper. (Patients had no prior encounters for acne and no history of acne treatments.)
Antibiotics, acne itself
Research on antibiotic use for acne and risk of IBD is scant, and the few studies that have been published show conflicting findings, Dr. Barbieri noted. In the meantime, studies and meta-analyses in the general medical literature – not involving acne – have identified an association between lifetime oral antibiotic exposure and IBD, he said.
While the results of the new study “are reassuring that oral tetracycline-class exposure for acne may not be associated with a significant absolute risk of inflammatory bowel disease, given the potential for antibiotic resistance and other antibiotic-associated complications, it remains important to be judicious” with their use in acne management, he and his coauthors wrote in the study.
The potential association between antibiotics for acne and IBD needs further study, preferably with longer follow-up duration, Dr. Barbieri said in the interview, but researchers are challenged by the lack of datasets with high-quality longitudinal data “beyond a few years of follow-up.”
The extent to which acne itself is associated with IBD is another area ripe for more research. Thus far, it seems that IBD and acne – and other chronic inflammatory skin diseases such as psoriasis – involve similar pathogenic pathways. “We know that in IBD Th17 and TNF immunologic pathways are important, so it’s not surprising that there may be associations,” he said.
In their paper, Dr. Barbieri and his coauthors emphasize, however, that the absolute risk difference between acne and IBD is small. It’s “unlikely that population level screening is warranted among patients with acne,” they wrote.
A second new study
The other study, also published recently in JAAD, used the same TriNetX research platform to identify approximately 77,000 patients with acne starting isotretinoin and matched them with patients starting oral antibiotics.
The investigators, Khalaf Kridin MD, PhD, and Ralf J. Ludwig, MD, of the Lübeck Institute of Experimental Dermatology, University of Lübeck (Germany), found that the lifetime risks (greater than 6 months) for patients on isotretinoin were not significantly elevated, compared with those on oral antibiotics for either CD (hazard ratio 1.05; 95% CI, 0.89-1.24, P = .583) or UC (HR, 1.13; 95% CI, 0.95-1.34; P = .162) They also looked at the risk of irritable bowel syndrome (IBS) and found a lower lifetime risk in the isotretinoin group.
In the short term, during the first 6 months after drug initiation, there was a significant, but slight increase in UC in the isotretinoin group. But this risk decreased to the level of the antibiotic group with longer follow up. “The absolute incidence rates [of IBD] and the risk difference of UC within the first 6 months are of limited clinical significance,” they wrote.
It may be, Dr. Weiss said in commenting on this study, “that isotretinoin unmasks an already-existing genetic tendency to UC early on in the course of treatment, but that it does not truly cause an increased incidence of any type of IBD.”
Both studies, said Dr. Barbieri, “add to an extensive body of literature that supports that isotretinoin is not associated with IBD.”
Dr. Barbieri had no disclosures for the study, for which Matthew T. Taylor served as first author. Coauthor Shawn Kwatra, MD, disclosed that he is an advisory board member/consultant for numerous pharmaceutical companies and has served as an investigator for several. Both are supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. The other authors had no disclosures. Dr. Kridin and Dr. Ludwig had no disclosures for their study. Dr. Weiss had no disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY