User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Hernia recurrence has improved only slightly
, according to a new research letter published March 1 in JAMA. Patients who underwent minimally invasive hernia repair had a higher incidence of reoperation than those who underwent open repairs.
In the United States, surgeons perform more than 1 million hernia repairs each year, according to the U.S. Food and Drug Administration. Despite hernias being such a common condition, it is “not at the forefront of many research agendas,” senior author Dana Telem, MD, an associate professor and section chief of general surgery at University of Michigan Health in Ann Arbor, said in an interview
While many surgical outcomes are measured within 30 days of operation, recurrences generally happen within 2 to 5 years after repair, she said. The last study that looked at reoperations for hernia repair at 10 years was published in 2003 and found that about 20% of patients needed surgery for reoccurrence over a decade. “We don’t really have a good understanding of what happened after these operations,” she explained. “Without knowing that piece, it is hard to go back retrospectively and understand what is the right operation for the right person at the right time.”
To understand rates of reoperation for hernia reoccurrence in today’s U.S. population of older adults, Dr. Telem and colleagues sorted through Medicare claims data to find adult patients who had undergone ventral or incisional and umbilical hernia repair from January 1, 2007 through December 31, 2018. They identified a total of 175,735 patients, 162,292 that underwent ventral or incisional hernia repair and 13,443 that underwent umbilical hernia repair. The average age of patients was 68.9 years and 39.2% were men. Most patients were White (87.2%), 8.1% were Black, 1.9% were Hispanic, and 0.5% were Asian. Median follow-up was 5.3 years.
Over the 10-year study period, 25,061 patients required reoperation for hernia recurrence with an adjusted cumulative incidence of 16.1% (95% CI, 16.1% - 16.2%). Patients who underwent open repair had a lower incidence of recurrence over 10 years than those who underwent minimally invasive repair for all hernia types (Table 1).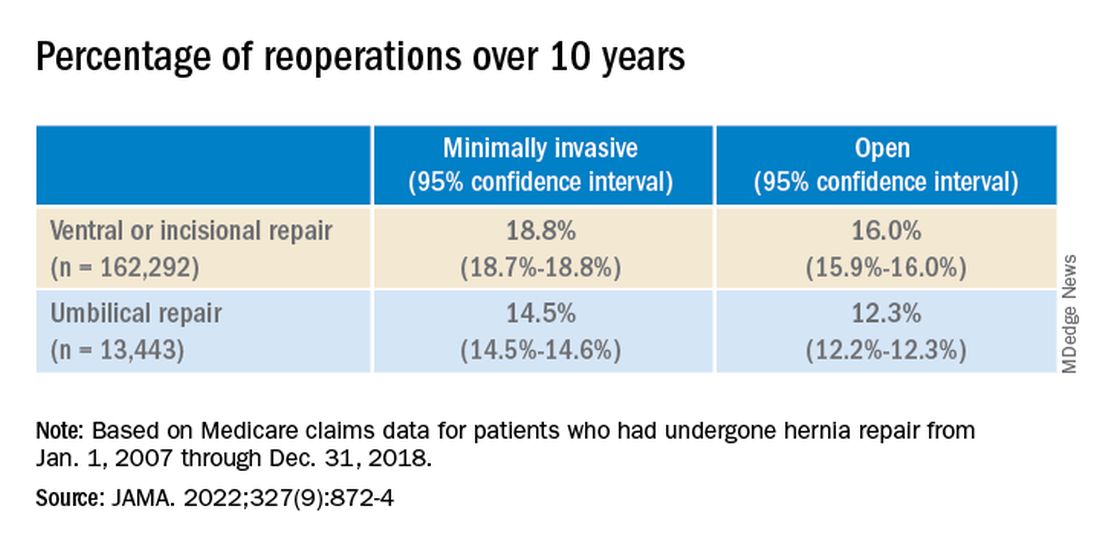
While it appears that hernia recurrence and reoperation have only marginally improved from 2003 to today, Vedra Augenstein, MD, an associate professor of surgery at the Atrium Health General & Complex Abdominal Surgery facility in Charlotte, N.C., suspects there is more to the story. “I think the reason it hasn’t gotten a whole lot better is just because we are operating on much tougher cases than we used to,” she said in an interview. “The way we are fixing hernias has changed and patients are being optimized differently.” Dr. Augenstein was not involved with the research.
To better understand how recurrence has changed over time, there needs to be more data about the comorbidities of patients, the techniques employed, and the meshes used in these surgeries, she said. Those numbers are not available in the published JAMA research letter, but Dr. Telem and colleagues will be submitting an article about this work with greater details.
Dr. Augenstein was also surprised that minimally invasive surgeries had higher incidences of reoperation for recurrence compared to open hernia surgeries. “I would think that patients who had minimally invasive repairs would actually have a lower chance of having postoperative complications because of wound issues,” she said. “Literature has shown that the recurrence rate is lower [in minimally invasive surgeries] because of fewer surgical site infections.”
While Dr. Telem also considers this research letter to be the first step in understanding modern hernia surgery outcomes, it is also a reminder that there is room for improvement in hernia repair surgeries. This includes advising patients on risk factors that may make them more likely to have a hernia recurrence, such as obesity, smoking, and diabetes, she added. “If we know it’s not a perfect science, then we have to do everything that we can upfront to help those numbers.”
Dr. Telem has reported receiving grants from the Agency for Healthcare Research and Quality and consulting fees from Medtronic. Dr. Augenstein has reported consulting for Intuitive Surgical, Medtronic, Allergan, Acelity, Vicarious Surgical, and Bard Pharmaceuticals and has received honoraria for speaking from Medtronic, Allergan, Intuitive Surgical, Acelity, and WL Gore.
A version of this article first appeared on Medscape.com.
, according to a new research letter published March 1 in JAMA. Patients who underwent minimally invasive hernia repair had a higher incidence of reoperation than those who underwent open repairs.
In the United States, surgeons perform more than 1 million hernia repairs each year, according to the U.S. Food and Drug Administration. Despite hernias being such a common condition, it is “not at the forefront of many research agendas,” senior author Dana Telem, MD, an associate professor and section chief of general surgery at University of Michigan Health in Ann Arbor, said in an interview
While many surgical outcomes are measured within 30 days of operation, recurrences generally happen within 2 to 5 years after repair, she said. The last study that looked at reoperations for hernia repair at 10 years was published in 2003 and found that about 20% of patients needed surgery for reoccurrence over a decade. “We don’t really have a good understanding of what happened after these operations,” she explained. “Without knowing that piece, it is hard to go back retrospectively and understand what is the right operation for the right person at the right time.”
To understand rates of reoperation for hernia reoccurrence in today’s U.S. population of older adults, Dr. Telem and colleagues sorted through Medicare claims data to find adult patients who had undergone ventral or incisional and umbilical hernia repair from January 1, 2007 through December 31, 2018. They identified a total of 175,735 patients, 162,292 that underwent ventral or incisional hernia repair and 13,443 that underwent umbilical hernia repair. The average age of patients was 68.9 years and 39.2% were men. Most patients were White (87.2%), 8.1% were Black, 1.9% were Hispanic, and 0.5% were Asian. Median follow-up was 5.3 years.
Over the 10-year study period, 25,061 patients required reoperation for hernia recurrence with an adjusted cumulative incidence of 16.1% (95% CI, 16.1% - 16.2%). Patients who underwent open repair had a lower incidence of recurrence over 10 years than those who underwent minimally invasive repair for all hernia types (Table 1).
While it appears that hernia recurrence and reoperation have only marginally improved from 2003 to today, Vedra Augenstein, MD, an associate professor of surgery at the Atrium Health General & Complex Abdominal Surgery facility in Charlotte, N.C., suspects there is more to the story. “I think the reason it hasn’t gotten a whole lot better is just because we are operating on much tougher cases than we used to,” she said in an interview. “The way we are fixing hernias has changed and patients are being optimized differently.” Dr. Augenstein was not involved with the research.
To better understand how recurrence has changed over time, there needs to be more data about the comorbidities of patients, the techniques employed, and the meshes used in these surgeries, she said. Those numbers are not available in the published JAMA research letter, but Dr. Telem and colleagues will be submitting an article about this work with greater details.
Dr. Augenstein was also surprised that minimally invasive surgeries had higher incidences of reoperation for recurrence compared to open hernia surgeries. “I would think that patients who had minimally invasive repairs would actually have a lower chance of having postoperative complications because of wound issues,” she said. “Literature has shown that the recurrence rate is lower [in minimally invasive surgeries] because of fewer surgical site infections.”
While Dr. Telem also considers this research letter to be the first step in understanding modern hernia surgery outcomes, it is also a reminder that there is room for improvement in hernia repair surgeries. This includes advising patients on risk factors that may make them more likely to have a hernia recurrence, such as obesity, smoking, and diabetes, she added. “If we know it’s not a perfect science, then we have to do everything that we can upfront to help those numbers.”
Dr. Telem has reported receiving grants from the Agency for Healthcare Research and Quality and consulting fees from Medtronic. Dr. Augenstein has reported consulting for Intuitive Surgical, Medtronic, Allergan, Acelity, Vicarious Surgical, and Bard Pharmaceuticals and has received honoraria for speaking from Medtronic, Allergan, Intuitive Surgical, Acelity, and WL Gore.
A version of this article first appeared on Medscape.com.
, according to a new research letter published March 1 in JAMA. Patients who underwent minimally invasive hernia repair had a higher incidence of reoperation than those who underwent open repairs.
In the United States, surgeons perform more than 1 million hernia repairs each year, according to the U.S. Food and Drug Administration. Despite hernias being such a common condition, it is “not at the forefront of many research agendas,” senior author Dana Telem, MD, an associate professor and section chief of general surgery at University of Michigan Health in Ann Arbor, said in an interview
While many surgical outcomes are measured within 30 days of operation, recurrences generally happen within 2 to 5 years after repair, she said. The last study that looked at reoperations for hernia repair at 10 years was published in 2003 and found that about 20% of patients needed surgery for reoccurrence over a decade. “We don’t really have a good understanding of what happened after these operations,” she explained. “Without knowing that piece, it is hard to go back retrospectively and understand what is the right operation for the right person at the right time.”
To understand rates of reoperation for hernia reoccurrence in today’s U.S. population of older adults, Dr. Telem and colleagues sorted through Medicare claims data to find adult patients who had undergone ventral or incisional and umbilical hernia repair from January 1, 2007 through December 31, 2018. They identified a total of 175,735 patients, 162,292 that underwent ventral or incisional hernia repair and 13,443 that underwent umbilical hernia repair. The average age of patients was 68.9 years and 39.2% were men. Most patients were White (87.2%), 8.1% were Black, 1.9% were Hispanic, and 0.5% were Asian. Median follow-up was 5.3 years.
Over the 10-year study period, 25,061 patients required reoperation for hernia recurrence with an adjusted cumulative incidence of 16.1% (95% CI, 16.1% - 16.2%). Patients who underwent open repair had a lower incidence of recurrence over 10 years than those who underwent minimally invasive repair for all hernia types (Table 1).
While it appears that hernia recurrence and reoperation have only marginally improved from 2003 to today, Vedra Augenstein, MD, an associate professor of surgery at the Atrium Health General & Complex Abdominal Surgery facility in Charlotte, N.C., suspects there is more to the story. “I think the reason it hasn’t gotten a whole lot better is just because we are operating on much tougher cases than we used to,” she said in an interview. “The way we are fixing hernias has changed and patients are being optimized differently.” Dr. Augenstein was not involved with the research.
To better understand how recurrence has changed over time, there needs to be more data about the comorbidities of patients, the techniques employed, and the meshes used in these surgeries, she said. Those numbers are not available in the published JAMA research letter, but Dr. Telem and colleagues will be submitting an article about this work with greater details.
Dr. Augenstein was also surprised that minimally invasive surgeries had higher incidences of reoperation for recurrence compared to open hernia surgeries. “I would think that patients who had minimally invasive repairs would actually have a lower chance of having postoperative complications because of wound issues,” she said. “Literature has shown that the recurrence rate is lower [in minimally invasive surgeries] because of fewer surgical site infections.”
While Dr. Telem also considers this research letter to be the first step in understanding modern hernia surgery outcomes, it is also a reminder that there is room for improvement in hernia repair surgeries. This includes advising patients on risk factors that may make them more likely to have a hernia recurrence, such as obesity, smoking, and diabetes, she added. “If we know it’s not a perfect science, then we have to do everything that we can upfront to help those numbers.”
Dr. Telem has reported receiving grants from the Agency for Healthcare Research and Quality and consulting fees from Medtronic. Dr. Augenstein has reported consulting for Intuitive Surgical, Medtronic, Allergan, Acelity, Vicarious Surgical, and Bard Pharmaceuticals and has received honoraria for speaking from Medtronic, Allergan, Intuitive Surgical, Acelity, and WL Gore.
A version of this article first appeared on Medscape.com.
FROM JAMA
Elective surgery should be delayed 7 weeks after COVID-19 infection for unvaccinated patients, statement recommends
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
.
For patients fully vaccinated against COVID-19 with breakthrough infections, there is no consensus on how vaccination affects the time between COVID-19 infection and elective surgery. Clinicians should use their clinical judgment to schedule procedures, said Randall M. Clark, MD, president of the American Society of Anesthesiologists (ASA). “We need all physicians, anesthesiologists, surgeons, and others to base their decision to go ahead with elective surgery on the patient’s symptoms, their need for the procedure, and whether delays could cause other problems with their health,” he said in an interview.
Prior to these updated recommendations, which were published Feb. 22, the ASA and the APSF recommended a 4-week gap between COVID-19 diagnosis and elective surgery for asymptomatic or mild cases, regardless of a patient’s vaccination status.
Extending the wait time from 4 to 7 weeks was based on a multination study conducted in October 2020 following more than 140,000 surgical patients. Patients with previous COVID-19 infection had an increased risk for complications and death in elective surgery for up to 6 weeks following their diagnosis, compared with patients without COVID-19. Additional research in the United States found that patients with a preoperative COVID diagnosis were at higher risk for postoperative complications of respiratory failure for up to 4 weeks after diagnosis and postoperative pneumonia complications for up to 8 weeks after diagnosis.
Because these studies were conducted in unvaccinated populations or those with low vaccination rates, and preliminary data suggest vaccinated patients with breakthrough infections may have a lower risk for complications and death postinfection, “we felt that it was prudent to just make recommendations specific to unvaccinated patients,” Dr. Clark added.
Although this guidance is “very helpful” in that it summarizes the currently available research to give evidence-based recommendations, the 7-week wait time is a “very conservative estimate,” Brent Matthews, MD, surgeon-in-chief of the surgery care division of Atrium Health, Charlotte, N.C., told this news organization. At Atrium Health, surgery is scheduled at least 21 days after a patient’s COVID-19 diagnosis, regardless of their vaccination status, Dr. Matthews said.
The studies currently available were conducted earlier in the pandemic, when a different variant was prevalent, Dr. Matthews explained. The Omicron variant is currently the most prevalent COVID-19 variant and is less virulent than earlier strains of the virus. The joint statement does note that there is currently “no robust data” on patients infected with the Delta or Omicron variants of COVID-19, and that “the Omicron variant causes less severe disease and is more likely to reside in the oro- and nasopharynx without infiltration and damage to the lungs.”
Still, the new recommendations are a reminder to re-evaluate the potential complications from surgery for previously infected patients and to consider what comorbidities might make them more vulnerable, Dr. Matthews said. “The real power of the joint statement is to get people to ensure that they make an assessment of every patient that comes in front of them who has had a recent positive COVID test.”
A version of this article first appeared on Medscape.com.
Nasal microbiota show promise as polyp predictor
A study of the nasal microbiome helped researchers predict recurrent polyps in chronic rhinosinusitis patients with more than 90% accuracy, based on data from 85 individuals.
Chronic rhinosinusitis with nasal polyps (CRSwNP) has a significant impact on patient quality of life, but the underlying mechanism of the disease has not been well studied, and treatment options remain limited, wrote Yan Zhao, MD, of Capital Medical University, Beijing, and study coauthors.
Previous research has shown that nasal microbiome composition differs in patients with and without asthma, and some studies suggest that changes in microbiota could contribute to CRSwNP, the authors wrote. The researchers wondered if features of the nasal microbiome can predict the recurrence of nasal polyps after endoscopic sinus surgery and serve as a potential treatment target.
In a study in Allergy, the researchers examined nasal swab samples from 85 adults with CRSwNP who underwent endoscopic sinus surgery between August 2014 and March 2016 at a single center in China. The researchers performed bacterial analysis and gene sequencing on all samples.
The patients ranged in age from 18-73 years, with a mean age of 46 years, and included 64 men and 21 women. The primary outcome was recurrence of polyps. Of the total, 39 individuals had recurrence, and 46 did not.
When the researchers compared microbiota from swab samples of recurrent and nonrecurrent patients, they found differences in composition based on bacterial genus abundance. “Campylobacter, Bdellovibrio, and Aggregatibacter, among others, were more abundant in swabs from CRSwNP recurrence samples, whereas Actinobacillus, Gemella, and Moraxella were more abundant in non-recurrence samples,” they wrote.
The researchers then tested their theory that distinct nasal microbiota could be a predictive marker of risk for future nasal polyp recurrence. They used a training set of 48 samples and constructed models from nasal microbiota alone, clinical features alone, and both together.
The regression model identified Porphyromonas, Bacteroides, Moryella, Aggregatibacter, Butyrivibrio, Shewanella, Pseudoxanthomonas, Friedmanniella, Limnobacter, and Curvibacter as the most important taxa that distinguished recurrence from nonrecurrence in the specimens. When the model was validated, the area under the curve was 0.914, yielding a predictor of nasal polyp recurrence with 91.4% accuracy.
“It is highly likely that proteins, nucleic acids, and other small molecules produced by nasal microbiota are associated with the progression of CRSwNP,” the researchers noted in their discussion of the findings. “Further, the nasal microbiota could maintain a stable community environment through the secretion of various chemical compounds and/or inflammatory factors, thus playing a central role in the development of CRSwNP.”
The study findings were limited by several factors, including the analysis of nasal flora only at the genus level in the screening phase, the use only of bioinformatic analysis for recurrence prediction, and the inclusion only of subjects from a single center, the researchers noted. Future studies should combine predictors to increase accuracy and include deeper sequencing, they said. However, the results support data from previous studies and suggest a strategy to meet the need for predictors of recurrence in CRSwNP, they concluded.
“There is a critical need to understand the role of the upper airway microbiome in different phenotypes of CRS,” said Emily K. Cope, PhD, assistant director at the Pathogen and Microbiome Institute, Northern Arizona University, Flagstaff, in an interview. “This was one of the first studies to evaluate the predictive power of the microbiome in recurrence of a common CRS phenotype – CRS with nasal polyps,” she said. “Importantly, the researchers were able to predict recurrence of polyps prior to the disease manifestation,” she noted.
“Given the nascent state of current upper airway microbiome research, I was surprised that they were able to predict polyp recurrence prior to disease manifestation,” Dr. Cope said. “This is exciting, and I can imagine a future where we use microbiome data to understand risk for disease.”
What is the take-home message for clinicians? Although the immediate clinical implications are limited, Dr. Cope expressed enthusiasm for additional research. “At this point, there’s not a lot we can do without validation studies, but this study is promising. I hope we can understand the mechanism that an altered microbiome might drive (or be a result of) polyposis,” she said.
The study was supported by the National Natural Science Foundation of China, the program for the Changjiang scholars and innovative research team, the Beijing Bai-Qian-Wan talent project, the Public Welfare Development and Reform Pilot Project, the National Science and Technology Major Project, and the CAMS Innovation Fund for Medical Sciences. The researchers and Dr. Cope disclosed no financial conflicts.
A version of this article first appeared on Medscape.com.
A study of the nasal microbiome helped researchers predict recurrent polyps in chronic rhinosinusitis patients with more than 90% accuracy, based on data from 85 individuals.
Chronic rhinosinusitis with nasal polyps (CRSwNP) has a significant impact on patient quality of life, but the underlying mechanism of the disease has not been well studied, and treatment options remain limited, wrote Yan Zhao, MD, of Capital Medical University, Beijing, and study coauthors.
Previous research has shown that nasal microbiome composition differs in patients with and without asthma, and some studies suggest that changes in microbiota could contribute to CRSwNP, the authors wrote. The researchers wondered if features of the nasal microbiome can predict the recurrence of nasal polyps after endoscopic sinus surgery and serve as a potential treatment target.
In a study in Allergy, the researchers examined nasal swab samples from 85 adults with CRSwNP who underwent endoscopic sinus surgery between August 2014 and March 2016 at a single center in China. The researchers performed bacterial analysis and gene sequencing on all samples.
The patients ranged in age from 18-73 years, with a mean age of 46 years, and included 64 men and 21 women. The primary outcome was recurrence of polyps. Of the total, 39 individuals had recurrence, and 46 did not.
When the researchers compared microbiota from swab samples of recurrent and nonrecurrent patients, they found differences in composition based on bacterial genus abundance. “Campylobacter, Bdellovibrio, and Aggregatibacter, among others, were more abundant in swabs from CRSwNP recurrence samples, whereas Actinobacillus, Gemella, and Moraxella were more abundant in non-recurrence samples,” they wrote.
The researchers then tested their theory that distinct nasal microbiota could be a predictive marker of risk for future nasal polyp recurrence. They used a training set of 48 samples and constructed models from nasal microbiota alone, clinical features alone, and both together.
The regression model identified Porphyromonas, Bacteroides, Moryella, Aggregatibacter, Butyrivibrio, Shewanella, Pseudoxanthomonas, Friedmanniella, Limnobacter, and Curvibacter as the most important taxa that distinguished recurrence from nonrecurrence in the specimens. When the model was validated, the area under the curve was 0.914, yielding a predictor of nasal polyp recurrence with 91.4% accuracy.
“It is highly likely that proteins, nucleic acids, and other small molecules produced by nasal microbiota are associated with the progression of CRSwNP,” the researchers noted in their discussion of the findings. “Further, the nasal microbiota could maintain a stable community environment through the secretion of various chemical compounds and/or inflammatory factors, thus playing a central role in the development of CRSwNP.”
The study findings were limited by several factors, including the analysis of nasal flora only at the genus level in the screening phase, the use only of bioinformatic analysis for recurrence prediction, and the inclusion only of subjects from a single center, the researchers noted. Future studies should combine predictors to increase accuracy and include deeper sequencing, they said. However, the results support data from previous studies and suggest a strategy to meet the need for predictors of recurrence in CRSwNP, they concluded.
“There is a critical need to understand the role of the upper airway microbiome in different phenotypes of CRS,” said Emily K. Cope, PhD, assistant director at the Pathogen and Microbiome Institute, Northern Arizona University, Flagstaff, in an interview. “This was one of the first studies to evaluate the predictive power of the microbiome in recurrence of a common CRS phenotype – CRS with nasal polyps,” she said. “Importantly, the researchers were able to predict recurrence of polyps prior to the disease manifestation,” she noted.
“Given the nascent state of current upper airway microbiome research, I was surprised that they were able to predict polyp recurrence prior to disease manifestation,” Dr. Cope said. “This is exciting, and I can imagine a future where we use microbiome data to understand risk for disease.”
What is the take-home message for clinicians? Although the immediate clinical implications are limited, Dr. Cope expressed enthusiasm for additional research. “At this point, there’s not a lot we can do without validation studies, but this study is promising. I hope we can understand the mechanism that an altered microbiome might drive (or be a result of) polyposis,” she said.
The study was supported by the National Natural Science Foundation of China, the program for the Changjiang scholars and innovative research team, the Beijing Bai-Qian-Wan talent project, the Public Welfare Development and Reform Pilot Project, the National Science and Technology Major Project, and the CAMS Innovation Fund for Medical Sciences. The researchers and Dr. Cope disclosed no financial conflicts.
A version of this article first appeared on Medscape.com.
A study of the nasal microbiome helped researchers predict recurrent polyps in chronic rhinosinusitis patients with more than 90% accuracy, based on data from 85 individuals.
Chronic rhinosinusitis with nasal polyps (CRSwNP) has a significant impact on patient quality of life, but the underlying mechanism of the disease has not been well studied, and treatment options remain limited, wrote Yan Zhao, MD, of Capital Medical University, Beijing, and study coauthors.
Previous research has shown that nasal microbiome composition differs in patients with and without asthma, and some studies suggest that changes in microbiota could contribute to CRSwNP, the authors wrote. The researchers wondered if features of the nasal microbiome can predict the recurrence of nasal polyps after endoscopic sinus surgery and serve as a potential treatment target.
In a study in Allergy, the researchers examined nasal swab samples from 85 adults with CRSwNP who underwent endoscopic sinus surgery between August 2014 and March 2016 at a single center in China. The researchers performed bacterial analysis and gene sequencing on all samples.
The patients ranged in age from 18-73 years, with a mean age of 46 years, and included 64 men and 21 women. The primary outcome was recurrence of polyps. Of the total, 39 individuals had recurrence, and 46 did not.
When the researchers compared microbiota from swab samples of recurrent and nonrecurrent patients, they found differences in composition based on bacterial genus abundance. “Campylobacter, Bdellovibrio, and Aggregatibacter, among others, were more abundant in swabs from CRSwNP recurrence samples, whereas Actinobacillus, Gemella, and Moraxella were more abundant in non-recurrence samples,” they wrote.
The researchers then tested their theory that distinct nasal microbiota could be a predictive marker of risk for future nasal polyp recurrence. They used a training set of 48 samples and constructed models from nasal microbiota alone, clinical features alone, and both together.
The regression model identified Porphyromonas, Bacteroides, Moryella, Aggregatibacter, Butyrivibrio, Shewanella, Pseudoxanthomonas, Friedmanniella, Limnobacter, and Curvibacter as the most important taxa that distinguished recurrence from nonrecurrence in the specimens. When the model was validated, the area under the curve was 0.914, yielding a predictor of nasal polyp recurrence with 91.4% accuracy.
“It is highly likely that proteins, nucleic acids, and other small molecules produced by nasal microbiota are associated with the progression of CRSwNP,” the researchers noted in their discussion of the findings. “Further, the nasal microbiota could maintain a stable community environment through the secretion of various chemical compounds and/or inflammatory factors, thus playing a central role in the development of CRSwNP.”
The study findings were limited by several factors, including the analysis of nasal flora only at the genus level in the screening phase, the use only of bioinformatic analysis for recurrence prediction, and the inclusion only of subjects from a single center, the researchers noted. Future studies should combine predictors to increase accuracy and include deeper sequencing, they said. However, the results support data from previous studies and suggest a strategy to meet the need for predictors of recurrence in CRSwNP, they concluded.
“There is a critical need to understand the role of the upper airway microbiome in different phenotypes of CRS,” said Emily K. Cope, PhD, assistant director at the Pathogen and Microbiome Institute, Northern Arizona University, Flagstaff, in an interview. “This was one of the first studies to evaluate the predictive power of the microbiome in recurrence of a common CRS phenotype – CRS with nasal polyps,” she said. “Importantly, the researchers were able to predict recurrence of polyps prior to the disease manifestation,” she noted.
“Given the nascent state of current upper airway microbiome research, I was surprised that they were able to predict polyp recurrence prior to disease manifestation,” Dr. Cope said. “This is exciting, and I can imagine a future where we use microbiome data to understand risk for disease.”
What is the take-home message for clinicians? Although the immediate clinical implications are limited, Dr. Cope expressed enthusiasm for additional research. “At this point, there’s not a lot we can do without validation studies, but this study is promising. I hope we can understand the mechanism that an altered microbiome might drive (or be a result of) polyposis,” she said.
The study was supported by the National Natural Science Foundation of China, the program for the Changjiang scholars and innovative research team, the Beijing Bai-Qian-Wan talent project, the Public Welfare Development and Reform Pilot Project, the National Science and Technology Major Project, and the CAMS Innovation Fund for Medical Sciences. The researchers and Dr. Cope disclosed no financial conflicts.
A version of this article first appeared on Medscape.com.
Autoantibodies may underpin clotting effects of COVID-19
Circulating antiphospholipid autoantibodies may contribute to endothelial cell activation and dysfunction in severe COVID-19, researchers report.
In 2020, the same researchers reported results from a preclinical study demonstrating that autoantibodies from patients with active COVID-19 caused clotting in mice.
The new study, published in Arthritis and Rheumatology, found higher-than-expected levels of antiphospholipid autoantibodies in the blood samples of 244 patients hospitalized with COVID-19.
“While endothelial dysfunction has been implicated in the widespread thromboinflammatory complications of COVID-19, the upstream mediators of endotheliopathy remain for the most part cryptic,” write Hui Shi, MD, PhD, and coauthors from the University of Michigan, Ann Arbor, and the National Heart, Lung, and Blood Institute.
When asked for comment on the study, Eline T. Luning Prak, MD, PhD, professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania in Philadelphia, said, “The autopsy cases for COVID-19 strongly point to thromboembolic complications in many individuals who succumbed to sequelae of the infection.
“Importantly, however, many factors can contribute to this pathology, including the inflammatory milieu, monocyte activation, neutrophil extracellular traps, immune complexes, complement, as well as effects on endothelial cells,” explained Dr. Luning Prak, who was not involved in the study.
“The findings in this paper nicely complement another study by Schmaier et al. that came out recently in JCI Insight that also suggests that endothelial cells can be activated by antibodies, she said.
‘Even stronger connection between autoantibody formation and clotting in COVID-19’
Dr. Shi and her team cultured human endothelial cells in serum or plasma from 244 patients hospitalized with COVID-19 and plasma from 100 patients with non-COVID sepsis. Using in-cell enzyme-linked immunosorbent assay, they measured levels of key cell adhesion molecules.
After analysis, the researchers found that serum from COVID-19 patients activated cultured endothelial cells to express surface adhesion molecules essential to inflammation and thrombosis, particularly E-selectin, ICAM-1, and VCAM-1.
“The presence of circulating antiphospholipid antibodies was a strong marker of the ability of COVID-19 serum to activate endothelium,” they explain.
Further analyses revealed that, for a subset of serum samples from patients with severe infection, this activation could be mitigated by depleting total immunoglobulin G.
In addition, supplementation of control serum with patient IgG was adequate to trigger endothelial activation.
On the basis of these results, the researchers hypothesize that antiphospholipid autoantibodies may characterize antibody profiles in severe COVID-19 that activate the endothelium and transition the usually quiescent blood-vessel wall interface toward inflammation and coagulation.
“[These findings] provide an even stronger connection between autoantibody formation and clotting in COVID-19,” Dr. Shi said in an accompanying press release.
Clinical implications
From a clinical perspective, Dr. Shi and her team question whether patients with severe COVID-19 should be tested for antiphospholipid antibodies to assess their risk of thrombosis and progression to respiratory failure.
Moreover, they question whether patients with high antiphospholipid antibody titers might benefit from therapies used in conventional cases of severe antiphospholipid syndrome, such as plasmapheresis, anticoagulation therapy, and complement inhibition, Dr. Shi added.
The researchers hope to answer these and other remaining questions in future studies. “Eventually, we may be able to repurpose treatments used in traditional cases of antiphospholipid syndrome for COVID-19.
“As we await definitive solutions to the pandemic, these findings add important context to the complex interplay between SARS-CoV-2 infection, the human immune system, and vascular immunobiology,” she concluded.
The study was supported by grants from the Rheumatology Research Foundation, the Michigan Medicine Frankel Cardiovascular Center, and the A. Alfred Taubman Medical Research Institute. One author is an inventor on an unrelated pending patent to the University of Michigan. The other authors and Dr. Luning Prak have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Circulating antiphospholipid autoantibodies may contribute to endothelial cell activation and dysfunction in severe COVID-19, researchers report.
In 2020, the same researchers reported results from a preclinical study demonstrating that autoantibodies from patients with active COVID-19 caused clotting in mice.
The new study, published in Arthritis and Rheumatology, found higher-than-expected levels of antiphospholipid autoantibodies in the blood samples of 244 patients hospitalized with COVID-19.
“While endothelial dysfunction has been implicated in the widespread thromboinflammatory complications of COVID-19, the upstream mediators of endotheliopathy remain for the most part cryptic,” write Hui Shi, MD, PhD, and coauthors from the University of Michigan, Ann Arbor, and the National Heart, Lung, and Blood Institute.
When asked for comment on the study, Eline T. Luning Prak, MD, PhD, professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania in Philadelphia, said, “The autopsy cases for COVID-19 strongly point to thromboembolic complications in many individuals who succumbed to sequelae of the infection.
“Importantly, however, many factors can contribute to this pathology, including the inflammatory milieu, monocyte activation, neutrophil extracellular traps, immune complexes, complement, as well as effects on endothelial cells,” explained Dr. Luning Prak, who was not involved in the study.
“The findings in this paper nicely complement another study by Schmaier et al. that came out recently in JCI Insight that also suggests that endothelial cells can be activated by antibodies, she said.
‘Even stronger connection between autoantibody formation and clotting in COVID-19’
Dr. Shi and her team cultured human endothelial cells in serum or plasma from 244 patients hospitalized with COVID-19 and plasma from 100 patients with non-COVID sepsis. Using in-cell enzyme-linked immunosorbent assay, they measured levels of key cell adhesion molecules.
After analysis, the researchers found that serum from COVID-19 patients activated cultured endothelial cells to express surface adhesion molecules essential to inflammation and thrombosis, particularly E-selectin, ICAM-1, and VCAM-1.
“The presence of circulating antiphospholipid antibodies was a strong marker of the ability of COVID-19 serum to activate endothelium,” they explain.
Further analyses revealed that, for a subset of serum samples from patients with severe infection, this activation could be mitigated by depleting total immunoglobulin G.
In addition, supplementation of control serum with patient IgG was adequate to trigger endothelial activation.
On the basis of these results, the researchers hypothesize that antiphospholipid autoantibodies may characterize antibody profiles in severe COVID-19 that activate the endothelium and transition the usually quiescent blood-vessel wall interface toward inflammation and coagulation.
“[These findings] provide an even stronger connection between autoantibody formation and clotting in COVID-19,” Dr. Shi said in an accompanying press release.
Clinical implications
From a clinical perspective, Dr. Shi and her team question whether patients with severe COVID-19 should be tested for antiphospholipid antibodies to assess their risk of thrombosis and progression to respiratory failure.
Moreover, they question whether patients with high antiphospholipid antibody titers might benefit from therapies used in conventional cases of severe antiphospholipid syndrome, such as plasmapheresis, anticoagulation therapy, and complement inhibition, Dr. Shi added.
The researchers hope to answer these and other remaining questions in future studies. “Eventually, we may be able to repurpose treatments used in traditional cases of antiphospholipid syndrome for COVID-19.
“As we await definitive solutions to the pandemic, these findings add important context to the complex interplay between SARS-CoV-2 infection, the human immune system, and vascular immunobiology,” she concluded.
The study was supported by grants from the Rheumatology Research Foundation, the Michigan Medicine Frankel Cardiovascular Center, and the A. Alfred Taubman Medical Research Institute. One author is an inventor on an unrelated pending patent to the University of Michigan. The other authors and Dr. Luning Prak have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Circulating antiphospholipid autoantibodies may contribute to endothelial cell activation and dysfunction in severe COVID-19, researchers report.
In 2020, the same researchers reported results from a preclinical study demonstrating that autoantibodies from patients with active COVID-19 caused clotting in mice.
The new study, published in Arthritis and Rheumatology, found higher-than-expected levels of antiphospholipid autoantibodies in the blood samples of 244 patients hospitalized with COVID-19.
“While endothelial dysfunction has been implicated in the widespread thromboinflammatory complications of COVID-19, the upstream mediators of endotheliopathy remain for the most part cryptic,” write Hui Shi, MD, PhD, and coauthors from the University of Michigan, Ann Arbor, and the National Heart, Lung, and Blood Institute.
When asked for comment on the study, Eline T. Luning Prak, MD, PhD, professor of pathology and laboratory medicine at the Hospital of the University of Pennsylvania in Philadelphia, said, “The autopsy cases for COVID-19 strongly point to thromboembolic complications in many individuals who succumbed to sequelae of the infection.
“Importantly, however, many factors can contribute to this pathology, including the inflammatory milieu, monocyte activation, neutrophil extracellular traps, immune complexes, complement, as well as effects on endothelial cells,” explained Dr. Luning Prak, who was not involved in the study.
“The findings in this paper nicely complement another study by Schmaier et al. that came out recently in JCI Insight that also suggests that endothelial cells can be activated by antibodies, she said.
‘Even stronger connection between autoantibody formation and clotting in COVID-19’
Dr. Shi and her team cultured human endothelial cells in serum or plasma from 244 patients hospitalized with COVID-19 and plasma from 100 patients with non-COVID sepsis. Using in-cell enzyme-linked immunosorbent assay, they measured levels of key cell adhesion molecules.
After analysis, the researchers found that serum from COVID-19 patients activated cultured endothelial cells to express surface adhesion molecules essential to inflammation and thrombosis, particularly E-selectin, ICAM-1, and VCAM-1.
“The presence of circulating antiphospholipid antibodies was a strong marker of the ability of COVID-19 serum to activate endothelium,” they explain.
Further analyses revealed that, for a subset of serum samples from patients with severe infection, this activation could be mitigated by depleting total immunoglobulin G.
In addition, supplementation of control serum with patient IgG was adequate to trigger endothelial activation.
On the basis of these results, the researchers hypothesize that antiphospholipid autoantibodies may characterize antibody profiles in severe COVID-19 that activate the endothelium and transition the usually quiescent blood-vessel wall interface toward inflammation and coagulation.
“[These findings] provide an even stronger connection between autoantibody formation and clotting in COVID-19,” Dr. Shi said in an accompanying press release.
Clinical implications
From a clinical perspective, Dr. Shi and her team question whether patients with severe COVID-19 should be tested for antiphospholipid antibodies to assess their risk of thrombosis and progression to respiratory failure.
Moreover, they question whether patients with high antiphospholipid antibody titers might benefit from therapies used in conventional cases of severe antiphospholipid syndrome, such as plasmapheresis, anticoagulation therapy, and complement inhibition, Dr. Shi added.
The researchers hope to answer these and other remaining questions in future studies. “Eventually, we may be able to repurpose treatments used in traditional cases of antiphospholipid syndrome for COVID-19.
“As we await definitive solutions to the pandemic, these findings add important context to the complex interplay between SARS-CoV-2 infection, the human immune system, and vascular immunobiology,” she concluded.
The study was supported by grants from the Rheumatology Research Foundation, the Michigan Medicine Frankel Cardiovascular Center, and the A. Alfred Taubman Medical Research Institute. One author is an inventor on an unrelated pending patent to the University of Michigan. The other authors and Dr. Luning Prak have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 vaccines do not trigger sudden hearing loss: Study
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
Anecdotal reports have linked the vaccines against COVID-19 to the sudden loss of hearing in some people. But a new study has found no evidence for such a connection with any of the three approved shots.
The analysis of data from the Centers for Disease Control and Prevention’s Vaccine Adverse Event Reporting System (VAERS) found that
“We’re not finding a signal,” said Eric J. Formeister, MD, a neurotology fellow at the Johns Hopkins University, Baltimore, and the first author of the U.S. study, which appeared Feb. 24 in JAMA Otolaryngology – Head and Neck Surgery.
Dr. Formeister and colleagues undertook the study in response to reports of hearing problems, including hearing loss and tinnitus, that occurred soon after COVID-19 vaccination.
They analyzed reports of sudden hearing loss, experienced within 21 days of vaccination, logged in VAERS. Anyone can report a potential event to the database, which does not require medical documentation in support of the adverse event. To minimize potential misdiagnoses, Dr. Formeister and colleagues reviewed only those reports that indicated that a doctor had diagnosed sudden hearing loss, leaving 555 cases (305 in women; mean age 54 years) between December 2020 and July 2021.
Dividing these reports by the total doses of vaccines administered in the United States during that period yielded an incidence rate of 0.6 cases of sudden hearing loss for every 100,000 people, Dr. Formeister and colleagues reported.
When the researchers divided all cases of hearing loss in the VAERS database (2,170) by the number of people who had received two doses of vaccine, the incidence rate increased to 28 per 100,000 people. For comparison, the authors reported, the incidence of sudden hearing loss within the United States population is between 11 and 77 per 100,000 people, depending on age.
“There was not an increase in cases of sudden [sensorineural] hearing loss associated with COVID-19 vaccination compared to previously published reports before the COVID-19 vaccination era,” study coauthor Elliott D. Kozin, MD, assistant professor of otolaryngology–head and neck surgery at Harvard Medical School, Boston, said in an interview.
Another reassuring sign: If hearing loss were linked to the vaccines, the researchers said, they would expect to see an increase in the number of complaints in lockstep with an increase in the number of doses administered. However, the opposite was true. “[T]he rate of reports per 100,000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week,” the researchers reported.
They also looked at case reports of 21 men and women who had experienced sudden hearing loss after having received COVID-19 vaccines, to see if they could discern any clinically relevant signs of people most likely to experience the adverse event. However, the group had a range of preexisting conditions and varying times after receiving a vaccine when their hearing loss occurred, leading Dr. Formeister’s team to conclude that they could find no clear markers of risk.
“When we examined patients across several institutions, there was no obvious pattern. The patient demographics and clinical findings were variable,” Dr. Kozin said. A provisional interpretation of this data, he added, is that no link exists between COVID-19 vaccination and predictable hearing deficits, although the analysis covered a small number of patients.
“Association does not necessarily imply a causal relationship,” said Michael Brenner, MD, FACS, associate professor of otolaryngology–head and neck surgery at the University of Michigan, Ann Arbor. Dr. Brenner, who was not involved in the study, said any hearing loss attributed to the COVID-19 vaccines could have had other causes besides the injections.
But a second study, also published in JAMA Otolaryngology – Head and Neck Surgery on Feb. 24, leaves open the possibility of a link. Researchers in Israel looked for increases in steroid prescriptions used to treat sudden hearing loss as vaccination with the Pfizer version of the shot became widespread in that country. Their conclusion: The vaccine might be associated with a slightly increased risk of sudden hearing loss, although if so, that risk is likely “very small” and the benefits of vaccination “outweigh its potential association” with the side effect.
Dr. Brenner agreed. “The evidence supports [the] clear public health benefit of COVID-19 vaccination, and the scale of those benefits dwarfs associations with hearing, which are of uncertain significance,” he said.
A version of this article first appeared on Medscape.com.
FROM JAMA OTOLARYNGOLOGY – HEAD AND NECK SURGERY
When physicians are the plaintiffs
Have you experienced malpractice?
No, I’m not asking whether you have experienced litigation. I’m asking whether you, as a physician, have actually experienced substandard care from a colleague. I have heard many such experiences over the years, and mistreatment doesn’t seem to be getting any less frequent.
The first is that, unlike the Pope, who has a dedicated confessor trained to minister to his spiritual needs, no one formally trains physicians to treat physicians. As a result, most of us feel slightly uneasy at treating other physicians. We naturally wish to keep our colleagues well, but at the same time realize that our clinical skills are being very closely scrutinized. What if they are found to be wanting? This discomfiture can make a physician treating a physician overly compulsive, or worse, overtly dismissive.
Second, we physicians are famously poor patients. We pretend we don’t need the advice we give others, to monitor our health and promptly seek care when something feels amiss. And, for the period during which we delay a medical encounter, we often attempt to diagnose and treat ourselves.
Sometimes we are successful, which reinforces this approach. Other times, we fail at being our own caregiver and present to someone else either too late, or with avoidable complications. In the former instance, we congratulate ourselves and learn nothing from the experience. In the latter, we may heap shame upon ourselves for our folly, and we may learn; but it could be a lethal lesson. In the worst scenario, our colleague gives in to frustration (or angst), and heaps even more shame onto their late-presenting physician patient.
Third, when we do submit to being a patient, we often demand VIP treatment. This is probably in response to our anxiety that some of the worst things we have seen happen to patients might happen to us if we are not vigilant to ensure we receive a higher level of care. But of course, such hypervigilance can lead to excessive care and testing, with all the attendant hazards, or alternatively to dilution of care if our caregivers decide we are just too much trouble.
Fourth, as a fifth-generation physician myself, I am convinced that physicians and physician family members are either prone to unusual manifestations of common diseases or unusual diseases, or that rare disease entities and complications are actually more common than literature suggests, and they simply aren’t pursued or diagnosed in nonphysician families.
No matter how we may have arrived in a position to need medical care, how often is such care substandard? And how do we respond when we suspect, or know, this to be the case? Are physicians more, or less, likely to take legal action in the face of it?
I certainly don’t know any statistics. Physicians are in an excellent position to take such action, because judges and juries will likely believe that a doctor can recognize negligence when we fall victim to it. But we may also be reluctant to publicly admit the way (or ways) in which we may have contributed to substandard care or outcome.
Based on decades of working with physician clients who have been sued, and having been sued myself (thus witnessing and also experiencing the effects of litigation), I am probably more reluctant than normal patients or physicians to consider taking legal action. This, despite the fact that I am also a lawyer and (through organized medicine) know many colleagues in all specialties who might serve as expert witnesses.
I have experienced serial substandard care, which has left me highly conflicted about the efficacy of my chosen profession. As a resident, I had my first odd pain condition and consulted an “elder statesperson” from my institution, whom I assumed to be a “doctor’s doctor” because he was a superb teacher (wrong!)
He completely missed the diagnosis and further belittled (indeed, libeled) me in the medical record. (Some years later, I learned that, during that period, he was increasingly demented and tended to view all female patients as having “wandering uterus” equivalents.) Fortunately, I found a better diagnostician, or at least one more willing to lend credence to my complaints, who successfully removed the first of several “zebra” lesions I have experienced.
As a young faculty member, I had an odd presentation of a recurring gynecologic condition, which was treated surgically, successfully, except that my fertility was cut in half – a possibility about which I had not been informed when giving operative consent. Would I have sued this fellow faculty member for that? Never, because she invariably treated me with respect as a colleague.
Later in my career after leaving academia, the same condition recurred in a new location. My old-school gynecologist desired to do an extensive procedure, to which I demurred unless specific pathology was found intraoperatively. Affronted, he subjected me to laparoscopy, did nothing but look, and then left the hospital leaving me and the PACU nurse to try to decipher his instructions (which said, basically, “I didn’t find anything; don’t bother me again.”). Several years of pain later, a younger gynecologist performed the correct procedure to address my problem, which has never recurred. Would I have sued him? No, because I believe he had a disability.
At age 59, I developed a new mole. My beloved general practitioner, in the waning years of his practice, forgot to consult a colleague to remove it for several months. When I forced the issue, the mole was removed and turned out to be a rare pediatric condition considered a precursor to melanoma. The same general practitioner had told me I needn’t worry about my “mild hypercalcemia.”
Ten years later I diagnosed my own parathyroid adenoma, in the interim losing 10% of my bone density. Would I have sued him? No, for he always showed he cared. (Though maybe, if I had fractured my spine or hip.)
If you have been the victim of physician malpractice, how did you respond?
Do we serve our profession well by how we handle substandard care – upon ourselves (or our loved ones)?
Dr. Andrew is a former assistant professor in the department of emergency medicine, Johns Hopkins University, Baltimore, and founder and principal of MDMentor, Victoria, B.C.
A version of this article first appeared on Medscape.com.
Have you experienced malpractice?
No, I’m not asking whether you have experienced litigation. I’m asking whether you, as a physician, have actually experienced substandard care from a colleague. I have heard many such experiences over the years, and mistreatment doesn’t seem to be getting any less frequent.
The first is that, unlike the Pope, who has a dedicated confessor trained to minister to his spiritual needs, no one formally trains physicians to treat physicians. As a result, most of us feel slightly uneasy at treating other physicians. We naturally wish to keep our colleagues well, but at the same time realize that our clinical skills are being very closely scrutinized. What if they are found to be wanting? This discomfiture can make a physician treating a physician overly compulsive, or worse, overtly dismissive.
Second, we physicians are famously poor patients. We pretend we don’t need the advice we give others, to monitor our health and promptly seek care when something feels amiss. And, for the period during which we delay a medical encounter, we often attempt to diagnose and treat ourselves.
Sometimes we are successful, which reinforces this approach. Other times, we fail at being our own caregiver and present to someone else either too late, or with avoidable complications. In the former instance, we congratulate ourselves and learn nothing from the experience. In the latter, we may heap shame upon ourselves for our folly, and we may learn; but it could be a lethal lesson. In the worst scenario, our colleague gives in to frustration (or angst), and heaps even more shame onto their late-presenting physician patient.
Third, when we do submit to being a patient, we often demand VIP treatment. This is probably in response to our anxiety that some of the worst things we have seen happen to patients might happen to us if we are not vigilant to ensure we receive a higher level of care. But of course, such hypervigilance can lead to excessive care and testing, with all the attendant hazards, or alternatively to dilution of care if our caregivers decide we are just too much trouble.
Fourth, as a fifth-generation physician myself, I am convinced that physicians and physician family members are either prone to unusual manifestations of common diseases or unusual diseases, or that rare disease entities and complications are actually more common than literature suggests, and they simply aren’t pursued or diagnosed in nonphysician families.
No matter how we may have arrived in a position to need medical care, how often is such care substandard? And how do we respond when we suspect, or know, this to be the case? Are physicians more, or less, likely to take legal action in the face of it?
I certainly don’t know any statistics. Physicians are in an excellent position to take such action, because judges and juries will likely believe that a doctor can recognize negligence when we fall victim to it. But we may also be reluctant to publicly admit the way (or ways) in which we may have contributed to substandard care or outcome.
Based on decades of working with physician clients who have been sued, and having been sued myself (thus witnessing and also experiencing the effects of litigation), I am probably more reluctant than normal patients or physicians to consider taking legal action. This, despite the fact that I am also a lawyer and (through organized medicine) know many colleagues in all specialties who might serve as expert witnesses.
I have experienced serial substandard care, which has left me highly conflicted about the efficacy of my chosen profession. As a resident, I had my first odd pain condition and consulted an “elder statesperson” from my institution, whom I assumed to be a “doctor’s doctor” because he was a superb teacher (wrong!)
He completely missed the diagnosis and further belittled (indeed, libeled) me in the medical record. (Some years later, I learned that, during that period, he was increasingly demented and tended to view all female patients as having “wandering uterus” equivalents.) Fortunately, I found a better diagnostician, or at least one more willing to lend credence to my complaints, who successfully removed the first of several “zebra” lesions I have experienced.
As a young faculty member, I had an odd presentation of a recurring gynecologic condition, which was treated surgically, successfully, except that my fertility was cut in half – a possibility about which I had not been informed when giving operative consent. Would I have sued this fellow faculty member for that? Never, because she invariably treated me with respect as a colleague.
Later in my career after leaving academia, the same condition recurred in a new location. My old-school gynecologist desired to do an extensive procedure, to which I demurred unless specific pathology was found intraoperatively. Affronted, he subjected me to laparoscopy, did nothing but look, and then left the hospital leaving me and the PACU nurse to try to decipher his instructions (which said, basically, “I didn’t find anything; don’t bother me again.”). Several years of pain later, a younger gynecologist performed the correct procedure to address my problem, which has never recurred. Would I have sued him? No, because I believe he had a disability.
At age 59, I developed a new mole. My beloved general practitioner, in the waning years of his practice, forgot to consult a colleague to remove it for several months. When I forced the issue, the mole was removed and turned out to be a rare pediatric condition considered a precursor to melanoma. The same general practitioner had told me I needn’t worry about my “mild hypercalcemia.”
Ten years later I diagnosed my own parathyroid adenoma, in the interim losing 10% of my bone density. Would I have sued him? No, for he always showed he cared. (Though maybe, if I had fractured my spine or hip.)
If you have been the victim of physician malpractice, how did you respond?
Do we serve our profession well by how we handle substandard care – upon ourselves (or our loved ones)?
Dr. Andrew is a former assistant professor in the department of emergency medicine, Johns Hopkins University, Baltimore, and founder and principal of MDMentor, Victoria, B.C.
A version of this article first appeared on Medscape.com.
Have you experienced malpractice?
No, I’m not asking whether you have experienced litigation. I’m asking whether you, as a physician, have actually experienced substandard care from a colleague. I have heard many such experiences over the years, and mistreatment doesn’t seem to be getting any less frequent.
The first is that, unlike the Pope, who has a dedicated confessor trained to minister to his spiritual needs, no one formally trains physicians to treat physicians. As a result, most of us feel slightly uneasy at treating other physicians. We naturally wish to keep our colleagues well, but at the same time realize that our clinical skills are being very closely scrutinized. What if they are found to be wanting? This discomfiture can make a physician treating a physician overly compulsive, or worse, overtly dismissive.
Second, we physicians are famously poor patients. We pretend we don’t need the advice we give others, to monitor our health and promptly seek care when something feels amiss. And, for the period during which we delay a medical encounter, we often attempt to diagnose and treat ourselves.
Sometimes we are successful, which reinforces this approach. Other times, we fail at being our own caregiver and present to someone else either too late, or with avoidable complications. In the former instance, we congratulate ourselves and learn nothing from the experience. In the latter, we may heap shame upon ourselves for our folly, and we may learn; but it could be a lethal lesson. In the worst scenario, our colleague gives in to frustration (or angst), and heaps even more shame onto their late-presenting physician patient.
Third, when we do submit to being a patient, we often demand VIP treatment. This is probably in response to our anxiety that some of the worst things we have seen happen to patients might happen to us if we are not vigilant to ensure we receive a higher level of care. But of course, such hypervigilance can lead to excessive care and testing, with all the attendant hazards, or alternatively to dilution of care if our caregivers decide we are just too much trouble.
Fourth, as a fifth-generation physician myself, I am convinced that physicians and physician family members are either prone to unusual manifestations of common diseases or unusual diseases, or that rare disease entities and complications are actually more common than literature suggests, and they simply aren’t pursued or diagnosed in nonphysician families.
No matter how we may have arrived in a position to need medical care, how often is such care substandard? And how do we respond when we suspect, or know, this to be the case? Are physicians more, or less, likely to take legal action in the face of it?
I certainly don’t know any statistics. Physicians are in an excellent position to take such action, because judges and juries will likely believe that a doctor can recognize negligence when we fall victim to it. But we may also be reluctant to publicly admit the way (or ways) in which we may have contributed to substandard care or outcome.
Based on decades of working with physician clients who have been sued, and having been sued myself (thus witnessing and also experiencing the effects of litigation), I am probably more reluctant than normal patients or physicians to consider taking legal action. This, despite the fact that I am also a lawyer and (through organized medicine) know many colleagues in all specialties who might serve as expert witnesses.
I have experienced serial substandard care, which has left me highly conflicted about the efficacy of my chosen profession. As a resident, I had my first odd pain condition and consulted an “elder statesperson” from my institution, whom I assumed to be a “doctor’s doctor” because he was a superb teacher (wrong!)
He completely missed the diagnosis and further belittled (indeed, libeled) me in the medical record. (Some years later, I learned that, during that period, he was increasingly demented and tended to view all female patients as having “wandering uterus” equivalents.) Fortunately, I found a better diagnostician, or at least one more willing to lend credence to my complaints, who successfully removed the first of several “zebra” lesions I have experienced.
As a young faculty member, I had an odd presentation of a recurring gynecologic condition, which was treated surgically, successfully, except that my fertility was cut in half – a possibility about which I had not been informed when giving operative consent. Would I have sued this fellow faculty member for that? Never, because she invariably treated me with respect as a colleague.
Later in my career after leaving academia, the same condition recurred in a new location. My old-school gynecologist desired to do an extensive procedure, to which I demurred unless specific pathology was found intraoperatively. Affronted, he subjected me to laparoscopy, did nothing but look, and then left the hospital leaving me and the PACU nurse to try to decipher his instructions (which said, basically, “I didn’t find anything; don’t bother me again.”). Several years of pain later, a younger gynecologist performed the correct procedure to address my problem, which has never recurred. Would I have sued him? No, because I believe he had a disability.
At age 59, I developed a new mole. My beloved general practitioner, in the waning years of his practice, forgot to consult a colleague to remove it for several months. When I forced the issue, the mole was removed and turned out to be a rare pediatric condition considered a precursor to melanoma. The same general practitioner had told me I needn’t worry about my “mild hypercalcemia.”
Ten years later I diagnosed my own parathyroid adenoma, in the interim losing 10% of my bone density. Would I have sued him? No, for he always showed he cared. (Though maybe, if I had fractured my spine or hip.)
If you have been the victim of physician malpractice, how did you respond?
Do we serve our profession well by how we handle substandard care – upon ourselves (or our loved ones)?
Dr. Andrew is a former assistant professor in the department of emergency medicine, Johns Hopkins University, Baltimore, and founder and principal of MDMentor, Victoria, B.C.
A version of this article first appeared on Medscape.com.
Twenty-three percent of health care workers likely to leave industry soon: Poll
according to a new poll.
About half of the respondents to the poll from USA Today/Ipsos reported feeling “burned out,” 43% said they were “anxious,” and 21% said they were “angry” about politics and abuse from patients and families.
“We’re trying to help people here, and we are getting verbally and physically abused for it,” Sarah Fried, a nurse in California who responded to the survey, told USA Today in a follow-up interview.
“Early in this pandemic, people were clapping for us and calling us heroes,” she said. “And what happened to that? What happened to them appreciating what nurses are doing?”
The poll was done Feb. 9-16 among 1,170 adults in the U.S. health care industry, including doctors, nurses, paramedics, therapists, home health aides, dentists, and other medical professionals.
A large majority of workers still reported being satisfied with their jobs, although that optimism has declined somewhat since early 2021 when the COVID-19 vaccine rollout was underway. About 80% of those in the recent poll said they were somewhat or very satisfied with their current job, which is down from 89% in an April 2021 poll from Kaiser Family Foundation/the Washington Post.
Most health care workers reported feeling “hopeful” (59%), “motivated” (59%), or “optimistic” (56%) about going to work. But “hopeful” is down from 76% and “optimistic” is down from 67%, compared with last year.
If they could pick a career over again, about 16% disagreed with the statement, “I would still decide to go into health care,” and 18% said they didn’t know how they felt about it.
“The pandemic has actually made me realize how important this career is and how I really do make a difference. I still love it,” Christina Rosa, a mental health counselor in Massachusetts, told USA Today.
During the pandemic, about 66% of those polled said they had treated a COVID-19 patient, which increased to 84% among nurses and 86% among hospital workers. Among those, 47% reported having a patient who died from COVID-19, including 53% of nurses and 55% of hospital workers.
What’s more, 81% of those who treated COVID-19 patients have cared for unvaccinated patients. Among those, 67% said their patients continued to express skepticism toward COVID-19 vaccines, and 38% said some patients expressed regret for not getting a vaccine. Beyond that, 26% said unvaccinated patients asked for unproven treatments, and 30% said the patient or family criticized the care they received.
Regarding coronavirus-related policy, most Americans working in health care expressed skepticism or criticism of the nation’s handling of the pandemic. About 39% agreed that the American health care system is “on the verge of collapse.”
Only 21% said the pandemic is mostly or completely under control. About 61% don’t think Americans are taking enough precautions to prevent the spread of the coronavirus.
Health care workers were slightly positive when it comes to the Centers for Disease Control and Prevention (54% approve, 34% disapprove), divided on the Biden administration (41% approve, 40% disapprove), and critical of the news media (20% approve, 61% disapprove) and the American public (18% approve, 68% disapprove).
Broadly, though, health care workers support public health efforts. About 85% back measures that provide N95 masks, and 83% back measures that provide COVID-19 tests.
A version of this article first appeared on WebMD.com.
according to a new poll.
About half of the respondents to the poll from USA Today/Ipsos reported feeling “burned out,” 43% said they were “anxious,” and 21% said they were “angry” about politics and abuse from patients and families.
“We’re trying to help people here, and we are getting verbally and physically abused for it,” Sarah Fried, a nurse in California who responded to the survey, told USA Today in a follow-up interview.
“Early in this pandemic, people were clapping for us and calling us heroes,” she said. “And what happened to that? What happened to them appreciating what nurses are doing?”
The poll was done Feb. 9-16 among 1,170 adults in the U.S. health care industry, including doctors, nurses, paramedics, therapists, home health aides, dentists, and other medical professionals.
A large majority of workers still reported being satisfied with their jobs, although that optimism has declined somewhat since early 2021 when the COVID-19 vaccine rollout was underway. About 80% of those in the recent poll said they were somewhat or very satisfied with their current job, which is down from 89% in an April 2021 poll from Kaiser Family Foundation/the Washington Post.
Most health care workers reported feeling “hopeful” (59%), “motivated” (59%), or “optimistic” (56%) about going to work. But “hopeful” is down from 76% and “optimistic” is down from 67%, compared with last year.
If they could pick a career over again, about 16% disagreed with the statement, “I would still decide to go into health care,” and 18% said they didn’t know how they felt about it.
“The pandemic has actually made me realize how important this career is and how I really do make a difference. I still love it,” Christina Rosa, a mental health counselor in Massachusetts, told USA Today.
During the pandemic, about 66% of those polled said they had treated a COVID-19 patient, which increased to 84% among nurses and 86% among hospital workers. Among those, 47% reported having a patient who died from COVID-19, including 53% of nurses and 55% of hospital workers.
What’s more, 81% of those who treated COVID-19 patients have cared for unvaccinated patients. Among those, 67% said their patients continued to express skepticism toward COVID-19 vaccines, and 38% said some patients expressed regret for not getting a vaccine. Beyond that, 26% said unvaccinated patients asked for unproven treatments, and 30% said the patient or family criticized the care they received.
Regarding coronavirus-related policy, most Americans working in health care expressed skepticism or criticism of the nation’s handling of the pandemic. About 39% agreed that the American health care system is “on the verge of collapse.”
Only 21% said the pandemic is mostly or completely under control. About 61% don’t think Americans are taking enough precautions to prevent the spread of the coronavirus.
Health care workers were slightly positive when it comes to the Centers for Disease Control and Prevention (54% approve, 34% disapprove), divided on the Biden administration (41% approve, 40% disapprove), and critical of the news media (20% approve, 61% disapprove) and the American public (18% approve, 68% disapprove).
Broadly, though, health care workers support public health efforts. About 85% back measures that provide N95 masks, and 83% back measures that provide COVID-19 tests.
A version of this article first appeared on WebMD.com.
according to a new poll.
About half of the respondents to the poll from USA Today/Ipsos reported feeling “burned out,” 43% said they were “anxious,” and 21% said they were “angry” about politics and abuse from patients and families.
“We’re trying to help people here, and we are getting verbally and physically abused for it,” Sarah Fried, a nurse in California who responded to the survey, told USA Today in a follow-up interview.
“Early in this pandemic, people were clapping for us and calling us heroes,” she said. “And what happened to that? What happened to them appreciating what nurses are doing?”
The poll was done Feb. 9-16 among 1,170 adults in the U.S. health care industry, including doctors, nurses, paramedics, therapists, home health aides, dentists, and other medical professionals.
A large majority of workers still reported being satisfied with their jobs, although that optimism has declined somewhat since early 2021 when the COVID-19 vaccine rollout was underway. About 80% of those in the recent poll said they were somewhat or very satisfied with their current job, which is down from 89% in an April 2021 poll from Kaiser Family Foundation/the Washington Post.
Most health care workers reported feeling “hopeful” (59%), “motivated” (59%), or “optimistic” (56%) about going to work. But “hopeful” is down from 76% and “optimistic” is down from 67%, compared with last year.
If they could pick a career over again, about 16% disagreed with the statement, “I would still decide to go into health care,” and 18% said they didn’t know how they felt about it.
“The pandemic has actually made me realize how important this career is and how I really do make a difference. I still love it,” Christina Rosa, a mental health counselor in Massachusetts, told USA Today.
During the pandemic, about 66% of those polled said they had treated a COVID-19 patient, which increased to 84% among nurses and 86% among hospital workers. Among those, 47% reported having a patient who died from COVID-19, including 53% of nurses and 55% of hospital workers.
What’s more, 81% of those who treated COVID-19 patients have cared for unvaccinated patients. Among those, 67% said their patients continued to express skepticism toward COVID-19 vaccines, and 38% said some patients expressed regret for not getting a vaccine. Beyond that, 26% said unvaccinated patients asked for unproven treatments, and 30% said the patient or family criticized the care they received.
Regarding coronavirus-related policy, most Americans working in health care expressed skepticism or criticism of the nation’s handling of the pandemic. About 39% agreed that the American health care system is “on the verge of collapse.”
Only 21% said the pandemic is mostly or completely under control. About 61% don’t think Americans are taking enough precautions to prevent the spread of the coronavirus.
Health care workers were slightly positive when it comes to the Centers for Disease Control and Prevention (54% approve, 34% disapprove), divided on the Biden administration (41% approve, 40% disapprove), and critical of the news media (20% approve, 61% disapprove) and the American public (18% approve, 68% disapprove).
Broadly, though, health care workers support public health efforts. About 85% back measures that provide N95 masks, and 83% back measures that provide COVID-19 tests.
A version of this article first appeared on WebMD.com.
Your heart doesn’t like peas any more than you do
Big Vegetable has lied to us all
Hear this, children of the world: Your parents have betrayed you. They tell you day in and day out that vegetables are necessary, that they’re healthy, that you need them, but it is not the truth. Behind their foul taste is nothing but empty lies.
Okay, before we get a full-blown child rebellion on our hands, let’s reel things in. Eating vegetables has many benefits, and will help prevent many nasty medical conditions, such as diabetes or cancer. However, cardiovascular disease is not among them.
For their study published in Frontiers in Nutrition, researchers analyzed the diet, lifestyle, and medical history of nearly 400,000 U.K. adults over a 5-year period, finding that 4.5% developed heart disease and that the average adult consumed about 5 tablespoons of vegetables per day. Those who consumed the most vegetables had a reduction in heart disease incidence of about 15%, compared with those who ate the least.
Hang on, you’re thinking, we just said that vegetables didn’t prevent cardiovascular disease. But the data show otherwise! Ah, but the data are unadjusted. Once the researchers took socioeconomic status, information level, and general lifestyle into account, that benefit disappeared almost completely. The benefit seems to come not from the vegetables themselves, but from being able to afford better food and medical care in general.
The researchers were quick to note the other benefits of eating vegetables, and that people should probably keep eating those five servings a day. But we’re onto you, scientists. You can’t fool us with your vegetable-based lies. Unless we’re talking about pizza. Pizza is the best vegetable.
The good old days of surgery?
Modern surgical instruments, techniques, and technological innovations are amazing. It’s hard to imagine what surgery was like before laparoscopes came along, or x-ray machines, or even anesthesia. But those days weren’t really that long ago. Modern anesthesia, after all, dates back to just 1846. We’ve got socks almost that old.
But suppose we go back even further … say 5,300 years. Older than the oldest sock. Scientists studying a funerary chamber in Burgos, Spain, which was built in the 4th millennium B.C., have come across what looks like “the first known radical mastoidectomy in the history of humankind,” Sonia Díaz-Navarro of the University of Valladolid (Spain) and associates wrote in Scientific Reports.
One of the skulls they uncovered shows signs of trepanation. “Despite the [evidence] of cut marks, it is difficult to conclude the type of tool used to remove the bone tissue, most likely a sharp instrument with a circular movement,” they investigators said.
What is clear, though, is that the patient survived the surgery, because there is evidence of bone regeneration at the surgical sites. Sites? “Based on the differences in bone remodelling between the two temporals, it appears that the procedure was first conducted on the right ear, due to an ear pathology sufficiently alarming to require an intervention, which this prehistoric woman survived,” they explained.
The same procedure was then performed on the left ear, “but whether this was performed shortly after the right ear, or several months or even years later can’t be concluded from the existing evidence,” IFL Science reported.
Located nearby was a small section of tree bark with some scratches on it. That, ladies and gentlemen, was the first prior authorization form.
I hate that song, with reason
Do you have a favorite song? You may have a million reasons for loving that song. And past research can tell you why. But it’s only in a recent study that researchers were able to tell you why you dislike a song. And you know the song we’re talking about.
Dislike breaks down into three major categories of rationale: subject-related reasons (how the song makes you feel emotionally and/or physically), object-related reasons (the lyrics or composition), and social reasons (do you relate to this?). Researchers at the Max Planck Institute for Empirical Aesthetics in Frankfurt, Germany, interviewed 21 participants and asked them to come up with a prepared list of music that they disliked and why they didn’t like it. And there was a lot that they didn’t like: 277 dislikes worth, to be exact.
“The most often mentioned type of dislike was musical style, followed by artist and genre,” senior author Julia Merrill explained on Eurekalert. Just over 40% of those rationales for not liking the music just had to do with the music itself, but 85% involved the music combined with one of the other categories.
Social reasoning played a big part in dislike. If the listener didn’t feel like a part of the target in-group for the music or the music didn’t have the same social values as those of the listener, it had an impact on dislike, they said.
But our dislike of certain types of music doesn’t just separate us from people in a negative way. Looking at the dislike of certain types of music helps us define our terms of having good taste, the researchers explained. Saying that one type of music is better than another can bring us closer with like-minded people and becomes a piece of how we identify ourselves. Cue the music snobs.
So if you can blast Barry Manilow but can’t bring yourself to play the Rolling Stones, there’s a reason for that. And if you love Aretha Franklin but not Frank Sinatra, there’s a reason for that, too. It’s all very personal. Just as music is meant to be.
Big Vegetable has lied to us all
Hear this, children of the world: Your parents have betrayed you. They tell you day in and day out that vegetables are necessary, that they’re healthy, that you need them, but it is not the truth. Behind their foul taste is nothing but empty lies.
Okay, before we get a full-blown child rebellion on our hands, let’s reel things in. Eating vegetables has many benefits, and will help prevent many nasty medical conditions, such as diabetes or cancer. However, cardiovascular disease is not among them.
For their study published in Frontiers in Nutrition, researchers analyzed the diet, lifestyle, and medical history of nearly 400,000 U.K. adults over a 5-year period, finding that 4.5% developed heart disease and that the average adult consumed about 5 tablespoons of vegetables per day. Those who consumed the most vegetables had a reduction in heart disease incidence of about 15%, compared with those who ate the least.
Hang on, you’re thinking, we just said that vegetables didn’t prevent cardiovascular disease. But the data show otherwise! Ah, but the data are unadjusted. Once the researchers took socioeconomic status, information level, and general lifestyle into account, that benefit disappeared almost completely. The benefit seems to come not from the vegetables themselves, but from being able to afford better food and medical care in general.
The researchers were quick to note the other benefits of eating vegetables, and that people should probably keep eating those five servings a day. But we’re onto you, scientists. You can’t fool us with your vegetable-based lies. Unless we’re talking about pizza. Pizza is the best vegetable.
The good old days of surgery?
Modern surgical instruments, techniques, and technological innovations are amazing. It’s hard to imagine what surgery was like before laparoscopes came along, or x-ray machines, or even anesthesia. But those days weren’t really that long ago. Modern anesthesia, after all, dates back to just 1846. We’ve got socks almost that old.
But suppose we go back even further … say 5,300 years. Older than the oldest sock. Scientists studying a funerary chamber in Burgos, Spain, which was built in the 4th millennium B.C., have come across what looks like “the first known radical mastoidectomy in the history of humankind,” Sonia Díaz-Navarro of the University of Valladolid (Spain) and associates wrote in Scientific Reports.
One of the skulls they uncovered shows signs of trepanation. “Despite the [evidence] of cut marks, it is difficult to conclude the type of tool used to remove the bone tissue, most likely a sharp instrument with a circular movement,” they investigators said.
What is clear, though, is that the patient survived the surgery, because there is evidence of bone regeneration at the surgical sites. Sites? “Based on the differences in bone remodelling between the two temporals, it appears that the procedure was first conducted on the right ear, due to an ear pathology sufficiently alarming to require an intervention, which this prehistoric woman survived,” they explained.
The same procedure was then performed on the left ear, “but whether this was performed shortly after the right ear, or several months or even years later can’t be concluded from the existing evidence,” IFL Science reported.
Located nearby was a small section of tree bark with some scratches on it. That, ladies and gentlemen, was the first prior authorization form.
I hate that song, with reason
Do you have a favorite song? You may have a million reasons for loving that song. And past research can tell you why. But it’s only in a recent study that researchers were able to tell you why you dislike a song. And you know the song we’re talking about.
Dislike breaks down into three major categories of rationale: subject-related reasons (how the song makes you feel emotionally and/or physically), object-related reasons (the lyrics or composition), and social reasons (do you relate to this?). Researchers at the Max Planck Institute for Empirical Aesthetics in Frankfurt, Germany, interviewed 21 participants and asked them to come up with a prepared list of music that they disliked and why they didn’t like it. And there was a lot that they didn’t like: 277 dislikes worth, to be exact.
“The most often mentioned type of dislike was musical style, followed by artist and genre,” senior author Julia Merrill explained on Eurekalert. Just over 40% of those rationales for not liking the music just had to do with the music itself, but 85% involved the music combined with one of the other categories.
Social reasoning played a big part in dislike. If the listener didn’t feel like a part of the target in-group for the music or the music didn’t have the same social values as those of the listener, it had an impact on dislike, they said.
But our dislike of certain types of music doesn’t just separate us from people in a negative way. Looking at the dislike of certain types of music helps us define our terms of having good taste, the researchers explained. Saying that one type of music is better than another can bring us closer with like-minded people and becomes a piece of how we identify ourselves. Cue the music snobs.
So if you can blast Barry Manilow but can’t bring yourself to play the Rolling Stones, there’s a reason for that. And if you love Aretha Franklin but not Frank Sinatra, there’s a reason for that, too. It’s all very personal. Just as music is meant to be.
Big Vegetable has lied to us all
Hear this, children of the world: Your parents have betrayed you. They tell you day in and day out that vegetables are necessary, that they’re healthy, that you need them, but it is not the truth. Behind their foul taste is nothing but empty lies.
Okay, before we get a full-blown child rebellion on our hands, let’s reel things in. Eating vegetables has many benefits, and will help prevent many nasty medical conditions, such as diabetes or cancer. However, cardiovascular disease is not among them.
For their study published in Frontiers in Nutrition, researchers analyzed the diet, lifestyle, and medical history of nearly 400,000 U.K. adults over a 5-year period, finding that 4.5% developed heart disease and that the average adult consumed about 5 tablespoons of vegetables per day. Those who consumed the most vegetables had a reduction in heart disease incidence of about 15%, compared with those who ate the least.
Hang on, you’re thinking, we just said that vegetables didn’t prevent cardiovascular disease. But the data show otherwise! Ah, but the data are unadjusted. Once the researchers took socioeconomic status, information level, and general lifestyle into account, that benefit disappeared almost completely. The benefit seems to come not from the vegetables themselves, but from being able to afford better food and medical care in general.
The researchers were quick to note the other benefits of eating vegetables, and that people should probably keep eating those five servings a day. But we’re onto you, scientists. You can’t fool us with your vegetable-based lies. Unless we’re talking about pizza. Pizza is the best vegetable.
The good old days of surgery?
Modern surgical instruments, techniques, and technological innovations are amazing. It’s hard to imagine what surgery was like before laparoscopes came along, or x-ray machines, or even anesthesia. But those days weren’t really that long ago. Modern anesthesia, after all, dates back to just 1846. We’ve got socks almost that old.
But suppose we go back even further … say 5,300 years. Older than the oldest sock. Scientists studying a funerary chamber in Burgos, Spain, which was built in the 4th millennium B.C., have come across what looks like “the first known radical mastoidectomy in the history of humankind,” Sonia Díaz-Navarro of the University of Valladolid (Spain) and associates wrote in Scientific Reports.
One of the skulls they uncovered shows signs of trepanation. “Despite the [evidence] of cut marks, it is difficult to conclude the type of tool used to remove the bone tissue, most likely a sharp instrument with a circular movement,” they investigators said.
What is clear, though, is that the patient survived the surgery, because there is evidence of bone regeneration at the surgical sites. Sites? “Based on the differences in bone remodelling between the two temporals, it appears that the procedure was first conducted on the right ear, due to an ear pathology sufficiently alarming to require an intervention, which this prehistoric woman survived,” they explained.
The same procedure was then performed on the left ear, “but whether this was performed shortly after the right ear, or several months or even years later can’t be concluded from the existing evidence,” IFL Science reported.
Located nearby was a small section of tree bark with some scratches on it. That, ladies and gentlemen, was the first prior authorization form.
I hate that song, with reason
Do you have a favorite song? You may have a million reasons for loving that song. And past research can tell you why. But it’s only in a recent study that researchers were able to tell you why you dislike a song. And you know the song we’re talking about.
Dislike breaks down into three major categories of rationale: subject-related reasons (how the song makes you feel emotionally and/or physically), object-related reasons (the lyrics or composition), and social reasons (do you relate to this?). Researchers at the Max Planck Institute for Empirical Aesthetics in Frankfurt, Germany, interviewed 21 participants and asked them to come up with a prepared list of music that they disliked and why they didn’t like it. And there was a lot that they didn’t like: 277 dislikes worth, to be exact.
“The most often mentioned type of dislike was musical style, followed by artist and genre,” senior author Julia Merrill explained on Eurekalert. Just over 40% of those rationales for not liking the music just had to do with the music itself, but 85% involved the music combined with one of the other categories.
Social reasoning played a big part in dislike. If the listener didn’t feel like a part of the target in-group for the music or the music didn’t have the same social values as those of the listener, it had an impact on dislike, they said.
But our dislike of certain types of music doesn’t just separate us from people in a negative way. Looking at the dislike of certain types of music helps us define our terms of having good taste, the researchers explained. Saying that one type of music is better than another can bring us closer with like-minded people and becomes a piece of how we identify ourselves. Cue the music snobs.
So if you can blast Barry Manilow but can’t bring yourself to play the Rolling Stones, there’s a reason for that. And if you love Aretha Franklin but not Frank Sinatra, there’s a reason for that, too. It’s all very personal. Just as music is meant to be.
Psychiatrist found guilty in $110 million fraud scheme
Mark Agresti, MD, 59, was convicted of unlawfully billing approximately $110 million of medically unnecessary urinalysis services for patients living at Good Decisions Sober Living (GDSL), a drug rehabilitation facility in West Palm Beach, Fla. Dr. Agresti was once the director of psychiatry at the former Columbia Hospital in West Palm Beach.
According to evidence presented at the trial, Dr. Agresti, as medical director of GDSL, agreed to provide standing orders for urinalysis for GDSL patients in exchange for a monthly fee. Three to four times a week, patients were required to submit to excessive and medically unnecessary urine tests, costing $6,000 to $9,000 per test, according to federal officials. Dr. Agresti did not review the results of these tests and did not use the tests to treat patients. The evidence also showed that he had also done this at other addiction facilities in the West Palm Beach area, resulting in thousands of additional fraudulent tests. Dr. Agresti had GDSL patients visit his private practice so he could fraudulently bill for services there as well.
“This defendant used his medical license to facilitate an egregious, multimillion-dollar fraud scheme that exploited vulnerable substance abusers,” said Juan Antonio Gonzalez, U.S. Attorney for the Southern District of Florida, in a statement announcing the conviction.
Three other defendants, including GDSL’s owner, Kenneth Bailynson, were previously indicted and pleaded guilty to related charges connected with the scheme. Dr. Agresti, however, insisted on his innocence throughout the trial, arguing that he was himself the victim of Mr. Bailynson. Mr. Bailynson, as a part of his plea deal, testified against Dr. Agresti at the trial.
According to a report in the Palm Beach Post, Dr. Agresti’s lawyers, in closing statements, argued, “The government did a fantastic job of proving that Dr. Agresti was negligent, maybe even grossly negligent, but that’s not the issue in this case.” The jury disagreed.
Dr. Agresti faces a maximum penalty of 20 years in prison for the healthcare fraud and wire fraud conspiracy charges and 10 years for each count of healthcare fraud. Sentencing is scheduled for April 21.
Ten people, including two physicians, charged in Texas kickback scheme
Jose Maldonado, MD, 48, a family medicine physician, and Eduardo Carlos Canova, MD, 44, an internal medicine specialist, both based in Laredo, Tex., were among ten people indicted in connection with a $300 million healthcare fraud scheme
According to the federal indictment, the founders of several lab companies, including Unified Laboratory Services, Spectrum Diagnostic Laboratory, and Reliable Labs, LLC, allegedly paid kickbacks to medical practitioners in exchange for orders for medically unnecessary lab tests, which were then billed to Medicare and other federal healthcare programs. The medical professionals, including Dr. Maldonado and Dr. Canova, are alleged to have accepted bribes for ordering millions of dollars’ worth of tests.
The scheme was facilitated by marketing firms, which paid doctors hundreds of thousands of dollars for “advisory services,” according to officials. Rather than providing advice, however, the physicians provided lab test referrals. The labs are accused of paying portions of the salaries of the doctors’ staffs as well as portions of their leases. In some instances, direct payments were made to the practitioner’s spouse. In one case, the labs informed one practitioner that the payments would cease if he didn’t refer more tests. He responded to the threat by immediately increasing his lab referrals, averaging 20 to 30 referrals a day.
Jeffrey Madison, the founder of Spectrum and Unified, is accused of convincing Reliable’s founders to convert their business to a physician-owned lab to disguise the kickbacks. Reliable offered physicians ownership opportunities only if they referred enough lab tests. In some cases, says the indictment, the labs made advance payments to physicians to ensure that those physicians would not send samples to other labs.
The scheme resulted in more than $300 million in billing to federal government healthcare programs. Between 2015 and 2018, Dr. Maldonado alone received more than $400,000 in kickbacks for ordering more than $4 million worth of lab tests, and Dr. Canova received more than $300,000 for ordering more than $12 million in tests, according to the indictment.
The 26-count indictment included charges of conspiracy to commit healthcare fraud, conspiracy to pay and receive healthcare kickbacks, offering or paying illegal kickbacks, and soliciting or receiving illegal kickbacks.
N.J. doc charged on 34 counts of illegally prescribing controlled substances
A N.J. orthopedic surgeon has been charged with 34 counts of illegally prescribing controlled substances.
According to documents filed in the case, from November 2016 to February 2018 Evangelos Megariotis, MD, 68, of Passaic County, N.J., prescribed opiates — including oxycodone, alprazolam (Xanax), and cough syrup with codeine — as well as stimulants without a legitimate medical purpose and outside the usual course of his practice.
Before the State of New Jersey permanently banned him from practicing medicine, Dr. Megariotis owned and operated Clifton Orthopedic Associates, in Clifton, N.J.
According to The Bergin Record, the state alleges that Dr. Megariotis prescribed thousands of doses of painkillers even though he knew his patients were abusing them and in some cases were getting similar drugs from other doctors. In addition, Dr. Megariotis failed to diagnose or treat these patients’ underlying conditions, treated illnesses outside his scope of practice, and performed surgery in the absence of medical need.
If convicted, Dr. Megariotis could face a lengthy prison sentence as well as substantial fines. The counts of illegally prescribing Schedule II opioids and stimulants are punishable by a maximum of 20 years in prison and a $1 million fine. Illegally prescribing Schedule IV controlled substances brings a maximum of 5 years in prison and a $250,000 fine. The counts of illegally prescribing cough syrup with codeine, a Schedule V controlled substance, are punishable by a maximum of 1 year in prison and a $100,000 fine.
Michigan surgeon pleads guilty to fraud scheme
A Bay City, Mich., vascular surgeon has pleaded guilty to engaging in a scheme to defraud Medicare, Medicaid, and Blue Cross/Blue Shield of Michigan out of approximately $19.5 million
According to the plea agreement, in 2009, Vasso Godiali, MD, 59, began false and fraudulent billing to the three insurers. False claims included billing for the placement of stents in dialysis patients and for the treatment of arterial blood clots. The surgeon admitted to billing for the placement of multiple stents in the same vessel and preparing medical records to support the necessity of placing those stents. However, he never placed the stents.
In the plea document, Dr. Godiali also admitted that his medical records would describe occluded arteries that would justify arterial thrombectomies. However, in many cases, there were no occluded arteries, and no thrombectomies were performed, even though Dr. Godiali billed insurers for them.
“When Dr Godiali submitted claims for medical services that were never provided, he violated the trust of his patients and defrauded taxpayer-funded health care programs,” Mario M. Pinto, special agent in charge, said in a statement announcing the plea.
In 2014, The Bay City Times interviewed Dr. Godiali after he was featured by USA Today as one of seven physicians in the United States to receive more than $10 million for Medicare reimbursements in 2012. He told The Times he was a workaholic. “I work a lot — I work probably more than surgeons that you meet,” he told the paper.
Dr. Godiali faces up to 10 years in prison. In addition, he will be required to pay $19.5 million in restitution to the three insurers. A civil forfeiture case against approximately $39.9 million seized from accounts controlled by Dr. Godiali is pending. Sentencing is scheduled for September 15.
A version of this article first appeared on Medscape.com.
Mark Agresti, MD, 59, was convicted of unlawfully billing approximately $110 million of medically unnecessary urinalysis services for patients living at Good Decisions Sober Living (GDSL), a drug rehabilitation facility in West Palm Beach, Fla. Dr. Agresti was once the director of psychiatry at the former Columbia Hospital in West Palm Beach.
According to evidence presented at the trial, Dr. Agresti, as medical director of GDSL, agreed to provide standing orders for urinalysis for GDSL patients in exchange for a monthly fee. Three to four times a week, patients were required to submit to excessive and medically unnecessary urine tests, costing $6,000 to $9,000 per test, according to federal officials. Dr. Agresti did not review the results of these tests and did not use the tests to treat patients. The evidence also showed that he had also done this at other addiction facilities in the West Palm Beach area, resulting in thousands of additional fraudulent tests. Dr. Agresti had GDSL patients visit his private practice so he could fraudulently bill for services there as well.
“This defendant used his medical license to facilitate an egregious, multimillion-dollar fraud scheme that exploited vulnerable substance abusers,” said Juan Antonio Gonzalez, U.S. Attorney for the Southern District of Florida, in a statement announcing the conviction.
Three other defendants, including GDSL’s owner, Kenneth Bailynson, were previously indicted and pleaded guilty to related charges connected with the scheme. Dr. Agresti, however, insisted on his innocence throughout the trial, arguing that he was himself the victim of Mr. Bailynson. Mr. Bailynson, as a part of his plea deal, testified against Dr. Agresti at the trial.
According to a report in the Palm Beach Post, Dr. Agresti’s lawyers, in closing statements, argued, “The government did a fantastic job of proving that Dr. Agresti was negligent, maybe even grossly negligent, but that’s not the issue in this case.” The jury disagreed.
Dr. Agresti faces a maximum penalty of 20 years in prison for the healthcare fraud and wire fraud conspiracy charges and 10 years for each count of healthcare fraud. Sentencing is scheduled for April 21.
Ten people, including two physicians, charged in Texas kickback scheme
Jose Maldonado, MD, 48, a family medicine physician, and Eduardo Carlos Canova, MD, 44, an internal medicine specialist, both based in Laredo, Tex., were among ten people indicted in connection with a $300 million healthcare fraud scheme
According to the federal indictment, the founders of several lab companies, including Unified Laboratory Services, Spectrum Diagnostic Laboratory, and Reliable Labs, LLC, allegedly paid kickbacks to medical practitioners in exchange for orders for medically unnecessary lab tests, which were then billed to Medicare and other federal healthcare programs. The medical professionals, including Dr. Maldonado and Dr. Canova, are alleged to have accepted bribes for ordering millions of dollars’ worth of tests.
The scheme was facilitated by marketing firms, which paid doctors hundreds of thousands of dollars for “advisory services,” according to officials. Rather than providing advice, however, the physicians provided lab test referrals. The labs are accused of paying portions of the salaries of the doctors’ staffs as well as portions of their leases. In some instances, direct payments were made to the practitioner’s spouse. In one case, the labs informed one practitioner that the payments would cease if he didn’t refer more tests. He responded to the threat by immediately increasing his lab referrals, averaging 20 to 30 referrals a day.
Jeffrey Madison, the founder of Spectrum and Unified, is accused of convincing Reliable’s founders to convert their business to a physician-owned lab to disguise the kickbacks. Reliable offered physicians ownership opportunities only if they referred enough lab tests. In some cases, says the indictment, the labs made advance payments to physicians to ensure that those physicians would not send samples to other labs.
The scheme resulted in more than $300 million in billing to federal government healthcare programs. Between 2015 and 2018, Dr. Maldonado alone received more than $400,000 in kickbacks for ordering more than $4 million worth of lab tests, and Dr. Canova received more than $300,000 for ordering more than $12 million in tests, according to the indictment.
The 26-count indictment included charges of conspiracy to commit healthcare fraud, conspiracy to pay and receive healthcare kickbacks, offering or paying illegal kickbacks, and soliciting or receiving illegal kickbacks.
N.J. doc charged on 34 counts of illegally prescribing controlled substances
A N.J. orthopedic surgeon has been charged with 34 counts of illegally prescribing controlled substances.
According to documents filed in the case, from November 2016 to February 2018 Evangelos Megariotis, MD, 68, of Passaic County, N.J., prescribed opiates — including oxycodone, alprazolam (Xanax), and cough syrup with codeine — as well as stimulants without a legitimate medical purpose and outside the usual course of his practice.
Before the State of New Jersey permanently banned him from practicing medicine, Dr. Megariotis owned and operated Clifton Orthopedic Associates, in Clifton, N.J.
According to The Bergin Record, the state alleges that Dr. Megariotis prescribed thousands of doses of painkillers even though he knew his patients were abusing them and in some cases were getting similar drugs from other doctors. In addition, Dr. Megariotis failed to diagnose or treat these patients’ underlying conditions, treated illnesses outside his scope of practice, and performed surgery in the absence of medical need.
If convicted, Dr. Megariotis could face a lengthy prison sentence as well as substantial fines. The counts of illegally prescribing Schedule II opioids and stimulants are punishable by a maximum of 20 years in prison and a $1 million fine. Illegally prescribing Schedule IV controlled substances brings a maximum of 5 years in prison and a $250,000 fine. The counts of illegally prescribing cough syrup with codeine, a Schedule V controlled substance, are punishable by a maximum of 1 year in prison and a $100,000 fine.
Michigan surgeon pleads guilty to fraud scheme
A Bay City, Mich., vascular surgeon has pleaded guilty to engaging in a scheme to defraud Medicare, Medicaid, and Blue Cross/Blue Shield of Michigan out of approximately $19.5 million
According to the plea agreement, in 2009, Vasso Godiali, MD, 59, began false and fraudulent billing to the three insurers. False claims included billing for the placement of stents in dialysis patients and for the treatment of arterial blood clots. The surgeon admitted to billing for the placement of multiple stents in the same vessel and preparing medical records to support the necessity of placing those stents. However, he never placed the stents.
In the plea document, Dr. Godiali also admitted that his medical records would describe occluded arteries that would justify arterial thrombectomies. However, in many cases, there were no occluded arteries, and no thrombectomies were performed, even though Dr. Godiali billed insurers for them.
“When Dr Godiali submitted claims for medical services that were never provided, he violated the trust of his patients and defrauded taxpayer-funded health care programs,” Mario M. Pinto, special agent in charge, said in a statement announcing the plea.
In 2014, The Bay City Times interviewed Dr. Godiali after he was featured by USA Today as one of seven physicians in the United States to receive more than $10 million for Medicare reimbursements in 2012. He told The Times he was a workaholic. “I work a lot — I work probably more than surgeons that you meet,” he told the paper.
Dr. Godiali faces up to 10 years in prison. In addition, he will be required to pay $19.5 million in restitution to the three insurers. A civil forfeiture case against approximately $39.9 million seized from accounts controlled by Dr. Godiali is pending. Sentencing is scheduled for September 15.
A version of this article first appeared on Medscape.com.
Mark Agresti, MD, 59, was convicted of unlawfully billing approximately $110 million of medically unnecessary urinalysis services for patients living at Good Decisions Sober Living (GDSL), a drug rehabilitation facility in West Palm Beach, Fla. Dr. Agresti was once the director of psychiatry at the former Columbia Hospital in West Palm Beach.
According to evidence presented at the trial, Dr. Agresti, as medical director of GDSL, agreed to provide standing orders for urinalysis for GDSL patients in exchange for a monthly fee. Three to four times a week, patients were required to submit to excessive and medically unnecessary urine tests, costing $6,000 to $9,000 per test, according to federal officials. Dr. Agresti did not review the results of these tests and did not use the tests to treat patients. The evidence also showed that he had also done this at other addiction facilities in the West Palm Beach area, resulting in thousands of additional fraudulent tests. Dr. Agresti had GDSL patients visit his private practice so he could fraudulently bill for services there as well.
“This defendant used his medical license to facilitate an egregious, multimillion-dollar fraud scheme that exploited vulnerable substance abusers,” said Juan Antonio Gonzalez, U.S. Attorney for the Southern District of Florida, in a statement announcing the conviction.
Three other defendants, including GDSL’s owner, Kenneth Bailynson, were previously indicted and pleaded guilty to related charges connected with the scheme. Dr. Agresti, however, insisted on his innocence throughout the trial, arguing that he was himself the victim of Mr. Bailynson. Mr. Bailynson, as a part of his plea deal, testified against Dr. Agresti at the trial.
According to a report in the Palm Beach Post, Dr. Agresti’s lawyers, in closing statements, argued, “The government did a fantastic job of proving that Dr. Agresti was negligent, maybe even grossly negligent, but that’s not the issue in this case.” The jury disagreed.
Dr. Agresti faces a maximum penalty of 20 years in prison for the healthcare fraud and wire fraud conspiracy charges and 10 years for each count of healthcare fraud. Sentencing is scheduled for April 21.
Ten people, including two physicians, charged in Texas kickback scheme
Jose Maldonado, MD, 48, a family medicine physician, and Eduardo Carlos Canova, MD, 44, an internal medicine specialist, both based in Laredo, Tex., were among ten people indicted in connection with a $300 million healthcare fraud scheme
According to the federal indictment, the founders of several lab companies, including Unified Laboratory Services, Spectrum Diagnostic Laboratory, and Reliable Labs, LLC, allegedly paid kickbacks to medical practitioners in exchange for orders for medically unnecessary lab tests, which were then billed to Medicare and other federal healthcare programs. The medical professionals, including Dr. Maldonado and Dr. Canova, are alleged to have accepted bribes for ordering millions of dollars’ worth of tests.
The scheme was facilitated by marketing firms, which paid doctors hundreds of thousands of dollars for “advisory services,” according to officials. Rather than providing advice, however, the physicians provided lab test referrals. The labs are accused of paying portions of the salaries of the doctors’ staffs as well as portions of their leases. In some instances, direct payments were made to the practitioner’s spouse. In one case, the labs informed one practitioner that the payments would cease if he didn’t refer more tests. He responded to the threat by immediately increasing his lab referrals, averaging 20 to 30 referrals a day.
Jeffrey Madison, the founder of Spectrum and Unified, is accused of convincing Reliable’s founders to convert their business to a physician-owned lab to disguise the kickbacks. Reliable offered physicians ownership opportunities only if they referred enough lab tests. In some cases, says the indictment, the labs made advance payments to physicians to ensure that those physicians would not send samples to other labs.
The scheme resulted in more than $300 million in billing to federal government healthcare programs. Between 2015 and 2018, Dr. Maldonado alone received more than $400,000 in kickbacks for ordering more than $4 million worth of lab tests, and Dr. Canova received more than $300,000 for ordering more than $12 million in tests, according to the indictment.
The 26-count indictment included charges of conspiracy to commit healthcare fraud, conspiracy to pay and receive healthcare kickbacks, offering or paying illegal kickbacks, and soliciting or receiving illegal kickbacks.
N.J. doc charged on 34 counts of illegally prescribing controlled substances
A N.J. orthopedic surgeon has been charged with 34 counts of illegally prescribing controlled substances.
According to documents filed in the case, from November 2016 to February 2018 Evangelos Megariotis, MD, 68, of Passaic County, N.J., prescribed opiates — including oxycodone, alprazolam (Xanax), and cough syrup with codeine — as well as stimulants without a legitimate medical purpose and outside the usual course of his practice.
Before the State of New Jersey permanently banned him from practicing medicine, Dr. Megariotis owned and operated Clifton Orthopedic Associates, in Clifton, N.J.
According to The Bergin Record, the state alleges that Dr. Megariotis prescribed thousands of doses of painkillers even though he knew his patients were abusing them and in some cases were getting similar drugs from other doctors. In addition, Dr. Megariotis failed to diagnose or treat these patients’ underlying conditions, treated illnesses outside his scope of practice, and performed surgery in the absence of medical need.
If convicted, Dr. Megariotis could face a lengthy prison sentence as well as substantial fines. The counts of illegally prescribing Schedule II opioids and stimulants are punishable by a maximum of 20 years in prison and a $1 million fine. Illegally prescribing Schedule IV controlled substances brings a maximum of 5 years in prison and a $250,000 fine. The counts of illegally prescribing cough syrup with codeine, a Schedule V controlled substance, are punishable by a maximum of 1 year in prison and a $100,000 fine.
Michigan surgeon pleads guilty to fraud scheme
A Bay City, Mich., vascular surgeon has pleaded guilty to engaging in a scheme to defraud Medicare, Medicaid, and Blue Cross/Blue Shield of Michigan out of approximately $19.5 million
According to the plea agreement, in 2009, Vasso Godiali, MD, 59, began false and fraudulent billing to the three insurers. False claims included billing for the placement of stents in dialysis patients and for the treatment of arterial blood clots. The surgeon admitted to billing for the placement of multiple stents in the same vessel and preparing medical records to support the necessity of placing those stents. However, he never placed the stents.
In the plea document, Dr. Godiali also admitted that his medical records would describe occluded arteries that would justify arterial thrombectomies. However, in many cases, there were no occluded arteries, and no thrombectomies were performed, even though Dr. Godiali billed insurers for them.
“When Dr Godiali submitted claims for medical services that were never provided, he violated the trust of his patients and defrauded taxpayer-funded health care programs,” Mario M. Pinto, special agent in charge, said in a statement announcing the plea.
In 2014, The Bay City Times interviewed Dr. Godiali after he was featured by USA Today as one of seven physicians in the United States to receive more than $10 million for Medicare reimbursements in 2012. He told The Times he was a workaholic. “I work a lot — I work probably more than surgeons that you meet,” he told the paper.
Dr. Godiali faces up to 10 years in prison. In addition, he will be required to pay $19.5 million in restitution to the three insurers. A civil forfeiture case against approximately $39.9 million seized from accounts controlled by Dr. Godiali is pending. Sentencing is scheduled for September 15.
A version of this article first appeared on Medscape.com.
Early in career, female academic docs earn less than males: study
Worse still, the earning potential of women in most specialties is $214,440 (or 10%) less than their male colleagues over the course of the first 10 years of their careers in academic medicine.
Among the vast majority of subspecialties, women’s starting salaries and their salaries 10 years into their careers were lower than their male colleagues in academic medicine, per the study in JAMA Network Open.
Eva Catenaccio, MD, an epilepsy fellow at Children’s Hospital of Philadelphia and the lead author of the study, told this news organization that the gender disparities in earning potential are “pervasive in academic medicine.” These earnings disparities, which occur in nearly all subspecialties and can reach hundreds of thousands of dollars in the first 10 years of an academic physician’s career, “are largely the result of gender differences in annual salary that start immediately after training,” she said.
Changing the timing of academic promotion and equalizing starting salary and salary growth can help close the salary gap, said Dr. Catenaccio.
The study also reveals that women could face a 1-year delay in promotion from assistant to associate professor, compared with men. This delay could reduce female physicians’ earning potential by a 10-year median of $26,042 (or 2%), whereas failure to be promoted at all could decrease the 10-year earning potential by a median of $218,724 (or 13%).
Across medicine more broadly, male physicians continue to earn 35% more than their female colleagues, according to the 2021 Medscape Physician Compensation Report. The biggest differences in take-home pay exist between male and female specialists, per the report. On average, male physicians earn $376,000, while women’s take-home pay is $283,000.
Medical schools and hospital leaders have a role to play
The earning potential during the first 10 years of post-training employment by gender was the most dramatic in neurosurgery, orthopedic surgery, and cardiology, per the study. Three subspecialties where women and men have similar earning potential include pediatric nephrology, pediatric neurology, and pediatric rheumatology.
The coauthors note that it’s commonly understood that women don’t negotiate as often or as successfully as their male colleagues. A 2019 study in JAMA Surgery of 606 male and female surgery residents revealed that while residents of both genders shared similar career goals, women had lower future salary expectations and a significantly more negative view of the salary negotiation process.
Dr. Catenaccio and her coauthors acknowledge that negotiation skills and financial literacy should be taught during medical school and postgraduate training. “However, the onus for ensuring salary equity should not fall on the individual candidate alone; rather, departmental and hospital leadership should take responsibility to ensure uniform starting salaries and prevent gender-based inequalities,” they wrote in the study.
“We hope that this study encourages academic medical institutions to increase transparency and equity around compensation, particularly for junior faculty,” Dr. Catenaccio said in an interview. “This will require both ensuring equal starting salaries and providing periodic adjustments throughout individuals’ careers to prevent divergence in earning potential by gender or any other individual characteristics.”
Harold Simon, MD, MBA, vice chair for faculty for the department of pediatrics and professor of pediatrics and emergency medicine at Emory University, Atlanta, told this news organization that “[i]ncreased transparency around compensation can enable women to advocate for equitable pay. However, the burden for ensuring equity should not fall on individuals but instead must be the primary responsibility of academic institutions.”
Specifically, Dr. Simon advocates for hospital leaders to “ensure equity among providers including compensation [as] a crucial part of maintaining a diverse workforce and, ultimately, providing balanced access to health care for patients.”
In addition, the authors call for periodic compensation evaluations and adjustments to help prevent gender-based salary differences among female and male physicians in academia. “This is absolutely necessary, both to develop future compensation plans and to address any pre-existing gender-based salary inequities for those women currently well into their careers,” they wrote in the study.
Data analysis was conducted from March to May 2021. Researchers used models to estimate the impacts of promotion timing and potential interventions, which include equalizing starting salaries and annual salary rates.
The study included compensation data for 24,593 female and 29,886 male academic physicians across 45 subspecialties. It relied on publicly available data from the Association of American Medical Colleges’ annual Medical School Faculty Salary Survey report.
A version of this article first appeared on Medscape.com.
Worse still, the earning potential of women in most specialties is $214,440 (or 10%) less than their male colleagues over the course of the first 10 years of their careers in academic medicine.
Among the vast majority of subspecialties, women’s starting salaries and their salaries 10 years into their careers were lower than their male colleagues in academic medicine, per the study in JAMA Network Open.
Eva Catenaccio, MD, an epilepsy fellow at Children’s Hospital of Philadelphia and the lead author of the study, told this news organization that the gender disparities in earning potential are “pervasive in academic medicine.” These earnings disparities, which occur in nearly all subspecialties and can reach hundreds of thousands of dollars in the first 10 years of an academic physician’s career, “are largely the result of gender differences in annual salary that start immediately after training,” she said.
Changing the timing of academic promotion and equalizing starting salary and salary growth can help close the salary gap, said Dr. Catenaccio.
The study also reveals that women could face a 1-year delay in promotion from assistant to associate professor, compared with men. This delay could reduce female physicians’ earning potential by a 10-year median of $26,042 (or 2%), whereas failure to be promoted at all could decrease the 10-year earning potential by a median of $218,724 (or 13%).
Across medicine more broadly, male physicians continue to earn 35% more than their female colleagues, according to the 2021 Medscape Physician Compensation Report. The biggest differences in take-home pay exist between male and female specialists, per the report. On average, male physicians earn $376,000, while women’s take-home pay is $283,000.
Medical schools and hospital leaders have a role to play
The earning potential during the first 10 years of post-training employment by gender was the most dramatic in neurosurgery, orthopedic surgery, and cardiology, per the study. Three subspecialties where women and men have similar earning potential include pediatric nephrology, pediatric neurology, and pediatric rheumatology.
The coauthors note that it’s commonly understood that women don’t negotiate as often or as successfully as their male colleagues. A 2019 study in JAMA Surgery of 606 male and female surgery residents revealed that while residents of both genders shared similar career goals, women had lower future salary expectations and a significantly more negative view of the salary negotiation process.
Dr. Catenaccio and her coauthors acknowledge that negotiation skills and financial literacy should be taught during medical school and postgraduate training. “However, the onus for ensuring salary equity should not fall on the individual candidate alone; rather, departmental and hospital leadership should take responsibility to ensure uniform starting salaries and prevent gender-based inequalities,” they wrote in the study.
“We hope that this study encourages academic medical institutions to increase transparency and equity around compensation, particularly for junior faculty,” Dr. Catenaccio said in an interview. “This will require both ensuring equal starting salaries and providing periodic adjustments throughout individuals’ careers to prevent divergence in earning potential by gender or any other individual characteristics.”
Harold Simon, MD, MBA, vice chair for faculty for the department of pediatrics and professor of pediatrics and emergency medicine at Emory University, Atlanta, told this news organization that “[i]ncreased transparency around compensation can enable women to advocate for equitable pay. However, the burden for ensuring equity should not fall on individuals but instead must be the primary responsibility of academic institutions.”
Specifically, Dr. Simon advocates for hospital leaders to “ensure equity among providers including compensation [as] a crucial part of maintaining a diverse workforce and, ultimately, providing balanced access to health care for patients.”
In addition, the authors call for periodic compensation evaluations and adjustments to help prevent gender-based salary differences among female and male physicians in academia. “This is absolutely necessary, both to develop future compensation plans and to address any pre-existing gender-based salary inequities for those women currently well into their careers,” they wrote in the study.
Data analysis was conducted from March to May 2021. Researchers used models to estimate the impacts of promotion timing and potential interventions, which include equalizing starting salaries and annual salary rates.
The study included compensation data for 24,593 female and 29,886 male academic physicians across 45 subspecialties. It relied on publicly available data from the Association of American Medical Colleges’ annual Medical School Faculty Salary Survey report.
A version of this article first appeared on Medscape.com.
Worse still, the earning potential of women in most specialties is $214,440 (or 10%) less than their male colleagues over the course of the first 10 years of their careers in academic medicine.
Among the vast majority of subspecialties, women’s starting salaries and their salaries 10 years into their careers were lower than their male colleagues in academic medicine, per the study in JAMA Network Open.
Eva Catenaccio, MD, an epilepsy fellow at Children’s Hospital of Philadelphia and the lead author of the study, told this news organization that the gender disparities in earning potential are “pervasive in academic medicine.” These earnings disparities, which occur in nearly all subspecialties and can reach hundreds of thousands of dollars in the first 10 years of an academic physician’s career, “are largely the result of gender differences in annual salary that start immediately after training,” she said.
Changing the timing of academic promotion and equalizing starting salary and salary growth can help close the salary gap, said Dr. Catenaccio.
The study also reveals that women could face a 1-year delay in promotion from assistant to associate professor, compared with men. This delay could reduce female physicians’ earning potential by a 10-year median of $26,042 (or 2%), whereas failure to be promoted at all could decrease the 10-year earning potential by a median of $218,724 (or 13%).
Across medicine more broadly, male physicians continue to earn 35% more than their female colleagues, according to the 2021 Medscape Physician Compensation Report. The biggest differences in take-home pay exist between male and female specialists, per the report. On average, male physicians earn $376,000, while women’s take-home pay is $283,000.
Medical schools and hospital leaders have a role to play
The earning potential during the first 10 years of post-training employment by gender was the most dramatic in neurosurgery, orthopedic surgery, and cardiology, per the study. Three subspecialties where women and men have similar earning potential include pediatric nephrology, pediatric neurology, and pediatric rheumatology.
The coauthors note that it’s commonly understood that women don’t negotiate as often or as successfully as their male colleagues. A 2019 study in JAMA Surgery of 606 male and female surgery residents revealed that while residents of both genders shared similar career goals, women had lower future salary expectations and a significantly more negative view of the salary negotiation process.
Dr. Catenaccio and her coauthors acknowledge that negotiation skills and financial literacy should be taught during medical school and postgraduate training. “However, the onus for ensuring salary equity should not fall on the individual candidate alone; rather, departmental and hospital leadership should take responsibility to ensure uniform starting salaries and prevent gender-based inequalities,” they wrote in the study.
“We hope that this study encourages academic medical institutions to increase transparency and equity around compensation, particularly for junior faculty,” Dr. Catenaccio said in an interview. “This will require both ensuring equal starting salaries and providing periodic adjustments throughout individuals’ careers to prevent divergence in earning potential by gender or any other individual characteristics.”
Harold Simon, MD, MBA, vice chair for faculty for the department of pediatrics and professor of pediatrics and emergency medicine at Emory University, Atlanta, told this news organization that “[i]ncreased transparency around compensation can enable women to advocate for equitable pay. However, the burden for ensuring equity should not fall on individuals but instead must be the primary responsibility of academic institutions.”
Specifically, Dr. Simon advocates for hospital leaders to “ensure equity among providers including compensation [as] a crucial part of maintaining a diverse workforce and, ultimately, providing balanced access to health care for patients.”
In addition, the authors call for periodic compensation evaluations and adjustments to help prevent gender-based salary differences among female and male physicians in academia. “This is absolutely necessary, both to develop future compensation plans and to address any pre-existing gender-based salary inequities for those women currently well into their careers,” they wrote in the study.
Data analysis was conducted from March to May 2021. Researchers used models to estimate the impacts of promotion timing and potential interventions, which include equalizing starting salaries and annual salary rates.
The study included compensation data for 24,593 female and 29,886 male academic physicians across 45 subspecialties. It relied on publicly available data from the Association of American Medical Colleges’ annual Medical School Faculty Salary Survey report.
A version of this article first appeared on Medscape.com.





