User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
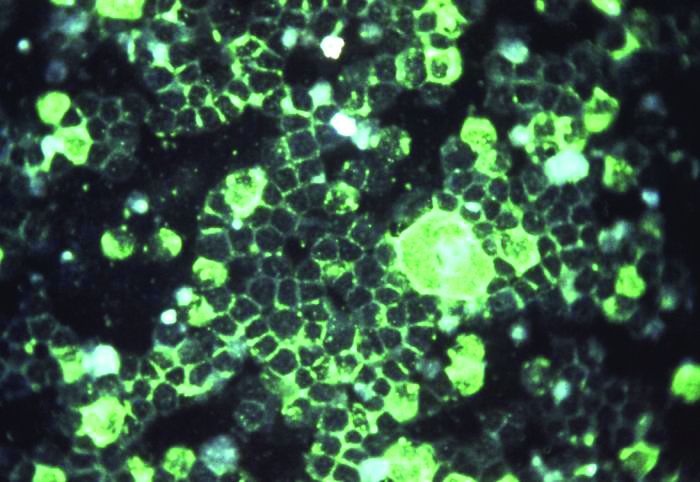
Getting closer to a lifesaving RSV vaccine
Louis Bont, MD, PhD, provided an overview of the most recent developments in the complex respiratory syncytial virus (RSV) vaccine landscape at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year.
RSV imposes significant burden worldwide, with 33 million patients, 3 million hospitalizations, and at least 120,000 deaths, reported Dr. Bont of the Wilhelmina Children’s Hospital, University Medical Centre, Utrecht, the Netherlands. Of those deaths, more than 50% are in infants younger than 5 months, and “about 99% of the children dying from RSV live in low- and middle-income countries.”
“There are high-risk populations, such as children with prematurity, congenital heart disease, lung disease, and Down syndrome, but about 73% of all children who are hospitalized for RSV infection were previously healthy children,” Dr. Bont explained. “So, we need to find a solution for all children to prevent RSV infection.”
As observed by Nienke Scheltema in a Lancet Global Health article, population distributions of RSV infection mortality show that, regardless of whether children have comorbidities or they are previously healthy, most children die at a very young age, Dr. Bont explained. These data suggest “that a maternal vaccine or an antibody prophylaxis approach from birth onwards or during the first RSV season is the solution for the problem.”
The path to developing an RSV vaccine has now narrowed its focus onto a structural element of RSV, the prefusion F protein. This shift started with the discovery by Jason McLellan (Science, 2013 [two papers]) that there are two variants of the RSV F-fusion protein: the very stable postfusion conformation and the prefusion active conformation, a metastable protein that exists for a “fraction of a second,” Dr. Bont said.
“The interesting thing is that epitopes that are visible at the prefusion, metastable state … induce highly neutralizing antibodies, whereas epitopes at the postfusion conformation do not,” Dr. Bont explained. “So, by stabilizing the prefusion state, we start inducing neutralizing antibodies that will protect against severe RSV infection, and this is the basic concept of all the vaccine developments currently ongoing.”
These RSV vaccine developments fall into five approach types: live-attenuated or chimeric vaccines, vector-based vaccines, monoclonal antibodies, particle-based vaccines, and subunit or protein-based vaccines.
One breakthrough, which was presented at last year’s ESPID meeting, is the monoclonal antibody nirsevimab. In addition to being nine times more potent than the broadly used antibody palivizumab, it is also more stable; whereas many antibodies have a half-life of 3 weeks, nirsevimab has a half-life of 100 days. “The idea is that a single injection at the start of the RSV season protects children in the first RSV season of their life, a dangerous episode for them.” Dr. Bont explained. The originators, AstraZeneca and Sanofi Pasteur, have “the vision that every child on this planet should receive a single injection with this antibody in the first season,” he explained.
Studies of nanoparticle-based maternal vaccines have also revealed interesting results: Although a phase 3 trial investigating such vaccines didn’t achieve its primary endpoint, “interestingly, 15% of all RSV infections were mild, and only 2% were very severe and leading to hypoxemia,” Dr. Bont noted. “But if we look at vaccine efficacy, we see the opposite – the vaccine was not very efficacious to prevent mild disease, but very efficacious to prevent severe hypoxemia; actually, this is exactly what you would like to see in a vaccine.”
Investigations into live-attenuated and vector-based vaccines have been promising as well, Dr. Bont shared. Studies of live-attenuated vaccines suggest they have a future and that we can move onto their next phase of clinical development, and a study investigating adenoviral vector-based vaccines has demonstrated safety, efficacy, and immunogenicity, though it has also shown that we should anticipate some side effects when using them.
Simple subunit vaccines for RSV are also being explored – a study of DS-Cav1, a stabilized prefusion F subunit protein candidate vaccine, has shown that it has a superior functional profile, compared with previous pre-F subunit vaccines. However, it seemed to be more efficacious against strains of RSV A than strains of RSV B, the dominant strain.
Dr. Bont also discussed exciting work by Sesterhenn et al., in which they used a computer-based program to develop their own vaccine. Using their in-depth knowledge of the RSV prefusion F protein and a computer program, Sesterhenn et al. developed a trivalent vaccine, produced it, and showed – both in vitro and in monkeys – that such vaccines can work up to the level of preclinical in vivo experiments.
“We can now make vaccines behind our computer,” Dr. Bont declared. “And the system doesn’t only work for RSV vaccines, but also for other pathogens – as long as you have an in-depth molecular knowledge of the target epitope,” he added.
Joanne Wildenbeest, MD, PhD, at the Utrecht University, the Netherlands commented: “Lower respiratory tract infections due to RSV are among the leading causes of death worldwide in children under the age of 5, especially young infants. The recent advances in the development of a vaccine and passive immunization are important steps towards the goal to reduce childhood mortality due to RSV worldwide. Since RSV-related mortality is mainly seen in developing countries it is important that, once a vaccine has been approved, it will also be made easily available to these countries.”
Dr. Bont reported the following disclosures: ReSViNET (a nonprofit foundation); investigator-initiated studies with the Bill & Melinda Gates Foundation, AbbVie, MedImmune, and MeMed; participation with Pfizer, Regeneron, and Janssen; and consultancy with GlaxoSmithKline, Ablynx, Novavax, and Janssen.
Louis Bont, MD, PhD, provided an overview of the most recent developments in the complex respiratory syncytial virus (RSV) vaccine landscape at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year.
RSV imposes significant burden worldwide, with 33 million patients, 3 million hospitalizations, and at least 120,000 deaths, reported Dr. Bont of the Wilhelmina Children’s Hospital, University Medical Centre, Utrecht, the Netherlands. Of those deaths, more than 50% are in infants younger than 5 months, and “about 99% of the children dying from RSV live in low- and middle-income countries.”
“There are high-risk populations, such as children with prematurity, congenital heart disease, lung disease, and Down syndrome, but about 73% of all children who are hospitalized for RSV infection were previously healthy children,” Dr. Bont explained. “So, we need to find a solution for all children to prevent RSV infection.”
As observed by Nienke Scheltema in a Lancet Global Health article, population distributions of RSV infection mortality show that, regardless of whether children have comorbidities or they are previously healthy, most children die at a very young age, Dr. Bont explained. These data suggest “that a maternal vaccine or an antibody prophylaxis approach from birth onwards or during the first RSV season is the solution for the problem.”
The path to developing an RSV vaccine has now narrowed its focus onto a structural element of RSV, the prefusion F protein. This shift started with the discovery by Jason McLellan (Science, 2013 [two papers]) that there are two variants of the RSV F-fusion protein: the very stable postfusion conformation and the prefusion active conformation, a metastable protein that exists for a “fraction of a second,” Dr. Bont said.
“The interesting thing is that epitopes that are visible at the prefusion, metastable state … induce highly neutralizing antibodies, whereas epitopes at the postfusion conformation do not,” Dr. Bont explained. “So, by stabilizing the prefusion state, we start inducing neutralizing antibodies that will protect against severe RSV infection, and this is the basic concept of all the vaccine developments currently ongoing.”
These RSV vaccine developments fall into five approach types: live-attenuated or chimeric vaccines, vector-based vaccines, monoclonal antibodies, particle-based vaccines, and subunit or protein-based vaccines.
One breakthrough, which was presented at last year’s ESPID meeting, is the monoclonal antibody nirsevimab. In addition to being nine times more potent than the broadly used antibody palivizumab, it is also more stable; whereas many antibodies have a half-life of 3 weeks, nirsevimab has a half-life of 100 days. “The idea is that a single injection at the start of the RSV season protects children in the first RSV season of their life, a dangerous episode for them.” Dr. Bont explained. The originators, AstraZeneca and Sanofi Pasteur, have “the vision that every child on this planet should receive a single injection with this antibody in the first season,” he explained.
Studies of nanoparticle-based maternal vaccines have also revealed interesting results: Although a phase 3 trial investigating such vaccines didn’t achieve its primary endpoint, “interestingly, 15% of all RSV infections were mild, and only 2% were very severe and leading to hypoxemia,” Dr. Bont noted. “But if we look at vaccine efficacy, we see the opposite – the vaccine was not very efficacious to prevent mild disease, but very efficacious to prevent severe hypoxemia; actually, this is exactly what you would like to see in a vaccine.”
Investigations into live-attenuated and vector-based vaccines have been promising as well, Dr. Bont shared. Studies of live-attenuated vaccines suggest they have a future and that we can move onto their next phase of clinical development, and a study investigating adenoviral vector-based vaccines has demonstrated safety, efficacy, and immunogenicity, though it has also shown that we should anticipate some side effects when using them.
Simple subunit vaccines for RSV are also being explored – a study of DS-Cav1, a stabilized prefusion F subunit protein candidate vaccine, has shown that it has a superior functional profile, compared with previous pre-F subunit vaccines. However, it seemed to be more efficacious against strains of RSV A than strains of RSV B, the dominant strain.
Dr. Bont also discussed exciting work by Sesterhenn et al., in which they used a computer-based program to develop their own vaccine. Using their in-depth knowledge of the RSV prefusion F protein and a computer program, Sesterhenn et al. developed a trivalent vaccine, produced it, and showed – both in vitro and in monkeys – that such vaccines can work up to the level of preclinical in vivo experiments.
“We can now make vaccines behind our computer,” Dr. Bont declared. “And the system doesn’t only work for RSV vaccines, but also for other pathogens – as long as you have an in-depth molecular knowledge of the target epitope,” he added.
Joanne Wildenbeest, MD, PhD, at the Utrecht University, the Netherlands commented: “Lower respiratory tract infections due to RSV are among the leading causes of death worldwide in children under the age of 5, especially young infants. The recent advances in the development of a vaccine and passive immunization are important steps towards the goal to reduce childhood mortality due to RSV worldwide. Since RSV-related mortality is mainly seen in developing countries it is important that, once a vaccine has been approved, it will also be made easily available to these countries.”
Dr. Bont reported the following disclosures: ReSViNET (a nonprofit foundation); investigator-initiated studies with the Bill & Melinda Gates Foundation, AbbVie, MedImmune, and MeMed; participation with Pfizer, Regeneron, and Janssen; and consultancy with GlaxoSmithKline, Ablynx, Novavax, and Janssen.
Louis Bont, MD, PhD, provided an overview of the most recent developments in the complex respiratory syncytial virus (RSV) vaccine landscape at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year.
RSV imposes significant burden worldwide, with 33 million patients, 3 million hospitalizations, and at least 120,000 deaths, reported Dr. Bont of the Wilhelmina Children’s Hospital, University Medical Centre, Utrecht, the Netherlands. Of those deaths, more than 50% are in infants younger than 5 months, and “about 99% of the children dying from RSV live in low- and middle-income countries.”
“There are high-risk populations, such as children with prematurity, congenital heart disease, lung disease, and Down syndrome, but about 73% of all children who are hospitalized for RSV infection were previously healthy children,” Dr. Bont explained. “So, we need to find a solution for all children to prevent RSV infection.”
As observed by Nienke Scheltema in a Lancet Global Health article, population distributions of RSV infection mortality show that, regardless of whether children have comorbidities or they are previously healthy, most children die at a very young age, Dr. Bont explained. These data suggest “that a maternal vaccine or an antibody prophylaxis approach from birth onwards or during the first RSV season is the solution for the problem.”
The path to developing an RSV vaccine has now narrowed its focus onto a structural element of RSV, the prefusion F protein. This shift started with the discovery by Jason McLellan (Science, 2013 [two papers]) that there are two variants of the RSV F-fusion protein: the very stable postfusion conformation and the prefusion active conformation, a metastable protein that exists for a “fraction of a second,” Dr. Bont said.
“The interesting thing is that epitopes that are visible at the prefusion, metastable state … induce highly neutralizing antibodies, whereas epitopes at the postfusion conformation do not,” Dr. Bont explained. “So, by stabilizing the prefusion state, we start inducing neutralizing antibodies that will protect against severe RSV infection, and this is the basic concept of all the vaccine developments currently ongoing.”
These RSV vaccine developments fall into five approach types: live-attenuated or chimeric vaccines, vector-based vaccines, monoclonal antibodies, particle-based vaccines, and subunit or protein-based vaccines.
One breakthrough, which was presented at last year’s ESPID meeting, is the monoclonal antibody nirsevimab. In addition to being nine times more potent than the broadly used antibody palivizumab, it is also more stable; whereas many antibodies have a half-life of 3 weeks, nirsevimab has a half-life of 100 days. “The idea is that a single injection at the start of the RSV season protects children in the first RSV season of their life, a dangerous episode for them.” Dr. Bont explained. The originators, AstraZeneca and Sanofi Pasteur, have “the vision that every child on this planet should receive a single injection with this antibody in the first season,” he explained.
Studies of nanoparticle-based maternal vaccines have also revealed interesting results: Although a phase 3 trial investigating such vaccines didn’t achieve its primary endpoint, “interestingly, 15% of all RSV infections were mild, and only 2% were very severe and leading to hypoxemia,” Dr. Bont noted. “But if we look at vaccine efficacy, we see the opposite – the vaccine was not very efficacious to prevent mild disease, but very efficacious to prevent severe hypoxemia; actually, this is exactly what you would like to see in a vaccine.”
Investigations into live-attenuated and vector-based vaccines have been promising as well, Dr. Bont shared. Studies of live-attenuated vaccines suggest they have a future and that we can move onto their next phase of clinical development, and a study investigating adenoviral vector-based vaccines has demonstrated safety, efficacy, and immunogenicity, though it has also shown that we should anticipate some side effects when using them.
Simple subunit vaccines for RSV are also being explored – a study of DS-Cav1, a stabilized prefusion F subunit protein candidate vaccine, has shown that it has a superior functional profile, compared with previous pre-F subunit vaccines. However, it seemed to be more efficacious against strains of RSV A than strains of RSV B, the dominant strain.
Dr. Bont also discussed exciting work by Sesterhenn et al., in which they used a computer-based program to develop their own vaccine. Using their in-depth knowledge of the RSV prefusion F protein and a computer program, Sesterhenn et al. developed a trivalent vaccine, produced it, and showed – both in vitro and in monkeys – that such vaccines can work up to the level of preclinical in vivo experiments.
“We can now make vaccines behind our computer,” Dr. Bont declared. “And the system doesn’t only work for RSV vaccines, but also for other pathogens – as long as you have an in-depth molecular knowledge of the target epitope,” he added.
Joanne Wildenbeest, MD, PhD, at the Utrecht University, the Netherlands commented: “Lower respiratory tract infections due to RSV are among the leading causes of death worldwide in children under the age of 5, especially young infants. The recent advances in the development of a vaccine and passive immunization are important steps towards the goal to reduce childhood mortality due to RSV worldwide. Since RSV-related mortality is mainly seen in developing countries it is important that, once a vaccine has been approved, it will also be made easily available to these countries.”
Dr. Bont reported the following disclosures: ReSViNET (a nonprofit foundation); investigator-initiated studies with the Bill & Melinda Gates Foundation, AbbVie, MedImmune, and MeMed; participation with Pfizer, Regeneron, and Janssen; and consultancy with GlaxoSmithKline, Ablynx, Novavax, and Janssen.
FROM ESPID 2020
Shared medical appointments may bridge the opioid treatment gap
Shared medical appointments (SMAs) are an acceptable way to receive treatment for opioid use disorder (OUD), new research suggests.
In a survey study, participants attending an urban outpatient buprenorphine clinic reported a high degree of satisfaction with SMAs. However, the majority also reported they preferred individual appointments.
Still, SMAs may serve a role in providing comprehensive care for certain subpopulations with OUD who are prone to isolation and may also increase capacity to treat more patients with a substance use disorder (SUD), said coinvestigator Serra Akyar, MD, Northwell Health Staten Island University Hospital, New York.
“By providing education and a forum for sharing, SMAs can lead to changes in behavior and enhance and reinforce coping and problem-solving skills,” Dr. Akyar said in an interview.
The findings were presented at the virtual American Academy of Addiction Psychiatry 31st Annual Meeting.
SMA vs. group therapy
SMA is not a form of group therapy, Dr. Akyar noted. Group therapy has a psychotherapy component and is led by a therapist. SMAs do not have a psychotherapeutic or a behavioral therapy component but provide education and an opportunity for sharing personal experiences of recovery.
“For example, the doctor participating in the group describes what happens in the brain to drive addiction and fellow participants share their personal anecdotes of recovery, including their struggles and successes,” Dr. Akyar said.
“ given the differences in the type of care each group provides,” she added.
Recent research on SMAs for OUD is limited. Although previous studies have shown that the practice is highly acceptable and has comparable or better retention in care rates with buprenorphine versus individual appointments, these studies have been conducted in predominantly White populations and in suburban settings.
For the new study, the investigators wanted to examine how acceptable SMAs for OUD would be in an urban setting involving predominantly racial and ethnic minorities.
They administered a 15-minute survey to patients with OUD who were attending the Comprehensive Addiction Resources and Education Center, an outpatient psychiatry clinic located at New Jersey Medical School, from December 2019 to February 2020.
Of the 42 participants who initially consented, 39 completed the survey. The majority of the responders were Black (64.1%), had an annual income that was less than $20,000 (61.5%), and/or were unemployed or disabled (69.3%).
Most of the participants agreed or strongly agreed with the following statements:
- Scheduling appointments for SMAs is easy.
- I gain valuable information from the responses to other patients’ questions in SMAs.
- There is enough time for questions during SMAs.
- I gain valuable information from the doctor and social worker in SMAs.
- My medical needs are met during SMAs.
- I would recommend an SMA to other patients.
- Since starting SMAs, I find it easier to stick to my treatment plan.
- I have a lot of support outside of SMAs.
- People in SMAs give me the support I need to stick to my treatment plan.
Interestingly, despite the overall high satisfaction with SMAs, just 33% of participants said they preferred them to one-on-one visits, Dr. Akyar noted.
Further analyses showed that total satisfaction scores were positively associated with older age, being on disability, or being in retirement.
Bridging the gap
In a comment, Philip Wong, MD, New Jersey Medical School, Newark, noted that a more widespread use of SMAs could potentially bridge the treatment gap that currently exists in the United States.
“For providers, SMAs help reduce costs, improve productivity, prevent repeating of common advice, and increase outreach. These are all important at a time when the need for OUD treatment is increasing. This is especially true for places like Newark, which is one of the prime epicenters of the opioid epidemic,” said Dr. Wong.
Although he was not involved with this research, he and his colleagues recently conducted a literature review of publications relating to SMAs and found seven peer-reviewed articles. However, none was appropriately designed to compare SMAs with traditional one-on-one recovery treatment.
“We definitely need more clinical studies to further our understanding of SMAs as a tool for the medication-assisted treatment of opioid use disorder,” Dr. Wong said.
“There are currently a very limited number of physicians who can prescribe medication-assisted treatment in the first place. So, if that one provider can reach a larger community by doing these SMAs, then the potential is very great in terms of addressing the opioid epidemic,” he said.
David Kan, MD, chief medical officer of Bright Heart Health, San Ramon, Calif., agreed.
“SMAs are promising because they are efficient and allow more people to access treatment,” Dr. Kan said in an interview.
“Although the mechanism of SMA satisfaction is unclear, other research shows peer support and groups helpful for SUD treatment as a whole. SMA takes the best of many worlds and increases the potential number of patients treated for SUD,” he said.
Also asked to comment, Lewei (Allison) Lin, MD, University of Michigan, Ann Arbor, said SMAs “are one of a number of important interventions that should be considered” in order to increase availability and access to medication providers for OUD.
However, more research is needed “to examine the impact on treatment uptake and patient and provider experiences,” said Dr. Lin.
Dr. Akyar, Dr. Wong, Dr. Kan, and Dr. Lin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Shared medical appointments (SMAs) are an acceptable way to receive treatment for opioid use disorder (OUD), new research suggests.
In a survey study, participants attending an urban outpatient buprenorphine clinic reported a high degree of satisfaction with SMAs. However, the majority also reported they preferred individual appointments.
Still, SMAs may serve a role in providing comprehensive care for certain subpopulations with OUD who are prone to isolation and may also increase capacity to treat more patients with a substance use disorder (SUD), said coinvestigator Serra Akyar, MD, Northwell Health Staten Island University Hospital, New York.
“By providing education and a forum for sharing, SMAs can lead to changes in behavior and enhance and reinforce coping and problem-solving skills,” Dr. Akyar said in an interview.
The findings were presented at the virtual American Academy of Addiction Psychiatry 31st Annual Meeting.
SMA vs. group therapy
SMA is not a form of group therapy, Dr. Akyar noted. Group therapy has a psychotherapy component and is led by a therapist. SMAs do not have a psychotherapeutic or a behavioral therapy component but provide education and an opportunity for sharing personal experiences of recovery.
“For example, the doctor participating in the group describes what happens in the brain to drive addiction and fellow participants share their personal anecdotes of recovery, including their struggles and successes,” Dr. Akyar said.
“ given the differences in the type of care each group provides,” she added.
Recent research on SMAs for OUD is limited. Although previous studies have shown that the practice is highly acceptable and has comparable or better retention in care rates with buprenorphine versus individual appointments, these studies have been conducted in predominantly White populations and in suburban settings.
For the new study, the investigators wanted to examine how acceptable SMAs for OUD would be in an urban setting involving predominantly racial and ethnic minorities.
They administered a 15-minute survey to patients with OUD who were attending the Comprehensive Addiction Resources and Education Center, an outpatient psychiatry clinic located at New Jersey Medical School, from December 2019 to February 2020.
Of the 42 participants who initially consented, 39 completed the survey. The majority of the responders were Black (64.1%), had an annual income that was less than $20,000 (61.5%), and/or were unemployed or disabled (69.3%).
Most of the participants agreed or strongly agreed with the following statements:
- Scheduling appointments for SMAs is easy.
- I gain valuable information from the responses to other patients’ questions in SMAs.
- There is enough time for questions during SMAs.
- I gain valuable information from the doctor and social worker in SMAs.
- My medical needs are met during SMAs.
- I would recommend an SMA to other patients.
- Since starting SMAs, I find it easier to stick to my treatment plan.
- I have a lot of support outside of SMAs.
- People in SMAs give me the support I need to stick to my treatment plan.
Interestingly, despite the overall high satisfaction with SMAs, just 33% of participants said they preferred them to one-on-one visits, Dr. Akyar noted.
Further analyses showed that total satisfaction scores were positively associated with older age, being on disability, or being in retirement.
Bridging the gap
In a comment, Philip Wong, MD, New Jersey Medical School, Newark, noted that a more widespread use of SMAs could potentially bridge the treatment gap that currently exists in the United States.
“For providers, SMAs help reduce costs, improve productivity, prevent repeating of common advice, and increase outreach. These are all important at a time when the need for OUD treatment is increasing. This is especially true for places like Newark, which is one of the prime epicenters of the opioid epidemic,” said Dr. Wong.
Although he was not involved with this research, he and his colleagues recently conducted a literature review of publications relating to SMAs and found seven peer-reviewed articles. However, none was appropriately designed to compare SMAs with traditional one-on-one recovery treatment.
“We definitely need more clinical studies to further our understanding of SMAs as a tool for the medication-assisted treatment of opioid use disorder,” Dr. Wong said.
“There are currently a very limited number of physicians who can prescribe medication-assisted treatment in the first place. So, if that one provider can reach a larger community by doing these SMAs, then the potential is very great in terms of addressing the opioid epidemic,” he said.
David Kan, MD, chief medical officer of Bright Heart Health, San Ramon, Calif., agreed.
“SMAs are promising because they are efficient and allow more people to access treatment,” Dr. Kan said in an interview.
“Although the mechanism of SMA satisfaction is unclear, other research shows peer support and groups helpful for SUD treatment as a whole. SMA takes the best of many worlds and increases the potential number of patients treated for SUD,” he said.
Also asked to comment, Lewei (Allison) Lin, MD, University of Michigan, Ann Arbor, said SMAs “are one of a number of important interventions that should be considered” in order to increase availability and access to medication providers for OUD.
However, more research is needed “to examine the impact on treatment uptake and patient and provider experiences,” said Dr. Lin.
Dr. Akyar, Dr. Wong, Dr. Kan, and Dr. Lin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Shared medical appointments (SMAs) are an acceptable way to receive treatment for opioid use disorder (OUD), new research suggests.
In a survey study, participants attending an urban outpatient buprenorphine clinic reported a high degree of satisfaction with SMAs. However, the majority also reported they preferred individual appointments.
Still, SMAs may serve a role in providing comprehensive care for certain subpopulations with OUD who are prone to isolation and may also increase capacity to treat more patients with a substance use disorder (SUD), said coinvestigator Serra Akyar, MD, Northwell Health Staten Island University Hospital, New York.
“By providing education and a forum for sharing, SMAs can lead to changes in behavior and enhance and reinforce coping and problem-solving skills,” Dr. Akyar said in an interview.
The findings were presented at the virtual American Academy of Addiction Psychiatry 31st Annual Meeting.
SMA vs. group therapy
SMA is not a form of group therapy, Dr. Akyar noted. Group therapy has a psychotherapy component and is led by a therapist. SMAs do not have a psychotherapeutic or a behavioral therapy component but provide education and an opportunity for sharing personal experiences of recovery.
“For example, the doctor participating in the group describes what happens in the brain to drive addiction and fellow participants share their personal anecdotes of recovery, including their struggles and successes,” Dr. Akyar said.
“ given the differences in the type of care each group provides,” she added.
Recent research on SMAs for OUD is limited. Although previous studies have shown that the practice is highly acceptable and has comparable or better retention in care rates with buprenorphine versus individual appointments, these studies have been conducted in predominantly White populations and in suburban settings.
For the new study, the investigators wanted to examine how acceptable SMAs for OUD would be in an urban setting involving predominantly racial and ethnic minorities.
They administered a 15-minute survey to patients with OUD who were attending the Comprehensive Addiction Resources and Education Center, an outpatient psychiatry clinic located at New Jersey Medical School, from December 2019 to February 2020.
Of the 42 participants who initially consented, 39 completed the survey. The majority of the responders were Black (64.1%), had an annual income that was less than $20,000 (61.5%), and/or were unemployed or disabled (69.3%).
Most of the participants agreed or strongly agreed with the following statements:
- Scheduling appointments for SMAs is easy.
- I gain valuable information from the responses to other patients’ questions in SMAs.
- There is enough time for questions during SMAs.
- I gain valuable information from the doctor and social worker in SMAs.
- My medical needs are met during SMAs.
- I would recommend an SMA to other patients.
- Since starting SMAs, I find it easier to stick to my treatment plan.
- I have a lot of support outside of SMAs.
- People in SMAs give me the support I need to stick to my treatment plan.
Interestingly, despite the overall high satisfaction with SMAs, just 33% of participants said they preferred them to one-on-one visits, Dr. Akyar noted.
Further analyses showed that total satisfaction scores were positively associated with older age, being on disability, or being in retirement.
Bridging the gap
In a comment, Philip Wong, MD, New Jersey Medical School, Newark, noted that a more widespread use of SMAs could potentially bridge the treatment gap that currently exists in the United States.
“For providers, SMAs help reduce costs, improve productivity, prevent repeating of common advice, and increase outreach. These are all important at a time when the need for OUD treatment is increasing. This is especially true for places like Newark, which is one of the prime epicenters of the opioid epidemic,” said Dr. Wong.
Although he was not involved with this research, he and his colleagues recently conducted a literature review of publications relating to SMAs and found seven peer-reviewed articles. However, none was appropriately designed to compare SMAs with traditional one-on-one recovery treatment.
“We definitely need more clinical studies to further our understanding of SMAs as a tool for the medication-assisted treatment of opioid use disorder,” Dr. Wong said.
“There are currently a very limited number of physicians who can prescribe medication-assisted treatment in the first place. So, if that one provider can reach a larger community by doing these SMAs, then the potential is very great in terms of addressing the opioid epidemic,” he said.
David Kan, MD, chief medical officer of Bright Heart Health, San Ramon, Calif., agreed.
“SMAs are promising because they are efficient and allow more people to access treatment,” Dr. Kan said in an interview.
“Although the mechanism of SMA satisfaction is unclear, other research shows peer support and groups helpful for SUD treatment as a whole. SMA takes the best of many worlds and increases the potential number of patients treated for SUD,” he said.
Also asked to comment, Lewei (Allison) Lin, MD, University of Michigan, Ann Arbor, said SMAs “are one of a number of important interventions that should be considered” in order to increase availability and access to medication providers for OUD.
However, more research is needed “to examine the impact on treatment uptake and patient and provider experiences,” said Dr. Lin.
Dr. Akyar, Dr. Wong, Dr. Kan, and Dr. Lin disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Hospital volumes start to fall again, even as COVID-19 soars
Hospital volumes, which had largely recovered in September after crashing last spring, are dropping again, according to new data from Strata Decision Technologies, a Chicago-based analytics firm.
For the 2 weeks that ended Nov. 28, inpatient admissions were 6.2% below what they’d been on Nov. 14 and 2.1% below what they’d been on Oct. 28. Compared with the same intervals in 2019, admissions were off 4.4% for the 14-day period and 3.7% for the 30-day period.
Although those aren’t large percentages, Strata’s report, based on data from about 275 client hospitals, notes that what kept the volumes up was the increasing number of COVID-19 cases. If COVID-19 cases are not considered, admissions would have been down “double digits,” said Steve Lefar, executive director of StrataDataScience, a division of Strata Decision Technologies, in an interview with this news organization.
“Hip and knee replacements, cardiac procedures, and other procedures are significantly down year over year. Infectious disease cases, in contrast, have skyrocketed,” Mr. Lefar said. “Many things went way down that hadn’t fully recovered. It’s COVID-19 that really brought the volume back up.”
Observation and emergency department visits also dropped from already low levels. For the 2 weeks that ended Nov. 28, observation visits were off 8.4%; for the previous month, 10.1%. Compared with 2019, they were down 22.3% and 18.6%, respectively.
ED visits fell 3.7% for the 2-week period, 0.6% for the month. They dropped 21% and 18.7%, respectively, compared with those periods from the previous year.
What these data reflect, Mr. Lefar said, is that people have avoided EDs and are staying away from them more than ever because of COVID-19 fears. This behavior could be problematic for people who have concerning symptoms, such as chest pains, that should be evaluated by an ED physician, he noted.
Daily outpatient visits were down 18.4% for the 14-day period and 9.3% for the 30-day period. But, compared with 2019, ambulatory visits increased 5.8% for the 2-week period and 4.7% for the previous month.
Long-term trends
The outpatient visit data should be viewed in the context of the overall trend since the pandemic began. Strata broke down service lines for the period between March 20 and Nov. 7. The analysis shows that evaluation and management (E/M) encounters, the largest outpatient visit category, fell 58% during this period, compared with the same interval in 2019. Visits for diabetes, hypertension, and minor acute infections and injuries were also way down.
Mr. Lefar observed that the E/M visit category was only for in-person visits, which many patients have ditched in favor of telehealth encounters. At the same time, he noted, “people are going in less for chronic disease visits. So there’s an interplay between less in-person visits, more telehealth, and maybe people going to other sites that aren’t on the hospital campus. But people are going less [to outpatient clinics].”
In the year-to-year comparison, volume was down substantially in other service lines, including cancer (–9.2%), cardiology (–20%), dermatology (–31%), endocrine (–18.8%), ENT (–42.5%), gastroenterology (–24.3%), nephrology (–15%), obstetrics (–15.6%), orthopedics (–28.2%), and general surgery (–22.2%). Major procedures decreased by 21.8%.
In contrast, the infectious disease category jumped 86% over 2019, and “other infectious and parasitic diseases” – i.e., COVID-19 – soared 222%.
There was a much bigger crash in admissions, observation visits, and ED visits last spring than in November, the report shows. “What happened nationally last spring is that everyone shut down,” Mr. Lefar explained. “All the electives were canceled. Even cancer surgery was shut down, along with many other procedures. That’s what drove that crash. But the provider community quickly learned that this is going to be a long haul, and we’re going to have to reopen. We’re going to do it safely, but we’re going to make sure people get the necessary care. We can’t put off cancer care or colonoscopies and other screenings that save lives.”
System starts to break down
The current wave of COVID-19, however, is beginning to change the definition of necessary care, he said. “Hospitals are reaching the breaking point between staff exhaustion and hospital capacity reaching its limit. In Texas, hospitals are starting to shut down certain essential non-COVID care. They’re turning away some nonurgent cases – the electives that were starting to come back.”
How about nonurgent COVID cases? Mr. Lefar said there’s evidence that some of those patients are also being diverted. “Some experts speculate that the turn-away rate of people with confirmed COVID is starting to go up, and hospitals are sending them home with oxygen or an oxygen meter and saying, ‘If it gets worse, come back.’ They just don’t have the critical care capacity – and that should scare the heck out of everybody.”
Strata doesn’t yet have the data to confirm this, he said, “but it appears that some people are being sent home. This may be partly because providers are better at telling which patients are acute, and there are better things they can send them home with. It’s not necessarily worse care, but we don’t know. But we’re definitely seeing a higher send-home rate of patients showing up with COVID.”
Hospital profit margins are cratering again, because the COVID-19 cases aren’t generating nearly as much profit as the lucrative procedures that, in many cases, have been put off, Mr. Lefar said. “Even though CMS is paying 20% more for verified COVID-19 patients, we know that the costs on these patients are much higher than expected, so they’re not making much money on these cases.”
For about a third of hospitals, margins are currently negative, he said. That is about the same percentage as in September. In April, 60% of health systems were losing money, he added. “The CARES Act saved some of them,” he noted.
A version of this article first appeared on Medscape.com.
Hospital volumes, which had largely recovered in September after crashing last spring, are dropping again, according to new data from Strata Decision Technologies, a Chicago-based analytics firm.
For the 2 weeks that ended Nov. 28, inpatient admissions were 6.2% below what they’d been on Nov. 14 and 2.1% below what they’d been on Oct. 28. Compared with the same intervals in 2019, admissions were off 4.4% for the 14-day period and 3.7% for the 30-day period.
Although those aren’t large percentages, Strata’s report, based on data from about 275 client hospitals, notes that what kept the volumes up was the increasing number of COVID-19 cases. If COVID-19 cases are not considered, admissions would have been down “double digits,” said Steve Lefar, executive director of StrataDataScience, a division of Strata Decision Technologies, in an interview with this news organization.
“Hip and knee replacements, cardiac procedures, and other procedures are significantly down year over year. Infectious disease cases, in contrast, have skyrocketed,” Mr. Lefar said. “Many things went way down that hadn’t fully recovered. It’s COVID-19 that really brought the volume back up.”
Observation and emergency department visits also dropped from already low levels. For the 2 weeks that ended Nov. 28, observation visits were off 8.4%; for the previous month, 10.1%. Compared with 2019, they were down 22.3% and 18.6%, respectively.
ED visits fell 3.7% for the 2-week period, 0.6% for the month. They dropped 21% and 18.7%, respectively, compared with those periods from the previous year.
What these data reflect, Mr. Lefar said, is that people have avoided EDs and are staying away from them more than ever because of COVID-19 fears. This behavior could be problematic for people who have concerning symptoms, such as chest pains, that should be evaluated by an ED physician, he noted.
Daily outpatient visits were down 18.4% for the 14-day period and 9.3% for the 30-day period. But, compared with 2019, ambulatory visits increased 5.8% for the 2-week period and 4.7% for the previous month.
Long-term trends
The outpatient visit data should be viewed in the context of the overall trend since the pandemic began. Strata broke down service lines for the period between March 20 and Nov. 7. The analysis shows that evaluation and management (E/M) encounters, the largest outpatient visit category, fell 58% during this period, compared with the same interval in 2019. Visits for diabetes, hypertension, and minor acute infections and injuries were also way down.
Mr. Lefar observed that the E/M visit category was only for in-person visits, which many patients have ditched in favor of telehealth encounters. At the same time, he noted, “people are going in less for chronic disease visits. So there’s an interplay between less in-person visits, more telehealth, and maybe people going to other sites that aren’t on the hospital campus. But people are going less [to outpatient clinics].”
In the year-to-year comparison, volume was down substantially in other service lines, including cancer (–9.2%), cardiology (–20%), dermatology (–31%), endocrine (–18.8%), ENT (–42.5%), gastroenterology (–24.3%), nephrology (–15%), obstetrics (–15.6%), orthopedics (–28.2%), and general surgery (–22.2%). Major procedures decreased by 21.8%.
In contrast, the infectious disease category jumped 86% over 2019, and “other infectious and parasitic diseases” – i.e., COVID-19 – soared 222%.
There was a much bigger crash in admissions, observation visits, and ED visits last spring than in November, the report shows. “What happened nationally last spring is that everyone shut down,” Mr. Lefar explained. “All the electives were canceled. Even cancer surgery was shut down, along with many other procedures. That’s what drove that crash. But the provider community quickly learned that this is going to be a long haul, and we’re going to have to reopen. We’re going to do it safely, but we’re going to make sure people get the necessary care. We can’t put off cancer care or colonoscopies and other screenings that save lives.”
System starts to break down
The current wave of COVID-19, however, is beginning to change the definition of necessary care, he said. “Hospitals are reaching the breaking point between staff exhaustion and hospital capacity reaching its limit. In Texas, hospitals are starting to shut down certain essential non-COVID care. They’re turning away some nonurgent cases – the electives that were starting to come back.”
How about nonurgent COVID cases? Mr. Lefar said there’s evidence that some of those patients are also being diverted. “Some experts speculate that the turn-away rate of people with confirmed COVID is starting to go up, and hospitals are sending them home with oxygen or an oxygen meter and saying, ‘If it gets worse, come back.’ They just don’t have the critical care capacity – and that should scare the heck out of everybody.”
Strata doesn’t yet have the data to confirm this, he said, “but it appears that some people are being sent home. This may be partly because providers are better at telling which patients are acute, and there are better things they can send them home with. It’s not necessarily worse care, but we don’t know. But we’re definitely seeing a higher send-home rate of patients showing up with COVID.”
Hospital profit margins are cratering again, because the COVID-19 cases aren’t generating nearly as much profit as the lucrative procedures that, in many cases, have been put off, Mr. Lefar said. “Even though CMS is paying 20% more for verified COVID-19 patients, we know that the costs on these patients are much higher than expected, so they’re not making much money on these cases.”
For about a third of hospitals, margins are currently negative, he said. That is about the same percentage as in September. In April, 60% of health systems were losing money, he added. “The CARES Act saved some of them,” he noted.
A version of this article first appeared on Medscape.com.
Hospital volumes, which had largely recovered in September after crashing last spring, are dropping again, according to new data from Strata Decision Technologies, a Chicago-based analytics firm.
For the 2 weeks that ended Nov. 28, inpatient admissions were 6.2% below what they’d been on Nov. 14 and 2.1% below what they’d been on Oct. 28. Compared with the same intervals in 2019, admissions were off 4.4% for the 14-day period and 3.7% for the 30-day period.
Although those aren’t large percentages, Strata’s report, based on data from about 275 client hospitals, notes that what kept the volumes up was the increasing number of COVID-19 cases. If COVID-19 cases are not considered, admissions would have been down “double digits,” said Steve Lefar, executive director of StrataDataScience, a division of Strata Decision Technologies, in an interview with this news organization.
“Hip and knee replacements, cardiac procedures, and other procedures are significantly down year over year. Infectious disease cases, in contrast, have skyrocketed,” Mr. Lefar said. “Many things went way down that hadn’t fully recovered. It’s COVID-19 that really brought the volume back up.”
Observation and emergency department visits also dropped from already low levels. For the 2 weeks that ended Nov. 28, observation visits were off 8.4%; for the previous month, 10.1%. Compared with 2019, they were down 22.3% and 18.6%, respectively.
ED visits fell 3.7% for the 2-week period, 0.6% for the month. They dropped 21% and 18.7%, respectively, compared with those periods from the previous year.
What these data reflect, Mr. Lefar said, is that people have avoided EDs and are staying away from them more than ever because of COVID-19 fears. This behavior could be problematic for people who have concerning symptoms, such as chest pains, that should be evaluated by an ED physician, he noted.
Daily outpatient visits were down 18.4% for the 14-day period and 9.3% for the 30-day period. But, compared with 2019, ambulatory visits increased 5.8% for the 2-week period and 4.7% for the previous month.
Long-term trends
The outpatient visit data should be viewed in the context of the overall trend since the pandemic began. Strata broke down service lines for the period between March 20 and Nov. 7. The analysis shows that evaluation and management (E/M) encounters, the largest outpatient visit category, fell 58% during this period, compared with the same interval in 2019. Visits for diabetes, hypertension, and minor acute infections and injuries were also way down.
Mr. Lefar observed that the E/M visit category was only for in-person visits, which many patients have ditched in favor of telehealth encounters. At the same time, he noted, “people are going in less for chronic disease visits. So there’s an interplay between less in-person visits, more telehealth, and maybe people going to other sites that aren’t on the hospital campus. But people are going less [to outpatient clinics].”
In the year-to-year comparison, volume was down substantially in other service lines, including cancer (–9.2%), cardiology (–20%), dermatology (–31%), endocrine (–18.8%), ENT (–42.5%), gastroenterology (–24.3%), nephrology (–15%), obstetrics (–15.6%), orthopedics (–28.2%), and general surgery (–22.2%). Major procedures decreased by 21.8%.
In contrast, the infectious disease category jumped 86% over 2019, and “other infectious and parasitic diseases” – i.e., COVID-19 – soared 222%.
There was a much bigger crash in admissions, observation visits, and ED visits last spring than in November, the report shows. “What happened nationally last spring is that everyone shut down,” Mr. Lefar explained. “All the electives were canceled. Even cancer surgery was shut down, along with many other procedures. That’s what drove that crash. But the provider community quickly learned that this is going to be a long haul, and we’re going to have to reopen. We’re going to do it safely, but we’re going to make sure people get the necessary care. We can’t put off cancer care or colonoscopies and other screenings that save lives.”
System starts to break down
The current wave of COVID-19, however, is beginning to change the definition of necessary care, he said. “Hospitals are reaching the breaking point between staff exhaustion and hospital capacity reaching its limit. In Texas, hospitals are starting to shut down certain essential non-COVID care. They’re turning away some nonurgent cases – the electives that were starting to come back.”
How about nonurgent COVID cases? Mr. Lefar said there’s evidence that some of those patients are also being diverted. “Some experts speculate that the turn-away rate of people with confirmed COVID is starting to go up, and hospitals are sending them home with oxygen or an oxygen meter and saying, ‘If it gets worse, come back.’ They just don’t have the critical care capacity – and that should scare the heck out of everybody.”
Strata doesn’t yet have the data to confirm this, he said, “but it appears that some people are being sent home. This may be partly because providers are better at telling which patients are acute, and there are better things they can send them home with. It’s not necessarily worse care, but we don’t know. But we’re definitely seeing a higher send-home rate of patients showing up with COVID.”
Hospital profit margins are cratering again, because the COVID-19 cases aren’t generating nearly as much profit as the lucrative procedures that, in many cases, have been put off, Mr. Lefar said. “Even though CMS is paying 20% more for verified COVID-19 patients, we know that the costs on these patients are much higher than expected, so they’re not making much money on these cases.”
For about a third of hospitals, margins are currently negative, he said. That is about the same percentage as in September. In April, 60% of health systems were losing money, he added. “The CARES Act saved some of them,” he noted.
A version of this article first appeared on Medscape.com.
Pregnant women should be offered COVID-19 vaccine, experts agree
according to guidance from the Centers for Disease Control and Prevention, the American College of Obstetricians and Gynecologists, and the Society for Maternal-Fetal Medicine.
Pregnant women who opt not to receive the vaccine should be supported in that decision as well, a practice advisory from ACOG recommends.
“Pregnant women who experience fever following vaccination should be counseled to take acetaminophen,” the advisory notes.
In addition, women do not need to avoid pregnancy after receiving the Pfizer-BioNTech COVID-19 vaccine, according to the CDC’s interim clinical considerations for its use. The U.S. Food and Drug Administration issued an emergency use authorization for the vaccine on Dec. 11.
Although investigators excluded pregnant women from clinical trials, experts believe that mRNA vaccines, which are not live vaccines, “are unlikely to pose a risk for people who are pregnant” and “are not thought to be a risk to the breastfeeding infant,” the CDC notes.
Meanwhile, women who are pregnant may be at greater risk of severe COVID-19, even though the absolute risk of severe illness is low. COVID-19 also may increase the risk of adverse pregnancy outcomes, such as preterm birth, although the data have been mixed with some studies finding an association and others not.
“If pregnant people are part of a group that is recommended to receive a COVID-19 vaccine (e.g., health care personnel), they may choose to be vaccinated,” the CDC advises. “A conversation between the patient and their clinical team may assist with decisions regarding the use of vaccines approved under EUA for the prevention of COVID-19. While a conversation with a health care provider may be helpful, it is not required prior to vaccination.”
Acknowledging side effects and uncertainty
ACOG’s advisory reiterates that approach. The group notes that, based on the mRNA vaccine’s mechanism of action and its safety and efficacy in clinical trials, “it is expected that the safety and efficacy profile of the vaccine for pregnant individuals would be similar to that observed in nonpregnant individuals ... That said, there are no safety data specific to mRNA vaccine use in pregnant or lactating individuals and the potential risks to a pregnant individual and the fetus are unknown.”
In clinical trials, most participants experienced mild influenza-like symptoms following vaccination, including injection site reactions, fatigue, chills, muscle and joint pain, and headache. Among participants aged 18-55 years, fever greater than 38°C occurred in 3.7% of participants after the first dose and in 15.8% after the second dose. Most symptoms resolved within a few days.
Women who are pregnant should treat fever with acetaminophen because “fever has been associated with adverse pregnancy outcomes,” according to the ACOG guidance. “Acetaminophen has been proven to be safe for use in pregnancy and does not appear to impact antibody response to COVID-19 vaccines.” Patients may treat other vaccine side effects, such as injection-site soreness with acetaminophen as well.
When counseling patients, clinicians should explain that side effects are a normal part of developing antibodies to protect against COVID-19. “Regardless of their decision,” the group says, “these conversations provide an opportunity to remind patients about the importance of other prevention measures such as hand washing, physical distancing, and wearing a mask.”
More data expected
Data from developmental and reproductive toxicity studies in animals are expected soon, the CDC said. In addition, the manufacturer is following clinical trial participants who became pregnant during the study.
Women who are pregnant and their physicians should weigh factors such as the extent of COVID-19 transmission in the community, the patient’s risk of contracting COVID-19, risks of COVID-19 to the patient and fetus, vaccine efficacy and side effects, and the lack of data about COVID-19 vaccination during pregnancy.
The Society for Maternal-Fetal Medicine recommends that pregnant and lactating women have access to COVID-19 vaccines in general and has advocated for the inclusion of women who are pregnant or lactating in vaccine trials. The society has suggested that health care professionals “counsel their patients that the theoretical risk of fetal harm from mRNA vaccines is very low.” It published resources this week for physicians and patients focused on COVID-19 vaccination and pregnancy.
In a review published online Dec. 10 in the American Journal of Obstetrics & Gynecology MFM, Amanda M. Craig, MD, of Duke University Health System in Durham, N.C., and coauthors note that there “is a theoretical risk for fetal harm from any untested medical intervention and this is no different for COVID-19 vaccines.”
“Pregnant individuals should be given the opportunity, along with their obstetric provider, to weigh the potential risk of severe maternal disease against the unknown risk of fetal exposure, and make an autonomous decision about whether or not to accept vaccine until pregnancy safety data are available,” they write.
A version of this article first appeared on Medscape.com.
according to guidance from the Centers for Disease Control and Prevention, the American College of Obstetricians and Gynecologists, and the Society for Maternal-Fetal Medicine.
Pregnant women who opt not to receive the vaccine should be supported in that decision as well, a practice advisory from ACOG recommends.
“Pregnant women who experience fever following vaccination should be counseled to take acetaminophen,” the advisory notes.
In addition, women do not need to avoid pregnancy after receiving the Pfizer-BioNTech COVID-19 vaccine, according to the CDC’s interim clinical considerations for its use. The U.S. Food and Drug Administration issued an emergency use authorization for the vaccine on Dec. 11.
Although investigators excluded pregnant women from clinical trials, experts believe that mRNA vaccines, which are not live vaccines, “are unlikely to pose a risk for people who are pregnant” and “are not thought to be a risk to the breastfeeding infant,” the CDC notes.
Meanwhile, women who are pregnant may be at greater risk of severe COVID-19, even though the absolute risk of severe illness is low. COVID-19 also may increase the risk of adverse pregnancy outcomes, such as preterm birth, although the data have been mixed with some studies finding an association and others not.
“If pregnant people are part of a group that is recommended to receive a COVID-19 vaccine (e.g., health care personnel), they may choose to be vaccinated,” the CDC advises. “A conversation between the patient and their clinical team may assist with decisions regarding the use of vaccines approved under EUA for the prevention of COVID-19. While a conversation with a health care provider may be helpful, it is not required prior to vaccination.”
Acknowledging side effects and uncertainty
ACOG’s advisory reiterates that approach. The group notes that, based on the mRNA vaccine’s mechanism of action and its safety and efficacy in clinical trials, “it is expected that the safety and efficacy profile of the vaccine for pregnant individuals would be similar to that observed in nonpregnant individuals ... That said, there are no safety data specific to mRNA vaccine use in pregnant or lactating individuals and the potential risks to a pregnant individual and the fetus are unknown.”
In clinical trials, most participants experienced mild influenza-like symptoms following vaccination, including injection site reactions, fatigue, chills, muscle and joint pain, and headache. Among participants aged 18-55 years, fever greater than 38°C occurred in 3.7% of participants after the first dose and in 15.8% after the second dose. Most symptoms resolved within a few days.
Women who are pregnant should treat fever with acetaminophen because “fever has been associated with adverse pregnancy outcomes,” according to the ACOG guidance. “Acetaminophen has been proven to be safe for use in pregnancy and does not appear to impact antibody response to COVID-19 vaccines.” Patients may treat other vaccine side effects, such as injection-site soreness with acetaminophen as well.
When counseling patients, clinicians should explain that side effects are a normal part of developing antibodies to protect against COVID-19. “Regardless of their decision,” the group says, “these conversations provide an opportunity to remind patients about the importance of other prevention measures such as hand washing, physical distancing, and wearing a mask.”
More data expected
Data from developmental and reproductive toxicity studies in animals are expected soon, the CDC said. In addition, the manufacturer is following clinical trial participants who became pregnant during the study.
Women who are pregnant and their physicians should weigh factors such as the extent of COVID-19 transmission in the community, the patient’s risk of contracting COVID-19, risks of COVID-19 to the patient and fetus, vaccine efficacy and side effects, and the lack of data about COVID-19 vaccination during pregnancy.
The Society for Maternal-Fetal Medicine recommends that pregnant and lactating women have access to COVID-19 vaccines in general and has advocated for the inclusion of women who are pregnant or lactating in vaccine trials. The society has suggested that health care professionals “counsel their patients that the theoretical risk of fetal harm from mRNA vaccines is very low.” It published resources this week for physicians and patients focused on COVID-19 vaccination and pregnancy.
In a review published online Dec. 10 in the American Journal of Obstetrics & Gynecology MFM, Amanda M. Craig, MD, of Duke University Health System in Durham, N.C., and coauthors note that there “is a theoretical risk for fetal harm from any untested medical intervention and this is no different for COVID-19 vaccines.”
“Pregnant individuals should be given the opportunity, along with their obstetric provider, to weigh the potential risk of severe maternal disease against the unknown risk of fetal exposure, and make an autonomous decision about whether or not to accept vaccine until pregnancy safety data are available,” they write.
A version of this article first appeared on Medscape.com.
according to guidance from the Centers for Disease Control and Prevention, the American College of Obstetricians and Gynecologists, and the Society for Maternal-Fetal Medicine.
Pregnant women who opt not to receive the vaccine should be supported in that decision as well, a practice advisory from ACOG recommends.
“Pregnant women who experience fever following vaccination should be counseled to take acetaminophen,” the advisory notes.
In addition, women do not need to avoid pregnancy after receiving the Pfizer-BioNTech COVID-19 vaccine, according to the CDC’s interim clinical considerations for its use. The U.S. Food and Drug Administration issued an emergency use authorization for the vaccine on Dec. 11.
Although investigators excluded pregnant women from clinical trials, experts believe that mRNA vaccines, which are not live vaccines, “are unlikely to pose a risk for people who are pregnant” and “are not thought to be a risk to the breastfeeding infant,” the CDC notes.
Meanwhile, women who are pregnant may be at greater risk of severe COVID-19, even though the absolute risk of severe illness is low. COVID-19 also may increase the risk of adverse pregnancy outcomes, such as preterm birth, although the data have been mixed with some studies finding an association and others not.
“If pregnant people are part of a group that is recommended to receive a COVID-19 vaccine (e.g., health care personnel), they may choose to be vaccinated,” the CDC advises. “A conversation between the patient and their clinical team may assist with decisions regarding the use of vaccines approved under EUA for the prevention of COVID-19. While a conversation with a health care provider may be helpful, it is not required prior to vaccination.”
Acknowledging side effects and uncertainty
ACOG’s advisory reiterates that approach. The group notes that, based on the mRNA vaccine’s mechanism of action and its safety and efficacy in clinical trials, “it is expected that the safety and efficacy profile of the vaccine for pregnant individuals would be similar to that observed in nonpregnant individuals ... That said, there are no safety data specific to mRNA vaccine use in pregnant or lactating individuals and the potential risks to a pregnant individual and the fetus are unknown.”
In clinical trials, most participants experienced mild influenza-like symptoms following vaccination, including injection site reactions, fatigue, chills, muscle and joint pain, and headache. Among participants aged 18-55 years, fever greater than 38°C occurred in 3.7% of participants after the first dose and in 15.8% after the second dose. Most symptoms resolved within a few days.
Women who are pregnant should treat fever with acetaminophen because “fever has been associated with adverse pregnancy outcomes,” according to the ACOG guidance. “Acetaminophen has been proven to be safe for use in pregnancy and does not appear to impact antibody response to COVID-19 vaccines.” Patients may treat other vaccine side effects, such as injection-site soreness with acetaminophen as well.
When counseling patients, clinicians should explain that side effects are a normal part of developing antibodies to protect against COVID-19. “Regardless of their decision,” the group says, “these conversations provide an opportunity to remind patients about the importance of other prevention measures such as hand washing, physical distancing, and wearing a mask.”
More data expected
Data from developmental and reproductive toxicity studies in animals are expected soon, the CDC said. In addition, the manufacturer is following clinical trial participants who became pregnant during the study.
Women who are pregnant and their physicians should weigh factors such as the extent of COVID-19 transmission in the community, the patient’s risk of contracting COVID-19, risks of COVID-19 to the patient and fetus, vaccine efficacy and side effects, and the lack of data about COVID-19 vaccination during pregnancy.
The Society for Maternal-Fetal Medicine recommends that pregnant and lactating women have access to COVID-19 vaccines in general and has advocated for the inclusion of women who are pregnant or lactating in vaccine trials. The society has suggested that health care professionals “counsel their patients that the theoretical risk of fetal harm from mRNA vaccines is very low.” It published resources this week for physicians and patients focused on COVID-19 vaccination and pregnancy.
In a review published online Dec. 10 in the American Journal of Obstetrics & Gynecology MFM, Amanda M. Craig, MD, of Duke University Health System in Durham, N.C., and coauthors note that there “is a theoretical risk for fetal harm from any untested medical intervention and this is no different for COVID-19 vaccines.”
“Pregnant individuals should be given the opportunity, along with their obstetric provider, to weigh the potential risk of severe maternal disease against the unknown risk of fetal exposure, and make an autonomous decision about whether or not to accept vaccine until pregnancy safety data are available,” they write.
A version of this article first appeared on Medscape.com.
Moderna’s COVID-19 vaccine deemed ‘highly effective,’ but further studies needed
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) evaluated
The panel acknowledged that further studies will be required post issuance of an Emergency Use Authorization (EUA) to collect additional data on the safety and effectiveness of the vaccine. A briefing document released by the FDA on Dec. 17, 2020, summarized interim results and included recommendations from VRBPAC on use of Moderna’s mRNA-1273 COVID-19 vaccine.
“On November 30, 2020, ModernaTX (the Sponsor) submitted an EUA request to FDA for an investigational COVID-19 vaccine (mRNA-1273) intended to prevent COVID-19,” the committee wrote.
The mRNA-1273 vaccine trial
Among 30,351 individuals aged 18 years and older, the efficacy, safety, and immunogenicity of the mRNA-1273 vaccine candidate was evaluated in a randomized, stratified, observer-blind, placebo-controlled phase 3 study. Participants were randomly assigned (1:1) to receive two injections of either 100 mcg of mRNA-1273 (n = 15,181) or saline placebo (n = 15,170) administered intramuscularly on day 1 and day 29.
The primary efficacy endpoint was efficacy of mRNA-1273 against PCR-confirmed COVID-19 with onset at least 14 days following the second dose. The primary safety endpoint was to characterize the safety of the vaccine following one or two doses.
Efficacy
Among 27,817 subjects included in the first interim analysis (data cutoff: Nov. 7, 2020), 5 cases of COVID-19 with onset at least 14 days after the second dose occurred among vaccine recipients and 90 case occurred among placebo recipients, corresponding to 94.5% vaccine efficacy (95% confidence interval, 86.5%-97.8%).
“Subgroup analyses of the primary efficacy endpoint showed similar efficacy point estimates across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe COVID-19,” they reported.
Data from the final scheduled analysis of the primary efficacy endpoint (data cutoff: Nov. 21, 2020; median follow-up of >2 months after dose 2), demonstrated 94.1% vaccine efficacy (95% confidence interval, 89.3%-96.8%), corresponding to 11 cases of COVID-19 in the vaccine group and 185 cases in the placebo group.
When stratified by age, the vaccine efficacy was 95.6% (95% CI, 90.6%-97.9%) for individuals 18-64 years of age and 86.4% (95% CI, 61.4%-95.5%) for those 65 years of age or older.
In addition, results from secondary analyses indicated benefit for mRNA-1273 in preventing severe COVID-19 cases, COVID-19 in those with prior SARS-CoV-2 infection, and infection after the first dose, but these data were not conclusive.
Safety
Among 30,350 subjects included in the first interim analysis (data cutoff: Nov. 11, 2020; median follow-up of 7 weeks post second dose), no specific safety concerns were observed that would prevent issuance of an EUA.
Additional safety data (data cutoff: Nov. 25, 2020; median follow-up of 9 weeks post second dose) were provided on Dec. 7, 2020, but did not change the conclusions from the first interim analysis.
The most common vaccine-related adverse reactions were injection site pain (91.6%), fatigue (68.5%), headache (63.0%), muscle pain (59.6%), joint pain (44.8%), and chills (43.4%).
“The frequency of serious adverse events (SAEs) was low (1.0% in the mRNA-1273 arm and 1.0% in the placebo arm), without meaningful imbalances between study arms,” they reported.
Myocardial infarction (0.03%), nephrolithiasis (0.02%), and cholecystitis (0.02%) were the most common SAEs that were numerically greater in the vaccine arm than the placebo arm; however, the small number of cases does not infer a casual relationship.
“The 2-dose vaccination regimen was highly effective in preventing PCR-confirmed COVID-19 occurring at least 14 days after receipt of the second dose,” the committee wrote. “[However], it is critical to continue to gather data about the vaccine even after it is made available under EUA.”
The associated phase 3 study was sponsored by ModernaTX.
SOURCE: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) evaluated
The panel acknowledged that further studies will be required post issuance of an Emergency Use Authorization (EUA) to collect additional data on the safety and effectiveness of the vaccine. A briefing document released by the FDA on Dec. 17, 2020, summarized interim results and included recommendations from VRBPAC on use of Moderna’s mRNA-1273 COVID-19 vaccine.
“On November 30, 2020, ModernaTX (the Sponsor) submitted an EUA request to FDA for an investigational COVID-19 vaccine (mRNA-1273) intended to prevent COVID-19,” the committee wrote.
The mRNA-1273 vaccine trial
Among 30,351 individuals aged 18 years and older, the efficacy, safety, and immunogenicity of the mRNA-1273 vaccine candidate was evaluated in a randomized, stratified, observer-blind, placebo-controlled phase 3 study. Participants were randomly assigned (1:1) to receive two injections of either 100 mcg of mRNA-1273 (n = 15,181) or saline placebo (n = 15,170) administered intramuscularly on day 1 and day 29.
The primary efficacy endpoint was efficacy of mRNA-1273 against PCR-confirmed COVID-19 with onset at least 14 days following the second dose. The primary safety endpoint was to characterize the safety of the vaccine following one or two doses.
Efficacy
Among 27,817 subjects included in the first interim analysis (data cutoff: Nov. 7, 2020), 5 cases of COVID-19 with onset at least 14 days after the second dose occurred among vaccine recipients and 90 case occurred among placebo recipients, corresponding to 94.5% vaccine efficacy (95% confidence interval, 86.5%-97.8%).
“Subgroup analyses of the primary efficacy endpoint showed similar efficacy point estimates across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe COVID-19,” they reported.
Data from the final scheduled analysis of the primary efficacy endpoint (data cutoff: Nov. 21, 2020; median follow-up of >2 months after dose 2), demonstrated 94.1% vaccine efficacy (95% confidence interval, 89.3%-96.8%), corresponding to 11 cases of COVID-19 in the vaccine group and 185 cases in the placebo group.
When stratified by age, the vaccine efficacy was 95.6% (95% CI, 90.6%-97.9%) for individuals 18-64 years of age and 86.4% (95% CI, 61.4%-95.5%) for those 65 years of age or older.
In addition, results from secondary analyses indicated benefit for mRNA-1273 in preventing severe COVID-19 cases, COVID-19 in those with prior SARS-CoV-2 infection, and infection after the first dose, but these data were not conclusive.
Safety
Among 30,350 subjects included in the first interim analysis (data cutoff: Nov. 11, 2020; median follow-up of 7 weeks post second dose), no specific safety concerns were observed that would prevent issuance of an EUA.
Additional safety data (data cutoff: Nov. 25, 2020; median follow-up of 9 weeks post second dose) were provided on Dec. 7, 2020, but did not change the conclusions from the first interim analysis.
The most common vaccine-related adverse reactions were injection site pain (91.6%), fatigue (68.5%), headache (63.0%), muscle pain (59.6%), joint pain (44.8%), and chills (43.4%).
“The frequency of serious adverse events (SAEs) was low (1.0% in the mRNA-1273 arm and 1.0% in the placebo arm), without meaningful imbalances between study arms,” they reported.
Myocardial infarction (0.03%), nephrolithiasis (0.02%), and cholecystitis (0.02%) were the most common SAEs that were numerically greater in the vaccine arm than the placebo arm; however, the small number of cases does not infer a casual relationship.
“The 2-dose vaccination regimen was highly effective in preventing PCR-confirmed COVID-19 occurring at least 14 days after receipt of the second dose,” the committee wrote. “[However], it is critical to continue to gather data about the vaccine even after it is made available under EUA.”
The associated phase 3 study was sponsored by ModernaTX.
SOURCE: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
The Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) evaluated
The panel acknowledged that further studies will be required post issuance of an Emergency Use Authorization (EUA) to collect additional data on the safety and effectiveness of the vaccine. A briefing document released by the FDA on Dec. 17, 2020, summarized interim results and included recommendations from VRBPAC on use of Moderna’s mRNA-1273 COVID-19 vaccine.
“On November 30, 2020, ModernaTX (the Sponsor) submitted an EUA request to FDA for an investigational COVID-19 vaccine (mRNA-1273) intended to prevent COVID-19,” the committee wrote.
The mRNA-1273 vaccine trial
Among 30,351 individuals aged 18 years and older, the efficacy, safety, and immunogenicity of the mRNA-1273 vaccine candidate was evaluated in a randomized, stratified, observer-blind, placebo-controlled phase 3 study. Participants were randomly assigned (1:1) to receive two injections of either 100 mcg of mRNA-1273 (n = 15,181) or saline placebo (n = 15,170) administered intramuscularly on day 1 and day 29.
The primary efficacy endpoint was efficacy of mRNA-1273 against PCR-confirmed COVID-19 with onset at least 14 days following the second dose. The primary safety endpoint was to characterize the safety of the vaccine following one or two doses.
Efficacy
Among 27,817 subjects included in the first interim analysis (data cutoff: Nov. 7, 2020), 5 cases of COVID-19 with onset at least 14 days after the second dose occurred among vaccine recipients and 90 case occurred among placebo recipients, corresponding to 94.5% vaccine efficacy (95% confidence interval, 86.5%-97.8%).
“Subgroup analyses of the primary efficacy endpoint showed similar efficacy point estimates across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe COVID-19,” they reported.
Data from the final scheduled analysis of the primary efficacy endpoint (data cutoff: Nov. 21, 2020; median follow-up of >2 months after dose 2), demonstrated 94.1% vaccine efficacy (95% confidence interval, 89.3%-96.8%), corresponding to 11 cases of COVID-19 in the vaccine group and 185 cases in the placebo group.
When stratified by age, the vaccine efficacy was 95.6% (95% CI, 90.6%-97.9%) for individuals 18-64 years of age and 86.4% (95% CI, 61.4%-95.5%) for those 65 years of age or older.
In addition, results from secondary analyses indicated benefit for mRNA-1273 in preventing severe COVID-19 cases, COVID-19 in those with prior SARS-CoV-2 infection, and infection after the first dose, but these data were not conclusive.
Safety
Among 30,350 subjects included in the first interim analysis (data cutoff: Nov. 11, 2020; median follow-up of 7 weeks post second dose), no specific safety concerns were observed that would prevent issuance of an EUA.
Additional safety data (data cutoff: Nov. 25, 2020; median follow-up of 9 weeks post second dose) were provided on Dec. 7, 2020, but did not change the conclusions from the first interim analysis.
The most common vaccine-related adverse reactions were injection site pain (91.6%), fatigue (68.5%), headache (63.0%), muscle pain (59.6%), joint pain (44.8%), and chills (43.4%).
“The frequency of serious adverse events (SAEs) was low (1.0% in the mRNA-1273 arm and 1.0% in the placebo arm), without meaningful imbalances between study arms,” they reported.
Myocardial infarction (0.03%), nephrolithiasis (0.02%), and cholecystitis (0.02%) were the most common SAEs that were numerically greater in the vaccine arm than the placebo arm; however, the small number of cases does not infer a casual relationship.
“The 2-dose vaccination regimen was highly effective in preventing PCR-confirmed COVID-19 occurring at least 14 days after receipt of the second dose,” the committee wrote. “[However], it is critical to continue to gather data about the vaccine even after it is made available under EUA.”
The associated phase 3 study was sponsored by ModernaTX.
SOURCE: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
Key clinical point: The FDA’s Vaccines and Related Biological Products Advisory Committee regarded Moderna’s COVID-19 vaccine as highly effective with a favorable safety profile, based on interim phase 3 results.
Major finding: The two-dose vaccine regimen had a low frequency of serious adverse events (1.0% each in the mRNA-1273 and placebo arms, respectively) and demonstrated 94.1% (95% CI, 89.3%-96.8%) vaccine efficacy.
Study details: A briefing document summarized interim data and recommendations from the FDA’s VRBPAC on Moderna’s mRNA-1273 COVID-19 vaccine.
Disclosures: The associated phase 3 study was sponsored by ModernaTX.
Source: FDA Briefing Document: Moderna COVID-19 Vaccine. FDA Vaccines and Related Biological Products Advisory Committee. Published Dec. 17, 2020.
Strategies for tracking SARS-CoV-2 could help detect next pandemic
Two recently published studies indicate that COVID-19 infections were already circulating in the United States in December 2019. The question is whether these methodologies that could be applied to track the next pandemic.
One study evaluating blood donations found antibodies on the West coast as early as Dec. 13, 2019, and in blood donated on the East Coast by early January 2020 (Clin Infect Dis. 2020; Nov 30. doi: 10.1093/cid/ciaa1785). Both preceded the first documented COVID-19 infection in the United States, which has been widely reported as occurring on Jan. 19, 2020, in a traveler returning from China.
The other study, utilizing electronic medical record (EMR) analytics, demonstrated a spike in visits or hospitalizations for cough, a trend that persisted from Dec. 22, 2019, onward, exceeding norms for seasonal flu ( J Med Internet Res. 2020;22:e21562). This spike was interpreted as evidence that the SARS-CoV-2 pandemic was already underway before the first case was established.
While the ongoing serologic testing of blood donations for viral antibodies “will advance understanding of the epidemiology” for SARS-CoV-2 and “inform allocation of resources and public health prevention interventions to mitigate morbidity and mortality,” it might also be a strategy for disease surveillance in the next pandemic, according to a team led by investigators at the Centers for Disease Control and Prevention.
Blood donation surveillance is not now used routinely to monitor for population-based health threats, but it is not a new idea, according to the lead author of the study, Sridhar V. Basavaraju, MD, of Emory University and director of the CDC’s Office of Blood, Organ, and Other Tissue Safety, Atlanta, and his coinvestigators. Most recently, blood donation surveillance was used in the United States to track the penetration of the Zika virus.
For early detection of respiratory infections, blood donations might have unique advantages over alternatives, such as surveillance of respiratory specimens from symptomatic patients. Not least, blood donation surveillance captures individuals who are not seeking medical care, according to the investigators.
EMR surveillance might also have unique advantages for population-based monitoring of health threats. For one, aggregate data from large EMR systems have the potential to reveal symptom patterns before they become apparent at level of clinical care, according to a team of collaborating investigators from the University of California, Los Angeles, and the University of Washington, Seattle.
Emphasizing an urgent need for “agile healthcare analytics” to enable “disease surveillance in real time,” the first author of the EMR study, Joann G. Elmore, MD, professor in the department of health policy and management at the University of California, Los Angeles, expressed the hope that the approach will “lead to better preparation and the ability to quickly provide warnings and track the next pandemic.”
In the blood donation surveillance study, the goal was simply to determine whether SARS-CoV-2 reactive antibodies could be found in blood donations before the first case was identified. Of the 7,389 archived blood samples tested between Dec. 13, 2019, and Jan. 17, 2020, 106 (1.4%) were reactive.
These were not true positives, acknowledged the investigators. True positives would require reactive antibodies in the context of a positive molecular diagnostic test or paired acute convalescent sera with rising titers. The investigators also cautioned that false positives could not be completely ruled out, particularly in light of cross-reactivity that has been reported with other human coronaviruses.
Nevertheless, the monitoring of blood donations offers substantial promise for “understanding the dynamics of SARS-CoV-2 pandemic from early introduction,” and the CDC is now collaborating on ongoing surveillance with the goal of contributing information that could be applied “to mitigate morbidity and mortality.”
Lessons learned from this pandemic are potentially relevant to the next.
The EMR study simply looked at whether the word “cough” was included more often in the notes from visits or hospitalizations between December 2019 and February 2020 relative to the preceding 5 years. The investigators drew on data from three hospitals and more than 180 clinics.
From Dec. 22, 2019, onward, cough was noted above the 95% prediction interval for all 10 weeks of the study. The excess was seen in the outpatient setting and among hospitalized patients. There was also significant excess in the number of patients hospitalized with acute respiratory failure during the study period.
“Our approach to analyzing electronic records could be helpful in the future as we included consideration of data from the outpatient clinics in addition to the emergency departments and inpatient settings,” Dr. Elmore reported.
Surveillance of influenza and influenza-like infections has been undertaken in the United States for more than 20 years, but Dr. Elmore contends that EMR data, particularly data from outpatient clinics are “usually a harbinger of what is to come” for emergency department visits and, ultimately, hospitalizations. She thinks that this is a resource not yet fully exploited.
“There are always opportunities to better harness EMR data,” Dr. Elmore said.
These are intriguing studies and “useful” for reconsidering when SARS-CoV-2 was introduced in the United States, according to Janet G. Basemen, PhD, a professor of epidemiology and the associate dean of the University of Washington School of Public Health, Seattle. However, she noted that the task of translating data like these into actionable public health strategies has proven difficult in the past.
Symptom-based surveillance systems “have mostly served as situational awareness rather than early detection tools,” Dr. Baseman said. The problem is timely interpretation of a given signal.
Not that she doubts such tools “would be an incredible resource for humanity” if the current limitations can be resolved or that technological advances will lead to better methods of detecting and monitoring pandemics “at some point.” Rather, “we’re just not there yet,” she said.
SOURCE: Basavaraju SV et al. Clin Infect Dis. 2020 Nov 30. doi: 10.1093/cid/ciaa1785); Elmore JG et al. J Med Internet Res. 2020;22:e21562).
Two recently published studies indicate that COVID-19 infections were already circulating in the United States in December 2019. The question is whether these methodologies that could be applied to track the next pandemic.
One study evaluating blood donations found antibodies on the West coast as early as Dec. 13, 2019, and in blood donated on the East Coast by early January 2020 (Clin Infect Dis. 2020; Nov 30. doi: 10.1093/cid/ciaa1785). Both preceded the first documented COVID-19 infection in the United States, which has been widely reported as occurring on Jan. 19, 2020, in a traveler returning from China.
The other study, utilizing electronic medical record (EMR) analytics, demonstrated a spike in visits or hospitalizations for cough, a trend that persisted from Dec. 22, 2019, onward, exceeding norms for seasonal flu ( J Med Internet Res. 2020;22:e21562). This spike was interpreted as evidence that the SARS-CoV-2 pandemic was already underway before the first case was established.
While the ongoing serologic testing of blood donations for viral antibodies “will advance understanding of the epidemiology” for SARS-CoV-2 and “inform allocation of resources and public health prevention interventions to mitigate morbidity and mortality,” it might also be a strategy for disease surveillance in the next pandemic, according to a team led by investigators at the Centers for Disease Control and Prevention.
Blood donation surveillance is not now used routinely to monitor for population-based health threats, but it is not a new idea, according to the lead author of the study, Sridhar V. Basavaraju, MD, of Emory University and director of the CDC’s Office of Blood, Organ, and Other Tissue Safety, Atlanta, and his coinvestigators. Most recently, blood donation surveillance was used in the United States to track the penetration of the Zika virus.
For early detection of respiratory infections, blood donations might have unique advantages over alternatives, such as surveillance of respiratory specimens from symptomatic patients. Not least, blood donation surveillance captures individuals who are not seeking medical care, according to the investigators.
EMR surveillance might also have unique advantages for population-based monitoring of health threats. For one, aggregate data from large EMR systems have the potential to reveal symptom patterns before they become apparent at level of clinical care, according to a team of collaborating investigators from the University of California, Los Angeles, and the University of Washington, Seattle.
Emphasizing an urgent need for “agile healthcare analytics” to enable “disease surveillance in real time,” the first author of the EMR study, Joann G. Elmore, MD, professor in the department of health policy and management at the University of California, Los Angeles, expressed the hope that the approach will “lead to better preparation and the ability to quickly provide warnings and track the next pandemic.”
In the blood donation surveillance study, the goal was simply to determine whether SARS-CoV-2 reactive antibodies could be found in blood donations before the first case was identified. Of the 7,389 archived blood samples tested between Dec. 13, 2019, and Jan. 17, 2020, 106 (1.4%) were reactive.
These were not true positives, acknowledged the investigators. True positives would require reactive antibodies in the context of a positive molecular diagnostic test or paired acute convalescent sera with rising titers. The investigators also cautioned that false positives could not be completely ruled out, particularly in light of cross-reactivity that has been reported with other human coronaviruses.
Nevertheless, the monitoring of blood donations offers substantial promise for “understanding the dynamics of SARS-CoV-2 pandemic from early introduction,” and the CDC is now collaborating on ongoing surveillance with the goal of contributing information that could be applied “to mitigate morbidity and mortality.”
Lessons learned from this pandemic are potentially relevant to the next.
The EMR study simply looked at whether the word “cough” was included more often in the notes from visits or hospitalizations between December 2019 and February 2020 relative to the preceding 5 years. The investigators drew on data from three hospitals and more than 180 clinics.
From Dec. 22, 2019, onward, cough was noted above the 95% prediction interval for all 10 weeks of the study. The excess was seen in the outpatient setting and among hospitalized patients. There was also significant excess in the number of patients hospitalized with acute respiratory failure during the study period.
“Our approach to analyzing electronic records could be helpful in the future as we included consideration of data from the outpatient clinics in addition to the emergency departments and inpatient settings,” Dr. Elmore reported.
Surveillance of influenza and influenza-like infections has been undertaken in the United States for more than 20 years, but Dr. Elmore contends that EMR data, particularly data from outpatient clinics are “usually a harbinger of what is to come” for emergency department visits and, ultimately, hospitalizations. She thinks that this is a resource not yet fully exploited.
“There are always opportunities to better harness EMR data,” Dr. Elmore said.
These are intriguing studies and “useful” for reconsidering when SARS-CoV-2 was introduced in the United States, according to Janet G. Basemen, PhD, a professor of epidemiology and the associate dean of the University of Washington School of Public Health, Seattle. However, she noted that the task of translating data like these into actionable public health strategies has proven difficult in the past.
Symptom-based surveillance systems “have mostly served as situational awareness rather than early detection tools,” Dr. Baseman said. The problem is timely interpretation of a given signal.
Not that she doubts such tools “would be an incredible resource for humanity” if the current limitations can be resolved or that technological advances will lead to better methods of detecting and monitoring pandemics “at some point.” Rather, “we’re just not there yet,” she said.
SOURCE: Basavaraju SV et al. Clin Infect Dis. 2020 Nov 30. doi: 10.1093/cid/ciaa1785); Elmore JG et al. J Med Internet Res. 2020;22:e21562).
Two recently published studies indicate that COVID-19 infections were already circulating in the United States in December 2019. The question is whether these methodologies that could be applied to track the next pandemic.
One study evaluating blood donations found antibodies on the West coast as early as Dec. 13, 2019, and in blood donated on the East Coast by early January 2020 (Clin Infect Dis. 2020; Nov 30. doi: 10.1093/cid/ciaa1785). Both preceded the first documented COVID-19 infection in the United States, which has been widely reported as occurring on Jan. 19, 2020, in a traveler returning from China.
The other study, utilizing electronic medical record (EMR) analytics, demonstrated a spike in visits or hospitalizations for cough, a trend that persisted from Dec. 22, 2019, onward, exceeding norms for seasonal flu ( J Med Internet Res. 2020;22:e21562). This spike was interpreted as evidence that the SARS-CoV-2 pandemic was already underway before the first case was established.
While the ongoing serologic testing of blood donations for viral antibodies “will advance understanding of the epidemiology” for SARS-CoV-2 and “inform allocation of resources and public health prevention interventions to mitigate morbidity and mortality,” it might also be a strategy for disease surveillance in the next pandemic, according to a team led by investigators at the Centers for Disease Control and Prevention.
Blood donation surveillance is not now used routinely to monitor for population-based health threats, but it is not a new idea, according to the lead author of the study, Sridhar V. Basavaraju, MD, of Emory University and director of the CDC’s Office of Blood, Organ, and Other Tissue Safety, Atlanta, and his coinvestigators. Most recently, blood donation surveillance was used in the United States to track the penetration of the Zika virus.
For early detection of respiratory infections, blood donations might have unique advantages over alternatives, such as surveillance of respiratory specimens from symptomatic patients. Not least, blood donation surveillance captures individuals who are not seeking medical care, according to the investigators.
EMR surveillance might also have unique advantages for population-based monitoring of health threats. For one, aggregate data from large EMR systems have the potential to reveal symptom patterns before they become apparent at level of clinical care, according to a team of collaborating investigators from the University of California, Los Angeles, and the University of Washington, Seattle.
Emphasizing an urgent need for “agile healthcare analytics” to enable “disease surveillance in real time,” the first author of the EMR study, Joann G. Elmore, MD, professor in the department of health policy and management at the University of California, Los Angeles, expressed the hope that the approach will “lead to better preparation and the ability to quickly provide warnings and track the next pandemic.”
In the blood donation surveillance study, the goal was simply to determine whether SARS-CoV-2 reactive antibodies could be found in blood donations before the first case was identified. Of the 7,389 archived blood samples tested between Dec. 13, 2019, and Jan. 17, 2020, 106 (1.4%) were reactive.
These were not true positives, acknowledged the investigators. True positives would require reactive antibodies in the context of a positive molecular diagnostic test or paired acute convalescent sera with rising titers. The investigators also cautioned that false positives could not be completely ruled out, particularly in light of cross-reactivity that has been reported with other human coronaviruses.
Nevertheless, the monitoring of blood donations offers substantial promise for “understanding the dynamics of SARS-CoV-2 pandemic from early introduction,” and the CDC is now collaborating on ongoing surveillance with the goal of contributing information that could be applied “to mitigate morbidity and mortality.”
Lessons learned from this pandemic are potentially relevant to the next.
The EMR study simply looked at whether the word “cough” was included more often in the notes from visits or hospitalizations between December 2019 and February 2020 relative to the preceding 5 years. The investigators drew on data from three hospitals and more than 180 clinics.
From Dec. 22, 2019, onward, cough was noted above the 95% prediction interval for all 10 weeks of the study. The excess was seen in the outpatient setting and among hospitalized patients. There was also significant excess in the number of patients hospitalized with acute respiratory failure during the study period.
“Our approach to analyzing electronic records could be helpful in the future as we included consideration of data from the outpatient clinics in addition to the emergency departments and inpatient settings,” Dr. Elmore reported.
Surveillance of influenza and influenza-like infections has been undertaken in the United States for more than 20 years, but Dr. Elmore contends that EMR data, particularly data from outpatient clinics are “usually a harbinger of what is to come” for emergency department visits and, ultimately, hospitalizations. She thinks that this is a resource not yet fully exploited.
“There are always opportunities to better harness EMR data,” Dr. Elmore said.
These are intriguing studies and “useful” for reconsidering when SARS-CoV-2 was introduced in the United States, according to Janet G. Basemen, PhD, a professor of epidemiology and the associate dean of the University of Washington School of Public Health, Seattle. However, she noted that the task of translating data like these into actionable public health strategies has proven difficult in the past.
Symptom-based surveillance systems “have mostly served as situational awareness rather than early detection tools,” Dr. Baseman said. The problem is timely interpretation of a given signal.
Not that she doubts such tools “would be an incredible resource for humanity” if the current limitations can be resolved or that technological advances will lead to better methods of detecting and monitoring pandemics “at some point.” Rather, “we’re just not there yet,” she said.
SOURCE: Basavaraju SV et al. Clin Infect Dis. 2020 Nov 30. doi: 10.1093/cid/ciaa1785); Elmore JG et al. J Med Internet Res. 2020;22:e21562).
FDA OKs osimertinib as first adjuvant drug for NSCLC
Osimertinib was first approved in the US in 2018 for the first-line treatment of patients with metastatic EGFR-mutated NSCLC.
With this new indication, “patients may be treated with this targeted therapy in an earlier and potentially more curative stage of non-small cell lung cancer,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the FDA’s Center for Drug Evaluation and Research, said in a news release.
The expanded indication is based on results of the ADAURA clinical trial, which compared osimertinib with placebo following complete resection of localized or locally advanced NSCLC with negative margins.
In the trial, adjuvant osimertinib reduced the relative risk of disease recurrence or death by 83% in patients with stage II and IIIA disease (hazard ratio [HR], 0.17; 95% CI, 0.12 - 0.23; P < .0001).
Disease-free survival (DFS) in the overall trial population of patients with stage IB-IIIA disease showed osimertinib reduced the risk of disease recurrence or death by 80% (HR, 0.20; 95% CI, 0.15 - 0.27; P < .0001).
At 2 years, 89% of patients treated with the targeted agent remained alive and disease free vs 52% on placebo after surgery. The safety and tolerability of osimertinib in the adjuvant setting was consistent with previous trials in the metastatic setting.
The trial of 682 patients was unblinded early and halted on the recommendation of the independent data-monitoring committee, because of the efficacy of osimertinib.
“If I were on the committee, I would have done the same thing. These are extraordinary results,” study investigator Roy S. Herbst, MD, PhD, chief of medical oncology at the Yale Cancer Center, New Haven, Connecticut, said at a press briefing prior to the study presentation at the American Society of Clinical Oncology’s (ASCO) virtual scientific program last spring.
In a Medscape commentary, Mark Kris, MD, of Memorial Sloan Kettering Cancer Center in New York City, said the data with osimertinib in the adjuvant setting are “important and practice-changing.”
“The potential for this drug to improve outcomes has been there for a long time. This phase 3 randomized trial presented at the plenary session of ASCO showed a more than doubling of disease-free survival at 2 years. It shows that we can use therapies in the earlier stages of disease,” Kris noted.
“This approval dispels the notion that treatment is over after surgery and chemotherapy, as the ADAURA results show that Tagrisso can dramatically change the course of this disease,” Dave Fredrickson, executive vice president, AstraZeneca oncology business unit, said in a news release.
Osimertinib had orphan drug status and breakthrough therapy designation for treatment of EGFR mutation-positive NSCLC.
A version of this article first appeared on Medscape.com.
Osimertinib was first approved in the US in 2018 for the first-line treatment of patients with metastatic EGFR-mutated NSCLC.
With this new indication, “patients may be treated with this targeted therapy in an earlier and potentially more curative stage of non-small cell lung cancer,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the FDA’s Center for Drug Evaluation and Research, said in a news release.
The expanded indication is based on results of the ADAURA clinical trial, which compared osimertinib with placebo following complete resection of localized or locally advanced NSCLC with negative margins.
In the trial, adjuvant osimertinib reduced the relative risk of disease recurrence or death by 83% in patients with stage II and IIIA disease (hazard ratio [HR], 0.17; 95% CI, 0.12 - 0.23; P < .0001).
Disease-free survival (DFS) in the overall trial population of patients with stage IB-IIIA disease showed osimertinib reduced the risk of disease recurrence or death by 80% (HR, 0.20; 95% CI, 0.15 - 0.27; P < .0001).
At 2 years, 89% of patients treated with the targeted agent remained alive and disease free vs 52% on placebo after surgery. The safety and tolerability of osimertinib in the adjuvant setting was consistent with previous trials in the metastatic setting.
The trial of 682 patients was unblinded early and halted on the recommendation of the independent data-monitoring committee, because of the efficacy of osimertinib.
“If I were on the committee, I would have done the same thing. These are extraordinary results,” study investigator Roy S. Herbst, MD, PhD, chief of medical oncology at the Yale Cancer Center, New Haven, Connecticut, said at a press briefing prior to the study presentation at the American Society of Clinical Oncology’s (ASCO) virtual scientific program last spring.
In a Medscape commentary, Mark Kris, MD, of Memorial Sloan Kettering Cancer Center in New York City, said the data with osimertinib in the adjuvant setting are “important and practice-changing.”
“The potential for this drug to improve outcomes has been there for a long time. This phase 3 randomized trial presented at the plenary session of ASCO showed a more than doubling of disease-free survival at 2 years. It shows that we can use therapies in the earlier stages of disease,” Kris noted.
“This approval dispels the notion that treatment is over after surgery and chemotherapy, as the ADAURA results show that Tagrisso can dramatically change the course of this disease,” Dave Fredrickson, executive vice president, AstraZeneca oncology business unit, said in a news release.
Osimertinib had orphan drug status and breakthrough therapy designation for treatment of EGFR mutation-positive NSCLC.
A version of this article first appeared on Medscape.com.
Osimertinib was first approved in the US in 2018 for the first-line treatment of patients with metastatic EGFR-mutated NSCLC.
With this new indication, “patients may be treated with this targeted therapy in an earlier and potentially more curative stage of non-small cell lung cancer,” Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the FDA’s Center for Drug Evaluation and Research, said in a news release.
The expanded indication is based on results of the ADAURA clinical trial, which compared osimertinib with placebo following complete resection of localized or locally advanced NSCLC with negative margins.
In the trial, adjuvant osimertinib reduced the relative risk of disease recurrence or death by 83% in patients with stage II and IIIA disease (hazard ratio [HR], 0.17; 95% CI, 0.12 - 0.23; P < .0001).
Disease-free survival (DFS) in the overall trial population of patients with stage IB-IIIA disease showed osimertinib reduced the risk of disease recurrence or death by 80% (HR, 0.20; 95% CI, 0.15 - 0.27; P < .0001).
At 2 years, 89% of patients treated with the targeted agent remained alive and disease free vs 52% on placebo after surgery. The safety and tolerability of osimertinib in the adjuvant setting was consistent with previous trials in the metastatic setting.
The trial of 682 patients was unblinded early and halted on the recommendation of the independent data-monitoring committee, because of the efficacy of osimertinib.
“If I were on the committee, I would have done the same thing. These are extraordinary results,” study investigator Roy S. Herbst, MD, PhD, chief of medical oncology at the Yale Cancer Center, New Haven, Connecticut, said at a press briefing prior to the study presentation at the American Society of Clinical Oncology’s (ASCO) virtual scientific program last spring.
In a Medscape commentary, Mark Kris, MD, of Memorial Sloan Kettering Cancer Center in New York City, said the data with osimertinib in the adjuvant setting are “important and practice-changing.”
“The potential for this drug to improve outcomes has been there for a long time. This phase 3 randomized trial presented at the plenary session of ASCO showed a more than doubling of disease-free survival at 2 years. It shows that we can use therapies in the earlier stages of disease,” Kris noted.
“This approval dispels the notion that treatment is over after surgery and chemotherapy, as the ADAURA results show that Tagrisso can dramatically change the course of this disease,” Dave Fredrickson, executive vice president, AstraZeneca oncology business unit, said in a news release.
Osimertinib had orphan drug status and breakthrough therapy designation for treatment of EGFR mutation-positive NSCLC.
A version of this article first appeared on Medscape.com.
No benefit of cannabis on depression in pregnant women with OUD
Cannabis is ineffective at alleviating depression in pregnant women undergoing opioid agonist therapy (OAT), new research shows.
A study of more than 120 pregnant women undergoing treatment of opioid use disorder (OUD) showed that those who used cannabis to alleviate their depressive symptoms while undergoing OAT continued to have high depression scores at the end of opioid treatment.
In addition, depression scores improved for those who abstained from cannabis use after their first positive screen. Interestingly, cannabis use did not affect patient retention in treatment for OUD, the investigators note.
“To our knowledge, this is the first time looking at the impact of cannabis on the specific population of pregnant women with opioid use disorder, who are very vulnerable to depression,” lead author Abigail Richison, MD, University of Arkansas for Medical Sciences, Little Rock, said in an interview.
The findings were presented at the American Academy of Addiction Psychiatry (AAAP) 31st Annual Meeting, which was held online this year because of the COVID-19 pandemic.
A safer alternative?
Data from the National Survey on Drug Use and Health show that perinatal cannabis use increased by 62% between 2002 and 2014. Many women try to ameliorate their depression symptoms by using cannabis in the mistaken belief that it will help their depression, the investigators noted.
In addition, many women consider cannabis safer during pregnancy than prescribed medications for improving mood, said Dr. Richison. She said that cannabis does not alleviate depression and may even worsen it.
Dr. Richison noted that at her center, which has a women’s health program that treats pregnant women with OUDs, she was seeing a lot of patients who reported using cannabis to improve their mood.
“However, it didn’t seem like it was really helping, so I started researching about cannabis and depression,” Dr. Richison said.
“ and can be accused of perinatal substance use. I think it is very important to screen for depression as well as cannabis use in this population,” she added.
To shed some light on the impact of cannabis use by pregnant patients with OUD, the investigators conducted a retrospective chart review of 121 pregnant women with OUD who attended outpatient OAT. All were prescribed buprenorphine.
At each visit, Beck Depression Inventory (BDI) scores were obtained and urine drug screens were administered. The primary outcome was BDI score. Other measures included retention, urinary drug screen results, and antidepressant use.
The women were divided into two groups. The first comprised cannabis users, defined as having more than one urine drug screen that was positive for cannabis (n = 35). The other group comprised nonusers, defined as having urine drug screens that were negative for cannabis (n = 86).
Cannabis users were a little younger (mean age, 27 years) than non–cannabis users (mean age, 29.5 years; P = .006). Most of the participants were White (80.2%). Roughly half were on Medicaid, and most of the other participants had private insurance; a small number of women had no insurance.
Results showed that cannabis users had significantly higher BDI scores than non–cannabis users (mean scores, 16 vs. 9.3; P < .001).
Cannabis use continued to be associated with elevated scores for depression when controlling for opioid misuse and antidepressant use. There were no significant differences in retention or lapse to opioid misuse between the two groups.
More evidence of risk
Commenting on the findings in an interview, Carla Marienfeld, MD, professor of psychiatry at the University of California, San Diego, said there is a growing body of evidence about risks from cannabis use during pregnancy, “a time where we already know the endocannabinoid system is very active in the developing fetus.”
She noted that the current study’s design makes it hard to know whether marijuana use causes worse depression.
However, “it clearly is not associated with helping to improve mood the way people who are using it believe or hope for,” said Dr. Marienfeld, who was not part of the research.
“The risk for harm in terms of worse mood for the pregnant woman or risks for harm to the developing fetus are being better understood with many new studies,” she added.
Yet as more and more states legalize medical marijuana, cannabis use during pregnancy is only going to rise, experts fear.
Cornel Stanciu, MD, of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., who was asked for comment, noted that public endorsement for potential benefits of the marijuana plant is at an all-time high.
“To date, 33 states and the District of Columbia have responded by legalizing medical marijuana, with 10 states also having legalized recreational use of marijuana. The current practice is said to be ahead of science, as robust research has been hindered by strict regulations – and most epidemiological studies point toward harmful associations,” Dr. Stanciu said in an interview.
“Given the decreased perception of harm by the general public, women are certainly compelled to seek what they perceive as more natural self-management remedies,” he said.
A harmful habit
Dr. Stanciu cited a recent study conducted in Colorado in which researchers contacted cannabis dispensaries, identified themselves as being pregnant, and asked for guidance in managing pregnancy-related symptoms.
Almost 70% of dispensaries recommended products to treat symptoms, particularly in the vulnerable first trimester; 36% of them also provided reassurance of the safety profile. Very few encouraged a discussion with the physician.
“Consumption of cannabis during pregnancy results in cannabinoid placental crossing and accumulation in the fetal brain, as well as other organs, where it interferes with neurodevelopment and the endocannabinoid system,” he said.
In addition, retrospective studies have shown an association between prenatal cannabis ingestion and anemia in the mothers, low birth weight, greater risk for preterm and stillbirths, and increased need for neonatal ICU admissions.
“Children born to mothers who used cannabis during pregnancy have higher rates of impulsivity, delinquency, learning and memory impairment, as well as executive function deficits. There is also an increased association with proneness to psychosis during middle childhood,” Dr. Stanciu said.
When used during pregnancy, cannabis has been associated with increased anxiety in mothers, as well as increased risk for depressive disorders, incidence of suicidal ideations and behavior, and symptoms of mania and psychosis among those with bipolar and schizophrenia spectrum conditions. Cannabis has also been linked to coingestion of other substances and with alcohol use.
“So cannabis can pose harm, especially when used by those with affective disorders,” Dr. Stanciu said.
The study was funded by the National Institute on Drug Abuse. Dr. Richison, Dr. Marienfeld, and Dr. Stanciu have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com
Cannabis is ineffective at alleviating depression in pregnant women undergoing opioid agonist therapy (OAT), new research shows.
A study of more than 120 pregnant women undergoing treatment of opioid use disorder (OUD) showed that those who used cannabis to alleviate their depressive symptoms while undergoing OAT continued to have high depression scores at the end of opioid treatment.
In addition, depression scores improved for those who abstained from cannabis use after their first positive screen. Interestingly, cannabis use did not affect patient retention in treatment for OUD, the investigators note.
“To our knowledge, this is the first time looking at the impact of cannabis on the specific population of pregnant women with opioid use disorder, who are very vulnerable to depression,” lead author Abigail Richison, MD, University of Arkansas for Medical Sciences, Little Rock, said in an interview.
The findings were presented at the American Academy of Addiction Psychiatry (AAAP) 31st Annual Meeting, which was held online this year because of the COVID-19 pandemic.
A safer alternative?
Data from the National Survey on Drug Use and Health show that perinatal cannabis use increased by 62% between 2002 and 2014. Many women try to ameliorate their depression symptoms by using cannabis in the mistaken belief that it will help their depression, the investigators noted.
In addition, many women consider cannabis safer during pregnancy than prescribed medications for improving mood, said Dr. Richison. She said that cannabis does not alleviate depression and may even worsen it.
Dr. Richison noted that at her center, which has a women’s health program that treats pregnant women with OUDs, she was seeing a lot of patients who reported using cannabis to improve their mood.
“However, it didn’t seem like it was really helping, so I started researching about cannabis and depression,” Dr. Richison said.
“ and can be accused of perinatal substance use. I think it is very important to screen for depression as well as cannabis use in this population,” she added.
To shed some light on the impact of cannabis use by pregnant patients with OUD, the investigators conducted a retrospective chart review of 121 pregnant women with OUD who attended outpatient OAT. All were prescribed buprenorphine.
At each visit, Beck Depression Inventory (BDI) scores were obtained and urine drug screens were administered. The primary outcome was BDI score. Other measures included retention, urinary drug screen results, and antidepressant use.
The women were divided into two groups. The first comprised cannabis users, defined as having more than one urine drug screen that was positive for cannabis (n = 35). The other group comprised nonusers, defined as having urine drug screens that were negative for cannabis (n = 86).
Cannabis users were a little younger (mean age, 27 years) than non–cannabis users (mean age, 29.5 years; P = .006). Most of the participants were White (80.2%). Roughly half were on Medicaid, and most of the other participants had private insurance; a small number of women had no insurance.
Results showed that cannabis users had significantly higher BDI scores than non–cannabis users (mean scores, 16 vs. 9.3; P < .001).
Cannabis use continued to be associated with elevated scores for depression when controlling for opioid misuse and antidepressant use. There were no significant differences in retention or lapse to opioid misuse between the two groups.
More evidence of risk
Commenting on the findings in an interview, Carla Marienfeld, MD, professor of psychiatry at the University of California, San Diego, said there is a growing body of evidence about risks from cannabis use during pregnancy, “a time where we already know the endocannabinoid system is very active in the developing fetus.”
She noted that the current study’s design makes it hard to know whether marijuana use causes worse depression.
However, “it clearly is not associated with helping to improve mood the way people who are using it believe or hope for,” said Dr. Marienfeld, who was not part of the research.
“The risk for harm in terms of worse mood for the pregnant woman or risks for harm to the developing fetus are being better understood with many new studies,” she added.
Yet as more and more states legalize medical marijuana, cannabis use during pregnancy is only going to rise, experts fear.
Cornel Stanciu, MD, of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., who was asked for comment, noted that public endorsement for potential benefits of the marijuana plant is at an all-time high.
“To date, 33 states and the District of Columbia have responded by legalizing medical marijuana, with 10 states also having legalized recreational use of marijuana. The current practice is said to be ahead of science, as robust research has been hindered by strict regulations – and most epidemiological studies point toward harmful associations,” Dr. Stanciu said in an interview.
“Given the decreased perception of harm by the general public, women are certainly compelled to seek what they perceive as more natural self-management remedies,” he said.
A harmful habit
Dr. Stanciu cited a recent study conducted in Colorado in which researchers contacted cannabis dispensaries, identified themselves as being pregnant, and asked for guidance in managing pregnancy-related symptoms.
Almost 70% of dispensaries recommended products to treat symptoms, particularly in the vulnerable first trimester; 36% of them also provided reassurance of the safety profile. Very few encouraged a discussion with the physician.
“Consumption of cannabis during pregnancy results in cannabinoid placental crossing and accumulation in the fetal brain, as well as other organs, where it interferes with neurodevelopment and the endocannabinoid system,” he said.
In addition, retrospective studies have shown an association between prenatal cannabis ingestion and anemia in the mothers, low birth weight, greater risk for preterm and stillbirths, and increased need for neonatal ICU admissions.
“Children born to mothers who used cannabis during pregnancy have higher rates of impulsivity, delinquency, learning and memory impairment, as well as executive function deficits. There is also an increased association with proneness to psychosis during middle childhood,” Dr. Stanciu said.
When used during pregnancy, cannabis has been associated with increased anxiety in mothers, as well as increased risk for depressive disorders, incidence of suicidal ideations and behavior, and symptoms of mania and psychosis among those with bipolar and schizophrenia spectrum conditions. Cannabis has also been linked to coingestion of other substances and with alcohol use.
“So cannabis can pose harm, especially when used by those with affective disorders,” Dr. Stanciu said.
The study was funded by the National Institute on Drug Abuse. Dr. Richison, Dr. Marienfeld, and Dr. Stanciu have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com
Cannabis is ineffective at alleviating depression in pregnant women undergoing opioid agonist therapy (OAT), new research shows.
A study of more than 120 pregnant women undergoing treatment of opioid use disorder (OUD) showed that those who used cannabis to alleviate their depressive symptoms while undergoing OAT continued to have high depression scores at the end of opioid treatment.
In addition, depression scores improved for those who abstained from cannabis use after their first positive screen. Interestingly, cannabis use did not affect patient retention in treatment for OUD, the investigators note.
“To our knowledge, this is the first time looking at the impact of cannabis on the specific population of pregnant women with opioid use disorder, who are very vulnerable to depression,” lead author Abigail Richison, MD, University of Arkansas for Medical Sciences, Little Rock, said in an interview.
The findings were presented at the American Academy of Addiction Psychiatry (AAAP) 31st Annual Meeting, which was held online this year because of the COVID-19 pandemic.
A safer alternative?
Data from the National Survey on Drug Use and Health show that perinatal cannabis use increased by 62% between 2002 and 2014. Many women try to ameliorate their depression symptoms by using cannabis in the mistaken belief that it will help their depression, the investigators noted.
In addition, many women consider cannabis safer during pregnancy than prescribed medications for improving mood, said Dr. Richison. She said that cannabis does not alleviate depression and may even worsen it.
Dr. Richison noted that at her center, which has a women’s health program that treats pregnant women with OUDs, she was seeing a lot of patients who reported using cannabis to improve their mood.
“However, it didn’t seem like it was really helping, so I started researching about cannabis and depression,” Dr. Richison said.
“ and can be accused of perinatal substance use. I think it is very important to screen for depression as well as cannabis use in this population,” she added.
To shed some light on the impact of cannabis use by pregnant patients with OUD, the investigators conducted a retrospective chart review of 121 pregnant women with OUD who attended outpatient OAT. All were prescribed buprenorphine.
At each visit, Beck Depression Inventory (BDI) scores were obtained and urine drug screens were administered. The primary outcome was BDI score. Other measures included retention, urinary drug screen results, and antidepressant use.
The women were divided into two groups. The first comprised cannabis users, defined as having more than one urine drug screen that was positive for cannabis (n = 35). The other group comprised nonusers, defined as having urine drug screens that were negative for cannabis (n = 86).
Cannabis users were a little younger (mean age, 27 years) than non–cannabis users (mean age, 29.5 years; P = .006). Most of the participants were White (80.2%). Roughly half were on Medicaid, and most of the other participants had private insurance; a small number of women had no insurance.
Results showed that cannabis users had significantly higher BDI scores than non–cannabis users (mean scores, 16 vs. 9.3; P < .001).
Cannabis use continued to be associated with elevated scores for depression when controlling for opioid misuse and antidepressant use. There were no significant differences in retention or lapse to opioid misuse between the two groups.
More evidence of risk
Commenting on the findings in an interview, Carla Marienfeld, MD, professor of psychiatry at the University of California, San Diego, said there is a growing body of evidence about risks from cannabis use during pregnancy, “a time where we already know the endocannabinoid system is very active in the developing fetus.”
She noted that the current study’s design makes it hard to know whether marijuana use causes worse depression.
However, “it clearly is not associated with helping to improve mood the way people who are using it believe or hope for,” said Dr. Marienfeld, who was not part of the research.
“The risk for harm in terms of worse mood for the pregnant woman or risks for harm to the developing fetus are being better understood with many new studies,” she added.
Yet as more and more states legalize medical marijuana, cannabis use during pregnancy is only going to rise, experts fear.
Cornel Stanciu, MD, of Dartmouth-Hitchcock Medical Center, Lebanon, N.H., who was asked for comment, noted that public endorsement for potential benefits of the marijuana plant is at an all-time high.
“To date, 33 states and the District of Columbia have responded by legalizing medical marijuana, with 10 states also having legalized recreational use of marijuana. The current practice is said to be ahead of science, as robust research has been hindered by strict regulations – and most epidemiological studies point toward harmful associations,” Dr. Stanciu said in an interview.
“Given the decreased perception of harm by the general public, women are certainly compelled to seek what they perceive as more natural self-management remedies,” he said.
A harmful habit
Dr. Stanciu cited a recent study conducted in Colorado in which researchers contacted cannabis dispensaries, identified themselves as being pregnant, and asked for guidance in managing pregnancy-related symptoms.
Almost 70% of dispensaries recommended products to treat symptoms, particularly in the vulnerable first trimester; 36% of them also provided reassurance of the safety profile. Very few encouraged a discussion with the physician.
“Consumption of cannabis during pregnancy results in cannabinoid placental crossing and accumulation in the fetal brain, as well as other organs, where it interferes with neurodevelopment and the endocannabinoid system,” he said.
In addition, retrospective studies have shown an association between prenatal cannabis ingestion and anemia in the mothers, low birth weight, greater risk for preterm and stillbirths, and increased need for neonatal ICU admissions.
“Children born to mothers who used cannabis during pregnancy have higher rates of impulsivity, delinquency, learning and memory impairment, as well as executive function deficits. There is also an increased association with proneness to psychosis during middle childhood,” Dr. Stanciu said.
When used during pregnancy, cannabis has been associated with increased anxiety in mothers, as well as increased risk for depressive disorders, incidence of suicidal ideations and behavior, and symptoms of mania and psychosis among those with bipolar and schizophrenia spectrum conditions. Cannabis has also been linked to coingestion of other substances and with alcohol use.
“So cannabis can pose harm, especially when used by those with affective disorders,” Dr. Stanciu said.
The study was funded by the National Institute on Drug Abuse. Dr. Richison, Dr. Marienfeld, and Dr. Stanciu have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com
COVID-19 variant sparks U.K. travel restrictions
Researchers have detected a highly contagious coronavirus variant in the United Kingdom, leading Prime Minister Boris Johnson to shut down parts of the country and triggering other nations to impose travel and shipping restrictions on England.
Mr. Johnson held a crisis meeting with ministers Monday after Saturday’s shutdown announcement. The prime minister said in a nationally televised address that this coronavirus variant may be “up to 70% more transmissible than the old variant” and was probably responsible for an increase in cases in southeastern England.
“There is still much we don’t know. While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant,” he said.
Public Health England says it is working to learn as much about the variant as possible. “We know that mortality is a lagging indicator, and we will need to continually monitor this over the coming weeks,” the agency says.
That scientific uncertainty about the variant’s threat shook European nations that were rushing to ship goods to England in advance of a Dec. 31 Brexit deadline. Under Brexit, which is short for “British exit,” the United Kingdom will leave the European Union on Jan. 31, 2020. Until then, the two sides will come up with new trade and security relationships.
European Union members Austria, Belgium, Bulgaria, France, Germany, Ireland, Italy, and the Netherlands announced travel restrictions hours after Johnson’s speech.
Those restrictions created food uncertainty across the U.K., which imports about a quarter of its food from the EU, according to The New York Times. Long lines of trucks heading to ports in the U.K. came to a standstill on major roads such as the M20 near Kent and the Port of Dover.
Outside Europe, Canada, India, Iran, Israel, Hong Kong, Saudi Arabia, and Turkey banned all incoming flights from the U.K. And more bans could come.
The U.S. reaction
The United States has not imposed any new limits on travel with the United Kingdom, although New York Gov. Andrew Cuomo (D) has requested all passengers bound for John F. Kennedy International Airport from the U.K. be tested before boarding and a new travel ban be placed for Europe. He says the federal government must take action now to avoid a crisis situation like the one New York experienced in March and April.
“The United States has a number of flights coming in from the U.K. each day, and we have done absolutely nothing,” Mr. Cuomo said in a statement on the governor’s webpage. “To me, this is reprehensible because this is what happened in the spring. How many times in life do you have to make the same mistake before you learn?”
Leading U.S. health officials have downplayed the dangers of the virus.
“We don’t know that it’s more dangerous, and very importantly, we have not seen a single mutation yet that would make it evade the vaccine,” U.S. Assistant Secretary of Health and Human Services Adm. Brett Giroir, MD, said Sunday on ABC’s This Week with George Stephanopoulos. “I can’t say that won’t happen in the future, but right now it looks like the vaccine will cover everything that we see.”
Dr. Giroir said the HHS and other U.S. government agencies will monitor the variant.
“Viruses mutate,” he said. “We’ve seen almost 4,000 different mutations among this virus. There is no indication that the mutation right now that they’re talking about is overcoming England.”
Where did the variant come from?
Public Health England says the coronavirus variant had existed in the U.K. since September and circulated at very low levels until mid-November.
“The increase in cases linked to the new variant first came to light in late November when PHE was investigating why infection rates in Kent were not falling despite national restrictions. We then discovered a cluster linked to this variant spreading rapidly into London and Essex,” the agency said.
Public Health England says there’s no evidence the new variant is resistant to the Pfizer-BioNTech vaccine, which is now being given across the country to high-priority groups such as health care workers.
An article in The BMJ, a British medical journal, says the variant was first detected by Covid-19 Genomics UK, a consortium that tests the random genetic sequencing of positive COVID-19 samples around the U.K. The variant cases were mostly in the southeast of England.
A University of Birmingham professor said in a Dec. 15 briefing that the variant accounts for 20% of viruses sequenced in Norfolk, 10% in Essex, and 3% in Suffolk. “There are no data to suggest it had been imported from abroad, so it is likely to have evolved in the U.K.,” he said.
The variant is named VUI-202012/01, for the first “variant under investigation” in December 2020, BMJ says. It’s defined by a set of 17 mutations, with the most significant mutation in the spike protein the virus uses to bind to the human ACE2 receptor.
“Changes in this part of spike protein may, in theory, result in the virus becoming more infectious and spreading more easily between people,” the article says.
The European Centre for Disease Prevention and Control says the variant emerged during the time of year when people usually socialize more.
“There is no indication at this point of increased infection severity associated with the new variant,” the agency said. “A few cases with the new variant have to date been reported by Denmark and the Netherlands and, according to media reports, in Belgium.”
Mr. Johnson announced tighter restrictions on England’s hardest-hit areas, such as the southeast and east of England, where new coronavirus cases have continued to rise. And he said people must cut back on their Christmas socializing.
“In England, those living in tier 4 areas should not mix with anyone outside their own household at Christmas, though support bubbles will remain in place for those at particular risk of loneliness or isolation,” he said.
A version of this article first appeared on WebMD.com.
Researchers have detected a highly contagious coronavirus variant in the United Kingdom, leading Prime Minister Boris Johnson to shut down parts of the country and triggering other nations to impose travel and shipping restrictions on England.
Mr. Johnson held a crisis meeting with ministers Monday after Saturday’s shutdown announcement. The prime minister said in a nationally televised address that this coronavirus variant may be “up to 70% more transmissible than the old variant” and was probably responsible for an increase in cases in southeastern England.
“There is still much we don’t know. While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant,” he said.
Public Health England says it is working to learn as much about the variant as possible. “We know that mortality is a lagging indicator, and we will need to continually monitor this over the coming weeks,” the agency says.
That scientific uncertainty about the variant’s threat shook European nations that were rushing to ship goods to England in advance of a Dec. 31 Brexit deadline. Under Brexit, which is short for “British exit,” the United Kingdom will leave the European Union on Jan. 31, 2020. Until then, the two sides will come up with new trade and security relationships.
European Union members Austria, Belgium, Bulgaria, France, Germany, Ireland, Italy, and the Netherlands announced travel restrictions hours after Johnson’s speech.
Those restrictions created food uncertainty across the U.K., which imports about a quarter of its food from the EU, according to The New York Times. Long lines of trucks heading to ports in the U.K. came to a standstill on major roads such as the M20 near Kent and the Port of Dover.
Outside Europe, Canada, India, Iran, Israel, Hong Kong, Saudi Arabia, and Turkey banned all incoming flights from the U.K. And more bans could come.
The U.S. reaction
The United States has not imposed any new limits on travel with the United Kingdom, although New York Gov. Andrew Cuomo (D) has requested all passengers bound for John F. Kennedy International Airport from the U.K. be tested before boarding and a new travel ban be placed for Europe. He says the federal government must take action now to avoid a crisis situation like the one New York experienced in March and April.
“The United States has a number of flights coming in from the U.K. each day, and we have done absolutely nothing,” Mr. Cuomo said in a statement on the governor’s webpage. “To me, this is reprehensible because this is what happened in the spring. How many times in life do you have to make the same mistake before you learn?”
Leading U.S. health officials have downplayed the dangers of the virus.
“We don’t know that it’s more dangerous, and very importantly, we have not seen a single mutation yet that would make it evade the vaccine,” U.S. Assistant Secretary of Health and Human Services Adm. Brett Giroir, MD, said Sunday on ABC’s This Week with George Stephanopoulos. “I can’t say that won’t happen in the future, but right now it looks like the vaccine will cover everything that we see.”
Dr. Giroir said the HHS and other U.S. government agencies will monitor the variant.
“Viruses mutate,” he said. “We’ve seen almost 4,000 different mutations among this virus. There is no indication that the mutation right now that they’re talking about is overcoming England.”
Where did the variant come from?
Public Health England says the coronavirus variant had existed in the U.K. since September and circulated at very low levels until mid-November.
“The increase in cases linked to the new variant first came to light in late November when PHE was investigating why infection rates in Kent were not falling despite national restrictions. We then discovered a cluster linked to this variant spreading rapidly into London and Essex,” the agency said.
Public Health England says there’s no evidence the new variant is resistant to the Pfizer-BioNTech vaccine, which is now being given across the country to high-priority groups such as health care workers.
An article in The BMJ, a British medical journal, says the variant was first detected by Covid-19 Genomics UK, a consortium that tests the random genetic sequencing of positive COVID-19 samples around the U.K. The variant cases were mostly in the southeast of England.
A University of Birmingham professor said in a Dec. 15 briefing that the variant accounts for 20% of viruses sequenced in Norfolk, 10% in Essex, and 3% in Suffolk. “There are no data to suggest it had been imported from abroad, so it is likely to have evolved in the U.K.,” he said.
The variant is named VUI-202012/01, for the first “variant under investigation” in December 2020, BMJ says. It’s defined by a set of 17 mutations, with the most significant mutation in the spike protein the virus uses to bind to the human ACE2 receptor.
“Changes in this part of spike protein may, in theory, result in the virus becoming more infectious and spreading more easily between people,” the article says.
The European Centre for Disease Prevention and Control says the variant emerged during the time of year when people usually socialize more.
“There is no indication at this point of increased infection severity associated with the new variant,” the agency said. “A few cases with the new variant have to date been reported by Denmark and the Netherlands and, according to media reports, in Belgium.”
Mr. Johnson announced tighter restrictions on England’s hardest-hit areas, such as the southeast and east of England, where new coronavirus cases have continued to rise. And he said people must cut back on their Christmas socializing.
“In England, those living in tier 4 areas should not mix with anyone outside their own household at Christmas, though support bubbles will remain in place for those at particular risk of loneliness or isolation,” he said.
A version of this article first appeared on WebMD.com.
Researchers have detected a highly contagious coronavirus variant in the United Kingdom, leading Prime Minister Boris Johnson to shut down parts of the country and triggering other nations to impose travel and shipping restrictions on England.
Mr. Johnson held a crisis meeting with ministers Monday after Saturday’s shutdown announcement. The prime minister said in a nationally televised address that this coronavirus variant may be “up to 70% more transmissible than the old variant” and was probably responsible for an increase in cases in southeastern England.
“There is still much we don’t know. While we are fairly certain the variant is transmitted more quickly, there is no evidence to suggest that it is more lethal or causes more severe illness. Equally there is no evidence to suggest the vaccine will be any less effective against the new variant,” he said.
Public Health England says it is working to learn as much about the variant as possible. “We know that mortality is a lagging indicator, and we will need to continually monitor this over the coming weeks,” the agency says.
That scientific uncertainty about the variant’s threat shook European nations that were rushing to ship goods to England in advance of a Dec. 31 Brexit deadline. Under Brexit, which is short for “British exit,” the United Kingdom will leave the European Union on Jan. 31, 2020. Until then, the two sides will come up with new trade and security relationships.
European Union members Austria, Belgium, Bulgaria, France, Germany, Ireland, Italy, and the Netherlands announced travel restrictions hours after Johnson’s speech.
Those restrictions created food uncertainty across the U.K., which imports about a quarter of its food from the EU, according to The New York Times. Long lines of trucks heading to ports in the U.K. came to a standstill on major roads such as the M20 near Kent and the Port of Dover.
Outside Europe, Canada, India, Iran, Israel, Hong Kong, Saudi Arabia, and Turkey banned all incoming flights from the U.K. And more bans could come.
The U.S. reaction
The United States has not imposed any new limits on travel with the United Kingdom, although New York Gov. Andrew Cuomo (D) has requested all passengers bound for John F. Kennedy International Airport from the U.K. be tested before boarding and a new travel ban be placed for Europe. He says the federal government must take action now to avoid a crisis situation like the one New York experienced in March and April.
“The United States has a number of flights coming in from the U.K. each day, and we have done absolutely nothing,” Mr. Cuomo said in a statement on the governor’s webpage. “To me, this is reprehensible because this is what happened in the spring. How many times in life do you have to make the same mistake before you learn?”
Leading U.S. health officials have downplayed the dangers of the virus.
“We don’t know that it’s more dangerous, and very importantly, we have not seen a single mutation yet that would make it evade the vaccine,” U.S. Assistant Secretary of Health and Human Services Adm. Brett Giroir, MD, said Sunday on ABC’s This Week with George Stephanopoulos. “I can’t say that won’t happen in the future, but right now it looks like the vaccine will cover everything that we see.”
Dr. Giroir said the HHS and other U.S. government agencies will monitor the variant.
“Viruses mutate,” he said. “We’ve seen almost 4,000 different mutations among this virus. There is no indication that the mutation right now that they’re talking about is overcoming England.”
Where did the variant come from?
Public Health England says the coronavirus variant had existed in the U.K. since September and circulated at very low levels until mid-November.
“The increase in cases linked to the new variant first came to light in late November when PHE was investigating why infection rates in Kent were not falling despite national restrictions. We then discovered a cluster linked to this variant spreading rapidly into London and Essex,” the agency said.
Public Health England says there’s no evidence the new variant is resistant to the Pfizer-BioNTech vaccine, which is now being given across the country to high-priority groups such as health care workers.
An article in The BMJ, a British medical journal, says the variant was first detected by Covid-19 Genomics UK, a consortium that tests the random genetic sequencing of positive COVID-19 samples around the U.K. The variant cases were mostly in the southeast of England.
A University of Birmingham professor said in a Dec. 15 briefing that the variant accounts for 20% of viruses sequenced in Norfolk, 10% in Essex, and 3% in Suffolk. “There are no data to suggest it had been imported from abroad, so it is likely to have evolved in the U.K.,” he said.
The variant is named VUI-202012/01, for the first “variant under investigation” in December 2020, BMJ says. It’s defined by a set of 17 mutations, with the most significant mutation in the spike protein the virus uses to bind to the human ACE2 receptor.
“Changes in this part of spike protein may, in theory, result in the virus becoming more infectious and spreading more easily between people,” the article says.
The European Centre for Disease Prevention and Control says the variant emerged during the time of year when people usually socialize more.
“There is no indication at this point of increased infection severity associated with the new variant,” the agency said. “A few cases with the new variant have to date been reported by Denmark and the Netherlands and, according to media reports, in Belgium.”
Mr. Johnson announced tighter restrictions on England’s hardest-hit areas, such as the southeast and east of England, where new coronavirus cases have continued to rise. And he said people must cut back on their Christmas socializing.
“In England, those living in tier 4 areas should not mix with anyone outside their own household at Christmas, though support bubbles will remain in place for those at particular risk of loneliness or isolation,” he said.
A version of this article first appeared on WebMD.com.
New coalition demands urgent action on COVID-19 mental health crisis
Fourteen mental health organizations have formed a coalition to press federal and state officials to tackle the ongoing and growing mental health crisis that is accompanying the COVID-19 pandemic.
The coalition is offering a road map, A Unified Vision for Transforming Mental Health and Substance Abuse Care, which spells out “immediate and long-term changes that will lead to a mental health care system capable of saving our nation,” they said in a statement.
The group includes CEOs from the American Psychiatric Association, the American Psychological Association, the Massachusetts Association for Mental Health, Meadows Mental Health Policy Institute, Mental Health America, the National Association for Behavioral Healthcare, the National Alliance on Mental Illness, the National Council for Behavioral Health, One Mind, Peg’s Foundation, the Steinberg Institute, The Kennedy Forum, the Treatment Advocacy Center, and the Well Being Trust.
They have been meeting in weekly sessions since the beginning of the pandemic. The groups have come together in the spirit of previous efforts to address major health crises, including the 1970s war on cancer and the campaign to curtail the HIV/AIDS epidemic in the 1980s, they report.
The coalition reported that since the pandemic began the prevalence of depression symptoms has jumped threefold, overdose deaths have increased in 40 states, and 25% of young adults have had suicidal ideation.
“It requires immediate action by the new administration, as well as state and local governments in all 50 states, and an acknowledged, consistent commitment to fix what’s broken in our system of care,” Daniel H. Gillison Jr, CEO of the National Alliance on Mental Illness, said in a statement.
SAMHSA chief ‘grateful’
Elinore McCance-Katz, MD, PhD, who is the assistant secretary for mental health and substance use and leads the Substance Abuse and Mental Health Services Administration, U.S. Department of Health & Human Services, applauded the coalition.
“I am very grateful that these organizations are stepping up and putting out a report like this,” Dr. McCance-Katz told this news organization. “I hope that they will continue this kind of advocacy and leadership on these issues going forward,” she said, adding that the need for mental health care and substance use disorders will be much greater going forward because of the pandemic.
Seven policy areas
The group’s 17-page strategic plan emphasizes interventions and methods that have already been tried and tested, focusing on seven policy areas:
- Early identification and prevention, especially for families and young people, by, for instance, bringing telehealth into schools and community centers.
- Rapid deployment of emergency crisis response and prevention, including speeding up the implementation of the new 988 number for the National Suicide Prevention Lifeline.
- Leveling inequities in access to care by addressing social and political constructs and historical systemic injustices such as racism.
- Integrating physical and mental health care and substance use services to ensure “whole-person” well-being.
- Achieving parity in payment by health plans for mental health and substance-use coverage.
- Assuring evidence-based standards of treatments and care.
- Increasing the number and diversity of the mental health care workforce, peer support, and community-based programs.
and will be even more so in the near future as the effects of the pandemic continue to ripple out.
SAMHSA received $425 million in the first COVID-19 relief package signed into law in March – the CARES Act. The money was distributed to states and used for direct care for people with serious mental illness and substance-use disorders who could not otherwise get care because of virus-related restrictions, and for boosting support for mental health support lines, said Dr. McCance-Katz.
A senior SAMHSA spokesperson said the agency is “hopeful that we will see additional resources in the upcoming stimulus for mental health and substance abuse” that Congress is still working on.
“We need bold steps from our government and the business community alike,” former Rep. Patrick J. Kennedy, founder of The Kennedy Forum, said in the statement from the new coalition. “We encourage all state governments to engage with mental health leaders, bring them into pandemic-related responses, and actively facilitate their communication with communities across the country,” said Mr. Kennedy, who is a part of the new coalition.
Mr. Kennedy is also cochair of the Action Alliance’s Mental Health and Suicide Prevention National Response to COVID-19, which unveiled its own six-priority Action Plan earlier in December.
A version of this article first appeared on Medscape.com.
Fourteen mental health organizations have formed a coalition to press federal and state officials to tackle the ongoing and growing mental health crisis that is accompanying the COVID-19 pandemic.
The coalition is offering a road map, A Unified Vision for Transforming Mental Health and Substance Abuse Care, which spells out “immediate and long-term changes that will lead to a mental health care system capable of saving our nation,” they said in a statement.
The group includes CEOs from the American Psychiatric Association, the American Psychological Association, the Massachusetts Association for Mental Health, Meadows Mental Health Policy Institute, Mental Health America, the National Association for Behavioral Healthcare, the National Alliance on Mental Illness, the National Council for Behavioral Health, One Mind, Peg’s Foundation, the Steinberg Institute, The Kennedy Forum, the Treatment Advocacy Center, and the Well Being Trust.
They have been meeting in weekly sessions since the beginning of the pandemic. The groups have come together in the spirit of previous efforts to address major health crises, including the 1970s war on cancer and the campaign to curtail the HIV/AIDS epidemic in the 1980s, they report.
The coalition reported that since the pandemic began the prevalence of depression symptoms has jumped threefold, overdose deaths have increased in 40 states, and 25% of young adults have had suicidal ideation.
“It requires immediate action by the new administration, as well as state and local governments in all 50 states, and an acknowledged, consistent commitment to fix what’s broken in our system of care,” Daniel H. Gillison Jr, CEO of the National Alliance on Mental Illness, said in a statement.
SAMHSA chief ‘grateful’
Elinore McCance-Katz, MD, PhD, who is the assistant secretary for mental health and substance use and leads the Substance Abuse and Mental Health Services Administration, U.S. Department of Health & Human Services, applauded the coalition.
“I am very grateful that these organizations are stepping up and putting out a report like this,” Dr. McCance-Katz told this news organization. “I hope that they will continue this kind of advocacy and leadership on these issues going forward,” she said, adding that the need for mental health care and substance use disorders will be much greater going forward because of the pandemic.
Seven policy areas
The group’s 17-page strategic plan emphasizes interventions and methods that have already been tried and tested, focusing on seven policy areas:
- Early identification and prevention, especially for families and young people, by, for instance, bringing telehealth into schools and community centers.
- Rapid deployment of emergency crisis response and prevention, including speeding up the implementation of the new 988 number for the National Suicide Prevention Lifeline.
- Leveling inequities in access to care by addressing social and political constructs and historical systemic injustices such as racism.
- Integrating physical and mental health care and substance use services to ensure “whole-person” well-being.
- Achieving parity in payment by health plans for mental health and substance-use coverage.
- Assuring evidence-based standards of treatments and care.
- Increasing the number and diversity of the mental health care workforce, peer support, and community-based programs.
and will be even more so in the near future as the effects of the pandemic continue to ripple out.
SAMHSA received $425 million in the first COVID-19 relief package signed into law in March – the CARES Act. The money was distributed to states and used for direct care for people with serious mental illness and substance-use disorders who could not otherwise get care because of virus-related restrictions, and for boosting support for mental health support lines, said Dr. McCance-Katz.
A senior SAMHSA spokesperson said the agency is “hopeful that we will see additional resources in the upcoming stimulus for mental health and substance abuse” that Congress is still working on.
“We need bold steps from our government and the business community alike,” former Rep. Patrick J. Kennedy, founder of The Kennedy Forum, said in the statement from the new coalition. “We encourage all state governments to engage with mental health leaders, bring them into pandemic-related responses, and actively facilitate their communication with communities across the country,” said Mr. Kennedy, who is a part of the new coalition.
Mr. Kennedy is also cochair of the Action Alliance’s Mental Health and Suicide Prevention National Response to COVID-19, which unveiled its own six-priority Action Plan earlier in December.
A version of this article first appeared on Medscape.com.
Fourteen mental health organizations have formed a coalition to press federal and state officials to tackle the ongoing and growing mental health crisis that is accompanying the COVID-19 pandemic.
The coalition is offering a road map, A Unified Vision for Transforming Mental Health and Substance Abuse Care, which spells out “immediate and long-term changes that will lead to a mental health care system capable of saving our nation,” they said in a statement.
The group includes CEOs from the American Psychiatric Association, the American Psychological Association, the Massachusetts Association for Mental Health, Meadows Mental Health Policy Institute, Mental Health America, the National Association for Behavioral Healthcare, the National Alliance on Mental Illness, the National Council for Behavioral Health, One Mind, Peg’s Foundation, the Steinberg Institute, The Kennedy Forum, the Treatment Advocacy Center, and the Well Being Trust.
They have been meeting in weekly sessions since the beginning of the pandemic. The groups have come together in the spirit of previous efforts to address major health crises, including the 1970s war on cancer and the campaign to curtail the HIV/AIDS epidemic in the 1980s, they report.
The coalition reported that since the pandemic began the prevalence of depression symptoms has jumped threefold, overdose deaths have increased in 40 states, and 25% of young adults have had suicidal ideation.
“It requires immediate action by the new administration, as well as state and local governments in all 50 states, and an acknowledged, consistent commitment to fix what’s broken in our system of care,” Daniel H. Gillison Jr, CEO of the National Alliance on Mental Illness, said in a statement.
SAMHSA chief ‘grateful’
Elinore McCance-Katz, MD, PhD, who is the assistant secretary for mental health and substance use and leads the Substance Abuse and Mental Health Services Administration, U.S. Department of Health & Human Services, applauded the coalition.
“I am very grateful that these organizations are stepping up and putting out a report like this,” Dr. McCance-Katz told this news organization. “I hope that they will continue this kind of advocacy and leadership on these issues going forward,” she said, adding that the need for mental health care and substance use disorders will be much greater going forward because of the pandemic.
Seven policy areas
The group’s 17-page strategic plan emphasizes interventions and methods that have already been tried and tested, focusing on seven policy areas:
- Early identification and prevention, especially for families and young people, by, for instance, bringing telehealth into schools and community centers.
- Rapid deployment of emergency crisis response and prevention, including speeding up the implementation of the new 988 number for the National Suicide Prevention Lifeline.
- Leveling inequities in access to care by addressing social and political constructs and historical systemic injustices such as racism.
- Integrating physical and mental health care and substance use services to ensure “whole-person” well-being.
- Achieving parity in payment by health plans for mental health and substance-use coverage.
- Assuring evidence-based standards of treatments and care.
- Increasing the number and diversity of the mental health care workforce, peer support, and community-based programs.
and will be even more so in the near future as the effects of the pandemic continue to ripple out.
SAMHSA received $425 million in the first COVID-19 relief package signed into law in March – the CARES Act. The money was distributed to states and used for direct care for people with serious mental illness and substance-use disorders who could not otherwise get care because of virus-related restrictions, and for boosting support for mental health support lines, said Dr. McCance-Katz.
A senior SAMHSA spokesperson said the agency is “hopeful that we will see additional resources in the upcoming stimulus for mental health and substance abuse” that Congress is still working on.
“We need bold steps from our government and the business community alike,” former Rep. Patrick J. Kennedy, founder of The Kennedy Forum, said in the statement from the new coalition. “We encourage all state governments to engage with mental health leaders, bring them into pandemic-related responses, and actively facilitate their communication with communities across the country,” said Mr. Kennedy, who is a part of the new coalition.
Mr. Kennedy is also cochair of the Action Alliance’s Mental Health and Suicide Prevention National Response to COVID-19, which unveiled its own six-priority Action Plan earlier in December.
A version of this article first appeared on Medscape.com.