User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Heart failure: Medicare cost sharing may put quadruple therapy out of reach
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.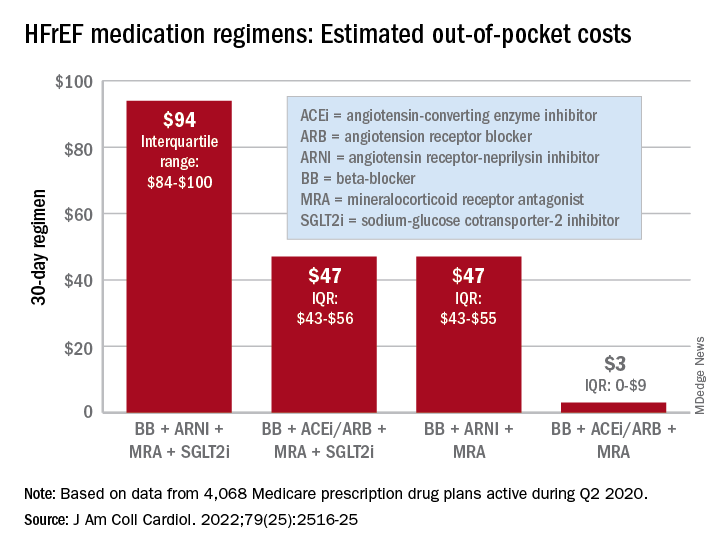
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
Out-of-pocket (OOP) costs for Medicare enrollees receiving quadruple drug therapy for heart failure with reduced ejection fraction were “substantially higher than regimens limited to generically available medications,” according to a new analysis of prescription drug plans.
“Despite the clinical benefit of quadruple therapy” consisting of beta-blockers, angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), and sodium-glucose cotransporter-2 (SGLT2) inhibitors, “coverage was restricted primarily through cost sharing, and estimated annual OOP costs for beneficiaries were [over $2,000] per year under most plans,” wrote Kamil F. Faridi, MD, and associates. The findings were published in the Journal of the American College of Cardiology.
For just 1 month of quadruple drug therapy for heart failure with reduced ejection fraction (HFrEF), the estimated median OOP cost was $94 for individuals covered by a Medicare prescription drug plan during the second quarter of 2020, with the majority coming from the ARNI (median, $47) and the SGLT2 inhibitor (median, $45). Alternative HFrEF regimens were significantly less costly, ranging from $3 to $47 OOP, the investigators reported.
Almost all of the 4,068 plans participating in Medicare at that time covered quadruple therapy for HFrEF, but more than 99% restricted coverage by instituting cost sharing for medications at tier level 3 and above on the drug formularies. Such restrictions for ARNIs and SGLT2 inhibitors “might not be readily apparent to prescribing physicians,” wrote Dr. Faridi of Yale University, New Haven, Conn., and associates.
Other methods of regulating coverage were less common. Prior authorization of ARNIs was invoked by about a quarter of the plans, but none required authorization for any of the other drugs involved, and few plans used step therapy-requirements involving lower-cost alternatives, they noted.
“The use of cost sharing restricts access through high OOP costs for patients. Furthermore, these policies likely disadvantage relatively poorer patients (although the poorest Medicare patients will tend to be dual-enrolled in Medicaid and protected from cost sharing),” Jason H. Wasfy, MD, and Anna C. O’Kelly, MD, said in an accompanying editorial comment .
Since acceptable cost-effectiveness has been demonstrated for dapagliflozin, an SGLT1 inhibitor, and for the ARNIs, and because these medications have no generic equivalents, health plans should “use the discretion they have under Medicare Part D to reduce cost sharing for patients with HFrEF,” Dr. Wasfy and Dr. O’Kelly wrote, adding that the current study “demonstrates that without consensus on cost effectiveness from the societal perspective, costs can be imposed directly on patients in ways that slow uptake of cost-effective drugs.”
Data for all Medicare Advantage plans (n = 3,167) and standalone Part D plans (n = 901) came from the Medicare Prescription Drug Plan Formulary and Pricing Information Files. Annual OOP costs were estimated “using each phase of a 2020 Medicare part D standard benefit,” including deductible, standard coverage, coverage gap, and catastrophic coverage, the investigators explained.
Dr. Faridi and associates did not report any direct funding sources for their study. Dr Faridi received a grant from the National Institutes of Health outside the scope of the present work, and other investigators disclosed ties to the Food and Drug Administration, the Centers for Medicare and Medicaid Services, Johnson & Johnson, AstraZeneca, Boehringer Ingelheim, Amgen, Cytokinetics, and the Institute for Clinical and Economic Review.
Dr. Wasfy is supported by the American Heart Association and has received consulting fees from Pfizer and honoraria from the Institute for Clinical and Economic Review. Dr. O’Kelly has no relevant disclosures.
FROM THE JOURNAL Of the AMERICAN COLLEGE OF CARDIOLOGY
How we treat acute pain could be wrong
In a surprising discovery that flies in the face of conventional medicine,
The paper, published in Science Translational Medicine, suggests that inflammation, a normal part of injury recovery, helps resolve acute pain and prevents it from becoming chronic. Blocking that inflammation may interfere with this process, leading to harder-to-treat pain.
“What we’ve been doing for decades not only appears to be wrong, but appears to be 180 degrees wrong,” says senior study author Jeffrey Mogil, PhD, a professor in the department of psychology at McGill University in Montreal. “You should not be blocking inflammation. You should be letting inflammation happen. That’s what stops chronic pain.”
Inflammation: Nature’s pain reliever
Wanting to know why pain goes away for some but drags on (and on) for others, the researchers looked at pain mechanisms in both humans and mice. They found that a type of white blood cell known as a neutrophil seems to play a key role.
“In analyzing the genes of people suffering from lower back pain, we observed active changes in genes over time in people whose pain went away,” says Luda Diatchenko, PhD, a professor in the faculty of medicine and Canada excellence research chair in human pain genetics at McGill. “Changes in the blood cells and their activity seemed to be the most important factor, especially in cells called neutrophils.”
To test this link, the researchers blocked neutrophils in mice and found the pain lasted 2-10 times longer than normal. Anti-inflammatory drugs, despite providing short-term relief, had the same pain-prolonging effect – though injecting neutrophils into the mice seemed to keep that from happening.
The findings are supported by a separate analysis of 500,000 people in the United Kingdom that showed those taking anti-inflammatory drugs to treat their pain were more likely to have pain 2-10 years later.
“Inflammation occurs for a reason,” says Dr. Mogil, “and it looks like it’s dangerous to interfere with it.”
Rethinking how we treat pain
Neutrophils arrive early during inflammation, at the onset of injury – just when many of us reach for pain medication. This research suggests it might be better not to block inflammation, instead letting the neutrophils “do their thing.” Taking an analgesic that alleviates pain without blocking neutrophils, like acetaminophen, may be better than taking an anti-inflammatory drug or steroid, says Dr. Mogil.
Still, while the findings are compelling, clinical trials are needed to directly compare anti-inflammatory drugs to other painkillers, the researchers said. This research may also lay the groundwork for new drug development for chronic pain patients, Dr. Mogil says.
“Our data strongly suggests that neutrophils act like analgesics themselves, which is potentially useful in terms of analgesic development,” Dr. Mogil says. “And of course, we need new analgesics.”
A version of this article first appeared on WebMD.com.
In a surprising discovery that flies in the face of conventional medicine,
The paper, published in Science Translational Medicine, suggests that inflammation, a normal part of injury recovery, helps resolve acute pain and prevents it from becoming chronic. Blocking that inflammation may interfere with this process, leading to harder-to-treat pain.
“What we’ve been doing for decades not only appears to be wrong, but appears to be 180 degrees wrong,” says senior study author Jeffrey Mogil, PhD, a professor in the department of psychology at McGill University in Montreal. “You should not be blocking inflammation. You should be letting inflammation happen. That’s what stops chronic pain.”
Inflammation: Nature’s pain reliever
Wanting to know why pain goes away for some but drags on (and on) for others, the researchers looked at pain mechanisms in both humans and mice. They found that a type of white blood cell known as a neutrophil seems to play a key role.
“In analyzing the genes of people suffering from lower back pain, we observed active changes in genes over time in people whose pain went away,” says Luda Diatchenko, PhD, a professor in the faculty of medicine and Canada excellence research chair in human pain genetics at McGill. “Changes in the blood cells and their activity seemed to be the most important factor, especially in cells called neutrophils.”
To test this link, the researchers blocked neutrophils in mice and found the pain lasted 2-10 times longer than normal. Anti-inflammatory drugs, despite providing short-term relief, had the same pain-prolonging effect – though injecting neutrophils into the mice seemed to keep that from happening.
The findings are supported by a separate analysis of 500,000 people in the United Kingdom that showed those taking anti-inflammatory drugs to treat their pain were more likely to have pain 2-10 years later.
“Inflammation occurs for a reason,” says Dr. Mogil, “and it looks like it’s dangerous to interfere with it.”
Rethinking how we treat pain
Neutrophils arrive early during inflammation, at the onset of injury – just when many of us reach for pain medication. This research suggests it might be better not to block inflammation, instead letting the neutrophils “do their thing.” Taking an analgesic that alleviates pain without blocking neutrophils, like acetaminophen, may be better than taking an anti-inflammatory drug or steroid, says Dr. Mogil.
Still, while the findings are compelling, clinical trials are needed to directly compare anti-inflammatory drugs to other painkillers, the researchers said. This research may also lay the groundwork for new drug development for chronic pain patients, Dr. Mogil says.
“Our data strongly suggests that neutrophils act like analgesics themselves, which is potentially useful in terms of analgesic development,” Dr. Mogil says. “And of course, we need new analgesics.”
A version of this article first appeared on WebMD.com.
In a surprising discovery that flies in the face of conventional medicine,
The paper, published in Science Translational Medicine, suggests that inflammation, a normal part of injury recovery, helps resolve acute pain and prevents it from becoming chronic. Blocking that inflammation may interfere with this process, leading to harder-to-treat pain.
“What we’ve been doing for decades not only appears to be wrong, but appears to be 180 degrees wrong,” says senior study author Jeffrey Mogil, PhD, a professor in the department of psychology at McGill University in Montreal. “You should not be blocking inflammation. You should be letting inflammation happen. That’s what stops chronic pain.”
Inflammation: Nature’s pain reliever
Wanting to know why pain goes away for some but drags on (and on) for others, the researchers looked at pain mechanisms in both humans and mice. They found that a type of white blood cell known as a neutrophil seems to play a key role.
“In analyzing the genes of people suffering from lower back pain, we observed active changes in genes over time in people whose pain went away,” says Luda Diatchenko, PhD, a professor in the faculty of medicine and Canada excellence research chair in human pain genetics at McGill. “Changes in the blood cells and their activity seemed to be the most important factor, especially in cells called neutrophils.”
To test this link, the researchers blocked neutrophils in mice and found the pain lasted 2-10 times longer than normal. Anti-inflammatory drugs, despite providing short-term relief, had the same pain-prolonging effect – though injecting neutrophils into the mice seemed to keep that from happening.
The findings are supported by a separate analysis of 500,000 people in the United Kingdom that showed those taking anti-inflammatory drugs to treat their pain were more likely to have pain 2-10 years later.
“Inflammation occurs for a reason,” says Dr. Mogil, “and it looks like it’s dangerous to interfere with it.”
Rethinking how we treat pain
Neutrophils arrive early during inflammation, at the onset of injury – just when many of us reach for pain medication. This research suggests it might be better not to block inflammation, instead letting the neutrophils “do their thing.” Taking an analgesic that alleviates pain without blocking neutrophils, like acetaminophen, may be better than taking an anti-inflammatory drug or steroid, says Dr. Mogil.
Still, while the findings are compelling, clinical trials are needed to directly compare anti-inflammatory drugs to other painkillers, the researchers said. This research may also lay the groundwork for new drug development for chronic pain patients, Dr. Mogil says.
“Our data strongly suggests that neutrophils act like analgesics themselves, which is potentially useful in terms of analgesic development,” Dr. Mogil says. “And of course, we need new analgesics.”
A version of this article first appeared on WebMD.com.
FROM SCIENCE TRANSLATIONAL MEDICINE
Bariatric surgery can be a tool to relieve migraine
DENVER – , a neurologist told colleagues at the annual meeting of the American Headache Society. “There’s evidence-based medicine to support bariatric surgery, a lot of it, and the outcomes are actually pretty good,” said Jennifer McVige, MD, MA, of Dent Neurologic Institute in Buffalo, N.Y.
As Dr. McVige noted, research has linked obesity to migraine even after adjustment for comorbidities. A 2007 analysis of a survey of 30,215 participants, for example, found that “the proportion of subjects with severe headache pain increased with BMI, doubling in the morbidly obese relative to the normally weighted (odds ratio [OR] = 1.9).” And a 2011 study of 3,733 pregnant women found that risk of migraine increased in line with level of obesity: “obese women had a 1.48-fold increased odds of migraine (OR = 1.48; 95% confidence interval [CI], 1.12-1.96). Severely obese (OR = 2.07; 95% CI, 1.27-3.39) and morbidly obese (OR = 2.75; 95% CI, 1.60-4.70) had the highest odds of migraines.”
The link between obesity and headaches is unclear, she said, but there are hints at possible factors. For one, calcitonin gene-related peptide (CGRP) is increased in people with obesity and is an important factor in migraines. Additionally, nausea is quite common in people with migraine, suggesting a possible gut-brain interaction – or not.
“Nausea is associated with a lot of the medicines that we give patients with migraine. Is it the nausea that’s associated with the migraine medicine, or is nausea occurring at the end of the migraine?” she asked. “That’s always been kind of a conundrum for us.”
Whatever the case, she said, bariatric surgery appears to be helpful for patients with headache. Some studies have been small, but a 2021 analysis of 1,680 patients with migraine found that 55% experienced remission with no need for medication at 180 days post surgery. Women, older patients, and those taking more migraine medications were less likely to reach remission.
Research also suggests that bariatric surgery can relieve headache symptoms in patients with idiopathic intracranial hypertension.
Dr. McVige cautioned, however, that medical professionals must take special care when they talk to patients about their weight. “I’ve learned from conversations with my patients that they don’t like hearing ‘obese,’ or ‘fat,’ or ‘diet,’ or ‘losing weight.’ What they do like is ‘maybe we could try to find ways to be more healthy, to help your body to look the way that you would like it to look in the future. Let me help you. Maybe we can talk about nutrition. Maybe we can talk about exercise. Let’s talk about energy. Let’s talk about those types of things.’”
Unfortunately, there’s little research into how to have these conversations, Dr. McVige said. Still, “we need to be the first people to come forward and say, ‘This is a tough topic for me to talk about too. It makes me uncomfortable as well. I know you’re uncomfortable, but I have to talk to you because I care about you as a patient.’”
She also advised colleagues to not bring up weight right out of the gate. “It’s easier to say some of those things after you develop a relationship,” she said, “when they know you care about them.”
Dr. McVige reported multiple disclosures related to research funding and speaker fees.
DENVER – , a neurologist told colleagues at the annual meeting of the American Headache Society. “There’s evidence-based medicine to support bariatric surgery, a lot of it, and the outcomes are actually pretty good,” said Jennifer McVige, MD, MA, of Dent Neurologic Institute in Buffalo, N.Y.
As Dr. McVige noted, research has linked obesity to migraine even after adjustment for comorbidities. A 2007 analysis of a survey of 30,215 participants, for example, found that “the proportion of subjects with severe headache pain increased with BMI, doubling in the morbidly obese relative to the normally weighted (odds ratio [OR] = 1.9).” And a 2011 study of 3,733 pregnant women found that risk of migraine increased in line with level of obesity: “obese women had a 1.48-fold increased odds of migraine (OR = 1.48; 95% confidence interval [CI], 1.12-1.96). Severely obese (OR = 2.07; 95% CI, 1.27-3.39) and morbidly obese (OR = 2.75; 95% CI, 1.60-4.70) had the highest odds of migraines.”
The link between obesity and headaches is unclear, she said, but there are hints at possible factors. For one, calcitonin gene-related peptide (CGRP) is increased in people with obesity and is an important factor in migraines. Additionally, nausea is quite common in people with migraine, suggesting a possible gut-brain interaction – or not.
“Nausea is associated with a lot of the medicines that we give patients with migraine. Is it the nausea that’s associated with the migraine medicine, or is nausea occurring at the end of the migraine?” she asked. “That’s always been kind of a conundrum for us.”
Whatever the case, she said, bariatric surgery appears to be helpful for patients with headache. Some studies have been small, but a 2021 analysis of 1,680 patients with migraine found that 55% experienced remission with no need for medication at 180 days post surgery. Women, older patients, and those taking more migraine medications were less likely to reach remission.
Research also suggests that bariatric surgery can relieve headache symptoms in patients with idiopathic intracranial hypertension.
Dr. McVige cautioned, however, that medical professionals must take special care when they talk to patients about their weight. “I’ve learned from conversations with my patients that they don’t like hearing ‘obese,’ or ‘fat,’ or ‘diet,’ or ‘losing weight.’ What they do like is ‘maybe we could try to find ways to be more healthy, to help your body to look the way that you would like it to look in the future. Let me help you. Maybe we can talk about nutrition. Maybe we can talk about exercise. Let’s talk about energy. Let’s talk about those types of things.’”
Unfortunately, there’s little research into how to have these conversations, Dr. McVige said. Still, “we need to be the first people to come forward and say, ‘This is a tough topic for me to talk about too. It makes me uncomfortable as well. I know you’re uncomfortable, but I have to talk to you because I care about you as a patient.’”
She also advised colleagues to not bring up weight right out of the gate. “It’s easier to say some of those things after you develop a relationship,” she said, “when they know you care about them.”
Dr. McVige reported multiple disclosures related to research funding and speaker fees.
DENVER – , a neurologist told colleagues at the annual meeting of the American Headache Society. “There’s evidence-based medicine to support bariatric surgery, a lot of it, and the outcomes are actually pretty good,” said Jennifer McVige, MD, MA, of Dent Neurologic Institute in Buffalo, N.Y.
As Dr. McVige noted, research has linked obesity to migraine even after adjustment for comorbidities. A 2007 analysis of a survey of 30,215 participants, for example, found that “the proportion of subjects with severe headache pain increased with BMI, doubling in the morbidly obese relative to the normally weighted (odds ratio [OR] = 1.9).” And a 2011 study of 3,733 pregnant women found that risk of migraine increased in line with level of obesity: “obese women had a 1.48-fold increased odds of migraine (OR = 1.48; 95% confidence interval [CI], 1.12-1.96). Severely obese (OR = 2.07; 95% CI, 1.27-3.39) and morbidly obese (OR = 2.75; 95% CI, 1.60-4.70) had the highest odds of migraines.”
The link between obesity and headaches is unclear, she said, but there are hints at possible factors. For one, calcitonin gene-related peptide (CGRP) is increased in people with obesity and is an important factor in migraines. Additionally, nausea is quite common in people with migraine, suggesting a possible gut-brain interaction – or not.
“Nausea is associated with a lot of the medicines that we give patients with migraine. Is it the nausea that’s associated with the migraine medicine, or is nausea occurring at the end of the migraine?” she asked. “That’s always been kind of a conundrum for us.”
Whatever the case, she said, bariatric surgery appears to be helpful for patients with headache. Some studies have been small, but a 2021 analysis of 1,680 patients with migraine found that 55% experienced remission with no need for medication at 180 days post surgery. Women, older patients, and those taking more migraine medications were less likely to reach remission.
Research also suggests that bariatric surgery can relieve headache symptoms in patients with idiopathic intracranial hypertension.
Dr. McVige cautioned, however, that medical professionals must take special care when they talk to patients about their weight. “I’ve learned from conversations with my patients that they don’t like hearing ‘obese,’ or ‘fat,’ or ‘diet,’ or ‘losing weight.’ What they do like is ‘maybe we could try to find ways to be more healthy, to help your body to look the way that you would like it to look in the future. Let me help you. Maybe we can talk about nutrition. Maybe we can talk about exercise. Let’s talk about energy. Let’s talk about those types of things.’”
Unfortunately, there’s little research into how to have these conversations, Dr. McVige said. Still, “we need to be the first people to come forward and say, ‘This is a tough topic for me to talk about too. It makes me uncomfortable as well. I know you’re uncomfortable, but I have to talk to you because I care about you as a patient.’”
She also advised colleagues to not bring up weight right out of the gate. “It’s easier to say some of those things after you develop a relationship,” she said, “when they know you care about them.”
Dr. McVige reported multiple disclosures related to research funding and speaker fees.
AT AHS 2022
FDA authorizes COVID vaccines in kids as young as 6 months
, one of the final steps in a long-awaited authorization process to extend protection to the youngest of Americans.
The agency’s move comes after a closely watched FDA advisory group vote earlier this week, which resulted in a unanimous vote in favor of the FDA authorizing both vaccines in this age group.
“The FDA’s evaluation and analysis of the safety, effectiveness, and manufacturing data of these vaccines was rigorous and comprehensive, supporting the EUAs,” the agency said in a news release.
The data show that the “known and potential benefits” of the vaccines outweigh any potential risks, the agency said.
The Moderna vaccine is authorized as a two-dose primary series in children 6 months to 17 years of age. The Pfizer vaccine is now authorized as a three-dose primary series in children 6 months up to 4 years of age. Pfizer’s vaccine was already authorized in children 5 years old and older.
Now all eyes are on the Centers for Disease Control and Prevention, which is expected to decide on the final regulatory hurdle at a meeting June 18. The CDC’s Advisory Committee on Immunization Practices has scheduled a vote on whether to give the vaccines the green light.
If ACIP gives the OK, CDC Director Rochelle Walensky, MD, MPH, is expected to issue recommendations for use shortly thereafter.
Following these final regulatory steps, parents could start bringing their children to pediatricians, family doctors, or local pharmacies for vaccination as early as June 20.
A version of this article first appeared on WebMD.com.
, one of the final steps in a long-awaited authorization process to extend protection to the youngest of Americans.
The agency’s move comes after a closely watched FDA advisory group vote earlier this week, which resulted in a unanimous vote in favor of the FDA authorizing both vaccines in this age group.
“The FDA’s evaluation and analysis of the safety, effectiveness, and manufacturing data of these vaccines was rigorous and comprehensive, supporting the EUAs,” the agency said in a news release.
The data show that the “known and potential benefits” of the vaccines outweigh any potential risks, the agency said.
The Moderna vaccine is authorized as a two-dose primary series in children 6 months to 17 years of age. The Pfizer vaccine is now authorized as a three-dose primary series in children 6 months up to 4 years of age. Pfizer’s vaccine was already authorized in children 5 years old and older.
Now all eyes are on the Centers for Disease Control and Prevention, which is expected to decide on the final regulatory hurdle at a meeting June 18. The CDC’s Advisory Committee on Immunization Practices has scheduled a vote on whether to give the vaccines the green light.
If ACIP gives the OK, CDC Director Rochelle Walensky, MD, MPH, is expected to issue recommendations for use shortly thereafter.
Following these final regulatory steps, parents could start bringing their children to pediatricians, family doctors, or local pharmacies for vaccination as early as June 20.
A version of this article first appeared on WebMD.com.
, one of the final steps in a long-awaited authorization process to extend protection to the youngest of Americans.
The agency’s move comes after a closely watched FDA advisory group vote earlier this week, which resulted in a unanimous vote in favor of the FDA authorizing both vaccines in this age group.
“The FDA’s evaluation and analysis of the safety, effectiveness, and manufacturing data of these vaccines was rigorous and comprehensive, supporting the EUAs,” the agency said in a news release.
The data show that the “known and potential benefits” of the vaccines outweigh any potential risks, the agency said.
The Moderna vaccine is authorized as a two-dose primary series in children 6 months to 17 years of age. The Pfizer vaccine is now authorized as a three-dose primary series in children 6 months up to 4 years of age. Pfizer’s vaccine was already authorized in children 5 years old and older.
Now all eyes are on the Centers for Disease Control and Prevention, which is expected to decide on the final regulatory hurdle at a meeting June 18. The CDC’s Advisory Committee on Immunization Practices has scheduled a vote on whether to give the vaccines the green light.
If ACIP gives the OK, CDC Director Rochelle Walensky, MD, MPH, is expected to issue recommendations for use shortly thereafter.
Following these final regulatory steps, parents could start bringing their children to pediatricians, family doctors, or local pharmacies for vaccination as early as June 20.
A version of this article first appeared on WebMD.com.
Diabetes tied to risk of long COVID, too
Individuals with diabetes who experience COVID-19 are at increased risk for long COVID compared to individuals without diabetes, according to data from a literature review of seven studies.
Diabetes remains a risk factor for severe COVID-19, but whether it is a risk factor for postacute sequelae of COVID-19 (PASC), also known as long COVID, remains unclear, Jessica L. Harding, PhD, of Emory University, said in a late-breaking poster session at the annual scientific sessions of the American Diabetes Association.
Long COVID is generally defined as “sequelae that extend beyond the 4 weeks after initial infection” and may include a range of symptoms that affect multiple organs, Dr. Harding said. A study conducted in January of 2022 suggested that type 2 diabetes was one of several strong risk factors for long COVID, she noted.
Dr. Harding and colleagues reviewed data from seven studies published from Jan. 1, 2020, to Jan. 27, 2022, on the risk of PASC in people with and without diabetes. The studies included patients with a minimum of 4 weeks’ follow-up after COVID-19 diagnosis. All seven studies had a longitudinal cohort design, and included adults from high-income countries, with study populations ranging from 104 to 4,182.
Across the studies, long COVID definitions varied, but included ongoing symptoms of fatigue, cough, and dyspnea, with follow-up periods of 4 weeks to 7 months.
Overall, three of the seven studies indicated that diabetes was a risk factor for long COVID (odds ratio [OR] greater than 4 for all) and four studies indicated that diabetes was not a risk factor for long COVID (OR, 0.5-2.2).
One of the three studies showing increased risk included 2,334 individuals hospitalized with COVID-19; of these about 5% had diabetes. The odds ratio for PASC for individuals with diabetes was 4.18. In another study of 209 persons with COVID-19, of whom 22% had diabetes, diabetes was significantly correlated with respiratory viral disease (meaning at least two respiratory symptoms). The third study showing an increased risk of long COVID in diabetes patients included 104 kidney transplant patients, of whom 20% had diabetes; the odds ratio for PASC was 4.42.
The findings were limited by several factors, including the relatively small number of studies and the heterogeneity of studies regarding definitions of long COVID, specific populations at risk, follow-up times, and risk adjustment, Dr. Harding noted.
More high-quality studies across multiple populations and settings are needed to determine if diabetes is indeed a risk factor for long COVID, she said.
In the meantime, “careful monitoring of people with diabetes for development of PASC may be advised,” Dr. Harding concluded.
Findings support need for screening
“Given the devastating impact of COVID on people with diabetes, it’s important to know what data has been accumulated on long COVID for future research and discoveries in this area,” Robert A. Gabbay, MD, chief science and medical officer for the American Diabetes Association, said in an interview. “The more information we have, the better we can understand the implications.”
Dr. Gabbay said he was surprised by the current study findings. “We know very little on this subject, so yes, I am surprised to see just how significant the risk of long COVID for people with diabetes seems to be, but clearly, more research needs to be done to understand long COVID,” he emphasized.
The take-home message for clinicians is the importance of screening patients for PASC; also “ask your patients if they had COVID, to better understand any symptoms they might have that could be related to PACS,” he noted.
“It is crucial that we confirm these results and then look at risk factors in people with diabetes that might explain who is at highest risk and ultimately understand the causes and potential cure,” Dr. Gabbay added.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Harding and Dr. Gabbay had no financial conflicts to disclose.
Individuals with diabetes who experience COVID-19 are at increased risk for long COVID compared to individuals without diabetes, according to data from a literature review of seven studies.
Diabetes remains a risk factor for severe COVID-19, but whether it is a risk factor for postacute sequelae of COVID-19 (PASC), also known as long COVID, remains unclear, Jessica L. Harding, PhD, of Emory University, said in a late-breaking poster session at the annual scientific sessions of the American Diabetes Association.
Long COVID is generally defined as “sequelae that extend beyond the 4 weeks after initial infection” and may include a range of symptoms that affect multiple organs, Dr. Harding said. A study conducted in January of 2022 suggested that type 2 diabetes was one of several strong risk factors for long COVID, she noted.
Dr. Harding and colleagues reviewed data from seven studies published from Jan. 1, 2020, to Jan. 27, 2022, on the risk of PASC in people with and without diabetes. The studies included patients with a minimum of 4 weeks’ follow-up after COVID-19 diagnosis. All seven studies had a longitudinal cohort design, and included adults from high-income countries, with study populations ranging from 104 to 4,182.
Across the studies, long COVID definitions varied, but included ongoing symptoms of fatigue, cough, and dyspnea, with follow-up periods of 4 weeks to 7 months.
Overall, three of the seven studies indicated that diabetes was a risk factor for long COVID (odds ratio [OR] greater than 4 for all) and four studies indicated that diabetes was not a risk factor for long COVID (OR, 0.5-2.2).
One of the three studies showing increased risk included 2,334 individuals hospitalized with COVID-19; of these about 5% had diabetes. The odds ratio for PASC for individuals with diabetes was 4.18. In another study of 209 persons with COVID-19, of whom 22% had diabetes, diabetes was significantly correlated with respiratory viral disease (meaning at least two respiratory symptoms). The third study showing an increased risk of long COVID in diabetes patients included 104 kidney transplant patients, of whom 20% had diabetes; the odds ratio for PASC was 4.42.
The findings were limited by several factors, including the relatively small number of studies and the heterogeneity of studies regarding definitions of long COVID, specific populations at risk, follow-up times, and risk adjustment, Dr. Harding noted.
More high-quality studies across multiple populations and settings are needed to determine if diabetes is indeed a risk factor for long COVID, she said.
In the meantime, “careful monitoring of people with diabetes for development of PASC may be advised,” Dr. Harding concluded.
Findings support need for screening
“Given the devastating impact of COVID on people with diabetes, it’s important to know what data has been accumulated on long COVID for future research and discoveries in this area,” Robert A. Gabbay, MD, chief science and medical officer for the American Diabetes Association, said in an interview. “The more information we have, the better we can understand the implications.”
Dr. Gabbay said he was surprised by the current study findings. “We know very little on this subject, so yes, I am surprised to see just how significant the risk of long COVID for people with diabetes seems to be, but clearly, more research needs to be done to understand long COVID,” he emphasized.
The take-home message for clinicians is the importance of screening patients for PASC; also “ask your patients if they had COVID, to better understand any symptoms they might have that could be related to PACS,” he noted.
“It is crucial that we confirm these results and then look at risk factors in people with diabetes that might explain who is at highest risk and ultimately understand the causes and potential cure,” Dr. Gabbay added.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Harding and Dr. Gabbay had no financial conflicts to disclose.
Individuals with diabetes who experience COVID-19 are at increased risk for long COVID compared to individuals without diabetes, according to data from a literature review of seven studies.
Diabetes remains a risk factor for severe COVID-19, but whether it is a risk factor for postacute sequelae of COVID-19 (PASC), also known as long COVID, remains unclear, Jessica L. Harding, PhD, of Emory University, said in a late-breaking poster session at the annual scientific sessions of the American Diabetes Association.
Long COVID is generally defined as “sequelae that extend beyond the 4 weeks after initial infection” and may include a range of symptoms that affect multiple organs, Dr. Harding said. A study conducted in January of 2022 suggested that type 2 diabetes was one of several strong risk factors for long COVID, she noted.
Dr. Harding and colleagues reviewed data from seven studies published from Jan. 1, 2020, to Jan. 27, 2022, on the risk of PASC in people with and without diabetes. The studies included patients with a minimum of 4 weeks’ follow-up after COVID-19 diagnosis. All seven studies had a longitudinal cohort design, and included adults from high-income countries, with study populations ranging from 104 to 4,182.
Across the studies, long COVID definitions varied, but included ongoing symptoms of fatigue, cough, and dyspnea, with follow-up periods of 4 weeks to 7 months.
Overall, three of the seven studies indicated that diabetes was a risk factor for long COVID (odds ratio [OR] greater than 4 for all) and four studies indicated that diabetes was not a risk factor for long COVID (OR, 0.5-2.2).
One of the three studies showing increased risk included 2,334 individuals hospitalized with COVID-19; of these about 5% had diabetes. The odds ratio for PASC for individuals with diabetes was 4.18. In another study of 209 persons with COVID-19, of whom 22% had diabetes, diabetes was significantly correlated with respiratory viral disease (meaning at least two respiratory symptoms). The third study showing an increased risk of long COVID in diabetes patients included 104 kidney transplant patients, of whom 20% had diabetes; the odds ratio for PASC was 4.42.
The findings were limited by several factors, including the relatively small number of studies and the heterogeneity of studies regarding definitions of long COVID, specific populations at risk, follow-up times, and risk adjustment, Dr. Harding noted.
More high-quality studies across multiple populations and settings are needed to determine if diabetes is indeed a risk factor for long COVID, she said.
In the meantime, “careful monitoring of people with diabetes for development of PASC may be advised,” Dr. Harding concluded.
Findings support need for screening
“Given the devastating impact of COVID on people with diabetes, it’s important to know what data has been accumulated on long COVID for future research and discoveries in this area,” Robert A. Gabbay, MD, chief science and medical officer for the American Diabetes Association, said in an interview. “The more information we have, the better we can understand the implications.”
Dr. Gabbay said he was surprised by the current study findings. “We know very little on this subject, so yes, I am surprised to see just how significant the risk of long COVID for people with diabetes seems to be, but clearly, more research needs to be done to understand long COVID,” he emphasized.
The take-home message for clinicians is the importance of screening patients for PASC; also “ask your patients if they had COVID, to better understand any symptoms they might have that could be related to PACS,” he noted.
“It is crucial that we confirm these results and then look at risk factors in people with diabetes that might explain who is at highest risk and ultimately understand the causes and potential cure,” Dr. Gabbay added.
The study was supported by the National Heart, Lung, and Blood Institute. Dr. Harding and Dr. Gabbay had no financial conflicts to disclose.
FROM ADA 2022
Eosinophils may predict outcomes in acute COPD exacerbations
High levels of eosinophils had a protective effect for individuals who experienced acute exacerbations of chronic obstructive pulmonary disease, based on data from nearly 1,000 patients.
Several blood biomarkers are under investigation for links to acute exacerbation of chronic obstructive pulmonary disease (AECOPD), which remains one of the top three causes of death worldwide, wrote Riuying Wang, MD, of Third Hospital of Shanxi Medical University, Taiyuan, China, and colleagues.
“Numerous studies have shown the relationship between eosinophilia and clinical outcomes of patients with AECOPD. However, the evidence lacks consensus, and the research thresholds are controversial,” they said.
In a study published in Heart & Lung, the researchers reviewed data from 984 adults with AECOPD over a 3-year follow-up period. The mean age of the patients was 71 years, and 78% were men. The patients’ blood eosinophil levels were grouped into three categories: EOS < 2%, EOS from 2% to < 3%, and 3% or higher. The researchers examined the association between eosinophilia and various comorbidities, treatment, and mortality.
Eosinophilia occurred in 477 cases. The prevalence of eosinophilia in the three groups was 36.48%, 22.87%, and 48.48% respectively, with eosinophilia defined as eosinophil counts of at least 100 cells per microliter, according to the report in Heart & Lung.
An EOS of 2% or higher was associated with significantly fewer cases of complicated pulmonary heart disease and atrial fibrillation than the lower EOS group. Similarly, patients in the EOS group of 2% or higher were less likely to use ventilators and systemic glucocorticoids and those in the EOS less than 2% group had significantly heavier airflow limitation, higher D-dimer, higher burden of infectious inflammation, and higher prevalence of respiratory failure than the other groups.
In addition, significantly fewer deaths occurred during the study period among patients with EOS of 2% or higher, compared with the lower EOS group (P < .01). The findings suggest that “Eosinophils can be used as a prognostic indicator of mortality in AECOPD,” the researchers said.
The researchers also used the area under the curve to examine the predictive value of EOS. The ROC curve showed that the indicators of AUC 0.5 included chest CT imaging, osteoporosis, mental illness, dust exposure, and being a former smoker; however, “the predictive value of EOS by the ROC curve was unstable. Further validation in large samples is needed,” the researchers wrote in their discussion.
The study findings were limited by several factors including the retrospective design and use of data from a single center, the researchers noted. Other limitations included the relatively small sample size and a lack of data on some clinical features and performance metrics, as well as lack of evaluation of chest CT subtypes.
However, the results are consistent with previous studies on infection and antibiotics and reviewed the optimal threshold of AECOPD, the researchers wrote. Based on their findings, “Eosinophils can not only guide clinical treatment but also be used as an index to predict the clinical outcome and prognosis of AECOPD patients,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
High levels of eosinophils had a protective effect for individuals who experienced acute exacerbations of chronic obstructive pulmonary disease, based on data from nearly 1,000 patients.
Several blood biomarkers are under investigation for links to acute exacerbation of chronic obstructive pulmonary disease (AECOPD), which remains one of the top three causes of death worldwide, wrote Riuying Wang, MD, of Third Hospital of Shanxi Medical University, Taiyuan, China, and colleagues.
“Numerous studies have shown the relationship between eosinophilia and clinical outcomes of patients with AECOPD. However, the evidence lacks consensus, and the research thresholds are controversial,” they said.
In a study published in Heart & Lung, the researchers reviewed data from 984 adults with AECOPD over a 3-year follow-up period. The mean age of the patients was 71 years, and 78% were men. The patients’ blood eosinophil levels were grouped into three categories: EOS < 2%, EOS from 2% to < 3%, and 3% or higher. The researchers examined the association between eosinophilia and various comorbidities, treatment, and mortality.
Eosinophilia occurred in 477 cases. The prevalence of eosinophilia in the three groups was 36.48%, 22.87%, and 48.48% respectively, with eosinophilia defined as eosinophil counts of at least 100 cells per microliter, according to the report in Heart & Lung.
An EOS of 2% or higher was associated with significantly fewer cases of complicated pulmonary heart disease and atrial fibrillation than the lower EOS group. Similarly, patients in the EOS group of 2% or higher were less likely to use ventilators and systemic glucocorticoids and those in the EOS less than 2% group had significantly heavier airflow limitation, higher D-dimer, higher burden of infectious inflammation, and higher prevalence of respiratory failure than the other groups.
In addition, significantly fewer deaths occurred during the study period among patients with EOS of 2% or higher, compared with the lower EOS group (P < .01). The findings suggest that “Eosinophils can be used as a prognostic indicator of mortality in AECOPD,” the researchers said.
The researchers also used the area under the curve to examine the predictive value of EOS. The ROC curve showed that the indicators of AUC 0.5 included chest CT imaging, osteoporosis, mental illness, dust exposure, and being a former smoker; however, “the predictive value of EOS by the ROC curve was unstable. Further validation in large samples is needed,” the researchers wrote in their discussion.
The study findings were limited by several factors including the retrospective design and use of data from a single center, the researchers noted. Other limitations included the relatively small sample size and a lack of data on some clinical features and performance metrics, as well as lack of evaluation of chest CT subtypes.
However, the results are consistent with previous studies on infection and antibiotics and reviewed the optimal threshold of AECOPD, the researchers wrote. Based on their findings, “Eosinophils can not only guide clinical treatment but also be used as an index to predict the clinical outcome and prognosis of AECOPD patients,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
High levels of eosinophils had a protective effect for individuals who experienced acute exacerbations of chronic obstructive pulmonary disease, based on data from nearly 1,000 patients.
Several blood biomarkers are under investigation for links to acute exacerbation of chronic obstructive pulmonary disease (AECOPD), which remains one of the top three causes of death worldwide, wrote Riuying Wang, MD, of Third Hospital of Shanxi Medical University, Taiyuan, China, and colleagues.
“Numerous studies have shown the relationship between eosinophilia and clinical outcomes of patients with AECOPD. However, the evidence lacks consensus, and the research thresholds are controversial,” they said.
In a study published in Heart & Lung, the researchers reviewed data from 984 adults with AECOPD over a 3-year follow-up period. The mean age of the patients was 71 years, and 78% were men. The patients’ blood eosinophil levels were grouped into three categories: EOS < 2%, EOS from 2% to < 3%, and 3% or higher. The researchers examined the association between eosinophilia and various comorbidities, treatment, and mortality.
Eosinophilia occurred in 477 cases. The prevalence of eosinophilia in the three groups was 36.48%, 22.87%, and 48.48% respectively, with eosinophilia defined as eosinophil counts of at least 100 cells per microliter, according to the report in Heart & Lung.
An EOS of 2% or higher was associated with significantly fewer cases of complicated pulmonary heart disease and atrial fibrillation than the lower EOS group. Similarly, patients in the EOS group of 2% or higher were less likely to use ventilators and systemic glucocorticoids and those in the EOS less than 2% group had significantly heavier airflow limitation, higher D-dimer, higher burden of infectious inflammation, and higher prevalence of respiratory failure than the other groups.
In addition, significantly fewer deaths occurred during the study period among patients with EOS of 2% or higher, compared with the lower EOS group (P < .01). The findings suggest that “Eosinophils can be used as a prognostic indicator of mortality in AECOPD,” the researchers said.
The researchers also used the area under the curve to examine the predictive value of EOS. The ROC curve showed that the indicators of AUC 0.5 included chest CT imaging, osteoporosis, mental illness, dust exposure, and being a former smoker; however, “the predictive value of EOS by the ROC curve was unstable. Further validation in large samples is needed,” the researchers wrote in their discussion.
The study findings were limited by several factors including the retrospective design and use of data from a single center, the researchers noted. Other limitations included the relatively small sample size and a lack of data on some clinical features and performance metrics, as well as lack of evaluation of chest CT subtypes.
However, the results are consistent with previous studies on infection and antibiotics and reviewed the optimal threshold of AECOPD, the researchers wrote. Based on their findings, “Eosinophils can not only guide clinical treatment but also be used as an index to predict the clinical outcome and prognosis of AECOPD patients,” they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM HEART & LUNG
Snoring may lead to a sedentary lifestyle
“People who snore are also likely to have sleep apnea, but those who snore and don’t have sleep apnea are a largely understudied group,” senior author Michael Grandner, PhD, told this news organization.
“We found that even just snoring alone can impact health and well-being,” said Dr. Grandner, director of the sleep and health research program at the University of Arizona, Tucson.
The findings were presented at the annual meeting of the Associated Professional Sleep Societies.
A viscous cycle
Frequent snoring can signal sleep-disordered breathing, which is associated with a myriad of comorbidities, including increased risk for cardiovascular disease.
Prior studies have shown that sleep-disordered breathing is associated with less physical activity, but few studies have examined this at the population level or in relation to primary snoring.
Dr. Grandner and colleagues evaluated the relationship between snoring frequency and minutes of sedentary activity using 3 years’ worth of data from the National Health and Nutrition Examination Survey. Participants reported snoring frequency and sedentary activity.
After adjusting for sex, age, race, education level, and marital status, adults who were frequent snorers (5+ nights per week) spent about 36 more minutes per day sedentary, compared with peers who reported never snoring.
In addition, those individuals who were determined to be at increased risk of having sleep apnea had about 54 more minutes per day of sedentary time in the adjusted model.
“Snoring is very common, and it doesn’t just affect the nighttime,” said Dr. Grandner.
Snoring can lead to “more tiredness and less energy, which can impact everything from mood to stress to – as we saw – activity level,” he noted.
Commenting on the results for this news organization, Raman Malhotra, MD, of the Washington University Sleep Center in St. Louis, said this study clearly demonstrates how people who snore and people who are at risk for sleep apnea are more sedentary.
This could explain the “vicious cycle” that these patients suffer from, inasmuch as having obesity can lead to sleep apnea, and having sleep apnea can lead to further sedentary lifestyle and weight gain, owing to lack of energy and feeling tired, Dr. Malhotra told this news organization.
“It is important to intervene and treat the sleep disorder to hopefully make people more active,” he added.
The study had no specific funding. Dr. Grandner and Dr. Malhotra disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“People who snore are also likely to have sleep apnea, but those who snore and don’t have sleep apnea are a largely understudied group,” senior author Michael Grandner, PhD, told this news organization.
“We found that even just snoring alone can impact health and well-being,” said Dr. Grandner, director of the sleep and health research program at the University of Arizona, Tucson.
The findings were presented at the annual meeting of the Associated Professional Sleep Societies.
A viscous cycle
Frequent snoring can signal sleep-disordered breathing, which is associated with a myriad of comorbidities, including increased risk for cardiovascular disease.
Prior studies have shown that sleep-disordered breathing is associated with less physical activity, but few studies have examined this at the population level or in relation to primary snoring.
Dr. Grandner and colleagues evaluated the relationship between snoring frequency and minutes of sedentary activity using 3 years’ worth of data from the National Health and Nutrition Examination Survey. Participants reported snoring frequency and sedentary activity.
After adjusting for sex, age, race, education level, and marital status, adults who were frequent snorers (5+ nights per week) spent about 36 more minutes per day sedentary, compared with peers who reported never snoring.
In addition, those individuals who were determined to be at increased risk of having sleep apnea had about 54 more minutes per day of sedentary time in the adjusted model.
“Snoring is very common, and it doesn’t just affect the nighttime,” said Dr. Grandner.
Snoring can lead to “more tiredness and less energy, which can impact everything from mood to stress to – as we saw – activity level,” he noted.
Commenting on the results for this news organization, Raman Malhotra, MD, of the Washington University Sleep Center in St. Louis, said this study clearly demonstrates how people who snore and people who are at risk for sleep apnea are more sedentary.
This could explain the “vicious cycle” that these patients suffer from, inasmuch as having obesity can lead to sleep apnea, and having sleep apnea can lead to further sedentary lifestyle and weight gain, owing to lack of energy and feeling tired, Dr. Malhotra told this news organization.
“It is important to intervene and treat the sleep disorder to hopefully make people more active,” he added.
The study had no specific funding. Dr. Grandner and Dr. Malhotra disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
“People who snore are also likely to have sleep apnea, but those who snore and don’t have sleep apnea are a largely understudied group,” senior author Michael Grandner, PhD, told this news organization.
“We found that even just snoring alone can impact health and well-being,” said Dr. Grandner, director of the sleep and health research program at the University of Arizona, Tucson.
The findings were presented at the annual meeting of the Associated Professional Sleep Societies.
A viscous cycle
Frequent snoring can signal sleep-disordered breathing, which is associated with a myriad of comorbidities, including increased risk for cardiovascular disease.
Prior studies have shown that sleep-disordered breathing is associated with less physical activity, but few studies have examined this at the population level or in relation to primary snoring.
Dr. Grandner and colleagues evaluated the relationship between snoring frequency and minutes of sedentary activity using 3 years’ worth of data from the National Health and Nutrition Examination Survey. Participants reported snoring frequency and sedentary activity.
After adjusting for sex, age, race, education level, and marital status, adults who were frequent snorers (5+ nights per week) spent about 36 more minutes per day sedentary, compared with peers who reported never snoring.
In addition, those individuals who were determined to be at increased risk of having sleep apnea had about 54 more minutes per day of sedentary time in the adjusted model.
“Snoring is very common, and it doesn’t just affect the nighttime,” said Dr. Grandner.
Snoring can lead to “more tiredness and less energy, which can impact everything from mood to stress to – as we saw – activity level,” he noted.
Commenting on the results for this news organization, Raman Malhotra, MD, of the Washington University Sleep Center in St. Louis, said this study clearly demonstrates how people who snore and people who are at risk for sleep apnea are more sedentary.
This could explain the “vicious cycle” that these patients suffer from, inasmuch as having obesity can lead to sleep apnea, and having sleep apnea can lead to further sedentary lifestyle and weight gain, owing to lack of energy and feeling tired, Dr. Malhotra told this news organization.
“It is important to intervene and treat the sleep disorder to hopefully make people more active,” he added.
The study had no specific funding. Dr. Grandner and Dr. Malhotra disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM SLEEP 2022
New law aims to meet crushing need for mental health care professionals
, say state leaders.
Governor J.B. Pritzker (D) signed the legislation, which took effect on June 10.
The law seeks to attract psychologists, social workers, and counselors who have left the workforce within the past 5 years by temporarily ending relicensing requirements, including the need for continuing education credit completion, passing new exams, and fee payments. It also eases the process for those practicing in other states to become licensed in Illinois.
State legislators said there is currently a crushing need for mental health providers, estimating that there are only 14 behavioral health care professionals for every 10,000 Illinois residents. The preamble to the law noted that there will be 8,353 unfilled mental health care jobs in Illinois by 2026.
“We need a mental health care workforce that is robust enough to get people help when they need it – not after months on a waiting list,” Governor Pritzker said in news release. “This legislation invests in mental health infrastructure – and that infrastructure is people,” he added.
Grant pathway
“Being told you have to wait weeks – or months – for care is extremely discouraging,” State Senator Laura Fine (D), a lead sponsor of the legislation, noted in the release.
“We need to support people struggling with mental and behavioral health issues, as well as address difficulties our mental health providers are facing trying to see as many patients as possible,” said Senator Fine.
Marvin Lindsey, CEO of the Community Behavioral Healthcare Association, added that the law would “accelerate the process for out-of-state professionals to obtain their Illinois licensure and [increase] the pipeline and diversity of the behavioral health workforce by implementing a funding mechanism that supports new or existing licensure training of interns.”
The law sets up a grant pathway for community mental health centers, which often serve as training sites. The grants would provide funds to establish or enhance training and supervision of interns and behavioral health providers-in-training seeking to become licensed clinical social workers, licensed clinical professional counselors, or licensed marriage and family therapists.
The money for those grants still has to be appropriated.
The law will also allow patient visits at Specialized Mental Health Rehabilitation Facilities conducted by either a psychiatrist or an advanced practice registered mental health or psychiatric nurse.
Finally, it would establish tax credits for employers who hire individuals in recovery from a substance use disorder or a behavioral disorder. Beginning in January 2023, employers will be eligible for up to $2,000 in credits per employee hired.
A version of this article first appeared on Medscape.com.
, say state leaders.
Governor J.B. Pritzker (D) signed the legislation, which took effect on June 10.
The law seeks to attract psychologists, social workers, and counselors who have left the workforce within the past 5 years by temporarily ending relicensing requirements, including the need for continuing education credit completion, passing new exams, and fee payments. It also eases the process for those practicing in other states to become licensed in Illinois.
State legislators said there is currently a crushing need for mental health providers, estimating that there are only 14 behavioral health care professionals for every 10,000 Illinois residents. The preamble to the law noted that there will be 8,353 unfilled mental health care jobs in Illinois by 2026.
“We need a mental health care workforce that is robust enough to get people help when they need it – not after months on a waiting list,” Governor Pritzker said in news release. “This legislation invests in mental health infrastructure – and that infrastructure is people,” he added.
Grant pathway
“Being told you have to wait weeks – or months – for care is extremely discouraging,” State Senator Laura Fine (D), a lead sponsor of the legislation, noted in the release.
“We need to support people struggling with mental and behavioral health issues, as well as address difficulties our mental health providers are facing trying to see as many patients as possible,” said Senator Fine.
Marvin Lindsey, CEO of the Community Behavioral Healthcare Association, added that the law would “accelerate the process for out-of-state professionals to obtain their Illinois licensure and [increase] the pipeline and diversity of the behavioral health workforce by implementing a funding mechanism that supports new or existing licensure training of interns.”
The law sets up a grant pathway for community mental health centers, which often serve as training sites. The grants would provide funds to establish or enhance training and supervision of interns and behavioral health providers-in-training seeking to become licensed clinical social workers, licensed clinical professional counselors, or licensed marriage and family therapists.
The money for those grants still has to be appropriated.
The law will also allow patient visits at Specialized Mental Health Rehabilitation Facilities conducted by either a psychiatrist or an advanced practice registered mental health or psychiatric nurse.
Finally, it would establish tax credits for employers who hire individuals in recovery from a substance use disorder or a behavioral disorder. Beginning in January 2023, employers will be eligible for up to $2,000 in credits per employee hired.
A version of this article first appeared on Medscape.com.
, say state leaders.
Governor J.B. Pritzker (D) signed the legislation, which took effect on June 10.
The law seeks to attract psychologists, social workers, and counselors who have left the workforce within the past 5 years by temporarily ending relicensing requirements, including the need for continuing education credit completion, passing new exams, and fee payments. It also eases the process for those practicing in other states to become licensed in Illinois.
State legislators said there is currently a crushing need for mental health providers, estimating that there are only 14 behavioral health care professionals for every 10,000 Illinois residents. The preamble to the law noted that there will be 8,353 unfilled mental health care jobs in Illinois by 2026.
“We need a mental health care workforce that is robust enough to get people help when they need it – not after months on a waiting list,” Governor Pritzker said in news release. “This legislation invests in mental health infrastructure – and that infrastructure is people,” he added.
Grant pathway
“Being told you have to wait weeks – or months – for care is extremely discouraging,” State Senator Laura Fine (D), a lead sponsor of the legislation, noted in the release.
“We need to support people struggling with mental and behavioral health issues, as well as address difficulties our mental health providers are facing trying to see as many patients as possible,” said Senator Fine.
Marvin Lindsey, CEO of the Community Behavioral Healthcare Association, added that the law would “accelerate the process for out-of-state professionals to obtain their Illinois licensure and [increase] the pipeline and diversity of the behavioral health workforce by implementing a funding mechanism that supports new or existing licensure training of interns.”
The law sets up a grant pathway for community mental health centers, which often serve as training sites. The grants would provide funds to establish or enhance training and supervision of interns and behavioral health providers-in-training seeking to become licensed clinical social workers, licensed clinical professional counselors, or licensed marriage and family therapists.
The money for those grants still has to be appropriated.
The law will also allow patient visits at Specialized Mental Health Rehabilitation Facilities conducted by either a psychiatrist or an advanced practice registered mental health or psychiatric nurse.
Finally, it would establish tax credits for employers who hire individuals in recovery from a substance use disorder or a behavioral disorder. Beginning in January 2023, employers will be eligible for up to $2,000 in credits per employee hired.
A version of this article first appeared on Medscape.com.
Hypothyroidism: No more waiting to eat or drink with liquid thyroxine?
ATLANTA -- Liquid formulations of levothyroxine offer the possibility of allowing patients with hypothyroidism to take their medication with meals or coffee and skip the currently recommended 30- to 60-minute waiting period before doing either, new data suggest.
Because food, coffee, and certain medications can interfere with intestinal absorption of levothyroxine (also known as LT4), current guidelines recommend that the drug be taken in a fasting state, typically 30-60 minutes before breakfast. However, compliance may be difficult for some patients.
Now, a potential solution may come from new evidence that liquid levothyroxine formulations that bypass the gastric dissolution phase of absorption may mitigate the interference with food and coffee.
Findings from two bioavailability studies showing no difference in comparisons of Thyquidity (levothyroxine sodium oral solution, Vertice Pharma) with or without waiting periods before consuming coffee or a high-fat meal were presented at the annual meeting of the Endocrine Society (ENDO 2022), by Vertice Pharma Medical Director Kris Washington, PharmD.
And just last month, similar data were published in Thyroid for another levothyroxine oral solution, Tirosint-SOL (IBSA). No difference in pharmacokinetic properties were found with this product with a shorter versus a longer waiting period before consuming a high-fat meal.
Liquid thyroxine may be less affected by food/drink but is expensive
Both products have been approved by the U.S. Food and Drug Administration, but current labeling for both still calls for a 30- to 60-minute waiting period between taking the medication and eating or drinking. Thyquidity is an oral solution of 100 µg/mL levothyroxine sodium that has been shown to be bioequivalent to one of the most popular branded levothyroxine tablets, Synthroid (AbbVie), under fasting conditions. Tirosint-SOL is also an oral solution that comes in 15 different dosage ampules.
“It is important to note that while these findings are exciting and encouraging, we do want you to continue to follow the current FDA-approved label for Thyquidity, recommending that it be taken on an empty stomach 30-60 minutes prior to breakfast and that patients continue to follow all other label instructions,” Dr. Washington said during a press briefing at ENDO 2022.
When asked whether the new data would be submitted to the FDA for a possible amendment to this message, she replied: “We’re still discussing that. We’re exploring all options. ... This is fairly new data. ... It makes sense and certainly solves a lot of the challenges for people who can’t swallow or don’t choose to swallow, or the challenges of splitting or crushing with tablets.”
Asked to comment, Benjamin J. Gigliotti, MD, a clinical thyroidologist at the University of Rochester, New York, told this news organization: “Liquid levothyroxine has the potential to be a clinically useful formulation,” noting that these recent data corroborate prior findings from Europe and elsewhere that liquid levothyroxine is absorbed more rapidly and thus may be less impacted by food or beverages.
However, Dr. Gigliotti also pointed out, “I don’t think malabsorption is a major contributor to suboptimal treatment because if [patients] malabsorb the hormone, we typically just increase their dose a little bit or ask them to take it separately, and that works just fine for most people.”
And the higher cost of the liquid products is a major issue, he noted.
A quick search on GoodRx shows that the lowest price of Tirosint-SOL is $115.52 for a 1 month supply and Thyquidity is $181.04/month. “In the few patients where I tried to obtain Tirosint-SOL, it was not covered by insurance, even with a prior authorization,” Dr. Gigliotti commented.
In contrast, generic levothyroxine tablets are about $4/month, while a common brand name of levothyroxine tablets are $47.81/month.
“Until these liquid formulations are more widely covered by insurance for a reasonable copay, or come down in price compared to generic levothyroxine tablets, most of my patients have voiced that they’d rather deal with the inconveniences of a tablet compared to higher medication cost, especially with rising economic insecurity imposed by the COVID-19 pandemic and recent world events,” Dr. Gigliotti said.
Bioequivalence with shorter versus longer waits before coffee/breakfast
The Thyquidity coffee study was a single-center open-label, randomized, crossover study of 40 healthy adults randomized after a 10-hour overnight fast to 600 µg Thyquidity with water under fasting conditions or to the same dose given 5 minutes prior to drinking an 8-ounce cup of American coffee without milk or sweeteners. After a 40-day washout period, the same participants received the other treatment.
Mean serum thyroxine (T4) concentrations over 48 hours were nearly identical, demonstrating comparable bioavailability. Pharmacokinetics parameters, including area under the curve (AUC) and Cmax, were also comparable for both groups. The geometric least square mean ratios for baseline-adjusted LT4 were 96.0% for Cmax and 94% for AUC. And the corresponding 90% confidence intervals fell within the 80%-125% FDA acceptance range for absence of a food effect on bioavailability, said Dr. Washington when presenting the findings.
There was one adverse event, a decrease in blood glucose level, which was deemed to be mild and unrelated to study treatment. No deaths, serious adverse events, or discontinuations due to adverse events were reported. There were no significant changes in vital signs or on ECG.
In the second Thyquidity study of 38 healthy adults, after a 10-hour fast, the same doses were given 10 or 30 minutes prior to the consumption of a 950-calorie standardized high-fat breakfast.
Again, over 48 hours, mean serum T4 levels were comparable between the two groups. The geometric least squares mean ratios for both AUC and Cmax for baseline-adjusted LT4 were 88.7% and 85.1%, respectively. Again, the corresponding 90% confidence intervals fell within the FDA’s noninterference definition, again demonstrating lack of a food effect on bioavailability, Dr. Washington noted.
Four adverse events were reported in three participants, with three deemed to be possibly related to the medication. All were isolated lab abnormalities without clinical symptoms and deemed to be mild. Three were normal on repeat testing.
There were no deaths or serious adverse events or study discontinuations for adverse events and no significant findings for vital signs or on ECG.
Similar findings for Tirosint-SOL but longer-term studies needed
The recently published Tirosint-SOL study included 36 healthy volunteers randomized to single 600-µg doses of the LT4 oral solution after a 10-hour fast, either 15 or 30 minutes before eating a standardized high-fat, high-calorie meal. Mean serum total thyroxine concentration profiles were similar for both the 15- and 30-minute waits, with similar AUCs.
Geometric mean ratios for AUCs at 48 and 72 hours were 90% and 92%, respectively, and the 90% confidence intervals fell within the 80%-125% FDA boundaries, suggesting similar exposures whether taken 15 or 30 minutes before a meal.
Senior author Francesco S. Celi, MD, chair of the division of endocrinology, diabetes, and metabolism at Virginia Commonwealth University, Richmond, told this news organization: “There is an interest in providing more opportunities for patients and improving adherence to the medication. ... Whatever makes life a bit easier for patients and results in a more predictable response to treatment means down the road there will be fewer visits to the doctor to make adjustments.”
However, he said that in addition to the cost and reimbursement issue, all of these studies have been short term and not conducted in real-life settings.
“Another question is: What happens if the patient goes on low-dose LT4? The studies were conducted on much higher pharmacologic doses. But at least from a safety standpoint, there’s no specific concern.”
Dr. Washington is an employee of Vertice Pharma. Dr. Celi has received unrestricted research grants and worked as a consultant for IBSA. Dr. Gigliotti has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ATLANTA -- Liquid formulations of levothyroxine offer the possibility of allowing patients with hypothyroidism to take their medication with meals or coffee and skip the currently recommended 30- to 60-minute waiting period before doing either, new data suggest.
Because food, coffee, and certain medications can interfere with intestinal absorption of levothyroxine (also known as LT4), current guidelines recommend that the drug be taken in a fasting state, typically 30-60 minutes before breakfast. However, compliance may be difficult for some patients.
Now, a potential solution may come from new evidence that liquid levothyroxine formulations that bypass the gastric dissolution phase of absorption may mitigate the interference with food and coffee.
Findings from two bioavailability studies showing no difference in comparisons of Thyquidity (levothyroxine sodium oral solution, Vertice Pharma) with or without waiting periods before consuming coffee or a high-fat meal were presented at the annual meeting of the Endocrine Society (ENDO 2022), by Vertice Pharma Medical Director Kris Washington, PharmD.
And just last month, similar data were published in Thyroid for another levothyroxine oral solution, Tirosint-SOL (IBSA). No difference in pharmacokinetic properties were found with this product with a shorter versus a longer waiting period before consuming a high-fat meal.
Liquid thyroxine may be less affected by food/drink but is expensive
Both products have been approved by the U.S. Food and Drug Administration, but current labeling for both still calls for a 30- to 60-minute waiting period between taking the medication and eating or drinking. Thyquidity is an oral solution of 100 µg/mL levothyroxine sodium that has been shown to be bioequivalent to one of the most popular branded levothyroxine tablets, Synthroid (AbbVie), under fasting conditions. Tirosint-SOL is also an oral solution that comes in 15 different dosage ampules.
“It is important to note that while these findings are exciting and encouraging, we do want you to continue to follow the current FDA-approved label for Thyquidity, recommending that it be taken on an empty stomach 30-60 minutes prior to breakfast and that patients continue to follow all other label instructions,” Dr. Washington said during a press briefing at ENDO 2022.
When asked whether the new data would be submitted to the FDA for a possible amendment to this message, she replied: “We’re still discussing that. We’re exploring all options. ... This is fairly new data. ... It makes sense and certainly solves a lot of the challenges for people who can’t swallow or don’t choose to swallow, or the challenges of splitting or crushing with tablets.”
Asked to comment, Benjamin J. Gigliotti, MD, a clinical thyroidologist at the University of Rochester, New York, told this news organization: “Liquid levothyroxine has the potential to be a clinically useful formulation,” noting that these recent data corroborate prior findings from Europe and elsewhere that liquid levothyroxine is absorbed more rapidly and thus may be less impacted by food or beverages.
However, Dr. Gigliotti also pointed out, “I don’t think malabsorption is a major contributor to suboptimal treatment because if [patients] malabsorb the hormone, we typically just increase their dose a little bit or ask them to take it separately, and that works just fine for most people.”
And the higher cost of the liquid products is a major issue, he noted.
A quick search on GoodRx shows that the lowest price of Tirosint-SOL is $115.52 for a 1 month supply and Thyquidity is $181.04/month. “In the few patients where I tried to obtain Tirosint-SOL, it was not covered by insurance, even with a prior authorization,” Dr. Gigliotti commented.
In contrast, generic levothyroxine tablets are about $4/month, while a common brand name of levothyroxine tablets are $47.81/month.
“Until these liquid formulations are more widely covered by insurance for a reasonable copay, or come down in price compared to generic levothyroxine tablets, most of my patients have voiced that they’d rather deal with the inconveniences of a tablet compared to higher medication cost, especially with rising economic insecurity imposed by the COVID-19 pandemic and recent world events,” Dr. Gigliotti said.
Bioequivalence with shorter versus longer waits before coffee/breakfast
The Thyquidity coffee study was a single-center open-label, randomized, crossover study of 40 healthy adults randomized after a 10-hour overnight fast to 600 µg Thyquidity with water under fasting conditions or to the same dose given 5 minutes prior to drinking an 8-ounce cup of American coffee without milk or sweeteners. After a 40-day washout period, the same participants received the other treatment.
Mean serum thyroxine (T4) concentrations over 48 hours were nearly identical, demonstrating comparable bioavailability. Pharmacokinetics parameters, including area under the curve (AUC) and Cmax, were also comparable for both groups. The geometric least square mean ratios for baseline-adjusted LT4 were 96.0% for Cmax and 94% for AUC. And the corresponding 90% confidence intervals fell within the 80%-125% FDA acceptance range for absence of a food effect on bioavailability, said Dr. Washington when presenting the findings.
There was one adverse event, a decrease in blood glucose level, which was deemed to be mild and unrelated to study treatment. No deaths, serious adverse events, or discontinuations due to adverse events were reported. There were no significant changes in vital signs or on ECG.
In the second Thyquidity study of 38 healthy adults, after a 10-hour fast, the same doses were given 10 or 30 minutes prior to the consumption of a 950-calorie standardized high-fat breakfast.
Again, over 48 hours, mean serum T4 levels were comparable between the two groups. The geometric least squares mean ratios for both AUC and Cmax for baseline-adjusted LT4 were 88.7% and 85.1%, respectively. Again, the corresponding 90% confidence intervals fell within the FDA’s noninterference definition, again demonstrating lack of a food effect on bioavailability, Dr. Washington noted.
Four adverse events were reported in three participants, with three deemed to be possibly related to the medication. All were isolated lab abnormalities without clinical symptoms and deemed to be mild. Three were normal on repeat testing.
There were no deaths or serious adverse events or study discontinuations for adverse events and no significant findings for vital signs or on ECG.
Similar findings for Tirosint-SOL but longer-term studies needed
The recently published Tirosint-SOL study included 36 healthy volunteers randomized to single 600-µg doses of the LT4 oral solution after a 10-hour fast, either 15 or 30 minutes before eating a standardized high-fat, high-calorie meal. Mean serum total thyroxine concentration profiles were similar for both the 15- and 30-minute waits, with similar AUCs.
Geometric mean ratios for AUCs at 48 and 72 hours were 90% and 92%, respectively, and the 90% confidence intervals fell within the 80%-125% FDA boundaries, suggesting similar exposures whether taken 15 or 30 minutes before a meal.
Senior author Francesco S. Celi, MD, chair of the division of endocrinology, diabetes, and metabolism at Virginia Commonwealth University, Richmond, told this news organization: “There is an interest in providing more opportunities for patients and improving adherence to the medication. ... Whatever makes life a bit easier for patients and results in a more predictable response to treatment means down the road there will be fewer visits to the doctor to make adjustments.”
However, he said that in addition to the cost and reimbursement issue, all of these studies have been short term and not conducted in real-life settings.
“Another question is: What happens if the patient goes on low-dose LT4? The studies were conducted on much higher pharmacologic doses. But at least from a safety standpoint, there’s no specific concern.”
Dr. Washington is an employee of Vertice Pharma. Dr. Celi has received unrestricted research grants and worked as a consultant for IBSA. Dr. Gigliotti has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
ATLANTA -- Liquid formulations of levothyroxine offer the possibility of allowing patients with hypothyroidism to take their medication with meals or coffee and skip the currently recommended 30- to 60-minute waiting period before doing either, new data suggest.
Because food, coffee, and certain medications can interfere with intestinal absorption of levothyroxine (also known as LT4), current guidelines recommend that the drug be taken in a fasting state, typically 30-60 minutes before breakfast. However, compliance may be difficult for some patients.
Now, a potential solution may come from new evidence that liquid levothyroxine formulations that bypass the gastric dissolution phase of absorption may mitigate the interference with food and coffee.
Findings from two bioavailability studies showing no difference in comparisons of Thyquidity (levothyroxine sodium oral solution, Vertice Pharma) with or without waiting periods before consuming coffee or a high-fat meal were presented at the annual meeting of the Endocrine Society (ENDO 2022), by Vertice Pharma Medical Director Kris Washington, PharmD.
And just last month, similar data were published in Thyroid for another levothyroxine oral solution, Tirosint-SOL (IBSA). No difference in pharmacokinetic properties were found with this product with a shorter versus a longer waiting period before consuming a high-fat meal.
Liquid thyroxine may be less affected by food/drink but is expensive
Both products have been approved by the U.S. Food and Drug Administration, but current labeling for both still calls for a 30- to 60-minute waiting period between taking the medication and eating or drinking. Thyquidity is an oral solution of 100 µg/mL levothyroxine sodium that has been shown to be bioequivalent to one of the most popular branded levothyroxine tablets, Synthroid (AbbVie), under fasting conditions. Tirosint-SOL is also an oral solution that comes in 15 different dosage ampules.
“It is important to note that while these findings are exciting and encouraging, we do want you to continue to follow the current FDA-approved label for Thyquidity, recommending that it be taken on an empty stomach 30-60 minutes prior to breakfast and that patients continue to follow all other label instructions,” Dr. Washington said during a press briefing at ENDO 2022.
When asked whether the new data would be submitted to the FDA for a possible amendment to this message, she replied: “We’re still discussing that. We’re exploring all options. ... This is fairly new data. ... It makes sense and certainly solves a lot of the challenges for people who can’t swallow or don’t choose to swallow, or the challenges of splitting or crushing with tablets.”
Asked to comment, Benjamin J. Gigliotti, MD, a clinical thyroidologist at the University of Rochester, New York, told this news organization: “Liquid levothyroxine has the potential to be a clinically useful formulation,” noting that these recent data corroborate prior findings from Europe and elsewhere that liquid levothyroxine is absorbed more rapidly and thus may be less impacted by food or beverages.
However, Dr. Gigliotti also pointed out, “I don’t think malabsorption is a major contributor to suboptimal treatment because if [patients] malabsorb the hormone, we typically just increase their dose a little bit or ask them to take it separately, and that works just fine for most people.”
And the higher cost of the liquid products is a major issue, he noted.
A quick search on GoodRx shows that the lowest price of Tirosint-SOL is $115.52 for a 1 month supply and Thyquidity is $181.04/month. “In the few patients where I tried to obtain Tirosint-SOL, it was not covered by insurance, even with a prior authorization,” Dr. Gigliotti commented.
In contrast, generic levothyroxine tablets are about $4/month, while a common brand name of levothyroxine tablets are $47.81/month.
“Until these liquid formulations are more widely covered by insurance for a reasonable copay, or come down in price compared to generic levothyroxine tablets, most of my patients have voiced that they’d rather deal with the inconveniences of a tablet compared to higher medication cost, especially with rising economic insecurity imposed by the COVID-19 pandemic and recent world events,” Dr. Gigliotti said.
Bioequivalence with shorter versus longer waits before coffee/breakfast
The Thyquidity coffee study was a single-center open-label, randomized, crossover study of 40 healthy adults randomized after a 10-hour overnight fast to 600 µg Thyquidity with water under fasting conditions or to the same dose given 5 minutes prior to drinking an 8-ounce cup of American coffee without milk or sweeteners. After a 40-day washout period, the same participants received the other treatment.
Mean serum thyroxine (T4) concentrations over 48 hours were nearly identical, demonstrating comparable bioavailability. Pharmacokinetics parameters, including area under the curve (AUC) and Cmax, were also comparable for both groups. The geometric least square mean ratios for baseline-adjusted LT4 were 96.0% for Cmax and 94% for AUC. And the corresponding 90% confidence intervals fell within the 80%-125% FDA acceptance range for absence of a food effect on bioavailability, said Dr. Washington when presenting the findings.
There was one adverse event, a decrease in blood glucose level, which was deemed to be mild and unrelated to study treatment. No deaths, serious adverse events, or discontinuations due to adverse events were reported. There were no significant changes in vital signs or on ECG.
In the second Thyquidity study of 38 healthy adults, after a 10-hour fast, the same doses were given 10 or 30 minutes prior to the consumption of a 950-calorie standardized high-fat breakfast.
Again, over 48 hours, mean serum T4 levels were comparable between the two groups. The geometric least squares mean ratios for both AUC and Cmax for baseline-adjusted LT4 were 88.7% and 85.1%, respectively. Again, the corresponding 90% confidence intervals fell within the FDA’s noninterference definition, again demonstrating lack of a food effect on bioavailability, Dr. Washington noted.
Four adverse events were reported in three participants, with three deemed to be possibly related to the medication. All were isolated lab abnormalities without clinical symptoms and deemed to be mild. Three were normal on repeat testing.
There were no deaths or serious adverse events or study discontinuations for adverse events and no significant findings for vital signs or on ECG.
Similar findings for Tirosint-SOL but longer-term studies needed
The recently published Tirosint-SOL study included 36 healthy volunteers randomized to single 600-µg doses of the LT4 oral solution after a 10-hour fast, either 15 or 30 minutes before eating a standardized high-fat, high-calorie meal. Mean serum total thyroxine concentration profiles were similar for both the 15- and 30-minute waits, with similar AUCs.
Geometric mean ratios for AUCs at 48 and 72 hours were 90% and 92%, respectively, and the 90% confidence intervals fell within the 80%-125% FDA boundaries, suggesting similar exposures whether taken 15 or 30 minutes before a meal.
Senior author Francesco S. Celi, MD, chair of the division of endocrinology, diabetes, and metabolism at Virginia Commonwealth University, Richmond, told this news organization: “There is an interest in providing more opportunities for patients and improving adherence to the medication. ... Whatever makes life a bit easier for patients and results in a more predictable response to treatment means down the road there will be fewer visits to the doctor to make adjustments.”
However, he said that in addition to the cost and reimbursement issue, all of these studies have been short term and not conducted in real-life settings.
“Another question is: What happens if the patient goes on low-dose LT4? The studies were conducted on much higher pharmacologic doses. But at least from a safety standpoint, there’s no specific concern.”
Dr. Washington is an employee of Vertice Pharma. Dr. Celi has received unrestricted research grants and worked as a consultant for IBSA. Dr. Gigliotti has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ENDO 2022
New National Lipid Association statement on statin intolerance
The U.S. National Lipid Association has issued a new scientific statement on the management of patients with statin intolerance, which recommends different strategies to help patients stay on statin medications, and also suggests alternatives that can be used in patients who really cannot tolerate statin drugs.
The statement was published online in the Journal of Clinical Lipidology.
It notes that, although statins are generally well tolerated, statin intolerance is reported in 5%-30% of patients and contributes to reduced statin adherence and persistence, as well as higher risk for adverse cardiovascular outcomes.
The statement acknowledges the importance of identifying modifiable risk factors for statin intolerance and recognizes the possibility of a “nocebo” effect, basically the patient expectation of harm resulting in perceived side effects.
To identify a tolerable statin regimen, it recommends that clinicians consider using several different strategies (different statin, dose, and/or dosing frequency), and to classify a patient as having statin intolerance, a minimum of two statins should have been attempted, including at least one at the lowest-approved daily dosage.
The statement says that nonstatin therapy may be required for patients who cannot reach therapeutic objectives with lifestyle and maximal tolerated statin therapy, and in these cases, therapies with outcomes data from randomized trials showing reduced cardiovascular events are favored.
In high and very high-risk patients who are statin intolerant, clinicians should consider initiating nonstatin therapy while additional attempts are made to identify a tolerable statin in order to limit the time of exposure to elevated levels of atherogenic lipoproteins, it suggests.
“There is strong evidence that statins reduce risk of cardiovascular events particularly in patients with atherosclerotic cardiovascular disease, but recent research shows that only about half of patients with ASCVD are on a statin,” Kevin C. Maki, PhD, coauthor of the statement and current president of the National Lipid Association, said in an interview.
“There is an urgent problem with underutilization of statins and undertreatment of ASCVD. And we know that perceived side effects associated with statins are a common reason for discontinuation of these drugs and the consequent failure to manage ASCVD adequately,” he said.
Dr. Maki noted that the NLA’s first message is that, when experiencing symptoms taking statins, a large majority of patients can still tolerate a statin. “They can try a different agent or a different dose. But for those who still can’t tolerate a statin, we then recommend nonstatin therapies and we favor those therapies with evidence from randomized trials.”
He pointed out that many patients who believe they are experiencing side effects from taking statins still experience the same effects on a placebo, a condition known as the nocebo effect.
“Several studies have shown that the nocebo effect is very common and accounts for more than half of perceived statin side effects. It is therefore estimated that many of the complaints of statin intolerance are probably not directly related to the pharmacodynamic actions of the drugs,” Dr. Maki said.
One recent study on the nocebo effect, the SAMSON study, suggested that 90% of symptoms attributed to statins were elicited by placebo tablets too.
But Dr. Maki added that it can be a losing battle for the clinician if patients think their symptoms are related to taking a statin.
“We suggest that clinicians inform patients that most people can tolerate a statin – maybe with a different agent or an alternative dose – and it is really important to lower LDL cholesterol as that will lower the risk of MI and stroke, so we need to find a regimen that works for each individual,” he said. “Most people can find a regimen that works. If this means taking a lower dose of a statin, they can take some additional therapy as well. This is a better situation than stopping taking statins altogether and allowing ASCVD to progress.”
Dr. Maki stressed that statins should still be the first choice as they are effective, taken orally, and inexpensive.
“Other medications do not have all these advantages. For example, PCSK9 inhibitors are very effective but they are expensive and injectable,” he noted. “And while ezetimibe [Zetia] is now generic so inexpensive, it has a more modest effect on LDL-lowering compared to statins, so by itself it is not normally enough for most patients to get to their target LDL, but it is an option for use in combination with a statin.”
He added that the NLA message is to do everything possible to keep patients on a statin, especially patients with preexisting ASCVD.
“We would like these patients to be on high-intensity statins. If they really can’t tolerate this, then they could be on a low-intensity statin plus an additional agent.”
Commenting on the NLA statement, SAMSON study coauthor James Howard, MB BChir, PhD, Imperial College London, said he had reservations about some of the recommendations.
“Whilst I think it is great news that the existence and importance of the nocebo effect is increasingly recognized in international guidelines and statements, I think we need to be very careful about recommending reduced doses and frequencies of statins,” Dr. Howard said.
“Studies such as SAMSON and StatinWISE indicate the vast majority of side effects reported by patients taking statins are not caused by the statin molecule, but instead are caused by either the nocebo effect, or ever-present background symptoms that are wrongly attributed to the statins,” he commented. “Therefore, to recommend that the correct approach in a patient with a history of MI suffering symptoms on 80 mg of atorvastatin is to reduce the dose or try alternate daily dosing. This reinforces the view that these drugs are side-effect prone and need to be carefully titrated.”
Dr. Howard suggested that patients should be educated on the possibility of the nocebo effect or background symptoms and encouraged to retrial statins at the same dose. “If that doesn’t work, then formal recording with a symptom diary might help patients recognize background symptoms,” he added.
Dr. Howard noted that, if symptoms still persist, an “n-of-1” trial could be conducted, in which the patient rotates between multiple periods of taking a statin and a placebo, but he acknowledged that this is expensive and time consuming.
Also commenting, Steve Nissen, MD, Cleveland Clinic, said he thought the NLA statement was “reasonable and thoughtful.”
“Regardless of whether the symptoms are due to the nocebo effect or not, some patients will just not take a statin no matter how hard you try to convince them to persevere, so we do need alternatives,” Dr. Nissen said.
He noted that current alternatives would include the PCSK9 inhibitors and ezetimibe, but a future candidate could be the oral bempedoic acid (Nexletol), which is currently being evaluated in a large outcomes trial (CLEAR Outcomes).
A version of this article first appeared on Medscape.com.
The U.S. National Lipid Association has issued a new scientific statement on the management of patients with statin intolerance, which recommends different strategies to help patients stay on statin medications, and also suggests alternatives that can be used in patients who really cannot tolerate statin drugs.
The statement was published online in the Journal of Clinical Lipidology.
It notes that, although statins are generally well tolerated, statin intolerance is reported in 5%-30% of patients and contributes to reduced statin adherence and persistence, as well as higher risk for adverse cardiovascular outcomes.
The statement acknowledges the importance of identifying modifiable risk factors for statin intolerance and recognizes the possibility of a “nocebo” effect, basically the patient expectation of harm resulting in perceived side effects.
To identify a tolerable statin regimen, it recommends that clinicians consider using several different strategies (different statin, dose, and/or dosing frequency), and to classify a patient as having statin intolerance, a minimum of two statins should have been attempted, including at least one at the lowest-approved daily dosage.
The statement says that nonstatin therapy may be required for patients who cannot reach therapeutic objectives with lifestyle and maximal tolerated statin therapy, and in these cases, therapies with outcomes data from randomized trials showing reduced cardiovascular events are favored.
In high and very high-risk patients who are statin intolerant, clinicians should consider initiating nonstatin therapy while additional attempts are made to identify a tolerable statin in order to limit the time of exposure to elevated levels of atherogenic lipoproteins, it suggests.
“There is strong evidence that statins reduce risk of cardiovascular events particularly in patients with atherosclerotic cardiovascular disease, but recent research shows that only about half of patients with ASCVD are on a statin,” Kevin C. Maki, PhD, coauthor of the statement and current president of the National Lipid Association, said in an interview.
“There is an urgent problem with underutilization of statins and undertreatment of ASCVD. And we know that perceived side effects associated with statins are a common reason for discontinuation of these drugs and the consequent failure to manage ASCVD adequately,” he said.
Dr. Maki noted that the NLA’s first message is that, when experiencing symptoms taking statins, a large majority of patients can still tolerate a statin. “They can try a different agent or a different dose. But for those who still can’t tolerate a statin, we then recommend nonstatin therapies and we favor those therapies with evidence from randomized trials.”
He pointed out that many patients who believe they are experiencing side effects from taking statins still experience the same effects on a placebo, a condition known as the nocebo effect.
“Several studies have shown that the nocebo effect is very common and accounts for more than half of perceived statin side effects. It is therefore estimated that many of the complaints of statin intolerance are probably not directly related to the pharmacodynamic actions of the drugs,” Dr. Maki said.
One recent study on the nocebo effect, the SAMSON study, suggested that 90% of symptoms attributed to statins were elicited by placebo tablets too.
But Dr. Maki added that it can be a losing battle for the clinician if patients think their symptoms are related to taking a statin.
“We suggest that clinicians inform patients that most people can tolerate a statin – maybe with a different agent or an alternative dose – and it is really important to lower LDL cholesterol as that will lower the risk of MI and stroke, so we need to find a regimen that works for each individual,” he said. “Most people can find a regimen that works. If this means taking a lower dose of a statin, they can take some additional therapy as well. This is a better situation than stopping taking statins altogether and allowing ASCVD to progress.”
Dr. Maki stressed that statins should still be the first choice as they are effective, taken orally, and inexpensive.
“Other medications do not have all these advantages. For example, PCSK9 inhibitors are very effective but they are expensive and injectable,” he noted. “And while ezetimibe [Zetia] is now generic so inexpensive, it has a more modest effect on LDL-lowering compared to statins, so by itself it is not normally enough for most patients to get to their target LDL, but it is an option for use in combination with a statin.”
He added that the NLA message is to do everything possible to keep patients on a statin, especially patients with preexisting ASCVD.
“We would like these patients to be on high-intensity statins. If they really can’t tolerate this, then they could be on a low-intensity statin plus an additional agent.”
Commenting on the NLA statement, SAMSON study coauthor James Howard, MB BChir, PhD, Imperial College London, said he had reservations about some of the recommendations.
“Whilst I think it is great news that the existence and importance of the nocebo effect is increasingly recognized in international guidelines and statements, I think we need to be very careful about recommending reduced doses and frequencies of statins,” Dr. Howard said.
“Studies such as SAMSON and StatinWISE indicate the vast majority of side effects reported by patients taking statins are not caused by the statin molecule, but instead are caused by either the nocebo effect, or ever-present background symptoms that are wrongly attributed to the statins,” he commented. “Therefore, to recommend that the correct approach in a patient with a history of MI suffering symptoms on 80 mg of atorvastatin is to reduce the dose or try alternate daily dosing. This reinforces the view that these drugs are side-effect prone and need to be carefully titrated.”
Dr. Howard suggested that patients should be educated on the possibility of the nocebo effect or background symptoms and encouraged to retrial statins at the same dose. “If that doesn’t work, then formal recording with a symptom diary might help patients recognize background symptoms,” he added.
Dr. Howard noted that, if symptoms still persist, an “n-of-1” trial could be conducted, in which the patient rotates between multiple periods of taking a statin and a placebo, but he acknowledged that this is expensive and time consuming.
Also commenting, Steve Nissen, MD, Cleveland Clinic, said he thought the NLA statement was “reasonable and thoughtful.”
“Regardless of whether the symptoms are due to the nocebo effect or not, some patients will just not take a statin no matter how hard you try to convince them to persevere, so we do need alternatives,” Dr. Nissen said.
He noted that current alternatives would include the PCSK9 inhibitors and ezetimibe, but a future candidate could be the oral bempedoic acid (Nexletol), which is currently being evaluated in a large outcomes trial (CLEAR Outcomes).
A version of this article first appeared on Medscape.com.
The U.S. National Lipid Association has issued a new scientific statement on the management of patients with statin intolerance, which recommends different strategies to help patients stay on statin medications, and also suggests alternatives that can be used in patients who really cannot tolerate statin drugs.
The statement was published online in the Journal of Clinical Lipidology.
It notes that, although statins are generally well tolerated, statin intolerance is reported in 5%-30% of patients and contributes to reduced statin adherence and persistence, as well as higher risk for adverse cardiovascular outcomes.
The statement acknowledges the importance of identifying modifiable risk factors for statin intolerance and recognizes the possibility of a “nocebo” effect, basically the patient expectation of harm resulting in perceived side effects.
To identify a tolerable statin regimen, it recommends that clinicians consider using several different strategies (different statin, dose, and/or dosing frequency), and to classify a patient as having statin intolerance, a minimum of two statins should have been attempted, including at least one at the lowest-approved daily dosage.
The statement says that nonstatin therapy may be required for patients who cannot reach therapeutic objectives with lifestyle and maximal tolerated statin therapy, and in these cases, therapies with outcomes data from randomized trials showing reduced cardiovascular events are favored.
In high and very high-risk patients who are statin intolerant, clinicians should consider initiating nonstatin therapy while additional attempts are made to identify a tolerable statin in order to limit the time of exposure to elevated levels of atherogenic lipoproteins, it suggests.
“There is strong evidence that statins reduce risk of cardiovascular events particularly in patients with atherosclerotic cardiovascular disease, but recent research shows that only about half of patients with ASCVD are on a statin,” Kevin C. Maki, PhD, coauthor of the statement and current president of the National Lipid Association, said in an interview.
“There is an urgent problem with underutilization of statins and undertreatment of ASCVD. And we know that perceived side effects associated with statins are a common reason for discontinuation of these drugs and the consequent failure to manage ASCVD adequately,” he said.
Dr. Maki noted that the NLA’s first message is that, when experiencing symptoms taking statins, a large majority of patients can still tolerate a statin. “They can try a different agent or a different dose. But for those who still can’t tolerate a statin, we then recommend nonstatin therapies and we favor those therapies with evidence from randomized trials.”
He pointed out that many patients who believe they are experiencing side effects from taking statins still experience the same effects on a placebo, a condition known as the nocebo effect.
“Several studies have shown that the nocebo effect is very common and accounts for more than half of perceived statin side effects. It is therefore estimated that many of the complaints of statin intolerance are probably not directly related to the pharmacodynamic actions of the drugs,” Dr. Maki said.
One recent study on the nocebo effect, the SAMSON study, suggested that 90% of symptoms attributed to statins were elicited by placebo tablets too.
But Dr. Maki added that it can be a losing battle for the clinician if patients think their symptoms are related to taking a statin.
“We suggest that clinicians inform patients that most people can tolerate a statin – maybe with a different agent or an alternative dose – and it is really important to lower LDL cholesterol as that will lower the risk of MI and stroke, so we need to find a regimen that works for each individual,” he said. “Most people can find a regimen that works. If this means taking a lower dose of a statin, they can take some additional therapy as well. This is a better situation than stopping taking statins altogether and allowing ASCVD to progress.”
Dr. Maki stressed that statins should still be the first choice as they are effective, taken orally, and inexpensive.
“Other medications do not have all these advantages. For example, PCSK9 inhibitors are very effective but they are expensive and injectable,” he noted. “And while ezetimibe [Zetia] is now generic so inexpensive, it has a more modest effect on LDL-lowering compared to statins, so by itself it is not normally enough for most patients to get to their target LDL, but it is an option for use in combination with a statin.”
He added that the NLA message is to do everything possible to keep patients on a statin, especially patients with preexisting ASCVD.
“We would like these patients to be on high-intensity statins. If they really can’t tolerate this, then they could be on a low-intensity statin plus an additional agent.”
Commenting on the NLA statement, SAMSON study coauthor James Howard, MB BChir, PhD, Imperial College London, said he had reservations about some of the recommendations.
“Whilst I think it is great news that the existence and importance of the nocebo effect is increasingly recognized in international guidelines and statements, I think we need to be very careful about recommending reduced doses and frequencies of statins,” Dr. Howard said.
“Studies such as SAMSON and StatinWISE indicate the vast majority of side effects reported by patients taking statins are not caused by the statin molecule, but instead are caused by either the nocebo effect, or ever-present background symptoms that are wrongly attributed to the statins,” he commented. “Therefore, to recommend that the correct approach in a patient with a history of MI suffering symptoms on 80 mg of atorvastatin is to reduce the dose or try alternate daily dosing. This reinforces the view that these drugs are side-effect prone and need to be carefully titrated.”
Dr. Howard suggested that patients should be educated on the possibility of the nocebo effect or background symptoms and encouraged to retrial statins at the same dose. “If that doesn’t work, then formal recording with a symptom diary might help patients recognize background symptoms,” he added.
Dr. Howard noted that, if symptoms still persist, an “n-of-1” trial could be conducted, in which the patient rotates between multiple periods of taking a statin and a placebo, but he acknowledged that this is expensive and time consuming.
Also commenting, Steve Nissen, MD, Cleveland Clinic, said he thought the NLA statement was “reasonable and thoughtful.”
“Regardless of whether the symptoms are due to the nocebo effect or not, some patients will just not take a statin no matter how hard you try to convince them to persevere, so we do need alternatives,” Dr. Nissen said.
He noted that current alternatives would include the PCSK9 inhibitors and ezetimibe, but a future candidate could be the oral bempedoic acid (Nexletol), which is currently being evaluated in a large outcomes trial (CLEAR Outcomes).
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL LIPIDOLOGY





