User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
CDC cuts back hospital data reporting on COVID
When the federal government’s public health emergency (PHE) ended on May 11, the Centers for Disease Control and Prevention scaled back the amount of COVID-related data that it had required hospitals to collect and report during the previous 3 years. The CDC had to do this, an agency spokesman said in an interview, because “CDC’s authorizations to collect certain types of public health data” expired with the PHE.
The CDC insists that it will have enough data to keep up with the virus, which repeatedly defied scientists’ expectations during the course of the pandemic. But some experts have doubts about whether this will turn out to be the case.
While the COVID pandemic is subsiding and transitioning to an endemic phase, many things about the coronavirus are still not understood, noted Marisa Eisenberg, PhD, associate professor of epidemiology at the University of Michigan, Ann Arbor.
“COVID is here to stay, and it ebbs and flows but is staying at fairly consistent levels across the country,” she said in an interview. “Meanwhile, we haven’t established a regular seasonality for COVID that we see for most other respiratory illnesses. We’re still seeing pretty rapidly invading new waves of variants. With flu and other respiratory illnesses, you often see a particular variant in each season. There’s an established pattern. For COVID, that’s still shifting.”
Similarly, Sam Scarpino, PhD, a public health expert at Northeastern University, Boston, told the New York Times: “The CDC is shuffling COVID into the deck of infectious diseases that we’re satisfied living with. One thousand deaths a week is just unacceptable.”
William Schaffner, MD, a professor of preventive medicine and health policy at Vanderbilt University Medical Center, Nashville, Tenn., said in an interview that “how we deal with influenza is something of a template or a model for what the CDC is trying to get to with COVID.” It’s not practical for physicians and hospitals to report every flu case, and the same is now true for COVID. However, “we’re still asking for data on people who are hospitalized with COVID to be reported. That will give us a measure of the major public health impact.”
Dr. Eisenberg doesn’t fully subscribe to this notion. “COVID and influenza are both respiratory illnesses, and our initial pandemic response was based on playbooks that we’d built for potential flu pandemics. But COVID is not the flu. We still have to grapple with the fact that it’s killing a lot more people than the flu does. So maybe it’s a template, but not a perfect one.”
What data is being deleted
The CDC is now requiring hospitals to submit COVID-related data weekly, rather than daily, as it previously had. In addition, the agency has cut the number of data elements that hospitals must report from 62 to 44. Among the data fields that are now optional for hospitals to report are the numbers of hospitalized children with suspected or lab-confirmed COVID; hospitalized and ventilated COVID patients; adults in the ICU with suspected or lab-confirmed COVID; adult and pediatric admissions with suspected COVID; COVID-related emergency department visits; and inpatients with hospital-acquired COVID.
Although widely feared by health care workers and the public, hospital-acquired COVID has never been a major factor in the pandemic, Dr. Schaffner said. “So why ask for something that’s actually not so critical? Let’s keep the emphasis on rapid, accurate reporting of people who are hospitalized because of this disease.”
Akin Demehin, senior director for quality and patient safety policy for the American Hospital Association, agreed that the rate of hospital-acquired COVID cases “has been very low throughout the pandemic.” That was one reason why CDC made this measure optional.
Dr. Eisenberg concurred with this view. “We worried about [hospital-acquired COVID] a lot, and then, because people were very careful, it wasn’t as much of a problem as we feared it would be.” But she added a note of caution: “Masking and other [preventive guidelines] are shifting in hospitals, so it will be interesting to see whether that affects things.”
CDC justifies its new policy
To put the hospital data reporting changes in context, it’s important to know that CDC will no longer directly track community levels of COVID and the percentage of tests that come back positive for COVID, which until now were used to measure transmission rates. (Laboratories no longer have to report these test data, whether they are in hospitals or in the community.) To track death rates, CDC will rely on the National Vital Statistics System, which is accurate but lags other kinds of surveillance by 2-3 weeks, according to the New York Times.
In a recent MMWR report, CDC defended its new COVID surveillance system, saying: “Weekly COVID-19 hospital admission levels and the percentage of all COVID-19–associated deaths will be primary surveillance indicators. Emergency department visits and percentage of positive SARS-CoV-2 laboratory test results will help detect early changes in trends. Genomic surveillance will continue to help identify and monitor SARS-CoV-2 variants.”
Clarifying the latter point, CDC said that national genomic surveillance, along with wastewater surveillance, will continue to be used to estimate COVID variant proportions. Dr. Eisenberg stressed the importance of genomic surveillance at the hundreds of sites that CDC now maintains across the country. But currently, many of these sites are only monitoring the level of COVID.
CDC also observed that COVID-19 hospital admission levels have been shown to be “concordant” with community levels of SARS-CoV-2 infection. Therefore, rates of COVID-associated admissions and the percentages of positive test results, COVID ED visits, and COVID deaths are “suitable and timely indicators of trends in COVID-19 activity and severity.”
Ready to shift to voluntary reporting?
In a news release, AHA praised the “streamlining” of CDC requirements for data reporting but said that it hoped that mandatory reporting would be phased out as soon as possible.
The association noted that this would require action by the Centers for Medicare & Medicaid Services. CMS now enforces the CDC requirements with a “condition of participation” (COP) provision, by which noncompliant hospitals could be excluded from Medicare. CMS has extended this COP to April 30, 2024, although it could choose to ask the Secretary of Health and Human Services to terminate it earlier.
If mandatory reporting were repealed, would most hospitals still report on the key COVID metrics? Mr. Demehin noted that before CMS implemented its COP, hospitals reported COVID data voluntarily, “and the participation rate was well over 90%. So setting up a mechanism similar to that is something we’ve encouraged CMS to consider.”
Dr. Eisenberg is skeptical. While bigger hospitals with more resources might continue reporting voluntarily, she said, safety-net hospitals in underserved areas might not, because they are especially short staffed. “Then you have disparities in which hospitals will report.”
Vaccinations: The sleeping dragon
COVID continues to ravage the nation. According to CDC statistics, there were 1,109 deaths from COVID in the U.S. in the week ending May 6, and total deaths have hit 1.13 million. There were 1,333 new COVID-related hospital admissions, and 7,261 people were in the hospital because of COVID.
Another eye-catching number: Only 16.9% of the U.S. population has received an updated COVID vaccine booster. Dr. Schaffner thinks that this is what we should really keep our eye on. While the combination of vaccinations and widespread SARS-CoV-2 infections has conferred herd immunity on most Americans, he said it’s temporary. “Whether your immunity comes from the virus and recovery from disease or from the vaccines, that immunity will wane over time. Unless we keep our vaccination rate up, we may see more future cases. We’ll have to see how that works out. But I’m nervous about that, because people do appear to be nonchalant.”
A version of this article first appeared on Medscape.com.
When the federal government’s public health emergency (PHE) ended on May 11, the Centers for Disease Control and Prevention scaled back the amount of COVID-related data that it had required hospitals to collect and report during the previous 3 years. The CDC had to do this, an agency spokesman said in an interview, because “CDC’s authorizations to collect certain types of public health data” expired with the PHE.
The CDC insists that it will have enough data to keep up with the virus, which repeatedly defied scientists’ expectations during the course of the pandemic. But some experts have doubts about whether this will turn out to be the case.
While the COVID pandemic is subsiding and transitioning to an endemic phase, many things about the coronavirus are still not understood, noted Marisa Eisenberg, PhD, associate professor of epidemiology at the University of Michigan, Ann Arbor.
“COVID is here to stay, and it ebbs and flows but is staying at fairly consistent levels across the country,” she said in an interview. “Meanwhile, we haven’t established a regular seasonality for COVID that we see for most other respiratory illnesses. We’re still seeing pretty rapidly invading new waves of variants. With flu and other respiratory illnesses, you often see a particular variant in each season. There’s an established pattern. For COVID, that’s still shifting.”
Similarly, Sam Scarpino, PhD, a public health expert at Northeastern University, Boston, told the New York Times: “The CDC is shuffling COVID into the deck of infectious diseases that we’re satisfied living with. One thousand deaths a week is just unacceptable.”
William Schaffner, MD, a professor of preventive medicine and health policy at Vanderbilt University Medical Center, Nashville, Tenn., said in an interview that “how we deal with influenza is something of a template or a model for what the CDC is trying to get to with COVID.” It’s not practical for physicians and hospitals to report every flu case, and the same is now true for COVID. However, “we’re still asking for data on people who are hospitalized with COVID to be reported. That will give us a measure of the major public health impact.”
Dr. Eisenberg doesn’t fully subscribe to this notion. “COVID and influenza are both respiratory illnesses, and our initial pandemic response was based on playbooks that we’d built for potential flu pandemics. But COVID is not the flu. We still have to grapple with the fact that it’s killing a lot more people than the flu does. So maybe it’s a template, but not a perfect one.”
What data is being deleted
The CDC is now requiring hospitals to submit COVID-related data weekly, rather than daily, as it previously had. In addition, the agency has cut the number of data elements that hospitals must report from 62 to 44. Among the data fields that are now optional for hospitals to report are the numbers of hospitalized children with suspected or lab-confirmed COVID; hospitalized and ventilated COVID patients; adults in the ICU with suspected or lab-confirmed COVID; adult and pediatric admissions with suspected COVID; COVID-related emergency department visits; and inpatients with hospital-acquired COVID.
Although widely feared by health care workers and the public, hospital-acquired COVID has never been a major factor in the pandemic, Dr. Schaffner said. “So why ask for something that’s actually not so critical? Let’s keep the emphasis on rapid, accurate reporting of people who are hospitalized because of this disease.”
Akin Demehin, senior director for quality and patient safety policy for the American Hospital Association, agreed that the rate of hospital-acquired COVID cases “has been very low throughout the pandemic.” That was one reason why CDC made this measure optional.
Dr. Eisenberg concurred with this view. “We worried about [hospital-acquired COVID] a lot, and then, because people were very careful, it wasn’t as much of a problem as we feared it would be.” But she added a note of caution: “Masking and other [preventive guidelines] are shifting in hospitals, so it will be interesting to see whether that affects things.”
CDC justifies its new policy
To put the hospital data reporting changes in context, it’s important to know that CDC will no longer directly track community levels of COVID and the percentage of tests that come back positive for COVID, which until now were used to measure transmission rates. (Laboratories no longer have to report these test data, whether they are in hospitals or in the community.) To track death rates, CDC will rely on the National Vital Statistics System, which is accurate but lags other kinds of surveillance by 2-3 weeks, according to the New York Times.
In a recent MMWR report, CDC defended its new COVID surveillance system, saying: “Weekly COVID-19 hospital admission levels and the percentage of all COVID-19–associated deaths will be primary surveillance indicators. Emergency department visits and percentage of positive SARS-CoV-2 laboratory test results will help detect early changes in trends. Genomic surveillance will continue to help identify and monitor SARS-CoV-2 variants.”
Clarifying the latter point, CDC said that national genomic surveillance, along with wastewater surveillance, will continue to be used to estimate COVID variant proportions. Dr. Eisenberg stressed the importance of genomic surveillance at the hundreds of sites that CDC now maintains across the country. But currently, many of these sites are only monitoring the level of COVID.
CDC also observed that COVID-19 hospital admission levels have been shown to be “concordant” with community levels of SARS-CoV-2 infection. Therefore, rates of COVID-associated admissions and the percentages of positive test results, COVID ED visits, and COVID deaths are “suitable and timely indicators of trends in COVID-19 activity and severity.”
Ready to shift to voluntary reporting?
In a news release, AHA praised the “streamlining” of CDC requirements for data reporting but said that it hoped that mandatory reporting would be phased out as soon as possible.
The association noted that this would require action by the Centers for Medicare & Medicaid Services. CMS now enforces the CDC requirements with a “condition of participation” (COP) provision, by which noncompliant hospitals could be excluded from Medicare. CMS has extended this COP to April 30, 2024, although it could choose to ask the Secretary of Health and Human Services to terminate it earlier.
If mandatory reporting were repealed, would most hospitals still report on the key COVID metrics? Mr. Demehin noted that before CMS implemented its COP, hospitals reported COVID data voluntarily, “and the participation rate was well over 90%. So setting up a mechanism similar to that is something we’ve encouraged CMS to consider.”
Dr. Eisenberg is skeptical. While bigger hospitals with more resources might continue reporting voluntarily, she said, safety-net hospitals in underserved areas might not, because they are especially short staffed. “Then you have disparities in which hospitals will report.”
Vaccinations: The sleeping dragon
COVID continues to ravage the nation. According to CDC statistics, there were 1,109 deaths from COVID in the U.S. in the week ending May 6, and total deaths have hit 1.13 million. There were 1,333 new COVID-related hospital admissions, and 7,261 people were in the hospital because of COVID.
Another eye-catching number: Only 16.9% of the U.S. population has received an updated COVID vaccine booster. Dr. Schaffner thinks that this is what we should really keep our eye on. While the combination of vaccinations and widespread SARS-CoV-2 infections has conferred herd immunity on most Americans, he said it’s temporary. “Whether your immunity comes from the virus and recovery from disease or from the vaccines, that immunity will wane over time. Unless we keep our vaccination rate up, we may see more future cases. We’ll have to see how that works out. But I’m nervous about that, because people do appear to be nonchalant.”
A version of this article first appeared on Medscape.com.
When the federal government’s public health emergency (PHE) ended on May 11, the Centers for Disease Control and Prevention scaled back the amount of COVID-related data that it had required hospitals to collect and report during the previous 3 years. The CDC had to do this, an agency spokesman said in an interview, because “CDC’s authorizations to collect certain types of public health data” expired with the PHE.
The CDC insists that it will have enough data to keep up with the virus, which repeatedly defied scientists’ expectations during the course of the pandemic. But some experts have doubts about whether this will turn out to be the case.
While the COVID pandemic is subsiding and transitioning to an endemic phase, many things about the coronavirus are still not understood, noted Marisa Eisenberg, PhD, associate professor of epidemiology at the University of Michigan, Ann Arbor.
“COVID is here to stay, and it ebbs and flows but is staying at fairly consistent levels across the country,” she said in an interview. “Meanwhile, we haven’t established a regular seasonality for COVID that we see for most other respiratory illnesses. We’re still seeing pretty rapidly invading new waves of variants. With flu and other respiratory illnesses, you often see a particular variant in each season. There’s an established pattern. For COVID, that’s still shifting.”
Similarly, Sam Scarpino, PhD, a public health expert at Northeastern University, Boston, told the New York Times: “The CDC is shuffling COVID into the deck of infectious diseases that we’re satisfied living with. One thousand deaths a week is just unacceptable.”
William Schaffner, MD, a professor of preventive medicine and health policy at Vanderbilt University Medical Center, Nashville, Tenn., said in an interview that “how we deal with influenza is something of a template or a model for what the CDC is trying to get to with COVID.” It’s not practical for physicians and hospitals to report every flu case, and the same is now true for COVID. However, “we’re still asking for data on people who are hospitalized with COVID to be reported. That will give us a measure of the major public health impact.”
Dr. Eisenberg doesn’t fully subscribe to this notion. “COVID and influenza are both respiratory illnesses, and our initial pandemic response was based on playbooks that we’d built for potential flu pandemics. But COVID is not the flu. We still have to grapple with the fact that it’s killing a lot more people than the flu does. So maybe it’s a template, but not a perfect one.”
What data is being deleted
The CDC is now requiring hospitals to submit COVID-related data weekly, rather than daily, as it previously had. In addition, the agency has cut the number of data elements that hospitals must report from 62 to 44. Among the data fields that are now optional for hospitals to report are the numbers of hospitalized children with suspected or lab-confirmed COVID; hospitalized and ventilated COVID patients; adults in the ICU with suspected or lab-confirmed COVID; adult and pediatric admissions with suspected COVID; COVID-related emergency department visits; and inpatients with hospital-acquired COVID.
Although widely feared by health care workers and the public, hospital-acquired COVID has never been a major factor in the pandemic, Dr. Schaffner said. “So why ask for something that’s actually not so critical? Let’s keep the emphasis on rapid, accurate reporting of people who are hospitalized because of this disease.”
Akin Demehin, senior director for quality and patient safety policy for the American Hospital Association, agreed that the rate of hospital-acquired COVID cases “has been very low throughout the pandemic.” That was one reason why CDC made this measure optional.
Dr. Eisenberg concurred with this view. “We worried about [hospital-acquired COVID] a lot, and then, because people were very careful, it wasn’t as much of a problem as we feared it would be.” But she added a note of caution: “Masking and other [preventive guidelines] are shifting in hospitals, so it will be interesting to see whether that affects things.”
CDC justifies its new policy
To put the hospital data reporting changes in context, it’s important to know that CDC will no longer directly track community levels of COVID and the percentage of tests that come back positive for COVID, which until now were used to measure transmission rates. (Laboratories no longer have to report these test data, whether they are in hospitals or in the community.) To track death rates, CDC will rely on the National Vital Statistics System, which is accurate but lags other kinds of surveillance by 2-3 weeks, according to the New York Times.
In a recent MMWR report, CDC defended its new COVID surveillance system, saying: “Weekly COVID-19 hospital admission levels and the percentage of all COVID-19–associated deaths will be primary surveillance indicators. Emergency department visits and percentage of positive SARS-CoV-2 laboratory test results will help detect early changes in trends. Genomic surveillance will continue to help identify and monitor SARS-CoV-2 variants.”
Clarifying the latter point, CDC said that national genomic surveillance, along with wastewater surveillance, will continue to be used to estimate COVID variant proportions. Dr. Eisenberg stressed the importance of genomic surveillance at the hundreds of sites that CDC now maintains across the country. But currently, many of these sites are only monitoring the level of COVID.
CDC also observed that COVID-19 hospital admission levels have been shown to be “concordant” with community levels of SARS-CoV-2 infection. Therefore, rates of COVID-associated admissions and the percentages of positive test results, COVID ED visits, and COVID deaths are “suitable and timely indicators of trends in COVID-19 activity and severity.”
Ready to shift to voluntary reporting?
In a news release, AHA praised the “streamlining” of CDC requirements for data reporting but said that it hoped that mandatory reporting would be phased out as soon as possible.
The association noted that this would require action by the Centers for Medicare & Medicaid Services. CMS now enforces the CDC requirements with a “condition of participation” (COP) provision, by which noncompliant hospitals could be excluded from Medicare. CMS has extended this COP to April 30, 2024, although it could choose to ask the Secretary of Health and Human Services to terminate it earlier.
If mandatory reporting were repealed, would most hospitals still report on the key COVID metrics? Mr. Demehin noted that before CMS implemented its COP, hospitals reported COVID data voluntarily, “and the participation rate was well over 90%. So setting up a mechanism similar to that is something we’ve encouraged CMS to consider.”
Dr. Eisenberg is skeptical. While bigger hospitals with more resources might continue reporting voluntarily, she said, safety-net hospitals in underserved areas might not, because they are especially short staffed. “Then you have disparities in which hospitals will report.”
Vaccinations: The sleeping dragon
COVID continues to ravage the nation. According to CDC statistics, there were 1,109 deaths from COVID in the U.S. in the week ending May 6, and total deaths have hit 1.13 million. There were 1,333 new COVID-related hospital admissions, and 7,261 people were in the hospital because of COVID.
Another eye-catching number: Only 16.9% of the U.S. population has received an updated COVID vaccine booster. Dr. Schaffner thinks that this is what we should really keep our eye on. While the combination of vaccinations and widespread SARS-CoV-2 infections has conferred herd immunity on most Americans, he said it’s temporary. “Whether your immunity comes from the virus and recovery from disease or from the vaccines, that immunity will wane over time. Unless we keep our vaccination rate up, we may see more future cases. We’ll have to see how that works out. But I’m nervous about that, because people do appear to be nonchalant.”
A version of this article first appeared on Medscape.com.
How providers are adjusting to clinical care post PHE
The first day of seeing patients without a mask was, for Sterling Ransone Jr., MD, “unsettling.”
“I can’t tell you how weird it was the first day that I walked down the hall from my office to where my exam rooms are, to not have a mask on after 3 years of the habit,” said Dr. Ransone, a family physician in Deltaville, Va., and board chair of the American Academy of Family Physicians.
The White House recently lifted the public health emergency order that overhauled the way health care providers operated and advised patients over the past 3 years.
For Dr. Ransone, this transition entails getting used to his bare face, reminding patients of the latest and varying symptoms of the virus, and parting ways with sick patients if they refuse to wear a mask.
As states, hospitals, and health care systems around the country relax their mask mandates for care providers, clinicians will have to fall back on their own policies that patients with potential symptoms mask up.
“Now that it’s up to our offices, we have to have a little bit more backbone,” Dr. Ransone said. “If they’re not willing to follow a health-related policy that will protect the vulnerable, we will not see them. And so for us, it’s been pretty straightforward.”
Despite the policy, Dr. Ransone has cared for patients who don’t disclose they are feeling sick until he enters the room.
“And I wasn’t masked,” Dr. Ransone said. So, “I will wear masks for the rest of the day just to try to protect the rest of my patients in case I was exposed.”
Masks are optional for both patients and staff at the University of Maryland Medical System, but Niharika Khanna, MD, MBBS, said she still wears one with her patients, and her office advises staff to do the same. If patients are experiencing respiratory symptoms, like a cough, they are asked to wear one.
“When the patient first walks up to you, you have no idea what they have,” Dr. Khanna said.
Dr. Khanna is especially mindful of immunocompromised patients who have cancer, and Dr. Ransone cares for several patients who have received kidney transplants and are on potent immunosuppressive drugs.
“I know they’re appreciating our efforts to protect them, and I think the other patients are realizing that it’s a wise thing to do,” Dr. Ransone said.
Some patients have anxiety about the end of masking in doctor offices, but others have been excited about interacting more with their care teams, according to William Dahut, MD, chief scientific officer for the American Cancer Society. Many clinicians will advise their most immunocompromised patients the same as they did prior to the COVID-19 pandemic.
“There’s always been guidelines that oncologists have given to patients who are immunocompromised – we always told them to avoid crowded places, crowded scenes, be outside more than inside,” Dr. Dahut said. “Those general recommendations will continue.”
The AAFP supports masking to limit COVID’s spread, but the “most important thing people can do is to get vaccinated,” Tochi Iroku-Malize, MD, MPH, MBA, president of the AAFP, said.
But the accessibility of vaccinations is also shifting.
Testing shifts
The government will continue to provide free COVID-19 vaccines because it still has supplies on hand. When this stock runs out, commercial insurance providers will be required to cover the immunizations, as they are considered preventive, but people without insurance will have to pay out of pocket.
The AAFP is pushing the Biden administration and Congress to keep the purchase price of those vaccines low enough that clinicians can keep them in stock, according to Dr. Iroku-Malize. Once the federal government transitions COVID-19 vaccines to the commercial market – as early as later in 2023 – it may pose some challenges for providers.
“If the price of the vaccines is too high, physician practices may struggle to make the upfront investment in COVID-19 vaccines,” Dr. Iroku-Malize said. “Patients often prefer to receive vaccine counseling and administration from their usual source of primary care, like their family physician.”
The federal government has also said it still has a supply of treatments for the public to access for free, but has not revealed how much it has on hand or given a timeline for the transition to the private market.
COVID-19 tests, meanwhile, are no longer covered because of the end of the public health emergency, and cost about $45 per kit on average, according to an analysis by the Kaiser Family Foundation.
Pediatrician Lisa Costello, MD, MPH, knows that price point will be a challenge for some families she cares for at West Virginia University Medicine Children’s Hospital in Morgantown. Many still ask her where they can access free tests.
“Testing if you’re a higher risk person is something we need to ensure that people continue to be educated about,” Dr. Costello said.
She’s hopeful that COVID-19 vaccines and treatments such as Paxlovid will stay free in the coming months so patients can continue to easily access them.
Future of telehealth
Relaxed regulations of prescribing controlled substances via telehealth and across state lines allowed clinicians to treat patients near and far during the pandemic. But many providers were worried about a proposal from the Drug Enforcement Administration to clamp down on the prescribing of controlled substances via telehealth, according to A. Mark Fendrick, MD, an internal medicine physician at the University of Michigan, Ann Arbor.
“We were all panicking about what was going to happen to what is for many clinicians a very valuable policy,” Dr. Fendrick said of the telehealth flexibilities introduced during COVID-19.
The DEA, after getting 38,000 comments on their proposed regulations, pulled back on that plan, delaying the cliff until November.
Dr. Fendrick said that telehealth has allowed clinicians to reach patients who have historically faced barriers to care, such as lacking transportation.
“The benefits of that outweigh the potential harms,” he said. “Every policy you make that tightens access because you want to decrease the untoward and unfortunate outcomes will also decrease access to clinical indications.”
The AAFP said it hopes for clear guidance from the DEA in the coming months on what the new telehealth landscape for prescribing will look like.
Medicaid changes
About half of the patients who see Dr. Khanna have insurance through Medicaid.
During the public health emergency, states were not allowed to remove anyone from Medicaid, regardless of whether they no longer qualified for the program or not. But a law passed by Congress last year requires states to once again check Medicaid eligibility. As many as 15 million people could lose their Medicaid coverage.
That could affect the treatments Dr. Khanna recommends for her patients who get kicked off because those who become uninsured or transition to private insurance will have to pay more out of pocket. Maryland will start removals in June.
“When you have an uninsured patient versus Medicaid, it’s a huge difference in what you can ask the patient to do – the medications you can provide, the testing you can provide,” Dr. Khanna said.
States were authorized to remove people from Medicaid as of April 1, with Arkansas, New Hampshire, and South Dakota starting right away. But many states are just now getting the review process going. About a dozen states, including Indiana, Ohio, Utah, and West Virginia, started removing people in May 2023.
Uninsurance rates hit record lows across the United States during the pandemic. Keeping Americans on health insurance is a top priority for the AAFP, Dr. Iroku-Malize said. “We know health care coverage disruptions prevent people from seeking and accessing the care they need.”
Many people who are removed from Medicaid will be eligible for health insurance through employers, or through the Affordable Care Act’s private marketplace. But premiums and deductibles are often higher in these plans, which studies have shown result in patients delaying medical visits and not filling prescriptions or receiving treatment.
Staying mindful
Hospitals that receive federal funds will still have to report COVID-19 test results to the Centers for Medicare & Medicaid Services through 2024, although private labs will no longer be obligated to do so. The Centers for Disease Control and Prevention will also continue to monitor virus levels in communities through wastewater. But some states will no longer collect data.
Gone are the days when clinicians and others would watch for daily totals of case counts with the type of fervor typically reserved for live scoring updates during sports games, according to Dr. Costello.
“We just have to be mindful of the numbers that might be coming in,” Dr. Costello said.
Dr. Ransone, however, cautioned that clinicians not become complacent. In early May, Dr. Ransone saw two patients with conjunctivitis, what patients thought was simply pink eye – a symptom of the latest COVID-19 variant. Both patients told him it wasn’t possible they had COVID-19 because they didn’t have coughs.
“I don’t want to see physician offices fall into that trap that it’s over and be a potential nidus for infection for other patients,” Dr. Ransone said. “It’s incumbent upon us to remind people of the current symptoms so that folks will know when they need to wear a mask when they’re around their grandmother.”
The move away from universal masking in the office has benefits. Many of his older patients have difficulty hearing and had used lip reading to help understand him, he said. During the pandemic, masks got in the way of that form of communication. Now they can see his mouth again and better decipher what he says.
“Being able to have that face-to-face contact, without a mask intervening, has been really beneficial for a lot of my older patients,” he said.
A version of this article first appeared on Medscape.com.
The first day of seeing patients without a mask was, for Sterling Ransone Jr., MD, “unsettling.”
“I can’t tell you how weird it was the first day that I walked down the hall from my office to where my exam rooms are, to not have a mask on after 3 years of the habit,” said Dr. Ransone, a family physician in Deltaville, Va., and board chair of the American Academy of Family Physicians.
The White House recently lifted the public health emergency order that overhauled the way health care providers operated and advised patients over the past 3 years.
For Dr. Ransone, this transition entails getting used to his bare face, reminding patients of the latest and varying symptoms of the virus, and parting ways with sick patients if they refuse to wear a mask.
As states, hospitals, and health care systems around the country relax their mask mandates for care providers, clinicians will have to fall back on their own policies that patients with potential symptoms mask up.
“Now that it’s up to our offices, we have to have a little bit more backbone,” Dr. Ransone said. “If they’re not willing to follow a health-related policy that will protect the vulnerable, we will not see them. And so for us, it’s been pretty straightforward.”
Despite the policy, Dr. Ransone has cared for patients who don’t disclose they are feeling sick until he enters the room.
“And I wasn’t masked,” Dr. Ransone said. So, “I will wear masks for the rest of the day just to try to protect the rest of my patients in case I was exposed.”
Masks are optional for both patients and staff at the University of Maryland Medical System, but Niharika Khanna, MD, MBBS, said she still wears one with her patients, and her office advises staff to do the same. If patients are experiencing respiratory symptoms, like a cough, they are asked to wear one.
“When the patient first walks up to you, you have no idea what they have,” Dr. Khanna said.
Dr. Khanna is especially mindful of immunocompromised patients who have cancer, and Dr. Ransone cares for several patients who have received kidney transplants and are on potent immunosuppressive drugs.
“I know they’re appreciating our efforts to protect them, and I think the other patients are realizing that it’s a wise thing to do,” Dr. Ransone said.
Some patients have anxiety about the end of masking in doctor offices, but others have been excited about interacting more with their care teams, according to William Dahut, MD, chief scientific officer for the American Cancer Society. Many clinicians will advise their most immunocompromised patients the same as they did prior to the COVID-19 pandemic.
“There’s always been guidelines that oncologists have given to patients who are immunocompromised – we always told them to avoid crowded places, crowded scenes, be outside more than inside,” Dr. Dahut said. “Those general recommendations will continue.”
The AAFP supports masking to limit COVID’s spread, but the “most important thing people can do is to get vaccinated,” Tochi Iroku-Malize, MD, MPH, MBA, president of the AAFP, said.
But the accessibility of vaccinations is also shifting.
Testing shifts
The government will continue to provide free COVID-19 vaccines because it still has supplies on hand. When this stock runs out, commercial insurance providers will be required to cover the immunizations, as they are considered preventive, but people without insurance will have to pay out of pocket.
The AAFP is pushing the Biden administration and Congress to keep the purchase price of those vaccines low enough that clinicians can keep them in stock, according to Dr. Iroku-Malize. Once the federal government transitions COVID-19 vaccines to the commercial market – as early as later in 2023 – it may pose some challenges for providers.
“If the price of the vaccines is too high, physician practices may struggle to make the upfront investment in COVID-19 vaccines,” Dr. Iroku-Malize said. “Patients often prefer to receive vaccine counseling and administration from their usual source of primary care, like their family physician.”
The federal government has also said it still has a supply of treatments for the public to access for free, but has not revealed how much it has on hand or given a timeline for the transition to the private market.
COVID-19 tests, meanwhile, are no longer covered because of the end of the public health emergency, and cost about $45 per kit on average, according to an analysis by the Kaiser Family Foundation.
Pediatrician Lisa Costello, MD, MPH, knows that price point will be a challenge for some families she cares for at West Virginia University Medicine Children’s Hospital in Morgantown. Many still ask her where they can access free tests.
“Testing if you’re a higher risk person is something we need to ensure that people continue to be educated about,” Dr. Costello said.
She’s hopeful that COVID-19 vaccines and treatments such as Paxlovid will stay free in the coming months so patients can continue to easily access them.
Future of telehealth
Relaxed regulations of prescribing controlled substances via telehealth and across state lines allowed clinicians to treat patients near and far during the pandemic. But many providers were worried about a proposal from the Drug Enforcement Administration to clamp down on the prescribing of controlled substances via telehealth, according to A. Mark Fendrick, MD, an internal medicine physician at the University of Michigan, Ann Arbor.
“We were all panicking about what was going to happen to what is for many clinicians a very valuable policy,” Dr. Fendrick said of the telehealth flexibilities introduced during COVID-19.
The DEA, after getting 38,000 comments on their proposed regulations, pulled back on that plan, delaying the cliff until November.
Dr. Fendrick said that telehealth has allowed clinicians to reach patients who have historically faced barriers to care, such as lacking transportation.
“The benefits of that outweigh the potential harms,” he said. “Every policy you make that tightens access because you want to decrease the untoward and unfortunate outcomes will also decrease access to clinical indications.”
The AAFP said it hopes for clear guidance from the DEA in the coming months on what the new telehealth landscape for prescribing will look like.
Medicaid changes
About half of the patients who see Dr. Khanna have insurance through Medicaid.
During the public health emergency, states were not allowed to remove anyone from Medicaid, regardless of whether they no longer qualified for the program or not. But a law passed by Congress last year requires states to once again check Medicaid eligibility. As many as 15 million people could lose their Medicaid coverage.
That could affect the treatments Dr. Khanna recommends for her patients who get kicked off because those who become uninsured or transition to private insurance will have to pay more out of pocket. Maryland will start removals in June.
“When you have an uninsured patient versus Medicaid, it’s a huge difference in what you can ask the patient to do – the medications you can provide, the testing you can provide,” Dr. Khanna said.
States were authorized to remove people from Medicaid as of April 1, with Arkansas, New Hampshire, and South Dakota starting right away. But many states are just now getting the review process going. About a dozen states, including Indiana, Ohio, Utah, and West Virginia, started removing people in May 2023.
Uninsurance rates hit record lows across the United States during the pandemic. Keeping Americans on health insurance is a top priority for the AAFP, Dr. Iroku-Malize said. “We know health care coverage disruptions prevent people from seeking and accessing the care they need.”
Many people who are removed from Medicaid will be eligible for health insurance through employers, or through the Affordable Care Act’s private marketplace. But premiums and deductibles are often higher in these plans, which studies have shown result in patients delaying medical visits and not filling prescriptions or receiving treatment.
Staying mindful
Hospitals that receive federal funds will still have to report COVID-19 test results to the Centers for Medicare & Medicaid Services through 2024, although private labs will no longer be obligated to do so. The Centers for Disease Control and Prevention will also continue to monitor virus levels in communities through wastewater. But some states will no longer collect data.
Gone are the days when clinicians and others would watch for daily totals of case counts with the type of fervor typically reserved for live scoring updates during sports games, according to Dr. Costello.
“We just have to be mindful of the numbers that might be coming in,” Dr. Costello said.
Dr. Ransone, however, cautioned that clinicians not become complacent. In early May, Dr. Ransone saw two patients with conjunctivitis, what patients thought was simply pink eye – a symptom of the latest COVID-19 variant. Both patients told him it wasn’t possible they had COVID-19 because they didn’t have coughs.
“I don’t want to see physician offices fall into that trap that it’s over and be a potential nidus for infection for other patients,” Dr. Ransone said. “It’s incumbent upon us to remind people of the current symptoms so that folks will know when they need to wear a mask when they’re around their grandmother.”
The move away from universal masking in the office has benefits. Many of his older patients have difficulty hearing and had used lip reading to help understand him, he said. During the pandemic, masks got in the way of that form of communication. Now they can see his mouth again and better decipher what he says.
“Being able to have that face-to-face contact, without a mask intervening, has been really beneficial for a lot of my older patients,” he said.
A version of this article first appeared on Medscape.com.
The first day of seeing patients without a mask was, for Sterling Ransone Jr., MD, “unsettling.”
“I can’t tell you how weird it was the first day that I walked down the hall from my office to where my exam rooms are, to not have a mask on after 3 years of the habit,” said Dr. Ransone, a family physician in Deltaville, Va., and board chair of the American Academy of Family Physicians.
The White House recently lifted the public health emergency order that overhauled the way health care providers operated and advised patients over the past 3 years.
For Dr. Ransone, this transition entails getting used to his bare face, reminding patients of the latest and varying symptoms of the virus, and parting ways with sick patients if they refuse to wear a mask.
As states, hospitals, and health care systems around the country relax their mask mandates for care providers, clinicians will have to fall back on their own policies that patients with potential symptoms mask up.
“Now that it’s up to our offices, we have to have a little bit more backbone,” Dr. Ransone said. “If they’re not willing to follow a health-related policy that will protect the vulnerable, we will not see them. And so for us, it’s been pretty straightforward.”
Despite the policy, Dr. Ransone has cared for patients who don’t disclose they are feeling sick until he enters the room.
“And I wasn’t masked,” Dr. Ransone said. So, “I will wear masks for the rest of the day just to try to protect the rest of my patients in case I was exposed.”
Masks are optional for both patients and staff at the University of Maryland Medical System, but Niharika Khanna, MD, MBBS, said she still wears one with her patients, and her office advises staff to do the same. If patients are experiencing respiratory symptoms, like a cough, they are asked to wear one.
“When the patient first walks up to you, you have no idea what they have,” Dr. Khanna said.
Dr. Khanna is especially mindful of immunocompromised patients who have cancer, and Dr. Ransone cares for several patients who have received kidney transplants and are on potent immunosuppressive drugs.
“I know they’re appreciating our efforts to protect them, and I think the other patients are realizing that it’s a wise thing to do,” Dr. Ransone said.
Some patients have anxiety about the end of masking in doctor offices, but others have been excited about interacting more with their care teams, according to William Dahut, MD, chief scientific officer for the American Cancer Society. Many clinicians will advise their most immunocompromised patients the same as they did prior to the COVID-19 pandemic.
“There’s always been guidelines that oncologists have given to patients who are immunocompromised – we always told them to avoid crowded places, crowded scenes, be outside more than inside,” Dr. Dahut said. “Those general recommendations will continue.”
The AAFP supports masking to limit COVID’s spread, but the “most important thing people can do is to get vaccinated,” Tochi Iroku-Malize, MD, MPH, MBA, president of the AAFP, said.
But the accessibility of vaccinations is also shifting.
Testing shifts
The government will continue to provide free COVID-19 vaccines because it still has supplies on hand. When this stock runs out, commercial insurance providers will be required to cover the immunizations, as they are considered preventive, but people without insurance will have to pay out of pocket.
The AAFP is pushing the Biden administration and Congress to keep the purchase price of those vaccines low enough that clinicians can keep them in stock, according to Dr. Iroku-Malize. Once the federal government transitions COVID-19 vaccines to the commercial market – as early as later in 2023 – it may pose some challenges for providers.
“If the price of the vaccines is too high, physician practices may struggle to make the upfront investment in COVID-19 vaccines,” Dr. Iroku-Malize said. “Patients often prefer to receive vaccine counseling and administration from their usual source of primary care, like their family physician.”
The federal government has also said it still has a supply of treatments for the public to access for free, but has not revealed how much it has on hand or given a timeline for the transition to the private market.
COVID-19 tests, meanwhile, are no longer covered because of the end of the public health emergency, and cost about $45 per kit on average, according to an analysis by the Kaiser Family Foundation.
Pediatrician Lisa Costello, MD, MPH, knows that price point will be a challenge for some families she cares for at West Virginia University Medicine Children’s Hospital in Morgantown. Many still ask her where they can access free tests.
“Testing if you’re a higher risk person is something we need to ensure that people continue to be educated about,” Dr. Costello said.
She’s hopeful that COVID-19 vaccines and treatments such as Paxlovid will stay free in the coming months so patients can continue to easily access them.
Future of telehealth
Relaxed regulations of prescribing controlled substances via telehealth and across state lines allowed clinicians to treat patients near and far during the pandemic. But many providers were worried about a proposal from the Drug Enforcement Administration to clamp down on the prescribing of controlled substances via telehealth, according to A. Mark Fendrick, MD, an internal medicine physician at the University of Michigan, Ann Arbor.
“We were all panicking about what was going to happen to what is for many clinicians a very valuable policy,” Dr. Fendrick said of the telehealth flexibilities introduced during COVID-19.
The DEA, after getting 38,000 comments on their proposed regulations, pulled back on that plan, delaying the cliff until November.
Dr. Fendrick said that telehealth has allowed clinicians to reach patients who have historically faced barriers to care, such as lacking transportation.
“The benefits of that outweigh the potential harms,” he said. “Every policy you make that tightens access because you want to decrease the untoward and unfortunate outcomes will also decrease access to clinical indications.”
The AAFP said it hopes for clear guidance from the DEA in the coming months on what the new telehealth landscape for prescribing will look like.
Medicaid changes
About half of the patients who see Dr. Khanna have insurance through Medicaid.
During the public health emergency, states were not allowed to remove anyone from Medicaid, regardless of whether they no longer qualified for the program or not. But a law passed by Congress last year requires states to once again check Medicaid eligibility. As many as 15 million people could lose their Medicaid coverage.
That could affect the treatments Dr. Khanna recommends for her patients who get kicked off because those who become uninsured or transition to private insurance will have to pay more out of pocket. Maryland will start removals in June.
“When you have an uninsured patient versus Medicaid, it’s a huge difference in what you can ask the patient to do – the medications you can provide, the testing you can provide,” Dr. Khanna said.
States were authorized to remove people from Medicaid as of April 1, with Arkansas, New Hampshire, and South Dakota starting right away. But many states are just now getting the review process going. About a dozen states, including Indiana, Ohio, Utah, and West Virginia, started removing people in May 2023.
Uninsurance rates hit record lows across the United States during the pandemic. Keeping Americans on health insurance is a top priority for the AAFP, Dr. Iroku-Malize said. “We know health care coverage disruptions prevent people from seeking and accessing the care they need.”
Many people who are removed from Medicaid will be eligible for health insurance through employers, or through the Affordable Care Act’s private marketplace. But premiums and deductibles are often higher in these plans, which studies have shown result in patients delaying medical visits and not filling prescriptions or receiving treatment.
Staying mindful
Hospitals that receive federal funds will still have to report COVID-19 test results to the Centers for Medicare & Medicaid Services through 2024, although private labs will no longer be obligated to do so. The Centers for Disease Control and Prevention will also continue to monitor virus levels in communities through wastewater. But some states will no longer collect data.
Gone are the days when clinicians and others would watch for daily totals of case counts with the type of fervor typically reserved for live scoring updates during sports games, according to Dr. Costello.
“We just have to be mindful of the numbers that might be coming in,” Dr. Costello said.
Dr. Ransone, however, cautioned that clinicians not become complacent. In early May, Dr. Ransone saw two patients with conjunctivitis, what patients thought was simply pink eye – a symptom of the latest COVID-19 variant. Both patients told him it wasn’t possible they had COVID-19 because they didn’t have coughs.
“I don’t want to see physician offices fall into that trap that it’s over and be a potential nidus for infection for other patients,” Dr. Ransone said. “It’s incumbent upon us to remind people of the current symptoms so that folks will know when they need to wear a mask when they’re around their grandmother.”
The move away from universal masking in the office has benefits. Many of his older patients have difficulty hearing and had used lip reading to help understand him, he said. During the pandemic, masks got in the way of that form of communication. Now they can see his mouth again and better decipher what he says.
“Being able to have that face-to-face contact, without a mask intervening, has been really beneficial for a lot of my older patients,” he said.
A version of this article first appeared on Medscape.com.
The antimicrobial peptide that even Pharma can love
Fastest peptide north, south, east, aaaaand west of the Pecos
Bacterial infections are supposed to be simple. You get infected, you get an antibiotic to treat it. Easy. Some bacteria, though, don’t play by the rules. Those antibiotics may kill 99.9% of germs, but what about the 0.1% that gets left behind? With their fallen comrades out of the way, the accidentally drug resistant species are free to inherit the Earth.
Antibiotic resistance is thus a major concern for the medical community. Naturally, anything that prevents doctors from successfully curing sick people is a priority. Unless you’re a major pharmaceutical company that has been loath to develop new drugs that can beat antibiotic-resistant bacteria. Blah blah, time and money, blah blah, long time between development and market application, blah blah, no profit. We all know the story with pharmaceutical companies.
Research from other sources has continued, however, and Brazilian scientists recently published research involving a peptide known as plantaricin 149. This peptide, derived from the bacterium Lactobacillus plantarum, has been known for nearly 30 years to have antibacterial properties. Pln149 in its natural state, though, is not particularly efficient at bacteria-killing. Fortunately, we have science and technology on our side.
The researchers synthesized 20 analogs of Pln149, of which Pln149-PEP20 had the best results. The elegantly named compound is less than half the size of the original peptide, less toxic, and far better at killing any and all drug-resistant bacteria the researchers threw at it. How much better? Pln149-PEP20 started killing bacteria less than an hour after being introduced in lab trials.
The research is just in its early days – just because something is less toxic doesn’t necessarily mean you want to go and help yourself to it – but we can only hope that those lovely pharmaceutical companies deign to look down upon us and actually develop a drug utilizing Pln149-PEP20 to, you know, actually help sick people, instead of trying to build monopolies or avoiding paying billions in taxes. Yeah, we couldn’t keep a straight face through that last sentence either.
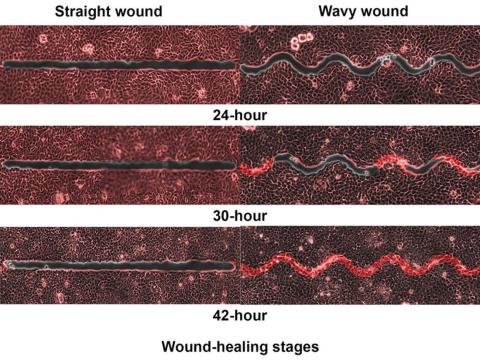
Speed healing: The wavy wound gets the swirl
Did you know that wavy wounds heal faster than straight wounds? Well, we didn’t, but apparently quite a few people did, because somebody has been trying to figure out why wavy wounds heal faster than straight ones. Do the surgeons know about this? How about you dermatologists? Wavy over straight? We’re the media. We’re supposed to report this kind of stuff. Maybe hit us with a tweet next time you do something important, or push a TikTok our way, okay?
You could be more like the investigators at Nanyang Technological University in Singapore, who figured out the why and then released a statement about it.
They created synthetic wounds – some straight, some wavy – in micropatterned hydrogel substrates that mimicked human skin. Then they used an advanced optical technique known as particle image velocimetry to measure fluid flow and learn how cells moved to close the wound gaps.
The wavy wounds “induced more complex collective cell movements, such as a swirly, vortex-like motion,” according to the written statement from NTU Singapore. In the straight wounds, cell movements paralleled the wound front, “moving in straight lines like a marching band,” they pointed out, unlike some researchers who never call us unless they need money.
Complex epithelial cell movements are better, it turns out. Over an observation period of 64 hours the NTU team found that the healing efficiency of wavy gaps – measured by the area covered by the cells over time – is nearly five times faster than straight gaps.
The complex motion “enabled cells to quickly connect with similar cells on the opposite site of the wound edge, forming a bridge and closing the wavy wound gaps faster than straight gaps,” explained lead author Xu Hongmei, a doctoral student at NTU’s School of Mechanical and Aerospace Engineering, who seems to have time to toss out a tumblr or two to keep the press informed.
As for the rest of you, would it kill you to pick up a phone once in a while? Maybe let a journalist know that you’re still alive? We have feelings too, you know, and we worry.
A little Jekyll, a little Hyde, and a little shop of horrors
More “Little Shop of Horrors” references are coming, so be prepared.
We begin with Triphyophyllum peltatum. This woody vine is of great interest to medical and pharmaceutical researchers because its constituents have shown promise against pancreatic cancer and leukemia cells, among others, along with the pathogens that cause malaria and other diseases. There is another side, however. T. peltatum also has a tendency to turn into a realistic Audrey II when deprived.
No, of course they’re not craving human flesh, but it does become … carnivorous in its appetite.
T. peltatum, native to the West African tropics and not found in a New York florist shop, has the unique ability to change its diet and development based on the environmental circumstances. For some unknown reason, the leaves would develop adhesive traps in the form of sticky drops that capture insect prey. The plant is notoriously hard to grow, however, so no one could study the transformation under lab conditions. Until now.
A group of German scientists “exposed the plant to different stress factors, including deficiencies of various nutrients, and studied how it responded to each,” said Dr. Traud Winkelmann of Leibniz University Hannover. “Only in one case were we able to observe the formation of traps: in the case of a lack of phosphorus.”
Well, there you have it: phosphorus. We need it for healthy bones and teeth, which this plant doesn’t have to worry about, unlike its Tony Award–nominated counterpart. The investigators hope that their findings could lead to “future molecular analyses that will help understand the origins of carnivory,” but we’re guessing that a certain singing alien species will be left out of that research.
Fastest peptide north, south, east, aaaaand west of the Pecos
Bacterial infections are supposed to be simple. You get infected, you get an antibiotic to treat it. Easy. Some bacteria, though, don’t play by the rules. Those antibiotics may kill 99.9% of germs, but what about the 0.1% that gets left behind? With their fallen comrades out of the way, the accidentally drug resistant species are free to inherit the Earth.
Antibiotic resistance is thus a major concern for the medical community. Naturally, anything that prevents doctors from successfully curing sick people is a priority. Unless you’re a major pharmaceutical company that has been loath to develop new drugs that can beat antibiotic-resistant bacteria. Blah blah, time and money, blah blah, long time between development and market application, blah blah, no profit. We all know the story with pharmaceutical companies.
Research from other sources has continued, however, and Brazilian scientists recently published research involving a peptide known as plantaricin 149. This peptide, derived from the bacterium Lactobacillus plantarum, has been known for nearly 30 years to have antibacterial properties. Pln149 in its natural state, though, is not particularly efficient at bacteria-killing. Fortunately, we have science and technology on our side.
The researchers synthesized 20 analogs of Pln149, of which Pln149-PEP20 had the best results. The elegantly named compound is less than half the size of the original peptide, less toxic, and far better at killing any and all drug-resistant bacteria the researchers threw at it. How much better? Pln149-PEP20 started killing bacteria less than an hour after being introduced in lab trials.
The research is just in its early days – just because something is less toxic doesn’t necessarily mean you want to go and help yourself to it – but we can only hope that those lovely pharmaceutical companies deign to look down upon us and actually develop a drug utilizing Pln149-PEP20 to, you know, actually help sick people, instead of trying to build monopolies or avoiding paying billions in taxes. Yeah, we couldn’t keep a straight face through that last sentence either.
Speed healing: The wavy wound gets the swirl
Did you know that wavy wounds heal faster than straight wounds? Well, we didn’t, but apparently quite a few people did, because somebody has been trying to figure out why wavy wounds heal faster than straight ones. Do the surgeons know about this? How about you dermatologists? Wavy over straight? We’re the media. We’re supposed to report this kind of stuff. Maybe hit us with a tweet next time you do something important, or push a TikTok our way, okay?
You could be more like the investigators at Nanyang Technological University in Singapore, who figured out the why and then released a statement about it.
They created synthetic wounds – some straight, some wavy – in micropatterned hydrogel substrates that mimicked human skin. Then they used an advanced optical technique known as particle image velocimetry to measure fluid flow and learn how cells moved to close the wound gaps.
The wavy wounds “induced more complex collective cell movements, such as a swirly, vortex-like motion,” according to the written statement from NTU Singapore. In the straight wounds, cell movements paralleled the wound front, “moving in straight lines like a marching band,” they pointed out, unlike some researchers who never call us unless they need money.
Complex epithelial cell movements are better, it turns out. Over an observation period of 64 hours the NTU team found that the healing efficiency of wavy gaps – measured by the area covered by the cells over time – is nearly five times faster than straight gaps.
The complex motion “enabled cells to quickly connect with similar cells on the opposite site of the wound edge, forming a bridge and closing the wavy wound gaps faster than straight gaps,” explained lead author Xu Hongmei, a doctoral student at NTU’s School of Mechanical and Aerospace Engineering, who seems to have time to toss out a tumblr or two to keep the press informed.
As for the rest of you, would it kill you to pick up a phone once in a while? Maybe let a journalist know that you’re still alive? We have feelings too, you know, and we worry.
A little Jekyll, a little Hyde, and a little shop of horrors
More “Little Shop of Horrors” references are coming, so be prepared.
We begin with Triphyophyllum peltatum. This woody vine is of great interest to medical and pharmaceutical researchers because its constituents have shown promise against pancreatic cancer and leukemia cells, among others, along with the pathogens that cause malaria and other diseases. There is another side, however. T. peltatum also has a tendency to turn into a realistic Audrey II when deprived.
No, of course they’re not craving human flesh, but it does become … carnivorous in its appetite.
T. peltatum, native to the West African tropics and not found in a New York florist shop, has the unique ability to change its diet and development based on the environmental circumstances. For some unknown reason, the leaves would develop adhesive traps in the form of sticky drops that capture insect prey. The plant is notoriously hard to grow, however, so no one could study the transformation under lab conditions. Until now.
A group of German scientists “exposed the plant to different stress factors, including deficiencies of various nutrients, and studied how it responded to each,” said Dr. Traud Winkelmann of Leibniz University Hannover. “Only in one case were we able to observe the formation of traps: in the case of a lack of phosphorus.”
Well, there you have it: phosphorus. We need it for healthy bones and teeth, which this plant doesn’t have to worry about, unlike its Tony Award–nominated counterpart. The investigators hope that their findings could lead to “future molecular analyses that will help understand the origins of carnivory,” but we’re guessing that a certain singing alien species will be left out of that research.
Fastest peptide north, south, east, aaaaand west of the Pecos
Bacterial infections are supposed to be simple. You get infected, you get an antibiotic to treat it. Easy. Some bacteria, though, don’t play by the rules. Those antibiotics may kill 99.9% of germs, but what about the 0.1% that gets left behind? With their fallen comrades out of the way, the accidentally drug resistant species are free to inherit the Earth.
Antibiotic resistance is thus a major concern for the medical community. Naturally, anything that prevents doctors from successfully curing sick people is a priority. Unless you’re a major pharmaceutical company that has been loath to develop new drugs that can beat antibiotic-resistant bacteria. Blah blah, time and money, blah blah, long time between development and market application, blah blah, no profit. We all know the story with pharmaceutical companies.
Research from other sources has continued, however, and Brazilian scientists recently published research involving a peptide known as plantaricin 149. This peptide, derived from the bacterium Lactobacillus plantarum, has been known for nearly 30 years to have antibacterial properties. Pln149 in its natural state, though, is not particularly efficient at bacteria-killing. Fortunately, we have science and technology on our side.
The researchers synthesized 20 analogs of Pln149, of which Pln149-PEP20 had the best results. The elegantly named compound is less than half the size of the original peptide, less toxic, and far better at killing any and all drug-resistant bacteria the researchers threw at it. How much better? Pln149-PEP20 started killing bacteria less than an hour after being introduced in lab trials.
The research is just in its early days – just because something is less toxic doesn’t necessarily mean you want to go and help yourself to it – but we can only hope that those lovely pharmaceutical companies deign to look down upon us and actually develop a drug utilizing Pln149-PEP20 to, you know, actually help sick people, instead of trying to build monopolies or avoiding paying billions in taxes. Yeah, we couldn’t keep a straight face through that last sentence either.
Speed healing: The wavy wound gets the swirl
Did you know that wavy wounds heal faster than straight wounds? Well, we didn’t, but apparently quite a few people did, because somebody has been trying to figure out why wavy wounds heal faster than straight ones. Do the surgeons know about this? How about you dermatologists? Wavy over straight? We’re the media. We’re supposed to report this kind of stuff. Maybe hit us with a tweet next time you do something important, or push a TikTok our way, okay?
You could be more like the investigators at Nanyang Technological University in Singapore, who figured out the why and then released a statement about it.
They created synthetic wounds – some straight, some wavy – in micropatterned hydrogel substrates that mimicked human skin. Then they used an advanced optical technique known as particle image velocimetry to measure fluid flow and learn how cells moved to close the wound gaps.
The wavy wounds “induced more complex collective cell movements, such as a swirly, vortex-like motion,” according to the written statement from NTU Singapore. In the straight wounds, cell movements paralleled the wound front, “moving in straight lines like a marching band,” they pointed out, unlike some researchers who never call us unless they need money.
Complex epithelial cell movements are better, it turns out. Over an observation period of 64 hours the NTU team found that the healing efficiency of wavy gaps – measured by the area covered by the cells over time – is nearly five times faster than straight gaps.
The complex motion “enabled cells to quickly connect with similar cells on the opposite site of the wound edge, forming a bridge and closing the wavy wound gaps faster than straight gaps,” explained lead author Xu Hongmei, a doctoral student at NTU’s School of Mechanical and Aerospace Engineering, who seems to have time to toss out a tumblr or two to keep the press informed.
As for the rest of you, would it kill you to pick up a phone once in a while? Maybe let a journalist know that you’re still alive? We have feelings too, you know, and we worry.
A little Jekyll, a little Hyde, and a little shop of horrors
More “Little Shop of Horrors” references are coming, so be prepared.
We begin with Triphyophyllum peltatum. This woody vine is of great interest to medical and pharmaceutical researchers because its constituents have shown promise against pancreatic cancer and leukemia cells, among others, along with the pathogens that cause malaria and other diseases. There is another side, however. T. peltatum also has a tendency to turn into a realistic Audrey II when deprived.
No, of course they’re not craving human flesh, but it does become … carnivorous in its appetite.
T. peltatum, native to the West African tropics and not found in a New York florist shop, has the unique ability to change its diet and development based on the environmental circumstances. For some unknown reason, the leaves would develop adhesive traps in the form of sticky drops that capture insect prey. The plant is notoriously hard to grow, however, so no one could study the transformation under lab conditions. Until now.
A group of German scientists “exposed the plant to different stress factors, including deficiencies of various nutrients, and studied how it responded to each,” said Dr. Traud Winkelmann of Leibniz University Hannover. “Only in one case were we able to observe the formation of traps: in the case of a lack of phosphorus.”
Well, there you have it: phosphorus. We need it for healthy bones and teeth, which this plant doesn’t have to worry about, unlike its Tony Award–nominated counterpart. The investigators hope that their findings could lead to “future molecular analyses that will help understand the origins of carnivory,” but we’re guessing that a certain singing alien species will be left out of that research.
Docs fervently hope federal ban on noncompete clauses goes through
The Federal Trade Commission’s proposed regulation that would ban noncompete agreements across the country seems like potential good news for doctors. Of course, many hospitals and employers are against it. As a result, the FTC’s sweeping proposal has tongues wagging on both sides of the issue.
Many physicians are thrilled that they may soon have more control over their career and not be stuck in jobs where they feel frustrated, underpaid, or blocked in their progress.
As of 2018, as many as 45% of primary care physicians had inked such agreements with their employers.
Typically, the agreements prevent physicians from practicing medicine with a new employer for a defined period within a specific geographic area. No matter how attractive an alternate offer of employment might be, doctors are bound by the agreements to say no if the offer exists in that defined area and time period.
The period for public comment on the proposed regulation ended on April 19, and there is currently no set date for a decision.
In a Medscape poll of 558 physicians, more than 9 out of 10 respondents said that they were either currently bound by a noncompete clause or that they had been bound by one in the past that had forced them to temporarily stop working, commute long distances, move to a different area, or switch fields.
The new proposal would make it illegal for an employer, such as a hospital or large group, to enter a noncompete with a worker; maintain a noncompete with a worker; or represent to a worker, under certain circumstances, that the worker is subject to a noncompete.
It also would not only ban future noncompete agreements but also retroactively invalidate existing ones. The FTC reasons that noncompete clauses could potentially increase worker earnings as well as lower health care costs by billions of dollars. If the ruling were to move forward, it would represent part of President Biden’s “worker-forward” priorities, focusing on how competition can be a good thing for employees. The President billed the FTC’s announcement as a “huge win for workers.”
In its statements on the proposed ban, the FTC claimed that it could lower consumer prices across the board by as much as $150 billion per year and return nearly $300 million to workers each year.
However, even if passed, the draft rule would keep in place nonsolicitation rules that many health care organizations have put into place. That means that, if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to switch to him or her in the new job.
Within that clause, however, the FTC has specified that if such nonsolicitation agreement has the “equivalent effect” of a noncompete, the agency would deem it such. That means, even if that rule stays, it could be contested and may be interpreted as violating the noncompete law. So there’s value in reading all the fine print should the ban move forward.
Could the ban bring potential downsides?
Most physicians view the potential to break free of a noncompete agreement as a victory. Peter Glennon, an employment litigation attorney with The Glennon Law Firm in Rochester, N.Y., says not so fast. “If you ask anyone if they’d prefer a noncompete agreement, of course they’re going to say no,” he said in an interview. “It sounds like a restriction, one that can hold you back.”
Mr. Glennon believes that there are actually upsides to physician noncompetes. For instance, many noncompetes come with sign-on bonuses that could potentially disappear without the agreements. There’s also the fact that when some physicians sign a noncompete agreement, they then receive pro bono training and continuing education along with marketing and promotion of their skills. Without signing a noncompete, employers may be less incentivized to provide all those benefits to their physician employers.
Those benefits – and the noncompetes – also vary by specialty, Mr. Glennon said. “In 2021, Washington, DC, banned noncompetes for doctors making less than $250,000. So, most generalists there can walk across the street and get a new job. For specialists like cardiologists or neurosurgeons, however, advanced training and marketing benefits matter, so many of them don’t want to lose noncompetes.”
Still, most physicians hope that the FTC’s ban takes hold. Manan Shah, MD, founder, and chief medical officer at Wyndly, an allergy relief startup practice, is one of them.
“Initially, it might disincentivize hospital systems from helping new physicians build up their name and practice because they might be concerned about a physician leaving and starting anew,” he said. “But in the long term, hospitals require physicians to bring their patients to them for care, so the best hospitals will always compete for the best physicians and support them as they build up their practice.”
Dr. Shah views noncompetes as overly prohibitive to physicians. “Right now, if a physician starts a job at a large hospital system and realizes they want to switch jobs, the noncompete distances are so wide they often have to move cities to continue practicing,” he said. “Picking up and starting over in a new city isn’t an option for everyone and can be especially difficult for someone with a family.”
Where Mr. Glennon argued that a physician leaving a team-based practice might harm patients, Shah takes a different perspective. “Imagine you have a doctor whom you trust and have been working with,” he said. “If something changes at their hospital and they decide to move, you literally have to find a new doctor instead of just being able to see them at another location down the street.”
Another potential burden of the noncompete agreements is that they could possibly squelch doctor’s desires to hang up their own shingle. According to Dr. Shah, the agreements make it so that if a physician wants to work independently, it’s nearly impossible to fly solo. “This is frustrating because independent practices have been shown to be more cost effective and allow patients to build better relationships with their doctors,” he claimed.
A 2016 study from Annals of Family Medicine supports that claim, at least for small general practices. Another study appearing in JAMA concurred. It does point out, however, that the cost equation is nuanced and that benefits of larger systems include more resilience to economic downturns and can provide more specialized care.
Will nonprofit hospitals be subject to this noncompete ban?
Further complicating the noncompete ban issue is how it might impact nonprofit institutions versus their for-profit peers. Most hospitals structured as nonprofits would be exempt from the rule because the FTC Act provides that it can enforce against “persons, partnerships, or corporations,” which are further defined as entities “organized to carry on business for their own profit or that of their members.”
The fallout from this, said Dr. Shah, is that it “would disproportionately affect health care providers, since many hospital systems are nonprofits. This is disconcerting because we know that many nonprofit systems make large profits anyway and can offer executive teams’ lucrative packages, while the nurses, assistants, and physicians providing the care are generally not well compensated.”
So far, about nine states plus Washington, D.C., have already put noncompete bans in place, and they may serve as a harbinger of things to come should the federal ban go into effect. Each varies in its specifics. Some, like Indiana, outright ban them, whereas others limit them based on variables like income and industry. “We’re seeing these states responding to local market conditions,” said Darryl Drevna, senior director of regulatory affairs at the American Medical Group Association. “Health care is a hyperlocal market. Depending on the situation, the bans adapt and respond specific to those states.”
Should the federal ban take hold, however, it will supersede whatever rules the individual states have in place.
Some opponents of the federal ban proposal question its authority to begin with, however, Mr. Glennon included. “Many people believe the FTC is overstepping,” he said. “Some people believe that Section 5 of the FTC Act does not give it the authority to police labor markets.”
Mr. Drevna noted that the FTC has taken an aggressive stance, one that will ultimately wind up in the courts. “How it works out is anyone’s guess,” he said. “Ideally, the FTC will consider the comments and concerns of groups like AMGA and realize that states are best suited to regulate in this area.”
In general, the ban’s supporters are employees/physicians; those who oppose it are their employers. Joining the AMGA in speaking out against the noncompete ban is the American Hospital Association, whereas the American College of Emergency Physicians has come out largely in support of the ban.
Still, doctors like Dr. Shah remain hopeful. “I am optimistic that perhaps my colleagues will not continue to be stuck in overrestrictive noncompetes, but I am also realistic,” he said. “Hospital systems are already coming out strongly against this and they have deep pockets, so I won’t be surprised if it does not come to pass.”
A version of this article first appeared on Medscape.com.
The Federal Trade Commission’s proposed regulation that would ban noncompete agreements across the country seems like potential good news for doctors. Of course, many hospitals and employers are against it. As a result, the FTC’s sweeping proposal has tongues wagging on both sides of the issue.
Many physicians are thrilled that they may soon have more control over their career and not be stuck in jobs where they feel frustrated, underpaid, or blocked in their progress.
As of 2018, as many as 45% of primary care physicians had inked such agreements with their employers.
Typically, the agreements prevent physicians from practicing medicine with a new employer for a defined period within a specific geographic area. No matter how attractive an alternate offer of employment might be, doctors are bound by the agreements to say no if the offer exists in that defined area and time period.
The period for public comment on the proposed regulation ended on April 19, and there is currently no set date for a decision.
In a Medscape poll of 558 physicians, more than 9 out of 10 respondents said that they were either currently bound by a noncompete clause or that they had been bound by one in the past that had forced them to temporarily stop working, commute long distances, move to a different area, or switch fields.
The new proposal would make it illegal for an employer, such as a hospital or large group, to enter a noncompete with a worker; maintain a noncompete with a worker; or represent to a worker, under certain circumstances, that the worker is subject to a noncompete.
It also would not only ban future noncompete agreements but also retroactively invalidate existing ones. The FTC reasons that noncompete clauses could potentially increase worker earnings as well as lower health care costs by billions of dollars. If the ruling were to move forward, it would represent part of President Biden’s “worker-forward” priorities, focusing on how competition can be a good thing for employees. The President billed the FTC’s announcement as a “huge win for workers.”
In its statements on the proposed ban, the FTC claimed that it could lower consumer prices across the board by as much as $150 billion per year and return nearly $300 million to workers each year.
However, even if passed, the draft rule would keep in place nonsolicitation rules that many health care organizations have put into place. That means that, if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to switch to him or her in the new job.
Within that clause, however, the FTC has specified that if such nonsolicitation agreement has the “equivalent effect” of a noncompete, the agency would deem it such. That means, even if that rule stays, it could be contested and may be interpreted as violating the noncompete law. So there’s value in reading all the fine print should the ban move forward.
Could the ban bring potential downsides?
Most physicians view the potential to break free of a noncompete agreement as a victory. Peter Glennon, an employment litigation attorney with The Glennon Law Firm in Rochester, N.Y., says not so fast. “If you ask anyone if they’d prefer a noncompete agreement, of course they’re going to say no,” he said in an interview. “It sounds like a restriction, one that can hold you back.”
Mr. Glennon believes that there are actually upsides to physician noncompetes. For instance, many noncompetes come with sign-on bonuses that could potentially disappear without the agreements. There’s also the fact that when some physicians sign a noncompete agreement, they then receive pro bono training and continuing education along with marketing and promotion of their skills. Without signing a noncompete, employers may be less incentivized to provide all those benefits to their physician employers.
Those benefits – and the noncompetes – also vary by specialty, Mr. Glennon said. “In 2021, Washington, DC, banned noncompetes for doctors making less than $250,000. So, most generalists there can walk across the street and get a new job. For specialists like cardiologists or neurosurgeons, however, advanced training and marketing benefits matter, so many of them don’t want to lose noncompetes.”
Still, most physicians hope that the FTC’s ban takes hold. Manan Shah, MD, founder, and chief medical officer at Wyndly, an allergy relief startup practice, is one of them.
“Initially, it might disincentivize hospital systems from helping new physicians build up their name and practice because they might be concerned about a physician leaving and starting anew,” he said. “But in the long term, hospitals require physicians to bring their patients to them for care, so the best hospitals will always compete for the best physicians and support them as they build up their practice.”
Dr. Shah views noncompetes as overly prohibitive to physicians. “Right now, if a physician starts a job at a large hospital system and realizes they want to switch jobs, the noncompete distances are so wide they often have to move cities to continue practicing,” he said. “Picking up and starting over in a new city isn’t an option for everyone and can be especially difficult for someone with a family.”
Where Mr. Glennon argued that a physician leaving a team-based practice might harm patients, Shah takes a different perspective. “Imagine you have a doctor whom you trust and have been working with,” he said. “If something changes at their hospital and they decide to move, you literally have to find a new doctor instead of just being able to see them at another location down the street.”
Another potential burden of the noncompete agreements is that they could possibly squelch doctor’s desires to hang up their own shingle. According to Dr. Shah, the agreements make it so that if a physician wants to work independently, it’s nearly impossible to fly solo. “This is frustrating because independent practices have been shown to be more cost effective and allow patients to build better relationships with their doctors,” he claimed.
A 2016 study from Annals of Family Medicine supports that claim, at least for small general practices. Another study appearing in JAMA concurred. It does point out, however, that the cost equation is nuanced and that benefits of larger systems include more resilience to economic downturns and can provide more specialized care.
Will nonprofit hospitals be subject to this noncompete ban?
Further complicating the noncompete ban issue is how it might impact nonprofit institutions versus their for-profit peers. Most hospitals structured as nonprofits would be exempt from the rule because the FTC Act provides that it can enforce against “persons, partnerships, or corporations,” which are further defined as entities “organized to carry on business for their own profit or that of their members.”
The fallout from this, said Dr. Shah, is that it “would disproportionately affect health care providers, since many hospital systems are nonprofits. This is disconcerting because we know that many nonprofit systems make large profits anyway and can offer executive teams’ lucrative packages, while the nurses, assistants, and physicians providing the care are generally not well compensated.”
So far, about nine states plus Washington, D.C., have already put noncompete bans in place, and they may serve as a harbinger of things to come should the federal ban go into effect. Each varies in its specifics. Some, like Indiana, outright ban them, whereas others limit them based on variables like income and industry. “We’re seeing these states responding to local market conditions,” said Darryl Drevna, senior director of regulatory affairs at the American Medical Group Association. “Health care is a hyperlocal market. Depending on the situation, the bans adapt and respond specific to those states.”
Should the federal ban take hold, however, it will supersede whatever rules the individual states have in place.
Some opponents of the federal ban proposal question its authority to begin with, however, Mr. Glennon included. “Many people believe the FTC is overstepping,” he said. “Some people believe that Section 5 of the FTC Act does not give it the authority to police labor markets.”
Mr. Drevna noted that the FTC has taken an aggressive stance, one that will ultimately wind up in the courts. “How it works out is anyone’s guess,” he said. “Ideally, the FTC will consider the comments and concerns of groups like AMGA and realize that states are best suited to regulate in this area.”
In general, the ban’s supporters are employees/physicians; those who oppose it are their employers. Joining the AMGA in speaking out against the noncompete ban is the American Hospital Association, whereas the American College of Emergency Physicians has come out largely in support of the ban.
Still, doctors like Dr. Shah remain hopeful. “I am optimistic that perhaps my colleagues will not continue to be stuck in overrestrictive noncompetes, but I am also realistic,” he said. “Hospital systems are already coming out strongly against this and they have deep pockets, so I won’t be surprised if it does not come to pass.”
A version of this article first appeared on Medscape.com.
The Federal Trade Commission’s proposed regulation that would ban noncompete agreements across the country seems like potential good news for doctors. Of course, many hospitals and employers are against it. As a result, the FTC’s sweeping proposal has tongues wagging on both sides of the issue.
Many physicians are thrilled that they may soon have more control over their career and not be stuck in jobs where they feel frustrated, underpaid, or blocked in their progress.
As of 2018, as many as 45% of primary care physicians had inked such agreements with their employers.
Typically, the agreements prevent physicians from practicing medicine with a new employer for a defined period within a specific geographic area. No matter how attractive an alternate offer of employment might be, doctors are bound by the agreements to say no if the offer exists in that defined area and time period.
The period for public comment on the proposed regulation ended on April 19, and there is currently no set date for a decision.
In a Medscape poll of 558 physicians, more than 9 out of 10 respondents said that they were either currently bound by a noncompete clause or that they had been bound by one in the past that had forced them to temporarily stop working, commute long distances, move to a different area, or switch fields.
The new proposal would make it illegal for an employer, such as a hospital or large group, to enter a noncompete with a worker; maintain a noncompete with a worker; or represent to a worker, under certain circumstances, that the worker is subject to a noncompete.
It also would not only ban future noncompete agreements but also retroactively invalidate existing ones. The FTC reasons that noncompete clauses could potentially increase worker earnings as well as lower health care costs by billions of dollars. If the ruling were to move forward, it would represent part of President Biden’s “worker-forward” priorities, focusing on how competition can be a good thing for employees. The President billed the FTC’s announcement as a “huge win for workers.”
In its statements on the proposed ban, the FTC claimed that it could lower consumer prices across the board by as much as $150 billion per year and return nearly $300 million to workers each year.
However, even if passed, the draft rule would keep in place nonsolicitation rules that many health care organizations have put into place. That means that, if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to switch to him or her in the new job.
Within that clause, however, the FTC has specified that if such nonsolicitation agreement has the “equivalent effect” of a noncompete, the agency would deem it such. That means, even if that rule stays, it could be contested and may be interpreted as violating the noncompete law. So there’s value in reading all the fine print should the ban move forward.
Could the ban bring potential downsides?
Most physicians view the potential to break free of a noncompete agreement as a victory. Peter Glennon, an employment litigation attorney with The Glennon Law Firm in Rochester, N.Y., says not so fast. “If you ask anyone if they’d prefer a noncompete agreement, of course they’re going to say no,” he said in an interview. “It sounds like a restriction, one that can hold you back.”
Mr. Glennon believes that there are actually upsides to physician noncompetes. For instance, many noncompetes come with sign-on bonuses that could potentially disappear without the agreements. There’s also the fact that when some physicians sign a noncompete agreement, they then receive pro bono training and continuing education along with marketing and promotion of their skills. Without signing a noncompete, employers may be less incentivized to provide all those benefits to their physician employers.
Those benefits – and the noncompetes – also vary by specialty, Mr. Glennon said. “In 2021, Washington, DC, banned noncompetes for doctors making less than $250,000. So, most generalists there can walk across the street and get a new job. For specialists like cardiologists or neurosurgeons, however, advanced training and marketing benefits matter, so many of them don’t want to lose noncompetes.”
Still, most physicians hope that the FTC’s ban takes hold. Manan Shah, MD, founder, and chief medical officer at Wyndly, an allergy relief startup practice, is one of them.
“Initially, it might disincentivize hospital systems from helping new physicians build up their name and practice because they might be concerned about a physician leaving and starting anew,” he said. “But in the long term, hospitals require physicians to bring their patients to them for care, so the best hospitals will always compete for the best physicians and support them as they build up their practice.”
Dr. Shah views noncompetes as overly prohibitive to physicians. “Right now, if a physician starts a job at a large hospital system and realizes they want to switch jobs, the noncompete distances are so wide they often have to move cities to continue practicing,” he said. “Picking up and starting over in a new city isn’t an option for everyone and can be especially difficult for someone with a family.”
Where Mr. Glennon argued that a physician leaving a team-based practice might harm patients, Shah takes a different perspective. “Imagine you have a doctor whom you trust and have been working with,” he said. “If something changes at their hospital and they decide to move, you literally have to find a new doctor instead of just being able to see them at another location down the street.”
Another potential burden of the noncompete agreements is that they could possibly squelch doctor’s desires to hang up their own shingle. According to Dr. Shah, the agreements make it so that if a physician wants to work independently, it’s nearly impossible to fly solo. “This is frustrating because independent practices have been shown to be more cost effective and allow patients to build better relationships with their doctors,” he claimed.
A 2016 study from Annals of Family Medicine supports that claim, at least for small general practices. Another study appearing in JAMA concurred. It does point out, however, that the cost equation is nuanced and that benefits of larger systems include more resilience to economic downturns and can provide more specialized care.
Will nonprofit hospitals be subject to this noncompete ban?
Further complicating the noncompete ban issue is how it might impact nonprofit institutions versus their for-profit peers. Most hospitals structured as nonprofits would be exempt from the rule because the FTC Act provides that it can enforce against “persons, partnerships, or corporations,” which are further defined as entities “organized to carry on business for their own profit or that of their members.”
The fallout from this, said Dr. Shah, is that it “would disproportionately affect health care providers, since many hospital systems are nonprofits. This is disconcerting because we know that many nonprofit systems make large profits anyway and can offer executive teams’ lucrative packages, while the nurses, assistants, and physicians providing the care are generally not well compensated.”
So far, about nine states plus Washington, D.C., have already put noncompete bans in place, and they may serve as a harbinger of things to come should the federal ban go into effect. Each varies in its specifics. Some, like Indiana, outright ban them, whereas others limit them based on variables like income and industry. “We’re seeing these states responding to local market conditions,” said Darryl Drevna, senior director of regulatory affairs at the American Medical Group Association. “Health care is a hyperlocal market. Depending on the situation, the bans adapt and respond specific to those states.”
Should the federal ban take hold, however, it will supersede whatever rules the individual states have in place.
Some opponents of the federal ban proposal question its authority to begin with, however, Mr. Glennon included. “Many people believe the FTC is overstepping,” he said. “Some people believe that Section 5 of the FTC Act does not give it the authority to police labor markets.”
Mr. Drevna noted that the FTC has taken an aggressive stance, one that will ultimately wind up in the courts. “How it works out is anyone’s guess,” he said. “Ideally, the FTC will consider the comments and concerns of groups like AMGA and realize that states are best suited to regulate in this area.”
In general, the ban’s supporters are employees/physicians; those who oppose it are their employers. Joining the AMGA in speaking out against the noncompete ban is the American Hospital Association, whereas the American College of Emergency Physicians has come out largely in support of the ban.
Still, doctors like Dr. Shah remain hopeful. “I am optimistic that perhaps my colleagues will not continue to be stuck in overrestrictive noncompetes, but I am also realistic,” he said. “Hospital systems are already coming out strongly against this and they have deep pockets, so I won’t be surprised if it does not come to pass.”
A version of this article first appeared on Medscape.com.
Could vitamin D supplementation help in long COVID?
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, in a retrospective, case-matched study.
The lower levels of vitamin D in patients with long COVID were most notable in those with brain fog.
These findings, by Luigi di Filippo, MD, and colleagues, were recently presented at the European Congress of Endocrinology and published in the Journal of Clinical Endocrinology & Metabolism.
“Our data suggest that vitamin D levels should be evaluated in COVID-19 patients after hospital discharge,” wrote the researchers, from San Raffaele Hospital, Milan.
“The role of vitamin D supplementation as a preventive strategy of COVID-19 sequelae should be tested in randomized controlled trials,” they urged.
The researchers also stressed that this was a controlled study in a homogeneous population, it included multiple signs and symptoms of long COVID, and it had a longer follow-up than most previous studies (6 vs. 3 months).
“The highly controlled nature of our study helps us better understand the role of vitamin D deficiency in long COVID and establish that there is likely a link between vitamin D deficiency and long COVID,” senior author Andrea Giustina, MD, said in a press release from the ECE.
“Our study shows that COVID-19 patients with low vitamin D levels are more likely to develop long COVID, but it is not yet known whether vitamin D supplements could improve the symptoms or reduce this risk altogether,” he cautioned.
“If confirmed in large, interventional, randomized controlled trials, [our data suggest] that vitamin D supplementation could represent a possible preventive strategy in reducing the burden of COVID-19 sequelae,” Dr. Giustina and colleagues wrote.
Reasonable to test vitamin D levels, consider supplementation
Invited to comment, Amiel Dror, MD, PhD, who led a related study that showed that people with a vitamin D deficiency were more likely to have severe COVID-19, agreed.
“The novelty and significance of this [new] study lie in the fact that it expands on our current understanding of the interplay between vitamin D and COVID-19, taking it beyond the acute phase of the disease,” said Dr. Dror, from Bar-Ilan University, Safed, Israel.
“It’s striking to see how vitamin D levels continue to influence patients’ health even after recovery from the initial infection,” he noted.
“The findings certainly add weight to the argument for conducting a randomized control trial [RCT],” he continued, which “would enable us to conclusively determine whether vitamin D supplementation can effectively reduce the risk or severity of long COVID.”
“In the interim,” Dr. Dror said, “given the safety profile of vitamin D and its broad health benefits, it could be reasonable to test for vitamin D levels in patients admitted with COVID-19. If levels are found to be low, supplementation could be considered.”
“However, it’s important to note that this should be done under medical supervision,” he cautioned, “and further studies are needed to establish the optimal timing and dosage of supplementation.”
“I anticipate that we’ll see more RCTs [of this] in the future,” he speculated.
Low vitamin D and risk of long COVID
Long COVID is an emerging syndrome that affects 50%-70% of COVID-19 survivors.
Low levels of vitamin D have been associated with increased likelihood of needing mechanical ventilation and worse survival in patients hospitalized with COVID-19, but the risk of long COVID associated with vitamin D has not been known.
Researchers analyzed data from adults aged 18 and older hospitalized at San Raffaele Hospital with a confirmed diagnosis of COVID-19 and discharged during the first pandemic wave, from March to May 2020, and then seen 6-months later for follow-up.
Patients were excluded if they had been admitted to the intensive care unit during hospitalization or had missing medical data or blood samples available to determine (OH) vitamin D levels, at admission and the 6-month follow-up.
Long COVID-19 was defined based on the U.K. National Institute for Health and Care Excellence guidelines as the concomitant presence of at least two or more of 17 signs and symptoms that were absent prior to the COVID-19 infection and could only be attributed to that acute disease.
Researchers identified 50 patients with long COVID at the 6-month follow-up and matched them with 50 patients without long COVID at that time point, based on age, sex, concomitant comorbidities, need for noninvasive mechanical ventilation, and week of evaluation.
Patients were a mean age of 61 years (range, 51-73) and 56% were men; 28% had been on a ventilator during hospitalization for COVID-19.
The most frequent signs and symptoms at 6 months in the patients with long COVID were asthenia (weakness, 38% of patients), dysgeusia (bad taste in the mouth, 34%), dyspnea (shortness of breath, 34%), and anosmia (loss of sense of smell, 24%).
Most symptoms were related to the cardiorespiratory system (42%), the feeling of well-being (42%), or the senses (36%), and fewer patients had symptoms related to neurocognitive impairment (headache or brain fog, 14%), or ear, nose, and throat (12%), or gastrointestinal system (4%).
Patients with long COVID had lower mean 25(OH) vitamin D levels than patients without long COVID (20.1 vs 23.2 ng/mL; P = .03). However, actual vitamin D deficiency levels were similar in both groups.
Two-thirds of patients with low vitamin D levels at hospital admission still presented with low levels at the 6-month follow-up.
Vitamin D levels were significantly lower in patients with neurocognitive symptoms at follow-up (n = 7) than in those without such symptoms (n = 93) (14.6 vs. 20.6 ng/mL; P = .042).
In patients with vitamin D deficiency (< 20 ng/mL) at admission and at follow-up (n = 42), those with long COVID (n = 22) had lower vitamin D levels at follow-up than those without long COVID (n = 20) (12.7 vs. 15.2 ng/mL; P = .041).
And in multiple regression analyses, a lower 25(OH) vitamin D level at follow-up was the only variable that was significantly associated with long COVID (odds ratio, 1.09; 95% confidence interval, 1.01-1.16; P = .008).
The findings “strongly reinforce the clinical usefulness of 25(OH) vitamin D evaluation as a possible modifiable pathophysiological factor underlying this emerging worldwide critical health issue,” the researchers concluded.
The study was supported by Abiogen Pharma. One study author is an employee at Abiogen. Dr. Giustina has reported being a consultant for Abiogen and Takeda and receiving a research grant to his institution from Takeda. Dr. Di Filippo and the other authors reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECE 2023
Hyperbaric oxygen may improve heart function in long COVID
.
Patients with reduced left ventricular global longitudinal strain (GLS) at baseline who received HBOT had a significant increase in GLS, compared with those who received sham treatment.
GLS is a measure of systolic function that is thought to be a predictor of heart failure–related outcomes.
The study also showed that global work efficiency (GWE) and the global work index (GWI) increased in HBOT-treated patients, though not significantly.
“HBOT is an effective treatment for diabetic foot ulcers, decompression sickness in divers, and other conditions, such as cognitive impairment after stroke,” Marina Leitman, MD, of the Sackler School of Medicine, Tel Aviv, said in an interview. Her team also studied HBOT in asymptomatic older patients and found that the treatment seemed to improve left ventricular end systolic function.
“We should open our minds to thinking about this treatment for another indication,” she said. “That is the basis of precision medicine. We have this treatment and know it can be effective for cardiac pathology.
“Now we can say that post-COVID syndrome patients probably should be evaluated with echocardiography and GLS, which is the main parameter that showed improvement in our study,” she added. “If GLS is below normal values, these patients can benefit from HBOT, although additional research is needed to determine the optimal number of sessions.”
Dr. Leitman presented the study at the European Association of Cardiovascular Imaging 2023, a scientific congress of the European Society of Cardiology.
Biomarker changes
The study enrolled 60 hospitalized and nonhospitalized post-COVID syndrome patients with ongoing symptoms for at least 3 months after having mild to moderate symptomatic COVID-19.
Participants were randomized to receive HBOT or a sham procedure five times per week for 8 weeks, for a total of 40 sessions. They underwent echocardiography at baseline and 1-3 weeks after the final session to assess GLS.
The HBOT group received 100% oxygen through a mask at a pressure of two atmospheres for 90 minutes, with 5-minute air breaks every 20 minutes.
The sham group received 21% oxygen by mask at one atmosphere for 90 minutes.
At baseline, 29 participants (48%) had reduced GLS, despite having a normal ejection fraction, Dr. Leitman said. Of those, 16 (53%) were in the HBOT group and 13 (43%) were in the sham group.
The average GLS at baseline across all participants was –17.8%; a normal value is about –20%.
In the HBOT group, GLS increased significantly from –17.8% at baseline to –20.2% after HBOT. In the sham group, GLS was –17.8% at baseline and –19.1% at the end of the study, with no statistically significant difference between the two measurements.
In addition, GWE increased overall after HBOT from 96.3 to 97.1.
Dr. Leitman’s poster showed GLS and myocardial work indices before and after HBOT in a 45-year-old patient. Prior to treatment, GLS was –19%; GWE was 96%; and GWI was 1,833 mm Hg.
After HBOT treatment, GLS was –22%; GWE, 98%; and GWI, 1,911 mm Hg.
Clinical relevance unclear
Scott Gorenstein, MD, associate professor in the department of surgery and medical director of wound care and hyperbaric medicine at NYU Langone–Long Island, New York, commented on the study for this news organization.
“The approach certainly warrants studying, but the benefit is difficult to assess,” he said. “We still don’t understand the mechanism of long COVID, so it’s difficult to go from there to say that HBOT will be an effective therapy.”
That said, he added, “This is probably the best study I’ve seen in that it’s a randomized controlled trial, rather than a case series.”
Nevertheless, he noted, “We have no idea from this study whether the change in GLS is clinically relevant. As a clinician, I can’t now say that HBOT is going to improve heart failure secondary to long COVID. We don’t know whether the participants were New York heart failure class 3 or 4, for example, and all of a sudden went from really sick to really good.”
“There are many interventions that may change markers of cardiac function or inflammation,” he said. “But if they don’t make a difference in quantity or quality of life, is the treatment really valuable?”
Dr. Gorenstein said he would have no problem treating a patient with mild to moderate COVID-related heart failure with HBOT, since his own team’s study conducted near the outset of the pandemic showed it was safe. “But HBOT is an expensive treatment in the U.S. and there still are some risks and side effects, albeit very, very low.”
The study received no funding. Dr. Leitman and Dr. Gorenstein have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
.
Patients with reduced left ventricular global longitudinal strain (GLS) at baseline who received HBOT had a significant increase in GLS, compared with those who received sham treatment.
GLS is a measure of systolic function that is thought to be a predictor of heart failure–related outcomes.
The study also showed that global work efficiency (GWE) and the global work index (GWI) increased in HBOT-treated patients, though not significantly.
“HBOT is an effective treatment for diabetic foot ulcers, decompression sickness in divers, and other conditions, such as cognitive impairment after stroke,” Marina Leitman, MD, of the Sackler School of Medicine, Tel Aviv, said in an interview. Her team also studied HBOT in asymptomatic older patients and found that the treatment seemed to improve left ventricular end systolic function.
“We should open our minds to thinking about this treatment for another indication,” she said. “That is the basis of precision medicine. We have this treatment and know it can be effective for cardiac pathology.
“Now we can say that post-COVID syndrome patients probably should be evaluated with echocardiography and GLS, which is the main parameter that showed improvement in our study,” she added. “If GLS is below normal values, these patients can benefit from HBOT, although additional research is needed to determine the optimal number of sessions.”
Dr. Leitman presented the study at the European Association of Cardiovascular Imaging 2023, a scientific congress of the European Society of Cardiology.
Biomarker changes
The study enrolled 60 hospitalized and nonhospitalized post-COVID syndrome patients with ongoing symptoms for at least 3 months after having mild to moderate symptomatic COVID-19.
Participants were randomized to receive HBOT or a sham procedure five times per week for 8 weeks, for a total of 40 sessions. They underwent echocardiography at baseline and 1-3 weeks after the final session to assess GLS.
The HBOT group received 100% oxygen through a mask at a pressure of two atmospheres for 90 minutes, with 5-minute air breaks every 20 minutes.
The sham group received 21% oxygen by mask at one atmosphere for 90 minutes.
At baseline, 29 participants (48%) had reduced GLS, despite having a normal ejection fraction, Dr. Leitman said. Of those, 16 (53%) were in the HBOT group and 13 (43%) were in the sham group.
The average GLS at baseline across all participants was –17.8%; a normal value is about –20%.
In the HBOT group, GLS increased significantly from –17.8% at baseline to –20.2% after HBOT. In the sham group, GLS was –17.8% at baseline and –19.1% at the end of the study, with no statistically significant difference between the two measurements.
In addition, GWE increased overall after HBOT from 96.3 to 97.1.
Dr. Leitman’s poster showed GLS and myocardial work indices before and after HBOT in a 45-year-old patient. Prior to treatment, GLS was –19%; GWE was 96%; and GWI was 1,833 mm Hg.
After HBOT treatment, GLS was –22%; GWE, 98%; and GWI, 1,911 mm Hg.
Clinical relevance unclear
Scott Gorenstein, MD, associate professor in the department of surgery and medical director of wound care and hyperbaric medicine at NYU Langone–Long Island, New York, commented on the study for this news organization.
“The approach certainly warrants studying, but the benefit is difficult to assess,” he said. “We still don’t understand the mechanism of long COVID, so it’s difficult to go from there to say that HBOT will be an effective therapy.”
That said, he added, “This is probably the best study I’ve seen in that it’s a randomized controlled trial, rather than a case series.”
Nevertheless, he noted, “We have no idea from this study whether the change in GLS is clinically relevant. As a clinician, I can’t now say that HBOT is going to improve heart failure secondary to long COVID. We don’t know whether the participants were New York heart failure class 3 or 4, for example, and all of a sudden went from really sick to really good.”
“There are many interventions that may change markers of cardiac function or inflammation,” he said. “But if they don’t make a difference in quantity or quality of life, is the treatment really valuable?”
Dr. Gorenstein said he would have no problem treating a patient with mild to moderate COVID-related heart failure with HBOT, since his own team’s study conducted near the outset of the pandemic showed it was safe. “But HBOT is an expensive treatment in the U.S. and there still are some risks and side effects, albeit very, very low.”
The study received no funding. Dr. Leitman and Dr. Gorenstein have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
.
Patients with reduced left ventricular global longitudinal strain (GLS) at baseline who received HBOT had a significant increase in GLS, compared with those who received sham treatment.
GLS is a measure of systolic function that is thought to be a predictor of heart failure–related outcomes.
The study also showed that global work efficiency (GWE) and the global work index (GWI) increased in HBOT-treated patients, though not significantly.
“HBOT is an effective treatment for diabetic foot ulcers, decompression sickness in divers, and other conditions, such as cognitive impairment after stroke,” Marina Leitman, MD, of the Sackler School of Medicine, Tel Aviv, said in an interview. Her team also studied HBOT in asymptomatic older patients and found that the treatment seemed to improve left ventricular end systolic function.
“We should open our minds to thinking about this treatment for another indication,” she said. “That is the basis of precision medicine. We have this treatment and know it can be effective for cardiac pathology.
“Now we can say that post-COVID syndrome patients probably should be evaluated with echocardiography and GLS, which is the main parameter that showed improvement in our study,” she added. “If GLS is below normal values, these patients can benefit from HBOT, although additional research is needed to determine the optimal number of sessions.”
Dr. Leitman presented the study at the European Association of Cardiovascular Imaging 2023, a scientific congress of the European Society of Cardiology.
Biomarker changes
The study enrolled 60 hospitalized and nonhospitalized post-COVID syndrome patients with ongoing symptoms for at least 3 months after having mild to moderate symptomatic COVID-19.
Participants were randomized to receive HBOT or a sham procedure five times per week for 8 weeks, for a total of 40 sessions. They underwent echocardiography at baseline and 1-3 weeks after the final session to assess GLS.
The HBOT group received 100% oxygen through a mask at a pressure of two atmospheres for 90 minutes, with 5-minute air breaks every 20 minutes.
The sham group received 21% oxygen by mask at one atmosphere for 90 minutes.
At baseline, 29 participants (48%) had reduced GLS, despite having a normal ejection fraction, Dr. Leitman said. Of those, 16 (53%) were in the HBOT group and 13 (43%) were in the sham group.
The average GLS at baseline across all participants was –17.8%; a normal value is about –20%.
In the HBOT group, GLS increased significantly from –17.8% at baseline to –20.2% after HBOT. In the sham group, GLS was –17.8% at baseline and –19.1% at the end of the study, with no statistically significant difference between the two measurements.
In addition, GWE increased overall after HBOT from 96.3 to 97.1.
Dr. Leitman’s poster showed GLS and myocardial work indices before and after HBOT in a 45-year-old patient. Prior to treatment, GLS was –19%; GWE was 96%; and GWI was 1,833 mm Hg.
After HBOT treatment, GLS was –22%; GWE, 98%; and GWI, 1,911 mm Hg.
Clinical relevance unclear
Scott Gorenstein, MD, associate professor in the department of surgery and medical director of wound care and hyperbaric medicine at NYU Langone–Long Island, New York, commented on the study for this news organization.
“The approach certainly warrants studying, but the benefit is difficult to assess,” he said. “We still don’t understand the mechanism of long COVID, so it’s difficult to go from there to say that HBOT will be an effective therapy.”
That said, he added, “This is probably the best study I’ve seen in that it’s a randomized controlled trial, rather than a case series.”
Nevertheless, he noted, “We have no idea from this study whether the change in GLS is clinically relevant. As a clinician, I can’t now say that HBOT is going to improve heart failure secondary to long COVID. We don’t know whether the participants were New York heart failure class 3 or 4, for example, and all of a sudden went from really sick to really good.”
“There are many interventions that may change markers of cardiac function or inflammation,” he said. “But if they don’t make a difference in quantity or quality of life, is the treatment really valuable?”
Dr. Gorenstein said he would have no problem treating a patient with mild to moderate COVID-related heart failure with HBOT, since his own team’s study conducted near the outset of the pandemic showed it was safe. “But HBOT is an expensive treatment in the U.S. and there still are some risks and side effects, albeit very, very low.”
The study received no funding. Dr. Leitman and Dr. Gorenstein have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM EACVI 2023
CDC: Drug-resistant ringworm reported in New York
BY ALICIA AULT
The in New York.
Tinea, or ringworm, one of the most common fungal infections, is responsible for almost 5 million outpatient visits and 690 hospitalizations annually, according to the CDC.
Over the past 10 years, severe, antifungal-resistant tinea has spread in South Asia, in part because of the rise of a new dermatophyte species known as Trichophyton indotineae, wrote the authors of a report on the two patients with the drug-resistant strain. This epidemic “has likely been driven by misuse and overuse of topical antifungals and corticosteroids,” added the authors, in Morbidity and Mortality Weekly Report.
The cases were detected by a New York City dermatologist. In the first case, a 28-year-old woman developed a widespread pruritic eruption in the summer of 2021. She did not consult a dermatologist until December, when she was in the third trimester of pregnancy. She had large, annular, scaly, pruritic plaques on her neck, abdomen, pubic region, and buttocks, but had no underlying medical conditions, no known exposures to someone with a similar rash, and no recent international travel history.
After she gave birth in January, she started oral terbinafine therapy but had no improvement after 2 weeks. Clinicians administered a 4-week course of itraconazole, which resolved the infection.
The second patient, a 47-year-old woman with no medical conditions, developed a rash while in Bangladesh in the summer of 2022. Other family members had a similar rash. She was treated with topical antifungal and steroid combination creams but had no resolution. Back in the United States, she was prescribed hydrocortisone 2.5% ointment and diphenhydramine, clotrimazole cream, and terbinafine cream in three successive emergency department visits. In December 2022, dermatologists, observing widespread, discrete, scaly, annular, pruritic plaques on the thighs and buttocks, prescribed a 4-week course of oral terbinafine. When the rash did not resolve, she was given 4 weeks of griseofulvin. The rash persisted, although there was 80% improvement. Clinicians are now considering itraconazole. The woman’s son and husband are also being evaluated, as they have similar rashes.
In both cases, skin culture isolates were initially identified as Trichophyton mentagrophytes. Further analysis at the New York State Department of Health’s lab, using Sanger sequencing of the internal transcribed spacer region of the ribosomal gene, followed by phylogenetic analysis, identified the isolates as T. indotineae.
The authors note that culture-based techniques used by most clinical laboratories typically misidentify T. indotineae as T. mentagrophytes or T. interdigitale. Genomic sequencing must be used to properly identify T. indotineae, they wrote.
Clinicians should consider T. indotineae in patients with widespread ringworm, especially if they do not improve with topical antifungals or oral terbinafine, said the authors. If T. indotineae is suspected, state or local public health departments can direct clinicians to testing.
The authors report no relevant financial relationships.
BY ALICIA AULT
The in New York.
Tinea, or ringworm, one of the most common fungal infections, is responsible for almost 5 million outpatient visits and 690 hospitalizations annually, according to the CDC.
Over the past 10 years, severe, antifungal-resistant tinea has spread in South Asia, in part because of the rise of a new dermatophyte species known as Trichophyton indotineae, wrote the authors of a report on the two patients with the drug-resistant strain. This epidemic “has likely been driven by misuse and overuse of topical antifungals and corticosteroids,” added the authors, in Morbidity and Mortality Weekly Report.
The cases were detected by a New York City dermatologist. In the first case, a 28-year-old woman developed a widespread pruritic eruption in the summer of 2021. She did not consult a dermatologist until December, when she was in the third trimester of pregnancy. She had large, annular, scaly, pruritic plaques on her neck, abdomen, pubic region, and buttocks, but had no underlying medical conditions, no known exposures to someone with a similar rash, and no recent international travel history.
After she gave birth in January, she started oral terbinafine therapy but had no improvement after 2 weeks. Clinicians administered a 4-week course of itraconazole, which resolved the infection.
The second patient, a 47-year-old woman with no medical conditions, developed a rash while in Bangladesh in the summer of 2022. Other family members had a similar rash. She was treated with topical antifungal and steroid combination creams but had no resolution. Back in the United States, she was prescribed hydrocortisone 2.5% ointment and diphenhydramine, clotrimazole cream, and terbinafine cream in three successive emergency department visits. In December 2022, dermatologists, observing widespread, discrete, scaly, annular, pruritic plaques on the thighs and buttocks, prescribed a 4-week course of oral terbinafine. When the rash did not resolve, she was given 4 weeks of griseofulvin. The rash persisted, although there was 80% improvement. Clinicians are now considering itraconazole. The woman’s son and husband are also being evaluated, as they have similar rashes.
In both cases, skin culture isolates were initially identified as Trichophyton mentagrophytes. Further analysis at the New York State Department of Health’s lab, using Sanger sequencing of the internal transcribed spacer region of the ribosomal gene, followed by phylogenetic analysis, identified the isolates as T. indotineae.
The authors note that culture-based techniques used by most clinical laboratories typically misidentify T. indotineae as T. mentagrophytes or T. interdigitale. Genomic sequencing must be used to properly identify T. indotineae, they wrote.
Clinicians should consider T. indotineae in patients with widespread ringworm, especially if they do not improve with topical antifungals or oral terbinafine, said the authors. If T. indotineae is suspected, state or local public health departments can direct clinicians to testing.
The authors report no relevant financial relationships.
BY ALICIA AULT
The in New York.
Tinea, or ringworm, one of the most common fungal infections, is responsible for almost 5 million outpatient visits and 690 hospitalizations annually, according to the CDC.
Over the past 10 years, severe, antifungal-resistant tinea has spread in South Asia, in part because of the rise of a new dermatophyte species known as Trichophyton indotineae, wrote the authors of a report on the two patients with the drug-resistant strain. This epidemic “has likely been driven by misuse and overuse of topical antifungals and corticosteroids,” added the authors, in Morbidity and Mortality Weekly Report.
The cases were detected by a New York City dermatologist. In the first case, a 28-year-old woman developed a widespread pruritic eruption in the summer of 2021. She did not consult a dermatologist until December, when she was in the third trimester of pregnancy. She had large, annular, scaly, pruritic plaques on her neck, abdomen, pubic region, and buttocks, but had no underlying medical conditions, no known exposures to someone with a similar rash, and no recent international travel history.
After she gave birth in January, she started oral terbinafine therapy but had no improvement after 2 weeks. Clinicians administered a 4-week course of itraconazole, which resolved the infection.
The second patient, a 47-year-old woman with no medical conditions, developed a rash while in Bangladesh in the summer of 2022. Other family members had a similar rash. She was treated with topical antifungal and steroid combination creams but had no resolution. Back in the United States, she was prescribed hydrocortisone 2.5% ointment and diphenhydramine, clotrimazole cream, and terbinafine cream in three successive emergency department visits. In December 2022, dermatologists, observing widespread, discrete, scaly, annular, pruritic plaques on the thighs and buttocks, prescribed a 4-week course of oral terbinafine. When the rash did not resolve, she was given 4 weeks of griseofulvin. The rash persisted, although there was 80% improvement. Clinicians are now considering itraconazole. The woman’s son and husband are also being evaluated, as they have similar rashes.
In both cases, skin culture isolates were initially identified as Trichophyton mentagrophytes. Further analysis at the New York State Department of Health’s lab, using Sanger sequencing of the internal transcribed spacer region of the ribosomal gene, followed by phylogenetic analysis, identified the isolates as T. indotineae.
The authors note that culture-based techniques used by most clinical laboratories typically misidentify T. indotineae as T. mentagrophytes or T. interdigitale. Genomic sequencing must be used to properly identify T. indotineae, they wrote.
Clinicians should consider T. indotineae in patients with widespread ringworm, especially if they do not improve with topical antifungals or oral terbinafine, said the authors. If T. indotineae is suspected, state or local public health departments can direct clinicians to testing.
The authors report no relevant financial relationships.
Here’s how we can rebuild trust in vaccines
When people ask Paul Offit, MD, what worries him the most about the COVID-19 pandemic, he names two concerns. “One is the lack of socialization and education that came from keeping kids out of school for so long,” Dr. Offit said in a recent interview. “And I think vaccines have suffered.”
Dr. Offit is director of the Vaccine Education Center and a professor of pediatrics at Children’s Hospital of Philadelphia. He has watched with alarm as the American public appears to be losing faith in the lifesaving vaccines the public health community has worked hard to promote. The Centers for Disease Control and Prevention estimates that the proportion of kids entering kindergarten who have received state-required vaccines dipped to 94% in the 2020-2021 school year – a full point less than the year before the pandemic – then dropped by another percentage point, to 93%, the following year.
Although a couple of percentage points may sound trivial, were only 93% of kindergarteners to receive the vaccine against measles, mumps, and rubella (MMR), approximately 250,000 vulnerable 5-year-olds could spark the next big outbreak, such as the recent measles outbreaks in Ohio and Minnesota.
Dr. Offit is one of many public health officials and clinicians who are working to reverse the concerning trends in pediatric vaccinations.
“I just don’t want to see an outbreak of something that we could have avoided because we were not protected enough,” Judith Shlay, MD, associate director of the Public Health Institute at Denver Health, said.
Official stumbles in part to blame
Disruptions in health care from the COVID-19 pandemic certainly played a role in the decline. Parents were afraid to expose their children to other sick kids, providers shifted to a telehealth model, and routine preventive care was difficult to access.
But Dr. Offit also blamed erosion of trust on mistakes made by government and public health institutions for the alarming trend. “I think that health care professionals have lost some level of trust in the Food and Drug Administration and CDC.”
He cited as an example poor messaging during a large outbreak in Massachusetts in summer 2021, when the CDC published a report that highlighted the high proportion of COVID-19 cases among vaccinated people. Health officials called those cases “breakthrough” infections, although most were mild or asymptomatic.
Dr. Offit said the CDC should have focused the message instead on the low rate (1%) of hospitalizations and the low number of deaths from the infections. Instead, they had to walk back their promise that vaccinated people didn’t need to wear masks. At other times, the Biden administration pressured public health officials by promising to make booster shots available to the American public when the FDA and CDC felt they lacked evidence to recommend the injections.
Rupali Limaye, PhD, an associate professor of international health at the Bloomberg School of Public Health at Johns Hopkins University, Baltimore, studies vaccine behavior and decision-making. She would go a step further in characterizing the roots of worsening vaccine hesitancy.
“In the last 20 years, we’ve seen there’s less and less trust in health care providers in general,” Dr. Limaye said. “More people are turning to their social networks or social contacts for that kind of information.” In the maelstrom of the COVID-19 pandemic, digital social networks facilitated the spread of misinformation about COVID-19 faster than scientists could unravel the mysteries of the disease.
“There’s always been this underlying hesitancy for some people about vaccines,” Dr. Shlay said. But she has noticed more resistance to the COVID-19 vaccine from parents nervous about the new mRNA technology. “There was a lot of politicization of the vaccine, even though the mRNA vaccine technology has been around for a long time,” she said.
Multipronged approaches
Dr. Shlay is committed to restoring childhood vaccination uptake to prepandemic levels now that clinics are open again. To do so, she is relying on a combination of quality improvement strategies and outreach to undervaccinated populations.
Denver Health, for instance, offers vaccinations at any inpatient or outpatient visit – not just well-child visits – with the help of alerts built into their electronic health records that notify clinicians if a patient is due for a vaccine.
COVID-19 revealed marked health inequities in underserved communities as Black, Hispanic, and people from other minority communities experienced higher rates of COVID-19 cases and deaths, compared with White people. The Public Health Institute, which is part of Denver Health, has responded with vaccine outreach teams that go to schools, shelters, churches, and community-based organizations to vaccinate children. They focus their efforts on areas where immunization rates are low. Health centers in schools throughout Colorado vaccinate students, and the Public Health Institute partners with Denver-area public schools to provide vaccines to students in schools that don’t have such centers. (They also provide dental care and behavioral health services.)
But it is unlikely that restoring clinic operations and making vaccines more accessible will fill the gap. After 3 years of fear and mistrust, parents are still nervous about routine shots. To help clinicians facilitate conversations about vaccination, Denver Health trains providers in communication techniques using motivational interviewing (MI), a collaborative goal-oriented approach that encourages changes in health behaviors.
Dr. Shlay, who stressed the value of persistence, advised, “Through motivational interviewing, discussing things, talking about it, you can actually address most of the concerns.”
Giving parents a boost in the right direction
That spirit drives the work of Boost Oregon, a parent-led nonprofit organization founded in 2015 that helps parents make science-based decisions for themselves and their families. Even before the pandemic, primary care providers needed better strategies for addressing parents who had concerns about vaccines and found themselves failing in the effort while trying to see 20 patients a day.
For families that have questions about vaccines, Boost Oregon holds community meetings in which parents meet with clinicians, share their concerns with other parents, and get answers to their questions in a nonjudgmental way. The 1- to 2-hour sessions enable deeper discussions of the issues than many clinicians can manage in a 20-minute patient visit.
Boost Oregon also trains providers in communication techniques using MI. Ryan Hassan, MD, a pediatrician in private practice who serves as the medical director for the organization, has made the approach an integral part of his day. A key realization for him about the use of MI is that if providers want to build trust with parents, they need to accept that their role is not simply to educate but also to listen.
“Even if it’s the wildest conspiracy theory I’ve ever heard, that is my opportunity to show them that I’m listening and to empathize,” Dr. Hassan said.
His next step, a central tenet of MI, is to make reflective statements that summarize the parent’s concerns, demonstrate empathy, and help him get to the heart of their concerns. He then tailors his message to their issues.
Dr. Hassan tells people who are learning the technique to acknowledge that patients have the autonomy to make their own decisions. Coercing them into a decision is unhelpful and potentially counterproductive. “You can’t change anyone else’s mind,” he said. “You have to help them change their own mind.”
Dr. Limaye reinforced that message. Overwhelmed by conflicting messages on the internet, people are just trying to find answers. She trains providers not to dismiss patients’ concerns, because dismissal erodes trust.
“When you’re dealing with misinformation and conspiracy, to me, one thing to keep in mind is that it’s the long game,” Dr. Limaye said, “You’re not going to be able to sway them in one conversation.”
Can the powers of social media be harnessed for pro-vaccine messaging? Dr. Limaye has studied social media strategies to promote vaccine acceptance and has identified several elements that can be useful for swaying opinions about vaccine.
One is the messenger – as people trust their physicians less, “it’s important to find influencers that people might trust to actually spread a message,” she said. Another factor is that as society has become more polarized, interaction with the leadership of groups that hold influence has become key. To promote vaccine acceptance, for example, leaders of moms’ groups on Facebook could be equipped with evidence-based information.
“It’s important for us to reach out and engage with those that are leaders in those groups, because they kind of hold the power,” Dr. Limaye said.
Framing the message is critical. Dr. Limaye has found that personal narratives can be persuasive and that to influence vaccine behavior, it is necessary to tailor the approach to the specific audience. Danish researchers, for example, in 2017 launched a campaign to increase uptake of HPV vaccinations among teenagers. The researchers provided facts about the safety and effectiveness of the vaccine, cited posts by clinicians about the importance of immunization against the virus, and relayed personal stories, such as one about a father who chose to vaccinate his daughter and another about a blogger’s encounter with a woman with cervical cancer. The researchers found that the highest engagement rates were achieved through personal content and that such content generated the highest proportion of positive comments.
According to Dr. Limaye, to change behavior, social media messaging must address the issues of risk perception and self-efficacy. For risk perception regarding vaccines, a successful message needs to address the parents’ questions about whether their child is at risk for catching a disease, such as measles or pertussis, and if they are, whether the child will wind up in the hospital.
Self-efficacy is the belief that one can accomplish a task. An effective message would provide information on where to find free or low-cost vaccines and would identify locations that are easy to reach and that have expanded hours for working parents, Dr. Limaye said.
What’s the best approach for boosting vaccination rates in the post-pandemic era? In the 1850s, Massachusetts enacted the first vaccine mandate in the United States to prevent smallpox, and by the 1900s, similar laws had been passed in almost half of states. But recent polls suggest that support for vaccine mandates is dwindling. In a poll by the Kaiser Family Foundation last fall, 71% of adults said that healthy children should be required to be vaccinated against measles before entering school, which was down from 82% in a similar poll in 2019.
So perhaps a better approach for promoting vaccine confidence in the 21st century would involve wider use of MI by clinicians and more focus by public health agencies taking advantage of the potential power of social media. As Dr. Offit put it, “I think trust is the key thing.”
Dr. Offit, Dr. Limaye, Dr. Shlay, and Dr. Hassan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When people ask Paul Offit, MD, what worries him the most about the COVID-19 pandemic, he names two concerns. “One is the lack of socialization and education that came from keeping kids out of school for so long,” Dr. Offit said in a recent interview. “And I think vaccines have suffered.”
Dr. Offit is director of the Vaccine Education Center and a professor of pediatrics at Children’s Hospital of Philadelphia. He has watched with alarm as the American public appears to be losing faith in the lifesaving vaccines the public health community has worked hard to promote. The Centers for Disease Control and Prevention estimates that the proportion of kids entering kindergarten who have received state-required vaccines dipped to 94% in the 2020-2021 school year – a full point less than the year before the pandemic – then dropped by another percentage point, to 93%, the following year.
Although a couple of percentage points may sound trivial, were only 93% of kindergarteners to receive the vaccine against measles, mumps, and rubella (MMR), approximately 250,000 vulnerable 5-year-olds could spark the next big outbreak, such as the recent measles outbreaks in Ohio and Minnesota.
Dr. Offit is one of many public health officials and clinicians who are working to reverse the concerning trends in pediatric vaccinations.
“I just don’t want to see an outbreak of something that we could have avoided because we were not protected enough,” Judith Shlay, MD, associate director of the Public Health Institute at Denver Health, said.
Official stumbles in part to blame
Disruptions in health care from the COVID-19 pandemic certainly played a role in the decline. Parents were afraid to expose their children to other sick kids, providers shifted to a telehealth model, and routine preventive care was difficult to access.
But Dr. Offit also blamed erosion of trust on mistakes made by government and public health institutions for the alarming trend. “I think that health care professionals have lost some level of trust in the Food and Drug Administration and CDC.”
He cited as an example poor messaging during a large outbreak in Massachusetts in summer 2021, when the CDC published a report that highlighted the high proportion of COVID-19 cases among vaccinated people. Health officials called those cases “breakthrough” infections, although most were mild or asymptomatic.
Dr. Offit said the CDC should have focused the message instead on the low rate (1%) of hospitalizations and the low number of deaths from the infections. Instead, they had to walk back their promise that vaccinated people didn’t need to wear masks. At other times, the Biden administration pressured public health officials by promising to make booster shots available to the American public when the FDA and CDC felt they lacked evidence to recommend the injections.
Rupali Limaye, PhD, an associate professor of international health at the Bloomberg School of Public Health at Johns Hopkins University, Baltimore, studies vaccine behavior and decision-making. She would go a step further in characterizing the roots of worsening vaccine hesitancy.
“In the last 20 years, we’ve seen there’s less and less trust in health care providers in general,” Dr. Limaye said. “More people are turning to their social networks or social contacts for that kind of information.” In the maelstrom of the COVID-19 pandemic, digital social networks facilitated the spread of misinformation about COVID-19 faster than scientists could unravel the mysteries of the disease.
“There’s always been this underlying hesitancy for some people about vaccines,” Dr. Shlay said. But she has noticed more resistance to the COVID-19 vaccine from parents nervous about the new mRNA technology. “There was a lot of politicization of the vaccine, even though the mRNA vaccine technology has been around for a long time,” she said.
Multipronged approaches
Dr. Shlay is committed to restoring childhood vaccination uptake to prepandemic levels now that clinics are open again. To do so, she is relying on a combination of quality improvement strategies and outreach to undervaccinated populations.
Denver Health, for instance, offers vaccinations at any inpatient or outpatient visit – not just well-child visits – with the help of alerts built into their electronic health records that notify clinicians if a patient is due for a vaccine.
COVID-19 revealed marked health inequities in underserved communities as Black, Hispanic, and people from other minority communities experienced higher rates of COVID-19 cases and deaths, compared with White people. The Public Health Institute, which is part of Denver Health, has responded with vaccine outreach teams that go to schools, shelters, churches, and community-based organizations to vaccinate children. They focus their efforts on areas where immunization rates are low. Health centers in schools throughout Colorado vaccinate students, and the Public Health Institute partners with Denver-area public schools to provide vaccines to students in schools that don’t have such centers. (They also provide dental care and behavioral health services.)
But it is unlikely that restoring clinic operations and making vaccines more accessible will fill the gap. After 3 years of fear and mistrust, parents are still nervous about routine shots. To help clinicians facilitate conversations about vaccination, Denver Health trains providers in communication techniques using motivational interviewing (MI), a collaborative goal-oriented approach that encourages changes in health behaviors.
Dr. Shlay, who stressed the value of persistence, advised, “Through motivational interviewing, discussing things, talking about it, you can actually address most of the concerns.”
Giving parents a boost in the right direction
That spirit drives the work of Boost Oregon, a parent-led nonprofit organization founded in 2015 that helps parents make science-based decisions for themselves and their families. Even before the pandemic, primary care providers needed better strategies for addressing parents who had concerns about vaccines and found themselves failing in the effort while trying to see 20 patients a day.
For families that have questions about vaccines, Boost Oregon holds community meetings in which parents meet with clinicians, share their concerns with other parents, and get answers to their questions in a nonjudgmental way. The 1- to 2-hour sessions enable deeper discussions of the issues than many clinicians can manage in a 20-minute patient visit.
Boost Oregon also trains providers in communication techniques using MI. Ryan Hassan, MD, a pediatrician in private practice who serves as the medical director for the organization, has made the approach an integral part of his day. A key realization for him about the use of MI is that if providers want to build trust with parents, they need to accept that their role is not simply to educate but also to listen.
“Even if it’s the wildest conspiracy theory I’ve ever heard, that is my opportunity to show them that I’m listening and to empathize,” Dr. Hassan said.
His next step, a central tenet of MI, is to make reflective statements that summarize the parent’s concerns, demonstrate empathy, and help him get to the heart of their concerns. He then tailors his message to their issues.
Dr. Hassan tells people who are learning the technique to acknowledge that patients have the autonomy to make their own decisions. Coercing them into a decision is unhelpful and potentially counterproductive. “You can’t change anyone else’s mind,” he said. “You have to help them change their own mind.”
Dr. Limaye reinforced that message. Overwhelmed by conflicting messages on the internet, people are just trying to find answers. She trains providers not to dismiss patients’ concerns, because dismissal erodes trust.
“When you’re dealing with misinformation and conspiracy, to me, one thing to keep in mind is that it’s the long game,” Dr. Limaye said, “You’re not going to be able to sway them in one conversation.”
Can the powers of social media be harnessed for pro-vaccine messaging? Dr. Limaye has studied social media strategies to promote vaccine acceptance and has identified several elements that can be useful for swaying opinions about vaccine.
One is the messenger – as people trust their physicians less, “it’s important to find influencers that people might trust to actually spread a message,” she said. Another factor is that as society has become more polarized, interaction with the leadership of groups that hold influence has become key. To promote vaccine acceptance, for example, leaders of moms’ groups on Facebook could be equipped with evidence-based information.
“It’s important for us to reach out and engage with those that are leaders in those groups, because they kind of hold the power,” Dr. Limaye said.
Framing the message is critical. Dr. Limaye has found that personal narratives can be persuasive and that to influence vaccine behavior, it is necessary to tailor the approach to the specific audience. Danish researchers, for example, in 2017 launched a campaign to increase uptake of HPV vaccinations among teenagers. The researchers provided facts about the safety and effectiveness of the vaccine, cited posts by clinicians about the importance of immunization against the virus, and relayed personal stories, such as one about a father who chose to vaccinate his daughter and another about a blogger’s encounter with a woman with cervical cancer. The researchers found that the highest engagement rates were achieved through personal content and that such content generated the highest proportion of positive comments.
According to Dr. Limaye, to change behavior, social media messaging must address the issues of risk perception and self-efficacy. For risk perception regarding vaccines, a successful message needs to address the parents’ questions about whether their child is at risk for catching a disease, such as measles or pertussis, and if they are, whether the child will wind up in the hospital.
Self-efficacy is the belief that one can accomplish a task. An effective message would provide information on where to find free or low-cost vaccines and would identify locations that are easy to reach and that have expanded hours for working parents, Dr. Limaye said.
What’s the best approach for boosting vaccination rates in the post-pandemic era? In the 1850s, Massachusetts enacted the first vaccine mandate in the United States to prevent smallpox, and by the 1900s, similar laws had been passed in almost half of states. But recent polls suggest that support for vaccine mandates is dwindling. In a poll by the Kaiser Family Foundation last fall, 71% of adults said that healthy children should be required to be vaccinated against measles before entering school, which was down from 82% in a similar poll in 2019.
So perhaps a better approach for promoting vaccine confidence in the 21st century would involve wider use of MI by clinicians and more focus by public health agencies taking advantage of the potential power of social media. As Dr. Offit put it, “I think trust is the key thing.”
Dr. Offit, Dr. Limaye, Dr. Shlay, and Dr. Hassan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
When people ask Paul Offit, MD, what worries him the most about the COVID-19 pandemic, he names two concerns. “One is the lack of socialization and education that came from keeping kids out of school for so long,” Dr. Offit said in a recent interview. “And I think vaccines have suffered.”
Dr. Offit is director of the Vaccine Education Center and a professor of pediatrics at Children’s Hospital of Philadelphia. He has watched with alarm as the American public appears to be losing faith in the lifesaving vaccines the public health community has worked hard to promote. The Centers for Disease Control and Prevention estimates that the proportion of kids entering kindergarten who have received state-required vaccines dipped to 94% in the 2020-2021 school year – a full point less than the year before the pandemic – then dropped by another percentage point, to 93%, the following year.
Although a couple of percentage points may sound trivial, were only 93% of kindergarteners to receive the vaccine against measles, mumps, and rubella (MMR), approximately 250,000 vulnerable 5-year-olds could spark the next big outbreak, such as the recent measles outbreaks in Ohio and Minnesota.
Dr. Offit is one of many public health officials and clinicians who are working to reverse the concerning trends in pediatric vaccinations.
“I just don’t want to see an outbreak of something that we could have avoided because we were not protected enough,” Judith Shlay, MD, associate director of the Public Health Institute at Denver Health, said.
Official stumbles in part to blame
Disruptions in health care from the COVID-19 pandemic certainly played a role in the decline. Parents were afraid to expose their children to other sick kids, providers shifted to a telehealth model, and routine preventive care was difficult to access.
But Dr. Offit also blamed erosion of trust on mistakes made by government and public health institutions for the alarming trend. “I think that health care professionals have lost some level of trust in the Food and Drug Administration and CDC.”
He cited as an example poor messaging during a large outbreak in Massachusetts in summer 2021, when the CDC published a report that highlighted the high proportion of COVID-19 cases among vaccinated people. Health officials called those cases “breakthrough” infections, although most were mild or asymptomatic.
Dr. Offit said the CDC should have focused the message instead on the low rate (1%) of hospitalizations and the low number of deaths from the infections. Instead, they had to walk back their promise that vaccinated people didn’t need to wear masks. At other times, the Biden administration pressured public health officials by promising to make booster shots available to the American public when the FDA and CDC felt they lacked evidence to recommend the injections.
Rupali Limaye, PhD, an associate professor of international health at the Bloomberg School of Public Health at Johns Hopkins University, Baltimore, studies vaccine behavior and decision-making. She would go a step further in characterizing the roots of worsening vaccine hesitancy.
“In the last 20 years, we’ve seen there’s less and less trust in health care providers in general,” Dr. Limaye said. “More people are turning to their social networks or social contacts for that kind of information.” In the maelstrom of the COVID-19 pandemic, digital social networks facilitated the spread of misinformation about COVID-19 faster than scientists could unravel the mysteries of the disease.
“There’s always been this underlying hesitancy for some people about vaccines,” Dr. Shlay said. But she has noticed more resistance to the COVID-19 vaccine from parents nervous about the new mRNA technology. “There was a lot of politicization of the vaccine, even though the mRNA vaccine technology has been around for a long time,” she said.
Multipronged approaches
Dr. Shlay is committed to restoring childhood vaccination uptake to prepandemic levels now that clinics are open again. To do so, she is relying on a combination of quality improvement strategies and outreach to undervaccinated populations.
Denver Health, for instance, offers vaccinations at any inpatient or outpatient visit – not just well-child visits – with the help of alerts built into their electronic health records that notify clinicians if a patient is due for a vaccine.
COVID-19 revealed marked health inequities in underserved communities as Black, Hispanic, and people from other minority communities experienced higher rates of COVID-19 cases and deaths, compared with White people. The Public Health Institute, which is part of Denver Health, has responded with vaccine outreach teams that go to schools, shelters, churches, and community-based organizations to vaccinate children. They focus their efforts on areas where immunization rates are low. Health centers in schools throughout Colorado vaccinate students, and the Public Health Institute partners with Denver-area public schools to provide vaccines to students in schools that don’t have such centers. (They also provide dental care and behavioral health services.)
But it is unlikely that restoring clinic operations and making vaccines more accessible will fill the gap. After 3 years of fear and mistrust, parents are still nervous about routine shots. To help clinicians facilitate conversations about vaccination, Denver Health trains providers in communication techniques using motivational interviewing (MI), a collaborative goal-oriented approach that encourages changes in health behaviors.
Dr. Shlay, who stressed the value of persistence, advised, “Through motivational interviewing, discussing things, talking about it, you can actually address most of the concerns.”
Giving parents a boost in the right direction
That spirit drives the work of Boost Oregon, a parent-led nonprofit organization founded in 2015 that helps parents make science-based decisions for themselves and their families. Even before the pandemic, primary care providers needed better strategies for addressing parents who had concerns about vaccines and found themselves failing in the effort while trying to see 20 patients a day.
For families that have questions about vaccines, Boost Oregon holds community meetings in which parents meet with clinicians, share their concerns with other parents, and get answers to their questions in a nonjudgmental way. The 1- to 2-hour sessions enable deeper discussions of the issues than many clinicians can manage in a 20-minute patient visit.
Boost Oregon also trains providers in communication techniques using MI. Ryan Hassan, MD, a pediatrician in private practice who serves as the medical director for the organization, has made the approach an integral part of his day. A key realization for him about the use of MI is that if providers want to build trust with parents, they need to accept that their role is not simply to educate but also to listen.
“Even if it’s the wildest conspiracy theory I’ve ever heard, that is my opportunity to show them that I’m listening and to empathize,” Dr. Hassan said.
His next step, a central tenet of MI, is to make reflective statements that summarize the parent’s concerns, demonstrate empathy, and help him get to the heart of their concerns. He then tailors his message to their issues.
Dr. Hassan tells people who are learning the technique to acknowledge that patients have the autonomy to make their own decisions. Coercing them into a decision is unhelpful and potentially counterproductive. “You can’t change anyone else’s mind,” he said. “You have to help them change their own mind.”
Dr. Limaye reinforced that message. Overwhelmed by conflicting messages on the internet, people are just trying to find answers. She trains providers not to dismiss patients’ concerns, because dismissal erodes trust.
“When you’re dealing with misinformation and conspiracy, to me, one thing to keep in mind is that it’s the long game,” Dr. Limaye said, “You’re not going to be able to sway them in one conversation.”
Can the powers of social media be harnessed for pro-vaccine messaging? Dr. Limaye has studied social media strategies to promote vaccine acceptance and has identified several elements that can be useful for swaying opinions about vaccine.
One is the messenger – as people trust their physicians less, “it’s important to find influencers that people might trust to actually spread a message,” she said. Another factor is that as society has become more polarized, interaction with the leadership of groups that hold influence has become key. To promote vaccine acceptance, for example, leaders of moms’ groups on Facebook could be equipped with evidence-based information.
“It’s important for us to reach out and engage with those that are leaders in those groups, because they kind of hold the power,” Dr. Limaye said.
Framing the message is critical. Dr. Limaye has found that personal narratives can be persuasive and that to influence vaccine behavior, it is necessary to tailor the approach to the specific audience. Danish researchers, for example, in 2017 launched a campaign to increase uptake of HPV vaccinations among teenagers. The researchers provided facts about the safety and effectiveness of the vaccine, cited posts by clinicians about the importance of immunization against the virus, and relayed personal stories, such as one about a father who chose to vaccinate his daughter and another about a blogger’s encounter with a woman with cervical cancer. The researchers found that the highest engagement rates were achieved through personal content and that such content generated the highest proportion of positive comments.
According to Dr. Limaye, to change behavior, social media messaging must address the issues of risk perception and self-efficacy. For risk perception regarding vaccines, a successful message needs to address the parents’ questions about whether their child is at risk for catching a disease, such as measles or pertussis, and if they are, whether the child will wind up in the hospital.
Self-efficacy is the belief that one can accomplish a task. An effective message would provide information on where to find free or low-cost vaccines and would identify locations that are easy to reach and that have expanded hours for working parents, Dr. Limaye said.
What’s the best approach for boosting vaccination rates in the post-pandemic era? In the 1850s, Massachusetts enacted the first vaccine mandate in the United States to prevent smallpox, and by the 1900s, similar laws had been passed in almost half of states. But recent polls suggest that support for vaccine mandates is dwindling. In a poll by the Kaiser Family Foundation last fall, 71% of adults said that healthy children should be required to be vaccinated against measles before entering school, which was down from 82% in a similar poll in 2019.
So perhaps a better approach for promoting vaccine confidence in the 21st century would involve wider use of MI by clinicians and more focus by public health agencies taking advantage of the potential power of social media. As Dr. Offit put it, “I think trust is the key thing.”
Dr. Offit, Dr. Limaye, Dr. Shlay, and Dr. Hassan report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Medical students gain momentum in effort to ban legacy admissions
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
Five ways docs may qualify for discounts on medical malpractice premiums
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.