User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


CRC screening disparities greatest among those under 55
Adults younger than 55 years were least likely to get screened for colorectal cancer over the past 2 decades, particularly if they were Hispanic or Asian or had a low income, lower education level, or no health insurance, according to a new study published online in Cancer Epidemiology, Biomarkers & Prevention.
The findings have raised concerns that disparities in screening rates will be even greater in adults aged 45-49 years, prompting the need for increased awareness and outreach to ensure that underserved groups have access to screenings.
“Differences in prevalence of screening by race and ethnicity, educational attainment, household income, and health insurance were most pronounced for those ages 50-54 years, whereas older adults experienced larger increases in prevalence across these groups,” wrote Po-Hong Liu, MD, MPH, a clinical investigator at Harvard University, Boston, and his colleagues. “The persistent and worsening disparities we observed in adults 50-54 years may extend to those ages 45-49 as they become eligible for screening.”
The U.S. Preventive Services Task Force shifted their recommendation for colorectal cancer screening in May 2021 to 5 years earlier, advising people to start screenings at 45 instead of 50, which aligns with the recommendations the American Cancer Society made 3 years earlier.
Both organizations made the change because of increasing rates of colorectal cancer in adults under age 50 and research indicating that beginning screenings at age 45 results in fewer cases, fewer deaths, and more life years gained.
“Across all age groups, colorectal cancer screening participation remains below national goals, and the benefits of screening are not equally realized across populations,” senior author Caitlin Murphy, PhD, MPH, associate professor, UTHealth School of Public Health, Houston, said in a prepared statement. “Extra care must be taken to ensure that expanding screening to younger ages does not negatively impact efforts to eliminate disparities in colorectal screening and outcomes nor jeopardize efforts to increase screening initiation among older adults who remain unscreened.”
Data analyzed from 8 years over 2 decades
The researchers analyzed data from the CDC’s cross-sectional National Health Interview Survey during 8 years over the past 2 decades: 2000, 2003, 2005, 2008, 2010, 2013, 2015, and 2018.
The number of participants each year ranged from a low of 21,781 in 2008 to a high of 34,557 in 2013. After excluding participants with a history of colorectal cancer or missing information on screenings, the total population sample included 80,220 participants 50-75 years old.
The researchers considered a person as having been screened if they received at least one recommended screening test within the year covered by the survey, regardless of why they underwent the test.
Recommended tests included sigmoidoscopy, colonoscopy, and stool-based tests for all survey years. In addition, the surveys for 2010, 2015, and 2018 included CT colonography, and the 2018 survey included FIT-DNA.
Screening across population groups
Colorectal cancer screening rates have doubled in the past 2 decades, from 36.7% in 2000 to 66.1% in 2018.
Rates are considerably lower, however, for several key groups, including the youngest group. Less than half (47.6%) of those aged 50-54 years received screenings in 2018, though this was still a nearly 20-point improvement over the 28.2% in this age group who were screened in 2000.
Separate from age, several other groups continue to have low screening rates in general, including Hispanics (56.5%, up from 25.9% in 2000), Asians (57.1%, up from 22.6% in 2000), those who have not received a high school degree (53.6%, up from 26.8% in 2000), and those from low income families (56.6%, up from 30.2% in 2000).
The group with the greatest need for more outreach and screenings are people without insurance, only 39.7% of whom were screened in 2018, a modest increase from 30.2% in 2000.
The biggest increase in screenings over time occurred in those aged 70-75 years, from 46.4% in 2000 to 78% in 2018 overall.
Racial/ethnic, economic, education, and insurance-based disparities were particularly evident the younger people were, including in terms of progress made over time.
For example, screenings of non-Hispanic White people aged 50-54 years improved 21 points (30.3% to 51%) between 2000 and 2018, compared with 19 points in Hispanics (16.7% to 35.5%) and 15 points in Asians (17.3% to 32.3%). Fortunately, Black Americans made even greater strides than White Americans with a 27-point increase during that time (23.4% to 50%).
Similarly, income correlated with expansion in screening rates for 50- to 54-year-olds: Those earning at least 400% over the federal poverty line improved 20 points (from 33.5% to 53.8%), compared with a 16-point improvement in those earning less than 200% above the poverty line (from 19.3% to 35%).
Those with private insurance likewise improved 21 points (from 30.7% to 51.7%), while those in this age group without insurance declined, with just 21.2% getting screened in 2018, compared with 28.2% in 2000. Those on public insurance saw a 15-point improvement, from 27.8% in 2000 to 43.1% in 2018.
“The individual and societal burden of colorectal cancer is especially great among younger adults,” the authors wrote.
The reasons for the much lower prevalence of screening in those under 55, the authors suggested, is likely due to less concern about colorectal cancer, less access to medical care (including being underinsured or uninsured), and the barriers created by competing priorities, such as work schedules, family responsibilities, and caregiving. The latter may be particularly true in underserved populations, the authors noted.
“Screening programs must consider the barriers unique to younger adults, ensuring the benefits of screening are equally realized by all population groups,” the authors concluded.
The research was funded by the National Institutes of Health and the Cancer Prevention and Research Institute of Texas. One author reported grants from Epigenomics and Freenome and personal fees from Guardant Health. Another author reported personal fees from Freenome, and a third author reported personal fees from Exact Sciences. No other authors had industry disclosures.
A version of this article first appeared on Medscape.com.
Adults younger than 55 years were least likely to get screened for colorectal cancer over the past 2 decades, particularly if they were Hispanic or Asian or had a low income, lower education level, or no health insurance, according to a new study published online in Cancer Epidemiology, Biomarkers & Prevention.
The findings have raised concerns that disparities in screening rates will be even greater in adults aged 45-49 years, prompting the need for increased awareness and outreach to ensure that underserved groups have access to screenings.
“Differences in prevalence of screening by race and ethnicity, educational attainment, household income, and health insurance were most pronounced for those ages 50-54 years, whereas older adults experienced larger increases in prevalence across these groups,” wrote Po-Hong Liu, MD, MPH, a clinical investigator at Harvard University, Boston, and his colleagues. “The persistent and worsening disparities we observed in adults 50-54 years may extend to those ages 45-49 as they become eligible for screening.”
The U.S. Preventive Services Task Force shifted their recommendation for colorectal cancer screening in May 2021 to 5 years earlier, advising people to start screenings at 45 instead of 50, which aligns with the recommendations the American Cancer Society made 3 years earlier.
Both organizations made the change because of increasing rates of colorectal cancer in adults under age 50 and research indicating that beginning screenings at age 45 results in fewer cases, fewer deaths, and more life years gained.
“Across all age groups, colorectal cancer screening participation remains below national goals, and the benefits of screening are not equally realized across populations,” senior author Caitlin Murphy, PhD, MPH, associate professor, UTHealth School of Public Health, Houston, said in a prepared statement. “Extra care must be taken to ensure that expanding screening to younger ages does not negatively impact efforts to eliminate disparities in colorectal screening and outcomes nor jeopardize efforts to increase screening initiation among older adults who remain unscreened.”
Data analyzed from 8 years over 2 decades
The researchers analyzed data from the CDC’s cross-sectional National Health Interview Survey during 8 years over the past 2 decades: 2000, 2003, 2005, 2008, 2010, 2013, 2015, and 2018.
The number of participants each year ranged from a low of 21,781 in 2008 to a high of 34,557 in 2013. After excluding participants with a history of colorectal cancer or missing information on screenings, the total population sample included 80,220 participants 50-75 years old.
The researchers considered a person as having been screened if they received at least one recommended screening test within the year covered by the survey, regardless of why they underwent the test.
Recommended tests included sigmoidoscopy, colonoscopy, and stool-based tests for all survey years. In addition, the surveys for 2010, 2015, and 2018 included CT colonography, and the 2018 survey included FIT-DNA.
Screening across population groups
Colorectal cancer screening rates have doubled in the past 2 decades, from 36.7% in 2000 to 66.1% in 2018.
Rates are considerably lower, however, for several key groups, including the youngest group. Less than half (47.6%) of those aged 50-54 years received screenings in 2018, though this was still a nearly 20-point improvement over the 28.2% in this age group who were screened in 2000.
Separate from age, several other groups continue to have low screening rates in general, including Hispanics (56.5%, up from 25.9% in 2000), Asians (57.1%, up from 22.6% in 2000), those who have not received a high school degree (53.6%, up from 26.8% in 2000), and those from low income families (56.6%, up from 30.2% in 2000).
The group with the greatest need for more outreach and screenings are people without insurance, only 39.7% of whom were screened in 2018, a modest increase from 30.2% in 2000.
The biggest increase in screenings over time occurred in those aged 70-75 years, from 46.4% in 2000 to 78% in 2018 overall.
Racial/ethnic, economic, education, and insurance-based disparities were particularly evident the younger people were, including in terms of progress made over time.
For example, screenings of non-Hispanic White people aged 50-54 years improved 21 points (30.3% to 51%) between 2000 and 2018, compared with 19 points in Hispanics (16.7% to 35.5%) and 15 points in Asians (17.3% to 32.3%). Fortunately, Black Americans made even greater strides than White Americans with a 27-point increase during that time (23.4% to 50%).
Similarly, income correlated with expansion in screening rates for 50- to 54-year-olds: Those earning at least 400% over the federal poverty line improved 20 points (from 33.5% to 53.8%), compared with a 16-point improvement in those earning less than 200% above the poverty line (from 19.3% to 35%).
Those with private insurance likewise improved 21 points (from 30.7% to 51.7%), while those in this age group without insurance declined, with just 21.2% getting screened in 2018, compared with 28.2% in 2000. Those on public insurance saw a 15-point improvement, from 27.8% in 2000 to 43.1% in 2018.
“The individual and societal burden of colorectal cancer is especially great among younger adults,” the authors wrote.
The reasons for the much lower prevalence of screening in those under 55, the authors suggested, is likely due to less concern about colorectal cancer, less access to medical care (including being underinsured or uninsured), and the barriers created by competing priorities, such as work schedules, family responsibilities, and caregiving. The latter may be particularly true in underserved populations, the authors noted.
“Screening programs must consider the barriers unique to younger adults, ensuring the benefits of screening are equally realized by all population groups,” the authors concluded.
The research was funded by the National Institutes of Health and the Cancer Prevention and Research Institute of Texas. One author reported grants from Epigenomics and Freenome and personal fees from Guardant Health. Another author reported personal fees from Freenome, and a third author reported personal fees from Exact Sciences. No other authors had industry disclosures.
A version of this article first appeared on Medscape.com.
Adults younger than 55 years were least likely to get screened for colorectal cancer over the past 2 decades, particularly if they were Hispanic or Asian or had a low income, lower education level, or no health insurance, according to a new study published online in Cancer Epidemiology, Biomarkers & Prevention.
The findings have raised concerns that disparities in screening rates will be even greater in adults aged 45-49 years, prompting the need for increased awareness and outreach to ensure that underserved groups have access to screenings.
“Differences in prevalence of screening by race and ethnicity, educational attainment, household income, and health insurance were most pronounced for those ages 50-54 years, whereas older adults experienced larger increases in prevalence across these groups,” wrote Po-Hong Liu, MD, MPH, a clinical investigator at Harvard University, Boston, and his colleagues. “The persistent and worsening disparities we observed in adults 50-54 years may extend to those ages 45-49 as they become eligible for screening.”
The U.S. Preventive Services Task Force shifted their recommendation for colorectal cancer screening in May 2021 to 5 years earlier, advising people to start screenings at 45 instead of 50, which aligns with the recommendations the American Cancer Society made 3 years earlier.
Both organizations made the change because of increasing rates of colorectal cancer in adults under age 50 and research indicating that beginning screenings at age 45 results in fewer cases, fewer deaths, and more life years gained.
“Across all age groups, colorectal cancer screening participation remains below national goals, and the benefits of screening are not equally realized across populations,” senior author Caitlin Murphy, PhD, MPH, associate professor, UTHealth School of Public Health, Houston, said in a prepared statement. “Extra care must be taken to ensure that expanding screening to younger ages does not negatively impact efforts to eliminate disparities in colorectal screening and outcomes nor jeopardize efforts to increase screening initiation among older adults who remain unscreened.”
Data analyzed from 8 years over 2 decades
The researchers analyzed data from the CDC’s cross-sectional National Health Interview Survey during 8 years over the past 2 decades: 2000, 2003, 2005, 2008, 2010, 2013, 2015, and 2018.
The number of participants each year ranged from a low of 21,781 in 2008 to a high of 34,557 in 2013. After excluding participants with a history of colorectal cancer or missing information on screenings, the total population sample included 80,220 participants 50-75 years old.
The researchers considered a person as having been screened if they received at least one recommended screening test within the year covered by the survey, regardless of why they underwent the test.
Recommended tests included sigmoidoscopy, colonoscopy, and stool-based tests for all survey years. In addition, the surveys for 2010, 2015, and 2018 included CT colonography, and the 2018 survey included FIT-DNA.
Screening across population groups
Colorectal cancer screening rates have doubled in the past 2 decades, from 36.7% in 2000 to 66.1% in 2018.
Rates are considerably lower, however, for several key groups, including the youngest group. Less than half (47.6%) of those aged 50-54 years received screenings in 2018, though this was still a nearly 20-point improvement over the 28.2% in this age group who were screened in 2000.
Separate from age, several other groups continue to have low screening rates in general, including Hispanics (56.5%, up from 25.9% in 2000), Asians (57.1%, up from 22.6% in 2000), those who have not received a high school degree (53.6%, up from 26.8% in 2000), and those from low income families (56.6%, up from 30.2% in 2000).
The group with the greatest need for more outreach and screenings are people without insurance, only 39.7% of whom were screened in 2018, a modest increase from 30.2% in 2000.
The biggest increase in screenings over time occurred in those aged 70-75 years, from 46.4% in 2000 to 78% in 2018 overall.
Racial/ethnic, economic, education, and insurance-based disparities were particularly evident the younger people were, including in terms of progress made over time.
For example, screenings of non-Hispanic White people aged 50-54 years improved 21 points (30.3% to 51%) between 2000 and 2018, compared with 19 points in Hispanics (16.7% to 35.5%) and 15 points in Asians (17.3% to 32.3%). Fortunately, Black Americans made even greater strides than White Americans with a 27-point increase during that time (23.4% to 50%).
Similarly, income correlated with expansion in screening rates for 50- to 54-year-olds: Those earning at least 400% over the federal poverty line improved 20 points (from 33.5% to 53.8%), compared with a 16-point improvement in those earning less than 200% above the poverty line (from 19.3% to 35%).
Those with private insurance likewise improved 21 points (from 30.7% to 51.7%), while those in this age group without insurance declined, with just 21.2% getting screened in 2018, compared with 28.2% in 2000. Those on public insurance saw a 15-point improvement, from 27.8% in 2000 to 43.1% in 2018.
“The individual and societal burden of colorectal cancer is especially great among younger adults,” the authors wrote.
The reasons for the much lower prevalence of screening in those under 55, the authors suggested, is likely due to less concern about colorectal cancer, less access to medical care (including being underinsured or uninsured), and the barriers created by competing priorities, such as work schedules, family responsibilities, and caregiving. The latter may be particularly true in underserved populations, the authors noted.
“Screening programs must consider the barriers unique to younger adults, ensuring the benefits of screening are equally realized by all population groups,” the authors concluded.
The research was funded by the National Institutes of Health and the Cancer Prevention and Research Institute of Texas. One author reported grants from Epigenomics and Freenome and personal fees from Guardant Health. Another author reported personal fees from Freenome, and a third author reported personal fees from Exact Sciences. No other authors had industry disclosures.
A version of this article first appeared on Medscape.com.
FROM CANCER EPIDEMIOLOGY, BIOMARKERS AN PREVENTION
FDA warning: Lymphoma drug heightens risk of death
The U.S. Food and Drug Administration issued a warning today that the cancer drug duvelisib (Copiktra, Verastem), a PI3 kinase inhibitor, may increase the risk of death and serious side effects.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib, compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced that it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib following a voluntary request by the drug manufacturer Secura Bio.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by this news organization, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com
The U.S. Food and Drug Administration issued a warning today that the cancer drug duvelisib (Copiktra, Verastem), a PI3 kinase inhibitor, may increase the risk of death and serious side effects.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib, compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced that it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib following a voluntary request by the drug manufacturer Secura Bio.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by this news organization, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com
The U.S. Food and Drug Administration issued a warning today that the cancer drug duvelisib (Copiktra, Verastem), a PI3 kinase inhibitor, may increase the risk of death and serious side effects.
Duvelisib was approved in 2018 to treat adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who had received at least two prior therapies that did not work or stopped working.
However, more recent 5-year overall survival results from the randomized phase 3 DUO clinical trial found a possible increased risk of death with duvelisib, compared with another drug used to treat leukemia and lymphoma, according to an FDA Drug Safety Communication.
“The trial also found Copiktra was associated with a higher risk of serious side effects, including infections, diarrhea, inflammation of the intestines and lungs, skin reactions, and high liver enzyme levels in the blood,” states the warning, which advises prescribers to weigh the risks and benefits of continued use versus use of other treatments.
More specifically, median 5-year overall survival among 319 patients with CLL or SLL in the DUO trial was 52.3 months with duvelisib versus 63.3 months with the monoclonal antibody ofatumumab (hazard ratio, 1.09 overall and 1.06 among patients who received at least two prior lines of therapy).
Serious adverse events of grade 3 or higher were also more common in those treated with duvelisib.
Of note, in April, the FDA also announced that it was withdrawing approval of the relapsed or refractory follicular lymphoma indication for duvelisib following a voluntary request by the drug manufacturer Secura Bio.
A public meeting will be scheduled to discuss the findings of the trial and whether the drug should continue to be prescribed.
This FDA warning follows the agency’s June 1 withdrawal of approval for umbralisib (Ukoniq), another PI3 kinase inhibitor, following an investigation into a “possible increased risk of death.”
As reported by this news organization, umbralisib had received accelerated approval in February 2021 to treat adults with relapsed or refractory marginal zone lymphoma following at least one prior therapy and those with relapsed or refractory follicular lymphoma who had received at least three prior therapies.
“These safety findings were similar for other medicines in the same PI3 kinase inhibitor class, which were discussed at an advisory committee meeting of non-FDA experts in April 2022,” according to the FDA warning.
The FDA urges patients and health care professionals to report side effects involving duvelisib or other medicines to the FDA MedWatch program.
A version of this article first appeared on Medscape.com
Fatty liver disease drives rise in liver cancer deaths
LONDON – Around the world, nonalcoholic fatty liver disease (NAFLD) has driven an increase in deaths from liver cancer over the past decade, overtaking alcoholic liver disease, hepatitis B, and hepatitis C, according to an analysis of the Global Burden of Disease Study 2019.
A global rise in liver cancer deaths and chronic liver disease reflects changes in underlying health patterns, said Zobair Younossi, MD, MPH, professor and chair, department of medicine, Inova Fairfax Medical Campus, Falls Church, Va., who presented the analysis at the International Liver Congress (ILC) 2022.
“NAFLD and NASH [nonalcoholic steatohepatitis] are rapidly becoming the main causes of cirrhosis and liver cancer in the world,” Dr. Younossi told this news organization. “We have known about the increasing prevalence for some time, but now the outcomes in terms of mortality are catching up,” he said.
“The bottom line of this study is that the burden of this disease [NAFLD] is going up, and it will be the most important disease of the next decade or so,” he said, adding that “the largest annual percentage increase in rates of mortality from liver cancer or chronic liver disease cirrhosis is related to NAFLD.”
Specifically, during the decade of 2009-2019, the annual percent change (APC) of +1.33% in the global liver cancer death rate was driven by the fact that the APC for NAFLD was +2.47%. By comparison, the APC for alcoholic liver disease was +1.91%; for hepatitis B, the APC was +0.21%; and for hepatitis C, the APC was +1.12%.
Aleksander Krag, MD, PhD, professor and senior consultant of hepatology and director of Odense Liver Research Centre at SDU and Odense University Hospital, Denmark, who chaired the session in which this presentation was a part, acknowledged the importance of recognizing the contribution of NAFLD to liver cancer mortality.
“Liver diseases are on the rise. They are the fastest rising cause of death in the United Kingdom, faster than heart disease and other cancers. NAFLD in particular is the fastest growing cause of liver cancer, and the leading cause in France and the United States,” he remarked.
Dr. Krag also highlighted the costs of disease management.
“Managing fatty liver disease in Europe is estimated at €35 billion in direct health care, so we need to do something now,” he stressed.
“The global burden of NAFLD is so high that we need both prevention and treatment tools,” Dr. Krag said. “Change to lifestyle is a ‘no-brainer’ and costs governments very little. For the sake of our young people, we need to take this very seriously. At a political level, we can easily implement this, for example, by banning junk food advertisements, but also educating young people and their families. Good drugs will also help.”
NAFLD: The liver manifestation of type 2 diabetes
About 25%-30% of the global population have NAFLD, and 3%-5% have NASH. Dr. Younossi highlighted that the U.S. transplant database shows that NAFLD was the second indication for all liver transplants in the country. NAFLD also was a leading cause of liver transplants for patients with hepatocellular carcinoma.
There are around two billion cases of chronic liver disease globally, he said. He noted that over time, there has been an increase in all kinds of liver diseases, as reflected in the annual percent change.
“The global epidemic of obesity and type 2 diabetes is driving the rise in NAFLD, but even among lean people, the prevalence of NAFLD is around 9%,” Dr. Younossi said. “Alongside the eye and kidney complications of diabetes, this is the liver manifestation of type 2 diabetes.”
To assess global liver disease and death, Dr. Younossi and his colleagues turned to the Global Burden of Disease Study, which gathered data from around 7,000 investigators located across 22 different regions of the world, comprising 156 countries.
They calculated the incidence, prevalence, mortality, and disability-adjusted life-years (DALYs) in relation to liver cancer and chronic liver disease, including the APC. They linked the data to changes in four liver diseases: NAFLD, alcoholic liver disease, hepatitis B infection, and hepatitis C infection.
The cases of NAFLD reported in the study had been diagnosed by ultrasound or other imaging. Importantly, the prevalence of NAFLD was adjusted for alcohol use in the various national populations, explained Dr. Younossi.
In 2019, they reported that globally, the overall prevalence of liver disease reached 1.69 billion (liver cancer, 0.04%; chronic liver disease, 99.96%), with an incidence of 2.59 million (liver cancer, 20.7%; chronic liver disease, 79.3%), mortality of 1.95 million (liver cancer, 24.8%; chronic liver disease, 75.3%), and DALYs of 58.7 million (liver cancer, 21.3%; chronic liver disease, 78.7%).
Between 2009 and 2019, deaths from liver cancer rose by 27.2%, and deaths from chronic liver disease rose by 10.6%. DALYs from liver cancer rose by 21.9%, and DALYs from chronic liver disease were up by 5.1%.
In contrast to the increase in liver cancer deaths, deaths from chronic liver disease decreased (APC, –0.18%). The decrease was driven by a decrease in hepatitis B (APC, –1.83%). APCs for hepatitis C (+0.37%), alcoholic liver disease (+0.45%), and NAFLD (+1.33%) increased.
“The burden of hepatitis B–related mortality has decreased because we have been so good at vaccinating people,” Dr. Younossi remarked.
NAFLD ‘exploding’ in Middle East, North Africa, and East Asia
The increase in NAFLD has been seen in all regions of the world, but a breakdown by region shows that NAFLD is primarily “exploding” with highest prevalence and mortality in the Middle East (mostly Egypt, Iran, and Turkey), North Africa, and East Asia, said Dr. Younossi. In addition, there are large increases in the West and South America.
“We knew that the prevalence was high in the Middle East, but we now know that mortality is also high, so we are connecting these data,” said Dr. Younossi.
Awareness lacking
Dr. Younossi pressed the fact that awareness among the general population, primary care providers, and policymakers is very low. “From my perspective, raising awareness of NAFLD is the number one priority, and that is the value of this study.”
He added that more people will become aware as testing becomes more manageable.
“There are some noninvasive tests being developed, so in the future, we won’t have to do liver biopsies to diagnose these patients,” he said. “Currently, there are some excellent treatments being developed.”
“The WHO [World Health Organization] does not mention NAFLD as an important noncommunicable disease, and this too has to change,” Dr. Younossi added.
Dr. Younossi has received research funds and/or has consulted for Abbott, Allergan, Bristol-Myers Squibb, Echosens, Genfit, Gilead Sciences, Intercept, Madrigal, Merck, and Novo Nordisk. Dr. Krag has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LONDON – Around the world, nonalcoholic fatty liver disease (NAFLD) has driven an increase in deaths from liver cancer over the past decade, overtaking alcoholic liver disease, hepatitis B, and hepatitis C, according to an analysis of the Global Burden of Disease Study 2019.
A global rise in liver cancer deaths and chronic liver disease reflects changes in underlying health patterns, said Zobair Younossi, MD, MPH, professor and chair, department of medicine, Inova Fairfax Medical Campus, Falls Church, Va., who presented the analysis at the International Liver Congress (ILC) 2022.
“NAFLD and NASH [nonalcoholic steatohepatitis] are rapidly becoming the main causes of cirrhosis and liver cancer in the world,” Dr. Younossi told this news organization. “We have known about the increasing prevalence for some time, but now the outcomes in terms of mortality are catching up,” he said.
“The bottom line of this study is that the burden of this disease [NAFLD] is going up, and it will be the most important disease of the next decade or so,” he said, adding that “the largest annual percentage increase in rates of mortality from liver cancer or chronic liver disease cirrhosis is related to NAFLD.”
Specifically, during the decade of 2009-2019, the annual percent change (APC) of +1.33% in the global liver cancer death rate was driven by the fact that the APC for NAFLD was +2.47%. By comparison, the APC for alcoholic liver disease was +1.91%; for hepatitis B, the APC was +0.21%; and for hepatitis C, the APC was +1.12%.
Aleksander Krag, MD, PhD, professor and senior consultant of hepatology and director of Odense Liver Research Centre at SDU and Odense University Hospital, Denmark, who chaired the session in which this presentation was a part, acknowledged the importance of recognizing the contribution of NAFLD to liver cancer mortality.
“Liver diseases are on the rise. They are the fastest rising cause of death in the United Kingdom, faster than heart disease and other cancers. NAFLD in particular is the fastest growing cause of liver cancer, and the leading cause in France and the United States,” he remarked.
Dr. Krag also highlighted the costs of disease management.
“Managing fatty liver disease in Europe is estimated at €35 billion in direct health care, so we need to do something now,” he stressed.
“The global burden of NAFLD is so high that we need both prevention and treatment tools,” Dr. Krag said. “Change to lifestyle is a ‘no-brainer’ and costs governments very little. For the sake of our young people, we need to take this very seriously. At a political level, we can easily implement this, for example, by banning junk food advertisements, but also educating young people and their families. Good drugs will also help.”
NAFLD: The liver manifestation of type 2 diabetes
About 25%-30% of the global population have NAFLD, and 3%-5% have NASH. Dr. Younossi highlighted that the U.S. transplant database shows that NAFLD was the second indication for all liver transplants in the country. NAFLD also was a leading cause of liver transplants for patients with hepatocellular carcinoma.
There are around two billion cases of chronic liver disease globally, he said. He noted that over time, there has been an increase in all kinds of liver diseases, as reflected in the annual percent change.
“The global epidemic of obesity and type 2 diabetes is driving the rise in NAFLD, but even among lean people, the prevalence of NAFLD is around 9%,” Dr. Younossi said. “Alongside the eye and kidney complications of diabetes, this is the liver manifestation of type 2 diabetes.”
To assess global liver disease and death, Dr. Younossi and his colleagues turned to the Global Burden of Disease Study, which gathered data from around 7,000 investigators located across 22 different regions of the world, comprising 156 countries.
They calculated the incidence, prevalence, mortality, and disability-adjusted life-years (DALYs) in relation to liver cancer and chronic liver disease, including the APC. They linked the data to changes in four liver diseases: NAFLD, alcoholic liver disease, hepatitis B infection, and hepatitis C infection.
The cases of NAFLD reported in the study had been diagnosed by ultrasound or other imaging. Importantly, the prevalence of NAFLD was adjusted for alcohol use in the various national populations, explained Dr. Younossi.
In 2019, they reported that globally, the overall prevalence of liver disease reached 1.69 billion (liver cancer, 0.04%; chronic liver disease, 99.96%), with an incidence of 2.59 million (liver cancer, 20.7%; chronic liver disease, 79.3%), mortality of 1.95 million (liver cancer, 24.8%; chronic liver disease, 75.3%), and DALYs of 58.7 million (liver cancer, 21.3%; chronic liver disease, 78.7%).
Between 2009 and 2019, deaths from liver cancer rose by 27.2%, and deaths from chronic liver disease rose by 10.6%. DALYs from liver cancer rose by 21.9%, and DALYs from chronic liver disease were up by 5.1%.
In contrast to the increase in liver cancer deaths, deaths from chronic liver disease decreased (APC, –0.18%). The decrease was driven by a decrease in hepatitis B (APC, –1.83%). APCs for hepatitis C (+0.37%), alcoholic liver disease (+0.45%), and NAFLD (+1.33%) increased.
“The burden of hepatitis B–related mortality has decreased because we have been so good at vaccinating people,” Dr. Younossi remarked.
NAFLD ‘exploding’ in Middle East, North Africa, and East Asia
The increase in NAFLD has been seen in all regions of the world, but a breakdown by region shows that NAFLD is primarily “exploding” with highest prevalence and mortality in the Middle East (mostly Egypt, Iran, and Turkey), North Africa, and East Asia, said Dr. Younossi. In addition, there are large increases in the West and South America.
“We knew that the prevalence was high in the Middle East, but we now know that mortality is also high, so we are connecting these data,” said Dr. Younossi.
Awareness lacking
Dr. Younossi pressed the fact that awareness among the general population, primary care providers, and policymakers is very low. “From my perspective, raising awareness of NAFLD is the number one priority, and that is the value of this study.”
He added that more people will become aware as testing becomes more manageable.
“There are some noninvasive tests being developed, so in the future, we won’t have to do liver biopsies to diagnose these patients,” he said. “Currently, there are some excellent treatments being developed.”
“The WHO [World Health Organization] does not mention NAFLD as an important noncommunicable disease, and this too has to change,” Dr. Younossi added.
Dr. Younossi has received research funds and/or has consulted for Abbott, Allergan, Bristol-Myers Squibb, Echosens, Genfit, Gilead Sciences, Intercept, Madrigal, Merck, and Novo Nordisk. Dr. Krag has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LONDON – Around the world, nonalcoholic fatty liver disease (NAFLD) has driven an increase in deaths from liver cancer over the past decade, overtaking alcoholic liver disease, hepatitis B, and hepatitis C, according to an analysis of the Global Burden of Disease Study 2019.
A global rise in liver cancer deaths and chronic liver disease reflects changes in underlying health patterns, said Zobair Younossi, MD, MPH, professor and chair, department of medicine, Inova Fairfax Medical Campus, Falls Church, Va., who presented the analysis at the International Liver Congress (ILC) 2022.
“NAFLD and NASH [nonalcoholic steatohepatitis] are rapidly becoming the main causes of cirrhosis and liver cancer in the world,” Dr. Younossi told this news organization. “We have known about the increasing prevalence for some time, but now the outcomes in terms of mortality are catching up,” he said.
“The bottom line of this study is that the burden of this disease [NAFLD] is going up, and it will be the most important disease of the next decade or so,” he said, adding that “the largest annual percentage increase in rates of mortality from liver cancer or chronic liver disease cirrhosis is related to NAFLD.”
Specifically, during the decade of 2009-2019, the annual percent change (APC) of +1.33% in the global liver cancer death rate was driven by the fact that the APC for NAFLD was +2.47%. By comparison, the APC for alcoholic liver disease was +1.91%; for hepatitis B, the APC was +0.21%; and for hepatitis C, the APC was +1.12%.
Aleksander Krag, MD, PhD, professor and senior consultant of hepatology and director of Odense Liver Research Centre at SDU and Odense University Hospital, Denmark, who chaired the session in which this presentation was a part, acknowledged the importance of recognizing the contribution of NAFLD to liver cancer mortality.
“Liver diseases are on the rise. They are the fastest rising cause of death in the United Kingdom, faster than heart disease and other cancers. NAFLD in particular is the fastest growing cause of liver cancer, and the leading cause in France and the United States,” he remarked.
Dr. Krag also highlighted the costs of disease management.
“Managing fatty liver disease in Europe is estimated at €35 billion in direct health care, so we need to do something now,” he stressed.
“The global burden of NAFLD is so high that we need both prevention and treatment tools,” Dr. Krag said. “Change to lifestyle is a ‘no-brainer’ and costs governments very little. For the sake of our young people, we need to take this very seriously. At a political level, we can easily implement this, for example, by banning junk food advertisements, but also educating young people and their families. Good drugs will also help.”
NAFLD: The liver manifestation of type 2 diabetes
About 25%-30% of the global population have NAFLD, and 3%-5% have NASH. Dr. Younossi highlighted that the U.S. transplant database shows that NAFLD was the second indication for all liver transplants in the country. NAFLD also was a leading cause of liver transplants for patients with hepatocellular carcinoma.
There are around two billion cases of chronic liver disease globally, he said. He noted that over time, there has been an increase in all kinds of liver diseases, as reflected in the annual percent change.
“The global epidemic of obesity and type 2 diabetes is driving the rise in NAFLD, but even among lean people, the prevalence of NAFLD is around 9%,” Dr. Younossi said. “Alongside the eye and kidney complications of diabetes, this is the liver manifestation of type 2 diabetes.”
To assess global liver disease and death, Dr. Younossi and his colleagues turned to the Global Burden of Disease Study, which gathered data from around 7,000 investigators located across 22 different regions of the world, comprising 156 countries.
They calculated the incidence, prevalence, mortality, and disability-adjusted life-years (DALYs) in relation to liver cancer and chronic liver disease, including the APC. They linked the data to changes in four liver diseases: NAFLD, alcoholic liver disease, hepatitis B infection, and hepatitis C infection.
The cases of NAFLD reported in the study had been diagnosed by ultrasound or other imaging. Importantly, the prevalence of NAFLD was adjusted for alcohol use in the various national populations, explained Dr. Younossi.
In 2019, they reported that globally, the overall prevalence of liver disease reached 1.69 billion (liver cancer, 0.04%; chronic liver disease, 99.96%), with an incidence of 2.59 million (liver cancer, 20.7%; chronic liver disease, 79.3%), mortality of 1.95 million (liver cancer, 24.8%; chronic liver disease, 75.3%), and DALYs of 58.7 million (liver cancer, 21.3%; chronic liver disease, 78.7%).
Between 2009 and 2019, deaths from liver cancer rose by 27.2%, and deaths from chronic liver disease rose by 10.6%. DALYs from liver cancer rose by 21.9%, and DALYs from chronic liver disease were up by 5.1%.
In contrast to the increase in liver cancer deaths, deaths from chronic liver disease decreased (APC, –0.18%). The decrease was driven by a decrease in hepatitis B (APC, –1.83%). APCs for hepatitis C (+0.37%), alcoholic liver disease (+0.45%), and NAFLD (+1.33%) increased.
“The burden of hepatitis B–related mortality has decreased because we have been so good at vaccinating people,” Dr. Younossi remarked.
NAFLD ‘exploding’ in Middle East, North Africa, and East Asia
The increase in NAFLD has been seen in all regions of the world, but a breakdown by region shows that NAFLD is primarily “exploding” with highest prevalence and mortality in the Middle East (mostly Egypt, Iran, and Turkey), North Africa, and East Asia, said Dr. Younossi. In addition, there are large increases in the West and South America.
“We knew that the prevalence was high in the Middle East, but we now know that mortality is also high, so we are connecting these data,” said Dr. Younossi.
Awareness lacking
Dr. Younossi pressed the fact that awareness among the general population, primary care providers, and policymakers is very low. “From my perspective, raising awareness of NAFLD is the number one priority, and that is the value of this study.”
He added that more people will become aware as testing becomes more manageable.
“There are some noninvasive tests being developed, so in the future, we won’t have to do liver biopsies to diagnose these patients,” he said. “Currently, there are some excellent treatments being developed.”
“The WHO [World Health Organization] does not mention NAFLD as an important noncommunicable disease, and this too has to change,” Dr. Younossi added.
Dr. Younossi has received research funds and/or has consulted for Abbott, Allergan, Bristol-Myers Squibb, Echosens, Genfit, Gilead Sciences, Intercept, Madrigal, Merck, and Novo Nordisk. Dr. Krag has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT ILC 2022
Simultaneous Cases of Carfilzomib-Induced Thrombotic Microangiopathy in 2 Patients With Multiple Myeloma
As a class of drugs, proteasome inhibitors are known to rarely cause drug-induced thrombotic microangiopathy (DITMA). In particular, carfilzomib is a second-generation, irreversible proteasome inhibitor approved for the treatment of relapsed, refractory multiple myeloma (MM) in combination with other therapeutic agents.1 Although generally well tolerated, carfilzomib has been associated with serious adverse events such as cardiovascular toxicity and DITMA.2-4 Thrombotic microangiopathy (TMA) is a life-threatening disorder characterized by thrombocytopenia, microangiopathic hemolytic anemia, and end-organ damage.5 Its occurrence secondary to carfilzomib has been reported only rarely in clinical trials of MM, and the most effective management of the disorder as well as the concurrent risk factors that contribute to its development remain incompletely understood.6,7 As a result, given both the expanding use of carfilzomib in practice and the morbidity of TMA, descriptions of carfilzomib-induced TMA from the real-world setting continue to provide important contributions to our understanding of the disorder.
At our US Department of Veterans Affairs (VA) medical center, 2 patients developed severe carfilzomib-induced TMA within days of one another. The presentation of simultaneous cases was highly unexpected and offered the unique opportunity to compare clinical features in real time. Here, we describe our 2 cases in detail, review their presentations and management in the context of the prior literature, and discuss potential insights gained into the disease.
Case Presentation
Case 1
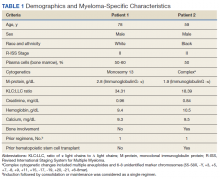
A 78-year-old male patient was diagnosed with monoclonal gammopathy of undetermined significance in 2012 that progressed to Revised International Staging System stage II IgG-κ MM in 2016 due to worsening anemia with a hemoglobin level < 10 g/dL (Table 1). He was treated initially with 8 cycles of first-line bortezomib, lenalidomide, and dexamethasone, to which he achieved a partial response with > 50% reduction in serum M-protein. He then received 3 cycles of maintenance bortezomib until relapse, at which time he was switched to second-line therapy consisting of carfilzomib 20 mg/m2 on days 1 and 2 and 56 mg/m2 on days 8, 9, 15, and 16 for cycle 1, followed by 56 mg/m2 on days 1, 2, 8, 9, 15, and 16 for subsequent cycles plus dexamethasone 20 mg twice weekly every 28 days.
After the patient received cycle 3, day 1 of carfilzomib, he developed subjective fevers, chills, and diarrhea. He missed his day 2 infusion and instead presented to the VA emergency department, where his vital signs were stable and laboratory tests were notable for the following levels: leukocytosis of20.3 K/µL (91.7% neutrophils), hemoglobin 12.4 g/dL (prior 13.5 g/dL), platelet count 171 K/µL, and creatinine 1.39 mg/dL (prior 1.13 g/dL). A chest X-ray demonstrated diffuse bilateral opacities concerning for edema vs infection, and he was started empirically on vancomycin, piperacillin-tazobactam, and azithromycin. His outpatient medications, which included acyclovir, aspirin, finasteride, oxybutynin, ranitidine, omega-3 fatty acids, fish oil, vitamin D, and senna, were continued as indicated.
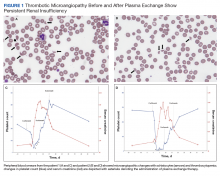
On hospital day 2, the patient’s platelet count dropped to 81 K/µL and creatinine level rose to 1.78 mg/dL. He developed dark urine (urinalysis [UA] 3+ blood, 6-11 red blood cells per high power field [RBC/HPF]) and had laboratory tests suggestive of hemolysis, including lactic dehydrogenase (LDH) > 1,200 IU/L (reference range, 60-250 IU/L), haptoglobin < 30 mg/dL (reference range, 44-215 mg/dL), total bilirubin 3.2 mg/dL (reference range, 0.2-1.3 mg/dL; indirect bilirubin, 2.6 mg/dL), and a peripheral blood smear demonstrating moderate microangiopathy (Figure 1).
Workup for alternative causes of thrombocytopenia included a negative heparin-induced thrombocytopenia panel and a disseminated intravascular coagulation (DIC) panel showing elevated fibrinogen (515 mg/dL; reference range, 200-400 mg/dL) and mildly elevated international normalized ratio (INR) (1.3). Blood cultures were negative, and a 22-pathogen gastrointestinal polymerase chain reaction (PCR) panel failed to identify viral or bacterial pathogens, including Escherichia coli O157:H7. C3 (81 mg/dL; reference range, 90-180 mg/dL) and C4 (16 mg/dL; reference range, 16-47 mg/dL) complement levels were borderline to mildly reduced.
Based on this constellation of findings, a diagnosis of TMA was made, and the patient was started empirically on plasma exchange and pulse-dosed steroids. After 4 cycles of plasma exchange, the platelet count had normalized from its nadir of 29 K/µL. ADAMTS13 activity (98% enzyme activity) ruled out thrombotic thrombocytopenic purpura (TTP), and the patient continued to have anuric renal failure (creatinine, 8.62 mg/dL) necessitating the initiation of hemodialysis. Given persistent renal insufficiency, a diagnosis of atypical hemolytic uremic syndrome (HUS) was considered, and eculizumab 900 mg was administered on days 8 and 15 with stabilization of renal function. By the time of discharge on day 18, the patient’s creatinine level had decreased to 3.89 mg/dL, and platelet count was 403 K/µL. Creatinine normalized to 1.07 mg/dL by day 46.
Outpatient genetic testing through the BloodCenter of Wisconsin Diagnostic Laboratories was negative for mutations in the following genes associated with atypical HUS: CFH, CFI, MCP (CD46), THBD, CFB, C3, DGKE, ADAMTS13, C4BPA, C4BPB, LMNA, CFHR1, CFHR3, CFHR4, and CFHR5. The patient subsequently remained off all antimyeloma therapy for > 1 year until eventually starting third-line pomalidomide plus dexamethasone without reinitiation of proteasome inhibitor therapy.
Case 2
A 59-year-old male patient, diagnosed in 2013 with ISS stage I IgG-κ MM after presenting with compression fractures, completed 8 cycles of cyclophosphamide, bortezomib, and dexamethasone before undergoing autologous hematopoietic stem cell transplantation with complete response (Table 1). He subsequently received single-agent maintenance bortezomib until relapse nearly 2 years later, at which time he started second-line carfilzomib 20 mg/m2 on days 1 and 2 and 27 mg/m2 on days 8, 9, 15, and 16 for cycle 1, followed by 27 mg/m2 on days 8, 9, 15, and 16 for cycles 2 to 8, lenalidomide 25 mg on days 1 to 21, and dexamethasone 40 mg weekly every 28 days. Serum free light chain levels normalized after 9 cycles, and he subsequently began maintenance carfilzomib 70 mg/m2 on days 1 and 15 plus lenalidomide 10 mg on days 1 to 21 every 28 days.
On the morning before admission, the patient received C6D17 of maintenance carfilzomib, which had been delayed from day 15 because of the holiday. Later that evening, he developed nausea, vomiting, and fever of 101.3 °F. He presented to the VA emergency department and was tachycardic (108 beats per minute) and hypotensive (86/55 mm Hg). Laboratory tests were notable for hemoglobin level 9.9 g/dL (prior 11.6 g/dL), platelet count 270 K/µL, and creatinine level 1.86 mg/dL (prior 1.12 mg/dL). A respiratory viral panel was positive for influenza A, and antimicrobial agents were eventually broadened to piperacillin-tazobactam, azithromycin, and oseltamivir. His outpatient medications, which included acyclovir, zoledronic acid, sulfamethoxazole/trimethoprim, aspirin, amlodipine, atorvastatin, omeprazole, zolpidem, calcium, vitamin D, loratadine, ascorbic acid, and prochlorperazine, were continued as indicated.
On hospital day 2, the patient’s platelet count declined from 211 to 57 K/µL. He developed tea-colored urine (UA 2+ blood, 0-2 RBC/HPF) and had laboratory tests suggestive of hemolysis, including LDH 910 IU/L (reference range, 60-250 IU/L), total bilirubin 3.3 mg/dL (reference range, 0.2-1.3 mg/dL; no direct or indirect available), and a peripheral blood smear demonstrating moderate microangiopathy. Although haptoglobin level was normal at this time (206 mg/dL; reference range, 44-215 mg/dL), it decreased to 42 mg/dL by the following day. Additional workup included a negative direct Coombs and a DIC panel showing elevated fibrinogen (596 mg/dL; reference range, 200-400 mg/dL) and mildly elevated INR (1.16). Blood cultures remained negative, and a 22-pathogen GI PCR panel identified no viral or bacterial pathogens, including E coli O157:H7. C3 (114 mg/dL; reference range, 90-180 mg/dL) and C4 (40 mg/dL; reference range, 16-47 mg/dL) complement levels were both normal.
Based on these findings, empiric treatment was started with plasma exchange and pulse-dosed steroids. The patient received 3 cycles of plasma exchange until the results of the ADAMTS13 activity ruled out TTP (63% enzyme activity). Over the next 6 days, his platelet count reached a nadir of 6 K/µL and creatinine level peaked at 10.36 mg/dL, necessitating the initiation of hemodialysis. Given severe renal insufficiency, a diagnosis of atypical HUS was again considered, and eculizumab 900 mg was administered on days 9 and 16 with stabilization of renal function. By the time of discharge on day 17, the patient’s creatinine level had decreased to 4.17 mg/dL and platelet count was 164 K/µL. Creatinine level normalized to 1.02 mg/dL by day 72.
Outpatient genetic testing through the BloodCenter of Wisconsin Diagnostic Laboratories was negative for gene mutations associated with atypical HUS. Approximately 1 month after discharge, the patient resumed maintenance lenalidomide alone without reinitiation of proteasome inhibitor therapy.
Discussion
In this case series, we describe the uncommon drug-related adverse event of TMA occurring in 2 patients with MM after receiving carfilzomib. Although the incidence of TMA disorders is low, reaching up to 2.8% in patients receiving carfilzomib plus cyclophosphamide and dexamethasone in the phase 2 CARDAMON trial, our experience suggests that a high index of suspicion for carfilzomib-induced TMA is warranted in the real-world setting.8 TMA syndromes, including TTP, HUS, and DITMA, are characterized by microvascular endothelial injury and thrombosis leading to thrombocytopenia and microangiopathic hemolytic anemia.5,9 Several drug culprits of DITMA are recognized, including quinine, gemcitabine, tacrolimus, and proteasome inhibitors (bortezomib, carfilzomib, ixazomib).10-12 In a real-world series of patients receiving proteasome inhibitor therapy, either carfilzomib (n=8) or bortezomib (n=3), common clinical features of DITMA included thrombocytopenia, microangiopathic hemolytic anemia, gastrointestinal symptoms, and renal insufficiency with or without a need for hemodialysis.2 Although DITMA has been described primarily as an early event, its occurrence after 12 months of proteasome inhibitor therapy has also been reported, both in this series and elsewhere, thereby suggesting an ongoing risk for DITMA throughout the duration of carfilzomib treatment.2,13
The diagnosis of DITMA can be challenging given its nonspecific symptoms that overlap with other TMA syndromes. Previous studies have proposed that for a drug to be associated with DITMA, there should be: (1) evidence of clinical and/or pathologic findings of TMA; (2) exclusion of alternative causes of TMA; (3) no other new drug exposures other than the suspected culprit medication; and (4) a lack of recurrence of TMA in absence of the drug.10 In the case of patients with MM, other causes of TMA have also been described, including the underlying plasma cell disorder itself and stem cell transplantation.14 In the 2 cases we have described, these alternative causes were considered unlikely given that only 1 patient underwent transplantation remotely and neither had a previous history of TMA secondary to their disease. With respect to other TMA syndromes, ADAMTS13 levels > 10% and negative stool studies for E coli O157:H7 suggested against TTP or typical HUS, respectively. No other drug culprits were identified, and the close timing between the receipt of carfilzomib and symptom onset supported a causal relationship.
Because specific therapies are lacking, management of DITMA has traditionally included drug discontinuation and supportive care for end-organ injury.5 The terminal complement inhibitor, eculizumab, improves hematologic abnormalities and renal function in patients with atypical HUS but its use for treating patients with DITMA is not standard.15 Therefore, the decision to administer eculizumab to our 2 patients was driven by their severe renal insufficiency without improvement after plasma exchange, which suggested a phenotype similar to atypical HUS. After administration of eculizumab, renal function stabilized and then gradually improved over weeks to months, a time course similar to that described in cases of patients with DITMA secondary to other anticancer therapies treated with eculizumab.16 Although these results suggest a potential role for eculizumab in proteasome inhibitor–induced TMA, distinguishing the benefit of eculizumab over drug discontinuation alone remains challenging, and well-designed prospective investigations are needed.
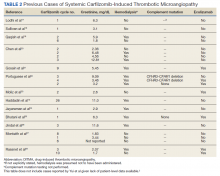
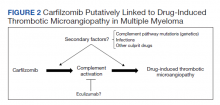
The clustered occurrence of our 2 cases is unique from previous reports that describe carfilzomib-induced TMA as a sporadic event (Table 2).13,17-28 Both immune-mediated and direct toxic effects have been proposed as mechanisms of DITMA, and while our cases do not differentiate between these mechanisms, we considered whether a combined model of initiation, whereby patient or environmental risk factors modulate occurrence of the disease in conjunction with the inciting drug, could explain the clustered occurrence of cases. In this series, drug manufacturing was not a shared risk factor as each patient received carfilzomib from different lot numbers. Furthermore, other patients at our center received carfilzomib from the same batches without developing DITMA. We also considered the role of infection given that 1 patient was diagnosed with influenza A and both presented with nonspecific, viral-like symptoms during the winter season. Interestingly, concurrent viral infections have been reported in other cases of carfilzomib-induced DITMA as well and have also been discussed as a trigger of atypical HUS.20,29 Finally, genetic testing was negative for complement pathway mutations that might predispose to complement dysregulation.
The absence of complement mutations in our 2 patients differs from a recent series describing heterozygous CFHR3-CHFR1 deletions in association with carfilzomib-induced TMA.22 In that report, the authors hypothesized that carfilzomib decreases expression of complement factor H (CFH), a negative regulator of complement activation, thereby leading to complement dysregulation in patients who are genetically predisposed. In a second series, plasma from patients with DITMA secondary to carfilzomib induced the deposition of the complement complex, C5b-9, on endothelial cells in culture, suggesting activation of the complement pathway.30 The effective use of eculizumab would also point to a role for complement activation, and ongoing investigations should aim to identify the triggers and mechanisms of complement dysregulation in this setting, especially for patients like ours in whom genetic testing for complement pathway mutations is negative (Figure 2).
Conclusions
DITMA is a known risk of proteasome inhibitors and is listed as a safety warning in the prescribing information for bortezomib, carfilzomib, and ixazomib.12 Given the overall rarity of this adverse event, the simultaneous presentation of our 2 cases was unexpected and underscores the need for heightened awareness in clinical practice. In addition, while no underlying complement mutations were identified, eculizumab was used in both cases to successfully stabilize renal function. Further research investigating the efficacy of eculizumab and the role of complement activation in proteasome inhibitor–induced TMA will be valuable.
Acknowledgments
The authors would like to thank the patients whose histories are reported in this manuscript as well as the physicians and staff who provided care during the hospitalizations and beyond. We also thank Oscar Silva, MD, PhD, for his assistance in reviewing and formatting the peripheral blood smear images.
1. McBride A, Klaus JO, Stockeri-Goldstein K. Carfilzomib: a second-generation proteasome inhibitor for the treatment of multiple myeloma. Am J Health Syst Pharm. 2015;72(5):353-360. doi:10.2146/ajhp130281
2. Yui JC, Van Keer J, Weiss BM, et al. Proteasome inhibitor associated thrombotic microangiopathy. Am J Hematol. 2016;91(9):E348-E352. doi:10.1002/ajh.24447
3. Dimopoulos MA, Roussou M, Gavriatopoulou M, et al. Cardiac and renal complications of carfilzomib in patients with multiple myeloma. Blood Adv. 2017;1(7):449-454. doi:10.1182/bloodadvances.2016003269
4. Chari A, Stewart AK, Russell SD, et al. Analysis of carfilzomib cardiovascular safety profile across relapsed and/or refractory multiple myeloma clinical trials. Blood Adv. 2018;2(13):1633-1644. doi:10.1182/bloodadvances.2017015545
5. George JN, Nester CM. Syndromes of thrombotic microangiopathy. N Engl J Med. 2014;371(7):654-666. doi:10.1056/NEJMra1312353
6. Dimopoulos MA, Moreau P, Palumbo A, et al. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016;17(1):27-38. doi:10.1016/S1470-2045(15)00464-7
7. Dimopoulos M, Quach H, Mateos MV, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396(10245):186-197. doi:10.1016/S0140-6736(20)30734-0
8. Camilleri M, Cuadrado M, Phillips E, et al. Thrombotic microangiopathy in untreated myeloma patients receiving carfilzomib, cyclophosphamide and dexamethasone on the CARDAMON study. Br J Haematol. 2021;193(4):750-760. doi:10.1111/bjh.17377
9. Masias C, Vasu S, Cataland SR. None of the above: thrombotic microangiopathy beyond TTP and HUS. Blood. 2017;129(21):2857-2863. doi:10.1182/blood-2016-11-743104
10. Al-Nouri ZL, Reese JA, Terrell DR, Vesely SK, George JN. Drug-induced thrombotic microangiopathy: a systemic review of published reports. Blood. 2015;125(4):616-618. doi:10.1182/blood-2014-11-611335
11. Saleem R, Reese JA, George JN. Drug-induced thrombotic-microangiopathy: an updated systematic review, 2014-2018. Am J Hematol. 2018;93(9):E241-E243. doi:10.1002/ajh.25208
12 Nguyen MN, Nayernama A, Jones SC, Kanapuru B, Gormley N, Waldron PE. Proteasome inhibitor-associated thrombotic microangiopathy: a review of cases reported to the FDA adverse event reporting system and published in the literature. Am J Hematol. 2020;95(9):E218-E222. doi:10.1002/ajh.25832
13. Haddadin M, Al-Sadawi M, Madanat S, et al. Late presentation of carfilzomib associated thrombotic microangiopathy. Am J Med Case Rep. 2019;7(10):240-243. doi:10.12691/ajmcr-7-10-5
14 Portuguese AJ, Gleber C, Passero Jr FC, Lipe B. A review of thrombotic microangiopathies in multiple myeloma. Leuk Res. 2019;85:106195. doi:10.1016/j.leukres.2019.106195
15. Legendre CM, Licht C, Muus P, et al. Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N Engl J Med. 2013;368(23):2169-2181. doi:10.1056/NEJMoa1208981
16. Olson SR, Lu E, Sulpizio E, Shatzel JJ, Rueda JF, DeLoughery TG. When to stop eculizumab in complement-mediated thrombotic microangiopathies. Am J Nephrol. 2018;48(2):96-107. doi:10.1159/000492033
17. Lodhi A, Kumar A, Saqlain MU, Suneja M. Thrombotic microangiopathy associated with proteasome inhibitors. Clin Kidney J. 2015;8(5):632-636. doi:10.1093/ckj/sfv059
18. Sullivan MR, Danilov AV, Lansigan F, Dunbar NM. Carfilzomib associated thrombotic microangiopathy initially treated with therapeutic plasma exchange. J Clin Apher., 2015;30(5):308-310. doi:10.1002/jca.21371
19. Qaqish I, Schlam IM, Chakkera HA, Fonseca R, Adamski J. Carfilzomib: a cause of drug associated thrombotic microangiopathy. Transfus Apher Sci. 2016;54(3):401-404. doi:10.1016/j.transci.2016.03.002
20. Chen Y, Ooi M, Lim SF, et al. Thrombotic microangiopathy during carfilzomib use: case series in Singapore. Blood Cancer J. 2016;6(7):e450. doi:10.1038/bcj.2016.62
21. Gosain R, Gill A, Fuqua J, et al. Gemcitabine and carfilzomib induced thrombotic microangiopathy: eculizumab as a life-saving treatment. Clin Case Rep. 2017;5(12):1926-1930. doi:10.1002/ccr3.1214
22. Portuguese AJ, Lipe B. Carfilzomib-induced aHUS responds to early eculizumab and may be associated with heterozygrous CFHR3-CFHR1 deletion. Blood Adv. 2018;2(23):3443-3446. doi:10.1182/bloodadvances.2018027532
23. Moliz C, Gutiérrez E, Cavero T, Redondo B, Praga M. Eculizumab as a treatment for atypical hemolytic syndrome secondary to carfilzomib. Nefrologia (Engl Ed). 2019;39(1):86-88. doi:10.1016/j.nefro.2018.02.005
24. Jeyaraman P, Borah P, Singh A, et al., Thrombotic microangiopathy after carfilzomib in a very young myeloma patient. Blood Cells Mol Dis. 2020;81:102400. doi:10.1016/j.bcmd.2019.102400
25. Bhutani D, Assal A, Mapara MY, Prinzing S, Lentzsch S. Case report: carfilzomib-induced thrombotic microangiopathy with complement activation treated successfully with eculizumab. Clin Lymphoma Myeloma Leuk. 2020;20(4):e155-e157. doi:10.1016/j.clml.2020.01.016
26. Jindal N, Jandial A, Jain A, et al. Carfilzomib-induced thrombotic microangiopathy: a case based review. Hematol Oncol Stem Cell Ther. 2020;S1658-3876(20)30118-7. doi:10.1016/j.hemonc.2020.07.001
27. Monteith BE, Venner CP, Reece DE, et al. Drug-induced thrombotic microangiopathy with concurrent proteasome inhibitor use in the treatment of multiple myeloma: a case series and review of the literature. Clin Lymphoma Myeloma Leuk. 2020;20(11):e791-e780. doi:10.1016/j.clml.2020.04.014
28. Rassner M, Baur R, Wäsch R, et al. Two cases of carfilzomib-induced thrombotic microangiopathy successfully treated with eculizumab in multiple myeloma. BMC Nephrol. 2021;22(1):32. doi:10.1186/s12882-020-02226-5
29. Kavanagh D, Goodship THJ. Atypical hemolytic uremic syndrome, genetic basis, and clinical manifestations. Hematology Am Soc Hematol Educ Program. 2011;2011:15-20. doi:10.1182/asheducation-2011.1.15
30. Blasco M, Martínez-Roca A, Rodríguez-Lobato LG, et al. Complement as the enabler of carfilzomib-induced thrombotic microangiopathy. Br J Haematol. 2021;193(1):181-187. doi:10.1111/bjh.16796
As a class of drugs, proteasome inhibitors are known to rarely cause drug-induced thrombotic microangiopathy (DITMA). In particular, carfilzomib is a second-generation, irreversible proteasome inhibitor approved for the treatment of relapsed, refractory multiple myeloma (MM) in combination with other therapeutic agents.1 Although generally well tolerated, carfilzomib has been associated with serious adverse events such as cardiovascular toxicity and DITMA.2-4 Thrombotic microangiopathy (TMA) is a life-threatening disorder characterized by thrombocytopenia, microangiopathic hemolytic anemia, and end-organ damage.5 Its occurrence secondary to carfilzomib has been reported only rarely in clinical trials of MM, and the most effective management of the disorder as well as the concurrent risk factors that contribute to its development remain incompletely understood.6,7 As a result, given both the expanding use of carfilzomib in practice and the morbidity of TMA, descriptions of carfilzomib-induced TMA from the real-world setting continue to provide important contributions to our understanding of the disorder.
At our US Department of Veterans Affairs (VA) medical center, 2 patients developed severe carfilzomib-induced TMA within days of one another. The presentation of simultaneous cases was highly unexpected and offered the unique opportunity to compare clinical features in real time. Here, we describe our 2 cases in detail, review their presentations and management in the context of the prior literature, and discuss potential insights gained into the disease.
Case Presentation
Case 1
A 78-year-old male patient was diagnosed with monoclonal gammopathy of undetermined significance in 2012 that progressed to Revised International Staging System stage II IgG-κ MM in 2016 due to worsening anemia with a hemoglobin level < 10 g/dL (Table 1). He was treated initially with 8 cycles of first-line bortezomib, lenalidomide, and dexamethasone, to which he achieved a partial response with > 50% reduction in serum M-protein. He then received 3 cycles of maintenance bortezomib until relapse, at which time he was switched to second-line therapy consisting of carfilzomib 20 mg/m2 on days 1 and 2 and 56 mg/m2 on days 8, 9, 15, and 16 for cycle 1, followed by 56 mg/m2 on days 1, 2, 8, 9, 15, and 16 for subsequent cycles plus dexamethasone 20 mg twice weekly every 28 days.
After the patient received cycle 3, day 1 of carfilzomib, he developed subjective fevers, chills, and diarrhea. He missed his day 2 infusion and instead presented to the VA emergency department, where his vital signs were stable and laboratory tests were notable for the following levels: leukocytosis of20.3 K/µL (91.7% neutrophils), hemoglobin 12.4 g/dL (prior 13.5 g/dL), platelet count 171 K/µL, and creatinine 1.39 mg/dL (prior 1.13 g/dL). A chest X-ray demonstrated diffuse bilateral opacities concerning for edema vs infection, and he was started empirically on vancomycin, piperacillin-tazobactam, and azithromycin. His outpatient medications, which included acyclovir, aspirin, finasteride, oxybutynin, ranitidine, omega-3 fatty acids, fish oil, vitamin D, and senna, were continued as indicated.
On hospital day 2, the patient’s platelet count dropped to 81 K/µL and creatinine level rose to 1.78 mg/dL. He developed dark urine (urinalysis [UA] 3+ blood, 6-11 red blood cells per high power field [RBC/HPF]) and had laboratory tests suggestive of hemolysis, including lactic dehydrogenase (LDH) > 1,200 IU/L (reference range, 60-250 IU/L), haptoglobin < 30 mg/dL (reference range, 44-215 mg/dL), total bilirubin 3.2 mg/dL (reference range, 0.2-1.3 mg/dL; indirect bilirubin, 2.6 mg/dL), and a peripheral blood smear demonstrating moderate microangiopathy (Figure 1).
Workup for alternative causes of thrombocytopenia included a negative heparin-induced thrombocytopenia panel and a disseminated intravascular coagulation (DIC) panel showing elevated fibrinogen (515 mg/dL; reference range, 200-400 mg/dL) and mildly elevated international normalized ratio (INR) (1.3). Blood cultures were negative, and a 22-pathogen gastrointestinal polymerase chain reaction (PCR) panel failed to identify viral or bacterial pathogens, including Escherichia coli O157:H7. C3 (81 mg/dL; reference range, 90-180 mg/dL) and C4 (16 mg/dL; reference range, 16-47 mg/dL) complement levels were borderline to mildly reduced.
Based on this constellation of findings, a diagnosis of TMA was made, and the patient was started empirically on plasma exchange and pulse-dosed steroids. After 4 cycles of plasma exchange, the platelet count had normalized from its nadir of 29 K/µL. ADAMTS13 activity (98% enzyme activity) ruled out thrombotic thrombocytopenic purpura (TTP), and the patient continued to have anuric renal failure (creatinine, 8.62 mg/dL) necessitating the initiation of hemodialysis. Given persistent renal insufficiency, a diagnosis of atypical hemolytic uremic syndrome (HUS) was considered, and eculizumab 900 mg was administered on days 8 and 15 with stabilization of renal function. By the time of discharge on day 18, the patient’s creatinine level had decreased to 3.89 mg/dL, and platelet count was 403 K/µL. Creatinine normalized to 1.07 mg/dL by day 46.
Outpatient genetic testing through the BloodCenter of Wisconsin Diagnostic Laboratories was negative for mutations in the following genes associated with atypical HUS: CFH, CFI, MCP (CD46), THBD, CFB, C3, DGKE, ADAMTS13, C4BPA, C4BPB, LMNA, CFHR1, CFHR3, CFHR4, and CFHR5. The patient subsequently remained off all antimyeloma therapy for > 1 year until eventually starting third-line pomalidomide plus dexamethasone without reinitiation of proteasome inhibitor therapy.
Case 2
A 59-year-old male patient, diagnosed in 2013 with ISS stage I IgG-κ MM after presenting with compression fractures, completed 8 cycles of cyclophosphamide, bortezomib, and dexamethasone before undergoing autologous hematopoietic stem cell transplantation with complete response (Table 1). He subsequently received single-agent maintenance bortezomib until relapse nearly 2 years later, at which time he started second-line carfilzomib 20 mg/m2 on days 1 and 2 and 27 mg/m2 on days 8, 9, 15, and 16 for cycle 1, followed by 27 mg/m2 on days 8, 9, 15, and 16 for cycles 2 to 8, lenalidomide 25 mg on days 1 to 21, and dexamethasone 40 mg weekly every 28 days. Serum free light chain levels normalized after 9 cycles, and he subsequently began maintenance carfilzomib 70 mg/m2 on days 1 and 15 plus lenalidomide 10 mg on days 1 to 21 every 28 days.
On the morning before admission, the patient received C6D17 of maintenance carfilzomib, which had been delayed from day 15 because of the holiday. Later that evening, he developed nausea, vomiting, and fever of 101.3 °F. He presented to the VA emergency department and was tachycardic (108 beats per minute) and hypotensive (86/55 mm Hg). Laboratory tests were notable for hemoglobin level 9.9 g/dL (prior 11.6 g/dL), platelet count 270 K/µL, and creatinine level 1.86 mg/dL (prior 1.12 mg/dL). A respiratory viral panel was positive for influenza A, and antimicrobial agents were eventually broadened to piperacillin-tazobactam, azithromycin, and oseltamivir. His outpatient medications, which included acyclovir, zoledronic acid, sulfamethoxazole/trimethoprim, aspirin, amlodipine, atorvastatin, omeprazole, zolpidem, calcium, vitamin D, loratadine, ascorbic acid, and prochlorperazine, were continued as indicated.
On hospital day 2, the patient’s platelet count declined from 211 to 57 K/µL. He developed tea-colored urine (UA 2+ blood, 0-2 RBC/HPF) and had laboratory tests suggestive of hemolysis, including LDH 910 IU/L (reference range, 60-250 IU/L), total bilirubin 3.3 mg/dL (reference range, 0.2-1.3 mg/dL; no direct or indirect available), and a peripheral blood smear demonstrating moderate microangiopathy. Although haptoglobin level was normal at this time (206 mg/dL; reference range, 44-215 mg/dL), it decreased to 42 mg/dL by the following day. Additional workup included a negative direct Coombs and a DIC panel showing elevated fibrinogen (596 mg/dL; reference range, 200-400 mg/dL) and mildly elevated INR (1.16). Blood cultures remained negative, and a 22-pathogen GI PCR panel identified no viral or bacterial pathogens, including E coli O157:H7. C3 (114 mg/dL; reference range, 90-180 mg/dL) and C4 (40 mg/dL; reference range, 16-47 mg/dL) complement levels were both normal.
Based on these findings, empiric treatment was started with plasma exchange and pulse-dosed steroids. The patient received 3 cycles of plasma exchange until the results of the ADAMTS13 activity ruled out TTP (63% enzyme activity). Over the next 6 days, his platelet count reached a nadir of 6 K/µL and creatinine level peaked at 10.36 mg/dL, necessitating the initiation of hemodialysis. Given severe renal insufficiency, a diagnosis of atypical HUS was again considered, and eculizumab 900 mg was administered on days 9 and 16 with stabilization of renal function. By the time of discharge on day 17, the patient’s creatinine level had decreased to 4.17 mg/dL and platelet count was 164 K/µL. Creatinine level normalized to 1.02 mg/dL by day 72.
Outpatient genetic testing through the BloodCenter of Wisconsin Diagnostic Laboratories was negative for gene mutations associated with atypical HUS. Approximately 1 month after discharge, the patient resumed maintenance lenalidomide alone without reinitiation of proteasome inhibitor therapy.
Discussion
In this case series, we describe the uncommon drug-related adverse event of TMA occurring in 2 patients with MM after receiving carfilzomib. Although the incidence of TMA disorders is low, reaching up to 2.8% in patients receiving carfilzomib plus cyclophosphamide and dexamethasone in the phase 2 CARDAMON trial, our experience suggests that a high index of suspicion for carfilzomib-induced TMA is warranted in the real-world setting.8 TMA syndromes, including TTP, HUS, and DITMA, are characterized by microvascular endothelial injury and thrombosis leading to thrombocytopenia and microangiopathic hemolytic anemia.5,9 Several drug culprits of DITMA are recognized, including quinine, gemcitabine, tacrolimus, and proteasome inhibitors (bortezomib, carfilzomib, ixazomib).10-12 In a real-world series of patients receiving proteasome inhibitor therapy, either carfilzomib (n=8) or bortezomib (n=3), common clinical features of DITMA included thrombocytopenia, microangiopathic hemolytic anemia, gastrointestinal symptoms, and renal insufficiency with or without a need for hemodialysis.2 Although DITMA has been described primarily as an early event, its occurrence after 12 months of proteasome inhibitor therapy has also been reported, both in this series and elsewhere, thereby suggesting an ongoing risk for DITMA throughout the duration of carfilzomib treatment.2,13
The diagnosis of DITMA can be challenging given its nonspecific symptoms that overlap with other TMA syndromes. Previous studies have proposed that for a drug to be associated with DITMA, there should be: (1) evidence of clinical and/or pathologic findings of TMA; (2) exclusion of alternative causes of TMA; (3) no other new drug exposures other than the suspected culprit medication; and (4) a lack of recurrence of TMA in absence of the drug.10 In the case of patients with MM, other causes of TMA have also been described, including the underlying plasma cell disorder itself and stem cell transplantation.14 In the 2 cases we have described, these alternative causes were considered unlikely given that only 1 patient underwent transplantation remotely and neither had a previous history of TMA secondary to their disease. With respect to other TMA syndromes, ADAMTS13 levels > 10% and negative stool studies for E coli O157:H7 suggested against TTP or typical HUS, respectively. No other drug culprits were identified, and the close timing between the receipt of carfilzomib and symptom onset supported a causal relationship.
Because specific therapies are lacking, management of DITMA has traditionally included drug discontinuation and supportive care for end-organ injury.5 The terminal complement inhibitor, eculizumab, improves hematologic abnormalities and renal function in patients with atypical HUS but its use for treating patients with DITMA is not standard.15 Therefore, the decision to administer eculizumab to our 2 patients was driven by their severe renal insufficiency without improvement after plasma exchange, which suggested a phenotype similar to atypical HUS. After administration of eculizumab, renal function stabilized and then gradually improved over weeks to months, a time course similar to that described in cases of patients with DITMA secondary to other anticancer therapies treated with eculizumab.16 Although these results suggest a potential role for eculizumab in proteasome inhibitor–induced TMA, distinguishing the benefit of eculizumab over drug discontinuation alone remains challenging, and well-designed prospective investigations are needed.
The clustered occurrence of our 2 cases is unique from previous reports that describe carfilzomib-induced TMA as a sporadic event (Table 2).13,17-28 Both immune-mediated and direct toxic effects have been proposed as mechanisms of DITMA, and while our cases do not differentiate between these mechanisms, we considered whether a combined model of initiation, whereby patient or environmental risk factors modulate occurrence of the disease in conjunction with the inciting drug, could explain the clustered occurrence of cases. In this series, drug manufacturing was not a shared risk factor as each patient received carfilzomib from different lot numbers. Furthermore, other patients at our center received carfilzomib from the same batches without developing DITMA. We also considered the role of infection given that 1 patient was diagnosed with influenza A and both presented with nonspecific, viral-like symptoms during the winter season. Interestingly, concurrent viral infections have been reported in other cases of carfilzomib-induced DITMA as well and have also been discussed as a trigger of atypical HUS.20,29 Finally, genetic testing was negative for complement pathway mutations that might predispose to complement dysregulation.
The absence of complement mutations in our 2 patients differs from a recent series describing heterozygous CFHR3-CHFR1 deletions in association with carfilzomib-induced TMA.22 In that report, the authors hypothesized that carfilzomib decreases expression of complement factor H (CFH), a negative regulator of complement activation, thereby leading to complement dysregulation in patients who are genetically predisposed. In a second series, plasma from patients with DITMA secondary to carfilzomib induced the deposition of the complement complex, C5b-9, on endothelial cells in culture, suggesting activation of the complement pathway.30 The effective use of eculizumab would also point to a role for complement activation, and ongoing investigations should aim to identify the triggers and mechanisms of complement dysregulation in this setting, especially for patients like ours in whom genetic testing for complement pathway mutations is negative (Figure 2).
Conclusions
DITMA is a known risk of proteasome inhibitors and is listed as a safety warning in the prescribing information for bortezomib, carfilzomib, and ixazomib.12 Given the overall rarity of this adverse event, the simultaneous presentation of our 2 cases was unexpected and underscores the need for heightened awareness in clinical practice. In addition, while no underlying complement mutations were identified, eculizumab was used in both cases to successfully stabilize renal function. Further research investigating the efficacy of eculizumab and the role of complement activation in proteasome inhibitor–induced TMA will be valuable.
Acknowledgments
The authors would like to thank the patients whose histories are reported in this manuscript as well as the physicians and staff who provided care during the hospitalizations and beyond. We also thank Oscar Silva, MD, PhD, for his assistance in reviewing and formatting the peripheral blood smear images.
As a class of drugs, proteasome inhibitors are known to rarely cause drug-induced thrombotic microangiopathy (DITMA). In particular, carfilzomib is a second-generation, irreversible proteasome inhibitor approved for the treatment of relapsed, refractory multiple myeloma (MM) in combination with other therapeutic agents.1 Although generally well tolerated, carfilzomib has been associated with serious adverse events such as cardiovascular toxicity and DITMA.2-4 Thrombotic microangiopathy (TMA) is a life-threatening disorder characterized by thrombocytopenia, microangiopathic hemolytic anemia, and end-organ damage.5 Its occurrence secondary to carfilzomib has been reported only rarely in clinical trials of MM, and the most effective management of the disorder as well as the concurrent risk factors that contribute to its development remain incompletely understood.6,7 As a result, given both the expanding use of carfilzomib in practice and the morbidity of TMA, descriptions of carfilzomib-induced TMA from the real-world setting continue to provide important contributions to our understanding of the disorder.
At our US Department of Veterans Affairs (VA) medical center, 2 patients developed severe carfilzomib-induced TMA within days of one another. The presentation of simultaneous cases was highly unexpected and offered the unique opportunity to compare clinical features in real time. Here, we describe our 2 cases in detail, review their presentations and management in the context of the prior literature, and discuss potential insights gained into the disease.
Case Presentation
Case 1
A 78-year-old male patient was diagnosed with monoclonal gammopathy of undetermined significance in 2012 that progressed to Revised International Staging System stage II IgG-κ MM in 2016 due to worsening anemia with a hemoglobin level < 10 g/dL (Table 1). He was treated initially with 8 cycles of first-line bortezomib, lenalidomide, and dexamethasone, to which he achieved a partial response with > 50% reduction in serum M-protein. He then received 3 cycles of maintenance bortezomib until relapse, at which time he was switched to second-line therapy consisting of carfilzomib 20 mg/m2 on days 1 and 2 and 56 mg/m2 on days 8, 9, 15, and 16 for cycle 1, followed by 56 mg/m2 on days 1, 2, 8, 9, 15, and 16 for subsequent cycles plus dexamethasone 20 mg twice weekly every 28 days.
After the patient received cycle 3, day 1 of carfilzomib, he developed subjective fevers, chills, and diarrhea. He missed his day 2 infusion and instead presented to the VA emergency department, where his vital signs were stable and laboratory tests were notable for the following levels: leukocytosis of20.3 K/µL (91.7% neutrophils), hemoglobin 12.4 g/dL (prior 13.5 g/dL), platelet count 171 K/µL, and creatinine 1.39 mg/dL (prior 1.13 g/dL). A chest X-ray demonstrated diffuse bilateral opacities concerning for edema vs infection, and he was started empirically on vancomycin, piperacillin-tazobactam, and azithromycin. His outpatient medications, which included acyclovir, aspirin, finasteride, oxybutynin, ranitidine, omega-3 fatty acids, fish oil, vitamin D, and senna, were continued as indicated.
On hospital day 2, the patient’s platelet count dropped to 81 K/µL and creatinine level rose to 1.78 mg/dL. He developed dark urine (urinalysis [UA] 3+ blood, 6-11 red blood cells per high power field [RBC/HPF]) and had laboratory tests suggestive of hemolysis, including lactic dehydrogenase (LDH) > 1,200 IU/L (reference range, 60-250 IU/L), haptoglobin < 30 mg/dL (reference range, 44-215 mg/dL), total bilirubin 3.2 mg/dL (reference range, 0.2-1.3 mg/dL; indirect bilirubin, 2.6 mg/dL), and a peripheral blood smear demonstrating moderate microangiopathy (Figure 1).
Workup for alternative causes of thrombocytopenia included a negative heparin-induced thrombocytopenia panel and a disseminated intravascular coagulation (DIC) panel showing elevated fibrinogen (515 mg/dL; reference range, 200-400 mg/dL) and mildly elevated international normalized ratio (INR) (1.3). Blood cultures were negative, and a 22-pathogen gastrointestinal polymerase chain reaction (PCR) panel failed to identify viral or bacterial pathogens, including Escherichia coli O157:H7. C3 (81 mg/dL; reference range, 90-180 mg/dL) and C4 (16 mg/dL; reference range, 16-47 mg/dL) complement levels were borderline to mildly reduced.
Based on this constellation of findings, a diagnosis of TMA was made, and the patient was started empirically on plasma exchange and pulse-dosed steroids. After 4 cycles of plasma exchange, the platelet count had normalized from its nadir of 29 K/µL. ADAMTS13 activity (98% enzyme activity) ruled out thrombotic thrombocytopenic purpura (TTP), and the patient continued to have anuric renal failure (creatinine, 8.62 mg/dL) necessitating the initiation of hemodialysis. Given persistent renal insufficiency, a diagnosis of atypical hemolytic uremic syndrome (HUS) was considered, and eculizumab 900 mg was administered on days 8 and 15 with stabilization of renal function. By the time of discharge on day 18, the patient’s creatinine level had decreased to 3.89 mg/dL, and platelet count was 403 K/µL. Creatinine normalized to 1.07 mg/dL by day 46.
Outpatient genetic testing through the BloodCenter of Wisconsin Diagnostic Laboratories was negative for mutations in the following genes associated with atypical HUS: CFH, CFI, MCP (CD46), THBD, CFB, C3, DGKE, ADAMTS13, C4BPA, C4BPB, LMNA, CFHR1, CFHR3, CFHR4, and CFHR5. The patient subsequently remained off all antimyeloma therapy for > 1 year until eventually starting third-line pomalidomide plus dexamethasone without reinitiation of proteasome inhibitor therapy.
Case 2
A 59-year-old male patient, diagnosed in 2013 with ISS stage I IgG-κ MM after presenting with compression fractures, completed 8 cycles of cyclophosphamide, bortezomib, and dexamethasone before undergoing autologous hematopoietic stem cell transplantation with complete response (Table 1). He subsequently received single-agent maintenance bortezomib until relapse nearly 2 years later, at which time he started second-line carfilzomib 20 mg/m2 on days 1 and 2 and 27 mg/m2 on days 8, 9, 15, and 16 for cycle 1, followed by 27 mg/m2 on days 8, 9, 15, and 16 for cycles 2 to 8, lenalidomide 25 mg on days 1 to 21, and dexamethasone 40 mg weekly every 28 days. Serum free light chain levels normalized after 9 cycles, and he subsequently began maintenance carfilzomib 70 mg/m2 on days 1 and 15 plus lenalidomide 10 mg on days 1 to 21 every 28 days.
On the morning before admission, the patient received C6D17 of maintenance carfilzomib, which had been delayed from day 15 because of the holiday. Later that evening, he developed nausea, vomiting, and fever of 101.3 °F. He presented to the VA emergency department and was tachycardic (108 beats per minute) and hypotensive (86/55 mm Hg). Laboratory tests were notable for hemoglobin level 9.9 g/dL (prior 11.6 g/dL), platelet count 270 K/µL, and creatinine level 1.86 mg/dL (prior 1.12 mg/dL). A respiratory viral panel was positive for influenza A, and antimicrobial agents were eventually broadened to piperacillin-tazobactam, azithromycin, and oseltamivir. His outpatient medications, which included acyclovir, zoledronic acid, sulfamethoxazole/trimethoprim, aspirin, amlodipine, atorvastatin, omeprazole, zolpidem, calcium, vitamin D, loratadine, ascorbic acid, and prochlorperazine, were continued as indicated.
On hospital day 2, the patient’s platelet count declined from 211 to 57 K/µL. He developed tea-colored urine (UA 2+ blood, 0-2 RBC/HPF) and had laboratory tests suggestive of hemolysis, including LDH 910 IU/L (reference range, 60-250 IU/L), total bilirubin 3.3 mg/dL (reference range, 0.2-1.3 mg/dL; no direct or indirect available), and a peripheral blood smear demonstrating moderate microangiopathy. Although haptoglobin level was normal at this time (206 mg/dL; reference range, 44-215 mg/dL), it decreased to 42 mg/dL by the following day. Additional workup included a negative direct Coombs and a DIC panel showing elevated fibrinogen (596 mg/dL; reference range, 200-400 mg/dL) and mildly elevated INR (1.16). Blood cultures remained negative, and a 22-pathogen GI PCR panel identified no viral or bacterial pathogens, including E coli O157:H7. C3 (114 mg/dL; reference range, 90-180 mg/dL) and C4 (40 mg/dL; reference range, 16-47 mg/dL) complement levels were both normal.
Based on these findings, empiric treatment was started with plasma exchange and pulse-dosed steroids. The patient received 3 cycles of plasma exchange until the results of the ADAMTS13 activity ruled out TTP (63% enzyme activity). Over the next 6 days, his platelet count reached a nadir of 6 K/µL and creatinine level peaked at 10.36 mg/dL, necessitating the initiation of hemodialysis. Given severe renal insufficiency, a diagnosis of atypical HUS was again considered, and eculizumab 900 mg was administered on days 9 and 16 with stabilization of renal function. By the time of discharge on day 17, the patient’s creatinine level had decreased to 4.17 mg/dL and platelet count was 164 K/µL. Creatinine level normalized to 1.02 mg/dL by day 72.
Outpatient genetic testing through the BloodCenter of Wisconsin Diagnostic Laboratories was negative for gene mutations associated with atypical HUS. Approximately 1 month after discharge, the patient resumed maintenance lenalidomide alone without reinitiation of proteasome inhibitor therapy.
Discussion
In this case series, we describe the uncommon drug-related adverse event of TMA occurring in 2 patients with MM after receiving carfilzomib. Although the incidence of TMA disorders is low, reaching up to 2.8% in patients receiving carfilzomib plus cyclophosphamide and dexamethasone in the phase 2 CARDAMON trial, our experience suggests that a high index of suspicion for carfilzomib-induced TMA is warranted in the real-world setting.8 TMA syndromes, including TTP, HUS, and DITMA, are characterized by microvascular endothelial injury and thrombosis leading to thrombocytopenia and microangiopathic hemolytic anemia.5,9 Several drug culprits of DITMA are recognized, including quinine, gemcitabine, tacrolimus, and proteasome inhibitors (bortezomib, carfilzomib, ixazomib).10-12 In a real-world series of patients receiving proteasome inhibitor therapy, either carfilzomib (n=8) or bortezomib (n=3), common clinical features of DITMA included thrombocytopenia, microangiopathic hemolytic anemia, gastrointestinal symptoms, and renal insufficiency with or without a need for hemodialysis.2 Although DITMA has been described primarily as an early event, its occurrence after 12 months of proteasome inhibitor therapy has also been reported, both in this series and elsewhere, thereby suggesting an ongoing risk for DITMA throughout the duration of carfilzomib treatment.2,13
The diagnosis of DITMA can be challenging given its nonspecific symptoms that overlap with other TMA syndromes. Previous studies have proposed that for a drug to be associated with DITMA, there should be: (1) evidence of clinical and/or pathologic findings of TMA; (2) exclusion of alternative causes of TMA; (3) no other new drug exposures other than the suspected culprit medication; and (4) a lack of recurrence of TMA in absence of the drug.10 In the case of patients with MM, other causes of TMA have also been described, including the underlying plasma cell disorder itself and stem cell transplantation.14 In the 2 cases we have described, these alternative causes were considered unlikely given that only 1 patient underwent transplantation remotely and neither had a previous history of TMA secondary to their disease. With respect to other TMA syndromes, ADAMTS13 levels > 10% and negative stool studies for E coli O157:H7 suggested against TTP or typical HUS, respectively. No other drug culprits were identified, and the close timing between the receipt of carfilzomib and symptom onset supported a causal relationship.
Because specific therapies are lacking, management of DITMA has traditionally included drug discontinuation and supportive care for end-organ injury.5 The terminal complement inhibitor, eculizumab, improves hematologic abnormalities and renal function in patients with atypical HUS but its use for treating patients with DITMA is not standard.15 Therefore, the decision to administer eculizumab to our 2 patients was driven by their severe renal insufficiency without improvement after plasma exchange, which suggested a phenotype similar to atypical HUS. After administration of eculizumab, renal function stabilized and then gradually improved over weeks to months, a time course similar to that described in cases of patients with DITMA secondary to other anticancer therapies treated with eculizumab.16 Although these results suggest a potential role for eculizumab in proteasome inhibitor–induced TMA, distinguishing the benefit of eculizumab over drug discontinuation alone remains challenging, and well-designed prospective investigations are needed.
The clustered occurrence of our 2 cases is unique from previous reports that describe carfilzomib-induced TMA as a sporadic event (Table 2).13,17-28 Both immune-mediated and direct toxic effects have been proposed as mechanisms of DITMA, and while our cases do not differentiate between these mechanisms, we considered whether a combined model of initiation, whereby patient or environmental risk factors modulate occurrence of the disease in conjunction with the inciting drug, could explain the clustered occurrence of cases. In this series, drug manufacturing was not a shared risk factor as each patient received carfilzomib from different lot numbers. Furthermore, other patients at our center received carfilzomib from the same batches without developing DITMA. We also considered the role of infection given that 1 patient was diagnosed with influenza A and both presented with nonspecific, viral-like symptoms during the winter season. Interestingly, concurrent viral infections have been reported in other cases of carfilzomib-induced DITMA as well and have also been discussed as a trigger of atypical HUS.20,29 Finally, genetic testing was negative for complement pathway mutations that might predispose to complement dysregulation.
The absence of complement mutations in our 2 patients differs from a recent series describing heterozygous CFHR3-CHFR1 deletions in association with carfilzomib-induced TMA.22 In that report, the authors hypothesized that carfilzomib decreases expression of complement factor H (CFH), a negative regulator of complement activation, thereby leading to complement dysregulation in patients who are genetically predisposed. In a second series, plasma from patients with DITMA secondary to carfilzomib induced the deposition of the complement complex, C5b-9, on endothelial cells in culture, suggesting activation of the complement pathway.30 The effective use of eculizumab would also point to a role for complement activation, and ongoing investigations should aim to identify the triggers and mechanisms of complement dysregulation in this setting, especially for patients like ours in whom genetic testing for complement pathway mutations is negative (Figure 2).
Conclusions
DITMA is a known risk of proteasome inhibitors and is listed as a safety warning in the prescribing information for bortezomib, carfilzomib, and ixazomib.12 Given the overall rarity of this adverse event, the simultaneous presentation of our 2 cases was unexpected and underscores the need for heightened awareness in clinical practice. In addition, while no underlying complement mutations were identified, eculizumab was used in both cases to successfully stabilize renal function. Further research investigating the efficacy of eculizumab and the role of complement activation in proteasome inhibitor–induced TMA will be valuable.
Acknowledgments
The authors would like to thank the patients whose histories are reported in this manuscript as well as the physicians and staff who provided care during the hospitalizations and beyond. We also thank Oscar Silva, MD, PhD, for his assistance in reviewing and formatting the peripheral blood smear images.
1. McBride A, Klaus JO, Stockeri-Goldstein K. Carfilzomib: a second-generation proteasome inhibitor for the treatment of multiple myeloma. Am J Health Syst Pharm. 2015;72(5):353-360. doi:10.2146/ajhp130281
2. Yui JC, Van Keer J, Weiss BM, et al. Proteasome inhibitor associated thrombotic microangiopathy. Am J Hematol. 2016;91(9):E348-E352. doi:10.1002/ajh.24447
3. Dimopoulos MA, Roussou M, Gavriatopoulou M, et al. Cardiac and renal complications of carfilzomib in patients with multiple myeloma. Blood Adv. 2017;1(7):449-454. doi:10.1182/bloodadvances.2016003269
4. Chari A, Stewart AK, Russell SD, et al. Analysis of carfilzomib cardiovascular safety profile across relapsed and/or refractory multiple myeloma clinical trials. Blood Adv. 2018;2(13):1633-1644. doi:10.1182/bloodadvances.2017015545
5. George JN, Nester CM. Syndromes of thrombotic microangiopathy. N Engl J Med. 2014;371(7):654-666. doi:10.1056/NEJMra1312353
6. Dimopoulos MA, Moreau P, Palumbo A, et al. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016;17(1):27-38. doi:10.1016/S1470-2045(15)00464-7
7. Dimopoulos M, Quach H, Mateos MV, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396(10245):186-197. doi:10.1016/S0140-6736(20)30734-0
8. Camilleri M, Cuadrado M, Phillips E, et al. Thrombotic microangiopathy in untreated myeloma patients receiving carfilzomib, cyclophosphamide and dexamethasone on the CARDAMON study. Br J Haematol. 2021;193(4):750-760. doi:10.1111/bjh.17377
9. Masias C, Vasu S, Cataland SR. None of the above: thrombotic microangiopathy beyond TTP and HUS. Blood. 2017;129(21):2857-2863. doi:10.1182/blood-2016-11-743104
10. Al-Nouri ZL, Reese JA, Terrell DR, Vesely SK, George JN. Drug-induced thrombotic microangiopathy: a systemic review of published reports. Blood. 2015;125(4):616-618. doi:10.1182/blood-2014-11-611335
11. Saleem R, Reese JA, George JN. Drug-induced thrombotic-microangiopathy: an updated systematic review, 2014-2018. Am J Hematol. 2018;93(9):E241-E243. doi:10.1002/ajh.25208
12 Nguyen MN, Nayernama A, Jones SC, Kanapuru B, Gormley N, Waldron PE. Proteasome inhibitor-associated thrombotic microangiopathy: a review of cases reported to the FDA adverse event reporting system and published in the literature. Am J Hematol. 2020;95(9):E218-E222. doi:10.1002/ajh.25832
13. Haddadin M, Al-Sadawi M, Madanat S, et al. Late presentation of carfilzomib associated thrombotic microangiopathy. Am J Med Case Rep. 2019;7(10):240-243. doi:10.12691/ajmcr-7-10-5
14 Portuguese AJ, Gleber C, Passero Jr FC, Lipe B. A review of thrombotic microangiopathies in multiple myeloma. Leuk Res. 2019;85:106195. doi:10.1016/j.leukres.2019.106195
15. Legendre CM, Licht C, Muus P, et al. Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N Engl J Med. 2013;368(23):2169-2181. doi:10.1056/NEJMoa1208981
16. Olson SR, Lu E, Sulpizio E, Shatzel JJ, Rueda JF, DeLoughery TG. When to stop eculizumab in complement-mediated thrombotic microangiopathies. Am J Nephrol. 2018;48(2):96-107. doi:10.1159/000492033
17. Lodhi A, Kumar A, Saqlain MU, Suneja M. Thrombotic microangiopathy associated with proteasome inhibitors. Clin Kidney J. 2015;8(5):632-636. doi:10.1093/ckj/sfv059
18. Sullivan MR, Danilov AV, Lansigan F, Dunbar NM. Carfilzomib associated thrombotic microangiopathy initially treated with therapeutic plasma exchange. J Clin Apher., 2015;30(5):308-310. doi:10.1002/jca.21371
19. Qaqish I, Schlam IM, Chakkera HA, Fonseca R, Adamski J. Carfilzomib: a cause of drug associated thrombotic microangiopathy. Transfus Apher Sci. 2016;54(3):401-404. doi:10.1016/j.transci.2016.03.002
20. Chen Y, Ooi M, Lim SF, et al. Thrombotic microangiopathy during carfilzomib use: case series in Singapore. Blood Cancer J. 2016;6(7):e450. doi:10.1038/bcj.2016.62
21. Gosain R, Gill A, Fuqua J, et al. Gemcitabine and carfilzomib induced thrombotic microangiopathy: eculizumab as a life-saving treatment. Clin Case Rep. 2017;5(12):1926-1930. doi:10.1002/ccr3.1214
22. Portuguese AJ, Lipe B. Carfilzomib-induced aHUS responds to early eculizumab and may be associated with heterozygrous CFHR3-CFHR1 deletion. Blood Adv. 2018;2(23):3443-3446. doi:10.1182/bloodadvances.2018027532
23. Moliz C, Gutiérrez E, Cavero T, Redondo B, Praga M. Eculizumab as a treatment for atypical hemolytic syndrome secondary to carfilzomib. Nefrologia (Engl Ed). 2019;39(1):86-88. doi:10.1016/j.nefro.2018.02.005
24. Jeyaraman P, Borah P, Singh A, et al., Thrombotic microangiopathy after carfilzomib in a very young myeloma patient. Blood Cells Mol Dis. 2020;81:102400. doi:10.1016/j.bcmd.2019.102400
25. Bhutani D, Assal A, Mapara MY, Prinzing S, Lentzsch S. Case report: carfilzomib-induced thrombotic microangiopathy with complement activation treated successfully with eculizumab. Clin Lymphoma Myeloma Leuk. 2020;20(4):e155-e157. doi:10.1016/j.clml.2020.01.016
26. Jindal N, Jandial A, Jain A, et al. Carfilzomib-induced thrombotic microangiopathy: a case based review. Hematol Oncol Stem Cell Ther. 2020;S1658-3876(20)30118-7. doi:10.1016/j.hemonc.2020.07.001
27. Monteith BE, Venner CP, Reece DE, et al. Drug-induced thrombotic microangiopathy with concurrent proteasome inhibitor use in the treatment of multiple myeloma: a case series and review of the literature. Clin Lymphoma Myeloma Leuk. 2020;20(11):e791-e780. doi:10.1016/j.clml.2020.04.014
28. Rassner M, Baur R, Wäsch R, et al. Two cases of carfilzomib-induced thrombotic microangiopathy successfully treated with eculizumab in multiple myeloma. BMC Nephrol. 2021;22(1):32. doi:10.1186/s12882-020-02226-5
29. Kavanagh D, Goodship THJ. Atypical hemolytic uremic syndrome, genetic basis, and clinical manifestations. Hematology Am Soc Hematol Educ Program. 2011;2011:15-20. doi:10.1182/asheducation-2011.1.15
30. Blasco M, Martínez-Roca A, Rodríguez-Lobato LG, et al. Complement as the enabler of carfilzomib-induced thrombotic microangiopathy. Br J Haematol. 2021;193(1):181-187. doi:10.1111/bjh.16796
1. McBride A, Klaus JO, Stockeri-Goldstein K. Carfilzomib: a second-generation proteasome inhibitor for the treatment of multiple myeloma. Am J Health Syst Pharm. 2015;72(5):353-360. doi:10.2146/ajhp130281
2. Yui JC, Van Keer J, Weiss BM, et al. Proteasome inhibitor associated thrombotic microangiopathy. Am J Hematol. 2016;91(9):E348-E352. doi:10.1002/ajh.24447
3. Dimopoulos MA, Roussou M, Gavriatopoulou M, et al. Cardiac and renal complications of carfilzomib in patients with multiple myeloma. Blood Adv. 2017;1(7):449-454. doi:10.1182/bloodadvances.2016003269
4. Chari A, Stewart AK, Russell SD, et al. Analysis of carfilzomib cardiovascular safety profile across relapsed and/or refractory multiple myeloma clinical trials. Blood Adv. 2018;2(13):1633-1644. doi:10.1182/bloodadvances.2017015545
5. George JN, Nester CM. Syndromes of thrombotic microangiopathy. N Engl J Med. 2014;371(7):654-666. doi:10.1056/NEJMra1312353
6. Dimopoulos MA, Moreau P, Palumbo A, et al. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016;17(1):27-38. doi:10.1016/S1470-2045(15)00464-7
7. Dimopoulos M, Quach H, Mateos MV, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396(10245):186-197. doi:10.1016/S0140-6736(20)30734-0
8. Camilleri M, Cuadrado M, Phillips E, et al. Thrombotic microangiopathy in untreated myeloma patients receiving carfilzomib, cyclophosphamide and dexamethasone on the CARDAMON study. Br J Haematol. 2021;193(4):750-760. doi:10.1111/bjh.17377
9. Masias C, Vasu S, Cataland SR. None of the above: thrombotic microangiopathy beyond TTP and HUS. Blood. 2017;129(21):2857-2863. doi:10.1182/blood-2016-11-743104
10. Al-Nouri ZL, Reese JA, Terrell DR, Vesely SK, George JN. Drug-induced thrombotic microangiopathy: a systemic review of published reports. Blood. 2015;125(4):616-618. doi:10.1182/blood-2014-11-611335
11. Saleem R, Reese JA, George JN. Drug-induced thrombotic-microangiopathy: an updated systematic review, 2014-2018. Am J Hematol. 2018;93(9):E241-E243. doi:10.1002/ajh.25208
12 Nguyen MN, Nayernama A, Jones SC, Kanapuru B, Gormley N, Waldron PE. Proteasome inhibitor-associated thrombotic microangiopathy: a review of cases reported to the FDA adverse event reporting system and published in the literature. Am J Hematol. 2020;95(9):E218-E222. doi:10.1002/ajh.25832
13. Haddadin M, Al-Sadawi M, Madanat S, et al. Late presentation of carfilzomib associated thrombotic microangiopathy. Am J Med Case Rep. 2019;7(10):240-243. doi:10.12691/ajmcr-7-10-5
14 Portuguese AJ, Gleber C, Passero Jr FC, Lipe B. A review of thrombotic microangiopathies in multiple myeloma. Leuk Res. 2019;85:106195. doi:10.1016/j.leukres.2019.106195
15. Legendre CM, Licht C, Muus P, et al. Terminal complement inhibitor eculizumab in atypical hemolytic-uremic syndrome. N Engl J Med. 2013;368(23):2169-2181. doi:10.1056/NEJMoa1208981
16. Olson SR, Lu E, Sulpizio E, Shatzel JJ, Rueda JF, DeLoughery TG. When to stop eculizumab in complement-mediated thrombotic microangiopathies. Am J Nephrol. 2018;48(2):96-107. doi:10.1159/000492033
17. Lodhi A, Kumar A, Saqlain MU, Suneja M. Thrombotic microangiopathy associated with proteasome inhibitors. Clin Kidney J. 2015;8(5):632-636. doi:10.1093/ckj/sfv059
18. Sullivan MR, Danilov AV, Lansigan F, Dunbar NM. Carfilzomib associated thrombotic microangiopathy initially treated with therapeutic plasma exchange. J Clin Apher., 2015;30(5):308-310. doi:10.1002/jca.21371
19. Qaqish I, Schlam IM, Chakkera HA, Fonseca R, Adamski J. Carfilzomib: a cause of drug associated thrombotic microangiopathy. Transfus Apher Sci. 2016;54(3):401-404. doi:10.1016/j.transci.2016.03.002
20. Chen Y, Ooi M, Lim SF, et al. Thrombotic microangiopathy during carfilzomib use: case series in Singapore. Blood Cancer J. 2016;6(7):e450. doi:10.1038/bcj.2016.62
21. Gosain R, Gill A, Fuqua J, et al. Gemcitabine and carfilzomib induced thrombotic microangiopathy: eculizumab as a life-saving treatment. Clin Case Rep. 2017;5(12):1926-1930. doi:10.1002/ccr3.1214
22. Portuguese AJ, Lipe B. Carfilzomib-induced aHUS responds to early eculizumab and may be associated with heterozygrous CFHR3-CFHR1 deletion. Blood Adv. 2018;2(23):3443-3446. doi:10.1182/bloodadvances.2018027532
23. Moliz C, Gutiérrez E, Cavero T, Redondo B, Praga M. Eculizumab as a treatment for atypical hemolytic syndrome secondary to carfilzomib. Nefrologia (Engl Ed). 2019;39(1):86-88. doi:10.1016/j.nefro.2018.02.005
24. Jeyaraman P, Borah P, Singh A, et al., Thrombotic microangiopathy after carfilzomib in a very young myeloma patient. Blood Cells Mol Dis. 2020;81:102400. doi:10.1016/j.bcmd.2019.102400
25. Bhutani D, Assal A, Mapara MY, Prinzing S, Lentzsch S. Case report: carfilzomib-induced thrombotic microangiopathy with complement activation treated successfully with eculizumab. Clin Lymphoma Myeloma Leuk. 2020;20(4):e155-e157. doi:10.1016/j.clml.2020.01.016
26. Jindal N, Jandial A, Jain A, et al. Carfilzomib-induced thrombotic microangiopathy: a case based review. Hematol Oncol Stem Cell Ther. 2020;S1658-3876(20)30118-7. doi:10.1016/j.hemonc.2020.07.001
27. Monteith BE, Venner CP, Reece DE, et al. Drug-induced thrombotic microangiopathy with concurrent proteasome inhibitor use in the treatment of multiple myeloma: a case series and review of the literature. Clin Lymphoma Myeloma Leuk. 2020;20(11):e791-e780. doi:10.1016/j.clml.2020.04.014
28. Rassner M, Baur R, Wäsch R, et al. Two cases of carfilzomib-induced thrombotic microangiopathy successfully treated with eculizumab in multiple myeloma. BMC Nephrol. 2021;22(1):32. doi:10.1186/s12882-020-02226-5
29. Kavanagh D, Goodship THJ. Atypical hemolytic uremic syndrome, genetic basis, and clinical manifestations. Hematology Am Soc Hematol Educ Program. 2011;2011:15-20. doi:10.1182/asheducation-2011.1.15
30. Blasco M, Martínez-Roca A, Rodríguez-Lobato LG, et al. Complement as the enabler of carfilzomib-induced thrombotic microangiopathy. Br J Haematol. 2021;193(1):181-187. doi:10.1111/bjh.16796
‘Unexpected’: Breast cancer spreads most during sleep
a discovery the investigators called “striking and unexpected.”
“This has not been shown before [and] we were surprised, indeed,” Nicola Aceto, PhD, professor of molecular oncology, Swiss Federal Institute of Technology Zürich, said in an interview.
The findings carry potential implications for the timing of biopsy and treatment of metastasis-prone cancers, the authors said.
The study was published online in Nature.
Circulating tumor cells (CTCs) are generally believed to shed constantly or following particular events such as surgery or physical activity; however, the processes that regulate tumor cell metastasis and how circadian rhythms may play into tumorigenesis remain unclear.
To better understand these processes, Dr. Aceto and colleagues collected blood samples from 30 women with breast cancer at 4:00 a.m. and 10:00 a.m. – times representing the body’s resting and active phases, respectively.
The researchers observed that more than 78% of all CTCs obtained were from samples taken during the resting phase.
This finding is astounding, Harrison Ball, a PhD candidate, and Sunitha Nagrath, PhD, with the University of Michigan, Ann Arbor, wrote in Nature News & Views .
Dr. Aceto and colleagues also found that CTCs generated at night divide more quickly and therefore have a higher potential to metastasize, compared with those generated during the day, which “are devoid of metastatic ability,” according to the authors, who obtained similar results in a series of mouse models.
The team also observed that key circadian rhythm hormones (such as melatonin, testosterone, and glucocorticoids) regulate CTC generation, and insulin promotes tumor cell proliferation in a time-dependent manner, suggesting a “need for time-controlled approaches for the characterization and treatment of breast cancer,” the authors wrote.
Practice changing?
Dr. Ball and Dr. Nagrath said the time-dependent nature of CTC dynamics could very well transform how cancer patients are assessed and treated.
“The data pointing to CTC proliferation and release during the rest phase suggest that doctors might need to become more conscious of when to administer specific treatments,” they wrote.
Both cautioned, however, that large clinical trials would be needed before any consideration of circadian rhythms is incorporated into standard practice. It’s also unclear whether these results in breast cancer hold true for other tumor types.
Mariana G. Figueiro, PhD, who was not involved in the research, agreed that, if studies confirm more metastatic spread at night, “there is an opportunity to treat patients at strategic times.”
Dr. Figueiro, of the Icahn School of Medicine at Mount Sinai, New York, also saw a potential impact on the timing of blood draws. “I think tightening up on how people do biopsies and bloodwork based on circadian time is important.”
Marleen Meyers, MD, agreed that these findings could have many clinical implications.
“The most obvious is that the time of day [that] treatment is administered may influence efficacy,” said Dr. Meyers, clinical professor of medicine at New York University Langone’s Perlmutter Cancer Center.
But, Dr. Meyers noted, the benefits of treating someone at night would need to be weighed against the downsides of interrupting a person’s normal sleep-wake cycle. “If this finding is clinically important it will be a challenge incorporating this into clinical care,” she said.
The study had no funding reported. Dr. Aceto is a cofounder and member of the board of PAGE Therapeutics, listed as an inventor in patent applications related to circulating tumor cells, a paid consultant for several companies, and a Novartis shareholder. One coauthor is a cofounder of PAGE Therapeutics. All other authors declare no competing interests. Dr. Meyers and Dr. Figueiro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a discovery the investigators called “striking and unexpected.”
“This has not been shown before [and] we were surprised, indeed,” Nicola Aceto, PhD, professor of molecular oncology, Swiss Federal Institute of Technology Zürich, said in an interview.
The findings carry potential implications for the timing of biopsy and treatment of metastasis-prone cancers, the authors said.
The study was published online in Nature.
Circulating tumor cells (CTCs) are generally believed to shed constantly or following particular events such as surgery or physical activity; however, the processes that regulate tumor cell metastasis and how circadian rhythms may play into tumorigenesis remain unclear.
To better understand these processes, Dr. Aceto and colleagues collected blood samples from 30 women with breast cancer at 4:00 a.m. and 10:00 a.m. – times representing the body’s resting and active phases, respectively.
The researchers observed that more than 78% of all CTCs obtained were from samples taken during the resting phase.
This finding is astounding, Harrison Ball, a PhD candidate, and Sunitha Nagrath, PhD, with the University of Michigan, Ann Arbor, wrote in Nature News & Views .
Dr. Aceto and colleagues also found that CTCs generated at night divide more quickly and therefore have a higher potential to metastasize, compared with those generated during the day, which “are devoid of metastatic ability,” according to the authors, who obtained similar results in a series of mouse models.
The team also observed that key circadian rhythm hormones (such as melatonin, testosterone, and glucocorticoids) regulate CTC generation, and insulin promotes tumor cell proliferation in a time-dependent manner, suggesting a “need for time-controlled approaches for the characterization and treatment of breast cancer,” the authors wrote.
Practice changing?
Dr. Ball and Dr. Nagrath said the time-dependent nature of CTC dynamics could very well transform how cancer patients are assessed and treated.
“The data pointing to CTC proliferation and release during the rest phase suggest that doctors might need to become more conscious of when to administer specific treatments,” they wrote.
Both cautioned, however, that large clinical trials would be needed before any consideration of circadian rhythms is incorporated into standard practice. It’s also unclear whether these results in breast cancer hold true for other tumor types.
Mariana G. Figueiro, PhD, who was not involved in the research, agreed that, if studies confirm more metastatic spread at night, “there is an opportunity to treat patients at strategic times.”
Dr. Figueiro, of the Icahn School of Medicine at Mount Sinai, New York, also saw a potential impact on the timing of blood draws. “I think tightening up on how people do biopsies and bloodwork based on circadian time is important.”
Marleen Meyers, MD, agreed that these findings could have many clinical implications.
“The most obvious is that the time of day [that] treatment is administered may influence efficacy,” said Dr. Meyers, clinical professor of medicine at New York University Langone’s Perlmutter Cancer Center.
But, Dr. Meyers noted, the benefits of treating someone at night would need to be weighed against the downsides of interrupting a person’s normal sleep-wake cycle. “If this finding is clinically important it will be a challenge incorporating this into clinical care,” she said.
The study had no funding reported. Dr. Aceto is a cofounder and member of the board of PAGE Therapeutics, listed as an inventor in patent applications related to circulating tumor cells, a paid consultant for several companies, and a Novartis shareholder. One coauthor is a cofounder of PAGE Therapeutics. All other authors declare no competing interests. Dr. Meyers and Dr. Figueiro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a discovery the investigators called “striking and unexpected.”
“This has not been shown before [and] we were surprised, indeed,” Nicola Aceto, PhD, professor of molecular oncology, Swiss Federal Institute of Technology Zürich, said in an interview.
The findings carry potential implications for the timing of biopsy and treatment of metastasis-prone cancers, the authors said.
The study was published online in Nature.
Circulating tumor cells (CTCs) are generally believed to shed constantly or following particular events such as surgery or physical activity; however, the processes that regulate tumor cell metastasis and how circadian rhythms may play into tumorigenesis remain unclear.
To better understand these processes, Dr. Aceto and colleagues collected blood samples from 30 women with breast cancer at 4:00 a.m. and 10:00 a.m. – times representing the body’s resting and active phases, respectively.
The researchers observed that more than 78% of all CTCs obtained were from samples taken during the resting phase.
This finding is astounding, Harrison Ball, a PhD candidate, and Sunitha Nagrath, PhD, with the University of Michigan, Ann Arbor, wrote in Nature News & Views .
Dr. Aceto and colleagues also found that CTCs generated at night divide more quickly and therefore have a higher potential to metastasize, compared with those generated during the day, which “are devoid of metastatic ability,” according to the authors, who obtained similar results in a series of mouse models.
The team also observed that key circadian rhythm hormones (such as melatonin, testosterone, and glucocorticoids) regulate CTC generation, and insulin promotes tumor cell proliferation in a time-dependent manner, suggesting a “need for time-controlled approaches for the characterization and treatment of breast cancer,” the authors wrote.
Practice changing?
Dr. Ball and Dr. Nagrath said the time-dependent nature of CTC dynamics could very well transform how cancer patients are assessed and treated.
“The data pointing to CTC proliferation and release during the rest phase suggest that doctors might need to become more conscious of when to administer specific treatments,” they wrote.
Both cautioned, however, that large clinical trials would be needed before any consideration of circadian rhythms is incorporated into standard practice. It’s also unclear whether these results in breast cancer hold true for other tumor types.
Mariana G. Figueiro, PhD, who was not involved in the research, agreed that, if studies confirm more metastatic spread at night, “there is an opportunity to treat patients at strategic times.”
Dr. Figueiro, of the Icahn School of Medicine at Mount Sinai, New York, also saw a potential impact on the timing of blood draws. “I think tightening up on how people do biopsies and bloodwork based on circadian time is important.”
Marleen Meyers, MD, agreed that these findings could have many clinical implications.
“The most obvious is that the time of day [that] treatment is administered may influence efficacy,” said Dr. Meyers, clinical professor of medicine at New York University Langone’s Perlmutter Cancer Center.
But, Dr. Meyers noted, the benefits of treating someone at night would need to be weighed against the downsides of interrupting a person’s normal sleep-wake cycle. “If this finding is clinically important it will be a challenge incorporating this into clinical care,” she said.
The study had no funding reported. Dr. Aceto is a cofounder and member of the board of PAGE Therapeutics, listed as an inventor in patent applications related to circulating tumor cells, a paid consultant for several companies, and a Novartis shareholder. One coauthor is a cofounder of PAGE Therapeutics. All other authors declare no competing interests. Dr. Meyers and Dr. Figueiro reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE
Good chemo vs. bad chemo: When too much is a bad thing
A new study finds that mortality is significantly higher among patients with advanced solid tumors who are admitted to the hospital for chemotherapy treatment.
The findings – released in a poster session at the annual meeting of the American Society of Clinical Oncology – found that patients with solid tumors were more likely to be treated for nonurgent indications, not be referred to palliative care, and die within 60 days, compared with patients with hematologic malignancies.
Decisions about inpatient chemotherapy should not be uniform and instead should be based on a case-by-case basis, said Natalie Berger, MD, a hematologist-oncologist at Mount Sinai Hospital,, New York, and the study’s lead author.
Inpatient chemotherapy can be appropriate in certain situations, such as when chemotherapy must be given in the hospital and when it must be administered quickly after a patient presents with cancer symptoms and needs relief, she said.
However, “sometimes patients are admitted due to infection, side effects of chemotherapy or cancer, or for reasons unrelated to their cancer, and chemotherapy may be administered when it is not appropriate. It is also overutilized at the end of life which can lead to more aggressive end-of-life care rather than focusing on quality of life and supportive care,” Dr. Berger said.
The study is based on a retrospective chart review of 880 patients admitted to Mount Sinai Hospital between January 2016 and December 2017 to receive chemotherapy.
They found that the type of tumor was used to determine the urgency of an in-hospital stay for chemotherapy (odds ratio, 0.42; 95% CI, 0.25-0.72; P = .001). Patients with solid tumors or older patients or patients with a functional impairment score (Karnofsky Performance Scale) of 50% were less likely to respond to chemotherapy. There was also a decrease in quality of life among these patients, but only 46% of patients with solid tumors and 15% of patients with hematologic malignancies met with a palliative care professional.
One-third (34%) of patients with solid tumors didn’t have urgent indications, 43% of patients had no response to inpatient chemotherapy, and 20% died within 60 days, compared with patients with hematologic malignancies (19%, 19%, and 9%, respectively).
“There are many reasons why this [high mortality rate in patients with solid tumors] may be happening. Solid tumor patients are more often admitted at a later stage of their cancer when they are sicker, and they were also less likely to have a response to inpatient chemotherapy. Older patients and patients with a poor performance status were also less likely to respond to chemotherapy. This indicates that these patients were sicker, and chemotherapy use may not have been appropriate and palliative care may be underutilized,” she said.
Dr. Berger and colleagues have created a standardized protocol to assess “the appropriateness” of inpatient chemotherapy, improve quality of life, and reduce chemotherapy and health care utilization at the end of life. The protocol has been implemented as a pilot program at Mount Sinai Hospital, Dr. Berger said.
“Any inpatient chemotherapy case that meets standard accepted criteria for required inpatient administration are auto-approved through the electronic survey. For cases outside of standard criteria, further information must be inputted to determine appropriateness of inpatient treatment and are then scored electronically and reviewed by committee physicians and pharmacists,” she said.
Gabriel A. Brooks, MD, MPH, an oncologist with Dartmouth Hitchcock Medical Center, Lebanon, N.H., who was not affiliated with the study, said that inpatient chemotherapy treatment is under scrutiny elsewhere as well.
“There has been recognition that patients who are otherwise sick enough to require hospital admission are often too sick to benefit from chemotherapy,” although there are exceptions. “There is certainly a movement to limit inpatient chemotherapy to situations where it is most likely to be beneficial. Some of this is driven by cost pressures. For instance, Medicare pays for inpatient hospitalizations using the DRG [diagnosis-related group] system. Hospitals cannot charge a la carte for treatments given in the hospital. Instead, they are reimbursed at a fixed rate based on the hospital diagnoses. This will often lead to poor reimbursement of high-cost cancer treatments.”
Dr. Brooks said the study offers insight into who’s getting inpatient chemotherapy. However, “what I can’t tell from this poster is how often the solid tumor patients are getting first-line chemotherapy [as] these patients may be presenting late or may have a potentially treatable cancer with a narrow closing window for treatment versus later-line chemotherapy.”
He also noted that patient and family wishes are missing from the research. “This is critical. Patients and families should be informed that inpatient chemotherapy may not provide the benefit they are hoping for, especially for patients with solid tumors starting later lines of therapy. Patients should be informed that there are alternatives to inpatient chemotherapy, such as hospice referral or waiting for possible outpatient treatment – if their condition improves. But when a patient wants to try inpatient chemotherapy and their doctor wants to offer it, then it is likely a reasonable thing to try.”
Going forward, he said, “qualitative study is needed to better understand when and why inpatient chemotherapy is used. There are likely some clear good uses and some clear bad uses of inpatient chemotherapy. Can outpatient regimens be substituted for the regimens where patients are directly admitted? Or, can outpatient protocols be devised for these regimens? Are there specific situations where inpatient chemotherapy is the right thing (leukemia, esophageal cancer with worsening dysphagia, etc.)?”
No study funding was received.
A new study finds that mortality is significantly higher among patients with advanced solid tumors who are admitted to the hospital for chemotherapy treatment.
The findings – released in a poster session at the annual meeting of the American Society of Clinical Oncology – found that patients with solid tumors were more likely to be treated for nonurgent indications, not be referred to palliative care, and die within 60 days, compared with patients with hematologic malignancies.
Decisions about inpatient chemotherapy should not be uniform and instead should be based on a case-by-case basis, said Natalie Berger, MD, a hematologist-oncologist at Mount Sinai Hospital,, New York, and the study’s lead author.
Inpatient chemotherapy can be appropriate in certain situations, such as when chemotherapy must be given in the hospital and when it must be administered quickly after a patient presents with cancer symptoms and needs relief, she said.
However, “sometimes patients are admitted due to infection, side effects of chemotherapy or cancer, or for reasons unrelated to their cancer, and chemotherapy may be administered when it is not appropriate. It is also overutilized at the end of life which can lead to more aggressive end-of-life care rather than focusing on quality of life and supportive care,” Dr. Berger said.
The study is based on a retrospective chart review of 880 patients admitted to Mount Sinai Hospital between January 2016 and December 2017 to receive chemotherapy.
They found that the type of tumor was used to determine the urgency of an in-hospital stay for chemotherapy (odds ratio, 0.42; 95% CI, 0.25-0.72; P = .001). Patients with solid tumors or older patients or patients with a functional impairment score (Karnofsky Performance Scale) of 50% were less likely to respond to chemotherapy. There was also a decrease in quality of life among these patients, but only 46% of patients with solid tumors and 15% of patients with hematologic malignancies met with a palliative care professional.
One-third (34%) of patients with solid tumors didn’t have urgent indications, 43% of patients had no response to inpatient chemotherapy, and 20% died within 60 days, compared with patients with hematologic malignancies (19%, 19%, and 9%, respectively).
“There are many reasons why this [high mortality rate in patients with solid tumors] may be happening. Solid tumor patients are more often admitted at a later stage of their cancer when they are sicker, and they were also less likely to have a response to inpatient chemotherapy. Older patients and patients with a poor performance status were also less likely to respond to chemotherapy. This indicates that these patients were sicker, and chemotherapy use may not have been appropriate and palliative care may be underutilized,” she said.
Dr. Berger and colleagues have created a standardized protocol to assess “the appropriateness” of inpatient chemotherapy, improve quality of life, and reduce chemotherapy and health care utilization at the end of life. The protocol has been implemented as a pilot program at Mount Sinai Hospital, Dr. Berger said.
“Any inpatient chemotherapy case that meets standard accepted criteria for required inpatient administration are auto-approved through the electronic survey. For cases outside of standard criteria, further information must be inputted to determine appropriateness of inpatient treatment and are then scored electronically and reviewed by committee physicians and pharmacists,” she said.
Gabriel A. Brooks, MD, MPH, an oncologist with Dartmouth Hitchcock Medical Center, Lebanon, N.H., who was not affiliated with the study, said that inpatient chemotherapy treatment is under scrutiny elsewhere as well.
“There has been recognition that patients who are otherwise sick enough to require hospital admission are often too sick to benefit from chemotherapy,” although there are exceptions. “There is certainly a movement to limit inpatient chemotherapy to situations where it is most likely to be beneficial. Some of this is driven by cost pressures. For instance, Medicare pays for inpatient hospitalizations using the DRG [diagnosis-related group] system. Hospitals cannot charge a la carte for treatments given in the hospital. Instead, they are reimbursed at a fixed rate based on the hospital diagnoses. This will often lead to poor reimbursement of high-cost cancer treatments.”
Dr. Brooks said the study offers insight into who’s getting inpatient chemotherapy. However, “what I can’t tell from this poster is how often the solid tumor patients are getting first-line chemotherapy [as] these patients may be presenting late or may have a potentially treatable cancer with a narrow closing window for treatment versus later-line chemotherapy.”
He also noted that patient and family wishes are missing from the research. “This is critical. Patients and families should be informed that inpatient chemotherapy may not provide the benefit they are hoping for, especially for patients with solid tumors starting later lines of therapy. Patients should be informed that there are alternatives to inpatient chemotherapy, such as hospice referral or waiting for possible outpatient treatment – if their condition improves. But when a patient wants to try inpatient chemotherapy and their doctor wants to offer it, then it is likely a reasonable thing to try.”
Going forward, he said, “qualitative study is needed to better understand when and why inpatient chemotherapy is used. There are likely some clear good uses and some clear bad uses of inpatient chemotherapy. Can outpatient regimens be substituted for the regimens where patients are directly admitted? Or, can outpatient protocols be devised for these regimens? Are there specific situations where inpatient chemotherapy is the right thing (leukemia, esophageal cancer with worsening dysphagia, etc.)?”
No study funding was received.
A new study finds that mortality is significantly higher among patients with advanced solid tumors who are admitted to the hospital for chemotherapy treatment.
The findings – released in a poster session at the annual meeting of the American Society of Clinical Oncology – found that patients with solid tumors were more likely to be treated for nonurgent indications, not be referred to palliative care, and die within 60 days, compared with patients with hematologic malignancies.
Decisions about inpatient chemotherapy should not be uniform and instead should be based on a case-by-case basis, said Natalie Berger, MD, a hematologist-oncologist at Mount Sinai Hospital,, New York, and the study’s lead author.
Inpatient chemotherapy can be appropriate in certain situations, such as when chemotherapy must be given in the hospital and when it must be administered quickly after a patient presents with cancer symptoms and needs relief, she said.
However, “sometimes patients are admitted due to infection, side effects of chemotherapy or cancer, or for reasons unrelated to their cancer, and chemotherapy may be administered when it is not appropriate. It is also overutilized at the end of life which can lead to more aggressive end-of-life care rather than focusing on quality of life and supportive care,” Dr. Berger said.
The study is based on a retrospective chart review of 880 patients admitted to Mount Sinai Hospital between January 2016 and December 2017 to receive chemotherapy.
They found that the type of tumor was used to determine the urgency of an in-hospital stay for chemotherapy (odds ratio, 0.42; 95% CI, 0.25-0.72; P = .001). Patients with solid tumors or older patients or patients with a functional impairment score (Karnofsky Performance Scale) of 50% were less likely to respond to chemotherapy. There was also a decrease in quality of life among these patients, but only 46% of patients with solid tumors and 15% of patients with hematologic malignancies met with a palliative care professional.
One-third (34%) of patients with solid tumors didn’t have urgent indications, 43% of patients had no response to inpatient chemotherapy, and 20% died within 60 days, compared with patients with hematologic malignancies (19%, 19%, and 9%, respectively).
“There are many reasons why this [high mortality rate in patients with solid tumors] may be happening. Solid tumor patients are more often admitted at a later stage of their cancer when they are sicker, and they were also less likely to have a response to inpatient chemotherapy. Older patients and patients with a poor performance status were also less likely to respond to chemotherapy. This indicates that these patients were sicker, and chemotherapy use may not have been appropriate and palliative care may be underutilized,” she said.
Dr. Berger and colleagues have created a standardized protocol to assess “the appropriateness” of inpatient chemotherapy, improve quality of life, and reduce chemotherapy and health care utilization at the end of life. The protocol has been implemented as a pilot program at Mount Sinai Hospital, Dr. Berger said.
“Any inpatient chemotherapy case that meets standard accepted criteria for required inpatient administration are auto-approved through the electronic survey. For cases outside of standard criteria, further information must be inputted to determine appropriateness of inpatient treatment and are then scored electronically and reviewed by committee physicians and pharmacists,” she said.
Gabriel A. Brooks, MD, MPH, an oncologist with Dartmouth Hitchcock Medical Center, Lebanon, N.H., who was not affiliated with the study, said that inpatient chemotherapy treatment is under scrutiny elsewhere as well.
“There has been recognition that patients who are otherwise sick enough to require hospital admission are often too sick to benefit from chemotherapy,” although there are exceptions. “There is certainly a movement to limit inpatient chemotherapy to situations where it is most likely to be beneficial. Some of this is driven by cost pressures. For instance, Medicare pays for inpatient hospitalizations using the DRG [diagnosis-related group] system. Hospitals cannot charge a la carte for treatments given in the hospital. Instead, they are reimbursed at a fixed rate based on the hospital diagnoses. This will often lead to poor reimbursement of high-cost cancer treatments.”
Dr. Brooks said the study offers insight into who’s getting inpatient chemotherapy. However, “what I can’t tell from this poster is how often the solid tumor patients are getting first-line chemotherapy [as] these patients may be presenting late or may have a potentially treatable cancer with a narrow closing window for treatment versus later-line chemotherapy.”
He also noted that patient and family wishes are missing from the research. “This is critical. Patients and families should be informed that inpatient chemotherapy may not provide the benefit they are hoping for, especially for patients with solid tumors starting later lines of therapy. Patients should be informed that there are alternatives to inpatient chemotherapy, such as hospice referral or waiting for possible outpatient treatment – if their condition improves. But when a patient wants to try inpatient chemotherapy and their doctor wants to offer it, then it is likely a reasonable thing to try.”
Going forward, he said, “qualitative study is needed to better understand when and why inpatient chemotherapy is used. There are likely some clear good uses and some clear bad uses of inpatient chemotherapy. Can outpatient regimens be substituted for the regimens where patients are directly admitted? Or, can outpatient protocols be devised for these regimens? Are there specific situations where inpatient chemotherapy is the right thing (leukemia, esophageal cancer with worsening dysphagia, etc.)?”
No study funding was received.
FROM ASCO 2022
My picks for best of ASCO 2022
CHICAGO – The American Society of Clinical Oncology recently wrapped its annual meeting in Chicago. Here, I highlight some presentations that stood out to me.
A first-line treatment for metastatic colorectal cancer
The plenary session did not disappoint. In abstract LBA1, investigators presented first-line treatment for patients with metastatic colorectal cancer who were randomized to receive mFOLFOX6 with either bevacizumab or panitumumab in RAS wild-type positive patients. This was the phase 3 PARADIGM trial.
The primary outcome for this study was overall survival. It included 823 patients who were randomized 1:1 with a subset analysis of whether the primary tumor was on the left or right side of the colon. At 61 months follow-up, the median overall survival results for left-sided colon cancer was 38 months versus 34 months. It was statistically significant favoring the panitumumab arm. It improved the curable resection rate for patients with left-sided tumors from 11% in the bevacizumab arm to 18% in the panitumumab arm. Interestingly, patients randomized with right-sided tumors showed no difference in overall survival. The investigator, Takayuki Yoshino, MD, PhD, National Cancer Center Hospital East, Kashiwa, Japan, said the study findings support the use of mFOLFOX6 with panitumumab in left-sided RAS wild type as first-line therapy in metastatic colorectal patients.
A possible new standard of care in breast cancer
Shanu Modi, MD, of Memorial Sloan Kettering Cancer Center, New York, received a standing ovation and deserved it. In the phase 3 clinical trial DESTINY-Breast04 (abstract LBA3), she demonstrated that trastuzumab deruxtecan (T-DXd) for patients with metastatic breast cancer who were HER2 low (IHC 1+ or 2+ ISH-), led to a statistically significant and clinically meaningful benefit in both progression free survival and overall survival. In this trial, patients were randomized 2:1 to receive trastuzumab deruxtecan or physician’s choice of chemotherapy. All patients had at least one to two lines of chemotherapy before entering the trial. Hormone-positive patients were allowed if they had already received and failed, or progressed on hormone therapy.
Previously, most patients were treated either with eribulin with some receiving capecitabine, gemcitabine or taxane, or hormone therapy if hormone positive.
The progression-free survival was 10.1 versus 5.4 months in hormone-positive patients, and in all patients (hormone receptor positive or negative), there was a likewise improvement of 9.9 versus 5.1 months progression free survival.
Overall survival was equally impressive. In the hormone receptor–positive patients, the hazard ratio was 0.64 with a 23.9 versus 17.5 month survival. If all patients were included, the HR was again 0.64 with 23.4 versus 16.8 month survival. Even the triple-negative breast cancer patients had a HR of 0.48 with 18.2 versus 8.3 months survival. Adverse events were quite tolerable with some nausea, some decreased white count, and only an interstitial lung disease of grade 2 or less in 12%.
Trastuzumab deruxtecan is a targeted treatment which, in addition to striking its target, also targets other tumor cells that are part of the cancer. The results of this study may lead to a new standard of care of this patient population.
The study by Dr. Modi and colleagues was simultaneously published in the New England Journal of Medicine.
Improving outcomes in multiple myeloma
In abstract LBA4, Paul G. Richardson, MD, of the Dana-Farber Cancer Institute, Boston, asks if autologous stem cell transplant (ASCT) can improve outcomes after induction with an RVD regimen (lenalidomide, bortezomib, and dexamethasone) and lenalidomide (Revlimid) maintenance for newly diagnosed patients with multiple myeloma in the DETERMINATION study.
The take home here was quite interesting. In fact, there is no difference in overall survival if patients get this standard RVD/lenalidomide maintenance induction with or without ASCT. However, the progression free survival was better with ASCT: 46 versus 67 months (improvement of 21 months). However, there were some caveats. There was toxicity and change in quality of life for a while in those patients receiving ASCT as would be expected. Furthermore, the study only allowed 65 years old or younger and ASCT may not be wise for older patients. The discussant made a strong point that African Americans tend to have higher risk disease with different mutations and might also be better served by have ASCT later.
The conclusion was that, given all the new therapies in myeloma for second line and beyond, ASCT should be a discussion with each new patient and not an automatic decision.
This study was simultaneously published in the New England Journal of Medicine.
Adagrasib promising for pretreated patients with NSCLC with KRAS mutation
In patients with advanced or metastatic non–small cell lung cancer (NSCLC), adagrasib was found to be well tolerated and “demonstrates promising efficacy” for patients with the KRAS G12C mutation (KRYSTAL-1, abstract 9002). This was a phase 2 registration trial of 116 patients who were treated with 600 mg of adagrasib twice orally. Patients all had previous chemotherapy or immunotherapy or both. The overall response rate was a surprisingly good 43% (complete response and partial response). Disease control was an incredible 80% if stable disease was included. The duration of response was 8.5 months, progression-free survival was 6.5 months, and overall survival was 12.6 months. Furthermore, 33% of those with brain metastases had a complete response or partial response.
The take-home message is that, since 15% of NSCLC metastatic patients are KRAS mutant G12C, we should be watching for such patients in our biomarker analysis. While we have sotorasib – approved by the Food and Drug Administration for NSCLC – the results of this study suggests we may have another new molecule in the same class.
Neoadjuvant chemotherapy with immunotherapy for NSCLC
It may be time to consider neoadjuvant chemotherapy with immunotherapy, such as nivolumab, for patients with NSCLC in order to achieve the best response possible.
In NADIM II, investigators led by Mariano Provencio-Pulla, MD, of the Hospital Universitario Puerta de Hierro-Majadahonda, Madrid, confirmed the superiority of chemotherapy with immunotherapy for patients with resectable stage IIIA NSCLC. NADIM included patients with resectable stage IIIA/B NSCLC who were randomized 2:1 to receive carboplatin taxol neoadjuvant therapy with or without nivolumab before and after surgery. The pathological complete response rates overall were 36% versus 7%, favoring the nivolumab arm, but even higher pCR rates occurred in patients with PD-L1 over 50%.
In closing, always check MMR, KRAS, BRAF, and HER2. For wild-type left-sided mCRC, consider FOLFOX or FOLFIRI with an anti-EGFR. For KRAS mutant or right-sided colon tumor, consider FOLFOX or FOLFIRI with bevacizumab, followed by maintenance 5FU or capecitabine, with or without bevacizumab.
CHICAGO – The American Society of Clinical Oncology recently wrapped its annual meeting in Chicago. Here, I highlight some presentations that stood out to me.
A first-line treatment for metastatic colorectal cancer
The plenary session did not disappoint. In abstract LBA1, investigators presented first-line treatment for patients with metastatic colorectal cancer who were randomized to receive mFOLFOX6 with either bevacizumab or panitumumab in RAS wild-type positive patients. This was the phase 3 PARADIGM trial.
The primary outcome for this study was overall survival. It included 823 patients who were randomized 1:1 with a subset analysis of whether the primary tumor was on the left or right side of the colon. At 61 months follow-up, the median overall survival results for left-sided colon cancer was 38 months versus 34 months. It was statistically significant favoring the panitumumab arm. It improved the curable resection rate for patients with left-sided tumors from 11% in the bevacizumab arm to 18% in the panitumumab arm. Interestingly, patients randomized with right-sided tumors showed no difference in overall survival. The investigator, Takayuki Yoshino, MD, PhD, National Cancer Center Hospital East, Kashiwa, Japan, said the study findings support the use of mFOLFOX6 with panitumumab in left-sided RAS wild type as first-line therapy in metastatic colorectal patients.
A possible new standard of care in breast cancer
Shanu Modi, MD, of Memorial Sloan Kettering Cancer Center, New York, received a standing ovation and deserved it. In the phase 3 clinical trial DESTINY-Breast04 (abstract LBA3), she demonstrated that trastuzumab deruxtecan (T-DXd) for patients with metastatic breast cancer who were HER2 low (IHC 1+ or 2+ ISH-), led to a statistically significant and clinically meaningful benefit in both progression free survival and overall survival. In this trial, patients were randomized 2:1 to receive trastuzumab deruxtecan or physician’s choice of chemotherapy. All patients had at least one to two lines of chemotherapy before entering the trial. Hormone-positive patients were allowed if they had already received and failed, or progressed on hormone therapy.
Previously, most patients were treated either with eribulin with some receiving capecitabine, gemcitabine or taxane, or hormone therapy if hormone positive.
The progression-free survival was 10.1 versus 5.4 months in hormone-positive patients, and in all patients (hormone receptor positive or negative), there was a likewise improvement of 9.9 versus 5.1 months progression free survival.
Overall survival was equally impressive. In the hormone receptor–positive patients, the hazard ratio was 0.64 with a 23.9 versus 17.5 month survival. If all patients were included, the HR was again 0.64 with 23.4 versus 16.8 month survival. Even the triple-negative breast cancer patients had a HR of 0.48 with 18.2 versus 8.3 months survival. Adverse events were quite tolerable with some nausea, some decreased white count, and only an interstitial lung disease of grade 2 or less in 12%.
Trastuzumab deruxtecan is a targeted treatment which, in addition to striking its target, also targets other tumor cells that are part of the cancer. The results of this study may lead to a new standard of care of this patient population.
The study by Dr. Modi and colleagues was simultaneously published in the New England Journal of Medicine.
Improving outcomes in multiple myeloma
In abstract LBA4, Paul G. Richardson, MD, of the Dana-Farber Cancer Institute, Boston, asks if autologous stem cell transplant (ASCT) can improve outcomes after induction with an RVD regimen (lenalidomide, bortezomib, and dexamethasone) and lenalidomide (Revlimid) maintenance for newly diagnosed patients with multiple myeloma in the DETERMINATION study.
The take home here was quite interesting. In fact, there is no difference in overall survival if patients get this standard RVD/lenalidomide maintenance induction with or without ASCT. However, the progression free survival was better with ASCT: 46 versus 67 months (improvement of 21 months). However, there were some caveats. There was toxicity and change in quality of life for a while in those patients receiving ASCT as would be expected. Furthermore, the study only allowed 65 years old or younger and ASCT may not be wise for older patients. The discussant made a strong point that African Americans tend to have higher risk disease with different mutations and might also be better served by have ASCT later.
The conclusion was that, given all the new therapies in myeloma for second line and beyond, ASCT should be a discussion with each new patient and not an automatic decision.
This study was simultaneously published in the New England Journal of Medicine.
Adagrasib promising for pretreated patients with NSCLC with KRAS mutation
In patients with advanced or metastatic non–small cell lung cancer (NSCLC), adagrasib was found to be well tolerated and “demonstrates promising efficacy” for patients with the KRAS G12C mutation (KRYSTAL-1, abstract 9002). This was a phase 2 registration trial of 116 patients who were treated with 600 mg of adagrasib twice orally. Patients all had previous chemotherapy or immunotherapy or both. The overall response rate was a surprisingly good 43% (complete response and partial response). Disease control was an incredible 80% if stable disease was included. The duration of response was 8.5 months, progression-free survival was 6.5 months, and overall survival was 12.6 months. Furthermore, 33% of those with brain metastases had a complete response or partial response.
The take-home message is that, since 15% of NSCLC metastatic patients are KRAS mutant G12C, we should be watching for such patients in our biomarker analysis. While we have sotorasib – approved by the Food and Drug Administration for NSCLC – the results of this study suggests we may have another new molecule in the same class.
Neoadjuvant chemotherapy with immunotherapy for NSCLC
It may be time to consider neoadjuvant chemotherapy with immunotherapy, such as nivolumab, for patients with NSCLC in order to achieve the best response possible.
In NADIM II, investigators led by Mariano Provencio-Pulla, MD, of the Hospital Universitario Puerta de Hierro-Majadahonda, Madrid, confirmed the superiority of chemotherapy with immunotherapy for patients with resectable stage IIIA NSCLC. NADIM included patients with resectable stage IIIA/B NSCLC who were randomized 2:1 to receive carboplatin taxol neoadjuvant therapy with or without nivolumab before and after surgery. The pathological complete response rates overall were 36% versus 7%, favoring the nivolumab arm, but even higher pCR rates occurred in patients with PD-L1 over 50%.
In closing, always check MMR, KRAS, BRAF, and HER2. For wild-type left-sided mCRC, consider FOLFOX or FOLFIRI with an anti-EGFR. For KRAS mutant or right-sided colon tumor, consider FOLFOX or FOLFIRI with bevacizumab, followed by maintenance 5FU or capecitabine, with or without bevacizumab.
CHICAGO – The American Society of Clinical Oncology recently wrapped its annual meeting in Chicago. Here, I highlight some presentations that stood out to me.
A first-line treatment for metastatic colorectal cancer
The plenary session did not disappoint. In abstract LBA1, investigators presented first-line treatment for patients with metastatic colorectal cancer who were randomized to receive mFOLFOX6 with either bevacizumab or panitumumab in RAS wild-type positive patients. This was the phase 3 PARADIGM trial.
The primary outcome for this study was overall survival. It included 823 patients who were randomized 1:1 with a subset analysis of whether the primary tumor was on the left or right side of the colon. At 61 months follow-up, the median overall survival results for left-sided colon cancer was 38 months versus 34 months. It was statistically significant favoring the panitumumab arm. It improved the curable resection rate for patients with left-sided tumors from 11% in the bevacizumab arm to 18% in the panitumumab arm. Interestingly, patients randomized with right-sided tumors showed no difference in overall survival. The investigator, Takayuki Yoshino, MD, PhD, National Cancer Center Hospital East, Kashiwa, Japan, said the study findings support the use of mFOLFOX6 with panitumumab in left-sided RAS wild type as first-line therapy in metastatic colorectal patients.
A possible new standard of care in breast cancer
Shanu Modi, MD, of Memorial Sloan Kettering Cancer Center, New York, received a standing ovation and deserved it. In the phase 3 clinical trial DESTINY-Breast04 (abstract LBA3), she demonstrated that trastuzumab deruxtecan (T-DXd) for patients with metastatic breast cancer who were HER2 low (IHC 1+ or 2+ ISH-), led to a statistically significant and clinically meaningful benefit in both progression free survival and overall survival. In this trial, patients were randomized 2:1 to receive trastuzumab deruxtecan or physician’s choice of chemotherapy. All patients had at least one to two lines of chemotherapy before entering the trial. Hormone-positive patients were allowed if they had already received and failed, or progressed on hormone therapy.
Previously, most patients were treated either with eribulin with some receiving capecitabine, gemcitabine or taxane, or hormone therapy if hormone positive.
The progression-free survival was 10.1 versus 5.4 months in hormone-positive patients, and in all patients (hormone receptor positive or negative), there was a likewise improvement of 9.9 versus 5.1 months progression free survival.
Overall survival was equally impressive. In the hormone receptor–positive patients, the hazard ratio was 0.64 with a 23.9 versus 17.5 month survival. If all patients were included, the HR was again 0.64 with 23.4 versus 16.8 month survival. Even the triple-negative breast cancer patients had a HR of 0.48 with 18.2 versus 8.3 months survival. Adverse events were quite tolerable with some nausea, some decreased white count, and only an interstitial lung disease of grade 2 or less in 12%.
Trastuzumab deruxtecan is a targeted treatment which, in addition to striking its target, also targets other tumor cells that are part of the cancer. The results of this study may lead to a new standard of care of this patient population.
The study by Dr. Modi and colleagues was simultaneously published in the New England Journal of Medicine.
Improving outcomes in multiple myeloma
In abstract LBA4, Paul G. Richardson, MD, of the Dana-Farber Cancer Institute, Boston, asks if autologous stem cell transplant (ASCT) can improve outcomes after induction with an RVD regimen (lenalidomide, bortezomib, and dexamethasone) and lenalidomide (Revlimid) maintenance for newly diagnosed patients with multiple myeloma in the DETERMINATION study.
The take home here was quite interesting. In fact, there is no difference in overall survival if patients get this standard RVD/lenalidomide maintenance induction with or without ASCT. However, the progression free survival was better with ASCT: 46 versus 67 months (improvement of 21 months). However, there were some caveats. There was toxicity and change in quality of life for a while in those patients receiving ASCT as would be expected. Furthermore, the study only allowed 65 years old or younger and ASCT may not be wise for older patients. The discussant made a strong point that African Americans tend to have higher risk disease with different mutations and might also be better served by have ASCT later.
The conclusion was that, given all the new therapies in myeloma for second line and beyond, ASCT should be a discussion with each new patient and not an automatic decision.
This study was simultaneously published in the New England Journal of Medicine.
Adagrasib promising for pretreated patients with NSCLC with KRAS mutation
In patients with advanced or metastatic non–small cell lung cancer (NSCLC), adagrasib was found to be well tolerated and “demonstrates promising efficacy” for patients with the KRAS G12C mutation (KRYSTAL-1, abstract 9002). This was a phase 2 registration trial of 116 patients who were treated with 600 mg of adagrasib twice orally. Patients all had previous chemotherapy or immunotherapy or both. The overall response rate was a surprisingly good 43% (complete response and partial response). Disease control was an incredible 80% if stable disease was included. The duration of response was 8.5 months, progression-free survival was 6.5 months, and overall survival was 12.6 months. Furthermore, 33% of those with brain metastases had a complete response or partial response.
The take-home message is that, since 15% of NSCLC metastatic patients are KRAS mutant G12C, we should be watching for such patients in our biomarker analysis. While we have sotorasib – approved by the Food and Drug Administration for NSCLC – the results of this study suggests we may have another new molecule in the same class.
Neoadjuvant chemotherapy with immunotherapy for NSCLC
It may be time to consider neoadjuvant chemotherapy with immunotherapy, such as nivolumab, for patients with NSCLC in order to achieve the best response possible.
In NADIM II, investigators led by Mariano Provencio-Pulla, MD, of the Hospital Universitario Puerta de Hierro-Majadahonda, Madrid, confirmed the superiority of chemotherapy with immunotherapy for patients with resectable stage IIIA NSCLC. NADIM included patients with resectable stage IIIA/B NSCLC who were randomized 2:1 to receive carboplatin taxol neoadjuvant therapy with or without nivolumab before and after surgery. The pathological complete response rates overall were 36% versus 7%, favoring the nivolumab arm, but even higher pCR rates occurred in patients with PD-L1 over 50%.
In closing, always check MMR, KRAS, BRAF, and HER2. For wild-type left-sided mCRC, consider FOLFOX or FOLFIRI with an anti-EGFR. For KRAS mutant or right-sided colon tumor, consider FOLFOX or FOLFIRI with bevacizumab, followed by maintenance 5FU or capecitabine, with or without bevacizumab.
AT ASCO 2022
Melanoma incidence is up, but death rates are down
CHICAGO – , according to a new analysis of the National Cancer Institute SEER database between 1975 and 2019.
“This is very encouraging data and represents the real-world effectiveness of these therapies. The cost of these therapies can be prohibitive for universal treatment access, so the ways to address the accessibility of these treatments and the health care costs need to be supported,” said lead author Navkirat Kaur Kahlon MD, a hematology/oncology fellow at the University of Toledo (Ohio). The study was presented at the annual meeting of the American Society of Clinical Oncology.
According to the American Cancer Society, the 5-year mortality for regional melanoma metastasis is 68%, and 30% for distant metastasis. However, these numbers may underestimate current survival. “People now being diagnosed with melanoma may have a better outlook than these numbers show. Treatments have improved over time, and these numbers are based on people who were diagnosed and treated at least 5 years earlier,” the American Cancer Society wrote.
Other studies have found similar trends. According to Cancer Research UK, 5-year melanoma skin cancer survival approximately doubled, from 46% to 90%, between 1971 and 2010. And, 1-year survival increased from 74% to 96%, but these improvements predated immune checkpoint inhibitors. An analysis of the Canadian Cancer Registry and Canadian Vital Statistics found an increasing incidence of melanoma, but a drop in mortality since 2013. A study of melanoma outcomes in Hungary also found increased incidence, while mortality declined by 16.55% between 2011 and 2019 (P =.013).
“These new drugs, which include immunotherapies and targeted therapies, are effective treatments in the clinical trial data, so the magnitude of drop seen in population mortality was not surprising but very exciting,” Dr. Kahlon said.
The findings are encouraging, but prevention remains the most important strategy. “The utility of sun-protective strategies and policies should be encouraged,” she added.
Cytotoxic chemotherapy has poor efficacy against metastatic melanoma, but novel therapies such as checkpoint inhibitors increased expected survival from months to years. “Given the magnitude of benefit compared to traditional chemotherapy in clinical trials, we decided to see if the real-world population is deriving the same benefit,” Dr. Kahlon said.
The researchers found that the annual percentage change (APC) melanoma mortality rate (MMR) was +1.65% between 1975 and 1988 (P < .01). The APC was 0.01% between 1988 and 2013, which was not statistically significant (P = .85). Between 2013 and 2017, APC was –6.24% (P < .01), and it was –1.56% between 2017 and 2019 (P = .53).
The increase in melanoma mortality between 1975 and 1988 may be due to changes in the way that SEER data was collected. “It is possible that this increase was at least in part due to better capturing of the data. There may also be a contribution of increased mortality due to increased incidence of diagnoses related to increased UV exposure. From the 1920s, increased sun exposure and bronzed skin became fashionable. In the 1940s-1960s, tanning oils and lotions became more popular, and there may have been an increase in UV exposure during that time, which later led to an increase in diagnosis and, without effective therapies, mortality. Further, the use of indoor tanning beds from the 1970s onward may have contributed to increased UV exposure, incidence, and mortality,” she said.
On the other hand, the researchers noted a slowing of mortality reduction between 2017 and 2019. This was not a surprise, Dr. Kahlon said, since by that time most novel therapies were being introduced in the adjuvant setting. “The mortality benefit, if any, from adjuvant treatments is seen over a longer period and may not yet be captured in SEER data. Even the clinical trial data for most of these treatments have not shown an overall survival advantage and require more time for the data to mature. It will be interesting to see how these trends change in the near future,” Dr. Kahlon said.
The study was limited by its retrospective nature. Dr. Kahlon has no relevant financial disclosures.
CHICAGO – , according to a new analysis of the National Cancer Institute SEER database between 1975 and 2019.
“This is very encouraging data and represents the real-world effectiveness of these therapies. The cost of these therapies can be prohibitive for universal treatment access, so the ways to address the accessibility of these treatments and the health care costs need to be supported,” said lead author Navkirat Kaur Kahlon MD, a hematology/oncology fellow at the University of Toledo (Ohio). The study was presented at the annual meeting of the American Society of Clinical Oncology.
According to the American Cancer Society, the 5-year mortality for regional melanoma metastasis is 68%, and 30% for distant metastasis. However, these numbers may underestimate current survival. “People now being diagnosed with melanoma may have a better outlook than these numbers show. Treatments have improved over time, and these numbers are based on people who were diagnosed and treated at least 5 years earlier,” the American Cancer Society wrote.
Other studies have found similar trends. According to Cancer Research UK, 5-year melanoma skin cancer survival approximately doubled, from 46% to 90%, between 1971 and 2010. And, 1-year survival increased from 74% to 96%, but these improvements predated immune checkpoint inhibitors. An analysis of the Canadian Cancer Registry and Canadian Vital Statistics found an increasing incidence of melanoma, but a drop in mortality since 2013. A study of melanoma outcomes in Hungary also found increased incidence, while mortality declined by 16.55% between 2011 and 2019 (P =.013).
“These new drugs, which include immunotherapies and targeted therapies, are effective treatments in the clinical trial data, so the magnitude of drop seen in population mortality was not surprising but very exciting,” Dr. Kahlon said.
The findings are encouraging, but prevention remains the most important strategy. “The utility of sun-protective strategies and policies should be encouraged,” she added.
Cytotoxic chemotherapy has poor efficacy against metastatic melanoma, but novel therapies such as checkpoint inhibitors increased expected survival from months to years. “Given the magnitude of benefit compared to traditional chemotherapy in clinical trials, we decided to see if the real-world population is deriving the same benefit,” Dr. Kahlon said.
The researchers found that the annual percentage change (APC) melanoma mortality rate (MMR) was +1.65% between 1975 and 1988 (P < .01). The APC was 0.01% between 1988 and 2013, which was not statistically significant (P = .85). Between 2013 and 2017, APC was –6.24% (P < .01), and it was –1.56% between 2017 and 2019 (P = .53).
The increase in melanoma mortality between 1975 and 1988 may be due to changes in the way that SEER data was collected. “It is possible that this increase was at least in part due to better capturing of the data. There may also be a contribution of increased mortality due to increased incidence of diagnoses related to increased UV exposure. From the 1920s, increased sun exposure and bronzed skin became fashionable. In the 1940s-1960s, tanning oils and lotions became more popular, and there may have been an increase in UV exposure during that time, which later led to an increase in diagnosis and, without effective therapies, mortality. Further, the use of indoor tanning beds from the 1970s onward may have contributed to increased UV exposure, incidence, and mortality,” she said.
On the other hand, the researchers noted a slowing of mortality reduction between 2017 and 2019. This was not a surprise, Dr. Kahlon said, since by that time most novel therapies were being introduced in the adjuvant setting. “The mortality benefit, if any, from adjuvant treatments is seen over a longer period and may not yet be captured in SEER data. Even the clinical trial data for most of these treatments have not shown an overall survival advantage and require more time for the data to mature. It will be interesting to see how these trends change in the near future,” Dr. Kahlon said.
The study was limited by its retrospective nature. Dr. Kahlon has no relevant financial disclosures.
CHICAGO – , according to a new analysis of the National Cancer Institute SEER database between 1975 and 2019.
“This is very encouraging data and represents the real-world effectiveness of these therapies. The cost of these therapies can be prohibitive for universal treatment access, so the ways to address the accessibility of these treatments and the health care costs need to be supported,” said lead author Navkirat Kaur Kahlon MD, a hematology/oncology fellow at the University of Toledo (Ohio). The study was presented at the annual meeting of the American Society of Clinical Oncology.
According to the American Cancer Society, the 5-year mortality for regional melanoma metastasis is 68%, and 30% for distant metastasis. However, these numbers may underestimate current survival. “People now being diagnosed with melanoma may have a better outlook than these numbers show. Treatments have improved over time, and these numbers are based on people who were diagnosed and treated at least 5 years earlier,” the American Cancer Society wrote.
Other studies have found similar trends. According to Cancer Research UK, 5-year melanoma skin cancer survival approximately doubled, from 46% to 90%, between 1971 and 2010. And, 1-year survival increased from 74% to 96%, but these improvements predated immune checkpoint inhibitors. An analysis of the Canadian Cancer Registry and Canadian Vital Statistics found an increasing incidence of melanoma, but a drop in mortality since 2013. A study of melanoma outcomes in Hungary also found increased incidence, while mortality declined by 16.55% between 2011 and 2019 (P =.013).
“These new drugs, which include immunotherapies and targeted therapies, are effective treatments in the clinical trial data, so the magnitude of drop seen in population mortality was not surprising but very exciting,” Dr. Kahlon said.
The findings are encouraging, but prevention remains the most important strategy. “The utility of sun-protective strategies and policies should be encouraged,” she added.
Cytotoxic chemotherapy has poor efficacy against metastatic melanoma, but novel therapies such as checkpoint inhibitors increased expected survival from months to years. “Given the magnitude of benefit compared to traditional chemotherapy in clinical trials, we decided to see if the real-world population is deriving the same benefit,” Dr. Kahlon said.
The researchers found that the annual percentage change (APC) melanoma mortality rate (MMR) was +1.65% between 1975 and 1988 (P < .01). The APC was 0.01% between 1988 and 2013, which was not statistically significant (P = .85). Between 2013 and 2017, APC was –6.24% (P < .01), and it was –1.56% between 2017 and 2019 (P = .53).
The increase in melanoma mortality between 1975 and 1988 may be due to changes in the way that SEER data was collected. “It is possible that this increase was at least in part due to better capturing of the data. There may also be a contribution of increased mortality due to increased incidence of diagnoses related to increased UV exposure. From the 1920s, increased sun exposure and bronzed skin became fashionable. In the 1940s-1960s, tanning oils and lotions became more popular, and there may have been an increase in UV exposure during that time, which later led to an increase in diagnosis and, without effective therapies, mortality. Further, the use of indoor tanning beds from the 1970s onward may have contributed to increased UV exposure, incidence, and mortality,” she said.
On the other hand, the researchers noted a slowing of mortality reduction between 2017 and 2019. This was not a surprise, Dr. Kahlon said, since by that time most novel therapies were being introduced in the adjuvant setting. “The mortality benefit, if any, from adjuvant treatments is seen over a longer period and may not yet be captured in SEER data. Even the clinical trial data for most of these treatments have not shown an overall survival advantage and require more time for the data to mature. It will be interesting to see how these trends change in the near future,” Dr. Kahlon said.
The study was limited by its retrospective nature. Dr. Kahlon has no relevant financial disclosures.
AT ASCO 2022
Adjuvant vs. neoadjuvant? What has ASCO 2022 taught us regarding resectable NSCLC?
for non–small cell lung cancer (NSCLC). While there has been some notable progress in this area, we need phase 3 trials that compare the two therapeutic approaches.
Investigators reporting at the 2022 annual meeting of American Society of Clinical Oncology focused primarily on neoadjuvant treatment, which I’ll address here.
In the randomized, phase 2 NADIM II clinical trial reported at the meeting, researchers expanded on the results of NADIM published in 2020 in the Lancet Oncology and in May 2022 in the Journal of Clinical Oncology along with CheckMate 816 results published in the New England Journal of Medicine.
In each of these three studies, researchers compared nivolumab plus chemotherapy versus chemotherapy alone (abstract 8501) as a neoadjuvant treatment for resectable stage IIIA NSCLC. In the study reported at ASCO 2022, patients with resectable clinical stage IIIA-B (per American Joint Committee on Cancer 8th edition) NSCLC and no known EGFR/ALK alterations, were randomized to receive preoperative nivolumab plus chemotherapy (paclitaxel and carboplatin; n = 57) or chemotherapy (n = 29) alone followed by surgery.
The primary endpoint was pathological complete response (pCR); secondary endpoints included major pathological response, safety and tolerability, impact on surgical issues such as delayed or canceled surgeries or length of hospital stay, overall survival and progression free survival. The pCR rate was 36.8% in the neoadjuvant nivolumab plus chemotherapy arm and 6.9% in the chemotherapy alone arm. (P = .0068). 25% of patients on the nivolumab plus chemo arm had grade 3-4 adverse events, compared with 10.3% in the control arm. 93% of patients on the nivolumab plus chemo arm underwent definitive surgery whereas 69.0% of the patients on the chemo alone arm had definitive surgery. (P = .008)
What else did we learn about neoadjuvant treatment at the meeting?
Investigators looking at the optimal number of neoadjuvant cycles (abstract 8500) found that three cycles of sintilimab (an investigational PD-1 inhibitor) produced a numerically higher major pathological response rate, compared with two cycles (when given in concert with platinum-doublet chemotherapy). And, neoadjuvant chemoradiotherapy does not result in significant survival benefits when compared with neoadjuvant chemotherapy alone (abstract 8503).
Of course, when it comes to resectable NSCLC, the goal of treatment is to increase the cure rate and improve survival. No randomized studies have reported yet on overall survival, probably because they are too immature. Instead, disease-free survival (DFS) or event-free survival (EFS) are often used as surrogate endpoints. Since none of the studies reported at ASCO reported on DFS or EFS, we need to look elsewhere. CheckMate 816 was a phase 3 study which randomized patients with stages IB-IIIA NSCLC to receive neoadjuvant nivolumab plus platinum-based chemotherapy or neoadjuvant platinum-based chemotherapy alone, followed by resection. The median EFS was 31.6 months with nivolumab plus chemotherapy and 20.8 months with chemotherapy alone (P = .005). The percentage of patients with a pCR was 24.0% and 2.2%, respectively (P < .001).
We all know one has to be careful when doing cross-trial comparisons as these studies differ by the percentage of patients with various stages of disease, the type of immunotherapy and chemotherapy used, etc. However, I think we can agree that neoadjuvant chemoimmunotherapy results in better outcomes than chemotherapy alone.
Of course, resectable NSCLC is, by definition, resectable. And traditionally, resection is followed by adjuvant chemotherapy to eradicate micrometastases. Unfortunately, the current standard of care for completely resected early-stage NSCLC (stage I [tumor ≥ 4 cm] to IIIA) involves adjuvant platinum-based combination chemotherapy which results in only a modest 4%-5% improvement in survival versus observation.
Given these modest results, as in the neoadjuvant space, investigators have looked at the benefit of adding immunotherapy to adjuvant chemotherapy. One such study has been reported. IMpower 010 randomly assigned patients with completely resected stage IB (tumors ≥ 4 cm) to IIIA NSCLC, whose tumor cells expressed at least 1% PD-L1, to receive adjuvant atezolizumab or best supportive care after adjuvant platinum-based chemotherapy. In the stage II-IIIA population whose tumors expressed PD-L1 on 1% or more of tumor cells, 3-year DFS rates were 60% and 48% in the atezolizumab and best supportive care arms, respectively (hazard ratio, 0·66 P =·.0039). In all patients in the stage II-IIIA population, the 3-year DFS rates were 56% in the atezolizumab group and 49% in the best supportive care group, (HR, 0.79; P = .020).
KEYNOTE-091, reported at the 2021 annual meeting of the European Society for Medical Oncology, randomized early-stage NSCLC patients following complete resection and adjuvant chemotherapy to pembrolizumab or placebo. Median DFS for the overall population was 53.6 months for patients in the pembro arm versus 42 months in the placebo arm (HR, 0.76; P = .0014). Interestingly, the benefit was not seen in patients with PD-L1 with at least 50%, where the 18-month DFS rate was 71.7% in the pembro arm and 70.2% in the placebo arm (HR, 0.82; P = .14). Although the contradictory results of PD-L1 as a biomarker is puzzling, I think we can agree that the addition of immunotherapy following adjuvant chemotherapy improves outcomes compared to adjuvant chemotherapy alone.
What to do when a patient presents with resectable disease?
Cross-trial comparisons are fraught with danger. Until we have a phase 3 study comparing concurrent neoadjuvant chemo/immunotherapy with concurrent adjuvant chemo/immunotherapy, I do not think we can answer the question “which is better?” However, there are some caveats to keep in mind when deciding on which approach to recommend to our patients: First, neoadjuvant treatment requires biomarker testing to ensure the patient does not have EGFR or ALK mutations. This will necessitate a delay in the operation. Will patients be willing to wait? Will the surgeon? Or, would patients prefer to proceed with surgery while the results are pending? Yes, neoadjuvant therapy gives you information regarding the pCR rate, but does that help you in subsequent management of the patient? We do not know.
Secondly, the two adjuvant studies used adjuvant chemotherapy followed by adjuvant immunotherapy, as contrasted to the neoadjuvant study which used concurrent chemo/immunotherapy. Given the longer duration of treatment in postoperative sequential adjuvant studies, there tends to be more drop off because of patients being unwilling or unfit postoperatively to receive long courses of therapy. In IMpower 010, 1,269 patients completed adjuvant chemotherapy; 1,005 were randomized, and of the 507 assigned to the atezolizumab/chemo group, only 323 completed treatment.
Finally, we must beware of using neoadjuvant chemo/immunotherapy to “down-stage” a patient. KEYNOTE-091 included patients with IIIA disease and no benefit to adjuvant chemotherapy followed by immunotherapy was found in this subgroup of patients, which leads me to wonder if these patients were appropriately selected as surgical candidates. In the NADIM II trials, 9 of 29 patients on the neoadjuvant chemotherapy were not resected.
So, many questions remain. In addition to the ones we’ve raised, there is a clear and immediate need for predictive and prognostic biomarkers. In the NADIM II trial, PD-L1 expression was a predictive biomarker of response. The pCR rate for patients with a PD-L1 tumor expression of less than 1%, 1%-49%, and 50% or higher was 15%, 41.7%, and 61.1%, respectively. However, in KEYNOTE-091, the benefit was not seen in patients with PD-L1 of at least than 50%, where the 18-month DFS rate was 71.7% in the pembro arm and 70.2% in the placebo arm.
Another possible biomarker: circulating tumor DNA. In the first NADIM study, three low pretreatment levels of ctDNA were significantly associated with improved progression-free survival and overall survival (HR, 0.20 and HR, 0.07, respectively). Although clinical response did not predict survival outcomes, undetectable ctDNA levels after neoadjuvant treatment were significantly associated with progression-free survival and overall survival (HR, 0.26 and HR0.04, respectively). Similarly, in CheckMate 816, clearance of ctDNA was associated with longer EFS in patients with ctDNA clearance than in those without ctDNA clearance in both the nivolumab/chemotherapy group (HR, 0.60) and the chemotherapy-alone group (HR, 0.63).
Hopefully, ASCO 2023 will provide more answers.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation.
for non–small cell lung cancer (NSCLC). While there has been some notable progress in this area, we need phase 3 trials that compare the two therapeutic approaches.
Investigators reporting at the 2022 annual meeting of American Society of Clinical Oncology focused primarily on neoadjuvant treatment, which I’ll address here.
In the randomized, phase 2 NADIM II clinical trial reported at the meeting, researchers expanded on the results of NADIM published in 2020 in the Lancet Oncology and in May 2022 in the Journal of Clinical Oncology along with CheckMate 816 results published in the New England Journal of Medicine.
In each of these three studies, researchers compared nivolumab plus chemotherapy versus chemotherapy alone (abstract 8501) as a neoadjuvant treatment for resectable stage IIIA NSCLC. In the study reported at ASCO 2022, patients with resectable clinical stage IIIA-B (per American Joint Committee on Cancer 8th edition) NSCLC and no known EGFR/ALK alterations, were randomized to receive preoperative nivolumab plus chemotherapy (paclitaxel and carboplatin; n = 57) or chemotherapy (n = 29) alone followed by surgery.
The primary endpoint was pathological complete response (pCR); secondary endpoints included major pathological response, safety and tolerability, impact on surgical issues such as delayed or canceled surgeries or length of hospital stay, overall survival and progression free survival. The pCR rate was 36.8% in the neoadjuvant nivolumab plus chemotherapy arm and 6.9% in the chemotherapy alone arm. (P = .0068). 25% of patients on the nivolumab plus chemo arm had grade 3-4 adverse events, compared with 10.3% in the control arm. 93% of patients on the nivolumab plus chemo arm underwent definitive surgery whereas 69.0% of the patients on the chemo alone arm had definitive surgery. (P = .008)
What else did we learn about neoadjuvant treatment at the meeting?
Investigators looking at the optimal number of neoadjuvant cycles (abstract 8500) found that three cycles of sintilimab (an investigational PD-1 inhibitor) produced a numerically higher major pathological response rate, compared with two cycles (when given in concert with platinum-doublet chemotherapy). And, neoadjuvant chemoradiotherapy does not result in significant survival benefits when compared with neoadjuvant chemotherapy alone (abstract 8503).
Of course, when it comes to resectable NSCLC, the goal of treatment is to increase the cure rate and improve survival. No randomized studies have reported yet on overall survival, probably because they are too immature. Instead, disease-free survival (DFS) or event-free survival (EFS) are often used as surrogate endpoints. Since none of the studies reported at ASCO reported on DFS or EFS, we need to look elsewhere. CheckMate 816 was a phase 3 study which randomized patients with stages IB-IIIA NSCLC to receive neoadjuvant nivolumab plus platinum-based chemotherapy or neoadjuvant platinum-based chemotherapy alone, followed by resection. The median EFS was 31.6 months with nivolumab plus chemotherapy and 20.8 months with chemotherapy alone (P = .005). The percentage of patients with a pCR was 24.0% and 2.2%, respectively (P < .001).
We all know one has to be careful when doing cross-trial comparisons as these studies differ by the percentage of patients with various stages of disease, the type of immunotherapy and chemotherapy used, etc. However, I think we can agree that neoadjuvant chemoimmunotherapy results in better outcomes than chemotherapy alone.
Of course, resectable NSCLC is, by definition, resectable. And traditionally, resection is followed by adjuvant chemotherapy to eradicate micrometastases. Unfortunately, the current standard of care for completely resected early-stage NSCLC (stage I [tumor ≥ 4 cm] to IIIA) involves adjuvant platinum-based combination chemotherapy which results in only a modest 4%-5% improvement in survival versus observation.
Given these modest results, as in the neoadjuvant space, investigators have looked at the benefit of adding immunotherapy to adjuvant chemotherapy. One such study has been reported. IMpower 010 randomly assigned patients with completely resected stage IB (tumors ≥ 4 cm) to IIIA NSCLC, whose tumor cells expressed at least 1% PD-L1, to receive adjuvant atezolizumab or best supportive care after adjuvant platinum-based chemotherapy. In the stage II-IIIA population whose tumors expressed PD-L1 on 1% or more of tumor cells, 3-year DFS rates were 60% and 48% in the atezolizumab and best supportive care arms, respectively (hazard ratio, 0·66 P =·.0039). In all patients in the stage II-IIIA population, the 3-year DFS rates were 56% in the atezolizumab group and 49% in the best supportive care group, (HR, 0.79; P = .020).
KEYNOTE-091, reported at the 2021 annual meeting of the European Society for Medical Oncology, randomized early-stage NSCLC patients following complete resection and adjuvant chemotherapy to pembrolizumab or placebo. Median DFS for the overall population was 53.6 months for patients in the pembro arm versus 42 months in the placebo arm (HR, 0.76; P = .0014). Interestingly, the benefit was not seen in patients with PD-L1 with at least 50%, where the 18-month DFS rate was 71.7% in the pembro arm and 70.2% in the placebo arm (HR, 0.82; P = .14). Although the contradictory results of PD-L1 as a biomarker is puzzling, I think we can agree that the addition of immunotherapy following adjuvant chemotherapy improves outcomes compared to adjuvant chemotherapy alone.
What to do when a patient presents with resectable disease?
Cross-trial comparisons are fraught with danger. Until we have a phase 3 study comparing concurrent neoadjuvant chemo/immunotherapy with concurrent adjuvant chemo/immunotherapy, I do not think we can answer the question “which is better?” However, there are some caveats to keep in mind when deciding on which approach to recommend to our patients: First, neoadjuvant treatment requires biomarker testing to ensure the patient does not have EGFR or ALK mutations. This will necessitate a delay in the operation. Will patients be willing to wait? Will the surgeon? Or, would patients prefer to proceed with surgery while the results are pending? Yes, neoadjuvant therapy gives you information regarding the pCR rate, but does that help you in subsequent management of the patient? We do not know.
Secondly, the two adjuvant studies used adjuvant chemotherapy followed by adjuvant immunotherapy, as contrasted to the neoadjuvant study which used concurrent chemo/immunotherapy. Given the longer duration of treatment in postoperative sequential adjuvant studies, there tends to be more drop off because of patients being unwilling or unfit postoperatively to receive long courses of therapy. In IMpower 010, 1,269 patients completed adjuvant chemotherapy; 1,005 were randomized, and of the 507 assigned to the atezolizumab/chemo group, only 323 completed treatment.
Finally, we must beware of using neoadjuvant chemo/immunotherapy to “down-stage” a patient. KEYNOTE-091 included patients with IIIA disease and no benefit to adjuvant chemotherapy followed by immunotherapy was found in this subgroup of patients, which leads me to wonder if these patients were appropriately selected as surgical candidates. In the NADIM II trials, 9 of 29 patients on the neoadjuvant chemotherapy were not resected.
So, many questions remain. In addition to the ones we’ve raised, there is a clear and immediate need for predictive and prognostic biomarkers. In the NADIM II trial, PD-L1 expression was a predictive biomarker of response. The pCR rate for patients with a PD-L1 tumor expression of less than 1%, 1%-49%, and 50% or higher was 15%, 41.7%, and 61.1%, respectively. However, in KEYNOTE-091, the benefit was not seen in patients with PD-L1 of at least than 50%, where the 18-month DFS rate was 71.7% in the pembro arm and 70.2% in the placebo arm.
Another possible biomarker: circulating tumor DNA. In the first NADIM study, three low pretreatment levels of ctDNA were significantly associated with improved progression-free survival and overall survival (HR, 0.20 and HR, 0.07, respectively). Although clinical response did not predict survival outcomes, undetectable ctDNA levels after neoadjuvant treatment were significantly associated with progression-free survival and overall survival (HR, 0.26 and HR0.04, respectively). Similarly, in CheckMate 816, clearance of ctDNA was associated with longer EFS in patients with ctDNA clearance than in those without ctDNA clearance in both the nivolumab/chemotherapy group (HR, 0.60) and the chemotherapy-alone group (HR, 0.63).
Hopefully, ASCO 2023 will provide more answers.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation.
for non–small cell lung cancer (NSCLC). While there has been some notable progress in this area, we need phase 3 trials that compare the two therapeutic approaches.
Investigators reporting at the 2022 annual meeting of American Society of Clinical Oncology focused primarily on neoadjuvant treatment, which I’ll address here.
In the randomized, phase 2 NADIM II clinical trial reported at the meeting, researchers expanded on the results of NADIM published in 2020 in the Lancet Oncology and in May 2022 in the Journal of Clinical Oncology along with CheckMate 816 results published in the New England Journal of Medicine.
In each of these three studies, researchers compared nivolumab plus chemotherapy versus chemotherapy alone (abstract 8501) as a neoadjuvant treatment for resectable stage IIIA NSCLC. In the study reported at ASCO 2022, patients with resectable clinical stage IIIA-B (per American Joint Committee on Cancer 8th edition) NSCLC and no known EGFR/ALK alterations, were randomized to receive preoperative nivolumab plus chemotherapy (paclitaxel and carboplatin; n = 57) or chemotherapy (n = 29) alone followed by surgery.
The primary endpoint was pathological complete response (pCR); secondary endpoints included major pathological response, safety and tolerability, impact on surgical issues such as delayed or canceled surgeries or length of hospital stay, overall survival and progression free survival. The pCR rate was 36.8% in the neoadjuvant nivolumab plus chemotherapy arm and 6.9% in the chemotherapy alone arm. (P = .0068). 25% of patients on the nivolumab plus chemo arm had grade 3-4 adverse events, compared with 10.3% in the control arm. 93% of patients on the nivolumab plus chemo arm underwent definitive surgery whereas 69.0% of the patients on the chemo alone arm had definitive surgery. (P = .008)
What else did we learn about neoadjuvant treatment at the meeting?
Investigators looking at the optimal number of neoadjuvant cycles (abstract 8500) found that three cycles of sintilimab (an investigational PD-1 inhibitor) produced a numerically higher major pathological response rate, compared with two cycles (when given in concert with platinum-doublet chemotherapy). And, neoadjuvant chemoradiotherapy does not result in significant survival benefits when compared with neoadjuvant chemotherapy alone (abstract 8503).
Of course, when it comes to resectable NSCLC, the goal of treatment is to increase the cure rate and improve survival. No randomized studies have reported yet on overall survival, probably because they are too immature. Instead, disease-free survival (DFS) or event-free survival (EFS) are often used as surrogate endpoints. Since none of the studies reported at ASCO reported on DFS or EFS, we need to look elsewhere. CheckMate 816 was a phase 3 study which randomized patients with stages IB-IIIA NSCLC to receive neoadjuvant nivolumab plus platinum-based chemotherapy or neoadjuvant platinum-based chemotherapy alone, followed by resection. The median EFS was 31.6 months with nivolumab plus chemotherapy and 20.8 months with chemotherapy alone (P = .005). The percentage of patients with a pCR was 24.0% and 2.2%, respectively (P < .001).
We all know one has to be careful when doing cross-trial comparisons as these studies differ by the percentage of patients with various stages of disease, the type of immunotherapy and chemotherapy used, etc. However, I think we can agree that neoadjuvant chemoimmunotherapy results in better outcomes than chemotherapy alone.
Of course, resectable NSCLC is, by definition, resectable. And traditionally, resection is followed by adjuvant chemotherapy to eradicate micrometastases. Unfortunately, the current standard of care for completely resected early-stage NSCLC (stage I [tumor ≥ 4 cm] to IIIA) involves adjuvant platinum-based combination chemotherapy which results in only a modest 4%-5% improvement in survival versus observation.
Given these modest results, as in the neoadjuvant space, investigators have looked at the benefit of adding immunotherapy to adjuvant chemotherapy. One such study has been reported. IMpower 010 randomly assigned patients with completely resected stage IB (tumors ≥ 4 cm) to IIIA NSCLC, whose tumor cells expressed at least 1% PD-L1, to receive adjuvant atezolizumab or best supportive care after adjuvant platinum-based chemotherapy. In the stage II-IIIA population whose tumors expressed PD-L1 on 1% or more of tumor cells, 3-year DFS rates were 60% and 48% in the atezolizumab and best supportive care arms, respectively (hazard ratio, 0·66 P =·.0039). In all patients in the stage II-IIIA population, the 3-year DFS rates were 56% in the atezolizumab group and 49% in the best supportive care group, (HR, 0.79; P = .020).
KEYNOTE-091, reported at the 2021 annual meeting of the European Society for Medical Oncology, randomized early-stage NSCLC patients following complete resection and adjuvant chemotherapy to pembrolizumab or placebo. Median DFS for the overall population was 53.6 months for patients in the pembro arm versus 42 months in the placebo arm (HR, 0.76; P = .0014). Interestingly, the benefit was not seen in patients with PD-L1 with at least 50%, where the 18-month DFS rate was 71.7% in the pembro arm and 70.2% in the placebo arm (HR, 0.82; P = .14). Although the contradictory results of PD-L1 as a biomarker is puzzling, I think we can agree that the addition of immunotherapy following adjuvant chemotherapy improves outcomes compared to adjuvant chemotherapy alone.
What to do when a patient presents with resectable disease?
Cross-trial comparisons are fraught with danger. Until we have a phase 3 study comparing concurrent neoadjuvant chemo/immunotherapy with concurrent adjuvant chemo/immunotherapy, I do not think we can answer the question “which is better?” However, there are some caveats to keep in mind when deciding on which approach to recommend to our patients: First, neoadjuvant treatment requires biomarker testing to ensure the patient does not have EGFR or ALK mutations. This will necessitate a delay in the operation. Will patients be willing to wait? Will the surgeon? Or, would patients prefer to proceed with surgery while the results are pending? Yes, neoadjuvant therapy gives you information regarding the pCR rate, but does that help you in subsequent management of the patient? We do not know.
Secondly, the two adjuvant studies used adjuvant chemotherapy followed by adjuvant immunotherapy, as contrasted to the neoadjuvant study which used concurrent chemo/immunotherapy. Given the longer duration of treatment in postoperative sequential adjuvant studies, there tends to be more drop off because of patients being unwilling or unfit postoperatively to receive long courses of therapy. In IMpower 010, 1,269 patients completed adjuvant chemotherapy; 1,005 were randomized, and of the 507 assigned to the atezolizumab/chemo group, only 323 completed treatment.
Finally, we must beware of using neoadjuvant chemo/immunotherapy to “down-stage” a patient. KEYNOTE-091 included patients with IIIA disease and no benefit to adjuvant chemotherapy followed by immunotherapy was found in this subgroup of patients, which leads me to wonder if these patients were appropriately selected as surgical candidates. In the NADIM II trials, 9 of 29 patients on the neoadjuvant chemotherapy were not resected.
So, many questions remain. In addition to the ones we’ve raised, there is a clear and immediate need for predictive and prognostic biomarkers. In the NADIM II trial, PD-L1 expression was a predictive biomarker of response. The pCR rate for patients with a PD-L1 tumor expression of less than 1%, 1%-49%, and 50% or higher was 15%, 41.7%, and 61.1%, respectively. However, in KEYNOTE-091, the benefit was not seen in patients with PD-L1 of at least than 50%, where the 18-month DFS rate was 71.7% in the pembro arm and 70.2% in the placebo arm.
Another possible biomarker: circulating tumor DNA. In the first NADIM study, three low pretreatment levels of ctDNA were significantly associated with improved progression-free survival and overall survival (HR, 0.20 and HR, 0.07, respectively). Although clinical response did not predict survival outcomes, undetectable ctDNA levels after neoadjuvant treatment were significantly associated with progression-free survival and overall survival (HR, 0.26 and HR0.04, respectively). Similarly, in CheckMate 816, clearance of ctDNA was associated with longer EFS in patients with ctDNA clearance than in those without ctDNA clearance in both the nivolumab/chemotherapy group (HR, 0.60) and the chemotherapy-alone group (HR, 0.63).
Hopefully, ASCO 2023 will provide more answers.
Dr. Schiller is a medical oncologist and founding member of Oncologists United for Climate and Health. She is a former board member of the International Association for the Study of Lung Cancer and a current board member of the Lung Cancer Research Foundation.
Artificial intelligence colonoscopy system shows promise
A new artificial intelligence (AI) system can help expert endoscopists improve their colonoscopies, a new study indicates.
Endoscopists using the computer program SKOUT (Iterative Scopes) achieved a 27% better detection rate of adenomas per colonoscopy, compared with endoscopists working without computer assistance, said lead author Aasma Shaukat, MD, MPH, director of outcomes research in the division of gastroenterology and hepatology at New York University.
The study showed that AI colonoscopy systems can work in a routine population of U.S. patients, Dr. Shaukat said in an interview.
“As gastroenterologists, we are very excited,” she said.
The study was published online in Gastroenterology and was presented at the annual Digestive Disease® Week.
Previous research has shown that experienced endoscopists miss many polyps. To improve their detection rate, multiple companies have used machine learning to develop algorithms to identify suspicious areas.
“Once the computer sees the polyp, it puts a bounding box around it,” said Dr. Shaukat. “It draws the attention of the endoscopist to it. It assists the endoscopist but doesn’t replace the endoscopist.”
The Food and Drug Administration has approved two such systems: EndoScreener (Wision AI) and GI Genius (Cosmo Pharmaceuticals).
The SKOUT algorithm was trained on 3,616 full-length colonoscopy procedure videos from multiple centers. In bench testing, it achieved a 93.5% polyp-level true positive rate and a 2.3% false positive rate.
Randomized trial pits AI against standard procedure
To see how well the system works in the clinic, Dr. Shaukat and colleagues recruited 22 U.S. board-certified gastroenterologists from five academic and community centers. The gastroenterologists all had a minimum adenoma detection rate of 25%, defined as the number of colonoscopies in which at least one adenoma is found, divided by the number of colonoscopies performed. All the gastroenterologists had performed a minimum of 1,000 colonoscopy procedures.
The researchers randomly assigned 682 patients to undergo colonoscopy with the SKOUT and 677 to undergo colonoscopy using the standard procedure. The patients were aged 40 years or older and were scheduled for either screening or surveillance.
The endoscopists who received computer assistance detected 1.05 adenomas per colonoscopy versus 0.83 for those who did not have computer assistance, a statistically significant difference.
The proportion of resections with clinically significant histology was 71.7% with standard colonoscopies versus 67.4% with computer-assisted colonoscopies. This fell within the 14% margin that the researchers had set to show noninferiority for the computer system.
“The important thing is not just detecting all polyps but the polyps we care about, which are adenomas, and doing so without increasing the false positive rate,” said Dr. Shaukat.
The adenoma detection rate was 43.9% for the standard procedure and 47.8% for the computer-assisted procedure. This difference was not statistically significant, but Dr. Shaukat argued that the adenoma detection rate is not the best measure of success, because endoscopists sometimes stop looking for polyps once they find one.
The overall sessile serrated lesion detection rate for the standard colonoscopies was 16.0% versus 12.6% for the computer-assisted colonoscopies, which also was not statistically significant.
Next steps
This study is important because it was a large, multicenter trial in the United States, said Omer Ahmad, BSc, MBBS, MRCP, a gastroenterologist and clinical researcher at University College London, who was not involved in the study. Most of the trials of AI have been in China or Europe. “It was very important just to see this replicated in the U.S. population.”
The average procedure time was 15.41 minutes for the standard colonoscopies versus 15.82 minutes for the computer-assisted colonoscopies, which was not statistically different.
“It is important to note that the studies so far suggest that false positives do not have a significant impact on workflow,” said Dr. Ahmad.
The next crucial step in evaluating AI colonoscopy will be to track the effects over the long term, said Dr. Shaukat.
“As these technologies get approved and we see them in practice, we need to see that it’s leading to some outcome, like reduced colon cancer,” she said.
That also may be necessary before payers in the United States are willing to pay the additional cost for this technology, she added.
In the meantime, Dr. Ahmad said computer assistance is improving his own colonoscopies.
“I have found the systems have spotted some polyps that I may have otherwise missed,” he said. “There is a false positive rate, but for me, it doesn’t distract from my workflow.”
He believes the systems will be particularly helpful in improving the performance of less-skilled endoscopists.
He is also looking forward to systems that can help complete the reports needed at the end of each colonoscopy. “Most of us dislike having to write a laborious report and having to code everything at the end of the procedure,” he said.
The study was funded by Iterative Scopes. Dr. Shaukat reported having received research funding to her institution for the current study from Iterative Scopes and consulting fees from Freenome and Medtronic. Dr. Ahmad reports receiving speaker fees from the Canadian Association of Gastroenterology/Medtronic.
A version of this article first appeared on Medscape.com.
A new artificial intelligence (AI) system can help expert endoscopists improve their colonoscopies, a new study indicates.
Endoscopists using the computer program SKOUT (Iterative Scopes) achieved a 27% better detection rate of adenomas per colonoscopy, compared with endoscopists working without computer assistance, said lead author Aasma Shaukat, MD, MPH, director of outcomes research in the division of gastroenterology and hepatology at New York University.
The study showed that AI colonoscopy systems can work in a routine population of U.S. patients, Dr. Shaukat said in an interview.
“As gastroenterologists, we are very excited,” she said.
The study was published online in Gastroenterology and was presented at the annual Digestive Disease® Week.
Previous research has shown that experienced endoscopists miss many polyps. To improve their detection rate, multiple companies have used machine learning to develop algorithms to identify suspicious areas.
“Once the computer sees the polyp, it puts a bounding box around it,” said Dr. Shaukat. “It draws the attention of the endoscopist to it. It assists the endoscopist but doesn’t replace the endoscopist.”
The Food and Drug Administration has approved two such systems: EndoScreener (Wision AI) and GI Genius (Cosmo Pharmaceuticals).
The SKOUT algorithm was trained on 3,616 full-length colonoscopy procedure videos from multiple centers. In bench testing, it achieved a 93.5% polyp-level true positive rate and a 2.3% false positive rate.
Randomized trial pits AI against standard procedure
To see how well the system works in the clinic, Dr. Shaukat and colleagues recruited 22 U.S. board-certified gastroenterologists from five academic and community centers. The gastroenterologists all had a minimum adenoma detection rate of 25%, defined as the number of colonoscopies in which at least one adenoma is found, divided by the number of colonoscopies performed. All the gastroenterologists had performed a minimum of 1,000 colonoscopy procedures.
The researchers randomly assigned 682 patients to undergo colonoscopy with the SKOUT and 677 to undergo colonoscopy using the standard procedure. The patients were aged 40 years or older and were scheduled for either screening or surveillance.
The endoscopists who received computer assistance detected 1.05 adenomas per colonoscopy versus 0.83 for those who did not have computer assistance, a statistically significant difference.
The proportion of resections with clinically significant histology was 71.7% with standard colonoscopies versus 67.4% with computer-assisted colonoscopies. This fell within the 14% margin that the researchers had set to show noninferiority for the computer system.
“The important thing is not just detecting all polyps but the polyps we care about, which are adenomas, and doing so without increasing the false positive rate,” said Dr. Shaukat.
The adenoma detection rate was 43.9% for the standard procedure and 47.8% for the computer-assisted procedure. This difference was not statistically significant, but Dr. Shaukat argued that the adenoma detection rate is not the best measure of success, because endoscopists sometimes stop looking for polyps once they find one.
The overall sessile serrated lesion detection rate for the standard colonoscopies was 16.0% versus 12.6% for the computer-assisted colonoscopies, which also was not statistically significant.
Next steps
This study is important because it was a large, multicenter trial in the United States, said Omer Ahmad, BSc, MBBS, MRCP, a gastroenterologist and clinical researcher at University College London, who was not involved in the study. Most of the trials of AI have been in China or Europe. “It was very important just to see this replicated in the U.S. population.”
The average procedure time was 15.41 minutes for the standard colonoscopies versus 15.82 minutes for the computer-assisted colonoscopies, which was not statistically different.
“It is important to note that the studies so far suggest that false positives do not have a significant impact on workflow,” said Dr. Ahmad.
The next crucial step in evaluating AI colonoscopy will be to track the effects over the long term, said Dr. Shaukat.
“As these technologies get approved and we see them in practice, we need to see that it’s leading to some outcome, like reduced colon cancer,” she said.
That also may be necessary before payers in the United States are willing to pay the additional cost for this technology, she added.
In the meantime, Dr. Ahmad said computer assistance is improving his own colonoscopies.
“I have found the systems have spotted some polyps that I may have otherwise missed,” he said. “There is a false positive rate, but for me, it doesn’t distract from my workflow.”
He believes the systems will be particularly helpful in improving the performance of less-skilled endoscopists.
He is also looking forward to systems that can help complete the reports needed at the end of each colonoscopy. “Most of us dislike having to write a laborious report and having to code everything at the end of the procedure,” he said.
The study was funded by Iterative Scopes. Dr. Shaukat reported having received research funding to her institution for the current study from Iterative Scopes and consulting fees from Freenome and Medtronic. Dr. Ahmad reports receiving speaker fees from the Canadian Association of Gastroenterology/Medtronic.
A version of this article first appeared on Medscape.com.
A new artificial intelligence (AI) system can help expert endoscopists improve their colonoscopies, a new study indicates.
Endoscopists using the computer program SKOUT (Iterative Scopes) achieved a 27% better detection rate of adenomas per colonoscopy, compared with endoscopists working without computer assistance, said lead author Aasma Shaukat, MD, MPH, director of outcomes research in the division of gastroenterology and hepatology at New York University.
The study showed that AI colonoscopy systems can work in a routine population of U.S. patients, Dr. Shaukat said in an interview.
“As gastroenterologists, we are very excited,” she said.
The study was published online in Gastroenterology and was presented at the annual Digestive Disease® Week.
Previous research has shown that experienced endoscopists miss many polyps. To improve their detection rate, multiple companies have used machine learning to develop algorithms to identify suspicious areas.
“Once the computer sees the polyp, it puts a bounding box around it,” said Dr. Shaukat. “It draws the attention of the endoscopist to it. It assists the endoscopist but doesn’t replace the endoscopist.”
The Food and Drug Administration has approved two such systems: EndoScreener (Wision AI) and GI Genius (Cosmo Pharmaceuticals).
The SKOUT algorithm was trained on 3,616 full-length colonoscopy procedure videos from multiple centers. In bench testing, it achieved a 93.5% polyp-level true positive rate and a 2.3% false positive rate.
Randomized trial pits AI against standard procedure
To see how well the system works in the clinic, Dr. Shaukat and colleagues recruited 22 U.S. board-certified gastroenterologists from five academic and community centers. The gastroenterologists all had a minimum adenoma detection rate of 25%, defined as the number of colonoscopies in which at least one adenoma is found, divided by the number of colonoscopies performed. All the gastroenterologists had performed a minimum of 1,000 colonoscopy procedures.
The researchers randomly assigned 682 patients to undergo colonoscopy with the SKOUT and 677 to undergo colonoscopy using the standard procedure. The patients were aged 40 years or older and were scheduled for either screening or surveillance.
The endoscopists who received computer assistance detected 1.05 adenomas per colonoscopy versus 0.83 for those who did not have computer assistance, a statistically significant difference.
The proportion of resections with clinically significant histology was 71.7% with standard colonoscopies versus 67.4% with computer-assisted colonoscopies. This fell within the 14% margin that the researchers had set to show noninferiority for the computer system.
“The important thing is not just detecting all polyps but the polyps we care about, which are adenomas, and doing so without increasing the false positive rate,” said Dr. Shaukat.
The adenoma detection rate was 43.9% for the standard procedure and 47.8% for the computer-assisted procedure. This difference was not statistically significant, but Dr. Shaukat argued that the adenoma detection rate is not the best measure of success, because endoscopists sometimes stop looking for polyps once they find one.
The overall sessile serrated lesion detection rate for the standard colonoscopies was 16.0% versus 12.6% for the computer-assisted colonoscopies, which also was not statistically significant.
Next steps
This study is important because it was a large, multicenter trial in the United States, said Omer Ahmad, BSc, MBBS, MRCP, a gastroenterologist and clinical researcher at University College London, who was not involved in the study. Most of the trials of AI have been in China or Europe. “It was very important just to see this replicated in the U.S. population.”
The average procedure time was 15.41 minutes for the standard colonoscopies versus 15.82 minutes for the computer-assisted colonoscopies, which was not statistically different.
“It is important to note that the studies so far suggest that false positives do not have a significant impact on workflow,” said Dr. Ahmad.
The next crucial step in evaluating AI colonoscopy will be to track the effects over the long term, said Dr. Shaukat.
“As these technologies get approved and we see them in practice, we need to see that it’s leading to some outcome, like reduced colon cancer,” she said.
That also may be necessary before payers in the United States are willing to pay the additional cost for this technology, she added.
In the meantime, Dr. Ahmad said computer assistance is improving his own colonoscopies.
“I have found the systems have spotted some polyps that I may have otherwise missed,” he said. “There is a false positive rate, but for me, it doesn’t distract from my workflow.”
He believes the systems will be particularly helpful in improving the performance of less-skilled endoscopists.
He is also looking forward to systems that can help complete the reports needed at the end of each colonoscopy. “Most of us dislike having to write a laborious report and having to code everything at the end of the procedure,” he said.
The study was funded by Iterative Scopes. Dr. Shaukat reported having received research funding to her institution for the current study from Iterative Scopes and consulting fees from Freenome and Medtronic. Dr. Ahmad reports receiving speaker fees from the Canadian Association of Gastroenterology/Medtronic.
A version of this article first appeared on Medscape.com.