User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
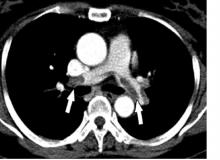
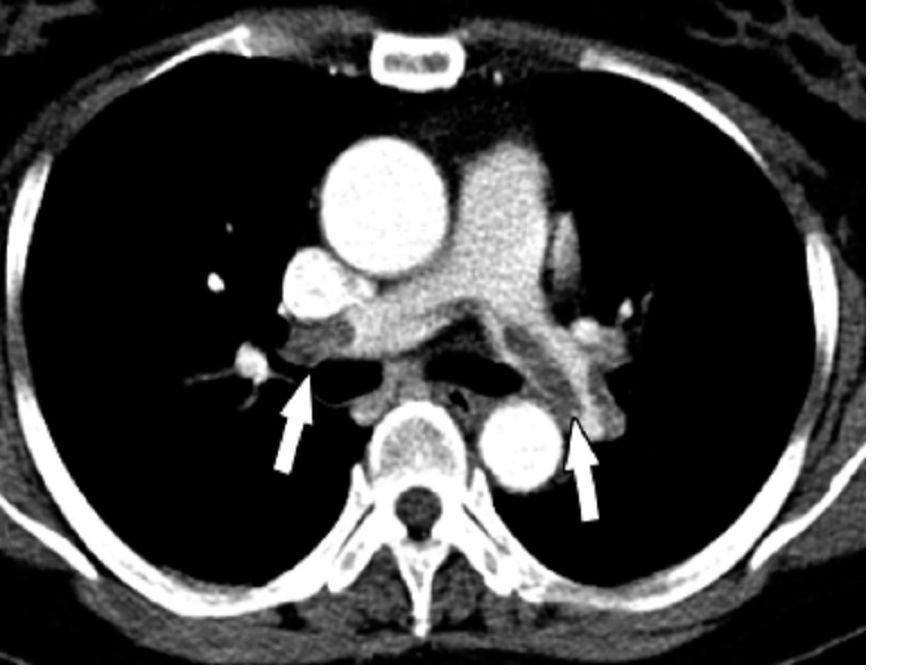
First randomized trial reassures on ACEIs, ARBs in COVID-19
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Three malpractice risks of video visits
During a telemedicine visit with his physician, a 62-year-old obese patient with an ankle injury reported new swelling of his leg. Three weeks had passed since the man visited an emergency department, where he underwent surgery and had a cast applied to the wound. The physician, during the telemedicine visit, advised the patient to elevate his leg and see an orthopedist within 24 hours. A Doppler ultrasound was ordered for 12:30 p.m. that same day.
The patient never made it to the appointment. He became unresponsive and went into full arrest hours later. His death fueled a lawsuit by his family that claimed failure to diagnose and treat deep venous thrombosis. The family contended the providers involved should have referred the patient to care immediately during the video visit.
The case, which comes from the claims database of national medical liability insurer The Doctors Company, illustrates the legal risks that can stem from video visits with patients, says Richard Cahill, JD, vice president and associate general counsel for The Doctors Company.
“By evaluating the patient remotely, the physician failed to appreciate the often subtle nuances of the clinical presentation, which undoubtedly could have been more accurately assessed in the office setting, and would probably have led to more urgent evaluation and intervention, thereby likely preventing the unfortunate and otherwise avoidable result,” said Mr. Cahill.
According to a Harris poll, 42% of Americans reported using video visits during the pandemic, a trend that is likely to continue as practices reopen and virtual care becomes the norm. But as physicians conduct more video visits, so grows their risk for lawsuits associated with the technology.
“We probably will see more malpractice suits filed the more telehealth is used,” said Mei Wa Kwong, JD, executive director of the Center for Connected Health Policy. “It’s a numbers game. The more it’s used, the higher likelihood that lawsuits occur.”
Three problems in not being able to touch the patient
1. The primary challenge with video visits “is the inability to directly observe and lay hands on the patient,” says Jonathan Einbinder, MD, assistant vice president of analytics for CRICO, a medical liability insurer based in Boston.
said Dr. Einbinder, a practicing internist.
Such incomplete pictures can lead to diagnostic errors and the potential for lawsuits, as demonstrated by a recent CRICO analysis. Of 106 telemedicine-related claims from 2014 to 2018, 66% were diagnosis related, according to the analysis of claims from CRICO’s national database. Twelve percent of the telemedicine-related claims were associated with surgical treatment, 11% were related to medical treatment, and 5% were associated with medication issues. A smaller number of claims resulted from patient monitoring, ob.gyn. care, and safety and security.
Another analysis by The Doctors Company similarly determined that diagnostic errors are the most common allegation in telemedicine-related claims. In the study of 28 telemedicine-related claims from The Doctors’ database, 71% were diagnosis related, 11% were associated with mismanagement of treatment, and 7% were related to improper management of a surgical patient. Other allegations included improper performance of treatment or procedure and improper performance of surgery.
“Because a ‘typical’ exam can’t be done, there is the potential to miss things,” said David L. Feldman, MD, chief medical officer for The Doctors Company Group. “A subtlety, perhaps a lump that can’t be seen but only felt, and only by an experienced examiner, for example, may be missed.”
2. Documentation dangers also loom, said William Sullivan, DO, JD, an emergency physician and an attorney who specializes in health care. The legal risk lies in documenting a video visit in the same way the doctor would document an in-person visit, he explained.
“Investigation into a potential lawsuit begins when there is some type of bad outcome related to medical care,” Dr. Sullivan explained. “To determine whether the lawsuit has merit, patients/attorneys review the medical records to retrospectively determine the potential cause of the bad outcome. If the documentation reflects an examination that could not have been performed, a lawyer might be more likely to pursue a case, and it would be more difficult to defend the care provided.”
Dr. Sullivan provided this example: During a video visit, a patient complains of acute onset weakness. The physician documents that the patient’s heart has a “regular rate and rhythm,” and “muscle strength is equal bilaterally.” The following day, the patient’s weakness continues, and the patient goes to the emergency department where he is diagnosed with stroke. An EKG in the ED shows that the patient is in atrial fibrillation.
“The telehealth provider would have a difficult time explaining how it was determined that the patient had normal muscle strength and a normal heart rhythm over a video visit the day before,” Dr. Sullivan said. “A lawyer in a subsequent malpractice case would present the provider as careless and would argue that if the provider had only sent the patient to the emergency department after the telehealth visit instead of documenting exam findings that couldn’t have been performed, the patient could have been successfully treated for the stroke.”
3. Poorly executed informed consent can also give rise to a lawsuit. This includes informed consent regarding the use of telehealth as the accepted modality for the visit rather than traditional on-site evaluations, as well as preprocedure informed consent.
“Inadequate and/or poorly documented informed consent can result in a claim for medical battery,” Mr. Cahill said.
A medical battery allegation refers to the alleged treatment or touching of a patient’s body without that person’s consent. As the AMA Journal of Ethics explains, a patient’s consent must be given, either expressly or implicitly, before a physician may legally “interfere” with the physical body of the patient.
Ideally, the informed consent process is undertaken during a first in-person visit, before virtual visits begin, Dr. Feldman said.
“There is a lot that a patient has to understand when a visit is done virtually, which is part of the informed consent process,” Dr. Feldman said. “The pandemic has forced some physicians to do their first visit virtually, and this makes the process of informing patients more onerous. It is not a simple matter of converting an in-person office practice to a remote office practice. The work flows are different, so there are definitely legal concerns as it relates to privacy and cybersecurity to name a few.”
Waivers may be weak protection
Since the pandemic started, a number of states have enacted emergency malpractice protections to shield health professionals from lawsuits. Some protections, such as those in Massachusetts, offer immunity to health professionals who provide general care to patients during the COVID-19 emergency, in addition to treatment of COVID-19 patients. Other protections, like those in Connecticut, apply specifically to care provided in support of the state’s pandemic response.
Whether that immunity applies both to in-person visits and video visits during the pandemic is not certain, said J. Richard Moore, JD, a medical liability defense attorney based in Indianapolis. Indiana’s immunity statute for example, does not make a specific provision for telehealth, he said.
“My best prediction is that if considered by the courts, the immunity would be applied to telehealth services, so long as they are being provided ‘in response to the emergency,’ which is the scope of the immunity,” he said. “I would not consider telehealth physicians to be either more or less protected than in-person providers.”
Regulatory scrutiny for telehealth providers has also been relaxed in response to COVID-19, but experts warn not to rely on the temporary shields for ultimate protection.
In March, the U.S. Department of Health and Human Service’s Office of Civil Rights (OCR) eased enforcement actions for noncompliance with Health Insurance Portability and Accountability Act requirements in connection with the good faith provision of telehealth during the COVID-19 health crisis. Under the notice, health providers can use popular applications such as Apple FaceTime, Facebook Messenger, Zoom, or Google Hangouts, to offer telehealth care without risk that OCR will impose fines or penalties for HIPAA violations.
But once the current health care emergency is mitigated, the waivers will likely be withdrawn, and enforcement actions will probably resume, Mr. Cahill said.
“It is recommended that, to avoid potential problems going forward, practitioners use due diligence and undertake best efforts to obey existing privacy and security requirements, including the use of technology that satisfies compliance regulations, despite the waiver by OCR,” he said.
In addition, a majority of states have relaxed state-specific rules for practicing telehealth and loosened licensure requirements during the pandemic. At least 47 states have issued waivers to alter in-state licensure requirements for telemedicine in response to COVID-19, according to the Federation of State Medical Boards. Most of the waivers allow physicians licensed in other states to provide care in states where they do not hold licenses, and some enable doctors to treat patients without first having had an in-person evaluation.
But at least for now, these are temporary changes, reminds Amy Lerman, JD, a health care attorney based in Washington, who specializes in telehealth and corporate compliance. Given the current pandemic environment, a significant concern is that physicians new to the telemedicine space are reacting only to the most recent rules established in the context of the pandemic, Ms. Lerman said.
“As previously noted, the recent developments are temporary in nature – states and various federal agencies have been pretty clear in setting this temporal boundary,” she said. “It is not advisable for providers to build telepractice models around temporary sets of rules.
“Furthermore, the recent developments are not necessarily comprehensive relative to all of the state-specific and other requirements that telemedicine providers are otherwise expected to follow, so relying only on the most recent guidance may cause providers to create telepractice models that have key gaps with respect to regulatory compliance.”
How you can avoid a lawsuit
As businesses reopen and practices resume treatments, physicians should weigh the choice between in-person care and video visits very carefully, said Joseph Kvedar, MD, president of the American Telemedicine Association and a dermatology professor at Harvard Medical School, Boston.
“We have to be very thoughtful about quality in this current phase, where we are doing what I call a hybrid model,” he said. “Some services are offered by telehealth and some require patients to come into the doctor’s office. We have to be very thoughtful about what types of care we determine to be appropriate for telehealth, and that has to be based on clinical quality. And if it is, it should follow that we’ll have low incidence of liability claims.”
Data should be at the center of that conclusion, Dr. Kvedar advises.
“Think about what data is needed to make a therapeutic or diagnostic decision,” he said. “If a health care provider can gather the information needed without touching the patient, then the provider is probably on safe, solid ground making that decision via a telehealth interaction. If the patient can come into the doctor’s office, and the provider deems it necessary to see the patient in person and touch the patient in order to make that clinical decision, then the patient should come in.”
An important step to preventing liability is also having strong telehealth systems and protocols in place and the necessary support to carry them out, said Dr. Einbinder of medical liability insurer CRICO.
For example, Dr. Einbinder, who practices in a 12-doctor internal medicine group, said when he finishes a virtual visit, he enters any orders into the electronic health record. Some of the orders will result in notifications to Dr. Einbinder if they are not executed, such as a referral appointment or a procedure that was not completed.
“I also can forward my orders to a front desk pool that is responsible for making sure things get done,” he said. “And, in our hospital system, we have good case management for complex patients and population management for a variety of chronic conditions. These represent additional safety nets.”
Another liability safeguard is sending patients a “visit summary” after each virtual visit, Dr. Sullivan said. This could be in the form of an email or a text that includes a brief template including items such as diagnosis, recommendations, follow-up, and a reminder to contact the doctor or go to the emergency department if symptoms worsen or new problems develop.
“Patients tend to remember about half of what physicians tell them and half of the information patients do remember is incorrect,” he said. “Consider a few sentences in an e-mail or text message as a substitute for the after-visit instructions from an office visit to enhance patient understanding. There are several inexpensive programs/services that allow text messages to be sent from a computer using a separate dedicated phone number and pretty much every patient has a cell phone to receive the instructions.”
Dr. Sullivan suggests having a documentation template specifically for telehealth visits. He also recommends the inclusion an “informed refusal of care” in the record when necessary. Dr. Sullivan’s wife, a family physician, has encountered several patients who fear contracting COVID-19 and who have refused her recommendations for in-person visits, he said. In such cases, he said it’s a good idea to document that the patient decided to forgo the recommendations given.
“If a patient suffers a bad outcome because of a failure to seek an in-person exam, a short note in the patient’s chart would help to establish that the lack of a follow-up physical exam was the patient’s informed decision, not due to some alleged negligence of the medical provider,” he said.
Concerning informed consent, Dr. Feldman says at a minimum physicians should discuss the following with patients:
- Names and credentials of staff participating.
- The right to stop or refuse treatment by telemedicine.
- Technology that will be used.
- Privacy and security risks.
- Technology-specific risks and permission to bill.
- Alternative care in case of an emergency or technology malfunction.
- Any state-specific requirements.
“Physicians can ensure they have a strong informed consent process during video visits by taking the time to cover these points at the beginning of the first visit, and being sure the patient understands and agrees to these,” Dr. Sullivan explained. “Ideally, this conversation can be recorded for future reference if necessary or at a minimum documented in the medical record.”
Consider these extra precautions
Mr. Cahill advises that practitioners be especially mindful of their “web-side manner” and the setting in which they are communicating with virtual patients to promote confidentiality, professionalism, and uninterrupted interactions.
“Use of a headset in a quiet home office is advisable,” he said. “Physicians must also be cognizant of their physical appearance and the background behind them when the visit includes both audio and visual capability. For ‘face-to-face’ telehealth encounters, it is recommended that a white lab jacket be worn as the appropriate attire; coat and tie are unnecessary.”
Some patients may need to be reminded of the need for confidentiality during a video visit, Mr. Moore added. Physicians are typically in a position to ensure confidentiality, but some patients may not understand how to protect their privacy on their end.
“If the physician sees on the screen or hears from an audio connection that there are other people around who may be able to overhear what is communicated, the physician probably has some responsibility to remind the patient that she or he may want to go to a more private place, close the door, etc.,” he said. “While I think a claim against a physician on this basis would be pretty weak, it is still a good practice for the physician to be cognizant of those kinds of concerns even if the patient is not.”
Finally, for physicians who set up telehealth operability during the pandemic – possibly in a hurry – consider using your actual case data to take a look backward, said Ms. Lerman, the Washington-based health care attorney. Reviewing the data can help determine whether you’re in compliance with relevant state laws, she said.
“If, for example, a provider set up telehealth operations during the pandemic and can see that most of [the] patients are based in a single state, or a small group of states, it is worthwhile to take [the] time and become familiar with the telemedicine laws in those states,” she said. “If there are modifications that need to be made, it may be easier to make them incrementally before the telehealth operations grow any larger in scope.”
A version of this article originally appeared on Medscape.com.
During a telemedicine visit with his physician, a 62-year-old obese patient with an ankle injury reported new swelling of his leg. Three weeks had passed since the man visited an emergency department, where he underwent surgery and had a cast applied to the wound. The physician, during the telemedicine visit, advised the patient to elevate his leg and see an orthopedist within 24 hours. A Doppler ultrasound was ordered for 12:30 p.m. that same day.
The patient never made it to the appointment. He became unresponsive and went into full arrest hours later. His death fueled a lawsuit by his family that claimed failure to diagnose and treat deep venous thrombosis. The family contended the providers involved should have referred the patient to care immediately during the video visit.
The case, which comes from the claims database of national medical liability insurer The Doctors Company, illustrates the legal risks that can stem from video visits with patients, says Richard Cahill, JD, vice president and associate general counsel for The Doctors Company.
“By evaluating the patient remotely, the physician failed to appreciate the often subtle nuances of the clinical presentation, which undoubtedly could have been more accurately assessed in the office setting, and would probably have led to more urgent evaluation and intervention, thereby likely preventing the unfortunate and otherwise avoidable result,” said Mr. Cahill.
According to a Harris poll, 42% of Americans reported using video visits during the pandemic, a trend that is likely to continue as practices reopen and virtual care becomes the norm. But as physicians conduct more video visits, so grows their risk for lawsuits associated with the technology.
“We probably will see more malpractice suits filed the more telehealth is used,” said Mei Wa Kwong, JD, executive director of the Center for Connected Health Policy. “It’s a numbers game. The more it’s used, the higher likelihood that lawsuits occur.”
Three problems in not being able to touch the patient
1. The primary challenge with video visits “is the inability to directly observe and lay hands on the patient,” says Jonathan Einbinder, MD, assistant vice president of analytics for CRICO, a medical liability insurer based in Boston.
said Dr. Einbinder, a practicing internist.
Such incomplete pictures can lead to diagnostic errors and the potential for lawsuits, as demonstrated by a recent CRICO analysis. Of 106 telemedicine-related claims from 2014 to 2018, 66% were diagnosis related, according to the analysis of claims from CRICO’s national database. Twelve percent of the telemedicine-related claims were associated with surgical treatment, 11% were related to medical treatment, and 5% were associated with medication issues. A smaller number of claims resulted from patient monitoring, ob.gyn. care, and safety and security.
Another analysis by The Doctors Company similarly determined that diagnostic errors are the most common allegation in telemedicine-related claims. In the study of 28 telemedicine-related claims from The Doctors’ database, 71% were diagnosis related, 11% were associated with mismanagement of treatment, and 7% were related to improper management of a surgical patient. Other allegations included improper performance of treatment or procedure and improper performance of surgery.
“Because a ‘typical’ exam can’t be done, there is the potential to miss things,” said David L. Feldman, MD, chief medical officer for The Doctors Company Group. “A subtlety, perhaps a lump that can’t be seen but only felt, and only by an experienced examiner, for example, may be missed.”
2. Documentation dangers also loom, said William Sullivan, DO, JD, an emergency physician and an attorney who specializes in health care. The legal risk lies in documenting a video visit in the same way the doctor would document an in-person visit, he explained.
“Investigation into a potential lawsuit begins when there is some type of bad outcome related to medical care,” Dr. Sullivan explained. “To determine whether the lawsuit has merit, patients/attorneys review the medical records to retrospectively determine the potential cause of the bad outcome. If the documentation reflects an examination that could not have been performed, a lawyer might be more likely to pursue a case, and it would be more difficult to defend the care provided.”
Dr. Sullivan provided this example: During a video visit, a patient complains of acute onset weakness. The physician documents that the patient’s heart has a “regular rate and rhythm,” and “muscle strength is equal bilaterally.” The following day, the patient’s weakness continues, and the patient goes to the emergency department where he is diagnosed with stroke. An EKG in the ED shows that the patient is in atrial fibrillation.
“The telehealth provider would have a difficult time explaining how it was determined that the patient had normal muscle strength and a normal heart rhythm over a video visit the day before,” Dr. Sullivan said. “A lawyer in a subsequent malpractice case would present the provider as careless and would argue that if the provider had only sent the patient to the emergency department after the telehealth visit instead of documenting exam findings that couldn’t have been performed, the patient could have been successfully treated for the stroke.”
3. Poorly executed informed consent can also give rise to a lawsuit. This includes informed consent regarding the use of telehealth as the accepted modality for the visit rather than traditional on-site evaluations, as well as preprocedure informed consent.
“Inadequate and/or poorly documented informed consent can result in a claim for medical battery,” Mr. Cahill said.
A medical battery allegation refers to the alleged treatment or touching of a patient’s body without that person’s consent. As the AMA Journal of Ethics explains, a patient’s consent must be given, either expressly or implicitly, before a physician may legally “interfere” with the physical body of the patient.
Ideally, the informed consent process is undertaken during a first in-person visit, before virtual visits begin, Dr. Feldman said.
“There is a lot that a patient has to understand when a visit is done virtually, which is part of the informed consent process,” Dr. Feldman said. “The pandemic has forced some physicians to do their first visit virtually, and this makes the process of informing patients more onerous. It is not a simple matter of converting an in-person office practice to a remote office practice. The work flows are different, so there are definitely legal concerns as it relates to privacy and cybersecurity to name a few.”
Waivers may be weak protection
Since the pandemic started, a number of states have enacted emergency malpractice protections to shield health professionals from lawsuits. Some protections, such as those in Massachusetts, offer immunity to health professionals who provide general care to patients during the COVID-19 emergency, in addition to treatment of COVID-19 patients. Other protections, like those in Connecticut, apply specifically to care provided in support of the state’s pandemic response.
Whether that immunity applies both to in-person visits and video visits during the pandemic is not certain, said J. Richard Moore, JD, a medical liability defense attorney based in Indianapolis. Indiana’s immunity statute for example, does not make a specific provision for telehealth, he said.
“My best prediction is that if considered by the courts, the immunity would be applied to telehealth services, so long as they are being provided ‘in response to the emergency,’ which is the scope of the immunity,” he said. “I would not consider telehealth physicians to be either more or less protected than in-person providers.”
Regulatory scrutiny for telehealth providers has also been relaxed in response to COVID-19, but experts warn not to rely on the temporary shields for ultimate protection.
In March, the U.S. Department of Health and Human Service’s Office of Civil Rights (OCR) eased enforcement actions for noncompliance with Health Insurance Portability and Accountability Act requirements in connection with the good faith provision of telehealth during the COVID-19 health crisis. Under the notice, health providers can use popular applications such as Apple FaceTime, Facebook Messenger, Zoom, or Google Hangouts, to offer telehealth care without risk that OCR will impose fines or penalties for HIPAA violations.
But once the current health care emergency is mitigated, the waivers will likely be withdrawn, and enforcement actions will probably resume, Mr. Cahill said.
“It is recommended that, to avoid potential problems going forward, practitioners use due diligence and undertake best efforts to obey existing privacy and security requirements, including the use of technology that satisfies compliance regulations, despite the waiver by OCR,” he said.
In addition, a majority of states have relaxed state-specific rules for practicing telehealth and loosened licensure requirements during the pandemic. At least 47 states have issued waivers to alter in-state licensure requirements for telemedicine in response to COVID-19, according to the Federation of State Medical Boards. Most of the waivers allow physicians licensed in other states to provide care in states where they do not hold licenses, and some enable doctors to treat patients without first having had an in-person evaluation.
But at least for now, these are temporary changes, reminds Amy Lerman, JD, a health care attorney based in Washington, who specializes in telehealth and corporate compliance. Given the current pandemic environment, a significant concern is that physicians new to the telemedicine space are reacting only to the most recent rules established in the context of the pandemic, Ms. Lerman said.
“As previously noted, the recent developments are temporary in nature – states and various federal agencies have been pretty clear in setting this temporal boundary,” she said. “It is not advisable for providers to build telepractice models around temporary sets of rules.
“Furthermore, the recent developments are not necessarily comprehensive relative to all of the state-specific and other requirements that telemedicine providers are otherwise expected to follow, so relying only on the most recent guidance may cause providers to create telepractice models that have key gaps with respect to regulatory compliance.”
How you can avoid a lawsuit
As businesses reopen and practices resume treatments, physicians should weigh the choice between in-person care and video visits very carefully, said Joseph Kvedar, MD, president of the American Telemedicine Association and a dermatology professor at Harvard Medical School, Boston.
“We have to be very thoughtful about quality in this current phase, where we are doing what I call a hybrid model,” he said. “Some services are offered by telehealth and some require patients to come into the doctor’s office. We have to be very thoughtful about what types of care we determine to be appropriate for telehealth, and that has to be based on clinical quality. And if it is, it should follow that we’ll have low incidence of liability claims.”
Data should be at the center of that conclusion, Dr. Kvedar advises.
“Think about what data is needed to make a therapeutic or diagnostic decision,” he said. “If a health care provider can gather the information needed without touching the patient, then the provider is probably on safe, solid ground making that decision via a telehealth interaction. If the patient can come into the doctor’s office, and the provider deems it necessary to see the patient in person and touch the patient in order to make that clinical decision, then the patient should come in.”
An important step to preventing liability is also having strong telehealth systems and protocols in place and the necessary support to carry them out, said Dr. Einbinder of medical liability insurer CRICO.
For example, Dr. Einbinder, who practices in a 12-doctor internal medicine group, said when he finishes a virtual visit, he enters any orders into the electronic health record. Some of the orders will result in notifications to Dr. Einbinder if they are not executed, such as a referral appointment or a procedure that was not completed.
“I also can forward my orders to a front desk pool that is responsible for making sure things get done,” he said. “And, in our hospital system, we have good case management for complex patients and population management for a variety of chronic conditions. These represent additional safety nets.”
Another liability safeguard is sending patients a “visit summary” after each virtual visit, Dr. Sullivan said. This could be in the form of an email or a text that includes a brief template including items such as diagnosis, recommendations, follow-up, and a reminder to contact the doctor or go to the emergency department if symptoms worsen or new problems develop.
“Patients tend to remember about half of what physicians tell them and half of the information patients do remember is incorrect,” he said. “Consider a few sentences in an e-mail or text message as a substitute for the after-visit instructions from an office visit to enhance patient understanding. There are several inexpensive programs/services that allow text messages to be sent from a computer using a separate dedicated phone number and pretty much every patient has a cell phone to receive the instructions.”
Dr. Sullivan suggests having a documentation template specifically for telehealth visits. He also recommends the inclusion an “informed refusal of care” in the record when necessary. Dr. Sullivan’s wife, a family physician, has encountered several patients who fear contracting COVID-19 and who have refused her recommendations for in-person visits, he said. In such cases, he said it’s a good idea to document that the patient decided to forgo the recommendations given.
“If a patient suffers a bad outcome because of a failure to seek an in-person exam, a short note in the patient’s chart would help to establish that the lack of a follow-up physical exam was the patient’s informed decision, not due to some alleged negligence of the medical provider,” he said.
Concerning informed consent, Dr. Feldman says at a minimum physicians should discuss the following with patients:
- Names and credentials of staff participating.
- The right to stop or refuse treatment by telemedicine.
- Technology that will be used.
- Privacy and security risks.
- Technology-specific risks and permission to bill.
- Alternative care in case of an emergency or technology malfunction.
- Any state-specific requirements.
“Physicians can ensure they have a strong informed consent process during video visits by taking the time to cover these points at the beginning of the first visit, and being sure the patient understands and agrees to these,” Dr. Sullivan explained. “Ideally, this conversation can be recorded for future reference if necessary or at a minimum documented in the medical record.”
Consider these extra precautions
Mr. Cahill advises that practitioners be especially mindful of their “web-side manner” and the setting in which they are communicating with virtual patients to promote confidentiality, professionalism, and uninterrupted interactions.
“Use of a headset in a quiet home office is advisable,” he said. “Physicians must also be cognizant of their physical appearance and the background behind them when the visit includes both audio and visual capability. For ‘face-to-face’ telehealth encounters, it is recommended that a white lab jacket be worn as the appropriate attire; coat and tie are unnecessary.”
Some patients may need to be reminded of the need for confidentiality during a video visit, Mr. Moore added. Physicians are typically in a position to ensure confidentiality, but some patients may not understand how to protect their privacy on their end.
“If the physician sees on the screen or hears from an audio connection that there are other people around who may be able to overhear what is communicated, the physician probably has some responsibility to remind the patient that she or he may want to go to a more private place, close the door, etc.,” he said. “While I think a claim against a physician on this basis would be pretty weak, it is still a good practice for the physician to be cognizant of those kinds of concerns even if the patient is not.”
Finally, for physicians who set up telehealth operability during the pandemic – possibly in a hurry – consider using your actual case data to take a look backward, said Ms. Lerman, the Washington-based health care attorney. Reviewing the data can help determine whether you’re in compliance with relevant state laws, she said.
“If, for example, a provider set up telehealth operations during the pandemic and can see that most of [the] patients are based in a single state, or a small group of states, it is worthwhile to take [the] time and become familiar with the telemedicine laws in those states,” she said. “If there are modifications that need to be made, it may be easier to make them incrementally before the telehealth operations grow any larger in scope.”
A version of this article originally appeared on Medscape.com.
During a telemedicine visit with his physician, a 62-year-old obese patient with an ankle injury reported new swelling of his leg. Three weeks had passed since the man visited an emergency department, where he underwent surgery and had a cast applied to the wound. The physician, during the telemedicine visit, advised the patient to elevate his leg and see an orthopedist within 24 hours. A Doppler ultrasound was ordered for 12:30 p.m. that same day.
The patient never made it to the appointment. He became unresponsive and went into full arrest hours later. His death fueled a lawsuit by his family that claimed failure to diagnose and treat deep venous thrombosis. The family contended the providers involved should have referred the patient to care immediately during the video visit.
The case, which comes from the claims database of national medical liability insurer The Doctors Company, illustrates the legal risks that can stem from video visits with patients, says Richard Cahill, JD, vice president and associate general counsel for The Doctors Company.
“By evaluating the patient remotely, the physician failed to appreciate the often subtle nuances of the clinical presentation, which undoubtedly could have been more accurately assessed in the office setting, and would probably have led to more urgent evaluation and intervention, thereby likely preventing the unfortunate and otherwise avoidable result,” said Mr. Cahill.
According to a Harris poll, 42% of Americans reported using video visits during the pandemic, a trend that is likely to continue as practices reopen and virtual care becomes the norm. But as physicians conduct more video visits, so grows their risk for lawsuits associated with the technology.
“We probably will see more malpractice suits filed the more telehealth is used,” said Mei Wa Kwong, JD, executive director of the Center for Connected Health Policy. “It’s a numbers game. The more it’s used, the higher likelihood that lawsuits occur.”
Three problems in not being able to touch the patient
1. The primary challenge with video visits “is the inability to directly observe and lay hands on the patient,” says Jonathan Einbinder, MD, assistant vice president of analytics for CRICO, a medical liability insurer based in Boston.
said Dr. Einbinder, a practicing internist.
Such incomplete pictures can lead to diagnostic errors and the potential for lawsuits, as demonstrated by a recent CRICO analysis. Of 106 telemedicine-related claims from 2014 to 2018, 66% were diagnosis related, according to the analysis of claims from CRICO’s national database. Twelve percent of the telemedicine-related claims were associated with surgical treatment, 11% were related to medical treatment, and 5% were associated with medication issues. A smaller number of claims resulted from patient monitoring, ob.gyn. care, and safety and security.
Another analysis by The Doctors Company similarly determined that diagnostic errors are the most common allegation in telemedicine-related claims. In the study of 28 telemedicine-related claims from The Doctors’ database, 71% were diagnosis related, 11% were associated with mismanagement of treatment, and 7% were related to improper management of a surgical patient. Other allegations included improper performance of treatment or procedure and improper performance of surgery.
“Because a ‘typical’ exam can’t be done, there is the potential to miss things,” said David L. Feldman, MD, chief medical officer for The Doctors Company Group. “A subtlety, perhaps a lump that can’t be seen but only felt, and only by an experienced examiner, for example, may be missed.”
2. Documentation dangers also loom, said William Sullivan, DO, JD, an emergency physician and an attorney who specializes in health care. The legal risk lies in documenting a video visit in the same way the doctor would document an in-person visit, he explained.
“Investigation into a potential lawsuit begins when there is some type of bad outcome related to medical care,” Dr. Sullivan explained. “To determine whether the lawsuit has merit, patients/attorneys review the medical records to retrospectively determine the potential cause of the bad outcome. If the documentation reflects an examination that could not have been performed, a lawyer might be more likely to pursue a case, and it would be more difficult to defend the care provided.”
Dr. Sullivan provided this example: During a video visit, a patient complains of acute onset weakness. The physician documents that the patient’s heart has a “regular rate and rhythm,” and “muscle strength is equal bilaterally.” The following day, the patient’s weakness continues, and the patient goes to the emergency department where he is diagnosed with stroke. An EKG in the ED shows that the patient is in atrial fibrillation.
“The telehealth provider would have a difficult time explaining how it was determined that the patient had normal muscle strength and a normal heart rhythm over a video visit the day before,” Dr. Sullivan said. “A lawyer in a subsequent malpractice case would present the provider as careless and would argue that if the provider had only sent the patient to the emergency department after the telehealth visit instead of documenting exam findings that couldn’t have been performed, the patient could have been successfully treated for the stroke.”
3. Poorly executed informed consent can also give rise to a lawsuit. This includes informed consent regarding the use of telehealth as the accepted modality for the visit rather than traditional on-site evaluations, as well as preprocedure informed consent.
“Inadequate and/or poorly documented informed consent can result in a claim for medical battery,” Mr. Cahill said.
A medical battery allegation refers to the alleged treatment or touching of a patient’s body without that person’s consent. As the AMA Journal of Ethics explains, a patient’s consent must be given, either expressly or implicitly, before a physician may legally “interfere” with the physical body of the patient.
Ideally, the informed consent process is undertaken during a first in-person visit, before virtual visits begin, Dr. Feldman said.
“There is a lot that a patient has to understand when a visit is done virtually, which is part of the informed consent process,” Dr. Feldman said. “The pandemic has forced some physicians to do their first visit virtually, and this makes the process of informing patients more onerous. It is not a simple matter of converting an in-person office practice to a remote office practice. The work flows are different, so there are definitely legal concerns as it relates to privacy and cybersecurity to name a few.”
Waivers may be weak protection
Since the pandemic started, a number of states have enacted emergency malpractice protections to shield health professionals from lawsuits. Some protections, such as those in Massachusetts, offer immunity to health professionals who provide general care to patients during the COVID-19 emergency, in addition to treatment of COVID-19 patients. Other protections, like those in Connecticut, apply specifically to care provided in support of the state’s pandemic response.
Whether that immunity applies both to in-person visits and video visits during the pandemic is not certain, said J. Richard Moore, JD, a medical liability defense attorney based in Indianapolis. Indiana’s immunity statute for example, does not make a specific provision for telehealth, he said.
“My best prediction is that if considered by the courts, the immunity would be applied to telehealth services, so long as they are being provided ‘in response to the emergency,’ which is the scope of the immunity,” he said. “I would not consider telehealth physicians to be either more or less protected than in-person providers.”
Regulatory scrutiny for telehealth providers has also been relaxed in response to COVID-19, but experts warn not to rely on the temporary shields for ultimate protection.
In March, the U.S. Department of Health and Human Service’s Office of Civil Rights (OCR) eased enforcement actions for noncompliance with Health Insurance Portability and Accountability Act requirements in connection with the good faith provision of telehealth during the COVID-19 health crisis. Under the notice, health providers can use popular applications such as Apple FaceTime, Facebook Messenger, Zoom, or Google Hangouts, to offer telehealth care without risk that OCR will impose fines or penalties for HIPAA violations.
But once the current health care emergency is mitigated, the waivers will likely be withdrawn, and enforcement actions will probably resume, Mr. Cahill said.
“It is recommended that, to avoid potential problems going forward, practitioners use due diligence and undertake best efforts to obey existing privacy and security requirements, including the use of technology that satisfies compliance regulations, despite the waiver by OCR,” he said.
In addition, a majority of states have relaxed state-specific rules for practicing telehealth and loosened licensure requirements during the pandemic. At least 47 states have issued waivers to alter in-state licensure requirements for telemedicine in response to COVID-19, according to the Federation of State Medical Boards. Most of the waivers allow physicians licensed in other states to provide care in states where they do not hold licenses, and some enable doctors to treat patients without first having had an in-person evaluation.
But at least for now, these are temporary changes, reminds Amy Lerman, JD, a health care attorney based in Washington, who specializes in telehealth and corporate compliance. Given the current pandemic environment, a significant concern is that physicians new to the telemedicine space are reacting only to the most recent rules established in the context of the pandemic, Ms. Lerman said.
“As previously noted, the recent developments are temporary in nature – states and various federal agencies have been pretty clear in setting this temporal boundary,” she said. “It is not advisable for providers to build telepractice models around temporary sets of rules.
“Furthermore, the recent developments are not necessarily comprehensive relative to all of the state-specific and other requirements that telemedicine providers are otherwise expected to follow, so relying only on the most recent guidance may cause providers to create telepractice models that have key gaps with respect to regulatory compliance.”
How you can avoid a lawsuit
As businesses reopen and practices resume treatments, physicians should weigh the choice between in-person care and video visits very carefully, said Joseph Kvedar, MD, president of the American Telemedicine Association and a dermatology professor at Harvard Medical School, Boston.
“We have to be very thoughtful about quality in this current phase, where we are doing what I call a hybrid model,” he said. “Some services are offered by telehealth and some require patients to come into the doctor’s office. We have to be very thoughtful about what types of care we determine to be appropriate for telehealth, and that has to be based on clinical quality. And if it is, it should follow that we’ll have low incidence of liability claims.”
Data should be at the center of that conclusion, Dr. Kvedar advises.
“Think about what data is needed to make a therapeutic or diagnostic decision,” he said. “If a health care provider can gather the information needed without touching the patient, then the provider is probably on safe, solid ground making that decision via a telehealth interaction. If the patient can come into the doctor’s office, and the provider deems it necessary to see the patient in person and touch the patient in order to make that clinical decision, then the patient should come in.”
An important step to preventing liability is also having strong telehealth systems and protocols in place and the necessary support to carry them out, said Dr. Einbinder of medical liability insurer CRICO.
For example, Dr. Einbinder, who practices in a 12-doctor internal medicine group, said when he finishes a virtual visit, he enters any orders into the electronic health record. Some of the orders will result in notifications to Dr. Einbinder if they are not executed, such as a referral appointment or a procedure that was not completed.
“I also can forward my orders to a front desk pool that is responsible for making sure things get done,” he said. “And, in our hospital system, we have good case management for complex patients and population management for a variety of chronic conditions. These represent additional safety nets.”
Another liability safeguard is sending patients a “visit summary” after each virtual visit, Dr. Sullivan said. This could be in the form of an email or a text that includes a brief template including items such as diagnosis, recommendations, follow-up, and a reminder to contact the doctor or go to the emergency department if symptoms worsen or new problems develop.
“Patients tend to remember about half of what physicians tell them and half of the information patients do remember is incorrect,” he said. “Consider a few sentences in an e-mail or text message as a substitute for the after-visit instructions from an office visit to enhance patient understanding. There are several inexpensive programs/services that allow text messages to be sent from a computer using a separate dedicated phone number and pretty much every patient has a cell phone to receive the instructions.”
Dr. Sullivan suggests having a documentation template specifically for telehealth visits. He also recommends the inclusion an “informed refusal of care” in the record when necessary. Dr. Sullivan’s wife, a family physician, has encountered several patients who fear contracting COVID-19 and who have refused her recommendations for in-person visits, he said. In such cases, he said it’s a good idea to document that the patient decided to forgo the recommendations given.
“If a patient suffers a bad outcome because of a failure to seek an in-person exam, a short note in the patient’s chart would help to establish that the lack of a follow-up physical exam was the patient’s informed decision, not due to some alleged negligence of the medical provider,” he said.
Concerning informed consent, Dr. Feldman says at a minimum physicians should discuss the following with patients:
- Names and credentials of staff participating.
- The right to stop or refuse treatment by telemedicine.
- Technology that will be used.
- Privacy and security risks.
- Technology-specific risks and permission to bill.
- Alternative care in case of an emergency or technology malfunction.
- Any state-specific requirements.
“Physicians can ensure they have a strong informed consent process during video visits by taking the time to cover these points at the beginning of the first visit, and being sure the patient understands and agrees to these,” Dr. Sullivan explained. “Ideally, this conversation can be recorded for future reference if necessary or at a minimum documented in the medical record.”
Consider these extra precautions
Mr. Cahill advises that practitioners be especially mindful of their “web-side manner” and the setting in which they are communicating with virtual patients to promote confidentiality, professionalism, and uninterrupted interactions.
“Use of a headset in a quiet home office is advisable,” he said. “Physicians must also be cognizant of their physical appearance and the background behind them when the visit includes both audio and visual capability. For ‘face-to-face’ telehealth encounters, it is recommended that a white lab jacket be worn as the appropriate attire; coat and tie are unnecessary.”
Some patients may need to be reminded of the need for confidentiality during a video visit, Mr. Moore added. Physicians are typically in a position to ensure confidentiality, but some patients may not understand how to protect their privacy on their end.
“If the physician sees on the screen or hears from an audio connection that there are other people around who may be able to overhear what is communicated, the physician probably has some responsibility to remind the patient that she or he may want to go to a more private place, close the door, etc.,” he said. “While I think a claim against a physician on this basis would be pretty weak, it is still a good practice for the physician to be cognizant of those kinds of concerns even if the patient is not.”
Finally, for physicians who set up telehealth operability during the pandemic – possibly in a hurry – consider using your actual case data to take a look backward, said Ms. Lerman, the Washington-based health care attorney. Reviewing the data can help determine whether you’re in compliance with relevant state laws, she said.
“If, for example, a provider set up telehealth operations during the pandemic and can see that most of [the] patients are based in a single state, or a small group of states, it is worthwhile to take [the] time and become familiar with the telemedicine laws in those states,” she said. “If there are modifications that need to be made, it may be easier to make them incrementally before the telehealth operations grow any larger in scope.”
A version of this article originally appeared on Medscape.com.
Pandemic worsens disparities in GI and liver disease
Suspension of disease screening and nonurgent procedures because of the COVID-19 pandemic will negatively impact long-term outcomes of GI and liver disease, and people of color will be disproportionately affected, according to a leading expert.
Novel, multipronged approaches are needed to overcome widening disparities in gastroenterology and hepatology, said Rachel Issaka, MD, of Fred Hutchinson Cancer Research Center in Seattle.
“The COVID-19 pandemic has led to unprecedented drops in breast, colorectal, and cervical cancer screenings,” Dr. Issaka said during an AGA FORWARD Program webinar. Screening rates for these diseases are down 83%-90%, she said.
“Certainly this creates a backlog of cancer screenings that need to occur, which poses very significant challenges for health systems as they’re adapting to this new state of health care that we have to provide,” Dr. Issaka said.
During her presentation, Dr. Issaka first addressed pandemic-related issues in colorectal cancer (CRC).
The sudden decrease in colonoscopies has already affected diagnoses, she said, as 32% fewer cases of CRC were diagnosed in April 2020 compared with April 2019, a finding that is “obviously very concerning.” All downstream effects remain to be seen; however, one estimate suggests that over the next decade, delayed screening may lead to an additional 4,500 deaths from CRC.
“These effects are particularly noticeable in medically underserved communities where CRC morbidity and mortality are highest,” Dr. Issaka wrote, as coauthor of a study published in Gastrointestinal Endoscopy.
Dr. Issaka and colleagues predict that the pandemic will likely worsen “persistent CRC disparities” in African-American and Hispanic communities, including relatively decreased screening participation, delayed follow-up of abnormal stool results, limited community-based research and partnerships, and limited community engagement and advocacy.
“COVID-19 related pauses in medical care, as well as shifts in resource allocation and workforce deployment, threaten decades worth of work to improve CRC disparities in medically underserved populations,” wrote Dr. Issaka and colleagues.
Dr. Issaka described similar issues in hepatology. She referred to a recent opinion article by Tapper and colleagues, which predicted that the COVID-19 pandemic will impact patients with liver disease in three waves: first, by delaying liver transplants, elective procedures, imaging, and routine patient follow-up; second, by increasing emergent decompensations, transplant wait-list dropouts, and care deferrals; and third, by losing patients to follow-up, resulting in missed diagnoses, incomplete cancer screening, and progressive disease.
“This could disproportionately impact Black, Hispanic, and Native-American populations, who may have already had difficulty accessing [liver care],” Dr. Issaka said.
To mitigate growing disparities, Dr. Issaka proposed a variety of strategies for CRC and liver disease.
For CRC screening, Dr. Issaka suggested noninvasive modalities, including mailed fecal immunochemical tests (FIT), with focused follow-up on patients with highest FIT values. For those conducting CRC research, Dr. Issaka recommended using accessible technology, engaging with community partners, providing incentives where appropriate, and other methods. For cirrhosis care, Dr. Issaka suggested that practitioners turn to telehealth and remote care, including weight monitoring, cognitive function testing, home medication delivery, and online education.
More broadly, Dr. Issaka called for universal health insurance not associated with employment, research funding for health disparities, sustainable employment wages, climate justice, desegregation of housing, and universal broadband Internet.
“The solutions to these problems are multipronged,” Dr. Issaka said. “Some will happen locally; for instance, well-executed planning around telehealth. Some will happen at the state level through opportunities like advocacy or even just reaching out to your own [congressional representative]. And then some will also happen programmatically – How can we as a health system begin to leverage something like mailed FIT?”
Finally, Dr. Issaka suggested that tools from another branch of science can help improve screening rates.
“We don’t, in medicine, tap into the benefits of behavioral psychology enough,” she said. “That’s a great discipline with really great tools that we can all use.”
Dr. Issaka described the power of community, in that people are more likely to undergo screening if they know how many others in their community are also being screened.
“I think as much as we can gather those kinds of data and share those with individuals to provide reassurance about the safety and importance of screening, I think [that] will help,” she said.
The AGA FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (DK118761). Dr. Issaka has no conflicts of interest.
SOURCES: Issaka. AGA FORWARD Program Webinar. 2020 Aug 27; Balzora et al. Gastrointestinal Endoscopy. 2020 June 20. doi: 10.1016/j.gie.2020.06.042; Tapper et al. Journal of Hepatology. 2020 Apr 13. doi: 10.1016/j.jhep.2020.04.005.
Suspension of disease screening and nonurgent procedures because of the COVID-19 pandemic will negatively impact long-term outcomes of GI and liver disease, and people of color will be disproportionately affected, according to a leading expert.
Novel, multipronged approaches are needed to overcome widening disparities in gastroenterology and hepatology, said Rachel Issaka, MD, of Fred Hutchinson Cancer Research Center in Seattle.
“The COVID-19 pandemic has led to unprecedented drops in breast, colorectal, and cervical cancer screenings,” Dr. Issaka said during an AGA FORWARD Program webinar. Screening rates for these diseases are down 83%-90%, she said.
“Certainly this creates a backlog of cancer screenings that need to occur, which poses very significant challenges for health systems as they’re adapting to this new state of health care that we have to provide,” Dr. Issaka said.
During her presentation, Dr. Issaka first addressed pandemic-related issues in colorectal cancer (CRC).
The sudden decrease in colonoscopies has already affected diagnoses, she said, as 32% fewer cases of CRC were diagnosed in April 2020 compared with April 2019, a finding that is “obviously very concerning.” All downstream effects remain to be seen; however, one estimate suggests that over the next decade, delayed screening may lead to an additional 4,500 deaths from CRC.
“These effects are particularly noticeable in medically underserved communities where CRC morbidity and mortality are highest,” Dr. Issaka wrote, as coauthor of a study published in Gastrointestinal Endoscopy.
Dr. Issaka and colleagues predict that the pandemic will likely worsen “persistent CRC disparities” in African-American and Hispanic communities, including relatively decreased screening participation, delayed follow-up of abnormal stool results, limited community-based research and partnerships, and limited community engagement and advocacy.
“COVID-19 related pauses in medical care, as well as shifts in resource allocation and workforce deployment, threaten decades worth of work to improve CRC disparities in medically underserved populations,” wrote Dr. Issaka and colleagues.
Dr. Issaka described similar issues in hepatology. She referred to a recent opinion article by Tapper and colleagues, which predicted that the COVID-19 pandemic will impact patients with liver disease in three waves: first, by delaying liver transplants, elective procedures, imaging, and routine patient follow-up; second, by increasing emergent decompensations, transplant wait-list dropouts, and care deferrals; and third, by losing patients to follow-up, resulting in missed diagnoses, incomplete cancer screening, and progressive disease.
“This could disproportionately impact Black, Hispanic, and Native-American populations, who may have already had difficulty accessing [liver care],” Dr. Issaka said.
To mitigate growing disparities, Dr. Issaka proposed a variety of strategies for CRC and liver disease.
For CRC screening, Dr. Issaka suggested noninvasive modalities, including mailed fecal immunochemical tests (FIT), with focused follow-up on patients with highest FIT values. For those conducting CRC research, Dr. Issaka recommended using accessible technology, engaging with community partners, providing incentives where appropriate, and other methods. For cirrhosis care, Dr. Issaka suggested that practitioners turn to telehealth and remote care, including weight monitoring, cognitive function testing, home medication delivery, and online education.
More broadly, Dr. Issaka called for universal health insurance not associated with employment, research funding for health disparities, sustainable employment wages, climate justice, desegregation of housing, and universal broadband Internet.
“The solutions to these problems are multipronged,” Dr. Issaka said. “Some will happen locally; for instance, well-executed planning around telehealth. Some will happen at the state level through opportunities like advocacy or even just reaching out to your own [congressional representative]. And then some will also happen programmatically – How can we as a health system begin to leverage something like mailed FIT?”
Finally, Dr. Issaka suggested that tools from another branch of science can help improve screening rates.
“We don’t, in medicine, tap into the benefits of behavioral psychology enough,” she said. “That’s a great discipline with really great tools that we can all use.”
Dr. Issaka described the power of community, in that people are more likely to undergo screening if they know how many others in their community are also being screened.
“I think as much as we can gather those kinds of data and share those with individuals to provide reassurance about the safety and importance of screening, I think [that] will help,” she said.
The AGA FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (DK118761). Dr. Issaka has no conflicts of interest.
SOURCES: Issaka. AGA FORWARD Program Webinar. 2020 Aug 27; Balzora et al. Gastrointestinal Endoscopy. 2020 June 20. doi: 10.1016/j.gie.2020.06.042; Tapper et al. Journal of Hepatology. 2020 Apr 13. doi: 10.1016/j.jhep.2020.04.005.
Suspension of disease screening and nonurgent procedures because of the COVID-19 pandemic will negatively impact long-term outcomes of GI and liver disease, and people of color will be disproportionately affected, according to a leading expert.
Novel, multipronged approaches are needed to overcome widening disparities in gastroenterology and hepatology, said Rachel Issaka, MD, of Fred Hutchinson Cancer Research Center in Seattle.
“The COVID-19 pandemic has led to unprecedented drops in breast, colorectal, and cervical cancer screenings,” Dr. Issaka said during an AGA FORWARD Program webinar. Screening rates for these diseases are down 83%-90%, she said.
“Certainly this creates a backlog of cancer screenings that need to occur, which poses very significant challenges for health systems as they’re adapting to this new state of health care that we have to provide,” Dr. Issaka said.
During her presentation, Dr. Issaka first addressed pandemic-related issues in colorectal cancer (CRC).
The sudden decrease in colonoscopies has already affected diagnoses, she said, as 32% fewer cases of CRC were diagnosed in April 2020 compared with April 2019, a finding that is “obviously very concerning.” All downstream effects remain to be seen; however, one estimate suggests that over the next decade, delayed screening may lead to an additional 4,500 deaths from CRC.
“These effects are particularly noticeable in medically underserved communities where CRC morbidity and mortality are highest,” Dr. Issaka wrote, as coauthor of a study published in Gastrointestinal Endoscopy.
Dr. Issaka and colleagues predict that the pandemic will likely worsen “persistent CRC disparities” in African-American and Hispanic communities, including relatively decreased screening participation, delayed follow-up of abnormal stool results, limited community-based research and partnerships, and limited community engagement and advocacy.
“COVID-19 related pauses in medical care, as well as shifts in resource allocation and workforce deployment, threaten decades worth of work to improve CRC disparities in medically underserved populations,” wrote Dr. Issaka and colleagues.
Dr. Issaka described similar issues in hepatology. She referred to a recent opinion article by Tapper and colleagues, which predicted that the COVID-19 pandemic will impact patients with liver disease in three waves: first, by delaying liver transplants, elective procedures, imaging, and routine patient follow-up; second, by increasing emergent decompensations, transplant wait-list dropouts, and care deferrals; and third, by losing patients to follow-up, resulting in missed diagnoses, incomplete cancer screening, and progressive disease.
“This could disproportionately impact Black, Hispanic, and Native-American populations, who may have already had difficulty accessing [liver care],” Dr. Issaka said.
To mitigate growing disparities, Dr. Issaka proposed a variety of strategies for CRC and liver disease.
For CRC screening, Dr. Issaka suggested noninvasive modalities, including mailed fecal immunochemical tests (FIT), with focused follow-up on patients with highest FIT values. For those conducting CRC research, Dr. Issaka recommended using accessible technology, engaging with community partners, providing incentives where appropriate, and other methods. For cirrhosis care, Dr. Issaka suggested that practitioners turn to telehealth and remote care, including weight monitoring, cognitive function testing, home medication delivery, and online education.
More broadly, Dr. Issaka called for universal health insurance not associated with employment, research funding for health disparities, sustainable employment wages, climate justice, desegregation of housing, and universal broadband Internet.
“The solutions to these problems are multipronged,” Dr. Issaka said. “Some will happen locally; for instance, well-executed planning around telehealth. Some will happen at the state level through opportunities like advocacy or even just reaching out to your own [congressional representative]. And then some will also happen programmatically – How can we as a health system begin to leverage something like mailed FIT?”
Finally, Dr. Issaka suggested that tools from another branch of science can help improve screening rates.
“We don’t, in medicine, tap into the benefits of behavioral psychology enough,” she said. “That’s a great discipline with really great tools that we can all use.”
Dr. Issaka described the power of community, in that people are more likely to undergo screening if they know how many others in their community are also being screened.
“I think as much as we can gather those kinds of data and share those with individuals to provide reassurance about the safety and importance of screening, I think [that] will help,” she said.
The AGA FORWARD program is funded by the National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health (DK118761). Dr. Issaka has no conflicts of interest.
SOURCES: Issaka. AGA FORWARD Program Webinar. 2020 Aug 27; Balzora et al. Gastrointestinal Endoscopy. 2020 June 20. doi: 10.1016/j.gie.2020.06.042; Tapper et al. Journal of Hepatology. 2020 Apr 13. doi: 10.1016/j.jhep.2020.04.005.
FROM THE AGA FORWARD PROGRAM
FDA OKs new ‘artificial pancreas’ Medtronic 770G
The Food and Drug Administration has approved the MiniMed 770G (Medtronic) automated insulin delivery system for children aged 2-6 years.
The 770G system adds Bluetooth smartphone connectivity to the SmartGuard technology that is present in the hybrid closed-loop MiniMed 670G system, which has been available in the United States since 2016 for individuals aged 14 years and older who have type 1 diabetes. It has been available since 2018 for children aged 7 years.
The 770G will also be available to older children and adults once it has been launched.
As with other so-called artificial pancreas systems, the 770G is made up of an insulin pump and continuous glucose monitor that are connected via software that allows the pump to deliver or withhold insulin on the basis of glucose readings.
It is a “hybrid closed-loop” system in that users or caregivers must still manually signal carbohydrate consumption.
The 770G includes a “share” feature that allows health care providers, users, and caregivers to follow the user’s glucose levels remotely via smartphones. In-app notices indicate when glucose levels are out of range. The data can be uploaded prior to telehealth visits.
The approval was based on a 3-month study of 151 children aged 2-6 years who showed improvement in outcomes comparable with those seen in 124 older adolescents and adults with the 770G system as compared to patients who used manual (nonlooped) mode over a 2-week period. There were no episodes of severe hypoglycemia or diabetic ketoacidosis and no serious device-related adverse events while in hybrid closed-loop mode.
The FDA will require Medtronic to conduct a postmarketing study to evaluate the 770G in real-world settings. It is not approved for use in children younger than 2 years nor in any patient who requires less than 8 units of insulin per day.
The next-generation Medtronic closed-loop system, the 780G, has already been approved in Europe. It improves on the technology by delivering automated bolus correction doses in addition to basal insulin every 5 minutes. The company is preparing to submit the 780G for approval in the United States.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has approved the MiniMed 770G (Medtronic) automated insulin delivery system for children aged 2-6 years.
The 770G system adds Bluetooth smartphone connectivity to the SmartGuard technology that is present in the hybrid closed-loop MiniMed 670G system, which has been available in the United States since 2016 for individuals aged 14 years and older who have type 1 diabetes. It has been available since 2018 for children aged 7 years.
The 770G will also be available to older children and adults once it has been launched.
As with other so-called artificial pancreas systems, the 770G is made up of an insulin pump and continuous glucose monitor that are connected via software that allows the pump to deliver or withhold insulin on the basis of glucose readings.
It is a “hybrid closed-loop” system in that users or caregivers must still manually signal carbohydrate consumption.
The 770G includes a “share” feature that allows health care providers, users, and caregivers to follow the user’s glucose levels remotely via smartphones. In-app notices indicate when glucose levels are out of range. The data can be uploaded prior to telehealth visits.
The approval was based on a 3-month study of 151 children aged 2-6 years who showed improvement in outcomes comparable with those seen in 124 older adolescents and adults with the 770G system as compared to patients who used manual (nonlooped) mode over a 2-week period. There were no episodes of severe hypoglycemia or diabetic ketoacidosis and no serious device-related adverse events while in hybrid closed-loop mode.
The FDA will require Medtronic to conduct a postmarketing study to evaluate the 770G in real-world settings. It is not approved for use in children younger than 2 years nor in any patient who requires less than 8 units of insulin per day.
The next-generation Medtronic closed-loop system, the 780G, has already been approved in Europe. It improves on the technology by delivering automated bolus correction doses in addition to basal insulin every 5 minutes. The company is preparing to submit the 780G for approval in the United States.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has approved the MiniMed 770G (Medtronic) automated insulin delivery system for children aged 2-6 years.
The 770G system adds Bluetooth smartphone connectivity to the SmartGuard technology that is present in the hybrid closed-loop MiniMed 670G system, which has been available in the United States since 2016 for individuals aged 14 years and older who have type 1 diabetes. It has been available since 2018 for children aged 7 years.
The 770G will also be available to older children and adults once it has been launched.
As with other so-called artificial pancreas systems, the 770G is made up of an insulin pump and continuous glucose monitor that are connected via software that allows the pump to deliver or withhold insulin on the basis of glucose readings.
It is a “hybrid closed-loop” system in that users or caregivers must still manually signal carbohydrate consumption.
The 770G includes a “share” feature that allows health care providers, users, and caregivers to follow the user’s glucose levels remotely via smartphones. In-app notices indicate when glucose levels are out of range. The data can be uploaded prior to telehealth visits.
The approval was based on a 3-month study of 151 children aged 2-6 years who showed improvement in outcomes comparable with those seen in 124 older adolescents and adults with the 770G system as compared to patients who used manual (nonlooped) mode over a 2-week period. There were no episodes of severe hypoglycemia or diabetic ketoacidosis and no serious device-related adverse events while in hybrid closed-loop mode.
The FDA will require Medtronic to conduct a postmarketing study to evaluate the 770G in real-world settings. It is not approved for use in children younger than 2 years nor in any patient who requires less than 8 units of insulin per day.
The next-generation Medtronic closed-loop system, the 780G, has already been approved in Europe. It improves on the technology by delivering automated bolus correction doses in addition to basal insulin every 5 minutes. The company is preparing to submit the 780G for approval in the United States.
A version of this article originally appeared on Medscape.com.
Obesity boosts risks in COVID-19 from diagnosis to death
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
A new analysis of existing research confirms a stark link between excess weight and COVID-19:
Obese patients faced the greatest bump in risk on the hospitalization front, with their odds of being admitted listed as 113% higher. The odds of diagnosis, ICU admission, and death were 46% higher (odds ratio [OR], 1.46; 95% confidence interval [CI], 1.30-1.65; P < .0001); 74% higher (OR, 1.74, CI, 1.46-2.08, P < .0001); 48% (OR, 1.48, CI, 1.22–1.80, P < .001, all pooled analyses and 95% CI), respectively. All differences were highly significantly different, investigators reported in a systematic review and meta-analysis published online Aug. 26 in Obesity Reviews.
“Essentially, these are pretty scary statistics,” nutrition researcher and study lead author Barry M. Popkin, PhD, of the University of North Carolina at Chapel Hill School of Public Health, said in an interview. “Other studies have talked about an increase in mortality, and we were thinking there’d be a little increase like 10% – nothing like 48%.”
According to the Johns Hopkins University of Medicine tracker, nearly 6 million people in the United States had been diagnosed with COVID-19 as of Aug. 30. The number of deaths had surpassed 183,000.
The authors of the new review launched their project to better understand the link between obesity and COVID-19 “all the way from being diagnosed to death,” Dr. Popkin said, adding that the meta-analysis is the largest of its kind to examine the link.
Dr. Popkin and colleagues analyzed 75 studies during January to June 2020 that tracked 399,461 patients (55% of whom were male) diagnosed with COVID-19. They found that 18 of 20 studies linked obesity with a 46% higher risk of diagnosis, but Dr. Popkin cautioned that this may be misleading. “I suspect it’s because they’re sicker and getting tested more for COVID,” he said. “I don’t think obesity enhances your likelihood of getting COVID. We don’t have a biological rationale for that.”
The researchers examined 19 studies that explored a link between obesity and hospitalization; all 19 found a higher risk of hospitalization in patients with obesity (pooled OR, 2.13). Twenty-one of 22 studies that looked at ICU admissions discovered a higher risk for patients with obesity (pooled OR, 1.74). And 27 of 35 studies that examined COVID-19 mortality found a higher death rate in patients with obesity (pooled OR, 1.48).
The review also looked at 14 studies that examined links between obesity and administration of invasive mechanical ventilation. All the studies found a higher risk for patients with obesity (pooled OR, 1.66; 95% CI, 1.38-1.99; P < .0001).
Could socioeconomic factors explain the difference in risk for people with obesity? It’s not clear. According to Dr. Popkin, most of the studies don’t examine factors such as income. While he believes physical factors are the key to the higher risk, he said “there’s clearly a social side to this.”
On the biological front, it appears that “the immune system is much weaker if you’re obese,” he said, and excess weight may worsen the course of a respiratory disease such as COVID-19 because of lung disorders such as sleep apnea.
In addition to highlighting inflammation and a weakened immune system, the review offers multiple explanations for why patients with obesity face worse outcomes in COVID-19. It may be more difficult for medical professionals to care for them in the hospital because of their weight, the authors wrote, and “obesity may also impair therapeutic treatments during COVID-19 infections.” The authors noted that ACE inhibitors may worsen COVID-19 in patients with type 2 diabetes.
The researchers noted that “potentially the vaccines developed to address COVID-19 will be less effective for individuals with obesity due to a weakened immune response.” They pointed to research that suggests T-cell responses are weaker and antibody titers wane at a faster rate in people with obesity who are vaccinated against influenza.
Pulmonologist Joshua L. Denson, MD, MS, of Tulane University, New Orleans, praised the review in an interview, but noted that some of the included studies have wide confidence intervals. One study that links COVID-19 to a sixfold higher mortality rate (OR, 6.29) has a confidence interval of 1.76-22.45.
Dr. Denson said he’s seen about 100 patients with COVID-19, and many are obese and have metabolic syndrome.
Like the authors of the study, he believes higher levels of inflammation play a crucial role in making these patients more vulnerable. “For whatever reason, the virus tends to really like that state. That’s driving these people to get sick,” he said.
Moving forward, Dr. Popkin urged physicians to redouble their efforts to warn patients about the risks of obesity and the importance of healthy eating. He also said COVID-19 vaccine researchers must stratify obese vs. nonobese subjects in clinical trials.
The review was funded by Bloomberg Philanthropies, the Carolina Population Center, World Bank, and Saudi Health Council. The review authors report no relevant disclosures. Dr. Denson reports no relevant disclosures.
SOURCE: Popkin BM et al. Obes Rev. 2020 Aug 26. doi: 10.1111/obr.13128.
FROM OBESITY REVIEWS
HOME-PE trial clarifies which pulmonary embolism patients to treat at home
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
The pragmatic Hestia criteria proved as safe as the more structured, points-based simplified Pulmonary Embolism Severity Index (sPESI) score for selection of patients with acute pulmonary embolism for outpatient care in the large, randomized HOME-PE trial presented at the virtual annual congress of the European Society of Cardiology.
“These results support outpatient management of acute pulmonary embolism patients using either the Hestia method or the sPESI score with the option for the physician-in-charge to override the decision. In hospitals organized for outpatient management, both triaging strategies enable more than a third of pulmonary embolism patients to be managed at home with a low rate of complications,” Pierre-Marie Roy, MD, said in presenting the HOME-PE findings.
The study clarifies a transatlantic controversy regarding how best to triage patients with acute pulmonary embolism (PE) for outpatient care. The answer? It’s basically a tie between the points-based sPESI score recommended in the current ESC guidelines (Eur Respir J. 2019 Oct 9;54[3]:1901647) and the Hestia method endorsed in the American College of Chest Physician guidelines (Chest. 2016 Feb;149[2]:315-52).
The sPESI is a validated tool that grants 1 point each for age over 80 years, background cardiopulmonary disease, a systolic blood pressure below 100 mm Hg, cancer, a heart rate of 110 bpm or more, and an oxygen saturation level below 90%. A patient needs a score of zero to be eligible for outpatient management. In contrast, the Hestia method relies upon 11 simple bedside criteria rather than a points system, explained Dr. Roy of University Hospital of Angers, France (J Thromb Haemost. 2011 Aug;9[8]:1500-7).
HOME-PE was a randomized, open-label, noninferiority trial conducted at 26 hospitals in France, Belgium, Switzerland, and the Netherlands. The study included 1,974 patients presenting to the emergency department with non–high-risk acute PE as defined by hemodynamic stability. About 39% of patients in the Hestia group were eligible for outpatient care on the basis of ‘no’ answers regarding all 11 criteria, while 48% of patients had an sPESI score of 0 and were thus initially considered appropriate for outpatient management.
However, the investigators recognized that no scoring system for acute PE is perfect, and that the judgment of a physician with extensive experience in managing this life-threatening condition counts for a lot. So they stipulated that a patient’s physician-in-charge could overrule a decision for early discharge. This happened 29% of the time in patients with a sPESI score of 0, as compared with a 3% overrule rate with the Hestia rule. The physician-in-charge also moved small numbers of patients who were Hestia or sPESI positive into the outpatient care group. As a result, a similar proportion of patients in both groups were discharged home within 24 hours for outpatient treatment: 38% of the total Hestia group and 37% in the sPESI arm.
Major adverse event rates were reassuringly low in both groups managed on an outpatient basis. The composite of recurrent venous thromboembolism, bleeding, or death within 30 days occurred in 1.3% of Hestia outpatients and 1.1% of sPESI outpatients. Among patients managed in the hospital, these rates were 5.6% in the Hestia group and 4.7% in the sPESI group.
Discussant Stavros V. Konstantinides, MD, who chaired the ESC guideline committee, asked rhetorically, “who’s happy with the HOME-PE trial? I think everybody.”
“The Hestia criteria integrate the feasibility of family support of the individual patient. This is a good thing. And eligibility based on the Hestia criteria, unlike sPESI, does not require age younger than 80 years or no cancer, and it appears from the HOME-PE study that this is okay,” observed Dr. Konstantinides of the Center for Thrombosis and Hemostasis at the University of Mainz (Germany).
In an interview, Hadley Wilson, MD, called the HOME-PE trial “transformative” and predicted it will change clinical practice. He was particularly impressed with the high quality of the trial, noting that 87% of participants managed as outpatients received a direct oral anticoagulant.
The Hestia rule is simpler and more user-friendly. And greater use of this triaging strategy might have advantages in terms of economics and health care utilization by potentially encouraging movement of decision-making regarding outpatient management of acute PE out of the hospital wards and into emergency departments, said Dr. Wilson, executive vice chair of the Sanger Heart and Vascular Institute and a cardiologist at the University of North Carolina at Chapel Hill.
Dr. Roy reported receiving research grants to conduct HOME-PE from the French Ministry of Health, the study sponsor. In addition, he is on scientific advisory boards and/or speakers’ panels for Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Pfizer, Aspen, Daiichi Sankyo, and Sanofi Aventis.
REPORTING FROM ESC CONGRESS 2020
ESC’s revised NSTE-ACS guidelines embrace hsT, personalized anti-ischemia treatments
The first revision since 2015 to the European Society of Cardiology’s guidelines for diagnosing and managing non ST-elevation acute coronary syndrome placed increased reliance on high-sensitivity cardiac troponin testing for diagnosis, and embraced coronary CT to rule out lower-risk patients.
It also highlighted the need for personalized antiplatelet regimens, systems of care, and quality improvement.
The society released the new guidelines on August 29 (Eur Heart J. 2020 Aug 29;doi: 10.1093/eurheartj/ehaa575), and then devoted a session to them the next day at the virtual annual congress of the European Society of Cardiology to highlight some of the key updates, starting with the further emphasis placed on high-sensitivity cardiac troponin (hs-cTn), a reliance that contrasts with what remains inconsistent use of this metric in U.S. practice.
An hs-cTn test is the “preferred” diagnostic test and a “key” testing element, said Marco Roffi, MD, professor and director of interventional cardiology at University Hospital, Geneva, and a member of the guideline committee. He also stressed an update to the time frame of initial hs-cTn testing, which now involves a baseline reading and then a second measure after 2 hours to discern how the marker level is evolving with time. The guidelines advise against measuring any other biomarker of myocardial injury.
U.S. lags in measuring high-sensitivity cardiac troponin
U.S. medical systems and centers “are not uniform in adopting hs-cTn,” noted Richard J. Kovacs, MD, professor of cardiology at the Indiana University School of Medicine in Indianapolis. “The new European guidelines should spur U.S. institutions to at least take a close look at the advantages of hs-cTn. There is a strong case that it leads to more efficient emergency care and allows for quicker decisions and triage,” added Dr. Kovacs in an interview.
The new guideline’s emphasis on hs-cTn should hasten broader uptake in U.S. practice, agreed Deepak L. Bhatt, MD, professor of medicine at Harvard Medical School in Boston and a member of the guideline-writing panel
Another plus of the guidelines is its endorsement of an “organized approach to risk assessment” early on in these patients, said Dr. Kovacs, who is also the immediate past-president of the American College of Cardiology (ACC). An ACC committee is developing a new set of recommendations for managing patients with cardiac chest pain and is on track for release in 2021. It would represent the first update to U.S. guidelines for non ST-elevation ACS patients since 2014.
The new ESC guidelines give coronary CT angiography a class Ia rating as an alternative to invasive coronary angiography for assessing patients with a low or intermediate risk of having coronary disease, a “tremendous upgrade,” commented Ashish Pershad, MD, an interventional cardiologist at Banner-University Medical Heart Institute in Phoenix. While he welcomes this support for using coronary CT angiography in this setting, he acknowledged that the method remains limited in availability as it requires highly trained technicians to obtain good images and experienced clinicians to interpret the results.
Personalizing antiplatelet and antithrombotic treatments
Notable revisions to medical treatments to minimize ischemia included an admonition not to use routine pretreatment with a P2Y12 receptor inhibitor (such as clopidogrel) before testing determines coronary anatomy.
Not using one of these antiplatelet drugs upfront on all patients “is a tremendous change,” Dr. Pershad said in an interview. Many patients currently get these drugs while awaiting an angiogram, but a more selected and deferred antiplatelet approach would be better when angiography shows that some patients need coronary bypass surgery, he noted. Recent study results have shown no added benefit from pretreatment, and its use can be especially problematic for patients who are slated for a planned invasive strategy, said Dr. Bhatt.
Dr. Pershad, Dr. Bhatt, and Dr. Kovacs all praised the overall emphasis on a personalized approach to treating patients with antiplatelet and antithrombotic drugs, with endorsement of a flexible approach to treatment intensity and duration. The guideline acknowledges the need to take into account a patient’s bleeding risk and comorbidities, and specifically endorsed the Academic Research Consortium’s formula for identifying and stratifying high bleeding risk (Eur Heart J. 2019 Aug 14;40[31]:2632-53).
The new guidelines also provide guidance on how to apply recent study results that addressed balancing efficacy and safety when pairing an antiplatelet drug with a direct-acting oral anticoagulant (DOAC) for patients who potentially need both drug classes, such as patients with atrial fibrillation and a recent ACS event. “It’s tremendous to get clarity on this issue; there’s been a lot of uncertainty,” said Dr. Pershad. The guidelines call for a week of triple therapy with a DOAC, aspirin, and a second antiplatelet drug, followed by 12 months on a DOAC plus a single antiplatelet drug, and then the DOAC alone as the “default” strategy for most patients, but also presents alternative options for patients with high risk for either bleeding or ischemia.
The new guidelines also give much-needed direction on how to apply an invasive strategy, with an emphasis on immediate intervention for within 2 hours for very-high-risk patients, and early intervention within 24 hours for high-risk patients. Adhering to this timetable can mean increasing catheter laboratory availability on an urgent basis over weekends, Dr. Bhatt noted.
Improving quality of care
A novel section in the new guidelines was devoted to nine quality measures that can help health systems and medical centers monitor their adherence to the guideline recommendations, track their performance relative to peer institutions, and follow changes in performance that result from quality improvement steps. It’s something of a “futuristic” step for a guideline to take, with a goal of persuading administrators to implement tracking of these measures and improve patient outcomes, noted Dr. Bhatt.
“It’s very important to see that this is not just a set of guidelines but also a tool to improve quality of care,” commented Dr. Kovacs. The key to success in this effort will be to follow registered patients, set benchmarks that systems can aspire to achieve, and use this to improve the quality of care.
Until now, optimizing care for patients with NSTE-ACS has been “challenging,” he said. “The focus must be on moving toward systems of care” that can provide consistent patient evaluation and care, and do it quickly, said Dr. Kovacs.
Dr. Roffi has received research funding from Biotronik, Boston Scientific, GE Healthcare, and Medtronic. Dr. Kovacs was formerly an employee of Eli Lilly. Dr. Bhatt has been a consultant to and has received research funding from several companies. Dr. Pershad had no disclosures.
The first revision since 2015 to the European Society of Cardiology’s guidelines for diagnosing and managing non ST-elevation acute coronary syndrome placed increased reliance on high-sensitivity cardiac troponin testing for diagnosis, and embraced coronary CT to rule out lower-risk patients.
It also highlighted the need for personalized antiplatelet regimens, systems of care, and quality improvement.
The society released the new guidelines on August 29 (Eur Heart J. 2020 Aug 29;doi: 10.1093/eurheartj/ehaa575), and then devoted a session to them the next day at the virtual annual congress of the European Society of Cardiology to highlight some of the key updates, starting with the further emphasis placed on high-sensitivity cardiac troponin (hs-cTn), a reliance that contrasts with what remains inconsistent use of this metric in U.S. practice.
An hs-cTn test is the “preferred” diagnostic test and a “key” testing element, said Marco Roffi, MD, professor and director of interventional cardiology at University Hospital, Geneva, and a member of the guideline committee. He also stressed an update to the time frame of initial hs-cTn testing, which now involves a baseline reading and then a second measure after 2 hours to discern how the marker level is evolving with time. The guidelines advise against measuring any other biomarker of myocardial injury.
U.S. lags in measuring high-sensitivity cardiac troponin
U.S. medical systems and centers “are not uniform in adopting hs-cTn,” noted Richard J. Kovacs, MD, professor of cardiology at the Indiana University School of Medicine in Indianapolis. “The new European guidelines should spur U.S. institutions to at least take a close look at the advantages of hs-cTn. There is a strong case that it leads to more efficient emergency care and allows for quicker decisions and triage,” added Dr. Kovacs in an interview.
The new guideline’s emphasis on hs-cTn should hasten broader uptake in U.S. practice, agreed Deepak L. Bhatt, MD, professor of medicine at Harvard Medical School in Boston and a member of the guideline-writing panel
Another plus of the guidelines is its endorsement of an “organized approach to risk assessment” early on in these patients, said Dr. Kovacs, who is also the immediate past-president of the American College of Cardiology (ACC). An ACC committee is developing a new set of recommendations for managing patients with cardiac chest pain and is on track for release in 2021. It would represent the first update to U.S. guidelines for non ST-elevation ACS patients since 2014.
The new ESC guidelines give coronary CT angiography a class Ia rating as an alternative to invasive coronary angiography for assessing patients with a low or intermediate risk of having coronary disease, a “tremendous upgrade,” commented Ashish Pershad, MD, an interventional cardiologist at Banner-University Medical Heart Institute in Phoenix. While he welcomes this support for using coronary CT angiography in this setting, he acknowledged that the method remains limited in availability as it requires highly trained technicians to obtain good images and experienced clinicians to interpret the results.
Personalizing antiplatelet and antithrombotic treatments
Notable revisions to medical treatments to minimize ischemia included an admonition not to use routine pretreatment with a P2Y12 receptor inhibitor (such as clopidogrel) before testing determines coronary anatomy.
Not using one of these antiplatelet drugs upfront on all patients “is a tremendous change,” Dr. Pershad said in an interview. Many patients currently get these drugs while awaiting an angiogram, but a more selected and deferred antiplatelet approach would be better when angiography shows that some patients need coronary bypass surgery, he noted. Recent study results have shown no added benefit from pretreatment, and its use can be especially problematic for patients who are slated for a planned invasive strategy, said Dr. Bhatt.
Dr. Pershad, Dr. Bhatt, and Dr. Kovacs all praised the overall emphasis on a personalized approach to treating patients with antiplatelet and antithrombotic drugs, with endorsement of a flexible approach to treatment intensity and duration. The guideline acknowledges the need to take into account a patient’s bleeding risk and comorbidities, and specifically endorsed the Academic Research Consortium’s formula for identifying and stratifying high bleeding risk (Eur Heart J. 2019 Aug 14;40[31]:2632-53).
The new guidelines also provide guidance on how to apply recent study results that addressed balancing efficacy and safety when pairing an antiplatelet drug with a direct-acting oral anticoagulant (DOAC) for patients who potentially need both drug classes, such as patients with atrial fibrillation and a recent ACS event. “It’s tremendous to get clarity on this issue; there’s been a lot of uncertainty,” said Dr. Pershad. The guidelines call for a week of triple therapy with a DOAC, aspirin, and a second antiplatelet drug, followed by 12 months on a DOAC plus a single antiplatelet drug, and then the DOAC alone as the “default” strategy for most patients, but also presents alternative options for patients with high risk for either bleeding or ischemia.
The new guidelines also give much-needed direction on how to apply an invasive strategy, with an emphasis on immediate intervention for within 2 hours for very-high-risk patients, and early intervention within 24 hours for high-risk patients. Adhering to this timetable can mean increasing catheter laboratory availability on an urgent basis over weekends, Dr. Bhatt noted.
Improving quality of care
A novel section in the new guidelines was devoted to nine quality measures that can help health systems and medical centers monitor their adherence to the guideline recommendations, track their performance relative to peer institutions, and follow changes in performance that result from quality improvement steps. It’s something of a “futuristic” step for a guideline to take, with a goal of persuading administrators to implement tracking of these measures and improve patient outcomes, noted Dr. Bhatt.
“It’s very important to see that this is not just a set of guidelines but also a tool to improve quality of care,” commented Dr. Kovacs. The key to success in this effort will be to follow registered patients, set benchmarks that systems can aspire to achieve, and use this to improve the quality of care.
Until now, optimizing care for patients with NSTE-ACS has been “challenging,” he said. “The focus must be on moving toward systems of care” that can provide consistent patient evaluation and care, and do it quickly, said Dr. Kovacs.
Dr. Roffi has received research funding from Biotronik, Boston Scientific, GE Healthcare, and Medtronic. Dr. Kovacs was formerly an employee of Eli Lilly. Dr. Bhatt has been a consultant to and has received research funding from several companies. Dr. Pershad had no disclosures.
The first revision since 2015 to the European Society of Cardiology’s guidelines for diagnosing and managing non ST-elevation acute coronary syndrome placed increased reliance on high-sensitivity cardiac troponin testing for diagnosis, and embraced coronary CT to rule out lower-risk patients.
It also highlighted the need for personalized antiplatelet regimens, systems of care, and quality improvement.
The society released the new guidelines on August 29 (Eur Heart J. 2020 Aug 29;doi: 10.1093/eurheartj/ehaa575), and then devoted a session to them the next day at the virtual annual congress of the European Society of Cardiology to highlight some of the key updates, starting with the further emphasis placed on high-sensitivity cardiac troponin (hs-cTn), a reliance that contrasts with what remains inconsistent use of this metric in U.S. practice.
An hs-cTn test is the “preferred” diagnostic test and a “key” testing element, said Marco Roffi, MD, professor and director of interventional cardiology at University Hospital, Geneva, and a member of the guideline committee. He also stressed an update to the time frame of initial hs-cTn testing, which now involves a baseline reading and then a second measure after 2 hours to discern how the marker level is evolving with time. The guidelines advise against measuring any other biomarker of myocardial injury.
U.S. lags in measuring high-sensitivity cardiac troponin
U.S. medical systems and centers “are not uniform in adopting hs-cTn,” noted Richard J. Kovacs, MD, professor of cardiology at the Indiana University School of Medicine in Indianapolis. “The new European guidelines should spur U.S. institutions to at least take a close look at the advantages of hs-cTn. There is a strong case that it leads to more efficient emergency care and allows for quicker decisions and triage,” added Dr. Kovacs in an interview.
The new guideline’s emphasis on hs-cTn should hasten broader uptake in U.S. practice, agreed Deepak L. Bhatt, MD, professor of medicine at Harvard Medical School in Boston and a member of the guideline-writing panel
Another plus of the guidelines is its endorsement of an “organized approach to risk assessment” early on in these patients, said Dr. Kovacs, who is also the immediate past-president of the American College of Cardiology (ACC). An ACC committee is developing a new set of recommendations for managing patients with cardiac chest pain and is on track for release in 2021. It would represent the first update to U.S. guidelines for non ST-elevation ACS patients since 2014.
The new ESC guidelines give coronary CT angiography a class Ia rating as an alternative to invasive coronary angiography for assessing patients with a low or intermediate risk of having coronary disease, a “tremendous upgrade,” commented Ashish Pershad, MD, an interventional cardiologist at Banner-University Medical Heart Institute in Phoenix. While he welcomes this support for using coronary CT angiography in this setting, he acknowledged that the method remains limited in availability as it requires highly trained technicians to obtain good images and experienced clinicians to interpret the results.
Personalizing antiplatelet and antithrombotic treatments
Notable revisions to medical treatments to minimize ischemia included an admonition not to use routine pretreatment with a P2Y12 receptor inhibitor (such as clopidogrel) before testing determines coronary anatomy.
Not using one of these antiplatelet drugs upfront on all patients “is a tremendous change,” Dr. Pershad said in an interview. Many patients currently get these drugs while awaiting an angiogram, but a more selected and deferred antiplatelet approach would be better when angiography shows that some patients need coronary bypass surgery, he noted. Recent study results have shown no added benefit from pretreatment, and its use can be especially problematic for patients who are slated for a planned invasive strategy, said Dr. Bhatt.
Dr. Pershad, Dr. Bhatt, and Dr. Kovacs all praised the overall emphasis on a personalized approach to treating patients with antiplatelet and antithrombotic drugs, with endorsement of a flexible approach to treatment intensity and duration. The guideline acknowledges the need to take into account a patient’s bleeding risk and comorbidities, and specifically endorsed the Academic Research Consortium’s formula for identifying and stratifying high bleeding risk (Eur Heart J. 2019 Aug 14;40[31]:2632-53).
The new guidelines also provide guidance on how to apply recent study results that addressed balancing efficacy and safety when pairing an antiplatelet drug with a direct-acting oral anticoagulant (DOAC) for patients who potentially need both drug classes, such as patients with atrial fibrillation and a recent ACS event. “It’s tremendous to get clarity on this issue; there’s been a lot of uncertainty,” said Dr. Pershad. The guidelines call for a week of triple therapy with a DOAC, aspirin, and a second antiplatelet drug, followed by 12 months on a DOAC plus a single antiplatelet drug, and then the DOAC alone as the “default” strategy for most patients, but also presents alternative options for patients with high risk for either bleeding or ischemia.
The new guidelines also give much-needed direction on how to apply an invasive strategy, with an emphasis on immediate intervention for within 2 hours for very-high-risk patients, and early intervention within 24 hours for high-risk patients. Adhering to this timetable can mean increasing catheter laboratory availability on an urgent basis over weekends, Dr. Bhatt noted.
Improving quality of care
A novel section in the new guidelines was devoted to nine quality measures that can help health systems and medical centers monitor their adherence to the guideline recommendations, track their performance relative to peer institutions, and follow changes in performance that result from quality improvement steps. It’s something of a “futuristic” step for a guideline to take, with a goal of persuading administrators to implement tracking of these measures and improve patient outcomes, noted Dr. Bhatt.
“It’s very important to see that this is not just a set of guidelines but also a tool to improve quality of care,” commented Dr. Kovacs. The key to success in this effort will be to follow registered patients, set benchmarks that systems can aspire to achieve, and use this to improve the quality of care.
Until now, optimizing care for patients with NSTE-ACS has been “challenging,” he said. “The focus must be on moving toward systems of care” that can provide consistent patient evaluation and care, and do it quickly, said Dr. Kovacs.
Dr. Roffi has received research funding from Biotronik, Boston Scientific, GE Healthcare, and Medtronic. Dr. Kovacs was formerly an employee of Eli Lilly. Dr. Bhatt has been a consultant to and has received research funding from several companies. Dr. Pershad had no disclosures.
FROM ESC CONGRESS 2020
FDA expands remdesivir use for all COVID-19 hospitalized patients
An EUA of remdesivir issued in May allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who need oxygen therapy or mechanical ventilation.
“Today, based on the Agency’s ongoing review of the EUA, including its review of the totality of scientific information now available, the FDA has determined that it is reasonable to believe Veklury may be effective for the treatment of suspected or laboratory-confirmed COVID-19 in all hospitalized adult and pediatric patients,” the FDA news release about the expanded EUA said. “The Agency’s review has also concluded that the known and potential benefits of Veklury outweigh the known and potential risks for these uses.”
‘Further evaluation’ needed
The EUA expansion is partially based on the results of a randomized, open-label trial that Gilead Sciences, remdesivir’s manufacturer, conducted at multiple sites.
The trial showed that a 5-day course of remdesivir was associated with statistically significant improvement among patients hospitalized with moderate COVID-19 in comparison with those receiving standard care. However, patients who were randomly assigned to a receive longer, 10-day remdesivir course had not improved significantly 11 days after treatment started, compared with those who received standard care.
Results with remdesivir in this trial and in two previously reported randomized trials varied, “raising the question of whether the discrepancies are artifacts of study design choices, including patient populations, or whether the drug is less efficacious than hoped,” wrote Erin K. McCreary, PharmD, and Derek C. Angus, MD, MPH, with the University of Pittsburgh School of Medicine, in an editorial that accompanied publication of the trials in JAMA.
Angus previously expressed concern that expanding remdesivir’s EUA could “interrupt or thwart efforts to execute the needed RCTs [randomized controlled trials].
“We think there really needs to be further evaluation of remdesivir in large-scale RCTs adequately powered to understand in which patients, at which dose, given at which point in the course of illness leads to what concrete and tangible improvement in clinical outcomes,” he told Medscape Medical News.
“At this point, remdesivir definitely holds promise, but given the cost to produce and distribute the drug, it seems crucial to know with more certainty how best to use it,” Angus said.
The EUA expansion is also partially based on results from a randomized, double-blind, placebo-controlled clinical trial that the National Institutes of Allergy and Infectious Diseases conducted. In that trial, there was a statistically significant reduction in median recovery time and higher odds of clinical improvement after 2 weeks for hospitalized patients who received remdesivir.
For hospitalized patients with mild to moderate disease, the results were consistent with the overall study results but were not statistically significant.
This article first appeared on Medscape.com.
An EUA of remdesivir issued in May allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who need oxygen therapy or mechanical ventilation.
“Today, based on the Agency’s ongoing review of the EUA, including its review of the totality of scientific information now available, the FDA has determined that it is reasonable to believe Veklury may be effective for the treatment of suspected or laboratory-confirmed COVID-19 in all hospitalized adult and pediatric patients,” the FDA news release about the expanded EUA said. “The Agency’s review has also concluded that the known and potential benefits of Veklury outweigh the known and potential risks for these uses.”
‘Further evaluation’ needed
The EUA expansion is partially based on the results of a randomized, open-label trial that Gilead Sciences, remdesivir’s manufacturer, conducted at multiple sites.
The trial showed that a 5-day course of remdesivir was associated with statistically significant improvement among patients hospitalized with moderate COVID-19 in comparison with those receiving standard care. However, patients who were randomly assigned to a receive longer, 10-day remdesivir course had not improved significantly 11 days after treatment started, compared with those who received standard care.
Results with remdesivir in this trial and in two previously reported randomized trials varied, “raising the question of whether the discrepancies are artifacts of study design choices, including patient populations, or whether the drug is less efficacious than hoped,” wrote Erin K. McCreary, PharmD, and Derek C. Angus, MD, MPH, with the University of Pittsburgh School of Medicine, in an editorial that accompanied publication of the trials in JAMA.
Angus previously expressed concern that expanding remdesivir’s EUA could “interrupt or thwart efforts to execute the needed RCTs [randomized controlled trials].
“We think there really needs to be further evaluation of remdesivir in large-scale RCTs adequately powered to understand in which patients, at which dose, given at which point in the course of illness leads to what concrete and tangible improvement in clinical outcomes,” he told Medscape Medical News.
“At this point, remdesivir definitely holds promise, but given the cost to produce and distribute the drug, it seems crucial to know with more certainty how best to use it,” Angus said.
The EUA expansion is also partially based on results from a randomized, double-blind, placebo-controlled clinical trial that the National Institutes of Allergy and Infectious Diseases conducted. In that trial, there was a statistically significant reduction in median recovery time and higher odds of clinical improvement after 2 weeks for hospitalized patients who received remdesivir.
For hospitalized patients with mild to moderate disease, the results were consistent with the overall study results but were not statistically significant.
This article first appeared on Medscape.com.
An EUA of remdesivir issued in May allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who need oxygen therapy or mechanical ventilation.
“Today, based on the Agency’s ongoing review of the EUA, including its review of the totality of scientific information now available, the FDA has determined that it is reasonable to believe Veklury may be effective for the treatment of suspected or laboratory-confirmed COVID-19 in all hospitalized adult and pediatric patients,” the FDA news release about the expanded EUA said. “The Agency’s review has also concluded that the known and potential benefits of Veklury outweigh the known and potential risks for these uses.”
‘Further evaluation’ needed
The EUA expansion is partially based on the results of a randomized, open-label trial that Gilead Sciences, remdesivir’s manufacturer, conducted at multiple sites.
The trial showed that a 5-day course of remdesivir was associated with statistically significant improvement among patients hospitalized with moderate COVID-19 in comparison with those receiving standard care. However, patients who were randomly assigned to a receive longer, 10-day remdesivir course had not improved significantly 11 days after treatment started, compared with those who received standard care.
Results with remdesivir in this trial and in two previously reported randomized trials varied, “raising the question of whether the discrepancies are artifacts of study design choices, including patient populations, or whether the drug is less efficacious than hoped,” wrote Erin K. McCreary, PharmD, and Derek C. Angus, MD, MPH, with the University of Pittsburgh School of Medicine, in an editorial that accompanied publication of the trials in JAMA.
Angus previously expressed concern that expanding remdesivir’s EUA could “interrupt or thwart efforts to execute the needed RCTs [randomized controlled trials].
“We think there really needs to be further evaluation of remdesivir in large-scale RCTs adequately powered to understand in which patients, at which dose, given at which point in the course of illness leads to what concrete and tangible improvement in clinical outcomes,” he told Medscape Medical News.
“At this point, remdesivir definitely holds promise, but given the cost to produce and distribute the drug, it seems crucial to know with more certainty how best to use it,” Angus said.
The EUA expansion is also partially based on results from a randomized, double-blind, placebo-controlled clinical trial that the National Institutes of Allergy and Infectious Diseases conducted. In that trial, there was a statistically significant reduction in median recovery time and higher odds of clinical improvement after 2 weeks for hospitalized patients who received remdesivir.
For hospitalized patients with mild to moderate disease, the results were consistent with the overall study results but were not statistically significant.
This article first appeared on Medscape.com.
Evolocumab safe and effective in pediatric FH
The PCSK9 monoclonal antibody evolocumab (Repatha) was well tolerated and effectively lowered LDL cholesterol by 38% compared with placebo in a randomized controlled trial in pediatric patients with heterozygous familial hypercholesterolemia (FH) already taking statins with or without ezetimibe.
“HAUSER-RCT is the largest study and the first placebo-controlled randomized trial of a PCSK9 inhibitor in pediatric FH,” senior author Daniel Gaudet, MD, PhD, Universite de Montreal, said in an interview.
“The study showed good safety and efficacy of the drug in this population, with an excellent 44% reduction in LDL cholesterol compared with 6% in the placebo group.”
The trial also found evolocumab to be well tolerated in this group, with adverse effects similar in the active and placebo groups.
“Some people have wondered about using a drug with a monthly injection in a pediatric population, but this was not an issue in our study,” Dr. Gaudet said. “The idea of a monthly injection was well received, and no patient withdrew because of this.”
The HAUSER-RCT trial was presented on Aug. 29 at the virtual annual congress of the European Society of Cardiology (ESC Congress 2020) and simultaneously published online in the New England Journal of Medicine.
“With patients recruited from 23 countries in five continents, the study provides an accurate picture of the safety and efficacy of evolocumab in pediatric FH patients worldwide,” Dr. Gaudet said.
The 24-week, randomized, double-blind, placebo-controlled trial involved 157 patients aged 10-17 years with heterozygous FH already taking statins with or without ezetimibe and who had an LDL cholesterol level of 130 mg/dL or more and a triglyceride level of 400 mg/dL or less.
They were randomly assigned in a 2:1 ratio to receive monthly subcutaneous injections of evolocumab (420 mg) or placebo.
Results showed that at week 24, the mean percentage change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group, giving a difference of −38.3 percentage points (P < .001).
The absolute change in the LDL cholesterol level was −77.5 mg/dL in the evolocumab group and −9.0 mg/dL in the placebo group, giving a difference of −68.6 mg/dL (P < .001).
Results for all secondary lipid variables were significantly better with evolocumab than with placebo. The incidence of adverse events that occurred during the treatment period was similar in the evolocumab and placebo groups. Laboratory abnormalities did not differ between groups.
Dr. Gaudet noted that FH is the most common genetic disease worldwide, affecting 1 in 250 people. “It is very treatable, so it is important to identify these patients, but it is massively underdiagnosed, with only around 15%-20% of patients with the condition having been identified,” he said.
“The vast majority of pediatric FH patients can reach target LDL levels with statins and ezetimibe, but there are 5%-10% of patients who may need additional therapy. We have now shown that evolocumab is safe and effective for these patients and can be used to fill this gap,” Dr. Gaudet said. “We can now say that we can cover all situations in treating FH whatever the severity of the disease.”
However, the challenge remains to improve the diagnosis of FH. “If there is one person with FH in a family, then it is essential that the whole extended family is tested. Our toolbox for treating this condition is now sufficiently effective, so there is no reason not to diagnose this disease,” Dr. Gaudet stressed.
The HAUSER-RCT study was supported by Amgen. Gaudet reports grants and personal fees from Amgen during the conduct of the study.
A version of this article originally appeared on Medscape.com.
The PCSK9 monoclonal antibody evolocumab (Repatha) was well tolerated and effectively lowered LDL cholesterol by 38% compared with placebo in a randomized controlled trial in pediatric patients with heterozygous familial hypercholesterolemia (FH) already taking statins with or without ezetimibe.
“HAUSER-RCT is the largest study and the first placebo-controlled randomized trial of a PCSK9 inhibitor in pediatric FH,” senior author Daniel Gaudet, MD, PhD, Universite de Montreal, said in an interview.
“The study showed good safety and efficacy of the drug in this population, with an excellent 44% reduction in LDL cholesterol compared with 6% in the placebo group.”
The trial also found evolocumab to be well tolerated in this group, with adverse effects similar in the active and placebo groups.
“Some people have wondered about using a drug with a monthly injection in a pediatric population, but this was not an issue in our study,” Dr. Gaudet said. “The idea of a monthly injection was well received, and no patient withdrew because of this.”
The HAUSER-RCT trial was presented on Aug. 29 at the virtual annual congress of the European Society of Cardiology (ESC Congress 2020) and simultaneously published online in the New England Journal of Medicine.
“With patients recruited from 23 countries in five continents, the study provides an accurate picture of the safety and efficacy of evolocumab in pediatric FH patients worldwide,” Dr. Gaudet said.
The 24-week, randomized, double-blind, placebo-controlled trial involved 157 patients aged 10-17 years with heterozygous FH already taking statins with or without ezetimibe and who had an LDL cholesterol level of 130 mg/dL or more and a triglyceride level of 400 mg/dL or less.
They were randomly assigned in a 2:1 ratio to receive monthly subcutaneous injections of evolocumab (420 mg) or placebo.
Results showed that at week 24, the mean percentage change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group, giving a difference of −38.3 percentage points (P < .001).
The absolute change in the LDL cholesterol level was −77.5 mg/dL in the evolocumab group and −9.0 mg/dL in the placebo group, giving a difference of −68.6 mg/dL (P < .001).
Results for all secondary lipid variables were significantly better with evolocumab than with placebo. The incidence of adverse events that occurred during the treatment period was similar in the evolocumab and placebo groups. Laboratory abnormalities did not differ between groups.
Dr. Gaudet noted that FH is the most common genetic disease worldwide, affecting 1 in 250 people. “It is very treatable, so it is important to identify these patients, but it is massively underdiagnosed, with only around 15%-20% of patients with the condition having been identified,” he said.
“The vast majority of pediatric FH patients can reach target LDL levels with statins and ezetimibe, but there are 5%-10% of patients who may need additional therapy. We have now shown that evolocumab is safe and effective for these patients and can be used to fill this gap,” Dr. Gaudet said. “We can now say that we can cover all situations in treating FH whatever the severity of the disease.”
However, the challenge remains to improve the diagnosis of FH. “If there is one person with FH in a family, then it is essential that the whole extended family is tested. Our toolbox for treating this condition is now sufficiently effective, so there is no reason not to diagnose this disease,” Dr. Gaudet stressed.
The HAUSER-RCT study was supported by Amgen. Gaudet reports grants and personal fees from Amgen during the conduct of the study.
A version of this article originally appeared on Medscape.com.
The PCSK9 monoclonal antibody evolocumab (Repatha) was well tolerated and effectively lowered LDL cholesterol by 38% compared with placebo in a randomized controlled trial in pediatric patients with heterozygous familial hypercholesterolemia (FH) already taking statins with or without ezetimibe.
“HAUSER-RCT is the largest study and the first placebo-controlled randomized trial of a PCSK9 inhibitor in pediatric FH,” senior author Daniel Gaudet, MD, PhD, Universite de Montreal, said in an interview.
“The study showed good safety and efficacy of the drug in this population, with an excellent 44% reduction in LDL cholesterol compared with 6% in the placebo group.”
The trial also found evolocumab to be well tolerated in this group, with adverse effects similar in the active and placebo groups.
“Some people have wondered about using a drug with a monthly injection in a pediatric population, but this was not an issue in our study,” Dr. Gaudet said. “The idea of a monthly injection was well received, and no patient withdrew because of this.”
The HAUSER-RCT trial was presented on Aug. 29 at the virtual annual congress of the European Society of Cardiology (ESC Congress 2020) and simultaneously published online in the New England Journal of Medicine.
“With patients recruited from 23 countries in five continents, the study provides an accurate picture of the safety and efficacy of evolocumab in pediatric FH patients worldwide,” Dr. Gaudet said.
The 24-week, randomized, double-blind, placebo-controlled trial involved 157 patients aged 10-17 years with heterozygous FH already taking statins with or without ezetimibe and who had an LDL cholesterol level of 130 mg/dL or more and a triglyceride level of 400 mg/dL or less.
They were randomly assigned in a 2:1 ratio to receive monthly subcutaneous injections of evolocumab (420 mg) or placebo.
Results showed that at week 24, the mean percentage change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group, giving a difference of −38.3 percentage points (P < .001).
The absolute change in the LDL cholesterol level was −77.5 mg/dL in the evolocumab group and −9.0 mg/dL in the placebo group, giving a difference of −68.6 mg/dL (P < .001).
Results for all secondary lipid variables were significantly better with evolocumab than with placebo. The incidence of adverse events that occurred during the treatment period was similar in the evolocumab and placebo groups. Laboratory abnormalities did not differ between groups.
Dr. Gaudet noted that FH is the most common genetic disease worldwide, affecting 1 in 250 people. “It is very treatable, so it is important to identify these patients, but it is massively underdiagnosed, with only around 15%-20% of patients with the condition having been identified,” he said.
“The vast majority of pediatric FH patients can reach target LDL levels with statins and ezetimibe, but there are 5%-10% of patients who may need additional therapy. We have now shown that evolocumab is safe and effective for these patients and can be used to fill this gap,” Dr. Gaudet said. “We can now say that we can cover all situations in treating FH whatever the severity of the disease.”
However, the challenge remains to improve the diagnosis of FH. “If there is one person with FH in a family, then it is essential that the whole extended family is tested. Our toolbox for treating this condition is now sufficiently effective, so there is no reason not to diagnose this disease,” Dr. Gaudet stressed.
The HAUSER-RCT study was supported by Amgen. Gaudet reports grants and personal fees from Amgen during the conduct of the study.
A version of this article originally appeared on Medscape.com.
ATPCI: Trimetazidine fizzles for post-PCI angina
Adding trimetazidine to optimal medical therapy does not improve outcomes following successful percutaneous coronary intervention (PCI) for stable angina or a non–ST-elevated myocardial infarction, results of the ATPCI trial show.
There was no benefit for the composite primary endpoint of cardiac death, hospitalization for cardiac events, or recurrent/persistent angina requiring an addition, switch, or increased dose of antianginal therapies, or requiring coronary angiography (hazard ratio, 0.98; 95% confidence interval, 0.88-1.09).
Further, there were no between-group differences in any of the individual components of the endpoint or any prespecified subgroups, Roberto Ferrari, MD, professor of cardiology at the University of Ferrara (Italy), reported in a Hot Line session at the digital European Society of Cardiology Congress 2020.
“I think one of the reasons why we couldn’t see any results was really due to this population was extremely well treated,” he said. “Almost all of them were receiving either a beta-blocker or calcium blocker and, on top of this, they had a successful angioplasty and that is what we should do, at least according to ESC guidelines.”
Research has shown that about 85% to 90% of patients have a change in New York Heart Association angina class within 30 days of PCI, leaving very little angina leftover to treat, observed Magnus Ohman, MD, director of the advanced coronary disease program at Duke University, Durham, N.C., who was not involved in the study.
“The fundamental question is whether this was the right study. Is this agent ineffective, or is it just that it was studied in the wrong population? That to me is really the crux of the matter,” he said in an interview.
There is potential benefit in chronic angina, which reflects the level II recommendation by the ESC, said Dr. Ohman. “Those patients typically require more therapy and, in the ideal world of treating angina, you need both physiological and metabolic agents to treat angina and trimetazidine is one metabolic agent.”
Trimetazidine is not available in the United States, but the anti-ischemic metabolic agent is recommended as second-line therapy for angina after beta-blockers and calcium-channel blockers in the 2019 ESC guidelines on chronic coronary syndrome.
Unlike other commonly used first- and second-line antianginal drugs, trimetazidine is devoid of hemodynamic effects, Dr. Ferrari said. It improves myocardial utilization by favoring glucose to fatty acids, thus allowing anaerobic adenosine triphosphate formation and preventing acidosis.
In the absence of contemporary data on the prognostic benefits of antianginal drugs in post-PCI patients, ATPCI investigators at 365 centers in 27 countries randomly assigned 6007 patients with stable angina or non–ST-segment MI after successful elective or urgent PCI to optimal medical therapy alone or with trimetazidine, 35 mg modified-release twice daily.
Patients with severe heart failure, valvular disease, arrhythmia, renal failure or acute ST-elevation MI were excluded.
Most patients (77% male) had Canadian Class Cardiovascular Society class III/IV angina (58%) and were receiving aspirin plus a P2Y12 inhibitor (97%), lipid-lowering agent (96.6%), renin-angiotensin inhibitors (82.2%), and beta-blockers (83.9%). A quarter were receiving calcium-channel blockers (27.6%). In all, 2517 patients had an urgent PCI and 3490 had an elective PCI.
After a median follow-up of 47.5 months, the composite primary endpoint occurred in 23.3% of the trimetazidine group and 23.7% of the control group, according to the study, which was published simultaneously in The Lancet.
The incidence of the individual components was similar:
- Cardiac death: 2.1% vs. 2.6% (HR, 0.81)
- Hospital admission for cardiac events: 13.4% vs. 13.4% (HR, 1.01)
- Angina leading to coronary angiography: 16.9% vs. 16.6% (HR, 1.02)
- Angina leading to increase/switch in antianginal drugs (HR, 1.01)
There was no between-group difference in the composite major secondary endpoint, which included the primary endpoint components plus ischemia leading to coronary angiography and an increase or switch in antianginal therapies. This outcome occurred in 23.5% and 24.0% of patients in the trimetazidine and control groups, respectively (HR, 0.98; 95% CI, 0.88-1.08).
Results were also similar when the primary endpoint was analyzed based on whether patients underwent elective PCI (HR, 0.94; 95% CI, 0.82-1.08) or urgent PCI (HR, 1.04; 95% CI, 0.88-1.22), Dr. Ferrari reported.
Given the lack of observed efficacy, trimetazidine has no use or place in the population studied, said Stephan Windecker, MD, the formal discussant for the study and chair of cardiology at Bern (Switzerland) University Hospital. “Notwithstanding, I think we have to recognize that the optimal medical therapy is so potent and has been well implemented in this trial that any additional medication beyond this is just unable to exploit additional benefit.”
The study was supported by Servier. Dr. Ferrari received fees, honoraria, and travel expenses from Servier. Dr. Ohman reports no relevant financial conflicts of interest. Dr. Windecker is an unpaid member of the steering/executive group for trials funded by Abbott, Abiomed, Amgen, BMS, Boston Scientific, Biotronik, Cardiovalve, Edwards Lifesciences, MedAlliancé, Medtronic, Polares, Sinomed, V-Wave, and Xeltis but has not received personal payments from any pharmaceutical or device company.
This article first appeared on Medscape.com.
Adding trimetazidine to optimal medical therapy does not improve outcomes following successful percutaneous coronary intervention (PCI) for stable angina or a non–ST-elevated myocardial infarction, results of the ATPCI trial show.
There was no benefit for the composite primary endpoint of cardiac death, hospitalization for cardiac events, or recurrent/persistent angina requiring an addition, switch, or increased dose of antianginal therapies, or requiring coronary angiography (hazard ratio, 0.98; 95% confidence interval, 0.88-1.09).
Further, there were no between-group differences in any of the individual components of the endpoint or any prespecified subgroups, Roberto Ferrari, MD, professor of cardiology at the University of Ferrara (Italy), reported in a Hot Line session at the digital European Society of Cardiology Congress 2020.
“I think one of the reasons why we couldn’t see any results was really due to this population was extremely well treated,” he said. “Almost all of them were receiving either a beta-blocker or calcium blocker and, on top of this, they had a successful angioplasty and that is what we should do, at least according to ESC guidelines.”
Research has shown that about 85% to 90% of patients have a change in New York Heart Association angina class within 30 days of PCI, leaving very little angina leftover to treat, observed Magnus Ohman, MD, director of the advanced coronary disease program at Duke University, Durham, N.C., who was not involved in the study.
“The fundamental question is whether this was the right study. Is this agent ineffective, or is it just that it was studied in the wrong population? That to me is really the crux of the matter,” he said in an interview.
There is potential benefit in chronic angina, which reflects the level II recommendation by the ESC, said Dr. Ohman. “Those patients typically require more therapy and, in the ideal world of treating angina, you need both physiological and metabolic agents to treat angina and trimetazidine is one metabolic agent.”
Trimetazidine is not available in the United States, but the anti-ischemic metabolic agent is recommended as second-line therapy for angina after beta-blockers and calcium-channel blockers in the 2019 ESC guidelines on chronic coronary syndrome.
Unlike other commonly used first- and second-line antianginal drugs, trimetazidine is devoid of hemodynamic effects, Dr. Ferrari said. It improves myocardial utilization by favoring glucose to fatty acids, thus allowing anaerobic adenosine triphosphate formation and preventing acidosis.
In the absence of contemporary data on the prognostic benefits of antianginal drugs in post-PCI patients, ATPCI investigators at 365 centers in 27 countries randomly assigned 6007 patients with stable angina or non–ST-segment MI after successful elective or urgent PCI to optimal medical therapy alone or with trimetazidine, 35 mg modified-release twice daily.
Patients with severe heart failure, valvular disease, arrhythmia, renal failure or acute ST-elevation MI were excluded.
Most patients (77% male) had Canadian Class Cardiovascular Society class III/IV angina (58%) and were receiving aspirin plus a P2Y12 inhibitor (97%), lipid-lowering agent (96.6%), renin-angiotensin inhibitors (82.2%), and beta-blockers (83.9%). A quarter were receiving calcium-channel blockers (27.6%). In all, 2517 patients had an urgent PCI and 3490 had an elective PCI.
After a median follow-up of 47.5 months, the composite primary endpoint occurred in 23.3% of the trimetazidine group and 23.7% of the control group, according to the study, which was published simultaneously in The Lancet.
The incidence of the individual components was similar:
- Cardiac death: 2.1% vs. 2.6% (HR, 0.81)
- Hospital admission for cardiac events: 13.4% vs. 13.4% (HR, 1.01)
- Angina leading to coronary angiography: 16.9% vs. 16.6% (HR, 1.02)
- Angina leading to increase/switch in antianginal drugs (HR, 1.01)
There was no between-group difference in the composite major secondary endpoint, which included the primary endpoint components plus ischemia leading to coronary angiography and an increase or switch in antianginal therapies. This outcome occurred in 23.5% and 24.0% of patients in the trimetazidine and control groups, respectively (HR, 0.98; 95% CI, 0.88-1.08).
Results were also similar when the primary endpoint was analyzed based on whether patients underwent elective PCI (HR, 0.94; 95% CI, 0.82-1.08) or urgent PCI (HR, 1.04; 95% CI, 0.88-1.22), Dr. Ferrari reported.
Given the lack of observed efficacy, trimetazidine has no use or place in the population studied, said Stephan Windecker, MD, the formal discussant for the study and chair of cardiology at Bern (Switzerland) University Hospital. “Notwithstanding, I think we have to recognize that the optimal medical therapy is so potent and has been well implemented in this trial that any additional medication beyond this is just unable to exploit additional benefit.”
The study was supported by Servier. Dr. Ferrari received fees, honoraria, and travel expenses from Servier. Dr. Ohman reports no relevant financial conflicts of interest. Dr. Windecker is an unpaid member of the steering/executive group for trials funded by Abbott, Abiomed, Amgen, BMS, Boston Scientific, Biotronik, Cardiovalve, Edwards Lifesciences, MedAlliancé, Medtronic, Polares, Sinomed, V-Wave, and Xeltis but has not received personal payments from any pharmaceutical or device company.
This article first appeared on Medscape.com.
Adding trimetazidine to optimal medical therapy does not improve outcomes following successful percutaneous coronary intervention (PCI) for stable angina or a non–ST-elevated myocardial infarction, results of the ATPCI trial show.
There was no benefit for the composite primary endpoint of cardiac death, hospitalization for cardiac events, or recurrent/persistent angina requiring an addition, switch, or increased dose of antianginal therapies, or requiring coronary angiography (hazard ratio, 0.98; 95% confidence interval, 0.88-1.09).
Further, there were no between-group differences in any of the individual components of the endpoint or any prespecified subgroups, Roberto Ferrari, MD, professor of cardiology at the University of Ferrara (Italy), reported in a Hot Line session at the digital European Society of Cardiology Congress 2020.
“I think one of the reasons why we couldn’t see any results was really due to this population was extremely well treated,” he said. “Almost all of them were receiving either a beta-blocker or calcium blocker and, on top of this, they had a successful angioplasty and that is what we should do, at least according to ESC guidelines.”
Research has shown that about 85% to 90% of patients have a change in New York Heart Association angina class within 30 days of PCI, leaving very little angina leftover to treat, observed Magnus Ohman, MD, director of the advanced coronary disease program at Duke University, Durham, N.C., who was not involved in the study.
“The fundamental question is whether this was the right study. Is this agent ineffective, or is it just that it was studied in the wrong population? That to me is really the crux of the matter,” he said in an interview.
There is potential benefit in chronic angina, which reflects the level II recommendation by the ESC, said Dr. Ohman. “Those patients typically require more therapy and, in the ideal world of treating angina, you need both physiological and metabolic agents to treat angina and trimetazidine is one metabolic agent.”
Trimetazidine is not available in the United States, but the anti-ischemic metabolic agent is recommended as second-line therapy for angina after beta-blockers and calcium-channel blockers in the 2019 ESC guidelines on chronic coronary syndrome.
Unlike other commonly used first- and second-line antianginal drugs, trimetazidine is devoid of hemodynamic effects, Dr. Ferrari said. It improves myocardial utilization by favoring glucose to fatty acids, thus allowing anaerobic adenosine triphosphate formation and preventing acidosis.
In the absence of contemporary data on the prognostic benefits of antianginal drugs in post-PCI patients, ATPCI investigators at 365 centers in 27 countries randomly assigned 6007 patients with stable angina or non–ST-segment MI after successful elective or urgent PCI to optimal medical therapy alone or with trimetazidine, 35 mg modified-release twice daily.
Patients with severe heart failure, valvular disease, arrhythmia, renal failure or acute ST-elevation MI were excluded.
Most patients (77% male) had Canadian Class Cardiovascular Society class III/IV angina (58%) and were receiving aspirin plus a P2Y12 inhibitor (97%), lipid-lowering agent (96.6%), renin-angiotensin inhibitors (82.2%), and beta-blockers (83.9%). A quarter were receiving calcium-channel blockers (27.6%). In all, 2517 patients had an urgent PCI and 3490 had an elective PCI.
After a median follow-up of 47.5 months, the composite primary endpoint occurred in 23.3% of the trimetazidine group and 23.7% of the control group, according to the study, which was published simultaneously in The Lancet.
The incidence of the individual components was similar:
- Cardiac death: 2.1% vs. 2.6% (HR, 0.81)
- Hospital admission for cardiac events: 13.4% vs. 13.4% (HR, 1.01)
- Angina leading to coronary angiography: 16.9% vs. 16.6% (HR, 1.02)
- Angina leading to increase/switch in antianginal drugs (HR, 1.01)
There was no between-group difference in the composite major secondary endpoint, which included the primary endpoint components plus ischemia leading to coronary angiography and an increase or switch in antianginal therapies. This outcome occurred in 23.5% and 24.0% of patients in the trimetazidine and control groups, respectively (HR, 0.98; 95% CI, 0.88-1.08).
Results were also similar when the primary endpoint was analyzed based on whether patients underwent elective PCI (HR, 0.94; 95% CI, 0.82-1.08) or urgent PCI (HR, 1.04; 95% CI, 0.88-1.22), Dr. Ferrari reported.
Given the lack of observed efficacy, trimetazidine has no use or place in the population studied, said Stephan Windecker, MD, the formal discussant for the study and chair of cardiology at Bern (Switzerland) University Hospital. “Notwithstanding, I think we have to recognize that the optimal medical therapy is so potent and has been well implemented in this trial that any additional medication beyond this is just unable to exploit additional benefit.”
The study was supported by Servier. Dr. Ferrari received fees, honoraria, and travel expenses from Servier. Dr. Ohman reports no relevant financial conflicts of interest. Dr. Windecker is an unpaid member of the steering/executive group for trials funded by Abbott, Abiomed, Amgen, BMS, Boston Scientific, Biotronik, Cardiovalve, Edwards Lifesciences, MedAlliancé, Medtronic, Polares, Sinomed, V-Wave, and Xeltis but has not received personal payments from any pharmaceutical or device company.
This article first appeared on Medscape.com.