User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
FDA okays new indication for alirocumab in homozygous FH
The Food and Drug Administration has approved alirocumab (Praluent, Regeneron Pharmaceuticals) injection as add-on therapy for adults with homozygous familial hypercholesterolemia, the agency announced.
The proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor was originally approved in the United States in 2015 as an adjunct to diet, alone or in combination with other lipid-lowering therapies, to reduce LDL cholesterol in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (FH).
Heterozygous FH is one of the most common genetic disorders, affecting 1 in every 200-500 people worldwide, whereas homozygous FH is very rare, affecting about 1 in 1 million people worldwide.
Alirocumab is also approved to reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with cardiovascular disease.
The new indication is based on a 12-week randomized trial in 45 adults who received 150 mg alirocumab every 2 weeks and 24 patients who received placebo, both on top of other therapies to reduce LDL cholesterol. At week 12, patients receiving alirocumab had an average 27% decrease in LDL cholesterol, compared with an average 9% increase among patients on placebo.
Common side effects of alirocumab are nasopharyngitis, injection-site reactions, and influenza. Serious hypersensitivity reactions have occurred among people taking alirocumab.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved alirocumab (Praluent, Regeneron Pharmaceuticals) injection as add-on therapy for adults with homozygous familial hypercholesterolemia, the agency announced.
The proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor was originally approved in the United States in 2015 as an adjunct to diet, alone or in combination with other lipid-lowering therapies, to reduce LDL cholesterol in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (FH).
Heterozygous FH is one of the most common genetic disorders, affecting 1 in every 200-500 people worldwide, whereas homozygous FH is very rare, affecting about 1 in 1 million people worldwide.
Alirocumab is also approved to reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with cardiovascular disease.
The new indication is based on a 12-week randomized trial in 45 adults who received 150 mg alirocumab every 2 weeks and 24 patients who received placebo, both on top of other therapies to reduce LDL cholesterol. At week 12, patients receiving alirocumab had an average 27% decrease in LDL cholesterol, compared with an average 9% increase among patients on placebo.
Common side effects of alirocumab are nasopharyngitis, injection-site reactions, and influenza. Serious hypersensitivity reactions have occurred among people taking alirocumab.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved alirocumab (Praluent, Regeneron Pharmaceuticals) injection as add-on therapy for adults with homozygous familial hypercholesterolemia, the agency announced.
The proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor was originally approved in the United States in 2015 as an adjunct to diet, alone or in combination with other lipid-lowering therapies, to reduce LDL cholesterol in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (FH).
Heterozygous FH is one of the most common genetic disorders, affecting 1 in every 200-500 people worldwide, whereas homozygous FH is very rare, affecting about 1 in 1 million people worldwide.
Alirocumab is also approved to reduce the risk of myocardial infarction, stroke, and unstable angina requiring hospitalization in adults with cardiovascular disease.
The new indication is based on a 12-week randomized trial in 45 adults who received 150 mg alirocumab every 2 weeks and 24 patients who received placebo, both on top of other therapies to reduce LDL cholesterol. At week 12, patients receiving alirocumab had an average 27% decrease in LDL cholesterol, compared with an average 9% increase among patients on placebo.
Common side effects of alirocumab are nasopharyngitis, injection-site reactions, and influenza. Serious hypersensitivity reactions have occurred among people taking alirocumab.
A version of this article first appeared on Medscape.com.
Starting April 5, patients can read your notes: 5 things to consider
Change in writing style is not mandated
The mandate, called “open notes” by many, is part of the 21st Century Cures Act, a wide-ranging piece of federal health care legislation. The previous deadline of Nov. 2, 2020, for enacting open notes was extended last year because of the exigencies of the COVID-19 pandemic.
Organizations must provide access via patient portals to the following types of notes: consultations, discharge summaries, histories, physical examination findings, imaging narratives, laboratory and pathology report narratives, and procedure and progress notes. Noncompliant organizations will eventually be subject to fines from the Department of Health & Human Services for “information blocking.”
This news organization reported on the mandate in 2020, and some readers said it was an unwelcome intrusion into practice. Since then, this news organization has run additional open notes stories about physician concerns, a perspective essay addressing those fears, and a reader poll about the phenomenon.
Now, as the legislation turns into a practical clinical matter, there are five key points clinicians should consider.
Clinicians don’t have to change writing style.
The new law mandates timely patient access to notes and test results, but it doesn’t require that clinicians alter their writing, said Scott MacDonald, MD, an internist and electronic health record medical director at University of California Davis Health in Sacramento.
“You don’t have to change your notes,” he said. However, patients are now part of the note audience and some health care systems are directing clinicians to make patient-friendly style changes.
Everyday experience should guide clinicians when writing notes, said one expert.
“When you’re not sure [of how to write a note], just mirror the way you would speak in the office – that’s going to get you right, including for mental health issues,” advised Leonor Fernandez, MD, an internist at Beth Deaconess Israel Medical Center, Boston, in her “take-away” comments in the online video, How to Write an Open Note.
According to a 2020 Medscape poll of 1,050 physicians, a majority (56%) anticipate that they will write notes differently, knowing that patients can read them via open notes. Nearly two-thirds (64%) believe that this new wrinkle in medical records will increase their workload. However, actual practice suggests that this is true for a minority of practitioners, according to the results from a recent study of more than 1,000 physicians in Boston, Seattle, and rural Pennsylvania, who already work in open notes settings. Only about one-third (37%) reported “spending more time on documentation.”
Note writing is going to change because of the addition of the patient reader, and something will be lost, argued Steven Reidbord, MD, a psychiatrist in private practice in San Francisco. By watering down the language for patients, “you are trading away the technical precision and other advantages of having a professional language,” commented Dr. Reidbord, who blogs for Psychology Today and has criticized the open notes movement in the past.
However, years of investigation from OpenNotes, the Boston-based advocacy and research organization, indicates that there are many gains with patient-accessible notes, including improved medical record accuracy, greater medication adherence, and potentially improved health care disparities among a range of patient types. In a 2019 study, researchers said that worry and confusion among note-reading patients are uncommon (5% and 3%, respectively), which addresses two criticisms voiced by multiple people last year.
Some clinical notes can be withheld.
The new rules from the federal government permit information blocking if there is clear evidence that doing so “will substantially reduce the risk of harm” to patients or to other third parties, Tom Delbanco, MD, and Charlotte Blease, PhD, of OpenNotes in Boston wrote in a commentary in February 2021.
There are also state-level laws that can supersede the new U.S. law and block access to notes, points out MacDonald. For example, California law dictates that providers cannot post cancer test results without talking with the patient first.
The OpenNotes organization also points out that, with regard to sensitive psychotherapy notes that are separated from the rest of a medical record, those notes “can be kept from patients without their permission, and such rules vary state by state.”
Some patients are more likely readers.
Some patients are more likely to peer into their files than others, said Liz Salmi, senior strategist at OpenNotes, who is also a brain cancer patient.
“Those patients who have more serious or chronic conditions ... are more likely to read their notes,” she said in an interview.
A new study of nearly 6,000 medical oncology patients at the University of Wisconsin confirmed that opinion. Patients with incurable metastatic disease were much more likely than those with early-stage, curable disease to read notes. Notably, younger patients were more likely than older ones to access notes, likely the result of generational tech savvy.
Despite the unpredictability of serious disease such as cancer, oncology patients find satisfaction in reading their notes, say experts. “We’ve overwhelmingly heard that patients like it,” Thomas LeBlanc, MD, medical oncologist at Duke University, Durham, N.C., where all patients already have access to clinicians’ notes, told this news organization in 2018.
You are part of the avant garde.
The United States and Scandinavian countries are the world leaders in implementing open notes in clinical practice, Dr. Blease said in an interview.
“It’s a phenomenal achievement” to have enacted open notes nationally, she said. For example, there are no open notes in Northern Ireland, Dr. Blease’s home country, or most of Europe.
In the United States, there are more than 200 medical organizations, including at least one in every state, that were voluntarily providing open notes before April 5, including interstate giants such as Banner Health and big-name medical centers such as Cleveland Clinic.
It may be hard for the United States to top Sweden’s embrace of the practice. The national open notes program now has 7.2 million patient accounts in a country of 10 million people, noted Maria Häggland, PhD, of Uppsala (Sweden) MedTech Science Innovation Center during a webinar last year.
The start day will come, and you may not notice.
“When April 5 happens, something brand new is going to happen symbolically,” Ms. Salmi said. Its importance is hard to measure.
“Patients say they trust their doctor more because they understand their thinking with open notes. How do you value that? We don’t have metrics for that,” she said.
Dr. MacDonald suggested that open notes are both new and not new. In the fall of 2020, he predicted that the launch day would come, and few clinicians would notice, in part because many patients already access truncated information via patient portals.
However, there are “sensitive issues,” such as with adolescents and reproductive health, where “we know that some parents have sign-in information for their teen’s portal,” he commented. With clinical notes now on full display, potential problems “may be out of our control.”
Still, the Sacramento-based physician and IT officer acknowledged that concerns about open notes may be a bit inflated. “I’ve been more worried about reassuring physicians that everything will be okay than what’s actually going to happen [as the law takes effect],” Dr. MacDonald said.
The OpenNotes organization is grant funded, and staff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Change in writing style is not mandated
Change in writing style is not mandated
The mandate, called “open notes” by many, is part of the 21st Century Cures Act, a wide-ranging piece of federal health care legislation. The previous deadline of Nov. 2, 2020, for enacting open notes was extended last year because of the exigencies of the COVID-19 pandemic.
Organizations must provide access via patient portals to the following types of notes: consultations, discharge summaries, histories, physical examination findings, imaging narratives, laboratory and pathology report narratives, and procedure and progress notes. Noncompliant organizations will eventually be subject to fines from the Department of Health & Human Services for “information blocking.”
This news organization reported on the mandate in 2020, and some readers said it was an unwelcome intrusion into practice. Since then, this news organization has run additional open notes stories about physician concerns, a perspective essay addressing those fears, and a reader poll about the phenomenon.
Now, as the legislation turns into a practical clinical matter, there are five key points clinicians should consider.
Clinicians don’t have to change writing style.
The new law mandates timely patient access to notes and test results, but it doesn’t require that clinicians alter their writing, said Scott MacDonald, MD, an internist and electronic health record medical director at University of California Davis Health in Sacramento.
“You don’t have to change your notes,” he said. However, patients are now part of the note audience and some health care systems are directing clinicians to make patient-friendly style changes.
Everyday experience should guide clinicians when writing notes, said one expert.
“When you’re not sure [of how to write a note], just mirror the way you would speak in the office – that’s going to get you right, including for mental health issues,” advised Leonor Fernandez, MD, an internist at Beth Deaconess Israel Medical Center, Boston, in her “take-away” comments in the online video, How to Write an Open Note.
According to a 2020 Medscape poll of 1,050 physicians, a majority (56%) anticipate that they will write notes differently, knowing that patients can read them via open notes. Nearly two-thirds (64%) believe that this new wrinkle in medical records will increase their workload. However, actual practice suggests that this is true for a minority of practitioners, according to the results from a recent study of more than 1,000 physicians in Boston, Seattle, and rural Pennsylvania, who already work in open notes settings. Only about one-third (37%) reported “spending more time on documentation.”
Note writing is going to change because of the addition of the patient reader, and something will be lost, argued Steven Reidbord, MD, a psychiatrist in private practice in San Francisco. By watering down the language for patients, “you are trading away the technical precision and other advantages of having a professional language,” commented Dr. Reidbord, who blogs for Psychology Today and has criticized the open notes movement in the past.
However, years of investigation from OpenNotes, the Boston-based advocacy and research organization, indicates that there are many gains with patient-accessible notes, including improved medical record accuracy, greater medication adherence, and potentially improved health care disparities among a range of patient types. In a 2019 study, researchers said that worry and confusion among note-reading patients are uncommon (5% and 3%, respectively), which addresses two criticisms voiced by multiple people last year.
Some clinical notes can be withheld.
The new rules from the federal government permit information blocking if there is clear evidence that doing so “will substantially reduce the risk of harm” to patients or to other third parties, Tom Delbanco, MD, and Charlotte Blease, PhD, of OpenNotes in Boston wrote in a commentary in February 2021.
There are also state-level laws that can supersede the new U.S. law and block access to notes, points out MacDonald. For example, California law dictates that providers cannot post cancer test results without talking with the patient first.
The OpenNotes organization also points out that, with regard to sensitive psychotherapy notes that are separated from the rest of a medical record, those notes “can be kept from patients without their permission, and such rules vary state by state.”
Some patients are more likely readers.
Some patients are more likely to peer into their files than others, said Liz Salmi, senior strategist at OpenNotes, who is also a brain cancer patient.
“Those patients who have more serious or chronic conditions ... are more likely to read their notes,” she said in an interview.
A new study of nearly 6,000 medical oncology patients at the University of Wisconsin confirmed that opinion. Patients with incurable metastatic disease were much more likely than those with early-stage, curable disease to read notes. Notably, younger patients were more likely than older ones to access notes, likely the result of generational tech savvy.
Despite the unpredictability of serious disease such as cancer, oncology patients find satisfaction in reading their notes, say experts. “We’ve overwhelmingly heard that patients like it,” Thomas LeBlanc, MD, medical oncologist at Duke University, Durham, N.C., where all patients already have access to clinicians’ notes, told this news organization in 2018.
You are part of the avant garde.
The United States and Scandinavian countries are the world leaders in implementing open notes in clinical practice, Dr. Blease said in an interview.
“It’s a phenomenal achievement” to have enacted open notes nationally, she said. For example, there are no open notes in Northern Ireland, Dr. Blease’s home country, or most of Europe.
In the United States, there are more than 200 medical organizations, including at least one in every state, that were voluntarily providing open notes before April 5, including interstate giants such as Banner Health and big-name medical centers such as Cleveland Clinic.
It may be hard for the United States to top Sweden’s embrace of the practice. The national open notes program now has 7.2 million patient accounts in a country of 10 million people, noted Maria Häggland, PhD, of Uppsala (Sweden) MedTech Science Innovation Center during a webinar last year.
The start day will come, and you may not notice.
“When April 5 happens, something brand new is going to happen symbolically,” Ms. Salmi said. Its importance is hard to measure.
“Patients say they trust their doctor more because they understand their thinking with open notes. How do you value that? We don’t have metrics for that,” she said.
Dr. MacDonald suggested that open notes are both new and not new. In the fall of 2020, he predicted that the launch day would come, and few clinicians would notice, in part because many patients already access truncated information via patient portals.
However, there are “sensitive issues,” such as with adolescents and reproductive health, where “we know that some parents have sign-in information for their teen’s portal,” he commented. With clinical notes now on full display, potential problems “may be out of our control.”
Still, the Sacramento-based physician and IT officer acknowledged that concerns about open notes may be a bit inflated. “I’ve been more worried about reassuring physicians that everything will be okay than what’s actually going to happen [as the law takes effect],” Dr. MacDonald said.
The OpenNotes organization is grant funded, and staff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The mandate, called “open notes” by many, is part of the 21st Century Cures Act, a wide-ranging piece of federal health care legislation. The previous deadline of Nov. 2, 2020, for enacting open notes was extended last year because of the exigencies of the COVID-19 pandemic.
Organizations must provide access via patient portals to the following types of notes: consultations, discharge summaries, histories, physical examination findings, imaging narratives, laboratory and pathology report narratives, and procedure and progress notes. Noncompliant organizations will eventually be subject to fines from the Department of Health & Human Services for “information blocking.”
This news organization reported on the mandate in 2020, and some readers said it was an unwelcome intrusion into practice. Since then, this news organization has run additional open notes stories about physician concerns, a perspective essay addressing those fears, and a reader poll about the phenomenon.
Now, as the legislation turns into a practical clinical matter, there are five key points clinicians should consider.
Clinicians don’t have to change writing style.
The new law mandates timely patient access to notes and test results, but it doesn’t require that clinicians alter their writing, said Scott MacDonald, MD, an internist and electronic health record medical director at University of California Davis Health in Sacramento.
“You don’t have to change your notes,” he said. However, patients are now part of the note audience and some health care systems are directing clinicians to make patient-friendly style changes.
Everyday experience should guide clinicians when writing notes, said one expert.
“When you’re not sure [of how to write a note], just mirror the way you would speak in the office – that’s going to get you right, including for mental health issues,” advised Leonor Fernandez, MD, an internist at Beth Deaconess Israel Medical Center, Boston, in her “take-away” comments in the online video, How to Write an Open Note.
According to a 2020 Medscape poll of 1,050 physicians, a majority (56%) anticipate that they will write notes differently, knowing that patients can read them via open notes. Nearly two-thirds (64%) believe that this new wrinkle in medical records will increase their workload. However, actual practice suggests that this is true for a minority of practitioners, according to the results from a recent study of more than 1,000 physicians in Boston, Seattle, and rural Pennsylvania, who already work in open notes settings. Only about one-third (37%) reported “spending more time on documentation.”
Note writing is going to change because of the addition of the patient reader, and something will be lost, argued Steven Reidbord, MD, a psychiatrist in private practice in San Francisco. By watering down the language for patients, “you are trading away the technical precision and other advantages of having a professional language,” commented Dr. Reidbord, who blogs for Psychology Today and has criticized the open notes movement in the past.
However, years of investigation from OpenNotes, the Boston-based advocacy and research organization, indicates that there are many gains with patient-accessible notes, including improved medical record accuracy, greater medication adherence, and potentially improved health care disparities among a range of patient types. In a 2019 study, researchers said that worry and confusion among note-reading patients are uncommon (5% and 3%, respectively), which addresses two criticisms voiced by multiple people last year.
Some clinical notes can be withheld.
The new rules from the federal government permit information blocking if there is clear evidence that doing so “will substantially reduce the risk of harm” to patients or to other third parties, Tom Delbanco, MD, and Charlotte Blease, PhD, of OpenNotes in Boston wrote in a commentary in February 2021.
There are also state-level laws that can supersede the new U.S. law and block access to notes, points out MacDonald. For example, California law dictates that providers cannot post cancer test results without talking with the patient first.
The OpenNotes organization also points out that, with regard to sensitive psychotherapy notes that are separated from the rest of a medical record, those notes “can be kept from patients without their permission, and such rules vary state by state.”
Some patients are more likely readers.
Some patients are more likely to peer into their files than others, said Liz Salmi, senior strategist at OpenNotes, who is also a brain cancer patient.
“Those patients who have more serious or chronic conditions ... are more likely to read their notes,” she said in an interview.
A new study of nearly 6,000 medical oncology patients at the University of Wisconsin confirmed that opinion. Patients with incurable metastatic disease were much more likely than those with early-stage, curable disease to read notes. Notably, younger patients were more likely than older ones to access notes, likely the result of generational tech savvy.
Despite the unpredictability of serious disease such as cancer, oncology patients find satisfaction in reading their notes, say experts. “We’ve overwhelmingly heard that patients like it,” Thomas LeBlanc, MD, medical oncologist at Duke University, Durham, N.C., where all patients already have access to clinicians’ notes, told this news organization in 2018.
You are part of the avant garde.
The United States and Scandinavian countries are the world leaders in implementing open notes in clinical practice, Dr. Blease said in an interview.
“It’s a phenomenal achievement” to have enacted open notes nationally, she said. For example, there are no open notes in Northern Ireland, Dr. Blease’s home country, or most of Europe.
In the United States, there are more than 200 medical organizations, including at least one in every state, that were voluntarily providing open notes before April 5, including interstate giants such as Banner Health and big-name medical centers such as Cleveland Clinic.
It may be hard for the United States to top Sweden’s embrace of the practice. The national open notes program now has 7.2 million patient accounts in a country of 10 million people, noted Maria Häggland, PhD, of Uppsala (Sweden) MedTech Science Innovation Center during a webinar last year.
The start day will come, and you may not notice.
“When April 5 happens, something brand new is going to happen symbolically,” Ms. Salmi said. Its importance is hard to measure.
“Patients say they trust their doctor more because they understand their thinking with open notes. How do you value that? We don’t have metrics for that,” she said.
Dr. MacDonald suggested that open notes are both new and not new. In the fall of 2020, he predicted that the launch day would come, and few clinicians would notice, in part because many patients already access truncated information via patient portals.
However, there are “sensitive issues,” such as with adolescents and reproductive health, where “we know that some parents have sign-in information for their teen’s portal,” he commented. With clinical notes now on full display, potential problems “may be out of our control.”
Still, the Sacramento-based physician and IT officer acknowledged that concerns about open notes may be a bit inflated. “I’ve been more worried about reassuring physicians that everything will be okay than what’s actually going to happen [as the law takes effect],” Dr. MacDonald said.
The OpenNotes organization is grant funded, and staff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
COVID-19 in 2020: Deaths and disparities
COVID-19 was the third-leading cause of death in the United States in 2020, but that mortality burden did not fall evenly along racial/ethnic lines, according to a provisional report from the Centers for Disease Control and Prevention.
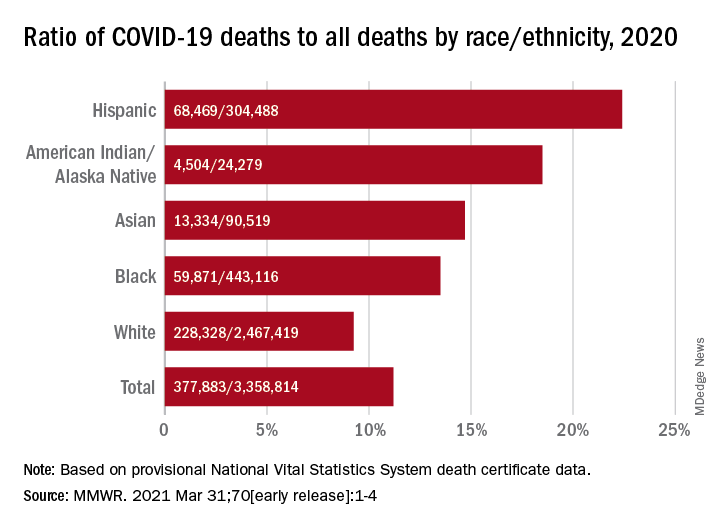
Only heart disease and cancer caused more deaths than SARS-CoV-2, which took the lives of almost 378,000 Americans last year, Farida B. Ahmad, MPH, and associates at the National Center for Health Statistics noted March 31 in the Morbidity and Mortality Weekly Report.
That represents 11.2% of the almost 3.36 million total deaths recorded in 2020. The racial/ethnics demographics, however, show that 22.4% of all deaths among Hispanic Americans were COVID-19–related, as were 18.6% of deaths in American Indians/Alaska Natives. Deaths among Asian persons, at 14.7%, and African Americans, at 13.5%, were closer but still above the national figure, while Whites (9.3%) were the only major subgroup below it, based on data from the National Vital Statistics System.
Age-adjusted death rates tell a somewhat different story: American Indian/Alaska native persons were highest with a rate of 187.8 COVID-19–associated deaths per 100,000 standard population, with Hispanic persons second at 164.3 per 100,000. Blacks were next at 151.1 deaths per 100,000, but Whites had a higher rate (72.5) than did Asian Americans (66.7), the CDC investigators reported.
“During January-December 2020, the estimated 2020 age-adjusted death rate increased for the first time since 2017, with an increase of 15.9% compared with 2019, from 715.2 to 828.7 deaths per 100,000 population,” they wrote, noting that “certain categories of race (i.e., AI/AN and Asian) and Hispanic ethnicity reported on death certificates might have been misclassified, possibly resulting in underestimates of death rates for some groups.”
COVID-19 was the third-leading cause of death in the United States in 2020, but that mortality burden did not fall evenly along racial/ethnic lines, according to a provisional report from the Centers for Disease Control and Prevention.

Only heart disease and cancer caused more deaths than SARS-CoV-2, which took the lives of almost 378,000 Americans last year, Farida B. Ahmad, MPH, and associates at the National Center for Health Statistics noted March 31 in the Morbidity and Mortality Weekly Report.
That represents 11.2% of the almost 3.36 million total deaths recorded in 2020. The racial/ethnics demographics, however, show that 22.4% of all deaths among Hispanic Americans were COVID-19–related, as were 18.6% of deaths in American Indians/Alaska Natives. Deaths among Asian persons, at 14.7%, and African Americans, at 13.5%, were closer but still above the national figure, while Whites (9.3%) were the only major subgroup below it, based on data from the National Vital Statistics System.
Age-adjusted death rates tell a somewhat different story: American Indian/Alaska native persons were highest with a rate of 187.8 COVID-19–associated deaths per 100,000 standard population, with Hispanic persons second at 164.3 per 100,000. Blacks were next at 151.1 deaths per 100,000, but Whites had a higher rate (72.5) than did Asian Americans (66.7), the CDC investigators reported.
“During January-December 2020, the estimated 2020 age-adjusted death rate increased for the first time since 2017, with an increase of 15.9% compared with 2019, from 715.2 to 828.7 deaths per 100,000 population,” they wrote, noting that “certain categories of race (i.e., AI/AN and Asian) and Hispanic ethnicity reported on death certificates might have been misclassified, possibly resulting in underestimates of death rates for some groups.”
COVID-19 was the third-leading cause of death in the United States in 2020, but that mortality burden did not fall evenly along racial/ethnic lines, according to a provisional report from the Centers for Disease Control and Prevention.

Only heart disease and cancer caused more deaths than SARS-CoV-2, which took the lives of almost 378,000 Americans last year, Farida B. Ahmad, MPH, and associates at the National Center for Health Statistics noted March 31 in the Morbidity and Mortality Weekly Report.
That represents 11.2% of the almost 3.36 million total deaths recorded in 2020. The racial/ethnics demographics, however, show that 22.4% of all deaths among Hispanic Americans were COVID-19–related, as were 18.6% of deaths in American Indians/Alaska Natives. Deaths among Asian persons, at 14.7%, and African Americans, at 13.5%, were closer but still above the national figure, while Whites (9.3%) were the only major subgroup below it, based on data from the National Vital Statistics System.
Age-adjusted death rates tell a somewhat different story: American Indian/Alaska native persons were highest with a rate of 187.8 COVID-19–associated deaths per 100,000 standard population, with Hispanic persons second at 164.3 per 100,000. Blacks were next at 151.1 deaths per 100,000, but Whites had a higher rate (72.5) than did Asian Americans (66.7), the CDC investigators reported.
“During January-December 2020, the estimated 2020 age-adjusted death rate increased for the first time since 2017, with an increase of 15.9% compared with 2019, from 715.2 to 828.7 deaths per 100,000 population,” they wrote, noting that “certain categories of race (i.e., AI/AN and Asian) and Hispanic ethnicity reported on death certificates might have been misclassified, possibly resulting in underestimates of death rates for some groups.”
FROM MMWR
Mishap ruins millions of J&J COVID vaccine doses
About 15 million doses of the Johnson & Johnson COVID-19 vaccine were ruined after workers at a manufacturing plant mixed up ingredients, The New York Times reported.
The Baltimore plant is operated by a company called Emergent BioSolutions, the Times said. The company works with both Johnson & Johnson and AstraZeneca.
The mistake has stopped shipments of the vaccine until the FDA investigates, the paper said. The mishap, however, does not affect doses of the J&J one-shot vaccine already delivered and being used.
The problem is that tens of millions of doses were supposed to come from the Baltimore plant.
The Associated Press reported that Emergent has had numerous problems with the FDA, with the agency citing the company for poorly trained employees, cracked vials and mold.
The records cover inspections at Emergent facilities, including Bayview, since 2017. Following a December 2017 inspection at an Emergent plant in Canton, Massachusetts, the FDA said the company hadn’t corrected “continued low level mold and yeast isolates” found in the facility. Nearly a year later, agency investigators questioned why Emergent had “an unwritten policy of not conducting routine compliance audits” at a separate plant in Baltimore, known as Camden, where an anthrax vaccine is filled into vials.
Meanwhile, in a statement, Johnson & Johnson said its own quality control process identified the problem in one batch of ingredients. The company said the Emergent plant in Baltimore is “not yet authorized to manufacture drug substance for our COVID-19 vaccine. This batch was never advanced to the filling and finishing stages of our manufacturing process.”
The company said it plans to still seek emergency use authorization for a different Emergent facility and will provide more experts on site at Emergent.
The Times reports that President Joe Biden’s team still believes the administration can meet its commitment to have enough vaccine doses to immunize every adult by the end of May.
Johnson & Johnson said it still plans to deliver an additional 24 million doses through April.
A version of this article first appeared on WebMD.com.
This article was updated 4/1/21.
About 15 million doses of the Johnson & Johnson COVID-19 vaccine were ruined after workers at a manufacturing plant mixed up ingredients, The New York Times reported.
The Baltimore plant is operated by a company called Emergent BioSolutions, the Times said. The company works with both Johnson & Johnson and AstraZeneca.
The mistake has stopped shipments of the vaccine until the FDA investigates, the paper said. The mishap, however, does not affect doses of the J&J one-shot vaccine already delivered and being used.
The problem is that tens of millions of doses were supposed to come from the Baltimore plant.
The Associated Press reported that Emergent has had numerous problems with the FDA, with the agency citing the company for poorly trained employees, cracked vials and mold.
The records cover inspections at Emergent facilities, including Bayview, since 2017. Following a December 2017 inspection at an Emergent plant in Canton, Massachusetts, the FDA said the company hadn’t corrected “continued low level mold and yeast isolates” found in the facility. Nearly a year later, agency investigators questioned why Emergent had “an unwritten policy of not conducting routine compliance audits” at a separate plant in Baltimore, known as Camden, where an anthrax vaccine is filled into vials.
Meanwhile, in a statement, Johnson & Johnson said its own quality control process identified the problem in one batch of ingredients. The company said the Emergent plant in Baltimore is “not yet authorized to manufacture drug substance for our COVID-19 vaccine. This batch was never advanced to the filling and finishing stages of our manufacturing process.”
The company said it plans to still seek emergency use authorization for a different Emergent facility and will provide more experts on site at Emergent.
The Times reports that President Joe Biden’s team still believes the administration can meet its commitment to have enough vaccine doses to immunize every adult by the end of May.
Johnson & Johnson said it still plans to deliver an additional 24 million doses through April.
A version of this article first appeared on WebMD.com.
This article was updated 4/1/21.
About 15 million doses of the Johnson & Johnson COVID-19 vaccine were ruined after workers at a manufacturing plant mixed up ingredients, The New York Times reported.
The Baltimore plant is operated by a company called Emergent BioSolutions, the Times said. The company works with both Johnson & Johnson and AstraZeneca.
The mistake has stopped shipments of the vaccine until the FDA investigates, the paper said. The mishap, however, does not affect doses of the J&J one-shot vaccine already delivered and being used.
The problem is that tens of millions of doses were supposed to come from the Baltimore plant.
The Associated Press reported that Emergent has had numerous problems with the FDA, with the agency citing the company for poorly trained employees, cracked vials and mold.
The records cover inspections at Emergent facilities, including Bayview, since 2017. Following a December 2017 inspection at an Emergent plant in Canton, Massachusetts, the FDA said the company hadn’t corrected “continued low level mold and yeast isolates” found in the facility. Nearly a year later, agency investigators questioned why Emergent had “an unwritten policy of not conducting routine compliance audits” at a separate plant in Baltimore, known as Camden, where an anthrax vaccine is filled into vials.
Meanwhile, in a statement, Johnson & Johnson said its own quality control process identified the problem in one batch of ingredients. The company said the Emergent plant in Baltimore is “not yet authorized to manufacture drug substance for our COVID-19 vaccine. This batch was never advanced to the filling and finishing stages of our manufacturing process.”
The company said it plans to still seek emergency use authorization for a different Emergent facility and will provide more experts on site at Emergent.
The Times reports that President Joe Biden’s team still believes the administration can meet its commitment to have enough vaccine doses to immunize every adult by the end of May.
Johnson & Johnson said it still plans to deliver an additional 24 million doses through April.
A version of this article first appeared on WebMD.com.
This article was updated 4/1/21.
Detroit cardiologists prevail in retaliation suit against Tenet
After losing at arbitration, as well as in federal court and partially on appeal, Tenet Healthcare is refusing to comment on whether it will continue to battle two Detroit-area cardiologists whom the hospital corporation fired from leadership positions in 2018.
The cardiologists were awarded $10.6 million from an arbitrator, who found that Detroit Medical Center (DMC) and its parent, Tenet, retaliated against Amir Kaki, MD, and Mahir Elder, MD, when the doctors repeatedly reported concerns about patient safety and potential fraud.
The award was made public when it was upheld in federal court in February 2021 and was partially upheld on appeal days later by the Sixth Circuit Court of Appeals.
The Sixth Circuit Court of Appeals denied Tenet’s motion to bar Dr. Kaki and Dr. Elder from returning to work with full privileges but said it would continue to consider the overall appeal. Tenet argued that it needed to keep the cardiologists out of DMC because of “behavioral issues.”
Those allegations are “complete nonsense,” said the cardiologists’ attorney, Deborah Gordon, of Bloomfield Hills, Mich. The alleged problems regarding Dr. Kaki and Dr. Elder were examined by an arbitrator, who “found that all of those complaints were unsubstantiated,” Ms. Gordon said in an interview.
In her final ruling, arbitrator Mary Beth Kelly wrote, “Both Kaki and Elder testified credibly regarding the humiliation, the emotional distress and the reputational damage they have suffered to their national reputations.”
A spokesperson for Tenet and DMC said the organizations had no further comment.
Ms. Gordon said she believes it’s unlikely Tenet will prevail in the Sixth Circuit Court of Appeals, noting that the court already had examined the merits of the case to determine whether Dr. Kaki and Dr. Elder could go back to work. In the court’s opinion, shared in an interview, nothing substantive in Tenet’s appeal prevented the doctors from returning to the hospital, she said.
As of now, both cardiologists have 1 year of privileges at the DMC-affiliated hospitals. Only Dr. Kaki has returned to work, said Ms. Gordon. Neither is speaking to the media, she said.
From respected to reviled
Both Dr. Kaki and Dr. Elder were respected at DMC, according to court filings.
Dr. Kaki was recruited from Weill Cornell Medical College by a Detroit mayor because of his expertise in interventional cardiology. He had staff privileges at DMC beginning in 2012 and was a clinical associate professor and assistant program director of the interventional cardiology fellowship program at Wayne State University in Detroit. He became director of the cardiac catheterization services unit at the new DMC Heart Hospital at Harper-Hutzel Hospital in Detroit in 2014, and 4 years later was appointed director of the facility’s anticoagulation clinic. Dr. Kaki was nominated for and completed Tenet’s Leadership Academy.
Dr. Elder was a clinical professor and assistant fellowship director at Wayne State and was a clinical professor of medicine at Michigan State University. Beginning in 2008, he held directorships at DMC’s cardiac care unit, ambulatory services program, cardiac CT angiogram program, PERT program, and carotid stenting program. Dr. Elder was voted Teacher of the Year for 10 consecutive years by DMC cardiology fellows.
The two doctors aimed high when it came to quality of care and ethics, according to legal filings. Over the years, Dr. Kaki and Dr. Elder repeatedly reported what they considered to be egregious violations of patient safety and of Medicare and Medicaid fraud laws. The clinicians complained about unsterile surgical instruments and the removal of a stat laboratory from the cardiac catheterization unit, noting that the removal would cause delays that would endanger lives.
At peer review meetings, as well as with administrators, they flagged colleagues who they said were performing unnecessary or dangerous procedures solely to generate revenue. At least one doctor falsified records of such a procedure after a patient died, alleged Dr. Kaki and Dr. Elder.
Tenet hired outside attorneys in the fall of 2018, telling Dr. Kaki and Dr. Elder that the legal team would investigate their complaints. However, the investigation was a sham: Filings allege that the investigation was used instead to build a case against Dr. Kaki and Dr. Elder and that Tenet leadership used the inquiry to pressure the cardiologists to resign.
They refused, and in October 2018, they were fired from their leadership positions. DMC and Tenet then held a press conference in which they said that Dr. Kaki and Dr. Elder had been dismissed for “violations” of the “Tenet Standards of Conduct.”
Cardiologists push back
Dr. Kaki and Dr. Elder, however, were not willing to just walk away. They sought reinstatement through an internal DMC appeals panel of their peers. The clinicians who participated on that panel ruled that neither firing was justified.
But DMC’s governing board voted in April 2020 to deny privileges to both cardiologists.
Tenet continued a campaign of retaliation, according to legal filings, by not paying the clinicians for being on call, by removing them from peer review committees, and by prohibiting them from teaching or giving lectures. DMC refused to give Dr. Kaki his personnel record, stating that he was never an employee when he was in the leadership position. Dr. Kaki sued, and a Wayne County Circuit Court judge granted his motion to get his file. DMC and Tenet appealed that ruling but lost.
Eventually, Ms. Gordon sued DMC and Tenet in federal court, alleging the hospital retaliated against the cardiologists, interfered with their ability to earn a living by disparaging them, refused to renew their privileges in 2019, and committed violations under multiple federal and state statutes, including the False Claims Act and the Fair Labor Standards Act.
Tenet successfully argued that the case should go to arbitration.
Arbitrator Mary Beth Kelly, though, ruled in December 2020 that the vast majority of the complaints compiled against the two physicians in the external investigation were not verified or supported and that Tenet and DMC had retaliated against Dr. Kaki and Dr. Elder.
For that harm, Ms. Kelly awarded each clinician $1 million, according to the final ruling shared in an interview.
In addition, she awarded Dr. Kaki $2.3 million in back pay and 2 years of front pay (slightly more than $1 million). She awarded Dr. Elder $2.3 million in back pay and $2.1 million in front pay for 4 years, noting that “his strong association with DMC may make it more difficult for him to successfully transition into the situation he enjoyed prior to termination and nonrenewal.”
The clinicians also were awarded legal fees of $623,816 and court costs of $110,673.
“Wholesale retrial”
To secure the award, Ms. Gordon had to seek a ruling from the U.S. District Court for Eastern Michigan. Tenet asked that court to overturn the arbitrator’s award and to keep it sealed from public view.
In his February ruling, Judge Arthur J. Tarnow wrote that Tenet and DMC “not only attempt to relitigate the legal issues, but also endeavor to introduce a factual counternarrative unmoored from the findings of the Arbitrator and including evidence which the Arbitrator specifically found inadmissible.
“By seeking a wholesale retrial of their case after forcing plaintiffs to arbitrate in the first place,” Tenet and DMC basically ignored the goal of arbitration, which is to relieve judicial congestion and provide a faster and cheaper alternative to litigation, he wrote.
Judge Tarnow also warned Tenet and DMC against taking too long to reinstate privileges for Dr. Kaki and Dr. Elder. If they “continue to delay the restoration of plaintiffs’ privileges in the hopes of a different result on appeal, they will be in violation of this Order,” said the judge.
Tenet, however, tried one more avenue to block the cardiologists from getting privileges, appealing to the Sixth Circuit, which again ordered the company to grant the 1-year privileges.
A version of this article first appeared on Medscape.com.
After losing at arbitration, as well as in federal court and partially on appeal, Tenet Healthcare is refusing to comment on whether it will continue to battle two Detroit-area cardiologists whom the hospital corporation fired from leadership positions in 2018.
The cardiologists were awarded $10.6 million from an arbitrator, who found that Detroit Medical Center (DMC) and its parent, Tenet, retaliated against Amir Kaki, MD, and Mahir Elder, MD, when the doctors repeatedly reported concerns about patient safety and potential fraud.
The award was made public when it was upheld in federal court in February 2021 and was partially upheld on appeal days later by the Sixth Circuit Court of Appeals.
The Sixth Circuit Court of Appeals denied Tenet’s motion to bar Dr. Kaki and Dr. Elder from returning to work with full privileges but said it would continue to consider the overall appeal. Tenet argued that it needed to keep the cardiologists out of DMC because of “behavioral issues.”
Those allegations are “complete nonsense,” said the cardiologists’ attorney, Deborah Gordon, of Bloomfield Hills, Mich. The alleged problems regarding Dr. Kaki and Dr. Elder were examined by an arbitrator, who “found that all of those complaints were unsubstantiated,” Ms. Gordon said in an interview.
In her final ruling, arbitrator Mary Beth Kelly wrote, “Both Kaki and Elder testified credibly regarding the humiliation, the emotional distress and the reputational damage they have suffered to their national reputations.”
A spokesperson for Tenet and DMC said the organizations had no further comment.
Ms. Gordon said she believes it’s unlikely Tenet will prevail in the Sixth Circuit Court of Appeals, noting that the court already had examined the merits of the case to determine whether Dr. Kaki and Dr. Elder could go back to work. In the court’s opinion, shared in an interview, nothing substantive in Tenet’s appeal prevented the doctors from returning to the hospital, she said.
As of now, both cardiologists have 1 year of privileges at the DMC-affiliated hospitals. Only Dr. Kaki has returned to work, said Ms. Gordon. Neither is speaking to the media, she said.
From respected to reviled
Both Dr. Kaki and Dr. Elder were respected at DMC, according to court filings.
Dr. Kaki was recruited from Weill Cornell Medical College by a Detroit mayor because of his expertise in interventional cardiology. He had staff privileges at DMC beginning in 2012 and was a clinical associate professor and assistant program director of the interventional cardiology fellowship program at Wayne State University in Detroit. He became director of the cardiac catheterization services unit at the new DMC Heart Hospital at Harper-Hutzel Hospital in Detroit in 2014, and 4 years later was appointed director of the facility’s anticoagulation clinic. Dr. Kaki was nominated for and completed Tenet’s Leadership Academy.
Dr. Elder was a clinical professor and assistant fellowship director at Wayne State and was a clinical professor of medicine at Michigan State University. Beginning in 2008, he held directorships at DMC’s cardiac care unit, ambulatory services program, cardiac CT angiogram program, PERT program, and carotid stenting program. Dr. Elder was voted Teacher of the Year for 10 consecutive years by DMC cardiology fellows.
The two doctors aimed high when it came to quality of care and ethics, according to legal filings. Over the years, Dr. Kaki and Dr. Elder repeatedly reported what they considered to be egregious violations of patient safety and of Medicare and Medicaid fraud laws. The clinicians complained about unsterile surgical instruments and the removal of a stat laboratory from the cardiac catheterization unit, noting that the removal would cause delays that would endanger lives.
At peer review meetings, as well as with administrators, they flagged colleagues who they said were performing unnecessary or dangerous procedures solely to generate revenue. At least one doctor falsified records of such a procedure after a patient died, alleged Dr. Kaki and Dr. Elder.
Tenet hired outside attorneys in the fall of 2018, telling Dr. Kaki and Dr. Elder that the legal team would investigate their complaints. However, the investigation was a sham: Filings allege that the investigation was used instead to build a case against Dr. Kaki and Dr. Elder and that Tenet leadership used the inquiry to pressure the cardiologists to resign.
They refused, and in October 2018, they were fired from their leadership positions. DMC and Tenet then held a press conference in which they said that Dr. Kaki and Dr. Elder had been dismissed for “violations” of the “Tenet Standards of Conduct.”
Cardiologists push back
Dr. Kaki and Dr. Elder, however, were not willing to just walk away. They sought reinstatement through an internal DMC appeals panel of their peers. The clinicians who participated on that panel ruled that neither firing was justified.
But DMC’s governing board voted in April 2020 to deny privileges to both cardiologists.
Tenet continued a campaign of retaliation, according to legal filings, by not paying the clinicians for being on call, by removing them from peer review committees, and by prohibiting them from teaching or giving lectures. DMC refused to give Dr. Kaki his personnel record, stating that he was never an employee when he was in the leadership position. Dr. Kaki sued, and a Wayne County Circuit Court judge granted his motion to get his file. DMC and Tenet appealed that ruling but lost.
Eventually, Ms. Gordon sued DMC and Tenet in federal court, alleging the hospital retaliated against the cardiologists, interfered with their ability to earn a living by disparaging them, refused to renew their privileges in 2019, and committed violations under multiple federal and state statutes, including the False Claims Act and the Fair Labor Standards Act.
Tenet successfully argued that the case should go to arbitration.
Arbitrator Mary Beth Kelly, though, ruled in December 2020 that the vast majority of the complaints compiled against the two physicians in the external investigation were not verified or supported and that Tenet and DMC had retaliated against Dr. Kaki and Dr. Elder.
For that harm, Ms. Kelly awarded each clinician $1 million, according to the final ruling shared in an interview.
In addition, she awarded Dr. Kaki $2.3 million in back pay and 2 years of front pay (slightly more than $1 million). She awarded Dr. Elder $2.3 million in back pay and $2.1 million in front pay for 4 years, noting that “his strong association with DMC may make it more difficult for him to successfully transition into the situation he enjoyed prior to termination and nonrenewal.”
The clinicians also were awarded legal fees of $623,816 and court costs of $110,673.
“Wholesale retrial”
To secure the award, Ms. Gordon had to seek a ruling from the U.S. District Court for Eastern Michigan. Tenet asked that court to overturn the arbitrator’s award and to keep it sealed from public view.
In his February ruling, Judge Arthur J. Tarnow wrote that Tenet and DMC “not only attempt to relitigate the legal issues, but also endeavor to introduce a factual counternarrative unmoored from the findings of the Arbitrator and including evidence which the Arbitrator specifically found inadmissible.
“By seeking a wholesale retrial of their case after forcing plaintiffs to arbitrate in the first place,” Tenet and DMC basically ignored the goal of arbitration, which is to relieve judicial congestion and provide a faster and cheaper alternative to litigation, he wrote.
Judge Tarnow also warned Tenet and DMC against taking too long to reinstate privileges for Dr. Kaki and Dr. Elder. If they “continue to delay the restoration of plaintiffs’ privileges in the hopes of a different result on appeal, they will be in violation of this Order,” said the judge.
Tenet, however, tried one more avenue to block the cardiologists from getting privileges, appealing to the Sixth Circuit, which again ordered the company to grant the 1-year privileges.
A version of this article first appeared on Medscape.com.
After losing at arbitration, as well as in federal court and partially on appeal, Tenet Healthcare is refusing to comment on whether it will continue to battle two Detroit-area cardiologists whom the hospital corporation fired from leadership positions in 2018.
The cardiologists were awarded $10.6 million from an arbitrator, who found that Detroit Medical Center (DMC) and its parent, Tenet, retaliated against Amir Kaki, MD, and Mahir Elder, MD, when the doctors repeatedly reported concerns about patient safety and potential fraud.
The award was made public when it was upheld in federal court in February 2021 and was partially upheld on appeal days later by the Sixth Circuit Court of Appeals.
The Sixth Circuit Court of Appeals denied Tenet’s motion to bar Dr. Kaki and Dr. Elder from returning to work with full privileges but said it would continue to consider the overall appeal. Tenet argued that it needed to keep the cardiologists out of DMC because of “behavioral issues.”
Those allegations are “complete nonsense,” said the cardiologists’ attorney, Deborah Gordon, of Bloomfield Hills, Mich. The alleged problems regarding Dr. Kaki and Dr. Elder were examined by an arbitrator, who “found that all of those complaints were unsubstantiated,” Ms. Gordon said in an interview.
In her final ruling, arbitrator Mary Beth Kelly wrote, “Both Kaki and Elder testified credibly regarding the humiliation, the emotional distress and the reputational damage they have suffered to their national reputations.”
A spokesperson for Tenet and DMC said the organizations had no further comment.
Ms. Gordon said she believes it’s unlikely Tenet will prevail in the Sixth Circuit Court of Appeals, noting that the court already had examined the merits of the case to determine whether Dr. Kaki and Dr. Elder could go back to work. In the court’s opinion, shared in an interview, nothing substantive in Tenet’s appeal prevented the doctors from returning to the hospital, she said.
As of now, both cardiologists have 1 year of privileges at the DMC-affiliated hospitals. Only Dr. Kaki has returned to work, said Ms. Gordon. Neither is speaking to the media, she said.
From respected to reviled
Both Dr. Kaki and Dr. Elder were respected at DMC, according to court filings.
Dr. Kaki was recruited from Weill Cornell Medical College by a Detroit mayor because of his expertise in interventional cardiology. He had staff privileges at DMC beginning in 2012 and was a clinical associate professor and assistant program director of the interventional cardiology fellowship program at Wayne State University in Detroit. He became director of the cardiac catheterization services unit at the new DMC Heart Hospital at Harper-Hutzel Hospital in Detroit in 2014, and 4 years later was appointed director of the facility’s anticoagulation clinic. Dr. Kaki was nominated for and completed Tenet’s Leadership Academy.
Dr. Elder was a clinical professor and assistant fellowship director at Wayne State and was a clinical professor of medicine at Michigan State University. Beginning in 2008, he held directorships at DMC’s cardiac care unit, ambulatory services program, cardiac CT angiogram program, PERT program, and carotid stenting program. Dr. Elder was voted Teacher of the Year for 10 consecutive years by DMC cardiology fellows.
The two doctors aimed high when it came to quality of care and ethics, according to legal filings. Over the years, Dr. Kaki and Dr. Elder repeatedly reported what they considered to be egregious violations of patient safety and of Medicare and Medicaid fraud laws. The clinicians complained about unsterile surgical instruments and the removal of a stat laboratory from the cardiac catheterization unit, noting that the removal would cause delays that would endanger lives.
At peer review meetings, as well as with administrators, they flagged colleagues who they said were performing unnecessary or dangerous procedures solely to generate revenue. At least one doctor falsified records of such a procedure after a patient died, alleged Dr. Kaki and Dr. Elder.
Tenet hired outside attorneys in the fall of 2018, telling Dr. Kaki and Dr. Elder that the legal team would investigate their complaints. However, the investigation was a sham: Filings allege that the investigation was used instead to build a case against Dr. Kaki and Dr. Elder and that Tenet leadership used the inquiry to pressure the cardiologists to resign.
They refused, and in October 2018, they were fired from their leadership positions. DMC and Tenet then held a press conference in which they said that Dr. Kaki and Dr. Elder had been dismissed for “violations” of the “Tenet Standards of Conduct.”
Cardiologists push back
Dr. Kaki and Dr. Elder, however, were not willing to just walk away. They sought reinstatement through an internal DMC appeals panel of their peers. The clinicians who participated on that panel ruled that neither firing was justified.
But DMC’s governing board voted in April 2020 to deny privileges to both cardiologists.
Tenet continued a campaign of retaliation, according to legal filings, by not paying the clinicians for being on call, by removing them from peer review committees, and by prohibiting them from teaching or giving lectures. DMC refused to give Dr. Kaki his personnel record, stating that he was never an employee when he was in the leadership position. Dr. Kaki sued, and a Wayne County Circuit Court judge granted his motion to get his file. DMC and Tenet appealed that ruling but lost.
Eventually, Ms. Gordon sued DMC and Tenet in federal court, alleging the hospital retaliated against the cardiologists, interfered with their ability to earn a living by disparaging them, refused to renew their privileges in 2019, and committed violations under multiple federal and state statutes, including the False Claims Act and the Fair Labor Standards Act.
Tenet successfully argued that the case should go to arbitration.
Arbitrator Mary Beth Kelly, though, ruled in December 2020 that the vast majority of the complaints compiled against the two physicians in the external investigation were not verified or supported and that Tenet and DMC had retaliated against Dr. Kaki and Dr. Elder.
For that harm, Ms. Kelly awarded each clinician $1 million, according to the final ruling shared in an interview.
In addition, she awarded Dr. Kaki $2.3 million in back pay and 2 years of front pay (slightly more than $1 million). She awarded Dr. Elder $2.3 million in back pay and $2.1 million in front pay for 4 years, noting that “his strong association with DMC may make it more difficult for him to successfully transition into the situation he enjoyed prior to termination and nonrenewal.”
The clinicians also were awarded legal fees of $623,816 and court costs of $110,673.
“Wholesale retrial”
To secure the award, Ms. Gordon had to seek a ruling from the U.S. District Court for Eastern Michigan. Tenet asked that court to overturn the arbitrator’s award and to keep it sealed from public view.
In his February ruling, Judge Arthur J. Tarnow wrote that Tenet and DMC “not only attempt to relitigate the legal issues, but also endeavor to introduce a factual counternarrative unmoored from the findings of the Arbitrator and including evidence which the Arbitrator specifically found inadmissible.
“By seeking a wholesale retrial of their case after forcing plaintiffs to arbitrate in the first place,” Tenet and DMC basically ignored the goal of arbitration, which is to relieve judicial congestion and provide a faster and cheaper alternative to litigation, he wrote.
Judge Tarnow also warned Tenet and DMC against taking too long to reinstate privileges for Dr. Kaki and Dr. Elder. If they “continue to delay the restoration of plaintiffs’ privileges in the hopes of a different result on appeal, they will be in violation of this Order,” said the judge.
Tenet, however, tried one more avenue to block the cardiologists from getting privileges, appealing to the Sixth Circuit, which again ordered the company to grant the 1-year privileges.
A version of this article first appeared on Medscape.com.
Nonfatal opioid overdose rises in teen girls
More adolescent girls than boys experienced nonfatal opioid overdose and reported baseline levels of anxiety, depression, and self-harm, according to data from a retrospective cohort study of more than 20,000 youth in the United States.
Previous studies have identified sex-based differences in opioid overdose such as a higher prevalence of co-occurring psychiatric disorders in women compared with men, wrote Sarah M. Bagley, MD, of Boston University, and colleagues. “However, few studies have examined whether such sex-based differences in opioid overdose risk extend to the population of adolescents and young adults,” they said.
In a retrospective cohort study published in JAMA Network Open, the researchers identified 20,312 commercially insured youth aged 11-24 years who experienced a nonfatal opioid overdose between Jan. 1, 2006, and Dec. 31, 2017, and reviewed data using the IBM MarketScan Commercial Database. The average age of the study population was 20 years and approximately 42% were female.
Females aged 11-16 years had a significantly higher incidence of nonfatal opioid overdose (60%) compared with males, but this trend reversed at age 17 years, after which the incidence of nonfatal opioid overdose became significantly higher in males. “Our finding that females younger than 17 years had a higher incidence of NFOD is consistent with epidemiologic data that have indicated changes in alcohol and drug prevalence among female youths,” the researchers wrote.
Overall, 57.8% of the cohort had mood and anxiety disorders, 12.8% had trauma- or stress-related disorders, and 11.7% had attention-deficit/hyperactivity disorder.
When analyzed by sex, females had a significantly higher prevalence than that of males of mood or anxiety disorders (65.5% vs. 51.9%) trauma or stress-related disorders (16.4% vs. 10.1%) and attempts at suicide or self-harm (14.6% vs. 9.9%). Males had significantly higher prevalence than that of females of opioid use disorder (44.7% vs. 29.2%), cannabis use disorder (18.3% vs. 11.3%), and alcohol use disorder (20.3% vs. 14.4%).
“Although in our study, female youths had a lower prevalence of all substance use disorders, including OUD [opioid use disorder], and a higher prevalence of mood and trauma-associated disorders, both male and female youths had a higher prevalence of psychiatric illness and substance use disorder than youths in the general population,” the researchers noted.
The study findings were limited by several factors including the inclusion only of youth with commercial insurance, with no uninsured or publicly insured youth, and only those youth who sought health care after a nonfatal opioid overdose, the researchers noted. The prevalence of substance use and mental health disorders may be over- or underdiagnosed, and race was not included as a variable because of unreliable data, they added. The database also did not allow for gender identity beyond sex as listed by the insurance carrier, they said.
However, the results indicate significant differences in the incidence of nonfatal opioid overdose and accompanying mental health and substance use disorders based on age and sex, they said.
“These differences may have important implications for developing effective interventions to prevent first-time NFOD and to engage youths in care after an NFOD,” they concluded.
The study was supported by grants to several researchers from the National Institute on Drug Abuse, National Institutes of Health, and the Charles A. King Trust. The researchers had no financial conflicts to disclose.
More adolescent girls than boys experienced nonfatal opioid overdose and reported baseline levels of anxiety, depression, and self-harm, according to data from a retrospective cohort study of more than 20,000 youth in the United States.
Previous studies have identified sex-based differences in opioid overdose such as a higher prevalence of co-occurring psychiatric disorders in women compared with men, wrote Sarah M. Bagley, MD, of Boston University, and colleagues. “However, few studies have examined whether such sex-based differences in opioid overdose risk extend to the population of adolescents and young adults,” they said.
In a retrospective cohort study published in JAMA Network Open, the researchers identified 20,312 commercially insured youth aged 11-24 years who experienced a nonfatal opioid overdose between Jan. 1, 2006, and Dec. 31, 2017, and reviewed data using the IBM MarketScan Commercial Database. The average age of the study population was 20 years and approximately 42% were female.
Females aged 11-16 years had a significantly higher incidence of nonfatal opioid overdose (60%) compared with males, but this trend reversed at age 17 years, after which the incidence of nonfatal opioid overdose became significantly higher in males. “Our finding that females younger than 17 years had a higher incidence of NFOD is consistent with epidemiologic data that have indicated changes in alcohol and drug prevalence among female youths,” the researchers wrote.
Overall, 57.8% of the cohort had mood and anxiety disorders, 12.8% had trauma- or stress-related disorders, and 11.7% had attention-deficit/hyperactivity disorder.
When analyzed by sex, females had a significantly higher prevalence than that of males of mood or anxiety disorders (65.5% vs. 51.9%) trauma or stress-related disorders (16.4% vs. 10.1%) and attempts at suicide or self-harm (14.6% vs. 9.9%). Males had significantly higher prevalence than that of females of opioid use disorder (44.7% vs. 29.2%), cannabis use disorder (18.3% vs. 11.3%), and alcohol use disorder (20.3% vs. 14.4%).
“Although in our study, female youths had a lower prevalence of all substance use disorders, including OUD [opioid use disorder], and a higher prevalence of mood and trauma-associated disorders, both male and female youths had a higher prevalence of psychiatric illness and substance use disorder than youths in the general population,” the researchers noted.
The study findings were limited by several factors including the inclusion only of youth with commercial insurance, with no uninsured or publicly insured youth, and only those youth who sought health care after a nonfatal opioid overdose, the researchers noted. The prevalence of substance use and mental health disorders may be over- or underdiagnosed, and race was not included as a variable because of unreliable data, they added. The database also did not allow for gender identity beyond sex as listed by the insurance carrier, they said.
However, the results indicate significant differences in the incidence of nonfatal opioid overdose and accompanying mental health and substance use disorders based on age and sex, they said.
“These differences may have important implications for developing effective interventions to prevent first-time NFOD and to engage youths in care after an NFOD,” they concluded.
The study was supported by grants to several researchers from the National Institute on Drug Abuse, National Institutes of Health, and the Charles A. King Trust. The researchers had no financial conflicts to disclose.
More adolescent girls than boys experienced nonfatal opioid overdose and reported baseline levels of anxiety, depression, and self-harm, according to data from a retrospective cohort study of more than 20,000 youth in the United States.
Previous studies have identified sex-based differences in opioid overdose such as a higher prevalence of co-occurring psychiatric disorders in women compared with men, wrote Sarah M. Bagley, MD, of Boston University, and colleagues. “However, few studies have examined whether such sex-based differences in opioid overdose risk extend to the population of adolescents and young adults,” they said.
In a retrospective cohort study published in JAMA Network Open, the researchers identified 20,312 commercially insured youth aged 11-24 years who experienced a nonfatal opioid overdose between Jan. 1, 2006, and Dec. 31, 2017, and reviewed data using the IBM MarketScan Commercial Database. The average age of the study population was 20 years and approximately 42% were female.
Females aged 11-16 years had a significantly higher incidence of nonfatal opioid overdose (60%) compared with males, but this trend reversed at age 17 years, after which the incidence of nonfatal opioid overdose became significantly higher in males. “Our finding that females younger than 17 years had a higher incidence of NFOD is consistent with epidemiologic data that have indicated changes in alcohol and drug prevalence among female youths,” the researchers wrote.
Overall, 57.8% of the cohort had mood and anxiety disorders, 12.8% had trauma- or stress-related disorders, and 11.7% had attention-deficit/hyperactivity disorder.
When analyzed by sex, females had a significantly higher prevalence than that of males of mood or anxiety disorders (65.5% vs. 51.9%) trauma or stress-related disorders (16.4% vs. 10.1%) and attempts at suicide or self-harm (14.6% vs. 9.9%). Males had significantly higher prevalence than that of females of opioid use disorder (44.7% vs. 29.2%), cannabis use disorder (18.3% vs. 11.3%), and alcohol use disorder (20.3% vs. 14.4%).
“Although in our study, female youths had a lower prevalence of all substance use disorders, including OUD [opioid use disorder], and a higher prevalence of mood and trauma-associated disorders, both male and female youths had a higher prevalence of psychiatric illness and substance use disorder than youths in the general population,” the researchers noted.
The study findings were limited by several factors including the inclusion only of youth with commercial insurance, with no uninsured or publicly insured youth, and only those youth who sought health care after a nonfatal opioid overdose, the researchers noted. The prevalence of substance use and mental health disorders may be over- or underdiagnosed, and race was not included as a variable because of unreliable data, they added. The database also did not allow for gender identity beyond sex as listed by the insurance carrier, they said.
However, the results indicate significant differences in the incidence of nonfatal opioid overdose and accompanying mental health and substance use disorders based on age and sex, they said.
“These differences may have important implications for developing effective interventions to prevent first-time NFOD and to engage youths in care after an NFOD,” they concluded.
The study was supported by grants to several researchers from the National Institute on Drug Abuse, National Institutes of Health, and the Charles A. King Trust. The researchers had no financial conflicts to disclose.
FROM JAMA NETWORK OPEN
Meta-analysis supports late thrombectomy in selected stroke patients
of data from six clinical trials.
Results of the AURORA analysis showed that for every 100 patients treated with thrombectomy, 33 patients will have less disability, and 27 patients will achieve an independent level of functioning compared with patients who receive only standard medical care.
The benefit of mechanical removal of the clot for selected patients who may have salvageable brain tissue, as identified through the use of various imaging modalities, was maintained whether the patient had a “wake-up stroke” or the onset of symptoms was witnessed, regardless of the point in time within the late window. In fact, the benefit of intervention was greater for patients who presented in the latter part of the late time window.
Never too late for urgent medical care
“While the findings of this analysis do not contradict the mantra that the earlier treatment is instituted, the higher the chance of a good outcome, they highlight the fact that it is never too late to seek urgent medical care,” said lead investigator Tudor Jovin, MD, chair of neurology at Cooper University Hospital, Cherry Hill, New Jersey.
“The implications of the findings from AURORA are that they could lead to a change in guidelines from endorsement of thrombectomy as level 1a recommendation in eligible patients presenting in the 6- to 16-hour time window to a 6- to 24-hour time window,” said Dr. Jovin.
“Furthermore, there are strong signals of benefit of thrombectomy in patients who are not selected based on volumetric analysis of baseline infarct (core) or extent of tissue at risk (penumbra), such that when those imaging modalities are not available or contraindicated, selection based on noncontrast CT and clinical information only may be acceptable,” he added. “Finally, the possibility of benefit from thrombectomy performed beyond 24 hours from last seen well is real and should be explored in future studies.”
The AURORA findings were presented at the virtual International Stroke Conference (ISC) 2021.
The objective of the study was to provide a more precise estimate of the benefit of thrombectomy for patients with stroke when performed within 6-24 hours after the patient was last seen well, Dr. Jovin explained.
He said the 6-hour cutoff was chosen somewhat arbitrarily, but added, “It is highly consequential, as it marks the point of demarcation between the early and late time window, and virtually all guidelines recommend different approaches, dependent on whether patients present before or after 6 hours from symptom onset.”
The 6+ hour window
Dr. Jovin pointed out that for patients who present beyond 6 hours, treatment options are more restricted, because the data on thrombectomy in this later period come mainly from two North American trials (DEFUSE 3 and DAWN) that had very stringent imaging criteria for enrollment.
“We wanted to create a heterogeneous dataset with regard to geography and selection criteria by forming the AURORA collaboration,” he commented. Their study involved an individual-level pooled analysis of all patients who underwent randomization after 6 hours from the time that they were last seen well. Patients were randomly assigned to receive either best medical therapy alone or best medical therapy plus thrombectomy (either with stent retrievers or aspiration) for anterior circulation proximal large-vessel occlusion stroke.
The data came from six trials: DAWN (which enrolled patients 6-24 hours from stroke onset), DEFUSE 3 (6-16 hours), ESCAPE (0-12 hours), REVASCAT (0-8 hours), RESILIENT (0-8 hours), and POSITIVE (0-12 hours). In total, 505 patients were included in the meta-analysis, 266 in the intervention group, and 239 in the control group.
“By pooling data on patients presenting after 6 hours from all these trails, we achieve greater precision for treatment effect estimation and increased the power for subgroup analysis,” Dr. Jovin noted.
Although the majority of the patients were in the DAWN and DIFFUSE 3 trials (n = 388), “there are still a good number from the other four trials (n = 117),” Dr. Jovin reported.
Most of the trials used Modified Rankin Scale (mRS) ordinal or shift analysis as their primary endpoint, which was the also the endpoint chosen for this meta-analysis.
Imaging selection criteria ranged from fully automated software-generated quantitative volumetric analysis of baseline infarct (core) or tissue at risk to CT perfusion and plain CT/CTA. The minimum ASPECTS score was 5 or 6.
There were no imbalances in baseline characteristics. The median NIH Stroke Scale score was 16, and the median ASPECTS score was 8. The median time to randomization was 10.5 hours.
With regard to safety, there was no significant difference in rates of symptomatic intracerebral hemorrhage (5.3% in the intervention group vs. 3.3% in the control group; P = .23) or in mortality at 90 days (16.5% vs. 19.3%; P = .87). Jovin noted that these results are very similar to those from the HERMES meta-analysis of patients treated in the early time window.
The primary outcome – ordinal analysis of the mRS distribution – showed an adjusted odds ratio of 2.54 (P < .0001) for benefit in the intervention group. The number needed to treat to reduce disability was 3. “This is again very similar to the HERMES meta-analysis of patients in the early window,” Dr. Jovin said.
The P value for heterogeneity of treatment effect across the six studies was nonsignificant.
Secondary outcome analysis showed an “almost 27%” difference in good functional outcome (MRS, 0-2) between the two groups (45.9% in the intervention group vs. 19.3% in the control group), which translates into a number needed to treat of 3.8, Dr. Jovin reported.
Subgroup analysis showed a treatment effect favoring intervention across all prespecified subgroup factors, including age, sex, occlusion location, mode of presentation (wake-up vs. witnessed), and ASPECTS score, with the caveat that most of the patients were enrolled with ASPECTS scores of 7 or greater.
Early versus late
Surprisingly, although thrombectomy was found to be beneficial in both the 6- to 12-hour and 12- to 24-hour time window, the magnitude of benefit was significantly higher in the later rather than the earlier time window. The odds ratio of a better outcome with thrombectomy on the mRS shift analysis in those presenting in the 6- to 12-hour period was 1.78, compared with 5.86 in the 12- to 24-hour time window.
“This should not be interpreted as a higher chance of a good outcome if treated late. In fact, the rate of good outcomes were numerically higher in the earlier treated patients, but the difference comes from the control group, which did much worse in patients randomized in the later time period,” Dr. Jovin said.
“Aurora was the goddess of dawn [in ancient Roman mythology], so this is a very fitting name, as it reminds us that we are in the dawn of a new era where patients are selected based on physiological data rather than on time, and we certainly hope that this work has brought us closer to this reality,” Dr. Jovin concluded.
Commenting on the study, Michael Hill, MD, University of Calgary (Alta.), said: “The work provides pooled empiric data to support the concept that time to treatment is no longer the sole threshold variable to be used in treatment decision-making. Instead, time is now simply another variable to consider in the context of clinical and imaging factors.”
Dr. Hill, who headed up the ESCAPE trial and was also involved in the current meta-analysis, added: “This meta-analysis supports the concept of patient selection using the ‘good scan’ model, rather than using a time-based concept of patient eligibility for endovascular therapy. It will further push changes in care, because the implication is that all patients with more severe acute stroke presentations need emergency neurovascular imaging to decide if they are eligible for treatment.”
The AURORA meta-analysis was funded by Stryker Neurovascular. Dr. Jovin reports stock holdings in Anaconda, Route 92, VizAi, FreeOx, Blockade Medical, Methinks, and Corindus; personal fees from Cerenovus and Contego Medical; travel support from Fundacio Ictus; and grant support from Medtronic and Stryker Neurovascular.
A version of this article first appeared on Medscape.com.
of data from six clinical trials.
Results of the AURORA analysis showed that for every 100 patients treated with thrombectomy, 33 patients will have less disability, and 27 patients will achieve an independent level of functioning compared with patients who receive only standard medical care.
The benefit of mechanical removal of the clot for selected patients who may have salvageable brain tissue, as identified through the use of various imaging modalities, was maintained whether the patient had a “wake-up stroke” or the onset of symptoms was witnessed, regardless of the point in time within the late window. In fact, the benefit of intervention was greater for patients who presented in the latter part of the late time window.
Never too late for urgent medical care
“While the findings of this analysis do not contradict the mantra that the earlier treatment is instituted, the higher the chance of a good outcome, they highlight the fact that it is never too late to seek urgent medical care,” said lead investigator Tudor Jovin, MD, chair of neurology at Cooper University Hospital, Cherry Hill, New Jersey.
“The implications of the findings from AURORA are that they could lead to a change in guidelines from endorsement of thrombectomy as level 1a recommendation in eligible patients presenting in the 6- to 16-hour time window to a 6- to 24-hour time window,” said Dr. Jovin.
“Furthermore, there are strong signals of benefit of thrombectomy in patients who are not selected based on volumetric analysis of baseline infarct (core) or extent of tissue at risk (penumbra), such that when those imaging modalities are not available or contraindicated, selection based on noncontrast CT and clinical information only may be acceptable,” he added. “Finally, the possibility of benefit from thrombectomy performed beyond 24 hours from last seen well is real and should be explored in future studies.”
The AURORA findings were presented at the virtual International Stroke Conference (ISC) 2021.
The objective of the study was to provide a more precise estimate of the benefit of thrombectomy for patients with stroke when performed within 6-24 hours after the patient was last seen well, Dr. Jovin explained.
He said the 6-hour cutoff was chosen somewhat arbitrarily, but added, “It is highly consequential, as it marks the point of demarcation between the early and late time window, and virtually all guidelines recommend different approaches, dependent on whether patients present before or after 6 hours from symptom onset.”
The 6+ hour window
Dr. Jovin pointed out that for patients who present beyond 6 hours, treatment options are more restricted, because the data on thrombectomy in this later period come mainly from two North American trials (DEFUSE 3 and DAWN) that had very stringent imaging criteria for enrollment.
“We wanted to create a heterogeneous dataset with regard to geography and selection criteria by forming the AURORA collaboration,” he commented. Their study involved an individual-level pooled analysis of all patients who underwent randomization after 6 hours from the time that they were last seen well. Patients were randomly assigned to receive either best medical therapy alone or best medical therapy plus thrombectomy (either with stent retrievers or aspiration) for anterior circulation proximal large-vessel occlusion stroke.
The data came from six trials: DAWN (which enrolled patients 6-24 hours from stroke onset), DEFUSE 3 (6-16 hours), ESCAPE (0-12 hours), REVASCAT (0-8 hours), RESILIENT (0-8 hours), and POSITIVE (0-12 hours). In total, 505 patients were included in the meta-analysis, 266 in the intervention group, and 239 in the control group.
“By pooling data on patients presenting after 6 hours from all these trails, we achieve greater precision for treatment effect estimation and increased the power for subgroup analysis,” Dr. Jovin noted.
Although the majority of the patients were in the DAWN and DIFFUSE 3 trials (n = 388), “there are still a good number from the other four trials (n = 117),” Dr. Jovin reported.
Most of the trials used Modified Rankin Scale (mRS) ordinal or shift analysis as their primary endpoint, which was the also the endpoint chosen for this meta-analysis.
Imaging selection criteria ranged from fully automated software-generated quantitative volumetric analysis of baseline infarct (core) or tissue at risk to CT perfusion and plain CT/CTA. The minimum ASPECTS score was 5 or 6.
There were no imbalances in baseline characteristics. The median NIH Stroke Scale score was 16, and the median ASPECTS score was 8. The median time to randomization was 10.5 hours.
With regard to safety, there was no significant difference in rates of symptomatic intracerebral hemorrhage (5.3% in the intervention group vs. 3.3% in the control group; P = .23) or in mortality at 90 days (16.5% vs. 19.3%; P = .87). Jovin noted that these results are very similar to those from the HERMES meta-analysis of patients treated in the early time window.
The primary outcome – ordinal analysis of the mRS distribution – showed an adjusted odds ratio of 2.54 (P < .0001) for benefit in the intervention group. The number needed to treat to reduce disability was 3. “This is again very similar to the HERMES meta-analysis of patients in the early window,” Dr. Jovin said.
The P value for heterogeneity of treatment effect across the six studies was nonsignificant.
Secondary outcome analysis showed an “almost 27%” difference in good functional outcome (MRS, 0-2) between the two groups (45.9% in the intervention group vs. 19.3% in the control group), which translates into a number needed to treat of 3.8, Dr. Jovin reported.
Subgroup analysis showed a treatment effect favoring intervention across all prespecified subgroup factors, including age, sex, occlusion location, mode of presentation (wake-up vs. witnessed), and ASPECTS score, with the caveat that most of the patients were enrolled with ASPECTS scores of 7 or greater.
Early versus late
Surprisingly, although thrombectomy was found to be beneficial in both the 6- to 12-hour and 12- to 24-hour time window, the magnitude of benefit was significantly higher in the later rather than the earlier time window. The odds ratio of a better outcome with thrombectomy on the mRS shift analysis in those presenting in the 6- to 12-hour period was 1.78, compared with 5.86 in the 12- to 24-hour time window.
“This should not be interpreted as a higher chance of a good outcome if treated late. In fact, the rate of good outcomes were numerically higher in the earlier treated patients, but the difference comes from the control group, which did much worse in patients randomized in the later time period,” Dr. Jovin said.
“Aurora was the goddess of dawn [in ancient Roman mythology], so this is a very fitting name, as it reminds us that we are in the dawn of a new era where patients are selected based on physiological data rather than on time, and we certainly hope that this work has brought us closer to this reality,” Dr. Jovin concluded.
Commenting on the study, Michael Hill, MD, University of Calgary (Alta.), said: “The work provides pooled empiric data to support the concept that time to treatment is no longer the sole threshold variable to be used in treatment decision-making. Instead, time is now simply another variable to consider in the context of clinical and imaging factors.”
Dr. Hill, who headed up the ESCAPE trial and was also involved in the current meta-analysis, added: “This meta-analysis supports the concept of patient selection using the ‘good scan’ model, rather than using a time-based concept of patient eligibility for endovascular therapy. It will further push changes in care, because the implication is that all patients with more severe acute stroke presentations need emergency neurovascular imaging to decide if they are eligible for treatment.”
The AURORA meta-analysis was funded by Stryker Neurovascular. Dr. Jovin reports stock holdings in Anaconda, Route 92, VizAi, FreeOx, Blockade Medical, Methinks, and Corindus; personal fees from Cerenovus and Contego Medical; travel support from Fundacio Ictus; and grant support from Medtronic and Stryker Neurovascular.
A version of this article first appeared on Medscape.com.
of data from six clinical trials.
Results of the AURORA analysis showed that for every 100 patients treated with thrombectomy, 33 patients will have less disability, and 27 patients will achieve an independent level of functioning compared with patients who receive only standard medical care.
The benefit of mechanical removal of the clot for selected patients who may have salvageable brain tissue, as identified through the use of various imaging modalities, was maintained whether the patient had a “wake-up stroke” or the onset of symptoms was witnessed, regardless of the point in time within the late window. In fact, the benefit of intervention was greater for patients who presented in the latter part of the late time window.
Never too late for urgent medical care
“While the findings of this analysis do not contradict the mantra that the earlier treatment is instituted, the higher the chance of a good outcome, they highlight the fact that it is never too late to seek urgent medical care,” said lead investigator Tudor Jovin, MD, chair of neurology at Cooper University Hospital, Cherry Hill, New Jersey.
“The implications of the findings from AURORA are that they could lead to a change in guidelines from endorsement of thrombectomy as level 1a recommendation in eligible patients presenting in the 6- to 16-hour time window to a 6- to 24-hour time window,” said Dr. Jovin.
“Furthermore, there are strong signals of benefit of thrombectomy in patients who are not selected based on volumetric analysis of baseline infarct (core) or extent of tissue at risk (penumbra), such that when those imaging modalities are not available or contraindicated, selection based on noncontrast CT and clinical information only may be acceptable,” he added. “Finally, the possibility of benefit from thrombectomy performed beyond 24 hours from last seen well is real and should be explored in future studies.”
The AURORA findings were presented at the virtual International Stroke Conference (ISC) 2021.
The objective of the study was to provide a more precise estimate of the benefit of thrombectomy for patients with stroke when performed within 6-24 hours after the patient was last seen well, Dr. Jovin explained.
He said the 6-hour cutoff was chosen somewhat arbitrarily, but added, “It is highly consequential, as it marks the point of demarcation between the early and late time window, and virtually all guidelines recommend different approaches, dependent on whether patients present before or after 6 hours from symptom onset.”
The 6+ hour window
Dr. Jovin pointed out that for patients who present beyond 6 hours, treatment options are more restricted, because the data on thrombectomy in this later period come mainly from two North American trials (DEFUSE 3 and DAWN) that had very stringent imaging criteria for enrollment.
“We wanted to create a heterogeneous dataset with regard to geography and selection criteria by forming the AURORA collaboration,” he commented. Their study involved an individual-level pooled analysis of all patients who underwent randomization after 6 hours from the time that they were last seen well. Patients were randomly assigned to receive either best medical therapy alone or best medical therapy plus thrombectomy (either with stent retrievers or aspiration) for anterior circulation proximal large-vessel occlusion stroke.
The data came from six trials: DAWN (which enrolled patients 6-24 hours from stroke onset), DEFUSE 3 (6-16 hours), ESCAPE (0-12 hours), REVASCAT (0-8 hours), RESILIENT (0-8 hours), and POSITIVE (0-12 hours). In total, 505 patients were included in the meta-analysis, 266 in the intervention group, and 239 in the control group.
“By pooling data on patients presenting after 6 hours from all these trails, we achieve greater precision for treatment effect estimation and increased the power for subgroup analysis,” Dr. Jovin noted.
Although the majority of the patients were in the DAWN and DIFFUSE 3 trials (n = 388), “there are still a good number from the other four trials (n = 117),” Dr. Jovin reported.
Most of the trials used Modified Rankin Scale (mRS) ordinal or shift analysis as their primary endpoint, which was the also the endpoint chosen for this meta-analysis.
Imaging selection criteria ranged from fully automated software-generated quantitative volumetric analysis of baseline infarct (core) or tissue at risk to CT perfusion and plain CT/CTA. The minimum ASPECTS score was 5 or 6.
There were no imbalances in baseline characteristics. The median NIH Stroke Scale score was 16, and the median ASPECTS score was 8. The median time to randomization was 10.5 hours.
With regard to safety, there was no significant difference in rates of symptomatic intracerebral hemorrhage (5.3% in the intervention group vs. 3.3% in the control group; P = .23) or in mortality at 90 days (16.5% vs. 19.3%; P = .87). Jovin noted that these results are very similar to those from the HERMES meta-analysis of patients treated in the early time window.
The primary outcome – ordinal analysis of the mRS distribution – showed an adjusted odds ratio of 2.54 (P < .0001) for benefit in the intervention group. The number needed to treat to reduce disability was 3. “This is again very similar to the HERMES meta-analysis of patients in the early window,” Dr. Jovin said.
The P value for heterogeneity of treatment effect across the six studies was nonsignificant.
Secondary outcome analysis showed an “almost 27%” difference in good functional outcome (MRS, 0-2) between the two groups (45.9% in the intervention group vs. 19.3% in the control group), which translates into a number needed to treat of 3.8, Dr. Jovin reported.
Subgroup analysis showed a treatment effect favoring intervention across all prespecified subgroup factors, including age, sex, occlusion location, mode of presentation (wake-up vs. witnessed), and ASPECTS score, with the caveat that most of the patients were enrolled with ASPECTS scores of 7 or greater.
Early versus late
Surprisingly, although thrombectomy was found to be beneficial in both the 6- to 12-hour and 12- to 24-hour time window, the magnitude of benefit was significantly higher in the later rather than the earlier time window. The odds ratio of a better outcome with thrombectomy on the mRS shift analysis in those presenting in the 6- to 12-hour period was 1.78, compared with 5.86 in the 12- to 24-hour time window.
“This should not be interpreted as a higher chance of a good outcome if treated late. In fact, the rate of good outcomes were numerically higher in the earlier treated patients, but the difference comes from the control group, which did much worse in patients randomized in the later time period,” Dr. Jovin said.
“Aurora was the goddess of dawn [in ancient Roman mythology], so this is a very fitting name, as it reminds us that we are in the dawn of a new era where patients are selected based on physiological data rather than on time, and we certainly hope that this work has brought us closer to this reality,” Dr. Jovin concluded.
Commenting on the study, Michael Hill, MD, University of Calgary (Alta.), said: “The work provides pooled empiric data to support the concept that time to treatment is no longer the sole threshold variable to be used in treatment decision-making. Instead, time is now simply another variable to consider in the context of clinical and imaging factors.”
Dr. Hill, who headed up the ESCAPE trial and was also involved in the current meta-analysis, added: “This meta-analysis supports the concept of patient selection using the ‘good scan’ model, rather than using a time-based concept of patient eligibility for endovascular therapy. It will further push changes in care, because the implication is that all patients with more severe acute stroke presentations need emergency neurovascular imaging to decide if they are eligible for treatment.”
The AURORA meta-analysis was funded by Stryker Neurovascular. Dr. Jovin reports stock holdings in Anaconda, Route 92, VizAi, FreeOx, Blockade Medical, Methinks, and Corindus; personal fees from Cerenovus and Contego Medical; travel support from Fundacio Ictus; and grant support from Medtronic and Stryker Neurovascular.
A version of this article first appeared on Medscape.com.
FROM ISC 2021
Pfizer: Vaccine shown 100% effective in children aged 12-15
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
The study enrolled 2,260 adolescents aged 12-15. No infections were reported in the group given the vaccine produced by Pfizer and its European partner, BioNTech, the release said. The placebo group reported 18 cases of COVID-19.
The vaccinated children showed a strong antibody response with no serious side effects.
Albert Bourla, PhD, chairman and CEO of Pfizer, said the company plans to seek Food and Drug Administration emergency use authorization, which could allow this age group to be vaccinated before the start of the next school year. Pfizer will also seek authorization from the European Medicines Agency.
“We share the urgency to expand the authorization of our vaccine to use in younger populations and are encouraged by the clinical trial data from adolescents between the ages of 12 and 15,” Dr. Bourla said in the release.
The clinical trials showed a stronger response in children aged 12-15 than the 95% effectiveness reported in clinical trials in adults. The Pfizer vaccine is now authorized to be given to people aged 16 and up in the United States.
Health experts said the clinical trials – while not peer-reviewed – amounted to very good news.
“The sooner that we can get vaccines into as many people as possible, regardless of their age, the sooner we will be able to really feel like we’re ending this pandemic for good,” Angela Rasmussen, PhD, a virologist affiliated with Georgetown University in Washington, told The New York Times.
Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, recently said that getting children vaccinated is an important step toward achieving herd immunity.
“We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape,” he said earlier in March during a hearing of the Senate Health, Education, Labor, and Pensions Committee. “We ultimately would like to get and have to get children into that mix.”
Pfizer said it started clinical trials during the week of March 23 with children aged 5-11 and will next start trials with children aged 2-5, followed by children aged 6 months to 2 years. Vaccine makers Moderna and AstraZeneca also have started clinical trials in younger children.
A version of this article first appeared on WebMD.com.
COVID vaccines could lose their punch within a year, experts say
In a survey of 77 epidemiologists from 28 countries by the People’s Vaccine Alliance, 66.2% predicted that the world has a year or less before variants make current vaccines ineffective. The People’s Vaccine Alliance is a coalition of more than 50 organizations, including the African Alliance, Oxfam, Public Citizen, and UNAIDS (the Joint United Nations Programme on HIV/AIDS).
Almost a third (32.5%) of those surveyed said ineffectiveness would happen in 9 months or less; 18.2% said 6 months or less.
Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, said in an interview that, while it’s hard to say whether vaccines could become ineffective in that time frame, “It’s perfectly reasonable to think it could happen.”
The good news, said Dr. Offit, who was not involved with the survey, is that SARS-CoV-2 mutates slowly, compared with other viruses such as influenza.
“To date,” he said, “the mutations that have occurred are not far enough away from the immunity induced by your natural infection or immunization such that one isn’t protected at least against severe and critical disease.”
That’s the goal of vaccines, he noted: “to keep people from suffering mightily.”
A line may be crossed
“And so far that’s happening, even with the variants,” Dr. Offit said. “That line has not been crossed. But I think we should assume that it might be.”
Dr. Offit said it will be critical to monitor anyone who gets hospitalized who is known to have been infected or fully vaccinated. Then countries need to get really good at sequencing those viruses.
The great majority of those surveyed (88%) said that persistently low vaccine coverage in many countries would make it more likely that vaccine-resistant mutations will appear.
Coverage comparisons between countries are stark.
Many countries haven’t given a single vaccine dose
While rich countries are giving COVID-19 vaccinations at the rate of a person a second, many of the poorest countries have given hardly any vaccines, the People’s Vaccine Alliance says.
Additionally, according to researchers at the Global Health Innovation Center at Duke University, Durham, N.C., high- and upper-middle–income countries, which represent one-fifth of the world’s population, have bought about 6 billion doses. But low- and lower-middle–income countries, which make up four-fifths of the population, have bought only about 2.6 billion, an article in Nature reports.
“You’re only as strong as your weakest country,” Dr. Offit said. “If we haven’t learned that what happens in other countries can [affect the global population], we haven’t been paying attention.”
Gregg Gonsalves, PhD, associate professor of epidemiology at Yale University, New Haven, Conn., one of the academic centers surveyed, didn’t specify a timeline for when vaccines would become ineffective, but said in a press release that the urgency for widespread global vaccination is real.
“Unless we vaccinate the world,” he said, “we leave the playing field open to more and more mutations, which could churn out variants that could evade our current vaccines and require booster shots to deal with them.”
“Dire, but not surprising”
Panagis Galiatsatos, MD, MHS, a pulmonologist at John Hopkins University, Baltimore, whose research focuses on health care disparities, said the survey findings were “dire, but not surprising.”
Johns Hopkins was another of the centers surveyed, but Dr. Galiatsatos wasn’t personally involved with the survey.
COVID-19, Dr. Galiatsatos pointed out, has laid bare disparities, both in who gets the vaccine and who’s involved in trials to develop the vaccines.
“It’s morally concerning and an ethical reckoning,” he said in an interview.
Recognition of the borderless swath of destruction the virus is exacting is critical, he said.
The United States “has to realize this can’t be a U.S.-centric issue,” he said. “We’re going to be back to the beginning if we don’t make sure that every country is doing well. We haven’t seen that level of uniform approach.”
He noted that scientists have always known that viruses mutate, but now the race is on to find the parts of SARS-CoV-2 that don’t mutate as much.
“My suspicion is we’ll probably need boosters instead of a whole different vaccine,” Dr. Galiatsatos said.
Among the strategies sought by the People’s Vaccine Alliance is for all pharmaceutical companies working on COVID-19 vaccines to openly share technology and intellectual property through the World Health Organization COVID-19 Technology Access Pool, to speed production and rollout of vaccines to all countries.
In the survey, 74% said that open sharing of technology and intellectual property could boost global vaccine coverage; 23% said maybe and 3% said it wouldn’t help.
The survey was carried out between Feb. 17 and March 25, 2021. Respondents included epidemiologists, virologists, and infection disease specialists from the following countries: Algeria, Argentina, Australia, Belgium, Bolivia, Canada, Denmark, Ethiopia, France, Guatemala, India, Italy, Kenya, Lebanon, Norway, Philippines, Senegal, Somalia, South Africa, South Sudan, Spain, United Arab Emirates, Uganda, United Kingdom, United States, Vietnam, Zambia, and Zimbabwe.
Dr. Offit and Dr. Galiatsatos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a survey of 77 epidemiologists from 28 countries by the People’s Vaccine Alliance, 66.2% predicted that the world has a year or less before variants make current vaccines ineffective. The People’s Vaccine Alliance is a coalition of more than 50 organizations, including the African Alliance, Oxfam, Public Citizen, and UNAIDS (the Joint United Nations Programme on HIV/AIDS).
Almost a third (32.5%) of those surveyed said ineffectiveness would happen in 9 months or less; 18.2% said 6 months or less.
Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, said in an interview that, while it’s hard to say whether vaccines could become ineffective in that time frame, “It’s perfectly reasonable to think it could happen.”
The good news, said Dr. Offit, who was not involved with the survey, is that SARS-CoV-2 mutates slowly, compared with other viruses such as influenza.
“To date,” he said, “the mutations that have occurred are not far enough away from the immunity induced by your natural infection or immunization such that one isn’t protected at least against severe and critical disease.”
That’s the goal of vaccines, he noted: “to keep people from suffering mightily.”
A line may be crossed
“And so far that’s happening, even with the variants,” Dr. Offit said. “That line has not been crossed. But I think we should assume that it might be.”
Dr. Offit said it will be critical to monitor anyone who gets hospitalized who is known to have been infected or fully vaccinated. Then countries need to get really good at sequencing those viruses.
The great majority of those surveyed (88%) said that persistently low vaccine coverage in many countries would make it more likely that vaccine-resistant mutations will appear.
Coverage comparisons between countries are stark.
Many countries haven’t given a single vaccine dose
While rich countries are giving COVID-19 vaccinations at the rate of a person a second, many of the poorest countries have given hardly any vaccines, the People’s Vaccine Alliance says.
Additionally, according to researchers at the Global Health Innovation Center at Duke University, Durham, N.C., high- and upper-middle–income countries, which represent one-fifth of the world’s population, have bought about 6 billion doses. But low- and lower-middle–income countries, which make up four-fifths of the population, have bought only about 2.6 billion, an article in Nature reports.
“You’re only as strong as your weakest country,” Dr. Offit said. “If we haven’t learned that what happens in other countries can [affect the global population], we haven’t been paying attention.”
Gregg Gonsalves, PhD, associate professor of epidemiology at Yale University, New Haven, Conn., one of the academic centers surveyed, didn’t specify a timeline for when vaccines would become ineffective, but said in a press release that the urgency for widespread global vaccination is real.
“Unless we vaccinate the world,” he said, “we leave the playing field open to more and more mutations, which could churn out variants that could evade our current vaccines and require booster shots to deal with them.”
“Dire, but not surprising”
Panagis Galiatsatos, MD, MHS, a pulmonologist at John Hopkins University, Baltimore, whose research focuses on health care disparities, said the survey findings were “dire, but not surprising.”
Johns Hopkins was another of the centers surveyed, but Dr. Galiatsatos wasn’t personally involved with the survey.
COVID-19, Dr. Galiatsatos pointed out, has laid bare disparities, both in who gets the vaccine and who’s involved in trials to develop the vaccines.
“It’s morally concerning and an ethical reckoning,” he said in an interview.
Recognition of the borderless swath of destruction the virus is exacting is critical, he said.
The United States “has to realize this can’t be a U.S.-centric issue,” he said. “We’re going to be back to the beginning if we don’t make sure that every country is doing well. We haven’t seen that level of uniform approach.”
He noted that scientists have always known that viruses mutate, but now the race is on to find the parts of SARS-CoV-2 that don’t mutate as much.
“My suspicion is we’ll probably need boosters instead of a whole different vaccine,” Dr. Galiatsatos said.
Among the strategies sought by the People’s Vaccine Alliance is for all pharmaceutical companies working on COVID-19 vaccines to openly share technology and intellectual property through the World Health Organization COVID-19 Technology Access Pool, to speed production and rollout of vaccines to all countries.
In the survey, 74% said that open sharing of technology and intellectual property could boost global vaccine coverage; 23% said maybe and 3% said it wouldn’t help.
The survey was carried out between Feb. 17 and March 25, 2021. Respondents included epidemiologists, virologists, and infection disease specialists from the following countries: Algeria, Argentina, Australia, Belgium, Bolivia, Canada, Denmark, Ethiopia, France, Guatemala, India, Italy, Kenya, Lebanon, Norway, Philippines, Senegal, Somalia, South Africa, South Sudan, Spain, United Arab Emirates, Uganda, United Kingdom, United States, Vietnam, Zambia, and Zimbabwe.
Dr. Offit and Dr. Galiatsatos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a survey of 77 epidemiologists from 28 countries by the People’s Vaccine Alliance, 66.2% predicted that the world has a year or less before variants make current vaccines ineffective. The People’s Vaccine Alliance is a coalition of more than 50 organizations, including the African Alliance, Oxfam, Public Citizen, and UNAIDS (the Joint United Nations Programme on HIV/AIDS).
Almost a third (32.5%) of those surveyed said ineffectiveness would happen in 9 months or less; 18.2% said 6 months or less.
Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia, said in an interview that, while it’s hard to say whether vaccines could become ineffective in that time frame, “It’s perfectly reasonable to think it could happen.”
The good news, said Dr. Offit, who was not involved with the survey, is that SARS-CoV-2 mutates slowly, compared with other viruses such as influenza.
“To date,” he said, “the mutations that have occurred are not far enough away from the immunity induced by your natural infection or immunization such that one isn’t protected at least against severe and critical disease.”
That’s the goal of vaccines, he noted: “to keep people from suffering mightily.”
A line may be crossed
“And so far that’s happening, even with the variants,” Dr. Offit said. “That line has not been crossed. But I think we should assume that it might be.”
Dr. Offit said it will be critical to monitor anyone who gets hospitalized who is known to have been infected or fully vaccinated. Then countries need to get really good at sequencing those viruses.
The great majority of those surveyed (88%) said that persistently low vaccine coverage in many countries would make it more likely that vaccine-resistant mutations will appear.
Coverage comparisons between countries are stark.
Many countries haven’t given a single vaccine dose
While rich countries are giving COVID-19 vaccinations at the rate of a person a second, many of the poorest countries have given hardly any vaccines, the People’s Vaccine Alliance says.
Additionally, according to researchers at the Global Health Innovation Center at Duke University, Durham, N.C., high- and upper-middle–income countries, which represent one-fifth of the world’s population, have bought about 6 billion doses. But low- and lower-middle–income countries, which make up four-fifths of the population, have bought only about 2.6 billion, an article in Nature reports.
“You’re only as strong as your weakest country,” Dr. Offit said. “If we haven’t learned that what happens in other countries can [affect the global population], we haven’t been paying attention.”
Gregg Gonsalves, PhD, associate professor of epidemiology at Yale University, New Haven, Conn., one of the academic centers surveyed, didn’t specify a timeline for when vaccines would become ineffective, but said in a press release that the urgency for widespread global vaccination is real.
“Unless we vaccinate the world,” he said, “we leave the playing field open to more and more mutations, which could churn out variants that could evade our current vaccines and require booster shots to deal with them.”
“Dire, but not surprising”
Panagis Galiatsatos, MD, MHS, a pulmonologist at John Hopkins University, Baltimore, whose research focuses on health care disparities, said the survey findings were “dire, but not surprising.”
Johns Hopkins was another of the centers surveyed, but Dr. Galiatsatos wasn’t personally involved with the survey.
COVID-19, Dr. Galiatsatos pointed out, has laid bare disparities, both in who gets the vaccine and who’s involved in trials to develop the vaccines.
“It’s morally concerning and an ethical reckoning,” he said in an interview.
Recognition of the borderless swath of destruction the virus is exacting is critical, he said.
The United States “has to realize this can’t be a U.S.-centric issue,” he said. “We’re going to be back to the beginning if we don’t make sure that every country is doing well. We haven’t seen that level of uniform approach.”
He noted that scientists have always known that viruses mutate, but now the race is on to find the parts of SARS-CoV-2 that don’t mutate as much.
“My suspicion is we’ll probably need boosters instead of a whole different vaccine,” Dr. Galiatsatos said.
Among the strategies sought by the People’s Vaccine Alliance is for all pharmaceutical companies working on COVID-19 vaccines to openly share technology and intellectual property through the World Health Organization COVID-19 Technology Access Pool, to speed production and rollout of vaccines to all countries.
In the survey, 74% said that open sharing of technology and intellectual property could boost global vaccine coverage; 23% said maybe and 3% said it wouldn’t help.
The survey was carried out between Feb. 17 and March 25, 2021. Respondents included epidemiologists, virologists, and infection disease specialists from the following countries: Algeria, Argentina, Australia, Belgium, Bolivia, Canada, Denmark, Ethiopia, France, Guatemala, India, Italy, Kenya, Lebanon, Norway, Philippines, Senegal, Somalia, South Africa, South Sudan, Spain, United Arab Emirates, Uganda, United Kingdom, United States, Vietnam, Zambia, and Zimbabwe.
Dr. Offit and Dr. Galiatsatos reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
National Psoriasis Foundation recommends some stop methotrexate for 2 weeks after J&J vaccine
The , Joel M. Gelfand, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
The new guidance states: “Patients 60 or older who have at least one comorbidity associated with an increased risk for poor COVID-19 outcomes, and who are taking methotrexate with well-controlled psoriatic disease, may, in consultation with their prescriber, consider holding it for 2 weeks after receiving the Ad26.COV2.S [Johnson & Johnson] vaccine in order to potentially improve vaccine response.”
The key word here is “potentially.” There is no hard evidence that a 2-week hold on methotrexate after receiving the killed adenovirus vaccine will actually provide a clinically meaningful benefit. But it’s a hypothetical possibility. The rationale stems from a small randomized trial conducted in South Korea several years ago in which patients with rheumatoid arthritis were assigned to hold or continue their methotrexate for the first 2 weeks after receiving an inactivated-virus influenza vaccine. The antibody response to the vaccine was better in those who temporarily halted their methotrexate, explained Dr. Gelfand, cochair of the NPF COVID-19 Task Force and professor of dermatology and of epidemiology at the University of Pennsylvania, Philadelphia.
“If you have a patient on methotrexate who’s 60 or older and whose psoriasis is completely controlled and quiescent and the patient is concerned about how well the vaccine is going to work, this is a reasonable thing to consider in someone who’s at higher risk for poor outcomes if they get infected,” he said.
If the informed patient wants to continue on methotrexate without interruption, that’s fine, too, in light of the lack of compelling evidence on this issue, the dermatologist added at the conference, sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The NPF task force does not extend the recommendation to consider holding methotrexate in recipients of the mRNA-based Moderna and Pfizer vaccines because of their very different mechanisms of action. Nor is it recommended to hold biologic agents after receiving any of the available COVID-19 vaccines. Studies have shown no altered immunologic response to influenza or pneumococcal vaccines in patients who continued on tumor necrosis factor inhibitors or interleukin-17 inhibitors. The interleukin-23 inhibitors haven’t been studied in this regard.
The task force recommends that most psoriasis patients should continue on treatment throughout the pandemic, and newly diagnosed patients should commence appropriate therapy as if there was no pandemic.
“We’ve learned that many patients who stopped their treatment for psoriatic disease early in the pandemic came to regret that decision because their psoriasis flared and got worse and required reinstitution of therapy,” Dr. Gelfand said. “The current data is largely reassuring that if there is an effect of our therapies on the risk of COVID, it must be rather small and therefore unlikely to be clinically meaningful for our patients.”
Dr. Gelfand reported serving as a consultant to and recipient of institutional research grants from Pfizer and numerous other pharmaceutical companies.
MedscapeLIVE and this news organization are owned by the same parent company.
The , Joel M. Gelfand, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
The new guidance states: “Patients 60 or older who have at least one comorbidity associated with an increased risk for poor COVID-19 outcomes, and who are taking methotrexate with well-controlled psoriatic disease, may, in consultation with their prescriber, consider holding it for 2 weeks after receiving the Ad26.COV2.S [Johnson & Johnson] vaccine in order to potentially improve vaccine response.”
The key word here is “potentially.” There is no hard evidence that a 2-week hold on methotrexate after receiving the killed adenovirus vaccine will actually provide a clinically meaningful benefit. But it’s a hypothetical possibility. The rationale stems from a small randomized trial conducted in South Korea several years ago in which patients with rheumatoid arthritis were assigned to hold or continue their methotrexate for the first 2 weeks after receiving an inactivated-virus influenza vaccine. The antibody response to the vaccine was better in those who temporarily halted their methotrexate, explained Dr. Gelfand, cochair of the NPF COVID-19 Task Force and professor of dermatology and of epidemiology at the University of Pennsylvania, Philadelphia.
“If you have a patient on methotrexate who’s 60 or older and whose psoriasis is completely controlled and quiescent and the patient is concerned about how well the vaccine is going to work, this is a reasonable thing to consider in someone who’s at higher risk for poor outcomes if they get infected,” he said.
If the informed patient wants to continue on methotrexate without interruption, that’s fine, too, in light of the lack of compelling evidence on this issue, the dermatologist added at the conference, sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The NPF task force does not extend the recommendation to consider holding methotrexate in recipients of the mRNA-based Moderna and Pfizer vaccines because of their very different mechanisms of action. Nor is it recommended to hold biologic agents after receiving any of the available COVID-19 vaccines. Studies have shown no altered immunologic response to influenza or pneumococcal vaccines in patients who continued on tumor necrosis factor inhibitors or interleukin-17 inhibitors. The interleukin-23 inhibitors haven’t been studied in this regard.
The task force recommends that most psoriasis patients should continue on treatment throughout the pandemic, and newly diagnosed patients should commence appropriate therapy as if there was no pandemic.
“We’ve learned that many patients who stopped their treatment for psoriatic disease early in the pandemic came to regret that decision because their psoriasis flared and got worse and required reinstitution of therapy,” Dr. Gelfand said. “The current data is largely reassuring that if there is an effect of our therapies on the risk of COVID, it must be rather small and therefore unlikely to be clinically meaningful for our patients.”
Dr. Gelfand reported serving as a consultant to and recipient of institutional research grants from Pfizer and numerous other pharmaceutical companies.
MedscapeLIVE and this news organization are owned by the same parent company.
The , Joel M. Gelfand, MD, said at Innovations in Dermatology: Virtual Spring Conference 2021.
The new guidance states: “Patients 60 or older who have at least one comorbidity associated with an increased risk for poor COVID-19 outcomes, and who are taking methotrexate with well-controlled psoriatic disease, may, in consultation with their prescriber, consider holding it for 2 weeks after receiving the Ad26.COV2.S [Johnson & Johnson] vaccine in order to potentially improve vaccine response.”
The key word here is “potentially.” There is no hard evidence that a 2-week hold on methotrexate after receiving the killed adenovirus vaccine will actually provide a clinically meaningful benefit. But it’s a hypothetical possibility. The rationale stems from a small randomized trial conducted in South Korea several years ago in which patients with rheumatoid arthritis were assigned to hold or continue their methotrexate for the first 2 weeks after receiving an inactivated-virus influenza vaccine. The antibody response to the vaccine was better in those who temporarily halted their methotrexate, explained Dr. Gelfand, cochair of the NPF COVID-19 Task Force and professor of dermatology and of epidemiology at the University of Pennsylvania, Philadelphia.
“If you have a patient on methotrexate who’s 60 or older and whose psoriasis is completely controlled and quiescent and the patient is concerned about how well the vaccine is going to work, this is a reasonable thing to consider in someone who’s at higher risk for poor outcomes if they get infected,” he said.
If the informed patient wants to continue on methotrexate without interruption, that’s fine, too, in light of the lack of compelling evidence on this issue, the dermatologist added at the conference, sponsored by MedscapeLIVE! and the producers of the Hawaii Dermatology Seminar and Caribbean Dermatology Symposium.
The NPF task force does not extend the recommendation to consider holding methotrexate in recipients of the mRNA-based Moderna and Pfizer vaccines because of their very different mechanisms of action. Nor is it recommended to hold biologic agents after receiving any of the available COVID-19 vaccines. Studies have shown no altered immunologic response to influenza or pneumococcal vaccines in patients who continued on tumor necrosis factor inhibitors or interleukin-17 inhibitors. The interleukin-23 inhibitors haven’t been studied in this regard.
The task force recommends that most psoriasis patients should continue on treatment throughout the pandemic, and newly diagnosed patients should commence appropriate therapy as if there was no pandemic.
“We’ve learned that many patients who stopped their treatment for psoriatic disease early in the pandemic came to regret that decision because their psoriasis flared and got worse and required reinstitution of therapy,” Dr. Gelfand said. “The current data is largely reassuring that if there is an effect of our therapies on the risk of COVID, it must be rather small and therefore unlikely to be clinically meaningful for our patients.”
Dr. Gelfand reported serving as a consultant to and recipient of institutional research grants from Pfizer and numerous other pharmaceutical companies.
MedscapeLIVE and this news organization are owned by the same parent company.
FROM INNOVATIONS IN DERMATOLOGY